Abstract
Introduction; Pulmonary fibrosis is a frequently reported COVID-19 sequela in which the exact prevalence and risk factors are yet to be established. This meta-analysis aims to investigate the prevalence of post-COVID-19 pulmonary fibrosis (PCPF) and the potential risk factors.
Methods; CINAHL, PubMed/MEDLINE, Cochrane Library, Web of Science, and EMBASE databases were searched to identify English language studies published up to December 3, 2021.
Results; The systematic search initially revealed a total of 618 articles - of which only 13 studies reporting 2018 patients were included in this study. Among the patients, 1047 (51.9%) were male and 971 (48.1%) were female. The mean age was 54.5 years (15–94). The prevalence of PCPF was 44.9%. The mean age was 59 years in fibrotic patients and 48.5 years in non-fibrotic patients. Chronic obstructive pulmonary disease was the only comorbidity associated with PCPF. Fibrotic patients more commonly suffered from persistent symptoms of dyspnea, cough, chest pain, fatigue, and myalgia (p-value < 0.05). Factors related to COVID-19 severity that were associated with PCPF development included computed tomography score of ≥18, ICU admission, invasive/non-invasive mechanical ventilation, longer hospitalization period, and steroid, antibiotic and immunoglobulin treatments (p-value < 0.05). Parenchymal bands (284/341), ground-glass opacities (552/753), interlobular septal thickening (220/381), and consolidation (197/319) were the most common lung abnormalities found in fibrotic patients.
Conclusion, About 44.9% of COVID-19 survivors appear to have developed pulmonary fibrosis. Factors related to COVID-19 severity were significantly associated with PCPF development.
Keywords: COVID-19, Pulmonary fibrosis, Post-COVID, Meta-analysis, Prevalence
Highlights
-
•
Pulmonary fibrosis is a severe and frequently reported COVID-19 sequela.
-
•
Its prevalence in COVID-19 survivors and risk factors are yet to be established.
-
•
This meta-analysis aims to investigate the prevalence of post-COVID-19 pulmonary fibrosis and the potential risk factors.
1. Introduction
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a recently emerged viral pathogen that leads to coronavirus disease-2019 (COVID-19) [1]. It belongs to the Coronaviridae family; these viruses have crown-like spikes on their outer surfaces [2]. The viral disease was first recorded in China in late 2019, and since then, has quickly spread across the globe, affecting every country [3]. The World Health Organization (WHO) officially announced COVID-19 as a pandemic disease in March 2020 [4]. This infectious disease is highly contagious and mainly spreads by coughing, talking within proximity, or sneezing [5]. A large proportion of infected COVID-19 cases have very mild symptoms - such as loss of taste or smell, fever, fatigue, and dry cough - or are completely asymptomatic. However, in about 14% of the cases, acute respiratory distress syndrome (ARDS) can develop which is a potentially fatal condition [6]. ARDS can especially develop in patients predisposed to certain risk factors, such as diabetes mellitus, old age, and hypertension [7]. COVID-19 is considered a systemic disease as it can adversely affect various organs, including the cardiovascular, gastrointestinal, nervous, hematopoietic, and cardiovascular systems; however, it is primarily considered a respiratory condition. This can be due to the angiotensin-converting enzyme 2 (ACE2)'s abundant presence in many organs of the body [8]. Even though it has been reported that most COVID-19 cases will fully recover after the infection, various new studies estimate that nearly 70–80% of the patients will continue to suffer from a wide range of short- or long-term post-infectious complications, especially in severe COVID-19 cases [9,10]. Pulmonary fibrosis is one of the more severe and frequently reported COVID-19 sequelae. The exact prevalence of this sequela in COVID-19 survivors and its link to the viral condition are yet to be completely established [11]. Studies investigating the prevalence of pulmonary fibrosis in previous SARS pandemics have shown a prevalence of 62% and 33% after SARS-CoV and MERS-CoV, respectively [12,13].
There is currently no systematic review or meta-analysis in the literature regarding post-COVID-19 pulmonary fibrosis (PCPF). Hence, this meta-analysis aims to shed light on the prevalence of PCPF and the potential risk factors for the disease.
2. Methods
2.1. Study protocol
This meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines [14].
2.2. Data sources and search strategy
A systematic literature search was performed through the CINAHL, PubMed/MEDLINE, Cochrane Library, Web of Science, and EMBASE databases to identify studies published up to December 3, 2021. The search was done using these keywords: (SARS-CoV-2 OR COVID-19 OR Coronavirus 2019 OR 2019-nCoV) AND (fibrotic OR fibrosis OR interstitial lung OR lung scarring OR lung injury). Article titles containing the words “cystic-fibrosis” or “liver” were filtered out of the search to easily exclude a large portion of irrelevant studies. The search was limited to humans and the English language.
2.3. Eligibility criteria
Only studies that met these inclusion criteria were included in this meta-analysis:
1) inclusion of reverse transcriptase-polymerase chain reaction (RT-PCR) confirmed COVID-19 cases who have recovered (confirmed via negative RT-PCR results, or discharged/asymptomatic at least 1 month after COVID-19 onset), 2) prospective and retrospective observational studies (cross-sectional and cohort), and 3) studies with a clear definition of fibrosis that contained both fibrotic and non-fibrotic groups for comparison. Studies published in predatory journals (not properly peer-reviewed) and those that only included recovered severe/critical COVID-19 patients were excluded in this meta-analysis to minimize bias.
2.4. Study selection process
Two independent researchers initially screened the titles and abstracts of the identified studies and then conducted a full-text screening according to the inclusion and exclusion criteria. After this, the studies eligible for inclusion were selected. In the event of disagreement, a third author stepped in to resolve it through discussion and debate.
2.5. Data items
Multiple data were collected from the included articles, including the year of publication, first author, country, study design, sex, age, smoking status, comorbidities, body mass index (BMI), the prevalence of pulmonary fibrosis, assessment time, the severity of COVID-19, and fibrotic lung abnormalities.
2.6. Data analysis and synthesis
The extracted data were used in qualitative synthesis. They were re-analyzed using the Statistical Package for Social Sciences (SPSS) 25.0 software for quantitative synthesis. Summary tables containing relevant variables were produced and presented as frequency, mean, percentage, odds ratio, and p-value. The statistical level of significance was set at 0.05; thus, a p-value of less than 0.05 was considered significant.
3. Results
3.1. Study selection
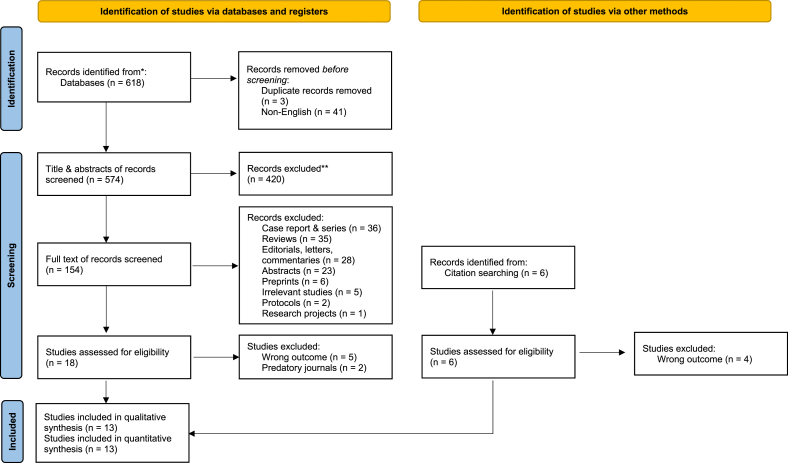
The systematic search initially revealed a total of 618 articles. Before further screening, three duplicates and 41 non-English articles were removed. The titles and abstracts of 574 articles were screened, of which 420 were excluded due to irrelevancy. Citation searching was performed for the remaining 154 articles, which revealed another 6 relevant articles. The additional full-text screening was carried out to remove wrong study designs, leading to the exclusion of 136 articles. The remaining 24 articles were assessed against the full eligibility criteria, of which 9 were excluded due to a wrong outcome and 2 were excluded due to their publication in predatory journals. Finally, 13 eligible articles were included in the meta-analysis. The detailed PRISMA flow chart is shown in Fig. 1.
Fig. 1.
Study selection PRISMA flow chart.
3.2. Characteristics of the included studies
All the included studies were observational (cohort and cross-sectional), and amongst them, seven were conducted in China, two in Egypt, one in Iran, one in the USA, one in Italy, and one in the UK. Zou et al. reported the highest prevalence of PCPF (84.15%), whereas Aul and colleagues reported the lowest prevalence (9.3%). More details regarding the characteristics of the included studies are shown in Table 1.
Table 1.
Characteristics of the included studies.
| Author | Country | Study Design | Publication date | Number of included participants | Gender |
Age | Smoking N (%) |
Comorbidities | Prevalence of pulmonary fibrosis N (%) |
Assessment time | Severe cases N (%) |
Non-severe cases N (%) |
|
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Male N (%) |
Female N (%) |
||||||||||||
| Ali et al. [22] | Egypt | Cross-sectional | 2021 | 80 | 40 (50%) | 40 (50%) | 43.2 (25–75) | 30 (37.5%) | – | 25 (31.25%) | 4–6 weeks and 9–12 weeks after recovery | 42 (52.5%) | 38 (47.5%) |
| Aul et al. [21] | UK | Cohort | 2021 | 387 | 219 (56.6%) | 168 (43.4%) | 63 (50–75) | 251 (65.5%) | - Chronic lung disease 74 (19.32%) - COPD 14 (3.65%) - Asthma 32 (8.35%) - Diabetes mellitus 101 (26.16%) - Hypertension 144 (37.4%) - Ischemic heart disease 41 (10.6%) |
36 (9.3%) | 6 weeks after discharge | – | – |
| Hu et al. [23] | China | Cohort | 2020 | 76 | 34 (44.7%) | 42 (55.3%) | 50.5 (15–84) | – | - Cardiovascular disease 9 (11.8%) - Hypertension 17 (22.4%) - Diabetes 8 (10.5%) |
46 (60.5%) | At discharge | 13 (17.1%) | 63 (82.9%) |
| Li et al. [24] | China | Cohort | 2021 | 289 | 141 (49.1%) | 148 (50.9%) | – | – | - Coronary heart disease 16 (5.57%) - Hypertension 37 (12.9%) - Diabetes 13 (4.5%) Respiratory disease 13 (4.53%) |
173 (59.86%) | 90–150 days after COVID-19 onset | 56 (19.4%) | 233 (80.6%) |
| Marvisi et al. [25] | Italy | Cohort | 2020 | 90 | 47 (52.2%) | 43 (47.8%) | 66 (±15) | 42 (46.6%) | - Hypertension 17 (18.9%) - Diabetes 10 (11.1%) - COPD 5 (5.5%) - Asthma 4 (4.4%) - Liver disease 1 (1.1%) - Chronic renal failure 3 (3.3%) |
23 (25.5%) | 8 weeks after admission | – | – |
| McGroder et al. [26] | USA | Cohort | 2021 | 76 | 45 (61%) | 31 (39%) | 54 (±13.7) | 33 (43%) | - Hypertension 41 (54%) - Diabetes 25 (33%) - COPD 4 (5%) - Asthma 18 (27%) - Heart disease 2 (3%) - Chronic Kidney Disease 7 (9%) |
32 (42%) | 4 months after admission | – | – |
| Nabahati et al. [27] | Iran | Cross-sectional | 2021 | 173 | 57 (32.9%) | 116 (67.1%) | 53.6 (18–93) | – | - CVDs 72 (41.6%) - Asthma/COPD 15 (8.6%) - Diabetes 27 (15.6%) |
90 (52%) | 3 and 6 months after discharge | 93 (53.75%) | 80 (46.25%) |
| Yang et al. [28] | China | Cohort | 2020 | 166 | 69 (41.6%) | 97 (58.4%) | 57 (±15) | – | – | 76 (46%) | 56 days after COVID-19 onset | 127 (77%) | 39 (23%) |
| Yasin et al. [29] | Egypt | Cohort | 2021 | 210 | 149 (71%) | 61 (29%) | 53.85 (18–94) | – | – | 101 (48.1%) | 41.5 days after discharge | – | – |
| Yu et al. [30] | China | Cohort | 2020 | 32 | 22 (68.7%) | 10 (31.3%) | – | – | - Diabetes 2 (6.25%) - Hypertension 4 (12.5%) - Cardiac disease 2 (6.25%) - COPD 1 (3.1%) |
14 (43.7%) | 9 days after discharge | – | – |
| Zou et al. [31] | China | Cohort | 2021 | 284 | 122 (43%) | 162 (57%) | – | – | – | 239 (84.15%) | 30, 60 and 90 days after discharge | 70 (24.7%) | 214 (75.3) |
| Han et al. [32] | China | Cohort | 2021 | 114 | 80 (70%) | 34 (30%) | 54 (±12) | 16 (14%) | - Diabetes 13 (11%) - Hypertension 32 (28%) - Chronic pulmonary disease 16 (14%) |
40 (35.1%) | 6 months after COVID-19 onset | 31 (27%) | 93 (73%) |
| Liu et al. [33] | China | Cohort | 2021 | 41 | 22 (53.7%) | 19 (46.3%) | 50 (±14) | – | - Hypertension 3 (7%) - Diabetes 1 (2%) - COPD 2 (5%) - Cardiovascular disease 1 (2%) - Hepatic disease 1 (2%) |
12 (29%) | 7 months after discharge | 15 (36.6%) | 26 (63.4%) |
3.3. Participants
In total, 2018 COVID-19 survivors with a mean age of 54.5 years (15–94) were included, of whom 1047 (51.9%) were male and 971 (48.1%) were female. Three studies did not report the average age of the patients. Smoking status was presented in only 5 studies, showing a positive history in 372 patients. Nine studies presented data regarding comorbidities, including asthma, diabetes mellitus, chronic obstructive pulmonary disease (COPD), hypertension, cardiac disease, renal disease, and chronic pulmonary disease. Eight studies determined the proportion of severe and non-severe COVID-19 patients.
3.4. Assessment time
The assessment time varied greatly between the included studies, ranging from the day of hospital discharge to 7 months after discharge. In some studies, follow-up timing was calculated after hospital discharge, in others, after symptom onset or hospital admission.
3.5. Main findings
The participants were divided into two groups based on the presence of fibrosis: the “fibrotic group” (n = 907) and the “non-fibrotic group” (n = 1111). The prevalence of PCPF was 44.9%, without a significant gender difference (53.8% were males and 46.2% were females). The fibrotic patients had a mean age of 59 years, while the non-fibrotic patients were considerably younger, with a mean age of 48.5 years (p-value = 0.0033). Only two studies presented BMI, without noticeable differences between the fibrotic and non-fibrotic patients: 25.23 and 24.75, respectively. Five studies identified the smoking history of the patients, which did not show an effect on the development of PCPF (positive history in 53.2% of fibrotic patients and 48.9% of non-fibrotic patients). PCPF was 2.88 times (95% CI 1.27, 6.52) more prevalent in patients with COPD, and it was 0.51 (95% CI 0.37, 0.70) and 0.69 (95% CI 0.53, 0.90) times less prevalent in patients with diabetes and hypertension, respectively. However, asthma, cardiac diseases, renal diseases, and chronic pulmonary disease comorbidities were irrelevant to the prevalence of PCPF (p-value > 0.05) (Table 2). Symptoms of coughing (47.4%), chest pain (27.6%), and fever (72.4%) during COVID-19 were more common amongst the fibrotic group than in the non-fibrotic group (p-value < 0.05) (Table 3). Additionally, it was observed that fibrotic patients more commonly suffered from persistent symptoms of dyspnea (50%), cough (31.6%), chest pain (30.5%), fatigue (80%), and myalgia (58.3%) when compared to non-fibrotic patients (p-value < 0.05) (Table 4). Factors related to COVID-19 severity which were significantly associated with the development of PCPF included computed tomography (CT) score of ≥18 (45.3%), ICU admission (48.3%), invasive (12%) and non-invasive (17.3%) mechanical ventilation, longer hospitalization period (a mean of 21.58 days in fibrotic patients and 12.65 days in non-fibrotic patients), and steroid (57.1%), antibiotic (60%) and immunoglobulin (36.1%) administrations (p-value < 0.05) (Table 5). The lung abnormalities found in fibrotic patients were reticulations (148/399), honeycombing (100/477), traction bronchiectasis (121/514), parenchymal bands (284/341), irregular interfaces (134/352), interlobular septal thickening (220/381), lung distortion (19/76), ground-glass opacity (552/753), meshwork (23/163), consolidation (197/319), and crazy paving (104/191) (Table 6).
Table 2.
Demographic and comorbidity profiles in fibrotic and non-fibrotic patients.
| N. patients | Fibrotic | Non-Fibrotic | P-Value | Odds Ratio (95% CI) | References | |
|---|---|---|---|---|---|---|
| Overall | 2018 | 907 (44.9%) | 1111 (55.1%) | 21–33 | ||
| Age (Grand mean) | 1938 | 59 | 48.5 | 0.0033 | 21,23-33 | |
| Gender (Male) | 1047 | 488/907 (53.8%) | 559/1111 (50.3%) | 0.1188 | 1.15 (0.96, 1.37) | 21–33 |
| Gender (Female) | 971 | 419/907 (46.2%) | 552/1111 (49.7%) | 21–33 | ||
| BMI (Grand mean) | 626 | 25.23 | 24.75 | 0.8836 | 21,24 | |
| Smoking history | 372 | 83/156 (53.2%) | 289/591 (48.9%) | 0.3391 | 1.19 (0.83, 1.69) | 21,22,25,26,32 |
| Diabetes | 201 | 64/693 (9.2%) | 137/828 (16.5%) | <0.0001 | 0.51 (0.37, 0.70) | 21,23–27,30,32,33 |
| Hypertension | 295 | 111/603 (18.4%) | 184/745 (24.7%) | 0.0056 | 0.69 (0.53, 0.90) | 21,23,24–26,30,32,33 |
| Asthma | 54 | 12/91 (13.2%) | 42/462 (9.1%) | 0.2317 | 1.52 (0.77, 3.01) | 21,25,26 |
| COPD | 26 | 10/117 (8.5%) | 16/509 (3.1%) | 0.0112 | 2.88 (1.27, 6.52) | 21,25,26,30,33 |
| Cardiac diseases | 143 | 58/403 (14.4%) | 85/671 (12.7%) | 0.4209 | 1.16 (0.81, 1.66) | 21,23,24,26,27,30,33 |
| Renal Diseases | 10 | 5/55 (9.1%) | 5/111 (4.5%) | 0.2515 | 2.12 (0.59, 7.66) | 25,26 |
| Chronic pulmonary disease | 103 | 27/249 (10.8%) | 76/541 (14%) | 0.2152 | 0.74 (0.47, 1.19) | 21,24,32 |
Table 3.
COVID-19 symptoms in fibrotic and non-fibrotic patients.
| N. patients | Fibrotic | Non-Fibrotic | P-Value | Odds Ratio (95% CI) | References | |
|---|---|---|---|---|---|---|
| Overall | 614 | 424 (69.1%) | 190 (30.9%) | 24,31,33 | ||
| Dyspnea (Breathlessness) | 5 | 3/12 (25%) | 2/29 (6.9%) | 0.1290 | 4.50 (0.65, 31.37) | 33 |
| Cough | 259 | 202/426 (47.4%) | 57/190 (30%) | 0.0001 | 2.10 (1.46, 3.03) | 24,31,33 |
| Chest Pain | 72 | 66/239 (27.6%) | 6/45 (13.3%) | 0.0492 | 2.48 (1.00, 6.13) | 31 |
| Fatigue | 39 | 23/185 (12.4%) | 16/145 (11%) | 0.6964 | 1.14 (0.58, 2.26) | 24,33 |
| Myalgia (Muscle pain) | 12 | 3/12 (25%) | 9/29 (31%) | 0.6998 | 0.74 (0.16, 3.40) | 33 |
| Fever | 213 | 134/185 (72.4%) | 79/145 (54.5%) | 0.0008 | 2.19 (1.39, 3.47) | 24,33 |
| Diarrhea | 11 | 10/173 (5.8%) | 1/116 (1%) | 0.0643 | 7.05 (0.89, 55.88) | 24 |
| Headache | 10 | 3/12 (25%) | 7/29 (24.1%) | 0.9534 | 1.05 (0.22, 4.98) | 33 |
| Expectoration (Sputum) | 28 | 10/185 (5.4%) | 18/145 (12.4%) | 0.0272 | 0.40 (0.18, 0.90) | 24,33 |
Table 4.
Persistent symptoms in fibrotic and non-fibrotic patients.
| N. patients | Fibrotic | Non-Fibrotic | P-Value | Odds Ratio (95% CI) | References | |
|---|---|---|---|---|---|---|
| Overall | 501 | 76 (15.2%) | 425 (84.8%) | 21,32 | ||
| Dyspnea (Breathlessness) | 151 | 38/76 (50%) | 113/425 (26.6%) | 0.0001 | 2.76 (1.68, 4.55) | 21,32 |
| Cough | 90 | 24/76 (31.6%) | 66/425 (15.5%) | 0.0010 | 2.51 (1.45, 4.35) | 21,32 |
| Chest Pain | 39 | 11/36 (30.5%) | 28/351 (8%) | 0.0001 | 5.08 (2.26, 11.38) | 21 |
| Fatigue | 165 | 29/36 (80%) | 136/351 (38.7%) | <0.0001 | 6.55 (2.79, 15.37) | 21 |
| Myalgia (Muscle pain) | 75 | 21/36 (58.3%) | 54/351 (15.4%) | <0.0001 | 7.70 (3.74, 15.87) | 21 |
| Expectoration (Sputum) | 11 | 5/40 (12.5%) | 6/74 (8.1%) | 0.4517 | 1.62 (0.46, 5.68) | 32 |
Table 5.
Factors related to COVID-19 severity in fibrotic and non-fibrotic patients.
| N. patients | Fibrotic | Non-Fibrotic | P-Value | Odds Ratio (95% CI) | References | |
|---|---|---|---|---|---|---|
| Overall | 1928 | 884 (45.9%) | 1044 (54.1%) | 21-24,26-33 | ||
| Severe-cases (CT Score ≥18) | 434 | 297/655 (45.3%) | 137/492 (27.8%) | <0.0001 | 2.15 (1.67, 2.76) | 22,24,27,28,31-33 |
| Non-Severe cases (CT Score <18) | 713 | 358/655 (54.6%) | 355/492 (72.1%) | 22,24,27,28,31-33 | ||
| ICU admission | 141 | 73/151 (48.3%) | 68/478 (14.2%) | <0.0001 | 5.64 (3.75, 8.50) | 21,29,30 |
| Invasive Mechanical ventilation | 52 | 40/333 (12%) | 12/353 (3.4%) | 0.0001 | 3.88 (2.0, 7.53) | 24,26,28,32,33 |
| Non-invasive Mechanical ventilation | 57 | 50/289 (17.3%) | 7/280 (2.5%) | <0.0001 | 8.16 (3.63, 18.34) | 24,28,32 |
| Days of Hospitalization (Grand mean) | 1122 | 21.58 | 12.65 | 0.00261 | 23,24,26,29-33 | |
| Steroid treatment | 439 | 282/494 (57.1%) | 157/555 (28.3%) | <0.0001 | 3.37 (2.60, 4.36) | 23,24,26,28–30,32,33 |
| Antiviral treatment | 280 | 122/163 (74.8%) | 158/212 (74.5%) | 0.9440 | 1.02 (0.64, 1.63) | 23,28,30,32 |
| Antibiotic treatment | 181 | 90/149 (60%) | 91/194 (46.9%) | 0.0134 | 1.73 (1.12, 2.66) | 23,28,32 |
| Immunoglobulin treatment | 117 | 90/249 (36.1%) | 27/206 (13.1%) | <0.0001 | 3.75 (2.32, 6.07) | 24,28 |
Table 6.
Lung abnormalities found in fibrotic patients.
| Frequency | References | |
|---|---|---|
| N. patients | 753 | 24,26–29,31-33 |
| Reticulations | 148/399 | 26,28,31-33 |
| Honeycombing | 100/477 | 26-28,31,32 |
| Traction Bronchiectasis | 121/514 | 24,26–29,32,33 |
| Parenchymal bands | 284/341 | 24,27,28,33 |
| Irregular interfaces | 134/352 | 24,28,29,33 |
| Interlobular septal thickening | 220/381 | 27,31-33 |
| lung distortion | 19/76 | 28 |
| Ground-glass opacity | 552/753 | 24,26–29,31-33 |
| Meshwork | 23/163 | 24 |
| Consolidation | 197/319 | 27-29,32,33 |
| Crazy paving | 104/191 | 27,29 |
4. Discussion
Since the beginning of the COVID-19 outbreak, more than 293 million people have been infected worldwide, of whom 256 million have recovered [15]. As the number of recovered patients is increasing, it is necessary to acquire information about pulmonary sequelae that can persist or develop after the initial recovery at least 4 weeks post COVID-19 onset. Pulmonary sequelae of COVID-19 are currently little understood; hence, information in this regard can help us in identifying the risk groups that need close follow-up. The development of pulmonary fibrosis is considered one of the key concerns regarding COVID-19 pulmonary sequelae as it is associated with architectural distortion of the lung parenchyma and overall impairment of lung function resulting in decreased quality of life [9]. Many studies have been conducted on PCPF; however, their results have been heterogeneous in some aspects. Until the time of writing this paper, no other meta-analysis to summarize the findings of these studies had been published in the literature.
The pathogenic progression of pulmonary fibrosis post-COVID-19 is yet to be fully illuminated; however, it is thought to be multifactorial. Whatever the cause, fibrosis is considered to be due to the abnormal healing of the injured lung parenchyma [16]. In COVID-19 patients, possible sources of injury include cytokine storm due to improper inflammatory response, bacterial co-infections, and thromboembolic events causing microvascular damage and endothelial dysfunction [17]. The renin-angiotensin system is also believed to be involved due to the high affinity of SARS-CoV-2 viral spike protein to the angiotensin-converting enzyme-2 (ACE2) receptor [18].
To date, there is no proven treatment for PCPF, which makes the condition even more concerning. However, anti-fibrotic drugs may be used to decrease pulmonary injury in cases with severe COVID-19 [19,20].
Previous studies have been quite heterogeneous regarding the prevalence of PCPF. Aul et al. reported a low prevalence of 9.3% [21]. Meanwhile, the rest of the available studies have reported a much higher prevalence, ranging from 25.5% to 84.15% [[22], [23], [24], [25], [26], [27], [28], [29], [30], [31], [32], [33]], with the highest being reported by Zou and associates [31].
In the current meta-analysis, the overall PCPF prevalence of all the studies was 44.9%. This is lower than that of the previous SARS epidemic (62%) and higher than that of MERS (33%) [12,13]. In our study, the mean age of the fibrotic patients was much higher (59 years) than that of those with no fibrotic changes (48.5 years). Most of the available literature shared similar findings regarding this aspect [[22], [23], [24], [25], [26], [27], [28], [29], [30], [31], [32], [33]]. However, Aul et al. reported that the age difference was insignificant between the fibrotic and non-fibrotic patients [21]. The literature has had contradictory findings regarding the effect of gender and smoking status on the development of fibrosis. Despite some studies suggesting a higher prevalence of PCPF in males [21,22,25,26], the majority of the studies disregard it [23,24,[27], [28], [29], [30], [31], [32], [33]]. Multiple studies consider smoking as a risk factor for PCPF [21,22,25]; meanwhile, others do not highlight this connection [26,32]. In our meta-analysis, male gender, smoking status, and BMI were not found to be significant risk factors for the development of PCPF.
Among all the comorbidities studied in this meta-analysis, only COPD was linked with the risk of developing PCPF. However, the findings of a few studies showed hypertensive patients as a risk group for PCPF [23,24,30].
According to two studies by Han et al. and Aul et al., recovered COVID-19 patients who have developed pulmonary fibrosis, suffer more frequently from persistent symptoms, including dyspnea, cough, chest pain, fatigue, and myalgia when compared to non-fibrotic patients [21,32]. This is in line with the findings of this meta-analysis.
According to the literature, pulmonary fibrosis can develop right after discharge or several weeks later [33]. However, a portion of these cases has been reported to improve in the months after COVID-19 recovery. In their study, Zou and colleagues showed that 30-, 60-, and 90-day follow-up of PCPF patients have confirmed that pulmonary fibrosis in some patients will resolve over time; however, fibrosis in the majority of the patients will not resolve [31]. In another study by Nabahati and associates, 6-month follow-up of PCPF patients showed that in 33.9% of the patients, pulmonary fibrosis was nearly diminished in comparison to 3-month follow-up CT scans; however, in 66.1% of the patients, no considerable changes were noticed [27]. Additionally, in a follow-up study by Han et al., it was observed that even after 1-year post-COVID-19, pulmonary fibrosis persisted in the majority of the fibrotic patients [34]. The exact reason why some patients heal after the damage done to them during COVID-19 and others develop pulmonary fibrosis is yet to be known [20].
Based on the data provided by WHO, 14% of COVID-19 cases are severe and 6% are considered critical. Previous reports, without dispute, have highlighted the severity of COVID-19 as a risk factor for PCPF development. The reported severity factors include invasive and non-invasive mechanical ventilation, a CT score of more than 18, ICU admission, and a longer hospitalization period [[21], [22], [23], [24],[26], [27], [28], [29], [30], [31], [32], [33]]. The same findings were observed in this meta-analysis. Furthermore, Zou et al. discovered that the severity of PCPF was correlational to the severity of COVID-19, with critically ill COVID-19 patients developing more severe PCPF [31]. Although it has been suggested that steroid therapy prevents irreversible lung injury [35], the current meta-analysis showed that PCPF was more common in those patients who had received steroid treatment by threefolds.
Among the lung abnormalities found in fibrotic patients, parenchymal bands, interlobular septal thickening, and coarse reticulations have been reported to be the most frequent CT findings [29,36]. Our meta-analysis showed similar findings, in addition to frequent ground-glass opacities and consolidations.
The current meta-analysis faces multiple limitations. The assessment time varied greatly between the included studies, both in terms of scale and duration, which may result in bias in the overall prevalence of PCPF. Some of the studies included a small number of participants. Only a few studies were included in some comparisons. Finally, in some aspects, there was significant heterogenicity between the included studies.
In conclusion, a significant portion of recovered COVID-19 patients (44.9%) appear to have developed pulmonary fibrosis, which may mostly persist over time. Factors related to COVID-19 severity and the requirement for steroid, immunoglobulin, and antibiotic administration were significantly associated with the development of PCPF. Hence, close follow-up of severe or critically ill COVID-19 patients is recommended. Parenchymal bands and ground-glass opacities were the most common lung abnormalities found in fibrotic patients. More comprehensive longitudinal studies are still required with larger sample sizes.
Ethical approval
Not applicable.
Please state any sources of funding for your research
No source to be stated.
Author contribution
Fahmi H. Kakamad: literature review, writing the manuscript, final approval of the manuscript. Gasha S. Ahmed, Berwn A. Abdulla, Shvan H. mohammed. Tomas M. Mikael, Rawezh Q. Salih, Razhan k. Ali, Dahat A. Hussein, Shaho F.Ahmed: literature review and final approval of the manuscript. Abdulwahid M. Salh: major contribution of the idea, final approval of the manuscript.
Please state any conflicts of interest
There is no conflict to be declared.
Registration of research studies
N/A.
Guarantor
Fahmi Hussein Kakamad is Guarantor of this submission.
Consent
Not applicable.
Provenance and peer review
Not commissioned, externally peer-reviewed.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.amsu.2022.103590.
Appendix A. Supplementary data
The following are the Supplementary data to this article:
References
- 1.Ahmad S.A., Salih K.H., Ahmed S.F., Kakamad F.H., Salh A.M., Hassan M.N., et al. Post COVID-19 transverse myelitis; a case report with review of literature. Ann. Med. Surg. 2021;69(1):1–3. doi: 10.1016/j.amsu.2021.102749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Amin A.A., Awakhti A.H., Hussein L.A., Fattah F.H., Baba H.O., Kakamad F.H., et al. Survived COVID-19 patient presented with death on arrival: a case report. Int. J. Surg. Case Rep. 2021;81(1):1–3. doi: 10.1016/j.ijscr.2021.105826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ahmad S.A., Salih B.K., Hussein K.F., Mikael T.M., Kakamad F.H., Salih A.M. Aseptic meningoencephalitis after COVID-19 vaccination: a case report. Ann. Med. Surg. 2021;71(1):1–3. doi: 10.1016/j.amsu.2021.103028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Abdullah H.M., Hama-Ali H.H., Ahmed S.N., Ali K.M., Karadakhy K.A., Mahmood S.O., et al. Severe refractory COVID-19 patients responding to convalescent plasma; A case series. Ann. Med. Surg. 2020;56(1):125–127. doi: 10.1016/j.amsu.2020.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Salih R.Q., Salih G.A., Abdulla B.A., Ahmed A.D., Mohammed H.R., Kakamad F.H., et al. False-positive HIV in a patient with SARS-CoV-2 infection; a case report. Ann. Med. Surg. 2021;71(1):1–3. doi: 10.1016/j.amsu.2021.103027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kakamad F.H., Mahmood S.O., Rahim H.M., Abdulla B.A., Abdullah H.O., Othman S., et al. Post covid-19 invasive pulmonary Aspergillosis: a case report. Int. J. Surg. Case Rep. 2021;82(1):1–3. doi: 10.1016/j.ijscr.2021.105865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ahmad S.A., Kakamad F.H., Mohamad H.S., Salih B.K., Mohammed S.H., Abdulla B.A., et al. Post COVID-19 cerebral venous sinus thrombosis; a case report. Ann. Med. Surg. 2021;72(1):1–3. doi: 10.1016/j.amsu.2021.103031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Baram A., Kakamad F.H., Abdullah H.M., Mohammed-Saeed D.H., Hussein D.A., Mohammed S.H., et al. Large vessel thrombosis in patient with COVID-19, a case series. Ann. Med. Surg. 2020;60(1):526–530. doi: 10.1016/j.amsu.2020.11.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ahmed O.F., Amin B.J., Abdullah B.A., Hassan M.N., Salih R.Q., Mohammed S.H., et al. Post COVID-19 pulmonary complications; a single center experience. Ann. Med. Surg. 2021;72(1):1–4. doi: 10.1016/j.amsu.2021.103052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Carfì A., Bernabei R., Landi F. Persistent symptoms in patients after acute COVID-19. JAMA. 2020;324(6):603–605. doi: 10.1001/jama.2020.12603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Delpino M., Quarleri J. SARS-CoV-2 pathogenesis: imbalance in the renin-angiotensin system favors lung fibrosis. Front. Cell. Infect. Microbiol. 2020;10:340. doi: 10.3389/fcimb.2020.00340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Antonio G.E., Wong K.T., Hui D.S., Wu A., Lee N., Yuen E.H., et al. Thin-section CT in patients with severe acute respiratory syndrome following hospital discharge: preliminary experience. Radiology. 2003;228(3):810–815. doi: 10.1148/radiol.2283030726. [DOI] [PubMed] [Google Scholar]
- 13.Das K.M., Lee E.Y., Singh R., Enani M.A., Al Dossari K., Van Gorkom K., et al. Follow-up chest radiographic findings in patients with MERS-CoV after recovery. Indian J. Radiol. Imag. 2017;27(3):342–349. doi: 10.4103/ijri.IJRI_469_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Page M.J., McKenzie J.E., Bossuyt P.M., Boutron I., Hoffmann T.C., Mulrow C.D., et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Int. J. Surg. 2021;88:105906. doi: 10.1016/j.ijsu.2021.105906. [DOI] [PubMed] [Google Scholar]
- 15.World Health Organization 2022. https://covid19.who.int/ January 5.
- 16.Fernandez I.E., Eickelberg O. New cellular and molecular mechanisms of lung injury and fibrosis in idiopathic pulmonary fibrosis. Lancet. 2012;380(9842):680–688. doi: 10.1016/S0140-6736(12)61144-1. [DOI] [PubMed] [Google Scholar]
- 17.Phua J., Weng L., Ling L., Egi M., Lim C.M., Divatia J.V., et al. Intensive care management of coronavirus disease 2019 (COVID-19): challenges and recommendations. Lancet Respir. Med. 2020;8(5):506–517. doi: 10.1016/S2213-2600(20)30161-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang H., Penninger J.M., Li Y., Zhong N., Slutsky A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46(4):586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wu C., Chen X., Cai Y., Zhou X., Xu S., Huang H., et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern. Med. 2020;180(7):934–943. doi: 10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.George P.M., Wells A.U., Jenkins R.G. Pulmonary fibrosis and COVID-19: the potential role for antifibrotic therapy. Lancet Respir. Med. 2020;8(8):807–815. doi: 10.1016/S2213-2600(20)30225-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Aul R., Gates J., Draper A., Dunleavy A., Ruickbie S., Meredith H., et al. Complications after discharge with COVID-19 infection and risk factors associated with development of post-COVID pulmonary fibrosis. Respir. Med. 2021;188(1):1–9. doi: 10.1016/j.rmed.2021.106602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ali R.M., Ghonimy M.B. Post-COVID-19 pneumonia lung fibrosis: a worrisome sequelae in surviving patients. Egyptian J. Radiol. Nucl. Med. 2021;52(1):1–8. [Google Scholar]
- 23.Hu Z.J., Xu J., Yin J.M., Li L., Hou W., Zhang L.L., et al. Lower circulating interferon-gamma is a risk factor for lung fibrosis in COVID-19 patients. Front. Immunol. 2020;11(1):1–10. doi: 10.3389/fimmu.2020.585647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li X., Shen C., Wang L., Majumder S., Zhang D., Deen M.J., et al. Pulmonary fibrosis and its related factors in discharged patients with new corona virus pneumonia: a cohort study. Respir. Res. 2021;22(1):1–11. doi: 10.1186/s12931-021-01798-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Marvisi M., Ferrozzi F., Balzarini L., Mancini C., Ramponi S., Uccelli M. First report on clinical and radiological features of COVID-19 pneumonitis in a Caucasian population: factors predicting fibrotic evolution. Int. J. Infect. Dis. 2020;99(1):485–488. doi: 10.1016/j.ijid.2020.08.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.McGroder C.F., Zhang D., Choudhury M.A., Salvatore M.M., D'Souza B.M., Hoffman E.A., et al. Pulmonary fibrosis 4 months after COVID-19 is associated with severity of illness and blood leucocyte telomere length. Thorax. 2021;76(1):1242–1245. doi: 10.1136/thoraxjnl-2021-217031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nabahati M., Ebrahimpour S., Khaleghnejad Tabari R., Mehraeen R. Post-COVID-19 pulmonary fibrosis and its predictive factors: a prospective study. Egyptian J. Radiol. Nucl. Med. 2021;52(1):1–7. [Google Scholar]
- 28.Yang Z.L., Chen C., Huang L., Zhou S.C., Hu Y.N., Xia L.M., et al. Fibrotic changes depicted by thin-section CT in patients with COVID-19 at the early recovery stage: preliminary experience. Front. Med. 2020;7(1):1–10. doi: 10.3389/fmed.2020.605088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yasin R., Gomaa A.A., Ghazy T., Hassanein S.A., latif Ibrahem R.A., Khalifa M.H. Predicting lung fibrosis in post-COVID-19 patients after discharge with follow-up chest CT findings. Egyptian J. Radiol. Nucl. Med. 2021;52(1):1–3. [Google Scholar]
- 30.Yu M., Liu Y., Xu D., Zhang R., Lan L., Xu H. Prediction of the development of pulmonary fibrosis using serial thin-section CT and clinical features in patients discharged after treatment for COVID-19 pneumonia. Korean J. Radiol. 2020;21(6):746–755. doi: 10.3348/kjr.2020.0215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zou J.N., Sun L., Wang B.R., Zou Y., Xu S., Ding Y.J., et al. The characteristics and evolution of pulmonary fibrosis in COVID-19 patients as assessed by AI-assisted chest HRCT. PLoS One. 2021;16(3):1–12. doi: 10.1371/journal.pone.0248957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Han X., Fan Y., Alwalid O., Li N., Jia X., Yuan M., et al. Six-month follow-up chest CT findings after severe COVID-19 pneumonia. Radiology. 2021;299(1):E177–E186. doi: 10.1148/radiol.2021203153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Liu M., Lv F., Huang Y., Xiao K. Follow-up study of the chest CT characteristics of COVID-19 survivors seven months after recovery. Front. Med. 2021;8(1):1–8. doi: 10.3389/fmed.2021.636298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Han X., Fan Y., Alwalid O., Zhang X., Jia X., Zheng Y., et al. Fibrotic interstitial lung abnormalities at 1-year follow-up CT after severe COVID-19. Radiology. 2021;301(3):E438–E440. doi: 10.1148/radiol.2021210972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Myall K.J., Mukherjee B., Castanheira A.M., Lam J.L., Benedetti G., Mak S.M., et al. Persistent post–COVID-19 interstitial lung disease. An observational study of corticosteroid treatment. Ann. Am. Thoracic Soc. 2021;18(5):799–806. doi: 10.1513/AnnalsATS.202008-1002OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang D., Zhang C., Li X., Zhao J., An C., Peng C., et al. Thin-section computed tomography findings and longitudinal variations of the residual pulmonary sequelae after discharge in patients with COVID-19: a short-term follow-up study. Eur. Radiol. 2021;31(1):7172–7183. doi: 10.1007/s00330-021-07799-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.