Abstract
Background
The sleep reduction can change healthy people's hemodynamic control and cardiovascular regulation through increased inflammatory response and altered endothelial function. The objective the study to analyze the association between sleep duration and cardiometabolic risk factors in adolescents in the birth cohort of São Luís (1997/98).
Methods
This is a cross-sectional study with adolescents participating in the birth cohort of São Luís (1997/98). Sleep duration was evaluated using accelerometer data (Actigraph wGT3X-BT). Glycemia, systolic blood pressure (SBP), diastolic blood pressure (DBP), total cholesterol, low-density lipoprotein cholesterol (LDL-c), high-density lipoprotein cholesterol (HDL-c) and triglycerides were considered cardiometabolic factors. The Directed Acyclic Graph (DAG) was used to identify the minimum set of adjustment for confounding.
Results
Out of 1,268 adolescents, 50.3% of them were male. The prevalence of sleep duration of less than 6 h per day was 31.1%. The mean glycemia value was 91.8 mg/dL (± 15.9), DBP was 71.3 mmHg (± 7.5), SBP was 114.9 mmHg (± 12.3), HDL was 48.5 mg/dL (± 11.6), LDL was 89.0 mg/dL (± 25.7), the total cholesterol was 156.0 mg/dL (± 31.1), and triglycerides was 93.6 mg/dL (± 47.2). The crude analysis showed an association between sleep duration and SBP and LDL-c. In the adjusted analysis, the associations did not remain.
Conclusion
Our study showed no association between sleep duration and cardiometabolic outcomes in adolescents.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12889-022-13119-7.
Keywords: Sleep, Glycemia, Arterial Pressure, Cholesterol HDL, Cholesterol LDL, Triglycerides
Background
Sleep is considered an important psychological and biochemical factor, which contributes to optimal health and vitality [1]. According to the literature, the variability of sleep patterns, especially sleep duration, can disrupt circadian cycles of biological processes, resulting in metabolic and endocrine dysfunction, which increase cardiovascular risk [2–4].
In contrast to those observed during wakefulness, the hemodynamic changes found during the sleep period are one of the possible explanations of the relationship between sleep and cardiovascular risk [5, 6]. Thus, sleep reduction can change healthy people's hemodynamic control and cardiovascular regulation through increased inflammatory response and altered endothelial function [7, 8].
The National Sleep Foundation (NSF), in consensus with a multidisciplinary panel of experts, recommends that individuals between 18 and 25 years old sleep 7 to 9 h per night. This sleep time is considered adequate for health and well-being, emphasizing that some individuals may sleep more or less than this time without adverse effects. However, people with sleep durations too far outside the recommended range may either be involved in sleep deprivation or have health problems [9].
The short duration of sleep has become increasingly widespread due to the effect it has on health and quality of life. Current evidence indicates that inadequate sleep duration may play a role in cardiometabolic risk for children and adolescents and with consequences later age [10, 11]. There is evidence of an association between inadequate sleep and abdominal adiposity [12], decreased insulin sensitivity [13], and high blood pressure [14]. However, evidence of potential associations between sleep and lipids, and inflammatory markers, are less convincing [10–13, 15].
Although national [13, 15, 16] and international studies [6, 17, 18] have investigated cardiometabolic risk factors, researches involving adolescents are still scarce. The adverse effects of inadequate sleep duration on physiological functions and cardiovascular risk factors that may underlie the association with increased morbidity and mortality are not fully understood [19].
Thus, understanding the possible adverse effects of sleep duration on these risk factors can help implement interventions aimed at improving adolescents’ sleep habits and, consequently, reduce the emergence of cardiometabolic risk factors throughout life. Therefore, this study aims to analyze the association between sleep duration and cardiometabolic factors in adolescents in the birth cohort of São Luís (1997/1998), in the northeast of Brazil (1997/98).
Methods
This cross-sectional study is part of the birth cohort study (1997/98) in São Luís, State of Maranhão. The methods related to the sample planning of the cohort baseline and follow-ups were previously published and will be explained briefly [20].
The perinatal study of the São Luís cohort was initiated in 10 public and private hospitals in the city, from March 1997 to February 1998, entitled “Determinants throughout the life cycle of obesity, precursors of chronic diseases, human capital and mental health”. After the exclusion of stillbirths, the final sample consisted of 2,493 births. We used data from the third phase of the cohort conducted between January and November 2016, in which participants were already 18/19 years of age. In total, 687 individuals were located and agreed to participate in the study. In this follow-up, a retrospective component was included (with application of a fundamental part of the perinatal questionnaire to mothers of adolescents), conducted with 1,828 adolescents born in São Luís in 1997 and who were initially not drawn to participate in the cohort in the previous phases of the study were included totaling 2,515 adolescents. The new members were selected by random sampling from the System of Information about Liveborns restricting for children born in 1997. Adolescents identified in schools and universities and by means of the social network were also included [20, 21]. Information from 1,538 individuals who used the accelerometer was collected. Of the 1,538 individuals, 179 were excluded due to invalid data and 35 excluded due to some error or defect (calibration error greater than 0.02, patterns not compatible with human movement, incomplete 24-h cycle, and presented quality problems after visual inspection of the plots) (Figure S1).
Furthermore, according to Spearman Brown's formula, 56 adolescents were excluded because they had incomplete data (use of fewer than four days). Thus, the final sample of the São Luís birth cohort in this study was composed of 1,268 adolescents.
Cardiometabolic risk factors
The following cardiometabolic risk factors were considered: blood pressure, glycemia, total cholesterol, triglycerides, low-density lipoprotein cholesterol (LDL-c) and high-density lipoprotein cholesterol (HDL-c).
Arterial Pressure
Both systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured with the Omron HEM 742INT device (Omron, São Paulo, Brazil), three times in each arm, after five minutes at rest [21]. The mean of the three measurements was used.
Glycemia
Without fasting, the glycemia was determined by the enzymatic colorimetric method automated by the Cobas equipment® (Module cobas c 501) from Roche®. The variable was used continuously (mg/dL).
Total cholesterol, triglycerides, low-density lipoprotein cholesterol (LDL-c) and high-density lipoprotein cholesterol (HDL-c)
Total cholesterol, triglycerides, LDL-c, HDL-c were dosed the enzymatic colorimetric method automated by the Cobas equipment® (Module cobas c 501), Roche®.
Blood collection was performed through venipuncture, strictly following the recommendations of good practices in clinical analysis laboratories (http://www.ufma.br/portalUFMA/arquivo/3c85c88c4fc6e33.pdf), and identification is performed at the front of the individuals with barcode labels with the patient's name and exams to be performed, as well as the material is all disposable. Upon arrival at the laboratory, the samples are checked and sent from the technical sectors to carry out the necessary analyzes. In addition, the laboratory follows the requirements of Anvisa (Agência Nacional de Vigilância Sanitária—National Health Surveillance Agency), provided for in the Resolution of the Collegiate Board (RDC—Resolução da Diretoria Colegiadas) 302/2005, for quality control, safety and efficiency in exams. For laboratory tests, fasting was not required. Continuous values were considered for each factor.
Sleep Duration (independent variable)
Sleep duration was evaluated using an accelerometer (GT3X and GTX3 + models) of Actigraf®, collected with a frequency of 60 Hz and epochs of 5 s. Calibration of all accelerometers and the training of those involved in the collection was performed. The survey supervisors explained the usage procedures and invited the individuals to use the accelerometer for 7 continuous days. The instrument should be used 24 h a day, except in bathing and water activities. The crude data were extracted using the Software ActiLife version 6.12, resulting in a spreadsheet (.csv) for each adolescent. Data processing after extraction was performed using the R statistical package (GGIR package version 6.12). The non-human movement filtering, the validation of time of use, calibration, the data quality graphs, and others were performed. Of the 1,538 individuals, 179 were excluded due to invalid data and 35 excluded due to some error or defect (calibration error greater than 0.02, patterns not compatible with human movement, incomplete 24-h cycle, and presented quality problems after visual inspection of the plots). Moreover, we excluded 56 due to insufficient data (use of less than 4 days). Spearman Brown’s formula was applied to test the reliability for the minimum days of use of the accelerometer for sleep [22], parameters, being defined at least 4 nights, with a value of 0.52, being the best result considering the loss of many observations. Sleep duration was identified based on the algorithm proposed by Van Hess et al. [23]. It was categorized into 6 h or more and less than 6 h of sleep, depending on the population's average, since less than 20 participants had 8 h or more of sleep.
Complementary variables
The variables age (in continuous years), gender (male and female), skin color (white, black and mixed-race), adolescent's years of study, socioeconomic classification assessed by Economic Classification Brazil 2016 (Classificação Econômica Brasil – CEB) criterion A/B (B1 + B2), C(C1 + C2), D/E, class A/B being the richest and schooled and D/E the poorest and least educated [24]. Current work and smoking (yes/no), and alcohol consumption (yes/no) were also evaluated [25]. The consumption of in natura or minimally processed foods and ultra-processed foods [26] (%kcal) was assessed using a validated food frequency questionnaire (FFQ) for Brazilian adolescents [27], categorized into tertiles. Physical activity was assessed by accelerometer, in minutes/day of moderate and vigorous physical activity [28], being categorized into tertile (1st tertile: < 15 min 50 s; 2nd tertile: ≥ 15 min 50 s and < 36 min 15 s; 3rd tertile: ≥ 36 min 15 s). Screen time (non-use < 2 hs; ≥ 2 hs to ≤ 5 hs; > 5 hs) was considered exposure to electronic devices, such as mobile phones, tablets, computers, video games, and television during weekdays, whereas weekends were disregarded in the evaluation. Body mass index (BMI) was estimated from the ratio of body mass, in kilograms, by height, in meters, squared (BMI kg/m2 = body mass/height2). To evaluate the major depressive episode or depression (yes/no), the M.I.N.I. Questionnaire was used. (Mini International Neuropsychiatric Interview – Brazilian version 5.0.0—DSM IV) [24].
Data analysis
Descriptive analyses were performed with estimates of absolute frequencies and percentages. The mean outcomes between the groups were compared using the Student’s t-test or Wilcoxon Test and one-way ANOVA or Kruskal–Wallis Test.
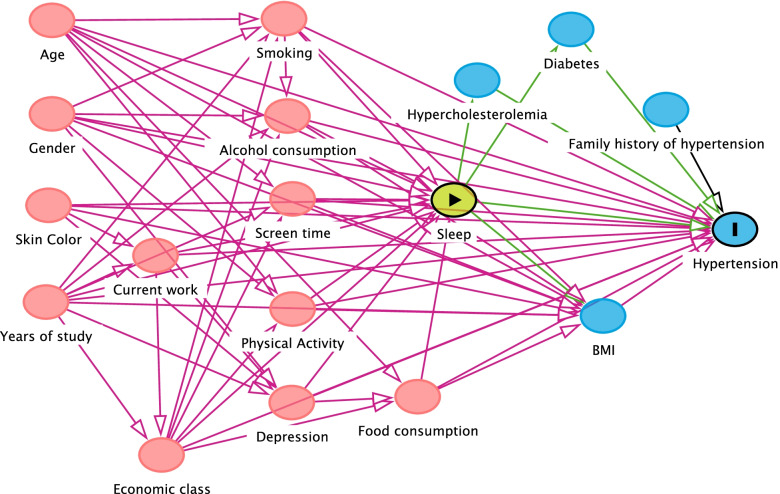
Regarding the covariates of the study, a directed acyclic graph (DAG) was built from the literature review on the study theme, which shows the complex relationship between the different risk factors (Figs. 1, 2, and 3). DAG is a visual and qualitative tool for selecting confounding variables identified from a theoretical model. The arrowheads inform a path between two variables, and it is possible, through pre-established rules, to identify a minimum set of variables for adjustment. After application of DAG’s rules, the minimum adjustment set of variables for analysis of association between sleep duration and cardiometabolic outcomes (blood pressure, dyslipidemia and glycemia) were: For the associations between sleep and diabetes, the following variables were selected: age, gender, adolescent years of study, skin color, economic class, current work, alcohol consumption, smoking, physical activity, screen time, food consumption. For sleep and blood pressure, in turn, the variables selected were adjusted for: age, gender, adolescent years of study, skin color, economic class, current work, alcohol consumption, smoking, physical activity, screen time, food consumption, depression. In the association between sleep and dyslipidemias, the adjustment variables were: age, gender, adolescent years of study, skin color, economic class, current work, alcohol consumption, smoking, physical activity, screen time, food consumption.
Fig. 1.
Acyclic graph directed to the association between sleep and glycemia
Fig. 2.
Acyclic graph directed to the association between sleep and dyslipidemias
Fig. 3.
Acyclic graph directed to the association between sleep and blood pressure
To analyze the relationship between sleep duration and cardiometabolic outcomes, a weighted by propensity score was used from the Inverse Probability of Treatment Weighting (IPTW). This study used the propensity score as a way to correct the imbalance in the distribution of predictors (covariate) that occur in observational studies, thus making the groups comparable and avoiding biased estimates of the effect of sleep duration and cardiometabolic outcomes.
The propensity score calculation was performed using the linear regression. The application of linear regression allowed the estimation of the selection probability of adolescents, conditional on the values of covariates identified in the DAG (Figs. 1, 2 and 3).
Checking the common support area and balancing the groups verified adequate propensity score estimation. A balance in the distribution of observed covariates was achieved, obtaining standardized differences between means and proportions less than 0.1 and for variances between 0.18–1.2. The effect of of sleep duration on cardiometabolic outcomes was calculated using the beta coefficient with 95% confidence intervals (95% CI) for each cardiometabolic outcome considered in the analysis. No interaction was observed between sleep duration and gender concerning glycemia, blood pressure, and dyslipidemia outcomes evaluated by the gender/sleep duration ratio. Therefore, the sample was not stratified by gender.
Statistical analysis was conducted in Stata 14.0 (Stata Corporation, College Station, Texas, USA).
Ethical aspects
The project referring to the birth cohort of São Luís (1997/98) was approved by the Research Ethics Committee of the University Hospital—Federal University of Maranhão (UFMA), under Opinion no. 1.302.489. In all phases of the cohort, the Informed Consent Form was signed by the individual or guardian. All projects meet the criteria in Resolution (466/2012) of the National Health Council and its complementary regulations.
Results
Studies have assessed the distribution of participants according to the use or not of the accelerometer, with differences between the categories of age (p ≤ 0.001), gender (p ≤ 0.001), schooling (p ≤ 0.001), skin color (p = 0.016), economic class (p ≤ 0.001), leisure-time physical activity (p ≤ 0.001), drug use (p = 0.008). Most participants who used the accelerometer were 18 years old (86.8%), had lower schooling (71.6%), were male (50.45%), the poorest (Class C) (49.5%), and the most physically active (42.9%) [29].
The mean glycemia value was 91.8 mg/dL (± 15.9), diastolic blood pressure was 71.3 mmHg (± 7.5), and systolic blood pressure was 114.9 mmHg (± 12.3), HDL was 48.5 mg/dL (± 11.6), LDL was 89.0 mg/dL (± 25.7), total cholesterol was 156.0 mg/dL (± 31.1), triglycerides was 93.6 mg/dL (± 47.2) and mean sleep time was 6 (± 1.0) hours per day. The prevalence of sleep less than 6 h per day was 31.1% (Table 1).
Table 1.
Characterization of the sample and mean and standard deviation of outcomes according to demographic, socioeconomic, lifestyle, and sleep duration characteristics in adolescents (n = 1,268). São Luís, Maranhão, Brazil, 2016/2017
| Variable |
Glycemia (mg/dL) |
DBP (mmHg) |
SBP (mmHg) |
HDL-c (mg/dL) |
LDL-c (mg/dL) |
Cholesterol total (mg/dL) |
Triglycerides (mg/dL) |
|
|---|---|---|---|---|---|---|---|---|
| n (%) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | |
| Total | 1,268 | 91.8(15.9) | 71.3 (7.5) | 114.9 (12.3) | 48.5(11.6) | 89.0 (25.7) | 156.0 (31.1) | 93.6 (47.2) |
| Age (n = 1,268) | p = 0.327** | p = 0.839* | p = 0.011** | p = 0.010** | p = 0.171* | p = 0.038* | p = 0.986* | |
| 18 years | 1105(87.1) | 91.9 (16.1) | 71.4 (7.5) | 115.2 (12.4) | 48.2 (11.5) | 88.5(25.2) | 155.2(30.4) | 93.3 (46.2) |
| 19 years | 163 (12.9) | 90.6 (1.2) | 70.5 (7.2) | 112.0 (11.6) | 50.7 (11.9) | 92.7 (28.7) | 161.8 (35.1) | 95.4 (53.6) |
| Gender (n = 1,268) | < 0.001* | p = 0.030** | < 0.001* | < 0.001* | p < 0.001** | p < 0.001** | p < 0.001** | |
| Male | 638 (50.3) | 92.7 (17.0) | 72.2 (7.7) | 121.4 (10.9) | 45.0 (10.2) | 85.9 (24.9) | 150.6 (29.2) | 99.7 (49.2) |
| Female | 630 (49.7) | 90.8 (14.7) | 70.3 (7.2) | 108.2 (9.9) | 52.1(11.8) | 92.2 (26.1) | 161.7(31.9) | 87.2(44.2) |
| Adolescent years of study (n = 1,261) | p = 0.090* | p = 0.042** | p = 0.606** | p = 0.207** | p = 0.408** | p = 0.094** | p = 0.830 | |
| No schooling at 11 years | 1,202 (95.3) | 92.0 (16.0) | 71.2 (7.5) | 114.9 (12.4) | 48.4 (11.5) | 88.9(25.5) | 155.6 (31.1) | 93.4 (47.3) |
| 12 or more | 59(4.7) | 87.6(11.3) | 71.7(7.3) | 114.1 (11.6) | 50.3 (12.5) | 91.8 (29.7) | 162.7(31.1) | 94.8 (44.4) |
| Skin Color (n = 1,261) | p = 0.107**** | p = 0.175**** | p = 0.213**** | p = 0.034*** | p = 0.107*** | p = 0.177* | p = 0.137**** | |
| White | 235 (18.6) | 92.5 (15.2) | 70.6 (7.0) | 114.5 (11.6) | 48.2(11.9) | 89.9(28.0) | 156.0(33.4) | 93.1 (46.6) |
| Black | 202 (16.0) | 89.6 (14.8) | 71.3 (7.2) | 114.9 (13.1) | 50.8(12.8) | 89.5(14.9) | 159.2(31.1) | 92.6 (51.7) |
| Mixed race | 824 (65.4) | 92.1 (16.4) | 71.4 (7.7) | 114.9 (12.3) | 48.0 (11.1) | 88.7 (25.2) | 155.3 (30.3) | 93.9 (46.4) |
| Economic class (n = 1,110) | p = 0.367**** | p = 0.536**** | p = 0.703***** | p = 0.483**** | p = 0.150*** | p = 0.087**** | p = 0.069**** | |
| A/B | 308 (27.8) | 90.4 (14.5) | 70.8 (7.1) | 115.0 (12.2) | 49.0(11.2) | 89.8(26.5) | 157.5(31.7) | 93.4 (45.3) |
| C | 552 (49.6) | 91.6 (14.1) | 71.3 (7.5) | 115.1 (12.5) | 48.7 (11.8) | 88.0 (24.5) | 154.6 (29.7) | 92.3 (46.7) |
| D/E | 251 (22.6) | 92.6 (15.2) | 71.2 (7.3) | 113.1(12.0) | 48.5 (12.0) | 91.1(26.8) | 158.7(33.4) | 95.0 (51.7) |
| Current works (n = 1,268) | p = 0.568** | p = 0.242** | p = 0.727** | p = 0.909** | p = 0.631** | p = 0.811** | p = 0.584** | |
| No | 1,066 (84.1) | 91.9 (16.2) | 71.4 (7.6) | 11.8 (12.4) | 48.5(11.5) | 88.9(25.7) | 155.9(31.0) | 93.2 (47.1) |
| Yes | 2,020 (15.9) | 91.2 (14.6) | 70.7 (7.2) | 115.4(11.9) | 48.6(11.8) | 89.8(25.5) | 156.5 (31.2) | 95.3(47.8) |
| Alcohol consumption (n = 1,257) | p = 0.742* | p = 0.906** | p = 0.347** | p = 0.705** | p = 0.304** | p = 0.896** | p = 0.179** | |
| No | 744 (59.1) | 91.7 (14.8) | 71.1 (7.5) | 114.6 (12.1) | 48.4(11.9) | 89.7(25.6) | 155.9(31.5) | 91.3(45.4) |
| Yes | 513 (40.9) | 91.8 (17.1) | 71.5 (7.6) | 115.3 (12.6) | 48.7(1.1) | 88.2(25.6) | 156.2(30.3) | 96.4 (48.0) |
| Smoking (n = 1,268) | p = 0.189** | p = 0.704** | p = 0.528* | p = 0.127** | p = 0.876** | p = 0.512** | p = 0.044** | |
| No | 1,228 (96.9) | 91.7 (14.6) | 71.2 (7.5) | 114.8 (12.2) | 48.6 (11.5) | 89.0 (25.6) | 155.9 (30.9) | 93.1 (47.3) |
| Yes | 40 (3.1) | 94.3 (38.7) | 72.0 (8.3) | 116.9 (15.1) | 45.7(11.7) | 89.6(28.9) | 159.2 (34.7) | 108.3 (42.8) |
| Tertile physical activity (n = 1,268) | p = 0.148*** | p = 0.113**** | p = 0.297**** | p = 0.710**** | p = 0.522**** | p = 0.009*** | p = 0.585**** | |
| 1st tertile (< 15 min 50 s) | 426 (33.6) | 91.0 (15.9) | 71.1 (7.2) | 113.0(11.7) | 48.6(11.6) | 90.8(25.5) | 157.7(30.5) | 92.9 (46.6) |
| 2nd tertile (≥ 15 min 50 s and < 36 min 15 s) | 427 (33.7) | 92.1 (17.7) | 71.1 (7.4) | 114.5 (12.5) | 49.3(11.7) | 89.9(26.4) | 157.8(33.2) | 93.1 (48.6) |
| 3rd tertile (≥ 36 min 15 s) | 415 (32.7) | 92.3 (13.9) | 71.5 (9.9) | 117.2 (12.5) | 47.5(11.3) | 86.3(25.0) | 152.4(29.2) | 94.7 (46.5) |
| BMI (n = 1,253) | p = 0.664*** | p = 0.120**** | p = 0.077**** | < 0.001*** | p < 0.001*** | < 0.001*** | p < 0.001*** | |
| Adequate | 1,011 (80.7) | 91.9 (16.6) | 70.5 (7.3) | 113.3 (11.6) | 49.2(11.3) | 87.2 (24.2) | 153.8 (28.6) | 87.8 (40.9) |
| Overweight | 190 (15.2) | 91.7 (12.3) | 73.4 (7.0) | 120.7(12.6) | 46.2(12.6) | 94.3(27.7) | 162.1 (35.3) | 110.6 (56.6) |
| Obesity | 52 (4.1) | 92.3 (14.3) | 78.3 (8.7) | 124.4 (14.0) | 40.6(9.5) | 103.0(35.0) | 170.2(39.6) | 132.3 (62.9) |
| Food ultra-processed (tertile) (n = 1,254) | p = 0.917*** | p = 0.424**** | p = 0.404**** | p = 0.588**** | p = 0.781**** | p = 0.496**** | p = 0.070**** | |
| 1st tertile | 426 (34.0) | 91.9 (16.7) | 70.9 (7.6) | 115.7 (12.5) | 47.9(10.9) | 88.5 (25.3) | 154.7 (30.1) | 93.4 (44.5) |
| 2nd tertile | 396 (31.6) | 92.0 (14.0) | 71.4 (7.7) | 115.3 (12.6) | 48.8 (12.4) | 88.2 (26.2) | 156.1 (31.9) | 94.0 (49.9) |
| 3rd tertile | 432 (34.4) | 16.8 (16.8) | 71.4(7.2) | 16.8 (16.8) | 48.7 (11.4) | 90.1(25.5) | 157.2 (31.3) | 93.3 (47.7) |
| Food in natura or minimaly processed (tertile) (n = 1,254) | p = 0.980*** | p = 0.252**** | p = 0.146*** | p = 0.172**** | p = 0.753**** | p = 0.510**** | p = 0.944*** | |
| 1st tertile | 439 (35.0) | 91.4 (12.9) | 71.4(7.2) | 113.9 (11.7) | 48.9 (11.5) | 90.4 (26.1) | 157.8 (32.0) | 94.4 (49.6) |
| 2nd tertile | 384 (30.6) | 92.6 (18.4) | 71.2 (7.4) | 114.8 (12.2) | 48.6 (12.15) | 87.6 (25.2) | 155.2 (30.6) | 93.8 (48.2) |
| 3rd tertile | 431 (34.4) | 91.6 (16.5) | 71.1 (7.8) | 116.1 (12.9) | 48.0 (11.0) | 88.9 (25.6) | 154.8 (30.4) | 92.5 (44.1) |
| Depression | p = 0.027* | p = 0.152* | p < 0.001* | p = 0.113** | p = 0.858** | p = 0.307** | p = 0.514** | |
| No | 1.128(88.9) | 92.2(16.3) | 71.4(7.6) | 115.4(12.5) | 48.3(11.5) | 89.0(25.5) | 155.7(30.9) | 93.3(47.3) |
| Yes | 140(11.1) | 88.7(11.6) | 70.2(6.6) | 110.4(9.7) | 50,0(12.1) | 89.4(26.8) | 158.6(32.0) | 96.1(46.3) |
| Sleep duration (n = 1,268) | p = 0.857* | p = 0.202** | 0.001** | p = 0.145** | p = 0.036** | p = 0.098** | p = 0.980* | |
| ≥ 6 h | 874 (68.9) | 91.5 (14.2) | 71.1 (7.5) | 114.0 (12.5) | 48.8 (11.4) | 90.1 (26.2) | 157.0(31.4) | 92.6 (45.1) |
| < 6 h | 394 (31.1) | 92.3 (19.2) | 71.7 (7.5) | 116.9(11.7) | 47.8(11.9) | 86.7 (24.4) | 153.8 (30.3) | 95.6 (51.6) |
Caption: SD Standard deviation, DBP Diastolic arterial pressure, SBP Systolic arterial pressure, LDL-c Low-density lipoprotein cholesterol, HDL-c high density lipoprotein cholesterol, *Wilcoxon rank Sum test, ** Student t test, *** Kruskal Wallis test, **** one-way ANOVA
We evaluated 1,268 individuals; 50.3% were men, 87.1% were 18 years old, 95.3% had none to 11 years of schooling, 55.3% were mixed-raced, 49.6% belonged to socioeconomic class C, 84.1% did not work, 59.2% did not consume alcohol, 96.9% did not smoke, 80.7% had adequate BMI, 11.1% have depression (Table 1).
Table 1 also shows the difference in the means of the outcomes considering the sociodemographic and lifestyle variables. Men had a higher mean glycemia than women. Those with higher schooling had higher mean diastolic blood pressure than their peers. Men and those aged 18 years had higher mean systolic blood pressure when compared to 19 years of age and women, respectively. Adolescents aged 19 years, women, black, and those with adequate nutritional status had higher means of HDL. Women and obese had higher LDL averages. Adolescents aged 19 years, women, obese, and those belonging to the 2nd quartile of PA had higher total cholesterol means. Concerning triglycerides, the highest averages were found among men aged 19 years or older, smokers and obese. Individuals with a sleep duration less than 6 h had higher mean SBP, while those that sleep more than 6 h had a higher mean LDL.
The data in Table 2 show the results of the crude and adjusted analyses concerning the sleep duration associated with the outcomes. The crude analysis showed an association between sleep duration and systolic blood pressure and LDL. Adolescents who have < 6 h of sleep per day have 2.90 mmHg more systolic blood pressure when compared to those who have ≥ 6 h of sleep. Those with < 6 h of sleep per day have 3.33 mg/dL less LDL-c when compared to those with ≥ 6 h of sleep. However, after adjustments to sociodemographic and lifestyle variables, the associations did not remain. That is, there was no association between sleep duration and cardiometabolic risk outcomes.
Table 2.
Crude and adjusted analysis of the association between sleep duration and cardiometabolic risk factors in adolescents. São Luís, Maranhão, Brazil, 2016/2017
| Variables | Crude analysis | Adjusted analysis | ||
|---|---|---|---|---|
| β (95%IC) | p | β (95%IC) | p | |
| Glycemia (mg/dL) | ||||
| Sleep duration | 0.407 | 0.760 | ||
| ≥ 6 h | 0 | 0a | ||
| < 6 h | 0.82(-1.11;2.75) | 0.33(-1.80;2.47) | ||
| SBP (mmHg) | ||||
| Sleep duration | 0.001 | 0.910 | ||
| ≥ 6 h | 0 | 0b | ||
| < 6 h | 2.90(1.43;4.37) | -0.08(-1.50;1.34) | ||
| DBP (mmHg) | ||||
| Sleep duration | 0.202 | 0.898 | ||
| ≥ 6 h | 0 | 0b | ||
| < 6 h | 0.58(-0.31;1.48) | 0.06(-0.92;1.05) | ||
| HDL-c (mg/dL) | ||||
| Sleep duration | 0.145 | 0.286 | ||
| ≥ 6 h | 0 | 0a | ||
| < 6 h | -1.04(-2.44;0.36) | 0.93(-0.78;2.63) | ||
| LDL-c (mg/dL) | ||||
| Sleep duration | 0.036 | 0.303 | ||
| ≥ 6 h | 0 | 0a | ||
| < 6 h | -3.33(-6.45;-0.22) | -1.90(-5.51;1.71) | ||
| Total cholesterol (mg/dL) | ||||
| Sleep duration | 0.098 | 0.941 | ||
| ≥ 6 h | 0 | 0a | ||
| < 6 h | -3.18(-6.94;0.59) | -0.16(-4.55;4.22) | ||
| Triglycerides (mg/dL) | ||||
| Sleep duration | 0.308 | 0.969 | ||
| ≥ 6 h | 0 | 0a | ||
| < 6 h | 2.98(-2.75;8.71) | 0.13(-6.36;6.62) | ||
95%IC 95% confidence interval; DBP Diastolic blood pressure; SBP systolic blood pressure; LDL-c low-density lipoprotein cholesterol; HDL-c high density lipoprotein cholesterol. aModel 1 = Adjusted for age, gender, adolescent years of study, skin color, economic class, current work, alcohol consumption, smoking, physical activity, screen time, food consumption; bModel 2 = Adjusted Model 1 + depression
Discussion
We aimed to analyze the association between sleep duration and cardiometabolic factors in adolescents of the birth cohort (1997/98) in São Luís, Maranhão. In this study, we observed no associations between sleep duration and cardiometabolic factors in adolescents.
Like this study, some studies that used accelerometry in the evaluation of sleep duration also did not observe significant associations with cardiometabolic outcomes in adolescents. The study by Sung et al. [30] with obese American adolescents (10 to 16 years) showed no association between sleep duration and metabolic outcomes. Concerning lipid profile, the presence of heteroscedasticity in the data limited the analysis of the associations. Hjorth et al. [31], reported negative associations between sleep duration and metabolic syndrome scores in Danish children and adolescents aged 8 to 11 years. However, the associations did not remain in the analyses adjusted for gender, age, pubertal stage, leisure physical activity, sedentary behavior, and fat mass index. With the inclusion of the fat mass index in the adjustment model, the authors suggest adiposity could mediate this relationship.
On the other hand, other studies that have also used accelerometers have reported significant associations between sleep duration and blood pressure in adolescents. Javaheri et al. [32] reported a negative association between sleep duration (each one-hour increase) and SBP in American adolescents aged 13 to 16 years and no association for DBP. In the study by Meininger et al., [33] American adolescents aged 11 to 16 years showed lower SBP with each one-hour increase in night sleep, and lower SBP and DBP for each additional hour of daytime sleep. Another study conducted with American adolescents aged 14 to 19 years showed an association between the shorter sleep duration and lower SBP and DBP in the 24 h throughout the 2 days of outpatient monitoring [34].
Although our study showed no association between sleep duration and cardiometabolic factors, studies have suggested some mechanisms that could explain this relationship, such as circadian misalignment, increased sympathetic nervous system activity, and renal sodium retention. Hyperinsulinemia is believed to raise blood pressure by increasing renal sodium retention or stimulating the sympathetic nervous system or a combination of both, which could change cardiometabolic factors [11].
According to the systematic review of observational studies (including questionnaires, self-report of parents and accelerometry) conducted by Quist et al. [11], we observed some evidence of association with cardiometabolic outcomes in children and adolescents. Regarding glycemia (n = 21 studies) and blood pressure (n = 30 studies), the evidence was more conclusive, whereas regarding the associations with serum lipids (n = 14 studies) it was inconsistent. However, the authors [11] point out that it should be considered that most studies relating sleep with published cardiometabolic outcomes were cross-sectional in nature, and subjective measures for sleep assessment often evaluated sleep.
The difference between sleep duration measured by accelerometer and by other methods is an important point. From the total minutes without movement in the wrist in a given period of time [23, 35], accelerometer provides objective information about daily variability and sleep duration, recording information in the home sleep environment, not being influenced by patient expectations and memory bias [35]. However, although lack of body movement is an important indicator of the presence of sleep, accelerometer does not allow the evaluation of subjective aspects of sleep, such as the quality or satisfaction with sleep, [35, 36] and the results are limited to sleep architecture. In this sense, the lack of association may be due to the sleep indicator used in this study (accelerometer), and the results should be interpreted with caution. According to evidence in the literature, the evaluation of sleep by subjective measures may be associated with cardiometabolic outcomes [11], thus, further studies investigating aspects of sleep and these outcomes in adolescence are needed.
The study has some limitations. One of them refers to collecting food consumption data using FFQ, whose quantification is often inaccurate and subjected to memory bias [37]. However, these food consumption data were obtained through a validated instrument for the study population. Another limitation may be related to the losses of participants who used the accelerometer, which may lead to selection bias. It is the adolescents with better health who may have attended to evaluate cardiometabolic factors, which may underestimate the values. In addition, we have the limitation that we cannot distinguish fasting versus non-fasting blood samples.
On the other hand, this study also has strengths. The main strength was the extensive training of the team of interviewers, performed to reduce the biases during data collection. Another strong point was the construction and use of DAG to identify confounding factors, which indicated the minimum set of variables for adjustment, seeking to avoid inadequate or unnecessary adjustments.
It is important to note that the study cannot have the results extrapolated to the population because the sample was not probabilistic.
Conclusions
These results allow us to observe no association between sleep duration and cardiometabolic risk factors in adolescence. However, these associations may not have occurred at this stage yet and may arise in other life phases. Despite the inexistence of association of factors in this phase, monitoring sleep duration and cardiometabolic risk profile is important to prevent damage in the next phases of the life.
Supplementary Information
Additional file 1. Legend: Sistema de Informações sobre Nascidos Vivos (SINASC)/ Information System on Live Births (SINASC)
Acknowledgements
All authors are grateful to all teenagers, their fathers, and their mothers. We also wish to thank research assistants involved.
Abbreviations
- SBP
Systolic blood pressure
- DBP
Diastolic blood pressure
- LDL-c
Low-density lipoprotein cholesterol
- HDL-c
High-density lipoprotein cholesterol
- DAG
Directed Acyclic Graph
- mg/dl
Milligrams per deciliter
- mmHg
Millimeters of mercury
- NSF
National Sleep Foundation
- FFQ
Food frequency questionnaire
- Classificação Econômica Brasil – CEB
Economic Classification Brazil
- BMI
Body mass index
- DSM IV
Diagnostic and Statistical Manual of Mental Disorders
- MINI
Mini International Neuropsychiatric Interview
- UFMA
Federal University of Maranhão
- MA
Maranhão
- DECIT
Department of Science and Technology
- SD
Standard deviation
- 95%IC
95% Confidence interval
Authors' contributions
SCC, CCCR, RFLB, and AAMS designed the research; SCC, LYGA, EISM, and AAMS conducted the research; SCC, LYGA, EISM, and AAMS analyzed data; and SCC, LYGA, EISM, ARB, CCCR, RFLB, and AAMS wrote the paper. SCC had primary responsibility for the final content. All authors read and approved the final draft.
Funding
This research was funded by Department of Science and Technology – DECIT (Ministry of Health)
Availability of data and materials
The data that support the findings of this study are available from e-mail rosangela.flb@ufma.br, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of Rosangela Fernandes Lucena Batista.
Declarations
Ethics approval and consent to participate
The project referring to the birth cohort of São Luís (1997/98) was approved by the Research Ethics Committee of the University Hospital - Federal University of Maranhão (UFMA), under Opinion no. 1.302.489. In all phases of the cohort, the Informed Consent Form was signed by the individual or guardian. All projects meet the criteria in Resolution (466/2012) of the National Health Council and its complementary regulations.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Susana Cararo Confortin, Email: susanaconfortin@gmail.com.
Liliana Yanet Gómez Aristizábal, Email: lilianayanetgomez@gmail.com.
Elma Izze da Silva Magalhães, Email: elma_izze@hotmail.com.
Aline Rodrigues Barbosa, Email: alinerb13@yahoo.com.br.
Cecilia Claudia Costa Ribeiro, Email: cecilia_ribeiro@hotmail.com.
Rosângela Fernandes Lucena Batista, Email: rosangela.flb@ufma.br.
Antônio Augusto Moura da Silva, Email: aamouradasilva@gmail.com.
References
- 1.Wang L, Li J, Du Y, Sun T, Na L, Wang Z. The relationship between sleep onset time and cardiometabolic biomarkers in Chinese communities: A cross-sectional study. BMC Public Health. 2020;20(1):1–9. doi: 10.1186/s12889-019-7969-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Makarem N, Zuraikat FM, Aggarwal B, Jelic S, St-Onge MP. Variability in Sleep Patterns: an Emerging Risk Factor for Hypertension. Curr Hypertens Rep. 2020;22(2):19. doi: 10.1007/s11906-020-1025-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Feng X, Liu Q, Li Y, Zhao F, Chang H, Lyu J. Longitudinal study of the relationship between sleep duration and hypertension in Chinese adult residents (CHNS 2004–2011) Sleep Med. 2019;58:88–92. doi: 10.1016/j.sleep.2019.01.006. [DOI] [PubMed] [Google Scholar]
- 4.Javaheri S, Storfer-Isser A, Rossen CL, Redline S. Sleep Quality and Elevated Blood Pressure in Adolescents. Circulation. 2008;118(10):1034–1040. doi: 10.1161/CIRCULATIONAHA.108.766410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Trinder J, Waloszek J, Woods MJ, Jordan AS. Sleep and cardiovascular regulation. Pflügers Arch - Eur J Physiol. 2012;463(1):161–8. 10.1007/s00424-011-1041-3. [DOI] [PubMed]
- 6.Spiegel K, Tasali E, Leproult R, Van Cauter E. Effects of poor and short sleep on glucose metabolism and obesity risk. Nat Rev Endocrinol. 2009;5(5):253–61. Available from: http://www.nature.com/articles/nrendo.2009.23 [DOI] [PMC free article] [PubMed]
- 7.Pillai V, Cheng P, Kalmbach DA, Roehrs T, Roth T, Drake CL. Prevalence and Predictors of Prescription Sleep Aid Use among Individuals with DSM-5 Insomnia: The Role of Hyperarousal. Sleep. 2016;39(4):825–32. Available from: https://academic.oup.com/sleep/article/39/4/825/2453997 [DOI] [PMC free article] [PubMed]
- 8.Canto CB, Onuki Y, Bruinsma B, van der Werf YD, De Zeeuw CI. The Sleeping Cerebellum. Trends Neurosci. 2017;40(5):309–23. Available from: https://linkinghub.elsevier.com/retrieve/pii/S0166223617300309 [DOI] [PubMed]
- 9.Hirshkowitz M, Whiton K, Albert SM, Alessi C, Bruni O, DonCarlos L, et al. National Sleep Foundation’s sleep time duration recommendations: methodology and results summary. Sleep Heal. 2015;1(1):40–3. Available from: http://dx.doi.org/10.1016/j.sleh.2014.12.010 [DOI] [PubMed]
- 10.Fobian AD, Elliott L, Louie T. A Systematic Review of Sleep, Hypertension, and Cardiovascular Risk in Children and Adolescents. Curr Hypertens Rep. 2018;20(5):42. doi: 10.1007/s11906-018-0841-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Quist JS, Sjödin A, Chaput JP, Hjorth MF. Sleep and cardiometabolic risk in children and adolescents. Sleep Med Rev. 2016;29:76–100. doi: 10.1016/j.smrv.2015.09.001. [DOI] [PubMed] [Google Scholar]
- 12.Seo SH, Shim YS. Association of Sleep Duration with Obesity and Cardiometabolic Risk Factors in Children and Adolescents: A Population-Based Study. Sci Rep. 2019;9(1):1–10. doi: 10.1038/s41598-019-45951-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.De Bernardi Rodrigues AM, De Cassia Da Silva C, Vasques ACJ, Camilo DF, Barreiro F, Cassani RSL, et al. Association of sleep deprivation with reduction in insulin sensitivity as assessed by the hyperglycemic clamp technique in adolescents. JAMA Pediatr. 2016;170(5):487–94. doi: 10.1001/jamapediatrics.2015.4365. [DOI] [PubMed] [Google Scholar]
- 14.Guo X, Zheng L, Wang J, Zhang X, Zhang X, Li J, et al. Epidemiological evidence for the link between sleep duration and high blood pressure: A systematic review and meta-analysis. Sleep Med. 2013;14(4):324–332. doi: 10.1016/j.sleep.2012.12.001. [DOI] [PubMed] [Google Scholar]
- 15.De Oliveira LC, Passos MAZ, Vellozo EP, Dos Santos Quaresma MVL, De Piano Ganen A. Associação entre o padrão de sono e marcadores de risco cardiometabólicos de adolescentes. DEMETRA Aliment Nutr Saúde. 2020;15:e45177. doi: 10.12957/demetra.2020.45177. [DOI] [Google Scholar]
- 16.Wells JCK, Hallal PC, Reichert FF, Menezes AMB, Araújo CLP, Victora CG. Sleep patterns and television viewing in relation to obesity and blood pressure: Evidence from an adolescent Brazilian birth cohort. Int J Obes. 2008;32(7):1042–1049. doi: 10.1038/ijo.2008.37. [DOI] [PubMed] [Google Scholar]
- 17.Azadbakht L, Kelishadi R, Khodarahmi M, Qorbani M, Heshmat R, Motlagh ME, et al. The association of sleep duration and cardiometabolic risk factors in a national sample of children and adolescents: The CASPIAN III Study. Nutrition. 2013;29(9):1133–1141. doi: 10.1016/j.nut.2013.03.006. [DOI] [PubMed] [Google Scholar]
- 18.Crowley SJ, Acebo C, Carskadon MA. Sleep, circadian rhythms, and delayed phase in adolescence. Sleep Med. 2007;8(6):602–12. Available from: https://linkinghub.elsevier.com/retrieve/pii/S1389945706007076 [DOI] [PubMed]
- 19.Covassin N, Singh P. Sleep Duration and Cardiovascular Disease Risk. Sleep Med Clin. 2016;11(1):81–9. Available from: https://linkinghub.elsevier.com/retrieve/pii/S1556407X15001381 [DOI] [PMC free article] [PubMed]
- 20.Confortin SC, Ribeiro MRC, Barros AJD, Menezes AMB, Horta BL, Victora CG, et al. RPS Brazilian Birth Cohorts Consortium (Ribeirão Preto, Pelotas and São Luís): history, objectives and methods. Cad Saude Publica. 2021;37(4). Available from: http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0102-311X2021000400601&tlng=en [DOI] [PubMed]
- 21.Simões VMF, Batista RFL, de Britto e Alves MTSS, Ribeiro CCC, Thomaz EBAF, de Carvalho CA, et al. Health of adolescents in the 1997/1998 birth cohort in São Luís, Maranhão State, Brazil. Cad Saude Publica. 2020;36(7):e00164519. doi: 10.1590/0102-311x00164519. [DOI] [PubMed] [Google Scholar]
- 22.Eisinga R, Te GM, Pelzer B. The reliability of a two-item scale: Pearson, Cronbach, or Spearman-Brown? Int J Public Health. 2013;58(4):637–642. doi: 10.1007/s00038-012-0416-3. [DOI] [PubMed] [Google Scholar]
- 23.Van Hees VT, Sabia S, Anderson KN, Denton SJ, Oliver J, Catt M, et al. A novel, open access method to assess sleep duration using a wrist-worn accelerometer. PLoS ONE. 2015;10(11):1–13. doi: 10.1371/journal.pone.0142533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.ABEP. Critério Brasil e Alterações na aplicação do Critério Brasil, válidas a partir de 16/04/2018. Assoc Bras Empres Pesqui Critério Classif econômica Bras. 2018;1–6. Available from: http://www.abep.org/criterio-brasil
- 25.Moretti-Pires RO, Corradi-Webster CM. Adaptation and validation of the Alcohol Use Disorders Identifi cation Test (AUDIT) for a river population in the Brazilian Amazon. Cad Saude Publica. 2011;27(3):497–509. doi: 10.1590/S0102-311X2011000300010. [DOI] [PubMed] [Google Scholar]
- 26.Monteiro CA, Moubarac JC, Levy RB, Canella DS, Da Costa Louzada ML, Cannon G. Household availability of ultra-processed foods and obesity in nineteen European countries. Public Health Nutr. 2018;21(1):18–26. doi: 10.1017/S1368980017001379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bogea EG, França AKTC, Bragança MLBM, Vaz JS, Assunção MC, Barbieri MA, et al. Relative validity of a food frequency questionnaire for adolescents from a capital in the Northeastern region of Brazil. Brazilian J Med Biol Res. 2021;54(1):1–9. doi: 10.1590/1414-431x20209991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hildebrand M, Hansen BH, van Hees VT, Ekelund U. Evaluation of raw acceleration sedentary thresholds in children and adults. Scand J Med Sci Sport. 2017;27(12):1814–1823. doi: 10.1111/sms.12795. [DOI] [PubMed] [Google Scholar]
- 29.Confortin SC, Batista RFL, Barbosa AR, Wendt A, Silva I, MTSS de B e Alves, et al. Is sleep time associated with handgrip strength in adolescentsthe 1997/1998 São Luís Birth Cohort? Cien Saude Colet. 2021;27(3):1147–1155. doi: 10.1590/1413-81232022273.03132021. [DOI] [PubMed] [Google Scholar]
- 30.Sung V, Beebe DW, VanDyke R, Fenchel MC, Crimmins NA, Kirk S, et al. Does sleep duration predict metabolic risk in obese adolescents attending tertiary services? A cross-sectional study Sleep. 2011;34(7):891–898. doi: 10.5665/SLEEP.1122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hjorth MF, Chaput JP, Damsgaard CT, Dalskov SM, Andersen R, Astrup A, et al. Low physical activity level and short sleep duration are associated with an increased cardio-metabolic risk profile: A longitudinal study in 8–11 year old Danish children. PLoS One. 2014;9(8):e104677. doi: 10.1371/journal.pone.0104677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Javaheri S, Storfer-Isser A, Rosen CL, Redline S. Sleep Quality and Elevated Blood Pressure in Adolescents. Circulation. 2008;118(10):1034–40. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3624763/pdf/nihms412728.pdf [DOI] [PMC free article] [PubMed]
- 33.Meininger JC, Gallagher MR, Eissa MA, Nguyen TQ, Chan W. Sleep duration and its association with ambulatory blood pressure in a school-based, diverse sample of adolescents. Am J Hypertens. 2014;27(7):948–955. doi: 10.1093/ajh/hpt297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mezick EJ, Hall M, Matthews KA. Sleep Duration and Ambulatory Blood Pressure in Black and White Adolescents. Hypertension. 2012;59(3):747–52. Available from: 10.1161/HYPERTENSIONAHA.111.184770 [DOI] [PMC free article] [PubMed]
- 35.Martin JL, Hakim AD. Wrist Actigraphy. Chest. 2011;139(6):1514–27. Available from: 10.1378/chest.10-1872 [DOI] [PMC free article] [PubMed]
- 36.Buysse DJ, Reynolds CF, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric pratic and research. Vol. 28(2), Psychiatry Research. 1989. p. 193–213. Available from: https://www.psychiatry.pitt.edu/sites/default/files/inline-files/PSQI Article.pdf [DOI] [PubMed]
- 37.Augusta A, Cavalcante M, Priore SE, Do Carmo S, Franceschini C. Food consumption studies: general methodological aspects and its use in the evaluation of children and adolescents aged. Rev Bras Saúde Matern Infant. 2004;4(3):229–240. doi: 10.1590/S1519-38292004000300002. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. Legend: Sistema de Informações sobre Nascidos Vivos (SINASC)/ Information System on Live Births (SINASC)
Data Availability Statement
The data that support the findings of this study are available from e-mail rosangela.flb@ufma.br, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of Rosangela Fernandes Lucena Batista.