Abstract
The scientific community has been alarmed by the possible immunological evasion, higher infectivity, and severity of disease caused by the newest variants of SARS-CoV-2. The spike protein has an important role in the cellular invasion of viruses and is the target of several vaccines and therapeutic resources, such as monoclonal antibodies. In addition, some of the most relevant mutations in the different variants are on the spike (S) protein gene sequence that leads to structural alterations in the predicted protein, thus causing concern about the protection mediated by vaccines against these new strains. The present review highlights the most recent knowledge about COVID-19 and vaccines, emphasizing the different spike protein structures of SARS-CoV-2 and updating the reader about the emerging viral variants and their classifications, the more common viral mutations described and their distribution in Brazil. It also compiles a table with the most recent knowledge about all of the Omicron spike mutations.
Keywords: Brazil, COVID-19, Public health, SARS-CoV-2, Variants, Viral diseases
Introduction
The present report highlights recent knowledge about the different variants of the causative agent of coronavirus disease 2019 (COVID-19), severe acute respiratory syndrome-related coronavirus 2 (SARS-CoV-2), throughout 2021, with an emphasis on those variants that could further compromise the critical public health scenario in different parts of the globe. The focus of the present report is (1) the multiple structural features of the spike (S) protein, invariably present in the capsid of all viral variants; (2) understanding how these variations can potentially decrease the protection offered by vaccines and therapeutic measures, such as monoclonal antibodies; and (3) highlighting the most recent knowledge about Omicron’s mutations.
COVID-19
COVID-19, a disease caused by the new severe acute respiratory disease 2 (SARS-CoV-2), was first reported as a case of pneumonia in December 2019 in Wuhan, China; since then, it has progressed from a local outbreak to an unprecedented pandemic. Two other insurgences in the Coronaviridae family have occurred in the last two decades: severe acute respiratory disease (SARS), caused by SARS-CoV in 2002 [1], and Middle East respiratory syndrome (MERS), caused by MERS-CoV in 2012 [2, 3]. Even though the mortality rate decreased [4] when compared with the previous viruses, the virus that emerged in 2019 showed a major increase in infectivity [2, 4, 5].
Past vaccine knowledge and advances in technology have helped to accelerate the already well-established process of vaccine development [6]. The response to the disease outbreak was similar to that which followed the 2009–2010 H1N1 pandemic, for instance, but the number of vaccine developers was 30–50 times greater [6], and the technology level had been developing over the past 10 years. The funding allocated worldwide for COVID-19 testing and vaccination is unprecedented, gathering several billion dollars to all stages of development [7]. Additionally, even though cooperation between governmental funders and research institutes or international alliances was previously seen in other vaccines, COVID-19 exceeded previous cooperation levels, leading to rapid information sharing among public and private partnerships, providing risk sharing, and improving the efficiency of resource use [7]. Nonetheless, at the final stages of development, the high number of infected people helped to show the effectiveness of the vaccines, which is challenging with diseases of low prevalence. The clinical trials were not reduced [6], and some efficacy and safety studies for COVID-19 vaccines included more than 40,000 subjects [8–15]. In contrast, other approved vaccines, such as Haemophilus influenzae [16], had approximately 5000 subjects, varicella vaccines [17] had approximately 1000 and hepatitis A had approximately 40,000 subjects [18, 19]. Fast regulatory review and approval processes were important, as the criteria of approval in multiple countries were the same, and several documents were issued to clarify the licensure pathway [6].
Fast information sharing has been strongly discussed by scientists in recent years, since it helps researchers gather information quickly, and the results of health care and public policy are widespread in this urgent time. However, it might also cause the spread of biased data and questionable results without peer review [7]. Even though the scientific community might understand the importance of taking precautions with this kind of information and checking for further publication of results, the mainstream and social media might interpret them as generating fake news [20, 21].
The symptoms of this infection may vary between inapparent or mild cases of respiratory insufficiency and sepsis, with the major symptoms including fever, dry cough, and difficulty breathing [22–24]. Additionally, host genetics might be key to understanding the high dissimilarity of symptoms between patients [25]. The disease relates to an aggressive immune response that may damage the airways. As a result, severe cases must consider controlling the infection itself as well as managing the host response, since it might evolve into a cytokine storm, which leads to multiple organ failure and death [26]. Nonetheless, minimizing secondary bacterial or fungal infections is also very important [26]. Disease transmission occurs mainly through contact with contaminated people. The incubation period can range between 0 and 14 days after virus contact and is usually between 5 and 6 days [27–29]. /The viral load is usually high at the beginning of symptoms but is still similar in patients with or without symptoms, suggesting that even asymptomatic patients can be contagious, emphasizing the relevance of preventive measures [30].
Host genetics may be important for understanding the large range of symptoms between patients, but researchers [2] have also reported several host genes that are critical to viral infection, such as ACE2, RAB7A and four members of the ARP2/3 complex, which are crucial for attachment and endocytosis, or CTSL and 13 vacuolar-ATPase proton pumps, which are important for spike protein cleavage and viral membrane fusion. From invasion to the exit of the infected cell, they are mainly involved in the initial binding, endocytosis and cleavage of the spike protein but also play roles in fusion with the viral membrane, endosome, Golgi apparatus, and transcriptional modulators. These genes are important regardless of viral load and do not strongly depend on the cell type or tissue. Nonetheless, among the main genes found, only the ACE2 receptor demonstrated tissue-specific expression, with increased expression in the testicles, small intestine, kidneys, and heart. This expression might help to explain the viral tropism (as S interacts primarily with ACE2), and more studies should be performed to better understand the potential damage caused by COVID-19 in these tissues.
Some evidence has shown the possibility of long-term, nonproductive persistence of SARS-CoV-2 infection in tissues, such as epithelial, myeloid, and neural cells [31]. The persistence might also be related to mutational events in the virus, as related by a study analyzing a variant (GZ69) isolated from an asymptomatic individual who reported an unprecedented capability of replication in Vero E6 cells in the absence of any evident cytopathic effect. This strain might favor cell survival and, eventually, viral persistence due to a mutation in residues 203 and 204 in the N protein [32]. This evidence, along with others [33–35], suggests that prolonged viral shedding and viral persistence might be present in COVID-19.
A nasopharyngeal or oropharyngeal swab followed by quantitative real-time reverse transcriptase-polymerase chain reaction (qRT–PCR) is the standard procedure for diagnosis [36–40]. Some immunochromatography assays and other technologies, such as reverse transcription loop-mediated isothermal amplification (RT-LAMP) [41–43], can be used as complements.
According to data published by the World Health Organization (WHO) in February 25, 2022 [44], 146 vaccines are in clinical development. Of these, 33% use a protein subunit platform, and 24% use RNA or mRNA to elicit an immune response, a great number of which rely on the form or sequence of the spike protein of SARS-CoV-2, including the Pfizer/BioNTech® and Moderna® vaccines [45, 46].
SARS-CoV-2 biology
SARS-CoV-2 is an enveloped virus with a viral particle of 60–140 nm [47, 48] in diameter with a single positive-strand RNA (( +) ssRNA) of approximately 26–33 kb in length [49], and it may depend on host factors at all stages of its viral cycle. It belongs to the order Nidovirales, suborder Cornidovirineae, family Coronaviridae, subfamily Orthocoronavirinae, genus Betacoronavirus, and subgenus Sarbecovirus [50]. The coronavirus family is so named thanks to a spiculated glycoprotein membrane that resembles a solar corona [51].
The virus has four main structural proteins: spike protein (S), membrane (M), envelope (E), and nucleocapsid protein (N) [52]. The M protein is the most abundant and, in addition to defining the shape of the viral envelope, anchors the envelope to the nucleocapsid [52, 53]. Protein E, which is also the smallest structural protein, has viroporin action and forms an ionic channel [52, 54]. Both E and M proteins are essential to the assembly and release of the virion of host cells[52]. The N protein is the only protein that binds directly to viral RNA and has a high diagnostic value [52, 55].
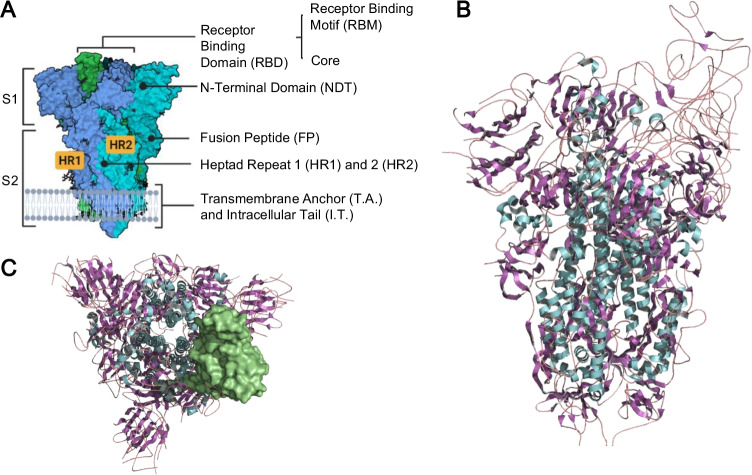
The spike protein (S) shapes trimers in the viral envelope and has a crucial function in fusion with the host cell and viral pathogenicity. Its N-terminal part has an external globular domain called S1, where the receptor-binding domain (RBD) is also located. The C-terminal part, called S2, forms the spicule rod and includes the membrane fusion peptide (FP) [49, 52]. The structure of the spike protein of a D614G mutant at pH 5.5 (PDB: 6XM0) is shown in Fig. 1, constructed with PyMOL 2.5.2® (The PyMOL Molecular Graphics System [56]) and Biorender® [57]. Different from other external fusion proteins, the SARS-CoV-2 spike protein is very flexible and articulated in at least three points, improving the scanning capacity over the human cell and multiple binding sites [58].
Fig. 1.
A Side view of the demarcating protein S2 and S1 domains and subdomains. B SARS-CoV-2 spike protein structure of the D614G mutant viral strain (PDB: 6XM0) constructed through PyMOL® and BioRender. The colors refer to the secondary structures: alpha-helix in cyan, beta sheets in purple and turns in magenta. C Top view of the trimeric protein with the receptor binding domain (RBD) demarcated on the surface
The receptor binding domain (RBD), located in the S1 part of protein S, has approximately 220 residues and is stabilized by eight disulfide bridges and two N-glycosylation sites (N331 and N343). It has a potential role in protein folding, dynamics, stability, and accessibility to the receptor and is composed mainly of random structures (35.6%) and β-sheets (33%), followed by turns (19.1%) and alpha helices (12.4%) [59].
The RBD is also subdivided into two subdomains: the core, rich in nonpolar residues, and the receptor binding motif (RBM), corresponding to residues 443–503 of the S protein, which is mostly polar. Specifically, the RBM mediates angiotensin converting enzyme-2 (ACE2) receptor binding and must maintain sufficient affinity with the receptor for entry into the host cell to be effective [59]; however, this region is highly targeted by neutralizing antibodies, presenting itself as a crucial location for mutations that promote viral immune evasion. It has been demonstrated [60] that RBM might have variability and a high degree of plasticity, supporting variations in its sequence without losing the ability to connect with ACE2. Additionally, A475 within RBM seems to be a crucial residue for the connection between S and ACE-2 [61].
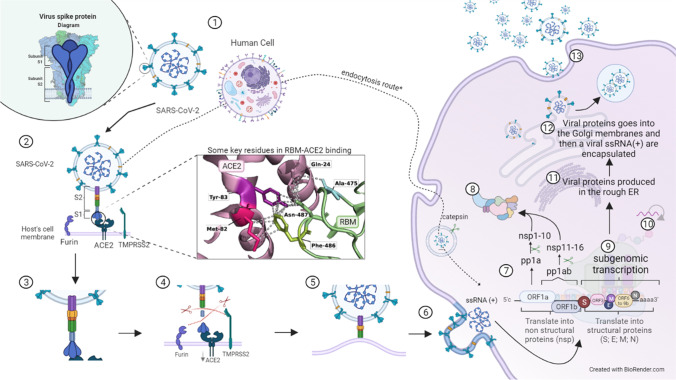
The viral entry process and the replication mechanism of SARS-CoV-2 will be described in the next paragraphs. Figure 2 provides a visual guide and might be helpful to better understand the process.
Fig. 2.
Role of spike protein in the SARS-CoV-2 infection mechanism. 1 The S protein is used by SARS-CoV-2 to interact with the host cell receptor. The host cell has several different receptors and polysaccharides in its membranes. In this step-by-step figure of the cycle, we focus on the major receptor used, which is the ACE2 receptor, but SARS-CoV-2 may also interact with other cell receptors. 2 Protein S attacks its target cells: the RBM (in RBD) interacts with the ACE2 receptor. In this process, A475 and F486 in the RBM seem to be the key residues to connect. In this step, it is important to note 2 additional receptors important for viral entry: furin and TMPRSS2. 3 Interaction between HR1 and HR2: the connection causes conformational changes in S2, where HR1 and HR2 motifs interact to start the formation of the six-helix bundle. 4 Proteolytic activation of spike: after S-ACE2 binding, proteolytic activation of spike occurs when the additional receptors cleave the protein, exposing the fusion peptide (FP). While furin cleaves the polybasic cleavage site (PRRAR) between S1 and S2, TMPRSS2 cleaves an S2 site. 5 Approximation between viral and host membranes: the FP approximates the viral and host membranes together with the six-helix bundle, leading to fusion and viral entry. 6 Fusion and viral entry: fusion of the membranes releases the viral RNA into the cytoplasm of the host cell. 7 Translation of replicase genes in the viral RNA genome: since the RNA of SARS-CoV-2 is a + ssRNA, it can be translated directly into polyproteins: ORFa will be translated in pp1a, which will be further cleaved in nspn 1 to 10, and by a − 1 ribosomal frame shifting, ORF1a and ORF1b will translate to pp1b, which will generate nsps 11 to 16. 8 Replicase-transcriptase complex: the viral RNA-dependent RNA polymerase (RdRp), composed of nsp12, nsp7, and 8 along with other nsps, condenses and assembles in the rough endoplasmic reticulum (ER) membrane-bound ribonucleoprotein complex, the so-called replicase-transcriptase complex (RTC) that directs replication, transcription, and maturation of the viral genome and subgenomic mRNAs. 9 Subgenomic transcription: the direct translation of the viral genome produces several ( −) subgenomic RNAs (sgRNAs) from the structural protein genes and ORF3 and ORF6 to 9, which are synthesized in a full-length negative strand RNA combining varying lengths of the 3′ end of the genome with the 5′ leader sequence. 10 Replication: the full-length ( −) RNA is also used as a mold to replicate the ( +) ssRNA of the virus that will form the new virion. 11 Structural protein synthesis: the ( −) sgRNA is used as a mold to synthesize subgenomic ( +) mRNAs, which are then translated into M, S, E, and N proteins and processed as needed. 12 Encapsulation: after translation, the structural proteins are inserted into the endoplasmic reticulum and continue to their intermediate compartment, where the replicated ( +) ssRNA will interact with N protein, forming the nucleocapsid that will be enveloped in the ER-Golgi intermediate. 13 Transport and exocytosis: the virions are transported by vesicles and released by exocytosis
Protein S attacks target cells, such as nasal epithelial cells, bronchial cells, and pneumocytes, by binding to ACE2. The RBM-ACE2 connection causes proteolytic activation of the S protein, exposing the fusion peptide and leading to the adoption of a more favorable conformation with epistatic regions, leading to viral entry into the host cell. This cleavage between S1 and S2 is usually performed by the TMPRSS2 protein, another protease that causes protein-dependent proteolysis of S or through the furin cleavage site or polybasic cleavage site (PRRAR) [23, 25, 49, 52, 62, 63]. Both ACE2 and TMPRSS2 have been studied as molecular markers that confer genetic susceptibility or resistance [25].
This furin site, a single Arg in other coronaviruses, has an unusual sequence of amino acids between S1 and S2 in SAR-CoV-2, composed of Pro-Arg-Arg-Ala-Arg (Pro: proline; Arg: Arginine; Ala: Alanine). The cleavage of this sequence is important for the efficient uptake of lung cells [58, 64]. SARS-CoV-2 virus without this site seems to be less infective [62, 65], and some variations in this sequence seem to improve viral transmissibility, as seen in the alpha and delta variants [58]. In alpha, proline is changed to histidine (P681H), and in delta, proline is changed to arginine (P681R). Both alterations make the sequence more basic, improving recognition by furin. This furin cleavage primes the newly made S protein (and consequently the virion) to be more effective at infecting host cells. For instance, for SARS-CoV, these primed proteins represent only 10%, while for the SARS-CoV-2 alpha variant, they represent more than 50%, and for the Delta variant, they represent more than 75% [58].
After binding ACE2, two domains interact within the S2 subunit: heptad repeats 1 and 2 (HR1 and HR2). These domains are structural motifs characterized by seven amino acids in a specific configuration (hydrophobic-polar-polar-hydrophobic-charged-polar-charged) that prone them to form alpha helices. They form a six-helix bundle responsible for approximating the viral and host membranes, leading to fusion and viral entry [66]. Following fusion, the inner contents of the virus are released into the cytoplasm of the host cell. Then, replicase genes are translated into the viral RNA genome.
As SARS-CoV-2 has a ( +) ssRNA, it can act as an mRNA and be directly translated into the ribosome. The first two open reading frames (ORFs) in the viral genome, called ORF1a and ORF1b, are immediately translated into two polyproteins that then undergo proteolysis into 16 nonstructural proteins (nspns). Polyprotein 1a (pp1a) originates from the translation of ORF1a and is cleaved in nspn 1 to 10, while polyprotein 1ab (pp1ab) is encoded by ORF1a and ORF1b through a − 1 ribosomal frame-shift mechanism and is then cleaved in nsps 11 to 16.
SARS-CoV-2 has two cysteine proteases [67] encoded by ORF1a: one papain-like protease (PLpro or nsp3) that cleaves pp1a at three sites liberating nspn1-4 and the main protease (Mpro or nsp5), sometimes referred to as 3Clpro for having a chymotrypsin-like fold, which cleaves both polyproteins liberating nsps4-16 [67, 68]. After this process, the viral RNA-dependent RNA polymerase (RdRp), composed of nsp12, nsp7, and 8 along with other nsps, condenses and assembles in the rough endoplasmic reticulum (ER) membrane-bound ribonucleoprotein complex, the so-called replicase-transcriptase complex (RTC), which directs replication, transcription, and maturation of the viral genome and subgenomic mRNAs [52, 67–70].
One important viral protein in this early step is Nsp1, which blocks host mRNA entry in ribosomes, undermining translation by approximately 70%, and also induces selective endonucleolytic cleavage and degradation of host mRNAs, especially cytosolic mRNAs, by binding to the 40S ribosome subunit. Thus, this protein helps to accelerate cell turnover, as the remaining translation activity is used by the virus. Another important action of this protein is to jam up nuclear exit channels, inhibiting nuclear alerts to the immune system and leading to a lower secretion of interferons [58, 71].
The structural proteins of SARS-CoV-2, however, are produced through a more complex process, performed by the RTC, of discontinuous transcription and then translation into proteins. In this process, translation of the ( +) ssRNA of the virus produces several ( −) subgenomic RNAs (sgRNAs) from the structural protein’s genes and ORFs 3 and ORF6 to 9, which are synthesized in a full-length negative strand RNA combining varying lengths of the 3′ end of the genome with the 5′ leader sequence. Then, this ( −) sgRNA is used as a mold to synthesize subgenomic ( +) mRNAs, which are then translated into M, S, E, and N proteins and processed as needed [52, 69, 72]. Full-length ( −) RNA is also used as a model for replication of the ( +) ssRNA of the virus that will form the new virion [69] (Fig. 2).
After replication and synthesis of subgenomic RNA, structural proteins are translated and inserted into the endoplasmic reticulum and continue to an intermediate compartment, where the replicated ( +) ssRNA will interact with the N protein. This forms the nucleocapsid that will be enveloped in the ER-Golgi intermediate, and vesicles containing the virion will bind to the membrane surface, resulting in the release of the virus [69]. Protein M directs most of the protein–protein interactions involved in viral assembly; however, the presence of protein E is also essential to produce the viral envelope, in addition to being related to the induction of membrane curvature and in preventing the formation of aggregates by M protein. After assembly, the virions are transported by vesicles and released by exocytosis [52, 69].
Additionally, infection by SARS-CoV-2 can induce syncytia [73, 74], as seen in MERS-CoV[75] and with some evidence in SARS-CoV [76]. In this mechanism, S proteins shown on the surface of the infected host cell activate a host cell calcium-ion channel, leading to the production and secretion of a fatty coating that leads to fusion with nearby cells expressing ACE2. This multinuclear structure may allow longer survival of the infected cell. SARS-CoV-2 is even able to form this structure with lymphocytes, important immune cells, similar to what is seen in tumor cells, which also helps the virus avoid host immune detection [58].
Most of the sequence variations reported in the new strains are single nucleotide polymorphisms or variants, along with other point mutations. In addition, some reports have suggested that variations in the virus genome might occur following infection, so-called intrahost mutations [25, 77–79].
The arrival of new viral variants has worsened the scenario. Viruses change constantly, and these mutations might result in new variants, some of which may have mutations that could allow them to spread more easily or increase disease severity. The RNA replication of SARS-CoV-2 has a moderate intrinsic error rate [60], as shown by the small number of mutations that reach high frequency between sequenced genomes. Nevertheless, the high number of infected individuals and the range of susceptible hosts increase the risk of new variants that could directly impact vaccines and therapeutic tools [60]. Additionally, coronaviruses possess a NSP14 exoribonuclease, which has a repair proofreading function [25].
The spike protein (S) of SARS-CoV-2: clinical interest and new viral variants
Due to its critical role in host invasion, the spike protein (S) of the virus has been the target of numerous studies, many related to possible therapeutic targets, whether by blocking binding with the receptor [60], DNA vaccines [80] or RNA based on the S protein sequence [46, 81], among others. However, there has been an increasing number of variants with divergences in their S protein sequences that can affect the efficiency of therapeutic measures based on transmission rates and disease severity [82, 83].
In addition, some authors [60] have indicated that, depending on the variants, long-term control of the pandemic will require systematic monitoring and complementary preparations for circulating strains. GISAID, which refers to the Global Initiative on Sharing Avian Influenza Data, has also monitored SARS-CoV-2 variants through its database of gene sequences since the beginning of the COVID-19 pandemic [82, 84–86]. The WHO also regularly monitors new variants and their potential impact on transmissibility; severity; and resistance to therapeutic tools, vaccines, and diagnosis to assess the risk they impose on public health. Weekly update reports are made available on the organization’s website and aim to assess the overall state of the pandemic worldwide [82, 87, 88].
The CDC-US [89] and WHO[90] have proposed classifying the variants into four classes based on the main strains in circulation in the USA and globally: variants of high consequence (VOHC), variants of concern (VOC), variants of interest (VOI), and variants being monitored (VBM). As it is not restricted to the strains circulating in the USA, the current WHO classification of the variants will be adopted [87, 90].
In the first stage, we include VOHC, a level for lineages with a proven advantage over preventative measures or medical treatments over previously circulating strains [89]. The measurements considered are the proof of failure in diagnosis or a decrease in vaccination effectiveness, a disproportional number of infections in vaccinated individuals, or low vaccine-induced protection for critical conditions, as well as a more severe prognosis and a spike in the proportion of hospitalizations. Neither the CDC-US nor WHO reported any variants in the VOHC category as of March 2022.
The VBM list, in contrast, involves variants that have genetic mutations that are suspected to affect virus characteristics, with some indication that they may pose a future risk, but for which evidence is still unclear and needs further monitoring and assessment [90]. By March 1, 2022, only the lineages B.1.640, C.1.2, and B.1.1.318 are on the VBM list.
Variants of interest (VOIs) have specific genetic markers associated with changes in receptor binding, mainly mediated by S protein, considerable potential for avoidance of neutralizing antibodies generated by previous infection or vaccination and a reduction in the efficacy of available treatments, a potential impact on diagnosis, or the possibility of increased transmissibility or severity of the disease [89]. Only the strains Lambda (C.37) and Mu (B.1.621) are currently (March 1, 2022) classified as VOI by the WHO [90].
The Mu variant seems to be highly resistant to COVID-19 convalescent sera, even more so than beta or delta, potentially induced by YY144-145TSN (mutation of two Y (tyrosine) by one T (threonine) and S (serine) in positions 144 and 145, with a genetic insertion of an N (asparagine) in position 146) and E484K mutations in the spike protein. Additionally, Mu-infected individual serum has been shown to be resistant to Mu as well as other variants [91].
Lambda has one deletion (del246-252) and seven nonsynonymous mutations in S (G75V, T76I, D253N, L452Q, F490S, D614G, T859N) [92]. Of these, F491S showed some evasion capacity of antibody neutralization in vitro [93–95] and seems to be highly infectious, characteristics associated mainly with T76I and L452Q [96].
On the other hand, VOC characterizes variants with evidence of increased transmissibility and severity of disease course (increase in hospitalization or deaths), a significant reduction in the efficiency of neutralizing antibodies, a decrease in the effectiveness of treatments or vaccination, and failures or interferences in diagnostic targets.
The following lineages are considered VOC by WHO as of March 1, 2022, with the correspondent’s name in order of Pango lineage and WHO label (a summary of data can be found in Table 1):
B.1.1.7 or Alpha: detected in the UK on 20 September 2020, with a high number of mutations, including 8 in the S protein. The main [82, 88] mutations in protein S are H69/V70 and N501Y changes, A570D, and P681H, according to the weekly report of 16 March 2021 from WHO. The strain, already found in 118 [88] countries, has a known 50% increase in viral transmission and a potential increase in disease severity but no to minimal impacts on monoclonal antibody therapies or neutralizing antibodies in convalescent serum or postvaccination [82, 88, 89, 97–99]. Recent data suggest that individuals infected by this strain are, on average, 61% more likely to die, based on 1,146,534 patients in community tests [100].
P.1 or B.1.1.28.1 or Gamma: detected in Japan in December 2020, but based on data in Brazil, has a moderate impact on therapies based on monoclonal antibodies and a reduction in the neutralizing power of convalescent sera and postvaccination [89]. The main mutations highlighted in the recent WHO [88] report are the changes K417T, E484K, and N501Y. It is currently found in 38 countries.
B.1.351 or Beta: the first report of this strain used to be considered early August 2020 in South Africa and has already been reported in 64 countries [88], but the WHO now considers that this strain emerged in South Africa in May 2020. This variant might have a 50% increase in its transmission rate [101], a moderate impact on monoclonal antibody therapies [73, 102], especially to the combination of Bamlanivimab® and Etesevimab® [102], and reduced neutralization by convalescent and postvaccination antibodies [73, 102].
B.1.617.2 or Delta: it was first reported in India. It has at least nine mutations in the S protein [25] This variant has higher transmissibility and a potential reduction in neutralization postvaccination and against some of the monoclonal antibody treatments currently used in the USA. Two strains were initially detected, AY.1 and AY.2, but now they are both considered under B.1.617.2. Another study showed that the sensitivity to vaccines in this strain is only modestly different than that of the alpha variant after complete vaccination (double- or single-dose vaccines) [103]. Another study, however, showed lower protection when compared with the alpha strain. In this study, Delta was resistant to some anti-NTD and anti-RBD monoclonal antibodies, the sera of convalescent individuals after 12 months were fourfold less potent against this variant, and the sera of individuals immunized with one dose of Pfizer® or AstraZeneca® were barely inhibitory to the strain. However, they improved with a second dose, being neutralized in 95% of the individuals, but with titers at least threefold lower than the alpha variant [104]
B.1.1.529 or Omicron: it was first reported in South Africa. It has at least 32 mutations only on the S protein, which is markedly different from other strains. Initial data show that this variant has a potential increase in transmissibility and a potential reduction by some monoclonal antibody treatments and postvaccination sera [89]. A genome-based phylogeny and mutational analysis, currently in preprint, concluded that this variant shares a common ancestry with the VOI lambda [105].
Table 1.
WHO’S classification of SARS-CoV-2 Variants of Concern (March 2022): Who label, Pango lineage, local of origin, quantification and descriptions of spike mutations and important characteristics highlighted in the literature
| WHO label | Pango lineage | Origin [90] | Number of mutations in spike | Spike mutations | Important characteristics |
|---|---|---|---|---|---|
| Beta | B.1.351 | South Africa on May 2020 | 10 [106] | D80A, D215G, 241del, 242del, 243del, K417N, E484K, N501Y, D614G, A701V |
- Enhanced transmissivity and higher mortality rates [107–109] - Evidence of non-protection against mild-to-moderate COVID-19 with two-doses of ChAdOx1 nCoV-19 vaccine/AstraZeneca-Oxford (multicenter, double-blind, randomized trial, n = 2026) [110] - Further evidence of immune escape was founded [102, 111, 112], and the infection by this variant might confer potent lineage-specifical and cross-reactive antibodies [113] |
| Alpha | B.1.1.7 | UK on 20 September 2020 | 13 [106] | 69del, 70del, 144del, E484K, S494P, N501Y, A570D, D614G, P681H, T716I, S982A, D1118H, K1191N | |
| Delta | B.1.617.2 | India on October 2020 | 15 [89] | T19R, V70Fa, T95I, G142D, E156-, F157-, R158G, A222Va, W258La, K417Na, L452R, T478K, D614G, P681R, D950N |
- Increased transmissibility [116] - Immune evasion of response triggered by infection and vaccination, and also Bamlanivimab® [104, 117–119] - It might have a lower reduction in transmission after vaccination, compared to Alpha [120] - Some evidence of similar prevalence between vaccinated and unvaccinated groups (n = 539) [121] |
| Gamma | P.1 or B.1.1.28.1 | Brazil on November 2020 | 11 [106] | L18F, T20N, P26S, D138Y, R190S, K417T, E484K, N501Y, D614G, H655Y, T1027I, | -Resistant to multiple neutralizing mAbs, convalescent plasma, and vaccinee sera [118, 122–124] |
| Omicron | B.1.1.529 | South Africa on November 2021 | 34 [89] | A67V, del69-70, T95I, del142-144, Y145D, del211, L212I, ins214EPE, G339D, S371L, S373P, S375F, K417N, N440K, G446S, S477N, T478K, E484A, Q493R, G496S, Q498R, N501Y, Y505H, T547K, D614G, H655Y, N679K, P681H, N764K, D796Y, N856K, Q954H, N969K, L981F |
- Potential of increased transmissibility [89] -Potential of reduction in neutralization by some EUA monoclonal antibody treatments [89] - Potential of reduction in neutralization by post-vaccination sera [89] Omicron’s mutation characteristics are further discussed in Tables 2, 3 and 4 |
aMight be detected in some sequences but not all
Interestingly, most VOCs have the same additions in nonstructural protein 6 (NSP6), a potential scaffold transmembrane protein S106, G107, and F108 [52], and all variants monitored have at least one common mutation: the alteration of a negatively charged aspartate by a glycine at position 614 of the spike protein [8, 83, 86, 125–127].
In the sequence, we will describe important mutations frequently described in the literature.
D614G
One month after being identified in March and April 2020, D614G became the most prevalent mutation worldwide due to positive selection [63, 83, 86, 128]. Travelers may have played a major role in introducing the mutation in several locations. Another hypothesis [63] is that the genetic variability in ACE2 expression may play a role in the greater success of this mutation, since its expression is significantly higher in Asian populations than in European, miscegenated American and African populations, which show lower expression of the receptor. A correlation analysis showed a significant positive relationship between ACE2 expression and the prevalence of D614, which could explain the higher prevalence of the wild-type strain in the Asian population and the rapid spread of the G614 variant throughout the globe, since it might lead to greater transmission in populations with lower ACE2 expression [22].
Infections by strains with this mutation are associated with a greater number of nucleic acids found in the upper respiratory tract of patients, suggesting greater viral load and infectivity, which may suggest the need for higher antibody titers for protection [8, 86, 129]. G614 is also transmitted significantly faster than the wild strain between hamsters through aerosols and droplets [130].
However, no evidence has linked this mutation to the greater severity of the disease, data also supported by other authors [8, 86], nor is there any difference between the sensitivity between the two strains (D614 and G614) against monoclonal antibodies [8, 127] or convalescent serum [130].
Although one study [8] indicated that it decreases affinity for ACE2, the main SARS-CoV-2 receptor, increasing the dissociation rate by 4 × and decreasing binding affinity by 5.7 × at 25 °C in relation to the D614 strain, the higher G614 infectivity could not be explained by increased protein–receptor affinity. Other authors [127] found an increase of 1.5 to 2 × in affinity between the mutated protein and ACE2 receptor, also based on the dissociation rate, at 30 °C and 37 °C, suggesting that the mutation increased affinity. Ozono et al. [127], the authors of the second experiment, indicated that the difference between the results could have occurred due to the use of different S proteins and that further studies should be performed.
Additionally, structural changes seem to favor the G614 mutation: the exchange for glycine at position 614 leads to the breakdown of a usual bond between D614 in the S1 part of the protein and T859 in the S2 part. This leads to an important conformational change, increasing the proportion of proteins in the open conformation (greater interaction with ACE2) in relation to the closed conformation, from 18% open to 58% in proteins with the G614 mutation. Additionally, other intermediate states were found in this case: open-2 conformation (~ 39% of the observed forms) and fully open state (~ 20%), which also favors the connection to the ACE2 receptor [8]. The highest efficiency of entry into the host cell by strains with this mutation was also found by [128, 130] and Ozono et al. [127].
N439K and N439R
Two recurrent mutations in the RBM region, N439K and N439R, showed a 2 × increase in ACE2 binding affinity compared to the wild strain (N439) [60]. The N439K variants seem to be more successful since they appeared independently in multiple lineages due to convergent evolution and rapidly reached numerous countries. This mutation, although unrelated to the severity of disease, enables immune evasion, conferring resistance to various monoclonal antibodies and some polyclonal responses generated by previous infection or vaccination. Compared with N439, the N439K variant strain showed a twofold lower antibody-mediated immune response in 442 convalescent individuals [60]. This substitution does not seem to be present in any of the strains under monitoring [89]; however, Omicron bears an N440K mutation, close enough to have similar effects, as shown in Table 3.
Table 3.
Mutations N440K to ins214EPE in spike protein of the Omicron variant: locations, previous association to other SARS-CoV-2 variants and current knowledge about them (December 2021)
| S protein substitutions | Location in spike | Previous variants | What is already known about this substitution |
|---|---|---|---|
| N440K | RBD | Reported in India [185] and UK [186], Delta |
- Clinical: seem to increase infectivity enhancing biding free energy by 0.62 kcal/mol [147]. Might emerge during replication in the presence of C135 mAbs [186] - Immune: some evidence linking it to immune escape [177, 186] and reinfection. Resistant to class 3 mAbs C135 and REGN10987, that focus on this N440 residue [185, 186] - Structural: the Docking Energy of Omicron RBD-ACE2 for this mutation was − 496.38 and decreased protein stability − 0.50 [119] |
| G446S | RBM |
- Immune: might relate to immune evasion of NAbs in Group A-D, which target the ACE2-binding motif [177] - Structural: reduced RBD stability and High ACE2 biding affinity [119, 183]. Also, shows a high destabilizing effect on RBD [183] |
|
| S477Nb | RBD |
20A.EU2 strain [187] B.1.620 |
- Immune: attenuate neutralization by mAb and convalescent sera [187] - Structural: the Docking Energy of Omicron RBD-ACE2 for this mutation was − 500.05 and decreased protein stability − 0.45 [119]. The highest frequency mutation in RBD (reported 3400 times) increases biding affinity compared with S477 [158, 188] |
| T478Kb | RBD |
Multiple VOI and VOC [147] Delta [89] |
- Clinical: seem to increase infectivity [147, 158, 182] and enhance biding free energy by 1.00 kcal/mol [147] - Immune: linked to immune evasion [158] - Structural: the Docking Energy of Omicron RBD-ACE2 for this mutation was − 517.03 and decreased protein stability − 0.74 [119] |
| E484A | RBD |
- Clinical: some evidence showing an increase in infectivity is available [94] - Immune: multiple reports of mAbs escapes[192]. Markedly resistant to LY-CoV555 mAbs [193]. Together with K417N and Y505H, increases the evasion power of Omicron, compared with Delta variant, against 132 antibodies [179]. Alone, may disrupt Rockefeller University mAbs and together with Q493R and Q498R may disrupt the antibody Regdanvimab by Celltrion, and with K417N and Q493R may seriously reduce the efficacy of Eli Lilly mAB cocktail [179]. Other reports of immune evasion against NAbs of the A-D group [180] and bamlanivimab [193, 194], CT-p59 [194], and convalescent sera [95] were also made. A study mapping RBS’s mutations affecting biding to Regeneron’s REGN-CO2 cocktail and Eli Lilly’s LY-CoV016 antibodies did not consider this mutation as an escape mutation, though [195]. - Structural: the Docking Energy of Omicron RBD-ACE2 for this mutation was − 478.49 and decreased protein stability − 0.79 [120]. Part of an “interface-destabilizing” group and independently can strongly impact hACE2 biding[187, 196]a. Also can be related to the acquirement of the ability to biding with the DDP4 receptor, the main receptor in MERS-CoV and abundantly distributed in human tissues [197] |
|
| Q493R | RBD |
- Clinical: might be linked to mild but prolonged cases of COVID-19 [198] - Immune: High level of C144 antibody resistance [189, 199]a. Together with E484A and Q498R may disrupt the antibody Regdanvimab by Celltrion, and together with K417N and E484A may seriously reduce the efficacy of Eli Lilly mAB cocktail [179]. It also seems to be selected and confers simultaneous resistance to bamlanivimab and etesivimab[200–203]and Ly-CoV555[193]. - Structural: the Docking Energy of Omicron RBD-ACE2 for this mutation was − 581.53 and decreased protein stability − 0.17 [120]. A high negative impact on biding was also reported for this mutation [204]a. Lower biding free energy when compared with Q493 [205] |
|
| G496S | RBD |
- Immune: it might be related to immune evasion of NAbs in Group A-D, which target the ACE2-bindind motif [180] - Structural: enhanced the S1-RBD binding by 13 folds in Alpha and Beta models [204]a. The Docking Energy of Omicron RBD-ACE2 for this mutation was − 505.58 and decreased protein stability − 1.22 [120] |
The mutations were searched in Scielo®, Google academic®, and CAPES Periodicals by the name of the mutation and the words “mutation” and “spike”. Only the articles that were specifically about the mutation were selected, along with other relevant papers that appeared during the reading
aArticles still under peer review (preprints), please, analyze data with caution
abMutations discussed in the text, also the research step was not done to these mutations, the data inserted in the table was a summarization of the data since an extensive work was already done and published about these mutations
K417T, E484K, and N501Y
Other mutations in RBD are present in most strains considered VOC, such as K417T (K417N in Omicron), E484K (in omicron E484A, [89]), and N501Y, the latter two directly in RBM, shared by Gamma and Beta. The Beta strain is reported [124, 131] to be more resistant to some monoclonal antibodies, convalescent plasma and patients vaccinated due to the E484K mutation, which also emerged independently in more than 50 strains and are currently present in all strains under monitoring. The gamma strain also has an evasion capacity, but it is apparently less impactful than the beta strain [132]. It is important to emphasize that the N501Y mutation, in addition to assisting in immune evasion, confers greater affinity for the ACE2 receptor. Several monoclonal antibody treatments under analysis target E484 and K417, other mutations shared by the strains, and may not be as effective in these two variants [124, 131].
S477N
The S477N mutation in Iota (former VBM) and Omicron showed a decrease in protein stability in computer-based analysis, which is still under peer review, compared with N501Y, which stabilizes the protein structure [133]. This study also showed that, while D614G leads to reduced stability and more disease, the variation S477N is less prone to disease [134]. In the same computational study, the N477 mutant showed lower binding affinity, but other authors found that this mutation could increase ACE2 binding affinity [135]. Additionally, there is some evidence linking this mutation with escape from multiple mAbs [94].
T478K
The T478K mutation, also seen in Delta strains and Mexican B.1.1.222 and B.1.1.519, located in the RBD part of the spike protein, forms three additional hydrogen bonds with F486, leading to a stabilization effect in the protein. Furthermore, it is predicted to increase the electrostatic potential of the S protein in a region of ACE2 contact, which might impact the RBD-ACE2 interaction [136, 137]. This mutation was considered a unique mutation site for the Delta variant and was used in screening tests for this variant. However, T478K is also mutated in the Omicron variant, which calls for caution and careful study when adopting this target for variant screening [138, 139]. Immunologically, this mutation might also be related to the success of immune evasion seen in the Delta variant against convalescent and, to a lesser extent, vaccine-elicited sera as well as against mAbs, such as imdevimab [140].
Effects on mAbs and immune response triggered by natural infection or vaccines
As stated by Moore et al. (2021), it is important to keep in mind that reduced sensitivity to neutralization does not necessarily translate into lower vaccines effectiveness, especially those that generate high levels of neutralizing antibodies, such as mRNA vaccines. These vaccines also induce specific cellular immune responses, such as TCD4 + and TCD8 + lymphocytes, which can aid in the response to the virus [131].
Garcia-Beltran et al. (2021) showed the neutralizing power of Pfizer® and Moderna® vaccines against some variants. While Alpha, B.1.1.298, and B.1.429 continued to be neutralized, variants P.2 and Gamma significantly reduced the neutralization capacity of vaccine-induced antibodies. The Beta variant demonstrated high dropout power, being as efficient as other distant coronaviruses.
Another study [124] evaluated the neutralizing efficacy of serum from convalescent and immunized patients with the Pfizer vaccine (25 patients 4–14 days after the second dose) and AstraZeneca (25 patients 14 to 28 days after the second dose), with results consistent with those found by Garcia-Beltran et al. (2021). The authors concluded that the Beta is/was the variant of greatest concern at the time of study, since the data suggest a large reduction in neutralization and even evidence of complete failure in neutralization in some cases. The neutralizing power against gamma and alpha was similar, and few samples failed to demonstrate 100% neutralization in serum dilutions of 1:20, while the neutralization of variant beta (B.1.351) was reduced 7.6-fold in patients immunized with the Pfizer vaccine and ninefold with the AstraZeneca vaccine. Another study [118] similarly demonstrated a reduction in the power of neutralizing antibodies of 1.7-fold in alpha, fivefold in gamma, and 7.9-fold in beta.
Another study [123] mentioned a possible failure in the neutralization of the gamma strain in natural infection convalescent patients and after 5 months of immunization with CoronaVac®; in the latter case, the data suggest a sixfold reduction in neutralization of the strain.
Decreased neutralization is common in vaccines, and a reduction of 4 × in a vaccine for influenza, for example, would signal a need for updating [143]. More robust studies with a higher number of vaccinated individuals should provide more compelling information about the true immunizing power of vaccines and the need for booster doses. Thus, the data seem to suggest the need for caution in discarding preventive measures, individual and collective, even after vaccination.
Epidemiology and reinfection rates
In the matter of epidemiology, it is important to note that the distribution of strains in Brazil used to be very unequal between regions, but delta has been the most prevalent in all states for several months. It is also important to note that the testing and genomic vigilance in the country is low despite all of the efforts made by scientists in the area. Even so, the country ranked 11th worldwide in SARS-CoV-2 sequencing performance published in December 2020 [144]. The country has already registered at least 2000 cases of the omicron variant in Brazil, and the possible increase in infectivity and reinfection rates in this strain may rapidly change the current state if preventive measures are not ensured [145].
Regarding reinfection rates, a study conducted in South Africa, still under peer review, did not find any evidence of increased reinfection risk associated with the beta or delta variant. In contrast, Omicron showed a marked immune evasion ability at the population level, which increases the risk of reinfection by this variant [146]. These data are supported by a computer-based study that predicted the vaccine escape capability of Omicron to be approximately twice as high as that seen in Delta, and its mutations could impact the efficacy of the Eli Lilly antibody cocktail and mAbs from Celltrion® and Rockefeller University® [147].
Omicron
Despite multiple studies seeking to assess the risk of this new Omicron variant, the available data are still preliminary. What is known and was reinforced in the last update of the WHO regarding Omicron is that all variants of SARS-CoV-2 can cause severe cases and death from COVID-19; thus, taking the right preventive measures is still the best option [148]. Even though there are no data to support an increase in the severity of disease by this variant, only the higher number of infections, which are doubling every 2 to 3 days, could represent a new wave of contamination [149].
The concern related to Omicron lies mostly in the fact it has a lot of mutations, some already studied, as previously discussed, some new and some still under investigation. It is also important to understand how these mutations work individually and in combination to better assess the effects of the new variant of interest, as some combinations might further increase receptor binding affinity [150]. More than 30 mutations have been described only on the S protein [89, 151, 152]. What is known about theses variations is described in Tables 2, 3, and 4; most of the articles included in this table are preprint versions, as knowledge about these mutations, especially the ones only seen in Omicron, is very new and published in the last months (Tables 2, 3, and 4). Additionally, a visual demonstration of all the catalogued mutations in the Omicron variant can be seen in Fig. 3.
Table 2.
Mutations A67V to K417N in spike protein of the Omicron variant: locations, previous association to other SARS-CoV-2 variants and current knowledge about them (December 2021)
| S protein substitutions | Location in spike | Previous variants | What is already known about this substitution |
|---|---|---|---|
| A67V | β3-β4 loop of NTD [153] | Eta [153] |
Decreased protein stability by − 0.01 [119] Might promote new hydrophobic interconnections in the β3-β4 loop [153] |
| del69-70 | β3-β4 loop of NTD [153] | Alpha, Beta, Eta, and B.1.375 [153, 154] |
- Clinical: in a study (n = 25), this single mutation showed prolonged inflammation, viral excretion, and late exacerbation 18 days after [155]. Increased infectivity compared to wild-type [156] Immune: Associated with immune escape [153, 157, 158] - Diagnostic: linked to diagnostic failure in at least one RT-PCR assay [159]; Can be used in diagnostic methods, allied with other mutations, to help to screen high profile variants [154, 160] - Structural: might be related to allosteric changes in S1 conformation [161, 162] |
| T95I | S1 NTD | Mu; Iota [163]; B.1.617.1; Delta or Delta plus [89, 164] |
- Structural: induces changes in the protein [165] e decrease protein stability − 0.78 [119] - Immune: showed a minor increase in resistance to convalescent sera [91] and some evidence linked it to vaccine breakthrough infection [166, 167] - Clinical: does not seem to be relevant to infectivity [19]a, but can be associated with higher viral load, especially in co-occurrence of G142D [20]a. Also, it might confer adaptative advantages if combined with E484Q [21]a; |
| del142-144 | N3 loop in NTD | – | It might be related to vaccine breakthrough infection [166] |
| Y145D | N3 loop in NTD | – | It might be related to reduced neutralization of convalescent sera and loos of biding by ADI-56479 MAb to the mutant protein [171] |
| del211 | NTD | – | No articles were found |
| L212I | NTD | B.1.1.33 [172] | Reported in a case study 12 days after administration of convalescent plasma [173] |
| ins214EPE | – | – |
Present in 86,3% of the Omicron cases (of a 131 sequence pool) [25]a Might have occurred because of template switching during viral co-infection or from prevalent templates in the human genome, such as transcripts expressed in alveolar cells or enterocytes [26]a - Structural: decreased protein stability − 0.71 [119] |
| G339D | RBD | Gamma [176]a |
- Immune: creates a mild impact on various antibody-RBD complexes [147], and some evidence of immune escape from a subset of NAbs were also observed [177] - Structural: the Docking Energy of Omicron RBD-ACE2 for this mutation was − 507.06 and decreased protein stability − 1.16 [119] |
| S371L | RBD | – |
- Immune: creates a mild impact on various antibody-RBD complexes [147], and some evidence of immune escape from a subset of NAbs were observed [177] - Structural: the Docking Energy of Omicron RBD-ACE2 for this mutation was − 549.34 and decreased protein stability − 0.22 [119] |
| S373P | RBD | Gamma, Lambda [178] |
- Immune: reported to confer escape immunity elicited by mRNA vaccines [178, 179]. It creates a mild impact on various antibody-RBD complexes [147] - Structural: the Docking Energy of Omicron RBD-ACE2 for this mutation was − 541.87 and decreased protein stability − 0.53 [119] |
| S375F | RBD | – |
- Immune: creates a mild impact on various antibody-RBD complexes because it lies far away from the biding interfaces of most known antibodies [147]. Also, seem to strengthen the biding with the H014 antibody present in some cocktails [180]. Despite that, some evidence of immune escape from a subset of NAbs were also observed[177] - Structure: the Docking Energy of Omicron RBD-ACE2 for this mutation was − 530.07 and decreased protein stability − 0.33 [119] |
| K417Nb | RBD |
Delta plus [164] Multiple VOI and VOC by WHO [147] |
- Immune: likely to scape vaccine-induced antibodies predicted by TopNetmAb model and together with E484A and Y505H, increased the evasion power of Omicron, compared with Delta variant, against 132 antibodies [147]. Other evidence of immune evasion was also reported [177, 181, 182] - Structural: reduced RBD stability and High ACE2 binding affinity [119, 183]. Part of an “interface-destabilizing” group and independently can strongly impact hACE2 biding [184]a |
The mutations were searched in Scielo®, Google academic®, and CAPES Periodicals by the name of the mutation and the words “mutation” and “spike”. Only the articles that were specifically about the mutation were selected, along with other relevant papers that appeared during the reading
aArticles still under peer review (preprints), please, analyze data with caution
bMutations discussed in the text, also the research step was not done to these mutations, the data inserted in the table was a summarization of the data since an extensive work was already done and published about these mutations
Table 4.
Mutations Q498R to L981F in spike protein of the Omicron variant: locations, previous association to other SARS-CoV-2 variants and current knowledge about them (December 2021)
| S protein substitutions | Location in spike | Previous variants | What is already known about this substitution |
|---|---|---|---|
| Q498R | RBD (ACE2-RBD interface) [206] | Some strains from USA, UK and Italy [207] |
- Clinical: it could relate to a previous appearance of N501Y mutation, and the affinity for ACE-2 seems to increase dramatically [206]. This pair appeared in GISAID sequences from France, Netherlands, South Africa, Spain, the UK, and the USA between March and September 2021 [208]a - Immune: could be linked with immune evasion [206, 209, 210]. The presence of neutralizing antibodies C121 and C141 enriched the appearance of Q493R mutation [211]. Also, together with E484A and Q493R may disrupt the antibody Regdanvimab by Celltrion [179] - Structural: enhanced the S1-RBD biding[210] by 98 folds in Alpha and Beta models [204]a. The Docking Energy of Omicron RBD-ACE2 for this mutation was − 527.38 and decreased protein stability − 0.17 [120]. It promotes a significant change in free energy (− 1.92 kcal/mol), contributing to a higher binding to the receptor, using Alpha and Beta variants as a model [204]. Helped, along with N460K and E484K, to strengthen de electrostatic complementarity between RBD and ACE2 [206, 212, 213]. In contrast, another study found low effects on biding by this mutation alone [211]. Also, despite arginine being a non-aromatic amino acid, it is involved with π-stacking interactions and conformation flexibility; therefore, the author suggested that this could be the reason why this combination is more tolerated [208]a |
| N501Yb | RBD | Beta and Gamma |
- Clinical: it seems to increase infectivity, improving binding free energy by 0.55 kcal/mol [179, 190] - Immune: evidence of immune evasion to mAbs [184, 190] - Structural: increases RBD stability and High ACE2 binding affinity by 10-folds [120, 160, 186, 190, 214]. Paired up with Q498R mutation enhances affinity for ACE-2 dramatically, about fourfold above N501Y alone [137, 206]. Also part of an “interface-destabilizing” group and independently can strongly impact hACE2 biding [187] |
| Y505H | RBD |
- Immune: together with E484A and Y505H, increased the evasion power of Omicron, compared with Delta variant, against 132 antibodies [179]. Evasion against other antibodies, such as CT-q59, was also reported[194] - Structure: enhanced the S1-RBD biding by 14 folds in Alpha and Beta models [204]a. Increases RBD stability and low ACE2 binding affinity [120, 186], in SIFT tool protein stability analysis, this mutation affected protein function [120]. Another study shows that Y505H reduced the usage of ACE2 receptors, mainly because of the loss of hydrogen bonds and hydrophobic interactions [215] |
|
| T547K | Decreased protein stability − 1.05 [120] | ||
| D614Gb |
- Clinical: Some evidence linked to vaccine breakthrough infection [168] - Structural: Decreased protein stability − 0.93 [120] |
||
| H655Y | Furin site between S1/S2 [216]. Between the RBD and the FP [217] |
Strains from USA, Scotland, China, South Africa strains [218] |
- Clinical: could be related to persistent infection [221]a . It might play a potential role in replication, transmissibility, and pathogenicity [217, 219] a. It might not increase viral infectivity alone [217, 222], but the cluster with N679K+P681H is associated with an increase in cell invasion and improved metastasis [223]. Also, it might relate to tissue culture adaptations [224], which also appeared in Rhesus Macaques treated with Normal plasma [225] - Immune: some evidence linking it to mAb evasion [224, 226]a - Structural: decreased protein stability − 0.08 [120]. In contrast, another study found that this mutations increases the structural stability in S protein, also associating this mutation with decreased molecular flexibility [73]. Evidence shows that may be related to improving spike glycoprotein fusion efficiency and host cell entry [217, 219, 224] a. Enhances spike cleavage and viral growth in both Vero E6 and human-like Vero-TMPRSS2 cells [219]a. It seems to increase ACE2 binding interaction by 1.2-fold [227] - Diagnostic: used before to discriminate between Beta and Gamma [228] |
| N679K | Furin site between S1/S2 | B.1.1.433, AT.1 B.1.258, B.1.1.7, P.1 and C.1.2 |
- Clinical: the cluster H655Y+N679K+P681H is associated with a higher in cell invasion and improved metastasis [223] - Structural: decreased protein stability − 0.32 [120]. Along with P681H, it might relate to beneficial enzyme-substrate coupling [229] |
| P681Hb | Furin site between S1/S2 | Alpha, Mu, Gamma, B.1.1.318 |
- Clinical: the cluster H655Y+N679K+P681H is associated with a higher cell invasion and improved metastasis [223]. It might facilitate the efficient spread and infection of the virus [220] - Structural: decreased protein stability − 1.27 [120]. Likely to improve furin cleavage [230]a |
| N764K | N-terminal of S2 | P.1 | - Structural: decreased protein stability − 0.21, and in SIFT tool protein stability analysis, this mutation affected protein function[120] |
| D796Y | HMN.19B [231] and A.27 [232] | - Structural: decreased protein stability − 0.09 [120]. It might be associated with ligand binding surfaces changes[233]. Appears in only 0.05% of global sequences [158] | |
| N856K | Between FP and HR1 | Decrease protein stability − 0.38 [120] | |
| Q954H | HR1 | Delta [234] a |
- Clinical: potential enhanced infectivity and transmission by affecting the fusion state of spike protein[234] a It might improve interaction affinity between HR1 and HR2, together with N969K and L981F, increasing membrane fusion and infectivity[196]a - Structural: decreased protein stability − 0.86 [120] |
| N969K | HR1 |
- Clinical: it might enhance interaction affinity between HR1 and HR2, together with Q954H and L981F, increasing membrane fusion and infectivity [196] a - Structural: decreased protein stability − 0.63 [120] |
|
| L981F | HR1 |
- Clinical: it might enhance interaction affinity between HR1 and HR2, together with Q954H and N969K, increasing membrane fusion and infectivity [196] a Decreased protein stability − 1.24 [120] |
The mutations were searched in Scielo®, Google academic®, and CAPES Periodicals by the name of the mutation and the words “mutation” and “spike”. Only the articles that were specifically about the mutation were selected, along with other relevant papers that appeared during the reading
aArticles still under peer review (preprints), please, analyze data with caution
bMutations discussed in the text, also the research step was not done to these mutations, the data inserted in the table was a summarization of the data since an extensive work was already done and published about these mutations
Fig. 3.
Visual representation of Tables 2, 3, and 4, showing all catalogued mutations of Omicron’s spike proteins. They were constructed using PyMOL 2.5.2® (The PyMOL Molecular Graphics System) and Biorender®
Another computational study comparing Delta and Omicron suggests that the S protein and its RBD are more hydrophobic, as the contents of leucine and phenylalanine and other hydrophobic amino acids are higher in the Omicron variant. Additionally, alpha-helix structures are more present in omicron than in the Delta variant in the whole S protein, suggesting, according to the authors, a more stable structure [119].
The same study [119] also compared the docking energy of the strains in relation to the Wuhan wild type. The wild type reached − 500.37 of docking energy using HEX software, whereas Delta reached − 529.62 and Omicron − 539.81; thus, the Omicron variant S protein seems to hold a higher affinity and fitness with the ACE2 receptor. Additionally, regarding the Omicron RBD mutations, E484A showed the lowest energy interaction with the receptor (− 478.49), and a higher energy interaction occurred with Q439R (− 581.53). The values for the other mutations are shown in Table 2. These data show that the Omicron variant transmission rate may be greater than that seen in the Delta strain, which is currently responsible for more than 90% of new COVID cases in the world.
Some results, one still under peer review, suggest that Omicron is very good at evading immunologic defense and mAbs [177, 232, 233]. Regarding vaccines, the emergence of omicron has heated the debate about booster doses. Pfizer®, for example, released statements saying that 3 doses of Pfizer-BioNTech COVID-19 vaccine are sufficient to neutralize Omicron, while two doses, which was the initial plan, show significantly reduced neutralization titers [234, 235]
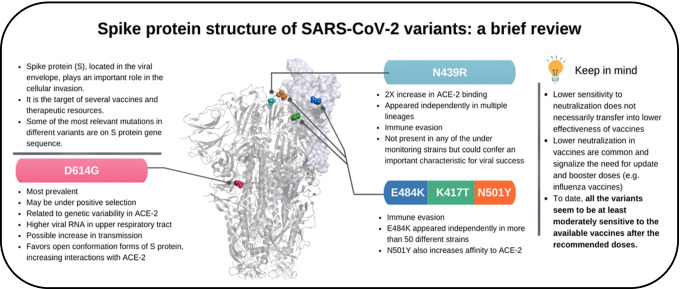
In summary, the new variants of SARS-CoV-2 are a major issue, and mass vaccination is the best way to control the pandemic (Fig. 4). Even so, the appearance of strains capable of evading the immune response is inevitable because vaccination progress can also provide selective pressure to these strains, hence the need for maintaining preventive measures. It is also important to guarantee vaccine doses to everybody [236]. Despite efforts, this is not the reality, as African vaccination data reports indicate that some countries still have less than 20 doses per 100 population as of March 3, 2022 [237]. This vaccine inequity [238], which may arise from difficulties in the four dimensions of effective global immunization (development and production, allocation, affordability and deployment) [239, 240], will probably lead to a more prolonged pandemic and might even culminate in a more resistant variant [239, 241]. Thus far, all of the variants seem to be at least moderately sensitive to the available vaccines after the recommended doses, especially blocking symptomatic and severe cases, but booster doses will be necessary, as already seen in some countries, especially with the emergence of Omicron.
Fig. 4.
Summary of the main information regarding the spike protein and its mutations. The box summarizes data considering common characteristics of the protein and its major mutations, such as D614G, N439R, E484K, K417T, and N501Y
Final remarks
The S protein has a crucial role in viral infectivity, and all strains considered of interest or concern have mutations in the S sequence. These mutations seem to confer immune evasion and higher infectivity properties, especially those linked to conformational changes in its structure. Surprisingly, the mRNA vaccines, such Pfizer® or Moderna®, which use this protein sequence, seem to trigger a strong immune response [242, 243] to vaccination that can protect against most variants with multiple mutations in its sequence [103], even though some variants, such as Gamma [73, 122], Beta [112, 244], or Delta [104], have been demonstrated to be more tolerant to postvaccine serum, which could cause more infections among vaccinated people.
Omicron variant risk is still under preliminary assessment but appears to be more effective in evading immune responses, largely because of the many mutations in its S protein, and it also seems to transmit better than Delta. To date, there is no evidence of an increase in the severity of disease, even though the number of vaccinated people infected can mask these results, as vaccination tends to protect individuals from more severe cases or progression. Additionally, some preliminary evidence showing major outbreaks among vaccinated people, indicating the need for booster doses, is already available. Many of these strain mutations, highlighted in Table 1, are new, and there is almost no knowledge about them, making these genetic variations good subjects for further research.
This review summarizes the disease process, biology and replication cycle of SARS-CoV-2 and the important mutations carried by the most worrisome variants thus far. Additionally, this study highlights the most recent knowledge about Omicron’s spike mutations.
Genomic sequencing of the different SARS-CoV-2 variants and their hosts has undoubtedly led to the rapid development of vaccines by pharmaceutical companies around the world, allowing people to experience the pandemic in a slightly less catastrophic way. Of course, this happened differently in each country across the globe, mainly because different countries adopted distinct testing procedures, social distancing, and specific vaccination schedules in an uncoordinated way. However, there is a consensus that Science has saved lives around the world and has provided different people with the possibility of being better prepared to face similar public health problems that may occur in the future.
Acknowledgements
The authors thank Unioeste (Universidade Estadual do Oeste do Paraná), Fundep (Fundação de Apoio ao Ensino, Extensão, Pesquisa Post-Graduate), PEL (Program for Teaching Languages), Cardiff University and Global Challenges Research Fund for insistently promoting research at the Laboratory of Molecular Biochemistry at Unioeste.
Authors’ contributions
Conceptualization: KLC and CRE. Data curation: KLC and AM. Formal analysis: KLC and RCGS. Funding acquisition: LOF and RCGS. Methodology: KLC. Project administration: RCGS. Visualization: KLC, JLCS, MKK. Writing—original draft: KLC; Writing–review and editing: KLC and RCGS. All authors read and approved the final manuscript.
Funding
Despite the drastic reduction of investments in research by the Brazilian government in recent years, this project received the following support: K.L. Candido was fellow of CAPES-Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-Brasil (CAPES)-Finance Code 001; C.R. Eich was fellow of CNPq - Conselho Nacional de Desenvolvimento Científico e Tecnológico (Pibic 2021/2022); LaBioqMol was partially supported by Instituto São João and Sicoob Bank from Cascavel, PR, Brazil.
Declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Kattlyn Laryssa Candido, Email: k.candido@hotmail.com.
Rita de Cássia Garcia Simão, Email: rita.simao@unioeste.br, Email: ritabioq@icloud.com.
References
- 1.Rosling L, Rosling M. Pneumonia causes panic in Guangdong province. BMJ. 2003;326:416. doi: 10.1136/bmj.326.7386.416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Daniloski Z, Jordan TX, Wessels HH, et al. Identification of required host factors for SARS-CoV-2 infection in human cells. Cell. 2021;184:92–105.e16. doi: 10.1016/j.cell.2020.10.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zaki AM, van Boheemen S, Bestebroer TM, et al. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med. 2012;367:1814–1820. doi: 10.1056/nejmoa1211721. [DOI] [PubMed] [Google Scholar]
- 4.Abdelghany TM, Ganash M, Bakri MM, et al. SARS-CoV-2, the other face to SARS-CoV and MERS-CoV: Future predictions. Biomed J. 2021;44:86–93. doi: 10.1016/J.BJ.2020.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Liu Y, Gayle AA, Wilder-Smith A, Rocklöv J. The reproductive number of COVID-19 is higher compared to SARS coronavirus. J Travel Med. 2020 doi: 10.1093/JTM/TAAA021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kuter BJ, Offit PA, Poland GA. The development of COVID-19 vaccines in the United States: Why and how so fast? Vaccine. 2021;39:2491–2495. doi: 10.1016/j.vaccine.2021.03.077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bloom DE, Cadarette D, Ferranna M, et al. How new models of vaccine development for COVID-19 have helped address an epic public health crisis. Health Aff. 2021;40:410–418. doi: 10.1377/hlthaff.2020.02012. [DOI] [PubMed] [Google Scholar]
- 8.Yurkovetskiy L, Wang X, Pascal KE, et al. Structural and functional analysis of the D614G SARS-CoV-2 spike protein variant. Cell. 2020;183:739–751.e8. doi: 10.1016/j.cell.2020.09.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Thomas SJ, Moreira ED, Kitchin N, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine through 6 months. N Engl J Med. 2021;385:1761–1773. doi: 10.1056/NEJMOA2110345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383:2603–2615. doi: 10.1056/NEJMOA2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mulligan MJ, Lyke KE, Kitchin N, et al. Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults. Nature. 2020;586:589–593. doi: 10.1038/S41586-020-2639-4. [DOI] [PubMed] [Google Scholar]
- 12.Janssen Vaccines & Prevention B.V (2022) A study of Ad26.COV2.S for the prevention of SARS-CoV-2-mediated COVID-19 in adult participants (ENSEMBLE). In: ClinicalTrials. https://clinicaltrials.gov/ct2/show/study/NCT04505722. Accessed 3 Mar 2022
- 13.Sadoff J, Gray G, Vandebosch A et al (2022) Final analysis of efficacy and safety of single-dose Ad26.COV2.S. N Engl J Med 386. 10.1056/NEJMOA2117608 [DOI] [PMC free article] [PubMed]
- 14.El Sahly HM, Baden LR, Essink B, et al. Efficacy of the mRNA-1273 SARS-CoV-2 Vaccine at Completion of Blinded Phase. N Engl J Med. 2021;385:1774–1785. doi: 10.1056/NEJMOA2113017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Baden LR, El Sahly HM, Essink B, et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N Engl J Med. 2021;384:403–416. doi: 10.1056/NEJMOA2035389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Santosham M, Wolff M, Reid R, et al. The efficacy in Navajo infants of a conjugate vaccine consisting of Haemophilus influenzae type b polysaccharide and Neisseria meningitidis outer-membrane protein complex. N Engl J Med. 1991;324:1767–1772. doi: 10.1056/NEJM199106203242503. [DOI] [PubMed] [Google Scholar]
- 17.Weibel RE, Neff BJ, Kuter BJ, et al. Live attenuated varicella virus vaccine: efficacy trial in healthy children. N Engl J Med. 1984;310:1409–1415. doi: 10.1056/NEJM198405313102201. [DOI] [PubMed] [Google Scholar]
- 18.Innis BL, Snitbhan R, Kunasol P, et al. Protection against hepatitis A by an inactivated vaccine. JAMA. 1994;271:1328–1334. doi: 10.1001/jama.1994.03510410040030. [DOI] [PubMed] [Google Scholar]
- 19.Werzberger A, Mensch B, Kuter B, et al. A controlled trial of a formalin-inactivated hepatitis a vaccine in healthy children. N Engl J Med. 1992;327:453–457. doi: 10.1056/NEJM199208133270702. [DOI] [PubMed] [Google Scholar]
- 20.Koerber A (2020) Is It Fake News or Is It Open Science? Science Communication in the COVID-19 Pandemic: 35:22–27. 10.1177/1050651920958506
- 21.King A (2020) Fast news or fake news? EMBO Rep 21:e50817. 10.15252/EMBR.202050817 [DOI] [PMC free article] [PubMed]
- 22.Baloch S, Baloch MA, Zheng T, Pei X. The coronavirus disease 2019 (COVID-19) pandemic. Tohoku J Exp Med. 2020;250:271–278. doi: 10.1620/tjem.250.271. [DOI] [PubMed] [Google Scholar]
- 23.Wiersinga WJ, Rhodes A, Cheng AC, et al. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;324:782–793. doi: 10.1001/jama.2020.12839. [DOI] [PubMed] [Google Scholar]
- 24.Zhu N, Zhang D, Wang W, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Serpeloni JM, Lima Neto QA, Lucio LC, et al. Genome interaction of the virus and the host genes and non-coding RNAs in SARS-CoV-2 infection. Immunobiology. 2021;226:152130. doi: 10.1016/j.imbio.2021.152130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tay MZ, Poh CM, Rénia L, et al. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol. 2020;20:363–374. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hui DSC, Zumla A. Severe acute respiratory syndrome. Infect Dis Clin North Am. 2019;33:869–889. doi: 10.1016/j.idc.2019.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bouadma L, Lescure F-X, Lucet J-C, et al. Severe SARS-CoV-2 infections: practical considerations and management strategy for intensivists. Intensive Care Med. 2020;46:579–582. doi: 10.1007/s00134-020-05967-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kampf G. Potential role of inanimate surfaces for the spread of coronaviruses and their inactivation with disinfectant agents. Infect Prev Pract. 2020;2:100044. doi: 10.1016/j.infpip.2020.100044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ra SH, Lim JS, Kim GU, et al. Upper respiratory viral load in asymptomatic individuals and mildly symptomatic patients with SARS-CoV-2 infection. Thorax. 2021;76:61–63. doi: 10.1136/thoraxjnl-2020-215042. [DOI] [PubMed] [Google Scholar]
- 31.Gaspar-Rodríguez A, Padilla-González A, Rivera-Toledo E (2021) Coronavirus persistence in human respiratory tract and cell culture: an overview. Braz J Infect Dis 25:101632 [DOI] [PMC free article] [PubMed]
- 32.Caccuri F, Zani A, Messali S, et al. A persistently replicating SARS-CoV-2 variant derived from an asymptomatic individual. J Transl Med. 2020 doi: 10.1186/s12967-020-02535-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pinho JRR, Oliveira KG de, Sitnik R et al (2021) Long term persistence of coronavirus SARS-CoV-2 infection. Einstein (Sao Paulo) 19:eRC6369. 10.31744/EINSTEIN_JOURNAL/2021RC6369 [DOI] [PMC free article] [PubMed]
- 34.Osman AA, Al Daajani MM, Alsahafi AJ. Re-positive coronavirus disease 2019 PCR test: could it be a reinfection? New Microbes New Infect. 2020 doi: 10.1016/J.NMNI.2020.100748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Li Q, Zheng XS, Shen XR, et al. Prolonged shedding of severe acute respiratory syndrome coronavirus 2 in patients with COVID-19. Emerg Microbes Infect. 2020;9:2571–2577. doi: 10.1080/22221751.2020.1852058/SUPPL_FILE/TEMI_A_1852058_SM4982.DOCX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Corman V, Bleicker T, Brünink S et al (2020) Diagnostic detection of 2019-nCoV by real-time RT-RCR. Charité Berlin 1–13
- 37.Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020;323:1843–1844. doi: 10.1001/jama.2020.3786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.ANVISA (2020) Produtos para diagnóstico in vitro de COVID-19 regularizados pela ANVISA. https://www.gov.br/anvisa/pt-br/assuntos/produtosparasaude/coronavirus/produtos-diagnostico-aprovados. Accessed 23 Mar 2021
- 39.CDC Real-Time RT-PCR diagnostic panel for emergency use only. CDC EUA. 2020;3:1–42. [Google Scholar]
- 40.CDC (2021) In Vitro Diagnostics EUAs | FDA. In: Cdc. https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/in-vitro-diagnostics-euas. Accessed 23 Mar 2021
- 41.Artik Y, Coşğun AB, Cesur NP, et al. Comparison of COVID-19 laboratory diagnosis by commercial kits: Effectivity of RT-PCR to the RT-LAMP. J Med Virol. 2022 doi: 10.1002/JMV.27559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Varlamov DA, Blagodatskikh KA, Smirnova EV, et al. Combinations of PCR and isothermal amplification techniques are suitable for fast and sensitive detection of SARS-CoV-2 viral RNA. Front Bioeng Biotechnol. 2020;8:1294. doi: 10.3389/FBIOE.2020.604793/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.R Augustine A Hasan S Das (2020) Loop-mediated isothermal amplification (LAMP): A rapid, sensitive, specific, and cost-effective point-of-care test for coronaviruses in the context of COVID-19 pandemic. Biol et al 2020 Vol 9 Page 182 9 182 10.3390/BIOLOGY9080182 [DOI] [PMC free article] [PubMed]
- 44.World Health Organization (2022) COVID-19 vaccine tracker and landscape. In: Who. https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines. Accessed 28 Feb 2022
- 45.Speiser DE, Bachmann MF. COVID-19: mechanisms of vaccination and immunity. Vaccines. 2020;8:404. doi: 10.3390/vaccines8030404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Pfizer (2021) The Facts About Pfizer and BioNTech’s COVID-19 Vaccine | Pfizer. In: Pfizer. https://www.pfizer.com/news/hot-topics/the_facts_about_pfizer_and_biontech_s_covid_19_vaccine. Accessed 16 Mar 2021
- 47.National Health Commission & State Administration of, Medicine TC (2021) Diagnosis and Treatment Protocol for Novel Coronavirus Pneumonia. In: Use of Patented Traditional Chinese Medicine against COVID-19. National Health Commission & State Administration of Traditional Chin Med 13–34
- 48.Jin Y, Yang H, Ji W, et al. Virology, epidemiology, pathogenesis, and control of COVID-19. Viruses. 2020 doi: 10.3390/V12040372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kasmi Y, Khataby K, Souiri A, Ennaji MM (2020) Coronaviridae: 100,000 years of emergence and reemergence. In: Emerging and Reemerging Viral Pathogens. Elsevier 127–149
- 50.Coronaviridae Study Group of the International Committee on Taxonomy of Viruses The species Severe acute respiratory syndrome-related coronavirus: classifying 2019-nCoV and naming it SARS-CoV-2. Nat Microbiol. 2020;5:536–544. doi: 10.1038/s41564-020-0695-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Velavan TP, Meyer CG. The COVID-19 epidemic. Trop Med Int Heal. 2020;25:278. doi: 10.1111/TMI.13383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Malik YA. Properties of Coronavirus and SARS-CoV-2. Malays J Pathol. 2020;42:3–11. [PubMed] [Google Scholar]
- 53.Neuman BW, Kiss G, Kunding AH, et al. A structural analysis of M protein in coronavirus assembly and morphology. J Struct Biol. 2011;174:11–22. doi: 10.1016/J.JSB.2010.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Cao Y, Yang R, Lee I, et al. Characterization of the SARS-CoV-2 E Protein: Sequence, Structure, Viroporin, and Inhibitors. Protein Sci. 2021;30:1114. doi: 10.1002/PRO.4075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zeng W, Liu G, Ma H, et al. Biochemical characterization of SARS-CoV-2 nucleocapsid protein. Biochem Biophys Res Commun. 2020;527:618. doi: 10.1016/J.BBRC.2020.04.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Schrödinger L The PyMOL Molecular Graphics System
- 57.BioRender. biorender.com. Accessed 3 Mar 2022
- 58.Scudellari M. How the coronavirus infects cells — and why Delta is so dangerous. Nature. 2021;595:640–644. doi: 10.1038/d41586-021-02039-y. [DOI] [PubMed] [Google Scholar]
- 59.Consortium AA Structural and functional comparison of SARS-CoV-2-spike receptor binding domain produced in Pichia pastoris and mammalian cells. Sci Rep. 2020;10:21779. doi: 10.1038/s41598-020-78711-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Thomson EC, Rosen LE, Shepherd JG, et al. Circulating SARS-CoV-2 spike N439K variants maintain fitness while evading antibody-mediated immunity. Cell. 2021;184:1171–1187.e20. doi: 10.1016/j.cell.2021.01.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Yi C, Sun X, Ye J, et al. Key residues of the receptor binding motif in the spike protein of SARS-CoV-2 that interact with ACE2 and neutralizing antibodies. Cell Mol Immunol. 2020;17:621–630. doi: 10.1038/s41423-020-0458-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Peacock TP, Goldhill DH, Zhou J, et al. (2021) The furin cleavage site in the SARS-CoV-2 spike protein is required for transmission in ferrets. Nat Microbiol. 2021;67(6):899–909. doi: 10.1038/s41564-021-00908-w. [DOI] [PubMed] [Google Scholar]
- 63.Huang S-W, Miller SO, Yen C-H, Wang S-F. Impact of genetic variability in ACE2 expression on the evolutionary dynamics of SARS-CoV-2 spike D614G mutation. Genes (Basel) 2020;12:16. doi: 10.3390/genes12010016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hoffmann M, Kleine-Weber H, Pöhlmann S. A multibasic cleavage site in the spike protein of SARS-CoV-2 is essential for infection of human lung cells. Mol Cell. 2020;78:779–784.e5. doi: 10.1016/J.MOLCEL.2020.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Mykytyn AZ, Breugem TI, Riesebosch S, et al. Sars-cov-2 entry into human airway organoids is serine protease-mediated and facilitated by the multibasic cleavage site. Elife. 2021;10:1–23. doi: 10.7554/ELIFE.64508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zhang Q, Xiang R, Huo S, et al. Molecular mechanism of interaction between SARS-CoV-2 and host cells and interventional therapy. Signal Transduct Target Ther. 2021;6:1–19. doi: 10.1038/s41392-020-00451-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Osipiuk J, Azizi SA, Dvorkin S, et al. Structure of papain-like protease from SARS-CoV-2 and its complexes with non-covalent inhibitors. Nat Commun. 2021;12:1–9. doi: 10.1038/s41467-021-21060-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Snijder EJ, Decroly E, Ziebuhr J. The nonstructural proteins directing coronavirus RNA synthesis and processing. Adv Virus Res. 2016;96:59–126. doi: 10.1016/BS.AIVIR.2016.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.de Wit E, van Doremalen N, Falzarano D, Munster VJ. SARS and MERS: recent insights into emerging coronaviruses. Nat Rev Microbiol. 2016;14:523–534. doi: 10.1038/nrmicro.2016.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hillen HS, Kokic G, Farnung L, et al. (2020) Structure of replicating SARS-CoV-2 polymerase. Nat. 2020;5847819(584):154–156. doi: 10.1038/s41586-020-2368-8. [DOI] [PubMed] [Google Scholar]
- 71.Finkel Y, Gluck A, Nachshon A, et al. (2021) SARS-CoV-2 uses a multipronged strategy to impede host protein synthesis. Nat. 2021;5947862(594):240–245. doi: 10.1038/s41586-021-03610-3. [DOI] [PubMed] [Google Scholar]
- 72.da Santos FR, S, de Azevedo MSP, Bielavsky M, , et al. Mutational profile confers increased stability of SARS-CoV-2 spike protein in Brazilian isolates. J Biomol Struct Dyn. 2021 doi: 10.1080/07391102.2021.1982775. [DOI] [PubMed] [Google Scholar]
- 73.Hoffmann M, Prerna A, Ru ¨ diger G, , et al. SARS-CoV-2 variants B.1.351 and P.1 escape from neutralizing antibodies | Elsevier Enhanced Reader. Cell. 2021;184:1–10. doi: 10.1016/j.cell.2020.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Buchrieser J, Dufloo J, Hubert M, et al (2020) Syncytia formation by SARS‐CoV‐2‐infected cells. EMBO J 39. 10.15252/embj.2020106267 [DOI] [PMC free article] [PubMed]
- 75.Chan JFW, Chan KH, Choi GKY, et al. Differential cell line susceptibility to the emerging novel human betacoronavirus 2c EMC/2012: Implications for disease pathogenesis and clinical manifestation. J Infect Dis. 2013;207:1743–1752. doi: 10.1093/infdis/jit123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Zeng C, Evans JP, King T, et al. SARS-CoV-2 spreads through cell-to-cell transmission. Proc Natl Acad Sci USA. 2022 doi: 10.1073/PNAS.2111400119/-/DCSUPPLEMENTAL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Rocheleau L, Laroche G, Fu K, et al. Identification of a high-frequency intrahost SARS-CoV-2 spike variant with enhanced cytopathic and fusogenic effects. MBio. 2021;12:e0078821. doi: 10.1128/MBIO.00788-21/SUPPL_FILE/MBIO.00788-21-ST001.TXT. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Li J, Du P, Yang L, et al. Two-step fitness selection for intra-host variations in SARS-CoV-2. Cell Rep. 2022;38:110205. doi: 10.1016/J.CELREP.2021.110205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wang Y, Wang D, Zhang L, et al. Intra-host variation and evolutionary dynamics of SARS-CoV-2 populations in COVID-19 patients. Genome Med. 2021;13:1–13. doi: 10.1186/S13073-021-00847-5/FIGURES/4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.World Health Organization (2020) Draft landscape of COVID-19 candidate vaccines - 15 May 2020. Who 3
- 81.Precision Vaccinations (2021) Ad26.COV2-S vaccine — precision vaccinations. https://www.precisionvaccinations.com/vaccines/cvncov-vaccine. Accessed 16 Mar 2021
- 82.WHO (2021) SARS-CoV-2 virus mutations & variants
- 83.Mittal A, Verma V. Connections between biomechanics and higher infectivity: a tale of the D614G mutation in the SARS-CoV-2 spike protein. Signal Transduct Target Ther. 2021;6:11. doi: 10.1038/s41392-020-00439-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.GISAID/FIOCRUZ (2021) GISAID – Genomahcov – Fiocruz. http://www.genomahcov.fiocruz.br/gisaid/. Accessed 21 Mar 2021
- 85.GISAID (2021) GISAID - Mission. https://www.gisaid.org/about-us/mission/. Accessed 21 Mar 2021
- 86.Korber B, Fischer WM, Gnanakaran S, et al. Tracking changes in SARS-CoV-2 spike: evidence that D614G increases infectivity of the COVID-19 virus. Cell. 2020;182:812–827.e19. doi: 10.1016/j.cell.2020.06.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ministry of Health (2021) COVID-19 Weekly Epidemiological Update. World Heal Organ 1–33
- 88.Organisation Mondiale de la Santé (2021) Weekly epidemiological update on COVID-19 - 23 March 2021. In: Who. https://www.who.int/publications/m/item/weekly-epidemiological-update---16-march-2021. Accessed 21 Mar 2021
- 89.CDC (2021) SARS-CoV-2 Variants of Concern. In: Centers Dis. Control Prev. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/variant-surveillance/variant-info.html#Concern. Accessed 21 Mar 2021
- 90.WHO Tracking SARS-CoV-2 variants. In: 2021. https://www.who.int/en/activities/tracking-SARS-CoV-2-variants/. Accessed 12 May 2021
- 91.Uriu K, Cárdenas P, Muñoz E, et al (2022) Characterization of the immune resistance of SARS-CoV-2 Mu variant and the robust immunity induced by Mu infection. J Infect Dis 2021.11.23.469770. 10.1093/infdis/jiac053 [DOI] [PMC free article] [PubMed]
- 92.Romero PE, Dávila-Barclay A, Salvatierra G, et al (2021) The Emergence of Sars-CoV-2 Variant Lambda (C.37) in South America. Microbiol Spectr 9. 10.1128/SPECTRUM.00789-21/SUPPL_FILE/REVIEWER-COMMENTS.PDF [DOI] [PMC free article] [PubMed]
- 93.Weisblum Y, Schmidt F, Zhang F, et al. Escape from neutralizing antibodies 1 by SARS-CoV-2 spike protein variants. Elife. 2020;9:1. doi: 10.7554/ELIFE.61312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Liu Z, VanBlargan LA, Bloyet LM, et al. Identification of SARS-CoV-2 spike mutations that attenuate monoclonal and serum antibody neutralization. Cell Host Microbe. 2021;29:477–488.e4. doi: 10.1016/J.CHOM.2021.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Tchesnokova V, Kulasekara H, Larson L, et al. Acquisition of the L452R Mutation in the ACE2-Binding Interface of Spike Protein Triggers Recent Massive Expansion of SARSCoV- 2 Variants. J Clin Microbiol. 2021 doi: 10.1128/JCM.00921-21/SUPPL_FILE/JCM.00921-21-S0007.PDF. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kimura I, Kosugi Y, Wu J, et al. The SARS-CoV-2 Lambda variant exhibits enhanced infectivity and immune resistance. Cell Rep. 2022;38:110218. doi: 10.1016/J.CELREP.2021.110218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.ATCC BEI Reagent Search. https://www.beiresources.org/Catalog/animalviruses/NR-54000.aspx. Accessed 21 Mar 2021
- 98.V Borges C Sousa L Menezes (2021) Tracking SARS-CoV-2 lineage B.1.1.7 dissemination: insights from nationwide spike gene target failure (SGTF) and spike gene late detection (SGTL) data, Portugal, week 49, et al 2020 to week 3 2021 Eurosurveillance. 10.2807/1560-7917.ES.2021.26.10.2100130 [DOI] [PMC free article] [PubMed]
- 99.Galloway SE, Paul P, MacCannell DR, et al (2021) Emergence of SARS-CoV-2 B.1.1.7 Lineage — United States, December 29, 2020–January 12, 2021. MMWR Morb Mortal Wkly Rep 70:95–99. 10.15585/mmwr.mm7003e2 [DOI] [PMC free article] [PubMed]
- 100.Davies NG, Jarvis CI, CMMID COVID-19 Working Group, et al (2021) Increased mortality in community-tested cases of SARS-CoV-2 lineage B.1.1.7. Nature 1–5. 10.1038/s41586-021-03426-1 [DOI] [PMC free article] [PubMed]
- 101.Tegally H, Wilkinson E, Giovanetti M, et al. (2021) Detection of a SARS-CoV-2 variant of concern in South Africa. Nat. 2021;5927854(592):438–443. doi: 10.1038/s41586-021-03402-9. [DOI] [PubMed] [Google Scholar]
- 102.Zhou D, Dejnirattisai W, Supasa P, et al. Evidence of escape of SARS-CoV-2 variant B.1.351 from natural and vaccine-induced sera. Cell. 2021;184:2348–2361.e6. doi: 10.1016/j.cell.2021.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Lopez Bernal J, Andrews N, Gower C, et al. Effectiveness of Covid-19 Vaccines against the B.1.617.2 (Delta) Variant. N Engl J Med. 2021;385:585–594. doi: 10.1056/nejmoa2108891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Planas D, Veyer D, Baidaliuk A, et al. Reduced sensitivity of SARS-CoV-2 variant Delta to antibody neutralization. Nature. 2021;596:276–280. doi: 10.1038/s41586-021-03777-9. [DOI] [PubMed] [Google Scholar]
- 105.Bansal K, Kumar S (2021) Mutational cascade of SARS-CoV-2 leading to evolution and emergence of omicron variant. bioRxiv 2021.12.06.471389. 10.1101/2021.12.06.471389 [DOI] [PMC free article] [PubMed]
- 106.Chakraborty C, Bhattacharya M, Ashish |, et al (2021) Present variants of concern and variants of interest of severe acute respiratory syndrome coronavirus 2: their significant mutations in S-glycoprotein, infectivity, re-infectivity, immune escape and vaccines activity. 10.1002/rmv.2270
- 107.Jassat W, Mudara C, Ozougwu L, et al. Difference in mortality among individuals admitted to hospital with COVID-19 during the first and second waves in South Africa: a cohort study. Lancet Glob Heal. 2021;9:e1216–e1225. doi: 10.1016/S2214-109X(21)00289-8/ATTACHMENT/A9D1F970-C4E2-4A9A-8A9A-B9488222081F/MMC1.PDF. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Abu-Raddad LJ, Chemaitelly H, Ayoub HH, et al (2021) Severity, criticality, and fatality of the SARS-CoV-2 Beta variant. Clin Infect Dis 2021.08.02.21261465. 10.1093/cid/ciab909 [DOI] [PMC free article] [PubMed]
- 109.Callaway E. Remember Beta? New data reveal variant’s deadly powers Nature. 2021 doi: 10.1038/D41586-021-02177-3. [DOI] [PubMed] [Google Scholar]
- 110.Madhi SA, Baillie V, Cutland CL, et al. Efficacy of the ChAdOx1 nCoV-19 Covid-19 Vaccine against the B.1.351 Variant. N Engl J Med. 2021;384:1885–1898. doi: 10.1056/NEJMOA2102214/SUPPL_FILE/NEJMOA2102214_DATA-SHARING.PDF. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Wang Z, Schmidt F, Weisblum Y, et al. (2021) mRNA vaccine-elicited antibodies to SARS-CoV-2 and circulating variants. Nat. 2021;5927855(592):616–622. doi: 10.1038/s41586-021-03324-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wang P, Nair MS, Liu L, et al. Antibody resistance of SARS-CoV-2 variants B.1.351 and B.1.1.7. Nature. 2021;593:130–135. doi: 10.1038/S41586-021-03398-2. [DOI] [PubMed] [Google Scholar]
- 113.Reincke SM, Yuan M, Kornau HC, et al (2022) SARS-CoV-2 Beta variant infection elicits potent lineage-specific and cross-reactive antibodies. Science (80- ) 375:782–787. 10.1126/SCIENCE.ABM5835/SUPPL_FILE/SCIENCE.ABM5835_SM_MDAR_REPRODUCIBILITY_CHECKLIST.PDF [DOI] [PMC free article] [PubMed]
- 114.Liu Y, Liu J, Plante KS, et al. (2021) The N501Y spike substitution enhances SARS-CoV-2 infection and transmission. Nat. 2021;6027896(602):294–299. doi: 10.1038/s41586-021-04245-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Challen R, Brooks-Pollock E, Read JM, et al. Risk of mortality in patients infected with SARS-CoV-2 variant of concern 202012/1: matched cohort study. BMJ. 2021 doi: 10.1136/BMJ.N579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Allen H, Vusirikala A, Flannagan J, et al (2022) Household transmission of COVID-19 cases associated with SARS-CoV-2 delta variant (B.1.617.2): national case-control study. Lancet Reg Heal Eur 12. 10.1016/J.LANEPE.2021.100252/ATTACHMENT/F4B45EE0-D911-41E2-9313-241D342AAED9/MMC2.DOCX [DOI] [PMC free article] [PubMed]
- 117.Hoffmann M, Hofmann-Winkler H, Krüger N, et al (2021) SARS-CoV-2 variant B.1.617 is resistant to bamlanivimab and evades antibodies induced by infection and vaccination. Cell Rep 36:109415. 10.1016/J.CELREP.2021.109415/ATTACHMENT/39B1E666-BE93-41A9-A42B-526240B8B305/MMC1.PDF [DOI] [PMC free article] [PubMed]
- 118.Hoffmann M, Arora P, Groß R, et al. SARS-CoV-2 variants B.1.351 and P.1 escape from neutralizing antibodies. Cell. 2021;184:2384–2393.e12. doi: 10.1016/j.cell.2021.03.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Kumar S, Thambiraja TS, Karuppanan K, Subramaniam G. Omicron and Delta variant of SARS-CoV-2: a comparative computational study of spike protein. J Med Virol. 2022;94:1641–1649. doi: 10.1002/jmv.27526. [DOI] [PubMed] [Google Scholar]
- 120.Eyre DW, Taylor D, Purver M, et al. Effect of Covid-19 vaccination on transmission of alpha and delta variants. N Engl J Med. 2022;386:744–756. doi: 10.1056/nejmoa2116597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Thangaraj JWV, Yadav P, Kumar CG, et al. Predominance of delta variant among the COVID-19 vaccinated and unvaccinated individuals, India, May 2021. J Infect. 2022;84:94–118. doi: 10.1016/J.JINF.2021.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Wang P, Casner RG, Nair MS, et al. Increased resistance of SARS-CoV-2 variant P.1 to antibody neutralization. Cell Host Microbe. 2021;29:747–751.e4. doi: 10.1016/j.chom.2021.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.de Souza WM, Amorim MR, Sesti-Costa R, et al (2021) Levels of SARS-CoV-2 Lineage P.1 Neutralization by antibodies elicited after natural infection and vaccination. SSRN Electron J. 10.2139/ssrn.3793486
- 124.Dejnirattisai W, Zhou D, Supasa P, et al. Antibody evasion by the P.1 strain of SARS-CoV-2. Cell. 2021;184:2939–2954.e9. doi: 10.1016/j.cell.2021.03.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Los Alamos National Laboratory (2021) Tracking SARS-CoV-2 spike mutations. https://www.lanl.gov/updates/sars-cov-2-variant.php. Accessed 21 Mar 2021
- 126.Xu W, Wang M, Yu D, Zhang X. Variations in SARS-CoV-2 spike protein cell epitopes and glycosylation profiles during global transmission course of COVID-19. Front Immunol. 2020 doi: 10.3389/fimmu.2020.565278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Ozono S, Zhang Y, Ode H, et al. SARS-CoV-2 D614G spike mutation increases entry efficiency with enhanced ACE2-binding affinity. Nat Commun. 2021;12:848. doi: 10.1038/s41467-021-21118-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Zhang L, Jackson CB, Mou H, et al. SARS-CoV-2 spike-protein D614G mutation increases virion spike density and infectivity. Nat Commun. 2020;11:6013. doi: 10.1038/s41467-020-19808-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Korber B, Fischer WM, Gnanakaran S, et al (2020) Spike mutation pipeline reveals the emergence of a more transmissible form of SARS-CoV-2. bioRxiv
- 130.Hou YJ, Chiba S, Halfmann P, et al (2021) SARS-CoV-2 D614G variant exhibits efficient replication ex vivo and transmission in vivo. Science (80- ) 370:1464–1468. 10.1126/science.abe8499 [DOI] [PMC free article] [PubMed]
- 131.Wibmer CK, Ayres F, Hermanus T, et al. SARS-CoV-2 501Y.V2 escapes neutralization by South African COVID-19 donor plasma. Nat Med. 2021;27:622–625. doi: 10.1038/s41591-021-01285-x. [DOI] [PubMed] [Google Scholar]
- 132.Sun P, Lu X, Xu C, et al. Understanding of COVID-19 based on current evidence. J Med Virol. 2020;92:548–551. doi: 10.1002/jmv.25722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Mathavan S, Kumar S (2020) Evaluation Of The Effect Of D614G, N501Y AND S477N mutation in SARS-CoV-2 through computational approach. Preprints 2020120710. 10.20944/PREPRINTS202012.0710.V1
- 134.AL Alaofi M Shahid (2021) Mutations of SARS-CoV-2 RBD may alter its molecular structure to improve its infection efficiency. Biomol, 2021 Vol 11 Page 1273 11 1273 10.3390/BIOM11091273 [DOI] [PMC free article] [PubMed]
- 135.Zahradník J, Marciano S, Shemesh M, et al. SARS-CoV-2 variant prediction and antiviral drug design are enabled by RBD in vitro evolution. Nat Microbiol. 2021;6:1188–1198. doi: 10.1038/s41564-021-00954-4. [DOI] [PubMed] [Google Scholar]
- 136.Cherian S, Potdar V, Jadhav S, et al. Sars-cov-2 spike mutations, l452r, t478k, e484q and p681r, in the second wave of covid-19 in Maharashtra. India Microorganisms. 2021;9:1542. doi: 10.3390/MICROORGANISMS9071542/S1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Di Giacomo S, Mercatelli D, Rakhimov A, Giorgi FM. Preliminary report on severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Spike mutation T478K. J Med Virol. 2021;93:5638–5643. doi: 10.1002/JMV.27062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Jhun H, Park HY, Hisham Y, et al (2021) SARS-CoV-2 Delta (B.1.617.2) Variant: a unique T478K mutation in receptor binding motif (RBM) of spike gene. Immune Netw 21. 10.4110/IN.2021.21.E32 [DOI] [PMC free article] [PubMed]
- 139.Aoki A, Adachi H, Mori Y, et al. A rapid screening assay for L452R and T478K spike mutations in SARS-CoV-2 Delta variant using high-resolution melting analysis. J Toxicol Sci. 2021;46:471–476. doi: 10.2131/jts.46.471. [DOI] [PubMed] [Google Scholar]
- 140.A Wilhelm T Toptan C Pallas (2021) Antibody-mediated neutralization of authentic SARS-CoV-2 B.1.617 Variants Harboring L452R and T478K, E484Q. Viruses, et al 2021 Vol 13 Page 1693 13 1693 10.3390/V13091693 [DOI] [PMC free article] [PubMed]
- 141.Moore JP, Offit PA. SARS-CoV-2 vaccines and the growing threat of viral variants. JAMA - J Am Med Assoc. 2021;325:821–822. doi: 10.1001/jama.2021.1114. [DOI] [PubMed] [Google Scholar]
- 142.Garcia-Beltran WF, Lam EC, St. Denis K, et al (2021) Multiple SARS-CoV-2 variants escape neutralization by vaccine-induced humoral immunity. Cell. 10.1016/j.cell.2021.03.013 [DOI] [PMC free article] [PubMed]
- 143.Souza WM, Amorim MR, Sesti-Costa R, et al. Neutralisation of SARS-CoV-2 lineage P.1 by antibodies elicited through natural SARS-CoV-2 infection or vaccination with an inactivated SARS-CoV-2 vaccine: an immunological study. The Lancet Microbe. 2021;2:e527–e535. doi: 10.1016/s2666-5247(21)00129-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Furuse Y. Genomic sequencing effort for SARS-CoV-2 by country during the pandemic. Int J Infect Dis. 2021;103:305–307. doi: 10.1016/J.IJID.2020.12.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Fundação Oswaldo Cruz (2021) Dashboard – Genomahcov – Fiocruz. http://www.genomahcov.fiocruz.br/dashboard/. Accessed 27 Feb 2022
- 146.Pulliam JRC, Van Schalkwyk C, Govender N, et al (2021) Increased risk of SARS-CoV-2 reinfection associated with emergence of the Omicron variant in South Africa. Preprints. 10.1101/2021.11.11.21266068
- 147.Chen J, Wang R, Gilby NB, Wei G-WW. Omicron Variant (B.1.1.529): Infectivity, vaccine breakthrough, and antibody resistance. J Chem Inf Model. 2022;62:412–422. doi: 10.1021/acs.jcim.1c01451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.OMS (2021) Update on Omicron. https://www.who.int/news/item/28-11-2021-update-on-omicron. Accessed 9 Dec 2021
- 149.OMS (2021) Enhancing readiness for Omicron (B.1.1.529): technical brief and priority actions for member states
- 150.Barton MI, Macgowan S, Kutuzov M, et al. Effects of common mutations in the sars-cov-2 spike rbd and its ligand the human ace2 receptor on binding affinity and kinetics. Elife. 2021 doi: 10.7554/eLife.70658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.First confirmed case of omicron variant detected in the United States | CDC Online Newsroom | CDC. https://www.cdc.gov/media/releases/2021/s1201-omicron-variant.html. Accessed 8 Dec 2021
- 152.Classification of Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern. https://www.who.int/news/item/26-11-2021-classification-of-omicron-(b.1.1.529)-sars-cov-2-variant-of-concern. Accessed 1 Mar 2022
- 153.Klinakis A, Cournia Z, Rampias T. N-terminal domain mutations of the spike protein are structurally implicated in epitope recognition in emerging SARS-CoV-2 strains. Comput Struct Biotechnol J. 2021;19:5556–5567. doi: 10.1016/j.csbj.2021.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Nörz D, Grunwald M, Olearo F, et al (2021) Evaluation of a fully automated high-throughput SARS-CoV-2 multiplex qPCR assay with built-in screening functionality for del-HV69/70- and N501Y variants such as B.1.1.7. J Clin Virol 141:104894. 10.1016/J.JCV.2021.104894 [DOI] [PMC free article] [PubMed]
- 155.Takano K, Watanabe Y, Hariu M, Seki M. Detection of representative mutant strains and a case of prolonged infection by SARS-CoV-2 with spike 69/70 deletion in Japan. Infect Drug Resist. 2021;14:2579. doi: 10.2147/IDR.S320658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Kemp SA, Collier DA, Datir RP, et al. (2021) SARS-CoV-2 evolution during treatment of chronic infection. Nat. 2021;5927853(592):277–282. doi: 10.1038/s41586-021-03291-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.McCarthy KR, Rennick LJ, Nambulli S, et al (2021) Recurrent deletions in the SARS-CoV-2 spike glycoprotein drive antibody escape. Science (80- ) 371:1139–1142. 10.1126/science.abf6950 [DOI] [PMC free article] [PubMed]
- 158.Qin S, Cui M, Sun S, et al (2021) Genome characterization and potential risk assessment of the novel SARS-CoV-2 Variant Omicron (B.1.1.529). Zoonoses 1. 10.15212/ZOONOSES-2021-0024
- 159.Nörz D, Pfefferle S, Grunwald M, et al. Modifying a diagnostic SARS-CoV-2 spike PCR to turn a Del69/70 dropout into a discriminatory on-target assay. J Mol Diagnostics. 2021;23:777–778. doi: 10.1016/J.JMOLDX.2021.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Vega-Magaña N, Sánchez-Sánchez R, Hernández-Bello J, et al. RT-qPCR assays for rapid detection of the N501Y, 69–70del, K417N, and E484K SARS-CoV-2 mutations: a screening strategy to identify variants with clinical impact. Front Cell Infect Microbiol. 2021;11:434. doi: 10.3389/FCIMB.2021.672562/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Xie X, Liu Y, Liu J, et al. (2021) Neutralization of SARS-CoV-2 spike 69/70 deletion, E484K and N501Y variants by BNT162b2 vaccine-elicited sera. Nat Med. 2021;274(27):620–621. doi: 10.1038/s41591-021-01270-4. [DOI] [PubMed] [Google Scholar]
- 162.Wrapp D, Wang N, Corbett KS, et al (2020) Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science (80- ) 367:1260–1263. 10.1126/science.abb2507 [DOI] [PMC free article] [PubMed]
- 163.West AP, Wertheim JO, Wang JC, et al (2021) Detection and characterization of the SARS-CoV-2 lineage B.1.526 in New York. Nat Commun 12. 10.1038/s41467-021-25168-4 [DOI] [PMC free article] [PubMed]
- 164.Kannan SR, Spratt AN, Cohen AR, et al. Evolutionary analysis of the Delta and Delta Plus variants of the SARS-CoV-2 viruses. J Autoimmun. 2021;124:102715. doi: 10.1016/J.JAUT.2021.102715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Aktas E. Bioinformatics analysis unveils certain mutations implicated in spike structure damage and ligand-binding site of severe acute respiratory syndrome coronavirus 2. Bioinform Biol Insights. 2021;15:11779322211018200. doi: 10.1177/11779322211018200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Hacisuleyman E, Hale C, Saito Y, et al. Vaccine breakthrough infections with SARS-CoV-2 variants. N Engl J Med. 2021;384:2212–2218. doi: 10.1056/NEJMOA2105000/SUPPL_FILE/NEJMOA2105000_DISCLOSURES.PDF. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Srivastava M, Hall D, Omoru OB, et al. Mutational landscape and interaction of sars-cov-2 with host cellular components. Microorganisms. 2021;9:1794. doi: 10.3390/microorganisms9091794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Mishra T, Joshi G, Kumar A, et al (2021) B.1.617.3 SARS CoV-2 spike E156G/Δ157–158 mutations contribute to reduced neutralization sensitivity and increased infectivity. bioRxiv. 10.1101/2021.10.04.463028
- 169.Shen L, Triche TJ, Bard JD, et al (2021) Spike protein NTD mutation G142D in SARS-CoV-2 Delta VOC lineages is associated with frequent back mutations, increased viral loads, and immune evasion. medRxiv. 10.1101/2021.09.12.21263475
- 170.Verdurme L, Danesh G, Trombert-Paolantoni S, et al (2021) Spread of SARS-CoV-2 Delta variant infections bearing the S:E484Q and S:T95I mutations in July and August 2021 in France. medRxiv 2021.09.13.21263371. 10.1101/2021.09.13.21263371
- 171.Haslwanter D, Dieterle ME, Wec AZ, et al. A combination of receptor-binding domain and N-terminal domain neutralizing antibodies limits the generation of SARS-CoV-2 spike neutralization-escape mutants. MBio. 2021 doi: 10.1128/MBIO.02473-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Resende PC, Gräf T, Neto LGL, et al (2021) Identification of a new B.1.1.33 SARS-CoV-2 Variant of Interest (VOI) circulating in Brazil with mutation E484K and multiple deletions in the amino (N)-terminal domain of the spike protein. Virological.com April 7
- 173.Lodi L, Moriondo M, Pucci A, et al. Chronic asymptomatic severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Infection in the immunocompromised patient: new challenges and urgent needs. Clin Infect Dis. 2021 doi: 10.1093/CID/CIAB538. [DOI] [PubMed] [Google Scholar]
- 174.Yeh T-Y, Contreras GP (2021) Tajima D test accurately forecasts Omicron / COVID-19 outbreak. medRxiv. 10.1101/2021.12.02.21267185
- 175.Venkatakrishnan A, Anand P, Lenehan PJ, et al (2021) Omicron variant of SARS-CoV-2 harbors a unique insertion mutation of putative viral or human genomic origin. OSF Prepr. 10.31219/osf.io/f7txy
- 176.Zimerman RA, Aline P, Ferrareze G, et al (2021) Comparative genomics and characterization of SARS-CoV-2 P.1 (Gamma) Variant of concern (VOC) from Amazonas, Brazil comparative genomics and characterization of SARS-CoV-2 P.1 (Gamma) Variant of. medRxiv Prepr Serv Heal Sci. 10.1101/2021.10.30.21265694
- 177.Cao Y, Wang J, Jian F, et al. Omicron escapes the majority of existing SARS-CoV-2 neutralizing antibodies. Nature. 2022;602:657–663. doi: 10.1038/s41586-021-04385-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Oróstica KY, Contreras S, Mohr SB, et al (2021) Mutational signatures and transmissibility of SARS-CoV-2 Gamma and Lambda variants [DOI] [PMC free article] [PubMed]
- 179.Long SW, Olsen RJ, Christensen PA, et al. Molecular architecture of early dissemination and massive second wave of the SARS-CoV-2 virus in a major metropolitan area. MBio. 2020;11:1–30. doi: 10.1128/MBIO.02707-20/SUPPL_FILE/MBIO.02707-20-SF006.PDF. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Chen J, Gao K, Wang R, Wei GW. Prediction and mitigation of mutation threats to COVID-19 vaccines and antibody therapies. Chem Sci. 2021;12:6929–6948. doi: 10.1039/d1sc01203g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Fratev F. N501Y and K417N mutations in the spike protein of SARS-CoV-2 alter the interactions with Both hACE2 and human-derived antibody: a free energy of perturbation retrospective study. J Chem Inf Model. 2021;61:6079–6084. doi: 10.1021/acs.jcim.1c01242. [DOI] [PubMed] [Google Scholar]
- 182.Wang R, Chen J, Gao K, Wei GW. Vaccine-escape and fast-growing mutations in the United Kingdom, the United States, Singapore, Spain, India, and other COVID-19-devastated countries. Genomics. 2021;113:2158–2170. doi: 10.1016/J.YGENO.2021.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Verma J, Subbarao N. Insilico study on the effect of SARS-CoV-2 RBD hotspot mutants’ interaction with ACE2 to understand the binding affinity and stability. Virology. 2021;561:107–116. doi: 10.1016/j.virol.2021.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Genovese L, Zaccaria M, Farzan M, et al (2021) Investigating the mutational landscape of the SARS-CoV-2 Omicron variant via ab initio quantum mechanical modeling. bioRxiv. 10.1101/2021.12.01.470748
- 185.Rani PR, Imran M, Lakshmi JV, et al. Symptomatic reinfection of SARS-CoV-2 with spike protein variant N440K associated with immune escape. J Med Virol. 2021;93:4163–4165. doi: 10.1002/jmv.26997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Weisblum Y, Schmidt F, Zhang F, et al. Escape from neutralizing antibodies by SARS-CoV-2 spike protein variants. Elife. 2020;9:1. doi: 10.7554/eLife.61312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Focosi D, Maggi F. Neutralising antibody escape of SARS-CoV-2 spike protein: risk assessment for antibody-based Covid-19 therapeutics and vaccines. Rev Med Virol. 2021;31:e2231. doi: 10.1002/RMV.2231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Singh A, Steinkellner G, Köchl K, et al. (2021) Serine 477 plays a crucial role in the interaction of the SARS-CoV-2 spike protein with the human receptor ACE2. Sci Reports. 2021;111(11):1–11. doi: 10.1038/s41598-021-83761-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Peiffer-Smadja N, Bridier-Nahmias A, Ferré VM, et al. Emergence of e484k mutation following bamlanivimab monotherapy among high-risk patients infected with the alpha variant of sars-cov-2. Viruses. 2021;13:1642. doi: 10.3390/V13081642/S1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Laurini E, Marson D, Aulic S, et al. (2021) Molecular rationale for SARS-CoV-2 spike circulating mutations able to escape bamlanivimab and etesevimab monoclonal antibodies. Sci Reports. 2021;111(11):1–20. doi: 10.1038/s41598-021-99827-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Shah M, Woo HG (2022) Omicron: a heavily mutated SARS-CoV-2 variant exhibits stronger binding to ACE2 and potently escapes approved COVID-19 therapeutic antibodies. Front Immunol 12:2021.12.04.471200. 10.3389/fimmu.2021.830527 [DOI] [PMC free article] [PubMed]
- 192.Starr TN, Greaney AJ, Addetia A, et al (2021) Prospective mapping of viral mutations that escape antibodies used to treat COVID-19. Science (80- ) 371:850–854. 10.1126/science.abf9302 [DOI] [PMC free article] [PubMed]
- 193.Sarkar R, Lo M, Saha R, et al (2021) S glycoprotein diversity of the Omicron variant. medRxiv. 10.1101/2021.12.04.21267284
- 194.Li Y, Zhang Z, Yang L, et al (2020) The MERS-CoV Receptor DPP4 as a candidate binding target of the SARS-CoV-2 spike. iScience 23:101160. 10.1016/j.isci.2020.101160 [DOI] [PMC free article] [PubMed]
- 195.Neuman BW, Brashear WA, Brun M, et al. Case report: paucisymptomatic college-age population as a reservoir for potentially neutralization-resistant severe acute respiratory syndrome Coronavirus 2 variants. Am J Trop Med Hyg. 2021;105:1227–1229. doi: 10.4269/ajtmh.21-0542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Muecksch F, Weisblum Y, Barnes CO, et al (2021) Development of potency, breadth and resilience to viral escape mutations in SARS-CoV-2 neutralizing antibodies. bioRxiv. 10.1101/2021.03.07.434227 [DOI] [PMC free article] [PubMed]
- 197.Focosi D, Novazzi F, Genoni A, et al. Emergence of sars-cov-2 spike protein escape mutation q493r after treatment for covid-19. Emerg Infect Dis. 2021;27:2728–2731. doi: 10.3201/eid2710.211538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Guigon A, Faure E, Lemaire C, et al. Emergence of Q493R mutation in SARS-CoV-2 spike protein during bamlanivimab/etesevimab treatment and resistance to viral clearance. J Infect. 2021 doi: 10.1016/J.JINF.2021.08.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Vellas C, Del Bello A, Debard A, et al. Influence of treatment with neutralizing monoclonal antibodies on the SARS-CoV-2 nasopharyngeal load and quasispecies. Clin Microbiol Infect. 2022;28:139.e5–139.e8. doi: 10.1016/j.cmi.2021.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Truffot A, Andréani J, Le Maréchal M, et al. Sars-cov-2 variants in immunocompromised patient given antibody monotherapy. Emerg Infect Dis. 2021;27:2725–2728. doi: 10.3201/eid2710.211509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Fratev F (2021) The High Transmission of SARS-CoV-2 Omicron (B.1.1.529) Variant is Not Only Due to Its hACE2 binding: A Free Energy of Perturbation Study. bioRxiv. 10.1101/2021.12.04.471246
- 202.Ortega JT, Pujol FH, Jastrzebska B, Rangel HR (2021) Mutations in the SARS-CoV-2 spike protein modulate the virus affinity to the human ACE2 receptor, an in silico analysis. EXCLI J 20:585–600. 10.17179/EXCLI2021-3471 [DOI] [PMC free article] [PubMed]
- 203.Zahradník J, Marciano S, Shemesh M, et al (2021) SARS-CoV-2 RBD in vitro evolution follows contagious mutation spread, yet generates an able infection inhibitor. bioRxiv. 10.1101/2021.01.06.425392
- 204.Lennerstrand J, Palanisamy N. Global prevalence of adaptive and prolonged infections’ mutations in the receptor-binding domain of the sars-cov-2 spike protein. Viruses. 2021;13:1974. doi: 10.3390/V13101974/S1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Kuiper MJ, Wilson L, Mangalaganesh S, et al (2021) But Mouse, you are not alone: on some severe acute respiratory syndrome coronavirus 2 variants infecting mice. bioRxiv. 10.1101/2021.08.04.455042 [DOI] [PMC free article] [PubMed]
- 206.Kramer KJ, Johnson NV, Shiakolas AR, et al. Potent neutralization of SARS-CoV-2 variants of concern by an antibody with an uncommon genetic signature and structural mode of spike recognition. Cell Rep. 2021 doi: 10.1016/j.celrep.2021.109784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.L Queirós-Reis PG Silva da J Gonçalves (2021) SARS-CoV-2 virus−host interaction: currently available structures and implications of variant emergence on infectivity and immune response. Int J Mol Sci, et al 2021 Vol 22 Page 10836 22 10836 10.3390/IJMS221910836 [DOI] [PMC free article] [PubMed]
- 208.Ahmed S, Bhasin M, Manjunath K, Varadarajan R (2022) Prediction of residue-specific contributions to binding and thermal stability using yeast surface display. Front Mol Biosci 8:2021.05.31.446445. 10.3389/fmolb.2021.800819 [DOI] [PMC free article] [PubMed]
- 209.Valdes-Balbin Y, Santana-Mederos D, Paquet F, et al. Molecular aspects concerning the use of the SARS-CoV-2 receptor binding domain as a target for preventive vaccines. ACS Cent Sci. 2021;7:757–767. doi: 10.1021/acscentsci.1c00216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Xue T, Wu W, Guo N, et al. Single point mutations can potentially enhance infectivity of SARS-CoV-2 revealed by in silico affinity maturation and SPR assay. RSC Adv. 2021;11:14737–14745. doi: 10.1039/D1RA00426C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Liu H, Zhang Q, Wei P, et al. The basis of a more contagious 501Y.V1 variant of SARS-CoV-2. Cell Res. 2021;31:720–722. doi: 10.1038/s41422-021-00496-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Conceicao C, Thakur N, Human S, et al. The SARS-CoV-2 spike protein has a broad tropism for mammalian ACE2 proteins. PLOS Biol. 2020;18:e3001016. doi: 10.1371/JOURNAL.PBIO.3001016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Scheepers C, Everatt J, Amoako DG, et al (2021) The continuous evolution of SARS-CoV-2 in South Africa: a new lineage with rapid accumulation of mutations of concern and global detection. medRxiv. 10.1101/2021.08.20.21262342
- 214.Braun KM, Moreno GK, Halfmann PJ, et al. Transmission of SARS-CoV-2 in domestic cats imposes a narrow bottleneck. PLOS Pathog. 2021;17:e1009373. doi: 10.1371/JOURNAL.PPAT.1009373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Pulakuntla S, Lokhande KB, Padmavathi P, et al. Mutational analysis in international isolates and drug repurposing against SARS-CoV-2 spike protein: molecular docking and simulation approach. VirusDisease. 2021;32:690–702. doi: 10.1007/S13337-021-00720-4/FIGURES/4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Escalera A, Gonzalez-Reiche AS, Aslam S, et al (2021) SARS-CoV-2 variants of concern have acquired mutations associated with an increased spike cleavage. bioRxiv. 10.1101/2021.08.05.455290
- 217.Gupta D, Sharma P, Singh M, et al. (2021) Structural and functional insights into the spike protein mutations of emerging SARS-CoV-2 variants. Cell Mol Life Sci. 2021;7824(78):7967–7989. doi: 10.1007/S00018-021-04008-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Williamson MK, Hamilton F, Hutchings S, et al (2021) Chronic SARS-CoV-2 infection and viral evolution in a hypogammaglobulinaemic individual. medRxiv. 10.1101/2021.05.31.21257591
- 219.Shiliaev N, Lukash T, Palchevska O, et al. Natural and recombinant SARS-CoV-2 isolates rapidly evolve in vitro to higher infectivity through more efficient binding to heparan sulfate and reduced S1/S2 cleavage. J Virol. 2021 doi: 10.1128/JVI.01357-21/ASSET/834DCF96-5CD9-4BF7-A649-CA0D1BFFB959/ASSETS/IMAGES/LARGE/JVI.01357-21-F011.JPG. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Sahoo JP, Samal KC. World on Alert: WHO designated South African new COVID strain (Omicron/B.1.1.529) as a variant of concern. Biot Res Today. 2021;3:1086–1088. [Google Scholar]
- 221.Dieterle ME, Haslwanter D, Bortz RH, et al. A replication-competent vesicular stomatitis virus for studies of SARS-CoV-2 spike-mediated cell entry and its inhibition. Cell Host Microbe. 2020;28:486–496.e6. doi: 10.1016/j.chom.2020.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Deere JD, Carroll TD, Dutra J, et al. SARS-CoV-2 infection of rhesus macaques treated early with human COVID-19 convalescent plasma. Microbiol Spectr. 2021 doi: 10.1128/SPECTRUM.01397-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Islam SR, Prusty D, Manna SK (2021) Structural basis of fitness of emerging SARS-COV-2 variants and considerations for screening, testing and surveillance strategy to contain their threat. medRxiv. 10.1101/2021.01.28.21250666
- 224.Focosi D. SARS-CoV-2 spike protein convergent evolution. Cham: Springer International Publishing; 2021. [Google Scholar]
- 225.Mertens J, Coppens J, Loens K, et al. Monitoring the SARS-CoV-2 pandemic: screening algorithm with single nucleotide polymorphism detection for the rapid identification of established and emerging variants. Clin Microbiol Infect. 2022;28:124–129. doi: 10.1016/j.cmi.2021.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Naveca F, Nascimento V, Souza V, et al (2021) Emergence and spread of SARS-CoV-2 P. 1 (Gamma) lineage variants carrying spike mutations ?141–144, N679K or P681H during persistent viral circulation in Amazonas, Brazil. Virological.org 420:75–77
- 227.Yang X-J (2021) SARS-COV-2 C.1.2 variant is highly mutated but may possess reduced affinity for ACE2 receptor. bioRxiv. 10.1101/2021.10.16.464644
- 228.Fourati S, Decousser J-W, Khouider S, et al. Novel SARS-CoV-2 Variant Derived from Clade 19B, France. Emerg Infect Dis. 2021;27:1540–1543. doi: 10.3201/eid2705.210324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Calvignac-Spencer S, Budt M, Huska M, et al (2021) Rise and fall of sars-cov-2 lineage a.27 in germany. Viruses 13:1491. 10.3390/v13081491 [DOI] [PMC free article] [PubMed]
- 230.Liu L, Luo T, Lei G, et al Notes from the Field. 10.46234/ccdcw2021.155
- 231.Sadek A, Zaha D, Salama M, et al (2021) Structural insights of SARS-CoV-2 spike protein from delta and omicron variants. bioRxiv 2021.12.08.471777. 10.1101/2021.12.08.471777
- 232.Zhang L, Li Q, Liang Z, et al. The significant immune escape of pseudotyped SARS-CoV-2 variant Omicron. Emerg Microbes Infect. 2022;11:1–5. doi: 10.1080/22221751.2021.2017757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Wilhelm A, Widera M, Grikscheit K, et al (2021) Reduced neutralization of SARS-CoV-2 omicron variant by vaccine sera and monoclonal antibodies. medRxiv. 10.1101/2021.12.07.21267432
- 234.Pfizer (2021) Pfizer and Biontech Provide Update on Omicron Variant. In: Pfizer. https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-provide-update-omicron-variant. Accessed 15 Dec 2021
- 235.Cele S, Jackson L, Khoury DS, et al. Omicron extensively but incompletely escapes Pfizer BNT162b2 neutralization. Nature. 2022;602:654–656. doi: 10.1038/s41586-021-04387-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Lancet T. Access to COVID-19 vaccines: looking beyond COVAX. Lancet (London, England) 2021;397:941. doi: 10.1016/S0140-6736(21)00617-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.World Health Organization (2021) WHO Coronavirus (COVID-19) Dashboard. WHO Coronavirus (COVID-19) Dashboard With Vaccination Data. Who 1–5
- 238.Asundi A, O’Leary C, Bhadelia N. Global COVID-19 vaccine inequity: The scope, the impact, and the challenges. Cell Host Microbe. 2021;29:1036–1039. doi: 10.1016/J.CHOM.2021.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Wouters OJ, Shadlen KC, Salcher-Konrad M, et al. Challenges in ensuring global access to COVID-19 vaccines: production, affordability, allocation, and deployment. Lancet (London, England) 2021;397:1023. doi: 10.1016/S0140-6736(21)00306-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Massinga Loembé M, Nkengasong JN. COVID-19 vaccine access in Africa: Global distribution, vaccine platforms, and challenges ahead. Immunity. 2021;54:1353–1362. doi: 10.1016/J.IMMUNI.2021.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Kim JH, Marks F. Clemens JD (2021) Looking beyond COVID-19 vaccine phase 3 trials. Nat Med. 2021;272(27):205–211. doi: 10.1038/s41591-021-01230-y. [DOI] [PubMed] [Google Scholar]
- 242.Hussain A, Hasan A, Nejadi Babadaei MM, et al (2020) Targeting SARS-CoV2 spike protein receptor binding domain by therapeutic antibodies. Biomed. Pharmacother 130 [DOI] [PMC free article] [PubMed]
- 243.Pilishvili T, Gierke R, Fleming-Dutra KE, et al (2021) Effectiveness of mRNA Covid-19 Vaccine among U.S. Health Care Personnel. N Engl J Med 385:e90. 10.1056/NEJMOA2106599/SUPPL_FILE/NEJMOA2106599_DISCLOSURES.PDF [DOI] [PMC free article] [PubMed]
- 244.Liu Y, Liu J, Xia H, et al. Neutralizing Activity of BNT162b2-Elicited Serum. N Engl J Med. 2021;384:1466–1468. doi: 10.1056/NEJMC2102017. [DOI] [PMC free article] [PubMed] [Google Scholar]