ABSTRACT
Background
Patients receiving dialysis for end-stage kidney disease (ESKD) commonly co-exhibit risk factors for hepatic impairment. This systematic review and meta-analysis aimed to quantify the coexistence of chronic liver disease (CLD) and characterize risk factors and outcomes.
Methods
We searched the following databases from inception to May 2021: CINAHL, Cochrane Library, Embase, Kings Fund Library, MEDLINE and PubMed. The protocol was pre-registered on PROSPERO (study ID: CRD42020206486). Studies were assessed against three inclusion criteria: adults (>18 years) with ESKD receiving dialysis, primary outcome involving CLD prevalence and publications in English. Moderator analysis was performed for age, gender, study size and publication year. Sensitivity analysis was performed where applicable by removing outlier results and studies at high risk of bias.
Results
Searches yielded 7195 articles; of these 15 met the inclusion criteria. A total of 320 777 patients were included. The prevalence of cirrhosis and non-alcoholic fatty liver disease (NAFLD) was 5% and 55%, respectively. Individuals with CLD had 2-fold higher mortality than those without {odds ratio [OR] 2.19 [95% confidence interval (CI) 1.39–3.45]}. Hepatitis B [OR 13.47 (95% CI 1.37–132.55)] and hepatitis C [OR 7.05 (95% CI 4.00–12.45)], but not diabetes, conferred increased cirrhosis risk. All studies examining NAFLD were judged to be at high risk of bias. We found no data on non-alcoholic steatohepatitis (NASH). Deaths from CLD, cancer and infection were greater among cirrhotic patients.
Conclusions
CLD is prevalent in dialysis patients. Hepatitis B and C confer increased risk of CLD. The impact of NAFLD and NASH cirrhosis requires further study. CLD is associated with an increased risk of mortality in this setting.
Keywords: dialysis, ESRD, haemodialysis, peritoneal dialysis, systematic review
INTRODUCTION
End-stage kidney disease (ESKD) and chronic liver disease (CLD) share common cardiometabolic and pathogenic risk factors. For example, diabetes mellitus (DM) is one of the leading causes of ESKD worldwide [1] and, in addition to other metabolic risk factors, is a common finding in patients with non-alcoholic fatty liver disease (NAFLD). NAFLD, the most common liver disorder in the Western world, affects 17–46% of adults [2]. NAFLD progresses to non-alcoholic steatohepatitis (NASH) in ∼20% of cases [3]. NASH can lead to liver fibrosis and cirrhosis [4] and, with a increasing prevalence predicted over the next decade, is soon expected to overtake chronic infection with hepatitis B and C as the leading cause of cirrhosis worldwide [5–7]. Nevertheless, hepatitis B and C remain prevalent particularly within haemodialysis (HD) populations globally despite improvements in infection control, uptake of hepatitis B vaccines and the emergence of novel therapeutic options for hepatitis C [8–10].
The prevalence of combined advanced liver and kidney impairment is increasing, as evidenced by increasing numbers of simultaneous liver–kidney transplant referrals over the past 2 decades [11]. This is partly due to increased emphasis on kidney impairment in liver transplant scoring systems, but the increasing prevalence of liver and kidney disease in the context of DM and obesity is also a probable contributor.
Despite the increasing burden of combined liver and kidney dysfunction, relatively little attention has been paid to this patient group, despite high risks of haemodynamic instability, malnutrition, bleeding and infection [12]. The primary aim of this systematic review and meta-analysis was to summarize available evidence on the prevalence of CLD in patients treated with dialysis. It also assessed known aetiological factors, severity of CLD and clinical outcomes. This is the first systematic review and meta-analysis to evaluate CLD not limited to viral hepatitis and outcomes in dialysis patients.
MATERIALS AND METHODS
The systematic review and meta-analyses were performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines [13]. The methods were pre-registered on PROSPERO (study ID: CRD42020206486).
Search strategy and review process
The search strategy is available from https://www.crd.york.ac.uk/PROSPEROFILES/206486_STRATEGY_20201008.pdf. In brief, a combination of terms for ESKD, HD, peritoneal dialysis (PD), liver disease and outcomes was used. Articles were searched across the following databases from inception until 4 May 2021: CINAHL, Cochrane Library, Embase, Kings Fund Library, MEDLINE and PubMed. A total of 7181 records were retrieved with a further 14 identified through lateral searches. After removing duplicates, 5808 papers were screened by the first author against the following inclusion criteria: adults (>18 years) with ESKD treated with dialysis, primary outcome related to the prevalence of CLD (defined as the presence of hepatic inflammation, steatosis, fibrosis or cirrhosis) and publication in English.
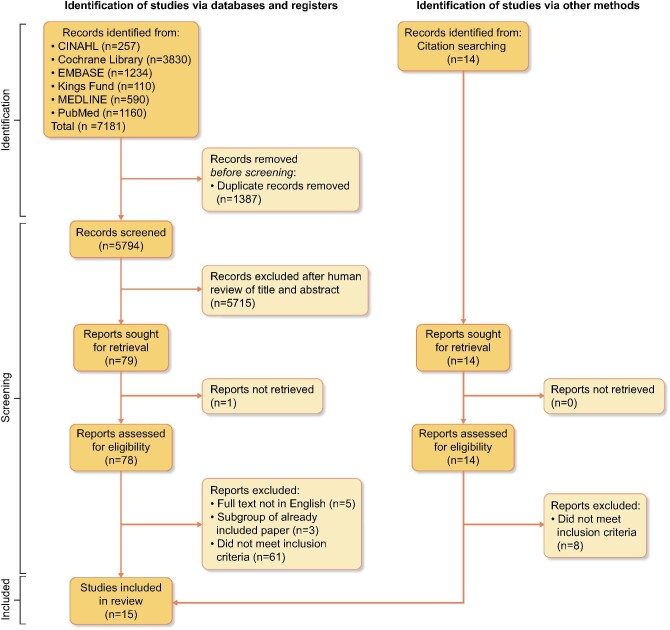
A total of 93 articles were selected for full-text review. Two authors (O.S. and S.R.) screened articles independently for consensus on inclusion. Discrepancies were resolved through discussion with members of the study team (S.S. and K.F.). A total of 15 studies met the inclusion criteria and were used in the analysis [14–28] (see Figure 1). Detailed reasons for exclusion were noted.
FIGURE 1:
Study selection flowchart.
Baseline study data summarizing study characteristics are outlined in Table 1. All data extracted are incorporated into this article and the online supplementary material.
Table 1.
Study and participant characteristics
| Patients included, n | Demographics | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Study | Country | Length of follow-up | Study period | Population source | Age (years) | Male (%) | Ethnicity | HD (%) | PD (%) | Study design | Inclusion criteria | Exclusion criteria | Method of liver disease diagnosis | ||
| Artru 2019 [14] | France | 2 years | 2000–2014 | All patients in Nephronor database, Nord Pas du Calais | Regional population | 7658 | 70.5 | 58.1 | Not described | 100 | 0 | Cohort study | >18 years, HIV negative, no previous transplant | None described | Histological, clinical or radiological findings |
| Chien 2012 [16] | Taiwan | 7.6 years (PD) 6.9 (HD) | 1999–2008 | Taiwan National Health Insurance Research Database | National population database | 12 054 | 59.5 | 46.6 | Not described | 93.7 | 6.3 | Cohort study | >18 years, >90 days on dialysis | Kidney transplant (prior to or during follow up), multiple modality switches | ICD-9 CM codes |
| Che-Yi 2016 [15] | China | 6 years | 2004–2013 | China Medical University Hospital | Single hospital population | 425a | 62.5 | 64.7 | Not described | 80 | 20 | Cohort study | >3 months on dialysis | Patients receiving HDF | ICD-9 CM codes |
| b | Taiwan | Up to 15 years | 1996–2011 | Longitudinal Health Insurance Database for Catastrophic Illness Patients of the Taiwan National Health Research Institute | National population database | 1395c | 60.3 | 62.1 | Not described | 80 | 20 | Cohort study | ESKD with liver cirrhosis | None described | |
| Deshpande 2019 [18] | USA | 1 year | 2005–2016 | 5% random sample of USA Medicare beneficiaries receiving inpatient or outpatient care | National population database | 291 663 | 67.3 | 55 | 55% White, 32.5% Black, 3.0% Asian, 5.8% Hispanic, 1.4% Native American | 100 | 0 | Cohort study | ESKD treated with dialysis with HCV infection/cirrhosis | None described | ICD-9 or ICD-10 CM codes |
| De Vecchi 2002 [17] | Italy | 3.2 years (CLD) 2.8 years (controls) | 1985–1999 | Nephrology Department, University of Milan | Single hospital population | 62d | 57.3 | 67.7 | Not described | 0 | 100 | Case control study | ESKD treated with PD | None described | Histological, clinical or radiological findings |
| Espinosa 2001 [19] | Spain | 8 years | 1992–1999 | 5 dialysis units, Cordoba | Regional population | 175 | 53.2 | 53.1 | Not described | 100 | 0 | Cohort study | Treatment with thrice weekly standard HD | <6 months on HD, HCV negative initially but then developed hepatitis, HbeAg positive | Histological, clinical or radiological findings |
| Huang 2011 [20] | Taiwan | 3.3 years (CLD) 2.6 years (controls) | 1984–2008 | Department of Nephrology, Taichung Veterans General Hospital | Single hospital population | 90e | 48.9 | 43.3 | Not described | 0 | 100 | Case control study | ESKD treated with PD | None described | Histological, clinical or radiological findings |
| Kim 2016 [21] | South Korea | 2.9 years (CLD) 3.2 years (controls) | 2000–2011 | Gachon University Gil Medical Center, Korea | Single hospital population | 1069 | 55 | 56 | Not described | 69.4 | 30.6 | Cohort study | ESKD treated with dialysis | <18 years, recovery of native kidney function, kidney transplant during follow-up period, incomplete medical records | Clinical or radiological findings |
| Lee 2017 [22] | South Korea | 4.8 years (CLD) 6.4 years (controls) | 2007–2014 | Dong-A University Hospital, Busan | Single hospital population | 295 | 59.1 | 59 | Not described | 0 | 100 | Case control study | >18 years, PD catheter in situ | HD catheter sited prior to treatment with PD | Histological, clinical or radiological findings |
| Marcelli 1996 [23] | USA | Up to 5 years | 1986–1991 | US Renal Data System | National population database | 2900 | 59.9 | 53.7 | White only | 78.8 | 21.2 | Cohort study | White patients starting dialysis | Patients who died within 30 days of starting RRT | USRDS database |
| Italy | Up to 5 years | 1986–1991 | Lombardi Dialysis and Transplant Registry | Regional population | 1296 | 55.9 | 62.1 | White only | 69.3 | 30.7 | Cohort study | White patients starting dialysis | Patients who died within 30 days of starting RRT | RLDT database | |
| Marcus 1992 [24] | USA | 2.1 years | 1980–1990 | Division of Nephrology, University of Michigan | Single hospital population | 9e | Not described | Not described | Not described | 0 | 100 | Case series | Chronic liver disease and ascites at time of starting PD | None described | Histological, clinical or radiological findings |
| Mikolasevic 2015 [25] | Croatia | 1.5 years | Not specified | Department of Nephrology, Dialysis and Kidney Transplantation, Rijeka | Single hospital population | 94 | 69.1 | 54.3 | Not described | 100 | 0 | Cohort study | Treatment with thrice weekly HD 4–5 h per session | Active HBV/HCV infection, alcohol abuse, other liver diseases, hepatotoxic medications, failed transient elastography, AKI or metastatic cancer, kidney transplant during follow-up period | Transient elastography |
| Stolic 2016 [26] | Serbia | Cross-sectional study | Not specified | HD patients at Clinical Center Kragujevac | Single hospital population | 72 | All > 65 | 56.9 | Not described | 100 | 0 | Cross-sectional study | >65 years, ESKD treated with HD, previous liver USS imaging | <3 months on HD, hospitalization within last 6 months, infection with hepatotrophic virus, DM, previous HPB surgery, BMI >30, statin/glucocorticoid therapy | USS comparison of echogenicity of right kidney/spleen with liver |
| Nakayama 2000 [28] | Japan | 6 years | 1993–1999 | 16 dialysis centres mostly in Tokyo | Regional population | 1470 | 55.4 | 60.5 | Not described | 100 | 0 | Cohort study | ESKD treated with HD | None described | Clinical or radiological findings |
| Behairy 2021 [27] | Egypt | Cross-sectional study | 2018–2019 | 2 dialysis centres in Egypt | Regional population | 50 | 48.6 | 60 | Not described | 100 | 0 | Cross-sectional study | >18 years, thrice weekly HD > 6 months, 4 h per session | HBV/HCV/HIV positive, decompensated or known liver disease, alcohol intake, diabetes mellitus, BMI >30, use of drugs known to induce liver steatosis | Transient elastography |
A total of 425 patients included in final analyses however prevalence data based on data from 9975 patients prior to propensity matching.
Where data from Chien et al. [16] are also analysed, these data have not been included due to risk of double counting.
A total of 1395 patients included in final analyses, however group selected from 5365 patients prior to propensity matching.
A total of 41 controls and 21 cirrhotic PD patients included in the final analysis from 381 patients prior to matching.
A total of 60 controls and 30 cirrhotic PD patients included in the final analysis from 728 prior to matching.
A total of 9 patients included in the final analysis with prevalence data calculated from information about the total number of records reviewed (n = 465).
Outcome measures
The primary clinical outcome measure of interest was the prevalence of CLD among patients treated with dialysis. Other secondary outcome measures included differences in mortality rates of patients treated with dialysis both with and without coexistent CLD and differences in mortality from cardiovascular disease, infection, malignancy and liver disease. Other prespecified measurements of interest included rates of comorbidities associated with an increased risk of CLD (DM, obesity and viral hepatitis), the proportion of patients with systemic inflammation, anaemia and depression and the underlying aetiology and severity of liver disease.
Risk of bias assessment
Risk of bias assessment was performed by two authors (O.S. and H.U.) independently using the Newcastle–Ottawa Scale (NOS) as recommended by Cochrane [29, 30]. This tool includes assessment of the quality of patient selection, comparability and exposure. Results were then discussed and discrepancies were resolved by consensus.
Statistical methods
Data analysis was undertaken using Review Manager 5 software, version 5.4 (Cochrane Collaboration, London, UK) and Meta-Essentials: workbooks for meta-analysis, version 1.5 (Erasmus Research Institute of Management, Rotterdam, The Netherlands) [31]. All relevant data from individual studies were extracted and pooled manually. A random effects model was applied to the data undergoing meta-analysis in order to account for heterogeneity and the impact of differing sample sizes within the populations of interest [32]. Data pertaining to clinical outcomes were handled using odds ratios (ORs) with 95% confidence intervals (CIs). Statistical heterogeneity between combined studies was measured using the I2 test, which defines the percentage of total variation across studies due to heterogeneity rather than chance [33]. Levels of heterogeneity are typically defined as follows: low (<25%), moderate (25–75%) and high (>75%). Moderator analysis was performed for age, gender, study size and year of publication only, due to insufficient data on other shared risk factors. Sensitivity analyses were performed where applicable by removing outlier results and studies at high risk of bias to assess the robustness of synthesized results.
RESULTS
Study and patient characteristics
This meta-analysis selected 15 studies published between 1992 and 2021 (Figure 1). Baseline study characteristics are outlined in Table 1. Six studies were performed in East Asia [15, 16, 20–22, 28], five in Europe [14, 17, 19, 25, 26], two in the USA [18, 24] and one in North Africa [27]. One study was multinational [23]. Follow-up ranged from 1 to 15 years (two studies were cross-sectional). The number of included patients was 320 777. The mean age of participants ranged from 49 to 71 years. The proportion of male participants ranged from 43 to 68%. Only two studies [18, 23] described participant ethnicity. Seven studies [14, 18, 19, 25–28], including the largest study involving 291 663 patients drawn from a 5% random sample of US Medicare beneficiaries [18], involved HD patients only; four [17, 20, 22, 24] involved (PD) patients only. The remaining four studies [15, 16, 21, 23] involved both PD and HD patients. Three studies evaluated patients on dialysis with NAFLD [25–27]. The remaining studies evaluated patients with cirrhosis. A total of 21 523 patients had cirrhosis. Information on cirrhosis diagnosis came from abstraction from coding records, patient databases and clinical notes in 98% and from review of histological, clinical or radiological findings in 2%. NAFLD was identified in 119 patients based on ultrasound or FibroScan imaging findings.
Risk of bias
Risk of bias was assessed using the NOS (Supplemental Tables S1–S3). Eleven studies were of high quality, with a score ≥7. Four scored ≤6 [24–27] and therefore were at high risk of exhibiting bias. Sensitivity analyses were performed, where applicable, to examine data excluding studies exhibiting high risk of bias (Supplemental Table S4). All studies evaluating NAFLD exhibited a high risk of bias.
Prevalence of CLD among patients with ESKD receiving dialysis
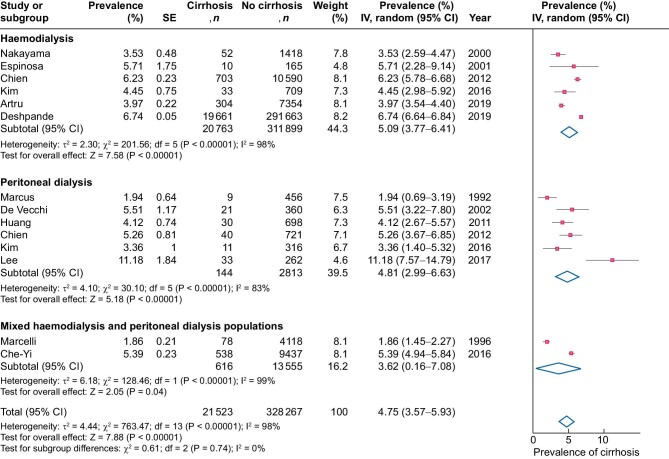
The prevalence of cirrhosis ranged from 2 to 11%. The overall pooled prevalence of cirrhosis across studies using a random effects model was 5% (Figure 2). There was no significant difference in prevalence between those treated with HD (5%) and PD (5%). Sensitivity analyses performed excluding outlier, case–control and case series studies did not yield significantly different results (Supplemental Figure S3 and Supplemental Table S4). Seven studies assessed cirrhosis severity [14, 15, 17, 20–22, 24]. A total of 60% had Child–Pugh A, 23% Child–Pugh B and 17% Child–Pugh C cirrhosis (Table 2). Cirrhosis aetiology was only available from a minority of studies but was pooled from the six studies where there were available data [15, 17, 19, 21, 22, 24]. A total of 35% had cirrhosis secondary to hepatitis B, 32% secondary to hepatitis C and 17% secondary to alcohol-related liver disease. The aetiology of cirrhosis was undefined in 15% of patients across these six studies (Table 3). Heterogeneity across these studies was high (I2 = 98%). Sensitivity analyses excluding studies at high risk of bias did not yield significantly different results in terms of the severity and aetiology of cirrhosis or heterogeneity (Supplemental Table S4).
FIGURE 2:
Prevalence of cirrhosis in dialysis patients by modality. SE: standard error; IV: inverse variance; df: degrees of freedom; I2: total variability due to heterogeneity.
Table 2.
Severity of cirrhosis in ESKD patients
| Study | Child–Pugh A, n | Child–Pugh B, n | Child–Pugh C, n | Total, N | |
|---|---|---|---|---|---|
| Haemodialysis | |||||
| Che-Yi 2016 [15] (HD cohort)a | 199 | 59 | 82 | 340 | |
| Kim 2016 [21] (HD cohort) | 15 | 13 | 5 | 33 | |
| Artru 2019 [14] | 150 | 52 | 11 | 213 | |
| Subtotal (n) | 364 | 124 | 98 | 586 | |
| % | 62 | 21 | 17 | ||
| Peritoneal dialysis | |||||
| Marcus 1992 [24] | 0 | 7 | 2 | 9 | |
| De Vecchi 2002 [17] | 10 | 5 | 6 | 21 | |
| Huang 2011 [20] | 16 | 12 | 2 | 30 | |
| Che-Yi 2016 [15] (PD cohort)a | 48 | 17 | 20 | 85 | |
| Kim 2016 [21] (PD cohort) | 6 | 3 | 2 | 11 | |
| Lee 2017 [22] | 20 | 10 | 3 | 33 | |
| Subtotal (n) | 100 | 54 | 35 | 189 | |
| % | 53 | 29 | 18 | ||
| Total | 464 | 178 | 133 | 775 | |
| % Overall | 60 | 23 | 17 |
Data on severity only provided for CMUH cohort.
Table 3.
Aetiology of cirrhosis in ESKD patients
| Study | HBV, n | HCV, n | ArLD, n | Multifactorial disease (HBV/HCV/ArLD combination), n | Other, n | Unknown, n | Total, N | |
|---|---|---|---|---|---|---|---|---|
| Haemodialysis | ||||||||
| Espinosa 2001 [19] | 9 | 1 | 10 | |||||
| Che-Yi 2016 [15] (HD cohort—full dataset)a | 500 | 478 | 253 | 225 | 1456 | |||
| Kim 2016 [21] (HD cohort) | 25 | 5 | 3 | 33 | ||||
| Subtotal (n) | 525 | 487 | 258 | 4 | 225 | 1499 | ||
| % | 35 | 32 | 17 | <1 | 15 | |||
| Peritoneal dialysis | ||||||||
| Che-Yi 2016 [15] (PD cohort—full dataset)a | 117 | 117 | 66 | 64 | 364 | |||
| De Vecchi 2002 [17] | 4 | 9 | 1 | 2 | 5 | 21 | ||
| Kim 2016 [21] (PD cohort) | 8 | 1 | 0 | 2 | 11 | |||
| Lee 2017 [22] | 16 | 9 | 5 | 3 | 33 | |||
| Marcus 1992 [24] | 1 | 5 | 1 | 2 | 9 | |||
| Subtotal (n) | 146 | 136 | 77 | 4 | 1 | 74 | 438 | |
| % | 33 | 31 | 18 | <1 | <1 | 17 | ||
| Total | 671 | 623 | 335 | 8 | 1 | 299 | 1937 | |
| Overall % | 35 | 32 | 17 | <1 | <1 | 15 | ||
HBV: hepatitis B; HCV: hepatitis C; ArLD: alcohol-related liver disease.
All Che-Yi data included (no risk of duplication).
Data from Artru, Deshpande, Nakayama, Huang excluded as incomplete data set on aetiology of cirrhosis.
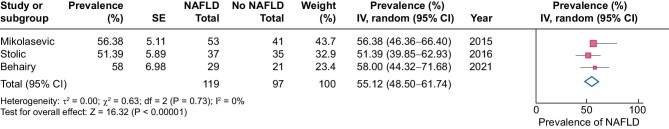
The overall prevalence of NAFLD across studies using a random effects model was 55% (Figure 3). Heterogeneity across these studies was low (I2 = 0%). It was not possible to perform sensitivity analyses for this subgroup, as all studies exhibited a high risk of bias.
FIGURE 3:
Prevalence of NAFLD in dialysis patients. SE: standard error; IV: inverse variance; df: degrees of freedom; I2: total variability due to heterogeneity.
Mortality risk and the effect of CLD in ESKD receiving dialysis
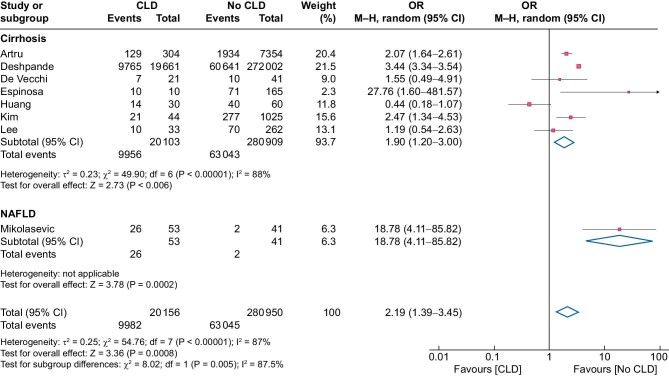
Patients with CLD had a >2-fold higher likelihood of mortality compared with those without CLD [OR 2.19 (95% CI 1.39–3.45)] (Figure 4). In the cirrhosis group, the OR for death was 1.90 (95% CI 1.20–3.00) and in the NAFLD group (one study), the OR for death was 18.78 (95% CI 4.11–85.82) (Figure 4). The heterogeneity observed in the analyses for the cirrhosis group was high (I2 = 88%) and remained so (I2 = 88%) in the sensitivity analysis, which excluded outlier studies [19, 20], with minimal change in the OR for death (Supplemental Table S4). Data on the mortality risk of cirrhosis among dialysis patients were also described in three other studies [16, 21, 23], but these data were excluded, as the raw data were unobtainable. In these three studies, cirrhosis conferred a significant increased risk of mortality, despite adjustment for other comorbidities. The severity of cirrhosis and its relationship with mortality were measured in four studies [14, 15, 17, 24]. An increasing severity of cirrhosis (classified by the Child–Pugh score or the presence of liver disease decompensation) was associated with increased mortality in the studies where this information was available (Supplemental Table S5).
FIGURE 4:
Association between death in dialysis patients with and without cirrhosis or NAFLD. M-H: Mantel–Haenszel; df: degrees of freedom; I2: total variability due to heterogeneity.
Patients with cirrhosis had an increased risk of death compared with those without, from infection [OR 2.17 (95% CI 1.51–3.11)], cancer (including hepatocellular carcinoma) [OR 5.42 (95% CI 1.01–28.96)] and liver disease [OR 28.46 (95% CI 16.52–49.03)], but not from cardiovascular disease [OR 0.96 (95% CI 0.54–1.73)] (Supplemental Figure S1).
Risk factors
Both hepatitis B [OR 13.47 (95% CI 1.37–132.55)] and hepatitis C [OR 7.05 (95% CI 4.00–12.45)] conferred an increased risk of cirrhosis, although this was not the case with DM [OR 1.19 (95% CI 0.97–1.46)] (Supplemental Figure S2).
Moderator analyses
Meta-regression was performed to consider the effect of age, gender, study size and year of study publication on cirrhosis prevalence in dialysis patients (Supplemental Figures S4–S7). The year of study publication was significantly associated with cirrhosis prevalence (P = 0.027), with increasing prevalence found in more recently published studies. Age, gender and study size were not significantly associated with cirrhosis prevalence. Other moderator analyses planned a priori were not performed due to the small number of studies assessing the variables of interest [34].
Assessment for publication bias
Publication biases were considered by examining funnel plot asymmetry (Supplemental Figure S8). There was no evidence of publication bias in the reporting of CLD across studies (Egger regression test P = 0.058).
DISCUSSION
This systematic review and meta-analysis assessed the prevalence of CLD in patients treated with dialysis. Risk factors for CLD in dialysis patients were explored, along with associations with all-cause mortality and cardiovascular, infectious, cancer and liver mortality. Cirrhosis and NAFLD prevalence was 5 and 55%, respectively. Outcomes for dialysis patients with CLD were worse, with a >2-fold higher likelihood of mortality for patients with CLD compared with those without.
We found a stronger association with mortality for NAFLD than for cirrhosis, although this should be interpreted with significant caution. It is based on data from a single study with a high risk of bias and small sample size (Figure 4). Patients with cirrhosis were at increased risk of death from infection, cancer and liver disease, but not from cardiovascular disease. In most studies, the exact cause of liver death was not detailed.
Risk of infectious death in cirrhosis patients receiving dialysis was lower than previously described among all patients with cirrhosis [35]. This may relate to high rates of infectious death among dialysis patients without cirrhosis. Additionally, deaths attributed to liver disease may have been due to bacterial infection, a well-recognized trigger for liver decompensation [36].
Interestingly, DM did not confer an increased risk of cirrhosis, although there was a trend towards significance. The high rates of hepatitis observed in these populations may have masked the impact of DM.
It is currently unclear what proportion of dialysis patients have undiagnosed CLD. Liver enzymes are often normal in advanced CLD [37], and the majority of patients with NASH in the general population remain undiagnosed [38]. The difficulties in obtaining a diagnosis of CLD and the impact of the method of liver disease diagnosis on coded diagnostic information may explain the high level of heterogeneity observed across studies. This may be a factor in the differences in prevalence rates of cirrhosis observed in two of the largest studies in this analysis. The prevalence was 4.0% (95% CI 3.5–4.4) in Artru et al. [14] but was 6.7% (95% CI 6.6–6.8) in Deshpande et al. [18]. Differences in dialysis vintage may also be an important factor. This parameter was reported in five studies [19, 22, 25, 26, 28] and ranged between 5 and 7 years.
In patients not on dialysis, Child–Pugh A cirrhosis is associated with better outcomes and Child–Pugh C cirrhosis with the worst [39, 40]. An increasing severity of cirrhosis in patients with ESKD, as evidenced by a higher Child–Pugh classification or the presence of decompensated liver disease, was similarly associated with poorer outcomes. Furthermore, patients with decompensated cirrhosis with coexistent advanced kidney disease (e.g. those with hepatorenal syndrome) have a very poor short-term prognosis without liver transplantation [41] and may not have survived the minimum period on dialysis to meet study inclusion criteria. This may have led to an underestimation of survival rates in this systematic review, as six studies included in this review considered only patients who had been dialysing for a minimum period of 30 days–6 months [15, 16, 19, 23, 26, 27]. Other studies that abstracted registry data may not have included inpatients initiating dialysis who did not survive to hospital discharge. In addition, patients with decompensated cirrhosis may not have been deemed suitable for long-term dialysis if they were not considered potential candidates for liver transplantation.
Where the cause of liver disease was available, two-thirds had cirrhosis secondary to hepatitis B or C, although these analyses were influenced by the results of a large Taiwanese data set [15]. As a result, hepatitis B and C were the most relevant factors for the development of cirrhosis in dialysis patients in this systematic review. However, these results may not be generalizable to current populations where patients are now vaccinated against hepatitis B virus and treatments to eradicate hepatitis C are easily accessible.
There were no data on NASH cirrhosis or obesity, despite the increasing impact of these conditions on the global burden of CLD [42]. Therefore the impact of NASH cirrhosis among dialysis populations remains to be determined. Three articles with small sample sizes reported on the prevalence of NAFLD in the dialysis setting and indicated that the prevalence of NAFLD in dialysis patients was greater than that in the general population [2]. The true prevalence of NAFLD may be even higher in dialysis populations, as two of these three studies excluded patients with obesity and DM [26, 27]. It was not possible to establish NAFLD as a risk factor for cardiovascular mortality as part of this study due to insufficient data.
The studies involving patients with cirrhosis and ESKD exhibited significant heterogeneity. This is likely to have been influenced by reporter bias, the variation in the prevalence of cirrhosis and hepatitis B and C in the populations studied based on geographical location, variations in follow-up times and the different study sample sizes. No significant evidence of publication bias was identified. Sensitivity analyses that excluded the studies with a high risk of bias did not alter heterogeneity or interpretation of the overall data on cirrhosis prevalence. It was not possible to perform moderator analyses to adequately explore possible factors that may explain the high level of heterogeneity observed (other than age, gender, study size and year of publication), as there was insufficient consistency on the reporting of other characteristics of interest (at least 10 observations per characteristic are recommended for a moderator analysis to be performed) [34]. The year of study publication was associated with an increasing reported prevalence of cirrhosis, reflecting the increasing numbers of patients with combined kidney and liver dysfunction and potentially improved diagnosis. A requirement for future studies to report on risk factors for liver disease, including DM, obesity and ethnicity, would enable better stratification of patients at high risk of NAFLD and NASH cirrhosis.
This is the first systematic review and meta-analysis to evaluate CLD in dialysis patients. While offering the largest evidence synthesis to date, some caveats apply when interpreting the findings. These mainly relate to the degree of heterogeneity observed across studies, which, as discussed, we could only explore in a limited fashion because of scarce reporting of moderators of interest. Hence some of the analyses outlined in our PROSPERO preregistration document were not possible to pursue. These included relationships with anaemia, hypoalbuminemia, quality of life, hospital admission and length of hospital stay. All these variables could provide important information about current clinical status, morbidity and patient-reported outcomes. However, the strengths of this review include the provision of evidence-based assessments for the co-occurrence of CLD and ESKD and, importantly, in detailing its impact on mortality in this patient group. The review also highlights important avenues for future research, especially on the impact of NAFLD and NASH in dialysis patients. Both NAFLD and NASH [43, 44] are associated with DM, obesity, hypertension and systemic inflammation, all of which are commonly encountered in the dialysis population [45]. Given the high prevalence of NAFLD we have observed, further investigation is required on the role of NAFLD and NASH in systemic inflammation in ESKD, the risk of developing cirrhosis among dialysis patients with NAFLD and NASH and their impact on outcomes, including mortality.
In conclusion, this systematic review and meta-analysis has demonstrated that CLD is an important comorbidity associated with increased risks of mortality in this already vulnerable patient group. Close collaborative work between nephrologists and hepatologists to manage these patients is required, alongside further studies to identify those at highest risk of developing these complications and to define the additional burdens they impose on dialysis patients.
Supplementary Material
Contributor Information
Oscar Swift, Department of Renal Medicine, Lister Hospital, East and North Hertfordshire NHS Trust, Stevenage, UK; School of Life and Medical Sciences, University of Hertfordshire, Hatfield, Hertfordshire, UK.
Shivani Sharma, School of Life and Medical Sciences, University of Hertfordshire, Hatfield, Hertfordshire, UK.
Sivaramakrishnan Ramanarayanan, Department of Renal Medicine, Lister Hospital, East and North Hertfordshire NHS Trust, Stevenage, UK.
Hamza Umar, College of Medical and Dental Sciences, University of Birmingham, Birmingham, UK.
Keith R Laws, School of Life and Medical Sciences, University of Hertfordshire, Hatfield, Hertfordshire, UK.
Enric Vilar, Department of Renal Medicine, Lister Hospital, East and North Hertfordshire NHS Trust, Stevenage, UK; School of Life and Medical Sciences, University of Hertfordshire, Hatfield, Hertfordshire, UK.
Ken Farrington, Department of Renal Medicine, Lister Hospital, East and North Hertfordshire NHS Trust, Stevenage, UK; School of Life and Medical Sciences, University of Hertfordshire, Hatfield, Hertfordshire, UK.
CONFLICT OF INTEREST STATEMENT
None declared. The results presented have not been published previously in whole or part, except in abstract format.
REFERENCES
- 1. Webster AC, Nagler EV, Morton RL et al. Chronic kidney disease. Lancet 2017; 389: 1238–1252 [DOI] [PubMed] [Google Scholar]
- 2. Vernon G, Baranova A, Younossi ZM. Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther 2011; 34: 274–285 [DOI] [PubMed] [Google Scholar]
- 3. Estes C, Razavi H, Loomba R et al. Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease. Hepatology 2018; 67: 123–133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Singh S, Allen AM, Wang Z et al. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: a systematic review and meta-analysis of paired-biopsy studies. Clin Gastroenterol Hepatol 2015; 13: 643–654.e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. GBD 2017 Cirrhosis Collaborators . The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol 2020; 5: 245–266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Hazlehurst JM, Woods C, Marjot T et al. Non-alcoholic fatty liver disease and diabetes. Metabolism 2016; 65: 1096–1108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Maurice J, Manousou P. Non-alcoholic fatty liver disease. Clin Med 2018; 18: 245–250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Jadoul M, Bieber BA, Martin P et al. Prevalence, incidence, and risk factors for hepatitis C virus infection in hemodialysis patients. Kidney Int 2019; 95: 939–947 [DOI] [PubMed] [Google Scholar]
- 9. Edey M, Barraclough K, Johnson DW. Review article: hepatitis B and dialysis. Nephrology 2010; 15: 137–145 [DOI] [PubMed] [Google Scholar]
- 10. Burdick RA, Bragg-Gresham JL, Woods JD et al. Patterns of hepatitis B prevalence and seroconversion in hemodialysis units from three continents: the DOPPS. Kidney Int 2003; 63: 2222–2229 [DOI] [PubMed] [Google Scholar]
- 11. Miles CD, Westphal S, Liapakis A et al. Simultaneous liver-kidney transplantation: impact on liver transplant patients and the kidney transplant waiting list. Curr Transplant Rep 2018; 5: 1–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Khan S, Rosner MH. Peritoneal dialysis for patients with end-stage renal disease and liver cirrhosis. Perit Dial Int 2018; 38: 397–401 [DOI] [PubMed] [Google Scholar]
- 13. Page MJ, McKenzie JE, Bossuyt PM et al. Updating guidance for reporting systematic reviews: development of the PRISMA 2020 statement. J Clin Epidemiol 2021; 134: 103–112 [DOI] [PubMed] [Google Scholar]
- 14. Artru F, Louvet A, Bellati S et al. The prognostic impact of cirrhosis on patients receiving maintenance haemodialysis. Aliment Pharmacol Ther 2019; 50: 75–83 [DOI] [PubMed] [Google Scholar]
- 15. Che-Yi C, Shu-Ming W, Chih-Chia L et al. Peritoneal dialysis is associated with a better survival in cirrhotic patients with chronic kidney disease. Medicine (Baltimore) 2016; 95: e2465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Chien CC, Wang JJ, Sun YM et al. Long-term survival and predictors for mortality among dialysis patients in an endemic area for chronic liver disease: a national cohort study in Taiwan. BMC Nephrol 2012; 13: 43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. De Vecchi AF, Colucci P, Salerno F et al. Outcome of peritoneal dialysis in cirrhotic patients with chronic renal failure. Am J Kidney Dis 2002; 40: 161–168 [DOI] [PubMed] [Google Scholar]
- 18. Deshpande R, Stepanova M, Golabi P et al. Prevalence, mortality and healthcare utilization among Medicare beneficiaries with hepatitis C in haemodialysis units. J Viral Hepat 2019; 26: 1293–1300 [DOI] [PubMed] [Google Scholar]
- 19. Espinosa M, Martin-Malo A, Alvarez de Lara MA et al. Risk of death and liver cirrhosis in anti-HCV-positive long-term haemodialysis patients. Nephrol Dial Transplant 2001; 16: 1669–1674 [DOI] [PubMed] [Google Scholar]
- 20. Huang ST, Chuang YW, Cheng CH et al. Outcome of peritoneal dialysis in cirrhotic patients with end-stage renal disease—a 24-years' experience in Taiwan. Clin Nephrol 2011; 76: 306–313 [DOI] [PubMed] [Google Scholar]
- 21. Kim AJ, Lim HJ, Ro H et al. Liver cirrhosis leads to poorer survival in patients with end-stage renal disease. Korean J Intern Med 2016; 31: 730–738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Lee SM, Son YK, Kim SE et al. Clinical outcomes of peritoneal dialysis in end-stage renal disease patients with liver cirrhosis: a propensity score matching study. Perit Dial Int 2017; 37: 314–320 [DOI] [PubMed] [Google Scholar]
- 23. Marcelli D, Stannard D, Conte F et al. ESRD patient mortality with adjustment for comorbid conditions in Lombardy (Italy) versus the United States. Kidney Int 1996; 50: 1013–1018 [DOI] [PubMed] [Google Scholar]
- 24. Marcus RG, Messana J, Swartz R. Peritoneal dialysis in end-stage renal disease patients with preexisting chronic liver disease and ascites. Am J Med 1992; 93: 35–40 [DOI] [PubMed] [Google Scholar]
- 25. Mikolasevic I, Racki S, Devcic B et al. Relationship between non-alcoholic fatty liver disease and MIA syndrome. Hemodial Int 2015; 19: 472–481 [DOI] [PubMed] [Google Scholar]
- 26. Stolic RV, Trajkovic GZ, Kostic MM et al. Correlation between nonalcoholic fatty liver and cardiovascular disease in elderly hemodialysis patients. Int Urol Nephrol 2016; 48: 883–889 [DOI] [PubMed] [Google Scholar]
- 27. Behairy MA, Sherief AF, Hussein HA. Prevalence of non-alcoholic fatty liver disease among patients with non-diabetic chronic kidney disease detected by transient elastography. Int Urol Nephrol 2021; 53: 2593–2601 [DOI] [PubMed] [Google Scholar]
- 28. Nakayama E, Akiba T, Marumo F et al. Prognosis of anti-hepatitis C virus antibody-positive patients on regular hemodialysis therapy. J Am Soc Nephrol 2000; 11: 1896–1902 [DOI] [PubMed] [Google Scholar]
- 29. Wells GA, Shea B, O'Connell D et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of non-randomised studies in meta-analysis. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (14 April 2021, date last accessed) [Google Scholar]
- 30. Stang A. Critical evaluation of the Newcastle-Ottawa Scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol 2010; 25: 603–605 [DOI] [PubMed] [Google Scholar]
- 31. Suurmond R, van Rhee H, Hak T. Introduction, comparison, and validation of meta-essentials: a free and simple tool for meta-analysis. Res Synth Methods 2017; 8: 537–553 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. DerSimonian R, Kacker R. Random-effects model for meta-analysis of clinical trials: an update. Contemp Clin Trials 2007; 28: 105–114 [DOI] [PubMed] [Google Scholar]
- 33. Higgins JP, Thompson SG, Deeks JJ et al. Measuring inconsistency in meta-analyses. BMJ 2003; 327: 557–560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Higgins J, Thomas J, Chandler J et al. (eds). Cochrane Handbook for Systematic Reviews of Interventions, Version 6.2 (updated February 2021). London: Cochrane Collaboration, 2021 [Google Scholar]
- 35. Arvaniti V, D'Amico G, Fede G et al. Infections in patients with cirrhosis increase mortality four-fold and should be used in determining prognosis. Gastroenterology 2010; 139: 1246–1256.e5 [DOI] [PubMed] [Google Scholar]
- 36. Jalan R, Fernandez J, Wiest R et al. Bacterial infections in cirrhosis: a position statement based on the EASL Special Conference 2013. J Hepatol 2014; 60: 1310–1324 [DOI] [PubMed] [Google Scholar]
- 37. Lominadze Z, Kallwitz ER. Misconception: you can't have liver disease with normal liver chemistries. Clin Liver Dis 2018; 12: 96–99 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Morgan A, Hartmanis S, Tsochatzis E et al. Disease burden and economic impact of diagnosed non-alcoholic steatohepatitis (NASH) in the United Kingdom (UK) in 2018. Eur J Health Econ 2021; 22: 505–518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Pugh RN, Murray-Lyon IM, Dawson JL et al. Transection of the oesophagus for bleeding oesophageal varices. Br J Surg 1973; 60: 646–649 [DOI] [PubMed] [Google Scholar]
- 40. D'Amico G, Garcia-Tsao G, Pagliaro L. Natural history and prognostic indicators of survival in cirrhosis: a systematic review of 118 studies. J Hepatol 2006; 44: 217–231 [DOI] [PubMed] [Google Scholar]
- 41. Allegretti AS, Parada XV, Eneanya ND et al. Prognosis of patients with cirrhosis and AKI who initiate RRT. Clin J Am Soc Nephrol 2018; 13: 16–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Paik JM, Golabi P, Younossi Y et al. Changes in the global burden of chronic liver diseases from 2012 to 2017: the growing impact of NAFLD. Hepatology 2020; 72: 1605–1616 [DOI] [PubMed] [Google Scholar]
- 43. Byrne CD, Targher G. NAFLD: a multisystem disease. J Hepatol 2015; 62(1 Suppl): S47–S64 [DOI] [PubMed] [Google Scholar]
- 44. Luci C, Bourinet M, Leclère PS et al. Chronic inflammation in non-alcoholic steatohepatitis: molecular mechanisms and therapeutic strategies. Front Endocrinol 2020; 11: 597648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Cobo G, Lindholm B, Stenvinkel P. Chronic inflammation in end-stage renal disease and dialysis. Nephrol Dial Transplant 2018; 33(Suppl 3): iii35–iii40 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.