Abstract
Background:
The aim of our study is to determine the clinical availability accessibility of cancer ratio and cancer ratio plus formulations, previously validated and reported to have clinical value in distinguishing malignant pleural effusion from tuberculosis pleurisy and parapneumonic effusion.
Materials and Methods:
Retrospective study of patients hospitalized with Malignant Pleural Effusion (MPE), tuberculosis (TPE) and pararapneumonic effusion (PPE) between 2009 and 2018.
Results:
Totally 232 patients, 101(43.5 %) having MPE, 86 (37.1 %) having PPE and 45 (19.4 %) TPE were examined. When compared with each other, “serum LDH / PS Lymphocyte %”, “Cancer ratıo” and “Cancer ratıo plus” values were statistically different between the groups (p = 0.021, p <0.001 and p = 0.015, respectively). In multivariate logistic regression analysis, cancer ratio, serum LDH: pleural fluid lymphocyte count ratio was in positive correlation with MPE. The sensitivity and specificity of “cancer ratio”, “cancer ratio plus” and “ratio of serum LDH: pleural fluid lymphocyte count” were 84.2 % (95% CI 75.6– 90.7) and 52.7 (95% CI 43.8– 61.5), and 82.2 % (95% CI 73.3– 89.1) and 45.8 (95%CI 37.1– 54.7), 53.5% (95% CI 43.3– 63.5) and 67.2% (95% CI 0.68–0.94) at the cut-off level of >14.25, >28.7, and >636, respectively. When considering only MPE and TPE patients, the specificity of cancer ratıo and cancer ratıo plus increased.
Conclusion:
The cancer ratio plus rate (the ratio of “cancer ratio”formulation to the percentage of differential pleural lymphocyte count) was almost the same as the cancer ratio in separating the malignant pleural effusion from the TPE and PPE, while it has better specificity only in differentiating malignant effusions from tuberculosis effusions.
Keywords: Pleural Effusion, Malignant, Tuberculosis, Parapneumonic effusion, Cancer ratio, Cancer ratio plus
INTRODUCTION
Pleural Effusion (PE) is frequent in patients admitted to emergency departments of respiratory or thoracic diseases (1). Etiologies of PE are different, and common causes are tuberculous pleural effusion (TPE), parapneumonic effusion (PPE), malignant pleural effusion (MPE), hearth failure (HF) and others (2). Differentiatiol diagnosis between specific diseases in exudative effusion requires detailed evaluation of pleural fluid, total and differential cell count, Ph and glucose levels, adenosine deaminase (ADA) activity, as well as cytological and microbiological examination. If the diagnosis is not certaine, more invasive diagnostic procedures is necessary. MPE is usually diagnosed with PE cytology or thoracentesis with pleural biopsy. Cytology is an inexpensive diagnostic tool with high specificity but 0.6 sensitivity (3), depending on size and stage of primary tumour. Pleural biopsy is often employed to diagnose MPE but it is an invasive tool and has complications (e.g. pain, subcutaneous emphysema and bleeding) (2).
Recently some parameters have been used to differentiate malignant pleural effusion from non-malignant effusion. In 2016, Verma et al. argued that serum lactate dehydrogenase (LDH) to pleural fluid adenosine deaminase (ADA) ratio (named as cancer ratio, CR) had high diagnostic accuracy for MPE (4). At the cut off level of more than 20, CR yielded high sensitivity and related to the observations that MPE usually associates with high serum LDH levels, while TPE -with elevated pleural fluid ADA levels (4)
Also there is more lymphocyte dominance in TPE than MPE. Though in the initial stage of TPE, neutrophil dominance is known, therefore, it is claimed that the cancer ratıo plus obtained by adding pleural fluid lymphocyte count to the cancer ratio can help clinicians in distinguishing the causes from benign pleural effusion (TPE,PPE) from MPE (4). In the study of Verma et al., only patients who had MPE and TPE were included.(4) In our study, in addition to MPE and TPE, we included PPEs also, which are the most common diseases in the etiology of pleural effusion to see its potential prediction in the nearly all pleural effusions except transudates and emphysema. The aim of our study is to determine the clinical availability of cancer ratio and cancer ratio plus, previously validated, in distinguishing malignant pleural effusion from tuberculosis pleurisy and parapneumonic effusion.
MATERIALS AND METHODS
Study Population
The study was planned as a retrospective, cross sectional study. Between January 2009–December 2018 in only one of the inpatient services of Health Sciences Izmir Suat Seren Chest Diseases and Surgery Research Center of, a total of 232 patients, 101 patients with MPE, 86 patients with PPE and 45 patients who had TPE were included in the study.
We analyzed pleural fluid values taken only by first thoracentesis in patients presenting with pleural effusion. Recurrent thoracentesis were not considered.
For the diagnosis of malignant pleurisy, the detection of malignancy was accepted in the pathological examinations of samples taken from patients with thoracentesis, pleural biopsy, transthoracic fine needle aspiration biopsy or video assisted thoracoscopy.
For the diagnosis of tuberculosis pleurisy, at least one of the following criteria was required:
Tuberculosis bacilli isolation from pleural fluid or pleural tissue
Detection of Acid Fast Bacili (AFB) positivity or caseous granuloma structure in pleural tissue
Response to anti-tuberculosis treatment even though AFB is negative in pleural tissue
Positive for tuberculosis bacillus in sputum culture and exclusion of other causes in a patient with pleural fluid
A significant decline in pleurisy by anti-tuberculous therapy and clinical improvement
Criteria for the diagnosis of PPE: No malignant cell in pleural fluid, diagnosis of TBP and lung TB excluded, the dominance of neutrophil, bacterial growth in nonspecific culture, nonspecific inflammation detected in the pleural biopsy, and exudative fluid responsive to antibiotic therapy.
Exclusion Criteria
Patients below 15 years of age
Patients with suspected pregnancy or pregnancy
Patients with collagen tissue disease and other etiologies.
Clinical conditions that increase serum LDH (such as sepsis, cerebrovascular disease, hepatitis, hemolytic anemia)
Patients with complicated PPE (emphysema)
There was only one case with pleural effusion due to lymphoma; ıt was not included into the data set, because the lymphocyte count was too high.
Academic Board approval was obtained from the training planning board of T.R. Izmir University of Health Sciences Suat Seren Chest Diseases and Surgery Research Center with protocol number 48865165-302.14.01. The names, protocol numbers, age, and gender of the patients included in the study were recorded. LDH, ADA, LDH/ADA ratio, glucose, albumin, protein, pH and serum albumin, protein, LDH, glucose, ADA levels, pleural fluid examination, culture, and cytology were recorded.
Pleural fluid ADA level measurement
A minimum of 2 ml pleural fluid obtained by thoracentesis was transferred to SST plastic gel flat biochemistry tube with yellow cap and delivered to our hospital biochemistry laboratory on the same day. After 10 minutes of centrifugation at 3000 rpm, the BENBiochemicalenterprise ADA kit was quantitatively assayed using the kinetic method in the autoanalyzer (RocheCobas ® 6000 c-501). The value range was 4–20 U/L.
Pleural fluid LDH measurement
A minimum of 2 ml pleural fluid obtained by thoracentesis was transferred to SST plastic gel flat biochemistry tube with yellow cap and delivered to the biochemical laboratory of our hospital on the same day. After 10 minutes of centrifugation at 3000 rpm, the Roche ® LDH kit was run by the enzymatic method on the same day in the autoanalyzer (RocheCobas ® 6000 c-501). The value range was 135–225 U/L.
We calculated and analysed four ratios:
Cancer ratıo: The ratio between serum LDH - pleural ADA
Cancer ratio plus: The ratio of cancer ratio to the percentage of differential pleural lymphocyte count
Pleural Neutrofil/pleural lymphocyte ratio: The ratio of pleural neutrophil count to pleural lymphocyte.
Serum LDH/pleural lymphocyte %: The ratıo of serum LDH to the percentage of pleural lymphocyte.
Statistical Analysis
Analyses of data was made with Statistical Package for the Social Sciences (SPSS, Inc., Chicago IL), version 22, software for Windows. Shapiro-Wilk test was employed for determining whether the data were distributed normally. It was decided that the data were distributed normally because p value was greater than 0.05. One-way ANOVA test was used for comparing serum and pleural fluid parameters between the groups. After the evaluation of the One-way ANOVA results, Tukey and Games-Howell tests were used for post-hoc analysis. The results are presented as mean±standard deviation. Chi square test and Exact test were used to compare qualitative data between the groups and the results were presented in n and (%). ROC analysis was made for evaluating the diagnostic strength of the parameters obta ined by calculating from the data of serum and pleural fluid. The area under the curve (AUC), sensitivity and selectivity values, negative and positive predictive values and negative and positive likelihood ratio were determined for each parameter. Cut-off values were found for each parameter using Youden Index calculation. The results were presented with 95% CI. P<0.05 was accepted statistically significant in all tests.
RESULTS
Demographic characteristics of patients
Totaly 232 patients who had exudative pleural effusion were analysed: 101(43.5 %) had MPE, 86 (37.1 %) had PPE and 45 (19.4 %) had TPE. Of those who had MPE, the etiology of malignancy was; primary lung cancer (n = 82), mesothelioma (n = 7), and metastatic carcinoma (n = 15). Characteristics and laboratory values of patient are given in Table 1.
Table 1.
Patient characteristics
| Age | (15,0–94,0) | 61,3±18,8 |
| Gender (N,%) | ||
| Male | 149 | %64,2 |
| Female | 83 | %35,8 |
| Diagnosis | ||
| Pneumonia | 86 | %37,1 |
| Malignancy | 101 | %43,5 |
| -Adenocarcinoma | 55 | %23.7 |
| -Non small cell carcinoma | 14 | %13.5 |
| -Squamous carcinoma | 3 | %2.9 |
| -Small cell carcinoma | 10 | %4.3 |
| -Mesothelioma | 7 | %3 |
| - Metastatic carcinoma | 15 | %6.5 |
| Tuberculosis | 45 | %19,4 |
| Additional disease | 112 | %48,3 |
| Hypertension | 34 | %14,7 |
| Diabetes mellitus | 19 | %8,2 |
| Chronıc Obstructive Pulmonary | 53 | %22,8 |
| Disaese | ||
| Congestive Failure | 30 | %12,9 |
| A history of malignancy | 108 | %46,6 |
| Cytology | ||
| Malignant | 101 | %43,5 |
| Benign | 131 | %56,5 |
PostHOC Analysis between the Groups
When the patient groups with PPE, MPE and TPE were compared, serum LDH / PS Lymphocyte %, Cancer ratio and Cancer ratıo plus values were all statistically different between the groups. (p = 0.021, p <0.001 and p = 0.015, respectively). Post HOC analyzes were applied to statistically reveal the differences between the pairs of each groups. According to Post HOC analysis results, there was a statistical difference in comparison of MPE – TPE and comparison of PPE-TPE (p=0.010 and p=0.037, respectively) for serum LDH / PS Lymphocyte %, but conversely no statistical difference was detected in PPEMPE comparison. In terms of cancer ratio (for cancer ratio), there was a statistical difference in comparison of MPE-TPE and comparison of TPE-PPE (p <0.001 and p <0.001, respectively), and no statistical difference in PPE-MPE comparison was revealed.
Serum LDH/PS lymphocyte % rate was statistically higher in MPE than TPE and PPE. Cancer ratıo plus was 340.5 ± 819.1 in MPE, 35.2±125.4 ın TPE and 171.3±468.0. While cancer ratio and cancer ratio plus parameters are not statistically significant between PPE and MPE; it was statistically higher in MPE compared to TPE. There was a statistical difference in the comparison of MPE-TPE and TPE-PPE for cancer ratio plus (p = 0.001 and p = 0.034, respectively), and no statistical difference in the comparison of PPE-MPE (Table 2).
Table 2.
Comparison of demographic data and clinical parameters of patients according to their last diagnosis (post hoc evaluation P1, P2, P3)
| PPE n=86 | MPE n=101 | TPE n=45 | P değeri | P1 | P2 | P3 | |
|---|---|---|---|---|---|---|---|
| Age | 64,6±17,7 | 66,5±13,1 | 43,4±21,2 | <0,001 | 0,697 | <0,001 | <0,001 |
| Gender | |||||||
| Male | 64 %74,4 | 58 %57,4 | 27 %60,0 | 0,043 | |||
| Female | 22 %25,6 | 43 %42,6 | 18 %40,0 | ||||
| Serum parameters | |||||||
| Glucose | 151,6±97,4 | 128,0±54,5 | 111,0±37,3 | 0,006 | 0,117 | 0,002 | 0,077 |
| Protein | 6,8±0,8 | 6,6±0,7 | 7,0±0,7 | 0,005 | 0,115 | 0,053 | 0,001 |
| Albumin | 3,4±0,7 | 3,5±0,6 | 3,6±0,5 | 0,449 | 0,352 | 0,241 | 0,659 |
| Lactate dehydrogenase | 231,3±230,5 | 349,6±421,6 | 231,3±112,2 | 0,020 | 0,043 | 1,000 | 0,026 |
| Neutrophil | 7323,5±3949,7 | 8524,0±5326,7 | 6080,9±3301,2 | 0,009 | 0,184 | 0,141 | 0,003 |
| Lymphocytes | 1412,3±749,7 | 1534,8±2517,6 | 1818,7±2896,8 | 0,587 | 0,697 | 0,304 | 0,461 |
| RDW | 15,3±2,8 | 15,3±2,7 | 14,1±2,7 | 0,031 | 0,980 | 0,018 | 0,014 |
| MPV | 8,3±1,1 | 8,2±1,0 | 8,1±1,1 | 0,507 | 0,450 | 0,261 | 0,592 |
| Neutrophil/Lymphocyte | 7,5±7,5 | 8,6±9,7 | 5,5±6,2 | 0,096 | 0,301 | 0,203 | 0,032 |
| Pleural effusion parameters | |||||||
| Glucose | 132,7±77,0 | 109,1±58,5 | 81,1±41,6 | <0,001 | 0,055 | <0,001 | 0,004 |
| Protein | 4,3±1,0 | 4,4±0,8 | 5,0±0,8 | <0,001 | 0,921 | <0,001 | <0,001 |
| Albumin | 2,3±0,7 | 2,5±0,6 | 2,7±0,5 | 0,002 | 0,034 | 0,001 | 0,070 |
| ADA | 14,9±24,0 | 11,5±9,0 | 37,1±21,3 | <0,001 | 0,425 | <0,001 | <0,001 |
| LDH | 412,0±538,8 | 627,4±591,3 | 695,3±755,5 | 0,015 | 0,017 | 0,012 | 0,534 |
| Neutrophil | 1307,2±1835,1 | 1147,7±2685,4 | 1336,2±2933,7 | 0,872 | 0,659 | 0,949 | 0,669 |
| Lymphocytes | 1148,5±1345,6 | 943,6±1125,2 | 2100,0±1961,5 | <0,001 | 0,504 | 0,013 | 0,001 |
| Lymphocytes% | 0,4±0,3 | 0,4±0,3 | 0,7±0,3 | <0,001 | 0,799 | <0,001 | <0,001 |
| Neutrophil/Lymphocyte | 3,8±7,8 | 4,2±10,9 | 1,3±3,1 | 0,159 | 0,948 | 0,026 | 0,037 |
| Calculated parameters | |||||||
| Serum Lactate dehydrogenase/Pleural fluid lymphocyte % | 1288,1±2339,7 | 2424,3±5513,0 | 597,1±756,1 | 0,021 | 0,148 | 0,037 | 0,010 |
| Cancer ratio | 30,2±33,8 | 44,1±48,0 | 8,8±7,4 | <0,001 | 0,057 | <0,001 | <0,001 |
| Cancer ratio plus | 171,3±468,0 | 340,5±819,1 | 35,2±125,4 | 0,015 | 0,185 | 0,034 | 0,001 |
Parameters of Pleural Effusion between Malignant and Benign Pleural Effusion
When we look at malignant effusion and pleural effusion due to benign causes, cancer ratio, cancer ratio plus and serum LDH / PS Lymphocyte % were found to be statistically significantly higher in MPE (Table 3).
Table 3.
Demographic and laboratory characteristics of patients between malignant and benign pleural effusion
| Malignant Effusion (n=101) | Benign effusion (n=131) | P değeri | |
|---|---|---|---|
| Age | 66,5±13,1 | 57,3±21,4 | <0,001 |
| Gender | |||
| Male | 58 %57,4 | 91 %69,5 | 0,058 |
| Female | 43 %42,6 | 40 %46,9 | |
| Serum parameters | |||
| Glucose | 128,0±54,5 | 137,6±84,0 | 0,291 |
| Protein | 6,6±0,7 | 6,8±0,8 | 0,008 |
| Albumin | 3,5±0,6 | 3,5±0,7 | 0,637 |
| Lactate dehydrogenase | 349,6±421,6 | 231,3±197,5 | 0,010 |
| neutrophil | 8524,0±5326,7 | 6896,6±3773,5 | 0,010 |
| lymphocytes | 1534,8±2517,6 | 1551,9±1801,5 | 0,952 |
| RDW | 15,3±2,7 | 14,9±2,8 | 0,247 |
| MPV | 8,2±1,0 | 8,2±1,1 | 0,764 |
| Neutrophil/Lymphocytes | 8,6±9,7 | 6,8±7,1 | 0,080 |
| Pleural fluid parameters | |||
| Glucose | 109,1±58,5 | 115,0±71,2 | 0,501 |
| Protein | 4,4±0,8 | 4,6±1,0 | 0,062 |
| Albumin | 2,5±0,6 | 2,5±0,6 | 0,490 |
| Adenosine deaminase | 11,5±9,0 | 22,5±25,3 | <0,001 |
| Lactate dehydrogenase | 627,4±591,3 | 509,3±633,4 | 0,149 |
| Neutrophil | 1147,7±2685,4 | 1317,2±2261,7 | 0,603 |
| Lymphocytes | 943,6±1125,2 | 1475,3±1640,7 | 0,004 |
| Lymphocytes % | 0,4±0,3 | 0,5±0,3 | 0,061 |
| Neutrophil/Lymphocytes | 4,2±10,9 | 3,0±6,6 | 0,267 |
| Calculated parameters | |||
| Serum Lactate dehydrogenase/Pleural fluid lymphocyte % | 2424,3±5513,0 | 1050,8±1970,1 | 0,018 |
| Cancer ratıo | 44,1±48,0 | 22,8±29,5 | <0,001 |
| Cancer ratıo plus | 340,5±819,1 | 124,6±390,8 | 0,016 |
ROC Analysis of Parameters In Distinguishing Malignant Pleural Effusion From Tuberculosis Effusion And Parapneumonic Effusion Cut-off Level for Cancer Ratio (Serum LDH: Pleural Fluid ADA)
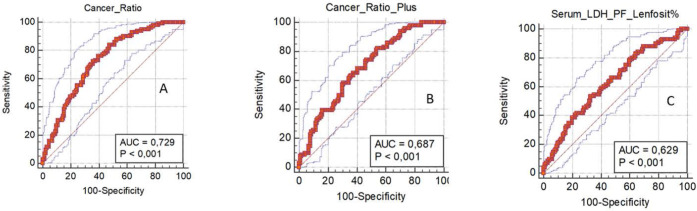
At cut-off level of > 14,25, the sensitivity and specificity of “cancer ratio” were 84.2 % (95% CI 75.6– 90.7) and 52.7 (95% CI 43.8– 61.5), respectively. The positive likelihood ratio (PLR) value was 1.7, while the negative likelihood ratio (NLR) at this cut-off was found to be 0.30 (Table 4). Area under the curve (AUC) was 0.729 (Figure 1).
Table 4.
ROC analysis of serum and pleural fluid parameters In Distinguishing Malignant Pleural Effusion From Tuberculosis Effusion And Parapneumonic Effusion
| AUC (95% Cl) | P value | Cut off value | Sensitivity (95% Cl) | Specificity (95% Cl) | PPV (95% Cl) | NPV (95% Cl) | Positive Likelihood Ratio (95% Cl) | Negative Likelihood Ratio (95% Cl) | |
|---|---|---|---|---|---|---|---|---|---|
| Serum NLR | 0,592 (0,517–0,665) | 0,015 | >6,27 | 49,50% (39,4–59,6) | 69,5% (60,8–77,2) | 55,6% (47,5–63,4) | 64,1% (58,8–69,1) | 1,62 (1,2–2,2) | 0,73 (0,6–0,9) |
| Pleural fluid NLR | 0,535 (0,512–0,658) | 0,023 | >0,27 | 83,2% (74,4–89,9) | 32,1% (24,2–40,8) | 48,6% (44,9–52,2) | 71,2% (60,0–80,3) | 1,22 (1,1–1,4) | 0,52 (0,3–0,9) |
| Serum LDH/Pleural Fluid lymphocytes % | 0,629 (0,557–0,701) | 0,001 | >636,51 | 53,5% (43,3–63,5 | 67,2% (58,4–75,1 ) | 55,7% (48,1–63,0) | 65,2% (59,5–70,4) | 1,63 (1,2–2,2) | 0,69 (0,5–0,9) |
| Cancer ratio | 0,729 (0,665–0,793) | <0,001 | >14,25 | 84,2% (75,6–90,7) | 52,7% (43,8–61,5) | 61,6% (54,8–68,0) | 73,3% (66,8–79,0) | 1,78 (1,5–2,2) | 0,30 (0,5–0,6) |
| Cancer ratio plus | 0,687 (0,620–0,754) | <0,001 | >28,71 | 82,2% (73,3–89,1) | 45,8% (37,1–54,7) | 53,9% (49,4–58,4) | 76,9% (67,8–84,1) | 1,52 (1,3–1,8) | 0,39 (0,2–0,6) |
NLR: Neutrophil lymphocytes ratio. AUC: Area under curve PPV: Positive predictive value NPV: Negative predictive value CI:Confidence Interval
Figure 1.
ROC analysis for Cancer Ratio (Serum LDH: Pleural Fluid ADA) In Distinguishing Malignant Pleural Effusion From Tuberculosis Effusion and Parapneumonic Effusion B) ROC analysis for Cancer Ratio Plus (Cancer Ratio: Pleural Fluid Lymphocyte Count) In Distinguishing Malignant Pleural Effusion From Tuberculosis Effusion and Parapneumonic Effusion C) ROC analysis for Serum LDH: Pleural Lymphocyte Count Ratio In Distinguishing Malignant Pleural Effusion From Tuberculosis Effusion and parapneumonic Effusion
Cut-Off Level for Cancer Ratio Plus (Cancer Ratio: Pleural Fluid Lymphocyte Count).
At cut-off level of > 28.7, the sensitivity and specificity of “cancer ratio plus” were 82.2 % (95% CI 73.3– 89.1) and 45.8 (95%CI 37.1– 54.7), respectively. The PLR value was 1.52, while NLR at this cut-off was found to be 0.39. AUC was 0.68 (Table 4) (Figure 1).
Cut-Off Level for Serum LDH: Pleural Lymphocyte Count Ratio
In serum LDH: pleural lymphocyte count ratio, the optimum sensitivity and specificity was at cut-off level of ≥ 636. The sensitivity was 53.5% (95% CI 43.3– 63.5) and specificity was 67.2% (95% CI 0.68–0.94). These values were lower than the sensitivity but higher than specificity of “cancer ratio” and “cancer ratio plus.” Area under the curve on the ROC curve was 0.629 (Figure 1).
ROC Analysis Of Parameters In Distinguishing Malignant Pleural Effusion From Tuberculosis Effusion Cut-off Level for Cancer Ratio (Serum LDH: Pleural Fluid ADA)
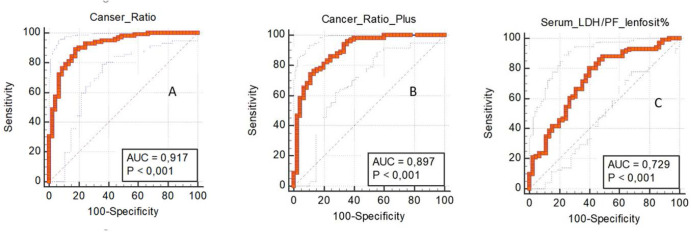
For “cancer ratio” at cut-off level of >12,13, the sensitivity and specificity were 89,1% (81,3-94,4) and 82,2% (67,9–92,0) respectively. The positive likelihood ratio (PLR) value was 5,01 (2,7–9,4) while the negative likelihood ratio (NLR) at this cut-off was found to be 0,13 (Table 5). Area under the curve (AUC) was 0.917 (Table 5) (Figure 2).
Table 5.
ROC analysis of serum and pleural fluid parameters In Distinguishing Malignant Pleural Effusion From Tuberculosis Effusion
| AUC (95% Cl) | P value | Cut off value | Sensitivity (95% Cl) | Specificity (95% Cl) | PPV (95% Cl) | NPV (95% Cl) | Positive Likelihood Ratio (95% Cl) | Negative Likelihood Ratio (95% Cl) | |
|---|---|---|---|---|---|---|---|---|---|
| Serum NLR | 0,661 (0,578–0,737) | <0,001 | >6,1 | 50,5% (40,4–60,6) | 80,0% (65,4–90,4) | 85,0%% (75,4–91,3) | 41,9% (36,0–47,9) | 2,52 (1,4–4,7) | 0,62 (0,5–0,8) |
| Pleural fluid NLR | 0,738 (0,659–0,807) | <0,001 | >0,14 | 93,1% (86,2–97,2) | 46,7% (31,7–62,1) | 79,7% (74,8–83,8) | 75,0% (57,9–86,7) | 1,75 (1,3–2,3) | 0,15 (0,07-0,3) |
| Serum LDH/Pleural Fluid lymphocytes % | 0,729 (0,649–0,799) | <0,001 | >313,55 | 80,2% (71,1–87,5) | 60,0% (44,3–74,3) | 81,8% (75,6–86,7) | 57,4% (46,0–68,1) | 2,00 (1,4–2,9) | 0,33 (0,2–0,5) |
| Cancer ratio | 0,917 (0,860–0,956) | <0,001 | >12,13 | 89,1% (81,3–94,4) | 82,2% (67,9–92,0) | 91,8% (85,7–95,6) | 77,1% (65,4–85,7) | 5,01 (2,7–9,4) | 0,13 (0,07–0,2) |
| Cancer ratio plus | 0,897 (0,836–0,941) | <0,001 | >36,88 | 74,3% (64,6–82,4) | 88,9% (75,9–96,3) | 93,7% (86,7–97,2) | 60,6 (52,1–68,5) | 6,68 (2,9–15,4) | 0,29 (0,2–0,4) |
NLR: Neutrophil lymphocytes ratio. AUC: Area under curve PPV: Positive predictive value NPV: Negative predictive value CI: Confidence interval
Figure 2.
A) ROC analysis for Cancer Ratio (Serum LDH: Pleural Fluid ADA) In Distinguishing Malignant Pleural Effusion From Tuberculosis Effusion B) ROC analysis for Cancer Ratio Plus (Cancer Ratio: Pleural Fluid Lymphocyte Count) In Distinguishing Malignant Pleural Effusion From Tuberculosis Effusion C) ROC analysis for Serum LDH: Pleural Lymphocyte Count Ratio In Distinguishing Malignant Pleural Effusion From Tuberculosis Effusion
Cut-Off Level for Cancer Ratio Plus (Cancer Ratio: Pleural Fluid Lymphocyte Count).
For “cancer ratio plus” at cut-off level of > 36,88, the sensitivity and specificity were 74,3% (64,6–82,4) and 88,9% (75,9–96,3) respectively. The PLR value was 6,68, while NLR at this cut-off was found to be 0.29. AUC was 0.897 (Table 5) (Figure 2).
Cut-Off Level for Serum LDH: Pleural Lymphocyte Count Ratio.
For the formulation of serum LDH: pleural lymphocyte count ratio, the optimum sensitivity and specificity was obtained at the cut-off level of ≥ 313,5. The sensitivity was 80,2% (71,1–87,5) and specificity was 60,0% (44,3–74,3). AUC was 0.629 (Table 5) (Figure 2).
Logıstic Regression Analysis
“Cancer ratio”, serum LDH: pleural fluid lymphocyte count ratio”, supplied significance as positive predictors of MPE in multivariate logistic regression analysis, Table 6.
Table 6.
Logistic regression analysis for prediction of malignancy
| Beta | OR | 95% Cl for OR | P value | |
|---|---|---|---|---|
| Serum NLR | 0,023 | 1,023 | 0,983–1,065 | 0,261 |
| Pleural fluid NLR | −0,114 | 0,892 | 0,787–1,011 | 0,075 |
| Serum LDH/Pleural Fluid lymphocytes % | 0,000 | 1,000 | 1,000–1,001 | 0,034 |
| Cancer ratio | 0,013 | 1,013 | 1,001–1,025 | 0,029 |
| Cancer ratio plus | 0,000 | 1,000 | 0,999–1,001 | 0,817 |
NLR: Neutrophil lymphocytes ratio OR: Odds Ratio CI: Confidence Interval
DISCUSSION
In this study, we found that serum LDH, Cancer Ratio (serum LDH: Pleural fluid ADA) and Cancer Ratio Plus (Cancer Ratio: Pleural Fluid Lymphocyte Count) are significantly higher in patients with malignant pleural effusion and may be usefull to differ malignant effusions from non-malignant effusion. Particularly, in separating MPE from TPE and PPE a cut-off level for cancer ratio > 14,25 and a cut off level for Cancer Ratio Plus > 28.7 are highly predictive of malignancy with high sensitivity but low specificity. However, when we received (considered) only MPE and TPE patient groups, we found that the cancer ratio and cancer ratio plus increased the specificity of separating MPE from TPE. This shows us that cancer ratio and cancer ratio plus can be used in the clinic especially to separate MPE from TPE, but both parameters are not superior to each other. This finding highlighted the idea that it can help clinician’s decision to manage early treatment especially in MPE and TPE.
Serum LDH is a ubiquitous cellular enzyme that rises non-specifically in response to tissue damage. Consequently, serum LDH level may increase in many clinical situations (5). But isolated serum LDH elevation may be a marker for specific diagnostic groups. Its diagnotic and prognostic roles were reported as a poor prognostic marker for sepsis and cancer patients (6–12). The proposed explanation for the elevated levels in cancer is the preferred use of glycolysis by tumor cells for energy rather than oxidative phosphorylation, which is a key in LDH-mediated ATP production pathway (13). We found similar correlation between increased serum LDH and malignant pleural effusion as in previously reported studies (14,15).
ADA is secreted by mononuclear cells, lymphocytes, neutrophils and red blood cells (16,17). There are two types, ADA-1 and ADA-2, but in routine clinical practice, only total ADA is measured. High levels are associated with infectious conditions such as TB (ADA-2) and empyema (ADA-1). In addition, ADA is employed to diagnose tuberculosis, and ADA level in pleural effusion helps in early diagnosis of tuberculosis pleurisy, and is also an important indicator for distinguishing tuberculosis pleural effusion and MPE (16). In our study, ADA level was 14.9 ± 24, 11.5 ± 9 and 37.1 ± 21.3 in PPE, MPE and TPE, respectively. The statistically significant lower ADA levels in MPE was compatible with other studies (4,18, 19). In addition, in another study, the level of ADA in PPE was higher at significant levels than that of MPE, as in our study (20). For this reason, low ADA level shows the possibility of high MPE. On the other hand, previous studies have shown that the level of LDH in serum is elevated in MPE than TPE and PPE, and that elevated serum LDH is associated with the possibility of high MPE (21,22).
The number of median lymphocytes in our study was higher in TPE than in MPE, which is in line with the results of studies conducted earlier. High lymphocyte levels in pleural fluid was reported to be related to TPE; and 67% of patients who had TPE in one study had pleural lymphocyte rate by >95% (23). In a previously-conducted study with 245 patients who had TPE, >50% of leukocytes were lymphocytes in pleural fluid with mean ± SD of 77 ± 19.9 and median (range) of 80.5 (2–100%) (24). In a study conducted with 382 patients who had TPE, median lymphocyte percentage was 84% in total cells (25).
In our study, we showed that cancer ratio and cancer ratio plus can be used to define MPEs from TPE and PPE with high sensitivity (84%, 82%, respectively) and AUC (0.729, 0.687, respectively). However, in our study, the specificity of cancer ratio was found lower than other studies (4,18,19,26). The specificity calculated for cancer ratio and cancer ratio plus in distinguishing MPE from TPE and PPE were 52.7% and 45.8%, respectively in our study. Verma et al. reported the specificity as 0.94 and 0.85 in their two previous studies. The difference can be explained with different inclusion criteria and characteristics in the study groups. Unlike our study, in the study in which the cancer ratio plus assessment was performed by Verma et al., the study population included only MPE and TPE patients (26). In addition, in both studies of Verma et al. most MPE patients were lung cancer patients (95% and 97.6%, respectively), while the number of TPE patients was less than in our study. In our study, 88.11% (101/89) of MPE patients were with lung cancer, while the number of patients with tuberculosis-related pleural effusion (TPE) was also higher (19.4%, 45/232). In addition, MPE and TPE patients were present in our study population, as well as PPE patients. But when we evaluated only patients with MPE and TPE, we found the specificity of the cancer ratio and cancer ratio plus were high in distinguishing MPE from TPE, as in the study by Verma and colleagues.
In Zhang et al.’s study in which 987 patients were included, the rate of patients with lung cancer was 91.8%; sensitivity was 94.03%, and specificity was 72.65% and AUC was 0.841 and these values were also found to be close to values of our study if the cut off value reported for cancer ratio was taken 10.6 and above (18). Contrary to our study, the number of patient populations of the two other studies was lower (19,27). In the study by Elmahalawy et al., 60 patients (20 malignant, 20 PPE, 20 TPE) were evaluated, and the cut off value was reported as 5.03, and the specificity, sensitivity and AUC values were found to be 100%, 87% and 1.0, respectively (19). As seen, when different diagnosis groups included ın the study, so patient population affects the specificity and sensitivity of the cancer ratio. Repeating the study with more patients will help to show the diagnostic significance of this value. In our study, while the cancer ratio plus had almost the same sensitivity in differentiating MPE from TPE and PPE compared to the cancer ratio, the specificity was found to be lower. But when we exclude PPEs, we found that the specificity of cancer ratio and cancer ratio plus parameters increased in distinguishing MPE from TPE.
It has been shown that cancer ratio can be used as the result of our study and of a meta-analysis performed by Han et al. which includes all of these studies mentioned above in diagnosing MPE (28). The sensitivity, specificity, PPD, NPD and AUC values determined in our study were lower than the values obtained in this meta-analysis. Although sensitivity and specificity are two basic diagnostic tools, they are not effective alone. The area under the ROC curve has also been recognized worldwide to support diagnosis. The area under the ROC curve is between 0.5 and 1.0 and the higher it is, the higher the diagnostic accuracy. When our study with meta-analysis was evaluated, our values were found to be low, but it was found to be close to the specified values except for specificity. This shows that the cancer ratio and cancer ratio plus values we found in this retrospective study in terms of MPE can be diagnostic.
Also, the multivariate analysis revealed that the cancer ratio was significant in predicting MPE, while the cancer ratio plus was not significant. In conclusion, we also showed that cancer ratio plus formulation created by adding pleural fluid lymphocyte value to cancer ratio does not add any extra value in distinguishing MPE. We attribute the reason for this to the existence of PPE cases other than TPE and MPE in our study.
The most important limitation of our study was that it is performed retrospectively and single-centered. Therefore, only routine biomarkers of blood and pleural fluid were included in this study. The use of additional new potential biomarkers that reflect systemic inflammatory and pleural responses (like serum CRP, pleural fluid ADA / serum CRP, pCEA….) could be more useful in predicting the diagnosis. Because of the retrospective nature of the study, the timing of thoracentesis and sampling pleural fluid could not be standardized. Therefore, especially in TPEs the lymphocytic dominance of pleural fluid and the level of ADA may change in an increasing direction in a week. In addition, we cannot completely exclude the potential effect of empirical antibiotics given to all patients who had TPE and patients who had PPE at the beginning before the implementation of thoracentesis on pleural fluid analysis. Another limitation is that our study consisted only of patients with MPE, TPE and PPE; it did not include patients with other causes of exudative pleural effusion. Patients with comorbidities were not included as they were relatively low in number and was heterogeneous. Thirdly, the cytological type of the primary tumour and the stage of cancer with malignant pleural effusion or presence of liver, bone metastases and the amount of pleural fluid that may affect serum LDH in all patients is unspecified. However, by seeing that such subgroup analyzes have not been conducted in other previous studies as well, and prospectively, we think that new studies whose methodology will be constructed in this way may yield interesting data.
In conclusion the cancer ratio plus rate: the ratio of cancer ratio to the percentage of differential pleural lymphocyte count was almost the same the cancer ratio in separating the malignant pleural effusion from the TPE and PPE, while it has better specificity only in differentiating malignant effusions from tuberculosis effusions.
REFERENCES
- 1.Porcel JM, Azzopardi M, Koegelenberg CF, Maldonado F, Rahman NM, Lee YC. The diagnosis of pleural effusions. Expert Rev Respir Med 2015;9(6):801–15. [DOI] [PubMed] [Google Scholar]
- 2.Wang XJ, Yang Y, Wang Z, Xu LL, Wu YB, Zhang J, et al. Efficacy and safety of diagnostic thoracoscopy in undiagnosed pleural effusions. Respiration 2015;90(3):251–5. [DOI] [PubMed] [Google Scholar]
- 3.Assawasaksakul T, Boonsarngsuk V, Incharoen P. A comparative study of conventional cytology and cell block method in the diagnosis of pleural effusion. J Thorac Dis 2017;9(9):3161–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Verma A, Abisheganaden J, Light RW. Identifying Malignant Pleural Effusion by A Cancer Ratio (Serum LDH: Pleural Fluid ADA Ratio). Lung 2016;194(1):147–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lott JA, Nemensanszky E. Lactate dehydrogenase. In: Lott JA, Wolf PL. (eds) Clinical enzymology, a case oriented approach. Year Book Medical, New York, 1987; pp 213–244. [Google Scholar]
- 6.Suárez-Santamaría M, Santolaria F, Pérez-Ramírez A, Alemán-Valls MR, Martínez-Riera A, González-Reimers E, et al. Prognostic value of inflammatory markers (notably cytokines and procalcitonin), nutritional assessment, and organ function in patients with sepsis. Eur Cytokine Netw 2010;21(1):19–26. [DOI] [PubMed] [Google Scholar]
- 7.Trédan O, Ray-Coquard I, Chvetzoff G, Rebattu P, Bajard A, Chabaud S, et al. Validation of prognostic scores for survival in cancer patients beyond first-line therapy. BMC Cancer 2011;11:95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Steyerberg EW, Keizer HJ, Fosså SD, Sleijfer DT, Bajorin DF, et al. Resection of residual retroperitoneal masses in testicular cancer: evaluation and improvement of selection criteria. The ReHiT study group. Re-analysis of histology in testicular cancer. Br J Cancer 1996;74(9):1492–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.You B, Tranchand B, Girard P, Falandry C, Ribba B, Chabaud S, et al. Etoposide pharmacokinetics and survival in patients with small cell lung cancer: a multicentre study. Lung Cancer 2008;62(2):261–72. [DOI] [PubMed] [Google Scholar]
- 10.Spiess PE, Pettaway CA, Vakar-Lopez F, Kassouf W, Wang X, Busby JE, et al. Treatment outcomes of small cell carcinoma of the prostate: a single-center study. Cancer 2007;110(8):1729–37. [DOI] [PubMed] [Google Scholar]
- 11.Füssenich LM, Desar IM, Peters ME, Teerenstra S, van der Graaf WT, Timmer-Bonte JN, et al. A new, simple and objective prognostic score for phase I cancer patients. Eur J Cancer 2011;47(8):1152–60. [DOI] [PubMed] [Google Scholar]
- 12.Terpos E, Katodritou E, Roussou M, Pouli A, Michalis E, Delimpasi S, et al. High serum lactate dehydrogenase adds prognostic value to the international myeloma staging system even in the era of novel agents. Eur J Haematol 2010;85(2):114–9. [DOI] [PubMed] [Google Scholar]
- 13.Goldman RD, Kaplan NO, Hall TC. Lactic Dehydrogenase in Human Neoplastic Tissues. Cancer Res 1964;24389–99 [PubMed] [Google Scholar]
- 14.Nishio K, Jacobson KB, Jenkins VK, Upton AC. Studies on plasma lactic dehydrogenase in mice with myeloid leukemia. I. Relation of enzyme level to course of disease. Cancer Res 1963;23340–3 [PubMed] [Google Scholar]
- 15.Porcel JM, Esquerda A, Bielsa S. Diagnostic performance of adenosine deaminase activity in pleural fluid: a single-center experience with over 2100 consecutive patients. Eur J Intern Med 2010;21(5):419–23. [DOI] [PubMed] [Google Scholar]
- 16.Boonyagars L, Kiertiburanakul S. Use of adenosine deaminase for the diagnosis of tuberculosis: a review. J Infect Dis Antimicrob Agents 2010;27(2):111–8. [Google Scholar]
- 17.Valdés L, San José E, Alvarez D, Valle JM. Adenosine deaminase (ADA) isoenzyme analysis in pleural effusions: diagnostic role, and relevance to the origin of increased ADA in tuberculous pleurisy. Eur Respir J 1996;9(4):747–51. [DOI] [PubMed] [Google Scholar]
- 18.Zhang F, Hu L, Wang J, Chen J, Chen J, Wang Y. Clinical value of jointly detection serum lactate dehydrogenase/pleural fluid adenosine deaminase and pleural fluid carcinoembryonic antigen in the identification of malignant pleural effusion. J Clin Lab Anal 2017;31(5):e22106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Elmahalawy II, Abdelaal GA, Alhanafy AM, Montaser BA. Role of cancer ratio and carcinogenic embryonic antigen ratio in diagnosis of pleural effusion. Egypt J Chest Dis Tuberc 2017;66(4):671–4. [Google Scholar]
- 20.Chung W, Jung Y, Lee K, Park J, Sheen S, Park K. CXCR3 ligands in pleural fluid as markers for the diagnosis of tuberculous pleural effusion. Int J Tuberc Lung Dis 2017;21(12):1300–6. [DOI] [PubMed] [Google Scholar]
- 21.Pan Y, Bai W, Chen J, Mao Y, Qian X, Xu K, et al. Diagnosing malignant pleural effusion using clinical and analytical parameters. J Clin Lab Anal 2019;33(2):e22689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Elsammak MY, Attia A, Hassan HA, Zaytoun TM, Shorman M, Suleman M. Evaluation of pleural fluid human epididymis 4 (HE4) as a marker of malignant pleural effusion. Tumour Biol 2012;33(5):1701–7. [DOI] [PubMed] [Google Scholar]
- 23.Berger HW, Mejia E. Tuberculous pleurisy. Chest 1973;63(1):88–92. [DOI] [PubMed] [Google Scholar]
- 24.Valdés L, Alvarez D, San José E, Penela P, Valle JM, García-Pazos JM, et al. Tuberculous pleurisy: a study of 254 patients. Arch Intern Med 1998;158(18):2017–21. [DOI] [PubMed] [Google Scholar]
- 25.Ruan SY, Chuang YC, Wang JY, Lin JW, Chien JY, Huang CT, et al. Revisiting tuberculous pleurisy: pleural fluid characteristics and diagnostic yield of mycobacterial culture in an endemic area. Thorax 2012;67(9):822–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Verma A, Dagaonkar RS, Marshall D, Abisheganaden J, Light RW. Differentiating Malignant from Tubercular Pleural Effusion by Cancer Ratio Plus (Cancer Ratio: Pleural Lymphocyte Count). Can Respir J 2016;2016:7348239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Korczyński P, Mierzejewski M, Krenke R, Safianowska A, Light RW. Cancer ratio and other new parameters for differentiation between malignant and nonmalignant pleural effusions. Pol Arch Intern Med 2018;128(6):354–361. [DOI] [PubMed] [Google Scholar]
- 28.Han YQ, Zhang L, Yan L, Ouyang PH, Li P, Hu ZD. Diagnostic accuracy of cancer ratio for malignant pleural effusion: a systematic review and meta-analysis. Ann Transl Med 2019;7(20):554. [DOI] [PMC free article] [PubMed] [Google Scholar]