Abstract
Wu Wei Zi is the dried fruit of Schisandra chinensis (Turcz.) Baill. or Schisandra sphenanthera Rehd. et Wils. (family Magnoliaceae). As a homology of medicine and food, it has been widely used in China for thousands of years, to tonify the kidney, and ameliorate neurological, cardiovascular, liver, and gastrointestinal disorders. As its increasing health benefits and pharmacological value, many literatures have reported that the combination of Wu Wei Zi in patients has led to fluctuations in the blood level of the combined drug. Therefore, it is extremely important to evaluate its safety concern such as drug-drug interactions (DDIs) when patients are under the poly-therapeutic conditions. This review summarized the effects of Wu Wei Zi extract and its major lignan components on cytochrome P450 and P-glycoprotein activities, the change of which could induce metabolic DDIs. Our review also elaborated on the differences of the major lignan components of the two Schisandra species, as well as the absorption, distribution, metabolism, and elimination of the major lignans. In conclusion, these results would enhance our understanding of the DDI mechanisms involving Wu Wei Zi, and may potentially untangle some differing and conflicting results in the future.
Keywords: Schisandra sphenanthera, Schisandraceae, lignans, drug-drug interactions, ADME, Schisandra chinensis
Introduction
“Wu Wei Zi” refers to the ripe fruits of two plants in the genus Schisandra, S. chinensis (Turcz.) Baill. (North Wu Wei Zi) or S. sphenanthera Rehd. et Wils (South Wu Wei Zi). It has been long used as nutritional and fatigue-fighting food supplement in China, Japan, Korea, and Russia. Records suggest that Russian hunters would eat Wu Wei Zi to alleviate fatigue, improve night vision, and replenish body fluids, while Ainu used Wu Wei Zi to protect against seasickness and cold weather (Panossian and Wikman, 2008). Both North Wu Wei Zi and South Wu Wei Zi were described with functions of astringency, reinforcing qi to generate body fluids, and tonifying the kidney to calm the mind (Chinese Pharmacopoeia Commission, 2020), along with hepatoprotective and anti-diabetic activities (Mu et al., 2006; Liu et al., 2017). Additionally, the two species also host unique properties. For instance, South Wu Wei Zi is more suitable for the treatment against chronic cough, mild breathlessness, and skin inflammation (Huyke et al., 2007), while North Wu Wei Zi has better cardiovascular-protective, neuroprotective, and tonic activities (Panossian and Wikman, 2008; Chun et al., 2014; Chen et al., 2015). More than 100 traditional Chinese medicine prescriptions composed of North Wu Wei Zi and South Wu Wei Zi are documented in the Pharmacopoeia of the People’s Republic of China (Ali et al., 2018), with various pharmacological activities. With their increasing consumption, it is extremely important to evaluate the safety concern such as drug-drug interactions (DDIs) when patients are under the poly-therapeutic conditions.
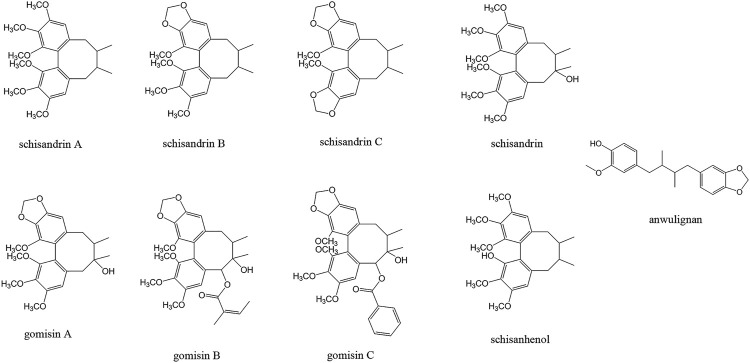
Recent studies revealed that both North Wu Wei Zi and South Wu Wei Zi are enriched in dibenzocyclooctadiene lignan components (Schisandra lignans, Figure 1), which are responsible for the major bioactivities of the two species (Sowndhararajan et al., 2018). However, the schisandra lignans are found to interfere with cytochrome P450 isoenzymes (CYPs) and P-glycoprotein (P-gp) activities, thus affecting the absorption, transportation, or metabolism of these or other drugs (Zhou et al., 2005; Qin et al., 2014a; Wang Cai-Ping et al., 2014), resulting metabolic DDIs. Depending on different medication circumstances, the “clinically relevant” DDI could lead to risk of adverse events or could be an advantageous for the therapy appropriateness processes (Palleria et al., 2020).
FIGURE 1.
Structure of the major lignan components of Wu Wei Zi.
In recent years, the researchers paid more attention to the review of the botany, phytochemistry, chemical composition, in-depth in vivo pharmacological study, and clinical application of North Wu Wei Zi or South Wu Wei Zi (Szopa et al., 2017; Li et al., 2018; Huang et al., 2021), and only one review covered both North Wu Wei Zi and South Wu Wei Zi (Yang et al., 2022). However, little information was presented about their metabolic DDIs. In this review, we mainly focus on the main components of North Wu Wei Zi and South Wu Wei Zi, and identified CYP- and P-gp-interfering effects of the schisandra lignans, in order to better understand the quality control and potential DDIs of Wu Wei Zi products. By creating a full picture of the potential impacts of Wu Wei Zi, this review might facilitate investigations on DDIs for new chemical products and traditional Chinese medicines.
Literature Search Strategy
Literature research was conducted in May 2021 for articles published from 2000 to 2021, without restriction to regions and publication type. Eligible literatures for inclusion are written in English or Chinese. Electronic databases such as PubMed, Web of Science, ScienceDirect, Springer, Wiley, China National Knowledge Infrastructure (CNKI) and WANFANG Data were searched by two authors independently. Detailed PubMed strategy was listed as follows: key words were composed of Wu Wei Zi/Schisandra chinensis/Schisandra sphenanthera/schisandra lignans and contents/pharmacokinetics/DDI/CYP/P-gp. Articles dealing with the geographical, history, botany, ethnomedicine were excluded. Most articles were from PubMed (85 articles), supplemented by Science Direct (2 articles), Web of Science (2 articles), CNKI (26 articles, 6 dissertations), WANFANG Data (4 articles).
The Main Lignan Components of North Wu Wei Zi and South Wu Wei Zi
Schisandra lignans have a basic dibenzocyclooctadiene structure (Figure 1). They showed different contents in North Wu Wei Zi and South North Wu Wei Zi. The quantity and quality determinations of schisandra lignans to date are based on high-performance liquid chromatography (HPLC) methods such as HPLC-ultraviolet (UV), HPLC-capillary electrophoresis (CE), and HPLC-mass spectrometry (MS), or on gas chromatography-mass spectrometry (GC-MS) (Table 1). HPLC-UV has become the predominant method used to study Wu Wei Zi as there is a strong UV absorbance of schisandra lignans between 230 and 255 nm, resulting in high sensitivity and specificity (Zhang Hai et al., 2009).
TABLE 1.
Contents of major schisandra lignans of Wu Wei Zi.
| Type | Schisandrin A (mg/g) | Schisandrin B (mg/g) | Schisandrin C (mg/g) | Schisandrin (mg/g) | Gomisin A (mg/g) | Gomisin B (mg/g) | Gomisin C (mg/g) | Schisanhenol (mg/g) | Anwulignan (mg/g) | Det. | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Synonyms | deoxyschisandrin | γ-schisandrin | — | schisandrol A | schisandrol B | schisantherin B | schisantherin A | — | macelignan | — | — |
| Abbr. | Sch A | Sch B | Sch C | SCH | Gom A | Gom B | Gom C | — | — | — | — |
| North | — | 8.00∼23.50 | — | 22.40∼37.50 | 7.40∼12.80 | — | — | — | — | UV | Hu et al. (2014) |
| North | 0.77∼2.40 | 0.57∼2.31 | 0.08∼1.88 | 2.13∼14.02 | 0.36∼6.14 | — | 0.38∼2.48 | — | — | MS | Deng et al. (2008) |
| North | 0.26∼2.83 | 0.34∼2.05 | 0.03∼0.65 | 1.29∼11.32 | 2.10∼32.96 | — | — | — | — | FLD | Xia et al. (2014a) |
| North | 0.45∼2.92 | 0.08∼0.90 | 0.05∼0.97 | 3.35∼10.59 | 0.75∼3.72 | — | — | — | — | GC-MS | Xia et al. (2014b) |
| North | 0.30∼3.10 | 0.10∼4.60 | 2.40 | 0.10∼9.50 | — | — | 1.40∼7.80 | — | — | UV | Geng and Yang, (2007) |
| North | 0.90∼1.60 | 2.10∼4.40 | 0.20∼1.00 | 5.30∼7.60 | 1.30∼3.40 | — | 0.20∼0.40 | — | — | UV | Wang et al. (2015) |
| North | 0.97∼1.78 | 2.06∼3.76 | 0.27∼0.83 | 3.93∼6.81 | 1.40∼2.59 | — | 0.20∼0.44 | — | — | UV | Dou et al. (2014) |
| North | 0.60∼0.80 | — | 2.20∼2.90 | 5.70∼6.40 | 2.00∼2.90 | 0.30∼0.60 | 0.04∼0.08 | — | — | UV | Avula et al. (2005) |
| North | 0.66∼1.44 | 0.51∼0.81 | 0.24∼1.27 | 3.73∼6.37 | 1.32∼2.61 | — | — | — | — | UV | Lee and Kim, (2010) |
| North | 1.10 | 3.20 | — | 6.00 | — | — | — | — | — | UV | Tian et al. (2007) |
| North | 0.60∼2.25 | 0.09∼4.52 | 0.12∼0.72 | 0.13∼5.53 | 0.07∼3.11 | — | 0.83∼6.14 | — | — | UV | Zhang Hai et al. (2009) |
| North | 0.002∼0.235 | 0.02∼1.79 | — | 0.47∼5.44 | 0.17∼1.71 | — | — | — | — | UV | Halstead et al. (2007) |
| North | — | 2.50 | — | 5.00 | — | — | — | — | — | UV | Liu et al. (2015) |
| North | 1.60∼4.00 | — | — | 0.10∼0.70 | — | — | 0.30∼3.00 | — | — | UV | Zhu et al. (2007) |
| South | 3.00∼7.30 | 0.10∼0.80 | 0.10∼0.90 | 0.10∼0.50 | 0.10∼0.90 | — | 2.90∼8.30 | — | 2.10∼5.20 | UV | Gao et al. (2003) |
| South | — | — | — | — | — | — | 5.50∼7.70 | — | — | UV | Wang et al. (2017) |
| South | — | — | — | — | — | — | 5.38∼5.81 | — | — | Gu et al. (2008) | |
| South | 1.54∼6.63 | <0.21 | 0.03∼0.89 | <0.76 | <0.51 | — | 0.80∼5.66 | 0.01∼1.48 | 1.57∼3.82 | UV | Wei et al. (2010) |
| South | 1.95∼4.63 | <0.15 | <0.23 | <0.16 | <0.42 | — | 0.37∼3.13 | 0.11∼0.53 | 1.02∼2.21 | UV | Yuan et al. (2011) |
| South | — | — | — | — | — | — | 0.15∼1.71 | 0.11∼8.53 | 0.33∼1.35 | MS | Yang et al. (2014) |
| South | 1.90 | 2.30 | 5.90 | 4.80 | 2.20 | — | — | — | — | MS | Song, (2004) |
The content results of North Wu Wei Zi (S. chinensis) were listed in descending order according to the maximum value of schisandrin (SCH) content in each literature.
The content results of South Wu Wei Zi (S. sphenanthera) were listed in descending order according to the maximum value of gomisin C (Gom C) content in each literature.
Det. is short for detection method. Abbr. is short for abbreviation.
The major schisandra lignans in North Wu Wei Zi were found to be schisandrin B (Sch B), schisandrin (SCH) and gomisin A (Gom A) (Ren and Zhang, 2020), while South Wu Wei Zi contains higher levels of anwulignan, schisandrin A (Sch A) and gomisin C (Gom C) (Sun et al., 2014). As such, SCH and Gom C are defined as the quality markers of North Wu Wei Zi and South Wu Wei Zi, respectively, according to the Chinese pharmacopoeia, where the SCH content is required to be above 0.40% (North Wu Wei Zi), and Gom C content should be higher than above 0.20% (South Wu Wei Zi).
In addition to species, the place of origin and associated climatic conditions could also affect the lignan contents. It was found that North Wu Wei Zi has a higher enrichment with schisandra lignans when collected from Zhongtiao Mountain, south and east of Qinling mountain, and south of Taihang mountain than from other locations. Similarly, the content of Gom C decreases as rainfall increases, and anwulignan content was reduced by higher maximum temperatures (Sun et al., 2014). It has also been reported that cultivated North Wu Wei Zi that were planted in a north-south orientation had higher lignan accumulation, with SCH, Sch A, and Sch B content in north-south planted North Wu Wei Zi 10.2, 25.8, and 26.8% higher than in those planted in an east-west orientation (Yang, 2011).
The “ADME” of the Primary Schisandra Lignans
Absorption
Pharmacokinetic studies have found that the lignans of Wu Wei Zi are readily absorbed into the bloodstream after oral ingestion. Following intragastric (i.g.) administration of Wuzhi capsule (a Chinese patent medicine composed of the ethanol extract of South Wu Wei Zi), Sch A, SCH, Gom A, Gom C, and schisanhenol were rapidly absorbed into the blood and were subsequently metabolized fully before elimination (Wei et al., 2010b; Wei et al., 2014; Wang et al., 2016).
Previous studies showed that SCH, the main lignan in North Wu Wei Zi, could be quickly absorpted after oral administration, with Tmax less lower than 1 h (Wang et al., 2003; Yan et al., 2006). Further study revealed that it is primarily absorbed in the duodenum and jejunum, followed by the colon and rectum, with absorption percentage of 37.4, 36.1, 24.9 and 31.4%, respectively (Zhang W et al., 2009). In contrast, Sch A and Sch B showed relative slower absorption. For example, the Tmax of Sch A was found to be between 2 and 4 h after i.g. of South Wu Wei Zi ethanol extract oil in rats (Wei et al., 2010a; Li H. L. et al., 2013), and a secondary absorption could be observed around 8 h (Li WL. et al., 2013). Sch B exhibited the first plasma concentration peak at 5.5–6.0 h, closely followed by a second absorption concentration peak 0.5 h after the first peak. Currently available data showed that the absorption rates are much faster in mice compared with rats, with observed secondary absorption of Sch C, Gom A, Gom B, and Gom C appearing at 8–25 h after North Wu Wei Zi extract administration. Meanwhile, the secondary Cmax was often lower that the primary one (Wei Hua et al., 2013; Liu et al., 2017). It is suggested that the secondary peak may be caused either by enterohepatic circulation or by the metabolic transformation from other schisandra lignans.
The majority of the schisandra lignans could change their absorption characteristics when Wu Wei Zi was administrated along with other medical materials. For example, the absorption rate of SCH decreased slightly (Tmax 0.32∼1.00 h) when North Wu Wei Zi was administrated in the prescription “Shengmai Powder” which also contains Radix Ophipogonis and Radix et Rhizoma Ginseng (Wang et al., 2003; Li et al., 2005; Guo et al., 2012). Moreover, SCH showed a significantly decreased Tmax at 7.0 h when administrated in the prescription “Longlingchun mixture”, which is composed of North Wu Wei Zi and other ten Chinese medicines (Zhao et al., 2006).
Distribution
After absorption, or after tail vein injection, the highest concentrations of the main schisandra lignans (such as SCH, Sch A, and Sch B) were presented in the lungs and liver, followed by the heart, kidneys, and lastly the spleen (Zhou et al., 2013). Sch A and SCH were also both found in rat brain at a very low concentration level, suggesting that they were able to cross the blood-brain barrier (Hu, 2012). Within the different rat brain regions, SCH showed the highest concentrations in the hypothalamus, striatum, and hippocampus, followed by the brain stem, and the lowest concentration of which was found in the cerebral cortex and cerebellum. The high distribution in hypothalamus, striatum, and hippocampus may explain the neuro-protective effects of SCH and North Wu Wei Zi on mice with Alzheimer’s disease and depression (Wang et al., 2013; Wang Baolian et al., 2014).
Metabolism
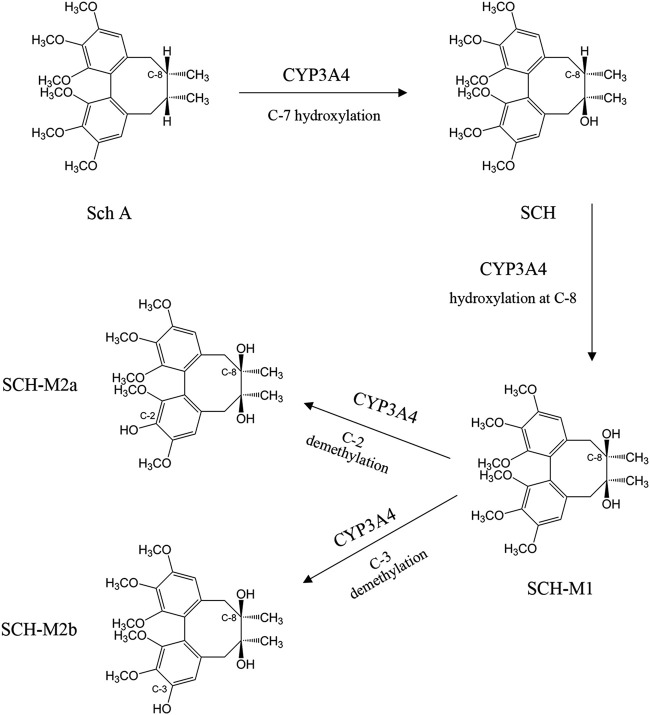
The major phase I metabolism pathways of schisandra lignans are demethylation and hydroxylation (Zhang et al., 2014; Zhang et al., 2017a), including a total of 33 metabolites characterized from six lignans following administration of a South Wu Wei Zi preparation. In detail, the enzyme CYP3A4 catalyzes Sch A to SCH in the first step (Cao, 2010). Secondly, SCH formed SCH-M1, mediated by CYP3A4 again at hydroxylation of C8 position, and then biotransformed by demethylation of C2 position (SCH-M2a) or C3 position (SCH-M2b) (Figure 2). The production of SCH-M2b was characterizes two-thirds of that of SCH-M2a in the rat liver microsomes study. The main metabolic pathways of Sch B have been revealed as the mono-oxygenation of C4 or C11 position, the demethylation of the methoxy group on C12 position, or demethylation at the methylenedioxy group connected with C2 and C3 positions which opens the five-member ring, followed by glucuronidation on C3 position (Qian et al., 2015).
FIGURE 2.
The metabolism of schisandrin A (Sch A) and schisandrin (SCH).
Elimination
Wu et al. (2014a) reported that the main schisandra lignans SCH, Sch A, and Sch B, were fully eliminated within 25 h in male rats following South Wu Wei Zi extract administration. Basically, schisandra lignans and their metabolites are gradually excreted through feces and urine (Qian et al., 2015).
Wu Wei Zi Induced Drug-Drug Interactions
The Biphasic Effect of Schisandra Lignans on CYPs
DDIs have been implicated in some studies for both North Wu Wei Zi and South Wu Wei Zi. Interestingly, recent studies have identified seemingly contradictory inductive and inhibitory effects of their water or ethanol extracts, and more specifically schisandra lignans, on CYPs and P-gp.
Both Wu et al. (2007a) and Mao and Tay (2016) found that North Wu Wei Zi extract inhibited CYP3A in human liver microsomes (HLMs; in vitro) and in healthy volunteers (in vivo). In contrast, Hu (2011) reported that North Wu Wei Zi extract increased CYP450 contents in Kunming mice after 7-day-oral administration. The bi-directional effect on CYPs was first identified by Cheng et al., and it was found that CYP3A activity in rats was inhibited within 12 h after North Wu Wei Zi water extract administration yet was increased after 6-day-oral administration. The phenomenon was further discussed by Chen et al. (2010), suggesting that the bi-directional effect of North Wu Wei Zi extracts on CYPs depend on the pre- or co-treatment time period. The inhibition effect could be observed immediately after administration, while the induction effect could only be monitored till the episode of DNA transcription and protein synthesis. Besides, a few schisandra lignans showed contradictory effects on CYP activities when involving individually in different experimental conditions. For example, Sch B was identified as a CYP inducer when administrated to mice in vivo (Liu, 1988), yet could inhibit CYP3A activity when incubated with rat liver microsomes in vitro (Li et al., 2010). In general, both North Wu Wei Zi and South Wu Wei Zi extracts, Sch A, Sch B, and SCH are recognized as activators when administered in vivo with long-term administration (>6 days), and as inhibitors when incubated with liver microsomes or CYP isoforms in vitro (Tables 2, 3).
TABLE 2.
Biphasic effect on regulating CYP activities by Wu Wei Zi extract.
| Extract | in vivo/in vitro a | Bi-directional effect on CYPs b | IC50 (μg/ml) | Dosage/Application | CYP Substrate d | Note | Ref. |
|---|---|---|---|---|---|---|---|
| North Wu Wei Zi water extract | in vitro, RLM | CYP1A2 -47% | — | 500 μg/ml | PNT-CYP1A2 | — | Wang et al. (2011) |
| CYP2C6 -53% | DFS-CYP2C6 | ||||||
| CYP2C11 -48% | MPT-CYP2C11 | ||||||
| CYP2D2 -40% | DTM-CYP2D2 | ||||||
| CYP2E1 -57% | CLZ-CYP2E1 | ||||||
| CYP3A-70% | MDZ-CYP3A | ||||||
| in vitro, RLM | CYP3A- | 487.8 | — | TST | — | Chen et al. (2010) | |
| in vitro, RH | CYP3A+55% | — | 2.5 mg/ml, 3 days | TST | — | Chen et al. (2010) | |
| in vivo, rat | CYP2D2—CYP2E1 + | — | 1.5 g/kg/d, 7 days | DTM-CYP2D2 | — | Wang et al. (2011) | |
| EDH c : 13.1 g/d | CLZ-CYP2E1 | ||||||
| in vivo, rat | CYP3A-51∼60% | — | 1∼3 g/kg/d, 1 day | TST | — | Chen et al. (2010) | |
| EDH d : 8.6∼25.7 g/d | |||||||
| in vivo, rat | CYP3A+36∼58% | — | 1∼3 g/kg/d, 3 days | TST | — | ||
| EDH c : 8.6∼25.7 g/d | |||||||
| in vivo, rat | total CYP + | — | 3 g/kg/d, 1 day, tested after 6 days | — | mRNA transcription + | Mu et al. (2006) | |
| EDH c , 18.9 g/d | |||||||
| North Wu Wei Zi ethanol extract | in vitro, RLM | CYP1A2 -80% | — | 120 μg/ml | PNT-CYP1A2 | — | Wang et al. (2011) |
| CYP2C6 -90% | DFS-CYP2C6 | ||||||
| CYP2C11–90% | MPT-CYP2C11 | ||||||
| CYP2D2 -80% | DTM-CYP2D2 | ||||||
| CYP2E1 -50% | CLZ-CYP2E1 | ||||||
| CYP3A-90% | MDZ-CYP3A | ||||||
| in vitro, HH | CYP3A4 + | — | — | — | mRNA transcription + | Mu et al. (2006) | |
| CYP2C9 + | |||||||
| in vitro, MH | CYP3A+ | — | — | — | mRNA transcription + | ||
| CYP2C29 + | |||||||
| in vivo, rat | CYP2D2 – | — | 1.5 g/kg/d, 7 days | MPT-CYP2C11 | — | Wang et al. (2011) | |
| CYP2C11– | DTM-CYP2D2 | ||||||
| CYP2E1 + | EDH c : 13.1 g/d | CLZ-CYP2E1 | |||||
| CYP3A+ | MDZ-CYP3A | ||||||
| Schisandra lignan extract of South Wu Wei Zi | in vitro, RLM | CYP3A- | 40.0 without preincubation | — | MDZ | Mechanism-based inhibition suggested | Lai et al. (2009) |
| 35.0 with preincubation | |||||||
| in vitro, RIM | CYP3A- | 25 without preincubation | — | MDZ | Mechanism-based inhibition suggested | ||
| 6.3 with preincubation | |||||||
| in vivo, rat | CYP3A+ | — | 0.15 g/kg/d, 14 days | MDZ | — | ||
| EDH d : 1.3 g/d | |||||||
| South Wu Wei Zi ethanol extract | in vivo, rat | CYP3A- | — | 0.25 g/kg/d, 1 day | paclitaxel | blood exposure +1.9 fold | Jin et al. (2011) |
| EDH c : 2.1 g/d |
In vivo/in vitro: HH: human hepatocytes; HLM: human liver microsomes; RH: rat hepatocytes; RLM: rat liver microsomes; RIM: rat intestinal microsomes; MH: mouse hepatocytes.
Bi-directional effect on CYPs: −, inhibition; +, induction.
EDH: equivalent dosage for human.
CYP probe substrates: phenacetin (PNT), diclofenac sodium (DFS), mephenytoin (MPT), dextromethorphan (DTM), chlorzoxazone (CLZ), midazolam (MDZ), and testosterone (TST).
TABLE 3.
Inhibitory of main CYP450 isoform activity by the major schisandra lignans.
| Component (abbreviation) | in vivo/in vitro | Enzyme source a | CYP enzyme | Substrate b | IC50 (μM) | K i (μM) | K I (μM) | k inact (min−1) | Inhibition type | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| Schisandrin A (Sch A) | in vitro | HLM | CYP3A4 | TST | — | 1.51 | — | — | Reversible, competitive | Zhang et al. (2017b) |
| in vitro | HLM | CYP3A4 | TST | — | 74.1 ± 10.1 | — | — | Mixed | Wu et al. (2014) | |
| in vitro | RLM | CYP3A | MDZ | 70 | 45 | — | — | — | Lai et al. (2009) | |
| in vitro | RLM | CYP3A | MDZ | 12.5 | 4.8 | — | — | — | Lai et al. (2009) | |
| in vitro | HLM | CYP2C19 | OME | 2.77 | — | — | — | — | Wei (2010) | |
| in vivo | rat | CYP3A | MDZ | — | 30.67 mg/kg | — | — | Reversible, non-competitive | Li Wei-Liang et al. (2012) | |
| in vivo | rat | CYP3A | MDZ | 12.5 ± 2.5 | 30.67 | — | — | — | Li Ruidong et al. (2012) | |
| Schisandrin B (Sch B) | in vitro | RLM | CYP3A | MDZ | — | 4.24 | — | — | Reversible, non-competitive | Li et al. (2010) |
| in vitro | RLM | CYP3A | MDZ | 6.25 | 5 | — | — | — | Lai et al. (2009) | |
| in vivo | rat | CYP3A | MDZ | — | 16.64 mg/kg | — | — | Reversible, non-competitive | Li WL. et al. (2013) | |
| in vitro | HLM | CYP3A | MDZ, NIF, TST | 1.3–4.5 | — | — | — | — | Seo et al. (2021) | |
| in vitro | HLM | CYP2C19 | DTM | 10.4 | — | — | — | — | Seo et al. (2021) | |
| in vitro | HLM | CYP2E1 | CZX | >50 | — | — | — | — | Seo et al. (2021) | |
| Schisandrin C (Sch C) | in vitro | HLM | CYP3A | OME | 4.01 | — | — | — | — | Wei, (2010) |
| in vitro | HLM | CYP2C19 | DTM | 2.7 | — | — | — | — | Seo et al. (2021) | |
| in vitro | HLM | CYP2E1 | CZX | >50 | Seo et al. (2021) | |||||
| Schisandrin (SCH) | in vitro | RLM | CYP3A | MDZ | 70 | 45 | — | — | — | Lai et al. (2009) |
| in vitro | HLM | CYP2C19 | DTM | 5.3 | — | — | — | — | Seo et al. (2021) | |
| in vitro | HLM | CYP2E1 | CZX | 4.2 | Seo et al. (2021) | |||||
| Gomisin A (Gom A) | in vitro | RLM | CYP3A | TST | 0.35 | 1.96 | irreversible | Zhai et al. (2017) | ||
| in vitro | HLM | CYP3A | ERY | 10.1 | — | — | — | — | Iwata et al. (2004) | |
| in vitro | HLM | CYP3A | TST | 6.71 | — | — | — | — | Iwata et al. (2004) | |
| in vitro | HLM | CYP3A | MDZ | 2.40 | — | — | — | — | Wei (2010) | |
| in vitro | HLM | CYP3A | MDZ, NIF, TST | 1.8–2.3 | — | — | — | — | Seo et al. (2021) | |
| in vitro | HLM | CYP2C9 | TBT | 3.44 | — | — | — | — | Wei (2010) | |
| in vitro | HLM | CYP2C19 | OME | 5.91 | — | — | — | — | Wei (2010) | |
| in vitro | HLM | CYP2C19 | DTM | 11.2 | — | — | — | reversible | Seo et al. (2021) | |
| in vitro | HLM | CYP2D6 | DTM | 8.58 | — | — | — | — | Wei, (2010) | |
| in vitro | HLM | CYP2E1 | CZX | >50 | — | — | — | reversible | Seo et al. (2021) | |
| Gomisin B (Gom B) | in vitro | HLM | CYP3A | ERY | 0.399 | 0.131 | — | — | — | Iwata et al. (2004) |
| in vitro | HLM | CYP3A | TST | 0.624 | — | — | — | — | Iwata et al. (2004) | |
| in vitro | HLM | CYP3A | MDZ, NIF, TST | 0.28–0.42 | — | — | — | — | Seo et al. (2021) | |
| in vitro | HLM | CYP2C19 | DTM | >50 | — | — | — | — | Seo et al. (2021) | |
| in vitro | HLM | CYP2E1 | CZX | >50 | — | — | — | — | Seo et al. (2021) | |
| Gomisin C (Gom C) | in vitro | HLM | CYP3A | ERY | 0.254 | 0.049 | 0.399 | 0.092 | irreversible | Iwata et al. (2004) |
| in vitro | HLM | CYP3A | TST | 0.257 | — | — | — | — | Iwata et al. (2004) | |
| in vitro | RLM | CYP3A | MDZ | 0.3 | 0.06 | — | — | — | Lai et al. (2009) | |
| in vitro | HLM | CYP3A | MDZ, NIF, TST | 0.19–0.30 | — | — | — | — | Seo et al. (2021) | |
| in vitro | HLM | CYP2C19 | DTM | 16.3 | — | — | — | — | Seo et al. (2021) | |
| in vitro | HLM | CYP2E1 | CZX | >50 | — | — | — | — | Seo et al. (2021) |
RH: rat hepatocytes; HH, human hepatocytes; RLM, rat liver microsomes; HLM, humanliver microsomes.
CYP probe substratesb: testosterone (TST), midazolam (MDZ), omeprazole (OME), nifedipine (NIF), tolbutamide (TBT), dextromethorphan (DTM), and erythromycin (ERY), chlorzoxazone (CZX).
Zhai et al. (2015) refined the theory, suggesting that Wu Wei Zi extracts or their major schisandra lignans exhibit inhibitory or inductive effect on CYPs activities depending on the experimental conditions. Specifically, it is suspected that the main schisandra lignans could induce CYP DNA expression and inhibit CYP protein activity at the same time; however, the CYP inhibition would be observed immediately, explaining CYP-inhibitory effects were only seen in in vitro experiments when using liver microsomes or CYP isoforms, which did not rely on DNA transcription and translation. On the other hand, the lignan-induced CYP expression could only occur after DNA transcription and translation had taken place, a process of which apparently could take 5 days or more in vivo. The modified theory has been supported by other studies. For example, North Wu Wei Zi water extract inhibited CYP3A activity with single-dose administration and induced CYP3A activity with multiple administration in rats (1–3 g/kg, qd for 3 days) (Chen et al., 2010) (Table 2). This study also showed that the CYP3A induction was caused by an increase of mRNA transcription with Sch B and Gom A.
The inhibition or induction of Wu Wei Zi and its major schisandra lignans is summarized in Tables 2–4. For ease of comparison, the dose of Wu Wei Zi in the in vivo studies were converted into human dosage according to an average body weight of 54 kg.
The Inhibitory Effects of Wu Wei Zi and its Major Lignans on CYP Enzymes
Wu Wei Zi extracts were found with extensive inhibitory effects on CYPs (Table 2). CYP inhibition can generally be classified as reversible or irreversible interactions (Berg et al., 2012), with reversible CYP inhibitions further divided into competitive, non-competitive, and un-competitive interactions according to the interaction between inhibitors and CYP enzymes. For irreversible inhibition, inhibitors are first catalyzed by CYPs into reactive intermediates which then form metabolite-inhibitor complexes with CYP that causes deactivation. As a result, irreversible inhibitions are time-dependent and NADPH-dependent process when the inhibitors exposed to the CYP. This phenomenon could be reinforced by multiple dosing of the inhibitors in vivo.
Wang et al. (2011) reported that CYP1A2, CYP2D2, CYP2C6, CYP2C11, CYP2E1, and CYP3A activities could be inhibited by North Wu Wei Zi water/ethanol extract in rat liver microsomes (RLMs). The activities of the CYPs were reduced by 50–90% at a concentration of 120 μg/ml when incubated with North Wu Wei Zi ethanol extract, which was recognized as a stronger CYP inhibitor than North Wu Wei Zi water extract. Moreover, the activities of CYP2D2 and CYP2C11 remained inhibited 1 week after North Wu Wei Zi ethanol administration (i.g., 1.5 g/kg). South Wu Wei Zi was also found to be a potent CYP3A inhibitor in rat hepatic or intestinal microsomes, the lignan extract of which showed IC50 values of 25–40 μM. It was further confirmed that Gom C, the most abundant lignan in South Wu Wei Zi, exhibited a strong inhibitory effect in a time-dependent manner, indicating a mechanism-based inhibition (Lai et al., 2009).
Both North and South Wu Wei Zi and their major lignan components have been investigated as CYP inhibitors in DDI studies with clinical-practiced medicines for financial burden reducing, toxicity-alleviating, or pharmacodynamics-enhancing purposes. Take tacrolimus (FK506), a natural immunosuppressant as an example, which is metabolized by CYP3A (Harding et al., 1989; Chen et al., 2020). Its blood concentration can be largely increased by the combination of Wuzhi tablet, a commercially available medicine composed of South Wu Wei Zi ethanol extract, owing to the schisandra lignans’ higher affinity to CYP3A than that of FK506 (Qin et al., 2010a; Qin et al., 2014b; Li et al., 2017; Qin et al., 2020). Thus, Wuzhi tablet is recognized to reduce the oral dosage of FK506 as well as relieve FK506-induced hepatotoxicity, which is similar to Wuzhi capsule (Wei et al., 2014; Wang et al., 2016; Cheng et al., 2021). Many clinical practices have evidenced that both Wuzhi tablet and Wuzhi capsule could effectively increase the blood exposure of FK506 in renal, heart and liver transplanted patients (Jiang et al., 2010; Xin et al., 2011; Liu et al., 2012; Zhou et al., 2017). Similarly, as csyclosporin A was a substrate of CYP3A and P-gp, Wuzhi Tablet could also dramatically increase its Cmax and the blood exposure of by 84.1 and 293.1%, respectively (Xue et al., 2013). What’s more, Wuzhi tablet was also effective in increasing the blood exposure of paclitaxel by 1.9˗fold in rats, while Gom A could effectively increase the Cmax of oral-administered paclitaxel by 2.8˗fold at an oral dose of 25 mg/kg (Jin et al., 2011). Meanwhile, the North Wu Wei ethyl acetate extract was found to block the CYP3A-mediated toxic metabolic pathway of cyclophosphamide, and thus reduce the metabolite related liver, kidney, and brain toxicity in rats (Zhai et al., 2018).
With further investigation, it was found that the CYP-inhibitory efficacy comes from several major lignans of south and north Wu Wei Zi. Zhai et al. have identified Gom A as an efficient CYP3A blocker (K I = 0.35 µM, k inact = 1.96 min−1) (Zhai et al., 2017), while Iwata et al. reported that the CYP3A-inhibiting efficacy of Gom C was found stronger than that of ketoconazole (Iwata et al., 2004). Additionally, other schisandra lignans also take roles as CYP inhibitors (Table 3). For example, Sch A and Sch B have both been found to decrease CYP3A activity with IC50 ranging from 6 to 70 μM (Li Ruidong et al., 2012; Wu et al., 2014), and the inhibition effect was not reversed by activation after 3 days of continuous administration (Li Wei-Liang et al., 2012; Li WL. et al., 2013).
So far, the major schisandra lignans were found working as CYP-inhibitiors with different mechanisms. The in vivo evidences reported that Sch A and B could dose-dependently inhibited hepatic microsomal CYP3A activity as noncompetitive inhibitors (Ki Sch A = 30.67 mg/kg; Ki Sch B = 16.64 mg/kg) (Li Ruidong et al., 2012; Li H. L. et al., 2013). Meanwhile, Gom A was found as a mix-type CYP3A inhibitor, which showed characteristics of competitive as well as time- and NADPH- dependent inhibition (Zhai et al., 2017), while Gom C could irreversibly inactivate CYP3A by forming metabolite-inhibitor complexes (Iwata et al., 2004). It is suspected that the potent inhibition on CYP3A may be caused by the metabolite-intermediate complexes formed by the methylenedioxyphenyl structure with P450 enzyme (Iwata et al., 2004; Lai et al., 2009; Seo et al., 2021). In all, lignans with one methylenedioxyphenyl group such as Gom A, Gom B, Gom C, and Sch B most strongly inhibited CYP3A activity, with IC50 values as low as 0.19–0.28 μM, which was much lower than those of the other P450 isoforms. For these schisandra lignans, some researches obtained larger IC50 values, the discrepancy of which could be due to differences in incubation conditions including CYP3A probe substrates and/or the enzyme source. Among those schisandra lignans, most of them showed potent or moderate inhibition of on CYP2C19, with IC50 values ≤16.3 μM, except Gom B (>50 μM). Sch C with two methylenedioxyphenyl groups showed most inhibitory effect on CYP2C19, with a IC50 value of 2.7 μM. Only SCH, containing none methylenedioxyphenyl group but one hydroxyl group, moderately inhibited CYP2E1(IC50 value 4.2 μM).
The Inductive Effects of Wu Wei Zi and its Major Lignans on CYP Enzyme Activity
Since the late 1960s, both North Wu Wei Zi and South Wu Wei Zi extracts have shown hepatic-protective effects in clinical practice (Zuo et al., 2019). Apart from the alleviation of inflammatory response, it was found that North Wu Wei Zi extract significantly increased the smooth endoplasmic reticulum in hepatocytes of rats after continuous ingestion (≥3 days, qd), which was consistent with the increased liver CYP content and the metabolism acceleration of toxic components in experimental animals (Qiu et al., 2018). More studies found that multiple prolonged administration of Wu Wei Zi extract (≥3 days, qd) could lead to CYP induction, increasing activities of CYP3A, CYP2C, and CYP2E (Chen et al., 2010; Mu et al., 2006) (Table 4), and its administration at high dose could be more likely to cause CYP induction (≥1 g/kg/d for rats, which is equivalent to≥8.6 g/d for human, Table 2). Besides, continuous ingestion of Wu Wei Zi extract at low dose (0.15 g/kg/d for rats, which is equivalent to 1.3 g/d for human) for 2 weeks was also found to cause CYP3A induction in rats (Lai et al., 2009).
TABLE 4.
Induction of CYP3A activity by the major schisandra lignans.
| Component (Synonym) | in vitro/in vivo a | Induction | Substrate b | Dosage/Applicationd | Increased | Ref. |
|---|---|---|---|---|---|---|
| On CYPs | ||||||
| Schisandrin A (deoxyschisandrin) | in vitro, HH | CYP3A4 + | — | — | mRNA transcription | Mu et al. (2006) |
| Schisandrin B (γ-schisandrin) | in vitro, HH | CYP3A4 + | — | — | mRNA transcription | Mu et al. (2006) |
| in vitro, RH | CYP3A+21∼42% | TST | 0.01∼0.1 µM, 3 days incubation | mRNA transcription | Chen et al. (2010) | |
| in vivo, rat | CYP3A | MDZ | — | — | Li H.L. et al. (2013) | |
| Gomisin A (schisandrol B) | in vitro, HH | CYP3A4 + | — | — | mRNA transcription | Mu et al. (2006) |
| in vitro, RH | CYP3A+27% | TST | 10 µM | mRNA transcription | Chen et al. (2010) | |
| in vivo, rat | CYP3A- | paclitaxel | 25 mg/kg/d, 1 day | 2.8-fold blood exposure | Jin et al. (2010) | |
| EDH c : 214 mg/d |
In vitro/in vivo: RH: rat hepatocytes; HH, human hepatocytes.
CYP probe substrates: midazolam (MDZ); testosterone (TST).
EDH: equivalent dosage for human.
Mu et al. (2006) further suggested that the mechanism of CYP induction of Wu Wei Zi extract may be caused by the activation of the pregnane X receptor, with Sch A, Sch B, and Gom B as major responsible agonists. Interestingly, recent investigations revealed that pregnane X receptor might also be the key target for the hepato-protective efficacy of Gom A. By activating the pregnane X receptor, Gom A could accelerate the bile acid metabolism, promote bile acid efflux and induce hepatic expression of Cyp3a in mice (Zeng et al., 2017).
Effects of Wu Wei Zi and Their Major Lignans on P-Glycoprotein Activity
As well as the CYP-dependent metabolism changes, phytochemical-mediated alterations in P-glycoprotein (P-gp) activity may also produce DDIs by altering drug absorption, distribution, and elimination, which could be achieved either by decreasing P-gp expression or by inhibiting P-gp activity. For example, North Wu Wei Zi extract could effectively increase the blood exposure of P-gp substrate taninolol by 47% in healthy volunteers (Fan et al., 2009). A number of studies have shown that Gom A, Gom C, SCH, Sch A, and Sch B could reverse P-gp-mediated multidrug resistance (MDR) and decrease the efflux of P-gp substrate in cancer cells (Wan et al., 2006; Fong et al., 2007; Li et al., 2007; Yoo et al., 2007; Qin et al., 2010b).
Interestingly, a subsequent transport study using Caco-2 cells also found that Gom A, Gom C, Sch A, and SCH were not substrates of P-gp (Qin et al., 2014a). It was found that instead of directly inhibiting P-gp activity, the underlying mechanism of the schisandra lignans may be intervening with the substrate-P-gp complex and blocking the active P-gp transport sites (Fong et al., 2007). What’s more, continuous treatment with schisandra lignan extract for 10 days (or >10 days) would cause massive P-gp expression decrease in rat intestinal and brain tissues (Liang et al., 2013).
Discussion and Conclusion
In clinical setting, combination with traditional Chinese medicine is usually used to enhance the efficacy and/or reduce adverse reactions of modern medicines. Over the past few years, there have been many reports concerning the pharmacokinetic interactions involving the components of Wu Wei Zi in the form of herb extract, herb decoction, or herb preparation (Feng et al., 2017). Therefore, Table 5 summarized some DDI in animal experiments and clinical researches. It was found that Wu Wei Zi and its preparations have the capacity of affecting the blood concentration/exposure and pharmacokinetic profiles of other drugs. For example, the blood concentration of FK506 in vivo mostly contributed to the inhibition of CYP3A4 and/or P-gp via schisandra lignans when co-administration both in single and multiple doses. Meanwhile, the blood concentrations of the schisandra lignans were decreased because of their higher affinity to CYP3A. The schisandra lignans could also enhance the blood exposure of the compounds which were metabolized by CYP2D6 and CYP2D2, or transported by P-gp. The same compound showed different increases in the Cmax or AUC when the schisandra lignans co-administrated in different situation, indicating the occurrence of DDI was due to be the overall effect of all of the complex chemical components rather than the activity of an individual component. Considering the similarity of the chemical structure of the major schisandra lignans and their transformation during the episode of their metabolism, it is possible that the CYP3A/P-gp inhibitory activity of Wu Wei Zi should be considered as an overall and comprehensive effect of all schisandra lignans and their metabolites. It is important to point out that although schisandra lignans exhibit the potential for other CYPs-associated DDI based mostly on in vitro experimental results (Tables 2-4), “clinically relevant” DDI mediated by these CYPs (such as 2C19 and CYP2E1) are limited.
TABLE 5.
Reported DDIs in animal experiments and clinical researches.
| Subjects | Drug dosage and administration route | Some key pharmacokinetic parameters | Underlying mechanism | Ref. |
|---|---|---|---|---|
| SD male rats | Wuzhi Tablet: 0.25 g/kg, i.g., once | FK506: Cmax +80.1%, AUC0−t +2.1-fold | The inhibition of FK506 metabolism mostly contributed to the increase of oral FK506 exposure for the interplay of CYP3A and P-gp in liver by Wuzhi Tablet. | Qin et al. (2010a) |
| FK506: 1.89 mg/kg, i.g., once | ||||
| SD male rats | Wuzhi tablet: 250 mg/kg, i.g., once | FK506: Cmax +1.64-fold | Wuzhi tablet could enhance the blood concentration of FK506 in vivo, which might be due to inhibition of CYP3A4 and/or P-gp via substances | Qin et al. (2010b) |
| FK506: 3.78 mg/kg/day, i.g., once | ||||
| SD male rats | Schisandra lignans (Sch A, Sch B, Sch C, SCH, Gom A, and Gom C): 0.024 mg/kg, i.g., once | FK506: AUC0-t +152.0%, +109.6%, +46.4%, +41.4%, +598.4%, and +159.5%, by a single oral dose co-administration of Sch A, Sch B, Sch C, SCH, Gom A, and Gom C, respectively | The exposure of FK506 in rats was increased when co-administered with the lignans, which inhibited P-gp–mediated efflux and CYP3A-mediated metabolism of FK506, and reduced the intestinal first-pass action | Qin et al. (2014a) |
| FK506: 1.89 mg/kg, i.g., once | ||||
| SD male rats | Wuzhi tablet: 0.25 g/kg, i.g., once FK506: 1.89 mg/kg, i.g., once | With FK506 co-administration | The blood concentrations of the lignans were decreased and their CYP3A-mediated metabolisms were increased in the presence of FK506 since these lignans had higher affinity to CYP3A | Qin et al. (2014b) |
| Sch A: AUC0-t -47.2% | It was suggested that the lignans have higher affinity to CYP3A, so that their blood concentrations were decreased while the CYP3A-mediated metabolisms were increased in the presence of FK506. Transport study in Caco-2 cells showed that these lignans were not substrates of P-gp, suggesting decreased blood concentration of lignans by FK506 was not via P-gp pathway | |||
| SCH: AUC0-t -55.1% | ||||
| Gom A: AUC0-t -57.4% | ||||
| Gom C: AUC0-t -64.5% | ||||
| SD male rats | Wuzhi tablet: 62.5∼750 mg/kg, i.g., once | FK506: AUC0-t +1.76, +1.26, +2.48, +0.97, +0.87 and +0.36-fold, respectively, with Wuzhi tablet pretreatment (0, 0.5, 2, 6, 12 and 24 h before FK506 administration, 250 mg/kg) | The CYP3A activity was irreversibly inactivated by Wuzhi tablet, although the mRNA and protein expression of CYP3A was significantly induced after the long-term WZ treatment | Qin et al. (2020) |
| FK506: 1.89 mg/kg, i.g., once | FK506: AUC0-t +0.07, +0.44, +1.60, +1.32 and +1.42-fold, respectively, with Wuzhi tablet co-administration (62.5, 125, 250, 500 or 750 mg/kg) | |||
| SD male rats | Wuzhi capsule: 0.25 g/kg/day, i.g., once | FK506: AUC0–24 h + 128%, clearance -68% | The increase in blood FK506 concentration is caused by the strong inhibition of Wuzhi capsule on P-gp-mediated efflux and CYP3A metabolism | Qin et al. (2013) |
| FK506: 1.89 mg/kg, i.g., once | ||||
| SD male rats | Wuzhi capsule: 450 mg/kg, i.g., once and qd×12 days | FK506: Cmax +5.0-fold, AUC0-∞+4.9-fold by single dose of Wuzhi capsule | By inhibiting CYP3A and P-gp activity, the reduction of intestinal first-pass effect of FK506 is extensive and contributes greatly to the increase in FK506 bioavailability | Wei Bibbin et al. (2013) |
| FK506:1.2 mg/kg, i.g., once | FK506: Cmax +1.17-fold, AUC0-∞+1.29-foldby consecutive 12-day Wuzhi capsule pretreatment | |||
| SD male rats | North Wu Wei Zi water extract 1.5 g/kg/day, i.g., qd×7 days | By North Wu Wei Zi ethanol extract pretreatment | Multiple administrations of North Wu Wei Zi water extract increased the activities, mRNA and protein expressions of CYP2E1, and meanwhile, inhibited the activities and mRNA expression of CYP2D2 in vivo. The multiple administration might also inhibit the activity or expression of P-gp, which prevailed over CYP3A induction of and resulted in the elevation of FK506 plasma concentration | Wang Baolian et al. (2014) |
| North Wu Wei Zi ethanol extract 1.5 g/kg/day, i.g., qd×7 days | FK506: Cmax +9-fold, AUC0-t +6-fold | |||
| FK506, 1 mg/kg, i.g.., once | Chlorzoxazone: AUC0-t -35%, Cmax -20% | |||
| Chlorzoxazone, 100 mg/kg, i.g., once | Sertraline: Cmax +30% | |||
| Sertraline, 20 mg/kg, i.g., once | ||||
| SD male rats | Wuzhi capsule: 0.25 g/kg/day, i.g., once | Paclitaxel, i.g.: Cmax +95%, AUC0-∞+94% | Inhibition of the activity of P-gp and/or cytochrome P450 enzymes may contribute to the decrease of clearance of paclitaxel | Jin et al. (2011) |
| Paclitaxel: 30 mg/kg, i.g., once; or 0.5 mg/kg, i.v., once | Paclitaxel, i.v.: AUC0-∞+ 30% | |||
| SD male rats | Wuzhi capsule: 0.25 g/kg/day, i.g., once | Cyclosporine A (37.8 mg/kg): Cmax +13.1%, AUC0-∞+40.1% | That cyclosporine A concentration is slightly increased in the presence of concomitant Wuzhi capsule due to inhibition of CYP3A4 and/or P-gp | Xue et al. (2013) |
| Cyclosporin A: 37.8 or 1.89 mg/kg, i.g., once | Cyclosporine A (1.89 mg/kg): Cmax +84.1%, AUC0-∞+2.9-fold | |||
| SD male rats | North Wu Wei Zi ethanol extract: 54, 108, 216 mg/kg, i.g., once | Cyclosporine A: Cmax +26%, AUC0−t +2-fold | The increase in systemic exposure of cyclosporine A was probably caused by the strong inhibition of North Wu Wei Zi ethanol extract on P-gp-mediated efflux and CYP3A-mediated metabolism of cyclosporine A | Lai et al. (2015) |
| Cyclosporine A: 25 mg/kg, i.g., once | ||||
| SD male rats | North Wu Wei Zi water extract 0.75 g/kg/day, i.g., once and qd×7 days | By North Wu Wei Zi water extract pretreatment | Inhibition on liver microsomal CYP3A in vivo. with multiple administration | Zhai et al. (2018) |
| Cyclophosphamide: 300 mg/kg, i.v., once | DCCTX (Cyclophosphamide metabolite) | |||
| Cmax -69%, AUC0-t -49% when North Wu Wei Zi was administrated once | ||||
| Cmax -25%, AUC0-t -37% when North Wu Wei Zi was administrated by consecutive 7-day | ||||
| SD male rats | Wuzhi capsule: 300 mg/kg/day, i.g., once | DCCTX: Cmax -33.10%, AUC0-t -35.51% | The decreased DCCTX concentration was attributed by CYP3A inhibition effect of Wuzhi capsule | Chen et al. (2019) |
| Cyclophosphamide: 300 mg/kg, i.v | ||||
| SD male rats | Wuzhi capsule: 450 mg/kg, i.g., once and qd×7 days | Methotrexate: Cmax +1.18-fold, AUC0-t +79% with single dose of Wuzhi capsule | The inhibition of OAT1/3 and P-gp expression by Wuzhi capsule led to decrease in the clearance and increase in the blood exposure of methotrexate | Fu et al. (2021) |
| Methotrexate: 2 mg/kg, i.g., once | Methotrexate: Cmax +1.53-fold, AUC0-t +61% by consecutive 7-day North Wu Wei Zi ethanol extract pretreatment | |||
| SD male rats | Wuzhi capsule: 450 mg/kg, i.g., qd×7 days | With Wuzhi capsule pretreatment | The increase in blood exposure was caused by the P-gp inhibition in intestine, along with the inhibition of metabolic enzymes | Cui et al. (2021) |
| Lenvatinib:1.2 mg/kg, i.g., once | Lenvatinib: Cmax +1.69-fold, AUC0−t +62.7% | |||
| SD male rats | North Wu Wei Zi: 3, 10 g/kg, i.g., once | North Wu Wei Zi extract did not significantly alter the pharmacokinetics of lamivudine | A possible reason is that lamivudine is mainly excreted through the kidney and is not significantly metabolized by CYPs | Li et al. (2020) |
| Lamivudine: 10 mg/kg, i.v., once | ||||
| SD male rats | North Wu Wei Zi: 500 mg/kg, i.g., once | Warfarin: AUC0-∞ -29% | Wu Wei Zi activated PXR and induced CYP3As and 2Cs and then increased warfarin metabolism | Mu et al. (2006) |
| Warfarin, 2 mg/kg, i.v., once | ||||
| SD male rats | DZSM (containing 20% North Wu Wei Zi): 97.2 mg/kg, i.g., once Clopidogrel: 6.75 mg/kg, i.g., once | Clopidogrel: AUCc +1.25-fold, Cmax + 18% | DZSM extract inhibited clopidogrel metabolism in rat liver microsomes in a dose-dependent manner | Chen et al. (2016) |
| Clopidogrel metabolite: Cmax -65.8%, AUC0-∞ -64.8% IC50 of DZSM extract against clopidogrel metabolism is 0.02 mg/ml | ||||
| SD female rats | Schisandra lignan extract of North Wu Wei Zi: 500 mg/kg, i.g., once and qd×10 days | With Schisandra lignan extract single-dose pretreatment | Schisandra lignan extract may enhance the exposure of ginsenosides through inhibiting the activity and expression of P-gp | Liang et al. (2014) |
| Digoxin: 0.5 mg/kg, i.g., once | Digoxin: AUC0−∞t +1.25-fold; ginsenoside Rb2: AUC0−∞+2.18-fold | |||
| Ginsenosides extract: 120 mg/kg, i.g., once | ginsenoside Rc: AUC0−∞+1.49-fold | |||
| ginsenoside Rd: AUC0−∞ +1.86-fold | ||||
| ginsenoside Rb1: AUC0−∞ +1.29-fold | ||||
| With Schisandra lignan extract multi-dose pretreatment (qd×10 days) | ||||
| Digoxin: AUC0−∞t +2.00-fold; ginsenoside Rb2: AUC0−∞ +1.33-fold | ||||
| ginsenoside Rc: AUC0−∞ +1.49-fold | ||||
| ginsenoside Rd: AUC0−∞ +1.60-fold | ||||
| ginsenoside Rb1: AUC0−∞ +1.82-fold | ||||
| SD male rats | ginsenoside Rg1: 10 mg/kg, i.g., once | Ginsenoside Rg1: AUCc + 3.61-fold; ginsenoside Rb1: AUCc + 1.17-fold | Inhibition on liver microsomal CYP3A4 and CYP2D6 in vivo | Zhan et al. (2014) |
| ginsenoside Rb1: 10 mg/kg, i.g., once | schisandrin: AUCc + 86% | |||
| schisandrin: 10 mg/kg, i.g., once | compared with administrated alone | |||
| i.g. separately or in combination | ||||
| Healthy adults (n = 18, male and female) | Wuzhi capsule: 33.75 mg/d, p.o. bid×13d | Rapamycin: Cmax +96.3%, AUC0−∞ +106.8% | The variability of rapamycin pharmacokinetic parameters by Wuzhi capsule is probably due to the inhibition of CYP3A and (or) P-gp and (or) intestinal enzymes, resulting in increased absorption and decreased gut metabolism and increased rapamycin bioavailability | Li Wei-Liang et al. (2012) |
| Rapamycin: 2 mg, p.o., once | ||||
| Healthy male volunteers (n = 12, male) | Wuzhi capsule: 33.75 mg/d, p.o. bid×13d | FK506: Cmax + 227.1%, AUC0−t +164.2% | The increased systemic exposure of FK506 induced by North Wu Wei Zi ethanol extract could be due to inhibition of CYP3A4 and/or P-gp in the intestine, resulting in increased absorption and decreased gut metabolism | Xin et al. (2007) |
| FK506: 2 mg, p.o., once | ||||
| Renal transplant recipients (n = 64, male and female) | Wuzhi capsule: 22.5 mg/d, p.o | The blood trough concentration/dosage of FK506 in the Wuzhi capsule group was significantly higher than the non-Wuzhi capsule group at each time point from1 month to 1 year | Not mentioned | Cheng et al. (2021) |
| FK506: 3.0–5.0 mg/d, bid | ||||
| Liver transplant recipients (n = 46, male and female) | Patients were initially administered FK506 (first phase) and then South Wu Wei Zi extract was provided (second phase) | FK506: Cmax + 183%, AUC0-12 + 212%, with co-administration of South Wu Wei Zi extract | Not mentioned | Jiang et al. (2010) |
| Renal transplant recipients (n = 64, male and female) | Wuzhi capsule, 11.25 mg/d, p.o., bid×6 months | Following treatment with Wuzhi capsule for 6 months, the dosage of FK506 in Wuzhi capsule-treated patients had decreased by 34.0%, and the blood trough concentration had risen by 100.5% | Wuzhi capsule inhibited P-gp-mediated efflux and CYP3A-mediated metabolism of FK506 and that the reduction of the intestinal first-pass effect by Wuzhi capsule was the major cause of the increased FK506 oral bioavailability | Xin et al. (2011) |
| FK506: 1.42∼2.05 mg/d, p.o., once |
AUCc: (X0-single/X0-mix)*AUC0-t. X0-single, the dose of single compound administration; X0-mix, the relative dose of each compound in co-administration mixture.
AUC0−t (the area under the curve of 0 to t hours) and AUC0-∞ (the area under the curve of 0 to infinity).
Nowadays, a few in vitro studies have evaluated the modulatory effects of individual schisandra lignans on CYP3A/P-gp activities, the interactions and synergistic impact of schisandra lignans on CYP3A/P-gp activities are still remains to be further investigated. Besides, the discovery of a family of nuclear receptors such as pregnane X receptor (PXR), constitutive androstane receptor (CAR) and glucocorticoid receptor (GR) had given insight into the molecular explanation of CYP3A induction by xenobiotics (Qin and Wang, 2019). Regarding that Sch A, Sch B, SCH and Gom C could activate human PXR and induce the CYP3A4 reporter gene assay (Zhang et al., 2020), whether the same phenomenon could be observed for CAR and GR was not known. Therefore, studies should begin to explore the potential of these nuclear receptors as targets for those schisandra lignans to elucidate the ability to modulate the expression of CYP/P-gp whether they directly or through the transcriptional activation of nuclear receptors.
According to “Pharmacopoeia of the People’s Republic of China”, the suggested daily dosage of Wu Wei Zi is 2∼6 g. Based on the above discussion, the dosage is unlikely to affect the CYP activity according to Table 2 (the equivalent CYP inhibitory/inductive dosage of North- and South-Wu Wei Zi extract for human were annotated by “EDH”). Even so, the risk of CYP inhibition is high when patients are taking other CYP-blocking chemical drugs at the same time (especially CYP3A), such as nefazolone, ketoconazole, erythromycin and clarithromycin (Manikandan and Nagini, 2018). On the other hand, long-term use of Wu Wei Zi along with CYP-inducers (such as nevirapine and rifampin) could cause significant CYP induction (Finkelstein et al., 2010). In the above cases, the clinicians should put extra caution in applying drugs with narrow theraputic windows, like theophylline, tizanidine, warfarin, phenytoin, tacrolimus and quinidine (Schutte-Nutgen et al., 2018; Patocka et al., 2020). Meanwhile, clinicians must be aware of the contents and pharmacokinetic features of the schisandra lignans in Wu Wei Zi, which is helpful to understand the potential change of chemical drug efficacy with Wu Wei Zi co-administration, as well as the opposite after discontinuation of Wu Wei Zi. Therefore, this paper aimed to provide enough published data about DDIs induced by Wu Wei Zi and the schisandra lignans, and expected to provide guidance for rational use of chemical drugs or herbal medicines in order to avoid the occurrence of adverse side effects via CYP3A or P-gp inhibition and/or induction. Of note, the information and knowledge that were given in this review were the present practical methods and rules for screening and characterization of CYP3A/P-gp inhibitors and inductors and for facilitating the investigations on CYP3A/P-gp mediated DDI interactions.
Author Contributions
FZ and JZ wrote this manuscript. JZ and NW designed tables and figures. JG checked this manuscript. WC and JY supervised the project and critically revised the manuscript.
Funding
This project was supported by Innovative Clinical Research Funding Project (2020YLCYJ-Y25), the Three-year Action Plan for the Development of Traditional Chinese Medicine of Shanghai (ZY (2021-2023)-0215), Shanghai Municipal Health Commission (ZYCC2019018), Jin-Zi-Ta Talent project (0806 and 1016), Science and Technology Commission of Shanghai Municipality (22S21901900), National Natural Science Foundation of China (81830109, 81573793) and Shanghai Science and Technology Development Funds (19QB1404500).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
- Ali M., Khan T., Fatima K., Ali Q. U. A., Ovais M., Khalil A. T., et al. (2018). Selected Hepatoprotective Herbal Medicines: Evidence from Ethnomedicinal Applications, Animal Models, and Possible Mechanism of Actions. Phytother. Res. 32 (2), 199–215. 10.1002/ptr.5957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avula B., Choi Y. W., Srinivas P. V., Khan I. A. (2005). Quantitative Determination of Lignan Constituents from Schisandra Chinensis by Liquid Chromatyography. Chromatographia 61 (9-10), 515–518. 10.1365/s10337-005-0547-2 [DOI] [Google Scholar]
- Berg J., Tymoczko J., Stryer L. (2012). Editor Freeman W. H. (Basingstoke.Biochemistry [Google Scholar]
- Cao Y. (2010)., (Vol Doctor). Shenyang: Shenyang Pharmaceitical Univiersity. In Vitro metabolic Properties of Schisandrae Lignans Sch. traditional Chin. materia [Google Scholar]
- Chen L., Xiong X., Hou X., Wei H., Zhai J., Xia T., et al. (2019). Wuzhi Capsule Regulates Chloroacetaldehyde Pharmacokinetics Behaviour and Alleviates High-Dose Cyclophosphamide-Induced Nephrotoxicity and Neurotoxicity in Rats. Basic Clin. Pharmacol. Toxicol. 125 (2), 142–151. 10.1111/bcpt.13211 [DOI] [PubMed] [Google Scholar]
- Chen L., He L., Yu C., Chen J. (2015). Research Progress of Neuroprotective Effects of Schisandra Chinensis Baill Lignans. J. Beihua Univ. (Natural Science), [Google Scholar]
- Chen Q., Wu Y. J., Cheng N. N., Li Y. L., Wang Y. M. (2010). Dual Effects of Extract of Schisandra Chinensis Baill on Rat Hepatic CYP3A. Yao Xue Xue Bao 45 (9), 1194–1198. [PubMed] [Google Scholar]
- Chen X., Wang Y., Ma N., Tian J., Shao Y., Zhu B., et al. (2020). Target Identification of Natural Medicine with Chemical Proteomics Approach: Probe Synthesis, Target Fishing and Protein Identification. Signal. Transduct. Target. Ther. 5 (1), 72. 10.1038/s41392-020-0186-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X., Zhao Z., Chen Y., Gou X., Zhou Z., Zhong G., et al. (2016). Mechanistic Understanding of the Effect of Dengzhan Shengmai Capsule on the Pharmacokinetics of Clopidogrel in Rats. J. Ethnopharmacol. 192, 362–369. 10.1016/j.jep.2016.07.066 [DOI] [PubMed] [Google Scholar]
- Cheng F., Li Q., Wang J., Zeng F., Zhang Y. (2021). Effects and Safety Evaluation of Wuzhi Capsules Combined with Tacrolimus for the Treatment of Kidney Transplantation Recipients. J. Clin. Pharm. Ther. 46 (6), 1636–1649. 10.1111/jcpt.13493 [DOI] [PubMed] [Google Scholar]
- Chinese Pharmacopoeia Commission (2020). Pharmacopoeia of the People’s Republic of China, (Vol. 1). Beijing: People’s Medical Publishing House. [Google Scholar]
- Chun J. N., Cho M., So I., Jeon J. H. (2014). The Protective Effects of Schisandra Chinensis Fruit Extract and its Lignans against Cardiovascular Disease: a Review of the Molecular Mechanisms. Fitoterapia 97, 224–233. 10.1016/j.fitote.2014.06.014 [DOI] [PubMed] [Google Scholar]
- Cui Y., Li Y., Fan L., An J., Wang X., Fu R., et al. (2021). UPLC-MS/MS Method for the Determination of Lenvatinib in Rat Plasma and its Application to Drug-Drug Interaction Studies. J. Pharm. Biomed. Anal. 206, 114360. 10.1016/j.jpba.2021.114360 [DOI] [PubMed] [Google Scholar]
- Deng X., Chen X., Cheng W., Shen Z., Bi K. (2008). Simultaneous LC-MS Quantification of 15 Lignans in Schisandra Chinensis (Turcz.) Baill. Fruit. Chroma 67 (s7-8), 559–566. 10.1365/s10337-008-0589-3 [DOI] [Google Scholar]
- Dou Z. H., An L. P., Chen M., Miao Y. X., Feng Y. (2014). Simultaneous Determination of Six Lignanoids in Schisandra Chinensis by Quantitative Analysis of Multi-Components by Single Marker. Chin. Pharm. J. (Chinese) 49 (2), 147–151. 10.11669/cpj.2014.02.016 [DOI] [Google Scholar]
- Fan L., Mao X. Q., Tao G. Y., Wang G., Jiang F., Chen Y., et al. (2009). Effect of Schisandra Chinensis Extract and Ginkgo Biloba Extract on the Pharmacokinetics of Talinolol in Healthy Volunteers. Xenobiotica 39 (3), 249–254. 10.1080/00498250802687657 [DOI] [PubMed] [Google Scholar]
- Feng G., Zhai J. X., Chen W. S., Xiong X. J., Gao S. H., Yang Y., et al. (2017). Study Progress of the Interactions between the Schisandra Chinensis and the Schisandra Sphenanthera as Well as Thier Preparations with Other Drugs. Chin. J. Hosp. Pharm. (Chinese) 37 (21), 2206–2208. [Google Scholar]
- Finkelstein Y., Aks S. E., Hutson J. R., Juurlink D. N., Nguyen P., Dubnov-Raz G., et al. (2010). Colchicine Poisoning: the Dark Side of an Ancient Drug. Clin. Toxicol. (Phila) 48 (5), 407–414. 10.3109/15563650.49534810.3109/15563650.2010.495348 [DOI] [PubMed] [Google Scholar]
- Fong W. F., Wan C. K., Zhu G. Y., Chattopadhyay A., Dey S., Zhao Z., et al. (2007). Schisandrol A from Schisandra Chinensis Reverses P-Glycoprotein-Mediated Multidrug Resistance by Affecting Pgp-Substrate Complexes. Planta Med. 73 (3), 212–220. 10.1055/s-2007-967120 [DOI] [PubMed] [Google Scholar]
- Fu R., Wang X. N., Guo C. H., Li Y., Ding C. Y., Li Y. J., et al. (2021). Wuzhi Capsule Increased Systemic Exposure to Methotrexate by Inhibiting the Expression of OAT1/3 and P-Gp. Ann. Transl. Med. 9 (10), 845. 10.21037/atm-21-1303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao J. P., Wang Y. H., Yu Y. Q. (2003). Determination by HPLC and Variation Regularity of Lignan Constituents in Chinese Crude Drug Fructus Schisandrae Sphenantherae. Chin. J. Nat. Medicines (Chinese) 1, 89–93. [Google Scholar]
- Geng L. C., Yang Z. L. (2007). Determination of Lignanoids in Fructus Schisandrae from Different Regions by HPLC. Acta Chin. Med. Pharmacol. 35 (2), 47–48. 10.3969/j.issn.1002-2392.2007.02.017 [DOI] [Google Scholar]
- Gu W., Wei N., Wang Z. (2008). LC analysis of lignans from Schisandra sphenanthera Rehd. et Wils. Chroma 67 (11-12), 979–983. 10.1365/s10337-008-0621-7 [DOI] [Google Scholar]
- Guo W. J., Shao Q., Zhang Y. F., Fan X. H. (2012). Pharmacokinetics Interaction Among Three Major Active Compounds of Shengmai Formula in Rats. Zhejiang Da Xue Xue Bao Yi Xue Ban 41 (1), 6–12. 10.3785/j.issn.1008-9292.2012.01.002 [DOI] [PubMed] [Google Scholar]
- Halstead C. W., Lee S., Khoo C. S., Hennell J. R., Bensoussan A. (2007). Validation of a Method for the Simultaneous Determination of Four Schisandra Lignans in the Raw Herb and Commercial Dried Aqueous Extracts of Schisandra Chinensis (Wu Wei Zi) by RP-LC with DAD. J. Pharm. Biomed. Anal. 45 (1), 30–37. 10.1016/j.jpba.2007.05.016 [DOI] [PubMed] [Google Scholar]
- Harding M. W., Galat A., Uehling D. E., Schreiber S. L. (1989). A Receptor for the Immunosuppressant FK506 Is a Cis-Trans Peptidyl-Prolyl Isomerase. Nature 341, 758–760. 10.1038/341758a0 [DOI] [PubMed] [Google Scholar]
- Hu D., Han N., Yao X., Liu Z., Wang Y., Yang J., et al. (2014). Structure-activity Relationship Study of Dibenzocyclooctadiene Lignans Isolated from Schisandra Chinensis on Lipopolysaccharide-Induced Microglia Activation. Planta Med. 80 (08/09), 671–675. 10.1055/s-0034-1368592 [DOI] [PubMed] [Google Scholar]
- Hu D. (2012). Studies on Active Lignans from Schisandra Chinensis for Anti-alzheimer's Disease. Shenyang: Shenyang Pharmaceitical Univiersity (Vol Doctor). [Google Scholar]
- Hu F. (2011). The Effect of Vinegar-Preparation on the Schisandra Chinensis Lignan Contents and its Effect on CYP450 Activity, (Vol Doctor). Nanjing: Nanjing University of Chinese Medicine. [Google Scholar]
- Huang S., Zhang D., Li Y., Fan H., Liu Y., Huang W., et al. (2021). Schisandra Sphenanthera: A Comprehensive Review of its Botany, Phytochemistry, Pharmacology, and Clinical Applications. Am. J. Chin. Med. 49 (7), 1577–1622. 10.1142/S0192415X21500749 [DOI] [PubMed] [Google Scholar]
- Huyke C., Engel K., Simon-Haarhaus B., Quirin K. W., Schempp C. M. (2007). Composition and Biological Activity of Different Extracts from Schisandra Sphenanthera and Schisandra Chinensis. Planta Med. 73 (10), 1116–1126. 10.1055/s-2007-981559 [DOI] [PubMed] [Google Scholar]
- Iwata H., Tezuka Y., Kadota S., Hiratsuka A., Watabe T. (2004). Identification and Characterization of Potent CYP3A4 Inhibitors in Schisandra Fruit Extract. Drug Metab. Dispos. 32 (12), 1351–1358. 10.1124/dmd.104.000646 [DOI] [PubMed] [Google Scholar]
- Jiang W., Wang X., Xu X., Kong L. (2010). Effect of Schisandra Sphenanthera Extract on the Concentration of Tacrolimus in the Blood of Liver Transplant Patients. Int. J. Clin. Pharmacol. Ther. 48 (3), 224–229. 10.5414/cpp48224 [DOI] [PubMed] [Google Scholar]
- Jin J., Bi H., Hu J., Zeng H., Zhong G., Zhao L., et al. (2011). Effect of Wuzhi Tablet (Schisandra Sphenanthera Extract) on the Pharmacokinetics of Paclitaxel in Rats. Phytother. Res. 25 (8), 1250–1253. 10.1002/ptr.3407 [DOI] [PubMed] [Google Scholar]
- Jin J., Bi H., Hu J., Zhong G., Zhao L., Huang Z., et al. (2010). Enhancement of Oral Bioavailability of Paclitaxel after Oral Administration of Schisandrol B in Rats. Biopharm. Drug Dispos. 31 (4), 264–268. 10.1002/bdd.705 [DOI] [PubMed] [Google Scholar]
- Lai L., Hao H., Wang Q., Zheng C., Zhou F., Liu Y., et al. (2009). Effects of Short-Term and Long-Term Pretreatment of Schisandra Lignans on Regulating Hepatic and Intestinal CYP3A in Rats. Drug Metab. Dispos. 37 (12), 2399–2407. 10.1124/dmd.109.027433 [DOI] [PubMed] [Google Scholar]
- Lai Q., Wei J., Mahmoodurrahman M., Zhang C., Quan S., Li T., et al. (2015). Pharmacokinetic and Nephroprotective Benefits of Using Schisandra Chinensis Extracts in a Cyclosporine A-Based Immune-Suppressive Regime. Drug Des. Devel. Ther. 9, 4997–5018. 10.2147/DDDT.S89876 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H. J., Kim C. Y. (2010). Simultaneous Determination of Nine Lignans Using Pressurized Liquid Extraction and HPLC-DAD in the Fruits of Schisandra Chinensis. Food Chem. 120 (4), 1224–1228. 10.1016/j.foodchem.2009.11.068 [DOI] [Google Scholar]
- Li C. L., Hsieh C. H., Tsai T. H. (2020). Preclinical Pharmacokinetics of Lamivudine and its Interaction with Schisandra Chinensis Extract in Rats. ACS Omega 5 (4), 1997–2004. 10.1021/acsomega.9b03922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H. L., Ji D., Mao C. Q., Lu T. L., Wang J. (2013). Rapid Determination of Three Lignans in the Fruits of Schisandra Chinensis by LC-MS Methods and Analysis of Their Pharmacokinetics in Rat Plasma. Chin. Pharmacol. Bull. 29 (9), 1313–1317. 10.3969/j.issn.1001-1978.2013.09.031 [DOI] [Google Scholar]
- Li J., Chen S., Qin X., Fu Q., Bi H., Zhang Y., et al. (2017). Wuzhi Tablet (Schisandra Sphenanthera Extract) Is a Promising Tacrolimus-Sparing Agent for Renal Transplant Recipients Who Are CYP3A5 Expressers: a Two-phase Prospective Study. Drug Metab. Dispos. 45 (11), 1114–1119. 10.1124/dmd.117.076737 [DOI] [PubMed] [Google Scholar]
- Li L., Pan Q., Sun M., Lu Q., Hu X. (2007). Dibenzocyclooctadiene Lignans: a Class of Novel Inhibitors of Multidrug Resistance-Associated Protein 1. Life Sci. 80 (8), 741–748. 10.1016/j.lfs.2006.11.002 [DOI] [PubMed] [Google Scholar]
- Li R., Guo W., Fu Z., Ding G., Wang Z., Fu H. (2012). A Study about Drug Combination Therapy of Schisandra Sphenanthera Extract and Rapamycin in Healthy Subjects. Can. J. Physiol. Pharmacol. 90 (7), 941–945. 10.1139/y2012-080 [DOI] [PubMed] [Google Scholar]
- Li W-L., Xin H. W., Su M. W. (2012). Inhibitory Effects of Continuous Ingestion of Schisandrin A on CYP3A in the Rat. Basic Clin. Pharmacol. Toxicol. 110 (2), 187–192. 10.1111/j.1742-7843.2011.00787.x [DOI] [PubMed] [Google Scholar]
- Li W. L., Xin H. W., Yu A. R., Wu X. C. (2013). In Vivo effect of Schisandrin B on Cytochrome P450 Enzyme Activity. Phytomedicine 20 (8-9), 760–765. 10.1016/j.phymed.2013.02.005 [DOI] [PubMed] [Google Scholar]
- Li W. L., Xin H. W., Su M. W. (2010). Investigation on the Effect of Schisandrin B in Rat Liver Microsomal CYP3A Activity and its Inhibition Mechanism in. Vitro. Chin. Pharmacol. Bull. (Chinese) 26 (12), 1616–1621. CNKI: SUN:YAOL.0.2010-12-021. [Google Scholar]
- Li Y. H., Yang Y. H., Liu X. (2005). Study on Comparison between Shengmai Ultramicro Granule and Shengmai Powders. China J. Traditional Chin. Med. Pharm. (Chinese) 20, 588–590. 10.2217/nnm.14.169 [DOI] [Google Scholar]
- Li Z., He X., Liu F., Wang J., Feng J. (2018). A Review of Polysaccharides from Schisandra Chinensis and Schisandra Sphenanthera: Properties, Functions and Applications. Carbohydr. Polym. 184, 178–190. 10.1016/j.carbpol.2017.12.058 [DOI] [PubMed] [Google Scholar]
- Liang Y., Zhou Y., Zhang J., Liu Y., Guan T., Wang Y., et al. (2013). In Vitro to In Vivo Evidence of the Inhibitor Characteristics of Schisandra Lignans toward P-Glycoprotein. Phytomedicine 20 (11), 1030–1038. 10.1016/j.phymed.2013.04.005 [DOI] [PubMed] [Google Scholar]
- Liang Y., Zhou Y., Zhang J., Rao T., Zhou L., Xing R., et al. (2014). Pharmacokinetic Compatibility of Ginsenosides and Schisandra Lignans in Shengmai-San: from the Perspective of P-Glycoprotein. PloS One 9 (6), e98717. 10.1371/journal.pone.0098717 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu G. T. (1988). Biochemical and Pharmacological Effects of Schisandra chinensis and its Components on Animal Liver. Prog. Physiol. Sci. (Chinese) 19 (3), 197–203. [Google Scholar]
- Liu H. S., Zhang Y. M., Wang S. S., Shen Z. Y., Liu H. L. (2015). Determination of Deoxyschizandrin, Schisandrin B, Schisandrin C, and Ursolic Acid in Baogan Pill by RP-HPLC. Chin. Traditional Herbal Drugs (Chinese) 46 (24), 3691–3694. 10.7501/j.issn.0253-2670.2015.24.014 [DOI] [Google Scholar]
- Liu K., Song Y., Liu Y., Peng M., Li H., Li X., et al. (2017). An Integrated Strategy Using UPLC-QTOF-MSE and UPLC-QTOF-MRM (Enhanced Target) for Pharmacokinetics Study of Wine Processed Schisandra Chinensis Fructus in Rats. J. Pharm. Biomed. Anal. 139, 165–178. 10.1016/j.jpba.2017.02.043 [DOI] [PubMed] [Google Scholar]
- Liu L. S., Wang C. X., Qian F. U., Chen S. Y., Li J. L., Li J., et al. (2012). Effect of Wuzhi Tablet on Pharmacokinetics of Tacrolimus and Long-Term Clinical Outcome in Renal Transplant Patients. Organ. Transplant. (Chinese) 3 (1), 33–36. 10.1016/j.jep.2008.04.020 [DOI] [Google Scholar]
- Manikandan P., Nagini S. (2018). Cytochrome P450 Structure, Function and Clinical Significance: a Review. Curr. Drug Targets 19 (1), 38–54. 10.2174/1389450118666170125144557 [DOI] [PubMed] [Google Scholar]
- Mao J., Tay S., Khojasteh C. S., Chen Y., Hop C. E. C. A., Kenny J. R., et al. (2016). Revaluation of Time Dependent Inhibition Assays for Marketed Oncology Drugs: Comparison of Human Hepatocytes and Liver Microsomes in the Presence and Absence of Human Plasma. Pharm. Res. 33 (5), 1204–1219. 10.1007/s11095-016-1865-9 [DOI] [PubMed] [Google Scholar]
- Mu Y., Zhang J., Zhang S., Zhou H. H., Toma D., Ren S., et al. (2006). Traditional Chinese Medicines Wu Wei Zi (Schisandra Chinensis Baill) and Gan Cao (Glycyrrhiza Uralensis Fisch) Activate Pregnane X Receptor and Increase Warfarin Clearance in Rats. J. Pharmacol. Exp. Ther. 316 (3), 1369–1377. 10.1124/jpet.105.094342 [DOI] [PubMed] [Google Scholar]
- Palleria C., Roberti R., Iannone L. F., Tallarico M., Barbieri M. A., Vero A., et al. (2020). Clinically Relevant Drug Interactions between Statins and Antidepressants. J. Clin. Pharm. Ther. 45 (2), 227–239. 10.1111/jcpt.13058 [DOI] [PubMed] [Google Scholar]
- Panossian A., Wikman G. (2008). Pharmacology of Schisandra Chinensis Bail.: an Overview of Russian Research and Uses in Medicine. J. Ethnopharmacol. 118 (2), 183–212. 10.1016/j.jep.2008.04.020 [DOI] [PubMed] [Google Scholar]
- Patocka J., Wu Q., Nepovimova E., Kuca K. (2020). Phenytoin - an Anti-seizure Drug: Overview of its Chemistry, Pharmacology and Toxicology. Food Chem. Toxicol. 142, 111393. 10.1016/j.fct.2020.111393 [DOI] [PubMed] [Google Scholar]
- Qian T., Leong P. K., Ko K. M., Chan W. (2015). Investigation of In Vitro and In Vivo Metabolism of Schisandrin B from Schisandrae Fructus by Liquid Chromatography Coupled Electrospray Ionization Tandem Mass Spectrometry. Pp 06 (8), 363–373. 10.4236/pp.2015.68037 [DOI] [Google Scholar]
- Qin X., Wang X. (2019). Role of Vitamin D Receptor in the Regulation of CYP3A Gene Expression. Acta Pharm. Sin. B 9 (6), 1087–1098. 10.1016/j.apsb.2019.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qin X. L., Bi H. C., Wang C. X., Li J. L., Wang X. D., Liu L. S., et al. (2010b). Study of the Effect of Wuzhi Tablet (Schisandra Sphenanthera Extract) on Tacrolimus Tissue Distribution in Rat by Liquid Chromatography Tandem Mass Spectrometry Method. Biomed. Chromatogr. 24 (4), 399–405. 10.1002/bmc.1305 [DOI] [PubMed] [Google Scholar]
- Qin X. L., Bi H. C., Wang X. D., Li J. L., Wang Y., Xue X. P., et al. (2010a). Mechanistic Understanding of the Different Effects of Wuzhi Tablet (Schisandra Sphenanthera Extract) on the Absorption and First-Pass Intestinal and Hepatic Metabolism of Tacrolimus (FK506). Int. J. Pharm. 389 (1-2), 114–121. 10.1016/j.ijpharm.2010.01.025 [DOI] [PubMed] [Google Scholar]
- Qin X. L., Chen X., Wang Y., Xue X. P., Wang Y., Li J. L., et al. (2014a). In Vivo to In Vitro Effects of Six Bioactive Lignans of Wuzhi Tablet (Schisandra Sphenanthera Extract) on the CYP3A/P-Glycoprotein-Mediated Absorption and Metabolism of Tacrolimus. Drug Metab. Dispos. 42 (1), 193–199. 10.1124/dmd.113.053892 [DOI] [PubMed] [Google Scholar]
- Qin X. L., Chen X., Zhong G. P., Fan X. M., Wang Y., Xue X. P., et al. (2014b). Effect of Tacrolimus on the Pharmacokinetics of Bioactive Lignans of Wuzhi Tablet (Schisandra Sphenanthera Extract) and the Potential Roles of CYP3A and P-Gp. Phytomedicine 21 (5), 766–772. 10.1016/j.phymed.2013.12.006 [DOI] [PubMed] [Google Scholar]
- Qin X. L., Li J. L., Wang S. H., Chen X., Huang M., Bi H. C. (2020). Co-administration of Wuzhi Tablet (Schisandra Sphenanthera Extract) Alters Tacrolimus Pharmacokinetics in a Dose- and Time-dependent Manner in Rats. J. Ethnopharmacol. 263, 113233. 10.1016/j.jep.2020.113233 [DOI] [PubMed] [Google Scholar]
- Qin X. L., Yu T., Li L. J., Wang Y., Gu H. M., Wang Y. T., et al. (2013). Effect of Long-Term Co-administration of Wuzhi Tablet (Schisandra Sphenanthera Extract) and Prednisone on the Pharmacokinetics of Tacrolimus. Phytomedicine 20 (3-4), 375–379. 10.1016/j.phymed.2012.11.008 [DOI] [PubMed] [Google Scholar]
- Qiu B. X., Liu K., Zou L., Zhu H. F., Feng S. (2018). Effects of Schisandrae Chinensis Fructus Induced CYPs and Nrf2 Activation on Acute Liver Injury Induced by Acetaminophen. Zhongguo Zhong Yao Za Zhi 43 (24), 4908–4915. 10.19540/j.cnki.cjcmm.20180625.006 [DOI] [PubMed] [Google Scholar]
- Ren W., Zhang C. (2019). Research Progress of Schisandra chinensis and Prediction Analysis of Q-Maker. Chin. Trad. Herbal Drugs 51 (11), 3110–3116. [Google Scholar]
- Schutte-Nutgen K., Tholking G., Suwelack B., Reuter S. (2018). Tacrolimus - Pharmacokinetic Considerations for Clinicians. Curr. Drug Metab. 19 (4), 342–350. 10.2174/1389200219666180101104159 [DOI] [PubMed] [Google Scholar]
- Seo H. J., Ji S. B., Kim S. E., Lee G. M., Park S. Y., Wu Z., et al. (2021). Inhibitory Effects of Schisandra Lignans on Cytochrome P450s and Uridine 5'-Diphospho-Glucuronosyl Transferases in Human Liver Microsomes. Pharmaceutics 13 (3), 371. 10.3390/pharmaceutics13030371 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song H. X. (2004). Studies on HPLC–MS Analysis on the Lignans of Fructus Schisandrae Chinensis and Clustering Analysis on the Chief Active Constituents of Chinese Herbal Medicine, Vol Master. Changsha: Hunan Normal University. [Google Scholar]
- Sowndhararajan K., Deepa P., Kim M., Park S. J., Kim S. (2018). An Overview of Neuroprotective and Cognitive Enhancement Properties of Lignans from Schisandra Chinensis. Biomed. Pharmacother. 97, 958–968. 10.1016/j.biopha.2017.10.145 [DOI] [PubMed] [Google Scholar]
- Sun Y. X., Cong Y. L., Liu Y., Jin B., Si L., Wang A. B., et al. (2014). Schisandrin A and B Affect Subventricular Zone Neurogenesis in Mouse. Eur. J. Pharmacol. 740, 552–559. 10.1016/j.ejphar.2014.06.032 [DOI] [PubMed] [Google Scholar]
- Szopa A., Ekiert R., Ekiert H. (2017). Current Knowledge of Schisandra Chinensis (Turcz.) Baill. (Chinese magnolia Vine) as a Medicinal Plant Species: a Review on the Bioactive Components, Pharmacological Properties, Analytical and Biotechnological Studies. Phytochem. Rev. 16 (2), 195–218. 10.1007/s11101-016-9470-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian F., Dou D. Q., Cheng D. R., Wang L. N., Li M., Qin Y., et al. (2007). Comparison of Lignan Content in Crude and Processed Fructus. Schisandrae Chinensis. Mod. Chin. Med. (Chinese) 9 (9), 7–8. 10.3969/j.issn.1673-4890.2007.09.003 [DOI] [Google Scholar]
- Wan C. K., Zhu G. Y., Shen X. L., Chattopadhyay A., Dey S., Fong W. F. (2006). Gomisin A Alters Substrate Interaction and Reverses P-Glycoprotein-Mediated Multidrug Resistance in HepG2-DR Cells. Biochem. Pharmacol. 72 (7), 824–837. 10.1016/j.bcp.2006.06.036 [DOI] [PubMed] [Google Scholar]
- Wang B., Yang S., Hu J., Li Y. (2014). Multifaceted Interaction of the Traditional Chinese Medicinal Herb Schisandra Chinensis with Cytochrome P450-Mediated Drug Metabolism in Rats. J. Ethnopharmacol. 155 (3), 1473–1482. 10.1016/j.jep.2014.07.026 [DOI] [PubMed] [Google Scholar]
- Wang B., Hu J., Sheng L., Li Y. (2011). The Effects of Schisandra Chinensis on the Isozyme of Cytochrome P450 in Rat Liver Determined by Mixed Probe Substrate Method. Acta Pharmaceutica Sinica (Chinese) 46 (8), 922–927. [PubMed] [Google Scholar]
- Wang C-P., Li G. C., Shi Y. W., Zhang X. C., Li J. L., Wang Z. W., et al. (2014). Neuroprotective Effect of Schizandrin A on Oxygen and Glucose Deprivation/reperfusion-Induced Cell Injury in Primary Culture of Rat Cortical Neurons. J. Physiol. Biochem. 70 (3), 735–747. 10.1007/s13105-014-0342-3 [DOI] [PubMed] [Google Scholar]
- Wang L. J., Liu S. M., Mao C. Q., Tu-Lin L. U., Zhou Y., Xiao-Nan S. U., et al. (2015). Simultaneous Determination of 8 Lignans in Schisandra Chinensis by Quantitative Analysis of Multi-Components by Single Marker. Chin. J. Pharm. Anal. (Chinese) 35 (7), 1191–1197. [Google Scholar]
- Wang S., Zhao H., Huangpu M., Li X. (2003). Pharmacokinetic Effect of the Combination of Fructus Schisandrae and Radix Salvia Miltiorrhizae on Schizandrol A and Schizandrin B. Chin. Traditional Patent Med. (Chinese) 26 (12), 987–991. [Google Scholar]
- Wang W., Zhu S., Guo W., Fei T., Wei H., Chen W. (2016). Simultaneous Determination of Tacrolimus and Five Main Compounds of Wuzhi Capsule in Liver Transplant Patients' Whole Blood by LC-MS/MS. Chin. J. Mod. Appl. Pharm. (Chinese) 33 (7), 854–859. 10.13748/j.cnki.issn1007-7693.2016.07.005 [DOI] [Google Scholar]
- Wang Y. C., Ren K., Shen N., Huang X. D., Fan H. Y. (2013). Improvement of Impaired Memory in Mice by Schisandrin. Amr 750-752, 1533–1538. 10.4028/www.scientific.net/amr.750-752.1533 [DOI] [Google Scholar]
- Wang Z., Jin L., Ma Y., Cui Z., Li Q., Huang D. (2017). Quality Evaluation of Schisandra Sphenanthera Based on the Content of Gomisin C. China J. Chin. Materia Med. (Chinese) 42 (19), 3728–3732. [DOI] [PubMed] [Google Scholar]
- Wei B., Li Q., Fan R., Su D., Ou X., Chen K., et al. (2013). UFLC-MS/MS Method for Simultaneous Determination of Six Lignans of Schisandra Chinensis (Turcz.) Baill. In normal and Insomniac Rats Brain Microdialysates and Homogenate Samples: towards an In-Depth Study for its Sedative-Hypnotic Activity. J. Mass. Spectrom. 48 (4), 448–458. 10.1002/jms.3176 [DOI] [PubMed] [Google Scholar]
- Wei B. B., Chen X. H., Su D., Qian Z. Z., Yuan R. H., Bi K. S. (2010). Pharmacokinetics of Deoxyschizandrin and γ-Schizandrin in Schisandra Chinensis Oil in Rat Plasma. Chin. J. Hosp. Pharm. (Chinese) 9 012. [Google Scholar]
- Wei H., Miao H., Yun Y., Li J., Qian X., Wu R., et al. (2014). Validation of an LC-MS/MS Method for Quantitative Analysis of the 5 Bioactive Components of Wuzhi Capsule in Human Plasma Samples. Ther. Drug Monit. 36 (6), 781–788. 10.1097/FTD.0000000000000079 [DOI] [PubMed] [Google Scholar]
- Wei H. (2010). Study on the Pharmacokinetics of Wuzhi-Capsule and its Herb-Drug Interaction with Tacrolimus, Vol Doctor. Shanghai: Second Military Medical University. [Google Scholar]
- Wei H., Sun L., Tai Z., Gao S., Xu W., Chen W. (2010a). A Simple and Sensitive HPLC Method for the Simultaneous Determination of Eight Bioactive Components and Fingerprint Analysis of Schisandra Sphenanthera. Anal. Chim. Acta 662 (1), 97–104. 10.1016/j.aca.2009.12.039 [DOI] [PubMed] [Google Scholar]
- Wei H., Tao X., Di P., Yang Y., Li J., Qian X., et al. (2013). Effects of Traditional Chinese Medicine Wuzhi Capsule on Pharmacokinetics of Tacrolimus in Rats. Drug Metab. Dispos. 41 (7), 1398–1403. 10.1124/dmd.112.050302 [DOI] [PubMed] [Google Scholar]
- Wei H., Xu W., Cai F., Zhao G., Feng J., Sun L., et al. (2010b). Simultaneous Determination of Five Lignan Constituents of Wuzhi Capsule in Rat Plasma by LC-MS/MS: Application to Pharmacokinetic Study. J. Pharm. Biomed. Anal. 52 (5), 741–746. 10.1016/j.jpba.2010.02.003 [DOI] [PubMed] [Google Scholar]
- Wu J., Cao Y., Zhang Y., Liu Y., Hong J. Y., Zhu L., et al. (2014). Deoxyschizandrin, a Naturally Occurring Lignan, Is a Specific Probe Substrate of Human Cytochrome P450 3A. Drug Metab. Dispos. 42 (1), 94–104. 10.1124/dmd.113.053884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu X., Zhou Y., Yin F., Dai G., Li L., Xu B., et al. (2014a). Comparative Pharmacokinetics and Tissue Distribution of Schisandrin, Deoxyschisandrin and Schisandrin B in Rats after Combining Acupuncture and Herb Medicine (Schisandra Chinensis). Biomed. Chromatogr. 28 (8), 1075–1083. 10.1002/bmc.3122 [DOI] [PubMed] [Google Scholar]
- Wu Y. J., Cheng N. N., Hu Z. H., Chen B. Y. (2007a). Effects of Schisandra Chinensis Baill and Lignans on Enzyme Activity of Hepatic CYP3A. Chin. J. Clin. Pharm. (Chinese) 5, 267–271. 10.3969/j.issn.1007-4406.2007.05.001 [DOI] [Google Scholar]
- Xia Y.-G., Yang B.-Y., Liang J., Wang J.-S., Kuang H.-X. (2014a). Simultaneous Quantification of Five Dibenzocyclooctadiene Lignans in Schisandra Chinensis by HPLC Separation and Fluorescence Detection. Anal. Methods 6 (15), 5981–5985. 10.1039/c4ay00017j [DOI] [Google Scholar]
- Xia Y. G., Yang B. Y., Liang J., Yang Q., Wang D., Kuang H. X. (2014b). Quantitative Analysis and Fingerprint Profiles for Quality Control of Fructus Schisandrae by Gas Chromatography: Mass Spectrometry. ScientificWorldJournal 2014, 806759–806768. 10.1155/2014/806759 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xin H. W., Li Q., Wu X. C., He Y., Yu A. R., Xiong L., et al. (2011). Effects of Schisandra Sphenanthera Extract on the Blood Concentration of Tacrolimus in Renal Transplant Recipients. Eur. J. Clin. Pharmacol. 67 (12), 1309–1311. 10.1007/s00228-011-1075-7 [DOI] [PubMed] [Google Scholar]
- Xin H. W., Wu X. C., Li Q., Yu A. R., Zhu M., Shen Y., et al. (2007). Effects of Schisandra Sphenanthera Extract on the Pharmacokinetics of Tacrolimus in Healthy Volunteers. Br. J. Clin. Pharmacol. 64 (4), 469–475. 10.1111/j.1365-2125.2007.02922.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue X. P., Qin X. L., Xu C., Zhong G. P., Wang Y., Huang M., et al. (2013). Effect of Wuzhi Tablet (Schisandra Sphenanthera Extract) on the Pharmacokinetics of Cyclosporin A in Rats. Phytother Res. 27 (8), 1255–1259. 10.1002/ptr.4849 [DOI] [PubMed] [Google Scholar]
- Yan J., Ma Y., Wang T., Ji L. (2006). Pharmacokinetics Study of Schisandrin in Shengmai Granule. Traditional Chin. Drug Res. Clin. Pharmacol. (Chinese) 1 012. [Google Scholar]
- Yang G. (2011). The Key Technique Research of the High Quality and High Yield of Schisandra Chinensis, Vol Master. Haerbin: Heilongjiang University of Chinese Medicine. [Google Scholar]
- Yang J., Duan J. A., Li G. L., Zhu Z. H., Zhu T. L., Qian D. W., et al. (2014). Determination of Lignans in Schisandrae Sphenantherae Fructus from Different Regions. Zhongguo Zhong Yao Za Zhi 39 (23), 4647–4652. [PubMed] [Google Scholar]
- Yang K., Qiu J., Huang Z., Yu Z., Wang W., Hu H., et al. (2022). A comprehensive review of ethnopharmacology, phytochemistry, pharmacology, and pharmacokinetics of Schisandra chinensis (Turcz.) Baill. and Schisandra sphenanthera Rehd. et Wils. J. Ethnopharmacol. 284, 114759. 10.1016/j.jep.2021.114759 [DOI] [PubMed] [Google Scholar]
- Yoo H. H., Lee M., Lee M. W., Lim S. Y., Shin J., Kim D. H. (2007). Effects of Schisandra Lignans on P-Glycoprotein-Mediated Drug Efflux in Human Intestinal Caco-2. Planta Med. 73 (5), 444–450. 10.1055/s-2007-967178 [DOI] [PubMed] [Google Scholar]
- Yuan G., Liu Y., Li T., Wang Y., Sheng Y., Guan M. (2011). Simultaneous and Rapid Determination of Main Lignans in Different Parts of Schisandra Sphenanthera by Micellar Electrokinetic Capillary Chromatography. Molecules 16 (5), 3713–3722. 10.3390/molecules16053713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng H., Jiang Y., Chen P., Fan X., Li D., Liu A., et al. (2017). Schisandrol B Protects against Cholestatic Liver Injury through Pregnane X Receptors. Br. J. Pharmacol. 174 (8), 672–688. 10.1111/bph.13729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai J., Zhang F., Gao S., Chen L., Feng G., Yin J., et al. (2018). Schisandra Chinensis Extract Decreases Chloroacetaldehyde Production in Rats and Attenuates Cyclophosphamide Toxicity in Liver, Kidney and Brain. J. Ethnopharmacol. 210, 223–231. 10.1016/j.jep.2017.08.020 [DOI] [PubMed] [Google Scholar]
- Zhai J., Zhang F., Gao S., Chen L., Feng G., Yin J., et al. (2017). Time- and NADPH-dependent Inhibition on CYP3A by Gomisin A and the Pharmacokinetic Interactions between Gomisin A and Cyclophosphamide in Rats. Molecules 22 (8), 1298. 10.3390/molecules22081298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai J. X., Liu Z. H., Han N., Yin J. (2015). The Study of the CYP-Interfering Effects of Schisandra Chinensis and its Mechanism. Modernization Traditional Chin. Med. Materia Materia-World Sci. Tech. (Chinese) 1, 52–55. 10.11842/wst.2015.01.008 [DOI] [Google Scholar]
- Zhan S., Guo W., Shao Q., Fan X., Li Z., Cheng Y. (2014). A Pharmacokinetic and Pharmacodynamic Study of Drug-Drug Interaction between Ginsenoside Rg1, Ginsenoside Rb1 and Schizandrin after Intravenous Administration to Rats. J. Ethnopharmacol. 152 (2), 333–339. 10.1016/j.jep.2014.01.014 [DOI] [PubMed] [Google Scholar]
- Zhang H., Jiang Y., Wu J., Zheng C., Ran X., Li D., et al. (2017b). Metabolic Mapping of Schisandra Sphenanthera Extract and its Active Lignans Using a Metabolomic Approach Based on Ultra High Performance Liquid Chromatography with High-Resolution Mass Spectrometry. J. Sep. Sci. 40 (2), 574–586. 10.1002/jssc.201600954 [DOI] [PubMed] [Google Scholar]
- Zhang H., Bu F., Li L., Jiao Z., Ma G., Cai W., et al. (2017a). Prediction of Drug-Drug Interaction between Tacrolimus and Principal Ingredients of Wuzhi Capsule in Chinese Healthy Volunteers Using Physiologically-Based Pharmacokinetic Modelling. Basic Clin. Pharmacol. Toxicol. 122, 331–340. 10.1111/bcpt.12914 [DOI] [PubMed] [Google Scholar]
- Zhang H., Zhang G., Zhu Z., Zhao L., Fei Y., Jing J., et al. (2009). Determination of Six Lignans in Schisandra Chinensis (Turcz.) Baill. Fruits and Related Chinese Multiherb Remedies by HPLC. Food Chem. 115 (2), 735–739. 10.1016/j.foodchem.2008.12.010 [DOI] [Google Scholar]
- Zhang H., Zhou Y.-n., Sun S., Sun F.-f., Chai Y.-f., Zhang G.-q. (2014). Metabolic Rates and Metabolites of Three Lignans in Schisandra Chinensis (Turcz.) Baill in Rat Liver Microsomes. Acad. J. Second Mil. Med. Univ. 35 (4), 426–431. 10.3724/sp.j.1008.2014.00426 [DOI] [Google Scholar]
- Zhang W., Zhu Y. D., Zhang Q. Y., Ma L. J., Yang L., Guo W. Z., et al. (2020). Research Progress in Application and Mechanism of Schisandrae Chinensis Fructus for Prevention and Treatment of Liver Diseases. Zhongguo Zhong Yao Za Zhi 45 (16), 3759–3769. 10.19540/j.cnki.cjcmm.20200513.601 [DOI] [PubMed] [Google Scholar]
- Zhang W., Wang Y., Zou G. (2009). Study on Gastrointestinal Dynamics of Schisandrin. J. Liaoning Univ. Traditional Chin. Med. (Chinese) 36, 1965–1966. [Google Scholar]
- Zhao H. Q., Zhao R. S., Zhang C. (2006). Pharmacokinetic Studies of Schisandrin in Rat Plasma. Chin. J. Pharm. Anal. (Chinese) 26, 605–608. [Google Scholar]
- Zhou H., Zhang J., Wu S., Huang Y., Shi S., Zhang Y., et al. (2017). Effects of Wuzhi Capsules on Tacrolimus Trough Concentration in Heart Transplant Recipients. Herald Med. (Chinese) 36 (2), 158–162. 10.1080/00926230290001565 [DOI] [Google Scholar]
- Zhou S., Feng X., Kestell P., Paxton J. W., Baguley B. C., Chan E. (2005). Transport of the Investigational Anti-cancer Drug 5,6-Dimethylxanthenone-4-Acetic Acid and its Acyl Glucuronide by Human Intestinal Caco-2 Cells. Eur. J. Pharm. Sci. 24 (5), 513–524. 10.1016/j.ejps.2005.01.006 [DOI] [PubMed] [Google Scholar]
- Zhou Y., Mao C. Q., Hu J. Y. (2013). Tissue Distribution of Schisandrin, Deoxyschizandrin and Schisandrin B in Rat Determined by HPLC-MS. Chin. J. Pharm. Anal. (Chinese) 33, 1121–1126. 10.1056/NEJMoa1307361 [DOI] [Google Scholar]
- Zhu M., Chen X.-s., Wang K.-x. (2007). Variation of the lignan content of Schisandra chinensis (Turcz.) Baill. and Schisandra sphenanthera Rehd. et Wils. Chroma 66 (1-2), 125–128. 10.1365/s10337-007-0261-3 [DOI] [Google Scholar]
- Zuo X. B., Sun X. M., Wang C. Y., Liu J., Xiao X. H., Bai Z. F. (2019). China J. Chin. Materia Med. (Chinese) 44 (6), 1238–1245. 10.19540/j.cnki.cjcmm.20181210.001 [DOI] [PubMed] [Google Scholar]