Abstract
Background
The increased warfarin sensitivity observed after mechanical mitral valve replacement (MVR) operations dictates clinical discretion in warfarin dose initiation. Evidence is still lacking with regard to anticoagulation management of MVR patients.
Objective
This study aimed to compare initiating warfarin at the recommended dosing regimen versus empirically lowered doses intended to account for the variation in warfarin sensitivity.
Methods
A prospective, single-blind, randomized, comparative study was conducted in postoperative MVR patients. Patients were randomly assigned to either the 5 mg group (n = 25) or the 3 mg group (n = 25) and were initiated on a 5 or 3 mg warfarin dose, respectively. Time to target international normalized ratio (INR), time in therapeutic range, occurrence of bleeding/thromboembolic events, and cost of bridging with enoxaparin were assessed for both groups.
Results
Target INR was achieved earlier in the 5 mg group than in the 3 mg group (p = 0.033), with a mean ± SD of 5.3 ± 2.0 and 6.6 ± 2.0, respectively (95% confidence interval of the mean difference 1.022–1.890). Bleeding events did not differ significantly between the two groups. The cost of enoxaparin consumption per patient was significantly higher in the 3 mg group versus the 5 mg group (p = 0.002).
Conclusions
The initiation of warfarin at a 5 mg dose in MVR patients was more efficacious than the 3 mg dose in terms of time to reach the target INR. Moreover, the cost of enoxaparin bridging was significantly reduced with a 5 mg warfarin initiation dose. Bleeding events were comparable.
ClinicalTrials.gov ID
NCT04235569, 22 January 2020.
Key Points
| Optimal warfarin initiation dosing in patients with mechanical mitral valve replacements (MVRs) is debatable. |
| Warfarin initiation dose is lowered empirically in clinical institutions to balance the early increased postoperative warfarin sensitivity, leading to increased consumption of enoxaparin as a bridging agent. |
| This study compared two initiation dosing protocols of warfarin 5 mg versus 3 mg using low molecular weight heparin (enoxaparin) as a bridging agent in the MVR patient population at a single institution. |
| Patients receiving the 5 mg initiation dose achieved therapeutic international normalized ratio more rapidly, hence minimizing the cost of enoxaparin bridging. |
Introduction
It has been estimated that 300,000 prosthetic heart valve replacements are performed each year worldwide [1]. Mechanical heart valves are more durable than bio-prostheses but more thrombogenic. Hence, postoperative warfarin anticoagulation is recommended for all patients receiving mechanical aortic or mitral heart valve replacement [2]. Mechanical heart valves necessitate lifetime anticoagulation, with an international normalized ratio (INR) target range of 2.0–3.0 for aortic valves, and 2.5–3.5 for mitral valves. Warfarin starting doses between 5 and 10 mg/day administered on the first 1 or 2 days of warfarin treatment were consistently accepted in earlier medical practice and were recommended by the American College of Chest Physicians (ACCP) to be applied for individuals who need long-term anticoagulation with consequent dose adjustment based on the INR findings [3]. The exaggerated warfarin sensitivity is defined as an increased warfarin dose response that people may experience as a result of certain conditions or factors [4], and it is well recognized after heart valve surgery [5–7].
Many factors contribute to this postoperative sensitivity, including patient’s age, sex, body weight, concomitant medications such as amiodarone and cefazoline, baseline INR, and serum albumin level [8]. Owing to the lack of evidence in this field, currently available guidelines offer no clear recommendation concerning warfarin initiation after heart valve surgery [9–11].
The existing warfarin commencement protocols were addressed for use in populations other than mitral valve replacement (MVR) patients [12–16]. Those protocols investigated different warfarin initiation dosing techniques (5 mg, 10 mg, empirical, or age-adjusted) in patient populations with various warfarin indications such as atrial fibrillation, deep vein thrombosis (DVT), pulmonary embolus, malignancy, or vascular surgeries. Although there is still considerable uncertainty regarding using a 10 mg or 5 mg loading dose for warfarin initiation [17], using the 10 mg dose is not recommended and is not usually used in clinical practice for postoperative warfarin initiation after prosthetic heart valve replacement due to the increased warfarin sensitivity and risk of bleeding [18, 19]. As a result of these recommendations, the starting dose of 10 mg is no longer used in our hospital, and lower initiation doses, such as 5 mg, may be preferred following prosthetic valve replacement surgery.
In clinical practice, it has been observed that when starting with the commonly used warfarin loading dose of 5 mg, many patients exceed the upper limit of the therapeutic range and thus require several dose reductions; occasionally, warfarin may be discontinued, resulting in irregular anticoagulation management [20]. This may result in a higher risk of bleeding, a delay in achieving therapeutic INR, the removal of the pacing wires, and, consequently, prolonged hospitalization [21]. Hence, many clinical practices recently favored loading warfarin using reduced empirical dosing (e.g., 3 mg), particularly in the early postoperative period [16, 22, 23]. This practice has hindered the use of the warfarin initiation protocols, with a 5 mg dose in our hospital after heart valve surgery. Consequently, warfarin initiation in this group has sometimes reverted to empirical dosing depending on clinical judgment, with the commencement of a 3 mg dose targeting a more conservative initiation and transition to a stable INR.
The current study aimed to compare the efficacy and safety of warfarin initiation at two dose levels (5 vs. 3 mg), in terms of reaching optimum anticoagulation and occurrence of bleeding/thromboembolic events, as well as to calculate the enoxaparin cost used as the bridging agent.
Methods
Study Design
This was a prospective, single-blinded, randomized controlled study conducted in patients subjected to isolated mechanical MVR at the Cardiovascular Hospital, Ain Shams University (ASU). The study protocol was approved by the Research Ethics Committee for Experimental and Clinical Studies at the Faculty of Pharmacy, ASU. Before participation in the study, written informed consent was obtained from all participants. The study was conducted in accordance with the Declaration of Helsinki and was registered at ClinicalTrials.gov (ID: NCT04235569).
Patients and Methods
All patients presenting to the Cardiovascular Hospital for MVR were screened for eligibility. Patients were included if they were between 18 and 65 years of age. Exclusion criteria comprised pregnancy or lactation, renal dysfunction (glomerular filtration rate ≤ 45 mL/min), hepatic insufficiency (Child–Pugh class B or C), clinically significant active bleeding, recurrent DVT or pulmonary embolism, baseline INR > 1.2, cancer, Asian ancestry, malnutrition, or excessive alcohol consumption.
Patients were randomly assigned to either the 5 mg group (n = 25) or the 3 mg group (n = 25) and initiated on warfarin 5 or 3 mg, respectively. A blocked randomization list for patient allocation was produced using an online software application available at https://www.sealedenvelope.com/simple-randomiser. An allocation ratio of 1:1 and a block size of 2 was determined. Participant randomization assignment remained concealed in sealed envelopes.
The principal investigator was the on-site clinical pharmacist responsible for generating the allocation sequence, participants’ enrollment, and randomly assigning participants to interventions, as well as the collection of patients’ data during the study period.
Patients’ demographic data [age, sex, body mass index (BMI), smoking status] and laboratory data (liver and kidney function tests, platelet count, serum albumin), along with comorbid diseases, allergies, comedication, and family history, were all collected from the patient daily notes using a predesigned follow-up sheet.
The study patients were initiated on warfarin for postoperative anticoagulation combined with enoxaparin as a bridging agent 24 h after surgery. Once the lower limit of the therapeutic INR range (2.5) was reached, enoxaparin was discontinued. Enoxaparin injections were administered twice daily using therapeutic doses adapted to the patient’s body weight (1 mg/kg).
Warfarin dose adjustments and enoxaparin injection doses were determined using the anticoagulation toolkit (version 1.7) [24] produced by the Michigan Anticoagulation Quality Improvement Initiative (MAQI2) [18].
There is no validated initiation nomogram for warfarin doses < 5 mg or for the target INR range (2.5–3.5). Hence, some healthcare facilities (such as the University of Connecticut Health Center/John Dempsey Hospital, 2015 [25] and University of Iowa Healthcare Warfarin Tips & Dosing Nomograms, 2013 [26]) previously applied dose adjustment techniques that used the same percentage increase or decrease in warfarin initiation dose nomogram stated in MAQI2 to fill the gap between the published guidelines and clinical practice. We decided to adopt the same MAQI2 accustomed technique used by other health care institutions [25, 26] to standardize the dose adjustment processes and make the results reproducible.
Regarding postoperative care, all patients were monitored in the Intensive Care Unit (ICU) of the Cardiovascular Hospital, ASU, and received postoperative care by the ICU staff as per the hospital management protocols for postoperative oral diet introduction, fluid administration, and extubation.
Follow-Up and Endpoints
All patients were followed up for a minimum of 4 consecutive days from the start of postoperative anticoagulation until the INR was within the therapeutic range for at least 2 consecutive days [22, 27]. According to the routine practice of our institution, follow-up was scheduled daily for INR monitoring and dose adjustments. Warfarin dose was administered at 5:00 pm, while INR laboratory monitoring was done on the morning of the following day. The INR measurement methodology and techniques were unified and assessed at the same laboratory for all patients in both groups. Dose adjustments were made accordingly by the attending physician together with the responsible clinical pharmacist until the first in-range target INR reading (2.5–3.5). All patients’ comedications during the follow-up period were recorded.
Primary Outcome
The quality of anticoagulation was evaluated using time to reach therapeutic INR, defined as the number of days required to reach the lower limit (2.5) of the target INR range.
Secondary Outcomes
The proportion of patients who reached the target INR range within 3–5 days was recorded. The 3- to 5-day range was the time required for the usual onset of the anticoagulant effect of warfarin that matches the target time crucial for cardiac surgeons to allow the removal of pacing wires.
The proportion of time spent in the therapeutic range, defined as the percentage of time (days) that the patient’s INR value was within the therapeutic INR range, was also reported. It was calculated as a percentage ratio of the number of in-range INR readings to the total number of INR readings recorded throughout the follow-up period.
The count of INR readings per patient that were in range or that exceeded four during the follow-up period were also recorded. Furthermore, any major or minor bleeding events, as well as thromboembolic events, were reported during the follow-up period.
Minor bleedings were mild bleeding events that required minimal medical intervention only to stop or treat the bleeding without the need to temporarily discontinue warfarin [28]. Major bleedings were serious bleeding events that necessitated extensive medical intervention to stop or treat bleeding, including temporarily discontinuing or changing the dose of warfarin [28]. They were defined using the National Cancer Institute’s Common Terminology Criteria for Adverse Events (CTCAE v4.03 [minor update 2014]) [29].
Cost of Bridging
During the anticoagulation initiation period, the duration of bridging with enoxaparin administration until its discontinuation was reported.
The cost consumed, from the time enoxaparin was initiated until it was discontinued after reaching the first therapeutic INR, was calculated and was estimated in US dollars.
Statistical Analysis
Sample size was calculated using the STATA program (StataCorp LLC, College Station, TX, USA), setting the type-1 error (α) at 0.05 and the power (1 − β) at 0.9. Results from a previous study by Roberts et al. [22] showed that the protocol group (n = 37) achieved a stable INR more rapidly than the empiric group (n = 77); median times of 5.1 and 8.7 days, respectively, with a pooled standard deviation (SD) of 3.4 days. Median was transformed to mean according to Hozo et al. formula [30]. Calculation according to these values produced a sample size of 25 cases per group (50 total), considering a 20% dropout rate.
Statistical analysis was performed using IBM SPSS® Statistics version 22 (IBM® Corporation, Armonk, NY, USA). Quantitative data were expressed as mean and SD or median and interquartile range as appropriate, while qualitative data were expressed as frequency and percentage. Pearson’s Chi-square test or Fisher’s exact test was used to examine the relation between qualitative variables. Quantitative data were tested for normality using the Kolmogorov–Smirnov test and Shapiro–Wilk test. Comparison between two groups for normally distributed data was performed using Student’s t test, and the Mann–Whitney test was used for data that were not normally distributed. Because there was a significant difference in age between the two groups, the outcome measures were adjusted for age using logistic regression for qualitative outcome variables or analysis of covariance (ANCOVA) for quantitative outcome variables. The other patient characteristics that could have affected warfarin sensitivity, such as BMI, albumin level, and some of the concomitantly used drugs, were comparable between the two study groups and did not require statistical adjustment. All tests were two-tailed. A p value < 0.05 was considered significant.
Results
From March 2018 to January 2019, out of a total of 85 MVR patients screened, only 50 patients fulfilled the inclusion criteria and were recruited, as detailed in Fig. 1. The analysis included all randomized patients.
Fig. 1.
Study flow diagram: enrollment, randomization, and follow-up
Patient Characteristics
Baseline characteristics and demographics were comparable between the two groups except for age, which was statistically higher in the 3 mg group versus the 5 mg group (p = 0.048). No allergic reactions were observed in either group of patients.
Serum creatinine level was within the normal range in both groups, but the 5 mg group had a significantly higher level than the 3 mg group (p = 0.001). The identified comorbidities included diabetes mellitus, hypertension, rheumatic heart disease, atrial fibrillation, hyperlipidemia, and hypothyroidism. Medications coadministered included cephazolin, amiodarone, non-steroidal anti-inflammatory drugs, paracetamol, omeprazole, levothyroxine, simvastatin, and propranolol. Among those medications, amiodarone and cefazoline are known to affect warfarin response [4]. Medication use was comparable between groups, particularly for amiodarone (28% vs. 16%) and cefazoline (92% vs. 92%). Patients’ demographics, baseline characteristics, and comedications affecting warfarin response are presented in Table 1.
Table 1.
Demographics and baseline characteristics of the study groups
| Parameter | 5 mg group [n = 25] | 3 mg group [n = 25] | p value |
|---|---|---|---|
| Age, years [mean ± SD] | 43.2 ± 12.7 | 49.3 ± 8.0 | 0.048a,e |
| Male [n (%)] | 13 (52) | 11 (44) | 0.571b |
| BMI, kg/m2 [mean ± SD] | 30.6 ± 5.9 | 27.8 ± 3.7 | 0.061a |
| Smoking status [n (%)] | 6 (24) | 5 (20) | 0.733b |
| INR [mean ± SD] | 1.1 ± 0.1 | 1.1 ± 0.1 | 0.279a |
| Serum creatinine, mg/dL [median (IQR)] | 1.10 (0.90–1.16) | 0.83 (0.79–0.94) | 0.001c,e |
| Platelet count, ×1000/μL [mean ± SD] | 381 ± 66 | 390 ± 59 | 0.613a |
| ALT, IU/L [mean ± SD] | 23.7 ± 5.1 | 19.0 ± 5.0 | 0.052a |
| AST, IU/L [mean ± SD] | 21.8 ± 5.4 | 20.1 ± 3.9 | 0.090a |
| Serum albumin, g/dL [mean ± SD] | 4.1 ± 0.4 | 4.2 ± 0.5 | 0.608a |
| Number of patient comorbidities [median (IQR)] | 1 (0–1) | 1 (0–1) | 0.142c |
| Comorbid diseases [n (%)] | |||
| Diabetes mellitus | 7 (28) | 7 (28) | 1.00b |
| Hypertension | 2 (8) | 5 (20) | 0.417 |
| Rheumatic heart disease | 4 (16) | 4 (16) | 1.00b |
| Atrial fibrillation | 2 (8) | 2 (8) | d |
| Hyperlipidemia | 0 (0) | 2 (8) | d |
| Hypothyroidism | 1 (4) | 2 (8) | d |
| Medications [n (%)] | |||
| Amiodarone | 7 (28) | 4 (16) | 0.306b |
| Cefazolin | 23 (92) | 23 (92) | 1.00b |
ALT alanine aminotransferase, AST aspartate aminotransferase, BMI body mass index, INR international normalized ratio, IQR interquartile range, SD standard deviation
aIndependent t test
bChi-square test
cMann–Whitney test
dNo p value given because of the small number of cases within groups
ep value < 0.05: significant
Primary Outcome Analysis
At the end of the follow-up period, comparison between groups indicated that the 5 mg group achieved a therapeutic INR more rapidly than the 3 mg group (95% confidence interval [CI] of the mean difference 1.02–1.89), with a mean ± SD of 5.3 ± 2.0 and 6.6 ± 2.0 days for the 5 mg and 3 mg groups, respectively (p = 0.033) (Table 2).
Table 2.
Comparison of outcomes and endpoints between the study groups
| Parameter | 5 mg group [n = 25] | 3 mg group [n = 25] | p value |
|---|---|---|---|
| TRT, days [mean ± SD] | 5.3 ± 2.0 | 6.6 ± 2.0 | 0.033a,c |
| Patients reached target INR in the first 3–5 days [n (%)] | 14 (56) | 7 (28) | 0.080b |
| Proportion of time spent in therapeutic INR, % [median (IQR)] | 38 (25–44) | 29 (24–33) | 0.091a |
| Number of INR readings > 4 [median (IQR)] | 0.0 (0.0–2.0) | 0.0 (0.0–0.5) | 0.009a,c |
| Number of in-range readings [median (IQR)] | 3 (2–4) | 2 (2–3) | 0.023a,c |
| Patients experienced major bleeding event [n (%)] | 4 (16) | 5 (20) | 0.589b |
| Patients experienced minor bleeding event [n (%)] | 11 (44) | 5 (20) | 0.022b,c |
| Patients experienced any bleeding event [n (%)] | 13 (52) | 8 (32) | 0.094b |
| Total number of bleeding events [median (IQR)] | 1 (0–2) | 0 (0–1) | 0.052a |
| Number of bridging days [median (IQR)] | 3.0 (2.0–4.0) | 6.0 (3.5–6.5) | < 0.001a,c |
| Enoxaparin overall cost, USD [median (IQR)] | 24.8 (16.5–35.6) | 49.6 (30.8–57.5) | 0.002a,c |
| Follow-up period, days [mean ± SD] | 9.0 ± 2.8 | 8.6 ± 2.5 | 0.557a |
TRT time required to reach the lower limit of therapeutic INR, INR international normalized ratio, IQR interquartile range, SD standard deviation
aAnalysis of covariance
bLogistic regression
cp value < 0.05: significant
Endpoints are presented in Table 2. A dot plot to show the 25 individual time to reach therapeutic INR data points for each group is presented in Fig. 2.
Fig. 2.
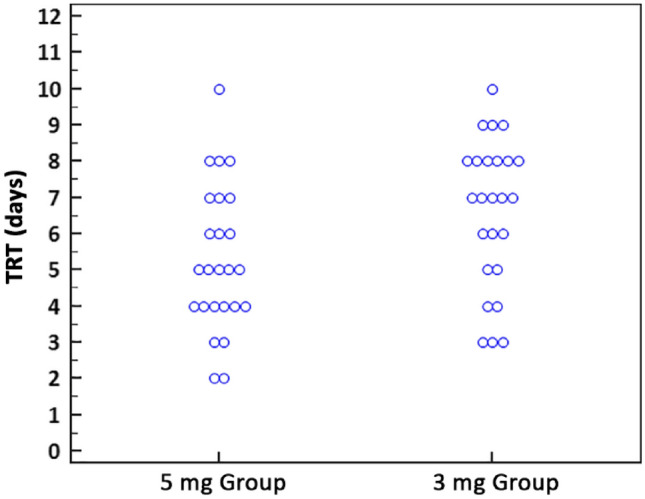
Dot plot showing individual TRT for the 25 patients in each group. TRT time to reach therapeutic international normalized ratio
Secondary Outcome Analysis
The proportion of patients who reached the target INR range within 3–5 days was not significantly different between the two study groups (5 mg group vs. 3 mg group: 14 [56%] vs. 7 [28%]; p = 0.08). Furthermore, the proportion of time spent in the therapeutic range was comparable between the two groups. During the follow-up period, the median number of in-range readings, as well as the number of INR readings that were > 4 per patient, were statistically higher in the 5 mg group.
Four patients experienced major bleeding events in the 5 mg group compared with five patients in the 3 mg group (p = 0.589). Eleven patients experienced minor bleeding events in the 5 mg group compared with five patients in the 3 mg group (p = 0.022). The percentages of patients who experienced any type of bleeding events, as well as the total number of bleeding events experienced for each patient in each group, were not significantly different between the groups.
The major bleeding events reported were hematuria, hematochezia, hematemesis, and wound bleed, while the minor bleeding events were epistaxis, bleeding gum, and localized bruises.
No thromboembolic events were reported in patients of either group throughout the follow-up period, and there was no significant difference in the length of follow-up periods between the two groups (p = 0.120).
Cost Analysis
The median duration of enoxaparin administration as bridging therapy was significantly higher in the 3 mg group (p < 0.001) (Table 2).
The cost of enoxaparin consumption per patient was significantly lower in the 5 mg group compared with the 3 mg group, with a median USD (IQR) of 24.8 (16.5–35.6) versus 49.6 (30.8–57.5), respectively (p = 0.002) (Table 2).
Discussion
After mechanical MVR, anticoagulation is usually initiated with warfarin in addition to low molecular weight heparin (LMWH) for bridging until the therapeutic INR is achieved [31]. Warfarin initiation at a 5 mg dose may sometimes result in irregular anticoagulation management [20] and a delay in pacing wire removal [21]. As a result, many clinical practices have recently favored loading warfarin with lower empirical dosing, such as 3 mg. In light of the complete lack of clinical trials specifically addressing this issue, this was considered a safe strategy [16, 22, 23].
Currently, there are no available data from randomized controlled trials addressing the effective and well tolerated warfarin loading dose for use in MVR patients.
In this study, we compared the conventional protocols [17, 32] that recommend warfarin initiation at a 5 mg dose versus the safe strategy of using the 3 mg dose.
Our results showed that the efficacy of initiating warfarin at 3 mg was found to be less than that of the 5 mg dose, demonstrated by the longer time required to reach the therapeutic INR range, with a comparable safety profile. Furthermore, the shorter mean time to reach the therapeutic INR achieved with the 5 mg dose (5.3 days) was comparable with that observed by Roberts et al., who reported a mean time to reach therapeutic INR of 5.1 days in their study that compared empirical warfarin initiation at 5 mg in MVR patients with a designed protocol that adopted lower warfarin initiation doses [22].
On the contrary, the 5 mg group in the study by Ageno et al. [16], comparing 5 mg and 2.5 mg warfarin initiation in heart valve replacement patients, reported a lower mean time to reach therapeutic INR (1.98) than our 5 mg group (5.3). This discrepancy could be attributed to their lower target INR range (1.5–2.6), which differs from the defined target INR range in our population. Moreover, the shorter time to therapeutic INR attained in our 5 mg group (5.3 days) was comparable with that reported in previous studies by Kovacs et al. (5.6 days) in acute VTE [33] and Shine et al. (5 days) in acute clotting and atrial fibrillation [34].
The longer time to reach therapeutic INR in the 3 mg group resulted in longer bridging days, with higher consumption of enoxaparin and, consequently, higher bridging cost. Although earlier studies have suggested an increased cost of bridging when using reduced loading warfarin doses [33, 35], to the best of our knowledge none of them have estimated the value of this increased cost of bridging therapy. Healthcare cost reduction is of utmost importance, especially when addressing countries with limited healthcare resources and high expenditures such as Egypt.
The proportion of time spent in the therapeutic range may provide a good presentation for anticoagulation management, however this is dependent on the length of the follow-up period after treatment initiation [36]. Several meta-analyses concluded that the higher proportion of time spent in the therapeutic range could be detected after 1 or more months of follow-up and that there was a significant improvement when follow-up periods were extended to 2 or 3 months [34–36].
In the current study, the proportion of time spent in the therapeutic range was not significantly different between the two groups, yet the lack of difference could be attributed to the short period of follow-up in our study that aimed to focus on the early initiation phase of anticoagulation.
Following the warfarin 5 mg loading dose for initiation, the proportion of patients achieving therapeutic INR by day 5 in previous studies varied from 46 to 88% [14, 33, 35]—56% in the current study, compared with 46% reported by Kovacs et al. [33] and 63% reported by Shine et al. [34], although involving patients of different indications. On the contrary, Crowther et al. reported a higher proportion of 88%, which might be attributed to the fact that 44% of their patients suffered malignancy, which could contribute to an increase in coagulability status [14]. In the 3 mg group in our study, the percentage of patients reaching target INR within 3–5 days (28%) was comparable with that previously reported by a retrospective study on adult Thai patients (29%) that studied the same dose for long-term anticoagulation in different indications [23].
In the present study, there was no significant difference between the two groups in the percentage of patients achieving the therapeutic INR within 3–5 days; however, the duration of enoxaparin bridging was significantly lower in the 5 mg group. This reveals that warfarin initiation at 5 mg accelerated reaching target INR, and consequently permitted the earlier removal of pacing wires by cardiac surgeons. Moreover, the inconvenience and pain associated with parenteral bridging and, subsequently, overall treatment costs were reduced with the 5 mg loading dose.
It is worth noting that despite the number of patients with laboratory evidence of over-anticoagulation (INR occasions >4), major bleeding events were only reported in four patients in the 5 mg group and five patients in the 3 mg group. Similarly, a recent study concluded that a rapid rise in INR following warfarin administration was not associated with an increased risk of major bleeding in those patients during the immediate postoperative period [37]. This could be explained by the phenomenon that a rapid initial increase in INR inhibits the clotting cascade, resulting in a relatively net neutral effect on the total risk of bleeding [9].
Minor bleeding events were more common in the 5 mg group. Nonetheless, as evidenced by comparable follow-up periods between groups, this significant difference did not delay attaining stable INR.
Patients achieved stable therapeutic INR in a variable number of days. As a result, each patient’s follow-up period was unique, ranging from 4 to 16 days.
The mean number of days of follow-up did not differ statistically between groups and was comparable with the average postoperative time (7 days) required to achieve a postoperative stable INR value that allows ICU discharge, as previously reported in the literature [38].
Patients’ age, concurrent drugs such as amiodarone and cefazoline, baseline INR, and serum albumin level were all shown to contribute to postoperative warfarin sensitivity [8]. Hence, all patients with a baseline INR > 1.2 were initially excluded and statistical analyses were adjusted for age. Furthermore, there was no significant difference between groups in terms of amiodarone or cefazoline use, as well as baseline serum albumin levels.
Prior guidelines published by the ACCP recommended against routine genotyping [39], but the Clinical Pharmacogenetics Implementation Consortium guidelines recommended incorporating existing genotype information into warfarin dose decisions [40].
Genotype-guided warfarin dosing is not performed at our hospital. Published clinical trials have generated variable results regarding the benefit of genotype-guided warfarin dosing [32, 41]. Some studies showed better safety [42] or shorter time to reach therapeutic INR [43] with genotype-guided warfarin, while others have highlighted the need to develop race-specific dosing algorithms [44, 45]. However, further studies are still needed to determine the cost effectiveness of routine warfarin‐related genotype testing [46].
Currently, it would appear more practical to assign the limited financial resources to the founding of better infrastructure for INR testing, improving patient–provider communication, implementing validated clinical anticoagulation protocols, and enhancing patient adherence rather than using genotype-based dosing algorithms [47, 48]. Accordingly, we presume that genetic-guided dosing is not currently recommended, especially in developing countries.
Based on the findings of the current study in MVR patients, using a safe strategy of warfarin initiation at 3 mg did not improve the effectiveness of anticoagulation or add benefit to safety profiles and resulted in a higher cost of bridging therapy versus the regular warfarin initiation at 5 mg.
Study Limitations
Although our study was powered and the sample size was calculated, the number of patients is still limited. Further studies may demonstrate the effect of a wider range of doses for warfarin initiation. Larger randomized controlled trials that are powered to show differences in bleeding/thromboembolic events are required to confirm the results and to provide more information about anticoagulation complications of heart valve replacement patients. Furthermore, our study can be useful as an hypothesis generator for other randomized studies addressing the different warfarin initiation dosing in other patients with different types of prostheses and its impact on other patient clinical data such as the timing of chest tube and pacing wire removal. In the current study, we roughly estimated the cost of bridging with enoxaparin, yet a full pharmacoeconomic study can better interpret the overall costs.
Conclusion
The efficacy of anticoagulation management using the 5 mg warfarin initiation in MVR patients appeared to be more favorable than that of the 3 mg group. No significant difference was found between groups in terms of major bleeding events or total number of bleeding events. Only minor events were higher in the 5 mg group, and the cost of bridging was higher in the 3 mg group. Further larger randomized studies are required to examine the efficacy and safety of 5 mg versus higher warfarin initiation doses in MVR patients.
Acknowledgements
The authors would like to acknowledge all study patients for their participation in this study. All authors take full responsibility for all aspects of the reliability and freedom from bias of the data presented herein and their discussed interpretation.
Declarations
Conflict of interest
Sarah Sabry, Lamia Mohamed El Wakeel, Ayman Saleh, and Marwa Adel Ahmed have no conflicts of interest to declare that are relevant to the contents of this article. All authors certify that they are not involved in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki and was registered at ClinicalTrials.gov (ID: NCT04235569). The study protocol was approved by the Research Ethics Committee for Experimental and Clinical Studies at the Faculty of Pharmacy, ASU [ethics approval number: Ph.D. (No. 51), 8 October 2017].
Consent to participate
Written informed consent was obtained from all participants prior to participation in this study.
Consent for publication
Not applicable.
Availability of data and material
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Author contributions
All authors contributed to the study’s conception and design. Material preparation and data collection were performed by SS. Analysis was performed by SS, LMEW, MAA, and AS. The first draft of the manuscript was written by SS and all authors edited and revised previous versions of the manuscript. All authors read and approved the final manuscript.
References
- 1.Sun JC, Davidson MJ, Lamy A, Eikelboom JW. Antithrombotic management of patients with prosthetic heart valves: current evidence and future trends. Lancet. 2009;374(9689):565–576. doi: 10.1016/s0140-6736(09)60780-7. [DOI] [PubMed] [Google Scholar]
- 2.Whitlock RP, Sun JC, Fremes SE, Rubens FD, Teoh KH. Antithrombotic and thrombolytic therapy for valvular disease: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e576S–e600S. doi: 10.1378/chest.11-2305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ansell J, Hirsh J, Hylek E, Jacobson A, Crowther M, Palareti G. Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th edition) Chest. 2008;133(6 Suppl):160S–S198. doi: 10.1378/chest.08-0670. [DOI] [PubMed] [Google Scholar]
- 4.White PJ. Patient factors that influence warfarin dose response. J Pharm Pract. 2010;23(3):194–204. doi: 10.1177/0897190010362177. [DOI] [PubMed] [Google Scholar]
- 5.Rahman M, BinEsmael TM, Payne N, Butchart EG. Increased sensitivity to warfarin after heart valve replacement. Ann Pharmacother. 2006;40(3):397–401. doi: 10.1345/aph.1G407. [DOI] [PubMed] [Google Scholar]
- 6.Ageno W, Turpie AG. Exaggerated initial response to warfarin following heart valve replacement. Am J Cardiol. 1999;84(8):905–908. doi: 10.1016/s0002-9149(99)00463-4. [DOI] [PubMed] [Google Scholar]
- 7.Rose JP, Rihn TL, Long SF. Warfarin sensitivity after mechanical heart valve replacement. Pharmacotherapy. 1998;18(4):856–859. [PubMed] [Google Scholar]
- 8.Mohammadi K, Kargar M. Sensitivity to warfarin following cardiac surgery. Ther Adv Drug Saf. 2018;9(12):673–674. doi: 10.1177/2042098618804488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ageno W, Gallus AS, Wittkowsky A, Crowther M, Hylek EM, Palareti G. Oral anticoagulant therapy: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e44S–e88S. doi: 10.1378/chest.11-2292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vahanian A, Alfieri O, Andreotti F, Antunes MJ, Baron-Esquivias G, Baumgartner H, et al. Guidelines on the management of valvular heart disease (version 2012): the Joint Task Force on the Management of Valvular Heart Disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS) Eur J Cardiothorac Surg. 2012;42(4):S1–44. doi: 10.1093/ejcts/ezs455. [DOI] [PubMed] [Google Scholar]
- 11.Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP, 3rd, Guyton RA, et al. 2014 AHA/ACC guideline for the management of patients with valvular heart disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014;63(22):2438–2488. doi: 10.1016/j.jacc.2014.02.537. [DOI] [PubMed] [Google Scholar]
- 12.Roberts GW, Helboe T, Nielsen CB, Gallus AS, Jensen I, Cosh DG, et al. Assessment of an age-adjusted warfarin initiation protocol. Ann Pharmacother. 2003;37(6):799–803. doi: 10.1345/aph.1A372. [DOI] [PubMed] [Google Scholar]
- 13.Roberts GW, Druskeit T, Jorgensen LE, Wing LM, Gallus AS, Miller C, et al. Comparison of an age adjusted warfarin loading protocol with empirical dosing and Fennerty's protocol. Aust N Z J Med. 1999;29(5):731–736. doi: 10.1111/j.1445-5994.1999.tb01623.x. [DOI] [PubMed] [Google Scholar]
- 14.Crowther MA, Ginsberg JB, Kearon C, Harrison L, Johnson J, Massicotte MP, et al. A randomized trial comparing 5-mg and 10-mg warfarin loading doses. Arch Intern Med. 1999;159(1):46–48. doi: 10.1001/archinte.159.1.46. [DOI] [PubMed] [Google Scholar]
- 15.Harrison L, Johnston M, Massicotte MP, Crowther M, Moffat K, Hirsh J. Comparison of 5-mg and 10-mg loading doses in initiation of warfarin therapy. Ann Intern Med. 1997;126(2):133–136. doi: 10.7326/0003-4819-126-2-199701150-00006. [DOI] [PubMed] [Google Scholar]
- 16.Ageno W, Turpie AG, Steidl L, Ambrosini F, Cattaneo R, Codari RL, et al. Comparison of a daily fixed 2.5-mg warfarin dose with a 5-mg, international normalized ratio adjusted, warfarin dose initially following heart valve replacement. Am J Cardiol. 2001;88(1):40–44. doi: 10.1016/s0002-9149(01)01582-x. [DOI] [PubMed] [Google Scholar]
- 17.Heneghan C, Tyndel S, Bankhead C, Wan Y, Keeling D, Perera R, et al. Optimal loading dose for the initiation of warfarin: a systematic review. BMC Cardiovasc Disord. 2010;10:18. doi: 10.1186/1471-2261-10-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Barnes GD, Kline-Rogers E. Engaging with quality improvement in anticoagulation management. J Thromb Thrombolysis. 2015;39(3):403–409. doi: 10.1007/s11239-015-1184-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kulik A, Rubens FD, Wells PS, Kearon C, Mesana TG, van Berkom J, et al. Early postoperative anticoagulation after mechanical valve replacement: a systematic review. Ann Thorac Surg. 2006;81(2):770–781. doi: 10.1016/j.athoracsur.2005.07.023. [DOI] [PubMed] [Google Scholar]
- 20.Sanaani A, Yandrapalli S, Harburger JM. Antithrombotic management of patients with prosthetic heart valves. Cardiol Rev. 2018;26(4):177–186. doi: 10.1097/crd.0000000000000189. [DOI] [PubMed] [Google Scholar]
- 21.Zhou S, Sheng XY, Xiang Q, Wang ZN, Zhou Y, Cui YM. Comparing the effectiveness of pharmacist-managed warfarin anticoagulation with other models: a systematic review and meta-analysis. J Clin Pharm Ther. 2016;41(6):602–611. doi: 10.1111/jcpt.12438. [DOI] [PubMed] [Google Scholar]
- 22.Roberts G, Razooqi R, Quinn S. Comparing usual care with a warfarin initiation protocol after mechanical heart valve replacement. Ann Pharmacother. 2017;51(3):219–225. doi: 10.1177/1060028016676830. [DOI] [PubMed] [Google Scholar]
- 23.Suwanawiboon B, Kongtim P, Chinthammitr Y, Ruchutrakool T, Wanachiwanawin W. The efficacy of 3-mg warfarin initiating dose in adult Thai patients, who required long-term anticoagulant therapy. J Med Assoc Thai. 2011;94(Suppl 1):S225–S231. [PubMed] [Google Scholar]
- 24.MAQI2. Anticoagulation toolkit: a Consortium-Developed Quick Reference for Anticoagulation. 2018. https://cdn.ymaws.com/sites/opso.site-ym.com/resource/resmgr/2018_Winter/Magnuson_MAQIAC_toolkitfull.pdf.
- 25.University of Connecticut. Warfarin Collaborative Practice Dosing Protocol. 2018. https://cupdf.com/document/protocol-warfarin-collaborative-practice-dosing-protocol-warfarin-should.html.
- 26.Dumont Z, Kosar L, Regier L, Jensen B. Warfarin tips & dosing nomograms. 2013. https://www.healthcare.uiowa.edu/familymedicine/fpinfo/Docs/Warfarin_Tips.pdf.
- 27.Kuruvilla M, Gurk-Turner C. A review of warfarin dosing and monitoring. Proc (Bayl Univ Med Cent). 2001;14(3):305–306. doi: 10.1080/08998280.2001.11927781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mehran R, Rao SV, Bhatt DL, Gibson CM, Caixeta A, Eikelboom J, et al. Standardized bleeding definitions for cardiovascular clinical trials. Circulation. 2011;123(23):2736–2747. doi: 10.1161/CIRCULATIONAHA.110.009449. [DOI] [PubMed] [Google Scholar]
- 29.Basch E, Reeve BB, Mitchell SA, Clauser SB, Minasian LM, Dueck AC, et al. Development of the National Cancer Institute's patient-reported outcomes version of the common terminology criteria for adverse events (PRO-CTCAE) J Natl Cancer Inst. 2014;106(9):dju244. doi: 10.1093/jnci/dju244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5(1):13. doi: 10.1186/1471-2288-5-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Saksena D, Muralidharan S, Mishra YK, Kanhere V, Mohanty BB, Srivastava CP, et al. Anticoagulation management in patients with valve replacement. J Assoc Physicians India. 2018;66(1):59–74. [PubMed] [Google Scholar]
- 32.Mahtani KR, Heneghan CJ, Nunan D, Bankhead C, Keeling D, Ward AM, et al. Optimal loading dose of warfarin for the initiation of oral anticoagulation. Cochrane Database Syst Rev. 2012;12:CD008685. 10.1002/14651858.CD008685.pub2. [DOI] [PMC free article] [PubMed]
- 33.Kovacs MJ, Rodger M, Anderson DR, Morrow B, Kells G, Kovacs J, et al. Comparison of 10-mg and 5-mg warfarin initiation nomograms together with low-molecular-weight heparin for outpatient treatment of acute venous thromboembolism. A randomized, double-blind, controlled trial. Ann Intern Med. 2003;138(9):714–719. doi: 10.7326/0003-4819-138-9-200305060-00007. [DOI] [PubMed] [Google Scholar]
- 34.Shine D, Patel J, Kumar J, Malik A, Jaeger J, Maida M, et al. A randomized trial of initial warfarin dosing based on simple clinical criteria. Thromb Haemost. 2003;89(2):297–304. doi: 10.1055/s-0037-1613446. [DOI] [PubMed] [Google Scholar]
- 35.Fanikos J, Tsilimingras K, Kucher N, Rosen AB, Hieblinger MD, Goldhaber SZ. Comparison of efficacy, safety, and cost of low-molecular-weight heparin with continuous-infusion unfractionated heparin for initiation of anticoagulation after mechanical prosthetic valve implantation. Am J Cardiol. 2004;93(2):247–250. doi: 10.1016/j.amjcard.2003.09.054. [DOI] [PubMed] [Google Scholar]
- 36.Erkens PM, ten Cate H, Büller HR, Prins MH. Benchmark for time in therapeutic range in venous thromboembolism: a systematic review and meta-analysis. PLoS ONE. 2012;7(9):e42269. doi: 10.1371/journal.pone.0042269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Olson LM, Nei AM, Dierkhising RA, Joyce DL, Nei SD. Warfarin-induced rapid rise in INR post-cardiac surgery is not associated with increased bleeding risk. Ann Pharmacother. 2019;53(12):1184–1191. doi: 10.1177/1060028019858677. [DOI] [PubMed] [Google Scholar]
- 38.Almashrafi A, Elmontsri M, Aylin P. Systematic review of factors influencing length of stay in ICU after adult cardiac surgery. BMC Health Serv Res. 2016;16:318. doi: 10.1186/s12913-016-1591-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Holbrook A, Schulman S, Witt DM, Vandvik PO, Fish J, Kovacs MJ, et al. Evidence-based management of anticoagulant therapy: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e152S–e184. doi: 10.1378/chest.11-2295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Johnson JA, Gong L, Whirl-Carrillo M, Gage BF, Scott SA, Stein CM, et al. Clinical Pharmacogenetics Implementation Consortium Guidelines for CYP2C9 and VKORC1 genotypes and warfarin dosing. Clin Pharmacol Ther. 2011;90(4):625–629. doi: 10.1038/clpt.2011.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cavallari LH, Nutescu EA. Warfarin pharmacogenetics: to genotype or not to genotype, that is the question. Clin Pharmacol Ther. 2014;96(1):22–24. doi: 10.1038/clpt.2014.78. [DOI] [PubMed] [Google Scholar]
- 42.Yang T, Zhou Y, Chen C, Lu M, Ma L, Cui Y. Genotype-guided dosing versus conventional dosing of warfarin: A meta-analysis of 15 randomized controlled trials. J Clin Pharm Ther. 2019;44(2):197–208. doi: 10.1111/jcpt.12782. [DOI] [PubMed] [Google Scholar]
- 43.Hao Y, Yang J, Zheng X, Hu Y, Yan X, Zhang L. Chinese patients with heart valve replacement do not benefit from warfarin pharmacogenetic testing on anticoagulation outcomes. Ther Drug Monit. 2019;41(6):748–754. doi: 10.1097/ftd.0000000000000664. [DOI] [PubMed] [Google Scholar]
- 44.Shendre A, Dillon C, Limdi NA. Pharmacogenetics of warfarin dosing in patients of African and European ancestry. Pharmacogenomics. 2018;19(17):1357–1371. doi: 10.2217/pgs-2018-0146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kimmel SE, French B, Kasner SE, Johnson JA, Anderson JL, Gage BF, et al. A pharmacogenetic versus a clinical algorithm for warfarin dosing. N Engl J Med. 2013;369(24):2283–2293. doi: 10.1056/NEJMoa1310669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tse G, Gong M, Li G, Wong SH, Wu WKK, Wong WT, et al. Genotype-guided warfarin dosing vs. conventional dosing strategies: a systematic review and meta-analysis of randomized controlled trials. Br J Clin Pharmacol. 2018;84(9):1868–1882. doi: 10.1111/bcp.13621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Syn NL, Wong AL, Lee SC, Teoh HL, Yip JWL, Seet RC, et al. Genotype-guided versus traditional clinical dosing of warfarin in patients of Asian ancestry: a randomized controlled trial. BMC Med. 2018;16(1):104. doi: 10.1186/s12916-018-1093-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Stergiopoulos K, Brown DL. Genotype-guided vs clinical dosing of warfarin and its analogues: meta-analysis of randomized clinical trials. JAMA Intern Med. 2014;174(8):1330–1338. doi: 10.1001/jamainternmed.2014.2368. [DOI] [PubMed] [Google Scholar]