Abstract
Purpose
Adult-onset disease risks associated with carriers of recessive disease have and will continue to be identified. As carrier screening becomes more broadly utilized, providers face the dilemma of whether they should discuss these risks during discussions with prospective parents. This study aimed to understand whether preconception/prenatal genetic counselors (PPGCs) were aware of the risk of Parkinson disease in carriers of, and persons with, Gaucher disease and the reasons behind choosing whether to discuss this risk with patients.
Methods
Eligible participants included board-certified or board-eligible genetic counselors who had counseled preconception/prenatal patients within the past 3 years. An online survey was distributed via the National Society of Genetic Counselors in November of 2017.
Results
One hundred twenty genetic counselors completed the quantitative survey, distributed in Fall of 2017. While the majority of respondents knew of the Gaucher-related Parkinson’s link (n = 78; 65%), just over one-third reported discussing it in preconception/prenatal settings (n = 30; 38.5%). Respondents reported discussing these links more consistently when disclosing positive results or when the patient/family approached the topic. Respondents cited the lack of professional guidelines as one of the main reasons for not discussing the link.
Conclusion
These results highlight an inconsistency in PPGCs’ discussions of the Parkinson’s risk in Gaucher disease carriers, and the need to develop guidelines regarding these issues to help standardize the care and education of patients.
Supplementary Information
The online version contains supplementary material available at 10.1007/s10815-022-02430-2.
Keywords: Informed consent, Pregnancy, Carrier risks, Secondary findings, Prenatal testing, Carrier screening
Introduction
The American College of Obstetricians and Gynecologists (ACOG) recommends that information about carrier screening be provided to every pregnant woman [1]. However, while the goal of carrier screening is to provide individuals and couples with information about their risk of having a child with a serious genetic condition, carrier screening also has the potential to reveal health risks to the individuals undergoing carrier screening themselves. As the number of conditions for which individuals may be offered carrier screening for increases, the potential for discovering carriers at risk for symptoms also increases.
Historically, carrier screening was provided to individuals based on their self-reported race or ethnicity. However, with advances in technology and costs of sequencing decreasing, there has been a shift from single-gene ethnicity-based screening to pan-ethnic expanded carrier screening where individuals are screened for hundreds of genes simultaneously. These technological advances along with increased access to genetic testing has led to the discovery of risks associated with being a carrier of certain diseases. One example of this is fragile X syndrome in which premutation carriers are at risk for primary ovarian insufficiency (FXPOI; 20% lifetime risk in females) and/or tremor-ataxia syndrome (FXTAS; 17% of females and 46% of males older than 50) [2], as well as neuropsychiatric illness (FXAND) [3]. Over the years, additional risks in carriers of both autosomal recessive and X-linked diseases have been identified, such as the risk of cardiomyopathy in carriers of Duchenne and Becker muscular dystrophy, the risk of myelopathy in carriers of X-linked adrenoleukodystrophy and the increased risk of cancer in carriers for ataxia telangiectasia (ATM), Nijmegen breakage syndrome (NBN), Fanconi anemia type C, and Bloom syndrome [33, 34,4–7]. As carrier screening becomes more widely utilized and genetic knowledge increases, additional disorders with adult-onset risks to carriers will be identified.
A more recent addition to this list of conditions with risks to carriers is Gaucher disease (GD), an autosomal recessive lysosomal storage disorder that causes a buildup of lipids in macrophages, potentially leading to symptoms such as hepatosplenomegaly, anemia, and bone pain [8]. GD is most seen in the Ashkenazi Jewish population, where the carrier rate is approximately 1 in 16 [9]. Individuals affected with GD, as well as carriers of this condition, have an increased risk to develop GBA-associated Parkinson disease (GBA-PD) [10–12]. Although the actual numbers vary greatly depending on study population and specific GBA variant, a robust study from Gan Or et al. [13] found a 2.84 to 21.29 increased likelihood to develop Parkinson disease (PD) compared to the 1–4% lifetime risk in the general population [14, 15]. Additionally, GBA-PD is associated with a younger age of onset and more dementia symptoms compared to idiopathic PD [11, 16].
Carrier screening is also moving beyond targeted ethnic-based panels to expanded universal panels [17]. With up to thousands of conditions tested on these panels, position statements on carrier screening by the American College of Medical Genetics and Genomics (ACMG) recognizes that a generic consent is needed during pre-test counseling [18, 19]. However, this generic consent is often insufficient for patients who prefer more detailed pre-test counseling [20–22]. Still, the desire for detail must be balanced with the risk of information overload, which can prevent individuals from making informed decisions about testing [23]. Yet even with a generic consent, ACMG recommends that adult-onset conditions—particularly those with risks to the prospective parents—be explicitly stated as a possibility to the individual(s) being tested as part of the consenting process [18]. Whether providers have these discussions with patients is not well documented.
Preconception/prenatal genetic counselors (PPGCs) are specially trained to provide education and support throughout the carrier screening process. Little is known on the knowledge, practices, and attitudes of PPGCs surrounding the potential to identify health risks in prospective parents undergoing carrier screening. This study focuses specifically on the risk of Parkinson’s disease in carriers of and individuals with Gaucher disease, caused by variants in the GBA gene. This GBA-PD link is especially relevant given the high carrier rate for GD within the Ashkenazi Jewish population, the absence of reliable preventive treatments or cures for PD, and its emergent relevance from adult neurology into the preconception/prenatal field.
Methods
Study population and recruitment
The study population consisted of board-certified or board-eligible genetic counselors who had counseled preconception/prenatal patients within the past three years. An email invitation was sent to all members of the National Society of Genetic Counselors (NSGC). Two reminder emails were sent approximately 3 weeks and 6 weeks following the initial email. The survey was open for 6 weeks, from November 10, 2017 through December 22, 2017. As an incentive to participate, a $2 donation was made to St. Jude Children’s Research Hospital for every survey submitted. Study data was collected and managed using REDCap, a secure web-based application hosted through Northwestern University [24].
Survey development
A validated survey tool assessing PPGC’s knowledge in this area was not available. Thus, the survey instrument used in this study (Online Resource 1) was developed by the research committee. The survey consisted of questions aimed to capture information about the main objectives: knowledge, practices, and attitudes regarding carrier screening for GBA and its link to PD. Respondents with knowledge of the GBA-PD link were asked further questions on their practices and attitudes for or against discussing it during carrier screening consultations. All questions were designed in multiple-response multiple choice, yes/no, and free-response formats. This study was deemed exempt by the Northwestern University Institutional Review Board.
Data analysis
Data analysis was conducted with SPSS version 25 and LibreOffice Calc version 6.3. Chi-square, ANOVA, binary logistic regression, or Wilcoxon signed rank tests were performed to determine if demographics and practices were significant predictors of knowledge. Binary logistic regression tests were completed to establish whether demographics were significant predictors of knowledge and practices. Descriptive analyses were performed to summarize the data.
Results
Demographics
A total of 210 individuals began the survey with 120 completing the survey (dropout rate of 42.9%). All points of dropout were on questions at the beginning of a survey page. The majority of respondents had less than seven years of genetic counseling experience (60.8%; mean = 6.95, SD = 7.06) and less than 5 years of experience in the preconception/prenatal field (54.2%). More than 50% (n = 65; select all format) of the respondents worked at an academic medical center during their career in the preconception/prenatal specialty. Demographic characteristics of the cohort are summarized in Table 1.
Table 1.
Demographic information
| N (%) | |
|---|---|
| Years of genetic counseling experience | N = 120 |
| 0–4 years | 59(49.2) |
| 5–9 years | 29 (24.2) |
| 10–14 years | 12(10) |
| 15–19 years | 11 (9.2) |
| > 20 years | 9 (7.5) |
| Years in prenatal practice | N = 120 |
| 0–4 years | 65 (54.2) |
| 5–9 years | 28 (23.3) |
| 10–14 years | 11 (9.2) |
| 15–19 years | 10 (8.3) |
| > 20 years | 6 (5.0) |
| Work setting(s) when in prenatal practice* | N = 185 |
| Academic medical center | 65 (35.5) |
| Private hospital | 34 (18.6) |
| Public hospital | 31 (16.9) |
| Physicians private practice | 23 (12.6) |
| Commercial diagnostic laboratory | 19 (10.4) |
| Telemedicine company | 4 (2.2) |
| Other‡ | 7 (3.8) |
*Individuals were allowed to select more than one work setting
‡Nonprofit medical center, private company (gamete donor facility), private GC practice, government funded hospital (VA), government hospital (VA), biotechnology company providing carrier screening, private corporate clinic
Knowledge
Sixty-five percent (n = 78/120) of respondents were aware of the risk for GBA-PD in GD carriers and affected individuals. Working in a commercial diagnostic laboratory was significantly associated with a higher likelihood of knowledge of the risk for GBA-PD (100%, n = 19, p < 0.000). This group was also more likely to report choosing to discuss the risk for GBA-PD in the preconception/prenatal setting (63.2%, n = 12/19, p = 0.058). Experience with patients who had either a personal or family history of GD was significantly associated with a higher likelihood of knowledge of the link (p = 0.013); however, the same experiences with PD did not predict whether respondents were knowledgeable about the link (p = 0.581).
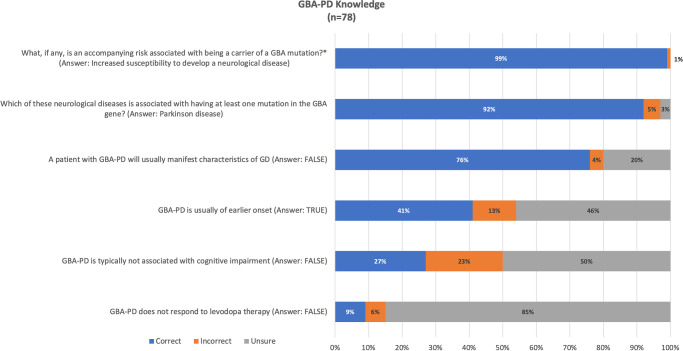
The average GD knowledge score of all one-hundred twenty respondents was 90.83% (out of 5 questions; SD = 0.11) and for PD was 40.21% (out of 4 questions; SD = 0.26). There was not a statistical difference in GD or PD knowledge scores between the cohorts who were aware of the link compared to those who were unaware. The GBA-PD knowledge level per each of the four true or false questions is depicted in Fig. 1 (N = 78).
Fig. 1.
GBA-PD knowledge level. Participants were asked whether they were aware of any accompanying risk associated with being a carrier of GBA mutation and additional questions regarding the type of risk, symptoms, and onset
Practices
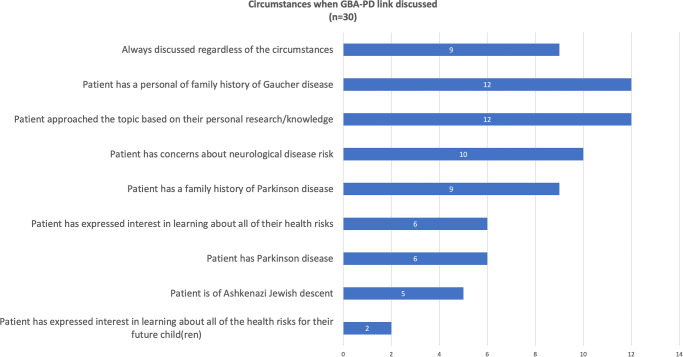
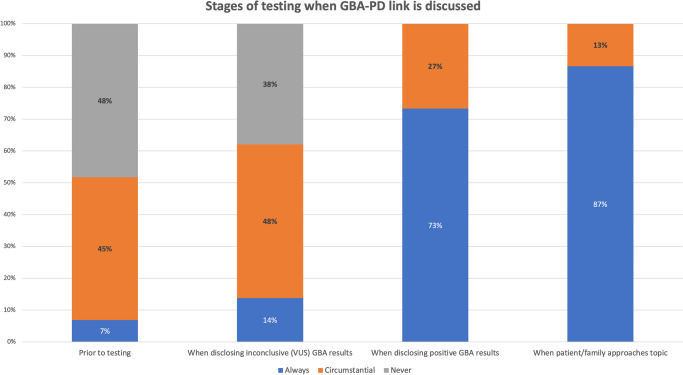
Of the seventy-eight respondents aware of the risk for GBA-PD, 38.5% (n = 30/78) reported discussing the link in the preconception/prenatal setting. A majority of this cohort had prior experience with patients with a personal or family history of GD and PD (n = 29, p < 0.000; n = 22, p = 0.011, respectively). Nine of these 30 respondents (30%) reported they always discuss the link regardless of circumstances. Figure 2 further illustrates the circumstances when GBA-PD is discussed. Respondents rarely discuss the link prior to testing. Respondents most often discuss the link when disclosing positive GBA results or at the point when the patient/family approached the topic. Figure 3 illustrates how often the link was discussed at each stage of counseling. There was no common phrasing used to discuss the link (between “an association has been found,” “there might be,” or “there is” an increased risk). Only two respondents indicated that they provide specific risk estimates (10% and 15%) for developing PD up to the age of 80.
Fig. 2.
Circumstances when the risk of Parkinson’s disease in GBA carriers is discussed. Participants were asked to share under which circumstances they discuss the GBA-PD risk with patients undergoing testing
Fig. 3.
Timing of GBA carrier and Parkinson’s disease risk discussion. Participants were asked at what stage of testing do they discuss the risk of Parkinson’s disease in GBA carriers
Attitudes
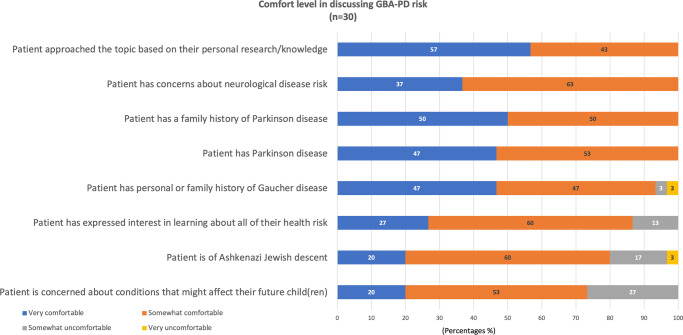
The top three identified reasons for discussing the risk of GBA-PD were the following: (1) the presence of peer-reviewed literature regarding the link (n = 22/30, 73.3%), (2) respecting a patient’s right-to-know (n = 17/30, 56.7%), and (3) learning that patients desire to know of the link (33.3%; N = 30; additional reasons listed in Table 2). Seventy percent or more of this cohort felt at least somewhat comfortable in multiple scenarios when discussing the link. The only two scenarios where respondents entered “very uncomfortable” was when the patient has a personal or family history of Gaucher disease or when they were of Ashkenazi Jewish descent. Figure 4 illustrates these scenarios and comfort levels in detail.
Table 2.
Reasons for discussing GBA-PD risk
| N = 30 | ||
|---|---|---|
| n | % | |
| There is peer-reviewed primary literature which addresses this risk | 22 | 73.3% |
| I respect a patient’s right-to-know | 17 | 56.7% |
| I feel that patients would want to know | 5 | 16.7% |
| I have found that patients want to know | 10 | 33.3% |
| I would want to know myself | 3 | 10.0% |
| Other† | 6 | 20.0% |
†Other explanations verbatim: So that they may get proper medical care if and when they experience symptoms. It is on the genetic testing report and I don’t feel it is responsible not to discuss it. I have had some patients specifically ask me about this topic. I usually don’t bring it up unless they want to know. Their child has 50% chance of also being a carrier, therefore this is relevant to the current or future pregnancy as well. I have found that most patients do NOT want to know, so I do not discuss on a routine basis. However, if a patient asks, that indicates to me that they want to know and I respect this right. Studies have shown that overwhelmingly patients want to know this information
Fig. 4.
Comfort level in discussing GBA-PD link. Participants were asked how comfortable they were with discussing a result that reveals an increased risk for Parkinson disease during preconception/prenatal indications
The top three identified reasons for not discussing the risk of GBA-PD were the following: (1) the GC had not counseled patients with a personal or family history of GD (n = 20/48, 41.7%), (2) the lack of guidelines on how to approach this topic (n = 18/48, 37.5%), and (3) not knowing enough about the link (n = 17/48, 35.4%; N = 48; additional reasons listed in Table 3). When asked specifically whether these respondents would discuss the link if professional guidelines were released, almost all respondents (n = 47/48) reported they would discuss the risk of GBA-PD in the preconception/prenatal setting.
Table 3.
Reasons for not discussing GBA-PD risk
| N = 48 | ||
|---|---|---|
| n | % | |
| I have not counseled patients with personal or family history of GD | 20 | 41.7% |
| I have not counseled patients with personal or family history of PD | 10 | 20.8% |
| There are no guidelines from professional organizations about how to approach this link | 18 | 37.5% |
| I do not know enough about this link | 17 | 35.4% |
| It is too difficult to find current and consistent increased risk values | 8 | 16.7% |
| There is no cure | 4 | 8.3% |
| The lifetime risk is not high enough | 4 | 8.3% |
| I do not feel comfortable discussing late-onset disease in a preconception/prenatal setting | 10 | 20.8% |
| It was not the reason for the referral | 6 | 12.5% |
| Other | 9 | 18.8% |
Discussion
This is the first study to survey the knowledge, practices, and attitudes of PPGCs regarding the risk for PD in carriers of and patients with GD. It serves as a snapshot for the discussions of GBA-PD by genetic counselors during prenatal/preconception indications; during a period when the link was becoming more widely known among those in this cohort. This study found that although a majority of respondents were aware of the link, only a small proportion chose to discuss it. This inconsistency raises a concern that patients are not receiving the same care and education across the field.
Results suggest that experience and guidelines steer genetic counseling knowledge and practice. Of those who choose to discuss GBA-PD, all but one had experience with GD or PD. Limited knowledge and absence of guidelines on the topic were cited among the top reasons why this cohort chooses not to discuss the link. If guidelines were established, all but one of the respondents who indicated they did not discuss the link answered that they would discuss the GBA-PD risk. This is consistent with the attitudes and practices of genetics professionals before and after fragile X guidelines were established that specifically mentioned adult-onset FXTAS and FXPOI risks in premutation carriers [25]. In 2009, a survey of geneticists and genetic counselors found that only a quarter had this discussion, whereas one hundred percent of PPGCs reported discussing FXTAS and FXPOI in this study [25]. Overall, we speculate that education and guidelines on GBA-PD would lead to an uptake in discussions amongst genetic healthcare providers of this and other adult-onset risks to carriers.
The most striking observation is the incongruence between what providers feel patients want to know and what literature suggests patients desire to know with respect to PD risk. Respondents who discuss GBA-PD risk chose “respect[ing] a patient’s right-to-know” and finding “that patients want to know” among the three most common reasons why they discuss the link. Patient autonomy is a central tenet of the NSGC Code of Ethics [26], and is represented in the reasoning listed above. However, it is surprising that the PPGCs who inform patients only represent a third of those who are aware of GBA-PD. Indeed, research suggests that patients want to know this risk information. For example, individuals with PD want genetic testing to learn of the risk to their children [27], and individuals with GD want to learn of their risk for PD from their healthcare providers rather than through other sources [28]. In 2011, during a time when respondents with PD were not well aware that genetic testing was available, there was perceived interest and utility in learning of genetic cause [9]. It is also important to consider the timing of the discussion. The 2011 study found that GBA-PD is discussed only when disclosing positive results or if the patient/family bring up the topic. However, in a retrospective survey of individuals who underwent carrier screening for GD and tested negative, they specified they would have wanted to learn of the potential to identify PD risk before undergoing testing [29]. Other studies have also highlighted the patient desire to learn and know more about their potential carrier status and risks. A publication studying nulliparous females on their awareness of and interest in expanded carrier screening revealed that 78% were interested in screening and preferred to learn the information prior to pregnancy and from their healthcare provider [30]. A study among X-linked carriers revealed that most individuals learned about their carrier status when having a child with an X-linked condition but the majority believed that they should have learned this information ahead of time including their potential for personal risk as carriers of X-linked conditions [31]. These studies highlight that it is important for healthcare providers to consider what patients want and how to approach potential risks during the pre-test session.
This study also highlights a need for continued education among healthcare providers to better inform them of the potential to discover health risks to carriers during the carrier screening process. The Ashkenazi Jewish population is the group most likely to be carriers for GD [9]; however, this was one of the only scenarios in which some (10%) respondents indicated they felt very uncomfortable discussing GBA-PD. This, in-part, may be due to providers feeling less comfortable bringing up topics that their patients are more likely to be familiar with. Furthermore, one of the top three reasons counselors listed they do not discuss GBA-PD risk is because they do not have enough knowledge about it. Increased education on this topic could inspire a greater level of confidence when discussing risks with patients, possibly making providers more likely to bring up the topic. This study found that GeneReviews and primary literature were the main sources of information PPGCs used when preparing to discuss GBA-PD. However, with the hundreds of conditions available on carrier screening panels, it is not always feasible for providers to thoroughly research all potential disease risks to carriers prior to a pre-test discussion. Nor is it practical to discuss all potentially identified risks to carriers in addition to the already complex pre-test counseling. In addition, patients rarely request screening for just one condition like GD. As carrier screening panels continue to expand and new carrier risks identified, there will be increasing barriers to discussing every disease-specific risk with patients. As a suggested alternative, this education could instead be delivered to providers and patients in a more accessible format, such as video or webinar. General pre-carrier screen statements highlighting the possibility of identifying health risks related to carriers with a few examples may also be helpful.
It is important to note that the respondents of this study are not representative of the entire genetic counseling community [32]. There is an overrepresentation of individuals employed by commercial diagnostic laboratories and those with less than four years of experience. And it is possible that this overrepresentation may skew towards more participants being aware of the link as laboratories include this information in their reports. Additionally, more recent graduates may be more aware of these additional carrier risks as they are learning about carrier screening and recent developments through their coursework. Additionally, as the survey was communicated to the listserv as focusing on adult-onset disease risks in carriers for recessive conditions, there may have been self-selection bias of individuals completing the survey who had particular interest on this topic. And as with most retrospective surveying, there was opportunity for recall bias. However, this research adds to an important conversation on whether healthcare providers should be discussing risks associated with being a carrier for patients undergoing carrier screening. Further research interviewing various stakeholders, and quantitatively assessing psychosocial effects on patients before and after testing, is needed to design an evidence-based approach to genetic counseling for this topic.
Conclusion
As the availability and uptake of carrier screening increases along with the number of conditions on carrier screening panels, it is important to be prepared for incidental findings such as carrier risks. As we have already seen, there are several genes on expanded carrier screening panels, such as ATM, NBN, and FMR1, that confer an additional risk to the carrier themselves. As our knowledge and understanding of genetics expands, future research will uncover additional health risks linked to carrier statuses. It is important to understand how genetic counselors and other healthcare providers learn of these links, whether they choose to discuss, and how they discuss them with patients. Considerations must be made to include what information the patients want to know, and whether this is important to discuss during the pre-test session. With better understanding of all these factors, the process of disseminating knowledge and recommendations may be streamlined to provide up-to-date, comprehensive resources for healthcare providers, and high-quality care to patients.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The authors would like to thank Lisa Kinsley, neurology genetic counselor, for serving on the thesis committee. The authors would also like to thank Amy Yang, biostatistician, who provided guidance on the statistical analyses. Funding for the donation incentive was provided by Northwestern University Graduate Program in Genetic Counseling. Research presented in this paper was conducted to fulfill a requirement for a Master of Science degree in genetic counseling of first author Tara Jones.
Author contribution
All authors played a role in study design and data interpretation. TJ reviewed literature, analyzed data, designed figures, and drafted the manuscript. AA and JS edited the manuscript and approved the final version for publication. JR and SA provided manuscript feedback. JS served as subject expert. AA served as principal investigator. All authors read and approved the final manuscript.
Funding
The research was conducted as part of a Master’s thesis project for the Northwestern University Graduate Program in Genetic Counseling. Funding for the study was provided by Northwestern University.
Data availability
Access to data is restricted to certain individuals in accordance to IRB protocol.
Code availability
Not applicable.
Declarations
Conflict of interest
AA was an employee and shareholder of Myriad Genetics, a laboratory that offers carrier screening at the time of study. TJ holds shares in Invitae and Myriad, both genetic laboratories offering carrier screening. The other authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Committee Opinion No. 691 Carrier screening for genetic conditions. Obstet Gynecol. 2017;129(3):e41–e55. doi: 10.1097/AOG.0000000000001952. [DOI] [PubMed] [Google Scholar]
- 2.Finucane B, Abrams L, Cronister A, Archibald AD, Bennett RL, McConkie-Rosell A. Genetic counseling and testing for FMR1 gene mutations: practice guidelines of the national society of genetic counselors. J Genet Couns. 2012;21(6):752–760. doi: 10.1007/s10897-012-9524-8. [DOI] [PubMed] [Google Scholar]
- 3.Hagerman RJ, Protic D, Rajaratnam A, Salcedo-Arellano MJ, Aydin EY, Schneider A. Fragile X-associated neuropsychiatric disorders (FXAND) Front Psychiatry. 2018;13(9):564. doi: 10.3389/fpsyt.2018.00564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Berwick M, Satagopan JM, Ben-Porat L, Carlson A, Mah K, Henry R, Diotti R, Milton K, Pujara K, Landers T, Dev Batish S, Morales J, Schindler D, Hanenberg H, Hromas R, Levran O, Auerbach AD. Genetic heterogeneity among Fanconi anemia heterozygotes and risk of cancer. Cancer Res. 2007;67(19):9591–9596. doi: 10.1158/0008-5472.CAN-07-1501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bogdanova N, Feshchenko S, Schürmann P, Waltes R, Wieland B, Hillemanns P, Rogov YI, Dammann O, Bremer M, Karstens JH, Sohn C, Varon R, Dörk T. Nijmegen breakage syndrome mutations and risk of breast cancer. Int J Cancer. 2008;122(4):802–806. doi: 10.1002/ijc.23168. [DOI] [PubMed] [Google Scholar]
- 6.de Voer RM, Hahn MM, Mensenkamp AR, Hoischen A, Gilissen C, Henkes A, Spruijt L, van Zelst-Stams WA, Kets CM, Verwiel ET, Nagtegaal ID, Schackert HK, van Kessel AG, Hoogerbrugge N, Ligtenberg MJ, Kuiper RP. Deleterious germline BLM mutations and the risk for early-onset colorectal cancer. Sci Rep. 2015;11(5):14060. doi: 10.1038/srep14060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Geoffroy-Perez B, Janin N, Ossian K, Laugé A, Croquette MF, Griscelli C, Debré M, Bressac-de-Paillerets B, Aurias A, Stoppa-Lyonnet D, Andrieu N. Cancer risk in heterozygotes for ataxia-telangiectasia. Int J Cancer. 2001;93(2):288–293. doi: 10.1002/ijc.1329. [DOI] [PubMed] [Google Scholar]
- 8.Kannai R, Chertok IR. Prenatal panel screening considerations for non-neuronopathic Gaucher disease in the Ashkenazi-Jewish population. Isr Med Assoc J. 2006;8(5):347–350. [PubMed] [Google Scholar]
- 9.Falcone D, Wood EM, Mennuti M, Xie SX, Van Deerlin VM. Prenatal healthcare providers’ Gaucher disease carrier screening practices. Genet Med. 2012;14(10):844–851. doi: 10.1038/gim.2012.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Goker-Alpan O, Schiffmann R, LaMarca ME, Nussbaum RL, McInerney-Leo A, Sidransky E. Parkinsonism among Gaucher disease carriers. J Med Genet. 2004;41(12):937–940. doi: 10.1136/jmg.2004.024455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rana HQ, Balwani M, Bier L, Alcalay RN. Age-specific Parkinson disease risk in GBA mutation carriers: information for genetic counseling. Genet Med. 2013;15(2):146–149. doi: 10.1038/gim.2012.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rosenbloom B, Balwani M, Bronstein JM, Kolodny E, Sathe S, Gwosdow AR, Taylor JS, Cole JA, Zimran A, Weinreb NJ. The incidence of Parkinsonism in patients with type 1 Gaucher disease: data from the ICGG Gaucher Registry. Blood Cells Mol Dis. 2011;46(1):95–102. doi: 10.1016/j.bcmd.2010.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gan-Or Z, Amshalom I, Kilarski LL, Bar-Shira A, Gana-Weisz M, Mirelman A, Marder K, Bressman S, Giladi N, Orr-Urtreger A. Differential effects of severe vs mild GBA mutations on Parkinson disease. Neurology. 2015;84(9):880–887. doi: 10.1212/WNL.0000000000001315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.de Lau LM, Breteler MM. Epidemiology of Parkinson’s disease. Lancet Neurol. 2006;5(6):525–535. doi: 10.1016/S1474-4422(06)70471-9. [DOI] [PubMed] [Google Scholar]
- 15.Tysnes OB, Storstein A. Epidemiology of Parkinson’s disease. J Neural Transm (Vienna) 2017;124(8):901–905. doi: 10.1007/s00702-017-1686-y. [DOI] [PubMed] [Google Scholar]
- 16.Lopez G, Sidransky E. Autosomal recessive mutations in the development of Parkinson’s disease. Biomark Med. 2010;4(5):713–721. doi: 10.2217/bmm.10.96. [DOI] [PubMed] [Google Scholar]
- 17.Lazarin GA, Haque IS. Expanded carrier screening: a review of early implementation and literature. Semin Perinatol. 2016;40(1):29–34. doi: 10.1053/j.semperi.2015.11.005. [DOI] [PubMed] [Google Scholar]
- 18.Grody WW, Thompson BH, Gregg AR, Bean LH, Monaghan KG, Schneider A, Lebo RV. ACMG position statement on prenatal/preconception expanded carrier screening. Genet Med. 2013;15(6):482–483. doi: 10.1038/gim.2013.47. [DOI] [PubMed] [Google Scholar]
- 19.Gregg AR, Aarabi M, Klugman S, Leach NT, Bashford MT, Goldwaser T, Chen E, Sparks TN, Reddi HV, Rajkovic A, Dungan JS, ACMG Professional Practice and Guidelines Committee Screening for autosomal recessive and X-linked conditions during pregnancy and preconception: a practice resource of the American College of Medical Genetics and Genomics (ACMG) Genet Med. 2021 doi: 10.1038/s41436-021-01203-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Reeves A, Trepanier A. Comparison of informed consent preferences for multiplex genetic carrier screening among a diverse population. J Genet Couns. 2016;25(1):166–178. doi: 10.1007/s10897-015-9854-4. [DOI] [PubMed] [Google Scholar]
- 21.Ormond KE, Iris M, Banuvar S, Minogue J, Annas GJ, Elias S. What do patients prefer: informed consent models for genetic carrier testing. J Genet Couns. 2007;16(4):539–550. doi: 10.1007/s10897-007-9094-3. [DOI] [PubMed] [Google Scholar]
- 22.Ormond KE, Banuvar S, Daly A, Iris M, Minogue J, Elias S. Information preferences of high literacy pregnant women regarding informed consent models for genetic carrier screening. Patient Educ Couns. 2009;75(2):244–250. doi: 10.1016/j.pec.2008.09.020. [DOI] [PubMed] [Google Scholar]
- 23.Allen PM, Edwards JA, Snyder FJ, Makinson KA, Hamby DM. The effect of cognitive load on decision making with graphically displayed uncertainty information. Risk Anal. 2014;34(8):1495–1505. doi: 10.1111/risa.12161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Harris PA, Taylor R, Thielke R, Payne J, Gonzales N, et al. Research electronic data capture (REDCap) – a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–81. doi: 10.1016/j.jbi.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lieberman S, Zuckerman S, Levy-Lahad E, Altarescu G. Conflicts regarding genetic counseling for fragile X syndrome screening: a survey of clinical geneticists and genetic counselors in Israel. Am J Med Genet A. 2011;155A(9):2154–2160. doi: 10.1002/ajmg.a.34155. [DOI] [PubMed] [Google Scholar]
- 26.National Society of Genetic Counselors (NSGC), Inc. (2017). NSGC Code of Ethics. Accessed August 29, 2020, from https://www.nsgc.org/p/cm/ld/fid=12
- 27.Scuffham TM, McInerny-Leo A, Ng SK, Mellick G. Knowledge and attitudes towards genetic testing in those affected with Parkinson’s disease. J Community Genet. 2014;5:167–177. doi: 10.1007/s12687-0168-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zaretsky L, Zeid N, Naik H, Alcalay RN, Balwani M. Knowledge and attitudes of Parkinson’s disease risk in the Gaucher population. J Genet Couns. 2019;29:105–111. doi: 10.1002/jgc4.1185. [DOI] [PubMed] [Google Scholar]
- 29.Mulhern M, Bier L, Alcalay RN, Balwani M. Patients’ opinions on genetic counsel ing on the increased risk of Parkinson disease among Gaucher disease carriers. J Genet Counsel. 2017 doi: 10.1007/s10897-017-0161-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rabkina L, Swanson A, Aufox S, Propst L, Fiddler M, Wagner A, Arjunan A. What women want: general population perspectives and access to preconception expanded carrier screening. Prenat Diagn. 2021;41(9):1057–1065. doi: 10.1002/pd.5995. [DOI] [PubMed] [Google Scholar]
- 31.Choi J, Kane T, Propst L, Spencer S, Kostialik J, Arjunan A. Not just carriers: experiences of X-linked female heterozygotes. J Assist Reprod Genet. 2021;38(10):2757–2767. doi: 10.1007/s10815-021-02270-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.National Society of Genetic Counselors. 2021 Professional status survey: executive summary. 2021. Accessed January 8, 2022.https://www.nsgc.org/Portals/0/Executive%20Summary%202021%20FINAL%2005-03-21.pdf
- 33.Bogue Lauren, Peay Holly, Martin Ann, Lucas Ann, Ramchandren Sindhu. Knowledge of carrier status and barriers to testing among mothers of sons with Duchenne or Becker muscular dystrophy. Neuromuscular Disorders. 2016;26(12):860–864. doi: 10.1016/j.nmd.2016.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Marc, Engelen Mathieu, Barbier Inge M. E., Dijkstra Remmelt, Schür Rob M. A., de Bie Camiel, Verhamme Marcel G. W., Dijkgraaf Patrick A., Aubourg Ronald J. A., Wanders Bjorn M., van Geel Marianne, de Visser Bwee T., Poll–The Stephan, Kemp (2014) (2014) (2014) X-linked adrenoleukodystrophy in women: a cross-sectional cohort study. Brain 137(3) 693-706 10.1093/brain/awt361 [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Access to data is restricted to certain individuals in accordance to IRB protocol.
Not applicable.