Abstract
Identifying Helicobacter pylori (H. pylori, Hp) infection in animals before and after artificial infection influences the subsequent experiment. We established effective and noninvasive detection methods, including the gastric fluid nested polymerase chain reaction (PCR) method and the 13C-urea breath test, which can detect Hp before modeling Hp infection in Mongolian gerbils. We designed a gas collection equipment for gerbils. Hp nested PCR was also performed on gastric fluid, gastric mucosa, duodenal contents, and faeces of gerbils challenged with Hp. Conventional Hp detection methods, including rapid urease assay and immunohistochemistry, were compared. Moreover, we assessed the natural infection of Hp in 135 gerbils that had never been exposed to Hp artificially from the major laboratory gerbil groups in China. In 10 Hp infected gerbils, the positive detection results were 100%, 100%, 90%, and 10% in gastric fluid, gastric mucosa, duodenal contents, and faeces with nested PCR, respectively. A rapid urease test performed on gastric mucosa showed that all animals were infected with Hp. Immunohistochemical detection and bacteria culture of gastric mucosa samples that were positive by the nested PCR method also confirmed the presence of Hp. 9% (3/35) and 6% (2/31) natural infection rates were found in conventional gerbil groups from the Capital Medical University and Zhejiang Laboratory Animal Center. In conclusion, we established two noninvasive Hp detection methods that can be performed before modelingHp infection, including the gastric fluid nested PCR method and the 13C-urea breath test.
1. Introduction
Helicobacter pylori (H. pylori, Hp) is a gram-negative bacterium that infects more than half of the world's people and is associated with a variety of severe gastric diseases, including gastritis, peptic ulcer, and even adenocarcinoma [1]. Further understanding of the specific mechanism of H. pylori-induced gastric cancer is needed.
Researchers have tried to replicate H. pylori infection model in animals to study the pathogenesis of H. pylori infection in vivo. The colonization efficiency is unsatisfactory in large animals such as monkeys, cats, and dogs [2]. In mice, H. pylori infection results in development of gastric adenocarcinoma with some genetically engineered mice or with mice that had been induced with chemical carcinogens in advance [3, 4]. H. pylori infection in rats only induces inflammation with preinjury of the gastric mucosa [5]. In 1998, Watanabe and Honda found that Mongolian gerbils can develop gastric adenocarcinoma after long-term H. pylori infection without additional chemical carcinogens [6, 7]. Mongolian gerbils are currently recognized as an efficient, cost-effective, and robust rodent model of H. pylori infection.
The gerbil model is often used to study the inflammatory response, pathogenic mechanism and therapeutic strategies of H. pylori infection. The cancer progression associated with H. pylori in gerbils is characterized by erosion, inflammation with neutrophil infiltration, chronic superficial gastritis, atrophic gastritis, intestinal metaplasia, and finally dysplasia and adenocarcinoma, which are similar to those of humans [8, 9]. Nevertheless, in many studies, H. pylori-free gerbils were not used or gerbils were not evaluated for natural infection before infection with H. pylori. It impacts the reliability and consistency of previous studies [10, 11].
A greater variety of H. pylori detection methods have been applied to detect H. pylori in human and experimental animals [12]. Bacterial culture was considered the most specific method for the detection of H. pylori [12]. However, animals need to be sacrificed first. Immunohistochemistry also requires the animals to be sacrificed. Serum antibody detection and real-time PCR have been widely used clinically [13]. Nested PCR is often performed in gastric mucosa, gastric contents and duodenum to detect H. pylori infection, whereas gastric tissue and duodenum can only be obtained after the animals were euthanized. Hence, we developed a gastric fluid nested PCR method that can perform noninvasive Hp detection on live gerbils.
13C-urea breath test is one of the most reliable noninvasive methods for detecting H. pylori infection in humans. H. pylori produces urease enzyme and then hydrolyses urea to release CO2 and NH3. 13C-urea breath test can detect the urease production and has been used for the clinical detection of bacteria in antral biopsies [14, 15]. Therefore, we designed a 13C-urea breath test equipment for gerbils that are suitable for the size of animals and the volume of expired gas. The 13C-urea breath test can detect Hp before modelling Hp infection in Mongolian gerbils.
In this study, we established two noninvasive H. pylori detection methods including the gastric fluid nested PCR method and the 13C-urea breath test. We compared these two new methods with conventional Hp detection methods and assessed the natural infection of H. pylori in gerbils from the major laboratory gerbil groups in China.
2. Materials and Methods
2.1. Bacterial Strains
H. pylori ATCC 43504, Staphylococcus aureus ATCC 25923, Pseudomonas aeruginosa ATCC 27853, Escherichia coli ATCC 25922, Klebsiella pneumonia CMCC 46114, Proteus mirabilis CMCC 49005, and Campylobacter jejuni subsp. ATCC 700819 were provided by the National Institutes for Food and Drug Control. H. pylori SS1 was provided by the Capital Medical University. H. bilis ATCC51630 was provided by Guangdong Laboratory Animals Monitoring Institute.
2.2. Culture of H. pylori from the Gastric Mucosa of Mongolian Gerbils
For bacterial culture, the gastric tissue was grounded in Brucella broth (Difco, Detroit, MI, USA) and plated onto Columbia agar (Difco) supplemented with 5% sheep blood and Dent antibiotic supplement (Oxoid, Basingstoke, UK). Incubating the plates at 37°C in a microaerophilic condition for 72 h. Organisms were identified as H. pylori by modified Gram staining, oxidase, catalase, and urease reactions and kept frozen at -80°C.
2.3. Animals
A total of 69 Mongolian gerbils (6-8 weeks old and 50-60 g) comprising 59 conventional gerbils and 10 specific pathogen-free males were included to establish the H. pylori detection methods. The H. pylori infection model was replicated in an additional 94 conventional gerbils (6-8 weeks old and 50-60 g). All rodents were from Capital Medical University and fed at a Level II Biosafety laboratory in the Chinese Center for Disease Control and Prevention. Microbiological status of the conventional gerbils and specific pathogen-free gerbils were strictly controlled according to local standards of Beijing. In order to establish H. pylori-infected gerbil model, we used 2 × 109 CFU/mLH. pylori ATCC 43504 to gavage gerbils for 5 times, 0.5 mL each time. Oral gavage was performed at an interval of 48 h, and the gerbils were fasted for 12 h prior to challenge. The infection method is based on previous reports [6, 9].
In China, the main laboratory gerbils are separately located at the Capital Medical University (CMU), Zhejiang Laboratory Animal Centre (ZJLAC), and Dalian Medical University (DMU) [16, 17]. All colonies originated from Inner Mongolia in China. The screening groups were from CMU (35 conventional gerbils and 23 clean gerbils), ZJCLA (31 conventional gerbils and 26 clean gerbils), and DMU (20 conventional gerbils) [18, 19].
All gerbils are housed in individual ventilated cages (IVC; 461 × 274 × 229 mm; Tecniplast, Milan, Italy) on a 12 h light/dark cycle, with 2 to 3 gerbils in each cage. Females and males were separated and housed separately. The room temperature was maintained at 22~24°C, and the humidity was maintained at 60~70%. The animal experiments were conducted in accordance with the Guidelines of the CMU Animal Experiments and Experimental Animals Management Committee under a protocol approved by the Animal Experiments and Experimental Animal Welfare Committee of CMU (Permit number: AEEI-2016-152).
2.4. Sample Extraction and Bacterial Genome
To collect gastric fluid, gerbils were gavaged with 0.5 mL distilled water after fasting for 12 h. A gavage needle was inserted into the stomach, and gastric fluid was extracted into a centrifuge tube within 1 min. The gastric fluid was centrifuged, and the genome was extracted using a microsample genomic DNA extraction kit (Tiangen, Beijing, China). DNA concentration and quality were verified by Nanodrop 2000c (Thermo, USA).
After 10 weeks of infection, the animals were anesthetized with isoflurane (induced with 3%, maintained with 2%, in 30% O2/70% N2O) and then euthanized. Samples of gastric tissue, gastric contents, duodenal contents, and colonic faeces (0.4 g/each) were collected into 1.5 mL sterile centrifuge tubes, respectively. Genomic DNA was extracted by a microsample genomic DNA extraction kit containing centrifugal adsorption columns (Tiangen, Beijing, China).
Bacterial DNA was extracted using a bacterial genomic DNA extraction kit (Tiangen, Beijing, China).
2.5. Nested PCR Primer Design, Amplification, and DNA Sequencing
According to previous reports, we compared different H. pylori strains, and the conserved region of H. pylori (GenBank ID: NC-000915.1) genome Urea gene was selected.
Primers were designed by the primer design software Primer Premier 5. The primers for the first PCR were F1: 5′-AGTAGGGCCATACATAGAAA-3′ and R1: 5′-GACAAAACTCGTAACCGT-3′. Each reaction was performed in a 20 μL reaction volume containing 10 μL Dream Taq Green PCR Master Mix (Thermo Fisher Scientific, Massachusetts, MA), 10 pmol each primer, and 50 ng of the extracted template. The PCR protocol was as follows: 95°C for 5 min, followed by 35 cycles of 95°C for 30 s, 51.3°C for 30 s, and 72°C for 30 s, and a final extension at 72°C for 5 min. The expected PCR product was 499 bp.
The primers for the second PCR were F2: 5′-CATAGTTGTCATCGCTTTT-3′ and R2: 5′-GCGTTGGTTGATAGGC-3′. Each reaction was performed in a 20 μL reaction volume containing 10 μL Dream Taq Green PCR Master Mix (Thermo Fisher Scientific, Massachusetts, MA), 10 pmol of each primer, and 50 ng of the extracted template. The PCR was performed at 95°C for 5 min, followed by 35 cycles of 95°C for 30 s, 51.3°C for 30 s, and 72°C for 30 s, and a final extension at 72°C for 5 min. The expected PCR product was 100 bp.
After matching primers 1 and primers 2 of nested PCR with Helicobacter species (taxid: 209) on the GenBank database, we confirmed that the matching sequences only exist in H. pylori.
The nested PCR amplification product was electrophoresed in 2% agarose gel (Amresco, USA); Takara 50 bp DNA Ladder was used as a molecular mass marker.
DNA sequencing was completed by Beijing Tianyihuiyuan Life Science & Technology Inc. Alignments were performed between nested PCR production sequences and sequences in GenBank.
H. pylori ATCC 43504 with a concentration of 2 × 108 ~ 2 × 101 CFU/mL, Staphylococcus aureus ATCC 25923, Pseudomonas aeruginosa ATCC 27853, Escherichia coli ATCC 25922, Klebsiella pneumonia CMCC 46114, Proteus mirabilis CMCC 49005, Campylobacter jejuni subsp. ATCC 700819, and H. pylori SS1 were used as templates for nested PCR according to the above conditions, respectively.
2.6. Establishment of the 13C-Urea Breath Test Detection Threshold
Gerbils were challenged with H. pylori by oral gavage at concentrations of 2 × 109 CFU/mL, 2 × 107 CFU/mL, 2 × 105 CFU/mL, 2 × 103 CFU/mL, and 2 × 101 CFU/mL and sterile saline. Each concentration treatment group contained three animals. Gerbils were placed immediately into a holder tube. The holder tube was connected to a gas propulsion device and a gas collecting device. The gas exhaled by a gerbil for 10 min was collected as the background gas with a ventilation of 0.5 mL/s. The gerbil was removed and was gavaged with 0.8 mg/mL urease solution (Haidewei, Shenzhen, China), and the gerbil was placed back into the holder tube immediately. Sample gas was collected in the same way. Finally, the 13C tester (Haidewei, Shenzhen, China) was inserted into the gas bag, and the delta-over-baseline (DOB) value was measured.
2.7. Other H. pylori Detection Methods
A rapid urease detection kit (SanQiang, China) was performed on gerbil gastric tissue.
Immunohistochemistry and Warthin-Starry silver staining were also performed with gastric tissue to detect H. pylori. The gastric tissue was embedded in paraffin and cut into 4 μm-thick sections. Pyloric gastric sections were immunostained for H. pylori with primary antibodies (1 : 30 dilution, Batch B-0471, DAKO, Glostrup, Denmark). The Warthin-Starry silver staining kit was purchased from Solarbio (Beijing, China).
2.8. Statistical Analysis
ROC (receiver-operating characteristic) curve analysis was used to determine the cut-off value of the detection threshold of the 13C-urea breath test. The sensitivity, accuracy, and specificity of the 13C-urea breath test methods and nested PCR were compared with several conventional detection methods. All analyses were performed using Graph Pad Prism 8.
3. Results
3.1. Sensitivity and Specificity Assay of Nested PCR
The results showed that target fragments could be amplified from H. pylori at a concentration of 2 × 102 ~ 108 CFU/mL (Figure 1(a)). The lowest concentration of H. pylori that can be detected is 2 × 102 CFU/mL and the total PCR reaction volume is 20 μL, containing 4 CFU of bacteria. The expected band can only be observed in H. pylori but not in other bacteria (Figures 1(b) and 1(c)). In order to exclude the influence of other bacteria including Helicobacter species, we compared the target fragments amplified by nested PCR. The GenBank database was used to perform BLAST analysis on the sequencing results to confirm that the nested PCR amplified fragments were specific fragments of H. pylori. Taken together, nested PCR detection method for H. pylori has high sensitivity and specificity.
Figure 1.
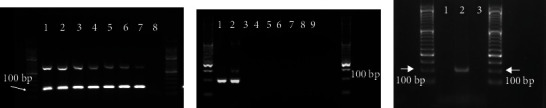
Sensitivity and specificity assays of nested PCR. (a) The nested PCR electrophoresis map of different concentrations of H. pylori. The target band was 100 bp. The concentrations of H. pylori in wells 1~8 were 2 × 108 CFU/mL, 2 × 107 CFU/mL, 2 × 106 CFU/mL, 2 × 105 CFU/mL, 2 × 104 CFU/mL, 2 × 103 CFU/mL, 2 × 102 CFU/mL, and 2 × 101 CFU/mL. (b) The nested PCR electrophoresis map of different bacteria. The bacterial samples in wells 1-9 were H. pylori ATCC 43504, H. pylori SS1, Staphylococcus aureus ATCC 25923, Pseudomonas aeruginosa ATCC 27853, Escherichia coli ATCC 25922, Klebsiella pneumonia CMCC 46114, Proteus mirabilis CMCC 49005, Campylobacter jejuni subsp. ATCC 700819, and nucleotide-free water. (c) The nested PCR electrophoresis map of different bacteria. The bacterial samples in wells 1-3 were nucleotide-free water, H. pylori ATCC 43504, and H. bilis ATCC51630.
3.2. Detection of H. pylori Infection by Nested PCR
After 10 weeks of infection with H. pylori, the gastric fluid of 10 pathogen-free gerbils was extracted. Gastric tissue, duodenal content, and faecal samples were collected after euthanizing the gerbils. DNA extraction, PCR amplification, and gel electrophoresis were performed on all the samples.
The expected band of the first PCR amplification was 499 bp, and 30% (3/10) of the gastric fluid samples were positive (Figure 2(a)) The expected band of the second PCR amplification was 100 bp. After agarose gel electrophoresis analysis, 100% (10/10), 100% (10/10), 90% (9/10), and 10% (1/10) of the gastric fluid, gastric tissue, duodenal content, and faecal samples were positive for the 100 bp band (Figures 2(b), 2(c), 2(d) and 2(e)).
Figure 2.
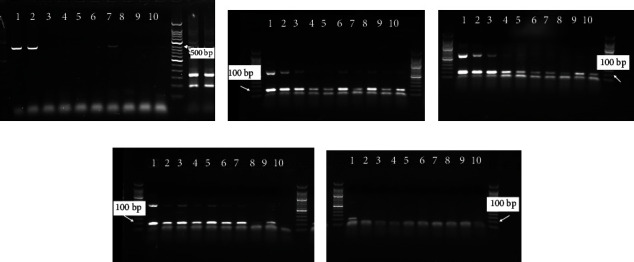
The detection of H. pylori in Mongolian gerbils by different PCR methods. (a) Electrophoresis of PCR products from gastric fluid samples; (b) electrophoresis of nested PCR products from gastric fluid samples; (c) electrophoresis of nested PCR products from gastric mucosa samples; (d) electrophoresis of nested PCR products from duodenum content samples; and (e) electrophoresis of nested PCR products from colonic stool samples. 1-10 are the numbers of 10 different gerbils.
3.3. Results from the Mongolian Gerbil Screening Groups
In order to investigate the natural infection of H. pylori in Chinese laboratory gerbils, we performed nested PCR detection of H. pylori in gastric fluid and gastric tissue of the main Chinese laboratory gerbil populations. There were three conventional gerbils infected with H. pylori at CMU, while the positive rate was 9% (2/35); two conventional gerbils were infected with H. pylori at ZJCLA with a 6% (1/31) positive rate. However, the clean gerbils at CMU and ZJCLA and the conventional gerbils at DMU were not infected with H. pylori as determined by evaluation of gastric fluid samples with the nested PCR detection method (Table 1). These observations suggested the presence of natural infection of H. pylori among laboratory gerbils in China, although the positive rate was low.
Table 1.
The H. pylori infection results in different Mongolian gerbil groups.
| Name of group | Capital Medical University conventional animal | Capital Medical University clean animal | Dalian Medical University conventional animal | Zhejiang Provincial Experimental Animal Center conventional animal | Zhejiang Provincial Experimental Animal Center clean animal |
|---|---|---|---|---|---|
| Number of positive samples from gastric fluid nested PCR | 1 | 0 | 0 | 1 | 0 |
| Number of positive samples from gastric tissue nested PCR | 2 | 0 | 0 | 1 | 0 |
| Total number | 35 | 23 | 20 | 31 | 26 |
| Infection rate | 9% | 0% | 0% | 6% | 0% |
3.4. Establishment of the Mongolian Gerbil 13C-Urea Breath Test and Detection Threshold
H. pylori can produce abundant urease enzymes that hydrolyse urea to release CO2 and NH3. The hydrolysed urea will form 13CO2 after taking 13C-urea capsules orally and enter the lungs with blood and be discharged by gas. Based on this principle, we designed a 13C-urea breath test gas collection device (Figure 3(a)) for gerbils that consists of a gerbil holder tube, a gas propulsion device and a gas collecting device. We can determine whether the gerbils are infected with H. pylori by detecting the 13C in the exhaled gas before and after taking the 13C-urea solution. We then studied the relationship between the DOB value and the dose of 13C-urea, H. pylori concentration and time. The DOB value increased with increasing doses of 13C-urea (Figure 3(b)). With the increase in detection time after gavage, the DOB value reached a peak and then gradually decreased (Figure 3(c)). The DOB value increased with increasing H. pylori concentration (Figure 3(d)). By detecting the change in DOB with different amounts of H. pylori (Figure 3(e)), we determined that by collecting the exhaled gas of gerbils within 10 min after a gavage of 0.5 mg/mL 13C-urea solution, the gerbils were considered to be H. pylori-positive if DOB > 7.
Figure 3.
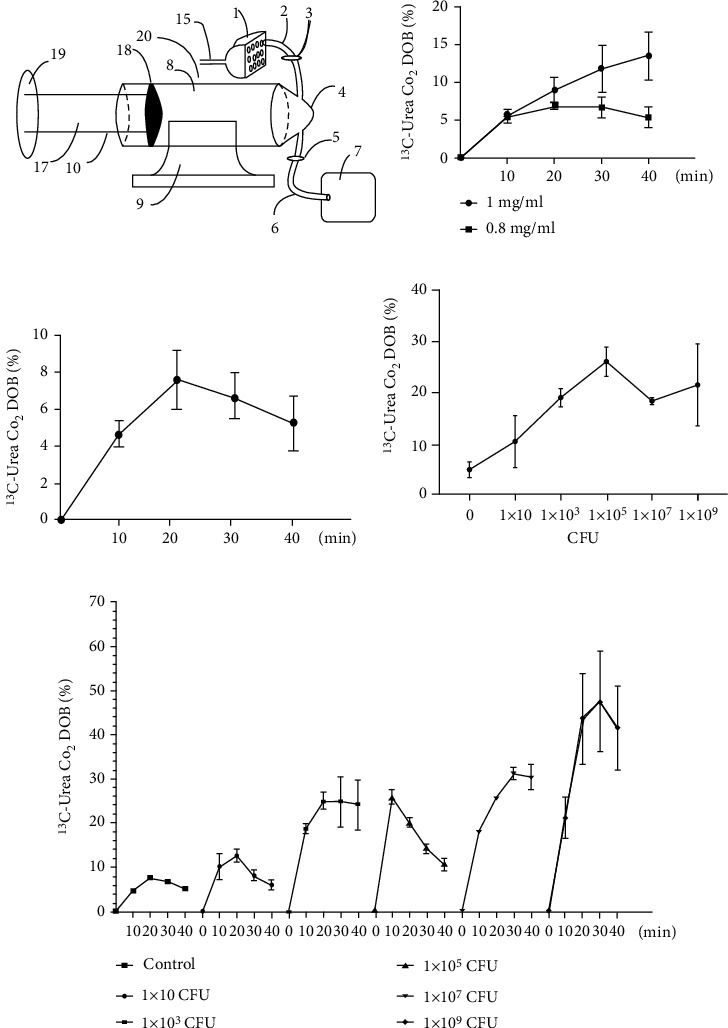
The detection threshold of gastric fluid nested PCR ((b–d) n = 3; (c) n = 5). (a) Mongolian gerbil gas collection; (b) the relationship between CO2 DOB value and 13C-urea concentrations; (c) the relationship between CO2 DOB value and test time; (d) the relationship between CO2 DOB value and H. pylori concentrations; and (e) the relationship between different H. pylori concentrations in Mongolian gerbils and test time with the 13C-urea breath test.
3.5. Detection of H. pylori-Infected Gerbils
We established an H. pylori-infected Mongolian gerbil model in which the presence of H. pylori was evaluated every 5 weeks after infection for 85 weeks (Table 2). The 13C-urea breath test showed that 50% (47/94) of gerbils were infected with H. pylori, while the positive rate of detection in the gastric mucosa by nested PCR was 95.2% (89/94). Nested PCR showed a 100% positive rate of H. pylori infection after 15 weeks. 47 animals that were considered positive by the 13C-urea breath test also tested positive by nested PCR. The detection rate of nested PCR in gastric tissue was higher than that of the 13C detection method.
Table 2.
The H. pylori infection results in different Mongolian gerbil groups every 5 weeks after infection for 85 weeks.
| Week after infection | Total number of samples | Number of positive samples via 13C detection methods | Number of positive gastric tissue samples via nested PCR |
|---|---|---|---|
| 5 | 6 | 2 | 2 |
| 10 | 6 | 2 | 5 |
| 15 | 6 | 3 | 6 |
| 20 | 5 | 0 | 5 |
| 25 | 6 | 4 | 6 |
| 30 | 6 | 0 | 6 |
| 35 | 6 | 6 | 6 |
| 40 | 5 | 2 | 5 |
| 45 | 5 | 5 | 5 |
| 50 | 4 | 3 | 4 |
| 55 | 5 | 1 | 5 |
| 60 | 6 | 0 | 6 |
| 65 | 6 | 4 | 6 |
| 70 | 6 | 1 | 6 |
| 75 | 6 | 5 | 6 |
| 80 | 5 | 5 | 5 |
| 85 | 5 | 4 | 5 |
| Total | 94 | 47 | 89 |
| % | 100.00% | 50.00% | 95.20% |
3.6. Other H. pylori Detection Methods
To verify the accuracy of the nested PCR method, immunohistochemistry and rapid urease test were used for comparison in the present study. The rapid urease test showed that the detection rate of 10 animals infected with H. pylori was 100% (Table 3). We observed H. pylori in gerbils that were H. pylori-positive by nested PCR and the 13C-urea breath test at the same time through immunohistochemistry (Figure 4). H. pylori was cultured and identified from the gastric mucosa of 5 gerbils that were determined to be H. pylori-positive by nested PCR performed on gastric fluid and gastric tissue (Figure 5). Bacterial culture proves the presence of H. pylori.
Table 3.
Comparison of the results of different methods for the detection of H. pylori in Mongolian gerbils.
| No. | Gastric fluid PCR | Gastric fluid nested PCR | Gastric mucosa nested PCR | Duodenum content nested PCR | Faeces nested PCR | Rapid urease assay |
|---|---|---|---|---|---|---|
| 1 | + | + | + | + | + | + |
| 2 | + | + | + | + | - | + |
| 3 | - | + | + | + | - | + |
| 4 | - | + | + | + | - | + |
| 5 | - | + | + | + | - | + |
| 6 | - | + | + | + | - | + |
| 7 | + | + | + | + | - | + |
| 8 | - | + | + | + | - | + |
| 9 | - | + | + | + | - | + |
| 10 | - | + | + | - | - | + |
| Positive | 30% | 100% | 100% | 90% | 10% | 100% |
Notes: “+” means positive; “-” means negative.
Figure 4.
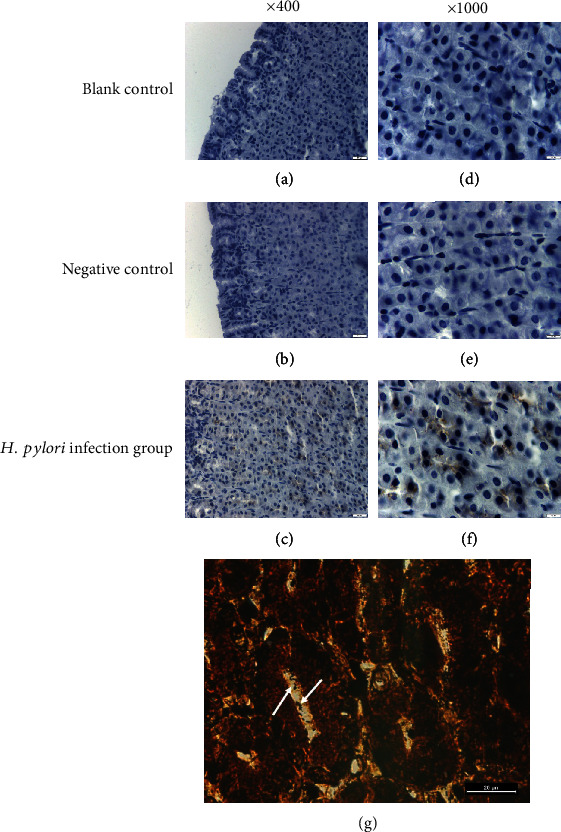
Immunohistogram of H. pylori in gastric mucosa. (a) Blank control, ×400; (b) negative control, ×400; (c) experimental group, ×400; (d) blank control, ×1000; (e) negative control, ×1000; and (f) experimental group, ×1000. (g) Warthin-Starry silver staining on gastric mucosa of H. pylori infected gerbil, ×1000.
Figure 5.
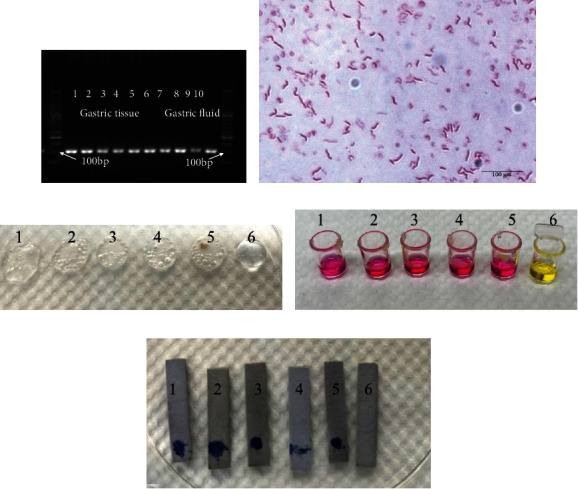
(a). Electrophoresis of nested PCR products from gastric tissue and gastric fluid samples of 5 H. pylori-infected gerbils. 1–5 are the nested PCR products of gastric tissue, 6-10 are the nested PCR products of gastric fluid, respectively. (b) Gram staining of bacteria cultured from H. pylori-infected gerbils' gastric mucosa, ×1000. (c–e) Oxidase, catalase, and urease reactions of bacteria cultured from H. pylori-infected gerbils' gastric mucosa. 1–5 is the number of gerbils. 6 is the negative control. (c) Oxidase reaction. (d) Catalase reaction. (e) Urease reaction.
4. Discussion
Though the transmission route of H. pylori infection remains unclear, it is generally believed that the transmission of H. pylori person-to-person mainly occurs via the oral-oral or faecal-oral route [20]. Mongolian gerbils are a good animal model for studying H. pylori infection and transmission because of the tumor progression associated with H. pylori in gerbils is similar to that of humans [9, 21]. Besides, gerbils are often kept as pets for children who are susceptible to H. pylori [22].
In Charles River Laboratories International, Inc. (Wilmington, USA)'s latest Gerbil VAF Report, 100.0% (72/72) of gerbils were positive for Helicobacter species through PCR detection. Our general survey of the major Mongolian gerbil population in China also showed the natural infection of H. pylori through the gastric fluid nested PCR detection method. The positive rate was highest (9%) in the conventional gerbils at the Capital Medical University. In rodents, direct transmission of H. pylori occurs from challenged mice to unchallenged mice via saliva and faeces in a single cage [23]. Studies have found that H. pylori infection in Mongolian gerbil pups are transmitted by the faecal-oral route from an infected mother [21]. Mongolian gerbils are susceptible to H. pylori as previously reported [6]. We also found that the rate of colonization in the Mongolian gerbils infected with H. pylori was 100% after 10 weeks. Furthermore, Mongolian gerbils have been kept as pets for children, and the transmission of H. pylori between humans and pets has been reported [24]. It is necessary to determine the prevalence of H. pylori in pet gerbils through noninvasive detection methods. In addition to being used in the investigation of H. pylori, Mongolian gerbils are also considered to be a good animal model for studying hearing loss and brain ischemia. In these studies, unexpected results may be produced since H. pylori infection leads to increased expression of IL12 and IFNγ in Mongolian gerbils, which seriously affects the accuracy of experimental results [25–27]. Besides, H. pylori infection may induce extragastric diseases, making it difficult to determine the health status of experimental gerbils [28]. Though H. pylori detection before replicating the H. pylori-infected model in gerbils is necessary, we found few studies that used H. pylori-free gerbils or detected the natural infection before challenge with H. pylori [10, 11]. However, we cannot confirm whether the animal had been infected after modelling for a long time unless the animal was sacrificed, which affected the efficiency of modelling and did not conform to the “3R principle” (reduce, refine, and replace). Therefore, there is an urgent need for the detection of H. pylori infection before modelling through a more accurate and noninvasive detection method and for long-term detection [29].
Hence, we developed a method of gastric fluid nested PCR that only needs to extract the gastric fluid of live gerbils through gavage needle without harming the animals [12]. The positive rate of detection from gastric fluid with nested PCR was consistent with the results of the rapid urease detection method, indicating that the gastric fluid nested PCR method had convincing accuracy.
Chronic H. pylori infection can reduce the secretion of gastric acid and allow the growth of the gastric bacterial community [30]. The extracted gastric fluid may contain other bacteria besides H. pylori. We amplified several common bacterial DNAs including H. bilis with nested PCR primers, and no target fragments were detected [31]. After using GenBank database to match primer 1 and primer 2 of nested PCR to Helicobacter species (taxid:209), we found that the matching sequence of both primer 1 and primer 2 only existed in H. pylori. Thus, we confirmed that the nested PCR method is specific to H. pylori. The gastric fluid nested PCR method has high specificity. Considering the primers are a target to bacteria themselves, nested PCR could theoretically be applied in rodents which needs further investigation.
Nested PCR amplification was then performed with genomes from different concentrations of H. pylori. Target fragments could be amplified successfully from H. pylori at a concentration of 2 × 102 ~ 2 × 108 CFU/mL, that is, the copies of the bacterial genome are 106~1, indicating that the nested PCR method has a high sensitivity. In Figure 1(a), agarose gel electrophoresis was performed for the second PCR product of nested PCR. Two bands of PCR products were shown, the lower band was the target band of the second PCR, with a size of 100 bp, and the upper band was the unreacted DNA template, with a size of 499 bp from the first PCR.
The positive rate of H. pylori infection determined by nested PCR was different in different parts of the alimentary canal. During the gavage process, H. pylori entered the junction of the duodenum and stomach with water or food, which caused a high positive rate of detection in the duodenal contents. The positive rate of H. pylori in faeces was low which may be due to the PCR inhibitors contained in the faecal material. Bacterial culture was considered the most specific method for the detection of H. pylori; therefore, we performed bacterial culture from gastric mucosa of 5 gerbils. Nested PCR of gastric tissue and gastric fluid has determined that all the gerbils are H. pylori positive. Gram staining, oxidase, catalase, and urease reactions confirmed the presence of H. pylori. These results demonstrate the reliability of the nested PCR method.
13C-urea breath test has been applied in clinic and in many experimental animals except for gerbils, such as H. pylori infected mice model and barrier born pigs [32, 33]. Although the detection standards are different due to the differences in size of animals and in volume of expired gas, the 13C-urea breath test is harmless and reliable in these animals. Hence, we designed a gas collection equipment suitable for gerbils and defined the detection method and threshold of gerbil 13C-urea breath test.
In our research, when gerbils were examined every 5 weeks after infection for 85 weeks, the 13C-urea breath test showed that 50% of gerbils were infected with H. pylori, while the positive rate of gastric mucosa by nested PCR was 95.2%. The accuracy of the 13C-urea breath test is unsatisfactory because the urease test basically depends on bacterial density [34]. It is difficult to detect the presence of H. pylori by 13C-urea breath test at a very low density. Meanwhile, in our study, the air in the holder tube of gerbil gas collection equipment diluted the 13CO2 concentration. As there is no special 13C-urea breath equipment for rodents, we have no choice but to select 13CO2 detector for humans. We aim to improve the detection method and establish a better detection threshold in the future.
5. Conclusions
In conclusion, there is a strong need to detect H. pylori with a noninvasive method in gerbils before replicating the model of H. pylori infection because of the considerable natural infection rate and acute effects in animal colonies and individuals after infection. It can improve the validity, reliability, and consistency of subsequent experiments and lay the foundation for research on the pathogenic mechanism and a therapeutic strategy for gastric disease caused by H. pylori.
Acknowledgments
We do appreciate Pengju Guo from the Guangdong Laboratory Animals Monitoring Institute for providing DNA of H. bilis ATCC51630. This work was supported by National Science Foundation of China (32070537, 31772545, 31672375, and 31572348) and Beijing Natural Science Foundation Program and Scientific Research Key Program of Beijing Municipal Commission of Education (KZ20210025037). Thanks to these funds for their support.
Contributor Information
Zhenwen Chen, Email: czwen@ccmu.edu.cn.
Changlong Li, Email: licl@ccmu.edu.cn.
Data Availability
All data, models, and code generated or used during the study appear in the submitted article.
Conflicts of Interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Authors' Contributions
Xiulin Zhang, Cunlong Wang, and Yang He contributed equally to this work.
References
- 1.Bauer B., Meyer T. F. The human gastric pathogen Helicobacter pylori and its association with gastric cancer and ulcer disease. Ulcers . 2011;2011:23. doi: 10.1155/2011/340157. [DOI] [Google Scholar]
- 2.Fox J. G., Batchelder M., Marini R., et al. Helicobacter pylori-induced gastritis in the domestic cat. Infection and Immunity . 1995;63(7):2674–2681. doi: 10.1128/iai.63.7.2674-2681.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Aziz F., Xin M., Gao Y., et al. Induction and prevention of gastric cancer with combined Helicobacter pylori and capsaicin administration and DFMO treatment, respectively. Cancers (Basel). . 2020;12(4):p. 816. doi: 10.3390/cancers12040816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Miao Z. F., Sun J. X., Adkins-Threats M., et al. DDIT4 licenses only healthy cells to proliferate during injury-induced metaplasia. Gastroenterology . 2021;160(1):260–271.e10. doi: 10.1053/j.gastro.2020.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ross J. S., Bui H. X., del Rosario A., Sonbati H., George M., Lee C. Y. Helicobacter pylori. Its role in the pathogenesis of peptic ulcer disease in a new animal model. The American Journal of Pathology . 1992;141(3):721–727. [PMC free article] [PubMed] [Google Scholar]
- 6.Honda S., Fujioka T., Tokieda M., Satoh R., Nishizono A., Nasu M. Development of Helicobacter pylori-induced gastric carcinoma in Mongolian gerbils. Cancer Research . 1998;58(19):4255–4259. [PubMed] [Google Scholar]
- 7.Watanabe T., Tada M., Nagai H., Sasaki S., Nakao M. Helicobacter pylori infection induces gastric cancer in Mongolian gerbils. Gastroenterology . 1998;115(3):642–648. doi: 10.1016/S0016-5085(98)70143-X. [DOI] [PubMed] [Google Scholar]
- 8.Sawada Y., Yamamoto N., Sakagami T., et al. Comparison of pathologic changes in Helicobacter pylori-infected Mongolian gerbils and humans. Journal of Gastroenterology . 1999;34(Suppl 11):55–60. [PubMed] [Google Scholar]
- 9.Noto J. M., Romero-Gallo J., Piazuelo M. B., Peek R. M. The Mongolian gerbil: a robust model of Helicobacter pylori-induced gastric inflammation and cancer. Methods in Molecular Biology . 2016;1422:263–280. doi: 10.1007/978-1-4939-3603-8_24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Noto J. M., Gaddy J. A., Lee J. Y., et al. Iron deficiency accelerates Helicobacter pylori-induced carcinogenesis in rodents and humans. The Journal of Clinical Investigation . 2013;123(1):479–492. doi: 10.1172/JCI64373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kuo C. H., Lu C. Y., Yang Y. C., et al. Does long-term use of silver nanoparticles have persistent inhibitory effect on H. pylori based on Mongolian gerbil's model? BioMed Research International . 2014;2014:7. doi: 10.1155/2014/461034.461034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Patel S. K., Pratap C. B., Jain A. K., Gulati A. K., Nath G. Diagnosis of Helicobacter pylori: what should be the gold standard? World Journal of Gastroenterology . 2014;20(36):12847–12859. doi: 10.3748/wjg.v20.i36.12847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bénéjat L., Ducournau A., Lehours P., Mégraud F. Real-time PCR for Helicobacter pylori diagnosis. The best tools available. Helicobacter . 2018;23(5, article e12512) doi: 10.1111/hel.12512. [DOI] [PubMed] [Google Scholar]
- 14.Savarino V., Vigneri S., Celle G. The 13C urea breath test in the diagnosis of Helicobacter pylori infection. Gut . 1999;45(Supplement 1):I18–I22. doi: 10.1136/gut.45.2008.i18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Graham D. Y., Malaty H. M., Cole R. A., Martin R. F., Klein P. D. Simplified 13C-urea breath test for detection of Helicobacter pylori infection. The American Journal of Gastroenterology . 2001;96(6):1741–1745. doi: 10.1111/j.1572-0241.2001.03867.x. [DOI] [PubMed] [Google Scholar]
- 16.Du X. Y., Li W., Sa X. Y., et al. Selection of an effective microsatellite marker system for genetic control and analysis of gerbil populations in China. Genetics and Molecular Research . 2015;14(3):11030–11042. doi: 10.4238/2015.September.21.16. [DOI] [PubMed] [Google Scholar]
- 17.Wang Y., Zhao P., Song Z., et al. Generation of gene-knockout Mongolian gerbils via CRISPR/Cas9 system. Frontiers in Bioengineering and Biotechnology . 2020;8, article 780 doi: 10.3389/fbioe.2020.00780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Du X., Wang D., Li Y., et al. Newly breeding an inbred strain of ischemia-prone Mongolian gerbils and its reproduction and genetic characteristics. Experimental Animals . 2018;67(1):83–90. doi: 10.1538/expanim.17-0071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ding X. M., Qian B. Z., Matsuda J., Koura M., Sa X. Y., Shi Z. K. Genetic diversity of Mongolian gerbils (Meriones unguiculatus) Yi Chuan= Hereditas . 2008;30(7):877–884. doi: 10.3724/SP.J.1005.2008.00877. [DOI] [PubMed] [Google Scholar]
- 20.Goh K. L., Chan W. K., Shiota S., Yamaoka Y. Epidemiology of Helicobacter pylori infection and public health implications. Helicobacter . 2011;16(Suppl 1):1–9. doi: 10.1111/j.1523-5378.2011.00874.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oshio I., Osaki T., Hanawa T., et al. Vertical Helicobacter pylori transmission from Mongolian gerbil mothers to pups. Journal of Medical Microbiology . 2009;58(5):656–662. doi: 10.1099/jmm.0.008185-0. [DOI] [PubMed] [Google Scholar]
- 22.Rowland M., Daly L., Vaughan M., Higgins A., Bourke B., Drumm B. Age-specific incidence of Helicobacter pylori. Gastroenterology . 2006;130(1):65–72. doi: 10.1053/j.gastro.2005.11.004. [DOI] [PubMed] [Google Scholar]
- 23.Karita M., Matsumoto S., Kamei T., Shinohara K., Sugiyama T. Direct transmission of H. pylori from challenged to nonchallenged mice in a single cage. Digestive Diseases and Sciences . 2005;50(6):1092–1096. doi: 10.1007/s10620-005-2710-x. [DOI] [PubMed] [Google Scholar]
- 24.Kubota-Aizawa S., Matsubara Y., Kanemoto H., et al. Transmission of Helicobacter pylori between a human and two dogs: a case report. Helicobacter . 2021;26(3, article e12798) doi: 10.1111/hel.12798. [DOI] [PubMed] [Google Scholar]
- 25.Devi S., Ansari S. A., Vadivelu J., Mégraud F., Tenguria S., Ahmed N. Helicobacter pylori antigen HP0986 (TieA) interacts with cultured gastric epithelial cells and induces IL8 secretion via NF-κB mediated pathway. Helicobacter . 2014;19(1):26–36. doi: 10.1111/hel.12100. [DOI] [PubMed] [Google Scholar]
- 26.Heeringa A. N., Zhang L., Ashida G., Beutelmann R., Steenken F., Köppl C. Temporal coding of single auditory nerve fibers is not degraded in aging gerbils. The Journal of Neuroscience . 2020;40(2):343–354. doi: 10.1523/JNEUROSCI.2784-18.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kim D. H., Kim D. W., Jung B. H., et al. Ginsenoside Rb2 suppresses the glutamate-mediated oxidative stress and neuronal cell death in HT22 cells. Journal of Ginseng Research . 2019;43(2):326–334. doi: 10.1016/j.jgr.2018.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Suzuki H., Ataka K., Asakawa A., et al. _Helicobacter pylori_ vacuolating cytotoxin A causes anorexia and anxiety via hypothalamic urocortin 1 in mice. Scientific Reports . 2019;9(1):p. 6011. doi: 10.1038/s41598-019-42163-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Whary M. T., Fox J. G. Detection, eradication, and research implications of Helicobacter infections in laboratory rodents. Lab Animal . 2006;35(7):25–36. doi: 10.1038/laban0706-25. [DOI] [PubMed] [Google Scholar]
- 30.Ferreira R. M., Pereira-Marques J., Pinto-Ribeiro I., et al. Gastric microbial community profiling reveals a dysbiotic cancer-associated microbiota. Gut . 2018;67(2):226–236. doi: 10.1136/gutjnl-2017-314205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mishra S., Singh V., Rao G. R., Dixit V. K., Gulati A. K., Nath G. Prevalence of Helicobacter pylori in asymptomatic subjects--a nested PCR based study. Infection, Genetics and Evolution . 2008;8(6):815–819. doi: 10.1016/j.meegid.2008.08.001. [DOI] [PubMed] [Google Scholar]
- 32.Santos A. M., Lopes T., Oleastro M., et al. Role of 13C-urea breath test in experimental model of Helicobacter pylori infection in mice. Helicobacter . 2011;16(4):320–326. doi: 10.1111/j.1523-5378.2011.00847.x. [DOI] [PubMed] [Google Scholar]
- 33.Meyer-Rosberg K., Gustavsson S. 13C-urea breath test for diagnosis of experimental Helicobacter pylori infection in barrier born pigs. Gut . 1993;34(5):594–598. doi: 10.1136/gut.34.5.594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mégraud F. Advantages and disadvantages of current diagnostic tests for the detection of Helicobacter pylori. Scandinavian Journal of Gastroenterology. Supplement . 1996;215:57–62. doi: 10.3109/00365529609094536. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data, models, and code generated or used during the study appear in the submitted article.


