Abstract
Background
Control interventions in randomised trials provide a frame of reference for the experimental interventions and enable estimations of causality. In the case of randomised trials assessing patients with mental health disorders, many different control interventions are used, and the choice of control intervention may have considerable impact on the estimated effects of the treatments being evaluated.
Objectives
To assess the benefits and harms of typical control interventions in randomised trials with patients with mental health disorders. The difference in effects between control interventions translates directly to the impact a control group has on the estimated effect of an experimental intervention. We aimed primarily to assess the difference in effects between (i) wait‐list versus no‐treatment, (ii) usual care versus wait‐list or no‐treatment, and (iii) placebo interventions (all placebo interventions combined or psychological, pharmacological, and physical placebos individually) versus wait‐list or no‐treatment. Wait‐list patients are offered the experimental intervention by the researchers after the trial has been finalised if it offers more benefits than harms, while no‐treatment participants are not offered the experimental intervention by the researchers.
Search methods
In March 2018, we searched MEDLINE, PsycInfo, Embase, CENTRAL, and seven other databases and six trials registers.
Selection criteria
We included randomised trials assessing patients with a mental health disorder that compared wait‐list, usual care, or placebo interventions with wait‐list or no‐treatment .
Data collection and analysis
Titles, abstracts, and full texts were reviewed for eligibility. Review authors independently extracted data and assessed risk of bias using Cochrane’s risk of bias tool. GRADE was used to assess the quality of the evidence. We contacted researchers working in the field to ask for data from additional published and unpublished trials.
A pre‐planned decision hierarchy was used to select one benefit and one harm outcome from each trial. For the assessment of benefits, we summarised continuous data as standardised mean differences (SMDs) and dichotomous data as risk ratios (RRs). We used risk differences (RDs) for the assessment of adverse events. We used random‐effects models for all statistical analyses. We used subgroup analysis to explore potential causes for heterogeneity (e.g. type of placebo) and sensitivity analyses to explore the robustness of the primary analyses (e.g. fixed‐effect model).
Main results
We included 96 randomised trials (4200 participants), ranging from 8 to 393 participants in each trial. 83 trials (3614 participants) provided usable data. The trials included 15 different mental health disorders, the most common being anxiety (25 trials), depression (16 trials), and sleep‐wake disorders (11 trials).
All 96 trials were assessed as high risk of bias partly because of the inability to blind participants and personnel in trials with two control interventions. The quality of evidence was rated low to very low, mostly due to risk of bias, imprecision in estimates, and heterogeneity.
Only one trial compared wait‐list versus no‐treatment directly but the authors were not able to provide us with any usable data on the comparison.
Five trials compared usual care versus wait‐list or no‐treatment and found a SMD –0.33 (95% CI ‐0.83 to 0.16, I² = 86%, 523 participants) on benefits.
The difference between all placebo interventions combined versus wait‐list or no‐treatment was SMD −0.37 (95% CI −0.49 to −0.25, I² = 41%, 65 trials, 2446 participants) on benefits. There was evidence of some asymmetry in the funnel plot (Egger’s test P value of 0.087). Almost all the trials were small. Subgroup analysis found a moderate effect in favour of psychological placebos SMD ‐0.49 (95% CI −0.64 to −0.30; I² = 53%, 39 trials, 1656 participants). The effect of pharmacological placebos versus wait‐list or no‐treatment on benefits was SMD ‐0.14 (95% CI −0.39 to 0.11, 9 trials, 279 participants) and the effect of physical placebos was SMD −0.21 (95% CI −0.35 to −0.08, I² = 0%, 17 trials, 896 participants). We found large variations in effect sizes in the psychological and pharmacological placebo comparisons. For specific mental health disorders, we found significant differences in favour of all placebos for sleep‐wake disorders, major depressive disorder, and anxiety disorders, but the analyses were imprecise due to sparse data.
We found no significant differences in harms for any of the comparisons but the analyses suffered from sparse data.
When using a fixed‐effect model in a sensitivity analysis on the comparison for usual care versus wait‐list and no‐treatment, the results were significant with an SMD of –0.46 (95 % CI –0.64 to –0.28). We reported an alternative risk of bias model where we excluded the blinding domains seeing how issues with blinding may be seen as part of the review investigation itself. However, this did not markedly change the overall risk of bias profile as most of the trials still included one or more unclear bias domains.
Authors' conclusions
We found marked variations in effects between placebo versus no‐treatment and wait‐list and between subtypes of placebo with the same comparisons. Almost all the trials were small with considerable methodological and clinical variability in factors such as mental health population, contents of the included control interventions, and outcome domains. All trials were assessed as high risk of bias and the evidence quality was low to very low.
When researchers decide to use placebos or usual care control interventions in trials with people with mental health disorders it will often lead to lower estimated effects of the experimental intervention than when using wait‐list or no‐treatment controls. The choice of a control intervention therefore has considerable impact on how effective a mental health treatment appears to be. Methodological guideline development is needed to reach a consensus on future standards for the design and reporting of control interventions in mental health intervention research.
Keywords: Humans; Anxiety; Anxiety Disorders; Depressive Disorder, Major; Mental Health; Psychotherapy; Randomized Controlled Trials as Topic
Plain language summary
Control interventions in randomised trials for people with a mental health disorder
This systematic review assesses the effects of different control interventions in randomised trials including patients with a mental health disorder. In randomised trials, patients are assigned by chance to one of two or more groups – usually an experimental intervention and a control intervention. There are many types of control interventions in mental health intervention research. Some of the most common are different types of placebos that lack what is assumed to be the active component in the experimental intervention, and usual care, where patients receive the standard treatment for their mental health disorder in the area where they live. Two other types of control interventions are wait‐list or no‐treatment where patients receive no trial‐related care during the study (although some patients may receive care outside the studies). Wait‐list patients are often offered the experimental intervention after the trial has been finalised if it is likely to provide more benefits than harms, while no‐treatment participants are not offered the experimental intervention by the researchers.
We searched for randomised trials with patients with mental health disorders where wait‐list, usual care, or placebo interventions were compared with either wait‐list or no‐treatment. We looked at differences between all the types of control interventions on beneficial effects and whether they caused any adverse effects. We included 96 trials with a total of 4200 participants. Only 83 trials (3614 participants) provided usable data. Fifteen different mental health disorders were included. We found that all the trials were at high risk of bias in how they had been conducted, which reduced the interpretability of our findings. However, the risk of bias was mostly due to lack of blinding in the placebo studies, which may be seen as an aspect of the review's methodological question rather than a flaw with the review itself. We found no clinically important differences for usual care or wait‐list control interventions in the main analyses, however in our secondary analyses we found a clinically important favourable difference for usual care. In general, placebo control interventions tended to be favourable over no‐treatment or wait‐list control interventions across mental health disorders. We found no clinically important differences on adverse events.
This review suggests that different control interventions have a tendency to yield very different estimates for the effects of the experimental intervention and that the choice of control intervention has a large impact on how effective a mental health treatment appears to be. Control interventions in trials with patients with mental health disorders are often poorly reported upon, and guidelines are needed to inform researchers on how to properly design, report, and interpret these trials.
Summary of findings
Summary of findings 1. Summary of findings.
| Benefits and harms of wait‐list compared with no‐treatment for mental health disorders | ||||||
|
Patient or population: patients with mental health disorders Settings: inpatient and outpatient Intervention: wait‐list Comparison: no‐treatment | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Wait‐list/no‐treatment | Usual care | |||||
| Wait‐list compared with no‐treatment | Only one cluster‐randomised trial compared a wait‐list intervention to a no‐treatment intervention was included (Howlin 2007). However, no usable data were provided in the full report, and the authors did engage in correspondence. Eighty‐four elementary school children with a autism spectrum disorder were randomised to either, i) immediate treatment, ii) delayed treatment (wait‐list), and iii) no‐treatment. Conclusions were that Picture Exchange Communication System (PECS) training indicated modest effectiveness for children with autism spectrum disorder. In general there were no differences on across outcome measures between the wait‐list and no‐treatment intervention groups. | |||||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk Ratio; RD: Risk Difference; RCT: Randomised clinical trial | ||||||
| GRADE Working Group grades of evidence High quality: further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: we are very uncertain about the estimate. | ||||||
Summary of findings 2. Summary of findings.
| Benefits and harms of usual care compared with wait‐list or no‐treatment for mental health disorders | ||||||
|
Patient or population: patients with mental health disorders Settings: inpatient and outpatient Intervention: usual care Comparison: wait‐list or no‐treatment | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Wait‐list/no‐treatment | Usual care | |||||
|
Usual care compared with wait‐list/no‐treatment (Variety of continuous outcome) (Post‐treatment) |
The mean score in the usual care group was 0.33 points lower (0.83 lower to 0.16 higher) | 523 (5 RCTs) |
⊕⊝⊝⊝ very low a,b,c | TSA adjusted CI = ‐2.32 to 1.15 TSA RIS = 1536 |
||
| Serious adverse events for all placebos | No data | |||||
| Non‐serious adverse events for all placebos | No data | |||||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RR: risk ratio; RD: risk difference; RCT: randomised clinical trial; TSA: Trial Sequential Analysis | ||||||
|
GRADE Working Group grades of evidence
High quality: further research is very unlikely to change our confidence in the estimate of effect.
Moderate quality: further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate.
Low quality: further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate.
Very low quality: we are very uncertain about the estimate. a We downgraded the quality of evidence by one level due to risk of bias b We downgraded the quality of evidence by one level due to inconsistency (in terms of either clinical and methodological heterogeneity) c We downgraded the quality of evidence by one level due to imprecision (wide confidence intervals) | ||||||
Summary of findings 3. Summary of findings.
| Benefits and harms of placebos compared with wait‐list or no‐treatment for mental health disorders | ||||||
|
Patient or population: patients with mental health disorders Settings: inpatient and outpatient Intervention: all placebos combined, psychological, pharmacological and physical placebos Comparison: wait‐list or no‐treatment | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of Participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Wait‐list/no‐treatment | Placebos | |||||
|
All placebos compared with wait‐list or no‐treatment (Variety of continuous outcome) (Post‐treatment) |
The mean score in the placebo group was 0.37 points lower (0.49 lower to 0.25 lower) | 2446 (65 RCTs) |
⊕⊕⊝⊝ low a,b | TSA adjusted Cl = −1.85 to −0.84 TSA RIS = 397 | ||
|
Psychological placebos compared with wait‐list or no‐treatment (Variety of continuous outcome) (Post‐treatment) |
The mean score in the placebo group was 0.49 points lower (0.66 lower to 0.31 lower) | 1263 (38 RCTs) |
⊕⊕⊝⊝ low a,b | TSA adjusted Cl = −2.54 to −1.02 TSA RIS = 454 | ||
|
Pharmacological placebos compared with wait‐list or no‐treatment (Variety of continuous outcome) (Post‐treatment) |
The mean score in the placebo group was 0.14 points lower (0.39 lower to 0.11 higher) | 279 (9 RCTs) |
⊕⊝⊝⊝ very low a,b,c | TSA adjusted Cl = −9.43 to 6.15 TSA RIS = 229 | ||
|
Physical placebos compared with wait‐list or no‐treatment (Variety of continuous outcome) (Post‐treatment) |
The mean score in the placebo group was 0.21 points lower (0.35 lower to 0.08 lower) | 896 (17 RCTs) |
⊕⊕⊝⊝ low a,b | TSA adjusted Cl = −3.64 to −0.49 TSA RIS = 194 | ||
|
Serious adverse events for all placebos compared with wait‐list or no treatment (Spontaneous reporting of dichotomous outcomes) (Post‐treatment) |
43 per 1000 | 27 per 1000 (95% CI 32 fewer to 23 higher) |
RD ‐0.00 (95% CI ‐0.03 to 0.03) |
517 (11 RCTs) |
⊕⊝⊝⊝ very low a,b,c | Not possible to calculate TSA on serious adverse events due to too little information use |
|
Non‐serious adverse events for all placebos compared with wait‐list or no treatment (Spontaneous reporting of dichotomous outcomes) (Post‐treatment) |
93 per 1000 |
96 per 1000 (95% CI 2 fewer to 7 higher) |
RD 0.03 (95% CI ‐0.02 to 0.08) |
590 (14 trials) |
⊕⊝⊝⊝ very low a,b,c | Not possible to calculate TSA on serious adverse events due to too little information use |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk Ratio; RD: Risk Difference; RCT: Randomised clinical trial; RIS: required information size; TSA: trial sequential analysis | ||||||
|
GRADE Working Group grades of evidence
High quality: further research is very unlikely to change our confidence in the estimate of effect.
Moderate quality: further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate.
Low quality: further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate.
Very low quality: we are very uncertain about the estimate. a We downgraded the quality of evidence by one level due to risk of bias b We downgraded the quality of evidence by one level due to inconsistency (in terms of either clinical and methodological heterogeneity) c We downgraded the quality of evidence by one level due to imprecision (wide confidence intervals) | ||||||
Background
Description of the methods being investigated
Control interventions in randomised trials provide a frame of reference for the experimental intervention and allow causal estimations of treatment efficacy and adverse events (Higgins 2019; Kazdin 2016; Sibbald 1998). This systematic review assesses the benefits and harms of different control interventions in randomised trials that include patients with a mental health disorder.
We included the following experimental interventions in the review (which are often described as control interventions — see Table 4): (a) wait‐list, (b) usual care, (c) psychological placebo, (d) pharmacological placebo, and (e) physical placebo. We included the following control interventions: (a) wait‐list and (b) no‐treatment. We also planned to compare wait‐list with no‐treatment. We conducted analyses across all included patient populations and within specific mental health disorders. We made direct comparisons between the control interventions by including trials with more than one control arm (often three‐armed randomised trials).
1. Description of experimental interventions.
| Name of intervention | Other common names reported in the literature | Definition | Examples |
| Psychological placebo | Attention placebo, credible placebo, common‐factor treatment control, sham intervention, pseudo control | Psychological placebos target the non‐specific or shared components of psychological treatments, such as human interaction variables, attending sessions, and patient expectations | In an example from Tan 1986, the psychological placebo participants were exposed to sessions of supportive group counselling, which were thought to represent the non‐specific component of the active intervention, which was cognitive behavioural therapy |
| Pharmacological placebo | Pill placebo, placebo tablet, dummy pill | Pharmacological placebos are inert substances, typically in pill or liquid form, which do not contain the active ingredients of a given drug treatment | Participants typically receive a pill containing starch, sugar, or lactose (Double 1993; Meissner 2011b). |
| Physical placebo | Sham intervention, credible placebo, pseudo control | Physical placebos target the inert components of a given physical treatment (such as acupuncture, needle injection, exercise regimens, surgery, or electromagnetic stimulation) | This could be sham acupuncture where the needles are blunted ( Tough 2009 ), or sham electromagnetic stimulation, where the machine is not turned on or electrodes are attached to inactive sites ( Sommer 2006 ) |
| Usual care | Treatment as usual (TAU), standard care, outpatient care, standard practice, support as usual, clinical care, routine care, existing‐practice control | Usual care reflects locally accepted treatment practices for a given mental health disorder. It is provided either by private or public practitioners and may involve both pharmacological and psychological treatment. | Patients allocated to usual care might receive a large variety of therapies with a theoretical blend of psychodynamic, humanistic and behavioural approaches ( Borduin 2009 ) |
| Wait‐list | Minimal contact control, delayed treatment | Wait‐list participants are assessed on repeated occasions, but are promised the "active" intervention after the trial has ended | In Ertl 2011, participants allocated to the wait‐list group were reassessed at baseline and follow‐up, and subsequently offered the active treatment, which was narrative exposure therapy |
| No‐treatment | Minimal contact control | No‐treatment participants are assessed on repeated occasions without receiving the active treatment intervention. Unlike wait‐list interventions, no‐treatment participants are not promised the "active" intervention after trial completion | No‐treatment participants in Miranda 2003 did not receive any mental‐health related treatment, and were not promised the active intervention (antidepressants or psychotherapy) after trial completion |
Wait‐list participants are typically assessed before and after a given time period, and they receive the experimental intervention after the final research assessment if it provides more benefits than harms. No‐treatment participants are also assessed on repeated occasions but are not promised the experimental intervention after the final assessment (Comer 2013). Furukawa and colleagues have proposed that wait‐list participants could become motivated to remain in poor health in order to receive a desired therapy after the trial has ended, and that those receiving no‐treatment might actively seek out other forms of care outside the trial during the trial period ( Furukawa 2014). Wait‐list participants could therefore be subject to so‐called nocebo effects (i.e. negative effects from inert interventions) (Colloca 2020), but the evidence on this is preliminary (Greville‐Harris 2015; Furukawa 2014). Wait‐list and no‐treatment comparators control for maturation, spontaneous improvement, regression to the mean, and observer‐expectancy effects (Comer 2013 ; Kienle 1997). Careful monitoring of participants in wait‐list and no‐treatment interventions is important to ensure toleration of treatment delays and ethical compliance Comer 2013; Mohr 2009).
Usual care (sometimes also referred to as treatment as usual) is a control intervention that attempts to mirror the locally accepted treatment practices for a given mental health disorder. This control intervention may include both pharmacological and psychological treatments that are administered by relevant practitioners (Freedland 2011). The research teams are often not involved in the care of these patients. Usual care control groups are typically subject to large clinical and methodological heterogeneity, the practitioners receive little supervision, and the interventions often use a mixture of different theoretical approaches (Comer 2013 ; Kazdin 2015 ; Löfholm 2013 ). Despite these issues, usual care arguably reflects routine practice better than highly controlled psychiatric interventions ( Kazdin 2015; Mohr 2014 ) and when delivered well this type of control intervention is useful for determining whether novel psychiatric treatments are favourable to current practices (Mohr 2009). Usual care is sometimes standardised (Bateman 2009; Chanen 2008), which may involve manualisation, optimising of treatment structure, and adherence procedures (e.g. through supervision) (Bateman 2017; Cristea 2017; Kongerslev 2015).
This Cochrane methodology review distinguishes between three types of placebos. First, psychological placebos are designed to target the shared components of psychological treatments, such as attending sessions, the therapeutic relationship and patient expectations (Frank 1991; Hróbjartsson 2012; Rosenzweig 1936). It is both methodologically and theoretically difficult to discriminate between psychological placebos and psychological treatments (Borkovec 2005; Hróbjartsson 2012; Locher 2018; Mohr 2014; Wampold 2010; Wampold 2016). However, psychological placebos can be methodologically useful for differentiating between the proposed active and non‐active components in psychological treatments (Mohr 2009). Second, pharmacological placebos are inert substances in pill, liquid or other forms that do not contain the active ingredients of a given pharmacological treatment. Participants typically receive a pill containing starch, sugar, or lactose (Double 1993; Meissner 2011). The pharmacological placebo will need to match the active drug treatment (e.g. antidepressant medication) in size, form, colour, weight, smell, texture, solubility and taste, but not include any of the active components in the experimental intervention (Wager 2015). Third, physical placebos target the inert components of physical treatments (e.g. acupuncture, exercise regimens, or surgery). Here an example could be a staged electromagnetic stimulation procedure where the machine is not turned on or electrodes are attached to inactive sites (Sommer 2006).
Why it is important to do this review
The need to improve and develop treatments for mental health disorders is great (Holmes 2018; Karterud 2020; Leichsenring 2019; Weisz 2019). The type of comparator used in randomised trials with patients with mental health disorders may influence estimates of the effects of the experimental intervention, and it is important to know comparative benefits and harms of different types of comparator. However, these is a lack of consensus on how to design and report control interventions in randomised trials with these patients and evidence‐based guidelines are needed (Erlen 2015; Freedland 2011; Gold 2018; Kube 2017; Lund 2014; Mohr 2009). One aim of this review was to provide an empirical basis for future methodological guideline development in this field (Hoffmann 2013; Tajika 2015).
Wait‐list control and no‐treatment interventions may yield different effects in favour of experimental treatments depending on how they are structured, designed, and delivered, and it is very important to describe such factors. Wait‐list and no‐treatment conditions are also some of the most commonly used control interventions in psychiatric research (Mohr 2014) but may induce unwanted adverse events in participants, for instance from waiting to receive a treatment that patients may critically need (Furukawa 2014). If participants allocated to wait‐list and no‐treatment interventions show significantly more adverse events than those allocated to other control interventions, the ethical concerns and risks of overestimating the effects of clinical interventions in randomised trials should also be investigated (Cunningham 2013; Furukawa 2014).
We need more evidence on the content and effects of usual care as a control intervention in randomised trials (Rosenberg 2014; Swanson 2014) given the lack of discussion on how to design usual care conditions properly and how the use of usual care as a control condition may influence the reported effects (e.g. in favour of experimental treatments in study reports) (Kazdin 2015).
In a series of prior reviews, Hróbjartsson and Gøtzsche found, in general, no clinically important effects of psychological, pharmacological, and physical placebos versus wait‐list and no‐treatment interventions for various medical and psychiatric conditions (Hróbjartsson 2001; Hróbjartsson 2002; Hróbjartsson 2004; Hróbjartsson 2010). For example, the most recent update of their review (2010) included 44 trials with dichotomous outcomes and 158 trials with continuous outcomes, and they found moderate heterogeneity for both outcome domains (I 2 = 45% and 42%, respectively). For continuous outcomes, they also found large variation in effects between small and large trials (asymmetric funnel plots). Although the design of Hróbjartsson's and Gøtzsche's reviews is similar to the design of this review, their objective was to investigate the clinical relevancy of placebos, whereas this review is focused on methodological questions related to control interventions in randomised trials with patients with mental health disorders. It is, however, relevant to compare the two reviews methodologically. The present review is also interested in how placebo interventions may depend on factors such as type of mental health disorder, context of administration, information given to participants, and type of outcome measure (Charlesworth 2017; Fässler 2015; Holmes 2016; Hróbjartsson 2010; Howick 2019; Jensen 2017; Meissner 2011; O'Leary 1978; Rutherford 2014; Vase 2019Walach 2011; We 2012; Weimer 2015; Yeung 2017), which were also investigated by Hróbjartsson and Gøtzsche.
This review is based on our published protocol (Faltinsen 2019).
Objectives
Our objectives were to assess the comparative benefits and harms of different control interventions used in randomised trials with patients with mental health disorders. We specifically wanted to assess whether different control interventions yield different effect estimates compared with wait‐list or no‐treatment. We included the most common control interventions in mental health intervention research: wait‐list, usual care and placebos and compared these with wait‐list or no‐treatment. We also wanted to compare wait‐list with no‐treatment interventions.
We compared the following interventions:
wait‐list versus no‐treatment interventions;
usual care versus wait‐list or no‐treatment interventions;
all placebos combined, psychological, pharmacological, and physical placebos versus wait‐list or no‐treatment interventions.
Methods
Criteria for considering studies for this review
Types of studies
Randomised trials comparing wait‐list, usual care, or placebo interventions with either wait‐list or no‐treatment interventions were eligible. Parallel trials irrespective of language, publication year, and publication type were eligible. We included one cross‐over trial, but only used data from the first phase of the trial as a regular parallel trial. We included one cluster‐randomised trial. In case of articles published in languages other than English, we sought translation of the relevant sections. Unpublished studies where methods and results could be assessed in written form were eligible.
Types of data
All patients in each included trial were required to have a formal diagnosis of a mental health disorder according to the Diagnostic and Statistical Manual of Mental Disorders (DSM), First Edition (DSM‐I; APA 1952), Second Edition (DSM‐II; APA 1968), Third Edition (DSM‐III; APA 1980), Third Edition Revised (DSM‐III‐R; APA 1987), Fourth Edition (DSM‐IV; APA 1994), Fourth Edition Text Revision (DSM‐IV‐TR; APA 2000), and Fifth Edition (DSM‐5; APA 2013), or according to the International Classification of Diseases and Related Health Problems (ICD), Sixth Edition (ICD‐6; WHO 1949), Seventh Edition (ICD‐7; WHO 1955), Eighth Edition (ICD‐8; WHO 1967), Ninth Edition (ICD‐9; WHO 1975), 10th Edition (ICD‐10; WHO 1993), or 11th Edition (ICD‐11; WHO 2018). In some instances, the diagnostic classification system was not mentioned in the full report, but the participants fulfilled all symptoms to receive a diagnosis of a mental health disorder or they were formally diagnosed by a mental health professional. For trials published before the introduction of DSM or ICD criteria in 1949, participants were eligible if they had received a formal diagnostic assessment of a mental health disorder by a health professional.
We categorised the different mental health disorders according to the current nomenclature in the DSM‐5 (APA 2013). If all participants in a trial had a mental health disorder, but not the same one, we included the trial in all the analyses except those on specific mental health disorders (see Types of outcome measures). We included participants with or without comorbid conditions. Eligible participants were included irrespective of location, setting, and other demographic variables (including age).
Types of methods
Experimental interventions
We defined wait‐list, usual care, and placebo interventions as any interventions that were clearly labelled or reflected the properties of wait‐list, usual care, or placebo interventions, according to the criteria below (and in Table 4). We anticipated that most of the included interventions would be control interventions in three‐group randomised trials. The properties of the interventions deemed experimental for this methodology review were defined as the following (based on the work by Hróbjartsson 2010 ; Comer 2013; and Kazdin 2016.
Wait‐list: an intervention where participants are assessed on one or more occasions, and are promised the 'active' intervention after the trial has ended.
Usual care: an intervention that reflects locally accepted treatment practices for a given mental health disorder. It is provided either by private or public practitioners and may involve pharmacological, psychological treatment or both.
Psychological placebo: an intervention that targets the non‐specific or shared components of psychological treatments, such as treatment exposure and human interaction variables, attending sessions, and patient expectations.
Pharmacological placebo: an intervention that includes an inert substance, typically in the form of a pill or liquid, which does not contain the active ingredients of a given medication.
Physical placebo: an intervention that includes the inert components of a physical treatment (such as acupuncture, exercise regimens, surgery, or electromagnetic stimulation).
Comparator interventions
We included two comparators: wait‐list and no‐treatment (see Table 4). When wait‐list was the experimental intervention, we only compared it with no‐treatment interventions. We defined these comparator interventions as any interventions that were clearly labelled as, or reflected the properties of wait‐list and no‐treatment interventions. The properties of no‐treatment interventions were defined as the following (based on the work by Comer 2013).
No‐treatment: an intervention where participants are assessed on repeated occasions without receiving the experimental intervention. Unlike wait‐list interventions, no‐treatment participants are not promised the experimental treatment after trial completion.
Description of main comparisons
We conducted the comparisons on placebo and usual care interventions in the following order.
We first pooled wait‐list and no‐treatment interventions when compared with placebo and usual care interventions.
We then conducted subgroup analyses (see Subgroup analysis and investigation of heterogeneity) between wait‐list and no‐treatment interventions for all these pooled comparisons. If there were significant differences or substantial heterogeneity between the wait‐list and no‐treatment interventions for a given comparison, we conducted separate main analyses for the two comparison interventions. We expressed low confidence in these analyses if they had insufficient statistical power.
Types of outcome measures
Primary outcomes
Outcomes measuring the efficacy of wait‐list, usual care, and placebo interventions versus wait‐list or no‐treatment interventions for all mental health disorders combined.
Serious adverse events in wait‐list, usual care, and placebo interventions versus wait‐list or no‐treatment interventions for all mental health disorders combined and for specific mental health disorders.
Secondary outcomes
Outcomes measuring the efficacy of wait‐list, usual care, and placebo interventions versus wait‐list or no‐treatment interventions for specific mental health disorders.
Non‐serious adverse events in wait‐list, usual care, and placebo interventions versus wait‐list or no‐treatment interventions for all mental health disorders and for specific mental health disorders.
Description of outcomes
We conducted analyses across all included mental health disorders and within specific disorders. We grouped the specific disorders according to the classification in the DSM‐5 (APA 2013). We only calculated the efficacy for specific mental health disorders that had been included in at least three included trials. This was a pragmatic threshold inspired by Hróbjartsson and Gøtzsche ( Hróbjartsson 2010 ) to reduce spurious positive and negative findings in single trials.
For the outcomes measuring efficacy, we selected one outcome from each trial report. We conducted separate analyses on dichotomous and continuous outcomes (see Measures of the effect of the methods ). We used the following decision hierarchy to select the outcomes measuring effect.
We first included the outcome indicated as the primary outcome in the trial report (e.g. the one used for the sample size calculation). We preferred data from end of treatment over follow‐up data. This choice was inspired by Hróbjartsson 2010.
If the trial did not differentiate between primary and secondary outcomes or if more than one primary outcome was stated, we preferred continuous over dichotomous outcomes.
If there were multiple continuous outcomes, we preferred observer‐reported over patient‐reported outcomes, and blinded over non‐blinded outcomes.
If trials reported several observer‐reported outcomes, we included the outcomes that best captured the core symptoms of the mental health population being treated. Here, we preferred global scores over sub‐scores.
We then identified the outcome measure with the best psychometric properties (e.g. validity and reliability).
If still undecided, we randomly selected the outcome measure to use.
Serious adverse events were defined as any event that lead to death (e.g. suicide), is life‐threatening (e.g. suicidality), required in‐patient hospitalisation (e.g. self‐harm), prolonged hospitalisation, resulted in persistent or significant disability, or was any other important event that jeopardised the patient’s life or required intervention for prevention (ICH 2005 ). All other adverse events were considered non‐serious adverse events (ICH 2005). We conducted separate analyses for specific serious adverse events (e.g. suicide and self‐harm). We combined all non‐serious adverse events into a single estimate.
We extracted adverse events from studies as measured by standardised psychometric rating scales, such as laboratory values, or spontaneous reporting. We also located adverse events as described in the International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) (ICH 2005 ). Most of the adverse events data from the reports were spontaneously reported. Adverse events in randomised trials generally (Allen 2018) and for psychiatric treatments in particular, can be difficult to detect, and valid instruments to detect them are lacking (Lilienfeld 2007; Linden 2014; Pagsberg 2017; Storebø 2018). However, strategic searches for adverse events using standardised questionnaires are becoming more common (Pagsberg 2017; Storebø 2018). We corresponded with trial authors if they did not report data on adverse events.
Search methods for identification of studies
Electronic searches
We searched the electronic databases and trial registries listed below (guided by Bramer 2017) using the search strategies shown in Appendix 1 . The strategy for MEDLINE was used as a template for the other databases and trial registries, with modified syntax and controlled terms as necessary.
Bibliographic databases (April 2018)
MEDLINE Ovid (1946 to current) (see Appendix 1 for search strategy)
PsycINFO Ovid (1806 to current)
Embase Ovid (1974 to current)
Cochrane Central Register of Controlled Trials (CENTRAL; current issue), in The Cochrane Library.
Allied and Complementary Medicine Database (AMED; 1900 to current)
Web of Science Core Collection (1900 to current)
ProQuest Dissertations and Theses A&I (1743 to current)
Sociological Abstracts ProQuest (1952 to current)
Google Scholar ( https://scholar.google.no/)
BIOSIS Previews/Thomson Reuters (969 to current)
Open Grey (1997 to current)
Clinical trial registries (March 2019)
Australian New Zealand Clinical Trials Registry (ANZCTR; www.anzctr.org.au/BasicSearch.aspx ).
Clinical Trials ( clinicaltrials.gov ).
EU Clinical Trials Register ( www.clinicaltrialsregister.eu/ctr-search/search ).
ISRCTN Registry ( www.isrctn.com ).
UK Clinical Trials Gateway ( www.ukctg.nihr.ac.uk/#popoverSearchDivId ).
World Health Organization International Clinical Trials Registry Platform (WHO ICTRP; http://apps.who.int/trialsearch/ )
Searching other resources
We searched other resources at the end of the screening process. We surveyed relevant journals such as ACTA Psychiatrica Scandinavica, the American Journal of Psychiatry, Biological Psychiatry, the British Journal of Psychiatry, the BMJ, the International Journal of Clinical Psychopharmacology, JAMA Psychiatry, Journal of the American Academy of Child and Adolescent Psychiatry, Journal of Clinical Psychiatry, Journal of Clinical Psychopharmacology, Journal of Psychopharmacology, Lancet Psychiatry, Psychopharmacology, Psychotherapy Research and the Scandinavian Journal of Child and Adolescent Psychiatry and Psychology. We also reviewed abstracts of key psychiatric conferences, given the large proportion of conference abstracts that do not go on to full publication (Scherer 2018) and asked for relevant unpublished studies from experts in the field. We also checked the references in relevant literature.
Data collection and analysis
We conducted this review according to the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2019), and performed analyses using the latest version of RevMan (Review Manager 5) .
Selection of studies
Because we expected to retrieve large numbers of records from the electronic literature search, titles and abstracts were screened only once (divided equally between review authors EF and AT). For quality assurance, an additional review author (OJS) screened a random sample of the retrieved records to check whether there were differences in the included and excluded records between screeners. Three review authors (EF, AT and LB) independently screened the full‐text reports for studies judged to be potentially eligible. They discussed any disagreements, and an arbiter (OJS) made the final decision if agreement was not reached. Full‐text reports were obtained and assessed for inclusion based on the eligibility criteria (see Criteria for considering studies for this review). Randomised trials in this general topic area that do not fulfil the inclusion criteria are listed as excluded studies. We used EPPI Reviewer 4, an online software application for systematic review development, for screening of abstracts and full‐text reports (Thomas 2010). We included a PRISMA flow diagram to show the flow of included and excludes studies in the full review (Moher 2009).
Data extraction and management
Three review authors (EF, AT and LB) independently extracted data from the included studies. We resolved disagreements by discussion or using an arbiter (OJS), if necessary. Two review authors (EF and AT) entered data into Review Manager 5. We requested missing information by contacting relevant authors (Young 2011). We developed a data extraction form to facilitate standardisation of the data extraction process. The form included the following items: methods (e.g. trial design, setting, and country), types of participants (e.g. baseline demographics, inclusion and exclusion criteria), description of experimental and comparator interventions and their components (e.g. duration and intensity), outcome measures, and risk of bias assessment (see Appendix 2).
Assessment of risk of bias in included studies
Three review authors (EF, AT and LB) assessed the risk of bias using Cochrane’s risk of bias tool (RoB) 1.0 (Higgins 2011). There is an updated version of this tool (Eldridge 2016 ; Higgins 2017), but because it was still at the pilot stage when we rated risk of bias, we used the original version. For each included study, the data extractors independently categorised the risk of bias domains listed in Appendix 3 as being low, unclear (uncertain), or high risk of bias, according to the guidelines in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). Potential disagreements were resolved by discussion or using an arbiter (OJS), if necessary.
We defined trials at 'low risk of bias' as having low risk of bias on all domains. We defined trials with one or more unclear risk of bias domain as trials at ‘high risk of bias’. We evaluated the influence of risk of bias on our results (see Sensitivity analysis) due to the risk of overestimating beneficial intervention effects and underestimating adverse events in randomised trials with unclear or inadequate methodological quality (Kjaergard 2001; Lundh 2017; Moher 1998; Savović 2012; Savovic 2018; Schulz 1995; Wood 2008; Savovic 2018).
At the protocol stage ( Faltinsen 2019 ), we decided to include all the domains in RoB 1.0 including the blinding domains when assessing risk of bias in the included studies. However, we recognise that the blinding domains are the subject of the investigation in this review in the placebo comparisons, (i.e. one goal of our review is to assess differences in blinding between placebos and no‐treatment or wait‐list seeing) and we therefore decided to report two solutions to the bias assessment post hoc for placebo interventions: one bias assessment including the blinding domains and one without.
We assessed conflicts of interest in the included studies as a separate bias category outside of Cochrane's risk of bias tool. We assessed both financial and non‐financial conflicts of interest. Conflicts of interest were defined as situations in which professional judgments or actions regarding a primary interest are unduly influenced by a secondary interest (Institute of Medicine 2009). Examples of financial conflicts could be when a study's authors had received payment from a company manufacturing one of the study interventions. A non‐financial conflict of interest (often termed affiliation bias in psychotherapy research) could be if a study's authors had developed the treatment manual for the intervention being evaluated (Munder 2013).
Dichotomous data
We summarised dichotomous data as risk ratios (RR) for outcomes for efficacy and risk differences (RD) for adverse events. We used 95% confidence intervals (CIs) for both, and Trial Sequential Analysis (TSA)‐adjusted CIs if possible (see Subgroup analysis and investigation of heterogeneity).
Continuous data
For continuous data, we estimated standardised mean differences (SMD). We used SMD because we anticipated variation in the types of outcome measures. We calculated SMDs using scores from the end of intervention. We considered a statistical significant SMD effect size of: 0.15 or less to have no clinically meaningful effect; 0.15 to 0.40 to have a clinical meaningful but small effect; 0.40 to 0.75 to have a moderate effect; and greater than 0.75 to have a large treatment effect (Cohen1988 ). When the trials only reported change data, we pooled these with scores from the end of intervention (da Costa 2013). We explored whether inclusion of change data affected the outcomes by performing a sensitivity analysis (see Sensitivity analysis). If the direction of a given scale was opposite to that of most other scales, we multiplied the corresponding mean values by ‐1.00 to ensure adjusted values. If the trials did not report means and standard deviations (SDs), but reported other values such as t‐tests and P values, we attempted to transform these into means and SDs.
We used data from means and SDs in intention‐to‐treat (ITT) analyses as well as replacing missing values when available. We otherwise conducted the analyses based on the available data. We performed all calculations using RevMan software (Review Manager 5).
We summarised the outcomes measuring adverse events from count data (e.g. spontaneous reporting) as RD (see Subgroup analysis and investigation of heterogeneity).
Unit of analysis issues
We only included the first phase of cross‐over trials. We calculated study estimates on the basis of post‐treatment group results. If trials were cluster‐randomised we planned to appropriately control for cluster effects (robust standard errors or hierarchical linear models). If the necessary information was unclear or not available in the trial reports, we attempted to contact the original authors for further information. We used sensitivity analyses to assess the potential biases of inadequately controlled cluster‐randomised trials (Donner 2002) (see Sensitivity analysis).
Dealing with missing data
We contacted trial authors for relevant missing data on our primary and secondary outcomes (Young 2011). However, we did not contact authors of trials published before 1990 because of a lack of reliable contact information and the probability that these data would not have been preserved. If authors did not respond after two attempts to contact them, we stopped communications. If we were not able to obtain missing data, we used the available data (incomplete data) in our analyses. If data were not reported in a usable way, we consulted a statistician to explore its transformation. For a description of each trial with missing data see Table 5 .
2. Description of trials with missing data.
| Trial ID | Missing data | Methods of data generation |
| Borden 1986 | Missing information regarding how many participants randomised to each group | Since 30 patients was included in total, we assumed due to randomisation that there was an equal distribution of patients in each group. |
| Borkovec 1975 | The standard deviation (SD) was not reported on the Daily Sleep Questionnaire (DSQ) – Subscale difficulty experienced in falling asleep | An artificial SD was imported from Steinmark 1974 due to similar outcome, population, and control interventions |
| Brill 1964a ; Brill 1964b | No usable data reported | Not possible to generate data |
| Carlson 1993 | Missing information regarding how many participants randomised to each group | Since 28 patients was included in total, we assumed due to randomisation and ethical principles that the active arm (in this case methylphenidate) included an additional patient compared with the placebo and no‐treatment group |
| Crouch 1988 | No usable data reported | Not possible to generate data, and could not get in contact with authors |
| Doty 1975 | No usable data reported | Not possible to generate data |
| Fuchs 1977 | The standard deviation (SD) on the improvement of the mean on Beck Depression Inventory was not reported. | The SD was calculated from a F‐test statistic |
| Goldwasser 1987 | The SD was not reported on the Mini‐Mental State (MMS) | Not possible to generate data |
| Hekmat 1984 | The SD was not reported on the Timed Behavior Checklist (BCL) | Not possible to generate data |
| Howlin 2007 | No usable data reported | Not possible to generate data, and could not get in contact with authors |
| Krapfl 1970 | Mean was only reported in a figure, and the SD on the Behavioral Avoidance Test (BAT) was not reported | Mean value was generated from figure 1, and the SD was generated from Etringer 1982 ; Rosen 1976 , due to that both trials included similar outcome, population, and control interventions |
| Lang 1965 | No usable data reported | Not possible to generate data |
| Pelham 1992 | Missing information regarding how many participants randomised to each group | Since 38 patients was included in total, we assumed due to randomisation and ethical principles that the active arm (methylphenidate) and placebo included an additional patient each |
| Pendleton 1983 | Missing information regarding how many participants randomised to each group | Since 62 patients was included in total, we assumed due to randomisation and ethical principles that the two active arms (Negative practice and Desensitization) included an additional patient each |
| Roth 1964 | The standard deviation (SD) was not reported on either Global estimate of the severity of the patient’s illness and a global measure of over‐all improvement | Not possible to generate data |
| Rupert 1978 | No usable data reported | Not possible to generate data |
| Shealy 1979 | Separate means missing mild to moderate group. The standard deviation (SD) was not reported on the Daily Sleep and Relaxation Practice Questionnaire (DSRQ) |
We pooled the means from mild and moderate due to no information of patients in each group. A mean score was generated from the two groups. SDs was generated from Ascher 1979 ; Steinmark 1974 due to that these trials hade the same outcomes and population |
| Sibilio 1957 | No usable data reported | Not possible to generate data |
| Sommerness 1955 | No usable data reported | Not possible to generate data |
| Tan 1986 | The standard deviation (SD) on the improvement of the mean on Beck Depression Inventory was not reported | The SD was imported from Fuchs 1977 due to similar outcome and population |
| Trexler 1972 | The standard deviation (SD) was not reported on the Behavioral Checklist | Not possible to generate data |
| Matson 1980 | No usable data reported | Not possible to generate data, and could not get in contact with authors |
| Pearl 1956 | No usable data reported | Not possible to generate data, and could not get in contact with authors |
Assessment of heterogeneity
We expected to find evidence of substantial heterogeneity. We created subgroups based on study characteristics such as different control intervention, study duration, participants etc. (see Subgroup analysis and investigation of heterogeneity). We evaluated methodological heterogeneity by comparing trial designs. Assessment of statistical heterogeneity was carried out for comparisons by visual inspection of the graphs and the I² statistic (Higgins 2003). I² values between 0% and 40% indicated little heterogeneity; between 30% and 60% indicated moderate heterogeneity: between 50% and 90% indicated substantial heterogeneity; and between 75% and 100% indicated considerable heterogeneity (Higgins 2019). We also assessed statistical heterogeneity by Chi² tests (P < 0.10) and tau², an estimate of between‐study variability.
Assessment of reporting biases
Funnel plots were provided for comparisons that had a sufficient number of included trials. Asymmetry in the funnel plot could be due to publication bias or other reasons for heterogeneity between small and large trials (Higgins 2019). Egger’s statistical test was performed for primary outcomes included in the Table 3 to test for small‐study effects (Egger 1997). A visual inspection of funnel plots and Egger’s statistical test was not applied if there were fewer than 10 trials in the meta‐analysis, in keeping with the recommendations in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2019).
Data synthesis
We performed statistical analyses according to the recommendations in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2019). We applied the inverse variance method to give estimates from trials with less variance (mostly, larger studies) more weight. We used the random‐effects model for meta‐analysis because some clinical heterogeneity was expected to be present in most cases. We tested whether a fixed‐effect model provided different effect estimates in a sensitivity analysis (see Sensitivity analysis). If pooling of data seemed feasible, we combined the included study effects and calculated the associated 95% CIs.
Subgroup analyses (pre‐specified)
Type of active interventions: i) psychological intervention, ii) pharmacological intervention, iii) physical intervention, or iv) other or combination of interventions.
Overall risk of bias: i) high risk of bias compared with ii) low risk of bias.
Type of outcome domain: i) blinded observer‐reported, ii) non‐blinded observer‐reported, or iii) patient‐reported.
Type of comparator intervention: i) wait‐list or ii) no‐treatment.
Awareness of placebo intervention: i) participants were aware that they might receive a placebo or ii) participants were not aware of this.
Trial objective: i) a trial’s objective was clearly to assess the effects of placebo, usual care, or wait‐list interventions, or ii) no such objectives were stated.
Mean age of participants: i) < 18 years, ii) 18 to 50 years, or iii) > 50 years.
Duration of intervention: i) three months or above or ii) below three months.
Type of usual care: i) pharmacological, ii) psychological, iii) physical, or iv) other.
Standardised usual care: i) the usual care intervention was intentionally standardised or manualised or ii) no standardisation or manualisation.
Mode of psychological treatment in usual care and psychological placebo: i) individual psychological treatment or ii) group psychological treatment.
Subgroup analyses (post hoc)
Mental health diagnoses: i) formal diagnosis according to DSM/ICD, ii) fulfil symptoms of disorder ICD/DSM while not stating classifications systems, or iii) population is classified as having a mental disorder, but full diagnostic criteria not reported.
Type of psychological placebo: i) interaction placebo, ii) educational placebo, or iii) exposure placebo.
Type of physical placebo: i) acupuncture or acupressure placebo, ii) exercise and relaxation placebo, iii) technical device placebo, or iv) electromagnetic stimulation placebo.
Conflicts of interest: i) risk of non‐financial and financial conflicts of interest, or ii) no risk of conflicts of interest (Leichsenring 2019).
Imputed data: i) analyses with available outcome data or ii) analyses following the ITT principle.
Diversity‐adjusted required information size (RIS) and Trial Sequential Analysis (TSA)
Trial Sequential Analysis (TSA) is a methodology that combines a required information size (RIS) calculation for meta‐analyses with a threshold for statistical significance ( Brok 2009 ; Thorlund 2009; Wetterslev 2008; Wetterslev 2009; Wetterslev 2017). The TSA enables quantification of the statistical reliability of the data in cumulative meta‐analysis, and adjusted P values for sparse data and for repetitive testing on accumulating data (Brok 2008; Brok 2009; Thorlund 2009; Wetterslev 2008; Wetterslev 2017). Similar to an a priori sample size estimation in a single randomised trial, a meta‐analysis should include a RIS at least as large as the sample size of an adequately powered single trial to control the risks of random error. The TSA program can calculate the RIS in a meta‐analysis and provide an alpha‐spending boundary to adjust the significance level for sparse data and repetitive testing (Copenhagen Trial Unit 2018; Wetterslev 2008; Wetterslev 2017). This enables one to control for the risk of random error.
Multiple analyses of accumulating data when new trials emerge lead to repeated significance testing and introduces multiplicity issues. Therefore, the use of a conventional naïve P value exacerbates the risk of random errors (Berkey 1996; Thorlund 2011; Wetterslev 2017). By analysing meta‐analyses that do not reach the RIS with trial sequential alpha‐spending monitoring boundaries (analogous to interim monitoring boundaries in a single trial), this can be controlled for (Wetterslev 2008; Wetterslev 2017).
We calculated a RIS on the outcomes reported in the summary of findings tables in this review (i.e. the major findings of the review). If the TSA does not find significant results (no crossing of the alpha‐spending boundary and no crossing of the conventional boundary of P = 0.05) before the RIS has been reached, several conclusions may be inferred. We will either conclude that more trials are needed to reject or accept an intervention effect used for the calculation of the required sample size, or reject the anticipated effect, if the cumulative Z‐curve enters the futility area. We used an assumption that the minimal relevant clinical difference (MIREDIF) was approximately ½ SD on the used scale, which can be used as a MIREDIF (Norman 2003).
We calculated the diversity‐adjusted required information size (DARIS; that is the number of participants required to detect or reject a specific intervention effect in a meta‐analysis), and performed TSAs for the primary outcomes reporting continuous data at the end of treatment, based on the following a priori assumptions:
the SD of the primary outcomes;
an anticipated MIREDIF as a ½ SD on the used scale;
a maximum type I error of 3.3% (due to two primary outcomes; Jakobsen 2014);
a maximum type II error of 10% (minimum 90% power; Castellini 2018); and
the diversity observed in the meta‐analysis.
For the outcomes 'total serious adverse events' (dichotomous data), we calculated the diversity‐adjusted required information size (DARIS; i.e. number of participants in the
proportion of participants in the control group with serious adverse events;
relative risk reduction of 25%;
type I error of 3.3%;
type II error of 10%;
observed diversity of the meta‐analysis; and
we included trials with zero events by substituting 0.5 for zero ( Thorlund 2011 ).
It was not possible to calculate TSA on 'total serious adverse events' and 'non‐serious adverse events' due to a lack of information.
Summary of findings tables
We used the GRADE approach to construct three summary of findings tables to document primary review outcomes. GRADE evaluates the quality of a body of evidence based on the confidence that an effect estimate or association reflects the item being assessed. These considerations were based on within‐trial risk of bias, directness of evidence, heterogeneity of data, precision of effect estimates and risk of publication bias (Andrews 2013a; Andrews 2013b; Balshem 2018; Brunetti 2013; GRADE Working Group 2004; Guyatt 2011a; Guyatt 2011b; Guyatt 2011c; Guyatt 2011d; Guyatt 2011e; Guyatt 2011f ; Guyatt 2011g; Guyatt 2011h; Guyatt 2013a; Guyatt 2013b; Mustafa 2013). When possible, we used the SMD or the RR for the summary of findings table. We used the TSA as the rating for imprecision (Jakobsen 2014).
Sensitivity analysis
Trials contributing to statistical heterogeneity (‘outliers’) were removed to evaluate their impact on the overall pooled effect estimate. We removed outliers one by one and assessed the impact on the overall outcome.
We conducted sensitivity analyses to determine whether findings were sensitive to the following decisions made during the review process.
Analytical technique (e.g. fixed‐effect compared with random‐effects models)
Combination of data in continuous outcomes (end of intervention or compared with change scores)
Trial Sequential Analysis (TSA) as a sensitivity analysis for the imprecision rated with GRADE (Castellini 2018)
Including wait‐list interventions described as no‐interventions
Including no‐interventions described as wait‐list interventions
More information on the sensitivity analyses that we were not able to conduct is given in Differences between protocol and review .
Results
Description of studies
See Characteristics of included studies , Characteristics of excluded studies , Characteristics of studies awaiting classification , and Characteristics of ongoing studies .
Results of the search
All electronic databases and search periods are listed in the Methods section (see Electronic searches). The search was conducted in April 2018. The search strategy was comprehensive (see Appendix 1 and generated 64,529 records, but only 58,943 records could be exported from the databases and imported to Endnote. We consulted our research librarian and identified some records that did not include any title, abstract, or keywords, and they could therefore not be retrieved. Another 1034 records of clinical trials across the remaining trial registries could not be imported and had to be manually screened. Ten records were identified from references in other reviews.
Figure 1 shows our PRISMA flowchart. After duplicate check, 13,134 studies were excluded. We used an EPPI Reviewer 4 text mining software filter to identify reports that with 97% certainty was a systematic review or a randomised trial. This filter was used in three phases. In total, 10,167 reports were excluded because they did not fulfil the criteria for being a randomised trial, leaving 35,642 reports for abstract screening in EPPI reviewer 4, and 1034 clinical trials. Following screening of titles and abstracts, 1243 records were identified for full‐text screening. Six trials are still awaiting classification due to difficulties locating the trial reports (see Studies awaiting classification), while four trials are ongoing (see Ongoing studies). In total, 96 randomised trials described in 122 reports were eligible for the full review (see Figure 1 for a more detailed description).
1.
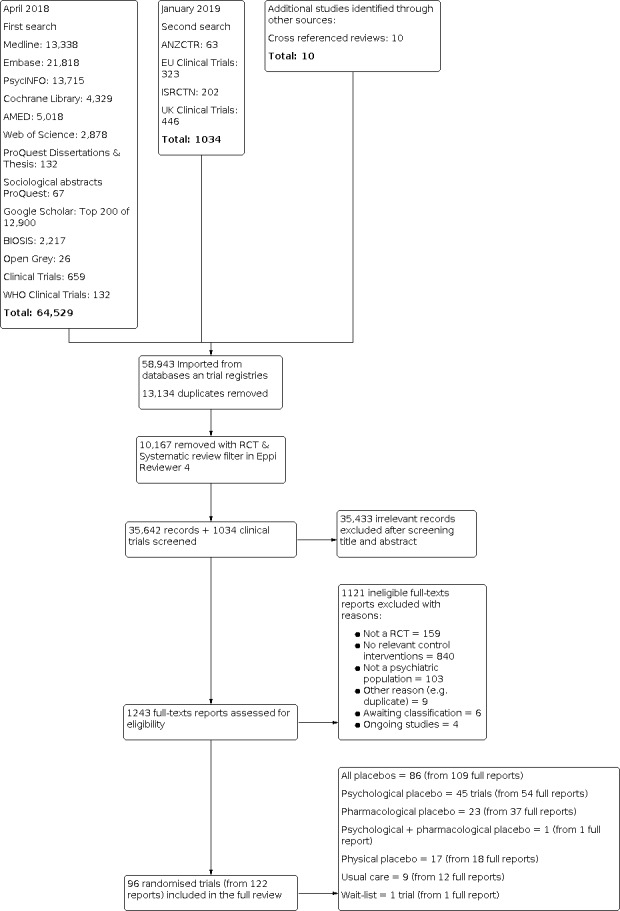
Study flow diagram.
* Powers 2008 was included in both psychological and pharmacological placebo
" Brill 1964 was included in both pharmacological placebo and usual care
< Klerman 1974 was divided into two different trials
‐: Peck 1974 was included only in all placebos analyses due to that the placebo group was a mix of psychological and pharmacological placebo
Author correspondence
We contacted authors from 35 trials with unclear or missing data and requested the necessary data but only 16 responded (Table 6) The other trials did not provide contact information or were below the threshold for contact.
3. Key demographic characteristics of the included studies.
Included studies
Here we summarise the key characteristics of the 96 included trials. Further detail can also be found in Characteristics of included studies.
Design
We included 96 trials (94 parallel‐group trials, one cluster‐randomised trial, and one cross‐over trial). The only cross‐over trial did not provide any usable data (Sibilio 1957). Only one trial compared wait‐list with no‐treatment (Howlin 2007). This was the only cluster‐randomised trial identified. Nine trials compared usual care versus either wait‐list or no‐treatment (Brill 1964b; Crisp 1991; Glogowska 2000; Matson 1980; Milby 1980; Rapee 2006; Rapee 2007; Robin 1976; Teri 1997). We included 45 trials on psychological placebos, 23 trials on pharmacological placebos and 17 trials on physical placebos.
One parallel‐group trial compared a pooled group of psychological and pharmacological placebo with no‐treatment (Peck 1976). One trial included three control groups (wait‐list, usual care and pharmacological placebo) and was split into two trials ( Brill 1964a; Brill 1964b).
Settings
Seventy‐four trials were conducted in outpatient settings and 20 trials were conducted in inpatient settings. Two trials were conducted combining inpatient and outpatient settings (Table 6).
Sample sizes
There was considerable variation in sample sizes between the trials. The total number of participants ranged from eight participants ( Kilmann 1987; Peck 1976) to 393 participants (Proudfoot 2013). Only five trials included more than 100 participants (Table 6).
Participants
The 96 trials included a total of 4200 participants but 586 participants could not be included due to missing data. The mean age ranged from 2.9 years (Glogowska 2000) to 86.5 years (Kwan 2017). Nineteen trials only included females, and 14 trials only included males. Seven trials did not state the sex of the participants (Table 6). All remaining trials included both sexes.
Diagnostic criteria
Participants were diagnosed as having a formal mental health disorder according to DSM‐II (two trials), DSM‐III (four trials), DSM‐III‐R (eight trials), DSM‐IV (15 trials), DSM‐IV‐TR (three trials) and ICD 9th edition (one trial) (Table 6). The most commonly used assessment instrument was Structured Clinical Interview for DSM (SCID; Spitzer 1989), which was used by eight trials. Twenty‐four trials fulfilled the symptoms of a mental health disorder from the available diagnostic classifications system at the time of the trial, but did not report a classification system. Thirty‐five trials reported a population classified as having a mental health disorder, but full diagnostic criteria were not reported (for more information see Table 6).
Diagnoses
The 96 trials included participants with the following 15 diagnoses: different forms of anxiety disorders (such as specific anxiety, social anxiety, or panic disorder, 25 trials); depression (16 trials); sleep‐wake disorders (11 trials); substance use disorders of different kind such as cocaine and alcohol dependency (eight trials); other unspecified disorders mentioned as 'psychiatric patients' only (eight trials); neurodegenerative diseases (six trials); schizophrenia (five trials); attention deficit hyperactivity disorder (ADHD) or attention deficit disorder (ADD) (five trials); post traumatic stress disorder (PTSD) (four trials); learning disability (three trials); intellectual disability (two trials); and one trial each for anorexia, autism, bulimia, encopresis, and erectile dysfunction (Table 6).
Experimental interventions
In the original trials, the placebo groups were all control interventions. We turned these control interventions into our experimental interventions in this systematic review.
Types of interventions
Only one trial (cluster‐randomised) compared a wait‐list intervention versus a no‐treatment intervention.
Three trials included usual care as a standard treatment, three trials included it as a form of outpatient psychotherapy, one trial as community‐based therapy, one trial as typical care control, and one trial did not specify its format (Robin 1976).
The 44 trials with psychological placebo included seven different labels for the psychological placebos. Thirteen trials used the term attention placebo control, 11 trials used non‐specific placebo counselling or treatment; seven trials used a quasi‐desensitisation placebo; four trials used a non‐specific educational placebo; two trials used a form of active treatment such as present‐centred therapy (Foa 2018), or emotion‐focused supportive therapy (Ehlers 2014). The other trials used different variations of placebo definitions, such as credible placebo or imagery relief placebo (Table 6).
Twenty trials used a psychological placebo with an interactive component; 16 trials included a psychological placebo with an exposure component; nine trials had a psychoeducational character (Table 6); and one trial combined psychological and pharmacological placebo (Peck 1976 ).
Sixteen trials provided pharmacological placebos in pill form, one trial used implants, and one trial used injection (Table 6).
Four trials used a pharmacological placebo with psychological treatment as an add‐on treatment. Six trials provided a physical placebo as a technical device, five trials as either acupuncture or acupressure, three trials as exercise and relaxation, and two trials as electromagnetic stimulation (Table 6).
Format of interventions
Twenty‐five trials administered psychological placebos individually, whereas 18 trials administered them in groups. Three trials used a combination of individual and group administration of psychological placebos (Table 6). All pharmacological and physical treatments were provided on an individual basis, except for one trial that combined a pharmacological treatment with group psychological treatment (Crouch 1988).
Duration of interventions
Seventy‐four trials had a duration of less than three months, while 21 had a duration of three months or more (Table 6). One trial did report the duration of the interventions (Hippman 2016). Where reported, the duration of treatment ranged from a single session (Etringer 1982; Karst 2007; Powers 2004; Powers 2008a; Powers 2008b; Wilson 1980; Wolitzky 2009) to two years of treatment (McLachlan 1991).
Control comparators
Fifty‐six trials included a no‐treatment control and 39 trials used a wait‐list control intervention. Ten trials labelled their comparator as a wait‐list intervention, but their description and definition of it led us to classify it as a no‐treatment control intervention. Four trials labelled their comparator as no‐treatment, but their description and definition led us to classify it as a wait‐list control intervention, and four trials received an add‐on psychotherapeutic treatment to the wait‐list group. One trial labelled their wait‐list as a 'minimal contact group', three trials labelled their wait‐list as a 'delayed treatment group', and one trial received an add‐on drug treatment (Table 6).
Concomitant treatment
Twenty trials did not allow concomitant psychotherapy to the placebo, treatment as usual, no‐intervention, or wait‐list groups, and 18 trials allowed the participant to receive a concomitant psychotherapy to the placebo, treatment as usual, no‐intervention, or wait‐list groups, (Table 6). The remaining trials did not report any information about concomitant treatments.
Twenty‐nine trials allowed the participants to receive a concomitant pharmacotherapy to the placebo, treatment as usual, no‐intervention or wait‐list groups, while 19 trials did not allow any sort of concomitant pharmacotherapy (Table 6). The other trials did not report any information about concomitant treatments.
Outcomes
Benefits
We followed our hierarchy for selecting outcomes measuring potential benefits (see Types of outcome measures). For more information for the individual trials, see Characteristics of included studies . We included 59 different outcomes for the placebo analyses (see Characteristics of included studies). The most common outcomes were Behavioral Avoidance Test (BAT) in eight trials, Daily Sleep Questionnaire (DSQ) in six trials, and Beck Depression Inventory (BDI) in four trials (Table 6). All outcomes included in the usual care analysis were different. The outcome in the cluster‐randomised trial of wait‐list versus no‐treatment included an outcome that was not used in any of the other analyses.
Adverse events
Only 11 trials reported serious adverse events, and only 14 trials reported non‐serious adverse events (Table 6). This was reported in the following ways: one trial used a complaint list ( Ayen 2004 ), another trial used an assessment with clinician‐administered posttraumatic stress disorder (PTSD) Scale for DSM‐5 (CAPS‐5), a third trial used a disulfiram‐ethanol reaction (DER), and the remaining 11 trials reported adverse events as a spontaneous reporting.
Excluded studies
In total, we excluded 1121 full‐text reports. Of the excluded full‐text reports, 159 were not a randomised trial, 840 did not compare a placebo or usual care control intervention versus either wait‐list or no‐treatment intervention. One hundred and three studies were excluded because the participants did not belong to a psychiatric population. Lastly, nine duplicates were identified in the full‐text screening and excluded (Figure 1). Thirty‐three excluded studies were close enough to the inclusion criteria to be listed in Characteristics of excluded studies.
Studies awaiting classification
Six full reports are awaiting classification due to difficulties in retrieving them (Studies awaiting classification). We were not able to locate the full text for these trials. Three were reported as an abstract (Bommert 1978; McLachlan 1993; Trianes Torres 1991), and three were only reported as a title ( Brandes 2010; Newton‐Cross 2017; Schwarzler 1999). We tried to contact the authors of the most recent studies (Brandes 2010 ; Newton‐Cross 2017), but did not receive any response. After two attempts, we terminated our correspondence.
Ongoing studies
We identified four ongoing studies that assessed different type of placebos or usual care versus wait‐list or no‐treatment (Heitman 2017; ISRCTN21392756; ISRCTN35717198; NCT00044629) (Characteristics of ongoing studies).
Risk of bias in included studies
Figure 2 and Figure 3 show our assessment of the risk of bias for each included study (see also Characteristics of included studies).
2.

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
We judged all trials to be at high risk of bias overall. All trials were rated at high risk of bias on blinding of participants and personnel because of the difficulties with blinding a trial with a no‐treatment or wait‐list comparator. However, the remaining risk of bias domains also had a large proportion of unclear risk of biases. We used all eligible trials in the meta‐analysis, as recommended in the Cochrane Handbook for Systematic Reviews of Interventions when all trials are assigned the same risk of bias ( Higgins 2011; Higgins 2019). We incorporated our risk of bias assessment when considering the quality of the evidence using the GRADE approach (Higgins 2011). Below is a breakdown of how the included trials scored on each risk of bias domain.
Allocation
Random sequence generation
Evidence suggests that trials which lack sufficient reporting of randomisation processes are more likely to present larger effect estimates for beneficial outcomes (Chalmers 1983; Schulz 1995; Kjaergard 2001; Savović 2012; Savovic 2018; Wood 2008). Trials were regarded as low risk of bias if they provided detailed description of their randomisation process such as stratification methods rather than just stating that it was randomised. Twenty‐seven trials provided sufficient information on how the randomisation sequence had been generated, and were rated at low risk of bias. The random sequence generation was rated at high risk of bias in four trials (Table 7). Examples included patients being randomised according to when they entered the treatment program (Poland 2013) or allowing participants to decline up to two treatments arms (Shalev 2012). The remaining 65 trials did not provide sufficient information on how the treatment allocation had been conducted and were assessed as unclear risk of bias.
4. Risk of bias in included studies.
Allocation concealment
We classified trials as low risk of bias if the allocation was conducted off‐site (centralised) by computer software or by an independent research coordinator not involved in delivering the therapy. Eighteen trials provided information about how the allocation was concealed and were therefore rated at low risk of bias. The other 78 trials did not provide any information regarding allocation concealment and were assessed as unclear risk of bias (Table 7)
Blinding
Blinding of outcome assessors
Forty‐five trials that reported that outcomes assessors were kept blind to treatment allocation were rated as low risk of bias. Thirty‐two trials were rated as high risk of bias due to reporting of inadequate blinding of outcomes assessors. The remaining 19 trials were assessed as unclear risk of bias due to a lack of sufficient information (Table 7).
Blinding of participants and personnel
We judged all trials as high risk of bias in this domain because the participants would be aware of whether they received treatment or not (e.g. allocated to either placebo or wait‐list).
Incomplete outcome data
Incomplete outcome data
Forty‐one trials were assessed as low risk of bias, due to the use of appropriate methods for handling missing data, such as intention‐to‐treat (ITT) analyses. Twenty‐nine trials either reported data on completers only or did not address the missing data and were considered as high risk of bias. The other trials did not report adequately information regarding missing data, and were therefore assessed as unclear risk of bias (Table 7).
Selective reporting
Most trials (n = 85) did not have a published protocol prior to initiation or did not provide sufficient information in the report to judge reporting bias and were considered as unclear risk of reporting bias. Six trials had a prior published protocol that provided sufficient information about all outcomes and did not exclude any of these in the full report. These trials were rated as low risk of biased reporting. Five trials published a protocol before the start of the trial, but we found discrepancies such as missing outcomes or an addition of outcomes and we rated them as high risk of bias.
Other potential sources of bias
Eighty‐seven trials were rated as low risk of other biases, eight trials were rated as high risk of bias in other potential sources of bias. These included four trials with researchers or authors who provided the treatment, two trials with attention bias or differences in duration of treatment, one trial with potential carry‐over effects, one trial that exceeded the passivity of placebo, and one trial with a time bias or assessment at different point for the groups. One trial was rated as unclear risk of others bias because of confounding differences between groups on the number of medical diagnoses other than dementia among participants (Table 7).
Conflicts of interest
We assessed six trials to be at risk of bias because of conflicts of interest. This could be a non‐financial affiliation bias, for instance if one of the investigators had developed a treatment evaluated in the trial, or bias from trials funded by a company manufacturing one of the interventions. We included subgroup analyses to test the difference between trials judged to be at risk of affiliation bias and those judged not to be at this risk (Analysis 16.17).
16.17. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 17: Affiliation bias
Effect of methods
Effects of interventions for all mental health disorders
Here, we present the results for each of the primary and secondary outcomes for the 19 comparisons. Seventy‐one trials reported data as continuous and 11 trials reported dichotomous data, whereas 13 trials did not report usable data. It was only possible to generate missing data for nine trials. For more information see Table 5 .
Wait‐list versus no‐treatment
We included a single cluster‐randomised trial that compared a wait‐list intervention versus a no‐treatment intervention (Howlin 2007). However, no usable data were provided in the full report and the trial authors did not respond to our request for additional data. In this trial, 84 elementary school children with autism spectrum disorder were randomised to either immediate treatment, delayed treatment (wait‐list) or no‐treatment. The study's conclusion was that Picture Exchange Communication System (PECS) training showed modest effectiveness for these children. In general, there were no differences across outcome measures between the wait‐list and the no‐treatment intervention groups.
Outcomes measuring benefits for usual care versus wait‐list or no‐treatment
Nine trials compared usual care versus wait‐list or no‐treatment. Two of these did not report usable data (Table 8). Five trials reported continuous data and two trials reported dichotomous data.
5. Effects of methods.
Usual care versus wait‐list or no‐treatment (continuous data)
No differences were found for beneficial effects comparing usual care versus wait‐list or no‐treatment when using a random‐effects model (standardised mean difference (SMD) −0.33, 95% confidence interval (CI) −0.83 to 0.16; 5 trials, 523 participants; P = 0.13; I 2 = 86% ; very low‐quality evidence; Analysis 2.1). No differences were found between the subgroups: usual care versus wait‐list (SMD −0.53, 95% CI −1.17 to 0.10; 3 trials, 443 participants; P = 0.10; I 2 = 91%), and usual care versus no‐treatment (SMD 0.08, 95% CI −0.38 to 0.53; 2 trials, 80 participants; P = 0.74; I 2 = 0%). Test for subgroup difference: Chi 2 = 2.33, df = 1, P = 0.13, I 2 = 57.1%.
2.1. Analysis.

Comparison 2: Usual care versus wait‐list or no‐treatment for all mental health disorders, Outcome 1: Usual care compared with wait‐list/no‐treatment for continuous data
When using a fixed‐effect model, usual care had a beneficial effect compared with wait‐list or no‐treatment (SMD −0.45, 95% CI −0.62 to −0.27; 5 trials, 523 participants; P = 0.01; I 2 = 83.3%; Analysis 25.1), and there were differences between the subgroups: usual care versus wait‐list (SMD −0.54, 95% CI −0.73 to −0.35; 3 trials, 443 participants; P < 0.00001; I 2 = 91%) and usual care versus no‐treatment (SMD 0.08, 95% CI −0.38 to 0.53; 2 trials, 80 participants; P = 0.74; I 2 = 0%). Test for subgroup differences: Chi 2 = 5.99, df = 1; P = 0.01; I 2 = 83.3%.
25.1. Analysis.

Comparison 25: Sensitivity analyses for usual care compared with wait‐list or no‐treatment, Outcome 1: Fixed effects
The TSA showed the cumulated Z curve enters the futility area, and therefore the anticipated intervention effect can be rejected (TSA‐adjusted confidence interval −2.32 to 1.15) (see Figure 4). Inspection of the funnel plot and Egger's test were not possible due to insufficient data.
4.
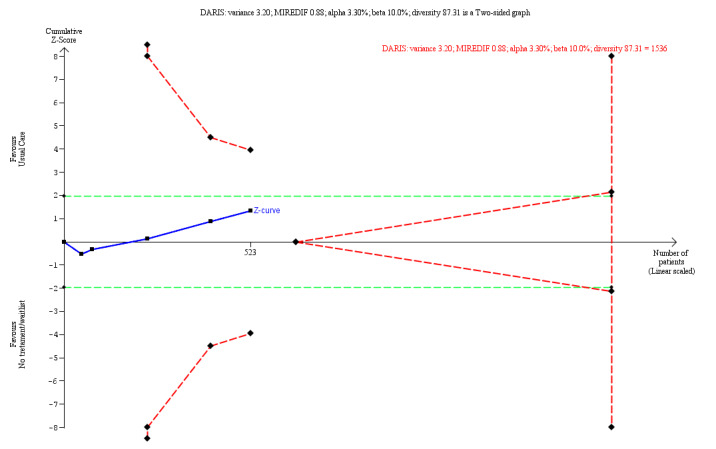
When we compared usual care with wait‐list and no‐treatment, we performed trial sequential analysis (TSA) on the primary outcome. The analysis shows that the required information size was not reached. See Figure 4 above. MIREDIF: Minimum relevant difference
Usual care versus wait‐list or no‐treatment (dichotomous data)
No differences were found for beneficial effects comparing usual care versus wait‐list or no‐treatment (RR 0.93, 95% CI 0.34 to 2.57; 2 trials, 260 participants; P = 0.89; I 2 = 79%; very low‐quality evidence; Analysis 2.2). Tests for subgroup differences were not done because both these trials were versus wait‐list.
2.2. Analysis.

Comparison 2: Usual care versus wait‐list or no‐treatment for all mental health disorders, Outcome 2: Usual care compared with wait‐list/no‐treatment for dichotomous data
Serious adverse events of usual care versus wait‐list or no‐treatment
None of the trials in this comparison reported data on serious adverse events.
Non‐serious adverse events of usual care versus wait‐list or no‐treatment
None of the trials in this comparison reported data on non‐serious adverse events.
Outcomes measuring benefits for all placebos versus wait‐list or no‐treatment
86 trials compared all placebos versus wait‐list or no‐treatment. 12 of these did not report usable data (Table 8).
All placebos versus wait‐list or no‐treatment (continuous data)
All placebo interventions showed beneficial effect compared with wait‐list or no‐treatment (SMD −0.37, 95% CI −0.49 to −0.25; 65 trials, 2446 participants; P < 0.00001, I 2 = 41% ; low‐quality evidence; Analysis 6.1) . Differences were identified between subgroups: all placebos versus wait‐list (SMD −0.55, 95% CI −0.76 to −0.35; 31 trials, 1410 participants; P < 0.00001; I 2 = 62%), and all placebos versus no‐treatment (SMD −0.18, 95% CI −0.30 to −0.05; 34 trials, 1036 participants; P = 0.005; I 2 = 0%). Test for subgroup differences: Chi 2 = 9.63, df = 1 ; P = 0.002; I 2 = 89.6%).
6.1. Analysis.

Comparison 6: All placebos versus waitl‐list or no‐treatment for people with mental health disorders, Outcome 1: All placebos compared with wait‐list/no‐treatment for continuous data
The Trial Sequential Analysis (TSA) showed that the required information size (RIS) was reached (n = 397) and that there was no risk of type 1 error (TSA adjusted confidence interval −1.85 to −0.84) (see Figure 5 in Appendix 4). Inspection of the funnel plot (see Figure 6 in Appendix 4) suggested a small potential bias (asymmetry), but we found no evidence of possible publication bias: Egger’s regression intercept (bias) −0.699 (two tailed, P = 0.087).
All placebos versus wait‐list or no‐treatment (dichotomous data)
We found no differences for beneficial effect comparing all placebos versus wait‐list or no‐treatment (risk ratio (RR) 1.05, 95% CI 0.74 to 1.48; 9 trials, 385 participants; P = 0.79; I 2 = 58%; very low‐quality evidence; Analysis 6.2) . Tests for subgroup differences were not done because all these trials were versus no‐treatment.
6.2. Analysis.

Comparison 6: All placebos versus waitl‐list or no‐treatment for people with mental health disorders, Outcome 2: All placebos compared with wait‐list/no‐treatment for dichotomous data
Serious adverse events of all placebos versus wait‐list or no‐treatment
Eleven trials compared versus placebos with wait‐list or no‐treatment and reported serious adverse events (Table 8).
All placebos versus wait‐list or no‐treatment (dichotomous data)
We found no differences for serious adverse events comparing all placebos versus wait‐list or no‐treatment (risk difference (RD) −0.00, 95% CI −0.03 to 0.03; 11 trials, 517 participants; P = 0.89; I 2 = 0%; very low‐quality evidence; Analysis 7.1)
7.1. Analysis.

Comparison 7: Serious adverse events of placebos versus wait‐list or no‐treatment for people with mental health disorders, Outcome 1: All placebos compared with wait‐list/no‐treatment for dichotomous data
It was not possible to construct a TSA‐figure on serious adverse events due to insufficient data. Inspection of the funnel plot (Figure 7 in Appendix 4 ) suggested no potential bias (asymmetry). We found no evidence of possible publication bias: Egger’s regression intercept (bias) −1.192 (two tailed, P = 0.408).
Psychological placebos versus wait‐list (dichotomous data)
We found no differences for serious adverse events comparing psychological placebos versus wait‐list (RD −0.01, 95% CI −0.07 to 0.04; 2 trials, 207 participants; P = 0.68; I 2 = 0%; very low‐quality evidence; Analysis 7.2)
7.2. Analysis.

Comparison 7: Serious adverse events of placebos versus wait‐list or no‐treatment for people with mental health disorders, Outcome 2: Psychological placebos compared with wait‐list/no‐treatment for dichotomous data
Pharmacological placebos versus no‐treatment (dichotomous data)
We found no differences for serious adverse events comparing pharmacological placebos versus no‐treatment (RD 0.01, 95% CI −0.08 to 0.09; 4 trials, 125 participants; P = 0.89; I 2 = 0%; very low‐quality evidence; Analysis 7.3)
7.3. Analysis.

Comparison 7: Serious adverse events of placebos versus wait‐list or no‐treatment for people with mental health disorders, Outcome 3: Pharmacological placebos compared with wait‐list/no‐treatment for dichotomous data
Physical placebos versus wait‐list or no‐treatment (dichotomous data)
We found no differences for serious adverse events comparing physical placebos versus wait‐list or no‐treatment (RD 0.00, 95% CI −0.04 to 0.04; 5 trials, 185 participants; P = 1.00; I 2 = 0%; very low‐quality evidence; Analysis 7.4)
7.4. Analysis.

Comparison 7: Serious adverse events of placebos versus wait‐list or no‐treatment for people with mental health disorders, Outcome 4: Physical placebos compared with wait‐list/no‐treatment for dichotomous data
Non‐serious adverse events of all placebos versus wait‐list or no‐treatment
Fourteen trials compared all placebos versus wait‐list or no‐treatment and reported non‐serious adverse events (Table 8).
All placebos compared with wait‐list or no‐treatment (dichotomous data)
We found no differences for non‐serious adverse events comparing all placebos versus wait‐list or no‐treatment (RD 0.03, 95% CI −0.02 to 0.08; 14 trials, 590 participants; P = 0.27; I 2 = 33% ; very low‐quality evidence; Analysis 8.1).
8.1. Analysis.

Comparison 8: Non‐serious adverse events of all placebos versus wait‐list or no‐ treatment for people with mental health disorders, Outcome 1: All placebos compared with wait‐list/no‐treatment for dichotomous data
Psychological placebos versus wait‐list or no‐treatment (dichotomous data)
We found no differences for non‐serious adverse events comparing psychological placebos versus wait‐list or no‐treatment (RD 0.01, 95% CI −0.18 to 0.19; 5 trials, 280 participants; P = 0.96; I 2 = 66%; very low‐quality evidence; Analysis 8.2).
8.2. Analysis.

Comparison 8: Non‐serious adverse events of all placebos versus wait‐list or no‐ treatment for people with mental health disorders, Outcome 2: Psychological placebos compared with wait‐list/no‐treatment for dichotomous data
Pharmacological placebos versus wait‐list or no‐treatment (dichotomous data)
We found no differences for non‐serious adverse events comparing pharmacological placebos versus no‐treatment (RD 0.08, 95% CI −0.04 to 0.21; 4 trials, 125 participants; P = 0.18; I 2 = 46%; very low‐quality evidence; Analysis 8.3).
8.3. Analysis.

Comparison 8: Non‐serious adverse events of all placebos versus wait‐list or no‐ treatment for people with mental health disorders, Outcome 3: Pharmacological placebos compared with wait‐list/no‐treatment for dichotomous data
Physical placebos versus wait‐list or no‐treatment (dichotomous data)
We found no differences for non‐serious adverse events comparing physical placebos versus wait‐list or no‐treatment (RD 0.00, 95% CI −0.04 to 0.04; 5 trials, 185 participants; P = 1.00; I 2 = 0%; very low‐quality evidence; Analysis 8.4).
8.4. Analysis.

Comparison 8: Non‐serious adverse events of all placebos versus wait‐list or no‐ treatment for people with mental health disorders, Outcome 4: Physical placebos compared with wait‐list/no‐treatment for dichotomous data
Outcomes measuring benefits for psychological placebos versus wait‐list or no‐treatment
Forty‐four trials compared psychological placebos versus wait‐list or no‐treatment. Five trials did not report usable data.(Table 8). One trial reported dichotomous data and 39 trials reported continuous data.
Psychological placebos versus wait‐list or no‐treatment (continuous data)
Psychological placebos showed a beneficial effect compared with wait‐list or no‐treatment interventions (SMD −0.49, 95% CI −0.64 to −0.30; 38 trials, 1656 participants; P < 0.00001; I 2 = 56%; low‐quality evidence; Analysis 9.1). Differences were identified between subgroups: psychological placebos versus wait‐list (SMD −0.66, 95% CI −0.92 to −0.40; 23 trials, 721 participants; P < 0.00001; I 2 = 41%), and psychological placebos versus no‐treatment (SMD −0.21, 95% CI −0.38 to −0.04; 15 trials, 542 participants; P = 0.02; I 2 = 0%). Test for subgroup difference: Chi 2 = 8.03, df = 1, P = 0.005; I 2 = 87.5%.
9.1. Analysis.

Comparison 9: Psychological placebos versus wait‐list or no‐treatment for people with mental health disorders, Outcome 1: Psychological placebos compared with wait‐list/no‐treatment for continuous data
The TSA showed that the RIS was reached (n = 454), and that there was no risk of type 1 error (TSA‐adjusted confidence interval −2.54 to −1.02) (Figure 8 in Appendix 4). Inspection of the funnel plot (Figure 9 in Appendix 4) suggested no potential bias (asymmetry), and we found no evidence of possible publication bias: Egger’s regression intercept (bias) −0.915 (two tailed, P = 0.259).
Psychological placebos versus wait‐list or no‐treatment (dichotomous data)
No data were applicable; Analysis 9.2 .
9.2. Analysis.

Comparison 9: Psychological placebos versus wait‐list or no‐treatment for people with mental health disorders, Outcome 2: Psychological placebos compared with wait‐list/no‐treatment for dichotomous data
Outcomes measuring benefits for pharmacological placebos versus wait‐list or no‐treatment
Twenty‐three trials compared pharmacological placebos versus wait‐list or no‐treatment. Six trials did not report usable data (Table 8). Nine trials reported continuous data and eight trials reported dichotomous data.
Pharmacological placebos versus wait‐list or no‐treatment (continuous data)
We found no differences for beneficial effect comparing pharmacological placebos versus wait‐list or no‐treatment (SMD −0.14, 95% CI −0.39 to 0.11; 9 trials, 279 participants; P = 0.28; I 2 = 0%; very low‐quality evidence; Analysis 10.1) No differences were identified between subgroups: pharmacological placebos versus wait‐list (SMD −0.51, 95% CI −1.41 to 0.38; 1 trial, 20 participants; P = 0.26; I 2 = not applicable), and pharmacological placebos versus no‐treatment (SMD −0.11, 95% CI −0.37 to 0.16; 8 trials, 259 participants; P = 0.43; I 2 = 0%). Test for subgroup differences: Chi 2 = 0.73, df = 1, P = 0.39; I 2 = 0%.
10.1. Analysis.

Comparison 10: Pharmacological placebos versus wait‐list or no‐treatment for people with mental health disorders, Outcome 1: Pharmacological placebos compared with wait‐list/no‐treatment for continuous data
The TSA shows that the cumulated Z curve enters the futility area, and therefore the anticipated intervention effect can be rejected (TSA adjusted confidence interval −9.43 to 6.15) (Figure 10 in Appendix 4). Inspection of the funnel plot was not possible due to insufficient data. We found no evidence of possible publication bias: Egger’s regression intercept (bias) −1.192 (two tailed, P = 0.408).
Pharmacological placebos versus wait‐list or no‐treatment (dichotomous data)
We found no differences for beneficial effect comparing pharmacological placebos versus wait‐list or no‐treatment (RR 1.05, 95% CI 0.74 to 1.48; 8 trials, 366 participants; P = 0.79; I 2 = 58%; very low‐quality evidence; Analysis 10.2 ). Test for subgroup differences: not applicable.
10.2. Analysis.

Comparison 10: Pharmacological placebos versus wait‐list or no‐treatment for people with mental health disorders, Outcome 2: Pharmacological placebos compared with no‐treatment for continuous data
Outcomes measuring benefits for physical placebos versus wait‐list or no‐treatment
17 trials compared physical placebos versus wait‐list or no‐treatment. One of these did not report usable data (Table 5 in Appendix 4).
Physical placebos compared with wait‐list or no‐treatment for continuous data
Physical placebos had a beneficial effect compared with wait‐list or no‐treatment (SMD −0.21, 95% CI −0.35 to −0.08; 17 trials, 896 participants; P = 0.002; I 2 = 0%; low‐quality evidence; Analysis 11.1). No differences were found between subgroups: physical placebos versus wait‐list (SMD −0.30, 95% CI −0.54 to −0.06; 7 trials, 669 participants; P = 0.02; I 2 = 37%), and physical placebos versus no‐treatment (SMD −0.15, 95% CI −0.42 to 0.11; 10 trials, 227 participants; P = 0.26; I 2 = 0%). Test for subgroup differences: Chi 2 = 0.64, df = 1, P = 0.42, I 2 = 0%.
11.1. Analysis.

Comparison 11: Physical placebo versus wait‐list or no‐treatment for people with mental health disorders, Outcome 1: Physical placebos compared with wait‐list/no‐treatment for continuous data
The TSA showed that the RIS was reached (n = 194), and that there was no risk of type 1 error (TSA‐adjusted confidence interval −3.64 to −0.49) (Figure 11 in Appendix 4). Inspection of the funnel plot (Figure 12 in Appendix 4) suggested no potential bias (asymmetry), and we found no evidence of possible publication bias: Egger’s regression intercept (bias) −0.078 (two tailed, P = 0.860).
Subgroup analyses
We found significant subgroup differences between using wait‐list or no‐treatment as comparators in the analyses on all placebos and psychological placebos (Appendix 5). We found larger differences in favour of all placebos or psychological placebos when comparing them with wait‐list rather than no‐treatment.
For specific mental health disorders: on all placebos versus wait‐list or no‐treatment, we found differences in favour of placebos for sleep‐wake disorders (SMD −0.34, 95% CI −0.60 to −0.07, I 2 = 0%), depression (SMD −0.42, 95% CI −0.78 to −0.05, I 2 = 51%), post‐traumatic stress disorder (SMD −0.54, 95% CI −1.06 to −0.02, I 2 = 74%), and anxiety disorders (SMD −0.57, 95% CI −0.93 to −0.21, I 2 = 66%) . However, sparse data in these analyses made the results imprecise. Psychological placebos showed a beneficial effect for patients with sleep‐wake disorders (SMD −0.44, 95% CI −0.76 to −0.12, I 2 = 0%) and for patients with post‐traumatic stress disorder (SMD −0.75, 95% CI −1.23 to −0.27, I 2 = 55%) versus wait‐list or no‐treatment.
In the other subgroup analyses, we only found significant differences in the analyses comparing non‐blinded observer‐reported outcomes with blinded observer‐rated and patient‐reported outcomes. We also conducted a post‐hoc subgroup analysis on types of psychological placebos: those with an interactional component (e.g. talking to a counsellor in a non‐directive manner), psychological placebos with a psychoeducational component and those with an exposure element. For this subgroup analysis, we found that interactional placebos yielded significantly higher effects than the other two types of psychological placebo.
There were no significant differences in the other subgroup analyses (see Appendix 5 for all the estimates of the subgroup analyses).
Sensitivity analyses
Due to a lack of sufficient data, it was not possible to conduct some of our predefined sensitivity analyses (see Table 9). We used both the fixed‐effect and the random‐effects models in all meta‐analyses. Statistical significance did not change when we applied a fixed‐effect model to analyses regarding all placebos, psychological placebos, pharmacological placebos and physical placebos. However, the statistical significance did change for usual care versus no‐treatment or wait‐list (Analysis 25.1). We therefore report the results of the random‐effects model for placebo interventions versus wait‐list or no treatment, and both the random‐effects and fixed‐effect models for usual care versus wait‐list or no treatment.
6. Differences from protocol and review.
| Section | Protocol | Review |
| Pooling of placebos | We planned to pool psychological, pharmacological, and physical placebos into one placebo for the first primary outcome. | We pooled psychological, pharmacological, and physical placebos into a group called 'all placebos' for all outcomes. This was done to increase the power of the analysis regarding adverse events and specific mental health diagnosis. |
| Dealing with missing data | We planned to contact study authors for relevant missing data on our prImary and secondary outcomes. If the authors dId not respond after two attempts to contact them, we planned to stop communications. If we are not able to obtain missing data, we will use the available data (incomplete data) in the analyses. If data are not reported in a usable way, we will consult a statistician to explore its transformation. | We contacted study authors for relevant missing data. However, we made a pragmatic decision not to contact authors from studies before 1990. This was due to lack of probability that these data would have been preserved. In trials that did not report standard deviation (SD), we created an artificially SD from trials with the same population, outcome, experimental and control intervention. Furthermore, if trials did not report the amount of participants included in each group, and the total amount of participant could be equally divided, we expected the groups to have the same amount of participants in each group. However, if the number could not be equally divided, we anticipated that the active interventions and active control interventions included more. |
| Subgroup analysis and investigation of heterogeneity | We planned to conduct subgroup analyses to make hypotheses about the subgroups mentioned below.
|
We added four post‐hoc subgroup analyses:
|
| Sensitivity analysis | Studies contributing to heterogeneity (‘outliers’) was planned to be removed to evaluate the impact of their statistical heterogeneity on the overall pooled effect estimate. We will remove outliers one by one and assess the impact on the overall outcome. We will conduct sensitivity analyses to determine whether findings are sensitive to the following decisions made during the review process.
|
We were only able to perform the analyses 2, 3, 4, and 5 due to a lack of sufficient data. Furthermore, we included a test for imprecision, as assessed by GRADE, by conducting Trial Sequential Analysis (TSA) on our primary outcomes included in the Table 3 . We also included these two sensitivity analyses:
|
| Meta‐regression | It was planned that we would conduct supplementary meta‐regression analyses on continuous outcome, based on the findings from the subgroup analyses. We planned to choose covariates based on relevant subgroup analyses, such as type of intervention, risk of bias, type of outcome domain, mean participant age and duration of interventions. | Due to a lack of data, meta‐regression analyses were not possible to conduct. |
| Selections of studies (participants) | All patients in each included trial was required to have a formal diagnosis of a mental health disorder, according to the Diagnostic and Statistical Manual of Mental Disorders (DSM), First Edition (DSM‐I;APA 1952), SecondEdition (DSM‐II;APA 1968),Third Edition (DSM‐III; APA 1980), Third Edition Revised (DSM‐IIIR; APA 1987), Fourth Edition (DSM‐IV; APA 1994), Fourth Edition Text Revision (DSM‐IV‐TR; APA 2000), and Fifth Edition (DSM‐5; APA 2013), or according to the International Classification ofDiseases andRelatedHealth Problems (ICD), Sixth Edition (ICD‐6; WHO 1949), Seventh Edition (ICD‐7; WHO 1955), Eighth Edition (ICD‐8; WHO 1967), Ninth Edition (ICD‐9; WHO 1975), 10th Edition (ICD‐10; WHO 1993), or 11th Edition (ICD‐11; WHO 2018). | During the inclusion phase we identified trials, where patients fulfilled the symptoms of the disorder according to ICD or DSM, but where the trial did not explicitly state a classification system. Moreover, trials were also identified where the populations was classified as having a mental health disorder, but full diagnostic symptoms were not reported. We chose to include these in our analyses, and included a post‐hoc subgroup analysis testing the difference |
| Decision hierarchy of outcomes | We used the following decision hierarchy to select the outcomes measuring effect.
|
During the extraction phase, we had to extend our decision hierarchy due to the fact that the pre‐defined hierarchy proved not to be sufficient. We added:
|
We also tested if different type of data collection (e.g. measures of adverse events) impacted our results and found no differences. For another sensitivity analysis, for outcomes at the end of intervention, we removed change scores to see if it affected the results. No statistical significant differences were detected.
We performed Trial Sequential Analysis (TSA) on all relevant primary outcomes included in Table 2 and Table 3 . The required information size (RIS) was reached for all placebos, psychological, and physical placebos compared with wait‐list and no‐treatment. It was not reached for pharmacological placebos, where the cumulated Z curve entered the futility area, and therefore the anticipated intervention effect could be rejected. The RIS was also not reached for usual care compared with wait‐list or no‐treatment. It was not possible to calculate TSA on serious adverse events and non‐serious adverse events because of insufficient information. We also tested whether removing the trials named by trial authors as wait‐list or no‐treatment but fitting our criteria for no‐treatment or wait‐list respectively would change the subgroup analysis between these two groups and found no significant differences.
Summary of findings tables
We did not assess the quality of evidence and report effect estimates in Table 1 because it only included one study. In the second table, on usual care versus no‐treatment or wait‐list, we rated the quality of evidence as low using GRADE (Table 2). In the third table on placebo interventions, we included six comparisons and the quality of evidence was rated low to very low (Table 3).
Discussion
Summary of main results
This review includes 96 randomised trials, out of which 83 trials provided usable data (3614/4200 participants or 86%). The trials included 15 different mental health disorders. We only found one trial that compared wait‐list versus no‐treatment directly and the authors were not able to provide usable data for this comparison. The comparison on usual care versus wait‐list or no‐treatment was not significant with an standardised mean difference (SMD) of ‐0.33 (95% confidence interval (CI) ‐0.83 to 0.16, I² = 86%, 5 trials, 523 participants), although a sensitivity analysis showed significant differences when using a fixed‐effect instead of a random‐effect model with an SMD of ‐0.46 (95% CI ‐0.64 to ‐0.28). We found significant differences between all placebo interventions combined versus wait‐list and no‐treatment with an SMD of ‐0.37 (95% CI ‐0.49 to ‐0.25, I²= 41%, 65 trials, 2446 participants), but there was evidence of some asymmetry in the funnel plot and almost all the trials were small. We found a moderate effect in favour of psychological placebos (SMD ‐0.49, 95% CI ‐0.64 to ‐0.30, I² = 53%, 39 trials, 1656 participants) and small effects in favour of pharmacological placebos (SMD ‐0.14, 95% CI ‐0.39 to 0.11, I² = 0%, 9 trials, 279 participants) and physical placebos (SMD ‐0.21, 95% CI ‐0.35 to ‐0.08, I² = 0%, 17 trials, 896 participants). There were significant differences in favour of all placebos in the comparisons on specific mental disorders, but the analyses suffered from sparse data. No differences were found on harms in any of the analyses.
The present systematic review has many strengths. We developed a protocol for this review (Faltinsen 2019) in accordance with the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2019 ). We conducted extensive searches in relevant databases, with no restrictions to language, publication year, or publication type. Two independent review authors selected trials, extracted data, assessed the risk of bias, and graded the quality of the evidence. Disagreements were resolved by discussion. We used Trial Sequential Analysis (TSA) to estimate the required Information Size (RIS) needed to either accept or reject a certain intervention effect. Another strength of the review is the large number of included trials and the fact that we could make direct comparisons between the different control interventions. This enabled a comprehensive assessment of the effect of the included controls.
The results of this review are affected by the statistical heterogeneity in the analyses, which may be due in part to methodological and clinical heterogeneity in variables such as the included mental health populations, outcome domains, and the contents of the control interventions. To investigate some of the heterogeneity stemming from the pooled mental health populations in the first primary outcome, we conducted comparisons on specific mental health disorders. However, out of the 15 different identified diagnoses across the included trials, we were only able to run comparisons on seven mental health disorders, and these analyses were limited by sparse data, which made the results imprecise. The majority of the review’s meta‐analyses are therefore conducted across all included mental disorders.
Another example of methodological heterogeneity is the large variability in the contents of the included psychological placebos, which is a much‐discussed issue in the literature on mental health control interventions (Comer 2013; Kazdin 2015; Borkovec 2005; Hróbjartsson 2012). It is in part difficult to properly design a psychological placebo because of the issue of targeting hypothesised specific factors of complex psychological treatments Borkovec 2005; Hróbjartsson 2012; Mohr 2014). Out of the three types of placebo interventions, psychological placebos showed the largest reported difference compared with wait‐list and no‐treatment for all included mental health populations (SMD ‐0.49, 95% CI ‐0.65 to ‐0.30). In an attempt to further investigate the methodological heterogeneity within this control intervention, we conducted a post‐hoc subgroup analysis on psychological placebos and divided them into three separate groups: interactional, psychological placebos with a psychoeducational component, and those with an exposure element (Analysis 17.8). The term interactional placebo referred in this case to psychological placebos that control for human interaction variables in treatments such as psychotherapy. We found that interactional placebos yielded significantly higher effects than the other two groups, which may indicate that control interventions that involve a human interaction element yield higher effect sizes compared to no‐treatment or wait‐list. This is in line with previous frameworks on the shared factors of psychological treatments (Wampold 2010; Hafliðadóttir 2021), although it is not possible to draw strong conclusions on this matter with this exploratory subgroup analysis. Rather, it points to the fact that the psychological placebos as an intervention are methodologically heterogeneous in their contents.
17.8. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 8: Type of psychological placebo
Overall completeness and applicability of evidence
We were able to include data from 83 of 96 trials or 3614 participants out of 4200 in total (86% of the total participant pool). In order to include a sufficient number of trials and give a global estimate of efficacy, we combined different outcomes across the included trials. More specifically, one outcome was chosen from each trial based on our predetermined outcome hierarchy. We included 66 different outcomes, which is a source of methodological heterogeneity (Higgins 2019). When we looked at specific mental health disorders, the outcome measures were often more similar. For instance, for depression, the outcome was often a depression inventory to rate symptoms (see Table 6).
Quality of the evidence
We assessed all included trials as high risk of bias, partly due to lack of blinding of participants and personnel. It is not possible to maintain blinding when comparing a control intervention where participants receive some form of treatment with a control intervention where no treatment is provided (wait‐list or no‐treatment). This is because the participants and often the personnel will know what treatment is provided to whom, which makes the results prone to bias and systematic errors (Higgins 2019). However, the trials also suffered from other forms of risk of bias. In fact, only three trials (Allen 2006; Ehlers 2014; Kwan 2017) would have been rated at low risk of bias if the blinding of the participants and personnel rating were excluded from the risk of bias assessment. Blinding of participants and personnel is a persistent issue in randomised trials with psychosocial interventions (Guidi 2018; Juul 2020) and there is a need to consider how to address the specific methodological challenges relevant to these types of trials (Guidi 2018; Munder 2018). We also conducted a post‐hoc subgroup analysis and found no differences between the trials at low, unclear and high risk of bias, when we removed the blinding of participants and personnel from the assessment (Analysis 16.18 , test for subgroup differences P = 0.26, I 2 = 26%). However, this analysis was exploratory and many of the trials provided insufficient information on the bias domains, which makes it difficult to judge the overall true extent of systematic bias in the included studies. The reader should take these factors into consideration when evaluating the review's risk of bias profile and its impact on the quality of the evidence.
16.18. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 18: Risk of bias (participants and personnel excluded)
We intended for this review to provide support for an empirical and methodological reflection of the benefits and harms of control interventions in mental health intervention research. Realising that the subject matter is complex, one may look at the low to very low quality of evidence as a reflection of the state of control intervention design rather than a criticism of the interpretability of our review. Although the included trials were rated as low to very low quality of evidence, this is a reflection of the heterogeneous objectives of this review and to some extent the fact that blinding of participants is not possible with the included designs. As a consequence, we chose to report an alternative risk of bias profile where we excluded the blinding domains (see Figure 5; Figure 6 ) in addition to the conventional risk of bias assessment. However, this did not change the overall quality of evidence.
5.
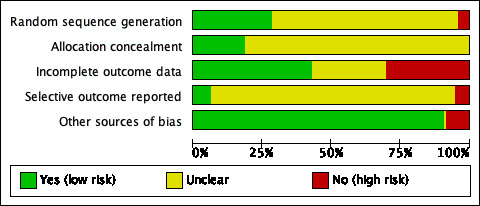
Alternative Risk of bias graph
6.

Alternative Risk of bias summary
Many trials had small sample sizes which led to imprecise estimates. The funnel plot for the comparison on all placebos was somewhat asymmetrical, which may reflect poor methodological quality, true heterogeneity, or selection bias (Higgins 2019). Upon visual inspection, the funnel plot indicated that some data points might be missing on the lower right corner of the plot, and this could have important implications for the interpretability of the data. The results in this comparison may be sensitive to trial size and should be interpreted with caution.
Almost all the included trials had three arms (interventions) with one experimental arm. Potential conflicts of interest when the researches had vested interests in the experimental intervention, financially or non‐financially, may have produced bias and threatened the validity of the results (Lundh 2017; Boutron 2021). We conducted a post‐hoc analysis to test conflicts of interest, but found no significant differences, but the data in the subgroup analysis were sparse. We should therefore not rule out the possibility of conflicts of interest towards the experimental intervention in some of the trials having impacted effect estimates. We graded all the overall results as either very low or low quality of evidence according to GRADE based on risk of bias, inconsistency of the evidence, and imprecision (Guyatt 2011a).
There are also many potential issues with reporting in the included trials. For instance, usual care, wait‐list and no‐treatment can vary in their contents and trial authors do not always specify how their control interventions were designed (Cuijpers 2021; Watts 2015), which makes it hard to determine how much overlap there is between the controls in the review. This should not, however, be an argument against the use of controls such as psychological placebos in trials with mental health populations because they may be methodologically useful for differentiating the active and non‐active factors in psychological treatments (Mohr 2009). Instead, controls such as psychological placebos should arguably be designed to control for everything but the hypothesised mechanism of causality in a psychological treatment (Hróbjartsson 2012; Locher 2018). Another potential issue is with the diagnostic classification systems used in the included studies, as some studies used older versions of the Diagnostic and Statistical Manual of Mental Disorders (DSM), which are not necessarily comparable to current versions.
Potential biases in the review process
Because of the large amount of records to screen, we chose to single‐screen records. This may have affected the selection of results and produced bias. It is very challenging to locate accurately all relevant records with such as broad search strategy (two control interventions in a three‐arm randomised trial with any mental health disorder), and we may have missed relevant trials. However, for the placebo interventions, we did include 50 more trials for mental health disorders than Hróbjartsson 2010 reported in an earlier review on placebos for all medical conditions.
During the inclusion phase, we identified trials where patients fulfilled the symptoms of a mental health disorder according to International Classification of Diseases (ICD) or DSM, but where the trial did not explicitly state what classification system was used. In some instances we also included trials where the population was classified as having a mental health disorder but the full diagnostic symptoms were not reported. We chose to include these trials in our analyses, and included a post‐hoc subgroup analysis to investigate potential differences.
There were some more minor changes from the protocol to the final report and these are all listed in Differences between protocol and review and Table 9.
Agreements and disagreements with other studies or reviews
The earlier reviews by Hróbjartsson and Gøtzche (Hróbjartsson 2001; Hróbjartsson 2002; Hróbjartsson 2004; Hróbjartsson 2010) found that the efficacy of their included placebo interventions yielded, on average, a small to moderate effect and that placebos may influence patient‐reported outcomes. Our analysis on all placebo types combined for continuous outcomes yielded an SMD of ‐0.37 (95% CI ‐0.49 to ‐0.25), which is not considerably different from the results found in Hróbjartsson and Gøtzsche's work (SMD ‐0.23, 95% CI ‐0.28 to ‐0.17) for all placebos combined on continuous outcomes. Both these effect estimates are small to moderate. They also found moderate statistical heterogeneity present in both the binary and continuous outcomes and funnel plot asymmetry in the analyses on continuous outcomes, which is similar to the findings in the present review.
We found some indication of funnel plot asymmetry in the comparison on all placebos, moderate heterogeneity in most analyses and high variability between effect sizes. In this review, psychological and physical placebos showed larger differences compared with no‐treatment or wait‐list than pharmacological placebos, which is also similar to what was found in Hróbjartsson 2010 . It could be true that placebo interventions yield larger differences compared with wait‐list or no‐treatment in randomised trials with mental health populations, perhaps because they involve more subjective outcome measures or that mental health disorders are more prone to be affected by placebo administration ( Weimer 2015 ). Our subgroup analyses for all placebos combined indicated that blinded and non‐blinded observer‐reported outcomes provided a higher placebo effect size compared with patient‐reported outcomes. Hróbjartsson 2010 found the opposite: a higher placebo effect for patient‐reported outcomes over observer‐reported. Here it should be noted that the observer‐reported outcomes in Hróbjartsson 2010 often measured a somatic variable, whereas observer‐reported outcomes in the present review were often psychometric instruments rated by an observer. Thus, the conflicting results from the reviews may not be directly comparable. Placebo and usual care control groups compared with wait‐list controls were found to yield higher effect estimates than compared with no‐treatment controls. The findings were only significant for all placebos combined and for psychological placebos. Our review found similarities to the work of Mohr and colleagues (Mohr 2014). In their meta‐analysis focusing on studies on depression, they found significant differences in effect sizes generated across different control interventions. Another recent network meta‐analysis that assessed control intervention's influence on effect estimates of active psychotherapies for depression found weaker effect estimates for wait‐list and no‐treatment than psychological and pharmacological placebos (Michopoulos 2021). Our review supports these findings and the importance of considering the type of control intervention in a randomised trial with mental health populations because it can drastically influence reported effect estimates. Cuijpers and Cristea has also proposed that to ensure a higher effect estimate of the active treatment in a randomised trial, a wait‐list intervention should be preferred (Cuijpers 2016). Wait‐list control interventions might bias the true effect of different active treatments and therefore potentially produce a skewed view of the effect of those treatments, but we are not able to make conclusions about this based on the results in this review.
Little research has been done on the harms of psychological treatments (Lilienfeld 2007; Linden 2014; Pagsberg 2017; Storebø 2018). We did not find indications of wait‐list or no‐treatment interventions being more harmful than any placebos. In the case of usual care, no trials reported or mentioned adverse events, and we are only able to give anecdotal evidence on adverse events here. Usual care is a highly heterogeneous control intervention and very few of our included trials accurately reported its contents because the researchers are often unaware of this themselves. Furukawa and colleagues previously speculated that wait‐list interventions could lead to negative effects in patients from waiting for an experimental treatment after the study period (Furukawa 2014), but our review was not able to confirm this because of sparse adverse event data. It should be a priority to identify whether wait‐list interventions might produce unfavourable harms in randomised trials with participants with mental health diagnoses in future research.
It has previously been argued that a decision framework should be put in place for how to properly choose a control intervention in trials with patients with mental health disorders, and that such a framework should take into account factors like trial phase, participation risk and available levels of resources (Gold 2018). It is evident from this review that the effect sizes in trials with patients with mental health disorders may vary widely depending on what control intervention is used, and it seems reasonable, therefore, to demand methodological standards for when it is appropriate to use a particular control in a trial. It may for instance be recommended that wait‐list or no‐treatment controls should only be used in the early stages of testing a new behavioural treatment, seeing that they often produce high effect sizes in favour of experimental interventions, which may give a misguided impression of the intervention’s effectiveness. Overall, it seems important that a control intervention should be properly designed and tailored to the specific objectives under investigation, and that there should be some agreement among researchers on when a type of control design is appropriate in a mental health trial and when it is not (Mohr 2009; Mohr 2014).
Authors' conclusions
Implication for systematic reviews and evaluations of healthcare.
The choice of a control intervention in randomised trials with patients with mental health disorders has a considerable impact on the reported estimate of benefits in published reports. When psychiatric interventions are compared with some kind of placebo intervention, the beneficial effect of the psychiatric intervention is lower than when compared with other control interventions. The difference in effect size for the experimental intervention might be approximately a standardised mean difference (SMD) value from 0.3 to 0.4 lower when using a placebo control intervention compared with wait‐list or no intervention.
Mental health systematic reviews and evaluations of healthcare should put equal emphasis on the reporting of the contents of control interventions, because they may have the same influence on effect size estimates as experimental interventions. This may be especially true for reviews dealing with psychosocial interventions, where the contents of control interventions such as usual care or psychological placebos are often underreported and unclear. People using reviews and evaluations should therefore be aware of how the choice of a control intervention (such as wait‐list controls or placebo controls) impacts on the reported effect estimates, both unfavourably and favourably. The evidence in the included trials in this review were rated as low to very low quality, only partly because of the inability to blind participants in randomised trials with a no‐treatment or wait‐list comparator. The issue of blinding may, however, be viewed as a methodological issue with the type of control design in mental health intervention research and not a flaw in the interpretability of this review itself.
The choice of a control intervention in a randomised trial in patients with mental health disorders has a considerable impact on the reported estimate of benefits in the published reports. Placebos or usual care tend to increase the differences compared with wait‐list or no‐treatment. Methodological guidelines need to be developed to reach a consensus on future standards for the design and reporting of control interventions in the field of mental health.
Implication for methodological research.
Currently, descriptions of both the experimental and control interventions are often poorly reported (Hoffmann 2013) and they need to be more adequately described (Guidi 2018). Methodological guidelines on how to properly report and design control interventions in randomised trials in mental health research are needed to advance the evidence base in the field. An adequate and systematised description of interventions would provide a platform for researchers to build on findings about control intervention design or replicate results (Hoffmann 2013; Tajika 2015). Control interventions should be developed to answer the specific research question at hand and should be chosen based on available resources, ethical concerns and the phase of research for a particular treatment (Gold 2018). Future research should support the development of a methodological guideline on how to properly design and report control interventions in randomised trials with patients with mental health disorders, to ensure the validity and reliability of future mental health trials.
History
Protocol first published: Issue 1, 2019
Acknowledgements
We thank the Cochrane Methodology Review Group's editorial team and peer reviewers. We thank Richard Kirubakaran for helping with the analysis regarding Egger's test. We also thank research librarian Trine Kæstel for her valuable guidance on the search strategy.
Appendices
Appendix 1. Search strategy
Database: Ovid MEDLINE(R) <1946 to March Week 5 2018 > Search Strategy:
exp mental disorders/ or exp anxiety disorders/ or exp agoraphobia/ or exp anxiety, separation/ or exp neurocirculatory asthenia/ or exp neurotic disorders/ or exp obsessive‐compulsive disorder/ or exp hoarding disorder/ or exp panic disorder/ or exp phobic disorders/ or exp phobia, social/ or exp "bipolar and related disorders"/ or exp bipolar disorder/ or exp "disruptive, impulse control, and conduct disorders"/ or exp firesetting behavior/ or exp gambling/ or exp trichotillomania/ or exp dissociative disorders/ or exp multiple personality disorder/ or exp elimination disorders/ or exp encopresis/ or exp enuresis/ or exp "feeding and eating disorders"/ or exp anorexia nervosa/ or exp binge‐eating disorder/ or exp bulimia nervosa/ or exp "feeding and eating disorders of childhood"/ or exp female athlete triad syndrome/ or exp food addiction/ or exp night eating syndrome/ or exp pica/ or exp mood disorders/ or exp depressive disorder/ or exp depression, postpartum/ or exp depressive disorder, major/ or exp depressive disorder, treatment‐resistant/ or exp dysthymic disorder/ or exp premenstrual dysphoric disorder/ or exp seasonal affective disorder/ or exp cyclothymic disorder/ or exp motor disorders/ or exp neurocognitive disorders/ or exp amnesia/ or exp alcoholic korsakoff syndrome/ or exp amnesia, anterograde/ or exp amnesia, retrograde/ or exp amnesia, transient global/ or exp cognition disorders/ or exp auditory perceptual disorders/ or exp huntington disease/ or exp cognitive dysfunction/ or exp consciousness disorders/ or exp delirium/ or exp emergence delirium/ or exp dementia/ or exp aids dementia complex/ or exp alzheimer disease/ or exp aphasia, primary progressive/ or exp primary progressive nonfluent aphasia/ or exp creutzfeldt‐jakob syndrome/ or exp dementia, vascular/ or exp dementia, multi‐infarct/ or exp diffuse neurofibrillary tangles with calcification/ or exp frontotemporal lobar degeneration/ or exp frontotemporal dementia/ or exp "pick disease of the brain"/ or exp kluver‐bucy syndrome/ or exp lewy body disease/ or exp dyslexia, acquired/ or exp alexia, pure/ or exp neurodevelopmental disorders/ or exp "attention deficit and disruptive behavior disorders"/ or exp attention deficit disorder with hyperactivity/ or exp conduct disorder/ or exp child behavior disorders/ or exp child development disorders, pervasive/ or exp autism spectrum disorder/ or exp asperger syndrome/ or exp autistic disorder/ or exp communication disorders/ or exp childhood‐onset fluency disorder/ or exp social communication disorder/ or exp speech sound disorder/ or exp developmental disabilities/ or exp intellectual disability/ or exp learning disorders/ or exp dyscalculia/ or exp dyslexia/ or exp specific learning disorder/ or exp motor skills disorders/ or exp mutism/ or exp reactive attachment disorder/ or exp schizophrenia, childhood/ or exp stereotypic movement disorder/ or exp tic disorders/ or exp paraphilic disorders/ or exp exhibitionism/ or exp "fetishism (psychiatric)"/ or exp masochism/ or exp pedophilia/ or exp sadism/ or exp transvestism/ or exp voyeurism/ or exp personality disorders/ or exp antisocial personality disorder/ or exp borderline personality disorder/ or exp compulsive personality disorder/ or exp dependent personality disorder/ or exp histrionic personality disorder/ or exp hysteria/ or exp paranoid personality disorder/ or exp passive‐aggressive personality disorder/ or exp schizoid personality disorder/ or exp schizotypal personality disorder/ or exp "schizophrenia spectrum and other psychotic disorders"/ or exp affective disorders, psychotic/ or exp capgras syndrome/ or exp delusional parasitosis/ or exp morgellons disease/ or exp paranoid disorders/ or exp psychotic disorders/ or exp psychoses, substance‐induced/ or exp schizophrenia/ or exp schizophrenia, catatonic/ or exp schizophrenia, disorganized/ or exp schizophrenia, paranoid/ or exp shared paranoid disorder/ or exp sexual dysfunctions, psychological/ or exp dyspareunia/ or exp erectile dysfunction/ or exp gender dysphoria/ or exp premature ejaculation/ or exp "sexual and gender disorders"/ or exp vaginismus/ or exp sleep wake disorders/ or exp dyssomnias/ or exp sleep deprivation/ or exp sleep disorders, circadian rhythm/ or exp sleep disorders, intrinsic/ or exp parasomnias/ or exp nocturnal paroxysmal dystonia/ or exp rem sleep parasomnias/ or exp restless legs syndrome/ or exp sleep arousal disorders/ or exp sleep bruxism/ or exp sleep‐wake transition disorders/ or exp somatoform disorders/ or exp body dysmorphic disorders/ or exp conversion disorder/ or exp factitious disorders/ or exp munchausen syndrome/ or exp munchausen syndrome by proxy/ or exp hypochondriasis/ or exp neurasthenia/ or exp substance‐related disorders/ or exp alcohol‐related disorders/ or exp alcohol amnestic disorder/ or exp alcohol withdrawal delirium/ or exp alcoholic intoxication/ or exp alcoholism/ or exp binge drinking/ or exp psychoses, alcoholic/ or exp wernicke encephalopathy/ or exp amphetamine‐related disorders/ or exp cocaine‐related disorders/ or exp inhalant abuse/ or exp marijuana abuse/ or exp "marijuana use"/ or exp neonatal abstinence syndrome/ or exp opioid‐related disorders/ or exp morphine dependence/ or exp opium dependence/ or exp phencyclidine abuse/ or exp substance abuse, intravenous/ or exp substance abuse, oral/ or exp substance withdrawal syndrome/ or exp "tobacco use disorder"/ or exp "trauma and stressor related disorders"/ or exp adjustment disorders/ or exp stress disorders, traumatic/ or exp battered child syndrome/ or exp combat disorders/ or exp psychological trauma/ or exp stress disorders, post‐traumatic/ or exp stress disorders, traumatic, acute/
exp PLACEBO EFFECT/ or exp Placebos/
(control* or compar* or nonspecific or non‐specific or un‐specific or unspecific or vehicle* or placebo* or credible or pseudo or sham or mock or fake or dumm* or attention or "common factor*").ab,hw,kf,ti.
(usual or clinic* or standard* or enhanc* or routine or outpatient* or convention* or gener* or local* or structur* or manual* or optim*).ab,hw,kf,ti.
(care or practi* or management* or treat* or intervention* or contact* or pill* or tablet* or medic* or therap* or surger* or operat*).ab,hw,kf,ti.
4 and 5
TAU.ab,hw,kf,ti.
exp Waiting Lists/
("no* care" or "no* practi*" or "no* management*" or "no* treat*" or "no* intervention*" or "no* contact*" or "no* pill*" or "no* tablet*" or "no* medic*" or "no* therap*" or "no* surger*" or "no* operat*" or "no* active*" or "no* experimental*").ab,hw,kf,ti.
(no*care or no*practi* or no*management* or no*treat* or no*intervention* or no*contact* or no*pill* or no*tablet* or no*medic* or no*therap* or no*surger* or no*operat* or no*active* or no*experimental*).ab,hw,kf,ti.
(uncare or unpracti* or unmanagement* or untreat* or unintervention* or uncontact* or unmedic* or untherap* or unsurger* or unoperat*or unactive* or unexperimental*).ab,hw,kf,ti.
("un care" or "un practi*" or "un management*" or "un treat*" or "un intervention*" or "un contact*" or "un medic*" or "un therap*" or "un surger*" or "un operat*" or "un active*" or "un experimental*").ab,hw,kf,ti.
("minim* care" or "minim* practi*" or "minim* management*" or "minim* treat*" or "minim* intervention*" or "minim* contact*" or "minim* medic*" or "minim* therap*" or "minim* surger*" or "minim* operat*" or "minim* active*" or "minim* experimental*" or "minim* period*" or "minim* time*").ab,hw,kf,ti.
("without care" or "without practi*" or "without management*" or "without treat*" or "without intervention*" or "without contact*" or "without pill*" or "without tablet*" or "without medic*" or "without therap*" or "without surger*" or "without operat*" or "without active*" or "without experimental*").ab,hw,kf,ti.
("delay* care" or "delay* practi*" or "delay* management*" or "delay* treat*" or "delay* intervention*" or "delay* contact*" or "delay* pill*" or "delay* tablet*" or "delay* medic*" or "delay* therap*" or "delay* surger*" or "delay* operat*" or "delay* active*" or "delay* experimental*" or "delay* list*" or "delay* period*" or "delay* time").ab,hw,kf,ti.
(await* or wait*).ab,hw,kf,ti.
randomi#ed controlled trial.pt.
controlled clinical trial.pt.
randomi#ed.ab.
"placebo* ".ab.
drug therapy.fs
randomly.ab.
trial.ab.
groups.ab.
exp Animals/
Humans/
25 not 26
17 or 18 or 19 or 20 or 21 or 22 or 23 or 24
28 not 27
8 or 9 or 10 or 11 or 12 or 13 or 14 or 15 or 16
2 or 3 or 6
1 and 29 and 30 and 31
Database: PsycINFO <1806 to April Week 1 2018> Search Strategy:
exp mental disorders/ or exp adjustment disorders/ or exp affective disorders/ or exp alexithymia/ or exp anxiety disorders/ or exp autism spectrum disorders/ or exp chronic mental illness/ or exp dementia/ or exp dissociative disorders/ or exp eating disorders/ or exp elective mutism/ or exp factitious disorders/ or exp gender identity disorder/ or exp hoarding disorder/ or exp hysteria/ or exp impulse control disorders/ or exp neurosis/ or exp paraphilias/ or exp personality disorders/ or exp pseudodementia/ or exp psychosis/ or exp schizoaffective disorder/ or exp abnormal psychology/ or exp adaptive behavior/ or exp attention deficit disorder/ or exp attention deficit disorder with hyperactivity/ or exp behavior disorders/ or exp borderline states/ or exp brain disorders/ or exp communication disorders/ or exp conduct disorder/ or exp consciousness disturbances/ or exp emotional disturbances/ or exp infantilism/ or exp intellectual development disorder/ or exp learning disorders/ or exp narcissism/ or exp personality processes/ or exp psychiatric patients/ or exp psychiatric symptoms/ or exp psychodiagnosis/ or exp psychopathology/ or exp sexual function disturbances/ or exp sleep disorders/
exp PLACEBO/
exp OUTPATIENTS/
(control* or compar* or nonspecific or non‐specific or un‐specific or unspecific or vehicle* or placebo* or fals* or credible or pseudo or sham or mock or fake or dumm* or neutral or attention or "common factor*").ab,hw,id,ti.
(usual or clinic* or standard* or enhanc* or routine or outpatient* or convention* or gener* or local* or structur* or manual* or optim*).ab,hw,id,ti.
(care or practi* or management* or treat* or intervention* or contact* or pill* or tablet* or medic* or therap* or surger* or operat* or period* or time).ab,hw,id,ti.
5 and 6
TAU.ab,hw,id,ti.
exp Experiment Controls/
("no* care" or "no* practi*" or "no* management*" or "no* treat*" or "no* intervention*" or "no* contact*" or "no* pill*" or "no* tablet*" or "no* medic*" or "no* therap*" or "no* surger*" or "no* operat*" or "no* active*" or "no* experimental*").ab,hw,id,ti.
(no*care or no*practi* or no*management* or no*treat* or no*intervention* or no*contact* or no*pill* or no*tablet* or no*medic* or no*therap* or no*surger* or no*operat* or no*active* or no*experimental*).ab,hw,id,ti.
(uncare or unpracti* or unmanagement* or untreat* or unintervention* or uncontact* or unmedic* or untherap* or unsurger* or unoperat*or unactive* or unexperimental*).ab,hw,id,ti.
("un care" or "un practi*" or "un management*" or "un treat*" or "un intervention*" or "un contact*" or "un medic*" or "un therap*" or "un surger*" or "un operat*" or "un active*" or "un experimental*").ab,hw,id,ti.
("minim* care" or "minim* practi*" or "minim* management*" or "minim* treat*" or "minim* intervention*" or "minim* contact*" or "minim* medic*" or "minim* therap*" or "minim* surger*" or "minim* operat*" or "minim* active*" or"minim* experimental*" or "minim* period*" or "minim* time*").ab,hw,id,ti.
("without care" or "without practi*" or "without management*" or "without treat*" or "without intervention*" or "without contact*" or "without pill*" or "without tablet*" or "without medic*" or "without therap*" or "without surger*" or "without operat*" or "without active*" or "without experimental*").ab,hw,id,ti.
("delay* care" or "delay* practi*" or "delay* management*" or "delay* treat*" or "delay* intervention*" or "delay* contact*" or "delay* pill*" or "delay* tablet*" or "delay* medic*" or "delay* therap*" or "delay* surger*" or "delay* operat*" or "delay* active*" or "delay* experimental*" or "delay* list*" or "delay* period*" or "delay* time").ab,hw,id,ti.
(await* or wait*).ab,hw,id,ti.
exp Clinical Trials/
(random* adj allocat*).ab.
randomi?ed.ab.
placebo.ab.
"random* ".ab.
"trial* ".ab.
"group* ".ab.
drug therapy.sh.
exp Animals/ not Humans/
18 or 19 or 20 or 21 or 22 or 23 or 24 or 25
27 not 26
2 or 3 or 4 or 7 or 8
9 or 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17
1 and 28 and 29 and 30
Database: Embase <1974 to 2018 April 09> Search Strategy:
exp mental disease/ or exp addiction/ or exp adjustment disorder/ or exp alexithymia/or exp anxiety disorder/ or exp autism/ or exp behavior disorder/ or exp delirium/ or exp dissociative disorder/ or exp emotional disorder/ or exp learning disorder/ or exp memory disorder/ or exp mental deficiency/ or exp mental infantilism/ or exp mental instability/ or exp mood disorder/ or exp neurosis/ or exp organic brain syndrome/ or exp personality disorder/ or exp psychosexual disorder/ or exp psychosis/ or exp psychosomatic disorder/ or exp psychotrauma/ or exp schizophrenia spectrum disorder/ or exp stupor/ or exp thought disorder/
exp placebo effect/ or exp placebo/
exp outpatient care/
(control* or compar* or nonspecific or non‐specific or un‐specific or unspecific or vehicle* or placebo* or credible or pseudo or sham or mock or fake or dumm* or attention or "common factor*").ab,hw,kw,ti.
(usual or clinic* or standard* or enhanc* or routine or outpatient* or convention* or gener* or local* or structur* or manual* or optim*).ab,hw,kw,ti.
(care or practi* or management* or treat* or intervention* or contact* or pill* or tablet* or medic* or therap* or surger* or operat* or period* or time).ab,hw,kw,ti.
5 and 6
TAU.ab,hw,kw,ti.
exp control group/
("no* care" or "no* practi*" or "no* management*" or "no* treat*" or "no* intervention*" or "no* contact*" or "no* pill*" or "no* tablet*" or "no* medic*" or "no* therap*" or "no* surger*" or "no* operat*" or "no* active*" or "no* experimental*").ab,hw,kw,ti.
(no*care or no*practi* or no*management* or no*treat* or no*intervention* or no*contact* or no*pill* or no*tablet* or no*medic* or no*therap* or no*surger* or no*operat* or no*active* or no*experimental*).ab,hw,kw,ti.
(uncare or unpracti* or unmanagement* or untreat* or unintervention* or uncontact* or unmedic* or untherap* or unsurger* or unoperat*or unactive* or unexperimental*).ab,hw,kw,ti.
("un care" or "un practi*" or "un management*" or "un treat*" or "un intervention*" or "un contact*" or "un medic*" or "un therap*" or "un surger*" or "un operat*" or "un active*" or "un experimental*").ab,hw,kw,ti.
("minim* care" or "minim* practi*" or "minim* management*" or "minim* treat*" or "minim* intervention*" or "minim* contact*" or "minim* medic*" or "minim* therap*" or "minim* surger*" or "minim* operat*" or "minim* active*" or "minim* experimental*" or "minim* period*" or "minim* time*").ab,hw,kw,ti.
("without care" or "without practi*" or "without management*" or "without treat*" or "without intervention*" or "without contact*" or "without pill*" or "without tablet*" or "without medic*" or "without therap*" or "without surger*"or "without operat*" or "without active*" or "without experimental*").ab,hw,kw,ti.
("delay* care" or "delay* practi*" or "delay* management*" or "delay* treat*" or "delay* intervention*" or "delay* contact*" or "delay* pill*" or "delay* tablet*" or "delay* medic*" or "delay* therap*" or "delay* surger*" or "delay* operat*" or "delay* active*" or "delay* experimental*" or "delay* list*" or "delay* period*" or "delay* time").ab,hw,kw,ti.
(await* or wait*).ab,hw,kw,ti.
controlled clinical trial/ or exp clinical trial/ or exp controlled study/ or exp randomized controlled trial/
(random* adj allocat*).ab.
randomi?ed.ab.
placebo.ab.
"random* ".ab.
"trial* ".ab.
drug therapy.fs.
exp Animals/ not Humans/
18 or 19 or 20 or 21 or 22 or 23 or 24
26 not 25
2 or 3 or 4 or 7 or 8
9 or 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17
1 and 27 and 28 and 29
Database: Cochrane Central Register of Controlled Trials; current issue:
MeSH descriptor: [Mental Disorders] explode all trees
MeSH descriptor: [Placebos] explode all trees
MeSH descriptor: [Placebo Effect] explode all trees
(control* or compar* or nonspecific or non‐specific or un‐specific or unspecific or vehicle* or placebo* or credible or pseudo or sham or mock or fake or dumm* or attention or "common factor*"):ti,ab,kw (Word variations have been searched)
(usual or clinic* or standard* or enhanc* or routine or outpatient* or convention* or gener* or local* or structur* or manual* or optim*) next (care or practi* or management* or treat* or intervention* or contact* or pill* or tablet* or medic* or therap* or surger* or operat* or period* or time):ti,ab,kw (Word variations have been searched)
TAU:ti,ab,kw (Word variations have been searched)
MeSH descriptor: [Waiting Lists] explode all trees
"no care" or "non care" or "no treat*" or "non treat*" or "no pract*" or "non pract*" or "no intervention" or "non intervention" or "no management" or "non management" or "no pill*" or "non pill" or "no contact" or "non contact" or "no tablet" or "non tablet" or "no medication*" or "non medication" or "no therap*" or "non therap*" or "no surger*" or "non surger*" or "no operat*" or "non operat*" or "no active" or "non active" or "no experimental" or "non experimental":ti,ab,kw (Word variations have been searched)
un near (care or practi* or management* or treat* or intervention* or contact* or pill* or tablet* or medic* or therap* or surger* or operat* or active* or experimental*):ti,ab,kw (Word variations have been searched)
minim* near (care or practi* or management* or treat* or intervention* or contact* or pill* or tablet* or medic* or therap* or surger* or operat* or active* or experimental*):ti,ab,kw (Word variations have been searched)
without near (care or practi* or management* or treat* or intervention* or contact* or pill* or tablet* or medic* or therap* or surger* or operat* or active* or experimental*):ti,ab,kw (Word variations have been searched)
delay near (care or practi* or management* or treat* or intervention* or contact* or pill* or tablet* or medic* or therap* or surger* or operat* or active* or experimental*):ti,ab,kw (Word variations have been searched)
(await* or wait*):ti,ab,kw (Word variations have been searched)
#2 or #3 or #4 or #5 or #6
#7 or #8 or #9 or #10 or #11 or #12 or #13
#1 and #14 and #15 in Trials
Database ‐ AMED ‐ The Allied and Complementary Medicine Database:
Interface ‐ EBSCOhost Research Databases
Search Screen ‐ Advanced Search
Search modes ‐ Boolean/Phrase
TX Mental* OR psych* OR Anxi* OR Bipolar OR Conduct disorder* OR Dissociative OR Elimination Disorder* OR Eat* OR Mood* OR Motor Disorder* OR Neuro* OR Paraphilic OR Personality OR Schizophren* OR Sexual Dys* OR Sleep* OR Somatoform* OR Substance* OR Trauma
placebo
(control* or compar* or nonspecific or non‐specific or un‐specific or unspecific or vehicle* or placebo* or credible or pseudo or sham or mock or fake or dumm* or attention or "common factor*")
(usual or clinic* or standard* or enhanc* or routine or outpatient* or convention* or gener* or local* or structur* or manual* or optim*)
(care or practi* or management* or treat* or intervention* or contact* or pill* or tablet* or medic* or therap* or surger* or operat*)
(S4 AND S5)
TAU
("no* care" or "no* practi*" or "no* management*" or "no* treat*" or "no* intervention*" or "no* contact*" or "no* pill*" or "no* tablet*" or "no* medic*" or "no* therap*" or "no* surger*" or "no* operat*" or "no* active*" or "no* experimental*")
(no*care or no*practi* or no*management* or no*treat* or no*intervention* or no*contact* or no*pill* or no*tablet* or no*medic* or no*therap* or no*surger* or no*operat* or no*active* or no*experimental*)
(uncare or unpracti* or unmanagement* or untreat* or unintervention* or uncontact* or unmedic* or untherap* or unsurger* or unoperat*or unactive* or unexperimental*)
("un care" or "un practi*" or "un management*" or "un treat*" or "un intervention*" or "un contact*" or "un medic*" or "un therap*" or "un surger*" or "un operat*" or "un active*" or "un experimental*")
("minim* care" or "minim* practi*" or "minim* management*" or "minim* treat*" or "minim* intervention*" or "minim* contact*" or "minim* medic*" or "minim* therap*" or "minim* surger*" or "minim* operat*" or "minim* active*" or "minim* experimental*" or "minim* period*" or "minim* time*")
("without care" or "without practi*" or "without management*" or "without treat*" or "without intervention*" or "without contact*" or "without pill*" or "without tablet*" or "without medic*" or "without therap*" or "without surger*" or "without operat*" or "without active*" or "without experimental*")
("delay* care" or "delay* practi*" or "delay* management*" or "delay* treat*" or "delay* intervention*" or "delay* contact*" or "delay* pill*" or "delay* tablet*" or "delay* medic*" or "delay* therap*" or "delay* surger*" or "delay* operat*" or "delay* active*" or "delay* experimental*" or "delay* list*" or "delay* period*" or "delay* time")
(await* or wait*)
Randomi#ed or controlled trial* or clinical trial* or placebo* or random* or trial or groups
(S2 OR S6 OR S7)
(S8 OR S9 OR S10 OR S11 OR S12 OR S13 OR S14 OR S15)
S1 AND S16 AND S17 AND S18
Database: Web of Science; 1900 to current:
TOPIC: (Mental* OR psych* OR Anxi* OR Bipolar OR Conduct disorder* OR Dissociative OR Elimination Disorder* OR Eat* OR Mood* OR Motor Disorder* OR Neuro* OR Paraphilic OR Personality OR Schizophren* OR Sexual Dys* OR Sleep* OR Somatoform* OR Substance* OR Trauma*) Indexes=SCI‐EXPANDED, SSCI, A&HCI, CPCI‐S, CPCI‐SSH, ESCI Timespan=All years
TS=(Placebo or control or comparison or vehicle or false or credible or pseudo or sham or mock or dummy or neutral or "standard care" or "usual intervention" or "routine care" or TAU or "treatment as usual" or "usual care" or "standard care" or "standard intervention" or "enhanced care" or "convention* care") Indexes=SCI‐EXPANDED, SSCI, A&HCI, CPCI‐S, CPCI‐SSH, ESCI Timespan=All years
TS=(("waiting list" or wait* or await* or "no*intervention" or "no therapy" or "no*treatment" or "no*care" or "no care" or "no treatment" or "minim*treatment" or "minim*care" or "minim* therapy" or "without care" or "without treatment" or "without intervention" or "without therapy" or "delayed care" or "delayed treatment" or "delayed therapy" or "delayed intervention" indexes=SCI‐EXPANDED, SSCI, A&HCI, CPCI‐S, CPCI‐SSH, ESCI Timespan=All years
TI=(randomized or randomised or controlled trial* or clinical trial* or placebo* or drug therapy or random* or trial or groups Indexes=SCI‐EXPANDED, SSCI, A&HCI, CPCI‐S, CPCI‐SSH, ESCI Timespan=All years
#4 AND #3 AND #2 AND #1 Indexes=SCI‐EXPANDED, SSCI, A&HCI, CPCI‐S, CPCI‐SSH, ESCI Timespan=All years
Database: ProQuest Dissertations and Theses A&I; 1743 to current:
((Mental* OR psych* OR Anxi* OR Bipolar OR Conduct disorder* OR Dissociative OR Elimination Disorder* OR Eat* OR Mood* OR Motor Disorder* OR Neuro* OR paedophilic OR Personality OR Schizophren* OR Sexual Dys* OR Sleep* OR Somatoform* OR Substance* OR Trauma*)
AND (placebo OR control OR comparison OR vehicle OR false OR credible OR pseudo OR sham OR mock OR dummy OR neutral OR "standard care" OR "usual intervention" OR "routine care" OR TAU OR "treatment as usual" OR "usual care" OR "standard care" OR "standard intervention" OR "enhanced care" OR "convention* care")
AND ("waiting list" OR wait* OR await* OR "no*intervention" OR "no therapy" OR "no*treatment" OR "no*care" OR "no care" OR "no treatment" OR "minim*treatment" OR "minim*care" OR "minim* therapy" OR "without care" OR "without treatment" OR "without intervention" OR "without therapy" OR "delayed care" OR "delayed treatment" OR "delayed therapy" OR "delayed intervention"))
AND diskw.Exact("PLACEBO" OR "Random" OR "Clinical Trial" OR "Controlled
trials" OR "randomised controlled trial" OR "Randomized Controlled Trial")
Database: Sociological Abstracts ProQuest; 1952 to current:
(Mental* OR psych* OR Anxi* OR Bipolar OR Conduct disorder* OR Dissociative OR Elimination Disorder* OR Eat* OR Mood* OR Motor Disorder* OR Neuro* OR paedophilic OR Personality OR Schizophren* OR Sexual Dys* OR Sleep* OR Somatoform* OR Substance* OR Trauma*)
AND ab(placebo OR control OR comparison OR vehicle OR false OR credible OR pseudo OR sham OR mock OR dummy OR neutral OR "standard care" OR "usual intervention" OR "routine care" OR TAU OR "treatment as usual" OR "usual care" OR "standard care" OR "standard intervention" OR "enhanced care" OR "convention* care")
AND ab("waiting list" OR wait* OR await* OR "no*intervention" OR "no therapy" OR"no*treatment" OR "no*care" OR "no care" OR "no treatment" OR "minim*treatment" OR "minim*care" OR "minim* therapy" OR "without care" OR "without treatment" OR "without intervention" OR "without therapy" OR "delayed care" OR "delayed treatment" OR "delayed therapy" OR "delayed intervention")
AND ab("PLACEBO" OR "Random" OR "Clinical Trial" OR "Controlled trials" OR "randomised controlled trial" OR "Randomized Controlled Trial")
Database: Google Scholar; top 200 of relevance according toBramer 2017:
Mental│psychiatric│psychological │no treatment│waitlist│placebo│usual care │random │clinical trials
Database: BIOSIS Previews; 1969 to current:
TOPIC: (mental disorder*) DocType=All document types; Language=All languages;
TOPIC: ("waiting list" or wait* or await* or "no*intervention" or "no therapy" or "no*treatment" or "no*care" or "no care" or "no treatment" or "minim*treatment" or "minim*care" or "minim* therapy" or "without care" or "without treatment" or "without intervention" or "without therapy" or "delayed care" or "delayed treatment" or "delayed therapy" or "delayed intervention") DocType=All document types; Language=All languages;
TOPIC: (Placebo or control or comparison or vehicle or false or credible or pseudo or sham or mock or dummy or neutral or “standard care” or “usual intervention” or “routine care” or TAU or “treatment as usual” or “usual care” or “standard care” or “standard intervention” or “enhanced care” or “convention* care”)DocType=All document types; Language=All languages;
#3 AND #2 AND #1DocType=All document types; Language=All languages;
Database: Open Grey;1997 to current
(mental* OR psych*) AND (placebo OR usual care OR "treatment as usual" OR wait‐list OR wait list OR await* OR wait*) AND (no treatment OR wait‐list OR wait list OR await* OR wait*) AND (random*)
Trial registry: Australian New Zealand Clinical Trials Registry (ANZCTR);www.anzctr.org.au/BasicSearch.aspx
Search terms: (mental OR psychiatric) AND (placebo OR usual care OR waitlist) AND (no treatment OR waitlist)
Allocation to treatment: Randomised
Condition category: Mental health
Healthy Volunteers: No
Trial registry: Clinical Trials;clinicaltrials.gov
Condition category: Mental Disorder
Other terms: (placebo OR usual care OR wait‐list)
Intervention/treatment: No treatment
Trial registry: EU Clinical Trials Register;www.clinicaltrialsregister.eu/ctr-search/search
(Mental disorder OR psychiatric) AND (placebo OR usual care OR wait‐list) AND (no treatment OR wait‐list)
Trial registry: ISRCTN;www.isrctn.com
Search 1:
Condition category: Mental and behavioural disorders
Interventions: No treatment
Search 2:
Condition category: Mental and behavioural disorders
Interventions: Waitlist
Trial registry: UK Clinical Trials Gateway;www.ukctg.nihr.ac.uk/#popoverSearchDivId
(Mental disorder OR psychiatric) AND (placebo OR usual care OR wait‐list) AND (no treatment OR wait‐list)
Trial registry: World Health Organization International Clinical Trials Registry Platform (WHO ICTRP);http://apps.who.int/trialsearch/
Condition: (Mental disorder OR psychiatric)
Intervention: No treatment OR wait‐list
Appendix 2. Template for extraction sheet
| Source | Trial ID (Original trial, e.g. Plizska 2000) Full citationOther publications on same studyAuthor contact informationPublication type (e.g. full report, abstract, letter) Form filled by (date, name)Ethical approval |
| Eligibility | Confirm eligibility: yes/no/awaiting |
| Correspondence | Correspondence required: yes/no |
| Methods | Design (number of arms): Sample calculation: Cluster randomised: (yes/no): Duration of trial (baseline to post): Duration of participation (trial + follow‐up): Setting: Purpose of trial: Open or closed placebo: |
| Participants | Number of participants screened: Number of participants included: Number of participants followed‐up: Number of participants randomly assigned to:
Number of withdrawals:
Diagnosis: Diagnostic manual (DSM/ICD) Means of assessment: Comorbidity: Age: mean years (range) IQ: Sex: (male/female) Ethnicity: Country: Country of treatment (where did the treatment take place): Inclusion criteria Exclusion criteria |
|
Interventions Remember to state (if applicable): 1. whether pharmacological, psychological other not specified 2. whether individual or group psychological treat |
Control intervention Treatment name (type): Description of intervention: Individual or group treatment: Exposure/intensity to treatment: Duration of treatment: Concomitant psychotherapy: Concomitant pharmacotherapy: Comparator intervention Comparison name (type): Description of intervention: Exposure/intensity to treatment: Duration treatment: Concomitant psychotherapy: Concomitant pharmacotherapy: |
|
Outcomes Remember to state: 1. whether self‐rated or observer‐rated 2. Hierarchy |
Relevant outcomes for effect: Relevant outcomes for adverse events: |
| Notes | Key conclusion from study authors Key limitations from study authors Other notes from review authors |
|
Risk of bias 1. Support for judgement 2. Risk of bias (low, unclear, high) |
Random sequence generation Allocation concealment Blinding of outcome assessment Blinding of participants and personnel Incomplete outcome data Selective outcome reporting(Trial registry ID (search: clinicaltrials.gov (from 2008) and who.int/ictrp/en (from 2004) Other sources of bias:
|
Appendix 3. Cochrane’s Risk of bias tool 1.0
Random sequence generation
Low risk of bias: an adequate method for randomisation sequence generation was used (e.g. computer‐generated random numbers or a table of random numbers), or the method was unlikely to introduce selection bias.
Unclear risk of bias: there was insufficient information to determine whether the applied randomisation method could introduce selection bias.
High risk of bias: the method applied was likely to introduce selection bias.
Allocation concealment
Low risk of bias: the method to conceal intervention allocations (e.g. central allocation) was unlikely to bias the results.
Unclear risk of bias: there was insufficient information to determine whether the applied method could bias allocation to interventions.
High risk of bias: the method applied (e.g. open random allocation schedule) could have biased the allocations to interventions.
Blinding of participants and personnel
Low risk of bias: the method of blinding was sufficiently described and blinding was conducted in a satisfactory way.
Unclear risk of bias: there was insufficient information to determine whether adequate blinding was used and whether it was likely to bias the effect estimates.
High risk of bias: no blinding procedures were used or the blinding procedures were incomplete.
It is important to highlight that blinding of participants and personnel were not possible in the included trials, since participants would be aware if they received any kind of care or no care. Therefore, all of the trials would be rated as high risk of bias.
Blinding of outcome assessment
Low risk of bias: the method of blinding was described and blinding was conducted in a satisfactory way.
Unclear risk of bias: there was insufficient information to determine whether the type of blinding was likely to bias the effect estimates.
High risk of bias: no blinding used or incomplete blinding was used.
Incomplete outcome data
Low risk of bias: missing data did probably not affect the outcome measures, as all missing data can be considered as missing at random or all data were reported.
Unclear risk of bias: there was insufficient information to determine whether missing data, or the method used to handle missing data was likely to bias the effect estimates.
High risk of bias: the crude estimate of effects could have been biased given the attrition rates, the reasons for the missing data, or the insufficient methods used to handle missing data.
Selective outcome reporting
Low risk of bias: the trial protocol was available and all pre‐specified outcomes of interest were reported.
Unclear risk of bias: there was insufficient information to determine whether selective outcome reporting could have occurred.
High risk of bias: not all of the primary outcomes specified beforehand were reported or participants were excluded after randomisation.
Other sources of bias
Low risk of bias: the trial appeared to be free of other sources of bias.
Unclear risk of bias: there was insufficient information to determine the extent of other possible sources of bias.
High risk of bias: other sources of bias were identified.
Appendix 4. Trial Sequential Analysis (TSA) and Funnel Plot figures
The analysis on usual care compared with wait‐list and no‐treatment showed that the required information size was not reached. See Table 2. See Figure 4. We were not able to draw funnel plots for Usual care due to lack of data.
We performed a TSA on the primary outcomes all placebos, psychological placebos, pharmacological placebos, and pharmacological placebos included in the Table 3.
The analysis on all placebos compared with wait‐list and no‐treatment showed that the required information size was reached. See Figure 132. We drew a funnel plot for the comparison between all placebos and wait‐list and no‐treatment. The funnel plot shows no signs of asymmetry. See Figure 133.
7.
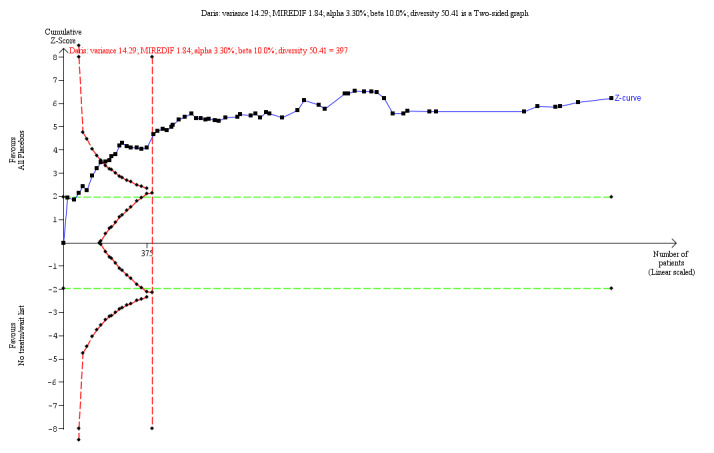
When comparing all placebos with wait‐list and no‐treatment on beneficial effects, we performed a trial sequential analysis on the primary outcome. The analysis shows that the required information size was reached. See Figure 5 above. MIREDIF: Minimum relevant difference
8.

Funnel plot of comparison: 6.1. Efficacy of all placebos compared with wait‐list/no‐treatment for continuous data.
We drew a funnel plot for the serious adverse events between all placebos and wait‐list and no‐treatment. It was not possible to perform a TSA for serious adverse events. The funnel plot shows no signs of asymmetry. See Figure 134.
9.

Funnel plot of comparison: 7.1 Serious adverse events of all placebos compared with wait‐list/no‐treatment for dichotomous data.
The analysis on psychological placebos compared with wait‐list and no‐treatment showed that the required information size was reached. See Figure 135. We drew a funnel plot for the comparison between all placebos and wait‐list and no‐treatment. The funnel plot shows no signs of asymmetry. See Figure 136.
10.
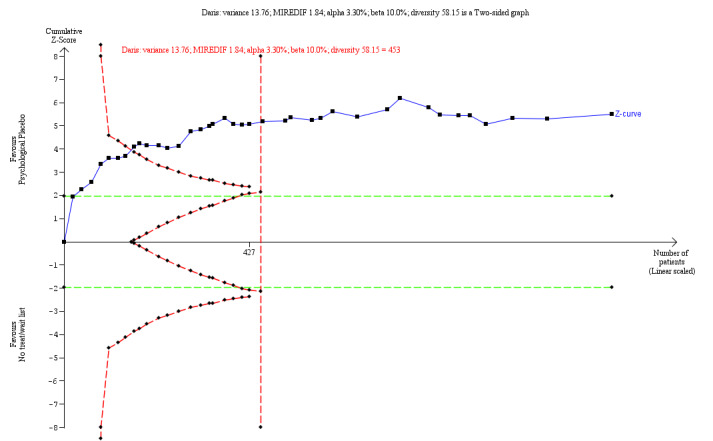
We performed a trial sequential analysis on the primary outcome for efficacy of psychological placebos compared with wait‐list and no‐treatment. The analysis shows that the required information size was reached. See Figure 8 above. MIREDIF: Minimum relevant difference
11.

Funnel plot of comparison: 9.1. Efficacy psychological placebos compared with wait‐list/no‐treatment for continuous data.
The analysis on pharmacological placebos compared with wait‐list and no‐treatment showed that the required information size was not reached. See Figure 137. A funnel plot for the comparison between all placebos and wait‐list and no‐treatment were not possible due to lack of data.
12.
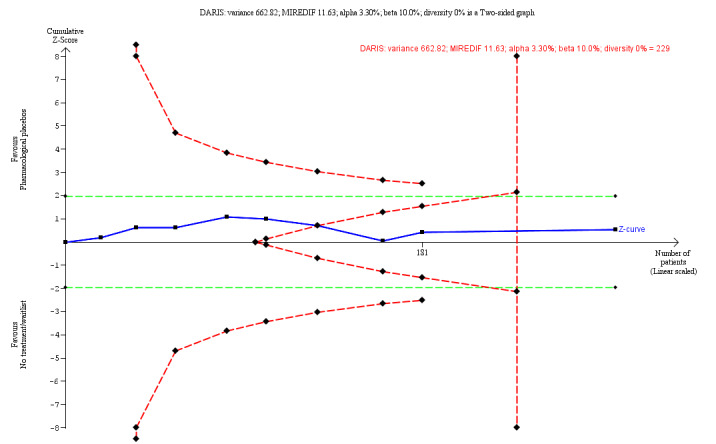
We performed a trial sequential analysis on the primary outcome for efficacy of pharmacological placebos compared with wait‐list and no‐treatment. The TSA showed the cumulated Z curve enters the futility area. See Figure 10 above. MIREDIF: Minimum relevant difference
The analysis on physical placebos compared with wait‐list and no‐treatment showed that the required information size was reached. See Figure 138. We drew a funnel plot for the comparison between all placebos and wait‐list and no‐treatment. The funnel plot shows no signs of asymmetry. See Figure 139.
13.
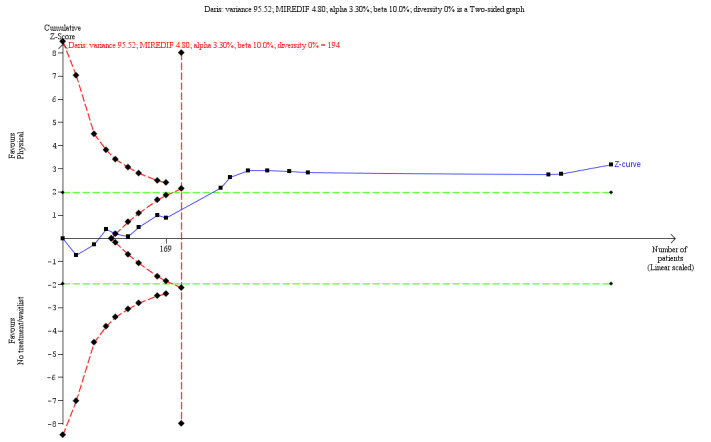
We performed a trial sequential analysis on the primary outcome for efficacy of physical placebos compared with wait‐list and no‐treatment. The analysis shows that the required information size was reached. See Figure 11 above. MIREDIF: Minimum relevant difference
14.

Funnel plot of comparison: 11.1 Efficacy of physical placebos compared with wait‐list/no‐treatment for continuous data.
Appendix 5. Subgroup analyses
Analyses for specific mental health disorders
Differences in outcomes measuring benefits between usual care versus wait‐list or no‐treatment for specific mental health disorders
There were not at least three trials for a specific mental health disorders regarding usual care.
Differences in outcomes measuring benefits between all placebos versus wait‐list or no treatment for specific mental health disorders
All placebos compared with wait‐list or no‐treatment for for people with substance use disorders
Four trials compared all placebos with no‐treatment (see Table 8).
All placebos compared with wait‐list or no‐treatment for continuous data: no differences were found for efficacy for patients with substance use disorders comparing all placebos with wait‐list or no‐treatment (SMD ‐0.15, 95% CI ‐0.49 to 0.19; 4 trials, 136 participants; P = 0.37; I 2 = 0%; low‐quality evidence; Analysis 12.1). Test for subgroup differences: not applicable.
12.1. Analysis.

Comparison 12: All placebos versus wait‐list or no‐treatment for people with specific mental health disorders for continuous data, Outcome 1: Efficacy of all placebos for people with substance use disorders
All placebos compared with wait‐list or no‐treatment for for people with sleep‐wake disorders
11 trials compared all placebos with wait‐list or no‐treatment (see Table 8).
All placebos compared with wait‐list or no‐treatment for continuous data:all placebos had a beneficial effect for patients with sleep‐wake disorders compared with wait‐list or no‐treatment (SMD ‐0.34, 95% CI ‐0.60 to ‐0.07; 11 trials, 229 participants; P = 0.01; I 2 = 0%; very low‐quality evidence; Analysis 12.2). No differences were found between subgroups: all placebos compared with wait‐list (SMD ‐0.58, 95% CI ‐1.10 to ‐0.05; 3 trials, 70 participants; P = 0.03; I 2 = 14%), and all placebos compared with no‐treatment (SMD ‐0.24, 95% CI ‐0.55 to 0.08; 8 trials, 159 participants;P = 0.14; I 2 = 0%). Test for subgroup differences: Chi 2 = 1.19, df = 1 (P = 0.28; I 2 = 15.7%)
12.2. Analysis.

Comparison 12: All placebos versus wait‐list or no‐treatment for people with specific mental health disorders for continuous data, Outcome 2: Efficacy of all placebos for people with sleep‐wake disorders
12.3. All placebos compared with wait‐list or no‐treatment for for people with depression
10 trials compared all placebos with wait‐list or no‐treatment (Table 8).
All placebos compared with wait‐list or no‐treatment for continuous data: all placebos had a beneficial effect for patients with depression compared with wait‐list or no‐treatment (SMD ‐0.42, 95% CI ‐0.78 to ‐0.05; 10 trials, 286 participants; P = 0.03; I 2 = 51%; low‐quality evidence; Analysis 12.3) No differences were found between subgroups: all placebos compared with wait‐list (SMD ‐0.65, 95% CI ‐1.15 to ‐0.15; 6 trials, 197 participants; P = 0.01; I 2 = 57%), and all placebos compared with no‐treatment (SMD ‐0.05, 95% CI ‐0.47 to 0.37; 4 trials, 89 participants; P = 0.82; I 2 = 0%). Test for subgroup differences: Chi 2 = 3.31, df = 1 (P = 0.07: I 2 = 69.8%)
12.3. Analysis.

Comparison 12: All placebos versus wait‐list or no‐treatment for people with specific mental health disorders for continuous data, Outcome 3: Efficacy of all placebos for people with depression
All placebos compared with wait‐list or no‐treatment for for people with post‐traumatic stress disorder (PTSD)
Four trials comparing all placebos with wait‐list or no‐treatment (Table 8).
All placebos compared with wait‐list or no‐treatment for continuous data: all placebos had a beneficial effect for patients with PTSD compared with wait‐list or no‐treatment (SMD ‐0.54, 95% CI ‐1.06 to ‐0.02; 4 trials, 329 participants; P = 0.04; I 2 = 74%; low‐quality evidence; Analysis 12.4). No differences were found between subgroups: all placebos compared with wait‐list (SMD ‐0.75, 95% CI ‐1.23 to ‐0.27; 3 trials, 231 participants; P = 0.002; I 2 = 55%), and all placebos compared with no‐treatment (SMD ‐0.08, 95% CI ‐0.56 to 0.40; 1 trial, 98 participants; I 2 = not applicable; P = 0.74). Test for subgroup differences: Chi 2 = 3.70, df = 1 (P = 0.05; I 2 = 73%).
12.4. Analysis.

Comparison 12: All placebos versus wait‐list or no‐treatment for people with specific mental health disorders for continuous data, Outcome 4: Efficacy of all placebos for people with post‐traumatic stress disorder (PTSD)
All placebos compared with wait‐list or no‐treatment for for people with anxiety disorder
Sixteen trials compared all placebos with wait‐list or no‐treatment (Table 8).
All placebos compared with wait‐list or no‐treatment for continuous data : All placebos had a beneficial effect for patients with anxiety disorders compared with wait‐list or no‐treatment (SMD ‐0.57, 95% CI ‐0.93 to ‐0.21; 16 trials, 401 participants; P = 0.002; I 2 = 66%; very low‐quality evidence; Analysis 12.5). No differences were found between subgroups: all placebos compared with wait‐list (SMD ‐0.81, 95% CI ‐1.66 to 0.05; 6 trials, 181 participants; I 2 = 85%; P = 0.07), and all placebos compared with no‐treatment (SMD ‐0.39, 95% CI ‐0.66 to ‐0.11; 10 trials, 220 participants; P = 0.006; I 2 = 0%). Test for subgroup differences: Chi 2 = 0.83, df = 1 (P = 0.36; I 2 = 0%).
12.5. Analysis.

Comparison 12: All placebos versus wait‐list or no‐treatment for people with specific mental health disorders for continuous data, Outcome 5: Efficacy of all placebos for people with anxiety disorders
12.6. All placebos compared with wait‐list or no‐treatment for for people with attention deficit hyperactivity disorder (ADHD) or attention deficit disorder (ADD)
Five trials compared all placebos with no‐treatment (Table 8)
All placebos compared with wait‐list or no‐treatment for continuous data: no differences were found for beneficial effect for patients with ADHD or ADD comparing all placebos with wait‐list or no‐treatment (SMD ‐0.31, 95% CI ‐0.64 to 0.02; 5 trials, 145 participants; P = 0.06; I 2 = 0%; very low‐quality evidence; Analysis 12.6). Test for subgroup differences: not applicable.
12.6. Analysis.

Comparison 12: All placebos versus wait‐list or no‐treatment for people with specific mental health disorders for continuous data, Outcome 6: Efficacy of all placebos for people with attention deficit hyperactivity disorder (ADHD) or attention deficit disorder (ADD)
All placebos compared with wait‐list or no‐treatment forfor people with neurodegenerative disorders
Four trials compared all placebos with no‐treatment (see Table 8).
All placebos compared with wait‐list or no‐treatment for continuous data: no differences were found for beneficial effect for patients with neurodegenerative disorders comparing all placebos with wait‐list or no‐treatment (SMD ‐0.21, 95% CI ‐0.47 to 0.05; 4 trials, 231 participants; P = 0.12; I 2 = 0%; very low‐quality evidence; Analysis 12.7). No differences were found between subgroups: all placebos compared with wait‐list (SMD ‐0.33, 95% CI ‐0.67 to 0.01; 2 trials, 124 participants; P = 0.06; I 2 = 0%), and all placebos compared with no‐treatment (SMD ‐0.03, 95% CI ‐0.43 to 0.36; 2 trials, 97 participants; P = 0.86; I 2 = 0%). Test for subgroup differences: Chi 2 = 1.24, df = 1 (P = 0.27; I 2 = 19.2%).
12.7. Analysis.

Comparison 12: All placebos versus wait‐list or no‐treatment for people with specific mental health disorders for continuous data, Outcome 7: Efficacy of all placebos for people with neurodegenerative disorders
Differences in outcomes measuring benefits between psychological placebos versus wait‐list or no treatment for specific mental health disorders
Psychological placebos compared with wait‐list or no‐treatment for people with substance use disorders
Three trials compared psychological placebos with no‐treatment (Table 8).
Psychological placebos compared with wait‐list or no‐treatment for continuous data: no differences were found for beneficial effect for patients with substance use disorders comparing psychological placebos with wait‐list or no‐treatment (SMD ‐0.19, 95% CI ‐0.54 to 0.17; 3 trials, 122 participants; P = 0.31; I 2 = 0%; very low‐quality evidence; Analysis 13.1). Test for subgroup differences: not applicable.
13.1. Analysis.

Comparison 13: Psychological placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data, Outcome 1: Efficacy of all placebos for people with substance use disorders
Psychological placebos compared with wait‐list or no‐treatment for people with sleep‐wake disorders
Seven trials compared psychological placebos with wait‐list or no‐treatment (Table 8).
Psychological placebos compared with wait‐list or no‐treatment for continuous data: psychological placebos had a beneficial effect for patients with sleep‐wake disorders compared with wait‐list or no‐treatment (SMD ‐0.44, 95% CI ‐0.76 to ‐0.12; 7 trials, 159 participants; P = 0.007; I 2 = 0%; low‐quality evidence; Analysis 13.2). No differences were found between subgroups: psychological placebos compared with wait‐list (SMD ‐0.63, 95% CI ‐1.49 to 0.24; 2 trials, 52 participants; P = 0.15; I 2 = 56%), and psychological placebos compared with no‐treatment (SMD ‐0.37, 95% CI ‐0.75 to 0.02; 5 trials, 107 participants; P = 0.06; I 2 = 0%). Test for subgroup differences: Chi 2 = 0.29, df = 1 (P = 0.59; I 2 = 0%).
13.2. Analysis.

Comparison 13: Psychological placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data, Outcome 2: Efficacy of all placebos for people with sleep‐wake disorders
13.3. Psychological placebos compared with wait‐list or no‐treatment for people with depression
Four trials compared psychological placebos with wait‐list or no‐treatment (Table 8).
Psychological placebos compared with wait‐list or no‐treatment for continuous data: no differences were found for beneficial effect for patients with depression comparing psychological placebos with wait‐list or no‐treatment (SMD ‐0.66, 95% CI ‐1.56 to 0.23; 4 trials, 86 participants; P = 0.15; I 2 = 73%; very low‐quality evidence; Analysis 13.3). There were differences between subgroups: psychological placebos compared with wait‐list (SMD ‐1.01, 95% CI ‐1.82 to ‐0.21; 3 trials, 66 participants; P = 0.01; I 2 = 55%), and psychological placebos compared with no‐treatment (SMD 0.34, 95% CI ‐0.55 to 1.22; 1 trial, 20 participants; P = 0.46; I 2 = not applicable). Test for subgroup differences: Chi 2 = 4.90, df = 1 (P = 0.03, I 2 = 79.6%).
13.3. Analysis.

Comparison 13: Psychological placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data, Outcome 3: Efficacy of all placebos for people with depression
Psychological placebos compared with wait‐list or no‐treatment for people with post‐traumatic stress disorder (PTSD)
Three trials compared psychological placebos with wait‐list or no‐treatment (Table 8).
Psychological placebos compared with wait‐list or no‐treatment for continuous data: psychological placebos had a beneficial effect for patients with PTSD compared with wait‐list or no‐treatment (SMD ‐0.75, 95% CI ‐1.23 to ‐0.27; 3 trials, 231 participants; P = 0.002; I 2 = 55%; very low‐quality evidence; Analysis 13.4). Test for subgroup differences: not applicable.
13.4. Analysis.

Comparison 13: Psychological placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data, Outcome 4: Efficacy of all placebos for people with post‐traumatic stress disorder (PTSD)
Psychological placebos compared with wait‐list or no‐treatment for people with anxiety disorders
Eleven trials compared psychological placebos with wait‐list or no‐treatment (Table 8).
Psychological placebos compared with wait‐list or no‐treatment for continuous data: psychological placebos had a beneficial effect for patients with anxiety disorders compared with wait‐list or no‐treatment (SMD ‐0.71, 95% CI ‐1.19 to ‐0.22; 11 trials, 258 participants; P = 0.005; I 2 = 70%; very low‐quality evidence; Analysis 13.5). No differences were found between subgroups: psychological placebos compared with wait‐list (SMD ‐0.94, 95% CI ‐1.83 to ‐0.05; 6 trials, 157 participants; P = 0.04); I 2 = 84%), and psychological placebos compared with no‐treatment (SMD ‐0.43, 95% CI ‐0.83 to ‐0.04; 5 trials, 101 participants; P = 0.03; I 2 = 0%). Test for subgroup differences: Chi 2 = 1.04, df = 1 (P = 0.31; I 2 = 3.7%).
13.5. Analysis.

Comparison 13: Psychological placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data, Outcome 5: Efficacy of all placebos for people with anxiety disorders
Differences in outcomes measuring benefits between pharmacological placebos versus wait‐list or no treatment for specific mental health disorders
Pharmacological placebos compared with wait‐list or no‐treatment for people with attention deficit hyperactivity disorder (ADHD) or attention deficit disorder (ADD)
Three trials compared pharmacological placebos with no‐treatment(Table 8).
Pharmacological placebos compared with wait‐list or no‐treatment for continuous data: no differences were found for beneficial effect for patients with ADHD or ADD comparing pharmacological placebos with wait‐list or no‐treatment (SMD ‐0.10, 95% CI ‐0.59 to 0.39; 3 trials, 64 participants; P = 0.68; I 2 = 0%; low‐quality evidence; Analysis 14.1). Test for subgroup differences: not applicable.
14.1. Analysis.

Comparison 14: Pharmacological placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data, Outcome 1: Efficacy of all placebos for people with attention deficit hyperactivity disorder (ADHD) or attention deficit disorder (ADD)
Differences in outcomes measuring benefits between physical placebos versus wait‐list or no treatment for specific mental health disorders
Physical placebos compared with wait‐list or no‐treatment for people with sleep‐wake disorders
Four trials comparing all placebos with wait‐list or no‐treatment (Table 8).
Physical placebos compared with wait‐list or no‐treatment for continuous data: no differences were found for beneficial effect for patients with sleep‐wake disorders comparing physical placebos with wait‐list or no‐treatment (SMD ‐0.11, 95% CI ‐0.58 to 0.36; 4 trials, 70 participants; P = 0.65; I 2 = 0%; very low‐quality evidence; Analysis 15.1). No differences were found between subgroups: physical placebos compared with wait‐list (SMD ‐0.52, 95% CI ‐1.46 to 0.43; 1 trial, 18 participants; P = 0.28; I 2 = not applicable), and physical placebos compared with no‐treatment (SMD 0.03, 95% CI ‐0.52 to 0.57; 3 trials, 52 participants; P = 0.93; I 2 = 0%). Test for subgroup differences: Chi 2 = 0.95, df = 1 (P = 0.33; I 2 = 0%).
15.1. Analysis.

Comparison 15: Physical placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data, Outcome 1: Efficacy of all placebos for people with sleep‐wake disorders
Physical placebos compared with wait‐list or no‐treatment for people with depression
Four trials compared physical placebos with wait‐list or no‐treatment (Table 8).
Physical placebos compared with wait‐list or no‐treatment for continuous data: no differences were found for beneficial effect for patients with depression comparing physical placebos with wait‐list or no‐treatment (SMD ‐0.24, 95% CI ‐0.73 to 0.25; 4 trials, 162 participants; P = 0.12; I 2 = 0%; very low‐quality evidence; Analysis 15.2). No differences were found between subgroups: physical placebos compared with wait‐list (SMD ‐0.27, 95% CI ‐1.28 to 0.75; 2 trials, 111 participants; P = 0.61; I 2 = 79%), and physical placebos compared with no‐treatment (SMD ‐0.10, 95% CI ‐0.65 to 0.45; 2 trials, 51 participants; P = 0.72; I 2 = 0%). Test for subgroup differences: Chi 2 = 0.08, df = 1 (P = 0.78; I 2 = 0%).
15.2. Analysis.

Comparison 15: Physical placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data, Outcome 2: Efficacy of all placebos for people with depression
Physical placebos compared with wait‐list or no‐treatment for people with anxiety disorders
Three trials compared physical placebos with wait‐list or no‐treatment (Table 8).
Physical placebos compared with wait‐list or no‐treatment for continuous data: no differences were found for beneficial effect for patients with anxiety disorders comparing physical placebos with wait‐list or no‐treatment (SMD ‐0.36, 95% CI ‐0.82 to 0.09; 3 trials, 84 participants; P = 0.002; I 2 = 66%; very low‐quality evidence; Analysis 15.3). No differences were found between subgroups: physical placebos compared with wait‐list (SMD ‐0.11, 95% CI ‐0.82 to 0.60; 1 trial, 36 participants; P = 0.76; I 2 = not applicable), and physical placebos compared with no‐treatment (SMD ‐0.54, 95% CI ‐1.14 to 0.06; 2 trials, 48 participants; P = 0.08; I 2 = 0%). Test for subgroup differences: Chi 2 = 0.82, df = 1 (P = 0.36, I 2 = 0%).
15.3. Analysis.

Comparison 15: Physical placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data, Outcome 3: Efficacy of all placebos for people with anxiety disorders
Ordinary Subgroup analyses
Subgroup analyses were conducted if two or more trials were included in each group. Therefore, some of the predefined subgroup analyses were not possible (see Table 9).
Subgroup analyses for all placebos
Type of active intervention (continuous data). Comparing psychological interventions to pharmacological, physical, and other or combination interventions
No differences were found between subgroups: psychological interventions (SMD ‐0.48, 95% CI ‐0.66 to ‐0.30; 38 trials, 1215 participants; I 2 = 54%; P < 0.0001), pharmacological interventions (SMD ‐0.29, 95% CI ‐0.62 to 0.04; 7 trials, 148 participants; I 2 = 0%; P = 0.08), physical interventions (SMD ‐0.21, 95% CI ‐0.35 to ‐0.07; 13 trials, 798 participants; I 2 = 0%; P = 0.004) and other or combination interventions (SMD ‐0.32, 95% CI ‐0.59 to ‐0.04; 6 trials, 252 participants; I 2 = 0%; P = 0.02). Test for subgroup differences: Chi 2 = 5.46, df = 3 (P = 0.14), I 2 = 45.1%; Analysis 16.1)
16.1. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 1: Type of active intervention
Type of active intervention (dichotomous data). Comparing psychological interventions to pharmacological, physical, and other or combination interventions
No differences were found between subgroups: psychological interventions (RR not estimable; 1 trial, 19 participants; I 2 = not applicable; P = not applicable), pharmacological interventions (RR 1.04, 95% CI 0.74 to 1.48; 7 trials, 316 participants; I 2 = 49%; P = 0.82), and other or combination interventions (RR 0.99, 95% CI 0.58 to 1.70; 1 trial, 50 participants; I 2 = not applicable; P = 0.98). Test for subgroup differences: Chi 2 = 0.02, df = 1 (P = 0.89), I 2 = 0%; Analysis 16.2)
16.2. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 2: Type of active intervention
Risk of bias (continuous data). Comparing trials of low risk of bias to trial of high risk of bias
Test for subgroup differences: not applicable (Analysis 16.3)
16.3. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 3: Risk of bias
Risk of bias (dichotomous data). Comparing trials of low risk of bias to trial of high risk of bias
Test for subgroup differences: not applicable (Analysis 16.4)
16.4. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 4: Risk of bias
Type of outcome domain (continuous data). Comparing blinded observer‐reported to non‐blinded observer‐reported and patient‐reported outcomes
There were differences between subgroups: blinded observer‐reported (SMD ‐0.50, 95% CI ‐0.70 to ‐0.31; 29 trials, 1046 participants; I 2 = 50%; P < 0.00001), non‐blinded observer‐reported (SMD ‐0.54, 95% CI ‐0.95 to ‐0.13; 2 trials, 96 participants; I 2 = 0%; P = 0.009), and patient‐reported (SMD ‐0.19, 95% CI ‐0.35 to ‐0.03; 25 trials, 1063 participants; I 2 = 26%; P = 0.02). Test for subgroup differences: Chi 2 = 6.73, df = 2 (P = 0.03), I 2 = 70.3%; Analysis 16.5)
16.5. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 5: Type of outcome domain
Type of outcome domain (dichotomous data). Comparing blinded observer‐reported to non‐blinded observer‐reported and patient‐reported outcomes
No differences were found between subgroups: blinded observer‐reported (RR 0.99, 95% CI 0.60 to 1.64; 3 trials, 113 participants; I 2 = 0%; P = 0.98), and patient‐reported (RR 1.00, 95% CI 0.60 to 1.65; 2 trials, 76 participants; I 2 = 73%; P = 0.99). Test for subgroup differences: Chi 2 = 0.00, df = 1 (P = 1.00), I 2 = 0%; Analysis 16.6)
16.6. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 6: Type of outcome domain
Awareness of placebo intervention (continuous data). Comparing open placebos to closed placebos
No differences were found between subgroups: open placebo (SMD ‐0.51, 95% CI ‐1.41 to 0.38; 1 trial, 20 participants; I 2 = not applicable; P = 0.26), and closed placebos (SMD ‐0.37, 95% CI ‐0.49 to ‐0.25; 64 trials, 2426 participants; I 2 = 42%; P < 0.0001). Test for subgroup differences: Chi 2 = 0.10, df = 1 (P = 0.75), I 2 = 0%; Analysis 16.7)
16.7. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 7: Awareness of placebo intervention
Awareness of placebo intervention (dichotomous data). Comparing open placebos to closed placebos
Test for subgroup differences: not applicable (Analysis 16.8)
16.8. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 8: Awareness of placebo intervention
The trial objective (continuous data). Comparing a trial’s objective is clearly to assess the effects of placebo to trials without no such objectives
Test for subgroup differences: not applicable (Analysis 16.9)
16.9. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 9: The trial objective
The trial objective (dichotomous data). Comparing a trial’s objective is clearly to assess the effects of placebo to trials without no such objectives
Test for subgroup differences: not applicable (Analysis 16.10)
16.10. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 10: The trial objective
Mean age of participants (continuous data). Comparing below 18 years of age to 18 to 50 years of age and to above 50 years of age
No differences were found between subgroups: below 18 years of age (SMD ‐0.47, 95% CI ‐0.98 to 0.04; 9 trials, 274 participants; I 2 = 75%; P = 0.07), 18 to 50 years of age (SMD ‐0.31, 95% CI ‐0.44 to ‐0.18; 40 trials, 1735 participants; I 2 = 31%; P < 0.00001), and above 50 years of age (SMD ‐0.41, 95% CI ‐0.75 to ‐0.06; 8 trials, 292 participants; I 2 = 45%; P = 0.02). Test for subgroup differences: Chi 2 = 0.54, df = 2 (P = 0.76), I 2 = 0%; Analysis 16.11)
16.11. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 11: Mean age of participants
Mean age of participants (dichotomous data). Comparing below 18 years of age to 18 to 50 years of age and to above 50 years of age
No differences were found between subgroups: 18 to 50 years of age (RR 1.07, 95% CI 0.70 to 1.62; 5 trials, 270 participants; I 2 = 53%; P = 0.76), and above 50 years of age (RR 0.73, 95% CI 0.43 to 1.27; 2 trials, 70 participants; I 2 = 0%; P = 0.27). Test for subgroup differences: Chi 2 = 1.14, df = 1 (P = 0.29), I 2 = 12.2%; Analysis 16.12)
16.12. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 12: Mean age of participants
Duration of treatment (continuous data). Comparing above three months of duration to below three months of duration
No differences were found between subgroups: above three months (SMD ‐0.68, 95% CI ‐1.09 to ‐0.27; 11 trials, 464 participants; I 2 = 74%; P = 0.001), and below three months (SMD ‐0.32, 95% CI ‐0.43 to ‐0.20; 53 trials, 1902 participants; I 2 = 18%; P < 0.00001). Test for subgroup differences: Chi 2 = 2.86, df = 1 (P = 0.09), I 2 = 65.1%; Analysis 16.13)
16.13. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 13: Duration of treatment
Duration of treatment (dichotomous data). Comparing above three months of duration to below three months of duration
No differences were found between subgroups: above three months (RR 1.45, 95% CI 0.84 to 2.49; 4 trials, 196 participants; I 2 = 40%; P = 0.18), and below three months (RR 0.82, 95% CI 0.68 to 0.98; 4 trials, 170 participants; I 2 = 0%; P < 0.03). Test for subgroup differences: Chi 2 = 3.88, df = 1 (P = 0.05), I 2 = 74.2%; Analysis 16.14)
16.14. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 14: Duration of treatment
Mental health diagnoses (continuous data). Comparing formal diagnosis according to DSM or ICD to fulfilment of symptoms according to DSM or ICD and population being classified as having a mental health disorder, but full diagnostic criteria not reported
No differences were found between subgroups: formal diagnosis according to DSM/ICD (SMD ‐0.42, 95% CI ‐0.62 to ‐0.21;29 trials, 1256 participants; I 2 = 63%; P < 0.001), fulfil symptoms of disorder according to DSM/ICD while not stating classifications systems (SMD ‐0.30, 95% CI ‐0.52 to ‐0.08; 14 trials, 326 participants; I 2 = 0%; P = 0.008), and population is classified as having a mental health disorder, but full diagnostic criteria not reported (SMD ‐0.27, 95% CI ‐0.42 to ‐0.12; 21 trials, 864 participants; I 2 = 4%; P = 0.0004). Test for subgroup differences: Chi 2 = 1.27, df = 2 (P = 0.53), I 2 = 0%; Analysis 16.15)
16.15. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 15: Mental health diagnoses
Mental health diagnoses (dichotomous data). Comparing formal diagnosis according to DSM or ICD to fulfilment of symptoms according to DSM or ICD, while not stating classifications system, and population being classified as having a mental health disorder, but full diagnostic criteria not reported
No differences were found between subgroups: formal diagnosis according to DSM/ICD (RR 1.31, 95% CI 0.72 to 2.37; 4 trials, 220 participants; I 2 = 48%; P = 0.38), fulfil symptoms of disorder according to ICD/DSM while not stating classifications systems (RR 1.01, 95% CI 0.62 to 1.63; 3 trials, 96 participants; I 2 = 31%; P = 0.98), and population is classified as having a mental health disorder, but full diagnostic criteria not provided (RR 0.80, 95% CI 0.65 to 0.99; 2 trials, 69 participants; I 2 = NA; P = 0.04). Test for subgroup differences: Chi 2 = 2.67, df = 2 (P = 0.26), I 2 = 25.2%; Analysis 16.16)
16.16. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 16: Mental health diagnoses
Affiliation bias (continuous data). Comparing risk of affiliation, industry and allegiance bias to no risk found of affiliation, industry and allegiance bias
No differences were found between subgroups: risk of affiliation bias (SMD ‐0.61, 95% CI ‐0.95 to ‐0.26; 6 trials, 363 participants; I 2 = 52%; P = 0.005), and no risk of affiliation bias (SMD ‐0.33, 95% CI ‐0.45 to ‐0.21; 59 trials, 2083 participants; I 2 = 34%; P < 0.00001). Test for subgroup differences: Chi 2 = 2.22, df = 1 (P = 0.14), I 2 = 55%; Analysis 16.17)
Risk of bias (participants and personnel excluded). Comparing low risk of bias with unclear risk of bias and high risk of bias.
No differences were found between subgroups: Low risk of bias (SMD ‐0.58, 95% CI ‐0.85 to ‐0.32; 3 trials, 230 participants; I 2 = 0%; P < 0.001), unclear risk of bias (SMD ‐0.12, 95% CI ‐0.95 to 0.71; 2 trials, 51 participants; I 2 = 52%; P = 0.58) and high risk of bias (SMD ‐0.36, 95% CI ‐0.48 to ‐0.23, 60 trials, 2165 participants; I 2 = 42%; P < 0.00001). Test for subgroup differences: Chi² = 2.70, df = 2 (P = 0.26), I² = 26.0%; Analysis 16.18)
Imputed data. Comparing available data with intention‐to‐treat and no‐attrition data
No differences were found between subgroups: available data (SMD ‐0.36, 95% CI ‐0.48 to ‐0.24, 63 trials, 2416 participants, I 2 = 19%; P < 0.00001), intention‐to‐treat (SMD ‐0.22, 95% CI ‐0.44 to ‐0.01, 9 trials, 891 participants, I 2 = 46%; P = 0.04), and no attrition (SMD ‐0.50, 95% CI ‐1.00 to 0.00, 9 trials, 289 participants, I 2 = 75%; P = 0.05 Test for subgroup differences: Chi² = 5.65, df = 2 (P = 0.06), I² = 64%; Analysis 16.19
16.19. Analysis.

Comparison 16: Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data, Outcome 19: Imputed data
Subgroup analyses for psychological placebos
Type of active intervention (continuous data). Comparing psychological interventions to pharmacological, physical, and other or combination interventions
No differences were found between subgroups: psychological interventions (SMD ‐0.48, 95% CI ‐0.67 to ‐0.29; 35 trials, 1174 participants; I 2 = 56%; P < 0.0001), and other or combination interventions (SMD ‐0.51, 95% CI ‐0.94 to ‐0.09; 3 trials, 89 participants; I 2 = 0%; P = 0.02). Test for subgroup differences: Chi 2 = 0.01, df = 1 (P = 0.91), I 2 = 0%; Analysis 17.1)
17.1. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 1: Type of active intervention
Risk of bias (continuous data). Comparing trials of low risk of bias to trial of high risk of bias
Test for subgroup differences: not applicable (Analysis 17.2)
17.2. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 2: Risk of bias
Type of outcome domain (continuous data). Comparing blinded observer‐reported to non‐blinded observer‐reported and patient‐reported outcomes
No differences were found between subgroups: blinded observer‐reported (SMD ‐0.72, 95% CI ‐1.08 to ‐0.36; 14 trials, 512 participants; I 2 = 68%; P < 0.00001), non‐blinded observer‐reported (SMD ‐0.54, 95% CI ‐0.95 to ‐0.13; 2 trials, 96 participants; I 2 = 0%; P = 0.009), and patient‐reported (SMD ‐0.27, 95% CI ‐0.47 to ‐0.08; 18 trials, 907 participants; I 2 = 36%; P = 0.007). Test for subgroup differences: Chi 2 = 5.02, df = 2 (P = 0.08), I 2 = 60.2%; Analysis 17.3)
17.3. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 3: Type of outcome domain
Awareness of placebo intervention (continuous data). Comparing open placebos to closed placebos
Test for subgroup differences: not applicable (Analysis 17.4)
17.4. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 4: Awareness of placebo intervention
The trial objective (dichotomous data). Comparing a trial’s objective is clearly to assess the effects of placebo to trials without no such objectives
Test for subgroup differences: not applicable (Analysis 17.5)
17.5. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 5: The trial objective
Mean age of participants (continuous data). Comparing below 18 years of age to 18 to 50 years of age and above 50 years of age
No differences were found between subgroups: below 18 years of age (SMD ‐0.85, 95% CI ‐2.02 to ‐0.32; 4 trials, 148 participants; I 2 = 89%; P = 0.16), 18 to 50 years of age (SMD ‐0.38, 95% CI ‐0.55 to ‐0.21; 25 trials, 1233 participants; I 2 = 40%; P < 0.00001), and above 50 years of age (SMD ‐0.59, 95% CI ‐1.33 to 0.14; 4 trials, 156 participants; I 2 = 75%; P = 0.11). Test for subgroup differences: Chi 2 = 0.88, df = 2 (P = 0.64), I 2 = 0%; Analysis 17.6)
17.6. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 6: Mean age of participants
Duration of treatment (continuous data). Comparing above three months of duration to below three months of duration
No differences were found between subgroups: above three months (SMD ‐1.02, 95% CI ‐1.68 to ‐0.36; 6 trials, 270 participants; I 2 = 83%; P = 0.003), and below three months (SMD ‐0.38, 95% CI ‐0.53 to ‐0.22; 32 trials, 1306 participants; I 2 = 33%; P < 0.00001). Test for subgroup differences: Chi 2 = 3.42, df = 1 (P = 0.06), I 2 = 70.8%; Analysis 17.7)
17.7. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 7: Duration of treatment
Type of psychological placebo (continuous data). Comparing interactive placebo to psychoeducational and exposure placebo
There were differences between subgroups: interaction placebo (SMD ‐0.70, 95% CI ‐1.04 to ‐0.36; 15 trials, 613 participants; I 2 = 70%; P < 0.0001), psychoeducational placebo (SMD ‐0.14, 95% CI ‐0.36 to 0.07; 9 trials, 329 participants; I 2 = 0%; P = 0.20), and exposure placebo (SMD ‐0.48, 95% CI ‐0.71 to ‐0.24; 14 trials, 321 participants; I 2 = 6%; P < 0.0001). Test for subgroup differences: Chi 2 = 8.68, df = 2 (P = 0.01), I 2 = 76.9%; Analysis 17.8)
Mode of psychological placebo (continuous data). Comparing individual treatment to group and a combination of both
No differences were found between subgroups: individual treatment (SMD ‐0.31, 95% CI ‐0.49 to ‐0.12; 19 trials, 1151 participants; I 2 = 45%; P = 0.001), group treatment (SMD ‐0.85, 95% CI ‐1.30 to ‐0.39; 13 trials, 308 participants; I 2 = 70%; P = 0.0003), and combination of individual and group (SMD ‐0.45, 95% CI ‐0.83 to ‐0.08; 3 trials, 112 participants; I 2 = 0%; P = 0.02). Test for subgroup differences: Chi 2 = 4.62, df = 2 (P = 0.10), I 2 = 56.7%; Analysis 17.9)
17.9. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 9: Mode of psychological placebo
Mental health diagnoses (continuous data). Comparing formal diagnosis according to DSM or ICD to fulfilment of symptoms according to DSM or ICD, while not stating classifications system, and population being classified as having a mental health disorder, but full diagnostic criteria not reported
No differences were found between subgroups: formal diagnosis according to DSM/ICD (SMD ‐0.53, 95% CI ‐0.86 to ‐0.21; 16 trials, 767 participants; I 2 = 76%; P = 0.001), fulfil symptoms of a disorder according to DSM/ICD while not stating classifications systems (SMD ‐0.39, 95% CI ‐0.66 to ‐0.12; 9 trials, 218 participants; I 2 = 0%; P = 0.005), and population is classified as having a mental health disorder, but full diagnostic criteria not reported (SMD ‐0.37, 95% CI ‐0.59 to ‐0.15; 14 trials, 671 participants; I 2 = 19%; P = 0.0008). Test for subgroup differences: Chi 2 = 0.70, df = 2 (P = 0.70), I 2 = 0%; Analysis 17.10)
17.10. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 10: Mental health diagnoses
Affiliation bias (continuous data). Comparing risk of affiliation, industry and allegiance bias to no risk found of affiliation, industry and allegiance bias
No differences were found between subgroups: risk of affiliation bias (SMD ‐0.61, 95% CI ‐0.95 to ‐0.26; 6 trials, 363 participants; I 2 = 52%; P = 0.005), and no risk of affiliation bias (SMD ‐0.33, 95% CI ‐0.45 to ‐0.21; 59 trials, 2083 participants; I 2 = 34%; P < 0.00001). Test for subgroup differences: Chi 2 = 2.22, df = 1 (P = 0.14), I 2 = 55%; Analysis 17.11)
17.11. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 11: Affiliation bias
Imputed data. Comparing available data with intention‐to‐treat and no‐attrition data.
No differences were found between subgroups: available data (SMD ‐0.50, 95% CI ‐0.68 to ‐0.33, 27 trials, 842 participants, I 2 = 27%; P < 0.00001), intention‐to‐treat (SMD ‐0.18, 95% CI ‐0.39 to ‐0.03, 6 trials, 682 participants, I 2 = 30%; P = 0.09), and no attrition (SMD ‐0.75, 95% CI ‐2.11 to 0.62, 4 trials, 102 participants, I 2 = 90%; P = 0.28. Test for subgroup differences: Chi² = 5.65, df = 2 (P = 0.28), I² = 64.6%; Analysis 17.12
17.12. Analysis.

Comparison 17: Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 12: Imputed data
Subgroup analyses for pharmacological placebos
Type of active intervention (continuous data). Comparing psychological interventions to pharmacological, physical, and other or combination interventions
No differences were found between subgroups: psychological interventions (SMD 0.44, 95% CI ‐0.26 to 1.13; 1 trial, 33 participants; I 2 = not applicable; P = 0.22), pharmacological interventions (SMD ‐0.29, 95% CI ‐0.62 to 0.04; 7 trials, 148 participants; I 2 = 0%; P = 0.08), and other or combination interventions (SMD ‐0.08, 95% CI ‐0.56 to 0.40; 1 trial, 98 participants; I 2 = not applicable; P = 0.74). Test for subgroup differences: Chi 2 = 3.55, df = 2 (P = 0.17), I 2 = 43.7%; Analysis 18.1)
18.1. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 1: Type of active intervention
Type of active intervention (dichotomous data). Comparing psychological interventions to pharmacological, physical, and other or combination interventions
No differences were found between subgroups: pharmacological interventions (RR 1.09, 95% CI 0.71 to 1.67; 7 trials, 316 participants; I 2 = 66%; P = 0.71), and other or combination interventions (RR 0.99, 95% CI 0.58 to 1.70; 1 trial, 50 participants; I 2 = not applicable; P = 0.98). Test for subgroup differences: Chi 2 = 0.06, df = 1 (P = 0.80), I 2 = 0%; Analysis 18.2)
18.2. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 2: Type of active intervention
Risk of bias (continuous data). Comparing trials of low risk of bias to trial of high risk of bias
Test for subgroup differences: not applicable (Analysis 18.3)
18.3. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 3: Risk of bias
Risk of bias (dichotomous data). Comparing trials of low risk of bias to trial of high risk of bias
Test for subgroup differences: not applicable (Analysis 18.4)
18.4. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 4: Risk of bias
Type of outcome domain (continuous data). Comparing blinded observer‐reported to non‐blinded observer‐reported and patient‐reported outcomes
No differences were found between subgroups: blinded observer‐reported (SMD ‐0.24, 95% CI ‐0.53 to 0.05; 7 trials, 220 participants; I 2 = 0%; P = 0.10), and patient‐reported (SMD 0.18, 95% CI ‐0.36 to 0.73; 2 trials, 59 participants; I 2 = 11%; P = 0.51). Test for subgroup differences: Chi 2 = 1.82, df = 1 (P = 0.18), I 2 = 45.0%; Analysis 18.5)
18.5. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 5: Type of outcome domain
Type of outcome domain (dichotomous data). Comparing blinded observer‐reported to non‐blinded observer‐reported and patient‐reported outcomes
No differences were found between subgroups: blinded observer‐reported (RR 0.99, 95% CI 0.60 to 1.64; 2 trials, 94 participants; I 2 = 0%; P = 0.98), and patient‐reported (RR 1.00, 95% CI 0.60 to 1.65; 2 trials, 76 participants; I 2 = 73%; P = 0.99). Test for subgroup differences: Chi 2 = 0.00, df = 1 (P = 1.00), I 2 = 0%; Analysis 18.6)
18.6. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 6: Type of outcome domain
Awareness of placebo intervention (continuous data). Comparing open placebos to closed placebos
No differences were found between subgroups: open placebo (SMD ‐0.51, 95% CI ‐1.41 to 0.38; 1 trial, 20 participants; I 2 = not applicable; P = 0.26), and closed placebos (SMD ‐0.11, 95% CI ‐0.37 to 0.16; 8 trials, 259 participants; I 2 = 0%; P = 0.43). Test for subgroup differences: Chi 2 = 0.73, df = 1 (P = 0.39), I 2 = 0%; Analysis 18.7)
18.7. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 7: Awareness of placebo intervention
Awareness of placebo intervention (dichotomous data). Comparing open placebos to closed placebos
Test for subgroup differences: not applicable (Analysis 18.8)
18.8. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 8: Awareness of placebo intervention
The trial objective (dichotomous data). Comparing a trial’s objective is clearly to assess the effects of placebo to trials without no such objectives
Test for subgroup differences: not applicable (Analysis 18.9)
18.9. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 9: The trial objective
The trial objective (dichotomous data). Comparing a trial’s objective is clearly to assess the effects of placebo to trials withoutno such objectives
Test for subgroup differences: not applicable (Analysis 18.10)
18.10. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 10: The trial objective
Mean age of participants (continuous data). Comparing below 18 years of age to 18 to 50 years of age and above 50 years of age
No differences were found between subgroups: below 18 years of age (SMD ‐0.10, 95% CI ‐0.59 to 0.39; 3 trials, 64 participants; I 2 = 0%; P = 0.68), and 18 to 50 years of age (SMD ‐0.19, 95% CI ‐0.68 to 0.31; 4 trials, 177 participants; I 2 = 50%; P = 0.46). Test for subgroup differences: Chi 2 = 0.88, df = 2 (P = 0.64), I 2 = 0%; Analysis 18.11)
18.11. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 11: Mean age of participants
Mean age of participants (dichotomous data). Comparing below 18 years of age to 18 to 50 years of age and above 50 years of age
No differences were found between subgroups: 18 to 50 years of age (RR 1.07, 95% CI 0.70 to 1.62; 5 trials, 270 participants; I 2 = 53%; P = 0.76), and above 50 years of age (RR 0.73, 95% CI 0.43 to 1.27; 2 trials, 70 participants; I 2 = 0%; P = 0.27), Test for subgroup differences: Chi 2 = 1.14, df = 1 (P = 0.29), I 2 = 12.2%; Analysis 18.12)
18.12. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 12: Mean age of participants
Duration of treatment (continuous data). Comparing above three months of duration to below three months of duration
No differences were found between subgroups: above three months (SMD ‐0.21, 95% CI ‐0.56 to 0.14; 4 trials, 164 participants; I 2 = 0%; P = 0.23), and below three months (SMD ‐0.06, 95% CI ‐0.43 to 0.31; 5 trials, 115 participants; I 2 = 0%; P = 0.77). Test for subgroup differences: Chi 2 = 0.35, df = 1 (P = 0.55), I 2 = 0%; Analysis 18.13)
18.13. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 13: Duration of treatment
Duration of treatment (dichotomous data). Comparing above three months of duration to below three months of duration
No differences were found between subgroups: above three months (RR 1.45, 95% CI 0.84 to 2.49; 4 trials, 196 participants; I 2 = 40%; P = 0.18), and below three months (RR 0.82, 95% CI 0.68 to 0.98; 4 trials, 170 participants; I 2 = 0%; P = 0.03). Test for subgroup differences: Chi 2 = 3.88, df = 1 (P = 0.05), I 2 = 74.2%; Analysis 18.14)
18.14. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 14: Duration of treatment
Mental health diagnoses (continuous data). Comparing formal diagnosis according to DSM or ICD to fulfilment of symptoms according to DSM or ICD, while not stating classifications system, and population being classified as having a mental health disorder, but full diagnostic criteria not reported
No differences were found between subgroups: formal diagnosis according to DSM/ICD (SMD ‐0.25, 95% CI ‐0.57 to 0.06; 5 trials, 188 participants; I 2 = 0%; P = 0.12), fulfil symptoms of disorder ICD/DSM while not stating classifications systems (SMD ‐0.07, 95% CI ‐0.69 to 0.56; 2 trials, 40 participants; I 2 = 0%; P = 0.83), and population is classified as having a mental health disorder, but full diagnostic criteria not reported (SMD 0.12, 95% CI ‐0.62 to 0.86; 2 trials, 51 participants; I 2 = 40%; P = 0.75). Test for subgroup differences: Chi 2 = 0.97, df = 2 (P = 0.62), I 2 = 0%; Analysis 18.15)
18.15. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 15: Mental health diagnoses
Mental health diagnoses (dichotomous data). Comparing formal diagnosis according to DSM or ICD to fulfilment of symptoms according to DSM or ICD, while not stating classifications system, and population being classified as having a mental health disorder, but full diagnostic criteria not reported
No differences were found between subgroups: formal diagnosis according to DSM/ICD (RR 1.31, 95% CI 0.72 to 2.37; 4 trials, 220 participants; I 2 = 48%; P = 0.38), and fulfil symptoms of disorder ICD/DSM while not stating classifications systems (RR 1.01, 95% CI 0.62 to 1.63; 3 trials, 96 participants; I 2 = 31%; P = 0.98). Test for subgroup differences: Chi 2 = 0.45, df = 1 (P = 0.50), I 2 = 0%; Analysis 18.16)
18.16. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 16: Mental health diagnoses
Imputed data. Comparing available data with intention‐to‐treat and no‐attrition data. +
No differences were found between subgroups: available able (SMD ‐0.17, 95% CI ‐0.49 to 0.16, 7 trials, 155 participants, I 2 = 3%; P = 0.31), intention‐to‐treat (SMD ‐0.08, 95% CI ‐0.56 to 0.40, 1 trial, 98 participants, I 2 = not applicable; P = 0.74), and no attrition (SMD ‐0.12, 95% CI ‐0.89 to 0.65, 1 trial, 26 participants, I 2 = not applicable; P = 0.76. Test for subgroup differences: Chi² = 0.09, df = 2 (P = 0.96), I² = 0%; Analysis 18.17)
18.17. Analysis.

Comparison 18: Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data, Outcome 17: Imputed data
Subgroup analyses for physical placebos
Type of active intervention (continuous data). Comparing psychological interventions to pharmacological, physical, and other or combination interventions
No differences were found between subgroups: psychological interventions (SMD ‐0.26, 95% CI ‐0.95 to 0.43; 2 trials, 33 participants; I 2 = 0%; P = 0.46), physical interventions (SMD ‐0.21, 95% CI ‐0.35 to ‐0.07; 13 trials, 798 participants; I 2 = 0%; P = 0.004), and other or combination interventions (SMD ‐0.30, 95% CI ‐0.82 to 0.23; 2 trials, 65 participants; I 2 = 0%; P = 0.26). Test for subgroup differences: Chi 2 = 0.13, df = 2 (P = 0.26), I 2 = 0%; Analysis 19.1)
19.1. Analysis.

Comparison 19: Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data, Outcome 1: Type of active intervention
Risk of bias (continuous data). Comparing trials of low risk of bias to trial of high risk of bias
Test for subgroup differences: not applicable (Analysis 19.2)
19.2. Analysis.

Comparison 19: Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data, Outcome 2: Risk of bias
Type of outcome domain (continuous data). Comparing blinded observer‐reported to non‐blinded observer‐reported and patient‐reported outcomes
No differences were found between subgroups: blinded observer‐reported (SMD ‐0.42, 95% CI ‐0.64 to ‐0.19; 8 trials, 314 participants; I 2 = 0%; P = 0.0004), and patient‐reported (SMD ‐0.00, 95% CI ‐0.40 to 0.40; 5 trials, 97 participants; I 2 = 0%; P = 1.00). Test for subgroup differences: Chi 2 = 3.11, df = 1 (P = 0.08), I 2 = 67.8%; Analysis 19.3)
19.3. Analysis.

Comparison 19: Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data, Outcome 3: Type of outcome domain
Awareness of placebo intervention (continuous data). Comparing open placebos to closed placebos
Test for subgroup differences: not applicable (Analysis 19.4)
19.4. Analysis.

Comparison 19: Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data, Outcome 4: Awareness of placebo intervention
The trial objective (continuous data). Comparing a trial’s objective is clearly to assess the effects of placebo to trials without no such objectives
Test for subgroup differences: not applicable (Analysis 19.5)
19.5. Analysis.

Comparison 19: Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data, Outcome 5: The trial objective
Mean age of participants (continuous data). Comparing below 18 years of age to 18 to 50 years of age and above 50 years of age
No differences were found between subgroups: below 18 years of age (SMD ‐0.32, 95% CI ‐0.82 to 0.19; 2 trials, 62 participants; I 2 = 0%; P = 0.22), 18 to 50 years of age (SMD ‐0.26, 95% CI ‐0.48 to ‐0.04; 11 trials, 325 participants; I 2 = 0%; P = 0.02), and above 50 years of age (SMD ‐0.40, 95% CI ‐0.77 to ‐0.03; 3 trials, 116 participants; I 2 = 0%; P = 0.03). Test for subgroup differences: Chi 2 = 0.40, df = 2 (P = 0.82), I 2 = 0%; Analysis 19.6)
19.6. Analysis.

Comparison 19: Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data, Outcome 6: Mean age of participants
Duration of treatment (continuous data). Comparing above three months of duration to below three months of duration
No differences were found between subgroups: above three months (SMD ‐0.48, 95% CI ‐1.21 to 0.25; 1 trial, 30 participants; I 2 = not applicable; P = 0.20), and below three months (SMD ‐0.21, 95% CI ‐0.35 to ‐0.08; 17 trials, 896 participants; I 2 = 0%; P = 0.002). Test for subgroup differences: Chi 2 = 0.49, df = 1 (P = 0.48), I 2 = 0%; Analysis 19.7)
19.7. Analysis.

Comparison 19: Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data, Outcome 7: Duration of treatment
Type of physical placebo (continuous data). Comparing acupuncture or acupressure placebo to exercise and relaxation, technical device and electromagnetic stimulation placebo
No differences were found between subgroups: acupuncture or acupressure placebo (SMD ‐0.42, 95% CI ‐0.73 to ‐0.11; 5 trials, 237 participants; I 2 = 21%; P = 0.008), exercise and relaxation placebo (SMD ‐0.21, 95% CI ‐0.70 to 0.27; 3 trials, 67 participants; I 2 = 0%; P = 0.39), technical device placebo (SMD ‐0.23, 95% CI ‐0.60 to 0.14; 5 trials, 119 participants; I 2 = 0%; P = 0.22), and electromagnetic stimulation placebo (SMD ‐0.02, 95% CI ‐0.57 to 0.54; 2 trials, 50 participants; I 2 = 0%; P = 0.95). Test for subgroup differences: Chi 2 = 1.80, df = 3 (P = 0.62), I 2 = 0%; Analysis 19.8)
19.8. Analysis.

Comparison 19: Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data, Outcome 8: Type of physical placebo
Mental health diagnoses (continuous data). Comparing formal diagnosis according to DSM or ICD to fulfilment of symptoms according to DSM or ICD, while not stating classifications system, and population being classified as having a mental health disorder, but full diagnostic criteria not reported
No differences were found between subgroups: formal diagnosis according to DSM/ICD (SMD ‐0.29, 95% CI ‐0.56 to ‐0.02; 7 trials, 293 participants; I 2 = 19%; P = 0.04), fulfil symptoms of disorder ICD/DSM while not stating classifications systems (SMD ‐0.15, 95% CI ‐0.62 to 0.33; 3 trials, 68 participants; I 2 = 0%; P = 0.55), and population is classified as having a mental health disorder, but full diagnostic criteria not reported (SMD ‐0.32, 95% CI ‐0.66 to 0.02; 6 trials, 142 participants; I 2 = 0%; P = 0.06). Test for subgroup differences: Chi 2 = 0.36, df = 2 (P = 0.84), I 2 = 0%; Analysis 19.9)
19.9. Analysis.

Comparison 19: Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data, Outcome 9: Mental health diagnoses
Imputed data. Comparing available data with intention‐to‐treat and no‐attrition data
No differences were found between subgroups: available data (SMD ‐0.13, 95% CI ‐0.40 to 0.13; 10 trials, 231 participants, I 2 = 0 %; P = 0.32), intention‐to‐treat (SMD ‐0.27, 95% CI ‐1.28 to 0.75;2 trials, 111 participants, I 2 = 79; P = 0.6), and no attrition (SMD ‐0.40, ‐0.71 to ‐0.08; 4 trials, 161 participants, I 2 = 0%; P = 0.01. Test for subgroup differences: Chi² = 1.59, df = 2 (P = 0.45), I² = 0%; Analysis 19.10)
19.10. Analysis.

Comparison 19: Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data, Outcome 10: Imputed data
Subgroup analyses for usual care
Type of active intervention (continuous data). Comparing psychological interventions to pharmacological, physical, and other or combination interventions
No differences were found between subgroups: psychological interventions (SMD ‐0.14, 95% CI ‐0.39 to 0.10; 4 trials, 346 participants; I 2 = 18%; P 0 0.25), physical interventions (SMD ‐1.10, 95% CI ‐1.42 to ‐0.79; 1 trial, 177 participants; I 2 = not applicable; P < 0.00001). Test for subgroup differences: Chi 2 = 22.02, df = 1 (P < 0.00001), I 2 = 95.5%; Analysis 20.1)
20.1. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 1: Type of active intervention
Type of active intervention (dichotomous data). Comparing psychological interventions to pharmacological, physical, and other or combination interventions
Test for subgroup differences: not applicable (Analysis 20.2)
20.2. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 2: Type of active intervention
Risk of bias (continuous data). Comparing trials of low risk of bias to trial of high risk of bias
Test for subgroup differences: not applicable (Analysis 20.3)
20.3. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 3: Risk of bias
Risk of bias (dichotomous data). Comparing trials of low risk of bias to trial of high risk of bias
Test for subgroup differences: not applicable (Analysis 20.4)
20.4. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 4: Risk of bias
Type of outcome domain (continuous data). Comparing blinded observer‐reported to non‐blinded observer‐reported and patient‐reported outcomes
No differences were found between subgroups: wait‐list (SMD ‐0.53, 95% CI ‐1.17 to 0.10; 3 trials, 443 participants; I 2 = 91%; P = 0.10), and no‐treatment (SMD 0.08, 95% CI ‐0.38 to 0.53; 2 trials, 80 participants; I 2 = 0%; P = 0.74). Test for subgroup differences: Chi 2 = 2.33, df = 1 (P = 0.13), I 2 = 57.1%; Analysis 20.6)
20.6. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 6: Mean age of participants
Mean age of participants (continuous data). Comparing below 18 years of age to 18 to 50 years of age and above 50 years of age
No differences were found between subgroups: below 18 years of age (SMD ‐1.10, 95% CI ‐1.42 to ‐0.79; 1 trial, 177 participants; I 2 = not applicable; P < 0.00001), 18 to 50 years of age (SMD ‐0.15, 95% CI ‐0.46 to 0.17; 3 trials, 316 participants; I 2 = 44%; P = 0.36), and above 50 years of age (SMD ‐0.06, 95% CI ‐0.82 to 0.70; 1 trial, 30 participants; I 2 = not applicable; P = 0.87). Test for subgroup differences: Chi 2 = 19.65, df = 2 (P < 0.00001), I 2 = 89.8%; Analysis 20.6)
Duration of treatment (continuous data). Comparing above three months of duration to below three months of duration
No differences were found between subgroups: above three months (SMD ‐0.53, 95% CI ‐1.17 to 0.10; 3 trials, 443 participants; I 2 = 91%; P = 0.10), and below three months (SMD 0.08, 95% CI ‐0.38 to 0.53; 2 trials, 80 participants; I 2 = 0%; P = 0.74). Test for subgroup differences: Chi 2 = 2.33, df = 1 (P = 0.13), I 2 = 57.1%;Analysis 20.7)
20.7. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 7: Duration of treatment
Duration of treatment (dichotomous data). Comparing above three months of duration to below three months of duration
Test for subgroup differences: not applicable (Analysis 20.8)
20.8. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 8: Duration of treatment
Type of usual care (continuous data). Comparing psychological usual care to pharmacological, physical, and other or combination usual care
No differences were found between subgroups: psychological usual care (SMD ‐0.44, 95% CI ‐0.99 to 0.10; 4 trials, 473 participants; I 2 = 87%; P = 0.11), and other or combination of usual care (SMD 0.15, 95% CI ‐0.41 to 0.72; 1 trial, 50 participants; I 2 = not applicable; P = 0.60). Test for subgroup differences: Chi 2 = 2.20, df = 1 (P = 0.14), I 2 = 54.6%; Analysis 20.9)
20.9. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 9: Type of usual care
Type of usual care (dichotomous data). Comparing psychological usual care to pharmacological, physical, and other or combination usual care
No differences were found between subgroups: psychological usual care (RR 0.50, 95% CI 0.20 to 1.26; 1 trial, 26 participants; I 2 = not applicable; P = 0.14), and other or combination of usual care (RR 1.42, 95% CI 1.17 to 1.73; 1 trial, 234 participants; I 2 = not applicable; P = 0.0005). Test for subgroup differences: Chi 2 = 4.72, df = 1 (P = 0.03), I 2 = 78.8%; Analysis 20.10)
20.10. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 10: Type of usual care
Standarised usual care (continuous data). Comparing trials of usual care intervention was intentionally standardised or manualised to trials of no standardisation or manualisation
No differences were found between subgroups: standardised or manualised (SMD ‐0.38, 95% CI ‐0.94 to 0.18; 4 trials, 493 participants; I 2 = 89%; P = 0.18), and no standardisation or manualisation (SMD ‐0.06, 95% CI ‐0.82 to 0.70; 1 trial, 30 participants; I 2 = not applicable; P = 0.87). Test for subgroup differences: Chi 2 = 0.43, df = 1 (P = 0.51), I 2 = 0%; Analysis 20.11)
20.11. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 11: Standarised usual care
Standarised usual care (dichotomous data). Comparing trials of usual care intervention was intentionally standardised or manualised to trials of no standardisation or manualisation
Test for subgroup differences: not applicable (Analysis 20.12)
20.12. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 12: Standarised usual care
Mode of psychological treatment in usual care (continuous data). Comparing individual treatment to group and a combination of both
No differences were found between subgroups: individual treatment (SMD ‐0.06, 95% CI ‐0.35 to 0.23; 2 trials, 185 participants; I 2 = 0%; P = 0.70), and group treatment (SMD ‐0.78, 95% CI ‐1.43 to ‐0.12; 2 trials, 288 participants; I 2 = 86%; P = 0.02). Test for subgroup differences: Chi 2 = 3.84, df = 2 (P = 0.05), I 2 = 73.9%; Analysis 20.13)
20.13. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 13: Mode of psychological treatment in usual care
Imputed data. Comparing available data with intention‐to‐treat and no‐attrition data
No differences were found between subgroups: available data (SMD ‐0.36, 95% CI ‐0.84 to 0.12; 2 trials, 80 participants, I 2 = 0%; P = 0.84), intention‐to‐treat (SMD ‐0.53, 95% CI ‐1.17 to 0.10; 3 trials, 443 participants, I 2 =91%; P = 0.10). Test for subgroup differences: Chi² = 1.76, df = 1 (P = 0.18), I² = 43.3%
Data and analyses
Comparison 1. Wait‐list versus no‐treatment for people with mental health disorder.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Wait‐list versus no‐treatment for people with mental health disorder | 1 | Other data | No numeric data |
1.1. Analysis.
Comparison 1: Wait‐list versus no‐treatment for people with mental health disorder, Outcome 1: Wait‐list versus no‐treatment for people with mental health disorder
| Wait‐list versus no‐treatment for people with mental health disorder | |
| Study | Heading 1 |
| Howlin 2007 | Only one cluster‐randomised trial compared a wait‐list intervention to a no‐treatment intervention was included ( Howlin 2007 ). However, no usable data was provided in the full report, and the authors did engage in correspondence. 84 elementary school children with a autism spectrum disorder was randomised to either, i) immediate treatment, ii) delayed treatment (wait‐list), and iii) no‐treatment. Conclusion were that Picture Exchange Communication System (PECS) training indicated modest effectiveness for children with autism spectrum disorder. In general there were no differences on across outcome measures between the wait‐list and no‐treatment intervention groups. |
Comparison 2. Usual care versus wait‐list or no‐treatment for all mental health disorders.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 2.1 Usual care compared with wait‐list/no‐treatment for continuous data | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.83, 0.16] |
| 2.1.1 Usual care compared with wait‐list | 3 | 443 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.53 [‐1.17, 0.10] |
| 2.1.2 Usual care compared with no‐treatment | 2 | 80 | Std. Mean Difference (IV, Random, 95% CI) | 0.08 [‐0.38, 0.53] |
| 2.2 Usual care compared with wait‐list/no‐treatment for dichotomous data | 2 | 260 | Risk Ratio (M‐H, Random, 95% CI) | 0.93 [0.34, 2.57] |
| 2.2.1 Usual care compared with wait‐list | 2 | 260 | Risk Ratio (M‐H, Random, 95% CI) | 0.93 [0.34, 2.57] |
| 2.2.2 Usual care compared with no‐treatment | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | Not estimable |
Comparison 6. All placebos versus waitl‐list or no‐treatment for people with mental health disorders.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 6.1 All placebos compared with wait‐list/no‐treatment for continuous data | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 6.1.1 All placebos compared with wait‐list | 31 | 1410 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.55 [‐0.76, ‐0.35] |
| 6.1.2 All placebos compared with no‐treatment | 34 | 1036 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.18 [‐0.30, ‐0.05] |
| 6.2 All placebos compared with wait‐list/no‐treatment for dichotomous data | 9 | 385 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.74, 1.48] |
| 6.2.1 All placebos compared with wait‐list | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | Not estimable |
| 6.2.2 All placebos compared with no‐treatment | 9 | 385 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.74, 1.48] |
Comparison 7. Serious adverse events of placebos versus wait‐list or no‐treatment for people with mental health disorders.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 7.1 All placebos compared with wait‐list/no‐treatment for dichotomous data | 11 | 517 | Risk Difference (IV, Random, 95% CI) | ‐0.00 [‐0.03, 0.03] |
| 7.2 Psychological placebos compared with wait‐list/no‐treatment for dichotomous data | 2 | 207 | Risk Difference (IV, Random, 95% CI) | ‐0.01 [‐0.07, 0.04] |
| 7.3 Pharmacological placebos compared with wait‐list/no‐treatment for dichotomous data | 4 | 125 | Risk Difference (M‐H, Random, 95% CI) | 0.01 [‐0.08, 0.09] |
| 7.4 Physical placebos compared with wait‐list/no‐treatment for dichotomous data | 5 | 185 | Risk Difference (M‐H, Random, 95% CI) | 0.00 [‐0.04, 0.04] |
Comparison 8. Non‐serious adverse events of all placebos versus wait‐list or no‐ treatment for people with mental health disorders.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 8.1 All placebos compared with wait‐list/no‐treatment for dichotomous data | 14 | Risk Difference (IV, Random, 95% CI) | 0.03 [‐0.02, 0.08] | |
| 8.1.1 All placebos compared with wait‐list | 7 | Risk Difference (IV, Random, 95% CI) | 0.02 [‐0.07, 0.10] | |
| 8.1.2 All placebos compared with no‐treatment | 7 | Risk Difference (IV, Random, 95% CI) | 0.04 [‐0.03, 0.10] | |
| 8.2 Psychological placebos compared with wait‐list/no‐treatment for dichotomous data | 5 | Risk Difference (IV, Random, 95% CI) | 0.01 [‐0.18, 0.19] | |
| 8.2.1 Psychological placebos compared with wait‐list | 5 | Risk Difference (IV, Random, 95% CI) | 0.01 [‐0.18, 0.19] | |
| 8.2.2 Psychological placebos compared with no‐treatment | 0 | Risk Difference (IV, Random, 95% CI) | Not estimable | |
| 8.3 Pharmacological placebos compared with wait‐list/no‐treatment for dichotomous data | 4 | 125 | Risk Difference (M‐H, Random, 95% CI) | 0.08 [‐0.04, 0.21] |
| 8.3.1 Pharmacological placebos compared with wait‐list | 0 | 0 | Risk Difference (M‐H, Random, 95% CI) | Not estimable |
| 8.3.2 Pharmacological placebos compared with no‐treatment | 4 | 125 | Risk Difference (M‐H, Random, 95% CI) | 0.08 [‐0.04, 0.21] |
| 8.4 Physical placebos compared with wait‐list/no‐treatment for dichotomous data | 5 | 185 | Risk Difference (M‐H, Random, 95% CI) | 0.00 [‐0.04, 0.04] |
| 8.4.1 Physical placebos compared with wait‐list | 2 | 111 | Risk Difference (M‐H, Random, 95% CI) | 0.00 [‐0.04, 0.04] |
| 8.4.2 Physical placebos compared with no‐treatment | 3 | 74 | Risk Difference (M‐H, Random, 95% CI) | 0.00 [‐0.09, 0.09] |
Comparison 9. Psychological placebos versus wait‐list or no‐treatment for people with mental health disorders.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 9.1 Psychological placebos compared with wait‐list/no‐treatment for continuous data | 38 | 1263 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐0.66, ‐0.31] |
| 9.1.1 Psychological placebos compared with wait‐list | 23 | 721 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.66 [‐0.92, ‐0.40] |
| 9.1.2 Psychological placebos compared with no‐treatment | 15 | 542 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.38, ‐0.04] |
| 9.2 Psychological placebos compared with wait‐list/no‐treatment for dichotomous data | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | Not estimable |
| 9.2.1 Psychological placebos compared with wait‐list | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | Not estimable |
| 9.2.2 Psychological placebos compared with no‐treatment | 1 | 19 | Risk Ratio (M‐H, Random, 95% CI) | Not estimable |
Comparison 10. Pharmacological placebos versus wait‐list or no‐treatment for people with mental health disorders.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 10.1 Pharmacological placebos compared with wait‐list/no‐treatment for continuous data | 9 | 279 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 10.1.1 Pharmacological placebos compared with wait‐list | 1 | 20 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.51 [‐1.41, 0.38] |
| 10.1.2 Pharmacological placebos compared with no‐treatment | 8 | 259 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.11 [‐0.37, 0.16] |
| 10.2 Pharmacological placebos compared with no‐treatment for continuous data | 8 | 366 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.74, 1.48] |
| 10.2.1 Pharmacological placebos compared with wait‐list | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | Not estimable |
| 10.2.2 Pharmacological placebos compared with no‐treatment | 8 | 366 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.74, 1.48] |
Comparison 11. Physical placebo versus wait‐list or no‐treatment for people with mental health disorders.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 11.1 Physical placebos compared with wait‐list/no‐treatment for continuous data | 17 | 896 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.08] |
| 11.1.1 Physical placebos compared with wait‐list | 7 | 669 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.30 [‐0.54, ‐0.06] |
| 11.1.2 Physical placebos compared with no‐treatment | 10 | 227 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.15 [‐0.42, 0.11] |
Comparison 12. All placebos versus wait‐list or no‐treatment for people with specific mental health disorders for continuous data.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 12.1 Efficacy of all placebos for people with substance use disorders | 4 | 136 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.15 [‐0.49, 0.19] |
| 12.1.1 All placebos compared with wait‐list | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 12.1.2 All placebos compared with no‐treatment | 4 | 136 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.15 [‐0.49, 0.19] |
| 12.2 Efficacy of all placebos for people with sleep‐wake disorders | 11 | 229 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.34 [‐0.60, ‐0.07] |
| 12.2.1 All placebos compared with wait‐list | 3 | 70 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.58 [‐1.10, ‐0.05] |
| 12.2.2 All placebos compared with no‐treatment | 8 | 159 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.24 [‐0.55, 0.08] |
| 12.3 Efficacy of all placebos for people with depression | 10 | 286 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.42 [‐0.78, ‐0.05] |
| 12.3.1 All placebos compared with wait‐list | 6 | 197 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.65 [‐1.15, ‐0.15] |
| 12.3.2 All placebos compared with no‐treatment | 4 | 89 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.05 [‐0.47, 0.37] |
| 12.4 Efficacy of all placebos for people with post‐traumatic stress disorder (PTSD) | 4 | 329 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.54 [‐1.06, ‐0.02] |
| 12.4.1 All placebos compared with wait‐list | 3 | 231 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.75 [‐1.23, ‐0.27] |
| 12.4.2 All placebos compared with no‐treatment | 1 | 98 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.08 [‐0.56, 0.40] |
| 12.5 Efficacy of all placebos for people with anxiety disorders | 16 | 401 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.57 [‐0.93, ‐0.21] |
| 12.5.1 All placebos compared with wait‐list | 6 | 181 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.81 [‐1.66, 0.05] |
| 12.5.2 All placebos compared with no‐treatment | 10 | 220 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.39 [‐0.66, ‐0.11] |
| 12.6 Efficacy of all placebos for people with attention deficit hyperactivity disorder (ADHD) or attention deficit disorder (ADD) | 5 | 145 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.31 [‐0.64, 0.02] |
| 12.6.1 All placebos compared with wait‐list | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 12.6.2 All placebos compared with no‐treatment | 5 | 145 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.31 [‐0.64, 0.02] |
| 12.7 Efficacy of all placebos for people with neurodegenerative disorders | 4 | 231 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.47, 0.05] |
| 12.7.1 All placebos compared with wait‐list | 2 | 134 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.67, 0.01] |
| 12.7.2 All placebos compared with no‐treatment | 2 | 97 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.03 [‐0.43, 0.36] |
Comparison 13. Psychological placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 13.1 Efficacy of all placebos for people with substance use disorders | 3 | 122 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.19 [‐0.54, 0.17] |
| 13.1.1 Psychological placebos compared with wait‐list | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 13.1.2 Psychological placebos compared with no‐treatment | 3 | 122 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.19 [‐0.54, 0.17] |
| 13.2 Efficacy of all placebos for people with sleep‐wake disorders | 7 | 159 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.44 [‐0.76, ‐0.12] |
| 13.2.1 Psychological placebos compared with wait‐list | 2 | 52 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.63 [‐1.49, 0.24] |
| 13.2.2 Psychological placebos compared with no‐treatment | 5 | 107 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.75, 0.02] |
| 13.3 Efficacy of all placebos for people with depression | 4 | 86 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.66 [‐1.56, 0.23] |
| 13.3.1 Psychological placebos compared with wait‐list | 3 | 66 | Std. Mean Difference (IV, Random, 95% CI) | ‐1.01 [‐1.82, ‐0.21] |
| 13.3.2 Psychological placebos compared with no‐treatment | 1 | 20 | Std. Mean Difference (IV, Random, 95% CI) | 0.34 [‐0.55, 1.22] |
| 13.4 Efficacy of all placebos for people with post‐traumatic stress disorder (PTSD) | 3 | 231 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.75 [‐1.23, ‐0.27] |
| 13.4.1 Psychological placebos compared with wait‐list | 3 | 231 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.75 [‐1.23, ‐0.27] |
| 13.4.2 Psychological placebos compared with no‐treatment | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 13.5 Efficacy of all placebos for people with anxiety disorders | 11 | 258 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.71 [‐1.19, ‐0.22] |
| 13.5.1 Psychological placebos compared with wait‐list | 6 | 157 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.94 [‐1.83, ‐0.05] |
| 13.5.2 Psychological placebos compared with no‐treatment | 5 | 101 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.43 [‐0.83, ‐0.04] |
Comparison 14. Pharmacological placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 14.1 Efficacy of all placebos for people with attention deficit hyperactivity disorder (ADHD) or attention deficit disorder (ADD) | 3 | 64 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.10 [‐0.59, 0.39] |
| 14.1.1 Pharmacological placebos compared with wait‐list | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 14.1.2 Pharmacological placebos compared with no‐treatment | 3 | 64 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.10 [‐0.59, 0.39] |
Comparison 15. Physical placebos versus wait‐list or no‐treatment for people with specific mental health disorders continuous data.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 15.1 Efficacy of all placebos for people with sleep‐wake disorders | 4 | 70 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.11 [‐0.58, 0.36] |
| 15.1.1 Physical placebos compared with wait‐list | 1 | 18 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.52 [‐1.46, 0.43] |
| 15.1.2 Physical placebos compared with no‐treatment | 3 | 52 | Std. Mean Difference (IV, Random, 95% CI) | 0.03 [‐0.52, 0.57] |
| 15.2 Efficacy of all placebos for people with depression | 4 | 162 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.24 [‐0.73, 0.25] |
| 15.2.1 Physical placebos compared with wait‐list | 2 | 111 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.27 [‐1.28, 0.75] |
| 15.2.2 Physical placebos compared with no‐treatment | 2 | 51 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.10 [‐0.65, 0.45] |
| 15.3 Efficacy of all placebos for people with anxiety disorders | 3 | 84 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.36 [‐0.82, 0.09] |
| 15.3.1 Physical placebos compared with wait‐list | 1 | 36 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.11 [‐0.82, 0.60] |
| 15.3.2 Physical placebos compared with no‐treatment | 2 | 48 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.54 [‐1.14, 0.06] |
Comparison 16. Subgroup analyses for all placebos compared with wait‐list or no‐treatment for continuous data.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 16.1 Type of active intervention | 64 | 2413 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.38 [‐0.50, ‐0.27] |
| 16.1.1 Psychological intervention | 38 | 1215 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.48 [‐0.66, ‐0.30] |
| 16.1.2 Pharmacological intervention | 7 | 148 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.29 [‐0.62, 0.04] |
| 16.1.3 Physical intervention | 13 | 798 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.07] |
| 16.1.4 Other or combination intervention | 6 | 252 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.32 [‐0.59, ‐0.04] |
| 16.2 Type of active intervention | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 16.2.1 Psychological intervention | 1 | 19 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 16.2.2 Pharmacological intervention | 7 | 316 | Risk Ratio (IV, Random, 95% CI) | 1.04 [0.74, 1.48] |
| 16.2.3 Physical intervention | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 16.2.4 Other or combination intervention | 1 | 50 | Risk Ratio (IV, Random, 95% CI) | 0.99 [0.58, 1.70] |
| 16.3 Risk of bias | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 16.3.1 Low risk of bias | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 16.3.2 High risk of bias | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 16.4 Risk of bias | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 16.4.1 Low risk of bias | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 16.4.2 High risk of bias | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 16.5 Type of outcome domain | 56 | 2205 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.38 [‐0.51, ‐0.24] |
| 16.5.1 Blinded observer‐reported | 29 | 1046 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.50 [‐0.70, ‐0.31] |
| 16.5.2 Non‐blinded observer‐reported | 2 | 96 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.54 [‐0.95, ‐0.13] |
| 16.5.3 Patient‐reported | 25 | 1063 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.19 [‐0.35, ‐0.03] |
| 16.6 Type of outcome domain | 5 | 189 | Risk Ratio (IV, Random, 95% CI) | 0.94 [0.73, 1.21] |
| 16.6.1 Blinded observer‐reported | 3 | 113 | Risk Ratio (IV, Random, 95% CI) | 0.99 [0.60, 1.64] |
| 16.6.2 Non‐blinded observer‐reported | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 16.6.3 Patient‐reported | 2 | 76 | Risk Ratio (IV, Random, 95% CI) | 1.00 [0.60, 1.65] |
| 16.7 Awareness of placebo intervention | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 16.7.1 Open placebo | 1 | 20 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.51 [‐1.41, 0.38] |
| 16.7.2 Closed placebo | 64 | 2426 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 16.8 Awareness of placebo intervention | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 16.8.1 Open placebo | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 16.8.2 Closed placebo | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 16.9 The trial objective | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 16.9.1 A trial’s objective is clearly to assess the effects of placebo | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 16.9.2 No such objectives are stated | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 16.10 The trial objective | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 16.10.1 A trial’s objective is clearly to assess the effects of placebo | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 16.10.2 No such objectives are stated | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 16.11 Mean age of participants | 57 | 2301 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.35 [‐0.47, ‐0.22] |
| 16.11.1 Below 18 years | 9 | 274 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.47 [‐0.98, 0.04] |
| 16.11.2 18 to 50 years | 40 | 1735 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.31 [‐0.44, ‐0.18] |
| 16.11.3 Above 50 years | 8 | 292 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.41 [‐0.75, ‐0.06] |
| 16.12 Mean age of participants | 7 | 340 | Risk Ratio (IV, Random, 95% CI) | 0.94 [0.70, 1.27] |
| 16.12.1 18 to 50 years | 5 | 270 | Risk Ratio (IV, Random, 95% CI) | 1.07 [0.70, 1.62] |
| 16.12.2 Above 50 years | 2 | 70 | Risk Ratio (IV, Random, 95% CI) | 0.73 [0.43, 1.27] |
| 16.13 Duration of treatment | 64 | 2366 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.38 [‐0.50, ‐0.26] |
| 16.13.1 Above 3 months | 11 | 464 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.68 [‐1.09, ‐0.27] |
| 16.13.2 Below 3 months | 53 | 1902 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.32 [‐0.43, ‐0.20] |
| 16.14 Duration of treatment | 8 | 366 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 16.14.1 Above 3 months | 4 | 196 | Risk Ratio (IV, Random, 95% CI) | 1.45 [0.84, 2.49] |
| 16.14.2 Below 3 months | 4 | 170 | Risk Ratio (IV, Random, 95% CI) | 0.82 [0.68, 0.98] |
| 16.15 Mental health diagnoses | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 16.15.1 Formal diagnosis according to DSM/ICD | 29 | 1256 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.42 [‐0.62, ‐0.21] |
| 16.15.2 Fulfil symptoms of disorder ICD/DSM while not stating systems | 14 | 326 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.30 [‐0.52, ‐0.08] |
| 16.15.3 Population is classified as having a mental disorder, but full diagnostic criteria not reported | 22 | 864 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.27 [‐0.42, ‐0.12] |
| 16.16 Mental health diagnoses | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 16.16.1 Formal diagnosis according to DSM/ICD | 4 | 220 | Risk Ratio (IV, Random, 95% CI) | 1.31 [0.72, 2.37] |
| 16.16.2 Fulfill symptoms of disorder ICD/DSM while not stating systems | 3 | 96 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.62, 1.63] |
| 16.16.3 Population is classified as having a mental disorder, but full diagnostic criteria not reported | 2 | 69 | Risk Ratio (IV, Random, 95% CI) | 0.80 [0.65, 0.99] |
| 16.17 Affiliation bias | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 16.17.1 Risk of affiliation, industry, and allegiance bias | 6 | 363 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.61 [‐0.95, ‐0.26] |
| 16.17.2 No risk found of affiliation, industry, and allegiance bias | 59 | 2083 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.45, ‐0.21] |
| 16.18 Risk of bias (participants and personnel excluded) | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 16.18.1 Low risk of bias | 3 | 230 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.58 [‐0.85, ‐0.32] |
| 16.18.2 Unclear risk of bias | 2 | 51 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.12 [‐0.95, 0.71] |
| 16.18.3 High risk of bias | 60 | 2165 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.36 [‐0.48, ‐0.23] |
| 16.19 Imputed data | 63 | 2416 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.36 [‐0.48, ‐0.24] |
| 16.19.1 Available data | 45 | 1236 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.39 [‐0.52, ‐0.25] |
| 16.19.2 Intent‐to‐treat | 9 | 891 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.22 [‐0.44, ‐0.01] |
| 16.19.3 No attrition | 9 | 289 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.50 [‐1.00, 0.00] |
Comparison 17. Subgroup analyses for psychological placebos compared with wait‐list or no‐treatment for continuous data.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 17.1 Type of active intervention | 38 | 1263 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐0.66, ‐0.31] |
| 17.1.1 Psychological intervention | 35 | 1174 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.48 [‐0.67, ‐0.29] |
| 17.1.2 Pharmacological intervention | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 17.1.3 Physical intervention | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 17.1.4 Other or combination intervention | 3 | 89 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.51 [‐0.94, ‐0.09] |
| 17.2 Risk of bias | 38 | 1263 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐0.66, ‐0.31] |
| 17.2.1 Low risk of bias | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 17.2.2 High risk of bias | 38 | 1263 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐0.66, ‐0.31] |
| 17.3 Type of outcome domain | 34 | 1515 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐0.68, ‐0.30] |
| 17.3.1 Blinded observer‐reported | 14 | 512 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.72 [‐1.08, ‐0.36] |
| 17.3.2 Non‐blinded observer‐reported | 2 | 96 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.54 [‐0.95, ‐0.13] |
| 17.3.3 Patient‐reported | 18 | 907 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.27 [‐0.47, ‐0.08] |
| 17.4 Awareness of placebo intervention | 38 | 1263 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐0.66, ‐0.31] |
| 17.4.1 Open placebo | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 17.4.2 Closed placebo | 38 | 1263 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐0.66, ‐0.31] |
| 17.5 The trial objective | 38 | 1263 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐0.66, ‐0.31] |
| 17.5.1 A trial’s objective is clearly to assess the effects of placebo | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 17.5.2 No such objectives are stated | 38 | 1263 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐0.66, ‐0.31] |
| 17.6 Mean age of participants | 33 | 1537 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.45 [‐0.64, ‐0.26] |
| 17.6.1 Below 18 years | 4 | 148 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.85 [‐2.02, 0.32] |
| 17.6.2 18 to 50 years | 25 | 1233 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.38 [‐0.55, ‐0.21] |
| 17.6.3 Above 50 years | 4 | 156 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.59 [‐1.33, 0.14] |
| 17.7 Duration of treatment | 38 | 1576 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.48 [‐0.66, ‐0.31] |
| 17.7.1 Above 3 months | 6 | 270 | Std. Mean Difference (IV, Random, 95% CI) | ‐1.02 [‐1.68, ‐0.36] |
| 17.7.2 Below 3 months | 32 | 1306 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.38 [‐0.53, ‐0.22] |
| 17.8 Type of psychological placebo | 38 | 1263 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐0.66, ‐0.31] |
| 17.8.1 Interaction placebo | 15 | 613 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.70 [‐1.04, ‐0.36] |
| 17.8.2 Psychoeducational placebo | 9 | 329 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.36, 0.07] |
| 17.8.3 Exposure placebo | 14 | 321 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.48 [‐0.71, ‐0.24] |
| 17.9 Mode of psychological placebo | 35 | 1571 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.48 [‐0.66, ‐0.30] |
| 17.9.1 Individual treatment | 19 | 1151 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.31 [‐0.49, ‐0.12] |
| 17.9.2 Group treatment | 13 | 308 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.85 [‐1.30, ‐0.39] |
| 17.9.3 Combination of individual and group | 3 | 112 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.45 [‐0.83, ‐0.08] |
| 17.10 Mental health diagnoses | 39 | 1656 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.47 [‐0.64, ‐0.30] |
| 17.10.1 Formal diagnosis according to DSM/ICD | 16 | 767 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.53 [‐0.86, ‐0.21] |
| 17.10.2 Fulfill symptoms of disorder ICD/DSM while not stating systems | 9 | 218 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.39 [‐0.66, ‐0.12] |
| 17.10.3 Population is classified as having a mental disorder, but full diagnostic criteria not reported | 14 | 671 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.59, ‐0.15] |
| 17.11 Affiliation bias | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 17.11.1 Risk of affiliation, industry, allegiance bias | 6 | 363 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.61 [‐0.95, ‐0.26] |
| 17.11.2 No risk of affiliation, industry, allegiance bias | 59 | 2083 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.45, ‐0.21] |
| 17.12 Imputed data | 37 | 1626 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.46 [‐0.63, ‐0.29] |
| 17.12.1 Available data | 27 | 842 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.50 [‐0.68, ‐0.33] |
| 17.12.2 Intent‐to‐treat | 6 | 682 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.18 [‐0.39, 0.03] |
| 17.12.3 No attrition | 4 | 102 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.75 [‐2.11, 0.62] |
Comparison 18. Subgroup analyses for pharmacological placebos compared with wait‐list or no‐treatment for continuous data.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 18.1 Type of active intervention | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 18.1.1 Psychological intervention | 1 | 33 | Std. Mean Difference (IV, Random, 95% CI) | 0.44 [‐0.26, 1.13] |
| 18.1.2 Pharmacological intervention | 7 | 148 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.29 [‐0.62, 0.04] |
| 18.1.3 Physical intervention | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 18.1.4 Other or combination intervention | 1 | 98 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.08 [‐0.56, 0.40] |
| 18.2 Type of active intervention | 8 | 366 | Risk Ratio (M‐H, Random, 95% CI) | 1.05 [0.74, 1.48] |
| 18.2.1 Psychological intervention | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | Not estimable |
| 18.2.2 Pharmacological intervention | 7 | 316 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.71, 1.67] |
| 18.2.3 Physical intervention | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | Not estimable |
| 18.2.4 Other or combination intervention | 1 | 50 | Risk Ratio (M‐H, Random, 95% CI) | 0.99 [0.58, 1.70] |
| 18.3 Risk of bias | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 18.3.1 Low risk of bias | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 18.3.2 High risk of bias | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 18.4 Risk of bias | 8 | 366 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 18.4.1 Low risk of bias | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 18.4.2 High risk of bias | 8 | 366 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 18.5 Type of outcome domain | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 18.5.1 Blinded observer‐reported | 7 | 220 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.24 [‐0.53, 0.05] |
| 18.5.2 Non‐blinded observer‐reported | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 18.5.3 Patient‐reported | 2 | 59 | Std. Mean Difference (IV, Random, 95% CI) | 0.18 [‐0.36, 0.73] |
| 18.6 Type of outcome domain | 4 | 170 | Risk Ratio (IV, Random, 95% CI) | 0.94 [0.73, 1.21] |
| 18.6.1 Blinded observer‐reported | 2 | 94 | Risk Ratio (IV, Random, 95% CI) | 0.99 [0.60, 1.64] |
| 18.6.2 Non‐blinded observer‐reported | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 18.6.3 Patient‐reported | 2 | 76 | Risk Ratio (IV, Random, 95% CI) | 1.00 [0.60, 1.65] |
| 18.7 Awareness of placebo intervention | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 18.7.1 Open placebo | 1 | 20 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.51 [‐1.41, 0.38] |
| 18.7.2 Closed placebo | 8 | 259 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.11 [‐0.37, 0.16] |
| 18.8 Awareness of placebo intervention | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 18.8.1 Open placebo | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 18.8.2 Closed placebo | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 18.9 The trial objective | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 18.9.1 A trial’s objective is clearly to assess the effects of placebo | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 18.9.2 No such objectives are stated | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 18.10 The trial objective | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 18.10.1 A trial’s objective is clearly to assess the effects of placebo | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 18.10.2 No such objectives are stated | 9 | 385 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 18.11 Mean age of participants | 7 | 241 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.13 [‐0.41, 0.14] |
| 18.11.1 Below 18 years | 3 | 64 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.10 [‐0.59, 0.39] |
| 18.11.2 18 to 50 years | 4 | 177 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.19 [‐0.68, 0.31] |
| 18.11.3 Above 50 years | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 18.12 Mean age of participants | 7 | 340 | Risk Ratio (IV, Random, 95% CI) | 0.94 [0.70, 1.27] |
| 18.12.1 Below 18 years | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 18.12.2 18 to 50 years | 5 | 270 | Risk Ratio (IV, Random, 95% CI) | 1.07 [0.70, 1.62] |
| 18.12.3 Above 50 years | 2 | 70 | Risk Ratio (IV, Random, 95% CI) | 0.73 [0.43, 1.27] |
| 18.13 Duration of treatment | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 18.13.1 Above 3 months | 4 | 164 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.56, 0.14] |
| 18.13.2 Below 3 months | 5 | 115 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.06 [‐0.43, 0.31] |
| 18.14 Duration of treatment | 8 | 366 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.76, 1.35] |
| 18.14.1 Above 3 months | 4 | 196 | Risk Ratio (IV, Random, 95% CI) | 1.45 [0.84, 2.49] |
| 18.14.2 Below 3 months | 4 | 170 | Risk Ratio (IV, Random, 95% CI) | 0.82 [0.68, 0.98] |
| 18.15 Mental health diagnoses | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 18.15.1 Formal diagnosis according to DSM/ICD | 5 | 188 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.25 [‐0.57, 0.06] |
| 18.15.2 Fulfill symptoms of disorder ICD/DSM while not stating systems | 2 | 40 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.07 [‐0.69, 0.56] |
| 18.15.3 Population is classified as having a mental disorder, but full diagnostic criteria not reported | 2 | 51 | Std. Mean Difference (IV, Random, 95% CI) | 0.12 [‐0.62, 0.86] |
| 18.16 Mental health diagnoses | 7 | 316 | Risk Ratio (IV, Random, 95% CI) | 1.12 [0.79, 1.59] |
| 18.16.1 Formal diagnosis according to DSM/ICD | 4 | 220 | Risk Ratio (IV, Random, 95% CI) | 1.31 [0.72, 2.37] |
| 18.16.2 Fulfill symptoms of disorder ICD/DSM while not stating systems | 3 | 96 | Risk Ratio (IV, Random, 95% CI) | 1.01 [0.62, 1.63] |
| 18.16.3 Population is classified as having a mental disorder, but full diagnostic criteria not reported | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 18.17 Imputed data | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 18.17.1 Available data | 7 | 155 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.17 [‐0.49, 0.16] |
| 18.17.2 Intent‐to‐treat | 1 | 98 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.08 [‐0.56, 0.40] |
| 18.17.3 No attrition | 1 | 26 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.12 [‐0.89, 0.65] |
Comparison 19. Subgroup analyses for physical placebos compared with wait‐list or no‐treatment for continuous data.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 19.1 Type of active intervention | 17 | 896 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.08] |
| 19.1.1 Psychological intervention | 2 | 33 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.26 [‐0.95, 0.43] |
| 19.1.2 Pharmacological intervention | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 19.1.3 Physical intervention | 13 | 798 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.07] |
| 19.1.4 Other or combination intervention | 2 | 65 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.30 [‐0.82, 0.23] |
| 19.2 Risk of bias | 17 | 896 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.08] |
| 19.2.1 Low risk of bias | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 19.2.2 High risk of bias | 17 | 896 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.08] |
| 19.3 Type of outcome domain | 13 | 411 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.31 [‐0.51, ‐0.12] |
| 19.3.1 Blinded observer‐reported | 8 | 314 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.42 [‐0.64, ‐0.19] |
| 19.3.2 Non‐blinded observer‐reported | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 19.3.3 Patient‐reported | 5 | 97 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.00 [‐0.40, 0.40] |
| 19.4 Awareness of placebo intervention | 17 | 896 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.08] |
| 19.4.1 Open placebo | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 19.4.2 Closed placebo | 17 | 896 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.08] |
| 19.5 The trial objective | 17 | 896 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.08] |
| 19.5.1 A trial’s objective is clearly to assess the effects of placebo | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 19.5.2 No such objectives are stated | 17 | 896 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.08] |
| 19.6 Mean age of participants | 16 | 503 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.30 [‐0.48, ‐0.12] |
| 19.6.1 Below 18 years | 2 | 62 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.32 [‐0.82, 0.19] |
| 19.6.2 18 to 50 years | 11 | 325 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.26 [‐0.48, ‐0.04] |
| 19.6.3 Above 50 years | 3 | 116 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.40 [‐0.77, ‐0.03] |
| 19.7 Duration of treatment | 17 | 926 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.22 [‐0.35, ‐0.09] |
| 19.7.1 Above 3 months | 1 | 30 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.48 [‐1.21, 0.25] |
| 19.7.2 Below 3 months | 17 | 896 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.35, ‐0.08] |
| 19.8 Type of physical placebo | 15 | 473 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.31 [‐0.50, ‐0.13] |
| 19.8.1 Acupuncture or acupressure placebo | 5 | 237 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.42 [‐0.73, ‐0.11] |
| 19.8.2 Excercise and relaxation placebo | 3 | 67 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.70, 0.27] |
| 19.8.3 Technical device placebo | 5 | 119 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.23 [‐0.60, 0.14] |
| 19.8.4 Electromagnetic stimulation placebo | 2 | 50 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.02 [‐0.57, 0.54] |
| 19.9 Mental health diagnoses | 16 | 503 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.30 [‐0.48, ‐0.12] |
| 19.9.1 Formal diagnosis according to DSM/ICD | 7 | 293 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.29 [‐0.56, ‐0.02] |
| 19.9.2 Fulfill symptoms of disorder ICD/DSM while not stating systems | 3 | 68 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.15 [‐0.62, 0.33] |
| 19.9.3 Population is classified as having a mental disorder, but full diagnostic criteria not reported | 6 | 142 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.32 [‐0.66, 0.02] |
| 19.10 Imputed data | 16 | 503 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.30 [‐0.48, ‐0.12] |
| 19.10.1 Available data | 10 | 231 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.13 [‐0.40, 0.13] |
| 19.10.2 Intent‐to‐treat | 2 | 111 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.27 [‐1.28, 0.75] |
| 19.10.3 No attrition | 4 | 161 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.40 [‐0.71, ‐0.08] |
Comparison 20. Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 20.1 Type of active intervention | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.83, 0.16] |
| 20.1.1 Psychological intervention | 4 | 346 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.10] |
| 20.1.2 Pharmacological intervention | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 20.1.3 Physical intervention | 1 | 177 | Std. Mean Difference (IV, Random, 95% CI) | ‐1.10 [‐1.42, ‐0.79] |
| 20.1.4 Other or combination intervention | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 20.2 Type of active intervention | 2 | 260 | Risk Ratio (IV, Random, 95% CI) | 0.93 [0.34, 2.55] |
| 20.2.1 Psychological intervention | 2 | 260 | Risk Ratio (IV, Random, 95% CI) | 0.93 [0.34, 2.55] |
| 20.2.2 Pharmacological intervention | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 20.2.3 Physical intervention | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 20.2.4 Other intervention | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 20.3 Risk of bias | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.83, 0.16] |
| 20.3.1 Low risk of bias | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 20.3.2 High risk of bias | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.83, 0.16] |
| 20.4 Risk of bias | 2 | 260 | Risk Ratio (IV, Random, 95% CI) | 0.93 [0.34, 2.55] |
| 20.4.1 Low risk of bias | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 20.4.2 High risk of bias | 2 | 260 | Risk Ratio (IV, Random, 95% CI) | 0.93 [0.34, 2.55] |
| 20.5 Type of outcome domain | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.83, 0.16] |
| 20.5.1 Blinded observer‐reported | 4 | 473 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.44 [‐0.99, 0.10] |
| 20.5.2 Non‐blinded observer‐reported | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 20.5.3 Patient‐reported | 1 | 50 | Std. Mean Difference (IV, Random, 95% CI) | 0.15 [‐0.41, 0.72] |
| 20.6 Mean age of participants | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.83, 0.16] |
| 20.6.1 Below 18 years | 1 | 177 | Std. Mean Difference (IV, Random, 95% CI) | ‐1.10 [‐1.42, ‐0.79] |
| 20.6.2 18 to 50 years | 3 | 316 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.15 [‐0.46, 0.17] |
| 20.6.3 Above 50 years | 1 | 30 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.06 [‐0.82, 0.70] |
| 20.7 Duration of treatment | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.36 [‐0.84, 0.12] |
| 20.7.1 Above 3 months | 3 | 443 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.53 [‐1.17, 0.10] |
| 20.7.2 Below 3 months | 2 | 80 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.00 [‐0.46, 0.45] |
| 20.8 Duration of treatment | 2 | 260 | Risk Ratio (IV, Random, 95% CI) | 0.93 [0.34, 2.55] |
| 20.8.1 Above 3 months | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 20.8.2 Below 3 months | 2 | 260 | Risk Ratio (IV, Random, 95% CI) | 0.93 [0.34, 2.55] |
| 20.9 Type of usual care | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.83, 0.16] |
| 20.9.1 Psychological usual care | 4 | 473 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.44 [‐0.99, 0.10] |
| 20.9.2 Pharmacological usual care | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 20.9.3 Physical usual care | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 20.9.4 Other or a combination usual care | 1 | 50 | Std. Mean Difference (IV, Random, 95% CI) | 0.15 [‐0.41, 0.72] |
| 20.10 Type of usual care | 2 | 260 | Risk Ratio (IV, Random, 95% CI) | 0.93 [0.34, 2.55] |
| 20.10.1 Psychological usual care | 1 | 26 | Risk Ratio (IV, Random, 95% CI) | 0.50 [0.20, 1.26] |
| 20.10.2 Pharmacological usual care | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 20.10.3 Physical usual care | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 20.10.4 Other or a combination usual care | 1 | 234 | Risk Ratio (IV, Random, 95% CI) | 1.42 [1.17, 1.73] |
| 20.11 Standarised usual care | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.83, 0.16] |
| 20.11.1 The usual care intervention was intentionally standardised or manualised | 4 | 493 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.38 [‐0.94, 0.18] |
| 20.11.2 No standardisation or manualisation | 1 | 30 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.06 [‐0.82, 0.70] |
| 20.12 Standarised usual care | 2 | 260 | Risk Ratio (IV, Random, 95% CI) | 0.93 [0.34, 2.55] |
| 20.12.1 The usual care intervention was intentionally standardised or manualised | 0 | 0 | Risk Ratio (IV, Random, 95% CI) | Not estimable |
| 20.12.2 No standardisation or manualisation | 2 | 260 | Risk Ratio (IV, Random, 95% CI) | 0.93 [0.34, 2.55] |
| 20.13 Mode of psychological treatment in usual care | 4 | 473 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.44 [‐0.99, 0.10] |
| 20.13.1 Individual treatment | 2 | 185 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.06 [‐0.35, 0.23] |
| 20.13.2 Group treatment | 2 | 288 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.78 [‐1.43, ‐0.12] |
| 20.13.3 Combination of individual and group | 0 | 0 | Std. Mean Difference (IV, Random, 95% CI) | Not estimable |
| 20.14 Imputed data | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.36 [‐0.84, 0.12] |
| 20.14.1 Available data | 2 | 80 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.00 [‐0.46, 0.45] |
| 20.14.2 Intent‐to‐treat | 3 | 443 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.53 [‐1.17, 0.10] |
20.5. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 5: Type of outcome domain
20.14. Analysis.

Comparison 20: Subgroup analyses for usual care compared with wait‐list or no‐treatment for continuous data, Outcome 14: Imputed data
Comparison 21. Sensitivity analyses for all placebos compared with wait‐list or no‐treatment.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 21.1 Fixed effects | 65 | 2446 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.33 [‐0.42, ‐0.25] |
| 21.2 Fixed effects | 9 | 385 | Risk Ratio (IV, Fixed, 95% CI) | 0.91 [0.77, 1.07] |
| 21.3 End of intervention (post‐treatment scores, changes scores excluded) | 65 | 2446 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.37 [‐0.49, ‐0.25] |
| 21.4 Type of data collection | 14 | Risk Difference (IV, Random, 95% CI) | 0.03 [‐0.02, 0.08] | |
| 21.4.1 Structured list of reporting | 1 | Risk Difference (IV, Random, 95% CI) | ‐1.54 [‐2.97, ‐0.11] | |
| 21.4.2 Spontaneous reporting | 13 | Risk Difference (IV, Random, 95% CI) | 0.03 [‐0.02, 0.07] | |
| 21.5 Transformed data for TSA analysis | 65 | 2456 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.34 [‐0.44, ‐0.23] |
21.1. Analysis.

Comparison 21: Sensitivity analyses for all placebos compared with wait‐list or no‐treatment, Outcome 1: Fixed effects
21.2. Analysis.

Comparison 21: Sensitivity analyses for all placebos compared with wait‐list or no‐treatment, Outcome 2: Fixed effects
21.3. Analysis.

Comparison 21: Sensitivity analyses for all placebos compared with wait‐list or no‐treatment, Outcome 3: End of intervention (post‐treatment scores, changes scores excluded)
21.4. Analysis.

Comparison 21: Sensitivity analyses for all placebos compared with wait‐list or no‐treatment, Outcome 4: Type of data collection
21.5. Analysis.

Comparison 21: Sensitivity analyses for all placebos compared with wait‐list or no‐treatment, Outcome 5: Transformed data for TSA analysis
Comparison 22. Sensitivity analyses for psychological placebo compared with wait‐list or no‐treatment.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 22.1 Fixed effects | 39 | 1656 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.37 [‐0.47, ‐0.27] |
| 22.2 End of intervention (post‐treatment scores) | 39 | 1656 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.47 [‐0.64, ‐0.30] |
| 22.3 Transformed data for TSA analysis | 38 | 1263 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.44 [‐0.59, ‐0.29] |
22.1. Analysis.

Comparison 22: Sensitivity analyses for psychological placebo compared with wait‐list or no‐treatment, Outcome 1: Fixed effects
22.2. Analysis.

Comparison 22: Sensitivity analyses for psychological placebo compared with wait‐list or no‐treatment, Outcome 2: End of intervention (post‐treatment scores)
22.3. Analysis.

Comparison 22: Sensitivity analyses for psychological placebo compared with wait‐list or no‐treatment, Outcome 3: Transformed data for TSA analysis
Comparison 23. Sensitivity analyses for pharmacological placebo compared with wait‐list or no‐treatment.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 23.1 Fixed effects | 9 | 279 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 23.2 Fixed effects | 8 | 366 | Risk Ratio (IV, Fixed, 95% CI) | 0.91 [0.77, 1.07] |
| 23.3 End of intervention (post‐treatment scores) | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.14 [‐0.39, 0.11] |
| 23.4 Transformed data for TSA analysis | 9 | 279 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.08 [‐0.33, 0.17] |
23.1. Analysis.

Comparison 23: Sensitivity analyses for pharmacological placebo compared with wait‐list or no‐treatment, Outcome 1: Fixed effects
23.2. Analysis.

Comparison 23: Sensitivity analyses for pharmacological placebo compared with wait‐list or no‐treatment, Outcome 2: Fixed effects
23.3. Analysis.

Comparison 23: Sensitivity analyses for pharmacological placebo compared with wait‐list or no‐treatment, Outcome 3: End of intervention (post‐treatment scores)
23.4. Analysis.

Comparison 23: Sensitivity analyses for pharmacological placebo compared with wait‐list or no‐treatment, Outcome 4: Transformed data for TSA analysis
Comparison 24. Sensitivity analyses for physical placebo compared with wait‐list or no‐treatment.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 24.1 Fixed effects | 16 | 503 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.30 [‐0.48, ‐0.12] |
| 24.2 End of intervention (post‐treatment scores) | 16 | 503 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.30 [‐0.48, ‐0.12] |
| 24.3 Transformed data for TSA analysis | 17 | 896 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.21 [‐0.34, ‐0.07] |
24.1. Analysis.

Comparison 24: Sensitivity analyses for physical placebo compared with wait‐list or no‐treatment, Outcome 1: Fixed effects
24.2. Analysis.

Comparison 24: Sensitivity analyses for physical placebo compared with wait‐list or no‐treatment, Outcome 2: End of intervention (post‐treatment scores)
24.3. Analysis.

Comparison 24: Sensitivity analyses for physical placebo compared with wait‐list or no‐treatment, Outcome 3: Transformed data for TSA analysis
Comparison 25. Sensitivity analyses for usual care compared with wait‐list or no‐treatment.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 25.1 Fixed effects | 5 | 523 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.46 [‐0.64, ‐0.28] |
| 25.2 Transformed data for TSA analysis | 5 | 523 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.32 [‐0.78, 0.14] |
25.2. Analysis.

Comparison 25: Sensitivity analyses for usual care compared with wait‐list or no‐treatment, Outcome 2: Transformed data for TSA analysis
Comparison 26. Sensitivity analysis for serious adverse events of placebos.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 26.1 Data in RR for TSA analysis | 11 | 517 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.25, 1.53] |
26.1. Analysis.

Comparison 26: Sensitivity analysis for serious adverse events of placebos, Outcome 1: Data in RR for TSA analysis
Comparison 27. Sensitivity analysis for non‐serious adverse events of placebos.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 27.1 Data in RR for TSA analysis | 11 | 362 | Odds Ratio (M‐H, Random, 95% CI) | 0.71 [0.27, 1.84] |
27.1. Analysis.

Comparison 27: Sensitivity analysis for non‐serious adverse events of placebos, Outcome 1: Data in RR for TSA analysis
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Abikoff 2004.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 1 year Duration of participation (trial + follow‐up): 1 year + 1 year follow‐up (switched to placebo) Setting: outpatient Purpose of trial: “To test that methylphenidate combined with intensive multimodal psychosocial intervention, which includes social skills training, significantly enhances social functioning in children with attention‐deficit/hyperactivity disorder (ADHD) compared with methylphenidate alone and methylphenidate plus nonspecific psychosocial treatment (attention control)” (p.820) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 332 Number of participants included: 103 Number of participants followed‐up at post treatment: 86 (after 1 year) Number of participants randomly assigned to:
Number of withdrawals: n = 17
Diagnosis: Attention Deficit Hyperactivity Disorder (ADHD) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd edition, revised (DSM‐III‐R) Means of assessment: the Diagnostic Interview Schedule for Children (DISC‐IV) Comorbidity: majority (55 of 103, 53.4%) met criteria for oppositional defiant disorder and 31 (30%) of 103 had one or two symptoms of conduct disorder. Relatively few (17 of 103, 16.5%) had an anxiety disorder (simple phobia, overanxious disorder, separation anxiety disorder) or major depression (four of 103, 3.9%) Age: 8.2 mean years (SD = 0.8) (Range 7 to 9.9) IQ: exclusion IQ less than 85. Mean WISC IQs were full scale, 109.5 (SD =14.5); verbal, 108.5, SD =4.0); and performance, 108.7 (SD =15.0) Sex: 7% female Ethnicity: 84% white, 13%, African American, 2% Hispanic, and 1% other. Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: attention control psychosocial treatment + methylphenidate (MPH) Description of intervention: “The attention control was designed to account for nonspecific treatment effects of the MPT intervention, including professional time and attention, extended interactions with peers, and parental attention. It contained components parallel to those of MPT but excluded specific remedial or therapeutic content.” ( Klein 2004 , p., 797) Individual or group treatment: group and Individual. Exposure/intensity to treatment: the groups’ mean daily methylphenidate dose did not differ at the end of year I, or year II The percentage of positive ritalinic acid assays was 87%, without differences between groups. ( Klein 2004 , p. 799) Duration of treatment: 1 year Concomitant psychotherapy: not stated Concomitant pharmacotherapy: all received MPH. Otherwise not stated No‐treatment Comparison name: methylphenidate (MPH) only (no‐treatment) Description of intervention: not stated Exposure/intensity to treatment: "The groups’ mean daily methylphenidate dose did not differ at the end of year I or year II. The percentage of positive ritalinic acid assays was 87%, without differences between groups.” ( Klein 2004 , p. 799) Duration treatment: 1 year Concomitant psychotherapy: not stated Concomitant pharmacotherapy: all got MPH. Otherwise not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest Potential industry bias: Disclosure: Dr. Abikoff is a member of the ADHD Advisory Board and a principal investigator in clinical trials, Shire Pharmaceutical Co., and a member of the Metadate CD Advisory Board of Celltech Pharmaceuticals. He is a recipient of an investigator‐initiated grant from McNeil Consumer and Specialty Pharmaceuticals. Dr. Hechtman received research funding from Eli Lilly, Janssen Ortho, Purdue,Shire Pharmaceutical Co., and GlaxoSmithKline Beecham and is on the speakers roster of Shire Pharmaceutical Co., Janssen Ortho, and Eli Lilly. Dr Klein is a member of the ADHD Advisory Board of Shire Pharmaceutical Co Judgment: no |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: “Children were randomly assigned to one of three treatments for 2 years: (1) methylphenidate alone (M), (2) methylphenidate and MPT (M + MPT), or (3) methylphenidate and attention control psychosocial treatment (M + ACT). Groups were balanced for ethnicity, sex, IQ, and oppositional defiant disorder. Assignment was done in blocks of four to enable group treatment components.” (p. 821) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Parents not blinded |
| Blinding of participants and personnel | No | Not possible to placebo and "no‐treatment" |
| Incomplete outcome data | No | Attrition >15% (21.4%). No ITT |
| Selective outcome reported | No | Differences in outcomes (adverse events) from the published description (Klein 2004) of the methods and full report |
| Other sources of bias | Yes | None found |
Allen 1998.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 8 weeks. Duration of participation (trial + follow‐up): no follow‐up Setting: outpatient Purpose of trial: “This design allowed us to test whether acupuncture designed to specifically treat symptoms of depression would demonstrate efficacy compared with a wait‐list control and nonspecific acupuncture treatments.” (p. 397) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 38 Number of participants followed‐up at post treatment: 34 Number of participants randomly assigned to: 38
Number of withdrawals: n = 4 (5 dropped out of the study but one of them had completed non‐specific treatment and is therefore included in analyses, p. 398).
Diagnosis: major depressive disorder Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM‐IV) Means of assessment: Structured Clinical Interview for DSM‐III‐R. (SCID‐R) Comorbidity: not stated Age: not stated (age between 18 and 45 years) IQ: not stated Sex: 100% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo
Treatment name: nonspecific treatment group
Description of intervention: acupuncture ‐ “(…) a placebo‐like treatment designed to treat a pattern of disharmony that was not related to the individual’s depression, but was characteristic of the individual.” (p. 398)
“Patients in the nonspecific‐treatment group received 8 weeks of nonspecific treatment first, and then 8 weeks of specific treatment.” (p. 398)
Individual or group treatment: individual
Exposure/intensity to treatment: “Each 8‐week treatment regimen (both specific and nonspecific) comprised 12 treatment sessions: 2 sessions a week for the first 4 weeks followed by 1 per week thereafter.” (p. 398)
Duration of treatment: 8 weeks
Concomitant psychotherapy: no – excluded if any current treatment
Concomitant pharmacotherapy: no – excluded if any current treatment Wait‐list Comparison name: wait‐list Description of intervention: “Patients in the wait‐list group waited 8 weeks before receiving 8 weeks of specific treatment.” (p. 398) Exposure/intensity to treatment: no treatment during the 8 weeks Duration treatment: 8 weeks. Concomitant psychotherapy : no – excluded if any current treatment Concomitant pharmacotherapy: no – excluded if any current treatment |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion
Key limitations
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “Patients were randomly assigned to one of three conditions (specific treatment: n=12, nonspecific treatment: n=11, or wait list: n=11.” (p. 398) Quote: “In 1996, unstratified randomization was implemented by creating a master randomized order in advance” (author correspondence) |
| Allocation concealment | Yes | Quote: "this was concealed until each new subject needed to be randomized.” ( Allen 1998 (pers comm) ) |
| Blinding of outcome assessors | Yes | ”Quote: All patients were interviewed by trained raters blind to treatment condition using the previously described 31‐item version of the HRSD.” (p. 399) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Quote: “To determine whether the results would be the same for the original intent‐to‐treat sample, we conducted two analyses. The simple intent‐to‐treat analysis took the last available DepHRSD score of each participant who dropped out and carried that score forward to count as the subsequent observation (…) The second strategy used a random regression model (Gibbons et al., 1993, implemented with BMDP 5V), which imputes missing values based on maximum‐likelihood estimates of missing parameters, thereby allowing for the analysis of all intent‐to‐treat subjects.” (p. 399) Attrition rate <15% (Specific treatment: 14%, Nonspecific treatment: 8%, Wait‐list: 8%). ITT used, but seems that they excluded non‐completers from reported data. |
| Selective outcome reported | Unclear | No trial registry was made ( Allen 1998 (pers comm) ) |
| Other sources of bias | Yes | No other sources found |
Allen 2006.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised : no Duration of trial (baseline to post): 8 weeks. Duration of participation (trial + follow‐up): “No follow‐up data. Patients in WL and non‐specific received specific treatment after 8 weeks" Setting: outpatient Purpose of trial: “The current study sought to test the efficacy of acupuncture as a monotherapy for MDD in a large randomized controlled trial of both men and women with a range in the severity of MDD by comparing the efficacy of acupuncture intervention specifically designed to target each individual’s depressive symptoms with an active valid acupuncture control that was not tailored to address an individual’s symptoms of depression and with a wait‐list control.” (p. 1666). Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened : 2965 Number of participants included: 157 Number of participants followed‐up at post treatment : 131 Number of participants randomly assigned to:
Number of withdrawals: n = 26
Diagnosis: major depressive disorder (MDD) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM‐IV) Means of assessment: Structured Clinical Interview for DSM‐IV (SCID‐R) Comorbidity: not stated Age: 42.1 mean years (SD = 11.0). (Range = 18 to 65) IQ: not stated Sex : 66.2% female Ethnicity: physical placebo: 88% white, wait‐list: 81% white, active treatment: 79% white. Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo
Treatment name (type): nonspecific acupuncture
Description of intervention: “A placebo‐like control intervention utilized a comparable number of valid acupuncture points that were not designed to treat the individual’s depression.” (p. 1667)
“Following the initial 8 weeks in 1 of these 3 intervention groups, all patients received SPEC intervention for the next 8 weeks.” (p. 1668)
Individual or group treatment: individual
Exposure/intensity to treatment: “Twice per week for 4 weeks, then once per week for 4 weeks for a given 8‐week regimen.” (p. 1667)
Duration of treatment: 8 weeks
Concomitant psychotherapy: no ‐ excluded if receiving any current relevant treatment
Concomitant pharmacotherapy: no ‐ excluded if receiving any current relevant treatment Wait‐list Comparison name: wait‐list. Description of intervention: “Following the initial 8 weeks in 1 of these 3 intervention groups, all patients received SPEC intervention for the next 8 weeks.” (p. 1668) Exposure/intensity to treatment : no treatment during the 8 weeks Duration treatment: 8 weeks Concomitant psychotherapy: no ‐ excluded if receiving any current relevant treatment Concomitant pharmacotherapy : no ‐ excluded if receiving any current relevant treatment |
|
| Outcomes |
Beneficial outcomes for effect
Relevant outcomes for adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “Patients were randomly assigned to 1 of 3 intervention groups following a stratified randomization schedule based on sex and severity of depression (…)” (p. 1668). “No significant differences emerged between intervention groups with respect to age, male/female ratio, ethnicity, age at onset, number of previous episodes, or symptom severity as assessed by the HAM‐D or the BDI.” (p. 1668) Quote: “In 2006, the randomization was stratified by severity, with two master records created in advance.” ( Allen 2006 (pers comm) ) |
| Allocation concealment | Yes | Quote: ”Randomization schedules were devised by the first author at study onset, with each client’s assignment becoming known to the assessing acupuncturist and the study coordinator only after the completion of the intake assessment” (p. 1668) Quote: "also concealed until each new subject needed to be randomized.” ( Allen 2006 (pers comm) ) |
| Blinding of outcome assessors | Yes | Quote: ”The primary outcome measure was the HAM‐D, administered at intake and at 4‐week intervals thereafter by trained raters blind to intervention condition.” (p. 1668) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Quote: “Change in depression severity over time was examined using random regression analyses using a mixed effects linear regression model with MIXREG software (…) The random regression approach utilizes all available data, estimating rate of change for each subject based on extant observations.” (p. 1668) Attrition >15% (Specific: 20.8%, Non‐specific: 8.2%, Wait‐list: 15.4%. Excluded 6 after randomisation due to that the did not fulfil the inclusion criteria. Uses intent‐to‐treat on the remaining. |
| Selective outcome reported | Yes |
NCT00010517 Protocol found but most information was not provided. Authors provided a descriptive protocol through author correspondence ( Allen 2006 (pers comm) ) |
| Other sources of bias | Yes | No other found |
Alvarez 1997.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 3 weeks Duration of participation (trial + follow‐up): 3 weeks (endpoint data only) Setting: drug‐free, residential therapeutic community located in a large northeastern metropolitan area (outpatient) Purpose of trial: ”This study tested the following hypotheses: (1) Anger management treatment reduces both experienced and expressed anger in a sample of drug addicts in a therapeutic community; (2) the acquisition of effective anger manager skills by these subjects increases self‐esteem; (3) the acquisition of positive anger management skills by these subjects decreases depression; (4) the acquisition of positive anger management skills by these subjects decreases addiction severity.” (p. 6) Open/closed placebo: closed |
|
| Data |
Number of participants screened: “350 to 400 clients.” (p. 49) Number of participants included: 119 Number of participants followed‐up at post treatment: 76 Number of participants randomly assigned to:
Completer data at post‐treatment
Number of withdrawals: n =
Diagnosis: Substance‐use disorder Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: not stated Comorbidity: substance‐use of different drugs (51.6% cocaine, 26.4% polydrug abusers, 13.2% heroin). Age : 33.91 mean years, (range 19 to 54) IQ: moderate to severe intellectual disability excluded from trial Sex : 47.1% female Ethnicity: 90% African‐Americans and Latinos and 10% Asian, European, or "other." Country : USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name : attention placebo Description of intervention: ”Subjects in the attention placebo group were presented an educational lecture from another standardized protocol, a copy of which appears in Appendix H. It consisted of a presentation of the clinical pharmacology of addictive drugs based on a training course for psychologists.” (p.60) Individual or group treatment: group Exposure/intensity to treatment: 6 hours Duration of treatment : 3 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: ”Prescription medications for medical conditions are allowed if they do not interfere with the individual's ability to fully participate in the TC program. Medications such as fluoxetine for depression are frowned upon, though sometimes tolerated.” (p.65) No‐treatment Comparison name: no‐treatment Description of intervention: "The control group (N = 40) received no treatment from the facilitators, except for the pre‐ and posttest batteries." (p.58) Exposure/intensity to treatment: none Duration treatment : 3 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: ”Prescription medications for medical conditions are allowed if they do not interfere with the individual's ability to fully participate in the TC program. Medications such as fluoxetine for depression are frowned upon, though sometimes tolerated.” (p.65) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found. Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”The list generated by the registration procedure was then randomly divided into three approximately even groups and each randomly assigned to an experimental condition.” (p.50) “Careful examination of procedures revealed no systematic bias that would interfere with the randomness of the groups.” (p. 58) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Quote:“The staff involved in the study were not blind to the paradigm” (p. 28) Patient‐reported outcomes only |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Attrition >15% (36.2%). No ITT. Only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Ascher 1979.
| Study characteristics | ||
| Methods |
Parallell randomised trial with 3 interventions
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 4 weeks Duration of participation (trial + follow‐up): “10 days pre trial + 4 weeks of experimental phase Setting: outpatient Purpose of trial: “The present study focused on a comparison of paradoxical intention with appropriate control procedures in reducing sleep onset insomnia.” (p. 408) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened : not stated Number of participants included: 25 Number of participants followed‐up at post treatment: not stated Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: sleep‐wake disorder (insomnia) Diagnostic manual: not stated Means of assessment: interview, not otherwise specified. “Following their initial phone call, clients who described themselves as experiencing a clinically significant level of sleep disturbance were invited for a pre‐treatment interview.” (p. 409) Comorbidity: not stated Age: 39 mean years, (range 24 to 67) IQ: not stated Sex : 60% female Ethnicity : not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo: Treatment name: placebo Description of intervention: “ Steinmark 1974 employed a placebo condition which served as a model for the present study. During the first session each subject was required to compose a hierarchy consisting of eighteen chronologically ordered bedtime activities. Then six neutral scenes were constructed. Finally, the client imagined each of the bedtime activities in the appropriate order, each being paired with one of the six neutral scenes. Homework involved practicing the procedure twice a day, but not within two hours of bedtime.” (p. 409) Individual or group treatment: individual Exposure/intensity to treatment: four weekly sessions of 30‐45 minutes Duration of treatment : 4 weeks Concomitant psychotherapy not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name : no treatment (in reality wait‐list) Description of intervention: “Clients in this condition were provided with no treatment throughout the four weeks of the experimental phase. Contact was maintained through brief telephone conversations once every 1 ½ weeks.” (p. 409) “Subjects in either of the control conditions who elected to continue with treatment were provided with a combination of behavioral techniques.” (p. 409.) Exposure/intensity to treatment: no treatment during the experimental phase Duration treatment: 4 weeks. Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”The experiment consisted of three groups of randomly assigned subjects. The paradoxical intention treatment was contrasted with no‐treatment and placebo treatment control conditions.” (p. 408) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Unclear | No information Attrition unclear |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Ayen 2004.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 3 months Duration of participation (trial + follow‐up): 3 months + 12 months Setting: outpatient Purpose of trial: assess short‐ and long‐term efficacy of CBT compared to supportive group program (UGT) and waiting list control for depressive problems among menopausal women. Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened : 85 Number of participants included : 51 Number of participants followed‐up at post treatment : 50 Number of participants randomly assigned to:
Number of withdrawals : n = 1
Diagnosis:depression Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV). 59 % fulfilled DSM diagnosis, 41 % had an unspecified depression Means of assessment: The Structured Clinical Interview for DSM‐IV Disorders (SCID) (German version) Comorbidity: 6 participants (12 %) had substance dependence. 27 (53%) had anxiety disorder Age: 51.3 means years (range = 46 – 56) IQ: not stated Sex: 100% females Ethnicity: not stated Country: Germany Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name : Unterstützende Gruppe (UGT) Description of intervention: The control group incorporated none of the specific techniques from the active intervention. Individual or group treatment: group exposure/intensity to treatment: 2 hours each session. Duration of treatment: 3 months Concomitant psychotherapy: not stated ‐ but many patients had already been in psychic or psycho‐therapeutic treatment Concomitant pharmacotherapy: 29 did not take drugs, 6 took antidepressants and 1 took anxiety medication. Wait‐list Comparison name: waiting list B Description of intervention: unclear Exposure/intensity to treatment: not stated Duration treatment: 3 months Concomitant psychotherapy: not stated ‐ but many patients had already been in psychic or psycho‐therapeutic treatment Concomitant pharmacotherapy: 29 did not take drugs, 6 took antidepressants and 1 took anxiety medication |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found. Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | quote: “However, initially (21 participants inside) between the KVT (N = 11) and the KG (N ‐ 10) was randomized, later (30 participants) then between the UGT (N = 20) and the KG (N 10)” (translated from, p. 293) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | Yes | Attrition <15% (1.96%) |
| Selective outcome reported | Unclear | No information |
| Other sources of bias | Yes | None other sources of bias found |
Berg 1983.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised : no Duration of trial (baseline to post): 3 months Duration of participation (trial + follow‐up): 3 months + 18 months Setting: outpatient Purpose of trial : “The study described in this paper was designed to see whether behaviour therapy would suffice on its own in the treatment of severe and persistent faecal soiling or would be improved by employing a laxative as well.” (p.544) Open/closed placebo: closed |
|
| Data |
Number of participants screened: not stated Number of participants included: 44 Number of participants followed‐up at post treatment 40 Number of participants randomly assigned to:
Number of withdrawals: 4 (only completed one or two visits) Diagnosis: faecal soiling (encopresis) Diagnostic manual: not stated Means of assessment: clinical interview (not otherwise specified) Comorbidity: not stated Age : 7.9 mean years (SD = 2.3) IQ: not stated Sex : not stated Ethnicity: not stated Country: UK Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name (type): Group B
Description of intervention: “The basic method of treatment given to all was behavioural, focusing on use of the toilet and freedom from soiling.” (p. 544)
“Group B was given placebo tablets in similar dosage…” (as group A, not specified). The children started at one tablet at night. On the next visit to the clinic, if there was no improvement in ‘use of the toilet’ and ‘being clean’ on the charts the dosage was increased to two tablets. The number of tablets was increased to three on the following visit if improvement had still not occurred by that time. When the soiling was getting better and the child was using the toilet the dosage was kept the same. Once the child was going regularly to the toilet and not soiling the tablets were stopped altogether.” (p. 544)
Individual or group treatment: individual.
Exposure/intensity to treatment: 1‐3 pills at night. ”Mother and child came to see the psychologist dealing with them every fortnight for three months,” (p. 544)
Duration of treatment: 3 months
Concomitant psychotherapy: not stated
Concomitant pharmacotherapy: not stated Wait‐list Comparison name: Group C (wait‐list – any child still soiling after the 3 months assessment was offered a further 3 months treatment by the same psychologist using a behavioural approach as before and explicitly labelled Senokot tablets, 1‐3 at night, in addition, (p. 546). Description of intervention: “The basic method of treatment given to all was behavioural, focusing on use of the toilet and freedom from soiling.” (p. 544) Exposure/intensity to treatment : No medical treatment. ”Mother and child came to see the psychologist dealing with them every fortnight for three months,” (p. 544) Duration treatment: 3 months Concomitant psychotherapy : not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found J judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”Cases were then randomly allocated to one of three treatment groups” (p. 544) “The process of random allocation was successful since the treatment groups did not differ on a whole variety of features” (p. 547) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Psychiatrist were outcome assessors Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Quote: ”Forty‐four children were included in the investigation but 4 dropped out after only one or two visits.” (p. 544)” Attrition > 15%. (9.1% dropped out after one or two visits) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Borden 1986.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised : no Duration of trial (baseline to post): 3 months Duration of participation (trial + follow‐up): “Treatment occurred over a three‐month period, with children beginning the program at various times throughout a one‐year period. All testing was completed during the three weeks preceding treatment, and then during the three weeks following treatment.” (p. 42) Setting: outpatient Purpose of trial: “The present study examined the attributional effects of combining medication with cognitive behavior therapy in the treatment of children diagnosed as having Attention Deficit Disorder (ADD).” (p. iii) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included : 37 (only 30 is stated in the text, but in notes at page 72, it is stated that seven additional children began the study). Number of participants followed‐up at post treatment: 30 Number of participants randomly assigned to: not stated Number of randomised completers
Number of withdrawals: "7 additional children began the study" (p. 72)
Diagnosis: Attention Deficit Hyperactivity Disorder (ADHD) (n = 25) or Attention Deficit Disorder without Hyperactivity (ADDNH) (n = 5). Diagnostic manual: not stated Means of assessment: clinical interview, laboratory tests, parent, teacher, and child questionnaires, achievement tests, intelligence tests and paediatric examination. Comorbidity: not stated Age: 107.1 months (SD = 20.6). (Range = 68 ‐ 143 months) IQ : Above 80 Sex: 16.7% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name : Placebo (pharmacological placebo) + cognitive behavior modification
Description of intervention: “The placebos were administered to placebo children in the same way and at the same time as was the active medication. Methylphenidate and placebo doses were both packaged by a University of Illinois Hospital pharmacist in identical opaque capsules to conceal their contents.” (p. 43) “Each child was randomly assigned to a therapist who was responsible for cognitive training.” (p. 43‐45)
Individual or group treatment: individual.
Exposure/intensity to treatment: two doses were administered per day: one at breakfast and one at lunch” (p. 43). Cognitive training sessions two times per week.
Duration of treatment: 3 months
Concomitant psychotherapy: cognitive behavior modification
Concomitant pharmacotherapy: not stated No‐treatment Comparison name : No pill (no treatment) + cognitive behaviour modification Description of intervention : No pharmacological treatment. “Cognitive training occurred over three months, with sessions held two times per week… Each child was randomly assigned to a therapist who was responsible for cognitive training.” (p. 43‐45) Exposure/intensity to treatment: no medication. Cognitive training sessions two times per week Duration treatment : 3 months Concomitant psychotherapy: cognitive behavior modification Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effects
Adverse events
|
|
| Notes |
Key conclusions from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: “Assignment to groups was random with the exception of stratification based upon sex and age.” (p. 33) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:”Examiners were blind to the treatment conditions of the children they tested.” (p. 42) Tests were administered by trained masters and doctoral level students who were blind to the subjects' medication conditions.” (p. 35) “Disagreements between raters were settled by a third graduate student in psychology who was blind to the responses of the first two.” (p. 39) |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | Unclear | Attrition > 15% (19%). No mention of ITT. Only completers included |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Borkovec 1975.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation : not stated Cluster randomised: no Duration of trial (baseline to post): 4 weeks Duration of participation (trial + follow‐up): 4 weeks of treatment + 5 months follow‐up Setting: outpatient Purpose of trial : “At the end of a counter demand period, progressive relaxation was compared to the same quasi‐desensitization placebo and no treatment conditions employed in the earlier investigation. As suggested elsewhere (Borkovec,1973), a treatment procedure demonstrated to reliably produce improvement greater than placebo under such (neutral) expectancy conditions is indeed a powerful modification technique and includes active ingredients independent of demand effects.” (p. 302) Open/closed placebo : closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included : 56 Number of participants followed‐up at post treatment: 41 N umber of participants randomly assigned to: 56 were randomised, but reports only data on completers
Number of withdrawals: n = 15
Diagnosis: sleep‐wake disorder (Insomnia) Diagnostic manual: not stated Means of assessment : clinical interview, not otherwise specified. “(…) were interviewed and screened, following the criteria of Steinmark 1974 .” (p. 302) Comorbidity : not stated Age: not stated IQ : not stated, but all were college students. Sex : not stated Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name : quasi‐desensitisation placebo Description of intervention: “During session 1, subjects in the P condition constructed an 18‐item hierarchy of chronological bedtime activities and chose six neutral images to be paired with the hierarchy items as a substitute for the relaxation ordinarily employed in conventional desensitization. Each hierarchy item was presented three times with intervening presentations of neutral images during session 2, 3 and 4. The P subjects were instructed to practice hierarchy and neutral image visualizations twice a day.” (p. 303) Individual or group treatment : group sessions + individual practice Exposure/intensity to treatment : 4 sessions of group treatment + individual practice twice a day Duration of treatment: 4 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy:: not stated Wait‐list Comparison name: waiting list no treatment (in reality wait‐list) Description of intervention: “Subjects in the NT condition were told by phone that current treatment groups were filled, new groups would begin in 4 wk, and they would receive priority if they filled out the daily questionnaires during the next 4 wk.” (p. 303) Exposure/intensity to treatment: no treatment during the 4 weeks experimental period Duration treatment: 4 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effects
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes
Conflicts of interest: none found Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”The subjects were ranked on latency to sleep onset obtained in the pretherapy interview and randomly assigned within levels of severity to one of the four conditions: (a) progressive relaxation (PR), (b) relaxation without tension‐release (NTR), (c) quasi‐desensitization placebo (P), and (d) waiting list no treatment (NT).” (p. 303) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | No | Attrition >15% (27%). No ITT. Only reports data on completers |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Borkovec 1976.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised no Duration of trial (baseline to post): 4 weeks (endpoint data) Duration of participation (trial + follow‐up): pre‐therapy week + 4 weeks of therapy + 1 year (follow‐up) Setting: outpatient Purpose of trial : “The purpose of the present study was to replicate the basic aspects of the earlier investigations using objective sleep measures.” (p. 174) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 36 Number of participants followed‐up at post treatment: 33 Number of participants randomly assigned to:
Number of withdrawals: n = 3
Diagnosis: sleep‐wake disorder (sleep disturbance) Diagnostic manual: not stated Means of assessment: Brief questionnaire ‐ ”A brief questionnaire on sleep behavior was given (…)” (p. 174) + pretherapy interview Comorbidity: not stated Age: not stated IQ : not stated, but psychology students at University of Iowa Sex : not stated Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo: Treatment name: placebo Description of intervention: “The placebo condition involved a quasi‐desensitization procedure. During Session 1, each subject constructed an 18‐item hierarchy of chronological bedtime activities and chose six neutral images to be paired with the hierarchy items and to be used as substitutes for relaxation” (p. 175) Individual or group treatment: group treatment + individual practice Exposure/intensity to treatment: 4 weekly group sessions + individual practice twice a day Duration of treatment: 4 weeks Concomitant psychotherapy: no – excluded if current contact with other professional services during the interview Concomitant pharmacotherapy: not stated Wait‐list Comparison name: no‐treatment (in reality a wait‐list) Description of intervention:“No‐treatment subjects were told that current treatment groups were filled but that new groups would be formed in 6 weeks and that they would receive priority if they continued to fill out the daily sleep questionnaires and attended the sleep evaluation nights.” (p. 174) Exposure/intensity to treatment: no treatment Duration treatment: 4 weeks Concomitant psychotherapy: no – excluded if current contact with other professional services during the interview Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”The subjects were ranked on latency to sleep onset obtained in the pretherapy interview and randomly assigned within levels of severity to one of the four conditions: (a) progressive relaxation (PR), (b) relaxation without tension‐release (NTR), (c) quasi‐desensitization placebo (P), and (d) waiting list no treatment (NT).” (p. 303) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:“Three research assistants, independent of each other and “blind” to the experimental condition and evaluation night of the subjects, scores the EEG records of the three evaluation nights (pretherapy, counterdemand, and positive demand). (p. 175‐6) |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | Yes | Attrition <15% (8.5%). Equal amount from each group. No ITT |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Bornovalova 2008.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 3 weeks Duration of participation (trial + follow‐up): 3 weeks Setting: inpatient Purpose of trial : “To develop a treatment for prevention of treatment drop‐out in a residential treatment setting.” (p. 1) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 68 Number of participants include : 66 Number of participants followed‐up at post treatment: 65 Number of participants randomly assigned to:
Number of withdrawals: n = 1
Diagnosis: substance use disorder Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: The Structured Clinical Interview for DSM‐IV Disorders (SCID) Comorbidity:
Age: 43.5 mean years (SD = 9.8) IQ : not stated ‐ but approximately 35.8% of the participants had an education level of “less then high school”, 34.3 % had a “high school or equivalent” level, and 30% had “some college and above” level Sex: 20.6% female Ethnicity: 90.1 % African American Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Supportive counselling Description of intervention: “To control for the non‐specific elements of therapist contact, approximately one‐third of the patients received SC, which also consisted of six individual sessions over 3 weeks. This treatment did not follow a clearly defined theoretical model, and was best described as unconditional support, combined with information and advice on managing current problems that a given patient may be experiencing. Although the format was rather open, therapists were provided with a manual providing a script for the initial session as well as potential topics for discussion and corresponding prompts. These included (but were not limited to) day to day annoyances/issues in the treatment center likes and dislikes about the centre, discussions of drug court status and concerns related to this, discussions of families and relationships, concerns about leaving the center, spirituality, relaxation and leisure time, and employment and finances. SC specifically avoided acceptance or mood induction techniques.” (p. 28) Individual or group treatment: individual Exposure/intensity to treatment: 6 sessions over 3 weeks Duration of treatment: 3 weeks Concomitant psychotherapy: “TAU is basically no treatment (from us). They are still receiving addiction TX from the Residential Facility (as are all patients regardless of condition); But they don’t get additional treatment from us. We just give them a pre and post” ( Bornovalova 2008 (pers comm) ) Concomitant pharmacotherapy: receiving Psychotropic Medication: 13.2%. Alcohol 32.4%, Cannabis 10.4%, Heroin 29.9%, Cocaine 58.2%, Phencyclidin (PCP) 7.5% Comparator intervention Comparison name: no‐treatment Description of intervention: “Of note, procedure for NTC was similar for baseline and post‐test. However, no therapy was given.” (p. 17). Sometimes labelled TAU. Exposure/intensity to treatment: Duration treatment: 3 weeks Concomitant psychotherapy: TAU is basically no treatment (from us). They are still receiving addiction TX from the Residential Facility (as are all patients regardless of condition); But they do not get additional treatment from us. We just give them a pre and post” (author correspondence) Concomitant pharmacotherapy: receiving Psychotropic Medication 13.2%. Alcohol 32.4%, Cannabis 10.4%, Heroin 29.9%, Cocaine 58.2%, Phencyclidin (PCP) 7.5% |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: potential n on‐financial conflict of interest. The principal investigator (MAB) conducted a large majority of the SIDI group . Judgment: no |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “I used one of the randomisation websites to pre‐create a randomisation list of subject numbers and conditions to which said numbers were assigned. ( Bornovalova 2008 (pers comm) ) |
| Allocation concealment | Yes | Concealed ( Bornovalova 2008 (pers comm) ) |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | No | Quote: “there were no dropouts in the SIDI or SC conditions (0%), and 4 individuals (16%) dropped out of the TAU group, resulting in an overall sample dropout rate of 6.1%. In contrast, when dropout at any point in treatment (thus including any time in their contract after SIDI was completed) was used as a dependent variable, the rates were somewhat different." (p. 74) ”Thus, the positive findings were likely inflated, as treatment completer rather then intent‐to‐ treat analyses were utilized in this study.” (p. 74) Attrition rate >15% (16% in no‐treatment group, 0% in other groups). No ITT. Reports data on completers only |
| Selective outcome reported | No |
NCT01741415. No congruent between trial registry outcomes and full report. Some outcomes added – some deleted. Control group added |
| Other sources of bias | Yes | No other sources found |
Bramston 1985.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post : 4 weeks Duration of participation (trial + follow‐up): 4 weeks + 3‐month follow‐up Setting: inpatient Purpose of trial: "This study was therefore designed to investigate the effectiveness of a traditional SST approach as compared to a cognitively based social‐problem‐solving (SPS) programme in enhancing the social competence of intellectually‐handicapped adults." (p. 240) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 48 Number of participants followed‐up at post treatment: 48 Number of participants randomised :
Number of withdrawals : n = 0 Diagnosis: intellectual disability Diagnostic manual: not stated Means of assessment: The Wechsler Adult Intelligence Scale (WAIS) Comorbidity: not stated Age: 28.06 mean years (SD = 7.08), (range = 18.03 to 46.5) IQ: a mean IQ of 40.06 and 55.20. Sex: not stated Ethnicity: not stated Country: Australia Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Attention‐Placebo Control Group Description of intervention: “In order to control for non‐specific treatment effects the 12 Ss in this condition received an equivalent degree of therapist input and small group attendance as for the BSST and SPS training groups. ” (p.242) Individual or group treatment: group Exposure/intensity to treatment: sessions occurred four times a week over 4 weeks, each lasting for 30 minutes Duration of treatment: 4 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy:: not stated Wait‐list Comparison name:: no‐treatment control group (in reality a wait‐list) Description of intervention: “Twelve Ss received no direct intervention other than that ordinarily offered by the centre during the daily routine. Assessments were made before and after the training phase on all measures applied to the experimental groups. Training was then made available following the post‐training assessment.” (p. 242) Exposure/intensity to treatment: no treatment provided Duration treatment: not stated Concomitant psychotherapy: ordinary treatment Concomitant pharmacotherapy:: ordinary treatment |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: "Subjects were randomly allocated to one of four groups: a behavioural social‐skills (BSST) programme, a social‐problem‐solving (SPS) programme, an attention‐placebo control (APC) group and a no‐treatment control (NTC) group." (p. 240) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:”In the present study one rater completed the scale for all Ss at each assessment session. Thisrater was highly experienced in use of the scale but was blind as to the treatment condition of Ss.” (p. 241) |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | Yes | Quote:”There was no other attrition throughout the study.” (p.240) Attrition <15% (0%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Brill 1964a.
| Study characteristics | ||
| Methods |
Parallel randomised trial with six arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): average 5 months (up to 12 months.) Duration of participation (trial + follow‐up): 5 + 10 ‐ 18 months follow‐up Setting: outpatient Purpose of trial: a controlled double‐blind study of 299 non‐psychotic female psychiatric clinic patients divided into six groups, with members of each group dealt with in a different manner from those in other groups. Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 299 Number of participants followed‐up at post treatment: 169 Number of participants randomly assigned to:
Number of withdrawals: n = 130
Diagnosis: psychiatric patients. (The sample included patients with personality disorders, psychoneuroses, psychosomatic disturbances, and borderline schizophrenic state) Diagnostic manual: not stated Means of assessment:: not stated Comorbidity: different psychiatric diagnoses Age: range = 20‐40 years IQ: average intelligence or better Sex: 100% female Ethnicit::100% Caucasian Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacologoical placebo Treatment name: placebo pill Description of intervention: “All capsules were identical in color and size.” (p. 583) Individual or group treatment: individual Exposure/intensity to treatment: not stated Duration of treatment: average 5 months Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: no‐treatment (in reality wait‐list) Description of intervention: “The no‐treatment group was assigned at random from the pool of patients who had been examined. These patients were told that they could not be accepted for treatment immediately but that treatment might be available after about four months to a year. They were told that they would be contacted in approximately that time. Four months after the date of initial evaluation, the same explanation was repeated. Some patients were re‐evaluated at that time ; others were again placed on a waiting list and were recalled during the year for re‐evaluation.” (p. 584) Exposure/intensity to treatment: no treatment during waiting Duration treatment: average 5 months Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | No information |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | Quote: ”the therapists had no knowledge of the names, types, or number of drugs involved, nor even if there was a placebo being used.” (p. 585). "it was evaluated blindly and because of the prejudice in favor of psychotherapy among patients and therapists." (p. 590) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | No | Attrition > 15% (43.5 %) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Brill 1964b.
| Study characteristics | ||
| Methods | See Brill 1964a | |
| Data | See Brill 1964a | |
| Comparisons |
Usual care Treatment name: psychotherapy Description of intervention: “The patients treated with psychotherapy were assigned to psychiatric residents who were closely supervised by the clinical staff of The Neuropsychiatrie Institute of the UCLA Center for Health Sciences. Each resident treated one or two psychotherapy patients. These were seen for 50‐minute sessions at least once a week and could be seen more often at the discretion of the resident and his supervisor. Psychotherapy, while psychoanalytically oriented and generally nondirective in keeping with the attitude of the supervisory staff, was in varying degrees supportive. The average length of treatment was five months.” (p. 583) Individual or group treatment: individual Exposure/intensity to treatment: 50 minutes at least once a week Duration of treatment: average 5 months Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: no‐treatment (in reality wait‐list) Description of intervention: “The no‐treatment group was assigned at random from the pool of patients who had been examined. These patients were told that they could not be accepted for treatment immediately but that treatment might be available after about four months to a year. They were told that they would be contacted in approximately that time. Four months after the date of initial evaluation, the same explanation was repeated. Some patients were re‐evaluated at that time ; others were again placed on a waiting list and were recalled during the year for re‐evaluation.” (p. 584) Exposure/intensity to treatment: no treatment during waiting Duration treatment: average 5 months Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes | See Brill 1964a | |
| Notes | See Brill 1964a | |
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | No information |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | Quote: ”the therapists had no knowledge of the names, types, or number of drugs involved, nor even if there was a placebo being used.” (p. 585). "it was evaluated blindly and because of the prejudice in favor of psychotherapy among patients and therapists." (p. 590) |
| Blinding of participants and personnel | No | Not possible to blind usual care and wait‐list |
| Incomplete outcome data | No | Attrition > 15% (43.5 %) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Carlson 1993.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 6 weeks. Duration of participation (trial + follow‐up): 6 weeks + 2 weeks Setting: outpatient (an 8‐week day‐treatment program) Purpose of trial: investigating MPH’s effect on the performance and perceptions of Attention Deficit Hyperactivity Disorder (ADHD) boys following solvable and insolvable puzzles. Closed/open placebo: closed placebo |
|
| Data |
Number of participants screened: not stated
Number of participants included: 28 Number of participants followed‐up at post treatment: 28 Number of participants randomly assigned to :
Number of withdrawals: n = 2
Diagnosis: Attention Deficit Hyperactivity Disorder (ADHD) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition, revised (DSM‐III‐R) Means of assessment: clinical interviews and standardised rating scales Comorbidity: ”Based on the interview, 17 of the boys also obtained a DSM‐III‐R diagnosis of oppositional/defiant disorder, and 8 boys met criteria for a DSM‐III‐R diagnosis of conduct disorder.” (p. 273) Ag:: 9.35 mean years (SD = 1.33) IQ: “On the Wechsler Intelligence Scale for Children‐Revised, subjects had a mean score of 108 (SD=10), and on the Woodcock‐Johnson Test of Achievement Revised, they obtained a mean reading standard score of 97 (SD =12). Sex: 100% male Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: placebo
Description of intervention: received placebo pill each day. “The MPH and placebo were packed in identical opaque gelatin capsules.” “Medication was given in the morning and at midday.” (p. 274)
Individual or group treatment: individual
Exposure/intensity to treatment: two times a day
Duration of treatment: 6 weeks.
Concomitant psychotherapy: not stated ‐ but was a part of a Summer Treatment Program
Concomitant pharmacotherapy: not stated No‐treatment Comparison name: no pill (no treatment) Description of intervention: no treatment Exposure/intensity to treatment: no treatment Duration treatment: 6 weeks Concomitant psychotherapy: not stated ‐ but was a part of a Summer Treatment Program Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effects
Adverse events
|
|
| Notes |
Key conclusions from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: “Order of the conditions was randomized, with the constraint that for all boys the first two sessions were either both solvable or both insolvable conditions.” (Milich 1991, p. 524) No information in Carlson 1993 if the study was randomised, but replicates ( Milich 1991 ), which was randomised. |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | No information but outcomes was objective measures (cognitive test) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Quote: “Two were dropped from analyses because on at least one solvable day they failed to find any solvable puzzles” (p. 272) Attrition <15% (7,1%). However, only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Carter 2003.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 8 weeks Duration of participation (trial + follow‐up): 8 weeks (endpoint data) Setting: inpatient Purpose of trial: “The aim of the present study was to evaluate the effectiveness of a cognitive behavior self‐help manual for patients with bulimia nervosa who were on a waiting list for treatment at a hospital‐based specialist clinic.” (p. 973) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 245 Number of participants included: 85 Number of participants followed‐up at post treatment: 65 Number of participants randomly assigned to:
Number of withdrawals: n = 20
Diagnosis: Bulimia nervosa Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: Eating Disorder Examination (EDE) Comorbidity: not stated Age: 27 mean years (SD = 8), (range=17 to 53) IQ: not stated Sex: 100% female Ethnicity: 83% Caucasian, 2% African Caribbean, 7% Asian, and 8% other. Country: Canada Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name : nonspecific self‐help Description of intervention: “The second self‐help condition (nonspecific self‐help) was designed to control for nonspecific factors, such as receiving a self‐help book, hearing a plausible rationale, and expecting to improve. It involved following the self‐help manual Self‐Assertion for Women. This self‐help manual focuses on developing assertiveness skills and does not in any way address the specific symptoms of bulimia nervosa. This control intervention was selected because it might be regarded by patients as a credible alternative treatment, since many women with eating disorders report experiencing significant interpersonal difficulties, including inhibited self‐assertion. Like the cognitive behavior self‐help manual, the nonspecific self‐help book contains both psychoeducational information and practical advice designed to foster behavioral change. Both books were similar in length and level of difficulty.” (p. 974) Individual or group treatment: individual (self‐help) Exposure/intensity to treatment: not stated Duration of treatment: 8 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “Patients who were taking an established dose of antidepressant medication were eligible to take part” (p. 974) No‐treatment Comparison name: waiting list control group (in reality a no treatment) Description of intervention: “Individuals assigned to the waiting list condition received no intervention.” (p. 974) Exposure/intensity to treatment: no exposure Duration treatment: 8 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: patients who were taking an established dose of antidepressant medication were eligible to take part” (p. 974) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote ”A restricted randomization procedure employing random permuted blocks of three people was used to ensure approximately equal numbers of participants in the three conditions.” (p. 974) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Quote: ”An assessor who was blind to the patients’ treatment assignment performed the posttreatment assessments 8 weeks later.” (p. 974) Patient‐reported outcomes used |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | Unclear | Quote: "Twenty participants (23.5%) dropped out of the study and did not attend the posttreatment assessment: five (17.9%) of these were from the cognitive behavior self help group (N=28), seven (25.0%) were from the nonspecific self‐help group (N=28), and eight (27.6%) were from the waiting list control group (N=29). There was no statistically significant difference between the three conditions in terms of the rate of attrition. This dropout rate is similar to those reported in previous treatment studies (...). There were no significant differences between the dropouts and completers in terms of baseline characteristics.” (p. 975) Attrition >15% (23.5%). ITT analyses made |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Crisp 1991.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): mean 20 weeks Duration of participation (trial + follow‐up): 20 weeks treatment, post treatment assessment at 1 year Setting: inpatient for usual care (outpatient for other group) Purpose of trial: “The present study involves an extension of this established treatment approach, allowing a controlled investigation of the effects of psychotherapy directed at the development and family psychopathology, together with dietary management, provided within three different treatment settings; and comparison with a no‐treatment group.” (p. 327) |
|
| Data |
Number of participants screened: not stated Number of participants included: 90 Number of participants followed‐up at post treatment: 73 Number of participants randomly assigned to:
Number of withdrawals: n = 17
Diagnosis: Anorexia nervosa Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition, revised (DSM‐III‐R) Means of assessment: diagnostic assessment interview Comorbidity: not stated Ag:: usual care 23.2 mean years (SD =4.9), no‐treatment; 21.9 mean years (SD = 4.5) IQ: not stated Sex: 100% female Ethnicity: not stated Country: UK Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Usual care Treatment name: inpatient treatment Description of intervention: “It was taken to be the established treatment (Crisp, 1980) and believed to be effective (...) In‐patient treatment was intensive and involved much greater patient contact than did the other two treatment options (…) Treatment involved weight restoration to the mean matched‐population weight (MMPW) at the age of onset of anorexia, supported by weekly individual therapy, family therapy, group therapy, dietary counselling and occupational therapy, including psychodrama and projective art techniques. Inpatient treatment was followed by 12 sessions of out‐patient psychotherapy involving the patient and family.” (p. 328) Individual or group treatment: both individual and group treatment Exposure/intensity to treatment : not stated Duration of treatment: mean stay was 20 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “Psychotropic drugs were not prescribed for or consumed by any of the patients in the three ongoing treatment groups during the period of study.” (p. 328) No‐treatment Comparison name: no further treatment Description of intervention: “Patients allocated to option 4, no further treatment (‘one‐off), were referred back to their family doctor or local consultant, who received a detailed report of the assessment together with advice on further management.” (p. 328) Exposure/intensity to treatment: no treatment Duration treatment: 12 months Concomitant psychotherapy: not stated, but probably, as most of the participants were treated elsewhere while enrolled in the study Concomitant pharmacotherapy: not stated, but probably, as most of the participants were treated elsewhere while enrolled in the study |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgment: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: “Following an assessment of the kind described above, 90 patients were randomly allocated to one or other of the four options (…)” (p. 327) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind usual care and no‐treatment |
| Incomplete outcome data | No | Attrition rate: 17/90 (18.9%). In‐patient: 12/30 (40%) Psychotherapy: 2/20 (10%). Group psychotherapy: 3/20 (15%). One‐off: 0. Quote:“Drop‐out occurred in all three treatment groups, especially the out‐patient group psychotherapy. One of the patients allocated to this treatment died as a consequence of her anorexia nervosa before the treatment could begin. This was the only death” (p. 331) “We were distressed to find patients refusing treatment or dropping out because of forced allocation when they would have preferred ‐ or we would have preferred to have offered them another therapy” (p. 331) Quote:“the sample will not include patients who refused in‐patient admission in the first instance or who dropped out of in‐patient treatment, refusing further intervention.” (p. 326) Attrition > 15% (18.9%). Drop‐out on usual care group was 40%. No ITT. Only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Crouch 1988.
| Study characteristics | ||
| Methods |
Parallel arm with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 10 weeks Duration of participation (trial + follow‐up): 10 weeks + 3 month follow‐up Setting: outpatient (withdrawal clinic) Purpose of trial: “We therefore present some characteristics of patients referred for treatment of tranquillizer dependence to a clinic specializing in its treatment” (p. 503‐4) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 91 Number of participants included: 44 Number of participants followed‐up at post treatment: 23 Number of participants randomly assigned to :
Number of withdrawals : n = 21
Diagnosis: substance dependence (benzodiazepine) Diagnostic manual: not stated Means of assessment: assessment interview. All patients assessed with The State‐Trait Anxiety Inventory (STAI). Otherwise, referred to by local general practitioners Comorbidity: Generalized anxiety disorder (GAD), n = 13; Panic disorder, n = 5; Agoraphobia; n = 1; somatic symptoms, n = 8; Insomnia, n =4; Drug abuse, n = 2; Alcohol abuse, n =1; Depression, n = 2. Age: 41.4 mean years (SD =10.8) IQ: not stated Sex: 14 male, 30 women Ethnicity: not stated Countr:: UK Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: placebo pill
Description of intervention: no information about placebo pill. “Group support was structured with active interventions, anxiety management training with a cognitive‐based component” (p. 504).
Individual or group treatment: Individual + group
Exposure/intensity to treatment: 160 mg per day + “group meetings were held weekly for 5 weeks, then after 2 and 4 weeks respectively and lasted approximately one hour“ (p. 505)
Duration of treatment: 10 weeks
Concomitant psychotherapy: all patients received group support
Concomitant pharmacotherapy: “No other medication was taken, apart from steady reduction of diazepam/lorazepam over a four‐week period” (p. 504) No‐treatment Comparison name: no pill Description of intervention: no information about no pill group. “Group support was structured with active interventions, anxiety management training with a cognitive‐based component” (p. 504). Exposure/intensity to treatment: no pill + “group meetings were held weekly for 5 weeks, then after 2 and 4 weeks respectively and lasted approximately one hour“ (p. 505) Duration treatment: 10 weeks Concomitant psychotherapy:: all patients received group support Concomitant pharmacotherapy: “No other medication was taken, apart from steady reduction of diazepam/lorazepam over a four week period” (p. 504) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”Patients were allocated randomly to either highly structured cognitive based group therapy or offered group therapy of a more supportive non‐interventionist nature. The patients were also randomly allocated to one of 3 treatment groups. Either propranolol (”Inderal LA”) 16mg per day, matching placebo or no medication (“no pills”).” (Hallström 1988, p. 41) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Attrition >15% (52,3%). Only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Doty 1975.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five interventions
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 2 weeks Duration of participation (trial + follow‐up): 2 weeks treatment + 2 weeks follow‐up Setting: inpatient Purpose of trial: ”Do training in social skills or incentives, or a combination of both contribute to either the daily social interaction rates or the social responsiveness of psychiatric patients?” (p. 677) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 96 Number of participants included : 56 Number of participants followed‐up at post treatment: 39 Number of participants randomly assigned to:
Number of withdrawals: n = 17
Diagnosis: open‐ward psychiatric patients
Diagnostic manual: not stated Means of assessment: not stated Comorbidity: not stated Age: 47.98 mean years IQ:: not stated Sex:: 100% male Ethnicity : not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: nonspecific control condition Description of intervention: “This treatment was modeled after the attention‐control group used by Wollersheim (1968) in that it was designed to control for the nonspecific therapy elements such as attention and positive regard from the therapist, knowledge of the target behaviors, and the expectancy that the four treatment sessions would lead to positive behavior change by the subjects. The vehicle for this control was lectures by the therapists following a transactional games analysis orientation and attempting to examine the supposed intrapsychic reasons why the subjects did not engage in more social interaction. Opportunities for the subjects to role play sample interactions or mention of concrete incentives for behavior change were specifically and intentionally avoided.” (p. 678) Individual or group treatment: group Exposure/intensity to treatment: 4 sessions Duration of treatment: 2 weeks Concomitant psychotherapy: “None of the subjects were participating in other forms of active psychological treatment except chemotherapy or assignment to recreational activities at the time of the study.” (p. 677) Concomitant pharmacotherapy: “52 of the 56 subjects were receiving psychotropic medication at the time of the study.” (p. 677) No‐treatment Comparison name: no‐treatment control Description of intervention: “The only contact that the subjects in this condition had with the therapists was in the pretreatment and posttreatment social responsiveness assessments. In fact, these subjects were never told that they would receive treatment and, therefore, received none of the nonspecific treatment elements such as knowledge of the target behaviors, encouragement to change, etc.” (p. 679) Exposure/intensity to treatment: not stated Duration treatment: 4 weeks Concomitant psychotherapy: “None of the subjects were participating in other forms of active psychological treatment except chemotherapy or assignment to recreational activities at the time of the study.” (p. 677) Concomitant pharmacotherapy “52 of the 56 subjects were receiving psychotropic medication at the time of the study.” (p. 677) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: ” Subjects were randomly assigned from stratified blocks formed on the basis of pretreatment levels of daily social interaction either to one of three active treatment conditions” (p. 677) “The success of the random stratified‐block subject‐assignment procedure in establishing the pre‐experimental group equation was checked using Treatment X Therapist analyses of variance for the group means and Bartlett's test of homogeneity of variance on the following variables both before treatment and rechecked after the data rejection mentioned above:” (p. 679) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: ”“The observers were blind to both the nature of the dependent variable to be extracted from their recordings and the group assignments of the subjects” (p. 678) ”Trained observers, unaware of the experimental group identity of the subjects,(...)” (p. 678) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Quote: ””In order to make the statistical analyses as meaningful as possible it was necessary in some cases to drop some subjects' data from consideration. For instance, eight subjects were dropped for failure to adequately expose themselves to the treatments (only subjects attending three or more sessions were included), and nine subjects were dropped from the analysis of the ward data because of incomplete data. Thus, criteria for data selection were as objective as possible, and attrition rates were approximately the same across the various subject groups” (p. 680) Attrition >15% (33.9%). No ITT. Excludes non‐completers |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Double 1993.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): phase one = 4 weeks (total 12 weeks) Duration of participation (trial + follow‐up) : not stated Setting: inpatient Purpose of trial:: “This study was rigorously designed to evaluate the efficacy of the long‐term use of anticholinergic agents in patients maintained on neuroleptics.” (p. 381) Open/closed placebo:: closed placebo |
|
| Data |
Number of participants screened: 96 Number of participants included: 27 Number of participants followed‐up at post treatment: 23 Number of participants randomly assigned to :
Number of withdrawals : n = 4
Diagnosis: psychiatric in‐patients Diagnostic manual: not stated Means of assessment: not stated Comorbidity: not stated Age: 54 mean years (range = 22‐76) IQ: not stated Sex: 29.6% female Ethnicity: not stated Country: UK Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo Treatment name: placebo Description of intervention: “Placebo and active medication for the trial were produced by the Pharmacy Manufacturing Department at the Royal Hallamshire Hospital, Sheffield, from raw materials obtained from manufacturers. The capsules looked identical and were tested for quality control. They were made in the strength that they are produced commercially, so that patients in the trial, instead of receiving tablets, were given capsules in the same number as usually prescribed.” (p. 382) Individual or group treatment: individual Exposure/intensity to treatment: up to 10 mg per day Duration of treatment: 4 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: on concomitant antiparkinsonian and neuroleptic medication for over one year No‐treatment Comparison name: no drug (no‐treatment) Description of intervention: not stated Exposure/intensity to treatment: not stated Duration treatment: 4 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: on concomitant antiparkinsonian and neuroleptic medication for over one year |
|
| Outcomes |
Beneficial effects
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | No information |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: ”Assessments were allocated equally between 3 assessors so that each patient was rated by a different assessor under each condition. There were, therefore, no carry‐over effects from one assessment to the next. Assessors remained blind to the previous ratings by other assessors.” (p. 382) Assessors remained blind to the previous ratings by other assessors. (p. 382) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Quote ”Complete assessments were unavailable for 4 patients (15%) because of drop‐out from the trial at different stages” (p. 382) Attrition = 15%. Only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Ehlers 2014.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 14 weeks Duration of participation (trial + follow‐up): 14 weeks + 27 weeks (follow‐up 1) and 40 weeks (follow‐up 2) Setting: outpatient Purpose of trial: “This clinical trial had two goals, (1) to investigate the acceptability and efficacy of a 7‐day intensive version of cognitive therapy for PTSD, and (2) to investigate whether cognitive therapy has specific treatment effects by comparing intensive and standard weekly cognitive therapy with an equally credible alternative treatment.” (p.1) Open/closed placebo : closed placebo |
|
| Data |
Number of participants screened: 253 Number of participants included: 125 (only reports data on 121 completers) Number of participants followed‐up at post treatment :112 Number of participants randomly assigned to:
Number of withdrawals: n =9
Diagnosis: Chronic Post‐traumatic Stress Disorder (PTSD) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DMSM‐IV) Means of assessment: the Structured Clinical Interview for DSM‐IV Disorders (SCID) Comorbidity: 63.6% had comorbid other Axis I disorders (mainly mood and anxiety disorders, substance abuse), and 19.8% had Axis II disorders (mainly obsessive‐compulsive, depressive, paranoid, avoidant). Age: placebo: 37.8 mean years (SD = 9.9), wait‐list; 36.8 mean years (SD =10.5) IQ: not stated Sex: 60% female Ethnicity: placebo 73.3% Caucasian, wait‐list 70 % Caucasion Country: UK Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pscyhological placebo Treatment name: Emotion‐focused Supportive Therapy Description of intervention: “This non‐directive treatment focused on patients’ emotional reactions rather than their cognitions. It was designed to provide a credible therapeutic alternative to control for nonspecific therapeutic factors so that observed effects of cognitive therapy could be attributed to its specific effects beyond the benefits of good therapy.” (p.4) Individual or group treatment: individual Exposure/intensity to treatment: “it comprised up to 12 weekly individual sessions (up to 20 hours in total) over three months and optional three monthly booster sessions.” (p.4) Duration of treatment: 12 weeks Concomitant psychotherapy: 3,3% started another psychological treatment during the study. Concomitant pharmacotherapy: “Patients taking psychotropic medication (29.8%) were required to be on a stable dose for two months before random allocation.” (p.3). No one started a new medication during the trial. Wait‐list Comparison name: wait‐list Description of intervention: patients allocated to wait‐list waited for 14 weeks before receiving treatment Exposure/intensity to treatment: no treatment during waiting Duration treatment: 14 weeks Concomitant psychotherapy: none reported Concomitant pharmacotherapy: “Patients taking psychotropic medication (29.8%) were required to be on a stable dose for two months before random allocation.” (p.3). No one started a new medication during the trial. |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: potential n on‐financial conflict of interest: ”The treatment follows Ehlers and Clark’s model of PTSD (...) ...” (p.3). First and last author developed the active treatment intervention Judgement: no |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: ”They were then randomly allocated (...) using the minimization procedure(...) to stratify for sex and severity of PTSD symptoms” (p.3) |
| Allocation concealment | Yes | Quote: ”They were then randomly allocated to one of the four trial conditions by an independent researcher who was not involved in assessing patients (...). Assessors determining the suitability of a patient for inclusion were not informed about the stratification variables and algorithm.” (p.3) |
| Blinding of outcome assessors | Yes | Quote: ”Assessments of treatment outcome were conducted by independent evaluators without knowledge of the patient’s treatment condition. Patients were asked not to reveal their group assignment to the evaluators. “(p. 3) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Quote: ”Dropouts were defined as attending fewer than 8 sessions (...), unless the earlier completion was agreed with the therapist. Dropout rates were low and did not differ between conditions (Table 2). Only one patient in the supportive therapy group reported symptom deterioration on the Posttraumatic Diagnostic Scale (Table 2). On the CAPS, fewer patients treated with intensive and cognitive therapy were rated as having deteriorated than those in the wait‐list condition. The supportive therapy group did not statistically differ from the other groups.” (p.7) Attrition <15% (4.1%). |
| Selective outcome reported | Yes | Trial registry: ISRCTN 48524925. No apparent differences in reporting between trial registry and full report. |
| Other sources of bias | Yes | No other sources found |
Espie 1989a.
| Study characteristics | ||
| Methods |
Parallel randomised trial with six arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 8 weeks Duration of participation (trial + follow‐up): 8 weeks treatment + follow‐up at 6 weeks, 3 months, 6 months and 17 months Setting: outpatient Purpose of trial: “Treatment process and outcome were investigated in terms of mean and standard deviation (night to night variability) measures of sleep pattern and sleep quality. (p. 80) Open/closed placebo: closed |
|
| Data |
Number of participants screened: 141 Number of participants included: 101 Number of participants followed‐up at post treatment: 84 (completers) Number of participants randomly assigned to :
Number of withdrawals:: n = 17
Diagnosis:: chronic insomniacs Diagnostic manual:: not stated Means of assessment: not stated Comorbidity: not stated Age: 45.5 mean years (SD = 15.9), (range 17 to 82) IQ: not stated Sex: 67.9% female Ethnicity: not stated Country: UK Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Imagery relief placebo Description of intervention: “Patients in this group were treated in accordance with the quasi—desensitisation placebo instructions commonly used in past research ( Steinmark 1974 ). The term "imagery relief" was, however, coined by the author. (...)The programme was, therefore, analogous to relaxation therapy/desensitisation but with the important omission of any known active ingredient, either theoretically or practically. Patientsreceived no instruction in dealing with sleeplessness per se.” ( Espie 1989b p. 105‐6,) Individual or group treatment: individual Exposure/intensity to treatment: not stated, but at least once weekly Duration of treatment: 8 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “Of the 84 patients, 58 (69%) were stated, by their GPs, to be "drugfree". Only 14 of these patients, however, had never been on hypnotic medication, and it transpired upon further assessment, that approximately one third of the remainder had not entirely discontinued medication. In most cases drug use was occasional and low dose. (…) Twenty—six patients were referred with persisting sleep difficulties who were also on nightly sleep medication. ” ( Espie 1989b , p. 101 ‐ 2) Wait‐list Comparison name: : no treatment (in reality a wait‐list) Description of intervention: "This group functioned as a waiting list control and had minimal therapist contact. Patients were seen after referral for the purposes of training in the use of the DSQ. This typically involved two appointments. Occasional contact by telephone was also made to ensure that sleep diaries were being completed as required, but at no time was advice or treatment offered. Subjects were seen again at the end of the ten week data collection period having thus provided data for the entire duration of the experimental period (...). Waiting—list subjects were, therefore, treated on an ad hoc individualised basis.” ( Espie 1989b , p. 107). Exposure/intensity to treatment: no treatment during waiting Duration treatment:: 10 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: "Of the 84 patients, 58 (69%) were stated, by their GPs, to be "drug free". Only 14 of these patients, however, had never been on hypnotic medication, and it transpired upon further assessment, that approximately one third of the remainder had not entirely discontinued medication. In most cases drug use was occasional and low dose. (…)Twenty—six patients were referred with persisting sleep difficulties who were also on nightly sleep medication. ” ( Espie 1989b , p. 101‐2) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: no |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”Ss were allocated according to a predetermined list of random numbers to either progressive relaxation (PR), stimulus control (SC), paradoxical intention (PI), imagery relief placebo (IR) or no treatment (NT).” (p. 81). |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported measures |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | No | Quote: ”The 84 included subjects represented 60% of the 141 referrals received. (...) In summary, half of the subject loss was due to the operation of strict selection criteria, and half due to subjects dropping out. Of the "drop‐outs", however, considerably more than one third failed to attend even the first appointment. The true "drop‐out" rate amongst assessed and suitable subjects was only 17 out of 101 patients. This attrition rate is at least comparable to clinical research studies in any field of application.” ( Espie 1989b , p. 180) Attrition >15% (15.5%). No ITT. Only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Etringer 1982.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three interventions
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 1 session (1 day – a maximum of 90 minutes) Duration of participation (trial + follow‐up): 1 day + 4 week follow‐up (but problems with assessment) Setting: outpatient (local community & college) Purpose of trial: “The present study compared PM to an attention‐placebo/treatment element control group that had been rated as initially equally credible for the treatment of severe snake‐avoidant behavior.” (p. 477) Open/Closed placebo: closed placebo |
|
| Data |
Number of participants screened: 41 Number of participants included: 38 Number of participants followed‐up at post treatment: not stated Number of participants randomly assigned to:
Number of withdrawals:: not stated Diagnosis: Specific anxiety (chronic fear of snakes) Diagnostic manual:: not stated Means of assessment: not stated Comorbidity: not stated Age: 23.7 mean years IQ: not stated‐ but 18/38 were college students Sex: : 81.6% female Ethnicity : not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Graduated subliminal modelling Description of intervention: “The subjects were given a theoretically neutral but highly credible placebo treatment couched in terms of modeling. The subjects were told that GSM was an effective method for getting rid of common fears. The stated rationale was that the method works by subjects' viewing tachistoscopically exposed slides of a model interacting with a snake. The model was ostensibly performing a graduated series of increasingly more threatening interaction behaviors with a fox snake. The subjects were told that by viewing these slides they would gradually come to learn that harmful consequences do not follow from interaction with the snake and that they would gradually overcome their fear. Subjects were also told that in order to optimize their progression through the interaction hierarchy, their heart rate and muscle tension would be monitored, thereby enabling the therapist to present the slides at such a pace that the subject would never become overly anxious.” (p. 478) Individual or group treatment: not stated Exposure/intensity to treatment : max 90 minutes Duration of treatment : 1 session (1 day) Concomitant psychotherapy : not stated Concomitant pharmacotherapy : not stated Wait‐list Comparison name: wait‐list Description of intervention: Subjects in the NTC condition participated in all assessment procedures without receiving any intervening treatment. Following pretreatment assessment, the subjects in this condition sat in the experimental room for the appropriate time period and were urged to read popular magazines that were made available.These subjects were given PM in the supplementary treatment phase of the experiment if they so desired.” (p. 479) Exposure/intensity to treatment : No treatment during waiting period Duration treatment: 1 session (1 day) Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Random assignment of subjects to conditions produced the following groups: PM contained 2 males and I I females (4 community and 9 college subjects) (p. 478) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Therapists were kept blind to the results of all assessment procedures. (p. 479) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Foa 1991.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 4.5 weeks Duration of participation (trial + follow‐up: 4.5 week. No follow‐up Setting: outpatient Purpose of trial: “We predicted that both PE and SIT would significantly reduce PTSD symptoms, more than would SC and WL.” (p. 716) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 55 Number of participants followed‐up at post treatment: 45 Number of participants randomly assigned to:
Number of patients reported in full report:
Number of withdrawals: n = 10
Diagnosis: Post‐traumatic stress‐disorder (PTSD) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition, Revised (DSM‐III‐R) Means of assessment: clinical interview Comorbidity: not stated, but several mental health diagnoses were excluded Age: 31.8 mean years (SD = 8.2) IQ:: not stated Sex: 100% female Ethnicity: Black 25%, White 72.7%, Hispanic 2.3%. Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name:: Supportive counselling Description of intervention: “Supportive counseling followed the nine‐session format, gathering information nine‐session format, gathering information through the initial interview in the first session and presenting the rationale for treatment in the second session. During the remaining sessions, patients were taught a general problem‐solving technique. Therapists played an indirect and unconditionally supportive role. Homework consisted of the patient's keeping a diary of daily problems and her attempts at problem solving. Patients were immediately redirected to focus on current daily problems if discussions of the assault occurred. No instructions for exposure or anxiety management were included.” (p. 718) Individual or group treatment: Individual Exposure/intensity to treatment: nine biweekly 90‐minute sessions Duration of treatment: 4.5 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: wait‐list (WL) Description of intervention: “WL subjects were informed that they would receive treatment in 5 weeks. During this period, they were contacted by a therapist between assessments to determine whether emergency services were required. Following an assessment at the end of the waitlist period, patients were randomly assigned to either PE or SIT.” (p. 718) Exposure/intensity to treatment: no treatment during waiting Duration treatment: 5 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflictsof interest: non‐financial conflict of interest: The first author (Foa EB) is the developer of the experimental intervention (prolonged exposure) Judgement: no |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ” After 10 patients were entered into the wait‐list condition, subsequent admissions were randomly assigned to one of the three treatment groups” (p. 716) |
| Allocation concealment | Unclear | No information. |
| Blinding of outcome assessors | Yes | Assessments at pretreatment, posttreatment, and follow‐up consisted of clinical interviews conducted by an independent assessor, who was blinded to treatment conditions, and self‐report questionnaires. (p. 717) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | No | Quote: “Dropout rates were not significantly different across the treatment groups, x^2 (3, N= 55) = 3.34, p > .30, and were as follows: PE 28.6%, SIT 17.6%, SC 21.4%, and WL 0%. (...) Subsequent analyses were conducted on data from the 45 completers.” (p. 718) Attrition >15% (PE 28.6%, SIT 17.6%, SC 21.4%, and WL 0%). No ITT. Only reports data on completers |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Foa 2018.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 2 weeks (available post‐treatment data) Duration of participation (trial + follow‐up): 8 weeks + 2 weeks follow‐up. There are also 12 weeks and 6 months follow‐up but no data on the minimal contact control group. Setting: outpatient Purpose of trial: to examine the effects of massed prolonged exposure therapy (massed therapy), spaced prolonged exposure therapy (spaced therapy), present‐centered therapy (PCT), and a minimal‐contact control (MCC) on PTSD severity Open/closed placebo : Closed placebo |
|
| Data |
Number of participants screened: 526 Number of participants included : 370 Number of participants followed‐up at post treatment: 245 Number of participants randomly assigned to:
Number of withdrawals before treatment: n = 55
Number of withdrawals post treatment: n = 70
Diagnosis: Post‐traumatic stress‐disorder (PTSD) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision (DSM‐IV‐TR) Means of assessment: clinical interview (not otherwise stated) Comorbidity: depressive symptoms Age:: mean psychological placebo: 32.54 years (SD = 7.45), wait‐list: 32.70 years (SD = 7.68) IQ:: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition Sex: 11.6% female Ethnicity: 32 Hispanic, 115 non‐Hispanic. 0 Asians, 29 Blacks, 95 whites, 23 other Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Present‐centered therapy Description of intervention: “Present‐centered therapy is a non–trauma‐focused, manualised treatment that controls for nonspecific therapeutic factors Ten 90‐minute sessions were scheduled similarly to spaced therapy and focused on current life problems that may or may not be PTSD‐related. Therapists helped participants identify stressors and discussed them in a supportive, nondirective manner.” (p. 356) Individual or group treatment: not stated Exposure/intensity to treatment: 10 sessions over 8 weeks for full treatment Duration of treatment: 2 weeks (data used). Full treatment was 8 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: 47.3% received 1 psychotropic medication or more Wait‐list Comparison name: Minimal contact control (wait‐list) Description of intervention: “The MCC condition consisted of 10‐ to 15‐minute therapist telephone calls once weekly for 4 weeks. Participants were asked about their well‐being, offered support as needed, and received contact information in case symptoms worsened.” (p. 356). “After the 2‐week follow‐up, participants in the MCC group were offered their choice of the other treatments” (p. 355) Exposure/intensity to treatment: minimal contact for 2 weeks. (post‐treatment) Duration treatment: 2 weeks + 2 weeks follow‐up Concomitant psychotherapy: not stated Concomitant pharmacotherapy: 30% received 1 psychotropic medication or more |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: non‐financial conflict of interest: the first author (Foa EB) is the developer of the experimental intervention (prolonged exposure) Judgment: no |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “Randomization was originally planned as 3:11:11:11 for MCC: massed therapy: spaced therapy: PCT. On January 5, 2012, enrolment in MCC was accelerated by changing the ratio to 1:1:1:1 to allow for preliminary massed therapy vs MCC comparison per Department of Defense request. After 40 participants were randomized to receive MCC, randomization to MCC was discontinued on March 19, 2014, and subsequent participants were assigned. To receive massed therapy, spaced therapy, or PCT(1:1:1).” (p. 355) Randomization pattern was dummy coded and then added as a moderator to the analyses. (p. 355) |
| Allocation concealment | Unclear | Quote: “The randomization sequence was entered by a study statistician into a secure, web‐based application using SAS version 9.4 (SAS Institute Inc), which was accessed by the project coordinator on enrollment of each participant.” (p. 355). Unclear whether project coordinator could have influenced allocation concealment. |
| Blinding of outcome assessors | Yes | Quote: “PTSD symptom severity was assessed by independent evaluators blinded to treatment condition, before and after treatment, and at 2‐week, 12‐week, and 6‐month follow‐up.” (p. 355) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Linear mixed models and generalised linear mixed models were used to analyse the data, using SPSS version 23 (IBM SPSS). These models are intent‐to‐treat and calculate results based on available data without imputation of missing data (p. 356) Attrition >15% (Active treatment 41% ‐ but only 12.1% in placebo and 0% in wait‐list). No ITT |
| Selective outcome reported | No | Trial registry: NCT01049516. Veterans RAND 12‐items HealthSurvey and Adverse events not mentioned in Clinical Trial Registry. |
| Other sources of bias | Yes | No other sources found |
Freire 2007.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 10 weeks Duration of participation (trial + follow‐up): 10 + 2 weeks = 12 weeks post‐treatment Setting: outpatient Purpose of trial: “To investigate the efficacy of acupuncture in the treatment of moderate obstructive sleep apnea syndrome (OSAS), assessed by polysomnography (PSG) and questionnaires of functional quality of life (SF‐36) and excessive daytime sleepiness (Epworth)” (p. 43) Open or closed placebo: closed placebo |
|
| Data |
Number of participants screened: 38
Number of participants included: 36
Number of participants followed‐up at post treatment: 26 Number of participants randomly assigned to:
Number of withdrawals: n = 10
Diagnosis: obstructive sleep apnoea (sleep‐wake disorder) Diagnostic manual:: not stated Means of assessment: diagnosis “confirmed by a full polysomnographic (PSG) study with an apnea/hypopnea index (AHI) >15/hour and <30/hour (moderate OSAS” (p. 44) + clinical interview Comorbidity: not stated Age: 54.67 mean years, (Range = 49 to 54) IQ :not stated – but participants with intellectual deficits were not eligible Sex: 55.6% female Ethnicity: not stated Country: Brazil Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo
Treatment name: sham acupuncture
Description of intervention: “The sham acupuncture group was stimulated with the same number of needles as the acupuncture group, and the points were localized 1 cun from the real point, in a region not related to any acupoints or meridians and was done following the standards of minimal acupuncture. For the sham acupuncture group, the needles were inserted and no manipulation was done. (…) All acupuncture procedures, as well as sham acupuncture, were performed by an experienced physician, who was a specialist in acupuncture, according to traditional Chinese acupuncture methods. All procedures were performed in the afternoon between 3 p.m. and 6 p.m. Body needles were left in situ for 30 min.” (p. 45) “Finally, patients were informed that at the end of the study all patients allocated to the sham acupuncture group would receive 10 sessions of acupuncture treatment if they so wanted.” (p. 44)
Individual or group treatment: individual
Exposure/intensity to treatment: once a week for 10 weeks
Duration of treatment: 10 weeks
Concomitant psychotherapy:: not stated
Concomitant pharmacotherapy: not stated – but excluded if taking hypnotic drugs Wait‐list Comparison name: control group (wait‐list) Description of intervention: “Patients assigned to the control group were offered weight reduction advice if overweight and sleep hygiene counseling. Given the usual waiting list for nCPAP at our service, which is about 6 months, the waiting time was not a matter of ethical concern.” (p. 44) Exposure/intensity to treatment: nothing during waiting Duration treatment: 3 months (12 weeks) Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated – but excluded if taking hypnotic drugs |
|
| Outcomes |
Beneficial outcomes :
Adverse events
|
|
| Notes |
Key conclusion
Key limitations
Other notes from review authors
Conflicts of interest: none Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “Randomisation was done by a blinded independent researcher and was conducted by selecting a closed piece of paper out of a box, with a treatment order written on it. (p. 44) |
| Allocation concealment | Yes | Only the physician applying the treatments (acupuncture/ sham acupuncture) was aware of which group each patient had been assigned to and did not participate in any phase of the subsequent evaluation.” (p. 44) |
| Blinding of outcome assessors | Yes | Quote:“All of the PSG recordings were assessed by two experienced sleep physicians (S.M. Togeiro and F.S.Chrispin), who were blind to the groups to which the patients had been assigned.” (p. 46) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Attrition >15% (27.8%). No ITT. Only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Fuchs 1977.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 6 weeks Duration of participation (trial + follow‐up): 6 weeks (Post‐treatment assessment at 7 weeks) + 6 weeks follow‐up (WL not assessed at follow‐up) Setting: outpatient Purpose of trial: “A behavior therapy program based on a self‐control model of depression was evaluated against a nonspecific group therapy condition and a waiting list control group.” (p. 206) Open/closed placebo : Closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 36 Number of participants followed‐up at post treatment: 28 N umber of participants randomly assigned to:
Number of withdrawals : n = 8
Diagnosis: depression Diagnostic manual: not stated Means of assessment: Minnesota Multiphasic Personality Inventory (MMPI) Comorbidity: not psychotic, suicidal, no history of psychiatric hospitalisation Age: 28.8 years (range = 18 to 48) IQ: not stated Sex: 100% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name (type): nonspecific therapy Description of intervention: “Session 1 began in the same way as the self‐control procedure with introductions, collection of deposits, a review of confidentiality issues, and a 10‐minute group interaction assessment procedure. As in the other groups, subjects were given an information sheet and a general introduction to group therapy concepts, generally from a nondirective framework. From that point on and throughout the ensuing sessions, therapists in this condition attempted to elicit discussion of past and current problems, to encourage group interaction, and to reflect and clarify feelings in an empathic manner. Although therapists at times suggested simple exercises within the group to facilitate open discussion, they were specifically instructed neither to recommend out‐of‐therapy activity nor explicitly to teach behavioral principles. These sessions lasted approximately 2 hours weekly, as did self‐control therapy sessions.” (p. 209) Individual or group treatment: group Exposure/intensity to treatment: 2 hours weekly Duration of treatment: :6 weeks Concomitant psychotherapy: no involvement in any other therapy for problems related to psychological functioning within the past month Concomitant pharmacotherapy: not stated Wait‐list Comparison name: wait‐list control Description of intervention : “Subjects in this condition were informed by phone that they had been accepted into the research program but that our present groups were filled, so that they would have to wait about 8 weeks before their groups would start. They were also told that they would be required to retake some of the screening tests just prior to beginning therapy; however, they were assured of being seen.” (p. 209) Exposure/intensity to treatment: no treatment during waiting Duration treatment: 6 weeks Concomitant psychotherapy: no involvement in any other therapy for problems related to psychological functioning within the past month Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: non‐financial conflict of interest: Authors developed the experimental intervention Judgement: no |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”Except where necessary to balance experimental conditions for mean age and severity of depression, subjects were randomly assigned to one of two therapists and one of three treatment conditions—self‐control therapy, nonspecific therapy, or waiting list control.” (p. 209) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | No | Quote: “Eight of the original 36 subjects dropped out of the study, all within the first 2 weeks. (...) Drop‐out rate did not differ significantly between conditions, x2 ( 2 ) = .29, p < .80. Dropouts did not differ from remainders on age, Depression Inventory, MMPl D, or MMPI total elevation scores.” (p. 210) Attrition >15% (22.2%). No ITT. Only reports data on completers |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Glogowska 2000.
| Study characteristics | ||
| Methods |
Parallel randomised trial with two arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): mean treatment 8,4 months (0,9 to 12 months). post treatment data at 12 months Duration of participation (trial + follow‐up):: 12 months ‐ no follow‐up Setting: outpatient Purpose of trial: “To compare routine speech and language therapy in preschool children with delayed speech and language against 12 months of “watchful waiting”. (p. 1) |
|
| Data |
Number of participants screened: 507 Number of participants included: 159 Number of participants followed‐up at post treatment: 155 Number of participants randomly assigned to:
Number of withdrawals: n= 4
Diagnosis: delayed speech and language Diagnostic manual: not stated Means of assessment: not stated Comorbidity: 13 diagnosed with hearing loss Age: age in months, usual care 34.2 months (range = 18 to 42), wait‐list 34.2 months (range = 24 to 42) IQ: not stated Sex: usual care: 23% female. wait‐list: 26% female Ethnicity: not stated Country: UK Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Usual care Treatment name: therapy Description of intervention: “Therapy provided in the study tended to focus on several areas of language simultaneously. Therapy techniques included Derbyshire language scheme tasks, as well as everyday play and games used as contexts for modelling language for the child. Goals covered a wide range of language stages – for example, understanding and building single words, using narratives, and identifying consonants in words.” (p. 4) Individual or group treatment: individual. “Children randomised to the therapy group received the one‐to‐one speech and language therapy (…)” (p. 2) Exposure/intensity to treatment : 6.2 (0‐15) hours of therapy. Frequency of therapy were once a month (range once a week to once every two and a half months) Duration of treatment: 8.4 months (range = 0.9‐12) ‐ number of months over which the therapy took place Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: watchful waiting Description of intervention: “Parents of children in the watchful waiting group could request therapy at any time if they were concerned about their child’s progress. All children in the study were reassessed by the research therapists after 12 months; if a child in the watchful waiting group were still experiencing difficulties, two research therapists (SR and MG) provided up to 12 therapy sessions.” (p. 2) Exposure/intensity to treatment: no treatment Duration treatment: 12 months (after all received treatment and assessments) Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: no |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: “Randomisation was stratified by the 16 clinics and by the three clinical criteria (general language, expressive language, and phonology) (…) The sequence of random numbers was generated before the trial independently of the therapists.” (p. 2) |
| Allocation concealment | Yes | Quote: “The allocation was implemented by the therapists opening sealed opaque envelopes (coloured according to the three clinical criteria) in the presence of the parents.” (p. 2) |
| Blinding of outcome assessors | Yes | Quote: “Assessors were blind to previous results, and every attempt was made to maintain blindness in terms of allocation. The presence of the parent meant that this was often inevitably compromised, but each child was seen by a different therapist for the two follow ups, and the language sample for the Bristol language development scales was analysed in a fully blinded manner.” (p. 2) |
| Blinding of participants and personnel | No | Not possible to blind usual care and wait‐list |
| Incomplete outcome data | Yes | Quote: “The trial arms were compared on an “intention to treat” basis.” (p. 2) “*Data were missing for all measures in both groups: analyses were based on 64 (therapy group) and 80 children (watchful waiting group) for auditory comprehension; 63 and 77 for expressive language; 57 and 62 for the phonology error rate; and 71 and 84 for improvement by 12 months.” (p. 4) Attrition <15% (2.5%). ITT used |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Goldstein 2000.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 4 weeks Duration of participation (trial + follow‐up): 4 weeks + 1 month follow‐up Setting : Outpatient Purpose of trial: “Accordingly, the purposes of the present study were twofold: (a) to conduct a replication of Feske and Goldstein's comparison of EMDR to a waiting list control group for PDA and (b) to contrast EMDR with a credible attention‐placebo.” (p. 948) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 46 Number of participants followed‐up at post treatment: 45 Number of participants randomly assigned to:
Number of withdrawals: n = 1
Diagnosis: agoraphobia Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: the Structured Clinical Interview for DSM‐IV Disorders (SCID) Comorbidity: 20 participants had at least one comorbid Axis I diagnosis: specific phobia (7), generalised anxiety disorder (6), social phobia (5), or obsessive—compulsive disorder (2). Of these, 5 had more than one Axis I comorbid condition. Three participants met criteria for obsessive— compulsive personality disorder, and 4 for avoidant personality disorder Age: 38.16 mean years, (range =22 to 63) IQ:: not stated, but 38 had attended at least some college Sex: 80.4% female Ethnicity: two were African American, and one was Asian American; the remainder were European American Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: attention placebo Description of intervention: “ART, the attention‐placebo treatment, included a combination of two relatively inert treatment procedures: 30—45 min of progressive muscle relaxation training and 45 60 min of association therapy” (p. 952) Individual or group treatment: Individual Exposure/intensity to treatment: six 90‐minute sessions held over an average of 4 weeks Duration of treatment: 4 weeks Concomitant psychotherapy: no other treatment. Excluded if they had Concomitant pharmacotherapy: excluded if taking medication. “Participants excluded on the basis of recent medication changes were eligible for reconsideration once medications were stabilized in appropriate limits.” (p. 950) Wait‐list Comparison name: wait‐list Description of intervention: “For 2 weeks prior to and after treatment or waiting list, as well as throughout the course of treatment or waiting period, participants completed anxiety forms every morning and evening and at the close of each week.” (p. 950) Exposure/intensity to treatment: waiting for treatment Duration treatment: 4 weeks. “Once the waiting list period ended, all those assigned to waiting list were randomized to EMDR (n = 6) or attention‐placebo ( n = 7).” (p. 949) Concomitant psychotherapy: no other treatment. Excluded if they had Concomitant pharmacotherapy: excluded if taking medication. “Participants excluded on the basis of recent medication changes were eligible for reconsideration once medications were stabilized in appropriate limits.” (p. 949) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: no |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Participants were initially randomly assigned to one of three groups: waiting list ( n = 14), EMDR ( n = 18), or an attention‐placebo condition ( n = 13) involving the same amount of therapist contact as EMDR (p. 3) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Quote: ”Raters were not blind to group assignment.” (950) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Quote: ”Dropouts were replaced with the next participant to enter the study” (p. 949) ”Intention‐to‐treat analyses were conducted at each assessment period by repeating ANOVAs and ANCOVAs with pretest scores carried forward to serve as posttest or follow‐up scores for those who failed to provide posttest data or who dropped out before the conclusion of treatment or before the follow‐up assessment. The findings of the EMDR versus waiting list and EMDR versus ART comparisons were unchanged.” (p. 955) Quote: “Fisher;s exact tests indicated that attrition was not significantly different across groups (EMDR vs. attention‐placebo p= .242; EMDR vs. waiting list p= 1.00). Of the 42 participants who completed treatment, 37 provided follow‐up data. Of those who dropped from follow‐up after EMDR, one required medical attention for an unrelated condition, one terminated because of increased distress during treatment, and a third refused assessment without explanation. Of those who dropped from attention‐ placebo, one dropped because of his disappointment with treatment and the other without explanation.” (950) Attrition > 15% (19.6%). ITT used |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Goldwasser 1987.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 5 weeks Duration of participation (trial + follow‐up): 5 weeks + 6 weeks follow‐up Setting: inpatient (resident population at Beth Sholom Home in Richmond, Virginia) Purpose of trial: “This article presents a controlled study designed to determine the degree to which reminiscence group therapy influences affective, cognitive, and behavioral functioning in demented elderly.” (p. 210) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 30 Number of participants included: 27 Number of participants followed‐up at post treatment: 24 Number of participants randomly assigned to:
Number of withdrawals : n = 3
Diagnosis: dementia Diagnostic manual: not stated Means of assessment: not stated Comorbidity: Alzheimers’s multi‐infarct, dementia secondary to medical disorder Age: 82.3 mean years (range = 70 to 97) IQ: not stated Sex: 74.1% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: attention‐placebo support group Description of intervention: “A second group consisted of a support group that focused on present or future events and problems. This group also met for a half hour twice weekly for a period of five weeks.” (p. 212); “In order to ensure that the reminiscence component of the intervention accounted for any observed changes, an attention‐placebo “support” group and a “no‐treatment” control group were also used.” (p. 210) Individual or group treatment:: group Exposure/intensity to treatment: 30 minutes twice weekly Duration of treatment: 5 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: no‐treatment Description of intervention: The third group served as a "no‐treatment" control group, and consequently did not participate in any group activity during the same period of time.” (p. 212) Exposure/intensity to treatment: no treatment Duration treatment: 5 weeks Concomitant psychotherapy:: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: "(...) randomly assigned to three groups of ten people each. (p. 210) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Quote: ”Since one participant in the reminiscence group died during the course of the study, one participant from each of the other treatment groups was randomly dropped from data analyses” (p. 210) Attrition <15% (11.1%). |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Hekmat 1984.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 3 sessions in total Duration of participation (trial + follow‐up): follow up occurred within 4 weeks. (p. 465) Setting: outpatient (college) Purpose of trial: “This study explored the clinical effectiveness of semantic desensitization in the treatment of public speaking anxiety. (p. 463) Closed/open placebo : Closed placebo |
|
| Data |
Number of participants screened: 239 Number of participants included: 30 Number of participants followed‐up at post treatment: not stated Number of participants randomly assigned: not stated Number of withdrawals: not stated Diagnosis: social anxiety (speech‐anxious students) Diagnostic manual: not stated Means of assessment: speech anxiety was measured by the following scales: Personal report of confidence as a speaker (PRCS), Affect Adjective Checklist (ACL), S‐R Inventory of Anxiousness, and Timed Behavior Checklist (BCL) Comorbidity: not stated Age: not stated IQ: not stated ‐ but university students Sex:: 60% female Ethnicity:: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: attention placebo Description of intervention: “Ss in this group were informed that they would receive a novel therapy called “systematic ventilization.” Ss were instructed that awareness of anxiety and the ways in which it would manifest itself in behavior is essential for cure.” (p. 464) Individual or group treatment: individual Exposure/intensity to treatment: 3 sessions Duration of treatment: not stated Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: waiting list control. (in reality no‐treatment) Description of intervention: “Ss in the no treatment waiting list control were instructed that periodic measurement of their anxiety reaction was essential to procure a reliable assessment of their problem. The no treatment waiting list control Ss also were given the pretreatment, posttreatment, and follow‐up anxiety measures.” (p. 464) Exposure/intensity to treatment: not stated Duration treatment: not stated Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: "Ss were volunteers who were matched on the basis of their pretreatment anxiety scores and randomly assigned to one of the following treatments: Group I, semantic desensitization therapy; Group 11, placebo control; and Group 111, waiting list control." (p. 463) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: ”The two behavioral assessors were blind to treatment assignments.” (p. 464) |
| Blinding of participants and personnel | No | Not possible to blind placebo and "wait‐list" |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Hippman 2016.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): not stated Duration of participation (trial + follow‐up:: not stated, but was done during September 2008–November 2011 Setting: inpatient and outpatient Purpose of trial: “We hypothesized that 1) mean scores for knowledge, risk perception accuracy, and perceived control over illness would be higher, and scores for internalized stigma would be lower for the GC group compared to an intervention group provided with an educational booklet (EB), and 2) mean differences in scale scores between outcome (T3) and baseline (T1) for the two intervention groups (GC, EB) would be significantly different than waitlist, with GC/EB mean scores being higher for knowledge, risk perception accuracy, and perceived control over illness, and lower for internalized stigma.” (p. 3) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 120 Number of participants followed‐up at post treatment: 112 Number of participants randomly assigned to:
Number of withdrawals (post‐treatment): n = 8
Diagnosis: serious mental illness: Bipolar disorder (69.2%), Schizophrenia (16.7%), Schizoaffective Disorder (10.8%), Other (Major depression and Major depression with psychosis) (3.3) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: the Structured Clinical Interview for DSM‐IV Disorders (SCID) Comorbidity: a variety of different mental health disorders Age: 41.6 mean years (range = 17 to 73) IQ: >70, 76.5% attended college or university Sex:: 60.7% female Ethnicity: not stated Country: Canada Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: educational booklet Description of intervention: “The EB intervention was designed as a rigorous control intervention; it was face‐to‐face and provided the same general information as GC, but without the ‘active ingredient’ of personalization of information/counseling by a BC/EGC.” (p. 4) “EB sessions (~30 minutes) were provided by the research coordinator (AR), who answered questions regarding literal interpretations of text, but responded to participants’ queries that aimed to make personal meaning of the material with responses such as: “I’m sorry, but I’m afraid I’m unable to answer that. If you’d like to meet with someone who can help you with questions like that, we can set up a GC appointment after you finish the study”. Thus, EB sessions did not evolve into GC, yet were a stringent control intervention. Through observation, the research coordinator confirmed participant adherence to the intervention. The booklet (16 color pages, reading grade level 8) was designed in collaboration with individuals with SMI and included: a graphical depiction of the concepts of vulnerability (genetic and environmental) and resilience (the “mental illness jar”), with specific examples and a table of general RRs for relatives of people with SMI.” (p. 4‐5) Individual or group treatment: individual Exposure/intensity to treatment: 30 minutes Duration of treatment: not stated Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: wait‐list (in reality no‐treatment) Description of intervention: “For the waitlist group, baseline and T1 occurred on the same day. Participants had the option of bringing a support person with them to appointments if they wished. In‐person visits were arranged for some participants to complete the outcome measures at one month follow‐up at their request. One of the participants in the waitlist group had received GC for SMI prior to the study. The trial was stopped once the pre‐determined number of participants had been recruited and those who were not lost to follow up had completed the study. The full protocol can be obtained from the corresponding author.” (p. 14) Exposure/intensity to treatment: none Duration treatment: not stated Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “For the randomization procedure, equally‐sized laminated cards were sorted into two opaque envelopes (one for males, containing 18 GC, and 17 of each EB and WL, and one for females, containing 22 GC, and 23 of each EB and WL). .” (p. 4) |
| Allocation concealment | Unclear | Quote: "Participants were asked to choose a card from the appropriate (male/female) envelope without looking (under the supervision of AR or AI).” (p. 4) |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and "wait‐list" |
| Incomplete outcome data | Yes | Quote: “While the nature of the study and interventions precluded blinding for participants or providers, an independent party blind to group status conducted data analyses.” (p. 4) Attrition <15% (6.7%). Analysed data on everyone that received the treatment. Linear mixed effects models used. Blinded data analyst |
| Selective outcome reported | Yes | NCT00713804. No differences in trial registry and full report |
| Other sources of bias | Yes | No other sources found |
Howlin 2007.
| Study characteristics | ||
| Methods |
Cluster‐randomised trial with three arms
Sample calculation: yes Cluster randomised: yes Duration of trial (baseline to post): 5 months Duration of participation (trial + follow‐up): 1 week + 5 months Setting: outpatient (school classroom) Purpose of trial: “To assess the effectiveness of expert training and consultancy for teachers of children with autism spectrum disorder in the use of the Picture Exchange Communication System (PECS).” (p. 473) |
|
| Data |
Number of participants screened: 38 classes Number of participants included: 18 classes (88 participants) Number of participants followed‐up at post treatment: not stated (84 participants) Number of participants randomly assigned to :
Number of withdrawals : n = 4
Diagnosis: autism Diagnostic manual: not stated, but “All children had received a clinical diagnosis of autism prior to enrolment in the study” (p. 476) Means of assessment: the Autism Diagnostic Observation Schedule‐Generic (ADOS‐G) Comorbidity: not stated Age: 6.8 mean years IQ: :not stated Sex: 17% female Ethnicity: not stated Country: UK Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Wait‐list Treatment name: delayed treatment group Description of intervention: DTG: Receiving PECS training 2 terms after initial baseline assessment. Exposure/intensity to treatment: no treatment during waiting Duration of treatment: 5 months Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name :no‐treatment group Description of intervention: receiving no PECS training Exposure/intensity to treatment: no treatment Duration treatment: 5 months Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: "In each stratum, classes were randomly allocated to one of the three treatment conditions using an online randomisation programme (http://www.random.org)" (p. 475) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Quote:"Fourthly, the assessors were not blinded to group allocation or treatment phase, as financial limitations precluded the use of additional blinded raters to code all the video recordings." (p. 479) |
| Blinding of participants and personnel | No | Not possible to blind wait‐list and no‐treatment |
| Incomplete outcome data | Yes | Quote:“Following random assignment, one class (ITG) subsequently withdrew from the study. One girl entered a DTG class one year into the study; thus her data were available from Time 2–Time 3 only. At baseline, one other girl (NTG) failed to meet criteria for ASD." (p. 477) Excluded from further analysis. Seven children moved out of the DTG during the watching waiting period and did not receive treatment but they were assessed at Times 2 and 3 and their data included in the analyses on an intention‐to‐treat basis. The final groups were: ITG (5 classes, 26 children, 21 boys, 5 girls); DTG (6 classes, 30 children, 27 boys, 3 girls); NTG (6 classes, 28 children, 25 boys, 3 girls).” (p. 478) Attrition <15% (5.6%). |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources of bias found |
Karst 2007.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation:: yes Cluster randomised:: no Duration of trial (baseline to post): 1 treatment (1 day) Duration of participation (trial + follow‐up): 1 treatment (1 day) Setting: outpatient Purpose of trial : “Therefore, we designed a study to determine whether auricular acupuncture can decrease acute dental anxiety and compared it with the standard pharmacological sedative medication midazolam, noninvasive placebo auricular acupuncture, and no treatment.” (p. 295) Open or closed placebo: closed placebo |
|
| Data |
Number of participants screened: 81 Number of participants included: 67 Number of participants followed‐up at post treatment: 67 Number of participants randomly assigned to:
Number of withdrawals: 0 Diagnosis: specific anxiety (dental anxiety) Diagnostic manual: not stated Means of assessment: not stated Comorbidity: not stated Age: 38 to 49 mean years (SD = 13.09) IQ: not stated Sex: 44.8% female Ethnicity: not stated Country: Germany Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo Treatment name: Placebo auricular acupuncture group Description of intervention: “In addition, patients in the placebo auricular acupuncture group were told that the needles would only be inserted gently and superficially and that an elastic cube would, therefore, be necessary to support the needle” (p. 296) “This group received placebo ear acupuncture by using the finger and liver points, which do not have any documented effects on anxiety reduction. A placebo needle system was used, in which the tip of the needle is blunt so as to cause a pricking sensation mimicking real acupuncture without actually puncturing the skin. To support the needle, an elastic foam was used which was fixed upon the area of the acupoint. In contrast to superficial sham acupuncture, this form of control may be associated with less unspecific physiological effects.” (p. 296) Individual or group treatment: individual Exposure/intensity to treatment: 1 treatment Duration of treatment: 1 day Concomitant psychotherapy:: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: no treatment Description of intervention: not stated Exposure/intensity to treatment: no treatment Duration treatment: not stated Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “The names of the recruited patients were transmitted to the Department of Biometrics, Hannover Medical School. A list with random numbers was prepared by one of its members (L.H.).” (p. 296) |
| Allocation concealment | Yes | Author L.H. made the list and randomised. “statistical procedures (L.H.) (…) were blind to treatment condition” (p. 297) |
| Blinding of outcome assessors | Yes | Quote: “Both the investigators performing follow‐up examinations (A.H.) and statistical procedures (L.H.), and the dentist were blind to treatment condition” (p. 297) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Attrition <15% (0%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Kelley 2012.
| Study characteristics | ||
| Methods |
Parallel randomised trial with two arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 2 weeks Duration of participation (trial + follow‐up): 2 weeks (post treatment data) + 2/4 weeks of placebo treatment (no follow‐up). Setting: outpatient Purpose of trial: investigating if open‐label placebo can be used as a first‐line treatment for depression Open or closed placebo: open placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 20 Number of participants followed‐up at post treatment: 15 Number of participants randomly assigned to:
Number of withdrawals: n = 5
Diagnosis: non‐psychotic Major Depressive Disorder Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: the Structured Clinical Interview for DSM‐IV Disorders (SCID) Comorbidity: “While some comorbid conditions resulted in patients being excluded (e.g., schizophrenia), many other comorbid conditions were allowed (e.g., Generalised anxiety disorder (GAD), so long as the GAD was not primary over major depressive disorder (MDD)).” ( Kelley 2012 (pers comm) ) Age: 38.8 mean years (SD = 12.6) IQ: not stated Sex: 70% female Ethnicity:: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: Open‐label placebo
Description of intervention: Patients were instructed to take two placebo pills, twice daily. The placebos were blue capsules containing microcrystalline cellulose” (p. 1).
Individual or group treatment individual
Exposure/intensity to treatment: 2 placebo pills twice daily
Duration of treatment: 2 weeks (post treatment data) + 2 weeks
Concomitant psychotherapy: not allowed (see exclusion criteria)
Concomitant pharmacotherapy: not allowed (see exclusion criteria) Wait‐list Comparison name: wait‐list control Description of intervention: waiting for treatment/placebo Exposure/intensity to treatment: waiting for treatment Duration treatment :2 weeks Concomitant psychotherapy: not allowed (see exclusion criteria) Concomitant pharmacotherapy: not allowed (see exclusion criteria) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “The randomization itself was created by the biostatistician using a computer to generate the sequence of assignments.” ( Kelley 2012 (pers comm) ) |
| Allocation concealment | Yes | Quote:“ prior to enrollment and the revelation of treatment assignment, the randomization was concealed from both the clinician and patient.” ( Kelley 2012 (pers comm) ) |
| Blinding of outcome assessors | Yes | Quote:“Blinded clinicians assessed patients at baseline and every two weeks thereafter. The primary outcome was the clinician‐rated 17‐item Hamilton Scale for Depression.” (p. 1). “In addition, all assessments were conducted by assessors who were blinded to treatment allocation.” ( Kelley 2012 (pers comm) ) |
| Blinding of participants and personnel | No | Open‐labelled placebo. Quote:“(1) Since this was a trial of open‐label placebo vs. no treatment control, patients and clinicians were not blinded during treatment.” ( Kelley 2012 (pers comm) ) |
| Incomplete outcome data | Unclear | Attrition >15% (25%). No mention of ITT |
| Selective outcome reported | No |
NCT01103271 Different primary outcome measure – feasibility (timeframe; one year) in protocol, while HAMD‐17 in the full report |
| Other sources of bias | Yes | No other sources found |
Kennedy 1974.
| Study characteristics | ||
| Methods |
Parallel randomised trial with six arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 1 year (max. of 6 sessions) Duration of participation (trial + follow‐up): 1 year Setting: outpatient (college) Purpose of trial: “Thus, another purpose of the present study was to obtain a more accurate picture of the relationship between anxiety decrements and approach behavior at various stages of performance on the behavioral avoidance test.” (p. 722‐3) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 81 Number of participants included: 74 Number of participants followed‐up at post treatment: 60 Number of participants randomly assigned to:
Number of withdrawals : n = 14
Diagnosis: specific anxiety (snake) Diagnostic manual: not stated Means of assessment:: behavioural avoidance test Comorbidity: not stated Age: not stated IQ: not stated – but college students Sex: 100% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Pseudo‐desensitisation Description of intervention: “Subjects in this control condition received the same type and amount of relaxation training as subjects in the desensitization groups. In contrast to the latter groups, however, relaxation was paired with snake‐irrelevant stimuli during the subsequent therapy sessions. That is, pseudo‐desensitization subjects were instructed to relax and imagine neutral, pleasant scenes such as walking in the mountains, sailing, frolicking at the beach, etc. Pseudo‐desensitization subjects were matched with subjects in the 100% desensitization group in terms of the number of treatment sessions and the time of the posttreatment assessment.” (p. 722) Individual or group treatment: individual Exposure/intensity to treatment: 40‐minute sessions for 6 sessions Duration of treatment: 1 year Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: untreated (no treatment) Description of intervention: “Untreated subjects participated only in the pretreatment and posttreatment assessment procedures. Posttreatment evaluation was conducted at approximately the same interval as for subjects in the 100% desensitization group.” (p. 722) Exposure/intensity to treatment: no treatment Duration treatment: 1 year Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: “Assignment to groups was random, with the constraint that extreme pretreatment BAT scores be distributed in order to keep pretreatment means reasonably equal.” (p. 722) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:“The experimental assistant who administered the BAT participated only in the assessment procedures and had no knowledge of which group the subjects represented.” (p. 722) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Attrition >15% (18.92%). No ITT. Only reports data on completers |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Kilmann 1987.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: not stated Cluster randomised: no Duration of trial: 4 weeks Duration of participation (trial + follow‐up):: 4 weeks + 6 months follow‐up. However, the same participants were used in Kilmann 1985 and Kilmann 1988 . In these they were compared to a healthy sample. Setting:: outpatient (WJB Dorn Veterans Hospital in Columbia) Purpose of trial: Testing the effect of Group treatment (Communication Technique Training, Sexual Technique Training, Combination Treatment) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 21 Number of participants followed‐up at post treatment: 20 Number of participants randomly assigned to:
Number of withdrawals: n = 1, one man dropped out after the first week of treatment due to an unexpected illness which required hospitalisation. Diagnosis: erectile dysfunction Diagnostic manual: not stated Mean of assessment: Clinical Interview (Sexual Interaction Inventory) Comorbidity: not stated Age:: 51 mean years, (range = 31 to 67) IQ: not stated – their education average was 15.3 years, with a range of 12 to 20. Sex: 100% male (and with their respective partners) Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pscyhological placebo Treatment name: Attention placebo control Description of intervention: “This format was conducted for eight 2‐hour sessions for a total of 16 hours. The format was designed to control for the therapist and treatment variables thought to be inherent in the Communication Technique Training, Sexual Technique Training, and the Combination Treatment formats. (p. 172) Individual or group treatment: group‐format. Exposure/intensity to treatment: 8x 2 hours sessions (total 16 sessions) Duration of treatment: 4 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “Nine of the 20 men were taking some type of medication on a regular basis; the medication was not presumed to interfere with the ability to gain and maintain an erection in sexual situations.” (p. 175) Wait‐list Comparison name: no treatment (in reality wait‐list) Description of intervention: ”The couples in the No‐Treatment Control group did not receive any treatment for a 5‐week waiting list period; they responded to the outcome measures in the same 5‐week pre‐ to posttesting time interval as the couples in the other experimental groups. After posttesting, these couples received two 2‐hour sessions of sex education followed by the Combination Treatment format described above.” (p. 172) Individual or group treatment: group‐format Exposure/intensity to treatment: none (wait‐listed) Duration treatment: 5 weeks of waiting Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “Nine of the 20 men were taking some type of medication on a regular basis; the medication was not presumed to interfere with the ability to gain and maintain an erection in sexual situations.” (p. 175) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote:”Their assignment to a particular group would be done on a random, statistical basis and would have nothing to do with them personally” (p. 175) “A table of random numbers determined that the therapist would conduct the treatment formats in the following sequence: Attention‐Placebo control, Combination Treatment, Communication Technique Training, No‐Treatment control, Sexual Technique Training.” (p. 176) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | Yes | Attrition <15% (5%). Only 1 person dropped out after 1 week of trial. Not stated which group he was a part of. No ITT |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Klein 1977.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 3 weeks Duration of participation (trial + follow‐up:: 3 weeks + 1 week follow‐up Setting: outpatient Purpose of trial: “This study explored the effectiveness of progressive relaxation and large muscle exercise in improving the cognitive performance of hyperactive, impulsive males..” (p. 1159) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 24 Number of participants followed‐up at post treatment: not stated Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: hyperactivity/impulsivity Diagnostic manual: not stated Means of assessment: “The rating scale was modeled after Conners' (1969) scale and consisted of short behavioral definitions of restlessness, impulsivity, distractibility, and short attention span. Employing a 1 to 4 rating scale (not at all, small degree, generally, very much) teachers rated the degree to which each behavioral description characterized the child's typical classroom behavior. Scores were summed across the four behaviors with greater scores representing greater hyperactivity.” (p. 1160) Comorbidity: not stated Age: not stated – but third grade IQ: not stated Sex:: 100% male Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Attention‐placebo Description of intervention: “The task of making various objects from "Play Doh" was assigned to the attentional‐training group. This task was chosen because it appeared neither especially rewarding nor boring. Each child in this control group worked by himself at a table inside the van.” (p. 1160) Individual or group treatment: individual Exposure/intensity to treatment: five sessions, 20 minutes each Duration of treatment: 3 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: No‐treatment Description of intervention : “Subjects in the no‐treatment and nonhyperactive controls received no intervention throughout the 3‐wk. period.” (p. 1160) Exposure/intensity to treatment: no treatment Duration treatment:: 3 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: The 24 most "hyperactive, impulsive" males (3 from each class) were randomly assigned to one of four treatments: (a) muscle relaxation, (b) large muscle exercise, (c) attention‐placebo control, (d) no‐ treatment control. (p. 1160) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:“The testing took place at the subject's school and was administered by an examiner who was not familiar with the subjects or the hypotheses of the study.” (p. 1160) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Klerman 1974a.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms High interpersonal contact:
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 8 months. Duration of participation (trial + follow‐up:: 8 months (no follow‐up) Setting: outpatients Purpose of trial: evaluate maintenance treatment of depression including feasibility, efficacy and safety. Closed or open placebo: closed placebo |
|
| Data |
Number of participants screened: 278 Number of participants included: 150 Number of participants followed‐up at post treatment: 139 (completed or relapsed). Number of participants randomly assigned to:
Number of withdrawals : n = 6
No‐treatment: n = 3
Diagnosis: depression Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition (DSM‐III) Means of assessment: Brief Psychiatric Rating Scale (BPRS) Comorbidity: ”85 percent of our patients were diagnosed as having neurotic depressions, according to criteria from the American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders” p. 187. Ag:: 39 mean years IQ: not stated Sex: 100% female Ethnicity: 83.3% white Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: Placebo group + high interpersonal contact
Description of intervention: no description of pharmacological placebo “The high contact group met with a social worker for a minimum of one hour a week” (p. 187).
Individual or group treatment: individual
Exposure/intensity to treatment: exposure to pharmacological placebo is not stated. High contact group: met with a social work for a minimum of one hour a week.
Duration of treatment: 8 months.
Concomitant psychotherapy: not allowed (exclusion criteria)
Concomitant pharmacotherapy: not stated No‐treatment Comparison name: No pill group (no treatment) + high interpersonal contact Description of intervention: “(…) a no pill group (a control for placebo effect), which received regular monthly visits from a psychiatrist and the same battery of rating scales as the other groups, but which did not receive any medication or pills.” (p. 187) Exposure/intensity to treatment: no pharmacological treatment. High contact group: met with a social work for a minimum of one hour a week Duration treatment: 8 months Concomitant psychotherapy: not allowed (exclusion criteria) Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”In the maintenance phase of our project, patients were assigned to treatment in a six‐cell design in a double‐blind controlled manner (…)” (p. 186) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | Quote: “Assessment ratings were carried out during a 1‐ to 1,5‐hour semi‐structured interview with the patient, by two Bachelor Degree‐level research assistants not involved in the treatment.” ( Weissman 1974 , p. 773) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Quote: “Patients terminated prior to month 9 for reasons other than relapse were counted for each completed month but for only one‐half of the month of termination.” Attrition <15% (7.3%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Klerman 1974b.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms Low interpersonal contact:
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 8 months. Duration of participation (trial + follow‐up): 8 months (no follow‐up) Setting : outpatients Purpose of trial: to evaluate maintenance treatment of depression including feasibility, efficacy and safety. Closed or open placebo: closed placebo |
|
| Data |
Number of participants screened: 278 Number of participants included: 150 Number of participants followed‐up at post treatment: 139 (completed or relapsed) Number of participants randomly assigned to:
Number of withdrawals : n = 5
Diagnosis: depression Diagnostic manual : Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition (DSM‐III) Means of assessment : Brief Psychiatric Rating Scale (BPRS) Comorbidity: ”85 percent of our patients were diagnosed as having neurotic depressions, according to criteria from the American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders” p. 187. Age: 39 mean years IQ: not stated Sex: 100% female Ethnicity:: 83.3% white Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: Placebo group + low interpersonal contact
Description of intervention: no description of pharmacological placebo The low contact group "saw the project psychiatrist for 15 minutes once a month for the completion of the rating scale, management of drug doses, and questions about possible side‐effects.” (p. 187) Individual or group treatment: individual Exposure/intensity to treatment: exposure to pharmacological placebo is not stated. low contact group: met with a project psychiatrist for 15 minutes once a month Duration of treatment: 8 months. Concomitant psychotherapy: not allowed (exclusion criteria) Concomitant pharmacotherapy: not stated No‐treatment Comparison name:No pill group (no treatment) + low interpersonal contact Description of intervention: “(…) a no pill group (a control for placebo effect), which received regular monthly visits from a psychiatrist and the same battery of rating scales as the other groups, but which did not receive any medication or pills.” (p. 187) Exposure/intensity to treatment: no pharmacological treatment. low contact group: met with a project psychiatrist for 15 minutes once a month Duration treatment: 8 months Concomitant psychotherapy: not allowed (exclusion criteria) Concomitant pharmacotherapy: not stated |
|
| Outcomes | See Klerman 1974a | |
| Notes | See Klerman 1974a | |
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”In the maintenance phase of our project, patients were assigned to treatment in a six‐cell design in a double‐blind controlled manner (…)” (p. 186) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | Quote: “Assessment ratings were carried out during a 1‐ to 1,5‐hour semi‐structured interview with the patient, by two Bachelor Degree‐level research assistants not involved in the treatment.” ( Weissman 1974 , p. 773) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Quote: “Patients terminated prior to month 9 for reasons other than relapse were counted for each completed month but for only one‐half of the month of termination.” Attrition <15% (7,3%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Klosko 1990.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 15 weeks. Duration of participation (trial + follow‐up): 15 weeks (no follow‐up) Setting: outpatient Purpose of trial: “The purpose of this study is to evaluate the relative effectiveness of each treatment in one setting using a group of clients diagnosed in an identical manner with outcome measured in precisely the same way (…) This study was seen as a precursor to studying possible integration or coordination of these treatments in panic disorder patients.” (p. 78) Closed or open placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included:: 69 Number of participants followed‐up at post treatment: 57 Number of participants randomly assigned to:
Number of withdrawals: n = 12
Diagnosis: Panic Disorder (with clinician’s severity rating of at least 4 on a 0‐8 scale). Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition (DSM‐III) Means of assessment: Anxiety Disorders Interview Schedule‐Revised (ADIS‐R). Comorbidity: not stated Age: 37 mean years (SD = 11.04) (Range: 18 to 65) IQ: not stated Sex: 74% female Ethnicity: “One dropout from the placebo group was Black; all other subjects were White.” (p. 80) Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: Placebo
Description of intervention: “Subjects received 15 individual treatment sessions in weekly meeting with a study psychiatrist experienced in alprazolam treatment of panic disorder. Medication was supplied by the Upjohn Company in matching 1‐mg tablets, packaged in matching bottles containing sufficient medication for 1 week (…) Medication was gradually increased following a standardized but flexible schedule until maximum benefit was achieved or dose‐limiting side effects occurred (…) The psychiatrist was instructed to limit interactions with subjects to discussion of clinical history, explanation of panic disorder, discussion of medication effects and side effects, and general support.” (p. 78‐9) Individual or group treatment : individual Exposure/intensity to treatment: not stated, but gradually increased dose to a maximum of 10 mg per day if required and at least three attempts made to titrate the medication to at least 6 mg per day. Duration of treatment: 15 weeks. Concomitant psychotherapy: “subjects who had been either in drug or psychotherapeutic treatment more than 6 months were excluded unless they agreed to stop such treatment for the duration of the study.” (p. 78) Concomitant pharmacotherapy: no – “Subjects in the three treatment groups withdrew from prestudy medications under the supervision of the study psychiatrist. Adherence to drug withdrawal was determined by analyses of plasma benzodiazepine screens.” (p. 78) Wait‐list Comparison name : Waiting list Description of intervention: “Subjects were placed on a 15‐week waiting list for treatment. They were told that they might contact the clinic by telephone during this time if they felt the need and that we would contact them approximately weekly by telephone.” (p. 79) Exposure/intensity to treatment: no treatment Duration treatment: 15 weeks Concomitant psychotherapy: “subjects who had been either in drug or psychotherapeutic treatment more than 6 months were excluded unless they agreed to stop such treatment for the duration of the study.” (p. 78) Concomitant pharmacotherapy: yes – ”Although inclusion criteria required all subjects to have been stabilized on medication, waiting‐list subjects were not required to withdraw from medication”. Therefore, this waiting‐list group might also be considered a minimal treatment condition, thereby providing a more conservative comparison with other treatment conditions.” (p. 79) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”Subjects remained off medication for at least 7 days before administration of psychophysiological, self‐report, and self‐monitoring measures and random assignment to one of the three treatment groups.” (p. 78) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: Patients“Posttreatment clinical assessment measures were gathered through administration of a short form of the ADIS‐R. The ADIS‐R administrators were blind to group assignment.” (p. 81) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Quote: ”A higher rate of dropout was observed in the placebo group compared with the other three groups.” “In any case, if one considers only completers, then 45% of placebo completers achieved high end state functioning posttreatment; but if one includes dropouts, only 28% of placebo subjects achieved high end state functioning.” (p. 84) Attrition >15% (17,4%). Only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Krapfl 1970.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 5 weeks (5 treatment sessions about a week apart) Duration of participation (trial + follow‐up): 5 weeks + 6 weeks follow‐up Setting: outpatient (University setting) Purpose of trial: “The present experiment attempted to examine whether or not SD would be successful if the aversive imagery were presented in random or decreasingly aversive hierarchical order.” (p. 333) Open/Closed: closed placebo |
|
| Data |
Number of participants screened: 1200 Number of participants included : 50 Number of participants followed‐up at post treatment: not stated Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis::specific anxiety (Snake phobia) Diagnostic manual: not stated Means of assessment:: exposure of snake Comorbidity: not stated Age:not stated IQ: not stated. College undergraduates Sex: 100% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Pseudodesensitisation Description of intervention: ”The imaginal stimuli used in treating this group were descriptions of 20 different snake‐irrelevant and pleasant landscape scenes. This group was included to control nonspecific factors, such as expectancy and commitment on the part of 5, relationship, attention, and suggestion.” (p. 334) Individual or group treatment: not stated Exposure/intensity to treatment: 30 minutes relaxation training and five sessions/week Duration of treatment: 5 weeks Concomitant psychotherapy: exclusion criteria not undergoing psychiatric treatment. Otherwise not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: No‐treatment Description of intervention: ”These 5s received only the pretreatment, posttreatment, and follow‐up test batteries which were administered to the other four groups” (p. 334) Exposure/intensity to treatment: none Duration treatment : 5 weeks Concomitant psychotherapy: exclusion criteria not undergoing psychiatric treatment. Otherwise not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Assignment of a matched 5 to one of the five groups was then done on a random basis. (p. 334) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Kwan 2017.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 3 weeks Duration of participation (trial + follow‐up): 3 weeks + 6 weeks follow‐up Setting: outpatient. Nursing Home Purpose of trial: “(…) the purpose of the present study is to examine the effect of acupressure on agitation and salivary cortisol by testing the following null hypotheses: (1) there is no difference in the level of agitation between the acupressure group and the control groups among nursing home residents with dementia over time; and (2) there is no significant difference in the salivary cortisol level between the acupressure group and the control groups among agitated nursing home residents with dementia over time.” (p. 93) Closed or open placebo: closed placebo |
|
| Data |
Number of participants screened: 2014
Number of participants included: 121
Number of participants followed‐up at post treatment: 118 Number of participants randomly assigned to:
Number of withdrawals : n = 3
Diagnosis: dementia Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision (DSM IV‐4‐TR) ( Kwan 2017 (pers comm) ) Means of assessment: not stated Comorbidity: number of chronic illness 4.1 (SD = 1.9) in the total population. Otherwise not stated Age: 86.5 mean years (SD = 6.3) IQ: not stated Sex:: 78% female Ethnicity: not stated Country: China Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo
Treatment name : Sham group + usual care
Description of intervention: “The sham protocol was identical to the acupressure protocol, except for the points on which pressure was applied. (…) They applied pressure on nonacupoints that were in an adequate distance from the acupoints. (…) These were located on (1) the nasal bone, (2) the olecranon, (3) the styloid process of the ulna, (4) the medial malleolus over the ankle, and (5) the head of the fibula.” (p. 95)
Individual or group treatment: individual
Exposure/intensity to treatment two 10‐minute acupressure sessions a day (morning session at 08.00‐12.00 and an afternoon session at 14.00‐18.00), 5 days per week
Duration of treatment: 2 weeks
Concomitant psychotherapy: received usual care provided by the RCH to manage agitated residents every day (not specified)
Concomitant pharmacotherapy: received usual care provided by the RCH to manage agitated residents every day (not specified). Number of psychotropic drugs used in the whole sample: 1.5 (SD =1.1) Wait‐list Comparison name: usual‐care group (in reality wait‐list) Description of intervention: “The participants in this group received only the usual care provided by the RCH to manage agitated residents every day, such as activity programs and the use of restraints if needed as judged by the nursing home staff. Such care was also provided to the participants in the acupressure and sham group.” (p 95) "In this group, participants receive no acupressure‐ related intervention. They receive a free course of acupressure sessions, identical to the one stipulated in the AG, after completing the study." ( Kwan 2014 , p. 5) Exposure/intensity to treatment: no treatment but the usual care Duration treatment: 2 weeks Concomitant psychotherapy: received usual care provided by the RCH to manage agitated residents every day (not specified) Concomitant pharmacotherapy: received usual care provided by the RCH to manage agitated residents every day (not specified). Number of psychotropic drugs used in the whole sample: 1.5 (SD =1.1) |
|
| Outcomes |
Beneficial effects
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: ”The participants were allocated by permuted block randomization to 3 parallel groups in a 1:1:1 ratio (...) The permuted block randomization list was generated by the web‐based generator at Randomization.com.” (p. 93) |
| Allocation concealment | Yes | Quote:”An independent research assistant, who did not participate in any other parts of the research and was blinded to participants’ demographics and clinical characteristics, allocated participants to groups according to the randomization list and subject codes provided by the data collectors.” (p. 93) |
| Blinding of outcome assessors | Yes | Quote:“Participants, RCH staff members, and data collectors who were not involved in providing any care services in the participating RCHs were blind to the group labels.”) (p. 93) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Quote:“Data analysis was performed using SPSS version 21.0 based on a modified intention‐to‐treat (mITT) principle, which means that all subjects were included after randomization except for those who withdrew from the study before undergoing the first session of the intervention. A generalized estimating equation (GEE) was used to answer the 2 hypotheses.(…) Participants with missing data points were included in the mITT as estimated by the GEE by following the missing‐at‐random assumption.” (p. 96) Attrition <15% (2.5%) |
| Selective outcome reported | Yes | CUHK_CCT003347 No apparent differences in reporting between trail registry and full report |
| Other sources of bias | Yes | No other sources found |
Lacy 1990.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial: 5 months in total Duration of participation (trial + follow‐up): 5 months. Post‐treatment data Setting: inpatient (hospital) Purpose of trial: “The purpose of the current investigation was to determine the effectiveness of short‐term autogenic training as a stress management intervention with hospitalized emotionally disturbed adolescents” (p. 96) Open/Closed placebo: closed placebo |
|
| Data |
Number of participants screened: 58 Number of participants included: 45 Number of participants followed‐up at post treatment: 45 Number of participants randomly assigned to:
Number of withdrawals: n = 0 Diagnosis: several psychiatric disorders. Diagnostic manual: not stated Means of assessment: Clinical interview (Global Assessment of Functioning; GAF): ranged from 28 to 55 years, average 37 Comorbidity: ” Adolescents admitted to the psychiatric hospital during the investigation exhibited various forms of dysfunctional behavior and were diagnosed with diverse psychiatric classifications. Many were determined to be a danger to self or others” (p. 59) “All subjects in the investigation were diagnosed with moderate to severe emotional problems; however, a number of subjects rated their anxiety levels quite low, below expected levels generally associated with normal adolescence.” (p. 102) Age:: 15.22 mean years (SD = 1.38). (range= 13 to 17) IQ: “The mean composite intelligence score of subjects was 95, falling within the average range 63‐125” (p. 61). Sex: 46.7% female Ethnicity: "Thirty‐eight subjects participating in the investigation were white. Three Native Americans, three Blacks (African Americans) and one Asian American also served as subjects" (p. 61) Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo
Treatment name:: a Self‐relaxation placebo control group
Description of intervention: “Subjects practicing self‐relaxation assumed a similar horizontal posture under the same experimental conditions as the autogenic trainees.” (p. 73)
Individual/group : Individual treatment
Exposure/intensity to treatment: six sessions were conducted over a two‐day period following the same time frame as the autogenic training sessions. Subjects practiced self‐relaxation for no longer than 10 minutes in one session
Duration of treatment: 5 months
Concomitant psychotherapy: “Treatment received prior to admission to the hospital varied for each subject. Most subjects had been involved in some level of counseling. Some subjects had been previously hospitalized for treatment.” (p. 59)
Concomitant pharmacotherapy: “Several subjects had histories of medication intervention. None of the subjects in the investigation were prescribed psychoactive medication during the evaluation period. Also, there were no controls for residual effects of prior medication.” (p. 59) Wait‐list Comparison name: No‐treatment (in reality wait‐list) Description of intervention: “Subjects in this control group participated in only pre‐ and posttesting measurement activity. No relaxation training was provided in the interim. Subjects were informed relaxation training was to begin with the completion of two measurement sessions. Control group subjects received brief instruction in autogenic training with the completion of posttesting. (p. 73) Exposure/intensity to treatment: no treatment during waiting Duration treatment: 5 months Concomitant psychotherapy: “Treatment received prior to admission to the hospital varied for each subject. Most subjects had been involved in some level of counseling. Some subjects had been previously hospitalized for treatment.” (p. 59) Concomitant pharmacotherapy: “Several subjects had histories of medication intervention. None of the subjects in the investigation were prescribed psychoactive medication during the evaluation period. Also, there were no controls for residual effects of prior medication.” (p. 59) |
|
| Outcomes |
Beneficial effects
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: A total of 45 adolescents were randomly assigned to participate as subjects in one of the three experimental conditions." (p. 62) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | Quote: ”No other assistance in completing the scale was provided. The scale was not scored by the investigator until the completion of posttesting.” (p. 65) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Quote: ”Attrition was not a problem as all 45 subjects complied with all treatment and measurement demands.” (p. 62) Attrition < 15% (0%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Lai 2004.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 6 weeks Duration of participation (trial + follow‐up): 6 weeks. No follow‐up Setting: inpatient (nursing homes) Purpose of trial: “This study is a randomized controlled trial aimed at finding out whether a specific reminiscence program would lead to any changes in social well‐being for nursing home residents with dementia.” (p. 34) Open/closed placebo:: closed placebo |
|
| Data |
Number of participants screened: 127 Number of participants included: 101 Number of participants followed‐up at post treatment: 89 Number of participants randomly assigned to
Number of withdrawals : n =12
Diagnosis: dementia Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: ”The physicians in charge of the homes confirmed that those residents diagnosed as having dementia met the criteria as specified by the DSM‐IV.” (p. 38) Comorbidity: number of other diagnoses: psychological placebo: 3.7 (SD=1.7). No‐treatment 4.5 (SD = 1.9). Not specified which Age: 85.45 mean years (SD = 7.35) IQ:: not stated, but years of education included Sex:: 77.5% female Ethnicity: not stated Country: China/USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name : Comparison group Description of intervention: “To control for the possibility that a resident’s improvement might have been the result of the attention and social contacts resulting from the intervention itself, the comparison program was designed to provide social contacts.” (p. 35). Individual or group treatment:: individual Exposure/intensity to treatment: a 30‐minute session weekly Duration of treatment: 6 weeks Concomitant psychotherapy: both groups receive their regular group sessions (Nursing home resident). It consisted of exercise, card games etc. Concomitant pharmacotherapy: not stated but regular medicine No‐treatment Comparison name: Control group (No treatment) Description of intervention: “Subjects assigned to the control group received no intervention.” (p. 36) Exposure/intensity to treatment: no intervention Duration treatment:: 6 weeks Concomitant psychotherapy: both groups received their regular group sessions (Nursing home resident). It consisted of exercise, card games etc. Concomitant pharmacotherapy: not stated, but regular medicine |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | No | Quote: ”It was also unclear whether a random assignment was used in group allocation” (p. 46) |
| Allocation concealment | Unclear | Concealed from authors Chi & Kayser‐Jones, but not Lai |
| Blinding of outcome assessors | Yes | Quote: ”The group of RAs who collected data on the participants included both raters (who rated only the WIB of the DCM) and assessors (who performed the rest of the assessments), and both groups were blinded to subject assignment.” (p. 37) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Quote: ” In the ITT sample, (Table 1) the percentage of data missing for the outcome variables of T0, T1, and T2 was 0.5%, whereas the missing data for two controlling variables, the MMSE and the MDS‐ADL, was 0.9%. Missing data for all other variables constituted 1.1%. In total, 2.5% of the data was missing in this dataset. For the per protocol sample, the percentages of missing data for the outcome variables was 0.2%, the MMSE and the MDS‐ADL–0.5%, and all other variables –0.8%. The total percentage of missing data for the per protocol sample was 1.5%. The mean value of the outcome variables for each respective group was used as a replacement for the missing data.” (p. 39) Attrition <15% /(2.5%). ITT used |
| Selective outcome reported | Unclear | Not registered beforehand ( Lai 2004 (pers comm) ) |
| Other sources of bias | Unclear | Quote: ”Concerning the per protocol population, significant differences were found between the control and comparison group in the number of medical diagnoses other than dementia and in whether they had any regular programs. It was difficult to explain the meaning of the differences, as these two groups were not significantly different in terms of their baseline (T0) and T1, C‐MMSE and MDS‐ADL scores.” (p. 45). |
Lang 1965.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 8‐16 weeks Duration of participation (trial + follow‐up): 8‐16 weeks Settin: : outpatient (University setting) Purpose of trial: “44 snake phobic Ss participated in laboratory experiments assessing the degree of fear change associated with systematic desensitization, no treatment, placebo treatment, and the trait of suggestibility” (p. 395) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 44 Number of participants followed‐up at post treatment: 44 Number of participants randomly assigned to:
Number of withdrawals: n = 0 Diagnosis: specific anxiety (snake phobia) Diagnostic manual: not stated Means of assessment: “They rated their fear of nonpoisonous snakes as "intense," on a fear questionnaire, and were included in this research only if a psychological interview corroborated this statement.” (p. 397) Comorbidity: not stated Age:: not stated – Introductory psychology students IQ:: not stated, Introductory psychology students Sex:: not stated Ethnicity:: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Pseudotherapy Description of intervention: “An effort was made to involve the subject in a treatment procedure, which was therapeutically neutral except for the therapist‐client relationship. Because desensitization was to be evaluated, all procedures employed in that method were included in pseudotherapy.” (p. 396) Individual or group treatment: Individual Exposure/intensity to treatment: the participants first experienced the same 5 training sessions as in desensitisation, followed by 11 pseudotherapy sessions. Duration of treatment : 16 sessions (8‐16 weeks) “Sessions lasted approximately 45 minutes at the rate of 1 or 2 per week” (p. 396) Concomitant psychotherapy : None of the participants in this study were being seen elsewhere because of psychological problems. Concomitant pharmacotherapy: not stated No‐treatment Comparison name:: No‐treatment Description of intervention: “Untreated subjects were not seen, except for evaluation sessions.” (p. 397) Exposure/intensity to treatment: no treatment Duration treatment:: not stated Concomitant psychotherapy: none of the participants in this study were being seen elsewhere because of psychological problems Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”Assignment to groups was essentially random, although some pretreatment effort to balance control variables was made. A more elaborate description of the selection battery has already been reported (Lang 1963).” (p. 397) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Attrition <15% (0%) |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Legrand 2016.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no. Duration of trial (baseline to post): 10 days. Duration of participation (trial + follow‐up): 10 days (no follow‐up) Setting: inpatient. Purpose of trial: “(…) in the present study we examined the efficacy of a 10‐days long aerobic exercise program as an add‐on treatment in severely depressed patients being treated with antidepressant medication (and no other form of therapy) for less than two weeks.” (p. 140) Closed/open placebo:: closed placebo |
|
| Data |
Number of participants screened: 124
Number of participants included: 35
Number of participants followed‐up at post treatment: 31 Number of participants randomly assigned to:
Number of withdrawals : n = 4
Diagnosis: Major depressive disorder (MDD) Diagnostic Manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision (DSM‐IV‐TR) Means of assessment: “Patients were assessed using the Beck Depression Inventory (BDI‐II)” ( Legrand 2016 (pers comm) ) Comorbidity : not stated Age:: 45.3 mean years (SD = 13.2) IQ: not stated Sex: 71.4% female Ethnicity: not stated Country:: France Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo
Treatment name: Placebo Stretching exercise
Description of intervention: “Patients in the stretching (ST) group also performed a daily 30 min exercise program for 10 consecutive days, but this consisted of stretching exercises instead of endurance training. Several muscle groups (thighs, calves, gluteal, shoulders, back) were stretched for 60 s. with equivalent resting intervals between stretching series. Training sessions were carried out in a room of the hospital restricted to these activities and were also supervised by the first author.” (p. 140‐2) Individual or group treatment: “(…) the format of delivery was mostly individual (only 4 of the 85 stretching sessions included 2 patients).” (p. 141) Exposure/intensity to treatment: 30 minutes daily Duration of treatment: 10 days Concomitant psychotherapy: no Concomitant pharmacotherapy: antidepressant medication No‐treatment Comparison name: No‐intervention (no treatment). Description of intervention: “(…) participants in the control (NI) group received no intervention other than the prescribed medication.” (p. 141) Exposure/intensity to treatment : no treatment other than the prescribed medication Duration treatment: 10 days Concomitant psychotherapy: no Concomitant pharmacotherapy: antidepressant medication |
|
| Outcomes |
Beneficial effects
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: ”One of the three study arms (aerobic exercise, stretching, no intervention) was randomly chosen for each participant at the end of an initial individual visit (…) This was done by running the rand between function of Microsoft Excel on our laptop, which generated a random number between 1 and 3: 1=aerobic exercise (AE), 2=stretching (ST), 3=no intervention.” (p. 140) |
| Allocation concealment | Yes | Quote: “Yes..... A can be understood from our text (p. 140, right‐hand column, 3rd paragraph) we did not know whether the next study patient would receive Aerobic Exercise, Stretching (sham), or No Intervention;...Consequently, it is possible to state that our random allocation procedure was concealed.” ( Legrand 2016 (pers comm) ) |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Quote: ”These analyses were performed using an intent‐to‐treat (ITT) approach in which all patients with baseline measures were included in analyses, even if they missed more than two training sessions. Missing data were imputed using the average change in depression score from baseline to post‐intervention in the control group. When drop‐out rates are less than 20% (which is the case in our study), this method keeps statistical power at higher levels compared to the last‐observation‐carried‐forward method.” (p. 141) Attrition <15% (11.5%) |
| Selective outcome reported | Yes |
NCT02612142 No apparent differences in reporting between trial registry and full report |
| Other sources of bias | Yes | No other sources found |
Lick 1975.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial: 6 weeks Duration of participation (trial + follow‐up): 6 weeks. 4‐week follow‐up, and 4‐month follow‐up Setting: outpatient Purpose of trial: “The investigation reported here compares two versions of (...) placebo manipulation with Wolpian systematic desensitization in the modification of snake and spider fear in volunteer adult subjects who manifest real‐life inhibitions as a result of their fear.” (p. 558) Open/Closed placebo : closed placebo |
|
| Data |
Number of participants screened: 48 Number of participants included: 36 Number of participants followed‐up at post treatment: post‐treatment not stated. (31 after 4 months) Number of participants randomly assigned to:
Number of withdrawals: post‐treatment: not stated, (5 in follow‐up) Diagnosis: specific anxiety (snakes & spiders) Diagnostic manual: not stated Means of assessment:: clinical interview Comorbidity: not stated Age: 29.77 mean years (range =18 to 59) IQ: not stated Sex: 100% females Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo
Treatment name: Placebo
Description of intervention: "This treatment was exactly the same as placebo feedback described above except that (a) the subject was not shown her GSR printouts, nor did the therapist comment about them at any point in treatment. If the subject asked about her GSR printouts, the therapist responded that they were not interpretable until analyzed by the computer, (b) The subject was also told that
the equipment reduced the number of shocks it delivered on each session automatically and independently of the subject's responses” (p. 559‐560)
Individual or group treatment: individual
Exposure/intensity to treatment: 20 minutes per session (except first one)
Duration of treatment: 8 biweekly sessions for 6 weeks
Concomitant psychotherapy: not stated
Concomitant pharmacotherapy: not stated Wait‐list Comparison name: No‐treatment (in reality wait‐list) Description of intervention: “The subjects in this group were sent a letter stating that because of staff shortages they could not be treated at this time but that they would receive treatment in several months when therapist time became available.” (p. 560) Exposure/intensity to treatment: no treatment during the period Duration treatment: 6 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effects
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: “(…) the subjects were stratified into three blocks of four snake phobics and six blocks of four spider phobics. One subject from each block was then randomly assigned to each of the four conditions, and the three therapists were randomly assigned one snake phobic and two spider phobics from each condition.” (p. 559) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:“The pretreatment Behavioral Approach Test was administered by the author; the posttreatment Behavioral Approach Test was administered by another male experimenter who was blind about the conditions to which subjects had been assigned”. (p. 560) |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | Unclear | Attrition unclear. 5 were lost to follow‐up. Unclear how many that were lost to post‐treatment. |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | No | Author (J.L.) was a therapist himself in the study |
Lick 1977.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 6 weeks Duration of participation (trial + follow‐up): 6 weeks of treatment + 1 month follow‐up Setting : Outpatient. University setting Purpose of trial: “The present study is designed to further evaluate the relative efficacy of relaxation training and attention placebo procedures in the treatment of severe insomnia.” (p. 154) “Finally, this study assesses whether giving people a tape with relaxation instructions to play in bed before retiring adds to the efficacy of relaxation training conducted in the clinic.” (p. 154) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 65
Number of participants included: 40 Number of participants followed‐up at post treatment: 35 Number of participants randomly assigned to :
Number of withdrawals: 5 – “Subjects approximately equivalent in age, sex, and time to fall asleep were substituted for these latter subjects.” (p. 155)
Diagnosis: Insomnia Diagnostic manual : none Means of assessment: averaging 50 minutes or longer to fall asleep during a 20‐day pre‐tratment baseline period Comorbidity: not stated Age: 47.48 mean years (SD = 10.88), (range = 29 to 72 years) IQ: not stated Sex: 65% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo
Treatment name: Placebo
Description of intervention: “The placebo procedures are a variant of those found credible and effective in the treatment of phobic behavior and were expected to work only because of placebo and other nonspecific factors.” (p. 154). “This procedure was a modified form of “T‐scope therapy”. Subjects were told that this procedure was designed to reduce autonomic arousal that was incompatible with sleep. Subjects were told that their autonomic arousal would be monitored with a polygraph and that whenever the polygraph detected a “high‐intensity autonomic response,” it would trigger a shock generator that would deliver a mildly unpleasant shock to the subject’s left index finger (…) Subjects received a decreasing number of shocks over the six sessions (…) Over the six sessions these printouts showed less and less autonomic activity (…)” (p. 155)
Individual or group treatment: individual.
Exposure/intensity to treatment: six sessions
Duration of treatment: 6 weeks.
Concomitant psychotherapy: not stated
Concomitant pharmacotherapy: participants were allowed to take sleep‐inducing drugs No‐treatment Comparison name: No‐treatment Description of intervention: no treatment – no other description of intervention. Exposure/intensity to treatment: no treatment. Duration treatment: 6 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: participants were allowed to take sleep‐inducing drug |
|
| Outcomes |
Beneficial effects
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”Within each wave, subjects were blocked according to sex, time to fall asleep, and whether they took sleeping medication. They were then randomly assigned to the following conditions: (a) progressive relaxation; (b) progressive relaxation plus taped relaxation; (c) placebo control; or (d) no‐treatment control. (p. 155) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Quote: "(…) 5 subjects dropped out of treatment; 2 dropped out because they repeatedly missed appointments, and 3 dropped out because of severe emotional or physical difficulties unrelated to treatment. Subjects approximately equivalent in age, sex, and time to fall asleep were substituted for these latter subjects.” (p. 155) Attrition <15% (12.5%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Liddle 1990.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial: 8 weeks Duration of participation (trial + follow‐up): 8 weeks + 5 week follow‐up Setting: Outpatient Purpose of trial: “The present study differs from other published studies cited in combining both a follow‐up assessment and a non‐specific treatment control group which will make it possible to determine if any beneficial effects are specific to the treatment.” (p. 88) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 380 Number of participants included:: 33 Number of participants followed‐up at post treatment: 31 Number of participants randomly assigned to: 31
Number of withdrawals: n = 2
Diagnosis: depression Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition (DSM‐III) Means of assessment: structured clinical interview Comorbidity: not stated Age:: 9.2 mean years (SD =1.15) IQ: not stated, but intellectually handicapped were excluded Sex:: 36.4% female Ethnicity: not stated Country:: Australia Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: attention placebo Description of intervention: “The APC group consisted of a drama programme which was adapted by the experimenter from one devised by Milneand Spence (1987) from educational literature on the teaching of drama to primary school children (...). Drama was selected for the APC because the following elements were shared with the social competence condition: withdrawal from class during school time; small group interaction;an equal amount of time and attention and homework assignments. Nospecific skill‐based instruction regarding the perception of interpersonal cues or methods of dealing with interpersonal situations was given. There is no evidence that drama sessions per se should improve social competence” (p. 93) Individual or group treatment: group Exposure/intensity to treatment:: eight, weekly, one‐hour group sessions Duration of treatment: 8 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name : No‐treatment Description of intervention: “Children in this condition did not participate in any "special activities". They attended regular school classes and were withdrawn for the pre‐, post‐ and follow‐up assessments only, on the pretext that they were helping the University with research into "how children feel about things".” (p. 94) Exposure/intensity to treatment: no treatment Duration treatment: 8 weeks of no treatment Concomitant psychotherapy: not stated Concomitant pharmacotherapy:: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: "The children were then assigned (it is unclear if this was random) to one of four conditions, namely role‐play to train interpersonal problem solving skills, cognitive restructuring, attention—placebo and classroom control. " (p. 86) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Attrition <15% (0% during treatment) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Matson 1980.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 6 weeks Duration of participation (trial + follow‐up): 6 weeks + 8 weeks (14 weeks) follow‐up Setting : outpatient Purpose of trial: “The present study was designed to compare variants of self‐control, self‐evaluation, and self‐reinforcement (independence training) to both a conventional treatment and a no‐treatment control group. (p. 488) |
|
| Data |
Number of participants screened: 164 Number of participants included: 75 Number of participants followed‐up at post treatment: 75 Number of participants randomly assigned to:
Number of withdrawals : n = 0 Diagnosis: mental retardation Diagnostic manual: not stated Means of assessment: not stated, but range of retardation was based on administration of Stanford‐Binet Intelligence Scale, Form L‐M, and the American Association on Mental Deficiency Adaptive Behavior Scale Comorbidity: not stated Age: 33.9 mean years (range= 22 to 57) IQ: not stated, but participants were mentally retarded. Moderate to profound range Sex: not stated Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Usual care Treatment name: standard‐treatment group Description of intervention: "The standard treatment consisted of procedures of choice for training of self‐help skills to retarded persons, determined by the frequency of cited research articles in the training of self‐help behavior. Training methods included verbal prompts, modeling, manual guidance, social reinforcement, shaping, fading, and chaining.” (p. 491) Individual or group treatment: individual Exposure/intensity to treatment: five training session weekly. “Each subject received five training sessions over a one‐week period.” (p. 491) Duration of treatment: 6 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: No‐treatment control group Description of intervention: “Target behaviors for these subjects was measures but not trained. The target behaviors were recorded at time periods in correspondence with the treatment groups without delivery of instructions, feedback, reinforcement, etc., specific to the behavior of being measured.” (p. 491) Exposure/intensity to treatment: no treatment Duration treatment: 6 weeks Concomitant psychotherapy: not stated, but possibly part of ambulatory treatment Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: "Twenty‐five subjects were randomly assigned to each of three conditions: no‐treatment control, standard treatment, and independence training.” (p. 491) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:“(…) and waiting until data collection had been completed before debriefing raters on the purpose of the study.” (p. 490) |
| Blinding of participants and personnel | No | Not possible to blind usual care and no‐treatment |
| Incomplete outcome data | Yes | Quote:“Group mean pre‐, post‐, and follow‐up scores for the 75 subjects who completed the experiment (25 in each group) are shown in Figure 1.” (p. 492) Attrition <15% (0%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
McLachlan 1991.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 24 months Duration of participation (trial + follow‐up): 24 months (no follow‐up) Setting: inpatient (nursing home) Purpose of trial: “We have completed a two year, singleblind study to investigate whether the progression of dementia could be slowed by the trivalent ion chelator, desferrioxamine.” (p. 1304) Closed/open placebo: closed placebo |
|
| Data |
Number of participants screened: 1510
Number of participants included:: 63
Number of participants followed‐up at post treatment: 41
Number of participants randomly assigned to:
Number of withdrawals: n = 22
Diagnosis: Alzheimer’s disease Diagnostic manual: not stated Means of assessment: brain tissue + cognitive tests Comorbidity: not stated Age: 63.1 mean years (SD = 6.3) IQ: 79.2 to ‐81.4 Sex: 39.7% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological Placebo
Treatment name: placebo
Description of intervention: oral lecithin
Individual or group treatment: Individual
Exposure/intensity to treatment: 500 mg lecithin twice daily,
Duration of treatment: 24 months
Concomitant psychotherapy: not stated – but lived in nursing home
Concomitant pharmacotherapy: not stated – but lived in nursing home No‐treatment Comparison name: No‐treatment Description of intervention: not stated Exposure/intensity to treatment: not stated Duration treatment: 24 months Concomitant psychotherapy: not stated – but lived in nursing home Concomitant pharmacotherapy: not stated – but lived in nursing home |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: ”Those eligible to enter the study were assigned to one of three groups from a table of random numbers:” (p. 1305) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: ”Test results collected by the behaviour evaluation team were not revealed to the medical management team; the behaviour evaluation team remained blind to the group to which participants were assigned until statistical analysis began.” (p. 1305) Quote: “Subsequent analysis was by trained raters who were not told about the nature of the study” (p. 1305) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Attrition >15% (23,8%). Only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Mealiea 1971.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 5 weeks Duration of participation (trial + follow‐up): 5 weeks + 4 weeks follow‐up Setting: outpatient (University setting) Purpose of trial: “Systematic desensitization (SD) and implosive therapy (IT) were compared for their effectiveness in modifying snake phobic behavior.” (p. 85) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 1200 Number of participants included: 50 Number of participants followed‐up at post treatment: 48 Number of participants randomly assigned to:
Number of withdrawals: n =1
Diagnosis: specific anxiety (snake phobia) Diagnostic manual: not stated Means of assessment: not stated Comorbidity: not stated Ag:: not stated IQ: not stated ‐ but university students Sex: 100% females Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Pseudotherapy Description of intervention: ”The PT group received treatment similar to that of the SD group except that relaxation was paired with 20 snake irrelevant scenes; i.e. relaxation was paired with descriptions of landscapes, beaches, clouds, etc. The PT group was included to control for possible placebo or expectancy effects.” (p. 88) Individual or group treatment: group Exposure/intensity to treatment: 5 sessions, 30 minutes each Duration of treatment:: 5 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: No‐treatment Description of intervention: “The Ss in the NT group participated only in the pre‐, post‐, and follow‐up treatment assessment sessions. The same programmed instructions regarding assessment procedure given the other groups were given the NT group." (p. 88) Exposure/intensity to treatment: no treatment Duration treatment: 5 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”The Ss were matched on the basis of both their BAT and FT scores and randomly assigned to one of five equal groups.” (p. 87) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Outcomes are objective |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Attrition < 15% (4%) |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources of bias found |
Milby 1980.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 3 months of treatment Duration of participation (trial + follow‐up): no follow‐up Setting: outpatient Purpose of trial: “This study assessed the efficacy of urine surveillance as an adjunct to outpatient psychotherapy.” (p. 994) |
|
| Data |
Number of participants screened: not stated Number of participants included: 29 Number of participants followed‐up at post treatment: 29 Number of participants randomly assigned to :
Number of withdrawals : n = 0 Diagnosis:substance use disorder Diagnostic manual: not stated Means of assessment: Menlo Park VA Drug Abuse Program Questionnaire, MMPI. Comorbidit: not stated ‐ previously addicted to narcotics or barbiturate‐like drugs Age: 25.8 mean years (range =16 to 54) IQ: not stated Sex: 34.5% females Ethnicity: 22 Whites, 7 Blacks Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Usual care Treatment name: group II ‐ therapy Description of intervention: “Group I received 3 months of outpatient individual or group therapy (…) Group II received the same treatment (…)” (p. 995) Individual or group treatment: individual and group therapy Exposure/intensity to treatment: not stated Duration of treatment: 3 months Concomitant psychotherapy: not stated. See exclusion criteria Concomitant pharmacotherapy: not stated Wait‐list Comparison name: No treatment (in reality a wait‐list) Description of intervention: “Group III was not seen for outpatient therapy or urine surveillance until after 3 months, thus serving as a control group.” (p. 996) Exposure/intensity to treatment : no treatment during waiting Duration treatment: 3 months Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse event
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”After the battery, patients were randomly assigned to one of three treatment groups.” (p. 995) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind usual care and wait‐list |
| Incomplete outcome data | Unclear | Quote: “Subjects were eliminated from the study if they: (1) attended less than 50% of the outpatient therapy sessions scheduled, (2) got more than 3 hours psychotherapy or other treatment elsewhere when they had been assigned to Group 3, and (3) failed to return for follow‐up testing (…) No subjects needed to be eliminated from the study.” (p. 996) Attrition unclear |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Miranda 1997.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 2.5 months Duration of participation (trial + follow‐up): 2.5 months ‐ unclear if followed up Setting: community outpatient Purpose of trial: This study investigates the efficiency of attributional retraining associated to a programme aimed at teaching problem‐solving strategies to students with learning disabilities (LD) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 173 Number of participants included: 41 Number of participants followed‐up at post treatment: 41 Number of participants randomly assigned to
Number of withdrawals: 0 Diagnosis: learning disabilities Diagnostic manual: not stated Means of assessment: arithmetic verbal task Comorbidity: not stated Age: mean range 10.1 to 11.9 IQ: within normality Sex: 47.6% female Ethnicity: not stated Country: Spain Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Placebo Description of intervention: “Carried out problem‐solving activities but without training in strategies.” Used the same materials as work in experimental group, but the instruction procedure consistent of independent practice of problems. Individual or group treatment: group Exposure/intensity to treatment: 3 weekly sessions lasting 50 minutes Duration of treatment: 2.5 months Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: No‐treatment Description of intervention: received no special training Exposure/intensity to treatment: no treatment Duration treatment: 2.5 months Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: “The selected subjects were randomly assigned to each of the four groups: (p. 37) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Attrition <15% (0%) |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | None other sources found |
Mitchell 2008.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 1 month Duration of participation (trial + follow‐up): 1 month (no follow‐up) Setting: outpatient Purpose of trial: “The current study investigated the effects of lens tinting by using a design which provided controls for various effects (e.g., Hawthorne, maturational, practice, and experimenter effects.” (p. 517) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 66
Number of participants included: 49
Number of participants followed‐up at post treatment: 49 N umber of participants randomly assigned to:
Number of withdrawals : n = 0 Diagnosis: visuoperceptual reading disabilities Diagnostic manual: not stated Means of assessment: Neale Analysis of Reading Ability Comorbidity: not stated Age: range 7 to 11 years IQ: not stated Sex: 28.6% female Ethnicity: not stated Country: South Africa Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo Treatment name: Placebo Description of intervention: “(…) the placebo group underwent the colour testing and received coloured filters in a colour complementary to that which was specified as the optimal colour for that child.” (p. 523) “All children who received no or placebo filters were supplied with correctly matched coloured filters.” (p. 524) Individual or group treatment: individual Exposure/intensity to treatment: “During this time the placebo and experimental groups wore their filters for all reading activities including homework (…)” (p. 523) Duration of treatment: one month Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: Control group (wait‐list). Description of intervention: “(…) the control group received no filters (…).” (p. 523) “(…) the control group received no special treatment.” (p. 523) “All children who received no or placebo filters were supplied with correctly matched coloured filters.” (p. 523) Exposure/intensity to treatment: no treatment Duration treatment: month Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”The researcher randomly placed the participants into three groups, as matching can create uncontrollable discrepancies.” (p. 524) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to placebo and no‐treatment |
| Incomplete outcome data | Yes | No mention of attrition rate or imputation methods, but it seems from table 2 that all participants is included in the analyses |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | No | Quote: “That group differences were seen on three of the six dependent measures may suggest problems with randomization.” (p. 529) |
Nandi 1976.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 28 days. Duration of participation (trial + follow‐up): not stated Setting: outpatient Purpose of trial: “To find an answer to the question, whether all of those who were labelled as depressives in this door‐to‐door study were treatable depressives as compared to those who sought treatment in clinics, a clinical trial with the following design was undertaken.” (p. 524) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 1078
Number of participants included: 41
Number of participants followed‐up at post treatment: 35
Number of participants randomly assigned to:
Number of withdrawals : n = 6
Diagnosis: depression Diagnostic manual: not stated (see other notes) Means of assessment: clinical interview. “The labelling of a case with an appropriate diagnosis was done on the basis of diagnostic criterion accepted for each diagnostic entity.” (p. 524) Comorbidity: “The patients were thoroughly examined and all were found to be free from any physical illness.” (p. 524) Age: not stated IQ: not stated Sex: not stated Ethnicity: not stated Country: India Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: Placebo
Description of intervention: “The second group, consisting of 10 patients, was given placebo and will be called the Placebo group. The placebo used was lactose in the form of tablets, which were administered in exactly the same manner as the drugs prescribed in the Medicine group” (p. 524)
“One 25 mg tablet twice daily for 2 days, then two tablets twice daily to continue for the remaining days; time of administration being morning and afternoon after some meal.” (p. 524)
Individual or group treatment: individual
Exposure/intensity to treatment: one 25 mg tablet twice daily for 2 days, then two tablets daily the remaining 26 days
Duration of treatment: 28 days
Concomitant psychotherapy: not stated
Concomitant pharmacotherapy: not stated No‐treatment Comparison name : Natural Process (no treatment) Description of intervention: “The third group consisting of 11 patients was given no treatment (and was left to nature for any change that might be observed in them after a given interval of time.” (p. 524) Exposure/intensity to treatment: no treatment Duration treatment: 28 days. Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”They were randomly divided into four groups. Three of these groups had 10 patients each and the fourth group had 11 patients. Two of them were randomly chosen and merged together which consisted of 20 patients” (p. 524) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:“The placebo tablets and the drugs used in the Medicine group did not look alike, but this dissimilarity was not a source of bias, as neither the patients nor the raters knew who were given what.” (p. 524) Quote:"All the assessments were made by two raters and inter‐rater agreement was found to be high. Both the raters worked independently, and the mean of their ratings was taken in each case.” (p. 524) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Attrition rate >15% (15% in medicine. 0% in Placebo, 27% in Natural process. Only reports data on completers. No mention of ITT |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Nicassio 1974.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 4 weeks Duration of participation (trial + follow‐up): 4 + 6 months follow‐up Setting : Outpatient Purpose of trial : “the objective of the present study is to compare and evaluate two direct, short‐term treatments for insomnia.” (p. 253) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 60
Number of participants included: 32
Number of participants followed‐up at post treatment: 30 Number of participants randomly assigned to :
Number of withdrawals : n = 2
Diagnosis: sleep‐wake disorder (insomnia) Diagnostic manual: not stated Means of assessment: daily sleep records (not otherwise specified) Comorbidity: not stated Age: 45.1 mean years (SD = 14.57 (range: 22 to 71 years) IQ: not stated Sex: 70% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo
Treatment name : Self‐relaxation control
Description of intervention: “(…) subjects received four one‐hour individual treatment sessions and a posttreatment session at which final assessments were made.” (p. 255) “Although subjects were seen for the same number of treatment sessions as those receiving bona fide relaxation instruction, no technique of relaxation was taught. Subjects were told that everyone knows how to relax; it is just a matter of scheduling time to do so.” (p. 255) Individual or group treatment: individual treatment. Exposure/intensity to treatment: “As in the other treatment groups, subjects were instructed to relax for one 20‐ to 30‐minute session at home during the day and again in bed while preparing for sleep.” (p. 255) Duration of treatment: 4 weeks. Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “An additional requirement was that all subjects stop taking sleeping pills or other forms of medication with soporific effects after obtaining the permission of their physicians.” (p. 254) Wait‐list Comparison name: No treatment control (wait‐list) Description of intervention: “Subjects in this group were seen at the beginning of the treatment period and were told that an extended baseline measurement period would be needed to accurately assess the nature and stability of their sleeping difficulties. For the next four weeks, subjects mailed in data and were not seen again until the posttreatment session at which several assessments were mad. Subjects then received either progressive relaxation or autogenic training for their insomnia.” (p. 255) Exposure/intensity to treatment: no treatment Duration treatment: 4 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “An additional requirement was that all subjects stop taking sleeping pills or other forms of medication with soporific effects after obtaining the permission of their physicians.” (p. 254) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote:”Within each wave, subjects were blocked according to time to fall asleep and then randomly assigned to the following conditions: (a) autogenic training, (b) progressive relaxation, (c) self‐relaxation control, and (d) no‐treatment control.” (p. 254) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Quote:“2 participants dropped out of the study for reasons unrelated to treatment.” (p. 254) Attrition <15% (6.25%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Pearl 1956.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 2 months Duration of participation (trial + follow‐up): 2 months. No follow‐up Setting: Inpatient (closed hospital ward) Purpose of trial: "The present investigation was undertaken to study the effectiveness of reserpine in the treatment of schizophrenic male patients within a controlled setting" (p. 198) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated
Number of participants included: 133
Number of participants followed‐up at post treatment: not stated Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: schizophrenia Diagnostic manual: not stated Means of assessment: Diagnostic interview: Lorr Multidimensional Scale for Rating Psychiatric Patients Comorbidity: different symptomatology of schizophrenia Age: not stated IQ: not stated Sex: 100% male Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: Placebo tablets
Description of intervention: "As a control factor, placebo tablets were obtained which were identical in physical characteristics with the drug tablets, e.g., shape, size, color, and taste. All personnel dispensing the placebo were told that it was a variant of reserpine named Plasepine. An equivalent number of placebo tablets was given to control patients, as were reserpine tablets to experimental subjects." (p. 199)
Individual or group treatment: individual treatment.
Exposure/intensity to treatment: "The actual reserpine dose varied from 2 to 10 mg. daily, the majority of patients receiving 4‐5 mg. a day." (p. 199)
Duration of treatment: 2 months
Concomitant psychotherapy: not stated, but inpatients
Concomitant pharmacotherapy: no adjunctive medication No‐treatment Comparison name: No‐treatment Description of intervention: not stated Exposure/intensity to treatment: no treatment Duration treatment: 2 months Concomitant psychotherapy: not stated, but inpatients Concomitant pharmacotherapy: no adjunctive medication |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote:"Within each ward, patients were randomly divided into three treatment groups." (p. 199) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | Quote:"In order to obtain as reliable judgments of condition as possible, in this study, the diagnostic interviews were jointly conducted by a team composed of psychiatrist, clinical psychologist, and psychological trainee. Each then rendered independent ratings based on his interview observations and inferences. Independent ratings of ward behavior were also secured from the nurse and charge aide on each of the experimental wards" (p. 199) No mention of blinding |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Peck 1976.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 5 weeks Duration of participation (trial + follow‐up): not stated Setting: inpatient Purpose of trial: “This study is an attempt to apply three types of desensitization procedures (Systematic Densitization, Vicarious Symbolic Desensitization, and Contact Densitization) to the mentally retarded.” (p. 138) Open/Closed placebo: closed placebo |
|
| Data |
Number of participants screened: 67 Number of participants included: 20 Number of participants followed‐up at post treatment: not stated Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: mild retardation + specific anxiety (heights/rats) Diagnostic manual: American Association of Mental Deficiency 1973 Means of assessment: Modified Fear Survey Schedule, Behavior Avoidance Test (BAT) Comorbidity: not stated Age: range = 19 to 61 years IQ:: 52 to 74 Sex: not stated Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological and pharmacological placebo Treatment name: Attention placebo + pill Description of intervention: “modified version of the “stress training” placebo rationale (...) was used. Ss were told that by learning to handle stress in one situation, they would learn not to be fearful in the presence of the phobic stimulus, Ss were instructed to take a “‘pill” (a passive placebo was used) which would relax them and left alone in the room for 5 minutes. The therapist subsequently reentered the room, checked the S’s pulse and eyelids and announced that he/she was relaxed. S was instructed to lie back, relax, and watch a cartoon on the videotape. The cartoons shown were interrupted visually and auditorily at the most exciting parts and a number was inserted as the presumed “stress training”. Ss were asked to read off these numbers as they appeared, and the therapist appeared to collect data on the number and latency of response. At the end of each session, S was told that he/she was progressing well and would soon be unafraid of the phobic stimulus (height or rat).” (p.139‐140) Individual or group treatment: individual Exposure/intensity to treatment: 15 sessions of 1,5 hours, three times a week Duration of treatment: 5 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “Fifteen of the Ss received tranquilizing medication of various types before and during the experiment.” (p. 139) No‐treatment Comparison name: No treatment Description of intervention: “No‐Treatment Control Ss were not contacted during the treatment phase.” (p. 140) Exposure/intensity to treatment: no contact Duration treatment: 5 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “Fifteen of the Ss received tranquilizing medication of various types before and during the experiment.” (p. 139) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote:“Ss were then randomly assigned within blocking variables (high and low anxiety) and height or rat stimuli to one of five conditions. … “ (p. 139) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Pelham 1992.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 6 weeks Duration of participation (trial + follow‐up): not stated Setting: outpatient Purpose of trial: “The present studies were designed to examine ADHD boys’ causal attributions in a double‐blind, within‐subject, placebo‐controlled medication trial.” (p. 283) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated
Number of participants included: 38
Number of participants followed‐up at post treatment: 38 Number of participants randomly assigned to:
Number of withdrawals : n= 0 Diagnosis: Attention Deficit/hyperactivity Disorder (ADHD) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition, Revised (DSM‐III‐R) Means of assessment: structured parent interview, standardised parent and teacher rating scales Comorbidity: “Eighteen also met criteria for conduct oppositional/defiant disorder,and another 8 met criteria for conduct disorder (…)” (p. 286) Age: 9 mean years and 11 months, (range =7 years and 3 months to 13 years and 9 months) IQ: Wechsler Intelligence Scale for children – mean: 106.2 (SD =13.1) Sex: 100% male Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name : Placebo
Description of intervention:
“Each subject received an identical capsule of either a low dose, a high dose, or
placebo before 8.00 a.m. and at midday, with the medication condition
randomized daily.” (p. 283)
“A day in the STP lasted from 8.00 a.m. until 5.00 p.m. on weekdays and was divided into the following activities: two academic classroom periods, each staffed by a special education teacher and an aide; an art class; swimming; three supervised, group, outdoor recreational activities (e.g., soccer); and lunch.” (p. 283)
Individual or group treatment: individual
Exposure/intensity to treatment: before 8.00 a.m. and at midday (dose not mentioned)
Duration of treatment: 6 weeks
Concomitant psychotherapy: not stated ‐ but was a part of a Summer Treatment Program
Concomitant pharmacotherapy: 22 of the 38 participants received pemoline, dexedrine spansule, slow‐release ritalin on days not included in the experiment. No carry‐over effect was present as concluded by the authors. No‐treatment Comparison name: No pill Description of intervention: "No treatment but attendance in the Summer Treatment Program. “A day in the STP lasted from 8.00 a.m. until 5.00 p.m. on weekdays and was divided into the following activities: two academic classroom periods, each staffed by a special education teacher and an aide; an art class; swimming; three supervised, group, outdoor recreational activities (e.g., soccer); and lunch.” (p. 283) Exposure/intensity to treatment: no pill Duration treatment: 6 weeks Concomitant psychotherapy: not stated ‐ but was a part of a Summer Treatment Program Concomitant pharmacotherapy: 22 of the 38 participants received pemoline, dexedrine spansule, slow‐release ritalin on days not included in the experiment. No carry‐over effect was present as concluded by the authors. |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | No information |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Outcome was patient‐reported |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Attrition < 15% (0%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | No | Quote: “Each subject received an identical capsule of either a low dose, a high dose, or placebo before 8.00 a.m. and at midday, with the medication condition randomized daily.” (p. 283) Potential carry‐over effect |
Pendleton 1983.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 6 weeks Duration of participation (trial + follow‐up): 6 weeks + 2 weeks post‐assessment Setting:outpatient (University setting) Purpose of trial : “The purpose of this study was to evaluate the effectiveness of a symptom scheduling technique in reducing excessive fear.” (p. 317) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 62 Number of participants followed‐up at post treatment: 58 Number of participants randomly assigned to :
Number of withdrawals: n= 4
Diagnosis : Specific anxiety (Acrophobia) Diagnostic manual: not stated Means of assessment: Acrophobia Behavioral Test Comorbidity: not stated Age: 25.1 mean years (SD = 6.75) (R =17 to 54) IQ: not stated Sex: 74.2% females Ethnicity: not stated Country : USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Relaxation training expectancy control Description of intervention: ”Subjects in the relaxation only treatment condition received a treatment rationale in which relaxation training was described as a passive, automatic process that acts in a cumulative manner.” (p. 319) “Although the relaxation only condition was included as an expectancy control, it appears in retrospect to have inadvertently contained specific treatment components.” (p. 321) Individual or group treatment: group Exposure/intensity to treatment: 45 minutes x 6 sessions Duration of treatment: 6 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: Wait‐list Description of intervention: ”Subjects in the waiting list control condition were contacted during the week following the pre‐testing session and informed that their treatment would be delayed. They were then retested during the regular posttesting period. After posttesting, these subjects were debriefed and then treated with the desensitization procedure.” (p. 319) Exposure/intensity to treatment: no treatment during waiting Duration treatment: 6 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”Acrophobic subjects were randomly assigned to one of three treatment conditions.” (p. 318) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:” Posttesting session. Posttesting appointments were scheduled one to two weeks after completion of all treatment sessions. All pretreatment measures were re‐ administered in the same manner as in pre‐testing by an experimenter who was blind to the subjects’ pretreatment performances and condition assignments.” (p. 319) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Quote:”Two community and two student subjects (one each from both the negative practice and relaxation only conditions) dropped out of the study due to scheduling conflicts.” (p. 318) Attrition <15% (6.5%). No ITT. Reports data on completers only |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | None other sources found |
Pillman 2001.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation : not stated Cluster randomised: no Duration of trial (baseline to post): participants averaged 22 days to complete the study (range between 18 to 35 days from pre‐ to post‐test) Duration of participation (trial + follow‐up): “All tests given on the 1 st day of testing (…) The final day of testing was administered 28 days after the initial testing.” (p. 56) Setting: inpatient substance abuse rehabilitation Purpose of trial: “Twenty‐one subjects with a primary diagnosis of alcohol abuse in a 30‐day inpatient treatment program were placed into three groups to determine the effectiveness of the computerized cognitive remediation treatment, NeurXercise.” (p. v) Closed/open placebo: closed placebo |
|
| Data |
Number of participants screened: 45
Number of participants included: 38
Number of participants followed‐up at post treatment: 22
Number of participants randomly assigned to:
Number of withdrawals: n = 20
Diagnosis: alcohol dependency Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: not stated Comorbidity: not stated (but excluded if they were polysubstance abusers) Age: Pharmacological placebo: 48.43 mean years (SD = 7.43), no‐treatment: 47.25 mean years (SD = 7.34) IQ: Wechsler Memory Scale, 117.8 Sex:: 100% male Ethnicity: only median given, 1 = Caucasian. It is not stated whether the other possibilities (2 = African American, 3 = Asia, 4 = Hispanic, and 5 = other) Country: USA Country of treatment (where did the treatment take place): USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo Treatment name: Placebo Description of intervention:“Those subjects in the placebo treatment group listened to several novels on audiotape. During the first two 1‐hour training sessions, the examiner supervised the patients to make certain that they were aware of the procedures and could operate the tape machine independently (p. 57) Individual or group treatment: individual Exposure/intensity to treatment: a total of 15 hours. “The examiners saw the patients in the placebo treatment group every 2 to 3 treatment hours to briefly discuss their progress. After 15 hours of placebo treatment, the patients were given the final battery of neuropsychological tests (…)” (p. 58) Duration of treatment: 22 days Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated – but the demographic questionnaire obtained information concerning current medications Wait‐list Comparison name: no treatment (in reality wait‐list) Description of intervention: “All subjects were informed of the 66% possibility that they would not be placed in the treatment group and that if the treatment was found to be successful, they had the right to return to undergo cognitive remediation treatment.” (p. 55) “Subjects in the no‐treatment group were given the initial battery of neuropsychological tests upon their agreement to participate. After 14 days, they were administered ANAM v3.11, and after 28 days they were administered the full battery of neuropsychological measures (…) They were allowed to ask questions and were informed as to their right to undergo cognitive remediation if the cognitive remediation was found to be effective.” (p. 58) Exposure/intensity to treatment: no treatment Duration treatment: 22 days Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated – but the demographic questionnaire obtained information concerning current medications |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | No | Quote: ”Subjects who met the initial criteria for participation in this study were systematically assigned to one of the three groups on an ongoing basis determined by when they entered the treatment program (i.e., Subject 1 was placed in Group 1, Subject 2 was placed in Group 2, Subject 3 was placed in group 3, and Subject 4 was placed in Group 1, etc.).” (p. 55) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | The outcome assessment is blinded, as the ANAM is computer‐based |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Quote: “(…) the chi‐square analysis of the demographic variables age, ethnicity, years of schooling, and handedness showed that those subjects completing the study did not differ significantly from those who terminated early.” (p 60) Attrition >15% (52.6%). No ITT used. |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Poland 2013.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 8 weeks Duration of participation (trial + follow‐up): 8 weeks of participation – no follow‐up Setting: outpatient Purpose of trial: “The study objectives were to determine whether massage therapy reduces symptoms of depression in subjects with human immunodeficiency virus (HIV) disease.” (p. 334) |
|
| Data |
Number of participants screened: 81 Number of participants included: 54 Number of participants followed‐up at post treatment: 37 Number of participants randomly assigned to:
Number of withdrawals: n =17
Diagnosis: major depressive disorder (in HIV infected subjects) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: The Structured Clinical Interview for DSM‐IV Disorders (SCID) Comorbidity: HIV. Otherwise not stated Age: Pharmacological placebo: 42.6 mean years (SD = 5.1), and No‐treatment 42.6 mean years (SD = 4.9) IQ: not stated Sex: 14.8% female Ethnicity: from ITT: African American: n = 6, White: n = 26, Hispanic: n = 8, other: n = 10 Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo Treatment name: Light touch Description of intervention: "Using a novel dual‐control group design,16 which included a light ‘‘touch’’ group to control for some of the nonspecific effects of massage and therapist–subject interaction,“ (p. 335) “The touch group had a massage therapist place both hands on the subject with slight pressure, but no massage, in a uniform distribution for 1 hour twice per week in the same pattern used for the massage subjects. Subjects were told that for study purposes, verbal communication between them and the therapist should be kept to a minimum (...).” (p. 335) Individual or group treatment: individual Exposure/intensity to treatment: twice per week for one hour (Monday/Thursday or Tuesday/Friday) Duration of treatment: 8 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: yes. “Subjects had to be on a stable neuropsychiatric, analgesic, and antiretroviral regimen for at least 4 weeks and planning to remain on the same regimen for the 8‐week duration of the study.” (p. 335) “Approximately 40% of the subjects also were currently taking antidepressants. As with the antiretroviral regimens, subjects had been on a stable antidepressant regimen for > 30 days and the regimen remained fixed for the duration of the study.” (p. 335) No‐treatment Comparison name: No intervention (no treatment) Description of intervention: no description of intervention Exposure/intensity to treatment: no treatment Duration treatment: 8 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: Yes ‐ “Subjects had to be on a stable neuropsychiatric, analgesic, and antiretroviral regimen for at least 4 weeks and planning to remain on the same regimen for the 8‐week duration of the study.” (p. 335) “Approximately 40% of the subjects also were currently taking antidepressants. As with the antiretroviral regimens, subjects had been on a stable antidepressant regimen for > 30 days and the regimen remained fixed for the duration of the study.” (p. 335) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: one found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: ”Subjects were enrolled by the study coordinators and then randomized using a random numbers table by a nurse practitioner whose sole role in the study was to randomize subjects.” (p. 335) |
| Allocation concealment | Yes | Quote:(…) randomized using a random numbers table by a nurse practitioner whose sole role in the study was to randomize subjects. Subjects were randomized 1:1:1 into one of three parallel groups” (p. 335) |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Quote:“50 subjects completed at least 1 week of the protocol (intent‐to‐treat; ITT) (…) The last information carried forward (LOCF) method was used for analyses of the ITT group.” (p. 335). Attrition >15% (31.5%) |
| Selective outcome reported | Unclear |
NCT00033852. No apparent differences in reporting between trail registry and full report, but information about outcome measures and eligibility criteria was not available. |
| Other sources of bias | Yes | No other sources found |
Powers 2004.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 1 day: (“They were also told that one effective strategy for reducing their fear is to be exposed to the feared situation repeatedly until the anxiety decreases. Participants in the three exposure conditions received a total of 30 min of self‐guided in vivo exposure to the claustrophobic chamber used for BAT 1.” (p. 450)) Duration of participation (trial + follow‐up): 1 day + 2 weeks follow‐up Setting: outpatient Purpose of trial: “The primary aim of the current study was to further investigate the deleterious effects of safety‐seeking behaviors on fear reduction by disentangling the effects of perceived availability of threat‐relevant safety behaviors during treatment versus their actual use.” (p. 449) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: pool of approximately 5000 Number of participants included: 72 Number of participants followed‐up at post treatment: 71 Number of participants randomly assigned to: 71
Number of withdrawals: n = 1
Diagnosis: claustrophobia Diagnostic manual : 75% met full Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV), 25% met all DSM‐IV criteria with the exception of Criterion E (i.e.the person must experience significant interference in social, academic, or work functioning or experience marked distress about having the phobia) Means of assessment: first potential participants rated their overall fear on a 5‐point Likert scale, afterwards participants were administered the CIDI‐Auto Comorbidity: not stated Age: 21.06 mean years (SD = 5.08), (range 18 to 49 years) IQ: not stated Sex: 86% female Ethnicity: 74% Caucasian, 13% Mexican American, 7% African American, 5% Asian American, and 1% Indian American Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Placebo Description of intervention: “Participants in the PL group returned 2 weeks after completing screening and received a similar rationale (…)”. (p. 450). “The DAVID developed by Comptronic Devices is used by health care professionals as a relaxation device. I is a small soundboard about the size of a stereo receiver, which includes a headset and plastic mask. The headset emits controllable ticking sounds, similar to those made by a metronome. The plastic mask resembles ski goggles and delivers pulsed orange lights at controllable rates. In this study, the audio and video stimulus frequency was set at 12 Hz (cycles per second), which is the rate at which the device is suggested to maximally produce relaxation and meditative states.” (p. 450) Individual or group treatment: individual Exposure/intensity to treatment: one treatment Duration of treatment: one treatment Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: Wait‐list Description of intervention: “This group was informed that they had been placed on a WL. They returned for assessment 2 weeks later and completed the post‐assessment. Following assessment, they received exposure treatment.” (p. 450) Exposure/intensity to treatment: no treatment Duration treatment: no treatment Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote:”Eligible participants were randomly assigned to one of five conditions: (a) EO, (b) exposure with SBU, (c) exposure with SBA, (d) credible psychological PL, or (e) WL control.” (p. 449) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Attrition <15% (1,41%) |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Powers 2008a.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 1 session (1 day) Duration of participation (trial + follow‐up): 1 day + 1‐week follow‐up Setting: outpatient Purpose of trial: “On the basis of the available evidence, we hypothesized the following: (a) Participants led to believe that they ingested a sedating herb with anxiety dampening effects would show significantly greater return of fear compared with those led to believe that they ingested a placebo; (b) participants led to believe that they ingested a stimulating herb with anxiety enhancing effects would show significantly enhanced maintenance of treatment gains at follow‐up compared with those led to believe that they ingested a placebo; and (c) the effects of the pill expectancy manipulation on changes in fear during the follow‐up period would be mediated by changes in coping self‐efficacy.” (p. 479) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 5326 Number of participants included: 95 Number of participants followed‐up at post treatment: 95 Number of participants randomly assigned to:
Number of withdrawals: n = 0 Diagnosis : Claustrophobia Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: Composite International Diagnostic Interview (CIDI‐Auto). Most participants (74%) met full Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM–IV; APA 1994 ) criteria for claustrophobia, whereas 26% met all DSM–IV criteria with the exception of Criterion E, which requires that the person experience significant interference in social, academic, or work functioning or marked distress about having the phobia Comorbidity : not stated Age: 20.11 mean years (SD = 6.23), (range = 18 to 60) IQ: not stated Sex: 71 % female Ethnicity: the ethnic breakdown of the sample was 73% Caucasian, 12% Hispanic, 9% African American, 4% Asian, and 2% Native American. Country: the Netherlands Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Credible Psychological Placebo Treatment Description of intervention: “Participants assigned to the psychological placebo condition returned 1 week following pretreatment assessment to receive 30 min of pulsed audio‐photic stimulation with a device called the Digital Audio Integration Device (DAVID) Paradise XL (Mind Alive Inc., Edmonton, Alberta, Canada). It consists of a headset, which emits controllable pulsing sounds, and plastic goggles, which produce pulsing lights at controllable rates. The number of treatment trials (N 6), the size and layout of the treatment room, the position of the participant (supine), and the duration of each trial (5 min) were equivalent to those receiving the exposure treatment. However, they received no exposure treatment.“ (p. 482) Individual or group treatment: individual Exposure/intensity to treatment: 1 session ‐ 30 minutes Duration of treatment: 1 day (1 session) Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name : Wait‐list Description of intervention: “Participants in the waitlist condition completed assessments at each of the three time points and were offered exposure treatment following study completion.” (p. 482) Exposure/intensity to treatment: 1 session Duration treatment: 1 day Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote:“Research assistants enrolled and randomized participants by cycling through a list consisting of a computer generated random sequence of the four treatment conditions (...). Three times as many participants were randomized to the exposure treatment plus inactive pill condition in anticipation of later randomization to the three perceived pill effect conditions.” (p. 482) ” Analyses showed no significant differences between groups at baseline on any of these measures (all ps = .20), suggesting that randomization was successful.” (p. 483) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Attrition <15% (0%) |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Powers 2008b.
| Study characteristics | ||
| Methods | See Powers 2008a | |
| Data | See Powers 2008a | |
| Comparisons |
Pharmacological placebo Treatment name: Exposure + inactive pill Description of intervention: “Prior to the start of exposure treatment, they were administered an inactive pill of 250 mg of Vitamin C and told that the experiment would be investigating an anxiety treatment while simultaneously examining the effects of an herbal supplement—“Adomoxin” (a fictitious name)— on memory.” (p. 482) Individual or group treatment: individual Exposure/intensity to treatment : Six 5‐minute trials Duration of treatment: 1 day (1 session) Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: Exposure + no pill Description of intervention: “In brief, this treatment consisted of several elements, including (a) brief education about the nature of claustrophobia, (b) rationale for exposure treatment, (c) six 5‐min trials of in vivo exposure to a claustrophobic chamber identical to that used in the BAT‐1 assessment, and (d) completion of treatment process ratings before and after each exposure trial.” (p. 482) Exposure/intensity to treatment: Six 5 min trials Duration treatment: 1 day Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes | See Powers 2008a | |
| Notes | See Powers 2008a | |
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote:“Research assistants enrolled and randomized participants by cycling through a list consisting of a computer generated random sequence of the four treatment conditions (...). Three times as many participants were randomized to the exposure treatment plus inactive pill condition in anticipation of later randomization to the three perceived pill effect conditions.” (p. 482) ” Analyses showed no significant differences between groups at baseline on any of these measures (all ps = .20), suggesting that randomization was successful.” (p. 483) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Attrition <15% (0%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Proudfoot 2013.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 7 weeks Duration of participation (trial + follow‐up): “Eligible participants completed a baseline questionnaire prior to randomization, a post‐intervention questionnaire administered at eight weeks, and a follow‐up questionnaire administered 12 weeks later for participants in the myCompass and AC groups, and 19 weeks later for the WL group.” (p. 4) Setting: outpatient Purpose of trial: “The aim of this paper is to report the outcomes of a CONSORT‐compliant randomised controlled trial (RCT) to evaluate the efficacy of the myCompass program in a large community sample of people experiencing mild‐to‐moderate depression, anxiety and/or stress. We predicted that symptoms of depression, anxiety and stress would reduce in participants randomly allocated to receive myCompass, relative to both attention control (AC) and waitlist (WL) conditions. We also predicted that use of myCompass would increase work and social functioning relative to the AC and WL conditions.” (p. 2) Open/closed placebo: Closed placebo |
|
| Data |
Number of participants screened: 2955 Number of participants included: 720 Number of participants followed‐up at post treatment: 515 Number of participants randomly assigned to:
Number of withdrawals: n = 205
Diagnosis: depression, anxiety or/and stress Diagnostic manual: not stated Means of assessment: Depression, Anxiety and Stress Scales (DASS) Comorbidity: 65% had comorbid symptoms. Not specified which Age: psychological placebo: 40 (SD = 11.42), wait‐list: 38 (SD = 10.26) IQ: not stated Sex: 68.2% female Ethnicity: not stated Country: Australia Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo Treatment name : Attention control Description of intervention: "myCompass is a fully‐automated, self‐help, public health intervention, that is tailored to the user and has no therapist input." (p. 3) “Attention control participants received a control mental health program matched to the active intervention on duration and mode of delivery. Each week for seven weeks, they received a fact sheet containing information about depression, anxiety or stress sent to their email address. The information was designed to be read on computer in approximately 10 minutes, and to be credible but void of management advice or treatment strategies. They also received on their mobile phones weekly SMS messages containing brief factual statements about depression, anxiety and stress. The mobile phone statements were also therapeutically inactive, but chosen to ensure that the control program had face validity.” (p. 3‐4) Individual or group treatment: individual Exposure/intensity to treatment: each week they received a fact sheet and weekly SMS messages containing brief factual statements Duration of treatment: 7 weeks Concomitant psychotherapy: not excluded if they did, but no records (author correspondence) Concomitant pharmacotherapy: probably but not stated. “(…) and use of antidepressant and anxiolytic medication were also assessed.” (p. 4) Wait‐list Comparison name: Wait‐list Description of intervention: “Waitlist participants did not receive emails or SMSs during the intervention phases, but received full access to the myCompass program at the end of the seven weeks.” (p. 3) Exposure/intensity to treatment: no treatment Duration treatment:: 7 weeks Concomitant psychotherapy: not excluded if they did, but no records (author correspondence) Concomitant pharmacotherapy: probably but not stated. “(…) and use of antidepressant and anxiolytic medication were also assessed.” (p. 4) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote:”A research assistant not involved in the RCT randomised participants after baseline using computerised random numbers. Allocation was either to the myCompass, AC or WL condition.” (p. 3) |
| Allocation concealment | Yes | Quote:”A research assistant not involved in the RCT randomised participants after baseline using computerised random numbers.” (p. 3) |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Unclear | Quote:“Effects of the myCompass intervention on study outcomes were evaluated using intention‐to‐treat (ITT) analyses that included data from all participants who completed the baseline assessment and any follow‐up assessment. Strategies for dealing with missing data in longitudinal studies vary, so we adopted two recommended techniques for analysing incomplete datasets, namely, mixed models repeated measures (MMRM) and multiple imputation (…)” (p. 4) Attrition >15% (MyCompass: 47.9%, Attention control: 22.2%, Wait‐list: 14.8%) |
| Selective outcome reported | No | ACTRN 12610000625077 Differences between protocol and report are found 1) 6 weeks of treatment in the protocol, 7 weeks in the report. 2) According to the protocol the assessments are made at baseline, and at 3, 6, 9, 12, 15 and 18 weeks after intervention commencement – in the report assessments are reported for baseline, 8 weeks (post treatment) and a 12 weeks follow‐up 3) It is stated in the protocol that masking is used – but this is not stated in the report |
| Other sources of bias | Yes | No other sources found |
Quayhagen 1995.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 12 weeks Duration of participation (trial + follow‐up): 12 weeks + 6 months Setting: outpatient Purpose of trial: “The objective of this study was to determine the impact over time on the cognitive and behavioral functioning of the care recipient from a home‐based intervention program of active cognitive stimulation implemented by the family caregiver.” (p. 154) Open/closed: closed placebo |
|
| Data |
Number of participants screened: 132 family units Number of participants included: 95 family units Number of participants followed‐up at post treatment: 79 (only 78 retained for analysis – one family unit was eliminated due to data inconsistency) Number of participants randomly assigned to:
Number of withdrawals: n = 16
Diagnosis : Alzheimer’s disease Diagnostic manual: not stated Means of assessment : Mattis Dementia Rating Scale Comorbidity: not stated Age: 73.6 mean years (SD = 8.0) IQ: not stated Sex: 28.4% female Ethnicity: white (85%), African American (3%), Hispanic (11%) Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: placebo Description of intervention:“With neuropsychological consultation, the decision was made to select activities similar to those in the experimental condition, but using a passive approach. Caregiver implementation relied heavily of the modeling work of Bandura (1977), where the impaired member was exposed to passive observation of the activity without enforced participation.” (p. 156) Individual or group treatment: individual Exposure/intensity to treatment: “All families in the treatment groups attended 12 consecutive weekly in‐home sessions with members of the intervention team. The caregiver and the care recipient were trained together in program implementation techniques.” (p. 155). “Following each weekly instruction session, the intervention was executed in the home by the family caregiver.” (p. 153) Duration of treatment: 12 weeks. Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “None were participating in pharmacological clinical trials.” (p. 154) Wait‐list Comparison name: Wait‐list control Description of intervention: “The families assigned to the control group were placed on a waiting list for complementary sessions on the cognitive stimulation program once the wave of the study in which they were participating was completed.” (p. 156) Exposure/intensity to treatment: no treatment Duration treatment: 12 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: ”None were participating in pharmacological clinical trials.” (p. 154) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: one found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”(...) the care recipients were stratified by degree of cognitive impairment to maintain initial comparability of functioning across groups. They were then randomly assigned to one of three conditions: (a) intervention (active cognitive stimulation), (b) placebo (passive cognitive stimulation), and (c) wait‐list control (no stimulation, but poststudy introduction to treatment).” (p. 155) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | Quote: “To control for experimenter bias, assessments were conducted by research assistant who, with rare exception, were blinded to the condition to which the family had been assigned.” (p. 155) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | No | Attrition >15% (17%). No mention of ITT |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | No | Quote: ”Return demonstrations by caregivers were required to validate training. At each session, the caregiver was reminded to give positive feedback and to complete a weekly log that included success or problems in implementation and the amount of time spent each day with the intervention.” (p. 155). Quote: “Through log recordings of the caregivers and the observation of the investigative team, it became apparent early in the study that a group of the care recipients in the placebo condition had exceeded the passivity parameters and were working toward self‐initiated improvement.” (p. 156) |
Rabkin 1990.
| Study characteristics | ||
| Methods |
Parallel randomised trial with two arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 6 weeks Duration of participation (trial + follow‐up): 12 week follow‐up Setting: outpatient Purpose of trial: to evaluate pill‐taking as determinant of placebo response Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated
Number of participants included: 50
Number of participants followed‐up at post treatment: 50
Number of participants randomly assigned to:
Number of withdrawals: n = 0 Diagnosis: major depression Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition (DSM III) Means of assessment: Hamilton rating scale for depression. Comorbidity: dysthymia = 18, major depressive disorder (MDD) =17, dysthymia + MDD = 15 Age: 37 mean years (SD = 10) (Range = 15 to 55) IQ: not stated Sex: 54% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name:: Placebo
Description of intervention: single‐blind pill placebo responders
Individual or group treatment: Individual
Exposure/intensity to treatment: not stated
Duration of treatment: 6 weeks.
Concomitant psychotherapy: not stated
Concomitant pharmacotherapy: not stated No‐treatment Comparison name: No‐treatment Description of intervention: received no intervention Exposure/intensity to treatment: no treatment Duration treatment: 6 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “The randomization process followed the order provided by our statistician in blocks of 4. I’m really not sure, but likely used a predefined list. (author correspondence) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: “All assessors were blinded” (author correspondence) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Quote: “Over the 5‐year period of the study, 58 patients were rated as 10‐day placebo responders; these patients constituted 10% of all patients who received single‐ blind medication in clinical trials during this period. Four of the 58 patients refused to participate in the study, one moved out of the area during the trial, one refused to stop taking the single‐blind placebo pills, one dropped out, and one denied 2 weeks later that he had been a placebo responder in the first place. The eight patients who did not complete the study did not differ from the 50 who did complete the study with respect to demographic characteristics, illness history, baseline illness characteristics, baseline symptoms, or severity of depression.” (p. 1623) Attrition <15% (0% in the second trial) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Rapee 2006.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 12 weeks of treatment Duration of participation (trial + follow‐up): 12 weeks of treatment + 24 weeks follow‐up Setting: outpatient Purpose of trial: “The aims of the current study was to examine the impact of using parents as therapists for their own child in a trial of bibliotherapy materials for parents of children with anxiety disorders.” (p. 437) |
|
| Data |
Number of participants screened: not stated Number of participants included: 267 Number of participants followed‐up at post treatment: 212 Number of participants randomly assigned to
Number of withdrawals : n = 55
Diagnosis: anxiety disorder Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: Anxiety Disorders Interview Schedule, Parent and Child Versions (ADIS‐CP) Comorbidity: main comorbid diagnoses = anxiety disorder (N = 219, 82%), externalising disorder (N = 72, 27%), mood disorder (N = 23,8.6%). Generalised anxiety disorder (N = 103). Social phobia (N = 64). Separation anxiety disorder (N = 51). Specific phobia (N = 33). Obsessive‐compulsive disorder (N = 13) Panic disorder (N = 3) Age: age in months: usual care: 113.7 (SD = 20.4) Waitlist: 114.1 (SD = 19.1) IQ: not stated Sex: usual care: 53.3% female. Wait‐list: 29.9% female Ethnicity: not stated Country: Australia Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Usual care Treatment name: Standard group treatment Description of intervention: “Group treatment was based on the Cool Kids Program, a nine‐session cognitive‐behavioral program for the management of broad‐based childhood anxiety disorders.” (p. 437) “Parents and children attend all nine sessions of the program on a weekly basis over 12 weeks (the final few sessions are biweekly) and cover recognition of emotion and anxiety, realistic thinking, child management strategies, exposure to feared cues, and additional skills such as assertiveness and dealing with teasing. (p. 438) Individual or group treatment: group treatment Exposure/intensity to treatment: nine sessions, approximately 2 hours per session Duration of treatment: 12 weeks Concomitant psychotherapy: “Generally we allow concomitant medication after stabilization; and we allow psychotherapy for unrelated problems also after stabilization” (author correspondence) Concomitant pharmacotherapy: yes. “Children on medication were included if the medication had been stable for the previous month.” (p. 437) “Generally we allow concomitant medication after stabilization; and we allow psychotherapy for unrelated problems also after stabilization” ( Rapee 2006 (pers comm) ) Wait‐list Comparison name: Wait‐list Description of intervention: “Participants in the waitlist were simply told that they had been randomly assigned to wait for treatment and that they would be recontacted for additional assessments in 3 months’ time, after which they would be offered the next available treatment group.” (p. 438) Exposure/intensity to treatment: no treatment during waiting Duration treatment: 12 weeks Concomitant psychotherapy: “Generally we allow concomitant medication after stabilization; and we allow psychotherapy for unrelated problems also after stabilization” (author correspondence) Concomitant pharmacotherapy: yes. “Children on medication were included if the medication had been stable for the previous month.” (p. 437). “Generally we allow concomitant medication after stabilization; and we allow psychotherapy for unrelated problems also after stabilization” ( Rapee 2006 (pers comm) ) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: ”Randomization occurred in block of eight to allow allocation to group treatment based on a predetermined random number schedule known only to the study coordinator.” (p. 438) |
| Allocation concealment | Yes | Quote: ”(...) known only to the study coordinator.” (p. 438) |
| Blinding of outcome assessors | Yes | Quote: ”Repeated interviews were conducted by clinicians who were masked to the child’s allocated treatment (…)” (p. 437) |
| Blinding of participants and personnel | No | Not possible to blind usual care and wait‐list |
| Incomplete outcome data | No | Quote: "Analyses were made on completers and on all randomized (intent‐to‐treat). “Intention‐to‐treat analyses included all participants who were allocated to a condition (aside from 7 participants who did not return any data at pretreatment) and used the last‐point‐carried‐forward method to deal with missing data.” (p. 439) Attrition >15% (20.6%). LOCF used. Only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Rapee 2007.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 12 weeks Duration of participation (trial + follow‐up): 12 weeks of treatment + 24 weeks follow‐up Setting : outpatient Purpose of trial: “Our study was designed to determine the value of two forms of self‐help through the use of bibliotherapeutic materials in the reduction of social phobia: pure bibliotherapy that involved almost no contact with the researchers, and therapist‐augmented bibliotherapy in which printed material was supplemented with five group sessions conducted by a therapist. Benchmarks for these conditions were provided by comparisons with a no‐treatment waiting list and standard ten‐session group therapy conducted by therapist.” (p. 246) |
|
| Data |
Number of participants screened: not stated Number of participants included: 224 Number of participants followed‐up at post treatment: 177 Number of participants randomly assigned to:
Number of withdrawals : n = 47
Diagnosis: 95.7% met criteria for generalized subtype of social phobia and 55.8% met criteria for a diagnosis of avoidant personality disorder Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: Anxiety Disorders Interview Schedule + International Statistical Classification of Diseases and Related Health Problems, 10th edition (ICD‐10) International Personality Disorder Examination Comorbidity: 42.9% met criteria for an additional anxiety disorder, 33.9% met criteria for an additional mood disorder and 4.0% met criteria for an additional substance use or alcohol disorder Age: 35.5 mean years (SD = 11.0) IQ: not stated Se:: 50.4% female Ethnicity: not stated Country: Australia Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Usual care Treatment name:Standard group treatment Description of intervention: “Treatment was conducted in groups of approximately six participants (..) Therapy extended for ten 2 h sessions across 12 weeks. (...) Components included those typically found in empirically validated treatments for social phobia including cognitive restructuring of negative evaluation beliefs, exposure to feared social situations, realistic feedback of social performance, and attention training. Participants engaged in home exercise and received various handouts as relevant.” (p. 247) Individual or group treatment: group Exposure/intensity to treatment: 10 two‐hour sessions Duration of treatment: 12 weeks Concomitant psychotherapy: no participant was in concurrent psychotherapy but they were allowed to. “Concurrent pharmacotherapy or psychotherapy was allowed as long as dosage had been consistent for 3 months and there was no plan to change.” (p. 246) Concomitant pharmacotherapy: “Concurrent pharmacotherapy or psychotherapy was allowed as long as dosage had been consistent for 3 months and there was no plan to change.” (p. 246).“(…) 6.8% were taking benzodiazepines or other anxiolytics, 21.2% were taking selective serotonin reuptake inhibitors or other antidepressants and 9.9% were taking other prescription medications.” (p. 246) Wait‐list Comparison name: Waiting list Description of intervention: “Participants on the waiting list were told that they had been randomly allocation to receive no treatment for 12 weeks. At the end of the 12‐week period they were offered out best available treatment.” (p. 247) Exposure/intensity to treatment: no treatment during waiting Duration treatment: 12 weeks Concomitant psychotherapy: no participant was in concurrent psychotherapy but they were allowed to. “Concurrent pharmacotherapy or psychotherapy was allowed as long as dosage had been consistent for 3 months and there was no plan to change.” (p. 246) Concomitant pharmacotherapy: “Concurrent pharmacotherapy or psychotherapy was allowed as long as dosage had been consistent for 3 months and there was no plan to change.” (p. 246) “(…) 6.8% were taking benzodiazepines or other anxiolytics, 21.2% were taking selective serotonin reuptake inhibitors or other antidepressants and 9.9% were taking other prescription medications.” (p. 246) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “Randomisation was done using a pre‐assigned random number generator in blocks of eight to allow for group delivery.” (p. 247) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: “As much as possible. That is – we do not tell assessor about the condition a patient is in and we instruct patients not to describe their treatments. But of course in some cases they do talk about it” ( Rapee 2007 (pers comm) ) |
| Blinding of participants and personnel | No | Not possible to blind usual care and wait‐list |
| Incomplete outcome data | Unclear | Quote: “Interpolation was used if post‐treatment data only were not available. As a precaution against biasing effects of these methods of handling missing data, analyses were conducted with and without missing data substituted. Analyses with missing data substituted are equivalent to intent‐to‐treat analyses.” (p. 248) "Means are calculated with missing data substituted by the last observed value or the interpolation of adjacent values (described in more detail in the method section)". (p. 250) Attrition >15% (21%) |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Robin 1976.
| Study characteristics | ||
| Methods |
Parallel randomised trial with two arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 1 week in immediate groups, but participants in the wait‐list group were on the list for 12 weeks (range 6 to 18 weeks) Duration of participation (trial + follow‐up): no follow‐up Setting: outpatient Purpose of trial: “This study examines the effect of the waiting list in a prospective controlled trial.” (p. 138) |
|
| Data |
Number of participants screened: not stated Number of participants included: 234 Number of participants followed‐up at post treatment: 234 Number of participants randomly assigned to:
Number of withdrawals: n = 0 Diagnosis: psychiatric patients with different diagnoses: affective disorder, neurosis, personality problem, alcoholism/drug dependence, schizophrenia, organic cerebral disease, mental handicap Diagnostic manual: not stated Means of assessment: not stated Comorbidity: different diagnoses Ag:: not stated IQ: 3% were mentally handicapped Sex: 50.4% female Ethnicity: not stated Country: UK Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Usual care Treatment name: immediate appointments Description of intervention: “(…) so that patient referred by general practitioners in the week before such a clinic could be offered appointments within seven days (‘immediate appointments’)” (p. 138) Individual or group treatment: not stated– various treatments Exposure/intensity to treatment: not stated – various treatments Duration of treatment: 1 week Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: Delayed appointment Description of intervention: “Those referred in the following weeks (‘delayed appointments’) would join a waiting list of, on average, 12 weeks’ duration (range 6‐18 weeks).” (p. 138) Exposure/intensity to treatment: no treatment during the wait‐list period Duration treatment: 12 weeks – range 6 to 18 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | No | Quote: “Without specific announcement, alternate out‐patient clinics were kept vacant so that patients referred by general practitioners in the week before such a clinic could be offered appointment within seven days (‘immediate appointments’). Those referred in the following weeks (‘delayed appointments’) would join a waiting list of, on average, 12 weeks’ duration (range 6‐18 weeks). Referrals were thus randomized between ‘immediate’ and ‘delayed’ appointments, but where urgency was specifically stressed by the family doctor after an appointment date had been offered, and effort was made to provide an earlier appointment for delayed patients.” (p. 138) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Outcome is ‘attended/did not attend’ |
| Blinding of participants and personnel | No | Not possible to blind usual care and wait‐list |
| Incomplete outcome data | Yes | Not relevant |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | No | Time bias in outcome measure. Patients in the immediate group was “assessed” for attenending immediately, but patients in the delayed was assessed after 12 weeks of wait‐list treatment. Attention bias: patients in TAU received treatment for 1 week, while WL received treatment for appr. 12 weeks (Range = 6‐18 weeks) |
Roehrich 1993.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 2 weeks Duration of participation (trial + follow‐up): 2 weeks (no follow‐up) Setting: inpatient alcohol treatment program Purpose of trial: “The current investigation focused on treatment‐relevant remediation (acquisition of the content of a relapse‐prevention [RP] program) using task administered by self‐guided workbooks.” (p. 812) Closed/open placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 80 Number of participants followed‐up at post treatment : 61 Number of participants randomly assigned to :
Number of withdrawals: n = 19 Diagnosis: substance use disorder (alcohol dependence) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition (DSM III) Means of assessment: not stated Comorbidity: “Forty percent of the subjects admitted to past drug abuse or dependence, and 21% of the sample had received prior treatment for depression, drug dependency, or posttraumatic stress disorder.” (p. 814) Age: psychological placebo: 42.38 mean years (SD = 10.65), no‐treatment: 43.13 mean years (SD =11.54) IQ: Wechsler Adult Intelligence Scale –, psychological placebo: 101.31 (SD = 12.85), no‐treatment: 96.07 (SD = 8.61) Sex: 100% male Ethnicity: 82% white, 16% black, 2% listed themselves as “other” Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Alcohol opinions and attitudes (PBO‐REM) Description of intervention: “Subjects received workbooks according to their treatment group status 10 to 12 days after admission. Instructions for the workbooks appeared inside each booklet and were not presented by the experimenter.” (p. 814) “The PBO‐REM workbook also contained elements of repetition and feedback. However, in this case, subjects were asked to respond to statements about alcoholism and then to provide a written rationale for their choices. This workbook relied heavily on automatic verbal skills that have been noted to be relatively unimpaired in alcoholic patients.” (p. 815). States on p. 814 that it is a placebo intervention. Individual or group treatment: individual – “All subjects were instructed to work independently and not share answers with one another.” (p. 814) Exposure/intensity to treatment: 4 sessions. “Four 1‐hr workbook sessions were spread out over the final 2 weeks of inpatient treatment.” (p. 814) Duration of treatment: 2 weeks. “A four‐group, pretest‐posttest design was overlaid on an operating 28‐day, inpatient alcohol treatment program.” (p. 813). “Four 1‐hr workbook sessions were spread out over the final 2 weeks of inpatient treatment.” (p. 814) Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “All patients had been abstinent for at least 7 days and were medication‐free except for vitamins, common analgesics (e.g., acetaminophen), and some use of antihypertensives and anti‐inflammatories (for arthritis).” (p. 813) No‐treatment Comparison name: No remediation Description of intervention: no description of the control group – only pre‐ and post‐testing Exposure/intensity to treatment: no treatment Duration treatment: 2 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: “All patients had been abstinent for at least 7 days and were medication‐free except for vitamins, common analgesics (e.g., acetaminophen), and some use of antihypertensives and anti‐inflammatories (for arthritis).” (p. 813) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”Each participant was then randomly assigned to one of four treatment groups; subjects younger than 40 years and those 40 years of age and older were assigned separately to each group to balance for age both within and across groups.” (p. 814) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Attrition >15% (24%). No ITT used |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Rosa‐Alcatraz 2009.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 3 months Duration of participation (trial + follow‐up): 3 months + 12 months follow‐up Setting: outpatient Purpose of trial: “The aim of this study is to analyze the specific effects of the Intervention in Adolescents with Social Phobia (IAFS) program together with the nonspecific factors of the interventions used in the treatment of adolescents with social phobia” (p. 44) Closed/open placebo: closed placebo |
|
| Data |
Number of participants screened: 2650 Number of participants included: 77 Number of participants followed‐up at post‐treatment: 77 Number of participants randomly assigned to:
Number of withdrawals: n = 0 Diagnosis: Social Anxiety disorder (SAD) + Generalized social phobia (GAD) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision ( DSM‐IV‐TR) Means of assessment: Anxiety Disorders Interview Schedule for Children, 4th edition (ADIS‐IV‐C) Comorbidity: Panic Disorder (4/77), Agoraphobia (7/77), Selective mutism (2/77), Generalized Anxiety Disorder (12/77), obsessive compulsive disorder (OCD) (2/77), Obsessive–compulsive personality disorder (10/77), Specific phobia (37/77), Post‐traumatic Stress disorder (PTSD) (2/77), Dysthymia (11/77), Substance use disorder: alcohol (11/77), Substance use disorder: other substances (4/77) Age: 14.87 mean years (SD = 0.80). (range = 14 to 17) IQ: not stated Sex: 71.4% female Ethnicity: not stated Country: Spain Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Placebo group Description of intervention: “Control Group Placebo (…) in order to provide empirical evidence both regarding the role of information transmission and in relation to the extent to which the effects generated by the treatment are due to the so‐called "spontaneous remission", that is, to specific elements of the treatment or to non‐specific factors such as, for example, of the patient or therapist, expectations towards treatment or care and support provided by the therapist.” (translated, p. 45) “The group Placebo received information on adequate nutrition (2 sessions), consumption of psychoactive substances (4 sessions), hygiene (1 session), sports (2 sessions), AIDS prevention (2 sessions), prevention of unwanted pregnancies (1 session). It was controlled that in no case were they taught or indicated how they should or could act in the face of the problems from which they received information. After the presentation of the contents, the adolescents commented as a group (of about 6 subjects) and discussed the problems that were presented in real life on the different topics, reaching agreements that should be presented to the large group (the 18 subjects together to the therapist) before the end of the session. The treatments were applied by therapists with experience in the application of the IAFS and in the transmission of specific and qualified information.” (translated, p. 50) Individual or group treatment: group Exposure/intensity to treatment: 12 sessions, 90 minutes weekly Duration of treatment: 12 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: Wait‐list Description of intervention: “Control Group (…) Waiting List, in order to provide empirical evidence both regarding the role of information transmission and in relation to the extent to which the effects generated by the treatment are due to the so‐called "spontaneous remission", that is, to specific elements of the treatment or to non‐specific factors such as, for example, of the patient or therapist, expectations towards treatment or care and support provided by the therapist.” (translated, p. 50) Exposure/intensity to treatment: no treatment during waiting Duration treatment: 3 months Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: "77 adolescents were selected at random and randomly distributed between four experimental conditions: psychological treatment group (IAFS multi‐component package), transmission of information or educational group, placebo and waiting list control group" (p. 44) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:”The evaluation was carried out by three independent groups of collaborators coordinated by the third author, previously trained for this purpose. The first group performed the preliminary evaluation and follow‐up at twelve months. The second, the posttest and the third, the follow‐ up at six months. The team was only aware of all the data referring to the different measures when the collection of information related to the second follow‐up ended.” (translated from p. 47) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Attrition <15% (0%, All patients completed posttreatment) |
| Selective outcome reported | Unclear | SEJ2004‐01471/PSIC Protocol: Olivares 2005: Programa IAFS. Protocolo para el tratamiento de la fobia social en adolescentes. Not able to locate trial registry or published protocol |
| Other sources of bias | Yes | No other sources found |
Rosen 1976.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 8 weeks Duration of participation (trial + follow‐up): 8 weeks + 2 months follow‐up Setting: outpatient Purpose of trial: “The present study evaluates the clinical efficacy of self‐administered desensitization in the context of a controlled outcome study” (p. 209) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 55 Number of participants followed‐up at post‐treatment: 43 Number of participants randomly assigned to
Number of withdrawals: n = 12
Diagnosis: specific anxiety (snakes) Diagnostic manual: not stated Means of assessment: self‐referred, scored high on Snake Attitude Questionnaire (SNAQ) and Fear Survey Schedule (FSS) Comorbidity: not stated Age: 33.5 mean years IQ: not stated Sex: 92.7% female Ethnicity: not stated Country: USA Inclusion criteria Subjects were self‐referred snake phobics who:
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Placebo control Description of intervention: “To control for initial therapeutic expectancies and other nonspecific treatment factors, a totally self‐administered bibliotherapy placebo called systematic relearning was included in the present study. The basic rationale of the treatment program was that people could substantially reduce their fears by replacing inaccurate perceptions with more accurate information about the feared object. To accomplish this goal, each individual studies a manual that organizes factual information about snakes into 10 chapters. Each chapter contains questions at the end to help subjects assess their mastery of the materials. As subjects work on the program they construct an "information hierarchy" from the relevant information in each chapter. Because of recent findings that question the adequacy of many placebo procedures (...), a number of steps were taken to assure adequate experimental control over subjects' expectancies. In addition, possible therapist expectancy effects were avoided by sending all self‐instructional materials through the mail. In effect, systematic relearning and self‐administered desensitization as previously described were administered under double‐blind conditions. “ (p. 210‐11) Individual or group treatment: individual Exposure/intensity to treatment: generally twice weekly Duration of treatment: up to 8 weeks Concomitant psychotherapy: were not currently receiving treatment for their phobia Concomitant pharmacotherapy: were not currently receiving treatment for their phobia Wait‐list Comparison name: No‐treatment (in reality wait‐list) Description of intervention: “No‐treatment control. Subjects in this group were informed that the large number of clients in the project necessitated a delay in treatment for some individuals. After posttesting, untreated controls were offered treatment.” (p. 211) Exposure/intensity to treatment: no treatment Duration treatment: up to 8 weeks Concomitant psychotherapy: were not currently receiving treatment for their phobia Concomitant pharmacotherapy: were not currently receiving treatment for their phobia |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote:”After pretreatment assessments had been completed, the subjects were matched on behavior approach scores and assigned by block randomization to one of four treatment groups or a no‐ treatment control.” (p. 210) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:”Pretreatment to posttreatment and follow‐up assessments of subjects' attitudes and reactions toward snakes were conducted by assistants blind to subjects' group assignment” (p. 209) |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | No | Quote:“Two subjects in each of the therapist‐aided treatment groups dropped out of their programs during the first week of therapy. Three self‐administered desensitization subjects, 1 placebo control, and 4 untreated controls could not be reached at time of posttesting primarily because of address changes. Accordingly, a final N of 43 was achieved, and group sizes were not equal (…)” (p. 211) Attrition >15% (38%). No ITT. Only reports data on completers. Very low sample size |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Roth 1964.
| Study characteristics | ||
| Methods |
Parallel randomised trial with six arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 4 weeks Duration of participation (trial + follow‐up): 4 weeks + 6 months follow‐up Setting: outpatient Purpose of trial: “The broad aim of the study was to determine some of the early effects of an ataractic agent, chlordiazepoxide, on anxiety and tension..” (p. 257) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 311
Number of participants included: 181
Number of participants followed‐up at post‐treatment: 150
Number of participants randomly assigned to
Number of withdrawals: n = 31
Diagnosis: Psychiatric outpatient, different diagnoses. “The therapists’ initial diagnosis classified 42 per cent of the total sample as Neurotics, 26 per cent as Personality Disorders, 15 per cent as Psychophysiologic Disorders, and 18 per cent as Psychotics. While there were some differences between treatment groups, these proved to be non‐significant. The mean global severity of illness rating for the sample was “Moderately Ill”.” p. 262. Diagnostic manual: not stated Means of assessment: intake interviews (clinical structured interview) Comorbidity: different disorders Age: 37.8 mean years IQ: not stated Sex: 100% male Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: Placebo
Description of intervention: ”The study medication which consisted of chlordiazepoxide (Librium) in 10 mg capsules or placebo were prescribed in the following manner. During the first week all patients scheduled for medication received a total daily dosage of four capsules. In the second week the physician prescribed four capsules if adjustment was not required. When required the dosage could be lowered to three or increased to five or six capsules per day. At the beginning of the third week, dosage could be lowered to two capsules or increased to eight capsules per day.
This final dosage remained fixed for a given patient for the last two weeks.” (p. 260)
Individual or group treatment: individual.
Exposure/intensity to treatment: between 2 and 8 capsules per day
Duration of treatment: 4 weeks
Concomitant psychotherapy: half of the patients started psychotherapy concomitantly with the medication treatment
Concomitant pharmacotherapy: not allowed Wait‐list Comparison name: Wait group Description of intervention: “All patients completed an inventory on the same day or within a week of initiation of treatment, but always before the first treatment. They were administered a IO‐mm adjective rating scale and completed a global improvement rating just before the second, third and fourth treatment visits to the therapist or prescribing physician. Just before the fifth treatment, each patient was re‐examined on a modified inventory. Since the Wait Group received no treatment for four weeks, initial testing was completed early. The adjective rating scale was given only once, a week after the initial inventory was completed. Wait Group cases then waited three weeks for a final re‐testing, given prior to the initial psychotherapeutic interview. The treatment groups did not differ on the number of days between their initial and final tests” (p. 260) Exposure/intensity to treatment: no pharmacological treatment. Some patients received psychotherapy. Duration treatment: 4 weeks Concomitant psychotherapy: half of the patients started psychotherapy. Concomitant pharmacotherapy: not allowed |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors A four‐week double‐blind study was made of the effects of chlordiazepoxide on a group of 150 male outpatients newly accepted for individual psychotherapy. Three major hypotheses were tested. The findings with respect to patient criteria were:
Key limitations from study authors
Other notes from the review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Patients were randomly assigned to six treatment groups receiving the combinations of psychotherapy and medication treatment (p. 283) |
| Allocation concealment | Yes | Quote:“Each clinic pharmacist dispensed the medication, which was delivered in individual bottles containing 250 capsules identical in size, color, appearance, and taste. Each bottle was labeled with the unique code number assigned to the patient. The identification label of the study medication sealed in small envelopes was placed in the hands of the pharmacist. Codes were not broken until after completion of the study.” (p. 260) |
| Blinding of outcome assessors | Yes | The therapist were outcome assessors‐ and were blinded. Quote:“Although therapists were eventually permitted to ascertain whether their patient had initially been on active drug or placebo, this was not allowed until many months after the medication was administered.” (p. 284) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Attrition >15% (17.1%) Only reports data on completers. Eliminates patients to ensure equal groups |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Rupert 1978.
| Study characteristics | ||
| Methods |
Parallel randomised trial with seven arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 4 to 7 days Duration of participation (trial + follow‐up): 4 to 7 days. No follow‐up Setting: inpatient Purpose of trial: “The present experiment examined the effects of multiple sessions of heart rate biofeedback training on the heart rate control and anxiety levels of anxious psychiatric patients.” (p. 583) Open/Closed placebo: closed |
|
| Data |
Number of participants screened: not stated
Number of participants included: 56
Number of participants followed‐up at post‐treatment: not stated
Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: anxious psychiatric patients Diagnostic manual: not stated Means of assessment: not stated Comorbidity: not stated Age: (range 19 to 55) IQ: not stated Sex: 100% male Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo 1 and Physical placebo 2
Treatment name: Placebo biofeedback
Description of intervention: “The other meter was constructed to give placebo (false positive) feedback and was a voltmeter wired in a circuit that generated a positive or negative signal. This signal was used to give the needle on the meter on the appropriate direction to indicate a steadily increasing or decreasing heart rate. The meters were either covered or uncovered depending on whether the subjects were in a biofeedback (true or placebo) or a no‐biofeedback condition.” (p. 584)
“Subjects in the true biofeedback and placebo biofeedback conditions were also told that the meter in front of them would give them information about changes in their heart rates and that they were to use this information to help them control their heart rates. It was explained that movements of the needle on the meter to the right and left reflected increases and decreases in their heart rates, respectively.” (p. 585)
Individual or group treatment: individual
Exposure/intensity to treatment: “All subjects participated in 4 expediential sessions which were conducted within a period of 4 to 7 days. Each session included a total of 25 min of heart rate training (or recording time) plus time required for directions, acquisition of initial heart rate levels, and rest periods.” (p. 584)
Duration of treatment: 4 to 7 days
Concomitant psychotherapy: not stated
Concomitant pharmacotherapy: no to little medication No‐treatment Comparison name: No‐treatment Description of intervention: assessed before and after treatment Exposure/intensity to treatment: no treatment Duration treatment: 4 to 7 days Concomitant psychotherapy: not stated Concomitant pharmacotherapy: no to little medication |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: "Subjects were randomly assigned in equal numbers to the six experimental conditions and the no‐treatment control condition." (p. 584) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | No information |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Shalev 2012.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 12 weeks of treatment and post treatment data at 5 months. Duration of participation (trial + follow‐up): 12 weeks of treatment + post treatment data at 5 month + follow‐up at 9 months. Setting: outpatient Purpose of trial: “To compare early and delayed exposure based, cognitive, and pharmacological interventions for preventing PTSD.” (p. 166) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 5286 Number of participants included: 242 Number of participants followed‐up at post‐treatment: 207 Number of participants randomly assigned to:
Number of withdrawals: n = 35
Diagnosis: post‐traumatic stress‐disorder (PTSD) or acute stress disorder (ASD) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV). Means of assessment: The PTSD Symptom Scale‐Interviewer Version (PSS‐I) and the ASD Scale (ASDS) Comorbidity: not stated Age: Pharmacological placebo: 36.26 mean years (SD = 12.39), wait‐list: 37.28 mean years (SD = 11.91) IQ: not stated Sex: not stated Ethnicity: not stated Country: Israel Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo Treatment name: Placebo Description of intervention: “Concealed tablets of either 10 mg escitalopram or placebo were prepared and coded by Lundbeck Pharmaceuticals (Copenhagen, Denmark) and were supplied to clinicians by a research associate. An initial dose of 1 tablet daily was increased to 2 tablets after 2 weeks of treatment. Trained psychiatrists provided 4 weekly sessions (weeks 1‐4) followed by 4 biweekly sessions (weeks 6‐12)." (p. 168) Individual or group treatment: individual Exposure/intensity to treatment: 1 tablet daily, increased to 2 tablets daily after 2 weeks of treatment Duration of treatment: 12 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: Waiting list Description of intervention: “The WL participants who met PTSD diagnostic criteria at 5 months received PE at that time (hereafter referred to as delayed PE.” (p. 167). “A telephone interview briefly contacted participant on the WL every 2 weeks to inquire about emerging needs or possible emergencies. These calls did not contain elements of PE or CT.” (p. 169) Exposure/intensity to treatment: no treatment Duration treatment: 12 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors 1. ”The results of our study show that there are significant and similar preventive effects of PE and CT.” (p. 174) 2. “The escitalopram subgroup did not differ from the placebo subgroup or the WL group at 5 months; however, the escitalopram subgroup fared worse than all the other groups at 9 months.” (p. 174) 3. “Delaying PE did not affect the 9‐month outcome (…) Our finding suggests that delaying the intervention does not increase the risk of chronic PTSD. Delaying treatment somewhat reduced the number of treatment candidates: about a third of those with initial PTSD recovered by 5 months (…) Thus, a delayed intervention is an acceptable option when early clinical interventions cannot be provided (e.g. during wars, disasters, or continuous hostilities” (p. 174) Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | No | Quote:“The equipoise‐stratified randomization is a method for randomly allocating participant to interventions in treatment studies that include more than two arms (…) In our study, participants who agreed to start treatment (n=269) were informed about the 4 treatment options (PE, CT, treatment with SSRI vs placebo, and WL and subsequent delayed PE) could decline up to 2 treatment options (including the WL for delayed PE), and were randomly assigned to the remaining treatment option.” (P. 168) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:”The clinical assessments were made by clinical psychology interns (…) They remained blind to treatment attendance and adherence.” (p. 167) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Quote:“To account for missing observations and the groups’ heterogeneities, we used a linear mixed model with covariance for significant initial group differences and for the time lag between the traumatic event of each participant and subsequent assessment.” (p. 171) Attrition <15% (PE: 11%, CT:17,5%, SSRI: 9%, Placebo: 22%. Waitlist, 15% |
| Selective outcome reported | Yes |
NCT00146900 No apparent differences in reporting between trial registry and full report |
| Other sources of bias | Yes | No other sources found |
Shealy 1979.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: NS Cluster randomised: (yes/no): no Duration of trial (baseline to post): 6 weeks of treatment Duration of participation (trial + follow‐up): 6 weeks treatment + 6 months follow‐up Setting: outpatient (college undergraduates) Purpose of trial: “There has been a relatively recent effort to develop behavioral treatments for sleep‐onset insomnia. The present research was designed as an extension of this previous work in order to examine four current methodological and theoretical issues” (p. 541) Closed/open: closed |
|
| Data |
Number of participants screened: not stated Number of participants included: 70 Number of participants followed‐up at post‐treatment: not stated Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: sleep‐wake disorder (insomnia) Diagnostic manual: not stated, but probably due to information provided: “Insomnia was considered mild if there was a sleep onset latency greater than 30 min present at least three nights per week and moderate if there was a sleep onset latency greater than 45 min present at least four nights per week. In both instances. a subjective indication of difficulty in falling asleep was also required, Three levels of insomnia duration were also examined: 3‐11 months, 11 years, and greater than 4 years. However, only mild insomniacs were eventually classified on the duration factor.” (p. 542) Means of assessment: not stated Comorbidity: not stated Age: 19.4 mean years (range =17 to 30) IQ: not stated, but all college students Sex: 100% female Ethnicity: not stated Countr:: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Placebo Description of intervention: “Insomniacs in this group also lay on their backs with heads on pillows in a dimly lighted room. Topics related to sleep such as sleep disturbances and dreams were discussed. There was no discussion on how to resolve sleep disturbances. It was assumed that this treatment would have little therapeutic value. Insomniacs in this group completed the DSQ every morning throughout the study.” (p. 543) Individual or group treatment: not stated, but seems like a group Exposure/intensity to treatment: self‐monitoring for 2 weeks assessing own sleeping patterns + two one‐half hours conducted for 3 weeks + 1 week of positive demand Duration of treatment: 6 weeks Concomitant psychotherapy: not allowed Concomitant pharmacotherapy: no hypnotic drugs Wait‐list Comparison name: Waiting list Description of intervention: “Subjects in these groups were told that all treatment groups were filled and that they would receive treatment as soon as possible. (…) The insomniacs in the waiting list group were required to fill out only two questionnaires‐ one at pretherapy and one at the end of the positive demand period.” (p. 543) Exposure/intensity to treatment: waiting for treatment Duration treatment: 6 weeks Concomitant psychotherapy: not allowed Concomitant pharmacotherapy: no hypnotic drugs |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from studyauthors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote:"First, insomniacs agreeing to participate were randomly assigned to the five groups on the basis of their answers to the pretherapy questionnaire. Group assignment occurred before baseline. because self‐monitoring itself was an independent variable. Each group consisted of nine mild (three of each duration) and five moderate insomniacs." (p. 542) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Sibilio 1957.
| Study characteristics | ||
| Methods |
Parallel randomised cross‐over trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 28 days Duration of participation (trial + follow‐up): 28 days + 3 months of other cross‐over phases Setting: inpatient Purpose of trial : “This paper gives the experimental design for the study and includes a report of the effects of promazine on the behavioral adjustment of patients. Future papers will report the effects of promazine on attention, as measured by the reaction‐time test, and on daily ward behavior, blood pressure, blood counts, and bone marrow.” (p. 419) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated
Number of participants included: 93
Number of participants followed‐up at post‐treatment: not stated
Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: chronic schizophrenia Diagnostic manual: not stated Means of assessment: not stated Comorbidity: not stated Age: (range = 32 to 61 years) IQ: all were considered to be of at least average intelligence (clinically determined) Sex: 100% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: Placebo
Description of intervention: “The pharmacologically inactive placebo was in every respect similar to the drug in appearance.” (p. 421)
Individual or group treatment: individual
Exposure/intensity to treatment: 4 times daily
Duration of treatment: 28 days
Concomitant psychotherapy: not allowed
Concomitant pharmacotherapy: not stated No‐treatment Comparison name: no treatment Description of intervention: not stated Exposure/intensity to treatment: no pill Duration of treatment: 28 days Concomitant psychotherapy: not allowed Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote:”To arrive at the research population for the study, each patient was randomly assigned to one of three groups in a manner which assured equivalence of the groups with respect to scores obtained on a behavioral adjustment rating scale.” (p. 419) |
| Allocation concealment | Yes | Quote:”The double‐blind technique was employed throughout the study. Patients received their medication (drug or placebo) in individual, sealed envelopes. Neither the patient nor the attendant dispensing the envelopes was cognizant of the kind and amount of medication contained in each envelope.” (p. 420‐1) |
| Blinding of outcome assessors | Yes | Quote:”Those attendants who rated the patients' behavioral adjustment did not dispense medication and were unaware of the experimental group to which a patient was assigned.” (p. 421) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Sommerness 1955.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 12 weeks Duration of participation (trial + follow‐up): 12 weeks treatment + 4 weeks follow‐up Setting: inpatient (hospital) Purpose of trial: “The effect of suggestion must be measured as well as the effect of an intercession of a break in ward routine when one is attempting to evaluate a drug. For these reasons, it was felt that experiments with the drug, placebo, and control groups must be carried out simultaneously in order to obtain reliable scientific results.” (p. 316) Closed/open placebo: closed placebo |
|
| Data |
Number of participants screened: 2000‐bed hospital Number of participants included: 90 Number of participants followed‐up at post‐treatment: not stated Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: chronic mental illness Diagnostic manual: not stated Means of assessment: “Patients were rated once every two weeks during the 20‐week period by two psychiatric aides independently, using the L‐M Fergus Falls Behavior Rating Scale” (p. 317) Comorbidity: not stated Age: not stated IQ: not stated Sex: 100% male Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo Treatment name: Placebo Description of intervention: “The second group was given an identical‐appearing placebo under identical conditions.” (p. 316) Individual or group treatment: individual Exposure/intensity to treatment: not stated Duration of treatment: 12 weeks Concomitant psychotherapy: “The usual hospital routine was continued on the ward. Other therapies were neither increased nor decreased during the period of the experiment” (p. 317) Concomitant pharmacotherapy: “The usual hospital routine was continued on the ward. Other therapies were neither increased nor decreased during the period of the experiment” (p. 317) No‐treatment Comparison name: No‐treated control group Description of intervention: “The third group received neither reserpine nor placebo but was otherwise under identical conditions.” (p. 316) Exposure/intensity to treatment: no treatment Duration of treatment: 12 weeks Concomitant psychotherapy: “The usual hospital routine was continued on the ward. Other therapies were neither increased nor decreased during the period of the experiment” (p. 317) Concomitant pharmacotherapy: “The usual hospital routine was continued on the ward. Other therapies were neither increased nor decreased during the period of the experiment” (p. 317) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote:”They were divided into three groups by a random numbers table. This randomization resulted in essential equality for diagnosis, behavior, weight, and blood pressure.” (p. 316) |
| Allocation concealment | Yes | Quote:"The hospital pharmacist alone knew which group received reserpine or placebo. This information was not available to the other experimenters until a complete analysis of the results of the experiment had been made." (p. 316) |
| Blinding of outcome assessors | Yes | See above |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Steinmark 1974.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 4 weeks Duration of participation (trial + follow‐up): 4 weeks + 5 months follow‐up Setting: outpatient (psychology students) Purpose of trial: “The present study was designed to critically test the demand and placebo interpretations of outcome improvement among subjects trained in relaxation and to evaluate whether any additional benefit would be achieved by the use of single‐item desensitization.” (p. 157‐8) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 519 Number of participants included: 52 Number of participants followed‐up at post‐treatment: 48 Number of participants randomly assigned to:
Number of withdrawals : n = 4
Diagnosis: sleep‐wake disorder Diagnostic manual: not stated Means of assessment: brief questionnaire on sleep behaviour Comorbidity: not stated Age: not stated, but psychology students IQ: not stated, but psychology students Sex: not stated Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name : Quasi‐desensitization placebo Description of intervention: “The placebo condition involved a quasi‐desensitization procedure. During Session 1 each subject constructed an IS‐item hierarchy of chronological bedtime activities and chose six neutral images to be paired with the hierarchy items and to be used as substitutes for relaxation. Viewing sleep disturbance as a problem in which bedtime stimuli elicit responses (physiological and/or cognitive) incompatible with sheep, then the imaginal pairing of such stimuli with varied, neutral images should not theoretically change that functional relationship. In Sessions 2, 3,and 4, each item was presented six times with intervening presentations of neutral images. The subjects in this condition were told to practice hierarchy and neutral image visualizations twice a day, the Iasi practice being at least two hours prior to retiring. The hatter instruction was included to insure that practice would not increase sleep disturbance.” (p. 159) Individual or group treatment: group Exposure/intensity to treatment: 4 sessions – 1 weekly Duration of treatment: 4 weeks Concomitant psychotherapy: not allowed Concomitant pharmacotherapy: not allowed Wait‐list Comparison name: Waiting list no‐treatment (wait‐list) Description of intervention: not stated Exposure/intensity to treatment: no treatment during waiting Duration of treatment: 4 weeks Concomitant psychotherapy: not allowed Concomitant pharmacotherapy: not allowed |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote:”The subjects were ranked on latency of sleep onset obtained in the pretreatment interview and were randomly assigned within severity blocks to one of four treatment conditions” (p. 159) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Attrition <15% (7.7%). One from each condition. Only reports data on completers |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | No | Quote:"The first author served as a therapist.” (p.158) |
Szymanski 1995.
| Study characteristics | ||
| Methods |
Parallel randomised trial with six arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 2 weeks Duration of participation (trial + follow‐up): 2 weeks. No follow‐up Setting: outpatient (college) Purpose of trial: “The first hypothesis was concerned with whether cognitive restructuring is an effective intervention with spider phobics. The second hypothesis was whether the presence of anxiety in subjects while learning the cognitive restructuring techniques would be more effective than teaching the techniques while the subjects were in a neutral emotional state.” “Finally, it was hypothesized that the addition of a cognitive intervention to a behavioral intervention would be more effective than the behavioral intervention alone” (p.134‐35) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 203 Number of participants included: 32 Number of participants follow‐up at post‐treatment: 32 Number of participants randomly assigned to:
Number of withdrawals: n = 0 Diagnosis: specific phobia (snake) Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd, Revised (DSM‐III‐R) Means of assessment: Clinical interview Comorbidity: not stated Age: 18.4 mean years IQ: not stated, but college students Sex: 71.9% female Ethnicity : not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Spider facts placebo Description of intervention: “This condition consisted of a spider facts lecture. It was included to test whether the content of the cognitive restructuring conditions was responsible for the treatment effects. In a related vein, the credibility of the placebo groups (as well as the cognitive restructuring groups)was monitored.” (p. 139) Individual or group treatment: group Exposure/intensity to treatment: “subjects were asked to participate in three, approximately one‐hour group sessions over a two‐week period.” (p. 139) Duration of treatment: 2 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy:: not stated No‐treatment Comparison name: No treatment Description of intervention: “Subjects in the no‐treatment control condition, the sixth condition, only participated in the two assessment sessions (i.e., pre‐ and post‐tests) at a similar interval to the experimental conditions.” (p. 139) Exposure/intensity to treatment: no treatment Duration of treatment: 2 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect outcome:
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: one found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: "Subjects who were identified as snake phobics were then randomly assigned to one of the six conditions." (p. 139) The groups were each given a number 1‐5. After each subject completed the screening they were then added to the next group. So the first subject went into group 1, the second subject went into group 2, etc. We did not try to match or even review information from their screen. ( Szymanski 1995 (pers comm) ) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote:“As they signed up they were randomly assigned to conditions by a research assistant who was blind to the conditions.” ( Szymanski 1995 (pers comm) ) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | No | Quote:“Due to the high levels of attrition it was important to determine whether the subjects who dropped out of the experiment were significantly different from those who remained in the study. Using two tailed t‐tests we found that subjects who had initially agreed to continue in the study but subsequently dropped out did not have significantly different pre‐test scores from subjects who completed the study.” (p.144) Attrition unclear. Unclear number of randomised participants. The authors report themselves high levels of attrition |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Tan 1986.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 8 weeks Duration of participation (trial + follow‐up): 8 weeks treatment + 4 month follow‐up Setting: outpatient Purpose of trial: “The present study evaluated the efficacy of group cognitive‐behavior therapy for the alleviation of psychosocial problems and reduction of seizures with adult epileptic patients”. (p.225) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 30 Number of participants followed‐up at post‐treatment: 27 Number of participants randomly assigned to:
Number of withdrawals: n = 3
Diagnosis: depression/anxiety Diagnostic manual: not stated, but were referred by a neurologic clinician Means of assessment: clinical interview ‐ The Minnesota Multiphasic Personality Inventory (MMPI), Beck Depression Inventory (BDI) Comorbidity: epilepsy Age: 33.4 mean years (SD = 11.1) IQ: not stated, but mentally retarded was excluded Sex: 63% female Ethnicity: not stated Country: Canada Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Supportive care Description of intervention: “The SC group as an attention‐placebo control group also received a total of eight 2‐h sessions of group counselling or discussion that was mainly supportive in nature. Techniques such as reflection and clarification of feelings were used, but no specific cognitive‐behavioral strategies were taught. This SC group intervention was meant to provide the “nonspecific” factors of any group psychological intervention or therapy, such as therapist attention, suggestion, group support,” (p.227) Individual or group treatment: group Exposure/intensity to treatment: 8 x 2 hours sessions Duration of treatment: 8 weeks Concomitant psychotherapy: 60% received concomitant therapy. “Six patients in each of the three groups did not receive any concomitant or other professional counselling or psychiatric treatment during the present study. The remaining patients received treatment from other therapists while participating in the present study, but such ongoing treatment could not be terminated for obvious ethical reasons” (p. 227) Concomitant pharmacotherapy: all patients received anticonvulsant medication for their epilepsy Wait‐list Comparison name: Wait‐list Description of intervention: “The WL group did not receive any group therapy until after the present study was completed. Patients assigned to this group were seen for the three assessments at about the time the CBT and SC groups had them (i.e., before therapy or pre, after therapy or post, and at a 4‐month follow‐up)” (p. 227) Exposure/intensity to treatment: waiting for therapy Duration of treatment: 8 weeks Concomitant psychotherapy: 60% received concomitant therapy. “Six patients in each of the three groups did not receive any concomitant or other professional counselling or psychiatric treatment during the present study. The remaining patients received treatment from other therapists while participating in the present study, but such ongoing treatment could not be terminated for obvious ethical reasons” (p. 227) Concomitant pharmacotherapy: all patients received anticonvulsant medication for their epilepsy |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: one found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | "Quote ‐ Twenty‐seven outpatients were randomly assigned to one of three groups: Cognitive‐Behavior Therapy, Supportive Counseling (attention‐placebo control), and Waiting list (no treatment control)". (p. 225) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Attrition <15% (10%). No ITT. On reports data on completers |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Teri 1997.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 9 weeks of treatment Duration of participation (trial + follow‐up): 9 weeks of treatment, follow‐up at 6 months. Setting: outpatient Purpose of trial: “The current study is a controlled clinical investigation of two non‐pharmacological treatments of depression in patients with Alzheimer’s disease. Two active behavioral treatments, one emphasizing patient pleasant events and one emphasizing caregiver problem solving, were compared to an equal‐duration typical care condition and a wait list control.” (p. 159) |
|
| Data |
Number of participants screened: not stated Number of participants included: 88 Number of participants followed‐up at post‐treatment: 72 Number of participants randomly assigned to:
Number of withdrawals : n = 16
Diagnosis: depression (in dementia patients) ‐ 75% were diagnosed with major depressive disorder, 25% were diagnosed with minor depressive disorder Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition, Revised (DSM‐III‐R) Means of assessment: clinical interview – Schedule for Affective Disorders and Schizophrenia Comorbidity: dementia Age: 76.4 mean years (SD = 8.2) IQ: not stated Sex: 47% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Usual care Treatment name: typical care control (TCC) Description of intervention: “Subjects in this condition were given information, advice, and support with their efforts to manage patient problems. No specified homework assignments or record keeping were provided. Specific problem solving or behavioral strategies were not implemented. Therapists have suggestions and advice of an unstructured nature.” (p. 161) Individual or group treatment: individual Exposure/intensity to treatment: one 60‐minute session per week. Duration of treatment: 9 weeks Concomitant psychotherapy: allowed to receive other treatment Concomitant pharmacotherapy: allowed to receive other treatment No‐treatment Comparison name: Wait list control (in reality no‐treatment) Description of intervention: “Subjects in this condition received no contact with therapists. Following assignment, they were informed that they would receive no active intervention during the 9‐week period. Immediately following the 9 weeks, they were post‐tested.” (p. 161) Exposure/intensity to treatment: no treatment Duration treatment: 9 weeks Concomitant psychotherapy: aAllowed to receive other treatment Concomitant pharmacotherapy: allowed to receive other treatment |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Yes | Quote: “Subjects were randomly assigned to one of four treatment conditions: behavior therapy‐pleasant events (BT‐PE); behavior therapy‐problem solving (BT‐PS); typical care control (TCC); and wait list control (WLC).” (p. 160) “Conducted independently by statistician using computer program” ( Teri 1997 (pers comm) ) |
| Allocation concealment | Yes | Quote: "Randomisation concealed from the researchers" ( Teri 1997 (pers comm) ) |
| Blinding of outcome assessors | Yes | Quote: “(…) assessed at pre‐, post‐, and 6‐ month follow‐up intervals by interviewers blind to treatment assignment.” (p. 160) Quote: “Interviews were conducted by experienced master’s‐ and PhD‐level clinical geriatric interviewers, blind to treatment condition.” (p. 160) |
| Blinding of participants and personnel | No | Not possible to blind usual care and wait‐list |
| Incomplete outcome data | No | Quote: “Eighty‐eight patient‐ caregiver pairs began the study; 72 (82%) completed the pretest, 9‐week intervention, and posttest. Subjects who discontinued treatment did so for the following reasons: serious medical illness (n = 4), change in living situation (n = 4), exclusionary medication prescribed during the intervention stage (n = 2), and caregiver stopped participating (n = 6). No significant differences were obtained on baseline measures between subjects who did and did not discontinue treatment.” (p. 160) Attrition >15% (18.2%). No ITT used. Reports data on completers only |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | Yes | No other sources found |
Tori 1973.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 2 weeks Duration of participation (trial + follow‐up): 2 weeks treatment + follow‐up at 30 days. Setting: outpatient Purpose of trial: “The present study was designed in order that comparisons could be made between counterconditioning, expectancy, and cognitive‐coping variables in the reduction of the fear and anxiety of snake‐phobic college students.” (p. 270) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: initial pool of 700 undergraduate students
Number of participants included: 47
Number of participants followed‐up at post‐treatment: not stated
Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: specific anxiety (snakes) Diagnostic manual: not stated Means of assessment: self‐rating on a 5‐point scale + approach test (excluded if they went beyond Point 8) Comorbidity: not stated Age: not stated IQ: not stated, college students Sex: 70.2% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo Treatment name: High expectancy placebo Description of intervention: “Electrodes were then attached to the subject’s wrists, left ankle, and left index finger. A polygraph and several other impressive but nonfunctional machines (all with flashing lights or moving dial indicators) were activated. Deep muscle relaxation was then induced, following the principles of progressive relaxation (...). The subjects then spent about 10 minutes imagining psychologically pleasant scenes, with the entire session lasting approximately 40 minutes” (p. 272) Individual or group treatment : individual Exposure/intensity to treatment: “Days spent in treatment were held constant by having the subjects meet either on Monday and Wednesday or Tuesday and Thursday for a two‐week period. (...) Each subject served individually in four treatment sessions lasting from 30 to 50 minutes each.” (p. 271) Duration of treatment: 2 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name: No‐treatment control Description of intervention: “The pretest, posttest, and follow‐up batteries were administered to these subjects (n=10, 7 females and 3 males) in order to determine the amount of improvement due to repeated exposure to the snakes and to control for any nonspecific changes that might occur over the time of the experiment.” (p. 271) Exposure/intensity to treatment : no treatment Duration of treatment: 2 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”The first 40 subjects were randomly assigned to the five groups employed in the experiment. In order to more closely equate groups for initial snake approach behavior, the last 7 subjects were assigned on the basis of their pretest approach scores.” (p. 271) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: ”The female experimenter who conducted post‐testing and follow‐up testing was not aware of the subjects’ treatment conditions.” (p. 273) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | No information |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Trexler 1972.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 1 week (4 sessions spaced several days apart) Duration of participation (trial + follow‐up): 1 week. No follow‐up Setting: outpatient (University) Purpose of trial: “In a partial replication and refinement of an earlier study by the authors, 33 college student volunteers reporting high levels of public‐speaking anxiety received rational‐emotive therapy (RET), attention placebo (AP), or no treatment (NT).” (p. 60) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 33 Number of participants followed‐up: not stated Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: specific anxiety (public‐speaking anxiety) Diagnostic manual: not stated Means of assessment: level 4 on a 10‐point behaviorally anchored rating scale of anxiety Comorbidity: not stated Age: not stated, undergraduates IQ: not stated ‐ but college students Sex: 51.5% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Attention placebo Description of intervention: ”AP treatment consisted mainly of the typical training in relaxation employed in systematic desensitization studies, without presentation of stimulus hierarchies. The expectancy was communicated continually that this procedure was a well‐regarded treatment for general anxiety and as such would be helpful in overcoming public‐speaking anxiety. Training was effected both with a tape recording and viva voce. Homework assignments for all sessions consisted of practicing relaxation skills and reading specially prepared materials (...), emphasizing both the purpose and techniques of relaxation.” (p. 61) Individual or group treatment: group Exposure/intensity to treatment: 4 sessions spaced several days apart Duration of treatment: 1 week Concomitant psychotherapy:: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: No‐treatment (in reality wait‐list) Description of intervention: “Those in the NT group were informed that there would be a short delay before they would begin therapy” (p. 61) Exposure/intensity to treatment: no treatment during waiting Duration of treatment: 1 week Concomitant psychotherapy : not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: "Treatment groups were matched approximately on the basis of the preliminary self‐ rating and randomly assigned to treatment conditions at the initial speech session" (p. 61) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: ”Pretherapy and posttherapy evaluations were made by a graduate student and an advanced undergraduate psychology honors student trained in observation prior to the study and kept blind as to 5s' treatment.” (p. 60) |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no treatment" |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | No | The first author served as therapist |
Turner 1979.
| Study characteristics | ||
| Methods |
Parallel randomised trial with five arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 4 weeks Duration of participation (trial + follow‐up): 4 weeks. No follow‐up Setting: outpatient Purpose of trial: “In light of these criticisms, the present experiment incorporates appropriate modifications in contrasting the effectiveness of stimulus control with progressive relaxation in ameliorating the complaints of severe insomniacs. A second purpose of the present study is to provide an initial experimental investigation of the efficacy of paradoxical intention for the treatment of insomnia and to compare this technique with the two more conventional procedures for reducing disorders of sleep (i.e., stimulus control and progressive relaxation)” (p. 501) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 115 Number of participants included: 50 Number of participants followed‐up: 50 Number of participants randomly assigned to:
Number of withdrawals : n = 0 Diagnosis: sleep‐wake disorder Diagnostic manual: not stated Duration of treatment: 1 week Means of assessment: Clinical interview (Monroe's Daily Sleep Questionnaire) Comorbidity: no physiological or psychological complaints Age: 39 mean years (range = 24 to 79) IQ: not stated Sex: 50% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Placebo control Description of intervention: “The placebo condition used was that described by Steinmark 1974 . Their "quasi‐desensitization" condition required clients to construct an individualized 18‐item hierarchy of chronological bedtime activities to be paired with six neutral images. During the first session clients were taught how to construct the hierarchy and develop six neutral images. Sessions 2, 3, and 4 consisted of imagining the hierarchies and alternating neutral images between scenes. Clients were instructed to practice the procedure twice a day, the last practice not being within 2 hours of bedtime.” (p. 504) Individual or group treatment: individual Exposure/intensity to treatment: 30 to 45 minutes once per week Duration of treatment: 4 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated Wait‐list Comparison name: Wait‐list Description of intervention:“The no‐treatment control clients were asked to forego treatment for 5 weeks and to serve as control subjects. These subjects were assured of receiving treatment in 4 weeks” (p. 504) Exposure/intensity to treatment: Waiting for treatment Duration treatment: 4 to 5 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: "Following the 10‐day baseline period, 10 clients were randomly assigned to each of the five treatments." (p. 502) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and "no‐treatment" |
| Incomplete outcome data | Yes | Attrition <15% (0%). |
| Selective outcome reported | Unclear | Protocol not found |
| Other sources of bias | No | Quote: “the first author served as the therapist in all conditions.” (p. 502‐3) |
Vanderplate 1983.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: yes Cluster randomised: not stated Duration of trial (baseline to post): for EMG‐groups: 3 weeks Duration of participation (trial + follow‐up): 3 weeks of treatment +2 months follow‐up Setting: outpatient Purpose of trial: “The purpose of the present study was to assess the efficacy of frontalis EMG biofeedback as a treatment for insomnia within a more adequately designed and rigorously controlled investigation. No previous study in this area has controlled for the reactive effects of sleep behavior self‐monitoring, or employed a pseudo‐treatment control group sufficiently equivalent on all non‐specific treatment variables. Therefore, success of EMG biofeedback in alleviating insomnia to date may attribute to self‐monitoring effects or demand and expectancy effects.” (p. 32) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 625
Number of participants included: 36
Number of participants followed‐up at post‐treatment: 24
Number of participants randomly assigned to:
Number of withdrawals: n = 12
Diagnosis: insomnia (sleep‐wake disorder) Diagnostic manual: not stated Means of assessment: self‐report – Sleep Behavior Questionnaire Comorbidity: “Thirteen coeds reported currently experiencing severe headaches, and all but one coed reported that these headaches were stress related.” (p. 40) Age: 20.38 mean years, (rRange = 18 to 25 years of age) IQ: not stated Sex: 100% female Ethnicity: all were Caucasian Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo
Treatment name (type): EMG Pseudo‐feedback Training Group
Description of intervention: “All coeds of this group received treatment identical to that of the EMG biofeedback training group with the exception that feedback provided was noncontingent upon their EMG levels (...) During the training phase, each daily training session conformed to the following procedure: the coed spent 2 ½ minutes resting quietly in the reclining chair in the training room, followed immediately by 2 ½ minutes with the electrodes attached to the forehead. Then, the tape recorder was turned on and each coed heard feedback recorded from the
training session of a randomly chosen biofeedback group coed.” (p. 47)
Individual or group treatment: individual
Exposure/intensity to treatment: “During the training phase, each daily training session conformed to the following procedure: the coed spent 2 ½ minutes resting quietly in the reclining chair in the training room, followed immediately by 2 ½ minutes with the electrodes attached to the forehead.” (p. 47)
Duration of treatment: 3 weeks
Concomitant psychotherapy: no ‐ exclusion if subject was under other treatment for insomnia
Concomitant pharmacotherapy: no – “Subjects were excluded from this study if they reported having taking any prescription medication for sleep induction within five weeks prior to the beginning of the study.” (p. 40) Wait‐list Comparison name: No‐treatment Waiting‐list Control group Description of intervention: “Coeds of this group were informed at the initial pre‐treatment meeting that all treatment groups were filled, and that they would receive treatment at the earliest possible time. They were requested to complete only the Sleep Behavior Questionnaire and the STAI following completion of this study Exposure/intensity to treatment: no treatment Duration treatment: not stated Concomitant psychotherapy: no – exclusion if participant was under other treatment for insomnia Concomitant pharmacotherapy: no – “Subjects were excluded from this study if they reported having taking any prescription medication for sleep induction within five weeks prior to the beginning of the study.” (p. 40) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: (…) 36 were assigned to one of four groups, with nine coeds per group. An attempt was made to make each group as equivalent as possible with regard to sleep behavior indices.” (p. 44) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | No | Quote: "Data from one coed of the biofeedback group were randomly excluded from the results to allow an equal N per group.” (p. 38) Attrition >15% (33,3%). No ITT. Only reports data on completers |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Watzl 1988.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 3 months Duration of participation (trial + follow‐up): 3 months + 1 year follow‐up Setting: inpatient Purpose of trial: “This study investigates the impact of both the strict surveillance of patient’s intake and of nonspecific medication on relapse rates.” (p. 197) Open or closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 80 Number of participants followed‐up at post‐treatment: 70 Number of participants randomly assigned to :
Number of withdrawals : n = 0 Diagnosis : alcohol substance use Diagnostic manual: International Statistical Classification of Diseases and Related Health Problems, 9th edition (ICD‐9) Means of assessment: not stated. The duration of dependence was 7.2 years on the average. Ninety per cent of the women reported daily or nearly daily consumption Comorbidity: not stated Age: 36.7,mean years (range = 21 to 59) I Q: not stated Sex: 100% female Ethnicity: not stated Country: Germany Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: Placebo Injection
Description of intervention : “A placebo condition is employed to assess the psychological sequelae of nonspecific medication.” (p. 197) Individual or group treatment : Individual Exposure/intensity to treatment: 2 injections every week. Duration of treatment : 3 months Concomitant psychotherapy : Behavioral treatment inpatient Concomitant pharmacotherapy : not stated No‐treatment Comparison name: No‐treatment Description of intervention: only assessment before and after. Exposure/intensity to treatment: no treatment Duration of treatment: 3 months Concomitant psychotherapy: behavioral treatment inpatient Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”To assign patients to one of the four conditions, quadruples matched for age and number of previous detoxifications were formed. First, two of these four patients were randomly assigned to the “placebo”‐ condition, the remaining two patients to the “no placebo”‐condition. Patients were informed that only those were to receive the medication (called EWOCA), for whom a certain blood factor indicates that they would profit from such a treatment.” (p. 191) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Yes | Quote: ”Of these, 10 were not considered in the final evaluation. Four of them were husbands of female patients. Six patients had dropped out of treatment due to reasons not related to the investigation.” (p. 198) Attrition <15% (12.5). Outcome were relapses |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Weingaertner 1971.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 2 weeks Duration of participation (trial + follow‐up): 2 weeks. No follow up Setting: inpatient Purpose of trial : “The present study employed a three‐group design in which hospitalized schizophrenics were given a shock box and told to shock themselves each time they experienced hallucinating voices.” (p. 422) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 95
Number of participants included: 45
Number of participants followed‐up at post‐treatment: not stated
Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: schizophrenia Diagnostic manual: not stated Means of assessment: not stated Comorbidity: none were physically handicapped. Two patients with brain damage. Age: 37.2 mean years IQ: not stated Sex: 100% male Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo
Treatment name: Shock placebo
Description of intervention: “Patients in the placebo group carried a box which gave no shock.” (p. 422)
Individual or group treatment: individual
Exposure/intensity to treatment: all patients in the self‐shock and placebo groups were told to retain the box for two weeks and to get in touch with E if they had any trouble. Any adjustments necessary during the two weeks were made promptly.
Duration of treatment: 2 weeks
Concomitant psychotherapy: not stated
Concomitant pharmacotherapy: no hallucinogenic drugs No‐treatment Comparison name: No‐treatment Description of intervention: “The no‐treatment group received only the pre and post evaluations which were given all Ss Exposure/intensity to treatment: “(p. 422) Duration of treatment: 2 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: no hallucinogenic drugs |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: "The 5s were randomly assigned to three groups: self‐shock, placebo, and no treatment" (p. 423) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: ”The Brief Psychiatric Rating Scale (...) was filled out by two raters independently on the basis of an 18‐minute interview. (...) The raters were not aware of the project for which the patient was being evaluated and in general did not know whether a rating was the first, last, or one of a series of routine ratings as required in some of the studies conducted simultaneously with this one.” (p. 424) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | No information |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Whittaker 1963.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 10 weeks Duration of participation (trial + follow‐up): not stated Setting: inpatient Purpose of trial: “Whilst a number of these studies was controlled, in none of the investigations was a separate control group receiving no medication employed. This is a technique which would help to gauge the relative importance of placebo and pharmacological effects.” (p. 422) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: not stated Number of participants included: 39 Number of participants followed‐up at post‐treatment: 39 Number of participants randomly assigned to:
Number of withdrawals : n= 0 Diagnosis: chronic schizophrenia Diagnostic manual: not stated, but diagnosed by at least two psychiatrists Means of assessment: clinical interview Comorbidity: all had a paranoid condition, but two had also catatonic tendencies, and another had hebephrenic features. Six had been leucotomised prior to 1952 Age: 50 mean years, (range = 27 to 66) IQ: not stated Sex: 100% male Ethnicity: not stated Country: Scotland Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name: Placebo elixir
Description of intervention:: not stated
Individual or group treatment: individual
Exposure/intensity to treatment: 8 mg (3 patients received 20/12/12mg)
Duration of treatment: 10 weeks
Concomitant psychotherapy: not stated – but inpatient
Concomitant pharmacotherapy: “They had all been receiving maintenance doses of perphenazine (Fentazin) for periods ranging from 8 to 30 months and the average duration was 16 months. Their condition had shown no significant recent changes: no E.C.T. had been used ‐ at least since perphenazine had been introduced, and none had an early prospect of discharge.” (p. 422)
“If a patient had required an anti‐Parkinsonism agent, orphenadrine (Disipal), this was continued in the same dosage irrespective of the trial group to which the patient was allocated.” (p. 422)
“No other psychotropic drugs were used during the trial which lasted for ten weeks from 4 October, 1961 until 12 December, 1961 (inclusive).” (p. 422) No‐treatment Comparison name: No‐medication Description of intervention: not stated Exposure/intensity to treatment: no medication Duration of treatment: 10 weeks Concomitant psychotherapy: not stated – but inpatient Concomitant pharmacotherapy: “They had all been receiving maintenance doses of perphenazine (Fentazin) for periods ranging from 8 to 30 months and the average duration was 16 months. Their condition had shown no significant recent changes: no E.C.T. had been used ‐ at least since perphenazine had been introduced, and none had an early prospect of discharge.” (p. 422) “If a patient had required an anti‐Parkinsonism agent, orphenadrine (Disipal), this was continued in the same dosage irrespective of the trial group to which the patient was allocated.” (p. 422) “No other psychotropic drugs were used during the trial which lasted for ten weeks from 4 October, 1961 until 12 December, 1961 (inclusive).” (p. 422) |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”Each patient was arbitrarily allocated to one of three groups of I 3 by the hospital pharmacist.” (p. 423) |
| Allocation concealment | Yes | Quote: “Individual bottles were used and, as far as the latter two groups were concerned, the trial was blind in that only the pharmacist knew which bottles were active.” (p. 423) |
| Blinding of outcome assessors | No | Quote: “Within the ten weeks of the trial, eleven patients were considered to have relapsed into their previous pattern (…) This was done by the doctor (C.B.W) acting upon his own observations, or upon those of the charge nurses.” (p. 424) |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐treatment |
| Incomplete outcome data | Unclear | Attrition not clear. Outcome were relapses |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Wilson 1980.
| Study characteristics | ||
| Methods |
Parallel randomised trial with three arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 1 day (one operation – 10 to 15 minutes). Duration of participation (trial + follow‐up): one operation + 48 weeks of follow‐up Setting: outpatient Purpose of trial: “In a recent study, we compared the effect of disulfiram and placebo implants in alcoholic patients. The present study is a partial replication using larger samples and including no‐operation controls and pseudocontrols (…) We designed the present study to evaluate the effectiveness of disulfiram implantation by comparing the duration of abstinence in patients receiving disulfiram or placebo implants and patient knowingly receiving neither type of implant” (p. 429) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 100 Number of participants included: 90 Number of participants followed‐up at post‐treatment: 90 Number of participants randomly assigned to:
Number of withdrawals: not stated Diagnosis: substance use disorder (alcoholic) Diagnostic manual: not stated Means of assessment: not stated Comorbidity: not stated Age: 36.1 mean years (SD = 4.9) IQ: not stated Sex: 11% female Ethnicity: not stated Country: Canada Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Pharmacological placebo
Treatment name : Placebo implant
Description of intervention: ”The disulfiram and placebo implant operations were carried out as in our previous study.” (p. 430) From Wilson 1978 : “The operations were carried out in an outpatient operating room using standard aseptic surgical technique (…) For patients implanted with placebo, 0.5 cc of physiological saline was deposited in the tract using a 10‐cc syringe and 19‐gauge needle. The procedure was repeated to a total of 8 insertions in 8 separate tracts radiating from the central incision.” (p. 812) Individual or group treatment: individual Exposure/intensity to treatment: one day one operation Duration of treatment: one operation – 10 to 15 minutes Concomitant psychotherapy : not stated Concomitant pharmacotherapy: not stated No‐treatment Comparison name : No‐operation control Description of intervention : “No‐operation control patients were told that an implant did not appear to be appropriate for them at this time and were asked to participate as though they had received an implant (…)” (p. 4309 Exposure/intensity to treatment: no treatment Duration treatment: one day (no treatment) Concomitant psychotherapy : not stated Concomitant pharmacotherapy: not stated |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”The 90 patients who met the selection criteria were block randomly assigned to a disulfiram implant (N=40), placebo implant (N=40), or no‐operation control (N=10) condition.” (p. 430) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | No | Patient‐reported outcomes |
| Blinding of participants and personnel | No | Not possible to blind placebo and no‐operation |
| Incomplete outcome data | Unclear | Attrition unclear |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Wolitzky 2009.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: yes Cluster randomised: no Duration of trial (baseline to post): 1 treatment: Duration of participation (trial + follow‐up): 1 treatment + 1 month follow‐up Setting: outpatient Purpose of trial: “The principal aim of the current study was to investigate the hypothesized facilitative effects of having acrophobic individuals engage in actions (i.e., running toward the rail of a balcony, spinning in place in the phobic situation to induce dizziness, holding one’s hands behind one’s back while looking over the edge of a railing) that are in direct opposition to their threat‐relevant fear action tendencies.” (p. 59) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened:: not stated Number of participants included: 89 Number of participants followed‐up at post‐treatment: 88 Number of participants randomly assigned to:
Number of withdrawals : n=1
Diagnosis: specific phobia (acrophobia) Diagnostic manual:: Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (DSM‐IV) Means of assessment: Composite International Diagnostic Interview (CIDI‐Auto). Comorbidity: not stated Age: 20.08 mean years (range =18 to 64) IQ: not stated, but 82% were university students Sex: 69% female Ethnicity: 49% Caucasians, 15% Hispanic/Latino, 12.5% African‐American, 19% Asian‐American, 3% Native American, and 2% multi‐racial or other race. Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Physical placebo Treatment name: Credible placebo, pulsed audio‐photic stimulation Description of intervention:“APS (Seiver, Mind Alive, Inc.) is typically used by health professionals to induce relaxation. The APS device resembles a small soundboard and is about the size of a MP3 player. The device consist of a headset, which emits controllable pulsing sounds like a metronome, and a plastic mask, which produces orange lights at controllable rates (…) Participants were told that introducing these lights and sounds would relax them by inducing alpha waves in the brain, which are typically associated with relaxation and meditation” (p. 62) Individual or group treatment: individual Exposure/intensity to treatment :“Treatment was delivered in six, 6‐min trials, for a total of one 36‐min session.” (p. 61‐2) Duration of treatment: one treatment (one day) Concomitant psychotherapy: not stated – but probably not as they were not in or seeking treatment Concomitant pharmacotherapy\; not stated – but probably not as they were not in or seeking treatment Wait‐list Comparison name: Wait list control Description of intervention: “Participants received no treatment until after the posttreatment assessment was completed. At that time, they received on 36‐min session of in vivo exposure treatment. In order to provide treatment as soon as possible, participants in this condition did not complete a follow‐up assessment.” (p. 63) Exposure/intensity to treatment: no treatment. Duration treatment: one day waiting Concomitant psychotherapy: not stated – but probably not as they were not in or seeking treatment Concomitant pharmacotherapy: not stated – but probably not as they were not in or seeking treatment |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”89 participants were randomized to one of the four treatment conditions (14 participants declined to participate in the treatment phase of the study).” (p. 60) “One‐way ANOVAs were used to assess whether randomization was successful in achieving equivalent groups at baseline. As seen in Table 1, the four experimental groups did not differ on the measures at baseline with the exception of BAT‐2 peak fear ratings (…)” (p. 63) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Yes | Quote: ”During each treatment trial, a trained undergraduate research assistant blind to the study hypotheses coded the presence or absence of nine possible oppositional actions.” (p. 61) |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | Yes | Quote: One participant dropped out during treatment. Thus, 88 participants completed treatment, which was conducted one week after the pretreatment assessment.” (p. 60) Attrition < 15% (1.1%). |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
Wollersheim 1991.
| Study characteristics | ||
| Methods |
Parallel randomised trial with four arms
Sample calculation: not stated Cluster randomised: no Duration of trial (baseline to post): 11 weeks Duration of participation (trial + follow‐up): 11 weeks + 6 months follow‐up Setting: outpatient Purpose of trial: “To evaluate differentially the effectiveness of cognitive‐behavioral group treatment the present investigation included a supportive group approach, a bibliotherapeutic approach, and a delayed treatment condition.” (p. 496‐7) Open/closed placebo: closed placebo |
|
| Data |
Number of participants screened: 99 Number of participants included: 32 Number of participants followed‐up at post‐treatment: 25 Number of participants randomly assigned to:
Number of withdrawals : n =7
Diagnosis: depression Diagnostic manual: Diagnostic and Statistical Manual of Mental Disorders, 3rd Edition (DSM‐III) Means of assessment: Clinical interview Comorbidity: not stated Age: 39.4 mean years (range= 22 to 68 years) IQ: not stated Sex: 71.9% female Ethnicity: not stated Country: USA Inclusion criteria
Exclusion criteria
|
|
| Comparisons |
Psychological placebo Treatment name: Supportive therapy Description of intervention: “The supportive treatment was designed to parallel the group format used in coping therapy, but supportive therapy techniques were much less directive and did not emphasize therapist‐generated problem‐solving tactics (p. 497) Individual or group treatment: Group Exposure/intensity to treatment: 10 weekly treatment sessions of 2 hours Duration of treatment: 11 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: medication not allowed Wait‐list Comparison name: Delayed treatment group Description of intervention: "Patients assigned to this condition were informed that their treatment would begin 11 weeks after their initial assessment session. Members of the delayed treatment group were asked to call if they needed more immediate treatment and told that in such cases treatment would be arranged but they would then not be involved in the treatment research program. In addition to receiving individual assessment sessions with a clinician, they were also given reassurance regarding prognosis and hope for improvement and were informed that they would receive the best approach available at the start of their treatment. After post‐treatment assessment, these patients were given group coping treatment." (p. 498) Exposure/intensity to treatment: waiting for treatment Duration of treatment: 11 weeks Concomitant psychotherapy: not stated Concomitant pharmacotherapy: medication not allowed |
|
| Outcomes |
Beneficial effect
Adverse events
|
|
| Notes |
Key conclusion from study authors
Key limitations from study authors
Other notes from review authors
Conflicts of interest: none found Judgement: yes |
|
| Risk of bias | ||
| Item | Authors' judgement | Support for judgement |
| Random sequence generation | Unclear | Quote: ”All 32 patients were blocked for age and sex and then randomly assigned to one of four treatment groups: coping therapy, supportive therapy, bibliotherapy, or a delayed treatment group.” (p. 498) |
| Allocation concealment | Unclear | No information |
| Blinding of outcome assessors | Unclear | No information |
| Blinding of participants and personnel | No | Not possible to blind placebo and wait‐list |
| Incomplete outcome data | No | Quote: “A total of seven patients (two each from coping and supportive group treatments, three from bibliotherapy) could not be located at follow up” (p. 499) Attrition >15% (21.9%). No ITT. |
| Selective outcome reported | Unclear | No protocol found |
| Other sources of bias | Yes | No other sources found |
CBT: Cognitive behavioral therapy; CPAP: continuous positive airway pressure; GAF: (Global Assessment of Functioning; IQ: intelligence quotient; ITT: intention‐to‐treat; MDD: Major depressive disorder; NCPAP: (nasal continuous positive airway pressure; SD: standard deviation; TAU: treatmnt as usual; WISC: Wechsler Intelligence Scale for children
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Adelman 1990 | Not a randomised clinical trial |
| Altenhofer 2007 | Not a randomised clinical trial |
| Andreasson 1990 | Not a randomised clinical trial |
| Banasiak 2005 | Did not fulfil inclusion criteria for a usual care |
| Bass 2016 | Did not fulfil inclusion criteria for a usual care |
| de Jongh 1995 | Did not fulfil inclusion criteria for a usual care |
| Dolhanty 2005 | Did not fulfil inclusion criteria for a physical placebo |
| Drozdek 2010 | Not a randomised clinical trial |
| Endicott 1964 | Not a randomised clinical trial |
| Ertl 2011 | Did not fulfil inclusion criteria for a usual care |
| Gitlin 2013 | Did not fulfil inclusion criteria for a usual care |
| Gournay 1994 | Wrong patient population (family distress) |
| Haertzen 1964 | Not a randomised clinical trial, and wrong population (post‐addicts) |
| Hartelius 1968 | Not a randomised clinical trial |
| Hinsberger 2017 | Some participants were not randomised but placed on a second wait list called “no camp”. |
| Keltner 1976 | Not a randomised clinical trial |
| Laessle 1988 | Not a randomised clinical trial |
| Litrownik 1978 | Did not fulfil inclusion criteria for a physical placebo |
| Mak 2008 | Did not fulfil inclusion criteria for a physical placebo |
| McCall 2005 | No wait‐list or no‐treatment group included |
| McNeil 1991 | No placebo group included |
| Otteson 1979 | Did not fulfil inclusion criteria for a usual care |
| Propst 1992 | Did not fulfil inclusion criteria for a usual care |
| Sackley 2018 | Did not fulfil inclusion criteria for a usual care |
| Salt 2002 | Not a randomised clinical trial |
| Schultz 2002 | Wrong patient population quote: ("to be interested in stress and depression" |
| Sheikh 1986 | Did not fulfil inclusion criteria for a physical placebo |
| Steiner 2011 | Did not fulfil inclusion criteria for a usual care |
| Timko 1995 | Not a randomised clinical trial |
| Tomporowski 1984 | Pools no‐treatment, usual care and placebo in one group. |
| Torkan 2014 | Did not fulfil inclusion criteria for a psychological placebo |
| Weber 1975 | Not a randomised clinical trial |
| Wilson 2002 | Did not fulfil inclusion criteria for a usual care |
Characteristics of studies awaiting classification [ordered by study ID]
Bommert 1978.
| Methods | Randomised controlled trial |
| Data | Not available Condition: Learning Disabled students |
| Comparisons |
Four‐armed randomised trial
|
| Outcomes | Not stated |
| Notes | Not able to locate the full report |
Brandes 2010.
| Methods | Not stated |
| Data | Not stated |
| Comparisons | Not stated |
| Outcomes | Not stated |
| Notes | Only the title located. Not possible to locate the full report. Authors did not respond to correspondence |
McLachlan 1993.
| Methods | Randomsied single‐blind, placebo‐controlled clinical trial |
| Data | Not available Condition : Alzheimer's disease |
| Comparisons |
Three‐armed randomised trial
|
| Outcomes |
Outcome
|
| Notes | Not able to locate the full report |
Newton‐Cross 2017.
| Methods | Not stated |
| Data | Not stated |
| Comparisons | Not stated |
| Outcomes | Not stated |
| Notes | Not able to locate the full report. Not able to find contact information of the authors |
Schwarzler 1999.
| Methods | Not stated |
| Data | Not stated |
| Comparisons | Not stated |
| Outcomes | Not stated |
| Notes | Not able to locate the full report |
Trianes Torres 1991.
| Methods | Not stated |
| Data | Not available Condition : Inhibited and impulsive preschool children |
| Comparisons |
Three‐armed randomised trial
|
| Outcomes |
Outcome
|
| Notes | Not able to locate the full report. Not able to find contact information of the authors |
Characteristics of ongoing studies [ordered by study ID]
Heitman 2017.
| Study name | Internet‐based attentional bias modification training as add‐on to regular treatment in alcohol and cannabis dependent outpatients: a study protocol of a randomised control trial |
| Methods | Randomised controlled trial |
| Data | Not available Condition: substance dependent (alcohol/cannabis) |
| Comparisons |
Two‐armed randomised trial
|
| Outcomes |
Primary outcomes
Secondary outcomes
|
| Starting date | 18/05/2015 |
| Contact information | Janika Heitmann email: J.Heitmann@vnn.nl |
| Notes | Quote: "We are currently processing the data, but unfortunately I do not yet have data to share (in case this is still of interest in a couple of moths it might be possible to share some results with you). Further, after reading your protocol I doubt whether our study is eligible for your review. That is, all our participants received psychological treatment (cognitive behavioural therapy), and only for the add‐on treatment we had three groups from which one is a group that received a placebo treatment and one was a treatment‐as‐usual group (only received cognitive behavioural therapy and no add‐on)." Heitmann 2017 (pers comm) |
ISRCTN21392756.
| Study name | Standardised stress management mental health training: does it have a beneficial effect? |
| Methods | Single‐centre open randomised controlled trial |
| Data | Not available Condition: adjustment disorder or mild depression |
| Comparisons |
Two‐armed randomised trial
|
| Outcomes |
Primary outcome
Secondary outcomes
|
| Starting date | 06/09/2010 |
| Contact information | Dr Trevor Hicks email: trevor@hicks65.freeserve.co.uk |
| Notes | No response to author correspondence |
ISRCTN35717198.
| Study name | Cognitive training with non‐invasive brain stimulation to treat binge eating disorder |
| Methods | Randomised controlled trial |
| Data | Not available Condition : Binge Eating Disorder |
| Comparisons |
Three‐armed randomised trial
|
| Outcomes |
Primary outcomes The primary outcome for this proof‐of‐concept feasibility study is to establish the feasibility of adding concurrent [ABM + tDCS] to treatment as usual in a binge eating disorder patient group and acquire key information to inform the development of a large‐scale randomised sham‐controlled trial (RCT). In line with recommendations of Eldridge et al. (2016). The primary outcomes of the proposed feasibility study are to:
Secondary outcomes
|
| Starting date | 03/05/2018 |
| Contact information | Gemma Gordon Email: gemma.gordon@kcl.ac.uk |
| Notes | No response to author correspondence |
NCT00044629.
| Study name | Combined behavioral/pharmacologicaltTherapy for insomnia |
| Methods | Randomised clinical trial |
| Data | Not available Condition: insomnia |
| Comparisons |
Three‐armed randomised trial
|
| Outcomes | Not stated |
| Starting date | 05/09/2002 |
| Contact information | Duke University |
| Notes | Not able to find any contact information |
Differences between protocol and review
There were some differences between the protocol and review. These included i) pooling of placebos, ii) dealing with missing data, iii) subgroup analyses, iv) sensitivity analyses, v) selection of studies, vi) an extended decision hierarchy, and vii) no meta‐regression analyses. For more information, see Table 9.
Contributions of authors
EF: protocol development, screening, data extraction, statistical analyses, writing, revisions
AT: protocol development, screening, data extraction, statistical analyses, writing, revisions
LB: screening, data extraction, statistical analyses, revisions
AH: protocol development, writing, revisions
CG: protocol development, statistical analyses, writing, revisions
MK: protocol development, writing, revisions
ES: protocol development, writing, revisions
OJ: protocol development, statistical analyses, writing, revisions
Sources of support
Internal sources
-
Psychiatric Research Unit, Region Zealand Psychiatry, Slagelse, Denmark
EF, AT, MTK, ES, and OJS we re personally salaried by the institution during the period of this review.
-
Center for Evidence‐Based Medicine, Odense University Hospital and University of Southern Denmark, Denmark
AH wa s personally salaried by the institution during the period of this review.
-
The Copenhagen Trial Unit, Centre for Clinical Intervention Research, Rigshospitalet, Copenhagen, Denmark
CG wa s personally salaried by the institution during the period of this review.
External sources
No sources of support provided
Declarations of interest
The review authors have no relevant interests to declare.
Shared first authorship
Shared first authorship
New
References
References to studies included in this review
Abikoff 2004 {published data only}
- Abikoff H, Hechtman L, Klein RG.Social functioning in children with ADHD treated with long-term methylphenidate and multimodal psychosocial treatment. Journal of the American Academy of Child and Adolescent Psychiatry 2004;43(7):820-9. [DOI] [PubMed] [Google Scholar]
- Abikoff H.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to A Todorovac 23rd of August 2019.
- Klein RG, Abikoff H, Hechtman L, Weiss G.Design and rationale of controlled study of long-term methylphenidate and multimodal psychosocial treatment in children with ADHD. Journal of the American Academy of Child and Adolescent Psychiatry 2004;43(7):792-801. [DOI] [PubMed] [Google Scholar]
Allen 1998 {published data only}
- Allen JB.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to A Todorovac 28th of August 2019.
- Allen JJ, Schnyer RN, Hitt SK.The efficacy of acupuncture in the treatment of major depression in women. Psychological Science 1998;9(5):397-401. [Google Scholar]
Allen 2006 {published data only}
- Allen JB.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 28th of August 2019.
- Allen JJ, Schnyer RN, Chambers AS, Hitt SK, Moreno FA, Manber R.Acupuncture for depression: a randomized controlled trial. Journal of Clinical Psychiatry 2006;67(11):1665-73. [DOI] [PubMed] [Google Scholar]
Alvarez 1997 {published data only}
- Alvarez JR.The effects of anger management training on depression, anger, self-concept, and treatment responsibility in a sample of drug-dependent individuals in a therapeutic community. Dissertation Abstracts International: Section B: the Sciences and Engineering 1997;58(4-B):2108. [Google Scholar]
Ascher 1979 {published data only}
- Ascher LM, Turner R.Paradoxical intention and insomnia: an experimental investigation. Behaviour Research and Therapy 1979;17(4):408-11. [DOI] [PubMed] [Google Scholar]
Ayen 2004 {published data only}
- Ayen I, Hautzinger M.Cognitive behavior therapy for depression in menopausal women. A controlled, randomized treatment study [Kognitive Verhaltenstherapie bei Depressionen im Klimakterium: Eine kontrollierte, randomiserte Interventionsstudie]. Zeitschrift für Klinische Psychologie und Psychotherapie 2004;33(4):290-9. [Google Scholar]
- Ayen I.Inclusion of study in Cochrane Systematic Review (personal communication). Email to EG Faltinsen 9th of September 2019.
Berg 1983 {published data only}
- Berg I, Forsythe I, Holt P, Watts J.A controlled trial of ‘Senokot’ in faecal soiling treated by behavioural methods. Journal of Child Psychology and Psychiatry 1983;24(4):543-9. [DOI] [PubMed] [Google Scholar]
Borden 1986 {published data only}
- Borden, KA.Attributional Outcomes: the Subtle Messages of Treatments for Attention Deficit Disorder. Dissertation, University of Illinois at Urbana-Champaign 1986.
- Borden KA, Brown RT.Attributional outcomes: The subtle messages of treatments for attention deficit disorder. Cognitive Therapy and Research 1989;13(2):147-60. [Google Scholar]
Borkovec 1975 {published data only}
- Borkovec TD, Kaloupek DG, Slama KM.The facilitative effect of muscle tension-release in the relaxation treatment of sleep disturbance. Behavior Therapy 1975;6(3):301-9. [Google Scholar]
Borkovec 1976 {published data only}
- Borkovec TD, Weerts TC.Effects of progressive relaxation on sleep disturbance: an electroencephalographic evaluation. Psychosomatic Medicine 1976;38(3):173-80. [DOI] [PubMed] [Google Scholar]
Bornovalova 2008 {published data only}
- Bornovalova M.Inclusion of study in Cochrane Systematic Review (personal communication). Email to A Todorovac. 21st of August 2019.
- Bornovalova MA.Distress tolerance treatment for inner-city drug users: a preliminary trial. Doctor’s Dissertation, University of Maryland 2008.
Bramston 1985 {published data only}
- Bramston P, Spence SH.Behavioural versus cognitive social-skills training with intellectually-handicapped adults. Behaviour Research and Therapy 1985;23(3):239-46. [DOI] [PubMed] [Google Scholar]
Brill 1964a {published data only}
- Brill NQ, Koegler RR, Epstein LJ, Forgy EW.Controlled study of psychiatric outpatient treatment. Archives of General Psychiatry 1964;10:581-95. [DOI] [PubMed] [Google Scholar]
- Brill NQ.Results of psychotherapy. California Medicine 1966;104(4):249-53. [PMC free article] [PubMed] [Google Scholar]
Brill 1964b {published data only}
- Brill NQ, Koegler RR, Epstein LJ, Forgy EW.Controlled study of psychiatric outpatient treatment. Archives of General Psychiatry 1964;10:581-95. [DOI] [PubMed] [Google Scholar]
Carlson 1993 {published data only}
- Carlson CL, Pelham WE, Milich R, Hoza B.ADHD boys’ performance and attributions following success and failure: drug effects and individual differences. Cognitive Therapy and Research 1993;17:269-87. [Google Scholar]
Carter 2003 {published data only}
- Carter JC, Olmsted MP, Kaplan AS, McCabe RE, Mills JS, Aime A.Self-help for bulimia nervosa: a randomized controlled trial. American Journal of Psychiatry 2003;160(5):973-8. [DOI] [PubMed] [Google Scholar]
Crisp 1991 {published data only}
- Crisp AH, Norton K, Gowers S, Halek C, Bowyer C, Yeldham D, et al.A controlled study of the effect of therapies aimed at adolescent and family psychopathology in Anorexia Nervosa. British Journal of Psychiatry: the Journal of Mental Science 1991;159:325-33. [DOI] [PubMed] [Google Scholar]
- Gowers S, Norton K, Halek C, Crisp AH.Outcome of outpatient psychotherapy in a random allocation treatment study of anorexia nervosa. International Journal of Eating Disorders 1994;15(2):165-77. [DOI] [PubMed] [Google Scholar]
- Gowers S, Norton K, Yelsham D.The St George's prospective treatment study of anorexia nervosa: a discussion of methodological problems. International Journal of Eating Disorders 1989;8:445-54. [Google Scholar]
Crouch 1988 {published data only}
- Crouch G, Robson M, Hallstrom C.Benzodiazepine dependent patients and their psychological treatment. Progress in Neuro-psychopharmacology & Biological Psychiatry 1988;12(4):503-10. [DOI] [PubMed] [Google Scholar]
- Hallström C, Crouch G, Robsen M, Shine P.The treatment of tranquillizer dependence by propranolol. Postgraduate Medical Journal 1988;64(Suppl 2):40-4. [PubMed] [Google Scholar]
Doty 1975 {published data only}
- Doty, DW.Role playing and incentives in the modification of the social interaction of chronic psychiatric patients. Journal of Consulting and Clinical Psychology 1975;43(5):676-82. [DOI] [PubMed] [Google Scholar]
Double 1993 {published data only}
- Double DB, Warren GC, Evans M, Rowlands MP.Efficacy of maintenance use of anticholinergic agents. Acta Psychiatrica Scandinavica 1993;88:381-4. [DOI] [PubMed] [Google Scholar]
Ehlers 2014 {published data only}
- Ehlers A, Hackmann A, Grey N, Wild J, Liness S, Albert I, et al.A randomized controlled trial of 7-day intensive and standard weekly cognitive therapy for PTSD and emotion-focused supportive therapy. American Journal of Psychiatry 2014;171(3):294-304. [DOI] [PMC free article] [PubMed] [Google Scholar]
Espie 1989a {published data only}
- Espie CA, Brooks DN, Lindsay WR.An evaluation of tailored psychological treatment of insomnia. Journal of Behavior Therapy and Experimental Psychiatry 1989;20(2):143-53. [DOI] [PubMed] [Google Scholar]
- Espie CA, Lindsay WR, Brooks DN, Hood EM, Turvey T.A controlled comparative investigation of psychological treatments for chronic sleep-onset insomnia. Behaviour Research and Therapy 1989;27(1):79-88. [DOI] [PubMed] [Google Scholar]
Etringer 1982 {published data only}
- Etringer BD, Cash TF, Rimm DC.Behavioral, affective, and cognitive effects of participant modeling and an equally credible placebo. Behavior Therapy 1982;13(4):476-85. [Google Scholar]
Foa 1991 {published data only}
- Foa EB, Rothbaum BO, Riggs DS, Murdock TB.Treatment of posttraumatic stress disorder in rape victims: a comparison between cognitive-behavioral procedures and counselling. Journal of Consulting and Clinical Psychology 1991;59(5):715-23. [DOI] [PubMed] [Google Scholar]
Foa 2018 {published data only}
- Foa EB, McLean CP, Zang Y, Rosenfield D, Yadin E, Yarvis JS, et al.Effect of prolonged exposure therapy delivered over 2 weeks vs 8 weeks vs present-centered therapy on PTSD symptom severity in military personnel: a randomized clinical trial. JAMA 2018;319(4):354-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
Freire 2007 {published data only}
- Freire AO, Sugai GC, Chrispin FS, Togeiro SM, Yamamura Y, Mello LE, et al.Treatment of moderate obstructive sleep apnea syndrome with acupuncture: a randomised, placebo-controlled pilot trial. Sleep Medicine 2007;8(1):43-50. [DOI] [PubMed] [Google Scholar]
Fuchs 1977 {published data only}
- Fuchs CZ, Rehn LP.A self-control behavior therapy program for depression. Journal of Consulting and Clinical Psychology 1977;45(2):206-15. [DOI] [PubMed] [Google Scholar]
Glogowska 2000 {published data only}
- Glogowska M, Roulstone S, Enderby P, Peters TJ.Randomised controlled trial of community based speech and language therapy in preschool children. BMJ 2000;321(7266):923-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Goldstein 2000 {published data only}
- Goldstein AJ, Beurs E, Chambless Dl, Wilson KA.EMDR for panic disorder with agoraphobia: comparison with waiting list and credible attention-placebo control conditions. Journal of Consulting and Clinical Psychology 2000;68(6):947-56. [PubMed] [Google Scholar]
Goldwasser 1987 {published data only}
- Goldwasser AN, Auerbach SM, Harkins SW.Cognitive, affective, and behavioural effects of reminiscence group therapy on demented elderly. Aging and Human Development 1987;25(3):209-22. [DOI] [PubMed] [Google Scholar]
Hekmat 1984 {published data only}
- Hekmat H, Lubitz R, Deal R.Semantic desensitization: a paradigmatic intervention approach to anxiety disorders. Journal of Clinical Psychology 1984;40(2):463-6. [PubMed] [Google Scholar]
Hippman 2016 {published data only}
- Hippman C, Ringrose A, Inglis A, Cheek J, Albert AY, Remick R, et al.A pilot randomized clinical trial evaluating the impact of genetic counselling for serious mental illnesses. Journal of Clinical Psychiatry 2016;77(2):e190-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Howlin 2007 {published data only}
- Howlin P, Gordon KR, Pasco G, Wade A, Charman T.The effectiveness of picture exchange communication system (PECS) training for teachers of children with autism: a pragmatic,group randomised controlled trial. Journal of Child Psychology and Psychiatry 2007;48(5):473-81. [DOI] [PubMed] [Google Scholar]
Karst 2007 {published data only}
- Karst M, Winterhalter M, Münte S, Francki B, Hondronikos A, Eckardt A, et al.Auricular acupuncture for dental anxiety: a randomized controlled trial. Anesthesia and Analgesia 2007;104(2):295-300. [DOI] [PubMed] [Google Scholar]
- Karst M.Inclusion of study in Cochrane Systematic Review (personal communication). Email to: EG Faltinsen 5th of September 2019.
Kelley 2012 {published data only}
- Kelley J.Inclusion of study in Cochrane Systematic Review (personal communication). Email to: A Todorovac 12th of August 2019.
- Kelley JM, Kaptchuk TJ, Cusin C, Lipkin S, Fava M.Open-label placebo for major depressive disorder: a pilot randomized controlled trial. Psychotherapy and Psychosomatics 2012;81(5):312-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kennedy 1974 {published data only}
- Kennedy TD, Kimura HK.Transfer, behavioral improvement, and anxiety reduction in systematic desensitization. Journal of Consulting and Clinical Psychology 1974;42(5):720-8. [DOI] [PubMed] [Google Scholar]
Kilmann 1987 {published data only}
- Kilmann PR, Milan RJ, Boland JP, Nankin HR, Davidson E, West MO, et al.Group treatment of secondary erectile dysfunction. Journal of Sex & Marital Therapy 1987;13(3):168-82. [DOI] [PubMed] [Google Scholar]
- Kilmann PR, Milan RJ Jr, Boland JP, Davidson E, West MO, Sablis RF, et al.The sexual interactions of men with secondary erectile dysfunction. Journal of Sex Education and Therapy 1988;14(1):46-9. [Google Scholar]
- Kilmann PR, Milan RJ Jr, Boland JP, Nankin HR, Sablis RF, West MO, et al.The sexual interactions of men with secondary erectile dysfunction. Paper presented at the annual meeting of the American Psychological Association in Los Angeles 1985;1:46-9.
Klein 1977 {published data only}
- Klein SA, Deffenbacher JL.Relaxation and exercise for hyperactive impulsive children. Perceptual and Motor Skills 1977;45:1159-62. [DOI] [PubMed] [Google Scholar]
Klerman 1974a {published data only}
- Klerman GL, Dimascio A, Weismann M, Prusoff B, Paykel ES.Treatment of depression by drugs and psychotherapy. American Journal of Psychiatry 1974;131(2):186-91. [DOI] [PubMed] [Google Scholar]
- Paykel ES, Dimascio A, Haskell D, Prusoff BA.Effects of maintenance amitriptyline and psychotherapy on symptoms of depression. Psychological Medicine 1975;5(1):67-77. [DOI] [PubMed] [Google Scholar]
- Paykel ES, DiMascio A, Klerman GL, Prusoff BA.Maintenance therapy of depression. Pharmakopsychiatrie, Neuro-Psychopharmakologie 1976;9:127-39. [DOI] [PubMed] [Google Scholar]
- Paykel ES, Weissman MM, Prusoff BA.Social maladjustment and severity of depression. Comprehensive Psychiatry 1978;19(2):121-8. [DOI] [PubMed] [Google Scholar]
- Weissman MM, Klerman GL, Paykel ES, Prusoff B.Treatment effects on the social adjustment of depressed patients. Archives of General Psychiatry 1974;30:771-8. [DOI] [PubMed] [Google Scholar]
Klerman 1974b {published data only}
- Klerman GL, Dimascio A, Weismann M, Prusoff B, Paykel ES.Treatment of depression by drugs and psychotherapy. American Journal of Psychiatry 1974;131(2):186-91. [DOI] [PubMed] [Google Scholar]
Klosko 1990 {published data only}
- Klosko JS, Barlow DH, Tassinari R, Cerny JA.A comparison of alprazolam and behavior therapy in treatment of panic disorder: correction. Journal of Psychotherapy Practice and Research 1994;3(2):163-79. [PMC free article] [PubMed] [Google Scholar]
- Klosko JS, Barlow DH, Tassinari R, Cerny JA.A comparison of alprazolam and behavior therapy in treatment of panic disorder. Journal of Consulting and Clinical Psychology 1990;58(1):77-84. [DOI] [PubMed] [Google Scholar]
Krapfl 1970 {published data only}
- Krapfl JE, Nawas MM.Differential ordering of stimulus presentation in systematic desensitization. Journal of Abnormal Psychology 1970;75(3):333-7. [DOI] [PubMed] [Google Scholar]
Kwan 2017 {published data only}
- Kwan R.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 26th of August 2019.
- Kwan RY, Leung MC, Lai CK.Acupressure for agitation in nursing home residents with dementia: study protocol for a randomized controlled trial. Trials 2014;15(410):1-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwan RY, Leung MCP, Lai CK.A randomized controlled trial examining the effect of acupressure on agitation and salivary cortisol in nursing home residents with dementia. Dementia and Geriatric Cognitive Disorders 2017;44(1-2):92-104. [DOI] [PubMed] [Google Scholar]
Lacy 1990 {published data only}
- Lacy MK.The effect of short term autogenic training on the stress reactivity of hospitalized emotionally disturbed adolescents. ProQuest Dissertations Publishing, Oklahoma State University 1996.
Lai 2004 {published data only}
- Lai CK, Chi I, Kayser-Jones J.A randomized controlled trial of a specific reminiscence approach to promote the well-being of nursing home residents with dementia. International Psychogeriatrics 2004;16(1):33-49. [DOI] [PubMed] [Google Scholar]
- Lai K.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 6th of September 2019.
Lang 1965 {published data only}
- Lang OJ, Lazovik AD.Experimental desensitization of a phobia. Journal of Abnormal and Social Psychology 1963;66:519-25. [DOI] [PubMed] [Google Scholar]
- Lang PJ, Lazovik AD, Reynolds DJ.Desensitization, suggestibility, and pseudotherapy. Journal of Abnormal Psychology 1965;70(6):395-402. [DOI] [PubMed] [Google Scholar]
Legrand 2016 {published data only}
- Legrand F.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 14th of August 2019.
- Legrand FD, Neff EM.Efficacy of exercise as an adjunct treatment for clinically depressed inpatients during the initial stages of antidepressant pharmacotherapy: an open randomized controlled trial. Journal of Affective Disorders 2016;191:139-44. [DOI] [PubMed] [Google Scholar]
Lick 1975 {published data only}
- Lick J.Expectancy, false galvanic skin response feedback, and systematic desensitization in the modification of phobic behavior. Journal of Consulting and Clinical Psychology 1975;43(4):557-67. [DOI] [PubMed] [Google Scholar]
Lick 1977 {published data only}
- Lick JR, Heffler D.Relaxation training and attention placebo in the treatment of severe insomnia. Journal of Consulting and Clinical Psychology 1977;45(2):153-61. [DOI] [PubMed] [Google Scholar]
Liddle 1990 {published data only}
- Liddle B, Spence SH.Cognitive-behaviour therapy with depressed primary school children: A cautionary note. Behavioural Psychotherapy 1990;18(2):85-102. [Google Scholar]
Matson 1980 {published data only}
- Matson JL, Marchetti A, Adkins JA.Comparison of operant- and independence-training procedures for mentally retarded adults. American Journal of Mental Deficiency 1980;84(5):487-94. [PubMed] [Google Scholar]
McLachlan 1991 {published data only}
- McLachlan DR, Dalton AJ, Kruck TP, Bell MY, Smith WL, Kalow W, et al.Intramuscular desferrioxamine in patients with Alzheimer's disease. Lancet 1991;337(8753):1304-8. [DOI] [PubMed] [Google Scholar]
- McLachlan DRC, Smith WL, Kruck TP.Desferrioxamine and Alzheimer’s disease: video home behavior assessment of clinical course and measures of brain aluminum. Therapeutic Drug Monitoring 1993;15(6):602-7. [PubMed] [Google Scholar]
Mealiea 1971 {published data only}
- Mealiea WL, Nawas MM.The comparative effectiveness of systematic desensitization and implosive therapy in the treatment of snake phobia. Journal of Behavior Therapy and Experimental Psychiatry 1971;2(2):85-94. [Google Scholar]
Milby 1980 {published data only}
- Milby JB, Clarke C, Toro C, Thornton S, Rickert D.Effectiveness of urine surveillance as an adjunct to outpatient psychotherapy for drug abusers. International Journal of the Addictions 1980;15(7):993-1001. [DOI] [PubMed] [Google Scholar]
Miranda 1997 {published data only}
- Miranda A, Arlandis P, Soriano M.Instruction in strategies and attributional training: Effects on the problem-solving and self-concept of students’ with learning disabilities [Instrucción en estrategias y entrenamiento atribucional: efectos sobre la resolución de problemas y el autoconcepto de los estudiantes con dificultades en el aprendizaje]. Infancia y Aprendizaje 1997;20(80):37-52. [Google Scholar]
Mitchell 2008 {published data only}
- Mitchell C, Mansfield D, Rautenbach S.Coloured filters and reading accuracy, comprehension and rate: a placebo-controlled study. Perceptual and Motor Skills 2008;106(2):517-32. [DOI] [PubMed] [Google Scholar]
Nandi 1976 {published data only}
- Nandi DN, Ajmany S, Ganguli H, Banerjee G, Boral GC, Ghosh A, et al.A clinical evaluation of depressives found in a rural survey in India. British Journal of Psychiatry 1976;128:523-7. [DOI] [PubMed] [Google Scholar]
Nicassio 1974 {published data only}
- Nicassio P, Bootzin R.A comparison of progressive relaxation and autogenic training as treatments for insomnia. Journal of Abnormal Psychology 1974;83(3):253-60. [DOI] [PubMed] [Google Scholar]
Pearl 1956 {published data only}
- Pearl D, Vander Kamp H, Olsen AL, Greenberg PD, Armitage SG.The effects of reserpine on schizophrenic patients. American Journal of Psychiatry 1956;112:198-204. [DOI] [PubMed] [Google Scholar]
Peck 1976 {published data only}
- Peck CL.Desensitization for the treatment of fear in the high level adult retardate. Behaviour Research and Therapy 1977;15(2):137-48. [DOI] [PubMed] [Google Scholar]
Pelham 1992 {published data only}
- Milich R, Carlson CL, Pelham WE Jr, Licht BG.Effects of methylphenidate on the persistence of ADHD boys following failure experiences. Journal of Abnormal Child Psychology 1991;19(5):519-36. [DOI] [PubMed] [Google Scholar]
- Pelham WE, Murphy DA, Vannatta K, Milich R, Licht BG, Gnagy EM, et al.Methylphenidate and attributions in boys with attention-deficit hyperactivity disorder. Journal of Consulting and Clinical Psychology 1992;60(2):282-92. [DOI] [PubMed] [Google Scholar]
Pendleton 1983 {published data only}
- Pedleton MG, Higgins RL.A comparison of negative practice and systematic desensitization in the treatment of acrophobia. Journal of Behavior Therapy and Experimental Psychiatry 1983;14(4):317-23. [DOI] [PubMed] [Google Scholar]
Pillman 2001 {published data only}
- Peterson MA, Patterson B, Pillman BM, Battista MA.Cognitive recovery following alcohol detoxification: a computerised remediation study. Neuropsychological Rehabilitation 2002;12(1):63-74. [Google Scholar]
- Pillman BM.Computerized cognitive remediation with recently detoxified alcoholics. Dissertation, California School of Professional Psychology, Fresno Campus 2000.
Poland 2013 {published data only}
- Poland RE, Gertsik L, Favreau JT, Smith SI, Mirocha JM, Rao U, et al.Open-label randomized parallel-group controlled clinical trial of massage for treatment of depression in HIV-infected subjects. Journal of Alternative and Complementary Medicine 2013;19(4):334-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
Powers 2004 {published data only}
- Powers MB, Smits JA, Telch MJ.Disentangling the effects of safety-behavior utilization and safety-behavior availability during exposure-based treatment: a placebo-controlled trial. Journal of Consulting and Clinical Psychology 2004;72(3):448-54. [DOI] [PubMed] [Google Scholar]
Powers 2008a {published data only}
- Powers MB, Smiths JA, Whitley D, Bystritsky A, Telch MJ.The effect of attributional processes concerning medication taking on return of fear. Journal of Consulting and Clinical Psychology 2008;76(3):478-90. [DOI] [PubMed] [Google Scholar]
Powers 2008b {published data only}
- Powers MB, Smiths JA, Whitley D, Bystritsky A, Telch MJ.The effect of attributional processes concerning medication taking on return of fear. Journal of Consulting and Clinical Psychology 2008;76(3):478-90. [DOI] [PubMed] [Google Scholar]
Proudfoot 2013 {published data only}
- Proudfoot J, Clarke J, Birch MR, Whitton AE, Parker G, Manicavasagar V, et al.Impact of a mobile phone and web program on symptom and functional outcomes for people with mild- to-moderate depression, anxiety and stress: a randomized controlled trial. BMC Psychiatry 2013;13(312):1-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Proudfoot J.Re: Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 26th of August 2019.
Quayhagen 1995 {published data only}
- Quayhagen MP, Quayhagen M, Corbeil RR, Roth PA, Rodgers JA.A dyadic remediation program for care recipients with dementia. Nursing Research 1995;44(3):153-9. [PubMed] [Google Scholar]
Rabkin 1990 {published data only}
- Liebowitz M, Quitkin FM, Stewart JW, McGrath PJ, Harrison WM, Markowitz JS, et al.Antidepressant efficacy in atypical depression. Archives of General Psychiatry 1988;45(2):129-37. [DOI] [PubMed] [Google Scholar]
- Quitkin FM, McGrath PJ, Stewart JW, Harrison W, Wager SG, Nunes E, et al.Phenelzine and imipramine in mood reactive depressives: further delineation of the syndrome of atypical depression. Archives of General Psychiatry 1989;46(9):787-93. [DOI] [PubMed] [Google Scholar]
- Rabkin JG, McGrath PJ, Quitkin FM, Tricamo E, Stewart JW, Klein DF.Effects of pill-giving on maintenance of placebo response in patients with chronic mild depression. American Journal of Psychiatry 1990;147(12):1622-6. [DOI] [PubMed] [Google Scholar]
Rapee 2006 {published data only}
- Rapee R.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 29th of August 2019.
- Rapee RM, Abbott MJ, Lyneham HJ.Bibliotherapy for children With anxiety disorders using written materials for parents: a randomized controlled trial. Journal of Consulting and Clinical Psychology 2006;74(3):436-44. [DOI] [PubMed] [Google Scholar]
Rapee 2007 {published data only}
- Rapee R.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 29th of August 2019.
- Rapee RM, Abbott MJ, Baillie AJ, Gaston JE.Treatment of social phobia through pure self-help and therapist-augmented self-help. British Journal of Psychiatry 2007;191:246-52. [DOI] [PubMed] [Google Scholar]
Robin 1976 {published data only}
- Robin A.Rationing out-patients: a defence of the waiting list. British Journal of Psychiatry 1976;129:138-41. [DOI] [PubMed] [Google Scholar]
Roehrich 1993 {published data only}
- Roehrich L, Goldman MS.Experience-dependent neuropsychological recovery and the treatment of alcoholism. Journal of Consulting and Clinical Psychology 1993;61(5):812-21. [DOI] [PubMed] [Google Scholar]
Rosa‐Alcatraz 2009 {published data only}
- Olivares J.Program IAFS. Protocol for the treatment of social phobia in adolescents [Programa IAFS. Protocolo para el tratamiento de la fobia social enadolescentes]. Madrid: Pirámide 2005;1.
- Rosa-Alcázar AI, Olivares-Olivares PJ, Olivares J.The role of non-specific effects in the psychological treatment of adolescents with social phobia [Papel de los efectos inespecíficos en el tratamiento psicológico de adolescentes con fobia social]. Anuario de Psicologia 2009;40(1):43-61. [Google Scholar]
Rosen 1976 {published data only}
- Rosen GM, Glasgow RE, Barrera M.A controlled study to assess the clinical efficacy of totally self-administrated systematic desensitization. Journal of Consulting and Clinical Psychology 1976;44(2):208-17. [DOI] [PubMed] [Google Scholar]
Roth 1964 {published data only}
- Lorr M, McNair DM, Weinstein GJ.Early effects of chlordiazepoxide (Librium) used with psychotherapy. Journal of Psychiatric Research 1963;1:257-70. [DOI] [PubMed] [Google Scholar]
- Roth I, Rhudick PJ, Shaskan DA, Slobin MS, Wilkinson AE, Young HH.Long-term effects on psychotherapy of initial treatment conditions. Journal of Psychiatric Research 1964;2:283-97. [DOI] [PubMed] [Google Scholar]
Rupert 1978 {published data only}
- Rupert PA, Holmes DS.Effects of multiple sessions of true and placebo heart rate biofeedback training on the heart rates and anxiety levels of anxious patients during and following treatment. Psychophysiology 1978;15(6):582-90. [DOI] [PubMed] [Google Scholar]
Shalev 2012 {published data only}
- Shalev AY, Ankri Y, Isreali-Shalev Y, Peleg T, Adessky R, Freedman S.Prevention of posttraumatic stress disorder by early treatment. Archives of General Psychiatry 2012;69(2):166-76. [DOI] [PubMed] [Google Scholar]
Shealy 1979 {published data only}
- Shealy RC.The effectiveness of various treatment techniques on different degrees and durations of sleep-onset insomnia. Behaviour Research and Therapy 1979;17(6):541-6. [DOI] [PubMed] [Google Scholar]
Sibilio 1957 {published data only}
- Sibilio JP, Andrew G, Dart D, Moore KB, Stehman A.Treatment of chronic schizophrenia with promazine hydrochloride. AMA Archives of Neurology and Psychiatry 1957;78(4):419-24. [DOI] [PubMed] [Google Scholar]
Sommerness 1955 {published data only}
- Sommerness MD, Lucero RJ, Hamlon JS, Erickson JL, Matthews R.A controlled study of reserpine on chronically disturbed patients. JAMA Archives of Neurology and Psychiatry 1955;74(3):316-9. [DOI] [PubMed] [Google Scholar]
Steinmark 1974 {published data only}
- Steinmark SW, Borkovec TD.Active and placebo treatment effects on moderate insomnia under counterdemand and positive demand instructions. Journal of Abnormal Psychology 1974;83(2):157-63. [DOI] [PubMed] [Google Scholar]
Szymanski 1995 {published data only}
- Szymanski J, O'Donohue W.The potential role of state-dependent learning in cognitive therapy with spider phobics. Journal of Rational-Emotive & Cognitive-Behavior Therapy 1995;13(2):131-50. [Google Scholar]
- Szymanski J.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 20th of August 2019.
Tan 1986 {published data only}
- Tan SY, Bruni J.Cognitive-behavior therapy with adult patients with epilepsy: a controlled outcome study. Epilepsia 1986;27(3):225-33. [DOI] [PubMed] [Google Scholar]
Teri 1997 {published data only}
- Teri L, Logsdon RG, Uomoto J, McCurry SM.Behavioral treatment of depression in dementia patients: a controlled clinical trial. Journals of Gerontology. Series B, Psychological Sciences and Social Sciences 1997;52(4):159-66. [DOI] [PubMed] [Google Scholar]
- Teri L.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 26th of August 2019.
Tori 1973 {published data only}
- Tori C, Worell L.Reduction of human avoidant behavior. A comparison of counterconditioning, expectancy, and cognitive information approaches. Clinical Psychology 1973;41(2):269-78. [DOI] [PubMed] [Google Scholar]
Trexler 1972 {published data only}
- Trexler, L.Rational-emotive therapy, placebo, and no-treatment effects on public-speaking anxiety. Doctoral dissertation, Temple University AnnArbor, Mich.: University Microfilms 1971;71-31(063).
- Trexler LD, Karst TO.Rational-emotive therapy, placebo, and no-treatment effects on public-speaking anxiety. Journal of Abnormal Psyhology 1972;79(1):60-7. [DOI] [PubMed] [Google Scholar]
Turner 1979 {published data only}
- Ascher ML, Turner RM.A comparison of two methods for the administration of paradoxical intention. Behaviour Research and Therapy 1980;18(2):121-6. [DOI] [PubMed] [Google Scholar]
- Turner RM, Ascher LM.Controlled comparison of progressive relaxation, stimulus control, and paradoxical intention therapies for insomnia. Journal of Consulting and Clinical Psychology 1979;47(3):500-8. [DOI] [PubMed] [Google Scholar]
Vanderplate 1983 {published data only}
- Vanderplate C.Electromyograph biofeedback Treatment of sleep onset Insomnia: a controlled Investigation. Dissertation, University of Southern Mississippi 1978.
Watzl 1988 {published data only}
- Watzl H, Rist F, Olbrich R, Cohen R.Placebo injections and surveillance of alcohol intake during inpatient treatment of female alcoholics. Addictive Behaviors 1988;13(2):197-200. [DOI] [PubMed] [Google Scholar]
Weingaertner 1971 {published data only}
- Weingaertner AH.Self-administered aversive stimulation with hallucinating hospitalized schizophrenics. Journal of Consulting and Clinical Psychology 1971;36(3):422-29. [DOI] [PubMed] [Google Scholar]
Whittaker 1963 {published data only}
- Whittaker CB, Hoy RM.Withdrawal of perphenazine in chronic schizophrenia. British Journal of Psychiatry 1963;109:422-7. [DOI] [PubMed] [Google Scholar]
Wilson 1980 {published data only}
- Wilson A, Davidson WJ, Blanchard R, White J.Disulfiram implantation. A placebo-controlled trial with two-year follow-up. Journal of Studies on Alcohol 1978;39(5):809-19. [DOI] [PubMed] [Google Scholar]
- Wilson A, Davidson WJ, Blanchard R.Disulfiram implantation: a trial using placebo implants and two types of controls. Journal of Studies on Alcohol 1980;41(5):429-36. [DOI] [PubMed] [Google Scholar]
Wolitzky 2009 {published data only}
- Wolitzky KB, Telch MJ.Augmenting in vivo exposure with fear antagonistic actions: a preliminary test. Behavior Therapy 2009;40(1):57-71. [DOI] [PubMed] [Google Scholar]
Wollersheim 1991 {published data only}
- Wollersheim JP, Wilson GL.Group treatment of unipolar depression: a comparison of coping, supportive, bibliotherapy, and delayed treatment groups. Professional Psychology: Research and Practice 1991;22(6):496-502. [Google Scholar]
References to studies excluded from this review
Adelman 1990 {published data only}
- Adelman HS, MacDonald VM, Nelson P, Smith DC, Taylor L.Motivational readiness and the participation of children with learning and behavior problems in psychoeducational decision making. Journal of Learning Disabilities 1990;23(3):171-6. [DOI] [PubMed] [Google Scholar]
Altenhofer 2007 {published data only}
- Altenhöfer A, Schulz W, Schwab R, Eckert J.Psychotherapy of adjustment disorders. Is psychotherapy if limited to 12 sessions sufficiently effective? [Psychotherapie von Anpassungsstörungen. Ist eine auf 12 Sitzungen begrenzte Gesprächspsychotherapie ausreichend wirksam?]. Psychotherapeut 2007;52:24-34. [Google Scholar]
Andreasson 1990 {published data only}
- Andréasson S, Parmander M, Allebeck P.A pilot study of treatment of alcohol abuse. Marked improvement of alcohol drinking habits of both treated and untreated persons. Läkartidningen 1990;87(9):627-30. [PubMed] [Google Scholar]
Banasiak 2005 {published data only}
- Banasiak SJ, Paxton SJ, Hay P.Guided self-help for bulimia nervosa in primary care: a randomized controlled trial. Psychological Medicine 2005;35(9):1283-94. [DOI] [PubMed] [Google Scholar]
Bass 2016 {published data only}
- Bass J, Murray SM, Mohammed TA, Bunn M, Gorman W, Ahmed AM, et al.A randomized controlled trial of a trauma-informed support, skills, and psychoeducation intervention for survivors of torture and related trauma in Kurdistan, Northern Iraq. Global Health, Science and Practice 2016;4(3):452-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
de Jongh 1995 {published data only}
- Jongh A, Muris P, ter Horst G, Zuuren F, Schoenmakers N, Makkes P.One-session cognitive treatment of dental phobia: preparing dental phobics for treatment by restructuring negative cognition. Behaviour Research and Therapy 1995;33(8):947-54. [DOI] [PubMed] [Google Scholar]
Dolhanty 2005 {published data only}
- Dolhanty J.Symptom control and self-efficacy with electronic self-monitoring of bulimia nervosa: a randomized controlled trial. Dissertation, York University, North York, Ontario 2005.
Drozdek 2010 {published data only}
- Drožđek B, Bolwerk N.Evaluation of group therapy With traumatized asylum seekers and refugees—the Den Bosch model. Traumatology 2010;16(4):117-27. [Google Scholar]
Endicott 1964 {published data only}
- Endicott NA, Endicott J.Prediction of improvement in treated and untreated patients using the Rorschaf Prognostic Rating Scale. Journal of Consulting Psychology 1964;28:342-8. [DOI] [PubMed] [Google Scholar]
Ertl 2011 {published data only}
- Ertl V, Pfeiffer A, Schauer E, Elbert T, Neuner F.Community-implemented trauma therapy for former child soldiers in northern Uganda. JAMA 2011;306(5):503-12. [DOI] [PubMed] [Google Scholar]
Gitlin 2013 {published data only}
- Gitlin LN, Harris LF, McCoy M, Chernett NL, Jutkowitz E, Pizzi LT, et al.A community-integrated home based depression intervention for older African Americans: [corrected] Description of the Beat the Blues randomized trial and intervention costs. BMC Geriatrics 2012;10(4):1-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gitlin LN, Harris LF, McCoy MC, Chernett NL, Pizzi LT, Jutkowitz E, et al.A home-based intervention to reduce depressive symptoms and improve quality of life in older African Americans: a randomized trial. Annals of Internal Medicine 2013;159(4):243-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pizzi LT, Jutkowitz E, Frick KD, Suh D, Prioli KM, Gitlin LN.Cost-effectiveness of a community-integrated home based depression intervention in older African Americans. Journal of the American Geriatrics Society 2014;62(12):2288-95. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gournay 1994 {published data only}
- Gournay K, Brooking J.Community psychiatric nurses in primary health care. British Journal of Psychiatry 1994;162(2):231-8. [DOI] [PubMed] [Google Scholar]
Haertzen 1964 {published data only}
- Haertzen CA.On the addiction research center inventory scores of former addicts receiving LSD and untreated schizophrenics. Psychological Reports 1964;14(1):483-8. [Google Scholar]
Hartelius 1968 {published data only}
- Hartelius H.Objective trial of sedatives: comparison between Noludar, Mogadon, placebo, and "no tablet at all" [Objektiv sömnmedelsprövning: Jämförelse mellan Noludar, Mogadon, placebo och ingen tablett alls]. Nordisk Medicin 1968;79(25):811-3. [PubMed] [Google Scholar]
Hinsberger 2017 {published data only}
- Hinsberger M, Holtzhausen L, Sommer J, Kaminer D, Elbert T, Seedat S, et al.Feasibility and effectiveness of narrative exposure therapy and cognitive behavioral therapy in a context of ongoing violence in South Africa. Psychological Trauma: Theory, Research, Practice and Policy 2017;9(3):282-91. [DOI] [PubMed] [Google Scholar]
Keltner 1976 {published data only}
- Keltner L, Marshall WL.Attribution and subject control factors in experimental desensitization. Behavior Therapy 1976;7(5):626-33. [Google Scholar]
Laessle 1988 {published data only}
- Laessle RG, Waadt S, Schindler L, Schweiger U, Pirke M.Structured ambulatory group therapy in bulimia [Strukturierte ambulante Gruppentherapie bei Bulimie - Vorläufige Ergebnisse einer kontrollierten Therapiestudie]. PPmP Psychotherapie, Psychosomatik und Medizinische Psychologie 1988;38:324-7. [PubMed] [Google Scholar]
Litrownik 1978 {published data only}
- Litrownik AJ, Freitas JL, Franzini LR.Self-regulation in mentally retarded children: assessment and training of self-monitoring skills. American Journal of Mental Deficiency 1978;82(5):499-506. [PubMed] [Google Scholar]
Mak 2008 {published data only}
- Mak MK, Hui-Chan CW.Cued task-specific training is better than exercise in improving sit-to-stand in patients with Parkinson’s Disease: a randomized controlled trial. Movement Disorders : Official Journal of the Movement Disorder Society 2007;23(4):501-9. [DOI] [PubMed] [Google Scholar]
McCall 2005 {published data only}
- Mccall WV, Perlis ML, Tu X, Groman AE, Krystal A, Walsh JK.A comparison of placebo and no-treatment during a hypnotic clinical trial. International Journal of Clinical Pharmacology and Therapeutics 2005;43(8):355-9. [DOI] [PubMed] [Google Scholar]
McNeil 1991 {published data only}
- McNeil JK, LeBlanc EM, Joyner M.The effect of exercise on depressive symptoms in the moderately depressed elderly. Psychology and Aging 1991;6(3):487-8. [DOI] [PubMed] [Google Scholar]
Otteson 1979 {published data only}
- Otteson JP.Curative caring: the use of buddy groups with chronic schizophrenics. Journal of Consulting and Clinical Psychology 1979;47(3):649-51. [DOI] [PubMed] [Google Scholar]
Propst 1992 {published data only}
- Propst LR, Ostrom R, Watkins P, Dean T, Mashburn D.Comparative efficacy of religious and nonreligious cognitive-behavioral therapy for the treatment of clinical depression in religious individuals. Journal of Consulting and Clinical Psychology 1992;60(1):94-103. [DOI] [PubMed] [Google Scholar]
Sackley 2018 {published data only}
- Sackley CM, Smith CH, Rick CE, Brady MC, Ives N, Patel S, et al.Lee Silverman Voice treatment versus standard speech and language therapy versus control in Parkinson’s disease: a pilot randomized controlled trial (PD COMM pilot). Pilot and Feasibility Studies 2018;4(30):1-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
Salt 2002 {published data only}
- Salt J, Shemilt J, Sellars V, Boyd S, Coulson T, McCool S.The Scottish Centre for autism preschool treatment programme: II. The results of a controlled treatment outcome study. International Journal of Research and Practice 2002;6(1):33-46. [DOI] [PubMed] [Google Scholar]
Schultz 2002 {published data only}
- Schultz DE.The effectiveness of stress management on anxiety and depression. Doctor’s Dissertation, California School of Professional Psychology Fresno Campus 2002.
Sheikh 1986 {published data only}
- Sheikh JI, Hill RD, Yesavage JA.Long-term efficacy of cognitive training for age-associated memory impairment: a six-month follow-up study. Developmental Neuropsychology 1986;2(4):413-21. [Google Scholar]
Steiner 2011 {published data only}
- Steiner NJ, Sheldrick RC, Gotthelf D, Perrin EC.Computer-based attention training in the schools for children with Attention-Deficit/Hyperactivity Disorder: a preliminary trial. Clinical Pediatrics 2011;50(7):615-22. [DOI] [PubMed] [Google Scholar]
Timko 1995 {published data only}
- Timko C, Moos RH, Finney JW, Moos BS, Kaplowitz MS.Long-term treatment careers and outcomes of previously untreated alcoholics. Journal of Studies on Alcohol 1995;60(4):437-47. [DOI] [PubMed] [Google Scholar]
Tomporowski 1984 {published data only}
- Tomporowski PD, Ellis NR.Effects of exercise on the physical fitness, intelligence, and adaptive behavior of institutionalized mentally retarded adults. Applied Research in Mental Retardation 1984;5(3):329-37. [DOI] [PubMed] [Google Scholar]
Torkan 2014 {published data only}
- Torkan H, Blackwell SE, Holmes EA, Kalantari M, Neshat-Doost HT, Maroufi M, et al.Positive imagery cognitive bias modification in treatment-seeking patients with major depression in Iran: a pilot study. Cognitive Therapy and Research 2014;38:132-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
Weber 1975 {published data only}
- Weber BA, Sulzbacher SI.Use of CNS stimulant medication in averaged electroencephalic audiometry with children with MBD. Journal of Learning Disabilities 1975;8(5):44-7. [Google Scholar]
Wilson 2002 {published data only}
- Wilson PH, Thomas PR, Maruff P.Motor imagery training ameliorates motor clumsiness in children. Journal of Child Neurology 2002;17(7):491-8. [DOI] [PubMed] [Google Scholar]
References to studies awaiting assessment
Bommert 1978 {published data only}
Brandes 2010 {published data only}
McLachlan 1993 {published data only}
Newton‐Cross 2017 {published data only}
Schwarzler 1999 {published data only}
Trianes Torres 1991 {published data only}
References to ongoing studies
Heitman 2017 {published data only}
- Internet-based attentional bias modification training as add-on to regular treatment in alcohol and cannabis dependent outpatients: a study protocol of a randomised control trial. Ongoing study. 18/05/2015. Contact author for more information. [DOI] [PMC free article] [PubMed]
ISRCTN21392756 {published data only}
- Standardised stress management mental health training: does it have a beneficial effect?. Ongoing study. 06/09/2010. Contact author for more information.
ISRCTN35717198 {published data only}
- Cognitive training with non-invasive brain stimulation to treat binge eating disorder. Ongoing study. 03/05/2018. Contact author for more information.
NCT00044629 {published data only}
- Combined behavioral/pharmacologicaltTherapy for insomnia. Ongoing study. 05/09/2002. Contact author for more information.
Additional references
Abikoff 2004 (pers comm)
- Abikoff H.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 23rd of August 2019.
Allen 1998 (pers comm)
- Allen JB.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 28th of August 2019.
Allen 2006 (pers comm)
- Allen JB.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 28th of August 2019.
Allen 2018
- Allen EN, Chandler CI, Mandimika N, Leisegang C, Barnes K.Eliciting adverse effects data from participants in clinical trials. Cochrane Database of Systematic Reviews 2018, Issue 1. Art. No: MR000039. [DOI: 10.1002/14651858.MR000039.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Andrews 2013a
- Andrews J, Guyatt G, Oxman AD, Alderson P, Dahm P, Falck-Ytter Y, et al.GRADE guidelines: 14. Going from evidence to recommendations: the significance and presentation of recommendations. Journal of Clinical Epidemiology 2013;66(7):719-25. [DOI] [PubMed] [Google Scholar]
Andrews 2013b
- Andrews JC, Schünemann HJ, Oxman AD, Pottie K, Meerpohl JJ, Coello PA, et al.GRADE guidelines: 15. Going from evidence to recommendation - determinants of a recommendation's direction and strength. Journal of Clinical Epidemiology 2013;66(7):726-35. [DOI] [PubMed] [Google Scholar]
APA 1952
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders. 1st edition. Washington (DC): American Psychiatric Association, 1952. [Google Scholar]
APA 1968
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders. 2nd edition. Washington (DC): American Psychiatric Association, 1968. [Google Scholar]
APA 1980
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders. 3rd edition. Washington (DC): American Psychiatric Association, 1980. [Google Scholar]
APA 1987
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders. 3rd revised edition. Washington (DC): American Psychiatric Association, 1987. [Google Scholar]
APA 1994
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders. 4th edition. Washington (DC): American Psychiatric Association, 1994. [Google Scholar]
APA 2000
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders. 4th text revision edition. Washington (DC): American Psychiatric Association, 2000. [Google Scholar]
APA 2013
- American Psychiatric Association.Diagnostic and Statistical Manual of Mental Disorders. 5th edition. Washington (DC): American Psychiatric Association, 2013. [https://www.psychiatry.org/psychiatrists/practice/dsm] [Google Scholar]
Ayen 2004 (pers comm)
- Ayen I.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: EG Faltinsen 9th of September 2019.
Balshem 2018
- Balshem H, Helfand M, Schünemann HJ, Oxman AD, Kunz R, Brozek J, et al.GRADE guidelines: 3. Rating the quality of evidence. Journal of Clinical Epidemiology 2018;64(4):401-6. [DOI] [PubMed] [Google Scholar]
Bateman 2009
- Bateman A, Fonagy P.Randomized controlled trial of outpatient mentalization-based treatment versus structured clinical management for borderline personality disorder. American Journal of Psychiatry 2009;166(12):1355-64. [DOI] [PubMed] [Google Scholar]
Bateman 2017
- Bateman A, Campbell C, Luyten P, Fonagy P.A mentalization-based approach to common factors in the treatment of borderline personality disorder. Current Opinion in Psychology 2017;21:44-9. [DOI] [PubMed] [Google Scholar]
Berkey 1996
- Berkey CS, Mosteller F, Lau J, Antman EM.Uncertainty of the time of first significance in random effects cumulative meta-analysis. Controlled Clinical Trials 1996;17(5):357-71. [DOI] [PubMed] [Google Scholar]
Borduin 2009
- Borduin CM, Schaeffer CM, Heiblum N.A randomized clinical trial of multisystemic therapy with juvenile sexual offenders: effects on youth social ecology and criminal activity. Journal of Consulting and Clinical Psychology 2009;77(1):26-37. [DOI] [PubMed] [Google Scholar]
Borkovec 2005
- Borkovec TD, Sibrava NJ.Problems with the use of placebo conditions in psychotherapy research, suggested alternatives, and some strategies for the pursuit of the placebo phenomenon. Journal of Clinical Psychology 2005;61(7):805-18. [DOI] [PubMed] [Google Scholar]
Bornovalova 2008 (pers comm)
- Bornovalova M.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 21st of August 2019.
Boutron 2021
- Boutron I, Page MJ, Higgins JP, Altman DG, Lundh A, Hróbjartsson A.Chapter 7: Considering bias and conflicts of interest among the included studies. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editors(s). Cochrane Handbook for Systematic Reviews of Interventions version 6.2. Cochrane, 2021. [Google Scholar]
Bramer 2017
- Bramer WM, Rethlefsen ML, Kleijnen J, Franco OH.Optimal database combinations for literature searches in systematic reviews: a prospective exploratory study. Systematic Reviews 2017;6:245. [DOI] [PMC free article] [PubMed] [Google Scholar]
Brok 2008
- Brok J, Thorlund K, Gluud C, Wetterslev J.Trial sequential analysis reveals insufficient information size and potentially false positive results in many meta-analyses. Journal of Clinical Epidemiology 2008;61(8):763-9. [DOI] [PubMed] [Google Scholar]
Brok 2009
- Brok J, Thorlund K, Wetterslev J, Gluud C.Apparently conclusive meta-analyses may be inconclusive -trial sequential analysis adjustment of random error risk due to repetitive testing of accumulating data in apparently conclusive neonatal meta-analyses. International Journal of Epidemiology 2009;38(1):287-98. [DOI] [PubMed] [Google Scholar]
Brunetti 2013
- Brunetti M, Shemilt I, Pregno S, Vale L, Oxman AD, Lord J, et al.GRADE guidelines: 10. Considering resource use and rating the quality of economic evidence. Journal of Clinical Epidemiology 2013;66(2):140-50. [DOI] [PubMed] [Google Scholar]
Castellini 2018
- Castellini G, Bruschettini M, Gianola S, Gluud C, Moja L.Assessing imprecision in Cochrane systematic reviews: a comparison of GRADE and Trial Sequential Analysis. Systematic Reviews 2018;7(1):110. [DOI] [PMC free article] [PubMed] [Google Scholar]
Chalmers 1983
- Chalmers TC, Celano P, Sacks HS, Smith H Jr.Bias in treatment assignment in controlled clinical trials. New England Journal of Medicine 1983;309(22):1358-61. [DOI] [PubMed] [Google Scholar]
Chanen 2008
- Chanen AM, Jackson HJ, McCutcheon LK, Jovev M, Dudgeon P, Hok PY, et al.Early intervention for adolescents with borderline personality disorder using cognitive analytic therapy: randomised controlled trial. British Journal of Psychiatry 2008;193(6):477-84. [DOI] [PubMed] [Google Scholar]
Charlesworth 2017
- Charlesworth JE, Petkovic G, Kelley JM, Hunter M, Onakpoya I, Roberts N, et al.Effects of placebos without deception compared with no treatment: a systematic review and meta-analysis. Journal of Evidence Based Medicine 2017;10(2):97-107. [DOI] [PubMed] [Google Scholar]
Cohen1988
- Cohen J.Statistical Power Analysis for the Behavioral Sciences. 2nd edition.. Hillsdale (NJ): Lawrence Erlbaum Associates, Publishers, 1988. [Google Scholar]
Colloca 2020
- Colloca L, Barsky AJ.Placebo and nocebo effects. New England Journal of Medicine 2020;382(6):554-61. [DOI] [PubMed] [Google Scholar]
Comer 2013
- Comer JS, Kendall PC.Methodology, design, and evaluation in psychotherapy research. In: Lambert MJ, editors(s). Bergin and Garfields’s Handbook of Psychotherapy and Behavior Change. 6th edition. New York: Wiley, 2013:21-48. [Google Scholar]
Copenhagen Trial Unit 2018
- TSA - Trial sequential analysis. Copenhagen Trial Unit. [www.ctu.dk/tsa (accessed April 23 2018)]
Cristea 2017
- Cristea IA, Gentili C, Cotet CD, Palomba D, Barbui C, Cuijpers P.Efficacy of psychotherapies for borderline personality disorder: A systematic review and meta-analysis. JAMA Psychiatry 2017;74(4):319-28. [DOI] [PubMed] [Google Scholar]
Cuijpers 2016
- Cuijpers P, Cristea IA.How to prove that your therapy is effective, even when it is not: a guideline. Epidemiology and Psychiatric Sciences 2016;25(5):428-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
Cuijpers 2021
- Cuijpers P, Quero S, Papola D, Cristea IA, Karyotaki E.Care-as-usual control groups across different settings in randomized trials on psychotherapy for adult depression: a meta-analysis. Psychological Medicine 2021;51(4):634–44. [DOI] [PubMed] [Google Scholar]
Cunningham 2013
- Cunningham JA, Kypri K, McCambridge J.Exploratory randomized controlled trial evaluating the impact of a waiting list control design. BMC Medical Research Methodology 2013;13(1):150. [DOI] [PMC free article] [PubMed] [Google Scholar]
da Costa 2013
- da Costa BR, Nüesch E, Rutjes AW, Johnston BC, Reichenbach S, Trelle S, et al.Combining follow-up and change data is valid in meta-analyses of continuous outcomes: a meta-epidemiological study. Journal of Clinical Epidemiology 2013;66(8):847-55. [DOI] [PubMed] [Google Scholar]
Donner 2002
- Donner A, Klar N.Issues in the meta-analysis of cluster randomized trials. Statistics in Medicine 2002;21(19):2971-80. [DOI] [PubMed] [Google Scholar]
Egger 1997
- Egger M, Smith GD, Phillips AN.Meta-analysis: principles and procedures. BMJ 1997;315(7121):1533-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Eldridge 2016
- Eldrige S, Campbell M, Campbell M, Dahota A, Giraudeau B, Higgins J, et al.Revised Cochrane risk of bias tool for randomized trials (RoB 2.0) Additional considerations for cluster-randomized trials. University of Bristol 2016.
Erlen 2015
- Erlen JA, Tamres LK, Reynolds N, Golin CE, Rosen MI, Remien RH, et al.Assessing usual care in clinical trials. Western Journal of Nursing Research 2015;37(3):288-98. [DOI] [PMC free article] [PubMed]
Espie 1989b
- Espie CA, Brooks DN, Lindsay WR.An evaluation of tailored psychological treatment of insomnia. Journal of Behavior Therapy and Experimental Psychiatry 1989;20(2):143-53. [DOI] [PubMed] [Google Scholar]
Fässler 2015
- Fässler M, Meissner K, Kleijnen J, Hróbjartsson A, Linde K.A systematic review found no consistent difference in effect between more and less intensive placebo interventions. Journal of Clinical Epidemiology 2015;68(4):442-51. [DOI] [PubMed] [Google Scholar]
Frank 1991
- Frank JD, Frank JB.Persuasion and Healing: a Comparative Study of Psychotherapy. 3rd edition. Baltimore: Johns Hopkins University Press, 1991. [Google Scholar]
Freedland 2011
- Freedland KE, Mohr DC, Davidson KW, Schwartz JE.Usual and unusual care: existing practice control groups in randomized controlled trials of behavioral interventions. Psychosomatic Medicine 2011;73(4):323-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
Furukawa 2014
- Furukawa TA, Noma H, Caldwell DM, Honyashiki M, Shinohara K, Imai H, et al.Waiting list may be a nocebo condition in psychotherapy trials: a contribution from network meta-analysis. Acta Psychiatrica Scandinavica 2014;130(3):181-92. [DOI] [PubMed] [Google Scholar]
Gold 2018
- Gold SM, Enck P, Hasselmann H, Friede T, Hegerl U, Mohr DC, et al.Control conditions for randomised trials of behavioural interventions in psychiatry: a decision framework. Lancet Psychiatry 2018;4(9):725-32. [DOI] [PubMed] [Google Scholar]
GRADE Working Group 2004
- GRADE Working Group.Grading quality of evidence and strength of recommendations. BMJ 2004;328(7454):1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
Greville‐Harris 2015
- Greville-Harris M, Dieppe P.Bad is more powerful than good: the nocebo response in medical consultations. American Journal of Medicine 2015;128(2):126-9. [DOI] [PubMed] [Google Scholar]
Guidi 2018
- Guidi J, Brakemeier EL, Bockting CL, Cosci F, Cuijpers P, Jarrett RB, et al.Methodological recommendations for trials of psychological interventions. Psychotherapy and Psychosomatics 2019;87(5):276-84. [DOI] [PubMed] [Google Scholar]
Guyatt 2011a
- Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al.GRADE guidelines: 1. Introduction - GRADE evidence profiles and summary of findings tables. Journal of Clinical Epidemiology 2011;64(4):383-94. [DOI] [PubMed] [Google Scholar]
Guyatt 2011b
- Guyatt GH, Oxman AD, Kunz R, Atkins D, Brozek J, Vist G, et al.GRADE guidelines: 2. Framing the question and deciding on important outcomes. Journal of Clinical Epidemiology 2011;64(4):395-400. [DOI] [PubMed] [Google Scholar]
Guyatt 2011c
- Guyatt GH, Oxman AD, Vist G, Kunz R, Brozek J, Alonso-Coello P, et al.GRADE guidelines: 4. Rating the quality of evidence - study limitations (risk of bias). Journal of Clinical Epidemiology 2011;64(4):407-15. [DOI] [PubMed] [Google Scholar]
Guyatt 2011d
- Guyatt GH, Oxman AD, Montori V, Vist G, Kunz R, Brozek J, et al.GRADE guidelines: 5. Rating the quality of evidence - publication bias. Journal of Clinical Epidemiology 2011;64(12):1277-82. [DOI] [PubMed] [Google Scholar]
Guyatt 2011e
- Guyatt GH, Oxman AD, Kunz R, Brozek J, Alonso-Coello P, Rind D, et al.GRADE guidelines 6. Rating the quality of evidence - imprecision. Journal of Clinical Epidemiology 2011;64(12):1283-93. [DOI] [PubMed] [Google Scholar]
Guyatt 2011f
- Guyatt GH, Oxman AD, Kunz R, Woodcock J, Brozek J, Helfand, et al.GRADE guidelines: 7. Rating the quality of evidence - inconsistency. Journal of Clinical Epidemiology 2011;64(12):1294-302. [DOI] [PubMed] [Google Scholar]
Guyatt 2011g
- Guyatt GH, Oxman AD, Kunz R, Woodcock J, Brozek J, Helfand M, et al.GRADE guidelines: 8. Rating the quality of evidence - indirectness. Journal of Clinical Epidemiology 2011;64(12):1303-10. [DOI] [PubMed] [Google Scholar]
Guyatt 2011h
- Guyatt GH, Oxman AD, Sultan S, Glasziou P, Akl EA, Alonso-Coello P, et al.GRADE guidelines: 9. Rating up the quality of evidence. Journal of Clinical Epidemiology 2011;64(12):1311-6. [DOI] [PubMed] [Google Scholar]
Guyatt 2013a
- Guyatt G, Oxman AD, Sultan S, Brozek J, Glasziou P, Alonso-Coello P, et al.GRADE guidelines: 11. Making an overall rating of confidence in effect estimates for a single outcome and for all outcomes. Journal of Clinical Epidemiology 2013;66(2):151-7. [DOI] [PubMed] [Google Scholar]
Guyatt 2013b
- Guyatt GH, Oxman AD, Santesso N, Helfand M, Vist G, Kunz R, et al.GRADE guidelines: 12. Preparing Summary of Findings tables - binary outcomes. Journal of Clinical Epidemiology 2013;66(2):158-72. [DOI] [PubMed] [Google Scholar]
Hafliðadóttir 2021
- Hafliðadóttir SH, Juhl CB, Nielsen SM, Henriksen M, Harris IA, Bliddal H, et al.Placebo response and effect in randomized clinical trials: meta-research with focus on contextual effects. Trials 2021;22(1):493. [DOI] [PMC free article] [PubMed] [Google Scholar]
Heitmann 2017 (pers comm)
- Heitmann J.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 5th of February 2019.
Higgins 2003
- Higgins JP, Thompson SG, Deeks JJ, Altman DG.Measuring inconsistency in meta-analyses. BMJ 2003;327(7414):557-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgin JP, Green S, editors.Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. The Cochrane Collaboration, 2011. [Available from handbook.cochrane.org.]
Higgins 2017
- Higgins JP, Altman DG, Sterne JA.Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Green S, Editors. Cochrane Handbook for Systematic Reviews of Interventions version 5.1.0 (updated June 2017). The Cochrane Collaboration, 2017. Available from www.training.cochrane.org/handbook.
Higgins 2019
- Higgin JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al (editors).Cochrane Handbook for Systematic Reviews of Interventions Version 6.0 (updated July 2019). Cochrane, 2019.. Available from www.training.cochrane.org/handbook.
Hoffmann 2013
- Hoffmann TC, Erueti C, Glasziou PP.Poor description of non-pharmacological interventions: analysis of consecutive sample of randomised trials. BMJ 2013;347(7924):e1-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
Holmes 2016
- Holmes RD, Tiwari AK, Kennedy JL.Mechanisms of the placebo effect in pain and psychiatric disorders. Pharmacogenomics Journal 2016;16(6):491. [DOI] [PubMed] [Google Scholar]
Holmes 2018
- Holmes EA, Ghaderi A, Harmer CJ, Ramchandani PG, Cuijpers P, Morrison AP, et al.The Lancet Psychiatry Commission on psychological treatments research in tomorrow's science. Lancet Psychiatry 2018;5(3):237-86. [DOI] [PubMed] [Google Scholar]
Howick 2019
- Howick J, Webster R, Kirby N, Hood K.Rapid overview of systematic reviews of nocebo effects reported by patients taking placebos in clinical trials. Trials 2018;19(1):674. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hróbjartsson 2001
- Hróbjartsson A, Gøtzsche PC.Is the placebo powerless? An analysis of clinical trials comparing placebo with no treatment. New England Journal of Medicine 2001;344:1594-602. [DOI] [PubMed] [Google Scholar]
Hróbjartsson 2002
- Hróbjartsson A, Götzsche PC.Placebo treatment versus no treatment. Cochrane Database of Systematic Reviews 2002, Issue 4. Art. No: CD003974. [DOI: 10.1002/14651858.CD003974] [DOI] [PubMed] [Google Scholar]
Hróbjartsson 2004
- Hróbjartsson A, Götzsche PC.Placebo interventions for all clinical conditions. Cochrane Database of Systematic Reviews 2004, Issue 2. Art. No: CD003974. [DOI: 10.1002/14651858.CD003974.pub2] [DOI] [PubMed] [Google Scholar]
Hróbjartsson 2010
- Hróbjartsson A, Götzsche PC.Placebo interventions for all clinical conditions. Cochrane Database of Systematic Reviews 2010, Issue 1. Art. No: CD003974. [DOI: 10.1002/14651858.CD003974.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Hróbjartsson 2012
- Hróbjartsson A, Miller FG.Placebo in nonpharmacological randomized trials. In: Boutron I, Ravaud P, Moher D, editors(s). Randomized Clincial Trials of Nonpharmacological Treatments. Taylor & Francis Group, LLC, 2012:11-26. [Google Scholar]
ICH 2005
- ICH Expert Working Group.International conference on harmonisation of technical requirements for registration of pharmaceuticals for human use. ICH harmonised tripartite guideline. Guideline for good clinical practice E6(R1). www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Efficacy/E6/E6_R1_Guideline.pdf (accessed 24 April 2018).
Institute of Medicine 2009
- Lo B, Field MJ.Institute of Medicine (US) Committee on Conflict of Interest in Medical Research, Education, and Practice. Conflict of Interest in Medical Research, Education, and Practice. Washington DC: National Academies Press (US), 2009. [PubMed] [Google Scholar]
Jakobsen 2014
- Jakobsen JC, Wetterslev J, Winkel P, Lange T, Gluud C.Thresholds for statistical and clinical significance in systematic reviews with meta-analytic methods. BMC Medical Research Methodology 2014;14(1):120. [DOI] [PMC free article] [PubMed] [Google Scholar]
Jensen 2017
- Jensen KB, Kirsch I, Pontén M, Rosén A, Yang K, Gollub RL, et al.Certainty of genuine treatment increases drug responses among intellectually disabled patients. Neurology 2017;88(20):1912-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Juul 2020
Karst 2007 (pers comm)
- Karst M.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: EG Faltinsen 5th of September 2019.
Karterud 2020
- Karterud S, Kongerslev MT.Psychotherapy of personality disorders needs an integrative theory of personality. Journal of Psychotherapy Integration 2020;31:34-53. [Google Scholar]
Kazdin 2015
- Kazdin AE.Treatment as usual and routine care in research and clinical practice. Clinical Psychology Review 2015;42:168. [DOI] [PubMed] [Google Scholar]
Kazdin 2016
- Kazdin AE.Methodological Issues & Strategies in Clinical Research. 4th edition. Washington DC: American Psychological Association, 2016. [Google Scholar]
Kelley 2012 (pers comm)
- Kelley J.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 12th of August 2019.
Kienle 1997
- Kienle GS, Kiene H.The powerful placebo effect - fact or fiction? Journal of Clinical Epidemiology 1997;50(12):1311-8. [DOI] [PubMed] [Google Scholar]
Kilmann 1985
- Kilmann PR, Milan RJ, Boland JP, Nankin HR, Sablis RF, West MO, et al.The sexual interactions of men with secondary erectile dysfunction. Paper presented at the annual meeting of the American Psychological Association in Los Angeles 1985.
Kilmann 1988
- Kilmann PR, Milan RJ, Boland JP, Davidson E, West MO, Sablis RF et al.The sexual interactions of men with secondary erectile dysfunction. Journal of Sex Education and Therapy 1988;14(1):46-7. [Google Scholar]
Kjaergard 2001
- Kjaergard LL, Villumsen J, Gluud C.Reported methodologic quality and discrepancies between large and small randomized trials in meta-analyses. Annals of Internal Medicine 2001;135(11):982. [DOI] [PubMed] [Google Scholar]
Kongerslev 2015
- Kongerslev MT, Chanen AM, Simonsen E.Personality disorder in childhood and adolescence comes of age: a review of the current evidence and prospects for future research. Scandinavian Journal of Child and Adolescent Psychiatry and Psychology 2015;3(1):31-48. [Google Scholar]
Kube 2017
- Kube T, Rief W.Are placebo and drug-specific effects additive? Questioning basic assumptions of double-blinded randomized clinical trials and presenting novel study designs. Drug Discovery Today 2017;22:729-35. [DOI] [PubMed] [Google Scholar]
Kwan 2014
- Kwan RY, Leung MC, Lai CK.Acupressure for agitation in nursing home residents with dementia:study protocol for a randomized controlled trial. Trials 2014;15(410):1-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kwan 2017 (pers comm)
- Kwan R.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 26th of August 2019.
Lai 2004 (pers comm)
- Lai K.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 6th of September 2019.
Legrand 2016 (pers comm)
- Legrand F.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 14th of August 2019.
Leichsenring 2019
- Leichsenring F, Steinert C, Ioannidis JPA.Toward a paradigm shift in treatment and research of mental disorders. Psychological Medicine 2019;49(13):2111-7. [DOI] [PubMed] [Google Scholar]
Lilienfeld 2007
- Lilienfeld SO.Psychological treatments that cause harm. Perspectives on Psychological Science 2007;2(1):53-70. [DOI] [PubMed] [Google Scholar]
Linden 2014
- Linden M, Schermuly-Haupt M.Definition, assessment and rate of psychotherapy side effects. World Psychiatry 2014;13(3):306-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Locher 2018
- Locher C, Gaab J, Blease C.When a placebo is not a placebo: problems and solutions to the gold standard in psychotherapy research. Frontiers in Psychology 2018;9:2317. [DOI] [PMC free article] [PubMed] [Google Scholar]
Löfholm 2013
- Löfholm AC, Brännström L, Olsson M, Hansson K.Treatment-as-usual in effectiveness studies: What is it and does it matter? International Journal of Social Welfare 2013;22(1):25-34. [Google Scholar]
Lund 2014
- Lund K, Vase L, Petersen GL, Jensen TS, Finnerup NB.Randomised controlled trials may underestimate drug effects: balanced placebo trial design. PLOS One 2014;9(1):1-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Lundh 2017
- Lundh A, Lexchin J, Mintzes B, Schroll JB, Bero L.Industry sponsorship and research outcome. Cochrane Database of Systematic Reviews 2017, Issue 2. Art. No: MR000033. [DOI: 10.1002/14651858.MR000033.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Meissner 2011
- Meissner K, Bingel U, Colloca L, Wager TD, Watson A, Flaten MA.The placebo effect: advances from different methodological approaches. Journal of Neuroscience 2011;31(45):16117-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
Michopoulos 2021
- Michopoulos I, Furukawa TA, Noma H, Kishimoto S, Onishi A, Ostinelli EG, et al.Different control conditions can produce different effect estimates in psychotherapy trials for depression. Journal of Clinical Epidemiology 2021;132:59–70. [DOI] [PubMed] [Google Scholar]
Milich 1991
- Milich R, Carlson CL, Pelham WE Jr, Licht BG.Effects of methylphenidate on the persistence of ADHD boys following failure experiences. Journal of Abnormal Child Psychology 1991;19(5):519-36. [DOI] [PubMed] [Google Scholar]
Miranda 1997 (pers comm)
- Miranda J.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: EG Faltinsen 9th of September 2019.
Miranda 2003
- Miranda J, Chung JY, Green BL, Krupnick J, Siddique J, Revicki DA, et al.Treating depression in predominantly low-income young minority women: a randomized controlled trial. JAMA 2003;290(1):57-65. [DOI] [PubMed] [Google Scholar]
Moher 1998
- Moher M, Moher D, Pham B, Jones Al, Cook DJ, Jadad AR, et al.Does quality of reports of randomised trials affect estimates of intervention efficacy reported in meta-analyses? Lancet 1998;352(9128):609-13. [DOI] [PubMed] [Google Scholar]
Moher 2009
- Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group.Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ (Clinical Research Ed.) 2009;339:b2535. [DOI: 10.1136/bmj.b2535] [DOI] [PMC free article] [PubMed] [Google Scholar]
Mohr 2009
- Mohr DC, Spring B, Freedland KE, Beckner V, Arean P, Hollon SD, et al.The selection and design of control conditions for randomized controlled trials of psychological interventions. Psychotherapy and Psychosomatics 2009;78(5):275-84. [DOI] [PubMed] [Google Scholar]
Mohr 2014
- Mohr DC, Ho J, Hart TL, Baron KG, Berendsen M, Beckner V, et al.Control condition design and implementation features in controlled trials: a meta-analysis of trials evaluating psychotherapy for depression. Translational Behavioral Medicine 2014;4(4):407-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
Munder 2013
- Munder T, Brütsch O, Leonhart R, Gerger H, Barth J.Researcher allegiance in psychotherapy outcome research: an overview of reviews. Clinical Psychology Review 2013;33(4):501-11. [DOI] [PubMed] [Google Scholar]
Munder 2018
- Munder T, Barth J.Cochrane’s risk of bias tool in the context of psychotherapy outcome research. Psychotherapy Research 2018;28(3):347-55. [DOI] [PubMed] [Google Scholar]
Mustafa 2013
- Mustafa RA, Santesso N, Brozek J, Akl EA, Walter SD, Norman G, et al.The GRADE approach is reproducible in assessing the quality of evidence of quantitative evidence syntheses. Journal of Clinical Epidemiology 2013;66(7):736-42.e5. [DOI] [PubMed] [Google Scholar]
Norman 2003
- Norman GR, Sloan JA, Wyrwich KW.Interpretation of changes in health-related quality of life: the remarkable universality of half a standard deviation. Medical Care 2003;41(5):582-92. [DOI] [PubMed] [Google Scholar]
O'Leary 1978
- O'Leary KD, Borkovec TD.Conceptual, methodological, and ethical problems of placebo groups in psychotherapy research. American Psychologist 1978;33(9):821-30. [DOI] [PubMed] [Google Scholar]
Pagsberg 2017
- Pagsberg AK, Jeppesen P, Klauber DG, Jensen KG, Rudå D, Stentebjerg-Oleson M, et al.Quetiapine extended release versus aripiprazole in children and adolescents with first-episode psychosis: the multicentre, double-blind, randomised tolerability and efficacy of antipsychotics (TEA) trial. Lancet Psychiatry 2017;4(8):605-18. [DOI] [PubMed] [Google Scholar]
Peterson 2002
- Peterson MA, Patterson B, Pillman BM, Battista MA.Cognitive recovery following alcohol detoxification: A computerised remediation study. Neuropsychological Rehabilitation 2002;12(1):63-74. [Google Scholar]
Proudfoot 2013 (pers comm)
- Proudfoot J.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 26th of August 2019.
Rapee 2006 (pers comm)
- Rapee R.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 29th of August 2019.
Rapee 2007 (pers comm)
- Rapee R.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 29th of August 2019.
Review Manager 5 [Computer program]
- Review Manager (RevMan 5).Version 5.3. Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Rosenberg 2014
- Rosenberg L.Assisted outpatient treatment: we can do better. Journal of Behavioral Health Services and Research 2014;41(3):251-3. [DOI] [PubMed] [Google Scholar]
Rosenzweig 1936
- Rosenzweig S.Some implicit common factors in diverse methods of psychotherapy. American Journal of Orthopsychiatry 1936;6(3):412-5. [Google Scholar]
Rutherford 2014
- Rutherford BR, Pott E, Tandler JM, Wall MM, Roose SP, Lieberman JA.Placebo response in antipsychotic clinical trials: A meta-analysis. JAMA Psychiatry 2014;71(12):1409-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
Savović 2012
- Savović J, Jones H, Altman D, Harris R, Jüni P, Pildal J, et al.Influence of reported study design characteristics on intervention effect estimates from randomised controlled trials: combined analysis of meta-epidemiological studies. Health Technology Assessment 2012;16(35):1-82. [DOI] [PubMed] [Google Scholar]
Savovic 2018
- Savovic J, Turner RM, Mawdsley D, Jones HE, Beynon R, Higgins JP, et al.Association between Risk-of-Bias assessments and results of randomized trials in Cochrane reviews: the ROBES meta-epidemiologic study. American Journal of Epidemiology 2018;187(5):1113-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
Scherer 2018
- Scherer RW, Meerpohl JJ, Pfeifer N, Schmucker C, Schwarzer G, Elm E.Full publication of results initially presented in abstracts. Cochrane Database of Systematic Reviews 2018, Issue 11. Art. No: MR000005. [DOI: 10.1002/14651858.MR000005.pub4] [DOI] [PMC free article] [PubMed] [Google Scholar]
Schulz 1995
- Schulz KF, Chalmers I, Hayes RJ, Altman DG.Empirical evidence of bias. Dimensions of methodological quality associated with estimates of treatment effects in controlled trials. JAMA 1995;273:408-12. [DOI] [PubMed] [Google Scholar]
Sibbald 1998
- Sibbald B, Roland M.Understanding controlled trials: why are randomised controlled trials important? BMJ 1998;316(7126):201. [DOI] [PMC free article] [PubMed] [Google Scholar]
Sommer 2006
- Sommer J, Jansen A, Dräger B, Steinsträter O, Breitenstein C, Deppe M, et al.Transcranial magnetic stimulation - a sandwich coil design for a better sham. Clinical Neurophysiology 2006;117(2):440-6. [DOI] [PubMed] [Google Scholar]
Spitzer 1989
- Spitzer RL, William JBW, Gibbon M, First MB.Structured Clinical Interview for DSM-III-R Personality Disorders (SCID-II). 1989: Biometrics Research Department, New York State Psychiatric Institute, 1989. [Google Scholar]
Storebø 2018
- Storebø OJ, Pedersen N, Ramstad E, Kielsholm ML, Nielsen SS, Krogh HB, et al.Methylphenidate for attention deficit hyperactivity disorder (ADHD) in children and adolescents – assessment of adverse events in non-randomised studies. Cochrane Database of Systematic Reviews 2018, Issue 5. Art. No: CD012069. [DOI: 10.1002/14651858.CD012069.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Swanson 2014
- Swanson JW, Swartz MS.Why the evidence for outpatient commitment is good enough. Psychiatric Services 2014;65(6):808-11. [DOI] [PubMed] [Google Scholar]
Szymanski 1995 (pers comm)
- Szymanski J.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 20th of August 2019.
Tajika 2015
- Tajika A, Ogawa Y, Takeshima N, Hayasaka Y, Furukawa TA.Replication and contradiction of highly cited research papers in psychiatry: 10-year follow-up. British Journal of Psychiatry 2015;207(4):357-62. [DOI] [PubMed] [Google Scholar]
Teri 1997 (pers comm)
- Teri L.Inclusion of study in Cochrane Systematic Review [personal communication]. Email to: A Todorovac 26th of August 2019.
Thomas 2010 [Computer program]
- EPPI-Reviewer 4: software for research synthesis..Thomas J, Brunton J, Graziosi S, Version 4.7.1.0. London, UK: EPPI-Centre Software. Social Science Research Unit, UCL Institute of Education, 2010. Available at www.eppi.ioe.ac.uk.
Thorlund 2009
- Thorlund K, Devereaux PJ, Wetterslev J, Guyatt G, Ioannidis JP, Thabane L, et al.Can trial sequential monitoring boundaries reduce spurious inferences from meta-analyses? International Journal of Epidemiology 2009;38(1):276-86. [DOI] [PubMed] [Google Scholar]
Thorlund 2011
- Thorlund K, Imberger G, Walsh M, Chu R, Gluud C, Wetterslev J, et al.The number of patients and events required to limit the risk of overestimation of intervention effects in meta-analysis - a simulation study. PLOS One 2011;6(10):e25491. [DOI] [PMC free article] [PubMed] [Google Scholar]
Tough 2009
- Tough EA, White AR, Richards SH, Lord B, Campbell JL.Developing and validating a sham acupuncture needle. Acupuncture in Medicine 2009;27(3):118-22. [DOI] [PubMed] [Google Scholar]
Vase 2019
- Vase L.Can insights from placebo and nocebo mechanism studies help improve randomized controlled trials? Clinical Pharmacology and Therapeutics 2019;106(6):1169-71. [DOI] [PubMed] [Google Scholar]
Wager 2015
- Wager TD, Atlas LY.The neuroscience of placebo effects: connecting context, learning and health. Nature Reviews Neuroscience 2015;16(7):403-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
Walach 2011
- Walach H.Placebo controls: historical, methodological and general aspects. Philosophical Transactions: Biological Sciences 2011;366(1572):1870-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wampold 2010
- Wampold BE, Imiel ZE.Great Psychotherapy Debate (Counseling and Psychotherapy) - The Evidence for What Makes Psychotherapy Work. 2 edition. Taylor & Francis Inc, 2010. [Google Scholar]
Wampold 2016
- Wampold BE, Frost ND, Yulish NE.Placebo effects in psychotherapy: a flawed concept and a contorted history. Psychology of Consciousness: Theory, Research, and Practice 2016;3(2):108-20.
Watts 2015
- Watts SE, Turnell A, Kladnitski N, Newby JM, Andrews G.Treatment-as-usual (TAU) is anything but usual: a meta-analysis of CBT versus TAU for anxiety and depression. Journal of Affective Disorders 2015;175:152–67. [DOI] [PubMed] [Google Scholar]
We 2012
- We SR, Koog YH, Park MS, Min B-Il.Placebo effect was influenced by publication year in three-armed acupuncture trials. Complementary Therapies in Medicine 2012;20(1-2):83-92. [DOI] [PubMed] [Google Scholar]
Weimer 2015
- Weimer K, Colloca L, Enck P.Placebo effects in psychiatry: mediators and moderators. Lancet Psychiatry 2015;2(3):246-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
Weissman 1974
- Weissman MM, Klerman GL, Paykel ES, Prusoff B, Hanson B.Treatment effects on the social adjustment of depressed patients. Archives of General Psychiatry 1974;30(6):771-8. [DOI] [PubMed] [Google Scholar]
Weisz 2019
- Weisz JR, Kuppens S, Ng MY, Vaughn-Coaxum RA, Ugueto AM, Eckshtain D, et al.Are psychotherapies for young people growing stronger? Tracking trends over time for youth anxiety, depression, attention-deficit/hyperactivity disorder, and conduct problems. Perspectives on Psychological Science 2019;14(2):216-37. [DOI] [PubMed] [Google Scholar]
Wetterslev 2008
- Wetterslev J, Thorlund K, Brok J, Gluud C.Trial sequential analysis may establish when firm evidence is reached in cumulative meta-analysis. Journal of Clinical Epidemiology 2008;61(1):64-75. [DOI] [PubMed] [Google Scholar]
Wetterslev 2009
- Wetterslev J, Thorlund K, Brok J, Gluud C.Estimating required information size by quantifying diversity in random-effects model meta-analyses. BMC Medical Research Methodology 2009;9(1):86. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wetterslev 2017
- Wetterslev J, Jakobsen JC, Gluud C.Trial Sequential Analysis in systematic reviews with meta-analysis. BMC Medical Research Methodology 2017;17(1):39. [DOI] [PMC free article] [PubMed] [Google Scholar]
WHO 1949
- Manual of the International Classification of Diseases, Sixth Revision (ICD-6). World Health Organization 1949.
WHO 1955
- Manual of the International Classification of Diseases, Seventh Revision (ICD-7). World Health Organization 1955.
WHO 1967
- Manual of the International Classification of Diseases, Eighth Revision (ICD-8). World Health Organization 1967.
WHO 1975
- Manual of the international classification of diseases, Ninth Revision (ICD-9). World Health Organization 1975.
WHO 1993
- Manual of the International Classification of Diseases, Tenth Revision (ICD-10). World Health Organization 1993.
WHO 2018
- Manual of the International Classification of Diseases, Eleventh Revision (ICD-11). World Health Organization 2018. [https://www.who.int/standards/classifications/classification-of-diseases]
Wilson 1978
- Wilson A, Davidson WJ, Blanchard R, WhiteJ.Disulfiram implantation. A placebo-controlled trial with two-year follow-up. Journal of Studies on Alcohol 1978;39(5):809-19. [DOI] [PubMed] [Google Scholar]
Wood 2008
- Wood L, Egger M, Gluud LL, Schulz KF, Jüni P, Altman DG, et al.Empirical evidence of bias in treatment effect estimates in controlled trials with different interventions and outcomes: meta-epidemiological study. BMJ 2008;336(7644):601. [DOI: 10.1136/bmj.39465.451748.AD] [DOI] [PMC free article] [PubMed] [Google Scholar]
Yeung 2017
- Yeung V, Sharpe L, Glozier N, Hackett ML, Colagiuri B.A systematic review and meta-analysis of placebo versus no treatment for insomnia symptoms. Sleep Medicine Reviews 2017;38:17-27. [DOI] [PubMed] [Google Scholar]
Young 2011
- Young T, Hopewell S.Methods for obtaining unpublished data. Cochrane Database of Systematic Reviews 2011, Issue 1110.1002/14651858.MR000027.pub2. Art. No: MR000027. [DOI: 10.1002/14651858.MR000027.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
References to other published versions of this review
Faltinsen 2019
- Faltinsen E, Todorovac A, Hróbjartsson A, Gluud C, Kongerslev MT, Simonsen E, Storebø OJ.Placebo, usual care and wait‐list interventions for all mental health disorders.. Cochrane Database of Systematic Reviews 2019, Issue 1. Art. No: MR000050. [DOI: 10.1002/14651858.MR000050] [DOI] [PMC free article] [PubMed] [Google Scholar]
Klein 2004
- Klein RG, Abikoff H, Hechtman L, Weiss G.Design and rationale of controlled study of long-term methylphenidate and multimodal psychosocial treatment in children with ADHD. Journal of the American Academy of Child and Adolescent Psychiatry 2004;43(7):792-801. [DOI] [PubMed] [Google Scholar]


