Abstract
Objectives
To evaluate the incidence and risk factors of postoperative pneumonia (POP) in geriatric patients with a hip fracture after surgery, to design a predictive nomogram, and to validate the accuracy of the nomogram.
Design
Retrospective study.
Setting
A tertiary hospital affiliated to a medical university.
Patients/Participants
We retrospectively studied 1285 surgical-treated geriatric patients with a hip fracture from April 2010 to April 2018.
Intervention
Surgical treatment was performed on the patients of this study. The procedure methods were classified as: total hip arthroplasty, hemiarthroplasty, percutaneous fixation, intramedullary nail fixation, and plate/screw fixation.
Main Outcome Measurement
The primary interest of end point of this study is the development of POP during the postoperative period. The postoperative period in this study was defined as the time from 24 hours after surgery to discharge. The diagnostic criteria for pneumonia were set according to the guidelines built by the Infectious Diseases Society of America and the American Thoracic Society (Guidelines for the Management of Adults with Hospital-Acquired, Ventilator-Associated, and Healthcare-Associated Pneumonia, 2005). Potential variables for developing POP were identified using logistic regression analyses initially and were further selected via the method of LASSO. Then the independent risk factors were identified by multivariable regression analyses. A predictive nomogram was built based on the multiple regression model, and the calibration abilities of the nomogram was measured by Harrel C-index, calibration plot and Hosmer–Lemeshow test, respectively. Decision curve analysis was carried out to assess the net benefit due to threshold probability and an on-line questionnaire survey was conducted among the clinicians to assess the applicability of the nomogram coherently.
Results
Of the 1285 patients, 70 (5.4%) developed POP. COPD, number of comorbidities, ASA classification >2, preoperative dependent functional status and cognitive impairment were identified as independent risk factors of POP. The nomogram built based on the results showed good accordance between the predicted probabilities and the observed frequency. The decision curve analysis confirmed the clinical utility of the nomogram when the threshold probabilities were between 5% and 65% due to the net benefit, while the results of on-line questionnaire among 200 clinicians showed that 91.5% of the participants had a mental threshold of intervention between 5-50%.
Conclusion
(1). COPD, number of comorbidities, ASA classification >2, preoperative dependent functional status and cognitive impairment are independent risk factors for POP. (2). The nomogram built in this study has a good accordance between the predictive risk and the observational incidence. The results of decision curve and questionnaire among clinicians show well applicability of the nomogram.
Keywords: hip fracture, geriatric, postoperative pneumonia, nomogram
Introduction
As a worldwide public health concern, hip fracture has ranked one of the top 10 causes of disability 1 and contributed to high mortalities and morbidities in the elderly population, which leads to increasing socioeconomic cost.2,3 The one-year mortality rate could be as high as 36% despite progressive surgical management and accelerated rehabilitation. 4 Many life-threatening complications have been investigated in current literature, such as cardiac infarction, stroke, deep vein thrombosis, and subsequent pulmonary embolism,5,6 while little has been studied regarding pneumonia, a troubling complication following hip fracture surgery. 7 Little is known for the occurrence of postoperative pneumonia (POP) during the hospital stay. The purpose of this study is to determine the incidence and independent risk factors of POP after hip fracture surgeries in geriatric patients, to construct a predictive nomogram for the development of POP and to evaluate the accuracy and clinical applicability of the nomogram.
Patients and Methods
Patient Population
In this study, we retrospectively studied 1285 patients with hip fractures and treated by surgical management between April 2010 and April 2018 in a single center. The inclusion criteria of patients were as follows: (1) age ≥65, (2) postoperative diagnosis of unilateral femoral neck fracture, or intertrochanteric fracture; (3) receiving total hip arthroplasty, or hemiarthroplasty, or percutaneous fixation, intramedullary nail fixation, or plate/screw fixation. The exclusion criteria included (1) pathological fractures or history of diagnostic malignant tumor, (2) multiple injuries, and (3) open fractures.
Patients were characterized at baseline according to sex, age, body mass index (BMI), comorbidities, current smoking status, fracture pattern, risk classification of anesthesia according to American Association of Anesthesiologists (ASA), type of anesthesia, surgery option, time to surgery, operation time, blood loss, transfusion, preoperative hemoglobin and serum albumin concentration, intensive care, and length of hospital stay, etc. (Table 1). Age was stratified into 65–74, 70–74, 75–79, 80–84, and ≥85 years. Hemoglobin was stratified into <120 g/L in male and <110 g/L in female (anemia) vs. ≥120 g/L in male and ≥110 g/L in female (normal). Serum albumin was stratified into <35 g/L (hypoalbuminemia) vs. ≥35 g/L (normal). Transfusion was stratified into >2 units of RBC or not. Time to surgery, which was calculated in days, was defined as the time elapsed from hospital admission to surgery. 8 Surgery procedure was stratified as described above.
Table 1.
Baseline characteristics of patients and univariable logistic regression.
| Risk Factors | All | No | Yes | OR | p.ratio |
|---|---|---|---|---|---|
| N=1285 | N=1215 | N=70 | |||
| Gender | |||||
| Female | 896 (69.7%) | 856 (70.5%) | 40 (57.1%) | Ref | Ref |
| Male | 389 (30.3%) | 359 (29.5%) | 30 (42.9%) | 1.79 [1.09; 2.92] | .022 |
| Age, years | 80.0 (75.0–84.0) | 79.0 (75.0–84.0) | 82.0 (79.0–86.8) | 1.06 [1.02; 1.09] | .002 |
| Age, years | |||||
| 65–69 | 130 (10.1%) | 127 (10.5%) | 3 (4.29%) | Ref | Ref |
| 70–74 | 176 (13.7%) | 170 (14.0%) | 6 (8.57%) | 1.46 [.36; 7.43] | .604 |
| 75–79 | 329 (25.6%) | 317 (26.1%) | 12 (17.1%) | 1.54 [.47; 7.18] | .498 |
| 80–84 | 330 (25.7%) | 308 (25.3%) | 22 (31.4%) | 2.89 [.97; 12.87] | .057 |
| ≥85 | 320 (24.9%) | 293 (24.1%) | 27 (38.6%) | 3.72 [1.28; 16.42] | .013 |
| BMI, Kg/m2 | 23.4 (21.1–25.6) | 23.4 (21.1–25.6) | 23.4 (21.5–26.0) | 1.05 [.98; 1.12] | .196 |
| BMI, Kg/m 2 | |||||
| (0, 18.5] | 53 (4.12%) | 48 (3.95%) | 5 (7.14%) | Ref | Ref |
| (18.5, 20] | 147 (11.4%) | 143 (11.8%) | 4 (5.71%) | .27 [.06; 1.11] | .069 |
| (20.0, 25.0] | 703 (54.7%) | 664 (54.7%) | 39 (55.7%) | .55 [.22; 1.68] | .267 |
| (25.0, 30.0] | 335 (26.1%) | 320 (26.3%) | 15 (21.4%) | .44 [.16; 1.44] | .163 |
| (30, +∞) | 47 (3.66%) | 40 (3.29%) | 7 (10.0%) | 1.66 [.48; 6.17] | .423 |
| Type of fracture | |||||
| Intertrochanteric fracture | 579 (45.1%) | 554 (45.6%) | 25 (35.7%) | Ref | Ref |
| Femoral neck fracture | 706 (54.9%) | 661 (54.4%) | 45 (64.3%) | 1.50 [.92; 2.52] | .107 |
| Cognitive impairment | |||||
| No | 1239 (96.4%) | 1180 (97.1%) | 59 (84.3%) | Ref | Ref |
| Yes | 46 (3.58%) | 35 (2.88%) | 11 (15.7%) | 6.32 [2.92; 12.77] | <.001 |
| Smoking | |||||
| never | 984 (76.6%) | 928 (76.4%) | 56 (80.0%) | Ref | Ref |
| current/former | 301 (23.4%) | 287 (23.6%) | 14 (20.0%) | .82 [.43; 1.45] | .499 |
| Comorbidities | |||||
| No | 515 (40.1%) | 508 (41.8%) | 7 (10.0%) | Ref | Ref |
| Yes | 770 (59.9%) | 707 (58.2%) | 63 (90.0%) | 6.33 [3.07; 15.42] | <.001 |
| Comorbidities | |||||
| <2 | 951 (74.0%) | 932 (76.7%) | 19 (27.1%) | Ref | Ref |
| ≥2 | 334 (26.0%) | 283 (23.3%) | 51 (72.9%) | 8.78 [5.18; 15.51] | <.001 |
| Hypertension | |||||
| No | 726 (56.5%) | 687 (56.5%) | 39 (55.7%) | Ref | Ref |
| Yes | 559 (43.5%) | 528 (43.5%) | 31 (44.3%) | 1.04 [.63; 1.68] | .889 |
| Diabetes mellitus | |||||
| No | 1074 (83.6%) | 1024 (84.3%) | 50 (71.4%) | Ref | Ref |
| Yes | 211 (16.4%) | 191 (15.7%) | 20 (28.6%) | 2.15 [1.22; 3.65] | .009 |
| COPD | |||||
| No | 1236 (96.2%) | 1186 (97.6%) | 50 (71.4%) | Ref | Ref |
| Yes | 49 (3.81%) | 29 (2.39%) | 20 (28.6%) | 16.28 [8.51; 30.77] | <.001 |
| Congestive heart failure | |||||
| No | 1249 (97.2%) | 1200 (98.8%) | 49 (70.0%) | Ref | Ref |
| Yes | 36 (2.80%) | 15 (1.23%) | 21 (30.0%) | 33.85 [16.50; 71.32] | <.001 |
| Hypotensive drugs | |||||
| No | 726 (56.5%) | 687 (56.5%) | 39 (55.7%) | Ref | Ref |
| Yes | 559 (43.5%) | 528 (43.5%) | 31 (44.3%) | 1.04 [.63; 1.68] | .889 |
| Hypoglycemic drugs | |||||
| No | 1184 (92.1%) | 1123 (92.4%) | 61 (87.1%) | Ref | Ref |
| Yes | 101 (7.86%) | 92 (7.57%) | 9 (12.9%) | 1.83 [.82; 3.63] | .133 |
| Cardiovascular drugs | |||||
| No | 1189 (92.5%) | 1132 (93.2%) | 57 (81.4%) | Ref | Ref |
| Yes | 96 (7.47%) | 83 (6.83%) | 13 (18.6%) | 3.13 [1.58; 5.80] | .002 |
| Respiratory drugs | |||||
| No | 1267 (98.6%) | 1204 (99.1%) | 63 (90.0%) | Ref | Ref |
| Yes | 18 (1.40%) | 11 (.91%) | 7 (10.0%) | 12.19 [4.28; 32.43] | <.001 |
| Other drugs | |||||
| No | 1218 (94.8%) | 1156 (95.1%) | 62 (88.6%) | Ref | Ref |
| Yes | 67 (5.21%) | 59 (4.86%) | 8 (11.4%) | 2.56 [1.08; 5.34] | .034 |
| Traction | |||||
| No | 1184 (92.1%) | 1121 (92.3%) | 63 (90.0%) | Ref | Ref |
| Yes | 101 (7.86%) | 94 (7.74%) | 7 (10.0%) | 1.35 [.55; 2.85] | .486 |
| ASA classification | 2.00 (2.00–3.00) | 2.00 (2.00–3.00) | 3.00 (2.00–3.00) | 2.83 [1.93; 4.15] | <.001 |
| ASA (2) | |||||
| 1–2 | 915 (71.2%) | 888 (73.1%) | 27 (38.6%) | Ref | Ref |
| 3–4 | 370 (28.8%) | 327 (26.9%) | 43 (61.4%) | 4.31 [2.63; 7.18] | <.001 |
| Preoperative RBC | 3.75 (3.33–4.18) | 3.76 (3.33–4.17) | 3.66 (3.23–4.20) | .79 [.55; 1.13] | .198 |
| Preoperative WBC | 8.20 (6.56–9.60) | 8.20 (6.56–9.60) | 8.21 (6.49–9.95) | .99 [.89; 1.09] | .827 |
| Preoperative ALB | 36.0 (33.0–38.0) | 36.0 (33.0–38.0) | 34.0 (30.0–37.0) | .91 [.87; 0.95] | <.001 |
| Preoperative ALB | |||||
| ≥35 | 807 (62.8%) | 776 (63.9%) | 31 (44.3%) | Ref | Ref |
| <35 | 478 (37.2%) | 439 (36.1%) | 39 (55.7%) | 2.22 [1.37; 3.64] | .001 |
| Preoperative Cr | 67.0 (56.0–82.0) | 67.0 (56.0–81.5) | 70.0 (61.0–90.5) | 1.00 [1.00; 1.01] | .922 |
| Preoperative HGB | 114 (102–125) | 115 (102–125) | 112 (98.0–124) | .99 [.98; 1.01] | .318 |
| Preoperative PLT | 173 (140–214) | 172 (140–214) | 174 (142–214) | 1.00 [1.00; 1.00] | .645 |
| Time to surgery | 4.00 (3.00–5.00) | 4.00 (3.00–5.00) | 4.50 (3.00–6.00) | 1.08 [.97; 1.19] | .165 |
| Anesthesia type | |||||
| Intra-spinal/nerve block | 1068 (83.1%) | 1012 (83.3%) | 56 (80.0%) | Ref | Ref |
| general | 217 (16.9%) | 203 (16.7%) | 14 (20.0%) | 1.26 [.66; 2.24] | .471 |
| Operation time | 80.0 (60.0–105) | 80.0 (60.0–105) | 87.5 (70.0–105) | 1.00 [1.00; 1.01] | .23 |
| Type of operation | |||||
| hemiarthroplasty | 347 (27.0%) | 325 (26.7%) | 22 (31.4%) | Ref | Ref |
| multiple screw | 31 (2.41%) | 29 (2.39%) | 2 (2.86%) | 1.08 [.15; 3.98] | .918 |
| plate/screw | 153 (11.9%) | 144 (11.9%) | 9 (12.9%) | .93 [.40; 2.03] | .865 |
| total hip arthroplasty | 298 (23.2%) | 280 (23.0%) | 18 (25.7%) | .95 [.49; 1.81] | .88 |
| intramedullary nail | 456 (35.5%) | 437 (36.0%) | 19 (27.1%) | .64 [.34; 1.21] | .172 |
| Blood loss | 200 (100–350) | 200 (100–350) | 200 (100–300) | 1.00 [1.00; 1.00] | .553 |
| Preoperative anemia | |||||
| No | 695 (54.1%) | 661 (54.4%) | 34 (48.6%) | Ref | Ref |
| yes | 590 (45.9%) | 554 (45.6%) | 36 (51.4%) | 1.26 [.78; 2.05] | .345 |
| Functional status | |||||
| independent | 993 (77.3%) | 956 (78.7%) | 37 (52.9%) | Ref | Ref |
| dependent | 292 (22.7%) | 259 (21.3%) | 33 (47.1%) | 3.29 [2.01; 5.37] | <.001 |
| Transfusion >2 iu RBC | |||||
| no | 690 (53.7%) | 653 (53.7%) | 37 (52.9%) | Ref | Ref |
| yes | 595 (46.3%) | 562 (46.3%) | 33 (47.1%) | 1.04 [.64; 1.68] | .884 |
| Postoperative ICU | |||||
| No | 1066 (83.0%) | 1017 (83.7%) | 49 (70.0%) | Ref | Ref |
| Yes | 219 (17.0%) | 198 (16.3%) | 21 (30.0%) | 2.21 [1.27; 3.72] | .006 |
| Length of stay | 10.0 (8.00–12.0) | 10.0 (8.00–12.0) | 11.5 (9.00–13.0) | 1.01 [1.00; 1.03] | .118 |
Patients were characterized according to whether or not they developed pneumonia in the postoperative period. The postoperative period in this study was defined as the time from 24 hours after surgery to discharge. The diagnostic criteria for pneumonia were set according to the guidelines built by the Infectious Diseases Society of America and the American Thoracic Society (Guidelines for the Management of Adults with Hospital-Acquired, Ventilator-Associated, and Healthcare-Associated Pneumonia, 2005). 9 In the guidelines, pneumonia was defined as the presence of “new lung infiltrate plus clinical evidence that the infiltrate is of an infectious origin, which includes the new onset of fever, purulent sputum, leukocytosis, and decline in oxygenation.” Accordingly, in our study, the identification of development of pneumonia should accord with the following events that could be confirmed in the patients’ medical/nursing records and radiological database: (1) a radiographic (X-ray or CT scan) infiltrate that was new or progressive, (2) observable clinical symptoms, which included a new onset of fever with a axillary temperature over 37.0°C, and/or purulent sputum with a positive sputum culture, (3) leukocytosis. Two experienced doctors (the co-first authors of this study) independently identified the diagnosis of POP for all patients following the diagnostic strategies. Any controversies in diagnosis between the two doctors were settled by discussion, and a final diagnosis for POP on each patient was made.
Statistical Analysis
The primary end point of interest in this study was the development of POP. Multivariable regression analysis was conducted by logistic regression model, and this model was the basis of the POP prediction model. The potential predictors of POP analyzed in this study included age, sex, body mass index, smoking status, hypertension, diabetes, COPD, congestive heart failure, preoperative functional status, fracture pattern, ASA classification, serum albumin, hemoglobin, time to surgery, anesthesia type, operation type, operation time, blood loss, transfusion, and ICU stay. An initial univariate logistic regression model was fitted to evaluate the association between the clinical factors and pneumonia. To identify independent risk factors, variables achieving a significance level of P < .05 were initially selected out. The method of LASSO was used to select potential strong factors and then independent predictors were identified by multiple regression analysis. The independent statistically significant predictors in the final model were determined with the backward method based on the AIC criteria. A predictive nomogram was established based on results of the multiple regression. A predictive discrimination was evaluated using Harrel Concordance index (C-index), which measured the probability that, given a pair of randomly selected patients, the model correctly predicted which patient will experience an event first. The C-index of the model can range between .5, which represents random chance, and 1.0, which represents a perfectly discriminating model (Harrell et al, 1982). The relatively correctional C-index of the nomogram was calculated by bootstraps with 1000 resamples. In calibration, the predicted probability of postoperative pneumonia generated by 10-fold cross validation was compared with the observed cumulative incidence estimates of pneumonia probability. The consistency of the curves was evaluated by Hosmer–Lemeshow (H-L) test. All statistical analyses were conducted using R software version 3.5.0 (http://www.r-project.org/), with rms package (Harrell, 2012). In addition, the decision curve analysis was carried out using the code found at https://www.mskcc.org/departments/epidemiology biostatistics/health-outcomes/decision-curve-analysis-01 according to its tutorials, and an on-line questionnaire (Appendix 1) was conducted on Tencent Questionnaire Platform from February 11, 2019, to February 15, 2019, combined with the decision curve to study the threshold for intervention among clinical practitioners, which was used to evaluate the clinical applicability of the nomogram comprehensively.
Results
Baseline Characteristics
There were 1409 patients who met the inclusion criteria for this study, of which 124 were excluded due to the exclusion criteria. Finally, 1285 patients were included in this study. The clinical and pathologic characteristics of patients were listed in Table 1. There were 389 (30.3%) male and 896 (69.7%) female patients in total with a median age of 80 (IQR, 75–84 years) at the time of diagnosis. 770 patients (59.9%) had comorbidities preoperatively. 334 patients (26.0%) had two or more than two kinds of comorbidities. Hypertension (43.5%) and diabetes (16.4%) were the most common comorbidities. COPD (3.8%) and congestive heart failure (2.8%) also constituted a small amount of comorbidities. Eventually, 70 (5.4%) patients developed POP, of which 30 (42.9%) were male and 40 (57.1%) were female.
Comparison of the Clinicopathological Features Between Patients With and Without POP
Compared to patients without POP, those with POP had more older age individuals (82.0 vs. 79.0, P = .002), a higher portion of male sex (42.9% vs. 29.5%, P = .022), a higher portion of patients who had an ASA classification higher than grade 2 (61.4% vs. 26.9%, P < .001), more individuals who had at least two kinds of comorbidities (72.9% vs. 23.3%, P < .001), more dependent functional status (47.1% vs. 21.3%, P < .001) and more preoperative hypoproteinemia (55.7% vs. 36.1%, P = .001), a higher rate of diabetes (28.6% vs. 15.7%, P = .009), more COPD (28.6% vs. 2.39%, P < .001), more congestive heart failure (30.0% vs. 1.23%, P < .001), more postoperative ICU stay (30.0% vs. 16.3%, p = .006). (Table 1).
Multiple Regression Analysis
The results of the univariate regression analysis were shown in Table 1. In the univariate logistic regression, male sex (P = .022), age (P = .002), preoperative cognitive impairment (P < .001), number of comorbidities ≥2 (P < .001), diabetes (P = .009), COPD (P < .001), ASA classification >2 (P < .001), preoperative hypoproteinemia (P = .001), preoperative dependent functional status (P < .001) and postoperative ICU stay (P = .006) were found to be significant predictors of POP. These factors were further filtrated by the method of LASSO. Comparatively weaker factors were eliminated so that the strength of influence of the factors was enhanced. Finally, in multiple regression analysis, COPD, number of comorbidities, ASA classification >2, preoperative dependent functional status and cognitive impairment were identified as independent risk factors of POP. (Table 2).2
Table 2.
Multiple Logistic Regression for risk factors of POP.
| Risk Factors | OR | 95% CI | P Value |
|---|---|---|---|
| COPD | <.001 | ||
| No | Ref | ||
| Yes | 10.38 | 4.90–22.00 | |
| Number of comorbidities | <.001 | ||
| <2 | Ref | ||
| ≥2 | 5.50 | 3.07–9.84 | |
| ASA classification | .001 | ||
| 1–2 | Ref | ||
| 3–4 | 2.92 | 1.68–5.09 | |
| Preoperative functional status | <.001 | ||
| Independent | Ref | ||
| Dependent | 3.33 | 1.90–5.84 | |
| Cognitive impairment | .002 | ||
| No | Ref | ||
| Yes | 4.26 | 1.69–10.70 |
C-index: .8396355 (.7838565–.8954145).
HL test: P = .8839044.
Development of a Predictive Nomogram and Utility of the Nomogram
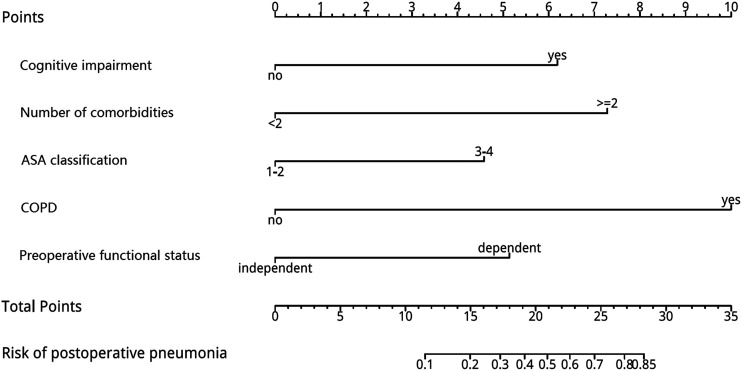
To provide the physicians with a quantitative method to predict the risk of postoperative pneumonia, we constructed a predictive nomogram (Figure 1) according to the strength of influence of each independent risk factors based on the multivariable logistic regression (Table 2).
Figure 1.
Predictive nomogram for postoperative pneumonia. This predictive nomogram was constructed based on the multivariable model. To use the nomogram, a vertical line is drawn up to the top point row to assign points for each variable. Then, the total number of points is calculated, and a vertical line is drawn downward from the total point row to obtain the probability of POP.
To use the nomogram, a vertical line is drawn up to the top point row to assign points for each variable. Then, the total number of points is calculated, and a vertical line is drawn downward from the total point row to obtain the probability of POP. The risk of POP increases with the increasing total points, due to the nomogram. The total points of the nomogram ranges from 0 to 35 points, corresponding to the predicted risk of POP from less than 10% to greater than 85%. (Figure 1). For example, when the total points is calculated to be 15, the corresponding risk of postoperative pneumonia is 20%.
Validation of the Nomogram
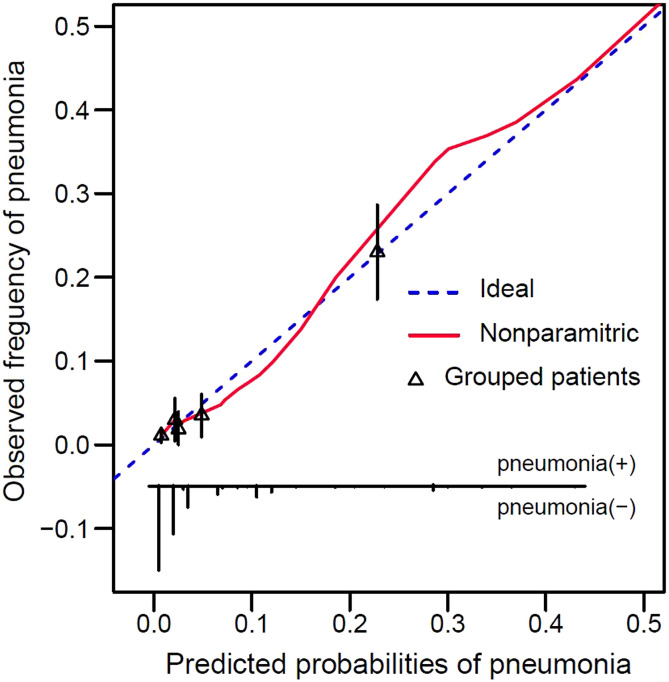
The nomogram was validated by the method of Harrel C-index and a calibration plot using the R software 3.0. H-L test was performed to evaluate the statistical significance of the consistency between the curves in the calibration plot.
The nomogram displayed a C-index of .84 (95% CI 0.78–.90) which indicated good accuracy of the predictive model. The calibration plot showed good concordance between predicted probability and actual probability. (Figure 2). The H-L test showed that the curves in the calibration plot had statistical significance. (P=.88)
Figure 2.
Calibration plot. The calibration curve showed good concordance between predicted probability and actual probability.
Decision Curve Analysis and Results of the On-Line Questionnaire
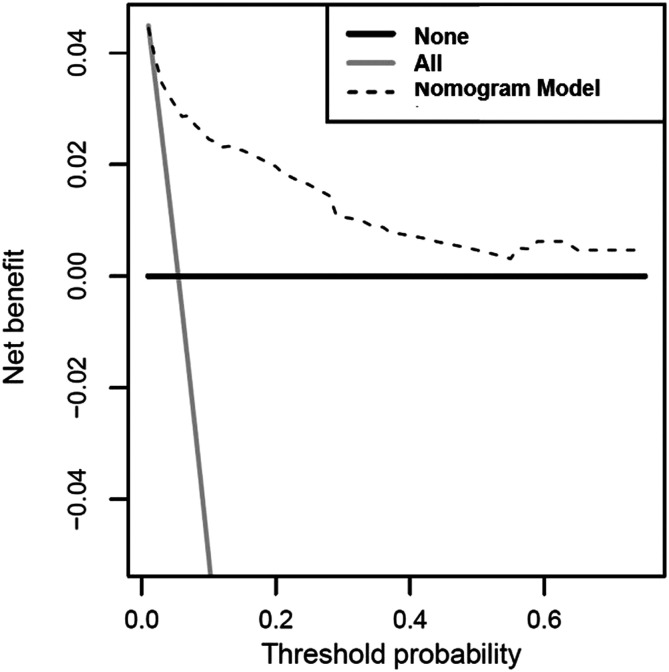
Based on the threshold probability, the decision analysis curve was leveraged to evaluate the clinical application of a prediction model. In the decision analysis curve, the nomogram was compared with the null model for its added value, which turned out that the nomogram was applicable when thresholds were in the range of .05–.65 due to the net benefit (Figure 3).
Figure 3.
Decision curve plot. In the decision analysis curve, the nomogram was compared with the null model for its added value, which turned out that the nomogram was applicable when thresholds were in the range between 0.05 and 0.65, due to the net benefit.
The results of on-line questionnaire among 200 clinicians showed that 89.5% of the participants had obtained Identification of Physician’s Qualification for more than 5 years. 52.5% had senior professional qualifications. 88.5% had been working as an orthopedist for more than 5 years. 62% had an experience of surgical treatment for geriatric hip fractures for more than 100 cases. 97% of the participants declared that they had encountered POP among geriatric hip fracture patients in their career. 91.5% had a threshold of intervention when predictive risk of POP ranged from 5% to 50%, which slickly conformed to the net-benefit threshold interval produced by the decision curve, indicating well clinical applicability of the nomogram (Table 3).
Table 3.
The results of On-Line Questionnaire
| All | Percent, % | |
|---|---|---|
| 1、The period of holding identification of Physician’s qualification, year | ||
| <3 | 4 | 2.0 |
| 3∼5 | 17 | 8.5 |
| 6∼10 | 31 | 15.5 |
| >10 | 148 | 74.0 |
| 2、Professional title | ||
| resident Doctor | 24 | 12 |
| attending Doctor | 71 | 35.5 |
| associate Chief | 77 | 38.5 |
| chief | 28 | 14 |
| 3、Years of working as a an orthopedic surgeon | ||
| <3 | 8 | 4.0 |
| 3∼5 | 15 | 7.5 |
| 6∼10 | 40 | 20.0 |
| >10 | 137 | 68.5 |
| 4、Experience of surgical treatment for geriatric patients with hip fractures | ||
| <50 | 28 | 14.0 |
| 50∼100 | 48 | 24.0 |
| 101∼300 | 60 | 30.0 |
| >300 | 64 | 32.0 |
| 5、Frequency of encountering POP in geriatric patients with hip fractures | ||
| Never | 6 | 3.0 |
| Rarely | 52 | 26.0 |
| Sometimes | 117 | 58.5 |
| Often | 25 | 12.5 |
| 6、The threshold of predicted probability to intervene | ||
| 5%∼10% | 78 | 39.0 |
| 11%∼25% | 65 | 32.5 |
| 26%∼50% | 40 | 20.0 |
| >50% | 17 | 8.5 |
Discussion
The increasing number of hip fractures worldwide predicts greater impact to health status in elderly population. In the United States, it has been estimated that the total number of hip fractures might double by the year 2040 compared to the 1980s. 2 Greater contributions should be made by Asian countries for the increasing number of osteoporotic hip fractures in future decades. 10 The risk of mortality and morbidity within one year of fracture remains high according to the existing literatures4,11-14.
The improvements in surgical and rehabilitation managements these years have improved clinical outcomes of hip fractures, while complications following hip fracture surgery remain frequent and troubling and are often complex, which might be caused by various factors, such as general conditions, comorbidities, traumatic impairment, surgery, anesthesia, and immobilization. Several studies have focused on the fatal adverse events during the postoperative periods, such as acute myocardial infarction, stroke, deep vein thrombosis,16-20 which were known as the most dangerous events.
Pneumonia is also known to be a devastating complication after hip fracture surgery, which can also be life-threatening when correlative respiratory and circulatory failure was induced subsequently, and is especially risky to elderly patients who are more easily affected by the impactions of trauma and immobilization after fractures and surgeries. The reported incidence of pneumonia in elderly patients with a hip fracture was 1.2% preoperatively 22 and approximately 4% ∼ 5% postoperatively according to recent literatures14,15,21 Although the overall incidence was not alarmingly great, it was found that patients who developed pneumonia would have a higher readmission rate, a higher rate of sepsis, and a higher mortality rate. 15 In our study, 70 (5.4%) of 1285 patients developed postoperative pneumonia, which involved the time from 24 hours after surgery to discharge. The latest guidelines built by the Infectious Diseases Society of America and the American Thoracic Society in 2016 23 has differed Ventilator-Associated Pneumonia (VAP) from the traditional concept of Hospital-Associated Pneumonia (HAP). However, in developing countries, such as China, situation remains different. Community mechanical ventilator equipment is far from popularized yet, so ventilators still could only be seen in the intensive care unit in hospital in most cases. The development of POP will significantly increase the length of stay, while clinical data would be hard to collect after discharge. Therefore, in this study, we defined POP as a new-onset pneumonia from 24 hours after surgery to discharge, so that all the clinical and radiological evidences could be exactly obtained.
Little was known regarding the occurrence of pneumonia in postoperative periods. In a recent retrospective study of 29,377 patients, male sex, older age, low body mass index (BMI), and chronic obstructive pulmonary disease (COPD) were identified to be the strongest risk factors for pneumonia. 15 Our study has also shown that COPD, ASA classification, number of comorbidities, cognitive impairment, and preoperative dependent functional status were identified to be the independent risk factors for the development of POP. We considered that the smaller sample and ethnological difference might play a role in the discrepancy of the results.
The impacts of pneumonia on clinical outcomes and rehabilitations in elderly patients with a hip fracture have been demonstrated, and it has been suggested that prevention of pneumonia is necessary to improve the outcome of hip fracture. 15 In a recent research, a nonrandomized, quasi-experimental study was designed to evaluate an inpatient pulmonary rehabilitation program by comparing 2 sequential time periods in a same center, 24 and a pulmonary rehabilitation program was demonstrated to be effective to reduce the incidence of pneumonia. For many older adults, prevention of complication may be a more important goal than cure. Thus, an effective prediction strategy is urgently needed clinically. However, previous researches have many limitations regarding the prediction strategies for POP, and prospective studies were rarely found in current literature.
The main advantages of this study include: first, a nomogram was built for the prediction for the development of POP, with good accuracy according to the statistical verifications. As a graphical presentation of predictive model, nomogram has been widely used in the area of oncology,25-28 with great advantages such as easy operation, convenience, and effectiveness. Previously, we have reported the predictive nomogram for the development of postoperative delirium in elderly patients with a hip fracture. 29 The nomogram created in this study is supposed to be used clinically as a prediction method in the preoperative assessment, thus further studies with prospective designs can be conducted based on the existing preliminary results. Second, we evaluated the clinical applicability of the nomogram through the method of decision curve analysis combining with the questionnaire of clinical practitioners. The decision curve showed that the nomogram was applicable when thresholds were in the range of .05–.65 due to the net benefit compared with the null model for its added value, while the results of questionnaire showed 91.5% of the clinicians had a threshold of intervention when predictive risk of POP ranged from 5% to 50%, which slickly conformed to the net-benefit threshold interval produced by the decision curve. It gives the presentation of the clinical utility of a predictive model more intuitively, making up for the deficiency of the traditional receiver operator characteristic curve.
The main limitations of this study include the following: firstly, it was a retrospective study, so the level of evidence was limited; secondly, it was programmed in a single institution, thus the sample size was small when compared to multi-center studies, and the selection of medical institution by patients might cause potential bias. Then, as the definition of the primary end point of this study—POP, the observation period of this study was limited to the duration of hospitalization, so the follow-up time was short.
Conclusion
This study shows that COPD, number of comorbidities, ASA classification >2, preoperative dependent functional status and cognitive impairment are independent risk factors of POP. The predictive nomogram established in this study shows a good accuracy in predicting the risk of POP and is applicable for clinical practice.
Acknowledgments
The authors of this research wish to thank the departments of Orthopedics and Record Room, Changhai Hospital affiliated to Second Military Medical University.
Appendix 1.
On-Line Questionnaire for Postoperative Pneumonia in geriatric patients with a hip fracture
1. I have obtained Identification of Physician’s Qualification for years. A. <3 B. 3∼5 C. 6∼10 D. >10
2. I am now a. A. resident doctor B. attending doctor C. associate chief physician D. archiater
3. I have been working as an orthopedist for years. A. <3 B. 3∼5 C. 6∼10 D. >10
4. The volume of my work place is a. A. Grade 3, A hospital B. Grade 3, B hospital C. Grade 2, A hospital D. Grade 2, B hospital
5. I have an experience of surgical treatment for geriatric hip fractures for. A. <50 cases B. 50∼100 cases C. 101∼300 cases D. >300 cases
6. In clinic practice, I have encountered POP in geriatric hip fracture patients. A. never B. rarely C. sometimes D. frequently
7. According to my threshold, if predicted probability for POP reaches, I will decide to intervene. A. 5%∼10% B. 11%∼25% C. 26%∼50% D. >50%
Footnotes
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval: This study was proved by Changhai Hospital of Shanghai with a waiver of requirement of patient consent because we covered patient data confidentiality and conducted this study in compliance with the Declaration of Helsinki.
Informed Consent: All patient data accessed complied with relevant data protection and privacy regulations. Following the national legislation and institutional requirements, written informed consent for participation was not required for this study.
ORCID iD
Da-ke Tong https://orcid.org/0000-0003-3257-3682
References
- 1.Marks R. Hip fracture epidemiological trends, outcomes, and risk factors, 1970-2009. Int J Gen Med. 2010;3(3):1-17. [PMC free article] [PubMed] [Google Scholar]
- 2.Cummings SR, Rubin SM, Black D. The future of hip fractures in the United States. Clin Orthop Relat Res. 1990;252:163-166. [PubMed] [Google Scholar]
- 3.Swart E, Vasudeva E, Makhni EC, Macaulay W, Bozic KJ. Dedicated perioperative hip fracture comanagement programs are cost-effective in high-volume centers: an economic analysis. Clin Orthop Relat Res. 2016. January;474(1):222-233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bhandari M, Devereaux PJ, Swiontkowski MF, et al. Internal fixation compared with arthroplasty for displaced fractures of the femoral neck. J Bone Jt Surg Am Vol. 2003;85:1673-1681. [DOI] [PubMed] [Google Scholar]
- 5.Sathiyakumar V, Avilucea FR, Whiting PS, et al. Risk factors for adverse cardiac events in hip fracture patients: an analysis of NSQIP data. Int Orthop. 2016. March;40(3):439-445. [DOI] [PubMed] [Google Scholar]
- 6.Sathiyakumar V, Greenberg SE, Jahangir AA, Mir HH, Obremskey WT, Sethi MK. Impact of type of surgery on deep venous thrombi and pulmonary emboli: a look at twenty seven thousand hip fracture patients. Int Orthop. 2015. October;39(10):2017-2022. [DOI] [PubMed] [Google Scholar]
- 7.Lv H, Yin P, Long A, et al. Clinical characteristics and risk factors of postoperative pneumonia after hip fracture surgery: a prospective cohort study. Osteoporos Int. 2016;27(10):3001-3009. [DOI] [PubMed] [Google Scholar]
- 8.Pincus D, Ravi B, Wasserstein D, et al. Association between wait time and 30-day mortality in adults undergoing hip fracture surgery. JAMA. 2017;318(20):1994-2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.American Thoracic Society (ATS) and Infectious Diseases Society of America (IDSA) . Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med. 2005;171:388-416. [DOI] [PubMed] [Google Scholar]
- 10.Dhanwal DK, Dennison EM, Harvey NC, Cooper C. Epidemiology of hip fracture: worldwide geographic variation. Indian J Orthop. 2011;45:15-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zuckerman JD. Hip fracture. N Engl J Med. 1996;334(23):1519-1525. [DOI] [PubMed] [Google Scholar]
- 12.Sathiyakumar V, Greenberg SE, Molina CS, Thakore RV, Obremskey WT, Sethi MK. Hip fractures are risky business: an analysis of the NSQIP data. Injury. 2015;46(4):703-708. [DOI] [PubMed] [Google Scholar]
- 13.Donegan DJ, Gay AN, Baldwin K, Morales EE, Esterhai JL, Jr, Mehta S. Use of medical comorbidities to predict complications after hip fracture surgery in the elderly. J Bone Jt Surg Am Vol. 2010. April;92(4):807-813. [DOI] [PubMed] [Google Scholar]
- 14.Vidán M, Serra JA, Moreno C, Riquelme G, Ortiz J. Efficacy of a comprehensive geriatric intervention in older patients hospitalized for hip fracture: a randomized, controlled trial. J Am Geriatr Soc. 2005;53:1476-1482. [DOI] [PubMed] [Google Scholar]
- 15.Bohl DD, Sershon RA, Saltzman BM, Darrith B, Della Valle CJ, Craig J. Incidence, risk factors, and clinical implications of pneumonia after surgery for geriatric hip fracture. J Arthroplasty. 2018;33(5):1552-1556. e1. [DOI] [PubMed] [Google Scholar]
- 16.Pedersen AB, Ehrenstein V, Szépligeti SK, Sørensen HT. Hip fracture, comorbidity, and the risk of myocardial infarction and stroke: a Danish nationwide cohort study, 1995-2015. J Bone Miner Res. 2017;V32N12:2339-2346. [DOI] [PubMed] [Google Scholar]
- 17.Huddleston JM, Gullerud RE, Smither F, et al. Myocardial infarction after hip fracture repair: a population-based study. J Am Geriatr Soc. 2012;60:2020-2026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Carnevale V, Fontana A, Scillitani A, Sinisi R, Romagnoli E, Copetti M. Incidence and all-cause mortality for hip fracture in comparison to stroke, and myocardial infarction: a fifteen years population-based longitudinal study. Endocrine. 2017;58(2):320-331. [DOI] [PubMed] [Google Scholar]
- 19.Durand WM, Goodman AD, Johnson JP, Daniels AH. Assessment of 30-day mortality and complication rates associated with extended deep vein thrombosis prophylaxis following hip fracture surgery. Injury. 2018;49(6):1141-1148. [DOI] [PubMed] [Google Scholar]
- 20.Zhang B-F, Xing W, Huang H, et al. Deep vein thrombosis in bilateral lower extremities after hip fracture, a retrospective study of 463 patients. Clin Interv Aging. 2018;13:681-689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lv H, Yin P, Long A, et al. Clinical characteristics and risk factors of postoperative pneumonia after hip fracture surgery: a prospective cohort study. Osteoporos Int. 2016;27(10):3001-3009. [DOI] [PubMed] [Google Scholar]
- 22.Patterson JT, Bohl DD, Basques BA, Arzeno AH, Grauer JN. Does preoperative pneumonia affect complications of geriatric hip fracture surgery?. Am J Orthoped. 2017;46(3):E177-E185. [PubMed] [Google Scholar]
- 23.Kalil AC, Metersky ML, Klompas M, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the infectious diseases society of America and the American thoracic society. Clin Infect Dis. 2016;63(5):e61-e111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chang SC, Lai JI, Lu MC, et al. Reduction in the incidence of pneumonia in elderly patients after hip fracture surgery: An inpatient pulmonary rehabilitation program. Med UK Ed. 2018;97(33):Article e11845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Graesslin O, Abdulkarim BS, Coutant C, et al. Nomogram to Predict Subsequent Brain Metastasis in Patients with Metastatic Breast Cancer; 2010. 2032-2037. [DOI] [PubMed] [Google Scholar]
- 26.Tsoutsou PG, Jeanneret Sozzi W, Matzinger O, Ozsahin M. Nomograms predicting locoregional recurrence in the subtype era of breast cancer. J Clin Oncol. 2013;31(5):647-648. [DOI] [PubMed] [Google Scholar]
- 27.Li J, Liu Y, Yan Z, et al. A nomogram predicting pulmonary metastasis of hepatocellular carcinoma following partial hepatectomy. Br J Cancer. 2014;110(5):1110-1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Semenkovich TR, Yan Y, Subramanian M, et al. A clinical nomogram for predicting node-positive disease in esophageal cancer. Ann Surg. 2019;273(3):e214-e221. doi: 10.1097/SLA.0000000000003450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang X, Tong D-k., Ji F, et al. Predictive nomogram for postoperative delirium in elderly patients with a hip fracture. Injury. 2019;50(2):392-397. [DOI] [PubMed] [Google Scholar]