Abstract
Purpose of Review
The aim of this review is to summarize and provide clear insights into studies that evaluate the interaction between air pollution, climate, and health in North Africa.
Recent Findings
Few studies have estimated the effects of climate and air pollution on health in North Africa. Most of the studies highlighted the evidence of the link between climate and air pollution as driving factors and increased mortality and morbidity as health outcomes. Each North African country prioritized research on a specific health factor. It was observed that the health outcome from each driving factor depends on the studied area and data availability. The latter is a major challenge in the region. As such, more studies should be led in the future to cover more areas in North Africa and when more data are available. Data availability will help to explore the applicability of different tools and techniques new to the region.
Summary
This review explores studies related to climate and air pollution, and their possible impacts on health in North Africa. On one hand, air quality studies have focused mainly on particulate matter exceedance levels and their long-term exposure impacts, namely, morbidity and mortality. The observed differences between the various studies are mainly due to the used exposure–response function, the studied population, background emissions, and natural emission from the Sahara Desert that characterize the region. On the other hand, climate studies have focused primarily on the impact of heat waves, vector-borne disease, and mental disorders. More than half of these studies have been on leishmaniasis disease. The review revealed unbalanced and insufficient research on health impacts from air pollution episodes and climate extremes across the region.
Keywords: Air pollution, Climate, Health, North Africa, Vector-borne disease, Climate extremes, Mental disorders
Introduction
In the recent years, climate change, extreme climate events, and the resulting deterioration in air quality have attracted worldwide attention. They had deserved special interest from the scientific community as their impact on human health and well-being becomes obvious. Climate change for example influences temperature and precipitation trends that affect the distribution of climate-sensitive diseases and impacts environmental and social conditions. Studies revealed a relationship between the incidence of certain diseases, especially cardiovascular and respiratory diseases, leishmaniases, arboviral diseases, mental disorders, typhoid fever and tick-borne diseases, and climate conditions in different regions of the world [1–7].
Cardiovascular and respiratory diseases are also due to air quality degradation that was widely recognized as a threat to both public health and economic progress. The World Health Organization (WHO) estimates that 4.2 million deaths annually can be attributed to outdoor air pollution [8]. Respiratory viral diseases are recognized as annual epidemics of the common cold. Influenza, also called flu or grippe, for example, is an acute viral infection of the upper or lower respiratory tract that is marked by fever, chills, and a generalized feeling of weakness and pain in the muscles, together with varying degrees of soreness in the head and abdomen. Studies have shown that climate factors are significantly associated with influenza seasonality [9]. Moreover, respiratory related epidemics caused by viruses such as severe acute respiratory syndrome coronavirus (SARS-CoV) and the newly emerging SARS-CoV-2 (COVID-19) occur during the winter months [10–13]. The mechanisms underlying the seasonal nature of respiratory viral infections have been examined and debated for many years. The two major contributing factors are the changes in environmental parameters and human behavior [14].
Leishmaniases are tropical endemic diseases, known in five epidemiological regions of the world: the Americas, East Africa, North Africa, West Asia, and Southeast Asia [15]. This group of diseases is caused by infection due to several types of parasites. In fact, there are at least twenty species of leishmania responsible for the three forms of the disease [16]: Mucocutaneous (ML), Visceral (VL), and Cutaneous (CL) [15]. Incidence of leishmaniasis in Mediterranean countries and across Europe has been on the rise [17]. Cases have been recently described in non-endemic areas, such as Germany [18], Sweden, or London [19, 20]. Leishmaniases are significantly affected by the change and instability of the climate in several regions [21]. The latter is expected to extend the geographical distribution and seasonality of phlebotomine vectors responsible for transmission of leishmaniases [22, 23].
Arboviral diseases are a global growing problem due to climate change, urbanization, population density, and global transportation [24]. They include a set of diseases such as Dengue, Chikungunya, Zika, and others. Incidence of diseases caused by arboviruses increased worldwide [25] and has aroused considerable concerns in the field of public health [26]. This growth may be explained by factors such as intensive growth of global transport systems; adaptation of vectors to increasing urbanization; inability to contain the mosquito population; and changes in environmental factors [25]. Five regions with populations of 100 million or more are projected to experience climate suitability for Zika transmission: East Africa, North America, East Asia, Western Europe, and North Africa and Middle East. A total of 737 million people worldwide could face their first exposure to year-round climate suitability for Zika transmission [27].
Research demonstrates that climate impacts human psychological health as well. Extreme weather events, for example, can impair mental health. They can lead to increases in depression, post-traumatic stress disorder, violence, and aggression. More gradual changes in climatic conditions, such as rising temperatures, are also harmful to mental health [5]. The study from Thoma and colleagues suggests that the climate and environmental crisis not only acts as direct stressors, but can also amplify the individual’s biopsychosocial vulnerability to develop mental illness [28]. In addition, there is increasing evidence that a significant proportion of people might be experiencing a harmful level of anxiety associated with their perception of climate change [5].
Typhoid is a serious and sometimes fatal disease caused by the bacterium Salmonella enterica serovar Typhi (S. Typhi). It spreads through contaminated food and water and poor sanitation. Although typhoid has been largely eliminated in industrialized countries, it continues to be a substantial public health problem in many low and middle-income countries. Globally, there are nearly 12 million cases and more than 128,000 deaths due to typhoid each year [29]. Increases in typhoid transmission are a possible consequence of climate change and the resulting increase in extreme weather events, including floods and droughts that lead to more severe surface water contamination [30].
Ticks exist in all continents and carry zoonotic pathogens that threaten human as well as livestock health. They spend most of their lives in the external environment outside their host and are thus expected to be affected by changes in climate [31]. Incidence of tick-borne diseases increases due to global warming, environmental and ecological changes, and availability of suitable habitat [32]. One of the main carriers of tick-borne infections is the Ixodes Ricinus (I. Ricinus) that was found related to climate parameters in many studies (Gilbert et al., 2014; Jore et al., 2014). The study of Rogulina and his team found that the parasitism peaks of I. Ricinus and their indicators are directly related to climatic conditions. They have observed increased humidity contributes to a decrease in the number of ixodids. Meanwhile, the increase of the average annual air temperature has the opposite effect and contributes to the increase of the arthropods [35].
Extreme weather events threaten lives, food security, and water quality and availability [36]. Prolonged heat waves causes extreme heat exposure that can result in a large number of deaths [37–39] and can lead to heatstroke and increased risks of hospitalization. Fluid disorders, renal failure, urinary tract infections, and septicemia were observed in vulnerable populations [36]. The intensity of these effects differs and relates to the nature of the geographical location. In the future between 2030 and 2050, climate change is expected to cause around 250,000 additional deaths per year due to heat stress and other climate-sensitive diseases [40].
Air pollution is the introduction of particulates, biological molecules, or other toxic chemicals into the Earth’s atmosphere. These particulates can take many forms, including solid particles, liquid droplets, or gasses. Many previous scientific studies assessed the impacts of air pollution on human health [41–44]. According to the Global Burden of Disease (GBD) study, environmental pollution was responsible for around 9 million premature deaths worldwide, with 2.9 million deaths caused by household air pollution and 4.2 million deaths caused by ambient air pollution [45]. Considering the environmental and economic shifts, pollution disproportionately affects both developed and developing countries [46]. For instance, in Europe in 2016, 7% of annual mortality was attributed to PM2.5 exposure, whereas 1% of annual mortality was attributed to NO2 exposure [47]. Of note, particulate matter (PM), which is one pollutant among others, is the most cited pollutant contributing to major harmful effects to global public health. It can induce various pathologies including bronchitis, asthma, lung cancer, preterm birth, inflammatory, cardiovascular problems, and chronic diseases [48–50].
North Africa, as a part of the MENA region (Middle East and North Africa), is particularly affected by the change in climate and is considered among the most water scarce regions in the world. Particularly, the whole region is expected to be one of the prominent and vulnerable climate change ‘‘hot spots’’ where health risks may be faced [51, 52]. In North Africa, climate change affects the susceptible sectors of the region and threatens the already increasing older population. This population contains a large percentage of those with chronic diseases, as well as poor people, who are therefore more vulnerable to the effects of climate [53]. The DALY (Disability Adjusted Life Years) estimates per million people are 3071.5 in Africa and 1586.5 in Eastern Mediterranean, compared to 8.9 in the developed countries [54].
Numerous review papers addressing the impact of climate and air pollution on health worldwide are available in the scientific literature. Yet, to the best of our knowledge, few studies have reviewed the literature in that field in North Africa. When doing a search of articles published in Africa on climate and air pollution up to December 31, 2016, we found a single journal article that only studied the relationship between asthma and Chronic Obstructive Pulmonary Disease (COPD) with road traffic–related ambient air pollution. We chose our review to include studies published since January 2016 [55]. To fill this gap, we conducted a scoping review of the literature with a focus on the impact of air pollution and climate on human health. The objectives of this study are to (1) provide a systemic overview of relevant studies related to the field; (2) identify the impact of air pollution and climate on health in North Africa; and (3) summarize the publication results and further explore research gaps and needs.
Methods
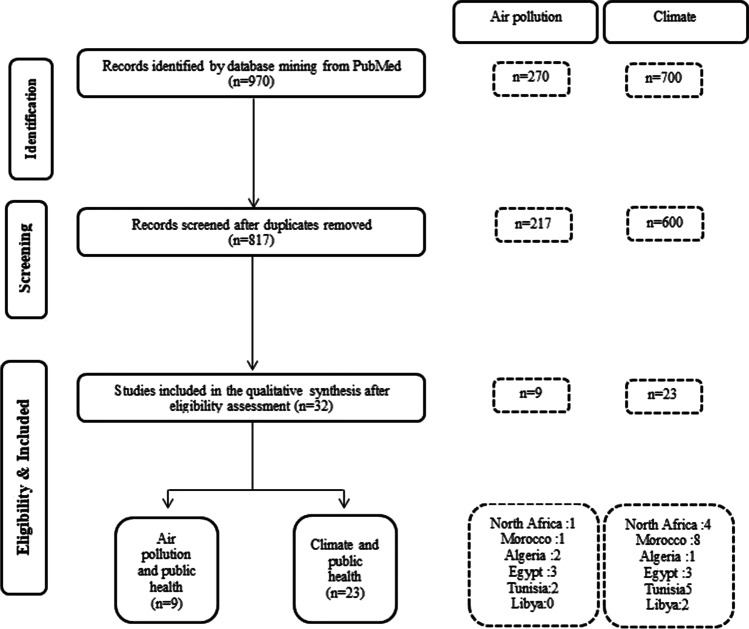
Figure 1 shows the search strategy used to identify the health impacts of air pollution and climate in North Africa (Fig. 2). This strategy consists of three steps:
Articles were identified through a literature exploration. First, an advanced search was performed through PubMed. We used the following keywords: “climate,” “health,” “air pollution,” “North Africa,” “Morocco,” “Algeria,” “Egypt,” “Tunisia.” and “Libya.” Then we searched for publications between 2016 and 2021. The last search was performed on June 08, 2021. A total of 970 articles were identified.
NBIB files, containing the resulting references, from PubMed were imported into Mendeley. We examined then the relevance of the papers by consulting the titles and abstracts and in case of uncertainty by evaluating the full texts.
The full texts of all records were reviewed for eligibility. The eligibility criteria are (1) the article is published after 2016, 2016 included; (2) the article is an original study of the impact of climate or air pollution on human health; (3) the study area of the article includes at least one of the following regions: North Africa, Morocco, Algeria, Tunisia, Libya, and Egypt; (4) and the articles selected should not be review articles. The selected studies were grouped according to two themes: air pollution and climate, and according to the region as well. The following information was extracted: authors, study area, calculated health effects, data, population and age, included periods, factors taken into account, methods, and the main results.
Fig. 1.
The search strategy
Fig. 2.
The study area
Results
Air Pollution and Public Health
Air Pollution Sources
In North Africa where there are a variety of sources of air pollution, air quality is mostly determined by location, meteorological conditions, season, and human activity. So far, there are limited accurate inventory studies in the region. Transportation, biomass burning, domestic combustion, energy, and industry are the main anthropogenic sources. Saharan dust and re-suspension of dust are the most common natural sources. The WHO has set pollution thresholds, whose exceeding can lead to numerous harmful effects on health. Nevertheless, some countries in the study region have established their own standards for monitoring air quality at the national level. In North Africa, the majority of available studies related to air pollution sources focus mainly on PM.
In Morocco, exceedance of WHO air quality guideline values was frequently observed in heavy traffic zones [56, 57], industrial zones, and in cities known by their important economic activities [57]. Moreover, due to the geographical position of the country, the natural process involving Sahara dust is an additive factor contributing to the increase in pollution precisely by PM10 [58]. Furthermore, the season has a significant impact on pollution levels. For example, during summer and spring, ozone (O3) concentration reached alarming levels in the city of Marrakech and the PM10 levels exceeded the threshold limits [59]. Other studies have shown that particulate pollution in the cities of Meknes and Kenitra is characterized by a high proportion of coarse particles, showing the importance of terrigenous inputs and soil resuspension [60–62]. In the Greater Casablanca region, a study concluded that road traffic and meteorological conditions are the main causes of air pollution [57]. In Algeria, data collected in urban and roadside stations at Algiers showed that the observed annual average concentration of PM1, PM2.5, and PM10 was 18.24, 32.23, and 60.01 µg m−3, respectively. The analysis revealed the anthropogenic and natural origins of PM. Desert dust influences the atmospheric composition of PM in Algiers [63]. PM10 samples collected between January 2015 and February 2016 at an urban site at Zouaghi, in the south of Constantine, showed that PM concentrations exceeded the WHO’s 24-h guideline value (50 µg m−3), linking such levels to resuspended dust and Saharan dust [64]. In Tunisia, the analysis of PM10 constituents revealed the anthropogenic and natural sources of PM, derived from traffic emissions, heavy industries, biomass burning, mechanic and electronic factories, and agricultural activities gathered with meteorological conditions [65]. In Egypt, a study in the residential-industrial area Shubra El-Kheima in the north of Greater Cairo revealed that fine aerosol particle concentrations, collected during the period from October 2010 to May 2011, exceed the WHO annual Air Quality Standard levels. The existence of PM2.5 unveiled the dominance of elements linked to mineral dust, heavy traffic, and industrial activities [66].
Overall, in North Africa, the main types of PM sources are industry, heavy traffic, and biomass combustion, as well as the Sahara Desert, and dust resuspension.
In the context of this review, 9 epidemiological studies were identified to examine the association between air pollution and mortality, morbidity and other health outcomes in North Africa. Details about these studies are given in Table 1.
Table 1.
The characteristics of the epidemiological studies included in the present review regarding air pollution and public health
| Authors, year | Study area | Calculated health effects | DATA | Population age | Included periods | Factors taken into account | Methods | Main results |
|---|---|---|---|---|---|---|---|---|
| Ghennam, Attou and Abdoun, 2021 | Algeria | Asthma and bronchitis hospital admissions | The index of atmospheric purity (IAP), the index of human impact (IHI), environmental parameters, and the respiratory illness data from Algerian hospital | Not specified | Between 2006 and 2016 | 976 cases taken into account including 771 with asthma and 205 with bronchitis | Redundancy analysis is used to quantify the relationship and the influence of air pollution and environmental parameters on respiratory diseases | Atmospheric pollutions contribute significantly in asthma and bronchitis with higher frequency related to hospital admission for asthma |
| Boussetta et al., 2020 | Tunisia | The impact of polluted area (PA) on the performance of athletes, cardiovascular, muscle damage, and oxidative stress | Data of cardiovascular parameters, damage markers, and oxidative stress markers | Mean age 22.45 | Randomized experimental protocol | Similarity of weather and climate | Comparison of measured parameters in two areas (i.e., polluted (PA) and non-polluted (NPA)) and the quantification of the impact of red orange juice supplementation (ROJS) | The impact of polluted areas on the athlete’s health could be mitigated using red orange juice supplementation |
| Marchetti et al., 2019 | Greater Cairo (Egypt) | Effects in human lung cells that are attributed to | The fine () | Not specified | From December 2016 to November 2017 | Seasonal variability | Correlations between Chemical Parameters and Biological Effects | displayed different biological effects in human lung cells exposed in vitro |
| Mohammed, Ibrahim and Saleh, 2019 |
Egypt: Shoubra El-Khaima and Ain Sokhna sectors |
- and -associated hospital admission respiratory cases | and | Not specified | From December 2015 to November 2016 | Shoubra El-Khaima and Ain Sokhna population | Air Q 2.2.3 Software | Results showed important differences in the ( and )-hospital admission association across two studied cities |
| Wheida et al., 2018 | Greater Cairo (Egypt) | Mortality attributable to long-term exposure to , NO2, and | , , NO2, and | All ages | From 2000 to 2004 and from 2010 to 2015 | Age and Greater Cairo population | AirQ + Software. The used concentration–response functions (CRF) are log-linear, linear, and log–log | causes a mortality of 62/100000 relative to the whole population of Greater Cairo. O3 gas is responsible for between 2.4 and 8.8% of mortality due to respiratory diseases and NO2 is thought to be responsible for 7850 to 10,470 deaths each year in the study area |
| Croitoru and Sarraf, 2017 | Morocco | Mortality and morbidity related to human exposure to ambient | All ages | The period 2012–2015 | Moroccan population, and age | Applying Integrated Exposure–Response Functions (IER) to | Adults over 55 years old are more vulnerable for premature deaths owing to air pollution due to ischemic heart disease and stroke and children under 5 years old are the ones most at risk of premature fatalities caused by air pollution due to acute lower respiratory infections | |
| Malley et al., 2017 | The globe including North Africa | -associated preterm births | National population-weighted, annual average ambient concentration, preterm birth rate, and number of livebirths | Not specified | 2010 | Data for 183 countries | Concentration response functions (CRF) from previous studies that are conducted in the USA and Europe | Regardless of the limitations and uncertainties in the estimations, results demonstrate that maternal exposure is a potentially significant global risk factor for preterm birth |
| Boussetta et al., 2017 | Tunisia | The effect of air pollution on the performance in anaerobic tests and cardiovascular and hematological parameters | Data of cardiovascular parameters, blood measurement, and anaerobic performances | Mean age: 21.8 [range: 20–24] | Test sessions in the morning (07:00–09:00 h) and in the evening (17:00–19:00 h) | Similarity of weather and climate | Comparison of measured parameters in two areas (i.e., polluted (PA) and non-polluted (NPA)) | Polluted area influences health and athletic training performances and the impact is more aggravated at the evening |
| Benaissa et al., 2016 | Bejaia City (Algeria) | All-cause mortality and respiratory and cardiovascular hospitalizations related to short-term exposure to ambient | , mortality data and data on hospitalization for cardiovascular and respiratory diseases | All ages | From 04/08/2015 to 07/14/2015 | Population of Bejaia (Algeria) | Traditional health impact assessment methods are used to analyze the impact of short-term exposure to . This is based on the parameters of Aphekom City Report Marseille | In 2014, the excess in concentrations was responsible for 29 deaths, 36 respiratory hospitalizations, and 23 cardiac hospitalizations per 100,000 of the total mortality, respiratory hospitalizations, and cardiac hospitalizations, respectively |
Mortality-Related Studies
In North Africa, there are a very limited number of studies that relate each pollutant to mortality. In particular, more interest is given to health effect of air pollution by ambient PM, which, according to its aerodynamic diameter, affects the respiratory tract and the cardiovascular system. Indeed, the impact of short-and long-term exposure to PM on overall mortality is well-documented.
The study carried out by Benaissa and colleagues in Algeria estimates the number of all-cause mortality that could be prevented by lowering annual mean levels of PM10 in Bejaia city to 5 µg/m3 that might prevent 4 deaths per year. Furthermore, a reduction in annual PM10 levels to 20 µg/m3 (WHO Air Quality Guidelines for PM10) could prevent 55 deaths per year [67]. Also, the authors emphasize the potential underestimation of the aforementioned risks, given the fact of the absence of a local concentration–response function.
Furthermore, a study conducted in Morocco that focused on the assessment of PM2.5 pollution health outcomes found that in 2014, exposure to ambient PM2.5 caused about 2200 deaths in the country, due to ischemic heart disease, stroke, and lung cancer, in which risk of premature mortality for adults over 55 is mainly due to ischemic heart disease and strokes. The risk of premature mortality for children under the age of 5 years old is mainly due to an acute lower respiratory tract infection [68].
Another study conducted by Wheida and colleagues [69] in Egypt assessed the number of deaths that can be attributed to long-term exposure to three pollutants: PM2.5, NO2, and O3, in the Greater Cairo area. The study claims that PM pollution causes mortality of 62/100000 relative to the whole population of the area. O3 is responsible for between 2.4 and 8.8% of mortality due to respiratory diseases and NO is thought to be responsible for 7850 to 10,470 deaths each year in the study area.
It is worth mentioning that studies investigating the potential health effects of air pollution do not focus on mortality only, but also on a wide range of human diseases, namely, hospital admission due to cardiovascular and respiratory diseases.
Morbidity-Related Studies
In various studies, cardiovascular and respiratory diseases are frequently linked to air pollution. An analysis of diverse populations in Bejaia city in Algeria shows possible prevention respectively of 5.2% and 3.2% from getting admitted to hospital for pulmonary and cardiovascular diseases for each 5 µg/m3 decrease in PM10 [67].
In another study in Algeria, Ghennam and colleagues have investigated the association between air pollution and the spatial prevalence of asthma and bronchitis based on lichen biomonitoring tools. The results have unveiled a significant link between air pollution levels and hospitalizations for asthma and bronchitis with increased hospitalization risk related to asthma [70]. In this same context, Mohammed and colleagues have estimated the number of hospital admission for respiratory diseases attributed to SO2 and NO2, in Shoubra El-Khaima and Ain Sokhna in Egypt, using Air Quality Health Impact Assessment (Air Q 2.2.3) tools. They showed important differences in the SO2 and NO2 hospital admission between the two studied cities [71]. For example, while Shoubra El-Khaima’s sector reported the largest risk estimates (311/234 cases attributed to exposure to SO2/NO2), smaller estimates were found in Ain Sokhna (18/15 cases attributed to exposure to SO2/NO2). These findings have been subsequently attributed to the fact that Shoubra El-Khaima has a higher density of population and industry than the Ain Sokhna coastal sector.
Other Health Outcome Studies
At a larger scale, a study covered 183 individual countries, including North Africa, found that the exposure of pregnant women to PM2.5 is a risk factor for preterm birth. Results show that the region has been a major contributor to the global PM2.5-associated preterm births [72].
Another study conducted in Egypt, precisely in Greater Cairo, investigated the in vitro toxicity of fine PM. The study found that this toxicity is linked to the season. Indeed, in warmer-season, possible genotoxic effects appear, while in colder seasons, potential cell xenobiotic responses and pro-inflammatory effects appear [73].
In sports science, two studies were conducted in Tunisia [74, 75]. They have assessed the impact of air pollution on soccer players. The first study investigated effects on performance variation, cardiovascular, hematological parameters, and blood gasses. The comparison between body reaction in polluted and non-polluted areas has reported that according to the time of the day, polluted areas influence health and then athletic training performances. Also, the impact is intensified in the evening. The second study has confirmed these results and has revealed that ROJS (Red Orange Juice Supplementation) unlike PLA (Red orange or placebo) is an appropriate measure to reduce the risk of pollution exposure, particularly on muscle damage and oxidative stress.
Climate and Public Health
For the purpose of this review, and considering the criteria adopted, we identified a total of 23 studies conducted in the region and relating climate to health; 3 focused on climate extremes, 18 on vector-borne diseases, and 2 on mental disorders. Details about the considered studies are gathered in Table 2.
Table 2.
The characteristics of the epidemiological studies included in the present review regarding climate and public health
| Authors, year | Study area | Calculated health effects | Data | Population, age | Included period | Factors taken into account | Methods | Main results |
|---|---|---|---|---|---|---|---|---|
| Outammassine et al., 2021 | Morocco | Modeling and mapping of the habitat suitability and the potential distribution of arboviruses vectors | Dataset records for Ae. albopictus and Ae. vittatus from the Global Biodiversity Information Facility (https://www.gbif.org) and the worldwide database compiled by Kraemer et al. ( 10.15468/dl.fygusa) | Not applicable | From 1916 to 2017 | Altitude, temperature, and precipitation | Maximum entropy (Maxent) modeling under current climatic conditions | Areas with maximum risk and high potential distribution were mainly located in the northwestern and central parts of Morocco |
| Gijón‐Robles et al., 2021 |
Morocco Anthroponotic cutaneous leishmaniasis (ACL) endemic area (El Borouj) and non-endemic area (Sidi Hajjaj) |
Comparison of the densities and genetic characteristics of P. sergenti populations in two nearby localities | Data from captured Sand flies | Not applicable | June 20 to July 10 and from September 20 to October 10, 2015 | Temperature, precipitation | Sand flies capture using CDC light traps inside households and sticky papers outside dwellings, Sand fly DNA extraction, Mitochondrial lineage determination by mt DNA Cyt b PCR–RFLP Polymerase | P. sergenti density was significantly higher in the endemic area than in the non-endemic town |
| Ryan et al., 2021 | Global including north Africa | First systematic assessment of where future temperatures are expected to become suitable for Zika virus (ZIKV) transmission | Thermal responses for mosquito and virus traits that drive transmission | Population at risk based on human population density data from 2015 | 2050 | Temperature, human population density | Temperature-dependent transmission model for Zika virus (ZIKV). Aedes transmission models | Over 1.3 billion new people could face suitable transmission temperatures for ZIKV by 2050 |
| Bettaieb et al., 2020 | Tunisia | Impact of heat on daily all-cause mortality |
Daily time series of mortality; daily mean, minimum, and maximum temperature (in °C); relative humidity (in %); wind speed (in m/s) and direction (in rose 368; sea level pressure (in hPa) NO2 (in µg/m3) |
Population of Tunis | Summer season (May–October) of a 3-year period (2005 to 2007) | Weather, air pollution |
The Generalized Additive Model (GAM) using cubic regression splines and Akaike’s Information Criterion (AIC) A segmented linear regression and the Muggeo’s approach to estimate the breakpoint. Poisson Generalized Estimating Equations (GEE) |
Daily mortality increased significantly by 2% for an 18 °C increase in daily maximum temperature above the breakpoint |
| Essayagh et al., 2020 | Meknes, Morocco | Cases of typhoid fever | Socio-demographic information, clinical, and paraclinical data; season; notion of contagiousness; food consumed; water supply; waste water disposal; notion of bathing and evolution of the case | Confirmed cases of typhoid fever ¨de at health centers or hospitals in Meknes | 2013–2016 | Seaonal study, temperature | Case series study using an epidemiological surveillance database | The number of cases of fever typhoid was highest in the dry season. Climatic conditions could facilitate the invasion of certain pathogens. The analysis during the different periods showed notable changes during the different seasons of the year with a high average incidence in the dry season (5 cases per 100,000 inhabitants in summer and 2.8 cases per 100,000 inhabitants in spring) |
| Elsobky et al., 2020 | Menoufia, Egypt | Climate impact on HPAI-H5N1disease variability | Domestic poultry HPAI-H5N1 outbreak | Not applicable | January 2006 to December 2016 | Temperature, relative humidity | General Linear Model (GLM) Generalized estimating equations (GEEs) | Effect of climate variability differs according to the timing of the outbreak occurrence |
| Maryam Hakkour et al., 2020 | Nine provinces located in the extreme and central north of Morocco (Aounate, Taza, Chefchaouen, Al Hoceima, Larache, T ´etouane,Tanger-Assilah, M’diq-Fnideq, and Fahs-Anjra Provinces) | Leishmaniasis cases | Human case. Environmental variables | A total of 6128 cases in the 9 provinces | 1997–2018 | Humidity, temperature | Linear regression model | Humidity was significant for both CL and VL (P < 0.05), while the temperature showed a significance only for VL. Wet season (October to April) had a higher incidence of leishmaniasis compared to the dry season (May to September) specifically for CL |
| Hajar El Omari et al., 2020 | Meknes prefecture, Morocco | The abundance of sandflies indicator of the risk of leishmaniasis | Meteorological data. climate factors | Phle-botomian population | From March 2016 until April 2017 | Temperature, humidity, precipitation | Principal component analysis (PCA) | The existence of a positive correlation between the temperature and the abundance of sandflies (r = 0.99) and a negative correlation with humidity and precipitation (r = − 0.87 and r = − 0.72) |
| Aly Zein Elabdeen Kassem et al., 2020 | Forty-three countries including Morocco and Tunisia | COVID-19 cases per million | Total number of confirmed cases | COVID-19 cases | January–July 2020 | Temperature | Non-linear least squares method | Inverse relationship between COVID-19 cases per million and the temperature in just two observations out of twelve other factors than temperature is the most influential on the transmission of COVID-19 |
| S.A. MEO et al., 2020 | 20 countries of the globe including Algeria | Mean daily case incidence, cumulative cases, and cumulative deaths of COVID-19 | Metrological data, daily new cases, and deaths of COVID-19 | population of the 20 countries including Algeria | Dec 29, 2019 to May 12, 2020 | Temperature | Simple linear regression analysis | The COVID-19 cases and deaths per million population were significantly low in countries with high temperature and low humidity (warmest countries), compared to those countries with low temperatures and high humidity (coldest countries) |
| humidity | ||||||||
| Saverio Bellizzi et al., 2020 | Eastern Mediterranean Region including Morocco, Tunisia, Egypt, and libya | The health consequence vulnerability index, mortality, morbidity | Publicly available articles in PubMed, data displayed on the WHO Regional Health Observatory, EMR specific program documents, and on WHO Country Office reports. Documentation on access to water and sanitation in the EMR countries. UNHCR figures on internally displaced Persons (IDPs) | Under-5-all population of Eastern Mediterranean Region | From 1990 through 2019 | Health status of the population before the disaster; infrastructure like water supply and sanitation systems; food insecurity; absence of warning systems; population displacement; and other concurrent situations like economic crisis, political instability, and armed conflict | A review of scientific literature and WHO EMR documentation. Calculation of index score for each country based on the vulnerability factor scores |
The WHO Eastern Mediterranean Region remains a highly vulnerable area of the world in terms of health consequences due to drought. The effects of drought have potentially greater impact on individual with underlying chronic medical conditions such as respiratory diseases as well as on persons with disabilities, who may not be able to access emergency response services due to the difficulties in mobility, hearing, seeing, and understanding Fifteen million people remain without even basic water services, live in Iraq, Islamic Republic of Iran, and Morocco. Morocco and Tunisia are the two countries among the 9 out of 22 EMR countries with safe drinking water management. Seventeen million people remain without basic sanitation services, living in Egypt, Iraq, and Morocco. Access to water and soap for hand washing varies widely, from 10% in Somalia to about 90% in Tunisia, Egypt, and Iraq |
| Caini et al., 2019 | A total of 31 countries including Morocco from north Africa | Epidemiological characteristics, pattern of circulation, and geographical distribution of influenza B viruses and its B/Victoria and B/Yamagata lineages | The GIBS database of epidemiological and virological influenza surveillance data from thirty-one countries around the world | All population | 2000–2018 | Age (exact age or age groups) | A statistical approach. The non-parametric Kruskal–Wallis test. EPIPOI software (Alonso WJ, McCormick BJ. EPIPOI: a user-friendly analytical tool for the extraction and visualization of temporal parameters from epidemiological time series. BMC Public Health. 2012; 12(1):982. 27.) | B/Yamagata was more frequent in countries that have temperate climate |
| Bauer et al., 2019 | Thirty-two countries including Tunisia | Association between solar insolation and a history of suicide attempt in patients with bipolar I disorder | A total of 3365 patients from 310 onset locations, 50 sites in 32 countries | A total of 1047 patients that had a history ofsuicide attempt | 2010 to 2016 | Sex, a history of alcohol or substance abuse, belonging to younger birth cohort, living in a country with a state-sponsored religion |
Diagnosis of bipolar disorder according to DSM-IV criteria from a psychiatrist, direct questioning, reviewing records, or both |
A significant inverse association between a history of suicide attempt and the ratio of mean winter solar insolation/mean summer solar insolation. Living in locations with large changes in solar insolation between winter and summer may be associated with increased suicide attempts in patients with bipolar disorder |
| Ahmed H. Salaheldin et al., 2018 | Four governorates from Egypt (Alexandria, Cairo, Minya, and Luxor) | Total number of A/H5N1 outbreaks per month | Total number of A/H5N1 outbreaks per month. Six viruses from the repository of the Friedrich-Loeffler-Institut | - | Ten seasons from 2006 to 2015. From the 1st of October to 31st of March | Climatic factors (temperature, relative humidity, and wind speed), biological fitness in vitro, and pathogenicity in domestic Pekin and Muscovy ducks | Negative binomial regression models | Ambient temperature in winter months influenced the spread of avian influenza virus A/H5N1 |
| Ait Kbaich et al., 2017 | Ouarzazate and Zagoura provinces in Morocco | Identification of Leishmania species causing CL in the two provinces | Eighty-one samples collected from patients | Eighty-one suspected CL patients. Four months to 56 years | 2015 & 2016 | Age, sex, travel history | Tissue sampling collected by dermal scraping from the 81 patients. DNA extraction and PCR–RFLP analysis. ITS1 PCR–RFLP of Leishmania species | Epidemiological pattern of CL in the studied areas appears to have changed, from a predominantly zoonotic CL caused by L. major to a polymorphic CL that can be due to either of the 3 leishmania species |
| Alkishe et al., 2017 | Different regions including North Africa | Effects of climate change on the spatial distribution of I. ricinus | Primary occurrence records for I. ricinus | Not applicable | 2050 and 2070 | Observed and projected Temperature, precipitation | Ecological niche modeling, future climate change scenarios | Present and future potential distributions of I. ricinus showed overlap in the region |
| Amro et al., 2017 | Lybia | Description of eco-epidemiological parameters of CL and spatiotemporal distributions of CL cases | A total of 312 CL patients | A total of 312 reported patients | The armed conflict period from January 2011 till December 2012 | Demographic data. Date of infection, age, gender place residence, number and location of lesions, and treatment response were documented. Household members previously diagnosed with CL, presence of rodents, and sandflies in the patient’s neighborhood | PCR–RFLP approach targeting the ITS1 region of the rDNA. Correlative modeling approach | Coastal regions have a higher level of risk. Future projection of CL until 2060 showed a trend of increasing incidence of CL in the northwestern part of the country. A spread along the coastal region and a possible emergence of new endemics in the north-eastern districts of the country |
| Bauer et al., 2017 | Thirty-two countries including Tunisia | To confirm prior findings that the larger the maximum monthly increase in solar insolation in springtime, the younger the age of onset of bipolar disorder | Data were collected from 5536 patients at 50 sites in 32 countries on six continents. Onset occurred at 456 locations in 57 countries | A total of 7392 patients with bipolar disorder | 2012, 2014, 2017 | Solar insolation, birth-cohort, family history, polarity of first episode, country physician density | Diagnosis of bipolar disorder according to DSM-IV criteria from a psychiatrist, direct questioning, reviewing records, or both | A large increase in springtime solar insolation may impact the onset of bipolar disorder, especially with a family history of mood disorders |
| Hmamouch et al., 2017 | Boulemane Province, Morocco | Distribution of CL in the study area | A total 1009 reported cases of CL | A total of 1009 reported patients | 2000 to 2015 | Poverty, vulnerability, population density, urbanization, and bioclimatic factors | PCR–RFLP method targeting the ITS1 of ribosomal DNA of leishmania. Ordinary least squares regression (OLSR) | Saharan microclimate, characterized by the presence of Leishmania major, was the most affected followed by semi-arid area where leishmania tropica was mostly identified |
| Horton et al., 2017 | Twenty hospitals in Egypt, Jordan, Oman, Qatar, and Yemen. Ten infectious disease hospitals in Egypt | Sentinel surveillance for SARI (Severe Acute Respiratory Infections) | Nasopharyngeal and oropharyngeal swabs | Patients meeting SARI case definitions | December 2007 through February 2014 | Climatic seasonality | Samples were tested by real time reverse transcriptase polymerase chain reaction (rtRT-PCR), MagMAX™ Pathogen RNA/DNA Kit. The chi-squared test. Logistic regression | Monthly variation, indicating seasonal differences in levels of infection, was observed for all pathogens. Viral respiratory pathogens are common among SARI patients in the Eastern Mediterranean Region |
| Talmoudi Khouloud et al., 2017 | Three rural areas in the governorate of Sidi Bouzid in Tunisia |
Relationship between ZCL occurrence and possible risk factors Apredicting model for ZCL's control and prevention purposes |
Monthly reported ZCL cases | The 1019 reported ZCL cases | 2009 to 2015 | Temperature, rainfall, relative humidity, wind speed, rodents’ density | Generalized additive model (GAM). Generalized additive mixed models (GAMM). Generalized cross-validation (GCV) score and residual test | Rodent density, average temperature, cumulative rainfall, and average relative humidity, with different time lags, all play role in sustaining and increasing the ZCL incidence in the region |
| H.Trablesi et al., 2017 | Tunisia | Frequency of allergic asthma attacks | The stages of asthma of patients | Forty-nine patients with allergic asthma hospitalized in Pneumology department. The environmental study was conducted for 30 patients in their homes | February 2014–October 2014 | Dust, humidity, cold, heat, climate change, seasonality | Epidemiological and environmental survey for fungal flora. Statistical analyses SPSS, Chi2tests, two tailed t-tests | The frequency of attacks was significantly associated with the seasonality, which was closely related to climate (P = 0.024) |
Climate Extremes
According to our search, 3 recent studies dealt with climate extremes and public health in some countries from North Africa. The study by Bellizzi and colleagues [76] was dedicated to the Eastern Mediterranean Region including Morocco, Tunisia, Egypt, and Libya and was performed from 1990 through 2019. It assessed the health consequence vulnerability index score and concluded that Libya is at risk of drought more than the other considered countries. The study highlighted that drought potentially has greater impact on people with underlying chronic conditions, such as respiratory diseases, as well as on people with disabilities. In Tunisia, two studies dealt with heat, seasonality, and public health. In 2020, Bettaieb and colleagues [77] conducted the study to estimate the impact of heat on total daily mortality in Tunis, the capital of Tunisia, during the summer season (May–October) over a 3-year period (2005 to 2007). They concluded that the daily mortality increased significantly by 2% for a 1 °C increase, above the breakpoint, in daily maximum temperature. The second study was conducted by Trablesi and colleagues [78]. They investigated the epidemiological and fungal environmental profile in 49 asthmatic patients, in 2014. Authors found that the frequency of attacks of asthma was significantly associated with seasonality, which is closely related to climate.
Vector-Borne Diseases
For the purpose of this review, we have studied 18 papers dealing with vector-borne diseases related to climate. Eight papers focus on leishmaniases studies, two on arboviral diseases, six on influenza and respiratory diseases, one on Tick-borne diseases, and one on Typhoid fever.
Leishmaniases
Leishmaniases are considered well-established in the North Africa region [79–89]. In Morocco, the disease occurs in unpredictable outbreaks in the south and the southeast regions of the Atlas Mountains and recently migrated from the west to the east of the country. In 2008, the Moroccan Ministry of Health reported 3414 CL cases caused by Leishmania major and Leishmania tropica (L. major and L. tropica which are species of parasites found in the Leishmania genus, and are associated with the CL) against 2028 cases in 2001. This shows a sharp increase in the incidence of leishmaniases in the country [90]. In 2020, a study led by Hakkour and colleagues investigated the relationship between climate and leishmaniasis in 9 provinces of Morocco [81]. Authors concluded that humidity was significant for CL and VL (caused by the species Leishmania donovani and Leishmania infantum) whereas temperature was significant only for VL. The incidence of leishmaniasis is higher during the wet season (October to April) than during the dry season (May to September), especially for CL. In 2017, Ait Kbaich and his team concluded that the epidemiological pattern of CL in two provinces from Morocco, Ouarzazate and Zagoura, has changed, from a predominantly Zoonotic CL (ZCL), transmissible from animals to humans caused by L. major, to a polymorphic CL that can be due to any one of the three Leishmania species (L. major, L. tropica, or L. infantum) [91]. In the same year, Hmamouch and colleagues examined the impact of several factors on the distribution of CL in Boulemane Province, Morocco. This province recorded a total of 1009 CL cases between the years 2000 and 2015. It is characterized by several microclimates, and Leishmania species [86]. The authors found that the Saharan microclimate, characterized by the presence of L. major, was the most affected and has recorded the highest number of CL cases followed by the semi-arid area where L. tropica was mostly identified. Another study was carried out in the prefecture of Meknes and showed the positive correlation of Leishmania sandfly abundance with temperature (r = 0.99) and the negative correlation with humidity and precipitation (r = − 0.87 and r = − 0.72) [89]. Another study compared the densities and genetic characteristics of Phlebotomus sergenti (kind of Leishmania sandfly) populations in two nearby localities, one in an anthroponotic cutaneous leishmaniasis (ACL, transmissible from humans) endemic area and another in a non-endemic area [92]. Authors found that P. sergenti density was significantly higher in the endemic area than in the non-endemic town. Bioclimatic differences were detected between both localities. Differences in both the density and the mitochondrial lineage of P. sergenti populations may explain the different epidemiological situation. P. sergenti in the locality without ACL cases seems sufficient to allow transmission but is not endemic; the non-endemic character could be justified by the absence of sufficient P. sergenti, which seems to prefer warmer and drier climates. Leishmaniases were also studied in Libya, by two researchers and their colleagues: Amro in 2017 and Dokhan in 2016 [80, 93]. The first study revealed that coastal regions have a higher level of risk due to more favorable conditions for the transmitting vectors and that future projection of CL until 2060 showed a trend of increasing incidence of CL in the Northwestern part of the country; a spread along the coastal region and a possible emergence of new endemic areas in the Northeastern districts of the country were highlighted as well. The second study, carried out in Al Rabta in the North-West of the country, indicated that the abundance of sandflies is influenced by climatological factors. It is directly related to temperature and relative humidity while inversely related to wind speed. In Tunisia, Talmoudi and his team built a regression model of ZCL cases to identify the relationship between ZCL occurrence and possible risk factors, and to develop a predicting model for ZCL’s control and prevention purposes [94]. The created model highlighted that rodent density, average temperature, cumulative rainfall, and average relative humidity, with different time lags, all play role in sustaining and increasing the ZCL incidence in the region. The abovementioned studies added to others out of the region confirm that the increase in leishmaniasis incidence and prevalence is mainly attributed to several risk factors mainly environmental conditions, socio-economic status, and demographic and human behaviors [95–97]. The climate change impacting different parameters and the population movements probably leads to alterations in the number, range, and density of the vectors and reservoirs and consequently may increase human exposure to infected sandflies [98].
Arboviral Diseases
Only a few studies focused on arboviral diseases in North Africa. Abdelkrim and colleagues have modeled and mapped the habitat suitability and the potential distribution of arboviruses vectors in Morocco using maximum entropy (Maxent) modeling under current climatic conditions. Results show that areas with maximum risk and high potential distribution were mainly located in the Northwestern and central parts of Morocco [99]. Some other authors focused on Zika as arbovirus; they investigated suitable temperatures for their transmission [27]. They used experimentally derived mechanistic model of Zika virus transmission by Aedes aegypti (the principal vector of Zika Virus) to map temperature-driven transmission risk in the North Africa and Middle East regions. Authors found that in the worst-case scenario, by 2050, over 270 million new people could be living in temperatures suitable for ZIKA virus transmission [27].
Influenza and Respiratory Diseases
Some studies were focused on Influenza in the North Africa region. Caini and colleagues have described the epidemiological characteristics, pattern of circulation, and geographical distribution of influenza B viruses and its B/Victoria and B/Yamagata lineages. They concluded that over 1.8 million influenza cases have occurred during 2000–2018 in thirty-one countries including Morocco from North Africa. The authors found that B/Yamagata was more frequent in the country that has a temperate climate [100]. In Egypt, two studies were found related to avian influenza. The study of Elsobky examined the degree to which a HPAI-H5N1 disease event is being influenced by climate variability as a potential risk factor [101]. Results revealed that the effect of climate variability differs according to the timing of the outbreak occurrence. Temperature and relative humidity could have both positive and negative effects on disease events. During the cold seasons, especially in the first quarter, higher minimum temperatures consistently show higher risks of disease occurrence, as this condition stimulates viral activity, while lower minimum temperatures support virus survival in the other quarters of the year with the highest negative effect in the third quarter. Relative humidity negatively affects the outbreak in the first quarter of the year as the humid weather does not support viral circulation, while the highest positive effect was found in the second quarter during which low humidity favors the disease event. The second study was from Salaheldin and his team [102]; the researchers explored factors contributing to the endemicity or re-emergence of A/H5N1 in Egypt; they showed that ambient temperature during winter months influenced the spread of the A/H5N1 avian flu virus.
Horton and colleagues conducted a sentinel surveillance for severe acute respiratory infections (SARI) from December 2007 through February 2014 at 20 hospitals in five countries including ten hospitals in Egypt from North Africa [103]. Nasopharyngeal and oropharyngeal swabs were collected from hospitalized patients meeting SARI case definitions and were analyzed for infection with influenza, respiratory syncytial virus (RSV), adenovirus (AdV), human metapneumovirus (hMPV), and human parainfluenza virus types 1 ± 3 (hPIV1-3). The surveillance observed monthly variation, indicating seasonal differences in levels of infection, for all pathogens. Influenza was the most common pathogen in Egypt.
Other studies involving the effect of climate are those related to COVID-19 cases and deaths. Two studies of that kind have interested our region. One study was conducted in 43 countries, including Morocco and Tunisia and showed an inverse relationship between COVID-19 per million and temperature in only two observations out of 12, indicating that factors other than temperature have greater influence on COVID-19 transmission [104]. The second study was conducted in 20 countries around the world, including Algeria, and showed that COVID-19 cases and deaths per million population were significantly lower in countries with high temperature and low humidity (the hottest countries), compared to countries with low temperature and high humidity (the coldest countries) [105].
Tick-Borne Diseases
The castor bean tick (I.Ricinus) is the most abundant and common tick in Europe [106]. It has a significant impact on public health [107]. It is the primary vector for Borrelia burgdorferi sensu lato spirochetes, causing Lyme borreliosis, and tick-borne encephalitis virus [106]. Moreover, many other tick-borne pathogens such as Borrelia miyamotoi, Rickettsia slovaca, Rickettsia helvetica, Rickettsia monacensis, Anaplasma phagocytophilum, Babesia divergens, Babesia venatorum, Babesia microti, Bartonella henselae, Coxiella burnetii, and Francisella tularensis have been detected in I. Ricinus [108, 109].
In North Africa, a study showed that current and potential future distributions of I.Ricinus largely overlap in parts of North Africa in the coastal regions of Morocco, Algeria, and Tunisia. The global warming is expected to increase the geographic distribution of I. Ricinus in the region and it could emerge in areas where they are currently absent, posing increased risks to human health in the region. For example, increasing temperatures lead to milder winter conditions, which prolongs the spring and fall seasons in Northern regions, making them more suitable for Ixodes Ricinus [110].
Typhoid Fever
In Meknes, a city of Morocco, Essayagh and his team showed that climatic conditions could facilitate the invasion of certain pathogens [111]. Authors showed that the average incidence of typhoid fever was highest during the dry season (5 cases per 100,000 inhabitants in summer and 2.8 cases per 100,000 inhabitants in spring).
Mental Disorders
In 2017, Bauer and colleagues explored the relationship between solar insolation in spring time and age of onset of bipolar I disorder [112]. They collected data from 5536 patients in 32 countries, including Tunisia, and applied the diagnosis of bipolar disorder according to DSM-IV criteria from a psychiatrist. Authors confirmed that the larger the maximum monthly increase in solar insolation in spring time, the younger the age of onset of bipolar disorder. In 2019, the same authors studied the association between solar insolation and the history of suicide attempts in bipolar I disorder using the same sample from the same region and the generalized estimating equations (GEE) statistical technique. Suicide-related data were available for 3365 patients from 310 onset locations in 51 countries including Tunisia. A large number of 1047 (31.1%) had a history of suicide attempt. The study showed that living in locations with large changes in solar insolation between winter and summer may be associated with increased suicide attempts in patients with bipolar disorder [113].
Discussion
We conducted a systematic review of the link between air pollution, climate, and human health. We identified 34 appropriate manuscripts published since 2016. Among these manuscripts, nine studied air pollution and health, and 24 investigated climate and health.
To the best of our knowledge, this is the only systematic review of published studies on climate and air pollution and their relationship with health in North Africa in the last 6 years.
Air Pollution and Public Health
Even though the North Africa region is missing accurate source apportionment studies, the location of the region in the North of the Saharan desert raised awareness about the region’s vulnerability to particulate pollution [30]. Thus, most of the studies related to air pollution and health in the region focused on PM10/PM2.5. These studies showed that PM pollution levels induce deaths due to pulmonary and cardiovascular diseases [39], ischemic heart disease, stroke, and lung cancer [38], and cause preterm births as well [43]. The extent of the air pollution impact in the region depends on the density of population in the different studied areas. Other pollutants such as O3, SO2, and NO2 were less examined in the literature and it is necessary to mention that they are in need of more attention. Indeed, the paucity of data for environmental and epidemiology makes it difficult to estimate the health effects of air pollution, particularly the long-term health effects of exposure which are difficult to detect. As a result, assumptions for estimating long-term air pollution exposure are frequently adopted. However, we can use some alternative data such as model outputs, satellite data [114], and data from some institutions such as the Institute for Health Metrics and Evaluation.
Health outcomes, mortality, and short-term exposures linked to PM have received the most attention. More research on the effects of exposure to all pollutants is required. Indeed, many studies were led outside the region to analyze the effect related to long-term exposure to pollution. For instance, people who have been exposed to PM for long-term periods have a significantly higher rate of cardiovascular and respiratory diseases, cerebrovascular disease, and mortality [115]. Also, chronic respiratory diseases (CRD), particularly asthma and chronic obstructive pulmonary disease (COPD), have been well-documented in developed countries. Hence, in comparison to developed countries, the burden related to these diseases is less documented in North Africa [55, 116].
As it is mentioned in Table 1, the methodology adopted in most North Africa studies used simple statistical methods or special tools (AirQ) to quantify the relationships between some diseases and PM pollutants. Hence, more studies are required in every country in terms of health impact assessment. A diversity of tools can be employed such as AIRQ2.2, AirCounts TM, BenMAP-CE, and LEAP-IBC. It is worth mentioning that not all of these tools are flexible in scope, thus each technique should be chosen according to the predefined study resolution (local, national, regional, global) and the tool’s configuration needs. Other factors define the choice of the most suitable method, talking of a trade-off between technical and operational characteristics. Overall, the AirQ tool is more frequently adopted in North Africa studies to explore the relationships between the short-term and long-term exposure to atmospheric pollutants mainly PM and morbidity and mortality cases. This tool has a global scope and therefore can be used for any city, country, or region. It is an open source software developed by the WHO. Like other methods of risk assessment, this method requires basically four inputs: (1) the number of people at risk, (2) the baseline health statistics, (3) the concentration–response association, and (4) the air pollution concentration [39]. The application of the Health Impact Assessment of Air Pollution (HIA-AP) method relies on the Concentration–response functions (CRF) to provide the number of health events that could be either prevented by reducing pollutant levels or increased due to increased concentrations of air pollutants.
To evaluate the impact of various levels of pollutants on health, many studies, according to pollution levels, extrapolated the shape of the concentration–response function generated from historical associations primarily conducted in the USA and Europe. The shape of the prior studies’ Concentration–Response functions (CRF) was almost linear, log-linear, and log–log. Understanding the true nature of the Concentration–Response functions between each pollutant and adverse health effects is a worthy endeavor.
None of the areas in the studied regions possess a CRF. Consequently, CRFs are frequently derived from epidemiological studies in other part of the world that differ basically on pollution levels, chemical composition, and health care system. Assumptions are often made which can lead to more uncertainty in the results. Hence, the absence of direct epidemiological evidence is an important limitation and it seems highly desirable for future work to define specific CRFs related to the studied areas. To assess PM2.5 pollutant, the IER (Integrated Exposure Response) curve function is generally well-used in the literature. This function relates PM2.5 to five causes of death, namely, ischemic heart disease, cerebrovascular disease, chronic obstructive pulmonary diseases, lung cancer, and acute lower respiratory infections. Consequently, it quantifies adverse health impact especially, where population-specific risk information is unavailable.
Results presented in this review display little evidence on the health impacts of air pollution. Regarding other studies outside the study region, the finding results underestimate the total mortality and morbidity related to air pollution due to the assumptions of the used models, namely, susceptible and non-susceptible populations, the thresholds of pollution effects, and health endpoints. Indeed, the literature covers a few ranges of possible adverse health effects linked to different groups of the population. The most reported cases are the number of deaths, hospital admission for pulmonary and cardiovascular diseases, and preterm birth. All of these findings indicate that additional efforts are required to close the gap. This review showed that air pollution and health studies did not equally cover all the countries within the region. In Libya, no similar research has been found. Furthermore, no studies linking air pollution episodes and health emergencies were found. Therefore, future research should fill this gap in order to improve health system preparedness and contribute in setting an alert system for a quick decision and air pollution management in the North Africa region.
The Impact of Climate on Public Health
To review studies related to climate and public health, we focused on climate extremes, vector-borne diseases, and mental disorders. Given the specificity of each study with its own parameters, we have opted in this part for a quantitative comparison rather than a qualitative one. Considering the number of studies related to climate and health taken into account from 2016 to 2021, we note on the one hand that Morocco ranks first with 50% of studies. On the other hand, 50% of total researches have focused on leishmaniasis.
Tunisia is the country studied in the three studies found in Northern Africa, in relation to climate extremes and health, during the selected period. These studies were interested in heat, seasonality, and drought. The study about drought included Morocco, Egypt, and Libya. However, the North African region lacks studies on climate extremes and public health, particularly the effects of floods, forest fires, and heat waves. The health impact of these phenomena was well-studied in many regions outside North Africa. Graeme Horton et al. [117] focused on the health impact of heat, drought, and dryness for aging Australians in rural areas. Carla Stanke et al. [118] used data from the following sources: MEDLINE, CINAHL, Embase, PsychINFO, and Cochrane Collection to investigate the health impact of drought in all countries.
Our work showed that most researches have focused on vector-borne diseases, especially those related to leishmaniasis. More than 50% of the studies related to leishmaniasis were performed in Morocco. The rest were carried out in Libya and Tunisia. Egypt and Algeria lack leishmaniasis-related studies during the study period. Even though the latter is a common health problem to all countries of the region, special areas are being affected within every country according to weather and climate conditions. Future climate change may modify areas of leishmaniasis and change its dispersion map in the region. More studies are needed to explore this aspect.
In the study region, we considered two studies on arboviral diseases in North Africa. One of them focused on Morocco. Concerning the studies related to influenza and respiratory diseases, Egypt was ranked first with 50% of the studies considered followed by Morocco (33%) then Algeria and Tunisia by studies related to COVID-19. Tick-borne diseases and typhoid fever have recorded each of them only one study and mental disorders have been considered only in Tunisia. In the other regions, a study presented some research on nutrition-related effects [119]; water-related diseases [120]; air- and dust-related diseases [121]; vector-borne diseases [122]; mental health effects [123]; and other health effects (including forest fires, migration effects, and infrastructure damage) [124]. Most health effects are indirect because of the link to other mediating circumstances such as loss of livelihoods. Some papers have studied the impact of drought on various vector-borne diseases such as [125]. Others have examined the relationship between climate variability and transmission of vector and rodent-borne diseases including malaria, dengue, Ross River virus infection, and hemorrhagic fever with renal syndrome [126]. The review of these papers showed that existing analysis methods have limitations. Research with different populations in various climatic/ecological regions using appropriate statistical models is needed. Walte Leal Filho et al. [127] agreed on the existence of important impacts of climate change on health and mentioned that climate change contributes to the spread of vector-borne diseases. The health effects of leishmaniasis [128], arboviral diseases [129], and influenza [130] have been studied and developed in different regions of the world. Shigemura and Kurosawa [131] studied the mental health impact of the COVID-19 pandemic. During our work, it was clear to us that researchers were interested in the most challenging pathogens in every country from the study region. Research related to other pathogens linked to climate and impacted by climate change is still needed.
Studies related to the joint impact of air pollution and climate on public health are missing as well. In fact, in studying the impacts of climate and air quality on health, it remains unclear whether air pollutants are confounders or effect modifiers of the association between the climate factor and mortality. In the North Africa region, only 2 studies tried to discover/control this association. Bettaieb et al., mentioned earlier [77], tried to estimate the impact of heat on daily all-cause mortality in Tunis while controlling for the effects of air pollution considering NO2. He has found that increase in NO2 was associated with a significant increase in daily mortality, which highlights the combined effect of heat and NO2 on daily mortality in the study area. Besides, a study carried out in Morocco [132] analyzed the potential concurrence of heat waves and O3 episodes and their human health impact based on the AQHI (Air Quality Heat Index) and the Heat Index. In light of the latter study results, local sources and large-scale atmospheric circulation are considered as the main sources behind the occurrence of these events. Overall, studies of the joint impact of air pollution and climate on public health are limited and further researches need to be carried out to fill this gap. Every further study linking climate/climate change to health will help decision makers to set appropriate adaptation strategies mainly for the health care system.
Future research directions could focus on studies related to air pollution and climate and their impact on health to fill this scientific gap. More research should be done in Algeria and Libya. Studies of health co-benefits; identification of vulnerable populations; prioritization of resource allocation, capacity, and skill building; development of appropriate risk communication strategies; and community health education packages for vulnerable communities in the context of climate change are desirable throughout the study region.
Conclusion
This work provides a timely guide to existing evidence, missing gaps, and potential next steps to researches related to climate, air pollution, and health in the North Africa region. The results of this review highlighted the great lack of scientific studies investigating the interaction between air pollution, climate, and health in North Africa in recent years. Each country in the study area prioritizes studies according to its own challenges in the field. However, the majority of studies relates to climate and vector-borne diseases especially leishmaniasis.
Air quality can affect climate change, climate can affect air quality, and both can directly or indirectly affect health. Given the importance of air pollution as a global risk factor for premature death and diseases, and the direct impact of climate on health and the strong link between climate and air quality, a huge need exists nowadays for studies discovering this link. Yet, these types of studies remain limited in North Africa. A direction in this research area should be taken in future scientific researches.
The effect of climate and damage caused by drought, flooding of infrastructure, forest fires, etc. can be increased by extremely hot weather as well. Assessing the impact of these extremes on health is a needed research direction in our region. Researches can provide tools to help decision makers manage the risks and emergencies in the health sector.
As for air quality, future studies using new tools dedicated to the assessment of air pollution are highly recommended. Furthermore, alternative solutions such as satellite data are strongly proposed in a future study to solve the problem of data scarcity.
The North African region also lacks empirical research to identify and quantify causal mechanisms as well as projection studies that encompass multiple climate-related parameters and outcomes. At the same time, obtaining results at spatial and temporal scales relevant to decision makers will remain a significant challenge.
Author Contribution
Conceptualization: Imane Sekmoudi, Oumaima Bouakline, and Kenza Khomsi; data curation: Imane Sekmoudi, Oumaima Bouakline, and Kenza Khomsi; formal analysis: Imane Sekmoudi, Oumaima Bouakline, and Kenza Khomsi; methodology: Imane Sekmoudi, Oumaima Bouakline, and Kenza Khomsi; supervision: Laila Idrissi, Youssef El merabet, and Zineb Souhaili; writing—review and editing: Imane Sekmoudi, Oumaima Bouakline, and Kenza Khomsi; cartography: Mohamed El jarmouni.
Declarations
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Conflict of Interest
The authors declare no competing interests.
Footnotes
This article is part of the Topical Collection on Global Environmental Health and Sustainability
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Sekmoudi Imane, Email: imane.sekmoudi@gmail.com.
Bouakline Oumaima, Email: Bouaklineoumaima1@gmail.com.
Khomsi Kenza, Email: k.khomsi@gmail.com.
Idrissi Laila, Email: idrissikam@gmail.com.
El merabet Youssef, Email: youssef.elmerabet@uit.ac.ma.
Souhaili Zineb, Email: z.souhaili@gmail.com.
El jarmouni Mohamed, Email: med.eljarmouni@gmail.com.
References
- 1.de Sousa TCM, Amancio F, De Sousa Hacon S, Barcellos C. Climate-sensitive diseases in Brazil and the world: systematic reviewEnfermedades sensibles al clima en Brasil y el mundo: revisión sistemática. Rev Panam Salud Publica Pan Am J Public Heal. 2018 doi: 10.26633/RPSP.2018.85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Iñiguez C, Royé D, Tobías A. Contrasting patterns of temperature related mortality and hospitalization by cardiovascular and respiratory diseases in 52 Spanish cities. Environ Res. 2021 doi: 10.1016/j.envres.2020.110191. [DOI] [PubMed] [Google Scholar]
- 3.Abdolahnejad A, Mousavi SH, Sofizadeh A, Jafari N, Shiravand B. Climate change and distribution of zoonotic cutaneous leishmaniasis (ZCL) reservoir and vector species in central Iran. Model Earth Syst Environ. 2021 doi: 10.1007/s40808-020-00860-4. [DOI] [Google Scholar]
- 4.Caldwell JM, LaBeaud AD, Lambin EF, et al. Climate predicts geographic and temporal variation in mosquito-borne disease dynamics on two continents. Nat Commun. 2021 doi: 10.1038/s41467-021-21496-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Clayton S. Climate change and mental health. Curr Environ Heal Reports. 2021 doi: 10.1007/s40572-020-00303-3. [DOI] [PubMed] [Google Scholar]
- 6.Indhumathi K, Sathesh Kumar K. A review on prediction of seasonal diseases based on climate change using big data. Mater Today Proc. 2020 doi: 10.1016/j.matpr.2020.08.517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.El-Sayed A, Kamel M. Climatic changes and their role in emergence and re-emergence of diseases. Environ Sci Pollut Res. 2020 doi: 10.1007/s11356-020-08896-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cohen AJ, Brauer M, Burnett R, et al. Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: an analysis of data from the Global Burden of Diseases Study 2015. Lancet. 2017 doi: 10.1016/S0140-6736(17)30505-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Su W, Liu T, Geng X, Yang G. Seasonal pattern of influenza and the association with meteorological factors based on wavelet analysis in Jinan City, Eastern China, 2013–2016. PeerJ. 2020 doi: 10.7717/peerj.8626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Li Q, Guan X, Wu P, et al. Early transmission dynamics in Wuhan, China, of novel Coronavirus–infected pneumonia. N Engl J Med. 2020 doi: 10.1056/nejmoa2001316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Paules CI, Marston HD, Fauci AS. Coronavirus infections-more than just the common cold. JAMA - J Am Med Assoc. 2020 doi: 10.1001/jama.2020.0757. [DOI] [PubMed] [Google Scholar]
- 12.Kuiken T, Fouchier RAM, Schutten M, et al. Newly discovered coronavirus as the primary cause of severe acute respiratory syndrome. Lancet. 2003 doi: 10.1016/S0140-6736(03)13967-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Peiris JSM, Lai ST, Poon LLM, Guan Y, Yam LYC, Lim W. Coronavirus as a possible cause of severe acute respiratory syndrome. J Tepecik Educ Res Hosp. 2003 doi: 10.5222/terh.2003.26734. [DOI] [Google Scholar]
- 14.Moriyama M, Hugentobler WJ, Iwasaki A. Seasonality of respiratory viral infections. Annu Rev Virol. 2020 doi: 10.1146/annurev-virology-012420-022445. [DOI] [PubMed] [Google Scholar]
- 15.Ruiz-postigo JA, Ruiz-postigo JA, Africa N, Asia W, Asia E. Global leishmaniasis surveillance 2017–2018 and first report on 5 additional indicators – surveillance mondiale de la leishmaniose, 2017–2018 premier rapport sur 5 indicateurs supplémentaires. Wkly Epidemiol Rec Relev épidémiologique Hebd. 2020;95:265–279. [Google Scholar]
- 16.Garrido-Jareño M, Sahuquillo-Torralba A, Chouman-Arcas R, et al. Cutaneous and mucocutaneous leishmaniasis: experience of a Mediterranean hospital. Parasit Vectors. 2020 doi: 10.1186/s13071-020-3901-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Inceboz T. Epidemiology and ecology of leishmaniasis. Curr Top Neglected Trop Dis. 2019 doi: 10.5772/intechopen.86359. [DOI] [Google Scholar]
- 18.Haeberlein S, Fischer D, Thomas SM, Schleicher U, Beierkuhnlein C, Bogdan C. First assessment for the presence of phlebotomine vectors in bavaria, southern germany, by combined distribution modeling and field surveys. PLoS ONE. 2013 doi: 10.1371/journal.pone.0081088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Söbirk SK, Inghammar M, Collin M, Davidsson L. Imported leishmaniasis in Sweden 1993–2016. Epidemiol Infect. 2018 doi: 10.1017/S0950268818001309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wall EC, Watson J, Armstrong M, Chiodini PL, Lockwood DN. Short report: epidemiology of imported cutaneous leishmaniasis at the hospital for tropical diseases, London. United Kingdom: use of polymerase chain reaction to identify the species Am J Trop Med Hyg. 2012 doi: 10.4269/ajtmh.2012.10-0558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.WHO (2021) Leishmaniasis.
- 22.Chalghaf B, Chemkhi J, Mayala B, Harrabi M, Benie GB, Michael E, Ben Salah A. Ecological niche modeling predicting the potential distribution of leishmania vectors in the Mediterranean basin: impact of climate change. Parasit Vectors. 2018 doi: 10.1186/s13071-018-3019-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ready PD. Leishmaniasis emergence in Europe. Eurosurveillance. 2010 doi: 10.2807/ese.15.10.19505-en. [DOI] [PubMed] [Google Scholar]
- 24.Pereira Cabral B, da Graça Derengowski Fonseca M, Mota FB. Long term prevention and vector control of arboviral diseases: what does the future hold? Int J Infect Dis. 2019 doi: 10.1016/j.ijid.2019.10.002. [DOI] [PubMed] [Google Scholar]
- 25.Gould E, Pettersson J, Higgs S, Charrel R, de Lamballerie X. Emerging arboviruses: why today? One Heal. 2017 doi: 10.1016/j.onehlt.2017.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Oscar Júnior AC, de Assis MF. Climate change and risk of arboviral diseases in the state of Rio de Janeiro (Brazil) Theor Appl Climatol. 2021 doi: 10.1111/gcb.15384. [DOI] [Google Scholar]
- 27.Ryan SJ, Carlson CJ, Tesla B, Bonds MH, Ngonghala CN, Mordecai EA, Johnson LR, Murdock CC. Warming temperatures could expose more than 13 billion new people to Zika virus risk by 2050. Glob Chang Biol. 2021 doi: 10.1111/gcb.15384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Thoma MV, Rohleder N, Rohner SL. Clinical ecopsychology: the mental health impacts and underlying pathways of the climate and environmental crisis. Front Psychiatry. 2021 doi: 10.3389/fpsyt.2021.675936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Antillón M, Warren JL, Crawford FW, Weinberger DM, Kürüm E, Pak GD, Marks F, Pitzer VE. The burden of typhoid fever in low- and middle-income countries: a meta-regression approach. PLoS Negl Trop Dis. 2017 doi: 10.1371/journal.pntd.0005376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Neuzil KM, Pollard AJ, Marfin AA. Introduction of typhoid conjugate vaccines in Africa and Asia. Clin Infect Dis. 2019 doi: 10.1093/cid/ciy878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gilbert L. The impacts of climate change on ticks and tick-borne disease risk. Annu Rev Entomol. 2021 doi: 10.1146/annurev-ento-052720-094533. [DOI] [PubMed] [Google Scholar]
- 32.Negi T, Kandari LS, Arunachalam K. Update on prevalence and distribution pattern of tick-borne diseases among humans in India: a review. Parasitol Res. 2021 doi: 10.1007/s00436-021-07114-x. [DOI] [PubMed] [Google Scholar]
- 33.Jore S, Vanwambeke SO, Viljugrein H, et al. Climate and environmental change drives Ixodes ricinus geographical expansion at the northern range margin. Parasit Vectors. 2014 doi: 10.1186/1756-3305-7-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gilbert L, Aungier J, Tomkins JL. Climate of origin affects tick (Ixodes ricinus) host-seeking behavior in response to temperature: implications for resilience to climate change? Ecol Evol. 2014 doi: 10.1002/ece3.1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rogulina M. Faunal and ecological features of the distribution of ixodid ticks in the Kursk Region. Theory Pract Parasit Dis Control. 2021 doi: 10.31016/978-5-6046256-1-3.2021.22.425-430. [DOI] [Google Scholar]
- 36.McMichael AJ. Globalization, climate change, and human health. N Engl J Med. 2013 doi: 10.1056/nejmra1109341. [DOI] [PubMed] [Google Scholar]
- 37.Can G, Şahin Ü, Sayılı U, et al. Excess mortality in istanbul during extreme heat waves between 2013 and 2017. Int J Environ Res Public Health. 2019 doi: 10.3390/ijerph16224348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Vaidyanathan A, Malilay J, Schramm P, Saha S. Heat-related deaths — United States, 2004–2018. MMWR Morb Mortal Wkly Rep. 2020 doi: 10.15585/mmwr.mm6924a1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Martínez-Solanas È, Basagaña X. Temporal changes in temperature-related mortality in Spain and effect of the implementation of a Heat Health Prevention Plan. Environ Res. 2019 doi: 10.1016/j.envres.2018.11.006. [DOI] [PubMed] [Google Scholar]
- 40.Kovats S, Hales S, Campbell-Lendrum D, Rocklov J, Honda Y, Lloyd S. Global risk assessment of the effect of climate change on selected causes of death in 2030s and 2050s. ISEE Conf Abstr. 2015 doi: 10.1289/isee.2015.2015-1204. [DOI] [Google Scholar]
- 41.Mathioudakis DG, Mathioudakis AG, Mathioudakis GA. Climate change and human health. Arch Hell Med. 2020 doi: 10.37547/tajmspr/volume02issue10-10. [DOI] [Google Scholar]
- 42.Pallagiano RA and C (2018) Climate change and air pollution: the impact on human health in developed and developing countries. Clim. Chang. Air Pollut.
- 43.Eguiluz-Gracia I, Mathioudakis AG, Bartel S, et al. The need for clean air: the way air pollution and climate change affect allergic rhinitis and asthma. Allergy Eur J Allergy Clin Immunol. 2020 doi: 10.1111/all.14177. [DOI] [PubMed] [Google Scholar]
- 44.Manisalidis I, Stavropoulou E, Stavropoulos A, Bezirtzoglou E. Environmental and health impacts of air pollution: a review. Front Public Heal. 2020 doi: 10.3389/fpubh.2020.00014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Forouzanfar MH, Alexander L, Anderson HR, et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks in 188 countries, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet (London, England) 2015;386:2287–2323. doi: 10.1016/S0140-6736(15)00128-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Landrigan PJ, Fuller R, Acosta NJR, et al (2018) The Lancet Commission on pollution and health. 391:2018 [DOI] [PubMed]
- 47.Khomenko S, Cirach M, Pereira-barboza E, Mueller N, Barrera-gómez J, Rojas-rueda D, Hoogh K De, Hoek G, Nieuwenhuijsen M (2016) Premature mortality due to air pollution in European cities : a health impact assessment.10.1016/S2542-5196(20)30272-2 [DOI] [PubMed]
- 48.Hoek G, Brunekreef B, Goldbohm S, Fischer P, Van Den Brandt PA. Association between mortality and indicators of traffic-related air pollution in the Netherlands: a cohort study. Lancet. 2002 doi: 10.1016/S0140-6736(02)11280-3. [DOI] [PubMed] [Google Scholar]
- 49.Pope CA, Dockery DW. Health effects of fine particulate air pollution: lines that connect. J Air Waste Manag Assoc. 2006 doi: 10.1080/10473289.2006.10464485. [DOI] [PubMed] [Google Scholar]
- 50.Mesquita SR, Van Drooge BL, Reche C, Guimarães L, Grimalt JO, Barata C, Piña B. Toxic assessment of urban atmospheric particle-bound PAHs: relevance of composition and particle size in Barcelona (Spain) Environ Pollut. 2014 doi: 10.1016/j.envpol.2013.09.034. [DOI] [PubMed] [Google Scholar]
- 51.Bettaieb J, Toumi A, Leffondre K, Chlif S, Ben Salah A. High temperature effect on daily all-cause mortality in Tunis 2005–2007. Rev Epidemiol Sante Publique. 2020;68:37–43. doi: 10.1016/j.respe.2019.09.007. [DOI] [PubMed] [Google Scholar]
- 52.Waha K, Krummenauer L, Adams S, et al. Climate change impacts in the Middle East and Northern Africa (MENA) region and their implications for vulnerable population groups. Reg Environ Chang. 2017 doi: 10.1007/s10113-017-1144-2. [DOI] [Google Scholar]
- 53.Linares C, Díaz J, Negev M, Martínez GS, Debono R, Paz S. Impacts of climate change on the public health of the Mediterranean Basin population - current situation, projections, preparedness and adaptation. Environ Res. 2020 doi: 10.1016/j.envres.2019.109107. [DOI] [PubMed] [Google Scholar]
- 54.Taha T. Climate change and potential impact on disease: what are the public health agenda? Saudi J Med Med Sci. 2016 doi: 10.4103/1658-631x.178285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sylla FK, Faye A, Fall M, Tal-Dia A. Air pollution related to traffic and chronic respiratory diseases (Asthma and COPD) in Africa. Health (Irvine Calif) 2017;09:1378–1389. [Google Scholar]
- 56.Sadiq A, El Fazziki A, Ouarzazi J, Sadgal M. Towards an agent based traffic regulation and recommendation system for the on-road air quality control. Springerplus. 2016;5:1604. doi: 10.1186/s40064-016-3282-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Inchaouh M, Tahiri M. Air pollution due to road transportation in Morocco : evolution and impacts. J Multidiscip Eng Sci Technol. 2017;4:7547–7552. [Google Scholar]
- 58.Khomsi K, Najmi H, Chelhaoui Y, Souhaili Z. The contribution of large-scale atmospheric patterns to pm10 pollution: the new saharan oscillation index. Aerosol Air Qual Res. 2020 doi: 10.4209/aaqr.2019.08.0401. [DOI] [Google Scholar]
- 59.Inchaouh M, Tahiri M, Johra B El, Abboubi R (2017) State of ambient air quality in Marrakech city (MOROCCO) over the period 2009 – 2012. 12:99–106
- 60.Zghaid M, Noack Y, Bounakla M, Benyaich F Pollution atmosphérique particulaire dans la ville de Kenitra ( Maroc ) Atmospheric particulate pollution in Kenitra ( Morocco ). 313–324
- 61.Bouh HA, Techniques N, Bounakhla M, Techniques N, Noack Y (2010) Caractérisations de la matière particulaire Atmosphérique dans la ville de Meknès ATMOSPHERIC PARTICULATE MATTER CHARACTERISATION IN MEKNES CITY.
- 62.Bounakhla M, Techniques N, Noack Y, Mounia T, Techniques N (2016) Chemical characterization and origin of suspended atmospheric particles in chemical characterization and origin of suspended atmospheric particles in Meknes city in Morocco.
- 63.Talbi A, Kerchich Y, Kerbachi R, Boughedaoui M. Assessment of annual air pollution levels with PM1, PM2.5, PM10 and associated heavy metals in Algiers Algeria. Environ Pollut. 2018 doi: 10.1016/j.envpol.2017.09.041. [DOI] [PubMed] [Google Scholar]
- 64.Bencharif-Madani F, Ali-Khodja H, Kemmouche A, Terrouche A, Lokorai K, Naidja L, Bouziane M. Mass concentrations seasonal variations chemical compositions and element sources of PM10 at an urban site in Constantine northeast Algeria. J Geochemical Explor. 2019;206:106356. doi: 10.1016/j.gexplo.2019.106356. [DOI] [Google Scholar]
- 65.Barhoumi B, Tedetti M, Heimbürger-Boavida L-E, Tesán Onrubia JA, Dufour A, Doan QT, Boutaleb S, Touil S, Scippo M-L. Chemical composition and in vitro aryl hydrocarbon receptor-mediated activity of atmospheric particulate matter at an urban agricultural and industrial site in North Africa (Bizerte Tunisia) Chemosphere. 2020;258:127312. doi: 10.1016/j.chemosphere.2020.127312. [DOI] [PubMed] [Google Scholar]
- 66.Shaltout AA, Boman J, Hassan SK, Abozied AM, Al-Ashkar EA, Abd-Elkader OH, Yassin MA, Al-Tamimi JH. Elemental Composition of PM(2.5) Aerosol in a residential-industrial area of a Mediterranean megacity. Arch Environ Contam Toxicol. 2020;78:68–78. doi: 10.1007/s00244-019-00688-9. [DOI] [PubMed] [Google Scholar]
- 67.Benaissa F, Maesano CN, Alkama R, Annesi-Maesano I. Short-term health impact assessment of urban PM10 in Bejaia city (Algeria) Can Respir J. 2016 doi: 10.1155/2016/8209485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Croitoru L, Sarraf M. Estimating the health cost of air pollution: the case of Morocco. J Environ Prot (Irvine Calif) 2017 doi: 10.4236/jep.2017.810069. [DOI] [Google Scholar]
- 69.Wheida A, Nasser A, El Nazer M, Borbon A, Abo El Ata GA, Abdel Wahab M, Alfaro SC. Tackling the mortality from long-term exposure to outdoor air pollution in megacities: lessons from the Greater Cairo case study. Environ Res. 2018 doi: 10.1016/j.envres.2017.09.028. [DOI] [PubMed] [Google Scholar]
- 70.Ghennam K, Attou F, Abdoun F. Impact of atmospheric pollution on asthma and bronchitis based on lichen biomonitoring using IAP, IHI and GIS in Algiers Bay (Algeria) Environ Monit Assess. 2021;193:198. doi: 10.1007/s10661-021-08965-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Mohammed AM, Ibrahim YH, Saleh IA. Estimation of hospital admission respiratory disease cases attributed to exposure to SO(2) and NO(2) in two different sectors of Egypt. Afr Health Sci. 2019;19:2892–2905. doi: 10.4314/ahs.v19i4.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Malley CS, Kuylenstierna JCI, Vallack HW, Henze DK, Blencowe H, Ashmore MR. Preterm birth associated with maternal fine particulate matter exposure: a global, regional and national assessment. Environ Int. 2017;101:173–182. doi: 10.1016/j.envint.2017.01.023. [DOI] [PubMed] [Google Scholar]
- 73.Marchetti S, Hassan SK, Shetaya WH, et al. Seasonal variation in the biological effects of PM2 5 from greater cairo. Int J Mol Sci. 2019 doi: 10.3390/ijms20204970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Boussetta N, Abedelmalek S, Khouloud A, Ben Anes A, Souissi N. Does red orange juice supplementation has a protective effect on performance, cardiovascular parameters, muscle damage and oxidative stress markers following the Yo-Yo Intermittent Recovery Test Level-1 under polluted air? Int J Environ Health Res. 2020;30:630–642. doi: 10.1080/09603123.2019.1614155. [DOI] [PubMed] [Google Scholar]
- 75.Boussetta N, Abedelmalek S, Aloui K, Souissi N. The effect of air pollution on diurnal variation of performance in anaerobic tests, cardiovascular and hematological parameters, and blood gases on soccer players following the Yo-Yo Intermittent Recovery Test Level-1. Chronobiol Int. 2017;34:903–920. doi: 10.1080/07420528.2017.1325896. [DOI] [PubMed] [Google Scholar]
- 76.Bellizzi S, Lane C, Elhakim M, Nabeth P. Health consequences of drought in the WHO Eastern Mediterranean Region: hotspot areas and needed actions. Environ Heal A Glob Access Sci Source. 2020 doi: 10.1186/s12940-020-00665-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Bettaieb J, Toumi A, Leffondre K, Chlif S, Ben Salah A. High temperature effect on daily all-cause mortality in Tunis 2005–2007. Rev Epidemiol Sante Publique. 2020 doi: 10.1016/j.respe.2019.09.007. [DOI] [PubMed] [Google Scholar]
- 78.Trablesi H, Hadrich I, Neji S, Fendri N, Ghorbel D, Makni F, Ayadi H, Kammoun S, Ayadi A. Environmental and molecular study of fungal flora in asthmatic patients. J Mycol Med. 2018;28:180–185. doi: 10.1016/j.mycmed.2017.09.001. [DOI] [PubMed] [Google Scholar]
- 79.Chahed MK, Bellali H, Ben Jemaa S, Bellaj T. Psychological and psychosocial consequences of zoonotic cutaneous leishmaniasis among women in Tunisia: preliminary findings from an exploratory study. PLoS Negl Trop Dis. 2016 doi: 10.1371/journal.pntd.0005090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Dokhan MR, Kenawy MA, Doha SA, El-Hosary SS, Shaibi T, Annajar BB. Entomological studies of phlebotomine sand flies (Diptera: Psychodidae) in relation to cutaneous leishmaniasis transmission in Al Rabta North West of Libya. Acta Trop. 2016 doi: 10.1016/j.actatropica.2015.11.004. [DOI] [PubMed] [Google Scholar]
- 81.Hakkour M, Hmamouch A, Mahmoud El Alem M, Bouyahya A, Balahbib A, El Khazraji A, Fellah H, Sadak A, Sebti F. Risk factors associated with leishmaniasis in the most affected provinces by leishmania infantum in Morocco. Interdiscip Perspect Infect Dis. 2020 doi: 10.1155/2020/6948650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Barhoumi W, Fares W, Cherni S, Derbali M, Dachraoui K, Chelbi I, Ramalho-Ortigao M, Beier JC, Zhioua E. Changes of sand fly populations and leishmania infantum infection rates in an irrigated village located in arid central Tunisia. Int J Environ Res Public Health. 2016 doi: 10.3390/ijerph13030329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Daoudi M, Boussaa S, Hafidi M, Boumezzough A. Potential distributions of phlebotomine sandfly vectors of human visceral leishmaniasis caused by leishmania infantum in Morocco. Med Vet Entomol. 2020 doi: 10.1111/mve.12434. [DOI] [PubMed] [Google Scholar]
- 84.Zouirech M, Rhajaoui M, Faraj C, El Guamri Y, Amahmid O, El Hachimi MY, Bouhout S, El Kharrim K, Belghyti D. Entomological inventory of phlebotomine sand flies and epidemiological study of leishmaniasis in afourer azilal province high Atlas Morocco. Med Sante Trop. 2018 doi: 10.1684/mst.2018.0840. [DOI] [PubMed] [Google Scholar]
- 85.Karmaoui A. Seasonal distribution of Phlebotomus papatasi, vector of zoonotic cutaneous leishmaniasis. Acta Parasitol. 2020 doi: 10.2478/s11686-020-00201-6. [DOI] [PubMed] [Google Scholar]
- 86.Hmamouch A, El Alem MM, Hakkour M, Amarir F, Daghbach H, Habbari K, Fellah H, Bekhti K, Sebti F. Circulating species of leishmania at microclimate area of Boulemane Province, Morocco: impact of environmental and human factors. Parasit Vectors. 2017;10:100. doi: 10.1186/s13071-017-2032-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Zarrouk A, Kahime K, Boussaa S, Belqat B. Ecological and epidemiological status of species of the Phlebotomus perniciosus complex (Diptera: Psychodidae Phlebotominae) in Morocco. Parasitol Res. 2016 doi: 10.1007/s00436-015-4833-0. [DOI] [PubMed] [Google Scholar]
- 88.Talbi FZ, El Ouali LA, Fadil M, et al. Entomological investigations, seasonal fluctuations and impact of bioclimate factors of phlebotomines sand flies (Diptera: Psychodidae) of an emerging focus of cutaneous leishmaniasis in Aichoun Central Morocco. J Parasitol Res. 2020 doi: 10.1155/2020/6495108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.El Omari H, Chahlaoui A, Ouarrak K, Faraj C, El Ouali LA. Surveillance of leishmaniasis: inventory and seasonal fluctuation of phlebotomine sandflies (Diptera: Psychodidae), at the prefecture of Meknes (center of Morocco) Bull la Soc Pathol Exot. 2018 doi: 10.3166/bspe-2019-0061. [DOI] [PubMed] [Google Scholar]
- 90.Kholoud K, Denis S, Lahouari B, El Hidan MA, Souad B. Management of leishmaniases in the era of climate change in Morocco. Int J Environ Res Public Health. 2018 doi: 10.3390/ijerph15071542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Ait Kbaich M, Mhaidi I, Ezzahidi A, Dersi N, El Hamouchi A, Riyad M, Akarid K, Lemrani M. New epidemiological pattern of cutaneous leishmaniasis in two pre-Saharan arid provinces, southern Morocco. Acta Trop. 2017;173:11–16. doi: 10.1016/j.actatropica.2017.05.016. [DOI] [PubMed] [Google Scholar]
- 92.Gijón-Robles P, Abattouy N, Merino-Espinosa G, El Khalfaoui N, Morillas-Márquez F, Corpas-López V, Jaaouani N, Díaz-Sáez V, Riyad M, Martín-Sánchez J. Understanding the factors that determine the emergence of anthroponotic cutaneous leishmaniasis due to leishmania tropica in Morocco: density and mitochondrial lineage of Phlebotomus sergenti in endemic and free areas of leishmaniasis. Transbound Emerg Dis. 2021 doi: 10.1111/tbed.14179. [DOI] [PubMed] [Google Scholar]
- 93.Amro A, Al-Dwibe H, Gashout A, Moskalenko O, Galafin M, Hamarsheh O, Frohme M, Jaeschke A, Schönian G, Kuhls K. Spatiotemporal and molecular epidemiology of cutaneous leishmaniasis in Libya. PLoS Negl Trop Dis. 2017;11:e0005873. doi: 10.1371/journal.pntd.0005873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Talmoudi K, Bellali H, Ben-Alaya N, Saez M, Malouche D, Chahed MK. Modeling zoonotic cutaneous leishmaniasis incidence in central Tunisia from 2009–2015: forecasting models using climate variables as predictors. PLoS Negl Trop Dis. 2017 doi: 10.1371/journal.pntd.0005844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Oryan A, Akbari M. Worldwide risk factors in leishmaniasis. Asian Pac J Trop Med. 2016 doi: 10.1016/j.apjtm.2016.06.021. [DOI] [PubMed] [Google Scholar]
- 96.Votýpka J, Kasap OE, Volf P, Kodym P, Alten B. Risk factors for cutaneous leishmaniasis in Cukurova region Turkey. Trans R Soc Trop Med Hyg. 2012 doi: 10.1016/j.trstmh.2011.12.004. [DOI] [PubMed] [Google Scholar]
- 97.Reveiz L, Maia-Elkhoury ANS, Nicholls RS, Sierra Romero GA, Yadon ZE. Interventions for American cutaneous and mucocutaneous leishmaniasis: a systematic review update. PLoS ONE. 2013 doi: 10.1371/journal.pone.0061843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Zavitsanou A, Koutis C, Babatsikou F (2008) Leishmaniasis: an overlooked public health concern. Heal. Sci. J.
- 99.Abdelkrim O, Samia B, Said Z, Souad L. Modeling and mapping the habitat suitability and the potential distribution of arboviruses vectors in Morocco. Parasite. 2021;28:37. doi: 10.1051/parasite/2021030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Caini S, Kusznierz G, Garate VV, et al. The epidemiological signature of influenza B virus and its B/Victoria and B/Yamagata lineages in the 21st century. PLoS ONE. 2019;14:1–17. doi: 10.1371/journal.pone.0222381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Elsobky Y, El AG, Abdalla E, Byomi A, Reddy G. Possible ramifications of climate variability on HPAI-H5N1 outbreak occurrence: case study from the Menoufia Egypt. PLoS One. 2020 doi: 10.1371/journal.pone.0240442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Salaheldin AH, Kasbohm E, El-Naggar H, et al. Potential biological and climatic factors that influence the incidence and persistence of highly pathogenic H5N1 avian influenza virus in Egypt. Front Microbiol. 2018 doi: 10.3389/fmicb.2018.00528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Horton KC, Dueger EL, Kandeel A, et al (2017) Viral etiology, seasonality and severity of hospitalized patients with severe acute respiratory infections in the Eastern Mediterranean Region, 2007–2014. PLoS One 12:e0180954 [DOI] [PMC free article] [PubMed]
- 104.Kassem AZE. Does temperature affect COVID-19 transmission? Front Public Heal. 2020 doi: 10.3389/fpubh.2020.554964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Meo SA, Abukhalaf AA, Alomar AA, Al-Beeshi IZ, Alhowikan A, Shafi KM, Meo AS, Usmani AM, Akram J. Climate and COVID-19 pandemic: effect of heat and humidity on the incidence and mortality in world’s top ten hottest and top ten coldest countries. Eur Rev Med Pharmacol Sci. 2020 doi: 10.26355/eurrev_202008_22513. [DOI] [PubMed] [Google Scholar]
- 106.Černý J, Lynn G, Hrnková J, Golovchenko M, Rudenko N, Grubhoffer L. Management options for Ixodes ricinus-associated pathogens: a review of prevention strategies. Int J Environ Res Public Health. 2020 doi: 10.3390/ijerph17061830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Sprong H, Azagi T, Hoornstra D, Nijhof AM, Knorr S, Baarsma ME, Hovius JW. Control of Lyme borreliosis and other Ixodes ricinus-borne diseases. Parasit Vectors. 2018 doi: 10.1186/s13071-018-2744-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Parola P, Raoult D. Tick-borne bacterial diseases emerging in Europe. Clin Microbiol Infect. 2001 doi: 10.1046/j.1469-0691.2001.00200.x. [DOI] [PubMed] [Google Scholar]
- 109.Barzon L. Ongoing and emerging arbovirus threats in Europe. J Clin Virol. 2018 doi: 10.1016/j.jcv.2018.08.007. [DOI] [PubMed] [Google Scholar]
- 110.Alkishe AA, Peterson AT, Samy AM. Climate change influences on the potential geographic distribution of the disease vector tick Ixodes ricinus. PLoS ONE. 2017 doi: 10.1371/journal.pone.0189092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Essayagh M, El RA, Essayagh S, Essayagh T. Profil épidémiologique de la fièvre typhoïde, Meknès, 2013–2016. Rev Epidemiol Sante Publique. 2020 doi: 10.1016/j.respe.2019.09.006. [DOI] [PubMed] [Google Scholar]
- 112.Bauer M, Glenn T, Alda M, et al. Solar insolation in springtime influences age of onset of bipolar I disorder. Acta Psychiatr Scand. 2017;136:571–582. doi: 10.1111/acps.12772. [DOI] [PubMed] [Google Scholar]
- 113.Bauer M, Glenn T, Alda M, et al. Association between solar insolation and a history of suicide attempts in bipolar I disorder. J Psychiatr Res. 2019;113:1–9. doi: 10.1016/j.jpsychires.2019.03.001. [DOI] [PubMed] [Google Scholar]
- 114.Sekmoudi I, Khomsi K, Faieq S, Idrissi L. Assessment of global and regional PM10 CAMSRA data: comparison to observed data in Morocco. Environ Sci Pollut Res. 2021 doi: 10.1007/s11356-021-12783-3. [DOI] [PubMed] [Google Scholar]
- 115.Jonathan A, Josef T, Andrew S. Clearing the air: a review of the effects of particulate matter air pollution on human health. J Med Toxicol. 2012;8:166–175. doi: 10.1007/s13181-011-0203-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Han X, Naeher LP. A review of traffic-related air pollution exposure assessment studies in the developing world. Environ Int. 2006;32:106–120. doi: 10.1016/j.envint.2005.05.020. [DOI] [PubMed] [Google Scholar]
- 117.Horton G, Hanna L, Kelly B. Drought, drying and climate change: emerging health issues for ageing Australians in rural areas. Australas J Ageing. 2010 doi: 10.1111/j.1741-6612.2010.00424.x. [DOI] [PubMed] [Google Scholar]
- 118.Stanke C, Kerac M, Prudhomme C, Medlock J, Murray V. Health effects of drought: a systematic review of the evidence. PLoS Curr. 2013 doi: 10.1371/currents.dis.7a2cee9e980f91ad7697b570bcc4b004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Arlappa N, Rao KM, Venkaiah K, Brahmam GNV, Vijayaraghavan K. Nutritional parameters and chronic energy deficiency in older adults of desert areas of Western Rajasthan, India. J Nutr Elder. 2009;28:61–71. doi: 10.1080/01639360802633987. [DOI] [PubMed] [Google Scholar]
- 120.Effler P, Isaäcson M, Arntzen L, et al. Factors contributing to the emergence of Escherichia coli O157 in Africa. Emerg Infect Dis. 2001;7:812–819. doi: 10.3201/eid0705.017507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Pavelchak N, Church L, Roerig S, London M, Welles W, Casey G. Silo gas exposure in New York State following the dry growing season of 1995. Appl Occup Environ Hyg. 1999;14:34–38. doi: 10.1080/104732299303395. [DOI] [PubMed] [Google Scholar]
- 122.Mutuku FM, King CH, Bustinduy AL, Mungai PL, Muchiri EM, Kitron U. Impact of drought on the spatial pattern of transmission of Schistosoma haematobium in coastal Kenya. Am J Trop Med Hyg. 2011;85:1065–1070. doi: 10.4269/ajtmh.2011.11-0186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Carnie TL, Berry HL, Blinkhorn SA, Hart CR. In their own words: young people’s mental health in drought-affected rural and remote NSW. Aust J Rural Health. 2011;19:244–248. doi: 10.1111/j.1440-1584.2011.01224.x. [DOI] [PubMed] [Google Scholar]
- 124.Sousa AQ, Pearson R. Drought, smallpox, and emergence of leishmania braziliensis in northeastern Brazil. Emerg Infect Dis. 2009;15:916–921. doi: 10.3201/eid1506.071331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Brown L, Medlock J, Murray V. Impact of drought on vector-borne diseases - how does one manage the risk? Public Health. 2014;128:29–37. doi: 10.1016/j.puhe.2013.09.006. [DOI] [PubMed] [Google Scholar]
- 126.Zhang Y, Bi P, Hiller JE. Climate change and the transmission of vector-borne diseases: a review. Asia Pac J Public Health. 2008;20:64–76. doi: 10.1177/1010539507308385. [DOI] [PubMed] [Google Scholar]
- 127.Leal Filho W, Bönecke J, Spielmann H, Azeiteiro UM, Alves F, Lopes de Carvalho M, Nagy GJ. Climate change and health: an analysis of causal relations on the spread of vector-borne diseases in Brazil. J Clean Prod. 2018;177:589–596. doi: 10.1016/j.jclepro.2017.12.144. [DOI] [Google Scholar]
- 128.Camargo LB, Langoni H. Impact of leishmaniasis on public health. J Venom Anim Toxins Incl Trop Dis. 2006;12:527–548. doi: 10.1590/S1678-91992006000400002. [DOI] [Google Scholar]
- 129.Gubler DJ. The global emergence/resurgence of arboviral diseases as public health problems. Arch Med Res. 2002;33:330–342. doi: 10.1016/S0188-4409(02)00378-8. [DOI] [PubMed] [Google Scholar]
- 130.Sullivan KM. Health impact of influenza in the United States. Pharmacoeconomics. 1996;9:26–33. doi: 10.2165/00019053-199600093-00008. [DOI] [PubMed] [Google Scholar]
- 131.Shigemura J, Kurosawa M. Mental health impact of the COVID-19 pandemic in Japan. Psychol Trauma Theory, Res Pract Policy. 2020;12:478–479. doi: 10.1037/tra0000803. [DOI] [PubMed] [Google Scholar]
- 132.Khomsi K, Chelhaoui Y, Alilou S, Souri R, Najmi H (2020) Concurrent heatwaves and extreme Ozone (O3) episodes : combined atmospheric patterns and impact on human health. 1–16 [DOI] [PMC free article] [PubMed]