Abstract
Introduction
Insulin resistance (IR) plays a key role in the pathogenesis and clinical course of patients with multiple metabolic diseases and diabetes. This study aimed to explore the effect of trimetazidine (TMZ) on skeletal muscle IR in mice fed a high-fat diet (HFD) and explore the possible underlying mechanism.
Research design and methods
In vivo, a HFD mouse IR model was adopted and TMZ and exercise were used to intervene. Postintervention the following were determined: blood levels of glucose and insulin, homeostasis model assessment of IR index, expression of skeletal muscle insulin signaling-related proteins phosphorylated insulin receptor substrate 1 (p-IRS1/IRS1) and phosphorylated protein kinase B (p-AKT/AKT), nuclear factor erythroid 2 related factor 2 (Nrf2) signaling pathway, and oxidative stress. In vitro, a palmitate-treated C2C12 myotube IR model was constructed. Cellular glucose uptake, p-IRS1/IRS1, and p-AKT/AKT were determined, and reactive oxygen species (ROS) production was analyzed based on treatments with specific small interfering RNA of Nrf2 with or without TMZ. Western blot was used to obtain the protein expression level and ROS production by functional analysis kits.
Results
In vivo, TMZ and exercise decreased the blood glucose and insulin levels and homeostasis model assessment of IR index, increased skeletal muscle insulin signaling-related protein ratios of p-IRS1/IRS1 and p-AKT/AKT, and both interventions activated Nrf2 signaling and reduced oxidative stress production in HFD mice. In vitro, TMZ reduced the oxidative stress reaction, increased the ratios of p-AKT/AKT and p-IRS1/IRS1, and attenuated the insulin stimulation of PA-induced glucose uptake. However, in the absence of Nrf2, TMZ failed to resist the effects of IR.
Conclusions
This study showed that TMZ, like exercise, brought about marked improvements to HFD-induced skeletal muscle IR through TMZ, a common pathway with exercise in the form of Nrf2, regulating oxidative stress. We provide new evidence to support the use of TMZ for diabetes treatment.
Keywords: exercise; drug therapy; insulin resistance; muscle, skeletal
Significance of this study.
What is already known about this subject?
Insulin resistance (IR) plays a major role in the pathogenesis and clinical course of patients with diabetes. Nuclear factor erythroid 2 related factor 2 (Nrf2) is a key regulator of antioxidant signaling, and there are indications that the redox balance could be a critical element that contributes toward the contradictory effects of Nrf2 on insulin sensitivity and resistance. Exercise training is a major therapeutic strategy against IR. Trimetazidine (TMZ), usually a treatment for coronary heart disease, has recently been evidenced to benefit patients with diabetes.
What are the new findings?
TMZ effectively improves skeletal muscle IR through regulating the Nrf2 signaling pathway against oxidative stress. TMZ and exercise may share a common pathway in the form of Nrf2, which regulates oxidative stress.
How might these results change the focus of research or clinical practice?
Both TMZ and exercise provide comparable improvements of skeletal muscle IR. This study provides further evidence to support the use of TMZ for the treatment of diabetes.
Introduction
Insulin resistance (IR) is considered to play a major role in the pathogenesis and clinical course of patients with diabetes.1 2 With an estimated 425 million (6%) patients with diabetes worldwide,3 gaining further insights into the pathogenesis of diabetes and developing novel treatments for IR is of importance.
Skeletal muscle is the key organ in glucose disposal. As such, it is the main site and most important tissue for insulin-stimulated glucose disposal.4 Accordingly, skeletal muscle IR is often referred to as the vital element of whole-body IR. Extensive research has identified oxidative stress as a major causative factor of IR in association with a high-fat diet (HFD) and reduced physical activity.5–7 The defined molecular mechanism of skeletal muscle IR pathogenesis remains largely unknown. It has been suggested that lipid overload, specifically in mitochondria, elicits mitochondrial dysfunction, increases oxidative stress, and, therefore, impairs insulin sensitivity in muscles.8 Nuclear factor erythroid 2 related factor 2 (Nrf2) is a main antioxidant signaling regulator that could prevent the development of metabolic syndrome and related cardiovascular diseases.9 Moreover, newly published data indicate that the redox balance contributes toward the effects of Nrf2 on insulin sensitivity and resistance.10 However, due to conflicting evidence, further validation is required.
Exercise training improves metabolic homeostasis, offering protection from IR, obesity, type 2 diabetes, and cardiovascular disease.11 12 Exercise can decrease oxidative stress and support mitochondrial function. Our previous study indicated that exercise enhances mitochondrial quality control (MQC) and improves skeletal muscle function.13 14 Existing evidence also shows that the benefit from exercise is mediated through oxidative stress via the Nrf2 pathway,15 wherein exercise activates Nrf2 in cardiac and skeletal muscle, increasing endurance capacity. In addition, Nrf2, an antioxidative modulator, alleviates muscle damage initiated by oxidative stress16 and often induces antioxidant gene expression involved in MQC.
Trimetazidine (TMZ), an antiangina agent, inhibits free fatty acid oxidation and simultaneously promotes glucose oxidation by selectively acting on the mitochondrial long-chain 3-ketoacyl-CoA thiolase.17 18 TMZ also has a role in regulating oxidative stress; it upregulates Nrf2/heme oxygenase-1 (HO-1) and downregulates nuclear factor kappa B signaling, which attenuates cardiac oxidative stress and cardiomyocyte apoptosis induced by exhaustive exercise.19 Improvement of glucose utilization as well as the reduction of glycemia and glycated hemoglobin by the “comparable-to-exercise” effect of TMZ have also been observed in patients with diabetes.20 21 Further, our previous study indicated that TMZ and exercise both improve muscle function, increasing loaded swimming time and inverted hanging time in healthy mice and HFD-induced obese mice through enhancing MQC.14 22 Uruno et al found that specific induction of Nfr2 in skeletal muscles markedly ameliorated IR induced via a HFD, protecting mice from obesity.23
The present study was designed to investigate the effect of TMZ on skeletal muscle IR in HFD mice and explore its underlying mechanisms. We hypothesized that TMZ, like exercise, which has pleiotropic actions could improve glycemic control and IR.
Research design and methods
Animal experiments
Thirty-two male C57BL/6J mice (8 weeks old) were purchased from the Animal Laboratory Centre of Xiangya Medical School (Changsha, Hunan, China). These mice were kept in temperature-controlled (22°C±2°C) quarters with 12-hour cycles of light and dark and had ad libitum access to food and water.
Following 1 week of adaptive feeding, mice were randomly separated into the following four groups (n=8 for each group): normal diet (ND), HFD, HFD+exercise (EX), and HFD+TMZ. Fat made up 45% and 18% of total calories in the HFD and ND groups, respectively. Mice in the HFD+EX group underwent an exercise training program as previously described,13 22 whereas mice in the HFD+TMZ group were intragastrically administered TMZ (10 mg/kg/day; Schweageer Pharmaceuticals). Mice in the remaining groups were administered an equal amount of saline. After 8 weeks, blood and skeletal muscle samples were obtained from each group.
Exercise training
The HFD+EX group was subjected to moderate-intensity swim training.24 In brief, an adaptive training protocol was employed; mice were placed in a Morris water maze pool (XR-XM101-R, ZSdichuang, Beijing, China, with a diameter of 120 cm and a depth of 60 cm) and swam for 10 min on the first day. Thereafter, this was increased 10 min daily until 60 min/day. After swimming, towels and blowers were used to dry the mice gently. Training was undertaken between 09:00 and 14:00 when mice exhibited minimal variations in aerobic capacity.
Biochemical analysis
Blood samples were obtained from each group after 12 hours of fasting and then assayed by an insulin assay kit (A042, Nanjing Jiancheng Bioengineering Institute, China).
Homeostasis model assessment of IR (HOMA-IR) index
Plasma insulin was determined by ELISA (CSB-E05071m; Wuhan, Hubei, China) and glucose by glucometer (Accu-Chek Performa; Roche Diagnostics, Indianapolis, Indiana, USA). The HOMA-IR index was calculated via the following formula:
HOMA-IR=fasting glucose (mmol/L)×fasting insulin (mU/L)/22.5
Gene expression assay
Total RNA was extracted from skeletal (quadriceps femoris) muscles by TRIzol (Invitrogen, Carlsbad, California, USA), and the expression of Nrf2 was quantified by real-time reverse transcription PCR analysis by using the primers shown in table 1.
Table 1.
Sequence of primers used for rtPCR
| Gene | Primer | Product length |
| Nrf2 | F→CCTATGCGTGAATCCCAATG R→AAGCGGCTTGAATGTTTGTC |
104 bp |
| GAPDH | F→GCGACTTCAACAGCAACTCCC R→CACCCTGTTGCTGTAGCCGTA |
122 bp |
GAPDH, glyceraldehyde-3-phosphate dehydrogenase; Nrf2, nuclear factor erythroid 2 related factor 2.
Manganese-dependent superoxide dismutase (MnSOD) and malondialdehyde (MDA) assay
The xanthine oxidase method was used to determine MnSOD activity in muscle tissues (A001-2; Jiancheng Bioengineering Institute, Nanjing, Jiangsu, China). Mitochondria were isolated (G006; Nanjing Jiancheng Bioengineering Institute) and the thiobarbituric acid method was applied to detect the content of MDA in mitochondria (A003-2; Nanjing Jiancheng Bioengineering Institute).
Cell culture
Cell culture was performed as previously described.2 Briefly, C2C12 mouse myoblasts (Cobioer Biotechnology, Nanjing, China) were cultured in Dulbecco’s modified Eagle’s medium containing 10% fetal bovine serum and penicillin/streptomycin (5000 U/5000 µg/mL; Gibco, Grand Island, New York, USA). Myotubes were incubated with 0.75 mmol/L of palmitate (PA) postdifferentiation for 24 hours followed with or without 50 µg/mL TMZ for 24 hours.
Small interfering (siRNA) transfection
Negative control and Nrf2 siRNAs (80 nmol/L; RiboBio, Guangzhou, China) were transfected into the cells using an FECTTM CP Transfection Kit (RiboBio). The cells were then treated with or without TMZ (50 µg/mL) for 24 hours.
Glucose uptake assay
Cells treated with PA were then stimulated with 30 mU/mL insulin or vehicle for 1 hour. Following stimulation, myotubes were incubated with 500 µM of 2-(N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)amino)-2-deoxyglucose (2-NBDG, Invitrogen) for 1 hour. To remove free 2-NBDG, phosphate-buffered saline was used to wash the myotubes three times. The fluorescence intensity of cells containing 2-NBDG was analyzed using a Synergy 2 Multi-Mode Microplate Reader (BioTek) with excitation and emission at 485 and 535 nm, respectively.
Western blot analysis
Muscle tissues or cells were lyzed, and proteins were extracted for immunoblotting with antibodies, including Nrf2, NAD(P)H:quinone oxidoreductase 1 (NQO-1), HO-1, phosphorylated protein kinase B (p-AKT), AKT, insulin receptor substrate 1 (IRS-1), phosphorylated IRS-1, and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Proteintech, Rosemont, Illinois, USA).
2,7-Dichlorodihydrofluorescein diacetate (DCF) and dihydroethidium (DHE) staining
DCF and DHE staining were performed to assess ROS generation in myotubes. Briefly, myotubes were stained with 100 µmol/L DCF and DHE (Yeasen) for 90 min. The fluorescence intensity of cells containing DCF and DHE was then estimated using a Synergy 2 Multi-Mode Microplate Reader (BioTek).
Statistical analysis
All results are expressed as the mean±SE of the mean. One-way analysis of variance with Newman-Keul’s tests was used to determine differences between group mean values. The statistical significance was set at p<0.05. Statistical analysis was performed using Prism 6 software (GraphPad, V.6.01).
Results
Effects of exercise and TMZ on IR and insulin signaling
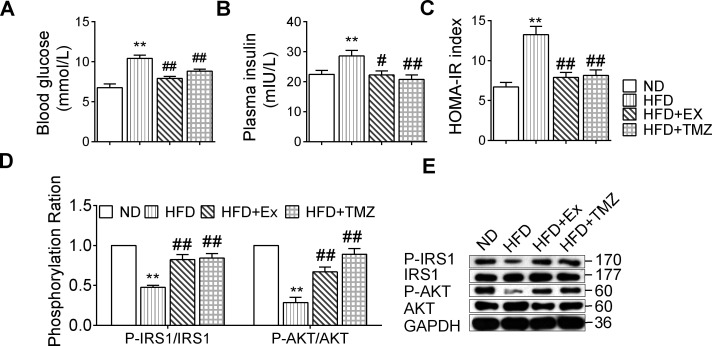
Compared with ND mice, the blood glucose in HFD mice increased, yet this decreased in HFD+EX and HFD+TMZ mice compared with the HFD group, though it remained significantly higher than in ND mice (all p<0.01; figure 1A). The insulin level in HFD mice increased (p<0.01) compared with ND mice, and this was lower in the HFD+EX (p<0.05) and HFD+TMZ (p<0.01) mice compared with the HFD group (figure 1B).
Figure 1.
Exercise and trimetazidine (TMZ) effects on insulin resistance (IR) and insulin signaling. Normal diet-group (ND), high-fat diet group (HFD), HFD+exercise group (HFD+Ex), and HFD+TMZ group (HFD+TMZ). (A) Blood glucose; (B) plasma insulin; (C) HOMA-IR index; (D, E) IRS1, p-IRS1, AKT, and p-AKT protein expression were found via western blot. *,**Represented p<0.05, p<0.01 in comparison with ND group, respectively; #,##represented p<0.05, p<0.01 in comparison with HFD group, respectively. GAPDH, glyceraldehyde-3-phosphate dehydrogenase; HOMA-IR, homeostasis model assessment of insulin resistance; p-AKT, phosphorylated protein kinase B (AKT); p-IRS1, phosphorylated insulin receptor substrate 1 (IRS-1).
The HOMA-IR index in HFD mice was significantly higher than in ND mice, and after exercise or TMZ treatment this decreased considerably to a level slightly higher than that of ND mice (p<0.01; figure 1C). The p-IRS1/IRS1 ratios, in addition to p-AKT/AKT, were significantly reduced following HFD feeding (figure 1D), while these were fully reversed by either exercise or TMZ treatment (all p<0.01; figure 1D, E).
Exercise and TMZ effects on the Nrf2 signaling pathway and oxidative stress in HFD mouse models
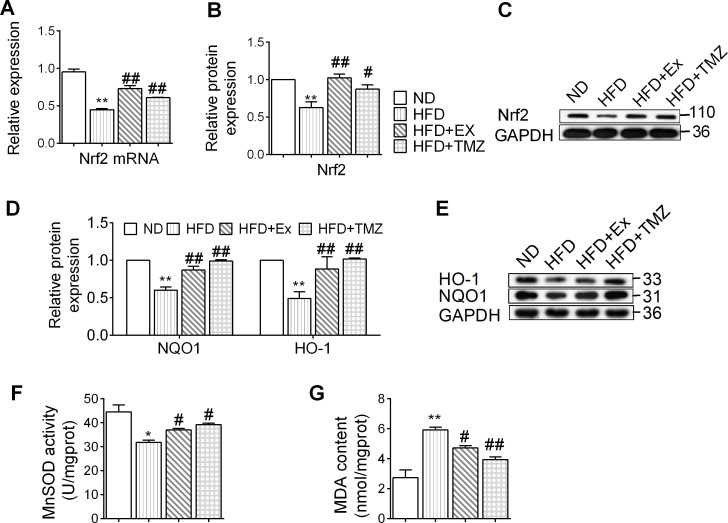
In animal models, a HFD downregulated the gene expression of Nrf2, which further contributed towards a reduction in Nrf2 protein levels (p<0.05 and p<0.01, respectively; figure 2A–C). The decreased Nrf2 expression was accompanied by a reduction in HO-1 and NQO-1 (p<0.01; figure 2D,E), suggesting that a HFD inhibits the Nrf2 signaling pathway. TMZ and exercise equally reversed the attenuated Nrf2 signaling pathway (p<0.05 and p<0.01, respectively). The HFD also induced oxidative stress characterized by a reduction in MnSOD and upregulation of MDA content. These were reversed by TMZ and exercise (p<0.05 and p<0.01, respectively; figure 2F, G).
Figure 2.
Exercise and trimetazidine (TMZ) effects on nuclear factor erythroid 2 related factor 2 (Nrf2) signaling pathway and oxidative stress in high-fat diet (HFD) mouse models. (A) Nrf2-mRNA expression was determined by quantitative PCR. (B, C) Nrf2 protein expression was determined by western blot. (D, E) Heme oxygenase-1 (HO-1), NAD(P)H:quinone oxidoreductase 1 (NQO-1) protein expression was determined by western blot. (F, G) Represented the manganese-dependent superoxide dismutase (MnSOD) activity and malondialdehyde (MDA) content, respectively. *,**Represented p<0.05, p<0.01 in comparison with normal diet (ND) group, respectively; #,##represents p<0.05, p<0.01 in comparison with HFD group, respectively. GAPDH, glyceraldehyde-3-phosphate dehydrogenase; HFD+Ex, HFD+exercise group; HFD+TMZ, HFD+TMZ group.
TMZ regulates the Nrf2 signaling pathway and resists the oxidative stress reaction induced by PA
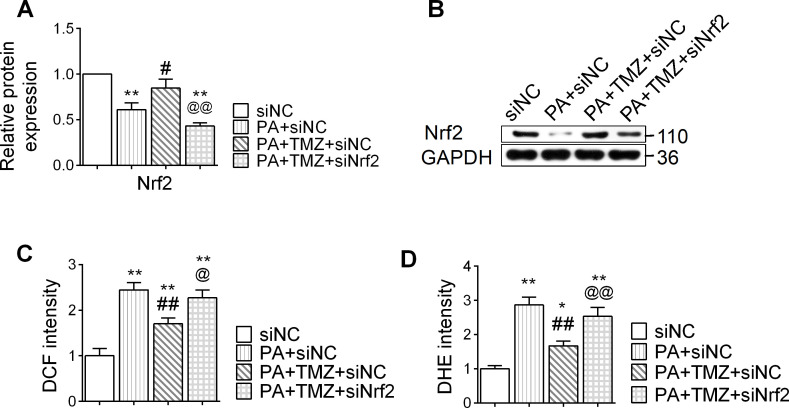
PA treatment suppressed the expression of Nrf2 in muscle cells, and TMZ treatment reversed this (p<0.05; figure 3A, B). In parallel, PA also upregulated the expression of oxidative stress markers, DHE and DCF in muscle cells, while TMZ significantly downregulated these changes (p<0.05 and p<0.01, respectively; figure 3C, D). However, this effect of TMZ was abolished by siNrf2 (Nrf2 small interfering RNA), providing direct evidence that TMZ-regulated oxidative stress was Nrf2-dependent. These results suggest that TMZ might prevent PA-induced oxidative stress through regulating the Nrf2 signaling pathway.
Figure 3.
Trimetazidine (TMZ) regulates the nuclear factor erythroid 2 related factor 2 (Nrf2) signaling pathway and resists oxidative stress reaction induced by palmitate (PA). PA, skeletal muscle cells treated with palmitate; siNC, treated with negative control small interfering RNA; siNrf2, treated with Nrf2 small interfering RNA; TMZ, skeletal muscle cells treated with TMZ. (A, B) Nrf2 protein expression was determined by western blot. (C, D) DCF: DCFH-DA (2,7-dichlorodihydrofluorescein diacetate), dihydroethidium (DHE) were detected for reactive oxygen species. *,**Represented p<0.05, p<0.01 in comparison with siNC group, respectively; #, ##represented p<0.05, p<0.01 in comparison with PA+siNC group, respectively; @,@@represented p<0.05, p<0.01 in comparison with PA+TMZ+siNC group, respectively. GAPDH, glyceraldehyde-3-phosphate dehydrogenase.
TMZ prevents PA-induced muscle cell IR via the Nrf2 signaling pathway
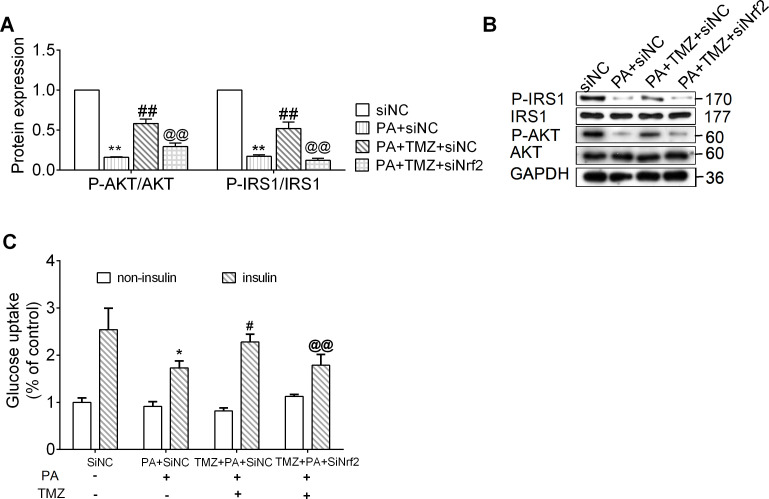
In the cell culture, we observed that p-IRS1/IRS1 and p-AKT/AKT ratios were significantly reduced in the PA-treated cells, while these were fully reversed by TMZ treatment (all p<0.01; figure 4A, B). Insulin-stimulated glucose uptake in the control cells was markedly attenuated in PA-treated cells (figure 4C). This could then be significantly counteracted by treatment with TMZ(p<0.05; figure 4C). However, in the absence of Nrf2, TMZ failed to resist the effects of IR (p<0.05 and p<0.01, respectively; figure 4A–C).
Figure 4.
Trimetazidine (TMZ) prevents muscle cells insulin resistance via the nuclear factor erythroid 2 related factor 2 (Nrf2) pathway, which regulates oxidative stress. PA, skeletal muscle cells treated with palmitate; siNC, treated with negative control small interfering RNA; siNrf2, treated with Nrf2 small interfering RNA; TMZ, skeletal muscle cells treated with TMZ. (A) Glucose uptake with insulin or with non-insulin; (B, C) IRS1, p-IRS1, AKT, and p-AKT protein expression were determined by western blot. *,**Represented p<0.05, p<0.01 in comparison with siNC group, respectively; #, ##represented p<0.05, p<0.01 in comparison with PA+siNC group, respectively; @,@@represented p<0.05, p<0.01 in comparison with PA+TMZ+ siNC group, respectively. GAPDH, glyceraldehyde-3-phosphate dehydrogenase; PA, palmitate; p-AKT, phosphorylated protein kinase B (AKT); p-IRS1, phosphorylated insulin receptor substrate 1 (IRS1).
Discussion
IR serves as a fundamental mechanism in the development of diabetes and is a vital issue for public health. In vivo, we found that TMZ successfully resisted IR induced by a HFD in mice, suggesting that TMZ has comparable effects to exercise and might form part of new complementary strategies to treat muscle IR. In addition, we found that TMZ could improve PA-induced cell IR in skeletal C2C12 cells by upregulating the expression of antioxidant factor Nrf2 and its downstream target genes, NQO-1 and HO-1. This study showed that TMZ and exercise provide analogous improvements to the skeletal muscle IR of mice through a shared Nrf2 signaling pathway.
Oxidative stress has a pivotal role in the pathogenesis of IR and diabetes.5–8 Excessive ROS production may inhibit insulin signaling, resulting in the reduced phosphorylation of IRS-1 and AKT.25 26 Lipotoxicity induced by saturated fatty acids contributes toward mitochondrial dysfunction and ROS production, leading to metabolic abnormalities and muscle IR in mice.27 28 Here, we found that, compared with an ND, 8 weeks of a HFD in mice resulted in hyperglycemia and hyperinsulinemia, with an ensuing increased HOMA-IR index. Additionally, in cultured cells, PA significantly impaired insulin-stimulated glucose uptake. It was observed that IRS1 and AKT phosphorylation decreased in HFD mice and PA-treated cells, which suggests that the IR model in mice and C2C12 cells was successfully established.
Nrf2 has been identified as a key regulator responsible for protection against oxidative stress and controlling the expression of several antioxidant genes engaged in metabolic homeostasis. The exact mechanisms underlying the role of Nrf2 in IR are not fully established, and studies on the role of Nrf2 in obesity and IR have presented conflicting findings.10 These studies, using Nrf2 KO and Keap1 KD mouse models, suggest that deletion or activation of Nrf2 in different specific tissues may lead to different effects on IR.23 29 30 In the present study, we found that PA and a HFD increased the expression of oxidative stress markers (DHE and DCF) and reduced MnSOD, leading to an upregulation in MDA content. Further, Nrf2 was also suppressed and accompanied by the reduced expressions of HO-1 and NQO-1.
Exercise is an effective strategy against IR induced by metabolic dysfunction.31 It corrects energy metabolism and increases IRS-1 and AKT phosphorylation.32 33 We confirmed that exercise significantly improves impaired metabolism and IR, as shown in previous studies,34 35 with a decreased HOMA-IR index and increased insulin sensitivity. Moreover, exercise could increase Nrf2 expression in skeletal muscles.23 The results of the present study are in agreement with previous findings showing that exercise and Stevia rebaudiana extracts attenuate diabetic cardiomyopathy by the upregulation of Nrf2 in type 2 diabetes rats.36 The findings of our study suggest that exercise may resist muscle IR following metabolic abnormalities through an Nrf2 mechanism, as demonstrated by the alleviation of muscle IR via activating the Nrf2 pathway.
TMZ, a fatty acid metabolic modulator, has also been shown to activate Nrf2 to improve oxidative stress,18 37 reduce inflammation,38 and protect cardiomyocytes from mitochondrial dysfunction induced by PA.39 Furthermore, Fragasso et al40 found that in patients with systolic heart failure, TMZ treatment was associated with improved left ventricular function and regulated whole-body energy metabolism. In a previous study, we found that the body composition in HFD mice significantly improved following TMZ treatment, with increased muscle mass, decreased visceral fat, and a trend for a decrease in body weight.22 These findings suggested that TMZ may bring about beneficial effects on IR.
Skeletal muscle is an important target organ in IR. We found that TMZ, similar to exercise, had a beneficial effect on skeletal muscle in healthy and obese mice by enhancing MQC.14 22 Previous studies on patients with diabetic have shown that TMZ improves glucose usage as well as reduces glycemia and glycated hemoglobin with effects comparable to exercise.20 21 In the present study, we observed that TMZ regulated the Nrf2 signaling pathway against oxidative stress markers MDA and increased MnSOD in HFD mice as well as decreased DCF and DHE levels in PA-treated muscle cells. The use of TMZ could repeal these impairments partially but does not influence glucose uptake without stimulating insulin. We also found that TMZ significantly reversed the lowered phosphorylation of IRS1 and AKT in obese mice and cells after treatment with PA. Therefore, it is probable that Nrf2 activation may ameliorate IR by counteracting HFD-induced oxidative stress and maintaining oxidant and antioxidant homeostasis. These results indicate that TMZ has an antioxidant effect attributed to IR reduction, and that regulation of ROS production is implicated in glucose metabolism and insulin sensitivity. Importantly, the combination treatment of TMZ with Nrf2 siRNA also led to the attenuation of the effect of TMZ, suggesting that the effects of TMZ and the signaling pathway of Nrf2 are associated. These results imply that the signaling pathways, including Nrf2 activation, oxidative stress regulation, and insulin signaling, are implicated in TMZ’s effect on IR.
Limitations and future directions
We only observed the effects of TMZ in comparison to exercise. Furthermore, in this study, we did not analyze energy expenditure. In future studies, the possible synergistic effect of TMZ along with exercise on IR could be analyzed, and further verification of the effect of TMZ on whole-body energy metabolism could be undertaken using an animal model.
Conclusions
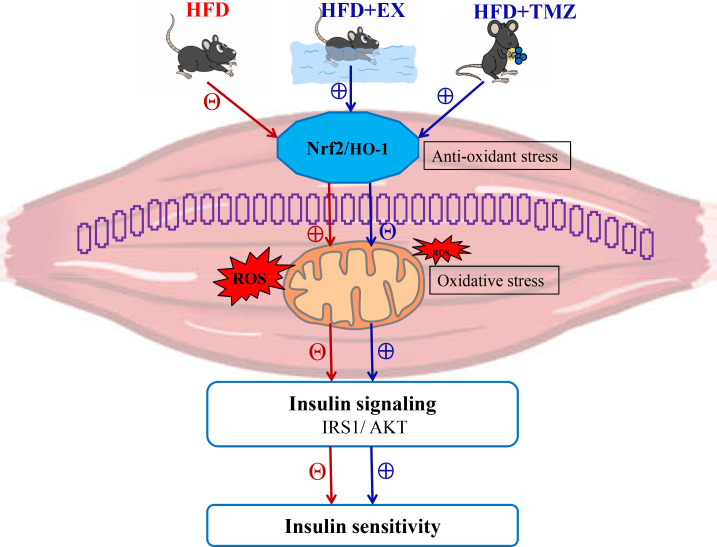
Our results show that TMZ, similar to exercise, brought about marked improvements to HFD-induced skeletal muscle IR (figure 5). Moreover, TMZ appeared to share a common pathway with exercise in the form of Nrf2, which regulated oxidative stress. These findings provide auxiliary evidence to support the use of TMZ in the treatment of diabetes.
Figure 5.
Proposed pathway of trimetazidine (TMZ) effects on high-fat diet (HFD)-induced muscle insulin resistance (IR). The blue and red arrows show the effects of a HFD and TMZ on physiological processes; ⊕ signals the facilitating effects and Θ the inhibiting effects. Fatty acids stimulate the mitochondrial production of reactive oxygen species (ROS) and inhibit the nuclear factor erythroid 2 related factor 2 (Nrf2) pathway in IR skeletal muscle, contributing to oxidative stress. The excessive ROS production leads to insulin signaling IRS1/AKT (insulin receptor substrate 1/protein kinase B) inhibition and adversely impacts insulin sensitivity. Though, exercise and TMZ were observed to activate Nrf2 signaling control, lowering mitochondrial ROS, resulting in improved insulin sensitivity. GAPDH, glyceraldehyde-3-phosphate dehydrogenase; HFD+Ex, HFD+exercise group.
Footnotes
Contributors: WZ, YD, and SL designed and performed most of the study and wrote the paper; BY analyzed and interpreted the data; SF, LQ, JC, and CL performed part of the study; JWR-G and YD critically revised and edited the manuscript; SL supervised the study; grants from SL, YD, and JC funded the study. SL is the guarantor of this work. All authors have read and agreed to the final version of the manuscript.
Funding: This work was supported by grants from the National Nature Science Foundation of China (grant no: 81672262 to SL; grant no: 82002403 to YD); and the Shenzhen Science and Technology Innovation Committee (grant no: JCYJ20180302150133743 to SL and JC).
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement
All data relevant to the study are included in the article.
Ethics statements
Patient consent for publication
Not required.
Ethics approval
All the procedures involving mice were conducted under the guidelines for the use of live animals of the National Institutes of Health and were approved by the Animal Ethics Committee of Xiangya Medical School, Central South University (Changsha, China) (approval ID: SYXK 2015-0017), and in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
References
- 1.Reaven GM. Relationships among insulin resistance, type 2 diabetes, essential hypertension, and cardiovascular disease: similarities and differences. J Clin Hypertens 2011;13:238–43. 10.1111/j.1751-7176.2011.00439.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.You B, Dun Y, Zhang W, et al. Anti-insulin resistance effects of salidroside through mitochondrial quality control. J Endocrinol 2020;244:383–93. 10.1530/JOE-19-0393 [DOI] [PubMed] [Google Scholar]
- 3.Glovaci D, Fan W, Wong ND. Epidemiology of diabetes mellitus and cardiovascular disease. Curr Cardiol Rep 2019;21:21. 10.1007/s11886-019-1107-y [DOI] [PubMed] [Google Scholar]
- 4.Turcotte LP, Fisher JS. Skeletal muscle insulin resistance: roles of fatty acid metabolism and exercise. Phys Ther 2008;88:1279–96. 10.2522/ptj.20080018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Anderson EJ, Lustig ME, Boyle KE, et al. Mitochondrial H2O2 emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans. J Clin Invest 2009;119:573–81. 10.1172/JCI37048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Krebs M, Roden M. Nutrient-induced insulin resistance in human skeletal muscle. Curr Med Chem 2004;11:901–8. 10.2174/0929867043455620 [DOI] [PubMed] [Google Scholar]
- 7.Popkin BM. Global nutrition dynamics: the world is shifting rapidly toward a diet linked with noncommunicable diseases. Am J Clin Nutr 2006;84:289–98. 10.1093/ajcn/84.2.289 [DOI] [PubMed] [Google Scholar]
- 8.Yazici D, Sezer H, Resistance I. Obesity and lipotoxicity. Adv Exp Med Biol 2017;960:277–304. [DOI] [PubMed] [Google Scholar]
- 9.Jiang S, Yang Y, Li T, et al. An overview of the mechanisms and novel roles of Nrf2 in cardiovascular diseases. Expert Opin Ther Targets 2016;20:1413–24. 10.1080/14728222.2016.1250887 [DOI] [PubMed] [Google Scholar]
- 10.Li S, Eguchi N, Lau H, et al. The role of the Nrf2 signaling in obesity and insulin resistance. Int J Mol Sci 2020;21:21186973. 10.3390/ijms21186973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cai Y, Xie K-L, Zheng F, et al. Aerobic exercise prevents insulin resistance through the regulation of miR-492/Resistin axis in aortic endothelium. J Cardiovasc Transl Res 2018;11:450–8. 10.1007/s12265-018-9828-7 [DOI] [PubMed] [Google Scholar]
- 12.Liu S, Zheng F, Cai Y, et al. Effect of long-term exercise training on lncRNAs expression in the vascular injury of insulin resistance. J Cardiovasc Transl Res 2018;11:459–69. 10.1007/s12265-018-9830-0 [DOI] [PubMed] [Google Scholar]
- 13.Dun Y, Liu S, Zhang W, et al. Exercise combined with Rhodiola sacra supplementation improves exercise capacity and ameliorates exhaustive exercise-induced muscle damage through enhancement of mitochondrial quality control. Oxid Med Cell Longev 2017;2017:8024857 10.1155/2017/8024857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Xie M, Jiang L, Dun Y, et al. Trimetazidine combined with exercise improves exercise capacity and anti-fatal stress ability through enhancing mitochondrial quality control. Life Sci 2019;224:157–68. 10.1016/j.lfs.2019.03.027 [DOI] [PubMed] [Google Scholar]
- 15.Kasai S, Shimizu S, Tatara Y, et al. Regulation of Nrf2 by mitochondrial reactive oxygen species in physiology and pathology. Biomolecules 2020;10:10020320. 10.3390/biom10020320 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kim Y, Kim C-S, Joe Y, et al. Quercetin reduces tumor necrosis factor alpha-induced muscle atrophy by upregulation of heme oxygenase-1. J Med Food 2018;21:551–9. 10.1089/jmf.2017.4108 [DOI] [PubMed] [Google Scholar]
- 17.Molinari F, Pin F, Gorini S, et al. The mitochondrial metabolic reprogramming agent trimetazidine as an 'exercise mimetic' in cachectic C26-bearing mice. J Cachexia Sarcopenia Muscle 2017;8:954–73. 10.1002/jcsm.12226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhao L. Protective effects of trimetazidine and coenzyme Q10 on cisplatin-induced cardiotoxicity by alleviating oxidative stress and mitochondrial dysfunction. Anatol J Cardiol 2019;22:232–9. 10.14744/AnatolJCardiol.2019.83710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang H, Liu M, Zhang Y, et al. Trimetazidine attenuates exhaustive exercise-induced myocardial injury in rats via regulation of the Nrf2/NF-κB signaling pathway. Front Pharmacol 2019;10:175. 10.3389/fphar.2019.00175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fragasso G, Piatti Md PM, Monti L, et al. Short- and long-term beneficial effects of trimetazidine in patients with diabetes and ischemic cardiomyopathy. Am Heart J 2003;146:854. 10.1016/S0002-8703(03)00415-0 [DOI] [PubMed] [Google Scholar]
- 21.Monti LD, Setola E, Fragasso G, et al. Metabolic and endothelial effects of trimetazidine on forearm skeletal muscle in patients with type 2 diabetes and ischemic cardiomyopathy. Am J Physiol Endocrinol Metab 2006;290:E54–9. 10.1152/ajpendo.00083.2005 [DOI] [PubMed] [Google Scholar]
- 22.Zhang W, You B, Qi D, et al. Trimetazidine and exercise provide comparable improvements to high fat diet-induced muscle dysfunction through enhancement of mitochondrial quality control. Sci Rep 2021;11:19116. 10.1038/s41598-021-98771-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Uruno A, Yagishita Y, Katsuoka F, et al. Nrf2-mediated regulation of skeletal muscle glycogen metabolism. Mol Cell Biol 2016;36:1655–72. 10.1128/MCB.01095-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jiang L, Shen X, Dun Y, et al. Exercise combined with trimetazidine improves anti-fatal stress capacity through enhancing autophagy and heat shock protein 70 of myocardium in mice. Int J Med Sci 2021;18:1680–6. 10.7150/ijms.53899 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Al-Lahham R, Deford JH, Papaconstantinou J. Mitochondrial-generated ROS down regulates insulin signaling via activation of the p38MAPK stress response pathway. Mol Cell Endocrinol 2016;419:1–11. 10.1016/j.mce.2015.09.013 [DOI] [PubMed] [Google Scholar]
- 26.Kanuri BN, Rebello SC, Pathak P, et al. Glucose and lipid metabolism alterations in liver and adipose tissue pre-dispose p47phox knockout mice to systemic insulin resistance. Free Radic Res 2018;52:568–82. 10.1080/10715762.2018.1453136 [DOI] [PubMed] [Google Scholar]
- 27.Bonnard C, Durand A, Peyrol S, et al. Mitochondrial dysfunction results from oxidative stress in the skeletal muscle of diet-induced insulin-resistant mice. J Clin Invest 2008;118:789–800. 10.1172/JCI32601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang J, Yang X, Zhang J. Bridges between mitochondrial oxidative stress, ER stress and mTOR signaling in pancreatic β cells. Cell Signal 2016;28:1099–104. 10.1016/j.cellsig.2016.05.007 [DOI] [PubMed] [Google Scholar]
- 29.Chartoumpekis DV, Palliyaguru DL, Wakabayashi N, et al. Nrf2 deletion from adipocytes, but not hepatocytes, potentiates systemic metabolic dysfunction after long-term high-fat diet-induced obesity in mice. Am J Physiol Endocrinol Metab 2018;315:E180–95. 10.1152/ajpendo.00311.2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu Z, Dou W, Ni Z, et al. Deletion of Nrf2 leads to hepatic insulin resistance via the activation of NF-κB in mice fed a high-fat diet. Mol Med Rep 2016;14:1323–31. 10.3892/mmr.2016.5393 [DOI] [PubMed] [Google Scholar]
- 31.Sampath Kumar A, Maiya AG, Shastry BA, et al. Exercise and insulin resistance in type 2 diabetes mellitus: a systematic review and meta-analysis. Ann Phys Rehabil Med 2019;62:98–103. 10.1016/j.rehab.2018.11.001 [DOI] [PubMed] [Google Scholar]
- 32.Pesta D, Roden M. The Janus head of oxidative stress in metabolic diseases and during physical exercise. Curr Diab Rep 2017;17:41. 10.1007/s11892-017-0867-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Qi J, Luo X, Ma Z, et al. Swimming exercise protects against insulin resistance via regulating oxidative stress through Nox4 and Akt signaling in high-fat diet-fed mice. J Diabetes Res 2020;2020:2521590 10.1155/2020/2521590 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 34.Liu S-X, Zheng F, Xie K-L, et al. Exercise reduces insulin resistance in type 2 diabetes mellitus via mediating the lncRNA MALAT1/MicroRNA-382-3p/Resistin axis. Mol Ther Nucleic Acids 2019;18:34–44. 10.1016/j.omtn.2019.08.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ryan BJ, Schleh MW, Ahn C, et al. Moderate-Intensity exercise and high-intensity interval training affect insulin sensitivity similarly in obese adults. J Clin Endocrinol Metab 2020;105:dgaa345:e2941–59. 10.1210/clinem/dgaa345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hussein AM, Eid EA, Bin-Jaliah I, et al. Exercise and Stevia rebaudiana (R) extracts attenuate diabetic cardiomyopathy in type 2 diabetic rats: possible underlying mechanisms. Endocr Metab Immune Disord Drug Targets 2020;20:1117–32. 10.2174/1871530320666200420084444 [DOI] [PubMed] [Google Scholar]
- 37.Wan P, Su W, Zhang Y, et al. Trimetazidine protects retinal ganglion cells from acute glaucoma via the Nrf2/HO-1 pathway. Clin Sci 2017;131:2363–75. 10.1042/CS20171182 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Liang Y, Ren K, Xu X-D, et al. Trimetazidine attenuates diabetic inflammation via Nrf2 activation. Int J Cardiol 2020;307:153. 10.1016/j.ijcard.2019.10.003 [DOI] [PubMed] [Google Scholar]
- 39.Kuzmicic J, Parra V, Verdejo HE, et al. Trimetazidine prevents palmitate-induced mitochondrial fission and dysfunction in cultured cardiomyocytes. Biochem Pharmacol 2014;91:323–36. 10.1016/j.bcp.2014.07.022 [DOI] [PubMed] [Google Scholar]
- 40.Fragasso G, Salerno A, Lattuada G, et al. Effect of partial inhibition of fatty acid oxidation by trimetazidine on whole body energy metabolism in patients with chronic heart failure. Heart 2011;97:1495–500. 10.1136/hrt.2011.226332 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data relevant to the study are included in the article.