OBJECTIVES:
To investigate the ICU survival of venovenous extracorporeal membrane oxygenation (ECMO) patients suffering from COVID-19–related acute respiratory distress syndrome (ARDS) versus ECMO patients without COVID-19 (non-COVID-19)–related ARDS.
DESIGN:
Preliminary analysis of data from two prospective ECMO trials and retrospective analysis of a cohort of ARDS ECMO patients.
SETTING:
Single-center ICU.
PATIENTS:
Adult ARDS ECMO patients, 16 COVID-19 versus 23 non-COVID-19 patients. Analysis of retrospective data from 346 adult ARDS ECMO patients.
INTERVENTIONS:
None.
MEASUREMENTS AND MAIN RESULTS:
COVID-19 and non-COVID-19 ARDS patients did not differ with respect to preexisting disease or body mass index. ICU survival rate was 62% for COVID-19 ECMO patients and 70% for non-COVID-19 ECMO patients. COVID-19 ECMO survivors were supported with ECMO for a median of 43 days (interquartile range [IQR], 18–58 d) versus 16 days (IQR, 19–39 d; p = 0.03) for non-COVID-19 patients. The median duration of ECMO therapy for all ARDS patients between 2007 and 2018 was 15 days (IQR, 6–28 d). The subgroup of patients suffering from any viral pneumonia received ECMO support for a median of 16 days (IQR, 9–27 d), survivors of influenza pneumonia received ECMO support for 13 days (IQR, 7–25 d).
CONCLUSIONS:
COVID-19 patients required significant longer ECMO support compared with patients without COVID-19 to achieve successful ECMO weaning and ICU survival.
Keywords: COVID-19, duration of extracorporeal membrane oxygenation therapy, extracorporeal membrane oxygenation survival, pulmonary fibrotic remodeling, venovenous extracorporeal membrane oxygenation
Venovenous extracorporeal membrane oxygenation (ECMO) can support systemic oxygenation as “bridge to recovery” in acute respiratory distress syndrome (ARDS)-related to COVID-19. The high global burden of COVID-19 cases resulted in a worldwide increase in ECMO use, although pooled data from the beginning of the pandemic demonstrated an ECMO survival rate below 10% in respective patients (1–3).
Barbaro et al (4) reported a 90-day inhospital mortality of only 37% for COVID-19 ECMO patients, but only 30% of these patients were discharged home or to a rehabilitation facility 90 days after initiation of ECMO.
Considering the potential of COVID-19–related ARDS to result in pulmonary fibrosis, prolonged ECMO support may be required compared with non-COVID-19 ECMO patients to generate a survival benefit (5, 6). Thus, we investigated preliminary data with respect to ECMO survival and ECMO duration of COVID-19 versus non-COVID-19 ECMO patients.
MATERIALS AND METHODS
Study Population
The study population consisted of 21 patients from a pilot trial (ClinicalTrials.gov: NCT03200314; written consent was obtained from the Charité ethics committee prior to each study start, ethics approval (EA) number: EA2/083/17) and 18 patients of a recruiting trial (NCT04754854; EA1/156/20) and was stratified to 16 COVID-19 versus 23 non-COVID-19 patients. All ARDS patients were 18 years or older, required venovenous ECMO, and were treated at the anesthesiological ICU of the ARDS ECMO center of the Charité—Universitätsmedizin Berlin.
The reports of the CT scans of all patients from both prospective studies were screened for findings of pulmonary fibrotic lesions, except for two non-COVID-19 patients who suffered from cystic fibrosis and were consequently not included into this analysis. All CT scans were interpreted by radiologists of the department of radiology of the Charité who were unaware of this study.
The retrospective ECMO cohort consists of 346 patients who suffered from ARDS and were treated between January 2007 and December 2018 with one round of venovenous ECMO therapy (EA1/223/12, EA4/054/21). Data were analyzed for the duration of ECMO therapy with respect to the underlying etiology.
Statistics
Metric data were tested with the nonparametric Mann-Whitney U test because of the small sample size and failure to meet normal distribution (D’Agostino-Pearson normality test) with GraphPad Prism 9.2.1 (GraphPad Software, San Diego, CA). Categorical data were tested with Fisher exact test except for the degree of mobilization at discharge (chi-square test). All tests were performed two-sided. Statistical significance was assumed for p value of less than 0.05. Comparisons of Kaplan-Meier estimates were performed with the log-rank test.
RESULTS
COVID-19 patients did not differ from non-COVID-19 ARDS patients with respect to preexisting disease or body mass index (Supplemental Table 1, http://links.lww.com/CCX/A962). All COVID-19 patients were at least 48 years old, with exception of a 27-year-old patient, who suffered from COVID-19–related ARDS after chemotherapy for Hodgkin lymphoma.
More COVID-19 patients were proned and treated with inhaled nitric oxide compared with non-COVID-19 patients before transfer to the ARDS ECMO center, albeit the difference did not reach statistical significance (Supplemental Table 1, http://links.lww.com/CCX/A962).
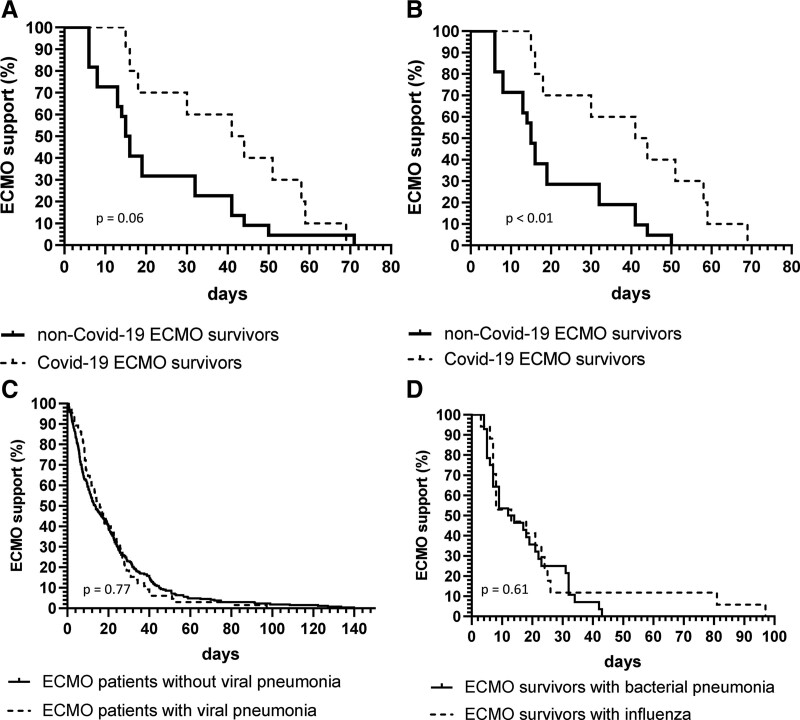
COVID-19 ECMO survivors received longer ECMO support by a median difference of 27 days compared with non-COVID-19 patients (Supplemental Table 1, http://links.lww.com/CCX/A962; and Fig. 1, A and B). The shortest ECMO treatment time of a COVID-19 ECMO survivor was 15 versus 6 days for a non-COVID-19 ECMO survivor.
Figure 1.
Probability of extracorporeal membrane oxygenation (ECMO) support over time with respect to the primary cause of acute respiratory distress syndrome (ARDS). Kaplan-Meier curves of the probability of continued venovenous ECMO support for patients with COVID-19 infection (COVID-19) (A) versus patients without COVID-19 infection (non-COVID-19) (B) after exclusion of one non-COVID-19 patient who was bridged to lung transplantation for 71 d with awake ECMO. C, Probability for non-COVID-19 ECMO patients with viral pneumonia versus non-COVID-19 ECMO patients with ARDS without viral pneumonia (D) for non-COVID-19 ECMO survivors with influenza versus non-COVID-19 ECMO survivors with community-acquired bacterial pneumonia. Comparisons were performed with the log-rank test.
The majority of all COVID-19 patients showed CT morphological signs of fibrotic pulmonary lesions. Fibrotic pulmonary lesions were only diagnosed in one out of 15 non-COVID-19 ECMO survivors but were present in the majority of non-COVID-19 patients who died on ECMO (Supplemental Table 1, http://links.lww.com/CCX/A962).
Patients treated between 2007 and 2018 were supported with venovenous ECMO for a median of 15 days (interquartile range [IQR], 6–28 d). The duration of ECMO therapy was comparable between all examined subgroups, namely: pneumonia (median, 16 d; IQR, 9–27 d); ARDS due to all other causes (14 d; IQR, 6–28 d) (Fig. 1C); ECMO survivors suffering from influenza (13 d; IQR, 7–25 d); and ECMO survivors suffering from community-acquired bacterial pneumonia(13 d; IQR, 6–29 d) (Fig. 1D).
DISCUSSION
This preliminary analysis indicates that COVID-19 ECMO patients may require prolonged ECMO support compared with non-COVID-19 patients to result in ICU survival rates as reported by Barbaro et al (4). The probability of continued ECMO support was significantly higher for COVID-19 patients versus non-COVID-19 after exclusion of one patient, who was bridged with awake venovenous ECMO to lung transplantation (Fig. 1, A and B). In fact, all COVID-19 ECMO survivors were still supported with ECMO by day 14, whereas 20% of non-COVID-19 ECMO survivors had already been weaned of ECMO.
The data of our retrospective ECMO cohort do not demonstrate prolonged duration of ECMO therapy for ARDS caused by viral pathogens different from COVID-19. Our clinical results are in accordance with recent findings, which emphasize the property of severe acute respiratory syndrome coronavirus 2 to induce pulmonary accumulation of macrophages with profibrotic transcriptome and proteome profiles and consequential pulmonary fibrosis (6).
Of note, COVID-19 patients who required mechanical ventilation in the state of Berlin were assigned to designated COVID-19 ICUs by a central organization unit (7). Daily telemedical consultations supported the widespread implementation of awake proning, proning of ventilated patients, and early dexamethasone treatment. As a consequence, the majority of hypoxemic COVID-19 patients may have been sufficiently treated with mechanical ventilation and conservative therapy alone. Thus, only a subgroup of patients with fibrotic pulmonary remodeling and/or progressive ventilator-induced lung injury may have been transferred to the ARDS ECMO centers (5, 6, 8). We speculate whether this “selection” of patients may have resulted in the observed high prevalence of pulmonary fibrotic lesions in the CT findings as well as the prolonged duration of ECMO therapy.
Limitations of the study are the low number of enrolled patients.
Second, due to study design and ethics committee approval, we could only compare ICU survival, but not overall survival at, for example, 90 days.
Third, the diagnosis of pulmonary fibrotic lesions was made with lung CT scans. It included a variety of CT findings from unspecific septal thickening, which may resolve over time, to bronchiectasis and honeycombing. Conceivably, the high burden of disease will have promoted interest in all respective clinical departments with a resulting frequent diagnosis of COVID-19–related pathologies. Hence, we cannot exclude a detection bias concerning the findings of pulmonary fibrotic lesions in our COVID-19 patients.
Last, but most importantly, we did not take lung biopsies from the investigated patients. Thus, we can neither verify the CT findings with histopathological examinations nor exclude further pathologies like COVID-19–related microthrombosis and microangiopathy, which may have also contributed to prolonged ECMO therapy durations (9). Further studies are needed to elucidate the true prevalence and disease burden of pulmonary fibrotic remodeling in COVID-19 ARDS.
In conclusion, the preliminary data presented here indicate that COVID-19 patients require markedly longer ECMO support compared with patients without COVID-19 to achieve successful ECMO weaning and ICU survival.
ACKNOWLEDGMENTS
We acknowledge the technical assistance of our extracorporeal membrane oxygenation (ECMO) perfusionists Andrej Dobbermann and trainee perfusionists Sascha Kirsten concerning the measurements of blood and gas flows on the ECMO circuits. Furthermore, we acknowledge the assistance of our medical student Victoria Bünger for data collection regarding the historic non-COVID ECMO cohort.
Supplementary Material
Footnotes
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal’s website (http://journals.lww.com/ccejournal).
The authors have disclosed that they do not have any potential conflicts of interest.
REFERENCES
- 1.World Health Organization: Weekly Epidemiological Update on COVID-19. 2021. Available at: https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---17-august-2021. Accessed August 20, 2021
- 2.Extracorporeal Life Support Organization: Extracorporeal Life Support Organization (ELSO) Registry. 2021. Available at: https://www.elso.org/. Accessed August 27, 2021
- 3.Henry BM, Lippi G: Poor survival with extracorporeal membrane oxygenation in acute respiratory distress syndrome (ARDS) due to coronavirus disease 2019 (COVID-19): Pooled analysis of early reports. J Crit Care 2020; 58:27–28 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Barbaro RP, MacLaren G, Boonstra PS, et al. ; Extracorporeal Life Support Organization: Extracorporeal membrane oxygenation support in COVID-19: An international cohort study of the Extracorporeal Life Support Organization registry. Lancet 2020; 396:1071–1078 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.George PM, Wells AU, Jenkins RG: Pulmonary fibrosis and COVID-19: The potential role for antifibrotic therapy. Lancet Respir Med 2020; 8:807–815 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wendisch D, Dietrich O, Mari T, et al. : SARS-CoV-2 infection triggers profibrotic macrophage responses and lung fibrosis. Cell 2021; 184:6243–6261.e27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wiesner B, Bachmann M, Blum TG, et al. : Responsibilities of weaning centers during the COVID-19 pandemic outbreak - recommendations for the assignment of ICU capacities in COVID-19 patients as shown by the Berlin-Brandenburg POST-SAVE-Model. Pneumologie 2020; 74:358–365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Grillo F, Barisione E, Ball L, et al. : Lung fibrosis: An undervalued finding in COVID-19 pathological series. Lancet Infect Dis 2021; 21:e72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Manolis AS, Manolis TA, Manolis AA, et al. : COVID-19 infection: Viral macro- and micro-vascular coagulopathy and thromboembolism/prophylactic and therapeutic management. J Cardiovasc Pharmacol Ther 2021; 26:12–24 [DOI] [PMC free article] [PubMed] [Google Scholar]