Abstract
Background
Post-stroke hand impairment is prevalent and persistent even after a full course of rehabilitation. Hand diminishes stroke survivors’ abilities for activities of daily living and independence. One way to improve treatment efficacy is to augment therapy with peripheral sensory stimulation. Recently, a novel sensory stimulation, TheraBracelet, has been developed in which imperceptible vibration is applied during task practice through a wrist-worn device. The objective of this trial is to determine if combining TheraBracelet with hand task practice is superior to hand task practice alone.
Methods
A double-blind randomized controlled trial will be used. Chronic stroke survivors will undergo a standardized hand task practice therapy program (3 days/week for 6 weeks) while wearing a device on the paretic wrist. The device will deliver TheraBracelet vibration for the treatment group and no vibration for the control group. The primary outcome is hand function measured by the Wolf Motor Function Test. Other outcomes include the Box and Block Test, Action Research Arm Test, upper extremity use in daily living, biomechanical measure of the sensorimotor grip control, and EEG-based neural communication.
Discussion
This research will determine clinical utility of TheraBracelet to guide future translation. The TheraBracelet stimulation is delivered via a wrist-worn device, does not interfere with hand motion, and can be easily integrated into clinical practice. Enhancing hand function should substantially increase stroke survivors' independence and quality of life and reduce caregiver burden.
Trial registration
NCT04569123. Registered on September 29, 2020
Keywords: Stroke, Upper extremity, Physical rehabilitation, Hand function, Subliminal stimulation, Physical stimulation, Stroke rehabilitation, Neurologic rehabilitation, Occupational therapy, Upper limb paresis, Hand, EEG, Paralysis, Randomized controlled trial
Administrative information
Note: the numbers in curly brackets in this protocol refer to SPIRIT checklist item numbers. The order of the items has been modified to group similar items (see http://www.equator-network.org/reporting-guidelines/spirit-2013-statement-defining-standard-protocol-items-for-clinical-trials/).
| Title {1} | Concomitant sensory stimulation during therapy to enhance hand functional recovery post stroke |
| Trial registration {2a and 2b}. | NCT04569123. Clinicaltrials.gov |
| Protocol version {3} | #6. 7/28/2021 |
| Funding {4} | NIH/NICHD 1R01HD094731-01A1 |
| Author details {5a} |
Na Jin Seo, PhD, Department of Rehabilitation Sciences, Department of Health Science and Research, Medical University of South Carolina, Charleston, SC, USA. Ralph H. Johnson VA Medical Center, Charleston, SC, USA. ORCID 0000-0001-6446-5905. seon@musc.edu. 151B Rutledge Ave, MSC 962, Charleston, SC 29425. 843-792-0084. Viswanathan Ramakrishnan, PhD, Department of Public Health Sciences, Medical University of South Carolina, Charleston, SC, USA. ORCID 0000-0002-4098-0539. ramakris@musc.edu. 135 Cannon St, Charleston, SC 29425. 843-876-1153. Michelle L. Woodbury, PhD, OTR/L, Department of Health Science and Research, Medical University of South Carolina, Charleston, SC, USA. 0000-0001-9289-570X. woodbuml@musc.edu. 77 President St, MSC 700, Charleston, SC 29425. 843-792-1671. Leonardo Bonilha, MD, PhD, Department of Neurology, Medical University of South Carolina, Charleston, SC, USA. bonilha@musc.edu. 96 Jonathan Lucas St, MSC 606, Charleston, SC 29425. 843-876-8311. Christian Finetto, PhD, Department of Health Sciences and Research, Medical University of South Carolina, Charleston, SC, USA. ORCID 0000-0003-0520-2034. finetto@musc.edu. 77 President St, MSC 700, Charleston, SC 29425. 843-792-5645. Christian Schranz, PhD, Department of Health Sciences and Research, Medical University of South Carolina, Charleston, SC, USA. ORCID: 0000-0003-1102-7180. schranz@musc.edu. 77 President St, MSC 700, Charleston, SC 29425. Gabrielle Scronce, PT, DPT, PhD, Department of Health Sciences and Research, Medical University of South Carolina, Charleston, SC, USA. ORCID 0000-0002-3861-1371. scronce@musc.edu. 77 President St, MSC 700, Charleston, SC 29425. Kristen Coupland, MS, OTR/L, CSRS, CDRS, Department of Health Sciences and Research, Medical University of South Carolina, Charleston, SC, USA. coupland@musc.edu. 77 President St, MSC 700, Charleston, SC 29425. Jenna Blaschke, BA, Division of Occupational Therapy, Department of Rehabilitation Sciences, Medical University of South Carolina, Charleston, SC, USA. blaschkj@musc.edu. 151B Rutledge Ave, MSC 962, Charleston, SC 29425. 803-459-6403. Adam Baker, BS, Department of Health Sciences and Research, Medical University of South Carolina, Charleston, SC, USA. bakerdon@musc.edu. 77 President St., MSC 700, Charleston, SC 29425 Keith Howard, BS, Department of Health Sciences and Research, Medical University of South Carolina, Charleston, SC, USA. howardke@musc.edu. 77 President St, MSC 700, Charleston, SC 29425. 843-792-2917. Caitlyn Meinzer, PhD, Department of Public Health Sciences, Medical University of South Carolina, Charleston, SC, USA. ellerbcn@musc.edu. 135 Cannon St, Charleston, SC 29425. 843-792-6588. Craig A. Velozo, PhD, Division of Occupational Therapy, Department of Rehabilitation Sciences, Medical University of South Carolina, Charleston, SC, USA. velozo@musc.edu. 151B Rutledge Ave, MSC 962, Charleston, SC 29425. 803-459-6403. Robert J. Adams, MD, Department of Neurology, Medical University of South Carolina, Charleston, SC, USA. adamsrj@musc.edu. 96 Jonathan Lucas St, MSC 606, Charleston, SC 29425. 843-792-3020. |
| Name and contact information for the trial sponsor {5b} |
Medical University of South Carolina, College of Health Professions 843-792-3328, 151B Rutledge Ave, Charleston, SC, 29425 NIH/NICHD, 1-800-370-2943 Email: NICHDInformationResourceCenter@mail.nih.gov Fax: 1-866-760-5947 Mail: P.O. Box 3006, Rockville, MD, 20847 |
| Role of sponsor {5c} | Oversight for regulatory requirements. |
Introduction
Background and rationale {6a}
Stroke is a leading cause of long-term disability in the USA [1]. More than two thirds of nearly 7 million stroke survivors in the USA [1] have persistent hand impairment even after a full course of rehabilitation [2]. Hand impairment diminishes stroke survivors’ abilities for activities of daily living including self-care, hygiene, leisure, and employment, which lowers their independence and increases caregiver burdens [3, 4]. Due to increasing limitations on access to therapy, improving the efficacy of rehabilitation treatment to improve hand function is of significant importance.
Recent meta-analysis shows that upper extremity motor function improves more when therapy is augmented by peripheral sensory stimulation, compared with therapy alone [5]. The scientific rationale is that afferent input is a powerful driver of change in the motor cortex [6, 7]. Specifically, direct projections from the cortical hand sensory areas to motor areas are evidenced in intracortical microstimulation studies in animals [8–11] and in the long-latency cutaneomuscular reflex in humans [12, 13]. These direct projections from the sensory to motor cortex enables afferent sensory stimulation to directly affect motor output [14–16]. For example, corticomotoneuronal excitability as measured using transcranial magnetic stimulation (TMS) has been shown to increase during muscle vibration, compared to no vibration [17, 18]. After 30 min of electrical stimulation of the whole hand via a mesh glove, functional magnetic resonance imaging blood-oxygen-level-dependent signals during finger movement in the primary motor and somatosensory area increased, compared to that before the stimulation [19]. After 2 h of transcutaneous electrical nerve stimulation (TENS), corticomotoneuronal excitability increases via GABAergic mechanism [20]. Daily 1-h TENS over 3 weeks led to increased cortical motor map area and volume of involved muscles (assessed using TMS) [21].
Based on this framework, peripheral sensory stimulation has been used in conjunction with therapy to increase neuroplasticity and motor recovery more than therapy alone in patients with neurologic motor impairment [22–34]. Specifically, 30-min vibration to the upper limb muscles followed by 1-h physiotherapy, repeated for 3 consecutive days, resulted in greater improvement in the Wolf Motor Function Test (WMFT) than dose-matched physiotherapy alone, which sustained 2 weeks after the intervention (n = 30 chronic stroke survivors, randomized to n = 15/group) [24]. This greater functional improvement was associated with greater corticomotor excitability and motor map areas (assessed using TMS) for the stimulation + therapy group compared with the therapy only group [24]. In another study, 2-h TENS followed by 4-h task practice therapy, repeated for 10 days, resulted in greater improvements in the Fugl-Meyer Upper Extremity Assessment and Action Research Arm Test (ARAT) than dose-matched therapy alone, which sustained at 1-month follow-up (n = 36 chronic stroke survivors randomized to n = 18/group) [29]. Meta-analysis supports this premise of using sensory stimulation to augment motor recovery [5].
Unfortunately, most modalities of peripheral sensory stimulation interfere with natural hand tasks. Specifically, suprathreshold stimulation causes sensation irrelevant to tasks at hand, including TENS-induced tingling sensation [19, 21]. Wearing of a glove or a finger cap hampers dexterous finger movement and causes a sense of discomfort [35, 36]. Thus, most modalities of sensory stimulation are administered prior to each therapy session while a person is in a sedentary posture, requiring additional time commitment ranging from 30 min to 2 h a day [22–30]. These constraints make it difficult for implementation and patient adherence to a stimulation regimen [37]. Furthermore, the effect diminishes 20–30 min after the stimulation [20, 38], weakening its effect during therapy. Studies that used sensory stimulation during therapy had no control group (with no stimulation) [31, 32] or did not test stroke survivors [33, 34] and used suprathreshold stimulation causing sensation irrelevant to tasks.
To address this practical limitation of the current sensory stimulation method and fully leverage the therapeutic benefits of sensory stimulation, we have developed an innovative sensory stimulation: “TheraBracelet” is imperceptible random-frequency vibration applied to wrist skin via a wrist-worn device. TheraBracelet does not interfere with natural hand tasks since the stimulation is imperceptible and delivered via a device worn on the wrist [39–41]. The theoretical framework is that imperceptible random-frequency vibration stimulates mechanoreceptors in wrist skin and afferents [42], adds small random currents to neurons in the sensorimotor cortex, triggering coherent [43] firing [44–46] at the peak of inputs related to hand tasks, and consequently enhances neural communication [47, 48] for hand tasks [49–52] and functional recovery. Pilot studies showed preliminary efficacy [53, 54].
A randomized controlled trial to determine clinical utility of TheraBracelet is described herein. This research is expected to result in an efficacious therapeutic adjunct to enhance upper extremity rehabilitation outcomes for people with stroke. Enhancing hand motor function and therapy outcome with TheraBracelet is expected to contribute to improving stroke survivors’ abilities to perform activities of daily living, increasing functional independence, and reducing caregiver burden, thereby leading to increased quality of life for both stroke survivors and caregivers.
Objectives {7}
The objective of this study is to determine if combining TheraBracelet with hand task practice is superior to hand task practice alone in an adequately powered study.
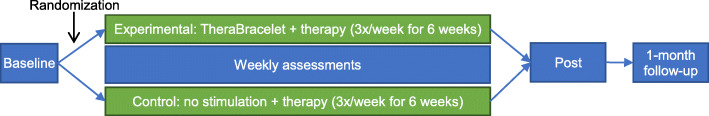
Trial design {8}
The trial design is a double-blind randomized controlled trial involving two parallel groups. Participants will be randomly assigned to either the experimental or control group. Half will be in the experimental group and the other half will be in the control group. Both groups will undergo standardized hand task practice therapy while wearing a device on the paretic wrist. The device will deliver TheraBracelet vibration for the treatment group and no vibration for the control group during therapy. The superiority of the experimental condition over the control condition will be examined.
Methods: participants, interventions, and outcomes
Study setting {9}
The study setting is the research laboratory of the Medical University of South Carolina, Charleston, SC, USA.
Eligibility criteria {10}
Inclusion criteria:
Adult (> = 18 years old)
Survived a stroke at least 6 months ago
WMFT [55] total average time > 10 s
WMFT hand task average time < 120 s
Exclusion criteria:
Concurrent upper limb therapy
Change in spasticity medication or botulinum toxin injection in the upper limb within 3 months prior to or during enrollment
Severe spasticity (Modified Ashworth Scale [56] = 4–5 out of 5) that prohibits engagement in task practice
Comorbidity such as complete upper extremity deafferentation, orthopedic conditions limiting motion [57], premorbid neurologic conditions, or compromised skin integrity of the wrist due to burn or long-term use of blood thinners
Language barrier or cognitive impairment that precludes following instructions and/or providing consent
Who will take informed consent? {26a}
Research staff approved by the Institutional Review Board (IRB) will take informed consent. The consent process will take place in a private room when the potential participant comes to the laboratory on a scheduled time agreed upon between the study personnel and the participant. The content of the consent will be verbally explained to the participant and the participant will be asked to raise any questions and concerns.
Additional consent provisions for collection and use of participant data and biological specimens {26b}
The consent includes sharing of de-identified data with the public and other investigators in publications and ClinicalTrials.gov. In addition, authorization for use and release of individually identifiable health information collected for research with the Medical University of South Carolina IRB and the funding agency will be obtained in writing. This trial does not involve collecting biological specimens for storage.
Interventions
Explanation for the choice of comparators {6b}
The control group will receive standardized task practice therapy while receiving no vibration from the device on the paretic wrist. The control condition represents the standard task practice therapy [58] without the TheraBracelet stimulation. For both groups, the device is worn during therapy only (not outside therapy).
Intervention description {11a}
Intervention schedule: All participants will undergo 6 weeks of standardized task practice therapy. The therapy schedule will be 3 sessions/week for 6 weeks, with each session lasting 1–2 h to complete 300 movement repetitions. This therapy schedule is chosen to simulate the outpatient rehabilitation model [59] and facilitate potential translation of the protocol for implementation in rehabilitation practice. This therapy schedule is also similar with other stroke upper limb rehabilitation programs with distributed practice schedules, enabling comparisons [60–64].
Task practice therapy: Therapists will be trained to administer the task practice therapy according to standardized procedures. The treatment manual, containing a menu of task practice activities, was developed by two experienced occupational therapists, based on the EXCITE trial [65] manual and Dr. Lang’s Task Specific Practice text [58]. The main principles of the therapy program are detailed below.
Specificity: All therapy tasks address hand/finger motions, because TheraBracelet has been shown to impact hand/finger control in our preliminary studies [51, 53]. Selected tasks naturally require several repetitions of the specified motion.
Structure: For organizational purposes, the tasks are categorized into self-care, household care, leisure, and vocation. Each category contains two types of tasks: tasks requiring (i) primarily in-hand manipulation and (ii) reaching. The in-hand manipulation tasks will be performed within the subject’s reachable workspace (e.g., in front of the torso) to exclusively focus on hand dexterity which is the intervention target using TheraBracelet. The reaching tasks will be performed by reaching to grasp/place objects throughout the reachable workspace to integrate hand dexterity skills with the proximal upper limb motion to maximize the functional relevance of the treatment. A total of 4 tasks (2 in-hand and 2 reaching, from each of 2 categories of choice) will be practiced in each session. Repetitions for in-hand vs. reaching will be balanced.
Saliency: To assure the task practice is meaningful, thus motivating for the subject, the therapist and subject will work together to select the tasks from the task menu. This maximizes the potential for the subject to apply skills learned in clinic to their real world tasks and goals [58].
Repetitions: To standardize therapy dosage, we will aim for each participant to achieve 300 movement repetitions per session. This dosage is feasible in a 1–2 h timeframe [61, 66] with no excessive pain or fatigue [60], results in functional improvements [61], and corresponds to the lower end of the repetitions in animal and motor learning studies that have been shown to promote neural plasticity and behavioral changes [67, 68]. One repetition will be defined for each activity in the task manual to ensure consistency in counting repetitions. For in-hand tasks, one repetition will be defined as one hand manipulation (e.g., place three coins in the paretic palm, and transfer to fingertips and insert into a slot with one coin at a time for 3 repetitions). For reaching tasks, one repetition will be reach-grasp-manipulate-release [61] (e.g., reach for/grasp a toothbrush, apply toothpaste, simulate brushing motions four times, and replace the toothbrush on the table, Fig. 1).
Difficulty: Brain reorganization occurs through motor learning that requires problem solving, not simple repetition of mastered skills, as shown in human [69, 70] and animal studies [67, 71, 72]. Thus, tasks will be at a “just-right” difficulty level that does not overwhelm or underwhelm participants. Specifically, we will implement tasks such that the participant practices movements that s/he can partially perform, as opposed to easily perform or cannot perform. We will increase or decrease difficulty based on the participant’s success rate of the task (≥ 90% or ≤ 50%), task pace, and patient input [58] following the Challenge Point Framework [73]. Grading of tasks will be accomplished by changing the object weight, size [74–76], shape [77, 78], slipperiness [79–84], compliance, stability [85], and location, use of adaptive materials (e.g., nonslip mat to prevent items from moving), task complexity, and speed and accuracy of movement.
Fig. 1.

An example therapy session with the wrist-worn device that can deliver TheraBracelet stimulation
TheraBracelet stimulation: TheraBracelet is imperceptible random-frequency vibration applied to wrist skin via a wrist-worn device. TheraBracelet stimulation parameters were determined based on literature and our previous data as detailed below.
Frequency: The vibrator will be driven by random-frequency signal low-pass filtered at 500 Hz for the following reasons. Literature in stochastic resonance collectively demonstrates that broadband random-frequency (white noise) stimulation enhances neural communication and signal detection (see reviews [43, 44, 86] and applications [47, 49, 87–92]). The benefit of the random-frequency stimulation compared to constant frequency stimulation is that it is less subject to desensitization (or sensory habituation). Literature comparing the two types of stimulation shows that temporally non-uniform stimulation improves tactile sensation and modulates the central nervous system excitability, while such effects could not be obtained with constant frequency stimulation [93, 94]. By preventing desensitization, TheraBracelet is expected to provide a benefit that persists throughout therapy sessions. As for the bandwidth, existing evidence shows that one’s ability to detect 25 Hz signal was enhanced when accompanied by random-frequency vibration bandwidth filtered at 0.125–50 Hz, and similarly, ability to detect 250 and 400 Hz signal was enhanced with random-frequency vibration of 50–500 Hz [87]. For various sensorimotor tasks including hand grip, touch detection, and texture discrimination, random-frequency vibration low-pass filtered at 300 Hz (i.e., 0–300 Hz) was used to successfully enhance sensorimotor performance [89]. Since human mechanoreceptors can detect vibration from 0.4 to 500 Hz [95], we decided to use random-frequency vibration low-pass filtered at 500 Hz to span the entire frequency spectrum for human mechanoreceptors.
Location: The vibration will be applied to the dorsal wrist, because the same beneficial effects on improved detection of fingertip touch during vibration vs. no vibration were obtained for all vibration locations we tested—volar wrist, dorsal wrist, dorsum of the hand near the first and the second knuckle, thenar area, and hypothenar area, in chronic stroke survivors [50] as well as healthy adults [52]. The wrist is optimal for a wearable device, as the device can be worn on the wrist similar to wearing a wristwatch.
Intensity: Vibration intensity 60% of the sensory threshold (imperceptible) will be used, because that intensity enhanced finger touch detection the most (as assessed by the monofilament test [96]), while 40% and 80% of the sensory threshold had lesser effects [50, 97]. Further, vibration 120% of the sensory threshold (perceptible) degraded finger sensation [52]. This intensity-effect relationship is consistent with the bell-shaped curve (i.e., inverted U-shape function) reported in stochastic resonance literature in which vibration intensities at 33–67% of the sensory threshold improved sensory perception to a greater extent than did intensities of 0%, 83%, and 100% of the sensory threshold [87], and suprathreshold vibration degraded sensation [88]. These results suggest that vibration intensity should be high enough to facilitate signal transmission but not too high to mask other signals [87].
For both groups, the participant’s sensory threshold will be determined at the beginning of each therapy session using a custom-developed software program that utilizes the staircase method [98]. Specifically, the vibration amplitude will be increased or decreased until the participant verbally indicates that s/he could or could not perceive the wrist vibration, respectively. The threshold will be determined as the average of 6 amplitudes that the participant could barely feel.
For the treatment group, TheraBracelet stimulation will be turned on when movement is detected at or higher than the acceleration magnitude that corresponds to movement of one finger only. Upon movement detection, TheraBracelet stimulation will be turned on for 1 s, after which the stimulation will continue if movement is detected again, or stop if no movement is detected, such that stimulation will be on during all movements but not for more than 1 s after the movement ends to minimize potential habituation to the stimulation. The consistency in the frequency characteristics from the device will be verified weekly using a laser vibrometer (OMS Corp, Laguna Hills, CA, USA).
Transfer package: To achieve greater independence at home using the participant’s improved motor capacities from therapy, we will implement a transfer package [99]. Transfer package is adopted from the behavioral intervention field specifically for physical neurorehabilitation, based on the principles of self-monitoring [100, 101], contracting/negotiating for specific behaviors [102], phone contacts to increase compliance [103–105], and problem-solving to overcome barriers [106–110]. Components of the transfer package are detailed below.
Behavioral contracts: The researcher will negotiate a contract with the participant and separately with the caregiver, if any, in which they agree to comply with the intervention including completion of all assignments and using the paretic hand on specific activities of daily living as much as possible outside the lab. The negotiated document will be signed by the participant, caregiver, and researcher to emphasize the character of the document as a contract.
Activity tracking: The Motor Activity Log (MAL) [111] will be administered at baseline and reviewed weekly (in person during 6-week intervention and in weekly phone contacts following intervention) to track amount and quality of paretic hand use in daily living and to keep participants’ attention on use of the paretic hand at home.
Problem solving: The therapist will help participants think through barriers to using the paretic hand at home and ways to overcome them (e.g., use modified tools, ensure safe environment) at each visit during the 6-week intervention and in weekly phone contacts following intervention.
Daily home diary: Specific activities that the participant can try using the paretic hand at home will be determined and written down. The participant will record how many times s/he has used the paretic hand for the specified activity.
Home skill assignment: Participants will be assigned to perform 4 specific tasks with the paretic hand daily with specified repetitions and record on a written check-off sheet during 6-week intervention. The tasks will be chosen to improve the participant’s most significant movement deficits. Following intervention, participants will be prescribed a written individualized home skill practice program entailing specific tasks to practice with specified repetitions for each day of the week for 4 weeks. When noncompliance is indicated for the daily home diary and home skill assignment, the therapist will inquire into the reasons and problem-solve with the participant/caregiver on how to reverse the trend.
Criteria for discontinuing or modifying allocated interventions {11b}
Participants with repeated no shows will be withdrawn.
Strategies to improve adherence to interventions {11c}
The intervention requires in-person visit to the laboratory. Support for parking and transportation assistance will be provided as necessary. The visit schedule will be printed and handed out to each participant. Reminder phone calls will be made. A waiting room will be provided within the building for caregivers. Remuneration for participation will be provided. All COVID-19 precautions will be taken to ensure health and safety of participants and study personnel.
Relevant concomitant care permitted or prohibited during the trial {11d}
Concurrent upper limb rehabilitation is prohibited during participation in the study. Change in spasticity medication or botulinum toxin injection in the upper limb is prohibited during the study. Concomitant care for other issues is permitted.
Provisions for post-trial care {30}
Participants will be followed up until 1 month post intervention for adverse events. Necessary medical treatment will be participants’ responsibilities.
Outcomes {12}
The primary outcome measure is hand function measured by the WMFT time [55]. The primary time point is from the baseline to 1-month follow-up. Hand function will also be assessed using the Box and Block Test (BBT) [112] and ARAT [113]. Paretic upper limb use in daily living will be measured using accelerometers [114] and MAL [111]. The patient-centered outcome measures of the Stroke Impact Scale, perceived meaningfulness of the intervention [115], and usability feedback on TheraBracelet will also be obtained. Other outcome measures will include sensorimotor grip control and neural communication for hand grip, quantified as the digit force directional control [116, 117] and EEG connectivity in the cortical sensorimotor network [118].
Participant timeline {13}
The participant timeline is shown in Fig. 2. A baseline assessment will take place prior to the intervention. The intervention will entail a total of 18 task practice sessions with or without TheraBracelet stimulation over 6 weeks. Hand function assessments will be performed weekly during the 6-week intervention to examine the pattern of progress. The post assessment will take place within a week from the last intervention session. The 1-month follow-up is to assess retention.
Fig. 2.
Participation timeline. All participants will have a baseline assessment, 6 weeks of task practice therapy with weekly hand function assessments, post assessment, and 1-month follow-up assessment
Sample size {14}
The primary objective is to determine clinical potential of TheraBracelet as measured by WMFT time. Insight on its minimal clinically important difference (MCID) is available in literature. Since the minimum detectable change ranges from 1.0 to 3.4 s [119], it could be interpreted that greater than 3.4 s change represents true clinical change. Anchor-based and distribution-based minimum detectable change and MCID range from 1.5 to 4.36 s [120]. Lang et al. (2008) report change of 19 ± 16 s was perceived as meaningful by acute stroke survivors, while 4 ± 7 s was not perceived as meaningful for the dominant hand [115]. Variability was high in that study, where patients’ perception of meaningfulness ranged between 3 and 11 s. In a recent ICARE trial [62], improvement in WMFT time of 8 ± 3 s after inpatient rehabilitation for acute stroke survivors (1.5 month post stroke) was associated with improvement in Stroke Impact Scale hand portion of 37 ± 5 (in percent points) which substantially exceeded MCID of 25 points (representing easement for tasks by a full category, e.g., from “very difficult” to “somewhat difficult”) [121]. Based on these, 4 to 7 s between-group difference in improvement in WMFT time will be considered minimally clinically meaningful.
This study will be powered to detect sustained difference at follow-up of at least 4 s in WMFT between the treatment and control groups. For the repeated measures design with a significance level of 0.017 (adjusted for simultaneously comparing 3 outcomes), at least 80% power, and 4 time points, for a standard deviation (SD) of 5.1 s and compound symmetry correlation of 0.92 (both based on our pilot study [53]), sample size of 32 per group will be adequate. We believe this is a very conservative estimate since we used data from a 2-week pilot intervention [53] to compute power, and this 6-week intervention is expected to yield greater effects [54]. Adjusting for 15% attrition, 38 per group is planned (total n = 76).
This sample size is sufficient for other outcome measures at follow-up by allowing detection of the minimum between-group difference in (i) BBT of 5.3 (with SD = 4.9 from our pilot study [53]) which is approximately the minimum detectable change of 5.5 [122], (ii) digit force directional control of 4.9° (SD = 4.54) which is approximately the group difference at follow-up observed in our pilot study and distinguishes grip control impairment levels [116], and (iii) EEG connectivity of 0.017 (SD = 0.016) which associates with psychophysical behavior change [123].
Recruitment {15}
Participants will be recruited from the MUSC Registry for Stroke Recovery (RESTORE). Currently, the RESTORE has information of more than a thousand stroke survivors who have agreed to be contacted for research (approved MUSC IRB PRO# 37803). The registry is growing every day, as approximately 500 new stroke cases are treated at the MUSC Stroke Center every year. All eligible stroke patients in the inpatient stroke units as well as the outpatient clinic are contacted by a dedicated recruiter supported by the NIH-funded Center of Biomedical Excellence for Stroke Recovery at MUSC to be enrolled in the registry. In addition to recruiting stroke survivors from the hospital, we recruit stroke survivors from the community by having a dedicated outreach therapist from the center visit local stroke support group meetings and develop relationships with stroke survivors, caregivers, and clinicians, and also by organizing community outreach events such as stroke caregiver summits and stroke recovery community engagement, to establish grassroots connections with stroke survivors and caregivers in the community. The center also sends newsletters to survivors, caregivers, local clinics, and local clinicians to inform them of news, new events, and new projects. With this effort, the registry has been growing with > 10 new enrollees per month. In addition, trial information is available via internet (e.g., ClinicalTrials.gov, South Carolina Research Studies Directory).
Assignment of interventions: allocation
Sequence generation {16a}
A computer-generated random allocation sequence will be used. Block randomization will be used to ensure balance (half in the experimental, half in the control). Block sizes will be randomly chosen between 8 and 38.
Concealment mechanism {16b}
At the beginning of each intervention session, the custom-developed software program is used to determine the sensory threshold. The program requires a participant number. Upon completion of the sensory threshold determination, the program will access the computer-generated random allocation sequence, find the assignment information for the participant number, and apply that group assignment information to the device’s vibration output. Therefore, the research personnel will not be involved in assigning groups and thus be blinded to the group assignment. Participants will be blinded, because TheraBracelet stimulation is subthreshold and both groups will not feel any vibration.
Implementation {16c}
The allocation sequence will be generated by the computer. The approved study staff will enroll participants. The custom-developed program will assign participants to the group according to the computer-generated allocation sequence.
Assignment of interventions: blinding
Who will be blinded {17a}
Participants, care providers, intervention providers, outcome assessors, and data analysts except for the primary biostatistician will be blinded to the group assignment.
Procedure for unblinding if needed {17b}
A permission for unblinding will be deliberated and reviewed by the Data and Safety Monitoring Board (DSMB) if unanticipated intervention-related serious adverse events warrant investigation using the group assignment information.
Data collection and management
Plans for assessment and collection of outcomes {18a}
The outcome assessment timeline is provided in Fig. 2. All clinical hand function tests (WMFT, BBT, and ARAT) will be administered by a blinded research therapist, videotaped, coded in names, and scored by raters who are blinded to the group assignment as well as timing of the videos (i.e., before or how many weeks after the intervention). Raters will be trained until excellent intra/interrater reliability is met with correlation greater than 0.9.
WMFT time has been validated for test-retest and interrater reliability in chronic stroke survivors [124, 125], and its minimal detectable change has been established [119]. WMFT time showed responsiveness to TheraBracelet treatment in our pilot study [54]. BBT and ARAT have also been validated for test-retest and interrater reliability [122, 126–128] and shown to be responsive to changes [112, 129] in stroke survivors. Minimal detectable change for BBT [122] and minimal clinically important difference for ARAT have been reported [115].
To gauge what participants perceive they can do functionally that they could not do before, we will obtain the Stroke Impact Scale hand and activities of daily living subscales [121, 130]. MAL [111] is used to assess the perceived quantity and quality of the paretic hand use in activities of daily living as part of the transfer package. The perceived meaningfulness of the intervention will be obtained on a 7-point Likert scale [115] (1 = much better, 2 = a little better, meaningful, 3 = a little better, not meaningful, 4 = about the same, 5 = a little worse, not meaningful, 6 = a little worse, meaningful, 7 = much worse) after completion of the intervention. Usability feedback [131] will also be obtained for TheraBracelet.
Accelerometers have been used to objectively capture changes in stroke survivors’ upper limb usage in daily living before and after therapy [132]. Although accelerometer data can be influenced by the whole body motion (e.g., walking) or non-purposeful upper limb movement, accelerometer data have been shown to significantly correlate with the number of observed purposeful repetitions [133–135] and adequately represent changes in the amount of functional upper limb use [114]. Tri-axial accelerometers (GT9X Link, ActiGraph, Pensacola, FL, USA) will be worn on each wrist for 3 days to capture upper limb activity of an average day [114]. Accelerometers will not interfere with TheraBracelet, as accelerometers are worn outside the laboratory and TheraBracelet is worn in the laboratory during therapy only. From 3-dimensional limb acceleration data recorded in 1 s bins over days [132], we will compute the duration and intensity of the paretic upper limb movement relative to the nonparetic limb (“use ratio” and “magnitude ratio,” respectively) [132, 135] as objective outcome measures.
Sensorimotor grip control will be quantified using the well-established biomechanical sensorimotor integration measures of digit force directional control [116, 117, 136–138] and efficient scaling of grip force [139–144]. Neural communication for hand grip will be quantified as EEG connectivity [118, 145] in the cortical sensorimotor network during the paretic hand grip. Accelerometer and sensorimotor grip control will be analyzed using a custom-scripted code in MATLAB (The MathWorks, Natick, MA, USA), and neural communication will be analyzed using Brainstorm [146] to obtain the final metrics by the blinded researcher post data collection.
Plans to promote participant retention and complete follow-up {18b}
To promote participant retention and complete follow-up, effective communication will be maintained between study staff and participants. Schedules, changes to schedules, and expectation for each visit will be clearly communicated.
Data management {19}
All electronic data will be stored in a password-protected secure research server. Visit records in paper will be scanned and stored in the password-protected secure research server. Data will be entered into a computer-based database. Quarterly data quality assessments will be performed by examining the outcomes databases for missing data, unexpected distributions or responses, irregularities, and outliers. Accuracy and completeness of the data collected will also be ensured.
Confidentiality {27}
The consent and HIPAA forms where personally identifiable information is recorded will be stored in a locked cabinet in a locked office. Only study personnel will have access to this personally identifiable information. For the video recording of the upper limb function tests, we will set the camera angle such that the video recording does not capture the participant’s face, while capturing the hand and arm movements and the interaction between the hand and objects in hand manipulation. All data will be coded with a participant code, and no personally identifiable information will be used to label the data. This means individual results would not be able to be linked to the participant by others who review the results of this research. De-identified paper data including testing sheets documenting testing sequences and notes will be stored in a cabinet in a key-locked room that is accessible to study personnel only. The linkage between the participant identities and participant codes will be stored in a locked cabinet in a locked room and will be accessible to study personnel only.
Plans for collection, laboratory evaluation, and storage of biological specimens for genetic or molecular analysis in this trial/future use {33}
This trial does not involve collecting biological specimens for storage.
Statistical methods
Statistical methods for primary and secondary outcomes {20a}
The primary analysis will be a repeated measures general linear model with compound symmetry covariance structure (although other structures will be compared) for each outcome. The dependent variables will be the change from baseline at the evaluation times. The primary independent variables are group (treatment vs. control), evaluation time, and their interaction. We will also include sex as a biological independent variable along with its interactions to study sex difference. If the group×time interaction is significant, the main hypothesis that there is a group difference at follow-up will be tested using post hoc tests. Diagnostics will be performed on the residuals and appropriate actions will be taken if assumptions are not met. Summary statistics and graphs with a scatter plot matrix of the outcomes across time will be reviewed. Analyses will be performed using SAS© v.9.4. Greater increase in hand function for the treatment than control (with a significant group×time interaction) will support the hypothesis.
In secondary analysis, we will examine the time course of effects by analyzing the group differences in outcomes longitudinally (baseline at week 0, intervention week 1–6, post, and follow-up at 10 weeks) and finding the best fit regression model. This analysis will also inform the minimum duration to observe a significant effect. We will also test if the treatment group perceives the intervention more meaningful than the control group. An ordinal logistic regression assuming proportional odds will be considered.
Interim analyses {21b}
No interim analysis is planned. The DSMB may recommend stopping the study if the study has unanticipated safety concerns that warrant stopping.
Methods for additional analyses (e.g., subgroup analyses) {20b}
There will be no other additional analysis other than the analyses mentioned above.
Methods in analysis to handle protocol non-adherence and any statistical methods to handle missing data {20c}
We will use intent-to-treat analysis. If missing data arise, multiple imputation methods will be applied under the assumption of missing at random.
Plans to give access to the full protocol, participant level-data and statistical code {31c}
The protocol will be shared in ClinicalTrials.gov. De-identified participant-level dataset and/or statistical code will be shared upon reasonable request in writing.
Oversight and monitoring
Composition of the coordinating center and trial steering committee {5d}
Study oversight will be provided by the DSMB. The DSMB will be composed of a board-certified stroke neurologist, a registered and licensed occupational therapist, and a biostatistician with expertise in design and analysis of clinical trials. The DSMB members will be experienced in care of stroke survivors and/or stroke recovery research. The DSMB will convene semiannually to review enrollment and study progression.
The trial management will be performed by the principal investigator, co-investigators, and the IRB-approved study personnel. The trial team will meet weekly or as necessary to discuss the trial setup, operation, progression, data analysis, interpretation, and dissemination. The principal investigator and study personnel are responsible for day-to-day operation and organization of the trial including identifying potential recruits and taking consent.
Composition of the data monitoring committee, its role and reporting structure {21a}
The DSMB will also ensure the safety of participants and the validity and integrity of data collected during the study. The DSMB will review adverse event data and provide a report to the IRB. The DSMB will be independent from the sponsor and competing interests.
Adverse event reporting and harms {22}
Adverse events will be solicited at each visit, recorded, and coded in terms of frequency, severity, relatedness to the intervention, and unanticipated nature using established guidelines [147–149]. All serious adverse events will be investigated by an independent medical monitor to determine relatedness to the intervention. The report by the independent medical monitor will be reviewed by the DSMB. All related serious adverse events will be reported to the IRB as they occur. All adverse event data will be tabulated and reported to the DSMB and ClinicalTrials.gov.
Frequency and plans for auditing trial conduct {23}
The sponsor will audit the study at random. The sponsor will review the study progression, regulatory compliance, and training compliance of all study personnel.
Plans for communicating important protocol amendments to relevant parties (e.g., trial participants, ethical committees) {25}
Any changes will be approved by the IRB prior to being in effect. Changes will be updated in ClinicalTrials.gov.
Dissemination plans {31a}
Trial results will be disseminated in ClinicalTrials.gov, in publications, and in conferences, and in-service/community presentations.
Discussion
In case the sensory threshold changes over time, we will determine the participant’s sensory threshold at each intervention session for both groups. Participants might request wearing the device more than during therapy (e.g., at home) but will not be allowed based on the study need to control wear time. This research will determine clinical utility of TheraBracelet. If TheraBracelet is found efficacious, it is expected that TheraBracelet can be easily integrated in clinical practice. Enhancing hand function should increase stroke survivors’ independence and quality of life.
Trial status
Protocol #6. July 28, 2021. Recruitment began November 2, 2020, and is expected to conclude in November 2025.
Acknowledgements
We are grateful for the contribution from Dr. Ashley Wabnitz as an independent medical monitor. We are grateful for the contribution from the Zucker Institute of Applied Neurosciences for the device development. We are grateful for the intellectual input and infrastructure provided by the NIH/NIGMS P20GM109040 Center of Biomedical Research Excellence in Stroke Recovery at the Medical University of South Carolina directed by Dr. Steven Kautz.
Authors’ contributions {31b}
NS is the principal investigator and conceived the study. NS, VR, MW, and LB contributed to study design and development of the proposal. VR is a trial biostatistician and designed the sample size and statistical analysis plan. GS and JB administer the intervention. KC administers clinical assessments. CF developed and KH administers motion capture assessments. CS and AB administer EEG assessment. CM, CV, and RA serve in the Data and Safety Monitoring Board. All authors reviewed and approved the final manuscript.
Funding {4}
This work was supported by NIH/NICHD 1R01HD094731-01A1. The study funder is not involved nor has responsibility in the collection, analysis, and interpretation of data and writing a manuscript.
Availability of data and materials {29}
Reasonable requests for access to the final trial dataset should be submitted to the principal investigator.
Abbreviations
- ARAT
Action Research Arm Test
- AR(1)
Autoregressive
- BBT
Box and Block Test
- DSMB
Data and Safety Monitoring Board
- EEG
Electroencephalogram
- GABA
Gamma aminobytyric acid
- IRB
Institutional Review Board
- MAL
Motor Activity Log
- MCID
Minimal clinically important difference
- MUSC
Medical University of South Carolina
- NIH
National Institutes of Health
- RESTORE
MUSC Registry for Stroke Recovery
- SC
South Carolina
- SD
Standard deviation
- TENS
Transcutaneous electrical nerve stimulation
- TMS
Transcranial magnetic stimulation
- USA
United States of America
- WMFT
Wolf Motor Function Test
Declarations
Ethics approval and consent to participate {24}
The protocol has been approved by the Medical University of South Carolina IRB Pro00090790. Written informed consent to participate will be obtained from all participants.
Consent for publication {32}
Not applicable.
Competing interests {28}
NJS is an inventor of the TheraBracelet stimulation. All other authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Na Jin Seo, Email: seon@musc.edu.
Viswanathan Ramakrishnan, Email: ramakris@musc.edu.
Michelle L. Woodbury, Email: woodbuml@musc.edum
Leonardo Bonilha, Email: bonilha@musc.edu.
Christian Finetto, Email: finetto@musc.edu.
Christian Schranz, Email: schranz@musc.edu.
Gabrielle Scronce, Email: scronce@musc.edu.
Kristen Coupland, Email: coupland@musc.edu.
Jenna Blaschke, Email: blaschkj@musc.edu.
Adam Baker, Email: bakerdon@musc.edu.
Keith Howard, Email: howardke@musc.edu.
Caitlyn Meinzer, Email: ellerbcn@musc.edu.
Craig A. Velozo, Email: velozo@musc.edu
Robert J. Adams, Email: adamsrj@musc.edu
References
- 1.Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, de Ferranti S, Després JP, Fullerton HJ, Howard VJ, Huffman MD, Judd SE, Kissela BM, Lackland DT, Lichtman JH, Lisabeth LD, Liu S, Mackey RH, Matchar DB, McGuire D, Mohler ER, 3rd, Moy CS, Muntner P, Mussolino ME, Nasir K, Neumar RW, Nichol G, Palaniappan L, Pandey DK, Reeves MJ, Rodriguez CJ, Sorlie PD, Stein J, Towfighi A, Turan TN, Virani SS, Willey JZ, Woo D, Yeh RW, Turner MB, American Heart Association Statistics Committee and Stroke Statistics Subcommittee Heart Disease and Stroke Statistics-2015 Update: a report from the American Heart Association. Circulation. 2015;131(4):e29–e322. doi: 10.1161/CIR.0000000000000152. [DOI] [PubMed] [Google Scholar]
- 2.Lawrence ES, Coshall C, Dundas R, Stewart J, Rudd AG, Howard R, Wolfe CDA. Estimates of the prevalence of acute stroke impairments and disability in a multiethnic population. Stroke. 2001;32(6):1279–1284. doi: 10.1161/01.STR.32.6.1279. [DOI] [PubMed] [Google Scholar]
- 3.Nakayama H, Jorgensen HS, Raaschou HO, Olsen TS. Recovery of upper extremity function in stroke patients: the Copenhagen Stroke Study. Arch Phys Med Rehabil. 1994;75(4):394–398. doi: 10.1016/0003-9993(94)90161-9. [DOI] [PubMed] [Google Scholar]
- 4.Stewart JC, Cramer SC. Patient-reported measures provide unique insights into motor function after stroke. Stroke. 2013;44(4):1111–1116. doi: 10.1161/STROKEAHA.111.674671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Conforto AB, Dos Anjos SM, Bernardo WM, Silva AAD, Conti J, Machado AG, et al. Repetitive peripheral sensory stimulation and upper limb performance in stroke: a systematic review and meta-analysis. Neurorehabil Neural Repair. 2018;32(10):863–871. doi: 10.1177/1545968318798943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Baker SN. Oscillatory interactions between sensorimotor cortex and the periphery. Curr Opin Neurobiol. 2007;17(6):649–655. doi: 10.1016/j.conb.2008.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schabrun SM, Ridding MC, Galea MP, Hodges PW, Chipchase LS. Primary sensory and motor cortex excitability are co-modulated in response to peripheral electrical nerve stimulation. PLoS One. 2012;7(12):e51298. doi: 10.1371/journal.pone.0051298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kaneko T, Caria MA, Asanuma H. Information processing within the motor cortex. II. Intracortical connections between neurons receiving somatosensory cortical input and motor output neurons of the cortex. J Comp Neurol. 1994;345(2):172–184. doi: 10.1002/cne.903450203. [DOI] [PubMed] [Google Scholar]
- 9.Kaneko T, Caria MA, Asanuma H. Information processing within the motor cortex. I. Responses of morphologically identified motor cortical cells to stimulation of the somatosensory cortex. J Comp Neurol. 1994;345(2):161–171. doi: 10.1002/cne.903450202. [DOI] [PubMed] [Google Scholar]
- 10.Lemon RN. Functional properties of monkey motor cortex neurones receiving afferent input from the hand and fingers. J Physiol. 1981;311(1):497–519. doi: 10.1113/jphysiol.1981.sp013601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Matyas F, Sreenivasan V, Marbach F, Wacongne C, Barsy B, Mateo C, Aronoff R, Petersen CCH. Motor control by sensory cortex. Science. 2010;330(6008):1240–1243. doi: 10.1126/science.1195797. [DOI] [PubMed] [Google Scholar]
- 12.Jenner JR, Stephens JA. Cutaneous reflex responses and their central nervous pathways studied in man. J Physiol. 1982;333(1):405–419. doi: 10.1113/jphysiol.1982.sp014461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen R, Ashby P. Reflex responses in upper limb muscles to cutaneous stimuli. Can J Neurol Sci. 1993;20(4):271–278. doi: 10.1017/S0317167100048174. [DOI] [PubMed] [Google Scholar]
- 14.Ridding MC, Rothwell JC. Afferent input and cortical organisation: a study with magnetic stimulation. Exp Brain Res. 1999;126(4):536–544. doi: 10.1007/s002210050762. [DOI] [PubMed] [Google Scholar]
- 15.Shim JK, Karol S, Kim YS, Seo NJ, Kim YH, Kim Y, Yoon BC. Tactile feedback plays a critical role in maximum finger force production. J Biomech. 2012;45(3):415–420. doi: 10.1016/j.jbiomech.2011.12.001. [DOI] [PubMed] [Google Scholar]
- 16.Seo NJ, Shim JK, Engel AK, Enders LR. Grip surface affects maximum pinch force. Hum Factors. 2011;53(6):740–748. doi: 10.1177/0018720811420256. [DOI] [PubMed] [Google Scholar]
- 17.Rosenkranz K, Rothwell JC. Differential effect of muscle vibration on intracortical inhibitory circuits in humans. J Physiol. 2003;551(Pt 2):649–660. doi: 10.1113/jphysiol.2003.043752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rosenkranz K, Pesenti A, Paulus W, Tergau F. Focal reduction of intracortical inhibition in the motor cortex by selective proprioceptive stimulation. Exp Brain Res. 2003;149(1):9–16. doi: 10.1007/s00221-002-1330-3. [DOI] [PubMed] [Google Scholar]
- 19.Golaszewski SM, Siedentopf CM, Koppelstaetter F, Rhomberg P, Guendisch GM, Schlager A, Gallasch E, Eisner W, Felber SR, Mottaghy FM. Modulatory effects on human sensorimotor cortex by whole-hand afferent electrical stimulation. Neurology. 2004;62(12):2262–2269. doi: 10.1212/WNL.62.12.2262. [DOI] [PubMed] [Google Scholar]
- 20.Kaelin-Lang A, Luft AR, Sawaki L, Burstein AH, Sohn YH, Cohen LG. Modulation of human corticomotor excitability by somatosensory input. J Physiol. 2002;540(Pt 2):623–633. doi: 10.1113/jphysiol.2001.012801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Meesen RL, Cuypers K, Rothwell JC, Swinnen SP, Levin O. The effect of long-term TENS on persistent neuroplastic changes in the human cerebral cortex. Hum Brain Mapp. 2011;32(6):872–882. doi: 10.1002/hbm.21075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Conforto AB, Ferreiro KN, Tomasi C, dos Santos RL, Moreira VL, Marie SK, et al. Effects of somatosensory stimulation on motor function after subacute stroke. Neurorehabil Neural Repair. 2010;24(3):263–272. doi: 10.1177/1545968309349946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Celnik P, Hummel F, Harris-Love M, Wolk R, Cohen LG. Somatosensory stimulation enhances the effects of training functional hand tasks in patients with chronic stroke. Arch Phys Med Rehabil. 2007;88(11):1369–1376. doi: 10.1016/j.apmr.2007.08.001. [DOI] [PubMed] [Google Scholar]
- 24.Marconi B, Filippi GM, Koch G, Giacobbe V, Pecchioli C, Versace V, Camerota F, Saraceni VM, Caltagirone C. Long-term effects on cortical excitability and motor recovery induced by repeated muscle vibration in chronic stroke patients. Neurorehabil Neural Repair. 2011;25(1):48–60. doi: 10.1177/1545968310376757. [DOI] [PubMed] [Google Scholar]
- 25.Conforto AB, Cohen LG, dos Santos RL, Scaff M, Marie SK. Effects of somatosensory stimulation on motor function in chronic cortico-subcortical strokes. J Neurol. 2007;254(3):333–339. doi: 10.1007/s00415-006-0364-z. [DOI] [PubMed] [Google Scholar]
- 26.Sonde L, Gip C, Fernaeus SE, Nilsson CG, Viitanen M. Stimulation with low frequency (1.7 Hz) transcutaneous electric nerve stimulation (low-tens) increases motor function of the post-stroke paretic arm. Scand J Rehabil Med. 1998;30(2):95–99. doi: 10.1080/003655098444192. [DOI] [PubMed] [Google Scholar]
- 27.Yozbatiran N, Donmez B, Kayak N, Bozan O. Electrical stimulation of wrist and fingers for sensory and functional recovery in acute hemiplegia. Clin Rehabil. 2006;20(1):4–11. doi: 10.1191/0269215506cr928oa. [DOI] [PubMed] [Google Scholar]
- 28.Carrico C, Chelette KC, 2nd, Westgate PM, Salmon-Powell E, Nichols L, Sawaki L. Randomized trial of peripheral nerve stimulation to enhance modified constraint-induced therapy after stroke. Am J Phys Med Rehabil. 2016;95(6):397–406. doi: 10.1097/PHM.0000000000000476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Carrico C, Chelette KC, 2nd, Westgate PM, Powell E, Nichols L, Fleischer A, et al. Nerve stimulation enhances task-oriented training in chronic, severe motor deficit after stroke: a randomized trial. Stroke. 2016;47(7):1879–1884. doi: 10.1161/STROKEAHA.116.012671. [DOI] [PubMed] [Google Scholar]
- 30.Peurala SH, Pitkanen K, Sivenius J, Tarkka IM. Cutaneous electrical stimulation may enhance sensorimotor recovery in chronic stroke. Clin Rehabil. 2002;16(7):709–716. doi: 10.1191/0269215502cr543oa. [DOI] [PubMed] [Google Scholar]
- 31.Cordo P, Wolf S, Lou JS, Bogey R, Stevenson M, Hayes J, Roth E. Treatment of severe hand impairment following stroke by combining assisted movement, muscle vibration, and biofeedback. J Neurol Phys Ther. 2013;37(4):194–203. doi: 10.1097/NPT.0000000000000023. [DOI] [PubMed] [Google Scholar]
- 32.Cordo P, Lutsep H, Cordo L, Wright WG, Cacciatore T, Skoss R. Assisted movement with enhanced sensation (AMES): coupling motor and sensory to remediate motor deficits in chronic stroke patients. Neurorehabil Neural Repair. 2009;23(1):67–77. doi: 10.1177/1545968308317437. [DOI] [PubMed] [Google Scholar]
- 33.Gomes-Osman J, Field-Fote EC. Cortical vs. afferent stimulation as an adjunct to functional task practice training: a randomized, comparative pilot study in people with cervical spinal cord injury. Clin Rehabil. 2015;29(8):771–782. doi: 10.1177/0269215514556087. [DOI] [PubMed] [Google Scholar]
- 34.Hoffman L, Field-Fote E. Effects of practice combined with somatosensory or motor stimulation on hand function in persons with spinal cord injury. Top Spinal Cord Inj Rehabil. 2013;19(4):288–299. doi: 10.1310/sci1904-288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yu A, Yick KL, Ng SP, Yip J. Case study on the effects of fit and material of sports gloves on hand performance. Appl Ergon. 2019;75:17–26. doi: 10.1016/j.apergo.2018.09.007. [DOI] [PubMed] [Google Scholar]
- 36.Kinoshita H. Effect of gloves on prehensile forces during lifting and holding tasks. Ergonomics. 1999;42(10):1372–1385. doi: 10.1080/001401399185018. [DOI] [PubMed] [Google Scholar]
- 37.Morrow CM, Johnson E, Simpson KN, Seo NJ. Determining factors that influence adoption of new post-stroke sensorimotor rehabilitation devices in the USA. IEEE Trans Neural Syst Rehabil Eng. 2021;29:1213–1222. doi: 10.1109/TNSRE.2021.3090571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Smith L, Brouwer B. Effectiveness of muscle vibration in modulating corticospinal excitability. J Rehabil Res Dev. 2005;42(6):787–794. doi: 10.1682/JRRD.2005.02.0041. [DOI] [PubMed] [Google Scholar]
- 39.Lakshminarayanan K, Wang F, Webster JG, Seo NJ. Feasibility and usability of a wearable orthotic for stroke survivors with hand impairment. Disabil Rehabil Assist Technol. 2017;12(2):175–183. doi: 10.3109/17483107.2015.1111945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Seo NJ, Enders LR, Fortune A, Cain S, Vatinno AA, Schuster E, Ramakrishnan V, Feng W. Phase I Safety Trial: Extended daily peripheral sensory stimulation using a wrist-worn vibrator in stroke survivors. Transl Stroke Res. 2020;11(2):204–213. doi: 10.1007/s12975-019-00724-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang F, Lakshminarayanan K, Slota GP, Seo NJ, Webster JG. An MRI-compatible hand sensory vibrotactile system. Physiol Meas. 2015;36(1):N15–N21. doi: 10.1088/0967-3334/36/1/N15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vallbo AB. Microneurography: how it started and how it works. J Neurophysiol. 2018;120(3):1415–1427. doi: 10.1152/jn.00933.2017. [DOI] [PubMed] [Google Scholar]
- 43.Ward LM. Physics of neural synchronisation mediated by stochastic resonance. Contemporary Physics. 2009;50(5):563–574. doi: 10.1080/00107510902879246. [DOI] [Google Scholar]
- 44.Moss F, Ward LM, Sannita WG. Stochastic resonance and sensory information processing: a tutorial and review of application. Clin Neurophysiol. 2004;115(2):267–281. doi: 10.1016/j.clinph.2003.09.014. [DOI] [PubMed] [Google Scholar]
- 45.Seo NJ, Lakshminarayanan K, Lauer AW, Ramakrishnan V, Schmit BD, Hanlon CA, et al. Use of imperceptible wrist vibration to modulate sensorimotor cortical activity. Exp Brain Res. 2019;237(3):805–16. [DOI] [PMC free article] [PubMed]
- 46.Seo NJ, Lakshminarayanan K, Bonilha L, Lauer AW, Schmit BD. Effect of imperceptible vibratory noise applied to wrist skin on fingertip touch evoked potentials - an EEG study. Physiol Rep. 2015;3(11):e12624. doi: 10.14814/phy2.12624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ward LM, MacLean SE, Kirschner A. Stochastic resonance modulates neural synchronization within and between cortical sources. PLoS One. 2010;5(12):e14371. doi: 10.1371/journal.pone.0014371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Fries P. Rhythms for cognition: communication through coherence. Neuron. 2015;88(1):220–235. doi: 10.1016/j.neuron.2015.09.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Collins JJ, Imhoff TT, Grigg P. Noise-enhanced tactile sensation. Nature. 1996;383(6603):770. doi: 10.1038/383770a0. [DOI] [PubMed] [Google Scholar]
- 50.Enders LR, Hur P, Johnson MJ, Seo NJ. Remote vibrotactile noise improves light touch sensation in stroke survivors' fingertips via stochastic resonance. J Neuroeng Rehabil. 2013;10(1):105. doi: 10.1186/1743-0003-10-105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Seo NJ, Kosmopoulos ML, Enders LR, Hur P. Effect of remote sensory noise on hand function post stroke. Front Hum Neurosci. 2014;8:934. doi: 10.3389/fnhum.2014.00934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lakshminarayanan K, Lauer AW, Ramakrishnan V, Webster JG, Seo NJ. Application of vibration to wrist and hand skin affects fingertip tactile sensation. Physiol Rep. 2015;3(7):e12465. doi: 10.14814/phy2.12465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Seo NJ, Woodbury ML, Bonilha L, Ramakrishnan V, Kautz SA, Downey RJ, Dellenbach BHS, Lauer AW, Roark CM, Landers LE, Phillips SK, Vatinno AA. TheraBracelet stimulation during task-practice therapy to improve upper extremity function after stroke: a pilot randomized controlled study. Phys Ther. 2019;99(3):319–328. doi: 10.1093/ptj/pzy143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Vatinno AA, Hall L, Cox H, Fluharty A, Taylor C, Wease A, Davis A, Cain S, Ramakrishnan V, Woodbury M, Seo NJ. Using subthreshold vibratory stimulation during poststroke rehabilitation therapy: a case series. OTJR (Thorofare N J). 2022;42(1):30–39. doi: 10.1177/15394492211042275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Woodbury M, Velozo CA, Thompson PA, Light K, Uswatte G, Taub E, Winstein CJ, Morris D, Blanton S, Nichols-Larsen DS, Wolf SL. Measurement structure of the Wolf Motor Function Test: implications for motor control theory. Neurorehabil Neural Repair. 2010;24(9):791–801. doi: 10.1177/1545968310370749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bohannon RW, Smith MB. Interrater reliability of a modified Ashworth scale of muscle spasticity. Phys Ther. 1987;67(2):206–207. doi: 10.1093/ptj/67.2.206. [DOI] [PubMed] [Google Scholar]
- 57.Seo NJ, Sindhu BS, Shechtman O. Influence of pain associated with musculoskeletal disorders on grip force timing. J Hand Ther. 2011;24(4):335–343. doi: 10.1016/j.jht.2011.06.004. [DOI] [PubMed] [Google Scholar]
- 58.Lang CE, Birkenmeier RL. Upper-extremity task-specific training after stroke or disability: a manual for occupational therapy and physical therapy. 1st ed: AOTA Press; 2013.
- 59.Fleet A, Page SJ, MacKay-Lyons M, Boe SG. Modified constraint-induced movement therapy for upper extremity recovery post stroke: what is the evidence? Top Stroke Rehabil. 2014;21(4):319–331. doi: 10.1310/tsr2104-319. [DOI] [PubMed] [Google Scholar]
- 60.Birkenmeier RL, Prager EM, Lang CE. Translating animal doses of task-specific training to people with chronic stroke in 1-hour therapy sessions: a proof-of-concept study. Neurorehabil Neural Repair. 2010;24(7):620–635. doi: 10.1177/1545968310361957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lang CE, Strube MJ, Bland MD, Waddell KJ, Cherry-Allen KM, Nudo RJ, Dromerick AW, Birkenmeier RL. Dose response of task-specific upper limb training in people at least 6 months poststroke: A phase II, single-blind, randomized, controlled trial. Ann Neurol. 2016;80(3):342–354. doi: 10.1002/ana.24734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Winstein CJ, Wolf SL, Dromerick AW, Lane CJ, Nelsen MA, Lewthwaite R, et al. Effect of a task-oriented rehabilitation program on upper extremity recovery following motor stroke: the ICARE randomized clinical trial. JAMA. 2016;315(6):571–581. doi: 10.1001/jama.2016.0276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Page SJ, Sisto S, Levine P, McGrath RE. Efficacy of modified constraint-induced movement therapy in chronic stroke: a single-blinded randomized controlled trial. Arch Phys Med Rehabil. 2004;85(1):14–18. doi: 10.1016/S0003-9993(03)00481-7. [DOI] [PubMed] [Google Scholar]
- 64.Platz T, Eickhof C, van Kaick S, Engel U, Pinkowski C, Kalok S, Pause M. Impairment-oriented training or Bobath therapy for severe arm paresis after stroke: a single-blind, multicentre randomized controlled trial. Clin Rehabil. 2005;19(7):714–724. doi: 10.1191/0269215505cr904oa. [DOI] [PubMed] [Google Scholar]
- 65.Wolf SL, Winstein CJ, Miller JP, Taub E, Uswatte G, Morris D, et al. Effect of constraint-induced movement therapy on upper extremity function 3 to 9 months after stroke: the EXCITE randomized clinical trial. JAMA. 2006;296(17):2095–2104. doi: 10.1001/jama.296.17.2095. [DOI] [PubMed] [Google Scholar]
- 66.Waddell KJ, Birkenmeier RL, Moore JL, Hornby TG, Lang CE. Feasibility of high-repetition, task-specific training for individuals with upper-extremity paresis. Am J Occup Ther. 2014;68(4):444–453. doi: 10.5014/ajot.2014.011619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Nudo RJ, Milliken GW, Jenkins WM, Merzenich MM. Use-dependent alterations of movement representations in primary motor cortex of adult squirrel monkeys. J Neurosci. 1996;16(2):785–807. doi: 10.1523/JNEUROSCI.16-02-00785.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Carey JR, Kimberley TJ, Lewis SM, Auerbach EJ, Dorsey L, Rundquist P, Ugurbil K. Analysis of fMRI and finger tracking training in subjects with chronic stroke. Brain. 2002;125(Pt 4):773–788. doi: 10.1093/brain/awf091. [DOI] [PubMed] [Google Scholar]
- 69.Pascual-Leone A, Nguyet D, Cohen LG, Brasil-Neto JP, Cammarota A, Hallett M. Modulation of muscle responses evoked by transcranial magnetic stimulation during the acquisition of new fine motor skills. J Neurophysiol. 1995;74(3):1037–1045. doi: 10.1152/jn.1995.74.3.1037. [DOI] [PubMed] [Google Scholar]
- 70.Dayan E, Cohen LG. Neuroplasticity subserving motor skill learning. Neuron. 2011;72(3):443–454. doi: 10.1016/j.neuron.2011.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Plautz EJ, Milliken GW, Nudo RJ. Effects of repetitive motor training on movement representations in adult squirrel monkeys: role of use versus learning. Neurobiol Learn Mem. 2000;74(1):27–55. doi: 10.1006/nlme.1999.3934. [DOI] [PubMed] [Google Scholar]
- 72.Hosp JA, Luft AR. Cortical plasticity during motor learning and recovery after ischemic stroke. Neural Plast. 2011;2011:871296. doi: 10.1155/2011/871296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Guadagnoli MA, Lee TD. Challenge point: a framework for conceptualizing the effects of various practice conditions in motor learning. J Mot Behav. 2004;36(2):212–224. doi: 10.3200/JMBR.36.2.212-224. [DOI] [PubMed] [Google Scholar]
- 74.Seo NJ, Armstrong TJ, Ashton-Miller JA, Chaffin DB. The effect of torque direction and cylindrical handle diameter on the coupling between the hand and a cylindrical handle. J Biomech. 2007;40(14):3236–3243. doi: 10.1016/j.jbiomech.2007.04.023. [DOI] [PubMed] [Google Scholar]
- 75.Seo NJ, Armstrong TJ. Investigation of grip force, normal force, contact area, hand size, and handle size for cylindrical handles. Hum Factors. 2008;50(5):734–744. doi: 10.1518/001872008X354192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Park J, Seo NJ, Son J, Kim W, Cheong J. Postural variation of hand precision grips by object size. J Mech Sci Technol. 2014;28(5):1641–1651. doi: 10.1007/s12206-014-0309-x. [DOI] [Google Scholar]
- 77.Seo NJ, Armstrong TJ. Effect of elliptic handle shape on grasping strategies, grip force distribution, and twisting ability. Ergonomics. 2011;54(10):961–970. doi: 10.1080/00140139.2011.606923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hur P, Motawar B, Seo NJ. Hand breakaway strength model-effects of glove use and handle shapes on a person's hand strength to hold onto handles to prevent fall from elevation. J Biomech. 2012;45(6):958–964. doi: 10.1016/j.jbiomech.2012.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Seo NJ, Enders LR. Hand grip function assessed by the box and block test is affected by object surfaces. J Hand Ther. 2012;25(4):397–404. doi: 10.1016/j.jht.2012.04.004. [DOI] [PubMed] [Google Scholar]
- 80.Slota GP, Enders LR, Seo NJ. Improvement of hand function using different surfaces and identification of difficult movement post stroke in the Box and Block Test. Appl Ergon. 2014;45(4):833–838. doi: 10.1016/j.apergo.2013.10.014. [DOI] [PubMed] [Google Scholar]
- 81.Seo NJ, Armstrong TJ, Young JG. Effects of handle orientation, gloves, handle friction and elbow posture on maximum horizontal pull and push forces. Ergonomics. 2010;53(1):92–101. doi: 10.1080/00140130903389035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Enders LR, Seo NJ. Phalanx force magnitude and trajectory deviation increased during power grip with an increased coefficient of friction at the hand-object interface. J Biomech. 2011;44(8):1447–1453. doi: 10.1016/j.jbiomech.2011.03.020. [DOI] [PubMed] [Google Scholar]
- 83.Seo NJ, Armstrong TJ, Chaffin DB, Ashton-Miller JA. Inward torque and high-friction handles can reduce required muscle efforts for torque generation. Hum Factors. 2008;50(1):37–48. doi: 10.1518/001872008X250610. [DOI] [PubMed] [Google Scholar]
- 84.Seo NJ, Armstrong TJ, Chaffin DB, Ashton-Miller JA. The effect of handle friction and inward or outward torque on maximum axial push force. Hum Factors. 2008;50(2):227–236. doi: 10.1518/001872008X250692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Seo NJ, Armstrong TJ. Biomechanical analysis for handle stability during maximum push and pull exertions. Ergonomics. 2009;52(12):1568–1575. doi: 10.1080/00140130903287999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.McDonnell MD, Ward LM. The benefits of noise in neural systems: bridging theory and experiment. Nat Rev Neurosci. 2011;12(7):415–426. doi: 10.1038/nrn3061. [DOI] [PubMed] [Google Scholar]
- 87.Wells C, Ward LM, Chua R, Inglis JT. Touch noise increases vibrotactile sensitivity in old and young. Psychol Sci. 2005;16(4):313–320. doi: 10.1111/j.0956-7976.2005.01533.x. [DOI] [PubMed] [Google Scholar]
- 88.Collins JJ, Imhoff TT, Grigg P. Noise-mediated enhancements and decrements in human tactile sensation. Phys Rev E. 1997;56(1):923–926. doi: 10.1103/PhysRevE.56.923. [DOI] [Google Scholar]
- 89.Kurita Y, Shinohara M, Ueda J. Wearable sensorimotor enhancer for fingertip based on stochastic resonance effect. IEEE Transact Human Mach Syst. 2013;43(3):333–337. doi: 10.1109/TSMC.2013.2242886. [DOI] [Google Scholar]
- 90.Collins JJ, Priplata AA, Gravelle DC, Niemi J, Harry J, Lipsitz LA. Noise-enhanced human sensorimotor function. IEEE Eng Med Biol Mag. 2003;22(2):76–83. doi: 10.1109/MEMB.2003.1195700. [DOI] [PubMed] [Google Scholar]
- 91.Trevino M, De la Torre-Valdovinos B, Manjarrez E. Noise improves visual motion discrimination via a stochastic resonance-like phenomenon. Front Hum Neurosci. 2016;10:572. doi: 10.3389/fnhum.2016.00572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Liu W, Lipsitz LA, Montero-Odasso M, Bean J, Kerrigan DC, Collins JJ. Noise-enhanced vibrotactile sensitivity in older adults, patients with stroke, and patients with diabetic neuropathy. Arch Phys Med Rehabil. 2002;83(2):171–176. doi: 10.1053/apmr.2002.28025. [DOI] [PubMed] [Google Scholar]
- 93.Ragert P, Kalisch T, Bliem B, Franzkowiak S, Dinse HR. Differential effects of tactile high- and low-frequency stimulation on tactile discrimination in human subjects. BMC Neurosci. 2008;9(1):9. doi: 10.1186/1471-2202-9-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Perez MA, Field-Fote EC, Floeter MK. Patterned sensory stimulation induces plasticity in reciprocal Ia inhibition in humans. J Neurosci. 2003;23(6):2014–2018. doi: 10.1523/JNEUROSCI.23-06-02014.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Bolanowski SJ, Jr, Gescheider GA, Verrillo RT, Checkosky CM. Four channels mediate the mechanical aspects of touch. J Acoust Soc Am. 1988;84(5):1680–1694. doi: 10.1121/1.397184. [DOI] [PubMed] [Google Scholar]
- 96.Bell-Krotoski J, Tomancik E. The repeatability of testing with Semmes-Weinstein monofilaments. J Hand Surg Am. 1987;12(1):155–161. doi: 10.1016/S0363-5023(87)80189-2. [DOI] [PubMed] [Google Scholar]
- 97.Hur P, Wan Y-H, Seo NJ. Investigating the role of vibrotactile noise in early response to perturbation. IEEE Transact Biomed Engi. 2014;61(6):1628–1633. doi: 10.1109/TBME.2013.2294672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ehrenstein WH, Ehrenstein A. Psychophysical methods. In: Windhorst U, Johansson H, editors. Modern techniques in neuroscience research: Springer; 1999. p. 1211–41.
- 99.Taub E, Uswatte G, Mark VW, Morris DM, Barman J, Bowman MH, Bryson C, Delgado A, Bishop-McKay S. Method for enhancing real-world use of a more affected arm in chronic stroke: transfer package of constraint-induced movement therapy. Stroke. 2013;44(5):1383–1388. doi: 10.1161/STROKEAHA.111.000559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Rhode G, Morgan DP, Young KR. Generalization and maintenance of treatment gains of behaviorally handicapped students from resource rooms to regular classrooms using self-evaluation procedures. J Appl Behav Anal. 1983;16(2):171–188. doi: 10.1901/jaba.1983.16-171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Noland MP. The effects of self-monitoring and reinforcement on exercise adherence. Res Q Exerc Sport. 1989;60(3):216–224. doi: 10.1080/02701367.1989.10607443. [DOI] [PubMed] [Google Scholar]
- 102.Eyberg SM, Johnson SM. Multiple assessment of behavior modification with families: effects of contingency contracting and order of treated problems. J Consult Clin Psychol. 1974;42(4):594–606. doi: 10.1037/h0036723. [DOI] [PubMed] [Google Scholar]
- 103.Friedman RH. Automated telephone conversations to assess health behavior and deliver behavioral interventions. J Med Syst. 1998;22(2):95–102. doi: 10.1023/A:1022695119046. [DOI] [PubMed] [Google Scholar]
- 104.Sacco WP, Morrison AD, Malone JI. A brief, regular, proactive telephone “coaching” intervention for diabetes: rationale, description, and preliminary results. J Diabetes Complications. 2004;18(2):113–118. doi: 10.1016/S1056-8727(02)00254-4. [DOI] [PubMed] [Google Scholar]
- 105.Marcus AC, Heimendinger J, Wolfe P, Fairclough D, Rimer BK, Morra M, Warnecke R, Himes JH, Darrow SL, Davis SW, Julesberg K, Slevin-Perocchia R, Steelman M, Wooldridge J. A randomized trial of a brief intervention to increase fruit and vegetable intake: a replication study among callers to the CIS. Prev Med. 2001;33(3):204–216. doi: 10.1006/pmed.2001.0873. [DOI] [PubMed] [Google Scholar]
- 106.Wade SL, Michaud L, Brown TM. Putting the pieces together: preliminary efficacy of a family problem-solving intervention for children with traumatic brain injury. J Head Trauma Rehabil. 2006;21(1):57–67. doi: 10.1097/00001199-200601000-00006. [DOI] [PubMed] [Google Scholar]
- 107.Devellis BM, Blalock SJ, Hahn PM, Devellis RF, Hochbaum GM. Evaluation of a problem-solving intervention for patients with arthritis. Patient Educ Counsel. 1988;11(1):29–42. doi: 10.1016/0738-3991(88)90074-2. [DOI] [Google Scholar]
- 108.Glasgow RE, Toobert DJ, Barrera M, Jr, Strycker LA. Assessment of problem-solving: a key to successful diabetes self-management. J Behav Med. 2004;27(5):477–490. doi: 10.1023/B:JOBM.0000047611.81027.71. [DOI] [PubMed] [Google Scholar]
- 109.Writing Group for the Activity Counseling Trial Research G Effects of physical activity counseling in primary care: the Activity Counseling Trial: a randomized controlled trial. JAMA. 2001;286(6):677–687. doi: 10.1001/jama.286.6.677. [DOI] [PubMed] [Google Scholar]
- 110.Hallam J, Petosa R. A worksite intervention to enhance social cognitive theory constructs to promote exercise adherence. Am J Health Promot. 1998;13(1):4–7. doi: 10.4278/0890-1171-13.1.4. [DOI] [PubMed] [Google Scholar]
- 111.Chuang IC, Lin KC, Wu CY, Hsieh YW, Liu CT, Chen CL. Using Rasch analysis to validate the motor activity log and the lower functioning motor activity log in patients with stroke. Phys Ther. 2017;97(10):1030–1040. doi: 10.1093/ptj/pzx071. [DOI] [PubMed] [Google Scholar]
- 112.Lin KC, Chuang LL, Wu CY, Hsieh YW, Chang WY. Responsiveness and validity of three dexterous function measures in stroke rehabilitation. J Rehabil Res Dev. 2010;47(6):563–571. doi: 10.1682/JRRD.2009.09.0155. [DOI] [PubMed] [Google Scholar]
- 113.van der Lee JH, Beckerman H, Lankhorst GJ, Bouter LM. The responsiveness of the Action Research Arm test and the Fugl-Meyer Assessment scale in chronic stroke patients. J Rehabil Med. 2001;33(3):110–113. doi: 10.1080/165019701750165916. [DOI] [PubMed] [Google Scholar]
- 114.Rand D, Eng JJ. Predicting daily use of the affected upper extremity 1 year after stroke. J Stroke Cerebrovasc Dis. 2015;24(2):274–283. doi: 10.1016/j.jstrokecerebrovasdis.2014.07.039. [DOI] [PubMed] [Google Scholar]
- 115.Lang CE, Edwards DF, Birkenmeier RL, Dromerick AW. Estimating minimal clinically important differences of upper-extremity measures early after stroke. Arch Phys Med Rehabil. 2008;89(9):1693–1700. doi: 10.1016/j.apmr.2008.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Seo NJ, Enders LR, Motawar B, Kosmopoulos ML, Fathi-Firoozabad M. The extent of altered digit force direction correlates with clinical upper extremity impairment in chronic stroke survivors. J Biomech. 2015;48(2):383–387. doi: 10.1016/j.jbiomech.2014.11.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Seo NJ, Rymer WZ, Kamper DG. Altered digit force direction during pinch grip following stroke. Exp Brain Res. 2010;202(4):891–901. doi: 10.1007/s00221-010-2193-7. [DOI] [PubMed] [Google Scholar]
- 118.Vatinno AA, Schranz C, Simpson A, Ramakrishnan V, Bonilha L, Seo NJ. Predicting upper extremity motor improvement following therapy using EEG-based connectivity in chronic stroke. NeuroRehabilitation. 2022;50(1):105–113. doi: 10.3233/NRE-210171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Fritz SL, Blanton S, Uswatte G, Taub E, Wolf SL. Minimal detectable change scores for the Wolf Motor Function Test. Neurorehabil Neural Repair. 2009;23(7):662–667. doi: 10.1177/1545968309335975. [DOI] [PubMed] [Google Scholar]
- 120.Lin KC, Hsieh YW, Wu CY, Chen CL, Jang Y, Liu JS. Minimal detectable change and clinically important difference of the Wolf Motor Function Test in stroke patients. Neurorehabil Neural Repair. 2009;23(5):429–434. doi: 10.1177/1545968308331144. [DOI] [PubMed] [Google Scholar]
- 121.Lin KC, Fu T, Wu CY, Wang YH, Liu JS, Hsieh CJ, Lin SF. Minimal detectable change and clinically important difference of the Stroke Impact Scale in stroke patients. Neurorehabil Neural Repair. 2010;24(5):486–492. doi: 10.1177/1545968309356295. [DOI] [PubMed] [Google Scholar]
- 122.Chen HM, Chen CC, Hsueh IP, Huang SL, Hsieh CL. Test-retest reproducibility and smallest real difference of 5 hand function tests in patients with stroke. Neurorehabil Neural Repair. 2009;23(5):435–440. doi: 10.1177/1545968308331146. [DOI] [PubMed] [Google Scholar]
- 123.Freyer F, Reinacher M, Nolte G, Dinse HR, Ritter P. Repetitive tactile stimulation changes resting-state functional connectivity-implications for treatment of sensorimotor decline. Front Hum Neurosci. 2012;6:144. doi: 10.3389/fnhum.2012.00144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Morris DM, Uswatte G, Crago JE, Cook EW, 3rd, Taub E. The reliability of the wolf motor function test for assessing upper extremity function after stroke. Arch Phys Med Rehabil. 2001;82(6):750–755. doi: 10.1053/apmr.2001.23183. [DOI] [PubMed] [Google Scholar]
- 125.Wolf SL, Catlin PA, Ellis M, Archer AL, Morgan B, Piacentino A. Assessing Wolf motor function test as outcome measure for research in patients after stroke. Stroke. 2001;32(7):1635–1639. doi: 10.1161/01.STR.32.7.1635. [DOI] [PubMed] [Google Scholar]
- 126.Platz T, Pinkowski C, van Wijck F, Kim IH, di Bella P, Johnson G. Reliability and validity of arm function assessment with standardized guidelines for the Fugl-Meyer Test, Action Research Arm Test and Box and Block Test: a multicentre study. Clin Rehabil. 2005;19(4):404–411. doi: 10.1191/0269215505cr832oa. [DOI] [PubMed] [Google Scholar]
- 127.Desrosiers J, Bravo G, Hebert R, Dutil E, Mercier L. Validation of the Box and Block Test as a measure of dexterity of elderly people: reliability, validity, and norms studies. Arch Phys Med Rehabil. 1994;75(7):751–755. doi: 10.1016/0003-9993(94)90130-9. [DOI] [PubMed] [Google Scholar]
- 128.Yozbatiran N, Der-Yeghiaian L, Cramer SC. A standardized approach to performing the action research arm test. Neurorehabil Neural Repair. 2008;22(1):78–90. doi: 10.1177/1545968307305353. [DOI] [PubMed] [Google Scholar]
- 129.Lin JH, Hsu MJ, Sheu CF, Wu TS, Lin RT, Chen CH, Hsieh CL. Psychometric comparisons of 4 measures for assessing upper-extremity function in people with stroke. Phys Ther. 2009;89(8):840–850. doi: 10.2522/ptj.20080285. [DOI] [PubMed] [Google Scholar]
- 130.Duncan PW, Bode RK, Min Lai S, Perera S. Glycine Antagonist in Neuroprotection Americans I. Rasch analysis of a new stroke-specific outcome scale: the Stroke Impact Scale. Arch Phys Med Rehabil. 2003;84(7):950–963. doi: 10.1016/S0003-9993(03)00035-2. [DOI] [PubMed] [Google Scholar]
- 131.Seo NJ, Arun Kumar J, Hur P, Crocher V, Motawar B, Lakshminarayanan K. Usability evaluation of low-cost virtual reality hand and arm rehabilitation games. J Rehabil Res Dev. 2016;53(3):321–334. doi: 10.1682/JRRD.2015.03.0045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Urbin MA, Waddell KJ, Lang CE. Acceleration metrics are responsive to change in upper extremity function of stroke survivors. Arch Phys Med Rehabil. 2015;96(5):854–861. doi: 10.1016/j.apmr.2014.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Connell LA, McMahon NE, Simpson LA, Watkins CL, Eng JJ. Investigating measures of intensity during a structured upper limb exercise program in stroke rehabilitation: an exploratory study. Arch Phys Med Rehabil. 2014;95(12):2410–2419. doi: 10.1016/j.apmr.2014.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Rand D, Givon N, Weingarden H, Nota A, Zeilig G. Eliciting upper extremity purposeful movements using video games: a comparison with traditional therapy for stroke rehabilitation. Neurorehabil Neural Repair. 2014;28(8):733–739. doi: 10.1177/1545968314521008. [DOI] [PubMed] [Google Scholar]
- 135.Coker-Bolt P, Downey RJ, Connolly J, Hoover R, Shelton D, Seo NJ. Exploring the feasibility and use of accelerometers before, during, and after a camp-based CIMT program for children with cerebral palsy. J Pediatr Rehabil Med. 2017;10(1):27–36. doi: 10.3233/PRM-170408. [DOI] [PubMed] [Google Scholar]
- 136.Enders LR, Seo NJ. Altered phalanx force direction during power grip following stroke. Exp Brain Res. 2015;233(6):1677–1688. doi: 10.1007/s00221-015-4241-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Enders LR, Seo NJ. Effects of sensory deficit on phalanx force deviation during power grip post stroke. J Mot Behav. 2017;49(1):55–66. doi: 10.1080/00222895.2016.1191416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Seo NJ, Fischer HW, Bogey RA, Rymer WZ, Kamper DG. Use of visual force feedback to improve digit force direction during pinch grip in persons with stroke: a pilot study. Arch Phys Med Rehabil. 2011;92(1):24–30. doi: 10.1016/j.apmr.2010.08.016. [DOI] [PubMed] [Google Scholar]
- 139.Blennerhassett JM, Matyas TA, Carey LM. Impaired discrimination of surface friction contributes to pinch grip deficit after stroke. Neurorehabil Neural Repair. 2007;21(3):263–272. doi: 10.1177/1545968306295560. [DOI] [PubMed] [Google Scholar]
- 140.Johansson RS, Westling G. Roles of glabrous skin receptors and sensorimotor memory in automatic control of precision grip when lifting rougher or more slippery objects. Exp Brain Res. 1984;56(3):550–564. doi: 10.1007/BF00237997. [DOI] [PubMed] [Google Scholar]
- 141.Hermsdorfer J, Hagl E, Nowak DA, Marquardt C. Grip force control during object manipulation in cerebral stroke. Clin Neurophysiol. 2003;114(5):915–929. doi: 10.1016/S1388-2457(03)00042-7. [DOI] [PubMed] [Google Scholar]
- 142.Nowak DA, Hermsdorfer J, Glasauer S, Philipp J, Meyer L, Mai N. The effects of digital anaesthesia on predictive grip force adjustments during vertical movements of a grasped object. Eur J Neurosci. 2001;14(4):756–762. doi: 10.1046/j.0953-816x.2001.01697.x. [DOI] [PubMed] [Google Scholar]
- 143.Nowak DA, Hermsdorfer J. Grip force behavior during object manipulation in neurological disorders: toward an objective evaluation of manual performance deficits. Mov Disord. 2005;20(1):11–25. doi: 10.1002/mds.20299. [DOI] [PubMed] [Google Scholar]
- 144.Seo NJ. Dependence of safety margins in grip force on isometric push force levels in lateral pinch. Ergonomics. 2009;52(7):840–847. doi: 10.1080/00140130802578555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Baldassarre A, Ramsey LE, Siegel JS, Shulman GL, Corbetta M. Brain connectivity and neurological disorders after stroke. Curr Opin Neurol. 2016;29(6):706–713. doi: 10.1097/WCO.0000000000000396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Tadel F, Baillet S, Mosher JC, Pantazis D, Leahy RM. Brainstorm: a user-friendly application for MEG/EEG analysis. Comput Intell Neurosci. 2011;2011:879716–879713. doi: 10.1155/2011/879716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.US Department of Health and Human Services National Institutes of Health National Cancer Institute. Common terminology criteria for adverse events (CTCAE) version 5.0 2017 [Accessed March 18th, 2022]. Available from: https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm#ctc_50.
- 148.NINDS. Adverse event relatedness scale 2017 [Accessed March 18th, 2022]. Available from: https://cde.nlm.nih.gov/deView?tinyId=XrsKeH6EJCC.
- 149.Reviewing and reporting unanticipated problems involving risks to subjects or others and adverse events: OHRP Guidance (2007) 2007 [Accessed March 18th, 2022]. Available from: http://www.hhs.gov/ohrp/policy/advevntguid.html.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Reasonable requests for access to the final trial dataset should be submitted to the principal investigator.