Dear Editor,
We read with interest Makinson et al.’s work [1], which found that opportunistic infection (OI) incidence rates in people with HIV (PWH) with cancer and controlled disease were similar to the incidence in HIV- controls with solid cancers in a study nested in a veterans cohort. In these patients, pneumocystis infections were rare, and mostly undocumented, and no cases of toxoplasmosis were described. Among 909 PWH with CD4 counts >200/mm3 and controlled viremia, only 2 possible pulmonary pneumocystosis (PCP) infections occurred, without microbiological confirmation.
We found similar results in an analysis from the DAT’AIDS French national cohort in PWH with solid cancers between January 2005 and September 2018. Our study aimed to describe PCP and toxoplasmosis cases in PWH with successful virological control with CD4 levels >200/mm3 who had been treated for solid malignancies.
DAT’AIDS is a prospective cohort of 71 141 subjects that covers PWH treated in 23 French public hospitals, based on a computerized real-time medical record that is used by clinicians who collect demographic, behavioral, epidemiological, clinical, and biological information in a database using anonymous, coded identification numbers. All subjects included in the cohort received oral information and gave written consent (ClinicalTrials.gov NCT02898987). We used International Classification of Diseases, Tenth Revision, codes to retrieve types of solid nonhematological cancers, excluding Kaposi sarcoma and basal cell carcinoma, and PCP or toxoplasmosis infections.
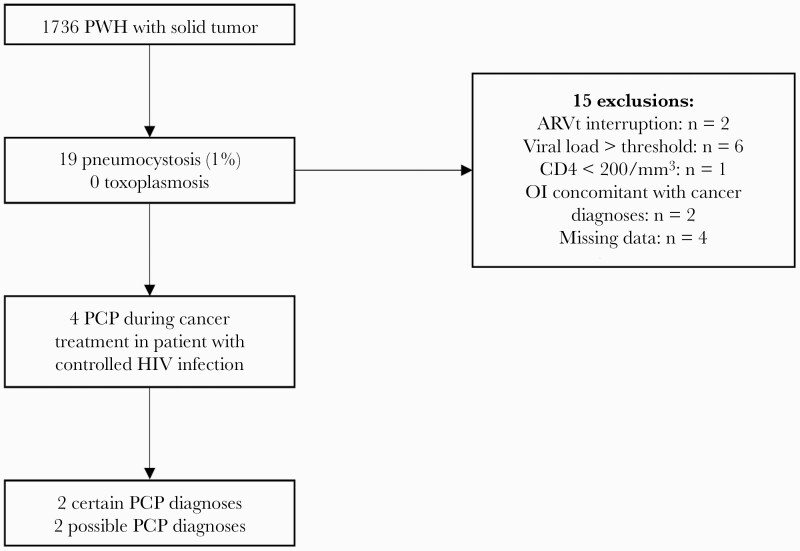
Among 1736 PWH with solid malignancies, 19 (1.1%) developed PCP, and none developed toxoplasmosis infection. All files of patients with PCP have been reviewed and validated. Only 4 PWH developed PCP during treatment despite HIV-controlled disease and CD4 levels >200 CD4/mm3 at malignancy diagnosis (data shown in Figure 1). Three had lung cancer, treated by chemotherapy for 1 (no available data on chemotherapy type), and 2 received a combination of radio-chemotherapy (carboplatin + pemetrexed followed by docetaxel for the first and cisplatin + etoposide and corticosteroids for the second). One had a thymoma treated by cisplatin + cyclophosphamide + doxorubicin followed by radiotherapy. Among patients with PCP, at cancer diagnosis the mean CD4 count was 544/mm3, and all viral loads were under the limit of detection. PCP occurred at a median of 306 days after cancer diagnosis. Only 1 PWH had a CD4 count <200 cells/mm3 at PCP diagnosis, and only 1 had received prophylaxis with pentamidine nebulization. Two PCP diagnoses were considered certain, with compatible clinical and radiological presentations and isolation of pneumocystis DNA in respiratory samples. No information regarding immunofluorescence or betaDglucan was available.
Figure 1.
Flowchart. Abbreviations: ARVt, antiretroviral therapy; OI, opportunistic infection; PCP, pulmonary pneumocystosis; PWH, people with HIV.
We surveyed 109 practitioners from 23 French centers participating in the DAT’AIDS cohort on their practice of PCP and toxoplasmosis prophylaxis: 6% prescribed systematic prophylaxis at cancer diagnosis, 61% only if chemotherapy and/or radiotherapy were started independently of CD4 T-cell count as recommended by French guidelines [2], 27% if the patient’s CD4 count fell <200/mm3 during follow-up, and 6% did not as long as the patient’s viral load remained undetectable.
We believe that our results are in line with a very low incidence of PCP risk in PWH treated for solid cancers, as in the general population. In patients without HIV infection, case reports of PCP infections in people with solid cancers have indeed been described with lung tumors, breast tumors, gastrointestinal solid tumors, and glioblastomas [3–6], particularly in patients treated with high doses of corticosteroids [5, 7, 8]. The association of chemotherapy and radiotherapy also seemed to increase risk of opportunistic infections [7].
To conclude, our data strengthen Makinson et al.’s results [1] and show low incidence of PCP and toxoplasmosis in PWH with solid cancers and controlled HIV disease, and systematic prophylaxis of these infections in patients with controlled HIV disease should probably not be systematic but individually assessed.
Acknowledgments
Financial support. No specific funding has been provided for this research.
Potential conflicts of interest. N.P., A.M., I.P.M., V.J., C.A., L.H., C.D., J.L., V.L.M., T.J., and J.R. do not have any associations that might pose a conflict of interest. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
Author contributions. N.P., A.M., and T.J. were responsible for the design and conception of the study, data collecting and analysis, and drafting, revision, and final approval of the manuscript. I.P.M., V.J., C.A., L.H., C.D., J.L., V.L.M., and J.R. made contributions to the data interpretation, critical revision of the manuscript, and final approval for publication.
Patient consent. All subjects included in the cohort received oral information and gave written consent (ClinicalTrials.gov NCT02898987). According to French law, this cohort was approved by French Commission Nationale Informatique et libertés (CNIL).
References
- 1. Makinson A, Park LS, Stone K, et al. Risks of opportunistic infections in people with human immunodeficiency virus with cancers treated with chemotherapy. Open Forum Infect Dis 2021; 8:XXX–XX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Morlat P. Prise en charge médicale des personnes vivant avec le VIH, recommandations du groupe d'experts. Rapport 2018. Available at: https://cns.sante.fr/actualites/prise-en-charge-du-vih-recommandations-du-groupe-dexperts. Accessed 25 March 2022. [Google Scholar]
- 3. Velcheti V, Govindan R.. Pneumocystis pneumonia in a patient with non-small cell lung cancer (NSCLC) treated with pemetrexed containing regimen. Lung Cancer Amst Neth 2007; 57:240–2. [DOI] [PubMed] [Google Scholar]
- 4. Lingaratnam SM, Slavin MA, Thursky KA, et al. Pneumocystis jirovecii pneumonia associated with gemcitabine chemotherapy: experience at an Australian center and recommendations for targeted prophylaxis. Leuk Lymphoma 2015; 56:157–62. [DOI] [PubMed] [Google Scholar]
- 5. De Vos FY, Gijtenbeek JM, Bleeker-Rovers CP, van Herpen CM.. Pneumocystis jirovecii pneumonia prophylaxis during temozolomide treatment for high-grade gliomas. Crit Rev Oncol Hematol 2013; 85:373–82. [DOI] [PubMed] [Google Scholar]
- 6. Watanabe H, Kitahara Y, Murakami Y, et al. Pneumocystis jirovecii pneumonia in a patient with breast cancer receiving neoadjuvant dose-dense chemotherapy. Intern Med Tokyo Jpn 2020; 59:987–0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lee EH, Kim EY, Lee SH, et al. Risk factors and clinical characteristics of Pneumocystis jirovecii pneumonia in lung cancer. Sci Rep 2019; 9:2094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Yale SH, Limper AH.. Pneumocystis carinii pneumonia in patients without acquired immunodeficiency syndrome: associated illness and prior corticosteroid therapy. Mayo Clin Proc 1996; 71:5–13. [DOI] [PubMed] [Google Scholar]