Abstract
Background
Infants born preterm (before 37 weeks' gestation) are at risk of respiratory distress syndrome (RDS) and need for respiratory support due to lung immaturity. One course of prenatal corticosteroids, administered to women at risk of preterm birth, reduces the risk of respiratory morbidity and improves survival of their infants, but these benefits do not extend beyond seven days. Repeat doses of prenatal corticosteroids have been used for women at ongoing risk of preterm birth more than seven days after their first course of corticosteroids, with improvements in respiratory outcomes, but uncertainty remains about any long‐term benefits and harms. This is an update of a review last published in 2015.
Objectives
To assess the effectiveness and safety, using the best available evidence, of a repeat dose(s) of prenatal corticosteroids, given to women who remain at risk of preterm birth seven or more days after an initial course of prenatal corticosteroids with the primary aim of reducing fetal and neonatal mortality and morbidity.
Search methods
For this update, we searched Cochrane Pregnancy and Childbirth's Trials Register, ClinicalTrials.gov, the WHO International Clinical Trials Registry Platform (ICTRP), and reference lists of retrieved studies.
Selection criteria
Randomised controlled trials, including cluster‐randomised trials, of women who had already received one course of corticosteroids seven or more days previously and were still at risk of preterm birth, randomised to further dose(s) or no repeat doses, with or without placebo. Quasi‐randomised trials were excluded. Abstracts were accepted if they met specific criteria. All trials had to meet criteria for trustworthiness, including a search of the Retraction Watch database for retractions or expressions of concern about the trials or their publications.
Data collection and analysis
We used standard Cochrane Pregnancy and Childbirth methods. Two review authors independently selected trials, extracted data, and assessed trial quality and scientific integrity. We chose primary outcomes based on clinical importance as measures of effectiveness and safety, including serious outcomes, for the women and their fetuses/infants, infants in early childhood (age two to less than five years), the infant in mid‐ to late childhood (age five to less than 18 years) and the infant as an adult. We assessed risk of bias at the outcome level using the RoB 2 tool and assessed certainty of evidence using GRADE.
Main results
We included 11 trials (4895 women and 5975 babies). High‐certainty evidence from these trials indicated that treatment of women who remain at risk of preterm birth seven or more days after an initial course of prenatal corticosteroids with repeat dose(s) of corticosteroids, compared with no repeat corticosteroid treatment, reduced the risk of their infants experiencing the primary infant outcome of RDS (risk ratio (RR) 0.82, 95% confidence interval (CI) 0.74 to 0.90; 3540 babies; number needed to treat for an additional beneficial outcome (NNTB) 16, 95% CI 11 to 29) and had little or no effect on chronic lung disease (RR 1.00, 95% CI 0.83 to 1.22; 5661 babies). Moderate‐certainty evidence indicated that the composite of serious infant outcomes was probably reduced with repeat dose(s) of corticosteroids (RR 0.88, 95% CI 0.80 to 0.97; 9 trials, 5736 babies; NNTB 39, 95% CI 24 to 158), as was severe lung disease (RR 0.83, 95% CI 0.72 to 0.97; NNTB 45, 95% CI 27 to 256; 4955 babies). Moderate‐certainty evidence could not exclude benefit or harm for fetal or neonatal or infant death less than one year of age (RR 0.95, 95% CI 0.73 to 1.24; 5849 babies), severe intraventricular haemorrhage (RR 1.13, 95% CI 0.69 to 1.86; 5066 babies) and necrotising enterocolitis (RR 0.84, 95% CI 0.59 to 1.22; 5736 babies).
In women, moderate‐certainty evidence found little or no effect on the likelihood of a caesarean birth (RR 1.03, 95% CI 0.98 to 1.09; 4266 mothers). Benefit or harm could not be excluded for maternal death (RR 0.32, 95% 0.01 to 7.81; 437 women) and maternal sepsis (RR 1.13, 95% CI 0.93 to 1.39; 4666 mothers). The evidence was unclear for risk of adverse effects and discontinuation of therapy due to maternal adverse effects. No trials reported breastfeeding status at hospital discharge or risk of admission to the intensive care unit.
At early childhood follow‐up, moderate‐ to high‐certainty evidence identified little or no effect of exposure to repeat prenatal corticosteroids compared with no repeat corticosteroids for primary outcomes relating to neurodevelopment (neurodevelopmental impairment: RR 0.97, 95% CI 0.85 to 1.10; 3616 children), survival without neurodevelopmental impairment (RR 1.01, 95% CI 0.98 to 1.04; 3845 children) and survival without major neurodevelopmental impairment (RR 1.02, 95% CI 0.98 to 1.05; 1816 children). An increase or decrease in the risk of death since randomisation could not be excluded (RR 1.06, 95% CI 0.81 to 1.40; 5 trials, 4565 babies randomised).
At mid‐childhood follow‐up, moderate‐certainty evidence identified little or no effect of exposure to repeat prenatal corticosteroids compared with no repeat corticosteroids on survival free of neurocognitive impairment (RR 1.01, 95% CI 0.95 to 1.08; 963 children) or survival free of major neurocognitive impairment (RR 1.00, 95% CI 0.97 to 1.04; 2682 children). Benefit or harm could not be excluded for death since randomisation (RR 0.93, 95% CI 0.69 to 1.26; 2874 babies randomised) and any neurocognitive impairment (RR 0.96, 95% CI 0.72 to 1.29; 897 children).
No trials reported data for follow‐up into adolescence or adulthood.
Risk of bias across outcomes was generally low although there were some concerns of bias. For childhood follow‐up, most outcomes had some concerns of risk of bias due to missing data from loss to follow‐up.
Authors' conclusions
The short‐term benefits for babies included less respiratory distress and fewer serious health problems in the first few weeks after birth with repeat dose(s) of prenatal corticosteroids for women still at risk of preterm birth seven days or more after an initial course. The current available evidence reassuringly shows no significant harm for the women or child in early and mid‐childhood, although no benefit.
Further research is needed on the long‐term benefits and risks for the baby into adulthood.
Keywords: Adolescent; Adult; Child; Child, Preschool; Female; Humans; Infant; Infant, Newborn; Pregnancy; Adrenal Cortex Hormones; Adrenal Cortex Hormones/therapeutic use; Odds Ratio; Outcome Assessment, Health Care; Premature Birth; Premature Birth/prevention & control; Vitamins
Plain language summary
Repeat doses of prenatal corticosteroids for women at risk of preterm birth for improving health outcomes in their babies
What is the issue?
Infants born preterm (before 37 weeks' of pregnancy) are at risk of difficulty breathing and lung disease because their lungs are not fully developed. One course of prenatal (administered during pregnancy) corticosteroids, given to women who may give birth early, helps develop the baby's lungs and improves survival. This benefit does not last beyond seven days so those babies born preterm more than seven days after the course of corticosteroid are still at risk of difficulty breathing and lung disease. This review details the evidence available for the benefits and harms of giving a further course(s) of corticosteroids with the aim of extending the benefits on lung development and breathing.
Why is this important?
Preterm birth is common, affecting approximately one in nine babies worldwide. In addition to breathing difficulties after birth, preterm babies who survive the early weeks after birth are at risk of long‐term disabilities such as delays in their development, epilepsy (fits) and cerebral palsy (weakness and problems in the muscles that affects movement and co‐ordination).
Corticosteroid medications are anti‐inflammatory and suppress growth and the production of cortisol (stress hormone) from the adrenal gland. As a result, repeat prenatal corticosteroid treatment could increase the risk of infection and suppress production of cortisol for the mother and her baby, and reduce the baby's growth. It is, therefore, important that both the benefits and potential negative effects of repeat courses of corticosteroids are understood to allow the best treatment decisions to be made in clinical practice.
What evidence did we find?
We searched for evidence on 27 January 2021 and identified 11 randomised controlled trials (clinical studies where people are randomly put into one of two or more treatment groups), involving 4895 women and their 5975 babies who remained at risk of early birth more than seven days after an initial course of corticosteroids between 23 and 34 weeks' gestation at trial enrolment.
Overall, these trials showed that repeat dose(s) of prenatal corticosteroids reduced the risk of the baby having breathing difficulties, including severe difficulties, and serious health problems in the first few weeks after birth (moderate to high quality and low risk of bias evidence). There was probably no effect on chronic lung disease (usually defined by persistent breathing difficulty and need for oxygen treatment at the age they would have reached 36 weeks of pregnancy). The data available could not confirm if there was an increase, decrease or no change in the combination of fetal or newborn or infant death under one year of age, severe bleeding in the brain or severe bowel inflammation.
For the women, there was no increase in the likelihood of a caesarean birth but it was uncertain if there was an increase or decrease in maternal death, maternal infection, risk of maternal side effects or the need to stop treatment due to side effects. No trials reported data for breastfeeding at the time of leaving hospital or risk of the woman being admitted to the intensive care unit.
In five trials that follow the babies up to early childhood, there were no long‐term benefits or harms on later development. Similarly, the two trials that follow children up to mid‐childhood (five years in one trial and six to eight years in another trial) found no long‐term benefits or harms to development. For early and mid‐childhood follow‐up it was unclear if the total deaths after randomisation up to the time of follow‐up were increased, decreased or the same.
The evidence on which these statements were based was generally of moderate or high quality. Most results were based on information with low risk of bias or some concerns of risk of bias.
What does this mean?
This review shows that a repeat dose of prenatal corticosteroids given to women who remain at risk of an early birth after an initial course of prenatal corticosteroids helps the baby's lungs and reduces serious health problems in the first few weeks of life without harm on health or development up to mid‐childhood. Further research is needed on the long‐term benefits or harms for the baby into adulthood.
Summary of findings
Summary of findings 1. Summary of findings table ‐ Repeat dose(s) of prenatal corticosteroid compared to single course for the fetus/neonate/infant.
| Repeat dose(s) of prenatal corticosteroid compared to single course for the fetus/neonate/infant | ||||||
| Patient or population: the fetus/neonate/infant Setting: hospitals in low‐, middle‐ and high‐resource countries Intervention: repeat dose(s) Comparison: single course | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with single course | Risk with repeat dose(s) | |||||
| A1: Fetal or neonatal or infant death (< 1 year of age) – all fetuses randomised | 37 per 1000 | 35 per 1000 (27 to 46) | RR 0.95 (0.73 to 1.24) | 5849 (10 RCTs) | ⊕⊕⊕⊝ Moderatea | For fetal or neonatal or infant death (< 1 year of age), benefit or harm with repeat dose(s) of corticosteroids compared with placebo or standard care could not be excluded. |
| A5: Respiratory distress syndrome – all fetuses randomised | 340 per 1000 | 279 per 1000 (252 to 306) | RR 0.82 (0.74 to 0.90) | 3540 (9 RCTs) | ⊕⊕⊕⊕ High | Repeat dose(s) of prenatal corticosteroid reduces respiratory distress syndrome. |
| A7: Severe lung disease – all fetuses randomised | 130 per 1000 | 108 per 1000 (94 to 126) | RR 0.83 (0.72 to 0.97) | 4955 (6 RCTs) | ⊕⊕⊕⊝ Moderateb | Repeat dose(s) of prenatal corticosteroids probably results in a slight reduction in severe lung disease. |
| A8: Chronic lung disease – all fetuses randomised | 66 per 1000 | 66 per 1000 (54 to 80) | RR 1.00 (0.83 to 1.22) | 5661 (9 RCTs) | ⊕⊕⊕⊕ High | Repeat dose(s) of prenatal corticosteroid results in little to no difference in chronic lung disease. |
| A9: Severe intraventricular haemorrhage (grade 3 or 4) – all fetuses randomised | 11 per 1000 | 13 per 1000 (8 to 21) | RR 1.13 (0.69 to 1.86) | 5066 (7 RCTs) | ⊕⊕⊕⊝ Moderatec | For severe intraventricular haemorrhage (grade 3 or 4), benefit or harm with repeat dose(s) of prenatal corticosteroid could not be excluded. |
| A11: Necrotising enterocolitis – all fetuses randomised | 21 per 1000 | 18 per 1000 (13 to 26) | RR 0.84 (0.59 to 1.22) | 5736 (9 RCTs) | ⊕⊕⊕⊝ Moderated | For necrotising enterocolitis, benefit or harm with repeat dose(s) of prenatal corticosteroid could not be excluded. |
| A12: Composite of serious outcomes– all fetuses randomised | 211 per 1000 | 185 per 1000 (169 to 204) | RR 0.88 (0.80 to 0.97) | 5736 (9 RCTs) | ⊕⊕⊕⊝ Moderatee | Repeat dose(s) of prenatal corticosteroids probably reduces the composite of serious outcomes. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
| See interactive version of this table: https://gdt.gradepro.org/presentations/#/isof/isof_question_revman_web_427554148264309002. | ||||||
a Downgraded one level due to imprecision as the 95% confidence interval included both benefit and harm. b Downgraded one level due to inconsistency as evidenced by significant statistical heterogeneity, likely due to variation in the definition of serious lung disease. c Downgraded one level due to imprecision as the 95% confidence interval included both marked benefit and marked harm. d Downgraded one level due to imprecision as the 95% confidence interval included both marked benefit and harm. e Downgraded one level due to inconsistency as evidenced by significant statistical heterogeneity, likely due to variation in the definition of the composite serious outcome.
Summary of findings 2. Summary of findings table ‐ Repeat dose(s) of prenatal corticosteroids compared to single course for the woman.
| Repeat dose(s) of prenatal corticosteroids compared to single course for the woman | ||||||
| Patient or population: the woman Setting: hospitals in low‐, middle‐ and high‐resource countries Intervention: repeat dose(s) Comparison: single course | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with single course | Risk with repeat dose(s) | |||||
| B1: Maternal death – all women | 5 per 1000 | 1 per 1000 (0 to 36) | RR 0.32 (0.01 to 7.81) | 437 (1 RCT) | ⊕⊕⊝⊝ Lowa | For maternal death, benefit or harm with repeat dose(s) of prenatal corticosteroids compared with placebo/standard care could not be excluded. |
| B2: Maternal sepsis – an all women | 67 per 1000 | 76 per 1000 (62 to 93) | RR 1.13 (0.93 to 1.39) | 4666 (8 RCTs) | ⊕⊕⊕⊝ Moderateb | For maternal sepsis, benefit or harm with repeat dose(s) of prenatal corticosteroids compared with placebo/standard care could not be excluded. |
| B3: Caesarean section – an all women | 559 per 1000 | 576 per 1000 (548 to 609) | RR 1.03 (0.98 to 1.09) | 4266 (8 RCTs) | ⊕⊕⊕⊝ Moderatec | Repeat doses of prenatal corticosteroids likely results in little to no difference in the risk of caesarean section. |
| B4: Discontinuation of therapy due to maternal adverse effects – an all women | Not pooled | Not pooled | Not pooled | 485 (1 RCT) | ⊕⊕⊝⊝ Lowd | It was unclear if repeat dose prenatal corticosteroid had an effect on discontinuation of therapy due to maternal adverse effects as only 1 trial reported this outcome and the outcome occurred in 0 women. |
| Adverse effects of corticosteroids | Not pooled | Not pooled | Not pooled | 1477 (2 RCTs) | ⊕⊕⊝⊝ Lowe | It was unclear if repeat dose(s) of prenatal corticosteroids had an effect on the risk of adverse effects. Meta‐analysis was not performed as it was not considered appropriate to combine the results due to a marked differences in event rates and direction of effect. |
| Admission to the intensive care unit ‐ not reported | ‐ | ‐ | ‐ | ‐ | ‐ | No trials reported data for this outcome. |
| Breastfeeding at hospital discharge ‐ not reported | ‐ | ‐ | ‐ | ‐ | ‐ | No trials reported data for this outcome. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
| See interactive version of this table: https://gdt.gradepro.org/presentations/#/isof/isof_question_revman_web_427567765501157336. | ||||||
a Downgraded two levels due to imprecision given there was only one event and a wide 95% confidence interval included both significant benefit and significant harm. b Downgraded one level for imprecision as the 95% confidence interval included possible benefit and possible harm. c Downgraded one level for inconsistency as evidenced by statistical heterogeneity. d Downgraded two levels due to imprecision as there were no events recorded making it impossible to provide a relative effect estimate. e Downgraded two levels for marked inconsistency of effect as evidenced by very serious heterogeneity.
Summary of findings 3. Summary of findings table ‐ Repeat dose(s) of prenatal corticosteroids compared to single course for the child aged 2 to < 5 years.
| Repeat dose(s) of prenatal corticosteroids compared to single course for the child aged 2 to < 5 years | ||||||
| Patient or population: the child Setting: outpatient settings in high‐resource countries Intervention: repeat dose(s) Comparison: single course | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with single course | Risk with repeat dose(s) | |||||
| C1: Total deaths (after randomisation) up to early childhood follow‐up ‐ In all children follow‐up: range 2 years to 3 years | 42 per 1000 | 44 per 1000 (34 to 58) | RR 1.06 (0.81 to 1.40) | 4565 (5 RCTs) | ⊕⊕⊕⊝ Moderatea | For the risk of death after randomisation up to early childhood follow‐up, benefit or harm could not be excluded for repeat dose(s) of corticosteroids. |
| C2: Neurodevelopmental impairment at early childhood follow‐up ‐ In all children follow‐up: range 2 years to 3 years | 187 per 1000 | 181 per 1000 (159 to 205) | RR 0.97 (0.85 to 1.10) | 3616 (4 RCTs) | ⊕⊕⊕⊕ High | Repeat dose(s) of prenatal corticosteroids results in little to no difference in neurodevelopmental impairment at early childhood follow‐up. |
| C3: Survival free of neurodevelopmental impairment at early childhood follow‐up ‐ In all children follow‐up: range 2 years to 3 years | 777 per 1000 | 785 per 1000 (761 to 808) | RR 1.01 (0.98 to 1.04) | 3845 (4 RCTs) | ⊕⊕⊕⊕ High | Repeat dose(s) of prenatal corticosteroids results in little to no difference in survival free of neurodevelopmental impairment at early childhood follow‐up. |
| C4: Survival free of major neurodevelopmental impairment atearly childhood follow‐up ‐ In all children follow‐up: range 2 years to 3 years | 856 per 1000 | 873 per 1000 (839 to 899) | RR 1.02 (0.98 to 1.05) | 1816 (3 RCTs) | ⊕⊕⊕⊝ Moderateb | Repeat dose(s) of prenatal corticosteroids probably results in little to no difference in survival free of major neurodevelopmental impairment at early childhood follow‐up. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
| See interactive version of this table: https://gdt.gradepro.org/presentations/#/isof/isof_question_revman_web_427569318660056467. | ||||||
a Downgraded one level due to imprecision as the 95% confidence interval included possible benefit and harm. b Downgraded one level due to inconsistency as evidenced by marked statistical heterogeneity.
Summary of findings 4. Summary of findings table ‐ Repeat dose(s) of prenatal corticosteroids compared to single course for the child in mid‐ to late childhood (5 to < 18 years of age).
| Repeat dose(s) of prenatal corticosteroids compared to single course for the child in mid‐ to late childhood (5 to < 18 years of age) | ||||||
| Patient or population: the child Setting: outpatient settings in high‐resource countries Intervention: repeat dose(s) Comparison: single course | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with single course | Risk with repeat dose(s) | |||||
| D1: Total deaths (after randomisation) up to mid‐ to later childhood follow‐up ‐ In all children follow‐up: range 5 years to 8 years | 57 per 1000 | 53 per 1000 (39 to 72) | RR 0.93 (0.69 to 1.26) | 2874 (2 RCTs) | ⊕⊕⊕⊝ Moderatea | For total deaths after randomisation up to mid‐childhood follow‐up, benefit or harm could not be excluded for repeat dose(s) of corticosteroids. |
| D2: Neurocognitive impairment atmid‐ to later childhood follow‐up ‐ In all children follow‐up: range 5 years to 8 years | 167 per 1000 | 160 per 1000 (120 to 215) | RR 0.96 (0.72 to 1.29) | 897 (1 RCT) | ⊕⊕⊝⊝ Lowb,c | For neurocognitive impairment at mid‐childhood follow‐up, benefit or harm could not be excluded for repeat dose(s) of corticosteroids. |
| D3: Survival free of neurocognitive impairment atmid‐ to later childhood follow‐up ‐ In all children follow‐up: range 5 years to 8 years | 773 per 1000 | 780 per 1000 (734 to 835) | RR 1.01 (0.95 to 1.08) | 963 (1 RCT) | ⊕⊕⊕⊝ Moderated | Repeat dose(s) of prenatal corticosteroids likely results in little to no difference in survival free of neurocognitive impairment at mid‐childhood follow‐up. |
| D4: Survival free of major neurocognitive impairment atmid‐ to later childhood follow‐up ‐ In all children follow‐up: range 5 years to 8 years | 807 per 1000 | 807 per 1000 (783 to 839) | RR 1.00 (0.97 to 1.04) | 2682 (2 RCTs) | ⊕⊕⊕⊝ Moderateb | Repeat dose(s) of prenatal corticosteroids likely results in little to no difference in survival free of major neurocognitive impairment at mid‐childhood follow‐up. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
| See interactive version of this table: https://gdt.gradepro.org/presentations/#/isof/isof_question_revman_web_427569595635902654. | ||||||
a Downgraded one level for imprecision due to a wide 95% confidence interval that included benefit and possible harm. b Downgraded one level due to risk of bias as the included trials were judged to have some concerns of risk of bias due to missing outcome data. c Downgraded one level for imprecision as the 95% confidence interval included possible benefit and harm. d Downgraded one level due to risk of bias as the single included trial was judged to have some concerns of risk of bias due to missing outcome data.
Background
Description of the condition
Infants born preterm (before 37 weeks' gestation) are at high risk of neonatal lung disease and its sequelae. Worldwide in 2010, an estimated 11.1% of all live births were preterm (Blencowe 2013). The more preterm the baby, the greater are the risks, especially when birth occurs before 32 weeks' gestation. In Australia, in 2018, 1.6% of all births were before 32 weeks' gestation (AIHW 2020). Respiratory distress syndrome (RDS), as a consequence of immature lung development, is the principal cause of early neonatal mortality and morbidity and contributes significantly to the high costs of neonatal intensive care. Preterm babies who survive the early weeks after birth are at risk of long‐term neurological disability (Cheong 2017). Parents are understandably worried and distressed when their baby is born preterm. Strategies to reduce the risk of neonatal respiratory disease for infants who are born preterm have received considerable attention (McGoldrick 2020; Rojas‐Reyes 2012).
A single course of prenatal corticosteroids reduces the risk of RDS from 14.8% to 10.5% (risk ratio (RR) 0.71, 95% confidence interval (CI) 0.65 to 0.78; 26 trials, 11,183 infants) (McGoldrick 2020). Other beneficial effects include a reduced risk of neonatal death, intraventricular haemorrhage, necrotising enterocolitis and childhood developmental delay (McGoldrick 2020). Prenatal corticosteroids enhance the benefits of postnatal surfactant therapy (Jobe 1994), and reduce the need for blood pressure support (Moise 1995). Overall, there is a reduction in the cost and duration of neonatal care. The cost‐benefit of a single course of prenatal corticosteroids is estimated as USD 3000 (NIH 1995). Long‐term follow‐up into adulthood of infants exposed to prenatal corticosteroids in the first (New Zealand) trial (Liggins 1972), have shown no adverse clinical outcomes (Dalziel 2005a; Dalziel 2005b). However, even though prenatal corticosteroids remain the most effective known strategy for reducing the adverse consequences of preterm birth, and despite postnatal intensive care and exogenous surfactant, there is still significant neonatal morbidity (Rojas‐Reyes 2012).
Description of the intervention
Prenatal corticosteroid treatment compared with no prenatal corticosteroid treatment has been shown to be ineffective if babies are born more than seven days after the corticosteroid treatment has been given (Roberts 2006). Specifically, there is no reduction in the incidence of RDS or neonatal mortality (McLaughlin 2003; Roberts 2006). There may be benefit in repeating the dose of prenatal corticosteroids to women who remain at risk of preterm birth more than seven days after the initial course. This was suggested by Liggins and Howie in the first reported controlled trial of prenatal glucocorticoid treatment for the prevention of RDS in premature infants (Liggins 1972). Indeed, in some clinical centres this has become standard practice and has been incorporated into clinical guidelines (Antenatal Corticosteroids CPG Panel 2015).
How the intervention might work
Animal studies have suggested that repeat treatment with prenatal corticosteroids may be more effective than a single course in reducing the risk of RDS. In sheep fetuses, there is a dose‐dependent improvement in lung function with repeat doses of betamethasone (Ikegami 1997). In human infants, improved cardiovascular responses to preterm birth have been observed (Padbury 1996).
Why it is important to do this review
The potential benefits of repeat prenatal corticosteroid treatment on neonatal lung function and cardiovascular health may be balanced by increased maternal risks such as infection and suppression of hypothalamic‐pituitary‐adrenal (HPA) function (Ashwood 2006; McKenna 2000). In addition, experimental reports raise concerns about the use of repeat doses of prenatal corticosteroids because of potential adverse effects for the offspring.
It is well known that corticosteroids inhibit cell growth and DNA replication. Studies in both small and large animals demonstrate that exogenous steroids inhibit fetal growth and increase fetal blood pressure (Fowden 1996; Jensen 2002). In sheep, there is a dose‐dependent reduction in birthweight in lambs exposed to up to four doses of betamethasone administered to the ewe (Ikegami 1997), although exogenous steroids administered directly to the fetus do not inhibit fetal growth (Newnham 1999).
Other animal studies have shown that repeat doses of corticosteroids may have harmful effects on neuronal myelination, the development of insulation for nerve fibres, affecting nerve conduction (Dunlop 1997); the development of the alveolar septa (the sites of gas exchange in the lung) leaving 'emphysematous'‐like alveoli (Tschanz 1995) and HPA axis function (Ikegami 1997). Effects on the HPA axis can persist into adulthood.
In humans, similar concerns have been raised from non‐randomised cohort studies, with adverse effects after repeat doses of corticosteroids on measures of growth at birth (French 1999), risk of neonatal infection, fetal pituitary‐adrenal axis function, neonatal blood pressure (Mildenhall 2006), childhood behaviour (French 1998), and high levels of stress in parents (French 1998). Long‐term developmental follow‐up studies of infants exposed to repeat doses of prenatal corticosteroids are limited to date and have produced conflicting results. Some non‐randomised studies suggest delayed development (Esplin 2000) and adverse effects on childhood behaviour (French 1998), while other non‐randomised studies have shown no difference between exposed and non‐exposed children (French 1999; Hasbargen 2001; Thorp 2002), or possible reduced cerebral palsy (French 2004). Another long‐term potential adverse outcome that requires further investigation is the possibility that single or repeat doses of prenatal corticosteroids could programme cardiovascular settings in the fetus and lead to adult hypertension (Benediktsson 1993), and insulin resistance (Dalziel 2005a). Increased exposure of the fetus to glucocorticoids has been proposed as a possible mechanism underlying the epidemiological association between small size at birth and adult cardiovascular and metabolic disease (Seckl 2004).
There remains uncertainty, therefore, about whether there is overall benefit in repeating the dose of prenatal corticosteroids for women who remain at risk of preterm birth after an initial course. This review will assess the benefits and harms of repeat doses of prenatal corticosteroids for women at risk of preterm birth seven or more days after an initial course.
This is an update of a review first published in 2000 and updated in 2007, 2011 and 2015.
Objectives
To assess the effectiveness and safety, using the best available evidence, of a repeat dose(s) of prenatal corticosteroids, given to women who remain at risk of preterm birth seven or more days after an initial course of prenatal corticosteroids with the primary aim of reducing fetal and neonatal morbidity and mortality.
Methods
Criteria for considering studies for this review
Types of studies
All published, unpublished and ongoing randomised trials with reported data that compared outcomes for women at risk of preterm birth randomised to receive a repeat dose(s) of prenatal corticosteroids with outcomes in controls given a single course of prenatal corticosteroids, with or without additional placebo administration. Trials must have used some form of random allocation. Quasi‐randomised trials and cross‐over trials were not eligible for inclusion. Cluster‐randomised trials were eligible for inclusion. Trials published as abstracts were included if they met the criteria discussed in 'Selection of studies: Abstracts'.
Types of participants
Women considered at risk of preterm birth who had already received a single course of prenatal corticosteroid seven or more days previously.
Types of interventions
Corticosteroid administered to the women intravenously, intramuscularly or orally, compared with either placebo or no placebo. Both groups must have received their initial course of corticosteroids seven or more days earlier. We excluded trials in which the fetus received corticosteroids directly as these are included in another Cochrane Review ("Transplacental versus direct fetal corticosteroid treatment for accelerating fetal lung maturation where there is a risk of preterm birth"; Utama 2018).
Types of outcome measures
We prespecified outcomes for the review after each outcome was independently ranked for clinical importance by each review author using the scale described in Chapter 3 of the GRADE Handbook (gdt.gradepro.org/app/handbook/handbook.html). This scale uses scores from 1 to 9 to divide outcomes into those of critical importance, important but not critical and of limited importance to clinical decision‐making. Outcomes of critical importance to clinical decision‐making were included as primary outcomes for the review. Those outcomes classified as important but not critical were included as secondary outcomes.
Primary outcomes
We chose primary outcomes to be most representative of the clinically important measures of effectiveness and safety, including serious outcomes, for the women and their fetuses/infants, the infant in early childhood (age two to less than five years), the infant in mid‐ to late childhood (age five to less than 18 years) and the infant as an adult. These outcomes were changed from the previous version of the review after first identifying outcomes considered of clinical importance by the review authors, refining their definitions by discussion among the review authors, and ranking their importance using the criteria described in the GRADE handbook in Types of outcome measures. The changes to outcome descriptions are summarised in the Differences between protocol and review section.
For the fetus/neonate/infant
Fetal or neonatal or infant death (less than one year of age).
Fetal death.
Neonatal death.
Infant death (less than one year of age).
Respiratory distress syndrome (however defined by authors).
Severe respiratory distress syndrome (however defined by authors).
Severe lung disease (however defined by authors).
Chronic lung disease (however defined by authors).
Severe intraventricular haemorrhage (grade 3 or 4).
Intraventricular haemorrhage (any grade).
Necrotising enterocolitis (however defined by authors).
Composite serious outcome (however defined by authors).
For the woman
Maternal death.
Maternal sepsis (any of chorioamnionitis during labour, endometritis, pyrexia after trial entry requiring the use of antibiotics, puerperal sepsis, intrapartum fever requiring the use of antibiotics, postnatal pyrexia or however defined by authors).
Caesarean section.
Discontinuation of therapy because of maternal side effects.
Adverse effects of corticosteroids (including gastrointestinal upset, insomnia, local injection site adverse effects (pain, bruising, haematoma or infection at the injection site)).
Admission to the intensive care unit.
Breastfeeding at hospital discharge.
For the child in early childhood (aged two to less than five years)
Total deaths (after randomisation).
Neurodevelopmental impairment at age two to less than five years (any of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one standard deviation (SD) below the mean), motor impairment (mild or major impairment by appropriate mode of assessment), or cerebral palsy, or however defined by authors).
Survival free of neurodevelopmental impairment at age two to less than five years (none of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), motor impairment (mild or major impairment by appropriate mode of assessment), cerebral palsy or however defined by authors).
Survival free of major neurodevelopmental impairment at age two to less than five years (none of: moderate or severe intellectual impairment (developmental quotient or intelligence quotient more than two SDs below the mean), moderate or severe cerebral palsy, major motor impairment, blindness (corrected visual acuity worse than 6/60 in the better eye) or deafness (hearing loss requiring amplification or worse), or however defined by authors).
Cerebral palsy (categorised as nil, mild, moderate or severe, however defined by authors).
Developmental delay or intellectual impairment at age two to less than five years (categorised as mild (one SD below the mean), moderate (two SDs below the mean) or severe (three SDs below the mean) by an appropriate rating scale, or however defined by authors).
For the child in mid‐ to late childhood (aged five to less than 18 years)
Total deaths (after randomisation).
Neurocognitive impairment at age five to less than 18 years (any of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), executive dysfunction, learning difficulties, motor impairment (mild or major impairment by appropriate mode of assessment), or cerebral palsy, or however defined by authors).
Survival free of neurocognitive impairment at age five to less than 18 (none of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), executive dysfunction, learning difficulties, motor impairment (mild or major impairment by appropriate mode of assessment) cerebral palsy, or however defined by authors).
Survival free of major neurocognitive impairment at age five to less than 18 years (none of: moderate or severe intellectual impairment (developmental quotient or intelligence quotient more than two SD below the mean), major motor impairment, moderate or severe cerebral palsy, blindness (corrected visual acuity worse than 6/60 in the better eye) or deafness (hearing loss requiring amplification or worse), or however defined by authors).
Motor impairment at age five to less than 18 years (categorised as nil, mild impairment, major impairment by appropriate mode of assessment or however defined by authors).
Cognitive impairment at age five to less than 18 years (categorised as mild (one SD below the mean), moderate (two SDs below the mean) or severe (three SDs below the mean) by an appropriate rating scale, or however defined by authors).
Educational achievement (however defined by authors).
Cerebral palsy (categorised as nil, mild, moderate or severe by an appropriate rating scale or however defined by authors).
Hypertension (however defined by authors).
For the child as an adult (aged 18 years or greater)
Total deaths (after randomisation).
Neurodevelopmental disability at age 18 years or greater (any of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), cerebral palsy, or however defined by authors).
Cardiovascular disease (including diagnosis of ischaemic heart disease, cerebrovascular disease, heart failure or however defined by authors).
Cardiovascular death (ischaemic heart disease, stroke, arrhythmia or heart failure as cause of death or however defined by authors).
Type 2 diabetes mellitus (however defined by authors).
Glucose intolerance (however defined by authors).
Obesity/overweight (however defined by authors).
Hypertension (however defined by authors).
Secondary outcomes
These include other measures of effectiveness, complications, satisfaction with care and health service use.
For the fetus/neonate/infant
Birthweight (including weight for gestation if reported).
Small‐for‐gestational age (however defined by authors).
Length at birth (including length for gestation if reported).
Head circumference at birth (including head circumference for gestation if reported).
Growth assessments at primary hospital discharge (weight, head circumference and length including measurements standardised for postmenstrual age if reported).
Growth assessments at infant follow‐up (weight, head circumference, length, including measurements standardised for postmenstrual age if reported).
Gestational age at birth (term birth 37 weeks or greater, preterm birth less than 37 weeks, very preterm birth less than 34 weeks, extremely preterm birth less than 28 weeks and mean gestational age).
Interval between trial entry and birth.
Admission to the neonatal intensive care unit (NICU).
Proven neonatal infection while in the NICU.
Early systemic neonatal infection (less than 48 hours after birth or however defined by authors).
Late systemic neonatal infection (48 hours or greater after birth or however defined by authors).
Retinopathy of prematurity (however defined by authors).
Periventricular leukomalacia (however defined by authors).
Neonatal encephalopathy (however defined by authors).
Patent ductus arteriosus.
Use of respiratory support (any respiratory support including invasive (via an endotracheal tube) or non‐invasive modalities or however defined by authors).
Duration of respiratory support (any respiratory support, including any invasive (via an endotracheal tube) and non‐invasive modalities or however defined by authors).
Use of invasive respiratory support (any respiratory support via an endotracheal tube or however defined by authors).
Duration of invasive respiratory support (any respiratory support via an endotracheal tube or however defined by authors).
Use of non‐invasive respiratory support (any form of respiratory support that does not require an endotracheal tube including any non‐invasive ventilation, continuous positive airway pressure (CPAP), high or low flow gases or however defined by authors).
Duration of non‐invasive respiratory support (any form of respiratory support that does not require an endotracheal tube including any non‐invasive ventilation, CPAP and high or low flow gases or however defined by authors).
Use of oxygen supplementation.
Duration of oxygen supplementation.
Use of surfactant.
Use of postnatal corticosteroids.
Use of nitric oxide for respiratory support.
Pulmonary hypertension (however defined by authors).
Use of inotropic support.
Duration of inotropic support.
Air leak syndrome.
Measures of renal function (however reported by authors).
Feed tolerance (time to full feed tolerance, number of feed interruptions or however defined by authors).
Apgar score less than 7 at five minutes.
Cardiac hypertrophy (however defined by authors).
Measures of HPA function (however assessed by authors).
For the woman
Puerperal sepsis (aged 18 years or greater).
Chorioamnionitis during labour (however defined by authors).
Endometritis (however defined by authors).
Pyrexia after trial entry requiring the use of antibiotics.
Intrapartum fever requiring the use of antibiotics.
Postpartum haemorrhage.
Postnatal pyrexia (however defined by authors).
Preterm prelabour rupture of the membranes after trial entry.
Mode of birth.
Hypertension (however defined by authors).
Pulmonary oedema.
Glucose intolerance (however defined by authors).
Postnatal depression (however defined by authors).
Local injection site adverse effects (pain, bruising, haematoma or infection at the injection site).
Insomnia after treatment (however defined by authors).
Gastrointestinal adverse effects of treatment (however defined by authors).
Satisfaction with the therapy.
Quality of life.
For the child aged two to less than five years
Child behaviour (measured by an appropriate mode of behavioural assessment or however defined by authors).
Motor impairment at age two to less than five years (categorised as nil, mild impairment, major impairment by appropriate mode of assessment or however defined by authors).
Deafness/hearing impairment (however defined by authors).
Blindness/visual impairment (however defined by authors).
Growth assessments (weight, head circumference, height and assessments for age if available).
Body mass index (BMI).
Obesity/overweight.
Blood pressure (systolic, diastolic, mean arterial).
Hypertension (however defined by authors).
Measures of lung function (however defined by authors).
Chronic lung disease of infancy (however defined by authors).
Asthma or recurrent wheeze (however defined by authors).
Any respiratory disease (however defined by authors).
Measures of insulin and glucose homeostasis (however defined by authors).
Measures of lipid profile (however defined by authors).
Measures of HPA axis function (however assessed by authors).
Body composition (lean body mass corrected for height, fat mass corrected for height or however defined by authors).
Bone density (however assessed by authors).
For the child aged five to less than 18 years
Child behaviour (however defined by authors).
Deafness/hearing impairment (however defined by authors).
Blindness/visual impairment (however defined by authors).
Growth assessments (weight, head circumference, height and assessments for age if available).
BMI.
Obesity/overweight.
Blood pressure (systolic, diastolic, mean arterial).
Measures of lung function (however defined by authors).
Asthma or recurrent wheeze (however defined by authors).
Respiratory disease (however defined by authors).
Measure of insulin and glucose homeostasis (however defined by authors).
Lipid profile (however reported by authors).
Measures of HPA axis function (however defined by authors).
Bone density (however assessed by authors).
Body composition (lean body mass (fat free mass) for height, fat mass for height or however defined by authors).
For the child as an adult (aged 18 years or greater)
Educational achievement (however defined by authors).
Prediabetes (however defined by authors).
Mental health disorders (however defined by authors).
Diagnosis of depression (however defined by authors).
Diagnosis of bipolar affective disorder (however defined by authors).
Diagnosis of anxiety disorder (however defined by authors).
Ischaemic heart disease (however defined by authors).
Stroke (however defined by authors).
Heart failure.
Blindness/visual impairment (however defined by authors).
Deafness/hearing impairment (however defined by authors).
Measures of insulin and glucose homeostasis (however defined by authors).
Use of health services
Length of prenatal hospitalisation for the woman.
Length of postnatal hospitalisation for the woman.
Maternal admission to the intensive care unit.
Admission to and length of stay in NICU.
Length of infant hospitalisation.
Costs of maternal care.
Costs of neonatal care.
Hospital re‐admission at childhood follow‐up.
While we sought all the above outcomes from the included trials, only those with data appear in the analysis tables. We included outcomes in the analyses if data were available for analysis according to original allocation.
Search methods for identification of studies
The methods section of this review is based on a standard template used by the Cochrane Pregnancy and Childbirth Group.
Electronic searches
For this update, we searched Cochrane Pregnancy and Childbirth's Trials Register by contacting their Information Specialist (27 January 2021).
The Register is a database containing over 27,000 reports of controlled trials in the field of pregnancy and childbirth. It represents over 30 years of searching. For full current search methods used to populate Pregnancy and Childbirth's Trials Register including the detailed search strategies for the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase and CINAHL; the list of handsearched journals and conference proceedings, and the list of journals reviewed via the current awareness service, see pregnancy.cochrane.org/pregnancy-and-childbirth-groups-trials-register.
Briefly, Cochrane Pregnancy and Childbirth's Trials Register is maintained by their Information Specialist and contains trials identified from:
monthly searches of CENTRAL;
weekly searches of MEDLINE (Ovid);
weekly searches of Embase (Ovid);
monthly searches of CINAHL (EBSCO);
handsearches of 30 journals and the proceedings of major conferences;
weekly current awareness alerts for a further 44 journals plus monthly BioMed Central email alerts.
Search results are screened by two people and the full text of all relevant trial reports identified through the searching activities described above is reviewed. Based on the intervention described, each trial report is assigned a number that corresponds to a specific Pregnancy and Childbirth review topic (or topics), and is then added to the Register. The Information Specialist searches the Register for each review using this topic number rather than keywords. This results in a more specific search set that has been fully accounted for in the relevant review sections (Included studies; Excluded studies; Studies awaiting classification; Ongoing studies).
In addition, we searched ClinicalTrials.gov and the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP; trialsearch.who.int/) (27 January 2021) for unpublished, planned and ongoing trial reports using the search methods detailed in Appendix 1.
Searching other resources
We searched reference lists of trials and other review articles. We attempted to contact authors of two studies listed under 'ongoing studies' in the previous version of the review and two newly identified trials that are ongoing for further information for this update.
We applied no language restrictions.
Data collection and analysis
For methods used in the previous version of this review, see Crowther 2015.
For this update, we used the following methods for assessing the reports that were identified as a result of the updated search.
Selection of studies
We independently evaluated trials under consideration for inclusion without consideration of their results. We resolved any differences of opinion by discussion. There was no blinding of authorship.
Two review authors assessed the trials for eligibility and trustworthiness. Trials were not assessed blind, as we knew the author's names, institution and the source of publication. We resolved any disagreement by discussion until we reached consensus.
Screening eligible studies for scientific integrity/trustworthiness
All studies meeting our inclusion criteria were also evaluated by two review authors against predefined criteria to select studies that, based on available information, were deemed sufficiently trustworthy to be included in the analysis. Studies were considered at high risk for untrustworthiness if one or more of the following criteria were met.
Research governance
Are there any retraction notices or expressions of concern listed on the Retraction Watch Database relating to this study (retractiondatabase.org/RetractionSearch.aspx?)?
No prospective trial registration for studies published after 2010 without plausible explanation.
When requested, trial authors refused to provide/share the protocol or ethics approval letter, or both.
Trial authors refused to engage in communication with the Cochrane editorial group.
Trial authors refused to provide individual participant data upon request with no justifiable reason.
Baseline characteristics
Characteristics of the study participants being too similar (distribution of mean (SD) excessively narrow or excessively wide, as noted by Carlisle 2017).
Feasibility
Implausible numbers (e.g. 500 women with severe cholestasis of pregnancy recruited in 12 months).
(Close to) zero losses to follow‐up without plausible explanation.
Results
Implausible results (e.g. massive risk reduction for main outcomes with small sample size).
Concerns about the methods of randomisation such as unexpectedly even numbers of women 'randomised' including a mismatch between the numbers and the methods (e.g. if the authors say 'no blocking was used' but still have equal numbers, or if the authors say they used 'blocks of four' but the final numbers differ by six).
Studies assessed as being potentially 'high risk' were not included in the review. Where a study was classified as 'high risk' for one or more of the above criteria we attempted to contact the study authors to address any possible lack of information/concerns. If adequate information remained unavailable, the study remained in 'awaiting classification' and the reasons and communications with the author (or lack of) described in detail.
The process is described in full in Figure 1.
1.
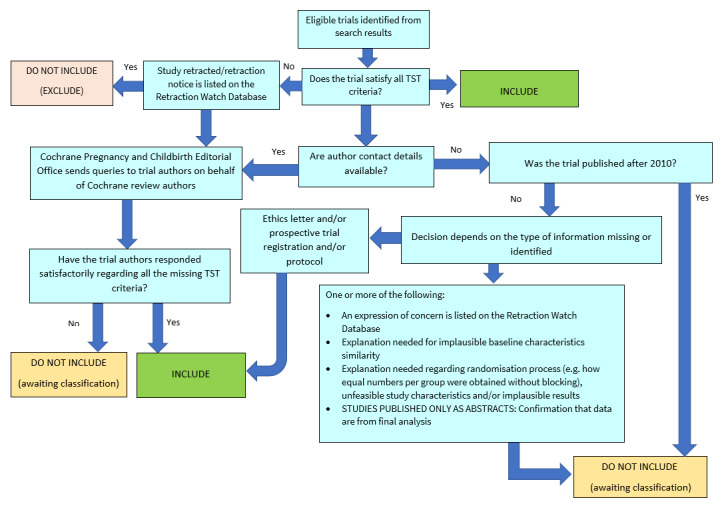
Applying the Cochrane Pregnancy and Childbirth Trustworthiness Screening Tool.
Abstracts
Data from abstracts were only included if, in addition to the trustworthiness assessment, the study authors confirmed in writing that the data to be included in the review had come from the final analysis and will not change. If such information was not available/provided, the study remained as 'awaiting classification' (as above).
Data extraction and management
Two review authors independently extracted study data, using a predesigned data form. Two review authors (AW and PM) independently extracted data for the ACTORDS (Australasian Collaborative Trial of Repeat Doses of Steroids) trial (Crowther 2006). We resolved discrepancies through discussion. When information was unclear, we attempted to contact authors of the original reports to request further details. We entered data into Review Manager software (RevMan Web 2021) and checked them for accuracy.
Assessment of risk of bias in included studies
Two review authors independently assessed risk of bias for each outcome for each study using the RoB 2 tool (accessed 30 June 2021, available from: sites.google.com/site/riskofbiastool/welcome/rob-2-0-tool), as detailed in Chapter 8 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2019). All primary and secondary outcomes were assessed (by outcome measure and time point of assessment where relevant) for risk of bias using the ROB 2 tool. Three review authors (CC, JH and CM) had authored publications for Crowther 2006 or its follow‐up studies and, therefore, they were not involved in risk of bias assessment related to this trial and its outcomes. Two review authors (AW and PM) assessed risk of bias for Crowther 2006 and its follow‐up studies. We resolved disagreements by discussion. The effect of interest was the effect of assignment to the intervention (intention‐to‐treat effect). The risk of bias assessments were recorded alongside forest plots for meta‐analyses. We used the RoB 2 Excel tool for managing assessments of risk of bias.
The assessments involved assessing five domains in which risk of bias may arise.
Bias arising from the randomisation process.
Bias due to deviations from intended interventions.
Bias due to missing outcome data.
Bias in measurement of the outcome.
Bias in selection of the reported result.
For each domain, signalling questions follow decision algorithms to arrive at a judgement of the risk of bias as described in the ROB 2 detailed guidance (Higgins 2019). For each signalling question the possible responses are 'yes', 'probably yes', 'probably no', 'no' or 'no information'. Supporting information was provided for answers to signalling questions and judgements of risk of bias. Risk of bias in each domain was determined to be low risk of bias, high risk of bias or some concerns. Based on the bias in each domain, an overall risk of bias was formulated using the same categories of low risk of bias, high risk of bias or some concerns. If any domain had a high risk of bias, then the outcome was considered to have a high risk of bias. If the risk of bias was assessed as some concerns in multiple domains of relevance to the outcome then the overall risk of bias was considered high.
When assessing risk of bias for outcomes from cluster‐randomised trials using the RoB 2 tool we included an additional domain 1b: bias arising from the timing of identification and recruitment of participants, as described in the ROB 2 guidance on additional considerations for cluster‐randomised trials (Eldridge 2021).
Measures of treatment effect
Dichotomous data
For dichotomous data, we presented results as summary risk ratios (RRs) with 95% confidence intervals (CI). We calculated number needed to treat for an additional beneficial outcome (NNTB) or harmful outcome (NNTH) from the summary risk differences and their 95% confidence limits (Higgins 2021).
Continuous data
For continuous data, we used the mean difference (MD) with 95% CIs. If median and interquartile range were reported, we estimated mean values and SDs to calculate MDs (Wan 2014).
Where studies used different instruments, we used the standardised mean difference (SMD) with 95% CI with the following interpretations:
SMD 0.8 or greater = large effect;
SMD greater than 0.49 and less than 0.8 = medium effect;
SMD greater than 0.19 and less than 0.5 = small effect;
SMD less than 0.2 = trivial or no effect.
Unit of analysis issues
Unit of analysis
The unit of analysis for maternal outcomes was the individual woman. To account for multiple pregnancies, the unit of analysis for outcomes in infants or children was the fetus alive at randomisation. In accordance with the Pregnancy and Childbirth Cochrane Group guidelines, for neonatal outcomes the number of fetuses alive at the point of randomisation was used as the denominator for analysis. This avoids bias by ensuring analysis is based on the total group of women and their fetuses initially randomised but may bias the analysis by underestimating the occurrence of neonatal morbidity, as the most unwell fetuses may die in utero or soon after birth and contribute to the denominator but not to measures of neonatal morbidity.
Cluster‐randomised trials
Cluster‐randomised trials were eligible for inclusion, but we did not identify any. Should we encounter cluster‐randomised trials subsequently, we will include them in the analyses along with individually randomised trials. We will adjust their sample sizes using the methods described in the Cochrane Handbook for Systematic Reviews of Interventions using an estimate of the intracluster correlation coefficient (ICC) derived from the trial (if possible), or from another source (Chapter 23.1.4; Higgins 2021). If ICCs from other sources are used, we will report this and conduct sensitivity analyses to investigate the effect of variation in the ICC. If we identify both cluster‐randomised trials and individually randomised trials, we plan to synthesise the relevant information. We consider it reasonable to combine the results from both if there is little heterogeneity between the study designs and an interaction between the effect of intervention and the choice of randomisation unit is considered unlikely. We will also acknowledge heterogeneity in the randomisation unit and perform a separate meta‐analysis.
When assessing risk of bias for outcomes from cluster‐randomised trials using the RoB 2 tool we will include an additional Domain 1b: bias arising from the timing of identification and recruitment of participants, as described in the ROB 2 guidance on additional considerations for cluster‐randomised trials (Eldridge 2021).
Cross‐over trials
Cross‐over trials were not eligible for inclusion.
Dealing with missing data
For included studies, we noted levels of attrition. Bias associated with missing outcome data was assessed using the Risk of Bias 2 Tool in Domain 3: bias due to missing outcome data.
For all outcomes, we carried out analyses as far as possible on an intention‐to‐treat basis. We attempted to include all participants randomised to each group in the analyses, and analysed all participants in the group to which they were allocated, regardless of whether or not they received the allocated intervention. The denominator for infant outcomes in each trial was taken as the number of fetuses randomised. For maternal outcomes and those at early and mid‐childhood follow‐up, the denominator was participants randomised, minus any participants whose outcomes were known to be missing ('available‐case' analysis).
Assessment of heterogeneity
We assessed statistical heterogeneity in each meta‐analysis using the I² and Chi² statistics. We regarded heterogeneity as substantial if the I² statistic was greater than 30% or there was a low P value (less than 0.10) in the Chi² test for heterogeneity. When we identified high levels of heterogeneity among the trials, we explored this in Sensitivity analysis.
Assessment of reporting biases
Where we identified high risk or some concerns for reporting bias (assessed in Domain 5: bias in selection of the reported result) we explored the impact of including such studies in the overall assessment of results by a sensitivity analysis.
Where there were 10 or more studies in the meta‐analysis, we investigated reporting biases (such as publication bias) using funnel plots. We assessed funnel plot asymmetry visually.
Data synthesis
We carried out statistical analysis using Review Manager software (RevMan Web 2021). We used fixed‐effect meta‐analysis for combining data because it was reasonable to assume that studies were estimating the same underlying treatment effect (i.e. where trials were examining the same intervention, and the trials' populations and methods were judged sufficiently similar).
Subgroup analysis and investigation of heterogeneity
Clinical subgroups
We prespecified secondary analyses of the primary outcomes for the infant, woman, child and adult to explore clinical diversity by examining interactions between the effect of repeat dose(s) of prenatal corticosteroids compared with women receiving no repeat prenatal corticosteroids/placebo and the following variables:
reasons the woman was considered at risk of preterm birth (e.g. presence or absence of ruptured membranes, prepartum haemorrhage, preterm labour, cervical incompetence, pre‐eclampsia and fetal growth restriction);
number of babies in utero (singleton, twins or higher order multiples);
type of corticosteroid given (betamethasone, dexamethasone);
planned interval between corticosteroid treatments (minimum interval of seven days or less, between eight and less than 14 days, 14 days or more);
planned number of repeat courses of corticosteroids to be given (one, two, three, four or more repeat courses);
planned dosage of corticosteroid given per treatment (12 mg or less, more than 12 mg to 24 mg, more than 24 mg);
planned dose of repeat dose of corticosteroid drug exposure/week (12 mg or less/week, more than 12 mg/week to 24 mg/week, more than 24 mg/week);
method of treatment administration (intramuscular, intravenous, intra‐amniotic); and
gestational age at which the first repeat treatment was given (less than 28, 28 to less than 32, 32 to 34, more than 34 completed weeks).
Sensitivity analysis
We carried out sensitivity analyses to explore the effect of risk of bias on outcomes of critical importance in the review (using the primary outcomes for each of the epochs). Where overall risk of bias was assessed as 'high risk' or 'some concerns' for a study outcome, we explored this by sensitivity analysis excluding these studies.
Sensitivity analysis was also performed to examine the effect of substantial levels of heterogeneity. We regarded heterogeneity as substantial if the I² statistic was greater than 30% or there was a low P value (less than 0.10) in the Chi² test for heterogeneity.
Summary of findings and assessment of the certainty of the evidence
For this update, we assessed the certainty of the evidence using the GRADE approach as outlined in the GRADE handbook) for the following outcomes (gdt.gradepro.org/app/handbook/handbook.html; Schünemann 2013).
Outcomes for the fetus/neonate/infant
Fetal or neonatal or infant death.
Respiratory distress syndrome (however defined by authors).
Severe lung disease (however defined by authors).
Chronic lung disease (however defined by authors).
Severe intraventricular haemorrhage (grade 3 or 4).
Necrotising enterocolitis (however defined by authors).
Composite serious outcome (however defined by authors).
Outcomes for the woman
Maternal death.
Maternal sepsis (any of chorioamnionitis during labour, endometritis, pyrexia after trial entry requiring the use of antibiotics, puerperal sepsis, intrapartum fever requiring the use of antibiotics, postnatal pyrexia or however defined by authors).
Caesarean section.
Discontinuation of therapy because of maternal side effects.
Adverse effects of corticosteroids (including gastrointestinal upset, insomnia, local injection site adverse effects (pain, bruising, haematoma or infection at the injection site)).
Admission to the intensive care unit.
Breastfeeding at hospital discharge.
For the child in early childhood (aged two to less than five years)
Total deaths (after randomisation).
Neurodevelopmental impairment at age two to less than five years (any of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), motor impairment (mild or major impairment by appropriate mode of assessment), or cerebral palsy, or however defined by authors).
Survival free of neurodevelopmental impairment at age two to less than five years (none of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), motor impairment (mild or major impairment by appropriate mode of assessment), cerebral palsy or however defined by authors).
Survival free of major neurodevelopmental impairment at age two to less than five years (none of: moderate or severe intellectual impairment (developmental quotient or intelligence quotient more than two SDs below the mean), moderate or severe cerebral palsy, major motor impairment, blindness (corrected visual acuity worse than 6/60 in the better eye) or deafness (hearing loss requiring amplification or worse), or however defined by authors).
For the child in mid‐ to late childhood (aged five to less than 18 years)
Total deaths (after randomisation).
Neurocognitive impairment at age five to less than 18 years (any of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), executive dysfunction, learning difficulties, motor impairment (mild or major impairment by appropriate mode of assessment), or cerebral palsy, or however defined by authors).
Survival free of neurocognitive impairment at age five to less than 18 years (none of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), executive dysfunction, learning difficulties, motor impairment (mild or major impairment by appropriate mode of assessment) cerebral palsy, or however defined by authors).
Survival free of major neurocognitive impairment at age five to less than 18 years (none of: moderate or severe intellectual impairment (developmental quotient or intelligence quotient more than two SD below the mean), major motor impairment, moderate or severe cerebral palsy, blindness (corrected visual acuity worse than 6/60 in the better eye) or deafness (hearing loss requiring amplification or worse), or however defined by authors).
For the child as an adult (aged 18 years or greater)
Total deaths (after randomisation).
Neurodevelopmental disability at age 18 years or greater (any of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), cerebral palsy, or however defined by authors).
Cardiovascular disease (including diagnosis of ischaemic heart disease, cerebrovascular disease, heart failure or however defined by authors).
Type 2 diabetes mellitus (however defined by authors).
Glucose intolerance (however defined by authors).
Obesity/overweight (however defined by authors).
Hypertension (however defined by authors).
We used GRADEpro GDT (GRADEpro GDT) to import data from Review Manager Web (RevMan Web 2021) to create summary of findings tables. We produced a summary of the intervention effect and a measure of certainty for each of the above outcomes using the GRADE approach. The GRADE approach uses five considerations (risk of bias, inconsistency of results, imprecision, indirectness of evidence and publication bias) to assess the certainty of the body of evidence. The evidence can be downgraded from 'high certainty' by one level for serious (or by two levels for very serious) limitations in any of the five considerations.
We used the GRADE approach to assess the evidence available for all secondary outcomes, with the level of certainty and individual domain judgements described in Table 5; Table 6; Table 7; Table 8; and Table 9.
1. GRADE certainty of evidence assessments for secondary outcomes for the fetus/neonate/infant.
| Secondary outcome | Certainty of evidence | Risk of bias | Inconsistency | Imprecision | Indirectness | Publication bias | Comments |
| Gestational age at birth ≥ 37 weeks | Low | Not serious | Serious | Serious | Serious | Undetected | Downgraded 1 level due to indirectness as participants in the trials varied markedly in their gestational age at entry, limiting the generalisability of this finding. Downgraded 1 level due to inconsistency of effect as evidenced by moderate statistical heterogeneity. |
| Gestational age at birth < 37 weeks | Low | Not serious | Serious | Not serious | Serious | Undetected | Downgraded 1 level due to indirectness as participants in the trials varied markedly in their gestational age at entry, limiting the generalisability of this finding. Downgraded 1 level due to inconsistency of effect as evidenced by moderate statistical heterogeneity. |
| Gestational age at birth < 34 weeks | Moderate | Not serious | Not serious | Not serious | Serious | Undetected | Downgraded 1 level due to indirectness as participants in the trials varied markedly in their gestational age at entry, limiting the generalisability of this finding. |
| Gestational age at birth < 28 weeks | Low | Not serious | Not serious | Serious | Serious | Undetected | Downgraded 1 level due to indirectness as participants in the trials varied markedly in their gestational age at entry, limiting the generalisability of this finding. Downgraded 1 level due to imprecision (95% CI including possible benefit and harm). |
| Mean gestational age at birth | Moderate | Not serious | Not serious | Not serious | Serious | Undetected | Downgraded 1 level due to indirectness as participants in the trials varied markedly in their gestational age at entry, limiting the generalisability of this finding. |
| Mean birthweight | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/10 trials had some concerns of risk of bias. Limiting the analysis to trials at low risk of bias did not markedly alter the findings. |
| Mean birthweight adjusted for gestational age | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Interval between trial entry and birth | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded due to imprecision due to an inadequate sample size for this outcome. |
| Small for gestational age | High | Not serious | Not serious | Not serious | Not serious | Undetected | 2/7 trials had some concerns of risk of bias. Limiting the analysis to trials at low risk of bias did not markedly alter the findings. |
| Mean length at birth | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/6 trials had some concerns of risk of bias. Limiting the analysis to trials at low risk of bias did not markedly alter the findings. |
| Mean length at birth adjusted for gestational age | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Mean head circumference at birth | High | Not serious | Not serious | Not serious | Not serious | Undetected | 2/10 trials had risk of bias (1 some concerns and 1 high risk). Limiting the analysis to trials at low risk of bias did not markedly alter the findings. |
| Mean head circumference Z score at birth | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Mean weight at primary hospital discharge | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/2 trials had some concerns for risk of bias. Limiting the analysis to
trials at low risk of bias did not markedly alter the findings. Not downgraded for inconsistency, as the degree of statistical heterogeneity was small, measured across only 2 studies with similar results. |
| Mean weight Z score at primary hospital discharge | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/2 trials had some concerns for risk of bias. Limiting the analysis to trials at low risk of bias did not markedly alter the findings. |
| Mean length at primary hospital discharge | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/2 trials had some concerns for risk of bias. Limiting the analysis to trials at low risk of bias did not markedly alter the findings. |
| Mean length Z score at primary hospital discharge | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/2 trials had some concerns for risk of bias. Limiting the analysis to trials at low risk of bias did not markedly alter the findings. |
| Mean head circumference at primary hospital discharge | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/2 trials had some concerns for risk of bias. Limiting the analysis to trials at low risk of bias did not markedly alter the findings. |
| Mean head circumference Z score at primary hospital discharge | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/2 trials had some concerns for risk of bias. Limiting the analysis to trials at low risk of bias did not markedly alter the findings. |
| Mean weight at infant follow‐up | Very low | Very serious | Not serious | Serious | Not serious | Undetected | Of 2 trials, 1 had some concerns of risk of bias and 1 had high risk of bias. Downgraded for imprecision due to an inadequate sample size for this outcome. |
| Mean weight Z score at infant follow‐up | Very low | Very serious | Not serious | Serious | Not serious | Undetected | The only included trial had high risk of bias. Downgraded for imprecision as the 95% CI included marked increase and marked decrease in Z score. |
| Mean length at infant follow‐up | Very low | Very serious | Not serious | Serious | Not serious | Undetected | Of 2 trials, 1 had some concerns of risk of bias and 1 had high risk of bias. Downgraded for imprecision due to inadequate sample size for this outcome. |
| Mean length Z score at primary infant follow‐up | Very low | Very serious | Not serious | Serious | Not serious | Undetected | The only included trial had high risk of bias. Downgraded for imprecision as the 95% CI included marked increase and marked decrease in Z score. |
| Mean head circumference at infant follow‐up | Very low | Very serious | Not serious | Serious | Not serious | Undetected | Of 2 trials, 1 had some concerns of risk of bias and 1 had high risk of bias. Downgraded for imprecision as the number of infants was too small for confident conclusions regarding this outcome. |
| Mean head circumference Z score at infant follow‐up | Very low | Very serious | Not serious | Serious | Not serious | Undetected | The only included trial had high risk of bias. Downgraded for imprecision as the 95% CI included marked increase and marked decrease in Z score. |
| Admission to neonatal intensive care unit | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Proven neonatal infection while in the neonatal intensive care unit | High | Not serious | Not serious | Not serious | Not serious | Undetected | 2/8 trials had some concerns of risk of bias. Limiting the analysis to trials at low risk of bias did not markedly change the findings. |
| Early systemic neonatal infection | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/4 trials had high risk of bias. Limiting the analysis to trials at low risk of bias did not markedly change the findings. |
| Late systemic neonatal infection | Low | Not serious | Not serious | Very Serious | Not serious | Undetected | 1/2 trials had high risk of bias. Limiting the analysis to trials at low risk of bias did not markedly change the findings. Downgraded for imprecision due to small numbers of events with a limited sample size and wide 95% CI including benefit and marked harm. |
| Retinopathy of prematurity | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | 2/8 trials had some concerns and 1 had high risk of bias. Limiting the analysis to trials at low risk of bias did not markedly change the findings. Downgraded for imprecision due to a 95% CI including benefit and harm. |
| Periventricular leukomalacia | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | 1/8 trials had some concerns and 1 had high risk of bias. Limiting the analysis to trials at low risk of bias did not markedly change the findings. Downgraded for imprecision due to a wide 95% CI including marked benefit and harm. |
| Neonatal encephalopathy | Very low | Very serious | Not serious | Very serious | Not serious | Undetected | Downgraded 2 levels as the only trial reporting this outcome had high risk of bias for lack of blinding and subjective outcome measurement. Downgraded 2 levels for imprecision due to small numbers of events with a small sample size and a wide 95% CI including marked benefit and marked harm. |
| Patent ductus arteriosus | Low | Very Serious | Not serious | Not serious | Not serious | Undetected | 1/7 trials had some concerns and 1 had high risk of bias. Limiting the analysis to trials at low risk of bias changed the findings. |
| Use of respiratory support | High | Not serious | Not serious | Not serious | Not serious | Undetected | |
| Use of invasive respiratory support | Moderate | Not serious | Serious | Not serious | Not serious | Undetected | 1/6 trials had some concerns of risk of bias. Limiting the analysis to trials at low risk of bias did not markedly change the findings. Downgraded for inconsistency as evidenced by marked statistical heterogeneity, possibly related to different types of invasive respiratory support in use. |
| Duration of invasive respiratory support | Moderate | Not serious | Serious | Not serious | Not serious | Undetected | Downgraded for inconsistency as evidenced by marked statistical heterogeneity, possibly related to different types of invasive respiratory support in use. |
| Use of non‐invasive respiratory support | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/3 trials had some concerns of risk of bias. Limiting the analysis to trials at low risk of bias did not markedly change the findings. |
| Duration of non‐invasive respiratory support | Low | Not serious | Not serious | Very serious | Not serious | Undetected | Downgraded 2 levels for imprecision due to small sample size and a wide 95% CI including benefit and minimal harm. |
| Use of oxygen supplementation | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Duration of oxygen supplementation | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision due to a 95% CI including benefit and harm. |
| Use of surfactant | Moderate | Not serious | Serious | Not serious | Not serious | Undetected | 2/10 trials had some concerns of risk of bias. Limiting the analysis to trials at low risk of bias did not markedly change the findings. Downgraded for Inconsistency as evidenced by marked statistical heterogeneity. |
| Use of nitric oxide for respiratory support | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision due to a 95% CI including marked benefit and harm. |
| Use of postnatal corticosteroids | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision due to a 95% CI including benefit and marked harm. |
| Use of inotropic support | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Air leak syndrome | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | 1/4 trials had some concerns of risk of bias. Limiting the analysis to trials at low risk of bias did not markedly change the findings. Downgraded for imprecision due to a 95% CI including marked benefit and marked harm. |
| Apgar score < 7 at 5 minutes | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded due to imprecision due to a 95% CI including marked benefit and harm. |
| Cardiac hypertrophy | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded for risk of bias as the only trial reporting data for this outcome had some concerns regarding missing outcome data. |
| Measures of hypothalamic‐pituitary‐adrenal axis function | Very low | Serious | Serious | Serious | Not serious | Undetected | Both trials had some concerns of risk of bias for this outcome. Downgraded for inconsistency as different measures had different findings. Downgraded for imprecision as all measures except cord blood cortisol had inadequate sample size. |
CI: confidence interval.
2. GRADE certainty of evidence assessments for secondary outcomes for the woman.
| Secondary outcome | Certainty of evidence | Risk of bias | Inconsistency | Imprecision | Indirectness | Publication bias | Comments |
| Puerperal sepsis | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision as 95% CI included both benefit and marked harm. |
| Chorioamnionitis during labour | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision as 95% CI included both benefit and marked harm. |
| Endometritis | Low | Not serious | Serious | Serious | Not serious | Undetected | Downgraded for imprecision as 95% CI included both benefit and marked harm. Downgraded for inconsistency as evidenced by marked statistical heterogeneity. |
| Pyrexia after trial entry requiring the use of antibiotics | Low | Not serious | Not serious | Very Serious | Not serious | Undetected | Downgraded 2 levels for imprecision as 95% CI included both marked benefit and harm and data were from a single trial of limited size. |
| Postpartum haemorrhage | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision as the number of events was too low for a reliable effect estimate. |
| Postnatal pyrexia | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision as 95% CI included both marked benefit and harm. |
| Preterm prelabour rupture of the membranes after trial entry | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision as 95% CI included both marked benefit and harm. |
| Mode of birth: vaginal birth | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Hypertension | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision as 95% CI included both possible benefit and harm. |
| Glucose intolerance | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | 1/2 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials only did not markedly change the findings. Downgraded for imprecision as 95% CI included both benefit and marked harm. |
| Postnatal depression | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded 1 level for imprecision as 95% CI included both benefit and harm. |
| Local injection site adverse effects | Low | Not serious | Very serious | Not serious | Not serious | Undetected | Downgraded 2 levels for inconsistency due to severe heterogeneity. |
| Insomnia after treatment | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/4 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials did not markedly change the findings. |
| Gastrointestinal adverse effects of treatment | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision as the number of events was too low and the study size too small for a reliable effect estimate. |
CI: confidence interval.
3. GRADE certainty of evidence assessments for secondary outcomes for the child in early childhood (aged two to less than five years).
| Secondary outcome | Certainty of evidence | Risk of bias | Inconsistency | Imprecision | Indirectness | Publication bias | Comments |
| Child behaviour | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | 1/3 trials reporting data for this outcome had some concerns of risk of bias for missing data. Not downgraded for inconsistency as clearly different methods of measurement were used. Downgraded for imprecision as 2/3 trials had 95% CIs that included both marked harm and either benefit or no effect. |
| Motor impairment | Moderate | Not serious | Serious | Not serious | Not serious | Undetected | 1/2 trials had some risks of bias but limiting the analysis to low risk of bias trials did not change the findings. Downgraded for inconsistency as evidenced by statistical heterogeneity. |
| Deafness/hearing impairment | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | 2/4 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials did not change the findings. Downgraded for imprecision as 95% CI included both marked benefit and marked harm. |
| Blindness/visual impairment | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | 1/3 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials did not change the findings. Downgraded for imprecision as 95% CI included both marked benefit and marked harm. |
| Mean weight at early childhood follow‐up | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/4 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials did not change the findings. |
| Weight for age at early childhood follow‐up | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/3 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials did not change the findings. |
| Low weight for age | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision as 95% CI included both benefit and possible harm. |
| Mean height at early childhood follow‐up | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/4 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials did not change the findings. |
| Height for age at early childhood follow‐up | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/3 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials did not change the findings. |
| Short for age | Low | Not serious | Serious | Serious | Not serious | Undetected | Downgraded for imprecision as 95% CI included both benefit and marked harm. Downgraded for inconsistency as evidenced by marked heterogeneity. |
| Mean head circumference at early childhood follow‐up | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/4 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials did not change the findings. |
| Head circumference for age at early childhood follow‐up | High | Not serious | Not serious | Not serious | Not serious | Undetected | 1/3 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials did not change the findings. |
| Small head circumference for age | Moderate | Not serious | Serious | Not serious | Not serious | Undetected | Downgraded for inconsistency as evidenced by marked heterogeneity. |
| Mean systolic blood pressure | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Systolic blood pressure Z score | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded for risk of bias as the only included trial had some concerns for risk of bias. |
| Mean diastolic blood pressure | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Diastolic blood pressure Z score | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded for risk of bias as the only included trial had some concerns for risk of bias. |
| Hypertension | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded for risk of bias as the only included trial had some concerns for risk of bias. |
| Asthma or recurrent wheeze | Moderate | Not serious | Serious | Serious | Not serious | Undetected | 2/3 trials had some concerns of risk of bias but limiting the analysis to
low‐risk trials did not change the findings. Downgraded for inconsistency as evidenced by marked heterogeneity. Downgraded for imprecision as 95% CI included both benefit and possible harm. |
| Any respiratory disease | High | Not serious | Not serious | Not serious | Not serious | Undetected | 2/3 trials had some concerns of risk of bias but limiting the analysis to low‐risk trials did not change the findings. |
CI: confidence interval.
4. GRADE certainty of evidence assessments for secondary outcomes for the child in mid‐ to late childhood (aged five to less than 18 years).
| Secondary outcome | Certainty of evidence | Risk of bias | Inconsistency | Imprecision | Indirectness | Publication bias | Comments |
| Child behaviour | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as both trials had some concerns of risk of bias due to missing data. |
| Deafness/hearing impairment | Low | Serious | Not serious | Serious | Not serious | Undetected | Downgraded 1 level as both trials had some concerns of risk of bias due to missing data. Downgraded for imprecision as the 95% CI included both benefit and marked harm. |
| Blindness/visual impairment | Low | Serious | Not serious | Serious | Not serious | Undetected | Downgraded 1 level as both trials had some concerns of risk of bias due to missing data. Downgraded for imprecision as the 95% CI included both benefit and marked harm. |
| Mean weight | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Mean weight Z score | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Mean height | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Mean height Z score | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Mean head circumference | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Mean head circumference Z score | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Body mass index Z score | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Mean systolic blood pressure | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Mean systolic blood pressure Z score | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Mean diastolic blood pressure | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Mean diastolic blood pressure Z score | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Forced expiratory volume in 1 second (FEV1) Z score | Very low | Very serious | Not serious | Serious | Not serious | Undetected | Downgraded 2 levels as the only included trial had high risk of bias due
to missing data. Downgraded 1 level for imprecision as 95% CI included both benefit and harm. |
| Forced vital capacity (FVC) Z score | Very low | Very serious | Not serious | Serious | Not serious | Undetected | Downgraded 2 levels as the only included trial had high risk of bias due to missing data. Downgraded 1 level for imprecision as 95% CI included both benefit and harm. |
| FEV1/FVC ratio Z score | Very low | Very serious | Not serious | Serious | Not serious | Undetected | Downgraded 2 levels as the only included trial had high risk of bias due to missing data. Downgraded 1 level for imprecision as 95% CI included both benefit and harm. |
| Asthma or recurrent wheeze | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Any respiratory disease | Very low | Very serious | Not serious | Serious | Not serious | Undetected | Downgraded 2 levels as the only included trial had high risk of bias due to missing data. Downgraded 1 level for imprecision as 95% CI included both benefit and harm. |
| Measures of insulin and glucose homeostasis | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Measures of hypothalamic‐pituitary‐adrenal (HPA) axis function | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Body composition | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
| Bone density | Moderate | Serious | Not serious | Not serious | Not serious | Undetected | Downgraded 1 level as the only included trial had some concerns of risk of bias due to missing data. |
CI: confidence interval.
5. GRADE certainty of evidence assessments for secondary outcomes for health services.
| Secondary outcome | Certainty of evidence | Risk of bias | Inconsistency | Imprecision | Indirectness | Publication bias | Comments |
| Length of postnatal hospitalisation for the woman | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Length of infant hospitalisation | Moderate | Not serious | Not serious | Serious | Not serious | Undetected | Downgraded for imprecision as 95% CI included both benefit and harm. |
| Hospital re‐admission by early childhood follow‐up | High | Not serious | Not serious | Not serious | Not serious | Undetected | — |
| Hospital re‐admission by mid‐ to late childhood follow‐up | Very low | Very serious | Not serious | Serious | Not serious | Undetected | Downgraded 2 levels for risk of bias as the only study included was at high risk of bias due to outcome measurement. Downgraded for imprecision as 95% CI included both benefit and marked harm. |
CI: confidence interval.
Results
Description of studies
Results of the search
See: Figure 2.
2.
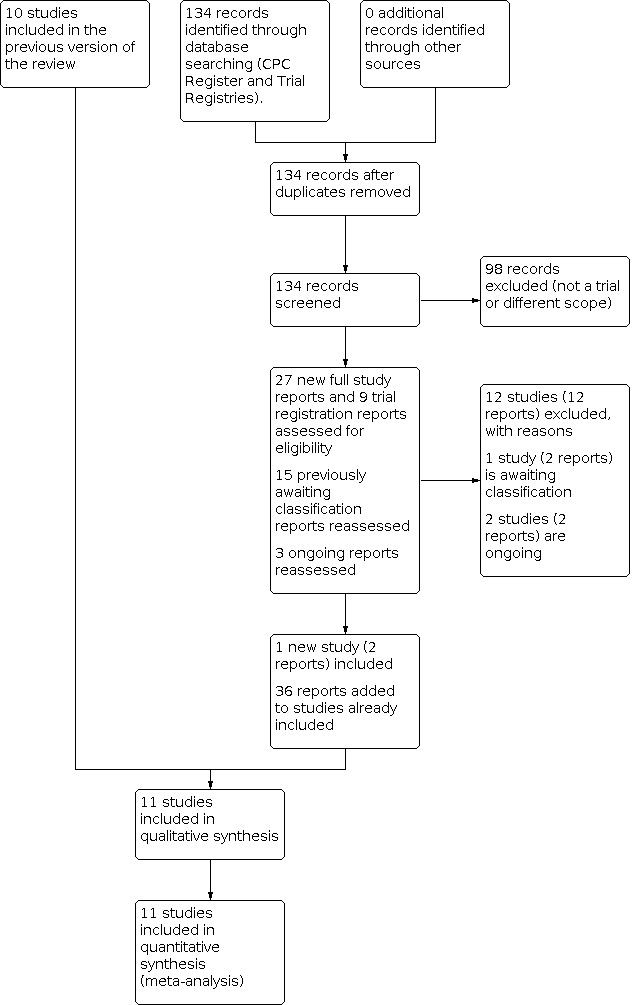
Study flow diagram.
The updated search identified 36 new study reports to assess in full. We also reassessed the three studies (15 reports) awaiting classification in the previous version of the review and the two ongoing trials (three reports). We included one new study (two reports) and excluded 12 new studies (12 reports). One study is awaiting classification (two reports), and two are ongoing. The remaining 36 were additional reports of studies already included and have been added under the main study report.
Screening eligible studies for trustworthiness
One study that was awaiting classification in the previous version of this review met our inclusion criteria, but we judged that it did not meet our criteria for trustworthiness due to concerns about randomisation processes as identical numbers were randomised to each group despite using a randomisation table for 1348 participants (Atarod 2014). There was also no explanation for the exclusion of 104 women after randomisation (Atarod 2014). We attempted to contact the authors, but we received no response to our queries; therefore, it remains as awaiting classification (see Studies awaiting classification).
Included studies
See Characteristics of included studies table for full details.
The 11 included trials randomised 4895 women (5975 babies) (Aghajafari 2002: 12 women, 16 fetuses; Crowther 2006: 982 women, 1146 fetuses; Garite 2009: 437 women, 577 fetuses; Guinn 2001: 502 women, 589 fetuses; Mazumder 2008: 76 women, 76 fetuses; McEvoy 2002: 37 women, 37 fetuses; McEvoy 2010: 85 women, 113 fetuses; Murphy 2008: 1858 women, 2309 fetuses; Peltoniemi 2007: 249 women, 328 fetuses; TEAMS 1999: 162 women, 188 fetuses; Wapner 2006: 495 women, 594 fetuses).
Five trials were conducted in the US (Garite 2009; Guinn 2001; McEvoy 2002; McEvoy 2010; Wapner 2006); one each in Canada (Aghajafari 2002), India (Mazumder 2008), the UK (TEAMS 1999), and Finland (Peltoniemi 2007); one in Australia and New Zealand (Crowther 2006); and one involved 20 countries (Murphy 2008).
Gestational age at trial entry varied between trials: 24 to 30 weeks in Aghajafari 2002; 25 to 32 weeks in Murphy 2008; 25 to less than 33 weeks in Guinn 2001 and Garite 2009; 26 to 33 weeks in Mazumder 2008 and McEvoy 2010; 25 to 33 weeks in McEvoy 2002; 23 to less than 32 weeks in Wapner 2006; less than 32 weeks in Crowther 2006 and TEAMS 1999; and less than 34 weeks in Peltoniemi 2007. All women were at increased risk of preterm birth and had received a single course of prenatal corticosteroids one week or more before trial entry. The type, amount and timing regimen for administration of the corticosteroid given for the pretrial course of prenatal corticosteroids varied between trials.
In seven trials, women were eligible for inclusion seven or more days after a pretrial course (Aghajafari 2002; Crowther 2006; Guinn 2001; Mazumder 2008; McEvoy 2002; TEAMS 1999; Peltoniemi 2007); in one trial between seven and 10 days after a pretrial course (Wapner 2006); in two trials 14 or more days after a pretrial course (Garite 2009: McEvoy 2010); and in one trial between 14 and 21 days after a pretrial course (Murphy 2008).
All trials started recruitment between 1996 and 2004. Four trials started recruitment prior to 2000 and seven trials started recruitment after 2000. All trials completed recruitment between 1999 and 2008.
Four trials were terminated early (Guinn 2001; McEvoy 2002; TEAMS 1999; Wapner 2006). Guinn 2001 and McEvoy 2002 were terminated early based on concern from reports in the literature of potential harm from repeat courses of prenatal corticosteroids and interim analyses suggesting that they were unlikely to detect a difference between groups for the primary outcomes. Wapner 2006 was terminated early due to a trend towards reduced birthweight in the repeat prenatal corticosteroids group without any suggestion of benefit for the primary outcome.
Exclusion criteria for recruitment to the included trials
Aghajafari 2002: chronic doses of corticosteroids secondary to medical conditions, contraindication to corticosteroids, clinical evidence of chorioamnionitis or fetus(es) had a known lethal congenital anomaly.
Crowther 2006: in second stage of labour, chorioamnionitis needing urgent delivery or if further corticosteroid therapy was judged essential.
Garite 2009: major fetal anomaly, cervical dilation 5 cm or more, multiple pregnancy of higher order than twins, ruptured membranes, documented lung maturity, receiving corticosteroids for other indications, HIV infection or active tuberculosis.
Guinn 2001: required immediate delivery, fetal anomalies incompatible with life, documented fetal lung maturity, maternal active tuberculosis or HIV infection.
Mazumder 2008: unreliable gestational age, frank chorioamnionitis, major fetal malformation or unavailable for follow‐up.
McEvoy 2002: insulin‐dependent diabetes, drug‐addiction, known lethal congenital anomaly or multiple pregnancy.
McEvoy 2010: insulin‐dependent diabetes, major documented fetal or chromosomal abnormality, multiple pregnancy of higher order than twins, clinical chorioamnionitis, first course of prenatal corticosteroids given before 24 weeks' gestation or chronic steroid use during pregnancy for clinical care.
Murphy 2008: contraindication to corticosteroid use, needed chronic doses of corticosteroid drugs, had evidence of chorioamnionitis, known lethal congenital abnormality, had an initial course of corticosteroids before 23 weeks' gestation, previously participated in MACS (Multiple Antenatal Corticosteroids) trial, women with a multiple pregnancy with fetal death after 13 weeks' gestation.
Peltoniemi 2007: long‐term maternal corticosteroid use, clinical chorioamnionitis or lethal disease of the fetus.
TEAMS 1999: maternal long‐term systemic corticosteroid therapy (not including inhaled or topical therapy).
Wapner 2006: preterm prelabour rupture of the membranes prior to randomisation, confirmed fetal lung maturity, chorioamnionitis, major fetal anomaly, non‐reassuring fetal status, systemic corticosteroid use during the current pregnancy or insulin‐dependent diabetes.
Interventions
The type of corticosteroid planned to be given as treatment was betamethasone for all the trialsa, although the gestational age at which treatment could begin or was continued until varied slightly between trials.
In six trials, a planned treatment course was two doses of betamethasone 12 mg/dose, intramuscularly, at weekly intervals (Aghajafari 2002; Guinn 2001; Mazumder 2008; McEvoy 2002; TEAMS 1999; Wapner 2006).
Aghajafari 2002 gave a weekly course of betamethasone (two doses of 12 mg/dose (Celestone Soluspan; Schering Canada Inc.) intramuscularly, 24 hours apart) until 33 weeks or birth if the woman remained at increased risk of preterm birth.
Guinn 2001 used a weekly course of betamethasone (two doses of 12 mg/dose, intramuscularly 24 hours apart) until 34 weeks or birth, whichever came first.
Mazumder 2008 used betamethasone 12 mg intramuscularly, two doses, 24 hours apart until the end of the 33rd week of gestation.
McEvoy 2002 used a weekly course of betamethasone (two doses of 12 mg/dose (Celestone Soluspan; Schering Corporation, Kenilworth, New Jersey)), intramuscularly, until 34 weeks or birth.
TEAMS 1999 used a weekly course of betamethasone (two doses of 12 mg given 12 or 24 hours apart) but allowed for a dosing interval of up to 14 days depending on local protocols.
Wapner 2006 used a weekly course of betamethasone (two doses of 12 mg as betamethasone sodium phosphate 6 mg and betamethasone acetate 6 mg, intramuscularly in 24 hours) until birth or 33 weeks and six days, limited to four repeat courses after the first 67 women.
Crowther 2006 used a single intramuscular injection of betamethasone 11.4 mg (Celestone Chronodose, Schering‐Plough, Sydney, Australia) containing betamethasone sodium phosphate 7.8 mg and betamethasone acetate 6 mg, repeated weekly if the woman remained undelivered, less than 32 weeks' gestation and the responsible clinician regarded her as at continued risk of preterm birth.
Murphy 2008 used a course of betamethasone (two doses of 12 mg/dose as betamethasone sodium phosphate 6 mg and betamethasone acetate 6 mg: Celestone Schering‐Plough Corporation, Madison, New Jersey, USA), intramuscularly, 24 hours apart, every 14 days (if the woman remained at risk of preterm birth after their first course of study treatment) until 33 weeks' gestation or birth. For women with preterm prelabour rupture of membranes, it was recommended that treatment stop at 32 weeks' gestation.
Three trials planned only a single repeat course of treatment; Garite 2009a used a single course consisting of two doses of betamethasone 12 mg, intramuscularly, 24 hours apart (preparation not specified); McEvoy 2010 used a single course of two doses of betamethasone 12 mg/dose (Celestone Soluspan; Schering Corporation, Kenilworth, New Jersey), intramuscularly, 24 hours apart; and Peltoniemi 2007 used a single intramuscular injection of intramuscular of betamethasone 12 mg (preparation not specified).
aOf note, Garite 2009, due to unavailability of betamethasone on occasion, used dexamethasone 6 mg, intramuscularly every 12 hours up to four doses or similar placebo regimen for 61 (14%) women.
Primary outcomes
Primary outcomes were predominantly focused on neonatal morbidity and mortality. Seven trials used a composite primary outcome incorporating both neonatal death, either RDS or severe RDS and other measures of neonatal morbidity (Aghajafari 2002; Garite 2009; Guinn 2001; Mazumder 2008; Murphy 2008; Peltoniemi 2007; Wapner 2006). Crowther 2006 reported primary outcomes of RDS, lung disease severity, measures of respiratory support and growth measurements. For TEAMS 1999, the primary outcomes were neonatal death and neurodevelopmental delay at age two years (corrected for gestational age at birth). Aghajafari 2002 reported primary outcomes focussed on the rate of recruitment over a 12‐month period, risk of complications requiring discontinuation of study treatment, and measures of maternal and fetal HPA function following birth. The primary outcomes for McEvoy 2002 and McEvoy 2010 were measures of lung function: functional residual capacity and respiratory compliance.
All trials had a range of secondary outcomes of clinical relevance.
At early childhood follow‐up, primary outcomes varied by trial. For Crowther 2006, the prespecified primary outcomes were survival at two years' corrected age free of major neurodisability (defined as survival free of moderate or severe disability); and body size (weight, height and head circumference). For Murphy 2008, the primary outcome was death or the presence of neurological impairment at 18 to 24 months' corrected age. Neurological impairment was defined as the presence of cerebral palsy or cognitive delay. For Peltoniemi 2007, the power analysis at follow‐up was based on survival without neurodevelopmental impairment. For Wapner 2006, the power analysis was based on the prespecified developmental outcome of the Bayley Mental Developmental Index score.
Two studies performed follow‐up in mid‐childhood (Crowther 2006; Murphy 2008). For Crowther 2006, the primary outcome for mid‐childhood follow‐up was survival free of any neurosensory disability at six to eight years' corrected age. For Murphy 2008, the primary outcome for mid‐childhood follow‐up was a composite of death or survival with a neurodevelopmental disability at five years of age.
Funding
All trials declared their sources of funding. One trial declared that it had no funding support (Mazumder 2008). The sources of funding support for the other 10 trials were as follows: the Canadian Institutes of Health Research Senior Scientist Award (Aghajafari 2002); the Australian National Health and Medical Research Council, the Channel 7 Research Foundation of South Australia, the Women's and Children's Hospital Research Foundation, Adelaide and The Department of Obstetrics and Gynaecology, The University of Adelaide, South Australia (Crowther 2006); the Pediatrix Medical Group (Garite 2009); March of Dimes grant, the Berlex Foundation, the Wisconsin Perinatal Association, the Perinatal Clinical Research Center at the University of Colorado Health Sciences Center (grant from the General Clinical Research Centers Program, National Centers for Research Resources, National Institutes of Health), and the participating departments (Guinn 2001); the American Lung Association (McEvoy 2002); Oregon Health and Science University, and the American Lung Association (McEvoy 2010); the Canadian Institutes of Health Research (Murphy 2008); the Foundation for Paediatric Research in Finland, the Alma and KA Snellmann Foundation, the Sigrid Juselius Foundation and hospital research funds (Peltoniemi 2007); Action Medical Research UK (TEAMS 1999); and the National Institute of Child Health and Human Development (Wapner 2006).
Declarations of interest
Seven trials declared there were no conflicts of interest (Aghajafari 2002; Crowther 2006; Garite 2009; Mazumder 2008; McEvoy 2010; Murphy 2008; Peltoniemi 2007).
For Wapner 2006, author Dr Mercer declared receiving consulting fees from Columbia Laboratories and reported no other conflicts of interest.
Three trials did not have an identified declaration of interest (Guinn 2001; McEvoy 2002; TEAMS 1999).
Trial registration
None of the trials were confirmed as being registered prospectively.
Five trials were registered during recruitment (Crowther 2006; Garite 2009; Murphy 2008; TEAMS 1999; Wapner 2006).
Two trials were registered retrospectively (McEvoy 2010; Peltoniemi 2007).
Four trials had no registration document identified (Aghajafari 2002; Guinn 2001; Mazumder 2008; McEvoy 2002).
Excluded studies
We excluded 16 studies; four were excluded in the last version of this review and 12 from the search for this update (Bontis 2011; CTRI/2017/04/008326; CTRI/2017/05/008721; Ernawati 2016; EUCTR2009‐010759‐29‐BE; Gyamfi‐Bannerman 2016; IRCT2014090912789N6; IRCT2015120415634N2; IRCT20191202045571N1; Kashanian 2018; Mercer 2001; NCT03446937; Romejko‐Wolniewicz 2013; Schmitz 2019; Sohrabvand 2001; Thorp 2000).
The reasons for study exclusion were women not receiving a first course of corticosteroids prior to trial entry, trials that did not compare repeat dose(s) of corticosteroid to a single course of corticosteroid, trials that were using a different treatment and one trial that was not truly randomised. Descriptions of excluded studies and the reasons for exclusion are described in the Characteristics of excluded studies table.
Studies awaiting classification
One trial from Iran is awaiting classification to establish its eligibility for inclusion as it did not meet trustworthiness criteria and we received no response from the authors for clarification of concerns (Atarod 2014). See Characteristics of studies awaiting classification table.
Ongoing studies
There are two ongoing trials of repeat prenatal corticosteroids in the context of preterm, prelabour rupture of membranes. The trials are set in the US and have yet to complete recruitment (NCT02469519; NCT02939742). See Characteristics of ongoing studies table.
Risk of bias in included studies
Overall and domain level risk of bias assessments for each outcome are included alongside forest plots for each outcome and in Figure 3. Domain level risk of bias judgements and supporting notes are included in the Risk of Bias tables (located after the Characteristics of included studies). To access further detailed risk of bias assessment data, including answers to signalling questions use the following link.
3.

Risk of bias for the randomisation process is a study‐level assessment and was low in all studies included.
The risk of bias for fetal/neonatal/infant outcomes was generally low for all outcomes. There were some concerns of risk of bias overall for Wapner 2006 for all outcomes due to missing data, as outcomes were only reported for one twin in the instance of multiple gestations. For Mazumder 2008, there were some concerns of risk of bias overall for all outcomes due to a lack of blinding of treatment allocation as this trial was open‐label and details of analysis plans were not clearly described.
For the women, the risk of bias for all primary outcomes was low across all studies except for some concerns of risk of bias for selection of the reported result for Garite 2009 for the outcome of maternal sepsis.
At early childhood follow‐up, there were some concerns of risk of bias for Wapner 2006 and high risk of bias for Peltoniemi 2007 for most of the primary outcomes. All trials had low risk of bias for the outcome of death since randomisation up to early childhood.
For outcomes at mid‐childhood, missing data was an important source of risk of bias, resulting in judgements of some concerns of bias in all primary outcomes in this category except for the outcome risk of death after randomisation.
Published prospective statistical analysis plans were not available for review. Primary publications for trials were all published in 2010 or earlier. The lack of a published statistical analysis plan was not considered to indicate a risk of bias unless it was felt likely that analysis methods or reported results were likely to have been influenced by a lack of prespecified plans or chosen on the basis of results.
Effects of interventions
See: Table 1; Table 2; Table 3; Table 4
Repeat dose(s) of prenatal corticosteroids versus placebo/standard care
Primary outcomes for the fetus/neonate/infant
Fetal or neonatal or infant death (less than one year of age)
For of fetal or neonatal or infant death less than one year of age, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.95, 95% CI 0.73 to 1.24; I2 = 0%, Chi2 P= 0.64; 10 trials, 5849 fetuses randomised; Analysis 1.1).
1.1. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 1: A1: Fetal or neonatal or infant death (< 1 year of age)
All trials reported death at the fetal or neonatal stage of life either separately or as a combined outcome. No trials specifically reported infant death before one year of age.
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of two trials with some concerns of bias (Mazumder 2008; Wapner 2006), but the result was similar to the overall analysis (RR 1.01, 95% CI 0.76 to 1.33; I2 = 0%, Chi2 P = 0.63; 8 trials, 5179 fetuses randomised).
Certainty of the evidence
The certainty of the evidence moderate. Certainty was downgraded one level due to imprecision as the 95% CI included both benefit and harm. Certainty was not downgraded for risk of bias as limiting the analysis to trials with low risk of bias did not markedly change the findings. There were no concerns regarding inconsistency or indirectness. Symmetry in the funnel plot was not suggestive of publication bias (Figure 4).
4.

Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.1; planned interval between corticosteroid treatments: Analysis 9.1; planned dose of corticosteroids to be given per treatment course: Analysis 10.1; planned dose of repeat dose of corticosteroid drug exposure per week: Analysis 11.1).
8.1. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 1: A1: Fetal or neonatal or infant death (< 1 year of age)
9.1. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 1: A1: Fetal or neonatal or infant death (< 1 year of age)
10.1. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 1: A1: Fetal or neonatal or infant death (< 1 year of age)
11.1. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 1: A1: Fetal or neonatal or infant death (< 1 year of age)
Fetal death
For fetal death, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.82, 95% CI 0.24 to 2.84; I2 = 0%, Chi2 P = 0.98; 7 trials, 2758 fetuses randomised; Analysis 1.2).
1.2. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 2: A2: Fetal death
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with some concerns of bias (Mazumder 2008), but the result was similar to the overall analysis (RR 1.01, 95% CI 0.25 to 4.01; I2 = 0%, Chi2 P = 1.00; 6 trials, 2682 fetuses randomised).
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded one level due to imprecision as evidenced by a few events and a 95% CI that included marked benefit and marked harm. Certainty was not downgraded for risk of bias as limiting analysis to only those trials with low risk of bias did not markedly change the findings. There were no concerns regarding inconsistency, indirectness or publication bias.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.2; planned interval between corticosteroid treatments: Analysis 9.2; planned dose of corticosteroids to be given per treatment course: Analysis 10.2; planned dose of repeat dose of corticosteroid drug exposure per week: Analysis 11.2).
8.2. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 2: A2: Fetal death
9.2. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 2: A2: Fetal death
10.2. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 2: A2: Fetal death
11.2. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 2: A2: Fetal death
Neonatal death
For neonatal death, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.91, 95% CI 0.62 to 1.34; I2 = 7%, Chi2 P = 0.37; 7 trials, 2758 fetuses randomised; Analysis 1.3).
1.3. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 3: A3: Neonatal death
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with some concerns of bias (Mazumder 2008), but the result was similar to the overall analysis (RR 0.97, 95% CI 0.64 to 1.45; I2 = 15%, Chi2 P = 0.32; 6 trials, 2682 fetuses randomised)
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded by one level due to imprecision as evidenced by a 95% CI that included possible benefit and possible harm. Certainty was not downgraded for risk of bias as limiting the analysis to trials with low risk of bias did not markedly change the findings. There were no concerns regarding inconsistency, indirectness or publication bias.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.3; planned interval between corticosteroid treatments: Analysis 9.3; planned dose of corticosteroids to be given per treatment course: Analysis 10.3; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.3).
8.3. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 3: A3: Neonatal death
9.3. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 3: A3: Neonatal death
10.3. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 3: A3: Neonatal death
11.3. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 3: A3: Neonatal death
Infant death (less than one year of age)
No trials specifically reported infant death before one year of age.
Respiratory distress syndrome
Repeat dose(s) of corticosteroids reduced the risk of RDS compared with placebo or standard care (RR 0.82, 95% CI 0.74 to 0.90; I2 = 25%, Chi2 P = 0.22; 9 trials, 3540 fetuses randomised; NNTB 16, 95% CI 11 to 29; Analysis 1.4).
1.4. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 4: A5: Respiratory distress syndrome
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Wapner 2006), and one trial with high risk of bias (Mazumder 2008), but the result was similar to the overall analysis (RR 0.82, 95% CI 0.75 to 0.91; I2 = 41%, Chi2 P = 0.12; 7 trials, 2870 fetuses randomised).
Certainty of the evidence
The certainty of the evidence was high. Certainty was not downgraded for risk of bias as limiting the analysis to trials with low risk of bias did not markedly change the findings. There were no concerns regarding inconsistency, imprecision, indirectness or publication bias.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.4; planned interval between corticosteroid treatments: Analysis 9.4; planned dose of corticosteroids to be given per treatment course: Analysis 10.4; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.4).
8.4. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 4: A5: Respiratory distress syndrome
9.4. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 4: A5: Respiratory distress syndrome
10.4. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 4: A5: Respiratory distress syndrome
11.4. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 4: A5: Respiratory distress syndrome
Severe respiratory distress syndrome
Repeat dose(s) of prenatal corticosteroid probably had little or no effect on the risk of severe RDS compared with placebo or standard care (RR 0.97, 95% CI 0.82 to 1.16; I2 = 61%, Chi2 P = 0.04; 5 trials, 3809 fetuses randomised; Analysis 1.5).
1.5. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 5: A6: Severe respiratory distress syndrome
Sensitivity analysis
Heterogeneity was not directly explained by differing definitions of severe RDS. Based on prespecified subgroup analyses, heterogeneity may be partially explained by differing weekly drug exposure between trials. Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (Mazumder 2008; Wapner 2006), but the result was similar to the overall analysis (RR 1.00, 95% CI 0.84 to 1.20; I2 = 76%, Chi2 P = 0.02; 3 trials, 3139 fetuses randomised).
Certainty of the evidence
The certainty of the evidence was moderate due to inconsistency as evidenced by high levels of heterogeneity, likely due to differences in the definition of severe RDS across different trials. Certainty was not downgraded for risk of bias as limiting the analysis to trials with low risk of bias did not markedly change the findings. There were no concerns regarding indirectness, imprecision or publication bias.
Subgroup analysis
Prespecified subgroup analysis identified an interaction between severe RDS and planned dose of repeat corticosteroid drug exposure per week (Chi2 = 6.25, P = 0.01; I2 = 84%; Analysis 11.5). Participants in those trials in which the planned dose of repeat corticosteroid was greater than 12 mg per week to 24 mg per week were more likely to experience benefit than participants in trials in which the planned dose was 12 mg per week or less.
11.5. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 5: A6: Severe respiratory distress syndrome
Interaction tests were not significant for other clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.5; planned interval between corticosteroid treatments: Analysis 9.5; planned dose of corticosteroids to be given per treatment course: Analysis 10.5).
8.5. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 5: A6: Severe respiratory distress syndrome
9.5. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 5: A6: Severe respiratory distress syndrome
10.5. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 5: A6: Severe respiratory distress syndrome
Severe lung disease
Repeat dose(s) of prenatal corticosteroids probably resulted in a reduction in the risk of severe lung disease compared with placebo or standard care (RR 0.83, 95% CI 0.72 to 0.97; I2 = 76%, Chi2 P = 0.0009; NNTB 45, 95% CI 27 to 256; 6 trials, 4955 fetuses randomised; Analysis 1.6).
1.6. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 6: A7: Severe lung disease
Sensitivity analysis
Heterogeneity was not explained by differing definitions of severe lung disease. Based on prespecified subgroup analyses, heterogeneity may be partially explained by differing planned number of treatment courses and treatment intervals between trials. Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (Mazumder 2008; Wapner 2006), with a result that was similar to the overall analysis (RR 0.85, 95% CI 0.73 to 0.98; I2 = 85%, Chi2 P = 0.0002; 4 trials, 4285 fetuses randomised).
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded due to inconsistency as evidenced by high levels of heterogeneity, likely due to differences in the definition of severe lung disease across different trials. Certainty was not downgraded for risk of bias as limiting the analysis to trials with low risk of bias did not markedly change the findings. There were no concerns regarding indirectness, imprecision or publication bias.
Subgroup analysis
Prespecified subgroup analysis identified an interaction between severe lung disease and the planned number of repeat courses of corticosteroids (Chi2 = 9.16, P = 0.002, I2 = 89%; Analysis 8.6). Participants in trials in which it was planned give one or more repeat treatment courses were more likely to experience benefit than participants in trials in which it was planned to give only one repeat treatment course.
8.6. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 6: A7: Severe lung disease
There was also an interaction between severe lung disease and the planned interval between corticosteroid treatments (Chi2 = 5.11, P = 0.02, I2 = 80%; Analysis 9.6). Participants in trials that gave the treatment at a minimum interval of seven days were more likely to experience benefit than participants in trials in which the interval was 14 days or greater.
9.6. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 6: A7: Severe lung disease
Interaction tests demonstrated no interactions between other clinical subgroups with available data (planned dose of corticosteroids to be given per treatment course: Analysis 10.6; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.6).
10.6. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 6: A7: Severe lung disease
11.6. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 6: A7: Severe lung disease
Chronic lung disease
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on the risk of chronic lung disease compared with placebo or standard care (RR 1.00, 95% CI 0.83 to 1.22; I2 = 0%, Chi2 P = 0.52; 9 trials, 5661 fetuses randomised; Analysis 1.7).
1.7. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 7: A8: Chronic lung disease
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (McEvoy 2010; Wapner 2006), and one with high risk of bias (Mazumder 2008), with a result that was similar to the overall analysis (RR 1.08, 95% CI 0.88 to 1.33; I2 = 0%, Chi2 P = 0.69; 6 trials, 4878 fetuses randomised).
Certainty of the evidence
The certainty of the evidence was high. Certainty was not downgraded for risk of bias as limiting the analysis to trials with low risk of bias did not markedly change the findings. There were no concerns regarding inconsistency, indirectness, imprecision or publication bias.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.7; planned interval between corticosteroid treatments: Analysis 9.7; planned dose of corticosteroids to be given per treatment course: Analysis 10.7; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.7).
8.7. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 7: A8: Chronic lung disease
9.7. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 7: A8: Chronic lung disease
10.7. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 7: A8: Chronic lung disease
11.7. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 7: A8: Chronic lung disease
Severe intraventricular haemorrhage (grade 3 or 4)
For severe intraventricular haemorrhage, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.13, 95% CI 0.69 to 1.86; I2 = 22%, Chi2 P = 0.27; 7 trials, 5066 fetuses randomised; Analysis 1.8).
1.8. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 8: A9: Severe intraventricular haemorrhage (grade 3/4)
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Peltoniemi 2007), with a result that was similar to the overall analysis (RR 1.06, 95% CI 0.61 to 1.83; I2 = 33%, Chi2 P = 0.20; 6 trials, 4738 fetuses randomised).
Certainty of the evidence
The certainty of the evidence was moderate due to imprecision as the total number of events was low and the 95% CI included both moderate benefit and moderate harm. Certainty was not downgraded for risk of bias as limiting the analysis to trials with low risk of bias did not markedly change the findings. There were no concerns regarding inconsistency, indirectness or publication bias.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.8; planned interval between corticosteroid treatments: Analysis 9.8; planned dose of corticosteroids to be given per treatment course: Analysis 10.8; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.8).
8.8. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 8: A9: Severe Intraventricular haemorrhage (grade 3 or 4)
9.8. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 8: A9: Severe intraventricular haemorrhage (grade 3 or 4)
10.8. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 8: A9: Severe intraventricular haemorrhage (grade 3 or 4)
11.8. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 8: A9: Severe intraventricular haemorrhage (grade 3 or 4)
Intraventricular haemorrhage (any grade)
Repeat dose(s) of prenatal corticosteroids had little or no effect on the risk of intraventricular haemorrhage (any grade) compared with placebo or standard care (RR 0.95, 95% CI 0.75 to 1.19; I2 = 0%, Chi2 P = 0.54; 6 trials, 3223 fetuses randomised; Analysis 1.9).
1.9. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 9: A10: Intraventricular haemorrhage
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (Peltoniemi 2007; Wapner 2006), and one trial with high risk of bias (Mazumder 2008), with a result that was similar to the overall analysis (RR 0.93, 95% CI 0.70 to 1.23; I2 = 0%, Chi2 P = 0.54; 3 trials, 2225 fetuses randomised).
Certainty of the evidence
The certainty of the evidence was high. Certainty was not downgraded for risk of bias as limiting the analysis to studies at low risk of bias did not markedly change the findings. There were no concerns regarding inconsistency, indirectness, imprecision or publication bias.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.9; planned interval between corticosteroid treatments: Analysis 9.9; planned dose of corticosteroids to be given per treatment course: Analysis 10.9; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.9).
8.9. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 9: A10: Intraventricular haemorrhage
9.9. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 9: A10: Intraventricular haemorrhage
10.9. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 9: A10: Intraventricular haemorrhage
11.9. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 9: A10: Intraventricular haemorrhage
Necrotising enterocolitis
For necrotising enterocolitis, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.84, 95% CI 0.59 to 1.22; I2 = 0%, Chi2 P = 0.43; 9 trials, 5736 fetuses randomised; Analysis 1.10).
1.10. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 10: A11: Necrotising enterocolitis
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with some concerns of bias (Wapner 2006), and one trial with high risk of bias (Mazumder 2008), with a result that was similar to the overall analysis (RR 0.86, 95% CI 0.56 to 1.28; I2 = 26%, Chi2 P = 0.24; 7 trials, 5066 fetuses randomised).
Certainty of the evidence
The certainty of the evidence was moderate due to imprecision as the 95% CI included both benefit and possible harm. Certainty was not downgraded for risk of bias as limiting the analysis to trials with low risk of bias did not markedly change the findings. There were no concerns regarding inconsistency, indirectness or publication bias.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.10; planned interval between corticosteroid treatments: Analysis 9.10; planned dose of corticosteroids to be given per treatment course: Analysis 10.10; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.10).
8.10. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 10: A11: Necrotising enterocolitis
9.10. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 10: A11: Necrotising enterocolitis
10.10. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 10: A11: Necrotising enterocolitis
11.10. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 10: A11: Necrotising enterocolitis
Composite serious outcome
Repeat dose(s) of prenatal corticosteroids reduced the risk of the composite of serious outcomes compared with placebo or standard care (RR 0.88, 95% CI 0.80 to 0.97; NNTB 39, 95% CI 24 to 158; I2 = 42%, Chi2 P = 0.09; 9 trials, 5736 fetuses randomised; Analysis 1.11).
1.11. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 11: A12: Composite of serious outcomes
The composite outcome was variously defined by the trialists.
Aghajafari 2002: one or more of: stillborn or neonatal death during the first 28 days of life or before hospital discharge, whichever was sooner; RDS; bronchopulmonary dysplasia (requiring oxygen at 36 corrected postnatal gestational age); grade 3 or 4 intraventricular haemorrhage and necrotising enterocolitis.
Crowther 2006: one or more of: air leak syndrome, patent ductus arteriosus, need for oxygen at 36 weeks' postmenstrual age, severe intraventricular haemorrhage (grade 3 or 4), periventricular leukomalacia, proven necrotising enterocolitis or retinopathy of prematurity.
Garite 2009: one or more of: perinatal death in babies born before 34 weeks' gestation, perinatal death (defined as stillbirth or death before neonatal discharge); RDS (oxygen requirement, clinical diagnosis and consistent chest radiograph); bronchopulmonary dysplasia (defined as a requirement for oxygen at 30 days of age); severe intraventricular haemorrhage (grades 3 or 4); periventricular leukomalacia; blood culture‐proven sepsis; or necrotising enterocolitis.
Guinn 2001: any of: severe RDS, bronchopulmonary dysplasia, severe intraventricular haemorrhage, periventricular leukomalacia, necrotising enterocolitis, proven sepsis or death between randomisation and nursery discharge.
Mazumder 2008: severe RDS or death (or both) within 28 days.
Murphy 2008: one of: stillbirth or neonatal death (defined as death during the first 28 days of life or before hospital discharge); severe RDS (defined as needing assisted ventilation via endotracheal tube and supplemental oxygen within the first 24 hours of life and for 24 hours or more, and either a radiographic scan compatible with RDS or surfactant given between the first two to 24 hours of life); bronchopulmonary dysplasia (defined as needing oxygen at a postmenstrual age of 36 completed weeks and radiographic scan compatible with bronchopulmonary dysplasia); intraventricular haemorrhage grade 3 or 4; cystic periventricular leukomalacia, necrotising enterocolitis.
Peltoniemi 2007: death or RDS or severe intraventricular haemorrhage (expressed as survival without RDS or severe intraventricular haemorrhage during first hospitalisation).
Wapner 2006: one of: severe RDS, intraventricular haemorrhage grade 3 or 4; periventricular leukomalacia, chronic lung disease (defined as the need for supplemental oxygen at 36 weeks' corrected age in infants born before 34 weeks' gestation), or stillbirth or neonatal death.
TEAMS 1999: a composite outcome was reported as part of the PRECISE‐IPD analysis Crowther 2019, which included death (fetal, neonatal or infant), severe respiratory disease, severe intraventricular haemorrhage (grade 3 and 4), chronic lung disease (oxygen dependent at 36 weeks' postnatal age), definite necrotising enterocolitis, severe retinopathy of prematurity (stage 3 or worse in the better eye) or cystic periventricular leukomalacia.
Sensitivity analysis
Heterogeneity was not explained by differing definitions of severe lung disease or the prespecified subgroup analyses. Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (Mazumder 2008; Wapner 2006), with a result that was similar to the overall analysis (RR 0.89, 95% CI 0.80 to 0.98; I2 = 51%, Chi2 P = 0.06; 7 trials, 5066 fetuses randomised).
Certainty of the evidence
The certainty of the evidence was moderate due to inconsistency as there was considerable statistical heterogeneity that may be due to differing definitions of the composite serious outcome between trials. Certainty was not downgraded for risk of bias as limiting the analysis to trials with low risk of bias did not markedly change the findings. There were no concerns regarding imprecision, indirectness or publication bias.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (number of babies in utero: Analysis 7.1; planned number of repeat courses of corticosteroids: Analysis 8.11; planned interval between corticosteroid treatments: Analysis 9.11; planned dose of corticosteroids to be given per treatment course: Analysis 10.11; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.11; gestational age at which the first repeat treatment was given: Analysis 12.1).
7.1. Analysis.

Comparison 7: Subgroup analysis for the number of babies in utero, Outcome 1: A12: Composite serious outcome (variously defined)
8.11. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 11: A12: Composite serious outcome (variously defined)
9.11. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 11: A12: Composite serious outcome (variously defined)
10.11. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 11: A12: Composite serious outcome (variously defined)
11.11. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 11: A12: Composite serious outcome (variously defined)
12.1. Analysis.

Comparison 12: Subgroup analysis for the gestational age at which the first repeat treatment was given, Outcome 1: A12: Composite serious outcome (variously defined)
Primary outcomes for the women
Maternal death
It was unclear if repeat dose(s) of prenatal corticosteroids had an effect on maternal death as there was one death reported for a woman allocated to the placebo group and withdrawn from the trial before study treatment was given (Garite 2009). As there was only one trial, meta‐analysis and sensitivity analysis was not possible (Analysis 2.1).
2.1. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 1: B1: Maternal death
Certainty of the evidence
The certainty of the evidence was low. It was downgraded two levels due to imprecision with only one event occurring and wide 95% CIs included both marked benefit and marked harm. There were no concerns regarding indirectness or inconsistency. Publication bias was unable to be assessed as only one trial reported this outcome.
Maternal sepsis
For maternal sepsis, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.13, 95% CI 0.93 to 1.39; I2 = 23%, Chi2 = 7.77,P = 0.26; 8 trials, 4666 mothers; Analysis 2.2).
2.2. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 2: B2: Maternal sepsis
Sensitivity analysis
All trials included in this analysis were at low risk of bias.
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded for imprecision as the 95% CIs included possible benefit and harm. There were no concerns regarding inconsistency, indirectness or publication bias.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.12; planned interval between corticosteroid treatments: Analysis 9.12; planned dose of corticosteroids to be given per treatment course: Analysis 10.12; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.12).
8.12. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 12: B2: Maternal sepsis
9.12. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 12: B2: Maternal sepsis
10.12. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 12: B2: Maternal sepsis
11.12. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 12: B2: Maternal sepsis
Caesarean section
Repeat dose(s) of prenatal corticosteroids had little or no effect on the risk of caesarean section compared with placebo or standard care (RR 1.03, 95% CI 0.98 to 1.09; I2 = 46%, Chi2 = 13.00,P = 0.07; 8 trials, 4266 mothers; Analysis 2.3)
2.3. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 3: B3: Caesarean section
Sensitivity analysis
Based on prespecified subgroup analyses, heterogeneity may be partially explained by differing planned number of treatment courses between trials. All trials included in this analysis were at low risk of bias.
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded by one level due to inconsistency of effect as evidenced by the degree of statistical heterogeneity. This inconsistency could have been due to differences in local practice or protocols regarding caesarean section. There were no concerns regarding indirectness, imprecision or publication bias.
Subgroup analysis
Prespecified subgroup analysis identified an interaction between caesarean section and the planned number of repeat courses of corticosteroids (Chi2 = 4.59, P = 0.03, I2 = 78%; Analysis 8.13). Participants in trials in which it was planned to give one or more repeat treatment courses had a higher likelihood of caesarean section than participants in trials in which it was planned to give only one repeat treatment course.
8.13. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 13: B3: Caesarean section
Interaction tests demonstrated no differences between the other clinical subgroups with available data (planned interval between corticosteroid treatments: Analysis 9.13; planned dose of corticosteroids to be given per treatment course: Analysis 10.13; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.13).
9.13. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 13: B3: Caesarean section
10.13. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 13: B3: Caesarean section
11.13. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 13: B3: Caesarean section
Discontinuation of therapy because of maternal side effects
It was unclear if repeat dose(s) of prenatal corticosteroids had an effect on discontinuation of therapy due to maternal adverse effects as only one trial reported this outcome and the outcome did not occur in any woman (Guinn 2001). As there was only one trial, 502 women and no events, meta‐analysis was not possible.
Certainty of the evidence
The certainty of the evidence was low. Certainty was downgraded two levels due to imprecision as no events were recorded making it impossible to provide a relative effect estimate. There were no concerns regarding risk of bias, indirectness or inconsistency. Publication bias was unable to be assessed as only one trial reported this outcome.
Adverse effects of corticosteroids
It was unclear if repeat dose(s) of prenatal corticosteroids had an effect on the risk of adverse effects. Two trials reported this outcome with opposite treatment effects and substantial difference in event rates and the definition of the outcome (Crowther 2006; Wapner 2006). Meta‐analysis was not performed as it was not considered appropriate to combine these results with such a marked difference in event rates and direction of effect.
Crowther 2006 defined adverse effects of treatment as any of pain/discomfort, haematoma, maternal distress, rash, sleeplessness, lethargy or other and identified a higher rate of adverse effects in the group who received repeat prenatal corticosteroid (10%) compared with the placebo group (5%) (RR 1.97, 95% CI 1.23 to 3.18; P = 0.005; 982 women).
Wapner 2006 defined any adverse effect of treatment as any of bruising, pain at the injection site, lump at the injection site, gastrointestinal upset, insomnia, contractions or cushingoid appearance and identified a lower rate of adverse effects in the group who received repeat prenatal corticosteroid (27%) compared with the placebo group (56%) (RR 0.49, 95% CI 0.39 to 0.61; P = 0.00001; 495 women).
The differences in adverse effects rates for intervention and placebo groups could be related to differences in placebo preparations. Crowther 2006 used a saline placebo. Wapner 2006 did not specify the placebo preparation
Certainty of the evidence
The certainty of the evidence was low. Certainty was downgraded two levels for marked inconsistency of effect as evidenced by marked heterogeneity in effect size and event rates. There were no concerns regarding risk of bias, imprecision or indirectness. Publication bias did not appear to be likely as there was one positive and one negative study for this outcome.
Admission to the intensive care unit
No trials reported data for admission to the intensive care unit.
Breastfeeding at hospital discharge
No trials reported data for breastfeeding at hospital discharge.
Primary outcomes for the child in early childhood (aged two to less than five years)
Five trials reported data in full for early childhood follow‐up, and we included these in the meta‐analysis (Crowther 2006; Murphy 2008; Peltoniemi 2007; TEAMS 1999; Wapner 2006). The outcome total deaths after randomisation up to early childhood follow‐up used all fetuses randomised as the denominator. Denominators for all other analyses were defined as all babies randomised minus those children for whom there were no outcome data available.
Total deaths (after randomisation)
For risk of death between randomisation and early childhood follow‐up, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.06, 95% CI 0.81 to 1.40; I2 = 0%, Chi2 P = 0.63; 5 trials, 4565 fetuses randomised; Analysis 3.1).
3.1. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 1: C1: Total deaths (after randomisation) up to early childhood follow‐up
Sensitivity analysis
All trials included in this analysis were at low risk of bias.
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded one level due to imprecision as the 95% CIs included possible benefit and harm. There were no concerns regarding imprecision, inconsistency or indirectness. Publication bias is unlikely as four of the five trials of varying size had effect estimates that were close to the no effect line.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.14; planned interval between corticosteroid treatments: Analysis 9.14; planned dose of corticosteroids to be given per treatment course: Analysis 10.14; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.14).
8.14. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 14: C1: Total deaths (after randomisation) up to early childhood follow‐up
9.14. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 14: C1: Total deaths after randomisation up to early childhood follow‐up
10.14. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 14: C1: Total deaths after randomisation up to early childhood follow‐up
11.14. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 14: C1: Total deaths after randomisation up to early childhood follow‐up
Neurodevelopmental impairment at age two to less than five years
Repeat dose(s) of prenatal corticosteroids had little or no effect on risk of neurodevelopmental impairment at early childhood follow‐up compared with placebo (RR 0.97, 95% CI 0.85 to 1.10; I2 = 0%, Chi2 P = 0.87; 4 trials, 3616 children; Analysis 3.2).
3.2. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 2: C2: Neurodevelopmental impairment at early childhood follow‐up
The result from each trial that was used in the meta‐analysis for neurodevelopmental impairment was as follows.
Crowther 2006: "Neurosensory disability" defined as any of cerebral palsy, developmental delay (Mental Development Index greater than one SD below the mean), blindness or deafness.
Murphy 2008: "Neurologic impairment" defined as either of cerebral palsy or cognitive delay (a score of two SD or more below the normative value for Bayley Scales of Infant Development‐II or the local equivalent).
TEAMS 1999: a composite outcome was reported as part of the PRECISE‐IPD analysis Crowther 2019 that included "Neurosensory Disability" defined as any of developmental delay or intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), cerebral palsy, blindness (corrected visual acuity worse than 6/60 in the better eye), deafness (hearing loss requiring amplification or worse).
Wapner 2006: children with Bayley Psychomotor Developmental Index less than 85 (one SD below normative data).
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with high risk of bias (TEAMS 1999), and one with some concerns of risk of bias (Wapner 2006), with a result that was similar to the overall analysis (RR 0.99, 95% CI 0.86 to 1.14; I2 = 0%, Chi2 P = 0.85; 2 trials, 3007 children).
Certainty of the evidence
The certainty of the evidence was high. Certainty was not downgraded due to risk of bias as limiting the analysis to trials with low risk of bias did not change the findings. There were no concerns of indirectness, imprecision or inconsistency. Publication bias was unlikely as the four trials were all clustered close to the no effect line except TEAMS 1999, which had wide 95% CIs.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned interval between corticosteroid treatments: Analysis 9.15; planned dose of corticosteroids to be given per treatment course: Analysis 10.15; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.15).
9.15. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 15: C2: Neurodevelopmental impairment at early childhood follow‐up
10.15. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 15: C2: Neurodevelopmental impairment at early childhood follow‐up
11.15. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 15: C2: Neurodevelopmental impairment at early childhood follow‐up
Survival free of neurodevelopmental impairment at age two to less than five years
Repeat dose(s) of prenatal corticosteroids had little or no effect on survival free of neurodevelopmental impairment at early childhood follow‐up compared with placebo (RR 1.01, 95% CI 0.98 to 1.04; I2 = 0%, Chi2 P = 0.89; 4 trials, 3845 children; Analysis 3.3).
3.3. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 3: C3: Survival free of neurodevelopmental impairment at early childhood follow‐up
Denominators were defined as all babies randomised minus those children for whom there were no outcome data available. The result from each trial that was used in the meta‐analysis for survival free of neurodevelopmental impairment was as follows.
Crowther 2006: total with available data minus those with "death or any neurosensory disability" (defined as any of cerebral palsy, developmental delay (Mental Development Index greater than one SD below the mean), blindness or deafness).
Murphy 2008: total children minus those with the outcome of "Death or neurologic impairment" (defined as either of cerebral palsy or cognitive delay (a score of two SD or more below the normative value for Bayley Scales of Infant Development‐II or the local equivalent)).
TEAMS 1999: total children with available data minus those with the outcome "death or neurosensory disability" reported as part of the PRECISE‐IPD analysis (Crowther 2019), which defined "neurosensory disability" as any of developmental delay or intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), cerebral palsy, blindness (corrected visual acuity worse than 6/60 in the better eye), deafness (hearing loss requiring amplification or worse).
Wapner 2006: total with available data minus those who had died prior to assessment and those children with Bayley Psychomotor Developmental Index less than 85 (one SD below normative data).
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with high risk of bias (TEAMS 1999), and one with some concerns of risk of bias (Wapner 2006), with a result that was similar to the overall analysis (RR 1.00, 95% CI 0.97 to 1.04; I2 = 0%, Chi2 P = 0.71; 2 trials, 3164 children).
Certainty of the evidence
The certainty of the evidence was high. Certainty was not downgraded due to risk of bias as limiting the analysis to trials with low risk of bias did not change the findings. There were no concerns of indirectness, imprecision or inconsistency. Publication bias was unlikely as the four trials were all clustered close to the no effect line.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (number of babies in utero: Analysis 7.2; planned interval between corticosteroid treatments: Analysis 9.16; planned dose of corticosteroids to be given per treatment course: Analysis 10.16; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.16).
7.2. Analysis.

Comparison 7: Subgroup analysis for the number of babies in utero, Outcome 2: C3: Survival free of neurodevelopmental impairment at early childhood follow‐up
9.16. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 16: C3: Survival free of neurodevelopmental impairment at early childhood follow‐up
10.16. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 16: C3: Survival free of neurodevelopmental impairment at early childhood follow‐up
11.16. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 16: C3: Survival free of neurodevelopmental impairment at early childhood follow‐up
Survival free of major neurodevelopmental impairment at age two to less than five years
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on survival free of major neurodevelopmental impairment at early childhood follow‐up compared with placebo (RR 1.02, 95% CI 0.98 to 1.05; I2 = 67%, Chi2 P = 0.05; 3 trials, 1816 children; Analysis 3.4).
3.4. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 4: C4: Survival free of major neurodevelopmental impairment at early childhood follow‐up
The result from each trial that was used in the meta‐analysis for survival free of major neurodevelopmental impairment was as follows.
Crowther 2006: total with available data minus those with "death or moderate to severe neurosensory disability" (defined as any of moderate‐to‐severe cerebral palsy, moderate‐to‐severe developmental delay (Mental Development Index greater than two SDs below the mean), blindness or deafness).
Peltoniemi 2007: survival without "severe neurological, cognitive, or sensory impairment" (defined as survival without cerebral palsy, Mental Developmental Index less than 70, developmental quotient less than 70, deafness or blindness).
Wapner 2006: total with available data minus those who had died prior to assessment and those children with Bayley Psychomotor Developmental Index less than 70 (two SDs below normative data).
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with high risk of bias (Peltoniemi 2007) and one with some concerns of risk of bias (Wapner 2006) which left only one trial at low risk of bias, with a result that was similar to the overall analysis (RR 1.04, 95% CI 0.99 to 1.10; 1060 children). Sensitivity analysis was not performed for heterogeneity as there were only three trials in this analysis.
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded due to inconsistency as evidenced by the high level of statistical heterogeneity. Certainty was not downgraded due to risk of bias as limiting the analysis to the one trial with low risk of bias did not change the findings. There were no concerns of indirectness or imprecision. Publication bias was unlikely as the three trials were all clustered close to the no effect line.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned number of repeat courses of corticosteroids: Analysis 8.15; planned dose of corticosteroids to be given per treatment course: Analysis 10.17; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.17).
8.15. Analysis.

Comparison 8: Subgroup analysis for the planned number of repeat courses of corticosteroids to be given, Outcome 15: C4: Survival free of major neurodevelopmental impairment at early childhood follow‐up
10.17. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 17: C4: Survival free of major neurodevelopmental impairment at early childhood follow‐up
11.17. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 17: C4: Survival free of major neurodevelopmental impairment at early childhood follow‐up
Cerebral palsy
For cerebral palsy at early childhood follow‐up, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.03, 95% CI 0.71 to 1.49; I2 = 0%, Chi2 P = 0.49; 5 trials, 3923 children; Analysis 3.5). There were too few children with cerebral palsy to analyse the subgroups of severity of cerebral palsy (mild, moderate and severe).
3.5. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 5: C5: Cerebral palsy at early childhood follow‐up
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of two trials with high risk of bias (Peltoniemi 2007; TEAMS 1999), with a result that was similar to the overall analysis (RR 1.01, 95% CI 0.69 to 1.47; I2 = 31%, Chi2 P = 0.24; 3 trials, 3541 children).
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded due to imprecision as evidenced by wide 95% CIs that included both benefit and harm. Certainty was not downgraded due to risk of bias as limiting the analysis to trials with low risk of bias did not change the result. There were no concerns of indirectness or inconsistency. Publication bias was unlikely as the five trials were all clustered close to the no effect line except Wapner 2006, which had very wide 95% CIs that included both benefit and marked harm.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned interval between corticosteroid treatments: Analysis 9.17; planned dose of corticosteroids to be given per treatment course: Analysis 10.18; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.18).
9.17. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 17: C5: Cerebral palsy at early childhood follow‐up
10.18. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 18: C5: Cerebral palsy at early childhood follow‐up
11.18. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 18: C5: Cerebral palsy at early childhood follow‐up
Developmental delay or intellectual impairment at age two to less than five years
Repeat dose(s) of prenatal corticosteroids had little or no effect on developmental delay at early childhood follow‐up compared with placebo (RR 0.95, 95% CI 0.84 to 1.09; I2 = 0%, Chi2 P = 0.86; 4 trials, 3581 children; Analysis 3.6).
3.6. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 6: C6: Developmental delay or intellectual impairment at early childhood follow‐up (any)
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with high risk of bias (Peltoniemi 2007), and one with some concerns of risk of bias (Wapner 2006), with a result that was similar to the overall analysis (RR 0.98, 95% CI 0.85 to 1.14; I2 = 0%, Chi2 P = 0.93; 2 trials, 2900 children).
Certainty of the evidence
The certainty of the evidence was high. Certainty was not downgraded due to risk of bias as limiting the analysis to trials with low risk of bias did not change the findings. There were no concerns of indirectness, imprecision or inconsistency. Publication bias was unlikely as the four trials were all clustered close to the no effect line.
Mental Developmental Index at age two to less than five years
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean Mental Developmental Index score at early childhood follow‐up compared with placebo (MD 0.89, 95% CI −0.61 to 2.39; I2 = 0%, Chi2 P = 0.46; 3 trials, 1627 children; Analysis 3.7).
3.7. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 7: Mental Developmental Index at early childhood follow‐up
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with high risk of bias (Peltoniemi 2007), and one with some concerns of risk of bias (Wapner 2006), with a result that was similar to the overall analysis (MD 0.60, 95% CI −1.59 to 2.79; 1 trial, 967 children).
Certainty of the evidence
The certainty of the evidence was high. Certainty was not downgraded due to risk of bias as limiting the analysis to trials with low risk of bias did not change the findings. There were no concerns of indirectness, imprecision, inconsistency or publication bias.
Subgroup analysis
Interaction tests demonstrated no differences between clinical subgroups with available data (planned interval between corticosteroid treatments: Analysis 9.18; planned dose of corticosteroids to be given per treatment course: Analysis 10.19; planned dose of repeat corticosteroid drug exposure per week: Analysis 11.19).
9.18. Analysis.

Comparison 9: Subgroup analysis for planned interval between corticosteroid treatments, Outcome 18: C6: Developmental delay or intellectual impairment at early childhood follow‐up (any)
10.19. Analysis.

Comparison 10: Subgroup analysis for the planned dose of corticosteroid given per treatment , Outcome 19: C6: Developmental delay or intellectual impairment at early childhood follow‐up (any)
11.19. Analysis.

Comparison 11: Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week, Outcome 19: C6: Developmental delay or intellectual impairment at early childhood follow‐up (any)
Primary outcomes for the child in mid‐ to late childhood (aged five to less than 18 years)
Two trials reported data for children in mid‐ to late childhood (Crowther 2006; Murphy 2008). The outcome total deaths after randomisation up to mid‐ to late childhood follow‐up used all fetuses randomised as the denominator. Denominators for all other analyses were defined as all babies randomised minus those children for whom there was no outcome data available.
Total deaths (after randomisation)
For risk of death between randomisation and mid‐ to late childhood follow‐up, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.93, 95% CI 0.69 to 1.26; I2 = 0%, Chi2 P = 0.84; 2 trials, 2874 fetuses randomised; Analysis 4.1).
4.1. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 1: D1: Total deaths (after randomisation) up to mid‐ to later childhood follow‐up
Sensitivity analysis
Both trials included in this analysis were at low risk of bias.
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded one level for imprecision due to wide 95% CIs that included benefit and possible harm. There were no concerns of indirectness or inconsistency. Publication bias was not formally assessed as there were only two trials and both had effect estimates close to the no effect line.
Neurocognitive impairment at age five to less than 18 years
For neurocognitive impairment at mid‐ to late childhood follow‐up, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.96, 95% CI 0.72 to 1.29; 1 trial, 897 children; Analysis 4.2).
4.2. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 2: D2: Neurocognitive impairment at mid‐ to later childhood follow‐up
Data came from the result "neurosensory disability", which was defined as any of cerebral palsy, intellectual quotient greater than one SD below the normative mean, blindness or deafness (Crowther 2006).
Sensitivity analysis
No sensitivity analysis was performed as only one trial reported this outcome.
Certainty of the evidence
The certainty of the evidence was low. Certainty was downgraded one level due to risk of bias as the single included trial had some concerns of risk of bias due to missing outcome data. Certainty was downgraded one further level for imprecision as the 95% CIs included both benefit and harm. There were no concerns of indirectness or inconsistency. Publication bias was not formally assessed as there was only one trial in this analysis.
Survival free of neurocognitive impairment at age five to less than 18 years
Repeat dose(s) of prenatal corticosteroids had little or no effect on survival free of neurocognitive impairment at mid‐ to late childhood follow‐up compared with placebo (RR 1.01, 95% CI 0.95 to 1.08; 1 trial, 963 children; Analysis 4.3).
4.3. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 3: D3: Survival free of neurocognitive impairment at mid‐ to later childhood follow‐up
Data came from the result "survival free of neurosensory disability" in which neurosensory disability was defined as any of cerebral palsy, intellectual quotient greater than one SD below the normative mean, blindness or deafness (Crowther 2006).
Sensitivity analysis
No sensitivity analysis was performed as only one trial reported this outcome.
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded one level due to risk of bias as the single included trial had some concerns of risk of bias due to missing outcome data. There were no concerns of indirectness, imprecision or inconsistency. Publication bias was not formally assessed as there was only one trial in this analysis.
Survival free of major neurocognitive impairment at age five to less than 18 years
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on survival free of major neurocognitive impairment at mid‐ to late childhood follow‐up compared with placebo (RR 1.00, 95% CI 0.97 to 1.04; I2 = 0%, Chi2 P = 0.69; 2 trials, 2682 children; Analysis 4.4).
4.4. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 4: D4: Survival free of major neurocognitive impairment at mid‐ to later childhood follow‐up
In the two trials that reported data, major neurocognitive impairment was defined as follows.
Crowther 2006: any of blindness, severe cerebral palsy, or severe intellectual impairment (intellectual quotient less than −3 SD).
Murphy 2008: "Severe disability", defined as any of neuromotor (non‐ambulatory cerebral palsy), neurosensory (blindness or deafness or need for visual or hearing aids), or neurocognitive disability (abnormal attention, memory or behaviour assessed by an abnormally elevated score (more than 1.5 SD greater than normative control sample) on either the Behaviour Rating Inventory of Executive Function – Preschool version and the Child Behaviour Checklist – 1.5–5).
Sensitivity analysis
Both trials included in this analysis were judged to have some concerns of risk of bias.
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded one level due to risk of bias as the included trials were judged to have some concerns of risk of bias due to missing outcome data. There were no concerns of indirectness, imprecision or inconsistency. Publication bias was not formally assessed as there were only two trials and both had effect estimates close to the no effect line.
Motor impairment at age five to less than 18 years
Neither of the trials reporting data for follow‐up at mid‐ to late childhood reported data for motor impairment. Murphy 2008 reported "neuromotor disability", which was classified as non‐ambulatory cerebral palsy and is included under the outcome section 'Cerebral palsy'.
Cognitive impairment at age five to less than 18 years
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on cognitive impairment at mid‐ to late childhood follow‐up compared with placebo (RR 1.00, 95% CI 0.81 to 1.22; I2 = 0%, Chi2 P = 0.59; 2 trials, 2504 children; Analysis 4.5).
4.5. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 5: D6: Cognitive impairment at mid‐ to later childhood follow‐up
Sensitivity analysis
Both trials included in this analysis were judged to have some concerns of risk of bias.
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded one level due to risk of bias as the included trials were judged to have some concerns of risk of bias due to missing outcome data. There were no concerns of indirectness, imprecision or inconsistency. Publication bias was not formally assessed as there were only two trials and both had effect estimates close to the no effect line.
Educational achievement at age five to less than 18 years
Only one trial reported data for educational achievement at mid‐ to late childhood (Crowther 2006). Educational achievement was reported using the Wide Range Achievement Test 4 for spelling, mathematics and reading standardised to a mean of 100 and SD of 15. The authors reported little or no difference between groups (spelling: MD −0.9, 95% CI −3.4 to 1.6; mathematics: MD 0.1, 95% CI −2.3 to 2.4; reading: MD −0.2, 95% CI −2.7 to 2.4).
Sensitivity analysis
No sensitivity analysis was performed as only one trial reported this outcome.
Certainty of the evidence
The certainty of the evidence was moderate. Certainty was downgraded one level due to risk of bias as the included trial was judged to have some concerns of risk of bias due to missing outcome data. There were no concerns of indirectness, imprecision or inconsistency. Publication bias was not formally assessed as there was only one trial that assessed this outcome.
Cerebral palsy
For cerebral palsy, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo (RR 0.74, 95% CI 0.43 to 1.26; I2 = 55%, Chi2 P = 0.13; 2 trials, 2622 children; Analysis 4.6).
4.6. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 6: D8: Cerebral palsy at mid‐ to later childhood follow‐up
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias due to missing data (Murphy 2008). The result from the remaining trial was similar to the overall analysis but with greater imprecision (RR 0.95, 95% CI 0.51 to 1.76; 987 children). Sensitivity analysis was not performed for heterogeneity as there were only two trials in this analysis.
Certainty of the evidence
The certainty of the evidence was low. Certainty was downgraded one level due to imprecision as the 95% CI included marked benefit and harm and the total number of events was low. It was downgraded one further level due to inconsistency as evidenced by the marked statistical heterogeneity. Heterogeneity may have been related to differing classifications of cerebral palsy as Crowther 2006 reported data for any cerebral palsy whereas Murphy 2008 reported non‐ambulatory cerebral palsy. Evidence was not downgraded due to risk of bias as limiting the analysis to only the trial with low risk of bias did not change the findings. There were no concerns of indirectness. Publication bias was not formally assessed as there were only two trials and both had effect estimates crossing the no effect line.
Hypertension
No trials reported hypertension. Crowther 2006 reported that the proportion of children in the pre‐hypertensive range (greater than 90th centile for systolic or diastolic blood pressure) was not different between the repeat doses of corticosteroids group and the placebo group (RR 1.07, 95% CI 0.64 to 1.80; 848 children).
Primary outcomes for the child as an adult (aged 18 years or greater)
No trials reported data for follow‐up into adulthood.
Secondary outcomes for the fetus/neonate/infant
Birthweight
Mean birthweight
Repeat dose(s) of prenatal corticosteroids resulted in a small reduction in mean birthweight compared with placebo or standard care (MD −74 g, 95% CI −116 to −33; I2 = 0%, Chi2 P = 0.82; 10 trials, 5808 infants; Analysis 1.38; high‐certainty evidence; Table 5).
1.38. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 38: L1: Mean birthweight (g)
Sensitivity analysis
Restricting the analysis to only those trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Mazumder 2008), with a result that was similar to the overall analysis (MD −73 g, 95% CI −116 to −32; I2 = 0%, Chi2 P = 0.74; 9 trials, 5734 infants).
Birthweight adjusted for gestational age
Repeat dose(s) of prenatal corticosteroids also resulted in a small reduction in birthweight adjusted for gestational age compared with placebo or standard care (SMD −0.15, 95% CI −0.23 to −0.06; I2 = 0%, Chi2 P = 0.64; 4 trials, 2028 infants; Analysis 1.41; high‐certainty evidence; Table 5). Data for this analysis were reported as birthweight Z scores for three trials (Crowther 2006; McEvoy 2010; TEAMS 1999), and multiples of the median for one trial (Wapner 2006).
1.41. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 41: L1: Birthweight adjusted for gestation (standardised mean difference)
Small‐for‐gestational age
Repeat dose(s) of prenatal corticosteroids resulted in an increase in infants born small‐for‐gestational age compared with placebo or standard care (RR 1.25, 95% CI 1.08 to 1.44; NNTH 29, 95% CI 16 to 90; I2 = 0%, Chi2 P = 0.69; 7 trials, 4013 fetuses randomised; Analysis 1.17; high‐certainty evidence; Table 5).
1.17. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 17: F2: Small‐for‐gestational age at birth
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of two trials at some concerns of risk of bias (Mazumder 2008; Wapner 2006), with a result similar to the overall analysis (RR 1.20, 95% CI 1.03 to 1.41; I2 = 0%, Chi2 P = 0.71; 5 trials, 3343 fetuses randomised).
Length at birth
Repeat dose(s) of prenatal corticosteroids resulted in a small reduction in mean length at birth compared with placebo or standard care (MD −0.6 cm, 95% CI −0.9 to −0.2; I2 = 0%, Chi2 P = 0.47; 6 trials, 4550 infants; Analysis 1.45; high‐certainty evidence; Table 5).
1.45. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 45: L4: Mean length at birth (cm)
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial judged at some concerns of risk of bias (Mazumder 2008), with a result that was similar to the overall analysis (MD −0.6 cm, 95% CI −0.9 to −0.2; I2 = 11%, Chi2 P = 0.34; 5 trials, 4476 infants).
Repeat dose(s) of prenatal corticosteroids resulted in little or no change in length at birth adjusted for gestational age compared with placebo or standard care (SMD −0.09, 95% CI −0.18 to 0.01; I2 = 0%, Chi2 P = 0.37; 3 trials, 1846 infants; Analysis 1.48; high‐certainty evidence; Table 5). Data for this analysis were reported as birthweight Z scores for two trials (Crowther 2006; McEvoy 2010) and multiples of the median for one trial (Wapner 2006).
1.48. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 48: L4: Length at birth adjusted for gestation (standardised mean difference)
Head circumference at birth
Mean head circumference at birth
Repeat dose(s) of prenatal corticosteroids resulted in a small reduction in mean head circumference at birth compared with placebo or standard care (MD −0.3 cm, 95% CI −0.5 to −0.2; I2 = 16%, Chi2 P = 0.30; 10 trials, 5731 infants; Analysis 1.43; high‐certainty evidence; Table 5).
1.43. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 43: L3: Mean head circumference at birth (cm)
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial judged at some concerns of risk of bias (Mazumder 2008) and one trial at high risk of bias (TEAMS 1999), with a result that was similar to the overall analysis (MD −0.3 cm, 95% CI −0.5 to −0.2; I2 = 30%, Chi2 P = 0.18; 8 trials, 5568 infants).
Mean head circumference at birth adjusted for gestational age
Repeat dose(s) of prenatal corticosteroids resulted in little or no change in mean head circumference at birth adjusted for gestational age compared with placebo or standard care (MD −0.14, 95% CI −0.27 to 0.00; I2 = 0%, Chi2 P = 0.43; 2 trials, 1251 infants; Analysis 1.44; high‐certainty evidence; Table 5).
1.44. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 44: L3: Mean head circumference Z score at birth
Growth assessments at primary hospital discharge
Mean weight at primary hospital discharge
Repeat dose(s) of prenatal corticosteroids had little or no effect on mean weight at primary hospital discharge compared with placebo or standard care (MD 18 g, 95% CI −52 to 88; I2 = 37%, Chi2 P = 0.21; 2 trials, 1195 infants; Analysis 1.49; high‐certainty evidence; Table 5).
1.49. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 49: L5i: Mean weight (g) at primary hospital discharge
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (McEvoy 2010), with a result that was similar to the overall analysis (MD −1 g, 95% CI −77 to 75; 1 trial, 1090 infants). Sensitivity analysis was not performed for heterogeneity as there were only two trials in this analysis.
Mean weight Z score at primary hospital discharge
Repeat dose(s) of prenatal corticosteroids had little or no effect on mean weight Z score at primary hospital discharge compared with placebo or standard care (MD −0.05, 95% CI −0.16 to 0.06; I2 = 0%, Chi2 P = 0.92; 2 trials, 1195 infants; Analysis 1.50; high‐certainty evidence; Table 5).
1.50. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 50: L5i: Mean weight Z score at primary hospital discharge
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (McEvoy 2010), with a result that was similar to the overall analysis (MD −0.05, 95% CI −0.16 to 0.06; 1 trial, 1090 infants).
Mean length at primary hospital discharge
Repeat dose(s) of prenatal corticosteroids had little or no effect on mean length at primary hospital discharge compared with placebo or standard care (MD 0.0 cm, 95% CI −0.4 to 0.5; I2 = 0%, Chi2 P = 0.87; 2 trials, 1189 infants; Analysis 1.53; high‐certainty evidence; Table 5).
1.53. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 53: L5iii: Mean length (cm) at primary hospital discharge
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (McEvoy 2010), with a result that was similar to the overall analysis (MD 0.0, 95% CI −0.5 to 0.5; 1 trial, 1090 infants).
Mean length Z score at primary hospital discharge
Repeat dose(s) of prenatal corticosteroids had little or no effect on mean length Z score at primary hospital discharge compared with placebo or standard care (MD −0.06, 95% CI −0.23 to 0.10; I2 = 0%, Chi2 P = 0.34; 2 trials, 1189 infants; Analysis 1.54; high‐certainty evidence; Table 5).
1.54. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 54: L5iii: Mean length Z score at primary hospital discharge
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (McEvoy 2010), with a result that was similar to the overall analysis (MD −0.03, 95% CI −0.21 to 0.15; 1 trial, 1090 infants).
Mean head circumference at primary hospital discharge
Repeat dose(s) of prenatal corticosteroids had little or no effect on mean head circumference at primary hospital discharge compared with placebo or standard care (MD 0.1 cm, 95% CI −0.1 to 0.4; I2 = 0%, Chi2 P = 0.56; 2 trials; 1195 infants; Analysis 1.51; high‐certainty evidence; Table 5).
1.51. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 51: L5ii: Mean head circumference (cm) at primary hospital discharge
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (McEvoy 2010), with a result that was similar to the overall analysis (MD 0.0, 95% CI −0.2 to 0.2; 1 trial, 1090 infants).
Mean head circumference Z score at primary hospital discharge
Repeat dose(s) of prenatal corticosteroids had little or no effect on mean head circumference Z score at primary hospital discharge compared with placebo or standard care (MD −0.03 cm, 95% CI −0.15 to 0.10; I2 = 0%, Chi2 P = 0.53; 2 trials, 1195 infants; Analysis 1.52; high‐certainty evidence; Table 5).
1.52. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 52: L5ii: Mean head circumference Z score at primary hospital discharge
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (McEvoy 2010), with a result that was similar to the overall analysis (MD −0.04, 95% CI −0.17 to 0.09; 1 trial, 1090 infants).
Growth assessments at infant follow‐up
Mean weight at infant follow‐up
Repeat dose(s) of prenatal corticosteroids possibly caused a small reduction in mean weight at infant follow‐up compared with placebo or standard care, but the evidence was very uncertain (MD −0.60 kg, 95% CI −0.87 to −0.34; I2 = 23%, Chi2 P = 0.25; 2 trials, 149 infants; Analysis 1.55; very low‐certainty evidence; Table 5).
1.55. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 55: L6i: Mean weight at infant follow‐up (kg)
Mean weight Z score at infant follow‐up
Repeat dose(s) of prenatal corticosteroids possibly had little or no effect on mean weight Z score at infant follow‐up compared with placebo or standard care, but the evidence was very uncertain (MD −0.12, 95% CI −0.84 to 0.6; 1 trial, 75 infants; Analysis 1.56; very low‐certainty evidence; Table 5).
1.56. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 56: L6i: Mean weight Z score at infant follow‐up
Mean length at infant follow‐up
Repeat dose(s) of prenatal corticosteroids possibly decreased mean length at infant follow‐up compared with placebo or standard care, but the evidence was very uncertain (MD −1.6 cm, 95% CI −2.3 to −0.8; I2 = 0%, Chi2 P = 0.59; 2 trials, 149 infants; Analysis 1.59; very low‐certainty evidence; Table 5).
1.59. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 59: L6iii: Mean length at infant follow‐up (cm)
Mean length Z score at infant follow‐up
Repeat dose(s) of prenatal corticosteroids possibly had little or no effect on mean length Z score at infant follow‐up compared with placebo or standard care, but the evidence was very uncertain (MD −0.24, 95% CI −1.21 to 0.73; 1 trial, 75 infants; Analysis 1.60; very low‐certainty evidence; Table 5).
1.60. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 60: L6iii: Mean length Z score at infant follow‐up
Mean head circumference at infant follow‐up
Repeat dose(s) of prenatal corticosteroids possibly decreased mean head circumference at infant follow‐up compared with placebo or standard care, but the evidence was very uncertain (MD −0.5 cm, 95% CI −0.9 to −0.1; I2 = 0%, Chi2 P = 0.59; 2 trials, 136 infants; Analysis 1.57; very low‐certainty evidence; Table 5).
1.57. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 57: L6ii: Mean head circumference at infant follow‐up (cm)
Mean head circumference Z score at infant follow‐up
Repeat dose(s) of prenatal corticosteroids possibly had little or no effect on mean head circumference Z scoreat infant follow‐up compared with placebo or standard care, but the evidence was very uncertain (MD −0.04, 95% CI −0.78 to 0.70; 1 trial, 62 infants; Analysis 1.58; very low‐certainty evidence; Table 5).
1.58. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 58: L6ii: Mean head circumference Z score at infant follow‐up
Gestational age at birth
Term birth
Repeat dose(s) of prenatal corticosteroids possibly had little or no effect of the likelihood of term birth compared with placebo or standard care (RR 0.96, 95% CI 0.86 to 1.06; I2 = 43%, Chi2 P = 0.12; 7 trials, 4068 fetuses randomised; Analysis 1.12; low‐certainty evidence; Table 5).
1.12. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 12: F1i: Term birth ≥ 37 weeks
Sensitivity analysis
Subgroup analysis based on prespecified subgroups suggests that some of the heterogeneity may be related to different planned numbers of treatment courses. Different gestational ages at trial entry could have contributed to heterogeneity but this was not directly assessed in sensitivity analysis.
Preterm birth
Repeat dose(s) of prenatal corticosteroids possibly had little or no effect of the likelihood of preterm birth compared with placebo or standard care (RR 1.02, 95% CI 0.98 to 1.05; I2 = 60%, Chi2 P = 0.02; 7 trials, 4068 fetuses randomised; Analysis 1.13; low‐certainty evidence; Table 5).
1.13. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 13: F1ii: Preterm birth before 37 weeks
Sensitivity analysis
Subgroup analysis based on prespecified subgroups suggests that some of the heterogeneity may be related to different planned numbers of treatment courses and the dose given per treatment course. Different gestational ages at trial entry could have contributed to heterogeneity but this was not directly assessed in sensitivity analysis.
Very preterm birth
Repeat dose(s) of prenatal corticosteroids probably had little or no effect of the likelihood of very preterm birth less than 34 weeks compared with placebo or standard care (RR 1.02, 95% CI 0.97 to 1.08; I2 = 0%, Chi2 P = 0.75; 6 trials, 2682 fetuses randomised; Analysis 1.14; moderate‐certainty evidence; Table 5).
1.14. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 14: F1iii: Very preterm birth before 34 weeks
Extremely preterm birth
For extremely preterm birth less than 28 weeks, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.13, 95% CI 0.92 to 1.38; I2 = 0%, Chi2 P = 0.87; 5 trials, 4022 fetuses randomised; Analysis 1.15; low‐certainty evidence; Table 5).
1.15. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 15: F1iv: Extremely preterm birth before 28 weeks
Mean gestational age at birth
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean gestational age at birth compared with placebo or standard care (MD −0.18 weeks, 95% CI −0.37 to 0.01; I2 = 0%, Chi2 P = 0.46; 10 trials, fetuses randomised; Analysis 1.16; moderate‐certainty evidence; Table 5). The funnel plot to investigate publication bias was symmetrical, with the exception of one small study showing a negative effect (Figure 5).
1.16. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 16: F1v: Mean gestational age at birth (weeks)
5.

Interval between trial entry and birth
Repeat dose(s) of prenatal corticosteroids probably resulted in a small reduction in the interval between trial entry and birth compared with placebo or standard care (MD −0.71 days, 95% CI −1.36 to −0.06; I2 = 0%, Chi2 P = 0.75; 4 trials, 1309 fetuses randomised; Analysis 1.42; moderate‐certainty evidence; Table 5).
1.42. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 42: L2: Interval between trial entry and birth (days)
Admission to neonatal intensive care unit
Repeat dose(s) of prenatal corticosteroids had little or no effect on risk of admission to the NICU compared with placebo or standard care (RR 1.01, 95% CI 0.95 to 1.07; I2 = 0%, Chi2 P = 0.36; 2 trials, 3455 fetuses randomised; Analysis 1.18; high‐certainty evidence; Table 5).
1.18. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 18: F3: Admission to the neonatal intensive care unit
Proven neonatal infection while in the neonatal intensive care unit
Repeat dose(s) of prenatal corticosteroids had little or no effect on risk of proven neonatal infection while in the NICU compared with placebo or standard care (RR 1.03, 95% CI 0.86 to 1.22; I2 = 0%, Chi2 P = 0.56; 8 trials, 5660 fetuses randomised; Analysis 1.19; high‐certainty evidence; Table 5).
1.19. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 19: F4: Proven neonatal infection while in the neonatal intensive care unit
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (TEAMS 1999; Wapner 2006), with a result that was similar to the overall analysis (RR 1.07, 95% CI 0.89 to 1.28; I2 = 0%, Chi2 P = 0.60; 6 trials, 4878 fetuses randomised).
Early systemic neonatal infection
Repeat dose(s) of prenatal corticosteroids had little or no effect on risk of early systemic neonatal infection compared with placebo or standard care (RR 0.93, 95% CI 0.79 to 1.11; I2 = 0%, Chi2 P = 0.80; 4 trials, 1738 fetuses randomised; Analysis 1.20; high‐certainty evidence; Table 5).
1.20. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 20: F5: Early systemic neonatal infection
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial at high risk of bias (Mazumder 2008), with a result that was similar to the overall analysis (RR 0.93, 95% CI 0.78 to 1.10; I2 = 0%, Chi2 P = 0.69; 3 trials, 1662 fetuses randomised).
Late systemic neonatal infection
For late systemic neonatal infection, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.37, 95% CI 0.78 to 2.41; I2 = 0%, Chi2 P = 0.38; 2 trials, 404 fetuses randomised; Analysis 1.21; low‐certainty evidence; Table 5).
1.21. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 21: F6: Late systemic neonatal infection
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with high risk of bias (Mazumder 2008), but did not markedly change the findings (RR 1.51, 95% CI 0.83 to 2.75; 1 trial, 328 fetuses randomised).
Retinopathy of prematurity
For retinopathy of prematurity, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.01, 95% CI 0.81 to 1.27; I2 = 0%, Chi2 P = 0.85; 8 trials, 5234 fetuses randomised; Analysis 1.22; moderate‐certainty evidence; Table 5).
1.22. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 22: F7: Retinopathy of prematurity
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (Murphy 2008; Wapner 2006) and one trial with high risk of bias (Mazumder 2008), but did not markedly change the results of the analysis (RR 0.95, 95% CI 0.71 to 1.27; I2 = 0%, Chi2 P = 0.94; 5 trials, 2255 fetuses randomised).
Periventricular leukomalacia
For periventricular leukomalacia, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.75, 95% CI 0.43 to 1.31; I2 = 0%, Chi2 P = 0.77; 8 trials, 5142 fetuses randomised; Analysis 1.23; moderate‐certainty evidence; Table 5).
1.23. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 23: F8: Periventricular leukomalacia
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Peltoniemi 2007) and one at high risk of bias (Mazumder 2008), but did not markedly change the findings (RR 0.67, 95% CI 0.38 to 1.20; I2 = 0%, Chi2 P = 0.93; 6 trials, 4738 fetuses randomised).
Neonatal encephalopathy
For neonatal encephalopathy, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.50, 95% CI 0.10 to 2.57; 1 trial, 76 fetuses randomised; Analysis 1.24; very low‐certainty evidence; Table 5).
1.24. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 24: F9: Neonatal encephalopathy
Patent ductus arteriosus
Repeat dose(s) of prenatal corticosteroids possibly reduced the risk of patent ductus arteriosus compared with placebo or standard care (RR 0.78, 95% CI 0.63 to 0.96; I2 = 30%, Chi2 P = 0.20; 7 trials, 4657 fetuses randomised; Analysis 1.25; low‐certainty evidence; Table 5).
1.25. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 25: F10: Patent ductus arteriosus
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Wapner 2006) and one trial with high risk of bias (Mazumder 2008), and moved the CIs of the effect estimate past the no effect line (RR 0.81, 95% CI 0.65 to 1.01; I2 = 40%, Chi2 P = 0.15; 5 trials, 3987 fetuses randomised).
Use of respiratory support
Repeat dose(s) of prenatal corticosteroids resulted in a small reduction in the use of respiratory support compared with placebo or standard care (RR 0.88, 95% CI 0.80 to 0.97; I2 = 5%, Chi2 P = 0.30; 2 trials, 2497 fetuses randomised; Analysis 1.26; high‐certainty evidence; Table 5).
1.26. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 26: F11: Use of respiratory support
Duration of respiratory support
No trials reported data for duration of respiratory support.
Use of invasive respiratory support
Repeat dose(s) of prenatal corticosteroids probably resulted in a small reduction in the use of invasive respiratory support compared with placebo or standard care (RR 0.86, 95% CI 0.80 to 0.93; I2 = 60%, Chi2 P = 0.03; 6 trials, 5067 fetuses randomised; Analysis 1.27; moderate‐certainty evidence; Table 5).
1.27. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 27: F12: Use of invasive respiratory support
Sensitivity analysis
Based on prespecified subgroup analyses, some of the heterogeneity may be associated with differences in the planned dose of corticosteroid drug exposure per week. Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Wapner 2006), with a result that was similar to the overall analysis (RR 0.88, 95% CI 0.82 to 0.95; I2 = 51%, Chi2 P = 0.09; 5 trials, 4473 fetuses randomised).
Duration of invasive respiratory support
Repeat dose(s) of prenatal corticosteroids probably slightly reduced the duration of invasive respiratory support compared with placebo or standard care (MD −0.27 days, 95% CI −0.38 to −0.16; I2 = 47%, Chi2 P = 0.13; 4 trials, 1620 neonates; Analysis 1.61; moderate‐certainty evidence; Table 5).
1.61. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 61: L8: Mean duration of invasive respiratory support (days)
Sensitivity analysis
Prespecified subgroup analyses did not appear to explain the observed heterogeneity.
Use of non‐invasive respiratory support
Repeat dose(s) of prenatal corticosteroids had little or no effect on the use of non‐invasive respiratory support compared with placebo or standard care (RR 0.91, 95% CI 0.82 to 1.01; I2 = 13%, Chi2 P = 0.32; 3 trials, 3231 fetuses randomised; Analysis 1.28; high‐certainty evidence; Table 5).
1.28. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 28: F13: Use of non‐invasive respiratory support
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Wapner 2006), with a result that was similar to the overall analysis (RR 0.94, 95% CI 0.84 to 1.05; I2 = 0%, Chi2 P = 0.64; 2 trials, 2637 fetuses randomised).
Duration of non‐invasive respiratory support
For duration of non‐invasive respiratory support, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (MD −0.59 days, 95% CI −1.29 to 0.11; 1 trial, 326 neonates; Analysis 1.62; low‐certainty evidence; Table 5).
1.62. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 62: L9: Mean duration of non‐invasive respiratory support (days)
Use of oxygen supplementation
Repeat dose(s) of prenatal corticosteroids resulted in a small reduction in the use of oxygen supplementation compared with placebo or standard care (RR 0.91, 95% CI 0.85 to 0.98; I2 = 9%, Chi2 P = 0.33; 3 trials, 3643 fetuses randomised; Analysis 1.29; high‐certainty evidence; Table 5).
1.29. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 29: F14: Use of oxygen supplementation
Duration of oxygen supplementation
For duration of oxygen supplementation, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (MD −0.32 days, 95% CI −0.94 to 0.30; I2 = 14%, Chi2 P = 0.32; 4 trials, 1619 fetuses randomised; Analysis 1.63; moderate‐certainty evidence; Table 5).
1.63. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 63: L10: Mean duration of oxygen supplementation (days)
Use of surfactant
Repeat dose(s) of prenatal corticosteroids probably reduced the use of surfactant compared with placebo or standard care (RR 0.80, 95% CI 0.73 to 0.89; I2 = 61%, Chi2 P = 0.006; 10 trials, 5870 fetuses randomised; Analysis 1.30; moderate‐certainty evidence; Table 5). Symmetry in the funnel plot was not suggestive of publication bias (Figure 6).
1.30. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 30: F15: Use of surfactant
6.

Sensitivity analysis
Based on prespecified subgroup analyses, some of the heterogeneity observed may be associated with the planned dose of corticosteroid per treatment course. Restricting the analysis to trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (Mazumder 2008; Wapner 2006), with a result that was similar to the overall analysis (RR 0.82, 95% CI 0.74 to 0.91; I2 = 65%, Chi2 P = 0.005; 8 trials, 5200 fetuses randomised).
Use of postnatal corticosteroids
For use of postnatal corticosteroids, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.37, 95% CI 0.99 to 1.91; I2 = 0%, Chi2 P = 0.82; 4 trials, 4145 fetuses randomised; Analysis 1.31; moderate‐certainty evidence; Table 5).
1.31. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 31: F17: Use of postnatal corticosteroids
Use of nitric oxide for respiratory support
For use of nitric oxide for respiratory support, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.58, 95% CI 0.29 to 1.17; 1 trial, 1146 fetuses randomised; Analysis 1.32; moderate‐certainty evidence; Table 5).
1.32. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 32: F16: Use of nitric oxide for respiratory support
Pulmonary hypertension
No trials reported data for pulmonary hypertension.
Use of inotropic support
Repeat dose(s) of prenatal corticosteroids resulted in a reduction in the use of inotropic support compared with placebo or standard care (RR 0.80, 95% CI 0.66 to 0.97; I2 = 0%, Chi2 P = 0.50; 2 trials, 1474 fetuses randomised; Analysis 1.33; high‐certainty evidence; Table 5).
1.33. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 33: F20: Use of inotropic support
Duration of inotropic support
No trials reported data for duration of inotropic support.
Air leak syndrome
For air leak syndrome, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.83, 95% CI 0.50 to 1.36; I2 = 14%, Chi2 P = 0.32; 4 trials, 2505 fetuses randomised; Analysis 1.34; moderate‐certainty evidence; Table 5).
1.34. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 34: F18: Air leak syndrome
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Wapner 2006), with a result that was similar to the overall analysis (RR 1.01, 95% CI 0.59 to 1.73; I2 = 0%, Chi2 P = 0.91; 3 trials, 1911 fetuses randomised).
Measures of renal function
No trials reported data for measures of renal function.
Feed tolerance
No trials reported data for feed tolerance.
Apgar score less than 7 at five minutes
For Apgar score less than seven at five minutes, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.83, 95% CI 0.64 to 1.09; I2 = 0%, Chi2 P = 0.89; 3 trials, 4032 fetuses randomised; Analysis 1.35; moderate‐certainty evidence; Table 5).
1.35. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 35: F21: Apgar score < 7 at 5 minutes
Cardiac hypertrophy
Only one trial reported neonatal cardiac hypertrophy, using measures of interventricular septal thickness and left ventricular wall thickness in diastole (Crowther 2006). Repeat dose(s) of prenatal corticosteroids probably had little or no effect on thickness of the interventricular septum (MD 0 mm, 95% CI −0.2 to 0.3; 1 trial, 175 neonates; Analysis 1.36) or of the left ventricular posterior wall (MD 0 mm, 95% CI −0.3 to 0.2; 1 trial, 175 neonates; Analysis 1.37) compared with placebo or standard care (moderate‐certainty evidence; Table 5).
1.36. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 36: F22: Neonatal cardiac hypertrophy as measured by interventricular septal thickness (IVS in mm)
1.37. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 37: F22: Neonatal cardiac hypertrophy as measured by left ventricular wall thickness in diastole (mm)
Measures of hypothalamic‐pituitary‐adrenal axis function
For measures of HPA axis function, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (very low‐certainty evidence; Table 5).
Five trials reported data relevant to this outcome.
Crowther 2006 and Wapner 2006 both reported cord blood cortisol concentrations (MD −50 nmol/L, 95% CI −73 to −28; I2 = 0%, Chi2 P = 0.66; 2 trials, 442 neonates; Analysis 1.64).
Crowther 2006 reported data for days three, seven, 14 and 21 unstressed and day three poststress (heel prick or nasopharyngeal suction) salivary cortisol. There were lower salivary cortisol concentrations with the repeat doses of corticosteroids compared with placebo on day three poststress samples (median: 11.9 nmol/L with repeat doses versus 21.3 nmol/L with placebo; P = 0.02; 27 neonates) and day seven unstressed samples (median: 11.7 nmol/L with repeat doses versus 18.2 nmol/L with placebo; P = 0.04; 39 neonates). Day 3 prestress (34 neonates, day 14 (39 neonates) and day 21 (35 neonates) salivary cortisol concentrations) and cortisol on did not differ between treatment groups.
Crowther 2006 also reported basal and post‐metyrapone cortisol concentrations at days two to three postbirth with no differences between baseline cortisol concentrations (63 neonates), baseline adrenocorticotrophic hormone (ACTH) concentrations or post‐metyrapone cortisol, ACTH or 11‐deoxycortisol concentrations (26 neonates).
Aghajafari 2002 reported median (5th/95th centiles) for cord cortisol concentration and cord ACTH concentration at birth. The authors reported no difference but the overall sample size was low (11 neonates) and data were unable to be included in the meta‐analysis as there were insufficient data to derive estimates of mean and SD.
McEvoy 2002 reported cortisol concentrations on day five at baseline and 30‐minute post‐ACTH stimulation test for 11 neonates, although the numbers in each treatment group were unclear. The authors described a lower post‐ACTH stimulation cortisol concentration and no difference in baseline cortisol concentrations.
TEAMS 1999 had data available for "HPA axis suppression" although the outcome occurred in only one neonate in the repeat dose(s) of prenatal corticosteroids group.
1.64. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 64: L14: Mean cord cortisol concentrations at birth (nmol/L)
Secondary outcomes for the woman
Puerperal sepsis
For puerperal sepsis, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.05, 95% CI 0.78 to 1.43; I2 = 29%, Chi2 = 5.65, P = 0.23; 6 trials, 3246 women; Analysis 2.5; moderate‐certainty evidence; Table 6).
2.5. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 5: G1: Puerperal sepsis
Chorioamnionitis during labour
For chorioamnionitis during labour, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.13, 95% CI 0.90 to 1.42; I2 = 0%, Chi2 = 3.31,P = 0.65; 7 trials, 4417 women; Analysis 2.6; moderate‐certainty evidence; Table 6).
2.6. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 6: G2: Chorioamnionitis during labour
Endometritis
For endometritis, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.17, 95% CI 0.74 to 1.84; I2 = 63%, Chi2 = 5.36,P = 0.07; 4 trials, 2842 women; Analysis 2.7; low‐certainty evidence; Table 6).
2.7. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 7: G3: Endometritis
Sensitivity analysis
Based on prespecified subgroup analyses, some of the heterogeneity may be associated with differences in the planned interval between treatment courses and the planned dose per treatment course.
Pyrexia after trial entry requiring the use of antibiotics
For pyrexia after trial entry requiring the use of antibiotics, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.56, 95% CI 0.25 to 1.25; 1 trial, 156 women; Analysis 2.8; low‐certainty evidence; Table 6).
2.8. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 8: G4: Pyrexia after trial entry requiring the use of antibiotics
Intrapartum fever requiring the use of antibiotics
No trials reported data for intrapartum fever requiring the use of antibiotics.
Postpartum haemorrhage
Repeat dose(s) of prenatal corticosteroids probably reduced the risk of postpartum haemorrhage compared with placebo or standard care (RR 0.54, 95% CI 0.31 to 0.96; I2 = 0%, Chi2 = 0.92,P = 0.34; 2 trials, 641 women; Analysis 2.9; moderate‐certainty evidence; Table 6).
2.9. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 9: G6: Postpartum haemorrhage
Postnatal pyrexia
For postnatal pyrexia, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.87, 95% CI 0.55 to 1.38; 1 trial, 982 women; Analysis 2.10; moderate‐certainty evidence; Table 6).
2.10. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 10: G7: Postnatal pyrexia
Preterm prelabour rupture of the membranes after trial entry
For preterm prelabour rupture of the membranes after trial entry, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.91, 95% CI 0.65 to 1.29; I2 = 0%, Chi2 = 0.75,P = 0.39; 2 trials, 977 women; Analysis 2.11; moderate‐certainty evidence; Table 6).
2.11. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 11: G8: Prelabour rupture of membranes after trial entry
Mode of birth
Vaginal birth was the only mode of birth reported by more than one study and able to be combined with meta‐analysis.
Repeat dose(s) of prenatal corticosteroids had little or no effect on the outcome of vaginal birth compared with placebo or standard care (RR 0.94, 95% CI 0.87 to 1.00; I2 = 32%, Chi2 = 7.34,P = 0.2; 6 trials, 4025 women; Analysis 2.12; high‐certainty evidence; Table 6).
2.12. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 12: G9: Mode of birth: vaginal birth
Sensitivity analysis
Prespecified subgroup analyses did not appear to explain the observed heterogeneity.
Hypertension
For hypertension, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.08, 95% CI 0.87 to 1.32; I2 = 14%, Chi2 = 2.31,P = 0.31; 3 trials, 3327 women; Analysis 2.13; moderate‐certainty evidence; Table 6).
2.13. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 13: G10: Hypertension
Pulmonary oedema
No trials reported data for pulmonary oedema.
Glucose intolerance
For glucose intolerance, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.23, 95% CI 0.90 to 1.67; I2 = 0%, Chi2 = 0.23,P = 0.63; 2 trials, 2345 women; Analysis 2.13; moderate‐certainty evidence; Table 6).
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Wapner 2006), with a result that was similar to the overall analysis (RR 1.12, 95% CI 0.68 to 1.85; 1 trial, 1853 women).
Postnatal depression
For postnatal depression, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.88, 95% CI 0.70 to 1.10; 1 trial, 1671 women; Analysis 2.15; moderate‐certainty evidence; Table 6).
2.15. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 15: G13: Postnatal depression
Local injection site adverse effects
Repeat dose(s) of prenatal corticosteroids possibly resulted in a lower risk of local injection site adverse effects compared with placebo (RR 0.44, 95% CI 0.32 to 0.60; I2 = 95%, Chi2 P = 0.00001; 2 trials, 1477 women; Analysis 2.16; low‐certainty evidence; Table 6).
2.16. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 16: G14: Local injection site adverse effects
Sensitivity analysis
Sensitivity analysis was not performed for heterogeneity as there were only two trials in this analysis.
Insomnia after treatment
Repeat dose(s) of prenatal corticosteroids increased the risk of insomnia after treatment compared with placebo (RR 1.21, 95% CI 1.04 to 1.40; I2 = 15%, Chi2 = 3.54, P = 0.32; 4 trials, 3198 women; Analysis 2.17; high‐certainty evidence; Table 6).
2.17. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 17: G15: Insomnia after treatment
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Aghajafari 2002), with a result that was similar to the overall analysis (RR 1.21, 95% CI 1.05 to 1.41; I2 = 23%, Chi2 P = 0.27; 3 trials, 3335 women).
Gastrointestinal adverse effects of treatment
Repeat dose(s) of prenatal corticosteroids probably reduced the risk of gastrointestinal adverse effects of treatment compared with placebo or standard care (RR 0.34, 95% CI 0.14 to 0.85; 1 trial, 495 women; Analysis 2.18; moderate‐certainty evidence; Table 6).
2.18. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 18: G16: Gastrointestinal adverse effects of treatment
Satisfaction with the therapy
No trials reported data for satisfaction with the therapy.
Quality of life
No trials reported data for quality of life.
Secondary outcomes for the child in early childhood (aged two to less than five years)
Child behaviour
For child behaviour, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (moderate‐certainty evidence; Table 7).
Three trials reported on child behaviour (Crowther 2006; Murphy 2008; Peltoniemi 2007). Crowther 2006 used the Child Behavior Checklist, a rating scale that screens for behavioural and emotional problems with higher scores indicating higher levels of behavioural and emotional problems. The trial showed no differences between groups in the total (MD 0.8, 95% CI −1.9 to 3.4; P = 0.56; 1045 children) or individual domain scores for internalising, externalising, emotional reactivity, anxiety or depression, somatic complaints, withdrawal, sleep problems, attention problems or aggressive behaviour between groups. The proportion of children in the clinical range (defined as the top 2.5th centile) did not differ between treatment groups in total (RR 1.11, 95% CI 0.79 to 1.57; P = 0.544; 1045 children) or across the individual domains, except for attention problems (RR 1.87, 95% CI 1.03 to 3.42; P = 0.04; 1045 children).
Murphy 2008 used the Behavior Rating Scale of the Bayley Scales of Infant Development II (BSID‐II) and found scores were non‐optimal or questionable for 285/902 (31.6%) children in the prenatal corticosteroid therapy group and 239/874 (27.3%) children in the placebo group (RR 1.16, 95% CI 1.00 to 1.34; Analysis 3.9).
3.9. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 9: H1: Child behaviour: Behaviour rating scale in the clinical range (BSID‐II)
Peltoniemi 2007 used the Early Childhood Behaviour Questionnaire, which uses a 201‐point scale to describe child behaviour in different daily situations. The trial found no differences between the treatment groups for 148 children for any of the variables in the questionnaire (activity, attentional focusing, attentional shifting, cuddliness, discomfort, fear, frustration, high‐intensity pleasure, impulsivity, inhibition, low‐intensity pleasure, motor activity, perceptual sensitivity, positive anticipation, sadness, shyness, soothability, sociability), including the summary subscales of extraversion, negative affectivity and effortful control (Analysis 3.10; Analysis 3.11; Analysis 3.12).
3.10. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 10: H1: Child behaviour at early childhood follow‐up as assessed by Early Child Behaviour Questionnaire Extraversion summary scale
3.11. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 11: H1: Child behaviour at early childhood follow‐up assessed by Early Child Behaviour questionnaire Negative affectivity summary scale
3.12. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 12: H1: Child behaviour at early childhood follow‐up assessed by Early Child Behaviour questionnaire Effortful control summary scale
Differences in the measurement of these outcomes precluded meaningful combination with meta‐analysis.
Motor impairment
Motor impairment at early childhood follow‐up was not reported separate to cerebral palsy, but three trials assessed motor development using the BSID II Psychomotor Developmental Index (Crowther 2006; Murphy 2008; Wapner 2006). Wapner 2006 used the median and range to estimate mean and SD as described by Wan 2014. Murphy 2008 reported a mean Psychomotor Developmental Index of 97.86 for the repeat doses of corticosteroid group and 98.98 for the placebo group, but the data were unable to be included in the meta‐analysis as SDs were not reported (1901 children).
Repeat dose(s) of prenatal corticosteroids probably has little or no effect on mean Psychomotor Developmental Index compared with placebo (MD 1.26, 95% CI −0.45 to 2.96; I2 = 39%, Chi2 P = 0.20; 2 trials, 1423 children; Analysis 3.13; moderate‐certainty evidence; Table 7).
3.13. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 13: H2: Psychomotor Developmental Index at early childhood follow‐up
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Wapner 2006), with a result that was similar to the overall analysis (RR 0.40, 95% CI −1.75 to 2.55; 1 trial, 958 children). Sensitivity analysis was not performed for heterogeneity as there were only two trials in this analysis.
Deafness/hearing impairment
For deafness/hearing impairment, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.97, 95% CI 0.56 to 1.71; I2 = 0%, Chi2 P = 0.74; 4 trials, 3528 children; Analysis 3.14; moderate‐certainty evidence; Table 7).
3.14. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 14: H3: Deafness/hearing impairment at early childhood follow‐up
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (Peltoniemi 2007; TEAMS 1999), with a result that was similar to the overall analysis (RR 0.97, 95% CI 0.56 to 1.71; I2 = 0%, Chi2 P = 0.74; 2 trials, 3146 children).
Blindness/visual impairment
For blindness/visual impairment, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.17, 95% CI 0.65 to 2.10; I2 = 0%, Chi2 P = 0.38; 3 trials, 3274 children; Analysis 3.15; moderate‐certainty evidence; Table 7).
3.15. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 15: H4: Blindness/visual impairment at early childhood follow‐up
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (TEAMS 1999), with a result that was similar to the overall analysis (RR 1.17, 95% CI 0.65 to 2.10; I2 = 0%, Chi2 P = 0.38; 2 trials, 3151 children).
Growth assessments
Mean weight at early childhood follow‐up
Repeat dose(s) of prenatal corticosteroids resulted in a small reduction in mean weight at early childhood follow‐up compared with placebo (MD −0.16 kg, 95% CI −0.25 to −0.07; I2 = 6%, Chi2 P = 0.36; 4 trials, 3784 children; Analysis 3.19; high‐certainty evidence; Table 7).
3.19. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 19: N1i: Mean weight at early childhood follow‐up (kg)
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Peltoniemi 2007), with a result that was similar to the overall analysis (MD −0.17 kg, 95% CI −0.26 to −0.08; I2 = 19%, Chi2 P = 0.29; 3 trials, 3541 children).
Weight for age at early childhood follow‐up
Repeat dose(s) of prenatal corticosteroids resulted in little or no effect on weight for age at early childhood follow‐up compared with placebo (SMD −0.06, 95% CI −0.15 to 0.04; I2 = 0%, Chi2 P = 0.54; 3 trials, 1776 children; Analysis 3.21; high‐certainty evidence; Table 7). Three trials measured weight for age at early childhood follow‐up (Crowther 2006; Peltoniemi 2007; Wapner 2006). Crowther 2006 reported weight Z scores. Peltoniemi 2007 reported relative weight as a percentage. Wapner 2006 reported weight as a percentile for age based on a published standard population.
3.21. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 21: N1i: Mean weight adjusted for age at early childhood follow‐up (standardised mean difference)
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Peltoniemi 2007), with a result that was similar to the overall analysis (SMD −0.06, 95% CI −0.16 to 0.04; I2 = 14%, Chi2 P = 0.28; 2 trials, 1514 children).
Low weight for age
For low weight for age, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.92, 95% CI 0.72 to 1.19; I2 = 0%, Chi2 P = 0.32; 2 trials, 1533 children; Analysis 3.22; moderate‐certainty evidence; Table 7). Two trials reported low weight for age, defined as weight less than 10th centile (Crowther 2006; Wapner 2006).
3.22. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 22: N1i: Weight small for age at early childhood follow‐up
High weight for age
Crowther 2006 also reported high weight for age (greater than 90th centile) with no difference reported between the repeat doses of corticosteroids group and placebo but with CIs that included both benefit and harm (RR 1.01, 95% CI 0.73 to 1.39; 1047 children).
Mean height at early childhood follow‐up
Repeat dose(s) of prenatal corticosteroids had little or no effect on mean height at early childhood follow‐up compared with placebo (MD −0.1 cm, 95% CI −0.3 to 0.2; I2 = 0%, Chi2 P = 0.77; 4 trials, 3784 children; Analysis 3.27; high‐certainty evidence; Table 7).
3.27. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 27: N1iii: Mean height at early childhood follow‐up (cm)
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Peltoniemi 2007), with a result that was similar to the overall analysis (MD −0.1 cm, 95% CI −0.3 to 0.2; I2 = 0%, Chi2 P = 0.63; 3 trials, 3541 children).
Height for age at early childhood follow‐up
Repeat dose(s) of prenatal corticosteroids had little or no effect on height for age at early childhood follow‐up compared with placebo (SMD −0.06, 95% CI −0.15 to 0.04; I2 = 0%, Chi2 P = 0.41; 3 trials, 1776 children; Analysis 3.29; high‐certainty evidence; Table 7). Three trials measured height for age at early childhood follow‐up (Crowther 2006; Peltoniemi 2007; Wapner 2006). Crowther 2006 and Peltoniemi 2007 reported height Z scores. Wapner 2006 reported height as a percentile for age based on a published standard population.
3.29. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 29: N1iii: Mean height adjusted for age at early childhood follow‐up (standardised mean difference)
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Peltoniemi 2007), with a result that was similar to the overall analysis (SMD −0.08, 95% CI −0.18 to 0.02; I2 = 0%, Chi2 P = 0.55; 2 trials, 1533 children).
Short for age
For short for age, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.03, 95% CI 0.77 to 1.36; I2 = 71%, Chi2 P = 0.07; 2 trials, 1526 children; Analysis 3.30; low‐certainty evidence; Table 7). Two trials reported short for age, defined as height less than 10th centile (Crowther 2006; Wapner 2006).
3.30. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 30: N1iii: Height small for age at early childhood follow‐up
Sensitivity analysis
Sensitivity analysis was not performed for heterogeneity as there were only two trials in this analysis.
Mean head circumference at early childhood follow‐up
Repeat dose(s) of prenatal corticosteroids had little or no effect on mean head circumference at early childhood follow‐up compared with placebo (MD −0.1 cm, 95% CI −0.2 to 0.1; I2 = 0%, Chi2 P = 0.86; 4 trials, 3784 children; Analysis 3.23; high‐certainty evidence; Table 7).
3.23. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 23: N1ii: Mean head circumference at early childhood follow‐up (cm)
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Peltoniemi 2007), with a result that was similar to the overall analysis (MD −0.1 cm, 95% CI −0.2 to 0.1; I2 = 0%, Chi2 P = 0.84; 3 trials, 3541 children).
Head circumference for age at early childhood follow‐up
Repeat dose(s) of prenatal corticosteroids had little or no effect head circumference for age at early childhood follow‐up compared with placebo (SMD 0.02, 95% CI −0.07 to 0.11; I2 = 0%, Chi2 P = 0.74; 3 trials, 1776 children; Analysis 3.25; high‐certainty evidence; Table 7). Three trials measured head circumference for age at early childhood follow‐up (Crowther 2006; Peltoniemi 2007; Wapner 2006). Crowther 2006 and Peltoniemi 2007 reported head circumference Z scores. Wapner 2006 reported head circumference as a percentile for age based on a published standard population.
3.25. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 25: N1ii: Mean head circumference adjusted for age at early childhood follow‐up (standardised mean difference)
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Peltoniemi 2007), with a result that was similar to the overall analysis (SMD 0.01, 95% CI −0.09 to 0.18; I2 = 0%, Chi2 P = 0.78; 2 trials, 1533 children).
Small head circumference for age
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on small head circumference for age compared with placebo (RR 1.02, 95% CI 0.87 to 1.21; I2 = 63%, Chi2 P = 0.10; 2 trials, 1527 children; Analysis 3.26; moderate‐certainty evidence; Table 7). Both trials reported small head circumference for age, defined as head circumference less than 10th centile (Crowther 2006; Wapner 2006).
3.26. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 26: N1ii: Head circumference small for age at early childhood follow‐up
Sensitivity analysis
Sensitivity analysis was not performed for heterogeneity as there were only two trials in this analysis.
Body mass index
No trials reported data for BMI.
Obesity/overweight
No trials reported data for obesity/overweight.
Blood pressure
Mean systolic blood pressure
Repeat dose(s) of prenatal corticosteroids probably resulted in a reduction in mean systolic blood pressure at early childhood follow‐up compared with placebo (MD −2.9 mmHg, 95% CI −5.4 to −0.4; 1 trial, 486 children; Analysis 3.31; high‐certainty evidence; Table 7).
3.31. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 31: N3: Mean systolic blood pressure at early childhood follow‐up (mmHg)
Mean systolic blood pressure Z score
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean systolic blood pressure Z score at early childhood follow‐up compared with placebo (MD −0.10, 95% CI −0.28 to 0.08; 1 trial, 672 children; Analysis 3.32; moderate‐certainty evidence; Table 7).
3.32. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 32: N3: Mean systolic blood pressure Z score at early childhood follow‐up
Mean diastolic blood pressure
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean diastolic blood pressure at early childhood follow‐up compared with placebo (MD −1.0 mmHg, 95% CI −2.9 to 0.9; 1 trial, 486 children; Analysis 3.33; high‐certainty evidence; Table 7).
3.33. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 33: N3: Mean diastolic blood pressure at early childhood follow‐up (mmHg)
Mean diastolic blood pressure Z score
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean diastolic blood pressure Z score at early childhood follow‐up compared with placebo (MD 0.10, 95% CI −0.05 to 0.25; 1 trial, 628 children; Analysis 3.34; moderate‐certainty evidence; Table 7).
3.34. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 34: N3: Mean diastolic blood pressure Z score at early childhood follow‐up
Hypertension
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on risk of hypertension at early childhood follow‐up compared with placebo (RR 0.97, 95% CI 0.77 to 1.23; 1 trial, 628 children; Analysis 3.16; moderate‐certainty evidence; Table 7).
3.16. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 16: H5: Hypertension at early childhood follow‐up
Measures of lung function
No trials reported data for measures of lung function.
Chronic lung disease of infancy
No trials reported data for chronic lung disease of infancy at early childhood follow‐up.
Asthma or recurrent wheeze
For asthma or recurrent wheeze, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.89, 95% CI 0.74 to 1.06; I2 = 62%, Chi2 P = 0.07; 3 trials, 1720 children; Analysis 3.17; moderate‐certainty evidence; Table 7).
3.17. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 17: H8: Asthma or recurrent wheeze at early childhood follow‐up
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (Peltoniemi 2007; Wapner 2006), with a result that was similar to the overall analysis (RR 0.87, 95% CI 0.70 to 1.07; 1 trial, 1060 children). Sensitivity analysis was not performed for heterogeneity as there were only three trials in this analysis.
Any respiratory disease
Three trials reported data on respiratory disease (Crowther 2006; Murphy 2008; Peltoniemi 2007). Crowther 2006 reported admissions for respiratory illness. Murphy 2008 reported admission for respiratory infections. Peltoniemi 2007 reported severe lung disease including pneumonia and recurrent wheeze or asthma. Repeat dose(s) of prenatal corticosteroids probably had little or no effect on risk of any respiratory disease compared with placebo (RR 1.04, 95% CI 0.92 to 1.18; I2 = 0%, Chi2 P = 0.52; 3 trials, 3423 children; Analysis 3.18; high‐certainty evidence; Table 7).
3.18. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 18: H9: Any respiratory disease at early childhood follow‐up
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of two trials with some concerns of risk of bias (Crowther 2006; Peltoniemi 2007), with a result that was similar to the overall analysis (RR 1.04, 95% CI 0.85 to 1.28; 1 trial, 2104 children).
Measures of insulin and glucose homeostasis
No trials reported data for measures of insulin and glucose homeostasis.
Measures of lipid profile
No trials reported data for lipid profile.
Measures of hypothalamic‐pituitary‐adrenal axis function
No trials reported data for measures of HPA axis function.
Body composition
No trials reported data for body composition.
Bone density
No trials reported data for bone density.
Secondary outcomes for the child in mid‐ to late childhood (aged five to less than 18 years)
Child behaviour
Two trials reported data for child behaviour from a parent‐administered questionnaire (Crowther 2006; Murphy 2008). Crowther 2006 reported mean scores on a parent administered Strengths and Difficulties Questionnaire (SDQ). Murphy 2008 reported mean scores for the Child Behaviour Checklist 1.5–5 years. For both questionnaires, higher scores represent more behavioural problems. Repeat dose(s) of prenatal corticosteroids probably had little or no effect on child behaviour compared with placebo (SMD 0.00, 95% CI −0.08 to 0.08; I2 = 0%, Chi2 P = 0.39; 2 trials, 2480 children; Analysis 4.10; moderate‐certainty evidence; Table 8).
4.10. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 10: I4: Child behaviour at mid‐ to later childhood follow‐up (standardised mean difference)
Murphy 2008 also reported the proportion with neurocognitive/neurobehavioural disability, which included those with elevated score on the Behaviour Rating Inventory of Executive Function (BRIEF) – preschool version (RR 0.96, 95% CI 0.75 to 1.22; 1615 children; Analysis 4.9).
4.9. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 9: I4: Abnormal child behaviour at mid‐ to later childhood follow‐up
Deafness/hearing impairment
For deafness/hearing impairment, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.62, 95% CI 0.77 to 3.41; I2 = 0%, Chi2 P = 0.75; 2 trials, 2532 children; Analysis 4.8; low‐certainty evidence; Table 8).
4.8. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 8: I3: Deafness/hearing impairment at mid‐ to later childhood follow‐up
Blindness/visual impairment
For blindness/visual impairment, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 1.12, 95% CI 0.79 to 1.59; I2 = 0%, Chi2 P = 0.56; 2 trials, 2532 children; Analysis 4.7; low‐certainty evidence; Table 8).
4.7. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 7: I2: Blindness/visual impairment at mid‐ to later childhood follow‐up
Growth assessments
Mean weight at mid‐ to late childhood follow‐up
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean weight at mid‐ to late childhood follow‐up compared with placebo (MD −0.2 kg, 95% CI −0.6 to 0.2; 1 trial, 1635 children; Analysis 4.13; moderate‐certainty evidence; Table 8).
4.13. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 13: O1i: Mean weight at mid‐ to later childhood follow‐up (kg)
Mean weight Z score at mid‐ to late childhood follow‐up
Repeat dose(s) of prenatal corticosteroids had little or no effect on mean weight Z score at mid‐ to late childhood follow‐up compared with placebo (MD −0.06, 95% CI −0.23 to 0.11; 1 trial, 940 children; Analysis 4.14; high‐certainty evidence; Table 8).
4.14. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 14: O1i: Mean weight Z score at mid‐ to later childhood follow‐up
Low weight for age
Crowther 2006 also reported low weight for age (less than 10th centile) (RR 0.96, 95% CI 0.67 to 1.39; P = 0.83; 940 children).
Mean height at mid‐ to late childhood follow‐up
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean height at mid‐ to late childhood follow‐up compared with placebo (MD −0.4 cm, 95% CI −1.1 to 0.3; 1 trial, 1635 children; Analysis 4.17; moderate‐certainty evidence; Table 8).
4.17. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 17: O1iii: Mean height at mid‐ to later childhood follow‐up (cm)
Mean height Z score at mid‐ to late childhood follow‐up
Repeat dose(s) of prenatal corticosteroids had little or no effect on mean height Z score at mid‐ to late childhood follow‐up compared with placebo (MD 0.02, 95% CI −0.13 to 0.17; 1 trial, 912 children; Analysis 4.18; high‐certainty evidence; Table 8).
4.18. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 18: O1iii: Mean height Z score at mid‐ to later childhood follow‐up
Short for age
Crowther 2006 also reported short for age (less than 10th centile) (RR 0.94, 95% CI 0.64 to 1.40; P = 0.78; 912 children).
Mean head circumference at mid‐ to late childhood follow‐up
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean head circumference at mid‐ to late childhood follow‐up compared with placebo (MD −0.1 cm, 95% CI −0.4 to 0.2; 1 trial, 1635 children; Analysis 4.15; moderate‐certainty evidence; Table 8).
4.15. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 15: O1ii: Mean head circumference at mid‐ to later childhood follow‐up (cm)
Mean head circumference Z score at mid‐ to late childhood follow‐up
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean head circumference Z score at mid‐ to late childhood follow‐up compared with placebo (MD −0.04, 95% CI −0.22 to 0.14; 1 trial, 885 children; Analysis 4.16; moderate‐certainty evidence; Table 8).
4.16. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 16: O1ii: Head circumference Z score at mid‐ to later childhood follow‐up
Body mass index
Repeat dose(s) of prenatal corticosteroids had little or no effect on BMI Z score compared with placebo (MD −0.13, 95% CI −0.30 to 0.04; 1 trial, 910 children; Analysis 4.19; high‐certainty evidence; Table 8).
4.19. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 19: O2: BMI Z scores at mid‐ to later childhood follow‐up
Obesity/overweight
No trials reported data for obesity/overweight.
Blood pressure
Mean systolic blood pressure
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean systolic blood pressure compared with placebo (MD 0.3 mmHg, 95% CI −1.1 to 1.7; 1 trial, 1635 children; Analysis 4.22; moderate‐certainty evidence; Table 8).
4.22. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 22: O6: Mean systolic blood pressure at mid‐ to later childhood follow‐up (mmHg)
Mean systolic blood pressure Z score
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean systolic blood pressure Z score compared with placebo (MD 0.07, 95% CI −0.06 to 0.20; 1 trial, 848 children; Analysis 4.24; moderate‐certainty evidence; Table 8).
4.24. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 24: O6: Mean systolic blood pressure Z score at mid‐ to later childhood follow‐up
Mean diastolic blood pressure
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean diastolic blood pressure compared with placebo (MD 0.7 mmHg, 95% CI −0.5 to 1.9; 1 trial, 1635 children; Analysis 4.23; moderate‐certainty evidence; Table 8).
4.23. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 23: O6: Mean diastolic blood pressure at mid‐ to later childhood follow‐up (mmHg)
Mean diastolic blood pressure Z score
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on mean diastolic blood pressure Z score compared with placebo (MD −0.09, 95% CI −0.25 to 0.07; 1 trial, 848 children; Analysis 4.25; moderate‐certainty evidence; Table 8).
4.25. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 25: O6: Mean diastolic blood pressure Z score at mid‐ to later childhood follow‐up
Measures of lung function
Forced expiratory volume in 1 second Z score
For forced expiratory volume in 1 second (FEV1) Z score, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (MD −0.06, 95% CI −0.34 to 0.22; 1 trial, 185 children; Analysis 4.26; very low‐certainty evidence; Table 8).
4.26. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 26: O8: Measures of lung function at mid‐ to later childhood follow‐up: mean FEV1 Z score
Forced vital capacity Z score
For forced vital capacity (FVC) Z score, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (MD −0.18, 95% CI −0.49 to 0.13; 1 trial, 185 children; Analysis 4.27; very low‐certainty evidence; Table 8).
4.27. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 27: O8: Measures of lung function at mid‐ to later childhood follow‐up: mean FVC Z score
FEV1/FVC ratio Z score
For FEV1/FVC ratio Z score, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (MD −0.19, 95% CI −0.44 to 0.06; 1 trial, 185 children; Analysis 4.28; very low‐certainty evidence; Table 8).
4.28. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 28: O8: Measures of lung function at mid‐ to later childhood follow‐up: mean FEV1/FVC Z score
Asthma or recurrent wheeze
Repeat dose(s) of prenatal corticosteroids had little or no effect on risk of asthma compared with placebo (RR 1.01, 95% CI 0.85 to 1.19; 1 trial, 979 children; Analysis 4.11; high‐certainty evidence; Table 8).
4.11. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 11: I5: Asthma or recurrent wheeze at mid‐ to later childhood follow‐up
Respiratory disease
For any respiratory disease, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (RR 0.79, 95% CI 0.36 to 1.72; 1 trial, 979 children; Analysis 4.12; very low‐certainty evidence; Table 8).
4.12. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 12: I6: Any respiratory disease at mid‐ to later childhood follow‐up
Measure of insulin and glucose homeostasis
Repeat dose(s) of prenatal corticosteroids had little or no effect on measures of insulin and glucose homeostasis compared with placebo (moderate‐certainty evidence; Table 8).
Only one trial reported measures of insulin and glucose homeostasis (Crowther 2006). Testing included a fasting plasma glucose (MD −0.1 mmol/L, 95% CI −0.2 to 0.0), fasting plasma insulin concentration (ratio of geometric means (RGM) 1.02, 95% CI 0.86 to 1.22) and a 90‐minute, frequently sampled intravenous glucose tolerance test (IVGTT) for 182 children. The IVGTT was used to derive measures of insulin sensitivity index (RGM 0.89, 95% CI 0.74 to 1.08), glucose effectiveness (RGM 0.93, 95% CI 0.81 to 1.06), acute insulin release (RGM 1.05, 95% CI 0.84 to 1.31) and glucose disappearance constant (RGM 0.92, 95% CI 0.80 to 1.07).
Lipid profile
No trials reported data for lipid profile.
Measures of hypothalamic‐pituitary‐adrenal axis function
Repeat dose(s) of prenatal corticosteroids possibly has little or no effect on mean salivary cortisol compared with placebo (moderate‐certainty evidence; Table 8). One trial reported data for measures of HPA axis function in 212 children (Crowther 2006). Morning and evening salivary cortisol concentrations were similar between those children who had received repeat doses of prenatal corticosteroids and those who received placebo after adjusting for time of sampling (morning RGM 1.05, 95% CI 0.91 to 1.20; P = 0.52; evening RGM 0.78, 95% CI 0.60 to 1.01; P = 0.11).
Bone density
Repeat dose(s) of prenatal corticosteroids probably had little or no effect on total body bone mineral content compared with placebo (moderate‐certainty evidence; Table 8). One trial reported data for measures of total body bone mineral content in 185 children (Crowther 2006). There were no differences between treatment groups for whole‐body bone mineral content adjusted for height (RGM 0.99, 95% CI 0.97 to 1.01), whole‐body bone area adjusted for height (RGM 0.99, 95% CI 0.95 to 1.02) and spinal bone mineral content (RGM 0.97, 95% CI 0.92 to 1.03).
Body composition
Repeat dose(s) of prenatal corticosteroids possibly has little or no effect on body composition compared with placebo (moderate‐certainty evidence; Table 8). One trial reported data for body composition assessed using dual energy x‐ray absorptiometry (DEXA) (Crowther 2006). There were no differences between treatment groups for whole‐body fat free mass adjusted for height (RGM 0.98, 95% CI 0.95 to 1.00) and whole‐body fat mass adjusted for height (RGM 0.96, 95% CI 0.76 to 1.20). There were also no differences for fat mass in gynoid (thigh) and android (abdominal) distributions.
Secondary outcomes for the child as an adult
No trials reported data for follow‐up into adulthood.
Secondary outcomes on use of health services
Length of prenatal hospitalisation for the woman
No trials reported data for length of prenatal hospitalisation for the woman.
Length of postnatal hospitalisation for the woman
Repeat dose(s) of prenatal corticosteroids had little or no effect on length of postnatal hospitalisation for the women compared with placebo (MD 0.00 days, 95% CI −0.22 to 0.22; 1 trial, 483 women; Analysis 6.3; high‐certainty evidence; Table 9).
6.3. Analysis.

Comparison 6: Repeat dose(s) of corticosteroids versus single course: outcomes for health services, Outcome 3: Q2: Length of postnatal hospitalisation for the woman (days)
Maternal admission to the intensive care unit
No trials reported data for maternal admission to the ICU.
Admission to and length of stay in neonatal intensive care unit
Admission to the NICU was reported under 'Secondary outcomes for the fetus/neonate/infant: admission to neonatal intensive care unit' and no trials reported data for the length of stay in the NICU to date.
Length of infant hospitalisation
For length of infant hospitalisation, benefit or harm could not be excluded for repeat dose(s) of corticosteroids compared with placebo or standard care (MD 0.18 days, 95% CI −2.60 to 2.96; I2 = 14%, Chi2 P = 0.31; 3 trials, 1733 infants; Analysis 6.4; moderate‐certainty evidence; Table 9).
6.4. Analysis.

Comparison 6: Repeat dose(s) of corticosteroids versus single course: outcomes for health services, Outcome 4: Q4: Length of infant hospitalisation (days)
Costs of maternal care
No trials reported data for costs of maternal care.
Costs of neonatal care
No trials reported data for costs of neonatal care.
Hospital re‐admission at childhood follow‐up
Early childhood follow‐up
Repeat dose of prenatal corticosteroid probably had little or no effect on hospital readmission by early childhood follow‐up compared with placebo (RR 1.02, 95% CI 0.93 to 1.11; I2 = 0%, Chi2 P = 0.39; 4 trials, 3824 children; Analysis 6.1; high‐certainty evidence; Table 9).
6.1. Analysis.

Comparison 6: Repeat dose(s) of corticosteroids versus single course: outcomes for health services, Outcome 1: K1: Hospital re‐admission by early childhood follow‐up
Sensitivity analysis
Restricting the analysis to trials at low risk of bias resulted in the exclusion of one trial with some concerns of risk of bias (Peltoniemi 2007), with a result that was similar to the overall analysis (RR 1.00, 95% CI 0.91 to 1.09; I2 = 0%, Chi2 P = 0.52; 3 trials, 35654 children).
Mid‐ to late childhood follow‐up
For hospital readmission by mid‐ to late childhood follow‐up, benefit or harm was unable to be excluded for repeat dose(s) of prenatal corticosteroids compared with placebo (RR 1.10, 95% CI 0.80 to 1.52; 1 trial, 980 children; Analysis 6.2; very low‐certainty evidence; Table 9).
6.2. Analysis.

Comparison 6: Repeat dose(s) of corticosteroids versus single course: outcomes for health services, Outcome 2: K1: Hospital re‐admission by mid to later childhood follow‐up
Discussion
Summary of main results
Outcomes for the fetus/neonate/infant
A total of 4895 women (5975 babies) who had received a course of prenatal corticosteroids seven or more days previously were randomised to repeat dose(s) of prenatal corticosteroids or placebo/standard care ("no repeat dose(s)"). The use of repeat dose(s) of corticosteroid for women at risk of preterm birth reduced the relative risk of neonatal RDS by 18% (NNTB 16, 95% CI 11 to 29 women; 62 fewer babies with RDS per 1000 women treated) and the relative risk of serious infant outcome by 12% (NNTB 39, 95% CI 24 to 158 women; 26 fewer babies with a serious health outcome per 1000 women treated). These are clinically important neonatal benefits, observed with high‐certainty (neonatal RDS) and moderate‐certainty (serious infant outcome) evidence. There is also high‐certainty evidence that repeat dose(s) results in no difference in the risk of chronic lung disease (RR 1.00, 95% CI 0.83 to 1.22; 5661 babies) and moderate‐certainty evidence that it reduces the risk of severe lung disease (RR 0.83, 95% CI 0.72 to 0.97, NNTB 45, 95% CI 27 to 256; 4955 babies). The information available could not exclude benefit or harm for the outcomes fetal or neonatal or infant death less than one year of age (RR 0.95, 95% CI 0.73 to 1.24; 5849 babies), severe intraventricular haemorrhage (RR 1.13, 95% CI 0.69 to 1.86; 5066 babies) and necrotising enterocolitis (RR 0.84, 95% CI 0.59 to 1.22; 5736 babies).
Outcomes for the woman
For the women, there was moderate‐certainty evidence of no increase in the likelihood of a caesarean birth, providing some reassurance about the safety of repeat prenatal corticosteroid treatment for the mothers. Benefit or harm was unable to be excluded for maternal sepsis. There was significant heterogeneity for the trials reporting on adverse effects and only one trial reported on each of maternal death and discontinuation of therapy due to adverse effects so it was not possible to determine if repeat dose(s) of corticosteroid resulted in benefit, harm or no effect on these outcomes. No trials reported data for breastfeeding status at hospital discharge or risk of admission to the intensive care unit.
Outcomes for the child in early childhood (age two to less than five years)
Moderate‐ to high‐certainty evidence identified little or no effect of repeat dose(s) of prenatal corticosteroids compared with no repeat doses for neurodevelopmental outcomes (neurodevelopmental impairment: RR 0.97, 95% CI 0.85 to 1.10; 3616 children; survival without neurodevelopmental impairment: RR 1.01, 95% CI 0.98 to 1.04; 3845 children; survival without major neurodevelopmental impairment; RR 1.02, 95% CI 0.98 to 1.05; 1816 children). An increase or decrease in the risk of death since randomisation could not be excluded (RR 1.06, 95% CI 0.81 to 1.40; 5 trials, 4565 babies randomised).
Outcomes for the child in mid‐ to late childhood (age five to less than 18 years)
Long‐term neurodevelopmental outcomes at mid‐ childhood follow‐up did not differ between treatment groups for survival free of neurocognitive impairment (RR 1.01, 95% CI 0.95 to 1.08; 963 children) and survival free of major neurocognitive impairment (RR 1.00, 95% CI 0.97 to 1.04; 2682 children) (moderate‐certainty evidence). Benefit or harm could not be excluded for death since randomisation (RR 0.93, 95% CI 0.69 to 1.26; 2874 babies randomised) and neurocognitive impairment (RR 0.96, 95% CI 0.72 to 1.29; 897 children).
Outcomes for the child in adulthood
No trials reported data for follow‐up into adolescence or adulthood.
Subgroup analyses
Prespecified subgroup analyses indicated significant interactions for three outcomes: severe RDS, severe lung disease and caesarean section. For severe RDS, participants in trials with corticosteroid exposure of greater than 12 mg to 24 mg per week were more likely to benefit from repeat doses than those with exposure of 12 mg or less per week. For severe lung disease, participants were more likely to experience benefit from repeat doses of corticosteroids in trials that planned to give one or more repeat treatment courses compared with trials that planned to give only one repeat treatment course, and in trials that planned to give the treatment at a minimum interval of seven days compared with trials in which the interval was 14 days or greater. Caesarean section was more likely to occur in trials that planned to give one or more repeat treatment courses than in trials that planned to give only one repeat treatment course, but it was not more likely to occur with repeat corticosteroids compared with placebo in the overall analysis. There were no significant interactions for the limited number of outcomes with data available between the effect of repeat doses of corticosteroids and the number of fetuses (singleton versus multiple) or gestation at the time of the first repeat dose (less than 28 weeks' gestation versus 28 weeks' gestation or greater).
Overall completeness and applicability of evidence
Women in the included trials were recruited in a range of countries and settings. The trials were conducted at either tertiary or provincial hospitals, with participants from high‐, middle‐ and low‐income countries. The trials included a wide variety of reasons for being at risk of preterm birth and all trials included women with both singleton and multiple pregnancies. The results are considered applicable to hospital settings worldwide. Betamethasone has been the only corticosteroid evaluated in randomised trials of repeat prenatal corticosteroids prior to preterm birth. Dexamethasone is often used because it is less expensive and often more easily available, particularly in low‐ to middle‐income countries. It is uncertain whether the effects seen for betamethasone would be the same for dexamethasone. Appropriate trials are needed.
There has been no cost‐effectiveness analysis of the use of repeat prenatal corticosteroids for women at risk of preterm birth seven or more days after initial corticosteroid treatment.
The evidence available provides reassurance about the health of infants into early to mid‐childhood when repeat prenatal corticosteroids are used for women at risk of preterm birth. However, there is as yet no information on health, neurodevelopmental, growth, cardiovascular or metabolic outcomes at older ages to assess the long‐term benefits or risks of this treatment.
Quality of the evidence
The risk of bias of the trials included in this review ranged from low to high risk across different outcomes, with most being assessed as low risk or having some concerns of risk of bias. All trials had adequate randomisation and all except one used a placebo. Losses to follow‐up to the time of primary hospital discharge after birth were nil or minimal. At early childhood follow‐up losses to follow‐up in the five trials that reported data ranged between 7.5% and 21.0%. At mid‐ to late childhood follow‐up, losses to follow‐up were 14% and 19% in the two trials that reported data. Four trials stopped recruitment early (Guinn 2001; Peltoniemi 2007; TEAMS 1999; Wapner 2006).
The evidence was mostly of moderate and high certainty for the primary outcomes with reasons for downgrading the certainty being predominantly inconsistency of effect or imprecision due to wide CIs including both appreciable benefit and harm. Secondary outcomes were also mostly of moderate to high certainty although for some outcomes the certainty was downgraded to low or very low because of risk of bias or marked imprecision. Outcomes assessed at mid‐ to late childhood follow‐up were mainly at risk of bias due to missing data, resulting in moderate certainty of evidence for most of these outcomes.
Potential biases in the review process
The evidence for this review is derived from trials identified from a detailed, systematic search process without language restriction. It is possible (but unlikely) that additional trials comparing the use of repeat corticosteroids prior to preterm birth versus placebo or no treatment have been published but not identified. It is also possible that there are other trials, additional to those of which we are aware, that have been conducted but are not yet published. Should any such trials be identified, we will include them in updates of this review.
Unit of analysis issues were considered as a potential source of bias. In accordance with the Pregnancy and Childbirth Cochrane Group guidelines, for neonatal outcomes we used the number of fetuses alive at the point of randomisation as the denominator for analysis. This avoids bias by ensuring analysis is based on the total group of women and their fetuses initially randomised but may bias the analysis by underestimating the occurrence of neonatal morbidity, as the most unwell fetuses may die in utero or soon after birth and contribute to the denominator but not to measures of neonatal morbidity. However, stillbirth was an infrequent occurrence, making this unlikely to be a major contributor to bias.
Not all trials reported data using the same unit of analysis. For multiple pregnancies, Guinn 2001 randomly selected one of the fetuses for inclusion in the analysis, an approach that may underestimate the occurrence of neonatal morbidity and therefore the effects of interventions because infants from multiple pregnancies have higher rates of many neonatal morbidities. Mazumder 2008 reported data using the pregnancy as the unit of analysis but did not detail which fetus or infant would be assessed for dichotomous outcomes. Wapner 2006 used the pregnancy as the unit of analysis and considered an outcome to have occurred if any of the fetuses randomised experienced the outcome. For these two trials the effect of interventions may be overestimated as the rate of occurrence of neonatal morbidity will be artificially elevated through the use of a smaller denominator and this may increase the apparent efficacy of interventions that may reduce neonatal morbidity.
Two trials have studied long‐term outcomes; Murphy 2008 at five years and Crowther 2006 at six to eight years of age; but the proportion of participants lost to follow‐up was high enough for there to be some concerns of bias due to missing outcome data for most outcomes reported. The relative paucity of studies that have performed long‐term follow‐up means that long‐term outcomes are unknown for most trials included in this review.
The large number of outcomes included in the review does introduce a risk of Type 1 error (i.e. finding a difference between treatment groups purely by chance rather than observing a true difference). The concordance between findings for risk of RDS and need for respiratory treatments such as surfactant makes this less likely for many of the respiratory outcomes but this is still an important potential bias for other outcomes.
The assessment of risk of bias and GRADE assessments have subjective elements that may be a source of bias in the review process. To reduce this risk, two review authors performed risk of bias assessments, and two review authors performed all GRADE assessments of certainty of evidence for completeness and accuracy.
Three review authors (CAC, JEH, CM) were investigators for the ACTORDS trial or its follow‐up studies (Crowther 2006). Other review authors (PM and AW) performed assessment of trustworthiness, risk of bias and data extraction for this trial without direct involvement of the investigators or authors of the trial or its follow‐up studies.
Agreements and disagreements with other studies or reviews
The results and conclusions of this systematic review are consistent with the results of an IPD meta‐analysis of trials of repeat doses of corticosteroids compared with placebo or standard care for woman with an ongoing risk of preterm labour (Crowther 2019). The results of the IPD subgroup analyses also suggested interactions between use of respiratory support and the repeat course interval, the number of courses and the total dose received; findings broadly consistent with the findings of the subgroup analyses in this review for severe RDS and severe lung disease.
The current international guideline from the World Health Organization regarding the use of interventions for preterm birth have used an earlier version of this review on which to base their recommendations (WHO 2015).
One systematic review performed for the production of clinical guidelines for practice in Australia and New Zealand had similar conclusions regarding repeat doses of prenatal corticosteroids and a number of the recommendations for further research have been addressed in the mid‐childhood follow‐up outcomes included in this review (Antenatal Corticosteroids CPG Panel 2015).
The American College of Obstetrics and Gynecology committee on obstetric practice produced a committee opinion statement in 2017, reaffirmed in 2020, that the use of repeat doses of prenatal corticosteroids in the context of threatened preterm birth should be considered for women with a pregnancy of less than 34 weeks' gestation for whom their last course of prenatal corticosteroids was 14 or more days prior (ACOG 2017).
Authors' conclusions
Implications for practice.
The short‐term benefits seen for babies support the use of repeat dose(s) of prenatal corticosteroids for women who have received an initial course of prenatal corticosteroids seven of more days previously and who remain at risk of preterm birth.
Repeat dose(s) of prenatal corticosteroids given to women at risk of preterm birth, compared to no repeat treatment, reduce the occurrence of respiratory distress syndrome (RDS) by 18% and the risk of serious health problems in the first few weeks of life for the infant by 12%. For one baby to benefit by not developing RDS, 16 women (95% confidence interval (CI) 11 to 29) would need to be treated with repeat prenatal corticosteroids, resulting in 62 fewer babies with RDS per 1000 women treated. The number needed to treat for an additional beneficial outcome (NNTB) for serious neonatal outcome is 39 women (95% CI 24 to 158), resulting in 26 fewer babies with a serious health outcome per 1000 women treated. At birth, the neonatal benefits are associated with a reduction in measures of mean body size (mean weight, head circumference and length) including weight adjusted for gestational age and an increase in the proportion of infants born small for gestational age (number needed to treat for an additional harmful outcome of 29, 95% CI 16 to 89). These effects were not observed for other anthropometric assessments that adjusted for gestational age (length and head circumference). Differences in body size measurements were no longer seen by hospital discharge in the two trials that report data. The limited evidence available from early and mid‐ childhood reassuringly shows no significant harm, although no benefit. There was a small reduction in mean weight at early childhood in those exposed to repeat dose(s) of prenatal corticosteroids, but this was not observed at mid‐childhood follow‐up. There were no differences in neurodevelopmental outcomes or childhood head circumference or height at early or mid‐childhood.
The reduction in RDS and serious neonatal outcome are of clinical importance, despite the absence of an effect on severe RDS or severe lung disease. The benefits of a reduction in neonatal morbidity with repeat dose(s) of prenatal corticosteroids must be weighed against the potential risks of the reduction in birthweight. Although the association of low birthweight with poorer health outcomes in later life is well established, long‐term follow‐up in adults exposed to prenatal corticosteroids has thus far shown minimal effects on health up to 30 years of age when compared with those exposed to placebo (Dalziel 2005a). The implications of a small reduction in birthweight on long‐term health for the infant exposed to repeat doses(s) of prenatal corticosteroids are unclear but there is little or no effect on health outcomes in follow‐up to eight years of age and, as yet, there is insufficient evidence in adolescence and adulthood.
The use of corticosteroids for women at risk of preterm birth is supported by evidence of overall benefit for its use in low‐, middle‐ and high‐income settings (McGoldrick 2020). Repeat prenatal corticosteroids should be considered for women who have received a course of prenatal corticosteroids seven or more days previously, and who remain at risk of preterm birth before 34 weeks' gestation. Women eligible for repeat prenatal corticosteroid treatment should be informed of the known benefits and risks and counselled about the available information about early and mid‐childhood health outcomes.
Implications for research.
We are aware of two ongoing trials in the US of repeat prenatal corticosteroids for women at risk of preterm birth in the setting of preterm prelabour rupture of membranes prior to trial entry (NCT02469519; NCT02939742).
There are still no data published for health and neurocognitive outcomes in adulthood. Such information would be valuable for assessing the overall benefits and risks of using repeat prenatal corticosteroids for women at risk of preterm birth.
Any further trials of repeat dose(s) of prenatal corticosteroids for women who remain at risk of preterm birth after a course of corticosteroids should be of high quality; be large enough to assess mortality and serious morbidity; consider comparing different corticosteroid preparations (all trials to date have used betamethasone for repeat treatment); provide further evaluation of the times between repeat courses; evaluate the optimal amount of corticosteroid given at each course; provide data on all relevant maternal and infant outcomes; and ensure assessment of neurodevelopmental status of the child at follow‐up and organise assessment of longer‐term outcomes including behaviour, educational achievement, cardiovascular status, bone density, hypothalamic‐pituitary‐adrenal axis function, glucose intolerance and lung function.
Cost‐effectiveness analysis of the use of repeat corticosteroids should be conducted.
Feedback
Murphy, 5 July 2012
Summary
As the authors of the Multiple courses of Antenatal Corticosteroids for preterm birth Study (MACS), which is one of the trials included in this review, we have a few comments that need to be addressed. 1. In MACS, we collected data on ‘severe respiratory distress syndrome (RDS)’ rather than ‘RDS’. In this review, data from MACS for ‘severe RDS’ are included in the meta‐analysis for ‘severe lung disease’ but not in the meta‐analysis for ‘RDS’. As a result, the data from MACS are currently not able to contribute to answering the question as to the effect of multiple courses of antenatal corticosteroids on RDS. Because RDS is defined differently in the various trials we believe it is reasonable that the outcome RDS should include both mild and severe disease. Subgroup analyses could then look at mild RDS and severe RDS separately. Given that MACS is the largest trial included in the review, it is particularly important that its results should contribute to answering the question about the effect of repeated doses of antenatal corticosteroids on RDS. 2. The review authors have chosen to include data for ‘severe RDS’ from the various trials in an outcome of ‘severe lung disease’. We find the labelling of severe RDS as ‘severe lung disease’ ambiguous. Severe lung disease could theoretically include meconium aspiration, pneumothorax, group B streptococcal pneumonia and other conditions that are not thought to benefit from antenatal corticosteroids. 3. In the Results text section ‘secondary outcomes for the child’, the review states that “Data were not able to be included in the meta‐analysis for Murphy 2008 for anthropometric assessments expressed as means, as standard deviations were not available in the published report.” In our report of the 18‐24 month follow‐up for MACS (Asztalos 20081, not Murphy 2008 as stated in the review) the data are expressed as means, mean differences and confidence intervals. The within‐group standard deviations are actually not needed for the meta‐analysis, as all that is required is the mean difference (which we reported) and its variance. The variance can be derived by squaring the width of the CI divided by 2*1.96. That is: Variance of Mean Difference = [(UL – LL)/(2*1.96)]2. In any case, an estimate of the within‐group standard deviations can be calculated from the confidence limits and the group sample sizes.
The table below shows the summary statistic for the mean difference in birth weight at early childhood follow‐up with and without the MACS data. When MACS data are included, the difference in birth weight between the two groups is statistically significant.. Table Summary statistic for the mean difference in birth weight at early childhood follow‐up.
| Mean Difference in Weight | Variance of Mean Difference | Lower Limit | Upper Limit | P value | |
| Summary without MACS | ‐0.0297 | 0.0086 | ‐0.2114 | 0.1520 | 0.7488 |
| Summary with MACS | ‐0.2003 | 0.0046 | ‐0.3327 | ‐0.0678 | 0.0030 |
The long term impact of weighing less in early childhood is not known. Nevertheless, that repeated doses of antenatal corticosteroids have adverse effects on growth is worrisome in that it signifies the potential for repeated doses to cause harm.
Follow‐up studies have thus far been reassuring, demonstrating no difference in death or neurologic impairment in early childhood. However, we should not be falsely reassured by these studies. The long‐term follow‐up studies have limited power to rule out the effect of repeated doses of antenatal corticosteroids on infrequent adverse events. Thus, finding no difference in long‐term outcome does not necessarily prove safety. We are reminded of the RCTs of postnatal corticosteroid treatment which initially demonstrated short term benefits and then over time, with the completion of long‐term studies, demonstrated the potential for harm.2
4. We believe that the findings of short term neonatal benefits from repeated courses of antenatal corticosteroids are important. However, given the adverse effect of antenatal corticosteroids on birth weight and weight at early childhood follow‐up, and the uncertain effect on long term outcomes, we believe that the conclusions and recommendations for clinical practice in this review should be more cautious than they currently are. We would suggest the review authors consider a statement such as “Although the short term neonatal benefits of repeated courses of antenatal corticosteroids support their use, long‐term benefits have not been demonstrated and long‐term adverse effects have not been ruled out. The adverse effect of repeated doses of antenatal corticosteroids on birth weight and weight at early childhood follow‐up is a concern. Caution should therefore be exercised to ensure that only those women who are at particularly high risk of very early preterm birth are offered treatment with repeated courses of antenatal corticosteroids.” References 1. Asztalos EV, Murphy KE, Hannah ME, Willan AR, Matthews SG, Ohlsson A, et al. Multiple courses of antenatal corticosteroids for preterm birth study: 2‐year outcomes. Pediatrics 2010;126:e1045‐e1055.
2. Halliday HL, Ehrenkranz RA, Doyle LW. Early (<8 days) postnatal corticosteroids for preventing chronic lung disease in preterm infants. Cochran Database of Sys Rev 2010, Issue 5.
Kellie E. Murphy, Andrew R Willan, Mary E. Hannah, Elizabeth Asztalos, Arne Ohlsson, Edmond N Kelly, Stephen G Matthews, Saroj Saigal, Susan Ross, Marie‐France Delisle, Kofi Amankwah, Patricia Guselle, Amiram Gafni, Shoo K Lee, B Anthony Armson, for the MACS Collaborative Group. July 2012
[Feedback received from Kellie E Murphy, 5 July 2012]
Reply
Many thanks to the authors of the Multiple courses of Antenatal Corticosteroids for preterm birth Study (MACS).
1. The MACS trial reported on the outcome of 'severe respiratory distress syndrome'. The authors query why the data from MACS were only included in the outcome for 'severe lung disease' rather than RDS (any).
We do not feel that it would be appropriate to combine data from trials reporting respiratory distress syndrome (mild or severe) with severe RDS. This is likely to result in significant heterogeneity and therefore we chose to report data for RDS and severe lung disease as separate outcomes. If the authors of the MACS trial can provide data for RDS (any) we would certainly be willing to add it to the appropriate analysis.
2. The authors of the MACS trial query the labelling of the outcome 'severe lung disease'.
This outcome was selected by the review authors and is defined in the review protocol.
3. The authors of the MACS trial query why their data for anthropometric assessments at 18 to 24 months were not included in the meta‐analysis.
Data were not included as SD's were not available in the trial report. We acknowledge that the author of this report was Asztalos 2008, however Cochrane methodology requires that it is the primary trial that is cited in the text. The reference to Asztalos can be found in the reference section of this review. In order to enter data in to the statistical programme we require mean values for each group with the associated standard deviation or standard error. If the authors of the MACS trial can provide us with the mean and standard deviations for the treatment and the control groups with the numbers of children in each group we will certainly be willing to add these data to the appropriate analyses.
4. The authors of the MACS trial query the conclusions of the review.
The review authors consider the conclusions and both the clinical and research recommendations to be appropriate summaries of the available evidence.
Contributors
Caroline Crowther
What's new
| Date | Event | Description |
|---|---|---|
| 26 January 2021 | New citation required and conclusions have changed | The updated review includes data from one an additional trial, with a total of 11 trials now included. Additional data now suggests that repeat doses of corticosteroids are associated with an increase in small for gestational age. In the last update, there was no increase identified. |
| 26 January 2021 | New search has been performed | Search updated with 48 additional reports. A total of 63 reports were assessed for this update (15 from awaiting classification in last published version plus 48 reports from updated searches). |
History
Protocol first published: Issue 4, 2002 Review first published: Issue 3, 2003
| Date | Event | Description |
|---|---|---|
| 5 January 2015 | New search has been performed | Search updated and two new trials excluded (Bontis 2011; Romejko‐Wolniewicz 2013). |
| 5 January 2015 | New citation required but conclusions have not changed | There are no changes to the conclusions of this review. |
| 24 July 2012 | Feedback has been incorporated | Comments from Kellie Murphy added ‐ see Feedback 1. |
| 18 July 2011 | Amended | Corrected reference error ‐ Ashworth 2006 amended to read Ashwood 2006. |
| 18 April 2011 | New citation required and conclusions have changed | Conclusions are now stronger; early benefit now without evidence of longer
term harm. Two additional authors joined the review team (C McKinlay and P Middleton). |
| 31 March 2011 | New search has been performed | Search updated in March 2011 and data from five new trials added (Garite 2009; Mazumder 2008; McEvoy 2010; Murphy 2008; Peltoniemi 2007). Published longer term follow‐up in early childhood now included for four trials (Crowther 2006; Murphy 2008; Peltoniemi 2007; Wapner 2006) and information on follow up for one further trial added (Mazumder 2008). Acknowledgement of unpublished information provided on caesarean section by Emeritus Professor Thomas Garite for the Garite 2009 trial. |
| 20 September 2008 | Amended | Converted to new review format. |
| 11 May 2007 | New search has been performed | Search updated in November 2006 and data from two trials now published added (Crowther 2006; Wapner 2006). We updated the search just before submission for publication and identified the published report for the previously listed Peltoniemi ongoing study. We have added it to the ' Studies awaiting assessment' section and will consider it for inclusion in the next update (Peltoniemi 2007). |
Risk of bias
Risk of bias for analysis 1.1 A1: Fetal or neonatal or infant death (< 1 year of age).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.1.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Treating team were blinded to allocation of patients. The outcome measure was objective. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome assessors were blinded to treatment allocation. Outcome measure was objective. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The injection of the study treatment was given by a designated research nurse who was not caring for the woman. The physical appearance of the study solutions was masked. Intention‐to‐treat analysis was performed. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | The outcome measure was objective. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. Assessment of mortality not able to be influenced by knowledge of intervention received. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | Some concerns | Some concerns overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label and some concerns of bias in selection of the reported result because of the limited detail of analysis methods and outcomes. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Analysis was by intention‐to‐treat. | Low risk of bias | Data available for this outcome for all except one infant. | Low risk of bias | Investigators were blinded to treatment allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. Outcome is objective. | Low risk of bias | Limited ways this outcome can be reported. Outcome specified in trial registration. Although no SAP or protocol was available to review, those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Study investigators were blinded to allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 3/94 fetuses (3.2%) in each group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. Objective outcome. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.2 A2: Fetal death.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.2.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Treating team were blinded to allocation of patients. The outcome measure was objective. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome assessors were blinded to treatment allocation. Outcome measure was objective. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The injection of the study treatment was given by a designated research nurse who was not caring for the woman. The physical appearance of the study solutions was masked. Intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | The outcome measure was objective. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. Assessment of mortality not able to be influenced by knowledge of intervention received. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | Some concerns | This was an open‐label trial and blinding of outcome assessors was not described but assessment of mortality is objective. No SAP available to review. Limited detail in analysis plans stated in methods. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | No missing data for this outcome. | Low risk of bias | Investigators were blinded to treatment allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Study investigators were blinded to allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.3 A3: Neonatal death.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.3.1 In all neonates | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Treating team were blinded to allocation of patients. The outcome measure was objective | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results . | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome assessors were blinded to treatment allocation. Outcome measure was objective. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The injection of the study treatment was given by a designated research nurse who was not caring for the woman. The physical appearance of the study solutions was masked. Intention‐to‐treat analysis was performed. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | The outcome measure was objective. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Overall Low as despite lack of detailed data analysis plan, outcomes were prespecified and absolute numbers of events were reported. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. Assessment of mortality not able to be influenced by knowledge of intervention received. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | Some concerns | This was an open‐label trial and blinding of outcome assessors was not described but assessment of mortality is objective. No SAP available to review. Limited detail in analysis plans stated in methods. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Data available for this outcome for all except one infant. | Low risk of bias | Investigators were blinded to treatment allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Study investigators were blinded to allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.4 A5: Respiratory distress syndrome.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.4.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as diagnosed by the treating team but case definition is unclear. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results . | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as per the diagnosis from case notes and defined by clinical and radiological features. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The primary outcome was specified in the trial registration document as neonatal respiratory outcomes. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The injection of the study treatment was given by a designated research nurse who was not caring for the woman. The physical appearance of the study solutions was masked. Intention‐to‐treat analysis was performed. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | RDS was based on clinical diagnosis, chest x‐ray findings and oxygen therapy. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Assessment not likely to be influenced by knowledge of allocation as outcome predefined and requiring minimal judgement. Based on clinical findings, radiography and oxygen requirement. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | High risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. Outcome ascertainment of clinical and radiological findings able to be influenced by knowledge of treatment allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, some concerns of bias in selection of the reported result due to limited detail of analysis methods and outcomes and high of risk of bias for measurement of the outcome as blinding of outcome assessors was not described for an outcome likely to be influenced by knowledge of allocation. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis. | Low risk of bias | Data available for this outcome for all except one infant. | Low risk of bias | RDS assessment based on clinical signs, radiographic evidence and oxygen requirement. Investigators were unaware of treatment allocation. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Diagnosis was based on clinical and radiological criteria, including need for oxygen or surfactant therapy. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 4/94 (4%) in intervention and 7/94 in placebo group (7%). | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Outcome based on clinical findings and radiographic findings. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.5 A6: Severe respiratory distress syndrome.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.5.1 All fetuses randomised | ||||||||||||
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Based on RDS diagnosis plus mechanical ventilation >24hours and surfactant treatment. Assessment not likely to be influenced by knowledge of allocation as outcome predefined and requiring minimal judgement. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. Assessment unlikely to be affected by knowledge of allocation as outcome grading was objective (based on the need for mechanical ventilation for 12 hours). | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | Some concerns | Some concerns overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label and some concerns of bias in selection of the reported result because of the limited detail of analysis methods and outcomes. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up for neonatal data | Low risk of bias | Outcome based on radiographic evidence of RDS or need for surfactant therapy, with severity defined by requirement for invasive ventilation. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | Outcome specified but not defined in registration document but well defined in methods. Although SAP was not reviewed, those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on RDS diagnosis along with ventilation requirement for >24hours despite surfactant therapy. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | An event was reported for multiple pregnancies if either or both infants had the outcome, with the more severe determining the overall severity assessment. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Outcome based on clinical findings, radiographic findings and treatment with surfactant/oxygen therapy. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as multiple pregnancies reported an event if either or both infants had the outcome, with the more severe determining the overall assessment. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.6 A7: Severe lung disease.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.6.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Severity of lung disease classified using defined thresholds. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The primary outcome was specified in the trial registration document as neonatal respiratory outcomes. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Based on RDS diagnosis plus mechanical ventilation and surfactant treatment. Assessment not likely to be influenced by knowledge of allocation as outcome predefined and requiring minimal judgement. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. Assessment unlikely to be affected by knowledge of allocation as outcome grading was objective and based on the need for mechanical ventilation for 12 hours. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | Some concerns | Some concerns overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label and some concerns of bias in selection of the reported result because of the limited detail of analysis methods and outcomes. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Outcome based on radiographic evidence of RDS or need for surfactant therapy, with severity defined by requirement for invasive ventilation. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | Outcome specified but not defined in registration document but well defined in methods. Although SAP was not reviewed, those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on RDS diagnosis along with ventilation requirement for >24hours despite surfactant therapy. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | An outcome was reported for multiple pregnancies if either or both infants had the outcome, with the more severe determining the overall severity assessment. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Outcome based on clinical findings, radiographic findings and treatment with surfactant/oxygen therapy. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as multiple pregnancies reported an event if either or both infants had the outcome, with the more severe determining the overall assessment. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.7 A8: Chronic lung disease.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.7.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as diagnosed by the treating team but case definition is unclear. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. Outcome definition was clear in methods. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Defined by need for a clinical treatment (oxygen requirement at 36 weeks post menstrual age). Unlikely to have differed by group due to blinding of clinical team. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The injection of the study treatment was given by a designated research nurse who was not caring for the woman. The physical appearance of the study solutions was masked. Intention‐to‐treat analysis was performed. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | Chronic lung disease was based on oxygen therapy at 30 days after birth. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Assessment not likely to be influenced by knowledge of allocation as outcome predefined (oxygen requirement for at least 28days after birth) and requiring minimal judgement. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | High risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome ascertainment is likely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, some concerns of bias in selection of the reported result due to limited detail of analysis methods and outcomes and high of risk of bias for measurement of the outcome as blinding of outcome assessors was not described for an outcome likely to be influenced by knowledge of allocation. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Data available for this outcome for all except one infant. | Low risk of bias | Outcome was not defined in methods but was unlikely to differ between treatment groups. Investigators were unaware of treatment allocation. | Some concerns | SAP and protocol were not available for review. Outcome was not in trial registration documentation. The outcome definition was unclear in the methods with multiple possible definitions available. | Some concerns | Some concerns in selection of the reported result as the outcome was not defined in methods or registration document, SAP was not available and multiple possible definitions exist. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% missing data for this outcome. | Low risk of bias | Outcome diagnosed based on oxygen requirement at 36 weeks post menstrual age. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | Outcome specified but not defined in trial registration but well defined in methods. Although SAP was not reviewed, those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on diagnosis of bronchopulmonary dysplasia(need for supplemental oxygen or ventilation at 36 weeks post menstrual age or 4 weeks postnatal age). Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Outcome based on oxygen requirement at 36 weeks post menstrual age. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.8 A9: Severe intraventricular haemorrhage (grade 3/4).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.8.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | For two babies the head ultrasound performed at the time of hospital discharge was missed. It is unclear whether they had been assigned to placebo or intervention but both were born at term and therefore lower likelihood of severe IVH. | Low risk of bias | Measured by ultrasonography. Ultrasonography was only performed for infants of birthweight less than 1500g. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. IVH grade 3 and 4 classified as severe is standard practice. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Information on this diagnosis was obtained from case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for 95% of those randomised. | Low risk of bias | Outcome measurement method not stated but unlikely to have differed between treatment groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | Only 44% had a cranial USS analysis performed. No sensitivity analysis performed. USS were performed if clinically indicated and this is unlikely to vary due to blinding of healthcare staff. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome unlikely to be affected by knowledge of allocation as grading criteria based on imaging or autopsy findings were clearly defined. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. Classification of grade 3 and 4 IVH as severe is standard practice. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | The number with ultrasounds was unclear. Centres were encouraged to perform at least 2 cranial US for infants with BW <1500grams. Those without US are less likely to have the outcome of interest as they are likely to be >1500grams BW. | Low risk of bias | Cranial US was routine if BW was <1500grams. Outcome assessors were blinded to allocation. | Low risk of bias | Outcome used a standard definition of severe IVH and was prespecified in trial registration document. Although SAP was not reviewed, those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Some concerns | "few infants" were not examined as transferred to a central hospital or were born at term and "appeared healthy". Transferred infants may have been more unwell and more likely to have the outcome of interest. | Low risk of bias | Cranial USS was performed routinely but "few infants" were not examined as transferred to a central hospital. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. IVH grading clearly defined. | Some concerns | Some concerns for missing outcome data as the infants transferred to a central hospital may have been more unwell and at higher risk of the outcome. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 8/94 (8.5%) in intervention and 7/94 (7.4%) in the placebo group. | Low risk of bias | Diagnosis by ultrasound. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.9 A10: Intraventricular haemorrhage.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.9.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Information on this diagnosis was obtained from case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for 95% of those randomised. | Low risk of bias | Outcome measurement method not stated but unlikely to have differed between treatment groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | Only 221/502(44%) of the infants had a cranial USS analysis performed. No sensitivity analysis performed. USS were performed if clinically indicated and this is unlikely to vary due to blinding of healthcare staff. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome unlikely to be affected by knowledge of allocation outcome was clearly defined based on imaging or autopsy findings and required little subjective judgement. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | High risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome ascertainment is likely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, some concerns of bias in selection of the reported result due to limited detail of analysis methods and outcomes and high of risk of bias for measurement of the outcome as blinding of outcome assessors was not described for an outcome likely to be influenced by knowledge of allocation. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Some concerns | A "few infants" were not examined as transferred to a central hospital or were born at term and "appeared healthy". Transferred infants may have been more unwell and more likely to have the outcome of interest. | Low risk of bias | Cranial USS was performed routinely but "few infants" were not examined as transferred to a central hospital or "appeared healthy". Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. IVH grading was clearly defined. | Some concerns | Some concerns of missing outcome data as the infants transferred to a central hospital may have been more unwell and at higher risk of the outcome. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | 93%(460/492) had data for this outcome. For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Head ultrasounds were read centrally by a panel of blinded radiologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.10 A11: Necrotising enterocolitis.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.10.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as diagnosed by the treating team but case definition is unclear. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Information on this diagnosis was obtained from case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | The definition of NEC was not clear in the report but the treatment was masked and outcome assessors were blinded so ascertainment is unlikely to have differed between groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Assessment not likely to be influenced by knowledge of allocation as outcome was predefined (based on clinical, radiological, surgical and autopsy findings) and required minimal judgement. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | High risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome ascertainment is likely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, some concerns of bias in selection of the reported result due to limited detail of analysis methods and outcomes and high of risk of bias for measurement of the outcome as blinding of outcome assessors was not described for an outcome likely to be influenced by knowledge of allocation. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | Outcome specified in trial registration and was clearly defined in methods. Although SAP was not reviewed, those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data for this outcome reported. | Low risk of bias | Necrotizing enterocolitis was diagnosed based on clearly defined staging criteria. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 5/94 (5%) in intervention and 7/94 (7%) in the placebo group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Outcome determined from case records. Deidentified charts underwent blinded central review by 2 neonatologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.11 A12: Composite of serious outcomes .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.11.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | For two babies the head ultrasound performed at the time of hospital discharge was missed however this assessed only a single component of the composite outcome and they still had USS on days 1, 3 and 7 | Low risk of bias | For multiple components of the composite, including retinopathy of prematurity, IVH and PVL, assessments were only performed for those infants <1500grams. Components were as diagnosed by the treating team but definitions were unclear. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | The individual components of the composite were obtained from case notes and ascertainment is unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | The outcome was not specified in the trial registration document. The composite outcome was stated to have been determined post‐hoc but did appear to include all relevant components. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | The composite components were clearly defined but methods of measurement of components were not all stated. The treatment was masked and outcome assessors were blinded so ascertainment is unlikely to have differed. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to treatment allocation. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome but components requiring cranial US assessment were only assessed in 221 participants as clinically indicated. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Components of the composite were all clearly defined and required minimal individual judgment by assessors. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. Outcome unlikely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | Some concerns | Some concerns overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label and some concerns of bias in selection of the reported result because of the limited detail of analysis methods and outcomes. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | Probably <1% with no data for this outcome. | Low risk of bias | Measurement of individual components was appropriate and due to blinded was unlikely to vary between groups. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | Registration document defined composite outcome. Although SAP was not reviewed, those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | A "few infants" were not examined for IVH as they had transferred to a central hospital or were healthy appearing term infants. | Low risk of bias | The composite outcome was defined based on occurrence of death, RDS or severe IVH. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. Composite outcome clearly defined in methods and registration document. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 3/94 fetuses (3.2%) in each group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | An outcome was reported for multiple pregnancies if either or both infants had the outcome, with the more severe determining the overall severity assessment. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Clearly defined composite including neonatal mortality/stillbirth, severe RDS, chronic lung disease, grade III/IV IVH, PVL | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as multiple pregnancies reported an event if either or both infants had the outcome, with the more severe determining the overall assessment. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.12 F1i: Term birth ≥ 37 weeks .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.12.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Assessment method for gestational age not stated. Likely to have followed standard methods and unlikely to have differed between groups. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Method of gestational age assessment not stated but unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2002 | Low risk of bias | Allocation was performed by the pharmacy using a randomisation table. The investigators and clinical care providers were unaware of the treatment provided and the placebo and intervention drug were noted to be identical in appearance. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical care providers were unaware of allocation. The administered medication for placebo and intervention were identical in appearance. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Gestational age was calculated from last normal menstrual period and confirmed by Ballard examination. Investigators were unaware of allocation. | Low risk of bias | No protocol, statistical analysis plan or registration document available for review. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Method of measurement of gestation was not stated but likely used standard methods and was unlikely to differ between groups. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Gestational age was measured by last menstrual period and confirmed with ultrasound at <20 weeks gestation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 3/94 fetuses (3.2%) in each group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. Gestational age likely measured using standard methods. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Gestational age determined from last menstrual period and confirmed with ultrasound findings. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.13 F1ii: Preterm birth before 37 weeks.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.13.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Assessment method for gestational age not stated. Likely to have followed standard methods and unlikely to have differed between groups. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Method of gestational age assessment not stated but unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2002 | Low risk of bias | Allocation was performed by the pharmacy using a randomisation table. The investigators and clinical care providers were unaware of the treatment provided and the placebo and intervention drug were noted to be identical in appearance. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical care providers were unaware of allocation. The administered medication for placebo and intervention were identical in appearance. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome | Low risk of bias | Gestational age was calculated from last normal menstrual period and confirmed by Ballard examination. Investigators were unaware of allocation. | Low risk of bias | No protocol, statistical analysis plan or registration document available for review. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Method of measurement of gestation was not stated but likely used standard methods and was unlikely to differ between groups. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Gestational age was measured by last menstrual period and confirmed with ultrasound at <20 weeks gestation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 3/94 fetuses (3.2%) in each group. | Low risk of bias | Gestational age likely measured using standard methods. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Gestational age determined from last menstrual period and confirmed with ultrasound findings. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.14 F1iii: Very preterm birth before 34 weeks.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.14.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Assessment method for gestational age not stated. Likely to have followed standard methods and unlikely to have differed between groups. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Method of gestational age assessment not stated but unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | Assessment method for gestational age not stated. Likely to have followed standard methods and unlikely to have differed between groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Assessment of gestational age was by last menstrual period and confirmed by ultrasound assessment. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Data available for this outcome for all except one infant. | Low risk of bias | Investigators were unaware of treatment allocation. Method of gestational age assessment not stated but unlikely to have differed between groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Gestational age was measured by last menstrual period and confirmed with ultrasound at <20 weeks gestation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.15 F1iv: Extremely preterm birth before 28 weeks.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.15.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Method of gestational age assessment not stated but unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Assessment not likely to be influenced by knowledge of allocation as outcome predefined and requiring minimal judgement. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Method of measurement of gestation was not stated but likely used standard methods and was unlikely to differ between groups. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Gestational age was measured by last menstrual period and confirmed with ultrasound at <20 weeks gestation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 3/94 fetuses (3.2%) in each group. | Low risk of bias | Gestational age likely measured using standard methods. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.16 F1v: Mean gestational age at birth (weeks).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.16.1 In all neonates | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Assessment method for gestational age not stated. Likely to have followed standard methods and unlikely to have differed between groups. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Method of gestational age assessment not stated but unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | Assessment method for gestational age not stated. Likely to have followed standard methods and unlikely to have differed between groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Assessment of gestational age was by last menstrual period and confirmed by ultrasound assessment. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2002 | Low risk of bias | Allocation was performed by the pharmacy using a randomisation table. The investigators and clinical care providers were unaware of the treatment provided and the placebo and intervention drug were noted to be identical in appearance. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical care providers were unaware of allocation. The administered medication for placebo and intervention were identical in appearance. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome | Low risk of bias | Gestational age was calculated from last normal menstrual period and confirmed by Ballard examination. Investigators were unaware of allocation. | Low risk of bias | No protocol, statistical analysis plan or registration document available for review. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Data available for this outcome for all except one infant. | Low risk of bias | Investigators were unaware of treatment allocation. Method of gestational age assessment not stated but unlikely to have differed between groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Method of measurement of gestation was not stated but likely used standard methods and was unlikely to differ between groups. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Gestational age was measured by last menstrual period and confirmed with ultrasound at <20 weeks gestation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 3/94 fetuses (3.2%) in each group. | Low risk of bias | Gestational age likely measured using standard methods. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Gestational age determined from last menstrual period and confirmed with ultrasound findings. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.17 F2: Small‐for‐gestational age at birth.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.17.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Ascertainment of birthweight is unlikely to have differed between groups. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. SGA classification <10th centile is standard. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | Ascertainment of birthweight was unlikely to have differed between treatment groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. Outcome classification as birthweight <10th centile was prespecified. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome is not able to be influenced by knowledge of treatment allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. The definition used for SGA was unclear. | Some concerns | Some concerns overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label and some concerns of bias in selection of the reported result because of the limited detail of analysis methods and outcomes. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Data for one infant missing. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. Definition of SGA used was standard (BW<10th centile). | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | Likely minimal losses: Authors report "a few participants were missing for this outcome" | Low risk of bias | Weight measurement likely followed standard methods. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | All expected measures were reported (BW <5th and <10th centile were reported). SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Study investigators were blinded to allocation. Weight assessment not stated but unlikely to differ between groups. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Growth measurements at birth were specified outcomes in the trial registration document. SGA definition used was standard. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Birthweight was an outcome in protocol documentation but not SGA. Both BW <10th and <5th centile were reported but only 10th centile was eligible for this review. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.18 F3: Admission to the neonatal intensive care unit.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.18.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome based on clinical course, obtained from case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | An objective outcome was used. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.19 F4: Proven neonatal infection while in the neonatal intensive care unit.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.19.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as diagnosed by the treating team but case definition is unclear. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement based on information in case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | Diagnosis of this outcome required positive blood cultures, making it more objective than pure clinical diagnosis. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Assessment not likely to be influenced by knowledge of allocation as outcome predefined (clinical findings, positive blood cultures and required antibiotics) and requiring minimal judgement. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Infection was defined by clinical features and either microbiological or radiological evidence. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome definition not stated but ascertainment unlikely to have differed, given blinding of treating team and investigators. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Some concerns | Proportion of missing data differs between groups. Missing data for 8/94 (8.5%) in intervention group and 16/94 (17%) in the placebo group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Some concerns | Some concerns due to missing outcome data that differed between groups. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Outcome determined from case records. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was defined in protocol documentation. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.20 F5: Early systemic neonatal infection.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.20.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement based on information in case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | High risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome ascertainment is likely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, some concerns of bias in selection of the reported result due to limited detail of analysis methods and outcomes and high of risk of bias for measurement of the outcome as blinding of outcome assessors was not described for an outcome likely to be influenced by knowledge of allocation. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome definition not stated but ascertainment unlikely to have differed, given blinding of treating team and investigators. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 8/94 (8.5%) in intervention and 13/94 (13.8%) in the placebo group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.21 F6: Late systemic neonatal infection.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.21.1 All fetuses randomised | ||||||||||||
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | High risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome ascertainment is likely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, some concerns of bias in selection of the reported result due to limited detail of analysis methods and outcomes and high of risk of bias for measurement of the outcome as blinding of outcome assessors was not described for an outcome likely to be influenced by knowledge of allocation. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome definition not stated but ascertainment unlikely to have differed, given blinding of treating team and investigators. | Low risk of bias | SAP not available for review but detailed analysis plans were included on the registered trial protocol and were overseen by a data safety monitoring committee. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.22 F7: Retinopathy of prematurity.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.22.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | Retinopathy of prematurity assessments were only performed for those infants <1500grams. | Low risk of bias | Method of measurement not stated but unlikely to have differed between groups. Retinopathy of prematurity assessments were only performed for those infants <1500grams. Median birthweight in the intervention group was 1840grams and 2420g in the placebo group. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | The method of measurement of retinopathy of prematurity was not specified but was unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for 91% of those randomised. | Low risk of bias | The method of assessment was not identified in the methods or protocol. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | High risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome ascertainment is likely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, some concerns of bias in selection of the reported result due to limited detail of analysis methods and outcomes and high of risk of bias for measurement of the outcome as blinding of outcome assessors was not described for an outcome likely to be influenced by knowledge of allocation. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | 773 in intervention and 821 in placebo group did not have an ophthalmologic assessment. No sensitivity analysis performed. | Low risk of bias | It is unclear how it was decided which infants had ophthalmologic assessments (likely local hospital protocols) but clinicians and researchers associated with the trial were unaware of allocation. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data: 773 in intervention and 821 in placebo group did not have an ophthalmologic assessment. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Method of assessment of ROP unlikely to have differed between groups, given blinding of the treating team. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 5/94 (5%) in intervention and 7/94 (7%) in the placebo group | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Outcome determined from case records. It was not described how ROP was assessed but ascertainment is unlikely to have differed given the blinding and deidentified charts underwent blinded central review by 2 neonatologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.23 F8: Periventricular leukomalacia.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.23.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | For two babies the head ultrasound performed at the time of hospital discharge was missed. It is unclear whether they had been assigned to placebo or intervention but as per the protocol each would have still had ultrasounds at day 1,3 and 7. | Low risk of bias | Measurement by ultrasonography. Ultrasonography was only performed for infants of birthweight less than 1500g. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Cranial imaging was performed as per usual clinical care. Definition of PVL not specified. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for 94% of those randomised. | Low risk of bias | The method of assessment was not identified in the methods or protocol but was unlikely to have differed between treatment groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | Only 221 of the infants had a cranial USS analysis performed. No sensitivity analysis performed. USS were performed if clinically indicated and this is unlikely to vary due to blinding of healthcare staff. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome unlikely to be affected by knowledge of allocation as the outcome was clearly defined (by imaging findings) and required little subjective judgement. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | High risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome ascertainment is likely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, some concerns of bias in selection of the reported result due to limited detail of analysis methods and outcomes and high of risk of bias for measurement of the outcome as blinding of outcome assessors was not described for an outcome likely to be influenced by knowledge of allocation. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | Centres were encouraged to perform at least 2 cranial US for infants with BW <1500grams only. | Low risk of bias | US routine if BW <1500g and encouraged to have 2 cranial US. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Some concerns | A "few infants" were not examined as transferred to a central hospital or were born at term and "appeared healthy". Transferred infants may have been more unwell and more | Low risk of bias | Cranial USS was performed routinely but "few infants" were not examined as transferred to a central hospital or "appeared healthy". Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Some concerns for missing outcome data as the infants transferred to a central hospital may have been more unwell and at higher risk of the outcome. Given the total number with this outcome was low, this could affect the overall assessment. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 5/94 (5%) in intervention and 7/94 (7%) in the placebo group | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.24 F9: Neonatal encephalopathy .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.24.1 All fetuses randomised | ||||||||||||
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | High risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome ascertainment is likely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, some concerns of bias in selection of the reported result due to limited detail of analysis methods and outcomes and high of risk of bias for measurement of the outcome as blinding of outcome assessors was not described for an outcome likely to be influenced by knowledge of allocation. |
Risk of bias for analysis 1.25 F10: Patent ductus arteriosus.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.25.1 All fetuses randomised | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as diagnosed by the treating team. Outcome defined as "indomethacin treated" PDA. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. Only indomethacin treated PDA was reported but this is a commonly used outcome. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was based on diagnosis of PDA stated in the case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | High risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome ascertainment is likely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, some concerns of bias in selection of the reported result due to limited detail of analysis methods and outcomes and high of risk of bias for measurement of the outcome as blinding of outcome assessors was not described for an outcome likely to be influenced by knowledge of allocation. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Measured as PDA requiring treatment. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | PDA defined by requirement for prostaglandin or surgical closure. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 5/94 (5%) in intervention and 7/94 (7%) in the placebo group | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Outcome determined from case records. Deidentified charts underwent blinded central review by 2 neonatologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.26 F11: Use of respiratory support.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.26.1 All fetuses randomised | ||||||||||||
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 4/94 (4%) in intervention and 7/94 (7%) in the placebo group. | Low risk of bias | Outcome based on care administered by the treating team, who were likely blinded to allocation. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.27 F12: Use of invasive respiratory support .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.27.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Measurement based on treatment administered by clinical team. Unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for 94% of those randomised. | Low risk of bias | Measurement based on treatment administered by clinical team. Unlikely to have differed between treatment groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Neonatal data missing for 1 infant. | Low risk of bias | Outcome based on treatment administered by the treating team, who were blinded to allocation. Investigators were unaware of treatment allocation. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Study investigators were also blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Outcome determined from case records. Based on care provided by the blinded treating team. Deidentified charts underwent blinded central review by 2 neonatologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation as part of the composite outcome. There are limited ways of reporting this outcome. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.28 F13: Use of non‐invasive respiratory support .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.28.1 All fetuses randomised | ||||||||||||
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Study investigators were also blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Outcome determined from case records. Based on care provided by the blinded treating team. Deidentified charts underwent blinded central review by 2 neonatologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation as part of the composite outcome. There are limited ways of reporting this outcome. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.29 F14: Use of oxygen supplementation.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.29.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Measurement based on treatment administered by clinical team. Unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 4/94 (4%) in intervention and 7/94 (7%) in the placebo group. | Low risk of bias | Outcome based on care administered by the treating team, who were likely blinded to allocation. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.30 F15: Use of surfactant.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.30.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Measurement based on treatment administered by clinical team. Unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | Measurement based on treatment administered by clinical team. Unlikely to have differed between treatment groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. Outcome based on treatment provided by clinical team who were blinded to allocation. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. The outcome ascertainment is unlikely to be influenced by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. Minimal alternative definitions possible. | Some concerns | Some concerns overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label and some concerns of bias in selection of the reported result because of the limited detail of analysis methods and outcomes. |
| McEvoy 2002 | Low risk of bias | Allocation was performed by the pharmacy using a randomisation table. The investigators and clinical care providers were unaware of the treatment provided and the placebo and intervention drug were noted to be identical in appearance. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical care providers were unaware of allocation. The administered medication for placebo and intervention were identical in appearance. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome | Low risk of bias | Outcome based on treatment administered by the treating team, who were blinded to allocation. | Low risk of bias | No protocol, statistical analysis plan or registration document available for review. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | 1 infant missing neonatal data. | Low risk of bias | Outcome based on treatment administered by the treating team, who were blinded to allocation. Investigators were unaware of treatment allocation. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data for this outcome reported. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Study investigators were also blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 4/94 (4%) in intervention and 7/94 (7%) in the placebo group. | Low risk of bias | Outcome based on care administered by the treating team, who were likely blinded to allocation. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Based on care provided by the blinded treating team. Deidentified charts underwent blinded central review by 2 neonatologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation as part of the composite outcome. There are limited ways of reporting this outcome. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.31 F17: Use of postnatal corticosteroids.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.31.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Measurement based on treatment administered by clinical team. Unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome based on treatment provided by clinical team who were blinded to allocation. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. | Low risk of bias | Missing data for 5/94 (5%) in intervention and 7/94 (7%) in the placebo group. | Low risk of bias | Outcome based on care administered by the treating team, who were likely blinded to allocation. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.32 F16: Use of nitric oxide for respiratory support.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.32.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Measurement based on treatment administered by clinical team. Unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.33 F20: Use of inotropic support.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.33.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Measurement based on treatment administered by clinical team. Unlikely to have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Study investigators were also blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was not in the trial registration but outcome definition and analysis were unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.34 F18: Air leak syndrome.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.34.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was based on diagnosis of air leak syndrome stated in the case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | Measurement was based on a diagnosis made by the treating team and was unlikely to have differed between treatment groups as the treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 5/94 (5.3%) in intervention and 8/94 (8.5%) in the placebo group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | For multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. | Low risk of bias | Outcome determined from case records. Deidentified charts underwent blinded central review by 2 neonatologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was not specified in protocol documentation. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Some concerns as for multiple pregnancies an event was reported if either or both infants had the outcome. This potentially biases the event incidence and treatment effect estimate. |
Risk of bias for analysis 1.35 F21: Apgar score < 7 at 5 minutes.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.35.1 All fetuses randomised | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Measurement of APGAR scores are standard and would not have differed between treatment groups. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. Cut‐off score of <7 is a standard measure and the only one eligible for this review. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | Measurement of APGAR scores are standard and would not have differed between treatment groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. Although not prespecified, this outcome is routinely collected data that was reported using a standard cut‐off of <7. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | 13 in ANC group and 14 in placebo group with unknown results. | Low risk of bias | Apgar measurement is standardised. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. The cut‐off of <7 at 5 minutes was the only one eligible for this analysis and is a standard cut‐off. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.36 F22: Neonatal cardiac hypertrophy as measured by interventricular septal thickness (IVS in mm).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.36.1 In all neonates | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Some concerns | Assessment of this outcome was only performed at one centre. Consent obtained for 145 (of 189 randomised at this centre) prior to birth. Missing data was noted as due to unsuccessful measurement and differed between groups (26% and 20% in intervention and placebo group). | Low risk of bias | Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Some concerns due to missing outcome data that differed in quantity between groups. |
Risk of bias for analysis 1.37 F22: Neonatal cardiac hypertrophy as measured by left ventricular wall thickness in diastole (mm).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.37.1 In all neonates | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Some concerns | Assessment of this outcome was only performed at one centre. Consent obtained for 145 (of 189 randomised at this centre) prior to birth. Missing data was noted as due to unsuccessful measurement and differed between groups (26% and 20% in intervention and placebo group). | Low risk of bias | Standard measurements of septum and left ventricular wall obtained by cardiac echocardiography. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Some concerns due to missing outcome data that differed in proportion between groups. |
Risk of bias for analysis 1.38 L1: Mean birthweight (g).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.38.1 In all neonates | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | Measurement method unlikely to have differed between treatment groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome assessment unlikely to be affected by knowledge of allocation. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | This was an open‐label trail and blinding of outcome assessors was not described. Outcome assessment not able to be influenced by knowledge of treatment allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | Some concerns | Some concerns overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label and some concerns of bias in selection of the reported result because of the limited detail of analysis methods and outcomes. |
| McEvoy 2002 | Low risk of bias | Allocation was performed by the pharmacy using a randomisation table. The investigators and clinical care providers were unaware of the treatment provided and the placebo and intervention drug were noted to be identical in appearance. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical care providers were unaware of allocation. The administered medication for placebo and intervention were identical in appearance. | Low risk of bias | No missing data for this outcome | Low risk of bias | Investigators were unaware of allocation. Method of measurement not stated but likely to be standard and unlikely to have differed between groups. | Low risk of bias | No protocol, statistical analysis plan or registration document available for review. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Missing neonatal data for 1 infant. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Measurement method not stated but likely standard. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome measurement method not stated but was unlikely to have differed between groups. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 3/94 fetuses (3.2%) in each group. | Low risk of bias | Measurements likely performed by standard methods. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Weight measurement likely followed standard methods. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.39 L1: Mean birthweight Z score.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.39.1 In all neonates | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 3/94 fetuses (3.2%) in each group. | Low risk of bias | Measurements likely performed by standard methods. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.40 L1: Birthweight multiples of the median.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.40.1 In all babies | ||||||||||||
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Weight measurement likely followed standard methods. | Low risk of bias | Protocol with planned statistical analysis was available for review. Birthweight was specified in protocol documentation. Multiples of the median was not specified but is unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains |
Risk of bias for analysis 1.41 L1: Birthweight adjusted for gestation (standardised mean difference).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.41.1 In all neonates | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 3/94 fetuses (3.2%) in each group. | Low risk of bias | Measurements likely performed by standard methods. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Weight likely measured using standard methods. | Low risk of bias | Protocol with planned statistical analysis was available for review. Birthweight was specified in protocol documentation. Multiples of the median was not specified but is unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.42 L2: Interval between trial entry and birth (days).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome unlikely to be affected by knowledge of allocation. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. Median was reported in one study report but mean and SD in another, however the conclusion was the same for both. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Study investigators were blinded to allocation. Outcome was objective. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was not in the trial registration but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation. | Low risk of bias | Low risk of bias for all domains |
Risk of bias for analysis 1.43 L3: Mean head circumference at birth (cm).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.43.1 In all neonates | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for >95% of those randomised. | Low risk of bias | Measurement method unlikely to have differed between treatment groups. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome unlikely to be affected by knowledge of allocation. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | This was an open‐label trial and blinding of outcome assessors was not described. Outcome assessment not able to be influenced by knowledge of treatment allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | Some concerns | Some concerns overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label and some concerns of bias in selection of the reported result because of the limited detail of analysis methods and outcomes. |
| McEvoy 2002 | Low risk of bias | Allocation was performed by the pharmacy using a randomisation table. The investigators and clinical care providers were unaware of the treatment provided and the placebo and intervention drug were noted to be identical in appearance. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical care providers were unaware of allocation. The administered medication for placebo and intervention were identical in appearance. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome | Low risk of bias | Investigators were unaware of allocation. Method of measurement not stated but likely to be standard and unlikely to have differed between groups. | Low risk of bias | No protocol, statistical analysis plan or registration document available for review. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Measurement method not stated but likely standard. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome measurement method not stated but was unlikely to have differed between groups. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | High risk of bias | Data available for fewer than 50% of those randomised. | Low risk of bias | Measurements likely performed by standard methods. Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | High risk of bias | High risk as data available for <50% randomised. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Measurement done by standardised methods. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Low risk of bias | Low risk of bias for all domains |
Risk of bias for analysis 1.44 L3: Mean head circumference Z score at birth.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.44.1 In all babies | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.45 L4: Mean length at birth (cm).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.45.1 In all neonates | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Low risk of bias | One mother with a singleton pregnancy was lost to follow‐up before delivery. | Low risk of bias | an open‐label trial and blinding of outcome assessors was not described. Outcome assessment not able to be influenced by knowledge of treatment allocation | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | Some concerns | Some concerns overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label and some concerns of bias in selection of the reported result because of the limited detail of analysis methods and outcomes. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Measurement method not stated but likely standard. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome measurement method not stated but was unlikely to have differed between groups. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Measurements done by standardised methods. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Low risk of bias | Low risk of bias for all domains |
Risk of bias for analysis 1.46 L4: Mean length Z score at birth.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.46.1 In all neonates | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.47 L4: Length multiples of the mean at birth.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.47.1 In all neonates | ||||||||||||
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Measurements done using standardised methods. | Low risk of bias | Protocol with planned statistical analysis was available for review. Neonatal measurements were specified in protocol documentation but multiples of the mean was not but is unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains |
Risk of bias for analysis 1.48 L4: Length at birth adjusted for gestation (standardised mean difference).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.48.1 In all neonates | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Deidentified charts underwent blinded central review by 2 neonatologists. Measurements done using standardised methods. | Low risk of bias | Protocol with planned statistical analysis was available for review. Neonatal measurements were specified in protocol documentation but multiples of the mean was not but is unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains |
Risk of bias for analysis 1.49 L5i: Mean weight (g) at primary hospital discharge.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.49.1 In all infants | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Some concerns | Missing data for 6% without explanation. No sensitivity analysis performed. Loss differed between groups. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Some concerns | Some concerns as proportion of missing data is high for assessments done at discharge and may have been related to health status. |
Risk of bias for analysis 1.50 L5i: Mean weight Z score at primary hospital discharge.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.50.1 In all infants | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Some concerns | Missing data for 6% without explanation. No sensitivity analysis performed. Loss differed between groups. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Some concerns | Some concerns as proportion of missing data is high for assessments done at discharge and may have been related to health status. |
Risk of bias for analysis 1.51 L5ii: Mean head circumference (cm) at primary hospital discharge.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.51.1 In all infants | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Some concerns | Missing data for 7% without explanation. No sensitivity analysis performed. Loss differed between groups. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Some concerns | Some concerns as proportion of missing data is high for assessments done at discharge and may have been related to health status. |
Risk of bias for analysis 1.52 L5ii: Mean head circumference Z score at primary hospital discharge.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.52.1 In all infants | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Some concerns | Missing data for 7% without explanation. No sensitivity analysis performed. Loss differed between groups. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Some concerns | Some concerns as proportion of missing data is high for assessments done as early as discharge and may have been related to health status. |
Risk of bias for analysis 1.53 L5iii: Mean length (cm) at primary hospital discharge.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.53.1 In all infants | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Some concerns | Missing data for 12% without explanation. No sensitivity analysis performed. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Some concerns | Some concerns as proportion of missing data is high for assessments done as early as discharge and may have been related to health status. |
Risk of bias for analysis 1.54 L5iii: Mean length Z score at primary hospital discharge.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.54.1 In all infants | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome measurement unlikely to have differed between treatment groups as outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Some concerns | Missing data for 12% without explanation. No sensitivity analysis performed. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Some concerns | Some concerns as proportion of missing data is high for assessments done as early as discharge and may have been related to health status. |
Risk of bias for analysis 1.55 L6i: Mean weight at infant follow‐up (kg).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Some concerns | 44/64 of those randomised and not known to be dead were assessed for this outcome (69%). Sensitivity analysis not performed. | Low risk of bias | This was an open‐label trail and blinding of outcome assessors was not described. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, selection of the reported result because of the limited detail of analysis methods and outcomes, for measurement of the outcome as blinding of outcome assessors was not described and missing outcome data as only 44/64(69%) of those randomised and not known to be dead were assessed for this outcome. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | High risk of bias | Data available for 75/113(66%). No sensitivity analysis performed. High risk due to marked loss to follow‐up, although proportion of missing data was similar across the two groups. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | High risk of bias | High risk due to very significant levels of loss to follow‐up at this early stage (34%), although proportion of missing data was similar across the two groups. |
Risk of bias for analysis 1.56 L6i: Mean weight Z score at infant follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | High risk of bias | Data available for 75/113(66%). No sensitivity analysis performed. High risk due to marked loss to follow‐up, although proportion of missing data was similar across the two groups. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | High risk of bias | High risk due to very significant levels of loss to follow‐up at this early stage (34%), although levels of missing data were similar across the two groups. |
Risk of bias for analysis 1.57 L6ii: Mean head circumference at infant follow‐up (cm).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Some concerns | 44/64 of those randomised and not known to be dead were assessed for this outcome (69%). Sensitivity analysis not performed. | Low risk of bias | This was an open‐label trail and blinding of outcome assessors was not described. Outcome not likely to be affected by knowledge of allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, selection of the reported result because of the limited detail of analysis methods and outcomes, for measurement of the outcome as blinding of outcome assessors was not described and missing outcome data as only 44/64(69%) of those randomised and not known to be dead were assessed for this outcome. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | High risk of bias | Data available for 62/113(55%). No sensitivity analysis performed. High risk due to marked loss to follow‐up, although proportion of missing data was similar across the two groups. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | High risk of bias | High risk due to very significant levels of loss to followup (45%), although proportion of missing data was similar across the two groups. |
Risk of bias for analysis 1.58 L6ii: Mean head circumference Z score at infant follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis. | High risk of bias | Data available for 62/113(55%) . No sensitivity analysis performed. High risk due to marked loss to follow‐up, although proportion of missing data was similar across the two groups. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | High risk of bias | High risk due to very significant levels of loss to follow‐up (45%), although proportion of missing data was similar across the two groups. |
Risk of bias for analysis 1.59 L6iii: Mean length at infant follow‐up (cm).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Mazumder 2008 | Low risk of bias | Simple randomisation used a website‐generated random number list. Allocation slips were placed in opaque, sealed envelopes. Limited baseline characteristics were recorded. | Some concerns | The trial was open‐label. No deviations from the intended intervention were reported. Methods did not specify if analysis was by intention‐to‐treat or per protocol. | Some concerns | 44/64 of those randomised and not known to be dead were assessed for this outcome (69%). Sensitivity analysis not performed. | Low risk of bias | This was an open‐label trail and blinding of outcome assessors was not described but the result was unlikely to be influenced by knowledge of treatment allocation. | Some concerns | No SAP or trial registration available to review. Limited detail in analysis plans stated in methods. | High risk of bias | High risk overall as there were some concerns of risk of bias for deviations from intended interventions because the trial was open label, selection of the reported result because of the limited detail of analysis methods and outcomes, for measurement of the outcome as blinding of outcome assessors was not described and missing outcome data as only 44/64(69%) of those randomised and not known to be dead were assessed for this outcome. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis. | High risk of bias | Data available for 75/113(66%). No sensitivity analysis performed. High risk due to marked loss to follow‐up, although proportion of missing data was similar across the two groups. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | High risk of bias | High risk due to high levels of loss to follow‐up, although levels of missing data were similar across the two groups. SAP was not available for review. An interim analysis was performed by an independent data safety monitoring committee. |
Risk of bias for analysis 1.60 L6iii: Mean length Z score at infant follow‐up .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | High risk of bias | Data available for 75/113(66%). No sensitivity analysis performed. High risk due to marked loss to follow‐up, although proportion of missing data was similar across the two groups. | Low risk of bias | Investigators were unaware of treatment allocation. Method of measurement likely standard and unlikely to have differed between treatment groups. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | High risk of bias | High risk due to high levels of loss to follow‐up (34%), although proportion of missing data was similar across the two groups. |
Risk of bias for analysis 1.61 L8: Mean duration of invasive respiratory support (days).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome based on care administered by treating team as per case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis but statistical analysis plan not available to review. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2002 | Low risk of bias | Allocation was performed by the pharmacy using a randomisation table. The investigators and clinical care providers were unaware of the treatment provided and the placebo and intervention drug were noted to be identical in appearance. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical care providers were unaware of allocation. The administered medication for placebo and intervention were identical in appearance. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome | Low risk of bias | Outcome based on treatment administered by the treating team, who were blinded to allocation. | Low risk of bias | No protocol, statistical analysis plan or registration document available for review. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis. | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. Outcome based on treatment administered by the treating team, who were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.62 L9: Mean duration of non‐invasive respiratory support (days).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.63 L10: Mean duration of oxygen supplementation (days).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome based on care administered by treating team as per case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis but statistical analysis plan not available to review. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2002 | Low risk of bias | Allocation was performed by the pharmacy using a randomisation table. The investigators and clinical care providers were unaware of the treatment provided and the placebo and intervention drug were noted to be identical in appearance. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical care providers were unaware of allocation. The administered medication for placebo and intervention were identical in appearance. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome based on treatment administered by the treating team, who were blinded to allocation. | Low risk of bias | No protocol, statistical analysis plan or registration document available for review. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis. | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. Outcome based on treatment administered by the treating team, who were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 1.64 L14: Mean cord cortisol concentrations at birth (nmol/L).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 1.64.1 In all neonates | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Some concerns | 67/102 infants randomised at the site of this sub‐study had cord blood available. Reasons for missing data noted as insufficient sample, "missed" or infants not born at this site. | Some concerns | Outcome assessors were blinded to allocation. Measurement of cord cortisol is not a reliable measure of HPA axis function. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Some concerns due to missing outcome data and some concerns of bias in measurement of the outcome because cord cortisol is not a reliable measure of HPA axis function. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | Data available for approximately 76% of pregnancies. Sensitivity analysis not performed. Proportion of missing data was similar between intervention and placebo groups. | Some concerns | Blinding of assessors not described for this outcome but was likely, and results unlikely to be influenced by knowledge of treatment allocation. Measurement of cord cortisol is not a reliable measure of HPA axis function. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was specified in protocol documentation. | Some concerns | Some concerns as data available for only 76% of pregnancies but the proportion of missing data was similar between intervention and placebo groups. Some concerns for measurement as cord cortisol is not a reliable measure of HPA axis function. |
Risk of bias for analysis 2.1 B1: Maternal death.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.1.1 All women | ||||||||||||
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The injection of the study treatment was given by a designated research nurse who was not caring for the woman. The physical appearance of the study solutions was masked. Intention‐to‐treat analysis was performed. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | The treatment was masked and outcome assessors were blinded. Outcome measure was objective. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration documentation. Outcome definition and analysis unlikely to have been selected based on results and has limited was this could be reported and analysed. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.2 B2: Maternal sepsis.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.2.1 In all women | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as diagnosed by the treating team. Case definition was not stated. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Diagnosis of the outcome was as recorded by the treating team in case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The injection of the study treatment was given by a designated research nurse who was not caring for the woman. The physical appearance of the study solutions was masked. Intention‐to‐treat analysis was performed. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Definition of sepsis unclear but ascertainment was unlikely to have differed between groups as it was based on diagnosis by the clinical team who were blinded to allocation. The treatment was masked and outcome assessors were blinded. | Some concerns | SAP and protocol were not available for review. Outcome was specified but not defined in trial registration documentation. Outcome definition not in methods and has multiple possible definitions. | Some concerns | Some concerns overall due to some concerns of risk of bias in selection of the reported results because the outcome was not defined and could have been selected from multiple definitions. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. The definition of this outcome was unclear but with the treating team blinded it is unlikely to have varied between treatment groups. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | <1% loss to follow‐up. | Low risk of bias | Outcome determined based on clinical, radiological and laboratory data. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. Outcome clearly defined in methods. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Ascertainment of outcome from clinical record unlikely to have differed between groups as treating team was blinded. Study investigators were also blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 1/82 in the intervention group and 3/80 in the placebo group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Assessors were blinded to allocation. Outcome ascertained from case notes. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.3 B3: Caesarean section.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.3.1 In all women | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as based on treatment administered by the treating team, who were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. This outcome is reported as standard practice. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome based on treatment administered by the treating team, who were blinded to allocation. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The injection of the study treatment was given by a designated research nurse who was not caring for the woman. The physical appearance of the study solutions was masked. Intention‐to‐treat analysis was performed. | Low risk of bias | Data available for 95% for this outcome. | Low risk of bias | Outcome determined by a treatment decision by the treating team, who were blinded to allocation. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration documentation but outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis | Low risk of bias | All women had data for this outcome. | Low risk of bias | Investigators were unaware of treatment allocation. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | <1% missing data for this outcome. | Low risk of bias | Outcome based on care administered by treating team. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was not in the trial registration but this outcome is reported as standard practice and is unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 1/82 in the intervention group and 3/80 in the placebo group. | Low risk of bias | Based on registration document and trial centre it is likely assessors were blinded. Objective outcome. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Assessors were blinded to allocation. Outcome ascertained from case notes. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was not in protocol documentation but is routinely reported in trials and unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.4 B4: Discontinuation of therapy due to maternal adverse effects.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.4.1 In all women | ||||||||||||
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. It is unlikely that this outcome could be markedly impacted by knowledge of the intervention. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.5 G1: Puerperal sepsis.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.5.1 In all women | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as diagnosed by the treating team. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. The definition of this outcome was unclear but with the treating team blinded it is unlikely to have varied between treatment groups. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | <1% missing data for this outcome. | Low risk of bias | Outcome determined based on clinical, radiological and laboratory data. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. Outcome clearly defined in methods. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Ascertainment of outcome from clinical record unlikely to have differed between groups as treating team was blinded. Study investigators were also blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 1/82 in the intervention group and 4/80 in the placebo group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Assessors were blinded to allocation. Outcome ascertained from case notes. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation but is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.6 G2: Chorioamnionitis during labour.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.6.1 In all women | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as diagnosed by the treating team. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Diagnosis of the outcome was as recorded by the treating team in case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome definition not clear but ascertainment unlikely to have differed between treatment groups as the treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration documentation but outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. The definition of this outcome was unclear but with the treating team blinded it is unlikely to have varied between treatment groups. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | <1% missing data for this outcome. | Low risk of bias | Outcome determined based on clinical and laboratory data. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. Outcome clearly defined in methods. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 1/82 in the intervention group and 3/80 in the placebo group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Assessors were blinded to allocation. Outcome ascertained from case notes. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation but is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.7 G3: Endometritis.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.7.1 In all women | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as diagnosed by the treating team. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. The definition of this outcome was unclear but with the treating team blinded it is unlikely to have varied between treatment groups. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | <1% missing data for this outcome. | Low risk of bias | Outcome determined based on clinical and laboratory data. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. Outcome clearly defined in methods. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Assessors were blinded to allocation. Outcome ascertained from case notes. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation but is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.8 G4: Pyrexia after trial entry requiring the use of antibiotics.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.8.1 In all women | ||||||||||||
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 1/82 in the intervention group and 3/80 in the placebo group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. Objective outcome. Definition of pyrexia not clear. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.9 G6: Postpartum haemorrhage.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.9.1 In all women | ||||||||||||
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. The definition of this outcome was unclear but with the treating team blinded it is unlikely to have varied between treatment groups. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 1/82 in the intervention group and 3/80 in the placebo group. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.10 G7: Postnatal pyrexia .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.10.1 In all women | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as reported in the case notes. Threshold of 38 degrees Celsius for pyrexia. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.11 G8: Prelabour rupture of membranes after trial entry.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.11.1 In all women | ||||||||||||
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. The definition of this outcome was unclear but with the treating team blinded it is unlikely to have varied between treatment groups. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Assessors were blinded to allocation. Outcome ascertained from case notes. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation but is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.12 G9: Mode of birth: vaginal birth.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.12.1 In all women | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome was as based on treatment administered by the treating team. Treating team were blinded to allocation of patients. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. This outcome is reported as standard practice. | Low risk of bias | Low risk of bias for all domains. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome based on treatment administered by the treating team, who were blinded to allocation. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Garite 2009 | Low risk of bias | A blocked, computer generated randomisation sequence was used that was prepared centrally. The research pharmacist (unblinded) at each site prepared the medication based on the randomisation sequence. No marked imbalance of baseline characteristics. | Low risk of bias | The study syringes (betamethasone or saline) were completely covered by a label to conceal the contents after preparation by a research pharmacist. A modified intention‐to‐treat analysis was used. | Low risk of bias | Data available for 95% for this outcome. | Low risk of bias | Outcome determined by a treatment decision by the treating team, who were blinded to allocation. The treatment was masked and outcome assessors were blinded. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration documentation but outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | <1% missing data for this outcome. | Low risk of bias | Outcome based on care provided by treating team. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Intention‐to‐treat analysis. | Low risk of bias | No missing data reported for this outcome. | Low risk of bias | Outcome based on care administered by the treating team, who were blinded to allocation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was not in the trial registration but this outcome is reported as standard practice and is unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Assessors were blinded to allocation. Outcome ascertained from case notes. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was not in protocol documentation but is routinely reported in trials and unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.13 G10: Hypertension .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.13.1 In all women | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Blood pressure was as recorded in case notes. Threshold of 140/90mmHg as threshold for hypertension. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | <1% loss to follow‐up ("one or two" values missing from this group). | Low risk of bias | Outcome as reported from case notes. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | >99% had data for this outcome. | Low risk of bias | Assessors were blinded to allocation. Outcome ascertained from case notes. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.14 G12: Glucose intolerance.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.14.1 In all women | ||||||||||||
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | <1% loss to follow‐up ("one or two" values missing from this group). | Low risk of bias | Outcome as reported from case notes. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | Data was available for 186/250(74%) in the intervention and 172/242(71%) for the placebo group. No sensitivity analysis. The amount of missing data was similar in both groups. | Low risk of bias | Measured from 1 hour glucose tolerance test. Assessors were blinded to allocation. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. Definition of abnormal GTT not provided but unlikely to have been chosen based on results. | Some concerns | Some concerns due to missing data as data available for only 72% of those randomised. |
Risk of bias for analysis 2.15 G13: Postnatal depression.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | 847/935(91%) in intervention group and 824/918(90%) in placebo group had data for this outcome. | Low risk of bias | Outcome obtained from maternal questionnaire. Researchers and participants associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. Outcome was reported using a standardised tool for assessing postnatal depression. . | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.16 G14: Local injection site adverse effects .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Injection adverse effects were recorded at the time of injection by the treating team. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Adverse effects were self reported by women but the treatment allocation was masked. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was not in protocol documentation but is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.17 G15: Insomnia after treatment.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 2.17.1 In all women | ||||||||||||
| Aghajafari 2002 | Low risk of bias | Randomisation sequence computer‐generated and centrally controlled by 1 pharmacist at each hospital who kept the randomisation code. Stratification by gestational age and by hospital. Baseline variation was as expected given the small sample size. | Low risk of bias | The allocated treatment was masked and intention‐to‐treat analysis was performed. | Some concerns | 1 out of 6 women in the intervention group did not complete a questionnaire. | Low risk of bias | Measurement was via a structured patient questionnaire. Participants were blinded to treatment allocation. | Low risk of bias | Statistical plan, protocol or trial registration document not available but outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Some concerns overall as some concerns of missing data because 1 out of 6 women in the intervention group did not complete a questionnaire and insomnia could have influenced this although participants were blinded to treatment allocation. |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Injection adverse effects were recorded by the treating team. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | 93% in intervention group and 92% in placebo group had data for this outcome. | Low risk of bias | Outcome obtained from maternal questionnaire. Researchers and participants associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. The questionnaire used to obtain data for this outcome was published with the results and data was reported as expected. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Adverse effects were self reported by women but the treatment allocation was masked. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was not in protocol documentation but is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 2.18 G16: Gastrointestinal adverse effects of treatment .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Adverse effects were self reported by women but the treatment allocation was masked. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was not in protocol documentation but is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.1 C1: Total deaths (after randomisation) up to early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.1.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were masked to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome assessment was objective. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | >90% follow‐up at 2 years. | Low risk of bias | Objective outcome. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | Death was a prespecified outcome in the trial registration. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Low risk of bias | Loss to follow‐up of 16% but mortality outcome appeared to be known for almost all randomised. | Low risk of bias | Study investigators were blinded to allocation. The outcome was objective. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Low risk of bias | Missing data for 3/94 (3.2%) in both groups. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. Objective outcome. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those randomised had data available. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.2 C2: Neurodevelopmental impairment at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.2.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 92% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Bayley Scales of infant development assessed by a psychologist and neurological examination performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | 91% of children were followed up for this outcome. | Low risk of bias | Assessment of the outcome was by a standardised neurological examination and standardised cognitive testing. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | High risk of bias | Only 68% of those randomised had data for this outcome. No sensitivity analysis performed. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. Developmental assessment by Bayley Scales of infant development specified in registration document. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | High risk of bias | High risk as only 68% of those randomised had data for this outcome. No sensitivity analysis performed. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | 80% of those alive at 2 year follow‐up had Bayley scales assessed (78% of those randomised). No sensitivity analysis performed. The amount of missing data was similar between groups. Missing participants differed slightly in proportions of different ethnic groups. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. Assessment included physical and neurological examination and Bayley scales of infant development. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Some concerns | Some concerns overall due to missing data (20% missing data). |
Risk of bias for analysis 3.3 C3: Survival free of neurodevelopmental impairment at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.3.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 92% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Bayley Scales of infant development assessed by a psychologist and neurological examination performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | 91% of children were followed up for this outcome. | Low risk of bias | Assessment of the outcome was by a standardised neurological examination and standardised cognitive testing. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | High risk of bias | data on neurodevelopmental outcome only available in 68%. | Low risk of bias | Based on registration document and trial centre it is likely assessors were blinded. Developmental assessment by Bayley Scales of infant development specified in registration document. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | High risk of bias | High risk as only 68% of those randomised had data for neurodevelopmental impairment. No sensitivity analysis performed. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | 80% of those alive at 2 year follow‐up had Bayley scales assessed (78% of those randomised). No sensitivity analysis performed. The amount of missing data was similar between groups. Missing participants differed slightly in proportions of different ethnic groups. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. Assessment included physical and neurological examination and Bayley scales of infant development. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Some concerns | Some concerns due to missing data for 20%. |
Risk of bias for analysis 3.4 C4: Survival free of major neurodevelopmental impairment at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.4.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 92% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Bayley Scales of infant development assessed by a psychologist and neurological examination performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | High risk of bias | Loss to follow‐up was 19% in the intervention group and 14% in the placebo group but the proportion with death or severe neurodevelopmental impairment was lower than this (9% and 4%). No sensitivity analysis performed. | Low risk of bias | Developmental assessments were performed by a neuropsychologist and either a paediatrician or paediatric neurologist. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | High risk of bias | High risk overall due to high risk of bias due to missing outcome data because of differential loss to follow up in the treatment groups and losses exceeded the number with the outcome. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | 80% of those alive at 2 year follow‐up had Bayley scales assessed (78% of those randomised). No sensitivity analysis performed. The amount of missing data was similar between groups. Missing participants differed slightly in proportions of different ethnic groups. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. Assessment included physical and neurological examination and Bayley scales of infant development. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Some concerns | Some concerns of risk of bias due to missing data. |
Risk of bias for analysis 3.5 C5: Cerebral palsy at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.5.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 92% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Neurological examination was performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. Cerebral palsy definition was standard. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | 91% of children were followed up for this outcome. | Low risk of bias | Outcome was assessed by a standardised neurological examination. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | High risk of bias | Loss to follow‐up was 19%in the intervention group and 14% in the placebo group, much higher than the proportion with the outcome of CP. No sensitivity analysis performed. | Low risk of bias | Neurological examination was performed by a paediatrician or a paediatric neurologist blinded. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was not specifically mentioned in the trial registration but "abnormalities of neurological status" were. Outcome definition and analysis unlikely to have been selected based on results. | High risk of bias | High risk overall due to high risk of bias due to missing outcome data because there was differential loss to follow‐up and losses exceeded the number with cerebral palsy. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | High risk of bias | 68% of those randomised had data for this outcome. Similar levels of missing data in both groups. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | High risk of bias | Only 68% of those randomised had data for this outcome. Similar proportion of missing data in both groups. Overall high risk as missing data may be more likely to include those with the outcome. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). Similar proportion of missing data in each group. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. Assessment included physical and neurological examination. | Some concerns | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation. Analysis plans were included in the methods but not in sufficient detail to explain the rationale behind multiple subgroup analyses being reported involving cerebral palsy that may not have been prespecified. | Some concerns | Some concerns in selection of reported result because analysis plans were included in the protocol but not in sufficient detail to explain the rationale behind multiple subgroup analyses being reported involving cerebral palsy that may not have been prespecified. Other domains low risk. |
Risk of bias for analysis 3.6 C6: Developmental delay or intellectual impairment at early childhood follow‐up (any).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.6.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 92% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Bayley Scales of infant development assessed by a psychologist. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. Cut‐off levels for developmental delay (on Mental development index) were those in standard usage. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | 86% of infants who were alive had an assessment for this outcome. | Low risk of bias | Assessment of the outcome was by standardised cognitive testing. Most were assessed using Bayley scales of infant development. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. Cut‐off of MDI <70 defined in trial registration document. | Low risk of bias | Low risk of bias for all domains |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | High risk of bias | Of those that completed follow‐up, only 73% of the intervention and 77% of the placebo group (59% of those randomised) had data for this outcome. No sensitivity analysis performed. No explanation provided for the difference in proportion with full psychological testing. | Low risk of bias | The Bayley scales of infant development were assessed by a neuropsychologist. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | High risk of bias | High risk overall due to high risk of bias due to missing outcome data as there was differential loss to follow‐up in the treatment groups and only 59% had data for this outcome. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | 80% of those alive at 2 year follow‐up had Bayley scales assessed (78% of those randomised). No sensitivity analysis performed. The amount of missing data was similar between groups. Missing participants differed slightly in proportions of different ethnic groups. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. Assessment used Bayley scales of infant development. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. All expected scales were reported (PDI and MDI) | Some concerns | Some concerns of risk of bias due to missing data. |
Risk of bias for analysis 3.7 Mental Developmental Index at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.7.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 89% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Bayley Scales of infant development assessed by a psychologist. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | High risk of bias | Of those that completed follow‐up, only 73% of the intervention and 77% (Overall 59% of those randomised) of the placebo group had data for this outcome. No sensitivity analysis performed. No explanation for the difference in proportion with full psychological testing. | Low risk of bias | The Bayley scales of infant development were assessed by a neuropsychologist. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | High risk of bias | High risk overall due to high risk of bias due to missing outcome data as there was differential loss to follow‐up in the treatment groups and only 59% had data for this outcome. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | 80% of those alive at 2 year follow‐up had Bayley scales assessed (78% of those randomised). No sensitivity analysis performed. The amount of missing data was similar between groups. Missing participants differed slightly in proportions of different ethnic groups. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Some concerns | Some concerns of risk of bias due to missing data. |
Risk of bias for analysis 3.8 H1: Child behaviour at early childhood follow‐up, Child Behaviour Checklist total score in the clinical range .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 92% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Child behaviour checklist was completed by parent or caregiver. Caregivers, parents and research staff were all blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. Using the top 2.5% as the clinical range is standard. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.9 H1: Child behaviour: Behaviour rating scale in the clinical range (BSID‐II).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | 80% of those randomised who were still alive had an assessment of this outcome. | Low risk of bias | Assessed using the behaviour rating scale in the Bayley scales of infant development. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome (and use of BSID) was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. The cut‐off used to define nonoptimal or within normal limits was not specified. | Some concerns | Some concerns due to missing data, as only 80% of those randomised who were still alive had an assessment of this outcome. |
Risk of bias for analysis 3.10 H1: Child behaviour at early childhood follow‐up as assessed by Early Child Behaviour Questionnaire Extraversion summary scale.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Low risk of bias | Although this group was a subset of the original study from one centre (71% of the total randomised) most of the group were followed up for this outcome (85% of those alive). | Low risk of bias | Assessed by parental completion of the Early Child Behaviour questionnaire. Parents and study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration but not the scale used. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.11 H1: Child behaviour at early childhood follow‐up assessed by Early Child Behaviour questionnaire Negative affectivity summary scale.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Low risk of bias | Although this group was a subset of the original study from one centre (71% of the total randomised) most of the group were followed up for this outcome (85% of those alive). | Low risk of bias | Assessed by parental completion of the Early Child Behaviour questionnaire. Parents and study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration but not the scale used. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.12 H1: Child behaviour at early childhood follow‐up assessed by Early Child Behaviour questionnaire Effortful control summary scale.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Low risk of bias | Although this group was a subset of the original study from one centre (71% of the total randomised) most of the group were followed up for this outcome (85% of those alive). | Low risk of bias | Assessed by parental completion of the Early Child Behaviour questionnaire. Parents and study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration but not the scale used. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.13 H2: Psychomotor Developmental Index at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.13.1 In all babies | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 88% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Bayley Scales of infant development assessed by a psychologist. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Some concerns | 80% of those alive at 2 year follow‐up had Bayley scales assessed (78% of those randomised). No sensitivity analysis performed. The amount of missing data was similar between groups. Missing participants differed slightly in proportions of different ethnicities. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | SAP not available for review. Analysis plans were included in the methods. This outcome was prespecified. | Some concerns | Some concerns due to missing data. Similar proportion of missing data in each group. Despite lack of SAP to review, outcome was well defined in the methods section and outcome was prespecified. |
Risk of bias for analysis 3.14 H3: Deafness/hearing impairment at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.14.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Hearing was assessed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | >90% follow‐up. | Low risk of bias | Method of measurement by clinical examination. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Loss to follow up‐was 19% in the intervention group and 14% in the placebo group. No sensitivity analysis performed. Documented reasons for loss to follow‐up were parental refusal or moving to an unknown address. | Low risk of bias | Medical history was obtained by a questionnaire. Parents/caregivers were blinded to allocation. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Some concerns overall due to some concerns of risk of bias due to missing outcome data because no events were seen for this outcome and the proportion of missing data was different between treatment groups. |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Some concerns | 68% of those randomised had data for this outcome. No sensitivity analysis. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Some concerns | Some concerns as only 68% of those randomised had data for this outcome. No sensitivity analysis. |
Risk of bias for analysis 3.15 H4: Blindness/visual impairment at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.15.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Vision was assessed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. Definition of blindness was standard. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | >90% follow‐up. | Low risk of bias | Method of measurement by clinical examination. . Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | Although SAP was not reviewed, detailed analysis plans were provided in the methods and were unlikely to have been chosen on the basis of results. Outcome definition prespecified in registration document. | Low risk of bias | Low risk of bias for all domains |
| TEAMS 1999 | Low risk of bias | Baseline information not available in detail. Limited information but based on registration document and trial centre standard protocols this was likely performed with random sequence generation and allocation concealment. | Low risk of bias | Limited information but based on registration document and trial centre likely participants were blinded. No analysis was published as the trial was stopped early. | Some concerns | 68% of those randomised had data for this outcome. No sensitivity analysis. | Low risk of bias | Limited information but based on registration document and trial centre likely assessors were blinded. | Low risk of bias | No analyses were published by author as trial stopped early. Unpublished trial data were included. | Some concerns | 68% of those randomised had data for this outcome. No sensitivity analysis. |
Risk of bias for analysis 3.16 H5: Hypertension at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.16.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | Approximately 58% of participants alive at follow‐up had assessments data for this outcome. Each group had similar proportions of missing data. | Low risk of bias | Blood pressure measured by a paediatrician according to local practice and hypertension defined as systolic or diastolic pressure above the 95th centile. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. The definition used is a standard definition for childhood hypertension. | Some concerns | Some concerns of bias due to missing data as only 58% of participants alive at follow‐up had assessments data for this outcome but treatment groups had similar proportions of missing data. |
Risk of bias for analysis 3.17 H8: Asthma or recurrent wheeze at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.17.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 98% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Outcome was assessed via a parental questionnaire. Parents were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Loss to follow‐up was 19%in the intervention group and 14% in the placebo group. No sensitivity analysis performed. The proportion of missing data was different between the groups but documented reasons for loss to follow‐up were parental refusal or moving to an unknown address. | Some concerns | Outcome assessors and parents were blinded to allocation. As this outcome is dependent on parental memory of a diagnosis, it is likely unreliable. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Overall some concerns due to some concerns of risk of bias due to missing outcome data because loss to follow‐up differed between groups and some concerns of risk of bias in the measurement of the outcome. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments(82% of those randomised). Similar proportion of missing data in intervention and placebo groups. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. Outcome ascertained from a detailed medical history. | Some concerns | Protocol available but did not include this outcome. Outcome in methods but appears to have been selected from a section on "chronic conditions" from an interview. It is unclear if the outcome or method of defining asthma was prespecified. | Some concerns | Some concerns for selection of the reported result. Protocol available but did not include this outcome. Outcome in methods but appears to have been selected from a section on "chronic conditions" from an interview. It is unclear if the outcome or method of defining asthma was prespecified. Other domains low risk. |
Risk of bias for analysis 3.18 H9: Any respiratory disease at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 98% of those alive at follow‐up had an assessment of this outcome. | Some concerns | Although outcome assessors were blinded to allocation, parental recall of reasons for hospital admissions may be unreliable. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Some concerns in the measurement of the outcome because, despite parents being blinded to allocation, parental recall of reasons for hospital admissions may be unreliable. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | >90% follow‐up. | Low risk of bias | Method of measurement not stated but unlikely to have differed between groups due to blinding. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Loss to follow‐up was 19%in the intervention group and 14% in the placebo group. No sensitivity analysis performed. The proportion of missing data was different between the groups but documented reasons for loss to follow‐up were parental refusal or moving to an unknown address. | Some concerns | Outcome assessors and parents were blinded to allocation. As this outcome is dependent on parental memory of a diagnosis, it is likely unreliable. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Overall some concerns due to some concerns of risk of bias due to missing outcome data because loss to follow‐up differed between groups and some concerns of risk of bias in the measurement of the outcome. |
Risk of bias for analysis 3.19 N1i: Mean weight at early childhood follow‐up (kg).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.19.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | Data available for 88% of those randomised. | Low risk of bias | Method of measurement not stated but likely standard methods were used. Researchers associated with the trial remained unaware of allocation up to 5 year followup. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Missing data for this outcome was similar between the two groups (24% and 22% of those alive in the intervention and placebo groups respectively). No sensitivity analysis performed. | Low risk of bias | Method of measurement not stated but is unlikely to differ between treatment groups. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Overall some concerns due to some concerns of risk of bias due to missing outcome data because data was missing for 23% of those alive for follow‐up but with similar levels of missing data between groups. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments(82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.20 N1i: Mean weight Z score at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.20.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.21 N1i: Mean weight adjusted for age at early childhood follow‐up (standardised mean difference).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.21.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow up‐had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Missing data for this outcome for this outcome was similar between the two groups (24% and 22% of those alive in the intervention and placebo groups respectively). No sensitivity analysis performed. | Low risk of bias | Method of measurement not stated but is unlikely to differ between treatment groups. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Overall some concerns due to some concerns of risk of bias due to missing outcome data because data was missing for 23% of those alive for follow‐up but with similar levels of missing data between groups. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.22 N1i: Weight small for age at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.22.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.23 N1ii: Mean head circumference at early childhood follow‐up (cm).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.23.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. One woman in the repeat antenatal steroids group had allocation unmasked at the request of her physician but completed the trial. Intention‐to‐treat analysis was used. | Low risk of bias | Data available for 88% of those randomised. | Low risk of bias | Method of measurement not stated but likely standard methods were used. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Missing data for this outcome for this outcome was similar between the two groups (24% and 22% of those alive in the intervention and placebo groups respectively). No sensitivity analysis performed. | Low risk of bias | Method of measurement not stated but is unlikely to differ between treatment groups. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Overall some concerns due to some concerns of risk of bias due to missing outcome data because data was missing for 23% of those alive for follow‐up but with similar levels of missing data between groups. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.24 N1ii: Mean head circumference Z score at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.24.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Methods state prespecified analysis but statistical analysis plan not available to review. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Missing data for this outcome for this outcome was similar between the two groups (24% and 22% of those alive in the intervention and placebo groups respectively). No sensitivity analysis performed. | Low risk of bias | Study investigators were blinded to allocation. | Low risk of bias | SAP not available for review but analysis plans were included on the registered trial protocol. | Some concerns | Missing data for 23% of those alive for follow‐up but similar levels of missing data between groups. |
Risk of bias for analysis 3.25 N1ii: Mean head circumference adjusted for age at early childhood follow‐up (standardised mean difference).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.25.1 In all babies | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Missing data for this outcome for this outcome was similar between the two groups (24% and 22% of those alive in the intervention and placebo groups respectively). No sensitivity analysis performed. | Low risk of bias | Method of measurement not stated but is unlikely to differ between treatment groups. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Overall some concerns due to some concerns of risk of bias due to missing outcome data because data was missing for 23% of those alive for follow‐up but with similar levels of missing data between groups. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.26 N1ii: Head circumference small for age at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.26.1 In all babies | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Methods state prespecified analysis but statistical analysis plan not available to review. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.27 N1iii: Mean height at early childhood follow‐up (cm).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.27.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | Data available for 88% of those randomised. | Low risk of bias | Method of measurement not stated but likely standard methods were used. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Missing data for this outcome for this outcome was similar between the two groups (24% and 22% of those alive in the intervention and placebo groups respectively). No sensitivity analysis performed. | Low risk of bias | Method of measurement not stated but is unlikely to differ between treatment groups. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Overall some concerns due to some concerns of risk of bias due to missing outcome data because data was missing for 23% of those alive for follow‐up but with similar levels of missing data between groups. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was not in protocol documentation but is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.28 N1iii: Height Z score at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.28.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Missing data for this outcome for this outcome was similar between the two groups (24% and 22% of those alive in the intervention and placebo groups respectively). No sensitivity analysis performed. | Low risk of bias | Study investigators were blinded to allocation. | Low risk of bias | SAP not available for review but analysis plans were included on the registered trial protocol. Outcome defined in trial registration prior to follow‐up study. | Some concerns | Missing data for 23% of those alive for follow‐up but similar levels of missing data between groups. |
Risk of bias for analysis 3.29 N1iii: Mean height adjusted for age at early childhood follow‐up (standardised mean difference).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.29.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Missing data for this outcome for this outcome was similar between the two groups (24% and 22% of those alive in the intervention and placebo groups respectively). No sensitivity analysis performed. | Low risk of bias | Method of measurement not stated but is unlikely to differ between treatment groups. Study investigators were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Overall some concerns due to some concerns of risk of bias due to missing outcome data because data was missing for 23% of those alive for follow‐up but with similar levels of missing data between groups. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was not in protocol documentation but is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.30 N1iii: Height small for age at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.30.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 96% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was not in protocol documentation but is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.31 N3: Mean systolic blood pressure at early childhood follow‐up (mmHg).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.31.1 In all children | ||||||||||||
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.32 N3: Mean systolic blood pressure Z score at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.32.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 62% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Some concerns of risk or bias due to missing outcome data as only 62% of those alive at follow‐up had an assessment of this outcome. Intervention and placebo groups had similar proportions of missing data. |
Risk of bias for analysis 3.33 N3: Mean diastolic blood pressure at early childhood follow‐up (mmHg).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.33.1 In all children | ||||||||||||
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessments (82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | Assessments were performed by centrally trained and certified study personnel who were unaware of the treatment assignment. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation and is unlikely to have been selected on the basis of results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 3.34 N3: Mean diastolic blood pressure Z score at early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 3.34.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 58% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Measurements performed by a developmental paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Some concerns of risk or bias due to missing outcome data as only 58% of those alive at follow‐up had an assessment of this outcome. Treatment groups had similar proportions of missing data. |
Risk of bias for analysis 4.1 D1: Total deaths (after randomisation) up to mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.1.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | >85% of those randomised had data for this outcome. | Low risk of bias | Outcome assessors were blinded to allocation. Outcome is objective. | Low risk of bias | Prospective data analysis plan included in protocol. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | >80% follow‐up at 5 years | Low risk of bias | The outcome was objective. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 4.2 D2: Neurocognitive impairment at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.2.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 78% of those randomised had assessments of this outcome. Outcome occurred in 16‐17%. No sensitivity analysis for missing data. Similar levels of missing data in both intervention and placebo group. | Low risk of bias | Outcome measurement performed by a pediatrician and psychologist unaware of allocation. Outcome assessors were blinded to allocation. | Low risk of bias | Prospective data analysis plan included in protocol. | Some concerns | Only 78% of those randomised had assessments of this outcome. Outcome occurred in only 16‐17%, a smaller proportion than the missing data. Similar levels of missing data in both intervention and placebo group. |
Risk of bias for analysis 4.3 D3: Survival free of neurocognitive impairment at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.3.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 78% of those randomised had data for this outcome. No sensitivity analysis for missing data. Loss to follow‐up was similar in both intervention and placebo groups. | Low risk of bias | Outcome measurement performed by a pediatrician and psychologist unaware of allocation. Outcome assessors were blinded to allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Some concerns | 78% of those randomised had assessments of this outcome. Loss to follow‐up was similar in both intervention and placebo groups. |
Risk of bias for analysis 4.4 D4: Survival free of major neurocognitive impairment at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.4.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 78% of those randomised had data for this outcome. No sensitivity analysis for missing data. Loss to follow‐up was similar in both intervention and placebo groups. | Low risk of bias | Outcome measurement performed by a pediatrician and psychologist unaware of allocation. Outcome assessors were blinded to allocation. | Low risk of bias | Prospective data analysis plan included in protocol. | Some concerns | 78% of those randomised had assessments of this outcome. Loss to follow‐up was similar in both intervention and placebo groups. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 75% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Assessment included a neurological examination for visual/hearing impairment and cerebral palsy along with 2 parental questionnaires. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data for 25% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.5 D6: Cognitive impairment at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.5.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 78% of those randomised had data for this outcome. No sensitivity analysis for missing data. Loss to follow‐up was similar in both intervention and placebo groups. | Low risk of bias | Outcome measurement performed by a psychologist unaware of allocation. Outcome assessors were blinded to allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Some concerns | 78% of those randomised had data for this outcome. Loss to follow‐up was similar in both intervention and placebo groups. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 75% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Assessment included the Behaviour rating inventory of executive function. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns as data not available for 25% of those randomised. Sensitivity analysis was not performed. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.6 D8: Cerebral palsy at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.6.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | >85% of those randomised had data for this outcome. | Low risk of bias | Outcome measurement performed by a pediatrician unaware of allocation. Outcome assessors were blinded to allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Low risk of bias | >85% of those randomised had data for this outcome. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 75% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Outcome was assessed by a neurological examination. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data for 25% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.7 I2: Blindness/visual impairment at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.7.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 78% of those randomised had data for this outcome. No sensitivity analysis for missing data. Proportion of missing data similar in intervention and placebo groups. | Low risk of bias | Outcome assessed by a pediatrician blinded to allocation. Outcome assessors were blinded to allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Some concerns | 78% of those randomised had data for this outcome. Proportion of missing data similar in intervention and placebo groups. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 75% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Outcome assessed as part of a targeted neurological examination. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data for 25% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.8 I3: Deafness/hearing impairment at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.8.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 78% of those randomised had data for this outcome. No sensitivity analysis performed. Similar levels of missing data in both intervention and placebo group. | Low risk of bias | Outcome assessed by a pediatrician blinded to allocation. Outcome assessors were blinded to allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Some concerns | 78% of those randomised had data for this outcome. Similar levels of missing data in both intervention and placebo group. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 75% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Outcome assessed as part of a targeted neurological examination. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data for 25% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.9 I4: Abnormal child behaviour at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 75% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Assessed by a parental questionnaire (Child behaviour checklist 1.5‐5year). Parents and researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. Outcome clearly defined in methods but the score cut‐off was not specified in trial registration. | Some concerns | Some concerns due to missing data for 25% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.10 I4: Child behaviour at mid‐ to later childhood follow‐up (standardised mean difference).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally and masked treatment packs were provided. Analysis was by intention‐to‐treat. | Some concerns | 75% of those randomised had data for this outcome. No sensitivity analysis for missing data. Proportion of missing data similar in intervention and placebo groups. | Low risk of bias | Outcome assessed by a parent/caregiver unaware of allocation. Outcome assessors were blinded to allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Some concerns | 75% of those randomised had data for this outcome. Proportion of missing data similar in intervention and placebo groups. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 75% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Assessed by a parental questionnaire (Child behaviour checklist 1.5‐5year). Parents and researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data for 25% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.11 I5: Asthma or recurrent wheeze at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.11.1 In all babies | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally and masked treatment packs were provided. Analysis was by intention‐to‐treat. | Low risk of bias | >85% of those randomised had data for this outcome. | Low risk of bias | Medical history collected as part of an assessment by a paediatrician. Outcome assessors were blinded to allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Low risk of bias | Low risk of bias for all domains |
Risk of bias for analysis 4.12 I6: Any respiratory disease at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally and masked treatment packs were provided. Analysis was by intention‐to‐treat. | Low risk of bias | >85% of those randomised had data for this outcome. | High risk of bias | Although parents were blinded to allocation, parental recall of reasons for hospital admissions may be unreliable. | Low risk of bias | Prospective statistical analysis plan included in protocol. | High risk of bias | Although outcome assessors were blinded to allocation, parental recall of reasons for hospital admissions may be unreliable and the number of respiratory admissions since neonatal discharge was lower at 6‐8 years than when assessed at 2 years. |
Risk of bias for analysis 4.13 O1i: Mean weight at mid‐ to later childhood follow‐up (kg).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.13.1 In all children | ||||||||||||
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 74% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Methods of measurement not stated but performed by trained staff unaware of allocation and likely used standard methods. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data for 26% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.14 O1i: Mean weight Z score at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.14.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally and masked treatment packs were provided. Analysis was by intention‐to‐treat. | Low risk of bias | 82% of those randomised had data for this outcome. | Low risk of bias | Outcome measurement performed by a pediatrician unaware of allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 4.15 O1ii: Mean head circumference at mid‐ to later childhood follow‐up (cm).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.15.1 In all children | ||||||||||||
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 74% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Methods of measurement not stated but performed by trained staff unaware of allocation and likely used standard methods. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data for 26% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.16 O1ii: Head circumference Z score at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.16.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally and masked treatment packs were provided. Analysis was by intention‐to‐treat. | Some concerns | 77% of those randomised had data for this outcome. No sensitivity analysis for missing data. Similar levels of loss to follow‐up in intervention and placebo group. | Low risk of bias | Outcome measurement performed by a pediatrician unaware of allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Some concerns | 77% of those randomised had data for this outcome. Similar levels of loss to follow‐up in intervention and placebo group. |
Risk of bias for analysis 4.17 O1iii: Mean height at mid‐ to later childhood follow‐up (cm).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.17.1 In all children | ||||||||||||
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 74% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Methods of measurement not stated but performed by trained staff unaware of allocation and likely used standard methods. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data for 26% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.18 O1iii: Mean height Z score at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.18.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally and masked treatment packs were provided. Analysis was by intention‐to‐treat. | Low risk of bias | 80% of those randomised had data. Similar levels of loss to follow‐up in intervention and placebo group. | Low risk of bias | Outcome measurement performed by a pediatrician unaware of allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Low risk of bias | Low risk of bias in all domains. |
Risk of bias for analysis 4.19 O2: BMI Z scores at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally and masked treatment packs were provided. Analysis was by intention‐to‐treat. | Low risk of bias | 80% of those randomised had data. Similar levels of loss to follow‐up in intervention and placebo group. | Low risk of bias | Outcome measurement performed by a pediatrician unaware of allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 4.20 O4: Body composition: total body fat‐free mass at mid‐ to later childhood follow‐up (kg) .
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally and masked treatment packs were provided. Analysis was by intention‐to‐treat. | Some concerns | 185/212 (87%) children living in centres with DEXA available were assessed. Most that were missing were unable to be traced or declined follow‐up or were at a centre without DEXA availability. 8 were excluded because of severe disability. | Low risk of bias | Measured using whole body dual energy x ray absorptiometry. Outcome assessors were blinded to allocation. | Some concerns | Data analysis plan included in methods. Other follow‐up studies of the trial had prespecified statistical analysis plans. | Some concerns | 185/212 (87%) children living in centres with DEXA available were assessed but this was already a subgroup of the overall study. |
Risk of bias for analysis 4.21 O5: Body composition: total body fat mass at mid‐ to later childhood follow‐up (kg).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 185/212 (87%) children living in centres with DEXA available were assessed. Most that were missing were unable to be traced or declined follow‐up or were at a centre without DEXA availability. 8 were excluded because of severe disability. | Low risk of bias | Measured using whole body dual energy x ray absorptiometry. Outcome assessors were blinded to allocation. | Low risk of bias | Data analysis plan included in methods. Other follow‐up studies of the trial had prespecified statistical analysis plans. | Some concerns | 185/212 (87%) children living in centres with DEXA available were assessed but this was already a subgroup of the overall study |
Risk of bias for analysis 4.22 O6: Mean systolic blood pressure at mid‐ to later childhood follow‐up (mmHg).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.22.1 In all children | ||||||||||||
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 74% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Methods of measurement not stated but performed by trained staff unaware of allocation and likely used standard methods. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data for 26% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.23 O6: Mean diastolic blood pressure at mid‐ to later childhood follow‐up (mmHg).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.23.1 In all children | ||||||||||||
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Some concerns | Data was available for 74% of those randomised. Sensitivity analysis was not performed. Similar levels of missing data in both groups. | Low risk of bias | Methods of measurement not stated but were performed by trained staff unaware of allocation and likely used standard methods. | Low risk of bias | SAP and protocol were not available for review. Outcome was specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Some concerns | Some concerns due to missing data for 26% of those randomised. Similar proportion of missing data in both groups. |
Risk of bias for analysis 4.24 O6: Mean systolic blood pressure Z score at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.24.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 74% of those randomised had data for this outcome. No sensitivity analysis performed. Similar proportions of missing data in both groups. | Low risk of bias | Outcome measurement performed by a pediatrician unaware of allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Some concerns | 74% of those randomised had data for this outcome but there were similar proportions of missing data in both groups. |
Risk of bias for analysis 4.25 O6: Mean diastolic blood pressure Z score at mid‐ to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 4.25.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Some concerns | 74% of those randomised had data for this outcome. No sensitivity analysis performed. Similar proportions of missing data in both groups. | Low risk of bias | Outcome measurement performed by a pediatrician unaware of allocation. | Low risk of bias | Prospective statistical analysis plan included in protocol. | Some concerns | 74% of those randomised had data for this outcome but there were similar proportions of missing data in both groups. |
Risk of bias for analysis 4.26 O8: Measures of lung function at mid‐ to later childhood follow‐up: mean FEV1 Z score.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | High risk of bias | Less than 20% of those alive at follow‐up had available lung function data, primarily as a large number did not have lung function tests of adequate quality. Proportion of missing data were similar between groups. | Low risk of bias | Outcome measurement performed by a pediatrician unaware of allocation. Spirogram was assessed for quality by independent examiners. | Low risk of bias | Prospective data analysis plan included in ethics approval. | High risk of bias | High risk due to a large proportion of missing data. It is possible that those children with poorer lung function were less likely to complete a satisfactory test. |
Risk of bias for analysis 4.27 O8: Measures of lung function at mid‐ to later childhood follow‐up: mean FVC Z score.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | High risk of bias | Less than 20% of those alive at follow‐up had available lung function data, primarily as a large number did not have lung function tests of adequate quality. Proportion of missing data were similar between groups. | Low risk of bias | Outcome measurement performed by a pediatrician unaware of allocation. Spirogram was assessed for quality by independent examiners. | Low risk of bias | Prospective data analysis plan included in ethics approval. | High risk of bias | High risk due to a large proportion of missing data. It is possible that those children with poorer lung function were less likely to complete a satisfactory test. |
Risk of bias for analysis 4.28 O8: Measures of lung function at mid‐ to later childhood follow‐up: mean FEV1/FVC Z score.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | High risk of bias | Less than 20% of those alive at follow‐up had available lung function data, primarily as a large number did not have lung function tests of adequate quality. Proportion of missing data were similar between groups. | Low risk of bias | Outcome measurement performed by a pediatrician unaware of allocation. Spirogram was assessed for quality by independent examiners. | Low risk of bias | Prospective data analysis plan included in ethics approval. | High risk of bias | High risk due to a large proportion of missing data. It is possible that those children with poorer lung function were less likely to complete a satisfactory test. |
Risk of bias for analysis 6.1 K1: Hospital re‐admission by early childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 6.1.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | 98% of those alive at follow‐up had an assessment of this outcome. | Low risk of bias | Outcome assessed by a parental questionnaire. Parents/caregivers were blinded to allocation. | Low risk of bias | Methods state prespecified analysis. The outcome was not specified in the trial registration document. Statistical analysis plan and protocol not available to review but outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| Murphy 2008 | Low risk of bias | A centrally controlled, computerised, telephone randomisation service was used. Randomisation was stratified by centre and gestational age. No marked imbalance of baseline characteristics. | Low risk of bias | The children’s families, clinicians and researchers associated with the trial remained unaware of assignment up to the 5 year follow‐up. Intention‐to‐treat analysis was used. | Low risk of bias | Over 90% follow‐up at 2 years . | Low risk of bias | Obtained from clinical records/participant history. Researchers associated with the trial remained unaware of allocation up to 5 year follow‐up. | Low risk of bias | SAP and protocol were not available for review. Outcome was not specified in trial registration document. Those analyses that were not prespecified were noted in the methods, making selection of this outcome definition and analysis based on results unlikely. | Low risk of bias | Low risk of bias for all domains |
| Peltoniemi 2007 | Low risk of bias | Randomisation performed centrally using opaque, sealed envelopes opened after informed consent was obtained. Nurses, doctors and participants were blinded to allocation. Method of random sequence generation was not stated. Stratified by centre for gestational age and multiple gestations. No marked imbalance of baseline characteristics. | Low risk of bias | Clinical staff, participants and study investigators were blinded to allocation. Study intervention and placebo were prepared in identical, masked syringes. Examiners and families were unaware of allocation at childhood follow‐up. Intention‐to‐treat analysis. | Some concerns | Loss to follow‐up was 19% in the intervention group and 14% in the placebo group. No sensitivity analysis performed. The proportion of missing data was different between the groups but documented reasons for loss to follow‐up were parental refusal or moving to an unknown address. | Low risk of bias | Outcome assessed by parental questionnaire. The outcome of any hospital readmission within 2 years is likely reliable. Study investigators and parents were blinded to allocation. | Low risk of bias | SAP and protocol were not available for review but analysis plans were included on the registered trial protocol. Outcome was in the trial registration. Outcome definition and analysis unlikely to have been selected based on results. | Some concerns | Overall some concerns due to some concerns of risk of bias due to missing outcome data for 23% of those alive for follow‐up, but similar levels of missing data between groups. |
| Wapner 2006 | Low risk of bias | Numbered kits were prepared using randomisation sequences created by an independent data co‐ordinating centre. Sequences were generated using the urn design. Woman were assigned to the next sequentially‐numbered kit. Randomisation was stratified by clinical centre, type of qualifying course and status as inpatient or outpatient. No marked imbalance of baseline characteristics. | Low risk of bias | Betamethasone or identical appearing placebo was prepared by a centralised research pharmacy. Intention‐to‐treat‐analysis. | Low risk of bias | 84% of those alive at 2 year follow‐up had assessmentsc(82% of those randomised). 248 /291 in intervention group and 238/288 in placebo. No sensitivity analysis performed. | Low risk of bias | The mother or healthcare provider were the source of information for this outcome but both were blinded to allocation. Although parental memory of healthcare events is likely unreliable, this only required memory of readmission or not. | Low risk of bias | Protocol with planned statistical analysis was available for review. Outcome was in protocol documentation. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 6.2 K1: Hospital re‐admission by mid to later childhood follow‐up.
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 6.2.1 In all children | ||||||||||||
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | >85% of those randomised had data for this outcome. | High risk of bias | Although parents were blinded to allocation, parental recall of hospital admissions may be unreliable. | Low risk of bias | Prospective statistical analysis plan included in protocol. | High risk of bias | High risk of bias in measurement of the outcome as, although parents were blinded to allocation, parental recall of hospital admissions earlier in life may be unreliable as number of readmissions total was lower than when assessed at 2 years. |
Risk of bias for analysis 6.3 Q2: Length of postnatal hospitalisation for the woman (days).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Subgroup 6.3.1 For all women | ||||||||||||
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. It is unlikely that this result could be impacted by assessor knowledge of treatment allocation (given the treating team were blinded). | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
Risk of bias for analysis 6.4 Q4: Length of infant hospitalisation (days).
| Study | Bias | |||||||||||
| Randomisation process | Deviations from intended interventions | Missing outcome data | Measurement of the outcome | Selection of the reported results | Overall | |||||||
| Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | Authors' judgement | Support for judgement | |
| Crowther 2006 | Low risk of bias | Random‐number sequence generated by computer with variable block sizes and stratification by centre, gestational age and number of fetuses. Allocation was performed centrally. No marked imbalance of baseline characteristics. | Low risk of bias | Treatment packs appeared identical with opaque syringes. Women and perinatal staff were blinded to allocation. Analysis was by intention‐to‐treat. | Low risk of bias | No missing data for this outcome. | Low risk of bias | Outcome ascertainment from case notes. Outcome assessors were blinded to treatment allocation. | Low risk of bias | Methods state prespecified analysis but statistical analysis plan not available to review. | Low risk of bias | Low risk of bias for all domains. |
| Guinn 2001 | Low risk of bias | Computer‐generated randomisation logs prepared centrally and distributed to the research pharmacist at each clinical site for preparation of the allocated treatment. Stratification by centre. No marked imbalance of baseline characteristics. | Low risk of bias | The placebo syringes were indistinguishable from the syringes containing betamethasone. Patients and healthcare workers were blinded to study group. Analysis was by intention‐to‐treat. | Low risk of bias | >95% had data available for this outcome. | Low risk of bias | Data on outcomes collected prospectively from clinical records. It was not stated if assessors were blinded to allocation. Outcome unable to be affected by knowledge of allocation. | Low risk of bias | SAP and protocol were not available to review. No registration document identified. Precoded forms were used to enter predefined outcomes. Outcome definition and analysis unlikely to have been selected based on results. | Low risk of bias | Low risk of bias for all domains. |
| McEvoy 2010 | Low risk of bias | Group assignment performed using a randomisation table. A staff pharmacist performed randomisation and study drug preparation at each institution. Stratification was performed for ≤ 28 versus > 28 weeks' gestation and for multiple gestation (twins versus singletons). No marked imbalance of baseline characteristics. | Low risk of bias | All patients, investigators and care providers were unaware of the treatment allocation. Intention‐to‐treat‐analysis. | Low risk of bias | Neonatal data missing for 1 infant | Low risk of bias | Investigators were unaware of treatment allocation. | Low risk of bias | SAP and protocol were not available for review. Outcome was not in trial registration documentation but the outcome definition and analysis was unlikely to have been selected based on results. | Low risk of bias | Low risk of bias in all domains. |
Acknowledgements
We acknowledge the support from the Cochrane Pregnancy and Childbirth Review editorial team in Liverpool, the Australia and New Zealand Satellite of Cochrane Pregnancy and Childbirth and the Liggins Institute, The University of Auckland, New Zealand.
As part of the prepublication editorial process, this review has been commented on by four peers (an editor and three referees who are external to the editorial team), a member of Cochrane Pregnancy and Childbirth's international panel of consumers and the Group's Statistical Adviser. The authors are grateful to the following peer reviewers for their time and comments: Liz Bickerdike, Associate Editor, Cochrane Editorial & Methods Department, UK; Doris Chou, Department of Sexual and Reproductive Health, Maternal Perinatal Health Team, World Health Organization (WHO), Geneva, Switzerland; Jim Thornton, Emeritus Professor, University of Nottingham, Nottingham, UK.
Unpublished trial data was provided for TEAMS 1999 by Professor Peter Brocklehurst.
This project was supported by the National Institute for Health Research (NIHR), via Cochrane Infrastructure funding to Cochrane Pregnancy and Childbirth. The views and opinions expressed herein are those of the review authors and do not necessarily reflect those of the Evidence Synthesis Programme, the NIHR, National Health Service (NHS) or the Department of Health and Social Care.
This review is supported by funding from the WHO and the UNDP‐UNFPA‐UNICEF‐WHO‐World Bank Special Programme of Research, Development and Research Training in Human Reproduction (HRP) to Cochrane Pregnancy and Childbirth (University of Liverpool). HRP supports and co‐ordinates research on a global scale, synthesises research through systematic reviews of literature, builds research capacity in low‐income countries and develops dissemination tools to make efficient use of ever‐increasing research information. In addition to its co‐sponsors, the International Planned Parenthood Federation (IPPF) and UNAIDS are both members of HRP's governing body.
We thank Anne Lawson for copyediting this review.
Appendices
Appendix 1. Search methods for ICTRP and ClinicalTrials.gov
ICTRP
(Each line was run separately and run 'with all synonyms')
corticosteroids AND premature
corticosteroids AND preterm
steroids AND premature
steroids AND preterm
ClinicalTrials.gov
Advanced search
Interventional Studies | Preterm Labor | Corticosteroid
Interventional Studies | Preterm Labor | Steroids
Data and analyses
Comparison 1. Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 A1: Fetal or neonatal or infant death (< 1 year of age) | 10 | 5849 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.73, 1.24] |
| 1.1.1 All fetuses randomised | 10 | 5849 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.73, 1.24] |
| 1.2 A2: Fetal death | 7 | 2758 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.24, 2.84] |
| 1.2.1 All fetuses randomised | 7 | 2758 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.24, 2.84] |
| 1.3 A3: Neonatal death | 7 | 2758 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.62, 1.34] |
| 1.3.1 In all neonates | 7 | 2758 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.62, 1.34] |
| 1.4 A5: Respiratory distress syndrome | 9 | 3540 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.74, 0.90] |
| 1.4.1 All fetuses randomised | 9 | 3540 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.74, 0.90] |
| 1.5 A6: Severe respiratory distress syndrome | 5 | 3809 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.82, 1.16] |
| 1.5.1 All fetuses randomised | 5 | 3809 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.82, 1.16] |
| 1.6 A7: Severe lung disease | 6 | 4955 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.72, 0.97] |
| 1.6.1 All fetuses randomised | 6 | 4955 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.72, 0.97] |
| 1.7 A8: Chronic lung disease | 9 | 5661 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.83, 1.22] |
| 1.7.1 All fetuses randomised | 9 | 5661 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.83, 1.22] |
| 1.8 A9: Severe intraventricular haemorrhage (grade 3/4) | 7 | 5066 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.69, 1.86] |
| 1.8.1 All fetuses randomised | 7 | 5066 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.69, 1.86] |
| 1.9 A10: Intraventricular haemorrhage | 6 | 3223 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.75, 1.19] |
| 1.9.1 All fetuses randomised | 6 | 3223 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.75, 1.19] |
| 1.10 A11: Necrotising enterocolitis | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.59, 1.22] |
| 1.10.1 All fetuses randomised | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.59, 1.22] |
| 1.11 A12: Composite of serious outcomes | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.80, 0.97] |
| 1.11.1 All fetuses randomised | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.80, 0.97] |
| 1.12 F1i: Term birth ≥ 37 weeks | 7 | 4068 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.86, 1.06] |
| 1.12.1 All fetuses randomised | 7 | 4068 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.86, 1.06] |
| 1.13 F1ii: Preterm birth before 37 weeks | 7 | 4068 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.98, 1.05] |
| 1.13.1 All fetuses randomised | 7 | 4068 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.98, 1.05] |
| 1.14 F1iii: Very preterm birth before 34 weeks | 6 | 2682 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.97, 1.08] |
| 1.14.1 All fetuses randomised | 6 | 2682 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.97, 1.08] |
| 1.15 F1iv: Extremely preterm birth before 28 weeks | 5 | 4022 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.92, 1.38] |
| 1.15.1 All fetuses randomised | 5 | 4022 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.92, 1.38] |
| 1.16 F1v: Mean gestational age at birth (weeks) | 10 | 5235 | Mean Difference (IV, Fixed, 95% CI) | ‐0.18 [‐0.37, 0.01] |
| 1.16.1 In all neonates | 10 | 5235 | Mean Difference (IV, Fixed, 95% CI) | ‐0.18 [‐0.37, 0.01] |
| 1.17 F2: Small‐for‐gestational age at birth | 7 | 4013 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.25 [1.08, 1.44] |
| 1.17.1 All fetuses randomised | 7 | 4013 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.25 [1.08, 1.44] |
| 1.18 F3: Admission to the neonatal intensive care unit | 2 | 3455 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.95, 1.07] |
| 1.18.1 All fetuses randomised | 2 | 3455 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.95, 1.07] |
| 1.19 F4: Proven neonatal infection while in the neonatal intensive care unit | 8 | 5660 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.86, 1.22] |
| 1.19.1 All fetuses randomised | 8 | 5660 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.86, 1.22] |
| 1.20 F5: Early systemic neonatal infection | 4 | 1738 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.79, 1.11] |
| 1.20.1 All fetuses randomised | 4 | 1738 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.79, 1.11] |
| 1.21 F6: Late systemic neonatal infection | 2 | 404 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.37 [0.78, 2.41] |
| 1.21.1 All fetuses randomised | 2 | 404 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.37 [0.78, 2.41] |
| 1.22 F7: Retinopathy of prematurity | 8 | 5234 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.81, 1.27] |
| 1.22.1 All fetuses randomised | 8 | 5234 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.81, 1.27] |
| 1.23 F8: Periventricular leukomalacia | 8 | 5142 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.75 [0.43, 1.31] |
| 1.23.1 All fetuses randomised | 8 | 5142 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.75 [0.43, 1.31] |
| 1.24 F9: Neonatal encephalopathy | 1 | 76 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.50 [0.10, 2.57] |
| 1.24.1 All fetuses randomised | 1 | 76 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.50 [0.10, 2.57] |
| 1.25 F10: Patent ductus arteriosus | 7 | 4657 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.78 [0.63, 0.96] |
| 1.25.1 All fetuses randomised | 7 | 4657 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.78 [0.63, 0.96] |
| 1.26 F11: Use of respiratory support | 2 | 2497 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.80, 0.97] |
| 1.26.1 All fetuses randomised | 2 | 2497 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.80, 0.97] |
| 1.27 F12: Use of invasive respiratory support | 6 | 5067 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.86 [0.80, 0.93] |
| 1.27.1 All fetuses randomised | 6 | 5067 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.86 [0.80, 0.93] |
| 1.28 F13: Use of non‐invasive respiratory support | 3 | 3231 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.82, 1.01] |
| 1.28.1 All fetuses randomised | 3 | 3231 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.82, 1.01] |
| 1.29 F14: Use of oxygen supplementation | 3 | 3643 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.85, 0.98] |
| 1.29.1 All fetuses randomised | 3 | 3643 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.85, 0.98] |
| 1.30 F15: Use of surfactant | 10 | 5870 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.73, 0.89] |
| 1.30.1 All fetuses randomised | 10 | 5870 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.73, 0.89] |
| 1.31 F17: Use of postnatal corticosteroids | 4 | 4145 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.37 [0.99, 1.91] |
| 1.31.1 All fetuses randomised | 4 | 4145 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.37 [0.99, 1.91] |
| 1.32 F16: Use of nitric oxide for respiratory support | 1 | 1146 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.58 [0.29, 1.17] |
| 1.32.1 All fetuses randomised | 1 | 1146 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.58 [0.29, 1.17] |
| 1.33 F20: Use of inotropic support | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.66, 0.97] |
| 1.33.1 All fetuses randomised | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.66, 0.97] |
| 1.34 F18: Air leak syndrome | 4 | 2505 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.50, 1.36] |
| 1.34.1 All fetuses randomised | 4 | 2505 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.50, 1.36] |
| 1.35 F21: Apgar score < 7 at 5 minutes | 3 | 4032 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.64, 1.09] |
| 1.35.1 All fetuses randomised | 3 | 4032 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.64, 1.09] |
| 1.36 F22: Neonatal cardiac hypertrophy as measured by interventricular septal thickness (IVS in mm) | 1 | 175 | Mean Difference (IV, Fixed, 95% CI) | 0.01 [‐0.24, 0.26] |
| 1.36.1 In all neonates | 1 | 175 | Mean Difference (IV, Fixed, 95% CI) | 0.01 [‐0.24, 0.26] |
| 1.37 F22: Neonatal cardiac hypertrophy as measured by left ventricular wall thickness in diastole (mm) | 1 | 175 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.27, 0.19] |
| 1.37.1 In all neonates | 1 | 175 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.27, 0.19] |
| 1.38 L1: Mean birthweight (g) | 10 | 5808 | Mean Difference (IV, Fixed, 95% CI) | ‐74.49 [‐115.80, ‐33.18] |
| 1.38.1 In all neonates | 10 | 5808 | Mean Difference (IV, Fixed, 95% CI) | ‐74.49 [‐115.80, ‐33.18] |
| 1.39 L1: Mean birthweight Z score | 3 | 1438 | Mean Difference (IV, Fixed, 95% CI) | ‐0.13 [‐0.25, ‐0.02] |
| 1.39.1 In all neonates | 3 | 1438 | Mean Difference (IV, Fixed, 95% CI) | ‐0.13 [‐0.25, ‐0.02] |
| 1.40 L1: Birthweight multiples of the median | 1 | 590 | Mean Difference (IV, Fixed, 95% CI) | 0.00 [‐0.03, 0.03] |
| 1.40.1 In all babies | 1 | 590 | Mean Difference (IV, Fixed, 95% CI) | 0.00 [‐0.03, 0.03] |
| 1.41 L1: Birthweight adjusted for gestation (standardised mean difference) | 4 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.41.1 In all neonates | 4 | 2028 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.15 [‐0.23, ‐0.06] |
| 1.42 L2: Interval between trial entry and birth (days) | 4 | 1309 | Mean Difference (IV, Fixed, 95% CI) | ‐0.71 [‐1.36, ‐0.06] |
| 1.43 L3: Mean head circumference at birth (cm) | 10 | 5731 | Mean Difference (IV, Fixed, 95% CI) | ‐0.32 [‐0.49, ‐0.15] |
| 1.43.1 In all neonates | 10 | 5731 | Mean Difference (IV, Fixed, 95% CI) | ‐0.32 [‐0.49, ‐0.15] |
| 1.44 L3: Mean head circumference Z score at birth | 2 | 1251 | Mean Difference (IV, Fixed, 95% CI) | ‐0.14 [‐0.27, ‐0.00] |
| 1.44.1 In all babies | 2 | 1251 | Mean Difference (IV, Fixed, 95% CI) | ‐0.14 [‐0.27, ‐0.00] |
| 1.45 L4: Mean length at birth (cm) | 6 | 4550 | Mean Difference (IV, Fixed, 95% CI) | ‐0.56 [‐0.89, ‐0.23] |
| 1.45.1 In all neonates | 6 | 4550 | Mean Difference (IV, Fixed, 95% CI) | ‐0.56 [‐0.89, ‐0.23] |
| 1.46 L4: Mean length Z score at birth | 2 | 1256 | Mean Difference (IV, Fixed, 95% CI) | ‐0.05 [‐0.19, 0.09] |
| 1.46.1 In all neonates | 2 | 1256 | Mean Difference (IV, Fixed, 95% CI) | ‐0.05 [‐0.19, 0.09] |
| 1.47 L4: Length multiples of the mean at birth | 1 | 590 | Mean Difference (IV, Fixed, 95% CI) | ‐0.01 [‐0.02, ‐0.00] |
| 1.47.1 In all neonates | 1 | 590 | Mean Difference (IV, Fixed, 95% CI) | ‐0.01 [‐0.02, ‐0.00] |
| 1.48 L4: Length at birth adjusted for gestation (standardised mean difference) | 3 | 1846 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.09 [‐0.18, 0.01] |
| 1.48.1 In all neonates | 3 | 1846 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.09 [‐0.18, 0.01] |
| 1.49 L5i: Mean weight (g) at primary hospital discharge | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.49.1 In all infants | 2 | 1195 | Mean Difference (IV, Fixed, 95% CI) | 17.91 [‐52.35, 88.18] |
| 1.50 L5i: Mean weight Z score at primary hospital discharge | 2 | 1195 | Mean Difference (IV, Fixed, 95% CI) | ‐0.05 [‐0.16, 0.06] |
| 1.50.1 In all infants | 2 | 1195 | Mean Difference (IV, Fixed, 95% CI) | ‐0.05 [‐0.16, 0.06] |
| 1.51 L5ii: Mean head circumference (cm) at primary hospital discharge | 2 | 1195 | Mean Difference (IV, Fixed, 95% CI) | 0.12 [‐0.10, 0.35] |
| 1.51.1 In all infants | 2 | 1195 | Mean Difference (IV, Fixed, 95% CI) | 0.12 [‐0.10, 0.35] |
| 1.52 L5ii: Mean head circumference Z score at primary hospital discharge | 2 | 1195 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐0.15, 0.10] |
| 1.52.1 In all infants | 2 | 1195 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐0.15, 0.10] |
| 1.53 L5iii: Mean length (cm) at primary hospital discharge | 2 | 1189 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.44, 0.47] |
| 1.53.1 In all infants | 2 | 1189 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.44, 0.47] |
| 1.54 L5iii: Mean length Z score at primary hospital discharge | 2 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 1.54.1 In all infants | 2 | 1189 | Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.23, 0.10] |
| 1.55 L6i: Mean weight at infant follow‐up (kg) | 2 | 149 | Mean Difference (IV, Fixed, 95% CI) | ‐0.60 [‐0.87, ‐0.34] |
| 1.56 L6i: Mean weight Z score at infant follow‐up | 1 | 75 | Mean Difference (IV, Fixed, 95% CI) | ‐0.12 [‐0.84, 0.60] |
| 1.57 L6ii: Mean head circumference at infant follow‐up (cm) | 2 | 136 | Mean Difference (IV, Fixed, 95% CI) | ‐0.45 [‐0.85, ‐0.05] |
| 1.58 L6ii: Mean head circumference Z score at infant follow‐up | 1 | 62 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.78, 0.70] |
| 1.59 L6iii: Mean length at infant follow‐up (cm) | 2 | 149 | Mean Difference (IV, Fixed, 95% CI) | ‐1.57 [‐2.32, ‐0.82] |
| 1.60 L6iii: Mean length Z score at infant follow‐up | 1 | 75 | Mean Difference (IV, Fixed, 95% CI) | ‐0.24 [‐1.21, 0.73] |
| 1.61 L8: Mean duration of invasive respiratory support (days) | 4 | 1620 | Mean Difference (IV, Fixed, 95% CI) | ‐0.27 [‐0.38, ‐0.16] |
| 1.62 L9: Mean duration of non‐invasive respiratory support (days) | 1 | 326 | Mean Difference (IV, Fixed, 95% CI) | ‐0.59 [‐1.29, 0.11] |
| 1.63 L10: Mean duration of oxygen supplementation (days) | 4 | 1619 | Mean Difference (IV, Fixed, 95% CI) | ‐0.32 [‐0.94, 0.30] |
| 1.64 L14: Mean cord cortisol concentrations at birth (nmol/L) | 2 | 442 | Mean Difference (IV, Fixed, 95% CI) | ‐50.49 [‐73.14, ‐27.85] |
| 1.64.1 In all neonates | 2 | 442 | Mean Difference (IV, Fixed, 95% CI) | ‐50.49 [‐73.14, ‐27.85] |
1.39. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 39: L1: Mean birthweight Z score
1.40. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 40: L1: Birthweight multiples of the median
1.46. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 46: L4: Mean length Z score at birth
1.47. Analysis.

Comparison 1: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the fetus/neonate/infant, Outcome 47: L4: Length multiples of the mean at birth
Comparison 2. Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 2.1 B1: Maternal death | 1 | 437 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.32 [0.01, 7.81] |
| 2.1.1 All women | 1 | 437 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.32 [0.01, 7.81] |
| 2.2 B2: Maternal sepsis | 8 | 4666 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.93, 1.39] |
| 2.2.1 In all women | 8 | 4666 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.93, 1.39] |
| 2.3 B3: Caesarean section | 8 | 4266 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.98, 1.09] |
| 2.3.1 In all women | 8 | 4266 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.98, 1.09] |
| 2.4 B4: Discontinuation of therapy due to maternal adverse effects | 1 | 502 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 2.4.1 In all women | 1 | 502 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 2.5 G1: Puerperal sepsis | 6 | 3246 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.05 [0.78, 1.43] |
| 2.5.1 In all women | 6 | 3246 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.05 [0.78, 1.43] |
| 2.6 G2: Chorioamnionitis during labour | 7 | 4417 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.90, 1.42] |
| 2.6.1 In all women | 7 | 4417 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.90, 1.42] |
| 2.7 G3: Endometritis | 4 | 2842 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.17 [0.74, 1.84] |
| 2.7.1 In all women | 4 | 2842 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.17 [0.74, 1.84] |
| 2.8 G4: Pyrexia after trial entry requiring the use of antibiotics | 1 | 156 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.25, 1.25] |
| 2.8.1 In all women | 1 | 156 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.25, 1.25] |
| 2.9 G6: Postpartum haemorrhage | 2 | 641 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.54 [0.31, 0.96] |
| 2.9.1 In all women | 2 | 641 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.54 [0.31, 0.96] |
| 2.10 G7: Postnatal pyrexia | 1 | 982 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.55, 1.38] |
| 2.10.1 In all women | 1 | 982 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.55, 1.38] |
| 2.11 G8: Prelabour rupture of membranes after trial entry | 2 | 977 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.65, 1.29] |
| 2.11.1 In all women | 2 | 977 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.65, 1.29] |
| 2.12 G9: Mode of birth: vaginal birth | 6 | 4025 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.94 [0.87, 1.00] |
| 2.12.1 In all women | 6 | 4025 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.94 [0.87, 1.00] |
| 2.13 G10: Hypertension | 3 | 3327 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.08 [0.87, 1.32] |
| 2.13.1 In all women | 3 | 3327 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.08 [0.87, 1.32] |
| 2.14 G12: Glucose intolerance | 2 | 2345 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.23 [0.90, 1.67] |
| 2.14.1 In all women | 2 | 2345 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.23 [0.90, 1.67] |
| 2.15 G13: Postnatal depression | 1 | 1671 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.70, 1.10] |
| 2.16 G14: Local injection site adverse effects | 2 | 1477 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.44 [0.32, 0.60] |
| 2.17 G15: Insomnia after treatment | 4 | 3198 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.21 [1.04, 1.40] |
| 2.17.1 In all women | 4 | 3198 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.21 [1.04, 1.40] |
| 2.18 G16: Gastrointestinal adverse effects of treatment | 1 | 495 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.34 [0.14, 0.85] |
2.4. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 4: B4: Discontinuation of therapy due to maternal adverse effects
2.14. Analysis.

Comparison 2: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the woman, Outcome 14: G12: Glucose intolerance
Comparison 3. Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 3.1 C1: Total deaths (after randomisation) up to early childhood follow‐up | 5 | 4565 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.06 [0.81, 1.40] |
| 3.1.1 In all children | 5 | 4565 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.06 [0.81, 1.40] |
| 3.2 C2: Neurodevelopmental impairment at early childhood follow‐up | 4 | 3616 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.85, 1.10] |
| 3.2.1 In all children | 4 | 3616 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.85, 1.10] |
| 3.3 C3: Survival free of neurodevelopmental impairment at early childhood follow‐up | 4 | 3845 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.98, 1.04] |
| 3.3.1 In all children | 4 | 3845 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.98, 1.04] |
| 3.4 C4: Survival free of major neurodevelopmental impairment at early childhood follow‐up | 3 | 1816 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.98, 1.05] |
| 3.4.1 In all children | 3 | 1816 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.98, 1.05] |
| 3.5 C5: Cerebral palsy at early childhood follow‐up | 5 | 3923 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.71, 1.49] |
| 3.5.1 In all children | 5 | 3923 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.71, 1.49] |
| 3.6 C6: Developmental delay or intellectual impairment at early childhood follow‐up (any) | 4 | 3581 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.84, 1.09] |
| 3.6.1 In all children | 4 | 3581 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.84, 1.09] |
| 3.7 Mental Developmental Index at early childhood follow‐up | 3 | 1627 | Mean Difference (IV, Fixed, 95% CI) | 0.89 [‐0.61, 2.39] |
| 3.7.1 In all children | 3 | 1627 | Mean Difference (IV, Fixed, 95% CI) | 0.89 [‐0.61, 2.39] |
| 3.8 H1: Child behaviour at early childhood follow‐up, Child Behaviour Checklist total score in the clinical range | 1 | 1045 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.09 [0.79, 1.51] |
| 3.9 H1: Child behaviour: Behaviour rating scale in the clinical range (BSID‐II) | 1 | 1776 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.16 [1.00, 1.34] |
| 3.10 H1: Child behaviour at early childhood follow‐up as assessed by Early Child Behaviour Questionnaire Extraversion summary scale | 1 | 142 | Mean Difference (IV, Fixed, 95% CI) | 0.00 [‐0.18, 0.18] |
| 3.11 H1: Child behaviour at early childhood follow‐up assessed by Early Child Behaviour questionnaire Negative affectivity summary scale | 1 | 142 | Mean Difference (IV, Fixed, 95% CI) | ‐0.10 [‐0.27, 0.07] |
| 3.12 H1: Child behaviour at early childhood follow‐up assessed by Early Child Behaviour questionnaire Effortful control summary scale | 1 | 142 | Mean Difference (IV, Fixed, 95% CI) | 0.00 [‐0.17, 0.17] |
| 3.13 H2: Psychomotor Developmental Index at early childhood follow‐up | 2 | 1423 | Mean Difference (IV, Fixed, 95% CI) | 1.26 [‐0.45, 2.96] |
| 3.13.1 In all babies | 2 | 1423 | Mean Difference (IV, Fixed, 95% CI) | 1.26 [‐0.45, 2.96] |
| 3.14 H3: Deafness/hearing impairment at early childhood follow‐up | 4 | 3528 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.56, 1.71] |
| 3.14.1 In all children | 4 | 3528 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.56, 1.71] |
| 3.15 H4: Blindness/visual impairment at early childhood follow‐up | 3 | 3274 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.17 [0.65, 2.10] |
| 3.15.1 In all children | 3 | 3274 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.17 [0.65, 2.10] |
| 3.16 H5: Hypertension at early childhood follow‐up | 1 | 628 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.77, 1.23] |
| 3.16.1 In all children | 1 | 628 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.77, 1.23] |
| 3.17 H8: Asthma or recurrent wheeze at early childhood follow‐up | 3 | 1720 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.74, 1.06] |
| 3.17.1 In all children | 3 | 1720 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.74, 1.06] |
| 3.18 H9: Any respiratory disease at early childhood follow‐up | 3 | 3423 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.92, 1.18] |
| 3.19 N1i: Mean weight at early childhood follow‐up (kg) | 4 | 3784 | Mean Difference (IV, Fixed, 95% CI) | ‐0.16 [‐0.25, ‐0.07] |
| 3.19.1 In all children | 4 | 3784 | Mean Difference (IV, Fixed, 95% CI) | ‐0.16 [‐0.25, ‐0.07] |
| 3.20 N1i: Mean weight Z score at early childhood follow‐up | 1 | 1047 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐0.19, 0.13] |
| 3.20.1 In all children | 1 | 1047 | Mean Difference (IV, Fixed, 95% CI) | ‐0.03 [‐0.19, 0.13] |
| 3.21 N1i: Mean weight adjusted for age at early childhood follow‐up (standardised mean difference) | 3 | 1776 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.15, 0.04] |
| 3.21.1 In all children | 3 | 1776 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.15, 0.04] |
| 3.22 N1i: Weight small for age at early childhood follow‐up | 2 | 1533 | Risk Ratio (IV, Fixed, 95% CI) | 0.92 [0.72, 1.19] |
| 3.22.1 In all children | 2 | 1533 | Risk Ratio (IV, Fixed, 95% CI) | 0.92 [0.72, 1.19] |
| 3.23 N1ii: Mean head circumference at early childhood follow‐up (cm) | 4 | 3784 | Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.18, 0.06] |
| 3.23.1 In all children | 4 | 3784 | Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.18, 0.06] |
| 3.24 N1ii: Mean head circumference Z score at early childhood follow‐up | 2 | 1290 | Mean Difference (IV, Fixed, 95% CI) | 0.04 [‐0.09, 0.18] |
| 3.24.1 In all children | 2 | 1290 | Mean Difference (IV, Fixed, 95% CI) | 0.04 [‐0.09, 0.18] |
| 3.25 N1ii: Mean head circumference adjusted for age at early childhood follow‐up (standardised mean difference) | 3 | Std. Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 3.25.1 In all babies | 3 | 1776 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.07, 0.11] |
| 3.26 N1ii: Head circumference small for age at early childhood follow‐up | 2 | 1527 | Risk Ratio (IV, Fixed, 95% CI) | 1.02 [0.87, 1.21] |
| 3.26.1 In all babies | 2 | 1527 | Risk Ratio (IV, Fixed, 95% CI) | 1.02 [0.87, 1.21] |
| 3.27 N1iii: Mean height at early childhood follow‐up (cm) | 4 | 3784 | Mean Difference (IV, Fixed, 95% CI) | ‐0.07 [‐0.31, 0.17] |
| 3.27.1 In all children | 4 | 3784 | Mean Difference (IV, Fixed, 95% CI) | ‐0.07 [‐0.31, 0.17] |
| 3.28 N1iii: Height Z score at early childhood follow‐up | 2 | 1290 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.17, 0.09] |
| 3.28.1 In all children | 2 | 1290 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.17, 0.09] |
| 3.29 N1iii: Mean height adjusted for age at early childhood follow‐up (standardised mean difference) | 3 | 1776 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.15, 0.04] |
| 3.29.1 In all children | 3 | 1776 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.15, 0.04] |
| 3.30 N1iii: Height small for age at early childhood follow‐up | 2 | 1526 | Risk Ratio (IV, Fixed, 95% CI) | 1.03 [0.77, 1.36] |
| 3.30.1 In all children | 2 | 1526 | Risk Ratio (IV, Fixed, 95% CI) | 1.03 [0.77, 1.36] |
| 3.31 N3: Mean systolic blood pressure at early childhood follow‐up (mmHg) | 1 | 486 | Mean Difference (IV, Fixed, 95% CI) | ‐2.90 [‐5.40, ‐0.40] |
| 3.31.1 In all children | 1 | 486 | Mean Difference (IV, Fixed, 95% CI) | ‐2.90 [‐5.40, ‐0.40] |
| 3.32 N3: Mean systolic blood pressure Z score at early childhood follow‐up | 1 | 672 | Mean Difference (IV, Fixed, 95% CI) | ‐0.10 [‐0.28, 0.08] |
| 3.32.1 In all children | 1 | 672 | Mean Difference (IV, Fixed, 95% CI) | ‐0.10 [‐0.28, 0.08] |
| 3.33 N3: Mean diastolic blood pressure at early childhood follow‐up (mmHg) | 1 | 486 | Mean Difference (IV, Fixed, 95% CI) | ‐1.00 [‐2.86, 0.86] |
| 3.33.1 In all children | 1 | 486 | Mean Difference (IV, Fixed, 95% CI) | ‐1.00 [‐2.86, 0.86] |
| 3.34 N3: Mean diastolic blood pressure Z score at early childhood follow‐up | 1 | 628 | Mean Difference (IV, Fixed, 95% CI) | 0.10 [‐0.05, 0.25] |
| 3.34.1 In all children | 1 | 628 | Mean Difference (IV, Fixed, 95% CI) | 0.10 [‐0.05, 0.25] |
3.8. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 8: H1: Child behaviour at early childhood follow‐up, Child Behaviour Checklist total score in the clinical range
3.20. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 20: N1i: Mean weight Z score at early childhood follow‐up
3.24. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 24: N1ii: Mean head circumference Z score at early childhood follow‐up
3.28. Analysis.

Comparison 3: Repeat dose(s) of corticosteroids versus single course: outcomes for the child in early childhood (2 to < 5 years of age), Outcome 28: N1iii: Height Z score at early childhood follow‐up
Comparison 4. Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 4.1 D1: Total deaths (after randomisation) up to mid‐ to later childhood follow‐up | 2 | 2874 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.69, 1.26] |
| 4.1.1 In all children | 2 | 2874 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.69, 1.26] |
| 4.2 D2: Neurocognitive impairment at mid‐ to later childhood follow‐up | 1 | 897 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.72, 1.29] |
| 4.2.1 In all children | 1 | 897 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.72, 1.29] |
| 4.3 D3: Survival free of neurocognitive impairment at mid‐ to later childhood follow‐up | 1 | 963 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.95, 1.08] |
| 4.3.1 In all children | 1 | 963 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.95, 1.08] |
| 4.4 D4: Survival free of major neurocognitive impairment at mid‐ to later childhood follow‐up | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 4.4.1 In all children | 2 | 2682 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.97, 1.04] |
| 4.5 D6: Cognitive impairment at mid‐ to later childhood follow‐up | 2 | 2504 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.81, 1.22] |
| 4.5.1 In all children | 2 | 2504 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.81, 1.22] |
| 4.6 D8: Cerebral palsy at mid‐ to later childhood follow‐up | 2 | 2622 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.74 [0.43, 1.26] |
| 4.6.1 In all children | 2 | 2622 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.74 [0.43, 1.26] |
| 4.7 I2: Blindness/visual impairment at mid‐ to later childhood follow‐up | 2 | 2532 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.12 [0.79, 1.59] |
| 4.7.1 In all children | 2 | 2532 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.12 [0.79, 1.59] |
| 4.8 I3: Deafness/hearing impairment at mid‐ to later childhood follow‐up | 2 | 2532 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.62 [0.77, 3.41] |
| 4.8.1 In all children | 2 | 2532 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.62 [0.77, 3.41] |
| 4.9 I4: Abnormal child behaviour at mid‐ to later childhood follow‐up | 1 | 1615 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.75, 1.22] |
| 4.10 I4: Child behaviour at mid‐ to later childhood follow‐up (standardised mean difference) | 2 | 2480 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.00 [‐0.08, 0.08] |
| 4.11 I5: Asthma or recurrent wheeze at mid‐ to later childhood follow‐up | 1 | 979 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.85, 1.19] |
| 4.11.1 In all babies | 1 | 979 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.85, 1.19] |
| 4.12 I6: Any respiratory disease at mid‐ to later childhood follow‐up | 1 | 979 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.79 [0.36, 1.72] |
| 4.13 O1i: Mean weight at mid‐ to later childhood follow‐up (kg) | 1 | 1635 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.62, 0.22] |
| 4.13.1 In all children | 1 | 1635 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐0.62, 0.22] |
| 4.14 O1i: Mean weight Z score at mid‐ to later childhood follow‐up | 1 | 940 | Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.23, 0.11] |
| 4.14.1 In all children | 1 | 940 | Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.23, 0.11] |
| 4.15 O1ii: Mean head circumference at mid‐ to later childhood follow‐up (cm) | 1 | 1635 | Mean Difference (IV, Fixed, 95% CI) | ‐0.10 [‐0.36, 0.16] |
| 4.15.1 In all children | 1 | 1635 | Mean Difference (IV, Fixed, 95% CI) | ‐0.10 [‐0.36, 0.16] |
| 4.16 O1ii: Head circumference Z score at mid‐ to later childhood follow‐up | 1 | 885 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.22, 0.14] |
| 4.16.1 In all children | 1 | 885 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.22, 0.14] |
| 4.17 O1iii: Mean height at mid‐ to later childhood follow‐up (cm) | 1 | 1635 | Mean Difference (IV, Fixed, 95% CI) | ‐0.40 [‐1.11, 0.31] |
| 4.17.1 In all children | 1 | 1635 | Mean Difference (IV, Fixed, 95% CI) | ‐0.40 [‐1.11, 0.31] |
| 4.18 O1iii: Mean height Z score at mid‐ to later childhood follow‐up | 1 | 912 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.13, 0.17] |
| 4.18.1 In all children | 1 | 912 | Mean Difference (IV, Fixed, 95% CI) | 0.02 [‐0.13, 0.17] |
| 4.19 O2: BMI Z scores at mid‐ to later childhood follow‐up | 1 | 910 | Mean Difference (IV, Fixed, 95% CI) | ‐0.13 [‐0.30, 0.04] |
| 4.20 O4: Body composition: total body fat‐free mass at mid‐ to later childhood follow‐up (kg) | 1 | 185 | Mean Difference (IV, Fixed, 95% CI) | ‐0.20 [‐1.43, 1.03] |
| 4.21 O5: Body composition: total body fat mass at mid‐ to later childhood follow‐up (kg) | 1 | 185 | Mean Difference (IV, Fixed, 95% CI) | 0.10 [‐0.71, 0.91] |
| 4.22 O6: Mean systolic blood pressure at mid‐ to later childhood follow‐up (mmHg) | 1 | 1635 | Mean Difference (IV, Fixed, 95% CI) | 0.30 [‐1.06, 1.66] |
| 4.22.1 In all children | 1 | 1635 | Mean Difference (IV, Fixed, 95% CI) | 0.30 [‐1.06, 1.66] |
| 4.23 O6: Mean diastolic blood pressure at mid‐ to later childhood follow‐up (mmHg) | 1 | 1635 | Mean Difference (IV, Fixed, 95% CI) | 0.70 [‐0.46, 1.86] |
| 4.23.1 In all children | 1 | 1635 | Mean Difference (IV, Fixed, 95% CI) | 0.70 [‐0.46, 1.86] |
| 4.24 O6: Mean systolic blood pressure Z score at mid‐ to later childhood follow‐up | 1 | 848 | Mean Difference (IV, Fixed, 95% CI) | 0.07 [‐0.06, 0.20] |
| 4.24.1 In all children | 1 | 848 | Mean Difference (IV, Fixed, 95% CI) | 0.07 [‐0.06, 0.20] |
| 4.25 O6: Mean diastolic blood pressure Z score at mid‐ to later childhood follow‐up | 1 | 848 | Mean Difference (IV, Fixed, 95% CI) | ‐0.09 [‐0.25, 0.07] |
| 4.25.1 In all children | 1 | 848 | Mean Difference (IV, Fixed, 95% CI) | ‐0.09 [‐0.25, 0.07] |
| 4.26 O8: Measures of lung function at mid‐ to later childhood follow‐up: mean FEV1 Z score | 1 | 185 | Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.34, 0.22] |
| 4.27 O8: Measures of lung function at mid‐ to later childhood follow‐up: mean FVC Z score | 1 | 185 | Mean Difference (IV, Fixed, 95% CI) | ‐0.18 [‐0.49, 0.13] |
| 4.28 O8: Measures of lung function at mid‐ to later childhood follow‐up: mean FEV1/FVC Z score | 1 | 185 | Mean Difference (IV, Fixed, 95% CI) | ‐0.19 [‐0.44, 0.06] |
4.20. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 20: O4: Body composition: total body fat‐free mass at mid‐ to later childhood follow‐up (kg)
4.21. Analysis.

Comparison 4: Repeat dose(s) of prenatal corticosteroids versus single course: outcomes for the child in mid‐ to later childhood (5 to < 18 years of age), Outcome 21: O5: Body composition: total body fat mass at mid‐ to later childhood follow‐up (kg)
Comparison 6. Repeat dose(s) of corticosteroids versus single course: outcomes for health services.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 6.1 K1: Hospital re‐admission by early childhood follow‐up | 4 | 3824 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.93, 1.11] |
| 6.1.1 In all children | 4 | 3824 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.93, 1.11] |
| 6.2 K1: Hospital re‐admission by mid to later childhood follow‐up | 1 | 980 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.10 [0.80, 1.52] |
| 6.2.1 In all children | 1 | 980 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.10 [0.80, 1.52] |
| 6.3 Q2: Length of postnatal hospitalisation for the woman (days) | 1 | 483 | Mean Difference (IV, Fixed, 95% CI) | 0.00 [‐0.22, 0.22] |
| 6.3.1 For all women | 1 | 483 | Mean Difference (IV, Fixed, 95% CI) | 0.00 [‐0.22, 0.22] |
| 6.4 Q4: Length of infant hospitalisation (days) | 3 | 1733 | Mean Difference (IV, Fixed, 95% CI) | 0.18 [‐2.60, 2.96] |
Comparison 7. Subgroup analysis for the number of babies in utero.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 7.1 A12: Composite serious outcome (variously defined) | 9 | 8373 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.85, 1.00] |
| 7.1.1 A12: Composite serious outcome (variously defined); intention to treat analysis | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.80, 0.97] |
| 7.1.2 In babies from singleton pregnancies | 2 | 1646 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.06 [0.86, 1.30] |
| 7.1.3 In babies from multiple pregnancies | 2 | 991 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.80, 1.31] |
| 7.2 C3: Survival free of neurodevelopmental impairment at early childhood follow‐up | 1 | 2104 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.96, 1.03] |
| 7.2.1 In babies from singleton pregnancies | 1 | 1333 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.96, 1.05] |
| 7.2.2 In babies from multiple pregnancies | 1 | 771 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.94, 1.05] |
Comparison 8. Subgroup analysis for the planned number of repeat courses of corticosteroids to be given.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 8.1 A1: Fetal or neonatal or infant death (< 1 year of age) | 10 | 5849 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.73, 1.24] |
| 8.1.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 7 | 4831 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.68, 1.19] |
| 8.1.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 3 | 1018 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.38 [0.65, 2.92] |
| 8.2 A2: Fetal death | 7 | 2758 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.24, 2.84] |
| 8.2.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 4 | 1740 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.14, 3.57] |
| 8.2.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 3 | 1018 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.14, 7.23] |
| 8.3 A3: Neonatal death | 7 | 2758 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.61, 1.31] |
| 8.3.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 4 | 1740 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.51, 1.23] |
| 8.3.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 3 | 1018 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.36 [0.58, 3.19] |
| 8.4 A5: Respiratory distress syndrome | 9 | 3540 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.74, 0.90] |
| 8.4.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 6 | 2522 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.71, 0.91] |
| 8.4.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 3 | 1018 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.72, 0.98] |
| 8.5 A6: Severe respiratory distress syndrome | 5 | 3809 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.82, 1.16] |
| 8.5.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 4 | 3481 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.70, 1.09] |
| 8.5.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 1 | 328 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.22 [0.94, 1.60] |
| 8.6 A7: Severe lung disease | 6 | 4955 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.72, 0.97] |
| 8.6.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 5 | 4627 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.75 [0.63, 0.89] |
| 8.6.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 1 | 328 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.22 [0.94, 1.60] |
| 8.7 A8: Chronic lung disease | 9 | 5661 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.83, 1.22] |
| 8.7.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 6 | 4643 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.77, 1.19] |
| 8.7.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 3 | 1018 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.20 [0.80, 1.82] |
| 8.8 A9: Severe Intraventricular haemorrhage (grade 3 or 4) | 7 | 5066 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.69, 1.86] |
| 8.8.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 5 | 4161 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.53, 1.79] |
| 8.8.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 2 | 905 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.53 [0.63, 3.71] |
| 8.9 A10: Intraventricular haemorrhage | 6 | 3223 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.75, 1.19] |
| 8.9.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 4 | 2318 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.69, 1.24] |
| 8.9.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 2 | 905 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.69, 1.42] |
| 8.10 A11: Necrotising enterocolitis | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.59, 1.22] |
| 8.10.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 7 | 4831 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.61, 1.39] |
| 8.10.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 2 | 905 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.60 [0.27, 1.37] |
| 8.11 A12: Composite serious outcome (variously defined) | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.80, 0.97] |
| 8.11.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 7 | 4831 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.78, 1.00] |
| 8.11.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 2 | 905 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.75, 1.02] |
| 8.12 B2: Maternal sepsis | 8 | 4666 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.93, 1.39] |
| 8.12.1 For women planned for ≥ 1 repeat course of prenatal corticosteroids | 7 | 4417 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.10 [0.89, 1.36] |
| 8.12.2 For women planned for only 1 repeat course of prenatal corticosteroids | 1 | 249 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.57 [0.80, 3.10] |
| 8.13 B3: Caesarean section | 8 | 4266 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.98, 1.09] |
| 8.13.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 5 | 3495 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.06 [1.00, 1.13] |
| 8.13.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 3 | 771 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.83, 1.03] |
| 8.14 C1: Total deaths (after randomisation) up to early childhood follow‐up | 5 | 4565 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.06 [0.81, 1.40] |
| 8.14.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 4 | 4237 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.75, 1.32] |
| 8.14.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 1 | 328 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.31 [0.82, 6.50] |
| 8.15 C4: Survival free of major neurodevelopmental impairment at early childhood follow‐up | 3 | 1816 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.98, 1.05] |
| 8.15.1 In babies planned for ≥ 1 repeat course of prenatal corticosteroids | 2 | 1559 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.98, 1.07] |
| 8.15.2 In babies planned for only 1 repeat course of prenatal corticosteroids | 1 | 257 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.95, 1.01] |
Comparison 9. Subgroup analysis for planned interval between corticosteroid treatments.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 9.1 A1: Fetal or neonatal or infant death (< 1 year of age) | 10 | 5849 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.73, 1.24] |
| 9.1.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 7 | 2850 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.62, 1.28] |
| 9.1.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.1.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 3 | 2999 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.69, 1.52] |
| 9.2 A2: Fetal death | 7 | 2758 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.24, 2.84] |
| 9.2.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 5 | 2068 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.78 [0.19, 3.15] |
| 9.2.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.2.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 2 | 690 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.06, 15.86] |
| 9.3 A3: Neonatal death | 7 | 2758 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.62, 1.34] |
| 9.3.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 5 | 2068 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.62, 1.39] |
| 9.3.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.3.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 2 | 690 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.27, 2.61] |
| 9.4 A5: Respiratory distress syndrome | 9 | 3540 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.74, 0.90] |
| 9.4.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 7 | 2850 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.76, 0.95] |
| 9.4.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.4.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 2 | 690 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.70 [0.57, 0.87] |
| 9.5 A6: Severe respiratory distress syndrome | 5 | 3809 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.82, 1.16] |
| 9.5.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 4 | 1500 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.72, 1.10] |
| 9.5.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.5.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 1 | 2309 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.11 [0.82, 1.49] |
| 9.6 A7: Severe lung disease | 6 | 4955 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.72, 0.97] |
| 9.6.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 5 | 2646 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.75 [0.63, 0.89] |
| 9.6.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.6.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 1 | 2309 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.11 [0.82, 1.49] |
| 9.7 A8: Chronic lung disease | 9 | 5661 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.83, 1.22] |
| 9.7.1 In babies where pregnancy complicated by preterm prelabour rupture of membranes | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.7.2 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 6 | 2662 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.74, 1.14] |
| 9.7.3 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.7.4 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 3 | 2999 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.39 [0.91, 2.12] |
| 9.8 A9: Severe intraventricular haemorrhage (grade 3 or 4) | 7 | 5066 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.69, 1.86] |
| 9.8.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 5 | 2180 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.32 [0.69, 2.53] |
| 9.8.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.8.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 2 | 2886 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.42, 1.99] |
| 9.9 A10: Intraventricular haemorrhage | 6 | 3223 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.75, 1.19] |
| 9.9.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 5 | 2646 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.77, 1.27] |
| 9.9.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.9.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 1 | 577 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.76 [0.43, 1.34] |
| 9.10 A11: Necrotising enterocolitis | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.59, 1.22] |
| 9.10.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 7 | 2850 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.56, 1.40] |
| 9.10.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.10.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 2 | 2886 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.42, 1.42] |
| 9.11 A12: Composite serious outcome (variously defined) | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.80, 0.97] |
| 9.11.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 7 | 2850 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.76, 0.99] |
| 9.11.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.11.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 2 | 2886 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.76, 1.04] |
| 9.12 B2: Maternal sepsis | 8 | 4666 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.93, 1.39] |
| 9.12.1 For women treated with repeat corticosteroids at a minimum interval of 7 days or less | 6 | 2376 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.90, 1.42] |
| 9.12.2 For women treated with repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.12.3 For women treated with repeat corticosteroids at a minimum interval of 14 days or more | 2 | 2290 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.15 [0.73, 1.80] |
| 9.13 B3: Caesarean section | 8 | 4266 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.98, 1.09] |
| 9.13.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 5 | 1891 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.05 [0.97, 1.14] |
| 9.13.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.13.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 3 | 2375 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.95, 1.09] |
| 9.14 C1: Total deaths after randomisation up to early childhood follow‐up | 5 | 4565 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.06 [0.81, 1.40] |
| 9.14.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 4 | 2256 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.10 [0.75, 1.61] |
| 9.14.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.14.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 1 | 2309 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.69, 1.51] |
| 9.15 C2: Neurodevelopmental impairment at early childhood follow‐up | 4 | 3616 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.85, 1.10] |
| 9.15.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 3 | 1608 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.82, 1.10] |
| 9.15.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.15.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 1 | 2008 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.77, 1.32] |
| 9.16 C3: Survival free of neurodevelopmental impairment at early childhood follow‐up | 4 | 3845 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.98, 1.04] |
| 9.16.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 3 | 1741 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.96, 1.09] |
| 9.16.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.16.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 1 | 2104 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.97, 1.03] |
| 9.17 C5: Cerebral palsy at early childhood follow‐up | 5 | 3923 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.71, 1.49] |
| 9.17.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 4 | 1915 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.12 [0.68, 1.85] |
| 9.17.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.17.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 1 | 2008 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.53, 1.62] |
| 9.18 C6: Developmental delay or intellectual impairment at early childhood follow‐up (any) | 4 | 3581 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.84, 1.09] |
| 9.18.1 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 3 | 1680 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.94 [0.81, 1.09] |
| 9.18.2 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 9.18.3 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 1 | 1901 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.75, 1.32] |
Comparison 10. Subgroup analysis for the planned dose of corticosteroid given per treatment .
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 10.1 A1: Fetal or neonatal or infant death (< 1 year of age) | 10 | 5849 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.73, 1.24] |
| 10.1.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.12 [0.70, 1.77] |
| 10.1.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 8 | 4375 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.64, 1.21] |
| 10.1.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.2 A2: Fetal death | 7 | 2758 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.24, 2.84] |
| 10.2.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.15, 7.31] |
| 10.2.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 5 | 1284 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.70 [0.14, 3.55] |
| 10.2.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.3 A3: Neonatal death | 7 | 2758 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.62, 1.34] |
| 10.3.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.12 [0.70, 1.80] |
| 10.3.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 5 | 1284 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.32, 1.20] |
| 10.3.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.4 A5: Respiratory distress syndrome | 9 | 3540 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.74, 0.90] |
| 10.4.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.86 [0.76, 0.98] |
| 10.4.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 7 | 2066 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.66, 0.89] |
| 10.4.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.5 A6: Severe respiratory distress syndrome | 5 | 3809 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.82, 1.16] |
| 10.5.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 1 | 328 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.22 [0.94, 1.60] |
| 10.5.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 4 | 3481 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.70, 1.09] |
| 10.5.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.6 A7: Severe lung disease | 6 | 4955 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.72, 0.97] |
| 10.6.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.66, 0.97] |
| 10.6.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 4 | 3481 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.70, 1.09] |
| 10.6.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.7 A8: Chronic lung disease | 9 | 5661 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.83, 1.22] |
| 10.7.1 In babies where planned dose per treatment course 12 mg or less of betamethasone or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.74, 1.27] |
| 10.7.2 In babies where planned dose per treatment course > 12 mg to 24 mg of betamethasone or equivalent | 7 | 4187 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.79, 1.38] |
| 10.7.3 In babies where planned dose per treatment course > 24 mg of betamethasone or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.8 A9: Severe intraventricular haemorrhage (grade 3 or 4) | 7 | 5066 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.69, 1.86] |
| 10.8.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.42, 2.12] |
| 10.8.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 5 | 3592 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.26 [0.67, 2.38] |
| 10.8.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.9 A10: Intraventricular haemorrhage | 6 | 3223 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.75, 1.19] |
| 10.9.1 In babies where planned dose per treatment course 12 mg or less of betamethasone or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.74, 1.40] |
| 10.9.2 In babies where planned dose per treatment course > 12 mg to 24 mg of betamethasone or equivalent | 4 | 1749 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.64, 1.22] |
| 10.9.3 In babies where planned dose per treatment course > 24 mg of betamethasone or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.10 A11: Necrotising enterocolitis | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.59, 1.22] |
| 10.10.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.41 [0.16, 1.05] |
| 10.10.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 7 | 4262 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.66, 1.47] |
| 10.10.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.11 A12: Composite serious outcome (variously defined) | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.80, 0.97] |
| 10.11.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.75, 1.03] |
| 10.11.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 7 | 4262 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.77, 1.00] |
| 10.11.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.12 B2: Maternal sepsis | 8 | 4666 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.93, 1.39] |
| 10.12.1 For women treated where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1231 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.19 [0.84, 1.69] |
| 10.12.2 For women where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 6 | 3435 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.10 [0.86, 1.42] |
| 10.12.3 For women where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.13 B3: Caesarean section | 8 | 4266 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.98, 1.09] |
| 10.13.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1231 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.10 [1.01, 1.20] |
| 10.13.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 6 | 3035 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.94, 1.07] |
| 10.13.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.14 C1: Total deaths after randomisation up to early childhood follow‐up | 5 | 4565 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.06 [0.81, 1.40] |
| 10.14.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.11 [0.72, 1.71] |
| 10.14.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 3 | 3091 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.73, 1.47] |
| 10.14.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.15 C2: Neurodevelopmental impairment at early childhood follow‐up | 4 | 3616 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.85, 1.10] |
| 10.15.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 1 | 999 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.83, 1.16] |
| 10.15.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 3 | 2617 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.77, 1.17] |
| 10.15.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.16 C3: Survival free of neurodevelopmental impairment at early childhood follow‐up | 4 | 3845 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.98, 1.04] |
| 10.16.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 1 | 1060 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.92, 1.12] |
| 10.16.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 3 | 2785 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.97, 1.04] |
| 10.16.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.17 C4: Survival free of major neurodevelopmental impairment at early childhood follow‐up | 3 | 1816 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.98, 1.05] |
| 10.17.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1317 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.98, 1.07] |
| 10.17.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 1 | 499 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.92, 1.06] |
| 10.17.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.18 C5: Cerebral palsy at early childhood follow‐up | 5 | 3923 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.71, 1.49] |
| 10.18.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1306 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.94 [0.55, 1.62] |
| 10.18.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 3 | 2617 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.12 [0.67, 1.86] |
| 10.18.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 10.19 C6: Developmental delay or intellectual impairment at early childhood follow‐up (any) | 4 | 3581 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.84, 1.09] |
| 10.19.1 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1194 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.81, 1.14] |
| 10.19.2 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 2 | 2387 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.77, 1.17] |
| 10.19.3 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
Comparison 11. Subgroup analysis for the planned dose of repeat dose of corticosteroid drug exposure/week.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 11.1 A1: Fetal or neonatal or infant death (< 1 year of age) | 7 | 4756 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.68, 1.21] |
| 11.1.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 3455 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.73, 1.40] |
| 11.1.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 5 | 1301 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.59 [0.31, 1.14] |
| 11.1.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.2 A2: Fetal death | 4 | 1740 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.14, 3.57] |
| 11.2.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 1 | 1146 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.06, 16.23] |
| 11.2.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 3 | 594 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.59 [0.08, 4.42] |
| 11.2.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.3 A3: Neonatal death | 4 | 1740 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.51, 1.23] |
| 11.3.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 1 | 1146 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.94 [0.56, 1.59] |
| 11.3.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 3 | 594 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.23, 1.19] |
| 11.3.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.4 A5: Respiratory distress syndrome | 5 | 2334 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.72, 0.93] |
| 11.4.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 1 | 1146 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.79 [0.68, 0.92] |
| 11.4.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 4 | 1188 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.69, 1.12] |
| 11.4.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.5 A6: Severe respiratory distress syndrome | 4 | 3481 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.70, 1.09] |
| 11.5.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 1 | 2309 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.11 [0.82, 1.49] |
| 11.5.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 3 | 1172 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.44, 0.88] |
| 11.5.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.6 A7: Severe lung disease | 5 | 4627 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.75 [0.63, 0.89] |
| 11.6.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 3455 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.65, 0.97] |
| 11.6.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 3 | 1172 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.44, 0.88] |
| 11.6.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.7 A8: Chronic lung disease | 6 | 4641 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.77, 1.19] |
| 11.7.1 In babies where planned repeat drug exposure was 12 mg or less/week of betamethasone or equivalent | 2 | 3455 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.79, 1.35] |
| 11.7.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of betamethasone or equivalent | 4 | 1186 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.56, 1.19] |
| 11.7.3 In babies where planned repeat drug exposure was > 24 mg/week of betamethasone or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.8 A9: Severe intraventricular haemorrhage (grade 3 or 4) | 4 | 3973 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.53, 1.79] |
| 11.8.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 3455 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.65 [0.30, 1.37] |
| 11.8.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 2 | 518 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.50 [0.76, 8.22] |
| 11.8.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.9 A10: Intraventricular haemorrhage | 4 | 2318 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.69, 1.24] |
| 11.9.1 In babies where planned repeat drug exposure was 12 mg or less/week of betamethasone or equivalent | 1 | 1146 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.57, 1.38] |
| 11.9.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of betamethasone or equivalent | 3 | 1172 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.95 [0.64, 1.41] |
| 11.9.3 In babies where planned repeat drug exposure was > 24 mg/week of betamethasone or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.10 A11: Necrotising enterocolitis | 6 | 4643 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.79 [0.51, 1.22] |
| 11.10.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 3455 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.65 [0.34, 1.24] |
| 11.10.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 4 | 1188 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.52, 1.68] |
| 11.10.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.11 A12: Composite serious outcome (variously defined) | 6 | 4643 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.76, 0.99] |
| 11.11.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 3455 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.77, 1.05] |
| 11.11.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 4 | 1188 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.79 [0.61, 1.02] |
| 11.11.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.12 B2: Maternal sepsis | 5 | 3824 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.21 [0.96, 1.52] |
| 11.12.1 For women where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 2835 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.18 [0.86, 1.62] |
| 11.12.2 For women where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 3 | 989 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.24 [0.90, 1.73] |
| 11.12.3 For women where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.13 B3: Caesarean section | 4 | 3339 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.08 [1.01, 1.14] |
| 11.13.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 2835 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.09 [1.02, 1.16] |
| 11.13.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 2 | 504 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.80, 1.25] |
| 11.13.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.14 C1: Total deaths after randomisation up to early childhood follow‐up | 3 | 4049 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.74, 1.33] |
| 11.14.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 3455 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.72, 1.33] |
| 11.14.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 1 | 594 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.15 [0.39, 3.38] |
| 11.14.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.15 C2: Neurodevelopmental impairment at early childhood follow‐up | 3 | 3493 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.85, 1.11] |
| 11.15.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 3007 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.86, 1.14] |
| 11.15.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 1 | 486 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.63, 1.22] |
| 11.15.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.16 C3: Survival free of neurodevelopmental impairment at early childhood follow‐up | 3 | 3663 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.97, 1.04] |
| 11.16.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 3164 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.97, 1.04] |
| 11.16.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 1 | 499 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.94, 1.14] |
| 11.16.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.17 C4: Survival free of major neurodevelopmental impairment at early childhood follow‐up | 2 | 1559 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.98, 1.07] |
| 11.17.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 1 | 1060 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.99, 1.10] |
| 11.17.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 1 | 499 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.92, 1.06] |
| 11.17.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.18 C5: Cerebral palsy at early childhood follow‐up | 3 | 3541 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.69, 1.47] |
| 11.18.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 3055 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.91 [0.61, 1.35] |
| 11.18.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 1 | 486 | Risk Ratio (M‐H, Fixed, 95% CI) | 5.76 [0.70, 47.47] |
| 11.18.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 11.19 C6: Developmental delay or intellectual impairment at early childhood follow‐up (any) | 3 | 3386 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.84, 1.10] |
| 11.19.1 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 2900 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.85, 1.14] |
| 11.19.2 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 1 | 486 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.65, 1.17] |
| 11.19.3 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
Comparison 12. Subgroup analysis for the gestational age at which the first repeat treatment was given.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 12.1 A12: Composite serious outcome (variously defined) | 9 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 12.1.1 A12: Composite serious outcome (variously defined); intention to treat analysis | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.80, 0.97] |
| 12.1.2 A12: Composite serious outcome (variously defined); per protocol analysis | 9 | 5599 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.80, 0.98] |
| 12.1.3 In babies where pregnancy complicated by preterm prelabour rupture of membranes | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 12.1.4 In babies from singleton pregnancies | 2 | 1646 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.06 [0.86, 1.30] |
| 12.1.5 In babies from multiple pregnancies | 2 | 991 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.80, 1.31] |
| 12.1.6 In babies exposed to repeat corticosteroids as betamethasone | 8 | 5159 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.82, 1.03] |
| 12.1.7 In babies exposed to repeat corticosteroids at a minimum interval of 7 days or less | 7 | 2850 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.76, 0.99] |
| 12.1.8 In babies exposed to repeat corticosteroids at a minimum interval between 8 and < 14 days | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 12.1.9 In babies exposed to repeat corticosteroids at a minimum interval of 14 days or more | 2 | 2886 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.76, 1.04] |
| 12.1.10 In babies where planned dose per treatment course 12 mg or less of prenatal corticosteroid or equivalent | 2 | 1474 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.75, 1.03] |
| 12.1.11 In babies where planned dose per treatment course > 12 mg to 24 mg of prenatal corticosteroid or equivalent | 7 | 4262 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.77, 1.00] |
| 12.1.12 In babies where planned dose per treatment course > 24 mg of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 12.1.13 In babies where planned repeat drug exposure was 12 mg or less/week of prenatal corticosteroid or equivalent | 2 | 3455 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.77, 1.05] |
| 12.1.14 In babies where planned repeat drug exposure was > 12 mg/week to 24 mg/week of prenatal corticosteroid or equivalent | 4 | 1188 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.79 [0.61, 1.02] |
| 12.1.15 In babies where planned repeat drug exposure was > 24 mg/week of prenatal corticosteroid or equivalent | 0 | 0 | Risk Ratio (M‐H, Fixed, 95% CI) | Not estimable |
| 12.1.16 In babies where prenatal corticosteroid was administered intramuscularly | 9 | 5736 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.86 [0.78, 0.95] |
| 12.1.17 In babies given their first repeat course at less than 28 completed weeks' gestational age | 1 | 181 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.71, 1.41] |
| 12.1.18 In babies planned for one repeat course of prenatal corticosteroids | 2 | 905 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.75, 1.02] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Aghajafari 2002.
| Study characteristics | ||
| Methods | Type of study: parallel, randomised, placebo‐controlled trial Small pilot study to determine the feasibility of a larger trial |
|
| Participants | Location: 2 hospitals in Toronto, Canada Dates of study: September 1999 to August 2000 Eligibility criteria: women at 24–30 weeks' gestation at continued increased risk of preterm birth who remained undelivered ≥ 7 days following a single course of prenatal corticosteroids (defined as 2 doses of intramuscular betamethasone 12 mg/dose, given at 12‐ or 24‐hour intervals; or 4 doses of intramuscular dexamethasone 5–6 mg/dose, given at 12‐hour intervals). At increased risk of preterm birth, women had to have ≥ 1 of the following: regular uterine contractions; shortened cervical length or cervical dilation; preterm prelabour rupture of the membranes; prepartum bleeding secondary to placental separation or placenta praevia; history of preterm birth; maternal hypertension; other medical condition increasing the risk of preterm delivery or intrauterine growth restriction; or other fetal conditions increasing the risk of preterm delivery Gestational age: range 24–30 weeks Exclusion criteria: required chronic doses of corticosteroids secondary to medical conditions; contraindication to corticosteroids; clinical evidence of chorioamnionitis or if their fetus(es) had a known lethal congenital anomaly Total recruited: 12 women (16 fetuses): 6 women (9 fetuses) in multiple course of prenatal corticosteroid group and 6 women (7 fetuses) in placebo group |
|
| Interventions | Multiple course of prenatal corticosteroid group: weekly course of
betamethasone (2 doses of betamethasone 12 mg/dose (Celestone Soluspan;
Schering Canada Inc) intramuscularly, 24 hours apart) until 33 weeks or
delivery if the woman remained at increased risk of preterm birth Placebo group: weekly course of placebo consisting of 2 doses of normal saline, intramuscularly 24 hours apart, until 33 weeks or delivery if the woman remained at increased risk of preterm birth |
|
| Outcomes | Rate of recruitment over 12‐month period, risk of complications requiring
discontinuation of study treatment, concentrations of plasma cortisol and
ACTH in cord blood and in maternal blood immediately following birth Perinatal or neonatal mortality or significant neonatal morbidity, defined as ≥ 1 of the following: stillborn or neonatal death during the first 28 days of life or prior to hospital discharge, whichever was sooner; RDS; BPD (requiring oxygen at 36 corrected postnatal gestational age); IVH (grade 3 or 4); and necrotising enterocolitis |
|
| Funding Support | Support from Canadian Institutes of Health Research Senior Scientist Award | |
| Notes | Sample‐size calculation: no. Declarations of interest: declared no conflict of interest. Ethical approval: approved by the research ethics committees of participating hospitals. Trial registration: not located. |
|
Crowther 2006.
| Study characteristics | ||
| Methods | Type of study: parallel, randomised, placebo‐controlled trial | |
| Participants | Location: 23 hospitals in Australia and New Zealand Dates of study: April 1998 to July 2004 Inclusion criteria: single, twin or triplet pregnancy at < 32 weeks' gestation if women had received an initial treatment of corticosteroid ≥ 7 days previously and their responsible clinician regarded them to be at continued risk of preterm birth, and there was no contraindication to further corticosteroid therapy Exclusion criteria: in second stage of labour, had chorioamnionitis needing urgent delivery, or if further corticosteroid therapy was judged essential Gestational age: recruited up to < 32 weeks Total recruited: 982 women (1146 fetuses); 489 women (568 fetuses) in repeat corticosteroid group and 493 women (578 fetuses) in placebo group |
|
| Interventions | Repeat corticosteroids group: betamethasone 11.4 mg (Celestone Chronodose)
(as betamethasone sodium phosphate 7.8 mg and betamethasone acetate 6 mg by
intramuscular injection Placebo group: saline intramuscular injection Every week, if the woman remained undelivered and < 32 weeks' gestation, and the responsible clinician regarded her as at continued risk of preterm birth, a further treatment pack from the same treatment group was allocated by the telephone randomisation service |
|
| Outcomes | To time of primary hospital discharge: primary outcomes: frequency and
severity of RDS (defined as clinical signs of respiratory distress and a
ground‐glass appearance on chest radiograph); weight, length and head
circumference at birth and primary discharge from hospital. Secondary
outcomes included clinical chorioamnionitis (defined as requiring
intrapartum antibiotics); maternal postpartum pyrexia (≥ 38.0 °C); any
adverse effects of the injection for the mother and other measures of
neonatal morbidity. Composite outcome was post hoc (defined as 1 of air leak
syndrome, patent ductus arteriosus, need for oxygen at 36 weeks'
postmenstrual age, severe IVH (grade 3 or 4), periventricular haemorrhage,
proven necrotising enterocolitis or retinopathy of prematurity) For the early childhood follow‐up at 2 years' corrected age: primary outcomes: survival at 2 years' corrected age free of major neurosensory disability, defined as survival free of moderate‐to‐severe disability. Body size (weight, height and head circumference). Secondary outcomes were general health, including the use of health services since primary hospital discharge; respiratory morbidity; blood pressure; child behaviour; incidence of neurosensory impairments and disabilities; total number of deaths by 2 years of corrected age; and the combined adverse outcome of death or any neurosensory disability at 2 years' corrected age For the mid‐childhood follow‐up at 6–8 years' corrected age: primary outcomes: survival free of any neurosensory disability and categorisation of neurosensory disability as none, mild, moderate or severe. Secondary outcomes were mortality; cerebral palsy; blindness or deafness; Z scores for height, weight, BMI and head circumference; expiratory flows on lung function; blood pressure Z scores and proportions in the abnormal ranges; IQ; attention and executive function; memory and learning; visual perception; academic achievement; behaviour; health service utilisation and reason for use; general health and health‐related quality of life |
|
| Funding Support | Funding: Australian National Health and Medical Research Council, The Channel 7 Research Foundation of South Australia, The Women's and Children's Hospital Research Foundation, Adelaide, and The Department of Obstetrics and Gynaecology, The University of Adelaide, South Australia | |
| Notes | Sample‐size calculation: yes. 980 women needed to detect a 25% reduction in
the risk of RDS from 30% to 22.5% with 80% power and a 2‐sided significance
level of 5%. Declarations of interest: declared no conflict of interest. Ethical approval: yes, obtained at each of the 23 collaborating hospitals. Trial registration: during recruitment. |
|
Garite 2009.
| Study characteristics | ||
| Methods | Type of study: parallel, randomised, placebo‐controlled trial | |
| Participants | Location: 15 private and 3 university centres, USA. Participants were
largely private/non‐government funded Dates of study: May 2003 to February 2008 Included: women with singleton or twin pregnancies from 25 weeks' to < 33 weeks' gestation who had received a course of betamethasone ≥ 14 days previously and who were judged to have recurrent or continued risk of preterm birth Exclusion criteria: major fetal anomaly, cervical dilatation ≥ 5 cm, higher order multiples, ruptured membranes, documented lung maturity, receiving corticosteroids for other indications, HIV infection or active tuberculosis Total recruited: 437 women; 223 in repeat corticosteroids group and 214 in placebo group. 577 infants enrolled; 289 in repeat corticosteroids group and 288 in placebo group, although 1 fetal twin in the corticosteroid group died before randomisation |
|
| Interventions | Repeat corticosteroid group: single course of intramuscular betamethasone
given as 2 doses of 12 mg, 24 hours apart (preparation not specified) Placebo group: similarly administered saline intramuscular injection In some centres betamethasone became unavailable and was replaced with dexamethasone 6 mg intramuscularly, 4 doses, every 12 hours. 31 women received dexamethasone and 30 women received an equivalent placebo |
|
| Outcomes | Primary outcome: composite neonatal mortality/morbidity in babies born
before 34 weeks' gestation. Composite outcome defined as ≥ 1 of: perinatal
death (defined as stillbirth or death before neonatal discharge); RDS
(oxygen requirement, clinical diagnosis and consistent chest radiograph);
BPD (defined as a requirement for oxygen at 30 days of age); severe IVH
(grades 3 or 4); PVL; blood culture‐proven sepsis; or necrotising
enterocolitis (not defined) Secondary outcomes: preterm birth before 34 weeks' gestation; RDS; gestational age at birth; small‐for‐gestational age (< 10th percentile); head circumference; birthweight; surfactant therapy; pneumothorax; maternal infectious morbidity |
|
| Funding Support | Funding: Pediatrix Medical Group | |
| Notes | Sample‐size calculation: yes, based on a 40% reduction in the primary
outcome, which was estimated to be 28% in the control group. The planned
sample size was 217 women in each arm (2‐tailed alpha 0.05, beta 0.2). Declarations of interest: declared no conflict of interest. Ethical approval: yes, at each institution. Trial registration: during recruitment. |
|
Guinn 2001.
| Study characteristics | ||
| Methods | Type of study: parallel, randomised, placebo‐controlled trial | |
| Participants | Location: 13 academic centres in the US Dates of study: February 1996 to April 2000 Inclusion criteria: women at 24 weeks' to < 33 weeks' gestation at high risk of preterm birth who remained undelivered 1 week following an initial course of prenatal corticosteroids (defined as 2 doses of intramuscular betamethasone 12 mg/dose, repeated at 24 hours; or 4 doses of intramuscular dexamethasone 6 mg/dose, given at 12 hour‐intervals). High risk of preterm birth qualifying criteria were: preterm labour with intact membranes (either a history of regular uterine contractions associated with cervical dilation of ≥ 2 cm and effacement ≥ 80% in a nulliparous participant or cervical dilation of ≥ 3 cm and ≥ 80% effacement in a multiparous participant at the time of presentation; or regular uterine contractions with documented cervical change); preterm prelabour rupture of the membranes (rupture of the membranes occurring > 1 hour prior to the onset of preterm labour); maternal medical illness (pre‐eclampsia, hypertension, diabetes, renal disease, systemic lupus erythematosus, trauma); or suspected fetal jeopardy (intrauterine growth restriction < 10th percentile, oligohydramnios, abnormal prepartum testing, progression of a fetal anomaly compatible with like, twin–twin transfusion syndrome) Gestational age range: 24 weeks to < 33 weeks' gestation Exclusion criteria: required immediate delivery, fetal anomalies incompatible with life, documented fetal lung maturity, maternal active tuberculosis or HIV infection Total recruited: 502 women (589 fetuses); 256 women in weekly course group and 246 women in single‐course group. Outcomes reported using the pregnancy as the unit of analysis. For infant outcomes from multiple gestations, a single infant was randomly selected for analysis |
|
| Interventions | Weekly course group: weekly course of betamethasone (2 doses of
betamethasone 12 mg/dose repeated after 24 hours, intramuscularly), until 34
weeks or birth whichever came first Single‐course group: similarly administered placebo |
|
| Outcomes | Primary outcomes: composite neonatal morbidity defined as presence of any
of the following: severe RDS, BPD, severe IVH, PVL, necrotising
enterocolitis, proven sepsis or death between randomisation and nursery
discharge Secondary outcomes: frequency and severity of RDS; need for and duration of oxygen therapy; need for and duration of ventilatory support; BPD (defined as need for oxygen > 21% and usually ventilatory therapy for ≥ 28 days of life; in cases were no additional ventilatory support was needed but oxygen was required, chest radiographs consistent with BPD were used; in the case of neonatal death, BPD was diagnosed on postmortem findings); severe IVH was defined as intraventricular bleeding with dilation of the cerebral ventricles (grade 3) or parenchymal haemorrhage (grade 4), as diagnosed with an imaging technique or postmortem, PVL was defined as the presence of > 1 obvious hypoechoic cyst in the periventricular white matter; necrotising enterocolitis (defined as proven); proven sepsis; perinatal death defined as death of a fetus or neonate at any time between randomisation and nursery discharge |
|
| Funding Support | Funding: March of Dimes grant, the Berlex Foundation, the Wisconsin Perinatal Association, the Perinatal Clinical Research Center at the University of Colorado Health Sciences Center (grant from the General Clinical Research Centers Program, National Centers for Research Resources, National Institutes of Health), and the participating departments | |
| Notes | Sample‐size calculation: yes. A sample of 1000 women was required to have a
90% power to detect a 1/3 reduction in composite morbidity from 25.0% to
16.5% (2‐tailed alpha = 0.05). 2 interim analyses were planned for efficacy
and safety. Recruitment was stopped early based on safety concerns. Declarations of interest: no declaration. Ethical approval: the investigational review board at each centre approved the study protocol. Trial registration: not located. |
|
Mazumder 2008.
| Study characteristics | ||
| Methods | Type of study: parallel, open‐label, randomised trial | |
| Participants | Location: tertiary hospital in northern India Dates of study: August 2004 onwards Included: mothers 26–33 weeks' gestation who were at risk of preterm birth and who had received a course of betamethasone ≥ 7 days previously. Mothers had to be available for follow‐up every week until birth Exclusion criteria: unreliable gestational age, frank chorioamnionitis and major fetal malformation Total recruited: 76 women (38 in repeat corticosteroids group and 38 in control group). 83 babies were born to 75 mothers but only the firstborn of multiple pregnancies were assessed (37 repeat corticosteroids, 38 control). The pregnancy was used as the unit of analysis but for multiple pregnancies it was not detailed how it was decided which infant would contribute to the outcome |
|
| Interventions | Repeat corticosteroid: betamethasone 12 mg intramuscularly, 2 doses, 24
hours apart. The course was repeated every 7 days until delivery or the end
of the 33rd week of gestation Control: no intervention |
|
| Outcomes | Primary outcome: severe RDS. RDS defined as respiratory distress within 6
hours of birth in a preterm infant with either a negative gastric shake test
or a typical chest radiograph. Severe RDS defined as requiring mechanical
ventilation for ≥ 12 hours. Mechanical ventilation started in infants with
hypoxaemia (PaO2 < 50 mmHg) or hypercapnic acidosis
(PaCO2 > 50 mmHg with pH < 7.25) or worsening acidosis or
clinically worsening respiratory fatigue/apnoea/work of breathing despite
continuous positive airway pressure (maximum 8 cmH2O
pressure) Secondary outcomes: RDS; IVH; necrotising enterocolitis (not defined); patent ductus arteriosus (not defined); BPD (not defined), sepsis (not defined), retinopathy of prematurity, stillbirth and neonatal death, weight, length and occipital‐frontal circumference at birth and at 6 months' corrected age, and body size and development (evaluated by the Denver Development Screening Test II) at 6 months' corrected age |
|
| Funding Support | None | |
| Notes | Sample‐size calculation: yes, based on a reduction in severe RDS from an
estimated 33% in the control group to 6% in the repeat corticosteroid group
(2‐tailed alpha 0.05, beta 0.2). The planned sample size was 70 (35 women in
each arm). Declarations of interest: declared no conflict of interest. Ethical approval: approved by the institute ethics committee. Trial registration: not located. |
|
McEvoy 2002.
| Study characteristics | ||
| Methods | Type of study: parallel, randomised, placebo‐controlled trial | |
| Participants | Location: single centre in the USA (Sacred Heart Hospital, University of
Florida, Pensacola, Florida) Dates of study: 3‐year period ending in December 1999 Eligibility criteria: women at 25–33 weeks' gestation who remained undelivered 1 week after a single course of prenatal corticosteroids (defined as 2 doses of betamethasone 12 mg/dose intramuscular), given because of increased risk of preterm delivery Gestational age range: 25–33 weeks Exclusion criteria: insulin‐dependent diabetes, drug‐addiction, fetus had a known lethal congenital anomaly Total recruited: 37 women (37 babies). 18 women in repetitive courses of prenatal corticosteroid group and 19 women in single course remote group |
|
| Interventions | Repeat course prenatal corticosteroid group: weekly course of betamethasone
(2 doses of betamethasone 12 mg/dose (Celestone Soluspan; Schering
Corporation, Kenilworth, New Jersey)), intramuscularly, until birth or 34
weeks' gestation Single‐course remote group: weekly courses of placebo intramuscularly, until 34 weeks or birth |
|
| Outcomes | Primary outcomes: functional residual capacity, respiratory compliance Secondary outcomes: admission head circumference, surfactant administration, days on oxygen and mechanical ventilation |
|
| Funding Support | American Lung Association | |
| Notes | Sample‐size calculation: yes. Based on 37 women the mean functional
residual capacity in the single course remote group was not > 12% smaller
than the functional residual capacity in the repetitive group (P = 0.05,
power 80%). Declarations of interest: no declaration of interest. Ethical approval: approved by the hospital's institutional review board. Trial registration: not located. |
|
McEvoy 2010.
| Study characteristics | ||
| Methods | Type of study: parallel, randomised, placebo‐controlled trial | |
| Participants | Location: 2 centres in the US (Sacred Heart Hospital, University of
Florida, Pensacola, Florida recruited first 8 participants; Oregon Health
and Science University) Dates of study: June 2001 to May 2007 Eligibility criteria: women at 26 to < 34 weeks' gestation; ≥ 14 days after first course of prenatal corticosteroids (93% received betamethasone); at continued risk of preterm delivery as determined by their care provider; who provided informed consent Gestational age range: 26 to < 34 weeks' gestation Exclusion criteria: insulin‐dependent diabetes, major documented fetal or chromosomal abnormality; multiple pregnancy greater than twins; clinical chorioamnionitis; first course of prenatal corticosteroids given < 24 weeks' gestation; chronic steroid use during pregnancy for clinical care Total recruited: 85 women randomised (113 babies alive at randomisation); 44 women (56 babies) in rescue corticosteroids group and 41 women (57 babies) in placebo group |
|
| Interventions | Rescue group: betamethasone 2 doses of 12 mg/dose (Celestone Soluspan;
Schering Corporation, Kenilworth, New Jersey), intramuscularly, 24 hours
apart Placebo group: placebo 2 doses (cortisone acetate 25 mg, an inactive steroid identical in appearance to betamethasone) |
|
| Outcomes | Primary outcomes: respiratory compliance and functional residual capacity
(measured within 72 hours of birth (before any surfactant) Secondary outcomes: growth measurements including weight, head circumference and length at birth and hospital discharge; surfactant administration; diagnosis of RDS (defined as clinical signs of respiratory distress with radiographic appearance and needing supplemental oxygen with FiO2 > 0.21); respiratory distress requiring ≥ 0.30 and ≥ 0.40 at 24 hours of age; days on mechanical ventilation and days on supplemental oxygen |
|
| Funding Support | Oregon Health and Science University, GCRC/PHS Grant 5 MO1 RR000334; OCTRI UL1 RR02414001; and The American Lung Association | |
| Notes | Recruitment was terminated early, after 85 women had been randomised,
primarily because of safety concerns of possible adverse effects of weekly
prenatal corticosteroid therapy. Declarations of interest: no conflict of interest. Ethics approval: approved by the institutional review board at each institution. Trial registration: retrospective. |
|
Murphy 2008.
| Study characteristics | ||
| Methods | Type of study: parallel, randomised, placebo‐controlled trial | |
| Participants | Location: 80 centres, 20 countries Dates of study: April 2001 to August 2006 Inclusion criteria: single, twin or triplet pregnancy 25–32 weeks' gestation if women remained undelivered 14–21 days after an initial course of prenatal corticosteroids (either betamethasone or dexamethasone) and continued to be at high risk of preterm birth Gestational age recruited: up to < 32 weeks Exclusion criteria: contraindication to corticosteroid use, needed chronic doses of these drugs, evidence of chorioamnionitis, carried a fetus with a known lethal congenital abnormality, had an initial course of corticosteroids before 23 weeks' gestation, previously participated in MACS, women with a multiple pregnancy with fetal death after 13 weeks' gestation Total recruited: 1858 women (2309 babies); 935 women (1166 infants) in prenatal corticosteroid group and 918 women (1143 infants) in placebo group |
|
| Interventions | Repeat corticosteroid group: each course consisted of 2 intramuscular
injections of betamethasone 12 mg (as betamethasone sodium phosphate 6 mg
and betamethasone acetate 6 mg; Celestone Schering‐Plough Corporation,
Madison, New Jersey) 24 hours apart Placebo group: similarly appearing intramuscular injection of dilute concentration of aluminium monostearate (an inert substance used as a filler in pharmaceutical preparations) Women who remained at risk of preterm birth after their first course of study medication continued to receive 2 doses of betamethasone 12 mg or placebo, 24 hours apart, every 14 days until 33 weeks' gestation or birth, whichever happened first. For women with preterm rupture of the membranes the recommendation was to stop the study medication at 32 weeks' gestation |
|
| Outcomes | Primary outcomes: composite of perinatal or neonatal mortality and neonatal
morbidity; 1 of the following: stillbirth or neonatal death (defined as
death during the first 28 days of life or before hospital discharge); severe
RDS (defined as needing assisted ventilation via endotracheal tube and
supplemental oxygen within the first 24 hours of life and for ≥ 24 hours,
and either a radiographic scan compatible with RDS or surfactant given
between the first 2 and 24 hours of life); BPD (defined as needing oxygen at
a postmenstrual age of 36 completed weeks and radiographic scan compatible
with BPD); IVH grade III or IV; cystic PVL, necrotising enterocolitis For the early childhood follow‐up at 2 years' corrected age: primary outcome was death or the presence of a neurological impairment at 18–24 months of age, corrected for gestational age at birth. Neurological impairment defined as presence of cerebral palsy or cognitive delay. Cerebral palsy diagnosed if child had a non‐progressive motor impairment characterised by abnormal muscle tone and decreased range of movements. Cognitive delay defined as Mental Developmental Index scores of 70 (2 SDs below the mean of 100) on the BSID‐II or equivalent scores on another standardised assessment Secondary outcomes: anthropometric measurements (weight, height and head circumference), general health, illnesses, and operations occurring after the primary hospitalisation was recorded, Psychomotor Developmental Index and the Behavior Rating Scale of the BSID‐II For the 5 years of age childhood follow‐up: primary outcome: composite of death or survival with a neurodevelopmental disability in ≥ 1 of the following domains: neuromotor (non‐ambulatory cerebral palsy), neurosensory (blindness, deafness, or need for visual or hearing aids), or neurocognitive/neurobehavioural function (abnormal attention, memory, or behaviour) Secondary outcomes: growth (height, weight and head circumference) and blood pressure. For those in Canadian centres, intelligence (The Wechsler Preschool and Primary Scale of Intelligence – Third Edition) and specific cognitive skills were assessed (Test of Visual‐Motor Integration – Fifth Edition for visual and motor abilities and integration; and the Peabody Picture Vocabulary Test – Third Edition for vocabulary knowledge development and receptive language abilities) |
|
| Funding Support | Funding: Canadian Institutes of Health Research (CIHR) | |
| Notes | Sample‐size calculation: yes. A sample size of 1900 women (950 per group)
was needed to have 80% probability of achieving a significant difference
between groups (2‐tailed type 1 error 0.05), if multiple courses of prenatal
corticosteroids reduced the risk of RDS from 12% to 8%. Declarations of interest: declared no conflict of interest. Ethics approval: from University of Toronto, Mount Sinai Hospital and all participating centres. Trial registration: during recruitment (ISRCTN 72654148). |
|
Peltoniemi 2007.
| Study characteristics | ||
| Methods | Type of study: parallel, placebo‐controlled, randomised trial | |
| Participants | Location: 5 Finnish university and 3 central hospitals Dates of study: May 2001 to March 2005 Included: women at < 34 weeks' gestation who had received a single course of betamethasone > 7 days previously and were to have elective delivery within 48 hours or were at very high risk of spontaneous delivery within 48 hours (cervical opening ≥ 3 cm, and regular contractions at 5‐ to 10‐minute intervals) Gestational ages: < 34 weeks' gestation Exclusion criteria: long‐term maternal corticosteroid use, clinical chorioamnionitis or lethal disease of the fetus Total recruited: 249 women (125 in betamethasone group and 124 in placebo group), 328 fetuses (160 in betamethasone group and 168 in placebo group) |
|
| Interventions | Repeat corticosteroid: a single dose betamethasone 12 mg
intramuscularly Placebo: isotonic saline intramuscularly |
|
| Outcomes | To time of primary hospital discharge: primary outcome: survival without
severe RDS or severe IVH (grade 3 or 4) during the first hospital admission.
RDS defined on the basis of typical chest radiograph findings, requirement
for continuous distending airway pressure, supplemental oxygen for ≥ 48
hours, or requirement for surfactant in cases of established respiratory
failure. Severe IVH defined as IVH with ventricular dilation (grade 3) or
parenchymal haemorrhage (grade 4). Cranial ultrasound performed for all
infants at 4–8 days of age and at 36 weeks postmenstrual age or before
discharge. The most severe grade of IVH was recorded Secondary outcomes: cystic PVL; necrotising enterocolitis grade ≥ 2; BPD (defined as a requirement for supplemental oxygen or any form of ventilation with continuous distending pressures at postnatal age of 36 weeks or at postnatal age of 4 weeks for those born after postmenstrual age of 31 weeks); patent ductus arteriosus requiring treatment (defined as a requirement for prostaglandin inhibitor therapy or surgery for closure) For the early childhood follow‐up at 2 years' corrected age: survival without serious neurological, cognitive or sensory impairment (NDI); respiratory problems; infections, medical history; child's weight, length and head circumference; cerebral palsy; speech; deafness; blindness; child behaviour |
|
| Funding Support | Funding: Foundation for Paediatric Research in Finland, Alma and KA Snellmann Foundation, Sigrid Juselius Foundation, hospital research funds | |
| Notes | Tocolytics were not used and 79% of mothers gave birth < 24 hours after
the intervention. All infants were born < 36 weeks' gestation. Sample‐size calculation: yes. Sample size based on a 25% increase in the primary outcome rate, from an estimated 50% in the control group to 62.5% in the repeat group. The planned sample size was 220 women in each arm (2‐tailed alpha 0.05, beta 0.2). Recruitment was terminated early, after 249 women had been enrolled, primarily because of safety concerns due a decrease in intact survival in the repeat corticosteroid group. In addition, recruitment was slower than expected. Declarations of interest: declared no conflict of interest. Ethical approval: approved by the ethics committee of Oulu University Hospital and the National Agency for Medicines. Trial registration: retrospective. |
|
TEAMS 1999.
| Study characteristics | ||
| Methods | Type of study: parallel, randomised, placebo‐controlled trial | |
| Participants | Location: hospitals in the UK Dates of study: January 2000 to April 2003 Inclusion criteria: women who had received 1 course of prenatal corticosteroids to improve foetal maturity and with clinical uncertainty that a second course of steroids is indicated, and gestational age < 32 weeks Gestational age: < 32 weeks' gestation Exclusion criteria: maternal long‐term systemic corticosteroid therapy (not including inhaled or topical therapy) Total recruited: 162 women (82 in repeat prenatal corticosteroid group and 80 in placebo group). 188 infants (94 in each group) |
|
| Interventions | Repeat prenatal corticosteroid group: 2 doses betamethasone 12 mg, 12 or 24
hours apart, usually repeated every 7 days but could be 10–14 days depending
on unit's protocol Placebo group: placebo administered to match corticosteroid group |
|
| Outcomes | Primary outcomes: neonatal death; neurodevelopmental delay at age 2 years
(corrected for gestational age at birth) Secondary outcomes: short‐term outcomes: stillbirth, death at any time before discharge from neonatal unit, diagnosis of RDS, pneumothorax or other pulmonary air leak, IVH confirmed by ultrasound, diagnosis of necrotising enterocolitis, chronic lung disease (oxygen dependency at 28 days of life), neonatal sepsis, birthweight, maternal sepsis. Long‐term outcomes: growth delay at age 2 years (corrected), respiratory symptoms at age 2 years (corrected), subscale scores for the Vineland Adaptive Behaviour Scales and Bayley II Scales at age 2 years (corrected), readmission to hospital Measures of health service utilisation: admission to, and duration of stay in, a neonatal intensive care unit, use of, and length of time on, mechanical ventilation; use of surfactant, postnatal corticosteroids, high‐frequency oscillation, nitric oxide and extracorporeal membrane oxygenation |
|
| Funding Support | Action Medical Research (UK) | |
| Notes | Declarations of interest: no declaration statement. Ethical approval: protocol was approved by the Multicentre Research Ethics Committee (ref: 98/5/70). Trial registration: during recruitment (ISRCTN46614711). |
|
Wapner 2006.
| Study characteristics | ||
| Methods | Type of study: parallel, randomised placebo‐controlled trial | |
| Participants | Location: 18 US hospitals (NICHD MFMU network centres) Dates of study: March 2000 to April 2003 Eligibility criteria: pregnant women with intact membranes between 23 weeks 0 days and 31 weeks and 6 days if they had received a single full course of betamethasone or dexamethasone between 7 and 10 days earlier and were at high risk for spontaneous preterm birth, or had the diagnosis of placenta praevia or chronic abruption Exclusions: preterm prelabour rupture of the membranes prior to randomisation, confirmed fetal lung maturity, chorioamnionitis, major fetal anomaly, non‐reassuring fetal status, systemic corticosteroid use during the current pregnancy, or insulin‐dependent diabetes. Gestational age determined from the last menstrual period provided that ultrasonography confirmed the estimate. When there was discordance, the duration of gestation at randomisation was determined from the first sonogram performed Gestational age range: 23 weeks 0 days to 31 weeks 6 days' gestation Total recruited: 495 women (planned for 2400), 594 fetuses/infants; 252 women (299 fetuses) in repeat corticosteroid group and 243 women (295 fetuses) in placebo The pregnancy was used as the unit of analysis and for infant outcomes from multiple pregnancies the outcome was considered to have occurred if either infant had the outcome of interest. For outcomes with a severity grading, the most severe of the infant outcomes contributed to the result |
|
| Interventions | Repeat corticosteroid group: each course consisted of 2 injections of
betamethasone 12 mg (as betamethasone sodium phosphate 6 mg and
betamethasone acetate 6 mg) repeated once in 24 hours Placebo group: 'matching placebo' – no other details of preparation given Initially women received courses until birth or 33 weeks 6 days' gestation, whichever was sooner. After 67 women had been enrolled, the number of courses (not including the qualifying course) was limited to 4 because of difficulty in recruitment and published literature suggesting possible harmful effects of multiple courses. 63.4% of women received ≥ 4 study courses |
|
| Outcomes | To time of primary hospital discharge: primary outcome: composite endpoint
of 1 of the following: severe RDS (defined as clinical features of RDS with
the need for oxygen and respiratory support for 6–24 hours or more of age,
an abnormal chest radiograph, and either administration of a full course of
surfactant or a fraction of inspired oxygen (FiO2 of ≥ 60%);
grade 3 or 3 IVH; PVL; chronic lung disease (defined as the need for
supplemental oxygen at 36 weeks' corrected age in infants born before 34
weeks' gestation); or stillbirth or neonatal death. Secondary outcomes not
stated in the paper For the early childhood follow‐up at 2 years' corrected age: the prespecified developmental outcome was the Bayley Mental Developmental Index Score. Other outcomes included Bayley Psychomotor Developmental Index Score; measurements of weight, height and head circumference; and the occurrence of cerebral palsy |
|
| Funding Support | Funding: National Institute of Child Health and Human Development | |
| Notes | Sample‐size calculation: yes. Planned sample size was 2400 women. Quote: "A
primary outcome rate of 11.5% was anticipated for patients assigned to
placebo. Detection of a 30% reduction for patients assigned to repeat
corticosteroids required a sample of 1200 patients in each group" (80% power
and type 1 error rate of 5% (2‐sided)). Recruitment was stopped early based on safety concerns (because of a tendency towards decreased birthweight in the repeat corticosteroid group without any reduction in the primary morbidity outcome and also because of difficulties in recruitment). Declarations of interest: author Dr Mercer declared receiving consulting fees from Columbia Laboratories. No other reported conflict of interest. Ethical approval: institutional board review approved at all participating centres. Trial registration: during recruitment. |
|
ACTH: adrenocorticotropic hormone; BMI: body mass index; BPD: bronchopulmonary dysplasia; BSID‐II: Bayley Scales of Infant Development II; FiO2: fraction of inspired oxygen; IQ: intelligence quotient; IVH: intraventricular haemorrhage; MFMU: Maternal Fetal Medicine Units; NICHD: National Institute of Child Health and Human Development; PaCO2: partial pressure of carbon dioxide; PaO2: partial pressure of oxygen; PVL: periventricular leukomalacia; RDS: respiratory distress syndrome; SD: standard deviation.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Bontis 2011 | 'Non‐randomised' trial. |
| CTRI/2017/04/008326 | Compared a single course of prenatal corticosteroid to placebo without prior corticosteroid. |
| CTRI/2017/05/008721 | Compared a single course of prenatal corticosteroid to placebo without prior corticosteroid. |
| Ernawati 2016 | Study of use of methylprednisolone in the management of pre‐eclampsia. |
| EUCTR2009‐010759‐29‐BE | Trial of prenatal corticosteroid compared with placebo in women with twin pregnancies without prior prenatal corticosteroid exposure. |
| Gyamfi‐Bannerman 2016 | Secondary analysis of prenatal corticosteroids compared with placebo for preterm birth in women who had not already received corticosteroid. |
| IRCT2014090912789N6 | Study comparing different doses of a single course of prenatal corticosteroid for women at risk of preterm birth. |
| IRCT2015120415634N2 | Compared different doses for a single course of prenatal corticosteroid for women at risk of preterm birth. |
| IRCT20191202045571N1 | Compared a single course of prenatal corticosteroid with placebo for women at risk of late preterm birth. |
| Kashanian 2018 | Compared different doses for a single course of prenatal corticosteroid for women at risk preterm birth. |
| Mercer 2001 | Women recruited to the trial did not have corticosteroids before entry. Objective of trial was to evaluate the need for and benefits of weekly prenatal corticosteroids in women at risk of preterm birth. 189 women between 23 and 32 weeks' gestation at risk of preterm birth were randomised to weekly prenatal corticosteroids or a control group where corticosteroids were given if indicated before 35 weeks, if the pregnancy was expected to last > 1 week. Primary outcome: prenatal corticosteroids given within 7 days of preterm birth (< 35 weeks) (optimal exposure). In control group, only 1/3 of infants < 35 weeks' gestation received optimal prenatal corticosteroid exposure. Weekly corticosteroids doubled optimal exposure although the most gave birth at > 34 weeks. |
| NCT03446937 | Trial of prenatal corticosteroid compared with placebo for late preterm birth, without prior corticosteroid exposure. |
| Romejko‐Wolniewicz 2013 | Head‐to‐head trial of 2 different prenatal corticosteroid regimens without prior prenatal corticosteroid exposure. |
| Schmitz 2019 | Trial of different prenatal corticosteroid regimens in women with twin pregnancies without prior prenatal corticosteroid exposure. |
| Sohrabvand 2001 | Women had not already received a single course of corticosteroid (randomisation was to 1 or > 1 course of prenatal corticosteroid). |
| Thorp 2000 | Women recruited to the trial were not randomised to receive repeat corticosteroids but prenatal phenobarbital. Abstract is a secondary multivariate analysis of this trial assessing if duration of prenatal betamethasone is associated with perinatal outcome. |
Characteristics of studies awaiting classification [ordered by study ID]
Atarod 2014.
| Methods | Parallel, randomised placebo‐controlled trial |
| Participants | Women at risk of preterm birth between 28 and 35 weeks' gestation Exclusion criteria: PROM prior to entering the study, major fetal anomalies, chorioamnionitis, systemic corticosteroid use during pregnancy, insulin‐dependent diabetes mellitus and IUGR. Total randomised 1348 women (674 to repeat prenatal corticosteroid group and 674 to placebo group). |
| Interventions | Repeat prenatal corticosteroid group: a treatment course including an
intramuscular injection of betamethasone 12 mg repeated after 24 hours,
followed by 2 other treatment doses every 10 days Placebo group: a treatment course including an intramuscular injection of betamethasone 12 mg repeated after 24 hours, followed by 2 doses of placebo every 10 days |
| Outcomes | RDS, need for oxygen, surfactant, need for ventilation, duration of hospital stay, mortality to hospital discharge, birthweight, height, head circumference |
| Notes | It is unclear as to the time point at which randomisation to repeat
betamethasone or no repeat occurred. The authors had been contacted for this
information in February 2015 and no response was received. This study was moved to awaiting classification after trustworthiness screening due to concerns about randomisation processes as identical numbers were randomised to each group while using simple randomisation for 1348 participants. There was also no explanation provided for exclusion of 104 women after randomisation. The corresponding author was contacted in July and August 2021 and a response was not received. |
IUGR: intrauterine growth restriction; PROM: Premature rupture of membranes; RDS: respiratory distress syndrome.
Characteristics of ongoing studies [ordered by study ID]
NCT02469519.
| Study name | Impact of a booster course of antenatal steroids on neonatal outcome in patients with premature rupture of the membranes (ACSinPROM) |
| Methods | Study type: parallel randomised, placebo‐controlled trial |
| Participants | Setting: 10 study sites in the US hospitals Inclusion criteria: all of: aged ≥ 18 years, 24 weeks and 0 days to 32 weeks and 6 days' gestation, singleton pregnancy, received first course of prenatal corticosteroids at or prior to 31 weeks 6 days' gestation, began first course of prenatal corticosteroids ≥ 7 days prior to randomisation, expectant management planned, PROM before onset of labour Exclusion criteria: any of: known major fetal anomalies, multiple gestation, not a candidate for expectant management, clinical chorioamnionitis (≥ 2 of: temperature > 38.0 °C; uterine tenderness; foul‐smelling vaginal discharge or amniotic fluid; maternal tachycardia > 100 beats per minute; fetal tachycardia > 160 beats per minute; maternal white blood cell count > 20 × 109/L; C‐reactive protein > 5.9 mg/L), already receiving corticosteroids for another condition, any contraindications to the maternal use of corticosteroids |
| Interventions | Experimental group: booster course of prenatal corticosteroids consisting
of betamethasone 12 mg intramuscular injection, 24 hours apart for 2 doses,
or if unavailable may give dexamethasone 6 mg intramuscularly 12 hours apart
for 4 doses Placebo: normal saline of equivalent volume given intramuscularly at the equivalent dosing regimens listed in experimental group |
| Outcomes | Primary outcomes: composite neonatal morbidity from birth through the first
28 days of life; composite neonatal morbidity includes ≥ 1 of: RDS,
bronchopulmonary dysplasia, severe IVH, periventricular leukomalacia, proven
sepsis, necrotising enterocolitis or neonatal death Secondary outcomes: gestational age of baby, baby's birth weight, IUGR, baby's head circumference within the first 24 hours following birth, mechanical ventilatory days, newborn oxygen support days, newborn surfactant therapy, newborn hospital days, pneumothorax, maternal infectious morbidity, time from first dose of study drug to birth |
| Starting date | 3 March 2016 |
| Contact information | As stated on trial registration document: kimberly_maurel@mednax.com |
| Notes | Status: currently recruiting. Confirmed after contact with study contact Kimberly Maurel. |
NCT02939742.
| Study name | Does a rescue course of betamethasone in pregnant women with PPROM decrease neonatal morbidity? |
| Methods | Parallel, randomised, placebo‐controlled trial |
| Participants | Setting: John Sealy Hospital in the US Inclusion criteria (all): maternal age ≥ 18 years, preterm prelabour rupture of membranes (demonstrated clinically by speculum examination), cervical dilation visually ≤ 5 cm on sterile speculum examination, planned delivery at John Sealy Hospital, gestational age of membrane rupture and initiation of first course of prenatal corticosteroids between 23 weeks 5 days to 32 weeks 5 days, planned pregnancy continuation with no indication for delivery for ≥ 7 days Exclusion criteria (any): maternal age > 50 years, gestational age < 23 weeks 5 days or > 32 weeks 5 days, known major congenital abnormalities, aneuploidy, or genetic syndrome, intrauterine fetal demise, any indication for expedited delivery, maternal chorioamnionitis, known allergy or adverse reaction to corticosteroids |
| Interventions | Experimental group: a second course of 2 betamethasone 12 mg intramuscular
injections given 24 hours apart Placebo group: intramuscular saline placebo, given as 2 injections 24 hours apart |
| Outcomes | Primary outcome: length of stay in the neonatal intensive care unit Secondary outcomes: composite neonatal morbidity; defined as ≥ 1 of the following: RDS (oxygen requirement, clinical diagnosis, and consistent chest radiograph), bronchopulmonary dysplasia (requirement for oxygen support at 30 days of life), severe IVH (grades III or IV), periventricular leukomalacia, blood culture‐proven sepsis, necrotising enterocolitis or perinatal death (stillbirth or death before neonatal hospital discharge), duration of oxygen and ventilatory support, development of RDS, grade III or IV IVH, neonatal sepsis, necrotising enterocolitis stage 2 or 3, perinatal death Other outcomes: labour latency, infectious morbidities including chorioamnionitis |
| Starting date | November 2016 |
| Contact information | As per trial registration: Antonio Saad, MD; 409‐772‐1571; afsaad@utmb.edu Sara O Jacobs, MD; 409‐772‐1571; sojacobs@utmb.edu |
| Notes | Status: ongoing recruitment |
IUGR: intrauterine growth restriction; IVH: intraventricular haemorrhage; PPROM: preterm prelabour rupture of the membranes; RDS: respiratory distress syndrome.
Differences between protocol and review
Previous review and updates
Amendments made to the protocol for the 2011 update that clarified the longer‐term health outcomes: total deaths added under outcomes for the child and the child as an adult; survival free of any disability and survival free of major disability added under primary outcomes for the child and for the child as an adult. In addition: periventricular haemorrhage changed to intraventricular haemorrhage; small‐for‐gestational age, intraventricular haemorrhage, intraventricular haemorrhage grade 3 or 4 and periventricular leukomalacia moved to secondary outcomes for the infant; "However defined by authors" added to the definitions of the following outcomes: necrotising enterocolitis, patent ductus arteriosus, retinopathy of prematurity and early systemic neonatal infection.
In the 2015 update, the prespecified groups for subgroup analysis were edited to describe specific populations of interest and methods were updated according to current Cochrane Pregnancy and Childbirth Group standard methods text (2015). An additional search of ClinicalTrials.gov, the WHO International Clinical Trials Registry Platform (ICTRP) for ongoing and unpublished studies was performed.
2021 update
We made the following changes for the 2021 update.
Revised the outcome measures
Grouped primary and secondary outcomes into those for the woman, the child as a fetus/neonate/infant, the child in early (two to less than five years of age) and mid‐ to late childhood (five to less than 18 years of age) and the child as an adult (greater than 18 years of age).
Added the following primary outcomes for the fetus/neonate/infant.
Infant death (less than one year of age).
Severe respiratory distress syndrome (however defined by authors).
Severe intraventricular haemorrhage (grade 3 or 4) (moved from secondary outcomes).
Necrotising enterocolitis (however defined by authors) (moved from secondary outcomes).
Added the following secondary outcomes for the fetus/neonate/infant.
Birthweight (moved from primary outcomes).
Late systemic neonatal infection (48 hours or greater after birth or however defined by authors).
Neonatal encephalopathy (however defined by authors).
Pulmonary hypertension (however defined by authors).
Duration of inotropic support.
Measures of renal function (however reported by authors).
Feed tolerance (time to full feed tolerance, number of feed interruptions or however defined by authors).
Removed the following secondary outcomes for the fetus/neonate/infant.
Placental weight.
Skin‐fold thickness at birth, at primary hospital discharge and at infant follow‐up.
Placental weight.
Neonatal blood pressure.
Developmental delay at infant follow‐up (moved to primary outcomes for the child in early childhood (aged two to less than five years)).
"Measures of HPA axis function (however assessed by authors)" has replaced "HPA axis suppression". "The use of respiratory support (mechanical ventilation or continuous positive airways pressure (CPAP), or both)" and "duration of respiratory support" have been clarified as "Use of respiratory support (any respiratory support including invasive (via an endotracheal tube) or non‐invasive modalities or however defined by authors)". Use and duration of "mechanical ventilation" have been replaced with use and duration of "use of invasive respiratory support (any respiratory support via an endotracheal tube or however defined by authors)". Use and duration of "CPAP" have been replaced with "Duration of non‐invasive respiratory support (any form of respiratory support that does not require an endotracheal tube including any non‐invasive ventilation, CPAP and high or low flow gases or however defined by authors)".
Added the following primary outcomes for the woman.
Maternal sepsis (any of chorioamnionitis during labour, endometritis, pyrexia after trial entry requiring the use of antibiotics, puerperal sepsis, intrapartum fever requiring the use of antibiotics, postnatal pyrexia or however defined by authors).
"Adverse effects of corticosteroid" has been clarified with the following text "including gastrointestinal upset, insomnia, local injection site adverse effects (pain, bruising, haematoma or infection at the injection site)".
Removed the following primary outcomes for the woman.
Puerperal sepsis (however defined by authors) (moved to secondary outcomes).
Chorioamnionitis during labour (however defined by authors) (moved to secondary outcomes).
Added the following secondary outcomes for the woman.
Puerperal sepsis (however defined by authors) (moved to secondary outcomes).
Chorioamnionitis during labour (however defined by authors) (moved to secondary outcomes).
Endometritis (however defined by authors).
Removed the following secondary outcomes for the woman.
Length of labour.
Parenting stress.
Adverse drug reaction.
Added the following secondary outcomes for the child in early childhood (two to less than five years of age).
Obesity/overweight.
Hypertension (however defined by authors).
Chronic lung disease of infancy (however defined by authors).
Asthma or recurrent wheeze (however defined by authors).
Measure of insulin and glucose homeostasis (however defined by authors).
Measures of lipid profile (however defined by authors).
Body composition (lean body mass corrected for height, fat mass corrected for height or however defined by authors).
Removed the following secondary outcomes for the child in early childhood (two to less than five years of age).
Skin fold thickness at childhood follow‐up.
Learning difficulties.
Dyslipidaemia.
Composite serious outcome.
Insulin sensitivity.
Developmental outcomes have been revised. "Survival free of any disability" has been replaced by "neurodevelopmental impairment at age two to less than five years", "survival free of major disability" by "survival free of neurodevelopmental impairment at age two to less than five years" and "disability at childhood follow‐up" by and "survival free of major neurodevelopmental impairment at age two to less than years". "Developmental delay" and "intellectual impairment" have been replaced by "developmental delay or intellectual impairment at age two to less than five years".
The following primary outcomes have been added for the child in mid‐ to late childhood (five to less than 18 years).
Total deaths (after randomisation).
Neurocognitive impairment at age five to less than 18 years (any of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), executive dysfunction, learning difficulties, motor impairment (mild or major impairment by appropriate mode of assessment), or cerebral palsy, or however defined by authors).
Survival free of neurocognitive impairment at age five to less than 18 (none of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), executive dysfunction, learning difficulties, motor impairment (mild or major impairment by appropriate mode of assessment) cerebral palsy, or however defined by authors).
Survival free of major neurocognitive impairment at age five to less than 18 years (none of: moderate or severe intellectual impairment (developmental quotient or intelligence quotient more than two SD below the mean), major motor impairment, moderate or severe cerebral palsy, blindness (corrected visual acuity worse than 6/60 in the better eye) or deafness (hearing loss requiring amplification or worse), or however defined by authors).
Motor impairment at age five to less than 18 years (categorised as nil, mild impairment, major impairment by appropriate mode of assessment or however defined by authors).
Cognitive impairment at age five to less than 18 years (categorised as mild (one SD below the mean), moderate (two SDs below the mean) or severe (three SDs below the mean) by an appropriate rating scale, or however defined by authors).
Educational achievement (however defined by authors).
Cerebral palsy (categorised as nil, mild, moderate or severe using an appropriate scale or however defined by authors).
Hypertension (however defined by authors).
Added the following secondary outcomes for the child in mid‐ to late childhood (five to less than 18 years).
Child behaviour (however defined by authors).
Deafness/hearing impairment (however defined by authors).
Blindness/visual impairment (however defined by authors).
Growth assessments (weight, head circumference, height).
BMI.
Obesity/overweight.
Blood pressure (systolic, diastolic, mean arterial).
Measures of lung function (however defined by authors).
Asthma or recurrent wheeze (however defined by authors).
Respiratory disease (however defined by authors).
Measure of insulin and glucose homeostasis (however defined by authors).
Lipid profile (however reported by authors).
Measures of HPA axis function (however defined by authors).
Bone density (however assessed by authors).
Body composition (lean body mass (fat free mass) for height, fat mass for height or however defined by authors).
Added the following primary outcomes for the child as an adult (greater than 18 years).
Neurodevelopmental disability at age 18 years or greater (any of: visual impairment, hearing impairment, intellectual impairment (developmental quotient or intelligence quotient more than one SD below the mean), cerebral palsy, or however defined by authors).
Cardiovascular disease (including diagnosis of ischaemic heart disease, cerebrovascular disease, heart failure or however defined by authors).
Cardiovascular death (ischaemic heart disease, stroke, arrhythmia or heart failure as cause of death or however defined by authors).
Type 2 diabetes mellitus (however defined by authors).
Glucose intolerance (however defined by authors).
Obesity/overweight (however defined by authors).
Hypertension (however defined by authors).
Removed the following primary outcomes for the child as an adult (greater than 18 years).
Survival free of any disability (however defined by authors).
Survival free of major disability (however defined by authors).
Disability at adult follow‐up (developmental delay or intellectual impairment, blindness, deafness, or cerebral palsy, however defined by authors).
Major sensorineural disability (defined as any of legal blindness, sensorineural deafness requiring hearing aids, moderate or severe cerebral palsy, or developmental delay or intellectual impairment (defined as developmental quotient or intelligence quotient more than two SDs below mean)).
Composite serious outcome (however defined by authors).
Added the following secondary outcomes for the child as an adult (aged 18 years or greater).
Educational achievement (however defined by authors).
Prediabetes (however defined by authors).
Mental health disorders (however defined by authors).
Diagnosis of depression (however defined by authors).
Diagnosis of bipolar affective disorder (however defined by authors).
Diagnosis of anxiety disorder (however defined by authors).
Ischaemic heart disease (however defined by authors).
Stroke (however defined by authors).
Heart failure.
Blindness/visual impairment (however defined by authors).
Deafness/hearing impairment (however defined by authors).
Measure of insulin and glucose homeostasis (however defined by authors).
Removed the following secondary outcomes for the child as an adult (aged 18 years or greater).
Insulin sensitivity.
Skin‐fold thickness in later life.
Blindness.
Deafness.
Insulin sensitivity.
Assessment of all eligible studies
Incorporated an assessment of scientific integrity (trustworthiness) as described in Selection of studies.
Risk of bias
Used the RoB 2 tool to assess risk of bias at the outcome level and replaced study level assessments as described in Assessment of risk of bias in included studies.
Sensitivity analyses
Modified planned sensitivity analyses to reflect changes in the risk of bias assessment and now state "Where overall risk of bias was assessed as 'high risk' or 'some concerns' for a study outcome, we explored this by sensitivity analysis excluding these studies".
Clinical subgroups for subgroup analysis
Removed "The number of repeat courses of corticosteroids actually given" from the list of clinical subgroups for subgroup analysis because it is a postrandomisation categorisation.
Unit of analysis
Included a comment on the unit of analysis. Changed the denominator for neonatal outcomes from live births to fetuses alive at randomisation.
Summary of findings tables
Included GRADE summary of findings tables to summarise the key outcomes for each group (for the fetus/neonate/infant, the woman, the child in early childhood (aged two to less than five years), the child in mid‐ to late childhood (aged five to less than 18 years) and the child as an adult (aged 18 years or greater).
Contributions of authors
For this 2021 updated, all authors contributed to updating the protocol.
AW, CM, JH, CAC assessed identified studies for eligibility.
AW and one additional author (CAC, JEH, CM or PM) performed data extraction, trustworthiness assessments and risk of bias assessments for each trial.
AW prepared the risk of bias tables from these assessments.
AW prepared the first draft.
All authors commented on subsequent drafts and approved the final version.
Previous versions and protocol
CAC and JEH prepared the original protocol published in 2000.
CAC wrote the draft of the original review, and both CAC and JEH commented on subsequent drafts and prepared the previous updates.
For the 2011 and 2015 updates, CAC prepared the first draft; CAC, CM and PM prepared the risk of bias tables, CAC and CM assessed identified studies for eligibility and CAC, CM and PM extracted data for the included trials.
All authors commented on subsequent drafts and approved the final version.
Sources of support
Internal sources
-
South Australian Health and Medical Research Institute, Australia
Salary support for PM
-
Liggins Institute, The University of Auckland, New Zealand
Salary support for CC, JH, CMcK
External sources
-
Aotearoa Foundation, USA
Scholarship support for AW
-
National Institute for Health Research, UK
NIHR Programme of centrally‐managed pregnancy and childbirth systematic reviews of priority to the NHS and users of the NHS:10/4001/02 (2015 update)
-
World Health Organization (WHO) and the UNDP‐UNFPA‐UNICEF‐WHO‐World Bank Special Programme of Research, Development and Research Training in Human Reproduction (HRP), Switzerland
This review is supported by funding to Cochrane Pregnancy and Childbirth (University of Liverpool) (2021 update)
Declarations of interest
AW: reports working as a health professional but in an unrelated area (as an Endocrinology advanced trainee).
CM: reports previously publishing on repeat prenatal corticosteroids, being an investigator in the six‐ to eight‐year follow‐up studies of the Australasian Collaborative Trial of Repeat Doses of Corticosteroid for the Prevention of Neonatal Respiratory Disease (Crowther 2006), and working as a health professional for Counties Manukau Health.
PM: reports being an Editor for Cochrane Pregnancy and Childbirth, but was not involved in the editorial process for this review.
JH: reports giving multiple lectures and publishing review articles which relate to the material included in this review, none directly reporting the contents of the review. Investigator in the two‐year and six‐ to eight‐year follow‐up studies of the Australasian Collaborative Trial of Repeat Doses of Corticosteroid for the Prevention of Neonatal Respiratory Disease (Crowther 2006). Funding for the ACTORDS follow‐up studies was updated received from: the National Health and Medical Research Council of Australia; Channel 7 Research Foundation, South Australia; the Women's and Children's Research Foundation, Adelaide, South Australia; Discipline of Obstetrics and Gynaecology, University of Adelaide, South Australia; Health Research Council of New Zealand; Auckland Medical Research Foundation, New Zealand. None of the funding bodies had any role in the conduct of the study, analysis or decision to publish.
CAC: reports being the lead investigator for the Australasian Collaborative Trial of Repeat Doses of Corticosteroid for the Prevention of Neonatal Respiratory Disease and subsequent follow‐up studies (Crowther 2006).
New search for studies and content updated (conclusions changed)
References
References to studies included in this review
Aghajafari 2002 {published data only}
- Aghajafari F, Murphy K, Ohlsson A, Amankwah K, Matthews S, Hannah M.Multiple versus single courses of antenatal corticosteroids for preterm birth: a pilot study. Journal of Obstetrics and Gynaecology Canada: JOGC 2002;24(4):321-9. [DOI] [PubMed] [Google Scholar]
Crowther 2006 {published data only}
- ACTRN12606000318583.Neonatal respiratory distress syndrome after repeat exposure to antenatal corticosteroids: a randomised controlled trial [A multicentred randomised controlled trial of repeat doses of prenatal corticosteroid given to women who remain at risk of preterm delivery for the prevention of neonatal morbidity]. www.who.int/trialsearch/Trial2.aspx?TrialID=ACTRN12606000318583 (first received 5 March 2001). [CENTRAL: CN-01861799]
- Ashwood PJ, Crowther CA, Willson KJ, Haslam RR, Kennaway DK, Hiller JE, et al.Neonatal adrenal function after repeat dose prenatal corticosteroids: a randomized controlled trial. American Journal of Obstetrics and Gynecology 2006;194:861-7. [DOI] [PubMed] [Google Scholar]
- Battin M, Bevan C, Harding J.Growth in the neonatal period after repeat courses of antenatal corticosteroids: data from the ACTORDS randomised trial. Archives of Disease in Childhood Fetal & Neonatal Edition 2012;97(2):F99-105. [DOI] [PubMed] [Google Scholar]
- Battin M, Bevan C, Harding J.Repeat doses of antenatal steroids and hypothalamic-pituitary-adrenal axis (HPA) function. American Journal of Obstetrics and Gynecology 2007;197:40.e1-40.e6. [DOI] [PubMed] [Google Scholar]
- Battin MR, Bevan C, Morton SM, Harding JE.Repeat courses of antenatal corticosteroids do not alter hypothalamic-pituitary-adrenal axis function after birth; results of a randomised controlled trial. In: Pediatric Academic Societies Annual Meeting; 2004 May 1-4; San Francisco, USA. 2004.
- Cartwright R, Crowther C, Anderson P, Harding J, Doyle L, McKinlay C.Influence of fetal growth restriction on neurocognitive function after repeat antenatal betamethasone: secondary analysis of a randomised trial. Journal of Paediatrics and Child Health 2019;55(Suppl 1):12-3. [CENTRAL: CN-01937295] [EMBASE: 627192578] [Google Scholar]
- Cartwright RD, Crowther CA, Anderson PJ, Harding JE, Doyle LW, McKinlay CJ.Association of fetal growth restriction with neurocognitive function after repeated antenatal betamethasone treatment vs placebo: secondary analysis of the ACTORDS randomized clinical trial. JAMA Network Open 2019;2(2):e187636. [CENTRAL: CN-01956584] [EMBASE: 627932277] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cartwright RD, Harding JE, Crowther CA, Cutfield WS, Battin MR, Dalziel SR, et al.Cardiometabolic function after repeat antenatal betamethasone: influence of fetal growth restriction. Journal of Paediatrics and Child Health 2018;54(Suppl 1):11-2. [CENTRAL: CN-01657057] [EMBASE: 621532745] [Google Scholar]
- Cartwright RD, Harding JE, Crowther CA, Cutfield WS, Battin MR, Dalziel SR, et al.Repeat antenatal betamethasone and cardiometabolic outcomes. Pediatrics 2018;142(1):e20180522. [CENTRAL: CN-01930328] [PMID: ] [DOI] [PubMed] [Google Scholar]
- Crosby DD, Ashwood PJ, Khong TY, Crowther CA.Effects of repeat dose prenatal corticosteroids on placental pathology: results from the ACTORDS trial. Journal of Paediatrics and Child Health 2011;47(Suppl 1):53. [Google Scholar]
- Crowther C, Doyle LW, Anderson P, Harding JE, Haslam RR, Hiller JE.Repeat dose(s) of prenatal corticosteroids for women at risk of preterm birth: early school-age outcomes (6 to 8 years) for the ACTORDS trial. In: Pediatric Academic Societies and Asian Society for Pediatric Research Joint Meeting; 2011 April 30-May 3; Denver, CO. 2011:3123.6.
- Crowther CA, Anderson PJ, McKinlay CJ, Harding JE, Ashwood PJ, Haslam RR, et al.Mid-childhood outcomes of repeat antenatal corticosteroids: a randomized controlled trial. Pediatrics 2016;138(4):e20160947. [CENTRAL: CN-01382393] [PMID: ] [DOI] [PubMed] [Google Scholar]
- Crowther CA, Doyle LW, Anderson P, Harding JE, Haslam RR, Hiller JE, et al.Repeat dose(s) of prenatal corticosteroids for women at risk of preterm birth: early school-age outcomes (6 to 8 years') for children in the ACTORDS trial. Journal of Paediatrics and Child Health 2011;47(Suppl 1):52-3. [Google Scholar]
- Crowther CA, Doyle LW, Haslam RR, Hiller JE, Harding JE, Robinson JS, et al.Outcome at 2 years of age after repeat doses of antenatal corticosteroids. New England Journal of Medicine 2007;357:1179-89. [DOI] [PubMed] [Google Scholar]
- Crowther CA, Haslam RR, Doyle LW, Harding JE, Hiller JE, Robinson JS, for the ACTORDS Study Group.Repeat doses of prenatal corticosteroids for women at risk of preterm birth: follow-up of children at 2 years corrected age in the ACTORDS trial. Journal of Paediatrics and Child Health 2007;43(Suppl 1):A76. [Google Scholar]
- Crowther CA, Haslam RR, Hiller JE, Doyle LW, Robinson JS, for the Australasian Collaborative Trial of Repeat Doses of Steroids (ACTORDS) Study Group.Neonatal respiratory distress syndrome after repeat exposure to antenatal corticosteroids: a randomised controlled trial. Lancet 2006;367:1913-9. [DOI] [PubMed] [Google Scholar]
- Crowther CA, Hiller JE, Haslam RR, Doyle LW, Robinson JS.Repeat doses of prenatal corticosteroids for women at risk of preterm birth: the ACTORDS Trial 12 month follow up. In: Perinatal Society of Australia and New Zealand 10th Annual Congress; 2006 April 3-6; Perth, Australia. 2006:179.
- McKinlay CJ, Cutfield WS, Battin MR, Dalziel SR, Crowther CA, Harding JE.Cardiovascular risk factors after exposure to repeat antenatal betamethasone: early school-age follow-up of a randomised trial (ACTORDS). Pediatric Academic Societies and Asian Society for Pediatric Research Joint Meeting; 2011 Apr 30-May 3; Denver (CO);1660.5.
- McKinlay CJ, Cutfield WS, Battin MR, Dalziel SR, Crowther CA, Harding JE.Cardiovascular risk factors in children after repeat doses of antenatal glucocorticoids: an RCT. Pediatrics 2015;135(2):e405-15. [DOI] [PubMed] [Google Scholar]
- McKinlay CJ, Cutfield WS, Battin MR, Dalziel SR, Crowther CA, Harding JE.Mid-childhood bone mass after exposure to repeat doses of antenatal glucocorticoids: a randomized trial. Pediatrics 2017;139(5):e20164250. [DOI] [PubMed] [Google Scholar]
- McKinlay CJ, Cutfield WS, Battin MR, Dalziel SR, Crowther CA, Harding JE.Repeat antenatal betamethasone does not affect basal salivary cortisol at early school-age: a randomised controlled trial (ACTORDS). Pediatric Academic Societies and Asian Society for Pediatric Research Joint Meeting; 2011 Apr 30-May 3; Denver (CO) 2011:2919.266. [Google Scholar]
- McKinlay CJ, Cutfield WS, Battin MR, Dalziel SR, Crowther CA, Harding JE.Repeat antenatal betamethasone does not affect bone mass at early school-age: a randomised controlled trial (ACTORDS). Journal of Paediatrics and Child Health 2013;49(Suppl 2):49. [Google Scholar]
- McKinlay CJ, Cutfield WS, Battin MR, Dalziel SR, Crowther CA.Cardiovascular risk factors after exposure to repeat antenatal betamethasone: early school-age follow-up of a randomised trial (ACTORDS). Journal of Paediatrics and Child Health 2011;47(Suppl 1):20. [Google Scholar]
- McKinlay CJ, Cutfield WS, Battin MR, Dalziel SR, Crowther CA.Repeat antenatal betamethasone does not affect basal salivary cortisol at early school-age: A randomised controlled trial (ACTORDS). Journal of Paediatrics and Child Health 2011;47(Suppl 1):19. [Google Scholar]
- McKinlay CJ, Harding JE, Ashwood PJ, Dalziel SR, Doyle LW, Haslam RR, et al.Effect of repeat antenatal betamethasone on childhood lung function: A randomised controlled trial (ACTORDS). Journal of Paediatrics and Child Health 2013;49(Suppl 2):92-3. [Google Scholar]
- Mildenhall L, Battin M, Bevan C, Kuschel C, Harding J.Repeat prenatal corticosteroid doses do not alter neonatal blood pressure or myocardial thickness: randomized, controlled trial. Pediatrics 2009;123(4):e646-52. [DOI] [PubMed] [Google Scholar]
- Mildenhall L, Battin M, Morton S, Bevan C, Kuschel C, Harding J.Exposure to repeat doses of antenatal glucocorticoids is associated with altered cardiovascular status after birth. Archives of Disease in Childhood. Fetal and Neonatal Edition 2006;91:F56-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mildenhall LF, Battin MR, Bevan C, Kuschel CA, Harding JE.Repeat doses of antenatal corticosteroids do not alter neonatal cardiovascular status after birth: a randomised controlled trial. In: Perinatal Society of Australia and New Zealand 10th Annual Congress; 2006 April 3-6; Perth, Australia. 2006:55.
Garite 2009 {published data only}
- Garite T, Kurtzman J, Maurel K, Clark R, for the Obstetrix Collaborative Research Network.Impact of a 'rescue course' of antenatal corticosteroids: a multicenter randomized placebo-controlled trial. American Journal of Obstetrics and Gynecology 2009;200:248.e1-9. [DOI] [PubMed] [Google Scholar]
- Kurtzman J, Garite T, Clark R, Maurel K, The Obstetrix Collaborative Research Network.Impact of a "rescue course" of antenatal corticosteroids (ACS): a multicenter randomized placebo controlled trial. American Journal of Obstetrics and Gynecology 2008;199(6 Suppl 1):S2. [DOI] [PubMed] [Google Scholar]
- NCT00201643.A randomized trial comparing the impact of one versus two courses of ACS on neonatal outcome. clinicaltrials.gov/ct2/show/NCT00201643 (first received 20 September 2005).
Guinn 2001 {published data only}
- Guinn D, Atkinson M, Sullivan L, Lee M, MacGregor S, Parilla B, et al.Single versus weekly courses of antenatal corticosteroids for women at risk of preterm delivery: a randomized controlled trial. Obstetrics & Gynecology 2003;101(1):195. [Google Scholar]
- Guinn D, BMZ Study Group.Multicenter randomized trial of single versus weekly courses of antenatal corticosteroids (ACS). American Journal of Obstetrics and Gynecology 2001;184(1):S6. [Google Scholar]
- Guinn DA, Atkinson MW, Sullivan L, Lee M, MacGregor S, Parilla B, et al.Single vs weekly courses of antenatal corticosteroids for women at risk of preterm delivery. JAMA 2001;286(13):1581-7. [DOI] [PubMed] [Google Scholar]
- Guinn DA, BMZ Study Group.Multicenter randomized trial of single versus weekly courses of antenatal corticosteroids (ACS): interim analysis. American Journal of Obstetrics and Gynecology 2000;182(1 Pt 2):S12. [Google Scholar]
- Lee M, Davies J, Atkinson MW, Guinn D, BMZ Study Group.Efficacy of weekly courses of antenatal corticosteroids (ACS) in preterm premature rupture of the membranes. American Journal of Obstetrics and Gynecology 2001;184(1):S8. [DOI] [PubMed] [Google Scholar]
- Lee MJ, Davies J, Guinn D, Sullivan L, Atkinson MW, McGregor S, et al.Single versus weekly courses of antenatal corticosteroids in preterm premature rupture of membranes. Obstetrics & Gynecology 2004;103(2):274-81. [DOI] [PubMed] [Google Scholar]
Mazumder 2008 {published data only}
- Mazumder P, Dutta S, Kaur J, Narang A.Single versus multiple courses of antenatal betamethasone and neonatal outcome: a randomized controlled trial. Indian Pediatrics 2008;45(8):661-7. [PMID: ] [PubMed] [Google Scholar]
McEvoy 2002 {published data only}
- McEvoy C, Bowling S, Williamson K, Lozano D, Tolaymat L, Collins J, et al.Effects of single versus weekly courses of antenatal steroids (AS) on functional residual capacity in preterm infants: a randomized trial. In: Pediatric Academic Societies Annual Meeting; 2001 Apr 28-May 1; Baltimore Convention Centre, Baltimore (MD). 2001:2228.
- McEvoy C, Bowling S, Williamson R, Lozano D, Tolaymat L, Izquierdo L, et al.The effect of a single remote course versus weekly courses of antenatal corticosteroids on functional residual capacity in preterm infants: a randomized trial. Pediatrics 2002;110:280-4. [DOI] [PubMed] [Google Scholar]
- McEvoy C.Effects of single versus weekly courses of antenatal steroids (AS) on functional residual capacity in preterm infants: a randomized trial. Pediatric Research 2001;49(4):388A. [DOI] [PubMed] [Google Scholar]
McEvoy 2010 {published data only}
- Jordan BK, Schilling D, McEvoy CT.Pulmonary function at hospital discharge in preterm infants randomized to a single rescue course of antenatal steroids. Journal of Pediatrics 2017;181:62-66.e1. [CENTRAL: CN-01379228] [EMBASE: 613767651] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan BK, Schilling D, McEvoy CT.The longitudinal impact of rescue antenatal steroids on neonatal respiratory compliance through hospital discharge: follow up of a randomized controlled trial (RCT). In: Pediatric Academic Societies Annual Meeting; 2016 Apr 30-May 3; Baltimore (MD). 2016:3828.264. [CENTRAL: CN-02011933]
- Jordan BK, Schilling D, McEvoy CT.The window of improved neonatal respiratory compliance after rescue antenatal steroids. Journal of Perinatology 2018;38(7):828-33. [CENTRAL: CN-02000541] [EMBASE: 622262146] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEvoy C, Schilling D, Clay N, Spitale P, Durand M.Neurodevelopmental outcome and growth in infants randomized to a single rescue course of antenatal steroids. In: Pediatric Academic Societies and Asian Society for Pediatric Research Joint Meeting; 2011 Apr 30-May 3; Denver (CO). 2011:3829.270.
- McEvoy C, Schilling D, Peters D, Tillotson C, Spitale P, Wallen L, et al.Respiratory compliance in preterm infants after a single rescue course of antenatal steroids: a randomized controlled trial. American Journal of Obstetrics and Gynecology 2010;202:544.e1-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEvoy C, Schilling D, Segel S, Spitale P, Wallen L, Bowling S, et al.Improved respiratory compliance in preterm infants after a single rescue course of antenatal steroids: a randomized trial. American Journal of Obstetrics and Gynecology 2008;199(Suppl 1):S228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEvoy C, Schilling D, Spitale P, Gravett M, Durand M.Pulmonary function and respiratory outcomes at 12-24 months in preterm infants randomized to a single rescue course of antenatal steroids. In: Pediatric Academic Societies' 2010 Annual Meeting; 2010 May 1-4; Vancouver, Canada. 2010.
- McEvoy C, Schilling D, Spitale P, O'Malley J, Bowling S, Durand M.Pulmonary function and outcomes in infants randomized to a rescue course of antenatal steroids. Pediatric Pulmonology 2017;52:1171-8. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEvoy C, Schilling D, Spitale P, Wallen L, Segel S, Bowling S, et al.Improved respiratory compliance after a single rescue course of antenatal steroids: a randomized controlled trial. In: Pediatric Academic Societies Annual Meeting; 2007 May 5-8; Toronto, Canada. 2007.
- McEvoy C, Schilling D, Spitale P, Wallen L, Segel S, Bowling S, et al.Improved respiratory compliance after a single rescue course of antenatal steroids: a randomized controlled trial. In: Pediatric Academic Societies Annual Meeting; 2008 May 2-6; Honolulu, Hawaii. 2008.
- McEvoy C, Schilling D, Spitale P, Wallen P, Segel S, Bowling S, et al.Growth and respiratory outcomes after a single rescue course of antenatal steroids: a randomized trial. In: Pediatric Academic Societies Annual Meeting; 2009 May 2-5; Baltimore (MD). 2009.
- McEvoy CT, Schilling D, Segal S, Spitale P, Wallen L, Bowling S, et al.Improved respiratory compliance in preterm infants < 34 weeks after a single rescue course of antenatal steroids. American Journal of Respiratory and Critical Care Medicine 2009;179:A4127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NCT00669383.Rescue antenatal steroids and pulmonary function tests in preterm infants [Rescue antenatal steroids and lung volumes in preterm infants]. clinicaltrials.gov/ct2/show/NCT00669383 (first received 30 April 2008). [CENTRAL: CN-02042396]
Murphy 2008 {published and unpublished data}
- Asztalos E, Murphy K, Hannah M, Willan A, MACS Collaborative Group.Outcomes of children at two years after multiple courses of antenatal corticosteroids for threatened preterm birth: the Multiple Antenatal Corticosteroids study (MACS). Pediatric Academic Societies' 2010 Annual Meeting; 2010 May 1-4; Vancouver, Canada.
- Asztalos E, Willan A, Murphy K, Matthews S, Ohlsson A, Saigal S, et al.Association between gestational age at birth, antenatal corticosteroids, and outcomes at 5 years: multiple courses of antenatal corticosteroids for preterm birth study at 5 years of age (MACS-5). BMC Pregnancy and Childbirth 2014;14(1):272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asztalos E, Willan A, Murphy K, Matthews S, Ohlsson A, Saigal S, et al.Multiple courses of antenatal corticosteroids for preterm birth study at 5 years of age (MACS-5): association between gestational age at birth, antenatal corticosteroids, and outcomes at 5 years of age (MACS-5). American Journal of Obstetrics and Gynecology 2014;210(1 Suppl):S321-2. [Google Scholar]
- Asztalos EV, Murphy KE, Hannah ME, Willan AR, Matthews SG, Ohlsson A, et al.Multiple courses of antenatal corticosteroids for preterm birth study: 2-year outcomes. Pediatrics 2010;126:e1045-55. [DOI] [PubMed] [Google Scholar]
- Asztalos EV, Murphy KE, Willan AR, Matthews SG, Ohlsson A, Saigal S, et al, for the MACS-5 Collaborative Group.Multiple courses of antenatal corticosteroids for preterm birth study: outcomes in children at 5 years of age (MACS-5). Journal of the American Medical Association Pediatrics 2013;167(12):1102-10. [DOI] [PubMed] [Google Scholar]
- Leung M, Asztalos E, Tomat L.Monitoring discrepancies between primary and follow-up databases. Clinical Trials (London, England) 2010;7(4):481. [CENTRAL: CN-01008729] [EMBASE: 70462866] [Google Scholar]
- Maternal, Infant and Reproductive Health Research Unit.Multiple courses of antenatal steroids for preterm birth study (MACS): study protocol. sunnybrook.ca/uploads/sri_cmicr_macs_protocol_en.pdf (accessed March 2022).
- Murphy K, Asztalos E, Hannah M, Willan A, Hewson S, Ohlsson A, et al.Multiple courses of antenatal corticosteroids for preterm birth study (MACS): results of the three month maternal post partum questionnaire. American Journal of Obstetrics and Gynecology 2009;201(6 Suppl 1):S173. [CENTRAL: CN-01743657] [EMBASE: 70129165] [Google Scholar]
- Murphy K, Asztalos E, Willan A, Ohlsson A, Saigal S, Kelly E, et al.Preterm premature rupture of membranes and adverse neurodevelopmental outcomes at 5 years of age: a multiple courses of antenatal corticosteroids for preterm birth study secondary analysis. American Journal of Obstetrics and Gynecology 2015;212(1 Suppl 1):S250. [Google Scholar]
- Murphy K, for the MACS Collaborative Group.Multiple courses of antenatal corticosteroids for preterm birth study. American Journal of Obstetrics and Gynecology 2007;197(6 Suppl 1):S2. [Google Scholar]
- Murphy K, Hannah M, Willan A, Hewson S, Ohlsson A, Kelly E, et al.Multiple courses of antenatal corticosteroids for preterm birth (MACS): a randomised controlled trial. Lancet 2008;372:2143-51. [DOI] [PubMed] [Google Scholar]
- Murphy K, Willan A, Hannah M, Ohlsson A, Kelly E, Matthews S, et al.Do antenatal corticosteroids reduce fetal growth or gestational age at birth? A secondary analysis from the multiple courses of antenatal corticosteroids for preterm birth study (MACS). American Journal of Obstetrics and Gynecology 2012;206(Suppl 1):S226. [Google Scholar]
- Murphy KE, Hannah ME, Willan AR, Ohlsson A, Kelly EN, Matthews SG, et al.Maternal side-effects after multiple courses of antenatal corticosteroids (MACS): the three-month follow-up of women in the randomized controlled trial of MACS for preterm birth study. Journal of Obstetrics and Gynaecology Canada: JOGC 2011;33(9):909-21. [PubMed] [Google Scholar]
- Murphy KE, Willan AR, Hannah ME, Ohlsson A, Kelly EN, Matthews SG, et al.Effect of antenatal corticosteroids on fetal growth and gestational age at birth. Obstetrics & Gynecology 2012;119(5):917-23. [DOI] [PubMed] [Google Scholar]
- NCT00187382.Multiple Courses of Antenatal Corticosteroids for Preterm Birth Study (MACS). clinicaltrials.gov/show/NCT00187382 (first received 13 September 2005). [CENTRAL: CN-02016501]
- Sanchez J, Mergler S, Mason D, Murphy K, Asztalos E.Developing a final analysis timeline for a large multicentre randomized controlled trial. Clinical Trials (London, England) 2011;8(4):543. [CENTRAL: CN-01006648] [EMBASE: 71191915] [Google Scholar]
- Shea A, Murphy KE, Asztalos E, Willan AR, Sanchez J.Postpartum depression assessment in the multiple courses of antenatal corticosteroids for preterm birth study (MACS). American Journal of Obstetrics and Gynecology 2017;216(1 Suppl 1):S462. [CENTRAL: CN-01475695] [EMBASE: 614089554] [Google Scholar]
Peltoniemi 2007 {published data only}
- Janer C, Helve O, Pitkänen OM, Kari MA, Peltoniemi OM, Hallman M, et al.Expression of airway epithelial sodium channel in the preterm infant is related to respiratory distress syndrome but unaffected by repeat antenatal betamethasone. Neonatology 2010;97(2):132-8. [DOI] [PubMed] [Google Scholar]
- Koivisto M, Peltoniemi OM, Saarela T, Tammela O, Jouppila P, Hallman M.Blood glucose level in preterm infants after antenatal exposure to glucocorticoid. Acta Paediatrica 2007;96(5):664-8. [DOI] [PubMed] [Google Scholar]
- NCT00295464.Antenatal rescue course of glucocorticoids in threatened premature birth [Randomized trial on efficacy and safety of the antenatal rescue course of glucocorticoids in threatened premature birth (ACG Trial)]. clinicaltrials.gov/show/NCT00295464 (first received 21 February 2006).
- Peltoniemi O, Kari M, Lano A, Yliherva A, Puosi R, Lehtonen L, et al.Two-year follow-up of a randomized trial with repeated antenatal betamethasone. Archives of Disease in Childhood. Fetal and Neonatal Edition 2009;94(6):F402-6. [DOI] [PubMed] [Google Scholar]
- Peltoniemi OM, Kari MA, Halmesmaki E, Raudaskoski T, Tammela O, Uotila J, et al.The effect of second course of antenatal betamethasone (BM) on neonatal morbidity in preterm infants: a randomised trial. Pediatric Research 2005;58(2):404. [Google Scholar]
- Peltoniemi OM, Kari MA, Jouppila P, Hallman M.The second rescue dose of antenatal betamethasone (BM) shortly before birth may increase respiratory morbidity in high risk preterm infants. In: Pediatric Academic Societies Annual Meeting; 2006 Apr 29-May 2; San Francisco (CA). 2006.
- Peltoniemi OM, Kari MA, Tammela O, Lehtonen L, Marttila R, Halmesmaki E, et al.Randomized trial of a single repeat dose of prenatal betamethasone treatment in imminent preterm birth. Pediatrics 2007;119(2):290-8. [DOI] [PubMed] [Google Scholar]
- Peltoniemi OM.2-year follow-up of randomized trial on a single repeat dose of antenatal betamethasone in imminent preterm birth. In: Pediatric Academic Societies and Asian Society for Pediatric Research Joint Meeting; 2008 May 2-6; Honolulu, Hawaii. 2008.
- Pesonen A, Raiikonen K, Lano A, Peltoniemi O, Hallman M, Kari MA.Antenatal betamethasone and fetal growth in prematurely born children: implications for temperament traits at the age of 2 years. Pediatrics 2009;123:e31-7. [DOI] [PubMed] [Google Scholar]
TEAMS 1999 {unpublished data only}ISRCTN46614711
- Brocklehurst P, Gates S, Johnson A.Effects of multiple courses of antenatal steroids are uncertain [letter]. BMJ 2000;321:47. [PMC free article] [PubMed] [Google Scholar]
- ISRCTN46614711.Trial of the effects of antenatal multiple courses of steroids versus a single course (TEAMS): pilot study. www.isrctn.com/ISRCTN46614711 (first received 1 March 2001).
Wapner 2006 {published data only}
- Borowski KS, Clark EA, Lai Y, Wapner RJ, Sorokin Y, Peaceman AM, et al.Neonatal genetic variation in steroid metabolism and key respiratory function genes and perinatal outcomes in single and multiple courses of corticosteroids. American Journal of Perinatology 2015;32(12):1126-32. [CENTRAL: CN-01169539] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carroll MA, Vidaeff AC, Mele L, Wapner RJ, Mercer B, Peaceman AM, et al.Bone metabolism in pregnant women exposed to single compared with multiple courses of corticosteroids. Obstetrics & Gynecology 2008;111(6):1352-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Church M.Auditory brainstem responses (ABRs) in neonates exposed to repeated courses of antenatal corticosteroids. In: Program of the Twenty Eighth Annual Midwinter Research Meeting of the Association for Research in Otolaryngology; 2005 Feb 19-24; New Orleans (LA). 2005:669.
- Church MW, Wapner RJ, Mele LM, Johnson F, Dudley DJ, Spong CY, et al.Repeated courses of antenatal corticosteroids: are there effects on the infant's auditory brainstem responses? Neurotoxicology and Teratology 2010;32(6):605-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dude C, Dude A, Gilner J, Swamy G, Grotegut C.Predicting preterm delivery: using the MFMU BEARS trial data to optimize corticosteroid use in women at risk for preterm delivery. Reproductive Sciences (Thousand Oaks, Calif.) 2016;23(Suppl 1):188A-9A. [CENTRAL: CN-02011932] [Google Scholar]
- Fonseca L, Ramin SM, Mele L, Wapner RJ, Johnson F, Peaceman AM, et al.Bone metabolism in fetuses of pregnant women exposed to single and multiple courses of corticosteroids. Obstetrics & Gynecology 2009;114(1):38-44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashima JN, Lai Y, Wapner RJ, Sorokin Y, Dudley DJ, Peaceman A, et al.The effect of maternal body mass index on neonatal outcome in women receiving a single course of antenatal corticosteroids. American Journal of Obstetrics and Gynecology 2010;202(3):263.e1-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NCT00015002.Repeat antenatal steroids trial [A randomized placebo-controlled trial of antenatal corticosteroid regimens]. clinicaltrials.gov/show/NCT00015002 (first received 17 April 2001). [CENTRAL: CN-02033229]
- Sawady J, Mercer B, Wapner R, Zhao Y, Sorokin Y, Johnson F, et al.The National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network beneficial effects of antenatal repeated steroids study: impact of repeated doses of antenatal corticosteroids on placental growth and histologic findings. American Journal of Obstetrics and Gynecology 2007;281:e1-8. [DOI] [PubMed] [Google Scholar]
- Sorokin Y, Romero R, for the NICHD MFMU Network.Elevated maternal serum IL-6 and CRP are associated with preterm delivery < 32 weeks and subsequent neonatal intraventricular hemorrhage. American Journal of Obstetrics and Gynecology 2005;193(6 Suppl):S62. [Google Scholar]
- Sorokin Y, Romero R, Mele L, Wapner RJ, Iams JD, Dudley DJ, et al.Maternal serum interleukin-6, C-reactive protein, and matrix metalloproteinase-9 concentrations as risk factors for preterm birth <32 weeks and adverse neonatal outcomes. American Journal of Perinatology 2010;27(8):631-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorokin Y for the NICHD MFMU Network.Effect of maternal BMI, number of courses and timing of antenatal corticosteroids: association with neonatal anthropometric measurements. American Journal of Obstetrics and Gynecology 2004;191(6 Suppl 1):S106. [Google Scholar]
- Wapner R, Sorokin Y, Mele L, Johnson F, Dudley D, Spong CY, et al.Long-term outcomes after repeat doses of antenatal corticosteroids. New England Journal of Medicine 2007;357:1190-8. [DOI] [PubMed] [Google Scholar]
- Wapner R.Long term follow-up of infants receiving single vs repeat courses of antenatal corticosteroids. American Journal of Obstetrics and Gynecology 2006;195(6 Suppl 1):S2. [Google Scholar]
- Wapner RJ, for the NICHD MFMU Network.A randomized trial of single versus weekly courses of corticosteroids. American Journal of Obstetrics and Gynecology 2003;189(6 Suppl 1):S56. [Google Scholar]
- Wapner RJ, for the NICHD MFMU Network.Maternal and fetal adrenal function following single and repeat courses of antenatal corticosteroids (ACS). American Journal of Obstetrics and Gynecology 2005;193(6 Suppl):S5. [Google Scholar]
- Wapner RJ, Sorokin Y, Thom EA, Johnson F, Dudley DJ, Spong CY, et al.Single versus weekly courses of antenatal corticosteroids: evaluation of safety and efficacy. American Journal of Obstetrics and Gynecology 2006;195(3):633-42. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Bontis 2011 {published data only}
- Bontis N, Vavilis D, Tsolakidis D, Goulis DG, Tzevelekis P, Kellartzis D, et al.Comparison of single versus multiple courses of antenatal betamethasone in patients with threatened preterm labor. Clinical and Experimental Obstetrics and Gynecology 2011;38(2):165-7. [PubMed] [Google Scholar]
CTRI/2017/04/008326 {published data only}
- CTRI/2017/04/008326.Improving newborn survival in preterm birth [A multi-country, multi-centre, two-arm, parallel, double-blind, placebo-controlled, randomized trial of antenatal corticosteroids for women at risk of imminent birth in the early preterm period in hospitals in low-resource countries to improve newborn outcomes – Antenatal CorticosTeroids for Improving Outcomes in preterm Newborns (ACTION-I TRIAL)]. www.who.int/trialsearch/Trial2.aspx?TrialID=CTRI/2017/04/008326 (first received 10 April 2017).
CTRI/2017/05/008721 {published data only}
- CTRI/2017/05/008721.Improving newborn survival in late preterm birth [A multi-country, multi-centre, two-arm, parallel, double-blind, placebo-controlled, randomized trial of antenatal corticosteroids for women at risk of imminent birth in the late preterm period in hospitals in low-resource countries to improve newborn outcomes – Antenatal CorticosTeroids for Improving Outcomes in preterm Newborns (ACTION-II TRIAL)]. www.who.int/trialsearch/Trial2.aspx?TrialID=CTRI/2017/05/008721 (first received 31 May 2017). [CENTRAL: CN-01884324]
Ernawati 2016 {published data only}
- Ernawati, Gumilar E, Kuntoro, Soeroso J, Dekker G.Expectant management of preterm preeclampsia in Indonesia and the role of steroids. Journal of Maternal-fetal & Neonatal Medicine 2016;29(11):1736-40. [CENTRAL: CN-01141034] [EMBASE: 2015249052] [PMID: ] [DOI] [PubMed] [Google Scholar]
EUCTR2009‐010759‐29‐BE {published data only}
- EUCTR2009-010759-29-BE.Etude clinique sur l'administration de 3 doses de betamethasone (12mg) a 18 heures d'intervalle lors d'une menace d'accouchement premature dans les grossesses gemellaires traitees par Atosiban, plutot que 2 doses de betamethasone (12mg) a 24 heures d'intervalle; afin de diminuer le risque de syndrome de detresse respiratoire chez les nouveau-nes – CORTGEM. www.who.int/trialsearch/Trial2.aspx?TrialID=EUCTR2009-010759-29-BE (first received 30 March 2009).
Gyamfi‐Bannerman 2016 {published data only}
- Viteri OA, Doty MS, Alrais MA, Sibai BM.471: intended administration of antenatal late preterm steroids: is a single dose enough? American Journal of Obstetrics and Gynecology 2019;220(1):S315. [CENTRAL: CN-01710606] [EMBASE: 2001405752] [Google Scholar]
IRCT2014090912789N6 {published data only}
- IRCT2014090912789N6.Impact of betamethasone on preterm infants outcomes [Comparing the impact of one versus two doses of betamethasone on preterm infants outcomes: a clinical trial study]. www.who.int/trialsearch/Trial2.aspx?TrialID=IRCT2014090912789N6 (first received 20 October 2014). [CENTRAL: CN-01862384]
IRCT2015120415634N2 {published data only}
- IRCT2015120415634N2.The effect of antenatal intramuscular administration of two doses of betamethasone every 12 hours on acute pulmonary complications in preterm infants born [Comparing of antenatal intramuscular administration of two doses of betamethasone every 12 hours with standard method every 24 hours on acute neonatal pulmonary complications in preterm infants born from women with preterm premature of membrane]. www.who.int/trialsearch/Trial2.aspx?TrialID=IRCT2015120415634N2 (first received 27 September 2017). [CENTRAL: CN-01886227]
IRCT20191202045571N1 {published data only}
- IRCT20191202045571N1.The effect of betamethasone injection on maternal and neonatal outcomes in pregnant women with gestational age 34–37 weeks exposed to preterm labor in Sayyad Shirazi Hospital, Gorgan. www.irct.ir/trial/44053 (first received 17 July 2020). [CENTRAL: CN-02148336]
Kashanian 2018 {published data only}
- Kashanian M, Eshraghi N, Sheikhansari N, Bordbar A, Khatami E.Comparison between two doses of betamethasone administration with 12 hours vs. 24 hours intervals on prevention of respiratory distress syndrome: a randomised trial. Journal of Obstetrics and Gynaecology 2018;38(6):770-6. [CENTRAL: CN-01665275] [EMBASE: 621208178] [PMID: ] [DOI] [PubMed] [Google Scholar]
Mercer 2001 {published data only}
- Mercer B, Egerman R, Beazley D, Sibai B, Carr T, Sepesi J.Steroids reduce fetal growth: analysis of a prospective trial. American Journal of Obstetrics and Gynecology 2001;184(1):S7. [Google Scholar]
- Mercer B, Egerman R, Beazley D, Sibai B, Carr T, Sepesi J.Weekly antenatal steroids trial in women at risk of preterm birth: a randomized trial. American Journal of Obstetrics and Gynecology 2001;184(1):S6. [Google Scholar]
- Sawady J, Mercer B.Impact of repeated doses of antenatal corticosteroids on placental growth and histology. American Journal of Obstetrics and Gynecology 2006;195(6 Suppl 1):S73. [Google Scholar]
NCT03446937 {published data only}
- NCT03446937.Effect of antenatal corticosteroids on neonatal morbidity. clinicaltrials.gov/show/NCT03446937 (first received 5 February 2018). [CENTRAL: CN-01483469]
Romejko‐Wolniewicz 2013 {published data only}
- Romejko-Wolniewicz E, Oleszczuk L, Zareba-Szczudlik J, Czajkowski K.Dosage regimen of antenatal steroids prior to preterm delivery and effects on maternal and neonatal outcomes. Journal of Maternal-Fetal and Neonatal Medicine 2013;26(3):237-41. [DOI] [PubMed] [Google Scholar]
Schmitz 2019 {published data only}
- Schmitz T, Alberti C, Ursino M, Baud O, Aupiais C.Full versus half dose of antenatal betamethasone to prevent severe neonatal respiratory distress syndrome associated with preterm birth: study protocol for a randomised, multicenter, double blind, placebo-controlled, non-inferiority trial (BETADOSE). BMC Pregnancy and Childbirth 2019;19(1):67. [CENTRAL: CN-01787973] [EMBASE: 626359019] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Sohrabvand 2001 {published data only}
- Sohrabvand F, Behbahani B, Kazeminejad A.Effects of single versus multiple courses of corticosteroid therapy on pregnancy results in women with PPROM. Journal of Perinatal Medicine 2001;29 Suppl 1(Pt 2):528. [Google Scholar]
Thorp 2000 {published data only}
- Thorp JA, Yeast JD, Cohen GR, Wickstrom EA, D'Angelo LJ.Repeated antenatal betamethasone and perinatal outcome. American Journal of Obstetrics and Gynecology 2000;182(1 Pt 2):S21. [Google Scholar]
References to studies awaiting assessment
Atarod 2014 {published data only}
- Atarod Z, Taghipour M, Roohanizadeh H, Fadavi S, Taghavipour M.Effects of single course and multicourse betamethasone prior to birth in the prognosis of the preterm neonates: a randomized, double-blind placebo-control clinical trial study. Journal of Research in Medical Sciences 2014;19(8):715-9. [PMC free article] [PubMed] [Google Scholar]
- Atarod Z.Single versus multiple courses of antenatal betamethasone: evaluation of preterm infant's outcome. www.irct.ir/trial/1175 (first received 16 May 2009).
References to ongoing studies
NCT02469519 {published data only}
- NCT02469519.Impact of a booster course of antenatal steroids on neonatal outcome in patients with premature rupture of the membranes [A randomized double-blinded trial comparing the impact of one versus two courses of antenatal steroids on neonatal outcome in the patient with prelabor premature rupture of the membranes]. clinicaltrials.gov/show/NCT02469519 (first received 5 June 2015). [CENTRAL: CN-01552815]
NCT02939742 {published data only}
- NCT02939742.Does a rescue course of betamethasone in pregnant women with PPROM decrease neonatal morbidity? [Does a repeat course of antenatal corticosteroids in pregnant women with preterm premature rupture of membranes decrease neonatal morbidity?]. clinicaltrials.gov/show/nct02939742 (first received 23 September 2016). [CENTRAL: CN-01583154]
Additional references
ACOG 2017
- American College of Obstetrics and Gynecology (ACOG) Committee on Obstetric Practice.Committee opinion No. 713: antenatal corticosteroid therapy for fetal maturation. Obstetrics and Gynecology 2017;130(2):e102-9. [DOI: ] [DOI] [PubMed] [Google Scholar]
AIHW 2020
- Australian Institute of Health and Welfare.Australia's Mothers and Babies 2018: in Brief. Perinatal Statistics Series no. 36. Canberra (Australia): Australian Institute of Health and Welfare, 2020. [ISBN: 978-1-76054-681-6] [Google Scholar]
Antenatal Corticosteroids CPG Panel 2015
- Antenatal Corticosteroids Clinical Practice Guidelines Panel.Antenatal corticosteroids given to women prior to birth to improve fetal, infant, child and adult health: Clinical Practice Guidelines. Auckland (New Zealand): Liggins Institute, The University of Auckland, 2015. [Google Scholar]
Ashwood 2006
- Ashwood PJ, Crowther CA, Willson KJ, Haslam RR, Kennaway DJ, Hiller JE, et al.Neonatal adrenal function after repeat dose prenatal corticosteroids: a randomized controlled trial. American Journal of Obstetrics and Gynecology 2006;194:861-7. [DOI] [PubMed] [Google Scholar]
Benediktsson 1993
- Benediktsson R, Lindsay RS, Noble J, Secki JR, Edwards CR.Glucocorticoid exposure in utero: new model for adult hypertension. Lancet 1993;341:339-41. [DOI] [PubMed] [Google Scholar]
Blencowe 2013
- Blencowe H, Cousens S, Chou D, Oestergaard M, Say L, Moller A-B, et al.Born too soon: the global epidemiology of 15 million preterm births. Reproductive Health 2013;10(Suppl 1 S2):1-14. [DOI: 10.1186/1742-4755-10-S1-S2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Carlisle 2017
- Carlisle JB.Data fabrication and other reasons for non-random sampling in 5087 randomised, controlled trials in anaesthetic and general medical journals. Anaesthesia 2017;72(8):944-52. [DOI: ] [DOI] [PubMed] [Google Scholar]
Cheong 2017
- Cheong JL, Anderson PJ, Burnett AC, Roberts G, Davis N, Hickey L, et al for the Victorian Infant Collaborative Study Group.Changing neurodevelopment at 8 Years in children born extremely preterm since the 1990s. Pediatrics 2017;139(6):e20164086. [DOI: ] [DOI] [PubMed] [Google Scholar]
Crowther 2019
- Crowther CA, Middleton PF, Voysey M, Askie L, Zhang S, Martlow TK, et al.Effects of repeat prenatal corticosteroids given to women at risk of preterm birth: an individual participant data meta-analysis. PLoS Medicine 2019;16(4):e1002771. [DOI: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Dalziel 2005a
- Dalziel SR, Walker NK, Parag V, Mantell C, Rea HH, Rodgers A, et al.Cardiovascular risk factors after antenatal exposure to betamethasone: 30-year follow-up of a randomised controlled trial. Lancet 2005;365(9474):1856-62. [DOI] [PubMed] [Google Scholar]
Dalziel 2005b
- Dalziel SR, Lim VK, Lambert A, McCarthy D, Parag V, Rodgers A, et al.Antenatal exposure to betamethasone: psychological functioning and health related quality of life 31 years after inclusion in randomised controlled trial. BMJ 2005;331(7518):665. [DOI] [PMC free article] [PubMed] [Google Scholar]
Dunlop 1997
- Dunlop SA, Archer MA, Quinlivan JA, Beazley LD, Newnham JP.Repeated prenatal corticosteroids delay myelination in the ovine central nervous system. Journal of Maternal-Fetal Medicine 1997;6:309-13. [DOI] [PubMed] [Google Scholar]
Eldridge 2021
- Eldridge S, Campbell MK, Campbell MJ, Drahota AK, Giraudeau B, Reeves BC, et al.Revised Cochrane risk of bias tool for randomized trials (RoB 2). Additional considerations for cluster-randomized trials (RoB 2 CRT). www.riskofbias.info/welcome/rob-2-0-tool/rob-2-for-cluster-randomized-trials (accessed prior to 11 March 2021).
Esplin 2000
- Esplin M, Fausett M, Smith S, Oshiro B, Porter TF, Branch DW, et al.Multiple courses of antenatal steroids associated with a delay in long-term psychomotor development in children with birth weight < 1500 grams. American Journal of Obstetrics and Gynecology 2000;182(1 Pt 2):S24. [Google Scholar]
Fowden 1996
- Fowden AL, Szemere J, Hughes RS, Forhead AJ.The effects of cortisol on the growth rate of the sheep fetus during late gestation. Journal of Endocrinology 1996;151:97-105. [DOI] [PubMed] [Google Scholar]
French 1998
- French N, Hagan R, Evans S, Godfrey M, Newnham J.Repeated antenatal corticosteroids: behaviour outcomes in a regional population of very preterm infants. Pediatric Research 1998;43:214A. [Google Scholar]
French 1999
- French N, Hagan R, Evans S, Godfrey M, Newnham J.Repeated antenatal corticosteroids: size at birth and subsequent development. American Journal of Obstetrics and Gynecology 1999;180:114-21. [DOI] [PubMed] [Google Scholar]
French 2004
- French N, Hagan R, Evans S, Mullan A, Newnham J.Repeated antenatal corticosteroids: effects on cerebral palsy and childhood behaviour. American Journal of Obstetrics and Gynecology 2004;190:588-95. [DOI] [PubMed] [Google Scholar]
GRADEpro GDT [Computer program]
- GRADEpro GDT.Hamilton (ON): McMaster University (developed by Evidence Prime), accessed 20 September 2021. Available at gradepro.org.
Hasbargen 2001
- Hasbargen U, Reber D, Versmold H, Schulze A.Growth and development of children to 4 years of age after repeated antenatal steroid administration. European Journal of Pediatrics 2001;160:552-5. [DOI] [PubMed] [Google Scholar]
Higgins 2019
- Higgins JP, Savovic J, Page MJ, Sterne JA.Revised Cochrane risk-of-bias tool for randomized trials (RoB 2) [ ]. www.riskofbias.info/welcome/rob-2-0-tool/current-version-of-rob-2 (accessed prior to 11 March 2022).
Higgins 2021
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook.
Ikegami 1997
- Ikegami M, Jobe AH, Newnham J, Polk DH, Willet KE, Sly P.Repetitive prenatal glucocorticoids improve lung function and decrease growth in preterm lambs. American Journal of Respiratory and Critical Care Medicine 1997;156:178-84. [DOI] [PubMed] [Google Scholar]
Jensen 2002
- Jensen EC, Gallaher BW, Breier BH, Harding JE.The effect of a chronic maternal cortisol infusion on the late gestation fetal sheep. Journal of Endocrinology 2002;174:27-36. [DOI] [PubMed] [Google Scholar]
Jobe 1994
- Jobe AH.National Institutes of Health. Report of the Consensus Development Conference on the Effect of Corticosteroids for Fetal Lung Maturation on Perinatal Outcomes. Bethesda (MD): US Department of Health and Human Services, Public Health Service, 1994. [Google Scholar]
Liggins 1972
- Liggins GC, Howie RN.A controlled trial of antepartum glucocorticoid treatment for prevention of the respiratory distress syndrome in premature infants. Pediatrics 1972;50:515-25. [PubMed] [Google Scholar]
McGoldrick 2020
- McGoldrick E, Stewart F, Parker R, Dalziel SR.Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database of Systematic Reviews 2020, Issue 12. Art. No: CD004454. [DOI: 10.1002/14651858.CD004454] [DOI] [PMC free article] [PubMed] [Google Scholar]
McKenna 2000
- McKenna DS, Witter GM, Nagaraja HN, Samuels P.The effects of repeat doses of antenatal corticosteroids on maternal adrenal function. American Journal of Obstetrics and Gynecology 2000;183:669-73. [DOI] [PubMed] [Google Scholar]
McLaughlin 2003
- McLaughlin KJ, Crowther CA, Walker N, Harding JE.Effects of a single course of corticosteroids given more than 7 days before birth: a systematic review. Australian and New Zealand Journal of Obstetrics and Gynaecology 2003;43:101-6. [DOI] [PubMed] [Google Scholar]
Mildenhall 2006
- Mildenhall LF, Battin MR, Morton SM, Bevan C, Kuschel CA, Harding JE.Exposure to repeat doses of antenatal glucocorticoids is associated with altered cardiovascular status after birth. Archives of Disease in Childhood Fetal and Neonatal Edition 2006;91:F56-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Moise 1995
- Moise AA, Wearden ME, Kozinetz CA, Gest AL, Welty SE, Hansen TN.Antenatal steroids are associated with less need for blood pressure support in extremely premature infants. Pediatrics 1995;95:845-50. [PubMed] [Google Scholar]
Newnham 1999
- Newnham JP, Evans S, Godfrey M, Huang W, Ikegami M, Jobe A.Maternal, but not fetal, administration of corticosteroids restricts fetal growth. Journal of Maternal-Fetal Medicine 1999;8(3):81-7. [DOI] [PubMed] [Google Scholar]
NIH 1995
- NIH Consensus Development Panel.Effect of corticosteroids for fetal maturation on perinatal outcomes. JAMA 1995;273:413-8. [DOI] [PubMed] [Google Scholar]
Padbury 1996
- Padbury JF, Ervin G, Polk D.Extrapulmonary effects of antenatally administered steroids. Journal of Pediatrics 1996;128:167-72. [DOI] [PubMed] [Google Scholar]
RevMan Web 2021 [Computer program]
- Review Manager Web (RevMan Web).Version 3.9.0. The Cochrane Collaboration, 2021. Available at revman.cochrane.org.
Roberts 2006
- Roberts D, Dalziel S.Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database of Systematic Reviews 2006, Issue 3. Art. No: CD004454. [DOI: 10.1002/14651858.CD004454.pub2] [DOI] [PubMed] [Google Scholar]
Rojas‐Reyes 2012
- Rojas-Reyes MX, Morley CJ, Soll R.Prophylactic versus selective use of surfactant in preventing morbidity and mortality in preterm infants. Cochrane Database of Systematic Reviews 2012, Issue 3. Art. No: CD000510. [DOI: 10.1002/14651858.CD000510.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Schünemann 2013
- Schünemann H, Brożek J, Guyatt G, Oxman A, editor(s).GRADE handbook for grading quality of evidence and strength of recommendations (updated October 2013). GRADE Working Group, 2013. Available from gdt.guidelinedevelopment.org/app/handbook/handbook.html.
Seckl 2004
- Seckl JR, Meaney MJ.Glucocorticoid programming. Annals of the New York Academy of Sciences 2004;1032:63-84. [DOI] [PubMed] [Google Scholar]
Thorp 2002
- Thorp JA, Etzenhouser J, O'Connor M, Jones A, Jones P, Belden B, et al.Effects of phenobarbital and multiple-dose antenatal/postnatal steroid on developmental outcome at age 7 years. American Journal of Obstetrics and Gynecology 2002;185(6):S87. [Google Scholar]
Tschanz 1995
- Tschanz SA, Danke BM, Burri PH.Influence of postnatally administered glucocorticoids on rat lung growth. Biology of the Neonate 1995;68:229-45. [DOI] [PubMed] [Google Scholar]
Utama 2018
- Utama DP, Crowther CA.Transplacental versus direct fetal corticosteroid treatment for accelerating fetal lung maturation where there is a risk of preterm birth. Cochrane Database of Systematic Reviews 2018, Issue 6. Art. No: CD008981. [DOI: 10.1002/14651858.CD008981] [DOI] [PMC free article] [PubMed] [Google Scholar]
Wan 2014
- Wan X, Wang W, Liu J, Tong T.Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Medical Research Methodology 2014;14:135. [DOI] [PMC free article] [PubMed] [Google Scholar]
WHO 2015
- World Health Organization.WHO recommendations on interventions to improve preterm birth outcomes: evidence base. www.who.int/reproductivehealth/publications/maternal_perinatal_health/preterm-birth-guideline/en/ (accessed prior to 11 March 2022). [PubMed]
References to other published versions of this review
Crowther 2000
- Crowther CA, Harding JE.Repeat doses of prenatal corticosteroids for women at risk of preterm birth for preventing neonatal respiratory disease. Cochrane Database of Systematic Reviews 2000, Issue 2. Art. No: CD003935. [DOI: 10.1002/14651858.CD003935] [DOI] [PubMed] [Google Scholar]
Crowther 2007
- Crowther CA, Harding JE.Repeat doses of prenatal corticosteroids for women at risk of preterm birth for preventing neonatal respiratory disease. Cochrane Database of Systematic Reviews 2007, Issue 3. Art. No: CD003935. [DOI: 10.1002/14651858.CD003935.pub2] [DOI] [PubMed] [Google Scholar]
Crowther 2011
- Crowther CA, McKinlay CJ, Middleton P, Harding JE.Repeat doses of prenatal corticosteroids for women at risk of preterm birth for improving neonatal health outcomes. Cochrane Database of Systematic Reviews 2011, Issue 6. Art. No: CD003935. [DOI: 10.1002/14651858.CD003935.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Crowther 2015
- Crowther CA, McKinlay CJ, Middleton P, Harding JE.Repeat doses of prenatal corticosteroids for women at risk of preterm birth for improving neonatal health outcomes. Cochrane Database of Systematic Reviews 2015, Issue 7. Art. No: CD003935. [DOI: 10.1002/14651858.CD003935.pub4] [DOI] [PMC free article] [PubMed] [Google Scholar]


