Key Points
Among patients who were eligible for a surgical fistula, 63% were suitable for endoAVF.
Older patients were less likely to have a vascular anatomy suitable for an endoAVF.
Patients with a vascular anatomy suitable for an endoAVF were similar in sex, race, DM, HTN, CAD, and peripheral artery disease.
Keywords: dialysis, arteriovenous fistula, AVF, dialysis access, endoAVF, feasibility studies, venous mapping
Visual Abstract
Abstract
Background
The first endovascular arteriovenous fistula (endoAVF) device (WavelinQ), a novel percutaneous technique of AVF creation, was approved by the Food and Drug Administration in 2018 and has been placed in a small number of United States patients on hemodialysis. It is unknown how often patients with advanced CKD have vascular anatomy suitable for WavelinQ creation. The goal of this study was to determine the proportion of patients with vascular anatomy suitable for WavelinQ creation and to assess patient characteristics associated with such suitability.
Methods
All patients referred for vascular access placement at a large academic medical center underwent standardized preoperative sonographic vascular mapping to assess suitability for an AVF. During a 2-year period (March 2019 to March 2021), we assessed the suitability of the vessels for creation of WavelinQ. We then compared the demographic characteristics, comorbidities, and vascular mapping measurements between patients who were or were not suitable for WavelinQ.
Results
During the study period, 437 patients underwent vessel mapping. Of these, 51% of patients were eligible for a surgical AVF, and 32% were eligible for a WavelinQ AVF; 63% of those suitable for a surgical AVF were also suitable for a WavelinQ AVF. Patients with a vascular anatomy suitable for WavelinQ were younger (age 55±15 versus 60±14 years, P=0.01) but similar in sex, race, diabetes, hypertension, coronary artery disease, and peripheral artery disease.
Conclusions
Among patients with CKD with vascular anatomy suitable for a surgical AVF, 63% are also suitable for a WavelinQ endoAVF. Older patients are less frequently suitable for WavelinQ.
Introduction
An arteriovenous fistula (AVF), the preferred type of vascular access for hemodialysis, is created by a direct surgical anastomosis between a peripheral artery and vein. To be eligible for a surgically created AVF, several preoperative vascular characteristics are used by many centers: an arterial diameter ≥2.0 mm, a venous diameter ≥2.5 mm, and absence of stenosis or thrombosis of the feeding artery and candidate draining vein (1–3). A preoperative vessel mapping is frequently obtained prior to AVF creation to ascertain whether these criteria are fulfilled. Specifically, patients may not be eligible for an AVF if the intended vein diameter is too small or if the draining vein has been damaged by prior cannulation.
Even when an AVF is created in a patient meeting these vascular parameters, it may fail to mature adequately for successful dialysis. One common cause of AVF nonmaturation is the development of a flow-limiting juxta-anastomotic stenosis (4,5). The stenosis is, in part, a consequence of aggressive neointimal hyperplasia, which may arise from surgical vascular injury or abnormal shear stress near the AVF anastomosis (6). Surgically created AVFs typically have a 30°–45° angle at the anastomosis. Computational fluid dynamics suggest that an anastomotic angle <30° improves the flow hemodynamics, and pilot clinical data demonstrated that a smaller surgical anastomotic angle reduced juxta-anastomotic stenosis (7,8). As a consequence, there has been great interest in novel AVF technologies that limit vascular injury, improve flow dynamics, and may theoretically lead to maturation rates superior to those of surgically created AVFs.
Endovascular arteriovenous fistula (endoAVF) is a novel percutaneous technique of AVF creation, with several devices recently approved for use in the United States by the Food and Drug Administration (FDA) (9,10). One of these devices, WavelinQ, is used at our medical center. In contrast to the surgical technique, WavelinQ potentially minimizes vascular injury at the time of AVF creation by using a minimally invasive technique. Moreover, by creating a side to side anastomosis between the artery and vein, it improves the flow hemodynamics near the anastomosis. Finally, it provides a dual outflow from the artery, enabling maturation of both the cephalic and basilic veins. These three features of the WavelinQ provide a potential rationale for its superior maturation, as compared with a surgical AVF.
Creation of WavelinQ requires two additional anatomic features beyond those required for surgical AVF creation. First, there must be an ulnar artery and ulnar vein of adequate diameter in close proximity to each other (within 2 mm). Second, there must be a patent and adequately sized perforator vein allowing drainage from the deep ulnar vein into the superficial veins in the upper arm. If the ulnar artery or vein is too small, an alternative approach entails creating an anastomosis between the radial artery and vein in the proximal forearm.
Publications on the WavelinQ device to date have consisted of small series and have not systematically determined how often patients with advanced CKD have the anatomy suitable for AVF creation (9–14). This study obtained routine preoperative vascular mapping in all patients referred for vascular access surgery at a single large dialysis center over a 2-year period, determined the proportion of patients who were suitable for WavelinQ, and compared clinical features between patients with or without anatomy suitable for WavelinQ. The importance of this study is to assess the proportion of patients suitable for WavelinQ and to identify patient characteristics that may limit the use of this type of endoAVF. It can also identify the type of patients in whom a surgically created AVF is preferred.
Materials and Methods
Preoperative Vascular Mapping Protocol for Surgical AVFs
All patients referred for vascular access placement at the University of Alabama at Birmingham (UAB) first underwent a standardized preoperative sonographic vascular mapping. The standard preoperative ultrasound for surgically created AVFs assesses the diameter of the superficial veins (cephalic and basilic) and the largest brachial vein, as well as assessing the potential inflow segments of the brachial and radial arteries. To be eligible for a surgically created AVF, our institutional protocol required an arterial diameter ≥2.0 mm, a venous diameter ≥2.5 mm, and exclusion of stenosis or thrombosis of the feeding artery and candidate draining vein (1–3).
All ultrasound measurements were performed by experienced sonographers in an American College of Radiology–accredited ultrasound department using a tourniquet. The nondominant upper extremity was first evaluated. If no criteria met for AVF was found, then the other extremity was evaluated. Duplex ultrasound mapping of the upper extremity with tourniquet included the basilic and cephalic vein diameters and depths at prescribed locations in the cranial and midupper arm, antecubital area, midforearm, and wrist. The presence of high brachial artery takeoff was also reported, if present (15).
Anatomic Prerequisites for an EndoAVF
One of the endoAVF technologies, the WavelinQ EndoAVF system (Bard Peripheral Vascular, Tempe, AZ), uses a percutaneous technique to create an anastomosis between the ulnar or radial artery and its adjacent vein in the proximal forearm (16). Briefly, arterial and deep venous magnetic catheters are placed under fluoroscopy. The magnets are then activated in both catheters to align the artery and vein. A spring-loaded radiofrequency electrode is released from the venous catheter and energized for 2 seconds, creating a channel between the vein and the artery (Figure 1). Thus, preoperative mapping for an endoAVF requires all of the vascular measurements obtained for a surgically created AVFs as well as two additional measurements of the ulnar artery, ulnar vein, and perforating vein. First, there must be an ulnar artery and an ulnar vein of adequate diameter in close proximity to each other (within 2 mm). Second, there must be an adequately sized (≥2 mm) patent perforator vein allowing drainage from the deep ulnar vein into the superficial venous veins in the upper arm. If the ulnar artery or vein is too small, an alternative approach entails creating an anastomosis between the radial artery and vein in the proximal forearm (Figure 2).
Figure 1.
Endovascular arteriovenous fistula (endoAVF) creation. (A) The precreation venogram shows a direct perforator (yellow arrow) arising from the lateral ulnar vein (white arrow) and draining into the cephalic vein (green arrow). (B) The precreation angiogram shows opacification of the radial (blue arrow), ulnar (red arrow), and interosseous (green arrow) arteries without stenosis. The lateral ulnar vein and ulnar artery were selected as target creation vessels. (C) Arterial and venous catheters were advanced to the desired position on the basis of precreation venogram/angiogram. Note that the arc of the electrode (green arrow) is congruent with the concave surface of the arterial backstop. (D) The fistulogram shows successful endoAVF creation (white arrow) and coil embolization of the brachial vein (blue arrow) to redirect the flow to the cephalic vein and support maturation of the cannulation area.
Figure 2.
Ultrasound vascular mapping showing suitable mapping for endoAVF. (A) Ulnar artery (UA) and ulnar veins (UVs) 2-cm caudal to the brachial artery bifurcation are shown. The UA diameter is 0.48 cm. (B) Radial artery (RA) and radial veins (RVs) 2 cm caudal to the brachial artery bifurcation are shown. The RA diameter is 0.43 cm. RV diameters are 0.37/0.44 cm. (C) Deep perforator. The diameter is 0.24 cm between the cephalic vein and the UV. (Note that the "Dist" annotations on the images are inner diameters.)
Data Collection
Two full-time access coordinators scheduled all vascular access procedures performed by surgeons, radiologists, or nephrologists and maintained a prospective, computerized database of these procedures (17). The data collection included all patients undergoing preoperative vascular mapping during a 2-year period. The UAB Institutional Review Board approved review of the electronic medical records for research and provided a waiver from obtaining informed consent. The focus of this study was to quantify the proportion of patients who were anatomically eligible for WavelinQ. In addition, each patient’s electronic medical record was reviewed to extract demographic and clinical information.
Statistical Analyses
Initially, we ascertained the proportion of patients with a preoperative vascular anatomy suitable for a surgically created AVF who were also suitable for WavelinQ. We then compared the demographic, clinical, and vascular properties of patients who were suitable for WavelinQ with those who were not suitable. Baseline features of the patients were compared by chi-squared test for categorical variables and by t tests or nonparametric statistics for continuous variables. P=0.05 was considered statistically significant.
Results
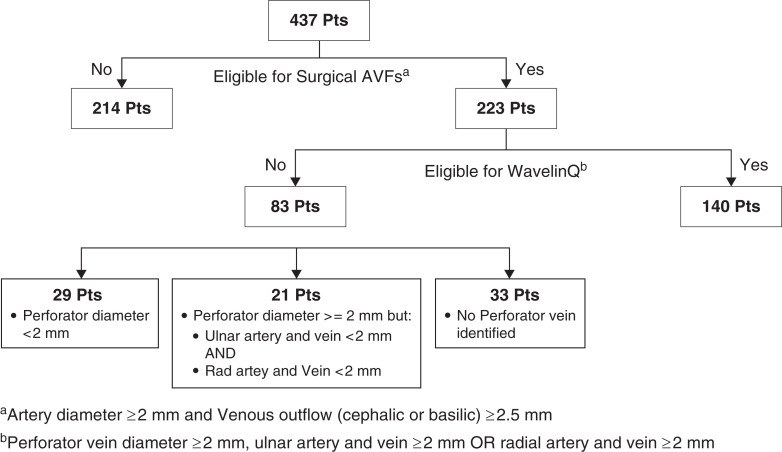
We identified 437 patients who underwent preoperative vascular mapping from March 2019 to March 2021 at UAB. Of these, we excluded 214 patients whose anatomy precluded creation of a surgical AVF. The remaining 223 patients were considered eligible for a surgical AVF (and 95% of them were receiving their first access in that extremity). Of this cohort, 140 (or 63%) were also suitable for endoAVF creation using the WavelinQ system. In other words, of all 437 patients referred for a vascular access, 140 (or 32%) were suitable for WavelinQ. The proportions of patients eligible for a surgical AVF who were also suitable for WavelinQ were very similar for the subset of patients who had ESKD (115 of 182 or 63%) and the subset of patients who were pre-ESKD (25 of 41 or 61%). Among the 83 patients who were not suitable for WavelinQ, 33 were due to the absence of a perforator vein, 29 were because the perforator vein was smaller than the minimal threshold, and 21 were because the ulnar and radial artery or vein was below the desired threshold (Figure 3).
Figure 3.
Schematic presentation of patients (Pts) who underwent preoperative vascular mapping between March 2019 and March 2021. AVF, arteriovenous fistula.
As compared with patients unsuitable for a surgical AVF, those who were suitable for a surgical AVF were less likely to be Black but did not differ in other demographic or clinical characteristics. Among the subset of patients suitable for a surgical AVF, those suitable for WavelinQ were younger than those not suitable for this access type (Table 1). The two groups were similar in terms of diabetes, hypertension, and cardiovascular comorbidities as well as laterality of the arm and presence of high radial artery takeoff. Most of patients (80%) had already initiated hemodialysis prior to undergoing preoperative vascular mapping.
Table 1.
Patients with preoperative vascular mapping suitable for a surgical arteriovenous fistula and for WavelinQ endovascular arteriovenous fistula
| Study Patients | Surgical Arteriovenous Fistula Eligibility in Patients Undergoing Vascular Mapping, n=437 | WavelinQ Endovascular Arteriovenous Fistula Eligibility in Patients Suitable for a Surgical Arteriovenous Fistula, n=223 | ||||
|---|---|---|---|---|---|---|
| Yes | No | P Value | Yes | No | P Value | |
| N of patients, % | 223 (51) | 214 (49) | 140 (63) | 83 (37) | ||
| Age, yr, mean±SD | 55±15 | 57±15 | 0.23 | 55±15 | 60±14 | 0.01 |
| Sex | 0.17 | 0.77 | ||||
| Men | 121 (54) | 102 (48) | 77 (55) | 44 (53) | ||
| Women | 102 (46) | 112 (52) | 63 (45) | 39 (47) | ||
| Racea | 0.002 | 0.13 | ||||
| Black | 150 (68) | 168 (79) | 90 (64) | 60 (74) | ||
| White | 71 (32) | 39 (21) | 50 (36) | 21 (26) | ||
| Diabetes | 118 (53) | 115 (54) | 0.86 | 71 (51) | 47 (57) | 0.39 |
| Hypertension | 210 (94) | 197 (92) | 0.38 | 130 (92) | 80 (96) | 0.28 |
| Coronary artery disease | 45 (20) | 42 (20) | 0.89 | 28 (20) | 17 (20) | 0.93 |
| Peripheral vascular disease | 15 (7) | 17 (8) | 0.63 | 8 (6) | 7 (8) | 0.43 |
| ESKD | 182 (82) | 187 (87) | 0.10 | 115 (82) | 67 (81) | 0.79 |
| AVF location, left arm | — | — | — | 98 (70) | 60 (72) | 0.72 |
| High radial takeoff | — | — | — | 18 (13) | 14 (17) | 0.41 |
AVF, arteriovenous fistula; —, no data.
Information is missing for nine patients.
Because WavelinQ suitability differed by patient age, we subsequently compared the individual characteristics of the deep venous system between older (age ≥60 years) and younger (age <60 years) patients (Table 2). Absence of a perforator vein was similar in both age groups, but older patients were more likely to have a perforator vein smaller than the 2-mm threshold (38% versus 20%, P=0.004). Among those patients with a perforator vein diameter below the minimum threshold, similar proportions in both age groups had ulnar and/or radial vessels below the minimum threshold.
Table 2.
Comparison of deep vein characteristics in older and younger patients evaluated for a WavelinQ endovascular arteriovenous fistula (n=223)
| Patients Deep Vein Characteristics | Age <60 yr, N=128 | Age >60 yr, N=95 | P Value |
|---|---|---|---|
| Absent perforator vein, n (%) | 16 (12) | 17 (18) | 0.26 |
| Perforator vein <2 mm, n (%) | 26 (20) | 36 (38) | 0.004 |
| Perforator vein ≥2 mm but radial or ulnar artery or vein <2 mm, n (%) | 14 (11) | 7 (7) | 0.34 |
| Unsuitable for WavelinQ for any reason, n (%) | 56 (44) | 60 (63) | 0.004 |
Discussion
This study focused on the anatomic suitability of vessels for the creation of WavelinQ. In our practice, patients referred for access surgery routinely undergo preoperative vascular mapping as the standard of care. There is a clinical preference for placing an AVF as the initial access when the vascular anatomy is suitable. After the WavelinQ device was approved by FDA in 2018, we modified our vascular mapping protocol to screen for eligibility for WavelinQ by adding measurements of the perforator vein (presence and diameter) as well as antecubital fossa measurements of the radial and ulnar veins and arteries. Of 223 patients who were eligible for a surgical fistula (minimum artery diameter of 2 mm and presence of a cephalic or basilic vein with minimum diameter of 2.5 mm), 63% were eligible for WavelinQ. Older patients were less likely to have a vascular anatomy suitable for WavelinQ, with the major limiting factor being a perforator vein smaller than the minimum diameter threshold of 2 mm.
Little has been published previously about the suitability of patients with ESKD or advanced CKD for an endoAVF. In the pilot Novel Endovascular Access Trial study that led to the FDA approval of the WavelinQ system, preoperative vascular mapping was not specifically addressed other than a vague statement that screening failure occurred in 25% of patients due to small target vessel size, without specific mention about the presence or diameter of the perforator vein (16). For a different type of endoAVF, the Elipsys EL system, the screening failure rate was reported as 28% (18). A recent study examined the anatomic suitability for both endoAVF types, and the percentage of patients anatomically suitable for endoAVF was slightly lower than the results of this study (63%). The former study was limited by measuring venous diameters without a tourniquet. In addition, it was performed at a Veteran Administration hospital, in which the patients were exclusively men and relatively old (19).
The major strengths of this study include assessment of anatomic suitability for WavelinQ using a standardized ultrasound technique, a multidisciplinary approach to optimize vascular access outcomes in a large academic center with a diverse patient population, reliance on a small number of highly experienced sonographers and radiologists, and the use of a prospective computerized database to optimize capture of data associated with vascular access care.
Our study also has some limitations. First, the patients were derived from a single large dialysis center, and the results may not generalize to some dialysis centers. Second, we lacked information on the dialysis vintage or number of previous arteriovenous access creations in the study population. Finally, this study focused on the technical feasibility of WavelinQ creation on the basis of meeting the minimal vascular diameters. It does not, however, answer the question about how often WavelinQ actually achieves clinical maturation (i.e., suitability for dialysis use). We know from published literature that a substantial proportion of surgically created AVFs fails to mature and that outcomes vary according to patient demographics or comorbidities. Future studies are required to determine how often endoAVFs fail to mature and what factors are associated with their success.
In a large academic center, 63% of patients whose vascular anatomy was suitable for creation of a surgical fistula were suitable for WavelinQ creation. Older patients were less likely to be suitable for WavelinQ, primarily due to the presence of small perforator veins. Thus, a surgical AVF may be preferred in such patients.
Disclosures
M. Allon reports consultancy agreements with CorMedix and scientific advisor or membership with the Kidney360 Editorial Board as Editor-in-Chief. M.L. Robbin reports research funding from Philips Medical and scientific advisor or membership with the Journal of Ultrasound in Medicine Editorial Board, the Radiology Editorial Board, and the Ultrasound Quarterly Editorial Board. R. Varma reports speakers bureau for Becton, Dickinson and Company. All remaining authors have nothing to disclose.
Funding
This work was supported by National Institute of Diabetes and Digestive and Kidney Diseases grant K23DK123319 (to A. Al-Balas) and National Institute on Minority Health and Health Disparities grant R01 MD013818 (to M. Allon).
Footnotes
See related editorial, “Vascular Suitability for an Endovascular AVF: Getting Beyond the Velvet Rope,” on pages 201–203.
Author Contributions
A.K. Abdel Aal, A. Al-Balas, and M. Allon conceptualized the study; A.K. Abdel Aal and A. Al-Balas were responsible for data curation; A. Al-Balas and H. Al-Balas were responsible for formal analysis; A.K. Abdel Aal and A. Al-Balas were responsible for methodology; H. Al-Balas and A. Almehmi were responsible for project administration; K. Sharbidre and R. Varma were responsible for resources; M. Allon, M.L. Robbin, and K. Sharbidre were responsible for validation; M. Allon was responsible for visualization; H. Al-Balas, M. Allon, A. Almehmi, and R. Varma provided supervision; A. Al-Balas wrote the original draft; and A.K. Abdel Aal, A. Al-Balas, H. Al-Balas, M. Allon, A. Almehmi, M.L. Robbin, K. Sharbidre, and R. Varma reviewed and edited the manuscript.
References
- 1.Robbin ML, Gallichio MH, Deierhoi MH, Young CJ, Weber TM, Allon M: US vascular mapping before hemodialysis access placement. Radiology 217: 83–88, 2000. 10.1148/radiology.217.1.r00oc2883 [DOI] [PubMed] [Google Scholar]
- 2.Allon M, Lockhart ME, Lilly RZ, Gallichio MH, Young CJ, Barker J, Deierhoi MH, Robbin ML: Effect of preoperative sonographic mapping on vascular access outcomes in hemodialysis patients. Kidney Int 60: 2013–2020, 2001. 10.1046/j.1523-1755.2001.00031.x [DOI] [PubMed] [Google Scholar]
- 3.Allon M, Robbin ML: Increasing arteriovenous fistulas in hemodialysis patients: Problems and solutions. Kidney Int 62: 1109–1124, 2002. 10.1111/j.1523-1755.2002.kid551.x [DOI] [PubMed] [Google Scholar]
- 4.Allon M, Robbin ML, Young CJ, Deierhoi MH, Goodman J, Hanaway M, Lockhart ME, Litovsky S: Preoperative venous intimal hyperplasia, postoperative arteriovenous fistula stenosis, and clinical fistula outcomes. Clin J Am Soc Nephrol 8: 1750–1755, 2013. 10.2215/CJN.02740313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cheung AK, Imrey PB, Alpers CE, Robbin ML, Radeva M, Larive B, Shiu YT, Allon M, Dember LM, Greene T, Himmelfarb J, Roy-Chaudhury P, Terry CM, Vazquez MA, Kusek JW, Feldman HI; Hemodialysis Fistula Maturation Study Group : Intimal hyperplasia, stenosis, and arteriovenous fistula maturation failure in the Hemodialysis Fistula Maturation Study. J Am Soc Nephrol 28: 3005–3013, 2017. 10.1681/ASN.2016121355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ene-Iordache B, Remuzzi A: Disturbed flow in radial-cephalic arteriovenous fistulae for haemodialysis: Low and oscillating shear stress locates the sites of stenosis. Nephrol Dial Transplant 27: 358–368, 2012. 10.1093/ndt/gfr342 [DOI] [PubMed] [Google Scholar]
- 7.Ene-Iordache B, Cattaneo L, Dubini G, Remuzzi A: Effect of anastomosis angle on the localization of disturbed flow in ‘side-to-end’ fistulae for haemodialysis access. Nephrol Dial Transplant 28: 997–1005, 2013. 10.1093/ndt/gfs298 [DOI] [PubMed] [Google Scholar]
- 8.Darcy M, Vachharajani N, Zhang T, Mani N, Kim SK, Matson S, Wood M, Shenoy S: Long-term outcome of upper extremity arteriovenous fistula using pSLOT: Single-center longitudinal follow-up using a protocol-based approach. J Vasc Access 18: 515–521, 2017. 10.5301/jva.5000764 [DOI] [PubMed] [Google Scholar]
- 9.Rajan DK, Ebner A, Desai SB, Rios JM, Cohn WE: Percutaneous creation of an arteriovenous fistula for hemodialysis access. J Vasc Interv Radiol 26: 484–490, 2015. 10.1016/j.jvir.2014.12.018 [DOI] [PubMed] [Google Scholar]
- 10.Hull JE, Jennings WC, Cooper RI, Waheed U, Schaefer ME, Narayan R: The pivotal multicenter trial of ultrasound-guided percutaneous arteriovenous fistula creation for hemodialysis access. J Vasc Interv Radiol 29: 149–158.e5, 2018. 10.1016/j.jvir.2017.10.015 [DOI] [PubMed] [Google Scholar]
- 11.Yan Wee IJ, Yap HY, Tang TY, Chong TT: A systematic review, meta-analysis, and meta-regression of the efficacy and safety of endovascular arteriovenous fistula creation. J Vasc Surg 71: 309–317.e5, 2020. 10.1016/j.jvs.2019.07.057 [DOI] [PubMed] [Google Scholar]
- 12.Berland TL, Clement J, Griffin J, Westin GG, Ebner A: Endovascular creation of arteriovenous fistulae for hemodialysis access with a 4 Fr device: Clinical experience from the EASE study. Ann Vasc Surg 60: 182–192, 2019. 10.1016/j.avsg.2019.02.023 [DOI] [PubMed] [Google Scholar]
- 13.Radosa CG, Radosa JC, Weiss N, Schmidt C, Werth S, Hofmockel T, Plodeck V, Gatzweiler C, Laniado M, Hoffmann RT: Endovascular creation of an arteriovenous fistula (endoAVF) for hemodialysis access: First results. Cardiovasc Intervent Radiol 40: 1545–1551, 2017. 10.1007/s00270-017-1750-x [DOI] [PubMed] [Google Scholar]
- 14.Shahverdyan R, Beathard G, Mushtaq N, Litchfield TF, Nelson PR, Jennings WC: Comparison of outcomes of percutaneous arteriovenous fistulae creation by Ellipsys and WavelinQ devices. J Vasc Interv Radiol 31: 1365–1372, 2020. 10.1016/j.jvir.2020.06.008 [DOI] [PubMed] [Google Scholar]
- 15.Pietryga JA, Little MD, Robbin ML: Sonography of arteriovenous fistulas and grafts. Semin Dial 30: 309–318, 2017. 10.1111/sdi.12599 [DOI] [PubMed] [Google Scholar]
- 16.Lok CE, Rajan DK, Clement J, Kiaii M, Sidhu R, Thomson K, Buldo G, Dipchand C, Moist L, Sasal J; NEAT Investigators : Endovascular proximal forearm arteriovenous fistula for hemodialysis access: Results of the prospective, multicenter Novel Endovascular Access Trial (NEAT). Am J Kidney Dis 70: 486–497, 2017. 10.1053/j.ajkd.2017.03.026 [DOI] [PubMed] [Google Scholar]
- 17.Allon M, Bailey R, Ballard R, Deierhoi MH, Hamrick K, Oser R, Rhynes VK, Robbin ML, Saddekni S, Zeigler ST: A multidisciplinary approach to hemodialysis access: Prospective evaluation. Kidney Int 53: 473–479, 1998. 10.1046/j.1523-1755.1998.00761.x [DOI] [PubMed] [Google Scholar]
- 18.Mallios A, Jennings WC, Boura B, Costanzo A, Bourquelot P, Combes M: Early results of percutaneous arteriovenous fistula creation with the Ellipsys Vascular Access System. J Vasc Surg 68: 1150–1156, 2018. 10.1016/j.jvs.2018.01.036 [DOI] [PubMed] [Google Scholar]
- 19.Popli K, Dittman JM, Amendola MF, Plum J, Newton DH: Anatomic suitability for commercially available percutaneous arteriovenous fistula creation systems. J Vasc Surg 73: 999–1004, 2021. 10.1016/j.jvs.2020.09.021 [DOI] [PubMed] [Google Scholar]