Abstract
Placenta accreta spectrum (PAS) is defined as abnormal placental adherence or invasion of the myometrium or extrauterine organs. This case series will analyze MRI findings and PAS grading, in addition to emergency situations like massive hematuria and placental invasion with rupture. We report 5 cases of pregnant women with placenta previa with suspected PAS. MRI revealed 1 case of placenta accreta, one case of placenta increta, and 3 cases of placenta percreta. Two cases were emergency situations. All cases were managed with cesarean section. PAS is frequently related to severe obstetric hemorrhage associated with high maternal morbidity and mortality, making diagnosis and management challenging. Ultrasound is the initial diagnostic modality for PAS. Although ultrasound is preferred for PAS diagnosis, MRI provides an effective modality for the analysis of the depth of placental invasion and can be helpful in emergency situations.
Keywords: Diagnostic, Emergency, MRI, Placenta accreta spectrum
Introduction
Placenta accreta spectrum (PAS) is an abnormally invasive placenta which does not detach spontaneously after delivery [1]. The frequency of PAS is rising worldwide [2,3], including in Indonesia. There has been a resurgence in cases since 2016, with the incidence reaching 2% and continuously increasing [4]. PAS is significantly associated with massive hemorrhage requiring multiple blood transfusions and increased maternal morbidity and mortality [5]. PAS can be classified based on the FIGO (International Federation of Gynecology and Obstetrics) classification, which focuses on the depth of placental invasion into the uterine wall during clinical and histopathological examination [6]. Ultrasound is a cheap and effective modality for PAS diagnosis, but MRI has emerged as the imaging modality of choice for PAS diagnosis in cases where the ultrasound is equivocal [7]. This case reports will analyze the MRI, clinical, and histopathological correlation in several PAS classification and emergency situations.
Case reports
Patient 1
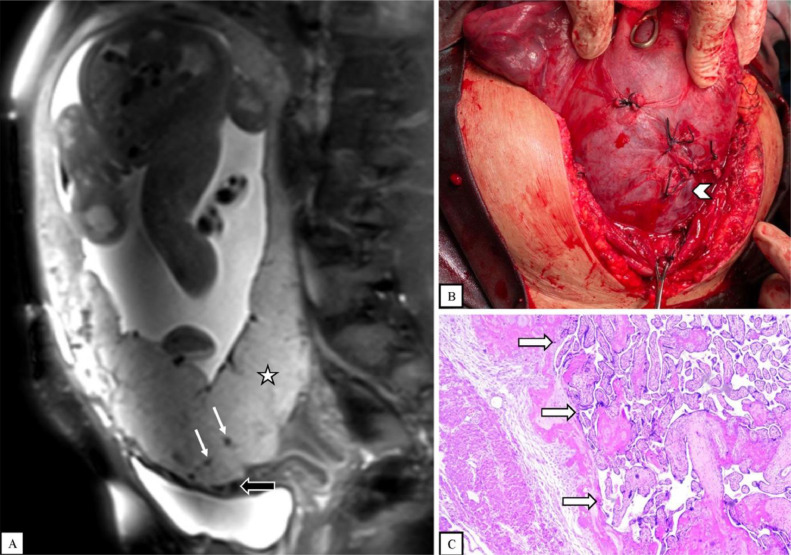
A 42-year-old woman at 30 weeks gestation of her third pregnancy was referred with placenta previa, 2 previous cesarean sections, and suspected of PAS. She complained of vaginal spotting without abdominal pain. Unenhanced pelvic MRI was performed and the result was placenta accreta with complete placenta previa. MR images showed an abnormal, heterogenous placenta with a rounded edge and dark intraplacental bands. There was mildly increased vascularity at the placental-myometrial interface (Fig. 1A). An elective cesarean delivery was planned at 34 weeks gestation followed by a hysterectomy. There was minimal neovascularity and no placental tissue was seen invading the surface of the uterus; FIGO grade 1 (placenta accreta) was confirmed (Fig. 1B). Histopathological examination confirmed the diagnosis of placenta accreta and showed placental villi attached to the endometrium (Fig. 1C). A healthy baby was delivered with a weight of 2300 g.
Fig. 1.
(A) Sagittal T2-weighted fat-saturated MR image shows an abnormal, heterogenous placenta with a rounded edge (star) and dark intraplacental bands (thin white arrows). There is mildly increased vascularity at the placental-myometrial interface (thick black arrow). (B) Cesarean section with hysterectomy shows minimal neovascularity and no placental tissue is seen invading the surface of the uterus (arrowhead). (C) Histopathological examination shows placental villi attached to the endometrium (thick white arrows).
Patient 2
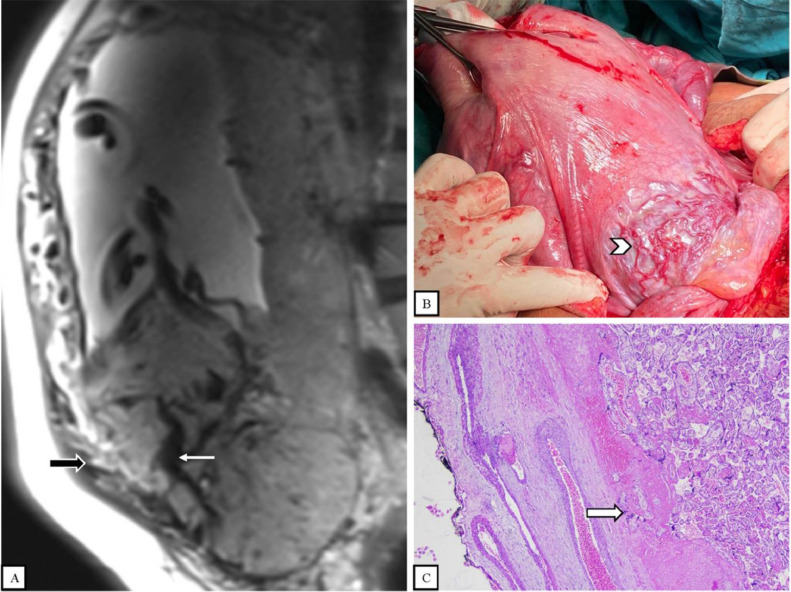
A 41-year-old woman at 34 weeks gestation of her fourth pregnancy was referred with placenta previa, 2 previous cesarean sections, and suspected of placenta percreta based on a previous ultrasound. She complained of spotting and mild abdominal pain. An unenhanced pelvic MRI revealed placenta increta. MR images showed an anteroinferior placental bulge into the myometrium. There were many dark intraplacental bands (Fig. 2A). Two days later, a cesarean section with hysterectomy was performed and confirmed the diagnosis of placenta increta. There was a placental bulge with purple/blue coloring and multiple hypervascularity (Fig. 2B). No placental tissue was seen invading the surface of the uterus. Histopathological examination showed placental villi invading the myometrium. FIGO grade 2 (placenta increta) was confirmed (Fig. 2C). A healthy baby was born with a weight of 2600 g.
Fig. 2.
(A) Sagittal T2-weighted MR image shows an anteroinferior placental bulge into the myometrium (thick black arrow) and dark intraplacental bands (thin white arrow). (B) Cesarean section with hysterectomy shows a placental bulge with purple/blue coloring and multiple hypervascularity (arrowhead). No placental tissue is seen invading the surface of the uterus. (C) Histopathological examination shows placental villi invading the myometrium (thick white arrow).
Patient 3
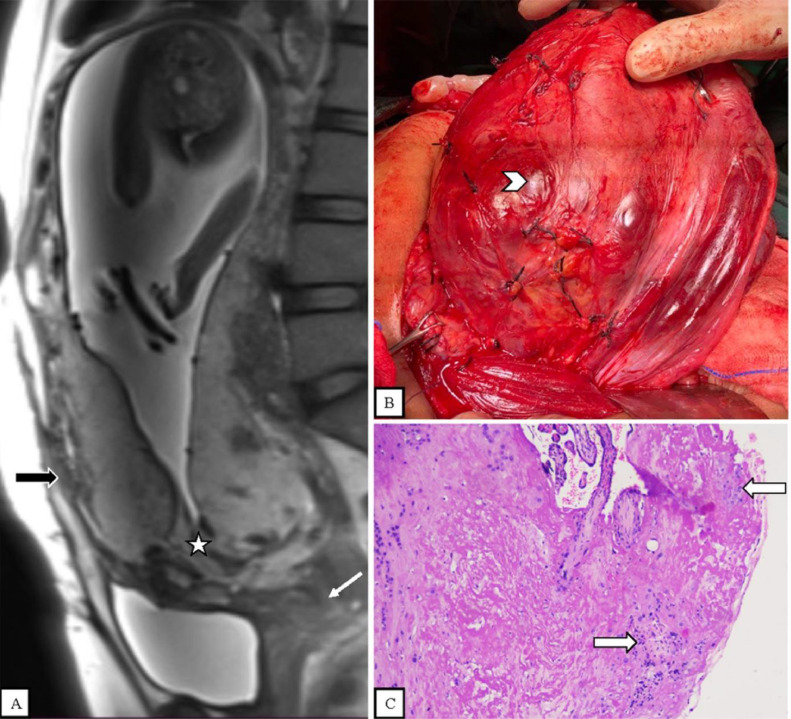
A 43-year-old woman at 24 weeks gestation of her fifth pregnancy was referred with placenta previa and suspected placenta percreta based on her ultrasound examination at 21 weeks gestation. She had 2 previous cesarean sections and complained of spotting without abdominal pain. MR images showed a placental bulge into the uterine serosa, focal disruption of the myometrium, and loss of the retroplacental dark line. Shortening of the cervix, an abnormal, heterogenous placenta with lumpy contour, a rounded edge, and dark intraplacental bands were also seen (Fig. 3A). Cesarean section with hysterectomy was performed at 34 weeks due to major vaginal bleeding. Placental tissue was seen invading the surface of the uterus during surgery (Fig. 3B). Histopathological examination showed placental villi invading the uterine serosa. PAS FIGO grade 3A (placenta percreta) was confirmed (Fig. 3C). A healthy baby was born with a weight of 2000 g.
Fig. 3.
(A) Sagittal T2-weighted MR image shows the placental bulge into the uterine serosa, focal disruption of the myometrium, and loss of the retroplacental dark line (thick black arrow). There is also shortening of the cervix (thin white arrow). An abnormal, heterogenous placenta with lumpy contour, a rounded edge, and dark intraplacental bands are also seen (star). (B) Cesarean section with hysterectomy shows placental tissue invading the surface of the uterus (arrowhead). (C) Histopathological examination shows placental villi invading the uterine serosa (thick white arrows).
Patient 4
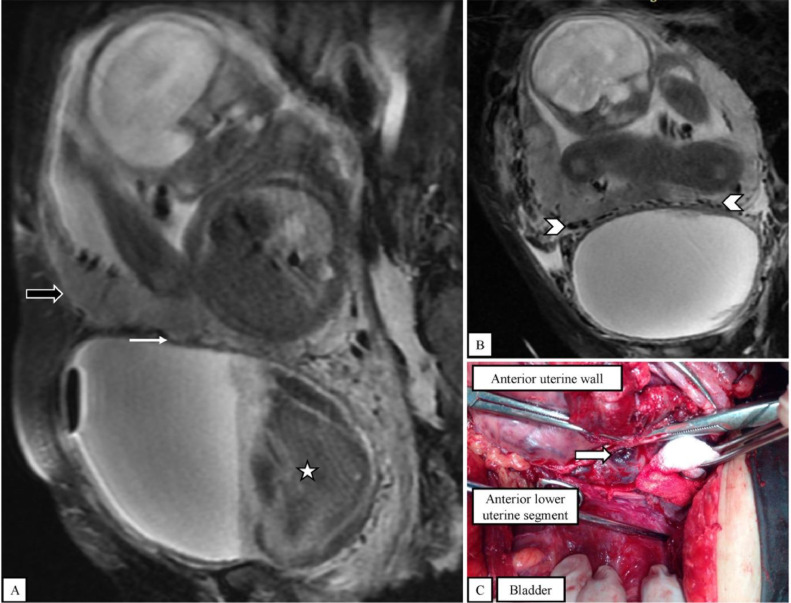
A 41-year-old woman at 28 weeks gestation was referred to our hospital with suspected placenta percreta based on her ultrasound examination at 26 weeks gestation. This was her fourth pregnancy, with one cesarean section performed 26 years ago. She complained of vaginal bleeding and hematuria. An unenhanced pelvic MRI was performed and showed a placental bulge with loss of the retroplacental dark line. There was a massive blood clot inside the bladder, but the vesicouterine fat plane was still effective, with no bladder infiltration (Fig. 4A). Increased vascularity between the bladder and the uterus was observed (Fig. 4B). After the MRI examination, an emergency cesarean section with hysterectomy confirmed the diagnosis of PAS FIGO grade 3A (placenta percreta). Placental tissue was seen invading the surface of the uterus, without bladder infiltration (Fig. 4C). The baby was born with a weight of 1000 g.
Fig. 4.
(A) Sagittal T2-weighted fat-saturated MR image shows the placental bulge with loss of the retroplacental dark line (thick black arrow). There is a massive blood clot inside the bladder (star). The vesicouterine fat plane is still effective (thin white arrow). (B) Coronal T2-weighted fat-saturated MR image shows increased vascularity between the bladder and the uterus (arrowheads). (C) Cesarean section with hysterectomy shows placental tissue invading the surface of the uterus, without bladder infiltration (thick white arrow).
Patient 5
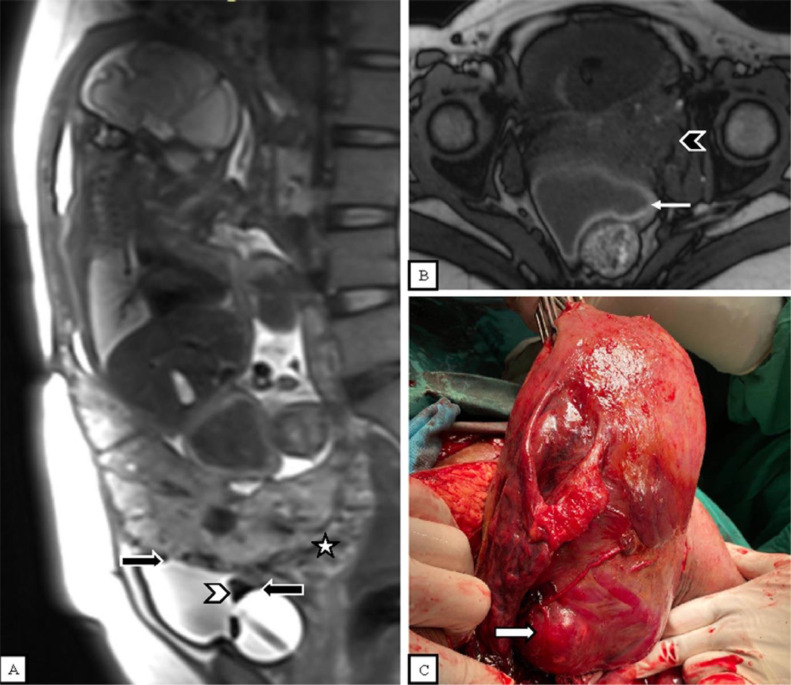
A 32-year-old woman at 28 weeks gestation was referred with placenta previa and placenta percreta was suspected based on her ultrasound examination at 27 weeks gestation. This was her second pregnancy, with one previous cesarean section due to placenta previa. She complained of hematuria and spotting without abdominal pain. The unenhanced pelvic MRI showed placental invasion of the S2 sector and bladder, with a blood clot inside the bladder (Fig. 5A). There was also placental invasion with rupture and invasion to the lower-left parametrium (Fig. 5B). Three days later, a cesarean section with hysterectomy was performed due to abdominal pain with high suspicion of placental rupture and the diagnosis of PAS FIGO grade 3C (placenta percreta in lower parametrium and bladder) was confirmed. A healthy baby was born with a weight of 1100 g.
Fig. 5.
(A) Sagittal T2-weighted MR image shows placental invasion to the bladder (thick black arrows). There is placental invasion of the S2 uterine sector (star) and a blood clot is seen inside the bladder (white arrowhead). (B) Axial T1-weighted MR image shows placental invasion with rupture (thin white arrow) and invasion to the lower-left parametrium (black arrowhead). (C) Cesarean section with hysterectomy shows placental tissue invading the surface of the bladder and left parametrium (thick white arrow).
Discussion
Dr. Soetomo General Academic Hospital is a tertiary center hospital and the main referral hospital for PAS. Most of these cases had advanced maternal age (4/5) and multiple cesarean sections (3/5). Two of these cases had a single cesarean section with an emergency due to massive hematuria and PAS with lower parametrium rupture, suggesting that the adverse outcomes of PAS are not always caused by multiple cesarean sections. The main etiological factor of PAS is a previous cesarean section, which has poor healing due to various causes [8].
Ultrasound is the first-line imaging modality for PAS because it is effective, inexpensive, widely available, and less time is required for exploration, with an overall sensitivity of 100% and specificity of 100% for multiple ultrasound signs; however, this modality has lower specificity for the analysis of the depth of placental invasion (specificity 64.8%-68.6%, sensitivity 100%) [9].
MRI is used for PAS evaluation and diagnosis when ultrasound examination is inconclusive, in cases of placental invasion of the posterior and lateral walls—which is difficult to assess in obese patients—and possible invasion of the parametrium and other adjacent organs (bladder, cervix, and pelvic side walls) [7,10]. MR imaging findings that suggest PAS are: placental bulge, a heterogenous placental signal intensity with lumpy contour and a rounded edge, dark T2 intraplacental bands, abnormal placental vascularity, loss of the retroplacental T2 dark zone, myometrial thinning, focal interruptions of the myometrium (highly specific for increta and percreta), and bladder tenting (highly specific for percreta) [7,11]. MRI has a high sensitivity and specificity for PAS diagnosis (sensitivity 100%, specificity 100%) and better accuracy for evaluating the depth of placental invasion when compared with ultrasound (specificity 96.8%-98.8%, specificity 86.5%-100%) [12].
Hematuria is one of the maternal signs of bladder invasion in PAS [10], although in one of our cases the MRI did not show bladder invasion and it was later confirmed during surgery. This case demonstrates the importance of MRI in differentiating between bladder invasion and rupture of the vesical vessel.
One case with lower parametrium showed internal bleeding due to placental invasion and rupture. In an emergency case such as this, MRI can be performed as long as the hemodynamics are still under control.
Sagittal MRI divides the anterior placental invasion using a plane perpendicular to the center of the upper bladder axis into 2 sectors: the S1 uterine sector is located in the uterine body, and the S2 uterine sector is located in the lower uterine segment or below it (cervix and upper vagina). Sector S1 is vascularized by vessels that are easy to access and exhibit rapid hemostasis. Sector S2 is vascularized by deep vessels, which significantly increases surgical complexity and presents an increased risk of hemorrhage [13]. S1/S2 uterine sector classification can provide important information for surgical strategies, especially the use of aortic control during surgery [14,15].
Conclusion
These case reports show the utility of MRI, especially for advanced grading of PAS, to give important information for surgical strategies. MRI can help to distinguish the depth of placental invasion alongside PAS diagnosis using ultrasound, especially for advanced grading and even in emergency situations as long as maternal hemodynamics are stable and there is no evidence of fetal distress.
Consent and ethic committee approval
Written consent has been obtained from the patient as there is no patient identifiable data included in this case reports. This study has met the ethical principle and already got approval from Research Ethics Committee from Dr. Soetomo General Hospital, Surabaya.
Patient consent
Informed consent obtained for publication of case reports: Written informed consent was obtained from the patient for the publication of this case reports.
Footnotes
Competing Interests: The author has nothing to disclose
References
- 1.Morlando M., Collins S. Placenta accreta spectrum disorders: challenges, risks, and management strategies. Int J Womens Health. 2020;12:1033–1045. doi: 10.2147/IJWH.S224191. Nov 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wu S., Kocherginsky M., Hibbard J.U. Abnormal placentation: twenty-year analysis. Am J Obstet Gynecol. 2005;192(5):1458–1461. doi: 10.1016/j.ajog.2004.12.074. May. [DOI] [PubMed] [Google Scholar]
- 3.Grechukhina O., Deshmukh U., Fan L., Kohari K., Abdel-Razeq S., Bahtiyar M.O., et al. Cesarean scar pregnancy, incidence, and recurrence: five-year experience at a single tertiary care referral center. Obstet Gynecol. 2018;132(5) doi: 10.1097/AOG.0000000000002940. [DOI] [PubMed] [Google Scholar]
- 4.Aryananda R.A., Akbar A., Wardhana M.P., Gumilar K.E., Wicaksono B., Ernawati E., et al. New three-dimensional/four-dimensional volume rendering imaging software for detecting the abnormally invasive placenta. J Clin Ultrasound. 2019;47(1) doi: 10.1002/jcu.22641. [DOI] [PubMed] [Google Scholar]
- 5.Nieto-Calvache A.J., Palacios-Jaraquemada J.M., Osanan G., Cortes-Charry R., Aryananda R.A., Bangal V.B., et al. Lack of experience is a main cause of maternal death in placenta accreta spectrum patients. Acta Obstet Gynecol Scand. 2021;100(8):1445–1453. doi: 10.1111/aogs.14163. Aug 1. [DOI] [PubMed] [Google Scholar]
- 6.Jauniaux E., Bhide A., Kennedy A., Woodward P., Hubinont C., Collins S., et al. FIGO consensus guidelines on placenta accreta spectrum disorders: Prenatal diagnosis and screening. Int J Gynecol Obstet. 2018;140(3):274–280. doi: 10.1002/ijgo.12408. Mar 1. [DOI] [PubMed] [Google Scholar]
- 7.Srisajjakul S., Prapaisilp P., Bangchokdee S. Magnetic resonance imaging of placenta accreta spectrum: a step-by-step approach. Korean J Radiol. 2021;22(2):198–212. doi: 10.3348/kjr.2020.0580. Feb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Vervoort A.J.M.W., Uittenbogaard L.B., Hehenkamp W.J.K., Brölmann H.A.M., Mol B.W.J., Huirne J.A.F. Why do niches develop in Caesarean uterine scars? Hypotheses on the aetiology of niche development. Hum Reprod. 2015;30(12):2695–2702. doi: 10.1093/humrep/dev240. Dec 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cali G., Forlani F., Timor-Trisch I., Palacios-Jaraquemada J., Foti F., Minneci G., et al. Diagnostic accuracy of ultrasound in detecting the depth of invasion in women at risk of abnormally invasive placenta: A prospective longitudinal study. Acta Obstet Gynecol Scand. 2018;97(10):1219–1227. doi: 10.1111/aogs.13389. Oct. [DOI] [PubMed] [Google Scholar]
- 10.Balan R., Radu ViD, Giușcă Si-E, Costache C., Ristescu C., Puia D., et al. A rare cause of massive hematuria: placenta percreta with bladder invasion. In Vivo (Brooklyn) 2021;35(6) doi: 10.21873/invivo.12670. Nov 13633 LP –3639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kilcoyne A., Shenoy-Bhangle A.S., Roberts D.J., Sisodia R.C., Gervais D.A., Lee S.I. MRI of placenta accreta, placenta increta, and placenta percreta: pearls and pitfalls. Am J Roentgenol. 2016;208(1):214–221. doi: 10.2214/AJR.16.16281. Oct 20. [DOI] [PubMed] [Google Scholar]
- 12.Familiari A., Liberati M., Lim P., Pagani G., Cali G., Buca D., et al. Diagnostic accuracy of magnetic resonance imaging in detecting the severity of abnormal invasive placenta: a systematic review and meta-analysis. Acta Obstet Gynecol Scand. 2018;97(5):507–520. doi: 10.1111/aogs.13258. May 1. [DOI] [PubMed] [Google Scholar]
- 13.Palacios Jaraquemada J.M., Bruno C.H. Magnetic resonance imaging in 300 cases of placenta accreta: surgical correlation of new findings. Acta Obstet Gynecol Scand. 2005;84(8):716–724. doi: 10.1111/j.0001-6349.2005.00832.x. Aug. [DOI] [PubMed] [Google Scholar]
- 14.Nieto-Calvache A.J., Palacios-Jaraquemada J.M., Aryananda R.A. Factors to consider when seeking better results in placenta accreta spectrum. Acta Obstet Gynecol Scand. 2021;100(10):1932–1933. doi: 10.1111/aogs.14214. Oct 1. [DOI] [PubMed] [Google Scholar]
- 15.Nieto-Calvache A.J., Aryananda R.A. The first step to evaluate the usefulness of arterial occlusion in placenta accreta spectrum is to establish its indications. Acta Obstet Gynecol Scand. 2021 doi: 10.1111/aogs.14307. Dec 14. [DOI] [PMC free article] [PubMed] [Google Scholar]