Abstract
Rectal cancer is a relatively common malignancy in the United States. Magnetic resonance imaging (MRI) of rectal cancer has evolved tremendously in recent years, and has become a key component of baseline staging and treatment planning. In addition to assessing the primary tumor and locoregional lymph nodes, rectal MRI can be used to help with risk stratification by identifying high risk features such as extramural vascular invasion and can assess treatment response for patients receiving neoadjuvant therapy. As the practice of rectal MRI continues to expand further into academic centers and private practices, standard MRI protocols and reporting are critical. In addition, it is imperative that the radiologists reading these cases work closely with surgeons, medical oncologists, radiation oncologists and pathologists to ensure we are providing the best possible care to patients. This review aims to provide a broad overview of the role of MRI for rectal cancer.
Keywords: Rectal cancer, MRI, Radiology
Introduction
Rectal cancer is a relatively common malignancy in the United States. Magnetic resonance imaging (MRI) of rectal cancer has evolved tremendously in recent years, and has become a key component of baseline staging and treatment planning. In addition to assessing the primary tumor and locoregional lymph nodes, rectal MRI can be used to help with risk stratification by identifying high risk features such as extramural vascular invasion and can assess treatment response for patients receiving neoadjuvant therapy. As the practice of rectal MRI continues to expand, standard MRI protocols and reporting are critical. In addition, it is imperative that the radiologists reading these cases work closely with surgeons, medical oncologists, radiation oncologists and pathologists to ensure we are providing the best possible care to patients. This review aims to provide a broad overview of the role of MRI for rectal cancer.
Staging
Local staging of rectal cancer has evolved considerably in recent decades, and now relies heavily on MRI of the rectum. Staging of rectal cancer by high-resolution MRI is considered reliable, with one meta-analysis finding diagnostic accuracy, sensitivity and specificity of 85%, 87% and 75% respectively (1).
Traditionally, the rectum has been divided into thirds for purposes of tumor localization. A tumor is described as low (0–5 cm from anal verge), middle (between 5–10 cm from the anal verge) or high (between 10–15 cm from anal verge). Above 15 cm, tumors are staged and treated as colon cancer.
The T category is defined by the depth of tumor penetration into and beyond the rectal wall. T1 tumors are confined to the mucosa and submucosa, whereas T2 tumors extend into to the muscularis propria. The distinction between T1 and T2 tumors is not always reliable on MRI, except in some cases when it is possible to visualize a well-defined submucosal layer that will appear hyperintense (bright) on T2-weighted images beneath the tumor (2, 3). Endorectal ultrasound, however, was found to have a better diagnostic performance in distinguishing T1 and T2 stages compared to MRI rectum (4).
Although the distinction of T1 versus T2 tumors is somewhat less important, identifying a tumor as T3 is imperative, as it has implications for treatment and often warrants neoadjuvant therapy prior to definitive surgical treatment. Tumors are categorized as T3 when there is disruption of the muscularis propria and extension of the tumor beyond the rectal wall into the mesorectal fat. T3 lesions are further sub-divided into four categories, based on the distance between the muscularis propria and maximum extramural invasion: T3a < 1mm, T3b is 1–5mm, T3c is 5–15mm and T3d >15mm. On T2-weighted images, the muscularis propria can be visualized as a low signal intensity line, which stands in contrast to the intermediate signal of the submucosa and surrounding high signal intensity mesorectal fat. The muscularis propria has two layers, the inner circular layer and outer longitudinal layer that can be disrupted by penetrating vessels. It is crucial for the radiologist to differentiate between those vessels and T3 tumors, which appear as nodular bulging lesions in continuity with the intramural component of the tumor (5). Another pitfall in T-staging is the desmoplastic reaction that can occur in the mesorectal fat adjacent to the rectal tumor, which appears as low intensity (dark) fine linear spicules on T2-weighted images, which can be mistaken as stage T3 disease (5). High resolution MRI rectum with reduced slice thickness can be useful in the differentiating desmoplastic reaction and stage T3 tumor. The sub-categories of T3 tumors do carry some prognostic significance, as the MERCURY trial, a multicenter rectal MRI study, showed that patients with stages T3c and T3d have a worse prognosis than patients with stages T3a and T3b (6). [FIGURE 1]
Figure 1:
47 year-old male with newly diagnosed rectal adenocarcinoma for baseline staging. (a) Sagittal T2 weighted image of the mid to low rectal tumor (arrowheads), with an enlarged suspicious superior rectal lymph node (dashed arrow). (b) Axial oblique T2 weighted image through the lower part of the tumor shows extramural extension up to 0.8 cm (dashed line), conferring T3 disease. Enlarged, rounded heterogeneous lymph nodes in the mesorectum (c, arrowheads) and superior rectal chain (d, arrowheads) are consistent with metastatic total mesocolic excision (TME) nodes.
T4 tumors are those that invade the anterior peritoneal reflection (stage T4a) or other pelvic organs or structures, including skeletal muscle (T4b). The anterior peritoneal reflection is visualized as a thin T2 hypointense line of 1mm or less in thickness that attaches on the anterior aspect of the rectum. The most inferior portion of the peritoneal reflection extends to the tip of the seminal vesicles in men and to the uterocervical junction in women. The anterior peritoneal reflection is reliably seen in most cases, and is better seen when there is trace amount of fluid in the pelvis (7). For mid and upper cancers, tumors can invade the bladder and uterus anteriorly, the pelvic sidewall laterally and the sacrum posteriorly. Low rectal tumors can invade the pelvic floor, anal sphincter, prostate, seminal vesicles, vagina, sacrum and coccyx (5).
Because of the proximity of low rectal tumors to the anal sphincter complex, it is imperative that radiologists indicate whether the tumor involves the internal sphincter, intersphincteric space, external sphincter and levator ani (8). The T2 weighted coronal oblique plane is the best for the evaluation of sphincter complex invasion (9).
MRI of the circumferential resection margin
The circumferential resection margin (CRM), also termed the lateral or radial resection margin, is defined as the surgical plane of resection during a total mesenteric excision (TME), the standard transabdominal surgical approach for the curative resection of rectal cancer (10). Whether a low anterior resection (LAR), intersphincteric resection (also known as the “ultra-LAR”), or abdominoperineal resection (11) are utilized, a TME involves the complete resection of the mesorectum along the plane of the mesorectal fascia. An intersphincteric resection may be indicated when a lower rectal tumor is confined to the inner anal sphincter, whereas an APR includes resection of the entire sphincter complex and variable portions of the levator ani and adjacent perineal soft tissues.
Surgical techniques which incorporate TME have significantly improved the outcome of rectal cancer patients, however, the single most important predictor of local recurrence and disease-free survival remains whether the CRM is involved by tumor (positive CRM) on surgical histopathology, correlating with the likelihood of residual disease beyond the resection margin (12–18). The measured distance of a tumor from the CRM is also an independent prognostic risk factor (19, 20). Locally advanced rectal cancers (usually T3c or greater) including tumors with predicted positive CRMs at imaging will usually benefit from neoadjuvant chemoradiotherapy (CRT) prior to reassessment for surgical resection, and this is generally determined by MRI of the rectum performed for baseline staging. [FIGURE 2]
Figure 2:
60 year-old male with newly diagnosed rectal adenocarcinoma for baseline staging. (a) Axial oblique T2-weighted image through the mid rectum shows a circumferential tumor (dashed arrow), with the mesorectal fascia noted (arrowheads). (b) More inferiorly in the rectum, there is extramural extension of tumor with multifocal involvement of the mesorectal fascia (arrowheads).
The anatomic correlate for the CRM is the mesorectal fascia (MRF), a term which is progressively being favored on MRI reports over CRM, which largely remains a surgical and pathologic descriptor. On MRI, the MRF is a thin T2 hypointense line surrounding the mesorectum. The distance from the MRF is measured as the shortest distance between the most peripheral margin of extramural rectal tumor and the MRF, and should be routinely included on all reports for relevant T3 tumors in the upper and mid rectum (21). MRI is the most accurate and reliable preoperative imaging modality for the measurement of this distance (1, 22). This distance is analogous and inversely related to the stratification of T3 tumors (T3a-T3d), with similar trends toward likelihoods of recurrence and disease-free survival. The most important distinction to make with this measurement is whether the tumor is within 1 mm of the MRF, which would predict a “positive CRM,” or between 1–2 mm from the MRF which is termed a “threatened” CRM (6, 23–26). It is also recommended to measure and document the shortest distance of any suspect lymphadenopathy and separate tumor deposits from the MRF although the prognosis of patients with a positive CRM solely related to these findings are not as poor (27, 28). The use of endorectal gel remains a topic of debate with at least one study purporting it may alter the measured distance of tumor from the MRF (29).
It is important to note that the measured distance to the MRF only applies to surgical resection planes and not to the peritonealized surfaces of the mid to upper rectum. At these levels, the MRF is no longer circumferential and only located along the posterior and posterolateral rectum. Tumor involvement of the anterior peritoneal reflection would instead confer a stage of T4a, and usually manifests as focal thickening or irregular nodularity of the peritoneal lining laterally rather than merely contact upon the peritoneal reflection.
Similarly, MRF distance measurements do not typically apply to lower rectal tumors which are located near the levator ani. Here the mesorectum narrows significantly as it extends inferiorly towards the anal sphincter complex. Given the proximity of the levator ani and sphincter complex to the rectal wall, the risk of MRF involvement by lower rectal tumors is much higher with surgical margins positive in up to 30% of cases (30). In these cases, preoperative chemoradiation therapy (CRT) can significantly downstage tumors in at least 74% of patients with complete pathologic response in 10% allowing for potential sphincter preservation (31). For lower rectal tumors, it is more important to measure the distance of the inferior extent of the tumor from the upper margin of the anal sphincter (important for planning in LAR) and to describe whether the intersphincteric space or external anal sphincter are involved to help the surgeon decide between an intersphincteric low anterior resection versus abdominoperineal resection.
Preoperative CRT has been shown to significantly improve survival by downstaging tumors and increasing the likelihood of CRM-negative surgical resection (32–34). Following neoadjuvant CRT, MRI restaging for T, N, and M categories is less accurate than on pre-treatment MRI, largely due to overestimation of disease related to inflammation and fibrosis (22, 35, 36). Accuracy, however, can be much improved when diffusion-weighted imaging is utilized. For further details regarding MR evaluation of post-treatment response, see the separate section below.
EMVI
Extramural venous invasion (EMVI) is diagnosed on pathology when malignant cells are seen within a vein, which extends beyond the muscularis propria of the rectal wall, and has been shown to be a poor prognostic indicator in rectal cancer patients. Studies have demonstrated an association between pathologically detected EMVI and local tumor recurrence, lymph node metastases, distant metastases, and overall survival (37–39). Brown et. al. were the first to describe EMVI on MRI (mrEMVI) and found it to correlate well with EMVI identified at histopathology (40). Since that early description, numerous studies have demonstrated the prognostic importance of mrEMVI to be similar to that of pathologically detected EMVI (41–45).
On MR imaging, EMVI results in the replacement of the normal dark vascular flow void on T2 weighted imaging by intermediate signal intensity similar to the primary rectal tumor. More advanced tumor invasion may result in vessel expansion and/or vessel irregularity or nodularity. EMVI of a primarily mucinous tumor will be markedly T2 hyperintense, similar to the primary tumor. A 5-point rating system was introduced by Smith et al (45) to determine the likelihood of the presence of EMVI. The system uses the tumor growth pattern, the location of the primary tumor relative to visible penetrating vessels, the caliber and contour of the vessel, and the intraluminal signal intensity to assign a likelihood score. Although such scoring systems have been used in various investigations, in our experience they are not routinely used in clinical practice. Although not an essential component of the MRI examination, in at least one study, use of intravenous contrast material increased radiologist confidence for detecting EMVI and increased sensitivity on post treatment exams, although the differences in the latter were not statistically significant (46). On post contrast images, EMVI can be identified either as a hypoenhancing intraluminal filling defect or as nodular/irregular enhancing tumor expanding the vessel. [FIGURE 3]
Figure 3:
54 year-old male with rectal adenocarcinoma. (a) Axial oblique T2-weighted image through the rectum shows intermediate signal tumor expanding a mesorectal vein (arrowhead). The presence of tumor within the vein is better appreciated on coronal oblique T2-weighted images (b, c; arrowheads). This extramural vascular invasion (EMVI) extended to the right internal iliac vein (not shown).
When EMVI is contiguous with the primary tumor, by definition it is associated with T3 or more advanced tumor and its extent should contribute to the determination of the extramural depth of invasion and to the T-stage. When present, the location should be documented, as well as its relationship to the mesorectal fascia (MRF). If EMVI that is contiguous with the primary tumor lies within 1 mm of the MRF at MRI, the anticipated operative margin should be considered invaded, conferring an increased risk for post-operative local recurrence. However, EMVI can be discontiguous and remote from the primary tumor as well. Although proximity of discontiguous EMVI to the MRF may not have the same prognostic significance regarding local recurrence, it should still be commented upon in the radiology report, as appropriate planning may help decrease the likelihood of tumor spillage at this location during surgery (27). Multiplanar evaluation is essential at MRI, as EMVI can be mistaken for a focal mesorectal metastatic lymph node or tumor deposit in a single plane of imaging, and this multiplanar evaluation is an advantage that MRI has over single planar blocked histopathology specimen evaluation.
Brown et al reported 85% agreement between MRI and pathologic detection of EMVI in vessels greater than 3 mm in diameter, however MRI is limited in detection of EMVI in small veins (40). On one of the earlier studies, MRI had a sensitivity of 62% and specificity of 88% when using histopathology as the reference standard (45). Several subsequent studies demonstrated a wide range of sensitivities (28–100%) with generally good specificity (88–94%)(39). A recent meta-analysis of 14 studies demonstrated a pooled sensitivity of 61% and specificity of 87% for MR detection of EMVI in colorectal cancer(47).
Although EMVI is not included in the TNM staging, its presence or absence should be included in the synoptic radiology report to aid in treatment planning and risk stratification. In the pretreatment setting, the presence of EMVI supports the need for neoadjuvant chemoradiotherapy. The importance of identifying persistent mrEMVI at restaging following chemoradiotherapy is not entirely understood, however, it has been reported that patients with mrEMVI positive tumors at baseline that regress to become mrEMVI negative at restaging have similar survival outcomes and recurrence rates as patients who were mrEMVI negative at baseline, and that patients who remained mrEMVI positive at restaging have shorter disease-free survival (48). Thus, post chemoradiotherapy, the presence of mrEMVI may contribute to consideration for intensified treatment and/or more aggressive surveillance strategies.
Lymph nodes
When evaluating the pelvic lymph nodes, it is important to consider the vascular supply to the rectum. The rectum is supplied by superior, middle, and inferior rectal (or hemorrhoidal) arteries. The superior rectal artery is a continuation of the inferior mesenteric artery, whereas the middle and inferior arteries are branches of the internal iliac arteries. The middle rectal artery is often absent or diminutive (49). Venous drainage follows the arterial system; the superior rectal vein cephalically continues as the inferior mesenteric vein and caudally communicates with the hemorrhoidal plexus with the middle and inferior rectal veins. The middle and inferior rectal veins drain to the internal iliac veins (49).
The lymphatic drainage of the rectum follows rectal vasculature and can also be subdivided into the drainage of the upper versus the lower rectum with three primary pathways via the superior, lateral, and inferior tracts (50–52).
The superior pathway follows the superior rectal arteries and drains the middle and upper rectum and eventually drains to the inferior mesenteric nodal group (53). The lateral pathway follows the path of the middle or inferior rectal arteries and eventually terminates at the subaortic nodes (52). The inferior pathway mostly drains the anal canal but may occasionally drain the lower rectum. This pathway ultimately drains to the superficial inguinal nodes.
The pelvic lymph nodes can be broadly classified into two groups: locoregional (N+) nodes and non-locoregional (M1) nodes (53). Though this classification is based on their relationship to the expected vascular supply to the rectum, in practical terms, it is more so based on the expected surgical resection margins for a routine TME. In general, mesorectal/superior rectal nodes (“central nodes”) are within the expected resection specimen of a TME, and non-locoregional (“pelvic side wall”) nodes are not.
Involvement of the locoregional nodes (N+) is an indication for neoadjuvant chemoradiation therapy. The presence of non-locoregional nodes (M1 disease) is potentially an indication that advanced treatment is needed in order to decrease recurrence and metastatic disease (54, 55). In general, locoregional (N stage) nodes include mesorectal, inferior mesenteric and superior rectal, internal iliac and obturator lymph nodes (55). Anatomically, the internal iliac and obturator nodes are both locoregional and lateral pelvic sidewall nodes. The internal iliac nodes are situated medial to the internal iliac artery and the obturator are lateral at the level of the obturator muscle (55). Non-locoregional (pelvic side wall + distant or M1) lymph nodes associated with rectal cancer include inguinal, external iliac, common iliac, and periaortic nodes (52, 55).
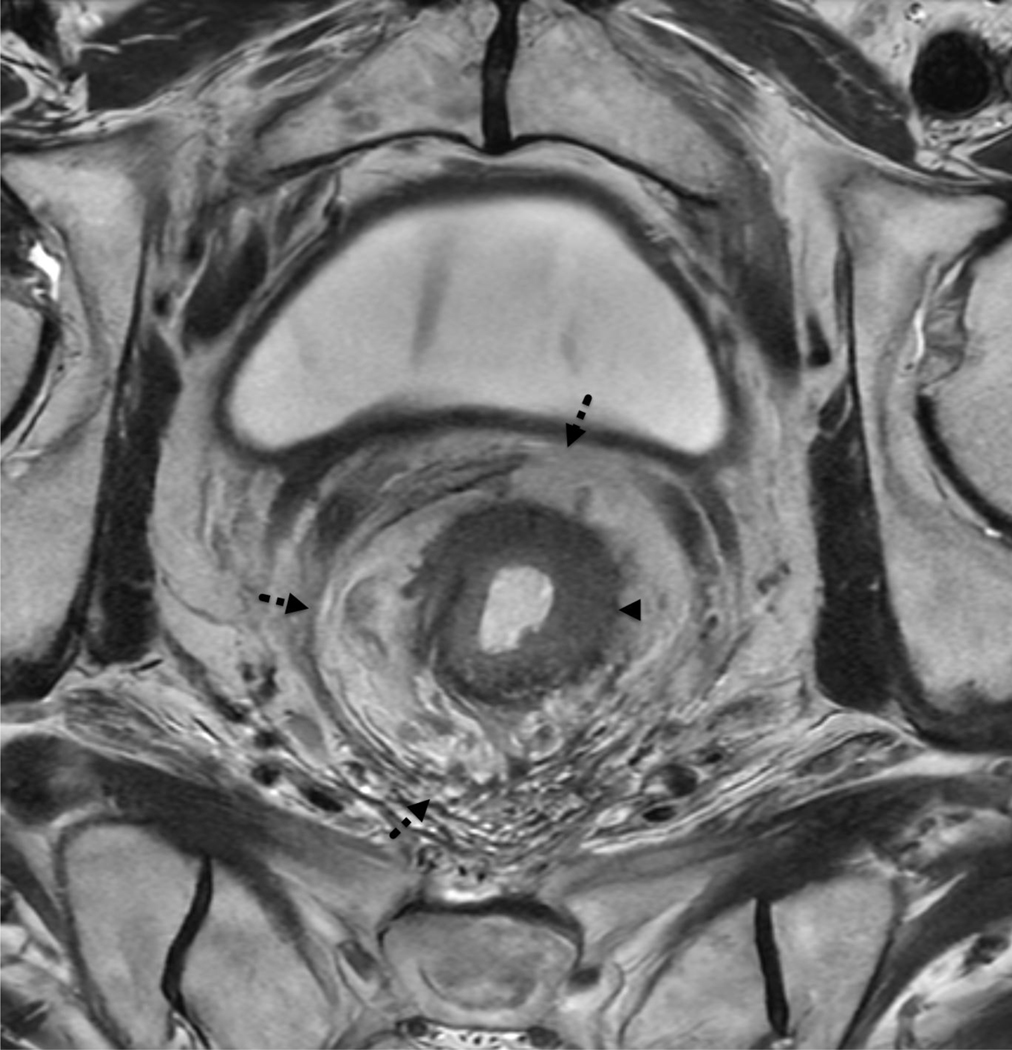
The presence of a suspicious locoregional lymph node (N+/stage III disease) is sufficient evidence to initiate neoadjuvant therapy, irrespective of the T stage. In general, all locoregional nodes with a short axis greater than 9 mm in length should be considered suspicious (56). Nodes with a short axis that are less than 9 mm in length should meet certain morphological or imaging criteria before being considered suspicious. The three major criteria include an irregular or indistinct outline, heterogeneous T2W signal intensity, and a round shape. All three criteria should be met for a locoregional node measuring less than 5 mm in length (56). Nodes measuring between 5–9 mm should meet at least two criteria. When mucinous rectal tumors are present, all T2W hyperintense nodes should be considered suspicious, irrespective of their size (57). In addition, all non-locoregional nodes should be considered suspicious if they measure longer than 10 mm in the short axis. Moreover, it is important to remember that DWI cannot differentiate between inflammatory or malignant nodes and may overestimate the disease burden (56, 58). [FIGURE 4]
Figure 4:
60 year-old male with rectal adenocarcinoma. (a) Coronal oblique T2 weighted image through the rectum shows a low rectal mass (dashed arrow) and a suspicious left obturator lymph node (arrowhead). (b) Axial oblique T2 image through the tumor (arrowhead) does not show definite extramural extension, but the 0.6 cm left obturator node (c, arrowhead) demonstrates a rounded morphology, heterogeneous signal and an irregular border, suspicious for nodal metastasis despite its small size.
A comprehensive MRI report should include the specific location of the suspicious nodes with a comment on locoregional vs. non-locoregional groups. It will allow the surgeons to better identify the type and extent of neoadjuvant treatment required and better determine extent of surgical resection needed (53).
Re-staging after neoadjuvant therapy
Complete surgical resection with negative margin is the standard curative treatment in rectal cancer. Total mesorectal excision (TME) in considered the gold standard surgical procedure and it is known that this approach significantly reduces the incidence of local recurrence. In addition, neoadjuvant chemoradiotherapy for select patients with locally advanced rectal cancer further decreased local recurrence rates(10, 59, 60). After neoadjuvant CRT, approximately 50–60% of patients show downstaging and 15–30% achieve a pathological complete response(61–64). Consequently, higher rates of complete (R0) surgical resection and sphincter-sparing techniques are achieved.
The NCCN guidelines panel recommends neoadjuvant treatment for 2 groups of patients: (a) T1-T2, N+ or T3, N any with clear CRM, and (b) T3, N any with involved or threatened CRM, T4 N any, locally unresectable disease or clinically inoperable. For the first group the options include: chemoradiation (chemoRT, concurrent radiation and fluoropyrimidine-based radiosensitizing chemotherapy over 6 weeks), short-course radiotherapy (25 Gy over 5 days) or total neoadjuvant therapy (TNT, induction chemotherapy followed by either chemoRT or short-course radiotherapy). For the second group, patients may undergo chemoRT alone or followed by 12–16 weeks of chemotherapy, if the CRM is still involved or bulky tumor after the 6 weeks of chemoradiation, or alternatively TNT. For patients with unresectable tumors, doses of radiation higher than 54 Gy may be required and intraoperative RT may be considered. TNT includes both oxaliplatin based chemotherapy as well as radiotherapy. The rational of TNT approach is to provide both local and systemic control of the disease, including for occult micro-metastatic disease (65).
After neoadjuvant treatment, patients with locally advanced rectal cancer are reassessed with digital rectal examination, rectal MRI, and direct endoscopic evaluation. The standard of care is definitive surgical resection, however when a complete clinical response occurs, a multidisciplinary team may decide if the patient is eligible for nonoperative management (so called “watch-and-wait”).
Watch-and-wait management can be considered for careful selected patients with clinical complete response (no evidence of residual tumor) who are being treated in a center with an experienced group (66). If surgery is the treatment of choice, the surgical team should also decide the best surgical approach.
Digital rectal examination and endoscopy provide an excellent luminal assessment of the rectum (67, 68). However, deeper structures relevant for surgical planning are not evaluated, such as bowel wall, mesorectum, lymph nodes and sphincter complex (69). Rectal MRI plays a critical role in the restaging scenario, by providing information regarding presence of residual tumor and which structures are threatened or involved using both morphological (70, 71) and functional sequences and functional sequences (62, 67).
In order to provide the most accurate re-staging MRI interpretation, it is important that the radiologist is aware of the type of neoadjuvant treatment, the time interval since the end of the treatment, endoscopic findings. It is also essential that they have the baseline rectal MRI from the patient available for review. After the radiologist has this information, they can begin to assess the response to therapy. [FIGURE 5]
Figure 5:
61 year-old male with rectal adenocarcinoma status post total neoadjuvant therapy with complete clinical response. (a) Axial oblique T2 weighted image shows semi-circumferential low signal scar in the anterior rectum with adjacent desmoplastic spiculations in the mesorectal fat. (b) A follow-up MRI 9 months later shows a new small intermediate signal mass in the scar (arrowhead), with high signal on b1500 s/mm2 diffusion weighted imaging (c, arrowhead) and corresponding hypointense signal on the apparent diffusion coefficient map (d, arrowhead), consistent with tumor re-growth in the scar.
Defining the tumor bed is the first step. A potential pitfall is that the normal rectal wall adjacent to the tumor may show post treatment changes, such as mucosal edema, wall thickening and high to intermediate signal intensity on T2 weighted images that protrude into the rectal lumen resulting in a “pseudo-tumor” appearance. Correlation with the baseline MRI to define where the tumor was before treatment is extremely helpful to avoid this pitfall.
For non-mucinous tumors, residual tumor appears as intermediate signal intensity on T2 weighted images and persistent restriction diffusion. In contrast, a fibrotic scar appears as markedly low signal intensity on T2 weighted images and demonstrates no residual restricted diffusion. Often variable degrees of residual tumor and fibrotic scar are present. For example, a fibrotic scar with some areas of heterogeneous signal on T2 weighted images and/or equivocal restricted diffusion may be considered a near complete response, although correlation with endoscopy is recommended for these cases. Occasionally no residual tumor and no fibrotic scar are seen, only a normal rectal wall, which can be considered a radiological complete response.
Patients with mucinous tumors on restaging MRI pose a challenge to radiologists, because MRI is not able to distinguish cellular mucin (residual tumor) from acellular mucin (complete response) (72). Mucinous (or colloid) degeneration, occurs when non-mucinous tumor on the baseline MRI becomes mucinous after treatment, but this also does not guarantee a complete pathologic response (73).
Although MR tumor regression grade (TRG) is used in some institutions, studies have suggested that it may not be a reliably reproducible or precise metric (74). Thus routine use of MR TRG in MRI reports has not reached widespread consensus among different organizations (75), and is included here for completeness of the discussion.
TRG on MR relies on assessment of T2 weighted images and is designed to correlate with pathological TRG (70, 76). The MR TRG groups are as follows: TRG1, thin scar and no evidence of tumor; TRG2, thick fibrosis without clear residual tumor or minimal residual tumor; TRG3, tumor and fibrosis (~50%); TRG4, few areas of fibrosis and mostly tumor; TRG5, unchanged or increased tumor without fibrosis.
Just as is done in baseline rectal MR reports, restaging MR reports should describe if there are suspicious lymph nodes by location. Mesorectal and superior rectal lymph nodes are routinely resected, whereas lateral lymph nodes are not unless explicitly identified prior to surgery. Lateral nodes represent an important cause of local recurrence (77).
The most reliable MR features that predict negative lymph node status on pathology are an absence of lymph nodes on DWI, decrease of at least 70% of node size, less than 5 mm in the short axis for mesorectal nodes (78–80) and less than 4 mm in the short axis for pelvic sidewall nodes (78, 79). However, none of these thresholds provides 100% accuracy and individualized discussion may be necessary.
Conclusion
In summary, MRI has become one of the key diagnostic tools in the assessment of patients with rectal cancer. It is of critical importance for baseline assessment, re-staging after neoadjuvant therapy, and for monitoring patients who achieve a complete clinical response who are pursuing “watch and wait” management under the guidance of an experienced multidisciplinary team. In an effort to improve the quality and standardization of rectal MRI, the Rectal and Anal Cancer Disease Focus Panel from the Society of Abdominal Radiology has shared educational resources online, including MR protocols1,2 and reporting templates3,4. The role of MRI in management of patients with rectal cancer is likely to expand, and can contribute to treatment planning and response assessment as multidisciplinary teams work together to best serve our patients.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Al-Sukhni E, Milot L, Fruitman M, Beyene J, Victor JC, Schmocker S, Brown G, McLeod R, Kennedy E. Diagnostic accuracy of MRI for assessment of T category, lymph node metastases, and circumferential resection margin involvement in patients with rectal cancer: a systematic review and meta-analysis. Ann Surg Oncol. 2012;19(7):2212–23. [DOI] [PubMed] [Google Scholar]
- 2.Costa-Silva L, Brown G. Magnetic resonance imaging of rectal cancer. Magn Reson Imaging Clin N Am. 2013;21(2):385–408. [DOI] [PubMed] [Google Scholar]
- 3.Furey E, Jhaveri KS. Magnetic resonance imaging in rectal cancer. Magn Reson Imaging Clin N Am. 2014;22(2):165–90, v-vi. [DOI] [PubMed] [Google Scholar]
- 4.Beets-Tan RGH, Lambregts DMJ, Maas M, Bipat S, Barbaro B, Curvo-Semedo L, Fenlon HM, Gollub MJ, Gourtsoyianni S, Halligan S, Hoeffel C, Kim SH, Laghi A, Maier A, Rafaelsen SR, Stoker J, Taylor SA, Torkzad MR, Blomqvist L. Magnetic resonance imaging for clinical management of rectal cancer: Updated recommendations from the 2016 European Society of Gastrointestinal and Abdominal Radiology (ESGAR) consensus meeting. Eur Radiol. 2018;28(4):1465–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Taylor FG, Swift RI, Blomqvist L, Brown G. A systematic approach to the interpretation of preoperative staging MRI for rectal cancer. AJR Am J Roentgenol. 2008;191(6):1827–35. [DOI] [PubMed] [Google Scholar]
- 6.Group MS. Extramural depth of tumor invasion at thin-section MR in patients with rectal cancer: results of the MERCURY study. Radiology. 2007;243(1):132–9. [DOI] [PubMed] [Google Scholar]
- 7.Gollub MJ, Maas M, Weiser M, Beets GL, Goodman K, Berkers L, Beets-Tan RG. Recognition of the anterior peritoneal reflection at rectal MRI. AJR Am J Roentgenol. 2013;200(1):97–101. [DOI] [PubMed] [Google Scholar]
- 8.Taylor FG, Quirke P, Heald RJ, Moran B, Blomqvist L, Swift I, St Rose S, Sebag-Montefiore DJ, Tekkis P, Brown G, group Ms. One millimetre is the safe cut-off for magnetic resonance imaging prediction of surgical margin status in rectal cancer. Br J Surg. 2011;98(6):872–9. [DOI] [PubMed] [Google Scholar]
- 9.Horvat N, Carlos Tavares Rocha C, Clemente Oliveira B, Petkovska I, Gollub MJ. MRI of Rectal Cancer: Tumor Staging, Imaging Techniques, and Management. Radiographics. 2019;39(2):367–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Benson AB, Venook AP, Al-Hawary MM, Arain MA, Chen YJ, Ciombor KK, Cohen S, Cooper HS, Deming D, Farkas L, Garrido-Laguna I, Grem JL, Gunn A, Hecht JR, Hoffe S, Hubbard J, Hunt S, Johung KL, Kirilcuk N, Krishnamurthi S, Messersmith WA, Meyerhardt J, Miller ED, Mulcahy MF, Nurkin S, Overman MJ, Parikh A, Patel H, Pedersen K, Saltz L, Schneider C, Shibata D, Skibber JM, Sofocleous CT, Stoffel EM, Stotsky-Himelfarb E, Willett CG, Gregory KM, Gurski LA. Colon Cancer, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2021;19(3):329–59. [DOI] [PubMed] [Google Scholar]
- 11.Jayaprakasam VS, Javed-Tayyab S, Gangai N, Zheng J, Capanu M, Bates DDB, Fuqua JL 3rd, Paroder V, Golia-Pernicka J, Gollub MJ, Petkovska I. Does microenema administration improve the quality of DWI sequences in rectal MRI? Abdom Radiol (NY). 2020; 10.1007/s00261-020-02718-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Adam IJ, Mohamdee MO, Martin IG, Scott N, Finan PJ, Johnston D, Dixon MF, Quirke P. Role of circumferential margin involvement in the local recurrence of rectal cancer. Lancet. 1994;344(8924):707–11. [DOI] [PubMed] [Google Scholar]
- 13.Caricato M, Borzomati D, Ausania F, Valeri S, Rosignoli A, Coppola R. Prognostic factors after surgery for locally recurrent rectal cancer: an overview. Eur J Surg Oncol. 2006;32(2):126–32. [DOI] [PubMed] [Google Scholar]
- 14.Gosens MJ, Klaassen RA, Tan-Go I, Rutten HJ, Martijn H, van den Brule AJ, Nieuwenhuijzen GA, van Krieken JH, Nagtegaal ID. Circumferential margin involvement is the crucial prognostic factor after multimodality treatment in patients with locally advanced rectal carcinoma. Clin Cancer Res. 2007;13(22 Pt 1):6617–23. [DOI] [PubMed] [Google Scholar]
- 15.Kelly SB, Mills SJ, Bradburn DM, Ratcliffe AA, Borowski DW, Northern Region Colorectal Cancer Audit G. Effect of the circumferential resection margin on survival following rectal cancer surgery. Br J Surg. 2011;98(4):573–81. [DOI] [PubMed] [Google Scholar]
- 16.Nagtegaal ID, Marijnen CA, Kranenbarg EK, van de Velde CJ, van Krieken JH, Pathology Review C, Cooperative Clinical I. Circumferential margin involvement is still an important predictor of local recurrence in rectal carcinoma: not one millimeter but two millimeters is the limit. Am J Surg Pathol. 2002;26(3):350–7. [DOI] [PubMed] [Google Scholar]
- 17.Nagtegaal ID, Quirke P. What is the role for the circumferential margin in the modern treatment of rectal cancer? J Clin Oncol. 2008;26(2):303–12. [DOI] [PubMed] [Google Scholar]
- 18.Pacelli F, Tortorelli AP, Rosa F, Bossola M, Sanchez AM, Papa V, Valentini V, Doglietto GB. Locally recurrent rectal cancer: prognostic factors and long-term outcomes of multimodal therapy. Ann Surg Oncol. 2010;17(1):152–62. [DOI] [PubMed] [Google Scholar]
- 19.Glynne-Jones R, Mawdsley S, Novell JR. The clinical significance of the circumferential resection margin following preoperative pelvic chemo-radiotherapy in rectal cancer: why we need a common language. Colorectal Dis. 2006;8(9):800–7. [DOI] [PubMed] [Google Scholar]
- 20.Quirke P, Durdey P, Dixon MF, Williams NS. Local recurrence of rectal adenocarcinoma due to inadequate surgical resection. Histopathological study of lateral tumour spread and surgical excision. Lancet. 1986;2(8514):996–9. [DOI] [PubMed] [Google Scholar]
- 21.Hermanek P, Junginger T. The circumferential resection margin in rectal carcinoma surgery. Tech Coloproctol. 2005;9(3):193–9; discussion 9–200. [DOI] [PubMed] [Google Scholar]
- 22.Oberholzer K, Junginger T, Heintz A, Kreft A, Hansen T, Lollert A, Ebert M, Duber C. Rectal Cancer: MR imaging of the mesorectal fascia and effect of chemoradiation on assessment of tumor involvement. J Magn Reson Imaging. 2012;36(3):658–63. [DOI] [PubMed] [Google Scholar]
- 23.Taylor FG, Quirke P, Heald RJ, Moran B, Blomqvist L, Swift I, Sebag-Montefiore DJ, Tekkis P, Brown G, group Ms. Preoperative high-resolution magnetic resonance imaging can identify good prognosis stage I, II, and III rectal cancer best managed by surgery alone: a prospective, multicenter, European study. Ann Surg. 2011;253(4):711–9. [DOI] [PubMed] [Google Scholar]
- 24.Park JS, Huh JW, Park YA, Cho YB, Yun SH, Kim HC, Lee WY, Chun HK. A circumferential resection margin of 1 mm is a negative prognostic factor in rectal cancer patients with and without neoadjuvant chemoradiotherapy. Dis Colon Rectum. 2014;57(8):933–40. [DOI] [PubMed] [Google Scholar]
- 25.Group MS. Diagnostic accuracy of preoperative magnetic resonance imaging in predicting curative resection of rectal cancer: prospective observational study. BMJ. 2006;333(7572):779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Brown G, Daniels IR, Richardson C, Revell P, Peppercorn D, Bourne M. Techniques and troubleshooting in high spatial resolution thin slice MRI for rectal cancer. Br J Radiol. 2005;78(927):245–51. [DOI] [PubMed] [Google Scholar]
- 27.Birbeck KF, Macklin CP, Tiffin NJ, Parsons W, Dixon MF, Mapstone NP, Abbott CR, Scott N, Finan PJ, Johnston D, Quirke P. Rates of circumferential resection margin involvement vary between surgeons and predict outcomes in rectal cancer surgery. Ann Surg. 2002;235(4):449–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shihab OC, Quirke P, Heald RJ, Moran BJ, Brown G. Magnetic resonance imaging-detected lymph nodes close to the mesorectal fascia are rarely a cause of margin involvement after total mesorectal excision. Br J Surg. 2010;97(9):1431–6. [DOI] [PubMed] [Google Scholar]
- 29.Nagtegaal I, Gaspar C, Marijnen C, Van De Velde C, Fodde R, Van Krieken H. Morphological changes in tumour type after radiotherapy are accompanied by changes in gene expression profile but not in clinical behaviour. J Pathol. 2004;204(2):183–92. [DOI] [PubMed] [Google Scholar]
- 30.Shihab OC, Brown G, Daniels IR, Heald RJ, Quirke P, Moran BJ. Patients with low rectal cancer treated by abdominoperineal excision have worse tumors and higher involved margin rates compared with patients treated by anterior resection. Dis Colon Rectum. 2010;53(1):53–6. [DOI] [PubMed] [Google Scholar]
- 31.Park JH, Kim JH, Ahn SD, Lee SW, Shin SS, Kim JC, Yu CS, Kim HC, Kang YK, Kim TW, Chang HM, Ryu MH, Choi EK. Prospective phase II study of preoperative chemoradiation with capecitabine in locally advanced rectal cancer. Cancer Res Treat. 2004;36(6):354–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sebag-Montefiore D, Stephens RJ, Steele R, Monson J, Grieve R, Khanna S, Quirke P, Couture J, de Metz C, Myint AS, Bessell E, Griffiths G, Thompson LC, Parmar M. Preoperative radiotherapy versus selective postoperative chemoradiotherapy in patients with rectal cancer (MRC CR07 and NCIC-CTG C016): a multicentre, randomised trial. Lancet. 2009;373(9666):811–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Roh MS, Colangelo LH, O’Connell MJ, Yothers G, Deutsch M, Allegra CJ, Kahlenberg MS, Baez-Diaz L, Ursiny CS, Petrelli NJ, Wolmark N. Preoperative multimodality therapy improves disease-free survival in patients with carcinoma of the rectum: NSABP R-03. J Clin Oncol. 2009;27(31):5124–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kapiteijn E, Marijnen CA, Nagtegaal ID, Putter H, Steup WH, Wiggers T, Rutten HJ, Pahlman L, Glimelius B, van Krieken JH, Leer JW, van de Velde CJ, Dutch Colorectal Cancer G. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer. N Engl J Med. 2001;345(9):638–46. [DOI] [PubMed] [Google Scholar]
- 35.van der Paardt MP, Zagers MB, Beets-Tan RG, Stoker J, Bipat S. Patients who undergo preoperative chemoradiotherapy for locally advanced rectal cancer restaged by using diagnostic MR imaging: a systematic review and meta-analysis. Radiology. 2013;269(1):101–12. [DOI] [PubMed] [Google Scholar]
- 36.Del Vescovo R, Trodella LE, Sansoni I, Cazzato RL, Battisti S, Giurazza F, Ramella S, Cellini F, Grasso RF, Trodella L, Beomonte Zobel B. MR imaging of rectal cancer before and after chemoradiation therapy. Radiol Med. 2012;117(7):1125–38. [DOI] [PubMed] [Google Scholar]
- 37.Talbot IC, Ritchie S, Leighton MH, Hughes AO, Bussey HJ, Morson BC. The clinical significance of invasion of veins by rectal cancer. Br J Surg. 1980;67(6):439–42. [DOI] [PubMed] [Google Scholar]
- 38.Talbot IC, Ritchie S, Leighton M, Hughes AO, Bussey HJ, Morson BC. Invasion of veins by carcinoma of rectum: method of detection, histological features and significance. Histopathology. 1981;5(2):141–63. [DOI] [PubMed] [Google Scholar]
- 39.Ale Ali H, Kirsch R, Razaz S, Jhaveri A, Thipphavong S, Kennedy ED, Jhaveri KS. Extramural venous invasion in rectal cancer: overview of imaging, histopathology, and clinical implications. Abdom Radiol (NY). 2019;44(1):1–10. [DOI] [PubMed] [Google Scholar]
- 40.Brown G, Radcliffe AG, Newcombe RG, Dallimore NS, Bourne MW, Williams GT. Preoperative assessment of prognostic factors in rectal cancer using high-resolution magnetic resonance imaging. Br J Surg. 2003;90(3):355–64. [DOI] [PubMed] [Google Scholar]
- 41.Zhang XY, Wang S, Li XT, Wang YP, Shi YJ, Wang L, Wu AW, Sun YS. MRI of Extramural Venous Invasion in Locally Advanced Rectal Cancer: Relationship to Tumor Recurrence and Overall Survival. Radiology. 2018;289(3):677–85. [DOI] [PubMed] [Google Scholar]
- 42.Siddiqui MRS, Simillis C, Hunter C, Chand M, Bhoday J, Garant A, Vuong T, Artho G, Rasheed S, Tekkis P, Abulafi AM, Brown G. A meta-analysis comparing the risk of metastases in patients with rectal cancer and MRI-detected extramural vascular invasion (mrEMVI) vs mrEMVI-negative cases. Br J Cancer. 2017;116(12):1513–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chand M, Bhangu A, Wotherspoon A, Stamp GWH, Swift RI, Chau I, Tekkis PP, Brown G. EMVI-positive stage II rectal cancer has similar clinical outcomes as stage III disease following pre-operative chemoradiotherapy. Ann Oncol. 2014;25(4):858–63. [DOI] [PubMed] [Google Scholar]
- 44.Bugg WG, Andreou AK, Biswas D, Toms AP, Williams SM. The prognostic significance of MRI-detected extramural venous invasion in rectal carcinoma. Clin Radiol. 2014;69(6):619–23. [DOI] [PubMed] [Google Scholar]
- 45.Smith NJ, Barbachano Y, Norman AR, Swift RI, Abulafi AM, Brown G. Prognostic significance of magnetic resonance imaging-detected extramural vascular invasion in rectal cancer. Br J Surg. 2008;95(2):229–36. [DOI] [PubMed] [Google Scholar]
- 46.Jhaveri KS, Hosseini-Nik H, Thipphavong S, Assarzadegan N, Menezes RJ, Kennedy ED, Kirsch R. MRI Detection of Extramural Venous Invasion in Rectal Cancer: Correlation With Histopathology Using Elastin Stain. AJR Am J Roentgenol. 2016;206(4):747–55. [DOI] [PubMed] [Google Scholar]
- 47.Kim TH, Woo S, Han S, Suh CH, Vargas HA. The Diagnostic Performance of MRI for Detection of Extramural Venous Invasion in Colorectal Cancer: A Systematic Review and Meta-Analysis of the Literature. AJR Am J Roentgenol. 2019;213(3):575–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chand M, Evans J, Swift RI, Tekkis PP, West NP, Stamp G, Heald RJ, Brown G. The prognostic significance of postchemoradiotherapy high-resolution MRI and histopathology detected extramural venous invasion in rectal cancer. Ann Surg. 2015;261(3):473–9. [DOI] [PubMed] [Google Scholar]
- 49.Corman ML, Corman ML. Corman’s colon and rectal surgery. 2013. [Google Scholar]
- 50.Kaur H, Ernst RD, Rauch GM, Harisinghani M. Nodal drainage pathways in primary rectal cancer: anatomy of regional and distant nodal spread. Abdom Radiol (NY). 2019;44(11):3527–35. [DOI] [PubMed] [Google Scholar]
- 51.Natsugoe S. Lymph Node Metastasis in Gastrointestinal Cancer. 1st ed. Singapore: Springer; 2019. [Google Scholar]
- 52.Harisinghani MG. Atlas of Lymph Node Anatomy. 1st ed. ed. New York, NY, USA: Springer; 2013. [Google Scholar]
- 53.Stoner RC, Korngold EK. Anatomy of Pelvic Lymph Nodal Stations and Their Role in Staging of Rectal Cancers. Semin Roentgenol. 2021;56(2):152–7. [DOI] [PubMed] [Google Scholar]
- 54.Kim DJ, Chung JJ, Yu JS, Cho ES, Kim JH. Evaluation of lateral pelvic nodes in patients with advanced rectal cancer. AJR Am J Roentgenol. 2014;202(6):1245–55. [DOI] [PubMed] [Google Scholar]
- 55.Hope TA, Gollub MJ, Arya S, Bates DDB, Ganeshan D, Harisinghani M, Jhaveri KS, Kassam Z, Kim DH, Korngold E, Lalwani N, Moreno CC, Nougaret S, Paroder V, Paspulati RM, Golia Pernicka JS, Petkovska I, Pickhardt PJ, Rauch GM, Rosenthal MH, Sheedy SP, Horvat N. Rectal cancer lexicon: consensus statement from the society of abdominal radiology rectal & anal cancer disease-focused panel. Abdom Radiol (NY). 2019;44(11):3508–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lalwani N, Bates DDB, Arif-Tiwari H, Khandelwal A, Korngold E, Lockhart M. Baseline MR Staging of Rectal Cancer: A Practical Approach. Semin Roentgenol. 2021;56(2):164–76. [DOI] [PubMed] [Google Scholar]
- 57.Nougaret S, Jhaveri K, Kassam Z, Lall C, Kim DH. Rectal cancer MR staging: pearls and pitfalls at baseline examination. Abdom Radiol (NY). 2019;44(11):3536–48. [DOI] [PubMed] [Google Scholar]
- 58.Mizukami Y, Ueda S, Mizumoto A, Sasada T, Okumura R, Kohno S, Takabayashi A. Diffusion-weighted magnetic resonance imaging for detecting lymph node metastasis of rectal cancer. World J Surg. 2011;35(4):895–9. [DOI] [PubMed] [Google Scholar]
- 59.Heald RJ, Ryall RD. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet. 1986;1(8496):1479–82. [DOI] [PubMed] [Google Scholar]
- 60.Krook JE, Moertel CG, Gunderson LL, Wieand HS, Collins RT, Beart RW, Kubista TP, Poon MA, Meyers WC, Mailliard JA, et al. Effective surgical adjuvant therapy for high-risk rectal carcinoma. N Engl J Med. 1991;324(11):709–15. [DOI] [PubMed] [Google Scholar]
- 61.Habr-Gama A, Perez RO, Nadalin W, Sabbaga J, Ribeiro U Jr., Silva e Sousa AH Jr., Campos FG, Kiss DR, Gama-Rodrigues J. Operative versus nonoperative treatment for stage 0 distal rectal cancer following chemoradiation therapy: long-term results. Ann Surg. 2004;240(4):711–7; discussion 7–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hotker AM, Tarlinton L, Mazaheri Y, Woo KM, Gonen M, Saltz LB, Goodman KA, Garcia-Aguilar J, Gollub MJ. Multiparametric MRI in the assessment of response of rectal cancer to neoadjuvant chemoradiotherapy: A comparison of morphological, volumetric and functional MRI parameters. Eur Radiol. 2016;26(12):4303–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Maas M, Nelemans PJ, Valentini V, Das P, Rodel C, Kuo LJ, Calvo FA, Garcia-Aguilar J, Glynne-Jones R, Haustermans K, Mohiuddin M, Pucciarelli S, Small W Jr., Suarez J, Theodoropoulos G, Biondo S, Beets-Tan RG, Beets GL. Long-term outcome in patients with a pathological complete response after chemoradiation for rectal cancer: a pooled analysis of individual patient data. Lancet Oncol. 2010;11(9):835–44. [DOI] [PubMed] [Google Scholar]
- 64.Pahlman L, Bohe M, Cedermark B, Dahlberg M, Lindmark G, Sjodahl R, Ojerskog B, Damber L, Johansson R. The Swedish rectal cancer registry. Br J Surg. 2007;94(10):1285–92. [DOI] [PubMed] [Google Scholar]
- 65.Petrelli F, Trevisan F, Cabiddu M, Sgroi G, Bruschieri L, Rausa E, Ghidini M, Turati L. Total Neoadjuvant Therapy in Rectal Cancer: A Systematic Review and Meta-analysis of Treatment Outcomes. Ann Surg. 2020;271(3):440–8. [DOI] [PubMed] [Google Scholar]
- 66.Smith JJ, Strombom P, Chow OS, Roxburgh CS, Lynn P, Eaton A, Widmar M, Ganesh K, Yaeger R, Cercek A, Weiser MR, Nash GM, Guillem JG, Temple LKF, Chalasani SB, Fuqua JL, Petkovska I, Wu AJ, Reyngold M, Vakiani E, Shia J, Segal NH, Smith JD, Crane C, Gollub MJ, Gonen M, Saltz LB, Garcia-Aguilar J, Paty PB. Assessment of a Watch-and-Wait Strategy for Rectal Cancer in Patients With a Complete Response After Neoadjuvant Therapy. JAMA Oncol. 2019;5(4):e185896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Maas M, Lambregts DM, Nelemans PJ, Heijnen LA, Martens MH, Leijtens JW, Sosef M, Hulsewe KW, Hoff C, Breukink SO, Stassen L, Beets-Tan RG, Beets GL. Assessment of Clinical Complete Response After Chemoradiation for Rectal Cancer with Digital Rectal Examination, Endoscopy, and MRI: Selection for Organ-Saving Treatment. Ann Surg Oncol. 2015;22(12):3873–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Perez RO, Habr-Gama A, Pereira GV, Lynn PB, Alves PA, Proscurshim I, Rawet V, Gama-Rodrigues J. Role of biopsies in patients with residual rectal cancer following neoadjuvant chemoradiation after downsizing: can they rule out persisting cancer? Colorectal Dis. 2012;14(6):714–20. [DOI] [PubMed] [Google Scholar]
- 69.Duldulao MP, Lee W, Streja L, Chu P, Li W, Chen Z, Kim J, Garcia-Aguilar J. Distribution of residual cancer cells in the bowel wall after neoadjuvant chemoradiation in patients with rectal cancer. Dis Colon Rectum. 2013;56(2):142–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Patel UB, Taylor F, Blomqvist L, George C, Evans H, Tekkis P, Quirke P, Sebag-Montefiore D, Moran B, Heald R, Guthrie A, Bees N, Swift I, Pennert K, Brown G. Magnetic resonance imaging-detected tumor response for locally advanced rectal cancer predicts survival outcomes: MERCURY experience. J Clin Oncol. 2011;29(28):3753–60. [DOI] [PubMed] [Google Scholar]
- 71.Patel UB, Brown G, Rutten H, West N, Sebag-Montefiore D, Glynne-Jones R, Rullier E, Peeters M, Van Cutsem E, Ricci S, Van de Velde C, Kjell P, Quirke P. Comparison of magnetic resonance imaging and histopathological response to chemoradiotherapy in locally advanced rectal cancer. Ann Surg Oncol. 2012;19(9):2842–52. [DOI] [PubMed] [Google Scholar]
- 72.Shia J, McManus M, Guillem JG, Leibold T, Zhou Q, Tang LH, Riedel ER, Weiser MR, Paty PB, Temple LK, Nash G, Kolosov K, Minsky BD, Wong WD, Klimstra DS. Significance of acellular mucin pools in rectal carcinoma after neoadjuvant chemoradiotherapy. Am J Surg Pathol. 2011;35(1):127–34. [DOI] [PubMed] [Google Scholar]
- 73.Horvat N, Hope TA, Pickhardt PJ, Petkovska I. Mucinous rectal cancer: concepts and imaging challenges. Abdom Radiol (NY). 2019;44(11):3569–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Nahas SC, Nahas CSR, Cama GM, de Azambuja RL, Horvat N, Marques CFS, Menezes MR, Junior UR, Cecconello I. Diagnostic performance of magnetic resonance to assess treatment response after neoadjuvant therapy in patients with locally advanced rectal cancer. Abdom Radiol (NY). 2019;44(11):3632–40. [DOI] [PubMed] [Google Scholar]
- 75.Gollub MJ, Arya S, Beets-Tan RG, dePrisco G, Gonen M, Jhaveri K, Kassam Z, Kaur H, Kim D, Knezevic A, Korngold E, Lall C, Lalwani N, Blair Macdonald D, Moreno C, Nougaret S, Pickhardt P, Sheedy S, Harisinghani M. Use of magnetic resonance imaging in rectal cancer patients: Society of Abdominal Radiology (SAR) rectal cancer disease-focused panel (DFP) recommendations 2017. Abdom Radiol (NY). 2018;43(11):2893–902. [DOI] [PubMed] [Google Scholar]
- 76.Trakarnsanga A, Gonen M, Shia J, Nash GM, Temple LK, Guillem JG, Paty PB, Goodman KA, Wu A, Gollub M, Segal N, Saltz L, Garcia-Aguilar J, Weiser MR. Comparison of tumor regression grade systems for locally advanced rectal cancer after multimodality treatment. J Natl Cancer Inst. 2014;106(10). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kim TH, Jeong SY, Choi DH, Kim DY, Jung KH, Moon SH, Chang HJ, Lim SB, Choi HS, Park JG. Lateral lymph node metastasis is a major cause of locoregional recurrence in rectal cancer treated with preoperative chemoradiotherapy and curative resection. Ann Surg Oncol. 2008;15(3):729–37. [DOI] [PubMed] [Google Scholar]
- 78.Ogura A, Konishi T, Cunningham C, Garcia-Aguilar J, Iversen H, Toda S, Lee IK, Lee HX, Uehara K, Lee P, Putter H, van de Velde CJH, Beets GL, Rutten HJT, Kusters M, Lateral Node Study C. Neoadjuvant (Chemo)radiotherapy With Total Mesorectal Excision Only Is Not Sufficient to Prevent Lateral Local Recurrence in Enlarged Nodes: Results of the Multicenter Lateral Node Study of Patients With Low cT3/4 Rectal Cancer. J Clin Oncol. 2019;37(1):33–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ogura A, Konishi T, Beets GL, Cunningham C, Garcia-Aguilar J, Iversen H, Toda S, Lee IK, Lee HX, Uehara K, Lee P, Putter H, van de Velde CJH, Rutten HJT, Tuynman JB, Kusters M, Lateral Node Study C. Lateral Nodal Features on Restaging Magnetic Resonance Imaging Associated With Lateral Local Recurrence in Low Rectal Cancer After Neoadjuvant Chemoradiotherapy or Radiotherapy. JAMA Surg. 2019;154(9):e192172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Beets-Tan RG, Lambregts DM, Maas M, Bipat S, Barbaro B, Caseiro-Alves F, Curvo-Semedo L, Fenlon HM, Gollub MJ, Gourtsoyianni S, Halligan S, Hoeffel C, Kim SH, Laghi A, Maier A, Rafaelsen SR, Stoker J, Taylor SA, Torkzad MR, Blomqvist L. Magnetic resonance imaging for the clinical management of rectal cancer patients: recommendations from the 2012 European Society of Gastrointestinal and Abdominal Radiology (ESGAR) consensus meeting. Eur Radiol. 2013;23(9):2522–31. [DOI] [PubMed] [Google Scholar]