Abstract
Objective:
this study aimed to evaluate and quantify this relationship using a uniquely large dataset from an international consortium of observational studies on gastric cancer, including data from 18 studies, for a total of 8,198 cases and 21,419 controls.
Methods:
A two-stage approach was used to obtain the pooled odds ratios (ORs) and the corresponding 95% confidence intervals (CIs) for coffee drinkers versus never or rare drinkers. A one-stage logistic mixed-effects model with a random intercept for each study was used to estimate the dose-response relationship. Estimates were adjusted for sex, age and the main recognized risk factors for gastric cancer.
Results:
Compared to never or rare coffee drinkers, the estimated pooled OR for coffee drinkers was 1.03 (95% CI: 0.94–1.13). When the amount of coffee intake was considered, the pooled ORs were 0.91 (95% CI: 0.81–1.03) for drinkers of 1–2 cups per day, 0.95 (95% CI: 0.82–1.10) for 3–4 cups, and 0.95 (95% CI: 0.79–1.15) for five or more cups. An OR of 1.20 (95% CI: 0.91–1.58) was found for heavy coffee drinkers (seven or more cups per day). A positive association emerged for high coffee intake (five or more cups per day) for gastric cardia cancer only.
Conclusions:
These findings better quantify the previously available evidence of the absence of a relevant association between coffee consumption and gastric cancer.
Keywords: cardia cancer, case-control study, coffee, gastric cancer, pooled analysis
Introduction
Gastric cancer is the fifth most common cause of cancer death in the world, and there were about one million (1,089,103) new cases of gastric cancer diagnosed in 2020 (Global Cancer Observatory: Cancer Today). Coffee is one of the most popular beverages worldwide, with an annual overall consumption of 1.27 kg per capita, which increased by 18.7% between 2014 and 2017 (Food and Agriculture Organization of the United Nations). Coffee is a complex mixture that includes many chemicals that can play different roles in the etiology of gastric cancer. Some substances, mainly antioxidants (such as phenolic compounds, diterpenes, melanoidins, and vitamin precursors) can have anti-cancer properties, while others can stimulate the carcinogenic process (including very small amounts of aromatic hydrocarbons and heterocyclic amines formed during the processing for the beans) (Borrelli et al., 2002,Gallus et al., 2009,Shen et al., 2015,Alicandro et al., 2017,Yu et al., 2019).
In 2018, the World Cancer Research Fund/American Institute for Research reported limited evidence for an association between the consumption of coffee and gastric cancer (World Cancer Research Fund/American Institute for Research, 2018). However, results of available studies were inconsistent. To better evaluate and quantify the association between coffee consumption and gastric cancer an individual participant pooled data analysis of gastric cancer studies included in an international consortium, the Stomach cancer Pooling (StoP) Project was carried out.
Methods
Study population
Data from the v.3.1 dataset release of the StoP Project (http://stop-project.org/) was used. This includes 34 case-control or cohort (participating through a nested case-control approach) studies for a total of about 13,500 gastric cancer cases and 32,000 controls. Detailed information on the aims and methods of the StoP Project is given elsewhere (Pelucchi et al., 2015). Principal investigators of the studies included in the StoP Project agreed to participate in the consortium by providing a signed data transfer agreement and the original dataset to the coordinating center, or by computing their own results locally (through standardized analyses) and then providing estimates for the second-stage meta-analysis to the StoP Project consortium (Finland (Cook et al., 2012) and Greece 2 (Benetou et al., 2008)). All centralized data were harmonized according to a pre-specified format. Ethical approval for the StoP Project was received by the University of Milan Review Board (reference 19/15 on January 4, 2015).
Overall, 21 studies collected data on coffee consumption. Three studies (López-Carrillo et al., 2003,Boccia et al., 2007,Pourfarzi et al., 2009) were excluded from the present analysis due to a high proportion (i.e., >60%) of missing values in coffee consumption. As such, 18 studies with data on coffee drinking conducted in Greece (Lagiou et al., 2004,Benetou et al., 2008) (two studies), Italy (Buiatti et al., 1989,La Vecchia et al., 1995,Lucenteforte et al., 2008) (three studies), Canada (Mao et al., 2002), Russia (Zaridze et al., 2000), USA (Zhang et al., 1999,Schatzkin et al., 2001,Ward et al., 2008) (three studies), Portugal (Lunet et al., 2007), Spain (Santibañez et al., 2012,Castaño-Vinyals et al., 2015) (two studies), Mexico (López-Carrillo et al., 1994,Hernández-Ramírez et al., 2009) (two studies), Brazil (Hamada et al., 2002,Nishimoto et al., 2002) (two studies), and Japan (Machida-Montani et al., 2004) were included. Out of the 18 studies, only two were cohort studies, one from USA (Schatzkin et al., 2001) and one from Greece (Benetou et al., 2008), and seven had information on decaffeinated coffee consumption: two from Italy (La Vecchia et al., 1995,Lucenteforte et al., 2008), one from Russia (Zaridze et al., 2000), two from Spain (Santibañez et al., 2012,Castaño-Vinyals et al., 2015), and two from the USA (Zhang et al., 1999,Schatzkin et al., 2001).
Coffee intake
Coffee intake was assessed through food frequency questionnaires (FFQs) that asked participants to report the amount of coffee consumed overall or according to specific types of coffee (e.g. caffeinated or decaffeinated coffee) before the gastric cancer diagnosis (for cases) or study recruitment (for controls). Coffee intake was collected either by face-to-face interview administered FFQs (in 12 studies) or by self-administered FFQs (in six studies).
Coffee consumption was expressed in standard unit of cups per day, by taking into account the number of coffee cups or times coffee was consumed or the frequency of consumption specified in each study. When coffee consumption was indicated in categories of consumption, the amount of coffee intake was converted into cups per day by considering the average number of coffee cups or times coffee was consumed reported in each category, and divided by the number of days considered. For the present analyses, the following three coffee consumption variables were considered: caffeinated coffee, decaffeinated coffee and their combined intake as total coffee. When the FFQ did not contain a specific variable for caffeinated coffee, the various types of caffeinated coffee reported separately were grouped together for the purposes of the present study. For example, espresso and cappuccino intake were considered as caffeinated coffee consumption in the Italy 2 study (Lucenteforte et al., 2008) while for the Russian study (Zaridze et al., 2000), black-instant coffee, coffee with milk and instant coffee with milk consumption were grouped together.
For total coffee consumption, never or rare coffee drinkers were defined as those who reported that they did not consume coffee at all or reported an amount of <1 cup/day, while ever coffee drinkers reported that they consumed coffee or reported an amount of ≥1 cup/day. Furthermore, for the 16 studies for which information on the amount of coffee consumed was available (i.e., all except the two studies from Greece (Lagiou et al., 2004,Benetou et al., 2008)), the consumption of caffeinated coffee and total coffee intake were categorized into the following eight categories: <1 cup/day: never or rare drinkers, ≥1 to <2 cups/day, ≥2 to <3 cups/day, ≥3 to <4 cups/day, ≥4 to <5 cups/day, ≥5 to <6 cups/day, ≥6 to <7 cups/day and ≥7 cups/day, that were also classified in four categories of drinking: <1 cup/day: never or rare drinkers, ≥1 to < 3 cups/day, ≥3 to <5 cups/day and ≥5 cups/day. For decaffeinated coffee consumption, the four following categories of drinking were defined: <1 cup/day: never or rare drinkers, ≥1 to <2 cups/day, ≥2 to < 3 cups/day and ≥3 cups/day, since decaffeinated coffee consumption was not reported as often as caffeinated coffee.
Statistical analysis
A two-stage modelling approach was adopted to estimate the pooled odds ratios (ORs) for ever coffee drinkers versus never or rare drinkers, including both the studies that provided original individual data and those that provided locally computed estimates.
First, the study-specific ORs and corresponding 95% confidence intervals (CIs) were estimated for the association between total coffee consumption (ever coffee drinkers versus never or rare drinkers) and gastric cancer, through multivariable conditional or unconditional logistic regression models, as appropriate. In the second stage, the summary (pooled) effect estimates were estimated using a random-effects model (DerSimonian and Laird, 1986).
A one-stage approach was used to evaluate the dose-response relationship between coffee consumption and gastric cancer and for stratified analyses (Burke et al., 2017) excluding two studies (Greece 1 (Lagiou et al., 2004) as the amount of coffee consumed was not reported and Greece 2 (Benetou et al., 2008) that provided locally computed estimates for the two-stage analysis only). One-stage ORs and the corresponding 95% CIs of gastric cancer were estimated across the categories of coffee consumption using generalized linear mixed effect models with a logistic link function and a random intercept for each study.
In both one-stage and two-stage approaches, never or rare coffee drinkers were used as the reference category. All models were adjusted for sex, five-year age groups (<40, 40–44, …, 70–74, ≥75), socioeconomic status (study-specific low, intermediate, high), smoking status (never, former, current low, current intermediate, current high), alcohol drinking (never, <1 drink/day, 1–3 drinks/day, ≥4 drinks/day), salt intake (study-specific low, intermediate, high), total fruit and vegetable intake (study-specific low, intermediate, high), and family history of gastric cancer. Missing values in the study-specific confounders were accounted for by either being included in the models as a separate category of each respective variable, or by being included in the lower levels of the categories when there was a low proportion missing (i.e., <1%).
For the stratified analyses, the effect of coffee drinking was explored across strata of sex, age (<65 and ≥65 years), geographical area (Europe, Asia, America), socioeconomic status (low, intermediate, high), smoking status (never smokers, former smokers, current smokers), alcohol drinking (<1 drink/day, 1–3 drinks/day, ≥4 drinks/day), total fruit and vegetable intake (low, intermediate, high), salt intake (low, intermediate, high), family history of gastric cancer (no, yes), H. pylori infection (no, yes), type of controls (hospital-based, population-based), cancer anatomical subsite (cardia, non-cardia) and histological type (intestinal, diffuse). For the strata of H. pylori infection, the Spain 2 (Santibañez et al., 2012) study was not included since the information was only available for the cases. For the stratifying variables of cancer anatomical subsite (cardia and non-cardia) and histological type (intestinal and diffuse by Lauren classification), multinomial mixed-effects models were used to estimate the ORs for each type of cancer separately. Heterogeneity between groups was assessed using the Q statistic.
The dose-response relationship was modelled using a one-stage linear random effects model with natural cubic splines and four knots at fixed percentiles of caffeinated coffee consumption (25th, 50th, 75th and 90th) (Desquilbet and Mariotti, 2010).
Results
Table 1 shows the main characteristics of the 8,198 cases of gastric cancer and 21,419 controls included in the present analysis. About 50% of cases and controls were from European studies. Compared to controls, cases were more frequently males (65.7% versus 57.4%), older (55.3% versus 48.3% ≥65 years old) and reported a low socioeconomic status more frequently (47.2% versus 37.1%). Cases were also more frequently high current smokers (7.6% versus 5.9%), heavy alcohol drinkers (14.3% versus 9.7%) and were more likely to have a first degree relative with a history of gastric cancer in (15.5% versus 6.8%) compared to controls.
Table 1.
Distribution of gastric cancer cases and controlsa according to study center, sex, age and other selected covariates in the Stomach cancer Pooling (StoP) Project consortium.
| Cases |
Controls |
|||
|---|---|---|---|---|
| N | % | N | % | |
|
| ||||
| Total | 8,198 | 100.0 | 21,419 | 100.0 |
| Study center (Reference) | ||||
| Europe | 4,191 | 51.0 | 10,470 | 48.9 |
| Greece 1 (Lagiou et al., 2004) | 110 | 1.3 | 100 | 0.5 |
| Greece 2 (Benetou et al., 2008) | 82 | 1.0 | 410 | 1.9 |
| Italy 1 (La Vecchia et al., 1995) | 769 | 9.4 | 2,081 | 9.7 |
| Italy 2 (Lucenteforte et al., 2008) | 230 | 2.8 | 547 | 2.6 |
| Italy 4 (Buiatti et al., 1989) | 1,016 | 12.4 | 1,159 | 5.4 |
| Portugal (Lunet et al., 2007) | 692 | 8.4 | 1,667 | 7.8 |
| Russia (Zaridze et al., 2000) | 450 | 5.5 | 611 | 2.9 |
| Spain 1 (Castaño-Vinyals et al., 2015) | 441 | 5.4 | 3,440 | 16.1 |
| Spain 2 (Santibañez et al., 2012) | 401 | 4.8 | 455 | 2.1 |
| Asia | ||||
| Japan 3 (Machida-Montani et al., 2004) | 153 | 1.9 | 303 | 1.4 |
| America | 3,854 | 47.0 | 10,646 | 49.7 |
| Brazil 1 (Nishimoto et al., 2002) | 226 | 2.8 | 226 | 1.1 |
| Brazil 2 (Hamada et al., 2002) | 93 | 1.1 | 186 | 0.9 |
| Canada (Mao et al., 2002) | 1,182 | 14.4 | 5,039 | 23.5 |
| Mexico 1 (Hernández-Ramírez et al., 2009) | 248 | 3.0 | 478 | 2.2 |
| Mexico 2 (López-Carrillo et al., 1994) | 220 | 2.7 | 752 | 3.5 |
| USA 1 (Zhang et al., 1999) | 132 | 1.6 | 132 | 0.6 |
| USA 3 (Ward et al., 2008) | 170 | 2.1 | 502 | 2.3 |
| USA 4 (Schatzkin et al., 2001) | 1,583 | 19.3 | 3,331 | 15.6 |
| Sex | ||||
| Male | 5,385 | 65.7 | 12,304 | 57.4 |
| Female | 2,813 | 34.3 | 9,115 | 42.6 |
| Age | ||||
| Missing | 41 | 0.5 | 18 | 0.1 |
| <40 | 240 | 2.9 | 1,462 | 6.8 |
| 40–44 | 256 | 3.1 | 1,144 | 5.3 |
| 45–49 | 458 | 5.6 | 1,549 | 7.2 |
| 50–54 | 615 | 7.5 | 1,774 | 8.3 |
| 55–59 | 885 | 10.8 | 2,161 | 10.3 |
| 60–64 | 1,167 | 14.2 | 2,943 | 13.7 |
| 65–69 | 1,626 | 19.8 | 3,779 | 17.6 |
| 70–74 | 1,698 | 20.7 | 3,672 | 17.1 |
| ≥75 | 1,212 | 14.8 | 2,917 | 13.6 |
| Socioeconomic status(study-specific) | ||||
| Missing | 184 | 2.2 | 309 | 1.5 |
| Low | 3,873 | 47.2 | 7,946 | 37.1 |
| Intermediate | 2,759 | 33.7 | 7,638 | 35.6 |
| High | 1,382 | 16.9 | 5,526 | 25.8 |
| Tobacco smoking | ||||
| Missing | 384 | 4.7 | 563 | 2.6 |
| Never | 3,092 | 37.7 | 9,094 | 42.5 |
| Former | 2,843 | 34.7 | 7,098 | 33.1 |
| Current | ||||
| Low | 512 | 6.2 | 1,603 | 7.5 |
| Intermedíate | 745 | 9.1 | 1,790 | 8.4 |
| High | 622 | 7.6 | 1,271 | 5.9 |
| Alcohol drinking | ||||
| Missing | 366 | 4.5 | 1513 | 7.1 |
| Never | 2,107 | 25.7 | 5,582 | 26.1 |
| Low (≤12 g/day) | 2,165 | 26.4 | 7,237 | 33.8 |
| Intermedíate (>12 and ≤ 47 g/day) | 2,388 | 29.1 | 5,010 | 23.4 |
| High (>47 g/day) | 1,172 | 14.3 | 2,077 | 9.7 |
| History of gastric cancer in first-degree relatives b | ||||
| Missing | 828 | 17.0 | 1,714 | 15.0 |
| No | 3,296 | 67.5 | 8,922 | 78.2 |
| Yes | 759 | 15.5 | 773 | 6.8 |
| Fruit and vegetable intake (study-specific tertiles) | ||||
| Missing | 179 | 2.2 | 745 | 3.5 |
| Low | 2,616 | 31.9 | 6,244 | 29.2 |
| Intermediate | 2,620 | 32.0 | 7,034 | 32.8 |
| High | 2,783 | 33.9 | 7,396 | 34.5 |
| Salt intake (study-specific tertiles) c | ||||
| Missing | 159 | 2.3 | 997 | 5.0 |
| Low | 2,794 | 40.0 | 7,501 | 38.0 |
| Intermediate | 2,221 | 31.8 | 6,192 | 31.4 |
| High | 1,816 | 26.0 | 5,060 | 25.6 |
Percentages may not add to 100% due to rounding.
The studies Canada (Mao et al., 2002), Greece 2 (Benetou et al., 2008), Mexico 1 (Hernández-Ramírez et al., 2009), Mexico 2 (López-Carrillo et al., 1994) and USA 4 (Schatzkin et al., 2001) were not included as they did not collect data on family history of gastric cancer.
The studies Greece 1 (Lagiou et al., 2004), Greece 2 (Benetou et al., 2008), and Italy 4 (Buiatti et al., 1989) were not included as they did not collect data on salt intake.
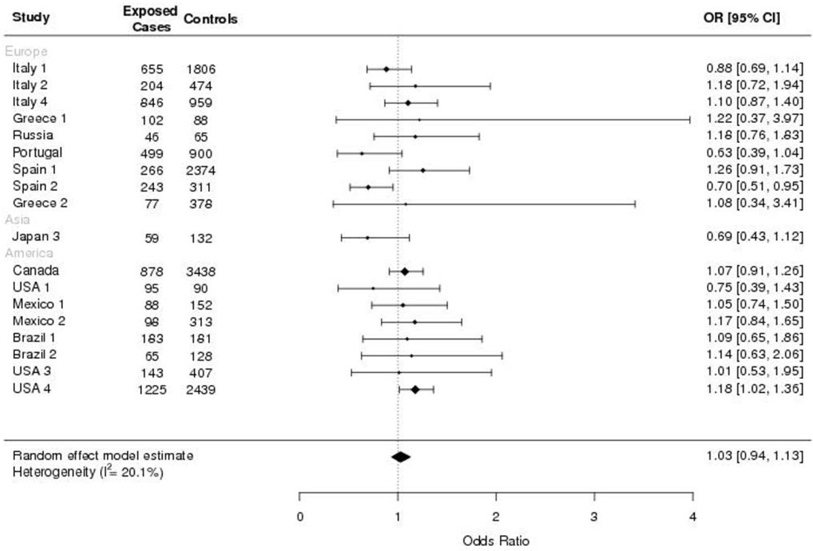
The study-specific and summary (pooled) ORs for gastric cancer, from the two-stage approach, according to total coffee drinking (drinkers versus never or rare drinkers) are presented in Figure 1. No association between total coffee consumption and gastric cancer risk was observed (OR=1.03, 95% CI: 0.94–1.13).
Figure 1.
Study-specific and two-stage pooled odds ratios (ORs) and corresponding 95% confidence intervals (CIs) of gastric cancer for total coffee drinkers compared with never or rare drinkers in the Stomach cancer Pooling (StoP) Project consortium.
The distribution of cases and controls according to the reported amounts for caffeinated, decaffeinated and total coffee consumption are presented in Table 2. About 63% of cases and 62% of controls reported a consumption of ≥1 cup per day of caffeinated coffee, and about 70% of cases and 68% of controls reported a consumption of ≥1 cup per day of total coffee. Compared with never or rare drinkers, the one-staged pooled ORs were 1.20 (95% CI: 0.91–1.58) and 1.01 (95% CI: 0.78–1.31) for ≥7 cups per day of caffeinated and total coffee, respectively. Data on decaffeinated coffee consumption were available for a total of seven studies, and approximately 15% of gastric cancer cases and 19% of controls reported drinking decaffeinated coffee. The pooled OR was 1.19 (95% CI: 0.76–1.85) for decaffeinated coffee consumers of ≥3 cups per day, compared to never or rare drinkers.
Table 2.
Distribution of gastric cancer cases and controlsa according to coffee consumption (in cups/day) categories, and one-stage pooled odds ratios (ORs) and corresponding 95% confidence intervals (CIs) for gastric cancer in the Stomach cancer Pooling (StoP) Project consortium.
| Cases |
Controls |
||||
|---|---|---|---|---|---|
| N | % | N | % | OR (CI 95%)b | |
|
| |||||
| Caffeinated coffee | 8,006 | 20,909 | |||
| Never/rarely | 2,726 | 34.0 | 6,753 | 32.3 | 1 |
| 1 | 1,441 | 18.0 | 3,752 | 17.9 | 0.84 (0.73–0.95) |
| 2 | 1,874 | 23.4 | 5,125 | 24.5 | 0.91 (0.80–1.04) |
| 3 | 582 | 7.3 | 1,345 | 6.4 | 0.87 (0.74–1.03) |
| 4 | 608 | 7.6 | 1,590 | 7.6 | 0.87 (0.71–1.07) |
| 5 | 111 | 1.4 | 281 | 1.3 | 0.95 (0.72–1.25) |
| 6 | 215 | 2.7 | 513 | 2.5 | 0.94 (0.68–1.31) |
| ≥7 | 172 | 2.1 | 318 | 1.5 | 1.20 (0.91–1.58) |
| Missing | 277 | 3.5 | 1,232 | 5.9 | |
| Decaffeinated coffee c | 4,006 | 10,597 | |||
| Never/rarely | 3,274 | 81.7 | 8,227 | 77.6 | 1 |
| 1 | 262 | 6.5 | 989 | 9.3 | 0.85 (0.69–1.05) |
| 2 | 252 | 6.3 | 717 | 6.8 | 1.19 (0.89–1.60) |
| ≥3 | 101 | 2.5 | 258 | 2.4 | 1.19 (0.76–1.85) |
| Missing | 117 | 2.9 | 406 | 3.8 | |
| Total coffee | 8,006 | 20,909 | |||
| Never/rarely | 2,128 | 26.6 | 5,462 | 26.1 | 1 |
| 1 | 1,615 | 20.2 | 3,901 | 18.7 | 0.88 (0.77–1.01) |
| 2 | 2,112 | 26.4 | 5,673 | 27.1 | 0.94 (0.82–1.08) |
| 3 | 629 | 7.9 | 1,433 | 6.9 | 0.96 (0.81–1.13) |
| 4 | 698 | 8.7 | 1,811 | 8.7 | 0.93 (0.76–1.14) |
| 5 | 121 | 1.5 | 353 | 1.7 | 0.96 (0.74–1.25) |
| 6 | 223 | 2.8 | 568 | 2.7 | 0.88 (0.64–1.20) |
| ≥7 | 195 | 2.4 | 430 | 2.1 | 1.01 (0.78–1.31) |
| Missing | 285 | 3.6 | 1,278 | 6.1 | |
Percentages may not add to 100% due to rounding.
One-stage pooled ORs were using mixed effects model adjusted for sex, age category, social class, smoking status, salt intake, fruit intake and vegetable, alcohol intake and family history of gastric cancer.
Information on decaffeinated coffee consumption was available for the studies Italy 1 (La Vecchia et al., 1995), Italy 2 (Lucenteforte et al., 2008), Russia (Zaridze et al., 2000), Spain 1 (Castaño-Vinyals et al., 2015), Spain 2 (Santibañez et al., 2012), USA 1 (Zhang et al., 1999) and USA 4 (Schatzkin et al., 2001).
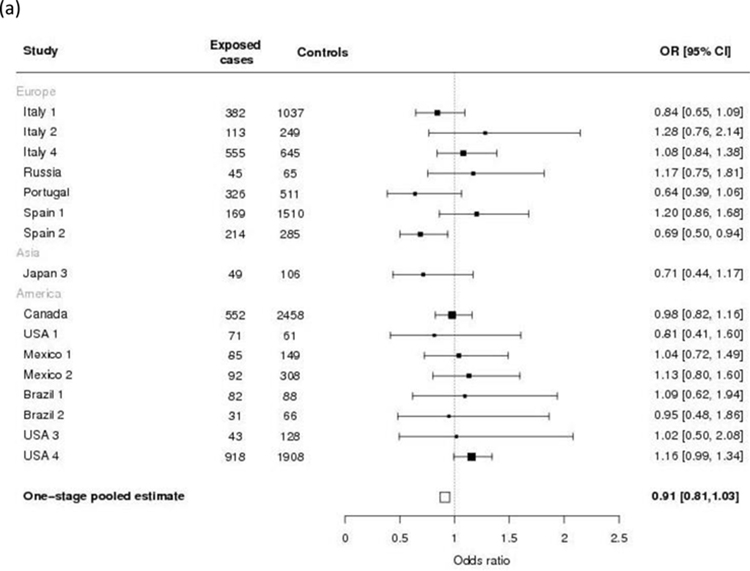
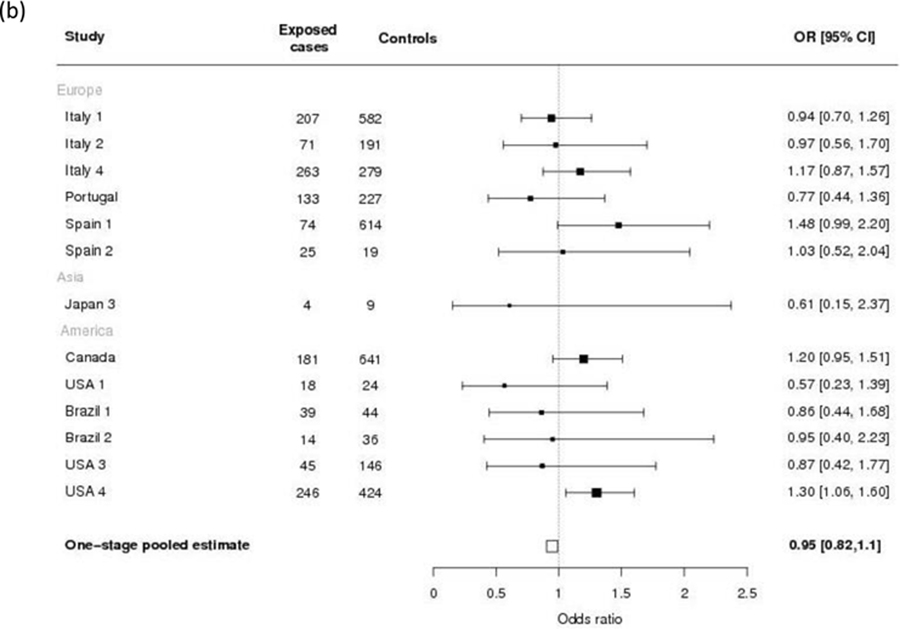
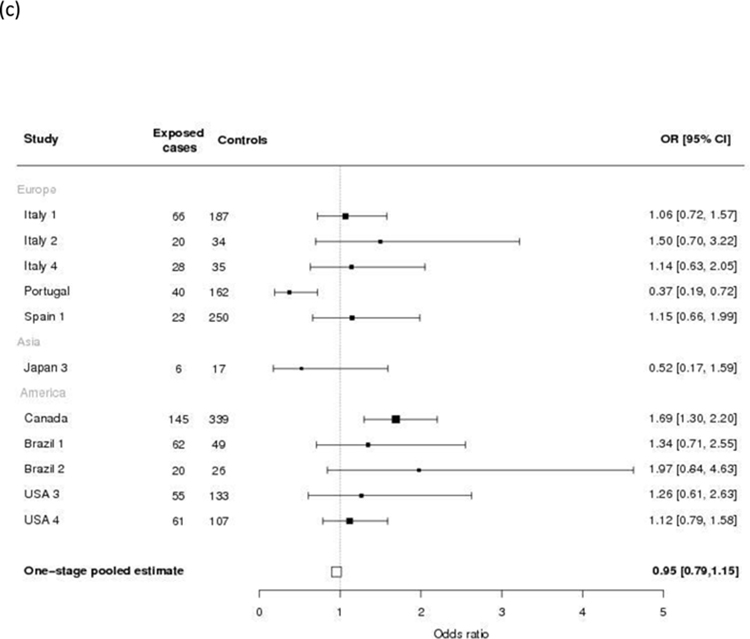
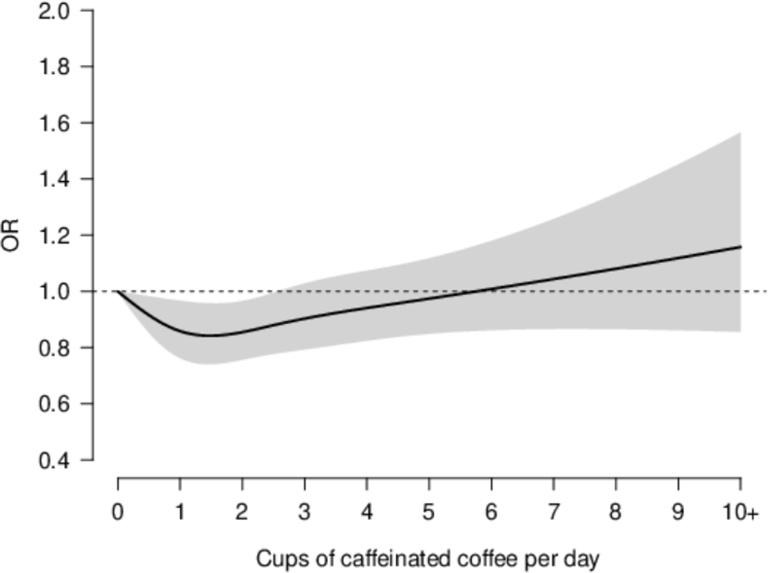
Figure 2 shows the forest plots of one-stage adjusted pooled ORs for gastric cancer according to the levels of total coffee drinking. There was no consistent association between levels of total coffee consumption and gastric cancer compared with never or rare coffee drinkers. Compared to never or rare coffee drinkers (the reference category), the pooled OR estimates were 0.91 (95% CI: 0.81–1.03) for light coffee drinkers (1–2 cups/day, Panel a), 0.95 (95% CI: 0.82–1.10) for moderate coffee drinkers (3–4 cups/day, Panel b), and 0.95 (95% CI: 0.79–1.15) for high coffee drinkers (≥ 5 cups/day, Panel c). Figure 3 shows the dose-response relationship, fitted by natural cubic splines, between consumption of caffeinated coffee and gastric cancer.
Figure 2.
Study-specific and one-stage pooled odds ratios (ORs) and corresponding 95% confidence intervals (CIs) of gastric cancer for total coffee drinkers of 1–2 cups per day (a), 3–4 cups per day (b) and ≥ 5 cups per day (c) compared with never or rare drinkers in the Stomach cancer Pooling (StoP) Project consortium. (Studies with more than five subjects in exposed cases or controls are shown in figures (b) and (c)).
Figure 3.
Dose-response relationship between caffeinated coffee consumption and gastric cancer (odds ratios, ORs, and corresponding 95% confidence intervals, CIs) fitted by natural cubic splines in one-stage linear random effects model in the Stomach cancer Pooling (StoP) Project consortium.
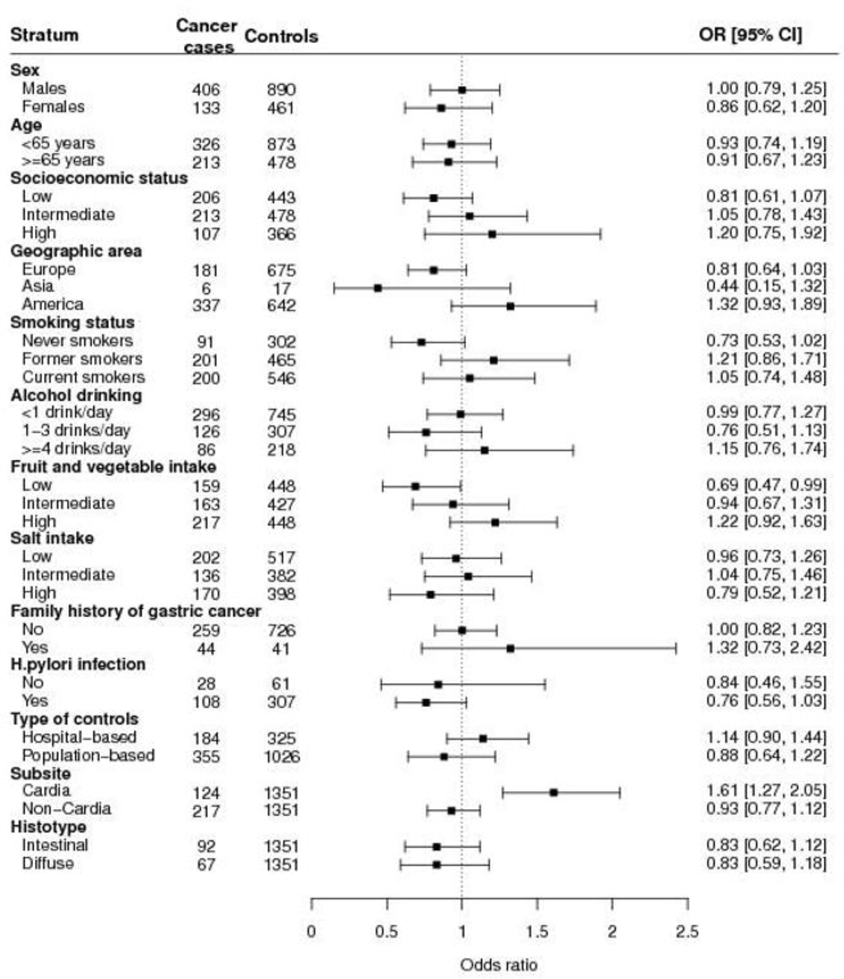
The results from the one-stage stratified analysis, according to levels of total coffee consumption are presented in Table 3 and Figure 4. Subgroup analyses by sex, age, socioeconomic status, alcohol drinking, salt intake, family history of gastric cancer, H. pylori infection, type of controls and histotype showed no heterogeneity. Heterogeneity was evident across categories of geographic area of the studies (Q=7.00, p<0.01), smoking status (Q= 4.83, p=0.03), fruit and vegetable intake (Q=5.58, p=0.02), and subsite of gastric cancer (Q=12.60, p<0.001). A positive association emerged for gastric cardia cancer (OR 1.61, 95% CI: 1.27–2.05) with a high consumption of total coffee (≥5 cups/day), while no association was found for non-cardia gastric cancer (OR=0.93, 95% CI: 0.77–1.12).
Table 3.
One-staged pooled odds ratios (ORs) and 95% confidence intervals (CIs) of gastric cancer according to caffeinated coffee consumption in strata of sex, age, socioeconomic status, geographic area, smoking status, alcohol drinking, fruit and vegetable intake, salt intake, family history of gastric cancer, Helicobacter pylori infection, control recruitment, cancer subsite and histotype in the Stomach cancer Pooling (StoP) Project consortium.
| Never/Rare | 1–2 cups/day |
3–4 cups/day |
≥5 cups/day |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ca; Co | Ca; Co | ORa (95% CI) | Q (p)b | Ca; Co | ORa (95% CI) | Q (p)b | Ca; Co | ORa (95% CI) | Q (p)b | |
|
| ||||||||||
| Overall | 2726; 6753 | 3315; 8877 | 1190; 2935 | 498; 1112 | ||||||
| Sex | 1.60 (0.21) | 3.5 (0.06) | 0.54 (0.46) | |||||||
| Men | 1273; 2993 | 2492;5567 | 0.97 (0.83–1.14) | 964; 1972 | 1.04 (0.86–1.26) | 406; 890 | 1.00 (0.79–1.25) | |||
| Women | 855; 2469 | 1235;4007 | 0.83 (0.69–1.00) | 363; 1272 | 0.77 (0.60–0.98) | 133; 461 | 0.86 (0.62–1.20) | |||
| Age | 0.62 (0.43) | 1.29 (0.26) | 0.01 (0.91) | |||||||
| <65 years | 945; 2856 | 1479; 4536 | 0.96 (0.81–1.13) | 719; 1930 | 0.99 (0.81–1.21) | 326; 871 | 0.93 (0.74–1.19) | |||
| ≥65 years | 1183; 2605 | 2248; 5032 | 0.87 (0.73–1.03) | 608; 1313 | 0.83 (0.66–1.04) | 213; 478 | 0.91 (0.67–1.23) | |||
| Socioeconomic status | 4.44 (0.04) | 0.58 (0.44) | 2.59 (0.11) | |||||||
| Low | 820; 1724 | 1844; 3564 | 0.83 (0.71–0.98) | 644; 1166 | 0.91 (0.74–1.12) | 206; 443 | 0.81 (0.61–1.07) | |||
| Intermedia | 875; 2188 | 1165; 3427 | 0.92 (0.74–1.13) | 443; 1141 | 0.96 (0.74–1.25) | 222; 526 | 1.05 (0.78–1.43) | |||
| High | 376; 1147 | 636; 2454 | 1.25 (0.88–1.79) | 225; 908 | 1.08 (0.73–1.61) | 107; 366 | 1.20 (0.75–1.92) | |||
| Geographic area | 1.70 (0.19) | 0.72 (0.40) | 7.00 (<0.01) | |||||||
| Europe | 990; 1908 | 1804; 4302 | 0.89 (0.77–1.02) | 774; 1912 | 0.97 (0.82–1.15) | 181; 675 | 0.81 (0.64–1.03) | |||
| Asia | 91; 168 | 49; 106 | 0.72 (0.44–1.18) | 4;9 | 0.66 (0.17–2.59) | 6; 17 | 0.44 (0.15–1.32) | |||
| America | 1047; 3386 | 1874; 5166 | 1.04 (0.77–1.40) | 549; 1323 | 0.85 (0.60–1.21) | 352; 659 | 1.32 (0.93–1.89) | |||
| Smoking status | 1.66 (0.20) | 1.81 (0.18) | 4.83 (0.03) | |||||||
| Never smokers | 1012; 2931 | 1423; 4029 | 0.85 (0.73–1.01) | 323; 942 | 0.85 (0.68–1.06) | 91; 302 | 0.73 (0.53–1.02) | |||
| Former smokers | 649; 1507 | 1410; 3513 | 1.03 (0.80–1.31) | 487; 1180 | 1.09 (0.81–1.45) | 201; 465 | 1.21 (0.86–1.71) | |||
| Current smokers | 385; 891 | 729; 1794 | 0.92 (0.70–1.21) | 457; 1043 | 0.98 (0.72–1.33) | 200; 546 | 1.05 (0.74–1.48) | |||
| Alcohol drinking | 0.83 (0.36) | 5.57 (0.02) | 2.10 (0.15) | |||||||
| < 1 drink/day | 1276; 3803 | 1933; 5755 | 0.89 (0.76–1.05) | 593; 1759 | 0.87 (0.71–1.06) | 296; 745 | 0.99 (0.77–1.27) | |||
| 1–3 drinks/day | 453; 890 | 1222; 2564 | 0.87 (0.68–1.11) | 478; 939 | 0.85 (0.63–1.15) | 126; 307 | 0.76 (0.51–1.13) | |||
| ≥ 4 drinks/day | 336; 497 | 477; 853 | 1.02 (0.77–1.35) | 222; 392 | 1.36 (0.97–1.90) | 86; 218 | 1.15 (0.76–1.74) | |||
| Fruit and vegetable intake | 1.19 (0.27) | 0.17 (0.68) | 5.58 (0.02) | |||||||
| Low | 624; 1615 | 1231; 2830 | 0.82 (0.65–1.04) | 460; 955 | 0.90 (0.68–1.19) | 159; 448 | 0.69 (0.47–0.99) | |||
| Intermediate | 667; 1172 | 1265; 3270 | 0.90 (0.73–1.11) | 433; 1129 | 0.92 (0.71–1.20) | 163; 427 | 0.94 (0.67–1.31) | |||
| High | 833; 2028 | 1203; 3342 | 0.97 (0.80–1.18) | 430; 1108 | 0.97 (0.76–1.25) | 217; 448 | 1.22 (0.92–1.63) | |||
| Salt intake c | 0.46 (0.50) | 1.29 (0.26) | 1.05 (0.31) | |||||||
| Low | 792; 2220 | 1318; 3420 | 0.94 (0.79–1.13) | 395;1117 | 0.86 (0.69–1.08) | 202; 530 | 0.96 (0.73–1.26) | |||
| Intermediate | 712; 1752 | 992; 2996 | 0.87 (0.71–1.08) | 353; 1010 | 0.93 (0.72–1.21) | 136; 382 | 1.04 (0.75–1.46) | |||
| High | 450; 1278 | 860; 2500 | 0.86 (0.66–1.12) | 316; 831 | 1.08 (0.78–1.49) | 170; 398 | 0.79 (0.52–1.21) | |||
| Family history of gastric cancer d | 0.08 (0.78) | 0.17 (0.68) | 0.76 (0.38) | |||||||
| No | 849; 1914 | 1349; 3746 | 0.94 (0.83–1.07) | 576; 1744 | 0.95 (0.81–1.12) | 259; 726 | 1.00 (0.82–1.23) | |||
| Yes | 191; 196 | 320; 325 | 0.89 (0.62–1.27) | 151; 158 | 1.05 (0.67–1.64) | 44; 41 | 1.32 (0.73–2.42) | |||
| H. pylori infection e | 0.07 (0.79) | 0.69 (0.41) | 0.08 (0.77) | |||||||
| No | 263; 434 | 129; 302 | 0.92 (0.66–1.29) | 27; 103 | 0.69 (0.38–1.24) | 28; 61 | 0.84 (0.46–1.55) | |||
| Yes | 469; 980 | 515; 1546 | 0.97 (0.80–1.17) | 174; 556 | 0.91 (0.70–1.19) | 108; 307 | 0.76 (0.56–1.03) | |||
| Type of controls | 0.68 (0.41) | 0.93 (0.33) | 1.64 (0.20) | |||||||
| Hospital-based | 885;1341 | 987; 1957 | 0.90 (0.78–1.04) | 379; 905 | 0.92 (0.76–1.10) | 184; 325 | 1.14 (0.90–1.44) | |||
| Population-based | 1243; 4121 | 2740; 7617 | 1.01 (0.80–1.28) | 948; 2339 | 1.08 (0.83–1.42) | 355; 1026 | 0.88 (0.64–1.22) | |||
| Subsite f | 0.93 (0.34) | 4.33 (0.04) | ||||||||
| Cardia | 365; 5462 | 721; 9574 | 1.09 (0.94–1.26) | 260; 3244 | 1.38 (1.15–1.67) | 124; 1351 | 1.61 (1.27–2.05) | 12.6 (<0.001) | ||
| Non-Cardia | 1035; 5462 | 1859; 9574 | 1.00 (0.91–1.10) | 650; 3244 | 1.09 (0.96–1.25) | 217; 1351 | 0.93 (0.77–1.12) | |||
| Histotype g | 0.03 (0.87) | 0.34 (0.56) | 0.00 (1.00) | |||||||
| Intestinal | 474; 5462 | 832; 9574 | 0.93 (0.79–1.10) | 302; 3244 | 1.04 (0.84–1.28) | 92; 1351 | 0.83 (0.62–1.12) | |||
| Diffuse | 362; 5462 | 447; 9574 | 0.91 (0.74–1.12) | 157; 3244 | 0.94 (0.72 –1.21) | 67; 1351 | 0.83 (0.59–1.18) | |||
One-stage pooled ORs were estimated using mixed effect models adjusted, where available and feasible, for sex, age category, social class, smoking status, salt intake, fruit and vegetable intake, alcohol intake and family history of gastric cancer.
p values for test of OR heterogeneity across strata.
The study Italy 4 (Buiatti et al., 1989) was not included as it did not collect data on salt intake.
The studies Canada (Mao et al., 2002), Mexico 1 (Hernández-Ramírez et al., 2009), Mexico 2 (López-Carrillo et al., 1994) and USA 4 (Schatzkin et al., 2001) were not included as they did not collect data on family history of gastric cancer.
The studies Italy 1 (La Vecchia et al., 1995), Italy 2 (Lucenteforte et al., 2008), Italy 4 (Buiatti et al., 1989), Canada (Mao et al., 2002), USA 1 (Zhang et al., 1999), Mexico 2 (López-Carrillo et al., 1994), USA 3 (Ward et al., 2008) and USA 4 (Schatzkin et al., 2001) were not included as they did not collect data on H. pylori infection. The study Spain 2 (Santibañez et al., 2012) was not included because no information on H. pylori infection was available for controls.
The studies Mexico 2 (López-Carrillo et al., 1994) and USA 3 (Ward et al., 2008) were not included as they did not collect data on cancer site.
The studies Italy 1 (La Vecchia et al., 1995), Mexico 2 (López-Carrillo et al., 1994), Japan 3 (Machida-Montani et al., 2004) and USA 3 (Ward et al., 2008) were not included as they did not collect data on histological type.
Ca: Cases, Co: Control
Figure 4.
One-stage pooled odds ratios (ORs) and corresponding 95% confidence intervals (CIs) of gastric cancer for total coffee consumption of ≥5 cups per day compared to never or rare coffee consumption, according to strata of selected variables in the Stomach cancer Pooling (StoP) Project consortium.
Discussion
The present analysis, based on 16 case-control studies and 2 cohort studies from the international StoP Project consortium, including 8,198 gastric cancer cases and 21,419 controls, found no material associations between caffeinated, decaffeinated, and total coffee consumption and gastric cancer. There was limited evidence of an inverse association for low to moderate consumption, whereas a non-significant 20% excess risk was observed for the highest level of consumption. A significant excess risk for high coffee intake emerged for gastric cardia cancer only.
Our findings are in broad agreement with previous reports. A meta-analysis (Poorolajal et al., 2020) reported an OR of 0.99 (95% CI: 0.88–1.11, based on 14 case-control studies) for ever drinkers versus non-drinkers. Results were similar when only cohort studies were considered (Fang et al., 2015,Li et al., 2015,Liu et al., 2015,Zeng et al., 2015). No significant relative risks (RR) for the highest compared with the lowest level of coffee consumption were reported, ranging from 1.13 to 1.18 (Li et al., 2015,Zeng et al., 2015), as well as for regular versus seldom coffee drinkers (RR: 1.05) (Liu et al., 2015). A few meta-analyses, which compared the highest levels of consumption with the lowest ones, found an increased risk, ranging from 1.16 to 1.24, although the highest levels of consumption varied substantially across the studies included (from two to more than seven cups per day) (Shen et al., 2015,Deng et al., 2016).
Only a few studies have investigated the relationship of coffee drinking and gastric cancer by anatomic site (cardia or non-cardia gastric cancer). These have suggested a modest excess of cardia risk of 23–50% for high coffee intake (Liu et al., 2015,Deng et al., 2016). Caffeine is a stimulant of gastric acid secretion (Schubert, 2010,Liszt et al., 2017) and coffee intake has been shown to be associated with an increased risk of gastroesophageal reflux symptoms, including heartburn and regurgitation (Mehta et al., 2020), two risk factors for cardia cancer (Derakhshan et al., 2008).
When we explored the effects of coffee consumption on gastric cancer according to geographical region, a non-significant excess risk emerged among studies from America (one of which was a prospective study (Schatzkin et al., 2001)), while an inverse association emerged among studies from Europe. This may be related to the amount and type of coffee consumed in America compared to Europe (Li et al., 2015), though chance or residual confounding may account for this apparent association. Coffee consumption varies among the geographic areas according to types of coffee, caffeine content, preparation as well as brewing methods. However, we were unable to consider these differences among the studies included due to lack information for most. Moreover, the methods used to measure coffee consumption, such as the number of cups of coffee or times coffee was consumed per day as well as the cup size, varied among the studies included.
Individuals with gastric cancer had gastritis or other gastric diseases more frequently, and patients with these conditions are often recommended to avoid or reduce their consumption of coffee. Therefore, subjects at high risk of gastric cancer may have reduced their consumption of coffee before cancer onset, thus inducing reverse causation. This could partially explain the slight inverse association of low/moderate consumption, since case-control studies collect data concerning a short period before the diagnosis. In fact, the only cohort study included in the analysis on amount of coffee consumed (Schatzkin et al., 2001) showed, if any, an increased rather than a decreased risk at low/moderate consumption levels. Nevertheless, the consistency of results between type of controls (population and hospital) strengthens the reliability of our findings.
The main strength of this study is the uniquely large sample size as well as the availability of information on several covariates, including potential confounders, such as alcohol drinking, smoking, salt intake, fruit and vegetable intake, and family history of gastric cancer. Residual confounding by tobacco smoking is however possible since tobacco is related to gastric cancer, and heavy coffee drinkers tend to be smokers more frequently (Praud et al., 2018).
In conclusion, using a unique pool of data of studies from different geographical areas, we provided evidence on the absence of a relevant association between coffee consumption and gastric cancer.
Source of funding:
This study was supported by the Associazione Italiana per la Ricerca sul Cancro (AIRC), Project no. 21378 (Investigator Grant), and by the Italian League for the Fight Against Cancer (LILT). The authors thank the European Cancer Prevention (ECP) Organization for providing support for the StoP meetings. The Unidade de Investigação em Epidemiologia – Instituto de Saúde Pública da Universidade do Porto (EPIUnit; UIDB/04750/2020) was funded by the Foundation for Science and Technology – FCT (Portuguese Ministry of Science, Technology and Higher Education). SM was also funded by the project “NEON-PC - Neuro-oncological complications of prostate cancer: longitudinal study of cognitive decline” (POCI-01-0145-FEDER-032358; ref. PTDC/SAU-EPI/32358/2017), which is funded by FEDER through the Operational Programme competitiveness and Internationalization, and national funding from FCT. We also thank all MCC-Spain study collaborators (CIBERESP, ISCIII, ISGlobal, ICO, University of Huelva, University of Oviedo, University of Cantabria, ibs.Granada, Instituto Salud Pública de Navarra, FISABIO, Murcia Regional Health Authority and cols).
Footnotes
Conflict of interest: none declared.
References
- Alicandro G, Tavani A, La Vecchia C (2017). Coffee and cancer risk: a summary overview. Eur J Cancer Prev 26:424–432. doi: 10.1097/cej.0000000000000341. [DOI] [PubMed] [Google Scholar]
- Benetou V, Trichopoulou A, Orfanos P, Naska A, Lagiou P, Boffetta P, et al. (2008). Conformity to traditional Mediterranean diet and cancer incidence: the Greek EPIC cohort. Br J Cancer 99:191–195. doi: 10.1038/sj.bjc.6604418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boccia S, Sayed-Tabatabaei FA, Persiani R, Gianfagna F, Rausei S, Arzani D, et al. (2007). Polymorphisms in metabolic genes, their combination and interaction with tobacco smoke and alcohol consumption and risk of gastric cancer: a case-control study in an Italian population. BMC cancer 7:206–206. doi: 10.1186/1471-2407-7-206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borrelli RC, Visconti A, Mennella C, Anese M, Fogliano V (2002). Chemical characterization and antioxidant properties of coffee melanoidins. J Agric Food Chem 50:6527–6533. doi: 10.1021/jf025686o. [DOI] [PubMed] [Google Scholar]
- Buiatti E, Palli D, Decarli A, Amadori D, Avellini C, Bianchi S, et al. (1989). A case-control study of gastric cancer and diet in Italy. Int J Cancer 44:611–616. doi: 10.1002/ijc.2910440409. [DOI] [PubMed] [Google Scholar]
- Burke DL, Ensor J, Riley RD (2017). Meta-analysis using individual participant data: one-stage and two-stage approaches, and why they may differ. Stat Med 36:855–875. doi: 10.1002/sim.7141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castaño-Vinyals G, Aragonés N, Pérez-Gómez B, Martín V, Llorca J, Moreno V, et al. (2015). Population-based multicase-control study in common tumors in Spain (MCC-Spain): rationale and study design. Gac Sanit 29:308–315. doi: 10.1016/j.gaceta.2014.12.003. [DOI] [PubMed] [Google Scholar]
- Cook MB, Kamangar F, Weinstein SJ, Albanes D, Virtamo J, Taylor PR, et al. (2012). Iron in relation to gastric cancer in the Alpha-tocopherol, Beta-carotene Cancer Prevention Study. Cancer Epidemiol Biomarkers Prev 21:2033–2042. doi: 10.1158/1055-9965.epi-12-0799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng W, Yang H, Wang J, Cai J, Bai Z, Song J, et al. (2016). Coffee consumption and the risk of incident gastric cancer--A meta-analysis of prospective cohort studies. Nutr Cancer 68:40–47. doi: 10.1080/01635581.2016.1115093. [DOI] [PubMed] [Google Scholar]
- Derakhshan MH, Malekzadeh R, Watabe H, Yazdanbod A, Fyfe V, Kazemi A, et al. (2008). Combination of gastric atrophy, reflux symptoms and histological subtype indicates two distinct aetiologies of gastric cardia cancer. Gut 57:298–305. doi: 10.1136/gut.2007.137364. [DOI] [PubMed] [Google Scholar]
- DerSimonian R, Laird N (1986). Meta-analysis in clinical trials. Control Clin Trials 7:177–188. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- Desquilbet L, Mariotti F (2010). Dose-response analyses using restricted cubic spline functions in public health research. Stat Med 29:1037–1057. doi: 10.1002/sim.3841. [DOI] [PubMed] [Google Scholar]
- Fang X, Wei J, He X, An P, Wang H, Jiang L, et al. (2015). Landscape of dietary factors associated with risk of gastric cancer: A systematic review and dose-response meta-analysis of prospective cohort studies. Eur J Cancer 51:2820–2832. doi: 10.1016/j.ejca.2015.09.010. [DOI] [PubMed] [Google Scholar]
- Food and Agriculture Organization of the United Nations. Food balance sheets. Available from: http://www.fao.org/faostat/en/#data/FBS, accessed [14–01-2021].
- Gallus S, Tramacere I, Tavani A, Bosetti C, Bertuccio P, Negri E, et al. (2009). Coffee, black tea and risk of gastric cancer. Cancer Causes Control 20:1303–1308. doi: 10.1007/s10552-009-9350-y. [DOI] [PubMed] [Google Scholar]
- Global Cancer Observatory: Cancer Today. Lyon, France: International Agency for Research on Cancer. Available from: https://gco.iarc.fr/today, accessed [14–01-2021]. [Google Scholar]
- Hamada GS, Kowalski LP, Nishimoto IN, Rodrigues JJG, Iriya K, Sasazuki S, et al. (2002). Risk factors for stomach cancer in Brazil (II): a case-control study among Japanese Brazilians in São Paulo. Japanese journal of clinical oncology 32:284–290. doi: 10.1093/jjco/hyf061. [DOI] [PubMed] [Google Scholar]
- Hernández-Ramírez RU, Galván-Portillo MV, Ward MH, Agudo A, González CA, Oñate-Ocaña LF, et al. (2009). Dietary intake of polyphenols, nitrate and nitrite and gastric cancer risk in Mexico City. Int J Cancer 125:1424–1430. doi: 10.1002/ijc.24454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- La Vecchia C, D’Avanzo B, Negri E, Decarli A, Benichou J (1995). Attributable risks for stomach cancer in northern Italy. Int J Cancer 60:748–752. doi: 10.1002/ijc.2910600603. [DOI] [PubMed] [Google Scholar]
- Lagiou P, Samoli E, Lagiou A, Peterson J, Tzonou A, Dwyer J, et al. (2004). Flavonoids, vitamin C and adenocarcinoma of the stomach. Cancer Causes Control 15:67–72. doi: 10.1023/B:CACO.0000016619.18041.b0. [DOI] [PubMed] [Google Scholar]
- Li L, Gan Y, Wu C, Qu X, Sun G, Lu Z (2015). Coffee consumption and the risk of gastric cancer: a meta-analysis of prospective cohort studies. BMC Cancer 15:733. doi: 10.1186/s12885-015-1758-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liszt KI, Ley JP, Lieder B, Behrens M, Stoger V, Reiner A, et al. (2017). Caffeine induces gastric acid secretion via bitter taste signaling in gastric parietal cells. Proc Natl Acad Sci U S A 114:E6260–E6269. doi: 10.1073/pnas.1703728114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu H, Hua Y, Zheng X, Shen Z, Luo H, Tao X, et al. (2015). Effect of coffee consumption on the risk of gastric cancer: a systematic review and meta-analysis of prospective cohort studies. PLoS One 10:e0128501. doi: 10.1371/journal.pone.0128501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- López-Carrillo L, Hernández Avila M, Dubrow R (1994). Chili pepper consumption and gastric cancer in Mexico: a case-control study. Am J Epidemiol 139:263–271. doi: 10.1093/oxfordjournals.aje.a116993. [DOI] [PubMed] [Google Scholar]
- López-Carrillo L, López-Cervantes M, Robles-Díaz G, Ramírez-Espitia A, Mohar-Betancourt A, Meneses-García A, et al. (2003). Capsaicin consumption, Helicobacter pylori positivity and gastric cancer in Mexico. Int J Cancer 106:277–282. doi: 10.1002/ijc.11195. [DOI] [PubMed] [Google Scholar]
- Lucenteforte E, Scita V, Bosetti C, Bertuccio P, Negri E, La Vecchia C (2008). Food Groups and Alcoholic Beverages and the Risk of Stomach Cancer: A Case-Control Study in Italy. Nutrition and Cancer 60:577–584. doi: 10.1080/01635580802054512. [DOI] [PubMed] [Google Scholar]
- Lunet N, Valbuena C, Vieira AL, Lopes C, Lopes C, David L, et al. (2007). Fruit and vegetable consumption and gastric cancer by location and histological type: case-control and meta-analysis. Eur J Cancer Prev 16:312–327. doi: 10.1097/01.cej.0000236255.95769.22. [DOI] [PubMed] [Google Scholar]
- Machida-Montani A, Sasazuki S, Inoue M, Natsukawa S, Shaura K, Koizumi Y, et al. (2004). Association of Helicobacter pylori infection and environmental factors in non-cardia gastric cancer in Japan. Gastric Cancer 7:46–53. doi: 10.1007/s10120-004-0268-5. [DOI] [PubMed] [Google Scholar]
- Mao Y, Hu J, Semenciw R, White K, Canadian Cancer Registries Epidemiology Research G (2002). Active and passive smoking and the risk of stomach cancer, by subsite, in Canada. European journal of cancer prevention : the official journal of the European Cancer Prevention Organisation (ECP) 11:27–38. doi: 10.1097/00008469-200202000-00005. [DOI] [PubMed] [Google Scholar]
- Mehta RS, Song M, Staller K, Chan AT (2020). Association Between Beverage Intake and Incidence of Gastroesophageal Reflux Symptoms. Clin Gastroenterol Hepatol 18:2226–2233 e2224. doi: 10.1016/j.cgh.2019.11.040. [DOI] [PubMed] [Google Scholar]
- Nishimoto IN, Hamada GS, Kowalski LP, Rodrigues JG, Iriya K, Sasazuki S, et al. (2002). Risk factors for stomach cancer in Brazil (I): a case-control study among non-Japanese Brazilians in São Paulo. Jpn J Clin Oncol 32:277–283. doi: 10.1093/jjco/hyf060. [DOI] [PubMed] [Google Scholar]
- Pelucchi C, Lunet N, Boccia S, Zhang ZF, Praud D, Boffetta P, et al. (2015). The stomach cancer pooling (StoP) project: study design and presentation. Eur J Cancer Prev 24:16–23. doi: 10.1097/CEJ.0000000000000017. [DOI] [PubMed] [Google Scholar]
- Poorolajal J, Moradi L, Mohammadi Y, Cheraghi Z, Gohari-Ensaf F (2020). Risk factors for stomach cancer: a systematic review and meta-analysis. Epidemiol Health 42:e2020004. doi: 10.4178/epih.e2020004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pourfarzi F, Whelan A, Kaldor J, Malekzadeh R (2009). The role of diet and other environmental factors in the causation of gastric cancer in Iran--a population based study. Int J Cancer 125:1953–1960. doi: 10.1002/ijc.24499. [DOI] [PubMed] [Google Scholar]
- Praud D, Rota M, Pelucchi C, Bertuccio P, Rosso T, Galeone C, et al. (2018). Cigarette smoking and gastric cancer in the Stomach Cancer Pooling (StoP) Project. Eur J Cancer Prev 27:124–133. doi: 10.1097/CEJ.0000000000000290. [DOI] [PubMed] [Google Scholar]
- Santibañez M, Alguacil J, de la Hera MG, Navarrete-Muñoz EM, Llorca J, Aragonés N, et al. (2012). Occupational exposures and risk of stomach cancer by histological type. Occup Environ Med 69:268–275. doi: 10.1136/oemed-2011-100071. [DOI] [PubMed] [Google Scholar]
- Schatzkin A, Subar AF, Thompson FE, Harlan LC, Tangrea J, Hollenbeck AR, et al. (2001). Design and serendipity in establishing a large cohort with wide dietary intake distributions : the National Institutes of Health-American Association of Retired Persons Diet and Health Study. Am J Epidemiol 154:1119–1125. doi: 10.1093/aje/154.12.1119. [DOI] [PubMed] [Google Scholar]
- Schubert ML (2010). Gastric secretion. Curr Opin Gastroenterol 26:598–603. doi: 10.1097/MOG.0b013e32833f2010. [DOI] [PubMed] [Google Scholar]
- Shen Z, Liu H, Cao H (2015). Coffee consumption and risk of gastric cancer: an updated meta-analysis. Clin Res Hepatol Gastroenterol 39:245–253. doi: 10.1016/j.clinre.2014.09.005. [DOI] [PubMed] [Google Scholar]
- Ward MH, Heineman EF, Markin RS, Weisenburger DD (2008). Adenocarcinoma of the Stomach and Esophagus and Drinking Water and Dietary Sources of Nitrate and Nitrite. International Journal of Occupational and Environmental Health 14:193–197. doi: 10.1179/oeh.2008.14.3.193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Cancer Research Fund/American Institute for Research. Continuous Update Project Expert Report 2018. Non-alcoholic drinks and the risk of cancer. 2018. [Google Scholar]
- Yu EY, Wesselius A, van Osch F, Stern MC, Jiang X, Kellen E, et al. (2019). The association between coffee consumption and bladder cancer in the bladder cancer epidemiology and nutritional determinants (BLEND) international pooled study. Cancer Causes Control 30:859–870. doi: 10.1007/s10552-019-01191-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaridze D, Borisova E, Maximovitch D, Chkhikvadze V (2000). Alcohol consumption, smoking and risk of gastric cancer: case-control study from Moscow, Russia. Cancer Causes Control 11:363–371. doi: 10.1023/a:1008907924938. [DOI] [PubMed] [Google Scholar]
- Zeng SB, Weng H, Zhou M, Duan XL, Shen XF, Zeng XT (2015). Long-Term Coffee Consumption and Risk of Gastric Cancer: A PRISMA-Compliant Dose-Response Meta-Analysis of Prospective Cohort Studies. Medicine (Baltimore) 94:e1640. doi: 10.1097/md.0000000000001640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang ZF, Kurtz RC, Klimstra DS, Yu GP, Sun M, Harlap S, et al. (1999). Helicobacter pylori infection on the risk of stomach cancer and chronic atrophic gastritis. Cancer Detect Prev 23:357–367. doi: 10.1046/j.1525-1500.1999.99041.x. [DOI] [PubMed] [Google Scholar]