Abstract
Background
Cancer remains a serious health concern worldwide, and different approaches are being developed for its treatment. The strategy to use the immune system as an approach for treating cancer has recently gained momentum. Messenger RNA (mRNA) has been assessed as an up-and-coming resource for the evolution of advanced cancer immunotherapies over the past decades. However, degradation in extracellular compartments and during endosomal escape remain obstacles for efficient mRNA delivery and limit the therapeutic applications of this approach.
Area covered
Lipid-based nanocarriers are gaining significant attention as non-viral mRNA vectors. Various lipid-based nanocarrier types have been developed to enhance the stability of mRNA molecules, facilitate their transfection, and ensure delivery to an intracellular compartment suitable for further processing. This review discusses the development of novel mRNA delivery systems using lipids for effective cancer immunotherapy.
Expert opinion
mRNAs are superior to other biomolecules for developing therapeutic drugs and vaccines with multiple medical applications that are currently being explored by researchers in various biomedical fields. Lipid-based mRNA nanoparticles can improve the potency of the mRNA by enhancing its stability, enabling its cellular uptake, and facilitating its endosomal escape. Targetability of these therapeutics can be increased by conjugating their surface with the desired ligands or targeting agents. Lipid–mRNA nanoparticles are increasingly being incorporated in cancer immunotherapy applications, including vaccines, monoclonal antibodies, and chimeric antigen receptor T-cell treatment, and several such nanoparticles are being assessed in clinical trials. Further research that assesses key variables for transfection efficiency of lipid–mRNA nanoparticles will expedite the development of improved therapeutics.
Keywords: mRNA, Lipids, Lipid nanoparticle, Cancer immunotherapy
Introduction
Cancer, one of the prime sources of death worldwide, comprises a large group of diseases, which are triggered by the uncontrolled proliferation of malignant cells. Surgery and radiation therapy are the primary treatment methods for local and non-metastatic cancers, whereas chemotherapy is used to suppress cancer cell proliferation and treat metastatic cancers. Depending on the cancer type and stage, the treatment is typically based on an appropriate combination of these methods, but the overall survival rate and therapeutic efficacy of these conventional approaches are still unsatisfactory. Thus, development of effective cancer treatment methods is of an utmost requirement. Recently, remarkable progress has been made in cancer treatment by deploying various combination therapies, including chemotherapy, photothermal therapy, photodynamic therapy, and hyperthermia therapy (Ou et al. 2018; Poudel et al. 2020).
Immunotherapy, which utilizes the capability of the body’s defense mechanism to concede and attack cancer cells, has gained substantial interest over the past decade for its applicability in cancer treatment. Two German physicians, Wilhelm Busch and Friedrich Fehleisen, recognized an epidemiological association of cancer and the immune system in the nineteenth century (Oiseth et al. 2017). Later, the American orthopedic surgeon William Coley, the father of cancer immunotherapy, medicated cancer patients with extracts containing heat-inactivated Streptococcus pyogenes and Serratia marcescens to induce sepsis and boost the immunity (Decker et al. 2017). This extract cocktail derived from gram-positive and gram-negative bacteria, which is widely known as ‘Coley’s toxins,’ had potent immunostimulatory properties and attained favorable responses in several types of malignancies. However, ‘Coley’s toxins’ did not become the standard practice in cancer therapy due to the lack of scientific rigor and the inability to obtain reproducible results. With the development of radiation therapy and chemotherapy, ‘Coley’s toxins’ usage was gradually discontinued (Waldman et al. 2020). However, with the discovery of immune checkpoints, such as cytotoxic T lymphocyte antigen 4 (CTLA-4) and programmed cell death 1 (PD-1), cancer immunotherapy research has seen a boost in the preceding decades and there has been an improved understanding of the process of immune surveillance by which innate immune cells eliminate cancer cells (Darvin et al. 2018; Esfahani et al. 2020). After the approval of six checkpoint blockade modulators (Hargadon et al. 2018) and two chimeric antigen receptor (CAR)-T cell immunotherapies (Pantin et al. 2020) by the US Food and Drug Administration (FDA), cancer immunotherapy is gaining tremendous prominence as a standard method for cancer treatment.
Although immunotherapy is evolving as an effective approach for the treatment of cancer, there are still some considerable obstacles. So far majority of immunotherapies have been utilized mostly for patients with advanced cancer, but response rates for diseases in less advanced cases have yet to be thoroughly determined. As the immune system response varies from person to person based on their genetic makeup, a hundred percent response rate is not guaranteed. Progress towards broader and more effective immunotherapeutic approaches requires deeper insights into the immunological relationships between tumors and hosts (Hiam-Galvez et al. 2021). Cancer vaccines which target antigens specific to tumor or associated with tumor, can precisely attack and eradicate malignant cells that overexpress these antigens, which is a safe and specific treatment option compared to other immunotherapy techniques. Despite these promising features, the clinical application of viral vector based, immune cell-based or peptide based vaccines has remained challenging for decades because of cumbersome preparation methods and modest clinical responses in patients. In this context, non-infectious nucleic acid (DNA and RNA) vaccines which are free of contaminations (protein or virus-derived) are emerging as promising vaccine platforms for cancer therapy. However, DNA needs to be translocated across the cell wall and nuclear membrane and needs to undergo transcription within the nucleus to transfect a cell. This process, along with a weak expression of the DNA-sensing mechanism and varying expression of nucleic acid-sensing pattern recognition receptors, contributes to the comparatively low immunogenicity of DNA vaccines. Unlike DNA, messenger RNA (mRNA) can efficiently transfect both mitotic and non-mitotic cells because it is directly translated into the antigen(s) of interest in the cytoplasm without requiring translocation into the nucleus (Huertas et al. 2003; Jahanafrooz et al. 2020). Typically, mRNA vaccines are linked with a higher protein expression and magnitude than DNA vaccines, and do not require genomic integration; mRNA is thus free from impending insertional mutagenesis (Sahin et al. 2014). The mRNA is prepared in a cell-free system, which facilitates the process of scaling up the production and implementing ‘Good Manufacturing Practices’ by optimizing the conditions. (Guevara et al. 2020; Sahin et al. 2014). mRNA molecules are demonstrably superior to other biomolecules, such as proteins, for developing new therapeutic and prophylactic vaccines, monoclonal antibodies, CAR T-cell therapies, and immunomodulatory drugs (Billingsley et al. 2020; Van Hoecke et al. 2019).
Though mRNA is an emerging platform for cancer therapy, it has disadvantage of being degraded by ubiquitous RNase and being unstable (Zhou et al. 2020). As a negatively charged and large molecule type (104–106 Da), it is challenging to translocate mRNA molecules across the anionic lipid bilayer of cell membranes. However, efficient mRNA delivery into cells is vital for the success of mRNA-based therapeutics. Several physical methods, including gene guns, microinjection, electroporation, hydrodynamic delivery, and microfluidic squeezing, have been applied for mRNA delivery (Hajj et al. 2017). Various nanomaterials, including polymers, protein derivatives, lipids and lipidoids have been designed for in vivo mRNA delivery. Other strategies, such as encapsulation of mRNA in viral vectors, have also been explored (Wadhwa et al. 2020). Non-viral lipid-based nano system exhibit promising properties to mediate mRNA transfection, such as protecting the mRNA from degradation and increasing its cellular internalization (Lee et al. 2020; Zhang et al. 2021a, b). With the recent approval of the lipid nanoparticle (LNP) mRNA vaccine for COVID-19, researchers have sought to harness this method for modulating cancer vaccines. Lipid based systems have been rigorously studied for mRNA cancer immunotherapy and are currently in clinical trials. This review presents a brief sketch of advancements in mRNA delivery by lipid nanocarriers for effective cancer immunotherapy.
Structure of mRNA
mRNA, a single-stranded RNA molecule, directs cytoplasmic protein expression. The mRNA types investigated for their use in cancer are non-replicating unmodified mRNA, modified mRNA and virus-derived self-amplifying RNA (Reichmuth et al. 2016). Traditional mRNA-based vaccines encode the antigen of interest with 5′ and 3′ untranslated regions (UTRs), while self-amplifying RNAs encode the viral replication machinery allowing intracellular RNA amplification and increased protein expression (Pardi et al. 2018). In vitro transcription (IVT) is used as a simple and quick method to synthesize the desired mRNA molecules in a cell-free system (Karikó 2019). IVT mRNA is synthesized from a linear DNA structure which uses bacteriophage T7, T3, or SP6 RNA polymerase. Preferentially, the product should have open reading frame that encodes the protein of interest, flanking UTRs, a 5′ cap, and a poly(A) tail (Pardi et al. 2018; Tossberg et al. 2020). Unprotected mRNA is subjected to rapid degradation by extracellular RNases and are not efficiently internalized. Hence, various transfection reagents have been advanced which enables cellular uptake of mRNA and thus provide protection against its degradation. (Wadhwa et al. 2020).
Lipid-based mRNA nanoparticle systems
Lipid-based vectors prepared from naturally derived or synthetic lipids are promising non-viral RNA delivery systems. These nanocarrier lipids consist of a polar head, a hydrophobic tail and a linker connecting the two regions. LNPs, which increase the mRNA stability and transfection efficiency, are broadly classified based on their charge and composition as cationic LNPs, ionizable LNPs, lipid–polymer hybrid nanoparticles (LPHNPs), and lipid calcium phosphate (LCP) nanoparticles.
Lipoplexes
Lipoplexes are formed through electrostatic interaction within positively charged liposomes and nucleic acids with negative charge (Fig. 1). The electrostatic interactions occur between the polar headgroup of cationic liposomes and mRNA phosphate groups. Lipoplexes were the earliest lipid-based delivery systems that carried mRNA molecules with distinct internal arrangements, which protected the mRNAs from degradation by cellular nucleases (Ewert et al. 2010). Studies have revealed that lipoplexes are released from endosomes via a flip-flop mechanism. An increase in transfection efficiency was achieved using lipids, such as 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine (DOPE), to facilitate the fusion of the lipoplex with the endosomal membrane and release of the mRNA (Simões et al. 1999). Overall, the formulation of mRNA lipoplexes can achieve enhanced immunogenicity, duration of protein expression, hemocompatibility, and storage stability. The positive charge of the formulation can increase the immune response after intravenous (i.v.) administration (Samaridou et al. 2020). Lipoplexes have limited applicability due to potency and tolerability issues. They have been subjected to extensive preclinical in vitro experiments; however, due to poor customization of their composition, relatively low transfection efficiency, tendency to aggregate, and rapid removal from blood circulation, they have been recently excluded from clinical studies (Guevara et al. 2020).
Fig. 1.
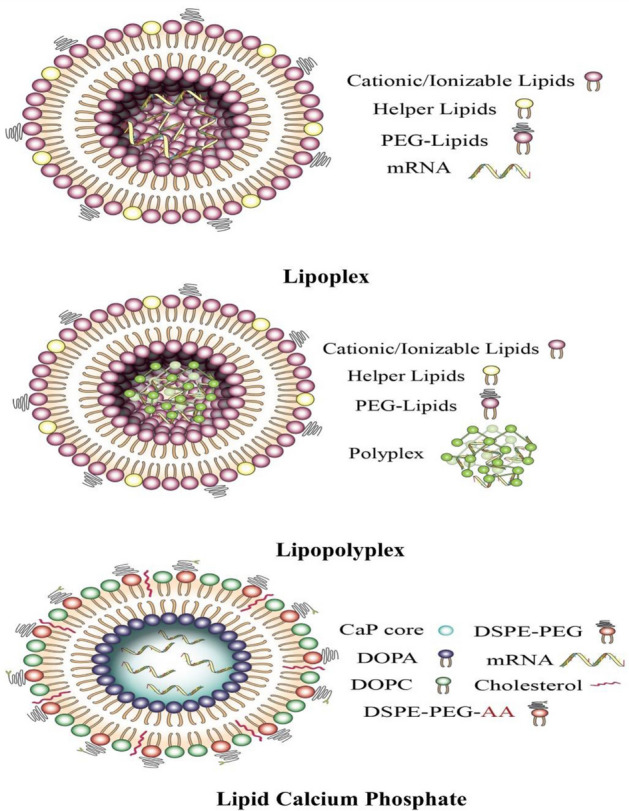
Lipid-based nanoparticles used for delivery of mRNA in cancer therapy
Cationic lipids
Cationic lipids feature a positively charged hydrophilic head that is joined to the hydrophobic chain by a linker. Electrostatic interaction of cationic lipids with the negatively charged phosphate groups of mRNA molecules can form complexes that are essential for mRNA in vitro transfection. The cationic lipid 1,2-di-O-octadecenyl-3-trimethylammonium-propane (DOTMA) and lipid DOPE were the first lipids used for mRNA delivery with relatively high transfection efficiency (Malone et al. 1989). DOTMA and its biodegradable analog 1,2-dioleoyl-3-trimethyl ammonium-propane (DOTAP) are employed in cancer immunotherapy. Spleen-targeted DOTMA–mRNA lipoplexes (RNA-LPX) were used to develop a systemic cancer vaccine (Kranz et al. 2016). Cheng et al. (2020) demonstrated that modifying the lipid-to-mRNA ratio can result in a change from cationic to anionic lipoplexes and this affects the in vivo delivery of mRNA. Anionic lipoplexes target the spleen, whereas cationic formulations target the lungs. Thus, the target organ can be changed by modifying the proportion of the cationic LNPs (Cheng et al. 2020). The combination of DOTAP with DOPE is more efficient in generating antigen-specific lipids, which appear to be associated with enhanced encapsulation and transfection efficiency. DOTAP, in combination with cholesterol, is even more efficient in transfecting primary bone marrow-derived dendritic cells (DCs; Liu et al. 2020). Recently, ethylphosphatidylcholine (ePC)-based LNPs have been investigated for use in mRNA-based cancer immunotherapies (Persano et al. 2017; Zhang et al. 2019).
Cationic lipid-based nanoparticles can trigger innate immune responses and act as immune adjuvants for mRNA vaccine (Lou et al. 2020). Especially the properties of the amine head group and the degree of acyl chain saturation of cationic liposomes can considerably affect their immunostimulatory activity (Lonez et al. 2012). Hence, nanoparticle delivery platforms containing lipids with permanent cationic charge can be designed to obtain mRNA-based immunotherapies with improved therapeutic efficacy and enhanced immunogenicity. Despite these beneficial properties, the disadvantages of cationic LNPs include a less efficient endosomal escape than other similar nanoplatforms and the induction of toxic pro-apoptotic and pro-inflammatory responses (Cui et al. 2018), which can induce liver damage and significantly increase leukocyte counts (Morille et al. 2008).
Ionizable lipoplexes
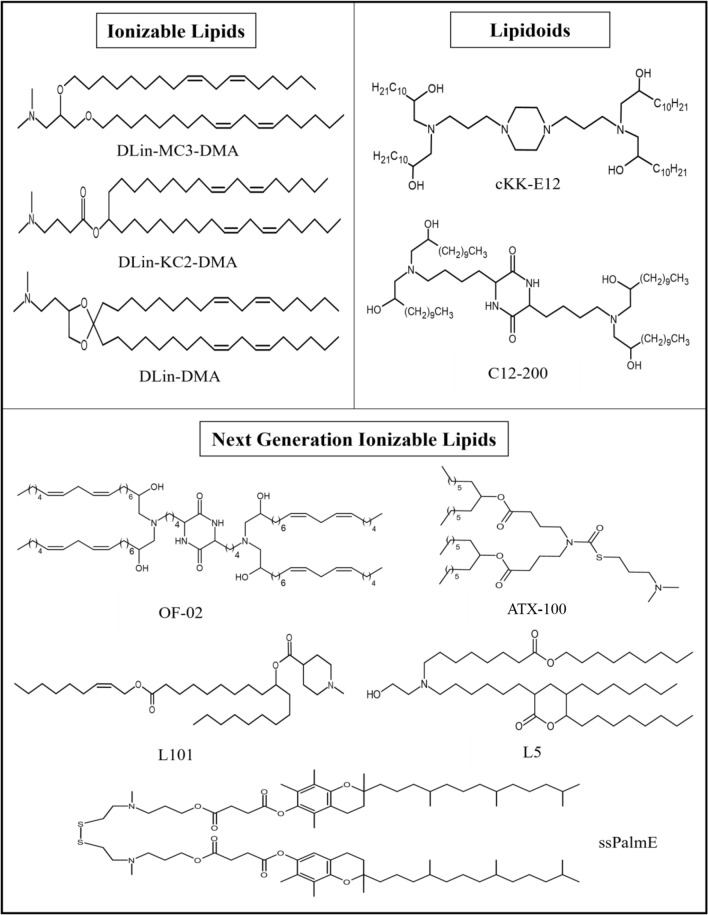
At physiological pH, ionizable lipids remain neutral, but at low pH, they become positively charged due to the protonation of free amines. Ionizable lipids were developed to overcome the limitations of cationic lipids and were initially used for small interfering RNA (siRNA) and DNA delivery (Guevara et al. 2020). The positive charge of ionizable lipids at low pH facilitates the complex formation or encapsulation of negatively charged mRNA, which enhances the therapeutic efficacy by improving endosomal escape and mRNA release. However, the ionizable lipids regain the neutral charge at physiological pH, which extends their circulation time, enhances stability, and decreases toxicity (Chaudhary et al. 2021; Samaridou et al. 2020). Moreover, the neutral charge of these lipids limits their interactions with the blood cell anionic membranes and thus improve their biocompatibility as a beneficial property for in vivo mRNA delivery. Once they enter the endosomes where the pH is lower compared to the extracellular environment, the ionizable lipids become protonated and acquire a positive charge, which may lead to membrane destabilization and assist in endosomal escape (Hou et al. 2021). The structures of some of the important next ionizable lipids are presented in Fig. 1. Next generation ionizable lipids such as the ssPalmE, L5, L101 and ATX-100 have been utilized for LNP synthesis for various application in RNA delivery. ssPalmE is an ionizable lipid-like material with vitamin E-scaffolds. ssPalmE in combination with cationic peptide “KALA” has been used as a lipoplex-mRNA nanoparticles as a DC-vaccine which stimulated higher protein expression and the production of proinflammatory cytokines from murine bone marrow derived DCs (BMDCs) and suggested to be the potent ex vivo DCs-based vaccine platform (Tateshita et al. 2019). L5, an amino lipid, has been shown to have efficient mRNA delivery in rodent and primate models with optimal pharmacokinetics and non-toxic side effects (Sabnis et al. 2018). L101 is a biodegradable lipid used to formulate effective LNP formulations. Biodegradable LNPs were prepared encapsulating an siRNA targeting proprotein convertase subtilisin/kexin type 9 (PCSK9) that demonstrated higher hepatic clearance and thus displayed improved toxicity profile (Suzuki et al. 2017). When new series of alkenyl amino alcohol (AAA) ionizable lipid materials for mRNA LNP delivery were developed, linoleic acid derivative OF-02 formulated with DOPE, C14-PEG-2000, and unmodified mRNA coding for human erythropoietin (EPO) into mRNA LNPs expressed higher levels of EPO expression. This formulation outperformed the benchmark lipid cKK-E12 LNPs yielding twofold increase in EPO production in vivo which represents tunable handle for in vivo protein expression significant in protein replacement therapies (Fenton et al. 2016).
The preparation of lipoplexes consisting of ionizable lipids and mRNA is carried out in an acidic buffer with the result that the lipids become positively charged and encapsulate the RNA cargo. The acid dissociation constant (pKa) of the ionizable group has a significant effect on the in vivo efficacy and immunogenicity of mRNA. Ideal immunogenicity is achieved with the following pKa ranges: i.v. mRNA delivery, pKa 6.2–6.5; intramuscular (i.m.) mRNA delivery, pKa 6.6–6.9 (Cullis and Hope 2017). 1,2-Dioleoyl-3-dimethylammonium propane (DODAP) and 1,2-dioleyloxy-N,N-dimethyl-3-aminopropane (DODMA) were the first ionizable lipids to be explored for mRNA delivery (Semple et al. 2001; Heyes et al. 2005). Various structural modifications of ionizable lipids have been used to enhance their mRNA delivery potency. For instance, enhanced degree of unsaturation in the hydrophobic domain of ionizable lipids increases mRNA delivery efficiency (Fenton et al. 2016). Specifically, addition of double bonds can enhance the fusogenic group formation, which promotes the fusion process and endosomal delivery (Heyes et al. 2005). Improving the efficacy of DODMA by rational design resulted in the development of the ionizable lipid 1,2-dilinoleyloxy-N,N-dimethyl-3-aminopropane (DLin-DMA) (Heyes et al. 2005). Further modification of the amine head group, linker, and hydrophobic regions of DLin-DMA led to the DLin-KC2-DMA lipid, which was further modified to obtain (6Z,9Z,28Z,31Z)-heptatriaconta-6,9,28,31-tetraen-19-yl4-(dimethylamino)butanoate (DLin-MC3-DMA) (Jayaraman et al. 2012) (Fig. 2). The ionizable lipid DLin-MC3-DMA (MC3) was utilized in the formulation of patisiran (Onpattro), the first FDA-approved siRNA drug (Akinc et al. 2019). For mRNA delivery, MC3 lipid is also explored (Hou et al. 2021; Chaudhary et al. 2021).
Fig. 2.
Various groups of ionizable lipids used for delivery of mRNA
A large library of ionizable lipids, which are termed lipid-like compounds or lipidoids, can be efficiently synthesized by combinatorial chemistry. High-throughput screening of a variety of lipidoids prepared by assembling diverse building blocks can rapidly identify ionizable lipids with better RNA delivery efficiency (Zhang et al. 2021a, b). For example, the lipid-like compound 1,1′-((2-(4-(2-((2-(bis(2-hydroxydodecyl)amino)ethyl)(2-hydroxydodecyl)amino) ethyl)piperazin-1-yl)ethyl)azanediyl)bis(dodecan-2-ol) (C12-200) has been developed for the delivery of mRNA molecules (Love et al. 2010). Lipidoid 3,6-bis(4-(bis(2-hydroxydodecyl)amino)butyl)piperazine-2,5-dione (cKK-E12) is incorporated into LNPs to deliver mRNA molecules for cancer immunotherapy (Oberli et al. 2017). The mRNA delivery efficacy of cKK-E12 can be improved by replacing its lipid chains with alkenyl amino alcohols, leading to 3,6-bis(4-(bis((9Z,12Z)-2-hydroxyoctadeca-9,12-dien-1-yl)amino)butyl)piperazine -2,5-dione (OF-02) (Fenton et al. 2016).
The tolerability of LNPs can be improved for frequent drug administration by using biodegradable ionizable lipids for the formulation. Enhance lipid biodegradability is achieved by introducing ester or disulfide motifs into the linker and lipid tails. Specifically, introducing an ester group into the MC3 lipid improves its mRNA delivery efficacy and accelerates its clearance from the liver and plasma. Compared with the properties of MC3, the biodegradable lipids ((4-hydroxybutyl)azanediyl)bis(hexane-6,1-diyl)bis(2-hexyldecanoate) (ALC-0315) and heptadecan-9-yl 8-((2-hydroxyethyl)(6-oxo-6-(undecyloxy)hexyl)amino)octanoate (Lipid H or SM-102) displayed improved pharmacokinetics and enhanced transfection efficiency and were used in the formulation of the COVID-19 vaccines BNT162b and mRNA-1273, respectively (Hou et al. 2021). Moreover, pH-switchable zwitterionic ionizable lipids, which can possess therapeutic properties, have been explored for mRNA delivery. LNP-mRNA formulations comprising zwitterionic ionizable lipids bearing a heterocyclic amine head group (A18-Iso5-2DC18) can target T cells in vivo. A18-Iso5-2DC18 strongly binds to the stimulator of interferon genes (STING) and induces potent cytolytic T lymphocyte responses, resulting in substantial antitumor immunity (Miao et al. 2019). Many ionizable lipids have been developed and screened to optimize antigen expression, elicit adaptive immune responses, and enhance RNA delivery.
Helper lipids
Along with cationic or ionizable lipids, helper lipids, such as cholesterol, PEGylated lipids, and phospholipids (phosphatidylcholine and phosphatidylethanolamine) are commonly included in mRNA-LNP formulations. These supplementary components with distinct functional properties are added to enhance the stability, cellular uptake, and mRNA delivery by LNPs. The inclusion of DOPE as helper lipid for cKK-E12 LNPs improved mRNA expression. Miao et al. (2020) used LNPs containing combinations of different ionizable lipids to evaluate mRNA expression and indicated that the combination of a protein-binding ionizable lipid with a lipid of high fusogenicity could enhance mRNA expression (Miao et al. 2020). The incorporation of cholesterol and 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC) improves lipid stability and endosomal escape. Inclusion of lipid-anchored polyethylene glycol (PEG) decreases macrophage-mediated clearance (Aldosari et al. 2021).
Lipopolyplexes
Lipopolyplexes consist of mRNA complexed with a polymer core and surrounded by a lipid shell (Fig. 1). This system was designed to utilize the advantages of lipoplexes and polymers to generate LNPs with better colloidal stability, decreased cytotoxicity, and enhanced transfection efficiency. Lipid and polymer can exert synergistic effects that promote cellular uptake and hence facilitate endosomal escape of the payload (Chen et al. 2016). Several polymers and lipids have been investigated for the formulation of stable lipopolyplexes. Polyethylenimine has excellent efficacy and is extensively used for nucleic acid delivery. However, its application is limited by its high charge density, which causes toxicity (Ulkoski et al. 2019). Several biodegradable polymers, such as polycaprolactone, PLGA and polylactic acid, along with their combinations are used for complexation with mRNA. DSPE, DOTAP, lecithin, 1,2-distearoyl-sn-glycero-3-phosphocholine, 1,2-dilauroyl-sn-glycero-3-phosphocholine, and PEG are the most commonly applied lipids (Wadhwa et al., 2020). The functional groups of lipid–polymer hybrid nanoparticles on the surface are accessible for modifications that alter their physicochemical properties and affect the delivery of mRNA. Because lipopolyplexes can be designed to induce a high degree of nucleic acid condensation, they are emerging as potential delivery vectors for mRNA. Although the endosome escape mechanism of polyplexes is not well understood, a possible mechanism suggests that proton buffering by the polymer causes osmotic swelling, which leads to the rupture of the endosome (Bus et al. 2018). Van der Jeught et al. (2018) investigated T cell and inflammatory responses to an advanced lipopolyplex platform enclosing a lipid shell decorated with mannose receptor-targeting moieties that demonstrated excellent hemocompatibility and largely limited the mRNA expression to splenic antigen-presenting cells. Immunization with mRNA-containing lipopolyplexes evoked robust T cell immunity and was more efficient at suppressing tumor growth than the i.v. immunization by antigen mRNA-electroporated DCs and lipoplexes (Van der Jeught et al., 2018). An ionizable lipid, a phospholipid, a PEG-containing lipid, cholesterol, and an additive for the administration of mRNA vaccines comprise the formulation. Phospholipid and cholesterol are important for stability. The PEGylated lipid reduce aggregation, assist in vivo biodistribution, and minimizes nonspecific interactions (Pichon et al. 2013; Van der Jeught et al. 2018).
A typical mRNA vaccine consisting of condensed mRNA in a positively charged core structure is not efficiently internalized by the antigen-presenting cells while the mRNA-based vaccine is superior in triggering a strong anticancer immunity. An mRNA vaccine consisting of a core with a poly-(β-amino ester) polymer and mRNA enclosed by 1,2-dioleoyl-sn-glycero-3-ethylphosphocholine/1,2-dioleoyl-sn-glycero-3-phosphatidyl-ethanolamine/1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[amino(polyethyleneglycol)-2000 (EDOPC/DOPE/DSPE-PEG) forms lipopolyplexes that enter DCs via micropinocytosis (Persano et al. 2017). By boosting interferon- and interleukin-12 expression in DCs via toll-like receptor 7/8 signaling, this formulation demonstrated intrinsic adjuvant action. DCs treated with this mRNA vaccination demonstrated improved antigen presentation abilities. (Diken et al. 2011). Collectively, these core–shell architectures constitute a suitable framework for the creation of mRNA vaccines. Moreover, pH-responsive polymer systems have been tested for mRNA delivery. Like ionizable lipids, the ionizable aminoethylene side chains linked to poly(aspartamide)s release the mRNA molecules when they become protonated due to the acidic pH inside endosomes. The degree of protonation and the delivery efficacy of poly(aspartamide) are affected by the length and hydrophobicity of its side chains. mRNA can be delivered to various parts of the body by PEGylated poly(aspartamide) with an ethylenediamine side chain (Chaudhary et al. 2021).
Lipid calcium phosphate nanoparticles
LCP nanoparticles are recently developed nanoparticle system with a core that can be loaded with mRNA, siRNA, DNA, or chemotherapeutic drugs (Tang et al. 2015; Wang et al. 2018) (Fig. 1). LCP nanoparticles consist of a calcium phosphate core enclosed by lipid coating. The acid-sensitive core of the calcium phosphate core facilitates endosomal escape thus directing to release of the enclosed ingredients. The dissolution of the calcium phosphate core in endolysosomal compartment increases osmotic pressure and ruptures the endosomal membrane. LCP nanoparticles have been used to co-deliver PD-L1, siRNA, and mRNA using mannose as a ligand for targeting DCs, which present tumor antigens, thus prompting T cell activation and proliferation (Wang et al. 2018).
Cellular internalization
After reaching the target cells, LNPs can be internalized via various mechanisms. The exact mechanism of cellular internalization of lipid-based mRNA systems has not been fully explored, leading to indeterminate conclusions (Maugeri et al. 2019). Recent studies have displayed that this process comprises phagocytosis, micropinocytosis, clathrin-mediated endocytosis, and caveolin-mediated endocytosis (De Haes et al. 2012; Rush et al. 2014; Yasar et al. 2018). Upon internalization, nanoparticles may enter the endolysosomal pathway from where some enter the cytosol for translation, whereas others escape from the endosome into the cytosol for translation. The mechanisms involved in endosomal escape are hypothesized to be the proton sponge effect, pore formation, disruption of membrane, or membrane fusion (Selby et al. 2017; Varkouhi et al. 2011).
Application of lipid-based mRNA nanoparticles
Lipid-based mRNA vaccine CAR T-cell therapy
CAR T-cell therapy is an incipient cancer immunotherapeutic approach. There are a few CD19-directed therapeutics with FDA approval, namely, brexucabtagene for mantle cell lymphoma, tisagenlecleucel for acute lymphoblastic leukemia, and axicabtagene ciloleucel for diffuse large B-cell lymphoma (Mohanty et al. 2019). This therapeutic technique is based on T-cell engineering, which involves manipulating T-cells generated from patient blood in vitro to express artificial receptors that target a specific tumor antigen (Mirzaei et al. 2019). The engineered T cells are expanded in the laboratory and then infused into the patients for treatment. However, CAR T-cell therapy is related with side effects, including cytokine release syndrome, neurological toxicity, tumor lysis syndrome, and anaphylaxis (Mohanty et al. 2019). In addition, CAR T cells are mostly produced by viral transduction, which limits manufacturing and in vivo translation.
To increase the safety of CAR T-cell immunotherapy for cancer, mRNA has emerged as a potential treatment option that enhances transfection efficiency, CAR expression, and the potency to kill melanoma target cells. Electroporation is clinically used to deliver mRNA to cells such as T cells (de Macedo Abdo et al. 2020). A lipid-nanoparticle-based mRNA system for CAR T-cell therapy provides a promising approach to overcome these challenges. Ionizable LNPs have been validated for lower cytotoxicity in various cell types, including immune cells (Billingsley et al., 2020). Billingsley et al. (2020) demonstrated the fabrication of various LNP-mRNA formulations using ionizable lipids as shown in Fig. 3. The refined C14-4 LNPs were employed to encapsulate CAR mRNA to create functional CAR T cells, resulting in better mRNA delivery. In comparison to the effects of electroporation-based mRNA delivery, C14-4 LNPs generated reduced T cell toxicity while achieving comparable levels of CAR surface expression; hence, C14-4 LNPs were verified as a potentially feasible alternative technique for mRNA-based ex vivo engineering of CAR T cells. (Billingsley et al. 2020).
Fig. 3.
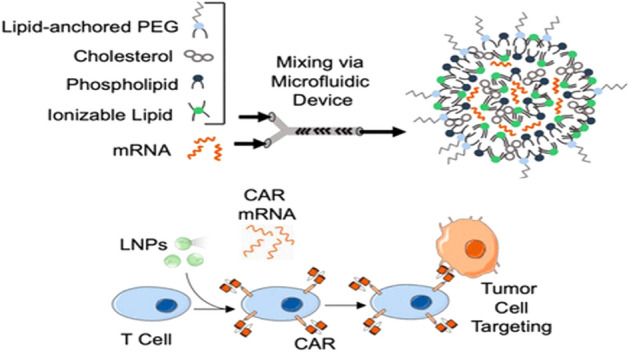
Schematic representation of preparation of lipid nanoparticles (LNPs) via microfluidic mixing encapsulating CAR mRNA. The LNP loaded with CAR mRNA induce CAR expression in T cells which results in the tumor targeting and cell death. Reprinted with permission from Billingsley et al. (2020).
Copyright© 2020 American Chemical Society
Lipid-based mRNA as cancer vaccine
Cancer vaccines can be prophylactic or therapeutic vaccines. Therapeutic vaccines contribute to boosting the immune system to detect and destroy antigens expressed in malignant tumors (Oberli et al. 2017). Different types of lipid-based mRNA vaccines are applied for effective treatment.
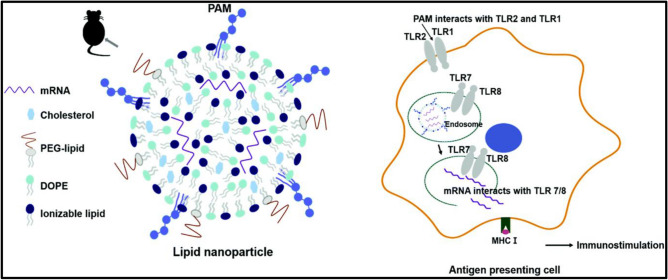
By activating various TLR subtypes, the combination of diverse adjuvant types can synergize immune stimulating effects. It is claimed that integrating additional adjuvants into LNPs can improve mRNA-mediated immune responses even further. Hydrophobic interaction between the lipid tails of Pam3, tri-palmitoyl-S-glyceryl cysteine connected to a pentapeptide (PAM3CSK4; Pam3) was introduced into LNPs (Lee et al. 2020). Pam3-LNPs induced an increase in immunological activation in cellular responses, resulting in a large antigen-specific CD8+ T cells. Co-stimulation of several TLR subtypes by additional adjuvants is likely to accelerate the evolution of LNP formulations as mRNA-based cancer immunotherapies as shown in Fig. 4 (Lee et al. 2020).
Fig. 4.
Lipid nanoparticle prepared using Pam3 lipid encapsulating mRNA for effective delivery of mRNA for cancer immunotherapy.
Reproduced from Lee et al. (2020) with permission from the Royal Society of Chemistry
As a cancer therapeutic alternative, an approach focused on evoking antigen-specific immunity is also being investigated (Benteyn et al. 2015). This work found that PD-L1 and PD-L2 siRNA delivery via DLin-KC2-DMA-containing LNPs resulted in efficient and specific reduction of PD-L expression in human monocyte-derived DCs. The established siRNA-LNP transfection approach had no effect on DC phenotypic or migratory capabilities and resulted in satisfactory DC vitality. siRNA-LNP transfection can be successfully paired with target antigen peptide loading and mRNA electroporation. Finally, these PD-L-silenced DCs, which were loaded with antigen mRNA, significantly increased ex vivo antigen-specific CD8+ T cell responses in cancer patients who had been transplanted. In conclusion, PD-L siRNA-LNP-modified DCs are appealing for clinical-level production and application to increase immune responses not only in transplanted cancer patients (Hobo et al. 2013).
Personalized cancer vaccines are also being explored in the field of mRNA-based nanovaccines. This system utilizes the release of neoantigens by utilizing various epitope sequences. Several clinical trials have been based on this system, as presented in Table 1.
Table 1.
mRNA based clinical trials
| NCT number | Cancer type | Components | Delivery system | Administration route | Phase |
|---|---|---|---|---|---|
| NCT03480152 | Metastatic melanoma, GI cancer, hepatocellular cancer | NCI-4650, a messenger ribonucleic acid (mRNA)-based | Cancer vaccine | Intramuscular | Phase I/II |
| NCT03164772 | Metastatic non-small-cell lung cancer | mRNA (BI 136,849) + durvalumab + tremelimumab | Combination immunotherapy and mRNA vaccine | N/A | Phase I/II |
| NCT01446731 | Prostatic neoplasms | mRNA + doxcetaxel | mRNA transfected dendritic cells | Intradermal | Phase II |
| NCT03948763 | Neoplasms, carcinoma, NSCL cancer, pancreatic neoplasm | mRNA-5671/V941 + pembrolizumab | N/A | Intramuscular | Phase I |
| NCT00890032 | Recurrent central nervous system neoplasm | BTSC mRNA loaded DCs | N/A | Intradermal | Phase I |
| NCT03468244 | Advanced esophageal squamous carcinoma, gastric adenocarcinoma, pancreatic adenocarcinoma, colorectal carcinoma | mRNA tumor vaccine encoding neoantigens | Personalized mRNA tumor vaccine | Subcutaneous | N/A |
| NCT00204516 | Malignant melanoma | Coding of mRNA for melanoma-associated antigens + GM-CSF | N/A | Intradermal | Phase I/II |
Lipid-based mRNA vaccine encoding a monoclonal antibody
Monoclonal antibodies are being utilized as frontline therapeutics for treating many ailments, including autoimmune disorders, cancer, and asthma. Antibodies are unique in that they can directly destroy tumor cells while also engaging the host immune system to create long-term effector responses against the tumor. The combination of a complex mechanism of action and target specificity distinguishes monoclonal antibody therapy from other conventional treatments, like as chemotherapy, and highlights antibodies' potential to generate robust antitumor responses while limiting toxicity and side effects (Van Hoecke et al. 2019).
Although monoclonal antibody treatment shows promising results for effective treatment regimens, there are several limits that affect their performance (Lu et al. 2020). The complex procedure of manufacturing and purifying the antibodies is vulnerable to post-translational modifications that can lead to altered therapeutic properties. Thus, this system needs to be optimized for better results. This mRNA-based alternative is critical for circumventing the limits of antibody-based drugs. It is conceivable to achieve in situ generation of therapeutic products by giving monoclonal antibody based mRNA without the need of an up-scaled synthesis and purification process (Jäger et al. 2013).
Conclusions and future perspective
Treatment options based on mRNA have emerged as promising approaches in cancer immunotherapy, with numerous ongoing clinical trials exploring various strategies. mRNA can be prepared in cell-free systems, and its expression can be controlled. It is safe for therapeutic applications because it avoids any risk associated with genomic integration. However, mRNA is unstable and undergoes hydrolysis. Therefore, suitable delivery vehicles are required to facilitate the therapeutic applications of mRNA. LNPs offer tremendous advantages over other delivery systems because they are expected to protect the mRNA and increase its stability and transfection efficiency to achieve improved therapeutic efficacy. The development of technologies for the preparation of mRNA and formulation of LNPs is expected to be scalable and robust, which can expedite the clinical implementation of cancer immunotherapy treatment using mRNA-based delivery systems.
The development of novel ionizable lipids is predicted to improve the efficacy of mRNA-based nanosystems. To improve the efficacy of mRNA-based anticancer therapy, a co-delivery method or specific targeting can be used. The integration of mRNA-based systems with existing immunotherapeutic strategies, as along with combination therapies, can enhance the immunotherapeutic responses against cancer.
However, the reasons for the low mRNA transfection efficiency observed in lymphocytes and monocytes must be addressed in future studies. The effect of the lipid composition, mRNA/lipid ratio, the biodegradability of the linker, the unsaturation status of the hydrophobic tail of the lipid, the fusogenicity, and other factors critical for transfection efficiency should be further explored. The modulation of various lipid structure components can ensure targeted delivery. Polymers or dendrimers can be incorporated into LNP-mRNA formulations to enhance their stability and target-specific delivery. The observations from the previous clinical studies will pave the way for the design of novel multifunctional lipids and the formulation of improved lipid–mRNA nanoparticles for application in immunotherapy for cancer.
Declarations
Conflict of interest
All authors (P. Karmacharya, B. P. Patil, and J. O. Kim) declare that they have no conflict of interest.
Statement of human and animal rights
This article does not contain any studies with human and animal subjects performed by any of the authors.
Acknowledgements
This research was supported by Yeungnam University Research Grant in 2021.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Prajeena Karmacharya and Basavaraj Rudragouda Patil have contributed equally to this study.
References
- Akinc A, Maier MA, Manoharan M, Fitzgerald K, Jayaraman M, et al. The Onpattro story and the clinical translation of nanomedicines containing nucleic acid-based drugs. Nat Nanotechnol. 2019;14:1084–1087. doi: 10.1038/s41565-019-0591-y. [DOI] [PubMed] [Google Scholar]
- Aldosari BN, Alfagih IM, Almurshedi AS. Lipid nanoparticles as delivery systems for RNA-based vaccines. Pharmaceutics. 2021;13:206. doi: 10.3390/pharmaceutics13020206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benteyn D, Heirman C, Bonehill A, Thielemans K, Breckpot K. mRNA-based dendritic cell vaccines. Expert Rev Vaccines. 2015;14:161–176. doi: 10.1586/14760584.2014.957684. [DOI] [PubMed] [Google Scholar]
- Billingsley MM, Singh N, Ravikumar P, Zhang R, June CH, et al. Ionizable lipid nanoparticle-mediated mRNA delivery for human CAR T cell engineering. Nano Lett. 2020;20:1578–1589. doi: 10.1021/acs.nanolett.9b04246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bus T, Traeger A, Schubert US. The great escape: how cationic polyplexes overcome the endosomal barrier. J Mater Chem B. 2018;6:6904–6918. doi: 10.1039/C8TB00967H. [DOI] [PubMed] [Google Scholar]
- Chaudhary N, Weissman D, Whitehead KA. mRNA vaccines for infectious diseases: principles, delivery and clinical translation. Nat Rev Drug Discov. 2021 doi: 10.1038/s41573-021-00283-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W, Li H, Liu Z, Yuan W. Lipopolyplex for therapeutic gene delivery and its application for the treatment of Parkinson's disease. Front Aging Neurosci. 2016;8:68. doi: 10.3389/fnagi.2016.00068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng Q, Wei T, Farbiak L, Johnson LT, Dilliard SA, et al. Selective organ targeting (SORT) nanoparticles for tissue-specific mRNA delivery and CRISPR–Cas gene editing. Nat Nanotechnol. 2020;15:313–320. doi: 10.1038/s41565-020-0669-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui S, Wang Y, Gong H, Lin X, et al. Correlation of the cytotoxic effects of cationic lipids with their headgroups. Toxicol Res. 2018;7:473–479. doi: 10.1039/C8TX00005K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cullis PR, Hope MJ. Lipid nanoparticle systems for enabling gene therapies. Mol Ther. 2017;25:1467–1475. doi: 10.1016/j.ymthe.2017.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darvin P, Toor SM, Sasidharan Nair V, Elkord E. Immune checkpoint inhibitors: recent progress and potential biomarkers. Exp Mol Med. 2018;50:1–11. doi: 10.1038/s12276-018-0191-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Decker WK, da Silva RF, Sanabria MH, Angelo LS, Guimarães F, et al. Cancer immunotherapy: historical perspective of a clinical revolution and emerging preclinical animal models. Front Immunol. 2017;8:829. doi: 10.3389/fimmu.2017.00829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Haes W, Van Mol G, Merlin C, De Smedt SC, Vanham G, et al. Internalization of mRNA lipoplexes by dendritic cells. Mol Pharm. 2012;9:2942–2949. doi: 10.1021/mp3003336. [DOI] [PubMed] [Google Scholar]
- de Macedo AL, Barros LRC, Viegas MS, Marques LVC, de Sousa FP, et al. Development of CAR-T cell therapy for B-ALL using a point-of-care approach. Oncoimmunology. 2020;9:1752592. doi: 10.1080/2162402X.2020.1752592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diken M, Kreiter S, Selmi A, Britten CM, Huber C, et al. Selective uptake of naked vaccine RNA by dendritic cells is driven by macropinocytosis and abrogated upon DC maturation. Gene Ther. 2011;18:702–708. doi: 10.1038/gt.2011.17. [DOI] [PubMed] [Google Scholar]
- Esfahani K, Roudaia L, Buhlaiga N, Del Rincon SV, Papneja N, et al. A review of cancer immunotherapy: from the past, to the present, to the future. Curr Oncol. 2020;27:S87–S97. doi: 10.3747/co.27.5223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ewert KK, Zidovska A, Ahmad A, Bouxsein NF, Evans HM, et al. Cationic liposome–nucleic acid complexes for gene delivery and silencing: pathways and mechanisms for plasmid DNA and siRNA. Top Curr Chem. 2010;296:191–226. doi: 10.1007/128_2010_70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenton OS, Kauffman KJ, McClellan RL, et al. Bioinspired alkenyl amino alcohol ionizable lipid materials for highly potent in vivo mRNA delivery. Adv Mater. 2016;28:2939–2943. doi: 10.1002/adma.201505822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guevara ML, Persano F, Persano S. Advances in lipid nanoparticles for mRNA-based cancer immunotherapy. Front Chem. 2020;8:589959. doi: 10.3389/fchem.2020.589959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hajj KA, Whitehead KA. Tools for translation: non-viral materials for therapeutic mRNA delivery. Nat Rev Mater. 2017;2:17056. doi: 10.1038/natrevmats.2017.56. [DOI] [Google Scholar]
- Hargadon KM, Johnson CE, Williams CJ. Immune checkpoint blockade therapy for cancer: an overview of FDA-approved immune checkpoint inhibitors. Int Immunopharmacol. 2018;62:29–39. doi: 10.1016/j.intimp.2018.06.001. [DOI] [PubMed] [Google Scholar]
- Heyes J, Palmer L, Bremner K, MacLachlan I. Cationic lipid saturation influences intracellular delivery of encapsulated nucleic acids. J Control Release. 2005;107:276–287. doi: 10.1016/j.jconrel.2005.06.014. [DOI] [PubMed] [Google Scholar]
- Hiam-Galvez KJ, Allen BM, Spitzer MH. Systemic immunity in cancer. Nat Rev Cancer. 2021;21:345–359. doi: 10.1038/s41568-021-00347-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hobo W, Novobrantseva TI, Fredrix H, Wong J, Milstein S, et al. Improving dendritic cell vaccine immunogenicity by silencing PD-1 ligands using siRNA-lipid nanoparticles combined with antigen mRNA electroporation. Cancer Immunol Immunother. 2013;62:285–297. doi: 10.1007/s00262-012-1334-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hou X, Zaks T, Langer R, et al. Lipid nanoparticles for mRNA delivery. Nat Rev Mater. 2021 doi: 10.1038/s41578-021-00358-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huertas P, Aguilera A. Cotranscriptionally formed DNA:RNA hybrids mediate transcription elongation impairment and transcription-associated recombination. Mol Cell. 2003;12:711–721. doi: 10.1016/j.molcel.2003.08.010. [DOI] [PubMed] [Google Scholar]
- Jäger V, Büssow K, Wagner A, Weber S, Hust M, et al. High level transient production of recombinant antibodies and antibody fusion proteins in HEK293 cells. BMC Biotechnol. 2013;13:52. doi: 10.1186/1472-6750-13-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jahanafrooz Z, Baradaran B, Mosafer J, Hashemzaei M, Rezaei T, et al. Comparison of DNA and mRNA vaccines against cancer. Drug Discov Today. 2020;25:552–560. doi: 10.1016/j.drudis.2019.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jayaraman M, Ansell SM, Mui BL, et al. Maximizing the potency of siRNA lipid nanoparticles for hepatic gene silencing in vivo. Angew Chem Int Ed Engl. 2012;51:8529–8533. doi: 10.1002/anie.201203263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karikó K. In vitro transcribed mRNA therapeutics: out of the shadows and into the spotlight. Mol Ther. 2019;27:691–692. doi: 10.1016/j.ymthe.2019.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kranz L, Diken M, Haas H, et al. Systemic RNA delivery to dendritic cells exploits antiviral defence for cancer immunotherapy. Nature. 2016;534:396–401. doi: 10.1038/nature18300. [DOI] [PubMed] [Google Scholar]
- Lee K, Kim SY, Seo Y, Kim MH, Chang J, et al. Adjuvant incorporated lipid nanoparticles for enhanced mRNA-mediated cancer immunotherapy. Biomater Sci. 2020;8:1101–1105. doi: 10.1039/C9BM01564G. [DOI] [PubMed] [Google Scholar]
- Liu C, Zhang L, Zhu W, Guo R, Sun H, et al. Barriers and strategies of cationic liposomes for cancer gene therapy. Mol Ther Methods Clin Dev. 2020;18:751–764. doi: 10.1016/j.omtm.2020.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lonez C, Vandenbranden M, Ruysschaert JM. Cationic lipids activate intracellular signaling pathways. Adv Drug Deliv Rev. 2012;64:1749–1758. doi: 10.1016/j.addr.2012.05.009. [DOI] [PubMed] [Google Scholar]
- Lou G, Anderluzzi G, Schmidt ST, Woods S, Gallorini S, et al. Delivery of self-amplifying mRNA vaccines by cationic lipid nanoparticles: the impact of cationic lipid selection. J Control Release. 2020;325:370–379. doi: 10.1016/j.jconrel.2020.06.027. [DOI] [PubMed] [Google Scholar]
- Love KT, Mahon KP, Levins CG, Whitehead KA, Querbes W, et al. Lipid-like materials for low-dose, in vivo gene silencing. Proc Natl Acad Sci USA. 2010;107:1864–1869. doi: 10.1073/pnas.0910603106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu RM, Hwang YC, Liu IJ, Lee CC, Tsai HZ, et al. Development of therapeutic antibodies for the treatment of diseases. J Biomed Sci. 2020;27:1. doi: 10.1186/s12929-019-0592-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malone RW, Felgner PL, Verma IM. Cationic liposome-mediated RNA transfection. Proc Natl Acad Sci USA. 1989;86:6077–6081. doi: 10.1073/pnas.86.16.6077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maugeri M, Nawaz M, Papadimitriou A, Angerfors A, Camponeschi A, et al. Linkage between endosomal escape of LNP-mRNA and loading into EVs for transport to other cells. Nat Commun. 2019;10:4333. doi: 10.1038/s41467-019-12275-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miao L, Li L, Huang Y, Delcassian D, Chahal J, et al. Delivery of mRNA vaccines with heterocyclic lipids increases anti-tumor efficacy by STING-mediated immune cell activation. Nat Biotechnol. 2019;37:1174–1185. doi: 10.1038/s41587-019-0247-3. [DOI] [PubMed] [Google Scholar]
- Miao L, Lin J, Huang Y, Li L, Delcassian D, et al. Synergistic lipid compositions for albumin receptor mediated delivery of mRNA to the liver. Nat Commun. 2020;11:2424. doi: 10.1038/s41467-020-16248-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirzaei HR, Jamali A, Jafarzadeh L, Masoumi E, Alishah K, et al. Construction and functional characterization of a fully human anti-CD19 chimeric antigen receptor (huCAR)-expressing primary human T cells. J Cell Physiol. 2019;234:9207–9215. doi: 10.1002/jcp.27599. [DOI] [PubMed] [Google Scholar]
- Mohanty R, Chowdhury CR, Arega S, Sen P, Ganguly P, et al. CAR T cell therapy: a new era for cancer treatment (Review) Oncol Rep. 2019;42:2183–2195. doi: 10.3892/or.2019.7335. [DOI] [PubMed] [Google Scholar]
- Morille M, Passirani C, Vonarbourg A, Clavreul A, Benoit JP. Progress in developing cationic vectors for non-viral systemic gene therapy against cancer. Biomaterials. 2008;29:3477–3496. doi: 10.1016/j.biomaterials.2008.04.036. [DOI] [PubMed] [Google Scholar]
- Oberli MA, Reichmuth AM, Dorkin JR, Mitchell MJ, Fenton OS, et al. Lipid nanoparticle assisted mRNA delivery for potent cancer immunotherapy. Nano Lett. 2017;17:1326–1335. doi: 10.1021/acs.nanolett.6b03329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oiseth SJ, Aziz MS. Cancer immunotherapy: a brief review of the history, possibilities, and challenges ahead. J Cancer Metastasis Treat. 2017;3:250–261. doi: 10.20517/2394-4722.2017.41. [DOI] [Google Scholar]
- Ou W, Jiang L, Thapa RK, Soe ZC, Poudel K, et al. Combination of NIR therapy and regulatory T cell modulation using layer-by-layer hybrid nanoparticles for effective cancer photoimmunotherapy. Theranostics. 2018;8:4574–4590. doi: 10.7150/thno.26758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pantin J, Battiwalla M. Upsetting the apple CAR-T (chimeric antigen receptor T-cell therapy)—sustainability mandates USA innovation. Br J Haematol. 2020;190:851–853. doi: 10.1111/bjh.16685. [DOI] [PubMed] [Google Scholar]
- Pardi N, Hogan MJ, Porter FW, Weissman D. mRNA vaccines—a new era in vaccinology. Nat Rev Drug Discov. 2018;17:261–279. doi: 10.1038/nrd.2017.243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Persano S, Guevara ML, Li Z, Mai J, Ferrari M, et al. Lipopolyplex potentiates anti-tumor immunity of mRNA-based vaccination. Biomaterials. 2017;125:81–89. doi: 10.1016/j.biomaterials.2017.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pichon C, Midoux P. Mannosylated and histidylated LPR technology for vaccination with tumor antigen mRNA. In: Rabinovich PM, editor. Synthetic messenger RNA and cell metabolism modulation (methods and protocols) Totowa: Humana Press; 2013. pp. 247–274. [DOI] [PubMed] [Google Scholar]
- Poudel K, Park S, Hwang J, Ku SK, Yong CS, et al. Photothermally modulatable and structurally disintegratable sub-8-nm Au1Ag9 embedded nanoblocks for combination cancer therapy produced by plug-in assembly. ACS Nano. 2020;14:11040–11054. doi: 10.1021/acsnano.9b09731. [DOI] [PubMed] [Google Scholar]
- Reichmuth AM, Oberli MA, Jaklenec A, Langer R, Blankschtein D. mRNA vaccine delivery using lipid nanoparticles. Ther Deliv. 2016;7:319–334. doi: 10.4155/tde-2016-0006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rush AM, Nelles DA, Blum AP, Barnhill SA, Tatro ET, et al. Intracellular mRNA regulation with self-assembled locked nucleic acid polymer nanoparticles. J Am Chem Soc. 2014;136:7615–7618. doi: 10.1021/ja503598z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabnis S, Kumarasinghe ES, Salerno T, Mihai C, Ketova T, et al. A novel amino lipid series for mRNA delivery: improved endosomal escape and sustained pharmacology and safety in non-human primates. Mol Ther J Am Soc Gene Ther. 2018;26(6):1509–1519. doi: 10.1016/j.ymthe.2018.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahin U, Karikó K, Türeci Ö. mRNA-based therapeutics—developing a new class of drugs. Nat Rev Drug Discov. 2014;13:759–780. doi: 10.1038/nrd4278. [DOI] [PubMed] [Google Scholar]
- Samaridou E, Heyes J, Lutwyche P. Lipid nanoparticles for nucleic acid delivery: current perspectives. Adv Drug Deliv Rev. 2020;154–155:37–63. doi: 10.1016/j.addr.2020.06.002. [DOI] [PubMed] [Google Scholar]
- Selby LI, Cortez-Jugo CM, Such GK, Johnston APR. Nanoescapology: progress toward understanding the endosomal escape of polymeric nanoparticles. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2017 doi: 10.1002/wnan.1452. [DOI] [PubMed] [Google Scholar]
- Semple SC, Klimuk SK, Harasym TO, Dos Santos N, Ansell SM, et al. Efficient encapsulation of antisense oligonucleotides in lipid vesicles using ionizable aminolipids: formation of novel small multilamellar vesicle structures. Biochim Biophys Acta. 2001;1510:152–166. doi: 10.1016/S0005-2736(00)00343-6. [DOI] [PubMed] [Google Scholar]
- Simões S, Slepushkin V, Pires P, Gaspar R, de Lima MP, et al. Mechanisms of gene transfer mediated by lipoplexes associated with targeting ligands or pH-sensitive peptides. Gene Ther. 1999;6:1798–1807. doi: 10.1038/sj.gt.3301015. [DOI] [PubMed] [Google Scholar]
- Suzuki Y, Hyodo K, Suzuki T, Tanaka Y, Kikuchi H, et al. Biodegradable lipid nanoparticles induce a prolonged RNA interference-mediated protein knockdown and show rapid hepatic clearance in mice and nonhuman primates. Int J Pharm. 2017;519(1):34–43. doi: 10.1016/j.ijpharm.2017.01.016. [DOI] [PubMed] [Google Scholar]
- Tang J, Li L, Howard CB, Mahler SM, Huang L, et al. Preparation of optimized lipid-coated calcium phosphate nanoparticles for enhanced in vitro gene delivery to breast cancer cells. J Mater Chem B. 2015;3:6805–6812. doi: 10.1039/C5TB00912J. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tateshita N, Miura N, Tanaka H, Masuda T, Ohtsuki S, et al. Development of a lipoplex-type mRNA carrier composed of an ionizable lipid with a vitamin E scaffold and the KALA peptide for use as an ex vivo dendritic cell-based cancer vaccine. J Control Release. 2019;310:36–46. doi: 10.1016/j.jconrel.2019.08.002. [DOI] [PubMed] [Google Scholar]
- Tossberg JT, Esmond TM, Aune TM. A simplified method to produce mRNAs and functional proteins from synthetic double-stranded DNA templates. Biotechniques. 2020;69:281–288. doi: 10.2144/btn-2020-0037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ulkoski D, Bak A, Wilson JT, Krishnamurthy VR. Recent advances in polymeric materials for the delivery of RNA therapeutics. Expert Opin Drug Deliv. 2019;16:1149–1167. doi: 10.1080/17425247.2019.1663822. [DOI] [PubMed] [Google Scholar]
- Van der Jeught K, De Koker S, Bialkowski L, Heirman C, Tjok Joe P, et al. Dendritic cell targeting mRNA lipopolyplexes combine strong antitumor T-cell immunity with improved inflammatory safety. ACS Nano. 2018;12:9815–9829. doi: 10.1021/acsnano.8b00966. [DOI] [PubMed] [Google Scholar]
- Van Hoecke L, Roose K. How mRNA therapeutics are entering the monoclonal antibody field. J Transl Med. 2019;17:54. doi: 10.1186/s12967-019-1804-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varkouhi AK, Scholte M, Storm G, Haisma HJ. Endosomal escape pathways for delivery of biologicals. J Control Release. 2011;151:220–228. doi: 10.1016/j.jconrel.2010.11.004. [DOI] [PubMed] [Google Scholar]
- Wadhwa A, Aljabbari A, Lokras A, Foged C, Thakur A. Opportunities and challenges in the delivery of mRNA-based vaccines. Pharmaceutics. 2020;12:102. doi: 10.3390/pharmaceutics12020102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waldman AD, Fritz JM, Lenardo MJ. A guide to cancer immunotherapy: from T cell basic science to clinical practice. Nat Rev Immunol. 2020;20:651–668. doi: 10.1038/s41577-020-0306-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Zhang L, Xu Z, Miao L, Huang L. mRNA vaccine with antigen-specific checkpoint blockade induces an enhanced immune response against established melanoma. Mol Ther. 2018;26:420–434. doi: 10.1016/j.ymthe.2017.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yasar H, Biehl A, De Rossi C, Koch M, Murgia X, et al. Kinetics of mRNA delivery and protein translation in dendritic cells using lipid-coated PLGA nanoparticles. J Nanobiotechnol. 2018;16:72. doi: 10.1186/s12951-018-0401-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang H, You X, Wang X, Cui L, Wang Z, et al. Delivery of mRNA vaccine with a lipid-like material potentiates antitumor efficacy through Toll-like receptor 4 signaling. Proc Natl Acad Sci USA. 2021;118:e2005191118. doi: 10.1073/pnas.2005191118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J, Shrivastava S, Cleveland RO, Rabbitts TH. Lipid–mRNA nanoparticle designed to enhance intracellular delivery mediated by shock waves. ACS Appl Mater Interfaces. 2019;11:10481–10491. doi: 10.1021/acsami.8b21398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou S, Chen W, Cole J, Zhu G. Delivery of nucleic acid therapeutics for cancer immunotherapy. Med Drug Discov. 2020;6:100023. doi: 10.1016/j.medidd.2020.100023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Sun C, Wang C, Jankovic KE, Dong Y. Lipids and lipid derivatives for RNA delivery. Chem Rev. 2021 doi: 10.1021/acs.chemrev.1c00244. [DOI] [PMC free article] [PubMed] [Google Scholar]