Abstract
Simple Summary
Tumor cells are highly resistant to oxidative stress, but beyond a certain threshold, it may lead to apoptosis/necrosis. Thus, induced loss of redox balance can be a strategy used in anticancer therapies. However, the effectiveness of drugs contrasts with unknown mechanisms involved in the loss of fertility. Considering that cancer patients’ life expectancy is increasing, it raises concerns about the unknown adverse effects. Therefore, new strategies should be pursued alongside explaining to the patients their options regarding the reproduction side effects.
Abstract
Tumor cells are highly resistant to oxidative stress resulting from the imbalance between high reactive oxygen species (ROS) production and insufficient antioxidant defenses. However, when intracellular levels of ROS rise beyond a certain threshold, largely above cancer cells’ capacity to reduce it, they may ultimately lead to apoptosis or necrosis. This is, in fact, one of the molecular mechanisms of anticancer drugs, as most chemotherapeutic treatments alter redox homeostasis by further elevation of intracellular ROS levels or inhibition of antioxidant pathways. In traditional chemotherapy, it is widely accepted that most therapeutic effects are due to ROS-mediated cell damage, but in targeted therapies, ROS-mediated effects are mostly unknown and data are still emerging. The increasing effectiveness of anticancer treatments has raised new challenges, especially in the field of reproduction. With cancer patients’ life expectancy increasing, many aiming to become parents will be confronted with the adverse effects of treatments. Consequently, concerns about the impact of anticancer therapies on reproductive capacity are of particular interest. In this review, we begin with a short introduction on anticancer therapies, then address ROS physiological/pathophysiological roles in both male and female reproductive systems, and finish with ROS-mediated adverse effects of anticancer treatments in reproduction.
Keywords: reactive oxygen species (ROS), oxidative stress, chemotherapy, targeted agents, (in)fertility
1. Introduction
Surgery and radiotherapy are commonly used in patients with local and non-metastatic cancer, as they are still the most effective. However, they are very inefficient when cancer is spread throughout the body. In this case, chemotherapeutic drugs or biological agents are among the first-line choice therapies, since they are capable of reaching all organs of the body, through the bloodstream [1].
Chemotherapeutic drugs act primarily by inhibiting the high proliferation rate of cancer cells. One of the molecular mechanisms by which these drugs act is by increasing intracellular levels of reactive oxygen species (ROS) [2] largely beyond cancer cells’ capacity to reduce it, leading to apoptosis or tissue necrosis [3], this has also been observed in the treatment of reproductive cancer [4,5,6]. Treatment-associated excess ROS production in healthy tissues can be the root of cell toxicity observed during chemotherapy treatment, such as anthracycline-mediated cardiotoxicity, and nephrotoxicity triggered by platinum-based compounds [7,8]. Additionally, undesirable side effects on fast renewing cells of the body, such as hair follicles, bone marrow, and gastrointestinal tract cells [1] are also frequently observed.
Biological agents, on the other hand, are more selective molecules that block specific transduction pathways or proteins that are overexpressed/mutated in cancer. The use of biologicals minimizes loss of viability of normal cells and avoids undesirable chemotherapy-associated side effects [1,9].
Although targeted therapies were promised to be magic bullets with single targets, the accumulated information obtained through their clinical use has shown side effects of such therapies [10,11,12] and wider mechanisms of action, including ROS-mediated ones [13,14,15].
The increasing effectiveness of anticancer treatments has raised new challenges, especially in the field of reproduction. As the life expectancy of cancer patients has been highly increased, patients who suffered from cancer in their youth will aim to become parents in adulthood and many will be confronted with fertility issues related to side effects of anticancer therapies. In parallel, patients that are living with cancer as a chronic illness through effective therapy may also be faced with this same issue. Therefore, concerns regarding the adverse effects of anticancer therapies on fertility have increased and are of particular interest, namely those impairing the adequate state of gonads and normal sexual functions. As male and female reproductive system(s) and associated gametogenesis, spermatogenesis, and oogenesis, respectively, show a wide range of differences, it is expected to find specific gonadal toxicity for each gender [16,17,18,19,20]. Male fertility dysfunction associated with anticancer therapies can occur by direct gonadotoxic effects and/or disruption of the hypothalamic–pituitary axis, the latest resulting in impaired libido, erection, sperm production, and ejaculation [21]. In females, anticancer drugs can have a direct toxic effect in the ovary and the uterus and cause dysregulation of the hypothalamic-pituitary axis. This may lead to a loss of libido, abnormal follicle development, and impairment of ovarian and uterine function [22,23]. Therefore, in individuals of child-bearing ages, it is necessary to inform them about the possible side-effects on fertility and available fertility preservation options. Post-treatment inability or difficulty to achieve pregnancy is a possible outcome. In fact, chemotherapeutic agents induce numeric or structural chromosomal abnormalities in the germline that, consequently, may impair reproduction by interfering with embryonic development, increasing miscarriages, or transmitting genetic anomalies to offspring [24,25,26,27].
Studies have reviewed the cytotoxic effects of chemotherapeutic drugs [16,18,21,23,24,28,29,30,31,32,33,34,35,36,37,38,39,40], and, to a lesser extent, target therapies [18,28,32,33] on fertility. However, the mechanisms by which they impair fertility are still largely unexplored. This knowledge will be important to define strategies to prevent or attenuate the impact of cancer therapies upon fertility. Thereby, in this review, we address the knowledge on the effects of anticancer treatments (traditional chemotherapy and targeted agents) on fertility, with a special focus on ROS physiological and pathological roles.
2. Oxidative Stress and Fertility
2.1. Reactive Oxygen Species and Oxidative Stress
ROS are reactive molecules that contain oxygen atoms and are subdivided into free radicals and non-radical oxidants [41]. ROS reactivity, half-lives, and diffusion capacity are widely variable [42,43,44]. ROS can be generated endogenously by mitochondrial respiratory chain enzymes, nicotinamide adenine dinucleotide phosphate oxidase (NADPH oxidase), microsomal cytochrome P450, or by xanthine oxidase [45]. Beyond endogenous sources, ROS can also have exogenous sources, including ultraviolet radiation, X- and gamma-rays, ultrasound, pesticides, herbicides, and xenobiotics [45].
To cope with the continued production of ROS, cells have developed antioxidant mechanisms that delay or prevent oxidation from happening. In cells, excess ROS are quenched by enzymatic antioxidants such as superoxide dismutase (SOD), catalase (Cat), and glutathione peroxidase (GPx) [46], or nonenzymatic antioxidants such as vitamins A, C, and E, glutathione, and plant polyphenols. Additionally, minerals such as zinc and selenium can have indirect antioxidant effects by acting as cofactors of antioxidant enzymes and other proteins that modulate cellular redox balance [47].
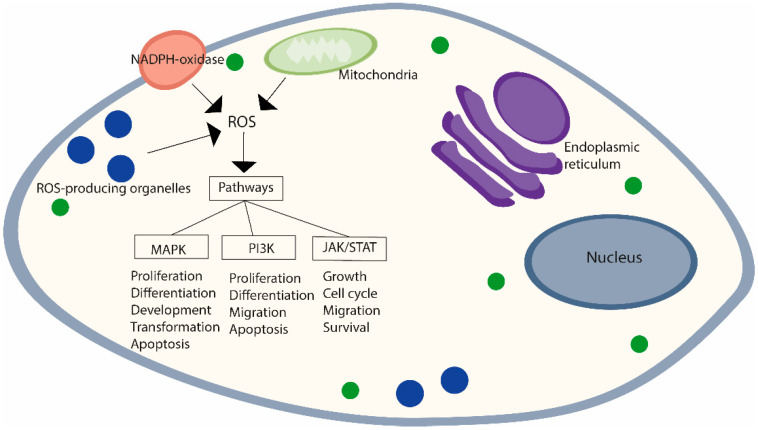
ROS are normal products of cell metabolism with known physiological roles. Even in small amounts, ROS are able to regulate a wide spectrum of signaling pathways, such as the mitogen-activated protein kinase (MAPK), Janus kinase (JAK)/signal transducer and activator of transcription proteins (STATs), and phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt) pathways [48]. They regulate these pathways by modifying the activity of structural proteins, transcription factors, membrane receptors, ion channels, and protein kinases/phosphatases [49]. The MAPK cascade, composed of at least three MAPKs, extracellular signal-regulated kinase (ERK), Janus kinase (JNK/SAPK), and p38 MAPK, plays important roles in cellular processes such as proliferation, differentiation, development, transformation, and apoptosis [50]. The JAK/STAT pathway is used to transduce a multitude of signals. Its activation stimulates cell proliferation, differentiation, migration, and apoptosis [51]. PI3K activation affects several cell events like growth, cell cycle entry, migration, and survival [52] (Figure 1).
Figure 1.
ROS-mediated activation of cell signaling pathways. Major sites of reactive oxygen species (ROS) production in cells, enzymes responsible for ROS production at each of the cellular compartments, and principal signaling pathways activated.
Despite ROS having a physiological function, when redox homeostasis is disturbed, due to an imbalance between their production and neutralization, a new state referred to as oxidative stress (OS) may arise. Cells have a graded response to OS. Minor or moderated changes allow cells to adapt and restore redox homeostasis while violent perturbations impair redox signaling, promote biomolecule modifications, and may even induce cell death [53,54].
2.2. Reactive Oxygen Species, Oxidative Stress, and Male (In)Fertility
Spermatogenesis lasts about 70 days, the time needed for the germ stem cell (spermatogonial stem cells) to give rise to the spermatozoon (about 70 million sperm daily), going through mitotic and meiotic divisions [55]. Spermatogonia, the precursors of all germ cell types, are located or “resting” on the basal lamina (a modified form of extracellular matrix (ECM) constituted by collagen and myoid cell layers). Basal lamina plays a significant role in spermatogenesis. It behaves as a physical support to the seminiferous epithelium, provides selected access of molecules to the seminiferous epithelium, and enables the crosstalk between the seminiferous epithelium, myoid cells, and interstitial cells, such as Leydig cells [56]. Spermatogonia present a continuous self-renewing capacity and are responsible for making a balance between germ and Sertoli cells [57].
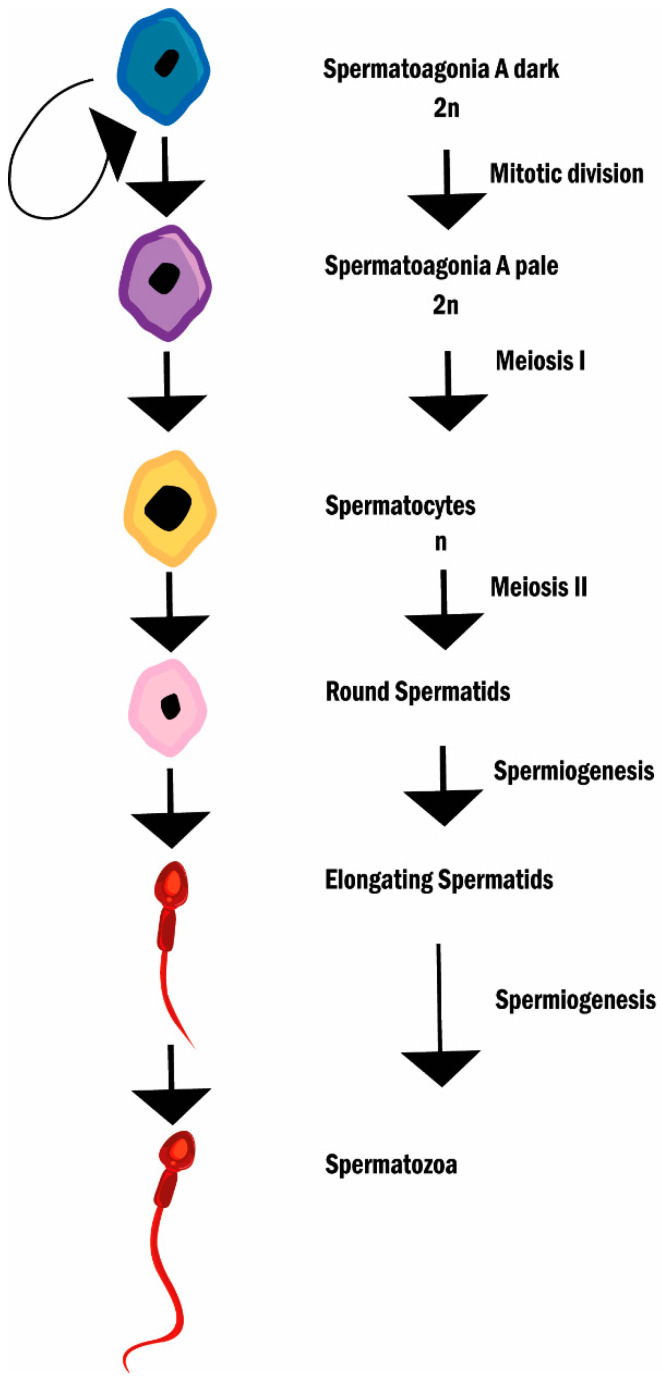
Spermatogonia A dark (SGAd) divides into spermatogonia A pale (SGAp) and SGAd thus, maintaining the stem cell pool. SGAp then divides to give origin to spermatogonia B and these to pre-leptotene spermatocytes. Pre-leptotene spermatocytes (primary spermatocytes) now enter meiosis and progressively turn into zygotene spermatocytes, and then into pachytene spermatocytes [58]. At this stage, the cell attains the biggest volume and starts crossing over (chromosome recombination). Primary spermatocytes then complete the first meiotic division with separation of chromosomes (not of chromatids as in mitosis) and give rise to secondary spermatocytes, that go through the second division (separation of chromatids) and originate haploid round spermatids. Spermatids progressively differentiate into spermatozoa [59], the nucleus condenses and elongates, and a flagellum is formed (spermiogenesis). The condensation of the nucleus is due to the substitution of histones by protamines, a basic protein that establishes bisulfite links enabling the compaction of DNA in order to protect against mechanical and oxidative stresses that sperm will encounter during the journey until the Fallopian tube where fertilization occurs [60]. From the Golgi, a series of vesicles fuse and give rise to an acrosomal vesicle that surrounds the anterior 2/3 of the nucleus. This vesicle contains the enzymes necessary to aid in the penetration of the zona pellucida [61]. When these steps finalize, the sperm retracts from Sertoli cell junctions (spermiation) and travels through the tubules of the rete testis to the epididymis where it matures and remains stored up to ejaculation [62] (Figure 2).
Figure 2.
Spermatogenesis and spermiogenesis. The diagram describes the different stages of spermatogenesis and spermiogenesis.
During human spermatozoa maturation, differentiated levels of ROS can be produced by plasmatic membrane NADPH oxidase and by mitochondrial nicotinamide adenine dinucleotide-dependent oxidoreductase [63,64], being highest in immature spermatozoa with abnormal head morphology and cytoplasmic retention and lowest in mature spermatozoa and immature germ cells [65,66]. Moreover, spermatogenesis appears to be paralleled by a differential expression of stress response genes, as suggested by an increase in the antioxidant enzyme Cu-Zn SOD [67]. This points to changes in susceptibility to OS through spermatogenesis, which can be rooted in distinct reasons. Late stages of spermatids and spermatozoa are vulnerable to the deleterious effects of ROS due to high levels of polyunsaturated fatty acids in the plasma membrane (essential for membrane fluidity), whereas differentiating spermatogonia and spermatocytes appear susceptible due to their high mitotic and meiotic activity, respectively [68,69]. ROS have a pivotal role in spermatogenesis, not only in the earliest stages of development, being involved in sperm chromatin condensation, in inducing apoptosis to adjust the number of germ cells or spermatogonia proliferation, but also in sperm maturation, participating in capacitation, acrosome reaction, mitochondrial sheath stability, and sperm motility [70,71,72,73].
ROS are also implicated in sperm-oocyte interaction [74] and participate in the activation of the steroidogenic pathway by inducing Ras and ERK1/2 activation in Leydig cells [75]. In the seminal fluid, leukocytes produce high levels of ROS (up to 1000 times more than spermatozoa) that play an important role in the cellular defense mechanism against infections and inflammation [76].
The pathophysiological role of ROS in spermatogenesis has also been studied. Intrinsic ROS overproduction depletes sperm antioxidant systems, leading to OS [77,78]. OS impinges on molecular components, inducing oxidation in lipids, proteins, carbohydrates, and DNA. Oxidative modifications to proteins alter their structure and function, with repercussions both on spermatogenesis and fertility [79]. Peroxidative damage to membrane lipids leads to membrane structure and fluidity instability and membrane-associated processes dysregulation [79,80]. Even sperm motility is affected by lipid peroxidation to mitochondrial membranes, leading to a decrease in mitochondrial membrane potential and defects in the sperm mid-piece and axonemal region [81]. OS also has deleterious effects on the spermatic nucleus, impinging on DNA integrity, increasing the rates of sperm DNA fragmentation [73,74,82]. Since spermatozoa lack DNA repair mechanisms, in case of excessive DNA damage apoptotic cascades are activated, leading to reduced sperm concentrations and consequently male infertility [83]. Recently, it has been observed that about 30–80% of infertile men have abnormal semen characteristics with elevated seminal ROS levels [84].
2.3. Reactive Oxygen Species, Oxidative Stress, and Female (In)Fertility
During early embryo development, primordial germ cells migrate to the developing gonads, undergo mitotic divisions (before entering meiosis), colonizing it. Primordial germ cells become oogonia and rearrange themselves in structures described as germ-cell nests. After entering meiosis, individual oocytes are encapsulated by a single layer of squamous pre-granulosa cells, forming primordial follicles [85]. After a programmed cell nest breakdown, only around 30% of the initial oogonia survive and become the pool of primordial follicles. Oocytes remain arrested in the first meiotic division until latter activation to proceed development [86]. Early activation, during childhood and until puberty, results in atresia (degeneration of follicle into scar tissue). For this reason, in puberty, ~300,000 primordial follicles remain [87,88]. Of this pool, as little as 400 follicles will complete development and ovulate; all the others will suffer atresia [89,90].
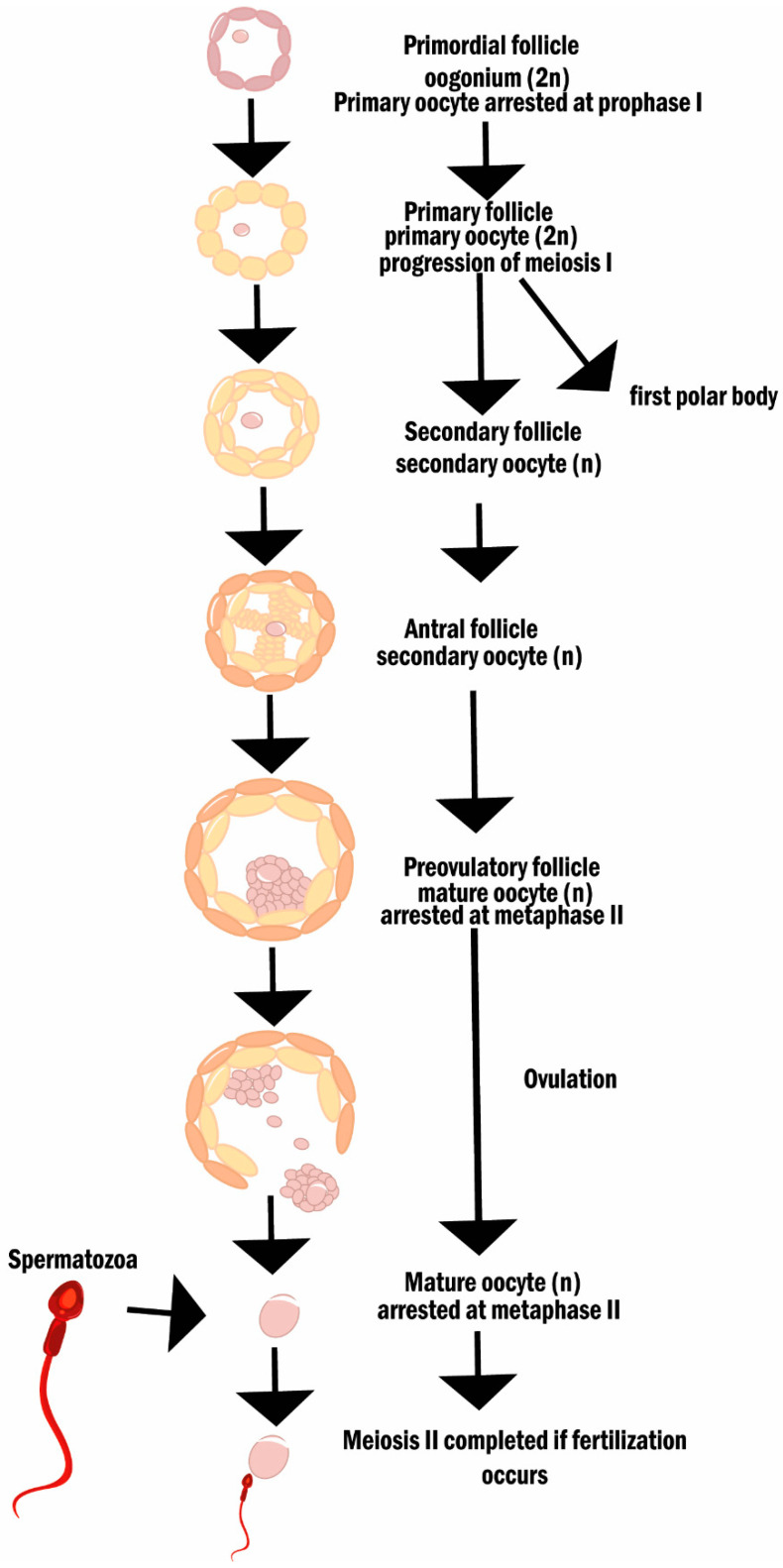
From puberty, and until menopause, hormones produced by the hypothalamus, pituitary gland, and the ovaries are the messengers responsible for the ovarian cycle, which can be divided into two phases: follicular and luteal. The follicular phase concerns the development of follicles until ovulation. In short, once a selected batch of primordial follicles is activated, their granulosa cells change shape and give rise to primary follicles. These follicles express cell proliferation markers which will allow their growth and ultimately the formation of a multi-layered follicle—the secondary follicle. The antral stage follows and is achieved by the formation of a cavity (the antrum) filled with fluid. In humans, only one dominant follicle will develop, until ovulation. The ovulated oocyte, arrested at metaphase I, will complete meiosis only if fertilization occurs (Figure 3).
Figure 3.
Oogenesis and folliculogenesis. The diagram describes the different stages of oogenesis and folliculogenesis.
The luteal phase starts after ovulation, with the formation of the corpus luteum, and is characterized by changes in hormone levels (an increase in progesterone and in estrogen and a decrease in follicle-stimulating hormone (FSH) and in luteinizing hormone). These hormonal fluctuations will regulate uterine transformations to enable implantation [91]. Upon fertilization, the zygote moves through the fallopian tube until reaching the uterine endometrium, where implantation may take place [92]. If a pregnancy does not occur, hormone production by corpus luteum declines, causing endometrial shedding, and marking the end of the luteal phase [91,93,94,95]. For a more detailed description see [91].
In the ovaries, ROS can be generated by macrophages, steroidogenic cells, and endothelial cells, modulating follicular fluid microenvironment and consequently oocyte development [96]. ROS are involved in the loss of sensitivity of granulosa cells to gonadotropins and steroidogenic function, thus influencing follicular atresia and having a role in the selection of the dominant follicle [97,98]. In the pre-ovulatory follicles, steroid production increases cytochrome P450 activity and consequently the levels of ROS, which are important inducers of ovulation. In fact, decreased ROS production impairs ovulation [99,100]. During oocyte maturation, the expression of enzymatic antioxidants such as Cu-ZnSOD and MnSOD revealed that oocytes are exposed to high levels of ROS and that the balance between ROS and antioxidant enzymes is an important modulator of this process [101]. Although ROS have important physiological roles, the cyclic production of these damaging agents over time and a reduction in ovarian antioxidant capacity may be the root of local inflammation and fibrosis and contribute to tissue dysfunction and the loss of fertility [102,103,104].
In the uterus, ROS have also been implicated in the regulation of the endometrial cycle alongside variations in the expression of SOD, GPx, and lipid peroxides (in response to sex hormones) [105,106,107]. NADPH-oxidase-derived O2 has been shown to activate the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB) signaling cascade promoting prostaglandin secretion, vasoconstriction, and ultimately endometrial shedding [108,109]. Thus, ROS have a determinant role in the regulation of angiogenesis and the endometrial cycle [110,111]. NF-kB exacerbated activation, due to increased uterine levels of ROS, may result in signaling pathways disruption and, consequently, in a broad spectrum of uterine-related infertility disorders (e.g., endometriosis) [112,113].
ROS play a bivalent role (physiological and pathophysiological) not only in the uterus and the ovaries but also in the process of placentation, as previously reviewed [114].
3. Current Evidence of OS-Mediated Effects on Fertility Derived from Cancer Therapies
3.1. Carcinogenesis, Anticancer Therapies and Oxidative Stress
Low-to-moderate ROS levels act as instigators of neoplastic transformation, by promoting genomic DNA mutations and increasing cell proliferation [115,116]. After neoplastic transformation, during hyperproliferation, cancer cells present uncontrolled metabolism and high basal levels of ROS [117]. Their survival under such adverse conditions is achieved due to antioxidant system adaptations [118]. However, if ROS levels increase above a certain threshold (even in neoplastic cells), it will lead to antioxidant system exhaustion and evoke irreversible oxidative damage. The majority of agents used in anticancer therapies aim to induce an accelerated and cumulative oxidative damage, which will surpass the cytotoxic threshold and “selectively” kill cancer cells [119].
There are two major approaches of eliciting intracellular ROS accumulation that are harnessed by anticancer therapies: direct ROS generation or cellular antioxidant system inhibition [120] (Table 1).
Table 1.
Major anticancer compounds and respective ROS-mediated actions.
| Name | Mechanism of Action | Role in Redox System | Ref |
|---|---|---|---|
| Direct ROS generation | |||
| 5-fluorouracil | Thymidylate synthase inhibitor | p53-dependent ROS | [121] |
| Bortezomib | Proteasome inhibitor | ER stress-induced ROS | [122] |
| Cisplatin | nDNA adducts generation | mtDNA and ETC damage | [123] |
| Doxorubicin | nDNA intercalation; topoisomerase-II-mediated nDNA repair disruption | Generation of free radical through iron chelation | [124] |
| Erlotinib | EGFR tyrosine kinase inhibition | Loss of MM potential | [125] |
| Imatinib | Bcr-Abl tyrosine kinase inhibition | Loss of MM potential | [126] |
| Rituximab | Anti-CD20 | Bcl-2 inhibition | [127] |
| Antioxidant process inhibition | |||
| Buthionine sulfoximine | - | GSH synthesis inhibitor | [128] |
| Imexon | Ribonucleotide reductase inhibitor | GSH activity disruption via thiol binding | [129] |
Abelson (Abl); B-cell lymphoma 2 (Bcl-2); breakpoint cluster region protein (Bcr); cluster of differentiate 20 (CD20); electron transport chain (ETC); endoplasmic reticulum (ER); epidermal growth factor receptor (EGFR); glutathione (GSH); mitochondrial DNA (mtDNA); mitochondrial membrane (MM); nuclear DNA (nDNA); reactive oxygen species (ROS).
ROS-promoting agents can: increase the production of O2 by impairing respiratory chain function and causing mitochondrial dysfunction [130] or by activating NADPH-oxidase activity [131]; increase radical intermediates by reacting with flavoprotein reductases (e.g., cytochrome P450 reductase) in the presence of reduced NADPH [132]; lead to hydroxyl radical formation by triggering Fenton-type reactions [133]. Doxorubicin promotes an increase in ROS by intracellular chelation of iron, which may trigger a Fenton-like reaction, generating the high reactive hydroxyl radical, and by interfering with cytochrome P450 forming radical derivatives, which can generate superoxide, in the presence of oxygen [134].
Agents that strategically interfere with ROS metabolism by inducing the depletion of the reduced glutathione (GSH) pool or restricting redox modulating enzymes (e.g., peroxidases and peroxidoxins) have a profound effect on the ability of cells to detoxify ROS. GSH-conjugating compounds, such as Imexon [135] or targeting its synthesis, such as Buthionine sulphoximine, fall into this group [128]. Specific inhibitors of other antioxidant enzymes are being identified, developed, and used in anticancer treatments. ATN-224, an inhibitor of SOD [136,137], and AT, an inhibitor of Cat [138], fall into this category.
Understanding the distinct mechanisms of action of each drug, either ROS-dependent or -independent is vital to attenuate their deleterious side effects. In fact, cotreatments with antioxidants have been used to ameliorate chemotherapy-mediated toxicity (e.g., nephrotoxicity and ototoxicity). Kilic and colleagues have demonstrated that cotreatment with melatonin significantly reduces NF-kB expression and is able to attenuate nephrotoxicity through the activation of nuclear factor-erythroid factor 2-related factor 2 (Nrf-2)/Heme oxygenase-1 pathway [139]. This pathway regulates the expression of several antioxidant genes and protects against OS and inflammation [139,140]. Additionally in the specific context of chemotherapeutical treatments, flavonoids and carotenoids (plant-derived phytochemicals) have been shown to have beneficial properties against ROS-associated secondary effects [141,142,143].
3.2. Treatment-Induced OS and Its Impact on Fertility
Although anticancer therapies aim to specifically disrupt the redox balance of cancer cells, unwanted effects on normal cells also occur. Encephalo-, cardio-, nephron-, oto-, hepato-, myeolo-, myo-, and gastrointestinal toxicities have been described [144,145,146,147,148]. Similarly, distinct gonad toxicity has also been reported [149,150], reflecting the differences between the two gametogenesis processes.
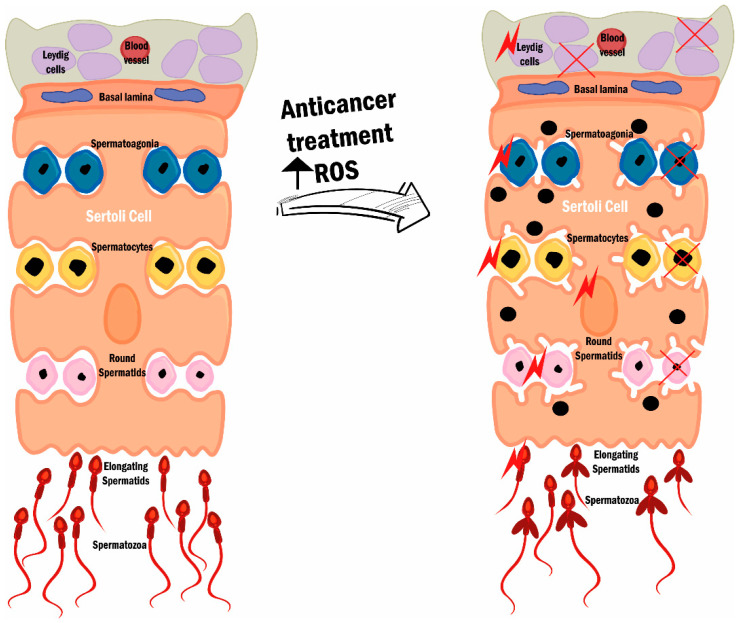
In males, depending on the type of spermatogonia affected, damage can result in transient or persistent oligozoospermia or azoospermia. Spermatogonia type B are more susceptible to cytotoxicity because of their active mitotic proliferation, whereas spermatogonia stem cells (type A) are less susceptible due to their low mitotic activity [16]. In severe gonad toxicity, all spermatogonia are destroyed and azoospermia is established. Additionally, even Sertoli or Leydig cells can be damaged. Leydig cell damage also affects hormone production [151,152,153] (Figure 4).
Figure 4.
Spermatogenesis dysfunction after anticancer treatment. ROS overproduction due to treatments depletes the antioxidant systems, leading to OS. Both the normal and abnormal spermatozoa can be damaged by ROS; however, in the treatment case (right side), damage is more prevalent since ROS are present/produced in higher quantity due to anticancer treatments. OS impinges on spermatozoa (represented by the red stars) and damages to cell/sperm and mitochondria membranes, DNA damage, and defects in the sperm mid-piece and axonemal region can be observed. The establishment of this compromised process leads to abnormal semen characteristics and is responsible for the fertility decline present in men submitted to anticancer treatments. Reactive oxygen species (ROS).
Females, unlike males, have a limited reproductive life span that is dependent on the number of primordial follicles (see Section 2.3 of this review). As such, conventional chemotherapeutic agents can lead to permanent ovarian failure and amenorrhea due to oocyte depletion [154,155]. This can occur by direct damage to granulosa cells, as these cells are an easy target for chemotherapeutic agents due to their highly proliferative rate. Reduced number of granulosa cells might deprive the oocyte of nutrient supply and disrupt granulosa/oocyte communication (vital for oocyte maturation), inducing oocyte apoptosis [156]. Even when gametes are spared, it is still possible that the damage caused to other ovarian components, such as ovarian vasculature and stroma, will also contribute to premature ovarian failure [157,158,159] (Figure 5).
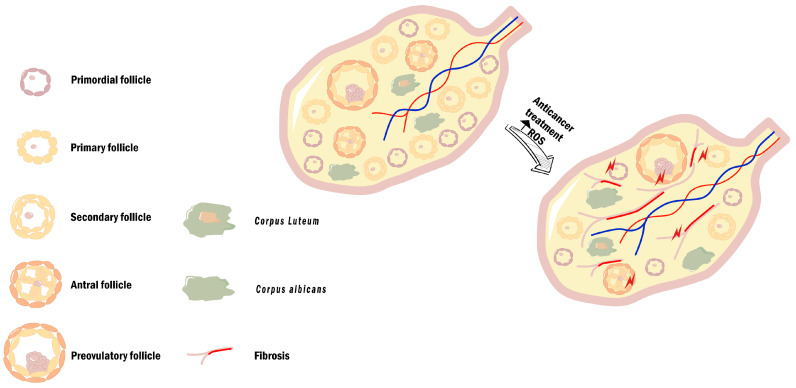
Figure 5.
Ovarian tissue dysfunction after anticancer treatments. Increase in OS-derived from anticancer treatment, due to increased ROS production and impaired antioxidant response leads to the establishment of an oxidative microenvironment. In a post-treatment ovarian stroma, a depletion in the number of primordial and primary follicles and the presence of collagen deposition can be observed (fibrosis). The establishment of this compromised microenvironment impairs ovarian function and is responsible for the fertility decline present in women submitted to anticancer treatments. Reactive oxygen species (ROS). Cisplatin and doxorubicin are two widely used chemotherapeutic drugs to treat several types of cancer, including those of the reproductive tract. Their ROS-mediated effects on fertility will now be revised.
Cisplatin is a highly reactive molecule that binds to DNA and forms nDNA (nuclear DNA) adducts (mechanism of cytotoxicity) and mitochondrial DNA (mtDNA) adducts (ROS-promoting mechanism). Cisplatin interacts with DNA by mainly forming Pt-d (GpG) di-adducts, which if not repaired by the DNA damage response will block replication and/or transcription and lead to apoptosis [123,160]. Cisplatin also binds to RNA and proteins. Mitochondrial membrane proteins, particularly voltage-dependent anion channels, are preferential binding sites [161]. Cisplatin also interferes with the activity of several proteins involved in the maintenance of redox balance. In the testicular tissue, cisplatin decreases GSH and Cat activity, which may increase the vulnerability of germ cells to ROS deleterious effects [162,163]. An increase in cisplatin-mediated ROS, at the testicular ECM, activates fibroblasts (by transforming growth factor-beta upregulation), and increases collagen accumulation with deleterious consequences in the structure of the seminiferous epithelium and a reduction in the spermatogenic activity [162,164]. In Leydig cells, dysfunction was also observed, as cisplatin exerts an inhibitory action at the level of cytochrome P450, inhibiting testosterone synthesis [165,166]. Broader cisplatin damage on spermatogenic parameters includes abnormalities in sperm motility and sperm morphology [167]. In the ovaries, cisplatin increases primordial follicle activation and granulosa cells apoptosis, leading to primordial follicles depletion [168,169]. It also increases the end-product of lipid peroxidation, malondialdehyde (MDA), and decreases SOD and GSH antioxidant activity. Chemoprotective effects have been observed with the use of molecules with antioxidant properties [170,171,172].
Doxorubicin, an antiproliferative (by inhibition of topoisomerase II), highly reactive (by DNA intercalation), and ROS promotor (by iron chelation) molecule, does not exclusively damage neoplastic cells, but also healthy dividing cells, as germ cells. In males, unwanted reproductive side effects occur through an increase in testicular oxidative stress, inflammation, and apoptosis [173,174,175]. A reduction in sperm quality (e.g., loss of acrosome integrity or morphological abnormalities, and motility) has been seen after doxorubicin treatment [176,177]. A decrease in body and relative testicular weights, reduced seminiferous tubule diameter, and germinal epithelium thickness have also been observed [178]. These changes probably result from atrophy of Leydig cells and the reduction in the germ cell number and spermatogenic proliferative rate [179,180]. Additionally, in the testicular tissue of doxorubicin-treated subjects, the levels of apoptotic-related genes (e.g., caspase 3 and B-cell lymphoma 2 genes) and MDA are increased, and the activities of antioxidant enzymes (SOD and GPx) are reduced [176,179]. In female mice, doxorubicin administration induced an imbalance in the redox state, by interfering with the activation of the antioxidant Nrf-2 pathway and the expression of antioxidant enzymes SOD, Cat, and GPx [181,182]. Divergent results regarding the expression of antioxidant enzymes in response to doxorubicin treatment have been reported and may result from experimental designs used and the specific self-protective response of cells (oocytes, granulosa, and cumulus) [181,182]. Nevertheless, beneficial effects were observed with the use of molecules that modulate redox balance [181,182,183]. Just like in males, doxorubicin influences the inflammatory response by inducing a significant increase in the expression of pro-inflammatory cytokines (e.g., tumor necrosis factor-alpha, interleukin 6 and 8) [184]. Inflammation activates matrix metalloproteinases (MMPs) and induces alterations in the degradation of the extracellular matrix that may favor an excessive collagen deposition and contribute to ovarian fibrosis [185]. Additionally, MMPs activation may also regulate the local recruitment and availability of inflammatory mediators, acting as a positive feedback loop of inflammation. Doxorubicin treatment is associated with local inflammatory responses and morphological damage to oocytes and stroma, and protective effects of antioxidant molecules were, once more, observed [184].
Although studies on the effects of molecular target therapy on fertility are still scarce, some data on male fertility are available [186,187]. Bortezomib, an antineoplastic agent and a proteasome inhibitor frequently used for multiple myeloma and mantle cell lymphoma, induces tumor cell apoptosis via the induction of endoplasmic reticulum stress (the capacity to fold proteins becomes saturated), increased expression of p53 (tumor suppressor), and activation of caspase-3 (cell death inductors) [188]. In males, bortezomib induces germ cell development arrest, impairing the spermatogenic process [189]. In fact, a study by Li and colleagues demonstrated that OS induced by bortezomib increased testicular MDA and decreased GPx and total SOD protein levels [190]. Besides, it also caused an imbalance in cell signaling, disrupted Sertoli-germ cell anchoring junctions, and interfered with spermatogenesis. Additionally, the study also provided evidence that FSH counteracted bortezomib’s negative effects by regulation of a pro-survival response to OS-mediated insults to Sertoli cells (via Akt/ERK pathway) [190].
Other less investigated anticarcinogenic agents, their ROS-dependent mechanisms of action, and effects on fertility are summarized in Table 2.
Table 2.
Other anticarcinogenic agents and their effects on fertility.
| Name | Fertility Effect | Ref | ROS-Known Effect | Ref |
|---|---|---|---|---|
| 5-fluorouracil | Decreased sperm count (rat) | [191] | Inflammation, autophagy, apoptosis, and senescence induction | [192,193,194] |
| Erlotinib | - | Increase radical’s production through NOX4 | [195] | |
| Imatinib | Reduces sperm count and density (human) | [196] | Reduces MMP and complex I activity of ETC, leading to mitochondrial OS | [197] |
| Decrease vasculature of placenta (mouse) | [198] | |||
| Diminishes primordial follicles (mouse) | [198] | |||
| Rituximab | No mentionable effects (human and mouse) | [199,200,201] | - | |
| Buthionine sulfoximine | - | Mitochondrial impairment | [202,203] | |
| Imexon | - | GSH depletion and induction of ER stress | [129,135] |
Electron transport chain (ETC); endoplasmic reticulum (ER); glutathione (GSH); metalloproteinases (MMP); NADPH oxidase 4 (NOX4); oxidative stress (OS).
4. Conclusions
Induced loss of redox balance can be a strategy used in anticancer therapies. However, the effectiveness of drugs contrasts with new problems and challenges that arise from the increase in patients’ survival and their aims to become parents. As ROS have a pivotal role in male and female gametogenesis processes, ROS-associated side effects of anticancer therapies on reproductive systems can compromise fertility. For this reason, there has been an increase in studies aiming to shed light on the mechanisms involved in the loss of fertility associated with anticancer treatments and innovative ways of ameliorating them.
It is important to continue the pursuit of such new strategies and in parallel explain to patients the available options to bypass anticancer treatment side effects on the reproductive system.
Author Contributions
Literature search and analysis, S.M.; conceptualization, R.S., M.S. and E.S.; manuscript writing, S.M.; critical discussion, S.M., R.S., M.M., F.M., M.S. and E.S.; review of the manuscript, R.S., M.M., F.M., M.S. and E.S. All authors have read and agreed to the published version of the manuscript.
Funding
Research from R.S. and M.S. are partially supported by the Unit for Multidisciplinary Research in Biomedicine (UMIB), funded by the Foundation for Science and Technology (FCT) Portugal (grant numbers UIDB/00215/2020, and UIDP/00215/2020), and the ITR-Laboratory for Integrative and Translational Research in Population Health (LA/P/0064/2020). E.S. contract is funded by the Portuguese Government and the European Union through DL57/2016/CP1327/CT0006.
Conflicts of Interest
We here state that the material contained in the submitted manuscript has not been published previously, and has not being submitted elsewhere for publication. We declare that we all participated in the present study and that we have seen and approved the final version. We also declare that if this publication is accepted, it will not be published elsewhere including electronically in the same form, in English or in any other language, without the written consent of the copyright-holder. There was also no Fabrication, falsification, plagiarism, repetitive publications, obfuscation, no human research or experimentation. All authors also agree the order of authorship. All Authors disclose any actual or potential conflict of interest including any financial, personal or other relationships with other people or organizations within three years of beginning the submitted work that could inappropriately influence, or be perceived to influence their work, such as manufacturers of pharmaceuticals, laboratory supplies, and/or medical devices. It also includes employment, consultancies, stock ownership, honoraria, paid expert testimony, patent applications/registrations, and grants or other funding, including any financial arrangement with a company whose product is prominent in the submitted manuscript or with a company making a competing product, and any commercial affiliations.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Chabner B., Roberts T. Timeline: Chemotherapy and the War on Cancer. Nat. Rev. Cancer. 2005;5:65–72. doi: 10.1038/nrc1529. [DOI] [PubMed] [Google Scholar]
- 2.Kim S.J., Kim H.S., Seo Y.R. Understanding of ROS-Inducing Strategy in Anticancer Therapy. Oxidative Med. Cell. Longev. 2019;2019:5381692. doi: 10.1155/2019/5381692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhuang B., Luo X., Rao H., Li Q., Shan N., Liu X., Qi H. Oxidative stress-induced C/EBPbeta inhibits beta-catenin signaling molecule involving in the pathology of Preeclampsia. Placenta. 2015;36:839–846. doi: 10.1016/j.placenta.2015.06.016. [DOI] [PubMed] [Google Scholar]
- 4.Panji M., Behmard V., Zare Z., Malekpour M., Nejadbiglari H., Yavari S., Nayerpour T., Safaeian A., Maleki N., Abbasi M., et al. Suppressing effects of green tea extract and Epigallocatechin-3-gallate (EGCG) on TGF-β- induced Epitheli-al-to-mesenchymal transition via ROS/Smad signaling in human cervical cancer cells. Gene. 2021;794:145774. doi: 10.1016/j.gene.2021.145774. [DOI] [PubMed] [Google Scholar]
- 5.Piktel E., Ościłowska I., Suprewicz Ł., Depciuch J., Marcińczyk N., Chabielska E., Wolak P., Wollny T., Janion M., Parlinska-Wojtan M., et al. ROS-Mediated Apoptosis and Autophagy in Ovarian Cancer Cells Treated with Peanut-Shaped Gold Nano-particles. Int. J. Nanomed. 2021;16:1993–2011. doi: 10.2147/IJN.S277014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cheng Q., Bao L., Li M., Chang K., Yi X. Erastin synergizes with cisplatin via ferroptosis to inhibit ovarian cancer growth in vitro and in vivo. J. Obstet. Gynaecol. Res. 2021;47:2481–2491. doi: 10.1111/jog.14779. [DOI] [PubMed] [Google Scholar]
- 7.Angsutararux P., Luanpitpong S., Issaragrisil S. Chemotherapy-Induced Cardiotoxicity: Overview of the Roles of Oxidative Stress. Oxidative Med. Cell. Longev. 2015;2015:795602. doi: 10.1155/2015/795602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Karasawa T., Steyger P.S. An integrated view of cisplatin-induced nephrotoxicity and ototoxicity. Toxicol. Lett. 2015;237:219–227. doi: 10.1016/j.toxlet.2015.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hanahan D., Weinberg R.A. Hallmarks of cancer: The next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 10.Bhatti M.T., Salama A. Neuro-ophthalmic side effects of molecularly targeted cancer drugs. Eye. 2017;32:287–301. doi: 10.1038/eye.2017.222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hansel T.T., Kropshofer H., Singer T., Mitchell J.A., George A.J.T. The safety and side effects of monoclonal antibodies. Nat. Rev. Drug Discov. 2010;9:325–338. doi: 10.1038/nrd3003. [DOI] [PubMed] [Google Scholar]
- 12.Kroschinsky F., on behalf of the Intensive Care in Hematological and Oncological Patients (iCHOP) Collaborative Group. Stölzel F., von Bonin S., Beutel G., Kochanek M., Kiehl M., Schellongowski P. New drugs, new toxicities: Severe side effects of modern targeted and immunotherapy of cancer and their management. Crit. Care. 2017;21:89. doi: 10.1186/s13054-017-1678-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Raso V. Antibodies in diagnosis and therapy. The magic bullet--nearing the century mark. Semin. Cancer Biol. 1990;1:227–242. [PubMed] [Google Scholar]
- 14.Dimitrov D.S., Marks J.D. Therapeutic antibodies: Current state and future trends—Is a paradigm change coming soon? Methods Mol. Biol. 2009;525:1–27. doi: 10.1007/978-1-59745-554-1_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Teppo H.-R., Soini Y., Karihtala P. Reactive Oxygen Species-Mediated Mechanisms of Action of Targeted Cancer Therapy. Oxidative Med. Cell. Longev. 2017;2017:1485283. doi: 10.1155/2017/1485283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Meistrich M.L. Effects of chemotherapy and radiotherapy on spermatogenesis in humans. Fertil. Steril. 2013;100:1180–1186. doi: 10.1016/j.fertnstert.2013.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Sabanegh E.S., Ragheb A.M. Male Fertility after Cancer. Urology. 2009;73:225–231. doi: 10.1016/j.urology.2008.08.474. [DOI] [PubMed] [Google Scholar]
- 18.Meirow D., Nugent D. The effects of radiotherapy and chemotherapy on female reproduction. Hum. Reprod. Update. 2001;7:535–543. doi: 10.1093/humupd/7.6.535. [DOI] [PubMed] [Google Scholar]
- 19.Meirow D., Biederman H., Anderson R.A., Wallace W.H.B. Toxicity of Chemotherapy and Radiation on Female Reproduction. Clin. Obstet. Gynecol. 2010;53:727–739. doi: 10.1097/GRF.0b013e3181f96b54. [DOI] [PubMed] [Google Scholar]
- 20.Chasle S., How C.C. The effect of cytotoxic chemotherapy on female fertility. Eur. J. Oncol. Nurs. 2003;7:91–98. doi: 10.1016/S1462-3889(02)00081-9. [DOI] [PubMed] [Google Scholar]
- 21.Nudell D.M., Monoski M.M., Lipshultz L.I. Common medications and drugs: How they affect male fertility. Urol. Clin. 2002;29:965–973. doi: 10.1016/S0094-0143(02)00079-4. [DOI] [PubMed] [Google Scholar]
- 22.Green D.M., Kawashima T., Stovall M., Leisenring W., Sklar C.A., Mertens A.C., Donaldson S.S., Byrne J., Robison L.L. Fertility of Female Survivors of Childhood Cancer: A Report From the Childhood Cancer Survivor Study. J. Clin. Oncol. 2009;27:2677–2685. doi: 10.1200/JCO.2008.20.1541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fleischer R.T., Vollenhoven B.J., Weston G.C. The Effects of Chemotherapy and Radiotherapy on Fertility in Premenopausal Women. Obstet. Gynecol. Surv. 2011;66:248–254. doi: 10.1097/OGX.0b013e318224e97b. [DOI] [PubMed] [Google Scholar]
- 24.Trottmann M., Becker A.J., Stadler T., Straub J., Soljanik I., Schlenker B., Stief C.G. Semen Quality in Men with Malignant Diseases before and after Therapy and the Role of Cryopreservation. Eur. Urol. 2007;52:355–367. doi: 10.1016/j.eururo.2007.03.085. [DOI] [PubMed] [Google Scholar]
- 25.Choy J.T., Brannigan R.E. The determination of reproductive safety in men during and after cancer treatment. Fertil. Steril. 2013;100:1187–1191. doi: 10.1016/j.fertnstert.2013.07.1974. [DOI] [PubMed] [Google Scholar]
- 26.Kim S.-Y., Cho G.J., Davis J.S. Consequences of chemotherapeutic agents on primordial follicles and future clinical applica-tions. Obstet. Gynecol. Sci. 2019;62:382–390. doi: 10.5468/ogs.2019.62.6.382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Stefansdottir A., Johnston Z.C., Powles-Glover N., Anderson R.A., Adams I.R., Spears N. Etoposide damages female germ cells in the developing ovary. BMC Cancer. 2016;16:482. doi: 10.1186/s12885-016-2505-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Van Der Kaaij M.A., van Echten-Arends J., Simmons A.H.M., Kluin-Nelemans H.C. Fertility preservation after chemotherapy for Hodgkin lymphoma. Hematol. Oncol. 2010;28:168–179. doi: 10.1002/hon.939. [DOI] [PubMed] [Google Scholar]
- 29.Sigman M. Introduction: Cancer treatment and male fertility: Effects of therapy and current and future management options. Fertil. Steril. 2013;100:1179. doi: 10.1016/j.fertnstert.2013.09.011. [DOI] [PubMed] [Google Scholar]
- 30.Forman E.J., Anders C.K., Behera M.A. A nationwide survey of oncologists regarding treatment-related infertility and fertility preservation in female cancer patients. Fertil. Steril. 2010;94:1652–1656. doi: 10.1016/j.fertnstert.2009.10.008. [DOI] [PubMed] [Google Scholar]
- 31.Meistrich M.L. Male gonadal toxicity. Pediatr. Blood Cancer. 2009;53:261–266. doi: 10.1002/pbc.22004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wallace W.H.B., Anderson R.A., Irvine D.S. Fertility preservation for young patients with cancer: Who is at risk and what can be offered? Lancet Oncol. 2005;6:209–218. doi: 10.1016/S1470-2045(05)70092-9. [DOI] [PubMed] [Google Scholar]
- 33.Grigg A. The Impact of Conventional and High-Dose Therapy for Lymphoma on Fertility. Clin. Lymphoma. 2004;5:84–88. doi: 10.3816/CLM.2004.n.013. [DOI] [PubMed] [Google Scholar]
- 34.Schrader M., Müller M., Straub B., Miller K. The impact of chemotherapy on male fertility: A survey of the biologic basis and clinical aspects. Reprod. Toxicol. 2001;15:611–617. doi: 10.1016/S0890-6238(01)00182-4. [DOI] [PubMed] [Google Scholar]
- 35.Lambertini M., Del Mastro L., Pescio M.C., Andersen C.Y., Azim H.A., Jr., Peccatori F.A., Costa M., Revelli A., Salvagno F., Gennari A., et al. Cancer and fertility preservation: International recommendations from an expert meeting. BMC Med. 2016;14:1. doi: 10.1186/s12916-015-0545-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rabaca A., Sousa M., Alves M.G., Oliveira P.F., Sá R. Novel drug therapies for fertility preservation in men undergoing chemotherapy: Clinical relevance of pro-tector agents. Curr. Med. Chem. 2015:3347–3369. doi: 10.2174/0929867322666150821094334. [DOI] [PubMed] [Google Scholar]
- 37.Blumenfeld Z. Chemotherapy and fertility. Best Pract. Res. Clin. Obstet. Gynaecol. 2012;26:379–390. doi: 10.1016/j.bpobgyn.2011.11.008. [DOI] [PubMed] [Google Scholar]
- 38.Ahmed M.R., Edmund S.S., Jr. Male fertility-implications of anticancer treatment and strategies to mitigate gonadotoxicity. Anti-Cancer Agents Med. Chem. 2010;10:92–102. doi: 10.2174/1871520611009010092. [DOI] [PubMed] [Google Scholar]
- 39.Puscheck E., Philip P.A., Jeyendran R.S. Male fertility preservation and cancer treatment. Cancer Treat. Rev. 2004;30:173–180. doi: 10.1016/j.ctrv.2003.07.005. [DOI] [PubMed] [Google Scholar]
- 40.Hobbie W.L., Ogle S.K., Ginsberg J.P. Fertility Concerns for Young Males Undergoing Cancer Therapy. Semin. Oncol. Nurs. 2009;25:245–250. doi: 10.1016/j.soncn.2009.08.001. [DOI] [PubMed] [Google Scholar]
- 41.Kohen R., Nyska A. Invited Review: Oxidation of Biological Systems: Oxidative Stress Phenomena, Antioxidants, Redox Reactions, and Methods for Their Quantification. Toxicol. Pathol. 2002;30:620–650. doi: 10.1080/01926230290166724. [DOI] [PubMed] [Google Scholar]
- 42.Halliwell B., Cross C.E. Oxygen-derived species: Their relation to human disease and environmental stress. Environ. Health Perspect. 1994;102:5–12. doi: 10.1289/ehp.94102s105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kehrer J.P. The Haber-Weiss reaction and mechanisms of toxicity. Toxicology. 2000;149:43–50. doi: 10.1016/S0300-483X(00)00231-6. [DOI] [PubMed] [Google Scholar]
- 44.Castro J.P., Jung T., Grune T., Almeida H. Actin carbonylation: From cell dysfunction to organism disorder. J. Proteom. 2013;92:171–180. doi: 10.1016/j.jprot.2013.05.006. [DOI] [PubMed] [Google Scholar]
- 45.Orient A., Donkó S.A., Leto T.L., Geiszt M. Novel sources of reactive oxygen species in the human body. Nephrol. Dial. Transplant. 2007;22:1281–1288. doi: 10.1093/ndt/gfm077. [DOI] [PubMed] [Google Scholar]
- 46.He L., He T., Farrar S., Ji L., Liu T., Ma X. Antioxidants Maintain Cellular Redox Homeostasis by Elimination of Reactive Oxygen Species. Cell. Physiol. Biochem. 2017;44:532–553. doi: 10.1159/000485089. [DOI] [PubMed] [Google Scholar]
- 47.Mironczuk-Chodakowska I., Witkowska A., Zujko M.E. Endogenous non-enzymatic antioxidants in the human body. Adv. Med. Sci. 2018;63:68–78. doi: 10.1016/j.advms.2017.05.005. [DOI] [PubMed] [Google Scholar]
- 48.Martindale J.L., Holbrook N.J. Cellular response to oxidative stress: Signaling for suicide and survival. J. Cell. Physiol. 2002;192:1–15. doi: 10.1002/jcp.10119. [DOI] [PubMed] [Google Scholar]
- 49.Zhang J., Wang X., Vikash V., Ye Q., Wu D., Liu Y., Dong W. ROS and ROS-mediated cellular signaling. Oxidative Med. Cell. Longev. 2016;2016:4350965. doi: 10.1155/2016/4350965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang W., Liu H.T. MAPK signal pathways in the regulation of cell proliferation in mammalian cells. Cell Res. 2002;12:9–18. doi: 10.1038/sj.cr.7290105. [DOI] [PubMed] [Google Scholar]
- 51.Rawlings J.S., Rosler K.M., Harrison D.A. The JAK/STAT signaling pathway. J. Cell Sci. 2004;117:1281–1283. doi: 10.1242/jcs.00963. [DOI] [PubMed] [Google Scholar]
- 52.Cantley L.C. The Phosphoinositide 3-Kinase Pathway. Science. 2002;296:1655–1657. doi: 10.1126/science.296.5573.1655. [DOI] [PubMed] [Google Scholar]
- 53.Sies H., Berndt C., Jones D.P. Oxidative Stress. Annu. Rev. Biochem. 2017;86:715–748. doi: 10.1146/annurev-biochem-061516-045037. [DOI] [PubMed] [Google Scholar]
- 54.Sies H. Oxidative stress: A concept in redox biology and medicine. Redox Biol. 2015;4:180–183. doi: 10.1016/j.redox.2015.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Clermont Y. The cycle of the seminiferous epithelium in man. Am. J. Anat. 1963;112:35–51. doi: 10.1002/aja.1001120103. [DOI] [PubMed] [Google Scholar]
- 56.Siu M.K.Y., Cheng C.Y. Extracellular Matrix and Its Role in Spermatogenesis. Mol. Mech. Spermatogenesis. 2009;636:74–91. doi: 10.1007/978-0-387-09597-4_5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Phillips B.T., Gassei K., Orwig K.E. Spermatogonial stem cell regulation and spermatogenesis. Philos. Trans. R. Soc. B Biol. Sci. 2010;365:1663–1678. doi: 10.1098/rstb.2010.0026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Hess R.A., Franca L.R.D. Spermatogenesis and cycle of the seminiferous epithelium. Adv. Exp. Med. Biol. 2008;636:1–15. doi: 10.1007/978-0-387-09597-4_1. [DOI] [PubMed] [Google Scholar]
- 59.Griswold M.D. Spermatogenesis: The Commitment to Meiosis. Physiol. Rev. 2016;96:1–17. doi: 10.1152/physrev.00013.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hao S.-L., Ni F.-D., Yang W.-X. The dynamics and regulation of chromatin remodeling during spermiogenesis. Gene. 2019;706:201–210. doi: 10.1016/j.gene.2019.05.027. [DOI] [PubMed] [Google Scholar]
- 61.Yoshinaga K., Toshimori K. Organization and modifications of sperm acrosomal molecules during spermatogenesis and epi-didymal maturation. Microsc. Res. Tech. 2003;61:39–45. doi: 10.1002/jemt.10315. [DOI] [PubMed] [Google Scholar]
- 62.Griswold M.D. The central role of Sertoli cells in spermatogenesis. Semin. Cell Dev. Biol. 1998;9:411–416. doi: 10.1006/scdb.1998.0203. [DOI] [PubMed] [Google Scholar]
- 63.Aitken R.J., Buckingham D.W., West K.M. Reactive oxygen species and human spermatozoa: Analysis of the cellular mecha-nisms involved in luminol- and lucigenin-dependent chemiluminescence. J. Cell. Physiol. 1992;151:466–477. doi: 10.1002/jcp.1041510305. [DOI] [PubMed] [Google Scholar]
- 64.Gavella M., Lipovac V. Nadh-Dependent Oxidoreductase (Diaphorase) Activity and Isozyme Pattern of Sperm in Infertile Men. Arch. Androl. 1992;28:135–141. doi: 10.3109/01485019208987691. [DOI] [PubMed] [Google Scholar]
- 65.Gil-Guzman E., Ollero M., Lopez M.C., Sharma R.K., Alvarez J.G., Thomas A.J., Jr., Agarwal A. Differential production of reactive oxygen species by subsets of human spermatozoa at different stages of maturation. Hum. Reprod. 2001;16:1922–1930. doi: 10.1093/humrep/16.9.1922. [DOI] [PubMed] [Google Scholar]
- 66.Fisher H.M., Aitken R.J. Comparative analysis of the ability of precursor germ cells and epididymal spermatozoa to generate reactive oxygen metabolites. J. Exp. Zool. 1997;277:390–400. doi: 10.1002/(SICI)1097-010X(19970401)277:5<390::AID-JEZ5>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 67.Aguilar-Mahecha A., Hales B.F., Robaire B. Expression of stress response genes in germ cells during spermatogenesis. Biol. Reprod. 2001;65:119–127. doi: 10.1095/biolreprod65.1.119. [DOI] [PubMed] [Google Scholar]
- 68.Aitken R.J. The amoroso lecture. The human spermatozoon--a cell in crisis? J. Reprod. Fertil. 1999;115:1–7. doi: 10.1530/jrf.0.1150001. [DOI] [PubMed] [Google Scholar]
- 69.Oldereid N.B., Thomassen Y., Purvis K. Selenium in human male reproductive organs. Hum. Reprod. 1998;13:2172–2176. doi: 10.1093/humrep/13.8.2172. [DOI] [PubMed] [Google Scholar]
- 70.Kemal D.N., Morshedi M., Oehninger S. Effects of hydrogen peroxide on DNA and plasma membrane integrity of human spermatozoa. Fertil. Steril. 2000;74:1200–1207. doi: 10.1016/S0015-0282(00)01591-0. [DOI] [PubMed] [Google Scholar]
- 71.Aitken R.J. Free radicals, lipid peroxidation and sperm function. Reprod. Fertil. Dev. 1995;7:659–668. doi: 10.1071/RD9950659. [DOI] [PubMed] [Google Scholar]
- 72.Aitken R.J., Baker M.A., Sawyer D. Oxidative stress in the male germ line and its role in the aetiology of male infertility and genetic disease. Reprod. Biomed. Online. 2003;7:65–70. doi: 10.1016/S1472-6483(10)61730-0. [DOI] [PubMed] [Google Scholar]
- 73.Agarwal A., Nallella K.P., Allamaneni S.S., Said T.M. Role of antioxidants in treatment of male infertility: An overview of the literature. Reprod. Biomed. Online. 2004;8:616–627. doi: 10.1016/S1472-6483(10)61641-0. [DOI] [PubMed] [Google Scholar]
- 74.Cocuzza M., Sikka S.C., Athayde K.S., Agarwal A. Clinical relevance of oxidative stress and sperm chromatin damage in male infertility: An evidence based analysis. Int. Braz. J. Urol. 2007;33:603–621. doi: 10.1590/S1677-55382007000500002. [DOI] [PubMed] [Google Scholar]
- 75.Tai P., Ascoli M. Reactive oxygen species (ROS) play a critical role in the cAMP-induced activation of Ras and the phos-phorylation of ERK1/2 in Leydig cells. Mol. Endocrinol. 2011;25:885–893. doi: 10.1210/me.2010-0489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Plante M., Lamirande E.D., Gagnon C. Reactive oxygen species released by activated neutrophils, but not by deficient spermatozoa, are sufficient to affect normal sperm motility. Fertil. Steril. 1994;62:387–393. doi: 10.1016/S0015-0282(16)56895-2. [DOI] [PubMed] [Google Scholar]
- 77.Agarwal A., Saleh R., Bedaiwy M. Role of reactive oxygen species in the pathophysiology of human reproduction. Fertil. Steril. 2003;79:829–843. doi: 10.1016/S0015-0282(02)04948-8. [DOI] [PubMed] [Google Scholar]
- 78.Safarnavadeh T., Rastegarpanah M. Antioxidants and infertility treatment, the role of Satureja Khuzestanica: A mini-systematic review. Iran. J. Reprod. Med. 2011;9:61–70. [PMC free article] [PubMed] [Google Scholar]
- 79.Gao S., Li C., Chen L., Zhou X. Actions and mechanisms of reactive oxygen species and antioxidative system in semen. Mol. Cell. Toxicol. 2017;13:143–154. doi: 10.1007/s13273-017-0015-8. [DOI] [Google Scholar]
- 80.Barati E., Nikzad H., Karimian M. Oxidative stress and male infertility: Current knowledge of pathophysiology and role of antioxidant therapy in disease management. Cell. Mol. Life Sci. 2019;77:93–113. doi: 10.1007/s00018-019-03253-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Bardaweel S.K., Gul M., Alzweiri M., Ishaqat A., ALSalamat H.A., Bashatwah R.M. Reactive Oxygen Species: The Dual Role in Physiological and Pathological Conditions of the Human Body. Eurasian J. Med. 2018;50:193–201. doi: 10.5152/eurasianjmed.2018.17397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Berby B., Bichara C., Rives-Feraille A., Jumeau F., Pizio P., Sétif V., Sibert L., Dumont L., Rondanino C., Rives N. Oxidative Stress Is Associated with Telomere Interaction Impairment and Chromatin Condensation Defects in Spermatozoa of Infertile Males. Antioxidants. 2021;10:593. doi: 10.3390/antiox10040593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Santi D., Spaggiari G., Simoni M. Sperm DNA fragmentation index as a promising predictive tool for male infertility diagnosis and treatment management-meta-analyses. Reprod. Biomed. Online. 2018;37:315–326. doi: 10.1016/j.rbmo.2018.06.023. [DOI] [PubMed] [Google Scholar]
- 84.Agarwal A., Parekh N., Selvam M.K.P., Henkel R., Shah R., Homa S.T., Ramasamy R., Ko E., Tremellen K., Esteves S., et al. Male oxidative stress infertility (MOSI): Proposed terminology and clinical practice guidelines for man-agement of idiopathic male infertility. World J. Mens Health. 2019;37:296–312. doi: 10.5534/wjmh.190055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Li H., Chian R.-C. Follicular Development and Oocyte Growth. In: Chian R.-C., Nargund G., Huang J.Y.J., editors. Development of In Vitro Maturation for Human Oocytes: Natural and Mild Approaches to Clinical Infertility Treatment. Springer International Publishing; Cham, Switzerland: 2017. pp. 37–57. [Google Scholar]
- 86.Von Stetina J.R., Orr-Weaver T.L. Developmental Control of Oocyte Maturation and Egg Activation in Metazoan Models. Cold Spring Harb. Perspect. Biol. 2011;3:a005553. doi: 10.1101/cshperspect.a005553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Baker T.G. A quantitative and cytological study of germ cells in human ovaries. Proc. R. Soc. Lond. Ser. B Boil. Sci. 1963;158:417–433. doi: 10.1097/00006254-196408000-00038. [DOI] [PubMed] [Google Scholar]
- 88.Gougeon A., Chainy G. Morphometric studies of small follicles in ovaries of women at different ages. Reproduction. 1987;81:433–442. doi: 10.1530/jrf.0.0810433. [DOI] [PubMed] [Google Scholar]
- 89.Sánchez F., Smitz J. Molecular control of oogenesis. Biochim. Biophys. Acta BBA Mol. Basis Dis. 2012;1822:1896–1912. doi: 10.1016/j.bbadis.2012.05.013. [DOI] [PubMed] [Google Scholar]
- 90.Rimon-Dahari N., Terushalmi-Heinemann L., Alyagor L., Dekel N. Ovarian Folliculogenesis. Results Probl. Cell Differ. 2016;58:167–190. doi: 10.1007/978-3-319-31973-5_7. [DOI] [PubMed] [Google Scholar]
- 91.Baerwald A.R., Adams G.P., Pierson R.A. Ovarian antral folliculogenesis during the human menstrual cycle: A review. Hum. Reprod. Update. 2011;18:73–91. doi: 10.1093/humupd/dmr039. [DOI] [PubMed] [Google Scholar]
- 92.Gellersen B., Brosens J. Cyclic Decidualization of the Human Endometrium in Reproductive Health and Failure. Endocr. Rev. 2014;35:851–905. doi: 10.1210/er.2014-1045. [DOI] [PubMed] [Google Scholar]
- 93.Messinis I.E. Ovarian feedback, mechanism of action and possible clinical implications. Hum. Reprod. Update. 2006;12:557–571. doi: 10.1093/humupd/dml020. [DOI] [PubMed] [Google Scholar]
- 94.Farage M.A., Neill S., MacLean A.B. Physiological changes associated with the menstrual cycle: A review. Obstet. Gynecol. Surv. 2009;64:58–72. doi: 10.1097/OGX.0b013e3181932a37. [DOI] [PubMed] [Google Scholar]
- 95.Bates G.W., Bowling M. Physiology of the female reproductive axis. Periodontology 2000. 2012;61:89–102. doi: 10.1111/j.1600-0757.2011.00409.x. [DOI] [PubMed] [Google Scholar]
- 96.Halliwell B., Gutteridge J. Free Radicals and Antioxidant Protection: Mechanisms and Significance in Toxicology and Disease. Hum. Toxicol. 1988;7:7–13. doi: 10.1177/096032718800700102. [DOI] [PubMed] [Google Scholar]
- 97.Sugino N., Nakamura Y., Takeda O., Ishimatsu M., Kato H. Changes in activities of superoxide dismutase and lipid peroxide in corpus luteum during pregnancy in rats. Reproduction. 1993;97:347–351. doi: 10.1530/jrf.0.0970347. [DOI] [PubMed] [Google Scholar]
- 98.Devine P.J., Perreault S.D., Luderer U. Roles of Reactive Oxygen Species and Antioxidants in Ovarian Toxicity. Biol. Reprod. 2012;86:27. doi: 10.1095/biolreprod.111.095224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Shkolnik K., Tadmor A., Ben-Dor S., Nevo N., Galiani D., Dekel N. Reactive oxygen species are indispensable in ovulation. Proc. Natl. Acad. Sci. USA. 2011;108:1462–1467. doi: 10.1073/pnas.1017213108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Ruder E.H., Hartman T.J., Goldman M.B. Impact of oxidative stress on female fertility. Curr. Opin. Obstet. Gynecol. 2009;21:219–222. doi: 10.1097/GCO.0b013e32832924ba. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Suzuki T., Sugino N., Fukaya T., Sugyama S., Uda T., Takaya R., Yajima A., Sasamo H. Superoxide dismutase in normal cycling human ovaries: Immunohistochemical localization and characterization. Fertil. Steril. 1999;72:720–726. doi: 10.1016/S0015-0282(99)00332-5. [DOI] [PubMed] [Google Scholar]
- 102.Shi L., Zhang J., Lai Z., Tian Y., Fang L., Wu M., Xiong J., Qin X., Luo A., Wang S. Long-Term Moderate Oxidative Stress Decreased Ovarian Reproductive Function by Reducing Follicle Quality and Progesterone Production. PLoS ONE. 2016;11:e0162194. doi: 10.1371/journal.pone.0162194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Silva E., Soares A.I., Costa F., Castro J.P., Matos L., Almeida H. Antioxidant Supplementation Modulates Age-Related Placental Bed Morphology and Reproductive Outcome in Mice. Biol. Reprod. 2015;93:56. doi: 10.1095/biolreprod.114.127746. [DOI] [PubMed] [Google Scholar]
- 104.Ferreira F.T., Mendes S., Rocha N.A., Matos L., Rodrigues A.R., Almeida H., Silva E. Apocynin Dietary Supplementation Delays Mouse Ovarian Ageing. Oxidative Med. Cell. Longev. 2019;2019:5316984. doi: 10.1155/2019/5316984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Cornelli U., Belcaro G., Cesarone M.R., Finco A. Analysis of oxidative stress during the menstrual cycle. Reprod. Biol. Endocrinol. 2013;11:74. doi: 10.1186/1477-7827-11-74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Strehlow K., Rotter S., Wassmann S., Adam O., Grohé C., Laufs K., Bohm M., Nickenig G. Modulation of Antioxidant Enzyme Expression and Function by Estrogen. Circ. Res. 2003;93:170–177. doi: 10.1161/01.RES.0000082334.17947.11. [DOI] [PubMed] [Google Scholar]
- 107.Xu X., Leng J.Y., Gao F., Zhao Z.A., Deng W.B., Liang X.H., Zhang Y.J., Zhang Z.R., Li M., Sha A.G., et al. Differential expression and anti-oxidant function of glutathione peroxidase 3 in mouse uterus during decidualization. FEBS Lett. 2014;588:1580–1589. doi: 10.1016/j.febslet.2014.02.043. [DOI] [PubMed] [Google Scholar]
- 108.Sugino N., Takiguchi S., Kashida S., Karube A., Nakamura Y., Kato H. Superoxide dismutase expression in the human corpus luteum during the menstrual cycle and in early pregnancy. Mol. Hum. Reprod. 2000;6:19–25. doi: 10.1093/molehr/6.1.19. [DOI] [PubMed] [Google Scholar]
- 109.Al-Gubory K.H., Garrel C., Faure P., Sugino N. Roles of antioxidant enzymes in corpus luteum rescue from reactive oxygen species-induced oxidative stress. Reprod. Biomed. Online. 2012;25:551–560. doi: 10.1016/j.rbmo.2012.08.004. [DOI] [PubMed] [Google Scholar]
- 110.Bedard K., Krause K.-H. The NOX Family of ROS-Generating NADPH Oxidases: Physiology and Pathophysiology. Physiol. Rev. 2007;87:245–313. doi: 10.1152/physrev.00044.2005. [DOI] [PubMed] [Google Scholar]
- 111.Davis J.M., Auten R.L. Maturation of the antioxidant system and the effects on preterm birth. Semin. Fetal Neonatal Med. 2010;15:191–195. doi: 10.1016/j.siny.2010.04.001. [DOI] [PubMed] [Google Scholar]
- 112.Mier-Cabrera J., Jimenez-Zamudio L.A., García-Latorre E., Cruz-Orozco O., Hernández-Guerrero C. Quantitative and qualitative peritoneal immune profiles, T-cell apoptosis and oxidative stress-associated characteristics in women with minimal and mild endometriosis. BJOG Int. J. Obstet. Gynaecol. 2010;118:6–16. doi: 10.1111/j.1471-0528.2010.02777.x. [DOI] [PubMed] [Google Scholar]
- 113.Mendes S., Timóteo-Ferreira F., Soares A.I., Rodrigues A.R., Silva A.M.N., Silveira S., Matos L., Saraiva J., Guedes-Martins L., Almeida H., et al. Age-related oxidative modifications to uterine albumin impair extravillous trophoblast cells function. Free Radic. Biol. Med. 2020;152:313–322. doi: 10.1016/j.freeradbiomed.2020.03.020. [DOI] [PubMed] [Google Scholar]
- 114.Mendes S., Timóteo-Ferreira F., Almeida H., Silva E. New Insights into the Process of Placentation and the Role of Oxidative Uterine Microenvironment. Oxidative Med. Cell. Longev. 2019;2019:9174521. doi: 10.1155/2019/9174521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Kong Q., Beel J., Lillehei K. A threshold concept for cancer therapy. Med. Hypotheses. 2000;55:29–35. doi: 10.1054/mehy.1999.0982. [DOI] [PubMed] [Google Scholar]
- 116.Klaunig J.E., Kamendulis L.M. The Role of Oxidative Stress in Carcinogenesis. Annu. Rev. Pharmacol. Toxicol. 2004;44:239–267. doi: 10.1146/annurev.pharmtox.44.101802.121851. [DOI] [PubMed] [Google Scholar]
- 117.Storz P. KRas, ROS and the initiation of pancreatic cancer. Small GTPases. 2016;8:38–42. doi: 10.1080/21541248.2016.1192714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Trachootham D., Alexandre J., Huang P. Targeting cancer cells by ROS-mediated mechanisms: A radical therapeutic ap-proach? Nat. Rev. Drug Discov. 2009;8:579–591. doi: 10.1038/nrd2803. [DOI] [PubMed] [Google Scholar]
- 119.Raza M.H., Siraj S., Arshad A., Waheed U., Aldakheel F., Alduraywish S., Arshad M. ROS-modulated therapeutic approaches in cancer treatment. J. Cancer Res. Clin. Oncol. 2017;143:1789–1809. doi: 10.1007/s00432-017-2464-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Wang J., Yi J. Cancer cell killing via ROS: To increase or decrease, that is the question. Cancer Biol. Ther. 2008;7:1875–1884. doi: 10.4161/cbt.7.12.7067. [DOI] [PubMed] [Google Scholar]
- 121.Hwang P.M., Bunz F., Yu J., Rago C., Chan T.A., Murphy M.P., Kelso G.F., Smith R.A., Kinzler K.W., Volgelstein B. Ferredoxin reductase affects p53-dependent, 5-fluorouracil-induced apoptosis in colorectal cancer cells. Nat. Med. 2001;7:1111–1117. doi: 10.1038/nm1001-1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Fribley A., Wang C.-Y. Proteasome inhibitor induces apoptosis through induction of endoplasmic reticulum stress. Cancer Biol. Ther. 2006;5:745–748. doi: 10.4161/cbt.5.7.2971. [DOI] [PubMed] [Google Scholar]
- 123.Marullo R., Werner E., Degtyareva N., Moore B., Altavilla G., Ramalingam S.S., Doetsch P.W. Cisplatin Induces a Mitochondrial-ROS Response That Contributes to Cytotoxicity Depending on Mitochondrial Redox Status and Bioenergetic Functions. PLoS ONE. 2013;8:e81162. doi: 10.1371/journal.pone.0081162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Thorn C.F., Oshiro C., Marsh S., Hernandez-Boussard T., McLeod H., Klein T.E., Altman R.B. Doxorubicin pathways: Pharmacodynamics and adverse effects. Pharmacogenet. Genomics. 2011;21:440–446. doi: 10.1097/FPC.0b013e32833ffb56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Shan F., Shao Z., Jiang S., Cheng Z. Erlotinib induces the human non-small-cell lung cancer cells apoptosis via activating ROS-dependent JNK pathways. Cancer Med. 2016;5:3166–3175. doi: 10.1002/cam4.881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Chang S.-P., Shen S.-C., Lee W.-R., Yang L.-L., Chen Y.-C. Imatinib mesylate induction of ROS-dependent apoptosis in melanoma B16F0 cells. J. Dermatol. Sci. 2011;62:183–191. doi: 10.1016/j.jdermsci.2011.03.001. [DOI] [PubMed] [Google Scholar]
- 127.Alas S., Ng C.P., Bonavida B. Rituximab modifies the cisplatin-mitochondrial signaling pathway, resulting in apoptosis in cisplatin-resistant non-Hodgkin’s lymphoma. Clin. Cancer Res. 2002;8:836–845. [PubMed] [Google Scholar]
- 128.Griffith O.W., Meister A. Potent and specific inhibition of glutathione synthesis by buthionine sulfoximine (S-n-butyl homo-cysteine sulfoximine) J. Biol. Chem. 1979;254:7558–7560. doi: 10.1016/S0021-9258(18)35980-5. [DOI] [PubMed] [Google Scholar]
- 129.Sheveleva E.V., Landowski T.H., Samulitis B.K., Bartholomeusz G., Powis G., Dorr R.T. Imexon Induces an Oxidative Endoplasmic Reticulum Stress Response in Pancreatic Cancer Cells. Mol. Cancer Res. 2012;10:392–400. doi: 10.1158/1541-7786.MCR-11-0359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Kim H.-J., Oridate N., Lotan R. Increased level of the p67phox subunit of NADPH oxidase by 4HPR in head and neck squamous carcinoma cells. Int. J. Oncol. 2005;27:787–790. [PubMed] [Google Scholar]
- 131.Wondrak G.T. NQO1-activated phenothiazinium redox cyclers for the targeted bioreductive induction of cancer cell apop-tosis. Free Radic. Biol. Med. 2007;43:178–190. doi: 10.1016/j.freeradbiomed.2007.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Pelicano H., Feng L., Zhou Y., Carew J.S., Hileman E.O., Plunkett W., Keating M.J., Huang P. Inhibition of mitochondrial respiration: A novel strategy to enhance drug-induced apoptosis in human leukemia cells by a reactive oxygen species-mediated mechanism. J. Biol. Chem. 2003;278:37832–37839. doi: 10.1074/jbc.M301546200. [DOI] [PubMed] [Google Scholar]
- 133.Kotamraju S., Chitambar C.R., Kalivendi S.V., Joseph J., Kalyanaraman B. Transferrin receptor-dependent iron uptake is responsible for doxorubicin-mediated apoptosis in endo-thelial cells: Role of oxidant-induced iron signaling in apoptosis. J. Biol. Chem. 2002;277:17179–17187. doi: 10.1074/jbc.M111604200. [DOI] [PubMed] [Google Scholar]
- 134.Kostrzewa-Nowak D., Paine M.J.I., Wolf C.R., Tarasiuk J. The role of bioreductive activation of doxorubicin in cytotoxic activity against leukaemia HL60-sensitive cell line and its multidrug-resistant sublines. Br. J. Cancer. 2005;93:89–97. doi: 10.1038/sj.bjc.6602639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Dvorakova K., Payne C.M., Tome M.E., Briehl M.M., McClure T., Dorr R.T. Induction of oxidative stress and apoptosis in myeloma cells by the aziridine-containing agent imexon. Biochem. Pharmacol. 2000;60:749–758. doi: 10.1016/S0006-2952(00)00380-4. [DOI] [PubMed] [Google Scholar]
- 136.Glasauer A., Chandel N.S. Targeting antioxidants for cancer therapy. Biochem. Pharmacol. 2014;92:90–101. doi: 10.1016/j.bcp.2014.07.017. [DOI] [PubMed] [Google Scholar]
- 137.Fang J., Sawa T., Akaike T., Greish K., Maeda H. Enhancement of chemotherapeutic response of tumor cells by a heme oxygenase inhibitor, pegylated zinc protoporphyrin. Int. J. Cancer. 2003;109:1–8. doi: 10.1002/ijc.11644. [DOI] [PubMed] [Google Scholar]
- 138.Smith P.S., Zhao W., Spitz D.R., Robbins M.E. Inhibiting catalase activity sensitizes 36B10 rat glioma cells to oxidative stress. Free Radic. Biol. Med. 2007;42:787–797. doi: 10.1016/j.freeradbiomed.2006.11.032. [DOI] [PubMed] [Google Scholar]
- 139.Kilic U., Kilic E., Tuzcu Z., Ozercan I.H., Yilmaz O., Sahin F., Kazim S. Melatonin suppresses cisplatin-induced nephrotoxicity via activation of Nrf-2/HO-1 pathway. Nutr. Metab. 2013;10:7. doi: 10.1186/1743-7075-10-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Surh Y.-J., Kundu J.K., Na H.-K. Nrf2 as a Master Redox Switch in Turning on the Cellular Signaling Involved in the Induction of Cytoprotective Genes by Some Chemopreventive Phytochemicals. Planta Med. 2008;74:1526–1539. doi: 10.1055/s-0028-1088302. [DOI] [PubMed] [Google Scholar]
- 141.Fiedor J., Burda K. Potential Role of Carotenoids as Antioxidants in Human Health and Disease. Nutrients. 2014;6:466–488. doi: 10.3390/nu6020466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Sak K. Cytotoxicity of dietary flavonoids on different human cancer types. Pharmacogn. Rev. 2014;8:122–146. doi: 10.4103/0973-7847.134247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Tavsan Z., Kayali H.A. Flavonoids showed anticancer effects on the ovarian cancer cells: Involvement of reactive oxygen species, apoptosis, cell cycle and invasion. Biomed. Pharmacother. 2019;116:109004. doi: 10.1016/j.biopha.2019.109004. [DOI] [PubMed] [Google Scholar]
- 144.Joshi G., Sultana R., Tangpong J., Cole M.P., Clair D.K.S., Vore M., Estus S., Butterfield D.A. Free radical mediated oxidative stress and toxic side effects in brain induced by the anti-cancer drug adriamycin: Insight into chemobrain. Free Radic. Res. 2005;39:1147–1154. doi: 10.1080/10715760500143478. [DOI] [PubMed] [Google Scholar]
- 145.Berthiaume J.M., Wallace K.B. Adriamycin-induced oxidative mitochondrial cardiotoxicity. Cell Biol. Toxicol. 2006;23:15–25. doi: 10.1007/s10565-006-0140-y. [DOI] [PubMed] [Google Scholar]
- 146.Zhao L., Qi Y., Xu L., Tao X., Han X., Yin L., Peng J. MicroRNA-140-5p aggravates doxorubicin-induced cardiotoxicity by promoting myocardial oxidative stress via targeting Nrf2 and Sirt2. Redox Biol. 2017;15:284–296. doi: 10.1016/j.redox.2017.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Gilliam L.A., St Clair D.K. Chemotherapy-Induced Weakness and Fatigue in Skeletal Muscle: The Role of Oxidative Stress. Antioxid. Redox Signal. 2011;15:2543–2563. doi: 10.1089/ars.2011.3965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Visacri M.B., Quintanilha J.C., Sousa V.M., Amaral L.S., Ambrósio R.F.L., Calonga L., Curi S.F.B.B., Leme M.F.T., Chone C.T., Altemani J.M.C., et al. Can acetylcysteine ameliorate cisplatin-induced toxicities and oxidative stress without decreasing antitumor efficacy? A randomized, double-blind, placebo-controlled trial involving patients with head and neck cancer. Cancer Med. 2019;8:2020–2030. doi: 10.1002/cam4.2072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zhang K., Weng H., Yang J., Wu C. Protective effect of Liuwei Dihuang Pill on cisplatin-induced reproductive toxicity and genotoxicity in male mice. J. Ethnopharmacol. 2019;247:112269. doi: 10.1016/j.jep.2019.112269. [DOI] [PubMed] [Google Scholar]
- 150.Kulhan N.G., Kulhan M., Turkler C., Ata N., Kiremitli T., Kiremitli S., Cimen F.K., Suleyman H., Toprak V. Effect of lycopene on oxidative ovary-damage induced by cisplatin in rats. Gen. Physiol. Biophys. 2019;38:253–258. doi: 10.4149/gpb_2019006. [DOI] [PubMed] [Google Scholar]
- 151.Lu C.C., Meistrich M.L. Cytotoxic effects of chemotherapeutic drugs on mouse testis cells. Cancer Res. 1979;39:3575–3582. [PubMed] [Google Scholar]
- 152.Howell S.J., Radford J.A., Ryder W., Shalet S.M. Testicular Function After Cytotoxic Chemotherapy: Evidence of Leydig Cell Insufficiency. J. Clin. Oncol. 1999;17:1493–1498. doi: 10.1200/JCO.1999.17.5.1493. [DOI] [PubMed] [Google Scholar]
- 153.Gerl A., Mühlbayer D., Hansmann G., Mraz W., Hiddemann W. The impact of chemotherapy on Leydig cell function in long term survivors of germ cell tumors. Cancer. 2001;91:1297–1303. doi: 10.1002/1097-0142(20010401)91:7<1297::AID-CNCR1132>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- 154.Bedoschi G., Navarro P.A., Oktay K. Chemotherapy-induced damage to ovary: Mechanisms and clinical impact. Future Oncol. 2016;12:2333–2344. doi: 10.2217/fon-2016-0176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Molina J.R., Barton D.L., Loprinzi C.L. Chemotherapy-induced ovarian failure: Manifestations and management. Drug Saf. 2005;28:401–416. doi: 10.2165/00002018-200528050-00004. [DOI] [PubMed] [Google Scholar]
- 156.Yang H., Xie Y., Yang D., Ren D. Oxidative stress-induced apoptosis in granulosa cells involves JNK, p53 and Puma. Oncotarget. 2017;8:25310–25322. doi: 10.18632/oncotarget.15813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Arnon J., Meirow D., Lewis-Roness H., Ornoy A. Genetic and teratogenic effects of cancer treatments on gametes and embryos. Hum. Reprod. Update. 2001;7:394–403. doi: 10.1093/humupd/7.4.394. [DOI] [PubMed] [Google Scholar]
- 158.Hales B.F., Barton T.S., Robaire B. Impact of Paternal Exposure to Chemotherapy on Offspring in the Rat. J. Natl. Cancer Inst. Monogr. 2005:28–31. doi: 10.1093/jncimonographs/lgi028. [DOI] [PubMed] [Google Scholar]
- 159.Signorello L.B., Mulvihill J.J., Green D.M., Munro H.M., Stovall M., Weathers R.E., Mertens A.C., Whitton J.A., Robison L.L., Boice J.D. Congenital Anomalies in the Children of Cancer Survivors: A Report From the Childhood Cancer Survivor Study. J. Clin. Oncol. 2012;30:239–245. doi: 10.1200/JCO.2011.37.2938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Yimit A., Adebali O., Sancar A., Jiang Y. Differential damage and repair of DNA-adducts induced by anti-cancer drug cisplatin across mouse organs. Nat. Commun. 2019;10:309. doi: 10.1038/s41467-019-08290-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Yang Z., Schumaker L.M., Egorin M.J., Zuhowski E.G., Guo Z., Cullen K.J. Cisplatin preferentially binds mitochondrial DNA and voltage-dependent anion channel protein in the mito-chondrial membrane of head and neck squamous cell carcinoma: Possible role in apoptosis. Clin. Cancer Res. 2006;12:5817–5825. doi: 10.1158/1078-0432.CCR-06-1037. [DOI] [PubMed] [Google Scholar]
- 162.Türk G., Ateşşahin A., Sönmez M., Ceribasi A.O., Yüce A. Improvement of cisplatin-induced injuries to sperm quality, the oxidant-antioxidant system, and the histologic structure of the rat testis by ellagic acid. Fertil. Steril. 2008;89:1474–1481. doi: 10.1016/j.fertnstert.2007.04.059. [DOI] [PubMed] [Google Scholar]
- 163.Fouad A.A., Qutub H.O., Fouad A.E.A., Audeh A.M., Al-Melhim W.N. Epigallocatechin-3-gallate counters cisplatin toxicity of rat testes. Pharm. Biol. 2017;55:1710–1714. doi: 10.1080/13880209.2017.1322618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Wang T.-E., Lai Y.-H., Yang K.-C., Lin S.-J., Chen C.-L., Tsai P.-S. Counteracting Cisplatin-Induced Testicular Damages by Natural Polyphenol Constituent Honokiol. Antioxidants. 2020;9:723. doi: 10.3390/antiox9080723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.García M.M.S., Acquier A., Suarez G., Gomez N.V., Gorostizaga A., Mendez C.F., Paz C. Cisplatin inhibits testosterone synthesis by a mechanism that includes the action of reactive oxygen species (ROS) at the level of P450scc. Chem. Interact. 2012;199:185–191. doi: 10.1016/j.cbi.2012.08.012. [DOI] [PubMed] [Google Scholar]
- 166.Afsar T., Razak S., Khan M.R., Almajwal A. Acacia hydaspica ethyl acetate extract protects against cisplatin-induced DNA damage, oxidative stress and testicular injuries in adult male rats. BMC Cancer. 2017;17:883. doi: 10.1186/s12885-017-3898-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Aksu E.H., Kandemir F.M., Özkaraca M., Ömür A.D., Küçükler S., Çomaklı S. Rutin ameliorates cisplatin-induced reproductive damage via suppression of oxidative stress and apoptosis in adult male rats. Andrologia. 2016;49:e12593. doi: 10.1111/and.12593. [DOI] [PubMed] [Google Scholar]
- 168.Qin Y., Iwase A., Murase T., Bayasula, Ishida C., Kato N., Nakamura T., Osuka S., Takikawa S., Goto M., et al. Protective effects of mangafodipir against chemotherapy-induced ovarian damage in mice. Reprod. Biol. Endocrinol. 2018;16:106. doi: 10.1186/s12958-018-0426-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Gouveia B.B., Barberino R.D.S., Silva R.L.D.S., Lins T.L.B.G., Guimarães V.D.S., Monte A.P.O.D., Palheta R.C., De Matos M.H.T. Involvement of PTEN and FOXO3a Proteins in the Protective Activity of Protocatechuic Acid Against Cisplatin-Induced Ovarian Toxicity in Mice. Reprod. Sci. 2020;28:865–876. doi: 10.1007/s43032-020-00305-4. [DOI] [PubMed] [Google Scholar]
- 170.Li X., Yang S., Lv X., Sun H., Weng J., Liang Y., Zhou D. The mechanism of mesna in protection from cisplatin-induced ovarian damage in female rats. J. Gynecol. Oncol. 2013;24:177–185. doi: 10.3802/jgo.2013.24.2.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Özcan P., Fıçıcıoğlu C., Yıldırım Ö.K., Özkan F., Akkaya H., Aslan İ. Protective effect of resveratrol against oxidative damage to ovarian reserve in female Sprague-Dawley rats. Reprod. Biomed. Online. 2015;31:410. doi: 10.1016/j.rbmo.2015.06.007. [DOI] [PubMed] [Google Scholar]
- 172.Altuner D., Gulaboglu M., Yapca O.E., Cetin N. The Effect of Mirtazapine on Cisplatin-Induced Oxidative Damage and Infertility in Rat Ovaries. Sci. World J. 2013;2013:627240. doi: 10.1155/2013/327240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.El-Maddawy Z.K., Abd El Naby W.S.H. Protective effects of zinc oxide nanoparticles against doxorubicin induced testicular toxicity and DNA damage in male rats. Toxicol. Res. 2019;8:654–662. doi: 10.1039/c9tx00052f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Aksu E.H., Kandemir F.M., Yıldırım S., Küçükler S., Dörtbudak M.B., Çağlayan C., Benzer F. Palliative effect of curcumin on doxorubicin-induced testicular damage in male rats. J. Biochem. Mol. Toxicol. 2019;33:e22384. doi: 10.1002/jbt.22384. [DOI] [PubMed] [Google Scholar]
- 175.Kabel A.M. Zinc/alogliptin combination attenuates testicular toxicity induced by doxorubicin in rats: Role of oxidative stress, apoptosis and TGF-β1/NF-κB signaling. Biomed. Pharm. 2018;97:439–449. doi: 10.1016/j.biopha.2017.10.144. [DOI] [PubMed] [Google Scholar]
- 176.Cabral R.E.L., Mendes T.B., Vendramini V., Miraglia S.M. Carnitine partially improves oxidative stress, acrosome integrity, and reproductive competence in doxo-rubicin-treated rats. Andrology. 2018;6:236–246. doi: 10.1111/andr.12426. [DOI] [PubMed] [Google Scholar]
- 177.Badkoobeh P., Parivar K., Kalantar S.M., Hosseini S.D., Salabat A. Effect of nano-zinc oxide on doxorubicin- induced oxidative stress and sperm disorders in adult male Wistar rats. Iran. J. Reprod. Med. 2013;11:355–364. [PMC free article] [PubMed] [Google Scholar]
- 178.Yang C.-C., Chen Y.-T., Chen C.-H., Chiang J.Y., Zhen Y.-Y., Yip H.-K. Assessment of doxorubicin-induced mouse testicular damage by the novel second-harmonic generation microscopy. Am. J. Transl. Res. 2017;9:5275–5288. [PMC free article] [PubMed] [Google Scholar]
- 179.Erdem G.E., Tektemur N.K., Tektemur A. Alpha-lipoic acid may ameliorate testicular damage by targeting dox-induced altered antioxidant parameters, mitofusin-2 and apoptotic gene expression. Andrology. 2021;53:e13990. doi: 10.1111/and.13990. [DOI] [PubMed] [Google Scholar]
- 180.Abdelaziz M.H., El-Din E.Y.S., El-Dakdoky M., Ahmed T.A. The impact of mesenchymal stem cells on doxorubicin-induced testicular toxicity and progeny outcome of male prepubertal rats. Birth Defects Res. 2019;111:906–919. doi: 10.1002/bdr2.1535. [DOI] [PubMed] [Google Scholar]
- 181.Niringiyumukiza J.D., Cai H., Chen L., Li Y., Wang L., Zhang M., Xu X., Xiang W. Protective properties of glycogen synthase kinase-3 inhibition against doxorubicin-induced oxi-dative damage to mouse ovarian reserve. Biomed. Pharmacother. 2019;116:108963. doi: 10.1016/j.biopha.2019.108963. [DOI] [PubMed] [Google Scholar]
- 182.Han J., Wang H., Zhang T., Chen Z., Zhao T., Lin L., Xia G., Wang C. Resveratrol attenuates doxorubicin-induced meiotic failure through inhibiting oxidative stress and apoptosis in mouse oocytes. Aging. 2020;12:7717–7728. doi: 10.18632/aging.103061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Gonzalez D.F., Young F. Gamma Tocopherol Reduced Chemotherapeutic-Induced ROS in an Ovarian Granulosa Cell Line, But Not in Breast Cancer Cell Lines In Vitro. Antioxidants. 2020;9:51. doi: 10.3390/antiox9010051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Fabbri R., Macciocca M., Vicenti R., Caprara G., Piccinni M., Paradisi R., Terzano P., Papi A., Seracchioli R. Epigallocatechin-3-gallate inhibits doxorubicin-induced inflammation on human ovarian tissue. Biosci. Rep. 2019;39:BSR20181424. doi: 10.1042/BSR20181424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Richter K., Kietzmann T. Reactive oxygen species and fibrosis: Further evidence of a significant liaison. Cell Tissue Res. 2016;365:591–605. doi: 10.1007/s00441-016-2445-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Holland B.C., Shetty Z., Alanee S. The effect of targeted therapy for genitourinary malignancies on sexual function and fer-tility. Curr. Urol. Rep. 2017;18:65. doi: 10.1007/s11934-017-0704-9. [DOI] [PubMed] [Google Scholar]
- 187.Lorenzi E., Simonelli M., Santoro A. Infertility risk and teratogenicity of molecularly targeted anticancer therapy: A chal-lenging issue. Crit. Rev. Oncol. Hematol. 2016;107:1–13. doi: 10.1016/j.critrevonc.2016.08.005. [DOI] [PubMed] [Google Scholar]
- 188.Lioni M., Noma K., Snyder A., Klein-Szanto A., Diehl J.A., Rustgi A.K., Herlyn M., Smalley K.S.M. Bortezomib induces apoptosis in esophageal squamous cell carcinoma cells through activation of the p38 mi-togen-activated protein kinase pathway. Mol. Cancer Ther. 2008;7:2866–2875. doi: 10.1158/1535-7163.MCT-08-0391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Hou M., Eriksson E., Svechnikov K., Jahnukainen K., Söder O., Meinhardt A., Sävendahl L. Bortezomib treatment causes long-term testicular dysfunction in young male mice. Mol. Cancer. 2014;13:155. doi: 10.1186/1476-4598-13-155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Li W., Fu J., Zhang S., Zhao J., Xie N., Cai G. The proteasome inhibitor bortezomib induces testicular toxicity by upregulation of oxidative stress, AMP-activated protein kinase (AMPK) activation and deregulation of germ cell development in adult murine testis. Toxicol. Appl. Pharmacol. 2015;285:98–109. doi: 10.1016/j.taap.2015.04.001. [DOI] [PubMed] [Google Scholar]
- 191.D’Souza U.J. Toxic effects of 5-Fluorouracil on sperm count in wistar rats. Malays. J. Med. Sci. 2003;10:43–45. [PMC free article] [PubMed] [Google Scholar]
- 192.Focaccetti C., Bruno A., Magnani E., Bartolini D., Principi E., Dallaglio K., Bucci E.O., Finzi G., Sessa F., Noonan D.M., et al. Effects of 5-Fluorouracil on Morphology, Cell Cycle, Proliferation, Apoptosis, Autophagy and ROS Production in Endothelial Cells and Cardiomyocytes. PLoS ONE. 2015;10:e0115686. doi: 10.1371/journal.pone.0115686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Fideles L.S., Miranda J.A.L., Martins C.S., Barbosa M.L.L., Pimenta H.B., Pimentel P.V.S., Teixeira C.S., Scafuri M.A.S., de Osterno Façanha S., Barreto J.E.F., et al. Role of Rutin in 5-Fluorouracil-Induced Intestinal Mucositis: Prevention of Histological Damage and Re-duction of Inflammation and Oxidative Stress. Molecules. 2020;25:2786. doi: 10.3390/molecules25122786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Bomfin L.E., Braga C.M., Oliveira T.A., Martins C.S., Foschetti D.A., Santos A.A., Costa D.V., Leitão R.F., Brito G.A. 5-Fluorouracil induces inflammation and oxidative stress in the major salivary glands affecting salivary flow and saliva composition. Biochem. Pharmacol. 2017;145:34–45. doi: 10.1016/j.bcp.2017.08.024. [DOI] [PubMed] [Google Scholar]
- 195.Orcutt K.P., Parsons A.D., Sibenaller Z.A., Scarbrough P.M., Zhu Y., Sobhakumari A., Wilke W.W., Kalen A.L., Goswami P., Miller F.J., et al. Erlotinib-Mediated Inhibition of EGFR Signaling Induces Metabolic Oxidative Stress through NOX4. Cancer Res. 2011;71:3932–3940. doi: 10.1158/0008-5472.CAN-10-3425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Chang X., Zhou L., Chen X., Xu B., Cheng Y., Sun S., Fang M., Xiang Y. Impact of Imatinib on the Fertility of Male Patients with Chronic Myelogenous Leukaemia in the Chronic Phase. Target. Oncol. 2017;12:827–832. doi: 10.1007/s11523-017-0521-6. [DOI] [PubMed] [Google Scholar]
- 197.Bouitbir J., Panajatovic M.V., Frechard T., Roos N.J., Krähenbühl S. Imatinib and Dasatinib Provoke Mitochondrial Dysfunction Leading to Oxidative Stress in C2C12 Myotubes and Human RD Cells. Front. Pharmacol. 2020;11:1106. doi: 10.3389/fphar.2020.01106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Salem W., Li K., Krapp C., Ingles S.A., Bartolomei M.S., Chung K., Paulson R.J., Nowak R.A., McGinnis L.K. Imatinib treatments have long-term impact on placentation and embryo survival. Sci. Rep. 2019;9:2535. doi: 10.1038/s41598-019-39134-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Das G., Damotte V., Gelfand J.M., Bevan C., Cree B.A.C., Do L., Green A.J., Hauser S.L., Bove R. Rituximab before and during pregnancy: A systematic review, and a case series in MS and NMOSD. Neurol. Neuroimmunol. Neuroinflamm. 2018;5:e453. doi: 10.1212/NXI.0000000000000453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Pendergraft W.F., McGrath M.M., Murphy A.P., Murphy P., Laliberte K.A., Greene M.F., Niles J.L. Fetal outcomes after rituximab exposure in women with autoimmune vasculitis. Ann. Rheum. Dis. 2013;72:2051–2053. doi: 10.1136/annrheumdis-2013-203833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Al-Mogairen S.M. Safety of rituximab on testicles, a double-blindedcontrolled trial in mice. J. Nat. Sci. Med. 2020;3:66. doi: 10.4103/JNSM.JNSM_38_19. [DOI] [Google Scholar]
- 202.Salvi A., Patki G., Khan E., Asghar M., Salim S. Protective Effect of Tempol on Buthionine Sulfoximine-Induced Mitochondrial Impairment in Hippocampal Derived HT22 Cells. Oxidative Med. Cell. Longev. 2016;2016:5059043. doi: 10.1155/2016/5059043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Takahashi K., Tatsunami R., Oba T., Tampo Y. Buthionine sulfoximine promotes methylglyoxal-induced apoptotic cell death and oxidative stress in en-dothelial cells. Biol. Pharm. Bull. 2010;33:556–560. doi: 10.1248/bpb.33.556. [DOI] [PubMed] [Google Scholar]