Abstract
A 60-year-old woman with a past medical history of asthma presented with fulminant myocarditis 9 days after testing positive for SARS-CoV-2 and 16 days after developing symptoms consistent with COVID-19. Her hospital course was complicated by the need for veno-arterial extracorporeal membrane oxygenation, ventricular arrhythmias, and pseudomonas bacteremia. She ultimately recovered and was discharged to home with normal left ventricular systolic function. Thereafter, she developed symptomatic ventricular tachycardia, for which she received an implantable cardioverter-defibrillator and antiarrhythmic drug therapy.
Key Words: COVID-19, fulminant myocarditis, ventricular arrhythmias
Abbreviations and Acronyms: CMR, cardiac magnetic resonance; CRRT, continuous renal replacement therapy; EMB, endomyocardial biopsy; hs-TnT, high-sensitivity troponin T; LVEF, left ventricular ejection fraction; VA ECMO, veno-arterial extracorporeal membrane oxygenation; VT, ventricular tachycardia
Case Presentation
A 60-year-old woman with a past medical history of asthma presented to the emergency department (ED) during the summer of 2020 with extreme fatigue, shortness of breath, and palpitations.
Nine days prior, she was seen by her primary care physician to evaluate a week of symptoms, including fever, decreased sense of smell and taste, body aches, sore throat, and congestion. These symptoms began several days after close contact with someone who was COVID-19 positive. She was diagnosed with SARS-CoV-2 infection by a PCR test and was started on supportive treatment with antipyretics. Although her symptoms improved initially, she came to the ED because of worsening fatigue, shortness of breath, and a heart rate of approximately 140 beats/min on a home monitor.
Initial vital signs included a heart rate of 141 beats/min, blood pressure of 138/63 mm Hg, temperature of 97.3 °F, respiratory rate of 16 breaths/min, and oxygen saturation by pulse oximetry of 98% on room air. Upon arrival, she was not in respiratory distress, her lungs were clear to auscultation, cardiac auscultation was unremarkable except for tachycardia, and neither jugular venous distention nor pedal edema was present.
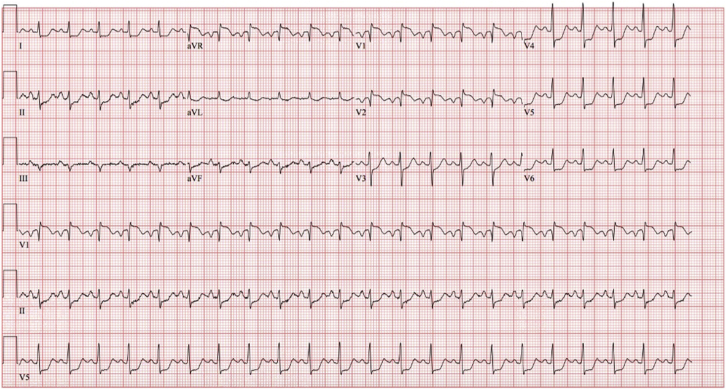
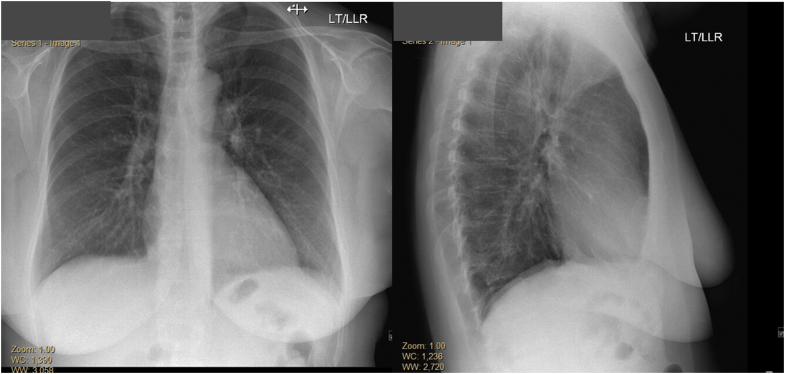
Her initial electrocardiogram demonstrated sinus tachycardia, ST-segment elevation in leads V1, V2, and aVR, and ST-segment depression in multiple other leads (Figure 1). Her chest X-ray showed a normal heart size; normal pulmonary vasculature; and absence of focal consolidation, pleural effusion, or pneumothorax (Figure 2). Laboratory testing at presentation revealed a significantly elevated high-sensitivity troponin T (hs-TnT) of 1,737 ng/L (normal 0-14 ng/L) (Table 1). Cardiology consultation was requested.
Figure 1.
Electrocardiogram at Presentation
A 12-lead electrocardiogram showing sinus tachycardia; ST-segment elevation in avR, V1, and V2; and ST-segment depression in multiple other leads.
Figure 2.
Chest X-Ray at Presentation
Chest X-ray, anteroposterior and lateral view showing normal heart size and pulmonary vasculature. No focal consolidations, pleural effusions, or pneumothorax.
Table 1.
Laboratory Testing at Different Time Points of the Hospitalization
| Laboratory Test | Presentation Day 1 |
Day 2 | ECMO Day 1 | ECMO Day 2 | Discharge | Normal Range |
|---|---|---|---|---|---|---|
| High-sensitivity troponin T, ng/L | 1,737 | 2,018 | 0-14 | |||
| NT-proBNP, pg/mL | 250 | 19,054 | 0-125 | |||
| C-reactive protein, mg/L | 44.79 | 88.61 | <10 | |||
| Sodium, mEq/L | 140 | 132 | 149 | 141 | 146 | 135-148 |
| Potassium, mEq/L | 4.0 | 5.4 | 3.6 | 5.1 | 4.7 | 3.4-5.3 |
| Chloride, mEq/L | 101 | 95 | 101 | 101 | 111 | 96-110 |
| Carbon dioxide, mEq/L | 23 | 5 | 21 | 26 | 24 | 19-32 |
| Glucose, mg/dL | 108 | 217 | 189 | 77 | 72-99 | |
| BUN, mg/dL | 10 | 23 | 33 | 28 | 26 | 3-29 |
| Creatinine, mg/dL | 0.6 | 1.4 | 1.86 | 2.28 | 1.2 | 0.5-1.2 |
| Calcium, mg/dL | 9.4 | 9.2 | 8.5-10.5 | |||
| Lactate, mmol/L | 14.9 | 17.5 | 1.56 | 0.5-1.6 | ||
| Anion gap | 16 | 32 | 31 | 19 | 5-15 | |
| Bilirubin, mg/dL | 0.4 | 1.0 | 0.2 | 0-1.5 | ||
| AST, U/L | 199 | 4,393 | 6,200 | 18 | 14-40 | |
| ALT, U/L | 118 | 3,240 | 2,480 | 24 | 9-42 | |
| ALP, U/L | 178 | 98 | 116 | 32-126 | ||
| WBC, K/mm3 | 10.3 | 11.2 | 26.9 | 12.3 | 3.8-10.8 | |
| Hemoglobin, g/dL | 14.5 | 12.8 | 9.6 | 8.6 | 12-15.6 | |
| Platelet count | 275 | 253 | 157 | 264 | 130-400 | |
| PT | 17.4 | Seconds | ||||
| INR | 1.4 | Seconds |
ALP = alkaline phosphatase; ALT = alanine transaminase; AST = aspartate transaminase; BUN = blood urea nitrogen; ECMO = extracorporeal membrane oxygenation; INR = international normalized ratio; NT-proBNP = N-terminal pro–B-type natriuretic peptide; PT = prothrombin time; WBC = white blood cell.
Out of concern for an acute coronary syndrome, she was treated with aspirin, intravenous heparin, intravenous nitroglycerin, metoprolol, and atorvastatin. A repeat hs-TnT, 4 hours after the initial troponin was 2,382 ng/L. She was taken for urgent coronary angiography, which demonstrated normal coronary arteries. Her left ventricular end-diastolic pressure was 17 mm Hg with an arterial blood pressure of 96/65 mm Hg. Left ventriculography demonstrated mild global hypokinesis. An echocardiogram done around the same time showed moderately increased left ventricular wall thicknesses, mild global left ventricular systolic dysfunction (left ventricular ejection fraction [LVEF] 49%, Simpsons), grade II diastolic dysfunction, and a trivial to small pericardial effusion. Based on these findings, a presumptive diagnosis of viral myocarditis was made.
Within a few hours of her cardiac catheterization, she developed worsening hypotension that was treated with a fluid bolus. A computed tomography scan of the abdomen and pelvis found no evidence of a retroperitoneal bleed. Over the next 12-24 hours, her clinical status deteriorated, with markedly increased work of breathing. Examination showed jugular venous distension and diffuse crackles on lung examination, and she was started on noninvasive positive pressure ventilation. She also showed signs of a low-output state, with cold, clammy extremities and drowsiness. A repeat echocardiogram showed worsening left ventricular systolic function with an ejection fraction of 30%-35%, moderately increased left ventricular wall thicknesses with hyperechoic myocardial echotexture, and a small pericardial effusion (Videos 1 and 2). For worsening hypoxemia despite noninvasive positive-pressure ventilation, invasive mechanical ventilation was initiated. Pertinent laboratory values at that time included pH 6.96, pCO2 20.7 mEq (normal 35-45 mEq), bicarbonate 4.7 mEq (normal 22-26 mEq), pO2 247 mm Hg (normal 80-100 mm Hg), oxygen saturation 98% by arterial blood gas, lactate 14.9 mmol/L (normal 0.5-1.6 mmol/L), sodium 132 mEq/L, creatinine 1.4 mg/dL, N-terminal pro–B-type natriuretic peptide 19,054 pg/mL (normal <300 pg/mL), interleukin-6 2,169 pg/mL (normal <6 pg/mL) and hs-TnT 5,810 ng/L. She was started on intravenous dobutamine and administered a dose of intravenous furosemide. Her clinical status continued to deteriorate, with persistent hypotension requiring escalating doses of norepinephrine, vasopressin, epinephrine, and phenylephrine. Within the next few hours, she had 2 episodes of sustained ventricular tachycardia (VT), for which she underwent synchronized cardioversions twice and was started on intravenous amiodarone. An arterial blood gas showed severe metabolic acidosis, for which she was started on intravenous bicarbonate.
On hospital day 3, she remained hypotensive with acidemia and was started on veno-arterial extracorporeal membrane oxygenation (VA ECMO), cannulating the right femoral artery and vein. Her rapid hemodynamic deterioration suggested fulminant myocarditis. Several forms of percutaneous temporary mechanical support were considered by the shock team, informed by our center’s experience. Primary bridge-to-decision VA ECMO was favored because of her rapid hemodynamic compromise, need for full circulatory support, risk of worsening right ventricular failure in the setting of myocarditis, and ability of ECMO to provide adequate oxygenation should COVID-19 acute respiratory distress syndrome evolve.
While on VA ECMO, continuous renal replacement therapy (CRRT) was initiated for worsening renal function. A bedside echocardiogram on hospital day 6 demonstrated severely reduced left ventricular systolic function with an ejection fraction <15%, along with increased left ventricular wall thicknesses and a small cavity size. Based on her presumed diagnosis of fulminant COVID-19 myocarditis, she was treated with 3 days of intravenous methylprednisolone (1 g/d), followed by a prednisone taper (60 mg daily initially, weaning 10 mg every 3 days). Concurrent with this, she experienced multiple episodes of nonsustained VT, for which she was continued on intravenous amiodarone and started on lidocaine. Over the next few days, her inotropic support was weaned off. On ECMO day 9, flow was weaned to 1.3 L/min, without worsening oxygenation, and a bedside echocardiogram demonstrated severely reduced but improving left ventricular systolic function with an ejection fraction of 25%-30%. On ECMO day 10, she was taken to the operating room, ECMO was weaned, and she was decannulated successfully. She required a brief course of vasopressin, epinephrine, and milrinone following decannulation. CRRT was discontinued on hospital day 20, transitioning to intermittent hemodialysis. Her urine output increased with intravenous furosemide within 6 days after CRRT discontinuation, and no further dialysis was required thereafter.
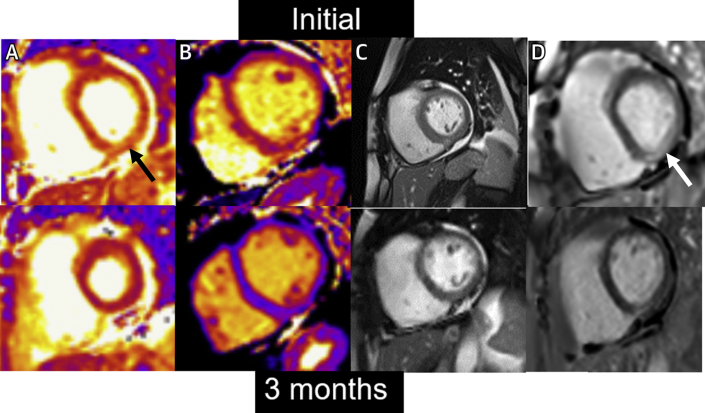
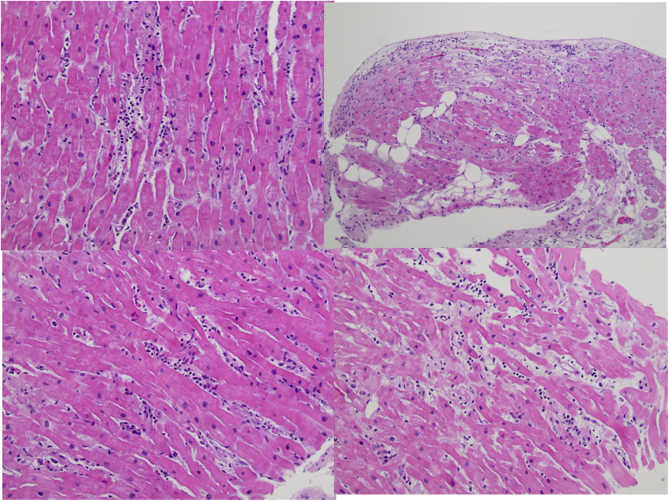
On hospital day 24, a repeat echocardiogram demonstrated improvement in her left ventricular systolic function with an ejection fraction of 50%-55%, normal left ventricular size, and reduced left ventricular wall thickness. Given stable and acceptable hemodynamics, she was started on metoprolol succinate, isosorbide dinitrate, and hydralazine. Cardiac magnetic resonance (CMR) was ordered to confirm the diagnosis of myocarditis and to assess more fully left and right ventricular systolic function. Diffuse hyperintensity on T2 mapping (up to 67 milliseconds, normal <52 milliseconds) suggestive of myocardial edema or inflammation was observed (Figure 3, Video 3). The myocardial extracellular volume fraction was significantly elevated at 39% (normal <29%), indicative of diffuse interstitial expansion. LVEF and right ventricular ejection fraction were 45% and 33%, respectively, with normal biventricular size. Right heart catheterization and endomyocardial biopsy (EMB) were also performed to exclude granulomatous myocarditis, given the arrhythmia burden. The biopsy showed scattered perivascular and interstitial inflammatory cells consisting of CD3-positive T-lymphocytes, CD20 positive B-lymphocytes, and histiocytes, along with interstitial and myocyte injury, suggestive of acute viral myocarditis. Viral immunostains for herpes simplex, adenovirus, and cytomegalovirus were negative. Congo red and iron stains were negative. There was no evidence of multinucleated giant cells, eosinophils, or granulomas (Figure 4).
Figure 3.
Cardiac Magnetic Resonance Imaging During Hospitalization and Follow-Up
(Top) Initial CMR. (A) Basal short-axis T2 map. Inferolateral wall (arrow) with elevated value of 67 ms (normal <52 ms). (B) Mid short-axis T1 map. Septal native T1 = 1,207 ms (normal <1,080 ms). (C) Basal short-axis cine. Quantitative LVEF = 45%. Quantitative RVEF = 33%. Small pericardial effusion. (D) Basal short-axis late gadolinium enhancement image. Note inferolateral epicardial late gadolinium enhancement (arrow). (Bottom) 3-month follow-up CMR. (A) Basal short-axis T2 map. Inferolateral wall 48 ms. (B) Mid short-axis T1 map. Septal native T1 = 967 ms. (C) Basal short-axis cine. Quantitative LVEF = 58%. Quantitative RVEF = 46%. (D) Basal short-axis late gadolinium enhancement image. Note improvement in inferolateral fibrosis compared to initial scan. CMR = cardiac magnetic resonance; LVEF = left ventricular ejection fraction; RVEF = right ventricular ejection fraction.
Figure 4.
Endomyocardial Biopsy
Endomyocardial biopsy showing scattered perivascular and interstitial inflammatory cell infiltrates, interstitial and myocyte injury, suggestive of acute myocarditis. Predominant CD3 T-lymphocytes fewer CD20 B-lymphocytes and CD68 histiocytes. Viral immunostains for herpes simplex virus, adenovirus, and cytomegalovirus were negative.
Her hospital course was further complicated by pseudomonas bacteremia from infection at the ECMO cannula site, for which she completed a course of antibiotics. She was discharged to home on hospital day 40 with improved ventricular function by echocardiography (Videos 4 and 5). Before discharge, she underwent placement of an implantable loop recorder (ILR) and an external defibrillator vest and guideline-directed medical therapy for heart failure (metoprolol succinate, lisinopril, amiodarone, and furosemide) was continued. An implantable cardioverter-defibrillator was considered; however, preference was given to a wearable defibrillator, as the LVEF was >45%, and her myocarditis was considered to be an acute and potentially reversible insult. The ILR was felt to be an important adjunct, given her significant arrhythmia burden during hospitalization and its ability to help guide her need for an implantable cardioverter-defibrillator in follow-up.
At her 3-month cardiology follow-up, she underwent repeat CMR (Figure 3, Video 6) that demonstrated normal left ventricular systolic function with an ejection fraction of 58%, a low-normal right ventricular ejection fraction of 46%, and residual linear epicardial nonischemic fibrosis involving the basal to mid inferolateral and lateral left ventricular walls, suggestive of prior myocarditis (Figure 3). Interval improvement was noted in right and left ventricular systolic function, along with resolution of myocardial edema/inflammation and diffuse interstitial expansion. As her left ventricular systolic function had improved and no significant arrhythmias were noted with interrogation of her ILR, implantation of a cardioverter-defibrillator was deferred. However, 6 months later (9 months after her initial presentation) she presented to the ED with lightheadedness and was found to be in a wide complex tachycardia (confirmed to be VT) at a rate of 176 beats/min. Interrogation of her ILR demonstrated persistence of this rhythm for 2 hours and 30 minutes. Thereafter, she underwent an electrophysiology study that demonstrated multiple VT morphologies. For this, a cardioverter-defibrillator was implanted, and she was started on antiarrhythmic drug therapy with mexiletine. She has done well in follow-up, with preserved left ventricular systolic function, improving functional capacity, and no further arrhythmias.
Overview of Fulminant Myocarditis
Epidemiology, presentation, evaluation, and management
Fulminant myocarditis is a life-threatening condition characterized by severe myocardial inflammation with myocardial edema and myocyte necrosis. Defining features include a rapidly progressive course, with cardiogenic shock, life-threatening cardiac arrhythmias, and multiorgan failure.
The incidence of viral myocarditis is estimated to be approximately 10 to 20 cases per 100,000 per year.1 Although the incidence of fulminant myocarditis is less well-defined, it is considered quite rare. Classically, fulminant myocarditis has a lymphocytic infiltrate on EMB, with further categorization by etiology (eg, infectious, autoimmune, toxic).2
Most commonly, patients with fulminant myocarditis have symptoms and signs of heart failure with impending cardiogenic shock. Electrocardiographic findings may include low voltage in the setting of myocardial edema, significant ST-segment changes, and both tachyarrhythmias and bradyarrhythmias.3 Laboratory abnormalities reflect compromised perfusion of various organs, with elevation of cardiac troponin being ubiquitous. A key feature of fulminant myocarditis is its rapid progression, with cardiogenic shock that requires vasoactive medications, temporary mechanical circulatory support (MCS), and/or extracorporeal life support (ECLS). Invasive evaluation with coronary angiography should be performed to assess for an ischemic etiology, along with hemodynamic monitoring to identify and address cardiogenic shock.3,4
Use of immunosuppressive therapy in fulminant viral myocarditis is not well supported by the literature and generally is reserved for patients with nonviral etiologies (eg, giant cell myocarditis, necrotizing eosinophilic myocarditis, and immune checkpoint inhibitor myocarditis).3 Despite this, patients with viral myocarditis are frequently treated with corticosteroids.
MCS, Imaging, EMB, and Arrhythmias in Fulminant Myocarditis
Mechanical circulatory support
Hemodynamic and respiratory support represent early goals of care for patients with fulminant myocarditis, with close monitoring of end-organ perfusion. Early transfer to an advanced heart failure center is recommended if fulminant myocarditis is suspected. Temporary MCS devices should be initiated in cases of refractory shock. Because biventricular failure is common, preference may instead be given to ECLS.3
The use of hemodynamic support devices in this population carries significant risks, including bleeding, coagulopathy, thrombosis, acute limb ischemia, infection, and neurological events. In most circumstances, however, the potential benefits of MCS and ECLS in fulminant myocarditis outweigh the risks.5,6
Liberating patients from MCS depends upon hemodynamics, cardiac output, and response to trials involving a reduction in circulatory support. Close monitoring for hemostasis and short-term complications is recommended thereafter.
Choice of Imaging
Echocardiography is the preferred imaging modality for initial evaluation of patients with suspected fulminant myocarditis, as it is widely available and can be performed at the bedside in rapidly deteriorating patients.3 Increased myocardial thickness caused by myocardial edema with regional wall motion abnormalities in a noncoronary distribution and normal left ventricular size represent findings consistent with myocarditis; in contrast, a dilated ventricle is more often suggestive of an alternative process.7 Presence of regional wall motion abnormalities may introduce challenges in discerning this from an acute coronary syndrome.
In all cases of myocarditis, exclusion of obstructive coronary artery disease is important, most often with invasive coronary angiography. This is particularly important in the setting of COVID-19, in which ischemic heart disease may be found at a higher rate.8
CMR can be used to confirm myocarditis and is the preferred noninvasive test for its evaluation.3,9, 10, 11 However, in fulminant cases, patient instability may prohibit its use, at least initially. Updated Lake Louise Criteria, which incorporate T1 abnormalities (elevated native T1 and/or late gadolinium enhancement [LGE]) and elevated native T2 signals (in a focal or global distribution representative of myocardial edema or inflammation), along with additional supportive criteria (eg, regional or global dysfunction, pericardial effusion) (Figure 3), are highly sensitive and specific for those with suspected myocarditis.12,13 T1 and T2 mapping improves diagnostic yield with CMR, and patterns of abnormalities can be used to identify specific types of myocarditis.14 CMR also has prognostic utility, with persistence and extent of LGE predicting adverse outcomes including mortality and arrhythmias.15 Finally, CMR can also be used to guide sites for endomyocardial biopsy (EMB).9
Role of biopsy
Traditionally, EMB has been considered the gold standard for diagnosis in patients with acute myocarditis. In cases of fulminant myocarditis, EMB can help discern the specific etiology and provide additional assistance in guiding therapy.9 In less severe cases of myocarditis, EMB has a lower sensitivity for diagnosis than CMR and is also less likely to inform management.2,16 EMB is also limited by low yield because of sampling error and lack of consensus on standard criteria for interpretation.10,11,16 In our case, EMB helped confirm the diagnosis of post-viral lymphocytic myocarditis, while ruling out alternative etiologies.
Arrhythmias
Atrial and ventricular arrhythmias are common in fulminant myocarditis and may have multiple underlying mechanisms (eg, myocardial inflammation or edema, scar).3,4,17 Exercise can trigger arrhythmias in acute myocarditis, and as such, current guidance cautions against exercise for 3-6 months to ensure resolution of myocardial inflammation.18 In our patient, ventricular arrhythmias were observed at 2 different time points. During her hospitalization, polymorphic VT was felt to be the result of severe myocardial inflammation. This arrhythmia continued in the early post-ECMO period and then abated as ventricular function improved later in her course. As the acute phase resolved, no further episodes of VT were detected on her ILR. Nine months after discharge, however, she presented with symptomatic VT. CMR done at that time showed persistent LGE, and her VT was likely driven by presence of myocardial scar or persistent inflammation. Importantly, persistent LGE on CMR is an independent predictor of arrhythmias independent of ventricular function.15,17
COVID-19 and Myocarditis
In a recent study that included ∼154,000 patients with COVID-19, the HR for myocarditis was 5.4 compared with uninfected contemporary and historical cohorts.8 Despite this, fulminant myocarditis with COVID-19 remains rare,19,20 with most reported cases being mild in severity. Autopsy studies have further suggested that although nonspecific cardiac inflammation is common with COVID-19, myocarditis meeting strict pathological criteria is rare.21 Multiple reports have also noted myocardial involvement by CMR among asymptomatic and minimally symptomatic individuals.22,23 Collectively, this has made estimation of the prevalence and incidence of myocarditis with COVID-19 quite challenging.
In addition to myocarditis, other forms of cardiovascular disease may manifest after SARS-CoV-2 infection, including pericarditis, myocardial ischemia, microvascular disease, nonischemic cardiomyopathy, stress cardiomyopathy, thromboembolism, and arrhythmia. For others, cardiovascular symptoms (eg, chest pain, dyspnea, palpitations) may occur or persist, with no specific cardiac etiology identified after testing. The 2022 ACC Expert Consensus Decision Pathway on Cardiovascular Sequelae of COVID-19 in Adults provides a framework by which to evaluate and manage these individuals.24
Conclusions
This case of fulminant myocarditis following SARS-CoV-2 infection highlights many of the severe clinical manifestations that may occur with this condition. Early recognition and implementation of supportive care (eg, ECLS) in cases such as this is critically important. Close monitoring of these patients after discharge is also paramount, as additional risks (eg, arrhythmic) may persist.
Funding Support and Author Disclosures
The authors have reported that they have no relationships relevant to the contents of this paper to disclose.
Footnotes
Muthiah Vaduganathan, MD, MPH, served as Guest Associate Editor for this paper. Javed Butler, MD, MPH, MBA, served as Guest Editor-in-Chief for this paper.
This paper is co-published in the Journal of the American College of Cardiology.
The authors attest they are in compliance with human studies committees and animal welfare regulations of the authors’ institutions and Food and Drug Administration guidelines, including patient consent where appropriate. For more information, visit the Author Center.
Appendix
For supplemental videos, please see the online version of this paper.
Appendix
Transthoracic Echocardiogram at Presentation Parasternal View. Two-dimensional echocardiogram in parasternal long-axis view showing left ventricular hypertrophy and severe left ventricular hypokinesis.
Transthoracic Echocardiogram at Presentation Apical View. Two-dimensional echocardiogram in apical 4-chamber view showing severe left ventricular hypokinesis.
Transthoracic Echocardiogram at Discharge Parasternal View. Two-dimensional echocardiogram in off axis apical 4-chamber view showing AST = normal left ventricular systolic function.
Cardiovascular Magnetic Resonance Cine Short Axis During Hospitalization. Basal short-axis cine postcontrast showing quantitative LVEF = 45%, quantitative RVEF = 33%, and contrast enhancement.
Cardiovascular Magnetic Resonance Cine Short Axis at 3-Month Follow-up. Basal short-axis cine postcontrast showing improvement in cardiac function (quantitative LVEF 58%, quantitative RVEF = 46%) and contrast enhancement.
Transthoracic Echocardiogram at Discharge Parasternal View. Two-dimensional echocardiogram in parasternal long-axis view showing normal left ventricular systolic function.
References
- 1.Tschöpe C., Ammirati E., Bozkurt B., et al. Myocarditis and inflammatory cardiomyopathy: current evidence and future directions. Nat Rev Cardiol. 2021;18(3):169–193. doi: 10.1038/s41569-020-00435-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Trachtenberg B.H., Hare J.M. Inflammatory cardiomyopathic syndromes. Circ Res. 2017;121(7):803–818. doi: 10.1161/CIRCRESAHA.117.310221. [DOI] [PubMed] [Google Scholar]
- 3.Kociol R.D., Cooper L.T., Fang J.C., et al. Recognition and initial management of fulminant myocarditis: a scientific statement from the American Heart Association. Circulation. 2020;141(6):e69–e92. doi: 10.1161/CIR.0000000000000745. [DOI] [PubMed] [Google Scholar]
- 4.Ammirati E., Cipriani M., Moro C., et al. Clinical presentation and outcome in a contemporary cohort of patients with acute myocarditis: Multicenter Lombardy Registry. Circulation. 2018;138(11):1088–1099. doi: 10.1161/CIRCULATIONAHA.118.035319. [DOI] [PubMed] [Google Scholar]
- 5.Subramaniam A.V., Barsness G.W., Vallabhajosyula S. Complications of temporary percutaneous mechanical circulatory support for cardiogenic shock: an appraisal of contemporary literature. Cardiol Ther. 2019;8(2):211–228. doi: 10.1007/s40119-019-00152-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.van Diepen S., Katz J.N., Albert N.M., et al. Contemporary management of cardiogenic shock: a scientific statement from the American Heart Association. Circulation. 2017;136(16):e232–e268. doi: 10.1161/CIR.0000000000000525. [DOI] [PubMed] [Google Scholar]
- 7.Skouri H.N., Dec G.W., Friedrich M.G., Cooper L.T. Noninvasive imaging in myocarditis. J Am Coll Cardiol. 2006;48(10):2085–2093. doi: 10.1016/j.jacc.2006.08.017. [DOI] [PubMed] [Google Scholar]
- 8.Xie Y., Xu E., Bowe B., Al-Aly Z. Long-term cardiovascular outcomes of COVID-19. Nat Med. 2022 doi: 10.1038/s41591-022-01689-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.McDonagh T.A., Metra M., Adamo M., et al. 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: developed by the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure of the European Society of Cardiology (ESC) Eur J Heart Fail. 2022;24(1):4–131. doi: 10.1002/ejhf.2333. [DOI] [PubMed] [Google Scholar]
- 10.Tschöpe C., Cooper L.T., Torre-Amione G., Van Linthout S. Management of myocarditis-related cardiomyopathy in adults. Circ Res. 2019;124(11):1568–1583. doi: 10.1161/CIRCRESAHA.118.313578. [DOI] [PubMed] [Google Scholar]
- 11.Ammirati E., Frigerio M., Adler E.D., et al. Management of acute myocarditis and chronic inflammatory cardiomyopathy: an expert consensus document. Circ Heart Fail. 2020;13(11) doi: 10.1161/CIRCHEARTFAILURE.120.007405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ferreira V.M., Schulz-Menger J., Holmvang G., et al. Cardiovascular magnetic resonance in nonischemic myocardial inflammation: expert recommendations. J Am Coll Cardiol. 2018;72(24):3158–3176. doi: 10.1016/j.jacc.2018.09.072. [DOI] [PubMed] [Google Scholar]
- 13.Messroghli D.R., Moon J.C., Ferreira V.M., et al. Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2∗ and extracellular volume: a consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imaging (EACVI) J Cardiovasc Magn Reson. 2017;19(1):75. doi: 10.1186/s12968-017-0389-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Friedrich M.G., Sechtem U., Schulz-Menger J., et al. Cardiovascular magnetic resonance in myocarditis: a JACC white paper. J Am Coll Cardiol. 2009;53(17):1475–1487. doi: 10.1016/j.jacc.2009.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yang F., Wang J., Li W., et al. The prognostic value of late gadolinium enhancement in myocarditis and clinically suspected myocarditis: systematic review and meta-analysis. Eur Radiol. 2020;30(5):2616–2626. doi: 10.1007/s00330-019-06643-5. [DOI] [PubMed] [Google Scholar]
- 16.Baughman K.L. Diagnosis of myocarditis: death of Dallas criteria. Circulation. 2006;113(4):593–595. doi: 10.1161/CIRCULATIONAHA.105.589663. [DOI] [PubMed] [Google Scholar]
- 17.Peretto G., Sala S., Rizzo S., et al. Arrhythmias in myocarditis: state of the art. Heart Rhythm. 2019;16(5):793–801. doi: 10.1016/j.hrthm.2018.11.024. [DOI] [PubMed] [Google Scholar]
- 18.Maron B.J., Udelson J.E., Bonow R.O., et al. Eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities: task force 3: hypertrophic cardiomyopathy, arrhythmogenic right ventricular cardiomyopathy and other cardiomyopathies, and myocarditis: a scientific statement from the American Heart Association and American College of Cardiology. Circulation. 2015;132(22):e273–e280. doi: 10.1161/CIR.0000000000000239. [DOI] [PubMed] [Google Scholar]
- 19.Naneishvili T., Khalil A., O'Leary R., Prasad N. Fulminant myocarditis as an early presentation of SARS-CoV-2. BMJ Case Rep. 2020;13(9) doi: 10.1136/bcr-2020-237553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guzik T.J., Mohiddin S.A., Dimarco A., et al. COVID-19 and the cardiovascular system: implications for risk assessment, diagnosis, and treatment options. Cardiovasc Res. 2020;116(10):1666–1687. doi: 10.1093/cvr/cvaa106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Halushka M.K., Vander Heide R.S. Myocarditis is rare in COVID-19 autopsies: cardiovascular findings across 277 postmortem examinations. Cardiovasc Pathol. 2021;50:107300. doi: 10.1016/j.carpath.2020.107300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rink L.D., Daniels C.J., Boersma D., et al. Competitive sports, the coronavirus disease 2019 pandemic, and big ten athletics. Circ Cardiovasc Qual Outcomes. 2020;13(12) doi: 10.1161/CIRCOUTCOMES.120.007608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Clark D.E., Parikh A., Dendy J.M., et al. COVID-19 Myocardial Pathology Evaluation in AthleTEs with Cardiac Magnetic Resonance (COMPETE CMR) Circulation. 2021;143(6):609–612. doi: 10.1161/CIRCULATIONAHA.120.052573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gluckman T.J., Bhave N.M., Allen L.A., et al. 2022 ACC expert consensus decision pathway on cardiovascular sequelae of COVID-19 in adults: myocarditis and other myocardial involvement, post-acute sequelae of SARS-CoV-2 infection, and return to play. J Am Coll Cardiol. 2022;79(17):1717–1756. doi: 10.1016/j.jacc.2022.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Transthoracic Echocardiogram at Presentation Parasternal View. Two-dimensional echocardiogram in parasternal long-axis view showing left ventricular hypertrophy and severe left ventricular hypokinesis.
Transthoracic Echocardiogram at Presentation Apical View. Two-dimensional echocardiogram in apical 4-chamber view showing severe left ventricular hypokinesis.
Transthoracic Echocardiogram at Discharge Parasternal View. Two-dimensional echocardiogram in off axis apical 4-chamber view showing AST = normal left ventricular systolic function.
Cardiovascular Magnetic Resonance Cine Short Axis During Hospitalization. Basal short-axis cine postcontrast showing quantitative LVEF = 45%, quantitative RVEF = 33%, and contrast enhancement.
Cardiovascular Magnetic Resonance Cine Short Axis at 3-Month Follow-up. Basal short-axis cine postcontrast showing improvement in cardiac function (quantitative LVEF 58%, quantitative RVEF = 46%) and contrast enhancement.
Transthoracic Echocardiogram at Discharge Parasternal View. Two-dimensional echocardiogram in parasternal long-axis view showing normal left ventricular systolic function.