Abstract
We have previously shown that the methicillin-resistance gene mecA of Staphylococcus aureus strain N315 is localized within a large (52-kb) DNA cassette (designated the staphylococcal cassette chromosome mec [SCCmec]) inserted in the chromosome. By sequence determination of the entire DNA, we identified two novel genes (designated cassette chromosome recombinase genes [ccrA and ccrB]) encoding polypeptides having a partial homology to recombinases of the invertase/resolvase family. The open reading frames were found to catalyze precise excision of the SCCmec from the methicillin-resistant S. aureus chromosome and site-specific as well as orientation-specific integration of the SCCmec into the S. aureus chromosome when introduced into the cells as a recombinant multicopy plasmid. We propose that SCCmec driven by a novel set of recombinases represents a new family of staphylococcal genomic elements.
Methicillin-resistant Staphylococcus aureus (MRSA) was first isolated in England in 1961 shortly after the development of methicillin, the first penicillinase-resistant semisynthetic penicillin (15). Since then, MRSA has become the most prevalent pathogen causing hospital infection throughout the world, and MRSA incidence is still increasing in many countries (1). MRSA is resistant to practically all β-lactam antibiotics, a class of antibiotics represented by penicillins and cephalosporins (3).
The β-lactam resistance of MRSA is caused by the production of a novel penicillin-binding protein (PBP) designated PBP 2′ (or PBP 2a), which, unlike the intrinsic set of PBPs (PBP 1 to 4) of S. aureus, has remarkably reduced binding affinities to β-lactam antibiotics (9, 24, 30). Despite the presence of otherwise inhibitory concentrations of β-lactam antibiotics, MRSA can continue cell wall synthesis solely depending upon the uninhibited activity of PBP 2′ (21). PBP 2′ is encoded by a mecA gene located on the chromosome of MRSA. In 1987, the mecA gene was cloned from a Japanese MRSA strain, and its sequence was determined (20, 26). The mecA gene is widely distributed among S. aureus as well as coagulase-negative staphylococci (13, 28). Therefore, it has been speculated that the methicillin resistance determinant (mec determinant) is freely transmissible among staphylococcal species. However, with a detailed molecular epidemiological study, Kreiswirth et al. have proposed that MRSA originated from a single or two ancestral clones (16). This led to the view that the frequency of inter- or intraspecies transmission of mecA is a rather limited process and that mec transmission may not be due to specialized transmission machinery, such as a transposon.
We have recently cloned and sequenced the entire chromosomal region surrounding the mecA gene, which is additionally present in the MRSA chromosome and is absent from the chromosome of methicillin-susceptible S. aureus (MSSA) (referred to herein as mecDNA), from a Japanese pre-MRSA strain, N315 (10). We identified the mecDNA-S. aureus chromosome junction points and the overall structure of mecDNA, and revealed that mecDNA constitutes a genomic island or antibiotic resistance island of S. aureus (14). In this study, based on the structure of mecDNA, we report that mecDNA is a new class of genomic element designated SCCmec (for staphylococcal cassette chromosome mec [12]) driven by two site-specific recombinase genes designated ccrA and ccrB (for cassette chromosome recombinases A and B).
MATERIALS AND METHODS
Bacteria and growth condition.
Pre-MRSA strain N315 and its SCCmec excising strain N315ex used in this study have been described previously (14). All the strains and their transformants were cultivated in brain heart infusion (BHI) broth (Becton Dickinson Microbiology Systems, Sparks, Md.). The antibiotics tetracycline (Sigma Chemical Co., St. Louis, Mo.) and tobramycin (Shionogi Co., Osaka, Japan) were used at the concentration of 10 μg/ml.
Construction of recombinant plasmids.
Recombinant plasmid pSR harboring intact ccrA and ccrB genes was constructed by cloning the BamHI-digested DNA fragment containing ccr genes into the unique BamHI site of the plasmid vector pYT3 (8). The DNA fragment containing ccr genes was prepared by PCR using the DNA extracted from N315 as a template. The two primers used were 5′-AAAAGGATCCATTAGCCGATTTGGTAATTGAA-3′ and 5′-AAAAGGATCCTCTGCTTCTTCGAATCTGCAAAT-3′ (introduced BamHI sites are underlined), which correspond to the nucleotides from base positions 24,004 to 24,025, and to the complementary nucleotides from positions 27,365 to 27,343 of the SCCmec sequence (accession no. D86934), respectively. To construct pSRA*, the BamHI fragment of pSR was subcloned into the BamHI site of pUC119ΔH, a derivative of pUC119 whose HindIII site had been eliminated by treatment with the Klenow fragment of DNA polymerase I and self-ligation. The resultant plasmid, pUCSR, was digested with BalI and HindIII and flush ended by Klenow treatment, which was followed by ligation to delete the BalI-HindIII fragment (344 bp) of the ccrA gene. Then, the BamHI fragment was cut out of the plasmid and was ligated into the BamHI site of pYT3 to obtain pSRA*. To construct pSRB*, pUCSR was cut with BstXI and MluI to remove the 252-bp BstXI-MluI fragment of the ccrB gene, and this was followed by Klenow treatment and self-ligation. The BamHI fragment of the resultant plasmid was cut out and ligated into the BamHI site of pYT3 to obtain pSRB*.
To construct attSCC-containing recombinant plasmids, the DNA fragment containing the attSCC sequence on the putative closed circular SCCmec cassette was amplified by PCR using the two primers mR7 and mL2 (see below), and the DNA was extracted from N315 (pSR) and used as a template. The amplified DNA was digested with SalI, and the resultant 933-bp fragment was cloned into a unique SalI site of plasmid pYT3 to obtain pYT3att. Then, the attSCC fragment was cut out of pYT3att with SalI and introduced into the SalI sites of pSR, pSRA*, and pSRB* to obtain pSRatt, pSRA*att, and pSRB*att, respectively.
Nucleotide sequencing.
The DNA fragments for sequencing were obtained by PCR amplification with genomic DNA as templates. After purification using a QIAquick PCR purification kit (QIAGEN, Hilden, Germany), the fragments of PCR products were directly sequenced with a set of synthetic oligonucleotide primers (see below) using a dye-labeled terminator–Taq DNA polymerase cycle sequencing kit (Applied Biosystems Inc., Foster City, Calif.). The sequence was read on a 373A automated fluorescent DNA sequencing system (Perkin-Elmer, Foster City, Calif.). All the computer analyses of nucleotide sequences were carried out using programs in The Wisconsin Package (version 9.0; Genetics Computer Group [GCG], Madison, Wis.). A homology search was performed using BLAST and TFastA programs accessed via the EMBL (release no. 55.0) and GenBank (release no. 107.0) databases and the FastA program accessed via the SWISS-PROT database. (release no. 35.0).
PFGE.
Pulsed-field gel electrophoresis (PFGE) was performed with a modification as described previously (32). For preparation of sample plugs, ca. 2 × 106 cells were embedded in 37.5 μl (1.5 by 5 by 5 mm) of 1% (wt/vol) low-temperature-melting agarose (Agarose Low Melt Preparative Grade; Bio-Rad Laboratories, Hercules, Calif.) containing 40 μg of lysostaphin (Sigma Chemical Co.) per ml. The sample plugs were incubated with 1% (wt/vol) N-lauroylsarcosine (Sigma Chemical Co.)–0.5 M EDTA, pH 7.5, at 37°C for 1 h and further incubated with a solution containing 2 mg of proteinase K (Sigma Chemical Co.) per ml, 1% (wt/vol) N-lauroylsarcosine (Sigma Chemical Co.), and 0.5 M EDTA, pH 7.5, at 50°C for 24 h. The plugs were then incubated with 1 mM phenylmethylsulfonyl fluoride (Sigma Chemical Co.) at 50°C for 30 min, followed by washing three times with Tris-EDTA (20 min each at 4°C). The samples were then treated with 60 U of SmaI (Takara Shuzo Co. Ltd., Kyoto, Japan) at 30°C for 24 h and next were treated with 200 μg of proteinase K at 37°C for 1 h. The final concentrations of SmaI and proteinase K were 0.4 U/μl and 100 μg/ml, respectively. They were further washed three times with Tris-EDTA at 4°C for 20 min and subjected to PFGE with 1.2% agarose gel (high-strength analysis-grade agarose; Bio-Rad Laboratories). The PFGE was performed at 4°C for 20 h in 0.5× Tris-borate-EDTA running buffer using a CHEF DRII apparatus (Bio-Rad Laboratories) with an electric field strength of ∼6 V/cm and a pulse time of 20 s.
Electroporation.
Exponentially growing cultures of strain N315 or N315ex were harvested at an optical density at 600 nm of 0.5 and washed with prechilled 1.1 M sucrose three times. The pellet was resuspended in 100 μl of EP buffer (1.1 M sucrose, 2 mM MgCl2, 14 mM KH2PO4-Na2HPO4 [pH 7.4]). To the cell suspension, 5 μg of the plasmid DNA was added, and the mixture was kept on ice for 25 min. An electric pulse of 25 μF, 2.5 kV, and 100 Ω was delivered by a Gene Pulser (Nippon Bio-Rad Laboratories, Tokyo, Japan). The cells were then transferred into 0.5 ml of 1.1 M sucrose in BHI broth and incubated for 2 h at 30°C (permissive temperature for plasmid replication) before aliquots were plated on BHI agar plates containing tetracycline (10 μg/ml). After a 48-h incubation at 30°C, mature colonies were picked and subjected to rapid plasmid analysis by digestion with various restriction enzymes to confirm the integrity of the introduced plasmid. The plasmid preparation procedure has been described previously (32). The transformants were further colony purified by streaking them onto the BHI agar plate with tetracycline (10 μg/ml) and incubating the plate overnight before establishing the transformant strains as those used in this study.
Southern blot hybridization.
Southern transfer of DNA from the PFGE gel to a nylon membrane was performed as described previously (32). Hybridization took place in a hybridization solution (50% formamide, 5× SSC [1× SSC is 0.15 M NaCl plus 0.015 M sodium citrate], 0.3% sodium dodecyl sulfate [SDS], 2% blocking reagent [Boehringer, Mannheim, Germany]) containing 20 ng of digoxigenin-labeled DNA probe per ml. Incubation was carried out for 16 h at 42°C. The orfX probe was prepared by using primers 5′-CCACGCATAATCTTAAATGCTCT-3′ and 5′-AAACGACATGAAAATCACCAT-3′ (primer cR2 [14]), which corresponded to the nucleotides from base positions 56,357 to 56,379 and complementary nucleotides from base positions 56,824 to 56,804 of the reported nucleotide sequence of orfX (accession no. D86934), respectively. The probe for the ermA gene was prepared using synthetic oligonucleotides 5′-TGAAACAATTTGTAACTATTGA-3′ and 5′-TGAACCAGAAAAACCCTAAAGA-3′ as primers, which corresponded to the nucleotides from positions 33,804 to 33,825 and the complementary nucleotides from position 34,530 to 34,509, respectively, of the reported nucleotide sequence of the N315 SCCmec (accession no. D86934 [14]). The cloned DNA fragment SJ2, which contains Tn554 of SCCmec, was used as the template for the PCR amplification of the ermA gene (14). These probes were labeled with digoxigenin using a DNA labeling kit (Boehringer). The hybridized filter (Biodyne A; Pall Biosupport Co., Glen Cove, N.Y.) was washed twice in 2× SSC with 0.1% SDS for 5 min at room temperature and then in 1× SSC with 0.1% SDS for 15 min at 68°C. Visualization of the signal was achieved using an alkaline phosphatase-conjugated antidigoxigenin antibody (Boehringer) and the chemiluminescent substrate CSPD (Boehringer) according to the procedure recommended by the manufacturers.
Synthetic oligonucleotides used as primers for PCR and nucleotide sequencing.
The primers used in this study and their nucleotide sequences with references, if not specified above, are listed below in the order of appearance in the text: cL1, 5′-ATTTAATGTCCACCATTTAACA-3′ (14); mL1, 5′-GAATCTTCAGCATGTGATTTA-3′ (corresponds to the complementary nucleotides from positions 4,977 to 4,957 of the nucleotide sequence [accession no. D86934]); mR8, 5′-ATGAAAGACTGCGGAGGCTAACT-3′ (corresponds to the nucleotides from base position 56,636 to 56,658 [accession no. D86934]); cR2, 5′-AAACGACATGAAAATCACCAT-3′ (14); mA1, 5′-TGCTATCCACCCTCAAACAGG-3′ (10); mA2, 5′-AACGTTGTAACCACCCCAAGA-3′ (10); mR7, 5′-AAAAAGTCGACACTGCTTGGGTAACTTATCATGGA-3′; mL2, 5′-AAAAAGTCGACATCACAGTAGTGCAAAGCACGTCGA-3′; α, 5′-TTTCACACAGGAAACAGCTATGAC-3′; and β, 5′-ATCACGATATTGCTTATAAGCA-3′ (corresponds to the nucleotides from base positions 26,673 to 26,694 of the reported sequence [accession no. D86934]).
RESULTS
ccrA and ccrB gene-mediated excision of SCCmec.
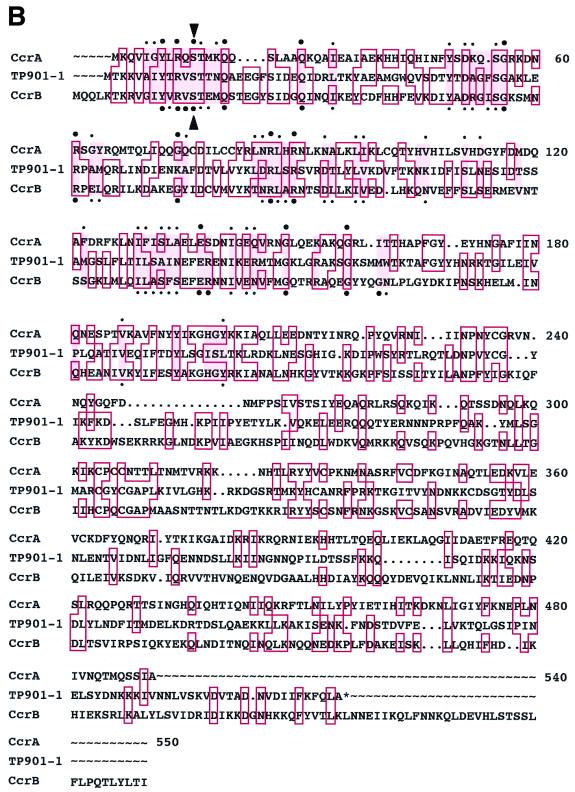
Two novel ORFs, ccrA and ccrB, were located in the midst of SCCmec (Fig. 1A). CcrA and CcrB polypeptides of 449 and 542 amino acids were potentially encoded by the ORFs. A Shine-Dalgarno sequence and −10 and −35 presumptive promoter sequences were identified immediately upstream of ccrA. On the other hand, although Shine-Dalgarno sequence was found for ccrB, no candidates for promoter sequences were identified in the adjacent upstream region of the gene. This suggests that the two ORFs may be transcribed as a single mRNA.
FIG. 1.
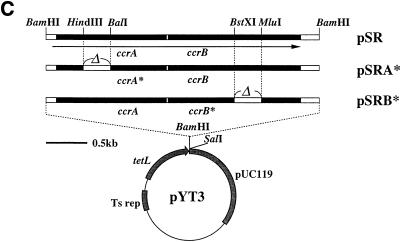
Identification of ccr genes. (A) Genomic structure of SCCmec. Locations of the essential genes are illustrated. The left and right chromosome-SCCmec junctions, attL and attR, were tentatively and operationally defined in this study by PCR primer combinations of cL1 and mL1 for attL, and mR8 and cR2 for attR. The MSSA chromosomal parts of the attL and attR were defined as attB-L and attB-R, delimited by primers cL1 and cR2, respectively. The SCCmec parts of these elements were defined as attSCC-L and attSCC-R, delimited by the primers mL1 and mR8, respectively: i.e., attL is attB-L plus attSCC-L, and attR is attB-R plus attSCC-R. Transposon Tn554 (23), encoding resistance to erythromycin and spectinomycin, was located in upstream of the mecI-mecR1-mecA gene complex (14). Plasmid pUB110, encoding resistance for kanamycin-tobramycin and bleomycin, was inserted between two insertion sequences IS431 (or IS257) (2, 7, 14, 22, 27). Nucleotide sequences around the left and right boundaries of SCCmec, attL and attR, are shown at the bottom of the panel. Inverted repeats, IRscc-L and IRscc-R, at both extremities of SCCmec are indicated by thin arrows. Thick arrows indicate the direct repeats, DR-B and DR-SCC. The nucleotide sequence of the N315 chromosome containing the entire SCCmec (56,939 bp) is deposited in DDBJ/EMBL/GenBank under accession number D86934. (B) Deduced amino acid sequences of ccrA and ccrB. CcrA and CcrB amino acid sequence were aligned with that of TP901-1 integrase (4). The alignment was performed using the Pile-Up program with a scoring matrix of pam 250 in the Wisconsin Package (version 9.0; Genetics Computer Group). Amino acids shared by the three peptides are boxed in red. CcrA and CcrB had 26.5 and 37.4% amino acid identity, respectively, with the TNP901-1 integrase. Large bullets indicate amino acid residues of CcrA or CcrB shared by site-specific recombinases of the invertase/resolvase family. Small bullets indicate amino acid substitution within the same class of amino acids. Of the 64 amino acid positions well conserved among the recombinases of the invertase/resolvase family (shown in pink) (25), 44 and 47 amino acids, respectively, were conserved in CcrA and CcrB. An arrowhead indicates the presumptive serine involved in the 5′ phosphoseryl linkage of the recombinase to DNA characteristically conserved in the NH2-terminal catalytic domain of site-specific recombinases of the invertase/resolvase family (25). (C) Construction of plasmids carrying intact and disrupted ccr genes. Restriction maps of pSR, pSRA* (a derivative of pSR with a partially deleted ccrA), and pSRB* (a derivative of pSR with a partially deleted ccrB) are shown. Arrows indicate the direction of transcription of the structural gene ccr and tetL. The respective set of ccrA and ccrB genes was inserted at the BamHI site of pYT3.
Two ORFs, ccrA and ccrB, were subcloned into a shuttle vector, pYT3, which has a temperature-sensitive origin of replication (copy numbers: 14 at 30°C, 1 at 37°C, and 0 at 42°C). The recombinant plasmids obtained were pSR, pSRA* (with a deletion introduced in ccrA), and pSRB* (with a deletion introduced in ccrB) (Fig. 1C). These recombinant plasmids were introduced into N315 by electroporation (17).
After cultivation of these strains in drug-free broth for 20 h at 30°C, we evaluated the proportion of the cells which had lost SCCmec in the culture by plating onto agar with and without tobramycin. The cultures of strains N315, N315(pYT3), N315(pSRA*), and N315(pSRB*) yielded essentially equal numbers of colonies on the BHI agar plates with or without tobramycin. On the other hand, the culture of strain N315(pSR), harboring both intact ccrA and ccrB genes, generated tobramycin-susceptible cells, which constituted 51 to 62% of the entire cell population in repeated experiments. A representative tobramycin-susceptible subclone, N315ex was established from the experiment.
Antibiotic susceptibility patterns of N315 and N315ex were compared on the basis of MICs determined using a standard procedure (17). The MICs of various antibiotics (in micrograms per milliliter) for N315 and N315ex were, respectively, 64 and 2 (oxacillin), 64 and 4 (ceftizoxime), 256 and 0.5 (tobramycin), 256 and 2 (kanamycin), and >512 and 16 (bleomycin). The data showed that all the resistance phenotypes were associated with the presence of SCCmec except for erythromycin resistance. Southern blot hybridization analysis of SmaI-digested DNAs of N315 and N315ex using the ermA gene-specific probe detected four restriction fragments (>436.5, 420, 320, and 110 kb in size) in both strains in addition to the 215-kb fragment of N315 on which SCCmec was localized (data not shown; see below for the PFGE-separated SmaI-restriction fragment pattern).
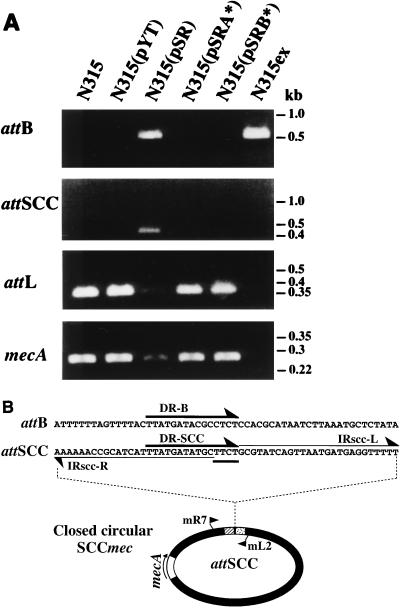
To confirm that the loss of resistance was caused by the excision of SCCmec, DNAs were extracted from transformants N315(pYT3), N315(pSR), N315(pSRA*), and N315(pSRB*), as well as from control strains N315 and N315ex. PCR using the combination of primers cL1 and cR2, which theoretically amplifies the 0.5-kb DNA fragment containing attB when SCCmec is excised from the chromosome [Fig. 2A), did not amplify any DNA fragment with N315, N315(pYT3), N315(pSRA*), and N315(pSRB*) DNAs as templates but the 0.5-kb DNA fragment was amplified with DNAs extracted from N315(pSR) and N315ex (Fig. 2A). The nucleotide sequencing of the amplified 0.5-kb DNA fragments from both N315(pSR) and N315ex DNAs showed that they contained the attB sequence theoretically expected to be generated by the precise excision of SCCmec from the chromosome (Fig. 2B). The SCCmec excision was considered to be precise since the nucleotide sequencing using the total DNA extracted from N315(pSR) as a template yielded a readable nucleotide sequence of attB which was identical to that yielded when the N315ex DNA was used as a template.
FIG. 2.
Precise excision of SCCmec mediated by ccrA and ccrB. (A) Detection of ccr-mediated SCCmec excision and appearance of attSCC. Template DNAs for PCR were extracted from overnight culture of the colony purified transformant strains in BHI broth with tetracycline (10 μg/ml). Four sets of primers were used to detect precise excision and the closed circle form of SCCmec. Primers cR2 and cL1 were used to detect attB (549 bp). Primers mL1 and mR8 were used to identify attSCC (456 bp). Primers cL1 and mL1 were used to detect attL (371 bp). Primers mA1 and mA2 were used to detect a part of mecA gene (286 bp). Note that precise excision of SCCmec occurred only in N315(pSR) and N315ex and that attSCC was detected only in N315(pSR). The molecular weight marker was a 1-kb ladder (Gibco-BRL, Gaithersburg, Md.) and only the relevant sizes are indicated. (B) Generation of attSCC and attB. The presumptive closed circular form of SCCmec is illustrated with attSCC, which appeared to be generated by head-to-tail ligation of attSCC-L (stippled box) and attSCC-R (striped box) (see Fig. 1A). Nucleotide sequences of attB in amplified DNA fragments from N315ex and N315(pSR) are also illustrated. The primers mR7 and mL2 were used for the cloning of attSCC into pYT3.
Concomitant appearance of a rearranged attachment sequence, attSCC, with SCCmec excision.
Upon the precise excision, the excised SCCmec was expected to form an extrachromosomal closed circular DNA. To test this possibility, PCR was performed using a pair of primers set divergently at both termini of SCCmec (primers mR8 and mL1 [Fig. 1A]). Figure 2A shows that a DNA fragment of 0.5 kb was specifically amplified from N315(pSR) but not from the other test strains. Nucleotide sequencing of the fragment showed a novel nucleotide sequence presumably generated by a head-to-tail ligation of attSCC-R and attSCC-L of both termini of SCCmec, forming a novel attachment sequence designated attSCC (Fig. 2B). The divergently arranged 27-bp inverted repeats, IR-L and IR-R, with a core of four nucleotides, TTCT (underlined in Fig. 2B), between them and the presence of direct repeats, DR-SCC, were characteristic of the structure of the attSCC. The data suggested that a closed circular form of SCCmec was produced as a result of the precise excision of SCCmec from the N315 chromosome.
Loss of SCCmec during passage of N315(pSR).
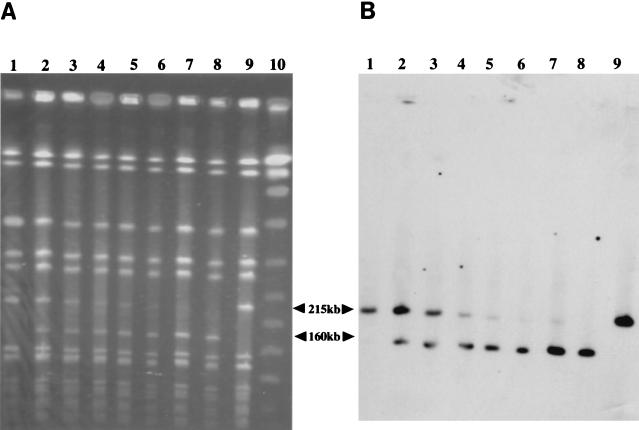
To see the rate of loss of SCCmec from the culture of N315(pSR) and confirm the precision of excision mediated by ccr genes, PFGE analysis on the total DNAs that were consecutively extracted from the culture was performed (Fig. 3A). Compared to the culture of N315 and N315(pYT3) (lanes 1 and 9, respectively), N315(pSR) had an extra band of about 160-kb (Fig. 3A, lane 2). The band was indistinguishable from that of N315ex, from which a 215-kb band of N315 was missing (lane 8). As long as the N315(pSR) was cultured in tobramycin-containing BHI broth, the proportions of the two bands were the same: densitometric measurement of the 215-, and 160-kb bands yielded a ratio of about 7 to 8:1 (data not shown). When, the cells were washed and resuspended in BHI broth without tobramycin, the intensity of the 215-kb band of N315(pSR) started decreasing and continued to do so during the course of the passage, while the 160-kb band continued increasing in intensity (Fig. 3A, lanes 2 to 7). Southern blot hybridization of the PFGE bands using orfX DNA as a probe showed that both the 215- and 160-kb bands carried orfX (Fig. 3B). Rehybridization of the membrane using the mecA gene probe showed that the 215-kb band hybridized with the probe, but the 160-kb band did not (data not shown). This clearly showed that the loss of mecA gene was caused by the precise excision of SCCmec. Greater than 99% of the cells in culture became susceptible to tobramycin and methicillin after 10 days' passage of N315(pSR) without tobramycin.
FIG. 3.
Loss of SCCmec during the cultivation of N315(pSR). (A) PFGE banding patterns of SmaI-digested total DNAs extracted from N315(pSR) on different days of passage. (B) Southern blot hybridization of the DNA fragments of the gel in panel A after transfer to a sheet of nylon membrane. The probe used was orfX. Lanes: 1, N315 cultivated for 24 h with tobramycin (10 μg/ml); 2, N315(pSR) cultivated for 24 h with tobramycin; 3 to 7, N315(pSR) after cultivation without tobramycin for 1, 4, 5, 7, and 9 days, respectively; 8, N315ex cultivated for 24 h without tobramycin; 9, N315(pYT3) cultivated for 24 h with tobramycin; 10, lambda DNA marker for PFGE (lambda ladder; Bio-Rad Laboratories). The sizes of the marker DNA (from top to bottom) were 485.0, 436.5, 388.0, 339.5, 291.0, 242.5, 194.0, 145.5, 97.0, and 48.5 kb. The arrows at the side of each figure indicate the DNA fragments hybridizable with the orfX probe.
Site-specific, orientation-specific integration of experimental plasmid mediated by ccr genes.
To investigate whether the putative closed circular form of SCCmec serves as a substrate for integration when introduced into a MSSA cell, we constructed experimental plasmids pSRatt and pSRtta on which both ccr genes and the presumptive attachment sequence, attSCC, are subcloned (14). The plasmid pSRtta was identical with pSRatt except that the attSCC fragment was cloned in the opposite orientation (Fig. 4). The resultant recombinant plasmids were introduced into strain N315ex by electroporation, and transformants were selected by overnight incubation on BHI agar plates containing tetracycline (10 μg/ml) at 30°C. The colony of each transformant was respread onto BHI agar plates containing tetracycline (10 μg/ml) and incubated overnight at 30 or 43°C (nonpermissive temperature for the plasmid replication) before enumeration of the generated colonies. Two strains, N315ex(pSRatt) and N315ex(pSRtta), generated significant number of colonies at 43°C, i.e., 17.7 and 21.2%, respectively, of those grown at 30°C, whereas the other strains generated only small number of colonies; i.e., <0.001% of those grown at 30°C. The result showed that the chromosomal integration of the plasmids occurred only when attSCC and an intact set of ccrA and ccrB genes were present on the plasmids.
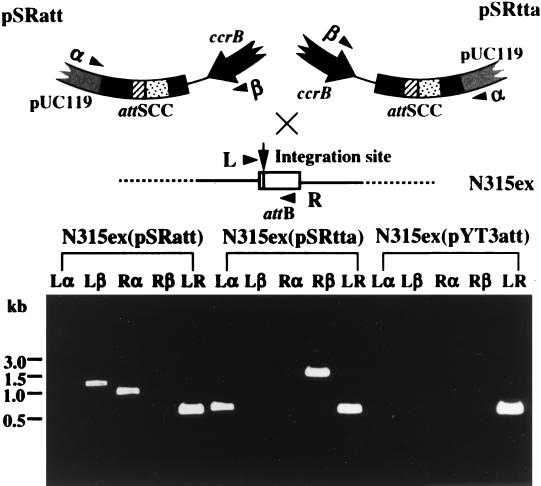
FIG. 4.
Site-, and orientation-specific integration of experimental plasmids mediated by ccrA and ccrB. PCR detection of the left and right plasmid-chromosome junctions was performed with four crisscross combinations of primers with DNAs extracted from the culture of N315ex(pSRatt), N315ex(pSRtta), and N315ex(pYT3att) as templates. Four primers L (cL1; see Fig. 1A), R (cR2; see Fig. 1A), α, and β were used. The location and direction of the four primers are illustrated. Only the regions adjacent to the introduced attSCC on the plasmids and the attB on N315ex chromosome are illustrated. The expected sizes of the amplified DNA fragments generated by a site-specific integration of the plasmids were 610 bp (L-α), 1,255 bp (L-β), 1,649 bp (R-α), 1004 bp (R-β), and 549 bp (L-R). The results show a strict site and orientation specificity of ccr-mediated integration. The molecular weight marker used was a 1-kb ladder.
To learn whether the integration of the experimental plasmids was site- and orientation-specific, i.e., whether a strand exchange occurs between the attB site of the chromosome and the attSCC site of pSRatt in a fixed orientation, a crisscross PCR experiment was designed (Fig. 4). The cultures of transformants N315ex(pSRatt) and N315ex(pSRtta), with N315ex(pYT3att) as a control, were analyzed with PCR amplification using crisscross combinations of four primers: L (cL1) and R (cR2) flanking the attB region on the chromosome, and α (RV in pUC119) and β (in the ccrB gene) flanking the attSCC region on the plasmids (Fig. 4). The results showed that the plasmid pSRatt generated plasmid-chromosome junctions which were detectable only by the two combinations of the primers, L-β and R-α. On the other hand, the plasmid-chromosome junctions generated by pSRtta were detected only by the combinations of the primers, L-α and R-β. Integration in the opposite orientation could not be detected with the sensitivity of PCR employed in this study: the PCR sensitivity was such that it was able to detect the integrated template in N315ex(pSRatt) DNA diluted up to 104 times (wt/wt) with N315ex(pYT3att) DNA (data not shown).
The amplified DNAs from N315ex(pSRatt) and from N315ex(pSRtta) contained sequences which were identical with those of the chromosome-SCCmec junction regions (attL and attR) of N315 as presented in Fig. 1A.
PCR with any combinations of the primers did not amplify DNA fragments on template DNAs extracted from any of the transformants [N315ex(pYT3), N315ex(pSRA*att), N315ex(pSRB*att), or N315ex(pSR) (not shown)]. This showed that all of ccrA, ccrB, and attSCC were required for the integration of the experimental plasmids.
DISCUSSION
The size of SCCmec (52 kb) is comparable to those of other genetic elements such as bacteriophages, conjugative transposons, and pathogenicity islands, but SCCmec does not have any structural similarity to those previously identified genetic elements: it is distinguishable from conjugative transposons since it lacks the tra gene complex (5), and from bacteriophages because it lacks the structure genes (or their remnants) for head or tail proteins of bacteriophages (29). Unlike SCCmec pathogenicity islands (18, 19), does not contain any ORFs predictably encoding virulence factors as far as we could judge from the homology search of extant gene products (14). Besides these structural characteristics of SCCmec, we demonstrated, in this study, that SCCmec carries a set of unique recombinase genes, ccrA and ccrB, that are specifically involved in the recombination events (integration and excision) of SCCmec with the S. aureus chromosome. The presence of the two genes was required for both excision and integration processes of SCCmec. In other genetic elements, such as bacteriophage lambda and conjugative transposons, two site-specific recombinases, designated integrase and excisionase, are reported to be required for the excision process. However, in these elements, the integration can be mediated by the integrase alone (5, 29).
The Ccr recombinases are also characteristic in that their NH2-terminal thirds had a substantial homology to the recombinases of the invertase/resolvase family, whereas the integrases of bacteriophage lambda and conjugative transposons belong to the integrase family of site-specific recombinases. The characteristic serine residue that is considered to offer the hydroxyl group that attacks the DNA molecule in the strand exchange reaction is conserved among the recombinases of the invertase/resolvase family and in Ccr proteins as well, but Ccr proteins also differ considerably from the recombinases of the invertase/resolvase family in that they possess much larger COOH-terminal domains than those possessed by the recombinases belonging to this family. A homology search of the COOH-terminal domains did not find any homologue in the extant proteins. Therefore, the molecular evolutionary relationship of Ccr proteins to other recombinases and their mode of action pose intriguing questions to be explored.
Since the 1960s, the spontaneous loss of the mecA gene has been observed during the storage or long-term cultivation of MRSA strains in antibiotic-free medium (11). Deletion of a large chromosomal region is identified in such mecA deletion strains. The deletion starts precisely from the left boundary of IS431mec and extends leftwards for various distances beyond the mecA gene. The precise excision of SCCmec demonstrated in this study is unrelated to the transposase-mediated mecA deletions (14). Curiously, spontaneous precise excision does not occur appreciably during cultivation of N315 despite the fact that the strain carries an intact pair of ccr genes on its chromosome. The excision does, however, seem to occur at an extremely low frequency and can be detected by nested-PCR amplification of the attB sequence (14). Therefore, it is an intriguing question whether the expression of the intact copy of ccr genes of SCCmec is down regulated to stabilize the SCCmec in the chromosome. Alternatively, even an unrestricted expression of a single copy of the ccr genes on the chromosome may not be enough to increase the concentration of Ccr proteins to a level high enough to trigger the excision of SCCmec. A study is under way to clarify these points.
It is well known that the mecA gene is carried by many staphylococcal species besides S. aureus (13, 28), so it has been suspected that mecA is transmissible among staphylococcal species. In fact, methicillin resistance can be experimentally transferred between S. aureus cells by phage transduction (6). The DNA element with similar structural features to SCCmec is also identified in the strains of various staphylococcal species with the methicillin resistance phenotype (T. Ito, unpublished observation). Thus, it seems that the methicillin resistance gene mecA is transferred from cell to cell as a part of the SCCmec across staphylococcal species. However, as far as we are aware, no transducing phage capable of transferring genetic information across the staphylococcal species barrier has been described. Thus, it remains to be seen if SCCmec is transmissible by phage transduction or if some other genetic transfer system specific for the movement of SCCmec exists.
In conclusion, we have demonstrated that the methicillin resistance gene of MRSA is carried by a novel genetic element, SCCmec, whose integration into and excision from the S. aureus chromosome are mediated by a unique set of recombinase genes, ccrA and ccrB. Further study of the distribution of SCCmec and its related elements among S. aureus and other staphylococcal species will be helpful to elucidate how staphylococcal species exchange useful genetic information. Also, future elucidation of the mechanism of regulation of SCCmec excision may lead to the attractive possibility of the development of a novel therapeutic measure to aid in antibiotic chemotherapy against MRSA infection by converting MRSA strains in vivo into MSSA strains against which many extant antibiotics are effective.
ACKNOWLEDGMENTS
We thank K. Tsutsumimoto for her excellent technical assistance and Yasmin Abu Hanifah for valuable suggestions concerning the worldwide epidemiology of MRSA.
This work was supported by the Core University Program under the auspices of the Japan Society for the Promotion of Science (JSPS), coordinated by the Graduate School of Medicine, University of Tokyo, and School of Medical Sciences, Universiti Sains Malaysia, and by the Specially Designated Research Promotion of Monbusho, Japan.
REFERENCES
- 1.Ayliffe G A. The progressive intercontinental spread of methicillin-resistant Staphylococcus aureus. Clin Infect Dis. 1997;24(Suppl. 1):74–79. doi: 10.1093/clinids/24.supplement_1.s74. [DOI] [PubMed] [Google Scholar]
- 2.Barberis-Maino L, Berger-Bachi B, Weber H, Beck W D, Kayser F H. IS431, a staphylococcal insertion sequence-like element related to IS26 from Proteus vulgaris. Gene. 1987;59:107–113. doi: 10.1016/0378-1119(87)90271-x. [DOI] [PubMed] [Google Scholar]
- 3.Chambers H F, Neu H C. Penicillins. In: Mandell G, Bennett J E, Dolin R, editors. Principles and practice of infectious diseases. New York, N.Y: Churchill Livingstone; 1995. pp. 233–246. [Google Scholar]
- 4.Christiansen B, Brondsted L, Vogensen F K, Hammer K. A resolvase-like protein is required for the site-specific integration of the temperate lactococcal bacteriophage TP901-1. J Bacteriol. 1996;178:5164–5173. doi: 10.1128/jb.178.17.5164-5173.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Clewell D B. Conjugative transposons. In: de Bruijn F J, Lupski J R, Weinstock G M, editors. Bacterial genomes. New York, N.Y: Chapman and Hall; 1998. pp. 130–139. [Google Scholar]
- 6.Cohen S, Sweeney H M. Transduction of methicillin resistance in Staphylococcus aureus dependent on an unusual specificity of the recipient strain. J Bacteriol. 1970;104:1158–1167. doi: 10.1128/jb.104.3.1158-1167.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dubin D T, Matthews P R, Chikramane S G, Stewart P R. Physical mapping of the mec region of an American methicillin-resistant Staphylococcus aureus strain. Antimicrob Agents Chemother. 1991;35:1661–1665. doi: 10.1128/aac.35.8.1661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hanaki H, Kuwahara-Arai K, Boyle-Vavra S, Daum R S, Labischinski H, Hiramatsu K. Activated cell-wall synthesis is associated with vancomycin resistance in methicillin-resistant Staphylococcus aureus clinical strains Mu3 and Mu50. J Antimicrob Chemother. 1998;42:199–209. doi: 10.1093/jac/42.2.199. [DOI] [PubMed] [Google Scholar]
- 9.Hartman B J, Tomasz A. Low-affinity penicillin-binding protein associated with beta-lactam resistance in Staphylococcus aureus. J Bacteriol. 1984;158:513–516. doi: 10.1128/jb.158.2.513-516.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hiramatsu K, Asada K, Suzuki E, Okonogi K, Yokota T. Molecular cloning and nucleotide sequence determination of the regulator region of mecA gene in methicillin-resistant Staphylococcus aureus (MRSA) FEBS Lett. 1992;298:133–136. doi: 10.1016/0014-5793(92)80039-j. [DOI] [PubMed] [Google Scholar]
- 11.Hiramatsu K, Suzuki E, Takayama H, Katayama Y, Yokota T. Role of penicillinase plasmids in the stability of the mecA gene in methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 1990;34:600–604. doi: 10.1128/aac.34.4.600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hiramatsu K, Ito T, Hanaki H. Evolution of methicillin and glycopeptide resistance in Staphylococcus aureus. In: Finch R G, Williams R J, editors. Bailliere's clinical infectious disease. London, United Kingdom: Bailliere Tindall; 1999. pp. 221–242. [Google Scholar]
- 13.Hurlimann-Dalei R L, Ryffel C, Kayser F H, Berger-Bachi B. Survey of the methicillin resistance-associated genes mecA, mecR1-mecI, and femA-femB in clinical isolates of methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 1992;36:2617–2621. doi: 10.1128/aac.36.12.2617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ito T, Katayama Y, Hiramatsu K. Cloning and nucleotide sequence determination of the entire mec DNA of pre-methicillin-resistant Staphylococcus aureus N315. Antimicrob Agents Chemother. 1999;43:1449–1458. doi: 10.1128/aac.43.6.1449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jevons M P. “Celbenin”-resistant staphylococci. Br Med J. 1961;1:124–125. [Google Scholar]
- 16.Kreiswirth B, Kornblum J, Arbeit R D, Eisner W, Maslow J N, McGeer A, Low D E, Novick P R. Evidence for a clonal origin of methicillin resistance in Staphylococcus aureus. Science. 1993;259:227–230. doi: 10.1126/science.8093647. [DOI] [PubMed] [Google Scholar]
- 17.Kuwahara-Arai K, Kondo-Hori N, Tateda-Suzuki E, Hiramatsu K. Suppression of methicillin resistance in a mecA-containing pre-methicillin-resistant Staphylococcus aureus strain is caused by the mecI-mediated repression of PBP2′ production. Antimicrob Agents Chemother. 1996;40:2680–2685. doi: 10.1128/aac.40.12.2680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee C A. Pathogenicity islands and the evolution of bacterial pathogens. Infect Agents Dis. 1996;5:1–7. [PubMed] [Google Scholar]
- 19.Lindsay J A, Ruzin A, Ross H F, Kurepina N, Novick R P. The gene for toxic shock toxin is carried by a family of mobile pathogenicity islands in Staphylococcus aureus. Mol Microbiol. 1998;29:527–543. doi: 10.1046/j.1365-2958.1998.00947.x. [DOI] [PubMed] [Google Scholar]
- 20.Matsuhashi M, Song M D, Ishino F, Wachi M, Doi M, Inoue M, Ubukata K, Yamashita N, Konno M. Molecular cloning of the gene of a penicillin binding protein supposed to cause high resistance to beta-lactam antibiotics in Staphylococcus aureus. J Bacteriol. 1986;167:975–980. doi: 10.1128/jb.167.3.975-980.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Matthews P, Tomasz A. Insertional inactivation of the mec gene in a transposon mutant of a methicillin-resistant clinical isolate of Staphylococcus aureus. Antimicrob Agents Chemother. 1990;34:1777–1779. doi: 10.1128/aac.34.9.1777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.McKenzie H T, Hoshino T, Tanaka T, Sueoka N. The nucleotide sequence of pUB110: some salient features in relation to replication and its regulation. Plasmid. 1986;15:93–103. doi: 10.1016/0147-619x(86)90046-6. [DOI] [PubMed] [Google Scholar]
- 23.Murphy, Huwyler E L, Bastos M C. Transposon Tn554: complete nucleotide sequence and isolation of transposition-defective and antibiotic-sensitive mutants. EMBO J. 1985;4:3357–3365. doi: 10.1002/j.1460-2075.1985.tb04089.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Reynolds P E, Brown D F J. Penicillin-binding proteins of beta-lactam-resistant strains of Staphylococcus aureus. FEBS Lett. 1985;192:28–32. doi: 10.1016/0014-5793(85)80036-3. [DOI] [PubMed] [Google Scholar]
- 25.Sherratt D. Tn3 and related transposable elements: site-specific recombination and transposition. In: Berg D E, Howe M M, editors. Mobile DNA. Washington, D.C.: American Society for Microbiology; 1989. pp. 163–184. [Google Scholar]
- 26.Song M D, Wachi M, Doi M, Ishino F, Matsuhashi M. Evolution of an inducible penicillin-target protein in methicillin-resistant Staphylococcus aureus by gene fusion. FEBS Lett. 1987;221:167–171. doi: 10.1016/0014-5793(87)80373-3. [DOI] [PubMed] [Google Scholar]
- 27.Stewart P R, Dubin D T, Chikramane S G, Ingli B, Matthews P R, Poston S M. IS257 and small plasmid insertions in the mec region of the chromosome of Staphylococcus aureus. Plasmid. 1994;31:12–20. doi: 10.1006/plas.1994.1002. [DOI] [PubMed] [Google Scholar]
- 28.Suzuki E, Hiramatsu K, Yokota T. Survey of methicillin-resistant clinical strains of coagulase-negative staphylococci for mecA gene distribution. Antimicrob Agents Chemother. 1992;36:429–434. doi: 10.1128/aac.36.2.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Thompson J F, Landy A. Regulation of bacteriophage lambda site-specific recombination. In: Berg D E, Howe M M, editors. Mobile DNA. Washington, D.C.: American Society for Microbiology; 1989. pp. 1–22. [Google Scholar]
- 30.Utsui Y, Yokota T. Role of an altered penicillin-binding protein in methicillin- and cephem-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 1985;28:397–403. doi: 10.1128/aac.28.3.397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wada A, Katayama Y, Hiramatsu K, Yokota T. Southern hybridization analysis of the mecA deletion from methicillin-resistant Staphylococcus aureus. Biochem Biophys Res Commun. 1991;176:1319–1325. doi: 10.1016/0006-291x(91)90430-f. [DOI] [PubMed] [Google Scholar]
- 32.Yoshida T, Kondo N, Hanifah Y A, Hiramatsu K. Combined use of ribotyping, PFGE typing and IS431 typing the discrimination of nosocomical strains of methicillin-resistant Staphylococcus aureus. Microbiol Immunol. 1997;41:687–695. doi: 10.1111/j.1348-0421.1997.tb01912.x. [DOI] [PubMed] [Google Scholar]