Summary
Background
Staphylococcus aureus bacteraemia (SAB) is one of the commonest bloodstream infections globally and is associated with a high mortality rate. Most published data comes from temperate, high-income countries. We describe the clinical epidemiology, microbiology, management and outcomes of patients with SAB treated in a tropical, middle-income setting at Fiji's largest hospital.
Methods
A prospective, observational study was performed of consecutive SAB cases admitted to Colonial War Memorial Hospital (CWMH) in Suva, between July 2020 and February 2021. Detailed demographic, clinical and microbiological data were collected, including the key outcome of in-patient mortality. To estimate the population incidence, all SAB cases diagnosed at the CWMH laboratory were included – even if not admitted to CWMH – with the population of Fiji's Central Division used as the denominator.
Findings
A total of 176 cases of SAB were detected over eight-months, which equated to an incidence of 68.8 cases per 100,000 population per year. Of these, 95 cases were admitted to CWMH within 48 h of index culture. Approximately 8.4% (8/95) of admitted cases were caused by methicillin-resistant Staphylococcus aureus (MRSA). All cause in-patient mortality was 25.3%, increasing to 55% among patients aged 60 or older.
Interpretation
This reported incidence of SAB in central Fiji is one of the highest in the world. SAB was associated with significant mortality, especially in those over 60 years of age, despite a relatively low frequency of methicillin resistance.
Funding
Supported by the National Health and Medical Research Council (Australia) and the GRAM (Global Research on Antimicrobial Resistance) Project, Oxford University (United Kingdom).
Keywords: Fiji, Pacific Islands, Staphylococcus aureus bacteraemia, Healthcare-associated infections, Methicillin-resistant staphylococcus aureus
Research in context.
Evidence before this study
Staphylococcus aureus bacteraemia (SAB) is a common cause of bloodstream infection and associated with high mortality. However, most knowledge about the epidemiology and outcomes of SAB comes from temperate, high-income countries. The only previous study of SAB from the Pacific Islands region was conducted over a decade ago in Fiji – this reported an incidence of 50 cases per 100,000 population per year however no outcome data was available.
Added value of this study
We analysed all cases of SAB from the largest hospital of a major Pacific Island Country (Fiji), and found an annualised incidence of 69 cases per 100,000 population. Among those admitted to CWMH, the rate of MRSA was low at only 8.4%, almost half of all adults had diabetes, and in-hospital mortality was over 25%. Healthcare-associated infections were responsible for almost one-third of SAB cases (n = 28, 29%), and are therefore potentially preventable.
Implications of the available evidence
Our study demonstrates that central Fiji has one of the highest reported rates of SAB in the world, and is associated with significant in-hospital mortality that is greater than high-income countries but comparable to equivalent estimates from other low- and middle-income countries. Efforts to reduce SAB in Fiji could focus on diabetes and skin disease management at the community level, and prevention of healthcare-associated infections at the hospital level.
Alt-text: Unlabelled box
Introduction
Staphylococcus aureus bacteraemia (SAB) is one of the commonest bloodstream infections worldwide.1 The all-cause mortality rate from SAB in high-income countries has been reported to be up to 20–30%,2,3 with even higher rates of over 50% seen in some low- and middle-income countries (LMICs).4,5
S. aureus infections – including SAB – are of great importance in the fields of both antimicrobial resistance (AMR) and healthcare-associated infections (HAIs). Regarding AMR, methicillin-resistant S. aureus (MRSA) has been identified as a High Priority Pathogen by the World Health Organization.6 Antibiotic treatment options for MRSA are more limited and may not always be available throughout LMIC settings. Until recently there has been a paucity of data on MRSA prevalence from LMICs,7 a situation that is slowly improving due to initiatives such as the Global Antimicrobial Resistance and Use Surveillance System (GLASS).8 Regarding HAIs, S. aureus is the most frequent cause of healthcare-associated bacteraemia in both high-income9 and LMIC settings.10 Healthcare-associated SAB (HA-SAB) has been used in surveillance programs as a marker of healthcare quality,11 in part as there are specific interventions – such as hand hygiene12 and line-insertion bundles13 – that are proven to reduce the rates of HA-SAB, including in LMICs.14,15
SAB is of particular importance to tropical regions such as the Pacific Islands. This region experiences high rates of skin disease (including impetigo, scabies and dermatophyte infections)16 and diabetes17 – both key risk factors for community-acquired SAB. A previous study of SAB from Fiji over a decade ago demonstrated a very high annual incidence of 50 cases per 100,000 population.18
Currently, most published data on SAB comes from high-income, temperate countries. There is little available data from low- and middle-income tropical countries,19,20 despite the majority of the global population residing in such settings. To address this knowledge gap, we describe the clinical epidemiology, microbiology, management and outcomes of patients with SAB treated at Fiji's largest hospital.
Methods
Study design
We conducted a prospective, observational study of SAB among inpatients at the Colonial War Memorial Hospital (CWMH) in Suva, over eight months from July 2020 until February 2021.
Setting
CWMH is a 500-bed tertiary hospital and the largest healthcare facility in Fiji. It serves a local catchment population of Fiji's Central Division (almost 400,000 people) and is also the national referral centre for Fiji's other two divisional hospitals. It has adult, paediatric and neonatal intensive care units (ICUs), and offers specialty services including cardiology, nephrology, orthopaedics, plastic surgery, urology and neurosurgery.
CWMH has a six-member Infection Prevention and Control team that conducts regular surveillance of select HAIs. Alcohol-based handrub is available on all CWMH wards, and is present at each bedside within ICUs. A recent point-prevalence survey of HAIs at CWMH reported that 8.7% of inpatients had an HAI, 69.4% had a peripheral vascular catheter and 5.5% had a central vascular catheter.21
Population
Patients with SAB were eligible if they were admitted to CWMH at the time of blood culture collection, or up to 48 h following blood culture collection. Patients were to be excluded if they were not being treated with curative intent for the entire calendar day on which the index blood culture was collected.
Patient identification
We identified patients with SAB by screening new positive blood culture results each day. The CWMH laboratory receives clinical samples not only from CWMH inpatients, but also from nearly every other healthcare centre in Fiji's Central Division. We noted all SAB results in order to estimate a local population incidence, but only collected clinical data for CWMH inpatients. Ward-based data collectors then determined whether patients with SAB were eligible. The notes of eligible patients were reviewed on the wards or in medical records as soon as practicable.
Data collection
Study data were collected and managed using REDCap (Research Electronic Data Capture) tools hosted at Monash University, Australia.22 Clinical and antibiotic data were collected at the initial chart review. Clinical data used definitions from the WHO GLASS Attributable Mortality of AMR Bloodstream Infections document,23 unless specified otherwise below and outlined in greater detail in the Supplementary Appendix Table S1. Clinical data included patient age, sex, admission date, admission source, admitting specialty, age-adjusted Charlson comorbidity index (CCI) score, Pitt bacteraemia score and high-risk quick Sequential Organ Failure Assessment (qSOFA) score at SAB onset, and whether the patient had any immunosuppression, hospitalisation, ICU admission, surgical procedure or antibiotic exposure preceding their SAB. Diverging from the WHO GLASS document, we also collected data on the likely source of infection and the epidemiological attribution of their SAB (using the categories of Hospital Acquired, Community Acquired, or Healthcare Associated24). Antibiotic data included all antibiotics received between Days 0 to 2 inclusive (‘initial antibiotic therapy’; Day 0 = day of positive blood culture), and the antibiotic susceptibility profile as reported by the Suva lab was also obtained. As the chart review occurred at one time point only, shortly after the diagnosis of SAB, we were unable to collect data on the duration of antibiotic therapy.
Effective initial antibiotic therapy was defined as receipt of at least one agent listed in Fiji's Therapeutic Guidelines:25 for methicillin-susceptible S. aureus (MSSA), intravenous (IV) (flu)cloxacillin or cephazolin; for MRSA, IV vancomycin. Broad-spectrum beta-lactams with anti-staphylococcal activity (such as piperacillin-tazobactam) were also counted as effective against MSSA. Chloramphenicol is commonly used in Fiji to treat non-invasive S. aureus infections, however we deemed chloramphenicol ‘ineffective’ therapy as it is not a recommended agent for SAB.25
Data on repeat blood cultures, echocardiography results and patients’ discharge date and status were collected retrospectively after patients had either been discharged or died, these data did not require further review of the paper medical record. Any patients discharged home to die had their discharge status recorded as moribund. During data analysis, moribund patients were grouped with those who had died in hospital.
Patients’ clinical management was entirely at the discretion of their CWMH clinicians, with no input from the research team. There were no specific study interventions, such as mandated echocardiography or repeat blood cultures.
Laboratory testing
Initial identification and antibiotic susceptibility testing (AST) for S. aureus were performed by the CMWH laboratory as per their standard procedures. Clinical samples were plated onto 5% sheep blood agar. Identification was performed using the deoxyribonuclease test and the staphylococcus latex agglutination test, and AST was performed for first-line laboratory antimicrobials using the disk-diffusion method with CLSI breakpoints.26 Alternatively, if an isolate was either identified as MRSA or was growing repeatedly from blood cultures, identification and AST were performed using the VITEK-2 GP ID and VITEK-2 AST cards (bioMérieux, Marcy-Étoile, France; version 9.01), respectively.
Where possible, the index S. aureus isolate from each patient was sub-cultured, stored at -80⁰C, and shipped to Melbourne, Australia for confirmatory testing. Confirmatory identification was performed using MALDI-TOF (Bruker, Hanau, Germany) and confirmatory AST was performed using VITEK-2 AST cards (bioMérieux, version 8.01) on all available isolates.
Echocardiography
Echocardiography was performed at the discretion of treating clinicians. Adult transthoracic echocardiograms (TTEs) are performed by dedicated technicians, whereas for children all TTEs are performed by paediatricians trained in echocardiography. There is no access to transoesophageal echocardiography.
Population estimates
To calculate the community incidence of SAB, we used the population of the Central Division of Fiji from the most recent 2017 National Population and Housing Census.27 This was corrected for subsequent population growth using estimates published by the Pacific Community Statistics for Development Division28 to arrive at mid-year population estimates for 2020 and 2021. To calculate an annualised incidence of SAB, the denominator of person-months was first obtained by multiplying each year's population by however many months of surveillance took place within that year (six in 2020, and two in 2021) and then summing these totals. For the numerator, we excluded any SABs among non-residents of Central Division. The remaining SABs detected at CWMH over the study period were then divided by the number of person-months and multiplied by 12 to determine the annualised incidence of SAB.
To calculate the rate of healthcare-associated SABs per 10,000 patient days, we assumed full occupancy of CWMH over the 8-month study period, as hospital occupancy and patient-day data were not available. While this assumption could lead to an inflated denominator and underestimation of the true rate, it is offset by the fact that CWMH is regularly operating at, or slightly above, its stated bed capacity.
Statistical analysis
Data analysis was performed using R (version 3.6.1). In bivariate comparisons, we used Fisher's exact test for categorical variables and Mann–Whitney U test for numeric variables. The Wilson method was used to calculate confidence intervals for proportions. For logistic regression analysis, Directed Acyclic Graphs (DAGs) were employed to identify a minimally sufficient adjustment set of variables to control confounding (Supplementary Appendix Fig. S1). The two relationships of interest were the impact of methicillin-resistance on mortality, and the impact of effective initial therapy on mortality. This second relationship was only assessed within the MSSA cohort, as effective therapy is on the causal pathway between AMR status and mortality. Due to concerns regarding over-parameterisation, Akike's information criterion was used to assess the impact of additional variables on model fit. The manuscript has been prepared according to the STROBE checklist.
Ethics
Ethics approval was provided by the Alfred Hospital Ethics Committee (593/19) and the Fiji National Health Research Ethics Review Committee (88/2019).
Role of the funding source
The funders had no role in the design, data collection, analysis, and interpretation, or preparation of this manuscript.
Results
Laboratory testing
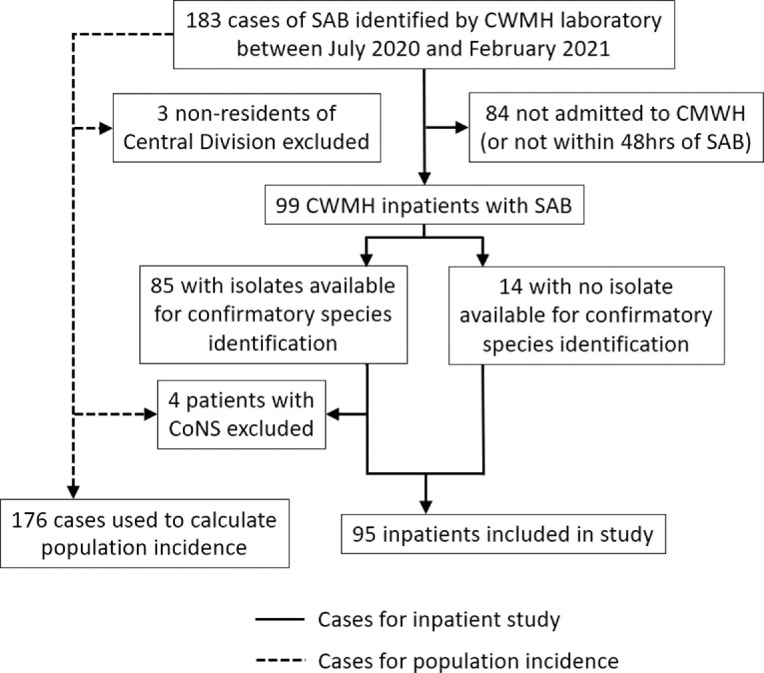
A total of 183 consecutive cases of SAB were identified at the CWMH laboratory during the study period, of which, 99 (54%) were associated with a patient admission to CWMH within 48 h of their index blood culture (Figure 1). 86% (85/99) of patients had an isolate available for confirmatory testing in Australia – four isolates were identified as coagulase-negative staphylococci (CoNS) and these patients were subsequently excluded from the study, leaving a total of 95 inpatients for analysis. Confirmatory AST was performed on all available isolates and found only one discrepancy – a patient with MRSA with that was initially identified in Suva as MSSA.
Figure 1.
Flowchart of patients included in the study.
SAB – S. aureus bacteraemia. CWMH – Colonial War Memorial Hospital. CoNS – Coagulase negative staphylococci.
Incidence of S. aureus bacteraemia
Excluding the four cases of CoNS identified on confirmatory testing, and three patients who did not reside in Fiji's Central Division, there were 176 separate instances of SAB detected at the CMWH laboratory over the eight-month study period. This equated to an annualised incidence of SAB for Fiji's Central Division of 68.8 cases per 100,000 population. Regarding HA-SAB, less than one-third of all SABs (28/95, 29.5%) were healthcare-associated (either hospital-acquired, or healthcare-associated but community-onset). This equates to a rate of HA-SAB of 2.3 per 10,000 patient days at CWMH across the study period.
Patient characteristics
Among the cohort of 95 inpatients with detailed clinical data, the median patient age was 44 years (IQR 16 – 59 years), with a range of 9 days to 89 years (Table 1). There were 25 (26%) paediatric patients <18 years, including two (2.1%) neonates <28 days (a full description of the paediatric cohort can be found in Supplementary Appendix Table S2). The median CCI score was 2, and the median Pitt Bacteraemia score was 0. The prevalence of diabetes among adult patients was 34/70 (48.6%). No paediatric patients had diabetes. All patients were being treated with curative intent for the entire calendar day of their index blood culture.
Table 1.
Patient characteristics.
| Community-Acquired SAB(n = 67) | Healthcare-Associated* SAB (n = 28) | P-value | |
|---|---|---|---|
| Demographics | |||
| Age in years on admission (median [IQR]) | 42.0 [11.5, 57.5] | 47.0 [24.7, 60.0] | 0.57 |
| Paediatric (age < 18 years) | 21 (31.3) | 4 (14.3) | 0.14 |
|
Sex Male Female |
42 (62.7) 25 (37.3) |
17 (60.7) 11 (39.3) |
>0.99 |
|
Admitting Unit Adult Medicine Adult Surgery Paediatrics Other |
26 (38.8) 20 (29.9) 17 (25.4) 4 (6.0) |
18 (64.3) 4 (14.3) 2 (7.1) 4 (14.3) |
0.035 |
| Age-adjusted Charlson comorbidity index score (median [IQR]) | 1.0 [0.0, 3.0] | 2.0 [0.8, 3.3] |
0.080 |
| Pitt Bacteraemia Score (median [IQR]) | 0.0 [0.0, 2.0] | 1.0 [0.0, 3.0] | 0.12 |
|
High-risk qSOFA score^ Yes No Not available |
8 (11.9) 50 (74.6) 9 (13.4) |
7 (25.0) 19 (67.9) 2 (7.1) |
0.23 |
| Infection | |||
|
Resistance profile of SAB MSSA MRSA |
63 (94.0) 4 (6.0) |
24 (85.7) 4 (14.3) |
0.36 |
|
Source of bacteraemia Primary Central line associated Secondary Skin and soft tissue infection Bone and joint infection Other |
23 (34.3) 0 (0.0) 44 (65.7) 34 (77.3) 7 (15.9) 3 (6.9) |
16 (57.1) 2 (7.1) 10 (35.7) 6 (60.0) 2 (20.0) 2 (20.0) |
0.0050 |
| Healthcare Exposures prior to SAB | |||
| Surgery# (n = 94) ICU stay > 48 h# (n = 93) Antibiotics in preceding 30 days (n = 93) Immunosuppressed (n = 93) Hospital LOS in days prior to SAB (median [IQR]) |
1 (1.5) 0 (0.0) 17 (26.2) 8 (12.3) 0 [0.0, 0.0] |
9 (32.1) 5 (18.5) 20 (71.4) 7 (25.0) 4 [0.0, 7.5] |
<0.0001 0.0010 <0.0001 0.20 <0.0001 |
| Effective Initial Antibiotics | 55 (82.1%) | 16 (57.1%) | 0.022 |
| Outcome | |||
| Hospital LOS (median [IQR]) | 17.00 [9.5, 26.0] | 21.00 [12.0, 27.0] | 0.29 |
|
Discharge Status Alive Moribund Died |
54 (80.6) 0 (0.0) 13 (19.4) |
17 (60.7) 1 (3.6) 10 (35.7) |
0.061 |
LOS = Length of Stay. qSOFA = quick Sequential Organ Failure Assessment.
Healthcare-associated SAB includes both Hospital-acquired and Healthcare-associated.
High-risk qSOFA score defined as ≥ 2 (out of a possible score of 3).
Between hospital admission and SAB.
MRSA was responsible for 8/95 (8.4%) cases of SAB, all of which occurred in adults. Just over two-thirds of all admitted SABs (67/95, 71%) were community-acquired. While only 6% (4/67) of community-acquired SABs were caused by MRSA, this rose to 14% (4/28) among healthcare-associated infections, although this was not statistically significant (p = 0.22).
Almost half of all SABs (39/95, 41%) were defined as primary infections, with a further two as central-line associated and the remaining 54 as secondary infections (Table 1). Skin and soft tissue (40/54, 74%) and bone and joint (9/54, 17%) sources accounted for almost all secondary infections.
Blood cultures
Overall, 219 blood cultures were collected; ≥1 repeat blood culture was performed in 77/95 (81.1%) patients and 33/95 (34.7%) patients had ≥2 repeat blood cultures. There was variability in the time to first repeat blood culture, with a median of 4 days (IQR 3-6 days) but ranging from 0 to 16 days.
Of the 77 patients with at least one repeat blood culture, a negative blood culture was achieved in 73 (94.8%). The remaining four patients all died before clearing their blood cultures. The median time to first negative blood culture was 6 days (IQR 4–8 days, range 0–17), and the median number of further blood cultures until first negative was 1 (IQR 1–2, range 1–5). The longest duration of bacteraemia was ten days and the most positive blood cultures for a single patient was five, across a nine-day period.
Microbiology
The antibiotic susceptibility seen among the SAB isolates is summarised in Table 2. Resistance to penicillin among MSSA isolates was almost universal, with only 4/73 (5.5%) remaining susceptible. All tested MSSA isolates were susceptible to clindamycin and tetracycline, with low (≤10%) rates of resistance seen to ciprofloxacin and trimethoprim-sulfamethoxazole (TMP-SMX). Of note, all MRSA isolates remained susceptible to vancomycin, clindamycin, and TMP-SMX.
Table 2.
Antibiotic susceptibility among S. aureus isolates with confirmatory testing in Australia (n = 81).
| Antibiotic | MSSA (n = 73) | MRSA (n = 8) |
|---|---|---|
| Number of susceptible isolates (%) | ||
| Ciprofloxacin | 44 (90%) | 4 (50%) |
| Clindamycin | 73 (100%) | 8 (100%) |
| Fusidic Acid | 73 (100%) | 8 (100%) |
| Gentamicin | 71 (97%) | 6 (75%) |
| Penicillin | 4 (5.5%) | 0 (0%) |
| Rifampicin | 73 (100%) | 8 (100%) |
| TMP-SMX | 68 (93%) | 8 (100%) |
| Tetracycline | 73 (100%) | 7 (88%) |
| Vancomycin | 73 (100%) | 8 (100%) |
TMP-SMX = Trimethoprim-sulfamethoxazole
Initial antibiotic therapy
The 95 patients were prescribed 247 antibiotics as part of their initial therapy – a time when blood culture results were generally not known – with a median of 3 antibiotics each (IQR 2–3, range 0–5). Overall, 71/95 (74.7%) patients received at least one effective antibiotic in their initial regimen. No patients with MRSA received effective initial antibiotic therapy, compared with 71/87 (81.6%) of those with MSSA. The rates of effective initial therapy were similar between paediatric (20/25, 80%) and adult (51/70, 72.9%) cases.
The antibiotics most commonly used as part of initial antibiotic therapy were cloxacillin (n = 73, 76.8% of patients), gentamicin (53, 55.8%), ceftriaxone (37, 38.9%) and metronidazole (30, 31.6%). Only five patients received vancomycin as part of their initial treatment regimen, all of whom had MSSA infection and also received empiric cloxacillin.
Echocardiography
Echocardiography status was determined for 91 patients (95.8%). Within this assessable cohort, a total of 62 echocardiograms were performed on 55 patients (55/91 = 60.4%). There was a median of 5 days (IQR 4–7 days) between a patient's index blood culture and their first echocardiogram. Of the 36 patients who did not undergo echocardiography, 15 (41.6%) died, with a median of 4 days (IQR 1.5–12.5 days) between index culture and death. Among 70 adult SAB cases, 45 echocardiograms were performed on 40 patients (57.1%); whereas among 21 assessable paediatric SAB cases, 17 echocardiograms were performed on 15 patients (71.4%). Four patients who underwent echocardiography had vegetations visualised (4/55 = 7.3%), three adults and one child. The affected valves were mitral (n = 2), tricuspid (n = 1), as well as one further case with a vegetation seen on the interatrial septum. All patients had definite endocarditis according to modified Duke criteria,29 and all survived with a median hospital length of stay of 34 days.
Outcomes
The overall in-hospital mortality rate was 25.3% (24/95). Unadjusted mortality was higher among patients with MRSA bacteraemia (37.5%, 3/8) compared to MSSA (24.4%, 21/87). Patients with resistant infections also experienced higher median CCI scores (3 vs 2, p = 0.042) and Pitt Bacteraemia scores (1.5 vs 0, p = 0.053).
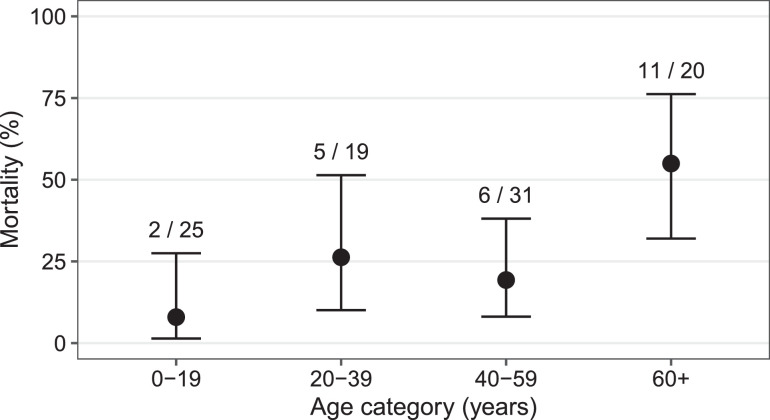
The crude in-hospital mortality rate increased with age (Figure 2), reaching 55.0% (11/20) among those aged 60 years or older, compared to only 17.3% (13/75) among the remainder. Overall, in-hospital mortality among all adults was 31.4% (22/70), versus 8.0% (2/25) among children <18 years (Relative risk 1.34, 95% CI 1.06- 1.64, p = 0.032). Both paediatric deaths were in patients <12 months of age but neither were neonates.
Figure 2.
In-hospital mortality of Staphylococcus aureus bacteraemia by age category.
Error bars represent 95% confidence intervals, using Wilson's method.
When assessing the relationship between MRSA and mortality using multivariable logistic regression, age-adjusted CCI score was identified as a key confounder on DAG. MRSA was not associated with significantly increased odds of dying in hospital (OR 1.32, p = 0.73), however higher CCI score was associated with increased mortality (OR 1.52, p = 0.001). Among the MSSA cohort, when assessing the impact of effective initial therapy on mortality, high Pitt score (≥4) was deemed a key confounder on DAG. Effective initial therapy did not significantly alter the odds of in-hospital mortality (OR 3.18, p = 0.21), whereas high Pitt score was associated with increased odds of dying in hospital (OR 17.1, p = 0.0002). For both models above, there was evidence of over-parameterisation when further potential confounders were added.
Discussion
Our research demonstrates that central Fiji has one of the highest reported incidence rates of SAB globally (68.8 cases per 100,000 population per year), which is associated with a significant in-hospital mortality rate over 25%.
Globally, most estimates of the population incidence of SAB come from high-income countries, ranging from 15 to 40 cases per 100,000 person-years.30,31 Estimates from LMICs have been far less frequent, often focus exclusively on paediatric populations, and potentially suffer from under ascertainment due to lower rates of microbiological testing.32 A Thai study of all age groups reported a SAB incidence of 7.3 per 100,000 person-years in two rural provinces,33 while two African studies in children reported incidences of 27 and 48 cases per 100 000 person-years in Kenya and Mozambique, respectively.34,35 Our observed rate of 68.8 cases per 100 000 person-years is higher than the 50.1 per 100 000 person-years reported by an earlier study of SAB cases in Suva over 2006–2007,18 and reinforces that Fiji has one of the highest global rates of SAB. This is likely driven by Fiji's high rates of both diabetes (almost half of all adults in our study) and concomitant skin diseases.16 One of the closest reported rates of SAB from the literature comes from Northern Australia, another tropical setting, with an incidence of 65 per 100,000 person-years among a cohort with high rates of diabetes (21.7%) and socioeconomic disadvantage.36
We determined a healthcare-associated SAB (HA-SAB) rate at CWMH of 2.3 cases per 10,000 patient days. HA-SAB is one of the commonest causes of nosocomial bacteraemia 37 and is associated with high mortality, however there are proven interventions – such as hand hygiene, or improved management of intravascular catheters – that can lower the HA-SAB rate.38 Although a meta-analysis suggested that healthcare-associated infections (HAIs) are 2–3 times more common in LMICs compared to high-income countries,10 a recent point-prevalence survey of HAIs in Fiji found a relatively low overall prevalence of HAIs at CMWH of 8.7%,21 slightly below estimates from similar studies in both Australia39 and Singapore.40 Despite this apparent low overall rate of HAIs, CWMH's HA-SAB rate of 2.3 per 10,000 patients days is higher than that seen in equivalent large hospitals in Australia (0.96 per 10,000 patient days)41 or in England (1.04 per 10,000 patient days).42 HA-SABs may represent a prime candidate for proven, targeted, infection prevention and control interventions at CWMH.
We observed an all-cause in-hospital mortality rate of 25.3% for all SABs in Fiji, rising to 31.4% among adult patients with SAB. This is slightly higher than rates of around 20% reported by high-income countries,2,43 however is similar to most other studies from LMICs which range from 27 to 33%.5,44 Interestingly, a case series from Thailand – which might have been expected to provide the closest comparison to Fiji as another tropical, upper-middle-income country – reported a far higher proportion of patients (40%) either dying in-hospital or being discharged moribund, and a 12-week all-cause mortality rate over 50%.4 Our finding of a significantly increased risk of death with rising age is consistent with the published literature from both high-income and LMIC settings.4,43
While the proportion of MRSA isolates in our study was relatively low at 8.4%, this is higher than the rate of only 2.3% from 128 consecutive SABs at CWMH 15 years ago.18 Reported rates of MRSA vary widely between Pacific Island countries, from very low rates of 0% and 12% in Vanuatu and Cook Islands, respectively,45,46 through to 30–42% in Kiribati, Tonga and Samoa.47, 48, 49 Patients with MRSA bacteraemia experienced a higher – but not statistically significant – crude mortality. This may be due to none of these patients receiving effective empiric antibiotics. Because of low numbers of MRSA cases we could not perform logistic regression to correct for confounders associated with both methicillin-resistance and mortality. We note that other studies from LMICs have found conflicting results regarding the impact of ineffective empiric therapy for S. aureus: in Thailand ineffective initial therapy was significantly associated with increased mortality on logistic regression,4 whereas such an association was not evident in a large multinational study across South America.50
Just over half of our assessable cohort underwent an echocardiogram; endocarditis was seen infrequently (7.3%, n = 4) with none of these patients dying. This contrasts with reported rates of endocarditis following SAB of up to 25%,51 with accompanying in-hospital mortality of 20–30%.52 Our prevalence could be falsely low in part due to the absence of transoesophageal echocardiography at CWMH, which has a higher sensitivity. Additionally, the low observed prevalence and mortality of endocarditis may be related to survivor bias, with healthier patients more likely to survive long enough to undergo an echocardiogram (a median of 5 days after their index culture). Other cohort studies from LMICs have demonstrated similarly low rates of endocarditis following SAB – between 7 and 14% – with under-ascertainment felt to be likely.4,5
Although our research was conducted during the coronavirus disease 2019 (COVID-19) pandemic, across the entire study period there was no community transmission of SARS-CoV-2 in Fiji and very few restrictions beyond the closure of the international border. We therefore feel our findings are representative of the outcomes and incidence of SAB during a non-pandemic period.
There are some limitations to our research. First, this single-centre analysis was from Fiji's largest hospital, located in the Central Division, and may not be representative of other hospitals or Divisions in Fiji. We do not have clinical or outcome data on SABs who were not admitted to CWMH within 48 h of their index culture, so cannot assess how this cohort might differ from the inpatient cohort presented above. Second, for pragmatic reasons, chart review occurred at a single time point shortly after index blood culture – this precluded data collection on antibiotic treatment duration, procedures for source control or presence of metastatic infection. All of these are important factors in determining SAB outcome, which could not subsequently be corrected for in our analyses. Third, our multivariable regression should be interpreted with caution as we were unable to include all variables suggested by the DAG process, due to small sample size and concerns regarding over-parameterisation. Finally, our measurement of SAB incidence may be a slight overestimate if there were further cases of CoNS incorrectly identified as S. aureus among patients with no isolates for confirmatory testing. However, conversely, our measurement could instead be an underestimate if there were further SAB cases that went undiagnosed, were misdiagnosed as CoNS, or were detected by one of the few small microbiology laboratories in Central Division outside of CWMH.
Conducting detailed, prospective, clinical and laboratory research in low- and middle-income countries is challenging, particularly during a global pandemic. Despite the limitations outlined above, our research contributes valuable new knowledge about SAB from a tropical, middle-income country. We have shown that central Fiji has one of the highest rates of SAB in the world. Based on the existing literature and our own new research, we believe efforts to reduce SAB in Fiji should focus on the prevention of both diabetes and skin diseases at the community level, and the optimisation of infection prevention activities at the hospital level.
Contributors
MJL, AJS, AWJJ, AYC, RN, ER and AYP conceived, designed and initiated study. MJL, AJS and AYC performed statistical analysis. ER, RN, LT, AP, TV, and VP provided technical and logistical advice. RN and LT coordinated and supervised the Fiji study staff performing data collection. TY-S and SW performed ward data collection. AP, TV and VP undertook all Suva laboratory work and data collection. JW, LB, SB, EvG and ZB undertook all Melbourne laboratory work and data collection. MJL wrote the first draft, prepared figures and tables, and made subsequent revisions based on co-authors’ feedback. All authors contributed to subsequent drafts and have all read and agreed to the published version of the manuscript.
Data sharing statement
A non-identifiable copy of the dataset will be loaded onto https://bridges.monash.edu.
Funding
MJL, AJS, and AYP are supported by Australian National Health and Medical Research Council Postgraduate Scholarship (APP1169220), Early Career Fellowship (GNT1141398) and Practitioner Fellowship (APP1117940), respectively.
The GRAM (Global Research on Antimicrobial Resistance) Project at Oxford University provided funding to support data collection in Suva. The GRAM project has been funded by the United Kingdom's Department of Health and Social Care, Fleming Fund, the Wellcome Trust (209142/Z/17/Z), and the Bill and Melinda Gates Foundation (OPP1176062).
Declaration of interests
The authors have no relevant conflicts of interest to declare.
Acknowledgments
We gratefully acknowledge the assistance of Maryanne Kora'ai from the CWMH Paediatric Department, and Sukafa Matanaicake from the CWMH Cardiology Department, who supported echocardiography data collection.
Footnotes
Supplementary material associated with this article can be found in the online version at doi:10.1016/j.lanwpc.2022.100438.
Appendix. Supplementary materials
References
- 1.Kern W.V., Rieg S. Burden of bacterial bloodstream infection-a brief update on epidemiology and significance of multidrug-resistant pathogens. Clin Microbiol Infect. 2020;26(2):151–157. doi: 10.1016/j.cmi.2019.10.031. [DOI] [PubMed] [Google Scholar]
- 2.Anantha R.V., Jegatheswaran J., Pepe D.L., et al. Risk factors for mortality among patients with Staphylococcus aureus bacteremia: a single-centre retrospective cohort study. CMAJ Open. 2014;2(4):E352–E359. doi: 10.9778/cmajo.20140018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kaasch A.J., Barlow G., Edgeworth J.D., et al. Staphylococcus aureus bloodstream infection: a pooled analysis of five prospective, observational studies. J Infect. 2014;68(3):242–251. doi: 10.1016/j.jinf.2013.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nickerson E.K., Hongsuwan M., Limmathurotsakul D., et al. Staphylococcus aureus bacteraemia in a tropical setting: patient outcome and impact of antibiotic resistance. PLoS One. 2009;4(1):e4308. doi: 10.1371/journal.pone.0004308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Eshwara V.K., Munim F., Tellapragada C., et al. Staphylococcus aureus bacteremia in an Indian tertiary care hospital: observational study on clinical epidemiology, resistance characteristics, and carriage of the Panton-valentine leukocidin gene. Int J Infect Dis. 2013;17(11):e1051–e1055. doi: 10.1016/j.ijid.2013.06.002. [DOI] [PubMed] [Google Scholar]
- 6.World Health Organization . World Health Organization; Geneva: 2017. Prioritization of Pathogens to Guide Discovery, Research and Development of New Antibiotics for Drug-Resistant Bacterial Infections, Including Tuberculosis. [Google Scholar]
- 7.Nickerson E.K., West T.E., Day N.P., Peacock S.J. Staphylococcus aureus disease and drug resistance in resource-limited countries in south and east Asia. Lancet Infect Dis. 2009;9(2):130–135. doi: 10.1016/S1473-3099(09)70022-2. [DOI] [PubMed] [Google Scholar]
- 8.World Health Organization . World Health Organization; Geneva: 2018. Global Antimicrobial Resistance Surveillance System (GLASS) Report: Early Implementation 2017-2018. [Google Scholar]
- 9.Magill S.S., O'Leary E., Janelle S.J., et al. Changes in Prevalence of health care-associated infections in U.S. hospitals. N Engl J Med. 2018;379(18):1732–1744. doi: 10.1056/NEJMoa1801550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Allegranzi B., Bagheri Nejad S., Combescure C., et al. Burden of endemic health-care-associated infection in developing countries: systematic review and meta-analysis. Lancet. 2011;377(9761):228–241. doi: 10.1016/S0140-6736(10)61458-4. [DOI] [PubMed] [Google Scholar]
- 11.Collignon P.J., Wilkinson I.J., Gilbert G.L., Grayson M.L., Whitby R.M. Health care-associated Staphylococcus aureus bloodstream infections: a clinical quality indicator for all hospitals. Med J Aust. 2006;184(8):404–406. doi: 10.5694/j.1326-5377.2006.tb00291.x. [DOI] [PubMed] [Google Scholar]
- 12.Barnett A.G., Page K., Campbell M., et al. Changes in healthcare-associated infections after the introduction of a national hand hygiene initiative. Healthc Infect. 2014;19(4):128–134. doi: 10.1086/677160. [DOI] [PubMed] [Google Scholar]
- 13.Rhodes D., Cheng A.C., McLellan S., et al. Reducing Staphylococcus aureus bloodstream infections associated with peripheral intravenous cannulae: successful implementation of a care bundle at a large Australian health service. J Hosp Infect. 2016;94(1):86–91. doi: 10.1016/j.jhin.2016.05.020. [DOI] [PubMed] [Google Scholar]
- 14.Loftus M.J., Guitart C., Tartari E., et al. Hand hygiene in low- and middle-income countries. Int J Infect Dis. 2019;86:25–30. doi: 10.1016/j.ijid.2019.06.002. [DOI] [PubMed] [Google Scholar]
- 15.Ista E., van der Hoven B., Kornelisse R.F., et al. Effectiveness of insertion and maintenance bundles to prevent central-line-associated bloodstream infections in critically ill patients of all ages: a systematic review and meta-analysis. Lancet Infect Dis. 2016;16(6):724–734. doi: 10.1016/S1473-3099(15)00409-0. [DOI] [PubMed] [Google Scholar]
- 16.Steer A.C., Jenney A.W., Kado J., et al. High burden of impetigo and scabies in a tropical country. PLoS Negl Trop Dis. 2009;3(6):e467. doi: 10.1371/journal.pntd.0000467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chan J.C., Cho N.H., Tajima N., Shaw J. Diabetes in the western pacific region-past, present and future. Diabetes Res Clin Pract. 2014;103(2):244–255. doi: 10.1016/j.diabres.2013.11.012. [DOI] [PubMed] [Google Scholar]
- 18.Jenney A., Holt D., Ritika R., et al. The clinical and molecular epidemiology of Staphylococcus aureus infections in Fiji. BMC Infect Dis. 2014;14:160. doi: 10.1186/1471-2334-14-160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tong S.Y., Steer A.C., Jenney A.W., Carapetis J.R. Community-associated methicillin-resistant Staphylococcus aureus skin infections in the tropics. Dermatol Clin. 2011;29(1):21–32. doi: 10.1016/j.det.2010.09.005. [DOI] [PubMed] [Google Scholar]
- 20.Tong S.Y., Davis J.S., Eichenberger E., Holland T.L., Fowler V.G. Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin Microbiol Rev. 2015;28(3):603–661. doi: 10.1128/CMR.00134-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Loftus M.J., Curtis S.J., Naidu R., et al. Prevalence of healthcare-associated infections and antimicrobial use among inpatients in a tertiary hospital in Fiji: a point prevalence survey. Antimicrob Resist Infect Control. 2020;9(1):146. doi: 10.1186/s13756-020-00807-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Harris P.A., Taylor R., Thielke R., Payne J., Gonzalez N., Conde J.G. Research electronic data capture (REDCap)-a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–381. doi: 10.1016/j.jbi.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.World Health Organization . World Health Organization; Geneva: 2020. GLASS Method for Estimating Attributable Mortality of Antimicrobial Resistant Bloodstream Infections. [Google Scholar]
- 24.Friedman N.D., Kaye K.S., Stout J.E., et al. Health care–associated bloodstream infections in adults: a reason to change the accepted definition of community-acquired infections. Ann Intern Med. 2002;137(10):791–797. doi: 10.7326/0003-4819-137-10-200211190-00007. [DOI] [PubMed] [Google Scholar]
- 25.Fiji Ministry of Health and Medical Services . 4th ed. Fiji Ministry of Health and Medical Services; 2019. Fiji Antibiotic Guidelines. [Google Scholar]
- 26.Clinical and Laboratory Standards Institute . 30th ed. Clinical and Laboratory Standards Institute; Wayne, PA: 2020. Performance Standards for Antimicrobial Susceptibility Testing. CLSI M100. [Google Scholar]
- 27.Fiji Bureau of Statistics. 2017 Fiji population and housing census [Internet]. Suva: Fiji Government, Bureau of Statistics; 2018 [cited 19 August 2021]. Available from: http://arks.princeton.edu/ark:/88435/dsp01n583xx76v.
- 28.Population Projections. In: Pacific Data Hub [database on the Internet]. Statistics for Development Division, Pacific Community [cited 19 August 2021]. Available from: https://sdd.spc.int/topic/population.
- 29.Li J.S., Sexton D.J., Mick N., et al. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis. 2000;30(4):633–638. doi: 10.1086/313753. [DOI] [PubMed] [Google Scholar]
- 30.Laupland K.B., Lyytikäinen O., Søgaard M., et al. The changing epidemiology of Staphylococcus aureus bloodstream infection: a multinational population-based surveillance study. Clin Microbiol Infect. 2013;19(5):465–471. doi: 10.1111/j.1469-0691.2012.03903.x. [DOI] [PubMed] [Google Scholar]
- 31.El Atrouni W.I., Knoll B.M., Lahr B.D., Eckel-Passow J.E., Sia I.G., Baddour L.M. Temporal trends in the incidence of Staphylococcus aureus bacteremia in Olmsted County, Minnesota, 1998 to 2005: a population-based study. Clin Infect Dis. 2009;49(12):e130–e138. doi: 10.1086/648442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wilson M.L., Fleming K.A., Kuti M.A., Looi L.M., Lago N., Ru K. Access to pathology and laboratory medicine services: a crucial gap. Lancet. 2018;391(10133):1927–1938. doi: 10.1016/S0140-6736(18)30458-6. [DOI] [PubMed] [Google Scholar]
- 33.Jaganath D., Jorakate P., Makprasert S., et al. Staphylococcus aureus bacteremia incidence and methicillin resistance in rural Thailand, 2006–2014. Am J Trop Med Hyg. 2018;99(1):155–163. doi: 10.4269/ajtmh.17-0631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Berkley J.A., Lowe B.S., Mwangi I., et al. Bacteremia among children admitted to a rural hospital in Kenya. N Engl J Med. 2005;352(1):39–47. doi: 10.1056/NEJMoa040275. [DOI] [PubMed] [Google Scholar]
- 35.Sigaúque B., Roca A., Mandomando I., et al. Community-acquired bacteremia among children admitted to a rural hospital in Mozambique. Pediatr Infect Dis J. 2009;28(2):108–113. doi: 10.1097/INF.0b013e318187a87d. [DOI] [PubMed] [Google Scholar]
- 36.Tong S.Y., Bishop E.J., Lilliebridge R.A., et al. Community-associated strains of methicillin-resistant Staphylococcus aureus and methicillin-susceptible S. aureus in indigenous Northern Australia: epidemiology and outcomes. J Infect Dis. 2009;199(10):1461–1470. doi: 10.1086/598218. [DOI] [PubMed] [Google Scholar]
- 37.Wisplinghoff H., Bischoff T., Tallent S.M., Seifert H., Wenzel R.P., Edmond M.B. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis. 2004;39(3):309–317. doi: 10.1086/421946. [DOI] [PubMed] [Google Scholar]
- 38.Mitchell B.G., Collignon P.J., McCann R., Wilkinson I.J., Wells A. A major reduction in hospital-onset Staphylococcus aureus bacteremia in Australia-12 years of progress: an observational study. Clin Infect Dis. 2014;59(7):969–975. doi: 10.1093/cid/ciu508. [DOI] [PubMed] [Google Scholar]
- 39.Russo P.L., Stewardson A.J., Cheng A.C., Bucknall T., Mitchell B.G. The prevalence of healthcare associated infections among adult inpatients at nineteen large Australian acute-care public hospitals: a point prevalence survey. Antimicrob Resist Infect Control. 2019;8:114. doi: 10.1186/s13756-019-0570-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cai Y., Venkatachalam I., Tee N.W., et al. Prevalence of healthcare-associated infections and antimicrobial use among adult inpatients in Singapore acute-care hospitals: results from the first national point prevalence survey. Clin Infect Dis. 2017;64 doi: 10.1093/cid/cix103. suppl_2S61-s7. [DOI] [PubMed] [Google Scholar]
- 41.Australian Institute of Health and Welfare. Bloodstream infections associated with hospital care 2019–20 [Internet]. Canberra: Australian Institute of Health and Welfare, 2021 [cited 12 August 2021]. Available from: https://www.aihw.gov.au/reports/health-care-quality-performance/bloodstream-infections-associated-with-hospital-ca.
- 42.Public Health England HCAI Mandatory Surveillance Team. Annual epidemiological commentary: Gram-negative bacteraemia, MRSA bacteraemia, MSSA bacteraemia and C. difficile infections [Internet]. London: Public Health England, 2021 [cited 12 August 2021]. Available from: https://www.gov.uk/government/statistics/mrsa-mssa-and-e-coli-bacteraemia-and-c-difficile-infection-annual-epidemiological-commentary?msclkid=d9560a85aa3f11ec823045e7ac9082d0.
- 43.van Hal S.J., Jensen S.O., Vaska V.L., Espedido B.A., Paterson D.L., Gosbell I.B. Predictors of mortality in Staphylococcus aureus Bacteremia. Clin Microbiol Rev. 2012;25(2):362–386. doi: 10.1128/CMR.05022-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fortuin-de Smidt M.C., Singh-Moodley A., Badat R., et al. Staphylococcus aureus bacteraemia in Gauteng academic hospitals, South Africa. Int J Infect Dis. 2015;30:41–48. doi: 10.1016/j.ijid.2014.10.011. [DOI] [PubMed] [Google Scholar]
- 45.Vanuatu Ministry of Health . Ministry of Health; Port Vila: 2018. Everts R. Antibiotic Guidelines Vanuatu 2018: Guidelines for Empiric and Targeted Antibiotic Treatment, Prophylaxis, Dosing and Allergies. [Google Scholar]
- 46.Cook Islands Ministry of Health . Ministry of Health; Rarotonga: 2018. Everts R. Antibiotic Guidelines Cook Islands 2018: Guidelines for Empiric and Targeted Antibiotic Treatment, Prophylaxis, Dosing and Allergies. [Google Scholar]
- 47.Kiribati Ministry of Health and Medical Services . Ministry of Health and Medical Services; Tarawa: 2017. Everts R. Antibiotic Guidelines Kiribati 2017: Guidelines for Empiric and Targeted Antibiotic Treatment, Prophylaxis, Dosing and Allergies. [Google Scholar]
- 48.Samoa Ministry of Health . Ministry of Health; Apia: 2016. Everts R. Antibiotic Guidelines Samoa 2016: Guidelines for Empiric and Targeted Antibiotic Treatment, Prophylaxis, Dosing and Allergies. [Google Scholar]
- 49.Tonga Ministry of Health . Ministry of Health; Nukuʿalofa: 2018. Everts R. Antibiotic Guidelines Tonga 2018: Guidelines for Empiric and Targeted Antibiotic Treatment, Prophylaxis, Dosing and Allergies. [Google Scholar]
- 50.Seas C., Garcia C., Salles M.J., et al. Staphylococcus aureus bloodstream infections in Latin America: results of a multinational prospective cohort study. J Antimicrob Chemother. 2018;73(1):212–222. doi: 10.1093/jac/dkx350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Incani A., Hair C., Purnell P., et al. Staphylococcus aureus bacteraemia: evaluation of the role of transoesophageal echocardiography in identifying clinically unsuspected endocarditis. Eur J Clin Microbiol Infect Dis. 2013;32(8):1003–1008. doi: 10.1007/s10096-013-1838-4. [DOI] [PubMed] [Google Scholar]
- 52.Asgeirsson H., Thalme A., Weiland O. Staphylococcus aureus bacteraemia and endocarditis - epidemiology and outcome: a review. Infect Dis. 2018;50(3):175–192. doi: 10.1080/23744235.2017.1392039. (Lond) [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.