Abstract
Background:
There are no effective therapies for right ventricular systolic dysfunction (RVD), hence the need to identify and modify risk factors for progressive RVD. The purpose of this study was to determine the risk factors for, and prognostic implications of progressive RVD in adults with congenitally corrected transposition of great arteries (cc-TGA).
Methods:
RV systolic function was assessed using RV longitudinal strain (RV-LS). The first echocardiogram (baseline echocardiogram) and all subsequent annual echocardiograms performed within 5 years from the baseline echocardiogram were analyzed. Progressive RVD (temporal decline in RV-LS) was assessed as the average annual change in RV-LS within the 5 years of imaging follow-up.
Results:
Of 186 patients (40±12 years), the RV-LS at baseline was-17±4%, and the annual decline in RV-LS was −4% (−6 – −2). The risk factors for progressive RVD were LV systolic dysfunction, LV pacing and systemic hypertension. Cardiovascular events (heart failure hospitalization, heart transplant, death) occurred in 57 (27%) patients. Progressive RVD was associated with cardiovascular events, independent of RV systolic function at baseline. In subgroup analyses assessing impact of therapies (medical therapy, cardiac resynchronization therapy, and tricuspid valve replacement), only tricuspid valve replacement was associated with improvement of RV systolic function when performed prior to onset of RVD.
Conclusions:
Patients with cc-TGA were at risk for progressive RVD, and the risk factors for progressive RVD were LV pacing, systemic hypertension and concomitant LV dysfunction. Further studies are required to determine whether strict blood pressure control and early tricuspid valve replacement will prevent progressive RVD.
Keywords: Transposition of great arteries, Systemic ventricular dysfunction, Cardiovascular outcomes
INTRODUCTION
Congenitally corrected transposition of great arteries (cc-TGA) is characterized by atrioventricular discordance and ventriculoarterial discordance, and as a result the right ventricle (RV) becomes the systemic ventricle.1, 2 The RV is not optimally adapted for systemic circulation because of its thin wall and a single coronary artery blood supply.1, 2 As a result, the systemic RV tends to fail overtime leading to a high prevalence of systemic RV systolic dysfunction (RVD) in adults with cc-TGA.3–6 Additionally, RV volume overload from tricuspid regurgitation, and ventricular dyssynchrony from left ventricular (LV) pacing further exacerbate systemic RVD in this population.7–9
The assessment of systemic RV systolic function by echocardiography is challenging because the complex shape of the RV (unlike the LV) does not allow for the geometric assumptions required for the calculation of ejection fraction.10, 11 However, speckle tracking imaging has emerged as a robust imaging tool for the assessment of RV systolic function across different disease groups, including cc-TGA.4 RV longitudinal strain can identify patients with cc-TGA who have RVD, and hence can be used for risk stratification and prognostication.4 Several therapies have been proposed for the treatment of RVD, but the efficacy of these therapies have not been consistent across different studies.6, 7, 12–15 Since there are no validated therapies to reverse RVD once it occurs, the goal of care should, therefore, be to prevent the occurrence and progression of RVD. As the next step towards this goal, there is a need to identify and treat reversible causes of progressive RVD, but such data are currently lacking. The purpose of this study was, therefore, to determine the risk factors for, and prognostic implications of progressive RVD (temporal decline in RV systolic function) in adults with cc-TGA.
METHODS
Study Population
This is a retrospective study of adults (≥18 years of age) with cc-TGA that underwent transthoracic echocardiogram at the Mayo Clinic Rochester from January 1, 2003 to December 31, 2020. Patients with Fontan palliation were excluded. From this cohort, we selected consecutive patients that had ≥2 echocardiograms from which RV longitudinal strain (RV-LS) can be calculated by offline image analyses. The patients were classified as simple cc-TGA or complex cc-TGA depending on whether they had associated congenital heart lesions such as ventricular septal defect, and/or LV outflow tract obstruction. The Institutional Review Board approved this study.
We reviewed clinic notes, surgical notes, and echocardiograms of all patients. Cardiovascular event was defined as the composite endpoint of heart failure hospitalization, heart transplant and cardiovascular death during follow-up. Cardiovascular events were ascertained from a review of the medical records and Accurint mortality database.
Echocardiography
Comprehensive 2-dimensional, Doppler and speckle tracking transthoracic echocardiogram was performed according to contemporary guidelines, with offline image analyses and measurements performed by 2 experienced research sonographers (J.W and K.T).10, 11, 16 The first echocardiogram performed within the study period was considered as the baseline study. Using the baseline echocardiogram as ‘time 0’, we reviewed all subsequent echocardiograms performed during annual clinical evaluations (±3 months) within 5 years from the baseline echocardiogram.
RV-LS was used as the primary metric of RV systolic function for this study. RV-LS was assessed by speckle tracking strain imaging with Vivid E9 and E95 (General Electric Co, Fairfield, Connecticut) with M5S and M5Sc-D transducers (1.5–4.6 MHz) at frame rate of 40 to 80 Hz. Three-beat cine-loop clips were obtained from RV-focused apical 4-chamber views, and the ventricular septum was included for the assessment of RV-LS. We assessed baseline RV systolic function using the RV-LS derived from the baseline echocardiogram, and we assessed for progressive RVD (temporal decline in RV systolic function) as the average annual change in RV systolic function (RV-LS) within the 5 years of imaging follow-up. The annual change in RV systolic function was calculated at each annual evaluation for each patient using this formula: ([RV-LS from previous echocardiogram − RV-LS from most recent echocardiogram] ÷ RV-LS from previous echocardiogram) ×100. RV fractional area change (RV FAC) was used as the secondary metric of RV systolic function, and the annual change in FAC was calculated using the same method described above. Other RV systic function indices such as tricuspid annular plane systolic excursion (TAPSE) and RV tissue Doppler systolic velocity (s’) were also assessed. Right atrial (RA) pressure was estimated based on respirophasic changes in the diameter of the inferior vena cava (IVC).11 IVC diameter in end-expiration (IVCexpiration) and inspiration (IVCinspiration) were assessed as an average of 3 measurements from 3 consecutive respiration cycles. Normal IVC size (IVC diameter ≤21mm) with normal collapsibility (≥50% collapsibility) during inspiration was assigned an estimated RA pressure of 3 mmHg. Abnormal IVC size or abnormal IVC collapsibility was assigned an estimated RA pressure of 8 mmHg, while abnormal IVC size and abnormal IVC collapsibility was assigned an estimated RA pressure of 15 mmHg
Subgroup Analysis
Four sets of subgroup analyses were performed to assess the effect of cardiac interventions on RV systolic function. Subgroup analysis #1 involved patients that underwent tricuspid valve replacement. Subgroup analysis #2 involved patients that underwent cardiac resynchronization therapy (CRT), and this includes patient without prior pacemaker implantation that underwent an initial implantation of a biventricular pacemaker, as well as patients that had an upgrade of a dual chamber pacemaker to a biventricular pacemaker. Subgroup analysis #3 involved patients that were not on heart failure therapy (defined as beta blocker and/or renin angiotensin aldosterone system [RAAS] antagonist) but were placed on these therapies after the baseline echocardiogram. Subgroup analysis #4 involved patients that were on heart failure therapy but had an increase in the intensity of therapy after the baseline echocardiogram (defined as increase in dose and/or addition of a new agent). For these subgroup analyses, we compared pre-intervention RV indices (derived from the last echocardiogram prior to therapeutic intervention) to post-intervention RV indices (derived from the first echocardiogram performed 24 months after therapeutic intervention).
For the purpose of this study, we classified all cardiac implantable electronic devises (CIED) into 3 mutually exclusive groups: (1) dual chamber pacemaker defined as having right atrial (RA) and LV leads only; (2) CRT defined as having a both RV and LV leads; (3) defibrillator defined as having a defibrillator with or without pacing capabilities.
Statistical Analysis
Data were presented count (%), mean ± standard deviation, median (interquartile range) or estimates (95% confidence interval [CI]). Between-group comparisons were performed using paired and unpaired t-test, Wilcoxon rank sum test, and Fischer’s exact test as appropriate. Multivariate regression models were created using stepwise backwards selection, with a p<0.25 required for entry, and p<0.1 required for a variable to remain in the model, and these variables were derived from univariate analysis. In order to control for the effect of therapies on progressive RVD and cardiovascular event, we adjusted for these therapies (tricuspid valve replacement, CRT, and heart failure therapy) in the models.
The prognostic power of RV-LS over conventional echocardiographic parameters (RV FAC, TAPSE, and RV end-diastolic area) was assessed using Cox regression. First, we created a multivariable Cox model (base model) using the following indices (RV FAC, TAPSE, and RV end-diastolic area, and age), and then we created a second model addition RV-LS to the indices in the first model. C-statistics and the integrated discrimination index (IDI)17 were calculated to quantify discriminatory ability and to evaluate the improvement in prediction accuracy of the combination of RV-LS and the conventional echocardiographic indices over the conventional indices alone. IDI (95% CI) and comparison p values were derived based from 1,000 bootstrap samples. Intraobserver, interobserver, and test-retest agreement were assessed in 20 randomly selected patients using intraclass correlation coefficient (ICC). All statistical analyses were performed with JMP software (version 14.1.0; SAS Institute Inc, Cary NC) and GraphPad software (version 9.0.1; San Diego, CA).
RESULTS
RV Systolic Function
Table 1 shows the baseline characteristics of the 186 patients (age 40±12 years; male 93 [50%]) that met the study inclusion criteria. All patients had adequate images for the assessment of RV-LS (inclusion criteria) while 184 (99%) patients had adequate images for the assessment of FAC. There was good intraobserver, interobserver, and test-retest agreement for RV-LS (0.91 [0.87–0.94], 0.87 [0.82–0.92], and 0.88 [0.84–0.92] respectively), and for FAC (0.90 [0.87–0.93], 0.83 [0.79–0.87], and 0.85 [0.81–0.89] respectively). The mean RV-LS and RV FAC were- 17±4% and 31±9% respectively. There was a modest correlation between RV-LS and RV FAC (r=−0.68, p=0.004). Cardiac magnetic resonance imaging data was available in 48 patients (Table 1). There was also a modest correlation between RV-LS and RV ejection fraction by magnetic resonance imaging (r=−0.61, p=0.01).
Table 1:
Baseline Characteristics
| Variables | All (n=186) |
|---|---|
| Demographic indices | |
| Age, years | 40±12 |
| Male sex | 93 (50%) |
| Body mass index, kg/m2 | 27±6 |
| Complex CC-TGA | 96 (52%) |
| Systolic blood pressure, mmHg | 129±14 |
| Prior cardiac procedures | 101 (54%) |
| Tricuspid valve repair | 12 (7%) |
| Tricuspid valve replacement | 31 (17%) |
| Ventricular septal defect closure | 47 (25%) |
| Infundibular stenosis resection | 2 (1%) |
| PVR/RV-PA conduit placement | 16 (9%) |
| Palliative shunts | 6 (3%) |
| CIED | 76 (41%) |
| Dual chamber pacemaker | 52 (30%) |
| Biventricular pacemaker | 7 (4%) |
| Defibrillators | 17 (9%) |
| Comorbidities | |
| Hypertension | 38 (20%) |
| Diabetes | 4 (2%) |
| Coronary artery disease | 6 (3%) |
| Medications | |
| Beta blockers | 62 (33%) |
| ACEI/ARB | 101 (54%) |
| Spironolactone | 21 (11%) |
| Loop diuretics | 61 (33%) |
| Digoxin | 49 (26%) |
| Echocardiography | |
| Systemic indices | |
| LA volume index, ml/m2 | 46 (32–69) |
| LA reservoir strain, % | 23±8 |
| RV longitudinal strain, % | −17±4 |
| RV fractional area change, % | 31±9 |
| RV end-diastolic area, cm2 | 37±10 |
| RV end-systolic area, cm2 | 27±10 |
| RV cardiac index, ml/m2 | 2.6±0.4 |
| ≥Mod tricuspid regurgitation* | 74 (40%) |
| ≥Mod aortic regurgitation* | 12 (7%) |
| TAPSE, cm [n=179] | 13±4 |
| RV s’, cm/s [n=65] | 8±13 |
| Non-systemic indices | |
| RA volume index, ml/m2 | 23±10 |
| RA reservoir strain, % | 32±11 |
| RA pressure, mmHg | 9±4 |
| LV longitudinal strain, % | −23±6 |
| LV systolic pressure, mmHg | 39 (32–54) |
| Cardiac MRI [n=48] | |
| RV end-diastolic volume index, ml/m2 | 114±39 |
| RV end-systolic volume index, ml/m2 | 62±28 |
| RV ejection fraction, % | 47±13 |
| LV end-diastolic volume index, ml/m2 | 93±35 |
| LV end-systolic volume index, ml/m2 | 45±24 |
| LV ejection fraction, % | 53±13 |
CC-TGA: congenitally corrected transposition of great arteries; PVR: pulmonary valve replacement; RV-PA: right ventricular to pulmonary artery; ACEI: angiotensin converting enzyme inhibitor; ARB: angiotensin-II receptor blocker; RA: right atrium; RV right ventricle; LV: left ventricle; LA: left atrium; CIED: cardiac implantable electronic device; MRI: magnetic resonance imaging; TAPSE: Tricuspid annular plane systolic excursion; s’: tissue Doppler systolic velocity
Table 2 shows the RV indices derived from the annual echocardiograms over a 5-year period, and the mean interval between studies was 13±2 months. The annual decline in RV-LS for the overall group was −4% (95%CI −6 – −2). Concordant with the temporal decrease in RV-LS, there was a temporal increase in N-terminal prohormone brain natriuretic peptide (analysis of variance p value 0.03), Table 2. The risk factors associated with progressive RVD (temporal decline in RV-LS) were LV systolic dysfunction (LV-LS), LV pacing, RA hypertension (RA pressure) and systemic hypertension (Table 3, and central illustration). Tricuspid regurgitation severity and history of complex cc-TGA were not associated with progressive RVD.
Table 2:
Serial Echocardiograms
| Variables | Baseline (n=186) | Year 1 (n=186) | Year 2 (n=159) | Year 3 (n=125) | Year 4 (n=108) | Year 5 (n=91) |
|---|---|---|---|---|---|---|
| RV systolic function | ||||||
| RV global longitudinal strain, % | −17±4 | −18±4 | −15±3 | −16±4 | −15±5 | −14±4 |
| RV fractional area change, % | 31±9 | 30±8 | 29±10 | 27±7 | 29±12 | 28±10 |
| Other systemic indices | ||||||
| RV end-diastolic area, cm2 | 37±10 | 35±9 | 39±11 | 36±10 | 39±8 | 41±9 |
| RV end-systolic area, cm2 | 25±8 | 25±8 | 27±8 | 26±10 | 28±7 | 30±8 |
| RV cardiac index, ml/m2 | 2.6±0.4 | 2.8±0.5 | 2.7±0.6 | 2.6±0.5 | 24.±0.7 | 2.5±0.4 |
| LA volume index, ml/m2 | 46 (32–69) | 48 (34–66) | 43 (28–69) | 44 (31–64) | 47 (34–72) | 49 (33–72) |
| LA reservoir strain, % | 20 (14–30) | 20 (15–31) | 18 (12–37) | 17 (12–35) | 18 (14–27) | 15 (11–19) |
| Neurohormonal activation | ||||||
| NT-proBNP, pg/ml | 265±143 [n=133] | 226±112 [n=103] | 273±153 [n=121] | 329±186 [n=84] | 305±216 [n=65] | 398±204 [n=69] |
RA: right atrium; RV right ventricle; LV: left ventricle; LA: left atrium; NT-proBNP: N-terminal pro hormone brain natriuretic peptide; [n] signifies number of patients with laboratory assay.
Table 3:
Risk Factors Associated with Temporal Decline in RV-LS (Progressive RVD)
| Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|
| Variables | β±SE | p | β±SE | p |
| Age, per year | 0.12±0.07 | 0.004 | ||
| Male sex | 0.19±0.11 | 0.02 | ||
| Body mass index, per km/m2 | --- | --- | ||
| Complex CC-TGA | --- | --- | ||
| LV pacing | 0.33±0.18 | 0.001 | 0.29±0.16 | 0.007 |
| CRT | --- | --- | ||
| Prior tricuspid valve surgery | --- | --- | ||
| Atrial fibrillation | --- | --- | ||
| Hypertension | 0.39±0.21 | <0.001 | 0.37±0.19 | <0.001 |
| ≥Moderate TR at baseline | 0.09±0.06 | 0.01 | ||
| Progression of TR | 0.07±0.07 | 0.08 | ||
| ≥Moderate aortic regurgitation | 0.11±0.09 | 0.03 | ||
| * LV-LS, per % | −0.14±0.06 | 0.004 | −0.11±0.05 | 0.01 |
| Annual change in LV-LS, per % | 0.11±0.09 | 0.03 | ||
| ≥Moderate mitral regurgitation | 0.07±0.05 | 0.004 | ||
| ≥Moderate pulmonary regurgitation | --- | --- | ||
| Pulmonary valve mean gradient, per % | --- | --- | ||
| RA pressure, mmHg | 0.18±0.12 | 0.01 | 0.10±0.06 | 0.004 |
| RA reservoir strain, % | −0.08±0.24 | 0.2 | ||
| LV systolic pressure, mmHg | --- | --- | ||
| Heart failure therapy | 0.24±0.17 | 0.009 | ||
RVD: right ventricular systolic dysfunction; RV-LS: right ventricular longitudinal strain; CC-TGA: congenitally corrected transposition of great arteries; β: beta coefficient; SE: standard error; CRT: cardiac resynchronization therapy; LV: left ventricle; RA: right atrium; TR: tricuspid regurgitation
--- denoted variables with p≥0.25; Note that only variables with p<0.25 on univariable analysis were imputed into the multivariable model
note that LS was modeled as absolute values (i.e. ignoring the negative sign); progression of tricuspid regurgitation was any increase in tricuspid regurgitation severity from moderate to moderate/severe or from moderate/severe to severe based on quantitative Doppler assessment
Central illustration: Temporal Changes in RV Systolic Function Indices.
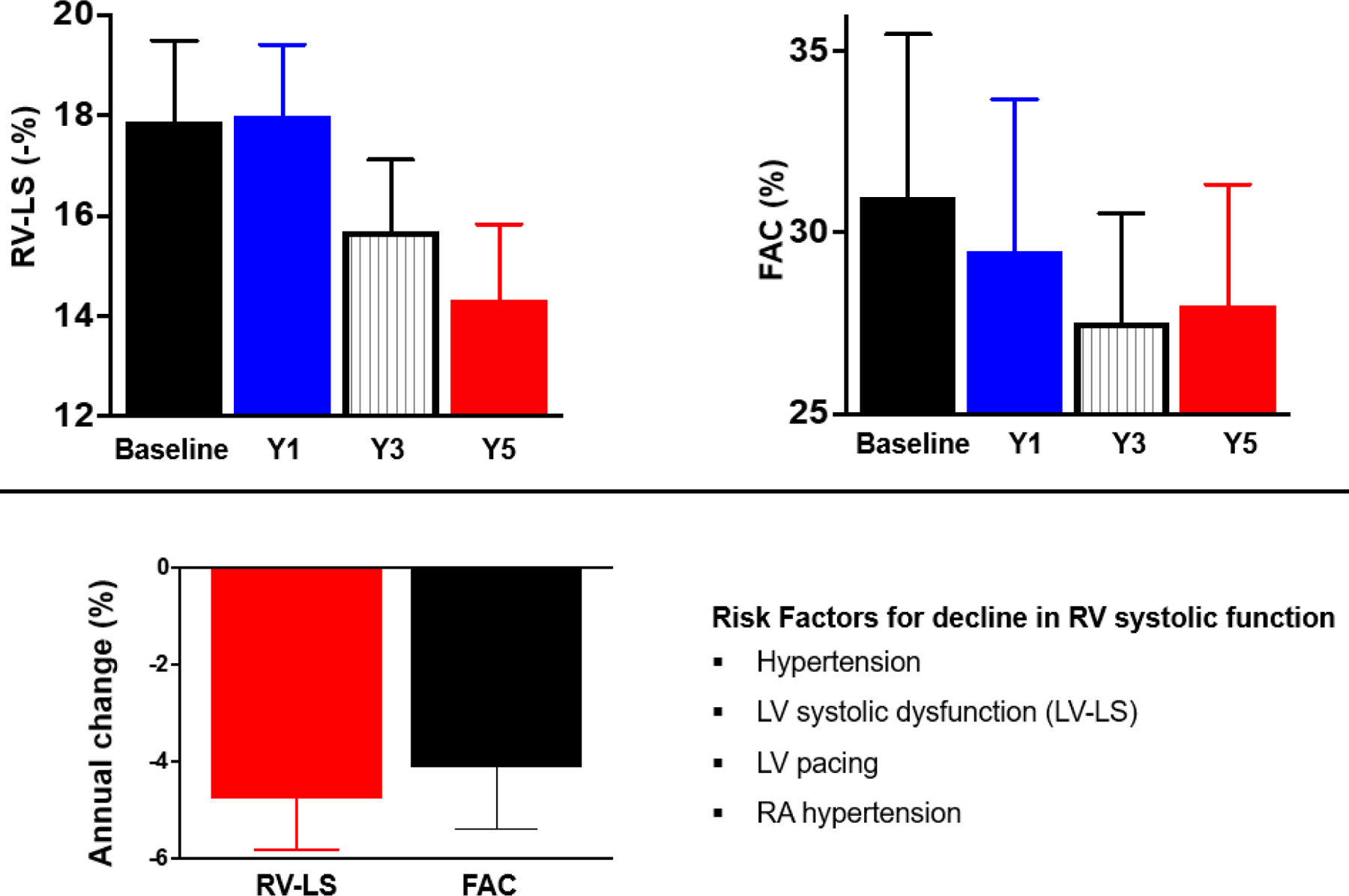
Top: bar graphs showing right ventricular longitudinal strain (RV-LS) and fractional area change (FAC) at baseline, and at 1, 3, and 5-year follow-up. Note that RV-LS is displayed as absolute numbers for ease (i.e. ignoring the negative sign)
Bottom: bar graphs showing the average annual change in RV-LS and FAC. Overall the RV-LS decrease by −4% per year while FAC decrease by −3% per year. The risk factors associated with temporal reduction in RV systolic function were hypertension, left ventricular longitudinal strain (LV-LS), LV pacing and right atrial (RA) hypertension
In an exploratory analysis using FAC as the metric for RV systolic function, we observed an annual decline in FAC was −3% (95%CI −6 – 0). Similarly, the risk factors associated with progressive RVD (temporal decline in FAC) were hypertension (beta coefficient t± standard error [β±SE] 0.32±0.16), LV-LS (β±SE −0.13±0.07 and LV pacing (β±SE 0.37±0.21), central illustration.
In a sensitivity analysis restricted to the patients that did not undergo tricuspid valve replacement between the baseline and final echocardiogram (n=133), the annual decline in RV-LS was −4% (95%CI −6 – −2). The rate of decline in RV systolic function derived from the sensitivity analysis was similar to that of the overall cohort (p=0.3).
Simple vs Complex cc-TGA
Of the 186 patients, 90 (48%) and 96 (52%) had simple cc-TGA and complex cc-TGA respectively. The simple cc-TGA group had better RV systolic function at baseline as compared to the complex cc-TGA group (RV-LS −18±4% vs −16±3%, p=0.04). The annual decline in RV-LS was −3% (95%CI −6 – 0) in the simple cc-TGA group, while the annual decline in RV-LS was −4% (95%CI −6 – −2) in the complex cc-TGA group. There was no significant difference in the rate of annual decline in RV-LS between the simple and complex cc-TGA groups (p=0.6).
Cardiovascular Events
The median duration of follow-up was 10.4 (4.2–16.1) years, and during this period, 34 (18%) patients were hospitalized for heart failure, 3 (2%) patients underwent heart transplant for end-stage heart failure, and 25 (13%) patients died from cardiovascular cause (end-stage heart failure n=20, sudden cardiac death n=4, stroke-related death n=1). The composite cardiovascular event endpoint occurred in 51 (27%) patients. Progressive RVD was a risk factor for cardiovascular events, independent of RV systolic function at baseline echocardiogram (Table 4).
Table 4:
Risk Factors Associated with Associated with Cardiovascular Events
| Univanate analysis | Multivanate analysis | |||
|---|---|---|---|---|
| Variables | HR (95%CI) | p | HR (95%CI) | p |
| * RV-LS at baseline, per % | 0.92 (0.88–0.96) | <0.001 | 0.94 (0.90–0.98) | 0.01 |
| Annual change in RV-LS, per % | 1.16 (1.08–1.31) | 0.005 | 1.11 (1.07–1.22) | <0.001 |
| Age, per year | 1.05 (1.02–1.08) | 0.004 | 1.03 (1.01–1.05) | 0.003 |
| Male sex | 1.11 (1.03–1.37) | 0.01 | ||
| Complex CC-TGA | --- | --- | ||
| LV pacing | 1.08 (1.01–1.15) | 0.03 | ||
| CRT | --- | --- | ||
| Tricuspid valve surgery | --- | --- | ||
| Atrial fibrillation | --- | --- | ||
| Hypertension | 1.36 (1.12–2.81) | 0.004 | 1.49 (1.16–2.68) | 0.04 |
| ≥Moderate tricuspid regurgitation | --- | --- | ||
| Heart failure therapy | --- | --- | ||
CC-TGA: congenitally corrected transposition of great arteries; HR: hazard ratio CI: confidence interval; CRT: cardiac resynchronization therapy; LV: left ventricle; RV: right ventricle; LS: longitudinal strain
--- denoted variables with p≥0.25; Note that only variables with p<0.25 on univariable analysis were imputed into the multivariable model
note that LS was modeled as absolute values (i.e. ignoring the negative sign)
RV Systolic Function Indices and Prognostication
In order to compare the prognostic power of RV-LS over conventional echocardiographic indices of RV hemodynamics (RV FAC, TAPSE, RV end-diastolic area), we created a base model using these conventional indices adjusted for age, and a second model by adding RV-LS to the base model (Table 5). The addition of RV-LS to the base model resulted in improvement in prognostic power as evidence by an increase in c-statistic from 0.676 (0.645–0.708) to 0.742 (0.722–0.789). Table 6 shows the reclassification analyses for prognostic value of RV-LS over conventional echocardiographic indices of RV hemodynamics.
Table 5:
Multivariable Cox Model Showing the Relationship Between RV Indices and Cardiovascular Events
| Base model | HR (95%CI) | p |
|---|---|---|
| RV FAC, per % | 0.97 (0.94–0.99) | 0.03 |
| TAPSE, per cm | 1.01 (0.93–1.10) | 0.7 |
| RV end-diastolic area, per cm2 | 1.03 (1.00–1.05) | 0.04 |
| Age, per year | 1.03 (1.01–1.05) | 0.02 |
| Base mode + RV-LS | HR (95%CI) | p |
| RV-LS, per % | 0.92 (0.88–0.96) | 0.002 |
| RV FAC, per % | 0.98 (0.95–1.02) | 0.3 |
| TAPSE, per cm | 1.05 (0.96–1.14) | 0.4 |
| RV end-diastolic area, per cm2 | 1.02 (0.99–1.04) | 0.2 |
| Age, per year | 1.03 (1.01–1.04) | 0.004 |
RV FAC: right ventricular fractional area change; TAPSE: tricuspid annular systolic plane excursion; HR: hazard ratio; CI: confidence interval; RV-LS: right ventricular longitudinal strain
note that LS was modeled as absolute values (i.e. ignoring the negative sign)
Table 6:
Reclassification Analyses for Prognostic Value of RV-LS for Clinical Outcomes
| Cardiovascular events | c-statistic (95%CI) | IDI (95%CI) | Relative IDI (95%CI) | p |
|---|---|---|---|---|
| Base model | 0.676 (0.645–0.708) | |||
| Base mode + RV-LS | 0.742 (0.711–0.789) | 0.028 (0.001–0.92) | 38.9% (0.3–144.8%)* | 0.02* |
The 95% CI and p values were calculated from 1,000 bootstrap samples. IDI: integrated discrimination index; CI: confidence interval;
indicate IDI analysis compared with base model
Subgroup Analysis
Of the 186 patients, 53 underwent tricuspid valve replacement during follow-up. There was an improvement in RV-LS, RV cardiac index, and left atrial strain with a reduction in RV end-diastolic area; but no significant improvement in FAC at 1-year post-operative echocardiogram, (Table 7). The average postoperative change in RV-LS was +2% (95%CI 1 – 3), and there was a correlation between preoperative RV-LS and postoperative increase in RV-LS (β±SE 0.19±0.08).
Table 7:
Subgroup Analyses
| Patients that underwent Tricuspid Valve Surgery (n=53) | |||
|---|---|---|---|
| Variables | Pre- | Post- | p |
| RV global longitudinal strain, % | −17±3 | −19±4 | 0.04 |
| RV fractional area change, % | 31±6 | 29±10 | 0.3 |
| RV end-diastolic area, cm2 | 39±8 | 33±9 | 0.02 |
| RV end-systolic area, cm2 | 28±6 | 26±8 | 0.4 |
| RV cardiac index, ml/m2 | 2.4±0.4 | 2.9±0.5 | 0.007 |
| Patients that underwent Cardiac Resynchronization Therapy (n=13) | |||
| Variables | Pre- | Post- | p |
| RV global longitudinal strain, % | −16±4 | −18±6 | 0.1 |
| RV fractional area change, % | 27±7 | 30±11 | 0.2 |
| RV end-diastolic area, cm2 | 37±6 | 34±9 | 0.3 |
| RV end-systolic area, cm2 | 26±6 | 22±7 | 0.09 |
| RV cardiac index, ml/m2 | 2.5±0.4 | 2.8±0.5 | 0.07 |
| Patients that initiation of heart failure therapy(n=31) | |||
| Variables | Pre- | Post- | p |
| RV global longitudinal strain, % | −17±3 | −16±4 | 0.3 |
| RV fractional area change, % | 30±7 | 31±8 | 0.4 |
| RV end-diastolic area, cm2 | 36±7 | 35±9 | 0.2 |
| RV end-systolic area, cm2 | 25±5 | 25±7 | 0.6 |
| RV cardiac index, ml/m2 | 2.5±0.3 | 2.7±0.6 | 0.09 |
| Patients that had escalation of heart failure therapy (n=48) | |||
| Variables | Pre- | Post- | p |
| RV global longitudinal strain, % | −18±3 | −16±4 | 0.09 |
| RV fractional area change, % | 31±10 | 28±8 | 0.4 |
| RV end-diastolic area, cm2 | 38±9 | 37±9 | 0.6 |
| RV end-systolic area, cm2 | 27±8 | 26±7 | 0.6 |
| RV cardiac index, ml/m2 | 2.4±0.6 | 2.5±0.7 | 0.7 |
RV: right ventricle
There were 13 patients that underwent CRT, 31 patients that had initiation of heart failure therapy after the baseline echocardiogram, and 48 patients that had escalation of heart failure therapy after the baseline echocardiogram. There was no improvement in RV-LS after CRT, or after the initiation or escalation of heart failure therapy (Table 7).
DISCUSION
We analyzed serial echocardiograms in 186 adults with cc-TGA to determine the risk factors for, and prognostic implications of progressive RVD in this population. The main findings are: (1) Overall, there was a temporal decline in RV systolic function (as measured by RV-LS) during a 5-year imaging follow-up, and the risk factors associated with progressive RVD (temporal decline in RV-LS) were hypertension, LV dysfunction, and LV pacing; (2) Progressive RVD was associated with cardiovascular events, independent of baseline RV systolic function; (3) Tricuspid valve replacement was associated with a postoperative improvement in RV systolic function, and the extent of postoperative improvement in RV systolic function was related to preoperative RV systolic function.
Previous studies have reported prevalence of RVD in adults with cc-TGA ranging from 55–80% depending on the age of the study population, and the imaging modality used for assessing RVD.4, 5, 7, 14, 18 Several risk factors for RVD have been described, and these factors include myocardial ischemia due to a single coronary artery blood supply of the RV, ventricular dyssynchrony from LV pacing, and ventriculo-ventricular interaction driven by LV systolic dysfunction especially in patients with associated pulmonary valve disease.4, 5, 7, 14, 18 Similar to these prior studies, we also identified LV pacing and LV systolic dysfunction as risk factors for progressive RVD.
An important observation from the current study was that RV systolic function was not ‘static’ but rather declined gradually overtime, and this is very concerning. The temporal decline in RV systolic function did not occur at the same rate for all patients, but it occurred more rapidly in patients with risk factors such as LV pacing, LV systolic dysfunction, and a diagnosis of systemic hypertension. Systemic hypertension has been postulated as a potential cause of RVD,19, 20 but this has not been empirical tested, hence the novelty of this result. Another interesting observation from the study was that the mean systolic blood pressure for the cohort was 129±14 mmHg, and this suggests that minimal blood pressure elevation can accelerate deterioration of systemic RV systolic function in this population. We postulate that this is most likely due to the inability of the RV to remodel adequately to maintain cardiac output is the setting of elevated afterload of the systemic circulation resulting in ‘ventricular-afterload mismatch’. It may be important to screen for hypertension meticulously using different modalities such as ambulatory and home blood pressure monitoring, and to have a lower threshold for initiating and optimizing antihypertensive/afterload reduction therapy in cc-TGA patients, especially those with declining RV systolic function.
Although tricuspid regurgitation is a common and well recognized complication of cc-TGA, our data suggested that tricuspid regurgitation severity is not directly linked to progressive RVD. Rather it is the RV adaptation to the volume load of tricuspid regurgitation that is important. We observed that patients with better preoperative RV systolic function were more likely to have a postoperative improvement of RV systolic function after tricuspid valve replacement. This has important clinical implications in the management of patients with moderate tricuspid regurgitation, especially when regurgitation is not considered severe enough to recommend tricuspid valve replacement. In such patients, the RV systolic function should be closely monitored, and such patients should, perhaps, undergo tricuspid valve replacement if there is a subtle decline in RV systolic function without waiting for the tricuspid regurgitation to become severe.
It is also important to highlight the negative findings from this study. We did not observe any improvement in RV systolic function following CRT. However, it is important to emphasize that only 13 patients underwent CRT in our cohort, and hence the study may not have been powered to detect a difference in outcomes for this particular analysis. Although CRT has been proposed as the preferred therapy in patients with cc-TGA requiring pacing,9, 20 the data supporting the use of CRT in cc-TGA are rather limited, and the largest trial to date was based on a cohort 13 patients with cc-TGA that showed improvement in RV systolic function following CRT.8, 12, 13
The role of medical therapy for treatment of RVD remain controversial, and the largest double blinded placebo-controlled clinical trial till date did not demonstrated any clinical benefit (RV ejection fraction, RV volumes and peak oxygen consumption) in 25 cc-TGA patients randomized to valsartan vs placebo.21 Consistent with these studies, we did not observe any improvement in the subset of patients that had initiation or intensification of heart failure therapy. However, we did not have a control arm, and we were unable to verify drug compliance or blood pressure changes because of the retrospective study design. Since RAAS antagonists, which are the cornerstone of heart failure therapy, are also the first line therapies for hypertension, perhaps, one should aim at using RAAS antagonist to maintain strict blood pressure control in this population.
Limitation
This is a retrospective single center study, and hence it is prone to selection and referral bias as well as other limitations inherent in retrospective study design. We assessed RV strain from the apical 4-chamber view only, rather than from multiple imaging planes, and as a result RV-LS assessed in this study may not refect the global RV systolic function. Our subgroup analyses assessing the effect of therapies (tricuspid valve replacement, CRT, and heart failure therapy) were based on a non-randomized sample, and the absence of a control arm limits the inference that can be drawn from the results.
CONCLUSION
Although RVD is a well-recognized complication of cc-TGA, the current study demonstrated that patients with cc-TGA were at risk for progressive RVD (temporal decline in RV systolic function). The rate of decline in RV systolic function was related to certain risk factors such as LV pacing, hypertension, and LV dysfunction. Additionally, tricuspid valve replacement was associated with a postoperative improvement in RV systolic function, and the extent of postoperative improvement in RV systolic function was related to preoperatives RV systolic function. Collectively, these data suggest that strict blood pressure control and tricuspid valve replacement prior to the onset of RVD may prevent progressive RVD. However, prospective studies are required to empirically validate these hypotheses.
PERSPECTIVES.
Competencies in Medical Knowledge
Patients with cc-TGA were at risk for progressive RVD (temporal decline in RV systolic function), and the rate of decline in RV systolic function was related to certain risk factors such as LV pacing, hypertension, and LV dysfunction. Tricuspid valve replacement was associated with a postoperative improvement in RV systolic function, and the extent of postoperative improvement in RV systolic function was related to preoperatives RV systolic function.
Translational Outlook
These data suggest that strict blood pressure control and tricuspid valve replacement prior to the onset of RVD may prevent progressive RVD. However, prospective studies are required to empirically validate these hypotheses.
Acknowledgement:
James Welper and Katrina Tollefsrud performed offline image analysis for this study.
Funding:
Dr. Egbe is supported by National Heart, Lung, and Blood Institute (NHLBI) grants (R01 HL158517 and K23 HL141448). The MACHD Registry is supported by the Al-Bahar Research grant.
Abbreviations:
- LV
left ventricle
- RV
right ventricle
- FAC
fractional area change
- LS
longitudinal strain
- cc-TGA
congenitally corrected transposition of great arteries
- RVD
right ventricular systolic dysfunction
- CRT
cardiac resynchronization therapy
- ICC
intraclass correlation coefficient
- RAAS
renin angiotensin aldosterone system
- CIED
cardiac implantable electronic devises
- TAPSE
tricuspid annular plane systolic excursion
- s’
tissue Doppler systolic velocity
- β±SE
beta coefficient ± standard error
- CI
confidence interval
- IDI
integrated discrimination index
- β±SE
beta coefficient ± standard error
- CI
confidence interval
- RA
right atrial
- IVC
inferior venae cava
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of Interest: none
Disclosures: none
REFERENCES
- 1.Van Praagh R. What is congenitally corrected transposition? The New England journal of medicine. 1970;282:1097–8. [DOI] [PubMed] [Google Scholar]
- 2.Van Praagh R, Papagiannis J, Grunenfelder J, Bartram U and Martanovic P. Pathologic anatomy of corrected transposition of the great arteries: medical and surgical implications. American heart journal. 1998;135:772–85. [DOI] [PubMed] [Google Scholar]
- 3.Graham TP Jr., Parrish MD, Boucek RJ Jr., Boerth RC, Breitweser JA, Thompson S, Robertson RM, Morgan JR and Friesinger GC. Assessment of ventricular size and function in congenitally corrected transposition of the great arteries. The American journal of cardiology. 1983;51:244–51. [DOI] [PubMed] [Google Scholar]
- 4.Diller GP, Radojevic J, Kempny A, Alonso-Gonzalez R, Emmanouil L, Orwat S, Swan L, Uebing A, Li W, Dimopoulos K, Gatzoulis MA and Baumgartner H. Systemic right ventricular longitudinal strain is reduced in adults with transposition of the great arteries, relates to subpulmonary ventricular function, and predicts adverse clinical outcome. American heart journal. 2012;163:859–66. [DOI] [PubMed] [Google Scholar]
- 5.Dobson R, Danton M, Nicola W and Hamish W. The natural and unnatural history of the systemic right ventricle in adult survivors. The Journal of thoracic and cardiovascular surgery. 2013;145:1493–501; discussion 1501–3. [DOI] [PubMed] [Google Scholar]
- 6.Barrios PA, Zia A, Pettersson G, Najm HK, Rajeswaran J, Bhimani S, Karamlou T and Members of the cc TGAWG. Outcomes of treatment pathways in 240 patients with congenitally corrected transposition of great arteries. The Journal of thoracic and cardiovascular surgery. 2021;161:1080–1093 e4. [DOI] [PubMed] [Google Scholar]
- 7.Mongeon FP, Connolly HM, Dearani JA, Li Z and Warnes CA. Congenitally corrected transposition of the great arteries ventricular function at the time of systemic atrioventricular valve replacement predicts long-term ventricular function. Journal of the American College of Cardiology. 2011;57:2008–17. [DOI] [PubMed] [Google Scholar]
- 8.Hofferberth SC, Alexander ME, Mah DY, Bautista-Hernandez V, del Nido PJ and Fynn-Thompson F. Impact of pacing on systemic ventricular function in L-transposition of the great arteries. The Journal of thoracic and cardiovascular surgery. 2016;151:131–8. [DOI] [PubMed] [Google Scholar]
- 9.Connolly HM, Miranda WR, Egbe AC and Warnes CA. Management of the Adult Patient With Congenitally Corrected Transposition: Challenges and Uncertainties. Seminars in thoracic and cardiovascular surgery Pediatric cardiac surgery annual. 2019;22:61–65. [DOI] [PubMed] [Google Scholar]
- 10.Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, Flachskampf FA, Foster E, Goldstein SA, Kuznetsova T, Lancellotti P, Muraru D, Picard MH, Rietzschel ER, Rudski L, Spencer KT, Tsang W and Voigt JU. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Journal of the American Society of Echocardiography : official publication of the American Society of Echocardiography. 2015;28:1–39 e14. [DOI] [PubMed] [Google Scholar]
- 11.Rudski LG, Lai WW, Afilalo J, Hua L, Handschumacher MD, Chandrasekaran K, Solomon SD, Louie EK and Schiller NB. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. Journal of the American Society of Echocardiography : official publication of the American Society of Echocardiography. 2010;23:685–713; quiz 786–8. [DOI] [PubMed] [Google Scholar]
- 12.Dubin AM, Janousek J, Rhee E, Strieper MJ, Cecchin F, Law IH, Shannon KM, Temple J, Rosenthal E, Zimmerman FJ, Davis A, Karpawich PP, Al Ahmad A, Vetter VL, Kertesz NJ, Shah M, Snyder C, Stephenson E, Emmel M, Sanatani S, Kanter R, Batra A and Collins KK. Resynchronization therapy in pediatric and congenital heart disease patients: an international multicenter study. Journal of the American College of Cardiology. 2005;46:2277–83. [DOI] [PubMed] [Google Scholar]
- 13.Diller GP, Okonko D, Uebing A, Ho SY and Gatzoulis MA. Cardiac resynchronization therapy for adult congenital heart disease patients with a systemic right ventricle: analysis of feasibility and review of early experience. Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2006;8:267–72. [DOI] [PubMed] [Google Scholar]
- 14.Liu R, Pang K, Li S, Zhang B, Rui L, Lin Y, Wang C and Ma K. The fate of congenitally corrected transposition of the great arteries unoperated before adulthood. The Annals of thoracic surgery. 2020. [DOI] [PubMed] [Google Scholar]
- 15.Chatterjee A, Miller NJ, Cribbs MG, Mukherjee A and Law MA. Systematic review and meta-analysis of outcomes of anatomic repair in congenitally corrected transposition of great arteries. World J Cardiol. 2020;12:427–436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mitchell C, Rahko PS, Blauwet LA, Canaday B, Finstuen JA, Foster MC, Horton K, Ogunyankin KO, Palma RA and Velazquez EJ. Guidelines for Performing a Comprehensive Transthoracic Echocardiographic Examination in Adults: Recommendations from the American Society of Echocardiography. Journal of the American Society of Echocardiography : official publication of the American Society of Echocardiography. 2019;32:1–64. [DOI] [PubMed] [Google Scholar]
- 17.Cook NR. Quantifying the added value of new biomarkers: how and how not. Diagn Progn Res. 2018;2:14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Graham TP Jr., Bernard YD, Mellen BG, Celermajer D, Baumgartner H, Cetta F, Connolly HM, Davidson WR, Dellborg M, Foster E, Gersony WM, Gessner IH, Hurwitz RA, Kaemmerer H, Kugler JD, Murphy DJ, Noonan JA, Morris C, Perloff JK, Sanders SP and Sutherland JL. Long-term outcome in congenitally corrected transposition of the great arteries: a multi-institutional study. Journal of the American College of Cardiology. 2000;36:255–61. [DOI] [PubMed] [Google Scholar]
- 19.Fredriksen PM, Chen A, Veldtman G, Hechter S, Therrien J and Webb G. Exercise capacity in adult patients with congenitally corrected transposition of the great arteries. Heart. 2001;85:191–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Filippov AA, Del Nido PJ and Vasilyev NV. Management of Systemic Right Ventricular Failure in Patients With Congenitally Corrected Transposition of the Great Arteries. Circulation. 2016;134:1293–1302. [DOI] [PubMed] [Google Scholar]
- 21.van der Bom T, Winter MM, Bouma BJ, Groenink M, Vliegen HW, Pieper PG, van Dijk AP, Sieswerda GT, Roos-Hesselink JW, Zwinderman AH and Mulder BJ. Effect of valsartan on systemic right ventricular function: a double-blind, randomized, placebo-controlled pilot trial. Circulation. 2013;127:322–30. [DOI] [PubMed] [Google Scholar]


