Abstract
HemoglobinA1c (HbA1c) is used to diagnose Diabetes mellitus and monitor glycemic control over the previous eight to twelve weeks in diabetic patients. Detection of HbA1c by cation exchange-high performance liquid chromatography (CE-HPLC) gives a chromatogram by which abnormal hemoglobin variants are also picked up. Some of these may interfere with HbA1c values affecting clinical management. Due to increased inter-state migration as well as medical tourism, there is a high possibility of finding various hemoglobin variants in any part of India. We did a prospective analysis over 1.5 years, of the hemoglobin variants detected during all the HbA1c runs. The HbA1c was tested on Bio-Rad D10 dual HbA2/F/A1c platform, which uses the CE-HPLC method. Every chromatogram was carefully studied to look for unknown peaks. The samples showing unknown peaks >6% were re-run in extended HbA2/F mode to categorize the hemoglobin variants. We had 9595 HbA1c samples, of which 70 cases showed a variant window. There were 40 males and 30 females, age ranging from 28 to 76 years. The different hemoglobin variants detected were HbD, HbE and HbS in heterozygous state, high HbF (with a differential diagnosis of HPFH heterozygous and delta-beta thalassemia heterozygous), HbE homozygous, HbQ heterozygous and HbJ heterozygous. We conclude that in the process of monitoring glycemic control using HbA1c, we can also pick-up hemoglobin variants. Hence, it is essential to review HbA1c graphs, so that the diagnosis of hemoglobin variants is not missed and the HbA1c reported is reliable.
Keywords: Chromatograms, Glycated hemoglobin (HbA1c), Serendipity, Hemoglobinopathy, Thalassemia
Introduction
Hemoglobin A1c (HbA1c) reflects the average plasma glucose over the previous eight to twelve weeks. A value of 6.5% or higher is diagnostic of diabetes mellitus (DM) [1]. National Glycohemoglobin Standardization Program (NGSP) has certified various methods such as iso-electric focusing, ion-exchange HPLC, boronate-affinity, immunoassay, enzymatic assay and capillary electrophoresis for the measurement of HbA1c [2]. Of these, ion-exchange HPLC and capillary electrophoresis have the advantage of graphical representation of each run. The graphs depict the various fractions of hemoglobins eluting at different retention times. In the process, additional pathology of hemoglobin variants is detected, which have clinical significance.
We performed a study to evaluate the incidence of such hemoglobin variants during HbA1c testing and categorized them.
Methods
We evaluated the chromatograms of all HbA1c tests done over a continuous 1.5-year period, in a hospital in South India. Repeat samples of patients on follow-up were excluded. HbA1c was tested by ion exchange—HPLC method on Bio-Rad D10 dual HbA2/F/A1c platform (Bio-Rad laboratories Inc., California, USA) with reportable range of 3.7–18.4% for HbA1c. In this equipment, samples can be run in two modes. The short program of 3 min (HbA1c mode) is exclusively for HbA1c quantification, while the extended program of 6.5 min (HbA2/F mode) is for β-thalassemia screening by quantification of HbA2 and HbF as well as for identification of other hemoglobin variants.
We prospectively reviewed the chromatograms of all the HbA1c tests (3 min run). Those samples having unknown peaks of >6% were further tested on the extended program (6.5 min run) to categorize the hemoglobin variants, based on the retention time and percentage of the variant peak. In these patients, the HbA1c values were reported only after confirming that the variant did not interfere with the HbA1c result. As per manufacturer guidelines, we did not report HbA1c values in patients with HbF and P3 peak of >10% and in homozygous and compound heterozygous cases. We also compared the HbA1c values in the two modes. The comparison was done using intra-class correlation (ICC).
Institutional review board approval was taken for this study.
Results
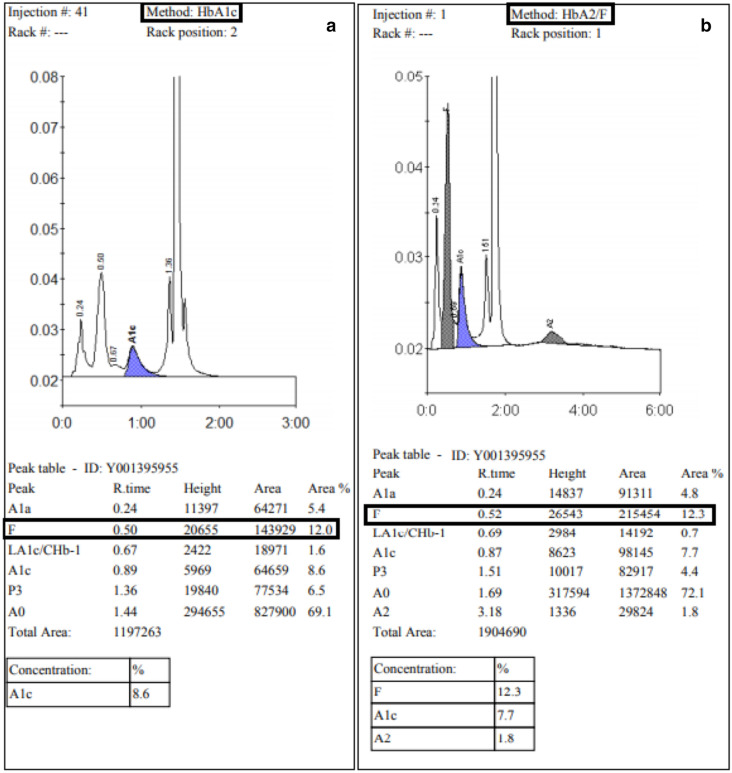
There were 9595 HbA1c samples; 70 cases (0.7%) showed a variant window (40 men and 30 women; aged 28–76 years; 60 Indians, 9 Africans and one West Asian). The HbA1c ranged from 4.1% to 15.6%. The variants were categorized on HbA2/F mode (Table 1). Examples of HbD (Fig. 1a and b) and HbF (Fig. 2a and b) heterozygous are shown. The reliability of measuring the HbA1c by short (A1c) and extended (A2/F) modes was found to be statistically significant (ICC = 0.9, p < 0.001).
Table 1.
Hemoglobin variants detected in patients tested for HbA1c
| Hemoglobin variants | No. of variants (n = 70) | % Among variants (n = 70) | % Among total HbA1c runs (n = 9595) |
|---|---|---|---|
| HbD heterozygous | 19 | 27.1 | 0.20 |
| HbE heterozygous | 18 | 25.7 | 0.19 |
| HbS heterozygous | 16 | 22.9 | 0.17 |
| High HbF | 10 | 14.3 | 0.10 |
| HbE homozygous | 4 | 5.7 | 0.04 |
| HbQ heterozygous | 1 | 1.4 | 0.01 |
| HbJ heterozygous | 1 | 1.4 | 0.01 |
| Compound heterozygous for HbD—β thalassemia | 1 | 1.4 | 0.01 |
Fig. 1.
(a and b) Variant window in HbA1c mode which was categorized as HbD heterozygous based on the percentage (39%) and retention time (3.97 min) on HbA2/F mode
Fig. 2.
(a and b) High HbF in HbA1c mode which was confirmed as HbF heterozygous (differential diagnosis of HPFH and δβ thalassemia heterozygous) based on the percentage (12.3%) in F window on HbA2/F mode
Discussion
Hemoglobinopathies are common in Southeast Asia, Mediterranean countries and Africa [3]. In India, the common hemoglobinopathies are β thalassemia, HbE, high HbF (with a differential diagnosis of HPFH and δβ-thalassemia trait), HbS and HbD. They are usually picked up based on RBC indices/screening programs for target population using HPLC or electrophoresis methods and confirmed by genetic testing. Hemoglobin variants can also be detected when diabetic patients undergo HbA1c testing [4, 5]. Bio-Rad D10 instrument uses HPLC method and gives a graph of each run, which should be reviewed for the presence of hemoglobin variant window before releasing the reports [6].
There are few case reports in indexed medical journals from India about hemoglobin variants detected during HbA1c run. These include HbQ [7], HPFH [8], HbD Iran-beta thalassemia [9], HbJ Meerut [10], HbE disease [11], Hb Hope [12, 13] and δβ-thalassemia trait [14]. However, ours is the largest series, where we have screened 9595 HbA1c runs and detected hemoglobin variants in 70 cases.
The most common variants we found were HbD, HbE and HbS in heterozygous states, similar to the distribution of hemoglobin variants in India [15–18]. The next common variant was high HbF [19, 20]. Hemoglobin variants are specific to certain ethnicities, who often inhabit a specific geographical area [15]. A review article on incidence of hemoglobinopathies by Balgir [16] in 2000 showed no cases of HbE in the south Indian state of Karnataka, whereas, our study done in the same state, shows that HbE is common. We suggest that this change in the distribution of cases in the past two decades could be due to inter-state migration. Hence, patients with hemoglobin variants may be seen in geographical areas, which were previously not known to harbor such variants. Further, more than half the patients with HbS trait in our study were non-Indians, due to medical tourism from Africa and West Asia.
Though β-thalassemia trait is the most common hemoglobinopathy in Indian population [20], it cannot be picked up in the HbA1c mode and hence there are no β-thalassemia cases in our study. However, one must suspect β-thalassemia trait when HbA1c does not correlate with the corresponding blood sugars. Such patients will have normal HbA1c graph, low MCV, Mentzer’s index <13 and normal serum iron profile. These cases should be further evaluated.
One should be aware that the HbA1c values of patients with hemoglobin variants in homozygous and compound heterozygous states are unreliable because of very low levels of adult hemoglobin (HbA). This could lead to misdiagnosis and mismanagement in patients with DM. Hence, HbA1c cannot be used for monitoring their glycemic control. Alternative non-hemoglobin methods such as fructosamine, glycated serum albumin or home glucose monitoring are recommended.
For variants in heterozygous state, interference in HbA1c estimation is dependent on the method used. Bio-Rad D10 instrument does not show an interference in heterozygous states, except for high HbF and P3 peak of >10% [21]. But these patients should be informed of the condition, since they need further evaluation to confirm the diagnosis and thus prevent homozygous or compound heterozygous states in the future generations. In our study, diagnosis was not confirmed by genetic testing due to financial constraints.
To summarize, in the process of monitoring glycemic control using HbA1c, we can also pick-up hemoglobin variants. Hence, it is essential to review HbA1c graphs, so that the diagnosis of hemoglobin variants is not missed and the HbA1c reported is reliable.
Acknowledgements
We thank Dr. Sanjay A. Pai and Dr. Rajeshwari Janakiraman for guidance and inputs during the study. We also thank Ms. Edna D’souza for technical expert advice and Mr. Shivaraj N S for statistical analysis.
Declarations
The material has not been published, submitted or accepted elsewhere.
Author contributions
JDK and Ss: Design, Data acquisition, Data analysis, Manuscript preparation, Manuscript editing, Manuscript review.
Funding
None.
Data Availability
Available.
Compliance with Ethical Standards
Conflict of interest
None.
Ethical Approval
Institutional review board approval taken.
Consent for Publication
All authors have consented for publication.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.American Diabetes Association. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2020. Diabetes Care. 2020 Jan;43(Supplement 1):S14-S31. [DOI] [PubMed]
- 2.List of NGSP Certified Methods, NGSP, 2020. Available from http://www.ngsp.org/docs/methods.pdf. Accessed on May 2020
- 3.Modell B, Darlison M. Global epidemiology of haemoglobin disorders and derived service indicators. Bull World Health Organ. 2008;86(6):480–487. doi: 10.2471/BLT.06.036673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Reeve J, Blake L, Griffin D, O'Shea P. Incidental detection of haemoglobin (Hb) variants during high performance liquid chromatography (HPLC) analysis of HbA1c: is it time for a standardised approach to reporting. Ir J Med Sci. 2015;184(2):353–355. doi: 10.1007/s11845-014-1118-z. [DOI] [PubMed] [Google Scholar]
- 5.Wilburn CR, Bernard DW, Zieske AW, Andrieni J, Miller T, Wang P. The prevalence and role of hemoglobin variants in biometric screening of a multiethnic population: one large health system’s experience. Am J Clin Pathol. 2017;147(6):589–595. doi: 10.1093/ajcp/aqx032. [DOI] [PubMed] [Google Scholar]
- 6.Lewis MR, Macauley RC, Sheehan PR, Staten MA, Phillips LS, Rasouli N, et al. Management of hemoglobin variants detected incidentally in HbA1c testing: a common problem currently lacking a standard approach. Diabetes Care. 2017;40(2):e8–e9. doi: 10.2337/dc16-1667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nigam PK, Sharma S, Sareen R, Paul V, Lal A. Lack of precision in HbA(1c) values on variant II in cases of hemoglobin Q India. Indian J Clin Biochem. 2006;21(2):72–75. doi: 10.1007/BF02912916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bhat VS, Dewan KK, Krishnaswamy PR. Diagnostic dilemma of HbA1c detection in presence of a hemoglobinopathy: a case report. Indian J Clin Biochem. 2011;26(1):91–95. doi: 10.1007/s12291-010-0076-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bhat VS, Mandal AK, Mathew B. Co-inheritance of HbD (Iran)/beta thalassemia IVS1-5 (G > C) trait in a Punjabi lady with diabetes. Indian J Clin Biochem. 2012;27(2):202–206. doi: 10.1007/s12291-012-0201-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sharma A, Marwah S, Buxi G, Yadav RB. Falsely low HbA1c value due to a rare hemoglobin variant (HEMOGLOBIN J-MEERUT) – a family study. Indian J Pathol Microbiol. 2012;55(2):270–271. doi: 10.4103/0377-4929.97912. [DOI] [PubMed] [Google Scholar]
- 11.Yedla N, Kuchay MS, Mithal A. Hemoglobin E disease and glycosylated hemoglobin. Indian J Endocrinol Metab. 2015;19(5):683–685. doi: 10.4103/2230-8210.163211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Singh B, Behera DD, Mehta N, Das S. Hb hope: a rare variant of haemoglobin-forged a rise in HbA1c on immunoturbidimetric assay. Indian J Clin Biochem. 2014;29(4):517–519. doi: 10.1007/s12291-013-0406-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chakraborty S, Chanda D, Gain M, Krishnan P. Interference of the hope hemoglobin with hemoglobin A1c results. Lab Med. 2015;46(3):221–225. doi: 10.1309/LME82XNY6SYVWDYQ. [DOI] [PubMed] [Google Scholar]
- 14.Kumar BV, Choccalingam C, Samuel P. Incidental identification of possible delta-beta thalassemia trait in a family: a rare cause of elevated Hb F. J Clin Diagn Res. 2016;10(3):BD01–BD02. doi: 10.7860/JCDR/2016/16352.7409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mohanty D, Colah RB, Gorakshakar AC, Patel RZ, Master DC, Mahanta J, et al. Prevalence of β-thalassemia and other haemoglobinopathies in six cities in India: a multicentre study. J Community Genet. 2013;4(1):33–42. doi: 10.1007/s12687-012-0114-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Balgir RS. The burden of hemoglobinopathies in India and challenges ahead. Curr Sci. 2000;79(11):1536–1547. [Google Scholar]
- 17.Mondal SK, Mandal S. Prevalence of thalassemia and hemoglobinopathy in eastern India: a 10-year high-performance liquid chromatography study of 119,336 cases. Asian J Transfus Sci. 2016;10(1):105–110. doi: 10.4103/0973-6247.175424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chandrashekar V, Soni M. Hemoglobin disorders in South India. ISRN Hematol. 2011;2011:748939. doi: 10.5402/2011/748939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rao S, Kar R, Gupta SK, Chopra A, Saxena R. Spectrum of haemoglobinopathies diagnosed by cation exchange-HPLC & modulating effects of nutritional deficiency anaemias from north India. Indian J Med Res. 2010;132:513–519. doi: 10.4103/0971-5916.73390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sachdev R, Dam AR, Tyagi G. Detection of Hb variants and hemoglobinopathies in Indian population using HPLC: report of 2600 cases. Indian J Pathol Microbiol. 2010;53(1):57–62. doi: 10.4103/0377-4929.59185. [DOI] [PubMed] [Google Scholar]
- 21.Little RR, Roberts WL. A review of variant hemoglobins interfering with hemoglobin A1c measurement. J Diabetes Sci Technol. 2009;3(3):446–451. doi: 10.1177/193229680900300307. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Available.