ABSTRACT.
Despite commendable progress in control of malaria in India and other countries, there are hidden reservoirs of parasites in human hosts that continually feed malaria transmission. Submicroscopic infections are a significant proportion in low-endemic settings like India, and these infections possess transmission potential. Hence, these reservoirs of infection add to the existing roadblocks for malaria elimination. It is crucial that this submerged burden of malaria is detected and treated to curtail further transmission. The currently used diagnostic tools, including the so-called “gold standard” microscopy, are incapable of detecting these submicroscopic infections and thus are suboptimal. It is an opportune time to usher in more sensitive molecular tools like polymerase chain reaction (PCR) for routine diagnosis at all levels of healthcare as an additional diagnostic tool in routine settings. PCR assays have been developed into user-friendly formats for field diagnostics and are near-point-of-collection. Because of the COVID-19 pandemic in India, these are being used rampantly across the country. The facilities created for COVID-19 diagnosis can easily be co-opted and harnessed for malaria diagnosis to augment surveillance by the inclusion of molecular techniques like PCR in the routine national malaria control program.
INTRODUCTION
Considerable progress was achieved in bringing down the malaria cases in most countries from 2000 to 2015, during which global malaria case incidence declined by 27%. However, it is worrisome that the rate of decline has plateaued from 2015 to 2019 with a decline of less than 2%. At the same time, with 3% of the global malaria burden, the southeast Asia region achieved a reduction from 23 million cases in the year 2000 to about 6.3 million in 2019. This decline was reflected in India’s malaria burden, which reduced from ∼20 million cases in 2000 to ∼5.6 million in 2019. 1 India’s national malaria control program reported 0.33 million cases and 77 deaths in 2019. 2 Despite an impressive overall decline of malaria in India over the years, the gains are not uniform across the country. Several districts (the administrative unit for malaria control operations) have been consistently reporting a high number of cases over the years and despite effective and well-implemented interventions continue to do so. Possible reasons for these parasite reservoirs could be operational inadequacies, continuous transmission sustained by submicroscopic infections, asymptomatic infections that escape detection, efficient vectors with behavior change of early and outdoor biting, drug and insecticide resistance, the suboptimal performance of diagnostics and general neglect by communities in malarious regions. Migration and population movement from endemic to nonendemic areas and vice versa can be a formidable challenge for surveillance and hence the migrant populations may feed the transmission cycle. These roadblocks will slow the progress of malaria elimination program.
The heterogeneity of the country and multiplicity of vectors and parasites enhance the complexity of malaria epidemiology in India. Plasmodium falciparum (Pf) and Plasmodium vivax (Pv) are major parasite species. Gradually Pv is becoming a cause of concern as > 50% of malaria cases reported by the national malaria control program in 2019 were because of Pv, and India is shouldering 47% of the global Pv burden. It is acknowledged worldwide that between the two, Pv would be much more difficult to eliminate because of several reasons like hypnozoites that lie latent in the liver and the affected becomes a reservoir, protracted radical cure of 14 days leading to poor compliance and contraindication of primaquine in people with glucose-6-phosphate dehydrogenase (G6PD) deficiency disorder. It is known that all stages of Pv infection can develop in the asymptomatic state which can sustain transmission. Pv hypnozoite forms add to the complexity of dealing with asymptomatic infections with their ability to sequester in the liver. Both light microscopy and rapid diagnostic tests (RDTs) fail to identify hypnozoite carriers. These factors thus contribute to the continued transmission of P. vivax. Two sequential genetically identical infections could either be from a failure to clear the bloodstream completely of parasites (recrudescence) or activation of a hypnozoite in the liver (relapse). Relapses because of activation of hypnozoites make vivax malaria a self-propagating infection, rendering vector control tools and other preventive measures ineffective. Hence, it is more important that each infected individual is identified and treated, thus curtailing the transmission. Strategies and tools for malaria elimination in India must be tailored-made keeping in view the endemicity at the state, district, and maybe sub-district level, given the size, population, and heterogeneity of malaria within a district. To develop and deploy interventions targeted at elimination and eventually eradication, it is essential to understand the true burden of malaria. In addition to patent and clinical malaria, which is routinely detected microscopically and by rapid diagnostics, malaria burden also includes asymptomatic (infection in people without overt symptoms and thus they escape detection and management), or infection below the detection limits of conventional microscopy and rapid diagnostic tests, that is, submicroscopic infections.
In addition, there are significant limitations of microscopy and rapid diagnostics. Diagnostics are a principal component of malaria surveillance which is considered as an intervention strategy. It is essential that all cases of malaria, be it asymptomatic, submicroscopic infections, or mixed species, are detected and treated not only for the individual benefit but for public health good. Even in diagnostics, elimination and eventual eradication of malaria will need additional tools and much more than the “business as usual” approach. Recent literature has revealed a significant burden of submicroscopic infections lingering undetected in the communities by routine diagnostics. 3 Studies have also indicated how vital submicroscopic infections are in the sustenance of transmission of malaria. 3 – 5
LIMITATIONS OF CURRENT DIAGNOSTIC METHODS
In 2010, WHO mandated parasitological confirmation (through microscopy or rapid diagnostic test) of malaria infection before initiating artemisinin combination therapy (ACT), also known as the “test-and-treat” strategy. Following WHO guidelines, the current Indian drug policy also demands that all cases of suspected malaria are to be tested and only those positive are administered anti-malarial treatment. 6 The two diagnostic methods approved by WHO, light microscopy and rapid diagnostics, are based on the premise of detecting patent infections. However, these diagnostic methods miss and sometimes misdiagnose submicroscopic and mixed parasite infections.
Diagnosis by routine microscopy has its limitations as a technique and additionally because of human factors like lack of expertise. The limit of detection may substantially vary depending on the experience and training of microscopists, with a range of 5–100 parasites/μL. 7 Also, microscopy is not best suited to differentiate between various non-Pf and non-Pv Plasmodium species (Plasmodium malariae, Plasmodium ovale, and Plasmodium knowlesi). Microscopy also misses detection of gametocytes present in low densities. In light of the above, microscopy though the current gold standard method thus suffers from several constraints. In addition, there are human factors that may cause errors in assessing the slides like lack of skill and fatigue. 8 The use of artificial intelligence to make microscopy more efficient may be harnessed to compensate for the dependence on a skilled workforce, but its use is still in infancy, even for the detection of Pf and Pv. 9, 10
The basic principle of diagnosis by RDTs is the identification of antigens, such as aldolase, lactate dehydrogenase (LDH), and histidine-rich protein-2 (HRP2). 11, 12 Of the three antigens, HRP2 specifically detects Pf in the blood-stage infection because it is expressed by Pf in both asexual and sexual stages. 13 – 15 Pf and Pv both produce LDH and aldolase, so these antigens are used for the diagnosis of both species. A growing body of evidence suggests that deletion of HRP2 and HRP3 genes in Pf impacts the performance of HRP-2-based RDTs and may result in a higher rate of false-negative results. Further, when compared with Pf, the lower density of parasitemia and lower pyrogenic threshold in Pv infections pose further challenges for the accurate diagnosis as microscopy and RDTs are not sensitive enough to detect the low parasitemia. 16, 17 According to reports, in the Indian states of Maharashtra, Madhya Pradesh, Jharkhand, Gujarat, and Odisha, the prevalence of Pf with HRP2 and HRP3 genes deletions ranges from 0% to 8%. 18, 19 Another study in Odisha, India, carried out from 2013 to 2016 in 25 districts, showed a 15% prevalence of HRP2 deletions. 20
National malaria control program relies on the above two methods and hence misses the true and submerged burden of malaria. India is experiencing a sustained decline in malaria burden and has API less than one since 2012, making India a low-transmission setting. Even at a more granular level, even though some districts may have higher API, none of them qualifies for being moderate or high-transmission areas as per WHO norms. 21 Therefore, it is important that countries like India, which are on an elimination pathway with a declining burden, invest in strengthening capacities in molecular tools for detecting submicroscopic infections, thus clearing the hidden reservoirs of infection from the community. In addition, these submicroscopic/asymptomatic infections are responsible for significant chronic anemia, maternal and neonatal mortality, cognitive impairment, and huge societal burden. 22 At the same time, several African countries fall under high-transmission settings as they have a high burden of patent infections with high parasitemia. And the healthcare settings in these countries are not up to the mark and hence routine interventions are deployed suboptimally. In these regions, before establishing molecular diagnostic capacities for program purposes, it is crucial to strengthen the routine surveillance and diagnostic structures through RDTs and microscopy in the first instance. 23 Since India is geared up for elimination by 2030, all programmatic activities, strategies, tools, and policies are directed at that goal. It is imperative that we revisit the strengths and weaknesses of the currently available diagnostic methods and assess the need for molecular techniques such as polymerase chain reaction (PCR), loop-mediated isothermal amplification (LAMP), and others to be included as an additional tool in the routine diagnosis for malaria. We base our arguments on PCR as an example of a molecular tool, which can be considered by the national malaria control program to be included as an additional diagnostic tool in India and in other malaria-endemic countries keeping malaria elimination in view. Table 1 compares the features and limitations of light microscopy, RDT, and PCR for malaria diagnostics. 24 – 26
Table 1.
Comparison between microscopy, PCR, and RDT
| Key features | Microscopy | PCR | Rapid diagnostic test | References |
|---|---|---|---|---|
| Level of detection (parasites/μL) | Skilled microscopist: < 10/μL Routine microscopy: ∼100/μL | 0.5–5/μL | 50–100/μL | 20, 21 |
| Sensitivity | 95% | 98–100% | 85–94.8%* | 20, 21 |
| Specificity | 98% | 88–94% | ∼95.2–99% | 20, 21 |
| Time | 60 min. | Standard > 6 hours | 20 min. | 21 |
| Requirements | Trained laboratory staff, microscope, electricity, slides, Giemsa stain | Trained laboratory staff, thermocycler, DNA extraction kit, cold chain, electricity, reagents | Immunochromatographic strip and appropriate storage conditions | 20, 21 |
| Cost per test | $0.20–$0.40 | $1–$5 | $0.85–$5.0 | 21 |
| Species detected | Majorly P. falciparum, P. vivax Difficult to detect mixed infection | All human malaria parasites Detects mixed infection | P. falciparum, P. vivax Cannot detect mixed infection | 19 |
| Type of detection | Parasite morphology, speciation and stages | Parasite nucleic acid 18S rRNA or others | Parasite specific antigen; pHRP-2 (specific for P. falciparum), LDH, aldolase (not specific) | 20, 21 |
| Sample type | Blood | Blood | Blood | 20 |
| Use | Used as gold standard in national programs for confirmation of diagnosis and clinical and research studies | Epidemiological studies and surveys | As a point-of-care test for diagnosis of malaria | 20 |
HRP = histidine-rich protein; LDH = lactate dehydrogenase; PCR = polymerase chain reaction.
The limit of sensitivity for RDT varies on the distribution of parasite density or HRP2 antigen in a given setting.
BURDEN AND TRANSMISSION POTENTIAL OF SUBMICROSCOPIC MALARIA INFECTIONS
Malarious countries typically show the presence of asymptomatic and submicroscopic malaria. Data from South American countries, like Brazil, Peru, Venezuela, Columbia, and South Asian countries, like Thailand, Myanmar, and Cambodia, have shown a high burden of asymptomatic malaria and malaria with low-density parasitemia not detected by microscopy. 27 Similarly, malarious regions in India have suggested a burden as high as 70–80% of asymptomatic infections and 30–45% as sub-patent. 28 Further, the submicroscopic infections have also been reported in moderate-to-high transmission regions with ∼20% proportion, whereas 70–80% in low-transmission settings. 29 Thus, the contribution of submicroscopic infections and asymptomatic infections should not be underestimated, and particular attention has to be diverted toward them in the current phase of malaria elimination. Table 2 depicts the proportion of submicroscopic malaria cases detected additionally by PCR in India and other countries.
Table 2.
Studies showing parasite positivity by microscopy and PCR
| S.N. | Year | Type of study | Sample | Location | Parasite positivity (%) | Microscopy (%) | PCR (%) | References |
|---|---|---|---|---|---|---|---|---|
| 1. | 2007–2008 | Clinic | 3,425 | Chhattisgarh, India | 1.3 | 3.4 | 68 | |
| 2. | 2009–2010 | Clinic | 281 | Karnataka, India | 59.7 | 49.8 | 9.9 | 25 |
| 3. | 2012 | Community | 3,501 | Central Islands Province, Solomon Islands | 13.4 | 3.7 | 9.7 | 69 |
| 4. | 2012 | Community | 963 | West Bengal, India | 8.4 | 7.8 | 0.6 | 70 |
| 5. | 2012–2014 | Community | 16,650 | Blantyre, Southern Malawi | 18.5 | 9.4 | 9.1 | 71 |
| 6. | 2012–2015 | Multi center, Community | 928 | Tamil Nadu, India | 2.5 | 0.7 | 1.8 | 43 |
| 796 | Gujarat, India | 7.5 | 7.3 | 0.2 | ||||
| 1,307 | Odisha, India | 8.3 | 5.7 | 2.6 | ||||
| Multi-center, Clinic | 1,054 | Tamil Nadu, India | 21.1 | 17.1 | 4.0 | |||
| 685 | Gujarat, India | 9.6 | 9.5 | 0.1 | ||||
| 1,875 | Odisha, India | 5.0 | 2.8 | 2.2 | ||||
| 7. | 2014–2015 | Community | 1,000 | Battambang Province, western Cambodia | 9.1 | 0.9 | 8.2 | 72 |
| 8. | 2015 | Community | 3,650 | Tak Province, northwestern Thailand | 2.60 | 0.27 | 2.33 | 73 |
| 9. | 2016–2017 | Community | 848 | Greater Accra region, Ghana | 11.67 | 5.42 | 6.25 | 74 |
| 10. | 2016–2020 | Community | 860 | Ethiopia | 21.2 | 12.2 | 9 | 75 |
| 11. | 2017 | Community | 2,236 | Chhattisgarh, India | 28.6 | 26.3 | 2.3 | 76 |
| 12. | 2017–2018 | Community | 1,114 | Punjab, India | 5.75 | 4.4 | 1.35 | 77 |
| 13. | 2017–2018 | Community | 422 | Odisha, India | 76.5 | 64.4 | 12 | 78 |
Transmission potential of asymptomatic and symptomatic Pf cases have been explored, and studies point out that such infections are transmissible. Molecular diagnostics have shown that the presence of gametocytes in low densities is widespread in malaria-endemic populations and these possess transmission potential. 30 In a study carried out in Papua New Guinea, the impact of malaria control operations (management and vector control) over a period of time was studied. Over 90% of gametocyte carriers were found to be asymptomatic, and a higher proportion of sub-patent infections made detection of gametocytes more difficult. 31 Transmission of Pf infection to mosquito from symptomatic and asymptomatic individuals was studied in a 14-month cohort study in Western Kenya. Unique parasite haplotypes were observed in human and mosquito infections. The odds of successful transmission of Pf from asymptomatic infections were more than double as compared with symptomatic cases. 5 In a longitudinal study in children aged 5–10 years in Burkina Faso, it was observed that Pf gametocyte infectivity was higher in chronic asymptomatic infections than in incident symptomatic infections, which required treatment before the gametocyte density reached sufficient density to infect mosquitoes. 4 Similarly, in low-transmission seasons in Chhattisgarh, a malaria-endemic state in India, a substantial number of asymptomatic (∼78%) and submicroscopic (∼36%) malaria infections were identified. 32 Similar results from low-transmission settings like South American countries and Vietnam also report largely asymptomatic infections in areas of low-seasonal malaria transmission. 33 – 35
The presence of mature gametocytes in the peripheral blood is required for malaria parasite transmission from the human host to the mosquito vector. Mosquitoes can be infected when feeding on people with submicroscopic gametocyte densities of about 4 gametocytes/μL, demonstrating that parasite transfer to the vector is a remarkably efficient mechanism. 36 In a study in Tanzania, the prevalence of gametocytes detected by pfs25 qRT-PCR was 10 times higher than that detected by light microscopy, indicating a large proportion of submicroscopic gametocytemia in the community. 37 It has been observed that individuals with negative peripheral smear for malaria parasite are infective to mosquitoes. 38, 39 In another paper, it was observed that gametocyte prevalence as low as 10.6% could sustain malaria in the presence of vectors. 40 Thus, detection of gametocytemia is vital in going forward in malaria elimination.
SIGNIFICANCE OF SUBMICROSCOPIC INFECTIONS
Aside from recent studies that have indicated the value of tracking low-density infections for stemming transmission of malaria using PCR-based tools of detection, compelling epidemiological reasons are pointing at the significance of submicroscopic infections:
-
1)
Adults have a higher frequency of submicroscopic infections than children. 29, 41 This is attributed to increased levels of immunity in adults and associated suppression of parasite densities by this immune response. In places where transmission intensity is low, the missed proportion of submicroscopic reservoir is usually higher. 3, 42 Also, people with repeated episodes of malaria have a lower density of parasites compared with less-exposed within the same area. 30, 43
-
2)
It has been observed that where successful malaria control operations have brought down the transmission intensity from high to low, submicroscopic infections become more common. This is because of the presence of pockets of hotspots where some communities continue to be exposed to malaria and thus rapidly acquire immunity, contributing to the disproportionately high submicroscopic parasite carriage. 44 Another cause for submicroscopic infections could be a decrease in genetic diversity under reduced transmission settings, as well as high levels of immunity against Plasmodium strains. 45
-
3)
If left untreated, the symptomatic and microscopically detected Pf infections may enter into a chronic phase which can last for months at a submicroscopic level. And in some situations, soon after treatment, residual asexual parasites and/or gametocytes persist for several weeks at sub-patent levels. In other cases, there is a period of low-level infection that may last > 3 weeks before the phase of patent parasitemia. Parasitemia in this phase is infective to others and is detectable by PCR but not by microscopy. 46
-
4)
In another scenario, parasitemia may never reach a patent level and remain sub-microscopic, which escapes detection by routine methods. A better understanding of these events is needed in low-transmission settings. 47
-
5)
Gametocytes make < 5% of parasite biomass in Pf malaria, which are routinely difficult to detect by microscopy. Individuals with submicroscopic infections frequently carry gametocytes and can contribute to the onward transmission of malaria. Individuals with submicroscopic infections have been shown to contribute to transmission across areas of perennial 39 and seasonal 48 settings as well as low 49 and high 50 transmission intensity, underscoring the potential relevance of this infection subgroup to malaria control efforts. 3 Hence, molecular tools can be used for the detection of gametocytes to understand the transmission potential of prevailing malaria infection in research mode and as specific surveillance activity.
-
6)
Identification of malaria in pregnancy is a challenging task as peripheral blood harbors a low level of parasitemia and Pf parasites may be sequestered in the placenta. Undiagnosed malaria in pregnancy and hence untreated has grave consequences for both mother and her newborn in the form of maternal anemia, prematurity, low birth weight, and neonatal mortality. 51
-
7)
Microscopy misses the mixed-species infection mainly because one of the parasite species is predominantly present. A study has revealed a substantial contribution of non Pf/Pv malaria parasite species to the overall burden of malaria. 8
-
8)
Population movement from/to malaria-endemic area to nonendemic area exposes the resident/traveler population to the risk of malaria. The immunologically naïve population can present as febrile cases at a low threshold of parasitemia. 52 Hence, they may be missed as malaria cases and are likely to be treated for other causes of febrile illness.
The above pointers indicate the significance of submicroscopic infections and the pressing need to clear the carriers of low-density infections to curtail further transmission of malaria.
NEED FOR PCR-BASED TOOLS
Polymerase chain reaction-based diagnostics have been used so far in research studies and epidemiological surveys only, but it is the opportune time in the backdrop of malaria elimination to consider the inclusion of PCR-like molecular tools in the diagnostic arsenal. It is very crucial at this juncture that more sensitive molecular tools are considered, not only for research studies but as an integral part of available diagnostics and for surveillance. 53
It is time to adopt and operationalize molecular tools like PCR in national malaria control program as an additional diagnostic tool in the following scenarios: 1) confirmatory diagnosis wherever feasible, 2) mass surveys in selected hotspot areas of persistent malaria to clear the reservoirs of infection, 54, 55 3) as a diagnostic for reactive active case detection as an elimination strategy, and 4) areas that receive travellers (like tourist places) and migrant population.
The PCR-based molecular devices being developed nowadays offer several advantages. These are 1) detection of low densities of malaria parasite of all species and their gametocytes, 56, 57 2) diagnosis of placental malaria in pregnancy during delivery and detection of low densities in peripheral parasitaemia, 58, 59 3) they are portable and handy, 4) they are battery operated and chargeable, thus obviating the need of continuous power supply, 5) they are digital devices, hence real-time data transfer is possible with the accessibility of performing offline in the absence or erratic Internet availability, 6) they are simple to operate, minimal training required, 7) they give quick results, 8) they show good throughput, 9) they are affordable—in the range of $1–5 per test, and 10) multiple Plasmodium species can be screened and diagnosed making the devices cost-effective.
Polymerase chain reaction-based tools have been always demanding, especially for resource-constraint settings, in terms of cost, requirement of infrastructure, equipment, and training, thus curtailing their use to mostly research studies mostly. But recognizing the potential and urgent need of these tools to be used in field settings, there have been attempts to develop these molecular tools as field-friendly, near point-of-care (PoC) tests for resource-limited countries/regions. Molecular tools like cartridge-based nucleic acid amplification test (CBNAAT) and Truenat are used for detection and confirmation of TB in India’s national program of tuberculosis. 60 These PCR-based diagnostics have paid rich dividends in improving detection rates of TB in India especially for extrapulmonary TB. 60 Similarly, for malaria, one such effort—TruenatTM (Bigtec Labs, Bangalore, India) Malaria Pv/Pf is a handheld, battery–operated, and chip-based real-time PCR test for detection of Pv and Pf. This technique has undergone laboratory validation and has shown promising early results on the parameters of sensitivity, specificity, and detection of mixed infection. 61
Further, simple and field-amenable techniques like LAMP that utilizes isothermal conditions for Plasmodium DNA amplification obviates the need for sophisticated and expensive thermal cyclers. 62 Over 40 different LAMP methods have been developed for detecting Plasmodium species with high sensitivity and specificity in comparison to microscopy, PCR, and RDTs in both endemic and nonendemic settings. 63 The sensitivity and specificity of LAMP has largely remained greater than 95% demonstrating that malaria LAMP has excellent diagnostic performance. 63 Similar to LAMP, other techniques like nested, quantitative (q), or real-time reverse transcription (RT-PCR), quantitative nucleic acid sequence-based amplification (QT-NASBA), and all key nucleic acid amplification tests (NAATs) have multiple advantages over microscopy and RDTs.
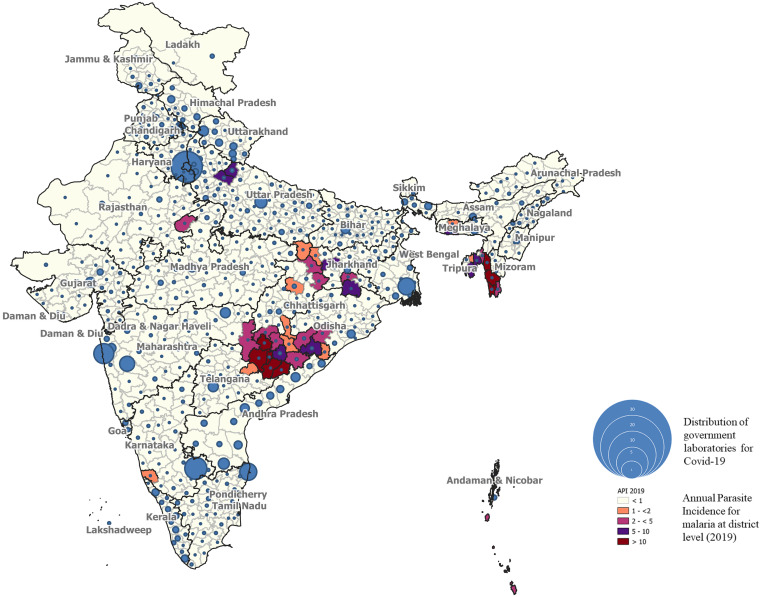
It is noteworthy to mention here that molecular tools such as RT-PCR, TruenatTM, and CBNAAT have been the major diagnostic tools in India’s fight against COVID-19 pandemic and these are widely deployed in diagnosis of the viral infection. Currently, 2,536 laboratories in India are performing these molecular tests for SARS-CoV-2 on a regular basis, and out of which 917 are using Truenat technology (Figure 1). The laboratories are present at the district level and have been upgraded with the necessary infrastructure, including trained manpower. These laboratories can be easily co-opted for malaria diagnosis utilizing these PCR machines in the malaria-endemic regions (Figure 1). 64 Furthermore, by making quick diagnostics available for free, more patients/healthcare workers will be able to choose confirmatory diagnostics, bringing malaria elimination one step closer. 65 For record-keeping purpose of the type of diagnostic techniques used, the “malaria-card” can be provided to the patients while making attempts to track by digital surveillance. 66, 67 Even if not all laboratories at the remote sites opt for PCR-based malaria diagnosis, the utilization of PCR for the confirmatory diagnosis could be implemented at least at the district level, which can later be taken forward to the block level in a phased manner. This will pave the way for the use of molecular tools like PCR for the diagnosis of sub-patent malaria and also for detection of non-Pf/Pv parasitemia. For adding PCR to the routine diagnosis along with the “gold standard” for malaria diagnosis, PCR can be deployed at the health centres where logistics, trainings, and technical cadre of people are available and can be reoriented to perform these molecular tests. Once these become fully field deployable the so-called limitations of using molecular techniques as routine diagnosis, such as a lack of skilled employees.
Figure 1.
Map of India depicting malaria incidence as per its annual parasite India (API) and the number of COVID-19 testing laboratories that can be used for malaria testing. This figure appears in color at www.ajtmh.org.
CONCLUSION
Challenges, such as infrastructure, equipment, and trained manpower will continue to exist and it would take considerable time and resources to expand the diagnostic network with availability of molecular tools including portable battery-operated PCR machines to percolate for malaria parasite detection. WHO approval of the first malaria vaccine RTS,S/AS01 has brought a lot of hope as it will reduce severity and mortality among young children in high to moderate transmission settings, though it may not be suitable for immediate deployment in India. 79 It is important to strengthen surveillance with detection of each and every case of malaria in the community, be it asymptomatic or case with low-density parasitemia in view of their continued contributions toward malaria transmission. It is exigent to expand the diagnostic arsenal and include PCR-based molecular tools not only for research but also as routine diagnostic test available under national malaria control program. For COVID-19 diagnosis, molecular tools are needed and thus appropriate laboratory infrastructure were established across countries including India. The staff were trained to use molecular tools such as PCR, Truenat, and others. The cost per test using these tools has also been rationalized with time. Moreover, COVID-19 pandemic has brought into forefront the significance of sub-clinical and asymptomatic infections in the transmission cycle. Hence, this has sensitized not only the scientific community but also program managers to take into cognizance the sub-merged infections in the community. The upgraded infrastructure and trained manpower can be leveraged for the detection of malaria at district and sub-district level using molecular methods. It is also the opportune time to contemplate on the traditional “gold standard” of malaria diagnosis, that is, microscopy, given its numerous limitations and drawbacks. The molecular tools are more accurate and far more sensitive to detect submerged burden of malaria. We will be turning a blind eye to the concealed case load of submicroscopic malaria infections if we detect the overtly presented cases via RDTs and microscopy only and that would be detrimental to our malaria elimination plans.
ACKNOWLEDGMENTS
The American Society of Tropical Medicine and Hygiene (ASTMH) assisted with publication expenses.
References
- 1. World Health Organization , 2020. World Malaria Report 2020. Geneva, Switzerland: WHO. [Google Scholar]
- 2. NVBDCP , 2019. National Vector Borne Disease Control Programme. Available at: https://nvbdcp.gov.in/index1.php?lang=1&level=1&sublinkid=5784&lid=3689. Accessed 2019.
- 3. Whittaker C Slater H Nash R Bousema T Drakeley C Ghani AC Okell LC , 2021. Global patterns of submicroscopic Plasmodium falciparum malaria infection: insights from a systematic review and meta-analysis of population surveys. Lancet Microbe 2: e366–e374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Barry A Bradley J Stone W Guelbeogo MW Lanke K Ouedraogo A Soulama I , 2021. Higher gametocyte production and mosquito infectivity in chronic compared to incident Plasmodium falciparum infections. Nat Commun 12: 2443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Sumner KM Freedman E Abel L Obala A Pence BW Wesolowski A Meshnick SR Prudhomme-O’Meara W Taylor SM , 2021. Genotyping cognate Plasmodium falciparum in humans and mosquitoes to estimate onward transmission of asymptomatic infections. Nat Commun 12: 909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Anon , 2013. National Drug Policy on Malaria 2013. India: Directorate of National Vector Borne Disease Control Programme, Directorate General of Health Services, Ministry of Health and Family Welfare, Government of India. [Google Scholar]
- 7. Zimmerman PA Howes RE , 2015. Malaria diagnosis for malaria elimination. Curr Opin Infect Dis 28: 446–454. [DOI] [PubMed] [Google Scholar]
- 8. Chaturvedi R Deora N Bhandari D Parvez S Sinha A Sharma A , 2021. Trends of neglected Plasmodium species infection in humans over the past century in India. One Health 11: 100190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Park HS Rinehart MT Walzer KA Chi J-TA Wax A , 2016. Automated detection of P. falciparum using machine learning algorithms with quantitative phase images of unstained cells. PLoS One 11: e0163045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Linder N Turkki R Wallliander M Martensson A Diwan V Rahtu E Pietikainen M Lundin M Lundin J , 2014. A malaria diagnostic tool based on computer vision screening and visualization of Plasmodium falciparum candidate areas in digitized blood smears. PLoS One 9: e104855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Moody AH Chiodini PL , 2002. Non-microscopic method for malaria diagnosis using OptiMAL IT, a second-generation dipstick for malaria pLDH antigen detection. Br J Biomed Sci 59: 228–231. [DOI] [PubMed] [Google Scholar]
- 12. Ndao M Bandyayera E Kokoskin E Gyorkos TW MacLean JD , 2004. Comparison of blood smear, antigen detection, and nested-PCR methods for screening refugees from regions where malaria is endemic after a malaria outbreak in Quebec, Canada. J Clin Microbiol 42: 2694–2700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Hayward RE Sullivan DJ Day KP , 2000. Plasmodium falciparum: histidine-rich protein II is expressed during gametocyte development. Exp Parasitol 96: 139–146. [DOI] [PubMed] [Google Scholar]
- 14. Rock EP Marsh K Saul AJ Wellems TE Taylor DW Maloy WL Howard RJ , 1987. Comparative analysis of the Plasmodium falciparum histidine-rich proteins HRP-I, HRP-II and HRP-III in malaria parasites of diverse origin. Parasitology 95: 209–227. [DOI] [PubMed] [Google Scholar]
- 15. Howard RJ Uni S Aikawa M Aley SB Leech JH Lew AM Wellems TE Rener J Taylor DW , 1986. Secretion of a malarial histidine-rich protein (Pf HRP II) from Plasmodium falciparum-infected erythrocytes. Cell Biol (Henderson NV) 103: 11269–11277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. McMorrow ML Aidoo M Kachur SP , 2011. Malaria rapid diagnostic tests in elimination settings—can they find the last parasite? Clin Microbiol Infect 17: 1624–1631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Price RN Tjitra E Guerra CA Yeung S White NJ Anstey NM , 2007. Vivax malaria: neglected and not benign. Am J Trop Med Hyg 77: 79–87. [PMC free article] [PubMed] [Google Scholar]
- 18. Verma AK Bharti PK Das A , 2018. HRP-2 deletion: a hole in the ship of malaria elimination. Lancet Infect Dis 18: 826–827. [DOI] [PubMed] [Google Scholar]
- 19. Kojom LP Singh V , 2020. Prevalence of Plasmodium falciparum field isolates with deletions in histidine-rich protein 2 and 3 genes in context with sub-Saharan Africa and India: a systematic review and meta-analysis. Malar J 19: 46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Pati P Dhangadamajhi G Bal M Ranjit M , 2018. High proportions of pfhrp2 gene deletion and performance of HRP2-based rapid diagnostic test in Plasmodium falciparum field isolates of Odisha. Malar J 17: 394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. World Health Organization , 2018. Malaria Surveillance, Monitoring and Evaluation: A Reference Manual. Geneva, Switzerland: WHO. [Google Scholar]
- 22. Hofmann N Mwingira F Shekalaghe S Robinson LJ Mueller I Felger I , 2015. Ultra-sensitive detection of Plasmodium falciparum by amplification of multi-copy subtelomeric targets. PLoS Med 12: e1001788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Jacobson JO Cueto C Smith JL Hwang J Gosling R Bennett A , 2017. Surveillance and response for high-risk populations: what can malaria elimination programmes learn from the experience of HIV? Malar J 16: 33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Han TZ Han KT Aye KH Hlaing T Thant KZ Vythilingam I , 2017. Comparison of microscopy and PCR for the detection of human Plasmodium species and Plasmodium knowlesi in southern Myanmar. Asian Pac J Trop Biomed 7: 680–685. [Google Scholar]
- 25. Mbanefo A Kumar N , 2020. Evaluation of malaria diagnostic methods as a key for successful control and elimination programs. Trop Med Infect Dis 5: 102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. World health Organization , 2021. WHO Guidelines for Malaria. Geneva, Switzerland: WHO. [Google Scholar]
- 27. Recht J Siqueira AM Monteiro WM Herrera S Herrera S Lacerda MVG , 2017. Malaria in Brazil, Colombia, Peru and Venezuela: current challenges in malaria control and elimination. Malar J 16: 273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Ranjha R Sharma A , 2021. Forest malaria: the prevailing obstacle for malaria control and elimination in India. BMJ Glob Health 6: e005391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Okell LC Bousema T Griffin JT Ouédraogo AL Ghani AC Drakeley CJ , 2012. Factors determining the occurrence of submicroscopic malaria infections and their relevance for control. Nat Commun 3: 1237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Slater H et al. 2019. The temporal dynamics and infectiousness of subpatent Plasmodium falciparum infections in relation to parasite density. Nat Commun 10: 1433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Koepfli C et al. 2017. Sustained malaria control over an 8-year period in Papua New Guinea: the challenge of low-density asymptomatic Plasmodium infections. J Infect Dis 216: 1434–1443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Chourasia MK et al. 2017. Additional burden of asymptomatic and sub-patent malaria infections during low transmission season in forested tribal villages in Chhattisgarh, India. Malar J 16: 320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Nguyen T-N et al. 2018. The persistence and oscillations of submicroscopic Plasmodium falciparum and Plasmodium vivax infections over time in Vietnam: an open cohort study. Lancet Infect Dis 18: 565–572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Ferreira IM Yokoo EM Souza-Santos R Galvão ND Atanaka-Santos M , 2012. Factors associated with the incidence of malaria in settlement areas in the district of Juruena, Mato Grosso state, Brazil. Cien Saude Colet 17: 2415–2424. [DOI] [PubMed] [Google Scholar]
- 35. da Silva-Nunes M Moreno M Conn JE Gamboa D Abeles S Vinetz JM Ferreira MU , 2012. Amazonian malaria: Asymptomatic human reservoirs, diagnostic challenges, environmentally driven changes in mosquito vector populations, and the mandate for sustainable control strategies. Acta Trop 121: 281–291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Churcher TS Bousema T Walker M Drakeley CJ Schneider P Ouedraogo AL Basáñez M-G , 2013. Predicting mosquito infection from Plasmodium falciparum gametocyte density and estimating the reservoir of infection. eLife 2: e00626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Shekalaghe SA Bousema T Kunei KK Lushino P Masokoto A Wolters LR Mwakalinga S Mosha FW Sauerwein RW Drakeley CJ , 2007. Submicroscopic Plasmodium falciparum gametocyte carriage is common in an area of low and seasonal transmission in Tanzania. Trop Med Int Health 12: 547–553. [DOI] [PubMed] [Google Scholar]
- 38. Coleman RE Kumpitak C Ponlawat A Maneechai N Phunkitchar V Rachapaew N Zollner G Sattabongkot J , 2004. Infectivity of asymptomatic Plasmodium-infected human populations to Anopheles virus mosquitoes in western Thailand. J Med Entomol 41: 201–208. [DOI] [PubMed] [Google Scholar]
- 39. Schneider P Bousema T Gouagna LC Otieno S van de Vegte-Bolmer M Omar SA Sauerwein RW , 2007. Submicroscopic Plasmodium falciparum gametocyte densities frequently result in mosquito infection. Am J Trop Med Hyg 76: 470–474. [PubMed] [Google Scholar]
- 40. Mwingira F Genton B Kabanywanyi A-NM Felger I , 2014. Comparison of detection methods to estimate asexual Plasmodium falciparum parasite prevalence and gametocyte carriage in a community survey in Tanzania. Malar J 13: 433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Tran TM et al. 2013. An intensive longitudinal cohort study of malian children and adults reveals no evidence of acquired immunity to Plasmodium falciparum infection. Clin Infect Dis 57: 40–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Rodriguez-Barraquer I et al. 2018. Quantification of anti-parasite and anti-disease immunity to malaria as a function of age and exposure. eLife 7: e38532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Eijk AV Sutton PL Ramanathapuram L Sullivan SA Kanagaraj D Priya GL , 2019. The burden of submicroscopic and asymptomatic malaria in India revealed from epidemiology studies at three varied transmission sites in India. Sci Rep 9: 17095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Robortella DR et al. 2020. Prospective assessment of malaria infection in a semi-isolated Amazonian indigenous Yanomami community: transmission heterogeneity and predominance of submicroscopic infection. PLoS One 15: e0230643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Björkman A Morris U , 2020. Why asymptomatic Plasmodium falciparum infections are common in low-transmission settings. Trends Parasitol 36: 898–905. [DOI] [PubMed] [Google Scholar]
- 46. Stresman G Kobayashi T Kamangi A Thuma PE Mharakurwa S Moss WJ Shiff C , 2012. Malaria research challenges in low prevalence settings. Malar J 11: 353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Bousema T Okell LC Felger I Drakeley CJ , 2014. Asymptomatic malaria infections: detectability, transmissibility and public health relevance. Nat Rev Microbiol 12: 833–840. [DOI] [PubMed] [Google Scholar]
- 48. Ouédraogo AL et al. 2009. Substantial contribution of submicroscopical Plasmodium falciparum gametocyte carriage to the infectious reservoir in an area of seasonal transmission. PLoS One 4: e8410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Taddesse FG et al. 2018. The relative contribution of symptomatic and asymptomatic Plasmodium vivax and Plasmodium falciparum infections to the infectious reservoir in a low-endemic setting in Ethiopia. Clin Infect Dis 66: 1883–1891. [DOI] [PubMed] [Google Scholar]
- 50. Ouédraogo AL et al. 2016. Dynamics of the human infectious reservoir for malaria determined by mosquito feeding assays and ultrasensitive malaria diagnosis in Burkina Faso. J Infect Dis 213: 90–99. [DOI] [PubMed] [Google Scholar]
- 51. Pandey M Rahi M Sharma A , 2021. The Indian burden of malaria in pregnancy needs assessment. Med 2: 464–469. [DOI] [PubMed] [Google Scholar]
- 52. Gatton ML Cheng Q , 2002. Evaluation of the pyrogenic threshold for Plasmodium falciparum malaria in naive individuals. Am J Trop Med Hyg 66: 467–473. [DOI] [PubMed] [Google Scholar]
- 53. Hemingway J, Shretta R, Wells TN, Bell D, Djimde AA, Achee N, Qi G, 2016. Tools and strategies for malaria control and elimination: what do we need to achieve a grand convergence in malaria? PLoS Biol 14: e1002380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Branch O Casapia WM Gamboa DV Hernandez JN Alava FF Roncal N Alvarez E Perez EJ Gotuzzo E , 2005. Clustered local transmission and asymptomatic Plasmodium falciparum and Plasmodium vivax malaria infections in a recently emerged, hypoendemic Peruvian Amazon community. Malar J 4: 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Cook J et al. 2015. Mass screening and treatment on the basis of results of a Plasmodium falciparum-specific rapid diagnostic test did not reduce malaria incidence in Zanzibar. J Infect Dis 211: 1476–1483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Santolamazza F Avellino P Siciliano G Yao FA Lombardo F Ouédraogo JB Modiano D Alano P Mangano VD , 2017. Detection of Plasmodium falciparum male and female gametocytes and determination of parasite sex ratio in human endemic populations by novel, cheap and robust RTqPCR assays. Malar J 16: 468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Karl S Davis TM St-Pierre T , 2009. A comparison of the sensitivities of detection of Plasmodium falciparum gametocytes by magnetic fractionation, thick blood film microscopy, and RT-PCR. Malar J 8: 98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Mbuyi MLT Bouyou-Akotet MK Mawili-Mboumba1 DP , 2014. Molecular detection of Plasmodium falciparum infection in matched peripheral and placental blood samples from delivering women in Libreville, Gabon. Malar Res Treat 014: 486042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Campos IM Uribe ML Cuesta C Franco-Gallego A Carmona-Fonseca J Maestre A , 2011. Diagnosis of gestational, congenital, and placental malaria in Colombia: comparison of the efficacy of microscopy, nested polymerase chain reaction, and histopathology. Am J Trop Med Hyg 84: 929–935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Central TB Division, Directorate General of Health Services, Ministry of Health & Family Welfare, New Delhi National Strategic Plan for Tuberculosis: 2017-25 Elimination by 2025.
- 61. Nair CB Manjula J Subramani PA Nagendrappa PB Manoj MN Malpani S Pullela PK , 2016. Differential diagnosis of malaria on Truelab Uno®, a portable, real-time, MicroPCR device for point-of-care applications. PLoS One 11: e0146961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Notomi T Okayama H Masubuchi H Yonekawa T Watanabe K Amino N Hase T , 2000. Loop-mediated isothermal amplification of DNA. Nucleic Acids Res 28: E63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Morris U Aydin-Schmidt B , 2021. Performance and application of commercially available loop-mediated isothermal amplification (LAMP) kits in malaria endemic and non-endemic settings. Diagnostics (Basel) 11: 336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Indian Council of Medical Research, New Delhi, India COVID-19 Testing Labs. Available at: https://www.icmr.gov.in/pdf/covid/labs/archive/COVID_Testing_Labs_11062020.pdf.
- 65. Rahi M Sharma A , 2021. Free market availability of rapid diagnostics will empower communities to eliminate malaria in India. Am J Trop Med Hyg 105: 281–283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Rahi M Mittal P Kaur J Sharma A , 2021. Malaria card: an empowering tool for patients and for epidemiological recording. J Glob Health Rep 5: e2021062. [Google Scholar]
- 67. Bhowmick I Chutia D Sharma A Rahi M Chhibber-Goel J , 2021. FeverTracker: validation of an mHealth technology platform for malaria surveillance in India. J Med Internet Res. 5: e28951 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Singh N Bharti PK Singh MP Singh R Yeboah-Antwi K Desai M Udhayakumar V Muniyandi M Hamer DH Wylie BJ , 2015. What is the burden of submicroscopic malaria in pregnancy in central India? Pathog Glob Health 109: 30–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Waltmann A Darcy AW Harris x I Koepfli, Cristian, Lodo J vahi V Piziki D Shanks GD Barry AE , 2015. High rates of asymptomatic, sub-microscopic Plasmodium vivax infection and disappearing Plasmodium falciparum malaria in an area of low transmission in Solomon Islands. PLoS Negl Trop Dis 9: e0003758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Ganguly S Saha P Guha SK Biswas A Das S Kundu PK Maji AK , 2013. High prevalence of asymptomatic malaria in a tribal population in eastern India. J Clin Microbiol 51: 1439–1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Vareta J Buchwald AG Barrall A Cohee LM Walldorf JA Coalson JE Seydel K Sixpence A , 2020. Submicroscopic malaria infection is not associated with fever in cross-sectional studies in Malawi. Malar J 19: 233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Tripura R et al. 2017. Submicroscopic Plasmodium prevalence in relation to malaria incidence in 20 villages in western Cambodia. Malar J 16: 56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Sattabongkot J Suansomjit C Nguitragool W Sirichaisinthop J Warit S Tiensuwan M Buates S , 2018. Prevalence of asymptomatic Plasmodium infections with sub-microscopic parasite densities in the northwestern border of Thailand: a potential threat to malaria elimination. Malar J 17: 329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Adu B Issahaque Q Sarkodie-Addo T Kumordjie S Kyei-Baafour E Sinclear CK Eyia-Ampah S Owusu-Yeboa E Theisen M Dodoo D , 2020. Microscopic and submicroscopic asymptomatic Plasmodium falciparum infections in Ghanaian children and protection against febrile malaria. Infect Immun 88: e00125–e20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Hailemeskel E et al. 2021. The epidemiology and detectability of asymptomatic Plasmodium vivax and Plasmodium falciparum infections in low, moderate and high transmission settings in Ethiopia. Malar J 20: 59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Krishna S Yadav A Bhandari S Vishwakarma AK Bharti PK Mandavi PL Bahgel P Basak S Sharma RK Singh N , 2017. Prevalence of malaria in two highly endemic Community Health Centers in the Bastar district, Chhattisgarh showing mixed infections with Plasmodium species. Sci Rep 7: 16860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Kaura T Kaur J Sharma A Dhiman A Pangotra M Upadhyay AK Grover GS Sharma SK , 2019. Prevalence of submicroscopic malaria in low transmission state of Punjab: a potential threat to malaria elimination. J Vector Borne Dis 56: 78–84. [DOI] [PubMed] [Google Scholar]
- 78. Ahmad A Soni P Kumar L Singh MP Verma AK Sharma A Das A Bharti PK , 2021. Comparison of polymerase chain reaction, microscopy, and rapid diagnostic test in malaria detection in a high burden state (Odisha) of India. Pathog Glob Health 115: 267–272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Rahi M, Sharma A. 2022. Should India be considering deployment of the first malaria vaccine RTS,S/AS01? BMJ Glob Health 7: e007870. [DOI] [PMC free article] [PubMed] [Google Scholar]