Abstract
Objective
Autoimmune inner ear disease (AIED) is a rare disorder characterized by rapidly progressive, sensorineural hearing loss that demonstrates good responsiveness to corticosteroid and immunosuppressive therapy. The pathophysiology is likely driven by chronic trafficking of immune cells into the inner ear, targeting inner ear proteins to coordinate inflammation. Suppression or modulation of the immune response can minimize cochleitis allowing for potential recovery of hearing. It is an otologic emergency requiring a multidisciplinary approach to management to commence immunosuppressive therapy. This can be achieved using steroids, immunomodulators, plasmapheresis, intravenous immunoglobulin, or biologic agents. Treatment decisions are further complicated in pregnancy and require supervision by an obstetrician and maternal-fetal medicine (MFM) specialist. Concerns include safe dosing of steroids and potential for transplacental migration of immune complexes. We provide the first comprehensive literature review on AIED and its implications in pregnancy. We frame our discussion in the context of the second reported case of primary AIED in pregnancy and the first to show excellent response to immunosuppressive therapy.
Methods
We reviewed the presented case and literature on AIED.
Results
A 27-year-old, pregnant, HSP-70 positive woman was diagnosed with AIED and had excellent recovery of hearing and balance following a combination of steroid treatment, augmented by oral immunomodulators, plasmapheresis, and IVIG.
Conclusion
AIED is a diagnostic challenge, and treatment considerations are complex when encountered in pregnancy. Management requires multidisciplinary involvement between otolaryngologists, immunologists, and obstetricians to balance maternal and fetal health outcomes.
1. Introduction
Autoimmune inner ear disease (AIED) is an exceptionally rare occurrence during pregnancy. It presents challenging diagnostic and management paradigms. AIED is defined as rapidly progressive, typically bilateral, hearing loss that demonstrates responsiveness to corticosteroid and immunosuppressive therapy as demonstrated by McCabe in 1979 [1, 2]. It was first described by Lehnhardt and Cogan in 1958 [3, 4]. Clinically, symptoms develop over weeks to months, which is too rapid for presbycusis and too gradual for sudden SNHL.
AIED may be primary, in which the disease process is confined to the inner ear, or secondary, accounting for 30% of cases, in which cochleovestibular damage occurs in the context of a systemic autoimmune process with circulating immune complexes [5]. Inner ear involvement secondary to multisystem inflammation has been seen in multiple conditions including Cogan's syndrome, rheumatologic conditions, and granulomatous disease [5]. The underlying immunological mechanisms are not well elucidated in the literature [6]. Autoimmune conditions may manifest during pregnancy, triggered by hormonal and physiological changes [7]. AIED should be recognized as an otologic emergency with urgent multidisciplinary management coordinated by otolaryngologists and immunologists in initiating immunosuppressive treatment that improves chance of hearing and balance recovery. Disease manifesting in pregnancy requires specialty input from obstetricians and MFM specialists [8] as treatment considerations must balance maternal and fetal health.
2. Case Presentation
A 27-year-old pregnant female, in the 19th week of gestation, presented to the outpatient ENT clinic with a 1-week history of bilateral SNHL, vertigo, and tinnitus. She had presented to the emergency department the previous week with flu-like symptoms, headache, photophobia, and neck stiffness, when she was discharged with a presumptive diagnosis of migraines. She was otherwise well with no medical history or regular medications. Her obstetric history was G9P1, including 6 miscarriages, 2 terminations, and 1 term pregnancy. There was no personal or family history of autoimmune disease, vasculitis, thrombophilia, or genetic conditions.
Otoscopy revealed patent external auditory canals, intact and noninflamed tympanic membranes, and no evidence of middle ear pathology. She was unsteady in her gait. There was horizontal nystagmus on left lateral gaze, and head impulse testing was positive to the left. Free field voice testing indicated bilateral hearing impairment. Tuning fork assessment was equivocal for Weber's test and Rinne's positive bilaterally. The remainder of the neurological examination was unremarkable.
Baseline pure tone audiogram showed bilateral moderate sensorineural hearing loss with flat morphology, affecting low and high frequencies (Figure 1). Speech discrimination was good with an appropriate level of amplification.
Figure 1.
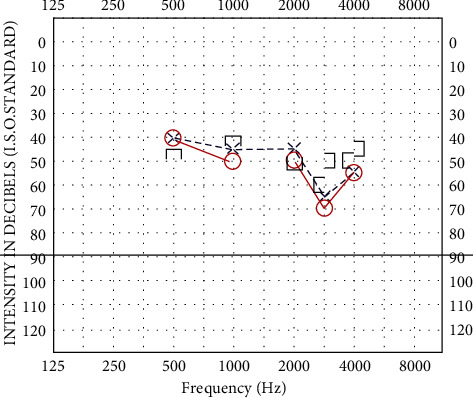
Audiogram pretreatment: bilateral mild sloping to moderate-severe sensorineural hearing loss.
The provisional diagnosis of bilateral acute cochleovestibular inflammation was made, and urgent multidisciplinary consultations were sought from obstetrics, MFM, neurology and immunology.
The lumbar puncture showed CSF-restricted oligoclonal bands and pleocytosis with mononuclear cells (Table 1). Autoimmune encephalitis was considered based on Graus criteria [9].
Table 1.
CSF studies.
| Cerebrospinal fluid analysis | ||
|---|---|---|
| CSF glucose | 2.8 mmol/L | 2.2–3.9 |
| CSF protein | 0.43 g/L | 0.15–0.45 |
| CSF LDH | 24 U/L | |
| CSF cell count | ||
| Leukocytes | 36 × 106/L | |
| Erythrocytes | 2 × 106/L | |
| Polymorphonuclear cells | Nil | |
| Mononuclear cells | 36 × 106/L | |
| Oligoclonal bands | Positive, CSF restricted | |
| Culture | No bacterial growth | |
| Mycobacterial culture | Negative | |
| Mycobacterial NAT | Negative | |
| Nucleic acid testing for EBV, VZV CMV, enterovirus, HSV, and polyomavirus | Negative | |
| Cerebrospinal fluid immunologic tests | ||
| Neuronal antibodies: Purkinje (Yo), PCA 2, ANNA-1, ANNA-2, Ma 1, Ma 2, amphiphysin, CV2, Tr, SOX-1 | Negative | |
| NMDA receptor antibodies | Negative | |
| CASPR2 antibodies | Negative | |
| LGI-1 antibodies | Negative | |
| GABA-B antibodies | Negative | |
| DPPX antibodies | Negative | |
| IgLON5 antibodies | Negative | |
| Albumin | 0.21 g/L | 0–0.35 |
| Immunoglobulin G | 0.03 g/L | 0–0.03 |
| IgG/albumin ratio | 0.14 | 0–0.25 |
An urgent MRI brain and spine was arranged which showed normal cerebellopontine angles, vestibulocochlear nerves, labyrinthine signal intensity, and architecture, with the absence of demyelination features. Serial imaging taken 2 weeks apart excluded interval cochlear fluid signal changes. Blood tests showed elevated acute phase reactants and excluded viral etiology (Tables 2–4). Autoimmune screen returned speckled antinuclear antibodies in 1 : 640 and positive HSP-70 antibodies (Table 5). The diagnosis of primary AIED was established.
Table 2.
Baseline laboratory tests.
| Full blood count | ||
| Haemoglobin | 127 g/L | 115–165 |
| White cell count | 5.5 × 109/L | 4.0–11.0 |
| Platelets | 159 × 109/L | 150–400 |
| Haematocrit | 0.37 L/L | 0.36–0.44 |
| Mean corpuscular volume | 87 fL | 82–98 |
| Mean corpuscular Hb | 30 pg | 27–32 |
| Mean corpuscular Hb concentration | 345 g/L | 300–350 |
| Red cell distribution width | 12.3% | 11.0–15.0 |
|
| ||
| Serum biochemistry | ||
| Sodium | 136 mmol/L | 135–145 |
| Potassium | 3.6 mmol/L | 3.2–5.0 |
| Chloride | 107 mmol/L | 95–110 |
| Bicarbonate | 20 mmol/L | 22–32 |
| Urea | 2.7 mmol/L | 2.5–6.5 |
| Creatinine | 52 umol/L | 45–90 |
| eGFR | >90 mL/min/1.73 m2 | >90 |
| Calcium | 2.42 mmol/L | 2.15–2.55 |
| Magnesium | 0.83 mmol/L | 0.70–1.10 |
| Phosphate | 1.09 mmol/L | 0.75–1.50 |
| C-reactive protein | <3 mg/L | <3 |
| Erythrocyte sedimentation rate | 50 mm/hr | 3–19 |
Table 3.
Viral and infectious diseases serology.
| Viral serology | ||
| CMV IgG | Not detected | |
| CMV IgM | Not detected | |
| EBV IgG | Detected | |
| EBV IgM | Equivocal | |
| HSV1 IgG | Detected | |
| HSV2 IgG | Not detected | |
| HSV IgM | Not detected | |
| Varicella zoster IgG | Detected | |
| Varicella zoster IgM | Not detected | |
| HIV Ag/Ab screen | Not detected | |
| HBsAg | Not detected | |
| HCV antibody | Not detected | |
| Ross River virus IgM | Not detected | |
| Barmah Forest virus IgM | Not detected | |
| Arbovirus serology | Not detected | |
|
| ||
| Infectious disease serology | ||
| Syphilis EIA total antibody | Nonreactive | |
| Anti-DNase B titre | <100 | <200 |
| Antistreptolysin O titre | 91.4 IU/mL | <200 |
| TB gamma interferon | Not detected | |
Table 4.
Metabolic and endocrinologic profile.
| Metabolic and endocrine profile | ||
|---|---|---|
| Iron level | 5.9 umol/L | 7.0–29.0 |
| Transferrin | 2.7 g/L | 1.8–3.3 |
| Transferrin saturation | 10% | 10–45 |
| Ferritin | 29 ug/L | 15–150 |
| HbA1c% | 5.0% | 4.0–6.0 |
| Vitamin B12 | 117 pmol/L | >150 |
| Holotranscobalamin level | 61 pmol/L | >36 |
| Serum folate | 18.9 nmol/L | >10.0 |
| TSH | 2.57 mIU/L | 0.40–3.50 |
| Free thyroxine | 14.0 pmol/L | 9.0–19.0 |
| Angiotensin converting enzyme | 32 UL | 20–70 |
| Urate | 0.18 mmol/L | 0.12–0.38 |
| Homocysteine | 6.2 umol/L | 4.5–13.5 |
| Chromogranin A | 1.9 nmol/L | <3.0 |
| Neopterin | 12 nmol/L | 0–13 |
| Total bile acids | 8 umol/L | ≤8.0 |
| Apolipoprotein A1 | 1.68 g/L | 1.11–2.09 |
| Apolipoprotein B | 1.18 g/L | 0.63–1.32 |
| Apolipoprotein A1/B ratio | 1.42 | 0.87–2.85 |
| Lipoprotein EPG | Normal limits | |
Table 5.
Immunologic and cell markers, including flow cytometry profile and genetic studies.
| Cell markers | ||
| CD19 pan B cell | 7% | |
| CD20 mature B cell | 7% | |
| Kappa light chain | 3% | |
| Lambda light chain | 3% | |
| CD3 T cells | 69% | |
| CD4 helper subset | 34% | |
| CD8 cytotoxic subset | 32% | |
| Flow comment | No evidence of lymphoproliferative disease | |
|
| ||
| Immunologic markers | ||
| Rheumatoid factor | <10 IU/mL | <15 |
| Anti-CCP antibodies | 3 U/mL | <5 |
| IgG | 8.7 g/L | 6.6–15.6 |
| IgA | 1.73 g/L | 0.75–3.80 |
| IM | 1.56 g/L | 0.40–3.10 |
| IgE | 19 U/mL | <113 |
| IgG1 | 3.76 g/L | 3.92–9.12 |
| IgG2 | 2.60 g/L | 1.50–6.40 |
| IgG3 | 0.48 g/L | 0.25–1.38 |
| IgG4 | 0.27 g/L | 0.04–0.70 |
| C1Q complement component | 155 mg/L | 118–244 |
| C2 complement | 29.9 mg/L | 14.0–5.0 |
| C3 complement | 1.30 g/L | 0.74–1.57 |
| C4 complement | 0.28 g/L | 0.13–0.41 |
| C5 complement | >200.0 mg/L | 100–169 |
| C6 complement | 120 mg/L | 45–96 |
| C7 complement | >110 mg/L | 55–85 |
| C8 complement | 172.0 mg/L | 112–172 |
| C9 complement | 500.0 mg/L | 125–265 |
| Haptoglobin | 1.53 g/L | 0.30–2.15 |
| A1 antitrypsin level | 2.38 g/L | 0.90–1.90 |
| Serum total protein | 63 g/L | 46–70 |
| Albumin EPG | 36 g/L | 37–51 |
| Alpha1 globulin | 2.9 g/L | 0.9–2.0 |
| Alpha2 globulin | 8.9 g/L | 2.8–7.7 |
| Beta globulin | 8.9 g/L | 5.1–14.0 |
| Gamma globulin | 6.9 g/L | 5.1–14.0 |
| B2 microglobulin | 1.9 mg/L | 1.0–2.6 |
| Glutamic acid decarboxylase antibody | <5 U/mL | ≤5 |
| Islet cell antigen 512 antibodies | <8 U/mL | 0–15 |
| Nuclear antibodies | Detected 1 : 640 speckled | |
| dsDNA antibodies | Pattern | |
| RNP antibodies | 7 IU/mL | 0–29 |
| SM (Smith) antigen antibodies | Not detected | |
| SSA/Ro 60 antibodies | Not detected | |
| Ro-52/TRIM 21 antibodies | Not detected | |
| SSB/La antibodies | Not detected | |
| Scleroderma 70 antibodies | Not detected | |
| Jo-1 antibodies | Not detected | |
| Neuronal antibodies: Purkinje | Not detected | |
| (Yo), PCA 2, ANNA-1, ANNA-2, Ma 1, Ma 2, amphiphysin, CV2, Tr, and SOX-1 | Not detected | |
| Smooth muscle antibodies | Not detected <1 : 40 | |
| Mitochondrial antibodies | Not detected <1 : 40 | |
| Parietal cell antibodies | Not detected <1 : 40 | |
| Intrinsic factor antibody | 4 U/mL | <20 |
| Thyroglobulin antibodies | <20 IU/mL | 0–60 |
| Thyroid peroxidase antibodies | <10 IU/mL | 0–35 |
| Heat shock protein 70 antibodies | Detected A | |
| Pancreatic islet cell antibodies | Not detected | |
| Gliadin IgG deamidated antibodies | 2 units | <20 |
| Transglutaminase IgA antibodies | <1 U/mL | <4 |
| Neutrophil cytoplasm PR3 and MPO antibodies | Not detected | |
| Ro-52, Mi-2, Ku, PM-Scl 100, PM-Scl 75, SRP, EJ, OJ, Jo-1, PL-7, and PL-12 | Negative | |
| Zinc transporter 8 antibodies | Negative <10 U/mL | |
|
| ||
| Genetic diagnostics | ||
| Thiopurine methyltransferase genotype | No TMPT variant alleles | |
| Factor V DNA | No factor V Leiden mutation | |
Systemic steroid treatment was commenced immediately following neurology, immunology, and MFM consultations. The patient received intravenous methylprednisolone pulsing with 1 g daily for three days. She received intravenous acyclovir 900 mg three times daily for three days as prophylactic HSV coverage on advice from neurologists. Obstetric and MFM advised on safety of medications in pregnancy. She received one dose of intratympanic dexamethasone, by which state there was marked improvement in her balance and hearing thresholds (Figure 2). Ophthalmology review excluded ocular inflammation. She was discharged on 100 mg daily azathioprine and 50 mg daily prednisolone, which was slowly weaned under immunology guidance. She remained under the combined supervision of the MFM, immunology, and ENT disciplines throughout her pregnancy. Her hearing and balance remained stable on maintenance therapy. Due to concerns of poor fetal growth, she received five sessions of plasmapheresis followed by monthly IVIG therapy.
Figure 2.
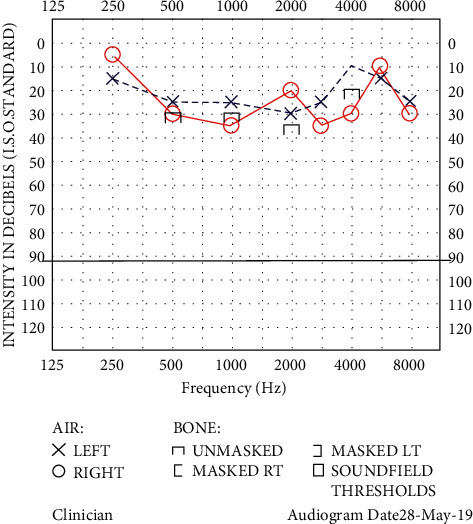
Audiogram posttreatment: mild sensorineural hearing loss (right greater than the left).
A healthy baby was born at term. The patient's newborn initially failed the newborn hearing test but passed the following week. There are no further concerns regarding the toddler's hearing. At three years, the patient remains on maintenance immunomodulator therapy, supervised by her immunologist, with azathioprine 100 mg and sirolimus 1 mg daily. Her recent audiogram showed hearing recovery stabilized to baseline (Figure 3).
Figure 3.
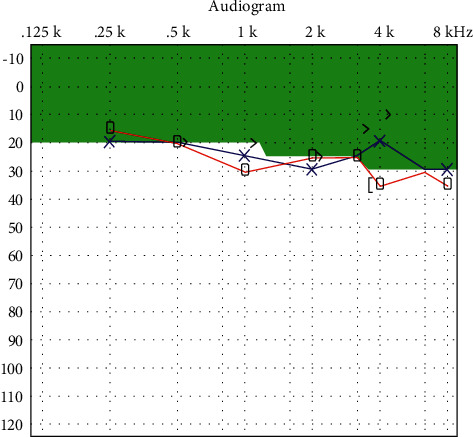
Audiogram two years posttreatment: hearing thresholds returned to baseline with mild sensorineural hearing loss at higher frequencies.
3. Discussion
3.1. Clinical Manifestations and Diagnosis of AIED
AIED is a rare entity first described by McCabe in a case series of 18 patients with progressive bilateral SNHL without identifiable etiology who were steroid responsive [1]. The incidence of AIED is estimated at <5 cases per 100,000 with an estimated prevalence of 45000 in the US [10]. Cochlear neuritis may initially be unilateral and then progress to the other ear, with patients often showing asymmetric audiometric profiles. The pattern of hearing loss can be fluctuant, but generally deteriorates with time. Vestibular involvement is common with 50% of patients experiencing ataxia or positional vertigo [5]. Balance disorders may be underestimated due to the slow development of vestibular dysfunction and compensation by somatosensory and visual systems [11]. Aural fullness and tinnitus are reported in 25–50% of patients [2].
Due to its low incidence combined with our limited understanding of pathophysiology and inability to establish reliable biomarkers, there is no formal diagnostic criterion [12]. AIED is a diagnosis of exclusion in a patient with a suggestive history, favorable response to immunosuppression, or known autoimmune disease. Diagnosis is suspected on audiogram with bilateral sensorineural hearing loss of at least 30 dB at any frequency and evidence of progression in at least one ear on two serial audiograms performed 3 months apart [13, 14]. Serial audiograms are required as the pattern of sensorineural hearing loss may be fluctuant and to monitor response to steroid therapy. The audiometric pattern is an important prognostic indicator for recovery of hearing. Isolated low-frequency hearing loss is associated with better treatment outcomes compared to a flat audiogram or high-frequency losses [15]. The severity of hearing loss prior to treatment has an inverse correlation with recovery. All patients should undergo MRI evaluation to exclude retrocochlear and demyelinating pathology.
3.2. Serological Markers in AIED
Laboratory testing can include nonspecific markers of inflammation and autoimmunity including C-reactive protein, erythrocyte sedimentation rate, antinuclear antibody, and complement protein. Specific inner ear antigen tests include the migration inhibition test (MIT), lymphocyte transformation test (LTT), and Western blot analysis. The MIT and LTT have inherent technical difficulties, and results may be heterogeneous [12]. More specific tests for autoreactivity to inner ear antigens such as HSP-70 should be considered. HSP-70 is a constitutively expressed protein that is upregulated in conditions of stress and found in the spiral limbus, spiral prominence, and organ of Corti within the inner ear, and in peripheral organs [6]. Suspicion of AIED should be raised in HSP-70 positive patients; however, it may also be present in the general population and in Meniere's disease—hence, its use as a biomarker is disputed. A diagnostic dilemma between AIED and Meniere's disease exists, and spontaneous recovery of hearing in Meniere's disease may be mistaken as a positive response to immunosuppression in AIED [16]. An extensive immunologic workup is not mandated and should be coordinated by an immunologist with expertise in interpretation of results, as testing can be expensive [17].
3.3. Pathogenesis of AIED
Our current understanding regarding the immunological basis of AIED is limited. The inner ear is not immunologically privileged as previously thought, and inflammatory stimuli can recruit immune competent lymphocytes into the labyrinth and endolymphatic sac [18]. Cells enter the scala tympani via the spiral modiolar vein, coordinating labyrinthitis [2, 5, 6, 12]. Studies have shown the presence of autoantibodies directed against inner ear proteins such as HSP-70, cochlin (spiral ganglion), type II collagen (spiral ligament, endolymphatic duct), and KHRI-3 (otolith organs and endolymphatic sac) [2, 6]. Chronic and persistent trafficking of immune cells into the inner ear results in destructive changes including cochlear ischemia and fibrosis, spiral ganglion degeneration, otic capsule spongiosis, and endolymphatic hydrops [2, 5]. Rarely, secondary AIED may induce arthritis of the incudostapedial joint causing conductive hearing loss [6]. Molecular mimicry and shared epitopes between viral antigens and inner ear proteins can cause cross-reactivity, directing an autoimmune response following viral infection [5, 12, 19]. Although plausible, there is currently no evidence of positive viral serology in AIED.
3.4. Treatment Modalities in AIED
Corticosteroids remain the primary medical therapy in autoimmune SNHL, with activation of glucocorticoid receptors within the cochlear hair cells causing downregulation of local cytokines and reducing autoantibody production and inflammation [20]. This attenuates cochleitis and hair cell death, with potential recovery of function. Steroids increase microvascular blood flow in the cochlea and reduce endolymphatic hydrops. Almost 70–90% are steroid responsive, although adjunctive immunosuppressive agents may be indicated in relapses of hearing loss during the maintenance or steroid weaning phase [14]. Azathioprine, a purine analog, has demonstrated improvement in hearing in combination with prednisone in a cohort study of 12 patients [21]. We used azathioprine because of compatibility with pregnancy. In later pregnancy after organogenesis, cyclophosphamide or tacrolimus can be considered [22]. Management of vestibular dysfunction and balance disorders involves balance physiotherapy and treatment of the autoimmune process.
Plasmapheresis removes circulating immune complexes and autoantibodies and is an adjunct in managing autoimmune diseases [5]. It can be considered in severe hearing impairment or as salvage treatment in steroid-resistant cases and involves thrice weekly treatment for 2 weeks and then weekly for four weeks [23]. IVIG therapy provides immunoregulatory action by altering complement-mediated destruction, neutralizing autoantibodies, and modulating autoreactive B cells [24].
Nonsteroidal immunomodulators and biologics have been used in treatment in patients who are not candidates for high-dose steroid therapy, who fail first-line steroid therapy, and to encourage steroid weaning. A recent systematic review revealed steroid nonresponders may benefit from biologics, although studies are limited by small cohorts and variable efficacy [20]. TNF-alpha inhibitors such as systemic etanercept 25 mg twice weekly have shown significant improvement in hearing thresholds [25]. Local instillation of infliximab weekly for 4 weeks revealed stable improvement in hearing at 10–38 weeks after treatment [26]. Anakinra, an interleukin-1β blocker, demonstrated improvement in hearing thresholds in a clinical trial of 14 patients with AIED who were nonresponders to steroids [27]. Rituximab, a CD20 antagonist, has been shown to reduce corticosteroid dosage and improve symptoms in AIED [28]. It has demonstrated ability to maintain hearing improvement after corticosteroid use [29], but it has limited use after the first trimester of pregnancy [30].
Patients with destructive inner ear changes have poor outcomes, even with maximal medical management [6, 17, 31]. Time sensitive cochlear implantation should be considered in cases when useful hearing is not restored and maintained [31]. Cochlear implantation does not address the underlying disease process and can be challenging in immune-mediated labyrinthitis ossificans and intracochlear fibrosis [32]. Future treatment strategies include gene and stem cell therapy aimed at neuronal preservation within the cochlea and hair cells [33]. In vivo models using human adipose-derived mesenchymal stem cells demonstrated improvement in hearing in AIED [34].
3.5. AIED in Pregnancy
Therapeutic decisions in pregnancy are complicated by the potential risk of treatment on maternal and fetal outcomes and the concern for autoimmune targeting of the fetal inner ear. Combination modalities should be incorporated to minimize systemic corticosteroid dosing. Long-term, high-dose steroid therapy has an unfavorable side effect profile. The literature is inconclusive on the safety of prolonged steroid administration in pregnancy, and there is currently insufficient evidence to suggest an increased risk of fetal malformations or gestational diabetes mellitus [35]. Multidisciplinary discussion between otolaryngology, immunology, and MFM facilitates sharing of expertise and ensuring optimal management of the immune-mediated hearing loss without compromising maternal or fetal outcomes. Due to limited evidence on the management of AIED, a collaborative approach is preferred [14].
The obstetrician provides expertise to guide safe dosing of steroids in pregnancy and whether steroid-sparing or steroid-minimizing treatment should be utilized. Alternate options include intratympanic dexamethasone, oral immunomodulator therapy, plasmapheresis, and IVIG [20]. Intratympanic steroid injection can achieve higher perilymph steroid concentrations compared to oral or intravenous administration [36, 37]. Systemic therapy is indicated to ameliorate the underlying multisystem autoimmune response. In pregnancy, there is concern for transplacental transfer of autoantibodies to the fetus-plasmapheresis or IVIG may provide benefit. Similar to our case, one study reported a higher incidence of abnormal auditory brainstem response findings in the children of women who developed AIED during pregnancy [38]. Hence, we must recognize that autoimmune hearing loss in pregnancy may also have implications on the newborn's hearing. There are no data on whether the hearing deficits persisted in childhood.
Hill et al. described the first case of primary AIED in pregnancy, reporting that oral steroids minimized further deterioration in hearing, but the patient did not recover to baseline levels [8]. In that case, the patient presented late and treated only with oral prednisone. In our case, early initiation of intensive steroids, including pulsed methylprednisolone and intratympanic dexamethasone, and multimodal long-term treatment showed great efficacy. Cogan's syndrome, a form of secondary AIED characterized by bilateral cochleovestibulitis and keratitis, has been described in pregnancy in two case reports. One study maintained disease stability with hydroxychloroquine and prednisone, whilst the other treated symptomatic flares with oral and ophthalmic steroids [39, 40].
4. Conclusion
AIED is an otologic emergency that rarely manifests during pregnancy and requires urgent treatment, supervised by otolaryngologists, immunologists, and MFM specialists. Multimodal combination therapy is effective and minimizes long-term systemic corticosteroid exposure. We report a rare case of primary AIED in pregnancy with remarkable hearing outcomes following combination (IV, oral, and intratympanic) steroid therapy, augmented by immunomodulators, plasmapheresis, and IVIG.
Abbreviations
- AIED:
Autoimmune inner ear disease
- CSF:
Cerebrospinal fluid
- ENT:
Ear, nose, and throat
- HSP:
Heat shock protein
- HSV:
Herpes simplex virus
- IVIG:
Intravenous immunoglobulin
- MRI:
Magnetic resonance imaging
- MFM:
Maternal-fetal medicine
- MIT:
Migration inhibition test
- LIT:
Lymphocyte transformation test
- SNHL:
Sensorineural hearing loss
- TNF:
Tumour necrosis factor.
Data Availability
No data were used to support this study.
Ethical Approval
This study does not require ethical committee review as per the Nepean Hospital Human Research Ethics Committee (HREC).
Consent
The subject of the case report provided written informed consent for the discussion of her case in the literature.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Supplementary Materials
The CARE checklist for presentation of a case report is provided.
References
- 1.McCabe B. F. Autoimmune sensorineural hearing loss. Annals of Otology, Rhinology & Laryngology . 1979;88(5):585–589. doi: 10.1177/000348947908800501. [DOI] [PubMed] [Google Scholar]
- 2.Ciorba A., Corazzi V., Bianchini C., et al. Autoimmune inner ear disease (AIED): a diagnostic challenge. International Journal of Immunopathology and Pharmacology . 2018;32 doi: 10.1177/2058738418808680.2058738418808680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lehnhardt E. Sudden hearing disorders occurring simultaneously or successively on both sides. Journal of Laryngology, Rhinology and Otology . 1958;37(1):1–16. [PubMed] [Google Scholar]
- 4.Norton E. W. D., Cogan D. G. Syndrome of nonsyphilitic interstitial keratitis and vestibuloauditory symptoms; a long-term follow-up. A.M.A. Archives of Ophthalmology . 1959;61(5):695–697. doi: 10.1001/archopht.1959.00940090697004. [DOI] [PubMed] [Google Scholar]
- 5.Bovo R., Aimoni C., Martini A. Immune-mediated inner ear disease. Acta Oto-Laryngologica . 2006;126(10):1012–1021. doi: 10.1080/00016480600606723. [DOI] [PubMed] [Google Scholar]
- 6.Das S., Bakshi S. S., Seepana R. Demystifying autoimmune inner ear disease. European Archives of Oto-Rhino-Laryngology . 2019;276(12):3267–3274. doi: 10.1007/s00405-019-05681-5. [DOI] [PubMed] [Google Scholar]
- 7.Angum F., Khan T., Kaler J., Siddiqui L., Hussain A. The prevalence of autoimmune disorders in women: a narrative review. Cureus . 2020;12(5) doi: 10.7759/cureus.8094.e8094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hill F. C. E., Iseli C., O’Leary S. Primary autoimmune inner ear disease in pregnancy. Australian Journal of Oto-Laryngology . 2018;1 doi: 10.21037/ajo.2018.09.06. [DOI] [Google Scholar]
- 9.Graus F., Titulaer M. J., Balu R., et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurology . 2016;15(4):391–404. doi: 10.1016/s1474-4422(15)00401-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vambutas A., Pathak S. AAO: autoimmune and autoinflammatory (disease) in otology: what is new in immune-mediated hearing loss. Laryngoscope Investigative Otolaryngology . 2016;1(5):110–115. doi: 10.1002/lio2.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ralli M., D’Aguanno V., Di Stadio A., et al. Audiovestibular symptoms in systemic autoimmune diseases. Journal of Immunology Research . 2018;2018:14. doi: 10.1155/2018/5798103.5798103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bovo R., Ciorba A., Martini A. The diagnosis of autoimmune inner ear disease: evidence and critical pitfalls. European Archives of Oto-Rhino-Laryngology . 2009;266(1):37–40. doi: 10.1007/s00405-008-0801-y. [DOI] [PubMed] [Google Scholar]
- 13.Rauch S. D. Clinical management of immune-mediated inner-ear disease. Annals of the New York Academy of Sciences . 1997;830:203–210. doi: 10.1111/j.1749-6632.1997.tb51891.x. [DOI] [PubMed] [Google Scholar]
- 14.Mijovic T., Zeitouni A., Colmegna I. Autoimmune sensorineural hearing loss: the otology-rheumatology interface. Rheumatology . 2013;52(5):780–789. doi: 10.1093/rheumatology/ket009. [DOI] [PubMed] [Google Scholar]
- 15.Choo O.-S., Yang S. M., Park H. Y., et al. Differences in clinical characteristics and prognosis of sudden low- and high-frequency hearing loss. Laryngoscope . 2016;127(8):1878–1884. doi: 10.1002/lary.26382. [DOI] [PubMed] [Google Scholar]
- 16.Ruckenstein M. J. Autoimmune inner ear disease. Current Opinion in Otolaryngology & Head and Neck Surgery . 2004;12(5):426–430. doi: 10.1097/01.moo.0000136101.95662.aa. [DOI] [PubMed] [Google Scholar]
- 17.García-Berrocal J. R., Trinidad A., Ramírez-Camacho R., Lobo D., Verdaguer J. M., Ibáñez A. Immunologic work-up study for inner ear disorders: looking for a rational strategy. Acta Oto-Laryngologica . 2005;125(8):814–818. doi: 10.1080/00016480510038059. [DOI] [PubMed] [Google Scholar]
- 18.Yehudai D., Shoenfeld Y., Toubi E. The autoimmune characteristics of progressive or sudden sensorineural hearing loss. Autoimmunity . 2006;39(2):153–158. doi: 10.1080/08916930500499599. [DOI] [PubMed] [Google Scholar]
- 19.Berrocal J. R. G., Ramírez-Camacho R. Sudden sensorineural hearing loss: supporting the immunologic theory. Annals of Otology, Rhinology & Laryngology . 2002;111(11):989–997. doi: 10.1177/000348940211101107. [DOI] [PubMed] [Google Scholar]
- 20.Breslin N. K., Varadarajan V. V., Sobel E. S., Haberman R. S. Autoimmune inner ear disease: a systematic review of management. Laryngoscope Investig Otolaryngol . 2002;5(6):1217–1226. doi: 10.1002/lio2.508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Saraçaydin A., Katircioğlu S., Katircioğlu S., Karatay M. C. Azathioprine in combination with steroids in the treatment of autoimmune inner-ear disease. Journal of International Medical Research . 1993;21(4):192–196. doi: 10.1177/030006059302100404. [DOI] [PubMed] [Google Scholar]
- 22.Götestam Skorpen C., Hoeltzenbein M., Tincani A., et al. The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Annals of the Rheumatic Diseases . 2016;75(5):795–810. doi: 10.1136/annrheumdis-2015-208840. [DOI] [PubMed] [Google Scholar]
- 23.Luetje C. M. Theoretical and practical implications for plasmapheresis in autoimmune inner ear disease. Laryngoscope . 1989;99(11):1137–1146. doi: 10.1288/00005537-198911000-00006. [DOI] [PubMed] [Google Scholar]
- 24.Gilardin L., Bayry J., Kaveri S. V. Intravenous immunoglobulin as clinical immune-modulating therapy. Canadian Medical Association Journal . 2015;187(4):257–264. doi: 10.1503/cmaj.130375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cohen S., Shoup A., Weisman M. H., Harris J. Etanercept treatment for autoimmune inner ear disease: results of a pilot placebo-controlled study. Otology & Neurotology . 2005;26(5):903–907. doi: 10.1097/01.mao.0000185082.28598.87. [DOI] [PubMed] [Google Scholar]
- 26.Van Wijk F., Staecker H., Keithley E., Lefebvre P. P. Local perfusion of the tumor Necrosis factor α blocker infliximab to the inner ear improves autoimmune neurosensory hearing loss. Audiology and Neuro-Otology . 2006;11(6):357–365. doi: 10.1159/000095897. [DOI] [PubMed] [Google Scholar]
- 27.Pathak S., Goldofsky E., Vivas E. X., Bonagura V. R., Vambutas A. IL-1β is overexpressed and aberrantly regulated in corticosteroid nonresponders with autoimmune inner ear disease. Journal of Immunology . 2011;186(3):1870–1879. doi: 10.4049/jimmunol.1002275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Matsuoka A. J., Harris J. P. Autoimmune inner ear disease: a retrospective review of forty-seven patients. Audiology and Neuro-Otology . 2013;18(4):228–239. doi: 10.1159/000351289. [DOI] [PubMed] [Google Scholar]
- 29.Cohen S., Roland P., Shoup A., et al. A pilot study of rituximab in immune-mediated inner ear disease. Audiology and Neuro-Otology . 2007;16(4):214–221. doi: 10.1159/000320606. [DOI] [PubMed] [Google Scholar]
- 30.Østensen M. The use of biologics in pregnant patients with rheumatic disease. Expert Review of Clinical Pharmacology . 2017;10(6):661–669. doi: 10.1080/17512433.2017.1305268. [DOI] [PubMed] [Google Scholar]
- 31.Shamriz O., Tal Y., Gross M. Autoimmune inner ear disease: immune biomarkers, audiovestibular aspects, and therapeutic modalities of cogan’s syndrome. Journal of immunology research . 2018;2018:8. doi: 10.1155/2018/1498640.1498640 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang J. R., Yuen H. W., Shipp D. B., et al. Cochlear implantation in patients with autoimmune inner ear disease including cogan syndrome: a comparison with age- and sex-matched controls. Laryngoscope . 2010;120(12):2478–2483. doi: 10.1002/lary.21060. [DOI] [PubMed] [Google Scholar]
- 33.Chien W. W., Monzack E. L., McDougald D. S., Cunningham L. L. Gene therapy for sensorineural hearing loss. Ear and Hearing . 2015;36(1):1–7. doi: 10.1097/aud.0000000000000088. [DOI] [PubMed] [Google Scholar]
- 34.Zhou Y., Yuan J., Zhou B., et al. The therapeutic efficacy of human adipose tissue-derived mesenchymal stem cells on experimental autoimmune hearing loss in mice. Immunology . 2011;133(1):133–140. doi: 10.1111/j.1365-2567.2011.03421.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bandoli G., Palmsten K., Forbess Smith C. J., Chambers C. D. A review of systemic corticosteroid use in pregnancy and the risk of select pregnancy and birth outcomes. Rheumatic Disease Clinics of North America . 2017;43(3):489–502. doi: 10.1016/j.rdc.2017.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Parnes L. S., Sun A. H., Freeman D. J. Corticosteroid pharmacokinetics in the inner ear fluids: an animal study followed by clinical application. Laryngoscope . 1999;109(7):1–17. doi: 10.1097/00005537-199907001-00001. [DOI] [PubMed] [Google Scholar]
- 37.Creber N. J., Eastwood H. T., Hampson A. J., Tan J., O’Leary S. J. A comparison of cochlear distribution and glucocorticoid receptor activation in local and systemic dexamethasone drug delivery regimes. Hearing Research . 2018;368:75–85. doi: 10.1016/j.heares.2018.03.018. [DOI] [PubMed] [Google Scholar]
- 38.Tan C.-Q., Dong W.-D., Guo L., Huang H., Wang D.-Y. Auditory function in women with autoimmune inner ear diseases and their offspring. International Journal of Pediatric Otorhinolaryngology . 2009;73(12):1702–1711. doi: 10.1016/j.ijporl.2009.09.003. [DOI] [PubMed] [Google Scholar]
- 39.Currie C., Wax J. R., Pinette M. G., Blackstone J., Cartin A. Cogan’s syndrome complicating pregnancy. Journal of Maternal-Fetal and Neonatal Medicine . 2009;22(10):928–930. doi: 10.1080/14767050902974236. [DOI] [PubMed] [Google Scholar]
- 40.Tarney C. M., Wilson K., Sewell M. F. Cogan syndrome in pregnancy. Obstetrics & Gynecology . 2014;124(2 Pt 2 Suppl 1):428–431. doi: 10.1097/AOG.0000000000000390. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The CARE checklist for presentation of a case report is provided.
Data Availability Statement
No data were used to support this study.


