Abstract
Humanin (HN) is a 24-amino acid mitochondrial-derived polypeptide with cyto-protective and anti-apoptotic effects that regulates the mitochondrial functions under stress conditions. Accumulating evidence suggests the role of HN against age-related diseases, such as Alzheimer’s disease. The decline in insulin action is a metabolic feature of aging and thus, type 2 diabetes mellitus is considered an age-related disease, as well. It has been suggested that HN increases insulin sensitivity, improves the survival of pancreatic beta cells, and delays the onset of diabetes, actions that could be deployed in the treatment of diabetes. The aim of this review is to present the in vitro and in vivo studies that examined the role of HN in insulin resistance and diabetes and to discuss its newly emerging role as a therapeutic option against those conditions.
Keywords: Diabetes mellitus, Insulin resistance, Humanin, Aging, Apoptosis, Oxidative stress
Core Tip: Humanin (HN) exerts cyto-protective and anti-apoptotic effects. Type 2 diabetes mellitus (T2DM) is considered an age-related disease. Beyond the role of HN against age-related diseases, it increases insulin sensitivity, improves the survival of pancreatic beta cells, and delays the onset of diabetes. Altered HN levels could serve as a potential biomarker in prediabetes and T2DM, since they seem to be an effect or a response to the increased reactive oxygen species production, oxidative stress, and reduced mitochondrial DNA copy number-A major and important question is whether HN could be used as a potential therapeutic option for diabetes.
INTRODUCTION
Twenty years ago, three independent laboratories discovered humanin (HN) (MTRNR2), the first mitochondrial small open reading frame (sORF)-encoded microprotein found to have biological activity. The Hashimoto laboratory discovered HN while searching for survival factors in the unaffected brain section of an Alzheimer’s patient[1]. The investigators revealed a cDNA fragment that mapped back to the mitochondrial 16S rRNA. This microprotein was named humanin because it displayed protection against Alzheimer’s disease (AD)-related neurotoxicity, an action that the original authors though potentially could retrieve the “humanity” of patients suffering from dementia. Second, Ikonen et al[2] found that HN bound insulin like growth factor binding protein 3 (IGFBP3) using a yeast two-hybrid screening system and intensified the protective effects of IGFBP3 against amyloid-β (Aβ) toxicity. Also, Guo et al[3] showed that HN can bind and suppress the apoptotic protein BAX and, subsequently, alleviate cell apoptosis.
Physiologically, HN is produced by tissues in several organs, including kidney, skeletal muscles, brain, heart, and liver[4-6]. Subsequently, it is secreted into the blood circulation and transported to various target cells, protecting in parallel cells against several diseases strongly associated with oxidative stress, mitochondrial dysfunction, and cytotoxicity[7]. Beyond the cytoprotection HN possesses a key role in cell metabolism and mediates the production and secretion of endocrine/ paracrine/autocrine protective stress response factors[8]. Additionally, it plays a role in age-related diseases and several metabolic disorders (e.g., cardiovascular diseases [CVD], memory loss, stroke, diabetes type 2 [T2DM]).
Diabetes is a chronic disease that occurs either due to autoimmune destruction of the pancreatic beta cells, leading to absolute insulin deficiency (T1DM) or due to progressive attenuation of insulin secretion on a background of insulin resistance resulting in relative insulin deficiency (T2DM). The number of people with diabetes rose from 108 million in 1980 to 422 million in 2014. Prevalence has been increasing faster in low- and middle-income countries than in high-income countries. The rising burden of T2DM is a major concern in health care worldwide. In 2017 6.28% of the worldwide population was affected by T2DM. It is disconcerting that the burden of the disease is rising globally, and at a more rapid rate in developed regions such as western Europe[9]. As for the T1DM, its incidence is estimated 15 per 100000 people and the global prevalence 9.5%[10]. Since diabetes and its complications affect individuals’ functional capacities and quality of life leading to significant morbidity and premature mortality, effective agents are required for its treatment.
STRUCTURE OF HUMANIN PEPTIDE
HN is encoded by a sORF within the gene for the 16S ribosomal subunit in the mitochondrial genome[11]. HN has a positively charged N-terminal (Met-Ala-Pro-Arg), central hydrophobic region (Gly-Phe-Ser-Cys-Leu-Leu-Leu-Leu-Thr-Ser-Glu-Ile-Asp-Leu), and negatively charged C-terminal (Pro-Val-Lys-Arg-Arg-Ala)[1]. Last three amino acid residues in the C-terminal are considered as dispensable because both 21 and 24-amino acid long peptides have indistinguishable intracellular and extracellular effects[12]. Thirteen nuclear-encoded HN isoforms have been identified. HN-like ORF has been named MTRNR2L1 to MTRNR2L13 after the original humanin MTRNR2 gene in the mitochondrial genome. MTRNR2L1—MTRNR2L10 are expressed in most human tissues, with MTRNR2 being expressed in higher proportion in comparison to the other isoforms. Molecular manipulations of HN at key amino acids lead to changes in chemical characteristics. Additionally, single amino acid substitutions can lead to significant modifications to its biological functions and potency[13].
MECHANISMS OF ACTION
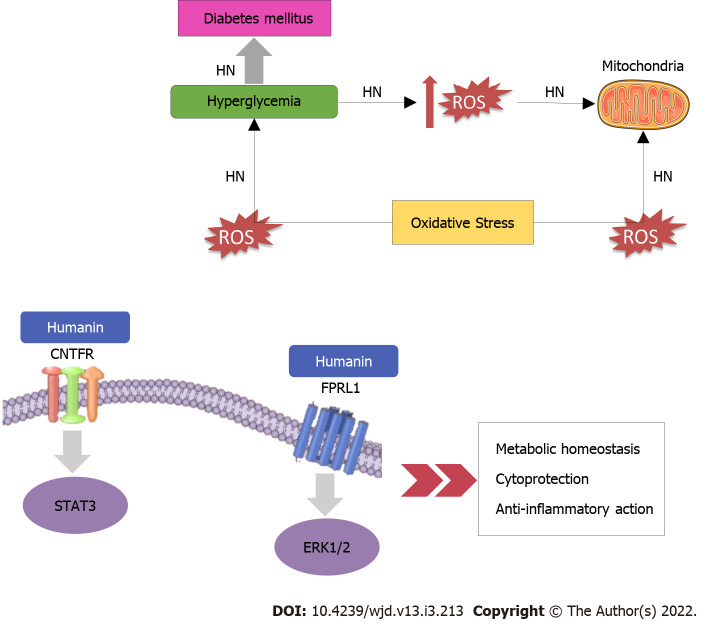
HN exerts its functions after connecting to either intracellular molecules or cell membrane receptors (Figure 1). Immediately after HN’s receptor binding, extracellular signal-regulated kinase 1/2 (ERK1/2) phosphorylation increases[14]. Once ERK1/2 is phosphorylated, it separates from its anchoring proteins, and transfers to other subcellular compartments. ERK1/2, a member of the mitogen-activated protein kinase pathway, participates in several essential cellular processes such as cell proliferation, survival, differentiation, mobility, and apoptosis[15,16]. HN behaves as a link to two different types of receptors: the seven-transmembrane G protein-coupled receptor formyl peptide receptor-like 1 (FPRL1) which plays a role in the cytoprotective properties of HN and a trimeric receptor, consisting of ciliary neurotrophic factor receptor (CNTFR), the cytokine receptor WSX-1 and the transmembrane glycoprotein 130 (GP130) (CNTFR/WSX-1/GP130) which is essential for HN activity and its neuroprotective effects[17]. As regards GP130, it is a transmembrane protein that acts as the signal transduction unit of the IL-6 receptor family[18]. Dimerization of GP130 receptors provokes the stimulation of janus kinases (JAK1 and JAK2), then subsequently provokes signal transducer and activator of transcription 3 (STAT3) and STAT1[19]. The dimerized STATs move to the nucleus and control transcription. The second signaling pathway directed by GP130 recruits SHP-2. SHP-2 is phosphorylated by JAK and interacts with growth-factor receptor bound protein 2 (Grb2), which allows the activation of mitogen-activated protein kinase (MAPK)[19].
Figure 1.
Mechanisms of action of Humanin in diabetes mellitus. CNTFR: Ciliary Neurotrophic Factor Receptor; ERK1/2: Extracellular signal-regulated protein kinases 1 and 2; FPRL1: Formyl peptide receptor-like 1; ΗΝ: Humanin; ROS: Reactive oxygen species; STAT3: Signal transducer and activator of transcription 3.
HN is regulated by insulin-growth factor 1 (IGF-1) and growth hormone (GH). HN and IGF-1 Levels decrease with age[20]. It has also been suggested that GH inhibits HN levels via IGF-1. Treatment with GH or IGF-1 reduces circulating HN levels in both mice and human subjects[21]. To date, HN has been suggested to play a role in various diseases like T2DM[22,23], CVD[4,5], memory loss[24], amyotrophic lateral sclerosis (ALS)[25], stroke[26], and inflammation[12,27]. The main mechanisms that dominate and link these age-related diseases are oxidative stress[28] and mitochondrial dysfunction[29]. Mitochondria are principal sources of reactive oxygen species (ROS) which can cause oxidative stress and injure of the lipids, proteins, and DNA of the cells. This can afflict mitochondrial function, and, subsequently, enhanced ROS production occurs[29]. These circumstances contribute to cellular damage, apoptosis, and cellular ageing, causing ageing and age-related diseases such as Parkinson’s disease[30], Alzheimer’s disease[31], atherosclerosis[32], heart failure[33], myocardial infarction[34], chronic inflammation[35], kidney disease[36], stroke[37], cancers[38], and many kinds of metabolic disorders[39,40].
Especially concerning diabetes, HN provides protection against apoptosis by binding pro-apoptotic Bax, inhibiting its mitochondrial localization, and lessening Bax-mediated apoptosis activation[3], acting either directly on Bax or through the FPRL-1 receptor[17]. As for its neuroprotective action, which has also a place in the neuroendocrine beta cells protection, it involves HN binding to a complex involving CNTFR/WSX-1/GP130[17] and activation of tyrosine kinases and STAT-3 phosphorylation[41]. Moreover, an important mechanism of cell protection may be via interfering with Jun N-terminal kinase (JNK) activity[42]. Important is also the interaction between HN and insulin-like growth factor binding protein-3 (IGFBP-3) which prevents the activation of caspases[2]. Furthermore, an alteration at position [Gly14]-HN (S14G, HNG) seems to induce neurosurvival activity and a substitution of phenylalanine in the 6th position with alanine (F6A, F6AHN) changes the binding of HN to IGFBP-3 and enhances its main effect on glucose metabolism and insulin sensitivity[5].
ROLE OF HUMANIN IN THE PATHOGENESIS OF TYPE 1 DIABETES
The role of HN in T1DM has been scarcely investigated. T1DM is characterized by the loss of pancreatic beta cells which results in insulin deficiency. The beta cells destruction, the dominant event in the pathogenesis of T1DM, occurs as a result of the IL-1, TNF-a, and IFN- γ actions which are originated from T cells and macrophages. Since HN is identified as a survival factor[43], it seems to serve also as a survival factor for neuroendocrine beta cells by decreasing cytokine-induced apoptosis and subsequently, improves glucose tolerance and onset of diabetes as it has been demonstrated in NOD mice in vivo[44]. Yet, no studies juxtaposing the HN levels in T1DM and T2DM have been published thus far.
ROLE OF HUMANIN IN THE PATHOGENESIS OF TYPE 2 DIABETES
T2DM is one of the most common metabolic diseases. This metabolic disorder and its comorbidities and complications, such as CVD, stroke, chronic kidney disease (CKD), and cancer, are global health problems which, noticeably, diminish quality of life and life expectancy[45-48].
Mitochondrial dysfunction and oxidative stress are involved in the pathogenesis of diabetes. Mitochondria are principal elements for the maintenance of metabolic health and cellular energy homeostasis. Mitochondrial dysfunction causes glycaemic dysregulation and metabolic derangement[49]. It causes inefficiency in the electron transport chain and beta-oxidation, thus trigging insulin resistance[50]. Furthermore, hyperglycemia provokes ROS generation which, in turn, causes oxidative stress in several tissues, cellular lipids, proteins, and DNA, and subsequently, provokes chronic inflammation[51]. The accumulation of oxidative damage leads to a decrement of mitochondrial function which can result in increased ROS production[29]. It has been suggested that mitochondrial dysfunction is implicated in diabetes-related complications impairing the kidneys, nervous system, heart and retina, and that mitochondrial dysfunction-related oxidative stress contributes to these complications[52]. Subsequently, an increase in ROS concentrations may provoke HN mobilization from various tissues to the impaired areas, where HN acts against oxidative stress, decreases ROS production, and promotes cell survival[51]. Mitochondrial derived peptides (MDPs), such as HN, have been suggested to play a critical role in reducing oxidative stress[53-55] and improving T2DM[56]. It has also been demonstrated that HN promotes mitochondrial biogenesis in pancreatic β-cells[57].
IN VITRO AND ANIMAL STUDIES
In vitro and animal studies
Considering that diseases related with ageing, named T2DM and neurodegeneration, have been suggested to be associated with mitochondrial dysfunction[58,59], it follows that the mitochondrial-derived peptide HN regulates them (Table 1). Based upon the molecular interaction between HN and IGFBP-3, that prevents the activation of caspases, and since IGFBP-3, independent of IGF-1, provokes IR both at the liver and periphery[60,61], Muzumdar et al[23] hypothesized that HN, besides its neuroprotective action, may regulate glucose homeostasis. Utilizing state of the art clamp technology, they investigated the role and the mechanism of action of central and peripheral HN in glucose metabolism. They finally demonstrated that infusion of HN improves both hepatic and peripheral insulin sensitivity and that hypothalamic STAT-3 activation is essential for the insulin-sensitizing action of HN. Moreover, treatment with a highly potent HN analog significantly lowered blood glucose in Zucker diabetic fatty rats. As for the levels of HN in tissues like hypothalamus, skeletal muscle, and cortex, they reduced with age in rodents, and its’ circulating levels were also diminished with age in humans and mice.
Table 1.
In vivo and in vitro studies on humanin and diabetes mellitus
|
Ref.
|
Study model
|
HN dose
|
Treatment duration
|
Results
|
| In vitro studies | ||||
| Rochette et al[51], 2014 (HN) | NIT-1 cells | 1-10000 nmol/L | 24 h | Reduced apoptosis caused by serum starvation in NIT-1 cells and decreased cytokine-induced apoptosis |
| Hunter and Jones[19], 2015 (HNGF6A) | Isolated islets and cultured murine β cell line | 50 ng/ml | 15-120 min | Enhanced glucose-stimulated insulin secretion |
| Qin et al[57], 2018 (HNG) | HUVECs | 1 μΜ | 3 h | Inhibited cell death, nucleus pyknosis and deformation. Diminished the expression of cleaved PARP (which reflects the level of apoptosis as well as ROS) Decreased the level of bax (a pro-apoptotic protein). Increased bcl-2 (an anti-apoptotic agent) |
| Miller et al[11], 2020 (HNG) | HEK293 and SH-SY5Y cells | 100 μM | 30 min | Is a major GP130 agonist which acts through the GP130/IL6ST receptor complex and activates AKT, ERK1/2, and STAT3 |
| Wang et al[50], 2010 (HN) | Pancreatic MIN6 β-cells | 25, 50 and 100 μΜ | 24h or48 h | Increased the expression of PGC-1α.Promoted mitochondrial biogenesis. Caused the phosphorylation of AMPK, improved mitochondrial respiration and stimulated ATP generation |
| Kim et al[60], 2007 (HN) | HUVECs | 200 μM | 24 h | Promoted the expression of KLF2.Reduced the expression of VCAM-1 and E-selectin; Impeded the secretion of TNF-α and IL-1β |
| In vivo studies | ||||
| Animals | ||||
| Hunter and Jones [19], 2015 (HNGF6A) | Sprague−Dawley rat | 0.07 mg/kg/h | 2-30 min | Improved insulin sensitivity and help in decreasing blood glucose level |
| Gong et al[20], 2014 (HN) | Sprague−Dawley rat | 0.375 mg/kg/h | 360 min | Decreased blood glucose in Sprague−Dawley rats by STAT-3 phosphorylation |
| Gong et al[20], 2014 (HNGF6A) | Zucker diabetic fatty rat | 0.05 mg/kg/h | 90-240 min | Decreased blood glucose in Zucker diabetic fatty rats |
| Rochette et al[51], 2014 (HN) | NOD mice | 0.7 mg/kg/day | 6 wk20 wk | Decreased lymphocyte infiltration in mice pancreata; Delayed or prevented the onset of diabetes in NOD mice (when the treatment was extended up to 20 wk) |
| Miller et al[11], 2020 (HNG) | Male C57BL/6 mice | 5 mg/kg/day | 2 wk | Old mice, but not young mice, showed an increase in phosphorylation in AKT and ERK1/2 in the hippocampus |
| Humans | ||||
| Muzumdar et al[61], 2006 | Participants attending a diabetes complications screening clinic | - | - | A significant decrease in HN was observed in the IFG group compared to control |
| Ha[71], 2006 | Uncomplicated T1DM patients | - | - | Plasma HN levels were significantly higher in T1D men by comparison with the healthy control men |
| do Nascimento et al[72], 2013 | Pregnant women with and without GDM | - | - | Serum HN levels were significantly lower in women with GDM compared to controls |
| Hashimoto et al[42], 2003 | Normal, prediabetes and diabetes subjects | - | - | Serum HN concentrations are lower in T2DM and correlate with HbA1c |
HUVECs: Human umbilical vein endothelial cells; PARP: Poly ADP-ribose polymerase; ROS: Reactive oxygen species; ERK1/2: Extracellular signal-regulated kinase 1/2; STAT3: Signal transducer and activator of transcription 3; PGC-1α: PPAR-γ coactivator-1α; KLF2: Krüppel-like factor 2; VCAM-1: Vascular cell adhesion molecule 1; TNF-α: Tumor necrosis factor-α; IL-1β: Interleukin-1β; IFG: Impaired fasting glucose; T1DM: Type 1 diabetes mellitus; GDM: Gestational diabetes mellitus.
A year later, a group from California[44] investigated whether HN could improve the survival of beta cells and delay or even treat diabetes in NOD mice. HN prevented apoptosis induced by serum starvation in NIT-1 cells and decreased cytokine exposure-related apoptosis (caused by interleukin [IL]-1β, tumor necrosis factor [TNF]α, and interferon[IFN]γ). STAT3 is considered as a principal survival signaling protein in beta cells, regulating the pro-survival effects of various growth factors and cytokines. HN activated STAT3 and ERK over a 24-hour time course. Interestingly, HN improved glucose tolerance in NOD mice and after 6 wk of treatment decreased lymphocyte infiltration was observed in their pancreata. When the treatment was extended up to 20 wk the investigators noted that HN delayed or prevented the onset of diabetes in NOD mice.
A few years later, the group we mentioned first[23] hypothesized that HNGF6A, a potent non-IGFBP-3 binding HN analog, may affect acutely and independently insulin secretion, since insulin concentrations were not reduced along with hypoglycemia caused by HNGF6A in Sprague Dawley rats[22]. Sprague Dawley rats that received HNGF6A presented higher insulin levels during hyperglycemic clamps compared to controls. Similarly, in vitro, HNGF6A enhanced glucose-stimulated insulin secretion in isolated islets and cultured murine β cell line. This effect was dose dependent, combined with ATP production in the β cell, related to the KATP-channel-independent augmentation phase of insulin release[62], and associated with amplified glucose metabolism. These potent effects on insulin secretion in combination with the effects on insulin action suggested a role of HN in the treatment of T2DM.
The protective effects of [Gly14]-Humanin (HNG) against high glucose-induced apoptosis were investigated in human umbilical vein endothelial cells (HUVECs). Pretreatment of HUVECs with HNG inhibited cell death, nucleus pyknosis and deformation[63]. Also, HNG diminished the expression of cleaved poly ADP-ribose polymerase (PARP) which reflects the level of apoptosis as well as reactive oxygen species (ROS). Regarding the level of bax, which is a pro-apoptotic protein, it decreased after pretreatment with HNG, while bcl-2, which exerts anti-apoptotic effects, it increased.
Another group identified a different sORF within the mitochondrial 12S rRNA encoding a 16-amino-acid peptide named MOTS-c (mitochondrial open reading frame of the 12S rRNA type-c) which also regulates insulin sensitivity and metabolic homeostasis[56]. Particularly, MOTS-c treatment in mice protected against age-dependent and high-fat-diet-induced insulin resistance and diet-induced obesity as well. Finally, they suggested that MDPs, like MOTS-c and HN, with such systemic effects may be useful in ameliorating the abnormal metabolism associated with aging in humans and regulating biological processes like weight and metabolic homeostasis.
Kim and his colleagues from California tried to elucidate the signaling pathways underlying HN’S cytoprotective roles in vitro and in vivo[14]. Utilizing multiple models, they showed that HN is a major GP130 agonist which acts through the GP130/IL6ST receptor complex and activates AKT, ERK1/2, and STAT3. PI3K, MEK, and JAK were suggested to be involved in the activation of those three signaling pathways, respectively.
Concerning the effects of HN on mitochondrial biogenesis in pancreatic β-cells, HN treatment in MIN6 β-cells increased the expression of peroxisome proliferator-activated receptor (PPAR) γ coactivator-1α (PGC-1α)[57] which promotes mitochondrial biogenesis by activating the expression of nuclear respiratory factor 1 (NRF-1) and mtDNA transcription factor A (TFAM)[64]. Also, HN treatment promoted mitochondrial biogenesis by increasing mitochondrial mass, elevating mitochondrial DNA (mtDNA)/nDNA ratio (reduced mtDNA copy number plays a key role in insulin resistance[65]), and increasing cytochrome B expression. Finally, HN treatment resulted in the phosphorylation of AMPK, which was involved in the induction of PGC-1α, NRF-1, and TFAM and improved mitochondrial respiration and stimulated ATP generation leading to a possible functional gain of the mitochondria.
In HUVECs also, HN displayed protective action against high-glucose-induced endothelial dysfunction and macrovascular complications[66]. HN treatment promoted the expression of Krüppel-like factor 2 (KLF2), a principal transcriptional regulator of endothelial function, by activating ERK5. In addition, HN significantly reduced the expression of vascular cell adhesion molecule 1 (VCAM-1) and E-selectin, which regulate the adhesion of circulating leukocytes to the endothelium, a principal procedure in the initiation of atherosclerosis. Furthermore, HN impeded the secretion of pro-inflammatory cytokines, such as TNF-α and IL-1β.
HUMAN SUBJECTS RESEARCH AND CLINICAL TRIALS
Human subjects research and clinical trials
The first attempt to measure HN levels in a clinical population with impaired fasting glucose (IFG) was made in participants attending a diabetes complications screening clinic (DiabHealth)[67,68]. Previous clinical studies reported noticeably increased HN levels in patients with mitochondrial encephalomyopathy, lactic acidosis, and stroke−like episodes (MELAS) and chronic progressive external ophthalmoplegia (CPEO), which are associated with excess oxidative stress[69,70]. However, a significant reduction (P = 0.0001) in HN was reported in the IFG group (n = 23; 204.84 ± 92.87 pg mL−1) compared to control (n = 58; 124.3 ± 83.91 pg mL−1) in accord with an adaptive cellular response by HN to a slight raise in fasting blood glucose level (BGL). As we described above, HN protects neuroendocrine β-cells[44] and increases glucose tolerance and insulin sensitivity[20,44]. Moreover, it is considered to interact with hydrogen peroxide and α-actinin-4 which rise during oxidative stress and IFG[71-73] and binds extracellularly with the CNTFR/WSX−1/GP130 receptor[69,74,75]. Interestingly, mild to moderate levels of ROS result in positive adaptive mechanisms of the mitochondria[76]. All these mechanisms, which benefit cell function and survival, lead to a reduction in HN levels, indicating a protective role of HN. However, with disease progression to T2DM and further oxidative stress, mitochondria may upregulate HN levels as observed in studies of Alzheimer's disease and in those of MELAS and CPEO.
These conditions are related to extensive oxidative stress which is also a key feature of DM. Particularly, hyperglycemia causes extended free radical activity and mitochondrial dysfunction which induce oxidative stress and release more ROS[76]. The advanced diseases MELAS and CPEO are associated with increased plasma HN levels. HN has a protective role and is upregulated with disease progression. On the contrary, the minor elevations of blood glucose levels are combined with a decrease in HN concentrations which supports the protective role of HN when levels are expected to decrease as a result of stimulation of oxidative stress-associated agents that are inhibited by HN. However, with disease progression to T2DM and further oxidative stress, mitochondria increase HN levels, as reported in MELAS and CPEO.
A few years earlier, another group from Toronto suggested that plasma HN levels were significantly higher in T1D men by comparison with the healthy control men (P < 0.0001)[77].
At the end of 2018 Ma et al[78] evaluated HN concentrations in pregnant women with and without gestational diabetes mellitus (GDM) aiming to define the role of HN in the development of GDM. 157 women were enrolled in the study. Serum HN levels were significantly lower in women with GDM compared to controls. Like Lee et al[21], who found that HN was regulated by IGF-1 in mice and humans, they suggested that the IGF axis influenced the HN levels and affected its normal function in GDM. By performing logistic regression analysis, they also showed that low HN levels were the independent risk factor of GDM and, therefore, might be a predictor for the GDM diagnosis. Additionally, HN levels were significantly negatively correlated with the body weight, body mass index (BMI) and homeostatic model assessment for insulin resistance (HOMA-IR).
The most recent study which attempted to evaluate MDP levels in normal, prediabetes and diabetes subjects enrolled 225 participants[49]. The investigators found that serum HN concentrations are lower in T2DM (P < 0.0001) and correlate with HbA1c. Interestingly, HN levels decreased by 62% in the prediabetes group, 66% in diabetes subjects with good control and 77% in uncontrolled diabetes patients compared to participants without diabetes. Also, this study confirmed that there are no significant differences in HN levels between healthy men and women and the levels of HN were not affected by the different anti-diabetic treatment (insulin, metformin, other hypoglycemic regimens) or the duration of therapy. Furthermore, since HN was associated with adiponectin, which has been suggested to be reduced in prediabetes and T2DM[79], it can be concluded that mitochondrial dysfunction contributes to glycemic dysregulation and metabolic effects in T2DM. Adiponectin levels were positively correlated with HN. Adiponectin concentrations decrease in pre-diabetes and DM[79]. It has also been demonstrated that adiponectin knockout mice have reduced mitochondrial content combined with insulin resistance[80]. In addition adiponectin may impair mitochondrial biogenesis[81]. Therefore, the affected mitochondrial function may arise from the low adiponectin levels.
As for the changes in HN levels with ageing, Voigt et al[67] showed that HN decreased with age among individuals attending a diabetes complications screening clinic suggesting a protective function of HN and this observation was consistent with a previous study among human and mice[23]. On the contrary, circulating levels of HN increase in age-associated diseases such as T2DM. With disease progression and additional oxidative stress, mitochondria may increase HN levels.
Besides the initial and principal lifestyle interventions for glycemic control in DM, currently, we have various oral and injectable pharmacologic agents at our disposal including metformin, thiazolidinediones, sulfonylureas, glucagon-like peptide 1 (GLP-1) receptor agonists, dipeptyl-peptidase 4 (DPP-4) inhibitors, sodium-glucose co-transporter 2 (SGLT-2) inhibitors, and insulin[82]. These medicines can be administered in various dosages and in many combinations in each patient diagnosed with DM. However, there is still room for additional new factors that could efficiently contribute to the management of the disease. Given HN’s protective properties, it may represent a novel treatment option to decrease the cellular damage caused by diabetes. Altered HN levels in diabetes could serve as a potential biomarker. Nevertheless, no clinical trials investigating the effects of HN or its analogues (e.g. HNGF6a) administration have thus far been published, albeit it would be an innovative and promising breakthrough in diabetes prevention and treatment.
CONCLUSION
In summary, HN shows cytoprotective effects in many biological processes, including oxidative stress and apoptosis. Altered HN levels could serve as a potential biomarker in prediabetes and T2DM, since they seem to be an effect or a response to the increased ROS production, oxidative stress, and reduced mtDNA copy number which all contribute to IR[83]. However, further study is needed to define the role of age and other modifiable confounding factors, like fitness level, adiposity, other metabolic comorbidities, such as CVD, stroke, inflammation. Undoubtedly, the major and important question is whether HN could be used as a potential therapeutic option for diabetes, that could even replace the current diabetes mellitus treatment strategies soon. Towards this direction, further studies are needed to identify the contribution of HN in the metabolic dysregulation of T2DM.
Footnotes
Conflict-of-interest statement: There are no conflicts of interest to declare.
Open-Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/Licenses/by-nc/4.0/
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Peer-review started: August 30, 2021
First decision: October 3, 2021
Article in press: February 23, 2022
Specialty type: Endocrinology and metabolism
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): A, A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Darenskaya MA, Lauro D, Liu D, Ma JH, Xia YK S-Editor: Wang LL L-Editor: A P-Editor: Wang LL
Contributor Information
Chrysoula Boutari, Second Propedeutic Department of Internal Medicine, Hippocration Hospital, Aristotle University of Thessaloniki, Thessaloniki 54642, Greece. chrisoulabgr@yahoo.gr.
Panagiotis D Pappas, First Department of Obstetrics and Gynaecology, Papageorgiou Hospital, Aristotle University of Thessaloniki, Thessaloniki 56429, Greece.
Theodoros D Theodoridis, First Department of Obstetrics and Gynaecology, Papageorgiou Hospital, Aristotle University of Thessaloniki, Thessaloniki 56429, Greece.
Dimitrios Vavilis, First Department of Obstetrics and Gynaecology, Papageorgiou Hospital, Aristotle University of Thessaloniki, Thessaloniki 56429, Greece; Medical School, University of Cyprus, Nicosia, Cyprus 20537 1678, Cyprus.
References
- 1.Hashimoto Y, Niikura T, Tajima H, Yasukawa T, Sudo H, Ito Y, Kita Y, Kawasumi M, Kouyama K, Doyu M, Sobue G, Koide T, Tsuji S, Lang J, Kurokawa K, Nishimoto I. A rescue factor abolishing neuronal cell death by a wide spectrum of familial Alzheimer's disease genes and Abeta. Proc Natl Acad Sci U S A . 2001;98:6336–6341. doi: 10.1073/pnas.101133498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ikonen M, Liu B, Hashimoto Y, Ma L, Lee KW, Niikura T, Nishimoto I, Cohen P. Interaction between the Alzheimer's survival peptide humanin and insulin-like growth factor-binding protein 3 regulates cell survival and apoptosis. Proc Natl Acad Sci U S A . 2003;100:13042–13047. doi: 10.1073/pnas.2135111100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Guo B, Zhai D, Cabezas E, Welsh K, Nouraini S, Satterthwait AC, Reed JC. Humanin peptide suppresses apoptosis by interfering with Bax activation. Nature . 2003;423:456–461. doi: 10.1038/nature01627. [DOI] [PubMed] [Google Scholar]
- 4.Zhang X, Urbieta-Caceres VH, Eirin A, Bell CC, Crane JA, Tang H, Jordan KL, Oh YK, Zhu XY, Korsmo MJ, Bachar AR, Cohen P, Lerman A, Lerman LO. Humanin prevents intra-renal microvascular remodeling and inflammation in hypercholesterolemic ApoE deficient mice. Life Sci . 2012;91:199–206. doi: 10.1016/j.lfs.2012.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Muzumdar RH, Huffman DM, Calvert JW, Jha S, Weinberg Y, Cui L, Nemkal A, Atzmon G, Klein L, Gundewar S, Ji SY, Lavu M, Predmore BL, Lefer DJ. Acute humanin therapy attenuates myocardial ischemia and reperfusion injury in mice. Arterioscler Thromb Vasc Biol . 2010;30:1940–1948. doi: 10.1161/ATVBAHA.110.205997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Brookmeyer R, Gray S, Kawas C. Projections of Alzheimer's disease in the United States and the public health impact of delaying disease onset. Am J Public Health . 1998;88:1337–1342. doi: 10.2105/ajph.88.9.1337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Widmer RJ, Flammer AJ, Herrmann J, Rodriguez-Porcel M, Wan J, Cohen P, Lerman LO, Lerman A. Circulating humanin levels are associated with preserved coronary endothelial function. Am J Physiol Heart Circ Physiol . 2013;304:H393–H397. doi: 10.1152/ajpheart.00765.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zuccato CF, Asad AS, Nicola Candia AJ, Gottardo MF, Moreno Ayala MA, Theas MS, Seilicovich A, Candolfi M. Mitochondrial-derived peptide humanin as therapeutic target in cancer and degenerative diseases. Expert Opin Ther Targets . 2019;23:117–126. doi: 10.1080/14728222.2019.1559300. [DOI] [PubMed] [Google Scholar]
- 9.Khan MAB, Hashim MJ, King JK, Govender RD, Mustafa H, Al Kaabi J. Epidemiology of Type 2 Diabetes-Global Burden of Disease and Forecasted Trends. J Epidemiol Glob Health . 2020;10:107–111. doi: 10.2991/jegh.k.191028.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mobasseri M, Shirmohammadi M, Amiri T, Vahed N, Hosseini Fard H, Ghojazadeh M. Prevalence and incidence of type 1 diabetes in the world: a systematic review and meta-analysis. Health Promot Perspect . 2020;10:98–115. doi: 10.34172/hpp.2020.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Miller B, Kim SJ, Kumagai H, Mehta HH, Xiang W, Liu J, Yen K, Cohen P. Peptides derived from small mitochondrial open reading frames: Genomic, biological, and therapeutic implications. Exp Cell Res . 2020;393:112056. doi: 10.1016/j.yexcr.2020.112056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zapała B, Kaczyński Ł, Kieć-Wilk B, Staszel T, Knapp A, Thoresen GH, Wybrańska I, Dembińska-Kieć A. Humanins, the neuroprotective and cytoprotective peptides with antiapoptotic and anti-inflammatory properties. Pharmacol Rep . 2010;62:767–777. doi: 10.1016/s1734-1140(10)70337-6. [DOI] [PubMed] [Google Scholar]
- 13.Bodzioch M, Lapicka-Bodzioch K, Zapala B, Kamysz W, Kiec-Wilk B, Dembinska-Kiec A. Evidence for potential functionality of nuclearly-encoded humanin isoforms. Genomics . 2009;94:247–256. doi: 10.1016/j.ygeno.2009.05.006. [DOI] [PubMed] [Google Scholar]
- 14.Kim SJ, Guerrero N, Wassef G, Xiao J, Mehta HH, Cohen P, Yen K. The mitochondrial-derived peptide humanin activates the ERK1/2, AKT, and STAT3 signaling pathways and has age-dependent signaling differences in the hippocampus. Oncotarget . 2016;7:46899–46912. doi: 10.18632/oncotarget.10380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Steelman LS, Chappell WH, Abrams SL, Kempf RC, Long J, Laidler P, Mijatovic S, Maksimovic-Ivanic D, Stivala F, Mazzarino MC, Donia M, Fagone P, Malaponte G, Nicoletti F, Libra M, Milella M, Tafuri A, Bonati A, Bäsecke J, Cocco L, Evangelisti C, Martelli AM, Montalto G, Cervello M, McCubrey JA. Roles of the Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR pathways in controlling growth and sensitivity to therapy-implications for cancer and aging. Aging (Albany NY) . 2011;3:192–222. doi: 10.18632/aging.100296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chang F, Steelman LS, Lee JT, Shelton JG, Navolanic PM, Blalock WL, Franklin RA, McCubrey JA. Signal transduction mediated by the Ras/Raf/MEK/ERK pathway from cytokine receptors to transcription factors: potential targeting for therapeutic intervention. Leukemia . 2003;17:1263–1293. doi: 10.1038/sj.leu.2402945. [DOI] [PubMed] [Google Scholar]
- 17.Hashimoto Y, Kurita M, Aiso S, Nishimoto I, Matsuoka M. Humanin inhibits neuronal cell death by interacting with a cytokine receptor complex or complexes involving CNTF receptor alpha/WSX-1/gp130. Mol Biol Cell . 2009;20:2864–2873. doi: 10.1091/mbc.E09-02-0168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bauer S, Kerr BJ, Patterson PH. The neuropoietic cytokine family in development, plasticity, disease and injury. Nat Rev Neurosci . 2007;8:221–232. doi: 10.1038/nrn2054. [DOI] [PubMed] [Google Scholar]
- 19.Hunter CA, Jones SA. IL-6 as a keystone cytokine in health and disease. Nat Immunol . 2015;16:448–457. doi: 10.1038/ni.3153. [DOI] [PubMed] [Google Scholar]
- 20.Gong Z, Tas E, Muzumdar R. Humanin and age-related diseases: a new link? Front Endocrinol (Lausanne) . 2014;5:210. doi: 10.3389/fendo.2014.00210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee C, Wan J, Miyazaki B, Fang Y, Guevara-Aguirre J, Yen K, Longo V, Bartke A, Cohen P. IGF-I regulates the age-dependent signaling peptide humanin. Aging Cell . 2014;13:958–961. doi: 10.1111/acel.12243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kuliawat R, Klein L, Gong Z, Nicoletta-Gentile M, Nemkal A, Cui L, Bastie C, Su K, Huffman D, Surana M, Barzilai N, Fleischer N, Muzumdar R. Potent humanin analog increases glucose-stimulated insulin secretion through enhanced metabolism in the β cell. FASEB J . 2013;27:4890–4898. doi: 10.1096/fj.13-231092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Muzumdar RH, Huffman DM, Atzmon G, Buettner C, Cobb LJ, Fishman S, Budagov T, Cui L, Einstein FH, Poduval A, Hwang D, Barzilai N, Cohen P. Humanin: a novel central regulator of peripheral insulin action. PLoS One . 2009;4:e6334. doi: 10.1371/journal.pone.0006334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mamiya T, Ukai M. [Gly(14)]-Humanin improved the learning and memory impairment induced by scopolamine in vivo. Br J Pharmacol . 2001;134:1597–1599. doi: 10.1038/sj.bjp.0704429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Matsuoka M, Hashimoto Y, Aiso S, Nishimoto I. Humanin and colivelin: neuronal-death-suppressing peptides for Alzheimer's disease and amyotrophic lateral sclerosis. CNS Drug Rev . 2006;12:113–122. doi: 10.1111/j.1527-3458.2006.00113.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xu X, Chua CC, Gao J, Hamdy RC, Chua BH. Humanin is a novel neuroprotective agent against stroke. Stroke . 2006;37:2613–2619. doi: 10.1161/01.STR.0000242772.94277.1f. [DOI] [PubMed] [Google Scholar]
- 27.Zhao ST, Zhao L, Li JH. Neuroprotective Peptide humanin inhibits inflammatory response in astrocytes induced by lipopolysaccharide. Neurochem Res . 2013;38:581–588. doi: 10.1007/s11064-012-0951-6. [DOI] [PubMed] [Google Scholar]
- 28.Dai DF, Chiao YA, Marcinek DJ, Szeto HH, Rabinovitch PS. Mitochondrial oxidative stress in aging and healthspan. Longev Healthspan . 2014;3:6. doi: 10.1186/2046-2395-3-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cui H, Kong Y, Zhang H. Oxidative stress, mitochondrial dysfunction, and aging. J Signal Transduct . 2012;2012:646354. doi: 10.1155/2012/646354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jenner P. Oxidative stress in Parkinson's disease. Ann Neurol . 2003;53 Suppl 3:S26–36; discussion S36. doi: 10.1002/ana.10483. [DOI] [PubMed] [Google Scholar]
- 31.Markesbery WR. Oxidative stress hypothesis in Alzheimer's disease. Free Radic Biol Med . 1997;23:134–147. doi: 10.1016/s0891-5849(96)00629-6. [DOI] [PubMed] [Google Scholar]
- 32.Singh U, Jialal I. Oxidative stress and atherosclerosis. Pathophysiology . 2006;13:129–142. doi: 10.1016/j.pathophys.2006.05.002. [DOI] [PubMed] [Google Scholar]
- 33.Tsutsui H, Kinugawa S, Matsushima S. Oxidative stress and heart failure. Am J Physiol Heart Circ Physiol . 2011;301:H2181–H2190. doi: 10.1152/ajpheart.00554.2011. [DOI] [PubMed] [Google Scholar]
- 34.Di Filippo C, Cuzzocrea S, Rossi F, Marfella R, D'Amico M. Oxidative stress as the leading cause of acute myocardial infarction in diabetics. Cardiovasc Drug Rev . 2006;24:77–87. doi: 10.1111/j.1527-3466.2006.00077.x. [DOI] [PubMed] [Google Scholar]
- 35.Federico A, Morgillo F, Tuccillo C, Ciardiello F, Loguercio C. Chronic inflammation and oxidative stress in human carcinogenesis. Int J Cancer . 2007;121:2381–2386. doi: 10.1002/ijc.23192. [DOI] [PubMed] [Google Scholar]
- 36.Ozbek E. Induction of oxidative stress in kidney. Int J Nephrol . 2012;2012:465897. doi: 10.1155/2012/465897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Allen CL, Bayraktutan U. Oxidative stress and its role in the pathogenesis of ischaemic stroke. Int J Stroke . 2009;4:461–470. doi: 10.1111/j.1747-4949.2009.00387.x. [DOI] [PubMed] [Google Scholar]
- 38.Sosa V, Moliné T, Somoza R, Paciucci R, Kondoh H, LLeonart ME. Oxidative stress and cancer: an overview. Ageing Res Rev . 2013;12:376–390. doi: 10.1016/j.arr.2012.10.004. [DOI] [PubMed] [Google Scholar]
- 39.Roberts CK, Sindhu KK. Oxidative stress and metabolic syndrome. Life Sci . 2009;84:705–712. doi: 10.1016/j.lfs.2009.02.026. [DOI] [PubMed] [Google Scholar]
- 40.Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, Nakayama O, Makishima M, Matsuda M, Shimomura I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest . 2004;114:1752–1761. doi: 10.1172/JCI21625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hashimoto Y, Suzuki H, Aiso S, Niikura T, Nishimoto I, Matsuoka M. Involvement of tyrosine kinases and STAT3 in Humanin-mediated neuroprotection. Life Sci . 2005;77:3092–3104. doi: 10.1016/j.lfs.2005.03.031. [DOI] [PubMed] [Google Scholar]
- 42.Hashimoto Y, Tsuji O, Niikura T, Yamagishi Y, Ishizaka M, Kawasumi M, Chiba T, Kanekura K, Yamada M, Tsukamoto E, Kouyama K, Terashita K, Aiso S, Lin A, Nishimoto I. Involvement of c-Jun N-terminal kinase in amyloid precursor protein-mediated neuronal cell death. J Neurochem . 2003;84:864–877. doi: 10.1046/j.1471-4159.2003.01585.x. [DOI] [PubMed] [Google Scholar]
- 43.Eizirik DL, Mandrup-Poulsen T. A choice of death--the signal-transduction of immune-mediated beta-cell apoptosis. Diabetologia . 2001;44:2115–2133. doi: 10.1007/s001250100021. [DOI] [PubMed] [Google Scholar]
- 44.Hoang PT, Park P, Cobb LJ, Paharkova-Vatchkova V, Hakimi M, Cohen P, Lee KW. The neurosurvival factor Humanin inhibits beta-cell apoptosis via signal transducer and activator of transcription 3 activation and delays and ameliorates diabetes in nonobese diabetic mice. Metabolism . 2010;59:343–349. doi: 10.1016/j.metabol.2009.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Grundy SM. Adipose tissue and metabolic syndrome: too much, too little or neither. Eur J Clin Invest . 2015;45:1209–1217. doi: 10.1111/eci.12519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fang X, Liu H, Zhang X, Zhang H, Qin X, Ji X. Metabolic Syndrome, Its Components, and Diabetes on 5-Year Risk of Recurrent Stroke among Mild-to-Moderate Ischemic Stroke Survivors: A Multiclinic Registry Study. J Stroke Cerebrovasc Dis . 2016;25:626–634. doi: 10.1016/j.jstrokecerebrovasdis.2015.11.017. [DOI] [PubMed] [Google Scholar]
- 47.Esser N, Paquot N, Scheen AJ. Inflammatory markers and cardiometabolic diseases. Acta Clin Belg . 2015;70:193–199. doi: 10.1179/2295333715Y.0000000004. [DOI] [PubMed] [Google Scholar]
- 48.Shimabukuro M, Kozuka C, Taira S-I, Yabiku K, Dagvasumberel M, Ishida M. Ectopic fat deposition and global cardiometabolic risk: New paradigm in cardiovascular medicine. J Med Invest . 2013 doi: 10.2152/jmi.60.1. [DOI] [PubMed] [Google Scholar]
- 49.Ramanjaneya M, Bettahi I, Jerobin J, Chandra P, Abi Khalil C, Skarulis M, Atkin SL, Abou-Samra AB. Mitochondrial-Derived Peptides Are Down Regulated in Diabetes Subjects. Front Endocrinol (Lausanne) . 2019;10:331. doi: 10.3389/fendo.2019.00331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wang CH, Wang CC, Wei YH. Mitochondrial dysfunction in insulin insensitivity: implication of mitochondrial role in type 2 diabetes. Ann N Y Acad Sci . 2010;1201:157–165. doi: 10.1111/j.1749-6632.2010.05625.x. [DOI] [PubMed] [Google Scholar]
- 51.Rochette L, Zeller M, Cottin Y, Vergely C. Diabetes, oxidative stress and therapeutic strategies. Biochim Biophys Acta . 2014;1840:2709–2729. doi: 10.1016/j.bbagen.2014.05.017. [DOI] [PubMed] [Google Scholar]
- 52.Blake R, Trounce IA. Mitochondrial dysfunction and complications associated with diabetes. Biochim Biophys Acta . 2014;1840:1404–1412. doi: 10.1016/j.bbagen.2013.11.007. [DOI] [PubMed] [Google Scholar]
- 53.Klein LE, Cui L, Gong Z, Su K, Muzumdar R. A humanin analog decreases oxidative stress and preserves mitochondrial integrity in cardiac myoblasts. Biochem Biophys Res Commun . 2013;440:197–203. doi: 10.1016/j.bbrc.2013.08.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Bachar AR, Scheffer L, Schroeder AS, Nakamura HK, Cobb LJ, Oh YK, Lerman LO, Pagano RE, Cohen P, Lerman A. Humanin is expressed in human vascular walls and has a cytoprotective effect against oxidized LDL-induced oxidative stress. Cardiovasc Res . 2010;88:360–366. doi: 10.1093/cvr/cvq191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Oh YK, Bachar AR, Zacharias DG, Kim SG, Wan J, Cobb LJ, Lerman LO, Cohen P, Lerman A. Humanin preserves endothelial function and prevents atherosclerotic plaque progression in hypercholesterolemic ApoE deficient mice. Atherosclerosis . 2011;219:65–73. doi: 10.1016/j.atherosclerosis.2011.06.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lee C, Zeng J, Drew BG, Sallam T, Martin-Montalvo A, Wan J, Kim SJ, Mehta H, Hevener AL, de Cabo R, Cohen P. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metab . 2015;21:443–454. doi: 10.1016/j.cmet.2015.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Qin Q, Jin J, He F, Zheng Y, Li T, Zhang Y, He J. Humanin promotes mitochondrial biogenesis in pancreatic MIN6 β-cells. Biochem Biophys Res Commun . 2018;497:292–297. doi: 10.1016/j.bbrc.2018.02.071. [DOI] [PubMed] [Google Scholar]
- 58.Frederiksen AL, Jeppesen TD, Vissing J, Schwartz M, Kyvik KO, Schmitz O, Poulsen PL, Andersen PH. High prevalence of impaired glucose homeostasis and myopathy in asymptomatic and oligosymptomatic 3243A>G mitochondrial DNA mutation-positive subjects. J Clin Endocrinol Metab . 2009;94:2872–2879. doi: 10.1210/jc.2009-0235. [DOI] [PubMed] [Google Scholar]
- 59.Loeb LA, Wallace DC, Martin GM. The mitochondrial theory of aging and its relationship to reactive oxygen species damage and somatic mtDNA mutations. Proc Natl Acad Sci U S A . 2005;102:18769–18770. doi: 10.1073/pnas.0509776102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kim HS, Ali O, Shim M, Lee KW, Vuguin P, Muzumdar R, Barzilai N, Cohen P. Insulin-like growth factor binding protein-3 induces insulin resistance in adipocytes in vitro and in rats in vivo. Pediatr Res . 2007;61:159–164. doi: 10.1203/pdr.0b013e31802d8a30. [DOI] [PubMed] [Google Scholar]
- 61.Muzumdar RH, Ma X, Fishman S, Yang X, Atzmon G, Vuguin P, Einstein FH, Hwang D, Cohen P, Barzilai N. Central and opposing effects of IGF-I and IGF-binding protein-3 on systemic insulin action. Diabetes . 2006;55:2788–2796. doi: 10.2337/db06-0318. [DOI] [PubMed] [Google Scholar]
- 62.Straub SG, Sharp GW. Glucose-stimulated signaling pathways in biphasic insulin secretion. Diabetes Metab Res Rev . 2002;18:451–463. doi: 10.1002/dmrr.329. [DOI] [PubMed] [Google Scholar]
- 63.Xie Y, Liu ZH, Li XY, Zhou YD, Xu X, Hu LF, Zhang YL, Liu CF. Protection effect of [Gly14]-Humanin from apoptosis induced by high glucose in human umbilical vein endothelial cells. Diabetes Res Clin Pract . 2014;106:560–566. doi: 10.1016/j.diabres.2014.09.020. [DOI] [PubMed] [Google Scholar]
- 64.Scarpulla RC. Nuclear activators and coactivators in mammalian mitochondrial biogenesis. Biochim Biophys Acta . 2002;1576:1–14. doi: 10.1016/s0167-4781(02)00343-3. [DOI] [PubMed] [Google Scholar]
- 65.Cheng Z, Almeida FA. Mitochondrial alteration in type 2 diabetes and obesity: an epigenetic link. Cell Cycle . 2014;13:890–897. doi: 10.4161/cc.28189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Wang X, Wu Z, He Y, Zhang H, Tian L, Zheng C, Shang T, Zhu Q, Li D. Humanin prevents high glucose-induced monocyte adhesion to endothelial cells by targeting KLF2. Mol Immunol . 2018;101:245–250. doi: 10.1016/j.molimm.2018.07.008. [DOI] [PubMed] [Google Scholar]
- 67.Voigt A, Jelinek HF. Humanin: a mitochondrial signaling peptide as a biomarker for impaired fasting glucose-related oxidative stress. Physiol Rep . 2016;4 doi: 10.14814/phy2.12796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Jelinek HF, Wilding C, Tinely P. An innovative Multi-disciplinary Diabetes Complications Screening Program in a Rural Community: A Description and Preliminary Results of the Screening. Aust J Prim Health . 2006;12:14–20. [Google Scholar]
- 69.Kariya S, Hirano M, Furiya Y, Sugie K, Ueno S. Humanin detected in skeletal muscles of MELAS patients: a possible new therapeutic agent. Acta Neuropathol . 2005;109:367–372. doi: 10.1007/s00401-004-0965-5. [DOI] [PubMed] [Google Scholar]
- 70.Kin T, Sugie K, Hirano M, Goto YI, Nishino I, Ueno S. Humanin expression in skeletal muscles of patients with chronic progressive external ophthalmoplegia. J Hum Genet . 2006;51:555–558. doi: 10.1007/s10038-006-0397-2. [DOI] [PubMed] [Google Scholar]
- 71.Ha TS. High glucose and advanced glycosylated end-products affect the expression of alpha-actinin-4 in glomerular epithelial cells. Nephrology (Carlton) . 2006;11:435–441. doi: 10.1111/j.1440-1797.2006.00668.x. [DOI] [PubMed] [Google Scholar]
- 72.do Nascimento JF, Canani LH, Gerchman F, Rodrigues PG, Joelsons G, dos Santos M, Pereira S, Veronese FV. Messenger RNA levels of podocyte-associated proteins in subjects with different degrees of glucose tolerance with or without nephropathy. BMC Nephrol . 2013;14:214. doi: 10.1186/1471-2369-14-214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Francés DE, Ronco MT, Monti JA, Ingaramo PI, Pisani GB, Parody JP, Pellegrino JM, Sanz PM, Carrillo MC, Carnovale CE. Hyperglycemia induces apoptosis in rat liver through the increase of hydroxyl radical: new insights into the insulin effect. J Endocrinol . 2010;205:187–200. doi: 10.1677/JOE-09-0462. [DOI] [PubMed] [Google Scholar]
- 74.Pagano G, Talamanca AA, Castello G, Cordero MD, d'Ischia M, Gadaleta MN, Pallardó FV, Petrović S, Tiano L, Zatterale A. Oxidative stress and mitochondrial dysfunction across broad-ranging pathologies: toward mitochondria-targeted clinical strategies. Oxid Med Cell Longev . 2014;2014:541230. doi: 10.1155/2014/541230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Katayama Y, Maeda K, Iizuka T, Hayashi M, Hashizume Y, Sanada M, Kawai H, Kashiwagi A. Accumulation of oxidative stress around the stroke-like lesions of MELAS patients. Mitochondrion . 2009;9:306–313. doi: 10.1016/j.mito.2009.04.002. [DOI] [PubMed] [Google Scholar]
- 76.Long YC, Tan TM, Takao I, Tang BL. The biochemistry and cell biology of aging: metabolic regulation through mitochondrial signaling. Am J Physiol Endocrinol Metab . 2014;306:E581–E591. doi: 10.1152/ajpendo.00665.2013. [DOI] [PubMed] [Google Scholar]
- 77.Lytvyn Y, Wan J, Lai V, Cohen P, Cherney DZ. The effect of sex on humanin levels in healthy adults and patients with uncomplicated type 1 diabetes mellitus. Can J Physiol Pharmacol . 2015;93:239–243. doi: 10.1139/cjpp-2014-0401. [DOI] [PubMed] [Google Scholar]
- 78.Ma Y, Li S, Wei X, Huang J, Lai M, Wang N, Huang Q, Zhao L, Peng Y, Wang Y. Comparison of serum concentrations of humanin in women with and without gestational diabetes mellitus. Gynecol Endocrinol . 2018;34:1064–1067. doi: 10.1080/09513590.2018.1482869. [DOI] [PubMed] [Google Scholar]
- 79.Kim JY, Bacha F, Tfayli H, Michaliszyn SF, Yousuf S, Arslanian S. Adipose Tissue Insulin Resistance in Youth on the Spectrum From Normal Weight to Obese and From Normal Glucose Tolerance to Impaired Glucose Tolerance to Type 2 Diabetes. Diabetes Care . 2019;42:265–272. doi: 10.2337/dc18-1178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Civitarese AE, Ukropcova B, Carling S, Hulver M, DeFronzo RA, Mandarino L, Ravussin E, Smith SR. Role of adiponectin in human skeletal muscle bioenergetics. Cell Metab . 2006;4:75–87. doi: 10.1016/j.cmet.2006.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.O'Brien LC, Graham ZA, Chen Q, Lesnefsky EJ, Cardozo C, Gorgey AS. Plasma adiponectin levels are correlated with body composition, metabolic profiles, and mitochondrial markers in individuals with chronic spinal cord injury. Spinal Cord . 2018;56:863–872. doi: 10.1038/s41393-018-0089-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.American Diabetes Association. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes-2020. Diabetes Care . 2020;43:S98–S110. doi: 10.2337/dc20-S009. [DOI] [PubMed] [Google Scholar]
- 83.Tan BL, Norhaizan ME, Liew WP. Nutrients and Oxidative Stress: Friend or Foe? Oxid Med Cell Longev . 2018;2018:9719584. doi: 10.1155/2018/9719584. [DOI] [PMC free article] [PubMed] [Google Scholar]