Abstract
Bronchiectasis is a debilitating chronic suppurative airway disease that confers a substantial burden globally. Despite the notable prevalence, research on bronchiectasis in mainland China remains in its infancy. Nevertheless, there has been a significant leap in the quantity and quality of research, which has contributed to the ever-improving clinical practice. A nationwide collaborative platform has been established to foster multicentre studies, which will help increase the level of evidence further. Here, we summarise the status quo of clinical management and consider the research priorities for bronchiectasis that have been published previously. We also highlight the efforts of the Chinese medical communities to outline the core tasks that need to be addressed within the next decade.
Short abstract
The remarkable progress made in bronchiectasis research has revolutionised clinical practice in mainland China https://bit.ly/3K8kQYr
Introduction
Bronchiectasis is defined as the pathological, and often irreversible, dilatation of the tracheobronchial tree that is due to recurrent airway infections and inflammation [1]. The earliest but incomplete documentation of bronchiectasis has been made by the ancient Chinese people, who had noticed patients producing a massive volume of sputum contained in a spittoon. It took a long time before the clinical impact of bronchiectasis was fully documented.
Bronchiectasis has traditionally been regarded as an orphan lung disease. In China, however, this was about to change when studies from Hong Kong and the first national epidemiological report [2–4] were published. These important publications aroused the interest of researchers in the exploration of bronchiectasis, improving the limited understanding about the disease. Additionally, a representative review that summarised the status quo of clinical management and research priorities for bronchiectasis has been published previously [5]. Despite the rapid development in bronchiectasis research, there are still considerable challenges and limitations that need to be resolved in China and other countries. Here, we focus on the latest progress in bronchiectasis research in China, which is relevant to the epidemiology, clinical diagnosis, pathogenesis and therapeutic management. Finally, we highlight the efforts made by the Chinese medical communities (including the establishment of the national bronchiectasis registry), the major opportunities and challenges for future research, and the implications for global clinical practice.
Epidemiology of bronchiectasis
Like COPD and asthma, bronchiectasis is a common chronic airway disease and has become an enormous social and economic burden owing to the high rates of hospital admission and mortality globally [2, 6–13]. However, the true incidence and prevalence remain poorly documented in China. An urban population-based cross-sectional survey from 2002 to 2004 [2] showed that the prevalence of physician-diagnosed bronchiectasis among individuals aged ≥40 years in China was 1200 per 100 000 persons, and it increased markedly with age. As the reported percentage included only those individuals diagnosed with bronchiectasis, the true prevalence remains to be further elucidated. Currently, bronchiectasis is diagnosed based on both clinical and radiological grounds [14]. The marked heterogeneity in the geographical regions, time periods and diagnostic criteria (codes only versus chest imaging), as well as study designs and settings [6–13], has meant that the incidence and prevalence estimates of different studies vary significantly (67–566 per 100 000 inhabitants, table 1).
TABLE 1.
Incidence and prevalence of bronchiectasis worldwide
| First author (ref.) | Methods | Country | Period of time | Incidence | Prevalence |
| Weycker et al. [6] | ICD (secondary care) | USA | 2013 | 34 and 23 per 100 000 person-years among women and men, respectively | 180/100 000 in women; 95/100 000 in men |
| Ringshausen et al. [7] and Diel et al. [8] | ICD (secondary care) | Germany | 2013 | 21.23 per 100 000 person-years | 67/100 000 persons Men aged 75–84 years: 228/100 000 |
| Quint et al. [9] | Read (primary care) | UK | 2013 | 35.2 and 26.9 per 100 000 person-years among women and men, respectively | 566.1/100 000 in women; 485.5/100 000 in men |
| Monteagudo et al. [10] | ICD (primary care) | Catalonia, Spain | 2012 | 49.3 and 46.9 per 100 000 person-years among women and men, respectively | 391/100 000 in women; 333/100 000 in men |
| Aliberti et al. [11] | ICD (primary care) | Italy | 2015 | 18.2 and 14.1 per 100 000 person-years among women and men, respectively | 178/100 000 in women; 147/100 000 in men |
| Zhou et al. [2] | Self-reported physician-diagnosed bronchiectasis | China | 2002–2004 | NA | 1100/100 000 and 1500/100 000 in women and men aged 40 years or older, respectively |
| Phua et al. [12] | ICD (primary care) | Singapore | 2017 | 11.2 and 9.9 per 100 000 person-years among women and men, respectively | 140.0/100 000 in women; 154.5/100 000 in men |
| Choi et al. [13] | ICD | South Korea | 2012 | NA | 464/100 000 in people aged 20 years or older |
ICD: International Coding of Disease; NA: not applicable.
Recently, most studies from Western countries have shown an increase in both the prevalence and incidence of bronchiectasis [6–11, 13], especially in the elderly. Given the heterogeneity of clinical manifestations and aetiologies, international registries, such as European Multicenter Bronchiectasis Audit and Research Collaboration (EMBARC) [15], the United States Bronchiectasis Research Registry (US registry) [16] and Bronchiectasis-China (clinical trial registry: NCT036436530), are warranted. These registries have provided valuable databases from which to study patients with bronchiectasis.
Advances of pathophysiology
Despite the fact that bronchiectasis has received increasing focus in the past few years, both the substantial heterogeneity between studies and lack of animal experimental models have contributed to the poor understanding of pathogenesis.
Currently, the most widely known model of the development of bronchiectasis is the “vicious cycle” hypothesis, which involves airway infection, inflammation, impaired mucociliary clearance and structural damage associated with the diverse aetiologies [17]. The theory posits that the disruption of any of these elements would result in progression of the disease. However, antibiotics and anti-inflammatory or airway clearance therapies used alone have demonstrated only modest effects on clinical outcomes in bronchiectasis [18–20]. Recently, Flume et al. [21] proposed a new concept called “vicious vortex” and suggested that the interactions between each pathophysiological step are far more complex. The core concept of this updated theory was that the individual elements were interrelated and that no uniform sequence of events would apply, suggesting that bronchiectasis resulted from the complicated interactions of the different key elements. Hence, targeting only one component of the vortex is probably insufficient to fully break the “vicious vortex” and halt the disease progression.
Scientists have made efforts to understand the vicious vortex. Airway infection with potentially pathogenic microorganisms (PPMs) is the main driver of progression in bronchiectasis. Several studies in China have shown that the most common PPM in patients with bronchiectasis when clinically stable was Pseudomonas aeruginosa, followed by Haemophilus parainfluenzae and H. influenzae [22]. Apart from the major factors for poor prognosis in bronchiectasis (e.g. being female, frequent exacerbations and cystic bronchiectasis) [23], P. aeruginosa was independently associated with an increased burden of disease including a higher frequency of exacerbations, worse health-related quality of life (QoL) and increased mortality [24]. Researchers have revealed that the virulence genes exoU and pldA in mucoid P. aeruginosa contributed to frequent exacerbations [25], but the isolation of antibiotic-resistant P. aeruginosa, which accounted for >50% of strains, was not associated with poor outcomes [26]. The advent of culture-independent techniques (e.g. 16srRNA sequencing) has helped characterise the respiratory microbiome. Consistent with previous studies [27], the Chinese bronchiectasis patients had a notably higher abundance of Proteobacteria and P. aeruginosa but a reduced diversity compared with healthy controls [28].
Nevertheless, the roles of viruses in bronchiectasis have been elusive. Compared with clinically stable state, viruses were more readily detected (∼50% versus 19%) at bronchiectasis exacerbations, providing the initial evidence that viruses might have triggered bronchiectasis exacerbations among adults [29]. Isolation of new bacterial strains, detection of any virus and these two combined were all associated with bronchiectasis exacerbations [30]. Epstein–Barr virus (EBV), another opportunistic virus, was commonly detected during the stable state compared with healthy controls. The detection of EBV DNA indicated a shorter time to the next exacerbation [31], suggesting that EBV might have contributed to bronchiectasis progression. Non-tuberculous mycobacteria (NTM) have also been commonly detected according to a national study (detection rate: 23.3%) [32], which showed that three or more abnormal features (nodular shadows, tree-in-bud signs, cavities) and bronchiectasis lesions predominantly located at the right middle or lingual lobe were risk factors associated with NTM pulmonary disease. With regard to fungal detection or infections, there remains a paucity of national studies and available data in China.
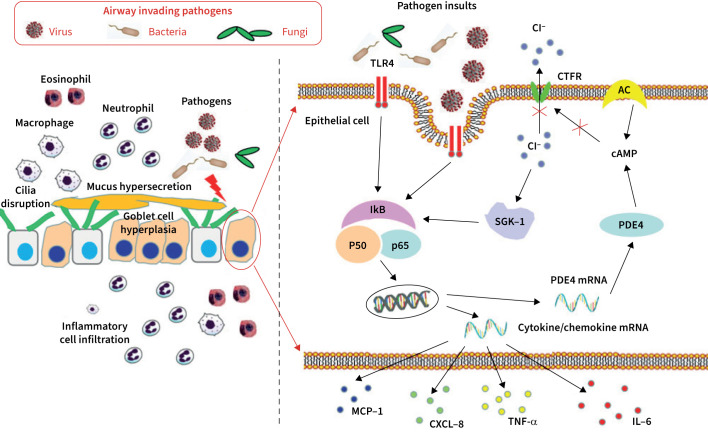
Although the pathogenesis of bronchiectasis is less well understood, the roles of components in the “vicious vortex” have been unveiled. Bronchiectasis patients had lower ciliary beat frequency, defects of central and peripheral microtubules, and lengthened cilia compared with healthy controls [33, 34]. A histopathological study revealed epithelial hyperplasia, including hyperplasia and/or hypertrophy of goblet cells in the bronchi and bronchioles [34]. Patients with bronchiectasis had prominent inflammatory cell (e.g. neutrophil, eosinophil) infiltration within airway epithelium [23, 34]. How airway inflammation becomes persistent has been an enigma for a long time. The observation that P. aeruginosa or lipopolysaccharide stimulation in airway epithelial cells could increase intracellular Cl− concentration via nuclear factor-κB–phosphodiesterase 4D–cAMP signalling pathways might address this issue and highlight the role of cystic fibrosis transmembrane regulator dysfunction as a potential target for therapeutic interventions [35]. These studies indicated the interactions among infection, inflammation, ciliary dysfunction and epithelial remodelling (figure 1).
FIGURE 1.
Novel insights into the pathogenesis of heightened inflammation in bronchiectasis. MCP-1: macrophage chemoattractant protein-1; IL-6: interleukin-6; CXCL-8: C-X-C motif chemokine ligand 8; TNF-α: tumor necrosis factor-α; IkB: nuclear factor κB inhibitor α; SGK-1: serum glucocorticoid kinase-1; PDE4: phosphodiesterase 4; TLR4: Toll-like receptor 4; CFTR: cystic fibrosis transmembrane regulator; AC: adenyl cyclase; cAMP: cyclic adenomonophosphate.
Advances in clinical diagnostics
Biomarker profiling
Despite the rapid development in modern medicine, no validated biomarker has been successfully applied in clinical practice for the diagnosis, exacerbation assessment or prognosis of bronchiectasis.
Neutrophil infiltration is predominant in bronchiectasis [36, 37]. Neutrophils secrete matrix metalloproteinase-8 and -9, which leads to airway destruction [38]. Interleukin (IL)-17 is an important pro-inflammatory cytokine, the expression of which is significantly elevated in serum, bronchoalveolar lavage fluid and lung tissues in patients with bronchiectasis [39].
Apart from activating the canonical nuclear factor-κB and mitogen-activated protein kinase/extracellular signal-regulated kinase 1/2 pathways, IL-17 plays a role in bronchiectasis-induced pulmonary hypertension [39]. The number as well as the migratory and proliferative activity of circulating endothelial progenitor cells in bronchiectasis was significantly reduced, which was inversely correlated with systemic inflammation [40]. Flow-mediated dilatation, as the indicator of vascular endothelial function, was significantly lower in bronchiectasis patients compared with healthy controls and was associated with the Bronchiectasis Severity Index and FACED score [41].
Imaging
High-resolution computed tomography (HRCT) of the chest is the “gold standard” for radiological diagnosis of bronchiectasis [42]. There are several scoring systems and objective criteria to evaluate the severity of chest HRCT in patients with bronchiectasis. A novel quantitative HRCT protocol has been developed to assess the extent of bronchiectasis, bronchial wall thickening, small-airway abnormalities and mosaic pattern. This protocol had a low intra-observer variation and high interobserver reliability. The extent of bronchiectasis, bronchial wall thickening and mosaic attenuation was inversely correlated with airway obstruction [43]. Besides, the Bhalla score may provide more insight compared with the modified Reiff score. A multicenter study showed that the type of bronchiectasis and Bhalla score correlated with the symptoms, lung function, frequency of exacerbations and hospitalisations [44].
Coexistence of bronchiectasis and COPD and/or asthma has been associated with worse prognosis. HRCT played a pivotal role in identifying this phenotype [45–47]. Bronchiectasis was detected with HRCT in 34.7% of COPD patients, with the isolation of P. aeruginosa being the major predictor [45]. In another study, COPD patients with bronchiectasis had poorer pulmonary function, higher emphysema index, and a higher proportion of pulmonary hypertension and cor pulmonale. Patients with coexistence of COPD and bronchiectasis had a higher pulmonary artery to aorta ratio, which correlated with worse nutritional status, higher erythrocyte sedimentation rate and fibrinogen levels [48].
In addition, artificial intelligence has introduced new algorithms based on machine learning for HRCT image analysis. Integration of artificial intelligence into the clinical practice is under development.
Pulmonary function
Because of structural changes, bronchiectasis patients may have different patterns of airflow limitation, including obstructive, restrictive or mixed lung function impairment. In bronchiectasis patients with airway obstruction, the bronchodilator response was negatively correlated with baseline forced expiratory volume in 1 s (FEV1) percentage predicted, but not blood or sputum eosinophil count [49]. Significant bronchodilator response correlated with a lower risk of exacerbations, but further exploration of the mechanisms involved is necessary.
Impulse oscillometry has been used to determine small-airway resistance. Greater abnormality of impulse oscillometric parameters was associated with a higher Bronchiectasis Severity Index, higher HRCT scores and more involved lobes [50]. Compared with spirometry, impulse oscillometric parameters (e.g. resonant frequency) could more readily distinguish patients with mild bronchiectasis. These results have shed light on applying lung function parameters to clinical assessment of bronchiectasis.
Lung clearance index may efficiently identify ventilation heterogeneity and is more sensitive than FEV1 in discriminating bronchiectasis patients from healthy subjects. Maximal mid-expiratory flow had a similar efficiency compared with lung clearance index in discriminating moderate-to-severe from mild bronchiectasis [51].
Unlike asthma, COPD and cystic fibrosis, the reduction in FEV1 and forced vital capacity was non-clinically significant (within 5% of baseline levels) at exacerbation onset and convalescence in bronchiectasis [52, 53]. There was no significant difference in impulse oscillometric parameters or lung clearance index at the onset of exacerbations [50–52]. Therefore, lung function changes might not accurately capture the onset of bronchiectasis exacerbations.
Non-pharmacological and pharmacological management
Despite major advances, evidence-based therapies for bronchiectasis are still in their infancy owing to the scarcity of large-scale randomised controlled trials (RCTs). The Chinese Thoracic Society published an updated expert consensus in 2021 [54]. Significant gaps regarding drug availability and implementation of airway clearance therapy existed between China and Western countries [1, 54, 55]. Since January 2020, a national registry platform has been initiated to improve bronchiectasis care in China to bridge these gaps (NCT03643653).
Airway clearance therapy
Impaired mucociliary clearance plays a crucial role in driving the progression of bronchiectasis. Airway clearance therapy is a cornerstone treatment both at steady-state and exacerbation that aims to promote mucus mobilisation and elimination. Airway clearance strategies have been developed into a range of physiotherapy techniques including assisted chest therapies/airway clearance techniques, specialised breathing techniques (e.g. “huff-cough” manoeuvre) and mechanical mobilisation.
Previous studies showed that airway clearance techniques led to short-term improvement in QoL, symptoms and sputum production compared with sham intervention or inactive controls [56]. However, there are limited available data regarding the benefits of specific technique. The European Respiratory Society (ERS) guideline endorsed an active cycle of breathing, autogenic drainage or oscillating positive expiratory pressure [55], which was suitable for domiciliary use. An RCT from Shanghai showed that airway clearance through bronchoscopy during exacerbation was safe and significantly prolonged the time to first exacerbation after discharge (198 versus 168 days), and it improved symptoms [57], which indicated bronchoscopy-assisted airway clearance could be an option for highly symptomatic patients at exacerbation.
In China, physiotherapy has not been widely accepted by both physicians and patients possibly due to a lack of awareness as well as resources and evidence, whereas mucoactive medications have been commonly prescribed. Among the various types of physiotherapy, chest percussion and gravity-assisted positioning drainage, with individualised frequency and duration, were the preferred techniques. However, in practice, airway clearance therapies have been mostly delivered by a respiratory physician rather than a trained respiratory physiotherapist. All these barriers hinder the implementation and affect the therapeutic efficacy of this backbone treatment advocated by the international bronchiectasis guidelines.
In addition, the ERS guideline recommended patients who have exertional limitation to exercise regularly and participate in pulmonary rehabilitation [55], which could markedly improve exercise tolerance and QoL [58]. However, the situation for pulmonary rehabilitation is similar to that of physiotherapy in China. Therefore, studies should be performed as soon as possible, and Chinese physicians should pay attention to these two therapies.
Mucoactive therapy
Mucoactive medications could be prescribed if the active cycle of breathing therapy becomes insufficient. Mucoactive drugs (e.g. hypertonic saline, mannitol, N-acetylcysteine, ambroxol, erdosteine, carbocisteine) could ease sputum expectoration and ameliorate mucus retention [1, 54, 55]. The 2017 ERS guideline summarised the findings of three systematic reviews of mucoactive drugs [55], none of which significantly reduced exacerbations. However, an RCT showed that 12 months of oral treatment with N-acetylcysteine reduced the risk of exacerbation and improved QoL compared with usual care [59].
Additionally, assessment of baseline symptom burden might help to evaluate therapeutic response. For instance, 12 months of inhaled treatment with mannitol (400 mg twice daily) prolonged the time to the next exacerbation but could not improve the rate of exacerbation compared with the control group [60]. A post hoc analysis showed that inhaled mannitol could reduce the rate of exacerbation among the highly symptomatic patients [61]. Recently, a multicentre RCT has been initiated to establish whether oral AnNingPai enteric soft capsules (300 mg, three times a day) could reduce exacerbation frequency and improve QoL in bronchiectasis patients with mucus hypersecretion (NCT04511897).
Bronchodilators and corticosteroids
Inhaled long-acting bronchodilators and corticosteroids are widely used in bronchiectasis, although the evidence base has been poor. Previous observational studies have shown that treatment with inhaled bronchodilators was effective in improving lung function in bronchiectasis patients with airflow limitation [62, 63], but the use of some inhalers (e.g. short-acting β-agonist and long-acting muscarinic antagonist) might be associated with increased risk of haemoptysis [64]. A recent RCT of tiotropium in stable bronchiectasis patients with airflow limitation over 6 months showed that tiotropium was associated with modest improvement in FEV1, but not exacerbation reduction or improvement in symptoms and QoL [65]. To add further evidence, an RCT of an inhaled long-acting dual bronchodilator (trial NCT04509661) in bronchiectasis with airflow limitation has been initiated.
Bronchiectasis has been regarded as a neutrophil-dominated inflammatory disease. However, eosinophilic infiltration is present in 30.8% of bronchi and 54.8% of bronchioles [34]. Therefore, inhaled corticosteroids might have a role in the treatment of patients with eosinophilic inflammation. Two post hoc analyses yielded mixed conclusions [66, 67], in which inhaled fluticasone rather than budesonide was associated with QoL improvement in patients with eosinophilic bronchiectasis (eosinophil count ≥150 cells·μL−1). These data emphasised the importance of a well-designed RCT to investigate the efficacy and safety of inhaled corticosteroids and its combination with long-acting β-agonists and dual bronchodilators in bronchiectasis.
Antibiotics therapy
We recommend antibiotics for: 1) treatment of pulmonary exacerbations; 2) eradication of P. aeruginosa; and 3) reduction of exacerbation risk and improvement of symptoms. Although there are clear standards for antibiotics treatment in guidelines [1, 54, 55], inappropriate use of antibiotics remains common in China. The lack of inhaled antibiotics has posed challenges for the management of bacterial infection. Choice of antibiotics depends on the aim of therapy, disease severity, microorganisms, susceptibility to antibiotics, the therapeutic responses to prior antibiotic use and adverse effects.
Physicians in China focused on the treatment of exacerbation of bronchiectasis, but the importance of management at steady-state seems to have been ignored [54]. Empirical therapy covering P. aeruginosa is often commenced while waiting for microbiology results in patients with moderate-to-severe bronchiectasis [22, 24]. The duration of antibiotics for an exacerbation is 14 days based on limited evidence. Intravenous antibiotics are commonly prescribed for patients with severe exacerbation, otherwise oral antibiotics are prescribed. Further studies regarding the duration, routine (oral, intravenous, or nebulised) and biomarker predictors of antibiotic use at exacerbation should be performed.
P. aeruginosa negatively affects the clinical outcomes [24]; therefore eradication should be considered upon new isolation. The recommended eradication regime is oral or intravenous antibiotics for 2 weeks followed by inhaled antibiotics for 3 months [1, 54, 55]. Owing to a lack of inhaled antibiotics in China, eradication of P. aeruginosa has not been implemented in practice. A nationwide RCT of inhaled tobramycin versus inhaled saline with 28 days on/off treatment for 4 months (No. NCT03715322) in bronchiectasis patients has been completed. The pending approval of tobramycin inhalation solution might provide an important arsenal for eradication of P. aeruginosa in China (table 2).
TABLE 2.
Representative clinical trials initiated within mainland China
| Trial identifier number | Study title | Interventions | Duration | Participants | Primary end-points | Recruitment status |
| NCT04601792 | A Series of N-of-1 Trials of Traditional Chinese Medicine Based on Bayesian Method | Drug: Individualised Decoction Drug: placebo Drug: Tested drug minus heat-clearing herbs |
4 weeks per period ×3 | 71 | Patient Self-Rated Symptom Score (Likert scale) | Recruiting |
| NCT03715322 | Tobramycin in Bronchiectasis Colonised With Pseudomonas aeruginosa (TORNASOL) | Drug: Tobramycin Inhalant Product Other: usual care Drug: Natural saline inhalation |
4 months | 350 | Changes in sputum load of Pseudomonas aeruginosa at day 29 compared with baseline; Changes in Quality of Life in Bronchiectasis-Respiratory Symptom Score at day 29 compared with baseline |
Completed |
| NCT02088216 | Effect of Long-term, High-dose N-acetylcysteine on Exacerbations of Bronchiectasis (BENE) | Drug: N-acetylcysteine Other: On-demand treatment |
12 months | 161 | Median Number of Exacerbations over 12 months | Completed |
| NCT03843541 | A Clinical Trial to Compare the Efficacy and Safety of 1-week Treatment of Intravenous N-acetylcysteine (NAC) 600 mg Twice Daily, Ambroxol Hydrochloride 30 mg Twice Daily and Placebo as Expectorant Therapies in Adult Chinese Patients With Respiratory Tract Diseases and Abnormal Mucus Secretions | Drug: N-acetylcysteine (NAC) 600 mg Drug: Ambroxol hydrochloride 30 mg Other: placebo |
4 weeks | 333 | Change from baseline to end of 1-week treatment of mean sputum viscosity score of NAC and placebo; Change from baseline to end of 1-week treatment of mean expectoration difficulty score of NAC and placebo |
Completed |
Long-term antibiotics
Long-term antibiotics (inhaled and/or oral) should be considered if patients have frequent exacerbations (three or more exacerbations per year) despite optimal airway clearance and management of underlying causes. Long-term macrolides have the highest level of evidence for reducing exacerbation.
Long-term macrolides should be the first-line therapy in patients who have frequent exacerbation [54, 55]. Several RCTs and meta-analyses showed that long-term oral macrolides was associated with a nearly 50% exacerbation risk reduction [19, 68–72]. Nevertheless, macrolides use in China is still suboptimal possibly due to the safety concerns and the insufficient awareness. Some frequent exacerbators were not treated with macrolides, whereas other patients received long-term macrolides treatment despite the low frequency of prior exacerbations. Active NTM infection should be excluded before macrolides are prescribed. However, this recommendation is hard for clinicians to follow owing to the lack of facilities for NTM detection in some relatively low-level hospitals within China.
An individual-data meta-analysis unveiled exacerbation benefit in patients with P. aeruginosa colonisation [19]. The precise mechanism in patients infected with macrolide-resistant P. aeruginosa is poorly understood. The efficacy of macrolides was partly associated with the anti-inflammatory and immunomodulatory effects rather than antibacterial effects. Azithromycin and erythromycin might exert an anti-inflammatory effect via inhibiting inflammasomes activation against P. aeruginosa infection [73]. Nevertheless, further understanding the potent non-antimicrobial properties of macrolides will contribute to better personalised care in practice.
Interventional therapy for haemoptysis
Haemoptysis is a devastating symptom both at steady-state and exacerbation, which contributes to significant psychological distress and therapeutic burden. Bronchial artery embolisation (BAE) is the first-line therapy in patients with massive haemoptysis (100–200 mL/24 h) [1, 54, 55]. However, a 0.7–2% peri-procedural risk of stroke suggested careful risk–benefit estimation before embolisation [74], and it should only be performed by an experienced interventional radiologist in a specialist centre. Furthermore, recurrent haemoptysis after successful BAE is common. The cumulative recurrence within 1, 2, 3 and 5 years was 9.5%, 17.2%, 22.3% and 25.6%, respectively [75]. The predictors including sex, lung destruction, and systemic arterial-pulmonary circulation shunts showed good discrimination (area under curve, 0.72) with low prediction error (integrated Brier score, 0.129). BAE should be the first-line treatment only in patients with massive haemoptysis, and close follow-up is necessary for patients at risk of recurrence.
Surgery
Surgical resection of the involved lobe is one optional strategy for bronchiectasis patients in China. The guidelines recommended that surgical resection should be reserved to severe localised bronchiectasis when all available treatments have failed or the patients have life-threatening haemoptysis [1, 54, 55]. A meta-analysis of patients with localised bronchiectasis showed the pooled mortality and post-operative morbidity of 1.4% and 16.7%, respectively. Symptoms were improved among 66.5% of patients. Successful surgical treatment as well as collaboration between respiratory physician and surgeons are needed.
Establishment of China Bronchiectasis Registry and Research Collaboration (BE-China)
The field of bronchiectasis developed slowly since the first description in 1819. Fortunately, a renewed interest in the past decade has transformed the field –Professor John Hurst referred to this era as the “age of bronchiectasis”. Large-scale registries such as EMBARC and US registry [15, 16] have contributed substantially to the latest advances. However, the clinicians still do not have licensed therapies for bronchiectasis, and clinical trials have been unsuccessful [1, 54, 55]. More data from outside Europe and the USA are required [76, 77]. The Establishment of China Bronchiectasis Registry and Research Collaboration (BE-China, www.chinabronchiectasis.com), formally launched in January 2020, will promote research in all areas of bronchiectasis by integrating the patient registry data from individual hospitals throughout China, and facilitate multidisciplinary collaborative research around the world. A series of studies have been conducted, including the understanding of the aetiology and microbiology of bronchiectasis throughout China. Longitudinal cohort studies are also underway. Additionally, the BE-China has become the avenue to foster multicentre trials such as the tobramycin and sitafloxacin clinical trials.
The expert consensus in China
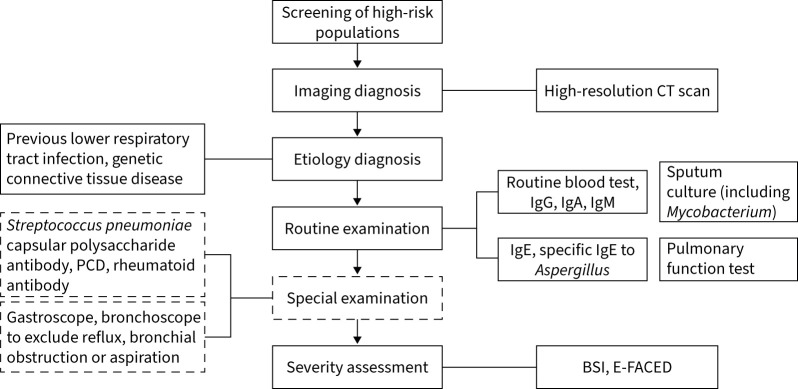
The latest expert consensus on the diagnosis and treatment of adult bronchiectasis in China was published by the Chinese Thoracic Society in 2021 [54]. The document summarised the epidemiology, pathogenesis, etiology, clinical diagnosis, treatment strategies and patient education of bronchiectasis in China according to a systematic review of the literature. The document endorsed a standard diagnostic procedure for clinicians to follow (figure 2).
FIGURE 2.
Standard diagnostic process of bronchiectasis proposed by the Chinese expert consensus document. PCD: primary ciliary dyskinesia; CT: computed tomography; BSI: bronchiectasis severity index.
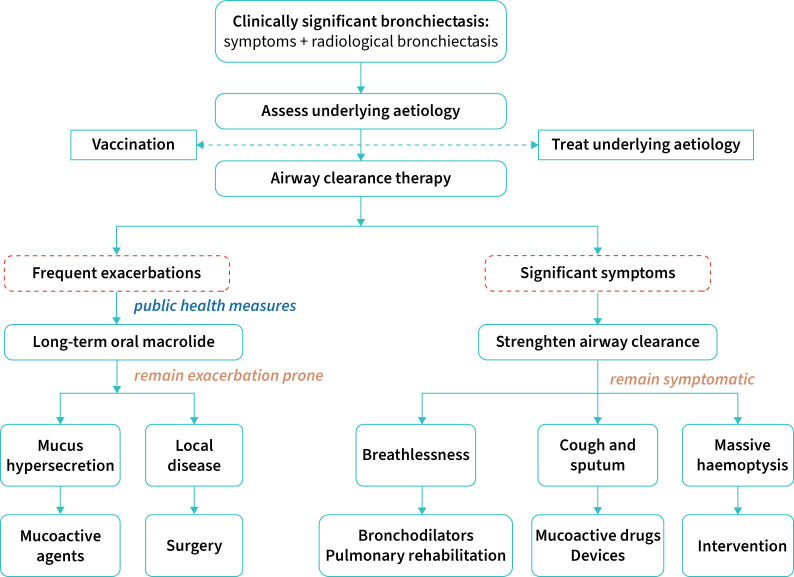
Meanwhile, the document has updated the patient management recommendations. At stage 1, clinicians should identify and treat underlying causes, initiate airway clearance and/or pulmonary rehabilitation, influenza vaccination, control infection and self-management. At stage 2, for adults with bronchiectasis who have three or more exacerbations per year despite optimisation of stage 1 management, long-term mucoactive drugs and oral macrolides should be suggested. At stage 3, for adults with bronchiectasis who have three or more exacerbations per year despite optimisation of stage 2 management, intravenous antibiotic therapy every 2–3 months or airway clearance by bronchoscopy should be considered. Figure 3 summarises the key recommended steps for the clinical management of bronchiectasis in clinical practice.
FIGURE 3.
Recommended therapeutic flow chart for the management of patients with bronchiectasis.
Impact of COVID-19 on the clinical outcomes and management of bronchiectasis
Coronavirus disease 2019 (COVID-19) has resulted in a global pandemic related to the transmission of various variants, causing substantial morbidity and mortality. The role of severe acute respiratory syndrome coronarvirus-2 (SARS-CoV-2) as the aetiology of bronchiectasis has been suggested by an observational study [78]. Nearly 30% of inpatients with COVID-19 developed radiological evidence of bronchiectasis at convalescence, and the area of severely infected regions correlated with a greater probability of developing bronchiectasis [78]. The key events driving the bronchial dilation remained unclear. Patients with pre-existing bronchiectasis might have an aggravated disease progression once contracting SARS-CoV-2.
Amid other intense debates was the impact of COVID-19 on the clinical outcomes of bronchiectasis. The underlying respiratory diseases might predispose to a greater susceptibility to contracting the virus and poorer clinical outcomes. The findings from different nationwide registry were, however, inconsistent. The Korean registry cohort showed that the prevalence of bronchiectasis was 1.6% among 8070 patients with COVID-19 and that patients with bronchiectasis had significantly greater symptom burden and more severe infections compared with those without bronchiectasis [79]. In support of these findings, the registry across England (QResearch database) reported that patients with pre-existing bronchiectasis had an elevated risk of hospitalisation but not death from COVID-19 [80]. By contrast, the Chinese nationwide registry cohort, which comprised nearly 40 000 patients with COVID-19, did not identify any positive correlation between bronchiectasis and the clinically meaningful outcomes of COVID-19, measured as the receipt of mechanical ventilation, intensive care unit admission or death [81]. The different findings might be attributable to the different study populations and outcome assessments.
Early in the outbreak, the pressure of the COVID-19 pandemic on daily management of bronchiectasis was unprecedented, especially when taking into account the interruption of out- and inpatient services. The interruption of outpatient services has stressed patients who relied on frequent follow-up owing to the challenges of re-filling maintenance therapy during outbreaks when the scale of routine respiratory clinics has been minimised. Research progress has been lagging behind schedule owing to the closure of many routine practices. However, recent epidemiological studies suggested a markedly reduced risk of acute exacerbations of bronchiectasis during COVID-19 outbreaks [82], possibly because of the strengthened public health measures such as wearing a face mask, more frequent hand washing and social distancing. Similarly, these findings were reminiscent of the remarkable decline in the frequency of bronchiectasis exacerbations compared with baseline levels in the tobramycin clinical trial (NCT03715322, unpublished data). These findings reinforced clinician's precaution of strengthening personal protection strategies among patients with pre-existing bronchiectasis amid the pandemic [83].
Implications for global practice
Although most of the above-mentioned findings have documented the latest advances in China, there are a number of findings that might be generalised to many other countries. We are witnessing more opportunities when the experts from China and other parts of the world could jointly battle against the enigma of bronchiectasis. For instance, airway hypersecretion is a hallmark presentation of bronchiectasis with a paucity of our understanding of the underlying pathophysiology. Our previous finding that the dysfunction of CFTR and increased intracellular chloride concentrations within the airway epithelium were implicated in the persistently heightened airway inflammation has shed light on a phase 2a ongoing clinical trial (No. NCT04396366), which sought to address whether a novel class of CFTR agonist will help alleviate bacterial infection and airway inflammation in adults with non-cystic fibrosis bronchiectasis. P. aeruginosa infection has been fairly common in China, but unfortunately the effective therapeutic approaches have been scarce. We and others have recently completed a national multicenter RCT (No. NCT03715322), which has revealed the efficacy of inhaled tobramycin solution in substantially improving the QoL while decreasing the sputum density of P. aeruginosa in adults with bronchiectasis (WJ Guan et al., personal communication). Because of the lack of marketed inhaled antibiotics in China, we believe that these new findings will be generalisable to many other developing countries or remote settings where essential treatments for P. aeruginosa infection are lacking. Similarly, there remain many other unanswered questions from clinical practice that await further research in other parts of the world. Finally, while the Chinese bronchiectasis registries would be helpful to facilitate multidisciplinary collaborative research around the world, there remain considerable differences in the settings and patient characteristics as compared with those from other Western countries. Therefore, findings from the Chinese bronchiectasis registries could provide valuable insights into geographical and ethnic differences of bronchiectasis and might be an important opportunity to demonstrate the generalizability of research work conducted in Western countries to the other parts of the globe (especially the developing countries).
Conclusion
The rapid pace of medical advances has greatly fuelled our ever-improving understanding of the pathophysiology, diagnosis and treatment of bronchiectasis. Improvement in awareness among physician and patient communities, and the availability of high-quality medical evidence and therapeutic options have contributed to better healthcare that aims to reduce the personal and social burden and ultimately translate into improved clinical outcomes.
Acknowledgements
We thank Jiayang Yan (Department of Respiratory and Critical Care Medicine, Affiliated Ruijin Hospital, School of Medicine, Shanghai Jiaotong University, Shanghai, China), Jing Bi and Lei Zhou (Zhongshan Hospital, Fudan University, Shanghai, China) for their preparation of the contents in the manuscript.
Footnotes
Author contributions: J-f. Xu, Y-h. Gao, Y-l. Song, J-m. Qu and W-j. Guan performed the literature review and drafted the manuscript; W-j. Guan contributed to study conception; J-f. Xu, Y-h. Gao, Y-l. Song, J-m. Qu and W-j. Guan provided critical review of the manuscript and approved the final submission.
Conflict of interest: W-j. Guan reports support for the present manuscript received from Zhongnanshan Medical Foundation of Guangdong Province (ZNSA-2020013) and National Natural Science Foundation grant 81870003. W-j. Guan is an associate editor of this journal. The remaining authors have nothing to disclose.
Support statement: This review was supported by National Natural Science Foundation grant 81870003, Zhongnanshan Medical Foundation of Guangdong Province grant ZNSA-2020013 (to W-j. Guan), National Natural Science Foundation grant 81925001 (to J-f. Xu) and the Shanghai Pujiang Program 2021 (21PJD061) (to Y-h. Gao). Funding information for this article has been deposited with the Crossref Funder Registry.
References
- 1.Hill AT, Sullivan AL, Chalmers JD, et al. British Thoracic Society Guideline for bronchiectasis in adults. Thorax 2019; 74: 1–69. [DOI] [PubMed] [Google Scholar]
- 2.Zhou YM, Wang C, Yao WZ, et al. [The prevalence and risk factors of bronchiectasis in residents aged 40 years old and above in seven cities in China]. Zhonghua Nei Ke Za Zhi 2013; 52: 379–382. [PubMed] [Google Scholar]
- 3.Tsang KW, Chan K, Ho P, et al. Sputum elastase in steady-state bronchiectasis. Chest 2000; 117: 420–426. doi: 10.1378/chest.117.2.420 [DOI] [PubMed] [Google Scholar]
- 4.Tsang KW, Ho PL, Lam WK, et al. Inhaled fluticasone reduces sputum inflammatory indices in severe bronchiectasis. Am J Respir Crit Care Med 1998; 158: 723–727. doi: 10.1164/ajrccm.158.3.9710090 [DOI] [PubMed] [Google Scholar]
- 5.Lin JL, Xu JF, Qu JM. Bronchiectasis in China. Ann Am Thorac Soc 2016; 13: 609–616. doi: 10.1513/AnnalsATS.201511-740PS [DOI] [PubMed] [Google Scholar]
- 6.Weycker D, Hansen GL, Seifer FD. Prevalence and incidence of noncystic fibrosis bronchiectasis among US adults in 2013. Chron Respir Dis 2017; 14: 377–384. doi: 10.1177/1479972317709649 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ringshausen FC, de Roux A, Diel R, et al. Bronchiectasis in Germany: a population-based estimation of disease prevalence. Eur Respir J 2015; 46: 1805–1807. doi: 10.1183/13993003.00954-2015 [DOI] [PubMed] [Google Scholar]
- 8.Diel R, Ewig S, Blaas S, et al. Incidence of patients with non-cystic fibrosis bronchiectasis in Germany – a healthcare insurance claims data analysis. Respir Med 2019; 151: 121–127. doi: 10.1016/j.rmed.2019.04.007 [DOI] [PubMed] [Google Scholar]
- 9.Quint JK, Millett ER, Joshi M, et al. Changes in the incidence, prevalence and mortality of bronchiectasis in the UK from 2004 to 2013: a population-based cohort study. Eur Respir J 2016; 47: 186–193. doi: 10.1183/13993003.01033-2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Monteagudo M, Rodríguez-Blanco T, Barrecheguren M, et al. Prevalence and incidence of bronchiectasis in Catalonia, Spain: a population-based study. Respir Med 2016; 121: 26–31. doi: 10.1016/j.rmed.2016.10.014 [DOI] [PubMed] [Google Scholar]
- 11.Aliberti S, Sotgiu G, Lapi F, et al. Prevalence and incidence of bronchiectasis in Italy. BMC Pulm Med 2020; 20: 15. doi: 10.1186/s12890-020-1050-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Phua HP, Lim WY, Ganesan G, et al. Epidemiology and economic burden of bronchiectasis requiring hospitalisation in Singapore. ERJ Open Res 2021; 7: 00334-2021. doi: 10.1183/23120541.00334-2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Choi H, Yang B, Nam H, et al. Population-based prevalence of bronchiectasis and associated comorbidities in South Korea. Eur Respir J 2019; 54: 1900194. doi: 10.1183/13993003.00194-2019 [DOI] [PubMed] [Google Scholar]
- 14.Aliberti S, Goeminne PC, O'Donnell AE, et al. Criteria and definitions for the radiological and clinical diagnosis of bronchiectasis in adults for use in clinical trials: international consensus recommendations. Lancet Respir Med 2022: 10: 298–306. [DOI] [PubMed] [Google Scholar]
- 15.Chalmers JD, Aliberti S, Polverino E, et al. The EMBARC European Bronchiectasis Registry: protocol for an international observational study. ERJ Open Res 2016; 2: 00081-2015. doi: 10.1183/23120541.00081-2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Aksamit TR, O'Donnell AE, Barker A, et al. Adult patients with bronchiectasis: a first look at the US Bronchiectasis Research Registry. Chest 2017; 151: 982–992. doi: 10.1016/j.chest.2016.10.055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chalmers JD, Chang AB, Chotirmall SH, et al. Bronchiectasis. Nat Rev Dis Primers 2018; 4: 45. doi: 10.1038/s41572-018-0042-3 [DOI] [PubMed] [Google Scholar]
- 18.Muñoz G, de Gracia J, Buxó M, et al. Long-term benefits of airway clearance in bronchiectasis: a randomised placebo-controlled trial. Eur Respir J 2018; 51: 1701926. doi: 10.1183/13993003.01926-2017 [DOI] [PubMed] [Google Scholar]
- 19.Chalmers JD, Boersma W, Lonergan M, et al. Long-term macrolide antibiotics for the treatment of bronchiectasis in adults: an individual participant data meta-analysis. Lancet Respir Med 2019; 7: 845–854. doi: 10.1016/S2213-2600(19)30191-2 [DOI] [PubMed] [Google Scholar]
- 20.Haworth CS, Bilton D, Chalmers JD, et al. Inhaled liposomal ciprofloxacin in patients with non-cystic fibrosis bronchiectasis and chronic lung infection with Pseudomonas aeruginosa (ORBIT-3 and ORBIT-4): two phase 3, randomised controlled trials. Lancet Respir Med 2019; 7: 213–226. doi: 10.1016/S2213-2600(18)30427-2 [DOI] [PubMed] [Google Scholar]
- 21.Flume PA, Chalmers JD, Olivier KN. Advances in bronchiectasis: endotyping, genetics, microbiome, and disease heterogeneity. Lancet 2018; 392: 880–890. doi: 10.1016/S0140-6736(18)31767-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guan WJ, Gao YH, Xu G, et al. Sputum bacteriology in steady-state bronchiectasis in Guangzhou. China Int J Tubercul Lung Dis 2015; 19: 610–619. doi: 10.5588/ijtld.14.0613 [DOI] [PubMed] [Google Scholar]
- 23.Guan WJ, Gao YH, Xu G, et al. Effect of airway Pseudomonas aeruginosa isolation and infection on steady-state bronchiectasis in Guangzhou, China. J Thorac Dis 2015; 7: 625–636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang H, Ji XB, Mao B, et al. Pseudomonas aeruginosa isolation in patients with non-cystic fibrosis bronchiectasis: a retrospective study. BMJ Open 2018; 8: e014613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Luo RG, Miao XY, Luo LL, et al. Presence of pldA and exoU in mucoid Pseudomonas aeruginosa is associated with high risk of exacerbations in non-cystic fibrosis bronchiectasis patients. Clin Microbiol Infect 2019; 25: 601–606. doi: 10.1016/j.cmi.2018.07.008 [DOI] [PubMed] [Google Scholar]
- 26.Gao YH, Guan WJ, Zhu YN, et al. Antibiotic-resistant Pseudomonas aeruginosa infection in patients with bronchiectasis: prevalence, risk factors and prognostic implications. Int J Chron Obstruct Pulm Dis 2018; 13: 237–246. doi: 10.2147/COPD.S150250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dicker AJ, Lonergan M, Keir HR, et al. The sputum microbiome and clinical outcomes in patients with bronchiectasis: a prospective observational study. Lancet Respir Med 2021; 9: 885–896. doi: 10.1016/S2213-2600(20)30557-9 [DOI] [PubMed] [Google Scholar]
- 28.Guan WJ, Yuan JJ, Li HM, et al. Proteobacteria community compositions correlate with bronchiectasis severity. Int J Tubercul Lung Dis 2018; 22: 1095–1105. doi: 10.5588/ijtld.18.0037 [DOI] [PubMed] [Google Scholar]
- 29.Gao YH, Guan WJ, Xu G, et al. The role of viral infection in pulmonary exacerbations of bronchiectasis in adults: a prospective study. Chest 2015; 147: 1635–1643. doi: 10.1378/chest.14-1961 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen CL, Huang Y, Yuan JJ, et al. The roles of bacteria and viruses in bronchiectasis exacerbation: a prospective study. Arch Bronconeumol 2020; 56: 621–629. [DOI] [PubMed] [Google Scholar]
- 31.Chen CL, Huang Y, Martinez-Garcia MA, et al. The role of Epstein-Barr virus in adults with bronchiectasis: a prospective cohort study. Open Forum Infect Dis 2020; 7: ofaa235. doi: 10.1093/ofid/ofaa235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yu S, Di C, Chen S, et al. Distinct immune signatures discriminate between asymptomatic and presymptomatic SARS-CoV-2(pos) subjects. Cell Res 2021; 31: 1148–1162. doi: 10.1038/s41422-021-00562-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tsang KW, Tipoe G, Sun J, et al. Clinical value of ciliary assessment in bronchiectasis. Lung 2005; 183: 73–86. doi: 10.1007/s00408-004-2520-5 [DOI] [PubMed] [Google Scholar]
- 34.Chen ZG, Li YY, Wang ZN, et al. Aberrant epithelial remodeling with impairment of cilia architecture in non-cystic fibrosis bronchiectasis. J Thorac Dis 2018; 10: 1753–1764. doi: 10.21037/jtd.2018.02.13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhang YL, Chen PX, Guan WJ, et al. Increased intracellular Cl(-) concentration promotes ongoing inflammation in airway epithelium. Mucosal Immunol 2018; 11: 1149–1157. doi: 10.1038/s41385-018-0013-8 [DOI] [PubMed] [Google Scholar]
- 36.Shum DK, Chan SC, Ip MS. Neutrophil-mediated degradation of lung proteoglycans: stimulation by tumor necrosis factor-alpha in sputum of patients with bronchiectasis. Am J Respir Crit Care Med 2000; 162: 1925–1931. doi: 10.1164/ajrccm.162.5.9907064 [DOI] [PubMed] [Google Scholar]
- 37.Chan SC, Shum DK, Ip MS. Sputum sol neutrophil elastase activity in bronchiectasis: differential modulation by syndecan-1. Am J Respir Crit Care Med 2003; 168: 192–198. doi: 10.1164/rccm.200208-829OC [DOI] [PubMed] [Google Scholar]
- 38.Zheng L, Lam WK, Tipoe GL, et al. Overexpression of matrix metalloproteinase-8 and -9 in bronchiectatic airways in vivo. Eur Respir J 2002; 20: 170–176. doi: 10.1183/09031936.02.00282402 [DOI] [PubMed] [Google Scholar]
- 39.Wang L, Liu J, Wang W, et al. Targeting IL-17 attenuates hypoxia-induced pulmonary hypertension through downregulation of β-catenin. Thorax 2019; 74: 564–578. doi: 10.1136/thoraxjnl-2018-211846 [DOI] [PubMed] [Google Scholar]
- 40.Liu Y, Huang X, Chen D, et al. The detrimental qualitative and quantitative alterations of circulating endothelial progenitor cells in patients with bronchiectasis. Respir Med 2021; 176: 106270. doi: 10.1016/j.rmed.2020.106270 [DOI] [PubMed] [Google Scholar]
- 41.Gao YH, Liu SX, Cui JJ, et al. Subclinical atherosclerosis in adults with steady-state bronchiectasis: a case-control study. Respir Med 2018; 134: 110–116. doi: 10.1016/j.rmed.2017.11.024 [DOI] [PubMed] [Google Scholar]
- 42.Wang H, Ji XB, Li CW, et al. Clinical characteristics and validation of bronchiectasis severity score systems for post-tuberculosis bronchiectasis. Clin Respir J 2018; 12: 2346–2353. doi: 10.1111/crj.12911 [DOI] [PubMed] [Google Scholar]
- 43.Ooi GC, Khong PL, Chan-Yeung M, et al. High-resolution CT quantification of bronchiectasis: clinical and functional correlation. Radiology 2002; 225: 663–672. doi: 10.1148/radiol.2253011575 [DOI] [PubMed] [Google Scholar]
- 44.Wu FJ, Qi Q, Hu Q, et al. [Correlation between chest CT features and clinical characteristics of patients with bronchiectasis]. Zhonghua Yi Xue Za Zhi 2019; 99: 2982–2988. [DOI] [PubMed] [Google Scholar]
- 45.Shi L, Wei F, Ma T, et al. Impact of radiographic bronchiectasis in COPD. Respir Care 2020; 65: 1561–1573. doi: 10.4187/respcare.07390 [DOI] [PubMed] [Google Scholar]
- 46.Dou S, Zheng C, Cui L, et al. High prevalence of bronchiectasis in emphysema-predominant COPD patients. Int J Chron Obstruct Pulm Dis 2018; 13: 2041–2047. doi: 10.2147/COPD.S163243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Jin J, Li S, Yu W, et al. Emphysema and bronchiectasis in COPD patients with previous pulmonary tuberculosis: computed tomography features and clinical implications. Int J Chron Obstruct Pulm Dis 2018; 13: 375–384. doi: 10.2147/COPD.S152447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Dou S, Zheng C, Ji X, et al. Co-existence of COPD and bronchiectasis: a risk factor for a high ratio of main pulmonary artery to aorta diameter (PA:A) from computed tomography in COPD patients. Int J Chron Obstruct Pulm Dis 2018; 13: 675–681. doi: 10.2147/COPD.S156126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Guan WJ, Gao YH, Xu G, et al. Bronchodilator response in adults with bronchiectasis: correlation with clinical parameters and prognostic implications. J Thorac Dis 2016; 8: 14–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Guan WJ, Gao YH, Xu G, et al. Impulse oscillometry in adults with bronchiectasis. Ann Am Thorac Soc 2015; 12: 657–665. doi: 10.1513/AnnalsATS.201406-280OC [DOI] [PubMed] [Google Scholar]
- 51.Guan WJ, Yuan JJ, Gao YH, et al. Maximal mid-expiratory flow is a surrogate marker of lung clearance index for assessment of adults with bronchiectasis. Sci Rep 2016; 6: 28467. doi: 10.1038/srep28467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Guan WJ, Gao YH, Xu G, et al. Characterization of lung function impairment in adults with bronchiectasis. PLoS One 2014; 9: e113373. doi: 10.1371/journal.pone.0113373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Guan WJ, Gao YH, Xu G, et al. Inflammatory responses, spirometry, and quality of life in subjects with bronchiectasis exacerbations. Respir Care 2015; 60: 1180–1189. doi: 10.4187/respcare.04004 [DOI] [PubMed] [Google Scholar]
- 54.Bronchiectasis Expert Consensus Writing Group, Pulmonary Infection Assembly, Chinese Thoracic Society . [Expert consensus on the diagnosis and treatment of adult bronchiectasis in China]. Zhonghua Jie He He Hu Xi Za Zhi 2021; 44: 311–321. [DOI] [PubMed] [Google Scholar]
- 55.Polverino E, Goeminne PC, McDonnell MJ, et al. European Respiratory Society guidelines for the management of adult bronchiectasis. Eur Respir J 2017; 50: 1700629. doi: 10.1183/13993003.00629-2017 [DOI] [PubMed] [Google Scholar]
- 56.Spinou A, Chalmers JD. Respiratory physiotherapy in the bronchiectasis guidelines: is there a loud voice we are yet to hear? Eur Respir J 2019; 54: 1901610. doi: 10.1183/13993003.01610-2019 [DOI] [PubMed] [Google Scholar]
- 57.Liu Y, Lu HW, Gu SY, et al. Bronchoscopic airway clearance therapy for acute exacerbations of bronchiectasis. EBioMedicine 2021; 72: 103587. doi: 10.1016/j.ebiom.2021.103587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Patel S, Cole AD, Nolan CM, et al. Pulmonary rehabilitation in bronchiectasis: a propensity-matched study. Eur Respir J 2019; 53: 1801264. doi: 10.1183/13993003.01264-2018 [DOI] [PubMed] [Google Scholar]
- 59.Qi Q, Ailiyaer Y, Liu R, et al. Effect of N-acetylcysteine on exacerbations of bronchiectasis (BENE): a randomized controlled trial. Respir Res 2019; 20: 73. doi: 10.1186/s12931-019-1042-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bilton D, Tino G, Barker AF, et al. Inhaled mannitol for non-cystic fibrosis bronchiectasis: a randomised, controlled trial. Thorax 2014; 69: 1073–1079. doi: 10.1136/thoraxjnl-2014-205587 [DOI] [PubMed] [Google Scholar]
- 61.Gao YH, Abo Leyah H, Finch S, et al. Relationship between symptoms, exacerbations, and treatment response in bronchiectasis. Am J Respir Crit Care Med 2020; 201: 1499–1507. doi: 10.1164/rccm.201910-1972OC [DOI] [PubMed] [Google Scholar]
- 62.Lee SY, Lee JS, Lee SW, et al. Effects of treatment with long-acting muscarinic antagonists (LAMA) and long-acting beta-agonists (LABA) on lung function improvement in patients with bronchiectasis: an observational study. J Thorac Dis 2021; 13: 169–177. doi: 10.21037/jtd-20-1282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Jeong HJ, Lee H, Carriere KC, et al. Effects of long-term bronchodilators in bronchiectasis patients with airflow limitation based on bronchodilator response at baseline. Int J Chron Obstruct Pulmon Dis 2016; 11: 2757–2764. doi: 10.2147/COPD.S115581 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Jang EJ, Lee CH, Yoon HI, et al. Association between inhaler use and risk of haemoptysis in patients with non-cystic fibrosis bronchiectasis. Respirology 2015; 20: 1213–1221. doi: 10.1111/resp.12618 [DOI] [PubMed] [Google Scholar]
- 65.Jayaram L, Vandal AC, Chang C, et al. Tiotropium treatment for bronchiectasis: a randomised, placebo-controlled, crossover trial. Eur Respir J 2021; 8: 2102184. doi: 10.1183/13993003.02184-2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Aliberti S, Sotgiu G, Blasi F, et al. Blood eosinophils predict inhaled fluticasone response in bronchiectasis. Eur Respir J 2020; 56: 2000453. doi: 10.1183/13993003.00453-2020 [DOI] [PubMed] [Google Scholar]
- 67.Aliberti S, Sotgiu G, Martinez Garcia MA. Blood eosinophils do not predict inhaled budesonide response in bronchiectasis. Eur Respir J 2020; 56: 2002210. doi: 10.1183/13993003.02210-2020 [DOI] [PubMed] [Google Scholar]
- 68.Wong C, Jayaram L, Karalus N, et al. Azithromycin for prevention of exacerbations in non-cystic fibrosis bronchiectasis (EMBRACE): a randomised, double-blind, placebo-controlled trial. Lancet 2012; 380: 660–667. doi: 10.1016/S0140-6736(12)60953-2 [DOI] [PubMed] [Google Scholar]
- 69.Altenburg J, de Graaff CS, Stienstra Y, et al. Effect of azithromycin maintenance treatment on infectious exacerbations among patients with non-cystic fibrosis bronchiectasis: the BAT randomized controlled trial. JAMA 2013; 309: 1251–1259. doi: 10.1001/jama.2013.1937 [DOI] [PubMed] [Google Scholar]
- 70.Serisier DJ, Martin ML, McGuckin MA, et al. Effect of long-term, low-dose erythromycin on pulmonary exacerbations among patients with non-cystic fibrosis bronchiectasis: the BLESS randomized controlled trial. JAMA 2013; 309: 1260–1267. doi: 10.1001/jama.2013.2290 [DOI] [PubMed] [Google Scholar]
- 71.Gao YH, Guan WJ, Xu G, et al. Macrolide therapy in adults and children with non-cystic fibrosis bronchiectasis: a systematic review and meta-analysis. PLoS One 2014; 9: e90047. doi: 10.1371/journal.pone.0090047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Fan LC, Lu HW, Wei P, et al. Effects of long-term use of macrolides in patients with non-cystic fibrosis bronchiectasis: a meta-analysis of randomized controlled trials. BMC Infect Dis 2015; 15: 160. doi: 10.1186/s12879-015-0872-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Fan LC, Lin JL, Yang JW, et al. Macrolides protect against Pseudomonas aeruginosa infection via inhibition of inflammasomes. Am J Physiol Lung Cell Mol Physiol 2017; 313: L677–l686. doi: 10.1152/ajplung.00123.2017 [DOI] [PubMed] [Google Scholar]
- 74.Panda A, Bhalla AS, Goyal A. Bronchial artery embolization in hemoptysis: a systematic review. Diag Intervent Radiol 2017; 23: 307–317. doi: 10.5152/dir.2017.16454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Yan HT, Lu GD, Huang XZ, et al. A nomogram to predict recurrence after bronchial artery embolization for hemoptysis due to bronchiectasis. Cardiovasc Intervent Radiol 2021; 44: 1609–1617. doi: 10.1007/s00270-021-02923-0 [DOI] [PubMed] [Google Scholar]
- 76.Chandrasekaran R, Mac Aogáin M, Chalmers JD, et al. Geographic variation in the aetiology, epidemiology and microbiology of bronchiectasis. BMC Pulm Med 2018; 18: 83. doi: 10.1186/s12890-018-0638-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Dhar R, Singh S, Talwar D, et al. Bronchiectasis in India: results from the European Multicentre Bronchiectasis Audit and Research Collaboration (EMBARC) and Respiratory Research Network of India Registry. Lancet Glob Health 2019; 7: e1269–e1279. doi: 10.1016/S2214-109X(19)30327-4 [DOI] [PubMed] [Google Scholar]
- 78.Wang Y, Mao K, Li Z, et al. Clinical study of pulmonary CT lesions and associated bronchiectasis in 115 convalescent patients with novel coronavirus pneumonia (COVID-19) in China. Can J Physiol Pharmacol 2021; 99: 328–331. doi: 10.1139/cjpp-2020-0522 [DOI] [PubMed] [Google Scholar]
- 79.Choi H, Lee H, Lee SK, et al. Impact of bronchiectasis on susceptibility to and severity of COVID-19: a nationwide cohort study. Ther Adv Respir Dis 2021; 15: 1753466621995043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Aveyard P, Gao M, Lindson N, et al. Association between pre-existing respiratory disease and its treatment, and severe COVID-19: a population cohort study. Lancet Respir Med 2021; 9: 909–923. doi: 10.1016/S2213-2600(21)00095-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Guan WJ, Liang WH, Shi Y, et al. Chronic Respiratory Diseases and the Outcomes of COVID-19: a nationwide retrospective cohort study of 39,420 cases. J Allergy Clin Immunol Pract 2021; 9: 2645–2655.e14. doi: 10.1016/j.jaip.2021.02.041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Crichton ML, Shoemark A, Chalmers JD. The impact of the COVID-19 pandemic on exacerbations and symptoms in bronchiectasis: a prospective study. Am J Respir Crit Care Med 2021; 204: 857–859. doi: 10.1164/rccm.202105-1137LE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.He ZF, Zhong NS, Guan WJ. Impact of chronic respiratory diseases on the outcomes of COVID-19. Arch Bronconeumol 2022; 58: 5–7. doi: 10.1016/j.arbres.2021.05.009 [DOI] [PMC free article] [PubMed] [Google Scholar]