Therapy failure of antiretroviral drugs may occur due to many factors, and one of the most important is drug resistance development (3). In the last few years, several authors have written about the phenomenon of multiple nucleoside resistance (MDR), which confers simultaneous resistance to many nucleoside reverse transcriptase inhibitors (NRTI) (2, 4). Different mechanisms are involved in this event; one of these is the codon 69 insertion, whose role continues to be controversial. Larder et al. (5) hypothesized that this insertion could effect nucleoside resistance particularly when combined with other mutated codons within the reverse transcriptase, and Winters et al. (10) reported that this insertion accounted for increases of up to 10-fold in the 50% inhibitory concentrations (IC50) of most nucleosides except stavudine.
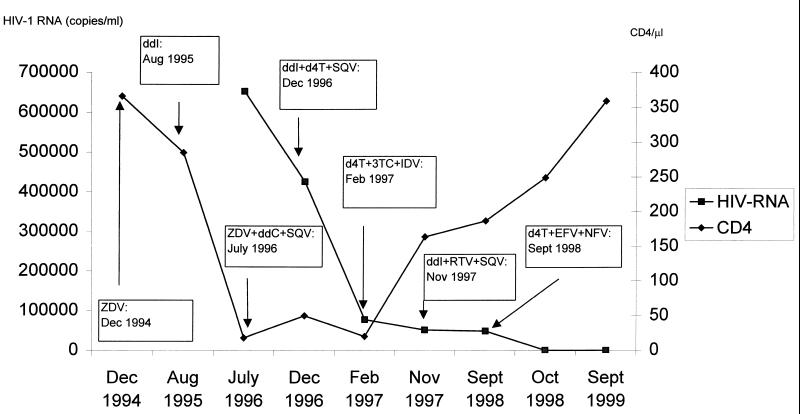
Here we report the case of a patient treated with various antiretroviral drugs in whom we found a viral mutant with an unusual codon 69 reverse transcriptase insertion. A 37-year-old heterosexual woman began therapy with zidovudine (ZDV) in December 1994. After 8 months we changed ZDV to didanosine (ddI) due to a CD4+ cell decline. In July 1996 we measured the viral load for the first time, which was very high (653,000 copies/ml; branched DNA), so we began triple therapy with ZDV–zalcitabine (ddC)–saquinavir (SQV). Three months later we again changed treatment due to a virological failure (Fig. 1), and the patient then began taking stavudine (d4T)-ddI-SQV. In November 1997, after a transient virological response there was a viral-load rebound; therefore, we started a double-protease-inhibitor regimen with ddI–ritonavir (RTV)–SQV, because it was the only alternative regimen available. Finally, after another virological failure and on the basis of a resistance test performed, in September 1998 we introduced a new treatment regimen: d4T–efavirenz (EFV)–nelfinavir (NFV). One month later, the viral load decreased to an undetectable level, and the situation has remained unchanged. Figure 1 shows CD4+ cell counts and HIV-1 RNA modifications in relation to drug changes.
FIG. 1.
Immunologic-virologic profile of the patient over time.
The patient was evaluated for drug resistance in March 1997 with both genotypic and phenotypic analyses, conducted as previously reported (6, 9). The results showed a genotypic profile for the protease gene typical of multiple mutations linked to protease inhibitor cross-resistance. Of note, the reverse transcriptase exhibited a resistance pattern that to our knowledge has been reported only once before (5). The reverse transcriptase of our patient (plasma RNA) showed mutations V106I and F214L and the T69S insertion VG (GenBank accession number, AF186771); the amino acid sequence from the culture supernatant showed a codon 210W mutation as well. The phenotypic analysis, repeated in several experiments, revealed a marked resistance to the protease inhibitors indinavir and RTV (IC50 > 0.1 μM), to the NRTI ZDV (IC50 > 10 μM), ddI (IC50 > 1 μM), and lamivudine (IC50 = 1.169 μM). On the other hand, a phenotypic susceptibility was evident for d4T (IC50 = 0.052 μM) and the new protease inhibitor tipranavir (IC50 = 0.191 μM).
This case offers additional evidence of newly described MDR insertions in heavily pretreated HIV-1-infected subjects in the absence of optimal viral suppression. Moreover, the finding of novel mutated codons can account for the emergence of resistance in the presence of potentially effective antiviral regimens, as described by Bloor et al. (1), who pointed out the role of the E44D and V118I mutations within the HIV-1 reverse transcriptase. As previously reported (10), our patient experienced the beneficial effect of d4T therapy, combined with EFV and NFV. Nevertheless, the potential development of d4T phenotypic resistance should be taken into account for NRTI-treated patients (7). Our report, as have others (5, 8), emphasizes the importance of continued monitoring for genotypic drug resistance in order to detect new mutations that can impair the efficacy of combination therapies against HIV-1.
REFERENCES
- 1.Bloor S, Hertogs K, De Vroey V, Miller V, Sturmer M, Larder B. Lamivudine-resistant HIV-1 clinical isolates lacking the Met184Val mutation have novel polymorphisms in RT. Antivir Ther. 1999;4(Suppl. 1):19. [Google Scholar]
- 2.de Jong J J, Jurrians S, Goudsmit J, Baan E, Huismans R, Danner S, Hillebrand M, Ten Veen J H, de Wolf F. Insertion of two amino acids in reverse transcriptase (RT) during antiretroviral combination therapy: implications for resistance against RT inhibitors. Antivir Ther. 1998;3(Suppl. 2):15. [Google Scholar]
- 3.Hirsch M S, Conway B, D'Aquila R T, Johnson V A, Brun-Vezinet F, Clotet B, Demeter L M, Hammer S M, Jacobsen D M, Kuritzkes D R, Loveday C, Mellors J W, Vella S, Richman D D for the AIDS Society-USA Panel. Antiretroviral drug resistance testing in adults with HIV infection: implications for clinical management. JAMA. 1998;279:1984–1991. doi: 10.1001/jama.279.24.1984. [DOI] [PubMed] [Google Scholar]
- 4.Iversen A K N, Shafer R W, Wehrly K, Winters M A, Mullins J I, Chesebro B, Merigan T C. Multidrug-resistant human immunodeficiency virus type 1 strains resulting from combination antiretroviral therapy. J Virol. 1996;70:1086–1090. doi: 10.1128/jvi.70.2.1086-1090.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Larder B A, Bloor S, Kemp S D, Hertogs K, Desmet R L, Miller V, Sturmer M, Staszewsky S, Ren J, Stammers K, Stuart D I, Pauwels R. A family of insertion mutations between codons 67 and 70 of human immunodeficiency virus type 1 reverse transcriptase confer multinucleoside analog resistance. Antimicrob Agents Chemother. 1999;8:1961–1967. doi: 10.1128/aac.43.8.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Merrill D P, Moonis M, Chou T-C, Hirsch M S. Lamivudine (3TC) or stavudine (d4T) in two- or three-drug combinations against HIV-1 replication in vitro. J Infect Dis. 1996;137:355–364. doi: 10.1093/infdis/173.2.355. [DOI] [PubMed] [Google Scholar]
- 7.Milazzo L, Rusconi S, Testa L, La Seta-Catamancio S, Galazzi M, Kurtagic S, Citterio P, Gianotto M, Grassini A, Adorni F, d'Arminio-Monforte A, Galli M, Moroni M. Evidence of stavudine-related phenotypic resistance among zidovudine-pretreated HIV-1-infected subjects receiving a therapeutic regimen of stavudine plus lamivudine. J Acquir Immune Defic Syndr Hum Retrovirol. 1999;22:101–103. doi: 10.1097/00042560-199909010-00013. [DOI] [PubMed] [Google Scholar]
- 8.Ross L, Johnson M, Graham N, Sheafer M, St. Clair M. The reverse transcriptase codon 69 insertion is observed in nucleoside reverse transcriptase inhibitor-experienced HIV-1-infected individuals, including those without prior or concurrent zidovudine therapy. J Hum Virol. 1999;2:290–295. [PubMed] [Google Scholar]
- 9.Rusconi S, De Pasquale M P, Milazzo L, Moscatelli G, Bulgheroni E, Citterio P, d'Arminio-Monforte A, Moroni M, Galli M. Loss of antiviral effect owing to zidovudine and lamivudine double resistance in HIV-1-infected patients in an ongoing open-label trial. Antivir Ther. 1997;2:39–46. [PubMed] [Google Scholar]
- 10.Winters M A, Cooley K I, Girard Y A, Leeve D J, Hamdan H, Katzenstein D A, Merigan T C. A 6-base pair insert in the reverse transcriptase gene of human immunodeficiency virus type 1 confers resistance to multiple nucleoside analogues. J Clin Investig. 1998;102:1769–1775. doi: 10.1172/JCI4948. [DOI] [PMC free article] [PubMed] [Google Scholar]