Abstract
Purpose
Hepatocellular carcinoma (HCC), has a very high mortality rate and is the most common type of liver cancer. Clotrimazole, a traditional antifungal drug, has garnered considerable attention as a therapeutic strategy for HCC. However, its effects against the migration and invasion of HCC cells as well as the associated underlying mechanisms remain unclear. Therefore, in this study, we investigated its effects on HCC and attempted to elucidate the underlying molecular mechanisms.
Methods
CCK-8 was used to investigate the inhibitory effect of clotrimazole on the proliferation of different types of HCC cells, and wound healing and transwell assays were performed to investigate its inhibitory effect on the invasion and migration of the HCC cells. Further, western blotting was employed to detect changes in the expression levels of epithelial mesenchymal transition (EMT)-related proteins, extracellular-regulated protein kinases (ERK), p-ERK, p65, and p-p65. We also used ERK activators in combination with clotrimazole to treat the HCC cell lines.
Results
Clotrimazole inhibited the invasion and migration of HCC cells, and mechanistically, it exerted these anti-tumor effects via EMT by repressing ERK phosphorylation.
Conclusion
These findings suggest that clotrimazole inhibits HCC metastasis by repressing EMT in an ERK dephosphorylation-dependent manner.
Keywords: hepatocellular carcinoma, clotrimazole, extracellular-regulated protein kinases, migration, invasion, cancer
Introduction
Liver cancer is the third leading cause of cancer-related deaths and the sixth most prevalent type of cancer worldwide.1 Particularly, hepatocellular carcinoma (HCC) is the most common type of liver cancer.2 Even though surgical resection is the first line of treatment for HCC, most patients with HCC are often diagnosed at an advanced stage of the disease; thus, they are poor candidates for surgical resection,3,4 and their treatment options are very limited.5 Despite recent advances in diagnostic and therapeutic strategies, such as the development of transcatheter arterial chemoembolization and the multi-kinase inhibitor, sorafenib, HCC survival rates remain low owing to high recurrence rates and metastasis.6,7 These limitations necessitate the development of novel treatment strategies that offer the possibility to effectively reduce HCC recurrence and metastasis to the end of prolonging the overall survival of patients with HCC.
In recent years, our understanding of the anti-tumor effects of antifungal drugs has advanced significantly.8–10 Increasing evidence suggest that antifungals, such as ketoconazole (used alone or in combination with other drugs), have strong anti-tumor effects.11 Specifically, the anti-tumor effects of antifungals were originally discovered in prostate cancer; however, similar effects have been observed in other cancers, including breast, colon, and bladder cancers.12–15 As a broad-spectrum antifungal agent, clotrimazole exerts a strong inhibitory effect against several fungi, especially Candida albicans.16 Additionally, it has attracted considerable attention as a cancer treatment option.17 Reportedly, it inhibits the ability of breast cancer cells to proliferate and metastasise in vitro.8,15 However, its effect on the invasion and metastasis of HCC is unclear, implying that studies are needed to determine whether it can be a potential treatment for HCC. In this study, we demonstrate that clotrimazole can inhibit the migration and invasion of HCC cells by inhibiting ERK/p65-mediated epithelial–mesenchymal transition (EMT). This finding may provide new ideas for the treatment of HCC.
Materials and Methods
Reagents
Primary antibodies against E-cadherin, N-cadherin, and vimentin, as well as goat anti-rabbit and goat anti-mouse secondary antibodies, were purchased from Proteintech Group (Wuhan, China). Primary antibodies that recognize NF-κB-p65, phospho-NF-κB p65 (Ser536), ERK1/2, and phospho-p44/42 MAPK (ERK1/2; Thr202/Tyr204) were purchased from Cell Signaling Technology (Danvers, MA, USA). Clotrimazole was purchased from Mecklin (Shanghai, China), while honokiol was purchased from MedChemExpress (Shanghai, China).
Cell Culture
Human liver cancer cell lines [HuH7, SMCC-7721 (7721), MHCC 97H (97H), and Hep3B] were obtained from the Chinese Academy of Sciences (Shanghai, China). The HuH7 and Hep3B cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) containing 100 μg/mL penicillin/streptomycin and 10% fetal bovine serum (FBS). The cells were cultured at 37 °C in a humidified atmosphere of 5% CO2.
Cell Growth Assay
The effect of clotrimazole on the proliferation of HCC cells was determined using the Cell Counting Kit-8 (CCK-8) (Beyotime; Shanghai, China). Briefly, HCC cells (1×103 cells/well) were cultured in 96-well plates and treated with different concentrations of clotrimazole (10, 20, 30, 40, 50, 60, 80, and 100 μM) dissolved in DMSO (0.1% v/v) or with DMEM only (0.1% v/v DMSO) as the control treatment. After 24 and 48 h, 10 μL of the CCK-8 reagent was added to each well, and after incubation for 1 h at 37 °C, absorbance measurements were performed at 450 nm using a micro-plate reader (Model 3550; Bio-Rad; Hercules, CA, USA). The relative proliferation ratio of the cells was calculated as follows: (experimental absorbance value/control absorbance value) × 100%. At least three independent tests, each involving six wells, were performed.
Wound Healing/Scratch Assay
HCC cell migration was examined by performing a wound healing assay. Briefly, HCC cells were seeded in 6-well plates. Thereafter, a long, vertical wound was inflicted on the monolayer using a 10 μL pipette tip. Following wound induction, the cells were treated with different concentrations of clotrimazole, honokiol, or DMEM only (0.1% v/v DMSO) as the control treatment for 48 h. After the cells reached confluence, they were observed microscopically (200×), and the wound closure ratio was calculated as follows: (0 h wound width - 48 h wound width)/0 h wound width.
Transwell Assay
Transwell assays were performed using 8-μm polycarbonate transwell filters (Coring; Cambridge, MA, USA). In brief, Matrigel (BD Biosciences; Bedford, MA, USA) was dissolved overnight at 4 °C. The next day, 150 μL of the Matrigel solution was added to the wells of a 24-well plate reader. Thereafter, the plates were incubated at 37 °C for 30 min followed by clotrimazole treatment for 24 h, HCC cells were added to the inner chamber and DMEM medium, containing 10% FBS, was added to the lower chamber. This was followed by incubation for 24 h, after which the non-invading cells were wiped off, while the invading cells were fixed in paraformaldehyde for 15 min, stained with crystal violet for 15 min, and counted (five randomly selected fields) at 200× magnification using a microscope.
Western Blot Analysis
Proteins were separated on a 12% SDS-polyacrylamide gel and thereafter transferred onto PVDF membranes (Merck Millipore; Carrigtwohill, Ireland), which were then blocked in skimmed milk at 37 °C for 1 h and thereafter incubated with different primary antibodies (E-cadherin, N-cadherin, vimentin, NF-κB- p65, p-p65, Erk1/2, and p-Erk1/2). In the next step, the membranes were washed three times with 0.1% TBST and further incubated in goat anti-rabbit or goat anti-mouse secondary antibodies for 2 h. Proteins of interest were then visualized using an enhanced chemiluminescence kit (Millipore; Billerica, MA, USA) and via autoradiography. Densitometric analysis was performed using Image J software (GE Healthcare; Buckinghamshire, UK).
Statistical Analysis
SPSS software v17.0 (IBM Inc., Chicago, IL, USA) was used for statistical analysis. All the data were expressed as mean ± standard deviation. Differences between two groups were assessed by performing Student’s t-tests, while one-way ANOVA was performed to compare multiple groups. Statistical significance was set at P < 0.05.
Results
Clotrimazole Inhibits the Proliferation of HCC Cells
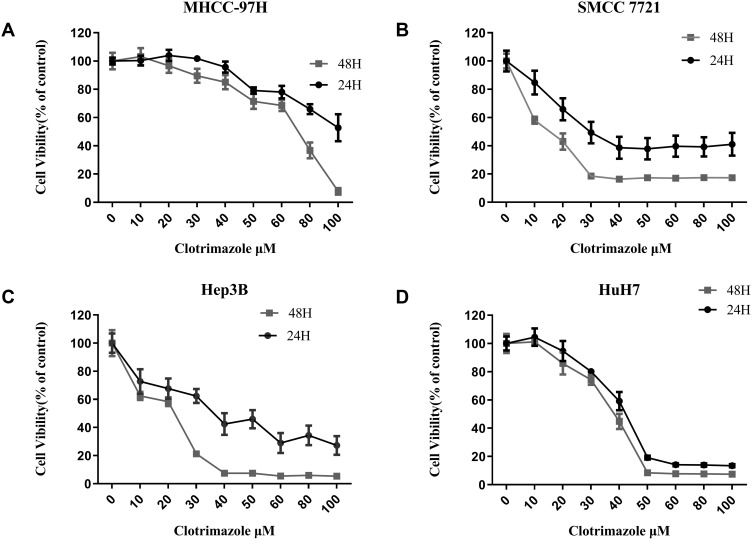
Several HCC cell lines were treated with different concentrations of clotrimazole for 24 h and 48 h to explore the effect of the drug on the proliferation of the cell lines. Thus, Cell Counting Kit-8 (CCK-8) assays demonstrated that the treatment of HCC cells with clotrimazole for 24 and 48 h inhibited their proliferation in a dose-dependent manner (Figure 1). The viabilities of 97H, 7721, Hep3B, and HuH7 cells treated for 48 h were lower than those of their counterparts that were treated for 24 h (Figure 1A–D). These findings suggested that clotrimazole inhibits the proliferation of HCC cells.
Figure 1.
Clotrimazole inhibits the proliferation of HCC cells. (A–D) The proliferation of 97H (A), 7721 (B), Hep3B (C), and HuH7 (D) cell lines with the different concentration of clotrimazole treatment assayed by CCK8. Absorbances were measured at 24 h and 48 h after treatment with clotrimazole.
Clotrimazole Suppresses the Migration and Invasion of HCC Cells
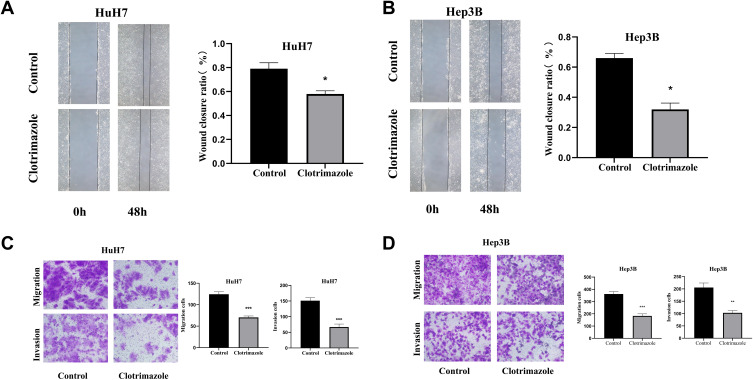
We next employed wound healing assays to examine the effect of clotrimazole on the invasion of HCC cells (Hep3B and HuH7 cell lines). Specifically, HCC cells were wounded, followed by clotrimazole (20 μM) treatment for 48 h and microscopic analysis. Compared with the control group, the scratches were wider in HCC cells after clotrimazole treatment (Figure 2A and B). Additionally, the assessment of the invasion and migration capacities of the HCC cells via Transwell assays showed that with or without the addition of Matrigel, fewer HCC cells crossed the membrane after 48 h of clotrimazole treatment compared with the control group (Figure 2C and D). These results demonstrated that clotrimazole inhibits the invasion and migration capacities of HCC cells.
Figure 2.
Clotrimazole inhibits migration and invasion of HCC cells. (A and B) The effect of clotrimazole (20μM) on the migration of HuH7 (A) and Hep3B (B) cell lines detected by wound healing assay. (C and D) The effect of clotrimazole on the migration and invasion of HuH7 (C) and Hep3B (D) cell lines detected by transwell assay. The results of quantification are expressed as the mean ± SD (n=3), *P<0.05, **P< 0.01, ***P<0.001 vs the control group.
Clotrimazole Regulates EMT and Represses ERK Phosphorylation
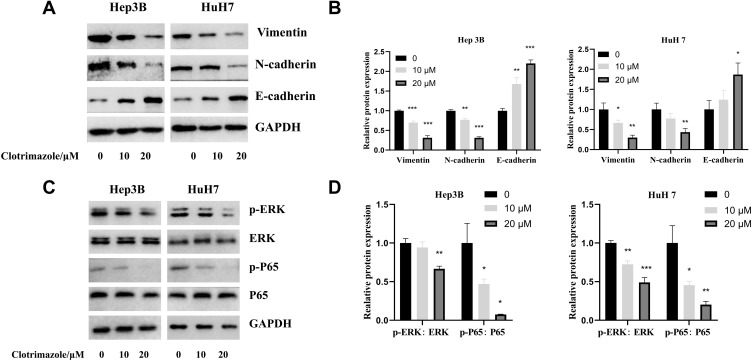
To further explore the mechanisms by which clotrimazole possibly inhibits the migration and invasion of HCC cells, we examined the molecules associated with EMT via western blot analysis. Thus, we observed that clotrimazole significantly inhibited the expression of vimentin and N-cadherin in HCC cells, while significantly enhancing the expression of E-cadherin (Figure 3A). Further, in clotrimazole-treated HCC cells (HuH7 and Hep3B), the expression of ERK and p65 did not change significantly. However, phosphorylated ERK and p65 were both significantly downregulated (Figure 3B). These findings suggested that clotrimazole regulates EMT and represses ERK phosphorylation.
Figure 3.
Clotrimazole regulates the EMT and ERK pathways. (A and B) The Vimentin, N-cadherin and E-cadherin levels in HCC cells treated with clotrimazole. (C and D) The ERK, p-ERK, p65 and p-p65 levels in HCC cells treated with clotrimazole. The results of quantification are expressed as the mean ± SD (n=3), *P<0.05, **P< 0.01, ***P<0.001 vs the control group (clotrimazole 0μM group).
Clotrimazole Inhibits the Migration and Invasion of HCC Cells via the ERK Pathway
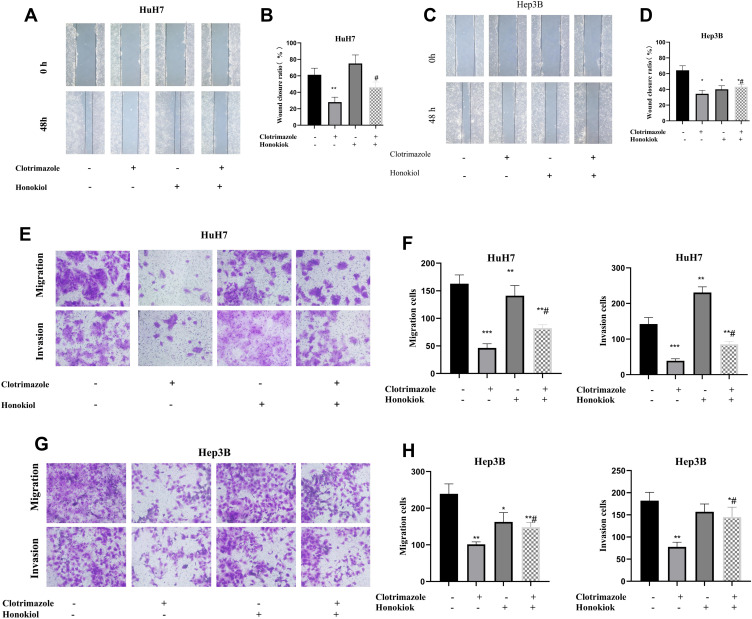
To verify whether clotrimazole inhibits HCC metastasis via the ERK pathway, we utilized the ERK activator honokiol. Treating wounded HCC cells with a combination of honokiol and clotrimazole resulted in narrower wounds than was the case with cells treated with clotrimazole only (Figure 4A–D). Additionally, transwell assays revealed that more HCC cells crossed the membrane after the honokiol-clotrimazole combined treatment compared with the observations made after the clotrimazole-only treatment (Figure 4E–H). These findings indicated that the activation of the ERK pathway can partially reverse the inhibitory effect of clotrimazole on HCC metastasis.
Figure 4.
Clotrimazole inhibits migration and invasion of HCC cells through the ERK pathway. (A and B) The effect of clotrimazole (20μM) alone or combinate with honokiol (10μM) on HuH7 cells migration detected by wound healing assay. (C and D) The effect of clotrimazole (20μM) alone or combinate with honokiol (10μM) on Hep3B cells migration detected by wound healing assay. (E and F) The effect of clotrimazole alone or combinate with honokiol (10μM) on HuH7 cells migration and invasion detected by transwell assay. (G and H) The effect of clotrimazole alone or combinate with honokiol (10μM) on Hep3B cells migration and invasion detected by transwell assay. The results of quantification are expressed as the mean ± SD (n=3), *P<0.05, **P< 0.01, ***P<0.001 vs the control group (without clotrimazole and honokiol group), #P<0.05 vs clotrimazole treated group.
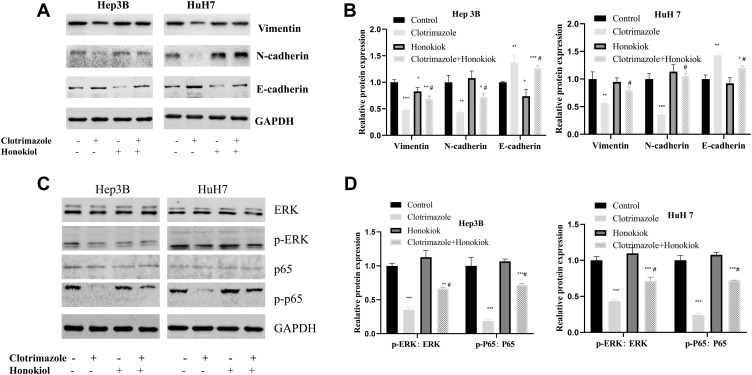
Clotrimazole Regulates EMT Through the ERK Pathway
The expression levels of vimentin, N-cadherin, and E-cadherin proteins (Figure 5A and B), as well as those of p-p65 and p-ERK (Figure 5C and D) were significantly upregulated in HCC cells treated with a combination of honokiol and clotrimazole compared with clotrimazole-only treated HCC cells treated. However, both treatments did not alter the expression levels of total p65 and ERK (Figure 5C and D). These findings suggested that honokiol reverses the effect of clotrimazole on EMT in HCC cells, indicating its involvement in the regulation of HCC cell-associated EMT through the regulation of the ERK pathway.
Figure 5.
Clotrimazole regulates EMT through the ERK pathway. (A and B) The Vimentin, N-cadherin, and E-cadherin levels in HCC cells treated with clotrimazole (20μM) alone or combinate with honokiol (10μM). (C and D) The ERK, p-ERK, p65 and p-p65 levels in HCC cells treated with clotrimazole alone or combinate with honokiol (10μM). The results of quantification are expressed as the mean ± SD (n=3), *P<0.05, **P< 0.01, ***P<0.001 vs the control group (without clotrimazole and honokiol group), #P<0.05 vs clotrimazole treated group.
Discussion
HCC is an aggressive form of cancer that is highly malignant.18 In this study, we investigated the effect of clotrimazole on HCC metastasis as well as the mechanisms underlying these effects. Specifically, clotrimazole is a broad-spectrum antifungal agent with strong inhibitory effects against a variety of fungi, especially C. albicans. Reportedly, it inhibits the development of malignant melanoma and breast cancer through its impacts on tumor proliferation and metastasis, which are mediated by the inhibition of PI3K and glycolysis.19–22 However, studies regarding its effects on HCC metastasis are limited.
Given that it has been previously reported that clotrimazole suppresses the invasion of human breast cancer cells,8,19 in this study, we investigated whether it could inhibit the migration and invasion of HCC cells. Thus, we observed that it inhibits the proliferation of a variety of HCC cell lines; this is consistent with its inhibitory effects on human breast cancer. Specifically, it preferentially inhibits human breast cancer cell proliferation, viability, and glycolysis.19 Our findings also indicated that clotrimazole-treated HCC cells heal more slowly and based on transwell assays, show reduced cell migration, suggesting that clotrimazole inhibits the metastasis and invasion of HCC cells.
Invasion and metastasis, features that are characteristic malignant tumors, involve complex multi-step processes that are regulated by a variety of genes that are involved in tumor cell adhesion, stromal degradation, and tumor angiogenesis.23,24 Although numerous genes involved in tumor invasion and metastasis have been identified, the specific mechanisms underlying their function remain largely uncharacterized. Further, EMT is known to promote the invasion and metastasis of HCC cells,25 and E-cadherin, an important component of intercellular adhesion junctions, is an important marker of EMT.26 Our results suggested that clotrimazole inhibits the expression of EMT markers, N-cadherin and vimentin, while promoting that of E-cadherin, suggesting that clotrimazole may modulate HCC invasion and metastasis by inhibiting the EMT.
There is evidence suggesting that the ERK pathway is involved in EMT in HCC, and thus, also influence HCC invasion and metastasis.27,28 An active ERK pathway promotes aggressiveness in HCC; however, inhibiting the ERK pathway can suppress HCC aggressiveness.29,30 Our results indicated that clotrimazole inhibits the phosphorylation of ERK and p65. To further confirm that clotrimazole inhibits HCC metastasis by inhibiting ERK, the ERK activator honokiol was used to activate ERK in clotrimazole-treated HCC cells, in which ERK activity is repressed.31 Thus, we observed that honokiol reversed the inhibitory effect of clotrimazole on HCC cell invasion and migration, suggesting that clotrimazole acts on HCC via the ERK pathway.
Several factors may be involved in the response of HCC to clotrimazole treatment. Our results showed that the effects of clotrimazole on different HCC cell lines are somewhat different, suggesting that different HCC cell types respond differently to clotrimazole treatment. In terms of clinical application, although clotrimazole has been used clinically as an antifungal agent, further studies are still needed to validate the possibility of its clinical application in the treatment of HCC. Further, our findings initially confirmed the role of EMT and ERK in mediating the inhibition of HCC migration and invasion by clotrimazole. However, the mechanism of the direct action of clotrimazole still needs to be further explored. Furthermore, gene mutations and genetic variants play a very important role in the development of HCC. For example, the role of mutations in CTNNB1 and TP53 in HCC has been extensively studied.32–34 Whether these mutations play a role in the application of clotrimazole in the treatment of HCC requires further exploration.
Additionally, the experiments performed in this study were limited to cellular assays, implying that further studies are needed to determine whether clotrimazole has anti-HCC effects in humans. The more direct mechanism of action of clotrimazole in inhibiting HCC metastasis also needs to be supported by further studies.
Conclusion
In summary, the results of this study indicated that clotrimazole can inhibit the proliferation, migration, and invasion of HCC via the suppression of EMT by modulating the ERK signaling pathway. To the best of our knowledge, this is the first study in which the mechanisms underlying clotrimazole-mediated HCC inhibition are characterized with a focus on the role of ERK in EMT. Our study provides novel avenues for the development of HCC therapeutics.
Acknowledgments
This work was funded by Science and Technology Research Plan of Henan Provincial Health Commission (SBGJ201802) and Henan Province Medical Science and Technology Research Plan (LHGJ20190135).
Abbreviations
HCC, hepatocellular carcinoma; TACE, transcatheter arterial chemoembolization; EMT, epithelial mesenchymal transition; DMSO, dimethyl sulfoxide; CCK-8, cell counting kit-8; DMEM, Dulbecco’s Modified Eagle’s Medium; FBS, fetal bovine serum.
Author Contributions
All the authors made a significant contribution to the work here reported, in conception, study design, and execution, and in the acquisition, analysis, and interpretation of data. All the authors also participated in drafting, revising, or critically reviewing the manuscript; gave their final approval of the version to be published; agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declared no conflict of interest.
References
- 1.Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–249. doi: 10.3322/caac.21660 [DOI] [PubMed] [Google Scholar]
- 2.Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet. 2018;391(10127):1301–1314. doi: 10.1016/S0140-6736(18)30010-2 [DOI] [PubMed] [Google Scholar]
- 3.Yamamoto S, Kondo S. Oral chemotherapy for the treatment of hepatocellular carcinoma. Expert Opin Pharmacother. 2018;19(9):993–1001. doi: 10.1080/14656566.2018.1479398 [DOI] [PubMed] [Google Scholar]
- 4.Yamamoto T, Kubo S, Shuto T, et al. Surgical strategy for hepatocellular carcinoma originating in the caudate lobe. Surgery. 2004;135(6):595–603. doi: 10.1016/j.surg.2003.10.015 [DOI] [PubMed] [Google Scholar]
- 5.Sangiovanni A, Colombo M. Treatment of hepatocellular carcinoma: beyond international guidelines. Liver Int. 2016;36(Suppl 1):124–129. doi: 10.1111/liv.13028 [DOI] [PubMed] [Google Scholar]
- 6.Worns MA, Galle PR. Hepatocellular carcinoma in 2017: two large steps forward, one small step back. Nat Rev Gastroenterol Hepatol. 2018;15(2):74–76. doi: 10.1038/nrgastro.2017.174 [DOI] [PubMed] [Google Scholar]
- 7.European Association for the Study of the Liver. EASL clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol. 2018;69(1):182–236. doi: 10.1016/j.jhep.2018.03.019 [DOI] [PubMed] [Google Scholar]
- 8.Bae SH, Park JH, Choi HG, et al. Imidazole antifungal drugs inhibit the cell proliferation and invasion of human breast cancer cells. Biomol Ther (Seoul). 2018;26(5):494–502. doi: 10.4062/biomolther.2018.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Moussa Z, El-Sharief M, Abbas SY. New imidazolidineiminothione derivatives: synthesis, spectral characterization and evaluation of antitumor, antiviral, antibacterial and antifungal activities. Eur J Med Chem. 2016;122:419–428. doi: 10.1016/j.ejmech.2016.06.051 [DOI] [PubMed] [Google Scholar]
- 10.Wei S, Li L, Shu Y, et al. Synthesis, antifungal and antitumor activity of two new types of imidazolin-2-ones. Bioorg Med Chem. 2017;25(24):6501–6510. doi: 10.1016/j.bmc.2017.10.033 [DOI] [PubMed] [Google Scholar]
- 11.Chen Y, Chen HN, Wang K, et al. Ketoconazole exacerbates mitophagy to induce apoptosis by downregulating cyclooxygenase-2 in hepatocellular carcinoma. J Hepatol. 2019;70(1):66–77. doi: 10.1016/j.jhep.2018.09.022 [DOI] [PubMed] [Google Scholar]
- 12.Herrera-Martinez AD, Feelders RA, de Herder WW, et al. Effects of ketoconazole on ACTH-producing and non-ACTH-producing neuroendocrine tumor cells. Horm Cancer. 2019;10(2–3):107–119. doi: 10.1007/s12672-019-00361-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lo EN, Beckett LA, Pan CX, et al. Prospective evaluation of low-dose ketoconazole plus hydrocortisone in docetaxel pre-treated castration-resistant prostate cancer patients. Prostate Cancer Prostatic Dis. 2015;18(2):144–148. doi: 10.1038/pcan.2015.2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Patel V, Liaw B, Oh W. The role of ketoconazole in current prostate cancer care. Nat Rev Urol. 2018;15(10):643–651. doi: 10.1038/s41585-018-0077-y [DOI] [PubMed] [Google Scholar]
- 15.Lim YW, Goh BC, Wang LZ, et al. Pharmacokinetics and pharmacodynamics of docetaxel with or without ketoconazole modulation in chemonaive breast cancer patients. Ann Oncol. 2010;21(11):2175–2182. doi: 10.1093/annonc/mdq230 [DOI] [PubMed] [Google Scholar]
- 16.Pais P, Costa C, Pires C, Shimizu K, Chibana H, Teixeira MC. Membrane proteome-wide response to the antifungal drug clotrimazole in Candida glabrata: role of the transcription factor CgPdr1 and the drug: h+Antiporters CgTpo1_1 and CgTpo1_2. Mol Cell Proteomics. 2016;15(1):57–72. doi: 10.1074/mcp.M114.045344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kadavakollu S, Stailey C, Kunapareddy CS, White S. Clotrimazole as a cancer drug: a short review. Med Chem (Los Angeles). 2014;4(11):722–724. doi: 10.4172/2161-0444.1000219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Akinyemiju T, Abera S, Ahmed M, et al. The burden of primary liver cancer and underlying etiologies from 1990 to 2015 at the global, regional, and national level: results from the global burden of disease study 2015. JAMA Oncol. 2017;3(12):1683–1691. doi: 10.1001/jamaoncol.2017.3055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Furtado CM, Marcondes MC, Sola-Penna M, et al. Clotrimazole preferentially inhibits human breast cancer cell proliferation, viability and glycolysis. PLoS One. 2012;7(2):e30462. doi: 10.1371/journal.pone.0030462 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Meira DD, Marinho-Carvalho MM, Teixeira CA, et al. Clotrimazole decreases human breast cancer cells viability through alterations in cytoskeleton-associated glycolytic enzymes. Mol Genet Metab. 2005;84(4):354–362. doi: 10.1016/j.ymgme.2004.11.012 [DOI] [PubMed] [Google Scholar]
- 21.Coelho RG, Calaca Ide C, Celestrini Dde M, et al. Clotrimazole disrupts glycolysis in human breast cancer without affecting non-tumoral tissues. Mol Genet Metab. 2011;103(4):394–398. doi: 10.1016/j.ymgme.2011.04.003 [DOI] [PubMed] [Google Scholar]
- 22.Furtado CM, Marcondes MC, Carvalho RS, et al. Phosphatidylinositol-3-kinase as a putative target for anticancer action of clotrimazole. Int J Biochem Cell Biol. 2015;62:132–141. doi: 10.1016/j.biocel.2015.03.004 [DOI] [PubMed] [Google Scholar]
- 23.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–674. doi: 10.1016/j.cell.2011.02.013 [DOI] [PubMed] [Google Scholar]
- 24.Valastyan S, Weinberg RA. Tumor metastasis: molecular insights and evolving paradigms. Cell. 2011;147(2):275–292. doi: 10.1016/j.cell.2011.09.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jayachandran A, Dhungel B, Steel JC. Epithelial-to-mesenchymal plasticity of cancer stem cells: therapeutic targets in hepatocellular carcinoma. J Hematol Oncol. 2016;9(1):74. doi: 10.1186/s13045-016-0307-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Venhuizen JH, Jacobs FJC, Span PN, Zegers MM. P120 and E-cadherin: double-edged swords in tumor metastasis. Semin Cancer Biol. 2020;60:107–120. doi: 10.1016/j.semcancer.2019.07.020 [DOI] [PubMed] [Google Scholar]
- 27.Chen L, Guo P, He Y, et al. HCC-derived exosomes elicit HCC progression and recurrence by epithelial-mesenchymal transition through MAPK/ERK signalling pathway. Cell Death Dis. 2018;9(5):513. doi: 10.1038/s41419-018-0534-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ying HY, Gong CJ, Feng Y, Jing DD, Lu LG. Serine protease inhibitor Kazal type 1 (SPINK1) downregulates E-cadherin and induces EMT of hepatoma cells to promote hepatocellular carcinoma metastasis via the MEK/ERK signaling pathway. J Dig Dis. 2017;18(6):349–358. doi: 10.1111/1751-2980.12486 [DOI] [PubMed] [Google Scholar]
- 29.Ding Q, Jiang C, Zhou Y, et al. Kinesin family member 2C promotes hepatocellular carcinoma growth and metastasis via activating MEK/ERK pathway. Biosci Biotechnol Biochem. 2021;85(11):2241–2249. doi: 10.1093/bbb/zbab154 [DOI] [PubMed] [Google Scholar]
- 30.Jia XW, Li ZW, Dong LY, et al. Lack of hepatic stimulator substance expression promotes hepatocellular carcinoma metastasis partly through ERK-activated epithelial-mesenchymal transition. Lab Invest. 2018;98(7):871–882. doi: 10.1038/s41374-018-0039-2 [DOI] [PubMed] [Google Scholar]
- 31.Bie N, Han L, Wang Y, Wang X, Wang C. A polysaccharide from Grifola frondosa fruit body induces HT-29 cells apoptosis by PI3K/AKT-MAPKs and NF-kappaB-pathway. Int J Biol Macromol. 2020;147:79–88. doi: 10.1016/j.ijbiomac.2020.01.062 [DOI] [PubMed] [Google Scholar]
- 32.Hagiwara S, Nishida N, Ueshima K, et al. Accumulation of genetic and epigenetic alterations in the background liver and emergence of hepatocellular carcinoma in patients with non-alcoholic. Fatty Liver Dis Cells. 2021;10. doi: 10.3390/cells10113257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Charawi S, Just PA, Savall M, et al. LKB1 signaling is activated in CTNNB1-mutated HCC and positively regulates β-catenin-dependent CTNNB1-mutated HCC. J Pathol. 2019;247:435–443. doi: 10.1002/path.5202 [DOI] [PubMed] [Google Scholar]
- 34.Wang H, Chen L, Zhou T, et al. p53 mutation at serine 249 and its gain of function are highly related to hepatocellular carcinoma after smoking exposure. Public Health Genom. 2021;24:171–181. doi: 10.1159/000516598 [DOI] [PubMed] [Google Scholar]