Abstract
Background
Given the severe infection, poor prognosis, and the low number of available effective drugs, potential prevention and treatment strategies for COVID-19 need to be urgently developed.
Main body
Herein, we present and discuss the possible protective and therapeutic mechanisms of human microbiota and probiotics based on the previous and recent findings. Microbiota and probiotics consist of mixed cultures of living microorganisms that can positively affect human health through their antiviral, antibacterial, anti-inflammatory, and immunomodulatory effect. In the current study, we address the promising advantages of microbiota and probiotics in decreasing the risk of COVID-19.
Conclusions
Thus, we recommend further studies be conducted for assessing and evaluating the capability of these microbes in the battle against COVID-19.
Keywords: Antiviral, Beneficial microbes, Gut microbiota, Immunomodulatory, Probiotics
Background
Respiratory infections could cause global high rates of morbidity and mortality. The viruses commonly associated with such infections include influenza viruses, parainfluenza viruses, coronaviruses, respiratory syncytial virus, rhinoviruses, and adenoviruses. In December 2019, an outbreak of pneumonia of unknown etiology was reported in Wuhan city in China [1]. WHO later identified this disease as Coronavirus disease (COVID-19) which is caused by a novel coronavirus called Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). As yet, SARS-CoV-2 has become a global pandemic virus causing unprecedented crises regarding health, economy, and high mortality rate [2].
At present, there is no sole medication for the treatment of COVID-19 thus, researchers all over the world are actively engaged to find out appropriate treatment for COVID-19. Using beneficial microbe-based drugs could be a novel approach to be used in the attempts being done for treatment and prevention of COVID-19. The recent research about microbiota has led to an improved understanding of the communities of the commensal microorganisms (including bacteria, fungi, viruses, phages, archaea, and helminths) which live within the human body. Besides the extensively studied gut microbiota, the lung microbiota, which is only considered in recent years, represents an important member of the whole human microbiota [3]. It has been observed that in COVID-19 patients, there is microbial dysbiosis in the gastrointestinal tract (GIT) and lung which could be involved in the severity of the disease [4].
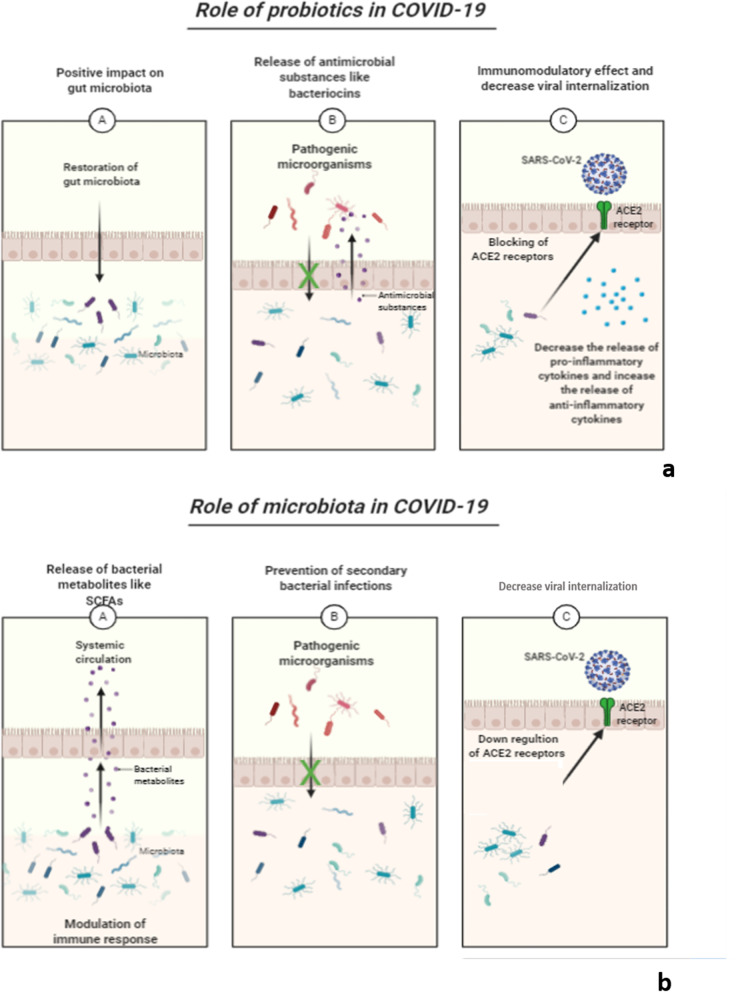
Probiotics are defined as living microorganisms that, when given in appropriate amounts, afford beneficial effects to the host [5]. The potential of probiotics to boost health benefits has been reported as they can regulate allergic reactions, alleviate inflammatory bowel disease, reduce tumor growth in some cancer models, prevent colon cancer, control the levels of blood cholesterol and protect hosts from bacterial and viral infections [6]. Human microbiota and probiotics have anti-inflammatory and immunomodulatory effects that could be beneficial in the treatment of the severely ill COVID-19 patient who always suffers from cytokine storm that results from the production of a large quantity of pro-inflammatory cytokines [7]. Besides, they have an antiviral and antibacterial activity which is necessary for our fight against the SARS-CoV-2 virus [8, 9]. The rationale of using biotherapeutic drugs based on beneficial microbes like human microbiota and probiotics for treatment and prevention of COVID-19 infection is attributed to their antiviral, anti-inflammatory, and immunomodulatory effect, they also can prevent secondary bacterial infections as presented in Fig. 1.
Fig. 1.
Role of a probiotics and b microbiota in treatment of COVID-19
Main text
Gut-lung axis and COVID-19
The alimentary tract hosts a complex group of the highly diverse microbial ecosystem which has a role in ensuring the establishment and persistence of immune homeostasis [10]. In addition to the widely investigated gut microbiota, the microbiota of other sites in the human body, especially the lungs, are crucial for host homeostasis. Interestingly, lung microbiota is now recognized to have an essential role in the physiopathology of many respiratory diseases [11]. Consequently, a group of researchers has investigated if the infection caused by SARS-CoV-2 affects the lung microbiota [12]. They observed a severe microbiota dysbiosis in the lungs of COVID-19 patients, with a high incidence of pathogenic species like Klebsiella oxytoca and Tobacco mosaic virus (TMV) a finding which could contribute to the complications that occur in SARS-CoV-2 infections. From birth and throughout the entire life span, a close correlation exists between the gut and lung microbiota [13]. For example, if the newborns’ diet is modified, the composition of their lung microbiota will be affected, and fecal transplantation in experimental rats can induce changes in their lung microbiota [3].
On the opposite side, the lung microbiota could affect the gut microbiota. In an experimental model, Looft and Allen [14] found that infection with influenza virus triggers an increased abundance of Enterobacteriaceae and decreased proportions of Lactobacilli in the gut. This connection is called the gut-lung axis and the mechanisms mediating this communication are still unclear [15]. Although respiratory distress is a main symptom of COVID-19, this disease is also associated with some other non-classical symptoms such as gastrointestinal symptoms. Noteworthy, patients with gastrointestinal symptoms had more serious respiratory complications. This could be associated with microbial dysbiosis in the lungs and GIT [16].
Human microbiota-virus interaction
Substantial interactions occur between the viruses invading the human body and commensal microbiota leading to certain suppressive outcomes for the viral infection [17]. This is based on the research carried out by Botic et al. who noticed that lactic acid bacteria (LAB) [18], which colonize the human gut, decreased the infectivity of vesicular stomatitis virus by direct binding to the virus, thus they blocked the entry of the viruses to the human cells. Also, Wang and his colleagues showed that Enterococcus faecium, a Gram-positive bacterium living in the human GIT, can prevent the influenza viral infection by direct adsorptive trapping of the viruses [19]. Furthermore, human microbiota can exert antiviral activity by its cellular components or through the production of several metabolites with antimicrobial activity [17].
An extracted cell wall-associated component from Lactobacillus brevis vaginal strain is an example of the antiviral activity of the microbiome cellular components. It has been found that this component potently inhibited the HSV-2 viral replication in an in vitro model [20, 21]. On the other hand, the extracellular matrix-binding protein which is produced by Staphylococcus epidermidis (bacterial commensal found in the human nasal cavity) can stably bind to the influenza virus thus, blocking further viral infection [22]. Microbiota may also have a role in decreasing the entry of SARS-CoV-2. It is well known that the SARS-CoV-2 virus enters human cells by transmembrane spike glycoprotein forming homotrimers expressed on its surface. This spike glycoprotein binds to trans-membrane angiotensin-converting enzyme (ACE2) receptor, which is expressed in different tissues in the human body like lung, kidney, and GIT [23]. Yang and his colleagues [24] studied the effect of microbiota on colonic ACE-2 receptors in a murine model and they noticed that gut microbiota regulated these receptors. Notably, various research articles have speculated the interaction of microbiota with ACE2 receptors in certain diseases like cardiovascular diseases [25] and intestinal inflammation [26, 27].
Anti-inflammatory and immunomodulatory effect of human microbiota
An indirect role of microbiota on viral infection is its anti-inflammatory and immunomodulatory effect. Microbiota, especially gut and lung microbiota have effects on the local immunity [28, 29]. Gut microbiota can trigger the local immune response through interactions with the immune cells expressing pattern recognition receptors (PRRs) (e.g., Toll-like receptors [TLRs]) [30]. They can also activate local dendritic cells through interactions with PRRs [31]. Then the activated dendritic cells travel from the GIT to mesenteric lymph nodes, where they induce the differentiation of the T cells into the effector T cells, mainly regulatory T cells (Tregs) and T helper 17 (Th17) cells. Some of these effector T cells migrate back to the GIT and affect the local immune responses [32]. Tregs can mediate the conversion of the immune system from the pro-inflammatory to the anti-inflammatory state via the release of anti-inflammatory cytokines (like IL-10, TGF-β) [33].
Besides, several microbiota-derived metabolites like short-chain fatty acids (SCFAs) were found to protect the integrity of the GIT barrier against the disrupting effects of the pro-inflammatory cytokines [34]. A vital role of the lung microbiota in both maturation and homeostasis of lung immunity has been revealed over the last few years [29]. Preclinical studies confirmed the impact of lung microbiota on the regulation and maturation of immune cells of the respiratory system [34–37]. On the other side, the gut microbiota has a long-reaching immune impact (systemic effect), mainly on the pulmonary immune system via the mesenteric lymphatic system through which the intact microbiota, their fragments, or metabolites (like SCFAs) may reach the systemic circulation and modulate the immune response of the lung [38]. Many researchers have studied the immunomodulatory impact of SCFAs impact on the pulmonary system [39–41]. They noticed that SCFAs act as signaling molecules on the antigen-presenting cells of the lungs leading to attenuation of the inflammatory and allergic responses. Yin and his colleagues [42] have conducted bacteria research on the segmented filamentous (SFB), members of the gut microbiota, in humans and mice and they noticed that SFB has a significant role in the modulation of the host immune systems.
Microbiota and prevention of secondary bacterial infections
One more important role of the human microbiota is colonization resistance where commensal microbiota protects the host against colonization with pathogenic organisms and inhibits the overgrowth of the pathogenic microbiota members. The postulated mechanisms of action for colonization resistance are: (1) directly by the interaction between human microbiota and different pathogens in competition for the shared niches and nutrients, and (2) enhancement of the host defense ability by the human microbiota to suppress pathogens (as discussed before). The dominant non-pathogenic microbiota plays an important role in both occupying the niche and inhibiting the colonization and growth of different pathogens [33]. Yet, if microbiota is disturbed for any reason, a decrease in the non-pathogenic dominant microbiota members decreases the capacity of colonization resistance, leading to colonization and overgrowth of the opportunistic pathogens in the empty niches. A classic example of this situation is the infection with Clostridium difficile which can cause pseudo-membranous colitis, sepsis, and death in severe cases [43].
Fecal microbiota transfer as an example for microbiome-based biotherapeutic drug
Fecal microbiota transfer (FMT) involves the suspension of the donor stool in certain solutions, homogenization then filtration and finally, it is delivered through upper and/or lower GIT as gelatin capsules after centrifugation [44]. FMT is approved as a therapy for the treatment of recurrent infection with Clostridium difficile [45, 46]. It is now under research to be used in the treatment of some other diseases like metabolic disorders [47] and hepatic encephalopathy [48]. The main benefit of the use of FMT is to restore gut health and to reverse the gut dysbiosis that is induced by either antibiotic [49] or microbial infection [50] like in the case of COVID-19 infection. Thus, depending on the previously mentioned associations between gut microbiota and respiratory diseases, FMT could be effective in the treatment of COVID-19 patients.
Immunomodulatory effect of probiotics
The effectiveness of probiotics in the treatment and prevention of a variety of diseases have been investigated like the prevention of allergy and certain intestinal diseases, in addition to the treatment of gastrointestinal diseases and certain types of cancers [51]. The health benefits conferred from probiotics are attributed to their effects on the immune system. Immunomodulators can be classified into immunostimulants or immunosuppressants [52]. The immunomodulatory effect of probiotics has been identified via the release of cytokines, interleukins, interferons, transforming growth factors, tumor necrosis factors (TNF), and chemokines from different immune cells such as lymphocytes, macrophages, mast cells, epithelial cells, granulocytes, and dendritic cells which boost the regulation of innate and adaptive immune system [53]. Several types of genera of bacteria have been identified as probiotics, among them, Lactobacillus and Bifidobacterium have been consumed as a part of fermented foods like those in dietary supplements [54]. It was found that L. reuteri and L. casei, can stimulate the production of IFN-gamma, and activate the pro-inflammatory Th1 cells [55]. Also, the oral administration of B. infantis into mice was noticed to stimulate dendritic cells that can suppress the biased responses of Th2 cells and stimulate the pro-inflammatory responses of Th1 that are required for virus elimination [56]. Besides, probiotics L. acidophilus, L. gasseri, L. delbrueckii, and B. bifidum strains can induce the production of IFN-alpha by monocytes [57]. The probiotic L. paracasei was found to increase the release of TNF-alpha, IL-6, IL-8 of the human monocyte cell line that is required for the antiviral effect [58].
Antiviral activity of probiotics
A growing interest in the effectiveness of probiotics as viral inhibitors has emerged in the treatment of diseases and infections associated with HIV [59]. Probiotics have exhibited a potential role as antiviral agents against several groups of viruses like rotavirus [60], coxsackieviruses, enterovirus, [61], and herpes simplex [62]. Interestingly, the exo-polysaccharides produced by Lactobacillus plantarum were shown to have an antiviral effect against human rotavirus-induced diarrhea [63] and transmissible gastroenteritis virus [64]. Probiotics have antiviral activity against many respiratory viruses like influenza and syncytial viruses via boosting the immunity of individuals through activating the secretion of IgA and enhancing the activity of neutrophils, natural killer cells, and macrophages [65, 66].
ACE inhibitory activity of probiotics
As previously mentioned, the entry of SARS-CoV-2 is facilitated by binding to ACE2 receptors and this interaction, when occurs in the gut, may be responsible for the GIT symptoms, which are reported in 12–60% cases of COVID-19 and it could be associated with increased disease severity [67]. In an interesting study [68], four metabolic products of Lactobacillus plantarum; Plantaricin BN, Plantaricin W, Plantaricin D, Plantaricin JLA-9 have been selected to design computer-based antiviral computational product for COVID-19. This study aimed to target and block the residual binding protein (RBP) on ACE2 receptor proteins by selected probiotics along with RNA-dependent RNA polymerase (RdRp). Three metabolic products of L. plantarum, significantly interacted with RdRp and ACE2, recording the lowest binding energy. These results suggest that probiotics could be used as a potential ACE2 receptor blocker, hence their importance in treating COVID-19 [68].
Antimicrobial substances produced by probiotics
Probiotics like LAB can produce antimicrobial substances such as bacteriocins that have a broad spectrum of antagonistic effects against many bacterial pathogens [69]. Bacteriocins have been considered as promising antimicrobial compounds with potential applications in the food, health, and veterinary sectors [70]. Novel applications of LAB bacteriocins are steadily increasing, with horizons of more fascinating roles to be played by these agents in the future in anti-quorum sensing strategies and site-specific drug delivery [71]. Additionally, LAB strains often produce polymeric substances such as exopolysaccharides (EPS) that are proven by several researchers to have the ability to express antagonistic effects against pathogenic bacteria [72–77]. LAB can also produce biosurfactant agents which have shown a broad range of antimicrobial activity against bacterial pathogens as well as anti-adhesion properties that can reduce the pathogens' adhesion to the gastric wall membrane [69]. This ability of probiotics is important to fight against the secondary bacterial infections that commonly occur in severely ill COVID-19 patients.
Impact of probiotics on gut microbiota and its link with COVID-19
Probiotics exert their beneficial effects via various mechanisms including treatment and restoration of gut microbiota, enhancement of intestinal barrier function, competition with pathogens for adhesion to gut epithelium and nutrition, suppression of opportunistic pathogens, production of antimicrobial substances, activation of mucosal immunity, and modulation of the innate and adaptive immune response. These actions of probiotics have been proven in various experimental and clinical studies [78]. As early mentioned, the respiratory viral infection is known to cause a disturbance in the gut microbiota, as in cases of COVID-19 infection, the gut microbiota is altered with severe hypoxemia. Some probiotic strains may restore the gut microbiota, maintain a healthy gut-lung axis, reduce translocation of pathogenic bacteria across gut mucosa and reduce the chances of secondary bacterial infection [79]. The most commonly used species in probiotics preparations are Lactobacillus sp, Bifidibacterium sp, Enterococcus sp, Streptococcus sp, Bacillus sp, and Pediococcus sp. Table 1 illustrated examples of different probiotics microbes, mechanisms of action, and their beneficial health effects [80–83]. It was reported that most of the patients with relatively mild symptoms of COVID-19 had received probiotics along with the established treatment protocols and this is in agreement with COVID-19 infection affecting the normal bacterial balance in the human intestine based on the observation of reduced numbers of Lactobacillus and Bifidobacterium species in patients with COVID-19 [84].
Table 1.
The commonly used species in probiotics, mechanisms of action, and effects
| Probiotics | Examples | Mechanism | Beneficial health effects |
|---|---|---|---|
| Lactobacillus sp |
L acidophilus L delbrueckii subsp bulgaricus L casei L cellobiosis L fermentum L curvatus L returi L plantarum L brevis L lactis |
Increasing mucin production via Increased expression of MUC 2 |
Improved mucosal immune function, mucin secretion, and disease prevention Adhering to human intestinal cells and balancing intestinal microflora mproved lactose digestion and decreased diarrhea |
| Bifidibacterium sp. |
B bifidum B adolescentis B thermophilum B animalis B infantis B longum |
Cytokine production Blocking proinflammatory |
Used in treating rotavirus diarrhea, balancing intestinal microflora, and treating viral diarrhea |
| Enterococcus sp. |
E faecalis E faecium |
Prevention of pathogenic strains from adhering to epithelial cells | Decreased duration of acute diarrhea from gastroenteritis |
| Streptococcus sp. |
S cremoris S salivarius S diaacetylactis S intermedius |
Immune modulation by attenuating IL-8 secretion or blocking the degradation of the counter-regulatory factor IκB |
Shortening of duration of acute gastroenteritis Prevention and treatment of C difficile diarrhea and traveler’s diarrhea |
| Bacillus sp. |
B licheniformis B subtilis B polyfermenticus B coagulans B laterosporus B polymyxa B pumilus B clausii B cereus var toyoi |
Enhanced antibody production Enhanced phagocytic activity |
Used as a prophylactic, and in prevention of GIT infections |
| Pediococcus sp. | P acidilactici |
Blocking proinflammatory molecules Increasing mucosal immunity |
Enhanced immune responses against infectious coccidioidal diseases |
Conclusion
Based on the aforementioned impacts of both microbiota and probiotics, we strongly believe that microbiota and probiotics-based drugs have antiviral potential which deserves more investigation of their role in the prevention and treatment of COVID-19. Preclinical and clinical trials should be carried out in the near future to get benefits from these beneficial bacteria in the treatment of COVID-19 pandemic. In addition, the COVID-19 prevention guidelines should include these bacteria as an important means to fight against COVID-19 infection. We will be so excited to see how they will be applied in the clinical practice and afford therapeutic benefits to patients and high-risk individuals.
Acknowledgements
None.
Abbreviations
- SARS-CoV-2
Severe Acute Respiratory Syndrome Coronavirus 2
- TMV
Tobacco mosaic virus
- LAB
Lactic acid bacteria
- SCFAs
Short-chain fatty acids
- ACE2
Angiotensin-converting enzyme
- TNF
Tumor necrosis factors
- PRRs
Pattern recognition receptors
- FMT
Fecal microbiota transfer
- RdRp
RNA-dependent RNA polymerase
- GIT
Gasterointestinal tract
- MUC 2
Mucin
Authors' contributions
EE and WAN writed and edited the manuscript. Both authors read and approved the final manuscript.
Funding
No fund received.
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Segal JP, Mak JW, Mullish BH, Alexander JL, Ng SC, Marchesi JR (2020) The gut microbiome: an under-recognized contributor to the COVID-19 pandemic? Ther Adv Gastroenterol 13:1756284820974914. 10.1177/1756284820974914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zeppa SD, Agostini D, Piccoli G, Stocchi V, Sestili P (2020) Gut microbiota status in COVID-19: an unrecognized player? Front Cell Infect Microbiol. 10.3389/fcimb.2020.576551 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Enaud R, Prevel R, Ciarlo E, Beaufils F, Wieërs G, Guery B et al (2020) The gut-lung axis in health and respiratory diseases: a place for inter-organ and inter-kingdom crosstalks. Front Cell Infect Microbiol 10:9. 10.3389/fcimb.2020.00009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Vodnar D-C, Mitrea L, Teleky B-E, Szabo K, Călinoiu L-F, Nemeş S-A et al (2020) Coronavirus disease (Covid-19) caused by (sars-cov-2) infections: a real challenge for human gut microbiota. Front Cell Infect Microbiol 10:786. 10.3389/fcimb.2020.575559 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nicolae C, Dan D, Lavinia S, Ioan L, Călin J, Oana M (2010) Probiotics–identification and ways of actions. Innov Romanian Food Biotechnol 6:1–11 [Google Scholar]
- 6.Hajavi J, Esmaeili SA, Varasteh AR, Vazini H, Atabati H, Mardani F et al (2019) The immunomodulatory role of probiotics in allergy therapy. J Cell Physiol 234(3):2386–2398. 10.1002/jcp.27263 [DOI] [PubMed] [Google Scholar]
- 7.Ragab D, Salah Eldin H, Taeimah M, Khattab R, Salem R (2020) The COVID-19 cytokine storm; what we know so far. Front Immunol 11:1446. 10.3389/fimmu.2020.01446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Baghbani T, Nikzad H, Azadbakht J, Izadpanah F, Kashani HH (2020) Dual and mutual interaction between microbiota and viral infections: a possible treat for COVID-19. Microb Cell Fact 19(1):1–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sundararaman A, Ray M, Ravindra P, Halami PM (2020) Role of probiotics to combat viral infections with emphasis on COVID-19. Appl Microbiol Biotechnol. 10.1007/s00253-020-10832-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Blander JM, Longman RS, Iliev ID, Sonnenberg GF, Artis D (2017) Regulation of inflammation by microbiota interactions with the host. Nat Immunol 18(8):851–860. 10.1038/ni.3780 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vandenborght L, Enaud R, Coron N, Denning D, Delhaes L (2019) From culturomics to metagenomics: the mycobiome in chronic respiratory diseases. Lung Microbiome. 10.1183/2312508X.10015918 [Google Scholar]
- 12.Han Y, Jia Z, Shi J, Wang W, He K (2020) The active lung microbiota landscape of COVID-19 patients. medRxiv. 10.1101/2020.08.20.20144014
- 13.Grier A, McDavid A, Wang B, Qiu X, Java J, Bandyopadhyay S et al (2018) Neonatal gut and respiratory microbiota: coordinated development through time and space. Microbiome 6(1):1–19. 10.1186/s40168-018-0566-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Looft T, Allen HK (2012) Collateral effects of antibiotics on mammalian gut microbiomes. Gut microbes 3(5):463–467. 10.4161/gmic.21288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Barcik W, Boutin RC, Sokolowska M, Finlay BB (2020) The role of lung and gut microbiota in the pathology of asthma. Immunity 52(2):241–255. 10.1016/j.immuni.2020.01.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gou W, Fu Y, Yue L, Chen G-d, Cai X, Shuai M et al (2020) Gut microbiota may underlie the predisposition of healthy individuals to COVID-19. MedRxiv. 10.1101/2020.04.22.20076091
- 17.Li N, Ma W-T, Pang M, Fan Q-L, Hua J-L (2019) The commensal microbiota and viral infection: a comprehensive review. Front Immunol 10:1551. 10.3389/fimmu.2019.01551 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Botić T, Danø T, Weingartl H, Cencič A (2007) A novel eukaryotic cell culture model to study antiviral activity of potential probiotic bacteria. Int J Food Microbiol 115(2):227–234. 10.1016/j.ijfoodmicro.2006.10.044 [DOI] [PubMed] [Google Scholar]
- 19.Wang Z, Chai W, Burwinkel M, Twardziok S, Wrede P, Palissa C et al (2013) Inhibitory influence of Enterococcus faecium on the propagation of swine influenza A virus in vitro. PLoS ONE 8(1):e53043. 10.1371/journal.pone.0053043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mastromarino P, Cacciotti F, Masci A, Mosca L (2011) Antiviral activity of Lactobacillus brevis towards herpes simplex virus type 2: role of cell wall associated components. Anaerobe 17(6):334–336. 10.1016/j.anaerobe.2011.04.022 [DOI] [PubMed] [Google Scholar]
- 21.Tuyama AC, Cheshenko N, Carlucci MJ, Li J-H, Goldberg CL, Waller DP et al (2006) ACIDFORM inactivates herpes simplex virus and prevents genital herpes in a mouse model: optimal candidate for microbicide combinations. J Infect Dis 194(6):795–803. 10.1086/506948 [DOI] [PubMed] [Google Scholar]
- 22.Chen H-W, Liu P-F, Liu Y-T, Kuo S, Zhang X-Q, Schooley RT et al (2016) Nasal commensal Staphylococcus epidermidis counteracts influenza virus. Sci Rep 6(1):1–12. 10.1038/srep27870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sharma RK, Stevens BR, Obukhov AG, Grant MB, Oudit GY, Li Q et al (2020) ACE2 (Angiotensin-Converting Enzyme 2) in cardiopulmonary diseases: ramifications for the control of SARS-CoV-2. Hypertension 76(3):651–661. 10.1161/HYPERTENSIONAHA.120.15595 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yang T, Chakraborty S, Saha P, Mell B, Cheng X, Yeo J-Y et al (2020) Gnotobiotic rats reveal that gut microbiota regulates colonic mRNA of Ace2, the receptor for SARS-CoV-2 infectivity. Hypertension 76(1):e1–e3. 10.1161/HYPERTENSIONAHA.120.15360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cole-Jeffrey CT, Liu M, Katovich MJ, Raizada MK, Shenoy V (2015) ACE2 and microbiota: emerging targets for cardiopulmonary disease therapy. J Cardiovasc Pharmacol 66(6):540. 10.1097/FJC.0000000000000307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Koester ST, Li N, Lachance DM, Morella NM, Dey N (2021) Variability in digestive and respiratory tract Ace2 expression is associated with the microbiome. PLoS ONE 16(3):e0248730. 10.1038/nature11228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hashimoto T, Perlot T, Rehman A, Trichereau J, Ishiguro H, Paolino M et al (2012) ACE2 links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature 487(7408):477–81. 10.1038/nature11228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Elson CO, Alexander KL (2015) Host-microbiota interactions in the intestine. Dig Dis 33(2):131–136. 10.1159/000369534 [DOI] [PubMed] [Google Scholar]
- 29.Dickson RP, Erb-Downward JR, Falkowski NR, Hunter EM, Ashley SL, Huffnagle GB (2018) The lung microbiota of healthy mice are highly variable, cluster by environment, and reflect variation in baseline lung innate immunity. Am J Respir Crit Care Med 198(4):497–508. 10.1164/rccm.201711-2180OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Skelly AN, Sato Y, Kearney S, Honda K (2019) Mining the microbiota for microbial and metabolite-based immunotherapies. Nat Rev Immunol 19(5):305–323. 10.1038/s41577-019-0144-5 [DOI] [PubMed] [Google Scholar]
- 31.Fulde M, Sommer F, Chassaing B, van Vorst K, Dupont A, Hensel M et al (2018) Neonatal selection by Toll-like receptor 5 influences long-term gut microbiota composition. Nature 560(7719):489–493. 10.1038/s41586-018-0395-5 [DOI] [PubMed] [Google Scholar]
- 32.Lawley TD, Walker AW (2013) Intestinal colonization resistance. Immunology 138(1):1–11. 10.1111/j.1365-2567.2012.03616.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kho ZY, Lal SK (2018) The human gut microbiome—a potential controller of wellness and disease. Front Microbiol 9:1835. 10.3389/fmicb.2018.01835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen T, Kim CY, Kaur A, Lamothe L, Shaikh M, Keshavarzian A et al (2017) Dietary fibre-based SCFA mixtures promote both protection and repair of intestinal epithelial barrier function in a Caco-2 cell model. Food Funct 8(3):1166–1173. 10.1039/c6fo01532h [DOI] [PubMed] [Google Scholar]
- 35.Gollwitzer ES, Saglani S, Trompette A, Yadava K, Sherburn R, McCoy KD et al (2014) Lung microbiota promotes tolerance to allergens in neonates via PD-L1. Nat Med 20(6):642–647. 10.1038/nm.3568 [DOI] [PubMed] [Google Scholar]
- 36.Allie SR, Bradley JE, Mudunuru U, Schultz MD, Graf BA, Lund FE et al (2019) The establishment of resident memory B cells in the lung requires local antigen encounter. Nat Immunol 20(1):97–108. 10.1038/s41590-018-0260-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Russell SL, Gold MJ, Hartmann M, Willing BP, Thorson L, Wlodarska M et al (2012) Early life antibiotic-driven changes in microbiota enhance susceptibility to allergic asthma. EMBO Rep 13(5):440–447. 10.1038/embor.2012.32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.McAleer JP, Kolls JK (2018) Contributions of the intestinal microbiome in lung immunity. Eur J Immunol 48(1):39–49. 10.1002/eji.201646721 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Anand S, Mande SS (2018) Diet, microbiota and gut-lung connection. Front Microbiol 9:2147. 10.3389/fmicb.2018.02147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cait A, Hughes M, Antignano F, Cait J, Dimitriu P, Maas K et al (2018) Microbiome-driven allergic lung inflammation is ameliorated by short-chain fatty acids. Mucosal Immunol 11(3):785–795. 10.1038/mi.2017.75 [DOI] [PubMed] [Google Scholar]
- 41.Trompette A, Gollwitzer ES, Yadava K, Sichelstiel AK, Sprenger N, Ngom-Bru C et al (2014) Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat Med 20(2):159–166. 10.1038/nm.3444 [DOI] [PubMed] [Google Scholar]
- 42.Yin Y, Wang Y, Zhu L, Liu W, Liao N, Jiang M et al (2013) Comparative analysis of the distribution of segmented filamentous bacteria in humans, mice and chickens. ISME J 7(3):615–621. 10.1038/ismej.2012.128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pruitt RN, Chumbler NM, Rutherford SA, Farrow MA, Friedman DB, Spiller B et al (2012) Structural determinants of Clostridium difficile toxin A glucosyltransferase activity. J Biol Chem 287(11):8013–8020. 10.1074/jbc.M111.298414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kelly CR, Kahn S, Kashyap P, Laine L, Rubin D, Atreja A et al (2015) Update on fecal microbiota transplantation 2015: indications, methodologies, mechanisms, and outlook. Gastroenterology 149(1):223–237. 10.1053/j.gastro.2015.05.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Papanicolas LE, Choo JM, Wang Y, Leong LE, Costello SP, Gordon DL et al (2019) Bacterial viability in faecal transplants: which bacteria survive? EBioMedicine 41:509–516. 10.1016/j.ebiom.2019.02.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chehoud C, Dryga A, Hwang Y, Nagy-Szakal D, Hollister EB, Luna RA et al (2016) Transfer of viral communities between human individuals during fecal microbiota transplantation. MBio 7(2):e00322-e416. 10.1128/mBio.00322-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Jayasinghe TN, Chiavaroli V, Holland DJ, Cutfield WS, O’Sullivan JM (2016) The new era of treatment for obesity and metabolic disorders: evidence and expectations for gut microbiome transplantation. Front Cell Infect Microbiol 6:15. 10.3389/fcimb.2016.00015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bajaj JS, Kassam Z, Fagan A, Gavis EA, Liu E, Cox IJ et al (2017) Fecal microbiota transplant from a rational stool donor improves hepatic encephalopathy: a randomized clinical trial. Hepatology 66(6):1727–1738. 10.1002/hep.29306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Le Bastard Q, Ward T, Sidiropoulos D, Hillmann BM, Chun CL, Sadowsky MJ et al (2018) Fecal microbiota transplantation reverses antibiotic and chemotherapy-induced gut dysbiosis in mice. Sci Rep 8(1):1–11. 10.1038/s41598-018-24342-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gupta S, Allen-Vercoe E, Petrof E (2016) Fecal microbiota transplantation: in perspective. Ther Adv Gastroenterol 9:229–239. 10.1177/1756283X15607414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kullar R, Johnson S, McFarland LV, Goldstein EJ (2021) Potential roles for probiotics in the treatment of COVID-19 patients and prevention of complications associated with increased antibiotic use. Antibiotics 10(4):408. 10.3390/antibiotics10040408 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Nair A, Chattopadhyay D, Saha B (2019) Plant-derived immunomodulators. New look to phytomedicine. Elsevier, Amsterdam, pp 435–499 [Google Scholar]
- 53.Foligné B, Dewulf J, Breton J, Claisse O, Lonvaud-Funel A, Pot B (2010) Probiotic properties of non-conventional lactic acid bacteria: immunomodulation by Oenococcus oeni. Int J Food Microbiol 140(2–3):136–145. 10.1016/j.ijfoodmicro.2010.04.007 [DOI] [PubMed] [Google Scholar]
- 54.Zhang Z, Lv J, Pan L, Zhang Y (2018) Roles and applications of probiotic Lactobacillus strains. Appl Microbiol Biotechnol 102(19):8135–8143. 10.1007/s00253-018-9217-9 [DOI] [PubMed] [Google Scholar]
- 55.de Roock S, van Elk M, Hoekstra MO, Prakken BJ, Rijkers GT, de Kleer IM (2011) Gut derived lactic acid bacteria induce strain specific CD4+ T cell responses in human PBMC. Clin Nutr 30(6):845–851. 10.1016/j.clnu.2011.05.005 [DOI] [PubMed] [Google Scholar]
- 56.Fu L, Song J, Wang C, Fu S, Wang Y (2017) Bifidobacterium infantis potentially alleviates shrimp tropomyosin-induced allergy by tolerogenic dendritic cell-dependent induction of regulatory T cells and alterations in gut microbiota. Front Immunol 8:1536. 10.3389/fimmu.2017.01536 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Balzaretti S, Taverniti V, Guglielmetti S, Fiore W, Minuzzo M, Ngo HN et al (2017) A novel rhamnose-rich hetero-exopolysaccharide isolated from Lactobacillus paracasei DG activates THP-1 human monocytic cells. Appl Environ Microbiol 83(3):e02702-e2716. 10.1128/AEM.02702-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Olaimat AN, Aolymat I, Al-Holy M, Ayyash M, Ghoush MA, Al-Nabulsi AA et al (2020) The potential application of probiotics and prebiotics for the prevention and treatment of COVID-19. NPJ Sci Food 4(1):1–7. 10.1038/s41538-020-00078-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Chang TL-Y, Chang C-H, Simpson DA, Xu Q, Martin PK, Lagenaur LA et al (2003) Inhibition of HIV infectivity by a natural human isolate of Lactobacillus jensenii engineered to express functional two-domain CD4. Proc Natl Acad Sci 100(20):11672–7. 10.1073/pnas.1934747100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Olaya Galán N, Ulloa Rubiano J, Velez Reyes F, Fernandez Duarte K, Salas Cardenas S, Gutierrez FM (2016) In vitro antiviral activity of Lactobacillus casei and Bifidobacterium adolescentis against rotavirus infection monitored by NSP 4 protein production. J Appl Microbiol 120(4):1041–1051. 10.1111/jam.13069 [DOI] [PubMed] [Google Scholar]
- 61.Ang LYE, Too HKI, Tan EL, Chow T-KV, Shek P-CL, Tham E et al (2016) Antiviral activity of Lactobacillus reuteri protectis against Coxsackievirus A and Enterovirus 71 infection in human skeletal muscle and colon cell lines. Virol J. 13(1):1–12. 10.1186/s12985-016-0567-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Khani S, Motamedifar M, Golmoghaddam H, Hosseini HM, Hashemizadeh Z (2012) In vitro study of the effect of a probiotic bacterium Lactobacillus rhamnosus against herpes simplex virus type 1. Braz J Infect Dis 16(2):129–135. 10.1590/S1413-86702012000200004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kim K, Lee G, Thanh HD, Kim J-H, Konkit M, Yoon S et al (2018) Exopolysaccharide from Lactobacillus plantarum LRCC5310 offers protection against rotavirus-induced diarrhea and regulates inflammatory response. J Dairy Sci 101(7):5702–5712. 10.3168/jds.2017-14151 [DOI] [PubMed] [Google Scholar]
- 64.Yang Y, Song H, Wang L, Dong W, Yang Z, Yuan P et al (2017) Antiviral effects of a probiotic metabolic products against transmissible gastroenteritis coronavirus. J Prob Health 5(3):1–6 [Google Scholar]
- 65.Khan R, Petersen FC, Shekhar S (2019) Commensal bacteria: an emerging player in defense against respiratory pathogens. Front Immunol 10:1203. 10.3389/fimmu.2019.01203 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Singh K, Rao A (2021) Probiotics: a potential immunomodulator in COVID-19 infection management. Nutr Res 87:1–12. 10.1016/j.nutres.2020.12.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Gupta A, Madhavan MV, Sehgal K, Nair N, Mahajan S, Sehrawat TS et al (2020) Extrapulmonary manifestations of COVID-19. Nat Med 26(7):1017–1032. 10.1038/s41591-020-0968-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Anwar F, Altayb HN, Al-Abbasi FA, Al-Malki AL, Kamal MA, Kumar V (2020) Antiviral effects of probiotic metabolites on COVID-19. J Biomol Struct Dyn. 10.1080/07391102.2020.1775123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kanmani P, Satish Kumar R, Yuvaraj N, Paari K, Pattukumar V, Arul V (2013) Probiotics and its functionally valuable products—a review. Crit Rev Food Sci Nutr 53(6):641–658. 10.1080/10408398.2011.553752 [DOI] [PubMed] [Google Scholar]
- 70.Soltani S, Hammami R, Cotter PD, Rebuffat S, Said LB, Gaudreau H et al (2020) Bacteriocins as a new generation of antimicrobials: toxicity aspects and regulations. FEMS Microbiol Rev. 10.1093/femsre/fuaa039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Mokoena MP (2017) Lactic acid bacteria and their bacteriocins: classification, biosynthesis and applications against uropathogens: a mini-review. Molecules 22(8):1255. 10.3390/molecules22081255 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wu M-H, Pan T-M, Wu Y-J, Chang S-J, Chang M-S, Hu C-Y (2010) Exopolysaccharide activities from probiotic bifidobacterium: Immunomodulatory effects (on J774A. 1 macrophages) and antimicrobial properties. Int J Food Microbiol 144(1):104–10. 10.1016/j.ijfoodmicro.2010.09.003 [DOI] [PubMed] [Google Scholar]
- 73.Trabelsi I, Ktari N, Slima SB, Triki M, Bardaa S, Mnif H et al (2017) Evaluation of dermal wound healing activity and in vitro antibacterial and antioxidant activities of a new exopolysaccharide produced by Lactobacillus sp. Ca6. Int J Biol Macromol. 103:194–201. 10.1016/j.ijbiomac.2017.05.017 [DOI] [PubMed] [Google Scholar]
- 74.Ayyash M, Abu-Jdayil B, Itsaranuwat P, Galiwango E, Tamiello-Rosa C, Abdullah H et al (2020) Characterization, bioactivities, and rheological properties of exopolysaccharide produced by novel probiotic Lactobacillus plantarum C70 isolated from camel milk. Int J Biol Macromol 144:938–946. 10.1016/j.ijbiomac.2019.09.171 [DOI] [PubMed] [Google Scholar]
- 75.Rajoka MSR, Jin M, Haobin Z, Li Q, Shao D, Jiang C et al (2018) Functional characterization and biotechnological potential of exopolysaccharide produced by Lactobacillus rhamnosus strains isolated from human breast milk. Lwt 89:638–647. 10.1016/j.lwt.2017.11.034 [Google Scholar]
- 76.Rani RP, Anandharaj M, Ravindran AD (2018) Characterization of a novel exopolysaccharide produced by Lactobacillus gasseri FR4 and demonstration of its in vitro biological properties. Int J Biol Macromol 109:772–783. 10.1016/j.ijbiomac.2017.11.062 [DOI] [PubMed] [Google Scholar]
- 77.Angelin J, Kavitha M (2020) Exopolysaccharides from probiotic bacteria and their health potential. Int J Biol Macromol 162:853–865. 10.1016/j.ijbiomac.2020.06.1907 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Angurana SK, Bansal A (2020) Probiotics and COVID-19: think about the link. Br J Nutr. 10.1017/S000711452000361X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Dhar D, Mohanty A (2020) Gut microbiota and Covid-19-possible link and implications. Virus Res. 10.1016/j.virusres.2020.198018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Gogineni VK, Morrow LE, Malesker MA (2013) Probiotics: mechanisms of action and clinical applications. J Prob Health 1(101):2. 10.1039/C8FO00376A [Google Scholar]
- 81.Parvez S, Malik KA, Ah Kang S, Kim HY (2006) Probiotics and their fermented food products are beneficial for health. J Appl Microbiol 100(6):1171–1185. 10.1111/j.1365-2672.2006.02963.x [DOI] [PubMed] [Google Scholar]
- 82.Fedorak RN, Madsen KL (2004) Probiotics and the management of inflammatory bowel disease. Inflamm Bowel Dis 10(3):286–299. 10.1097/00054725-200405000-00018 [DOI] [PubMed] [Google Scholar]
- 83.Amer M, Nadeem M, Nazir R, Ur S, Fakhar M, Abid F, Ain QU (2018) Probiotics and their use in inflammatory bowel disease. Altern Ther Health Med 24(3). [PubMed]
- 84.Xu K, Cai H, Shen Y, Ni Q, Chen Y, Hu S et al (2020) Management of corona virus disease-19 (COVID-19): the Zhejiang experience. J Zhejiang Univ (Med Sci). 10.3785/j.issn.1008-9292.2020.02.02 [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.