Abstract
Biotherapy has recently become a hotspot research topic with encouraging prospects in various fields due to a wide range of treatments applications, as demonstrated in preclinical and clinical studies. However, the broad applications of biotherapy have been limited by critical challenges, including the lack of safe and efficient delivery systems and serious side effects. Due to the unique potentials of biomaterials, such as good biocompatibility and bioactive properties, biomaterial-assisted biotherapy has been demonstrated to be an attractive strategy. The biomaterial-based delivery systems possess sufficient packaging capacity and versatile functions, enabling a sustained and localized release of drugs at the target sites. Furthermore, the biomaterials can provide a niche with specific extracellular conditions for the proliferation, differentiation, attachment, and migration of stem cells, leading to tissue regeneration. In this review, the state-of-the-art studies on the applications of biomaterials in biotherapy, including drug delivery, vaccine development, gene therapy, and stem cell therapy, have been summarized. The challenges and an outlook of biomaterial-assisted biotherapies have also been discussed.
Keywords: Biomaterial, Biotherapy, Drug delivery system, Vaccine development, Gene therapy, Stem cell therapy
Graphical abstract
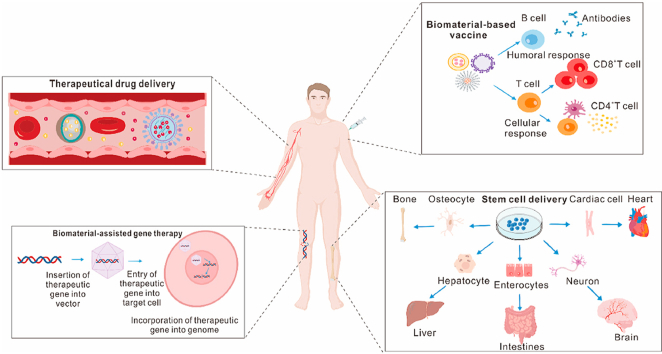
An overview of the latest pre-clinical and clinical studies on the applications of biomaterials in biotherapy, including drug delivery, vaccine development, gene therapy, and stem cell therapy.
Highlights
-
•
Biomaterials possess unique advantages to improve the efficacy and safety of biotherapy.
-
•
Various types of biomaterials can be used in a wide range of biotherapy.
-
•
The functions of biomaterials can be tuned by changing their inherent properties or the surrounding environment.
1. Introduction
Biomaterials are generally classified into three categories; organic, inorganic, and bio-based materials. Organic materials, such as the Food and Drug Administration (FDA)-approved polymers used in therapeutics, polyethylene glycol (PEG), and poly lactic-co-glycolic acid (PLGA), have been widely explored and applied in delivery systems, tissue engineering, etc. [1,2]. Inorganic materials have gotten increasing attention due to their controllable synthesis and rigid structures. The bio-based materials are mainly originated from cells, bacteria, and viruses, such as protein-based nanosystems, exosomes, outer membrane vesicles (OMVs), and virus-like particles (VLPs), etc. [[3], [4], [5], [6], [7]]. Furthermore, based on their origins, biomaterials can be divided into natural and synthetic materials. Natural biomaterials have been used for a long time due to their superior biocompatibility, biodegradability, low toxicity, and low allergenicity, and yield into the degradation products, which are less cytotoxic and more easily metabolized by the host tissues [8]. However, their extensive purification significantly limits the application of natural biomaterials. In contrast, synthetic materials are appealing due to their more tunable mechanical properties and easy manufacture on a large scale [9].
Biotherapy has become a hotspot research topic with encouraging prospects in various fields, such as cancer immunotherapy, gene therapy, stem cell therapy, and tissue engineering. In the field of cancer immunotherapy, cancer vaccines have been explored since the discovery of tumor-specific antigens by Georg Klein in the 1960s. However, its clinical translation is limited due to its weak immunogenicity, side effects, and systemic toxicity [10]. Gene therapies and stem cell therapies have revolutionized therapeutic modalities, offering an enormous potential to treat a wide range of diseases [11,12]. Nevertheless, a safe and efficient delivery system is still a critical challenge [13]. Moreover, the vital issues of poor cell engraftment, survival, and differentiation also limit the applications of stem cell therapy. Notably, the biomaterials can not only allow targeted delivery of multiple agents, such as drugs, proteins, and nucleic acids, but also ensure a sustained release at the target sites, thereby enhancing the therapeutic efficacy and reducing the systemic side effects [14]. Meanwhile, biomaterials can provide a biomimetic niche for stem cells to survive and proliferate [15]. With an increase in the aging population, the demand for implants, such as orthopedic devices, vascular scaffolds, and dental prostheses, is rapidly increasing worldwide [16,17]. The global market value for biomaterials was estimated to be USD 121.1 billion in 2020 and is projected to reach a global market value of USD 348.4 billion in 2027 [18].
In order to provide an overview of the current advances in biomaterial-assisted biotherapies, the latest pre-clinical and clinical studies, which were conducted on the applications of biomaterials in biotherapies, including drug delivery, vaccine delivery, gene therapy, and stem cell therapy, are documented in this study. For drug delivery, four systemic drug delivery strategies were focused on, which promote effective drug accumulation on target sites and minimize the off-target and undesirable side effects related to their long-term application. For vaccine development, different biomaterial categories, which are used for vaccination as carriers or adjuvants, were overviewed. For gene therapy, the contributions of biomaterials in delivering different nucleic acids, including encoding DNA, miRNA, chimeric antigen receptor (CAR) gene, and the CRISPR (clustered regularly interspaced short palindromic repeat)-Cas9 (CRISPR-associated) system, are highlighted. For stem cell therapy, the applications of biomaterials in creating 3D culture systems for guiding stem cell behaviors are discussed. The implantable scaffolds and injectable biomaterials for the delivery of biological factors, therapeutic agents, and immune cells are also described. Finally, the challenges, which are required to be addressed for the clinical applications of biomaterials, such as the selection of animal models to assess these biomaterial platforms, the need for a simple and standardized fabrication process, etc., are also highlighted.
2. Biomaterials in targeted drug delivery
Recent advances in biotherapies, including immunotherapy and molecular targeted therapy, have significantly improved the prognosis and life quality of cancer patients [19]. However, there remain plenty of challenges, concerning both the efficacy and safety, upon the systemic administration of these therapeutics alone due to poor tumor selectivity. Although the clinical use of biotherapy has been validated robustly, only a subset of patients responds to biotherapy. The administration of therapeutics in pure form might result in fast biodegradation in the body, undesirable biodistribution to non-target tissues and organs, and might not pass the permeabilization barriers in solid tumors, which significantly reduce their effective concentrations in targeted diseased tissues. Furthermore, some drugs do not remain contained within the lesions, thereby resulting in unavoidable systemic toxicity [19]. Hence, studies are continuously being conducted to develop more targeted and controlled systems for systemic drug delivery. The applications of biomaterials allow researchers and physicians to modulate drug pharmacokinetics after systemic drug administration. Numerous drugs, such as chemotherapeutics, immune checkpoint inhibitors, small molecule inhibitors, and cytokines, have been explored to be encapsulated in or conjugated to nanoparticles, which deliver and ensure the on-target release of drugs [[19], [20], [21]]. Currently, multiple materials, including lipids, polymers, metals, non-metallic inorganics, such as silica and graphene, and viruses, are being utilized to generate various drugs delivery systems (DDSs)with high loading capacity, excellent biocompatibility, biostability, biodegradability, and unique bioactivities [19,[22], [23], [24]]. In order to enhance the efficacy and safety, the targeted DDSs are under investigation to promote effective on-target drugs accumulation while minimizing the off-target and undesirable side effects related to the long-term drug's application (Fig. 1).
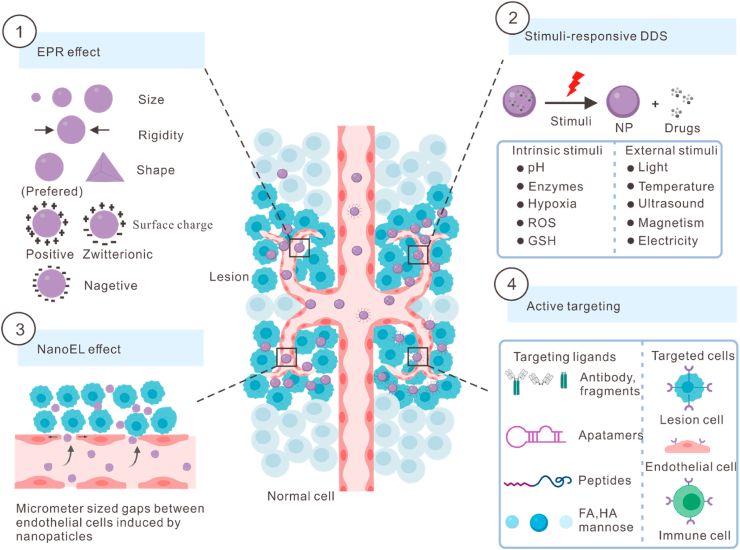
Fig. 1.
Four targeting strategies for systemic drug delivery. EPR, enhanced permeability and retention; NanoEL, nanomaterial-induced endothelial leakiness; DDS, drug delivery system; NP, nanoparticle; ROS, reactive oxygen species; GSH, glutathione; FA, folic acid; HA, hyaluronic acid; and R, receptor.
2.1. Passive targeting
The passive drug targeting mechanism is also known as the enhanced permeability and retention (EPR) effect [25]. The speed and rate of the drug's delivery to the on-target sites are influenced by the physicochemical characteristics of the carriers, such as size, shape, surface charge, rigidity, and contact angle [26]. The uptake efficiency of nanoparticles in macrophages has been demonstrated to be negatively correlated with the size of nanoparticles having <100 nm size and positively correlated with those having >100 nm size. The small-sized nanoparticles can enhance drug penetration into the hypovascular tumor core, thereby increasing the therapeutic potential of parent drugs against various multidrug-resistant tumors and decreasing their systemic toxicities. Aniruddha et al. demonstrated that the 20-nm nanoparticles, formed by the covalent conjugation of podophyllotoxin and PEG with acetylated carboxymethyl cellulose, showed a 5–20 fold increase in tumor-target-specific delivery compared to the larger nanoparticles (30 and 120 nm), which exhibited high uptake in the normal liver cells [27]. As for their surface charges, the highly positive or negative nanoparticles are more prone to be taken up by macrophages as compared to the neutral ones [28]. For instance, the positively charged poly β-amino esters (PBAEs) are used as a targeted drug-delivery system for cartilage by exploiting the electrostatic interactions between cationic nanocarriers and the negatively charged constituents of cartilage extracellular matrix (ECM) [29]. Generally, the rigid and spherical carriers tended to be internalized more easily than the soft and cylindrical ones [26]. However, these approaches are still far from ideal due to the off-target accumulation of a portion of drugs, such as those in the liver and spleen. Furthermore, the involvement of various material compositions or other uptake mechanisms might result in different outcomes among the incorporation patterns. Moreover, the promising EPR effects in animal cancer models failed to replicate in human cancer patients [30], which substantially limited their clinical translation.
2.2. Nanomaterial-induced endothelial leakiness
Nanomaterial-induced endothelial leakiness (NanoEL) was first coined by Setyawati and co-workers in 2013 and refers to the micrometer-sized gaps between the endothelial cells induced by certain nanoparticles with specific characteristics [31]. Unlike the EPR effect, which is a biological phenomenon characterized by an aberrant vascular network, the NanoEL is a controllable and tunable bioengineering effect, which is mainly caused by inorganic nanoparticles, such as TiO2-, gold- and SiO2-based nanoparticles [32]. This phenomenon is primarily attributed to the nanoparticle-induced cytoskeletal remodeling and disruption of intercellular junctional protein VE-cadherin [31,32]. The occurrence of NanoEL depends on the size of nanoparticles and the origin of endothelial cells. The gold nanoparticles (GNPs) with an average diameter of 10–30 nm are the potent inducers of NanoEL [33]. The human mammary and skin endothelial cells are more sensitive to the NanoEL as compared to the umbilical vein endothelial cells [33]. As the intravenously administered drugs need to pass through the endothelial barrier in order to penetrate the blood vessels at tumor sites, the NanoEL allows for the controlled release of nanomedicines into tumors or other vascular-related disease-target sites [32]. Nevertheless, the NanoEL could also cause leakiness at the unintended sites, leading to undesirable outcomes, such as metastasis, edema, inflammation, etc. [32]. Peng et al. found that the TiO2, silica, and gold nanoparticles-induced NanoEL could significantly promote cancer metastasis by accelerating the intravasation and extravasation of breast cancer cells in animal models [34]. Therefore, further studies are required to develop safer biomaterials to exploit the potential of NanoEL in drug delivery, especially for the treatment of cancer.
2.3. Stimuli-responsive DDS
Stimuli-responsive carriers can release payloads when triggered by intrinsic stimuli, such as pH, hypoxia, enzymes, glutathione (GSH), and reactive oxygen species (ROS), or extrinsic stimuli, such as light, temperature, ultrasound, magnetism, and electricity [30,35]. These strategies optimized the drug delivery by controlling the drug release accurately and flexibly at the target site, avoiding premature drug release in the healthy organs and tissues. The unique characteristics of diseased tissues and their microenvironments offer specific natural stimuli, which promote the drug unloading by triggering a conformational and solubility change in DDS or by cleaving the conjugation between drug and delivery platform [30]. For instance, the characteristics of the tumor microenvironment, such as acidic pH, overexpressed enzymes (matrix metalloproteinases, cathepsin B, and phospholipase A2), hypoxic state, and redox dysregulation (overproduction of GSH and ROS), can trigger the drug release in tumor tissues [30,36]. The pH-responsive nanocarriers are stable at physiological pH (7.35–7.45) but become unstable or fusogenic in response to the acidic environment in the extracellular compartments of tumors or endosomes (5.85–7.68), leading to the on-target release of drugs within tumors and cytosol [36]. The applications of external stimuli-based DDS depend on specialized equipment, which is easy to control. For instance, smart thermosensitive DDSs are stable at normal body temperature (37 °C) but release the loaded drugs in response to an increase in temperature (40–42 °C) [25]. The systemic administration of doxorubicin-loaded nanohybrid cerasomes followed by double high-intensity focused ultrasound (HIFU, a device to produce the required local high temperature) into mice showed a higher concentration of drugs delivered to the tumor target site, achieving the maximum therapeutic efficacy against adenocarcinoma (MDA-MB-231 cell line) with minimum side effects, as compared to the animals, receiving the same dose of nanomedicine without HIFU treatment [25].
However, there are some limitations in these stimuli-sensitive DDSs. For instance, although the infrared radiation (IR) light is one of the most commonly used external stimuli, its broad applications are significantly hindered by its limited tissue penetration and high phototoxicity [37]. Moreover, considering the remarkable heterogeneity within or between different target sites, the drug release is often mediated by a single-responsive delivery system, which is always insufficient and slow. Therefore, researchers are working on the development of multi-responsive materials to overcome these obstacles. For the delivery of doxorubicin, Zhong et al. designed a novel triple-responsive dendrimeric nanocage, which was responsive to temperature, pH, and oxidation-reduction states [38]. The nanocage was fabricated by the co-assembling and crosslinking of lipoic acid-modified dendrimers with PEG derivates. It exhibited an improved loading capacity (about 2-fold higher) at lower temperatures and passively accumulated at the tumor sites. Upon stimulation with temperature, lower pH, and reducing agent (high expression levels of GSH), the nanocage could release doxorubicin locally and deliver it through lysosomal disruption-mediated intracellular drug translocation, thereby achieving accurate targeted drug delivery. Therefore, the nanocarriers can be designed to release the drugs in response to the intrinsic stimuli, which are only or mainly located at the target diseased site. Moreover, external stimuli can also be provided using specialized equipment to guide the release of drugs at the targeted site.
2.4. Active targeting with specific antibodies or other ligands
Targeting ligands are used to enable the active delivery of drugs to tumor cells, immune cells, and tumor tissues via the specific ligand-receptor interaction. These ligands include specific antibodies, aptamers, peptides, and other ligands [30,36]. Monoclonal antibodies against various molecules, including human epidermal growth factor receptor 2 (HER2), epidermal growth factor receptor (EGFR), transferrin receptor (TfR), CD20, prostate-specific membrane antigen (PSMA), and mammaglobin-A (Mam-A), are the frequently used ligands for targeting the different types of tumors [39,40]. Due to their smaller size and easy conjugation, the antibody fragments, antigen-binding fragments (Fab), and single-chain variable fragments (scFV) are investigated to mediate the active targeting-based delivery of drugs [39]. Aptamers are versatile nucleic acid-based macromolecules with a high affinity for a specific target. The nanocarriers, including liposomes, polymeric micelles, and biomimetic nanocarriers, are functionalized by aptamers for the targeted anticancer drugs delivery [41]. Peptides are also molecularly recognized due to their unique amino acid sequences, cell-penetrating ability, and changeable conformations. The cancer cell-targeting peptides can be obtained through a structure-based design using molecular docking strategies [42]. Peptides, which target the transferrin receptors on the blood-brain barrier (T7 peptide) and diseased neurons (Tet1 peptide), are used to modify the polymeric nanoparticles for an increased drug accumulation at the target site and improved efficacy for Alzheimer's disease therapy [43]. Other ligands for the fabrication of targeted DDS, such as folic acid (anti-folate receptor) [26], hyaluronic acid (HA, anti-CD44) [44], and mannose (anti-CD206) [26], are vital candidates for elevating the intratumoral accumulation of drugs and increasing the cellular internalization.
Biomarkers are only expressed or highly expressed in the tumor cells. Tumor tissues are the most commonly investigated receptors for the ligand-targeting DDS. Theoretically, any abnormally expressed molecule in different tumors can be targeted. For example, Zhang et al. presented a conformational epitope imprinting strategy for the p32 receptor using a sub-40 nm-sized polymeric nanoparticle to selectively deliver the therapeutic drug to p32-positive breast cancer in-vivo [45]. A PSMA-targeting docetaxel nanoparticle could significantly improve tumor response in mice harboring PSMA-expressing human prostate cancer xenografts as compared to the passively targeted nanoparticles. On the contrary, among the breast cancer (MX-1) and non-small-cell lung cancer (NSCLC, NCI–H460) tumor models, which did not express PSMA, the efficacy of PSMA-targeting nanoparticles was similar to that of the non-PSMA-targeting nanoparticles, which further verified the remarkable effectiveness of actively targeting DDSs [46]. The nanoparticles, which carry therapeutic drugs, might diffuse poorly into the tumor parenchyma due to the vascular barrier. In order to deal with this, the molecules expressed on endothelial cells can be considered as target receptors. Yosi et al. developed a P-selectin-targeting nanotherapeutic delivery platform, which was composed of fucoidan (fucosylated polysaccharide) having a nanomolar affinity for P-selectin [21]. After encapsulation into these nanoparticles, the chemotherapeutics (paclitaxel and doxorubicin) and targeted therapeutics (MEK162) could be selectively delivered to the P-selectin-positive endothelial cells and multiple tumor cells. As compared to the passively delivered nanoparticles, this advanced DDS could mediate tumor-specific infiltration, prolong local drug release, improve antitumor efficacy, and reduce systemic toxicities [21]. For the tumors with low or without P-selectin expression, ionizing radiation-induced P-selectin expression could successfully redistribute the drugs and their susceptibility [21].
Another approach for the enhancement of diffusion rate is to specifically deliver the therapeutic drugs to a defined subset of endogenous immune cells, which can penetrate deeply into the tumor core. Daniela et al. developed a PLGA/PEG-based antibody-modified nanoparticle by conjugating the anti-CD8a F(ab’)2 fragments to the particle surface. This nanoparticle could specifically and efficiently deliver the immunomodulatory small molecules to programmed death-1 (PD-1)+ CD8+ T cells at the blood circulation, lymphoid tissues, and tumor microenvironment sites [47]. It could also successfully deliver transforming growth factor, β receptor I (TGFβRI) inhibitor (SD208) and toll-like receptor (TLR) 7/8 agonist (R848) to the targeted T cells, reduce the required dose and prolong the survival of tumor mouse models. Moreover, this nanoparticle also successfully enhanced the proportion of tumor-infiltrating CD8+ T cells and sensitized the MC38 and B16 tumor models, ensuring the anti-PD-1 therapy. In contrast, neither the free agents nor the non-targeting nanoparticles showed any of these efficacies [47]. Dendritic cells (DC), the most powerful antigen-presenting cells (APC), process and present antigens to lymphocytes for the activation of adaptive immunity [48]. Several ligands, which bind to the receptors, are highly expressed on the surface of DCs and can be used to modify the nanoparticles to specifically deliver nanomedicine to the DCs, such as DEC205 and 33D1 receptors [[49], [50], [51]]. Macrophages, another essential component of the innate immune system, can also be a high target cell for modified drug delivery [52]. Hence, fabricating the nanoparticles with molecules, which target various subtypes of immune cells, might allow the effective regulation of immune functions and prevent diseases. Theoretically, the active targeting strategies should possess the lowest off-target effects due to the use of specific ligand-receptor bindings.
3. Biomaterials in vaccine development
Vaccination has a major impact on disease prevention, including infectious diseases and cancer. Traditionally, vaccines are developed based on killed or live attenuated microbes or microbial components. Vaccines have greatly reduced the health burden of former epidemics, such as polio [53] and smallpox [54]. They prevent 2–3 million deaths every year from measles, influenza, tetanus, and pertussis, as estimated by the World Health Organization (WHO). However, the suboptimal immunogenicity and safety of conventional vaccination remain challenging. In addition, effective vaccines for immunization of several human pathogens, including human immunodeficiency virus (HIV) and malarial parasite Plasmodium falciparum, have not been developed yet [55]. With an increasing demand for high-quality vaccines, innovative approaches are urgently needed to develop vaccines. In recent years, a new class of biomimetic materials is widely explored as vaccine carriers because it can deliver the targeted and sustained release of multiple immune drugs and protects the drugs from enzymatic degradation and extreme pH conditions [56]. Here, different biomaterials, which are used for vaccine delivery, have been discussed. A more comprehensive introduction on the use of biomaterial in vaccine delivery can be read in other texts [56,57].
3.1. Polymeric particles
Numerous polymeric particles have been developed as carriers for vaccines due to their excellent biocompatibility and biodegradability, such as PLGA, PEG, polycaprolactone, dextran, chitosan, and self-assembled peptides [58]. These nanoparticles can not only entrap antigens for enhanced delivery but also allow a sustained antigen release with a slow biodegradation rate. In a recent study, the FDA-approved PLGA was selected for the delivery of thiol (–SH)–modified recombinant malarial antigen with an adjuvant to induce the high titers of antibody responses and increase antibody affinity [59]. The inhalable poly (lactic acid) (PLA) and PLGA nanoparticles, containing hepatitis B surface antigen (HBsAg), are reported to produce robust mucosal, humoral, and cytokine responses [60]. Other polymeric poly D, L-lactic-co-hydroxymethyl glycolic acid nanoparticles have been utilized for co-delivering the synthetic long peptides of human papillomavirus (HPV) and a TLR3 ligand (poly IC) in order to induce an effective immune response for the prolonged survival of mice [61]. Chitosan, which is a non-immunogenic deacetylated derivative of chitin, has been extensively used for the delivery of multiple vaccines, including DNA vaccines [62] and hepatitis B virus (HBV) vaccines [63]. The chitosan nanoparticles, loaded with Esat-6 three T cell epitopes (Esat-6/3e) and FMS-like tyrosine kinase 3 ligands (FL), could induce an enhanced T cell response and protect mice from Mycobacterium tuberculosis infection [64]. Peptide molecules can self-assemble into specific nanostructures with outstanding potential for vaccine delivery, as reviewed by Eskandari et al. [65]. On the other hand, the polymeric particles can also act as adjuvants for vaccines. For example, Advax, which is an adjuvant derived from insulin, could significantly enhance the immune response triggered by the HBV and influenza vaccines [66,67]. Recently, the PBAEs, which were synthesized via the Michael addition of amines to acrylates, could attract enormous attention as a delivery system due to their biodegradability and pH responsiveness [68]. PBAEs can be fabricated into versatile formulations alone or in combination with other polymers to fulfill different requirements for targeted delivery [69].
3.2. Liposomes
Liposomes are bi-layered vesicles, which are composed of plain lipids or the mixtures of lipids and cholesterol; they were first described by Alec Bangham in the mid-60s [70,71]. Liposomes have long been popular in the field of vaccinology either for their use as delivery vehicles or adjuvants due to their plasticity and versatility. Liposomes can be designed with desired properties by altering their lipid components, surface structures, particle sizes and charges, production processes, etc., as reviewed by Li et al. [72]. The cationic liposomes are more frequently used as vaccine carriers due to their more efficient uptake by macrophages and DCs as compared to the anionic liposomes [73]. They are also capable of bypassing the endosomal-lysosomal degradation in cells [73]. A liposomal vaccine, consisting of MUC1 peptide, monophosphoryl lipid A adjuvant, and the lipids of dimyristoyl phosphatidylglycerol (DMPG) and dipalmitoylphosphatidylcholine (DPPC), could increase the survival of patients with advanced unresectable NSCLC in phase III clinical trial [74]. Similarly, a lipid mRNA vaccine, which was designed to precisely and effectively target DCs, could induce the production of abundant interferon-α (IFN-α) and strong effector and memory T cell responses in-vivo [75]. These lipid nanoparticles (LNPs) protect mRNA from fast degradation, enable the translation of mRNA in DCs, and present the tumor antigens to T cells. Furthermore, using the mRNA, encoding a fluorescent protein, researchers found that the overall biodistribution of nanoparticles was more dependent on the particle charges rather than on the lipid species [75]. The application of liposomes in DNA vaccines could improve immunogenicity. A mannosylated zwitterionic-based cationic liposome (man-ZCL) was used for the construction of HIV DNA vaccines, which promoted the antigen presentation and enhanced the anti-HIV immune responses [76]. Moreover, this lipoplex showed lower toxicity than the cytosine-phosphodiester-guanine (CpG)/DNA and Lipo2000/DNA complexes [76].
3.3. Inorganic particles
Despite their low biodegradability, the inorganic nanoparticles are also considered as essential antigen delivery vehicles and vaccine adjuvants, such as silica nanoparticles (SNPs), carbon nanoparticles (CNPs), GNPs, iron oxide nanoparticles (IONPs), zinc oxide nanoparticles (ZNPs), aluminum and calcium phosphate, etc. Two major advantages of the inorganic particles are their controllable synthesis and rigid structures. The SNPs are known to be effective vaccine carriers due to their favorable biocompatibility, variable porosity, and high drug-carrying capacity. The mesoporous SNPs can deliver antigens in a controllable and sustainable manner [77]. The small-sized SNPs (30 nm) showed high efficiency in the targeted delivery of antigens and adjuvants to the draining lymph nodes and accumulated in APCs, which dramatically enhanced the antigen-specific B and T cell responses [78]. As powerful vaccine adjuvants, the SNPs could induce both the T-helper 1 (Th1)- and Th2-mediated immune responses [79]. Mooney's group developed a novel ciVAX vaccine, which is composed of mesoporous silica nanorods, absorbing the granulocyte-macrophage colony-stimulating factor (GM-CSF) and CpG-rich oligonucleotides, and superparamagnetic microbeads, capturing the pathogen-associated molecular patterns (PAMPs) [80]. This vaccine showed promising prospects for protection against Gram-negative and Gram-positive bacterial infections and septic shocks.
CNPs with large mesopores and macropores are excellent antigen carriers. When administered as oral vaccine adjuvants, they could induce strong IgG, IgA, and Th1- or Th2-mediated immune responses [81]. Furthermore, CNPs can also be synthesized into nanotubes, including single- and multiple-walled nanotubes. For instance, the single-walled carbon nanotubes could successfully deliver weak peptide antigens to DCs and induce significant IgG responses [82]. Nevertheless, the potential toxicity caused by carbon nanotubes in-vivo due to their irregular shapes and strong pro-inflammatory effects remains unknown.
GNPs can be easily synthesized into different core sizes and shapes, equipping them with unique tunable and biocompatible properties. The efficacy of GNPs has been demonstrated in influenza and foot-and-mouth disease as vaccine carriers [83]. For example, the GNPs were used as carriers for an antigen of influenza A virus in conjugation with CpG, which induced protective immunity [84]. The conjugation of gold nanorods with protein antigens derived from the respiratory syncytial virus could induce human T cell proliferation. GNPs can also be used as immunological vaccine adjuvants. As compared to the naked DNA vaccines, those conjugated with gold nanorods for HIV could efficiently promote the induction of both cellular and humoral immunity [85]. It is noteworthy that the GNPs could cause cytotoxicity at large doses (>8 mg/kg) and activate inflammatory responses [86].
IONPs are commonly used as drug carriers and contrast agents for magnetic resonance imaging (MRI). A variety of IONPs has already been translated into clinical settings. Ferumoxytol is an FDA-approved MRI contrast agent and iron supplement used for the treatment of iron-deficient anemia. Interestingly, ferumoxytol could significantly inhibit tumor growth by inducing the M1 polarization of macrophages in-vivo [87]. Furthermore, IONPs could also efficiently deliver antigens in the presence or absence of lipo-PEG. ZNPs have been widely developed in the cosmetic and food industries. Antigens can bind to the ZNPs through zinc-binding peptides [88]. Aluminum-based nanoparticles are the commonly used adjuvants, which can enhance antigen-specific cellular immunity [89]. Besides, the bioresorbable and non-toxic properties of calcium phosphate nanoparticles make them appealing vaccine carriers and adjuvants [90].
3.4. Virus-like particles (VLPs)
VLPs are self-assembled multiprotein nanoparticles, which are formed by biocompatible capsid proteins without a viral genome. VLPs are excellent vaccine candidates due to their favorable immunological characteristics and can activate both the innate and adaptive immune responses, avoiding viral replication. The first licensed malarial VLP-based vaccine (Mosquirix™) has been approved by the European Medicines Agency (EMA) [91]. EMA recommended Mosquirix™ to prevent malaria in children aged 6 weeks to 17 months. However, it is worth noting that Mosquirix™ requires a three-dose schedule followed by a booster after 18 months. The results of the phase III clinical trial indicated that the vaccine had no superiority over the existing malaria control measures without a booster dose, which might hinder its logistical implementation in the low-resource settings [92]. Recently, a handful of VLP-based vaccines have been commercialized, including the vaccines against HPV (Cervarix®, Gardasil® & Gardasil9®) and HBV (Engerix®). Several other VLP-based vaccines are currently undergoing preclinical evaluations and clinical trials (Table 1), covering a wide range of diseases, such as influenza, HIV, Chikungunya, cancer, hypertension, Alzheimer's disease, and coronavirus disease 2019 (COVID-19). For instance, Medicago's plant-derived VLPs vaccines for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) conjugated with AS03 or CpG1018 have been carried forward into an ongoing phase III clinical trial (NCT04636697) [93].
Table 1.
Clinical trials of VLP-based vaccine.
| Vaccine | Conditions | Phase | Sample size | Current status | ClinicalTrial.gov identifier |
|---|---|---|---|---|---|
| COVID-19 (adjuvant: AS03) | SARS-CoV-2 infection | II/Ⅲ | 30918 | Recruiting | NCT04662697 |
| VBI-2902a (adjuvant: Alum) | COVID-19 | Ⅰ/II | 780 | Active, not recruiting | NCT04773665 |
| NoV GI.1/GII.4 Bivalent VLP Vaccine | Norovirus | II | 840 | Completed | NCT02153112 |
| CHIKV VLP Vaccine | Chikungunya virus infection | II | 445 | Completed | NCT03483961 |
| 9-Valent HPV Vaccine | Cervical cancer/Vulvar cancer/Vaginal cancer/Genital warts/HPV infection | Ⅲ | 14840 | Completed | NCT00543543 |
| V501 | Cervical cancer/Genital warts | Ⅲ | 12167 | Active, not recruiting | NCT00092534 |
| Novartis Meningococcal ACWY Conjugate Vaccine | Meningococcal meningitis/HPV infection/Pertussis/Tetanus | Ⅲ | 1620 | Completed | NCT00518180 |
| Quadrivalent VLP Influenza Vaccine | Virus diseases/RNA virus infections/Respiratory tract diseases/Respiratory tract infections | Ⅲ | 10137 | Completed | NCT03301051 |
| A/H1N1 2009 Influenza VLP Vaccine | Seasonal influenza | II | 4560 | Completed | NCT01072799 |
| RSV-F Protein Nanoparticle Vaccine | RSV-F | II | 720 | Completed | NCT01960686 |
| Formalin-inactivated EV71 Vaccine | HFMD | Ⅲ | 12000 | Completed | NCT01569581 |
| CYT006-AngQb | Mild essential hypertension/Moderate essential hypertension | II | 83 | Completed | NCT00710372 |
| HIV p17/p24:Ty-VLP | HIV infections | Ⅰ | 36 | Completed | NCT00001053 |
| VRC-ZKADNA090-00-VP | Zika virus infection/Virus diseases/Flavivirus infections/Flaviviral diseases/Flaviviridae infections/RNA virus infections | II | 2338 | Completed | NCT03110770 |
COVID-19, coronavirus infectious disease 2019; SARS-CoV-2, severe acute respiratory syndrome-coronavirus 2; Nov, norovirus; VLP, virus-like particle; CHIKV, Chikungunya virus; HPV, human papillomavirus; HIV, human immunodeficiency virus; RSV-F, respiratory syncytial virus infections; EV71, enterovirus 71; and HFMD, hand, foot, and mouth disease.
3.5. Outer-membrane vesicles (OMVs)
OMVs are naturally derived from Gram-negative bacteria with sizes ranging from 20 to 250 nm. OMVs have a bi-layered lipid membrane nanostructure, containing lipopolysaccharide (LPS), phospholipids (PL), and membrane proteins, and a lumen, containing DNA, RNA, toxins, enzymes, periplasmic proteins, and peptidoglycan (PG) [94]. Forty years ago, OMVs were observed as membrane sacs produced by Vibrio cholerae and Neisseria meningitidis during their in-vitro growth [95,96]. OMVs are not only very stable when exposed to different treatments and temperatures [97] but are also highly versatile as they can be engineered to express different antigens [98]. In addition, as the OMVs contain immunogenic components from their parent bacterium, they can activate humoral and cell-mediated immune responses to antigens, which make them an appealing vaccine delivery adjuvant [99]. The OMVs-based vaccines, including the Meningitis type B (MenB) OMV vaccine, have been tested in multiple clinical studies, showing promising results. The multi-component MenB vaccine (4CMenB) could provide 66–91% protection against meningococcal serogroup B strain infection worldwide [100]. In addition to homologous antigens, OMVs can also be bioengineered to express heterologous antigens, making them even more applicable as vaccine carriers [101].
3.6. Immunostimulating complexes (ISCOMs)
The spherical ISCOMs can be spontaneously formed by mixing the hydrophobic antigens, saponin, phospholipids, and cholesterol; this was first described by Bror Morein in 1984 [102]. ISCOMs can be designed into stable vaccine carriers and adjuvants. For example, the Antigen 85 complex (Ag85)-loaded ISCOMs could improve humoral and cellular immune responses against Mycobacterium tuberculosis infection after pulmonary immunization [103]. However, a primary challenge for the synthesis of classical ISCOM was the incorporation of antigen proteins, which not only restricted the types of antigens but also was a complicated and not a well-controlled process. In order to address these issues, ISCOMATRIX has been developed [104], which is a pre-formed cage-like particulate, typically 40–50 nm in diameter, containing the purified fractions of Quillaia saponaria extract (ISCOPREP saponin), phospholipid, and cholesterol. ISCOMATRIX can be simply mixed with any antigen due to its Quil [105]. It is a potent immunomodulator with an impact on both the innate and adaptive immune systems. ISCOMATRIX vaccines can induce a wide range of antibodies and antigen-specific T cell responses in cancer immunotherapy and infectious diseases [106,107]. In addition, a series of ISCOMATRIX vaccines have been evaluated in human clinical trials, which are immunogenic, safe, and well-tolerated.
3.7. Nanoparticle-based vaccines for COVID-19
With the outbreak of SARS-CoV-2 in 2020, there was a great urgency to develop COVID-19 vaccines [108,109]. Until December 2nd, 2021, a total of 26 vaccines have been authorized for emergency use against SARS-CoV-2, and 165 vaccines candidates are under clinical trials, including 40 in phase Ⅰ, 56 in phase Ⅱ, and 61 in phase Ⅲ clinical trials [110]. Nanocarriers can enhance the stability of mRNA vaccines, transfection rate of DNA vaccines, and immunogenicity of protein vaccines while mimicking the size of viral particles, which increases their cellular uptake. Several different nanoparticles, including LNPs, self-assembling protein nanoparticles (SAPNs), and VLPs have been developed as adjuvants for COVID-19 vaccines. For example, a SARS-CoV-2 RNA vaccine was encapsulated in superparamagnetic iron oxide (Fe3O4) nanoparticles, which demonstrated robust antibody and cellular immune responses [111]. Until now, four LNP-formulated mRNA vaccines have been developed by BioNTech SE and Pfizer, including BNT162a1, BNT162b1, BNT162b2, and BNT162c2, and have been tested in phase Ⅱ (NCT04380701) and phase Ⅲ (NCT04368728) clinical trials. The BNT162b2 has finished the phase Ⅲ trials and achieved the emergency-use authorization of the FDA, which is the first FDA-approved mRNA vaccine globally [112]. Moderna TX, Inc, has also developed an LNP-based synthetic mRNA vaccine (mRNA-1273) through a modified ethanol-drop nanoprecipitation process [113], which was approved for emergency use by FDA on December 17, 2020 [114]. Currently, the McKay group is evaluating an LNP-encapsulated self-amplifying RNA vaccine against SARS-CoV-2 in a phase Ⅰ clinical trial (ISRCTN17072692) [115]. On the other hand, the SAPN-based coronavirus spike (S) protein vaccine could induce a higher titer of antibodies in mice in the presence of adjuvants [116]. Novavax applied the same strategy of using S protein nanoparticles conjugated with a Matrix M1 adjuvant to induce immunity against SARS-CoV-2 [117]. Currently, a VLP-based vaccine against SARS-CoV-2, developed by Medicago, is under phase Ⅲ clinical trial (NCT04636697) [110]. So far, a total of 20 LNP-based vaccines, 3 SAPN-based vaccines, and 5 VLP-based vaccines are under clinical trials for COVID-19.
In conclusion, the biomaterials offer unique opportunities to induce or alter the desired immune response from conventional vaccines, which have great potential to optimize the vaccination outcomes. However, several questions remain unclear. First, can biomaterials avoid the need for multiple-dose administrations through controlled drugs release? Recently, a pulsatile DDS (PDDS) has attracted great attention, as it allows the rapid release of a certain amount of drug after a lag time [118]. Remarkably, Langer lab at MIT has invented a new 3D fabrication method to generate a library of tiny and encased vaccine particles, which can release multiple doses of a vaccine at a precise and predictable time after being injected once, and has a significant impact on increasing the patients' compliance [119]. Second, the long-term health effects of biomaterials are ill-defined as they persist or degrade to specific byproducts in the body for a long time. Third, since most of the studies are conducted in mouse models, how the biomaterials-based vaccines can be extended to humans readily?
4. Biomaterials in gene therapy
Gene therapy encompasses the successful introduction of genetic materials, such as encoding DNA (including plasmid DNA, pDNA), mRNA, miRNA, small interfering RNA (siRNA), and small hairpin RNA (shRNA). Several gene therapeutics have been approved by FDA or EMA for treating all kinds of diseases, such as infections, cancer, and inherited disorders [120]. Nevertheless, their wide applications have been hindered by the lack of safe and efficient delivery vectors. The gene vectors can be divided into viral and non-viral vectors. The non-viral vectors outperform viral vectors, which might be particularly due to their greater capacity for delivering larger genetic materials and no integration into the host genome after transfection [121]. The non-viral gene vectors are mainly divided into three categories; inorganic materials, lipid or lipid-like materials, and polymeric materials [122]. The lipid (-like) and polymeric compounds are currently under the most intensive investigations for therapeutic gene delivery (Fig. 2).
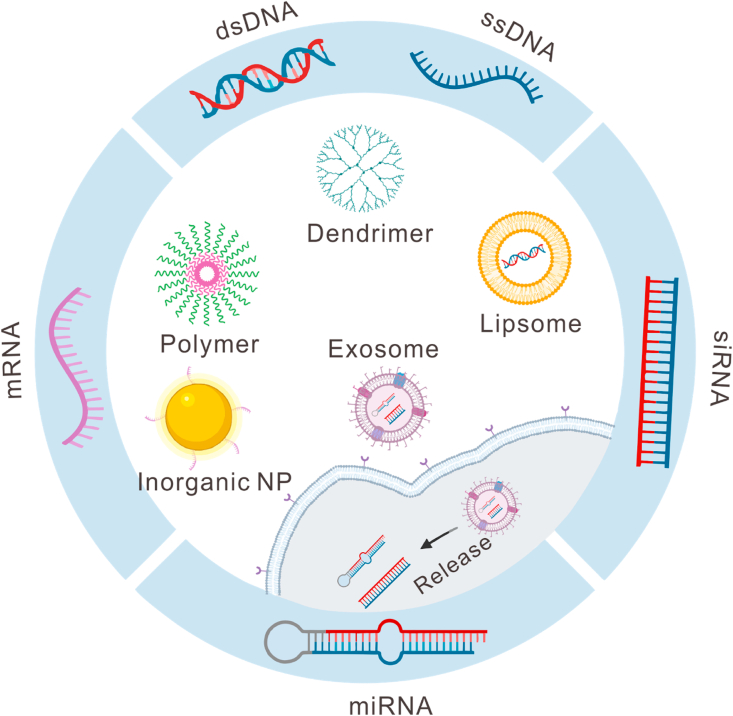
Fig. 2.
Application of biomaterials for the delivery of different nucleic acids. Biomaterials, such as lipid or lipid-like materials, polymeric materials, dendrimers, inorganic materials, and exosomes, are under intensive investigations for the delivery of different genes, including double-stranded DNA (dsDNA), single-stranded DNA (ssDNA), small interfering RNA (siRNA), microRNA (miRNA), messenger RNA (mRNA), etc.
4.1. Delivery of encoding DNA
The delivery of encoding DNA is a “knock-in” strategy, enabling the expression of therapeutic proteins in the target cells. A variety of DNA-based synthetic delivery materials are being developed to achieve gene delivery, which includes inorganic nanoparticles, lipid-based materials, polymers, and peptide-based materials [123]. Some lipid-based and polymeric DNA vectors have already reached the clinical evaluation stage, especially in cancer studies [123]. The cationic and ionizable headgroups of lipid-based nanoparticles can bind to and condense DNA. Then, the efficient delivery and cellular uptake of DNA are mediated via the electrostatic interactions between cationic lipids and negatively charged nucleic acid or plasma membrane components. The transfection efficiency can be improved by increasing the fluidity of the lipid membrane [122]. Rajala et al. created an artificial virus, using liposome-protamine-DNA nanoparticles for the delivery of pDNA, which promoted an efficient and long-lasting gene expression and further enhanced the gene deficiency-related problems in-vivo [124]. The lipid-based transfection reagents, such as lipofectin and lipofectamine, are commercially available for research use only at present and are still restricted for clinical use due to their toxicities. The polymers could enhance cellular delivery by forming a complex with DNA to form the nanosized polyplexes and protect DNA from enzymatic degradation. The amine-based cationic polymers, including polylysine, poly (ethyleneimine) (PEI), PBAEs, polyamidoamine (PAMAM), chitosan, and cationic dendrimers, have been exploited as attractive DNA carriers [125,126]. PEI and its derivatives are the widely used transfection reagents but their non-degradable nature makes them cytotoxic [127]. Currently, the highly branched PBAEs are the most developed gene transduction agents, which have a much higher transfection efficiency and lower toxicity than the commonly used PEI, SuperFect, and Lipofectamine [121,126]. Moreover, the PBAEs-encapsulated DNA maintains its full transfection capability after one year of freeze storage.
The dendritic or branched biomaterials perform better as gene vectors than their linear counterparts due to their 3D structure and multiple terminal groups. Zhou et al. found that the highly branched PBAEs could enhance the transfection efficiency by 8521-fold than their corresponding linear PBAEs [128]. A hybrid PBAEs combined with the linear and branched structures were also reported to show high efficiency in delivering the minicircle DNA into the hard-to-transfect fibroblast cells, showing potential therapeutic application for the treatment of hereditary skin diseases [129]. Most of the nonviral gene vectors developed to date are based on double-stranded DNA (dsDNA), such as pDNA. Researchers have focused on developing more compact-sized nanocarriers based on single-stranded DNA (ssDNA) in order to facilitate tissue penetration. Tockary et al. used the PEG-poly (l-lysine) block copolymer (PEG-PLys) to condense and encapsulate an unpaired ssDNA for constructing a polyplex micelle structure [130]. Through systemic administration, this delivery system could unprecedentedly overcome the thick stromal barriers of pancreatic tumor cell nest, stimulate the transgene expression in tumor cells, and eventually cause a significant antitumor effect. Therefore, the biomaterials can effectively deliver the encoding DNA in in-vitro, in-vivo, and clinical studies. Their structures can be refined to overcome the barriers at the tissue, cell, and intracellular levels for efficient gene expression. Moreover, the stability of biomaterial-DNA complexes improves the duration of gene expression and simplifies their preparation and storage conditions, thereby favoring the bench-to-bed translation. Notwithstanding, most of the popular gene transfection vectors, such as lipofectamine and PEI, are highly toxic, partly explaining their limited translation into clinical use. Huge efforts are still required to improve the safety of non-viral gene vectors without reducing their effectiveness.
4.2. Delivery of miRNA
The RNA-based gene therapy can intentionally modulate the gene expression patterns with exogenous RNAs. Naked RNAs are unstable because due to their rapid degradation by nucleases followed by renal excretion, thereby necessitating the delivery vehicles to enhance their stability and efficacy in RNA-based gene therapy. The non-viral gene vectors, optimized for DNA, cannot be simply adopted for RNA delivery due to their different sizes, structures, and intracellular sites of action (cytosol for RNA vs. nucleus for DNA). The ideal delivery vehicles of mRNA must be able to protect it from enzymatic degradation, evade immune recognition, reach the target tissue, and promote entry to the cells. The successful delivery of mRNA both intranasally and systemically has been reported in-vivo using biomaterials, such as hyperbranched PBAEs or the PBAEs coated with positively charged lipid-layer [[131], [132], [133]]. These polymeric carriers could enable the nanoformulation of stable and concentrated mRNA polyplexes for the noninvasive aerosol inhalation, which further achieved a uniform and localized distribution within all the lungs tissues in mice. For the delivery of siRNA, biomaterials must not only carry it to the cytoplasmic sites but also assemble it into the RNAi mechanisms [123]. The approval of the first siRNA therapeutics, patisiran (ONPATTRO™), by FDA in 2018 significantly inspired the development of biomaterial-based gene therapy. LNPs have been used to encapsulate patisiran for the targeted delivery to hepatocytes for the treatment of peripheral nerve disease (polyneuropathy) [134]. Other comprehensive reviews have summarized the delivery systems for mRNA and siRNA therapeutics [135,136]. There, this study focused on the design of miRNA delivery systems.
miRNAs are short endogenous non-coding RNAs, which negatively regulate the gene expression by partially complementing their target mRNAs [137]. The mature miRNAs are produced from the cleavage of pre-miRNAs in the cytosol [137]. The artificial polycations have drawn great attention for miRNA transfection. For instance, Li et al. demonstrated the poly (glycidyl methacrylate)-based star-like polycations as highly efficient miRNA delivery nanovectors both in the cultured primary cardiac fibroblasts and heart tissues [138]. The abundant flanking secondary amine and hydroxyl groups of these vectors can significantly reduce their toxicity. Naïve macrophages treated with chitosan-encapsulated miRNAs could regulate the expression of ATP-binding cassette transporters A1 (ABCA1) gene, which further controlled the cholesterol efflux and lesion progression of atherosclerosis [139]. Another nanoscale coordination polymer, containing platinum, has been designed to prolong survival in blood circulation and aid in endosomal escape for the miRNA-655–3p transfection into colorectal cells [140]. This approach effectively inhibits the proliferation and invasion of tumor cells, epithelial-to-mesenchymal transition, and liver metastases of colorectal cancer. In order to promote clinical utility, the PLGA nanoparticles could deliver anticancer miRNAs to the deep tissues of liver and kidney lesions in a pig model [141]. Collectively, the polymeric nanoparticles can be explored as the highly promising miRNA delivery vectors for in-vivo gene therapy with proper modifications.
In addition to polycations, natural materials with favorable biocompatibilities can also be modified to facilitate miRNA delivery. The miRNA-hyaluronan-sulfate complex with the aid of calcium ion bridges formed slightly anionic nanoparticles, which could selectively deliver miRNA to cardiac macrophages after myocardial infarction [142]. The addition of hyaluronan-sulfate could mediate the cardiac-targeted delivery and successful modulation of inflammatory and reparative state of lesions, resulting in increased angiogenesis and left ventricle remodeling. The peptide-based vectors could increase the miR199a-3p levels over 500-fold in hepatocellular carcinoma (HCC) cells in-vitro and promote their accumulation at the tumor site, resulting in the downregulation of the mTOR gene and over 50% decline in tumor growth [143]. These peptides can self-assemble into nanoparticles, which can be conjugated with targeting ligands for targeted delivery. As the smallest cell-derived extracellular vesicles, the exosomes can act as delivery vehicles for small RNAs, especially siRNA and miRNA, owing to their wide distribution and similarity to cell membranes; they have favorable biocompatibility, less toxicity, deep penetrating property, etc. [144]. For instance, miR-155 delivered by functionalized exosomes could efficiently reach the target cells in a mouse model to inhibit mRNA and alleviate CCL4-induced liver injury [145]. Collectively, the natural material-based delivery systems have obtained a significant interest in miRNA delivery due to their unique advantages, including their easy synthesis, decreased cost, flexible structures, inherent biocompatibility, and biodegradability.
Notably, the biomaterials can be modified to co-deliver multiple therapeutic nucleic acids simultaneously in order to make use of their diverse functions. Kim et al. used nanocarriers, consisting of PEI and dexamethasone, to co-deliver the pDNA and shRNA for the overexpression/knockdown of differentiation-associated proteins in human mesenchymal stem cells (MSC), causing morphological changes in MSC [146]. Additionally, the combination therapeutic delivery of siRNA and miRNA could precisely knock down a specific set of genes and target multiple disease-related pathways at the same time. Multifunctional tumor-penetrating mesoporous silica nanoparticles were developed for the co-delivery of siPlk1 siRNA and miR-200c [147]. For facilitating the endosomal escape and deeper tumor penetration, a photosensitizer indocyanine green and iRGD peptide were utilized, respectively. This approach showed a combined cell-killing activity and significant reduction of primary tumor growth as well as metastasis. In conclusion, the applications of biomaterials can significantly increase the therapeutic potential of RNA-based therapy. Different biomaterials, especially the lipid-based materials and polymeric nanoparticles, can be optimized to deliver the intact genetic materials to their site of action (cytosol/nucleus) both in-vitro and in-vivo, with some already clinical-stage studies. Still, some obstacles are needed to be overcome. Two of the general concerns include the potential toxicity and undesired biomaterial-related immune responses [120].
4.3. Delivery of CAR gene
The applications of CAR-modified T (CAR-T) cells have shown tremendous success in treating the hematologic malignancies and promising responses in solid tumors [148]. The CAR-T cells are genetically modified T cells, which stably express the cancer-targeting CAR on their surface, allowing them to specifically recognize and combat tumor cells. In recent years, the cationic polymers and LNPs have gained considerable interest in delivering the CAR gene to different lymphocytes. Yu et al. developed a self-assembled nanoparticle derived from the PEI and PAMAM for the transient transfection of Jurkat cells with a plasmid vector, expressing the EGFR variant III (EGFRvIII) CAR gene [148]. The expression of EGFRvIII-CAR on cell membrane could redirect the T cells to specifically recognize and destroy the EGFRvIII-positive tumor cells. The ionizable LNPs have been used in mRNA-based ex-vivo engineering of CAR-T cells, showing cancer-killing potency comparable to that of the electroporation- and virally-engineered CAR-T cells [149]. Of note, the natural killer (NK) cells are a special subtype of effector lymphocytes. The CAR-NK cells are considered as a next-generation cell-based cancer therapy. Kim et al. developed the multifunctional nanoparticles, which were synthesized with a cationic polymer-conjugated core-shell and a polydopamine coating layer, for the delivery of pDNA with EGFR-CAR gene into NK cells [150]. The engineered NK cells showed an enhanced expression of EGFR-CARs and cytotoxicity against human breast carcinoma [150]. Nevertheless, the current manufacturing approaches of CAR-T cells have been elaborated in clinical practice, including T cell isolation, ex-vivo activation, proliferation, genetic modification, and reinfusion of CAR-T cells. This makes CAR-T therapy too expensive for many patients. The cost for an annual course of the two commercial CAR-T products, Kymriah® and Yescarta®, is $475,000 and $373,000, respectively [151]. A promising solution could be the use of nanoparticle-based T cell programming in-situ.
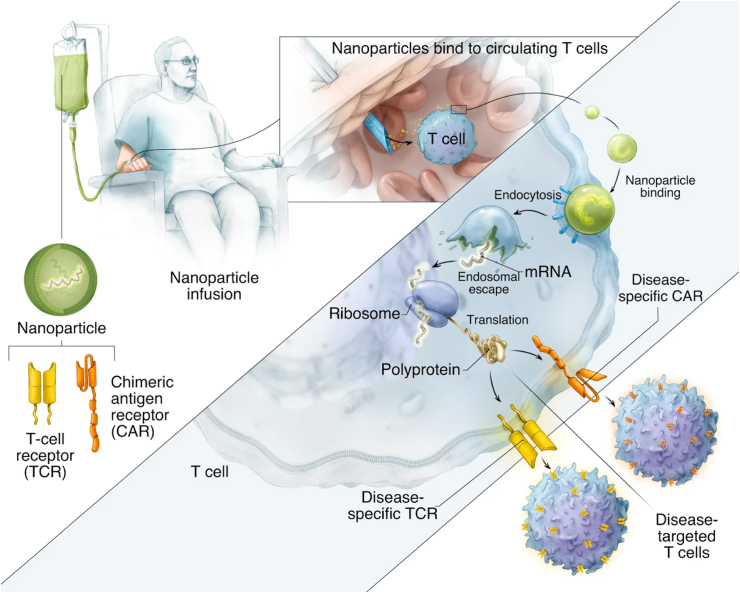
In 2017, Smith et al. revolutionarily delivered CD19-specific CAR genes into the circulating T cells in-situ using polymeric nanoparticles, consisting of PBAE 447, pDNA expressing CAR gene, T cell-targeting anti-CD3ε f(ab)2 fragments, nuclear localization peptide, and polyglutamic acid antibody [152]. These reprogrammed T cells could induce a long-term leukemia remission in mice with efficacy comparable to the conventional infusions of CAR-T cells transduced ex-vivo with viral vectors. They further utilized an injectable polymeric nanocarrier to deliver an in-vitro-transcribed mRNA for the transient expression of disease-specific receptors in circulating T cells, which included CD19-specific CAR and HB core-specific TCR. The repeated administration of mRNA nano-drug could induce antitumor responses against lymphoma, prostate cancer, and HBV-induced hepatocellular carcinoma in mice (Fig. 3) [153]. The in-situ T cell reprogramming might significantly simplify the complex procedures currently used in the clinic. The key features, including high efficiency, easy scale-up, low cost, and customizable properties, might help promote this nanomedicine toward clinical use. However, the requirement of repeated dosing and multicomponent manufacturing might potentially limit the nanoparticle-based CAR-T cell engineering; the latter might also draw concerns about safety issues.
Fig. 3.
Schematic illustration of in-situ T cells reprogramming to express the disease-specific CARs or TCRs using IVT mRNA carried by polymeric nanoparticles. Reproduced under the CC-BY license terms [153].
4.4. Delivery of CRISPR-Cas9 system
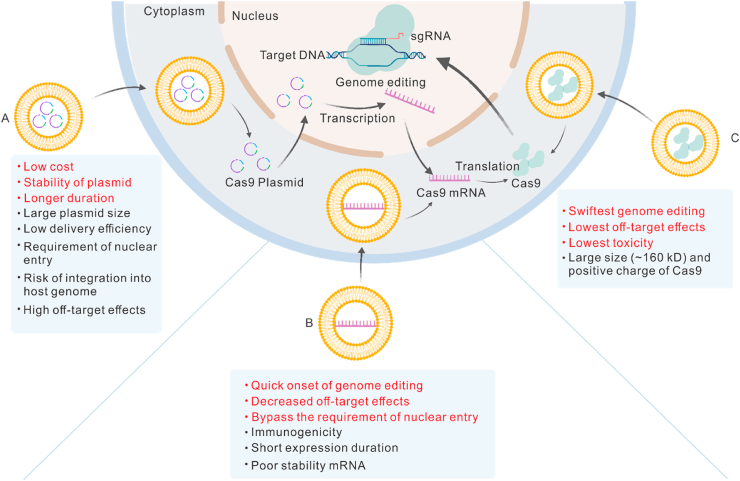
CRISPR-Cas9 system is a promising gene-editing tool, which is used in biomedical and clinical studies and can effectively ablate the disease-causing genes from a genome using a single guide RNA (sgRNA)-directed Cas9 nuclease [154]. Its development was recognized with the 2020 Nobel Prize in Chemistry. However, due to its large size, a major obstacle in its therapeutic application is the lack of safe and efficient delivery systems. Recently, biomaterials have shown a great potential for the delivery of CRISPR-Cas9, owing to their large capacity, versatility, biocompatibility, and high transfection efficiency. The Cas9 nuclease can be delivered in three different forms, including pDNA, mRNA, and protein (Fig. 4). Each of these delivery approaches has pros and cons.
Fig. 4.
Three strategies for the delivery of the CRISPR-Cas9 system. Cas9 nuclease can be delivered in plasmid DNA (A), mRNA (B), or protein (C) forms. Each of these delivery approaches has its pros (red) and cons (black). (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
The delivery of Cas9 nuclease encoding pDNA is attractive due to its lower cost, better stability, and longer duration [155]. The obstacles, such as the large plasmid size, low delivery efficiency, requirement of nuclear entry signal, risk of integration into host genome, and high off-target effects, can significantly limit its development and application. Different biomaterials have been investigated to overcome these challenges. Kretzmann et al. engineered a flexible dendritic polymer, which could successfully deliver large plasmids (Cas9: 9.8 kb; sgRNA: 3.2 kb) [156] with enhanced packaging capacity and transfection efficiency as compared to Lipofectamine 2000, which can only deliver small pDNA (∼5 kb). An artificial virus, consisting of a fluorinated polymer core and a multifunctional shell, which was acquired by modifying the hyaluronan polymer with PEG side chains and peptide, showed a more efficient transfection of Cas9 plasmid as compared to the commercially available Lipofectamine 2000 and Lipofectamine 3000 [157]. A microcarrier, which was composed of degradable polymers (polypeptides and polysaccharides) and modified by silica shell, also showed a higher Cas9 plasmid transfection efficiency than the liposome-based transfection reagents (>40% vs. 20%) in HEK 293 T cells [158]. In order to improve the targeted delivery, a GNP-condensed, lipid-encapsulated, and laser-controlled delivery system was developed for the selective delivery of the Cas9-sgRNA plasmid, targeting the Plk-1 gene [159]. The multifunctional lipid shell guaranteed the stability of the plasmid and facilitated cellular internalization. The inner GNP core permitted the plasmid condensation and thermo-triggered release of the CRISPR-Cas9 system into the cytosol of melanoma tumor cells. The modification of TAT peptide (a cell-penetrating peptide) mediated the entry of plasmid to the nucleus. These synergistic effects finally enabled an effective knockout of the Plk-1 gene and inhibited tumor growth both in-vitro and in-vivo.
An alternative strategy is the delivery of Cas9 mRNA and sgRNA. This method leads to the rapid action of genome editing and decreased off-target actions, bypassing the requirement of nuclear entry for transcription. Although this is a promising method for genome editing, the challenges, including immunogenicity, short-expression duration, and poor stability of mRNA, restrict its efficiency and safety. Therefore, the use of gene vectors especially lipid-based biomaterials and polymers are necessary. In a recent study, cationic lipid-assisted PEG and PLGA-based nanoparticles were used to deliver CRISPR-Cas9 components, targeting the costimulatory molecule CD40, in mRNA form. The intravenous injection of Cas9 mRNA and sgRNA encapsulated in nanoparticles could induce the disruption of CD40 in DCs, which inhibited the DC-mediated activation of T cells and significantly relieved the skin transplant rejection response in an acute rejection mouse model [160]. LNPs have also been used for the delivery of Cas9 mRNA and sgRNA, targeting the HBV-DNA and hypercholesterolemia-associated gene, for the treatment of liver-related diseases in mice [161]. Although most of the mRNA nanocarriers are cationic biomaterials, which can wrap the negatively charged nucleic acids via electrostatic interactions, Miller et al. synthesized zwitterionic amino LNPs for the delivery of Cas9 mRNA and sgRNAs both in-vitro and in-vivo [162]. Moreover, viral and non-viral vectors can be combined for the delivery of CRISPR-Cas9 components. Yin et al. combined the LNP-mediated Cas9 mRNA delivery with adeno-associated viruses, encoding a sgRNA, in a mouse model of human hereditary tyrosinemia [163]. This combined treatment was highly efficient by correcting >6% of the hepatocytes after a single dose and improved the disease symptoms, including weight loss and liver damage.
The most straightforward to deliver CRISPR CAS9 system is the delivery of native Cas9 protein and sgRNA, which enables the fastest genome editing with the lowest off-target effects and toxicity. However, the large size (∼160 kD) and positive charge of Cas9 protein have tremendously challenged its delivery. The widely used cationic lipids or polymers also face difficulty in encapsulating the Cas9 protein via electrostatic interaction [155]. The current strategies of using biomaterials for the delivery of Cas9 protein have focused on three approaches; 1) Direct conjugation of biomaterials with Cas9 protein by a covalent bond. The cell-penetrating peptide-based nanoparticles can conjugate with Cas9 protein by a thioether bond and form a complex with sgRNA in order to form condensed and positively charged nanoparticles for the CRISPR-Cas9 delivery in protein form [164]. 2) Make use of the native bind of Cas9 protein to anionic sgRNA. A single treatment dose of cationic lipid-mediated delivery of Cas9/sgRNA complexes could modify the genome in 80% cultured cells with about 10-fold higher specificity than the plasmid transfection [165]. The cationic lipid could also deliver unmodified Cas9/sgRNA complexes to hair cells in the inner ear of mice, which could disrupt the dominant deafness-associated gene defect and reduce progressive hearing loss [165,166]. DNA nanoclews were used to load Cas9/sgRNA complexes and deliver them to the cell nucleus [167]. The metal-containing nanoscale zeolitic imidazole frameworks could encapsulate Cas9/sgRNA complexes with a loading efficiency of 17% in 4 days [168]. 3) Modify Cas9 protein with negative charges. The surface charges of Cas9 protein can be altered to enhance its electrostatic interactions with cationic carriers. For example, the negatively charged glutamate peptide tag- [169] and boronic acid-rich dendrimers- [170] modified Cas9 protein could be more efficiently delivered by cationic nanocarriers, followed by more effective gene editing in numerous cell lines.
4.5. Co-delivery of nucleic acid and therapeutic drugs
The combined delivery strategies have also been investigated to improve the therapeutic efficacy and reduce the side effects. Lin et al. recently created a cationic peptide-based nano pro-drug co-delivery system for the delivery of both cisplatin and Beclin1 siRNA (an autophagy inhibitor factor). DSPE-PEG was used to improve the stability and biocompatibility of this delivery system through PEGylation, while cRGD was used to acquire the tumor cell-targeted delivery. After consuming the intracellular thiol-containing species, such as GSH, the nanoparticle decomposed, releasing active Pt (II) for chemotherapy and Beclin1 siRNA for the mediation of tumor cell autophagy. This co-delivery strategy could successfully reverse the acquired resistance and showed a more remarkable efficacy against cisplatin-resistant tumors after the intravenous injection as compared to the free cisplatin administration [20]. Similarly, the LNPs were used for the co-delivery of melphalan and miR-181a for treating the seeded retinoblastoma [171]. These two agents showed complementary effects on decreasing the viability of cultured retinoblastoma cells and enhancing the treatment efficacy in a retinoblastoma xenograft rat model as compared to monotherapy.
5. Biomaterials in stem cell therapy
Stem cells are undifferentiated cells, which are capable of self-perpetuation and differentiation into the specialized cells of various tissues and organs. By guiding their cell fates, stem cell therapy has gained great attention for its widespread biomedical applications, such as tissue engineering and regenerative medicine [172]. Nevertheless, the stem cells seeded in traditional two-dimensional (2D) cultures often show different characteristics from those after being transplanted in-vivo. This is because the flat and stiff 2D environment fails to replicate the endogenous 3D microenvironment [173]. However, the 3D culture systems are more physiologically relevant, providing cells with necessary physical and chemical supports. Additionally, the 3D culture systems are inherently more scalable and yield a higher number of cells than the 2D cultures with equal space. Natural materials, such as gelatin, laminin, collagen, alginate, HA, and decellularized ECM, have been used for the construction of 3D scaffolds for cell culture. However, the difficulty in biochemical modification and risk of immune rejection have limited their exploration and clinical applications [173,174]. The artificial materials, such as organic and inorganic porous materials, can be surface modified, mechanically tuned, or biochemically functionalized for the construction of 3D scaffolds, which might mimic the natural ECM (Fig. 5). Polymer-based hydrogels are highly attractive due to their ECM-like properties (tissue-similar elasticity, high water content, nutrients, as well as waste transportation capability) and optical clarity, enabling the nondestructive imaging of cell function [175].
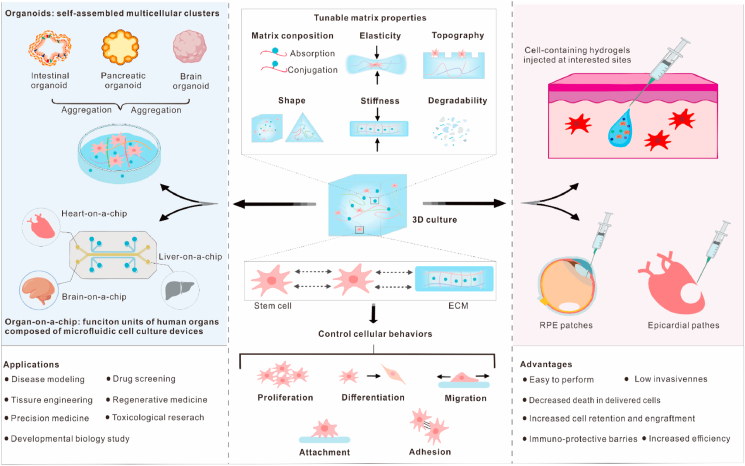
Fig. 5.
Application of biomaterials in stem cell therapy. By altering the tunable matrix properties of 3D cell culture, the cellular behaviors, including proliferation, differentiation, migration, attachment, and adhesion, can be controlled. The 3D culture with tunable properties can be spatiotemporally controlled to mimic the native ECM for the further guidance of stem cell behaviors to form desired organoids or organ-on-a-chip (OOC). Organoids and OOC have a wide range of applications, such as disease modeling, drug screening, etc. These properties of 3D culture can also be incorporated into designing the injectable hydrogels and stem cell patches for the delivery of stem cells. These cell delivery vehicles possess outstanding advantages, such as low invasiveness, high efficiency, decreased cell death, etc. ECM, extracellular matrix; and RPE, retinal pigment epithelium.
5.1. Biophysical cues of 3D culture and cell behaviors
Stem cells can proliferate, differentiate, attach, and migrate in specialized microenvironments with specific extracellular conditions. The current bottlenecks for the effectiveness of stem cell therapies include reproducible cell expansion for transplantation, maintenance of stemness (self-renewability and differentiation into multiple cell types), and prevention of cell death. The physiochemical properties of 3D culture systems, such as shape, porosity, topography, composition, elasticity, stiffness, and degradability, are closely related to a variety of cellular behaviors through the cell-cell and cell-ECM interactions in-vitro and in-vivo. By refining these factors, biomaterials can be used to control the behaviors and functions of stem cells. The matrix composition can be achieved by the adsorption or chemical conjugation of bioactive motifs and cell-binding domains with synthetic biomaterials. For instance, the biomaterials modified for integrin interactions could promote cell adhesion and induce stem cell pluripotency (iPSC) by activating the downstream kinase signaling pathway [176]. HA-based hydrogels could allow the invasion of highly expressing HA receptor CD44v6 cells through HA-CD44v6 binding [174]. The elasticity of both porous and non-porous materials has a profound effect on the behaviors of stem cells. Unlike the viscoelastic natural ECM, most of the non-degradable hydrogels were purely elastic despite adopting some physiological characteristics of the natural ECM [177]. Huebsch et al. proved that, by modifying the elastic modulus of void-forming alginate hydrogels, the behaviors of MSC osteogenesis, including cell deployment, differentiation, and proliferation in-vitro and expansion and dissemination in-vivo, was highly controllable [178]. Changing the hydrogel's elasticity could also regulate the efficacy of transplanted MSC for therapeutic bone regeneration in-situ [178].
Matrix stiffness is another key parameter for controlling the fate and activity of different stem cell populations in a 3D microenvironment. An increase in the onset of stress stiffening of synthetic polymer-based soft responsive hydrogels can readily alter the commitment and differentiation of human MSCs from adipogenesis to osteogenesis [179]. This effect is associated with the deformation and reorganization of stress stiffening-dependent microtubule cytoskeleton, which supports and controls the cell shape [179]. Of note, the alteration of initial hydrogel stiffness (∼0.5–50 kPa) could not affect the stemness maintenance of low contractible neural progenitor cells (NPCs) [180]. Therefore, it was speculated that the matrix stiffness only had an impact on the highly contractile stem cell types. The tunable stress relaxation and matrix remodeling of hydrogels can also regulate the fate and activity of stem cells. Unlike a 2D cell culture environment, where the cells are free to spread, the cells embedded within a 3D hydrogel require a matrix remodeling for migration and proliferation. An increase in the spreading, proliferation and differentiation of MSC was observed in an alginate gel with faster relaxation, which was mediated by the local adhesive ligands clustering and actomyosin contractility [177]. Madl et al. confirmed that matrix remodeling was necessary for the stemness maintenance of NPCs using three 3D biomaterial platforms, including two proteolytically degradable and physically changeable alginate hydrogels [180].
Therefore, there is an increasing interest in developing a dynamic adaptive biomaterial for controlling cellular fate and functions. ECM with reversible and tunable mechanical properties in response to the physical and chemical stimuli can facilitate the analysis of cells' interactions with their fluctuating mechanical environment. This is essential for understanding the physio-pathological processes, as well as regulating the growth and differentiation of stem cells in tissue engineering and regenerative medicine. Recently, an approach called 4D patterning has been used to reversibly pattern the cell-laden gel biomaterials by specific modification with bioactive site-specific proteins [175]. This strategy could unprecedentedly allow the precise regulation of advanced cellular functions in time and space. Hörner et al. synthesized a light-responsive polymer material to study the reaction of human MSCs to the dynamic mechanical environments and underlying signaling pathways [181]. The utilization of genetically encoded photoreceptors could allow a reversible, specific, precise, and spatiotemporal regulation of the matrix properties and cell behaviors in response to a tissue-penetrating light [181]. Additionally, the mutual interactions between stem cells and ECM inspired the construction of tunable materials. Jia et al. developed the adaptive liquid interfacially assembled protein nanosheets in order to dynamically control the behaviors and fate of MSCs [182]. In this system, stem cell traction force could induce the spatial rearrangement of ECM proteins, which in turn interfered with the cell fate.
5.2. Organoids and organ-on-a-chip
In recent years, organoids and OOC are the representatives of revolutionary technological breakthroughs in biomedicine and drug development and have enabled the construction of 3D tissues/organs to mimic the complex structures and functions of living human organs in-vitro. This development has bridged the gaps between animal models and significantly diverse human bodies [183]. The organoids and OOC technologies have potential applications in disease modeling, drug screening, developmental biology study, toxicological studies, tissue engineering, regenerative medicine, and precision medicine. Recent advances in the 3D cell culture system have tremendously facilitated the production of stem cell-derived organoids and OOC.
Organoids were first reported by the Clevers’ group by embedding the intestinal stem cells in ECM substitute to generate a crypt-like structure, resembling the native intestinal tissues [184]. Currently, they are defined as self-assembled multicellular clusters derived from the embryonic stem cells (ESC), iPSC, or tissue-specific stem cells [185]. The 3D cell culture, having increased cell-cell and cell-ECM interactions, might create organoids more efficiently than the conventional 2D culture. Biomaterials, especially hydrogels, with tunable properties, can be spatiotemporally controlled to mimic the native ECM for further guiding the stem cell behaviors to form the desired high-fidelity organoids. These biomaterials have been utilized in a variety of organoids, including lung, heart, intestine, pancreas, colon, liver, kidney, prostate, brain, blood vessel, etc. [185]. For instance, Tam et al. developed a well-defined biomimetic 3D hydrogel for the construction of an invasive lung disease model for the high-content drug screening [174]. A polymer-based scalable 3D tissue cultivation platform, named Biowire II, could enable the self-assembly of atrial and ventricular tissues derived from iPSC [186]. The alterations in the properties of culturing environments might interfere with the formation of organoids. Human iPSC differentiated on soft hydrogels showed an accelerated production of kidney organoids with more renal vesicles and nephron structures as compared to those produced under stiffer conditions [187]. The systematic substitution of physical components for removing the adherent forces or scaffolding components could dramatically increase and decrease the cyst formation in iPSC-derived kidney organoids, respectively [188]. Similarly, Shao et al. demonstrated that the physical signals originated from a bioengineered implantation-like niche, rather than a biochemical inductive cue, which could trigger the development of amnion-like tissues in iPSCs for modeling the human self-organized amniogenesis [189]. Therefore, it is a promising strategy to create the desired organoids with increased control over their morphogenesis and functions by intentionally modifying the in-vitro culture system for stem cells. However, the engineering and translational applications of organoids are primarily limited by variabilities in their cellular composition, phenotypes, and architecture, Future biomaterials and bioengineering approaches for designing the organoids should focus on the safety and stability concerns.
OOC is a miniaturized model of human organs’ functional units and is composed of microfluidic cell culture devices, where living human cells reside [185]. Hydrophilic hydrogels made from natural and synthetic biomaterials, especially polymers, are most frequently used for the construction of OOC devices. For the optimal engineering of sophisticated structures, various microfabrication approaches, including 3D photopatterning [190], 3D bioprinting, micro-molding, and microfluidic spinning [191], have been applied to build the perfusable microchannels in hydrogels. For instance, 3D bioprinting technology has allowed the precise pattern of living cells in pre-defined spatial positions. A 3D bioprinting-based hybrid method has been developed to efficiently engineer the endothelialized-myocardium- and heart-on-a-chip platforms using human iPSC-derived cardiomyocytes for screening the pharmaceutical compounds for their cardiovascular toxicities [192]. Other representatives of the biomaterial-supported OOC include liver-on-a-chip and brain-on-a-chip. A model brain-on-a-chip was produced in a novel microfluidic cell culture device for modeling epileptic seizures with iPSC-derived functionally connected cortical networks [193]. Christoffersson et al. used the hyaluronan-PEG-based hydrogels for the development of a perfused liver-on-a-chip device, where the human iPSC-derived hepatocytes remained highly viable and functional for a long time [194]. Collectively, the biomaterials are essential for the generation of OOC devices by serving as alternative chip materials, biomimetic tissue barrier, and an analogous cellular 3D matrix.
5.3. Stem cell carriers
The key determinants for the efficacy of stem cell therapy are the survival and function of cells during and post-transplantation. Biomaterials can be used to deliver and protect the stem cells from mechanical forces and provide a supportive microenvironment for their optimal function at a specific site. Apart from cells, these materials can also be used for the co-delivery of therapeutic agents, such as anti-inflammatory drugs, in order to promote the function of transplanted stem cells [195]. Recent advances in biomaterial-based stem cell delivery strategies have focused on highly efficient stem cell patches and less invasive injectable hydrogels. The biomaterial-based stem cell patches might be advantageous that outweigh the ease of injectable hydrogels in cell delivery for tissue engineering. These patches can be delivered through microsurgical tools, thereby offering the robust mechanical, contractile, and electrical properties to functionally support the transplanted stem cells in the targeted region [196]. A comparative study of biomaterials for the delivery of MSC in rat myocardial infarction models revealed that the epicardial patches could increase the cell viability 5-fold as compared to the injectable hydrogels [197]. Human iPSC-derived cardiac muscle patches fabricated with ECM-based scaffolds resulted in a significant decrease in the infarction size and enhanced the cardiac function without affecting the arrhythmogenicity in mice [198] and swine [199] models. These studies evaluated the therapeutic efficacy and safety of stem cell patches in ischemic myocardial injury in large animal models, which might help in guiding its clinical translation. The stem cell patches can also be used for the treatment of ophthalmic diseases. Human iPSC-derived retinal pigment epithelium (RPE) patches in biodegradable scaffolds showed an increase in the integration and function of RPE, thereby rescuing the degeneration of retina in rat and pig age-related macular degeneration (AMD) models [200]. The ESC-derived RPE patches could improve and stabilize the vision for at least 12 months in two AMD patients with serious vision loss [201].
Injectable hydrogels are preferred over the surgical intervention of stem cells into host tissues due to their low invasiveness. Numerous cells die due to mechanical disruption during the process of direct injection through a needle [202]. Hydrogels can protect the stem cells from membrane damage during injection by altering the fluid flow profile through the springe and avoiding the shear deformation [203]. After injection into tissues, the low retention and engraftment of cells and host immune response can significantly limit the use of stem cell transplantation. On the other hand, hydrogel carriers can serve as scaffolding materials to improve the retention of stem cells after injection [204,205]. They can also serve as immuno-protective barriers to inhibit immune cell infiltration and protect the encapsulated stem cells from the attack of immune cells [205]. In the central nervous system, hydrogels could increase the survival rate of injected human neural stem cells by inhibiting the microglial infiltration at the early and acute inflammatory stage of stroke [206]. The matrix properties used for the regulation of cell behaviors in cultures can be incorporated into hydrogel carriers in order to enhance the efficacy of stem cell therapy. In one example, altering the physical properties of nanoporous gel cocoons could promote the proliferation and migration of encapsulated human cardiac stem cells, eventually enhancing the generation of new blood vessels and cardiomyocytes [204]. When injectable hydrogels were used to deliver MSCs, their elasticity had an impact on bone regeneration with an optimal bone formation of 60 kPa [178]. In summary, biomaterials can provide 3D-organized carriers for supporting the viability and functions of stem cells, thereby improving the therapeutic impact of stem cell therapies. By changing the tunable cues of biomaterials, the cellular activities can be guided and personalized stem cell therapy can be promoted.
6. Implantable scaffolds and injectable biomaterials
As mentioned above, the biomaterial scaffolds have been widely developed for regenerative medicine in order to provide an optimal stem cell niche and modulate their gene expression, expansion and migration [207]. In addition to cellular regenerative medicine, biomaterial scaffolds are also developed for acellular regenerative therapeutics because they can provide an ECM and allow the infiltration of the body cells [208]. Furthermore, biomaterial scaffolds, including the implantable and injectable scaffolds, can be preloaded or functionalized using biological factors, therapeutic agents, or even cells. The implantable scaffolds can be implanted into a subcutaneous or excised tissue through minor surgery. The loaded bioactive agents remain at their implanted place for an extended period and enable a sustained release to perform various functions. The use of implantable scaffolds as DDS was originated in the 1960s, when silicones were used to prolong the therapeutic efficacy. The implantable DDSs are now widely applied in birth control and vascular/ocular disease treatment as reviewed by Kleiner et al. [209]. The present review describes a 3D-printed scaffold loaded with isoniazid (INH)/rifampin (RFP) for osteoarticular tuberculosis therapy [210]. Researchers loaded the high dosages of INH/RFP into mesoporous ceramics, which was then conjugated with poly 3-hydroxybutyrate-co-3-hydroxyhexanoate (PHBHHx), to form a composite scaffold using 3D printing. The hierarchical composite scaffolds showed a prolonged drug release for over 84 days as compared to the 5 days of commercial calcium phosphate scaffolds. Besides, the drug concentrations of INH and RFP were maintained above the threshold ensuring the effective Bacillus-killing in peripheral tissues and remained low in blood circulation, indicating minimal harm to the functions of normal organs. Researchers also observed a partial degradation of scaffolds and new bone growth in the cavity after debridement surgery. These promising results suggested that the 3D printed composite scaffolds had a good prospect for the treatment of osteoarticular tuberculosis. In addition, PBAEs are biocompatible and biodegradable, and their degradation time can be adjusted from hours to months. Clark and his colleagues created a novel hybrid scaffold by mixing the PLGA microspheres with PBAE porogen. The PBAE particles encapsulated with ketoprofen provided a rapid 12 h release, while the PLGA microspheres loaded with bone morphogenetic protein 2 (BMP-2) provided a sustained release lasting for 70 days [211].
Adoptive T cell (ATC) immunotherapy has shown significant survival benefits among patients with hematologic malignancies. However, the efficacy of ATC immunotherapy has been limited in solid tumors due to the inefficient expansion of T cells in the immunosuppressive microenvironment. It is valuable to develop feasible biomaterials capable of supporting the T cell viability and delivering tumor-reactive T cells. Stephan et al. integrated, which is a synthetic collagen-mimetic peptide, GFOGER with macroporous scaffolds, made from an FDA-approved polymerized alginate, add them to microparticles that could harbor and release the stimulatory antibodies and IL-15 superagonist for T cell proliferation. In the mouse breast and ovarian tumor resection models, the alginate scaffold could effectively promote the adhesion and migration of T cells, facilitate the expansion and dispersion of T cells into tumor-resected beds, and eventually enhance the tumor regression as compared to the conventional injection of T cells [212].
The implantable scaffolds can also be used to deliver activated DCs. Verma et al. developed a fibrin-derived scaffold, harboring the DCs to activate specific immune responses against primary and post-surgery secondary tumors [213]. Furthermore, scaffold-based vaccines have been widely developed. Hathaichanok and colleagues designed a porous 3D scaffold by cross-linking the collagen and HA for delivering the whole tumor lysate vaccine and its adjuvant anionic poly(I: C) agonists loaded in nanogels [214]. A chemotherapeutic agent gemcitabine (GEM) was co-delivered using this platform, which could deplete the myeloid-derived suppressor cells (MDSCs) for establishing a permissive immunogenic microenvironment. In a mouse breast tumor resection model, the implantation of this scaffold could increase the frequency of activated DCs, CD4+ T cells, CD8+ T cells, as well as the production of IFN-γ. It was shown that 25% of the treated mice experienced complete remission and 100% survival with reduced tumor metastasis [214]. The PLGA vaccine-loaded scaffold, consisting of tumor lysate, GM-CSF, and CPG, known as WDVAX, is currently in its phase I clinical trial for the treatment of stage IV melanoma (NCT01753089).
Besides, the utilization of implantable scaffolds requires invasive surgery, which cannot be performed in volume-sensitive or surgically inaccessible areas. Furthermore, the implantable scaffolds often remain at the implanted site for a long period, thereby compromising the function of normal organs, and struggle with immune rejection, causing fibrosis [215]. For example, the serum amylase and lipase levels were lower in the pancreatic tumor-bearing mice treated with the implants as compared to those of the untreated ones, suggesting an exocrine pancreatic insufficiency after the scaffold implantation [216]. In order to overcome these limitations of implantable scaffolds, injectable biomaterials have been developed, which can be administered through a conventional needle, subsequently forming a 3D scaffold after injection into the tissues [217]. This might avoid the complications associated with the surgical implantation and unnecessary tissue damages. Besides, they can be injected into any available space where a needle can reach. This injection requires less technical expertise and causes less distress in the patients as compared to surgical procedures. However, the injectable biomaterials are required to pass through the needle either as a liquid or a gel, which heavily limits its use. Table 2 summarizes the differences between implantable scaffolds and injectable biomaterials.
Table 2.
Differences between implantable scaffolds and injectable biomaterials.
| Implantable scaffold | Injectable biomaterial | |
|---|---|---|
| Advantages | Enable sustained release | Avoid surgery-related complications |
| Preload with biological factors, therapeutic agents, or even cells | Require less technical expertise | |
| Disadvantages | Require invasive surgery | Limit material types |
| Limit the desired implanted place | ||
| Cause fibrosis and compromise normal organ function | ||
| Material type | Drug delivery chips; osmosis pumps; polymer conduits for nerve regeneration, etc. | Self-assembling peptide hydrogels; macroporous cryogel sponges; liposome, etc. |
| Applications | Drug delivery system [210] | Drug delivery system [218] |
| Deliver adoptive T cells and activated dendritic cells [212] | Deliver tumor-specific CD8+ T lymphocytes [219] | |
| Provide an optimal stem cell niche [220] | Serve as an injectable vaccine platform [221] | |
| Re-establish tolerance to treat autoimmune diseases [222] |
Injectable biomaterials can serve as DDS. In one case, Li et al. utilized an alginate hydrogel system for the combined delivery of PD-1 monoclonal antibody (mAb) and celecoxib, which could improve the deleterious chronic inflammation induced by anti-PD-1 therapy [218]. In the B16–F10 melanoma and 4T1 metastatic breast cancer models, this co-delivery system could significantly improve the anti-tumor efficacy with complete tumor regression among 5 out of 9 mice as compared to only 1 out of 9 survivals in the mice treated with hydrogel-free celecoxib and PD-1 mAb. Further analysis indicated that the combination of hydrogel systems could increase the frequency of CD4+ IFN-γ+ and CD8+ IFN-γ+ T cells, thereby decreasing the presence of CD4+ FoxP3+ regulatory T cells (Tregs) and MDSCs within the tumor site. This system could also promote the expression of chemokines C-X-C motif ligand (CXCL) 9 and CXCL10 and inhibit the expression of IL-1, IL-6, and cycloxygenase-2 (COX2), thereby suggesting a repressed pro-tumor angiogenic and inflammatory microenvironment.
As combined with sodium hydrogen carbonate (SHC) and phosphate buffer (PB), the mechanical properties of chitosan hydrogels can be improved for maintaining their thermosensitive properties and rapid gelation at normal body temperature. Recently, Monette et al. developed injectable chitosan thermogels (CTGels) for the local delivery of tumor-specific CD8+ T lymphocytes [219]. They compared three different concentrations of SHC to form hydrogens; SHC 0.05 M (CTGel1), SHC 0.075 M (CTGel2), and SHC 0.12 M (CTGel3), among which, CTGel2 to found be superior. Therefore, the CTGel2 was further studied to investigate its potential for ATC immunotherapy. The results showed that the CTGel2 could encapsulate viable and effective CD8+ T lymphocytes and support their proliferation and gradual release for at least 24 days. Besides, the chitosan scaffolds degraded over time, thereby preventing any possible side effects induced by the long-term continuation of T cell proliferation.
Bencherif et al. designed a macroporous cryogel sponge as an injectable vaccination platform for the co-delivery of irradiated cells as an antigen source, GM-CSF, and a specific TLR agonist as DC enhancement factors, along with CpG ODN, which activates DCs [221]. This cryogel could release the loaded immunotherapeutic agents for over one month and significantly improve the anti-tumor efficacy. In a melanoma mouse model, the overall survival rate of mice vaccinated using the above cryogel was up to 80% as compared to less than 20% in the mice vaccinated with a bolus, suggesting the therapeutic potential of the 3D cryogel vaccination platform. No pathologic changes were observed in the vital organs of the vaccinated mice, indicating the safety of the injectable cryogel vaccination platform. Similarly, Li et al. developed a mesoporous silica micro-rod (MSR)-based vaccine [223]. The simple mixing of MSR with PEI, the MSR-PEI vaccine could significantly enhance the absorption and immunogenicity of antigens. Impressively, a single injection of MSR-PEI vaccine could completely eradicate the large established TC-1 tumors in 80% of the mice when immunized with E7 peptide was used and eradicate the established lung metastases when immunized with B16F10 or CT26 neoantigens pool. As compared to the standard MSR vaccine system, the MSR-PEI vaccine could significantly increase mice survival by two-fold. The biomaterial scaffolds not only activate the immune responses but also re-establish tolerance to autoimmune diseases by modulating the DC responses in a non-inflammatory context. Verbeke et al. reported an injectable, porous hydrogel to deliver GM-CSF and BDC peptide antigen, which could be recognized by BDC2.5 T cells, causing an induction of a Treg response in a NOD (non-obese diabetic) mouse model of type 1 diabetes [222].
With the advancements in scaffold fabrication techniques, the scaffold biocompatibility, bioactivity, biodegradability, and mechanical properties are tunable by modulating the material composition. Biomaterial scaffolds have become attractive candidates for drug and cell delivery due to their advantages of targetability, high loading efficiency, controllable content release, long duration of action, and protection of encapsulated molecules and cells. However, the issue of lacking imaging tools, which can monitor the cellular processes in-situ within the whole volume of scaffolds, remains unsolved. Therefore, the development of bio-imaging tools for maintaining a high temporal and spatial resolution to monitor the cellular events within 3D scaffolds, is critical in future studies [224].
7. Conclusions and outlook
In the past decades, biomaterial-assisted biotherapies have been a booming field of research accompanied by interdisciplinary developments in molecular biology, material science, nanotechnology, etc. With the improved therapeutic efficacy and limited side effects of biomaterial-assisted biotherapies, several products have been successfully commercialized (Table 3) or are currently being investigated in human clinical trials. Doxil® is the first FDA-approved nano-drug, which encapsulates doxorubicin in PEGylated-liposomes for a prolonged circulation time and passively targets the tumor sites [225]. Doxil® has shown an improved antitumor efficacy against a great variety of neoplastic disorders and a more remarkable reduction of cardiac toxicity than conventional doxorubicin [226]. LNPs are also attractive delivery vectors for genetic materials due to their higher tolerability and loading capacity than the other types of biomaterials. Lipid-based siRNA therapy (such as Onpattro®) and mRNA vaccines (such as Comirnaty®) have made a great success in the battle against hereditary and infectious diseases. Polymeric biomaterial-based local drug delivery systems could allow a sustained drug release, minimizing the requirement of a high-dose and repeated administration. Some examples of the commercialized polymeric products include PLGA microsphere-based hormone therapeutics (Leupron Depot® for advanced prostate cancer and Neutropin subcutaneous Depot® for growth hormone deficiency) and poly l-lactide-based vascular scaffold systems (Absorb GT1) for coronary artery diseases. VLPs are appealing vaccine carriers due to their high resemblance with natural viruses in their structures and functions, thereby significantly improving the immunogenicity of VLP-based vaccines. Cervarix®, Gardasil®, and Gardasil9® are the representatives of commercialized VLP vaccines, which have been reported to induce a life-long protective immunity against HPV infection [227,228]. Besides, some more advanced biomaterials are now under clinical evaluation. For example, the CRLX101, a pH-responsive cyclodextrin-based nanocarrier, containing camptothecin [229], has completed the phase II clinical trials (NCT01612546). In the case of biomaterial-assisted vaccines, the VLP-based HPV vaccine has been successfully commercialized (Cervarix®, Gardasil®, and Gardasil9®).
Table 3.
Several commercially available biomaterial-based products.
| Commercial name | License year | Biomaterials | Treatment | Indications |
|---|---|---|---|---|
| Leupron Depot® | 1989 | PLGA microspheres | Hormone therapy | Advanced prostate cancer |
| Norplant® implant | 1990 | 6 silicone rubber tubes, crosslinked polydimethylsiloxane | Implant (Levonorgestrel) | Contraceptive |
| Doxil® | 1995 | Lipid nanoparticles | Chemotherapy (Doxorubicin) | Breast neoplasms; multiple myeloma; ovarian neoplasms; Kaposi's sarcoma |
| Gliadel® | 1996 | PCPP-SA | Intracranial wafer | Treatment to surgery for recurrent glioblastoma multiforme |
| Neutropin subcutaneous Depot® | 1999 | PLGA | Microspheres | Growth hormone deficiency |
| Gardasil® | 2006 | Virus-like particle | Vaccine delivery | Cervical, vulvar and vaginal cancers |
| Absorb GT1 | 2016 | Poly (l-lactide) | Vascular scaffold system | Coronary artery disease |
| Onpattro® | 2018 | Lipid nanoparticles | Gene therapy (siRNA) | Polyneuropathy of hereditary transthyretin-mediated amyloidosis in adults |
| Dextenza® | 2018 | Polyethylene glycol-based Hydrogel conjugated with fluorescein |
Insert (Dexamethasone) | Ocular inflammation and pain following ophthalmic surgery |
| Comirnaty® | 2020 | Lipid nanoparticles | Vaccine delivery | SARS-CoV-2 infection |
PLGA, poly lactic-co-glycolic acid; PCPP-SA, poly-[1,3-bis carboxyphen-oxy propane-co-sebacic acid] anhydride; BVS, bioresorbable vascular scaffold system.
Nevertheless, there remain several challenges for biomaterial-assisted biotherapies. First, the animal models, which are chosen to assess these platforms, are of great significance to minimize the translational hurdles to human applications. These engineered biomaterials should be investigated in animal models, which must replicate human diseases most closely [230]. Therefore, researchers need to further explore the advantages and disadvantages of different animal models and the differences in immune responses between the humans and animal models [231]. In preclinical models, the factors, including animal housing, their diet, sex as well as commensal microbiota, might influence the response to biotherapies [232]. Second, simple and standardized fabrication processes are recommended to guarantee minimal batch-to-batch variability. Many essential factors for the clinical translation, including complexity, scalability, stability, production cost, etc., should be fully considered in the early stages of biomaterial designing [233]. Besides, the platforms using FDA-approved materials might simplify the regulatory processes and are more likely to get into the clinical practices. Third, although the biocompatibility of biomimetic nano-minerals has been demonstrated in animal models, the biosafety of most of the biomaterials has not been assessed in humans for clinical treatment. Challenges, including pharmacokinetics, pharmacodynamics, long-term efficacy, long-term side effects, and systemic toxicity, remain to be addressed. An adequate sample size and follow-up period, as well as tests in larger mammals, including dogs and non-human primates, in preclinical trials, are also essential [232].
A multimodal therapy, which combines biomaterial-assisted biotherapy with chemotherapy, radiotherapy, or photothermal therapy, has achieved increasing attention [234]. The development of biomaterial-based biotherapy is also expected to speed up with the advancements in big data and artificial intelligence (such as machine learning) for intelligent material design [235]. Besides, the combination of computer technology with organoids, genomics, and proteomics might facilitate the high throughput screening and preclinical studies [236]. Nevertheless, the dose-effect correlation in biomaterial-based biotherapy remains largely unknown. Future studies should investigate the dose window for more effective and safer delivery strategies [237]. Furthermore, biotheranostics (integration of biomaterial-assisted diagnosis and treatment) is also an innovative hotspot for precision medicine.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
This work was supported by the Natural Science Foundation of China (32122052, U19A2003, 82121003), the Key Research and Development Program of Science and Technology Department of Sichuan Province (2020YFS0570, 2020JDTD0028,2019YFS0514), the Science and Technology Project of Chengdu (2019-YF05-00784-SN), and the National Key Research and Development Program of China (2020YFC2005500).
Footnotes
Peer review under responsibility of KeAi Communications Co., Ltd.
Contributor Information
Xiawei Wei, Email: xiaweiwei@scu.edu.cn.
Lulu Cai, Email: cailulu@med.uestc.edu.cn.
References
- 1.Wei P.S., Chen Y.J., Lin S.Y., Chuang K.H., Sheu M.T., Ho H.O. In situ subcutaneously injectable thermosensitive PEG-PLGA diblock and PLGA-PEG-PLGA triblock copolymer composite as sustained delivery of bispecific anti-CD3 scFv T-cell/anti-EGFR Fab Engager (BiTEE) Biomaterials. 2021;278 doi: 10.1016/j.biomaterials.2021.121166. [DOI] [PubMed] [Google Scholar]
- 2.Lai Y., Li Y., Cao H., Long J., Wang X., Li L., et al. Osteogenic magnesium incorporated into PLGA/TCP porous scaffold by 3D printing for repairing challenging bone defect. Biomaterials. 2019;197:207–219. doi: 10.1016/j.biomaterials.2019.01.013. [DOI] [PubMed] [Google Scholar]
- 3.Kelly S.H., Wu Y., Varadhan A.K., Curvino E.J., Chong A.S., Collier J.H. Enabling sublingual peptide immunization with molecular self-assemblies. Biomaterials. 2020;241 doi: 10.1016/j.biomaterials.2020.119903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhang N., Mei K., Guan P., Hu X., Zhao Y. Protein-based artificial nanosystems in cancer therapy. Small. 2020;16 doi: 10.1002/smll.201907256. [DOI] [PubMed] [Google Scholar]
- 5.Xu Z., Zeng S., Gong Z., Yan Y. Exosome-based immunotherapy: a promising approach for cancer treatment. Mol. Cancer. 2020;19:160. doi: 10.1186/s12943-020-01278-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kulp A., Kuehn M.J. Biological functions and biogenesis of secreted bacterial outer membrane vesicles. Annu. Rev. Microbiol. 2010;64:163–184. doi: 10.1146/annurev.micro.091208.073413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Butkovich N., Li E., Ramirez A., Burkhardt A.M., Wang S.W. Advancements in protein nanoparticle vaccine platforms to combat infectious disease. Wiley interdiscipl. rev. Nanomed. nanobiotechnol. 2021;13 doi: 10.1002/wnan.1681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee K.Y., Mooney D.J. Alginate: properties and biomedical applications. Prog. Polym. Sci. 2012;37:106–126. doi: 10.1016/j.progpolymsci.2011.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Smanski M.J., Zhou H., Claesen J., Shen B., Fischbach M.A., Voigt C.A. Synthetic biology to access and expand nature's chemical diversity. Nat. Rev. Microbiol. 2016;14:135–149. doi: 10.1038/nrmicro.2015.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhang R., Billingsley M.M., Mitchell M.J. Biomaterials for vaccine-based cancer immunotherapy. J. Contr. Release : off. J. Control. Release Soc. 2018;292:256–276. doi: 10.1016/j.jconrel.2018.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vaegler M., Lenis A.T., Daum L., Amend B., Stenzl A., Toomey P., et al. Stem cell therapy for voiding and erectile dysfunction. Nat. Rev. Urol. 2012;9:435–447. doi: 10.1038/nrurol.2012.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dever D.P., Bak R.O., Reinisch A., Camarena J., Washington G., Nicolas C.E., et al. CRISPR/Cas9 β-globin gene targeting in human haematopoietic stem cells. Nature. 2016;539:384–389. doi: 10.1038/nature20134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Song X., Liu C., Wang N., Huang H., He S., Gong C., et al. Delivery of CRISPR/Cas systems for cancer gene therapy and immunotherapy. Adv. Drug Deliv. Rev. 2021;168:158–180. doi: 10.1016/j.addr.2020.04.010. [DOI] [PubMed] [Google Scholar]
- 14.Zou Y., Huang B., Cao L., Deng Y., Su J. Tailored mesoporous inorganic biomaterials: assembly, functionalization, and drug delivery engineering. Adv. Mater. (Deerfield Beach, Fla) 2021;33 doi: 10.1002/adma.202005215. [DOI] [PubMed] [Google Scholar]
- 15.Midgley A.C., Wei Y., Li Z., Kong D., Zhao Q. Nitric-oxide-releasing biomaterial regulation of the stem cell microenvironment in regenerative medicine. Adv. Mater. (Deerfield Beach, Fla) 2020;32 doi: 10.1002/adma.201805818. [DOI] [PubMed] [Google Scholar]
- 16.Hu C., Ashok D., Nisbet D.R., Gautam V. Bioinspired surface modification of orthopedic implants for bone tissue engineering. Biomaterials. 2019;219 doi: 10.1016/j.biomaterials.2019.119366. [DOI] [PubMed] [Google Scholar]
- 17.Amin Yavari S., Castenmiller S.M., van Strijp J.A.G., Croes M. Combating implant infections: shifting focus from bacteria to host. Adv. Mater. (Deerfield Beach, Fla) 2020;32 doi: 10.1002/adma.202002962. [DOI] [PubMed] [Google Scholar]
- 18.https://wwwgrandviewresearchcom/industry-analysis/biomaterials-industry
- 19.Riley R.S., June C.H., Langer R., Mitchell M.J. Delivery technologies for cancer immunotherapy. Nat. Rev. Drug Discov. 2019;18:175–196. doi: 10.1038/s41573-018-0006-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lin Y.X., Wang Y., An H.W., Qi B., Wang J., Wang L., et al. Peptide-based autophagic gene and cisplatin Co-delivery systems enable improved chemotherapy resistance. Nano Lett. 2019;19:2968–2978. doi: 10.1021/acs.nanolett.9b00083. [DOI] [PubMed] [Google Scholar]
- 21.Shamay Y., Elkabets M., Li H., Shah J., Brook S., Wang F., et al. P-selectin is a nanotherapeutic delivery target in the tumor microenvironment. Sci. Transl. Med. 2016;8:345ra387. doi: 10.1126/scitranslmed.aaf7374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang Y., Hsu B.Y., Ren C., Li X., Wang J. Silica-based nanocapsules: synthesis, structure control and biomedical applications. Chem. Soc. Rev. 2015;44:315–335. doi: 10.1039/c4cs00199k. [DOI] [PubMed] [Google Scholar]
- 23.Jia Q., Li Z., Guo C., Huang X., Song Y., Zhou N., et al. A gamma-cyclodextrin-based metal-organic framework embedded with graphene quantum dots and modified with PEGMA via SI-ATRP for anticancer drug delivery and therapy. Nanoscale. 2019;11:20956–20967. doi: 10.1039/c9nr06195a. [DOI] [PubMed] [Google Scholar]
- 24.Pitek A.S., Hu H., Shukla S., Steinmetz N.F. Cancer theranostic applications of albumin-coated tobacco mosaic virus nanoparticles. ACS Appl. Mater. Interfaces. 2018;10:39468–39477. doi: 10.1021/acsami.8b12499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liang X., Gao J., Jiang L., Luo J., Jing L., Li X., et al. Nanohybrid liposomal cerasomes with good physiological stability and rapid temperature responsiveness for high intensity focused ultrasound triggered local chemotherapy of cancer. ACS Nano. 2015;9:1280–1293. doi: 10.1021/nn507482w. [DOI] [PubMed] [Google Scholar]
- 26.Ngambenjawong C., Gustafson H.H., Pun S.H. Progress in tumor-associated macrophage (TAM)-targeted therapeutics. Adv. Drug Deliv. Rev. 2017;114:206–221. doi: 10.1016/j.addr.2017.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Roy A., Ernsting M.J., Undzys E., Li S.D. A highly tumor-targeted nanoparticle of podophyllotoxin penetrated tumor core and regressed multidrug resistant tumors. Biomaterials. 2015;52:335–346. doi: 10.1016/j.biomaterials.2015.02.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yu S.S., Lau C.M., Thomas S.N., Jerome W.G., Maron D.J., Dickerson J.H., et al. Size- and charge-dependent non-specific uptake of PEGylated nanoparticles by macrophages. Int. J. Nanomed. 2012;7:799–813. doi: 10.2147/IJN.S28531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Perni S., Prokopovich P. Poly-beta-amino-esters nano-vehicles based drug delivery system for cartilage. Nanomed. Nanotechnol. Biol. Med. 2017;13:539–548. doi: 10.1016/j.nano.2016.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.El-Sawy H.S., Al-Abd A.M., Ahmed T.A., El-Say K.M., Torchilin V.P. Stimuli-responsive nano-architecture drug-delivery systems to solid tumor micromilieu: past, present, and future perspectives. ACS Nano. 2018;12:10636–10664. doi: 10.1021/acsnano.8b06104. [DOI] [PubMed] [Google Scholar]
- 31.Setyawati M.I., Tay C.Y., Chia S.L., Goh S.L., Fang W., Neo M.J., et al. Titanium dioxide nanomaterials cause endothelial cell leakiness by disrupting the homophilic interaction of VE-cadherin. Nat. Commun. 2013;4:1673. doi: 10.1038/ncomms2655. [DOI] [PubMed] [Google Scholar]
- 32.Tee J.K., Yip L.X., Tan E.S., Santitewagun S., Prasath A., Ke P.C., et al. Nanoparticles' interactions with vasculature in diseases. Chem. Soc. Rev. 2019;48:5381–5407. doi: 10.1039/c9cs00309f. [DOI] [PubMed] [Google Scholar]
- 33.Setyawati M.I., Tay C.Y., Bay B.H., Leong D.T. Gold nanoparticles induced endothelial leakiness depends on particle size and endothelial cell origin. ACS Nano. 2017;11:5020–5030. doi: 10.1021/acsnano.7b01744. [DOI] [PubMed] [Google Scholar]
- 34.Peng F., Setyawati M.I., Tee J.K., Ding X., Wang J., Nga M.E., et al. Nanoparticles promote in vivo breast cancer cell intravasation and extravasation by inducing endothelial leakiness. Nat. Nanotechnol. 2019;14:279–286. doi: 10.1038/s41565-018-0356-z. [DOI] [PubMed] [Google Scholar]
- 35.Wang X., Li X., Mao Y., Wang D., Zhao Q., Wang S. Multi-stimuli responsive nanosystem modified by tumor-targeted carbon dots for chemophototherapy synergistic therapy. J. Colloid Interface Sci. 2019;552:639–650. doi: 10.1016/j.jcis.2019.05.085. [DOI] [PubMed] [Google Scholar]
- 36.Kanamala M., Wilson W.R., Yang M., Palmer B.D., Wu Z. Mechanisms and biomaterials in pH-responsive tumour targeted drug delivery: a review. Biomaterials. 2016;85:152–167. doi: 10.1016/j.biomaterials.2016.01.061. [DOI] [PubMed] [Google Scholar]
- 37.Zhao W., Zhao Y., Wang Q., Liu T., Sun J., Zhang R. Remote light-responsive nanocarriers for controlled drug delivery: advances and perspectives. Small. 2019;15 doi: 10.1002/smll.201903060. [DOI] [PubMed] [Google Scholar]
- 38.Zhong D., Wu H., Wu Y., Li Y., Xu X., Yang J., et al. Rational design and facile fabrication of biocompatible triple responsive dendrimeric nanocages for targeted drug delivery. Nanoscale. 2019;11:15091–15103. doi: 10.1039/c9nr04631c. [DOI] [PubMed] [Google Scholar]
- 39.Bazak R., Houri M., El Achy S., Kamel S., Refaat T. Cancer active targeting by nanoparticles: a comprehensive review of literature. J. Cancer Res. Clin. Oncol. 2015;141:769–784. doi: 10.1007/s00432-014-1767-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Liu Z., Yang X., Duan C., Li J., Tong R., Fan Y., et al. Identification and characterization of mammaglobin-A epitope in heterogenous breast cancers for enhancing tumor-targeting therapy. Signal Transduct Target Ther. 2020;5:82. doi: 10.1038/s41392-020-0183-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Alshaer W., Hillaireau H., Fattal E. Aptamer-guided nanomedicines for anticancer drug delivery. Adv. Drug Deliv. Rev. 2018;134:122–137. doi: 10.1016/j.addr.2018.09.011. [DOI] [PubMed] [Google Scholar]
- 42.Wang S.H., Yu J. Structure-based design for binding peptides in anti-cancer therapy. Biomaterials. 2018;156:1–15. doi: 10.1016/j.biomaterials.2017.11.024. [DOI] [PubMed] [Google Scholar]
- 43.Cai L.L., Yang C.Y., Jia W.F., Liu Y.W., Xie R., Lei T., et al. Endo/lysosome-escapable delivery Depot for improving BBB transcytosis and neuron targeted therapy of alzheimer's disease. Adv. Funct. Mater. 2020;30 [Google Scholar]
- 44.Du X., Yin S., Wang Y., Gu X., Wang G., Li J. Hyaluronic acid-functionalized half-generation of sectorial dendrimers for anticancer drug delivery and enhanced biocompatibility. Carbohydr. Polym. 2018;202:513–522. doi: 10.1016/j.carbpol.2018.09.015. [DOI] [PubMed] [Google Scholar]
- 45.Zhang Y., Deng C., Liu S., Wu J., Chen Z., Li C., et al. Active targeting of tumors through conformational epitope imprinting. Angew. Chem. 2015;54:5157–5160. doi: 10.1002/anie.201412114. [DOI] [PubMed] [Google Scholar]
- 46.Hrkach J., Von Hoff D., Mukkaram Ali M., Andrianova E., Auer J., Campbell T., et al. Preclinical development and clinical translation of a PSMA-targeted docetaxel nanoparticle with a differentiated pharmacological profile. Sci. Transl. Med. 2012;4:128ra139. doi: 10.1126/scitranslmed.3003651. [DOI] [PubMed] [Google Scholar]
- 47.Schmid D., Park C.G., Hartl C.A., Subedi N., Cartwright A.N., Puerto R.B., et al. T cell-targeting nanoparticles focus delivery of immunotherapy to improve antitumor immunity. Nat. Commun. 2017;8:1747. doi: 10.1038/s41467-017-01830-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Trombetta E.S., Mellman I. Cell biology of antigen processing in vitro and in vivo. Annu. Rev. Immunol. 2005;23:975–1028. doi: 10.1146/annurev.immunol.22.012703.104538. [DOI] [PubMed] [Google Scholar]
- 49.Irvine D.J., Aung A., Silva M. Controlling timing and location in vaccines. Adv. Drug Deliv. Rev. 2020;158:91–115. doi: 10.1016/j.addr.2020.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bonifaz L., Bonnyay D., Mahnke K., Rivera M., Nussenzweig M.C., Steinman R.M. Efficient targeting of protein antigen to the dendritic cell receptor DEC-205 in the steady state leads to antigen presentation on major histocompatibility complex class I products and peripheral CD8+ T cell tolerance. J. Exp. Med. 2002;196:1627–1638. doi: 10.1084/jem.20021598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Reddy S.T., van der Vlies A.J., Simeoni E., Angeli V., Randolph G.J., O'Neil C.P., et al. Exploiting lymphatic transport and complement activation in nanoparticle vaccines. Nat. Biotechnol. 2007;25:1159–1164. doi: 10.1038/nbt1332. [DOI] [PubMed] [Google Scholar]
- 52.Rodell C.B., Arlauckas S.P., Cuccarese M.F., Garris C.S., Li R., Ahmed M.S., et al. TLR7/8-agonist-loaded nanoparticles promote the polarization of tumour-associated macrophages to enhance cancer immunotherapy. Nat. Biomed. Eng. 2018;2:578–588. doi: 10.1038/s41551-018-0236-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kew O.M., Sutter R.W., de Gourville E.M., Dowdle W.R., Pallansch M.A. Vaccine-derived polioviruses and the endgame strategy for global polio eradication. Annu. Rev. Microbiol. 2005;59:587–635. doi: 10.1146/annurev.micro.58.030603.123625. [DOI] [PubMed] [Google Scholar]
- 54.Fenner F. Risks and benefits of vaccinia vaccine use in the worldwide smallpox eradication campaign. Res. Virol. 1989;140:465–466. doi: 10.1016/s0923-2516(89)80126-8. discussion 487-491. [DOI] [PubMed] [Google Scholar]
- 55.Kennedy R.B., Ovsyannikova I.G., Palese P., Poland G.A. Current challenges in vaccinology. Front. Immunol. 2020;11:1181. doi: 10.3389/fimmu.2020.01181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bookstaver M.L., Tsai S.J., Bromberg J.S., Jewell C.M. Improving vaccine and immunotherapy design using biomaterials. Trends Immunol. 2018;39:135–150. doi: 10.1016/j.it.2017.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chaudhary N., Weissman D., Whitehead K.A. mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nat. Rev. Drug Discov. 2021;20:817–838. doi: 10.1038/s41573-021-00283-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wibowo D., Jorritsma S.H.T., Gonzaga Z.J., Evert B., Chen S., Rehm B.H.A. Polymeric nanoparticle vaccines to combat emerging and pandemic threats. Biomaterials. 2021;268 doi: 10.1016/j.biomaterials.2020.120597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kasturi S.P., Skountzou I., Albrecht R.A., Koutsonanos D., Hua T., Nakaya H.I., et al. Programming the magnitude and persistence of antibody responses with innate immunity. Nature. 2011;470:543–547. doi: 10.1038/nature09737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Thomas C., Rawat A., Hope-Weeks L., Ahsan F. Aerosolized PLA and PLGA nanoparticles enhance humoral, mucosal and cytokine responses to hepatitis B vaccine. Mol. Pharm. 2011;8:405–415. doi: 10.1021/mp100255c. [DOI] [PubMed] [Google Scholar]
- 61.Rahimian S., Fransen M.F., Kleinovink J.W., Christensen J.R., Amidi M., Hennink W.E., et al. Polymeric nanoparticles for co-delivery of synthetic long peptide antigen and poly IC as therapeutic cancer vaccine formulation. J. Contr. Release : off. J. Control. Release Soc. 2015;203:16–22. doi: 10.1016/j.jconrel.2015.02.006. [DOI] [PubMed] [Google Scholar]
- 62.Zhao F., Zhang X., Liu S., Zeng T., Yu J., Gu W., et al. Assessment of the immune responses to Treponema pallidum Gpd DNA vaccine adjuvanted with IL-2 and chitosan nanoparticles before and after Treponema pallidum challenge in rabbits. Sci. China Life Sci. 2013;56:174–180. doi: 10.1007/s11427-012-4434-4. [DOI] [PubMed] [Google Scholar]
- 63.Borges O., Cordeiro-da-Silva A., Tavares J., Santarém N., de Sousa A., Borchard G., et al. Immune response by nasal delivery of hepatitis B surface antigen and codelivery of a CpG ODN in alginate coated chitosan nanoparticles. Eur. J. Pharm. Biopharm. : off. J. Arbeitsgemeinschaft fur Pharmazeutische Verfahrenstechnik eV. 2008;69:405–416. doi: 10.1016/j.ejpb.2008.01.019. [DOI] [PubMed] [Google Scholar]
- 64.Feng G., Jiang Q., Xia M., Lu Y., Qiu W., Zhao D., et al. Enhanced immune response and protective effects of nano-chitosan-based DNA vaccine encoding T cell epitopes of Esat-6 and FL against Mycobacterium tuberculosis infection. PLoS One. 2013;8 doi: 10.1371/journal.pone.0061135. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 65.Eskandari S., Guerin T., Toth I., Stephenson R.J. Recent advances in self-assembled peptides: implications for targeted drug delivery and vaccine engineering. Adv. Drug Deliv. Rev. 2017;110–111:169–187. doi: 10.1016/j.addr.2016.06.013. [DOI] [PubMed] [Google Scholar]
- 66.Gordon D., Kelley P., Heinzel S., Cooper P., Petrovsky N. Immunogenicity and safety of Advax™, a novel polysaccharide adjuvant based on delta inulin, when formulated with hepatitis B surface antigen: a randomized controlled Phase 1 study. Vaccine. 2014;32:6469–6477. doi: 10.1016/j.vaccine.2014.09.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Gordon D.L., Sajkov D., Honda-Okubo Y., Wilks S.H., Aban M., Barr I.G., et al. Human Phase 1 trial of low-dose inactivated seasonal influenza vaccine formulated with Advax™ delta inulin adjuvant. Vaccine. 2016;34:3780–3786. doi: 10.1016/j.vaccine.2016.05.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Green J.J., Langer R., Anderson D.G. A combinatorial polymer library approach yields insight into nonviral gene delivery. Accounts chem. res. 2008;41:749–759. doi: 10.1021/ar7002336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Liu Y., Busscher H.J., Zhao B., Li Y., Zhang Z., van der Mei H.C., et al. Surface-adaptive, antimicrobially loaded, micellar nanocarriers with enhanced penetration and killing efficiency in staphylococcal biofilms. ACS Nano. 2016;10:4779–4789. doi: 10.1021/acsnano.6b01370. [DOI] [PubMed] [Google Scholar]
- 70.Papahadjopoulos D., Nir S., Oki S. Permeability properties of phospholipid membranes: effect of cholesterol and temperature. Biochim. Biophys. Acta. 1972;266:561–583. doi: 10.1016/0006-3002(72)90001-7. [DOI] [PubMed] [Google Scholar]
- 71.Papahadjopoulos D., Cowden M., Kimelberg H. Role of cholesterol in membranes. Effects on phospholipid-protein interactions, membrane permeability and enzymatic activity. Biochim. Biophys. Acta. 1973;330:8–26. doi: 10.1016/0005-2736(73)90280-0. [DOI] [PubMed] [Google Scholar]
- 72.Li M., Du C., Guo N., Teng Y., Meng X., Sun H., et al. Composition design and medical application of liposomes. Eur. J. Med. Chem. 2019;164:640–653. doi: 10.1016/j.ejmech.2019.01.007. [DOI] [PubMed] [Google Scholar]
- 73.Simões S., Filipe A., Faneca H., Mano M., Penacho N., Düzgünes N., et al. Cationic liposomes for gene delivery. Expet Opin. Drug Deliv. 2005;2:237–254. doi: 10.1517/17425247.2.2.237. [DOI] [PubMed] [Google Scholar]
- 74.Butts C., Socinski M.A., Mitchell P.L., Thatcher N., Havel L., Krzakowski M., et al. Tecemotide (L-BLP25) versus placebo after chemoradiotherapy for stage III non-small-cell lung cancer (START): a randomised, double-blind, phase 3 trial. Lancet Oncol. 2014;15:59–68. doi: 10.1016/S1470-2045(13)70510-2. [DOI] [PubMed] [Google Scholar]
- 75.Kranz L.M., Diken M., Haas H., Kreiter S., Loquai C., Reuter K.C., et al. Systemic RNA delivery to dendritic cells exploits antiviral defence for cancer immunotherapy. Nature. 2016;534:396–401. doi: 10.1038/nature18300. [DOI] [PubMed] [Google Scholar]
- 76.Qiao C., Liu J., Yang J., Li Y., Weng J., Shao Y., et al. Enhanced non-inflammasome mediated immune responses by mannosylated zwitterionic-based cationic liposomes for HIV DNA vaccines. Biomaterials. 2016;85:1–17. doi: 10.1016/j.biomaterials.2016.01.054. [DOI] [PubMed] [Google Scholar]
- 77.Mekaru H., Lu J., Tamanoi F. Development of mesoporous silica-based nanoparticles with controlled release capability for cancer therapy. Adv. Drug Deliv. Rev. 2015;95:40–49. doi: 10.1016/j.addr.2015.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.An M., Li M., Xi J., Liu H. Silica nanoparticle as a lymph node targeting platform for vaccine delivery. 2017;9:23466–23475. doi: 10.1021/acsami.7b06024. [DOI] [PubMed] [Google Scholar]
- 79.Wang T., Jiang H., Zhao Q., Wang S., Zou M., Cheng G. Enhanced mucosal and systemic immune responses obtained by porous silica nanoparticles used as an oral vaccine adjuvant: effect of silica architecture on immunological properties. Int. J. Pharm. 2012;436:351–358. doi: 10.1016/j.ijpharm.2012.06.028. [DOI] [PubMed] [Google Scholar]
- 80.Super M., Doherty E.J., Cartwright M.J., Seiler B.T., Langellotto F., Dimitrakakis N., et al. Biomaterial vaccines capturing pathogen-associated molecular patterns protect against bacterial infections and septic shock. Nat. Biomed. Eng. 2021 doi: 10.1038/s41551-021-00756-3. [DOI] [PubMed] [Google Scholar]
- 81.Wang T., Zou M., Jiang H., Ji Z., Gao P., Cheng G. Synthesis of a novel kind of carbon nanoparticle with large mesopores and macropores and its application as an oral vaccine adjuvant. Eur. J. Pharmaceut. Sci.: off. J. Eur. Feder. Pharmaceut. Sci. 2011;44:653–659. doi: 10.1016/j.ejps.2011.10.012. [DOI] [PubMed] [Google Scholar]
- 82.Villa C.H., Dao T., Ahearn I., Fehrenbacher N., Casey E., Rey D.A., et al. Single-walled carbon nanotubes deliver peptide antigen into dendritic cells and enhance IgG responses to tumor-associated antigens. ACS Nano. 2011;5:5300–5311. doi: 10.1021/nn200182x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Niikura K., Matsunaga T., Suzuki T., Kobayashi S., Yamaguchi H., Orba Y., et al. Gold nanoparticles as a vaccine platform: influence of size and shape on immunological responses in vitro and in vivo. ACS Nano. 2013;7:3926–3938. doi: 10.1021/nn3057005. [DOI] [PubMed] [Google Scholar]
- 84.Tao W., Ziemer K.S., Gill H.S. Gold nanoparticle-M2e conjugate coformulated with CpG induces protective immunity against influenza A virus. Nanomedicine. 2014;9:237–251. doi: 10.2217/nnm.13.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Xu L., Liu Y., Chen Z., Li W., Liu Y., Wang L., et al. Surface-engineered gold nanorods: promising DNA vaccine adjuvant for HIV-1 treatment. Nano Lett. 2012;12:2003–2012. doi: 10.1021/nl300027p. [DOI] [PubMed] [Google Scholar]
- 86.Yen H.J., Hsu S.H., Tsai C.L. Cytotoxicity and immunological response of gold and silver nanoparticles of different sizes. Small. 2009;5:1553–1561. doi: 10.1002/smll.200900126. [DOI] [PubMed] [Google Scholar]
- 87.Zanganeh S., Hutter G., Spitler R., Lenkov O., Mahmoudi M., Shaw A., et al. Iron oxide nanoparticles inhibit tumour growth by inducing pro-inflammatory macrophage polarization in tumour tissues. Nat. Nanotechnol. 2016;11:986–994. doi: 10.1038/nnano.2016.168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ha N.Y., Shin H.M., Sharma P., Cho H.A., Min C.K., Kim H.I., et al. Generation of protective immunity against Orientia tsutsugamushi infection by immunization with a zinc oxide nanoparticle combined with ScaA antigen. J. Nanobiotechnol. 2016;14:76. doi: 10.1186/s12951-016-0229-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Sun B., Ji Z., Liao Y.P., Wang M., Wang X., Dong J., et al. Engineering an effective immune adjuvant by designed control of shape and crystallinity of aluminum oxyhydroxide nanoparticles. ACS Nano. 2013;7:10834–10849. doi: 10.1021/nn404211j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Temchura V.V., Kozlova D., Sokolova V., Uberla K., Epple M. Targeting and activation of antigen-specific B-cells by calcium phosphate nanoparticles loaded with protein antigen. Biomaterials. 2014;35:6098–6105. doi: 10.1016/j.biomaterials.2014.04.010. [DOI] [PubMed] [Google Scholar]
- 91.Morrison C. Landmark green light for Mosquirix malaria vaccine. Nat. Biotechnol. 2015;33:1015–1016. doi: 10.1038/nbt1015-1015. [DOI] [PubMed] [Google Scholar]
- 92.Efficacy and safety of RTS,S/AS01 malaria vaccine with or without a booster dose in infants and children in Africa: final results of a phase 3, individually randomised, controlled trial. Lancet. 2015;386:31–45. doi: 10.1016/S0140-6736(15)60721-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Ward B.J., Gobeil P., Séguin A., Atkins J., Boulay I., Charbonneau P.Y., et al. Phase 1 randomized trial of a plant-derived virus-like particle vaccine for COVID-19. Nat. Med. 2021;27:1071–1078. doi: 10.1038/s41591-021-01370-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Kaparakis-Liaskos M., Ferrero R.L. Immune modulation by bacterial outer membrane vesicles. Nat. Rev. Immunol. 2015;15:375–387. doi: 10.1038/nri3837. [DOI] [PubMed] [Google Scholar]
- 95.Chatterjee S.N., Das J. Electron microscopic observations on the excretion of cell-wall material by Vibrio cholerae. J. Gen. Microbiol. 1967;49:1–11. doi: 10.1099/00221287-49-1-1. [DOI] [PubMed] [Google Scholar]
- 96.Devoe I.W., Gilchrist J.E. Release of endotoxin in the form of cell wall blebs during in vitro growth of Neisseria meningitidis. J. Exp. Med. 1973;138:1156–1167. doi: 10.1084/jem.138.5.1156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Arigita C., Jiskoot W., Westdijk J., van Ingen C., Hennink W.E., Crommelin D.J., et al. Stability of mono- and trivalent meningococcal outer membrane vesicle vaccines. Vaccine. 2004;22:629–642. doi: 10.1016/j.vaccine.2003.08.027. [DOI] [PubMed] [Google Scholar]
- 98.Baker J.L., Chen L., Rosenthal J.A., Putnam D., DeLisa M.P. Microbial biosynthesis of designer outer membrane vesicles. Curr. Opin. Biotechnol. 2014;29:76–84. doi: 10.1016/j.copbio.2014.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Ellis T.N., Kuehn M.J. Virulence and immunomodulatory roles of bacterial outer membrane vesicles. Microbiol. Mol. Biol. Rev. : MMBR (Microbiol. Mol. Biol. Rev.) 2010;74:81–94. doi: 10.1128/MMBR.00031-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.O'Ryan M., Stoddard J., Toneatto D., Wassil J., Dull P.M. A multi-component meningococcal serogroup B vaccine (4CMenB): the clinical development program. Drugs. 2014;74:15–30. doi: 10.1007/s40265-013-0155-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Gerritzen M.J.H., Martens D.E., Wijffels R.H., van der Pol L., Stork M. Bioengineering bacterial outer membrane vesicles as vaccine platform. Biotechnol. Adv. 2017;35:565–574. doi: 10.1016/j.biotechadv.2017.05.003. [DOI] [PubMed] [Google Scholar]
- 102.Morein B., Sundquist B., Höglund S., Dalsgaard K., Osterhaus A. Iscom, a novel structure for antigenic presentation of membrane proteins from enveloped viruses. Nature. 1984;308:457–460. doi: 10.1038/308457a0. [DOI] [PubMed] [Google Scholar]
- 103.Pabreja S., Garg T., Rath G., Goyal A.K. Mucosal vaccination against tuberculosis using Ag85A-loaded immunostimulating complexes. Artificial cells, nanomed. biotechnol. 2016;44:532–539. doi: 10.3109/21691401.2014.966195. [DOI] [PubMed] [Google Scholar]
- 104.Pearse M.J., Drane D. ISCOMATRIX adjuvant for antigen delivery. Adv. Drug Deliv. Rev. 2005;57:465–474. doi: 10.1016/j.addr.2004.09.006. [DOI] [PubMed] [Google Scholar]
- 105.Brito L.A., Malyala P., O'Hagan D.T. Vaccine adjuvant formulations: a pharmaceutical perspective. Semin. Immunol. 2013;25:130–145. doi: 10.1016/j.smim.2013.05.007. [DOI] [PubMed] [Google Scholar]
- 106.Baz Morelli A., Becher D., Koernig S., Silva A., Drane D., Maraskovsky E. ISCOMATRIX: a novel adjuvant for use in prophylactic and therapeutic vaccines against infectious diseases. J. Med. Microbiol. 2012;61:935–943. doi: 10.1099/jmm.0.040857-0. [DOI] [PubMed] [Google Scholar]
- 107.Nicholaou T., Ebert L.M., Davis I.D., McArthur G.A., Jackson H., Dimopoulos N., et al. Regulatory T-cell-mediated attenuation of T-cell responses to the NY-ESO-1 ISCOMATRIX vaccine in patients with advanced malignant melanoma. Clin. Cancer Res. : Off. J. Am. Ass. Cancer Res. 2009;15:2166–2173. doi: 10.1158/1078-0432.CCR-08-2484. [DOI] [PubMed] [Google Scholar]
- 108.Yang J., Wang W., Chen Z., Lu S., Yang F., Bi Z., et al. A vaccine targeting the RBD of the S protein of SARS-CoV-2 induces protective immunity. Nature. 2020;586:572–577. doi: 10.1038/s41586-020-2599-8. [DOI] [PubMed] [Google Scholar]
- 109.Ku M.W., Bourgine M., Authié P., Lopez J., Nemirov K., Moncoq F., et al. Intranasal vaccination with a lentiviral vector protects against SARS-CoV-2 in preclinical animal models. Cell Host Microbe. 2021;29:236–249. doi: 10.1016/j.chom.2020.12.010. e236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Vaccines–COVID19 Vaccine Tracker Home Page https://covid19.trackvaccines.org/vaccines/ Available online.
- 111.Erasmus J.H., Khandhar A.P., O'Connor M.A., Walls A.C., Hemann E.A., Murapa P., et al. An Alphavirus-derived replicon RNA vaccine induces SARS-CoV-2 neutralizing antibody and T cell responses in mice and nonhuman primates. Sci. Transl. Med. 2020;12 doi: 10.1126/scitranslmed.abc9396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Pfizer-BioNTech COVID-19 vaccine. https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/pfizer-biontech-covid-19-vaccine Available online:
- 113.Hassett K.J., Benenato K.E., Jacquinet E., Lee A., Woods A., Yuzhakov O., et al. Optimization of lipid nanoparticles for intramuscular administration of mRNA vaccines. Mol. Ther. Nucleic Acids. 2019;15:1–11. doi: 10.1016/j.omtn.2019.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Moderna COVID vaccine becomes second to get US authorization. https://www.nature.com/articles/d41586-020-03593-7 Available online:
- 115.Ward D., McCormack S. Clinical trial to assess the safety of a coronavirus vaccine in healthy men and women (ISRCTN17072692) BioMedCentral. 2020 [Google Scholar]
- 116.Coleman C.M., Liu Y.V., Mu H., Taylor J.K., Massare M., Flyer D.C., et al. Purified coronavirus spike protein nanoparticles induce coronavirus neutralizing antibodies in mice. Vaccine. 2014;32:3169–3174. doi: 10.1016/j.vaccine.2014.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Clinical trials for covid-19. EU Clinical Trials Register. 2020 https://www.clinicaltrialsregister.eu/ Available online: [Google Scholar]
- 118.Richards Grayson A.C., Choi I.S., Tyler B.M., Wang P.P., Brem H., Cima M.J., et al. Multi-pulse drug delivery from a resorbable polymeric microchip device. Nat. Mater. 2003;2:767–772. doi: 10.1038/nmat998. [DOI] [PubMed] [Google Scholar]
- 119.McHugh K.J., Nguyen T.D., Linehan A.R., Yang D., Behrens A.M., Rose S., et al. Fabrication of fillable microparticles and other complex 3D microstructures. Science. 2017;357:1138–1142. doi: 10.1126/science.aaf7447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Kulkarni J.A., Witzigmann D., Thomson S.B., Chen S., Leavitt B.R., Cullis P.R., et al. The current landscape of nucleic acid therapeutics. Nat. Nanotechnol. 2021;16:630–643. doi: 10.1038/s41565-021-00898-0. [DOI] [PubMed] [Google Scholar]
- 121.Zeng M., Xu Q., Zhou D., A S., Alshehri F., Lara-Saez I., et al. Highly branched poly(beta-amino ester)s for gene delivery in hereditary skin diseases. Adv. Drug Deliv. Rev. 2021;176 doi: 10.1016/j.addr.2021.113842. [DOI] [PubMed] [Google Scholar]
- 122.Buck J., Grossen P., Cullis P.R., Huwyler J., Witzigmann D. Lipid-based DNA therapeutics: hallmarks of non-viral gene delivery. ACS Nano. 2019;13:3754–3782. doi: 10.1021/acsnano.8b07858. [DOI] [PubMed] [Google Scholar]
- 123.Yin H., Kanasty R.L., Eltoukhy A.A., Vegas A.J., Dorkin J.R., Anderson D.G. Non-viral vectors for gene-based therapy. Nat. Rev. Genet. 2014;15:541–555. doi: 10.1038/nrg3763. [DOI] [PubMed] [Google Scholar]
- 124.Rajala A., Wang Y., Zhu Y., Ranjo-Bishop M., Ma J.X., Mao C., et al. Nanoparticle-assisted targeted delivery of eye-specific genes to eyes significantly improves the vision of blind mice in vivo. Nano Lett. 2014;14:5257–5263. doi: 10.1021/nl502275s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Zhou Z., Liu X., Zhu D., Wang Y., Zhang Z., Zhou X., et al. Nonviral cancer gene therapy: delivery cascade and vector nanoproperty integration. Adv. Drug Deliv. Rev. 2017;115:115–154. doi: 10.1016/j.addr.2017.07.021. [DOI] [PubMed] [Google Scholar]
- 126.Zeng M., Alshehri F., Zhou D., Lara-Sáez I., Wang X., Li X., et al. Efficient and robust highly branched poly(β-amino ester)/minicircle COL7A1 polymeric nanoparticles for gene delivery to recessive dystrophic epidermolysis bullosa keratinocytes. ACS Appl. Mater. Interfaces. 2019;11:30661–30672. doi: 10.1021/acsami.9b13135. [DOI] [PubMed] [Google Scholar]
- 127.Lv H., Zhang S., Wang B., Cui S., Yan J. Toxicity of cationic lipids and cationic polymers in gene delivery. J. Contr. Release : off. J. Control. Release Soc. 2006;114:100–109. doi: 10.1016/j.jconrel.2006.04.014. [DOI] [PubMed] [Google Scholar]
- 128.Zhou D., Cutlar L., Gao Y., Wang W., O'Keeffe-Ahern J., McMahon S., et al. The transition from linear to highly branched poly(β-amino ester)s: branching matters for gene delivery. Sci. Adv. 2016;2 doi: 10.1126/sciadv.1600102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Zeng M., Zhou D., Alshehri F., Lara-Sáez I., Lyu Y., Creagh-Flynn J., et al. Manipulation of transgene expression in fibroblast cells by a multifunctional linear-branched hybrid poly(β-amino ester) synthesized through an oligomer combination approach. Nano Lett. 2019;19:381–391. doi: 10.1021/acs.nanolett.8b04098. [DOI] [PubMed] [Google Scholar]
- 130.Tockary T.A., Foo W., Dirisala A., Chen Q., Uchida S., Osawa S., et al. Single-stranded DNA-packaged polyplex micelle as adeno-associated-virus-inspired compact vector to systemically target stroma-rich pancreatic cancer. ACS Nano. 2019;13:12732–12742. doi: 10.1021/acsnano.9b04676. [DOI] [PubMed] [Google Scholar]
- 131.Su X., Fricke J., Kavanagh D.G., Irvine D.J. In vitro and in vivo mRNA delivery using lipid-enveloped pH-responsive polymer nanoparticles. Mol. Pharm. 2011;8:774–787. doi: 10.1021/mp100390w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Phua K.K., Leong K.W., Nair S.K. Transfection efficiency and transgene expression kinetics of mRNA delivered in naked and nanoparticle format. J. Contr. Release : off. J. Control. Release Soc. 2013;166:227–233. doi: 10.1016/j.jconrel.2012.12.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Patel A.K., Kaczmarek J.C., Bose S., Kauffman K.J., Mir F., Heartlein M.W., et al. Inhaled nanoformulated mRNA polyplexes for protein production in lung epithelium. Adv. Mater. 2019;31 doi: 10.1002/adma.201805116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Hoy S.M. Patisiran: first global approval. Drugs. 2018;78:1625–1631. doi: 10.1007/s40265-018-0983-6. [DOI] [PubMed] [Google Scholar]
- 135.Hajj K.A., Whitehead K.A. Tools for translation: non-viral materials for therapeutic mRNA delivery. Nat. Rev. Mater. 2017;2:17056. [Google Scholar]
- 136.Dong Y., Siegwart D.J., Anderson D.G. Strategies, design, and chemistry in siRNA delivery systems. Adv. Drug Deliv. Rev. 2019;144:133–147. doi: 10.1016/j.addr.2019.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Fernandez-Piñeiro I., Badiola I., Sanchez A. Nanocarriers for microRNA delivery in cancer medicine. Biotechnol. Adv. 2017;35:350–360. doi: 10.1016/j.biotechadv.2017.03.002. [DOI] [PubMed] [Google Scholar]
- 138.Li R.Q., Wu Y., Zhi Y., Yang X., Li Y., Xua F.J., et al. PGMA-based star-like polycations with plentiful hydroxyl groups act as highly efficient miRNA delivery nanovectors for effective applications in heart diseases. Adv. Mater. (Deerfield Beach, Fla) 2016;28:7204–7212. doi: 10.1002/adma.201602319. [DOI] [PubMed] [Google Scholar]
- 139.Nguyen M.A., Wyatt H., Susser L., Geoffrion M., Rasheed A., Duchez A.C., et al. Delivery of MicroRNAs by chitosan nanoparticles to functionally alter macrophage cholesterol efflux in vitro and in vivo. ACS Nano. 2019;13:6491–6505. doi: 10.1021/acsnano.8b09679. [DOI] [PubMed] [Google Scholar]
- 140.Chan C., Guo N., Duan X., Han W., Xue L., Bryan D., et al. Systemic miRNA delivery by nontoxic nanoscale coordination polymers limits epithelial-to-mesenchymal transition and suppresses liver metastases of colorectal cancer. Biomaterials. 2019;210:94–104. doi: 10.1016/j.biomaterials.2019.04.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Di Ianni T., Bose R.J.C., Sukumar U.K., Bachawal S., Wang H., Telichko A., et al. Ultrasound/microbubble-mediated targeted delivery of anticancer microRNA-loaded nanoparticles to deep tissues in pigs. J. Contr. Release : off. J. Control. Release Soc. 2019;309:1–10. doi: 10.1016/j.jconrel.2019.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Bejerano T., Etzion S., Elyagon S., Etzion Y., Cohen S. Nanoparticle delivery of miRNA-21 mimic to cardiac macrophages improves myocardial remodeling after myocardial infarction. Nano Lett. 2018;18:5885–5891. doi: 10.1021/acs.nanolett.8b02578. [DOI] [PubMed] [Google Scholar]
- 143.Varshney A., Panda J.J., Singh A.K., Yadav N., Bihari C., Biswas S., et al. Targeted delivery of microRNA-199a-3p using self-assembled dipeptide nanoparticles efficiently reduces hepatocellular carcinoma in mice. Hepatology. 2018;67:1392–1407. doi: 10.1002/hep.29643. [DOI] [PubMed] [Google Scholar]
- 144.Farooqi A.A., Desai N.N., Qureshi M.Z., Librelotto D.R.N., Gasparri M.L., Bishayee A., et al. Exosome biogenesis, bioactivities and functions as new delivery systems of natural compounds. Biotechnol. Adv. 2018;36:328–334. doi: 10.1016/j.biotechadv.2017.12.010. [DOI] [PubMed] [Google Scholar]
- 145.Li Z., Zhou X., Wei M., Gao X., Zhao L., Shi R., et al. In vitro and in vivo RNA inhibition by CD9-HuR functionalized exosomes encapsulated with miRNA or CRISPR/dCas9. Nano Lett. 2019;19:19–28. doi: 10.1021/acs.nanolett.8b02689. [DOI] [PubMed] [Google Scholar]
- 146.Kim H.J., Yi S.W., Oh H.J., Lee J.S., Park J.S., Park K.H. Transfection of gene regulation nanoparticles complexed with pDNA and shRNA controls multilineage differentiation of hMSCs. Biomaterials. 2018;177:1–13. doi: 10.1016/j.biomaterials.2018.05.035. [DOI] [PubMed] [Google Scholar]
- 147.Wang Y., Xie Y., Kilchrist K.V., Li J., Duvall C.L., Oupický D. Endosomolytic and tumor-penetrating mesoporous silica nanoparticles for siRNA/miRNA combination cancer therapy. ACS Appl. Mater. Interfaces. 2020;12:4308–4322. doi: 10.1021/acsami.9b21214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Yu Q., Zhang M., Chen Y., Chen X., Shi S., Sun K., et al. Self-assembled nanoparticles prepared from low-molecular-weight PEI and low-generation PAMAM for EGFRvIII-chimeric antigen receptor gene loading and T-cell transient modification. Int. J. Nanomed. 2020;15:483–495. doi: 10.2147/IJN.S229858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Billingsley M.M., Singh N., Ravikumar P., Zhang R., June C.H., Mitchell M.J. Ionizable lipid nanoparticle-mediated mRNA delivery for human CAR T cell engineering. Nano Lett. 2020;20:1578–1589. doi: 10.1021/acs.nanolett.9b04246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Kim K.S., Han J.H., Park J.H., Kim H.K., Choi S.H., Kim G.R., et al. Multifunctional nanoparticles for genetic engineering and bioimaging of natural killer (NK) cell therapeutics. Biomaterials. 2019;221 doi: 10.1016/j.biomaterials.2019.119418. [DOI] [PubMed] [Google Scholar]
- 151.Parayath N.N., Stephan M.T. In situ programming of CAR T cells. Annu. Rev. Biomed. Eng. 2021;23:385–405. doi: 10.1146/annurev-bioeng-070620-033348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Smith T.T., Stephan S.B., Moffett H.F., McKnight L.E., Ji W., Reiman D., et al. In situ programming of leukaemia-specific T cells using synthetic DNA nanocarriers. Nat. Nanotechnol. 2017;12:813–820. doi: 10.1038/nnano.2017.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Parayath N.N., Stephan S.B., Koehne A.L., Nelson P.S., Stephan M.T. In vitro-transcribed antigen receptor mRNA nanocarriers for transient expression in circulating T cells in vivo. Nat. Commun. 2020;11:6080. doi: 10.1038/s41467-020-19486-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Wang H.X., Li M., Lee C.M., Chakraborty S., Kim H.W., Bao G., et al. CRISPR/Cas9-Based genome editing for disease modeling and therapy: challenges and opportunities for nonviral delivery. Chem. Rev. 2017;117:9874–9906. doi: 10.1021/acs.chemrev.6b00799. [DOI] [PubMed] [Google Scholar]
- 155.Li L., Hu S., Chen X. Non-viral delivery systems for CRISPR/Cas9-based genome editing: challenges and opportunities. Biomaterials. 2018;171:207–218. doi: 10.1016/j.biomaterials.2018.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Kretzmann J.A., Ho D., Evans C.W., Plani-Lam J.H.C., Garcia-Bloj B., Mohamed A.E., et al. Synthetically controlling dendrimer flexibility improves delivery of large plasmid DNA. Chem. Sci. 2017;8:2923–2930. doi: 10.1039/c7sc00097a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Li L., Song L., Liu X., Yang X., Li X., He T., et al. Artificial virus delivers CRISPR-cas9 system for genome editing of cells in mice. ACS Nano. 2017;11:95–111. doi: 10.1021/acsnano.6b04261. [DOI] [PubMed] [Google Scholar]
- 158.Timin A.S., Muslimov A.R., Lepik K.V., Epifanovskaya O.S., Shakirova A.I., Mock U., et al. Efficient gene editing via non-viral delivery of CRISPR-Cas9 system using polymeric and hybrid microcarriers. Nanomed. Nanotechnol. Biol. Med. 2018;14:97–108. doi: 10.1016/j.nano.2017.09.001. [DOI] [PubMed] [Google Scholar]
- 159.Wang P., Zhang L., Zheng W., Cong L., Guo Z., Xie Y., et al. Thermo-triggered release of CRISPR-cas9 system by lipid-encapsulated gold nanoparticles for tumor therapy. Angew. Chem. 2018;57:1491–1496. doi: 10.1002/anie.201708689. [DOI] [PubMed] [Google Scholar]
- 160.Zhang Y., Shen S., Zhao G., Xu C.F., Zhang H.B., Luo Y.L., et al. In situ repurposing of dendritic cells with CRISPR/Cas9-based nanomedicine to induce transplant tolerance. Biomaterials. 2019;217 doi: 10.1016/j.biomaterials.2019.119302. [DOI] [PubMed] [Google Scholar]
- 161.Jiang C., Mei M., Li B., Zhu X., Zu W., Tian Y., et al. A non-viral CRISPR/Cas9 delivery system for therapeutically targeting HBV DNA and pcsk9 in vivo. Cell Res. 2017;27:440–443. doi: 10.1038/cr.2017.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Miller J.B., Zhang S., Kos P., Xiong H., Zhou K., Perelman S.S., et al. Non-viral CRISPR/Cas gene editing in vitro and in vivo enabled by synthetic nanoparticle Co-delivery of Cas9 mRNA and sgRNA. Angew. Chem. 2017;56:1059–1063. doi: 10.1002/anie.201610209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Yin H., Song C.Q., Dorkin J.R., Zhu L.J., Li Y., Wu Q., et al. Therapeutic genome editing by combined viral and non-viral delivery of CRISPR system components in vivo. Nat. Biotechnol. 2016;34:328–333. doi: 10.1038/nbt.3471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Ramakrishna S., Kwaku Dad A.B., Beloor J., Gopalappa R., Lee S.K., Kim H. Gene disruption by cell-penetrating peptide-mediated delivery of Cas9 protein and guide RNA. Genome Res. 2014;24:1020–1027. doi: 10.1101/gr.171264.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Zuris J.A., Thompson D.B., Shu Y., Guilinger J.P., Bessen J.L., Hu J.H., et al. Cationic lipid-mediated delivery of proteins enables efficient protein-based genome editing in vitro and in vivo. Nat. Biotechnol. 2015;33:73–80. doi: 10.1038/nbt.3081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Gao X., Tao Y., Lamas V., Huang M., Yeh W.H., Pan B., et al. Treatment of autosomal dominant hearing loss by in vivo delivery of genome editing agents. Nature. 2018;553:217–221. doi: 10.1038/nature25164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Sun W., Ji W., Hall J.M., Hu Q., Wang C., Beisel C.L., et al. Self-assembled DNA nanoclews for the efficient delivery of CRISPR-Cas9 for genome editing. Angew. Chem. 2015;54:12029–12033. doi: 10.1002/anie.201506030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Alsaiari S.K., Patil S., Alyami M., Alamoudi K.O., Aleisa F.A., Merzaban J.S., et al. Endosomal escape and delivery of CRISPR/Cas9 genome editing machinery enabled by nanoscale zeolitic imidazolate framework. J. Am. Chem. Soc. 2018;140:143–146. doi: 10.1021/jacs.7b11754. [DOI] [PubMed] [Google Scholar]
- 169.Mout R., Ray M., Yesilbag Tonga G., Lee Y.-W., Tay T., Sasaki K., et al. Direct cytosolic delivery of CRISPR/Cas9-Ribonucleoprotein for efficient gene editing. ACS Nano. 2017;11:2452–2458. doi: 10.1021/acsnano.6b07600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Liu C., Wan T., Wang H., Zhang S., Ping Y., Cheng Y. A boronic acid-rich dendrimer with robust and unprecedented efficiency for cytosolic protein delivery and CRISPR-Cas9 gene editing. Sci. Adv. 2019;5 doi: 10.1126/sciadv.aaw8922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Tabatabaei S.N., Derbali R.M., Yang C., Superstein R., Hamel P., Chain J.L., et al. Co-delivery of miR-181a and melphalan by lipid nanoparticles for treatment of seeded retinoblastoma. J. Contr. Release : off. J. Control. Release Soc. 2019;298:177–185. doi: 10.1016/j.jconrel.2019.02.014. [DOI] [PubMed] [Google Scholar]
- 172.Allemani C., Matsuda T., Di Carlo V., Harewood R., Matz M., Nikšić M., et al. Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): analysis of individual records for 37513025patients diagnosed with one of 18 cancers from 322 population -basedregistries in 71 countries. Lancet. 2018;391:1023–1075. doi: 10.1016/S0140-6736(17)33326-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Liu Z., Tang M., Zhao J., Chai R., Kang J. Looking into the future: toward advanced 3D biomaterials for stem-cell-based regenerative medicine. Adv. Mater. (Deerfield Beach, Fla) 2018;30 doi: 10.1002/adma.201705388. [DOI] [PubMed] [Google Scholar]
- 174.Tam R.Y., Yockell-Lelièvre J., Smith L.J., Julian L.M., Baker A.E.G., Choey C., et al. Rationally designed 3D hydrogels model invasive lung diseases enabling high-content drug screening. Adv. Mater. (Deerfield Beach, Fla) 2019;31 doi: 10.1002/adma.201806214. [DOI] [PubMed] [Google Scholar]
- 175.Shadish J.A., Benuska G.M., DeForest C.A. Bioactive site-specifically modified proteins for 4D patterning of gel biomaterials. Nat. Mater. 2019;18:1005–1014. doi: 10.1038/s41563-019-0367-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Caiazzo M., Okawa Y., Ranga A., Piersigilli A., Tabata Y., Lutolf M.P. Defined three-dimensional microenvironments boost induction of pluripotency. Nat. Mater. 2016;15:344–352. doi: 10.1038/nmat4536. [DOI] [PubMed] [Google Scholar]
- 177.Chaudhuri O., Gu L., Klumpers D., Darnell M., Bencherif S.A., Weaver J.C., et al. Hydrogels with tunable stress relaxation regulate stem cell fate and activity. Nat. Mater. 2016;15:326–334. doi: 10.1038/nmat4489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Huebsch N., Lippens E., Lee K., Mehta M., Koshy S.T., Darnell M.C., et al. Matrix elasticity of void-forming hydrogels controls transplanted-stem-cell-mediated bone formation. Nat. Mater. 2015;14:1269–1277. doi: 10.1038/nmat4407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Das R.K., Gocheva V., Hammink R., Zouani O.F., Rowan A.E. Stress-stiffening-mediated stem-cell commitment switch in soft responsive hydrogels. Nat. Mater. 2016;15:318–325. doi: 10.1038/nmat4483. [DOI] [PubMed] [Google Scholar]
- 180.Madl C.M., LeSavage B.L., Dewi R.E., Dinh C.B., Stowers R.S., Khariton M., et al. Maintenance of neural progenitor cell stemness in 3D hydrogels requires matrix remodelling. Nat. Mater. 2017;16:1233–1242. doi: 10.1038/nmat5020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Hörner M., Raute K., Hummel B., Madl J., Creusen G., Thomas O.S., et al. Phytochrome-based extracellular matrix with reversibly tunable mechanical properties. Adv. Mater. (Deerfield Beach, Fla) 2019;31 doi: 10.1002/adma.201806727. [DOI] [PubMed] [Google Scholar]
- 182.Jia X., Minami K., Uto K., Chang A.C., Hill J.P., Nakanishi J., et al. Adaptive liquid interfacially assembled protein nanosheets for guiding mesenchymal stem cell fate. Adv. Mater. (Deerfield Beach, Fla) 2020;32 doi: 10.1002/adma.201905942. [DOI] [PubMed] [Google Scholar]
- 183.Bhatia S.N., Ingber D.E. Microfluidic organs-on-chips. Nat. Biotechnol. 2014;32:760–772. doi: 10.1038/nbt.2989. [DOI] [PubMed] [Google Scholar]
- 184.Sato T., Vries R.G., Snippert H.J., van de Wetering M., Barker N., Stange D.E., et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009;459:262–265. doi: 10.1038/nature07935. [DOI] [PubMed] [Google Scholar]
- 185.Liu H., Wang Y., Cui K., Guo Y., Zhang X., Qin J. Advances in hydrogels in organoids and organs-on-a-chip. Adv. Mater. (Deerfield Beach, Fla) 2019;31 doi: 10.1002/adma.201902042. [DOI] [PubMed] [Google Scholar]
- 186.Zhao Y., Rafatian N., Feric N.T., Cox B.J., Aschar-Sobbi R., Wang E.Y., et al. A platform for generation of chamber-specific cardiac tissues and disease modeling. Cell. 2019;176:913–927. doi: 10.1016/j.cell.2018.11.042. e918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Garreta E., Prado P., Tarantino C., Oria R., Fanlo L., Martí E., et al. Fine tuning the extracellular environment accelerates the derivation of kidney organoids from human pluripotent stem cells. Nat. Mater. 2019;18:397–405. doi: 10.1038/s41563-019-0287-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Cruz N.M., Song X., Czerniecki S.M., Gulieva R.E., Churchill A.J., Kim Y.K., et al. Organoid cystogenesis reveals a critical role of microenvironment in human polycystic kidney disease. Nat. Mater. 2017;16:1112–1119. doi: 10.1038/nmat4994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Shao Y., Taniguchi K., Gurdziel K., Townshend R.F., Xue X., Yong K.M.A., et al. Self-organized amniogenesis by human pluripotent stem cells in a biomimetic implantation-like niche. Nat. Mater. 2017;16:419–425. doi: 10.1038/nmat4829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Nawroth J.C., Scudder L.L., Halvorson R.T., Tresback J., Ferrier J.P., Sheehy S.P., et al. Automated fabrication of photopatterned gelatin hydrogels for organ-on-chips applications. Biofabrication. 2018;10 doi: 10.1088/1758-5090/aa96de. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Xie R., Zheng W., Guan L., Ai Y., Liang Q. Engineering of hydrogel materials with perfusable microchannels for building vascularized tissues. Small. 2020;16 doi: 10.1002/smll.201902838. [DOI] [PubMed] [Google Scholar]
- 192.Zhang Y.S., Arneri A., Bersini S., Shin S.R., Zhu K., Goli-Malekabadi Z., et al. Bioprinting 3D microfibrous scaffolds for engineering endothelialized myocardium and heart-on-a-chip. Biomaterials. 2016;110:45–59. doi: 10.1016/j.biomaterials.2016.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Pelkonen A., Mzezewa R., Sukki L., Ryynänen T., Kreutzer J., Hyvärinen T., et al. A modular brain-on-a-chip for modelling epileptic seizures with functionally connected human neuronal networks. Biosens. Bioelectron. 2020;168 doi: 10.1016/j.bios.2020.112553. [DOI] [PubMed] [Google Scholar]
- 194.Christoffersson J., Aronsson C., Jury M., Selegård R., Aili D., Mandenius C.F. Fabrication of modular hyaluronan-PEG hydrogels to support 3D cultures of hepatocytes in a perfused liver-on-a-chip device. Biofabrication. 2018;11 doi: 10.1088/1758-5090/aaf657. [DOI] [PubMed] [Google Scholar]
- 195.Yang L., Conley B.M., Cerqueira S.R., Pongkulapa T., Wang S., Lee J.K., et al. Effective modulation of CNS inhibitory microenvironment using bioinspired hybrid-nanoscaffold-based therapeutic interventions. Adv. Mater. (Deerfield Beach, Fla) 2020 doi: 10.1002/adma.202002578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Facklam A.L., Volpatti L.R., Anderson D.G. Biomaterials for personalized cell therapy. Adv. Mater. (Deerfield Beach, Fla) 2020;32 doi: 10.1002/adma.201902005. [DOI] [PubMed] [Google Scholar]
- 197.Roche E.T., Hastings C.L., Lewin S.A., Shvartsman D., Brudno Y., Vasilyev N.V., et al. Comparison of biomaterial delivery vehicles for improving acute retention of stem cells in the infarcted heart. Biomaterials. 2014;35:6850–6858. doi: 10.1016/j.biomaterials.2014.04.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Gao L., Kupfer M.E., Jung J.P., Yang L., Zhang P., Da Sie Y., et al. Myocardial tissue engineering with cells derived from human-induced pluripotent stem cells and a native-like, high-resolution, 3-dimensionally printed scaffold. Circ. Res. 2017;120:1318–1325. doi: 10.1161/CIRCRESAHA.116.310277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Gao L., Gregorich Z.R., Zhu W., Mattapally S., Oduk Y., Lou X., et al. Large cardiac muscle patches engineered from human induced-pluripotent stem cell-derived cardiac cells improve recovery from myocardial infarction in swine. Circulation. 2018;137:1712–1730. doi: 10.1161/CIRCULATIONAHA.117.030785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Sharma R., Khristov V., Rising A., Jha B.S., Dejene R., Hotaling N., et al. Clinical-grade stem cell-derived retinal pigment epithelium patch rescues retinal degeneration in rodents and pigs. Sci. Transl. Med. 2019;11 doi: 10.1126/scitranslmed.aat5580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.da Cruz L., Fynes K., Georgiadis O., Kerby J., Luo Y.H., Ahmado A., et al. Phase 1 clinical study of an embryonic stem cell-derived retinal pigment epithelium patch in age-related macular degeneration. Nat. Biotechnol. 2018;36:328–337. doi: 10.1038/nbt.4114. [DOI] [PubMed] [Google Scholar]
- 202.Cai L., Dewi R.E., Heilshorn S.C. Injectable hydrogels with in situ double network formation enhance retention of transplanted stem cells. Adv. Funct. Mater. 2015;25:1344–1351. doi: 10.1002/adfm.201403631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Loebel C., Rodell C.B., Chen M.H., Burdick J.A. Shear-thinning and self-healing hydrogels as injectable therapeutics and for 3D-printing. Nat. Protoc. 2017;12:1521–1541. doi: 10.1038/nprot.2017.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Kanda P., Alarcon E.I., Yeuchyk T., Parent S., de Kemp R.A., Variola F., et al. Deterministic encapsulation of human cardiac stem cells in variable composition nanoporous gel cocoons to enhance therapeutic repair of injured myocardium. ACS Nano. 2018;12:4338–4350. doi: 10.1021/acsnano.7b08881. [DOI] [PubMed] [Google Scholar]
- 205.Tang J., Cui X., Caranasos T.G., Hensley M.T., Vandergriff A.C., Hartanto Y., et al. Heart repair using nanogel-encapsulated human cardiac stem cells in mice and pigs with myocardial infarction. ACS Nano. 2017;11:9738–9749. doi: 10.1021/acsnano.7b01008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Lee S., Yun S., Park K.I., Jang J.H. Sliding fibers: slidable, injectable, and gel-like electrospun nanofibers as versatile cell carriers. ACS Nano. 2016;10:3282–3294. doi: 10.1021/acsnano.5b06605. [DOI] [PubMed] [Google Scholar]
- 207.Yadid M., Feiner R., Dvir T. Gold nanoparticle-integrated scaffolds for tissue engineering and regenerative medicine. Nano Lett. 2019;19:2198–2206. doi: 10.1021/acs.nanolett.9b00472. [DOI] [PubMed] [Google Scholar]
- 208.Cho H., Blatchley M.R., Duh E.J., Gerecht S. Acellular and cellular approaches to improve diabetic wound healing. Adv. Drug Deliv. Rev. 2019;146:267–288. doi: 10.1016/j.addr.2018.07.019. [DOI] [PubMed] [Google Scholar]
- 209.Kleiner L.W., Wright J.C., Wang Y. Evolution of implantable and insertable drug delivery systems. J. Contr. Release : off. J. Control. Release Soc. 2014;181:1–10. doi: 10.1016/j.jconrel.2014.02.006. [DOI] [PubMed] [Google Scholar]
- 210.Zhu M., Li K., Zhu Y., Zhang J., Ye X. 3D-printed hierarchical scaffold for localized isoniazid/rifampin drug delivery and osteoarticular tuberculosis therapy. Acta Biomater. 2015;16:145–155. doi: 10.1016/j.actbio.2015.01.034. [DOI] [PubMed] [Google Scholar]
- 211.Clark A., Milbrandt T.A., Hilt J.Z., Puleo D.A. Mechanical properties and dual drug delivery application of poly(lactic-co-glycolic acid) scaffolds fabricated with a poly(β-amino ester) porogen. Acta Biomater. 2014;10:2125–2132. doi: 10.1016/j.actbio.2013.12.061. [DOI] [PubMed] [Google Scholar]
- 212.Stephan S.B., Taber A.M., Jileaeva I., Pegues E.P., Sentman C.L., Stephan M.T. Biopolymer implants enhance the efficacy of adoptive T-cell therapy. Nat. Biotechnol. 2015;33:97–101. doi: 10.1038/nbt.3104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Verma V., Kim Y., Lee M.C., Lee J.T., Cho S., Park I.K., et al. Activated dendritic cells delivered in tissue compatible biomatrices induce in-situ anti-tumor CTL responses leading to tumor regression. Oncotarget. 2016;7:39894–39906. doi: 10.18632/oncotarget.9529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Phuengkham H., Song C., Um S.H., Lim Y.T. Implantable synthetic immune niche for spatiotemporal modulation of tumor-derived immunosuppression and systemic antitumor immunity: postoperative immunotherapy. Adv. Mater. (Deerfield Beach, Fla) 2018;30 doi: 10.1002/adma.201706719. [DOI] [PubMed] [Google Scholar]
- 215.Welch N.G., Winkler D.A., Thissen H. Antifibrotic strategies for medical devices. Adv. Drug Deliv. Rev. 2020;167:109–120. doi: 10.1016/j.addr.2020.06.008. [DOI] [PubMed] [Google Scholar]
- 216.Smith T.T., Moffett H.F., Stephan S.B., Opel C.F., Dumigan A.G., Jiang X., et al. Biopolymers codelivering engineered T cells and STING agonists can eliminate heterogeneous tumors. J. Clin. Invest. 2017;127:2176–2191. doi: 10.1172/JCI87624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Han X., Li H., Zhou D., Chen Z., Gu Z. Local and targeted delivery of immune checkpoint blockade therapeutics. Accounts chem. res. 2020;53:2521–2533. doi: 10.1021/acs.accounts.0c00339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Li Y., Fang M., Zhang J., Wang J., Song Y., Shi J., et al. Hydrogel dual delivered celecoxib and anti-PD-1 synergistically improve antitumor immunity. OncoImmunology. 2016;5 doi: 10.1080/2162402X.2015.1074374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Monette A., Ceccaldi C., Assaad E., Lerouge S., Lapointe R. Chitosan thermogels for local expansion and delivery of tumor-specific T lymphocytes towards enhanced cancer immunotherapies. Biomaterials. 2016;75:237–249. doi: 10.1016/j.biomaterials.2015.10.021. [DOI] [PubMed] [Google Scholar]
- 220.Sheets K.T., Ewend M.G., Mohiti-Asli M., Tuin S.A., Loboa E.G., Aboody K.S., et al. Developing implantable scaffolds to enhance neural stem cell therapy for post-operative glioblastoma. Mol. Ther. : J. Am. Soc. Gene Ther. 2020;28:1056–1067. doi: 10.1016/j.ymthe.2020.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Bencherif S.A., Warren Sands R., Ali O.A., Li W.A., Lewin S.A., Braschler T.M., et al. Injectable cryogel-based whole-cell cancer vaccines. Nat. Commun. 2015;6:7556. doi: 10.1038/ncomms8556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Verbeke C.S., Gordo S., Schubert D.A., Lewin S.A., Desai R.M., Dobbins J., et al. Multicomponent injectable hydrogels for antigen-specific tolerogenic immune modulation. Adv. healthcare mater. 2017;6 doi: 10.1002/adhm.201600773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Li A.W., Sobral M.C., Badrinath S., Choi Y., Graveline A., Stafford A.G., et al. A facile approach to enhance antigen response for personalized cancer vaccination. Nat. Mater. 2018;17:528–534. doi: 10.1038/s41563-018-0028-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Vila-Parrondo C., García-Astrain C., Liz-Marzán L.M. Colloidal systems toward 3D cell culture scaffolds. Adv. Colloid Interface Sci. 2020;283 doi: 10.1016/j.cis.2020.102237. [DOI] [PubMed] [Google Scholar]
- 225.Barenholz Y. Doxil®--the first FDA-approved nano-drug: lessons learned. J. Contr. Release : off. J. Control. Release Soc. 2012;160:117–134. doi: 10.1016/j.jconrel.2012.03.020. [DOI] [PubMed] [Google Scholar]
- 226.Soloman R., Gabizon A.A. Clinical pharmacology of liposomal anthracyclines: focus on pegylated liposomal Doxorubicin. Clinical lymphoma & myeloma. 2008;8:21–32. doi: 10.3816/clm.2008.n.001. [DOI] [PubMed] [Google Scholar]
- 227.De Vincenzo R., Conte C., Ricci C., Scambia G., Capelli G. Long-term efficacy and safety of human papillomavirus vaccination. Int. J. Wom. Health. 2014;6:999–1010. doi: 10.2147/IJWH.S50365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Schiller J.T., Castellsagué X., Garland S.M. A review of clinical trials of human papillomavirus prophylactic vaccines. Vaccine. 2012;30(Suppl 5):F123–F138. doi: 10.1016/j.vaccine.2012.04.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Eliasof S., Lazarus D., Peters C.G., Case R.I., Cole R.O., Hwang J., et al. Correlating preclinical animal studies and human clinical trials of a multifunctional, polymeric nanoparticle. Proc. Natl. Acad. Sci. U.S.A. 2013;110:15127–15132. doi: 10.1073/pnas.1309566110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Tao L., Reese T.A. Making mouse models that reflect human immune responses. Trends Immunol. 2017;38:181–193. doi: 10.1016/j.it.2016.12.007. [DOI] [PubMed] [Google Scholar]
- 231.Mestas J., Hughes C.C. Of mice and not men: differences between mouse and human immunology. J. Immunol. 2004;172:2731–2738. doi: 10.4049/jimmunol.172.5.2731. [DOI] [PubMed] [Google Scholar]
- 232.Jiang W., Wang Y., Wargo J.A., Lang F.F., Kim B.Y.S. Considerations for designing preclinical cancer immune nanomedicine studies. Nat. Nanotechnol. 2021;16:6–15. doi: 10.1038/s41565-020-00817-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Hua S., de Matos M.B.C., Metselaar J.M., Storm G. Current trends and challenges in the clinical translation of nanoparticulate nanomedicines: pathways for translational development and commercialization. Front. Pharmacol. 2018;9:790. doi: 10.3389/fphar.2018.00790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Castro F., Pinto M.L., Pereira C.L., Serre K., Barbosa M.A., Vermaelen K., et al. Chitosan/γ-PGA nanoparticles-based immunotherapy as adjuvant to radiotherapy in breast cancer. Biomaterials. 2020;257 doi: 10.1016/j.biomaterials.2020.120218. [DOI] [PubMed] [Google Scholar]
- 235.Feng R., Yu F., Xu J., Hu X. Knowledge gaps in immune response and immunotherapy involving nanomaterials: databases and artificial intelligence for material design. Biomaterials. 2021;266 doi: 10.1016/j.biomaterials.2020.120469. [DOI] [PubMed] [Google Scholar]
- 236.Upadhya R., Kosuri S., Tamasi M., Meyer T.A., Atta S., Webb M.A., et al. Automation and data-driven design of polymer therapeutics. Adv. Drug Deliv. Rev. 2021;171:1–28. doi: 10.1016/j.addr.2020.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Ouyang B., Poon W., Zhang Y.N., Lin Z.P., Kingston B.R., Tavares A.J., et al. The dose threshold for nanoparticle tumour delivery. Nat. Mater. 2020;19:1362–1371. doi: 10.1038/s41563-020-0755-z. [DOI] [PubMed] [Google Scholar]