Abstract
With the emergence of various classes of blood glucose-lowering agents, choosing the appropriate drug for each patient is emphasized in diabetes management. Among incretin-based drugs, glucagon-like peptide 1 (GLP-1) receptor agonists are a promising therapeutic option for patients with diabetic kidney disease (DKD). Several cardiovascular outcome trials have demonstrated that GLP-1 receptor agonists have beneficial effects on cardiorenal outcomes beyond their blood glucose-lowering effects in patients with type 2 diabetes mellitus (T2DM). The renal protective effects of GLP-1 receptor agonists likely result from their direct actions on the kidney, in addition to their indirect actions that improve conventional risk factors for DKD, such as reducing blood glucose levels, blood pressure, and body weight. Inhibition of oxidative stress and inflammation and induction of natriuresis are major renoprotective mechanisms of GLP-1 analogues. Early evidence from the development of dual and triple combination agents suggests that GLP-1 receptor agonists will probably become popular treatment options for patients with T2DM.
Keywords: Diabetic nephropathies, Glucagon-like peptide 1, Type 2 diabetes mellitus
Introduction
The number of patients with diabetes mellitus (DM) continues to increase worldwide, and DM is the main cause of chronic kidney disease (CKD) and end-stage renal disease (ESRD) [1]. In Korea, the prevalence of diabetes was 13.8% in adults older than 30 years in 2018 [2], and it was predicted to be 29.2% in men and 19.7% in women by 2030 [3]. The total number of new patients who started renal replacement therapy (RRT) for ESRD increased from 10,000 in 2011 to 18,642 in 2019 [4], and the proportion of patients with DM as the underlying cause of ESRD increased from 19.5% in 1992 to 50.6% in 2012 [5], making DM the most common cause of ESRD in Korea [4]. Despite advances in medical technology and treatments, the need for RRT is increasing worldwide and is expected to more than double by 2030 compared with 2010 [6].
Diabetic kidney disease (DKD) is the main cause of morbidity and mortality in diabetes [7,8]. Therefore, inhibiting the onset and progression of DKD, in part by developing therapeutic approaches to prevent or delay it, is critical. Controlling blood sugar and blood pressure using angiotensin-converting enzyme inhibitors or angiotensin receptor blockers is the current goal in DKD management [9], and no special drugs or other therapeutic options are widely used to delay DKD progression. However, several cardiovascular outcome trials (CVOTs) have demonstrated that sodium-glucose cotransporter 2 (SGLT2) inhibitors and glucagon-like peptide 1 (GLP-1) receptor agonists have beneficial effects on cardiorenal outcomes, especially in patients with type 2 DM (T2DM) who are at high risk for cardiovascular disease (CVD) [10–12]. Based on the results of clinical trials, the current guidelines of the American Diabetes Association and Korean Diabetes Association recommend that clinicians consider prescribing SGLT2 inhibitors or GLP-1 receptor agonists after metformin as part of the glucose-lowering regimen for patients with T2DM and CKD [13,14]. In this review article, we focus on GLP-1 agonists and discuss the clinical and preclinical evidence for their nephroprotective effects and the potential mechanisms underlying those effects.
Physiology and metabolic effects of glucagon-like peptide 1
Oral intake of glucose causes the secretion of more insulin than does an injection of glucose due to the presence of gut hormones called incretins [15]. Gastrointestinal peptide (GIP) and the GLPs (GLP-1, GLP-2) are incretin hormones produced by enteroendocrine L-cells of the distal small bowel and colon [16]. In humans, fasting concentration of total GLP-1 ranges from 5 to 10 pmol/L and can increase to 40–50 pmol/L in response to meals [17]. Plasma concentration of biologically active, intact GLP-1 is much lower than that (fasting, <2 pmol/L; peak postprandial concentrations, 5–10 pmol/L) [18].
GLP-1 release after a meal occurs in a biphasic manner. An initial rapid rise in circulating GLP-1 level occurs 15 to 30 minutes after a meal, followed by a second minor peak at 90 to 120 minutes [19,20]. The rapid increase in GLP-1 secretion after meals is related to the proximal-distal loop regulated by neurotransmitters such as acetylcholine and gastrin-releasing peptide [21]. The second later peak of GLP-1 is believed to occur as ingested nutrients travel down the lumen and interact directly with distal L-cells [22,23].
Native GLP-1 has an extremely short half-life, less than 2 minutes, due to cleavage by dipeptidyl-peptidase IV (DPP IV) enzymes and renal elimination [24]. DPP IV enzymes cleave the active forms of GLP-17-36 and GLP-17-37 to produce inactive GLP-19-36 or GLP-19-37, respectively, which have low affinity for the GLP-1 receptor [25,26]. Only 10% to 15% of secreted GLP-1 reaches the pancreas via systemic circulation [25], and both the active and inactive forms of GLP-1 are rapidly cleared from the circulation via the kidneys. Although the initial DPP IV-mediated degradation of GLP-1 is unaffected by impairments in renal function, GLP-1 clearance is delayed in patients with renal insufficiency [24].
In humans, the GLP-1 receptor is expressed in the pancreas, lungs, brain, kidneys, stomach, and heart but not in the liver, skeletal muscle, or adipose tissue [27]. Binding between GLP-1 and its receptor activates adenylate cyclase, which is followed by increase in cyclic AMP level and cytoplasmic Ca+2 that induces insulin secretion [28]. In addition to GLP-1’s short-term effect of enhancing the glucose-dependent stimulation of insulin secretion, continuous GLP-1 activation also increases insulin synthesis [29], modulates β-cell proliferation [30], and inhibits β-cell apoptosis [31] and glucagon release [32]. Incretin hormones also decrease gastric emptying [33], inhibit food intake [34], and increase natriuresis and diuresis [35,36].
Classification of glucagon-like peptide 1 receptor agonists
GLP-1 receptor agonists have two main backbone structures and are classified as exendin-4- or human GLP-1-based compounds [37]. They are divided into short- and long-acting agents, and some formulations are mixed with insulin (Table 1). Exendin-4 is a protein isolated in 1992 from the saliva of the Gila monster lizard (Heloderma suspectum) [38]. This protein is composed of 39 amino acids and has 53% similarity in base sequence to native human GLP-1. Exenatide and lixisenatide are based on the structure of exendin-4. Exenatide is a recombinant form of the peptide exendin-4 and was the first GLP-1 receptor agonist to be developed for T2DM treatment. Lixisenatide is an exendin-4 analog with an additional six lysines attached to the C-terminus, which gives it a longer half-life than exenatide. These exendin-4-based agents have relatively short half-lives (~3 hours) and strongly inhibit gastric emptying [39], which can cause gastrointestinal side-effects such as nausea. But they also have robust postprandial antihyperglycemic effects and could potentially replace rapid-acting mealtime insulin [39]. These shorter-acting agents are less effective at decreasing fasting glucose levels because of their short half-lives.
Table 1.
Characteristics of GLP-1 receptor agonists
| Generic | Commercial | Backbone | Dosage | Administration | Half-life | Renal dose adjustment | Route of elimination |
|---|---|---|---|---|---|---|---|
| Short-acting compound | |||||||
| Exenatide | Byetta | Exendin-4 | 5 μg, 10 μg | Twice daily, SC | ~2.4 hr | Not recommended for patients with CrCl < 30 mL/min; caution needed for patients with CrCl 30–50 mL/min | Glomerular filtration followed by proteolysis; eliminated in the urine |
| Lixisenatide | Lyxumia | Exendin-4 | 10 μg, 20 μg | Once daily, SC | ~3 hr | Not recommended for patients with CrCl < 30 mL/min | Glomerular filtration and proteolysis; excreted in the urine |
| Long-acting compound | |||||||
| Liraglutide | Victoza | Human GLP-1 | 0.6–1.8 mg | Once daily, SC | ~13 hr | No dosage adjustment required; not recommended for patients with CrCl < 15 mL/min | Proteolysis; excreted via urine and feces |
| Liraglutide | Saxenda | Human GLP-1 | 0.6–3 mg | Once daily, SC | ~13 hr | No dosage adjustment required; not recommended for patients with CrCl < 15 mL/min | Proteolysis; excreted via urine and feces |
| Exenatide ER | Bydureon | Exendin-4 | 2 mg | Once weekly, SC | ~1 wk | Not recommended for patients with an eGFR < 45 mL/min/1.73 m2 or ESRD | Glomerular filtration followed by proteolysis; eliminated in the urine |
| Dulaglutide | Trulicity | Human GLP-1 | 0.75 mg, 1.5 mg | Once weekly, SC | ~5 day | No dosage adjustment required; not recommended for patients with CrCl < 15 mL/min | Proteolytic degradation |
| Semaglutide | Ozempic | Human GLP-1 | 0.5 mg, 1.0 mg | Once weekly, SC | ~1 wk | No dosage adjustment required; not recommended for patients with CrCl < 15 mL/min | Proteolysis; excreted via urine and feces |
| Albiglutidea | Tanzeum | Human GLP-1 | 30 mg, 50 mg | Once weekly, SC | ~5 day | No dosage adjustment required; not recommended for patients with CrCl < 15 mL/min | Not available |
| Oral agent | |||||||
| Semaglutide | Rybelsus | Human GLP-1 | 3 mg, 7 mg, 14 mg | Once daily, oral | ~1 wk | No dosage adjustment required | Proteolysis; excreted via urine and feces |
| Fixed-dose combination | |||||||
| Lixisenatide + glargine | Soliqua | Exendin-4 | 20 μg/iGlar 40 IU, 20 μg/iGlar 60 IU | Once daily, SC | ~3 hr | Closely monitor patients with CrCl 15–30 mL/min; not recommended for patients with CrCl < 15 mL/min | Glomerular filtration and proteolysis; excreted in the urine |
| Liraglutide + degludec | Xultophy | Human GLP-1 | 1.8 mg/iDeg 50 IU | Once daily, SC | ~13 hr | Not studied in severe renal impairment; liraglutide is not recommended for patients with CrCl < 15 mL/min | Proteolysis; excreted via urine and feces |
CrCl, creatinine clearance; eGFR, estimated glomerular filtration rate; ER, extended-release; ESRD, end-stage renal disease; GLP-1, glucagon-like peptide-1; iDeg, insulin degludec; iGlar, insulin glargine; SC, subcutaneous.
Marketing was discontinued in 2018.
Human GLP-1–based agents are more structurally similar to native GLP-1 than to those based on exendin-4. They have 90% to 97% amino-acid homology to endogenous human GLP-1 and an extended half-life conferred by DPP IV resistance and noncovalent binding to serum albumin. These longer-acting agents lead to a greater reduction of fasting plasma glucose and hemoglobin A1c (HbA1c) levels than the shorter-acting agents [39,40]. The human GLP-1 compounds are liraglutide, albiglutide, dulaglutide, and semaglutide, all of which are injectable agents. Albiglutide and dulaglutide are large molecules conjugated to large proteins, which extends their half-life and enables once-weekly administration. Semaglutide is available in both injectable and oral forms. With withdrawal of albiglutide from the market for commercial reasons, liraglutide, dulaglutide, and semaglutide (oral and subcutaneous) are the currently available, approved human GLP-1 receptor agonists.
Table 1 shows the recommended uses of GLP-1 receptor agonists according to estimated glomerular filtration rate (eGFR). Human GLP-1–derived dulaglutide, liraglutide, and semaglutide are not excreted via the kidneys and can be used down to an eGFR of 15 mL/min/1.73 m2; there is insufficient experience to recommend using those agents for eGFR values lower than that [41]. Conversely, exenatide and lixisenatide, which are eliminated by the kidneys, are contraindicated below an eGFR of 30 mL/min/1.73 m2 due to the risk of accumulation and toxicity [24]. Exenatide should be used with caution in patients with an eGFR of 30–50 mL/min/1.73 m2 (Table 1).
Renal effects of glucagon-like peptide 1 receptor agonists in patients with type 2 diabetes mellitus
Several CVOTs have examined GLP-1 receptor agonists; however, none have focused on the primary endpoint of renal events; renal outcomes have been reported as secondary outcomes after primary cardiovascular outcomes. This section focuses on the renal outcomes of GLP-1 receptor agonist treatment reported by randomized controlled trials (Table 2).
Table 2.
Renal endpoints in cardiovascular outcome trials of GLP-1 receptor agonists
| ELIXA [42] | LEADER [44] | SUSTAIN-6 [46] | EXSCEL [48] | HARMONY [51] | REWIND [53] | PIONEER-6 [55] | AMPLITUDE-O [56] | |
|---|---|---|---|---|---|---|---|---|
| Drug | Lixisenatide | Liraglutide | Semaglutide | Exenatide ER | Albiglutidea | Dulaglutide | Semaglutide (oral) | Efpeglenatide |
| Participants (n) | 6,068 | 9,340 | 3,297 | 14,752 | 9,463 | 9,901 | 3,183 | 4,076 |
| Median follow-up (yr) | 2.1 | 3.8 | 2.1 | 3.2 | 1.6 | 5.4 | 1.3 | 1.8 |
| Baseline HbA1c (%) | 7.7 | 8.7 | 8.7 | 8 | 8.7 | 7.2 | 8.2 | 8.9 |
| Baseline BP (mmHg) | 130 | 136/77 | 136/77 | 135/78 | 135/77 | 137/78 | 136/76 | 135/77 |
| Established CVD (%) | 100 | 81 | 83.0 | 73.1 | 100 | 31 | 84.7 | 89.6 |
| Baseline eGFR < 60 mL/min/1.73 m2 (%) | 23.2 | 23.1 | 24.1 | 21.6 | 23.5 | 22.2 | 26.9 | 31.6 |
| Baseline eGFR, mL/min/1.73 m2 | 78 | 80 | 80 | 77 | 79 | 75 | 74 | 72 |
| Albuminuria (%) | 25.3 | 11.0 | NA | 22.0 | NA | 34.5 | 33 | 48.5 |
| ACEI or ARB (%) | 85.0 | 82.8 | 83.5 | 79.9 | 81.6 | 81.5 | NA | 80.0 |
| Renal composite outcomesb | 0.84 (0.68–1.02) | 0.78 (0.67–0.92) | 0.64 (0.46–0.88) | 0.88 (0.76–1.01) | NA | 0.85 (0.77–0.93) | NA | 0.68 (0.57–0.79) |
| New-onset persistent macroalbuminuriab | 0.81 (0.66–0.99) | 0.74 (0.60–0.91) | 0.54 (0.37–0.77) | 0.87(0.70–1.07) | NA | 0.77 (0.68–0.87) | NA | 0.68 (0.58–0.80) |
| Persistent doubling of serum creatinineb | 1.16 (0.74–1.83) | 0.89 (0.67–1.19) | 1.28 (0.64–2.58) | NA | NA | NA | NA | NA |
| End-stage renal diseaseb | NA | 0.87 (0.61–1.24) | 0.91 (0.40–2.07) | NA | NA | 0.75 (0.39–1.44) | NA | NA |
| Death due to renal diseaseb | NA | 1.59 (0.52–4.87) | NA | NA | NA | NA | NA | NA |
ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; BP, blood pressure; CVD, cardiovascular disease; eGFR, estimated glomerular filtration rate; ER, extended-release; GLP-1, glucagon-like peptide-1; HbA1c, hemoglobin A1c; NA, not assessed.
Marketing was discontinued in 2018.
Hazard ratio (95% confidence interval).
The first CVOT for a GLP-1 receptor agonist was the ELIXA (Evaluation of Lixisenatide in Acute Coronary Syndrome) trial, the results of which were published in 2015 [42]. A total of 6,068 participants with T2DM, history of myocardial infarction or unstable angina, average baseline HbA1c of 7.7%, and a median follow-up of 25 months was enrolled. Although renal events were not investigated in the primary ELIXA trial, an exploratory analysis of renal outcomes was performed [43]. After a median follow-up of 108 weeks, lixisenatide reduced progression of the urinary albumin-to-creatinine ratio (UACR) in macroalbuminuric patients and was associated with a lower risk of new-onset macroalbuminuria after adjustment for baseline and on-trial HbA1c and other traditional renal risk factors. No significant differences in eGFR decline were identified between treatment groups. This study had a short follow-up period of 2 years, a high percentage of participants on statin therapy, and low compliance with the medication compared with the other trials in Table 2.
In the LEADER (Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results) trial published in 2016 [44], participants with T2DM were either 50 years of age or older with at least one cardiovascular condition or 60 years or older with at least one cardiovascular risk factor. A total of 9,340 participants with a median follow-up of 3.8 years was enrolled, and the average baseline HbA1c was 8.7%. Approximately 23% of participants had moderate-to-severe CKD, suggesting a very high-risk population. Of note, this trial included 220 individuals with an eGFR of 15–30 mL/min/1.73 m2. Liraglutide decreased the risk of the secondary composite renal endpoint (new-onset macroalbuminuria, sustained serum creatinine duplication, initiation of RRT, or renal death) by 22% (hazard ratio, 0.78; 95% confidence interval [CI], 0.67–0.92; p = 0.003) [45]. This finding was driven primarily by a reduction in new-onset persistent macroalbuminuria. That study was the first to show that a GLP-1 agonist had cardiovascular benefit, although it might not apply in patients with low cardiovascular risk.
The SUSTAIN-6 (Trial to Evaluate Cardiovascular and Other Long-term Outcomes with Semaglutide in Subjects with Type 2 Diabetes) was the next CVOT, also published in 2016 [46]. A total of 3,297 patients was randomly assigned, and 3,232 patients completed the trial over a median follow-up of 2.1 years. Eighty-three percent of participants had established CVD, CKD, or both, and the mean HbA1c of the total study population was 8.7%. Once-weekly semaglutide effected a 36% reduction (HR, 0.64; 95% CI, 0.46–0.88; p = 0.005) in the secondary combined renal endpoint (new-onset macroalbuminuria, doubling of serum creatinine, eGFR of <45 mL/min/1.73 m2, initiation of RRT, or renal death). This result was mainly driven by a reduction in new-onset macroalbuminuria. Across the SUSTAIN 1–7 trials [47], semaglutide lowered albuminuria compared with placebo beginning as early as 16 weeks and lasting over the entire treatment period.
The next trial, published in 2017, was the EXSCEL (Exenatide Study of Cardiovascular Event Lowering) trial to evaluate the effect of once-weekly exenatide extended-release (ER) on cardiovascular outcomes in participants with T2DM [48]. A total of 14,752 patients, of whom 73.1% had previous CVD, was followed for a median of 3.2 years. Exenatide ER had no significant effects on renal outcomes in an additional analysis of EXSCEL trial data [49]. Twice-daily exenatide also did not affect eGFR or albuminuria compared with insulin glargine over the 52-week study period [50].
The cardiovascular effects of albiglutide were evaluated in patients with T2DM and CVD in the HARMONY trial (NCT02465515) [51], published in 2018. A total of 9,463 participants with a median HbA1c of 8.7% was enrolled; this was a relatively high-risk population with high baseline glucose levels. After a median of 1.6 years of follow-up, albiglutide conferred no significant benefit in slowing the rate of eGFR decline.
The renal outcomes of dulaglutide treatment were investigated in two representative trials. The first study was the AWARD-7 trial (NCT01621178), published in 2018 [52]. Five hundred seventy-seven participants with T2DM and moderate-to-severe CKD were included in this trial. A once-weekly injection of dulaglutide was associated with a significantly smaller decline in eGFR compared with insulin glargine over 52 weeks. The mean eGFR decline with 1.5-mg dulaglutide was about 10% of that observed with insulin glargine (–0.5 mL/min/1.73 m2 in the 1.5-mg dulaglutide group compared with –5.5 mL/min/1.73 m2 in the insulin glargine group). This association between dulaglutide and reduced eGFR decline was most evident in participants with macroalbuminuria. Another study of the effects of injectable dulaglutide on cardiovascular outcomes in T2DM was the REWIND (Researching Cardiovascular Events with a Weekly Incretin in Diabetes) trial [53,54], published in 2019. This study was designed to demonstrate superiority, unlike the previous trials. A total of 9,901 participants with T2DM was followed up for a median of 5.4 years, a longer period than in the previous trials. This trial was unique in that the participants were low risk, with an average baseline HbA1c of 7.2%, median eGFR of 74.9 mL/min/1.73 m2, baseline prevalence of CVD of 31.5%, and baseline prevalence of albuminuria of 35.0%. The composite renal outcome occurred significantly less frequently in the dulaglutide group than in the placebo group (HR, 0.85; 95% CI, 0.77–0.93; p = 0.0004), and the largest effect was a reduction in the development of macroalbuminuria in the dulaglutide group (HR, 0.77; 95% CI, 0.68–0.87; p < 0.0001).
The PIONEER 6 (Peptide Innovation for Early Diabetes Treatment) trial was designed to evaluate the cardiovascular outcomes from once-daily oral semaglutide in T2DM patients at high cardiovascular risk [55], and its results were published in 2019. This study recruited 3,183 participants who were followed up for a median of 15.9 months, which is the shortest duration of the trials listed in Table 2. However, no renal endpoint was predefined for assessment in this trial.
The most recent CVOT for GLP-1 agonists was the AMPLITUDE-O (Effect of Efpeglenatide on Cardiovascular Outcomes) trial in patients with T2DM and a history of either CVD or CKD [56]; the results were published in 2021. Once-weekly injectable efpeglenatide is a new exendin-4-based GLP-1 receptor agonist. A total of 4,076 participants was enrolled and followed up for a median of 1.81 years. Compared with placebo, efpeglenatide led to a 32% lower risk of a composite renal outcome event (incident macroalbuminuria, increase in UACR of ≥30% from baseline, sustained decrease in eGFR of ≥40%, initiation of RRT, or sustained eGFR of <15 mL/min/1.73 m2), independently of baseline use of SGLT2 inhibitors or metformin and baseline eGFR (HR, 0.68; 95% CI, 0.57–0.79; p < 0.001). However, a kidney function outcome event, defined as a composite of a decrease in eGFR of at least 40% for ≥30 days, ESRD, or death from any cause, did not differ between the efpeglenatide group and the placebo group (HR, 0.77; 95% CI, 0.57–1.02; p = 0.07).
Suggested nephroprotective mechanisms of glucagon-like peptide 1 receptor agonists
Indirect effects by improving conventional risk factors for diabetic kidney disease
Hyperglycemia plays a critical role in the pathogenesis of DKD [57,58], and GLP-1 receptor agonists have potent glucose-lowering effects [59–62]. The Kidney Disease: Improving Global Outcomes (KDIGO) 2020 clinical practice guidelines recommend GLP-1 receptor agonists as an excellent option for patients with DKD who have not achieved their glycemic target or as an alternative for patients unable to tolerate metformin or an SGLT2 inhibitor [63]. Although glucose-independent mechanisms are also emphasized, the antihyperglycemic effects of GLP-1 receptor agonists are thought to contribute to their nephroprotective effects in patients with DKD. Furthermore, GLP-1 receptor agonists induce reduction in body weight, blood pressure, and dyslipidemia, which could also contribute to their antialbuminuric effects [64,65].
In the LEADER trial [44], the liraglutide group showed a 0.4% reduction in HbA1c compared with the placebo group. Weight loss was 2.3 kg higher and systolic blood pressure was 1.2 mmHg lower in the liraglutide group than in the placebo group. In the REWIND trial [54], participants in the once-weekly 1.5-mg dulaglutide group had a 0.61% lower HbA1c, 1.46 kg lower body weight, and 1.7 mmHg lower systolic blood pressure than participants in the placebo group. In the SUSTAIN-6 trial [46], the mean HbA1c level was 1.0 percentage point lower, mean body weight was decreased by 4.3 kg more, and mean systolic blood pressure was 2.6 mmHg lower in the group receiving 1.0 mg of semaglutide once weekly than in the placebo group. In the PIONEER 5 trial [55], once-daily oral semaglutide (14 mg) was superior to placebo at reducing HbA1c and body weight in patients with T2DM. However, statistical correction for on-trial HbA1c level, blood pressure change, and bodyweight decrease did not significantly alter the observed decreases in albuminuria induced by GLP-1 receptor agonists in several CVOTs [66], suggesting that the renal protective effects of GLP-1 receptor agonists are not entirely due to improvements in risk factors.
In addition to its actions on body weight, blood pressure, and glucose, GLP-1 also regulates lipid metabolism. Dyslipidemia is a strong risk factor for both CKD and DKD. Experimental studies have provided data to support the notion that lipid abnormalities contribute to initiation and progression of glomerular disease [67]. A systematic review and meta-analysis of 35 trials showed that GLP-1 receptor agonists are associated with reductions in total and low-density lipoprotein cholesterol and triglyceride levels [68]. GLP-1 inhibits gastric lipase secretion [69] and intestinal lipoprotein and chylomicron production in humans [70]. GLP-1 receptor signaling reduces hepatic triglyceride content and impairs lipogenesis in the liver by stimulating the AMP-activated protein kinase pathway [71,72]. It also increases peripheral use of triglyceride-rich lipoproteins through increased burning of fat and activation of brown adipose tissue function [73,74]. However, it is uncertain whether those actions directly contribute to the nephroprotective effects of GLP-1 receptor agonists.
Potential direct mechanisms accounting for the renal protective effects of glucagon-like peptide 1 receptor agonists
The GLP-1 receptor is expressed in the renal cortex and vasculature, as well as proximal tubular cells [75,76], although uncertainties remain regarding receptor localization in the kidney due to lack of antibodies with high sensitivity and specificity. Inhibition of oxidative stress and inflammation, induction of natriuresis, and reduction of intraglomerular pressure are potential direct mechanisms underlying the renal protective effects of GLP-1 analogues (Fig. 1). Systemic oxidative stress increases the stage of incipient DKD [77]. A study in diabetic rats revealed that recombinant human GLP-1 attenuated oxidative stress in the glomeruli and in glomerular microvascular endothelial cells by inhibiting protein kinase C and activating protein kinase A (PKA) [78]. Liraglutide also reduced oxidative stress and albuminuria in streptozotocin-induced type 1 DM rats via PKA-mediated inhibition of renal nicotinamide adenine dinucleotide phosphate oxidases [79]. Exendin-4 was shown to activate the Nrf2 signaling pathway, which plays a key role in preventing oxidative stress and maintaining redox homeostasis, in vascular smooth muscle cells [80,81].
Figure 1. The mechanisms underlying the nephroprotective effects of GLP-1 receptor agonists.
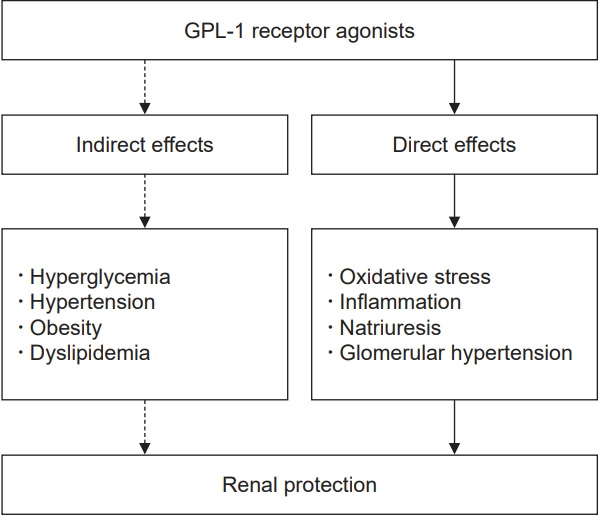
GLP-1, glucagon-like peptide 1.
Inflammation plays a central role in the development of DKD. Accumulating experimental data suggest that anti-inflammatory activity underlies the nephroprotective effects of GLP-1. GLP-1 receptor agonists decrease the production of proinflammatory cytokines, adhesion molecules, and profibrotic signaling [82–84]. Liraglutide inhibited renal tumor necrosis factor (TNF)-α-mediated nuclear factor kappa B (NF-κB) activation and mitogen-activated protein kinase pathway activation in the glomerular podocytes of an obesity-related glomerulopathy mouse model [82]. Exendin-4 attenuated albuminuria, glomerular hyperfiltration, glomerular hypertrophy, and mesangial matrix expansion without lowering blood glucose level in diabetic rats by inhibiting oxidative stress and NF-κB activation [83]. In humans, exenatide reduced reactive oxygen species generation and expression of NF-κB, TNF-α, interleukin-1β, c-Jun N-terminal kinase-1, toll-like receptor-4, and suppressor of cytokine signaling 3 in obese patients with T2DM, independent of weight loss [84]. Exenatide also reduced high-sensitivity C-reactive protein by 61% [85] and reduced urinary transforming growth factor-β1 and type IV collagen excretion in patients with T2DM [86]. Liraglutide treatment was associated with decreased levels of inflammatory cytokines and an increase in serum adiponectin level in obese patients with T2DM [87]. Liraglutide also improved oxidative stress by increasing glutathione concentration and decreasing serum lipid hydroperoxides and heme oxygenase-1 levels in subjects with T2DM, independent of its glucose-lowering effects [88].
The natriuretic effect of GLP-1 receptor agonists has been proposed to underly the GLP-1–induced reduction in blood pressure reported in large CVOTs. GLP-1–mediated natriuresis and diuresis appear to involve redistribution and reduction of Na+/H+ exchanger 3 (NHE3) activity, which is located at the brush border of renal proximal tubules [89]. GLP-1 receptor agonists phosphorylated NHE3 at the PKA consensus sites Ser552 and Ser605, reducing its activity [36]. GLP-1 receptor agonists also increased natriuresis and diuresis by increasing renal blood flow in rats [90]. Human studies have shown that GLP-1 infusion reduces proximal tubular sodium reabsorption and decreases plasma angiotensin II concentration [91]. In addition, a single subcutaneous injection of liraglutide increased sodium excretion in people with T2DM [92]. Inhibition of NHE3 by GLP-1 could also affect glomerular hemodynamics by activating tubuloglomerular feedback. The increase in sodium delivery to the macula densa due to low NHE3 activity results in afferent arteriolar vasoconstriction and lower glomerular hyperfiltration and pressure. Liraglutide is associated with an acute reduction in eGFR and subsequent stabilization over time, suggesting that GLP-1 has renal hemodynamic effects [93].
Ongoing studies and candidate drugs under development
The FLOW trial (NCT03819153) to evaluate the effect of once-weekly semaglutide on progression of renal impairment is currently in progress. The primary renal outcome comprises a persistent ≥50% reduction in eGFR or a persistent eGFR of <15 mL/min/1.73 m2, initiation of RRT, or death from kidney disease or CVD. This study recently began recruiting more than 3,000 T2DM patients with moderate/advanced CKD and albuminuria, and its estimated completion date is 2024. This trial will be the first to investigate the effects of a GLP-1 receptor agonist on primary kidney outcomes.
In addition, the SOUL trial (NCT03914326) is a currently ongoing CVOT to evaluate the hypothesis that oral semaglutide lowers the risk of cardiovascular events in T2DM patients at high risk for CVD. In this trial, the composite renal endpoint is a secondary outcome consisting of a persistent ≥50% reduction in eGFR or a persistent eGFR of <15 mL/min/1.73 m2, initiation of RRT, and renal death. Oral semaglutide received the approval of the U.S. Food and Drug Administration in September 2019.
Polypharmacology refers to the combination of several structurally related hormones into a single entity. Treatment with GLP-1/glucagon dual agonists produced weight loss and antihyperglycemic efficacy superior to that of GLP-1 selective agonists alone in mice with diet-induced obesity [94]. GLP-1 and glucagon are structurally similar, and glucagon also acts on the GLP-1 receptor [95], raising expectations that a combination of the two drugs could be more efficacious than the use of either drug on its own. Several phase 2 clinical trials of GLP-1/glucagon dual agonists are currently in progress. In addition, dual GLP-1/GIP agonists have prolonged half-lives due to fatty acylation or PEGylation. A once-weekly GLP-1/GIP co-agonist, named tirzepatide (LY3298176), was superior to dulaglutide in terms of weight loss and improved HbA1c level in a phase 2 study of patients with T2DM [96]. Phase 1 clinical trials for GLP-1/glucagon/GIP triple combination agents have been performed by Hanmi Pharmaceuticals (HM15211) and Novo Nordisk (NNC9204-1706).
GLP-1-based combination therapies have been found to offer metabolic benefits greater than those achieved by treatment with either compound alone. Based on the improved efficacy of GLP-1/glucagon and GLP-1/GIP co-agonists, it is reasonable to determine whether dual or triple agonists might provide greater efficacy than the respective mono-agonists. Various possible combinations are GLP-1 with GLP-2 [97], leptin [98], gastrin [99], amylin [100], peptide YY [101], cholecystokinin [102], insulin [103], adrenomedullin [104], fibroblast growth factor 21 [105], estrogen [106], dexamethasone [107], a proprotein convertase subtilisin/kexin type 9 antibody [108], melanocortin-4 agonist [109], farnesoid-x [110], or an SGLT2 inhibitor [111]. Future studies are required to evaluate whether any of these combinations of agents have nephroprotective effects superior to those of GLP-1 mono-agonists in DKD patients.
Conclusions and future perspectives
GLP-1 receptor agonists are promising therapeutic options for patients with DKD, with benefits beyond their blood glucose-lowering activity. These agents seem to predominantly affect macroalbuminuria, whereas their effects on hard renal endpoints are less clear. Although these agents can be used in CKD patients with an eGFR down to 15 mL/min/1.73 m2, the safety of GLP-1 receptor agonists in DKD patients with stage 5 CKD needs to be investigated. In terms of future research direction, more studies similar to the ongoing FLOW trial should be conducted to evaluate the primary kidney outcomes of GLP-1 receptor agonist treatment. In addition, it is necessary to explore whether combination treatment with GLP-1 receptor agonists and other classes of agents with beneficial effects on the kidney will have synergistic renoprotective effects in patients with DKD.
Footnotes
Conflicts of interest
All authors have no conflicts of interest to declare.
Funding
This research was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean government (MSIT) (2018R1D1A1B07049123, 2020R1F1A1074265) and by a Korea University grant (K1824431).
Authors’ contributions
Conceptualization, Funding acquisition: JHY, JAS
Investigation: SYP, DYL, NHK
Project administration: JAS
Writing–original draft: JHY
Writing–review & editing: SYP, DYL, NHK, JAS
All authors read and approved the final manuscript.
References
- 1.Alicic RZ, Rooney MT, Tuttle KR. Diabetic kidney disease: challenges, progress, and possibilities. Clin J Am Soc Nephrol. 2017;12:2032–2045. doi: 10.2215/CJN.11491116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jung CH, Son JW, Kang S, et al. Diabetes fact sheets in Korea, 2020: an appraisal of current status. Diabetes Metab J. 2021;45:1–10. doi: 10.4093/dmj.2020.0254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baik I. Projection of diabetes prevalence in Korean adults for the year 2030 using risk factors identified from national data. Diabetes Metab J. 2019;43:90–96. doi: 10.4093/dmj.2018.0043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hong YA, Ban TH, Kang CY, et al. Trends in epidemiologic characteristics of end-stage renal disease from 2019 Korean Renal Data System (KORDS) Kidney Res Clin Pract. 2021;40:52–61. doi: 10.23876/j.krcp.20.202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jin DC, Han JS. Renal replacement therapy in Korea, 2012. Kidney Res Clin Pract. 2014;33:9–18. doi: 10.1016/j.krcp.2014.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Liyanage T, Ninomiya T, Jha V, et al. Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet. 2015;385:1975–1982. doi: 10.1016/S0140-6736(14)61601-9. [DOI] [PubMed] [Google Scholar]
- 7.Li S, Wang J, Zhang B, Li X, Liu Y. Diabetes mellitus and cause-specific mortality: a population-based study. Diabetes Metab J. 2019;43:319–341. doi: 10.4093/dmj.2018.0060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Afkarian M, Sachs MC, Kestenbaum B, et al. Kidney disease and increased mortality risk in type 2 diabetes. J Am Soc Nephrol. 2013;24:302–308. doi: 10.1681/ASN.2012070718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yamazaki T, Mimura I, Tanaka T, Nangaku M. Treatment of diabetic kidney disease: current and future. Diabetes Metab J. 2021;45:11–26. doi: 10.4093/dmj.2020.0217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kristensen SL, Rørth R, Jhund PS, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol. 2019;7:776–785. doi: 10.1016/S2213-8587(19)30249-9. [DOI] [PubMed] [Google Scholar]
- 11.Neuen BL, Young T, Heerspink HJ, et al. SGLT2 inhibitors for the prevention of kidney failure in patients with type 2 diabetes: a systematic review and meta-analysis. Lancet Diabetes Endocrinol. 2019;7:845–854. doi: 10.1016/S2213-8587(19)30256-6. [DOI] [PubMed] [Google Scholar]
- 12.Oh TJ, Moon JY, Hur KY, et al. Sodium-glucose cotransporter-2 inhibitor for renal function preservation in patients with type 2 diabetes mellitus: a Korean Diabetes Association and Korean Society of Nephrology consensus statement. Kidney Res Clin Pract. 2020;39:269–283. doi: 10.23876/j.krcp.20.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hur KY, Moon MK, Park JS, et al. 2021 Clinical practice guidelines for diabetes mellitus of the Korean Diabetes Association. Diabetes Metab J. 2021;45:461–481. doi: 10.4093/dmj.2021.0156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.American Diabetes Association 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes-2021. Diabetes Care. 2021;44(Suppl 1):S111–S124. doi: 10.2337/dc21-S009. [DOI] [PubMed] [Google Scholar]
- 15.Elrick H, Stimmler L, Hlad CJ, Jr, Arai Y. Plasma insulin response to oral and intravenous glucose administration. J Clin Endocrinol Metab. 1964;24:1076–1082. doi: 10.1210/jcem-24-10-1076. [DOI] [PubMed] [Google Scholar]
- 16.Eissele R, Göke R, Willemer S, et al. Glucagon-like peptide-1 cells in the gastrointestinal tract and pancreas of rat, pig and man. Eur J Clin Invest. 1992;22:283–291. doi: 10.1111/j.1365-2362.1992.tb01464.x. [DOI] [PubMed] [Google Scholar]
- 17.Orskov C, Wettergren A, Holst JJ. Secretion of the incretin hormones glucagon-like peptide-1 and gastric inhibitory polypeptide correlates with insulin secretion in normal man throughout the day. Scand J Gastroenterol. 1996;31:665–670. doi: 10.3109/00365529609009147. [DOI] [PubMed] [Google Scholar]
- 18.Kuhre RE, Wewer Albrechtsen NJ, Hartmann B, Deacon CF, Holst JJ. Measurement of the incretin hormones: glucagon-like peptide-1 and glucose-dependent insulinotropic peptide. J Diabetes Complications. 2015;29:445–450. doi: 10.1016/j.jdiacomp.2014.12.006. [DOI] [PubMed] [Google Scholar]
- 19.Rask E, Olsson T, Söderberg S, et al. Impaired incretin response after a mixed meal is associated with insulin resistance in nondiabetic men. Diabetes Care. 2001;24:1640–1645. doi: 10.2337/diacare.24.9.1640. [DOI] [PubMed] [Google Scholar]
- 20.Elliott RM, Morgan LM, Tredger JA, Deacon S, Wright J, Marks V. Glucagon-like peptide-1 (7-36)amide and glucose-dependent insulinotropic polypeptide secretion in response to nutrient ingestion in man: acute post-prandial and 24-h secretion patterns. J Endocrinol. 1993;138:159–166. doi: 10.1677/joe.0.1380159. [DOI] [PubMed] [Google Scholar]
- 21.Roberge JN, Brubaker PL. Regulation of intestinal proglucagon-derived peptide secretion by glucose-dependent insulinotropic peptide in a novel enteroendocrine loop. Endocrinology. 1993;133:233–240. doi: 10.1210/endo.133.1.8319572. [DOI] [PubMed] [Google Scholar]
- 22.Lim GE, Brubaker PL. Glucagon-like peptide 1 secretion by the L-cell: the view from within. Diabetes. 2006;55(Suppl_2):S70–S77. [Google Scholar]
- 23.Plaisancie P, Bernard C, Chayvialle JA, Cuber JC. Regulation of glucagon-like peptide-1-(7-36) amide secretion by intestinal neurotransmitters and hormones in the isolated vascularly perfused rat colon. Endocrinology. 1994;135:2398–2403. doi: 10.1210/endo.135.6.7988423. [DOI] [PubMed] [Google Scholar]
- 24.Meier JJ, Nauck MA, Kranz D, et al. Secretion, degradation, and elimination of glucagon-like peptide 1 and gastric inhibitory polypeptide in patients with chronic renal insufficiency and healthy control subjects. Diabetes. 2004;53:654–662. doi: 10.2337/diabetes.53.3.654. [DOI] [PubMed] [Google Scholar]
- 25.Deacon CF, Johnsen AH, Holst JJ. Degradation of glucagon-like peptide-1 by human plasma in vitro yields an N-terminally truncated peptide that is a major endogenous metabolite in vivo. J Clin Endocrinol Metab. 1995;80:952–957. doi: 10.1210/jcem.80.3.7883856. [DOI] [PubMed] [Google Scholar]
- 26.Kieffer TJ, McIntosh CH, Pederson RA. Degradation of glucose-dependent insulinotropic polypeptide and truncated glucagon-like peptide 1 in vitro and in vivo by dipeptidyl peptidase IV. Endocrinology. 1995;136:3585–3596. doi: 10.1210/endo.136.8.7628397. [DOI] [PubMed] [Google Scholar]
- 27.Wei Y, Mojsov S. Tissue-specific expression of the human receptor for glucagon-like peptide-I: brain, heart and pancreatic forms have the same deduced amino acid sequences. FEBS Lett. 1995;358:219–224. doi: 10.1016/0014-5793(94)01430-9. [DOI] [PubMed] [Google Scholar]
- 28.Doyle ME, Egan JM. Mechanisms of action of glucagon-like peptide 1 in the pancreas. Pharmacol Ther. 2007;113:546–593. doi: 10.1016/j.pharmthera.2006.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang Y, Egan JM, Raygada M, Nadiv O, Roth J, Montrose-Rafizadeh C. Glucagon-like peptide-1 affects gene transcription and messenger ribonucleic acid stability of components of the insulin secretory system in RIN 1046-38 cells. Endocrinology. 1995;136:4910–4917. doi: 10.1210/endo.136.11.7588224. [DOI] [PubMed] [Google Scholar]
- 30.Buteau J. GLP-1 receptor signaling: effects on pancreatic beta-cell proliferation and survival. Diabetes Metab. 2008;34 Suppl 2:S73–S77. doi: 10.1016/S1262-3636(08)73398-6. [DOI] [PubMed] [Google Scholar]
- 31.Li Y, Hansotia T, Yusta B, Ris F, Halban PA, Drucker DJ. Glucagon-like peptide-1 receptor signaling modulates beta cell apoptosis. J Biol Chem. 2003;278:471–478. doi: 10.1074/jbc.M209423200. [DOI] [PubMed] [Google Scholar]
- 32.Hare KJ, Knop FK, Asmar M, et al. Preserved inhibitory potency of GLP-1 on glucagon secretion in type 2 diabetes mellitus. J Clin Endocrinol Metab. 2009;94:4679–4687. doi: 10.1210/jc.2009-0921. [DOI] [PubMed] [Google Scholar]
- 33.Little TJ, Pilichiewicz AN, Russo A, et al. Effects of intravenous glucagon-like peptide-1 on gastric emptying and intragastric distribution in healthy subjects: relationships with postprandial glycemic and insulinemic responses. J Clin Endocrinol Metab. 2006;91:1916–1923. doi: 10.1210/jc.2005-2220. [DOI] [PubMed] [Google Scholar]
- 34.Toft-Nielsen MB, Madsbad S, Holst JJ. Continuous subcutaneous infusion of glucagon-like peptide 1 lowers plasma glucose and reduces appetite in type 2 diabetic patients. Diabetes Care. 1999;22:1137–1143. doi: 10.2337/diacare.22.7.1137. [DOI] [PubMed] [Google Scholar]
- 35.Asmar A, Cramon PK, Simonsen L, et al. Extracellular fluid volume expansion uncovers a natriuretic action of GLP-1: a functional GLP-1-renal axis in man. J Clin Endocrinol Metab. 2019;104:2509–2519. doi: 10.1210/jc.2019-00004. [DOI] [PubMed] [Google Scholar]
- 36.Crajoinas RO, Oricchio FT, Pessoa TD, et al. Mechanisms mediating the diuretic and natriuretic actions of the incretin hormone glucagon-like peptide-1. Am J Physiol Renal Physiol. 2011;301:F355–F363. doi: 10.1152/ajprenal.00729.2010. [DOI] [PubMed] [Google Scholar]
- 37.Trujillo JM, Nuffer W. GLP-1 receptor agonists for type 2 diabetes mellitus: recent developments and emerging agents. Pharmacotherapy. 2014;34:1174–1186. doi: 10.1002/phar.1507. [DOI] [PubMed] [Google Scholar]
- 38.Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet. 2006;368:1696–1705. doi: 10.1016/S0140-6736(06)69705-5. [DOI] [PubMed] [Google Scholar]
- 39.Meier JJ. GLP-1 receptor agonists for individualized treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2012;8:728–742. doi: 10.1038/nrendo.2012.140. [DOI] [PubMed] [Google Scholar]
- 40.Neumiller JJ. Incretin pharmacology: a review of the incretin effect and current incretin-based therapies. Cardiovasc Hematol Agents Med Chem. 2012;10:276–288. doi: 10.2174/187152512803530379. [DOI] [PubMed] [Google Scholar]
- 41.Jespersen MJ, Knop FK, Christensen M. GLP-1 agonists for type 2 diabetes: pharmacokinetic and toxicological considerations. Expert Opin Drug Metab Toxicol. 2013;9:17–29. doi: 10.1517/17425255.2013.731394. [DOI] [PubMed] [Google Scholar]
- 42.Pfeffer MA, Claggett B, Diaz R, et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med. 2015;373:2247–2257. doi: 10.1056/NEJMoa1509225. [DOI] [PubMed] [Google Scholar]
- 43.Muskiet MH, Tonneijck L, Huang Y, et al. Lixisenatide and renal outcomes in patients with type 2 diabetes and acute coronary syndrome: an exploratory analysis of the ELIXA randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 2018;6:859–869. doi: 10.1016/S2213-8587(18)30268-7. [DOI] [PubMed] [Google Scholar]
- 44.Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311–322. doi: 10.1056/NEJMoa1603827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mann JF, Ørsted DD, Brown-Frandsen K, et al. Liraglutide and renal outcomes in type 2 diabetes. N Engl J Med. 2017;377:839–848. doi: 10.1056/NEJMoa1616011. [DOI] [PubMed] [Google Scholar]
- 46.Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834–1844. doi: 10.1056/NEJMoa1607141. [DOI] [PubMed] [Google Scholar]
- 47.Mann JF, Hansen T, Idorn T, et al. Effects of once-weekly subcutaneous semaglutide on kidney function and safety in patients with type 2 diabetes: a post-hoc analysis of the SUSTAIN 1-7 randomised controlled trials. Lancet Diabetes Endocrinol. 2020;8:880–893. doi: 10.1016/S2213-8587(20)30313-2. [DOI] [PubMed] [Google Scholar]
- 48.Holman RR, Bethel MA, Mentz RJ, et al. Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2017;377:1228–1239. doi: 10.1056/NEJMoa1612917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bethel MA, Mentz RJ, Merrill P, et al. Microvascular and cardiovascular outcomes according to renal function in patients treated with once-weekly exenatide: insights from the EXSCEL Trial. Diabetes Care. 2020;43:446–452. doi: 10.2337/dc19-1065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Muskiet MH, Bunck MC, Heine RJ, et al. Exenatide twice-daily does not affect renal function or albuminuria compared to titrated insulin glargine in patients with type 2 diabetes mellitus: a post-hoc analysis of a 52-week randomized trial. Diabetes Res Clin Pract. 2019;153:14–22. doi: 10.1016/j.diabres.2019.05.001. [DOI] [PubMed] [Google Scholar]
- 51.Hernandez AF, Green JB, Janmohamed S, et al. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (Harmony Outcomes): a double-blind, randomised placebo-controlled trial. Lancet. 2018;392:1519–1529. doi: 10.1016/S0140-6736(18)32261-X. [DOI] [PubMed] [Google Scholar]
- 52.Tuttle KR, Lakshmanan MC, Rayner B, et al. Dulaglutide versus insulin glargine in patients with type 2 diabetes and moderate-to-severe chronic kidney disease (AWARD-7): a multicentre, open-label, randomised trial. Lancet Diabetes Endocrinol. 2018;6:605–617. doi: 10.1016/S2213-8587(18)30104-9. [DOI] [PubMed] [Google Scholar]
- 53.Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and renal outcomes in type 2 diabetes: an exploratory analysis of the REWIND randomised, placebo-controlled trial. Lancet. 2019;394:131–138. doi: 10.1016/S0140-6736(19)31150-X. [DOI] [PubMed] [Google Scholar]
- 54.Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomized placebo-controlled trial. Lancet. 2019;394:121–130. doi: 10.1016/S0140-6736(19)31149-3. [DOI] [PubMed] [Google Scholar]
- 55.Husain M, Birkenfeld AL, Donsmark M, et al. Oral Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2019;381:841–851. doi: 10.1056/NEJMoa1901118. [DOI] [PubMed] [Google Scholar]
- 56.Gerstein HC, Sattar N, Rosenstock J, et al. Cardiovascular and renal outcomes with efpeglenatide in type 2 diabetes. N Engl J Med. 2021;385:896–907. doi: 10.1056/NEJMoa2108269. [DOI] [PubMed] [Google Scholar]
- 57.Coca SG, Ismail-Beigi F, Haq N, Krumholz HM, Parikh CR. Role of intensive glucose control in development of renal end points in type 2 diabetes mellitus: systematic review and meta-analysis intensive glucose control in type 2 diabetes. Arch Intern Med. 2012;172:761–769. doi: 10.1001/archinternmed.2011.2230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Tuttle KR, Bruton JL, Perusek MC, Lancaster JL, Kopp DT, DeFronzo RA. Effect of strict glycemic control on renal hemodynamic response to amino acids and renal enlargement in insulin-dependent diabetes mellitus. N Engl J Med. 1991;324:1626–1632. doi: 10.1056/NEJM199106063242304. [DOI] [PubMed] [Google Scholar]
- 59.Blonde L, Jendle J, Gross J, et al. Once-weekly dulaglutide versus bedtime insulin glargine, both in combination with prandial insulin lispro, in patients with type 2 diabetes (AWARD-4): a randomised, open-label, phase 3, non-inferiority study. Lancet. 2015;385:2057–2066. doi: 10.1016/S0140-6736(15)60936-9. [DOI] [PubMed] [Google Scholar]
- 60.Mosenzon O, Blicher TM, Rosenlund S, et al. Efficacy and safety of oral semaglutide in patients with type 2 diabetes and moderate renal impairment (PIONEER 5): a placebo-controlled, randomised, phase 3a trial. Lancet Diabetes Endocrinol. 2019;7:515–527. doi: 10.1016/S2213-8587(19)30192-5. [DOI] [PubMed] [Google Scholar]
- 61.Lingvay I, Catarig AM, Frias JP, et al. Efficacy and safety of once-weekly semaglutide versus daily canagliflozin as add-on to metformin in patients with type 2 diabetes (SUSTAIN 8): a double-blind, phase 3b, randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7:834–844. doi: 10.1016/S2213-8587(19)30311-0. [DOI] [PubMed] [Google Scholar]
- 62.Nauck M, Rizzo M, Johnson A, Bosch-Traberg H, Madsen J, Cariou B. Once-daily liraglutide versus lixisenatide as add-on to metformin in type 2 diabetes: a 26-week randomized controlled clinical trial. Diabetes Care. 2016;39:1501–1509. doi: 10.2337/dc15-2479. [DOI] [PubMed] [Google Scholar]
- 63.Kidney Disease: Improving Global Outcomes (KDIGO) Diabetes Work Group KDIGO 2020 clinical practice guideline for diabetes management in chronic kidney disease. Kidney Int. 2020;98(4S):S1–S115. doi: 10.1016/j.kint.2020.06.019. [DOI] [PubMed] [Google Scholar]
- 64.Vitale M, Haxhi J, Cirrito T, Pugliese G. Renal protection with glucagon-like peptide-1 receptor agonists. Curr Opin Pharmacol. 2020;54:91–101. doi: 10.1016/j.coph.2020.08.018. [DOI] [PubMed] [Google Scholar]
- 65.Dalsgaard NB, Vilsbøll T, Knop FK. Effects of glucagon-like peptide-1 receptor agonists on cardiovascular risk factors: a narrative review of head-to-head comparisons. Diabetes Obes Metab. 2018;20:508–519. doi: 10.1111/dom.13128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Mosterd CM, Bjornstad P, van Raalte DH. Nephroprotective effects of GLP-1 receptor agonists: where do we stand? J Nephrol. 2020;33:965–975. doi: 10.1007/s40620-020-00738-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kamanna VS, Roh DD, Kirschenbaum MA. Hyperlipidemia and kidney disease: concepts derived from histopathology and cell biology of the glomerulus. Histol Histopathol. 1998;13:169–179. doi: 10.14670/HH-13.169. [DOI] [PubMed] [Google Scholar]
- 68.Sun F, Wu S, Wang J, et al. Effect of glucagon-like peptide-1 receptor agonists on lipid profiles among type 2 diabetes: a systematic review and network meta-analysis. Clin Ther. 2015;37:225–241. doi: 10.1016/j.clinthera.2014.11.008. [DOI] [PubMed] [Google Scholar]
- 69.Wøjdemann M, Wettergren A, Sternby B, et al. Inhibition of human gastric lipase secretion by glucagon-like peptide-1. Dig Dis Sci. 1998;43:799–805. doi: 10.1023/a:1018874300026. [DOI] [PubMed] [Google Scholar]
- 70.Xiao C, Bandsma RH, Dash S, Szeto L, Lewis GF. Exenatide, a glucagon-like peptide-1 receptor agonist, acutely inhibits intestinal lipoprotein production in healthy humans. Arterioscler Thromb Vasc Biol. 2012;32:1513–1519. doi: 10.1161/ATVBAHA.112.246207. [DOI] [PubMed] [Google Scholar]
- 71.Ben-Shlomo S, Zvibel I, Shnell M, et al. Glucagon-like peptide-1 reduces hepatic lipogenesis via activation of AMP-activated protein kinase. J Hepatol. 2011;54:1214–1223. doi: 10.1016/j.jhep.2010.09.032. [DOI] [PubMed] [Google Scholar]
- 72.Ding X, Saxena NK, Lin S, Gupta NA, Anania FA. Exendin-4, a glucagon-like protein-1 (GLP-1) receptor agonist, reverses hepatic steatosis in ob/ob mice. Hepatology. 2006;43:173–181. doi: 10.1002/hep.21006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kooijman S, Wang Y, Parlevliet ET, et al. Central GLP-1 receptor signalling accelerates plasma clearance of triacylglycerol and glucose by activating brown adipose tissue in mice. Diabetologia. 2015;58:2637–2646. doi: 10.1007/s00125-015-3727-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Beiroa D, Imbernon M, Gallego R, et al. GLP-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic AMPK. Diabetes. 2014;63:3346–3358. doi: 10.2337/db14-0302. [DOI] [PubMed] [Google Scholar]
- 75.Pyke C, Heller RS, Kirk RK, et al. GLP-1 receptor localization in monkey and human tissue: novel distribution revealed with extensively validated monoclonal antibody. Endocrinology. 2014;155:1280–1290. doi: 10.1210/en.2013-1934. [DOI] [PubMed] [Google Scholar]
- 76.Schlatter P, Beglinger C, Drewe J, Gutmann H. Glucagon-like peptide 1 receptor expression in primary porcine proximal tubular cells. Regul Pept. 2007;141:120–128. doi: 10.1016/j.regpep.2006.12.016. [DOI] [PubMed] [Google Scholar]
- 77.Fujita H, Sakamoto T, Komatsu K, et al. Reduction of circulating superoxide dismutase activity in type 2 diabetic patients with microalbuminuria and its modulation by telmisartan therapy. Hypertens Res. 2011;34:1302–1308. doi: 10.1038/hr.2011.127. [DOI] [PubMed] [Google Scholar]
- 78.Yin W, Jiang Y, Xu S, et al. Protein kinase C and protein kinase A are involved in the protection of recombinant human glucagon-like peptide-1 on glomeruli and tubules in diabetic rats. J Diabetes Investig. 2019;10:613–625. doi: 10.1111/jdi.12956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hendarto H, Inoguchi T, Maeda Y, et al. GLP-1 analog liraglutide protects against oxidative stress and albuminuria in streptozotocin-induced diabetic rats via protein kinase A-mediated inhibition of renal NAD(P)H oxidases. Metabolism. 2012;61:1422–1434. doi: 10.1016/j.metabol.2012.03.002. [DOI] [PubMed] [Google Scholar]
- 80.Wang C, Li C, Peng H, et al. Activation of the Nrf2-ARE pathway attenuates hyperglycemia-mediated injuries in mouse podocytes. Cell Physiol Biochem. 2014;34:891–902. doi: 10.1159/000366307. [DOI] [PubMed] [Google Scholar]
- 81.Zhou T, Zhang M, Zhao L, Li A, Qin X. Activation of Nrf2 contributes to the protective effect of Exendin-4 against angiotensin II-induced vascular smooth muscle cell senescence. Am J Physiol Cell Physiol. 2016;311:C572–C582. doi: 10.1152/ajpcell.00093.2016. [DOI] [PubMed] [Google Scholar]
- 82.Ye Y, Zhong X, Li N, Pan T. Protective effects of liraglutide on glomerular podocytes in obese mice by inhibiting the inflammatory factor TNF-α-mediated NF-κB and MAPK pathway. Obes Res Clin Pract. 2019;13:385–390. doi: 10.1016/j.orcp.2019.03.003. [DOI] [PubMed] [Google Scholar]
- 83.Kodera R, Shikata K, Kataoka HU, et al. Glucagon-like peptide-1 receptor agonist ameliorates renal injury through its anti-inflammatory action without lowering blood glucose level in a rat model of type 1 diabetes. Diabetologia. 2011;54:965–978. doi: 10.1007/s00125-010-2028-x. [DOI] [PubMed] [Google Scholar]
- 84.Chaudhuri A, Ghanim H, Vora M, et al. Exenatide exerts a potent antiinflammatory effect. J Clin Endocrinol Metab. 2012;97:198–207. doi: 10.1210/jc.2011-1508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bunck MC, Diamant M, Eliasson B, et al. Exenatide affects circulating cardiovascular risk biomarkers independently of changes in body composition. Diabetes Care. 2010;33:1734–1737. doi: 10.2337/dc09-2361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Zhang H, Zhang X, Hu C, Lu W. Exenatide reduces urinary transforming growth factor-β1 and type IV collagen excretion in patients with type 2 diabetes and microalbuminuria. Kidney Blood Press Res. 2012;35:483–488. doi: 10.1159/000337929. [DOI] [PubMed] [Google Scholar]
- 87.Hogan AE, Gaoatswe G, Lynch L, et al. Glucagon-like peptide 1 analogue therapy directly modulates innate immune-mediated inflammation in individuals with type 2 diabetes mellitus. Diabetologia. 2014;57:781–784. doi: 10.1007/s00125-013-3145-0. [DOI] [PubMed] [Google Scholar]
- 88.Rizzo M, Abate N, Chandalia M, et al. Liraglutide reduces oxidative stress and restores heme oxygenase-1 and ghrelin levels in patients with type 2 diabetes: a prospective pilot study. J Clin Endocrinol Metab. 2015;100:603–606. doi: 10.1210/jc.2014-2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Yip KP, Tse CM, McDonough AA, Marsh DJ. Redistribution of Na+/H+ exchanger isoform NHE3 in proximal tubules induced by acute and chronic hypertension. Am J Physiol. 1998;275:F565–F575. doi: 10.1152/ajprenal.1998.275.4.F565. [DOI] [PubMed] [Google Scholar]
- 90.Ronn J, Jensen EP, Wewer Albrechtsen NJ, Holst JJ, Sorensen CM. Glucagon-like peptide-1 acutely affects renal blood flow and urinary flow rate in spontaneously hypertensive rats despite significantly reduced renal expression of GLP-1 receptors. Physiol Rep. 2017;5:e13503. doi: 10.14814/phy2.13503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Skov J, Dejgaard A, Frøkiær J, et al. Glucagon-like peptide-1 (GLP-1): effect on kidney hemodynamics and renin-angiotensin-aldosterone system in healthy men. J Clin Endocrinol Metab. 2013;98:E664–E671. doi: 10.1210/jc.2012-3855. [DOI] [PubMed] [Google Scholar]
- 92.Skov J, Pedersen M, Holst JJ, et al. Short-term effects of liraglutide on kidney function and vasoactive hormones in type 2 diabetes: a randomized clinical trial. Diabetes Obes Metab. 2016;18:581–589. doi: 10.1111/dom.12651. [DOI] [PubMed] [Google Scholar]
- 93.von Scholten BJ, Hansen TW, Goetze JP, Persson F, Rossing P. Glucagon-like peptide 1 receptor agonist (GLP-1 RA): long-term effect on kidney function in patients with type 2 diabetes. J Diabetes Complications. 2015;29:670–674. doi: 10.1016/j.jdiacomp.2015.04.004. [DOI] [PubMed] [Google Scholar]
- 94.Pocai A, Carrington PE, Adams JR, et al. Glucagon-like peptide 1/glucagon receptor dual agonism reverses obesity in mice. Diabetes. 2009;58:2258–2266. doi: 10.2337/db09-0278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Capozzi ME, Svendsen B, Encisco SE, et al. β Cell tone is defined by proglucagon peptides through cAMP signaling. JCI Insight. 2019;4:e126742. doi: 10.1172/jci.insight.126742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Frias JP, Nauck MA, Van J, et al. Efficacy and safety of LY3298176, a novel dual GIP and GLP-1 receptor agonist, in patients with type 2 diabetes: a randomised, placebo-controlled and active comparator-controlled phase 2 trial. Lancet. 2018;392:2180–2193. doi: 10.1016/S0140-6736(18)32260-8. [DOI] [PubMed] [Google Scholar]
- 97.Madsen KB, Askov-Hansen C, Naimi RM, et al. Acute effects of continuous infusions of glucagon-like peptide (GLP)-1, GLP-2 and the combination (GLP-1+GLP-2) on intestinal absorption in short bowel syndrome (SBS) patients: a placebo-controlled study. Regul Pept. 2013;184:30–39. doi: 10.1016/j.regpep.2013.03.025. [DOI] [PubMed] [Google Scholar]
- 98.Müller TD, Sullivan LM, Habegger K, et al. Restoration of leptin responsiveness in diet-induced obese mice using an optimized leptin analog in combination with exendin-4 or FGF21. J Pept Sci. 2012;18:383–393. doi: 10.1002/psc.2408. [DOI] [PubMed] [Google Scholar]
- 99.Suarez-Pinzon WL, Power RF, Yan Y, Wasserfall C, Atkinson M, Rabinovitch A. Combination therapy with glucagon-like peptide-1 and gastrin restores normoglycemia in diabetic NOD mice. Diabetes. 2008;57:3281–3288. doi: 10.2337/db08-0688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Trevaskis JL, Mack CM, Sun C, et al. Improved glucose control and reduced body weight in rodents with dual mechanism of action peptide hybrids. PLoS One. 2013;8:e78154. doi: 10.1371/journal.pone.0078154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Neary NM, Small CJ, Druce MR, et al. Peptide YY3-36 and glucagon-like peptide-17-36 inhibit food intake additively. Endocrinology. 2005;146:5120–5127. doi: 10.1210/en.2005-0237. [DOI] [PubMed] [Google Scholar]
- 102.Gutzwiller JP, Degen L, Matzinger D, Prestin S, Beglinger C. Interaction between GLP-1 and CCK-33 in inhibiting food intake and appetite in men. Am J Physiol Regul Integr Comp Physiol. 2004;287:R562–R567. doi: 10.1152/ajpregu.00599.2003. [DOI] [PubMed] [Google Scholar]
- 103.Balena R, Hensley IE, Miller S, Barnett AH. Combination therapy with GLP-1 receptor agonists and basal insulin: a systematic review of the literature. Diabetes Obes Metab. 2013;15:485–502. doi: 10.1111/dom.12025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Bech EM, Voldum-Clausen K, Pedersen SL, et al. Adrenomedullin and glucagon-like peptide-1 have additive effects on food intake in mice. Biomed Pharmacother. 2019;109:167–173. doi: 10.1016/j.biopha.2018.10.040. [DOI] [PubMed] [Google Scholar]
- 105.Pan Q, Lin S, Li Y, et al. A novel GLP-1 and FGF21 dual agonist has therapeutic potential for diabetes and non-alcoholic steatohepatitis. EBioMedicine. 2021;63:103202. doi: 10.1016/j.ebiom.2020.103202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Finan B, Yang B, Ottaway N, et al. Targeted estrogen delivery reverses the metabolic syndrome. Nat Med. 2012;18:1847–1856. doi: 10.1038/nm.3009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Quarta C, Clemmensen C, Zhu Z, et al. Molecular integration of incretin and glucocorticoid action reverses immunometabolic dysfunction and obesity. Cell Metab. 2017;26:620–632. doi: 10.1016/j.cmet.2017.08.023. [DOI] [PubMed] [Google Scholar]
- 108.Chodorge M, Celeste AJ, Grimsby J, et al. Engineering of a GLP-1 analogue peptide/anti-PCSK9 antibody fusion for type 2 diabetes treatment. Sci Rep. 2018;8:17545. doi: 10.1038/s41598-018-35869-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Clemmensen C, Finan B, Fischer K, et al. Dual melanocortin-4 receptor and GLP-1 receptor agonism amplifies metabolic benefits in diet-induced obese mice. EMBO Mol Med. 2015;7:288–298. doi: 10.15252/emmm.201404508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Jouihan H, Will S, Guionaud S, et al. Superior reductions in hepatic steatosis and fibrosis with co-administration of a glucagon-like peptide-1 receptor agonist and obeticholic acid in mice. Mol Metab. 2017;6:1360–1370. doi: 10.1016/j.molmet.2017.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Frías JP, Guja C, Hardy E, et al. Exenatide once weekly plus dapagliflozin once daily versus exenatide or dapagliflozin alone in patients with type 2 diabetes inadequately controlled with metformin monotherapy (DURATION-8): a 28 week, multicentre, double-blind, phase 3, randomised controlled trial. Lancet Diabetes Endocrinol. 2016;4:1004–1016. doi: 10.1016/S2213-8587(16)30267-4. [DOI] [PubMed] [Google Scholar]


