Summary
Lymphomas of the breast are rare neoplasms that arise from breast lymphoid tissue and are characterised by neoplastic B or T cells. Breast lymphomas arising from B cells include, but are not limited to, diffuse large B cell lymphoma, follicular lymphoma, extra-nodal marginal zone lymphoma and Burkitt lymphoma. Anaplastic large cell lymphoma (ALCL) is of a T cell origin and both anaplastic lymphoma kinase (ALK)-positive and ALK-negative presentations have been noted in the breast. In addition, there is a more recently identified presentation of ALK-negative ALCL that arises around textured breast implants and is usually confined to a periprosthetic fibrous capsule. Here, we discuss the clinical presentations, histological and immunohistochemical features and treatment options for each type of primary breast lymphoma. We hope that this review will highlight the importance of the timely and accurate diagnosis of breast lymphoma in order to tailor the most appropriate treatment. We also wish to raise awareness of the breast implant-associated lymphomas, with the goal of stimulating work that will aid our understanding of their epidemiology and pathogenesis.
Keywords: breast lymphoma, breast implant lymphoma, BIA-ALCL, non-Hodgkin lymphoma, breast implants
Introduction
Cancers of the breast affect millions of women worldwide and are a major cause of premature death.1 Breast cancer mostly arises from either the epithelial or stromal cell components of the breast parenchyma. In contrast, primary breast lymphoma (PBL) arises from breast lymphoid tissue and is characterised by neoplastic B or T cells and is therefore classified according to World Health Organisation (WHO) diagnostic criteria for non-Hodgkin lymphomas (NHL). Unlike breast cancer and some other nodal or extra-nodal NHL, PBL is rare accounting for just 0.04–1% of malignant breast neoplasms and less than 1% of NHL.2
PBL is diagnosed based on criteria proposed by Wiseman and Liao, which state that the primary site of lymphoma presentation should be the breast, the lymphoma should be in close association with the breast tissue, and the patient should have no history or evidence of disseminated disease within 6 months of diagnosis, whilst the simultaneous involvement of axillary lymph nodes is considered to be the part of the spectrum of PBL.3 Of note, these criteria exclude secondary breast lymphomas, i.e., those which arise elsewhere but manifest in the breast as part of secondary involvement. PBL can present as one of many different histological variants. Over 90% of reported PBL cases are of a B cell origin, of which over 50% are diagnosed as diffuse large B cell lymphomas (DLBCL), whilst follicular B cell lymphoma (FL), extra-nodal marginal zone lymphoma (MZL) and Burkitt lymphoma (BL) are amongst the less common B cell variants.4, 5, 6 The predominant PBL of a T cell origin is anaplastic large cell lymphoma (ALCL) which has been increasing in prevalence in the last 2 decades. Whilst not exclusively arising in the breast parenchyma, it is found surrounding textured breast implants and is usually confined by a periprosthetic fibrous capsule.6
Each histological variant of PBL seems to have a distinct epidemiology, prognosis and treatment requirement, and therefore each must be accurately diagnosed to enable timely administration of the most appropriate treatment. This is especially important as PBL is also easily misdiagnosed as breast carcinoma due to the fact that both often present as a painless, apparently innocuous lump and lack distinct imaging characteristics.7
In this review, the different types of PBL are discussed, focusing on breast implant-associated ALCL (BIA-ALCL), with reference to the clinical presentation, histological and immunohistochemical features, as well as treatment options available for each diagnostic entity. In compiling this review, our methodology included the review of case reports and small case series; large retrospective studies are lacking in the study of PBL due to the rarity of these neoplasms.
Primary breast lymphoma of a B cell origin
PBL arising from B cells accounts for up to 90% of all PBL cases and predominantly consists of the DLBCL, FL, MZL and BL diagnostic subtypes (Table 1). Rarer histological variants of B cell PBL include precursor B cell lymphoblastic lymphoma,4 classical Hodgkin lymphoma4 and Epstein–Barr virus (EBV)-positive Hodgkin lymphoma of mixed cellularity type,8 although these latter forms are not discussed in this review.
Table 1.
Incidences of PBL of a B cell origin according to individual case series
| PBL subtype | Study-specific prevalence amongst PBL cases analysed per study | Study references |
|---|---|---|
| Diffuse large B cell lymphoma | 64% of 50 cases | 4 |
| 76.6% of 30 cases | 5 | |
| 39.6% of 1,304 cases* | 6 | |
| Follicular lymphoma | 10% of 30 cases | 5 |
| 15.9% of 1,304 cases* | 6 | |
| Marginal zone lymphoma | 28% of 50 cases | 4 |
| 6.7% of 30 cases | 5 | |
| 24.7% of 1,304 cases* | 6 | |
| Burkitt lymphoma | 2% of 50 cases | 4 |
These studies also included systemic/nodal lymphoma presentations in their cohorts
Primary Breast Diffuse Large B Cell Lymphoma
Primary breast diffuse large B cell lymphoma (PB-DLBCL) is the most common presentation of PBL (Table 1) and is one of the more aggressive histological subtypes, often presenting as a rapidly enlarging breast lump, sometimes accompanied by multiple breast masses, diffuse breast enlargement and/or enlarged axillary lymph nodes.9 Systemic type-B symptoms including weight loss, fatigue and fever can also occur in some cases.10 The causes of PB-DLBCL remain unclear, although post-menopausal oestrogen levels,11 chronic inflammatory and autoimmune diseases12 and pregnancy and lactation 13 have been proposed as risk factors.
PB-DLBCL is histologically characterised by the presence of a diffuse infiltrate of large neoplastic B cells with round to oval or lobated nuclei and frequent prominent nucleoli (Figure 1A). Immunohistochemically, neoplastic cells are positive for B cell markers such as CD20 (Figure 1B), CD79α and/or PAX5 and have either a germinal centre-like phenotype positive for CD10 and BCL6 or a non-germinal centre-like phenotype of CD10 negative and MUM1 positive.7 The non-germinal centre phenotype is more common in PBL.5 The PB-DLBCL proliferation index detected with Ki-67 is usually high, commonly >80% 7,9,13,14 (Table 2).
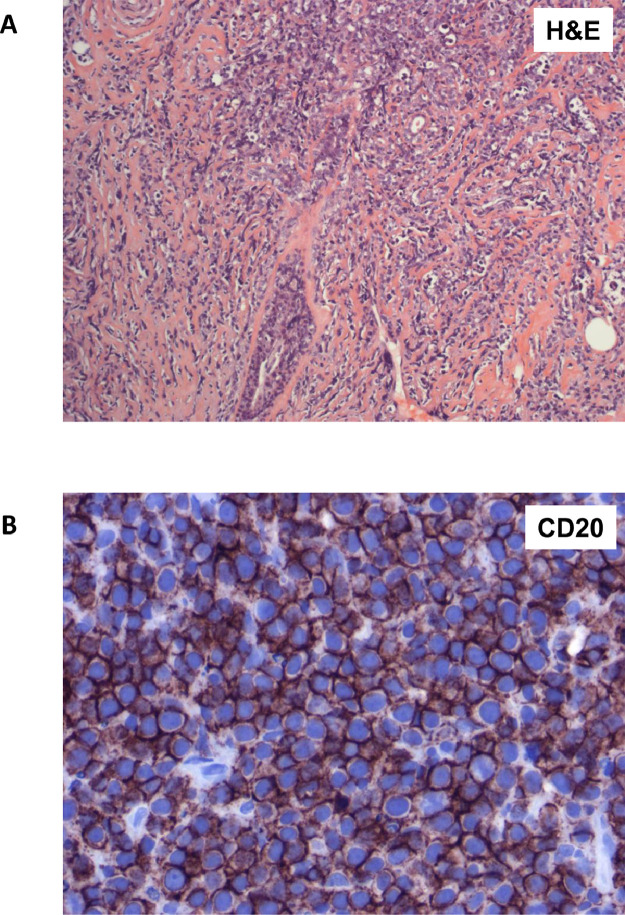
Figure 1.
Primary breast diffuse large B cell lymphoma. (A) H&E stain of breast ducts infiltrated by large cells with round to oval or lobated nuclei with vesicular chromatin. (B) Immunohistochemistry for the B cell marker CD20 demonstrates that most tumour cells are positive.
Table 2.
Immunohistochemical markers described in association with a PBL diagnosis of a B cell origin
| PBL B cell subtype | Immunohistochemical markers | References |
|---|---|---|
| DLBCL | CD20, BCL6, BCL2, MUM1, C-MYC, HGAL, CD79α, PAX5, CD10, LCA, CD19, Ki-67 (80–100%) | 7,9,10,12,13,14 |
| FL | CD20, CD10, BCL6, BCL2, CD79α, p53, Ki-67 (10–40%) | 16,18,19,20,21 |
| MZL | CD20, BCL2 | 22,23 |
| BL | CD20, BCL6, CD79α, MUM1, PAX5, CD43, CD10, CD19, HLA-DR, Ki-67 (100%) | 27,29,30,31 |
Owing to its aggressive nature, PB-DLBCL requires systemic treatments such as chemotherapy, radiotherapy and immunotherapy. Interestingly, in contrast with breast parenchyma-derived neoplasms, surgery and mastectomy seem to be associated with higher rates of recurrence and poorer prognosis.9 The most frequently administered treatment protocol for PB-DLBCL is six cycles of R-CHOP, consisting of rituximab, a monoclonal antibody targeting CD20 which acts to deplete CD20-positive cells and a regimen of four chemotherapy drugs: cyclophosphamide, doxorubicin, vincristine and prednisone. This is often followed by consolidative radiation therapy and/or five doses of intrathecal methotrexate as a prophylactic measure for central nervous system metastasis9,10,14,15.
Primary Breast Follicular Lymphoma
Primary breast follicular lymphoma (PB-FL) is less common and less aggressive than PB-DLBCL, and as such fewer case reports and case series exist. PB-FL often presents as an enlarging breast mass16 in the absence of type-B symptoms.17 Interestingly, PB-FL has been reported to co-occur with other breast malignancies; at least one case of PB-FL co-occurring with bilateral lobular breast carcinoma has been documented,18 and there have been multiple reports of secondary breast lymphomas co-occurring with a variety of breast cancers (reviewed in 19). The detailed pathophysiology of these co-occurring malignancies is unknown.
PB-FL is thought to arise from follicular B cells in breast lymphoid tissue and is histologically distinct from the other PBL variants due to the presence of small follicles composed of atypical small centrocytes with cleaved nuclei20 (Figure 2A, B). Immunohistochemically, PB-FL expresses the germinal centre markers CD10 and BCL6, as well as the anti-apoptotic protein BCL2 16,18, 19, 20 (Table 2), although some cases are CD10 negative.21 The proliferation index of PB-FL is much lower than PB-DLBCL, at around 10–40% Ki67 positivity in keeping with its less aggressive clinical presentation.20
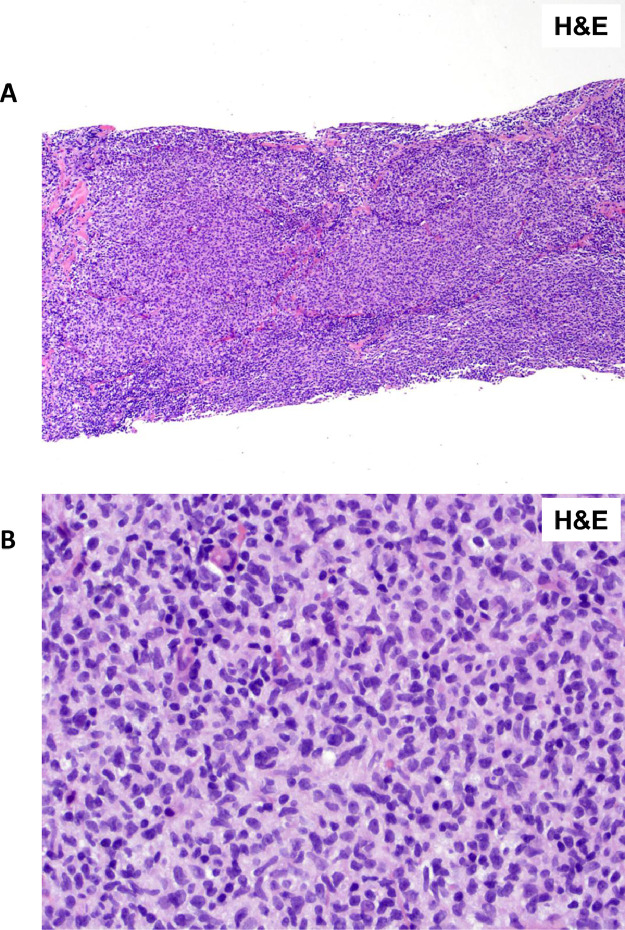
Figure 2.
Primary breast follicular lymphoma. (A) H&E stain of breast parenchyma infiltrated by ill-defined neoplastic follicles. (B) H&E stain of the neoplastic follicles which are composed predominantly of small centrocytes, supportive of a diagnosis of follicular lymphoma grade I.
PB-FL mostly has an excellent prognosis following treatment; a retrospective study of 36 patients with PB-FL showed an overall response rate of 97%.17 This study also found that radiation therapy alone, or in combination with surgery, was associated with a reduced risk of relapse compared to that of surgery alone.17 This has since been supported by others; in one case report, low-grade PB-FL was successfully treated with radiation therapy,21 and in another, high-grade PB-FL was successfully treated with radiation therapy in combination with Rituximab-Cyclophosphamide, hydroxydaunorubicin, oncovin, prednisone (R-CHOP).20
Primary Breast Extra-Nodal Marginal Zone Lymphoma
Primary breast extra-nodal marginal zone lymphoma (PB-MZL) has a similar incidence and prognosis to PB-FL but is more indolent.17 Although PB-MZL can present as a painful palpable mass,22 many cases are asymptomatic and are only identified as focal asymmetries on mammogram and/or ultrasound.23,24
PB-MZL is thought to arise from the marginal zone of reactive lymphoid follicles. It presents histologically as a vaguely nodular growth pattern (Figure 3A) of small atypical cells with moderately abundant pale cytoplasm and irregularly shaped nuclei, which are known as monocytoid lymphocytes22 (Figure 3B). Immunohistochemically, the defining feature of PB-MZL is the almost exclusive absence of CD10; PB-MZL stains positive for CD20 and BCL2 and has a low Ki67 proliferation index22,23 (Table 2). However, CD21, CD35 or CD23 highlight underlying remnants of follicular dendritic cell mesh-works suggestive of adjacent germinal centres (Figure 3C). PB-MZL has also been reported to harbour various chromosomal translocations involving the MALT1 gene, identifiable by fluorescent in situ hybridisation (FISH).25
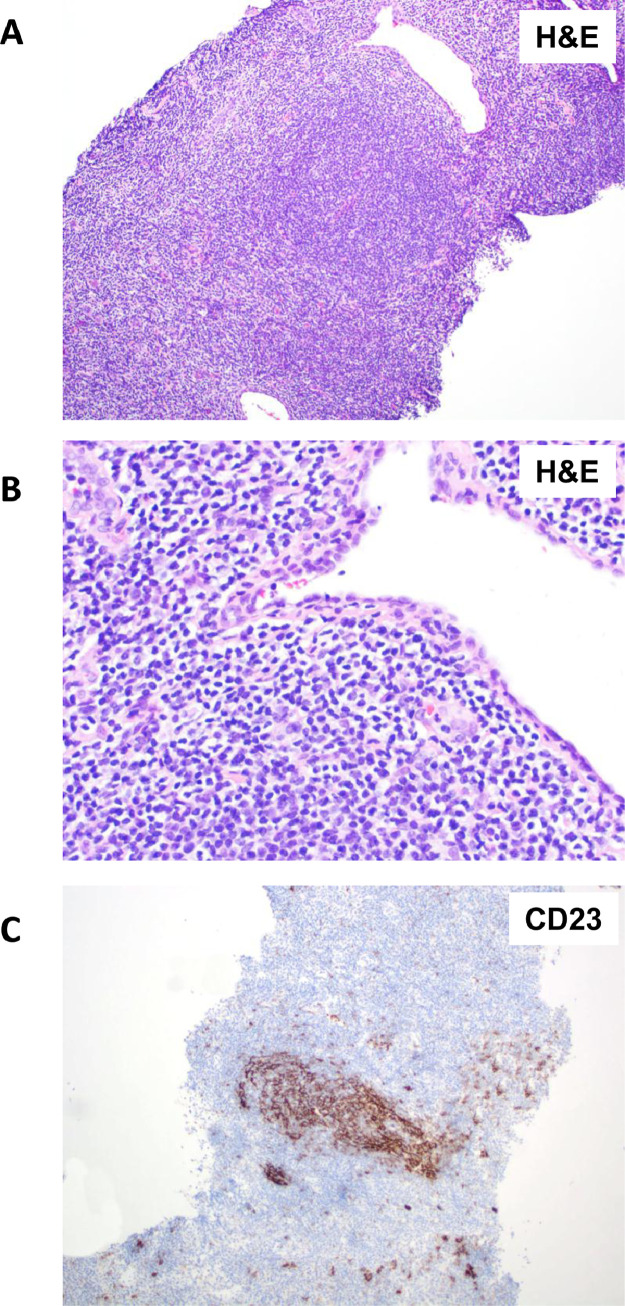
Figure 3.
Primary breast extra-nodal marginal zone lymphoma. (A) H&E stain of breast parenchyma infiltrated by a vaguely nodular infiltrate. (B) H&E stain of small to intermediate size lymphocytes known as monocytoid B cells with irregular nuclear contours and moderately abundant clear cytoplasm. (C) CD23 immunohistochemistry staining highlights remnants of follicular dendritic cell mesh-works, which is characteristic of all extra-nodal MZL.
Owing to its indolent nature, most of the PB-MZL can be treated by lumpectomy or localised radiation therapy.4 In some cases, women may opt for active surveillance. Interestingly, a recent case report detailed a woman who, due to the asymptomatic nature of her disease, opted for active surveillance even though the malignancy was at an advanced stage involving both breasts, spleen and bone marrow. However, the authors noted that irradiation and/or rituximab would be considered for this patient if local and/or systemic symptoms should occur.23 Furthermore, there have been cases where intervention with therapy was required. For example, one case report described a patient who opted for active surveillance but developed multiple new breast masses one year after her initial diagnosis which required the administration of rituximab monotherapy until a complete response was achieved.26 In another patient, 6 months after a low-grade PB-MZL was diagnosed, a high-grade PB-DLBCL developed; R-CHOP and consolidative radiotherapy were then administered,26 which may not have been required if the PB-MZL had been treated earlier. However, there is no method to determine which PB-MZL cases will progress to PB-DLBCL; this is also the case for MZL not associated with the breast and its transformation to DLBCL.
Primary Breast Burkitt Lymphoma
Primary breast Burkitt lymphoma (PB-BL) is one of the rarest histological variants of PBL, although its growth is extremely rapid and aggressive and can be fatal if not diagnosed early and treated appropriately.27 PB-BL commonly presents as a diffuse bilateral and rapid enlargement of the breasts.28 Pain, redness, itching, fever and axillary lymphadenopathy are also experienced by some patients.29 PB-BL mostly affects young women of childbearing age (the range is 12–36 years27) and cases often present in pregnancy28 or during lactation in the post-partum period.29,30 There have been reports, albeit very rarely, of males developing PB-BL; one case described a male with a history of basal cell carcinoma and spironolactone (anti-androgen) treatment presenting with high-grade PB-BL.31 Interestingly the common denominator in these cases is a history of a significant hormonal alteration, thus suggesting a role for hormonal changes in the development of PB-BL.
PB-BL is considered to arise from mature B cells. It presents histologically as a diffuse infiltrate of medium-sized B cells, with a uniform growth pattern, which forms a characteristic “starry sky” appearance27 (Figure 4A). Immunohistochemically, PB-BL is positive for CD20, as well as for the germinal centre markers CD10 and BCL6 27,30,31 (Table 2) and are almost exclusively 100% positive for Ki67 (Figure 4B), highlighting the high proliferative capacity of PB-BL.30 Both this high proliferation index and the presence of a chromosomal breakage and translocation of the MYC locus in over 90% of PB-BL cells 27,30 are characteristic of all forms of BL, not just those localised to the breast.
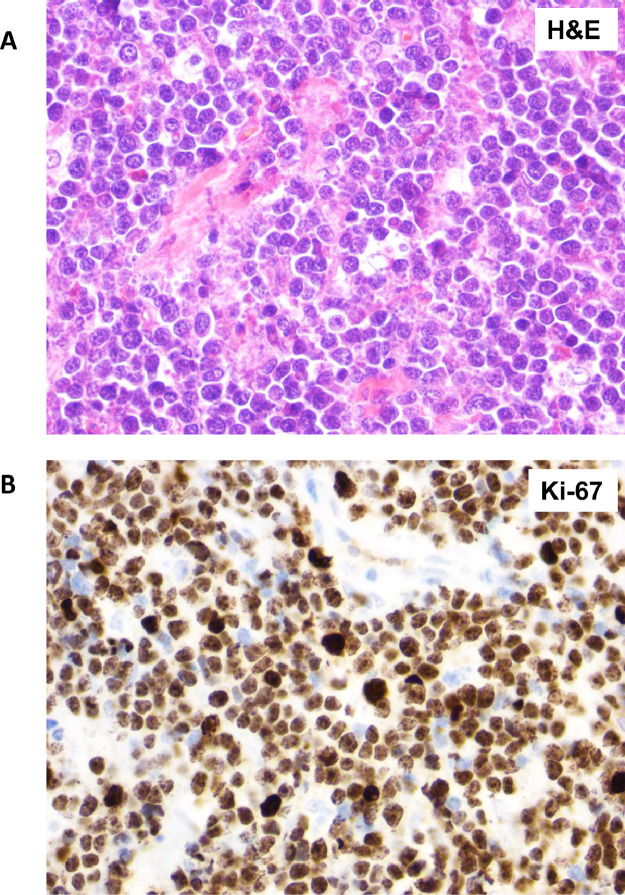
Figure 4.
Primary breast Burkitt lymphoma. (A) H&E stain of the breast parenchyma replaced by a diffuse infiltrate of intermediate size lymphoma cells associated with prominent phagocytic histiocytes that impart a “starry-sky” appearance as well as frequent mitoses and karyorrhexis. (B) Ki-67 immunohistochemistry staining of almost 100% of lymphoma cells indicates that almost all cells are in the process of cell division.
Successful PB-BL treatment outcomes have been achieved with bilateral mastectomy followed by adjuvant chemotherapy,27 although due to the aggressive nature of PB-BL, most cases require an intensive chemotherapy regimen. Furthermore, complications can arise when PB-BL arises during pregnancy. For example, one case report describes a woman with aggressive and late-stage PB-BL who suffered a miscarriage following a reduced-intensity chemotherapy regimen and consequently underwent aggressive chemotherapy combined with rituximab and methotrexate to achieve complete remission.28 PB-BL can also be fatal; in one case report, a patient progressed through the bleomycin, cyclophosphamide, doxorubicin, vincristine and prednisolone (BACOP) chemotherapy protocol, and despite being given further cycles of the modified chemotherapy regimen, CODOX- cyclophosphamide, oncovin, doxorubicin, methotrexate, ifosfamide, Vepesid and Ara-C (CODOX-M/IVAC) plus rituximab, the patient died shortly after.30 Another case report described a patient that relapsed 5 months after chemotherapy also with CODOX-M/IVAC and later died from treatment complications and sepsis.29 A detailed summary of the treatment options for all PBL of a B cell origin is described in Figure 5.
Figure 5.
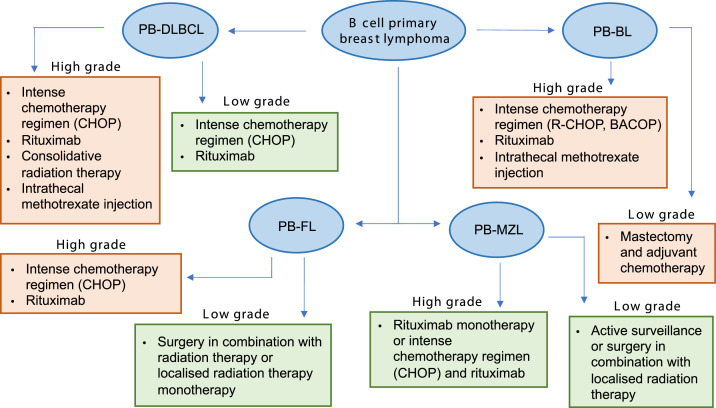
Treatment options for primary breast lymphomas of a B cell origin. PB-DLBCL: primary breast-diffuse large B cell lymphoma. PB-FL: primary breast-follicular lymphoma. PB-MZL: primary breast-marginal zone lymphoma. PB-BL: primary breast-Burkitt lymphoma. Green boxes represent subtypes with a relatively good prognosis following treatment, and orange boxes represent subtypes with a worse prognosis following treatment.
Primary breast lymphoma of a T cell origin
Primary breast-ALCL (PB-ALCL) is the most common T cell-derived PBL.6 Other T cell lymphomas including peripheral T cell lymphoma and follicular T cell lymphoma32 have been described in the breast, but as they are exceptionally rare, only PB-ALCL will be discussed in this section.
Most cases of PB-ALCL arise in association with textured breast implants (Section 4); less than 50 cases of ALCL arising in the breast in the absence of implants have been reported. Like systemic/nodal ALCL, PB-ALCL is characterised by the presence or absence of aberrant anaplastic lymphoma kinase (ALK) expression and activity which gives rise to both ALK-positive (ALK+) and ALK-negative (ALK-) ALCL entities. Of the ALCL cases arising in the absence of breast implants (PB-ALCL), both ALK+ and ALK- presentations have been reported, including cases of the more indolent ALK- primary cutaneous ALCL (pcALCL).
In general, PB-ALCL, like systemic/nodal ALCL are characterised by the presence of large pleomorphic “hallmark” cells with irregular nuclear contours, enlarged and misshapen nuclei and prominent Golgi bodies. Immunohistochemically, PB-ALCL almost invariably expresses CD30, as well as an unusual variety of cell markers (Table 3). Owing to the frequent cell surface expression of mature T cell characteristic proteins, such as CD4, CD8 and CD45RO, and the presentation of ALCL outside of the thymus, ALCL is thought to arise from mature peripheral T cells.33 Interestingly, although ALCL cases mostly harbour T cell receptor (TCR) rearrangements at the molecular level, confirming their T cell origin, not all ALCL express the CD3/TCR signalling complex on their surface.34,35
Table 3.
Immunohistochemical markers described in association with PB-ALCL
| PB-ALCL sub-type | Immunohistochemical markers | References |
|---|---|---|
| ALCL, ALK+ | CD30, CD5, EMA, ALK, MUM1-IRF4, CD31, BCL6, CD4, CD43, TIA-1, granzyme B, CD45RB, CD45RO, TCRβF1 | 38,40,41,44 |
| ALCL, ALK- | CD30, CD3, CD43, granzyme B, CD4, CD5, BCL2, MUM1 | 50,54 |
| BIA-ALCL | CD30, CD4, CD2, MUM1-IRF4, CD7, CD45, granzyme B, EMA, CD5, CD43, CD56, TIA-1 | 55,67,68,71,77,93,94 |
Primary Breast Anaplastic Large Cell Lymphoma, ALK-positive (PB-ALCL, ALK+)
Primary breast-ALCL (PB-ALCL), ALK+ is characterised by the presence of the t(2;5)(p23;q35) translocation juxtaposing the tyrosine kinase encoding domain of ALK to the nucleolar protein gene NPM1, resulting in the constitutive activation of ALK signalling36 and the upregulation of signalling pathways associated with cell proliferation and survival.33,37 Systemic/nodal ALCL, ALK+ mostly affects the paediatric population; this is also true for most cases of PB-ALCL, ALK+, although cases arising in pregnant women have also been reported.38 Systemic/nodal ALCL, ALK+ primarily presents within the lymph nodes,39 whilst PB-ALCL, ALK+ usually presents as an enlarging breast lump;40 both often occur in combination with type-B symptoms and axillary lymph node and/or extra-nodal involvement.39,41
PB-ALCL, ALK+ has variable treatment outcomes. For example, one patient was successfully treated with surgery and Cyclophosphamise, Hydroxydaunorubicin, Oncovin, Prednisone (CHOP) chemotherapy,40 and another case demonstrated an “excellent response” with cyclophosphamide, doxorubicin and prednisone (CHP) chemotherapy in combination with brentuximab vedotin (BV), even with antrum and bone marrow involvement.41 BV is an antibody-drug conjugate consisting of a monoclonal antibody targeting CD30 linked to the cytotoxic compound monomethyl auristatin E (MMAE).42 However, there are cases where PB-ALCL, ALK+ has been fatal; this mainly includes cases in young adolescents where systemic spread occurred, despite the intensive chemotherapeutic regimens used.43, 44, 45
Primary Breast Anaplastic Large Cell Lymphoma, ALK-negative (PB-ALCL, ALK-)
Systemic/nodal ALCL, ALK- comprises a heterogenous group of NHL which have a relatively poorly defined genetic profile, and in contrast to ALCL, ALK+, generally affect an older population.46,47 Based on the site of presentation, ALCL, ALK- can be classified into systemic/nodal, primary cutaneous and breast implant-associated (BIA) subtypes. Systemic/nodal ALCL, ALK- can be further classified based on the presence or absence of specific genetic abnormalities; for example, DUSP22 and TP63 rearrangements were found to be present in 30% and 8% of systemic ALCL, ALK- cases, respectively, and may serve as predictive biomarkers for treatment outcomes.48 Furthermore, JAK1 and/or STAT3 mutations have been reported in other systemic/nodal ALCL, ALK- cases,49 and it is likely that further subclassifications will be assigned as more genetic abnormalities are identified.46
Systemic/nodal ALCL, ALK- involves the lymph nodes, bone marrow and/or extra-nodal organs and has a relatively poor prognosis; thus, treatment often requires intensive chemotherapy regimens.46 There have been limited cases of systemic ALCL, ALK- presenting primarily within the breast (PB-ALCL, ALK-). Interestingly, cases of PB-ALCL, ALK- arising in a patient with a breast burn cicatrix,50 and in a patient with a history of localised ALCL, ALK-45 were recently reported. These case reports suggest that chronic inflammation may play a role in the development of these malignancies.50 Both cases responded well to treatment with BV and cyclophosphamide, doxorubicin, etoposide and prednisone (CHEP) chemotherapy50 or CHOP chemotherapy alone.45
pcALCL is a more indolent form of ALCL, ALK- and most commonly presents as single or multiple localised skin nodules.47 Whilst most cases of pcALCL do not express ALK, a handful of ALK-expressing pcALCL cases have been reported.51,52 Early-stage pcALCL has a relatively good prognosis with most cases treatable with surgery,43 and some cases have been reported in which the tumour spontaneously regresses after biopsy.53 Primary breast-pcALCL (PB-pcALCL) has been reported rarely in the literature; we found one case report of a 70-year-old woman who developed a single breast lesion, accompanied by skin discolouration, who was treated successfully with surgery and was followed up with active surveillance.54
Breast implant-associated lymphoma
Lymphoma has been reported for breasts in which breast implants are present. Indeed, ALCL (BIA-ALCL) is more common in this context, rather than when occurring in a breast without an implant (PB-ALCL). In this section, the different types of BIA lymphoma are discussed, with a focus on BIA-ALCL and its clinical presentations, treatment options, epidemiology and aetiology.
Breast Implant-Associated Anaplastic Large Cell Lymphoma
BIA-ALCL Clinical Presentation
BIA-ALCL is an ALK- ALCL that arises in the context of macrotextured breast implants. BIA-ALCL has been reported both in patients who have undergone breast augmentation for cosmetic purposes and reconstruction purposes after mastectomy.55 The median time to diagnosis is roughly 7–10 years after initial breast implantation.56,57 To date, no cases have been diagnosed in patients with an exclusive history of smooth implants, although there have been cases associated with smooth implants preceded by textured tissue expanders58 suggesting that BIA-ALCL pathogenesis could be related to a component of the implant texturing process.
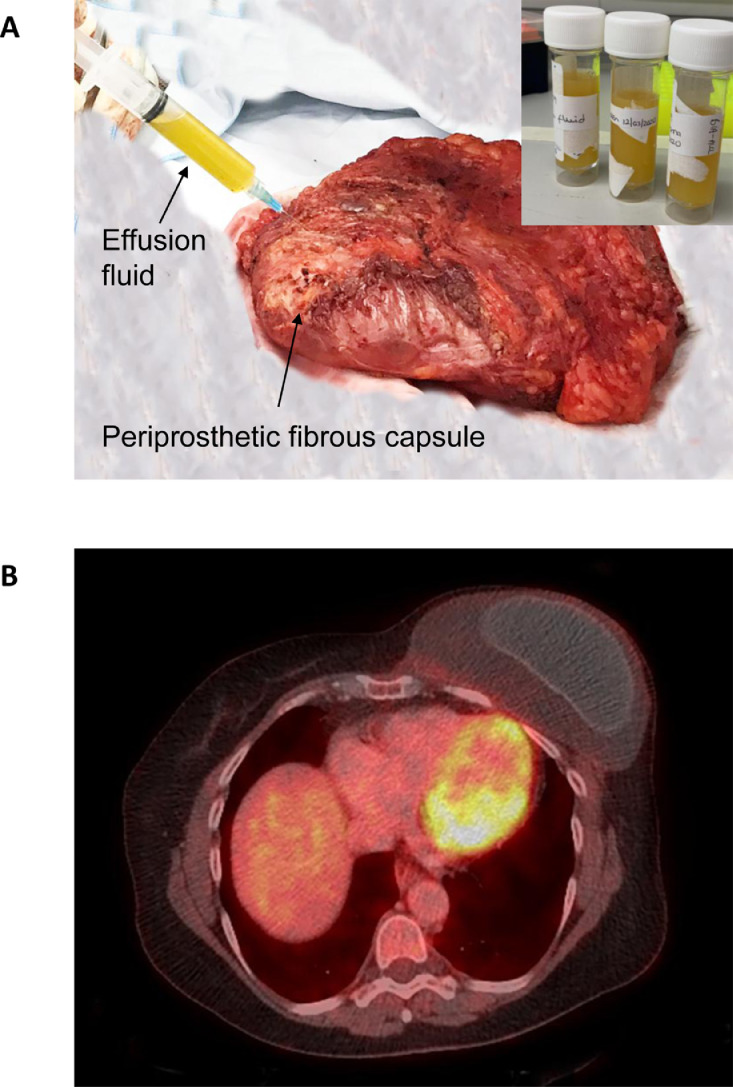
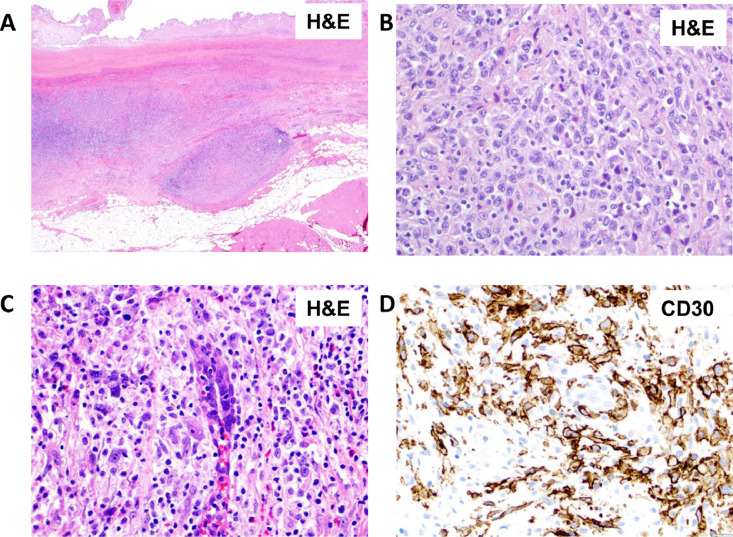
BIA-ALCL presents as a rapid and persistent unilateral or bilateral breast swelling, often along with breast discomfort and rarely with type-B symptoms.59 In most cases, BIA-ALCL presents as a yellowish effusion fluid that is found between the breast implant and periprosthetic fibrous capsule (Figure 6A) and can be detected by imaging techniques such as ultrasound computed tomography or positron emission tomography-computed tomography (PET-CT) (Figure 6B). The effusion fluid is yellow in colour and opaque due to extensive tumour cell necrosis. This can also be accompanied by one or several solid masses penetrating the capsule (clinical stage I according to the TNM staging system; as shown in Figure 7).60 In some cases, the lymphoma cells infiltrate beyond the fibrous capsule and invade the surrounding soft tissue (stage IIA; as shown in Figure 8A, B). In more aggressive cases, the lymphoma cells infiltrate surrounding breast ducts (Figure 8C, D). Roughly, 15% of BIA-ALCL cases show lymph node involvement;60 stage IIB describes the involvement of a single regional lymph node; stage III describes the involvement of multiple regional lymph nodes; and stage IV describes the disseminated spread of BIA-ALCL to multiple lymph nodes and organs60 (Figure 7). Whilst being uncommon, metastases to the liver, small bowel and bone have been observed.55
Figure 6.
Breast implant-associated anaplastic large cell lymphoma. (A) Gross appearance of an en bloc resection of a case with effusion suspicious of BIA-ALCL. One of the initial steps is to aspirate any peri-implant effusion surrounding a breast implant. The fluid (inset) is yellowish and opaque due to abundant cell debris and necrosis. (B) Positron emission tomography-computed tomography (PET-CT) shows a distorted implant surrounded by an effusion that is contained by a fibrous capsule.
Figure 7.
Breast implant-associated anaplastic large cell lymphoma clinical classifications according to the TNM staging system. Adapted from 55.
Figure 8.
Breast implant-associated anaplastic large cell lymphoma invading beyond the peri-implant fibrous capsule. (A) Neoplastic nodules are noted invading beyond the capsule into peri-implant fibrous tissue. (B) Lymphoma cells invading beyond the capsule. (C) Lymphoma cells invading a breast duct. (D) CD30 immunohistochemistry highlights the large neoplastic cells permeating along breast ducts.
Patients with stage I BIA-ALCL generally have an excellent prognosis with overall survival rates reaching 100%. However, cases presenting with lymph node involvement (stage IIB-IV) are associated with reduced overall survival and can be fatal in some circumstances.61 Fatalities have also occurred without lymph node involvement, when the lymphoma cells infiltrated through the chest wall, pleural space and mediastinum62.
BIA-ALCL Treatment
Treatment options for BIA-ALCL depend on the stage of the disease at diagnosis (Figure 9). For stage I disease, as the malignancy is localised to the capsule, treatment usually involves draining the effusion fluid followed by full bilateral capsulectomy with implant removal.(en bloc resection)55 Patients who do not undergo implant removal are more likely to experience persistent disease or progression, even if chemotherapy is administered.60,63 Furthermore, it is important to remove all of the fibrous capsule as the remaining capsule, and scar tissue has been associated with disease recurrence.58
Figure 9.
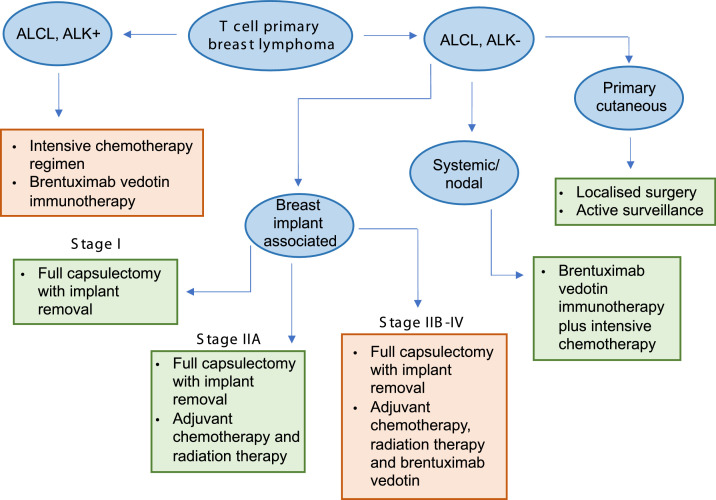
Treatment options for primary breast lymphomas of T cell origin. ALCL, ALK+: anaplastic large cell lymphoma, anaplastic lymphoma kinase positive. ALCL, ALK-: anaplastic large cell lymphoma, anaplastic lymphoma kinase negative. Green boxes represent subtypes with a relatively good prognosis following treatment, and orange boxes represent subtypes with a worse prognosis following treatment.
For stage IIA-IV disease, systemic treatments are required including radiotherapy, chemotherapy and the administration of immunotherapeutic agents such as BV. For patients with invasion beyond the fibrous capsule (stage IIA), successful treatment outcomes have been achieved with full capsulectomy in combination with radiotherapy64 or chemotherapy and radiotherapy65, 66, 67. The successful use of BV has been reported when a patient progressed through chemotherapy65 and prior to capsulectomy, in order to avoid more complicated and risky surgery involving the sub-pectoral muscles and ribs.68
For patients with lymph node involvement (stage IIB-IV), full tumour regression has been achieved with full capsulectomy, lymph node dissection plus post-operative chemotherapy such as CHOP 55 or localised radiation therapy to the lymph nodes.69 Chemotherapy has also been given prior to capsulectomy in an attempt to reduce the tumour load before surgery and negate the requirement for lymph node dissection.70 One case report described the successful use of 6 cycles of BV immunotherapy to treat BIA-ALCL which had invaded into the breast tissue and spread to the axillary lymph node.71 BV may also be used in patients who have a history of breast cancer as repeated anthracycline-based therapy is not recommended in these cases.55
BIA-ALCL Risk and Epidemiology
Whilst the first case of BIA-ALCL was reported in 1997,72 BIA-ALCL was only recognised as a distinct disease entity by the WHO in 2016.73,74 As of May 2021, there were 994 BIA-ALCL cases and 37 BIA-ALCL-related deaths reported worldwide,75 including cases seen for the first time in Thailand, Japan and Taiwan.67,76,77 However, this is likely an underestimation and it is possible that many cases have gone undiagnosed once the disease is disseminated and/or are not reported, perhaps more recently due to the COVID-19 pandemic. In this vein, the total number of BIA-ALCL cases worldwide is difficult to determine due to (i) a lack of information regarding the number of people with breast implants in situ and (ii) a limited awareness of BIA-ALCL by surgeons and patients leading to large discrepancies in the reporting of cases.78 This is likely exacerbated by the fact that BIA-ALCL has a long latency of around 5 to 10 years and can present with a wide variety of symptoms. As such, the absolute risk of developing BIA-ALCL is the subject of much debate. De Jong and co-workers were the first to identify an increased risk of developing BIA-ALCL in the context of textured implants; in 2008, they reported the absolute risk to be 0.1 to 0.3 per 100,000 women with implants per year.79 Since then, risk values ranging from 1 in 355 patients80 to 1 in 13,745 patients81 have been reported. These discrepancies could be due to factors such as differences in patient follow-up time, the size of the patient cohort selected for the retrospective study, patient susceptibility, the reason for breast implantation, the texturing of the implant and case collection methodology. As such, well-designed prospective studies are required to properly assess the absolute risk of BIA-ALCL.
BIA-ALCL Aetiology
The aetiology of BIA-ALCL is not very well understood. Multiple groups have suggested that a component of the implant texturing process may cause chronic inflammation which eventually leads to T cell hyperplasia and the eventual emergence of BIA-ALCL. This component has been suggested to be (i) small silicone particulates which are shed from the implant surface,82 (ii) a bacterial biofilm that manifests only on the surface of textured implants,83,84 (iii) toxic chemicals that leach through the implant shell85 and (iv) a chemical on the surface of textured implants which is recognised as a foreign antigen, an adjuvant or acts as a carcinogen.86 Others have also suggested a role for an allergic response to the breast implant82 and mechanical friction between the textured implant and the fibrous capsule85 in disease pathogenesis. Furthermore, it is likely that genetic susceptibility may play a role in BIA-ALCL development; germline BRCA1/2 mutations, mutations in the JAK3 pathway55 and specific germline HLA variants87 have all been associated to some extent with BIA-ALCL. However, many of the reported biological and genetic studies have been conducted with limited sample numbers and thus lack statistical power. We believe that better reporting and collection of patient samples may enable deeper insights into the biological mechanisms surrounding BIA-ALCL pathogenesis to be elucidated in the future.
Other Breast Implant-Associated Lymphomas
Whilst the majority of BIA malignancies are ALCL arising in the space between the breast implant and fibrous capsule, other less common presentations have been noted. For example, there have been cases of pcALCL arising on the skin overlying the breast in women with textured breast implants.88,89 However, a definitive causal relationship between pcALCL and breast implants has yet to be established. There have also been reports of B cell lymphomas arising in the context of textured breast implants, including cases of MZL, DLBCL and FL,90 as well as a case of bilateral chronic lymphocytic leukaemia (CLL)/small lymphocytic lymphoma 91 and multiple cases of EBV+ large B cell lymphoma.92 The clinical presentation of these can vary compared to BIA-ALCL, whereby thick fibrous capsules and more prominent layering of neoplastic cells were observed in the EBV+ large B cell lymphoma cases.92 However, interestingly, the majority of the B cell-associated cases had strikingly similar clinical presentations to BIA-ALCL, including primary presentation as solid masses attached to the fibrous capsule. Furthermore, successful treatment outcomes were achieved with full capsulectomy and implant removal, as well as R-CHOP and methotrexate treatment in some cases, thus suggesting that pathogenesis in these cases may also be specific to the textured breast implant. However, due to the limited number of B cell lymphomas arising in the context of breast implants, it is difficult to establish a causal relationship at present, and it remains to be elucidated whether the cause of breast implant-associated lymphoma in these incidences is distinct from that of BIA-ALCL.
Conclusions
Overall, primary lymphomas of the breast present with a variety of clinical manifestations with varying histological and immunohistochemical features. Each subtype has a different prognosis and thus requires a distinct treatment regimen. It is important to distinguish between the different PBL histological presentations in order to promptly administer the correct therapy and maximise the chances of treatment success.
Disclosures
Conflict of interest: SDT receives research funding from Allergan
Funding: None
Ethical approval: Not applicable
References
- 1.World Health Organisation. Breast cancer. https://www.who.int/news-room/fact-sheets/detail/breast-cancer [Accessibility verified November 3, 2021].
- 2.Moura C, Leite MI, Parreira R, Medeiros A. Primary breast lymphoma. J Surg Case Reports. 2020;2020(1):1–3. doi: 10.1093/jscr/rjz405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wiseman C, Liao KT. Primary lymphoma of the breast. Cancer. 1972;29(6):1705–1712. doi: 10.1002/1097-0142(197206)29:6<1705::aid-cncr2820290640>3.0.co;2-i. [DOI] [PubMed] [Google Scholar]
- 4.Talwalkar SS, Miranda RN, Valbuena JR, Routbort MJ, Martin AW, Medeiros LJ. Lymphomas involving the breast: A study of 106 cases comparing localized and disseminated neoplasms. Am J Surg Pathol. 2008;32(9):1299–1309. doi: 10.1097/PAS.0b013e318165eb50. [DOI] [PubMed] [Google Scholar]
- 5.Shao YB, Sun XF, He YN, Liu CJ, Liu H. Clinicopathological features of thirty patients with primary breast lymphoma and review of the literature. Med Oncol. 2015;32(2):1–6. doi: 10.1007/s12032-014-0448-z. [DOI] [PubMed] [Google Scholar]
- 6.Thomas A, Link BK, Altekruse S, Romitti PA, Schroeder MC. Primary Breast Lymphoma in the United States: 1975–2013. JNCI J Natl Cancer Inst. 2017;109(6) doi: 10.1093/jnci/djw294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Uenaka N, Yamamoto S, Sato S, et al. Primary breast lymphoma initially diagnosed as invasive ductal carcinoma: A case report. Clin Case Reports. 2021;9(6) doi: 10.1002/ccr3.4189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Marton I, Hrgović Z, Habek D. Primary hodgkin's lymphoma of the breast initially treated by surgical excision and axillary dissection. Acta Clin Croat. 2020;59(2):365. doi: 10.20471/acc.2020.59.02.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen M, Zhou J, Qu X. Primary Breast Diffuse Large B-Cell Lymphoma in a 42-Year-Old Female: A Case Report and Review of Literature. J Med Cases. 2021;12(5):181. doi: 10.14740/jmc3660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yoneyama K, Nakagawa M, Hara A. Primary lymphoma of the breast: A case report and review of the literature. Radiol Case Reports. 2021;16(1):55. doi: 10.1016/j.radcr.2020.10.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Teras LR, Patel A.V., Hildebrand JS, Gapstur SM. Postmenopausal unopposed estrogen and estrogen plus progestin use and risk of non-Hodgkin lymphoma in the American Cancer Society Cancer Prevention Study-II Cohort. Leuk. Lymphoma. 2012;54(4):720–725. doi: 10.3109/10428194.2012.722216. [DOI] [PubMed] [Google Scholar]
- 12.Shen F, Li G, Jiang H, Zhao S, Qi F. Primary breast diffuse large B-cell lymphoma in a patient with systemic lupus erythematosus: A case report and review of the literature. Medicine (Baltimore) 2020;99(33):e21736. doi: 10.1097/MD.0000000000021736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yin Y, Ye M, Chen L, Chen H. Giant primary breast diffuse large B-cell lymphoma during pregnancy and lactation. Breast J. 2019;25(5):996–997. doi: 10.1111/tbj.13403. [DOI] [PubMed] [Google Scholar]
- 14.Villalón-López JS, Souto-del Bosque R, Méndez-Sashida PG. Linfoma no Hodgkin primario de la glándula mamaria. Reporte de un caso. Cir Cir. 2017;85(1):70–75. doi: 10.1016/j.circir.2015.09.009. [DOI] [PubMed] [Google Scholar]
- 15.Wadhwa A, Senebouttarath K. Primary lymphoma of the breast: A case series. Radiol Case Reports. 2018;13(4):815. doi: 10.1016/j.radcr.2018.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Terada T. Follicular Lymphoma of Breast. Breast J. 2016;22(3):356–357. doi: 10.1111/tbj.12580. [DOI] [PubMed] [Google Scholar]
- 17.Martinelli G, Ryan G, Seymour JF, et al. Primary follicular and marginal-zone lymphoma of the breast: clinical features, prognostic factors and outcome: a study by the International Extranodal Lymphoma Study Group. Ann Oncol. 2009;20(12):1993–1999. doi: 10.1093/annonc/mdp238. [DOI] [PubMed] [Google Scholar]
- 18.Mirkheshti N, Mohebtash M. A rare case of bilateral breast lobular carcinoma coexisting with primary breast follicular lymphoma. J Community Hosp Intern Med Perspect. 2019;9(2):155. doi: 10.1080/20009666.2019.1579611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Michalinos A, Vassilakopoulos T, Levidou G, Korkolopoulou P, Kontos M. Multifocal Bilateral Breast Cancer and Breast Follicular Lymphoma: A Simple Coincidence? J Breast Cancer. 2015;18(3):296. doi: 10.4048/jbc.2015.18.3.296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tiong TYV, Alsannaa SM, Soon STG, Poon LM, Goh YG. A rare case of primary follicular lymphoma of the breast in a male patient. Breast J. 2020;26(7):1404–1406. doi: 10.1111/tbj.13772. [DOI] [PubMed] [Google Scholar]
- 21.Miura S, Hashimoto H, Sawada T, Ishida T, Usuki K, Morikawa T. CD10-Negative Primary Breast Follicular Lymphoma: A Rare Case of Primary Breast Lymphoma With an Atypical Immunophenotype Mimicking Marginal Zone Lymphoma. Int. J. Surg. 2020;29(4):400–403. doi: 10.1177/1066896920977690. [DOI] [PubMed] [Google Scholar]
- 22.Ismail S, Kherbek H, Skef J, Naser Eldine M, Alshehabi Z. Primary marginal zone lymphoma of the breast; a rare case report and review of the literature. Hum Pathol Case Reports. 2020;22 [Google Scholar]
- 23.Talluri S, Subramanian CR, Werner E, Kasireddy V, Philip L. Bilateral Primary Marginal Zone Lymphoma of the Breast – A Rare Breast Malignancy. In Vivo (Brooklyn) 2020;34(6):3597. doi: 10.21873/invivo.12204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Parashar K, Nepal P, Assaker R, Vargas J, Rosasco S. Incidentally detected Non-Hodgkin's lymphoma of the breast. A case report with review of literature. Radiol Case Reports. 2021;16(1):205–209. doi: 10.1016/j.radcr.2020.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liguori G, Cantile M, Cerrone M, et al. Breast MALT lymphomas: A clinicopathological and cytogenetic study of 9 cases. Oncol Rep. 2012;28(4):1211–1216. doi: 10.3892/or.2012.1942. [DOI] [PubMed] [Google Scholar]
- 26.Gluskin J, D'Alessio D, Kim AC, Morris EA, Chiu A, Noy A. Primary lymphoma of the breast: A report of two cases. Clin Imaging. 2020;68:295–299. doi: 10.1016/j.clinimag.2020.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wei Lin C, Xu C, Xi Q, Wang C. Primary Burkitt's lymphoma of the breast without Epstein-Barr virus infection: A case report and literature review. Indian J Pathol Microbiol. 2015;58(4):546. doi: 10.4103/0377-4929.168863. [DOI] [PubMed] [Google Scholar]
- 28.Testa AC, De Blasis I, Di Legge A, Belli P, Hohaus S, Ferrandina G. Burkitt's lymphoma of the breast metastatic to the ovary diagnosed during pregnancy. Ultrasound Obstet Gynecol. 2013;42(3):364–366. doi: 10.1002/uog.12533. [DOI] [PubMed] [Google Scholar]
- 29.Janbabai G, Kayedimajd S, Alian S, Naghshvar F, Rashidi M, Farazmandfar T. Bilateral breast swelling in a 23-year-old woman with Burkitt lymphoma. J Res Med Sci. 2012;17(12):1188. [PMC free article] [PubMed] [Google Scholar]
- 30.Negahban S, Ahmadi N, Oryan A, et al. Primary Bilateral Burkitt Lymphoma of the Lactating Breast. Mol Diagnosis Ther. 2012;14(4):243–250. doi: 10.1007/BF03256380. [DOI] [PubMed] [Google Scholar]
- 31.Elgaafary S, Nagel I, López C, et al. Double-hit lymphoma of the male breast: a case report. J Med Case Rep. 2020;14(1) doi: 10.1186/s13256-020-02526-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Meucci R, Pistolese CA, Perretta T, et al. Primary Extranodal Follicular T-Cell Lymphoma and Ductal Breast Carcinoma Diagnosed by a Magnetic Resonance Imaging-Guided Vacuum-Assisted Biopsy: A Case Report. Am J Case Rep. 2021;22(1) doi: 10.12659/AJCR.929309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Turner SD. An Exploration into the Origins and Pathogenesis of Anaplastic Large Cell Lymphoma. Anaplastic Lymphoma Kinase (ALK)-Positive. Cancers (Basel) 2017;9(10) [Google Scholar]
- 34.Bonzheim I, Geissinger E, Roth S, et al. Anaplastic large cell lymphomas lack the expression of T-cell receptor molecules or molecules of proximal T-cell receptor signaling. Blood. 2004;104(10):3358–3360. doi: 10.1182/blood-2004-03-1037. [DOI] [PubMed] [Google Scholar]
- 35.Malcolm TIM, Villarese P, Fairbairn CJ, et al. Anaplastic large cell lymphoma arises in thymocytes and requires transient TCR expression for thymic egress. Nat Commun. 2016;7(1):1–12. doi: 10.1038/ncomms10087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Morris SW, Kirstein MN, Valentine MB, et al. Fusion of a Kinase Gene, ALK, to a Nucleolar Protein Gene, NPM, in Non-Hodgkin's. Lymphoma. Science. 1994;263(5151):1281–1284. doi: 10.1126/science.8122112. [DOI] [PubMed] [Google Scholar]
- 37.Turner SD, Yeung D, Hadfield K, Cook SJ, Alexander DR. The NPM-ALK tyrosine kinase mimics TCR signalling pathways, inducing NFAT and AP-1 by RAS-dependent mechanisms. Cell Signal. 2007;19(4):740–747. doi: 10.1016/j.cellsig.2006.09.007. [DOI] [PubMed] [Google Scholar]
- 38.Iyengar P, Reid-Nicholson M, Moreira AL. Pregnancy-associated anaplastic large-cell lymphoma of the breast: A rare mimic of ductal carcinoma. Diagn Cytopathol. 2006;34(4):298–302. doi: 10.1002/dc.20442. [DOI] [PubMed] [Google Scholar]
- 39.Minard-Colin V, Brugières L, Reiter A, et al. Non-Hodgkin Lymphoma in Children and Adolescents: Progress Through Effective Collaboration, Current Knowledge, and Challenges Ahead. J Clin Oncol. 2015;33(27):2963. doi: 10.1200/JCO.2014.59.5827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sathyanarayanan V, Lokesh KN, Lakshmaiah K, et al. Anaplastic lymphoma kinase (ALK) positive anaplastic large cell lymphoma (ALCL) of breast in a patient without a breast implant. Ann Saudi Med. 2014;34(6):551. doi: 10.5144/0256-4947.2014.551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ivashkevich Y, Chernov Y, Chinenov D, et al. Unusual Case of Anaplastic Large Cell Lymphoma Presenting as a Breast Mass in a Patient with no History of Breast Implants. Case Rep Pathol. 2020;2020:1–4. doi: 10.1155/2020/7543836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yi JH, Kim SJ, Kim WS. Brentuximab vedotin: clinical updates and practical guidance. Blood Res. 2017;52(4):243. doi: 10.5045/br.2017.52.4.243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lazzeri D, Agostini T, Bocci G, et al. ALK-1–Negative Anaplastic Large Cell Lymphoma Associated With Breast Implants: A New Clinical Entity. Clin Breast Cancer. 2011;11(5):283–296. doi: 10.1016/j.clbc.2011.03.020. [DOI] [PubMed] [Google Scholar]
- 44.Aguilera NSI, Tavassoli FA, Chu WS, Abbondanzo SL. T-Cell Lymphoma Presenting in the Breast: A Histologic, Immunophenotypic and Molecular Genetic Study of Four Cases. Mod Pathol. 2000;13(6):599–605. doi: 10.1038/modpathol.3880103. [DOI] [PubMed] [Google Scholar]
- 45.Miranda RN, Lin L, Talwalkar SS, Manning JT, Medeiros LJ. Anaplastic Large Cell Lymphoma Involving the Breast: A Clinicopathologic Study of 6 Cases and Review of the Literature. Arch Pathol Lab Med. 2009;133(9):1383–1390. doi: 10.5858/133.9.1383. [DOI] [PubMed] [Google Scholar]
- 46.Pina-Oviedo S, Ortiz-Hidalgo C, Carballo-Zarate AA, Zarate-Osorno A. ALK-Negative Anaplastic Large Cell Lymphoma: Current Concepts and Molecular Pathogenesis of a Heterogeneous Group of Large T-Cell Lymphomas. Cancers (Basel) 2021;13(18) doi: 10.3390/cancers13184667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Brown RA, Fernandez-Pol S, Kim J. Primary cutaneous anaplastic large cell lymphoma. J Cutan Pathol. 2017;44(6):570–577. doi: 10.1111/cup.12937. [DOI] [PubMed] [Google Scholar]
- 48.Parrilla Castellar ER, Jaffe ES, Said JW, et al. ALK-negative anaplastic large cell lymphoma is a genetically heterogeneous disease with widely disparate clinical outcomes. Blood. 2014;124(9):1473–1480. doi: 10.1182/blood-2014-04-571091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lobello C, Tichy B, Bystry V, et al. STAT3 and TP53 mutations associate with poor prognosis in anaplastic large cell lymphoma. Leuk. 2020;35(5):1500–1505. doi: 10.1038/s41375-020-01093-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Dogan ZA, Miranda RN, Iyer S, Steiner RE, Singh P, Clemens MW. Anaplastic Large Cell Lymphoma of the Breast Arising in a Burn Cicatrix. Aesthetic Surg J. 2020;40(4):NP159. doi: 10.1093/asj/sjz245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sasaki K, Sugaya M, Fujita H, et al. A case of primary cutaneous anaplastic large cell lymphoma with variant anaplastic lymphoma kinase translocation. Br J Dermatol. 2004;150(6):1202–1207. doi: 10.1111/j.1365-2133.2004.05987.x. [DOI] [PubMed] [Google Scholar]
- 52.Pulitzer M, Ogunrinade O, Lin O, Steinherz P. ALK-positive (2p23 rearranged) anaplastic large cell lymphoma with localization to the skin in a pediatric patient. J Cutan Pathol. 2015;42(3):182–187. doi: 10.1111/cup.12446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Booken N, Goerdt S, Klemke CD. Clinical spectrum of primary cutaneous CD30-positive anaplastic large cell lymphoma: an analysis of the Mannheim Cutaneous Lymphoma Registry. JDDG J der Dtsch Dermatologischen Gesellschaft. 2012;10(5):331–339. doi: 10.1111/j.1610-0387.2011.07794.x. [DOI] [PubMed] [Google Scholar]
- 54.Bergsten TM, Principe DR, Raicu A, Rubin J, Ong AL, Hagen C. Non-implant associated primary cutaneous anaplastic large cell lymphoma of the breast. J Surg Case Reports. 2019;2019(5):1–3. doi: 10.1093/jscr/rjz139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mehta-Shah N, Clemens MW, Horwitz SM. How I treat breast implant–associated anaplastic large cell lymphoma. Blood. 2018;132(18):1889–1898. doi: 10.1182/blood-2018-03-785972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Loch-Wilkinson A, Beath KJ, Knight RJW, et al. Breast implant-associated anaplastic large cell lymphoma in Australia and New Zealand: High-surface-area textured implants are associated with increased risk. Plast Reconstr Surg. 2017;140(4):645–654. doi: 10.1097/PRS.0000000000003654. [DOI] [PubMed] [Google Scholar]
- 57.Miranda RN, Aladily TN, Prince HM, et al. Breast Implant–Associated Anaplastic Large-Cell Lymphoma: Long-Term Follow-Up of 60 Patients. J Clin Oncol. 2014;32(2):114. doi: 10.1200/JCO.2013.52.7911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Akhavan AA, Wirtz EC, Ollila DW, Bhatt N. An Unusual Case of BIA-ALCL Associated with Prolonged/Complicated Biocell-Textured Expander, followed by Smooth Round Breast Implant Exposure, and Concurrent Use of Adalimumab. Plast Reconstr Surg. 2021:299–303. doi: 10.1097/PRS.0000000000008155. [DOI] [PubMed] [Google Scholar]
- 59.Freedman J, Peredo A, Rosser J, Christian N, Kaoutzanis C. Biopsy Results Are Not Sufficient to Exclude Breast Implant–associated Anaplastic Large Cell Lymphoma: A Case Mistaken for Disseminated Silicosis. Plast Reconstr Surg Glob Open. 2020;8(9) doi: 10.1097/GOX.0000000000003153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Clemens MW, Medeiros LJ, Butler CE, et al. Complete Surgical Excision Is Essential for the Management of Patients With Breast Implant–Associated Anaplastic Large-Cell Lymphoma. J Clin Oncol. 2016;34(2):160. doi: 10.1200/JCO.2015.63.3412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Ferrufino-Schmidt MC, Jeffrey Medeiros L, Liu H, et al. Clinicopathologic Features and Prognostic Impact of Lymph Node Involvement in Patients With Breast Implant-associated Anaplastic Large Cell Lymphoma. Am J Surg Pathol. 2018;42(3):293. doi: 10.1097/PAS.0000000000000985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.D'Alessandris N, Lucatelli P, Tripodi D, Amabile MI, Ascoli V. Cytological features of breast implant-associated anaplastic large cell lymphoma in pleural effusion. Diagn Cytopathol. 2019;47(11):1213–1217. doi: 10.1002/dc.24287. [DOI] [PubMed] [Google Scholar]
- 63.Evans MG, Medeiros LJ, Marques-Piubelli ML, et al. Breast implant-associated anaplastic large cell lymphoma: clinical follow-up and analysis of sequential pathologic specimens of untreated patients shows persistent or progressive disease. Mod Pathol. 2021;34(12):2148–2153. doi: 10.1038/s41379-021-00842-6. [DOI] [PubMed] [Google Scholar]
- 64.Tripodi D, Amabile MI, Varanese M, D'Andrea V, Sorrenti S, Cannistrà C. Large cell anaplastic lymphoma associated with breast implant: a rare case report presentation and discussion of possible management. Gland Surg. 2021;10(6):2076–2080. doi: 10.21037/gs-20-853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Johnson L, O'Donoghue JM, McLean N, et al. Breast implant associated anaplastic large cell lymphoma: The UK experience. Recommendations on its management and implications for informed consent. Eur J Surg Oncol. 2017;43(8):1393–1401. doi: 10.1016/j.ejso.2017.05.004. [DOI] [PubMed] [Google Scholar]
- 66.Richardson K, Alrifai T, Grant-Szymanski K, et al. Breast implant-associated anaplastic large-cell lymphoma and the role of brentuximab vedotin (SGN-35) therapy: A case report and review of the literature. Mol Clin Oncol. 2017;6(4):539. doi: 10.3892/mco.2017.1170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Thienpaitoon P, Disphanurat W, Warnnissorn N. Breast implant-associated anaplastic large cell lymphoma in an Asian patient: The first case report from Thailand. Arch Plast Surg. 2020;47(5):478. doi: 10.5999/aps.2020.00108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Allchin RL, Wickenden K, Pilgrim S, Wilson-Morkeh I, Miall FM. The Successful Use of Neo Adjuvant Brentuximab Vedotin in the Treatment of BIA-ALCL. HemaSphere. 2020;4(6):e501. doi: 10.1097/HS9.0000000000000501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Berlin E, Singh K, Mills C, Shapira I, Bakst RL, Chadha M. Breast Implant-Associated Anaplastic Large Cell Lymphoma: Case Report and Review of the Literature. Case Rep Hematol. 2018;2018:1–6. doi: 10.1155/2018/2414278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Thibodeau R, Fan KL, Wehner PB. Stage IV Breast Implant–associated Anaplastic Large-cell Lymphoma with Complete Pathologic Response to Neoadjuvant Chemotherapy. Plast Reconstr Surg Glob Open. 2019;7(9):1. doi: 10.1097/GOX.0000000000002446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Alderuccio JP, Desai A, Yepes MM, Chapman JR, Vega F, Lossos IS. Frontline brentuximab vedotin in breast implant-associated anaplastic large-cell lymphoma. Clin Case Reports. 2018;6(4):634. doi: 10.1002/ccr3.1382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Keech JA, Creech BJ. Anaplastic T-cell lymphoma in proximity to a saline-filled breast implant. Plast Reconstr Surg. 1997;100(2):554–555. doi: 10.1097/00006534-199708000-00065. [DOI] [PubMed] [Google Scholar]
- 73.Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127(20):2375–2390. doi: 10.1182/blood-2016-01-643569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Miranda RN, Feldman AL, Soares FA. Breast implant-associated anaplastic large cell lymphoma. In: K.H. Allison et al. (eds). World Health Organization Breast Tumours 245-248 (IARC: Lyon, 2019).
- 75.CocoRuby Plastic Surgery. Updated Breast Implant Cancer (BIA-ALCL): Everything We Know In 2021. https://cocorubyplasticsurgery.com.au/blog/update-on-breast-implant-associated-alcl-bia-alcl-everything-we-know-in-2021/ [Accessibility verified November 12, 2021].
- 76.Ohishi Y, Mitsuda A, Ejima K, et al. Breast implant-associated anaplastic large-cell lymphoma: first case detected in a Japanese breast cancer patient. Breast Cancer. 2020;27(3):499. doi: 10.1007/s12282-020-01064-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Lai CS, Wu CY, Chen IC, Tsai YC. Breast implant-associated anaplastic large cell lymphoma (BIA-ALCL) – The first case in Taiwan. Asian J Surg. 2021;44(6):869–870. doi: 10.1016/j.asjsur.2021.03.049. [DOI] [PubMed] [Google Scholar]
- 78.Ionescu P, Vibert F, Amé S, Mathelin C. New Data on the Epidemiology of Breast Implant-Associated Anaplastic Large Cell Lymphoma. Eur J Breast Heal. 2021;17(4):302. doi: 10.4274/ejbh.galenos.2021.2021-5-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.De Jong D, Vasmel WLE, De Boer JP, et al. Anaplastic Large-Cell Lymphoma in Women With Breast Implants. JAMA. 2008;300(17):2030–2035. doi: 10.1001/jama.2008.585. [DOI] [PubMed] [Google Scholar]
- 80.Cordeiro PG, Ghione P, Ni A, et al. Risk of Breast Implant Associated Anaplastic Large Cell Lymphoma (BIA-ALCL) in a Cohort of 3546 Women Prospectively Followed Long Term After Reconstruction with Textured Breast Implants. J Plast Reconstr Aesthet Surg. 2020;73(5):841. doi: 10.1016/j.bjps.2019.11.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Santanelli Di Pompeo F, Sorotos M, Clemens MW, et al. Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL): Review of Epidemiology and Prevalence Assessment in Europe. Aesthetic Surg J. 2021;41(9):1014–1025. doi: 10.1093/asj/sjaa285. [DOI] [PubMed] [Google Scholar]
- 82.Lechner MG, Megiel C, Church CH, et al. Survival Signals and Targets for Therapy in Breast Implant–Associated ALK− Anaplastic Large Cell Lymphoma. Clin Cancer Res. 2012;18(17):4549–4559. doi: 10.1158/1078-0432.CCR-12-0101. [DOI] [PubMed] [Google Scholar]
- 83.Hu H, Jacombs A, Vickery K, Merten SL, Pennington DG, Deva AK. Chronic biofilm infection in breast implants is associated with an increased T-cell lymphocytic infiltrate: implications for breast implant-associated lymphoma. Plast Reconstr Surg. 2015;135(2):319–329. doi: 10.1097/PRS.0000000000000886. [DOI] [PubMed] [Google Scholar]
- 84.Mempin M, Hu H, Vickery K, et al. Gram-Negative Bacterial Lipopolysaccharide Promotes Tumor Cell Proliferation in Breast Implant-Associated Anaplastic Large-Cell Lymphoma. Cancers (Basel) 2021;13(21) doi: 10.3390/cancers13215298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Deva AK, Turner SD, Kadin ME, et al. Etiology of Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL): Current Directions in Research. Cancers (Basel) 2020;12(12):1–14. doi: 10.3390/cancers12123861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Turner SD. The Cellular Origins of Breast Implant–Associated Anaplastic Large Cell Lymphoma (BIA-ALCL): Implications for Immunogenesis. Aesthetic Surg J. 2019;39(Supplement_1):S21–S27. doi: 10.1093/asj/sjy229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Tevis SE, Hunt KK, Miranda RN, Lange C, Butler CE, Clemens MW. Differences in Human Leukocyte Antigen Expression Between Breast Implant-Associated Anaplastic Large Cell Lymphoma Patients and the General Population. Aesthetic Surg J. 2019;39(10):1065–1070. doi: 10.1093/asj/sjz021. [DOI] [PubMed] [Google Scholar]
- 88.Wang HY, Thorson JA, Hinds BR, et al. Cutaneous intralymphatic anaplastic lymphoma kinase-negative anaplastic large-cell lymphoma arising in a patient with multiple rounds of breast implants. J Cutan Pathol. 2021;48(5):659–662. doi: 10.1111/cup.13936. [DOI] [PubMed] [Google Scholar]
- 89.Alcalá R, Llombart B, Lavernia J, Traves V, Guillén C, Sanmartín O. Skin involvement as the first manifestation of breast implant-associated anaplastic large cell lymphoma. J Cutan Pathol. 2016;43(7):602–608. doi: 10.1111/cup.12697. [DOI] [PubMed] [Google Scholar]
- 90.Evans MG, Miranda RN, Young PA, et al. B-cell lymphomas associated with breast implants: Report of three cases and review of the literature. Ann Diagn Pathol. 2020;46 doi: 10.1016/j.anndiagpath.2020.151512. [DOI] [PubMed] [Google Scholar]
- 91.Bletsis PP, Janssen LE, Visser O, et al. Bilateral breast implant associated chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL): A case report. Int J Surg Case Rep. 2020;71:341. doi: 10.1016/j.ijscr.2020.05.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Medeiros LJ, Marques-Piubelli ML, Sangiorgio VFI, et al. Epstein–Barr-virus-positive large B-cell lymphoma associated with breast implants: an analysis of eight patients suggesting a possible pathogenetic relationship. Mod Pathol. 2021;34(12):2154–2167. doi: 10.1038/s41379-021-00863-1. [DOI] [PubMed] [Google Scholar]
- 93.Elia R, Maruccia M, De Pascale A, Di Napoli A, Ingravallo G, Giudice G. The management of breast implant-associated anaplastic large cell lymphoma in the setting of pregnancy: seeking for clinical practice guidelines. Arch Plast Surg. 2021;48(4):373. doi: 10.5999/aps.2021.00185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Misad S.C, Walker B.K, Valbuena JR, et al. Anaplastic large cell lymphoma associated with breast implants, diagnosed by fine needle aspiration. Report of one case. Rev Med Chil. 2020;148(8):1207–1212. doi: 10.4067/S0034-98872020000801207. [DOI] [PubMed] [Google Scholar]