Abstract
For decades, the use of secondary metabolites of various herbs has been an attractive strategy in combating human diseases. Rosmarinic acid (RA) is a bioactive phenolic compound commonly found in plants of Lamiaceae and Boraginaceae families. RA is biosynthesized using amino acids tyrosine and phenylalanine via enzyme-catalyzed reactions. However, the chemical synthesis of RA involves an esterification reaction between caffeic acid and 3,4-dihydroxy phenyl lactic acid contributing two phenolic rings to the structure of RA. Several studies have ascertained multiple therapeutic benefits of RA in various diseases, including cancer, diabetes, inflammatory disorders, neurodegenerative disorders, and liver diseases. Many previous scientific papers indicate that RA can be used as an anti-plasmodic, anti-viral and anti-bacterial drug. In addition, due to its high anti-oxidant capacity, this natural polyphenol has recently gained attention for its possible application as a nutraceutical compound in the food industry. Here we provide state-of-the-art, flexible therapeutic potential and biomedical features of RA, its implications and multiple uses. Along with various valuable applications in safeguarding human health, this review further summarizes the therapeutic advantages of RA in various human diseases, including cancer, diabetes, neurodegenerative diseases. Furthermore, the challenges associated with the clinical applicability of RA have also been discussed.
Keywords: Rosmarinic acid, Bioactive phenolic compounds, Natural products, Drug discovery, Anti-cancer therapy
Introduction
The phenolic compound rosmarinic acid (RA) is commonly produced by Boraginaeceae and subfamily Nepetoideae of the Lamiaceae plant species. The presence of RA is confined to the Lamiaceae family and in others, including monocotyledonous, dicotyledonous, and hornworts and ferns (Petersen et al. 2009). Originally RA, an attractive phytochemical, was extracted as a pure compound from rosemary (Rosmarinus officinalis), an evergreen perennial aromatic herb. In addition to RA, rosemary produces a variety of other secondary metabolites, including terpenes, essential oils, and flavonoids. Structurally, RA is an ester of caffeic acid and 3,4-dihydroxy phenyllactic acid originating from aromatic amino acids, phenylalanine, and tyrosine, respectively (Dewick 2002). Biosynthesis of RA from amino acid precursors phenylalanine and tyrosine involve eight enzymatic reactions and is deduced from studies conducted on cell suspensions of Coleus blumei (Razzaque and Ellis 1977). However, the increasing demand for RA has spurred efforts for the biotechnological production of RA through cell cultures that provide higher yields (Khojasteh et al. 2020; Li et al. 2019). RA and its derivatives such as lithospermic acid, rabdossin, and salvialonic acid exhibit potent biological actions combating various human diseases such as cancer, diabetes, apoptosis, neurodegenerative disorders, cardiovascular disease, and inflammatory disorders (Kim et al. 2015, 2017, 2020c; Jeong and Hwang 2021; Jin et al. 2021b; Lee et al. 2021; Koprivica et al. 2022). RA prevents ultraviolet rays-induced photodamage (Gupta et al. 2021; Huerta-Madroñal et al. 2021). Pattananandecha et al. (2021) reported anti-oxidant activity and anti-photoaging effects of RA extracts on UVA-mediated damage on human fibroblast from the Thunbergia laurifolia plant. Figure 1 demonstrates RA's anti-inflammatory, anti-oxidant, anti-microbial, anti-diabetic, anti-cancer, and anti-hypertensive potential (Bulgakov et al. 2012; Zhang et al. 2021a).
Fig. 1.
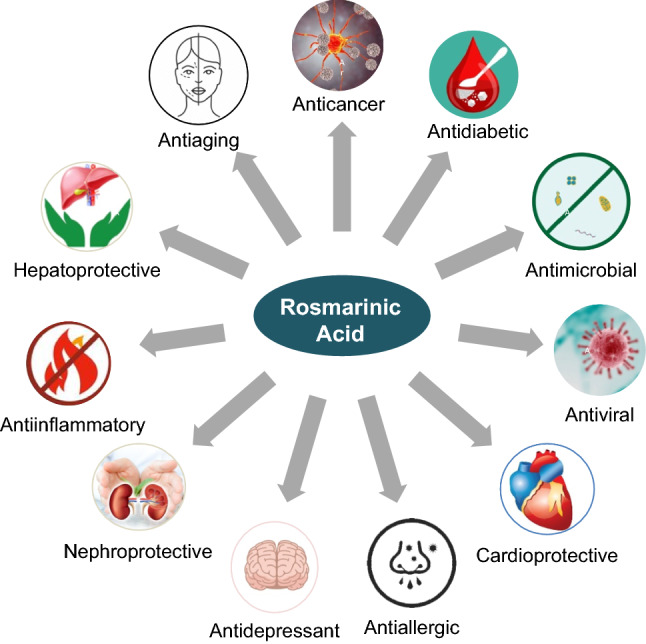
Pharmacological effects of RA on human health
In addition, several studies have reported that the dietary supplementation of herbs (rosemary, gingko, chamomile, and garlic) containing bioactive components (RA, quercetin, and thymol) improve immune responses, anti-oxidant status, and also lowered morbidity and mortality rates in animals and poultry (Alagawany and Abd El-Hack 2015). Bioinformatics and network-based analysis have demonstrated that RA could act on several target genes involved in inflammatory responses, anti-oxidant balance, tumor initiation, and progression to form a systemic pharmacological network (Guan et al. 2021). RA has been extensively studied for its therapeutic roles and nutraceutical properties to improve human and animal health based on its remarkable pharmacological and medicinal characteristics (Choi et al. 2016; Değer and Çavuş 2020; Kim et al. 2021b). In recent years, the design and development of potential nutraceutical and efficacious medicinal products from natural sources have attracted considerable interest (Alam et al. 2021, 2022). However, the effectiveness of such bioactive compounds depends on intake, their stability, and bioavailability. Here we attempt to summarize the recent updates on biological applications of RA and critically review its therapeutic potential and health benefits. This review also provides deeper mechanistic insights into the therapeutic effectiveness of RA in combating common metabolic diseases. We further provide a comprehensive understanding of RA's pharmacokinetic profile and updated clinical status as a potential drug molecule.
Structural features of RA
RA and related compounds were considered tannins of the Labiate plants earlier. They were later discovered as a pure compound from Rosmarinus officinalis by two Italian chemists in 1958. The chemical structure of RA (C18H16O8) is derived from hydroxycinnamic acid and has a chiral center with R and S enantiomers formed by rotation. RA appears as a crystalline solid with a molar mass of 360.2 g/mol and a melting point of 171–175 °C. RA is a highly lipophilic compound with a slight hydrophilic nature and therefore exhibits maximum solubility in organic solvents. The density of RA was assessed to be 1.54 g/cm3 and dissociation constant (pKa) of 3.57.
Biosynthesis of RA
Identified first in Coleus blumei, the biosynthesis of RA is a complex and non-linear enzyme-catalyzed process that begins with aromatic amino acids phenylalanine and tyrosine (Petersen and Simmonds 2003). Phenylalanine is deaminated to cinnamic acid by phenylalanine ammonia-lyase (PAL) enzyme of lignin through flavonoid biosynthetic pathway. Subsequently, the benzene ring of cinnamic acid is hydroxylated by cytochrome-P450 monooxygenase cinnamate-4 hydroxylase of flavonoid pathway forming 4-coumaric acid (Petersen 1997). Later hydroxycinnamic acid is activated by hydroxycinnamate: coenzyme A ligase (Petersen and Simmonds 2003). On the other hand, 4-hydroxyphenyllactic acid is derived from the precursor tyrosine by transamination to 4-hydroxy phenyl pyruvic acid. The transformation is catalyzed by tyrosine aminotransferase. 4-hydroxyphenylpyruvic acid is further reduced to 4-hydrophenyllactic acid by hydroxyphenylpyruvate reductase requiring NADH and NADPH as co-substrates (Trócsányi et al. 2020).
In the next biosynthetic step, 4-coumaryl CoA is derived by phenylalanine transformation and 4-hydroxyphenyllactate from tyrosine combined with tyrosine 4-coumaroyl-4′-hydroxyphenyllactic acid to release coenzyme A. The ester linkage between the carboxyl group of 4-coumaric acid and the hydroxyl group of 4-hydroxyphenyllactate is formed by hydroxycinnamoyl-CoA: hydroxyphenyllactate-hydroxycinnamoyl transferase, also known as rosmarinic acid synthase (Petersen et al. 1993). Lastly, 4-coumaryl-4-hydroxy phenyllactate is hydroxylated at 3 and 3′ position on aromatic rings by cytochrome P450 monooxygenases.
Pharmacological properties of RA
A multitude of biological roles of RA and similar compounds have been documented in earlier scientific literature (Marchev et al. 2021). These reports describe RA as an anti-inflammatory, anti-oxidant, and anti-microbial compound (Swarup et al. 2007). Since ancient times, RA has been used in folk medicine, cosmetics, and dietary supplements. The health-promoting benefits of RA could be based on scavenging reactive oxidation species, decreasing nitric oxide production, lipid peroxidation, increasing high-density lipoprotein, decreasing low-density lipoprotein levels, and deranged hemolysis (Baba et al. 2004; Guo et al. 2020). Nyandwi et al. (2021b) has reported lipid-lowering effects of RA through modulation of reverse cholesterol transport-related proteins, thereby combating hyperlipidemia-based disorders. RA also increased macrophage cholesterol influx via regulation of ABCA1 transporter expression through JAK2/STAT3, JNK, and PKC-p38 and ABCA2 expression JAK2/STAT3, JNK, and PKC-ERK1/2/p38 (Nyandwi et al. 2021a). Earlier scientific literature has also reported the role of RA in modulating several enzymes involved in the progression of several diseases, including diabetes, cancer, and inflammatory disease (Nadeem et al. 2019). Few studies elucidating RA-mediated enzyme inhibition have been listed in Table 1. The most interesting therapeutic role and mechanism of action of RA are detailed in the following subsections.
Table 1.
Enzyme Inhibition Constant Data of RA from Binding DB
| S. No | Target | Disease | IC50 (nM) | Assay description | Reference |
|---|---|---|---|---|---|
| 1 | HIV type 1 integrase | Acquired immuno-deficiency syndrome | 4000 | Inhibition of strand transfer activity of HIV-1 integrase | Mazumder et al. (1997) |
| 2 | Tyrosine-protein kinase Lck | Cancer, autoimmune disorders, chronic inflammatory diseases | 24,000 | Inhibition of binding to p56 Lck tyrosine kinase SH2 domain | Park et al. (2003) |
| 3 | Matrix metalloproteinase-2 | Cancer, arthritis, skin aging, blood vessel impairment | 27,200 | Inhibition of gelatinolytic activity of MMP2 in rat lung homogenate after 40 min by SDS-PAGE preincubated for 30 min | Murata et al. (2009) |
| 4 | Aldose reductase | Diabetes | 3910 | Inhibition of aldose reductase | Koukoulitsa et al. (2010) |
| 5 | Tyrosinase | Skin melanization | 250,000 | Inhibition of mushroom tyrosinase | Fujimoto et al. (2010) |
| 6 | Transcription factor AP-1 | Rheumatoid arthritis and cancer | 16,200 | Inhibition of transcription factor AP-1 binding to oligonucleotide containing TPA-responsive element in TPA-activated human HeLa cells after 1 h | Chen et al. (2014) |
| 7 | Interstitial collagenase | Rheumatoid arthritis, fibrotic disease | 5600 | Inhibition of human recombinant MMP1 catalytic domain using Dnp-Pro-beta-cyclohexyl-Ala-Gly-Cys(Me)-His-Lys-(Nma)-NH2 as substrate pre-incubated | Yuan et al. (2013) |
| 8 | Proto-oncogene tyrosine-protein kinase Src | Cancer | 26,000 | Inhibition of c-SRC SH2 domain | Sperl et al. (2009) |
| 9 | Estradiol 17-beta-dehydrogenase 2 | Osteoporosis | 3720 | Inhibition of human 17beta-HSD2 expressed in HEK293 cell lysates incubated for 10 min using [2,4,6,7-3H]-estradiol and NAD + by scintillation | Vuorinen et al. (2017) |
| 10 | Beta amyloid A4 protein | Alzheimer’s disease | 71,900 | Inhibition of amyloid beta (1 to 42 residues) (unknown origin) aggregation after 24 h by thioflavin T assay | Kwon et al. (2015) |
| 11 | Reverse transcriptase | Acquired immuno-deficiency syndrome | 100,000 | Inhibition of HIV1 reverse transcriptase | Dubois et al. (2008) |
| 12 | Integrase | Acquired immuno-deficiency syndrome | 63,500 | Inhibition of HIV1 integrase using labelled oligonucleotide substrate in presence of bovine serum albumin by ELISA | Dubois et al. (2008) |
| 13 | Aldose reductase | Diabetes complications | 3890 | Inhibition of Rattus norvegicus (rat) lens aldose reductase | Jain et al. (2012) |
| 14 | Tyrosine-protein kinase Fyn | Neurodegenerative disease | 1300 | Inhibition of human Fyn expressed in Sf9 cells after 20 min by ELISA in presence of 1 umol/L ATP | Jelić et al. (2007) |
| 15 | Lipoxygenase | Cancer, asthma, inflammatory disease | 1,150,000 | Inhibition of soybean lipoxygenase in vitro. Binding energy and IC-50 calculated to detect inhibitory activity | Koukoulitsa et al. (2007) |
| 16 | MARK-4 | Cancer | 6200 | Inhibition of MARK-4 validated in cell-free and cell-based enzyme assay. Increased apoptosis in MARK-4 overexpressing cancer cells | Anwar et al. (2020) |
| 17 | Glycogen Synthase kinase 3β | Alzheimer’s disease | 135 | Inhibition of GSK-3 β enzyme involved in several human diseases | Paudel et al. (2018) |
Anti-inflammatory potential
Inflammation is a vital component of innate immunity that initiates protective responses and healing cascades. The inflammatory process maintains immune system homeostasis and significantly impacts human health (Yi et al. 2017). The rapid recruitment of granulocytes marks acute inflammation, whereas macrophages and cytokine release by T cells are involved in chronic inflammation. The sustained inflammatory stage promotes chronic inflammation causing life-long debilitating illnesses such as irritable bowel syndrome, multiple sclerosis, systemic lupus erythematosus, and rheumatoid arthritis (Luo et al. 2020a). Most immunosuppressants or steroidal therapy treatments often prove inadequate and impose potent side effects. Bioactive components from natural herbs such as essential oils, flavonoids, phenols promote anti-inflammatory responses. As mentioned previously, RA exerts profound anti-inflammatory effects, and many in vivo and in vitro studies have reported the anti-inflammatory potential of RA in inflammatory diseases (Gonçalves et al. 2022).
T cell subset involved in the development and progression of rheumatoid arthritis is an important therapeutic target. RA induced T-cell apoptosis via cytochrome leakage blocks mitochondrial membrane potential disruption, and a reduced Bcl-2 production was observed in RA patients (Hur et al. 2007). RA application has been reported to influence collagen biosynthesis in a few studies (Sutkowska et al. 2021). Youn et al. (2003) found improved arthritic index and many affected hind paws in murine models of collagen-induced arthritis. In addition, prolonged treatment by RA significantly ameliorated collagen-induced arthritis judged by reduced synovitis and depleted COX-2 expressing cells in affected joints. Similarly, RA purified from Mexican mint was found to inhibit RANKL-induced osteoclastogenesis via downregulation of the NF-κB signaling pathway in cell culture (Phromnoi et al. 2021). Interestingly, RA enhanced osteo-integration of osteoblast cells on titanium surface by improving osteoblast cell differentiation, mineralization, and bone formation via the RANKL/RANK/OPG pathway (Jeong et al. 2021).
Moreover, RA significantly diminished IL-1β and TNF-α release ameliorating collagen-induced arthritis under in vivo conditions (Hsu et al. 2011). Hu et al. (2018) showed the protective effect of RA on rat chondrocytes by inhibiting extracellular matrix degradation in osteoarthritis. RA antagonized catabolic activity of IL-1β inhibited matrix-degrading enzymes and inflammatory molecules. Recently, RA isolated from Punica granatum showed anti-arthritic potential in Freund's complete adjuvant-induced arthritic rat model. The study concluded the implication of RA in clinical settings for the therapeutic management of arthritis (Gautam et al. 2019). The above findings suggest RA as a potential novel therapy for inflammatory joint disorders.
The incidence of chronic intestinal inflammation has increased worldwide, especially in developing countries. Current treatment modalities lack a complete cure for intestinal inflammatory diseases. Consequently, the need for effective and safe natural compounds becomes attractive in treating inflammatory bowel diseases. RA and other natural compounds have been extensively studied for clinical implications in intestinal inflammation (Formiga et al. 2020). Studies have reported ameliorative effects of RA on dextran sulfate-induced ulcerative colitis in mice models. RA markedly suppressed colonic inflammation via dual inhibition of NF-κB and STAT3 cascade activation and subsequent reduction of pro-survival genes. In addition to pleiotropic effects, RA inhibited pro-inflammatory cytokine release, thereby reducing colitis severity (Jin et al. 2017). In addition, researchers have detected that active ingredients of Perilla frutescens extracts, including apigenin, luteolin, and RA attenuated pro-inflammatory cytokine production, and RA specifically enhanced T-cell production, preventing dextran sulfate sodium (DSS) induced colitis in mice (Urushima et al. 2015). In another article on DSS-induced colitis models, RA and black-anthocyanin-rich extract alone or in combination alleviated inflammation through decreased myeloperoxidase and NO expression. In addition, reduced mRNA levels of IL-6, IL-1β, and iNOS (Zhao et al. 2018). Surprisingly, polyethylene-coated RA-loaded nano-formulation mitigated colonic inflammation and palliates pro-inflammatory cytokine in the dextran sulfate sodium-induced acute colitis animal models. Similarly, RA-loaded chitosan/nutriose-coated niosomes enhanced downregulated NLP3 proteins, an adaptor protein, caspase-1, and reduced IL-β1 levels in models of ulcerative colitis. On the other hand, RA enhanced Nrf2 translocation and hemeoxygenase expression exhibiting anti-inflammatory potential (Marinho et al. 2021).
Furthermore, the clinical efficacy of RA nano-formulation leads to an improved pharmacokinetic profile localized in the inflamed colon. However, the use of RA as therapeutic nano-medicine needs further consideration (Chung et al. 2020). Similarly, lipophilic modification of RA induces anti-colitic effects by activating the hypoxia-inducible factor 1-vascular endothelial growth factor pathway that improves ulcer healing (Jeong et al. 2015). Recently, Formiga et al. (2020) reported intestinal anti-inflammatory properties of RA in conjugation with p-Cymene. Combinatorial oral administration of RA and p-Cymene showed cytoprotective effects on trinitrobenzene sulphonic acid TNBS induced intestinal inflammation in rat models. This manifested as reduced mucosal injury and neutrophilic infiltrate. Both compounds exhibited immuno-modulatory effects by attenuating IL-1β, TNF-α levels and modulating the T-cell population. Moreover, RA down-regulated NF-κB transcription with subsequent induction of COX2 and iNOS. Few exciting research studies presented the gastro-protective effects of RA in ethanol-induced gastric lesions in vitro and in animal models. RA combated ulcer formation through modulation of oxidative parameters and inflammatory markers (do Nascimento et al. 2020; Wang et al. 2021b). Similar results were reported on ethanol-induced gastritis challenged rats. The RA-treated group showed increased antioxidant parameters and prostaglandin secretion, eventually protecting the mucosal barrier and gastric glands (Heidari et al. 2021). These findings corroborate and signify the general notion that RA possesses competent anti-inflammatory potential indicating that it can modulate inflammatory responses.
Given its anti-inflammatory attribute, RA exposure markedly increased inflammatory cells and Th2 cytokines in bronchoalveolar lavage fluid (BALF) with decreased total IgE and Ovalbumin (OVA) specific IgE levels and significantly improved airway hyperresponsiveness in OVA challenged the murine model of asthma (Liang et al. 2016). Histological findings reveal the presence of RA-mediated deranged mucus production and goblet cell hyperplasia. In another study reported by the same group, RA treatment significantly diminished inflammatory cells, upregulated SOD, glutathione peroxidase, and CAT activities, and a notable reduction in NOX-2 and NOX-4 expression in lung tissues. RA ameliorated oxidative lung injury and airway inflammation in asthmatic models (Liang et al. 2020). RA exhibited protective action against malathion (pesticide) induced lung damage through anti-inflammatory, antioxidant, and anti-apoptotic functions in albino Wistar rat models (Ahmed et al. 2021). However, future studies in vivo settings are required to unveil the underlying protective mechanism of RA. In another study, Shakeri et al. (2019) investigated the effects of RA on OVA-induced asthma and its influence on lung pathology of OVA-sensitized asthmatic rats. RA treatment significantly abridged IL-4, IgE, phospholipase A 2, and total protein levels but increased the IFN-γ/IL-4 ratio. Moreover, RA ameliorated pathological lung insults characterized by diminished interstitial fibrosis, bleeding, epithelial damage, and interstitial inflammation.
RA has been found to prevent inflammation and lipid accumulation in adipocytes indicating anti-adipogenic potential. RA significantly reduced C/EBPα protein levels and marked a decrease of sterol regulatory-element binding protein-1 expression. In adipocytes, attenuation of the sterol regulatory-element binding protein-1 mediated pathway is associated with decreased IL-6, TNF-α, resistin, and adiponectin levels (Vasileva et al. 2021). Another study reported that RA suppressed peroxisome proliferator-activated receptor-γ and CCAA/enhancer-binding protein, which is αT mediated. Additionally, RA halted TNF-α mRNA levels and activated p-ERK1/2 and pSmad3, exerting an inhibitory effect on adipogenesis, lipolysis, and inflammation. These findings conclude that RA is a potential molecule for managing obesity-related inflammation.
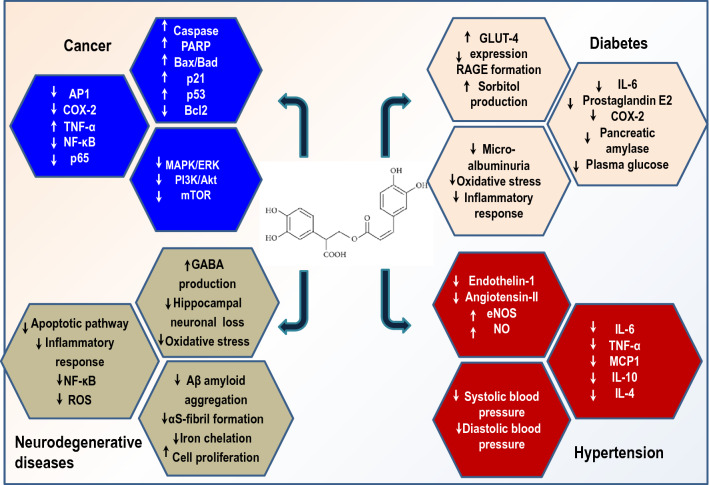
RA showed protection in other inflammatory diseases such as periodontal disease, pancreatitis, neuropathy, ischemia, and allergic rhinitis. In gingival fibroblast, RA-based therapy quenched IL-1β, IL-6, and TNF-α levels and inhibited iNOS. RA-based derangement of inflammatory response and improved anti-oxidant status alleviated progression of periodontitis. Also, RA pre-treatment was found to improve pathological alterations in sodium taurocholate-induced acute pancreatitis markedly and reduced inflammatory cytokine expression via downregulation of NF-κB translocation (Fan et al. 2015) (Fig. 2). In another study, therapeutic effects of RA were investigated in inflammation-induced acute kidney disease in Swiss albino mice injected with cisplatin. RA administered at 100 mg/kg for two consecutive days blocked the NLRP3 signaling pathway. Consequently, RA treatment down-regulated NF-κB and cyclooxygenase-2 expression and reduced renal tissue injury (Akhter et al. 2022). Similarly, RA diterpenes carsonic acid displayed effective countermeasures against acute and chronic COVID-19 complications and neurodegenerative diseases via inhibiting the NLRP3 inflammasome (Satoh et al. 2022). Recently, Gonçalves et al. (2022) conducted a database search on Pubmed, Scopus, and Web of Science to evaluate the anti-inflammatory potential of RA in preclinical in vivo models of inflammation. The most evaluated biomarkers included myeloperoxidase, catalase, glutathione, glutathione peroxidase, malionaldehyde and superoxide dismutase. Intraperitoneal dose of 10 mg/kg showed anti-inflammatory activity before and after induction of treatment.
Fig. 2.
Schematic diagram for possible mechanism of action of RA in ameliorating human diseases
Anti-oxidant potential
Oxidative stress due to mitochondrial dysfunction can induce organ damage and aggravate inflammation. Persistent production of reactive oxygen species causes cellular oxidative stress, which strongly augments the development and progression of several diseases. Therefore, several anti-oxidant compounds from natural sources have been expansively investigated as alternatives to current treatment strategies. Sevgi et al. (2015) evaluated the anti-oxidant and DNA damage protection ability of ten different phenolic acids such as RA, caffeic acid, chlorogenic acid, etc. RA exhibited maximum potential in β-carotene bleaching, 2,2-diphenyl-1-picrylhydrazyl free radical scavenging, and chelating effect assays.
Studies have found that RA exhibits strong anti-oxidant capacity in biological systems by scavenging free radical and reactive oxygen species (Ghorbani et al. 2019; Kim et al. 2020a). Recently, Sadeghi et al. (2020) investigated the impact of RA on lipopolysaccharide-induced oxidative damage and inflammation in peripheral blood mononuclear cells. The study demonstrated RA-mediated decline in lipid peroxidation and NO levels and restored anti-oxidant/oxidant balance. Furthermore, RA ameliorated lipopolysaccharide-induced inflammation by alleviating NF-κB and JNK MAPK signaling pathways. The beneficial role of RA in combating oxidative stress and associated diseases has been studied extensively. Tsai et al. (2019) showed protective effects of RA on selenite-induced cataract formation in Sprague–Dawley red pups. In addition, RA treatment significantly ameliorated oxidative damage by increasing anti-oxidant status (Nrf2, SOD, HO1, NQO1) and anti-oxidant enzymes (GSH-Px, GSH-Rd, and catalase). Moreover, protein levels of filensin and calpain2 were escalated along with a reduction in lipid peroxidation, calcium, and inflammation. Shahrestani et al. (2021) investigated the protective effects of RA encapsulated with ZnO/chitosan nanoparticles on methamphetamine-induced oxidative stress damage and changes in casp3a mRNA levels in zebra fish. RA/ZnO/chitosan nanoparticles exhibited high efficiency in combating methamphetamine elevation in casp3a mRNA levels and oxidative changes. Ilhan et al. (2022) reported protective effects of azoxymethane-induced rat colon carcinogenesis and anti-oxidant/oxidant balance. Fuster et al. (2021) formulated RA-loaded silk-fibroin nanoparticles to investigate the anti-tumor potential in human cervical and breast cancer cells. The study concluded increased cellular uptake of modified RA and enhanced anti-tumor activity in HeLa and MCF-7 cell lines.
Similarly, combination therapy of RA with sinapic acid improved anti-oxidant parameters (superoxide dismutase and glutathione-related enzymes) and reduced lipid and protein peroxidation, thereby reducing oxidative damage in the lenses of estrogen-deficient rats (Zych et al. 2019b). Another article validated the anti-oxidative role of RA on the liver and kidney of aging mice. RA supplementation at a 200 mg/kg dose significantly escalated the anti-oxidant enzyme activity of catalase, superoxide dismutase, and glutathione peroxidase, decreasing malondialdehyde levels. Histopathological images showed significant structural alterations in liver and kidney tissues (Zhang et al. 2015). These findings conclude a possible role of RA therapy in preventing or slowing the oxidative stress-induced aging process.
In several reports, RA has acted as hepatoprotective agent through modulation of inflammatory cascades and oxidative imbalances (Luo et al. 2021; Touiss et al. 2021). RA has attenuated chromium-induced hepatorenal oxidative damage and the formation of preneoplastic lesions in rats through upregulation of the Nrf2 signaling pathway and antioxidant actions (Khalaf et al. 2020). Recently, hepatoprotective effects of RA on acetaminophen-induced acute liver damage in mice were reported. The underlying mechanism of action was the antioxidant effect mediated by RA through RACK1/TNF-α pathway (Yu et al. 2021).
Similarly, RA exposure showed remarkable effects of RA in alleviating hepatic steatosis, oxidative stress, inflammation, and apoptosis in mice model challenged with non-alcoholic steatohepatitis (Komeili-Movahhed et al. 2021). RA also provided significant protection from concanavalin A (reversible-competitive AMPK inhibitor) induced auto-immune hepatitis in mice. After RA treatment, Serum levels of pro-inflammatory cytokines (IFN-γ, IL-2, IL-1β) were reduced, whereas anti-inflammatory cytokine (IL-10) was elevated. Furthermore, RA reduced hepatic damage via the activation of AMPK signaling (Wang et al. 2020). Another study found that RA markedly reduced Sertoli cell damage in rats caused by the electromagnetic field. The results showed that RA significantly improved testosterone levels, total anti-oxidant capacity, decreased malondialdehyde, and Sertoli cell apoptosis. These results support the view that RA administration as a food supplement could be a preventive measure against electromagnetic field-induced damages (Hajhosseini et al. 2013).
Oxidative damage is the causative factor for cognitive dysfunction in neurological disorders. Some researchers have demonstrated that incorporating RA as a dietary supplement could improve neurological issues and restore cognitive functions (Hassanzadeh-Taheri et al. 2021). Hasanein et al. (2017) reported ameliorative effects of RA in an animal model of ethanol-induced cognitive impairment. Prolonged and dose-dependent RA supplementation improved cognitive deficits in the alcoholic group. Decreased lipid peroxidation and nitrite levels disturbed oxidant/anti-oxidant levels have been suggested as a possible mechanism of chronic ethanol-induced amnesia. These results indicate RA as a potential therapeutic agent against alcoholic dementia.
Similarly, the beneficial effects of RA were evaluated in 4-aminopyridine and picrotoxin-induced epileptic seizures in mice. Although RA exposure could not prevent seizures, it decreased reactive oxygen species levels, catalase, and superoxide dismutase activity. DNA damage was measured in hippocampal tissues. The potential of RA in combating DNA damage and protection against ethanol-induced genotoxicity has already been reported. In addition, mitochondrial complex II activity was also found to be elevated post RA treatment. Since mitochondrial dysfunction due to deranged respiratory chain complexes is associated with seizure-induced brain damage, RA mitigated in epileptic models. Several studies have previously indicated the neuroprotective effect of RA due to its free radical scavenging properties and the ability to modulate mediators of signaling cascade such as NF-κB and c-fos that lead to neuronal apoptosis (Ma et al. 2020a). The study indicated that RA could exert anti-oxidant and neuroprotective effects in the treatment of epilepsy. Thus, RA could be incorporated with current anti-epileptic drugs in treating seizures (Luft et al. 2019). In another study, RA prevented oxidative stress and neuroinflammation in chemotherapy-induced peripheral neuropathy models. Dysregulated mitochondria aggravate oxidative stress that promotes an ongoing cycle of inflammatory damage.
Furthermore, an abrupt release of inflammatory cytokines can damage electron transport chain complexes, decrease ATP production and alter mitochondrial membrane potential. RA supplementation enhanced ATP production, restored mitochondrial membrane potential, tapered pro-inflammatory cytokine release, inhibited MAPK, and maintained AMPK levels, thereby exhibiting mitoprotective properties. A recent finding postulated bactericidal activity of RA to prevent MRSA pneumonia through modulation of Keap/Nrf2 signaling cascade. RA is a known inducer of Nrf2, inhibits its proteasomal degradation, and promotes nuclear translocation (Zhang et al. 2021b). Few studies have reported the bacteriostatic effects of RA treatment (Lu et al. 2021). However, in-depth in vivo screening of RA for translational success in clinical settings are needed (Areti et al. 2018).
Clinical implications of RA
RA shows various remarkable biological activities that reduce the likelihood of developing metabolic diseases such as cancer, diabetes, and neurodegenerative disorders (Feng et al. 2020). Several research groups have investigated the health benefits of RA and its extracts. Few studies depicting the pharmacological functions of RA have been summarized in Table 2. The subsections below provide the recent updates on the therapeutic benefits of RA in combating some common metabolic disorders.
Table 2.
Studies depicting various pharmacological functions of RA
| S. No | Pharmacological role | Function | Reference |
|---|---|---|---|
| 18 | Anti-inflammatory |
i. Reduced T-cell population, Bcl2 production in subjects with rheumatoid arthritis ii. Deranged inflammatory responses and improved acute pancreatitis in rat models iii. Decreased IgE production, mucus production and improved airway hyperresponsivness in OVA-challenged asthmatic rats iv. In conjugation with p-Cymene, modulated inflammatory cascade and reduced TNBS- induced intestinal inflammation in acute colitis rat models v. Ameliorated inflammation induced renal injury via inhibition of NLRP3 signaling in Cisplatin injected Swill abino mice |
Hur et al. (2007) Fan et al. (2015) Shakeri et al. (2019) Formiga et al. (2020) Akhter et al. (2022) |
| 19 | Anti-oxidant |
i. Slowed oxidative stress induced liver and kidney damage by enhancing anti-oxidant enzymes in mice models ii. Improved cognitive deficits in alcoholic dementia by reducing lipid peroxidation, nitrite levels and improving anti-oxidant status in rats iii. Ameliorated oxidative damage and exhibited protective effects in selenite-induced cataract animal models iv. Reduced acetaminophen-induced acute liver damage in mice through modulation of RACK1/TNF-α pathway v. Protective effects on methamphetamine-induced oxidative stress damage and changes in casp3a mRNA levels in zebra fish model |
Zhang et al. (2015) Hasanein et al. (2017) Tsai et al. (2019) Yu et al. (2021) Shahrestani et al. (2021) |
| 20 | Anti-cancer |
i. Induced prostate cancer cell apoptosis via modulation of intrinsic mitochondrial apoptotic pathway mediators ii. Inhibited proliferation and invasion in hepatocellular carcinoma cells via targeting PI3K/AkT/mTOR signalling pathway iii. Blocked FOXOM1 transcription factors, upregulated pro-apoptotic genes and exhibited anti-tumorigenic actions in triple negative breast cancer cells iv. Reduced pancreatic ductal adenocarcinoma via G1/S cycle arrest and inhibition of Gli translocation in mouse model of PDAC v. Propitiated TLR4/NF-κB/STAT3 activation and reduced colitis induced colon cancer in murine model |
Jang et al. (2018) Wang et al. (2019) Messeha et al. (2020) Zhou et al. (2022) Jin et al. (2021a) |
| 21 | Anti-diabetic |
i. Exhibited protective role via anti-inflammatory action in diabetic cerebral ischemic damage ii. Ameliorated hyperglycemia and insulin resistance by altering phosphoenol pyruvate carboxykinase levels and enhancing GLUT-4 expression iii. Prevented podocyte injury, reduced microalbuminuria and improved kidney function in rats with diabetic nephropathy iv. Attenuated inflammatory response and type 1 diabetes development in mice via modulatiom of innate and adaptive immune response v. Prevented podocyte injury, diabetic nephropathy alone and more efficiently in combination with Telmisartan in rat models |
Luan et al. (2013) Runtuwene et al. (2016) Samsu et al. (2019) Koprivica et al. (2022) Samsu et al. (2019) |
| 22 | Anti-neurodegenerative |
i. Attenuated motor neuron degeneration in rat models of amylotropic lateral sclerosis ii. Enhanced dopamine signalling pathway and suppressed Aβ aggregation in AD mice models iii. Prevented degeneration of nigrostriatal dopaminergic system, inhibited iron-induced α-synuclein aggregation against MPTP induced neurotoxicity iv. Regulated DJ-1/Akt/Nrf2 signaling and combated MPTP-induced neurotoxicity in zebrafish model of PD v. RA-chitosan nano-conjugates improved memory, neuroinflammation in Wistar rats |
Shimojo et al. (2010) Hase et al. (2019) Qu et al. (2019) Zhao et al. (2020) Fachel et al. (2020) |
| 23 | Anti-hypertensive |
i. Inhibited angiotensin-converting enzyme, reduced lipid profile, improved insulin sensitivity and lowered blood pressure in rats ii. Reduced vascular damage and blood pressure via inhibition of angiotensin-converting enzyme and pro-inflammatory cytokine release iii. Attenuated angiotensin converting enzyme and decreased hypertension in rats iv. Attenuated systolic blood pressure, increased eNOS production, improved vascular function in hypertensive rats |
Karthik et al. (2011) Alegría-Herrera et al. (2019) Ferreira et al. (2018) Pantam et al. (2019) |
Anti-cancer potential
Cancer, also called malignant tumor or neoplasm, is a large group of diseases characterized by the rapid creation of abnormal cells with uncontrolled proliferation (Kim et al. 2009, 2021c; Hwang et al. 2011; Son et al. 2013; Chung et al. 2015; da Silva et al. 2021). The ability of malignant cells to evade apoptosis or programmed cell death results from mutations in signaling pathways regulators. According to recent estimates released by WHO, 19.3 million new cases worldwide were diagnosed in 2020, with 10 million deaths. The global burden is expected to be 28.4 million cases in 2040. Socio-economic status and the aging population remain the primary factors responsible for increased cancer cases. Several medicinal properties, including anti-cancer activities, have been attributed to RA. Repeated use of chemotherapy drugs and radiotherapy often induces chemo-resistance that causes cytotoxicity to normal cells. Identifying novel therapeutic compounds of natural origin has gained scientific attention as anti-cancer agents (Jin et al. 2020; da Silva et al. 2021).
Numerous evidences suggest anti-proliferative and pro-apoptotic effects of RA on cancer cells (Luo et al. 2020b; Pagano et al. 2021). RA downregulated DSS-induced colitis-associated colon cancer by propitiating TLR4/NF-κB/STAT3 activation in murine model. RA significantly ameliorated colitis severity, inflammatory markers, and anti-apoptotic factors (Jin et al. 2021a). Anti-cancer effects of RA in prostate cancer cell lines were investigated. RA decreased cell proliferation halted colony and spheroid formation in OC3 and DU145 cell lines. Furthermore, RA induced early and late-stage apoptosis downregulated proliferating cell nuclear antigen, cyclin, D1, and cyclin E1, whereas it upregulated p21 expression. In addition, RA supplementation inhibited a histone-deacetylase enzyme that modulated the expression of intrinsic mitochondrial apoptotic-pathway related genes (Bax, bcl-2, caspase-3, and poly ADP ribose polymerase 1) via enhancement of p53 levels. However, further in-depth studies are required to evaluate RA-mediated cytotoxicity on normal cells (Jang et al. 2018). In another study, the anti-cancer effects of RA on hepatocellular carcinoma were investigated. The findings demonstrated that dose-dependent RA induced G1 arrest and apoptosis and inhibited the epithelial to mesenchymal transition of SMMC-7721 cells (Fig. 2). Additionally, RA treatment markedly decreased tumor growth and phosphorylated-phosphoinositide 3-kinase (PI3K), p-Akt, and phosphorylated-mammalian target of rapamycin (p-mTOR) expression levels. This leads to the suggestion that RA-mediated anti-tumorigenic role may be partially achieved via inhibition of PI3K/Akt/mTOR pathway. The above results provide the theoretical basis for considering RA as an anti-cancer agent (Wang et al. 2019).
Similarly, RA exhibited anti-proliferative and pro-apoptotic functions by DJ-1 inhibition via regulation of the PI3K-Akt signaling pathway in osteosarcoma cells (Ma et al. 2020b). In triple-negative breast cancer cells, RA altered the expression of genes involved in intrinsic and extrinsic apoptotic pathways. Noticeably, RA exposure upregulated pro-apoptotic gene BNIP-3, TNF-α, and GADD45A, causing S phase arrest and suppressing surviving protein BIRC5 and TNFRSF11B (Messeha et al. 2020). Another report concluded that RA mediated the inhibition of melanoma cell proliferation, migration and invasion and promoted apoptosis by ameliorating ADAM17/EGFR/AKT/GSK3β axis (Huang et al. 2021). The above findings reveal that PI3K-Akt signaling is a crucial biological target for RA in cancer cells (Lee et al. 2018a, 2018b; Ko et al. 2019; Park et al. 2020; Kim et al. 2020b, 2021a). On the contrary, in another study, RA acted as a radioprotector in healthy cells but caused significant damage to melanoma cells in irradiated cultures. The underlying mechanism could be RA-mediated eumelanin synthesis consuming intracellular glutathione inducing oxidative damage (Olivares et al. 2020).
RA exhibited chemo-preventive effects alone and combined with Paclitaxel drug in breast cancer mice models. RA significantly suppressed NF-κB, VEGF, TNF-α expression and restored P53, Bcl-2, Bax, and caspase-3 levels, causing apoptosis (Mahmoud et al. 2021). Interestingly, in another study, Zhou et al. (2022) reported RA-based reduction in pancreatic ductal adenocarcinoma by restraining Gli translocation and facilitating proteasomal degradation. In addition, RA induced G1/S cell cycle arrest and apoptosis in the pancreatic ductal adenocarcinoma cells by regulating apoptotic genes' expression. In Swiss BALB mice, RA exposure displayed protective effects in cisplatin-induced ovarian toxicity through modulation of anti-oxidant and anti-inflammatory parameters (Gui et al. 2021). RA exposure ameliorated doxorubicin-mediated cardiotoxicity in both in vitro and in vivo models and, therefore, also be a promising drug for combating anti-cancer drug-mediated toxicity (Rahbardar et al. 2021). Similar outcomes of RA pretreatment were reported on human cardiomyocyte cell lines and human-induced pluripotent stem-cell-derived cardiomyocytes exposed to doxorubicin.
RA significantly reduced doxorubicin-induced cell apoptosis and caspase9 activity. Furthermore, RA promotes hemeoxygenase expression and downregulated ROS production, displaying potential in combating cancer-therapy-related cardiac dysfunction (Zhang et al. 2020a). RA-mediated inhibition of NLRP3 inflammasome has been beneficial in cyclophosphamide triggered premature ovarian failure. The above findings reveals the mechanistic insights of therapeutic action of RA through inhibition of NLRP3 inflammasome and associated signalling pathway (Yao et al. 2020).
Repeated usage of chemotherapeutic drugs in clinical practice confers multidrug resistance (MDR) and harmful outcomes. Interestingly, RA acid methyl ester, a derivative of RA-induced therapeutic effects against cervical cancer, has been documented through transcriptome analysis. RA treatment downregulated oncogenic transcription factor forkhead box M1 (FOXM1) and target genes associated with abnormal cell proliferation and tumor growth. Moreover, combination treatment of cisplatin and RA methyl ester reversed the cisplatin resistance in ovarian cancer cells via possible inhibition of FOXM1 (Lim et al. 2020). Similarly, RA exposure alleviated MDR, increased Adriamycin and P-glycoprotein intracellular accumulation, and decreased MDR-1 gene transcription in gastric cancer cells (Li et al. 2013). Liao et al. (2020) investigated the potential of RA in reversing multidrug resistance in non-small cell lung cancer. These results postulate RA as a potent multidrug resistance reversal compound that induces remarkable apoptosis in non-small cell lung cancer (NSCLC) and G1 phase cell cycle arrest. The collective findings also provide evidence of a mechanistic link between RA and mitogen-activated protein kinase (MAPK) signaling, indicating RA as a potential drug for NSCLC treatment.
Ample evidence suggests RA's attenuating effects on morphological features of cancer cells, including invasion, metastasis, and angiogenesis (Hsieh et al. 2020). Mostly cancer cells represent altered glycosylation profiles with sialylated moieties and carbohydrate antigens. Recently, Radziejewska et al. (2018) reported RA administration's effect on gastric cancer's intrinsic characteristics and found that RA at 200 µM concentration showed an inhibitory effect on sialylated Tn antigen expression matrix-metalloprotease 9 activity and enhanced collagen type 1 expression. Similarly, RA inhibited epithelial to mesenchymal transition in metastatic colorectal cancer cells via p-38/AP1 signaling. The study also documents the involvement of mir-1225-5p expression in RA-mediated anti-metastatic potential (Yang et al. 2021). Another previous study conducted by Zhang et al. (2018) reported substantial changes in ovarian cancer cell morphology comprising appearance of cell surface projections, blebbing of the cell membrane, and formation of apoptotic bodies upon RA treatment. In addition, suppressed cell migration, chromatin condensation, and DNA fragmentation were also observed (Zhang et al. 2018). These results suggest the clinical usefulness of RA as a complementary compound supporting conventional cancer treatment by targeting pathological characteristics of tumor cells. Recently, our lab reported the anti-cancer effects of RA on neuroblastoma cells via microtubule affinity regulating kinase 4 (MARK-4) inhibition, a potential drug target in many cancers. Molecular docking and in-silico simulation analysis revealed excellent binding affinity of RA with active pockets of MARK-4, quenching its tau phosphorylation activity. The study reveals the mechanistic insights of RA-mediated therapeutic cancer management (Anwar et al. 2020). Previous literature documents the remarkable effects of RA in reducing tau phosphorylation and cognitive dysfunction via downregulation of the JNK signaling pathway (Yamamoto et al. 2021).
Interestingly RA has also been combined with siRNA therapy to detect its anti-apoptotic effects on human glioma cells. The heat shock protein (Hsp)-27 expression was attenuated with Hsp-siRNA alone with no significant apoptosis. However, combined treatment of RA and quercetin along with Hsp-siRNA transfection suppressed Hsp27 expression and enhanced caspase-3 activity, indicating RA to be a potent combinatorial agent for glioblastoma therapy (Şengelen and Önay-Uçar 2018). Liu et al. (2021) reported anti-apoptotic effects of RA on the human glioma cell line through inhibition of Fyn kinase. In addition, RA significantly reduced MMPs expression, cell invasion, and migration. In another study, RA in conjugation with anti-MUC1 antibody significantly inhibited biomarkers including Tn, sialylated Tn, and fucosylated sugar antigens in gastric cancer cells.
Moreover, RA and anti-MUC1 antibody's combined action enhanced pro-apoptotic proteins Bax and Bad with the reduction in anti-apoptotic marker Bcl2 mRNA expression (Radziejewska et al. 2021). RA's anti-tumor effects were demonstrated by a combination of Adriamycin on hepatocellular carcinoma cells. The synergistic effect of RA and Adriamycin enhanced apoptotic rates by triggering mitochondrial-mediated signaling pathways and alleviated DNA damage. The protective effects of RA on radiation-induced pulmonary fibrosis were investigated in rat models. RA-downregulated NF-κB expression and modulated inflammatory markers. Furthermore, RA upregulated RhoA/Rock signaling by upregulation of miR-19b-3p, inhibiting fibrosis (Zhang et al. 2020b).
Anti-diabetic potential
Diabetes mellitus is an endocrine disorder resulting from absolute or relative loss of insulin secretion and insulin resistance. Persistent hyperglycemia may lead to secondary organ damage in diabetes, causing retinopathy, nephropathy, neuropathy, and vascular diseases. Recently RA has gained pharmacological importance due to its anti-diabetic potential. Current anti-diabetic drugs have certain limitations in efficacy and safety, causing weight gain or sudden hyperglycemia (Ngo et al. 2018). Previous experimental evidence showed that RA treatment could alleviate diabetes-associated secondary complications (Rao et al. 2014).
Runtuwene et al. (2016) investigated the anti-diabetic role of RA on glucose homeostasis and insulin sensitivity in animal models with streptozotocin (STZ) induced type 1 diabetes and high fat-induced type 2 diabetes. The data suggest that RA ameliorated hyperglycemia and insulin resistance with decreased phosphoenolpyruvate carboxykinase expression in the liver and enhanced GLUT-4 expression in skeletal muscles. Interestingly, the study found that 200 mg/kg was the most effective RA dose to exhibit anti-diabetic potential. A similar dose of RA has been used in other studies; however, clinical trials should be conducted to evaluate the effectiveness in humans with diabetes mellitus. In another research, type1 diabetic mice model, phenylethyl ester of RA attenuated inflammatory response and type 1 diabetes development through specific interference with innate and adaptive immune responses (Koprivica et al. 2022). Another study validated enhanced glucose uptake in skeletal muscles at 2 µM RA concentrations. In this study, RA-mediated glucose uptake was PI3K/Akt independent as wortmannin (inhibitor of PI3K) did not modulate RA stimulated glucose uptake. Interestingly, RA inhibited DNA-glycation, thereby modulating Akt-1 and Akt-3 expression in the hippocampal region of STZ-induced diabetic rats (Alrubaye et al. 2021).
Additionally, AMPK phosphorylation was also increased; however, more profound insights into the underlying mechanism of RA in the regulation of glucose homeostasis need to be explored (Vlavcheski et al. 2017). Studies have also reported the inhibitory effect of RA on pancreatic amylase. As pancreatic amylase inhibitors are considered a good option for treating hyperglycemia by preventing glucose release from starch, these inhibitors can be utilized as anti-diabetic drugs (McCue and Shetty 2004).
Many scientific kinds of research have shown that RA and related constituents significantly improved glucose and lipid metabolism, oxidative parameters, and inflammatory status (Michalikova et al. 2021). Oxidative damage and inflammation play a crucial role in the pathophysiological mechanism of diabetes mellitus exerting harmful effects leading to progressive organ damage. The pharmacological effects of RA have been studied in combating diabetes micro-vascular and macro-vascular complications (Bao et al. 2020). In a recent study, RA supplementation improved oxidized lipid-induced endothelial dysfunction in diabetic atherosclerosis via regulation of the p38-FOXO1-TXNIP signaling pathway and inhibition of NLRP3 inflammasome formation (Nyandwi et al. 2020). Uncontrolled hyperglycemia increases the possibility of ischemic stroke, and NF-κB activation plays a pivotal role in diabetic stroke pathogenesis. Some researchers have documented neuroprotective effects of RA via blockade of inflammatory response. Luan et al. (2013) explored the neuroinflammatory potential of RA against diabetic cerebral ischemic damage. RA attenuated BBB degradation, decreased infarct volume, reduced HMGB1 expression, and enhanced NF-κB and IF-κB-α phosphorylation during diabetic cerebral ischemic injury. These findings indicate the anti-inflammatory potential of RA as a therapeutic drug in diabetic cerebral ischemic injury. Another group of researchers explored the protective role of RA in hepatic inflammation in the diabetic state. It examined the impact of RA on inflammatory stress in the liver of type 1 diabetic mice. RA supplementation alleviated diabetes progression, hepatic inflammation, and glycation stress via decreased levels of IL-6, prostaglandin E2, and cyclooxygenase-2 enzyme (Fig. 2).
Moreover, RA treatment markedly reduced receptors for the formation of advanced glycation end products formation and enhanced hepatic sorbitol production, consequently diminishing glycative stress. These results suggest that RA may ameliorate hepatic inflammation and glycative injuries under a diabetic state by regulating inflammatory cascades and polyol pathways (Wen and Yin 2017). Oxidative stress significantly contributes to diabetes mellitus that several anti-oxidants can reduce. According to a study, RA exerts anti-inflammatory effects and improves endothelial function (Sotnikova et al. 2013). In concordance with the above study, Sotnikova et al. (2015) investigated the effect of RA treatment on systemic oxidative stress in STZ-induced diabetic rats. RA administration (50 mg/kg) mitigated oxidative stress-induced tissue injury in the kidney and pancreas. Consistent RA exposure for 10 weeks depressed lipid peroxidation and raised GSH levels to be normal.
Similarly, RA exhibited a hepatoprotective and renoprotective role in combating oxidative stress in STZ induced diabetic rats. RA did not show any effect in improving glycemic levels; however, SOD and CAT, ascorbic acid, non-protein thiol, and delta-aminolevulinic acid dehydratase were increased in the liver and kidney of diabetic rats (Mushtaq et al. 2015). Similarly, RA treatment reduced methotrexate-induced hepatotoxicity and nephrotoxicity in the Wistar rat model. RA reduced necrosis and leukocyte infiltration through antioxidant and anti-inflammatory characteristics (Jafaripour et al. 2021). Such reports prove that naturally occurring compounds may contribute to diabetes management. In another report, RA attenuated high glucose-induced mitochondrial injury and improved cardiac function in diabetic mice models with cardiomyopathy via activating the SIRT1/PGC-1α pathway (Diao et al. 2021).
Samsu et al. (2019) observed the significance of RA monotherapy in preventing podocyte injury, detachment, and subsequent development of diabetic nephropathy in rats. It was found that treatment with RA alone was significantly more efficacious than the combination of telmisartan and RA in reducing microalbuminuria and improving kidney function. However, further studies are required to clarify the non-synergistic effects of RA and telmisartan in combination therapy. A group of researchers conducted a comparative analysis on hypoglycaemic, hyperlipidemic, and anti-oxidative effects of RA and another rosemary compound, carnosic acid. Both compounds showed anti-glycative, anti-oxidative roles, anti-inflammatory roles, and protected tissue damage. However, RA showed a stronger protective role than carnosic acid in alleviating diabetic symptoms (Ou et al. 2018). Another study evaluated the anti-oxidative effect of RA and sinapic acid in combination in type 2 diabetic rats (Zych et al. 2019a). The findings point to the beneficial impact of RA and sinapic acid treatment on oxidative stress parameters. Silva et al. (2021) demonstrated the hypoglycemic effect of RA extracts from Origanum vulgare on alloxan-induced diabetic. Diabetes is characterized by the deposition of amylin protein aggregates in pancreatic islets, contributing to diabetic pathology. RA oral administration prevented amylin aggregation, providing insight into RA's nutraceutical properties in diabetes management (Wu et al. 2021).
Anti-neurodegenerative potential
The progressive dysfunction and loss of a selectively vulnerable group of neuronal cells are classic neurodegenerative diseases (Kumar et al. 2016, 2022). Abnormal protein accumulation and deposition, such as neurofibrillary tangles, Lewy body, tau protein, and α-synuclein, constitute the major histological marker of neuropathologic diagnosis (Gibb and Lees 1988). Numerous in vivo and in vitro studies demonstrated that natural compounds with high anti-oxidative and anti-inflammatory properties might be essential in managing neurodegenerative diseases, including epilepsy, Alzheimer's disease (AD), Parkinson's disease (PD), memory impairment, and sclerosis (Ghasemzadeh and Hosseinzadeh 2020; Thingore et al. 2021). RA is well-known for combating ROS production and inflammatory response, highlighting its clinical significance in reducing neurodegenerative damage (Fachel et al. 2019). RA significantly abrogated LPS-induced mitochondrial dysregulation, oxidative/nitrosative damage, and neuroinflammation by promoting microglial M1/M2 repolarization (Wei et al. 2021). In another group, RA showed protective effects against LPS-mediated neuroinflammation by exhibiting anti- inflammatory potential in the adult zebrafish model (Fasolo et al. 2021). Fachel et al. (2020) formulated RA-loaded mucoadhesive chitosan-coated nanoemulsions against LPS induced neuroinflammation and memory defect in Wistar rats. RA bioavailability was enhanced with improvement in neuroprotection. Previous reports have affirmed the implication of RA in promoting neurogenesis and alleviation of cognition defects and synaptic regulation (Mirza et al. 2021). RA has been proven to be a promising, innovative, and effective pharmacotherapy agent for treating depressive disorders (Lataliza et al. 2021). micro-RNAs play a pivotal role in the pathogenesis of neurodegenerative diseases. Lv et al. (2020) demonstrated RA-mediated regulation of mi-miR-155-5p alleviating PD pathogenesis and concluded that suggesting that miR-155-5p could be used as a target for PD therapeutic management.
RA administration from presymptomatic stage attenuated motor neuron degeneration improved clinical score and extended lifespan in rat models of amyotrophic lateral sclerosis (Shimojo et al. 2010). In a mice model with PTZ induced seizures, RA exposure improved latency and reduced seizure frequency, reinforcing the neuroprotective effect of RA in epilepsy (Coelho et al. 2015). Several studies have reported that RA can modulate the gamma-aminobutyric acidergic (GABA) neurotransmission system via attenuating GABA transaminase enzyme. RA exposure enhances GABA production in the synaptic cleft, resulting in increased GABA transmission and an attractive strategy for epilepsy treatment (Ben‐Menachem 2011). Another study evaluated anticonvulsant, anti-oxidative, and neuroprotective effects of RA treatment in mice models of picrotoxin and 4-aminopyridine induced seizures (Luft et al. 2019). The findings demonstrated that RA significantly reduced neuronal cell damage and can be administered in conjugation with anti-epileptic drugs for improved outcomes (Fig. 2).
Several drugs targeting Aβ (amyloid β) gathering or reversals have failed clinical trials, demanding a timely search for polyphenolic compounds with anti-amyloidagenic properties. RA and its derivatives reduced protein aggregation in several neurological disorders, with immense preventive benefits (Lin et al. 2020). In this regard, Cornejo et al. (2017) investigated the anti-aggregating properties of some phenolic compounds, including RA, on heparin-induced tau aggregation in vitro. Structure-based analysis revealed that RA interacted with the hexapeptide region involved in fibrillization and β-sheet assembly and prevented tau-aggregation. Similarly, Hase et al. (2019) reported RA-mediated suppression of Aβ aggregation in AD mice models via enhancing dopamine signaling pathway through inhibition of monoamine oxidase. Salvia officinalis extracts containing RA reduced Aβ induced neurotoxicity in cultured rat pheochromocytoma cells. RA also reduced subsequent events associated with Aβ toxicity, including oxidative stress, DNA fragmentation, and tau-hyperphosphorylation (Iuvone et al. 2006).
Another study reported that RA exerted protective effects on Aβ evoked oxidative stress via Akt/GSK-3β/Fyn pathway through upregulation of nuclear factor E2-related factor (Nrf2) activity (Rong et al. 2018). Previous studies have reported the clinical effectiveness of RA-rich Melissa officinalis in preventing AD progression (Noguchi-Shinohara et al. 2020). The safety of RA (from Melissa officinalis) consumption was evaluated in a randomized-placebo controlled double-blind trial conducted for 24 weeks on subjects with mild dementia associated with AD. Melissa officinalis extract containing 500 mg RA was well tolerated by AD subjects with no adverse effects (Luft et al. 2019). The above findings could validate the implication of RA in the therapeutic management of AD. However, more comprehensive studies are needed to apply RA as a nutraceutical supplementation agent for AD treatment.
Derangement of dopaminergic neurons and dopamine production are classical neuropathological characteristics of PD. Additionally, increased oxidative markers and iron level-neuronal apoptosis are triggering factors. Hence compounds displaying multiple features, including iron-chelating, anti-apoptotic and free-radical scavenging, might serve as potential candidates for therapeutic management of PD. The ameliorative effects of RA on dopaminergic cell apoptosis and ROS production in PD have been evaluated in several studies. Wang et al. (2012) reported the neurorescue effect of RA on 6-hydroxydopamine (6–OHDA) induced dopamine neuronal cell toxicity in rat PD models. 6–OHDA treatment reduced dopamine in the striatum and increased nigral iron load and apoptotic mediators. RA rescued dopamine neuron degeneration, eliminated excessive iron-reducing redox activity, and regulated Bcl2-/Bax gene expression. The study indicates the potential application of RA as iron-chelators to encounter neurodegenerative diseases. The accumulation of α-synuclein in the brain is associated with several neurodegenerative disorders. RA has been extensively studied for its effects on the formation of α-synuclein fibrils and degradation of preformed α-synuclein aggregates. The studies concluded that RA displayed anti-oxidant and anti-fibrillogenic features and might be a key candidate for ameliorating PD and similar diseases. In this regard, protective effects of RA were studied against 1-methyl-4-phenyl-1,2,3, 6-tetrahydropyridine (MPTP) induced neurotoxicity on a mouse model of PD. RA could prevent degeneration of the nigrostriatal dopaminergic system by downgrading nigral iron levels, increasing SOD and tyrosine hydroxylase positive neurons.
Furthermore, RA exposure inhibited iron-induced α-synuclein aggregation via upregulation of hemeoxygenase 1 and inhibited α-synuclein expression (Qu et al. 2019). Another study reported neuroprotective action of RA in MPTP-induced neurotoxicity in dopaminergic neurons of a zebrafish model of PD through regulating the DJ-1/Akt/Nrf2 signaling pathway (Zhao et al. 2020). These findings project mechanistic insights of RA-mediated attenuation in the development and progression of neurodegenerative disorders.
Moreover, RA amended depressive behavior in rats exposed to chronic unpredictable stress. The anti-depressant-like effect exerted by RA may be attributed to astrocytic BDNF expression in the hippocampal region via enhancement of ERK1/2 phosphorylation. However, further in-depth studies are needed to validate the detailed underlying mechanism of the anti-depressive properties of RA (Jin et al. 2013). Interestingly, in another study, RA exerted neuroprotective effects against focal ischemic stroke and post-stroke depression in vivo through augmenting anti-oxidant response (Wang et al. 2021c). As the reduction in glutamate release is crucial for neuroprotection, RA treatment significantly tapered glutamate release in concentration-dependent manner via activation of GABAA receptors in cerebrocortical synaptosomes (Wang et al. 2021a). These findings propose that RA might motivate the future development of innovative anti-depressant drugs. However, more clinical studies are required to develop RA as an anti-depressant on depressing subjects.
Anti-hypertensive potential
As the data suggest, phenolic compounds like RA exhibited significant anti-hypertensive effects in-various studies, making it an alternative therapeutic agent for hypertension treatment (Kwon et al. 2006). Recently Karthik et al. (2011) evaluated investigated cardioprotective and anti-hypertensive effects of RA in fructose-fed rats. RA supplementation reduced lipid levels, improved insulin sensitivity, and lowered blood pressure by decreasing endothelin-1 and angiotensin-converting enzyme activity. Additionally, histopathological analysis revealed reduced myocardial damage through its anti-oxidant properties. RA supplementation displayed cardioprotective effects against myocardial ischemia/reperfusion (I/R) injury through amelioration of NF-κB signaling pathway and ROS changes in male C57BL/6 J mice (Quan et al. 2021). In conjugation with extracts from Ocimum basiculum and Ocimum selloi, RA ameliorated angiotensin-II mediated damage and alleviated pro-inflammatory cytokines and macrophage chemotactic protein 1 levels (Alegría-Herrera et al. 2019). These findings suggest the multifaceted action of RA in blood pressure management and associated vascular damage.
Interestingly, RA derivative, ethyl rosmarinate, has been evaluated for effects on Nω-nitro-L-arginine methyl ester (L-NAME)—induced hypertensive rat models. The data indicated that ethyl rosmarinate significantly attenuated systolic blood pressure, increased eNOS production, and improved vascular function (Pantan et al. 2019). Another group of researchers studied the inhibitory effect of RA on angiotensin-converting enzyme activity and hypertension. RA treatment caused a dose-dependent decrease in systolic blood pressure in the hypertensive group and angiotensin-converting enzyme activity in rat lung tissue (Ferreira et al. 2018). Angiotensin-II is a potent agent that generates oxidative damage and hypertension. In concordance with previous studies, acute and chronic RA treatment attenuated angiotensin-II mediated rise in systolic, diastolic, and arterial blood pressure and cardiometabolic abnormalities in rats (Fig. 2). In addition, acute RA reduced fasting plasma glucose and induced glucose transport activity in skeletal muscle. Therefore, it can be concluded that RA and its derivatives can be considered exciting candidates for alternative treatment against angiotensin-II associated hypertension and hyperglycemia (Prasannarong et al. 2019).
Pharmacokinetic profile of RA
The pharmacokinetic profile of RA was initially reported in animal models, including mice and rats, and later in humans (Hitl et al. 2021). Several studies investigated the pharmacokinetics of RA and later concluded that pharmacokinetic parameters differ in humans and animals (Nakazawa and Ohsawa 1998; Matsuno et al. 2002; Baba et al. 2005; Gamaro et al. 2011; Nunes et al. 2017). A study in healthy humans was performed to determine RA's absorption, metabolism, and urinary excretion. Six healthy men were enrolled in the study involving single intakes of perilla extract containing 200 mg RA and placebo with a 10-days interval. RA and its related metabolites methylated RA, caffeic acid, and ferulic acid was detected in the plasma and urine after intake of PE (Baba et al. 2005). The proportion of RA and its related metabolites excreted in the urine was 6.3 ± 2.2% of the total dose, with approximately 75% of these components being excreted within 6 h after the intake of perilla extract (Baba et al. 2005). Another study shows that RA was well absorbed from the gastrointestinal tract and the skin (al-Sereiti et al. 1999). The toxic effect of RA on inflammatory and nociception in rats indicated its non-toxicity compared with the control group (Gamaro et al. 2011). The pharmacokinetic properties of RA calculated through the pkCSM web tool (http://biosig.unimelb.edu.au/pkcsm/) are given in Table 3.
Table 3.
Pharmacokinetic properties of RA calculated through pkCSM web tool
| Absorption | Distribution | Metabolism | Excretion | Toxicity | |||
|---|---|---|---|---|---|---|---|
| GI absorption | Skin Permeability (log Kp) | BBB/CNS permeability | CYP2D6 substrate/inhibitor | Clearance (log ml/min/kg) | Renal OCT2 substrate | AMES | Hepatotoxicity |
| Low | − 2.73 | No | No | 0.25 | No | No | No |
Clinical trials on RA
Several herbal apothecaries have attributed the flora with various human health benefits. In this regard, two similar double-blind placebo-controlled crossover studies were performed to assess the effect of M. Officinalis extracts containing RA on cognitive functions and mood modulation in healthy young adults. The study concluded that foodstuffs containing lemon balm extracts might have positive behavioral effects, implied in clinical health settings (Scholey et al. 2014).
As described previously, RA is known to display anti-inflammatory and anti-oxidant potential. In this regard, the protective functions of RA were investigated on subjects with atopic dermatitis. Twenty-one subjects with atopic dermatitis were enrolled in the study. Topical application of RA (0.3%) emulsion improved skin dryness, pruritus, and general AD symptoms, indicating that RA can be incorporated as a therapeutic agent for atopic dermatitis treatment. However, the precise molecular mechanism of RA-mediated improvement in atopic dermatitis remains to be explored (Lee et al. 2008). A randomized, double-blind study investigated the anti-inflammatory effects of RA obtained from spearmint extracts on individuals with knee osteoarthritis. The findings reported significant improvement in mobility and stiffness in adults with knee osteoarthritis on daily high RA spearmint tea consumption for 16 weeks (Connelly et al.2014).
Similarly, supplementation of spearmint extract rich in polyphenols, including RA, was investigated in a placebo study on cognitive functions, sleep pattern, and mood in human subjects with age-associated memory impairment. The study reported that consumption of 900 mg/day for 90 days improved working memory and spatial memory quality, sleep patterns, and mood status in subjects with age-associated memory impairment. Another similar trial reported that daily consumption of 900 mg of proprietary spearmint extract containing 14.5% RA and 24% total phenolic content improved reactive agility in the young population with no adverse effects (NCT02518165). These findings suggest the benefits of phenolic compounds, including RA, in individuals with memory impairment (Herrlinger et al. 2018). A study reported beneficial effects of RA in curing insomnia via hypnotic effects targeting A1R agonists (Kim et al. 2022). Few placebo-controlled trials have monitored the efficacy and safety of polyphenolic compounds for treating allergic inflammatory diseases. Takano et al. (2004) conducted a randomized, double-blind placebo-controlled study on subjects with mid-seasonal allergic rhinoconjunctivitis supplemented with extracts of Perilla frutescens enriched with RA. The RA supplemented group displayed attenuated polymorphonuclear leukocytes in the nostrils compared to the placebo group. No adverse events and significant abnormalities were observed upon RA administration. The above reports provide a comprehensive understanding of introducing phenolic compounds as alternative medicines in treating inflammatory diseases.
Dietary supplementation of O. stamineus RA enriched Nuvastatic TM (1000 mg) ameliorated oxidative damage restored mitochondrial and cellular functions bring down fatigue in subjects with solid stage I-IV tumors. Also, the neuroprotective effects of RA potentially deranged pain, sleep loss, lethargy, and other symptoms associated with cancer-related fatigue in Phase III (NCT04546607). In another trial, mint tea with high content of RA blunted chronic eosinophilic inflammation in adults with bilateral nasal polyps (NCT00465543). The safety and efficacy of RA treatment were compared to the placebo group receiving the inactive substance. Recently, a randomized, double-blind clinical study was conducted in Phase II on oral nutraceutical LertalR having RA as an active ingredient in conjugation with standard therapy (antihistamine) among children with allergic rhinoconjunctivitis. The polycentric study concluded that prolonged LertalR consumption significantly reduced allergic rhinoconjunctivitis exacerbation and reduced usage of rescue medications with no adverse events. Therefore, LertalR can be envisioned as an effective add-on treatment with standard therapy in allergic rhinoconjunctivitis in pediatrics (Marseglia et al. 2019) (NCT03365648).
Conclusion and future prospects
Ample evidences validate the potential benefits of RA as a nutraceutical and therapeutic candidate (Alavi et al. 2021). Pure RA-containing products are also available commercially. The diverse biological functions of phenolic compounds like RA can provide various health benefits (Özevren et al. 2020). RA acts as a powerful anti-oxidant agent mitigating oxidative damage, which could reduce the induction of ROS production via the enhancement of anti-oxidant defense enzymes (Khamse et al. 2020). RA and its derivatives have been extensively investigated as pharmaceutical agents against several diseases, including cancer, diabetes, neurodegenerative disorders, hypertension, and inflammatory diseases (Shamsi et al. 2020).
From ancient times, RA has been used as anti-microbial, anti-fungal, and anti-allergic substance in several remedies. Due to its anti-oxidant and anti-inflammatory properties, RA has been an attractive chemotherapeutic agent and has effectively controlled tumors in various stages (Waer et al. 2020). RA has been beneficial in alleviating several pathological characteristics of neurodegenerative disease, including protein aggregation, iron chelation, neuronal apoptosis, and oxidative stress. RA balances the anti-oxidant/oxidant system, protecting the cell from ROS-mediated damage through its anti-oxidant properties. Further investigations are needed to explore the mechanistic insights of RA-mediated protective functions in various experimental models. Though several studies reported a lack of adverse effects upon RA intake, few recorded complaints, such as dry mouth, itchy scalp, abdominal discomfort, and headaches had been reported (Connelly et al. 2014). Hence, the complete pharmacokinetic profile of RA should be thoroughly explored to determine its safety and efficacy in healthy subjects.
Acknowledgements
Institutional Fund Project funded this research work under the grant (IFPRP: 350-130-1442). Therefore, the authors gratefully acknowledge technical and financial support from the Ministry of Education and King Abdulaziz University, DSR, Jeddah, Saudi Arabia. SN thanks the Council of Scientific and Industrial Research for the award of CSIR-Research Associate Fellowship (Award No. 09/466(0238)/2019-EMRI).
Data availability
All data generated or analyzed during this study are included in this published article.
Declarations
Conflict of interest
The authors declare no conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Dharmendra Kumar Yadav, Email: dharmendra30oct@gmail.com.
Md Imtaiyaz Hassan, Email: mihassan@jmi.ac.in.
References
- Ahmed AS, Mona MM, Abdel-Kareem MA, Elsisy RA. Potential of rosmarinic acid to ameliorate toxic effects of diethyl methoxy thio-phosphoryl thio-succinate on albino wistar rats' lung, mast cell infiltration inhibitory pathway. Food Sci Nutr. 2021;9(7):3593–3601. doi: 10.1002/fsn3.2316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akhter J, Khan J, Baghel M, Beg MMA, Goswami P, Afjal MA, Ahmad S, Habib H, Najmi AK, Raisuddin S. NLRP3 inflammasome in rosmarinic acid-afforded attenuation of acute kidney injury in mice. Sci Rep. 2022;12(1):1313. doi: 10.1038/s41598-022-04785-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alagawany M, Abd El-Hack ME. The effect of rosemary herb as a dietary supplement on performance, egg quality, serum biochemical parameters, and oxidative status in laying hens. J Anim Feed Sci. 2015;24:341–347. doi: 10.22358/jafs/65617/2015. [DOI] [Google Scholar]
- Alam M, Ali S, Ahmed S, Elasbali AM, Adnan M, Islam A, Hassan MI, Yadav DK. Therapeutic potential of ursolic acid in cancer and diabetic neuropathy diseases. Int J Mol Sci. 2021;22(22):12162. doi: 10.3390/ijms222212162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alam M, Ali S, Ashraf GM, Bilgrami AL, Yadav DK, Hassan MI. Epigallocatechin 3-gallate: from green tea to cancer therapeutics. Food Chem. 2022;379:132135. doi: 10.1016/j.foodchem.2022.132135. [DOI] [PubMed] [Google Scholar]
- Alavi MS, Fanoudi S, Ghasemzadeh Rahbardar M, Mehri S, Hosseinzadeh H. An updated review of protective effects of rosemary and its active constituents against natural and chemical toxicities. Phytother Res. 2021;35(3):1313–1328. doi: 10.1002/ptr.6894. [DOI] [PubMed] [Google Scholar]
- Alegría-Herrera E, Herrera-Ruiz M, Román-Ramos R, Zamilpa A, Santillán-Urquiza MA, Aguilar MI, Avilés-Flores M, Fuentes-Mata M, Jiménez-Ferrer E. Effect of Ocimum basilicum, Ocimum selloi, and rosmarinic acid on cerebral vascular damage in a chronic hypertension model. Biol Pharm Bull. 2019;42:201–211. doi: 10.1248/bpb.b18-00574. [DOI] [PubMed] [Google Scholar]
- Ali S, Alam M, Khatoon F, Fatima U, Elasbali AM, Adnan M, Islam A, Hassan MI, Snoussi M, De Feo V. Natural products can be used in therapeutic management of COVID-19: probable mechanistic insights. Biomed Pharmacother. 2022;147:112658. doi: 10.1016/j.biopha.2022.112658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alrubaye A, Motovali-Bashi M, Miroliaei M. Rosmarinic acid inhibits DNA glycation and modulates the expression of Akt1 and Akt3 partially in the hippocampus of diabetic rats. Sci Rep. 2021;11:20605. doi: 10.1038/s41598-021-99286-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Sereiti MR, Abu-Amer KM, Sen P. Pharmacology of rosemary (Rosmarinus officinalis Linn.) and its therapeutic potentials. Indian J Exp Biol. 1999;37:124–130. [PubMed] [Google Scholar]
- An Y, Zhao J, Zhang Y, Wu W, Hu J, Hao H, Qiao Y, Tao Y, An L. Rosmarinic acid induces proliferation suppression of hepatoma cells associated with NF-κB signaling pathway. Asian Pac J Cancer Prev. 2021;22:1623–1632. doi: 10.31557/APJCP.2021.22.5.1623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anwar S, Shamsi A, Shahbaaz M, Queen A, Khan P, Hasan GM, Islam A, Alajmi MF, Hussain A, Ahmad F, Hassan MI. Rosmarinic acid exhibits anticancer effects via MARK4 inhibition. Sci Rep. 2020;10:10300. doi: 10.1038/s41598-020-65648-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Areti A, Komirishetty P, Kalvala AK, Nellaiappan K, Kumar A. Rosmarinic acid mitigates mitochondrial dysfunction and spinal glial activation in oxaliplatin-induced peripheral neuropathy. Mol Neurobiol. 2018;55:7463–7475. doi: 10.1007/s12035-018-0920-4. [DOI] [PubMed] [Google Scholar]
- Baba S, Osakabe N, Natsume M, Terao J. Orally administered rosmarinic acid is present as the conjugated and/or methylated forms in plasma, and is degraded and metabolized to conjugated forms of caffeic acid, ferulic acid and m-coumaric acid. Life Sci. 2004;75:165–178. doi: 10.1016/j.lfs.2003.11.028. [DOI] [PubMed] [Google Scholar]
- Baba S, Osakabe N, Natsume M, Yasuda A, Muto Y, Hiyoshi T, Takano H, Yoshikawa T, Terao J. Absorption, metabolism, degradation and urinary excretion of rosmarinic acid after intake of Perilla frutescens extract in humans. Eur J Nutr. 2005;44:1–9. doi: 10.1007/s00394-004-0482-2. [DOI] [PubMed] [Google Scholar]
- Bao T-Q, Li Y, Qu C, Zheng Z-G, Yang H, Li P. Antidiabetic effects and mechanisms of rosemary (Rosmarinus officinalis L.) and its phenolic components. Am J Chin Med. 2020;48:1353–1368. doi: 10.1142/S0192415X20500664. [DOI] [PubMed] [Google Scholar]
- Ben-Menachem E. Mechanism of action of vigabatrin: correcting misperceptions. Acta Neurol Scand. 2011;124:5–15. doi: 10.1111/j.1600-0404.2011.01596.x. [DOI] [PubMed] [Google Scholar]
- Bulgakov VP, Inyushkina YV, Fedoreyev SA. Rosmarinic acid and its derivatives: biotechnology and applications. Crit Rev Biotechnol. 2012;32:203–217. doi: 10.3109/07388551.2011.596804. [DOI] [PubMed] [Google Scholar]
- Chen Y-S, Yu H-M, Shie J-J, Cheng T-JR, Wu C-Y, Fang J-M, Wong C-H. Chemical constituents of Plectranthus amboinicus and the synthetic analogs possessing anti-inflammatory activity. Bioorg Med Chem. 2014;22:1766–1772. doi: 10.1016/j.bmc.2014.01.009. [DOI] [PubMed] [Google Scholar]
- Choi SJ, Oh SS, Kim CR, Kwon YK, Suh SH, Kim JK, Park GG, Son SY, Shin DH. Perilla frutescens extract ameliorates acetylcholinesterase and trimethyltin chloride-induced neurotoxicity. J Med Food. 2016;19:281–289. doi: 10.1089/jmf.2015.3540. [DOI] [PubMed] [Google Scholar]
- Chung DH, Kim NR, Kim T, Ahn J, Lee S, Lee YD, Cho HY. Malignant glomus tumor of the thyroid gland where is heretofore an unreported organ: a case report and literature review. Endocr Pathol. 2015;26:37–44. doi: 10.1007/s12022-014-9352-5. [DOI] [PubMed] [Google Scholar]
- Chung CH, Jung W, Keum H, Kim TW, Jon S. Nanoparticles derived from the natural antioxidant rosmarinic acid ameliorate acute inflammatory bowel disease. ACS Nano. 2020;14:6887–6896. doi: 10.1021/acsnano.0c01018. [DOI] [PubMed] [Google Scholar]
- Coelho VR, Vieira CG, de Souza LP, Moysés F, Basso C, Papke DKM, Pires TR, Siqueira IR, Picada JN, Pereira P. Antiepileptogenic, antioxidant and genotoxic evaluation of rosmarinic acid and its metabolite caffeic acid in mice. Life Sci. 2015;122:65–71. doi: 10.1016/j.lfs.2014.11.009. [DOI] [PubMed] [Google Scholar]
- Connelly AE, Tucker AJ, Tulk H, Catapang M, Chapman L, Sheikh N, Yurchenko S, Fletcher R, Kott LS, Duncan AM. High-rosmarinic acid spearmint tea in the management of knee osteoarthritis symptoms. J Med Food. 2014;17:1361–1367. doi: 10.1089/jmf.2013.0189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornejo A, Aguilar Sandoval F, Caballero L, Machuca L, Muñoz P, Caballero J, Perry G, Ardiles A, Areche C, Melo F. Rosmarinic acid prevents fibrillization and diminishes vibrational modes associated to β sheet in tau protein linked to Alzheimer’s disease. J Enzyme Inhib Med Chem. 2017;32:945–953. doi: 10.1080/14756366.2017.1347783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- da Silva GB, Yamauchi MA, Zanini D, Bagatini MD. Novel possibility for cutaneous melanoma treatment by means of rosmarinic acid action on purinergic signaling. Purinergic Signal. 2022;18:61–81. doi: 10.1007/s11302-021-09821-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Değer U, Çavuş Y. Investigation of the role of rosmarinic acid treatment in regulating inflammation, cell damage, and angiogenesis in rat ovarian torsion and detorsion models. Acta Cir Bras. 2020;35:e202000304. doi: 10.1590/s0102-865020200030000004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dewick PM. Medicinal natural products: a biosynthetic approach. Chicester: Wiley; 2002. [Google Scholar]
- Diao J, Zhao H, You P, You H, Wu H, Shou X, Cheng G. Rosmarinic acid ameliorated cardiac dysfunction and mitochondrial injury in diabetic cardiomyopathy mice via activation of the SIRT1/PGC-1α pathway. Biochem Biophys Res Commun. 2021;546:29–34. doi: 10.1016/j.bbrc.2021.01.086. [DOI] [PubMed] [Google Scholar]
- do Nascimento RF, de Oliveira Formiga R, Machado FDF, de Sales IRP, de Lima GM, Alves Júnior EB, Vieira GC, Pereira RF, de Araújo AA, de Araújo Junior RF, Barbosa Filho JM, Batista LM. Rosmarinic acid prevents gastric ulcers via sulfhydryl groups reinforcement, antioxidant and immunomodulatory effects. Naunyn Schmiedebergs Arch Pharmacol. 2020;393:2265–2278. doi: 10.1007/s00210-020-01894-2. [DOI] [PubMed] [Google Scholar]
- Dubois M, Bailly F, Mbemba G, Mouscadet J-F, Debyser Z, Witvrouw M, Cotelle P. Reaction of rosmarinic acid with nitrite ions in acidic conditions: discovery of nitro-and dinitrorosmarinic acids as new anti-HIV-1 agents. J Med Chem. 2008;51:2575–2579. doi: 10.1021/jm7011134. [DOI] [PubMed] [Google Scholar]
- Fachel FNS, Schuh RS, Veras KS, Bassani VL, Koester LS, Henriques AT, Braganhol E, Teixeira HF. An overview of the neuroprotective potential of rosmarinic acid and its association with nanotechnology-based delivery systems: a novel approach to treating neurodegenerative disorders. Neurochem Int. 2019;122:47–58. doi: 10.1016/j.neuint.2018.11.003. [DOI] [PubMed] [Google Scholar]
- Fachel FNS, Michels LR, Azambuja JH, Lenz GS, Gelsleichter NE, Endres M, Scholl JN, Schuh RS, Barschak AG, Figueiró F, Bassani VL, Henriques AT, Koester LS, Teixeira HF, Braganhol E. Chitosan-coated rosmarinic acid nanoemulsion nasal administration protects against LPS-induced memory deficit, neuroinflammation, and oxidative stress in Wistar rats. Neurochem Int. 2020;141:104875. doi: 10.1016/j.neuint.2020.104875. [DOI] [PubMed] [Google Scholar]
- Fan Y-T, Yin G-J, Xiao W-Q, Qiu L, Yu G, Hu Y-L, Xing M, Wu D-Q, Cang X-F, Wan R. Rosmarinic acid attenuates sodium taurocholate-induced acute pancreatitis in rats by inhibiting nuclear factor-κB activation. Am J Chin Med. 2015;43:1117–1135. doi: 10.1142/S0192415X15500640. [DOI] [PubMed] [Google Scholar]
- Fasolo JMMA, Vizuete AFK, Rico EP, Rambo RBS, Toson NSB, Santos E, de Oliveira DL, Gonçalves CAS, Schapoval EES, Heriques AT. Anti-inflammatory effect of rosmarinic acid isolated from Blechnum brasiliense in adult zebrafish brain. Comp Biochem Physiol C. 2021;239:108874. doi: 10.1016/j.cbpc.2020.108874. [DOI] [PubMed] [Google Scholar]
- Feng TY, Lv DL, Zhang X, Du YQ, Yuan YT, Chen MJ, Xi HM, Li Y, Han N, Hu JH. Rosmarinic acid improves boar sperm quality, antioxidant capacity and energy metabolism at 17°C via AMPK activation. Reprod Domest Anim. 2020;55(12):1714–1724. doi: 10.1111/rda.13828. [DOI] [PubMed] [Google Scholar]
- Ferreira LG, Evora PRB, Capellini VK, Albuquerque AA, Carvalho MTM, da Silva Gomes RA, Parolini MT, Celotto AC. Effect of rosmarinic acid on the arterial blood pressure in normotensive and hypertensive rats: role of ACE. Phytomedicine. 2018;38:158–165. doi: 10.1016/j.phymed.2017.02.006. [DOI] [PubMed] [Google Scholar]
- Formiga RO, Alves Júnior EB, Vasconcelos RC, Guerra GCB, Antunes de Araújo A, Carvalho TG, Garcia VB, de Araújo Junior RF, Gadelha FAAF, Vieira GC, Sobral MV, Barbosa Filho JM, Spiller F, Batista LM. Cymene and rosmarinic acid ameliorate TNBS-induced intestinal inflammation upkeeping ZO-1 and MUC-2: role of antioxidant system and immunomodulation. Int J Mol Sci. 2020;21:5870. doi: 10.3390/ijms21165870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujimoto A, Shingai Y, Nakamura M, Maekawa T, Sone Y, Masuda T. A novel ring-expanded product with enhanced tyrosinase inhibitory activity from classical Fe-catalyzed oxidation of rosmarinic acid, a potent anti-oxidative Lamiaceae polyphenol. Bioorg Med Chem Lett. 2010;20:7393–7396. doi: 10.1016/j.bmcl.2010.10.040. [DOI] [PubMed] [Google Scholar]
- Fuster MG, Carissimi G, Montalbán MG, Víllora G. Antitumor activity of rosmarinic acid-loaded silk fibroin nanoparticles on HeLa and MCF-7 cells. Polymers. 2021;13:3169. doi: 10.3390/polym13183169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gamaro GD, Suyenaga E, Borsoi M, Lermen J, Pereira P, Ardenghi P. Effect of rosmarinic and caffeic acids on inflammatory and nociception process in rats. Int Sch Res Not. 2011 doi: 10.5402/2011/451682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gautam RK, Gupta G, Sharma S, Hatware K, Patil K, Sharma K, Goyal S, Chellappan DK, Dua K. Rosmarinic acid attenuates inflammation in experimentally induced arthritis in Wistar rats, using Freund's complete adjuvant. Int J Rheum Dis. 2019;22:1247–1254. doi: 10.1111/1756-185X.13602. [DOI] [PubMed] [Google Scholar]
- Ghasemzadeh RM, Hosseinzadeh H. Effects of rosmarinic acid on nervous system disorders: an updated review. Naunyn Schmiedebergs Arch Pharmacol. 2020;393(10):1779–1795. doi: 10.1007/s00210-020-01935-w. [DOI] [PubMed] [Google Scholar]
- Ghorbani A, Sadeghnia HR, Afshari AR, Hosseini A. Rosmarinic acid protects adipose tissue-derived mesenchymal stem cells in nutrient-deficient conditions. Prev Nutr Food Sci. 2019;24:449–455. doi: 10.3746/pnf.2019.24.4.449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibb W, Lees A (1988) The relevance of the Lewy body to the pathogenesis of idiopathic Parkinson's disease. J Neurol Neurosurg Psych 51:745–752 [DOI] [PMC free article] [PubMed]
- Gonçalves C, Fernandes D, Silva I, Mateus V. Potential anti-inflammatoryeffect of Rosmarinus officinalis in preclinical in vivo models of inflammation. Molecules. 2022;27:609. doi: 10.3390/molecules27030609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guan M, Guo L, Ma H, Wu H, Fan X. Network pharmacology and molecular docking suggest the mechanism for biological activity of rosmarinic acid. Evid Based Complement Altern Med. 2021;2021:5190808. doi: 10.1155/2021/5190808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gui H, Jin Y, Lin A, Wang P, Wang Y, Zhu H. Rosmarinic acid relieves cisplatin-induced ovary toxicity in female mice via suppression of oxidative stress and inflammation. J Biochem Mol Toxicol. 2021;35:e22839. doi: 10.1002/jbt.22839. [DOI] [PubMed] [Google Scholar]
- Guo C, Shangguan Y, Zhang M, Ruan Y, Xue G, Ma J, Yang J, Qiu L. Rosmarinic acid alleviates ethanol-induced lipid accumulation by repressing fatty acid biosynthesis. Food Funct. 2020;11:2094–2106. doi: 10.1039/c9fo02357g. [DOI] [PubMed] [Google Scholar]
- Gupta D, Archoo S, Naikoo SH, Abdullah ST. Rosmarinic acid: a naturally occurring plant based agent prevents impaired mitochondrial dynamics and apoptosis in ultraviolet-b-irradiated human skin cells. Photochem Photobiol. 2021 doi: 10.1111/php.13533. [DOI] [PubMed] [Google Scholar]
- Hajhosseini L, Khaki A, Merat E, Ainehchi N. Effect of rosmarinic acid on sertoli cells apoptosis and serum antioxidant levels in rats after exposure to electromagnetic fields. Afr J Tradit Complement Altern Med. 2013;10:477–480. doi: 10.4314/ajtcam.v10i6.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasanein P, Seifi R, Hajinezhad MR, Emamjomeh A. Rosmarinic acid protects against chronic ethanol-induced learning and memory deficits in rats. Nutr Neurosci. 2017;20:547–554. doi: 10.1080/1028415X.2016.1203125. [DOI] [PubMed] [Google Scholar]
- Hase T, Shishido S, Yamamoto S, Yamashita R, Nukima H, Taira S, Toyoda T, Abe K, Hamaguchi T, Ono K. Rosmarinic acid suppresses Alzheimer's disease development by reducing amyloid β aggregation by increasing monoamine secretion. Sci Rep. 2019;9:1–13. doi: 10.1038/s41598-019-45168-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hassanzadeh-Taheri M, Ahmadi-Zohan A, Mohammadifard M, Hosseini M. Rosmarinic acid attenuates lipopolysaccharide-induced neuroinflammation and cognitive impairment in rats. J Chem Neuroanat. 2021;117:102008. doi: 10.1016/j.jchemneu.2021.102008. [DOI] [PubMed] [Google Scholar]
- Heidari F, Komeili-Movahhed T, Hamidizad Z, Moslehi A. The protective effects of rosmarinic acid on ethanol-induced gastritis in male rats: antioxidant defense enhancement. Res Pharm Sci. 2021;16:305–314. doi: 10.4103/1735-5362.314829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrlinger KA, Nieman KM, Sanoshy KD, Fonseca BA, Lasrado JA, Schild AL, Maki KC, Wesnes KA, Ceddia MA. Spearmint extract improves working memory in men and women with age-associated memory impairment. J Altern Complement Med. 2018;24:37–47. doi: 10.1089/acm.2016.0379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hitl M, Kladar N, Gavarić N, Božin B. Rosmarinic acid–human pharmacokinetics and health benefits. Planta Med. 2021;87:273–282. doi: 10.1055/a-1301-8648. [DOI] [PubMed] [Google Scholar]
- Hsieh CF, Jheng JR, Lin GH, Chen YL, Ho JY, Liu CJ, Hsu KY, Chen YS, Chan YF, Yu HM, Hsieh PW, Chern JH, Horng JT. Rosmarinic acid exhibits broad anti-enterovirus A71 activity by inhibiting the interaction between the five-fold axis of capsid VP1 and cognate sulfated receptors. Emerg Microbes Infect. 2020;9(1):1194–1205. doi: 10.1080/22221751.2020.1767512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu Y-C, Cheng C-P, Chang D-M. Plectranthus amboinicus attenuates inflammatory bone erosion in mice with collagen-induced arthritis by downregulation of RANKL-induced NFATc1 expression. J Rheumatol. 2011;38:1844–1857. doi: 10.3899/jrheum.101223. [DOI] [PubMed] [Google Scholar]
- Hu Z-N, Huang L-J, Chen W-P. The inhibitory effects of rosmarinic acid on catabolism induced by IL-1β in rat chondrocyte. Acta Biochim Pol. 2018;65:535–538. doi: 10.18388/abp.2018_2607. [DOI] [PubMed] [Google Scholar]
- Huang L, Chen J, Quan J, Xiang D. Rosmarinic acid inhibits proliferation and migration, promotes apoptosis and enhances cisplatin sensitivity of melanoma cells through inhibiting ADAM17/EGFR/AKT/GSK3β axis. Bioengineered. 2021;12:3065–3076. doi: 10.1080/21655979.2021.1941699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huerta-Madroñal M, Caro-León J, Espinosa-Cano E, Aguilar MR, Chitosan VLB. Rosmarinic acid conjugates with antioxidant, anti-inflammatory and photoprotective properties. Carbohydr Polym. 2021;273:118619. doi: 10.1016/j.carbpol.2021.118619. [DOI] [PubMed] [Google Scholar]
- Hur Y-G, Suh C-H, Kim S, Won J. Rosmarinic acid induces apoptosis of activated T cells from rheumatoid arthritis patients via mitochondrial pathway. J Clin Immunol. 2007;27:36–45. doi: 10.1007/s10875-006-9057-8. [DOI] [PubMed] [Google Scholar]
- Hwang H, Park YN, Shim YS, Byun SS, Kim HS. Characteristic dynamic enhancement pattern of MR imaging for malignant thyroid tumor. Preliminary report. Neuroradiol J. 2011;24:392–394. doi: 10.1177/197140091102400307. [DOI] [PubMed] [Google Scholar]
- Ilhan N, Bektas I, Susam S, Ozercan IH. Protective effects of rosmarinic acid against azoxymethane-induced colorectal cancer in rats. J Biochem Mol Toxicol. 2022;36:e22961. doi: 10.1002/jbt.22961. [DOI] [PubMed] [Google Scholar]
- Iuvone T, De Filippis D, Esposito G, D'Amico A, Izzo AA. The spice sage and its active ingredient rosmarinic acid protect PC12 cells from amyloid-β peptide-induced neurotoxicity. J Pharmacol Exp Ther. 2006;317:1143–1149. doi: 10.1124/jpet.105.099317. [DOI] [PubMed] [Google Scholar]
- Jafaripour L, Naserzadeh R, Alizamani E, Javad Mashhadi SM, Moghadam ER, Nouryazdan N, Ahmadvand H. Effects of rosmarinic acid on methotrexate-induced nephrotoxicity and hepatotoxicity in wistar rats. Indian J Nephrol. 2021;31:218–224. doi: 10.4103/ijn.IJN_14_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jain SV, Bhadoriya KS, Bari SB. QSAR and flexible docking studies of some aldose reductase inhibitors obtained from natural origin. Med Chem Res. 2012;21:1665–1676. doi: 10.1007/s00044-011-9681-6. [DOI] [Google Scholar]
- Jang Y-G, Hwang K-A, Choi K-C. Rosmarinic acid, a component of rosemary tea, induced the cell cycle arrest and apoptosis through modulation of HDAC2 expression in prostate cancer cell lines. Nutrients. 2018;10:1784. doi: 10.3390/nu10111784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jelić D, Mildner B, Koštrun S, Nujić K, Verbanac D, Čulić O, Antolović R, Brandt W. Homology modeling of human Fyn kinase structure: discovery of rosmarinic acid as a new Fyn kinase inhibitor and in silico study of its possible binding modes. J Med Chem. 2007;50:1090–1100. doi: 10.1021/jm0607202. [DOI] [PubMed] [Google Scholar]
- Jeong JH, Hwang JH. Herbal medicine for the prevention of sarcopenia: a protocol for systematic review. Medicine. 2021;100:e25474. doi: 10.1097/MD.0000000000025474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeong S, Park H, Hong S, Yum S, Kim W, Jung Y. Lipophilic modification enhances anti-colitic properties of rosmarinic acid by potentiating its HIF-prolyl hydroxylases inhibitory activity. Eur J Pharmacol. 2015;747:114–122. doi: 10.1016/j.ejphar.2014.11.030. [DOI] [PubMed] [Google Scholar]
- Jeong MJ, Lim DS, Kim SO, Park C, Choi YH, Jeong SJ. Effect of rosmarinic acid on differentiation and mineralization of MC3T3-E1 osteoblastic cells on titanium surface. Anim Cells Syst. 2021;25:46–55. doi: 10.1080/19768354.2021.1886987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin X, Liu P, Yang F, Y-h Z, Miao D. Rosmarinic acid ameliorates depressive-like behaviors in a rat model of CUS and Up-regulates BDNF levels in the hippocampus and hippocampal-derived astrocytes. Neurochem Res. 2013;38:1828–1837. doi: 10.1007/s11064-013-1088-y. [DOI] [PubMed] [Google Scholar]
- Jin B-R, Chung K-S, Cheon S-Y, Lee M, Hwang S, Hwang SN, Rhee K-J, An H-J. Rosmarinic acid suppresses colonic inflammation in dextran sulphate sodium (DSS)-induced mice via dual inhibition of NF-κB and STAT3 activation. Sci Rep. 2017;7:1–11. doi: 10.1038/srep46252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin B, Liu J, Gao D, Xu Y, He L, Zang Y, Li N, Lin D. Detailed studies on the anticancer action of rosmarinic acid in human Hep-G2 liver carcinoma cells: evaluating its effects on cellular apoptosis, caspase activation and suppression of cell migration and invasion. J BUON. 2020;25:1383–1389. [PubMed] [Google Scholar]
- Jin BR, Chung KS, Hwang S, Hwang SN, Rhee KJ, Lee M, An HJ. Rosmarinic acid represses colitis-associated colon cancer: a pivotal involvement of the TLR4-mediated NF-κB-STAT3 axis. Neoplasia. 2021;23:561–573. doi: 10.1016/j.neo.2021.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin C, Jang BH, Jeon JP, Lee YS, Yang SB, Kwon S. Traditional east Asian herbal medicines for the treatment of poststroke constipation: a protocol for systematic review and meta analysis. Medicine. 2021;100:e25503 . doi: 10.1097/MD.0000000000025503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karthik D, Viswanathan P, Anuradha CV. Administration of rosmarinic acid reduces cardiopathology and blood pressure through inhibition of p22phox NADPH oxidase in fructose-fed hypertensive rats. J Cardiovasc Pharmacol. 2011;58:514–521. doi: 10.1097/FJC.0b013e31822c265d. [DOI] [PubMed] [Google Scholar]
- Khalaf AA, Hassanen EI, Ibrahim MA, Tohamy AF, Aboseada MA, Hassan HM, Zaki AR. Rosmarinic acid attenuates chromium-induced hepatic and renal oxidative damage and DNA damage in rats. J Biochem Mol Toxicol. 2020;34:e22579. doi: 10.1002/jbt.22579. [DOI] [PubMed] [Google Scholar]
- Khamse S, Sadr SS, Roghani M, Rashvand M, Mohammadian M, Marefati N, Harati E, Ebrahimi F. The effect of rosmarinic acid on apoptosis and nNOS immunoreactivity following intrahippocampal kainic acid injections in rats. Basic Clin Neurosci. 2020;11:41–48. doi: 10.32598/bcn.9.10.340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khojasteh A, Mirjalili MH, Alcalde MA, Cusido RM, Eibl R, Palazon J. Powerful plant antioxidants: a new biosustainable approach to the production of rosmarinic acid. Antioxidants. 2020;9(12):1273. doi: 10.3390/antiox9121273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim NR, Chung DH, Park CY, Ha SY. Malignant peripheral nerve sheath tumor of the uterine cervix expressing both S-100 protein and HMB-45. J Obstet Gynaecol Res. 2009;35:1136–1141. doi: 10.1111/j.1447-0756.2009.01078.x. [DOI] [PubMed] [Google Scholar]
- Kim KH, Moon E, Choi SU, Pang C, Kim SY, Lee KR. Identification of cytotoxic and anti-inflammatory constituents from the bark of Toxicodendron vernicifluum (Stokes) F.A. Barkley. J Ethnopharmacol. 2015;162:231–237. doi: 10.1016/j.jep.2014.12.07. [DOI] [PubMed] [Google Scholar]
- Kim KH, Jang S, Lee JA, Jang BH, Go HY, Park S, Jo HG, Lee MS, Ko SG. Experiences providing medical assistance during the sewol ferry disaster using traditional korean medicine. Evid Based Complement Altern Med. 2017;2017:3203768. doi: 10.1155/2017/3203768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim HK, Hwang S, Sung B, Kim YH, Chang Y. Gd-Complex of a rosmarinic acid conjugate as an anti-inflammatory theranostic agent via reactive oxygen species scavenging. Antioxidants. 2020;9:744. doi: 10.3390/antiox9080744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim KW, Cho HJ, Khaliq SA, Son KH, Yoon MS. Comparative analyses of mTOR/Akt and muscle atrophy-related signaling in aged respiratory and gastrocnemius muscles. Int J Mol Sci. 2020 doi: 10.3390/ijms21082862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SM, Park EJ, Kim JY, Choi J, Lee HJ. Anti-inflammatory effects of fermented lotus root and linoleic acid in lipopolysaccharide-induced RAW 264.7 cells. Life. 2020 doi: 10.3390/life10110293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim D, Kim HJ, Baek JO, Roh JY, Jun HS. Lysophosphatidic acid mediates imiquimod-induced psoriasis-like symptoms by promoting keratinocyte proliferation through LPAR1/ROCK2/PI3K/AKT signaling pathway. Int J Mol Sci. 2021 doi: 10.3390/ijms221910777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JS, Lee JH, Hong SM, Cho K, Kim SY. Salvia miltiorrhiza prevents methylglyoxal-induced glucotoxicity via the regulation of apoptosis-related pathways and the glyoxalase system in human umbilical vein endothelial cells. Biol Pharm Bull. 2021 doi: 10.1248/bpb.b21-00507. [DOI] [PubMed] [Google Scholar]
- Kim SW, Kim YI, Mustafa B, Kim MJ, Jeong G, Ahn SM, Lim SB, Yu CS, Kim JC, Hong SM, Park IJ. Distribution pattern of tumor infiltrating lymphocytes and tumor microenvironment composition as prognostic indicators in anorectal malignant melanoma. Mod Pathol. 2021;34:141–160. doi: 10.1038/s41379-020-0633-x. [DOI] [PubMed] [Google Scholar]
- Kim TH, Bormate KJ, Custodio RJP, Cheong JH, Lee BK, Kim HJ, Jung YS. Involvement of the adenosine A1 receptor in the hypnotic effect of rosmarinic acid. Biomed Pharmacother. 2022;146:112483. doi: 10.1016/j.biopha.2021.112483. [DOI] [PubMed] [Google Scholar]
- Ko YH, Shim KY, Kim SK, Seo JY, Lee BR, Hur KH, Kim YJ, Kim SE, Do MH, Parveen A, Kim SY, Lee SY, Jang CG. Lespedeza bicolor extract improves amyloid beta25—35-induced memory impairments by upregulating BDNF and activating Akt, ERK, and CREB signaling in mice. Planta Med. 2019;85:1363–1373. doi: 10.1055/a-1018-5402. [DOI] [PubMed] [Google Scholar]
- Kola A, Hecel A, Lamponi S, Valensin D. Novel perspective on Alzheimer's disease treatment: rosmarinic acid molecular interplay with copper(II) and amyloid β. Life. 2020;10:118. doi: 10.3390/life10070118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Komeili-Movahhed T, Bassirian M, Changizi Z, Moslehi A. SIRT1/NFκB pathway mediates anti-inflammatory and anti-apoptotic effects of rosmarinic acid on in a mouse model of nonalcoholic steatohepatitis (NASH) J Recept Signal Transduct Res. 2021;31:1–10. doi: 10.1080/10799893.2021.1905665. [DOI] [PubMed] [Google Scholar]
- Koprivica I, Jonić N, Diamantis D, Gajić D, Saksida T, Pejnović N, Tzakos AG, Stojanović I. Phenethyl ester of rosmarinic acid attenuates autoimmune responses during type 1 diabetes development in mice. Life Sci. 2022;288:120184. doi: 10.1016/j.lfs.2021.120184. [DOI] [PubMed] [Google Scholar]
- Koukoulitsa C, Hadjipavlou-Litina D, Geromichalos GD, Skaltsa H. Inhibitory effect on soybean lipoxygenase and docking studies of some secondary metabolites, isolated from Origanum vulgare L. ssp. hirtum. J Enzyme Inhib Med Chem. 2007;22:99–104. doi: 10.1080/14756360600991017. [DOI] [PubMed] [Google Scholar]
- Koukoulitsa C, Bailly F, Pegklidou K, Demopoulos VJ, Cotelle P. Evaluation of aldose reductase inhibition and docking studies of 6′-nitro and 6′, 6 ″-dinitrorosmarinic acids. Eur J Med Chem. 2010;45:1663–1666. doi: 10.1016/j.ejmech.2009.12.007. [DOI] [PubMed] [Google Scholar]
- Kumar V, Sami N, Kashav T, Islam A, Ahmad F, Hassan MI. Protein aggregation and neurodegenerative diseases: from theory to therapy. Eur J Med Chem. 2016;124:1105–1120. doi: 10.1016/j.ejmech.2016.07.054. [DOI] [PubMed] [Google Scholar]
- Kumar D, Ashraf GM, Hassan MI. Therapeutic integrity of microbiome-based medicines in neurodegenerative disorders. Curr Neuropharmacol. 2022 doi: 10.2174/1570159X20666220214113816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon Y-II, Vattem DA, Shetty K. Evaluation of clonal herbs of Lamiaceae species for management of diabetes and hypertension. Asia Pac J Clin Nutr. 2006;15:107. [PubMed] [Google Scholar]
- Kwon J, Seo YH, Lee J-E, Seo E-K, Li S, Guo Y, Hong S-B, Park S-Y, Lee D. Spiroindole alkaloids and spiroditerpenoids from Aspergillus duricaulis and their potential neuroprotective effects. J Nat Prod. 2015;78:2572–2579. doi: 10.1021/acs.jnatprod.5b00508. [DOI] [PubMed] [Google Scholar]
- Lataliza AAB, de Assis PM, da Rocha LL, Gonçalves ECD, Raposo NRB, Dutra RC. Antidepressant-like effect of rosmarinic acid during LPS-induced neuroinflammatory model: the potential role of cannabinoid receptors/PPAR-γ signaling pathway. Phytother Res. 2021;35:6974–6989. doi: 10.1002/ptr.7318. [DOI] [PubMed] [Google Scholar]
- Lee J, Jung E, Koh J, Kim YS, Park D. Effect of rosmarinic acid on atopic dermatitis. J Dermatol. 2008;35:768–771. doi: 10.1111/j.1346-8138.2008.00565.x. [DOI] [PubMed] [Google Scholar]
- Lee JH, Parveen A, Do MH, Lim Y, Shim SH, Kim SY. Lespedeza cuneata protects the endothelial dysfunction via eNOS phosphorylation of PI3K/Akt signaling pathway in HUVECs. Phytomedicine. 2018;48:1–9. doi: 10.1016/j.phymed.2018.05.005. [DOI] [PubMed] [Google Scholar]
- Lee YJ, Jung O, Lee J, Son J, Cho JY, Ryou C, Lee SY. Maclurin exerts anti-cancer effects on PC3 human prostate cancer cells via activation of p38 and inhibitions of JNK, FAK, AKT, and c-Myc signaling pathways. Nutr Res. 2018;58:62–71. doi: 10.1016/j.nutres.2018.07.003. [DOI] [PubMed] [Google Scholar]
- Lee BK, Jee HJ, Jung YS. Aβ1-40-induced platelet adhesion is ameliorated by rosmarinic acid through inhibition of NADPH oxidase/PKC-δ/integrin αIIbβ3 signaling. Antioxidants. 2021;23(10):1671. doi: 10.3390/antiox10111671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li F-R, Fu Y-Y, Jiang D-H, Wu Z, Zhou Y-J, Guo L, Dong Z-M, Wang Z-Z. Reversal effect of rosmarinic acid on multidrug resistance in SGC7901/Adr cell. J Asian Nat Prod Res. 2013;15:276–285. doi: 10.1080/10286020.2012.762910. [DOI] [PubMed] [Google Scholar]
- Li Z, Wang X, Zhang H. Balancing the non-linear rosmarinic acid biosynthetic pathway by modular co-culture engineering. Metab Eng. 2019;54:1–11. doi: 10.1016/j.ymben.2019.03.002. [DOI] [PubMed] [Google Scholar]
- Liang Z, Nie H, Xu Y, Peng J, Zeng Y, Wei Y, Wen X, Qiu J, Zhong W, Deng X. Therapeutic effects of rosmarinic acid on airway responses in a murine model of asthma. Int Immunopharmacol. 2016;41:90–97. doi: 10.1016/j.intimp.2016.10.010. [DOI] [PubMed] [Google Scholar]
- Liang Z, Wu L, Deng X, Liang Q, Xu Y, Deng R, Lv L, Ji M, Hao Z, He J. The antioxidant rosmarinic acid ameliorates oxidative lung damage in experimental allergic asthma via modulation of NADPH oxidases and antioxidant enzymes. Inflammation. 2020;43:1902–1912. doi: 10.1007/s10753-020-01264-3. [DOI] [PubMed] [Google Scholar]
- Liao XZ, Gao Y, Sun LL, Liu JH, Chen HR, Yu L, Chen ZZ, Chen WH, Lin LZ. Rosmarinic acid reverses non-small cell lung cancer cisplatin resistance by activating the MAPK signaling pathway. Phytother Res. 2020;34:1142–1153. doi: 10.1002/ptr.6584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim SH, Nam KH, Kim K, Yi SA, Lee J, Han JW. Rosmarinic acid methyl ester regulates ovarian cancer cell migration and reverses cisplatin resistance by inhibiting the expression of forkhead box M1. Pharmaceuticals. 2020;13:302. doi: 10.3390/ph13100302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin X, Watanabe K, Kuragano M, Kurotaki Y, Nakanishi U, Tokuraku K. Dietary intake of rosmarinic acid increases serum inhibitory activity in amyloid a aggregation and suppresses deposition in the organs of mice. Int J Mol Sci. 2020;21:6031. doi: 10.3390/ijms21176031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Xu X, Tang H, Pan Y, Hu B, Huang G. Rosmarinic acid inhibits cell proliferation, migration, and invasion and induces apoptosis in human glioma cells. Int J Mol Med. 2021;47:67. doi: 10.3892/ijmm.2021.4900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu P, Sui M, Zhang M, Wang M, Kamiya T, Okamoto K, Itoh H, Okuda S, Suzuki M, Asakura T, Fujiwara T, Nagata K. Rosmarinic acid and sodium citrate have a synergistic bacteriostatic effect against Vibrio species by inhibiting iron uptake. Int J Mol Sci. 2021;22(23):13010. doi: 10.3390/ijms222313010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luan H, Kan Z, Xu Y, Lv C, Jiang W. Rosmarinic acid protects against experimental diabetes with cerebral ischemia: relation to inflammation response. J Neuroinflamm. 2013;10:1–10. doi: 10.1186/1742-2094-10-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luft JG, Steffens L, Morás AM, da Rosa MS, Leipnitz G, Regner GG, Pflüger PF, Gonçalves D, Moura DJ, Pereira P. Rosmarinic acid improves oxidative stress parameters and mitochondrial respiratory chain activity following 4-aminopyridine and picrotoxin-induced seizure in mice. Naunyn Schmiedebergs Arch Pharmacol. 2019;392:1347–1358. doi: 10.1007/s00210-019-01675-6. [DOI] [PubMed] [Google Scholar]
- Luo C, Zou L, Sun H, Peng J, Gao C, Bao L, Ji R, Jin Y, Sun S. A review of the anti-inflammatory effects of rosmarinic acid on inflammatory diseases. Front Pharmacol. 2020;11:153. doi: 10.3389/fphar.2020.00153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo Y, Ma Z, Xu X, Qi H, Cheng Z, Chen L. Anticancer effects of rosmarinic acid in human oral cancer cells is mediated via endoplasmic reticulum stress, apoptosis, G2/M cell cycle arrest and inhibition of cell migration. J BUON. 2020;25:1245–1250. [PubMed] [Google Scholar]
- Luo C, Sun H, Peng J, Gao C, Bao L, Ji R, Zhang C, Zhu W, Jin Y. Rosmarinic acid exerts an antagonistic effect on nonalcoholic fatty liver disease by regulating the YAP1/TAZ-PPARγ/PGC-1α signaling pathway. Phytother Res. 2021;35:1010–1022. doi: 10.1002/ptr.6865. [DOI] [PubMed] [Google Scholar]
- Lv R, Du L, Zhou F, Yuan X, Liu X, Zhang L. Rosmarinic acid alleviates inflammation, apoptosis, and oxidative stress through regulating miR-155-5p in a mice model of Parkinson's disease. ACS Chem Neurosci. 2020;11:3259–3266. doi: 10.1021/acschemneuro.0c00375. [DOI] [PubMed] [Google Scholar]
- Ma Z, Lu Y, Yang F, Li S, He X, Gao Y, Zhang G, Ren E, Wang Y, Kang X. Rosmarinic acid exerts a neuroprotective effect on spinal cord injury by suppressing oxidative stress and inflammation via modulating the Nrf2/HO-1 and TLR4/NF-κB pathways. Toxicol Appl Pharmacol. 2020;397:115014. doi: 10.1016/j.taap.2020.115014. [DOI] [PubMed] [Google Scholar]
- Ma Z, Yang J, Yang Y, Wang X, Chen G, Shi A, Lu Y, Jia S, Kang X, Lu L. Rosmarinic acid exerts an anticancer effect on osteosarcoma cells by inhibiting DJ-1 via regulation of the PTEN-PI3K-Akt signaling pathway. Phytomedicine. 2020;68:153186. doi: 10.1016/j.phymed.2020.153186. [DOI] [PubMed] [Google Scholar]
- Mahmoud MA, Okda TM, Omran GA, Abd-Alhaseeb MM. Rosmarinic acid suppresses inflammation, angiogenesis, and improves paclitaxel induced apoptosis in a breast cancer model via NF3 κB-p53-caspase-3 pathways modulation. J Appl Biomed. 2021;19:202–209. doi: 10.32725/jab.2021.024. [DOI] [PubMed] [Google Scholar]
- Marchev AS, Vasileva LV, Amirova KM, Savova MS, Koycheva IK, Balcheva-Sivenova ZP, Vasileva SM, Georgiev MI. Rosmarinic acid-from bench to valuable applications in food industry. Trends Food Sci Technol. 2021;117:182–193. doi: 10.1016/j.tifs.2021.03.015. [DOI] [Google Scholar]
- Marinho S, Illanes M, Ávila-Román J, Motilva V, Talero E. Anti-inflammatory effects of rosmarinic acid-loaded nanovesicles in acute colitis through modulation of NLRP3 inflammasome. Biomolecules. 2021;11:162. doi: 10.3390/biom11020162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marseglia G, Licari A, Leonardi S, Papale M, Zicari AM, Schiavi L, Ciprandi G. A polycentric, randomized, parallel-group, study on Lertal®, a multicomponent nutraceutical, as preventive treatment in children with allergic rhinoconjunctivitis: phase II. Ital J Pediatr. 2019;45:1–6. doi: 10.1186/s13052-019-0678-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuno M, Nagatsu A, Ogihara Y, Ellis BE, Mizukami H. CYP98A6 from Lithospermum erythrorhizon encodes 4-coumaroyl-4′-hydroxyphenyllactic acid 3-hydroxylase involved in rosmarinic acid biosynthesis. FEBS Lett. 2002;514:219–224. doi: 10.1016/S0014-5793(02)02368-2. [DOI] [PubMed] [Google Scholar]
- Mazumder A, Neamati N, Sunder S, Schulz J, Pertz H, Eich E, Pommier Y. Curcumin analogs with altered potencies against HIV-1 integrase as probes for biochemical mechanisms of drug action. J Med Chem. 1997;40:3057–3063. doi: 10.1021/jm970190x. [DOI] [PubMed] [Google Scholar]
- McCue PP, Shetty K. Inhibitory effects of rosmarinic acid extracts on porcine pancreatic amylase in vitro. Asia Pac J Clin Nutr. 2004;13:101–106. [PubMed] [Google Scholar]
- Messeha SS, Zarmouh NO, Asiri A, Soliman KFA. Rosmarinic acid-induced apoptosis and cell cycle arrest in triple-negative breast cancer cells. Eur J Pharmacol. 2020;885:173419. doi: 10.1016/j.ejphar.2020.173419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michalikova D, Tyukos Kaprinay B, Brnoliakova Z, Sasvariova M, Krenek P, Babiak E, Frimmel K, Bittner Fialova S, Stankovicova T, Sotnikova R, Gasparova Z. Impact of improving eating habits and rosmarinic acid supplementation on rat vascular and neuronal system in the metabolic syndrome model. Br J Nutr. 2020;20:1–11. doi: 10.1017/S000711452000327X. [DOI] [PubMed] [Google Scholar]
- Mirza FJ, Amber S, Sumera HD, Ahmed T, Zahid S. Rosmarinic acid and ursolic acid alleviate deficits in cognition, synaptic regulation and adult hippocampal neurogenesis in an Aβ1-42-induced mouse model of Alzheimer's disease. Phytomedicine. 2021;83:153490. doi: 10.1016/j.phymed.2021.153490. [DOI] [PubMed] [Google Scholar]
- Murata T, Sasaki K, Sato K, Yoshizaki F, Yamada H, Mutoh H, Umehara K, Miyase T, Warashina T, Aoshima H. Matrix metalloproteinase-2 inhibitors from Clinopodium chinense var. parviflorum. J Nat Prod. 2009;72:1379–1384. doi: 10.1021/np800781t. [DOI] [PubMed] [Google Scholar]
- Mushtaq N, Schmatz R, Ahmed M, Pereira LB, da Costa P, Reichert KP, Dalenogare D, Pelinson LP, Vieira JM, Stefanello N. Protective effect of rosmarinic acid against oxidative stress biomarkers in liver and kidney of strepotozotocin-induced diabetic rats. J Physiol Biochem. 2015;71:743–751. doi: 10.1007/s13105-015-0438-4. [DOI] [PubMed] [Google Scholar]
- Nadeem M, Imran M, Aslam Gondal T, Imran A, Shahbaz M, Muhammad Amir R, Wasim Sajid M, Batool Qaisrani T, Atif M, Hussain G. Therapeutic potential of rosmarinic acid: a comprehensive review. Appl Sci. 2019;9:3139. doi: 10.3390/app9153139. [DOI] [Google Scholar]
- Nakazawa T, Ohsawa K. Metabolism of rosmarinic acid in rats. J Nat Prod. 1998;61:993–996. doi: 10.1021/np980072s. [DOI] [PubMed] [Google Scholar]
- Ngo YL, Lau CH, Chua LS. Review on rosmarinic acid extraction, fractionation and its anti-diabetic potential. Food Chem Toxicol. 2018;121:687–700. doi: 10.1016/j.fct.2018.09.064. [DOI] [PubMed] [Google Scholar]
- Nikshenas Shahrestani V, Haddadi M, Samzadeh Kermani AR. Behavioral and molecular analysis of antioxidative potential of rosmarinic acid against methamphetamine-induced augmentation of Casp3a mRNA in the zebrafish brain. Basic Clin Neurosci. 2021;12:243–254. doi: 10.32598/bcn.12.2.1777.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noguchi-Shinohara M, Ono K, Hamaguchi T, Nagai T, Kobayashi S, Komatsu J, Samuraki-Yokohama M, Iwasa K, Yokoyama K, Nakamura H, Yamada M. Safety and efficacy of Melissa officinalis extract containing rosmarinic acid in theprevention of Alzheimer's disease progression. Sci Rep. 2020;10:18627. doi: 10.1038/s41598-020-73729-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nunes S, Madureira AR, Campos D, Sarmento B, Gomes AM, Pintado M, Reis F. Therapeutic and nutraceutical potential of rosmarinic acid—cytoprotective properties and pharmacokinetic profile. Crit Rev Food Sci Nutr. 2017;57:1799–1806. doi: 10.1080/10408398.2015.1006768. [DOI] [PubMed] [Google Scholar]
- Nyandwi JB, Ko YS, Jin H, Yun SP, Park SW, Kim HJ. Rosmarinic acid inhibits oxLDL-induced inflammasome activation under high-glucose conditions through downregulating the p38-FOXO1-TXNIP pathway. Biochem Pharmacol. 2020;182:114246. doi: 10.1016/j.bcp.2020.114246. [DOI] [PubMed] [Google Scholar]
- Nyandwi JB, Ko YS, Jin H, Yun SP, Park SW, Kim HJ. Rosmarinic acid exhibits a lipid-lowering effect by modulating the expression of reverse cholesterol transporters and lipid metabolism in high-fat diet-fed mice. Biomolecules. 2021;11:1470. doi: 10.3390/biom11101470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nyandwi JB, Ko YS, Jin H, Yun SP, Park SW, Kim HJ. Rosmarinic acid increases macrophage cholesterol efflux through regulation of ABCA1 and ABCG1 in different mechanisms. Int J Mol Sci. 2021;22:8791. doi: 10.3390/ijms22168791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olivares A, Alcaraz-Saura M, Achel DG, Alcaraz M. Effect of rosmarinic acid and ionizing radiation on glutathione in melanoma B16F10 cells: a translational opportunity. Antioxidants. 2020;9:1291. doi: 10.3390/antiox9121291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ou J, Huang J, Zhao D, Du B, Wang M. Protective effect of rosmarinic acid and carnosic acid against streptozotocin-induced oxidation, glycation, inflammation and microbiota imbalance in diabetic rats. Food Funct. 2018;9:851–860. doi: 10.1039/c7fo01508a. [DOI] [PubMed] [Google Scholar]
- Özevren H, Deveci E, Tuncer MC. The effect of rosmarinic acid on deformities occurring in brain tissue by craniectomy method. Histopathological evaluation of IBA-1 and GFAP expressions. Acta Cir Bras. 2020;35:e202000406. doi: 10.1590/s0102-865020200040000006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pagano K, Carminati L, Tomaselli S, Molinari H, Taraboletti G, Ragona L. Molecular basis of the antiangiogenic action of rosmarinic acid, a natural compound targeting fibroblast growth factor-2/FGFR interactions. ChemBioChem. 2021;22:160–169. doi: 10.1002/cbic.202000610. [DOI] [PubMed] [Google Scholar]
- Pantan R, Tocharus J, Nakaew A, Suksamrarn A, Tocharus C. Ethyl rosmarinate prevents the impairment of vascular function and morphological changes in L-NAME-induced hypertensive rats. Medicina. 2019;55:777. doi: 10.3390/medicina55120777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park S-H, Kang S-H, Lim S-H, Oh H-S, Lee K-H. Design and synthesis of small chemical inhibitors containing different scaffolds for lck SH2 domain. Bioorg Med Chem Lett. 2003;13:3455–3459. doi: 10.1016/s0960-894x(03)00735-2. [DOI] [PubMed] [Google Scholar]
- Park SR, Kim SR, Hong IS, Lee HY. A novel therapeutic approach for colorectal cancer stem cells: blocking the PI3K/Akt signaling axis with caffeic acid. Front Cell Dev Biol. 2020;8:585987. doi: 10.3389/fcell.2020.585987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pattananandecha T, Apichai S, Julsrigival J, Ungsurungsie M, Samuhasaneetoo S, Chulasiri P, Kwankhao P, Pitiporn S, Ogata F, Kawasaki N, Saenjum C. Antioxidant activity and anti-photoaging effects on UVA-irradiated human fibroblasts of rosmarinic acid enriched extract prepared from Thunbergia laurifolia Leaves. Plants. 2021;10:1648. doi: 10.3390/plants10081648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paudel P, Seong SH, Zhou Y, Park CH, Yokozawa T, Jung HA, Choi JS. Rosmarinic acid derivatives’ inhibition of glycogen synthase kinase-3β is the pharmacological basis of Kangen-Karyu in Alzheimer’s disease. Molecules. 2018;23:2919. doi: 10.3390/molecules23112919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petersen M. Cytochrome P450-dependent hydroxylation in the biosynthesis of rosmarinic acid in Coleus. Phytochemistry. 1997;45:1165–1172. doi: 10.1016/S0031-9422(97)00135-0. [DOI] [Google Scholar]
- Petersen M, Simmonds MS. Rosmarinic acid. Phytochemistry. 2003;62:121–125. doi: 10.1016/s0031-9422(02)00513-7. [DOI] [PubMed] [Google Scholar]
- Petersen M, Häusler E, Karwatzki B, Meinhard J. Proposed biosynthetic pathway for rosmarinic acid in cell cultures of Coleus blumei benth. Planta. 1993;189:10–14. doi: 10.1007/BF00201337. [DOI] [Google Scholar]
- Petersen M, Abdullah Y, Benner J, Eberle D, Gehlen K, Hücherig S, Janiak V, Kim KH, Sander M, Weitzel C. Evolution of rosmarinic acid biosynthesis. Phytochemistry. 2009;70:1663–1679. doi: 10.1016/j.phytochem.2009.05.010. [DOI] [PubMed] [Google Scholar]
- Phromnoi K, Suttajit M, Saenjum C, Limtrakul DP. Inhibitory effect of a rosmarinic acid-enriched fraction prepared from Nga-Mon (Perilla frutescens) seed meal on osteoclastogenesis through the RANK signaling pathway. Antioxidants. 2021;10:307. doi: 10.3390/antiox10020307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prasannarong M, Saengsirisuwan V, Surapongchai J, Buniam J, Chukijrungroat N, Rattanavichit Y. Rosmarinic acid improves hypertension and skeletal muscle glucose transport in angiotensin II-treated rats. BMC Complement Altern Med. 2019;19:1–8. doi: 10.1186/s12906-019-2579-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qu L, Xu H, Jia W, Jiang H, Xie J. Rosmarinic acid protects against MPTP-induced toxicity and inhibits iron-induced α-synuclein aggregation. Neuropharmacology. 2019;144:291–300. doi: 10.1016/j.neuropharm.2018.09.042. [DOI] [PubMed] [Google Scholar]
- Quan W, Liu HX, Zhang W, Lou WJ, Gong YZ, Yuan C, Shao Q, Wang N, Guo C, Liu F. Cardioprotective effect of rosmarinic acid against myocardial ischaemia/reperfusion injury via suppression of the NF-κB inflammatory signalling pathway and ROS production in mice. Pharm Biol. 2021;59:222–231. doi: 10.1080/13880209.2021.1878236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radziejewska I, Supruniuk K, Nazaruk J, Karna E, Popławska B, Bielawska A, Galicka A. Rosmarinic acid influences collagen, MMPs, TIMPs, glycosylation and MUC1 in CRL-1739 gastric cancer cell line. Biomed Pharmacother. 2018;107:397–407. doi: 10.1016/j.biopha.2018.07.123. [DOI] [PubMed] [Google Scholar]
- Radziejewska I, Supruniuk K, Bielawska A. Anti-cancer effect of combined action of anti-MUC1 and rosmarinic acid in AGS gastric cancer cells. Eur J Pharmacol. 2021;902:174119. doi: 10.1016/j.ejphar.2021.174119. [DOI] [PubMed] [Google Scholar]
- Rahbardar MG, Eisvand F, Rameshrad M, Razavi BM, Hosseinzadeh H. In vivo and in vitro protective effects of rosmarinic acid against doxorubicin-induced cardiotoxicity. Nutr Cancer. 2021;4:1–15. doi: 10.1080/01635581.2021.1931362. [DOI] [PubMed] [Google Scholar]
- Rao NK, Bethala K, Sisinthy SP, Rajeswari KS. Anti-diabetic activity of Orthosiphon stamineus benth roots in streptozotocin induced type 2 diabetic rats. Asian J Pharm Clin Res. 2014;7:149–153. [Google Scholar]
- Razzaque A, Ellis B. Rosmarinic acid production in Coleus cell cultures. Planta. 1977;137:287–291. doi: 10.1007/BF00388164. [DOI] [PubMed] [Google Scholar]
- Rong H, Liang Y, Niu Y. Rosmarinic acid attenuates β-amyloid-induced oxidative stress via Akt/GSK-3β/Fyn-mediated Nrf2 activation in PC12 cells. Free Radic Biol Med. 2018;120:114–123. doi: 10.1016/j.freeradbiomed.2018.03.028. [DOI] [PubMed] [Google Scholar]
- Runtuwene J, Cheng K-C, Asakawa A, Amitani H, Amitani M, Morinaga A, Takimoto Y, Kairupan BHR, Inui A. Rosmarinic acid ameliorates hyperglycemia and insulin sensitivity in diabetic rats, potentially by modulating the expression of PEPCK and GLUT4. Drug Des Dev Ther. 2016;10:2193. doi: 10.2147/DDDT.S108539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadeghi A, Bastin AR, Ghahremani H, Doustimotlagh AH. The effects of rosmarinic acid on oxidative stress parameters and inflammatory cytokines in lipopolysaccharide-induced peripheral blood mononuclear cells. Mol Biol Rep. 2020;47:3557–3566. doi: 10.1007/s11033-020-05447-x. [DOI] [PubMed] [Google Scholar]
- Samsu N, Soeharto S, Rifai M, Rudijanto A. Rosmarinic acid monotherapy is better than the combination of rosmarinic acid and telmisartan in preventing podocyte detachment and inhibiting the progression of diabetic nephropathy in rats. Biologics. 2019;13:179. doi: 10.2147/BTT.S214820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satoh T, Trudler D, Oh CK, Lipton SA. Potential therapeutic use of the rosemary diterpene carnosic acid for Alzheimer's disease, Parkinson's disease, and long-COVID through NRF2 Activation to counteract the NLRP3 inflammasome. Antioxidants. 2022;11:124. doi: 10.3390/antiox11010124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scholey A, Gibbs A, Neale C, Perry N, Ossoukhova A, Bilog V, Kras M, Scholz C, Sass M, Buchwald-Werner S. Anti-stress effects of lemon balm-containing foods. Nutrients. 2014;6:4805–4821. doi: 10.3390/nu6114805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Şengelen A, Önay-Uçar E. Rosmarinic acid and siRNA combined therapy represses Hsp27 (HSPB1) expression and induces apoptosis in human glioma cells. Cell Stress Chaperones. 2018;23:885–896. doi: 10.1007/s12192-018-0896-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sevgi K, Tepe B, Sarikurkcu C. Antioxidant and DNA damage protection potentials of selected phenolic acids. Food Chem Toxicol. 2015;77:12–21. doi: 10.1016/j.fct.2014.12.006. [DOI] [PubMed] [Google Scholar]
- Shakeri F, Eftekhar N, Roshan NM, Rezaee R, Moghimi A, Boskabady M. Rosmarinic acid affects immunological and inflammatory mediator levels and restores lung pathological features in asthmatic rats. Allergol Immunopathol. 2019;47:16–23. doi: 10.1016/j.aller.2018.04.004. [DOI] [PubMed] [Google Scholar]
- Shamsi A, Ahmed A, Khan MS, Husain FM, Bano B. Rosmarinic acid restrains protein glycation and aggregation in human serum albumin: multi spectroscopic and microscopic insight—possible therapeutics targeting diseases. Int J Biol Macromol. 2020;161:187–193. doi: 10.1016/j.ijbiomac.2020.06.048. [DOI] [PubMed] [Google Scholar]
- Shimojo Y, Kosaka K, Noda Y, Shimizu T, Shirasawa T. Effect of rosmarinic acid in motor dysfunction and life span in a mouse model of familial amyotrophic lateral sclerosis. J Neurosci Res. 2010;88:896–904. doi: 10.1002/jnr.22242. [DOI] [PubMed] [Google Scholar]
- Silva MLAE, Lucarini R, Dos Santos FF, Martins CHG, Pauletti PM, Januario AH, Santos MFC, Cunha WR. Hypoglycemic effect of rosmarinic acid-rich infusion (RosCE) from Origanum vulgare in alloxan-induced diabetic rats. Nat Prod Res. 2021;14:1–7. doi: 10.1080/14786419.2021.1990282. [DOI] [PubMed] [Google Scholar]
- Son S, Lee SG, Jeong DH, Yoo CJ. Malignant solitary fibrous tumor of tandem lesions in the skull and spine. J Korean Neurosurg Soc. 2013;54:246–249. doi: 10.3340/jkns.2013.54.3.246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sotnikova R, Okruhlicova L, Vlkovicova J, Navarova J, Gajdacova B, Pivackova L, Fialova S, Krenek P. Rosmarinic acid administration attenuates diabetes-induced vascular dysfunction of the rat aorta. J Pharm Pharmacol. 2013;65:713–723. doi: 10.1111/jphp.12037. [DOI] [PubMed] [Google Scholar]
- Sotnikova R, Kaprinay B, Navarova J. Rosmarinic acid mitigates signs of systemic oxidative stress in streptozotocin-induced diabetes in rats. Gen Physiol Biophys. 2015;34:449–452. doi: 10.4149/gpb_2015025. [DOI] [PubMed] [Google Scholar]
- Sperl B, Seifert MH, Berg T. Natural product inhibitors of protein–protein interactions mediated by Src-family SH2 domains. Bioorg Med Chem Lett. 2009;19:3305–3309. doi: 10.1016/j.bmcl.2009.04.083. [DOI] [PubMed] [Google Scholar]
- Sutkowska J, Hupert N, Gawron K, Strawa JW, Tomczyk M, Forlino A, Galicka A. The stimulating effect of rosmarinic acid and extracts from rosemary and lemon balm on collagen type I biosynthesis in osteogenesis imperfecta type I skin fibroblasts. Pharmaceutics. 2021;13:938. doi: 10.3390/pharmaceutics13070938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swarup V, Ghosh J, Ghosh S, Saxena A, Basu A. Antiviral and anti-inflammatory effects of rosmarinic acid in an experimental murine model of Japanese encephalitis. Antimicrob Agents Chemother. 2007;51:3367–3370. doi: 10.1128/AAC.00041-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takano H, Osakabe N, Sanbongi C, Yanagisawa R, Inoue K-I, Yasuda A, Natsume M, Baba S, Ichiishi E-I, Yoshikawa T. Extract of Perilla frutescens enriched for rosmarinic acid, a polyphenolic phytochemical, inhibits seasonal allergic rhinoconjunctivitis in humans. Exp Biol Med. 2004;229:247–254. doi: 10.1177/153537020422900305. [DOI] [PubMed] [Google Scholar]
- Thingore C, Kshirsagar V, Juvekar A. Amelioration of oxidative stress and neuroinflammation in lipopolysaccharide-induced memory impairment using Rosmarinic acid in mice. Metab Brain Dis. 2021;36:299–313. doi: 10.1007/s11011-020-00629-9. [DOI] [PubMed] [Google Scholar]
- Touiss I, Ouahhoud S, Harnafi M, Khatib S, Bekkouch O, Amrani S, Harnafi H. Toxicological evaluation and hepatoprotective efficacy of rosmarinic acid-rich extract from Ocimum basilicum L. Evid Based Complement Altern Med. 2021;2021:6676998. doi: 10.1155/2021/6676998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trócsányi E, György Z, Zámboriné-Németh É. New insights into rosmarinic acid biosynthesis based on molecular studies. Curr Plant Biol. 2020;23:100162. doi: 10.1016/j.cpb.2020.100162. [DOI] [Google Scholar]
- Tsai C-F, Wu J-Y, Hsu Y-W. Protective effects of rosmarinic acid against selenite-induced cataract and oxidative damage in rats. Int J Med Sci. 2019;16:729. doi: 10.7150/ijms.32222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urushima H, Nishimura J, Mizushima T, Hayashi N, Maeda K, Ito T. Perilla frutescens extract ameliorates DSS-induced colitis by suppressing pro-inflammatory cytokines and inducing anti-inflammatory cytokines. Am J Physiol Gastrointest Liver Physiol. 2015;308:G32–G41. doi: 10.1152/ajpgi.00294.2014. [DOI] [PubMed] [Google Scholar]
- Vasileva LV, Savova MS, Tews D, Wabitsch M, Georgiev MI. Rosmarinic acid attenuates obesity and obesity-related inflammation in human adipocytes. Food Chem Toxicol. 2021;149:112002. doi: 10.1016/j.fct.2021.112002. [DOI] [PubMed] [Google Scholar]
- Vlavcheski F, Naimi M, Murphy B, Hudlicky T, Tsiani E. Rosmarinic acid, a rosemary extract polyphenol, increases skeletal muscle cell glucose uptake and activates AMPK. Molecules. 2017;22:1669. doi: 10.3390/molecules22101669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vuorinen A, Engeli RT, Leugger S, Bachmann F, Akram M, Atanasov AG, Waltenberger B, Temml V, Stuppner H, Krenn L. Potential antiosteoporotic natural product lead compounds that inhibit 17β-hydroxysteroid dehydrogenase type 2. J Nat Prod. 2017;80:965–974. doi: 10.1021/acs.jnatprod.6b00950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waer CN, Kaur P, Tumur Z, Hui DD, Le B, Guerra C, Henson B, Seleem D, Lewis J. Rosmarinic acid/blue light combination treatment inhibits head and neck squamous cell carcinoma in vitro. Anticancer Res. 2020;40:751–758. doi: 10.21873/anticanres.14006. [DOI] [PubMed] [Google Scholar]
- Wang J, Xu H, Jiang H, Du X, Sun P, Xie J. Neurorescue effect of rosmarinic acid on 6-hydroxydopamine-lesioned nigral dopamine neurons in rat model of Parkinson's disease. J Mol Neurosci. 2012;47:113–119. doi: 10.1007/s12031-011-9693-1. [DOI] [PubMed] [Google Scholar]
- Wang L, Yang H, Wang C, Shi X, Li K. Rosmarinic acid inhibits proliferation and invasion of hepatocellular carcinoma cells SMMC 7721 via PI3K/AKT/mTOR signal pathway. Biomed Pharmacother. 2019;120:109443. doi: 10.1016/j.biopha.2019.109443. [DOI] [PubMed] [Google Scholar]
- Wang Y, Meng J, Men L, An B, Jin X, He W, Lu S, Li N. Rosmarinic acid protects mice from concanavalin A-induced hepatic injury through AMPK signaling. Biol Pharm Bull. 2020;43:1749–1759. doi: 10.1248/bpb.b20-00477. [DOI] [PubMed] [Google Scholar]
- Wang CC, Hsieh PW, Kuo JR, Wang SJ. Rosmarinic acid, a bioactive phenolic compound, inhibits glutamate release from rat cerebrocortical synaptosomes through GABAA receptor activation. Biomolecules. 2021;11:1029. doi: 10.3390/biom11071029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang GY, Chen SY, Chen YY, Hong CJ, Hsu YH, Yen GC. Protective effect of rosmarinic acid-rich trichodesma khasianum clarke leaves against ethanol-induced gastric mucosal injury in vitro and in vivo. Phytomedicine. 2021;80:153382. doi: 10.1016/j.phymed.2020.153382. [DOI] [PubMed] [Google Scholar]
- Wang J, Wang S, Guo H, Li Y, Jiang Z, Gu T, Su B, Hou W, Zhong H, Cheng D. Rosmarinic acid protects rats against post-stroke depression after transient focal cerebral ischemic injury through enhancing antioxidant response. Brain Res. 2021;1757:147336. doi: 10.1016/j.brainres.2021.147336. [DOI] [PubMed] [Google Scholar]
- Wei Y, Chen J, Cai GE, Lu W, Xu W, Wang R, Lin Y, Yang C. Rosmarinic acid regulates microglial M1/M2 polarization via the PDPK1/Akt/HIF pathway under conditions of neuroinflammation. Inflammation. 2021;44:129–147. doi: 10.1007/s10753-020-01314-w. [DOI] [PubMed] [Google Scholar]
- Wen Y-J, Yin M-C. The anti-inflammatory and anti-glycative effects of rosmarinic acid in the livers of type 1 diabetic mice. Biomedicine. 2017;7:19. doi: 10.1051/bmdcn/2017070319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu L, Velander P, Brown AM, Wang Y, Liu D, Bevan DR, Zhang S, Xu B. Rosmarinic acid potently detoxifies amylin amyloid and ameliorates diabetic pathology in a transgenic rat model of type 2 diabetes. ACS Pharmacol Transl Sci. 2021;4:1322–1337. doi: 10.1021/acsptsci.1c00028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto S, Kayama T, Noguchi-Shinohara M, Hamaguchi T, Yamada M, Abe K, Kobayashi S. Rosmarinic acid suppresses tau phosphorylation and cognitivedecline by downregulating the JNK signaling pathway. NPJ Sci Food. 2021;5:1. doi: 10.1038/s41538-021-00084-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang K, Shen Z, Zou Y, Gao K. Rosmarinic acid inhibits migration, invasion, and p38/AP-1 signaling via miR-1225-5p in colorectal cancer cells. J Recept Signal Transduct Res. 2021;41:284–293. doi: 10.1080/10799893.2020.1808674. [DOI] [PubMed] [Google Scholar]
- Yao Y, Xu Y, Wang Y. Protective roles and mechanisms of rosmarinic acid in cyclophosphamide-induced premature ovarian failure. J Biochem Mol Toxicol. 2020;34(12):e22591. doi: 10.1002/jbt.22591. [DOI] [PubMed] [Google Scholar]
- Yi ZJ, Gong JP, Zhang W. Transcriptional co-regulator RIP140: an important mediator of the inflammatory response and its associated diseases. Mol Med Rep. 2017;16:994–1000. doi: 10.3892/mmr.2017.6683. [DOI] [PubMed] [Google Scholar]
- Youn J, Lee K-H, Won J, Huh S-J, Yun H-S, Cho W-G, Paik D-J. Beneficial effects of rosmarinic acid on suppression of collagen induced arthritis. J Rheumatol. 2003;30:1203–1207. [PubMed] [Google Scholar]
- Yu Y, Wu Y, Yan HZ, Xia ZR, Wen W, Liu DY, Wan LH. Rosmarinic acid ameliorates acetaminophen-induced acute liver injury in mice via RACK1/TNF-α mediated antioxidant effect. Pharm Biol. 2021;59:1286–1293. doi: 10.1080/13880209.2021.1974059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan H, Lu W, Wang L, Shan L, Li H, Huang J, Sun Q, Zhang W. Synthesis of derivatives of methyl rosmarinate and their inhibitory activities against matrix metalloproteinase-1 (MMP-1) Eur J Med Chem. 2013;62:148–157. doi: 10.1016/j.ejmech.2012.09.047. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Chen X, Yang L, Zu Y, Lu Q. Effects of rosmarinic acid on liver and kidney antioxidant enzymes, lipid peroxidation and tissue ultrastructure in aging mice. Food Funct. 2015;6:927–931. doi: 10.1039/c4fo01051e. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Hu M, Liu L, Cheng X-L, Cai J, Zhou J, Wang T. Anticancer effects of rosmarinic acid in OVCAR-3 ovarian cancer cells are mediated via induction of apoptosis, suppression of cell migration and modulation of lncRNA MALAT-1 expression. J BUON. 2018;23:763–768. [PubMed] [Google Scholar]
- Zhang Q, Li J, Peng S, Zhang Y, Qiao Y. Rosmarinic acid as a candidate in a phenotypic profiling cardio-/cytotoxicity cell model induced by doxorubicin. Molecules. 2020;25:836. doi: 10.3390/molecules25040836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang T, Ma S, Liu C, Hu K, Xu M, Wang R. Rosmarinic acid prevents radiation-induced pulmonary fibrosis through attenuation of ROS/MYPT1/TGFβ1 signaling via miR-19b-3p. Dose Response. 2020;18:1559325820968413. doi: 10.1177/1559325820968413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J, Cui X, Zhang M, Bai B, Yang Y, Fan S. The antibacterial mechanism of perilla rosmarinic acid. Biotechnol Appl Biochem. 2021 doi: 10.1002/bab.2248. [DOI] [PubMed] [Google Scholar]
- Zhang W, Cheng C, Sha Z, Chen C, Yu C, Lv N, Ji P, Wu X, Ma T, Cheng H, Shi L. Rosmarinic acid prevents refractory bacterial pneumonia through regulating Keap1/Nrf2-mediated autophagic pathway and mitochondrial oxidative stress. Free Radic Biol Med. 2021;168:247–257. doi: 10.1016/j.freeradbiomed.2021.03.038. [DOI] [PubMed] [Google Scholar]
- Zhao L, Zhang Y, Liu G, Hao S, Wang C, Wang Y. Black rice anthocyanin-rich extract and rosmarinic acid, alone and in combination, protect against DSS-induced colitis in mice. Food Funct. 2018;9:2796–2808. doi: 10.1039/C7FO01490B. [DOI] [PubMed] [Google Scholar]
- Zhao Y, Han Y, Wang Z, Chen T, Qian H, He J, Li J, Han B, Wang T. Rosmarinic acid protects against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine- induced dopaminergic neurotoxicity in zebrafish embryos. Toxicol in Vitro. 2020;65:104823. doi: 10.1016/j.tiv.2020.104823. [DOI] [PubMed] [Google Scholar]
- Zhou X, Wang W, Li Z, Chen L, Wen C, Ruan Q, Xu Z, Liu R, Xu J, Bai Y, Deng J. Rosmarinic acid decreases the malignancy of pancreatic cancer through inhibiting Gli1 signaling. Phytomedicine. 2022;95:153861. doi: 10.1016/j.phymed.2021.153861. [DOI] [PubMed] [Google Scholar]
- Zych M, Wojnar W, Borymski S, Szałabska K, Bramora P, Kaczmarczyk-Sedlak I. Effect of rosmarinic acid and sinapic acid on oxidative stress parameters in the cardiac tissue and serum of type 2 diabetic female rats. Antioxidants. 2019;8:579. doi: 10.3390/antiox8120579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zych M, Wojnar W, Dudek S, Kaczmarczyk-Sedlak I. Rosmarinic and sinapic acids may increase the content of reduced glutathione in the lenses of estrogen-deficient rats. Nutrients. 2019;11:803. doi: 10.3390/nu11040803. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.