Abstract
Background and Objectives
Loneliness is common, and its prevalence is rising. The relationship of loneliness with subsequent dementia and the early preclinical course of Alzheimer disease and related dementia (ADRD) remains unclear. Thus, the primary objective of this study was to determine the association of loneliness with 10-year all-cause dementia risk and early cognitive and neuroanatomic imaging markers of ADRD vulnerability.
Methods
This was a retrospective analysis of prospectively collected data from the population-based Framingham Study cohorts (September 9, 1948–December 31, 2018). Eligible participants had loneliness assessed and were dementia-free at baseline. Loneliness was recorded with the Center for Epidemiologic Studies Depression Scale, defined conservatively as feeling lonely ≥3 days in the past week. The main outcomes were incident dementia over a 10-year period, cognition, and MRI brain volumes and white matter injury.
Results
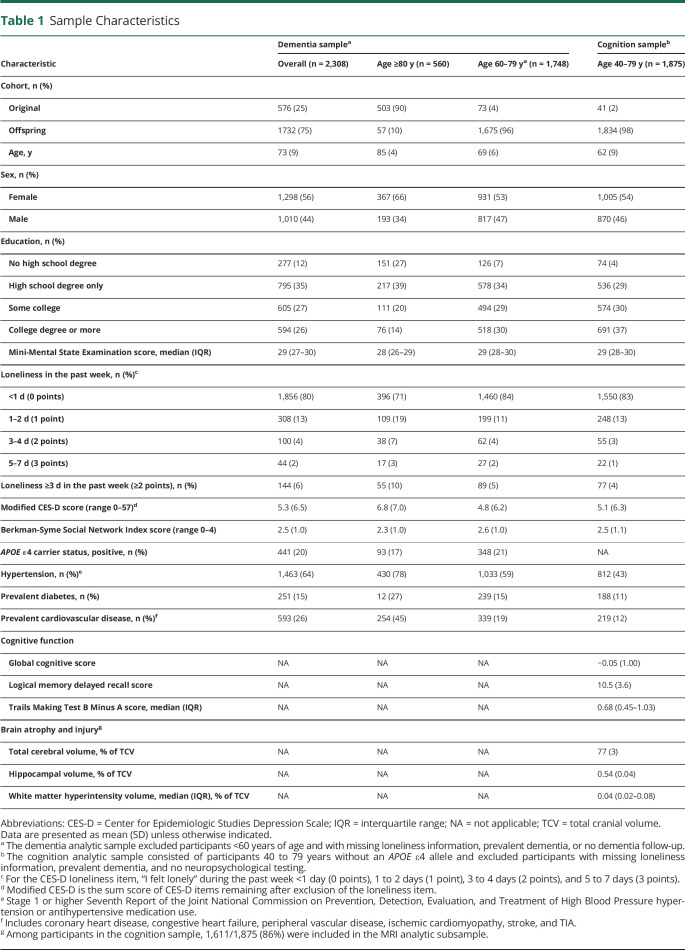
Of 2,308 participants (mean age 73 [SD 9] years, 56% women) who met eligibility in the dementia sample, 14% (329 of 2,308) developed dementia and 6% (144 of 2,308) were lonely. Lonely (versus not lonely) adults had higher 10-year dementia risk (age-, sex-, and education-adjusted hazard ratio 1.54, 95% CI 1.06–2.24). Lonely participants <80 years of age without APOE ε4 alleles had a 3-fold greater risk (adjusted hazard ratio 3.03, 95% CI, 1.63–5.62). Among 1,875 persons without dementia who met eligibility in the cognition sample (mean age 62 [SD 9] years, 54% women), loneliness associated with poorer executive function, lower total cerebral volume, and greater white matter injury.
Discussion
Over 10 years of close clinical dementia surveillance in this cohort study, loneliness was associated with increased dementia risk; this tripled in adults whose baseline risk would otherwise be relatively low on the basis of age and genetic risk, representing a majority of the US population. Loneliness was also associated with worse neurocognitive markers of ADRD vulnerability, suggesting an early pathogenic role. These findings may have important clinical and public health implications given observed loneliness trends.
Classification of Evidence
This study provides Class I evidence that loneliness increases the 10-year risk of developing dementia.
Loneliness is a risk factor for increased morbidity1 and mortality2 and can be reduced,3 yet its prevalence is rising, from 11% to 14% over a 2-year period extending early into the coronavirus disease 2019 pandemic in a US population–based sample,4 amounting to ≈46 million lonely Americans.5 Estimated loneliness prevalence ranges higher for adults ≥60 years of age in US and international cohorts (13%–43%).6-8 Given observed trends, a notable population health concern is that loneliness, especially if persistent, may be an important risk factor for dementia,6,9,10 but findings have been inconsistent.9,11,12
Cohort studies have associated loneliness with increased risk of cognitive decline,6 dementia,10,13 and Alzheimer disease and related dementia (ADRD) neuropathologic changes.14,15 Other results do not support these associations.9,11,12 Regardless of whether loneliness is an early ADRD contributor, symptom, or both, uncertainty about its association with incident dementia risk could underestimate the impact of increasing loneliness and limit understanding of ADRD development and underlying mechanisms. Estimates of dementia risk related to loneliness could be improved with longitudinal studies with more frequent comprehensive clinical and neuropsychological assessments, neurobiological measures that correspond with preclinical ADRD vulnerability, and continuous surveillance for the clinically important endpoint of dementia in a large population-based sample with long duration of extensive monitoring and minimal loss to follow-up. Thus, we analyzed data from the Framingham Study (FS), one of the largest and longest-running longitudinal cohorts in the United States, to determine how the baseline presence (versus absence) of loneliness in a population-based sample of adults associates with risk of developing dementia and indicators of susceptibility for neurocognitive decline. The primary research question of this study was to evaluate, among younger and older adult age groups, the prospective association of loneliness with 10-year incident dementia (confirmed by rigorous and continuous clinical dementia surveillance with adjudication by a dementia review panel of neurologists and neuropsychologists) and the cross-sectional association of loneliness with early markers of ADRD vulnerability: cognitive function (confirmed by standardized and comprehensive neuropsychological evaluations), brain volumes (confirmed by MRI measures of total cerebral volume and hippocampal volume), and small vessel cerebrovascular injury (confirmed by MRI measure of white matter hyperintensities).
Methods
Samples
This study included FS Original cohort participants attending their 25th biennial examination (1997–1999) and Offspring participants attending their seventh quadrennial examination (1998–2001). For the primary dementia analysis, eligible participants had a loneliness assessment and were at least 60 years of age (participants <60 years of age are unlikely to have substantial 10-year risk of dementia).16 Persons were excluded if they had prevalent dementia or no dementia follow-up. The subsequent analysis evaluated the association of loneliness with early cognitive and neuroimaging indicators of ADRD pathology. Because neuropsychological17 and MRI neuroanatomic18 changes are well known to accompany underlying accumulation of ADRD neuropathologic changes many years before its clinical manifestation as dementia, eligible participants were 40 to 79 years of age and had a loneliness assessment and cognitive testing. Participants were excluded if they had prevalent dementia or stroke. Given our predetermined approach for secondary biological validation with this sample and subsequent results of exploratory interaction analyses, participants were also excluded in this sample if they had no genotypic information or an APOE ε4 allele. Imaging analyses used a subset who also had a brain MRI.
Standard Protocol Approvals, Registrations, and Patient Consents
All participants provided written informed consent. The Institutional Review Board of Boston University Medical Center approved the consent form and study protocol.
Exposure
Methods quantifying loneliness with the Center for Epidemiologic Studies Depression Scale (CES-D)19 in large longitudinal cohorts like FS have previously been described.6,10 In brief, the CES-D asks respondents to report whether each of 20 depressive symptoms was present during the past week “rarely or none of the time (<1 day)” (0 points), “some or a little of the time (1–2 days)” (1 point), “occasionally or a moderate amount of time (3–4 days)” (2 points), or “most or all of the time (5–7 days)” (3 points). Identical to similar cohort studies that used the CES-D item asking how often the respondent felt lonely,6,10 participants were classified as lonely (3–7 days) or not lonely (0–2 days). This approach has yielded both positive and null results in similar community-based cohorts of older adults, and positive associations have demonstrated a pattern similar to exposure-response relationships whereby participants who have more severe and persistent loneliness are more likely to have greater ADRD risk.6,10
Although instruments assessing burden of depressive symptoms like the CES-D include items that ask about feelings of loneliness because of their co-occurrence,19 there is growing consensus that depression and loneliness are conceptually separate constructs that are statistically separable,6,20 represent distinct clinical phenomena,21 involve distinct brain circuitry,22,23 and have associations with ADRD neuropathologic changes that are independent from one another14,15,24 and, in this context, have limited to at most moderate overlap.6,10 However, because loneliness (as a predisposing variable in depression) has been observed to have consistently moderate effects on depressive symptoms across studies regardless of sampling strategies, publication type, and publication year,25 the CES-D loneliness item was selected as the main exposure variable while still accounting for depressive symptoms as a potential confounder with a modified version of the CES-D.
Incident Dementia Assessment
The primary outcome was clinical diagnosis of incident all-cause dementia during a 10-year follow-up period after baseline examination. The rigorous and continuous FS dementia surveillance methods have been published previously.26 In brief, cognitive status has been monitored with Mini-Mental State Examination27 and neuropsychological testing since 1975 in the Original cohort and since 1987 in the Offspring cohort. A dementia review panel of study neurologists and neuropsychologists reviewed every possible dementia case and determined the diagnosis date using extensive data such as serial and prompted assessments, telephone interviews with family/caregivers, and medical records.26 Diagnosis of dementia is based on criteria including the DSM-IV.28 Cases detected before 2001 had repeat reviews after 2001 so that updated diagnostic criteria could be applied. Continuous dementia surveillance occurred through December 31, 2018.
Cognitive Assessment
Clinic examination attendees had standardized neuropsychological testing administered by trained research assistants and neuropsychologists after clinical evaluation. Tests performed are reliable, are widely used, and cover all domains of the Alzheimer's Disease Center's Uniform Data Set.29 For the present study, we selected 3 separate measures to analyze that reflect key cognitive processes commonly affected early in ADRD17: Logical Memory Delayed Recall (memory), Trails Making Test B Minus A (executive function), and global cognition. For global cognition, we used a global cognitive score that was developed on a data sample collected at Offspring examination 7 with principal component analysis forcing a single component solution. The tasks included in the principal component analysis are the following: Trails Making Test A, Trails Making Test B, Logical Memory (Immediate and Delayed Recall), Visual Reproductions (Immediate and Delayed Recall), Paired Associate Learning (Delayed Recall), Hooper Visual Organization Test, and Similarities Test.30 The global cognitive score is a weighted sum of standardized scores; higher scores represent better performance. This method is identical to previous studies, is useful in addressing the neuropathologic and clinical heterogeneity of all-cause dementia in community-based samples of older adults,31 and is described in further detail elsewhere.32 Its creation is summarized in eTable 1, links.lww.com/WNL/B792.
Brain MRI Assessment
Preclinical ADRD MRI measures restricted only to cortical subregions might be less sensitive to the full range of underlying ADRD neuropathology in a population-based sample33; thus, our imaging analyses used the following key measures most likely to represent early ADRD cerebral vulnerability in the form of atrophy or microangiopathic white matter injury: total cerebral volume, hippocampal volume, and white matter hyperintensities. Participants were scanned during the baseline examination period with a Siemens Magnetom MRI (1.5T; Siemens Healthineers, Erlangen, Germany). Full details of acquisition, image analysis, and quantification of MRI variables have been described elsewhere, including imaging parameters and sequences, measurement protocols, segmentation methods, reliability, and reproducibility.34 Volume measures were corrected for head size by use of the ratio of each measure over total cranial volume multiplied by 100.34,35 All MRI variables were standardized in analyses. Additional details of imaging acquisition and quantification methods are provided in eMethods, links.lww.com/WNL/B792.
Covariates
Educational achievement was assessed with a 4-level variable (no high school degree, high school degree only, some college, college degree or more). Additional adjustments for depressive symptoms, social isolation, antidepressant medication use, and common vascular risk factors were performed in secondary sensitivity analyses. Regarding depressive symptoms, a re-evaluation of the factor structure of the original CES-D36 used a factor solution of 3 factors (somatic symptoms, negative affect, anhedonia) and demonstrated that, although the loneliness item was retained as a measure of negative affect, the weight of the loneliness item was the lowest within this factor and was relatively consistent across diverse participants, including undergraduate, community, rehabilitation, and clinical samples, as well as an epidemiologic sample comparable to the FS cohorts. Thus, depressive symptoms were accounted for with a modified CES-D score; it excluded the loneliness item, summed points for remaining questions (range 0–57 points), and was a continuous log-transformed variable to account for nonnormal distribution.37 Regarding social isolation, the self-reported Berkman-Syme Social Network Index was used to control for social isolation on the basis of social network size.1 Identical to prior methods,38,39 the Social Network Index classified individuals according to summed score cutoffs as socially isolated (0–1 points) or not socially isolated (2–4 points).
For interaction analyses, APOE ε4 allele carrier status was included as an independent genetic covariate instead of a polygenic risk score given its greater clinical and epidemiologic utility for considering the role of genetic predisposition in the present study.40 APOE genotype was determined by isoelectric focusing of collected plasma and confirmed by DNA genotype.41
Statistical Analysis
To characterize age differences in the association of loneliness with dementia risk, the primary dementia analysis was performed for the entire sample and stratified by age group: <80 and ≥80 years of age. The decision to characterize age differences by stratifying the sample at 80 years of age was made a priori and based on knowledge of extant literature reporting associations of loneliness with greater cortical amyloid and regional tau accumulation using PET imaging,14,15 previous FS investigations suggesting that loneliness-related dementia risk10,39 and risk of neurocognitive markers of early ADRD vulnerability42 varied between younger and older adults, and loneliness prevalence estimates demonstrating a slight increase by age for young-old adults (age 65–79 years) and a more notable increase for oldest-old adults ≥80 years of age.43
For the primary dementia analysis, overall and by age, Cox proportional hazards regression models estimated age-, sex-, and education-adjusted hazard ratios for 10-year dementia risk comparing participants who were lonely with those who were not lonely. To systematically assess for effect modification, interactions were evaluated using Cox models with interaction terms for each of the following variables by loneliness: sex, education level, and APOE ε4 status. Then, stratified Cox regression models were performed as indicated by significant interaction terms. The proportional hazards assumption was upheld for reported models, assessed by analyzing Schoenfeld residuals.
To characterize the role of loneliness in early ADRD susceptibility before the onset of dementia, a second sample was derived that was based on significant age-group-by-modifier subsets in the dementia analysis. The new sample consisted of participants who had neuropsychological testing at the baseline evaluation; most had a baseline brain MRI obtained. We used adjusted multivariable linear regression models in this second sample to assess whether loneliness status was an independent predictor of selected neurocognitive measures. All models adjusted for age, age2, sex, educational achievement, and time interval from loneliness assessment to when the cognitive or MRI measure was acquired. β Coefficient estimates were expressed in SD units (SDU), so each 1-unit change corresponds to a SD difference in each measure.
For sensitivity analysis, the primary dementia, cognition, and imaging models (model 1) were additionally adjusted in sequential models to account for baseline depressive symptoms (model 2), social isolation (Model 3), antidepressant medication use (model 4), and vascular risk factors (model 5). Participants with missing covariate data were excluded from these sensitivity analyses. To investigate the possibility that loneliness is a very early ADRD symptom (rather than an ADRD potentiator), we examined primary dementia models after excluding persons with prevalent mild cognitive impairment; separately, we also delayed the follow-up period to begin 5 years after baseline and to span 5 years, allowing a more conservative assessment of the role of loneliness before ADRD clinical expression. Additional sensitivity analyses used a 3-level ordinal term for loneliness in the primary dementia, cognition, and imaging models. To evaluate risk by dementia subtype, we used the primary dementia models to separately assess risk of developing Alzheimer disease and vascular dementia.
Statistical significance was determined with a 2-sided α level of 0.05 without appropriate indication to correct for multiple comparisons given that each planned comparison tested a different specific hypothesis in the main analyses of the present study.44 Exploratory interaction analyses in the dementia sample used an α level of 0.10 to increase sensitivity, consistent with prior FS studies assessing effect modification.45 Analyses were performed with SAS 9.4 (SAS Institute Inc, Cary, NC). This study followed Strengthening the Reporting of Observational Studies in Epidemiology reporting guidelines (eMethods, links.lww.com/WNL/B792).
Data Availability
The FS datasets analyzed for the present study are available through formal data-use agreements. Any investigator may access the data through the process outlined on the FS website (framinghamheartstudy.org).
Results
Association of Loneliness and 10-Year Risk of Incident Dementia
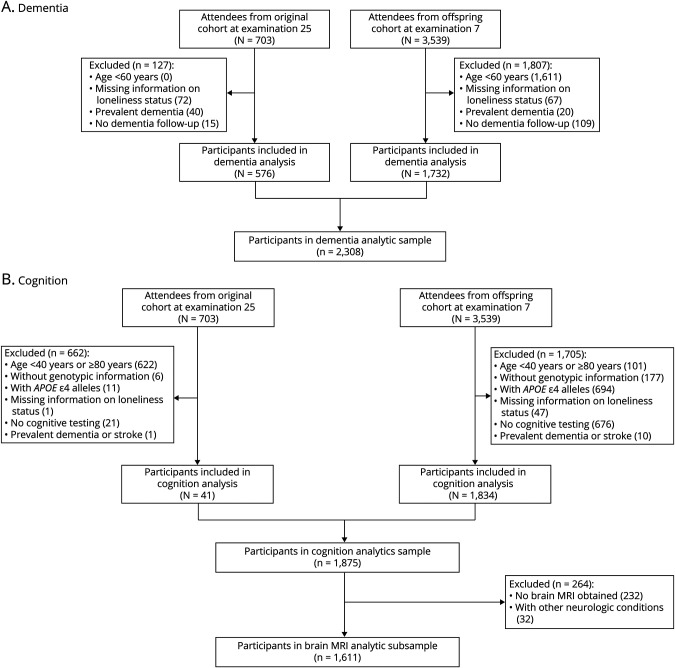
In the dementia analytic sample, 2,308 participants (576 Original and 1732 Offspring members) met inclusion criteria (Figure 1A). Mean age was 73 years (SD, 9 years); 56% were women; and 53% had at least some college education (Table 1). Participants ≥80 years of age were more likely to have prevalent cardiovascular disease and no college degree. At baseline, almost all Mini-Mental State Examination scores were in the cognitively unimpaired range (median score 29 [interquartile range 27–30], range 15–30). Twenty percent of participants had at least 1 APOE ε4 allele. Age-based subgroups had similar social network size and low depressive symptoms. The prevalence of participants reporting loneliness at least 1 day in the past week—452 of 2,308 (20%)—was comparable to that of similar cohorts of older adults.6-8,46,47 Overall, 144 of 2,308 (6%) participants felt lonely ≥3 days in the past week.
Figure 1. Sample Derivation.
Sample derivation flow diagrams for the Framingham Heart Study participants in the (A) dementia and (B) cognition analytic samples.
Table 1.
Sample Characteristics
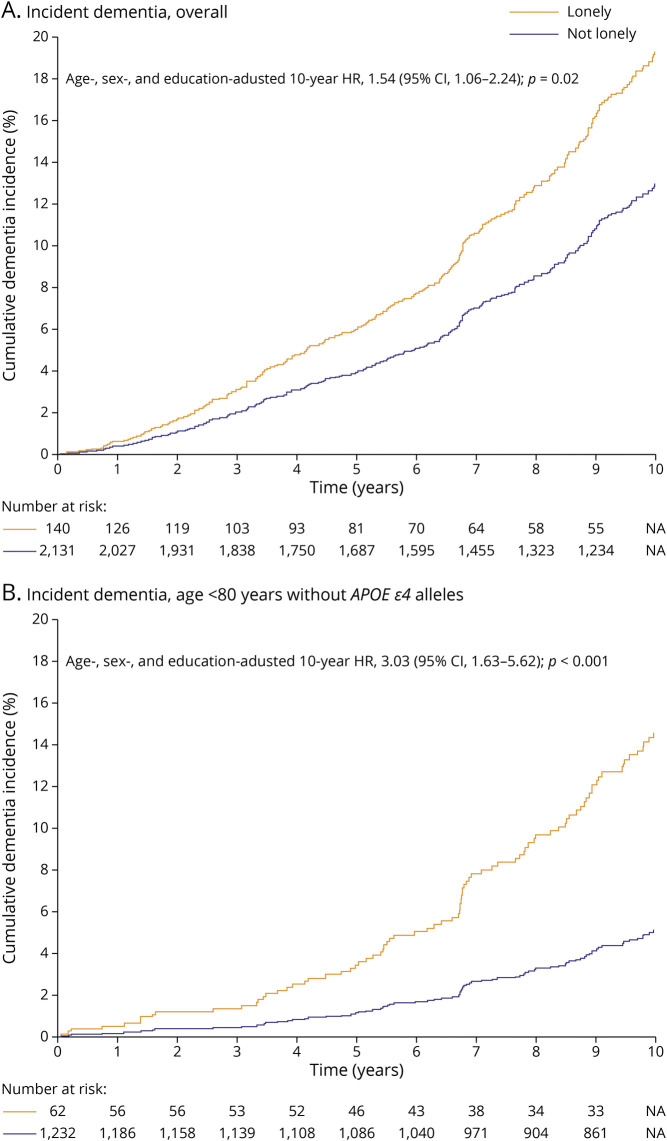
During the 10-year follow-up period (median 10.00 [interquartile range 5.50–10.00] years), 329 of 2,308 (14%) participants were diagnosed with dementia (189 Original and 140 Offspring members). Thirty-one of 144 (22%) lonely participants developed dementia. In the overall dementia sample, after adjustment for age, sex, and educational achievement, loneliness was significantly associated with higher dementia incidence (hazard ratio 1.54, 95% CI 1.06–2.24) (Table 2 and Figure 2A). There was no significant association between loneliness and incident dementia in participants ≥80 years of age; however, among participants <80 years of age, those who were lonely were more than twice as likely to develop dementia (hazard ratio 2.27, 95% CI 1.32–3.91). In the subsequent assessment of effect modification in the younger group, an interaction with APOE ε4 status was identified (Table 2). Loneliness was associated with a 3-fold increased hazard of incident dementia in the younger group of participants who did not carry an APOE ε4 allele (hazard ratio 3.03, 95% CI 1.63–5.62) (Figure 2B).
Table 2.
Adjusted 10-Year Risk of Incident All-Cause Dementia by Loneliness Status
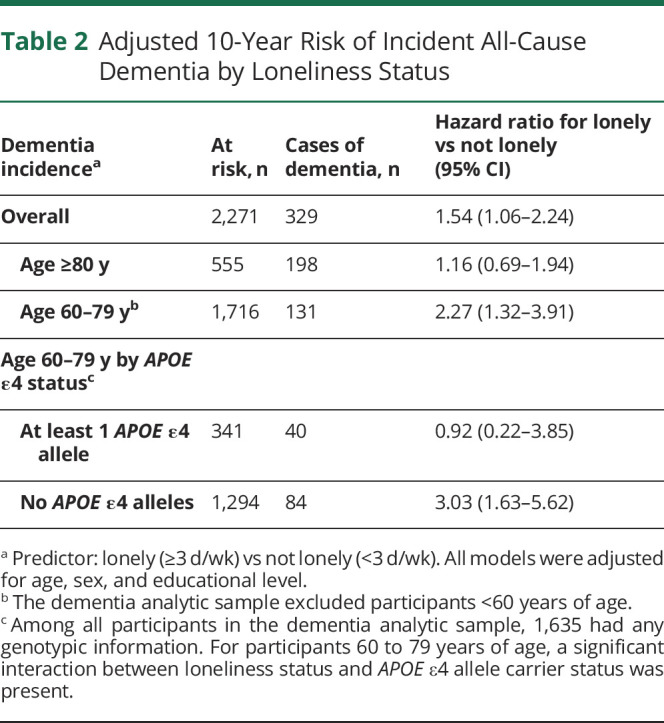
Figure 2. Ten-Year Cumulative Incidence of Dementia by Loneliness Status.
Cumulative incidence curves for the primary outcome, 10-year incident all-cause dementia (A) overall and (B) for the subgroup of participants 60 to 79 years of age without an APOE ε4 allele. Median follow-up time for lonely participants was 5.94 (interquartile range 2.90–10.00) years; for participants who were not lonely, 10.00 (interquartile range, 5.89–10.00) years. HR = hazard ratio; NA = not available.
Association of Loneliness With Early Cognitive and Neuroanatomic Markers of ADRD Vulnerability
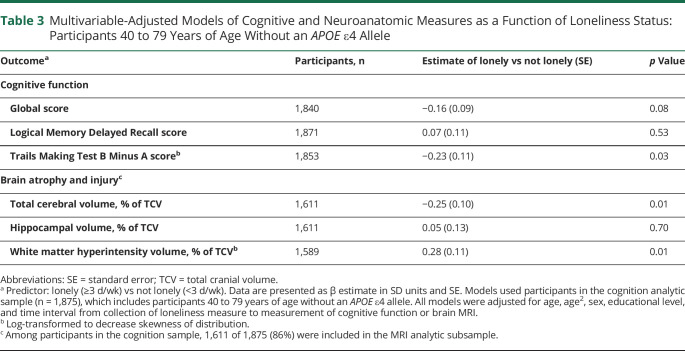
To better understand the relationship between loneliness and ADRD vulnerability in dementia-free adults, we used the cognition sample that consisted of 1,875 participants who met inclusion criteria (41 Original and 1834 Offspring members) (Figure 1B). Mean age was 62 years (SD 9 years); 54% were women; and 67% had at least some college education (Table 1). Loneliness was associated with poorer cognition in the executive function domain; lonely participants scored 0.23 SDU lower than participants who were not lonely (Table 3). A subset of the cognition sample was used to examine the relationship between loneliness and neuroanatomic measures. Baseline brain MRI was obtained for 1,643 of 1,875 (88%) participants (Figure 1B). The MRI subset also excluded 32 participants who had other neurologic conditions; the remaining 1,611 participants were representative of the full cognition sample (Table 1). Lonely (versus not lonely) participants were more likely to have lower total cerebral volume and greater white matter hyperintensity burden. Lonely participants had total cerebral volumes that were 0.25 SDU lower and white matter hyperintensity volumes that were 0.28 SDU greater (Table 3). There were no significant associations between loneliness and the global cognitive score, Logical Memory Delayed Recall score, or hippocampal volume.
Table 3.
Multivariable-Adjusted Models of Cognitive and Neuroanatomic Measures as a Function of Loneliness Status: Participants 40 to 79 Years of Age Without an APOE ε4 Allele
Sensitivity Analysis
Primary findings in the dementia analysis persisted after excluding 88 participants with baseline mild cognitive impairment (hazard ratio 1.92, 95% CI 1.29–2.85) and after advancing follow-up by 5 years (hazard ratio 1.83, 95% CI 1.08–3.12). Loneliness remained consistently associated for the younger group (including the APOE ε4-negative subset) after additional adjustments for depressive symptoms, social isolation, antidepressant medication use, and common vascular risk factors (eTable 2, links.lww.com/WNL/B792). Similarly, loneliness was still associated with greater white matter hyperintensities after additional adjustments (eTable 3). The magnitude of the association between loneliness and total cerebral volume reduced with adjustment for social isolation. Loneliness was no longer associated with cognition after adjustment for depressive symptoms. Using an ordinal term for loneliness in primary models produced similar results overall (eTables 4 and 5) except that reporting loneliness 1 to 2 d/wk in the older age group was also associated with increased dementia risk. In an examination of the association of loneliness with incidence of separate dementia subtypes, loneliness was associated with dementia due to Alzheimer disease. The association was not observed for dementia due to vascular dementia, although the hazard ratio remained in the expected direction (eTable 6).
Classification of Evidence
The primary research question of this study was to evaluate, among younger and older adult age groups, the prospective association of loneliness with 10-year incident dementia (confirmed by rigorous and continuous clinical dementia surveillance with adjudication by a dementia review panel of neurologists and neuropsychologists) and the cross-sectional association of loneliness with early markers of ADRD vulnerability: cognitive function (confirmed by standardized and comprehensive neuropsychological evaluations), brain volumes (confirmed by MRI measures of total cerebral volume and hippocampal volume), and small vessel cerebrovascular injury (confirmed by MRI measure of white matter hyperintensities). This study provides Class I evidence that loneliness increases the 10-year risk of developing dementia.
Discussion
This cohort study found that loneliness was associated with an increased 10-year incidence of all-cause dementia, reaffirming the elevated dementia risk of loneliness demonstrated by others9,12 but also revealing a tripling in risk for lonely adults <80 years of age without an APOE ε4 allele. This association between loneliness and 10-year dementia risk may be at least partly due to the involvement of loneliness in the earliest stages of ADRD neuropathogenesis, as suggested by our subsequent findings characterizing how loneliness in dementia-free adults relates to early cognitive and imaging measures of ADRD vulnerability. Among APOE ε4–negative adults <80 years of age, the highest-risk subgroup identified, loneliness was associated with (1) poorer executive function (a cognitive domain that includes processes such as attention, judgment, organization, planning, and decision-making), (2) lower total cerebral volume (a global neuroanatomic measure that is often more sensitive to the broad range of neuropathologies underlying cognitive decline in population-based samples than only cortical subregions),33 and (3) a greater volume of white matter hyperintensities (indicating greater extent of small vessel cerebrovascular injury). Taken together, these results implicate reporting loneliness ≥3 days in the past week as an important potentially modifiable psychosocial factor for middle-aged and young-old APOE ε4–negative adults early in the ADRD neuropathologic process leading up to the clinical presentation and diagnosis of dementia.
Although our results were consistent with the direction of prior associations reported between loneliness and dementia in population-based samples of older adults,9,12,13 the estimated risk of incident dementia in younger APOE ε4–negative participants was much higher than in other studies. For example, our estimate was greater than the 40% increased dementia risk found in the Health and Retirement Study among comparable lonely individuals ≥65 years of age who were also followed up for 10 years but had their cognitive status assessed with 3 tasks from a brief telephone interview designed for screening dementia.13 It is notable that our findings are also consistent with a recent FS study using only Offspring cohort participant data that observed that persistent loneliness in midlife (defined as reporting loneliness ≥1 day in the past week at the sixth and seventh quadrennial examinations) was associated with higher lifetime risk of dementia and Alzheimer disease (dementia hazard ratio 1.91, 95% CI 1.25–2.90).10 This risk with persistent loneliness was similarly elevated for APOE ε4 noncarriers (hazard ratio 2.32, 95% CI 1.49–3.62), although not as high as the 3-fold risk increase observed in the present study of loneliness reported at a single time point, at baseline, which may have clinical and public health relevance for brief screening assessments of risk. Compared to prior studies, the risk estimates in our investigation may be due to a combination of larger sample size, different loneliness assessments and definitions, parsimonious approach to enhancing sensitivity with the use of high-risk subgroups, longer follow-up period, and follow-up beginning at a time when individuals–based on age–were more likely to develop dementia in subsequent years. Although loneliness was measured with a single-item assessment from an instrument originally designed to evaluate depressive symptoms, our findings reasonably suggest that the increased dementia risk observed in lonely adults may be due to related increases in early ADRD vulnerability through pathways involving multifocal brain atrophy, extensive microvascular injury, and consequent cognitive decline.
Whether loneliness is an early symptom of neurodegenerative disease or an early contributor to neuropathology and cognitive decline remains unclear. On the one hand, if we consider loneliness as an early symptom of neurodegeneration, the present findings may reflect the earliest manifestations of cortical β-amyloid14 and tau15 accumulation in brain areas that both are affected early in the course of ADRD and are involved in the perception of loneliness and other neurobehavioral functions.23 On the other hand, if loneliness is mainly a causal factor in cognitive decline, the present findings may be indicative of loneliness driving (or compounding) vascular, neuroendocrine, inflammatory, and cytotoxic injury biological pathways that have been proposed to be specific to the distress of loneliness21 or increased functional communication in neural systems implicated in generating one's perception of loneliness (e.g., the default mode network).23 Regardless, although the present study is observational, it supports an upstream contributory role for loneliness given that participants without dementia were followed up for 10 years, which is longer than might typically be expected for dementia to develop if mild cognitive or neurobehavioral symptoms were present due to underlying ADRD pathologic changes. This was additionally supported by sensitivity analyses conducted to assess for the potential of reverse causality.
Our finding that lonely young-old adults without the APOE ε4 allele had elevated dementia risk may be due to APOE ε4 carriers having a higher genetic load for ADRD, so loneliness may not have been as influential in determining dementia risk. Loneliness also might have had a role in reducing APOE ε2 and ε3 expression through proposed stress and immune dysregulation pathways,48 leading to lower APOE in blood and the brain and consequently greater susceptibility to β-amyloid aggregation and ADRD pathogenesis. Moreover, the absent association between loneliness and dementia among individuals >80 years of age may be attributable to loneliness having a less prominent role in ADRD when other age-related biological factors account for a greater proportion of incident dementia after 80 years of age (e.g., tauopathy, TAR DNA-binding protein 43 proteinopathy, arteriosclerosis, and hippocampal sclerosis).49 Thus, for younger individuals without inherent genetic risk factors, loneliness and related psychosocial determinants of health may be more influential than traditional ADRD clinical risk factors, acting through pathways independent of or compounding biology.
Overall, these results have implications for dementia risk stratification, the relevance of basic loneliness screening, and the potential to underestimate dementia risk in lonely young-old adults without known genetic risk factors. As the proportion of lonely adults increases within societies, so does the potential to underestimate the associated risk of developing dementia. The current study observed not only a robust independent relationship among loneliness, incident dementia, and early ADRD vulnerability but also a notably higher risk for a specific subgroup that includes the estimated 61% of Black adults and 74% of White adults in the United States who do not possess an APOE ε4 allele.50 One of the strengths of neurologists and other health care providers is a broad perspective of the biological, clinical, and public health implications of the present findings, as well as an appreciation for the value of interdisciplinary strategies to address loneliness. These results raise the importance of future studies to further investigate mechanisms, possible interventions, and tools to efficiently screen for loneliness in routine clinical care (e.g., asking the question used in this study: In the past week, how often have you felt lonely?).
A strength of the present analysis is the rich data source including 2 distinct but similar cohorts followed up over many decades with minimal loss to follow-up. To minimize potential bias from unmeasured confounding that could influence loneliness patterns over time, analyses accounted for baseline differences between cohorts and other factors that could simultaneously affect loneliness and dementia risk or loneliness and ADRD-related neurocognitive measures.
Like other studies using data from the FS Original and Offspring cohorts, this sample is composed of mostly White individuals. Although the sample was smaller than in some prior studies,13 it was large enough to address the questions of interest and in a cohort with comprehensive follow-up spanning >6 decades. Although information on additional symptoms related to loneliness was not available for participants and was not included (e.g., feeling left out or misunderstood), the loneliness item of the CES-D is sufficiently distinct from the other CES-D items that the possibility of substantial overlap with depression or confounding from depressive symptoms that may manifest differently across age groups and various phenotypes of depression was likely low to moderate.36 Thus, this approach was considered to be adequate and appropriate. Although loneliness was slightly more prevalent in the older age group (consistent with similar community-based samples of older adults),6-8,46 it remains possible that depression without loneliness had a more influential role than loneliness without depression in this age group. Although the possibility for reverse causality cannot be ruled out in this cohort study (i.e., our findings could have been the result of underlying ADRD neuropathology manifesting as feelings of loneliness years or decades before clinical diagnosis), studying causal relationships between loneliness and incident dementia is not readily suitable for randomized clinical trials, and hence, conclusions may rely on cohort studies.
In this cohort study, loneliness was associated with higher 10-year risk of incident dementia; notably, lonely APOE ε4–negative younger adults had a 3-fold increased risk. This elevated risk is possibly due to associations identified between loneliness and early cognitive and neuroanatomic markers of ADRD vulnerability, raising potential population health implications for observed trends in loneliness.
Acknowledgment
The authors are grateful to the FS participants who have committed so much of their time and effort.
Glossary
- ADRD
Alzheimer disease and related dementia
- CES-D
Center for Epidemiologic Studies Depression Scale
- DSM-IV
Diagnostic and Statistical Manual of Mental Disorders, 4th edition
- FS
Framingham Study
- SDU
SD units
Appendix. Authors

Footnotes
Study Funding
This research was supported by the Boston University School of Medicine, Harvard Medical School, the Henry and Allison McCance Center for Brain Health, the Massachusetts General Hospital, and the New York University Grossman School of Medicine; contracts from the National Heart, Lung, and Blood Institute (N01-HC 25195, HHSN268201500001I, 75N92019D00031); grants from the National Institute on Neurological Disorders and Stroke (NS017950, UH2NS100605, T32NS048005) and National Institute on Aging [AG049505, AG052409, AG054076, AG049607, AG059421, K23AG057760]; and the Robert Katzman Research Training Fellowship in Alzheimer's and Dementia Research cosponsored by the American Academy of Neurology, the American Brain Foundation, and the Alzheimer's Association.
Disclosure
The authors report no disclosures relevant to the manuscript. Go to Neurology.org/N for full disclosures.
References
- 1.Valtorta NK, Kanaan M, Gilbody S, Ronzi S, Hanratty B. Loneliness and social isolation as risk factors for coronary heart disease and stroke: systematic review and meta-analysis of longitudinal observational studies. Heart. 2016;102(13):1009-1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rico-Uribe LA, Caballero FF, Martin-Maria N, Cabello M, Ayuso-Mateos JL, Miret M. Association of loneliness with all-cause mortality: a meta-analysis. PLoS One. 2018;13:e0190033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Masi CM, Chen HY, Hawkley LC, Cacioppo JT. A meta-analysis of interventions to reduce loneliness. Pers Soc Psychol Rev. 2011;15(3):219-266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.McGinty EE, Presskreischer R, Han H, Barry CL. Psychological distress and loneliness reported by US adults in 2018 and April 2020. JAMA. 2020;324(1):93-94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Population and housing unit estimates. Accessed September 23. census.gov/programs-surveys/popest.html
- 6.Donovan NJ, Wu Q, Rentz DM, Sperling RA, Marshall GA, Glymour MM. Loneliness, depression and cognitive function in older U.S. adults. Int J Geriatr Psychiatry. 2017;32(5):564-573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Perissinotto CM, Stijacic Cenzer I, Covinsky KE. Loneliness in older persons: a predictor of functional decline and death. Arch Intern Med. 2012;172(14):1078-1083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Victor CR, Yang K. The prevalence of loneliness among adults: a case study of the United Kingdom. J Psychol. 2012;146:85-104. [DOI] [PubMed] [Google Scholar]
- 9.Wilson RS, Krueger KR, Arnold SE, et al. Loneliness and risk of Alzheimer disease. Arch Gen Psychiatry. 2007;64(2):234-240. [DOI] [PubMed] [Google Scholar]
- 10.Akhter-Khan SC, Tao Q, Ang TFA, et al. Associations of loneliness with risk of Alzheimer's disease dementia in the Framingham Heart Study. Alzheimers Dement. 2021;17(10):1619-1627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Penninkilampi R, Casey AN, Singh MF, Brodaty H. The association between social engagement, loneliness, and risk of dementia: a systematic review and meta-analysis. J Alzheimers Dis. 2018;66(4):1619-1633. [DOI] [PubMed] [Google Scholar]
- 12.Lara E, Martin-Maria N, De la Torre-Luque A, et al. Does loneliness contribute to mild cognitive impairment and dementia? A systematic review and meta-analysis of longitudinal studies. Ageing Res Rev. 2019;52:7-16. [DOI] [PubMed] [Google Scholar]
- 13.Sutin AR, Stephan Y, Luchetti M, Terracciano A. Loneliness and risk of dementia. J Gerontol B Psychol Sci Soc Sci. 2020;75(7):1414-1422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Donovan NJ, Okereke OI, Vannini P, et al. Association of higher cortical amyloid burden with loneliness in cognitively normal older adults. JAMA Psychiatry. 2016;73(12):1230-1237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.d'Oleire Uquillas F, Jacobs HIL, Biddle KD, et al. Regional tau pathology and loneliness in cognitively normal older adults. Transl Psychiatry. 2018;8:282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chêne G, Beiser A, Au R, et al. Gender and incidence of dementia in the Framingham Heart Study from mid-adult life. Alzheimers Dement. 2015;11(3):310-320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Monsell SE, Mock C, Hassenstab J, et al. Neuropsychological changes in asymptomatic persons with Alzheimer disease neuropathology. Neurology. 2014;83(5):434-440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kaur B, Himali JJ, Seshadri S, et al. Association between neuropathology and brain volume in the Framingham Heart Study. Alzheimer Dis Assoc Disord. 2014;28:219-225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Radloff LS. The CES-D scale a self-report depression scale for research in the general population. Appl Psychol Meas. 1977;1:385-401. [Google Scholar]
- 20.Cacioppo S, Capitanio JP, Cacioppo JT. Toward a neurology of loneliness. Psychol Bull. 2014;140(6):1464-1504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kumar A, Salinas J. The long-term public health impact of social distancing on brain health: topical review. Int J Environ Res Public Health. 2021;18(14):7307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Padmanabhan JL, Cooke D, Joutsa J, et al. A human depression circuit derived from focal brain lesions. Biol Psychiatry. 2019;86(10):749-758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Spreng RN, Dimas E, Mwilambwe-Tshilobo L, et al. The default network of the human brain is associated with perceived social isolation. Nat Commun. 2020;11(1):6393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gonzales MM, Samra J, O'Donnell A, et al. Association of midlife depressive symptoms with regional amyloid-beta and tau in the Framingham Heart Study. J Alzheimers Dis. 2021;82:249-260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Erzen E, Çikrikci Ö. The effect of loneliness on depression: a meta-analysis. Int J Soc Psychiatry. 2018;64(5):427-435. [DOI] [PubMed] [Google Scholar]
- 26.Satizabal CL, Beiser AS, Chouraki V, Chêne G, Dufouil C, Seshadri S. Incidence of dementia over three decades in the Framingham Heart Study. N Engl J Med. 2016;374(6):523-532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Folstein MF, Folstein SE, McHugh PR. Mini-Mental State: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189-198. [DOI] [PubMed] [Google Scholar]
- 28.American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-IV. American Psychiatric Association; 1994. [Google Scholar]
- 29.Weintraub S, Salmon D, Mercaldo N, et al. The Alzheimer's Disease Centers' Uniform Data Set (UDS): the neuropsychologic test battery. Alzheimer Dis Assoc Disord. 2009;23:91-101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wechsler D. WAIS-R : Wechsler Adult Intelligence Scale-Revised. Psychological Corp; 1981:1981. [Google Scholar]
- 31.Raz L, Knoefel J, Bhaskar K. The neuropathology and cerebrovascular mechanisms of dementia. J Cereb Blood Flow Metab. 2016;36(1):172-186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pase MP, Beiser A, Enserro D, et al. Association of ideal cardiovascular health with vascular brain injury and incident dementia. Stroke. 2016;47(5):1201-1206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Farias ST, Mungas D, Reed B, et al. Maximal brain size remains an important predictor of cognition in old age, independent of current brain pathology. Neurobiol Aging. 2012;33(8):1758-1768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Albert M, Massaro J, DeCarli C, et al. Profiles by sex of brain MRI and cognitive function in the Framingham Offspring Study. Alzheimer Dis Assoc Disord. 2010;24:190-193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fletcher E, Singh B, Harvey D, Carmichael O, DeCarli C. Adaptive image segmentation for robust measurement of longitudinal brain tissue change. Annu Int Conf IEEE Eng Med Biol Soc. 2012;2012:5319-5322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Carleton RN, Thibodeau MA, Teale MJ, et al. The Center for Epidemiologic Studies Depression Scale: a review with a theoretical and empirical examination of item content and factor structure. PLoS One. 2013;8(3):e58067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vilagut G, Forero CG, Barbaglia G, Alonso J. Screening for depression in the general population with the Center for Epidemiologic Studies Depression (CES-D): a systematic review with meta-analysis. PLoS One. 2016;11(5):e0155431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Loucks EB, Sullivan LM, D'Agostino RB, Larson MG, Berkman LF, Benjamin EJ. Social networks and inflammatory markers in the Framingham heart study. J Biosoc Sci. 2006;38(6):835-842. [DOI] [PubMed] [Google Scholar]
- 39.Salinas J, Beiser A, Himali JJ, et al. Associations between social relationship measures, serum brain-derived neurotrophic factor, and risk of stroke and dementia. Alzheimers Dement. 2017;3:229-237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ware EB, Faul JD, Mitchell CM, Bakulski KM. Considering the APOE locus in Alzheimer's disease polygenic scores in the Health and Retirement Study: a longitudinal panel study. BMC Med Genomics. 2020;13(1):164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Welty FK, Lahoz C, Tucker KL, Ordovas JM, Wilson PW, Schaefer EJ. Frequency of ApoB and ApoE gene mutations as causes of hypobetalipoproteinemia in the Framingham Offspring population. Arterioscler Thromb Vasc Biol. 1998;18(11):1745-1751. [DOI] [PubMed] [Google Scholar]
- 42.Salinas J, O'Donnell A, Kojis DJ, et al. Association of social support with brain volume and cognition. JAMA Netw Open. 2021;4(8):e2121122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dykstra PA. Older adult loneliness: myths and realities. Eur J Ageing. 2009;6(2):91-100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ruxton GD, Beauchamp G. Time for some a priori thinking about post hoc testing. J Behav Ecol. 2008;19:690-693. [Google Scholar]
- 45.Nishtala A, Piers RJ, Himali JJ, et al. Atrial fibrillation and cognitive decline in the Framingham heart study. Heart Rhythm. 2018;15:166-172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Theeke LA. Predictors of loneliness in U.S. adults over age sixty-five. Arch Psychiatr Nurs. 2009;23(5):387-396. [DOI] [PubMed] [Google Scholar]
- 47.Cudjoe TKM, Roth DL, Szanton SL, Wolff JL, Boyd CM, Thorpe RJ. The epidemiology of social isolation: National Health and Aging Trends study. J Gerontol B Psychol Sci Soc Sci. 2020;75(1):107-113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hayley S, Hakim AM, Albert PR. Depression, dementia and immune dysregulation. Brain. 2021;144(3):746-760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Nelson PT, Trojanowski JQ, Abner EL, et al. New old pathologies: AD, PART, and cerebral age-related TDP-43 with sclerosis (CARTS). J Neuropathol Exp Neurol. 2016;75:482-498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rajan KB, Barnes LL, Wilson RS, et al. Racial differences in the association between apolipoprotein E risk alleles and overall and total cardiovascular mortality over 18 years. J Am Geriatr Soc. 2017;65(11):2425-2430. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The FS datasets analyzed for the present study are available through formal data-use agreements. Any investigator may access the data through the process outlined on the FS website (framinghamheartstudy.org).