Abstract
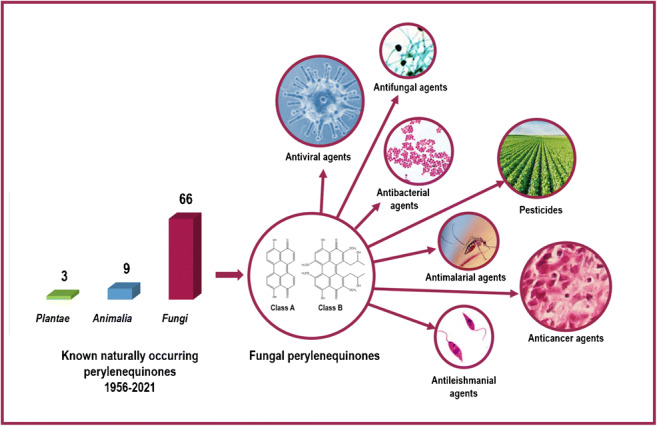
Perylenequinones (PQs) are aromatic polyketides with an oxidized pentacyclic core that make up a family of natural compounds. Naturally occurring PQs mostly are produced by phytopathogenic fungi, with few aphides, crinoids, and plants. PQs, also known as photosensitizers, absorb light energy which empowers them to produce reactive oxygen species that damage host cells. Therefore, PQs gained a considerable interest in pharmaceutical application notably in photodynamic therapy. This review presents a comprehensive overview of fungal PQs. Their occurrence, categorization, biosynthesis, structures, and bioactivities are all discussed in detail. After that, an analysis outlines their distribution across the kingdom of fungi. A total of 66 fungal PQs have been described from 22 ascomycete genera (Alternaria, Aspergillus, Bulgaria, Cenococcum, Cercospora, Cladosporium, Curvularia, Daldinia, Elsinoë, Hypocrella, Hypomyces, Parastagonospora, Phaeosphaeria, Phylacia, Pyrenochaeta, Rhopalostroma, Rubroshiraia, Setophoma, Shiraia, Stemphylium, Stagonospora, and Thamnomyces). Dothideomycetes account for the majority of documented fungal PQs (82%), followed by Sordariomycetes (14%), Leotiomycetes (3%), and Eurotiomycetes (1%). Herein, five families Pleosporaceae, Phaeosphaeriaceae, Cladosporiaceae, Shiraiaceae, and Hypoxylaceae are highlighted as potential sources of novel PQs due to their diversity. The review intends to pique bioprospectors’ interest in fungal PQs. Indeed, the pharmaceutical and agrochemical industries might gain greatly by exploiting fungal perylenequinones.
Graphical abstract
Keywords: Fungal natural products, Perylenequinones, Photodynamic therapy, Dothideomycetes, Sordariomycetes
Introduction
Fungal natural products have received a considerable attention. Several studies unveiled their amazing assortment. Perylenequinones (PQs) are members of natural products’ family characterized by an oxidized pentacyclic core represented by the parent perylenequinone 4,9-dihydroxyperylene-3,l0-quinone (25). Fungi are the best source for PQs as 85% of all known PQs are reported from fungi, compared to 12% for Animalia, and only 3% for Plantae. PQ pigments are produced by a wide variety of molds, the most of which are phytopathogens, with few marine sponge-derived, endolichenic, and endophytic fungi (Weiss et al. 1987; Pang et al. 2018; Tantry et al. 2018). Insects are an alternative source of PQs, such as rodoaphin, which is synthetized by aphids. In addition, the stalked crinoid Gymnocrinus richeri produced gymnochromes. PQs are also found in few plant species, such as scutiaquinones A and B were discovered in the root of Scutia myrtina. Besides, hypericin is one of Hypericum’s principal active constituents (Weiss and Altland 1965; Miskovsky 2002; Ayers et al. 2007). Moreover, PQs have been extracted from aquatic sediments and soil matter. 4,9-dihydroxyperylene-3,l0-quinone (25) and its derivatives were recovered from Japanese Andosols and Cambisol (Hanke et al. 2019; Kobayashi et al. 2019).
Perylenequinones are anticipated to be some of the most promising photodynamic therapy (PDT) agents (Olivo and Ali-Seyed 2007). PDT is a cancer and non-malignant condition treatment that comprises the delivery of a photosensitizing chemical such as PQs, followed by exposure of the tissue to non-thermal visible light (400–760 nm). When the molecule is stimulated by light of the appropriate wavelength, the photosensitizer is activated. This produces a series of molecular energy transfers leading to the release of singlet oxygen, a highly reactive and cytotoxic species, resulting in apoptosis. Drug uptake in malignant tissues combined with selective light treatment has the potential to give an effective cancer therapy with efficient cytotoxicity and minimal impairment to surrounding normal tissue (Ackroyd et al. 2001). PDT also provides several advantages over other standard clinical therapies, including more efficiency, greater safety, and decreased toxicity for surrounding normal tissues due to increasing accumulation of photosensitizers and irradiation on diseased targets (Yano et al. 2011). As compared to other photosensitizers investigated, such as porphyrin and phthalocyanine-like photosensitizers, PQs virtually exhibit all of the characteristics of ideal photosensitizers (Li et al. 2015). Hypocrellins A–B (50–51) have many advantages that might make them fascinating sensitizers for PDT. They are readily obtained, simply purified, very stable, low in toxicity, and do not form aggregates which would decrease their photodynamic activity. Although hypocrellin A (50) is phototoxic, it is rapidly removed from live organisms, usually within 24 h. Its photosensitizing side effects in PDT are considerably reduced as a result of this. In addition, hypocrellin A (50) is non-toxic in the absence of light (Diwu and William 1990). Several investigations have been conducted on the apoptotic and cytotoxic effects of some fungal PQs, including calphostin C (42), elsinochromes A (45), C (47), hypocrellin (49), and hypocrellin A (50). These studies highlighted their potential as promising anticancer medications and explored their application in cancer therapy (Dubauskas et al. 1998; Fang et al. 2006; Olivo and Ali-Seyed 2007; So et al. 2018; Tantry et al. 2018; Mastrangelopoulou et al. 2019). Furthermore, Sharma et al. (2013) have invented a patent (US 8,506,931 B2), that contains hypocrellin derivatives. These compounds can be particularly useful as photosensitizers or sononsensitizers in photodynamic or sonodynamic therapy. Also useful as therapeutic agents for treating various hyperproliferative disorders. Since two decades, the antiviral activity of various PQs has been studied (Hudson et al. 1997; Krishnamoorthy et al. 2005; Wiehe et al. 2019). Hudson and associates discovered a correlation between singlet oxygen (1O2) quantum yield and antiviral activity (Hudson et al. 1997). Other remarkable biological properties of PQs have been noted, such as the prevention of skin diseases caused by fungal infections, antileishmanial, antimalarial, antimicrobial properties, and mutagenicity (Wang and Bao 1985; Stack and Prival 1986; Diwu 1995; Ma et al. 2004; Fang et al. 2006; Tantry et al. 2018).
PQs were exploited in agriculture field as fungicides. In 2005, a patent (US 6,936,571 B2) was invented by Zhnag and Liu. The active constituents of the fungicide are selected perylenequinone derivatives including cercosporin (30), phleichrome (31), cladochromes A–D (33-36), elsinochromes A–C (45–47), hypocrellins A–B (50–51), and hypomycins A–B (53–54) (Zhnag and Liu 2005). PQs, on the other hand, have a promising environmental role as pesticides, due to their rapid degradation. Ahonsi et al. (2005) stated that, elsinochrome A (45) degrades rapidly in such conditions. They advocated for the release of elsinochrome A (45) into the environment, particularly in exposed niches such as spraying biocontrol product on plant surfaces, or release from shed diseased leave. This review gathers a comprehensive compilation of published findings on fungal PQs. The taxonomy of PQs and their biosynthesis come first in the introduction. Then, the review is divided into two sections, the first of which highlights a bibliography of fungal PQs and illustrates their structures. The distribution of PQ-producing fungi within the kingdom of fungi is the subject of section two, and the findings are summarized in a brief conclusion.
Classification of perylenequinones
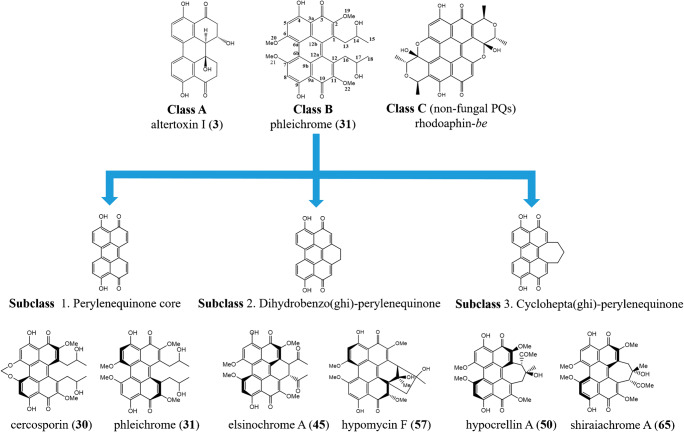
Natural PQs have been categorized into three broad classes (Fig. 1), according to Weiss et al. (1987).
-
A.
Class A: C20 compounds without carbon substituents
Fig. 1.
Classification of perylenequinones
Class A consists of the simple PQs including the parent perylenequinone 4,9-dihydroxyperylene-3,l0-quinone (25), which is found in Daldinia and other fungal taxa. Eleven genera (Table 1) produced partially reduced perylenequinones of class A.
-
B.
Class B: PQs carrying carbon substituents
Table 1.
List of PQ-producing genera
| Perylenequinone classes | Genera | References |
|---|---|---|
| Class A | Alternaria | (Idris et al. 2015) |
| Aspergillus | (Zhang et al. 2017) | |
| Bulgaria | (Xian et al. 2006) | |
| Cenococcum | (Peter et al. 2016) | |
| Curvularia | (Cruz et al. 2020) | |
| Daldinia | (Anderson and Murray 1956) | |
|
Phylacia Rhopalostroma |
(Wendt et al. 2018) (Stadler et al. 2010) |
|
| Setophoma | (Bazioli et al. 2020) | |
| Stemphylium | (Podlech et al. 2014) | |
| Thamnomyces | (Wendt et al. 2018) | |
| Class B | Cercospora | (Mastrangelopoulou et al. 2019) |
| Cladosporium | (Pettit 2011) | |
| Elsinoë | (Jiao et al. 2019) | |
| Hypocrella | (Li et al. 2021) | |
| Hypomyces | (Liu et al. 2001b, a) | |
| Parastagonospora | (Chooi et al. 2017) | |
| Phaeosphaeria | (Li et al. 2012) | |
| Pyrenochaeta | (Kurobane et al. 2006) | |
| Rubroshiraia | (Dai et al. 2019) | |
| Shiraia | (Fang et al. 2006) | |
| Stagonospora | (Ahonsi et al. 2005) |
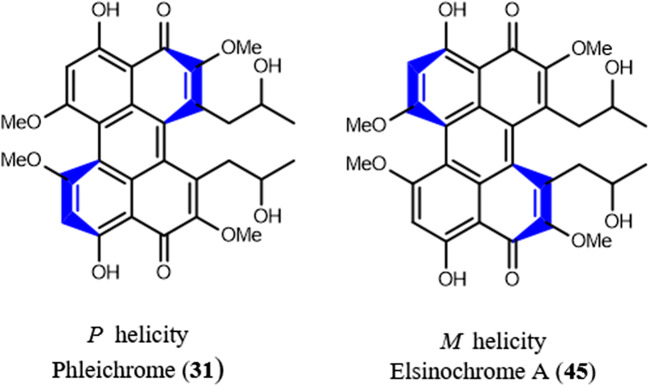
Class B is derived from the unit of cercosporin (30) and phleichrome (31), with or without additional alicyclic rings. It appears that PQs of class B are enriched in many phytopathogenic strains and have a role in plant pathogenicity. PQs of class B are produced via 11 genera (Table 1). 4,9-Dihydroxyperylene-3, l0-quinone (25) is an achiral planar substance. In most fungal perylenequinones, structural elements such as the two bulky methoxy groups or a strained seven-membered ring in positions 6 and 7, together with two C3 side chains in positions 1 and 12, are the source of enough steric interruption to force the pentacyclic perylenequinone system into a nonplanar helical shape (Weiss et al. 1987; Mulrooey et al. 2012; Hu et al. 2019). This helicity generates an element of asymmetry, the axial chirality M or P of the perylenequinone molecule (Fig. 2). More information concerning stereochemistry of PQs are available (Weiss et al. 1987). Class B was divided into three subclasses (Fig. 1) based on the core structures: (1) Perylenequinone core includes cercosporin, phleichrome, cladochromes, and calphostins (2) Dihydrobenzo (ghi)perylenequinone contains: elsinochromes, hypomycins, and phaeosphaerins (3) Cyclohepta(ghi)perylenequinone involves hypocrellins and shiraiachromes (Hu et al. 2019).
Fig. 2.
Axial chirality P and M of perylenequinones
-
C.
Class C
The parent perylenequinone 4,9-dihydroxyperylene-3, l0-quinone (25) with extra alicyclic rings is classified as class C. Other non-fungal species, such as aphids, crinoids, and plants formed PQs of class C, e.g., rhodoaphin-be, erythrouphins-fb and –sl, gymnochromes, scutiaquinones A and B (Weiss and Altland 1965; Miskovsky 2002; Ayers et al. 2007). The classification of fungal perylenequinones is shown in Fig. 1.
Biosynthesis of fungal perylenequinones
PQs captivated biologists with their role in fungal virulence, potent biological activities, as well as chemists due to their interesting axial chirality and photochemical characteristics (Diwu and Lown 1993; Mulrooey et al. 2010; Daub et al. 2013; So et al. 2018). Therefore, the biosynthetic gene clusters (BGCs) of some fungal PQs like cercosporin (30), elsinochrome C (47), and hypocrellin A (50) have been identified.
Cercosporin (30) has a typical pentacyclic perylenequinone core; however, it is distinguished by the presence of an asymmetric 1,3-dioxepine moiety. Cercosporin (30) was discovered in Cercospora kikuchii, the pathogenic fungus that causes soy bean purple speck disease (Kuyama and Tamura 1957). CTB cluster for biosynthesis of cercosporin (30) from Cercospora nicotianae was the first BGC to be found (Chen et al. 2007; Newman and Townsend 2016; Hu et al. 2019). Elsinochromes (45–48) are characterized by the hexacyclic dihydrobenzo(ghi)perylenequinone core. 1n 2017, Chooi and coworkers discovered the elc cluster for biosynthesis of elsinochrome C (47) in Parastagonospora nodorum, that causes septoria nodorum blotch in wheat, which has been proven to be important for its pathogenicity (Chooi et al. 2017). Three bamboo parasitic fungi produced hypocrellins, Hypocrella bambusae, Shiraia bambusicola, and Rubroshiraia bambusae. Hypocrellins (49–52) are considered by the hexacyclic cyclohepta(ghi)perylenequinone core, they are notorious to be present in traditional Chinese medicine (Yu et al. 1993). Based on gene expression correlation, the HYP cluster for hypocrellin A (50) production was discovered in Shiraia sp. Slf14 (Yang et al. 2014), and subsequently verified by targeted gene deletion, the monooxygenase-encoding gene (Mono) which is located in the hypocrellin gene cluster of Shiraia sp. (Deng et al. 2018).
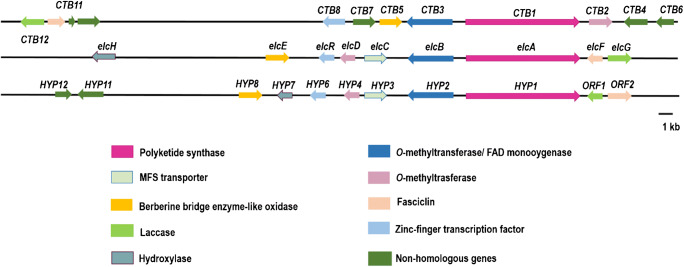
Biosynthesis of fungal PQs has been extensively investigated and revised several times to disclose the ambiguity. The CTB, elc, and HYP gene clusters contain multiple shared homologs (Fig. 3) (Chen et al. 2007; Yang et al. 2014; Newman and Townsend 2016; Chooi et al. 2017; Deng et al. 2018; Hu et al. 2019). The phylogenetic analysis of elcA and allied fungal non-reducing PKSs revealed that elcA is more closely related to Shiraia sp. HYP1 than to C. nicotianae CTB1. The elc gene cluster shares more homologues with HYP gene cluster compared with CTB cluster as well. Due to their structures, both hypocrellin A (50) and elsinochrome A (45) have a hexacyclic system while cercosporin (30) is pentacyclic, the additional homologous genes shared between elc and HYP gene clusters but not CTB could be responsible for the formation of the additional ring (Chooi et al. 2017).
Fig. 3.
The biosynthetic gene clusters of cercosporin CTB, elsinochrome A (45) elc, and hypocrellin A (50) HYP. Modified from Chooi et al. (2017) and Hu et al. (2019)
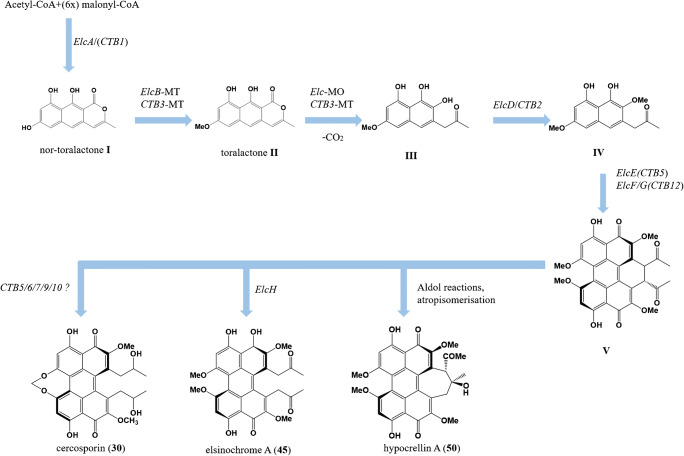
The most recent biosynthesis of perylenequinones was proposed by Hu et al. (2019) (Fig. 4). A common aromatic polyketide precursor serves as the starting point for biosynthesis, nor-toralactone I, which is synthesized by the non-reducing polyketide synthase (PKS) CTB1/elcA/HYP1. The subsequent step was achieved by bifunctional enzyme CTB3/elcB/HYP2 homolog, which possesses fused O-methyltransferase and Favin-dependent monooxygenase domains. This enzyme is responsible for methylation, hydroxylation, ring opening, decarboxylation of nor-toralactone I to afford toralactone II, then naphthol intermediate III. The O-methyltransferase CTB2/elcD/HYP4 is proposed to methylate the nascent OH-6 of intermediate III, blocking further oxidation at this site and yielding compound IV. The oxidative coupling steps to generate the pentacyclic core was elusive. Hu and collaborators have revealed the vagueness by demonstrating a heterologous biosynthesis, that cognate pairing of both Berberine Bridge Enzyme-like Oxidase (BBEO) elcE/CTB5 and Laccase-like Multi Copper Oxidase (LMCO) elcG/CTB12 for the double coupling step of two naphthol intermediates to afford the perylenequinone core V. Elsinochrome A (45) is synthesized via a radical process triggered by a single electron transfer from an enolate at the side chain to the FAD in elcH via the putative perylenequinone intermediate V. In the absence of elcH, hypocrellins (49–50) are formed, suggesting that they are most likely derived from the putative intermediate V via a transannular aldol reaction (Newman and Townsend 2016; Deng et al. 2018; Hu et al. 2019). CTB5, 6, 7, 9, and 10 afford cercosporin (30) through different reactions including reduction and homodimerization of the intermediates. The Zinc finger transcription factor CTB8 co-regulates expression of the cluster, while the Major Facilitator Superfamily (MFS) transporter CTB4 exports the final metabolite (Chen et al. 2007; Choquer et al. 2007; Newman and Townsend 2016).
Fig. 4.
Proposed biosynthetic pathway for three perylenequinones of class B: cercosporin (30), elsinochrome A (45), and hypocrellin (50). Modified from Hu et al. (2019)
Stimulation of PQs’ production
Light is an essential factor that regulates several physiological activities of fungi such as growth, reproduction, and biosynthesis of metabolites (Corrochano 2007). Conversely to plants, fungi use light as a source of information rather than energy (Tisch and Schmoll 2010). There is a strong evidence that light influences the synthesis of certain PQs. A significant increase in hypocrellin A (50) yield was observed, when Shiraia spp. were cultured under light-dark shift and red light (Sun et al. 2018; Ma et al. 2019a). The role of light in formation of cercosporin and elsinochromes by Cercospora nicotianae, C. kikuchii, and Elsinoë fawcettii was emphasized (Ehrenshaft and Upchurch 1991; Choquer et al. 2007). Light, on the other hand, is not required for biosynthesis of all PQs. Light seemed not to induce the synthesis of elsinochrome C (47) by Parastagonospora nodorum (Liao and Chung 2008). Several other factors, such as media, nutrition, growth conditions, and environmental variables, have an impact on PQs production. The synthesis of hypocrellin (49) increased on rice medium compared to Cheerios or oatmeal medium in solid-fermentation cultures (Al Subeh et al. 2020). The use of Triton X-100 and low-intensity ultrasound irradiation resulted in a significant increase in hypocrellin A (50) production (Lei et al. 2017; Sun et al. 2017). The formation of different PQs was boosted by co-cultivation of a hypocrellin-producing fungus with bacteria (Ma et al. 2019b).
Section one: fungal perylenequinones
Fungal PQs fall into two classes (A and B) of perylenequinones. This section includes the bibliography of known PQs between 1956 and 2021, as well as their structures and biological activities. A total of 66 fungal PQs have been found in 22 fungal genera. Table 2 shows a list of fungal PQs as well as their producers.
Table 2.
List of fungal PQs that have been discovered and their producers
| PQs’ class | Perylenequinone | Producer | Source | Reference |
|---|---|---|---|---|
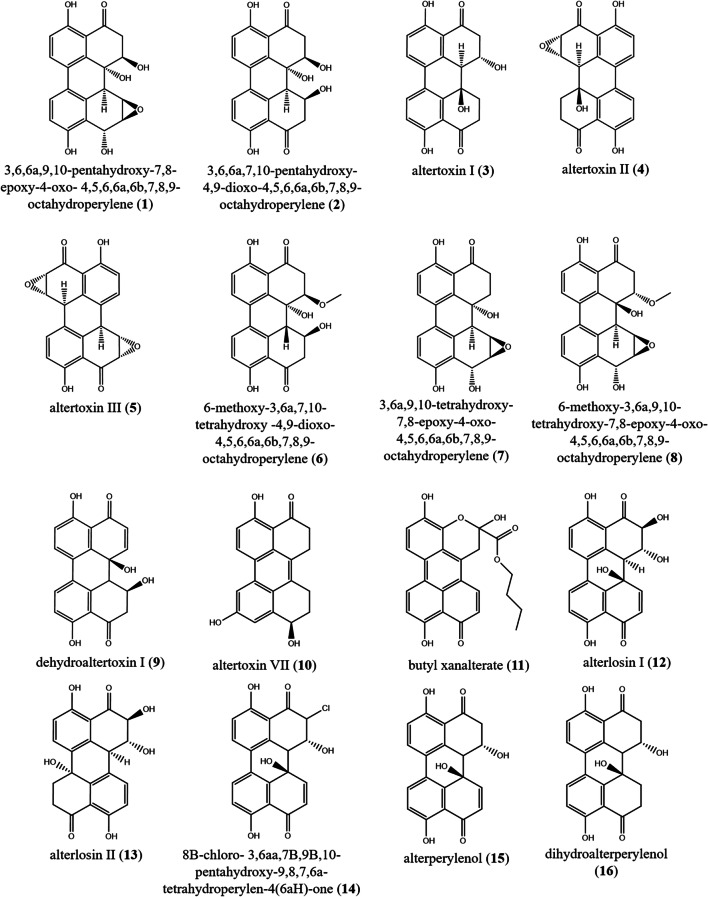
| Class A | 1) 3,6,6a,9,10-pentahydroxy-7,8-epoxy-4-oxo-4,5,6,6a,6b,7,8,9-octahydroperylene | Alternaria sp. |
Endophytic fungus from Pinus ponderosa |
(Tantry et al. 2018) |
| 2) 3,6,6a,7,10-pentahydroxy-4,9-dioxo-4,5,6,6a,6b,7,8,9-octahydroperylene | (Tantry et al. 2018) | |||
|
3) altertoxin I 4) altertoxin II 5) altertoxin III |
Alternaria alternata | – | (Stack and Prival 1986) | |
|
6) 6-methoxy-3,6a,7,10-tetrahydroxy-4,9-dioxo-4,5,6,6a,6b,7,8,9-octahydroperylene 7) 3,6a,9,10-tetrahydroxy-7,8-epoxy-4-oxo-4,5,6,6a,6b,7,8,9-octahydroperylene 8) 6-methoxy-3,6a,9,10-tetrahydroxy-7,8-epoxy-4-oxo-4,5,6,6a,6b,7,8,9-octahydroperylene 9) dehydroaltertoxin I altertoxin I (3) |
Alternaria sp. |
Endophytic fungus from Pinus ponderosa |
(Idris et al. 2015) | |
|
10) altertoxin VII 11) butyl xanalterate |
Alternaria sp. SCSIO41014 | Sponge-derived fungus | (Pang et al. 2018) | |
|
12) alterlosin I 13) alterlosin II |
Alternaria alternata | Pathogenic fungus from Centaurea maculosa, | (Stierle et al. 1989) | |
|
14) 8β-chloro-3,6aα,7β,9β,10-pentahydroxy-9,8,7,6 atetrahydroperylen-4(6aH)-one 15) alterperylenol 16) dihydroalterperylenol |
Alternaria sp. M6 Aspergillus fumigatus D |
Halotolerant fungus Endophytic of Edgeworthia chrysantha. |
(Zhang et al. 2012) (Zhang et al. 2017) |
|
|
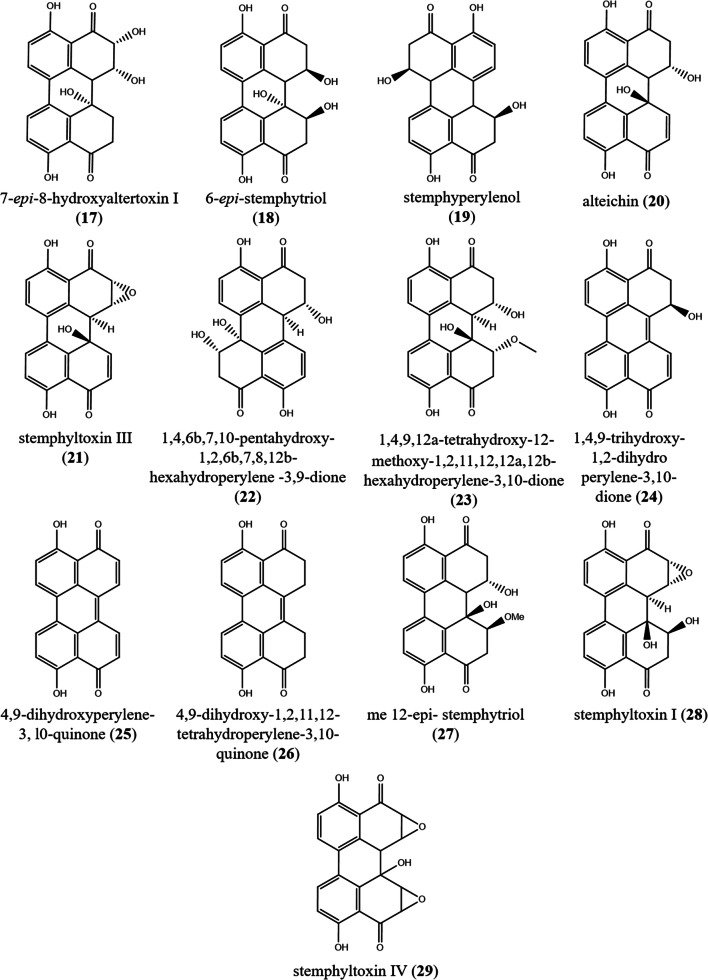
17) , 7-epi-8-hydroxyaltertoxin I 18) 6-epi- stemphytriol 19) stemphyperylenol altertoxin I (3) |
Alternaria alternata Setophoma sp. |
Marine endophytic fungus from algal species of the genus Laurencia. | (Gao et al. 2009; Bazioli et al. 2020) | |
| 20) alteichin | Alternaria eichhorniae | Phytopathogenic fungus | (Robeson et al. 1984) | |
| 21) stemphyltoxin III | Alternaria alternata | – | (Davis and Stack 1991) | |
|
22) 1,4,6b,7,10-pentahydroxy-1,2,6b,7,8,12b-hexahydroperylene-3,9-dione 23) 1,4,9,12a-tetrahydroxy-12-methoxy-1,2,11,12,12a,12b-hexahydroperylene-3,10-dione |
Alternaria tenuissima SS77 | Endophytic fungus | (Chagas et al. 2016) | |
| 24) 1,4,9-trihydroxy-1,2-dihydroperylene-3,10-dione | ||||
| 25) 4,9-dihydroxyperylene-3, l0-quinone |
Bulgaria inquinans Daldinia concentrica Thamnomyces sp. Phylacia sp. Cenococcum geophilum Rhopalostroma sp. |
– | (Anderson and Murray 1956; Weiss et al. 1987; Stadler et al. 2004; Itoh et al. 2012; Wendt et al. 2018) | |
| 26) 4,9-dihydroxy-1,2,11,12-tetrahydroperylene-3,10-quinone | Bulgaria inquinans | – | (Xian et al. 2006) | |
|
27) methylated 12-epi-stemphytriol alterperylenol (15) dihydroalterperylenol (16) |
Curvularia lunata LBQM-04 | – | (Cruz et al. 2020) | |
|
28) stemphyltoxin I 29) stemphyltoxin IV altertoxin II (4) stemphyltoxin III (21) |
Stemphylium botryosum | – | (Podlech et al. 2014) | |
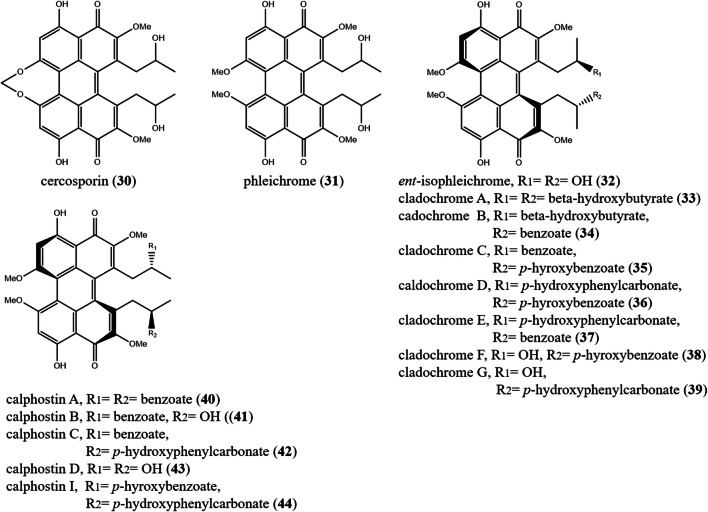
| Class B | 30) cercosporin |
Cercospora kikuchii C. hayii |
– | (Kuyama and Tamura 1957; Mumma et al. 1973; Mastrangelopoulou et al. 2019) |
| 31) phleichrome | Cladosporium phlei | Pathogenic fungus of Phleum pretense | (Seto et al. 2005) | |
|
32) ent-isopheichrome 33) cladochrome A 34) cladochrome B |
Cladosporium cucumerinum | Pathogen | (Arnone et al. 1988) | |
|
35) cladochrome C 36) cladochrome D 37) cladochrome E |
Cladosporium cladosporioides | – | (Arnone et al., 1990) | |
|
38) cladochrome F 39) cladochrome G |
Cladosporium spp. | – | (Williams et al. 2008; Pettit 2011) | |
|
40) calphostin A 41) calphostin B 42) calphostin C 43) calphostin D 44) calphostin I |
Cladosporium cladosporioides Phaeosphaeria sp. |
– |
(Iida et al. 2012) (Li et al. 2012) |
|
|
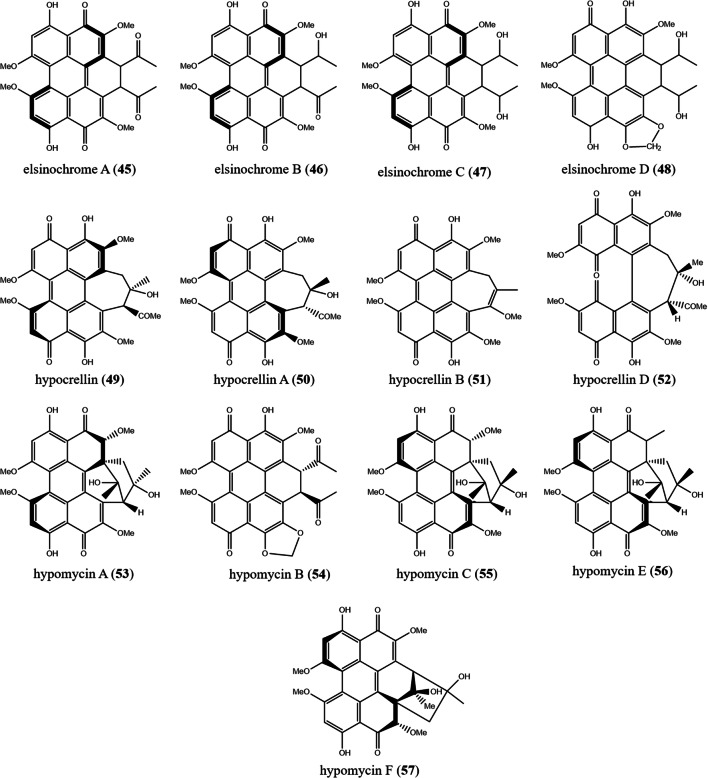
45) elsinochrome A 46) elsinochrome B 47) elsinochrome C 48) elsinochrome D |
Elsinoë annonae Stagonospora convolvuli Parastagonospora nodorum Pyrenochaeta terrestris Shiraia sp. Phaeosphaeria sp. |
Pathogen | (Weiss et al. 1987; Meille et al. 1989; Kurobane et al. 2006; Li et al. 2012; Chooi et al. 2017; Al Subeh et al. 2020) | |
|
49) hypocrellin 50) hypocrellin A 51) hypocrellin B 52) hypocrellin D |
Hypocrella bambusae Rubroshiraia bambusae Shiraia sp. |
Pathogen | (Wu et al. 1989; Ma et al. 2004; Dai et al. 2019; Al Subeh et al. 2020; Li et al. 2021) | |
|
53) hypomycin A 54) hypomycin B 55) hypomycin C 56) hypomycin E 57) hypomycin F 58) hypomycin D |
Hypomyces sp. Shiraia sp. |
– | (Liu et al. 2001b, a; Shen et al. 2003; Al Subeh et al. 2020) | |
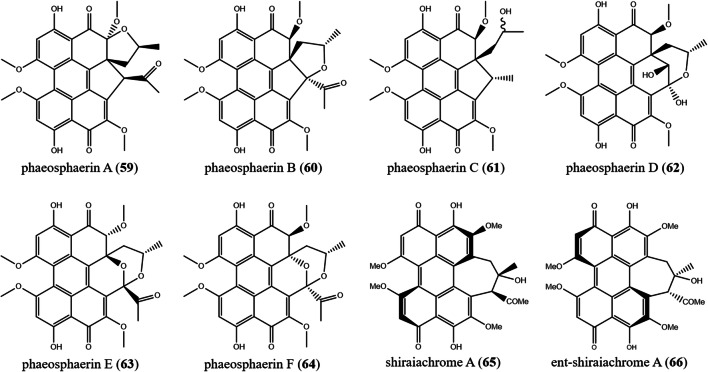
|
59) phaeosphaerin A 60) phaeosphaerin B 61) phaeosphaerin C 62) phaeosphaerin D 63) phaeosphaerin E 64) phaeosphaerin F |
Phaeosphaeria sp. | Endolichenic fungus from Heterodermia obscurata | (Li et al. 2012) | |
|
65) shiraiachrome A 66) ent-shiraiachrome A |
Shiraia bambusicola | Pathogenic fungus of bamboos | (Mulrooey et al. 2012) |
Class A perylenequinones
Eleven ascomycete genera (Table 1) have been reported to produce 29 PQs of class A (Figs. 5 and 6). Most of them (24 compounds) are synthetized by Alternaria spp. Class A perylenequinones displayed diverse biological properties such as cytotoxicity, antiviral, antimicrobial, antileishmanial, and antimalarial activities.
Fig. 5.
Perylenequinones described from the genera Alternaria and Setophoma
Fig. 6.
Reported perylenequinones from the genera Alternaria, Aspergillus, Bulgaria, Cenococcum, Curvularia, Daldinia, Phylacia, Rhopalostroma, Setophoma, Stemphylium, and Thamnomyces
Alternaria Nees ex Fr
The genus Alternaria (Pleosporaceae, Pleosporales) contains 366 accepted and recognizable species. Some species cause pre-harvest and post-harvest damage to cereal grains, fruits, and vegetables (Patriarca and Pinto 2018; Wijayawardene et al. 2020). Alternaria spp. produced several toxins including PQs. A total of 24 perylenequinones were recovered from Alternaria species. Recently, two dark PQ pigments, were isolated from the endophyte Alternaria sp. associated with Pinus ponderosa, identified as 3,6,6a,9,10-pentahydroxy-7,8-epoxy-4-oxo-4,5,6,6a,6b,7,8,9-octahydroperylene (1), and 3,6,6a,7,10-pentahydroxy-4,9-dioxo-4,5,6,6a,6b,7,8,9-octahydroperylene (2). Compound (1) and (2) displayed antileishmanial activity against Leishmania donovani with IC50 values of 2.55 and 4.40 μg/mL, respectively. Also compound (2) revealed antimalarial activity against both chloroquine sensitive (D6) (IC50 4.24 μg/mL) and chloroquine resistant (W2) clones of Plasmodium falciparum (3.65 μg/mL), as well as cytotoxicity against mammalian kidney fibroblasts (VERO cells) with IC50 value of 3.59 μg/mL (Tantry et al. 2018). Altertoxins I–III (3–5) were described from Alternaria alternata. Mutagenicity of compounds (3–5) was investigated using the Ames test with Salmonella typhimurium; these compounds were mutagenic to Salmonella typhimurium TA98, TA100, and TA1537 with and without metabolic activation (Stack and Prival 1986). Five perylenequinones (3, 6–9) were recovered from Alternaria sp. (DC401) an endophyte associated with Pinus ponderosa, altertoxin I (3), 6-methoxy-3,6a,7,10-tetrahydroxy-4,9-dioxo-4,5,6,6a,6b,7,8,9-octahydroperylene (6), 3,6a,9,10-tetrahydroxy-7,8-epoxy-4-oxo-4,5,6,6a,6b,7,8,9-octahydroperylene (7), 6-methoxy-3,6a,9,10-tetrahydroxy-7,8-epoxy-4-oxo-4,5,6,6a,6b,7,8,9-octahydroperylene (8), and dehydroaltertoxin I (9) (Idris et al. 2015). In 2018, two dark red pigments (PQs’ derivatives) altertoxin VII (10) and butyl xanalterate (11) were purified from a culture of the sponge-derived fungus, Alternaria sp. SCSIO41014. Compound (10) exhibited potent cytotoxicity against human gastric carcinoma cells (SGC-7901) (IC50 8.75 ± 0.13 μg/mL), hepatocellular carcinoma cells (BEL-7402) (13.11 ± 0.95 μg/mL), and human erythroleukemia (K562) (26.58 ± 0.80 μg/mL) (Pang et al. 2018). Two toxins identified as PQs, alterlosins I–II (12–13) were recovered from Alternaria alternata, a pathogen of spotted knapweed Centaurea maculosa. Compound (13) showed phytotoxicity (Stierle et al. 1989).
A monochloridated perylenequinone named 8β-chloro-3,6aα,7β,9β,10-pentahydroxy-9,8,7,6 atetrahydroperylen-4(6aH)-one (14), besides, two further PQs named alterperylenol (15), and dihydroalterperylenol (16) were isolated from a halotolerant Alternaria sp. M6 obtained from the solar salt field at the beach of Bohai Bay in China (Zhang et al. 2012). Four PQs, altertoxin I (3), 7-epi-8-hydroxyaltertoxin I (17), 6-epi-stemphytriol (18), and stemphyperylenol (19) were isolated from Alternaria alternata, a marine endophytic fungus derived from an unidentified algal species of the genus Laurencia. The antimicrobial activity of compounds (17) and (19) against Staphylococcus aureus, Escherichia coli, and Aspergillus niger was evaluated, but neither showed obvious activity (Gao et al. 2009). Alteichin (20) was recovered from liquid cultures of Alternaria eichhorniae Nag Raj & Ponnappa, a phytopathogen that attacks water hyacinth (Robeson et al. 1984). Stemphyltoxin III (21) was reported from Alternaria alternata. Compound (21) was tested for mutagenicity in the Ames Salmonella typhimurium plate incorporation assay with and without Aroclor 1254-induced rat S-9 metabolic activation. A positive response was noted in S. typhimurium TA98 and TA1537, also marginal response in strain TA100 (Davis and Stack 1991). Three yellow and yellow brownish PQs, 1,4,6b,7,10-pentahydroxy-1,2,6b,7,8,12b-hexahydroperylene-3,9-dione (22), 1,4,9,12a-tetrahydroxy-12-methoxy-1,2,11,12,12a,12b-hexahydroperylene-3,10-dione (23), and 1,4,9-trihydroxy-1,2-dihydroperylene-3,10-dione (24) were obtained from cultures of the endophytic fungus Alternaria tenuissima SS77 (Chagas et al. 2016).
Aspergillus P. Micheli ex Haller
The genus Aspergillus (Aspergillaceae, Eurotiales) has a significant economic and social impact. It comprises 428 species. Aspergillus spp. occur worldwide in various habitats such as food producing mycotoxins and are frequently reported as human and animal pathogens (Samson et al. 2014; Wijayawardene et al. 2020). Only one PQ is reported from the genus Aspergillus. Bioassay-guided fractionation of the crude extract of the salty broth of Aspergillus fumigatus D, an endophyte associated with Edgeworthia chrysantha led to the isolation of a chlorated perylenequinone, 8-chloro-3, 6a,7,9,10-pentahydroxy-9,8,7,6a-tetrahydroperylen-4(6aH)-one (14). Compound (14) revealed strong antimicrobial effects against Escherichia coli (MIC 0.78 μM) and Candida albicans (1.56 μM). Also exhibited moderate anti-proliferative activity against human lung cancer cell line A549 (IC50 12.79 ± 0. 33 μM) (Zhang et al. 2017).
Bulgaria Fr.
The genus Bulgaria (Phacidiaceae, Phacidiales) consists of 12 species (Wijayawardene et al. 2020). Bulgaria inquinans is a saprophyte that grows on felled oak trunks and fallen branches, and less frequently on ash. 4,9-dihydroxyperylene-3,l0-quinone (25), and 4,9-dihydroxy-1,2,11,12-tetrahydroperylene-3,10-quinone (26), were described from the ethanolic extract of B. inquinans fruiting bodies (Weiss et al. 1987; Xian et al. 2006).
Cenococcum Moug. & Fr.
There are five species of the genus Cenococcum (Gloniaceae, Gloniales). The most common and globally abundant ectomycorrhizal fungus is Cenococcum geophilum. This fungus often dominates the root systems of trees in extreme environments (Peter et al. 2016; Wijayawardene et al. 2020). Sclerotia of Cenococcum geophilum were abundant in compound (25) which interpreted the existence of compound (25) in sediment samples from Lake Biwa (Itoh et al. 2012; Hanke et al. 2019).
Curvularia Boedijn
The genus Curvularia (Pleosporaceae, Pleosporales) is commonly found in plants and soil of tropical and subtropical regions. There are 119 recognized species of Curvularia (Khiralla et al. 2019; Wijayawardene et al. 2020). In 2020, three PQs, the methylated 12-epi-stemphytriol (27), (15), and (16) were reported for the first time from malt extract broth of C. lunata (LBQM-04) (Cruz et al. 2020).
Daldinia Ces. & De Not.
The genus Daldinia (Hypoxylaceae, Xylariales) comprises 67 species. Daldinia concentrica is an inedible wood-rotting fungus (Lee et al. 2006; Wijayawardene et al. 2020). 4,9-dihydroxyperylene-3,l0-quinone (25) was the first perylenequinone discovered by Anderson and Murray (1956). They isolated compound (25) from the large black fruiting bodies of D. concentrica.
Phylacia Lév.
The genus Phylacia (Hypoxylaceae, Xylariales) was collected from different tropical countries such as Colombia, Brazil, Mexico and French Guiana. Twelve species are documented of the genus Phylacia (Fournier and Lechat 2015; Wijayawardene et al. 2020). Several members of Hypoxylaceae including Phylacia spp. were examined. Compound (25) was frequently encountered in stromata of Phylacia spp. (Wendt et al. 2018).
Rhopalostroma D. Hawksw.
The genus Rhopalostroma (Hypoxylaceae, Xylariales) contains currently 11 species. Rhopalostroma spp. have been collected exclusively from Africa and Asia (Daranagama et al. 2015; Wijayawardene et al. 2020). Stromata extracts of several Rhopalostroma spp. were screened using high-performance liquid chromatography with diode array and mass spectrometric detection (HPLC-DAD-MS). The pigment, 4,9-dihydroxyperylene-3,l0-quinone (25) was detected (Stadler et al. 2010).
Setophoma Gruyter, Aveskamp & Verkley
Wijayawardene et al. (2020) accepted six species of the genus Setophoma (Phaeosphaeriaceae, Pleosporales). The production of some PQs by Setophoma was reported (Bazioli et al. 2020). Stemphyperylenol (19) and derivatives were recovered from Setophoma sp., compound (19) displayed antifungal effects against Aspergillus fumigatus, Pencillium brasilianum, and P. digitatum.
Stemphylium Wallr.
Some species of Stemphylium are pathogens of several important crops including onion, alfalfa, sugar beet, and asparagus. The genus Stemphylium (Pleosporaceae, Pleosporales) contains 96 species (Wijayawardene et al. 2020). Four perylenequinones, stemphyltoxins I, IV (28–29), besides two further (4), and (21) were recovered from Stemphylium botryosum (Podlech et al. 2014).
Thamnomyces Ehrenb.
Thamnomyces (Hypoxylaceae, Xylariales) is a tropical genus known exclusively from the Neotropics and Africa. Only 11 species are recorded of the genus Thamnomyces (Wijayawardene et al. 2020). Compound (25) was reported in stromata extracts of Thamnomyces spp. (Wendt et al. 2018).
Class B perylenequinones
Thirty-seven fungal PQs of class B have been described to date (Figs. 7, 8, and 9). They are produced by 11 ascomycete genera (Table 1). The majority of them are plant pathogenic strains. Fungal PQs of class B were also investigated for their biological properties, some of them revealed cytotoxicity, antiviral, antimicrobial and antileishmanial activities.
Fig. 7.
Reported perylenequinones from the genera Cercospora and Cladosporium
Fig. 8.
Produced perylenequinones by the genera Elsinoë, Hypocrella, Hypomyces, Parastagonospora, Pyrenochaeta, and Stagonospora
Fig. 9.
Described perylenequinones from the genera Phaeosphaeria and Shiraia
Cladosporium link
The genus Cladosporium (Cladosporiaceae, Capnodiales) is frequently isolated from soil and organic matter; however, some species are pathogens. The genus Cladosporium comprises 237 accepted species (Bensch et al. 2018; Wijayawardene et al. 2020). Cladosporium spp. are distinguished by producing diverse PQs including cladochromes and calphostins. Phleichrome (31) was reported from the pathogenic fungus Cladosporium phlei associated with the timothy plant (Phleum pratense L.). In the presence of light, compound (31) demonstrated antifungal action against Epichloe typhina (Seto et al. 2005). Ent-isopheichrome (32) beside cladochromes A–G (33–39) were described from different Cladosporium spp. (Arnone et al. 1988, 1990; Williams et al. 2008; Pettit 2011). Calphostins A–D, I (40–44) were purified from the fermented broth of Cladosporium cladosporioides. Calphostin C (42), revealed potent and specific inhibition of protein kinase C, because it was 1000 times more inhibitory to protein kinase C (IC50 0.05 μM) than other protein kinases such as cAMP-dependent protein kinase and tyrosine-specific protein kinase (IC50, >50 μM) (Kobayashi et al. 1989; Iida et al. 2012).
Cercospora Fresen.
The genus Cercospora (Mycosphaerellaceae, Capnodiales) contains 1125 species. Most Cercospora spp. are pathogens (Wijayawardene et al. 2020). In 1957, cercosporin (30) was isolated from mycelial cultures of Cercospora kikuchii, a pathogen that causes soybean purple speck disease (Kuyama and Tamura 1957). Compound (30) demonstrated photocytotoxicity against two glioblastoma multiforme (T98G, U87) and one breast adenocarcinoma (MCF7) human cell lines. However, in the dark compound (30) displayed a synergistic cytotoxicity with copper only in the most respiratory cell lines of MCF7 and T98G. Cercosporin (30) is a powerful photosensitizer, but with a short activation wave-length, mostly appropriate for superficial PDT treatments (Mastrangelopoulou et al. 2019).
Elsinoë Racib.
Species of Elsinoë are phytopathogens, causing scab and spot anthracnose on several economically important crops. The genus Elsinoë (Elsinoaceae, Myriangiales) contains 40 species (Fan et al. 2017; Wijayawardene et al. 2020). Four PQs were reported from Elsinoë. Three bright red pigments, elsinochromes A–C (45–47), and one orange named elsinochrome D (48) were produced in cultures by Elsinoë annonae and its anamorph Sphaceloma randii (Weiss et al. 1987; Meille et al. 1989). Elsinochrome phytotoxins are generated also by E. arachidis, the responsible fungus of peanut scab. Elsinochrome A (45) is distinguished by a superior singlet oxygen quantum yield compared to other kinds of photosensitizes. Elsinochrome A (45) could be easily synthesized at present, which makes it an alternative candidate for PDT (Jiao et al. 2019).
Hypocrella Sacc.
Hypocrella spp. (Clavicipitaceae, Hypocreales) are common in tropical regions, particularly in moist old-growth forests. There are more than 170 recognized species in the genus Hypocrella. Some species of Hypocrella are parasites of scale insects and white flies (Mains 1959; Chaverri et al. 2008; Wijayawardene et al. 2020). H. bambusae, a parasite of living inflorescence of bamboo, was studied (Dai et al. 2019). Investigations revealed that H. bambusae produced several types of PQs: hypocrellins A–B (49–51), hypomycins A (53), F (57), and shiraiachrome A (65) (Weiss et al. 1987; Diwu and William 1990; Ma et al. 2004; Cheng et al. 2004; Li et al. 2021). Hypocrellins and shiraiachromes have so close structures. Formerly, there has been much confusion in the naming of the same compounds extracted from different sources, hypocrellin A (50) was referred as shiraiachrome B, and hypocrellin B (51) was named as either shiraiachrome C or hypocrellin C (Wu et al. 1989; Kishi et al. 1991; Liu et al. 2001a; Ma et al. 2019b). Herein, we followed the structures and nomenclatures (Fig. 8) proposed by Al Subeh et al. (2020).
Hypomyces (Fr.) Tul. & C. Tul.
The genus Hypomyces (Hypocreaceae, Hypocreales) consists exclusively of fungicolous fungi, parasitizing the fruiting bodies of some members of Agaricales, Boletales, Helotiales, Pezizales, and Polyporales. Hypomyces comprises of 150 species widely distributed in Australia, Asia, and Europe (Wijayawardene et al. 2020; Yu et al. 2020). Four perylenequinones, hypomycins A–C, D1 (53–55, 58) were discovered in the mycelia of Hypomyces spp. (Liu et al. 2001b, a; Shen et al. 2003).
Parastagonospora Quaedvl., Verkley & Crous
Parastagonospora is an important genus in Phaeosphaeriaceae that includes pathogens causing leaf and glume blotch on cereal crops. Nineteen species are recognized of the genus Parastagonospora (Goonasekara et al. 2019; Wijayawardene et al. 2020). Parastagonospora nodorum is a significant pathogen of wheat. The production of elsinochrome C (47) by P. nodorum was reported by Chooi et al. (2017). They stated that elsinochrome C contributes to the virulence of P. nodorum against wheat.
Phaeosphaeria I. Miyake
Phaeosphaeriais a member of Phaeosphaeriaceae. There are around 96 species of the genus Phaeosphaeria. Most of the Phaeosphaeria species are parasites of Poaceae and grass-like monocot plants (Stchigel et al. 2004; Wijayawardene et al. 2020). Twelve different PQs are produced by Phaeosphaeria: phaeosphaerins A–F (59–64), five are yellow (phaeosphaerins A–C, E, and F), while one is orange (phaeosphaerin D), as well as calphostin D (43) elsinochromes A–C (45–47), and hypocrellins A–B (50–51). These PQs were purified from an endolichenic fungus Phaeosphaeria sp. found in the lichen Heterodermia obscurata. The unusual α,β-unsaturated ketone moieties present in phaeosphaerins A−F resulted in their yellow coloration. Phaeosphaerins’ cytotoxicity against human prostate cancers intensified in the presence of light (Li et al. 2012).
Pyrenochaeta De Not.
Pyrenochaeta spp. (Pleosporales) inhabit soil and plant debris and are well-known as pathogen of plants and occasionally humans. The genus Pyrenochaeta contains five accepted species (Levic et al. 2013; Yadav et al. 2015; Wijayawardene et al. 2020). Pyrenochaeta terrestris, a pathogen that causes onion pink root disease, produced both elsinochrome C (47) and D (48) (Kurobane et al. 2006).
Rubroshiraia D.Q. Dai & K.D. Hyde
Rubroshiraia (Shiraiaceae, Pleosporales) contains one species named Rubroshiraia bambusae D.Q. Dai & K.D. Hyde, a common pathogen of bamboos. R. bambusae is a well-known taxon used in Chinese traditional medicine which is called “Zhuhongjun” (Dai et al. 2019; Wijayawardene et al. 2020). The HPLC profiles of methanol stromata extracts of R. bambusae contained high quantities of hypocrellin A and B (50–51). Stromata extracts of R. bambusae contains almost double the content of hypocrellin A and B compared to Shiraia bambusicola (Dai et al. 2019). Hypocrellins have gained much attention due to their light-induced antitumor, antimicrobial, antileishmanial, and antiviral activities (Diwu and William 1990; Hudson et al. 1994; Ma et al. 2004). Some clinical trials showed that hypocrellins have promising treatment for various skin diseases, such as skin cancer and white lesions of the vulva (Wan and Chen 1981; Li et al. 2000).
Shiraia Henn.
The genus Shiraia (Shiraiaceae, Pleosporales) comprises one species. Shiraia bambusicola is a pathogen of several genera of bamboos. Large stromata of S. bambusicola have been used in folk Chinese medicine (Wu et al. 1989; Wijayawardene et al. 2020). Nine PQs were reported from S. bambusicola: elsinochromes A–C (45–47), hypocrellins A–B, D (50–52), hypomycin E (56), shiraiachrome A (65), and ent-shiraiachrome A (66) (Wu et al. 1989; Fang et al. 2006; Ma et al. 2019b; Al Subeh et al. 2020; Li et al. 2021). Endophytes, identified as Shiraia spp., were also shown to produce hypocrellins on media (Morakotkarn et al. 2008; Shen et al. 2014). Compound (50) demonstrated promising antifungal activity against Candida albicans (0.65 ± 0.14 μg/mL), and exhibited potent antileishmanial activity (IC50 of 0.27 ± 0.03 μg/mL), while compound (51) revealed moderate antileishmanial activity (IC50 12.7 ± 2.1 μg/mL). Interestingly, the antileishmanial activity of hypocrellin A was three- and six fold more potent than that of amphotericin B and pentamidine, respectively. Compound (52) reduced tumor cell proliferation in Bel-7721 (IC50 1.8 mg/mL), A-549 (8.8 mg/mL), and Anip-973 (38.4 mg/mL) (Fang et al. 2006). Compound (56) inhibited pseudotyped SARS-CoV-2 infection in 293T-ACE2 cells (IC50 0.17μM), as well as compound (66) (0.038 μM), and both even suppressed live SARS-CoV-2 infection (EC50 0.22 and 0.21μM, respectively) (Li et al. 2021).
Stagonospora (Sacc.) Sacc.
The genus Stagonospora (Massarinaceae, Pleosporales) comprises 220 species. Some species of Stagonospora are commonly known as aggressive pathogens of wheat. While other Stagonospora spp. have been implicated in bioremediation of aromatic compounds and lignin derivatives (Bergbauer et al. 1992; Zeiner et al. 2016; Wijayawardene et al. 2020). Stagonospora convolvuli LA39 is a pathogen of field and hedge bindweeds, and was reported to produce elsinochrome A (45). Hence, S. convolvuli has been studied as a bindweed biocontrol agent (Ahonsi et al. 2005; Boss et al. 2007).
Section two: distribution of PQ-producing fungi among the kingdom of fungi
This section covers the main information gleaned from the aforementioned fungal PQ reports. In addition, classification of PQ-producing fungi as well as a conclusion.
In this review, 66 fungal PQs were reported from 22 fungal genera between 1956 and 2021. The review revealed that, despite appearances, the synthesis of fungal PQs is not exclusive to phytopathogens. PQ-producing fungi include endophytic, endolichenic, soil, and marine strains. Interestingly, color diversity of fungal PQ pigments was observed to be quite considerable, various colors including red, orange, yellow, and even dark blue are available (Weiss et al. 1987; Meille et al. 1989; Mulrooey et al. 2012; Li et al. 2012; Al Subeh et al. 2020).
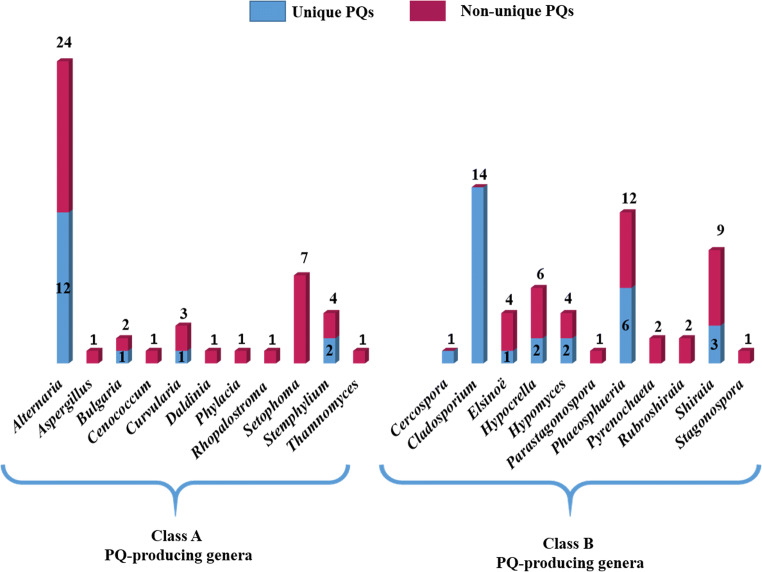
Obviously, 29 compounds belonging to class A have been reported from 11 ascomycete genera (Fig. 10). Twenty-four PQs were isolated from Alternaria (12 unique), seven from Setophoma, four from Stemphylium (two unique), three from Curvularia (one unique), two from Bulgaria (one unique) and one each from Aspergillus, Cenococcum, Daldinia, Rhopalostroma, Thamnomyces, and Phylacia, whereas 37 of the described fungal PQs belong to class B and were recovered from 11 ascomycete genera (Fig. 10). Cladosporium has 14 unique PQs, Phaeosphaeria has 12 (six unique), Shiraia has nine (three unique), Hypocrella has six (two unique), Elsinoë has four (one unique), Hypomyces has four (two unique), Pyrenochaeta and Rubroshiraia each has two, Cercospora has one unique, Stagonospora and Parastagonospora each has one PQ.
Fig. 10.
Number of fungal PQs per genus
The genera Alternaria, Cladosporium, Phaeosphaeria, and Shiraia have the most distinct PQs (Fig. 10). In comparison to class A, the uniqueness of PQs per genus was higher in class B. Remarkably, all PQ-producing fungi described in this review affiliate with one phylum Ascomycota, and only four classes. Dothideomycetes (14 genera) accounts for the bulk of the documented PQs 82%, followed by Sordariomycetes (six genera) 14%, Leotiomycetes (one genus) 3%, and Eurotiomycetes (one genus) 1%. The distribution of PQs among fungal genera is summarized in Fig. 11.
Fig. 11.
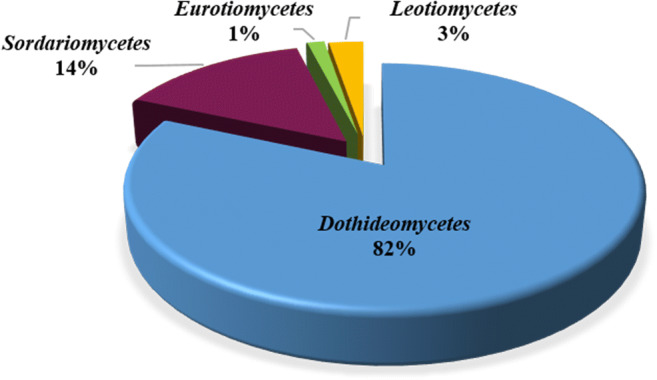
Distribution of PQs among fungal classes
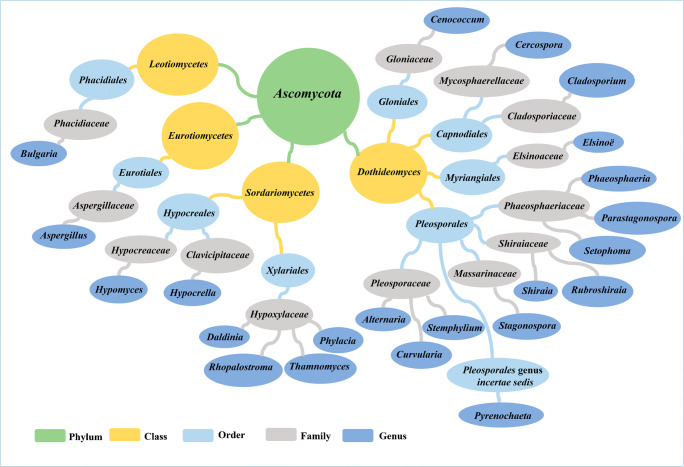
Fungal PQs were extracted from cultures, mycelia, stromata, and fruiting bodies, using various solvents including acetone, ethyl acetate, ethanol, and methanol. The tendency of fungal PQs appears to be extracted from cultures over stromata/fruiting bodies. Only 4,9-dihydroxyperylene-3,l0-quinone (25) was isolated from stromata/fruiting bodies of Bulgaria and Daldinia (Weiss et al. 1987; Anderson and Murray, 1956). There is a strong inconsistency in the chemotaxonomy of the PQ-producing fungi. They are described from a total of 13 separate families (Table 3). Fig. 12 shows the classification of PQ-producing fungi based on the recent outline of fungi (Wijayawardene et al. 2020). A chemotaxonomic approach is required to substantiate the hypothesis that members of a specific family could produce PQs. A number of fascinating Hypoxylaceae chemotaxonomic studies have been achieved (Helaly et al. 2018; Becker and Stadler 2021; Kuhnert et al. 2021). They documented the occurrence of several secondary metabolites in Daldinia, Phylacia, Rhopalostroma, and Thamnomyces stromata, including (25). In this review, five families, Pleosporaceae, Phaeosphaeriaceae, Cladosporiaceae, Shiraiaceae, and Hypoxylaceae are distinguished by their considerable number of PQs and genera. As a consequence, members of these families should be taken into account as potential sources of novel PQs.
Table 3.
List of families and number of PQ-producing genera
| Family | Number of genera* | Reference |
|---|---|---|
| Aspergillaceae | 1 | (Zhang et al. 2017) |
| Cladosporiaceae | 1 | (Arnone et al., 1990; Williams et al. 2008; Iida et al. 2012) |
| Clavicipitaceae | 1 | (Wu et al. 1989; Li et al. 2021) |
| Elsinoaceae | 1 | (Jiao et al. 2019) |
| Gloniaceae | 1 | (Itoh et al. 2012) |
| Hypocreaceae | 1 | (Liu et al. 2001b, a; Shen et al. 2003) |
| Hypoxylaceae | 4 | (Anderson and Murray 1956; Stadler et al. 2010) |
| Massarinaceae | 1 | (Boss et al. 2007) |
| Mycosphaerellaceae | 1 | (Mastrangelopoulou et al. 2019) |
| Phacidiaceae | 1 | (Weiss et al. 1987; Xian et al. 2006) |
| Phaeosphaeriaceae | 3 | (Li et al. 2012; Chooi et al. 2017; Bazioli et al. 2020) |
| Pleosporaceae | 3 | (Kurobane et al. 2006; Tantry et al. 2018; Cruz et al. 2020) |
| Shiraiaceae | 2 | (Wu et al. 1989; Al Subeh et al. 2020) |
*Pyrenochaeta (Pleosporales genus incertae sedis) is excluded from the table
Fig. 12.
Outline of PQ-producing fungi
Conclusion
Bioprospectors have been captivated by the diversity, novelty, and biological activity of fungal natural products. Perylenequinones (PQs) are naturally occurring aromatic polyketides. They are unique and fascinating compounds because of their chemical and biological properties. Fungi, aphids, crinoids, and plants are the sources of PQs. Fungal PQs have been first discovered in some phytopathogenic species, and it was subsequently recognized that PQs have a role in pathogenicity. Because of their capacity to absorb light and generate reactive oxygen species, fungal PQs possessed different bioactivities including antitumor, antiviral, antileishmanial, antimalarial, and antimicrobial properties, most notably their protein kinase C inhibitory activity. Fungal PQs are divided into two categories: class A simple PQs consists of the parent perylenequinone (4,9-dihydroxyperylene-3, l0-quinone) without carbon substituents. PQs with carbon substituents are compiled in class B. To date, 66 fungal PQs have been described from 22 genera. According to the analysis, fungi are the best source of PQs, accounting for 85 % of all known PQs, whereas only 15 % were obtained from both the kingdoms Animalia and Plantae. One phylum, Ascomycota, and four classes (Dothideomycetes, Sordariomycetes, Leotiomycetes, and Eurotiomycetes) are responsible for the documented fungal PQs. Dothideomycetes is distinguished by having the greatest diversity of PQ-producing fungi as well as the greatest amount of PQs. The families Pleosporaceae, Phaeosphaeriaceae, Cladosporiaceae, Shiraiaceae, and Hypoxylaceae could be significant sources of novel PQs. Fungal PQs merit more consideration as PDT agents, due to their advantages over other photosensitizers. Hypocrellins are an excellent example of the application of PQs. Certainly, more investigations are necessary to reveal novel fungal PQs using modern techniques.
Acknowledgements
The authors express their sincere gratitude to Prof. Marc Stadler and two anonymous reviewers for their contributions to the manuscript’s improvement.
Code availability
Not applicable.
Author contribution
Conceptualization: Afra Khiralla. Literature search and analysis: Afra Khiralla and Aisha O. Mohammed. Writing and review: Afra Khiralla. Editing and revision: Sakina Yagi.
Data availability
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Footnotes
No information available for hypomycin D structure.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Ackroyd R, Kelty C, Brown N, Reed M. The history of photodetection and photodynamic Therapy. Photochem Photobiol. 2001;74:656–669. doi: 10.1562/0031-8655(2001)074<0656:THOPAP>2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- Ahonsi MO, Maurhofer M, Boss D, Défago G. Relationship between aggressiveness of Stagonospora sp. isolates on field and hedge bindweeds, and in vitro production of fungal metabolites cercosporin, elsinochrome A and leptosphaerodione. Eur J Plant Pathol. 2005;111:203–215. doi: 10.1007/s10658-004-2878-5. [DOI] [Google Scholar]
- Al Subeh ZY, Raja HA, Monro S, et al. Enhanced production and anticancer properties of photoactivated perylenequinones. J Nat Prod. 2020;83:2490–2500. doi: 10.1021/acs.jnatprod.0c00492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson JM, Murray J (1956) lsolation of 4,9-dihytlroxy-peryecne-3,10-quinone from a fungus. Chem Ind 376–376
- Arnone A, Assante G, Di Modugno V, et al. Perylenequinones from cucumber seedlings infected with Cladosporium cucumerinum. Phytochemistry. 1988;27:1675–1678. doi: 10.1016/0031-9422(88)80424-2. [DOI] [Google Scholar]
- Arnone A, Assante G, Merlini L, Nasini G (1990) Structure and stereochemistry of cladochrome D and E, novel perylenequinone pigments from Cladosporium cladosporioides. Chem Inform 21. 10.1002/chin.199011284
- Ayers S, Zink DL, Mohn K, et al. Scutiaquinones A and B, perylenequinones from the roots of Scutia myrtina with antihelmintic activity. J Nat Prod. 2007;70:425–427. doi: 10.1021/np0604937. [DOI] [PubMed] [Google Scholar]
- Bazioli JM, Fill TP, Rocha MC, et al. Perylenequinones production induced by co-culturing Setophoma sp. and Penicillium brasilianum. Phytochem Lett. 2020;40:76–83. doi: 10.1016/j.phytol.2020.09.013. [DOI] [Google Scholar]
- Becker K, Stadler M. Recent progress in biodiversity research on the Xylariales and their secondary metabolism. J Antibiot (Tokyo) 2021;74:1–23. doi: 10.1038/s41429-020-00376-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bensch K, Groenewald JZ, Meijer M, et al. Cladosporium species in indoor environments. Stud Mycol. 2018;89:177–301. doi: 10.1016/j.simyco.2018.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bergbauer M, Eggert C, Kalnowski G. Biotreatment of pulp mill bleachery effluents with the coelomycetous fungus Stagonospora gigaspora. Biotechnol Lett. 1992;14:317–322. doi: 10.1007/BF01022331. [DOI] [Google Scholar]
- Boss D, Maurhofer M, Schläpfer E, Défago G. Elsinochrome A production by the bindweed biocontrol fungus Stagonospora convolvuli LA39 does not pose a risk to the environment or the consumer of treated crops. FEMS Microbiol Ecol. 2007;59:194–205. doi: 10.1111/j.1574-6941.2006.00207.x. [DOI] [PubMed] [Google Scholar]
- Chagas FO, Dias LG, Pupo MT. New perylenequinone derivatives from the endophytic fungus Alternaria tenuissima SS77. Tetrahedron Lett. 2016;57:3185–3189. doi: 10.1016/J.TETLET.2016.06.035. [DOI] [Google Scholar]
- Chaverri P, Liu M, Hodge KT (2008) A monograph of the entomopathogenic genera Hypocrella, Molleriella, and Samuelsia gen. nov. (Ascomycota, Hypocreales, Clavicipitaceae), and their aschersonia-like anamorphs. Stud Mycol 60. 10.3114/sim.2008.60.01 [DOI] [PMC free article] [PubMed]
- Chen H-Q, Lee M-H, Chung K-R. Functional characterization of three genes encoding putative oxidoreductases required for cercosporin toxin biosynthesis in the fungus Cercospora nicotianae. Microbiology. 2007;153:2781–2790. doi: 10.1099/mic.0.2007/007294-0. [DOI] [PubMed] [Google Scholar]
- Cheng TF, Jia XM, Ma XH, et al. Phylogenetic study on Shiraia bambusicola by rDNA sequence analyses. J Basic Microbiol. 2004;44:339–350. doi: 10.1002/jobm.200410434. [DOI] [PubMed] [Google Scholar]
- Chooi Y, Zhang G, Hu J, et al. Functional genomics-guided discovery of a light-activated phytotoxin in the wheat pathogen Parastagonospora nodorum via pathway activation. Environ Microbiol. 2017;19:1975–1986. doi: 10.1111/1462-2920.13711. [DOI] [PubMed] [Google Scholar]
- Choquer M, Lee M, Bau H, Chung K. Deletion of a MFS transporter-like gene in Cercospora nicotianae reduces cercosporin toxin accumulation and fungal virulence. FEBS Lett. 2007;581:489–494. doi: 10.1016/j.febslet.2007.01.011. [DOI] [PubMed] [Google Scholar]
- Corrochano LM. Fungal photoreceptors: sensory molecules for fungal development and behaviour. Photochem Photobiol Sci. 2007;6:725–736. doi: 10.1039/b702155k. [DOI] [PubMed] [Google Scholar]
- Cruz HJM, Boffo EF, Geris R. Perylenequinones from Curvularia lunata. Biochem Syst Ecol. 2020;92:104086. doi: 10.1016/j.bse.2020.104086. [DOI] [Google Scholar]
- Dai D, Wijayawardene NN, Tang L, et al. Rubroshiraia gen. nov., a second hypocrellin-producing genus in Shiraiaceae (Pleosporales) MycoKeys. 2019;58:1–26. doi: 10.3897/mycokeys.58.36723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daranagama DA, Liu X, Chamyuang S, et al. Rhopalostroma brevistipitatum sp. nov. from Thailand with an extended generic description for Rhopalostroma. Phytotaxa. 2015;227:229–242. doi: 10.11646/phytotaxa.227.3.2. [DOI] [Google Scholar]
- Daub ME, Herrero S, Chung K. Reactive oxygen species in plant pathogenesis : the role of perylenequinone photosensitizers. Antioxid Redox Signal. 2013;19:970–989. doi: 10.1089/ars.2012.5080. [DOI] [PubMed] [Google Scholar]
- Davis VM, Stack ME. Mutagenicity of stemphyltoxin III, a metabolite of Alternaria alternata. Appl Environ Microbiol. 1991;57:180–182. doi: 10.1128/aem.57.1.180-182.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng H, Gao R, Liao X, Cai Y. Characterisation of a monooxygenase in Shiraia bambusicola. Microbiology. 2018;164:1180–1188. doi: 10.1099/mic.0.000694. [DOI] [PubMed] [Google Scholar]
- Diwu Z. Novel therapeutic and diagnostic applications of hypocrellins and hypericins. Photochem Photobiol. 1995;61:529–539. doi: 10.1111/j.1751-1097.1995.tb09903.x. [DOI] [PubMed] [Google Scholar]
- Diwu Z, Lown JW. Photosensitization with anticancer agents: 15. Perylenequinonoid pigments as potential photodynamic therapeutic agents: Formation of semiquinone radicals and reactive oxygen species on illumination. J Photochem Photobiol B Biol. 1993;18:131–143. doi: 10.1016/1011-1344(93)80055-E. [DOI] [PubMed] [Google Scholar]
- Diwu Z, William LJ. Hypocrellins and their use in photosensitization. Photochem Photobiol. 1990;52:609–616. doi: 10.1111/j.1751-1097.1990.tb01807.x. [DOI] [PubMed] [Google Scholar]
- Dubauskas Z, Beck TP, Chmura SJ, et al. Activated calphostin C cytotoxicity is independent of p53 status and in vivo metastatic potential. Clin Cancer Res. 1998;4:2391–2398. [PubMed] [Google Scholar]
- Ehrenshaft M, Upchurch RG. Isolation of light-enhanced cDNAs of Cercospora kikuchii. Appl Environ Microbiol. 1991;57:2671–2676. doi: 10.1128/aem.57.9.2671-2676.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan XL, Barreto RW, Groenewald JZ, et al. Phylogeny and taxonomy of the scab and spot anthracnose fungus Elsinoë (Myriangiales, Dothideomycetes) Stud Mycol. 2017;87:1–41. doi: 10.1016/j.simyco.2017.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fang LZ, Qing C, Shao HJ, et al. Hypocrellin D, a Cytotoxic fungal pigment from fruiting bodies of the ascomycete Shiraia bambusicola. J Antibiot (Tokyo) 2006;59:351–354. doi: 10.1038/ja.2006.49. [DOI] [PubMed] [Google Scholar]
- Fournier J, Lechat C. Phylacia korfii sp. nov., a new species of Phylacia (Xylariaceae) from French Guiana, with notes on three other Phylacia spp. Ascomycete.org. 2015;7:315–319. doi: 10.25664/art-0154. [DOI] [Google Scholar]
- Gao SS, Li XM, Wang BG. Perylene derivatives produced by Alternaria alternata, an endophytic fungus isolated from Laurencia species. Nat Prod Commun. 2009;4:1477–1480. [PubMed] [Google Scholar]
- Goonasekara I, Erio C, Bulgakov T, et al. Two novel species of Parastagonospora (Phaeosphaeriaceae Pleosporales) on grasses from Italy and Russia. Asian J Mycol. 2019;2:170–182. doi: 10.5943/ajom/2/1/8. [DOI] [Google Scholar]
- Hanke UM, Lima-braun AL, Eglinton TI, et al. Significance of perylene for source allocation of terrigenous organic matter in aquatic sediments. Environ Sci Technol. 2019;53:8244–8251. doi: 10.1021/acs.est.9b02344. [DOI] [PubMed] [Google Scholar]
- Helaly SE, Thongbai B, Stadler M (2018) Diversity of biologically active secondary metabolites from endophytic and saprotrophic fungi of the ascomycete order Xylariales. Nat Prod Rep. 10.1039/C8NP00010G [DOI] [PubMed]
- Hu J, Sarrami F, Li H, et al. Heterologous biosynthesis of elsinochrome A sheds light on the formation of the photosensitive. Chem Sci. 2019;10:1457–1465. doi: 10.1039/c8sc02870b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudson JB, Imperial V, Haugland RP, Diwu Z. Antiviral activities of photoactive perylenequinones. Photochem Photobiol. 1997;65:352–354. doi: 10.1111/j.1751-1097.1997.tb08570.x. [DOI] [PubMed] [Google Scholar]
- Hudson JB, Zhou J, Chen J, et al. Hypocrellin, from Hypocrella bambusae, is phototoxic to human immunodeficiency virus. Photochem Photobiol. 1994;60:253–255. doi: 10.1111/j.1751-1097.1994.tb05100.x. [DOI] [PubMed] [Google Scholar]
- Idris A, Tantry MA, Ganai BA, et al. Reduced perylenequinone derivatives from an endophytic Alternaria sp. isolated from Pinus ponderosa. Phytochem Lett. 2015;11:264–269. doi: 10.1016/j.phytol.2015.01.008. [DOI] [Google Scholar]
- Iida T, Kobayashi E, Yoshida M, Sano H. Calphostins, novel and specific inhibitors of protein kinase C. II. Chemical structures. J Antibiot (Tokyo) 2012;42:1475–1481. doi: 10.7164/antibiotics.42.1475. [DOI] [PubMed] [Google Scholar]
- Itoh N, Sakagami N, Masaki T, Watanabe M. Perylene in Lake Biwa sediments originating from Cenococcum geophilum in its catchment area. Geochim Cosmochim Acta. 2012;95:241–251. doi: 10.1016/j.gca.2012.07.037. [DOI] [Google Scholar]
- Jiao WL, Liu L, Zhou RJ, et al. Elsinochrome phytotoxin production and pathogenicity of Elsinoë arachidis isolates in China. PLoS One. 2019;14:e0218391. doi: 10.1371/journal.pone.0218391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khiralla A, Spina R, Saliba S, Laurain-Mattar D. Diversity of natural products of the genera Curvularia and Bipolaris. Fungal Biol Rev. 2019;33:101–122. doi: 10.1016/J.FBR.2018.09.002. [DOI] [Google Scholar]
- Kishi T, Tahara S, Taniguchi N, et al. New perylenequinones from Shiraia bambusicola. Planta Med. 1991;57:376–379. doi: 10.1055/s-2006-960121. [DOI] [PubMed] [Google Scholar]
- Kobayashi E, Nakano H, Morimoto M, Tamaoki T. Calphostin C (UCN-1028C), a novel microbial compound, is a highly potent and specific inhibitor of protein kinase C. Biochem Biophys Res Commun. 1989;159:548–553. doi: 10.1016/0006-291X(89)90028-4. [DOI] [PubMed] [Google Scholar]
- Kobayashi T, Rasmussen C, Sumida H. Characterization of the perylenequinone pigments in Japanese Andosols and Cambisol. Soil Sci Plant Nutr. 2019;65:1–10. doi: 10.1080/00380768.2018.1536846. [DOI] [Google Scholar]
- Krishnamoorthy G, Webb SP, Nguyen T, et al. Synthesis of hydroxy and methoxy perylene quinones , their spectroscopic and computational characterization , and their antiviral activity. Photochem Photobiol Sci. 2005;81:924–933. doi: 10.1562/2004-11-23-RA-378. [DOI] [PubMed] [Google Scholar]
- Kuhnert E, Becker K, Stadler M, et al. Secondary metabolite biosynthetic diversity in the fungal family Hypoxylaceae and Xylaria hypoxylon. Stud Mycol. 2021;100118:1–43. doi: 10.1016/j.simyco.2021.100118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurobane I, Vining LC, McInnes AG, et al. Biosynthesis of elsinochromes C and D. Pattern of acetate incorporation determined by 13C and 2H NMR. Can J Chem. 2006;59:422–430. doi: 10.1139/v81-063. [DOI] [Google Scholar]
- Kuyama S, Tamura T. Cercosporin. A pigment of Cercosporina kikuchii Matsumoto et Tomoyasu. I. Cultivation of fungus, isolation and purification of pigment. J Am Chem Soc. 1957;79:5725–5726. doi: 10.1021/ja01578a038. [DOI] [Google Scholar]
- Lee I, Seok S, Kim W, Yun B. Diaporthin and orthosporin from the fruiting body of Daldinia concentrica. Mycobiology. 2006;34:38–40. doi: 10.4489/MYCO.2006.34.1.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei XY, Zhang MY, Ma YJ, Wang JW. Transcriptomic responses involved in enhanced production of hypocrellin A by addition of Triton X-100 in submerged cultures of Shiraia bambusicola. J Ind Microbiol Biotechnol. 2017;44:1–15. doi: 10.1007/s10295-017-1965-5. [DOI] [PubMed] [Google Scholar]
- Levic JT, Petrovic TM, Stankovic SC, Ivanovic DM. The incidence of Pyrenochaeta terrestris in root of different plant species in Serbia. Zb Matice Srp za Prir Nauk. 2013;125:21–30. doi: 10.2298/ZMSPN1325021L. [DOI] [Google Scholar]
- Li C, Wang HQ, Xie JL, et al. Analysis and comparisons of compounds among three medical fungi of Hypocreaceae. Chinese Tradit Herb Drugs. 2000;31:250–251. [Google Scholar]
- Li G, Wang H, Zhu R, et al. Phaeosphaerins A-F, cytotoxic perylenequinones from an endolichenic fungus, Phaeosphaeria sp. J Nat Prod. 2012;75:142–147. doi: 10.1021/np200614h. [DOI] [PubMed] [Google Scholar]
- Li T, Deng H, Zhao J, Gu Y. Elsinochrome A photosensitizers: alternative drugs for photodynamic therapy. J Innov Opt Health Sci. 2015;08:1530001. doi: 10.1142/s1793545815300013. [DOI] [Google Scholar]
- Li YT, Yang C, Wu Y, et al. Axial chiral binaphthoquinone and perylenequinones from the stromata of Hypocrella bambusae are SARS-CoV-2 entry inhibitors. J Nat Prod. 2021;84:436–443. doi: 10.1021/acs.jnatprod.0c01136. [DOI] [PubMed] [Google Scholar]
- Liao HL, Chung KR. Genetic dissection defines the roles of elsinochrome phytotoxin for fungal pathogenesis and conidiation of the citrus pathogen Elsinoe fawcettii. Mol Plant-Microbe Interact. 2008;21:469–479. doi: 10.1094/MPMI-21-4-0469. [DOI] [PubMed] [Google Scholar]
- Liu WZ, Ma LY, Li C, et al. Structural elucidation of a new perylenequinone. Acta Pharm Sin. 2001;36:313–314. [PubMed] [Google Scholar]
- Liu WZ, Shen YX, Liu XF, et al. A new perylenequinone from Hypomyces sp. Chin Chem Lett. 2001;12:431–432. [Google Scholar]
- Ma G, Khan SI, Jacob MR, et al. Antimicrobial and antileishmanial activities of hypocrellins A and B. Antimicrob Agents Chemother. 2004;48:4450–4452. doi: 10.1128/AAC.48.11.4450-4452.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma YJ, Sun CX, Wang JW. Enhanced production of hypocrellin A in submerged cultures of Shiraia bambusicola by red light. Photochem Photobiol. 2019;95:812–822. doi: 10.1111/php.13038. [DOI] [PubMed] [Google Scholar]
- Ma YJ, Zheng LP, Wang JW. Inducing perylenequinone production from a bambusicolous fungus Shiraia sp . S9 through co-culture with a fruiting body - associated bacterium Pseudomonas fulva. Microb Cell Factories. 2019;18:1–14. doi: 10.1186/s12934-019-1170-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mains EB. Species of Hypocrella. Mycopathol Mycol Appl. 1959;11:311–326. doi: 10.1007/BF02089506. [DOI] [PubMed] [Google Scholar]
- Mastrangelopoulou M, Grigalavicius M, Berg K, et al. Cytotoxic and photocytotoxic effects of cercosporin on human tumor cell lines. Photochem Photobiol. 2019;95:387–396. doi: 10.1111/php.12997. [DOI] [PubMed] [Google Scholar]
- Meille SV, Malpezzi L, Allegra G, et al. Structure of elsinochrome A: a perylenequinone metabolite. Acta Crystallogr C. 1989;45:628–632. doi: 10.1107/S0108270188012831. [DOI] [PubMed] [Google Scholar]
- Miskovsky P (2002) Hypericin, a new antiviral and antitumor photosensitizer: mechanism of action and interaction with biological macromolecules. Curr Drug Targets. 10.2174/1389450023348091 [DOI] [PubMed]
- Morakotkarn D, Kawasaki H, Tanaka K, et al. Taxonomic characterization of Shiraia-like fungi isolated from bamboos in Japan. Mycoscience. 2008;49:258–265. doi: 10.1007/S10267-008-0419-3. [DOI] [Google Scholar]
- Mulrooey CA, Morgan BJ, Li X, Kozlowski MC. Perylenequinone natural products: enantioselective synthesis of the oxidized pentacyclic core. J Organomet Chem. 2010;75:16–29. doi: 10.1021/jo9013832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulrooey CA, O’Brien EM, Morgan BJ, Kozlowski MC. Perylenequinones: isolation, synthesis, and biological activity. Eur J Org Chem. 2012;21:3887. doi: 10.1038/jid.2014.371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mumma RO, Lukezic FL, Kelly MG. Cercosporin from Cercospora hayii. Phytochemistry. 1973;12:917–922. doi: 10.1016/0031-9422(73)80703-4. [DOI] [Google Scholar]
- Newman AG, Townsend CA. Molecular characterization of the cercosporin biosynthetic pathway in the fungal plant pathogen Cercospora nicotianae. J Am Chem Soc. 2016;138:4219–4228. doi: 10.1021/jacs.6b00633.Molecular. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olivo M, Ali-Seyed M. Apoptosis signalling mechanisms in human cancer cells induced by calphostin PDT. Int J Oncol. 2007;30:537–548. [PubMed] [Google Scholar]
- Pang X, Lin X, Wang P, et al. Perylenequione derivatives with anticancer activities isolated from the marine sponge-derived fungus, Alternaria sp. SCSIO41014. Mar Drugs. 2018;16:1–13. doi: 10.3390/md16080280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patriarca A, Pinto VF. Reference Module in Food Science. Elsevier; 2018. Alternaria; pp. 1–8. [Google Scholar]
- Peter M, Kohler A, Ohm RA et al (2016) Ectomycorrhizal ecology is imprinted in the dominant symbiotic fungus Cenococcum geophilum. Nat Commun 7. 10.1038/ncomms12662 [DOI] [PMC free article] [PubMed]
- Pettit RK. Small-molecule elicitation of microbial secondary metabolites. Microb Biotechnol. 2011;4:471–478. doi: 10.1111/j.1751-7915.2010.00196.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Podlech J, Fleck SC, Metzler M, et al. Determination of the absolute configuration of perylene quinone-derived mycotoxins by measurement and calculation of electronic circular dichroism spectra and specific rotations. Chem - A Eur J. 2014;20:11463–11470. doi: 10.1002/chem.201402567. [DOI] [PubMed] [Google Scholar]
- Robeson D, Strobel G, Matusumoto GK, et al. Alteichin: an unusual phytotoxin from Alternaria eichorniae, a fungal pathogen of water hyacinth. Experientia. 1984;40:1248–1250. doi: 10.1007/BF01946657. [DOI] [PubMed] [Google Scholar]
- Samson RA, Visagie CM, Houbraken J, et al. Phylogeny, identification and nomenclature of the genus Aspergillus. Stud Mycol. 2014;78:141–173. doi: 10.1016/j.simyco.2014.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seto Y, Kogami Y, Shimanuki T, et al. Production of phleichrome by Cladosporium phlei as stimulated by diketopiperadines of Epichloe typhina. Biosci Biotechnol Biochem. 2005;69:1515–1519. doi: 10.1271/bbb.69.1515. [DOI] [PubMed] [Google Scholar]
- Sharma SK, Woo T, Naicker S (2013) Perylenequinoe drivatives and uses thereof. United States Patent No. US 8,506,931 B2. Quest Pharmatech Inc. https://patents.google.com/patent/US8506931B2/en. Accessed 20 June 2021
- Shen XY, Cheng YL, Cai CJ, et al. Diversity and antimicrobial activity of culturable endophytic fungi isolated from moso bamboo seeds. PLoS One. 2014;9:1–7. doi: 10.1371/journal.pone.0095838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen Y, Liu W, Rong X, Sun Y. Studies on the chemical constituents of a fungus producing perylenequinones. Acta Pharm Sin. 2003;38:834–837. [PubMed] [Google Scholar]
- So KK, Chun J, Kim DH. Antimicrobial and antitumor photodynamic effects of phleichrome from the phytopathogenic fungus Cladosporium Phlei. Mycobiology. 2018;46:448–451. doi: 10.1080/12298093.2018.1551599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stack ME, Prival MJ. Mutagenicity of the Alternaria metabolites altertoxins I, II, and III. Appl Environ Microbiol. 1986;52:718–722. doi: 10.1128/aem.52.4.718-722.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stadler M, Fournier J, Gardt S, Peršoh D. The phylogenetic position of Rhopalostroma as inferred from a polythetic approach. Persoonia. 2010;25:11–21. doi: 10.3767/003158510X524231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stadler M, Ju Y, Rogers JD, et al. Chemotaxonomy of Entonaema, Rhopalostroma and other Xylariaceae. Mycol Res. 2004;108:239–256. doi: 10.1017/S0953756204009347. [DOI] [PubMed] [Google Scholar]
- Stchigel AM, Calduch M, Mac Cormack W, Guarro J. Phaeosphaeria microscopica (Karsten) O. Erikss.: first report on Deschampsia antarctica Desv. Bol Micol. 2004;19:111–115. doi: 10.22370/bolmicol.2004.19.0.372. [DOI] [Google Scholar]
- Stierle AC, Caddlina JH, Strobel GA. Phytotoxins from Alternaria alternata, a pathogen of spotted knapweed. J Nat Prod. 1989;52:42–47. doi: 10.1021/np50061a003. [DOI] [Google Scholar]
- Sun CX, Ma YJ, Wang JW. Improved hypocrellin A production in Shiraia bambusicola by light-dark shift. J Photochem Photobiol B Biol. 2018;182:100–107. doi: 10.1016/j.jphotobiol.2018.04.004. [DOI] [PubMed] [Google Scholar]
- Sun CX, Ma YJ, Wang JW. Enhanced production of hypocrellin A by ultrasound stimulation in submerged cultures of Shiraia bambusicola. Ultrason Sonochem. 2017;38:214–224. doi: 10.1016/j.ultsonch.2017.03.020. [DOI] [PubMed] [Google Scholar]
- Tantry MA, Idris AS, Williamson JS, et al. Perylenequinones from an endophytic Alternaria sp. of Pinus ponderosa. Heliyon. 2018;4:1–11. doi: 10.1016/j.heliyon.2018.e01046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tisch D, Schmoll M. Light regulation of metabolic pathways in fungi. Appl Microbiol Biotechnol. 2010;85:1259–1277. doi: 10.1007/s00253-009-2320-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan XY, Chen YT. Hypocrellin A, a new drug for photochemotherapy. Chin Sci Bull. 1981;26:1040–1042. [Google Scholar]
- Wang JB, Bao JN. Clinical analysis and observation of hypocrellin photochemistry in treatment of Lichen amyloidosis in 37 patients. Acta Acad Med Sin. 1985;7:349–352. [PubMed] [Google Scholar]
- Weiss U, Altland HW. Red pigments of Dactynotus rudbeckiae and D. ambrosiae [Homoptera, Aphididae] Nature. 1965;207:1295–1297. doi: 10.1038/2071295b0. [DOI] [PubMed] [Google Scholar]
- Weiss U, Merlini L, Nasini G. Naturally occurring perylenequinones. Prog Chem Org Nat Prod. 1987;52:1–71. doi: 10.1007/978-3-7091-8906-1_1. [DOI] [PubMed] [Google Scholar]
- Wendt L, Sir EB, Kuhnert E, et al. Resurrection and emendation of the Hypoxylaceae, recognised from a multigene phylogeny of the Xylariales. Mycol Prog. 2018;17:115–154. doi: 10.1007/s11557-017-1311-3. [DOI] [Google Scholar]
- Wiehe A, O’Brien JM, Senge MO. Trends and targets in antiviral phototherapy. Photochem Photobiol Sci. 2019;18:2565–2612. doi: 10.1039/C9PP00211A. [DOI] [PubMed] [Google Scholar]
- Wijayawardene N, Hyde K, Lkt A, et al. Outline of fungi and fungus-like taxa. Mycospere. 2020;11:1060–1456. doi: 10.5943/mycosphere/11/1/8. [DOI] [Google Scholar]
- Williams RB, Henrikson JC, Hoover AR, et al. Epigenetic remodeling of the fungal secondary metabolome. Org Biomol Chem. 2008;6:1895–1997. doi: 10.1039/b804701d. [DOI] [PubMed] [Google Scholar]
- Wu H, Lao XF, Wang QW, et al. The shiraiachromes: novel fungal perylenequinone pigments from Shiraia bambusicola. J Nat Prod. 1989;52:948–951. doi: 10.1021/np50065a006. [DOI] [Google Scholar]
- Xian LP, Jing XN, Meng D, Sha Y. A new perylenequinone from the fruit bodies of Bulgaria inquinans. J Asian Nat Prod Res. 2006;8:743–746. doi: 10.1080/10286020500246626. [DOI] [PubMed] [Google Scholar]
- Yadav S, Agarwal R, Singh S, Goel S. Pyrenochaeta romeroi causing subcutaneous phaeohyphomycotic cyst in a diabetic female. Med Mycol Case Rep. 2015;8:47–49. doi: 10.1016/j.mmcr.2015.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang H, Wang BY, Zhang Z, et al. Whole-genome shotgun assembly and analysis of the genome of Shiraia sp . strain Slf14, a novel endophytic fungus producing huperzine A and hypocrellin A. Genome Announc. 2014;2:e00011–e00014. doi: 10.1128/genomeA.00011-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yano S, Hirohara S, Obata M, et al. Current states and future views in photodynamic therapy. J Photochem Photobiol C-Photochem Rev. 2011;12:46–67. doi: 10.1016/j.jphotochemrev.2011.06.001. [DOI] [Google Scholar]
- Yu C, Huang T, Ding Z, et al. Encyclopedia of Chinese Medicines, vol. 1, item 1187. Beijing, People’s Republic of China: Chinese Medicinal Science and Technology Press; 1993. [Google Scholar]
- Yu F, Jayawardena RS, Liu J, et al. Hypomyces pseudolactifluorum sp. nov. (Hypocreales: Hypocreaceae) on Russula sp. from Yunnan, PR China. Biodivers Data J. 2020;8:1–22. doi: 10.3897/BDJ.8.e53490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeiner CA, Purvine SO, Zink EM et al (2016) Comparative analysis of secretome profiles of manganese ( II )-oxidizing Ascomycete fungi. PLoS One 11. 10.1371/journal.pone.0157844 [DOI] [PMC free article] [PubMed]
- Zhang H, Bai X, Wang H. Isolation and identification of an antimicrobial and cytotoxic chlorated perylenequinone from the symbiotic fungus Aspergillus fumigatus D. Biochem Pharmacol. 2017;139:110. doi: 10.1016/J.BCP.2017.06.066. [DOI] [Google Scholar]
- Zhang SY, Li ZL, Bai J, et al. A new perylenequinone from a halotolerant fungus, Alternaria sp. M6. Chin J Nat Med. 2012;10:68–71. doi: 10.1016/S1875-5364(12)60015-6. [DOI] [PubMed] [Google Scholar]
- Zhnag H, Liu W (2005) Fungicidal composition containing PQDs. United States Patent No. US 6,936,571 B2. https://patents.google.com/patent/US6936571B2/en. Accessed 13 May 2020
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.