Abstract
Epigenetics including DNA and RNA modifications have always been the hotspot field of life sciences in the post-genome era. Since the first mapping of N6-methyladenosine (m6A) and the discovery of its widespread presence in mRNA, there are at least 160-170 RNA modifications have been discovered. These methylations occur in different RNA types, and their distribution is species-specific. 5-methylcytosine (m5C) has been found in mRNA, rRNA and tRNA of representative organisms from all kinds of species. As reversible epigenetic modifications, m5C modifications of RNA affect the fate of the modified RNA molecules and play important roles in various biological processes including RNA stability control, protein synthesis, and transcriptional regulation. Furthermore, accumulative evidence also implicates the role of RNA m5C in tumorigenesis. Here, we review the latest progresses in the biological roles of m5C modifications and how it is regulated by corresponding “writers”, “readers” and “erasers” proteins, as well as the potential molecular mechanism in tumorigenesis and cancer immunotherapy.
Keywords: Epigenetics, RNA methylation modification, 5-methylcytosine (m5C), Non-coding RNAs, cancer immunotherapy
Overview of RNA 5-methylcytosine (m5C) modification
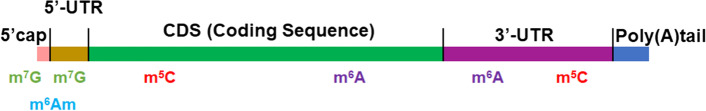
DNA and RNA modifications play crucial roles in various biological processes [1]. Compared with limited modifications detected in DNA, more than 170 modifications have been discovered in RNA including N6-methyladenosine (m6A), 5-methylcytosine (m5C) and 7-methylguanylate (m7G) (Fig. 1). They increase the complexity of the RNA species by acting on the tertiary structure, biogenesis, localization and function of RNA [2–4]. Among these modifications, mRNA N6-methyladenosine (m6A) is well acknowledged to be involved in translation, miRNA biosynthesis and gene attenuation [5, 6] (Fig. 1).
Fig. 1.
The distribution of methylation in mRNA. The preferential locations of each methylation within mRNA are shown
The discovery of DNA 5-Methylcytosine (m5C) can be traced back to the 1950s, earlier than the discovery of the double helix structure of DNA [7, 8]. In the 1970s, researchers discovered the m5C modification. Similar to DNA m5C [9], an active methyl-group from the donor, usually S-adenosyl-methionine (SAM), is added to the carbon-5 position of the cytosine base in RNA to form the m5C modification, which is likewise a widespread RNA modification detected in messenger RNA (mRNA) and non-coding RNAs including transfer RNA (tRNA), ribosomal RNA (rRNA), long non-coding RNA (lncRNA), small nuclear RNA (snRNA), micro RNA (miRNA) and enhancer RNA (eRNA) [10]. The m5C modifications have been reported in many species but its distribution seems to differ. For example, eukaryotic tRNA and mRNA have more m5C modification than bacterial mRNA and tRNA [9, 11, 12].
m5C modifications on tRNA and rRNA were extensively studied. In tRNA, m5C has been shown to maintain homeostasis, optimize codon–anticodon pairing, regulate stress response, and control translation efficiency and accuracy [13–18]. rRNA m5C modification is involved in glioma sensitivity to bioactive substrates of the stress-related enzyme NQO1 and structural stability of the tertiary rRNA–tRNA–mRNA complex under stress [19]. Until in the 1970s, researchers discovered the first m5C modification of eukaryotic mRNA, such as hamster cell mRNA and certain viral RNA but not SV40 RNA [20, 21]. Later, with the bisulfite sequencing in the whole transcripts of HeLa, a wide range of m5C modifications in mRNA and ncRNA were discovered. To date, mRNA m5C modifications have been associated with various biological processes, such as mRNA stability, splicing and nuclear–cytoplasmic shuttling [22–24]; DNA damage repair [25]; proliferation, and migration [26]; development, differentiation, and reprogramming of stem cells [27, 28]. Aberrant mRNA m5C modifications have been associated with the etiology of multiple diseases, including arteriosclerosis [29], autoimmune diseases [30] and cancer [31].
5-methylcytosine (m5C) modification in mRNA
With the development of high-throughput sequencing technology and the improvement of liquid chromatography sensitivity, the methods for the identification of the overall level of RNA methylation have been greatly developed. The current technical means used for the identification of the overall level of RNA methylation is mainly liquid chromatography coupled with MS (LC-MS). LC-MS/MS uses tandem mass spectrometry based on liquid-phase mass spectrometry, which is able to obtain both molecular and fragment ion peaks, allowing both qualitative and quantitative analysis of bases [32]. In contrast, bisulfite sequencing is a method that uses methylation to analyze different regions of DNA. In bisulfite sequencing, Bisulfite treatment can convert the unmethylated C bases in the genome into U, which becomes T after PCR amplification to distinguish them from the original C bases with methylation modifications, and then combined with high-throughput sequencing technology, a genome-wide DNA methylation map with single-base resolution can be drawn [33]. Moreover, a novel species-specific computational approach, Staem5, to accurately predict RNA m5C sites in Mus musculus and Arabidopsis thaliana was recently developed [34].
Bisulfite sequencing shows that m5C modification is another abundant mRNA modification and may be another RNA epigenetic marker [35]. Recently, enhanced liquid chromatography-mass spectrometry (LC-MS) has shown that eukaryotes do show methylation and hydroxymethylation of endocytosine. Due to the loss of information about the location of m5C modification in LC-MS, the adaptability of bisulfite sequencing with RNA opens a new possibility of mapping m5C with nucleotide resolution in RNA. Using this method, Lukas Trixl and Alexandra Lusser found ~ 10,000 sites showing > 20% methylation and mapping to ~ 8500 mRNAs resulting in a rate of 0.43% m5C of all sequenced Cs, and they also published the first cytosine methylome for human cells [36].
Recently, Amort et al. detected about 7500 m5C sites (> 20% methylation) corresponding to 1650 mRNAs in mouse embryonic stem cells and 2075 m5C sites corresponding to 486 mRNAs in mouse brain. Their concluded that m5Cs modification mainly exist in the coding region and are enriched around the translation initiation site [37]. Another study also found that m5Cs modification localized to the untranslated regions (UTRs) of mRNA transcripts [38].
Another recently published article on HeLa cells and mouse cytosine, a matrix, identified about 3600 loci in about 2000 genes in HeLa cells and 2500-4400 sites (1000-1655 genes) in different mouse tissues [37, 39]. About 100 m5C sites in Arabidopsis thaliana mRNA were detected by bisulfite sequencing [40], while another study used meRIP-seq and found that 6045 peaks correspond to 4465 expressed genes [41]. MeRIP-seq was also used to detect the level of m5C in the archaebacterium Sulfolobus solfataricus and budding yeast, and a single site in yeast and 14 m5C modified mRNAs in S. solfataricus [12] were discovered respectively.
However, the exploration of the function of m5C in mRNA is not comprehensive and thorough, while there are some interesting findings, such as Yang et al. demonstrated that during the maternal-to-zygotic transition (MZT) of zebrafish, RNA m5C modification regulates maternal mRNA stabilization, highlighting the key role of m5C mRNA modification in early development [23]. More efforts are needed to uncover the m5C pandora-box roles besides in regulating mRNA stability.
m5C modification in ncRNAs
It was found that m5C not only plays a role in regulating the stability of mRNA, but also regulating the stability of rRNA and tRNA. A large number of studies have reported that m5C modification is vital for translation regulation of tRNA and rRNA [42, 43].
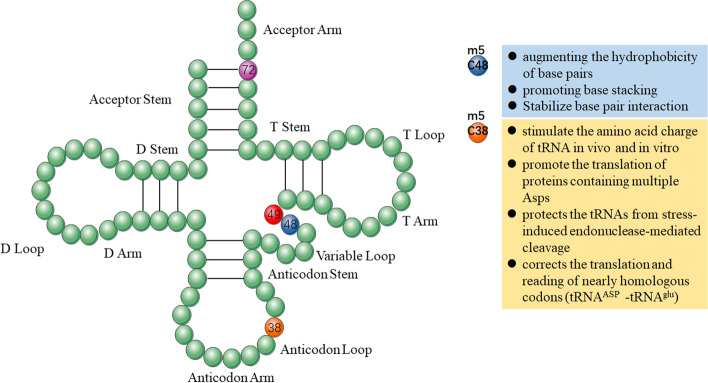
Most of the researches on m5C modification are focused on tRNA. Methylation of tRNA most often occurs on the cytosine at the junction region between the variable ring and the T-stem, as well as on one, two, or three Cs spanning the 47-50 position [44]. They participate in the composition of secondary structure, which is in connection with the codon recognition and stability of tRNA [45]. tRNA methylation mainly affects protein synthesis in mice [46]. Some studies have shown that methylation of C48 is based on augmenting the hydrophobicity of base pairs and promoting base stacking to stabilize this interaction. An unusual “Levitt pair” is formed by C48 and the nucleoside 15 on the D ring, resulting in a characteristic L-shaped three-dimensional structure [47]. Besides C48, C38 is another frequently methylated site in the anticodon loop. Studies have confirmed that methylation of C38 in mouse tRNAAsp can effectively stimulate tRNA charging Asps in vivo and in vitro, hereby promoting the translation of proteins harboring multiple Asps [48]. At the same time, C38 methylation also protects the tRNAs from stress-induced endonuclease-mediated cleavage and corrects the translation and reading of nearly homologous codons [48]. During the decoding of aspartate codons, the loss of m5C38 of tRNAMet results in a moderated translational fidelity [49]. At the same time, DNMT2-mediated m5C38 tRNAAsp can help to distinguish homologous and near homologous codons, such as different tRNAASP from tRNAglu, avoiding a false positive rate of amino acids, and improve the accuracy of translation [50]. In the late stage of tRNAThr and tRNACys transcription, the CCA sequence is added at the 3’end and thereafter C72 was confirmed to be methylated [51]. However, there has been no specific function reported about this modification. Other mitochondrial studies have shown that mitochondrial f5C modification stems from m5C34 of mt-tRNAmet, and it play a key role in mitochondrial translation as methionine in decoding unconventional AUA codons [52]. In yeast, wobble of m5C34 of tRNALeu is associated with translation regulation. In oxidative stress responses, its methylation level is significantly increased, which can enhance the translation of uug-codon-rich mRNAs, such as ribosomal protein El22A [47]. In addition, NSUN2-mediated tRNA m5C48 and m5C49 are located in the junction sequence of the V-loop (VL) and T-stem-loop (TSL), which is essential in accelerating tRNA stability and protein translation (Fig. 2).
Fig. 2.
The functions of m5C in tRNA
In rRNA, m5Cs modification in the ribosome function critical region help to keep its conformation stable. In yeast, a methylated nucleotide cluster in the fourth domain of the 25 s rRNA is critical to maintain structural stability of the 60S ribosomal subunits, whereby deletion of m5C2278 and ribose methylation of G2288 in 25 s rRNA resulted in severe instability of the 60S ribosomal subunits [53]. The modification of m5C in rRNA is associated with ribosome synthesis and protein translation. Under oxidative stress, m5C2278 of 25S rRNA in yeast which helps to maintain rRNA folding and facilitate selective recruitment and translation of mRNAs for participation in distinct cellular signals [53]. In mitochondrial ribosomes, NSUN4 is also required for mt-m5C911 12S rRNA. Depletion of NSUN4 caused functional assembly defect and disrupted mitochondrial protein translation [54]. NSUN5-mediated deletion of m5C3782 28S rRNA hinders the synthesis of whole proteins [55].
It has been reported that if cytosine-5 methylation is absent in the vault RNA, it will be abnormally processed into Argonaute-associated small RNA fragments, which have a similar function as microRNAs [56]. Although m5C is necessary for stability of vault ncRNAs, m5C is situated in lncRNA, X-inactive specific transcripts (XIST), and it prevents Polycomb repressing complex 2 (PRC2) complexes from binding in vitro [57]. Hu et al. have investigated the distribution and regulation of lncRNA in colorectal cancer in 5hmC, and they found that the hm5C is distributed in lncRNA and is positively correlated with lncRNA transcription. Studies have confirmed that hm5C directly or indirectly regulates dysregulated colorectal cancer lncRNAs by the abnormal activity of superenhancers and promoters modified by the hm5C. In addition, hm5C also participates in long-range chromatin interactions at the lncRNA sites. They also found that lncRNAs regulated by different hm5C markers were associated with different clinical outcomes and tumor status [58].
As a type of RNA modification, m5C has relatively rich but highly dynamic characteristics, and it constitutes a multifunctional and effective mechanism for coping with constantly changing internal and external environments. It acts by regulating the intracellular RNA metabolism and related functions. m5C modification is increasingly becoming mainstream in the field of epitranscriptomics.
Dynamic regulation of m5C by “writers”, “erasers”, and “readers”
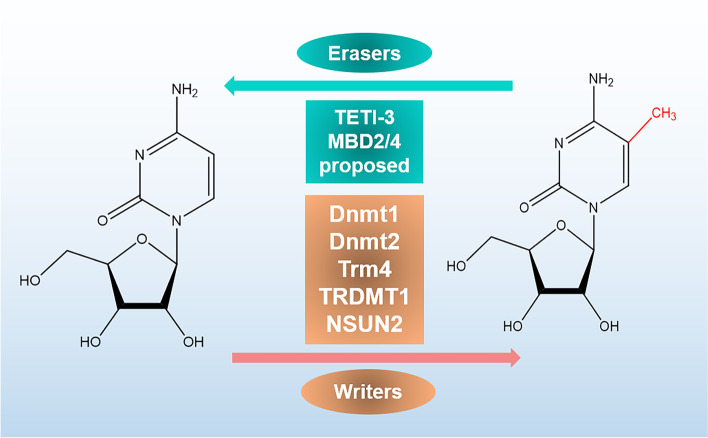
m5C modification is mainly mediated by 3 types of proteins, namely the methyltransferases (writers), the demethylases (erasers) and the binding proteins (readers) respectively. The m5Cs in RNAs are recruited by NOL1/NOP2/SUN (NSUN) protein family, including NSUN1-7 and DNA methyltransferase (DNMT) homologue DNMT2 [59], NSUN1, NSUN2, and NSUN5 are infrequently expressed in eukaryotes, whereas higher eukaryotes have high expressions of NSUN3, NSUN4, NSUN6 and NSUN7 [60](Fig. 3).
Fig. 3.
The summary of discovered writer and eraser proteins of m5C
m5C writers
The methyltransferase of m5C (RCMTs) uses adenosylmethionine as a methyl donor to form m5C by transferring the methyl group to cytosine [61]. RCMTs are believed to be the writers for catalyzing methylation of cytosine-5, including the NSUN methyltransferases and DNMT2 [62]. More than 10 types of RNA m5C methyltransferases have been found, including DNMT2, and tRNA-specific methyltransferase (TRDMT) family members, NOL1/NOP2/SUN domain (NSUN) family member. Among them, the NSUN methyltransferases includes several members (NSUN1 to NSUN7) and NSUN5a/b/c [63]. The TRDMT family includes TRM4A and TRM4B in Arabidopsis thaliana [64]. Both NSUN and DNMT family enzymes contain conserved motif IV and motif VI [65]. DNMT2 and NSUN2 have complementary target specificities [16]. A quinte essential example in the NSUN family should be cited; cysteine at the VI position of the motif in the NSUN family interferes with C6 of the target cytosine in RNA through nucleophilic attack, meanwhile, the motif IV proline and aspartate sidechain interact with the hydrogen atom, which positions the nucleobase in the active status and form bond in transient protonation. Then, the activated nucleobase accepts the SAM methyl group. Human NSUN6 forms complex with a full-length tRNA substrate and catalyze tRNA m5C modification. Liu and colleagues have solved the structures of NSUN6 [66]. A non-canonical conformation of the bound tRNA can be observed from these structures, and related enzymes methylate the base portion of the target cytosine. Further biochemical analysis revealed the key, but distinction, to two conserved cysteine residues of RNA: m5C methylation.
m5C erasers
Protein demethylases called erasers, such as the TET family of enzymes, have a reversible effect by mediating the demethylation of written RNA [67]. In recent years, quantitative analysis using LC–MS/MS/MS has shown that TET has the potential as an RNA demethylase, and its overexpression can significantly increase the level of RNA 5hmC in HEK293T cells [68]. The TET (ten-eleven translocation) proteins, TET1, TET2, and TET3, function as DNA dioxygenases catalyzing 5mC to 5hmC on DNA [69]. It has been found that oxidation of 5fC to 5caC in RNA is mediated by TET1 [70]. It is been reported that TET2 mediates the oxidation of m5C, which may inhibit the effect of 5-methylcytosine on the formation of double-stranded RNA in eukaryotes [71]. Besides, TET2 catalyzes RNA 5hmC, which involves degradation of RNA; this indicates that 5hmC plays a key role in post-transcriptional regulation [72]. In recent years, it has been revealed that the identified mitochondrial DNA and RNA dioxygenase Alpha-ketoglutarate-dependent dioxygenase ABH1 (ALKBH1) also participates in the demethylation of N1 methyladenosine (m1A) along the cytoplasmic tRNAs [73]. ALKBH1 catalyzes the anticodon modification of m5c34 to hm5cm34 (5-hydroxymethyl-20-o-methylcytidine) and f5cm34 (5-formyl-20-o-methylcytidine) along mt-tRNAmet and cytoplasmic tRNALeu [74]. Loss of ALKBH1 induce severe deficiency in mitochondrial translation and oxygen consumption, indicating that RNA m5C metabolism by ALKBH1 have great potential in regulating mitochondrial activity. Moreover, ALKBH1 can also specifically act on a histone dioxygenase-histone H2A [75].
m5C readers
Most of the biological functions of RNA modification are related to the protein to which it binds. RNA m5C binding proteins, such as ALYREF and YBX1, are considered to be a type of barcode reader, which exerts biological effects by recognizing and binding to m5C sites [63]. Recently, m5C-modified RNA oligos pull-down coupled with mass spectrometry analysis has been applied to a variety of m5C mRNAs, and two reading proteins of m5C mRNA, ALYREF (RNA and export factor binding protein 2) and YBX1, have been identified [76]. ALYREF is the first mRNA reading protein identified in the nucleus and has the key m5C recognition site of K171. m5C is recognized by the m5C reader ALYREF through the viral RNA transcript installed in the nucleus, which helps them export to the cytoplasm [31]. In order to determine whether m5C installation in viral transcripts occurred in the nucleus or the cytoplasm, Eckwahl and his colleagues performed RNAs isolation from nucleus and cytoplasm separately, followed by bisulfite treatment and Sanger sequencing; they focused their functional investigations on ALYREF reads on the protein. They found that knocking out the m5C reading protein ALYREF has significant impact on viral protein production and viral replication. The depletion of ALYREF decreased the MLV Gag protein and virus released cells by 5-fold. On the contrary, the overexpression of ALYREF increased the Gag protein level. They also examined whether using the plasmid encoding wild- type ALYREF or the known m5C to recognize the impaired ALYREF mutant (K171A) could rescue the 3 T3 cells lacking endogenous ALYREF. They found that wild- type ALYREF but not mutant constructs can rescue phenotypic effects [77]. Eckwahl also confirmed that m5C plays an important role in bridging the interaction between ALYREF and MLV RNA through endogenous RNA immunoprecipitation (RIP). They also confirmed that ALYREF knockout decreased the cytoplasmic and nuclear ratio of m5C in viral RNA, due to fewer m5C containing viral transcripts were exported to the cytoplasm.
Unlike ALYREF, YBX1 was identified as a cytoplasmic mRNA m5C reading protein in human cells [78]. Based on the structural analysis of YBX1 protein and isothermal titration thermal analysis, YBX1 recognizes m5C in its cold shock domain through the indole ring of W65 [79]. YBX1 specifically targets several m5C-containing oncogenes, such as HDGF, and promotes the stability of these oncogenes and the subsequent progression of cancer by recruiting the well-known mRNA stability maintenance factor ELAVL1.
Furthermore, YBX1 could recognize about 60% of m5C mRNAs in urothelial bladder cancer (UCB)-derived T24 cells. In zebrafish, the maternal YBX1 loss can increase overall translation, accumulation of unfolded proteins, leading to oogenesis defects and subsequent embryogenesis failure [80]. Recent studies have also found that 87.8% of the mRNAs modified by m5C were identified by YBX1. Upon the early development of zebrafish embryos, 397 maternal mRNAs with m5C showed significantly decreased expression when YBX1 was knocked out, indicating that these m5Cs mediated by YBX1 played a key role in regulating maternal gene clearance during the transition from the mother to the zygote [23]. In Ypsilon Schachtel (YPS) of Drosophila, the YBX1 homologue also acted as the RNA m5C reading protein [81]. In Drosophila ovary, YPS promotes the homeostasis, proliferation, and differentiation of germline stem cells (GSCs), which depends on the binding of m5C-containing RNAs. Interestingly, like humans and zebrafish YBX1, the highly conserved cold-shock domain (CSD) also contains the YPS m5C binding site. Mutation of this site can induce the defective development of GSC, which indicates that RNA m5C modification has an effect on the development of adult stem cells [82].
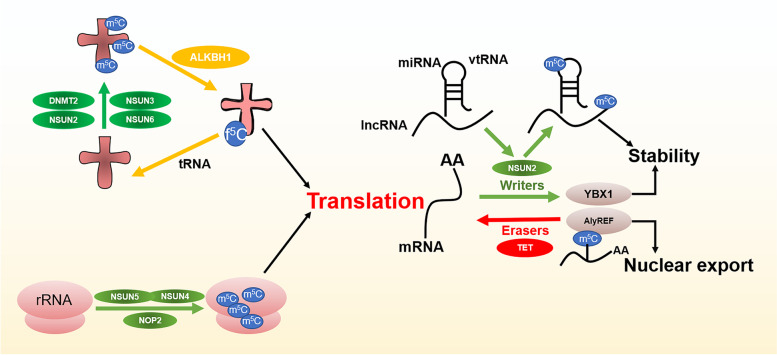
In summary, m5C modification extensively affects gene expression at multiple levels through interacting with a variety of writer, eraser and reader proteins (Fig. 4). Therefore, the dynamic modification of m5C plays multifaceted roles in various biological processes, including embryonic stem cells self-renewal and differentiation [83], circadian rhythm [84], heat shock [85] or DNA damage response [86], and sex determination [87].
Fig. 4.
The functional landscape of m5C writers, readers, and erasers. m5C modification in tRNA or rRNA regulated by NSUN family proteins (NSUN2, NSUN3, NSUN4, NSUN5, NSUN6), NOP2, DNMT2 and ALKBH1 affects the translation. m5C modification of lncRNA, miRNA, vtRNA and mRNA regulated by NSUN2, YBX1, AlyREF and TET family proteins affect their stability and nuclear export
Aberrant m5C in cancers
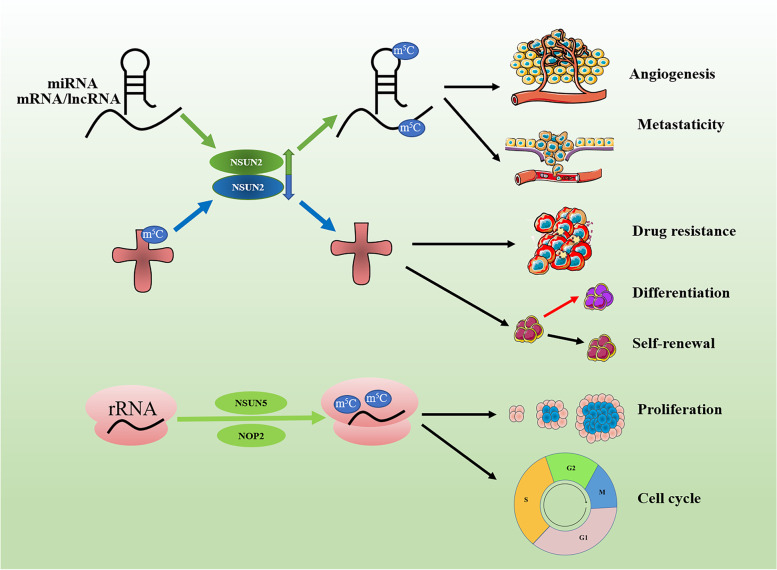
It appears clear that the overall modification m5C and its regulators, including writers, erasers, and readers, are aberrantly expressed in various types of cancers. Emerging evidence indicates that methylation status is closely associated with the pathogenesis of cancer involving initiation, metastasis, progression, as well as drug resistance and relapse (Fig. 5).
Fig. 5.
Aberrant m5C deposition in mRNA, miRNA and lncRNA promotes cancer angiogenesis and metastaticity. NSUN2-mediated tRNA m5C modification regulates cancer stem cell differentiation. m5C upregulation in rRNA by NOP2 or downregulation by NSUN5 disturb the cancer cell proliferation and cell cycle. Red arrows indicate induction. Blue arrows with flat end represent inhibition
Increased levels of RNA m5C can be detected in circulating tumor cells of lung cancer patients [88]. Increased gene expression of NSUN1 and NSUN2 can predict the occurrence and development of cancer [89]. Xue et al. selected m5C-regulated genes from the Cancer Genome Atlas (TCGA) database related to HNSCC and performed in-depth sequence analyses. They found that the expression of genes regulated by m5C was associated with the copy number variation (CNV) pattern. Gene set enrichment analysis (GSEA) showed that m5C reader protein ALYREF can actively promote mRNA export, thereby playing the role of an mRNA export adaptor in vivo and in vitro [90]. Meanwhile, DNMT1 and ALYREF expression levels effectively predicted the risk factors (morbidity and mortality) for patients with HNSCC. Similarly, based on the TCGA database, He et al. linked the abnormal changes of m5C regulatory genes in a variety of tumors with adverse clinical results, and provided a partial explanation for the discussion of cancer pathogenesis and/or survival. He and his colleagues assessed the correlation between the expression of m5C regulatory factors and related cancer pathways, and found that mutations in m5C regulatory genes and CNVs are significantly related in many cancer types. It is worth noting that this study verified the frequency of m5C regulatory patterns in 33 cancers and compared the expression levels of m5C regulatory factors in tumors and adjacent tissues, and found that they were overexpressed in tumors [91]. Glioma is the most common primary intracranial tumor, which is difficult to cure and often recurs. Wang et al. obtained the RNA sequence and clinicopathological data of RNA:m5C methyltransferase under glioma from the Chinese Glioma Genome Atlas (CGGA) and TCGA data sets. The results showed that RNA:m5C methyltransferase was significantly related to the malignant progression of glioma [92].
The NSUN2 gene has the highest mutation rate in gastrointestinal cancers [93]. A high frequency of mutations in m5C regulatory genes were found in HCC. Dysregulation of m5C-related genes was also associated with a higher HCC stage. Moreover, almost all patients with low survival rate were significantly associated with high expression of m5C regulatory factors. High expression of NSUN4 and ALYREF is highly coincident with the survival outcome. GSEA results indicate that methylation and demethylation will lead to high expression of NSUN4 [94]. In addition, the abnormal m5C modification of H19 lncRNA mediated by NSUN2 can affect its stability and interaction with the oncoprotein G3BP1, and it has been proved to be related to the poor differentiation of HCC. It indicates that this interaction may serve as a prognostic biomarker for HCC patients [95].
A recent study indicated that m5C promotes the pathogenesis of human bladder urothelial carcinoma. m5C stabilizes oncogene-mRNAs through the m5C-modified sites in their 3’ UTRs and promotes the pathogenesis of human bladder urothelial carcinoma by [31]. Zhang et al. found that NSUN2 and YBX1 is responsible for the m5C methylation in the 3’-untranslated region of HDGF and promote the urothelial carcinoma of the bladder. Moreover, upregulated NUSN2, YBX1, and HDGF expression levels predicted a poorer survival rate in patients with urothelial carcinoma of the bladder [96]. A recent study demonstrated the involvement of m5C in the development of urothelial carcinoma of the human bladder by stabilizing oncogene-mRNAs through m5C modified sites in their 3′ UTRs [31].
In addition, it has been shown that m5C and its writer proteins are associated with lineage-specific chromatin structure and treatment response/resistance, and they may be an epigenetic driver of leukemia cytochemical resistance. Studies have shown that NSUN3 and DNMT2 forms 5-azacytidine (5-AZA) sensitive chromatin structure through binding to the RNA binding protein hnRNPK, complexed with GATA1 and SPI1/PU.1, which interacts with CDK9/P-TEFb to recruit RNA polymerase-II in the nascent RNA. However, NSUN1 is recruited to the active chromatin structure formed by BRD4 and RNA polymerase-II. Though this chromatin structure is insensitive to 5-AZA, it is highly sensitive to the BRD4 inhibitors JQ1 and NSUN1 knockdown. Furthermore, RNA m5C and NSUN1−/BRD4 related active chromatin were enriched in 5-AZA-resistant leukemia cells and 5-AZA-resistant myelodysplastic syndrome and acute myeloid leukemia specimens [97].
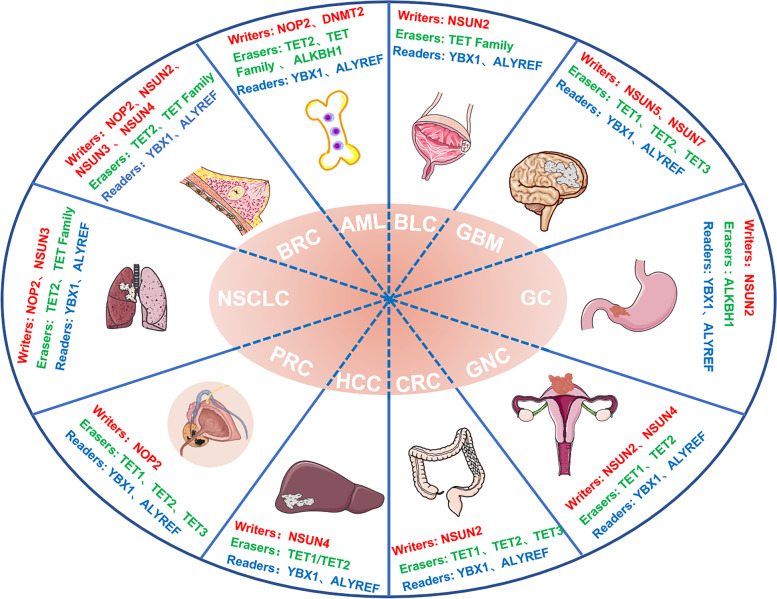
In summary, aberrant expressions of m5C writer, eraser and reader proteins are found in various human cancers (Fig. 6, Table 1).
Fig. 6.
Role of aberrant expressions of m5C writer, reader and eraser proteins in various cancer types. NSCLC: Non-Small Cell Lung Cancer; BRC: Breast Cancer. AML: Acute myeloid leukemia; BLC: Bladder Cancer; GBM: Glioblastoma; GC: Gastric Cancer; GNC: Gynecologic Cancer; CRC: Colorectal Cancer; HCC: Hepatocellular carcinoma; PRC: Prostate Cancer; HDFs: Human diploid fibroblasts; U87: Human glioma cell line U87; ATX: Autotaxin
Table 1.
The role and mechanism of m5C in cancer
| m5C regulators | Position | Function | Potential mechanism | Refs |
|---|---|---|---|---|
| ALYREF, NSUN5, DNMT1 | HNSCC | Participate in progress and predict prognosis | Involved in regulating tumor energy metabolism and protein synthesis | [90] |
| NSUN2 | Gastric Cancer | Cell Proliferation | Regulate downstream gene p57Kip2 | [98] |
| NSUN2 | HDFs | Cell Proliferation and Senescence | Regulate the expression of p27 and CDK1 | [99] |
| NSUN2 | U87 | Cell Migration | Participate in regulation by methylating ATX mRNA | [100] |
| NSUN2 | HCC | Cell Differentiation | Recruit oncoprotein G3BP1 | [95] |
| NSUN2, ALYREF | Bladder Cancer | Cell Proliferation and Migration | Regulation of mRNA stability through a new binding protein YBX1 in the cytoplasm | [31] |
| NSUN6 | Pancreatic Cancer | Cell Proliferation | Regulates cell cycle and G2M checkpoints | [101] |
| NSUN4, ALYREF | HCC | Tumor progression | High expression of NSUN4 and ALYREF is significantly correlated with survival outcome | [94] |
| NSUN2, NSUN6 | TNBC | Tumor proliferation Occurrence and Metastasis. | Affect tumor development and tumor immune microenvironment | [102] |
| NSUN2 | Gastrointestinal Cancer | Tumor progression and pathological staging | Regulates GSK3B and participates in the ErbB/PI3K-Akt signaling pathway | [103] |
| DNMT3A | Glioblastoma Multiforme | Tumor progression | Inhibit the formation of miRNA/mRNA duplexes, loss of tumor suppression kinetic energy | [104] |
| ALYREF | Bladder Cancer | Glycolysis and Tumorigenesis | Overexpression of ALYREF promotes the proliferation of bladder cancer cells through PKM2-mediated glycolysis. | [105] |
m5C and Cancer immunotherapy
The concept of protein replacement therapy using a gene was first proposed 30 years ago [106]. However, due to the fact that RNA molecule is instable and can induce immunogenicity, this approach did not gain propagation. Later, Kariko group incorporated m5C into mRNA therapy either separately or in combination, and it reduced the toll-like receptor (TLR)- mediated immune effects of mRNA [9]. This marks the breakthrough in the RNA and immunotherapy field that base modification in mRNA alters the immune activity. Andries et al. [107] believed that m5C-mRNA can be used as a new approach in the field of mRNA-based therapeutics, partially due to that m5C-modified mRNA may evade the innate immune system through inhibition of endosomal Toll-like receptor 3(TLR3) (Fig. 7).
Fig. 7.
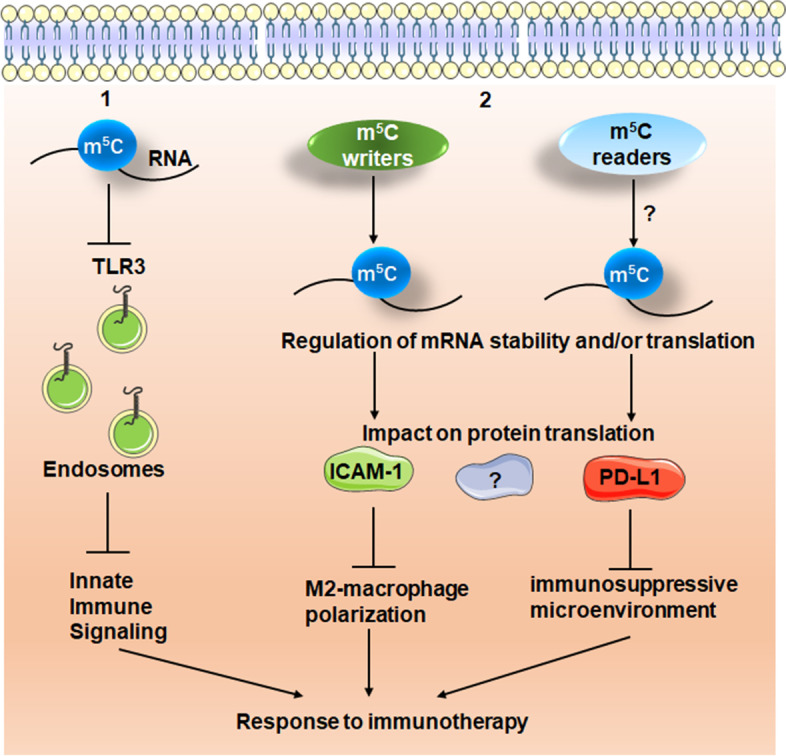
The identified functions of m5C in regulation of immune system
m5C implications in tumor immune microenvironment
Increasing evidence based on bioinformatic analysis implicate the significance of m5C in tumor immune microenvironment. Diverse methylation regulators can be used as prognostic and diagnostic markers of cancer [108, 109]. Oral Squamous Cell Carcinoma (OSCC) patients with higher expression levels of m5C regulators had lower immune activity in TILs such as DCs, NK cells, and CD8+ T-cells [110]. Papillary thyroid cancer (PTC) patients with low m5C-score contained higher resting CD4+ memory T cells and CD8+ T cells and had better prognosis, while activated NK cells and monocytes were mostly enriched in the high m5C-score patients with worse prognosis [111]. In triple-negative breast cancer, the changes in the expression of m5C RNA methylation regulators, the up-regulation of NSUN2 expression and the down-regulation of NSUN6 expression, which can objectively predict the clinical prognostic risk of TNBC patients to a large extent. Therefore, it may become new prognostic markers of TNBC and provide clues for understanding the RNA epigenetic modification of TNBC [102]. Related studies have also confirmed that NSUN3 and NSUN4 can predict the prognosis of lung squamous cell carcinoma and regulate the immune microenvironment [112]. In lung adenocarcinoma patients, different m5C pattern had different TME immune cell infiltration and high m5C score group had a better prognosis [113]. Besides, m5C-regulated lncRNAs also predict the overall survival of lung adenocarcinoma patients and affect the tumor immune microenvironment [114]. In pancreatic cancer patients, 3 m5C-related lncRNAs showed prognostic value. With TIDE (Tumor Immune Dysfunction and Exclusion) algorithm, high-m5C-lncRNA scores had a better response to immunotherapy [115]. In another study of pancreatic ductal adenocarcinoma (PDAC), relationships between m5C-related lncRNAs and PDAC-infiltrating immune cells were evaluated. Naïve B cells, CD8 + T cells, Treg cells, and resting NK cells had a higher expression level in the low-risk group while the M0 and M2 phenotype macrophages had a higher expression level in the high-risk group, suggesting that m5C-related lncRNAs may regulate pancreatic cancer progression via promoting M2 phenotype macrophage polarization or infiltration in PDAC [116]. Related expression also exists in the research of Head and Neck Squamous Cell Carcinoma [117], hepatocellular carcinoma [118], breast cancer [119], human high-grade serous ovarian cancer [120], cutaneous melanoma [121] and colon cancer [122].
m5C writers in regulation of cancer immunotherapy
Immune escape is a hallmark of human cancer. T-cell exhaustion mainly accounts for escape of tumors from immune surveillance. NSUN2, a tRNA methyltransferase, modifies tRNA and mRNA by increasing the methylation of m5C. NSUN2 is an important factor in maintaining stem cell self-renewal and differentiation [123]. In head and neck squamous cell carcinoma (HNSCC), the combination of NSUN2 expression and T-cell activation correlates with patient survival regardless of the HPV status, indicating that NSUN2 may serve as a potential biomarker for immune-checkpoint blockade [124]. It has been reported that NSUN2 methylates ICAM-1 (Intercellular Adhesion Molecule 1) mRNA and promotes its translation [29], thus inhibiting M2-macrophage polarization and suppressing tumor metastasis (Fig. 7). Moreover, targeting NSUN2 expression may also improve the outcome of immunotherapy in HNSCC [125]. Nonetheless, a larger sample size is necessary to further validate how the NSUN2 affects immune checkpoint blockade outcome [126].
m5C readers in regulation of cancer immunotherapy
YBX1 protein is the most common tumor-associated antigen and can also induce T cell response [127]. Therefore it can be used as a target of effector immunity and a candidate vaccine for evaluation [128]. Chemotherapy induces an immunosuppressive microenvironment in tumors and induces immune evasion through YBX1-mediated upregulation of PD-L1 (programmed death-1 ligand 1) (Fig. 7). YBX1 is upregulated in chemoresistant HCC cells. YBX1 knockout reverses chemoresistance by blocking PD-L1 expression and activating T cells in a tumor microenvironment [129]. Upregulation of functional cytotoxic CD8+ T cells and downregulation of myeloid-derived suppressor cells and regulatory T cells are associated with the overcome of the tumor immunosuppressive environment and immune escape status. YBX1 drives signal transduction in a tumor immunosuppressive microenvironment and immune escape pathway. In addition, YBX1 knockout can reverse HCC drug resistance by blocking PD-L1 expression and activating T cells in a tumor microenvironment. Data presented by Tao et al. showed that both tumor immune evasion and multidrug resistance can be reversed by targeting the YBX1 signaling cascade, suggesting an effective treatment regimen against tumor chemoresistance [130].
In conclusion, these data implicate that modulation of m5C regulators could be a useful strategy.
Conclusions and perspectives
m5C modifications of RNA have been demonstrated as a new molecular mechanism that controls eukaryotes gene expression. How the modification of m5C affects the ncRNA biogenesis, localization, and function, and how these effects are associated with etiology of cancer are to be elucidated. In recent years, extensive efforts have been devoted to the study of m5C modifications in cancer. In this review, particular attention was focused to Writer-NSUN2, the Eraser-TET family, and Reader-YBX1..
Some proof-of-concept studies have revealed that maladjusted m5C modulators targeted by small-molecule inhibitors have therapeutic potential for cancer treatment. As of now, specific m5C inhibitors have not been developed. In DNA methylation, the chemicals initially developed to target these proteins, such as 5-aza-2-deoxycytidine (decitabine) or 5-azacytidine (Vidaza), have been approved to treat myelodysplastic syndrome by the FDA, but their usage in solid tumor treatments is limited.
Bioinformatic research in RNA m5C modification will also be critical in both identifying the m5C sites from Single-molecule sequencing and in prediction of m5C sites in different RNA species [131]. Several machine-learning algorithms have recently been developed to predict the m5C sites in Homo sapiens, Mus musculus and Arabidopsis thaliana [34, 132]. With the fast development of computational approaches, it can be expected that the m5C epitranscriptomics will be fully understood and its relation with cancer will be uncovered.
In the future, the detailed roles of RNA m5C in immune system regulation and tumor immune microenvironment will be important directions. As for RNA m6A, depletion of m6A writers, readers or erasers had significant phenotypic consequences in the cellular response to infection and in a few immune cell types. Whether RNA m5C exerts similar functions in the immune system deserves deep efforts. More importantly, how different RNA modifications combat or combine with each other in immune regulation needs a long way to explore.
Different from m6A, the RNA m5C modification partially shares the same enzymatic machinery including DNMT1 with DNA m5C. This mechanism may also play critical roles during the tumor development and progression. m5C can operate with the tumor cells and immune microenvironment to brake or accelerate the tumorigenesis. It will be interesting to identify the immune checkpoint molecular target of m5C in the tumor microenvironment, so that targeted therapy can be used in combination with checkpoint immunotherapy to better treat cancer. In general, the investigation of novel m5C epigenetic modification in cancer not only will provide new insights into the molecular mechanisms of cancer biology and immune response, but also will pave the way to the develop new promising therapies.
Acknowledgements
Thanks to all the scientists working in this field and those public research funds that fund our research about this topic.
Abbreviations
- m6A
N6-methyladenosine
- m5C
5-methylcytosine
- UTRs
Untranslated regions
- tRNA
Transfer RNA
- rRNA
Ribosomal RNA
- mRNA
Messenger RNA
- snRNA
Small nuclear RNA
- miRNA
Micro RNA
- lncRNA
Long non-coding RNA
- eRNA
Enhancer RNA
- 5hmC
5-hydroxymethylcytosine
- 5fC
5-formylcytosine
- 5caC
5-carboxycytosine
- DNMT
DNA methyltransferase
- LC-MS
Liquid chromatography-mass spectrometry
- CDS
Coding sequence
- RCMTs
RNA:m5C methyltransferases
- TRDMT
TRNA-specific methyltransferase
- TET
Ten-eleven translocation
- ALKBH1
Alpha-ketoglutarate-dependent dioxygenase ABH1
- hm5cm34
5-hydroxymethyl-20-o-methylcytidine
- f5cm34
5-formyl-20-o-methylcytidine
- RIP
RNA immunoprecipitation
- GSCs
Germline stem cells
- CSD
Cold-shock domain
- VL
V-loop
- TSL
T-stem and loop
- XIST
X-inactive specific transcripts
- PRC2
Polycomb repressing complex 2
- CNV
Copy number variation
- GSEA
Enrichment analysis
- TFs
Transcription factors
- 5-AZA
5-azacytidine
- FDA
Food and Drug Administration
- MDS
Myelodysplastic syndrome
- AML
Acute myelogenous leukemia
- SGC
Structural Genome Consortium
- SAM
S-adenosylmethionine
- 5’FU
5-fluorouracil
- GSC
Germline stem cell
- S1P
Phingosine-1-phosphate
- SPHK
Sphingosine kinase
- BC
Breast cancer
- PCa
Prostate cancer
- TLR
Toll-like receptor
- SLE
Systemic lupus erythematosus
- CR
Coding region
- HNSCC
Head and neck squamous cell carcinoma
- CTLA4
Cytotoxic T lymphocyte antigen 4
- PD-1
Programmed cell death 1
- ICAM-1
Intercellular adhesion molecule 1
- PD-L1
Programmed death-1 ligand 1
Authors’ contributions
H.S., B.L., and T.M. conceived this review and critically revised the manuscript. H.S. and B.L. drafted the manuscript. J.X., B.C., H.Y. and J.S. drew the figures and collected the related references. J. Z. and X.Y. supervised and revised the manuscript. All authors read and approved the final manuscript.
Funding
National Natural Science Foundation of China (81802103).
Natural Science Foundation of Guangdong Province (2020A1515010605).
Project of High-Level Talents in AHUTCM (2019rcZD001).
National Key R&D Program of China (2021YFE0202000).
Excellent Young Scholars Project of Natural Science Foundation of Anhui Province in China(grant No.2108085Y29).
Natural Science Foundation cultivation project of Capital Medical University(No.PYZ21162).
Availability of data and materials
The authors confirm that the data support the findings of this study are available within the article.
Declarations
Ethical approval and consent to participate
Not applicable.
Consent for publication
All author consent for publication.
Competing interests
All authors have declared that there is no conflict of interest in this manuscript.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hang Song, Jianye Zhang and Bin Liu contributed equally to this work.
Contributor Information
Jianye Zhang, Email: jianyez@163.com.
Xiyong Yu, Email: yuxycn@aliyun.com.
Teng Ma, Email: mateng82913@163.com.
References
- 1.Zhao Y, Chen F, Li Q, Wang L, Fan C. Isothermal amplification of nucleic acids. Chem Rev. 2015;115(22):12491–12545. doi: 10.1021/acs.chemrev.5b00428. [DOI] [PubMed] [Google Scholar]
- 2.Schaefer M, Kapoor U, Jantsch MF. Understanding RNA modifications: the promises and technological bottlenecks of the ‘epitranscriptome’. Open Biol. 2017;7(5):170077. doi: 10.1098/rsob.170077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhao L-Y, Song J, Liu Y, Song C-X, Yi C. Mapping the epigenetic modifications of DNA and RNA. Protein Cell. 2020;11(11):792–808. doi: 10.1007/s13238-020-00733-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Frye M, Harada BT, Behm M, He C. RNA modifications modulate gene expression during development. Science (New York, NY) 2018;361(6409):1346–1349. doi: 10.1126/science.aau1646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang X, Lu Z, Gomez A, et al. N6-methyladenosine-dependent regulation of messenger RNA stability. Nature. 2014;505(7481):117–120. doi: 10.1038/nature12730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Niu Y, Wan A, Lin Z, Lu X, Wan G. N (6)-Methyladenosine modification: a novel pharmacological target for anti-cancer drug development. Acta Pharm Sin B. 2018;8(6):833–843. doi: 10.1016/j.apsb.2018.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hotchkiss RD. The quantitative separation of purines, pyrimidines, and nucleosides by paper chromatography. J Biol Chem. 1948;175(1):315–332. [PubMed] [Google Scholar]
- 8.Wyatt GR. Occurrence of 5-methylcytosine in nucleic acids. Nature. 1950;166(4214):237–238. doi: 10.1038/166237b0. [DOI] [PubMed] [Google Scholar]
- 9.Karikó K, Buckstein M, Ni H, Weissman D. Suppression of RNA recognition by toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity. 2005;23(2):165–175. doi: 10.1016/j.immuni.2005.06.008. [DOI] [PubMed] [Google Scholar]
- 10.Cantara WA, Crain PF, Rozenski J, et al. The RNA modification database, RNAMDB: 2011 update. Nucleic Acids Res. 2011;39(Database issue):D195–D201. doi: 10.1093/nar/gkq1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Shinoda S, Kitagawa S, Nakagawa S, et al. Mammalian NSUN2 introduces 5-methylcytidines into mitochondrial tRNAs. Nucleic Acids Res. 2019;47(16):8734–8745. doi: 10.1093/nar/gkz575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Edelheit S, Schwartz S, Mumbach MR, Wurtzel O, Sorek R. Transcriptome-wide mapping of 5-methylcytidine RNA modifications in bacteria, archaea, and yeast reveals m5C within archaeal mRNAs. PLoS Genet. 2013;9(6):e1003602. doi: 10.1371/journal.pgen.1003602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Strobel MC, Abelson J. Effect of intron mutations on processing and function of Saccharomyces cerevisiae SUP53 tRNA in vitro and in vivo. Mol Cell Biol. 1986;6(7):2663–2673. doi: 10.1128/mcb.6.7.2663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen Y, Sierzputowska-Gracz H, Guenther R, Everett K, Agris PF. 5-Methylcytidine is required for cooperative binding of Mg2+ and a conformational transition at the anticodon stem-loop of yeast phenylalanine tRNA. Biochemistry. 1993;32(38):10249–10253. doi: 10.1021/bi00089a047. [DOI] [PubMed] [Google Scholar]
- 15.Schaefer M, Pollex T, Hanna K, et al. RNA methylation by Dnmt2 protects transfer RNAs against stress-induced cleavage. Genes Dev. 2010;24(15):1590–1595. doi: 10.1101/gad.586710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tuorto F, Liebers R, Musch T, et al. RNA cytosine methylation by Dnmt2 and NSun2 promotes tRNA stability and protein synthesis. Nat Struct Mol Biol. 2012;19(9):900–905. doi: 10.1038/nsmb.2357. [DOI] [PubMed] [Google Scholar]
- 17.Chan CT, Pang YL, Deng W, et al. Reprogramming of tRNA modifications controls the oxidative stress response by codon-biased translation of proteins. Nat Commun. 2012;3:937. doi: 10.1038/ncomms1938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shanmugam R, Fierer J, Kaiser S, Helm M, Jurkowski TP, Jeltsch A. Cytosine methylation of tRNA-asp by DNMT2 has a role in translation of proteins containing poly-asp sequences. Cell discovery. 2015;1:15010. doi: 10.1038/celldisc.2015.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Janin M, Ortiz-Barahona V, de Moura MC, et al. Epigenetic loss of RNA-methyltransferase NSUN5 in glioma targets ribosomes to drive a stress adaptive translational program. Acta Neuropathol. 2019;138(6):1053–1074. doi: 10.1007/s00401-019-02062-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wildenauer D, Gross HJ, Riesner D. Enzymatic methylations: III. Cadaverine-induced conformational changes of E. coli tRNA fMet as evidenced by the availability of a specific adenosine and a specific cytidine residue for methylation. Nucleic Acids Res. 1974;1(9):1165–1182. doi: 10.1093/nar/1.9.1165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jank P, Gross HJ. Methyl-deficient mammalian transfer RNA: II. Homologous methylation in vitro of liver tRNA from normal and ethionine-fed rats: ethionine effect on 5-methyl-cytidine synthesis in vivo. Nucleic Acids Res. 1974;1(10):1259–1267. doi: 10.1093/nar/1.10.1259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yang X, Yang Y, Sun BF, et al. 5-methylcytosine promotes mRNA export - NSUN2 as the methyltransferase and ALYREF as an m (5) C reader. Cell Res. 2017;27(5):606–625. doi: 10.1038/cr.2017.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yang Y, Wang L, Han X, et al. RNA 5-Methylcytosine facilitates the maternal-to-zygotic transition by preventing maternal mRNA decay. Mol Cell. 2019;75(6):1188–202.e1111. [DOI] [PubMed]
- 24.Young JI, Hong EP, Castle JC, et al. Regulation of RNA splicing by the methylation-dependent transcriptional repressor methyl-CpG binding protein 2. Proc Natl Acad Sci U S A. 2005;102(49):17551–17558. doi: 10.1073/pnas.0507856102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen H, Yang H, Zhu X, et al. M (5) C modification of mRNA serves a DNA damage code to promote homologous recombination. Nat Commun. 2020;11(1):2834. doi: 10.1038/s41467-020-16722-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xue S, Xu H, Sun Z, et al. Depletion of TRDMT1 affects 5-methylcytosine modification of mRNA and inhibits HEK293 cell proliferation and migration. Biochem Biophys Res Commun. 2019;520(1):60–66. doi: 10.1016/j.bbrc.2019.09.098. [DOI] [PubMed] [Google Scholar]
- 27.Zou F, Tu R, Duan B, et al. Drosophila YBX1 homolog YPS promotes ovarian germ line stem cell development by preferentially recognizing 5-methylcytosine RNAs. Proc Natl Acad Sci U S A. 2020;117(7):3603–3609. doi: 10.1073/pnas.1910862117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Warren L, Manos PD, Ahfeldt T, et al. Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell. 2010;7(5):618–630. doi: 10.1016/j.stem.2010.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Luo Y, Feng J, Xu Q, Wang W, Wang X. NSun2 deficiency protects endothelium from inflammation via mRNA methylation of ICAM-1. Circ Res. 2016;118(6):944–956. doi: 10.1161/CIRCRESAHA.115.307674. [DOI] [PubMed] [Google Scholar]
- 30.Guo G, Wang H, Shi X, et al. Disease activity-associated alteration of mRNA m (5) C methylation in CD4(+) T cells of systemic lupus Erythematosus. Front Cell Dev Biol. 2020;8:430. doi: 10.3389/fcell.2020.00430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chen X, Li A, Sun BF, et al. 5-methylcytosine promotes pathogenesis of bladder cancer through stabilizing mRNAs. Nat Cell Biol. 2019;21(8):978–990. doi: 10.1038/s41556-019-0361-y. [DOI] [PubMed] [Google Scholar]
- 32.Motorin Y, Lyko F, Helm M. 5-methylcytosine in RNA: detection, enzymatic formation and biological functions. Nucleic Acids Res. 2010;38(5):1415–1430. doi: 10.1093/nar/gkp1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Selmi T, Hussain S, Dietmann S, et al. Sequence- and structure-specific cytosine-5 mRNA methylation by NSUN6. Nucleic Acids Res. 2021;49(2):1006–1022. doi: 10.1093/nar/gkaa1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chai D, Jia C, Zheng J, Zou Q, Li F. Staem5: a novel computational approachfor accurate prediction of m5C site. Mol Ther Nucleic Acids. 2021;26:1027–1034. doi: 10.1016/j.omtn.2021.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Squires JE, Preiss T. Function and detection of 5-methylcytosine in eukaryotic RNA. Epigenomics. 2010;2(5):709–715. doi: 10.2217/epi.10.47. [DOI] [PubMed] [Google Scholar]
- 36.Squires JE, Patel HR, Nousch M, et al. Widespread occurrence of 5-methylcytosine in human coding and non-coding RNA. Nucleic Acids Res. 2012;40(11):5023–5033. doi: 10.1093/nar/gks144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Amort T, Rieder D, Wille A, et al. Distinct 5-methylcytosine profiles in poly(a) RNA from mouse embryonic stem cells and brain. Genome Biol. 2017;18(1):1. doi: 10.1186/s13059-016-1139-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wei Z, Panneerdoss S, Timilsina S, et al. Topological characterization of human and mouse m (5) C Epitranscriptome revealed by bisulfite sequencing. Int J Genomics. 2018;2018:1351964. doi: 10.1155/2018/1351964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Huang T, Chen W, Liu J, Gu N, Zhang R. Genome-wide identification of mRNA 5-methylcytosine in mammals. Nat Struct Mol Biol. 2019;26(5):380–388. doi: 10.1038/s41594-019-0218-x. [DOI] [PubMed] [Google Scholar]
- 40.David R, Burgess A, Parker B, et al. Transcriptome-wide mapping of RNA 5-Methylcytosine in Arabidopsis mRNAs and noncoding RNAs. Plant Cell. 2017;29(3):445–460. doi: 10.1105/tpc.16.00751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cui X, Liang Z, Shen L, et al. 5-Methylcytosine RNA methylation in Arabidopsis Thaliana. Mol Plant. 2017;10(11):1387–1399. doi: 10.1016/j.molp.2017.09.013. [DOI] [PubMed] [Google Scholar]
- 42.Chen YS, Yang WL, Zhao YL, Yang YG. Dynamic transcriptomic m (5) C and its regulatory role in RNA processing. Wiley Interdiscip Rev RNA. 2021;12(4):e1639. doi: 10.1002/wrna.1639. [DOI] [PubMed] [Google Scholar]
- 43.Shen H, Ontiveros RJ, Owens MC, et al. TET-mediated 5-methylcytosine oxidation in tRNA promotes translation. J Biol Chem. 2021;296:100087. doi: 10.1074/jbc.RA120.014226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Reid R, Greene PJ, Santi DV. Exposition of a family of RNA m (5) C methyltransferases from searching genomic and proteomic sequences. Nucleic Acids Res. 1999;27(15):3138–3145. doi: 10.1093/nar/27.15.3138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Van Haute L, Lee S-Y, McCann BJ, et al. NSUN2 introduces 5-methylcytosines in mammalian mitochondrial tRNAs. Nucleic Acids Res. 2019;47(16):8720–8733. doi: 10.1093/nar/gkz559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Xu L, Liu X, Sheng N, et al. Three distinct 3-methylcytidine (mC) methyltransferases modify tRNA and mRNA in mice and humans. J Biol Chem. 2017;292(35):14695–14703. doi: 10.1074/jbc.M117.798298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Brzezicha B, Schmidt M, Makalowska I, Jarmolowski A, Pienkowska J, Szweykowska-Kulinska Z. Identification of human tRNA:m5C methyltransferase catalysing intron-dependent m5C formation in the first position of the anticodon of the pre-tRNA Leu (CAA) Nucleic Acids Res. 2006;34(20):6034–6043. doi: 10.1093/nar/gkl765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Goll MG, Kirpekar F, Maggert KA, et al. Methylation of tRNAAsp by the DNA methyltransferase homolog Dnmt2. Science (New York, NY) 2006;311(5759):395–398. doi: 10.1126/science.1120976. [DOI] [PubMed] [Google Scholar]
- 49.Zhang Y, Zhang X, Shi J, et al. Dnmt2 mediates intergenerational transmission of paternally acquired metabolic disorders through sperm small non-coding RNAs. Nat Cell Biol. 2018;20(5):535–540. doi: 10.1038/s41556-018-0087-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Burgess AL, David R, Searle IR. Conservation of tRNA and rRNA 5-methylcytosine in the kingdom Plantae. BMC Plant Biol. 2015;15:199. doi: 10.1186/s12870-015-0580-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Long T, Li J, Li H, et al. Sequence-specific and shape-selective RNA recognition by the human RNA 5-Methylcytosine methyltransferase NSun6. J Biol Chem. 2016;291(46):24293–24303. doi: 10.1074/jbc.M116.742569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Van Haute L, Powell CA, Minczuk M. Dealing with an unconventional genetic code in mitochondria: the biogenesis and pathogenic defects of the 5-Formylcytosine modification in mitochondrial tRNA. Biomolecules. 2017;7(1):24. [Google Scholar]
- 53.Sharma S, Yang J, Watzinger P, Kötter P, Entian K-D. Yeast Nop2 and Rcm1 methylate C2870 and C2278 of the 25S rRNA, respectively. Nucleic Acids Res. 2013;41(19):9062–9076. doi: 10.1093/nar/gkt679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Metodiev MD, Spåhr H, Loguercio Polosa P, et al. NSUN4 is a dual function mitochondrial protein required for both methylation of 12S rRNA and coordination of mitoribosomal assembly. PLoS Genet. 2014;10(2):e1004110. doi: 10.1371/journal.pgen.1004110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Heissenberger C, Liendl L, Nagelreiter F, et al. Loss of the ribosomal RNA methyltransferase NSUN5 impairs global protein synthesis and normal growth. Nucleic Acids Res. 2019;47(22):11807–11825. doi: 10.1093/nar/gkz1043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hussain S, Sajini AA, Blanco S, et al. NSun2-mediated cytosine-5 methylation of vault noncoding RNA determines its processing into regulatory small RNAs. Cell Rep. 2013;4(2):255–261. doi: 10.1016/j.celrep.2013.06.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kouznetsova VL, Tchekanov A, Li X, Yan X, Tsigelny IF. Polycomb repressive 2 complex-molecular mechanisms of function. Protein Sci. 2019;28(8):1387–1399. doi: 10.1002/pro.3647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Hu H, Shu M, He L, et al. Epigenomic landscape of 5-hydroxymethylcytosine reveals its transcriptional regulation of lncRNAs in colorectal cancer. Br J Cancer. 2017;116(5):658–668. doi: 10.1038/bjc.2016.457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Liu Y, Siejka-Zielińska P, Velikova G, et al. Bisulfite-free direct detection of 5-methylcytosine and 5-hydroxymethylcytosine at base resolution. Nat Biotechnol. 2019;37(4):424–429. doi: 10.1038/s41587-019-0041-2. [DOI] [PubMed] [Google Scholar]
- 60.Chi L, Delgado-Olguín P. Expression of NOL1/NOP2/sun domain (Nsun) RNA methyltransferase family genes in early mouse embryogenesis. Gene Expr Patterns. 2013;13(8):319–327. doi: 10.1016/j.gep.2013.06.003. [DOI] [PubMed] [Google Scholar]
- 61.Walbott H, Auxilien S, Grosjean H, Golinelli-Pimpaneau B. The carboxyl-terminal extension of yeast tRNA m5C methyltransferase enhances the catalytic efficiency of the amino-terminal domain. J Biol Chem. 2007;282(32):23663–23671. doi: 10.1074/jbc.M703818200. [DOI] [PubMed] [Google Scholar]
- 62.Govindaraju G, Jabeena CA, Sethumadhavan DV, Rajaram N, Rajavelu A. DNA methyltransferase homologue TRDMT1 in plasmodium falciparum specifically methylates endogenous aspartic acid tRNA. Biochim Biophys Acta Gene Regul Mech. 2017;1860(10):1047–1057. doi: 10.1016/j.bbagrm.2017.08.003. [DOI] [PubMed] [Google Scholar]
- 63.Chen Y-S, Yang W-L, Zhao Y-L, Yang Y-G. Dynamic transcriptomic m C and its regulatory role in RNA processing. Wiley Interdiscip Rev RNA. 2021;12(4):e1639. doi: 10.1002/wrna.1639. [DOI] [PubMed] [Google Scholar]
- 64.Krishnamohan A, Dodbele S, Jackman JE. Insights into catalytic and tRNA recognition mechanism of the dual-specific tRNA methyltransferase from. Genes (Basel) 2019;10(2):100. doi: 10.3390/genes10020100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Tao W, Zhao D, Sun M, et al. Intestinal absorption and activation of decitabine amino acid ester prodrugs mediated by peptide transporter PEPT1 and enterocyte enzymes. Int J Pharm. 2018;541(1-2):64–71. doi: 10.1016/j.ijpharm.2018.02.033. [DOI] [PubMed] [Google Scholar]
- 66.Liu RJ, Long T, Li J, Li H, Wang ED. Structural basis for substrate binding and catalytic mechanism of a human RNA:m5C methyltransferase NSun6. Nucleic Acids Res. 2017;45(11):6684–6697. doi: 10.1093/nar/gkx473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lio C-WJ, Yuita H, Rao A. Dysregulation of the TET family of epigenetic regulators in lymphoid and myeloid malignancies. Blood. 2019;134(18):1487–1497. doi: 10.1182/blood.2019791475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wojciechowski M, Rafalski D, Kucharski R, et al. Insights into DNA hydroxymethylation in the honeybee from in-depth analyses of TET dioxygenase. Open Biol. 2014;4(8). [DOI] [PMC free article] [PubMed]
- 69.Lio C-WJ, Rao A. TET enzymes and 5hmC in adaptive and innate immune systems. Front Immunol. 2019;10:210. doi: 10.3389/fimmu.2019.00210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ito S, Shen L, Dai Q, et al. Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science (New York, NY) 2011;333(6047):1300–1303. doi: 10.1126/science.1210597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Shen H, Ontiveros RJ, Owens MC, et al. TET-mediated 5-methylcytosine oxidation in tRNA promotes translation. J Biol Chem. 2020:296–100087. [DOI] [PMC free article] [PubMed]
- 72.Shen Q, Zhang Q, Shi Y, et al. Tet2 promotes pathogen infection-induced myelopoiesis through mRNA oxidation. Nature. 2018;554(7690):123–127. doi: 10.1038/nature25434. [DOI] [PubMed] [Google Scholar]
- 73.Liu F, Clark W, Luo G, et al. ALKBH1-mediated tRNA Demethylation regulates translation. Cell. 2016;167(3):816–828. doi: 10.1016/j.cell.2016.09.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Haag S, Sloan KE, Ranjan N, et al. NSUN3 and ABH1 modify the wobble position of mt-tRNAMet to expand codon recognition in mitochondrial translation. EMBO J. 2016;35(19):2104–2119. doi: 10.15252/embj.201694885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Ougland R, Jonson I, Moen MN, et al. Role of ALKBH1 in the Core transcriptional network of embryonic stem cells. Cell Physiol Biochem. 2016;38(1):173–184. doi: 10.1159/000438619. [DOI] [PubMed] [Google Scholar]
- 76.Makey DM, Shchurik V, Wang H, et al. Mapping the separation landscape in two-dimensional liquid chromatography: blueprints for efficient analysis and purification of pharmaceuticals enabled by computer-assisted modeling. Anal Chem. 2021;93(2):964–972. doi: 10.1021/acs.analchem.0c03680. [DOI] [PubMed] [Google Scholar]
- 77.Eckwahl M, Xu R, Michalkiewicz J, et al. 5-Methylcytosine RNA modifications promote retrovirus replication in an ALYREF reader protein-dependent manner. J Virol. 2020;94(13):e00544–e00520. doi: 10.1128/JVI.00544-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Zou F, Tu R, Duan B, et al. YBX1 homolog YPS promotes ovarian germ line stem cell development by preferentially recognizing 5-methylcytosine RNAs. Proc Natl Acad Sci U S A. 2020;117(7):3603–3609. doi: 10.1073/pnas.1910862117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lyabin DN, Eliseeva IA, Ovchinnikov LP. YB-1 protein: functions and regulation. Wiley Interdiscip Rev RNA. 2014;5(1):95–110. doi: 10.1002/wrna.1200. [DOI] [PubMed] [Google Scholar]
- 80.Huang C, Zhu B, Leng D, Ge W, Zhang XD. Long noncoding RNAs implicated in embryonic development in Ybx1 knockout zebrafish. FEBS Open Bio. 2021;11(4):1259–76. [DOI] [PMC free article] [PubMed]
- 81.Mansfield JH, Wilhelm JE, Hazelrigg T. Ypsilon Schachtel, a Drosophila Y-box protein, acts antagonistically to orb in the oskar mRNA localization and translation pathway. Development (Cambridge, England) 2002;129(1):197–209. doi: 10.1242/dev.129.1.197. [DOI] [PubMed] [Google Scholar]
- 82.Tatomer DC, Rizzardi LF, Curry KP, Witkowski AM, Marzluff WF, Duronio RJ. Drosophila Symplekin localizes dynamically to the histone locus body and tricellular junctions. Nucleus. 2014;5(6):613–625. doi: 10.4161/19491034.2014.990860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Frye M, Blanco S. Post-transcriptional modifications in development and stem cells. Development (Cambridge, England) 2016;143(21):3871–3881. doi: 10.1242/dev.136556. [DOI] [PubMed] [Google Scholar]
- 84.Hoernes TP, Erlacher MD. Translating the epitranscriptome. Wiley Interdiscip Rev RNA. 2017;8(1):e1375. [DOI] [PMC free article] [PubMed]
- 85.Tang Y, Gao C-C, Gao Y, et al. OsNSUN2-mediated 5-Methylcytosine mRNA modification enhances Rice adaptation to high temperature. Dev Cell. 2020;53(3):272–286.e7. doi: 10.1016/j.devcel.2020.03.009. [DOI] [PubMed] [Google Scholar]
- 86.Fehér Z, Schlagman SL, Miner Z, Hattman S. The UV excision-repair system of Saccharomyces cerevisiae is involved in the removal of methylcytosines formed in vivo by a cloned prokaryotic DNA methyltransferase. Curr Genet. 1989;16(5-6):461–464. doi: 10.1007/BF00340726. [DOI] [PubMed] [Google Scholar]
- 87.Okashita N, Kuroki S, Maeda R, Tachibana M. TET2 catalyzes active DNA demethylation of the Sry promoter and enhances its expression. Sci Rep. 2019;9(1):13462. doi: 10.1038/s41598-019-50058-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Mackenzie JW, Lewis RJ, Sisler GE, Lin W, Rogers J, Clark I. Urinary catabolites of ribonucleic acid as cancer markers: a preliminary report of their use in patients with lung cancer. Ann Thorac Surg. 1984;38(2):133–139. doi: 10.1016/s0003-4975(10)62220-5. [DOI] [PubMed] [Google Scholar]
- 89.Chellamuthu A, Gray SG. The RNA methyltransferase NSUN2 and its potential roles in Cancer. Cells. 2020;9(8). [DOI] [PMC free article] [PubMed]
- 90.Xue M, Shi Q, Zheng L, Li Q, Yang L, Zhang Y. Gene signatures of m5C regulators may predict prognoses of patients with head and neck squamous cell carcinoma. Am J Transl Res. 2020;12(10):6841–6852. [PMC free article] [PubMed] [Google Scholar]
- 91.He Y, Yu X, Zhang M, Guo W. Pan-cancer analysis of m (5) C regulator genes reveals consistent epigenetic landscape changes in multiple cancers. World J Sur Oncol. 2021;19(1):224. doi: 10.1186/s12957-021-02342-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wang P, Wu M, Tu Z, et al. Identification of RNA: 5-Methylcytosine Methyltransferases-related signature for predicting prognosis in Glioma. Front Oncol. 2020;10:1119. doi: 10.3389/fonc.2020.01119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Xie S, Chen W, Chen K, et al. Emerging roles of RNA methylation in gastrointestinal cancers. Cancer Cell Int. 2020;20(1):585. doi: 10.1186/s12935-020-01679-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.He Y, Yu X, Li J, Zhang Q, Zheng Q, Guo W. Role of m (5) C-related regulatory genes in the diagnosis and prognosis of hepatocellular carcinoma. Am J Transl Res. 2020;12(3):912–922. [PMC free article] [PubMed] [Google Scholar]
- 95.Sun Z, Xue S, Zhang M, et al. Aberrant NSUN2-mediated m (5) C modification of H19 lncRNA is associated with poor differentiation of hepatocellular carcinoma. Oncogene. 2020;39(45):6906–6919. doi: 10.1038/s41388-020-01475-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Zhang C, Chang X, Chen D, et al. Downregulation of HDGF inhibits the tumorigenesis of bladder cancer cells by inactivating the PI3K-AKT signaling pathway. Cancer Manag Res. 2019;11:7909–7923. doi: 10.2147/CMAR.S215341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Cheng JX, Chen L, Li Y, et al. RNA cytosine methylation and methyltransferases mediate chromatin organization and 5-azacytidine response and resistance in leukaemia. Nat Commun. 2018;9(1):1163. doi: 10.1038/s41467-018-03513-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Mei L, Shen C, Miao R, et al. RNA methyltransferase NSUN2 promotes gastric cancer cell proliferation by repressing p57(Kip2) by an m (5) C-dependent manner. Cell Death Dis. 2020;11(4):270. doi: 10.1038/s41419-020-2487-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Tang H, Fan X, Xing J, et al. NSun2 delays replicative senescence by repressing p27 (KIP1) translation and elevating CDK1 translation. Aging. 2015;7(12):1143–1158. doi: 10.18632/aging.100860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Xu X, Zhang Y, Zhang J, Zhang X. NSun2 promotes cell migration through methylating autotaxin mRNA. J Biol Chem. 2020;295(52):18134–18147. doi: 10.1074/jbc.RA119.012009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Yang R, Liang X, Wang H, et al. The RNA methyltransferase NSUN6 suppresses pancreatic cancer development by regulating cell proliferation. EBioMedicine. 2021;63:103195. doi: 10.1016/j.ebiom.2020.103195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Huang Z, Pan J, Wang H, et al. Prognostic significance and tumor immune microenvironment Heterogenicity of m5C RNA methylation regulators in triple-negative breast Cancer. Front Cell Dev Biol. 2021;9:657547. doi: 10.3389/fcell.2021.657547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Xiang S, Ma Y, Shen J, et al. M (5) C RNA methylation primarily affects the ErbB and PI3K-Akt signaling pathways in gastrointestinal Cancer. Front Mol Biosci. 2020;7:599340. doi: 10.3389/fmolb.2020.599340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Cheray M, Etcheverry A, Jacques C, et al. Cytosine methylation of mature microRNAs inhibits their functions and is associated with poor prognosis in glioblastoma multiforme. Mol Cancer. 2020;19(1):36. doi: 10.1186/s12943-020-01155-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Wang JZ, Zhu W, Han J, et al. The role of the HIF-1α/ALYREF/PKM2 axis in glycolysis and tumorigenesis of bladder cancer. Cancer Commun (London, England) 2021;41(7):560–575. doi: 10.1002/cac2.12158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Pezzilli R, Caccialanza R, Capurso G, Brunetti O, Milella M, Falconi M. Pancreatic enzyme replacement therapy in pancreatic Cancer. Cancers. 2020;12(2). [DOI] [PMC free article] [PubMed]
- 107.Andries O, Mc Cafferty S, De Smedt SC, Weiss R, Sanders NN, Kitada T. N (1)-methylpseudouridine-incorporated mRNA outperforms pseudouridine-incorporated mRNA by providing enhanced protein expression and reduced immunogenicity in mammalian cell lines and mice. J Control Release. 2015;217:337–344. doi: 10.1016/j.jconrel.2015.08.051. [DOI] [PubMed] [Google Scholar]
- 108.Sun L, Liu WK, Du XW, et al. Large-scale transcriptome analysis identified RNA methylation regulators as novel prognostic signatures for lung adenocarcinoma. Ann Transl Med. 2020;8(12):751. doi: 10.21037/atm-20-3744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Huang M, Zhang Y, Ou X, et al. m5C-related signatures for predicting prognosis in cutaneous melanoma with machine learning. J Oncol. 2021;2021:6173206. doi: 10.1155/2021/6173206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Gao L, Chen R, Sugimoto M, et al. The RNA methylation modification 5-Methylcytosine impacts immunity characteristics, prognosis and progression of Oral squamous cell carcinoma by bioinformatics analysis. Front Bioeng Biotechnol. 2021;9:760724. doi: 10.3389/fbioe.2021.760724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Li F, Deng Q, Pang X, et al. M (5) C regulator-mediated methylation modification patterns and tumor microenvironment infiltration characterization in papillary thyroid carcinoma. Front Oncol. 2021;11:729887. doi: 10.3389/fonc.2021.729887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Pan J, Huang Z, Xu Y. m5C RNA methylation regulators predict prognosis and regulate the immune microenvironment in lung squamous cell carcinoma. Front Oncol. 2021;11:657466. doi: 10.3389/fonc.2021.657466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Chen H, Ge XL, Zhang ZY, et al. M (5) C regulator-mediated methylation modification patterns and tumor microenvironment infiltration characterization in lung adenocarcinoma. Transl Lung Cancer Res. 2021;10(5):2172–2192. doi: 10.21037/tlcr-21-351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Pan J, Huang Z, Xu Y. m5C-related lncRNAs predict overall survival of patients and regulate the tumor immune microenvironment in lung adenocarcinoma. Front Cell Dev Biol. 2021;9:671821. doi: 10.3389/fcell.2021.671821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Liu X, Wang D, Han S, et al. Signature of m5C-related lncRNA for prognostic prediction and immune responses in pancreatic Cancer. J Oncol. 2022;2022:7467797. doi: 10.1155/2022/7467797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Yuan H, Liu J, Zhao L, et al. Prognostic risk model and tumor immune environment modulation of m5C-related LncRNAs in pancreatic ductal adenocarcinoma. Front Immunol. 2021;12:800268. doi: 10.3389/fimmu.2021.800268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Wang E, Li Y, Ming R, et al. The prognostic value and immune landscapes of a m (6) a/m (5) C/m (1) A-related LncRNAs signature in head and neck squamous cell carcinoma. Front Cell Dev Biol. 2021;9:718974. doi: 10.3389/fcell.2021.718974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Gu X, Zhou H, Chu Q, Zheng Q, Wang J, Zhu H. Uncovering the association between m (5) C regulator-mediated methylation modification patterns and tumour microenvironment infiltration characteristics in hepatocellular carcinoma. Front Cell Dev Biol. 2021;9:727935. doi: 10.3389/fcell.2021.727935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Huang Z, Li J, Chen J, Chen D. Construction of prognostic risk model of 5-Methylcytosine-related Long non-coding RNAs and evaluation of the characteristics of tumor-infiltrating immune cells in breast Cancer. Front Genet. 2021;12:748279. doi: 10.3389/fgene.2021.748279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Meng L, Zhang Q, Huang X. Comprehensive analysis of 5-Methylcytosine profiles of messenger RNA in human high-grade serous ovarian Cancer by MeRIP sequencing. Cancer Manag Res. 2021;13:6005–6018. doi: 10.2147/CMAR.S319312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Wu XR, Chen Z, Liu Y, et al. Prognostic signature and immune efficacy of m (1) A-, m (5) C- and m (6) A-related regulators in cutaneous melanoma. J Cell Mol Med. 2021;25(17):8405–8418. doi: 10.1111/jcmm.16800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Geng Q, Wei Q, Shen Z, et al. Comprehensive analysis of the prognostic value and immune infiltrates of the three-m5C signature in Colon carcinoma. Cancer Manag Res. 2021;13:7989–8002. doi: 10.2147/CMAR.S331549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Blanco S, Kurowski A, Nichols J, Watt FM, Benitah SA, Frye M. The RNA-methyltransferase Misu (NSun2) poises epidermal stem cells to differentiate. PLoS Genet. 2011;7(12):e1002403. doi: 10.1371/journal.pgen.1002403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Lu L, Gaffney SG, Cannataro VL, Townsend J. Transfer RNA methyltransferase gene NSUN2 mRNA expression modifies the effect of T cell activation score on patient survival in head and neck squamous carcinoma. Oral Oncol. 2020;101:104554. doi: 10.1016/j.oraloncology.2019.104554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Lu L, Zhu G, Zeng H, Xu Q, Holzmann K. High tRNA Transferase NSUN2 gene expression is associated with poor prognosis in head and neck squamous carcinoma. Cancer Investig. 2018;36(4):246–253. doi: 10.1080/07357907.2018.1466896. [DOI] [PubMed] [Google Scholar]
- 126.Saplaoura E, Perrera V, Kragler F. Methylated RNA Immunoprecipitation assay to study m5C modification in Arabidopsis. J Vis Exp. 2020;159. [DOI] [PubMed]
- 127.Trevarton A, Zhou Y, Yang D, et al. Orthogonal assays for the identification of inhibitors of the single-stranded nucleic acid binding protein YB-1. Acta Pharm Sin B. 2019;9(5):997–1007. doi: 10.1016/j.apsb.2018.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Johnson BD, Yan X, Schauer DW, Orentas RJ. Dual expression of CD80 and CD86 produces a tumor vaccine superior to single expression of either molecule. Cell Immunol. 2003;222(1):15–26. doi: 10.1016/s0008-8749(03)00079-0. [DOI] [PubMed] [Google Scholar]
- 129.Maurya PK, Mishra A, Yadav BS, et al. Role of Y box Protein-1 in cancer: as potential biomarker and novel therapeutic target. J Cancer. 2017;8(10):1900–1907. doi: 10.7150/jca.17689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Tao Z, Ruan H, Sun L, et al. Targeting the YB-1/PD-L1 Axis to enhance chemotherapy and antitumor immunity. Cancer Immunol Res. 2019;7(7):1135–1147. doi: 10.1158/2326-6066.CIR-18-0648. [DOI] [PubMed] [Google Scholar]
- 131.Anreiter I, Mir Q, Simpson JT, Janga SC, Soller M. New twists in detecting mRNA modification dynamics. Trends Biotechnol. 2021;39(1):72–89. doi: 10.1016/j.tibtech.2020.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Chen L, Li Z, Zhang S, Zhang YH, Huang T, Cai YD. Predicting RNA 5-Methylcytosine sites by using essential sequence features and distributions. Biomed Res Int. 2022;2022:4035462. doi: 10.1155/2022/4035462. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The authors confirm that the data support the findings of this study are available within the article.