Abstract
Objective
To explore the diagnostic value and prognostic evaluation of the autophagy-related protein expression level among patients with sepsis comorbid with acute respiratory distress syndrome.
Methods
A total of 182 sepsis patients were admitted to Naval Medical Center from March 2016 to April 2020 and divided into the acute respiratory distress syndrome and non-ARDS groups. Immunoblotting was employed to identify the expression of autophagy-associated protein from participants' peripheral blood mononuclear cells. Multivariate linear regression analysis was used to examine the association between mortality and the protein expression in sepsis complicated with acute respiratory distress syndrome.
Results
Among the 182 patients with sepsis included in this study, 82 patients had acute respiratory distress syndrome and 100 patients did not have acute respiratory distress syndrome. We observed that microtubule-related protein 1A/1B LC3II, Beclin-1, RAB7, and LAMP2 protein expression was significantly decreased in septic patients with ARDS, and p62 was significantly increased. Further receiver operating characteristic curve analysis showed that autophagy-related proteins had a high recognition ability in sepsis complicated with acute respiratory distress syndrome. LAMP2 protein was the best among them, and its specificity was up to 91.46%. In this study, 38 of the 82 patients with sepsis complicated with acute respiratory distress syndrome died, with a mortality rate of 46.34%. We found that the autophagy level was further inhibited in the patients with death, LC3II, Beclin-1, and RAB7. However, the lysosomal-associated membrane protein 2 levels in the survival patients were remarkably higher than that in the dead patients. In addition, the p62 level was lower in survival patients as well. Our results indicated age and SOFA score were the independent risk factors for mortality in septic patients with acute respiratory distress syndrome.
Conclusion
The autophagy level is significantly inhibited in septic patients with acute respiratory distress syndrome, and autophagy-associated proteins LC3II, Beclin-1, RAB7, LAMP2, and p62 have good value for the diagnosis and prognosis evaluation of sepsis comorbid with acute respiratory distress syndrome.
1. Introduction
Sepsis refers to immune dysfunction and systemic inflammation caused by microbial infection and is a syndrome characterized by organ dysfunction and abnormal immune response to infection [1, 2]. There are more than 19 million cases worldwide every year, and the in-hospital mortality rate of sepsis patients is about 15%-25% [3, 4]. Organ dysfunction is caused by the local or systemic release of harmful mediators from the site of infection or away from the organ stimulating infection [5]. The lung is one of the most affected organs during sepsis, which may lead to acute respiratory distress syndrome (ARDS) [6]. ARDS is a clinical syndrome in critical patients, including acute respiratory failure (ARF), hypoxemia, and noncardiogenic lung oedema [7, 8]. So far, there are no effective treatments for ARDS. Therefore, it is very important to assess the biomarkers associated with sepsis-induced ARDS and to understand the molecular mechanisms of ARDS caused by sepsis.
Autophagy participates in the etiopathogenesis of ARDS. The autophagic process is a remarkably conserved cell protection activity which involves the transport of damaged proteins to lysosomes for degradation and circulation, so as to maintain the stability of cell structure, function, and metabolism. It is considered to be one of the main pathways of protein and organelle degradation and circulation [9, 10]. Recently, researchers have discovered that autophagic activation participates in the pathophysiological process of sepsis, which alleviates the excessive release of cytokines in sepsis and lung injury. This function may protect mice from the effects of acute lung injury (ALI)/ARDS that was induced by cecal ligation perforation (CLP) [11, 12], improves arterial oxygenation and vascular function, and inhibits IL-1β production. Autophagy levels could be identified via assessing autophagy flux levels and detecting lipid-modified forms of microtubule-related protein 1A/1B light chain 3 (LC3) and autophagosomes [13]. Elevated autophagy levels can promote the accumulation of autophagy-associated proteins LC3II and Beclin-1 [14]. Beclin-1 is an important autophagic protein, including two different subcomplexes with regulators Atg 14 and UV radiation resistance-associated gene protein (UVRAG), which responds to autophagosome formation and maturation, respectively [15]. Lysosomal-associated membrane protein 2 (LAMP2) and Ras-related protein Rab-7a (RAB7) are necessary for late autophagosomal maturation and fusion between autophagosome and lysosome [16, 17]. p62, as an autophagic biomarker, is the first protein discovered to have such an adaptor role in the autophagic process and is decomposed via autophagic stimulation [18]. Evidence has shown that there is a potential association between the suppression of autophagic activity and elevated p62 levels [19]. At present, few studies have reported the clinical significance of autophagy-associated protein expression in ARDS.
In this study, we assess the association between autophagy-related protein expression level and the diagnostic value and prognostic evaluation in sepsis patients comorbid with ARDS. Our study will provide more data for clinical understanding of the biomarkers that might be potentially used for ARDS prediction among sepsis patients.
2. Methods
2.1. Research Object
A total of 182 sepsis patients admitted to an academic medical center, Naval Medical Center, from March 2016 to April 2020 were selected. Inclusion criteria include all patients that met the diagnostic criteria for sepsis in the 2012 Save Sepsis Campaign: International Guidelines for the Management of Sepsis and Septic Shock [20]. Patients in the ARDS group also met the diagnostic criteria for ARDS in the Berlin Definition. The entire sufferers were above 18 years old with complete medical records. Exclusion criteria include those infected with human immunodeficiency virus, who died within 24 hours after admission to ICU, combined with hematopoietic diseases or malignant tumors, pregnant and lactating women, combined with mental disorders, and with severe malnutrition. The inclusion and exclusion criteria were based on previously published studies [21, 22]. According to the occurrence of ARDS, the sufferers were assigned into 2 groups, including 82 patients with ARDS and 100 patients without ARDS. The present research was accepted by the Ethical Board of our hospital, and the whole sufferers offered informed consent.
2.2. Peripheral Blood Collection and Monocyte Separation
Within 24 hours after admission, 5 ml of fasting venous blood was harvested from the entire sufferers in the morning by vacuo venous blood collection. The density gradient centrifugation method was used to separate mononuclear cells, and 2 ml of lymph cell separation liquor was supplemented into the round-bottom test tube, and venous blood and Hanks fluid were pressed 1 : 1. The anticoagulant diluted blood was slowly added into the separation solution tube along the tube wall with a capillary straw and centrifuged in a horizontal centrifuge at room temperature (20°C) for 20 min at 2000 r/min. A capillary straw was inserted into the white cell layer at the interface, and the cells were sucked out and added 1 ml Hanks liquid, and the cells were suspended. All our blood sample collection was progressed by well-trained nurses, and the methods were based on previous similar published studies.
2.3. Autophagy-Related Protein Expression Detection
Western blot (WB) was employed to identify the expression of autophagy-associated proteins LC3II, Beclin-1, RAB7, LAMP2, and p62 in mononuclear cells. Lysis was performed in a cellular lysate buffering solution (1x RIPA, CST, America) for 0.5 h, followed by centrifugation at 12,000 g at 4°C for 15 min to harvest the supernate. The protein level was identified via the Bradford Assay (Bio-RAD, America); 30 μg protein was treated with SDS-PAGE and afterwards moved onto PVDF film (Millipore, America). Posterior to the sealing under RT for 120 min via 5% skim milk involving 0.1% Tween 20, it was cultivated nightlong via the first antisubstance at 4°C. Anti-mouse primary antibodies of LC3II, Beclin-1, RAB7, LAMP2, and p62 (1 : 1000) were obtained from Abcam (Cambridge, UK). After incubation with an anti-mouse secondary antibody bound to horseradish peroxidase (1 : 5000, Santa Cruz Biotechnology, America), the protein was developed using an enhanced chemiluminescence reagent (Thermo Pierce, USA). Quantitative analyses were completed via the Bio-RAD Gel Doc 2000 System (America). β-Actin was utilized as inner control.
2.4. Main Outcome Measures
The main outcome of interest was ARDS incidence in sepsis patients. In addition, sepsis patients with comorbid ARDS may have increased risks of all-cause mortality.
2.5. Covariates
Demographic and clinical characteristics were determined at the baseline including age, gender, smoking history, body mass index (BMI), and other comorbid conditions. The other comorbid conditions included severe pneumonia, severe pancreatitis, abdominal infection, biliary infection, surgical trauma, chronic obstructive pulmonary disease (COPD), cardiac disease, chronic kidney disease, and liver cirrhosis.
2.6. Statistical Analysis
The data are expressed as the average ± SD, and the χ2 test is used for comparison of measurement data. Continuous variates were contrasted via two-tailed Student's t-test or one-way ANOVA. The contrast of nonnormal distribution data is studied via the nonparametric test of two separate specimens. A two-tailed P value < 0.05 had significance on statistics. Multivariate linear regression analysis was used to examine the association between mortality and the protein expression in sepsis complicated with acute respiratory distress syndrome. We used the AUC as an overall measure of discrimination of each variable. The entire statistic analysis was finished via SPSS 17.0 (America). GraphPad Prism 5 (America) was used to draw relevant pictures.
3. Results
3.1. Clinical Information Statistics
Table 1 displays characteristics for both the ARDS and non-ARDS groups. Among the 182 patients with sepsis included in this study, 82 patients had ARDS and 100 patients did not have ARDS. The means (SD) of age were 55.96 (8.49) and 58.01 (9.45) years in the ARDS and non-ARDS groups, respectively. Compared with the non-ARDS group, the patients in the ARDS group were more likely to have smoking history and higher creatinine, CRP, and APACHE II scores (P < 0.05). There was no remarkable diversity in comparison to other variables (P > 0.05) (Table 1).
Table 1.
Analyses of clinic data of two groups of patients.
| ARDS (n = 82) | Non-ARDS (n = 100) | χ 2/t | P | |
|---|---|---|---|---|
| Gender | 0.533 | 0.465 | ||
| Male | 39 (47.56) | 53 (53.00) | ||
| Female | 43 (52.44) | 47 (47.00) | ||
| Age | 55.96 ± 8.49 | 58.01 ± 9.45 | 1.774 | 0.078 |
| BMI (kg/m2) | 23.15 ± 4.04 | 22.88 ± 3.64 | 0.475 | 0.635 |
| Smoking history | 10.957 | 0.001 | ||
| Yes | 48 (58.54) | 34 (34.00) | ||
| No | 34 (41.46) | 66 (66.00) | ||
| Primary disease | 0.314 | 0.997 | ||
| Severe pneumonia | 20 (24.39) | 24 (24.00) | ||
| Severe pancreatitis | 17 (20.73) | 20 (20.00) | ||
| Abdominal infection | 16 (19.51) | 19 (19.00) | ||
| Biliary infection | 12 (14.63) | 14 (14.00) | ||
| Surgical trauma | 10 (12.19) | 12 (12.00) | ||
| Others | 7 (8.54) | 11 (11.00) | ||
| Chronic complications | 2.705 | 0.439 | ||
| COPD | 26 (31.71) | 16 (16.00) | ||
| Cardiac disease | 30 (36.58) | 35 (35.00) | ||
| Chronic renal failure | 12 (14.63) | 10 (10.00) | ||
| Liver cirrhosis | 24 (29.27) | 19 (19.00) | ||
| Creatinine (mmol/l) | 125.59 ± 44.59 | 104.18 ± 30.92 | 3.812 | <0.001 |
| Albumin (g/l) | 27.96 ± 4.21 | 29.14 ± 4.46 | 1.833 | 0.068 |
| White blood cell count | 18.15 ± 10.48 | 15.68 ± 8.52 | 1.751 | 0.082 |
| CRP | 128.17 ± 30.78 | 97.00 ± 27.56 | 7.200 | <0.001 |
| PCT | 15.22 ± 4.15 | 16.12 ± 4.22 | 1.445 | 0.150 |
| APACHE II score | 18.81 ± 2.95 | 13.54 ± 2.24 | 13.685 | <0.001 |
| SOFA score | 9.44 ± 3.87 | 8.46 ± 3.78 | 1.724 | 0.086 |
3.2. Expression of Autophagy-Related Protein in Septic Patients with ARDS
Western blot outcomes revealed that in the peripheral blood monocytes of sepsis sufferers with ARDS, the relative expression levels of LC3II, Beclin-1, RAB7, and LAMP2 were remarkably lower in contrast to the non-ARDS group (P < 0.05). On the contrary, the relative expression level of p62 in the peripheral blood monocytes of sepsis sufferers with ARDS was remarkably greater in contrast to the non-ARDS group (P < 0.05) (Figure 1).
Figure 1.
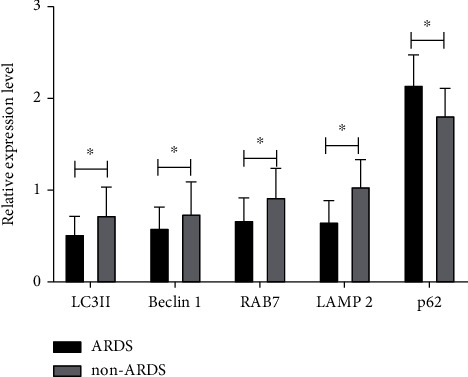
The expression of autophagy-related proteins in sepsis patients with ARDS. ∗P < 0.05.
3.3. Differential Diagnosis Value of Autophagy-Related Protein in Septic Patients with ARDS
The results of the ROC curve analysis are shown in Figure 2. The differential diagnosis value of five autophagy-associated proteins for sepsis sufferers with ARDS from high to low is LAMP2, p62, RAB7, LC3II, and Beclin-1, and the specific parameters are shown in Table 2. As shown in Table 2, the AUCs of LAMP2, p62, RAB7, LC3II, and Beclin-1 for the diagnosis of sepsis with ARDS are 0.853, 0.773, 0.746, 0.728, and 0.650, respectively.
Figure 2.
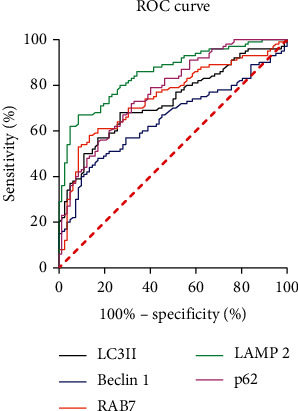
ROC curve of autophagy-associated protein in the diagnosis of sepsis with ARDS.
Table 2.
The differential diagnosis value of autophagy-related proteins in sepsis sufferers with ARDS.
| AUC | 95% CI | P | Sensitivity (%) | Specificity (%) | Diagnostic level | |
|---|---|---|---|---|---|---|
| LC3II | 0.728 | 0.656-0.801 | <0.001 | 68.00 | 73.17 | <0.631 |
| Beclin-1 | 0.650 | 0.570-0.729 | 0.001 | 48.00 | 82.93 | <0.815 |
| RAB7 | 0.746 | 0.675-0.818 | <0.001 | 58.00 | 86.59 | <0.888 |
| LAMP2 | 0.853 | 0.799-0.906 | <0.001 | 67.00 | 91.46 | <0.926 |
| p62 | 0.773 | 0.706-0.839 | <0.001 | 72.00 | 68.29 | >1.998 |
3.4. The Association between Autophagy-Related Protein and Death in Sepsis Patients with ARDS
Among the 82 sepsis sufferers with ARDS included in this research, the sufferers were separated into 2 groups as per whether there were deaths during hospitalization. Among them, there were 38 sufferers in the death group and 44 individuals in the survival group, with a mortality rate of 46.43%. The analysis found that the relative expression levels of LC3II, Beclin-1, RAB7, and LAMP2 in the mortality group were lower in contrast to the survival one (P < 0.05), whereas p62 was greater in contrast to the survival one (P < 0.05) (Figure 3).
Figure 3.
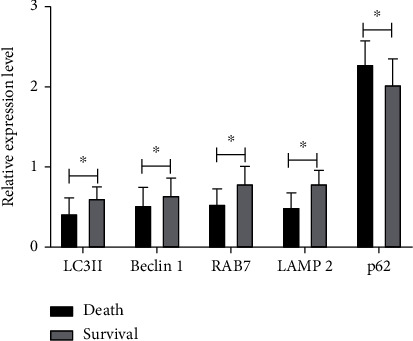
The association between autophagy-related proteins and the death of sepsis patients with ARDS. ∗P < 0.05.
3.5. The Prediction Significance of Autophagy-Related Protein in the Death of Sepsis Patients with ARDS
The results of the ROC curve analysis of autophagy-related proteins predicting the death of sepsis sufferers with ARDS are shown in Figure 4. The predictive value from high to low is LAMP2, RAB7, LC3II, p62, and Beclin-1, and the specific parameters are shown in Table 3. As shown, LAMP2, RAB7, LC3II, p62, and Beclin-1 predicted the AUC of death in sepsis patients with ARDS: 0.922, 0.833, 0.807, 0.745, and 0.708, respectively.
Figure 4.
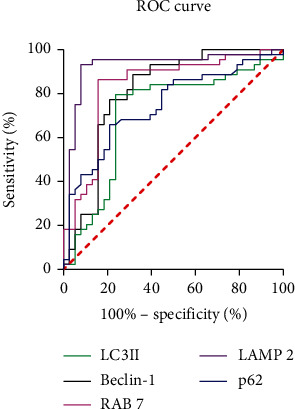
The ROC curve of autophagy-associated protein predicting the death of sepsis sufferers with ARDS.
Table 3.
The predictive value of autophagy-associated protein for the death of sepsis sufferers with ARDS.
| AUC | 95% CI | P | Sensitivity (%) | Specificity (%) | Diagnostic level | |
|---|---|---|---|---|---|---|
| LC3II | 0.807 | 0.706-0.909 | <0.001 | 88.64 | 68.42 | <0.465 |
| Beclin-1 | 0.708 | 0.588-0.829 | 0.001 | 79.55 | 76.32 | <0.592 |
| RAB7 | 0.833 | 0.739-0.928 | <0.001 | 86.36 | 84.21 | <0.639 |
| LAMP2 | 0.922 | 0.850-0.993 | <0.001 | 93.18 | 92.11 | <0.646 |
| p62 | 0.745 | 0.638-0.852 | <0.001 | 65.91 | 78.95 | >2.088 |
3.6. Analysis of Risk Factors for Death in Sepsis Patients with ARDS
Our team further studied the risk factors for mortality in sepsis sufferers with ARDS and incorporated all indicators into the logistics regression model for univariable and multivariable analyses. The outcomes of the univariable analyses are shown in Table 4. Age, CRP, PCT, APACHE II, SOFA, LC3II, Beclin-1, RAB7, LAMP2, and p62 are related to the death of patients with sepsis and ARDS. These indicators are further increased. For factor analysis, the results are shown in Table 5. In the end, only age and SOFA score were discovered to be independent risk factors for mortality in sepsis sufferers with ARDS.
Table 4.
Univariate analyses of death in sepsis sufferers with ARDS.
| B | S.E. | Wals | df | Sig. | Exp (B) | 95% CI | ||
|---|---|---|---|---|---|---|---|---|
| Lower limit | Upper limit | |||||||
| Sex | -0.610 | 0.449 | 1.842 | 1 | 0.175 | 0.543 | 0.225 | 1.311 |
| Age | 0.114 | 0.034 | 11.479 | 1 | 0.001 | 1.121 | 1.049 | 1.198 |
| BMI | 0.084 | 0.057 | 2.177 | 1 | 0.140 | 1.088 | 0.973 | 1.217 |
| Smoking history | 0.563 | 0.456 | 1.523 | 1 | 0.217 | 1.756 | 0.718 | 4.293 |
| Primary severe pneumonia | 0.194 | 0.515 | 0.142 | 1 | 0.706 | 1.214 | 0.443 | 3.331 |
| Primary severe pancreatitis | 0.334 | 0.546 | 0.374 | 1 | 0.541 | 1.397 | 0.479 | 4.074 |
| Primary abdominal infection | 0.495 | 0.562 | 0.776 | 1 | 0.378 | 1.640 | 0.546 | 4.932 |
| Primary biliary infection | -0.222 | 0.633 | 0.123 | 1 | 0.726 | 0.801 | 0.232 | 2.767 |
| Primary surgical trauma | -1.386 | 0.825 | 2.824 | 1 | 0.093 | 0.250 | 0.050 | 1.259 |
| Other primary diseases | 1.157 | 0.868 | 1.777 | 1 | 0.183 | 3.182 | 0.580 | 17.452 |
| Complicated with COPD | 0.671 | 0.481 | 1.947 | 1 | 0.163 | 1.957 | 0.762 | 5.022 |
| Complicated with cardiac disease | -0.191 | 0.461 | 0.172 | 1 | 0.678 | 0.826 | 0.334 | 2.040 |
| Complicated with chronic renal failure | 0.172 | 0.625 | 0.076 | 1 | 0.783 | 1.187 | 0.349 | 4.044 |
| Complicated with liver cirrhosis | -1.025 | 0.521 | 3.881 | 1 | 0.049 | 0.359 | 0.129 | 0.995 |
| Creatinine | 0.001 | 0.005 | 0.010 | 1 | 0.920 | 1.001 | 0.991 | 1.010 |
| Albumin | -0.033 | 0.053 | 0.377 | 1 | 0.539 | 0.968 | 0.872 | 1.074 |
| White blood cell count | 0.018 | 0.021 | 0.696 | 1 | 0.404 | 1.018 | 0.976 | 1.062 |
| CRP | 0.036 | 0.009 | 14.894 | 1 | 0.000 | 1.037 | 1.018 | 1.056 |
| PCT | 0.342 | 0.083 | 17.140 | 1 | 0.000 | 1.408 | 1.197 | 1.655 |
| APACHE II | 0.568 | 0.131 | 18.716 | 1 | 0.000 | 1.764 | 1.364 | 2.282 |
| SOFA | 0.583 | 0.131 | 19.646 | 1 | 0.000 | 1.791 | 1.384 | 2.317 |
| LC3II | -7.104 | 1.760 | 16.302 | 1 | 0.000 | 0.001 | 0.000 | 0.026 |
| Beclin-1 | -2.832 | 1.138 | 6.196 | 1 | 0.013 | 0.059 | 0.006 | 0.548 |
| RAB7 | -7.891 | 1.871 | 17.787 | 1 | 0.000 | 0.000 | 0.000 | 0.015 |
| LAMP2 | -11.109 | 2.318 | 22.962 | 1 | 0.000 | 0.000 | 0.000 | 0.001 |
| p62 | 2.912 | 0.887 | 10.783 | 1 | 0.001 | 18.401 | 3.235 | 104.659 |
Table 5.
Multivariate analyses of death in sepsis sufferers with ARDS.
| B | S.E. | Wals | df | Sig. | Exp (B) | 95% CI | ||
|---|---|---|---|---|---|---|---|---|
| Lower limit | Upper limit | |||||||
| Age | 0.154 | 0.068 | 5.152 | 1 | 0.023 | 1.166 | 1.021 | 1.332 |
| CRP | 0.001 | 0.021 | 0.002 | 1 | 0.961 | 1.001 | 0.961 | 1.042 |
| PCT | 0.123 | 0.133 | 0.854 | 1 | 0.356 | 1.131 | 0.871 | 1.468 |
| APACHE II | 0.395 | 0.273 | 2.098 | 1 | 0.148 | 1.485 | 0.870 | 2.534 |
| SOFA | 0.695 | 0.248 | 7.881 | 1 | 0.005 | 2.004 | 1.233 | 3.255 |
| LC3II | 0.313 | 3.156 | 0.010 | 1 | 0.921 | 1.367 | 0.003 | 663.833 |
| Beclin-1 | 6.623 | 3.399 | 3.796 | 1 | 0.051 | 751.852 | 0.961 | 588117.797 |
| RAB7 | -4.394 | 3.894 | 1.273 | 1 | 0.259 | 0.012 | 0.000 | 25.502 |
| LAMP2 | -4.721 | 3.328 | 2.012 | 1 | 0.156 | 0.009 | 0.000 | 6.059 |
| p62 | -2.896 | 2.514 | 1.327 | 1 | 0.249 | 0.055 | 0.000 | 7.624 |
4. Discussion
The attributable mortality of ARDS in sepsis sufferers is the most common risk factor for ARDS. Compared with sepsis sufferers without ARDS or non-sepsis-related ARDS, sepsis morbidity and mortality of toxicosis-related ARDS have increased significantly, even as high as 60% [20, 21]. After decades of research and a large number of preclinical and clinical trials, sepsis and ARDS still have no specific and effective drug treatments and basically supported management [22, 23]. Therefore, early diagnosis is essential to ameliorate the prognosis of sepsis sufferers.
In recent years, the autophagic causal links in sepsis sufferers have drawn more and more attention. The autophagic process includes autophagosome forming, autophagosomal-lysosomal fusion, and decomposition products [24]. The autophagic process not only realizes the elimination of damaged protein and cell organs but also realizes the elimination of microbes and causative agents in cytoplasm [25]. In sepsis sufferers, autophagic processes have always been considered an adaptive protection process limiting cellular injury and programmed cell death [26]. ARDS is a serious form of ALI. Pulmonary damage is one of the most commonly seen complicating diseases of sepsis sufferers clinically. It is featured by the releasing of substantial inflammation factors, accompanied by an elevation of programmed cell death and lung oedema. Studies have found that induction of autophagy can protect mice from LPS and mechanical ventilation-triggered ALI and improve artery oxygenation and vessel functions [27]. However, there are currently few clinical studies on autophagy sepsis sufferers with ARDS, and there is a lack of relevant clinical research evidence on whether autophagy-related markers have an effect on the early recognition and prognostic evaluation of sepsis complicated by ARDS.
Herein, our team assessed the diagnostic and prognostic significance of 5 autophagy-related proteins in sepsis complicated by ARDS. The expression of LC3II, Beclin-1, RAB7, and LAMP2 in sepsis patients with ARDS was significantly reduced; p62 was significantly increased, which suggests that autophagy was inhibited in patients with sepsis and ARDS. Further ROC curve analysis results showed that autophagy-related proteins have a higher ability to recognize sepsis with ARDS, and the best one was LAMP2, which had a specificity of 91.46%. Biomarkers can not only be used for early diagnosis of diseases but also predict the clinical outcome of patients. In this study, 38 deaths occurred in 82 patients with sepsis and ARDS, with a mortality rate of 46.34%. We found that the autophagy level in the patients who died was further inhibited, and the surviving patients had LC3II, Beclin-1, RAB7, and LAMP2 levels significantly higher than those in dead patients, and p62 was lower than that in dead patients. ROC analysis results show that the most valuable autophagy-related protein that predicts the death of sepsis sufferers with ARDS is still LAMP2. In the analysis of risk factors for mortality in sepsis sufferers with ARDS, only age and SOFA score were found to be independent risk factors. Previous studies indicated that environmental pollution, vitamin D deficiency, and smoking history may associate with ARDS. However, in our study, due to the limitation of relative information, we could not observe such associations [28]. LAMP2 is a 410 AA, greatly glycosylated protein, which along with LAMP1 accounts for approximately half of the lysosomal membranous protein. LAMP2 participates in lysosome biogenesis and mediates the fusion of lysosomes and autophagosomes [29]. A clinical data study in 2018 found that [29] increased lysosomal gene expression during sepsis was related to unsatisfactory prognostic results. The 5 genes they found that increased in death patients were ATP6AP1, CD63, LAMP1, LAMP2, and SLC1A1. This result is exactly the opposite of ours. The specific reasons have yet to be analyzed. Among their research subjects, they selected patients with SIRS and sepsis suspected of infection in the medical intensive care unit. There was a certain difference with us, and we had not found their sample collection time. In addition, their detection object was a lysosomal gene, which was not consistent with the protein we detected. During the process of gene translation into protein, it will be interfered with by many factors. But these speculations need further research and verification.
In this study, we did observe associations between several biomarkers and the risk of ARDS among sepsis patients. Thus, for the general population with sepsis, these results suggest that carefully and continually monitoring these biomarkers among patients with sepsis is important. Considering the additional benefits of these biomarkers in predicting the risk for long-term morbidity and mortality after sepsis patients comorbid with ARDS, monitoring these biomarkers in clinical practice is necessary.
In conclusion, the autophagy level is significantly inhibited in sepsis patients with ARDS, and autophagy-associated proteins LC3II, Beclin-1, RAB7, LAMP2, and p62 have good value for the diagnosis and prognosis evaluation of sepsis patients with ARDS.
Abbreviation
- ARDS:
Acute respiratory distress syndrome
- LC3:
Light chain 3
- RAB7:
Ras-related protein Rab-7a
- LAMP2:
Lysosomal-associated membrane protein 2
- ALI:
Acute lung injury
- CLP:
Cecal ligation perforation
- SIRS:
Systemic inflammatory response syndrome
- WB:
Western blot
- CST:
Cell Signaling Technology
- PVDF:
Polyvinylidene fluoride
- RT:
Room temperature
- AA:
Amino acid
- ARF:
Acute respiratory failure
- PAGE:
Polyacrylamide gel electrophoresis
- UVRAG:
UV radiation resistance-associated gene protein.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Authors' Contributions
Jia-Li Xu and Xue-Lu Yu are the co-first authors.
References
- 1.Englert J. A., Bobba C., Baron R. M. Integrating molecular pathogenesis and clinical translation in sepsis-induced acute respiratory distress syndrome. JCI Insight . 2019;4(2) doi: 10.1172/jci.insight.124061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.van der Poll T., van de Veerdonk F. L., Scicluna B. P., Netea M. G. The immunopathology of sepsis and potential therapeutic targets. Nature Reviews Immunology . 2017;17(7):407–420. doi: 10.1038/nri.2017.36. [DOI] [PubMed] [Google Scholar]
- 3.Cecconi M., Evans L., Levy M., Rhodes A. Sepsis and septic shock. The Lancet . 2018;392(10141):75–87. doi: 10.1016/S0140-6736(18)30696-2. [DOI] [PubMed] [Google Scholar]
- 4.Prescott H. C., Angus D. C. Enhancing recovery from sepsis: a review. Journal of the American Medical Association . 2018;319(1):62–75. doi: 10.1001/jama.2017.17687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Aziz M., Jacob A., Yang W. L., Matsuda A., Wang P. Current trends in inflammatory and immunomodulatory mediators in sepsis. Journal of Leukocyte Biology . 2013;93(3):329–342. doi: 10.1189/jlb.0912437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Martin-Loeches I., Levy M. M., Artigas A. Management of severe sepsis: advances, challenges, and current status. Drug Design, Development and Therapy . 2015;9, article 2079:2088. doi: 10.2147/DDDT.S78757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Force A. D. T., Ranieri V. M., Rubenfeld G. D., et al. Acute respiratory distress syndrome. JAMA . 2012;307(23):2526–2533. doi: 10.1001/jama.2012.5669. [DOI] [PubMed] [Google Scholar]
- 8.Matthay M. A., Zemans R. L., Zimmerman G. A., et al. Acute respiratory distress syndrome. Nature Reviews Disease Primers . 2019;5(1):p. 18. doi: 10.1038/s41572-019-0069-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dong A., Chen H., Xie K., Yu Y. Role of autophagy in lung injury in septic mice. Chinese Journal of Anesthesiology . 2015;35(9):1124–1127. [Google Scholar]
- 10.Monteleon C. L., Agnihotri T., Dahal A., et al. Lysosomes support the degradation, signaling, and mitochondrial metabolism necessary for human epidermal differentiation. Journal of Investigative Dermatology . 2018;138(9):1945–1954. doi: 10.1016/j.jid.2018.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhao H., Chen H., Xiaoyin M., et al. Autophagy activation improves lung injury and inflammation in sepsis. Inflammation . 2019;42(2):426–439. doi: 10.1007/s10753-018-00952-5. [DOI] [PubMed] [Google Scholar]
- 12.Wang X., Liu F., Xu M., Wu L. Penehyclidine hydrochloride alleviates lipopolysaccharide-induced acute respiratory distress syndrome in cells via regulating autophagy-related pathway. Molecular Medicine Reports . 2020;23(2) doi: 10.3892/mmr.2020.11739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Akhter M. S., Uddin M. A., Kubra K. T., Barabutis N. Autophagy, unfolded protein response and lung disease. Current research in cell biology . 2020;1, article 100003 doi: 10.1016/j.crcbio.2020.100003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mizushima N. Autophagy: process and function. Genes & Development . 2007;21(22):2861–2873. doi: 10.1101/gad.1599207. [DOI] [PubMed] [Google Scholar]
- 15.Qu Y., Wang X., Zhu Y., et al. ORF3a-mediated incomplete autophagy facilitates severe acute respiratory syndrome coronavirus-2 replication. Frontiers in Cell and Developmental Biology . 2021;9 doi: 10.3389/fcell.2021.716208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Choi A. M. K., Ryter S. W., Levine B. Autophagy in human health and disease. The New England Journal of Medicine . 2013;368(7):651–662. doi: 10.1056/NEJMra1205406. [DOI] [PubMed] [Google Scholar]
- 17.Gutierrez M. G., Munafó D. B., Berón W., Colombo M. I. Rab7 is required for the normal progression of the autophagic pathway in mammalian cells. Journal of Cell Science . 2004;117(13):2687–2697. doi: 10.1242/jcs.01114. [DOI] [PubMed] [Google Scholar]
- 18.Chen H., Hu Y., Xie K., et al. Effect of autophagy on allodynia, hyperalgesia and astrocyte activation in a rat model of neuropathic pain. International Journal of Molecular Medicine . 2018;42(4):2009–2019. doi: 10.3892/ijmm.2018.3763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang Q. J., Ding Y., Kohtz S., et al. Induction of autophagy in axonal dystrophy and degeneration. Journal of Neuroscience . 2006;26(31):8057–8068. doi: 10.1523/JNEUROSCI.2261-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dellinger R. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock, 2012. Intensive Care Medicine . 2013;39(2):165–228. doi: 10.1007/s00134-012-2769-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Panico F. F., Troster E. J., Oliveira C. S., et al. Risk factors for mortality and outcomes in pediatric acute lung injury/acute respiratory distress syndrome. Pediatric Critical Care Medicine . 2015;16(7):e194–e200. doi: 10.1097/PCC.0000000000000490. [DOI] [PubMed] [Google Scholar]
- 22.Brochard L., Roudot-Thoraval F., Roupie E., et al. Tidal volume reduction for prevention of ventilator-induced lung injury in acute respiratory distress syndrome. American Journal of Respiratory and Critical Care Medicine . 1998;158(6):1831–1838. doi: 10.1164/ajrccm.158.6.9801044. [DOI] [PubMed] [Google Scholar]
- 23.Klionsky D. J., Abdel-Aziz A. K., Abdelfatah S., et al. Guidelines for the use and interpretation of assays for monitoring autophagy (4th edition) Autophagy . 2021;17(1):1–382. doi: 10.1080/15548627.2020.1797280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Karagiannidis I., Kataki A., Glustianou G., et al. Extended cytoprotective effect of autophagy in the late stages of sepsis and fluctuations in signal transduction pathways in a rat experimental model of kidney injury. Shock . 2016;45(2):139–147. doi: 10.1097/SHK.0000000000000505. [DOI] [PubMed] [Google Scholar]
- 25.Oami T., Watanabe E., Hatano M., et al. Blocking liver autophagy accelerates apoptosis and mitochondrial injury in hepatocytes and reduces time to mortality in a murine sepsis model. Shock . 2018;50(4):427–434. doi: 10.1097/SHK.0000000000001040. [DOI] [PubMed] [Google Scholar]
- 26.Nosaka N., Martinon D., Moreira D., Crother T. R., Arditi M., Shimada K. Autophagy protects against developing increased lung permeability and hypoxemia by down regulating inflammasome activity and IL-1β in LPS plus mechanical ventilation-induced acute lung injury. Frontiers in Immunology . 2020;11:p. 207. doi: 10.3389/fimmu.2020.00207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Alcalai R., Arad M., Wakimoto H., et al. LAMP2cardiomyopathy: consequences of impaired autophagy in the heart. Journal of the American Heart Association . 2021;10(17, article e018829) doi: 10.1161/JAHA.120.018829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Confalonieri M., Salton F., Fabiano F. Acute respiratory distress syndrome. European Respiratory Review . 2017;26(144, article 160116) doi: 10.1183/16000617.0116-2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tolbert T., Marion C. R., Andrews N., et al. A49. Critical care: looking for clues-biomarkers in ARDS, sepsis, and acute critical illness . American Thoracic Society 2018 International Conference; 2018. Rising lysosomal gene expression during sepsis is associated with worse patient outcomes; p. p. A1809. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.


