Abstract
Background
Previous studies have reported associations between attention-deficit/hyperactivity disorder (ADHD) and lower socioeconomic status and intelligence. We aimed to evaluate the causal directions and strengths for these associations by use of a bi-directional two-sample Mendelian randomization (MR) design.
Methods
We used summary-level data from the largest available genome-wide association studies (GWAS) to identify genetic instruments for ADHD, intelligence, and markers of socioeconomic status including the Townsend deprivation index, household income, and educational attainment. Effect estimates from individual genetic variants were combined using inverse-variance weighted regression.
Results
A genetically predicted one standard deviation (SD) increment in the Townsend deprivation index conferred an odds ratio (OR) of 5.29 (95% confidence interval (CI) 1.89–14.76) for an ADHD diagnosis (p<0.001). A genetically predicted one SD higher education level conferred an OR of 0.30 (95% CI 0.25–0.37) (p<0.001), and a genetically predicted one SD higher family income provided an OR of 0.35 (95% CI 0.25–0.49; p<0.001). The associations remained after adjustment for intelligence whereas the lower odds of an ADHD diagnosis with higher intelligence did not persist after adjustment for liability to greater educational attainment (adjusted OR 1.03, 95% CI 0.68–1.56; p=0.87). The MR analysis of the effect of ADHD on socioeconomic markers found that genetic liability to ADHD was statistically associated with each of them (p<0.001) but not intelligence. However, the average change in the socioeconomic markers per doubling of the prevalence of ADHD corresponded only to 0.05–0.06 SD changes.
Conclusions
Our results indicate that an ADHD diagnosis may be a direct and strong intelligence-independent consequence of socioeconomic related factors, whereas ADHD appears to lead only to modestly lowered socioeconomic status. Low intelligence seems not to be a major independent cause or consequence of ADHD.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12916-022-02314-3.
Keywords: Attention-deficit/hyperactivity disorder, ADHD, Socioeconomic status, Education, Intelligence, Income, Townsend deprivation index, Mendelian randomization, Gene, GWAS
Background
The diagnosis of attention-deficit/hyperactivity disorder (ADHD) has become highly prevalent during the last two decades [1], but there are considerable regional differences, both within and between countries, in the proportion of children and adolescents with this diagnosis [2–4]. These differences may be explained by true variation in the prevalence or to be attributable to cultural contexts, diagnostic resources, and practice, as well as to biological factors like genetics [5, 6]. Hitherto, there is no established clinically valid biological marker for the neuropsychiatric diagnosis of ADHD [4].
Genetics clearly contribute to ADHD. Family studies indicate a five-fold higher risk in first-degree relatives of cases, and genetic liability to an ADHD diagnosis in children and adolescents is estimated to be 70–80% [7, 8]. Boys have a higher ADHD prevalence than girls [1, 6], but the heritability is higher for girls [8]. To date, genome-wide association studies have indicated single-nucleotide polymorphism (SNP) heritability, the proportion of phenotypic variance explained by all measured and statistically significant SNPs, to be 20%, leaving considerable missing heritability, i.e., the difference between SNP versus twin heritability [9]. This is not uncommon in genome-wide association study (GWAS) research and can be explained by rare or weak genetic variants, which are only captured by very large datasets [10]. Another conceivable explanation for the missing heritability can be moderately strong genetic factors not causally related to the ADHD diagnosis per se, but rather associated with the probability of being diagnosed. Two such contributing factors may be socioeconomic status (SES) and intelligence. Different aspects of SES, such as educational attainment and income, typically display twin heritabilities of 40% [11, 12]. The heritability of intelligence increases linearly, from 40% in childhood to 80% in late adulthood but declines to about 60% after age 80 years [13]. The relative importance of SES, including parental income, education, occupation, and marital status [14] as well as intelligence, in relation to the diagnosis of ADHD has long been debated. Several longitudinal cohort studies have reported a close relationship between socioeconomic disadvantage in childhood and a later diagnosis of an attention deficit/hyperactivity disorder (ADHD) [14–18]. Meta-analyses of measures of SES and their association with ADHD indicate that children in families of low SES are on average at doubled risk of receiving an ADHD diagnosis than their peers in high SES families [14]. Specifically, parental financial difficulties, housing conditions, education, occupation, and marital status have been shown to significantly be associated with children’s likelihood to be diagnosed with ADHD [14–18]. An ADHD diagnosis is also associated with relatively poor academic achievement and work performance [18–20] as well as with moderately lower intellectual capacity [21, 22].
Due to a complex web of interrelated associations, the independence and direction of the associations between a diagnosis of ADHD with specific markers of socioeconomic status and intelligence is unclear [14]. The observational nature of most studies of ADHD’s association with SES and intelligence hampers causal inference due to potential biases from residual confounding and reverse causation.
The Mendelian randomization (MR) design can overcome such biases, thereby strengthening causal inference regarding an exposure-outcome association by leveraging genetic variants as instrumental variables for an exposure [23]. In this approach, causality is inferred from associations between genetic instrument proxies for a putative modifiable risk factor and the outcome of interest. Bidirectional MR, with analyses assessing causality in both directions, can help tease apart causal temporal directions of two related variables [24]. In this case, if low SES or lower intelligence leads to a higher risk of an ADHD diagnosis, then genetic variants associated with lower SES or lower intelligence should be related to higher risk for a diagnosis of ADHD. Conversely, if ADHD is causally related to markers of low SES or intelligence, a genetic variant associated with a higher risk for a diagnosis of ADHD should be associated with lower SES or lower estimated intelligence. One recent Mendelian randomization study showed that genetic liability to higher education was associated with a lower risk of an ADHD diagnosis independently of intelligence but an analysis in the reverse direction displayed a modestly strong association [25]. This implies that lower education contributes as a cause and not a clear consequence of ADHD. Other aspects of SES in relation to an ADHD diagnosis have not been investigated by the use of a Mendelian randomization design.
Our aim was therefore to use a bi-directional two-sample MR design to assess the associations between markers of SES or intelligence and an ADHD diagnosis.
Methods
We used a two-sample MR design in which the genetic instruments for the exposure, and the outcome are extracted from independent GWAS data sources. Our analyses were bidirectional, first assessing the causal effect of the socio-economic variables on ADHD and then investigating the reverse relationship. Each analysis used multiple genetic variants obtained from publicly available GWAS summary data [26]. None of the data sources investigated both ADHD and socioeconomic status or intelligence, and there is no known subject overlap between the two sources of data [9].
Data sources
Data regarding the Townsend index of deprivation (TID) [27], household income, educational attainment, intelligence, and ADHD were obtained from four sources.
Genetic instruments for the TID (including the components unemployment, lack of car ownership, lack of home ownership, and household overcrowding) was retrieved from the UK Biobank (n=462,464; https://gwas.mrcieu.ac.uk/datasets/ukb-b-10011/) using a z score with a mean of −1.29 and a standard deviation (SD) of 3.10. A greater TID corresponds to a lower socioeconomic status. The UK Biobank was also used for a GWAS of self-reported household income (n=397,751; https://gwas.mrcieu.ac.uk/datasets/ukb-b-7408/), calculated as the average total household income in 2006–2010 before tax reduction.
We obtained genetic instrument data for education level from the Social Science Genetic Association Consortium (n=766,345), based on longitudinal assessment of cohort participants measured at an age of at least 30 [28]. Education level was defined as the number of years of education with durations harmonized across studies according to the International Standard Classification of Education categories [28]. The sample-size-weighted mean of years of education year was 16.8 years with a SD of 4.2 years.
We used Savage et al.’s GWAS meta-analysis of intelligence (children, young adults, middle-aged and older individuals with n=269,867) [29]. Included cohorts extracted a single sum score, mean score, or factor score from a multidimensional set of cognitive performance tests in a GWAS, with the exception of the High-Intelligence/Health and Retirement Study in which a logistic regression GWAS was run with “case” status (high intelligence, top 0.03% tail in the normal population) versus controls (a population sample of unselected individuals) [29].
Finally, we included genetic data from the international Psychiatric Genomics Consortium, which involved 20,183 individuals diagnosed with ADHD and 35,191 controls [9]. This GWAS did not include UK Biobank data. There was no heterogeneity of effects when the investigators compared different types of studies [9], including those based on children, those based on adults, and those who used the International Classification of Diseases (ICD) 10 clinical diagnosis of ADHD (code F90.0), ADHD treatment, and those employing continuous quantitative population measures of ADHD-related behaviors [9]. Specifically, in this GWAS [9], the genetic instruments associated with a clinical diagnosis of ADHD and ADHD treatment were also associated with population measures of ADHD-related behaviors. A genetic correlation analysis provided additional evidence that effects were consistent across cohorts included in the analysis [9]. Results based on data collected from populations in Western Europe and North America were accordingly similar. We extracted harmonized GWAS data for all phenotypes through the MR-Base platform [30]. In the current analyses, we restricted our analysis to results based on individuals of the European ancestry.
Genetic instruments
Genetic instruments, identified at a genome-wide significance threshold of p<5×10-8, were selected from the corresponding genome-wide association studies. Linkage disequilibrium (defined as R2>0.01 or clump distance <10,000 kb) between SNPs was assessed based on the 1000 Genomes European reference panel (https://www.internationalgenome.org/). For SNPs in linkage disequilibrium, those with the strongest association with the exposure were retained. Remaining independent SNPs who had met a GWAS-wide significance threshold were used as genetic instruments. SNPs that were unavailable in an outcome dataset were replaced by suitable proxies (minimum linkage disequilibrium R2=0.8) where available. We removed SNPs without an imputed substitute as well as all palindromic SNPs. Our genetic instruments included 17 SNPs for the TID, 42 SNPs for household income, 219 SNPs for educational attainment, and 132 SNPs for intelligence. These explained 0.13%, 0.53%, 2.8%, and 3.2% of the variance in the TID, household income, educational attainment, and intelligence, respectively. For the analysis with ADHD as the exposure, we used nine (out of 12) [9] conditionally independent SNPs associated with ADHD (Additional file 1: Table S1); these SNPs explained 0.6% of the variance in ADHD.
Statistical analysis
Since our different phenotypes are linked to each other, we first illustrate these relationships by the genetic correlations of ADHD with attained education, family income, TID, and intelligence using LD score regression [31].
In our main analysis, we assessed the direction and strengths of the associations of ADHD with socioeconomic markers and intelligence in a bi-directional design. We first estimated the impact of our SES variables and intelligence on the odds of ADHD, estimating both the total effect of each variable and the direct effect independent of intelligence and SES, respectively, by use of multivariable Mendelian randomization [32]. Secondly, we examined the association in the reverse direction, i.e., the influence of ADHD on SES and intelligence.
The principal analyses (Fig. 1A) were conducted using an inverse-variance weighted (IVW) approach in a multiplicative random-effects model, which assumes that all SNPs are valid instrumental variables [26, 33] and that the estimates can be interpreted to reflect the total effect of the exposure. In addition, we used multivariable MR analysis and the inverse-variance weighted method [32] with markers for SES adjusted for intelligence, and vice versa (Fig. 1B). This method was used to estimate the independent direct causal effect of each of the exposure on the outcome [32].
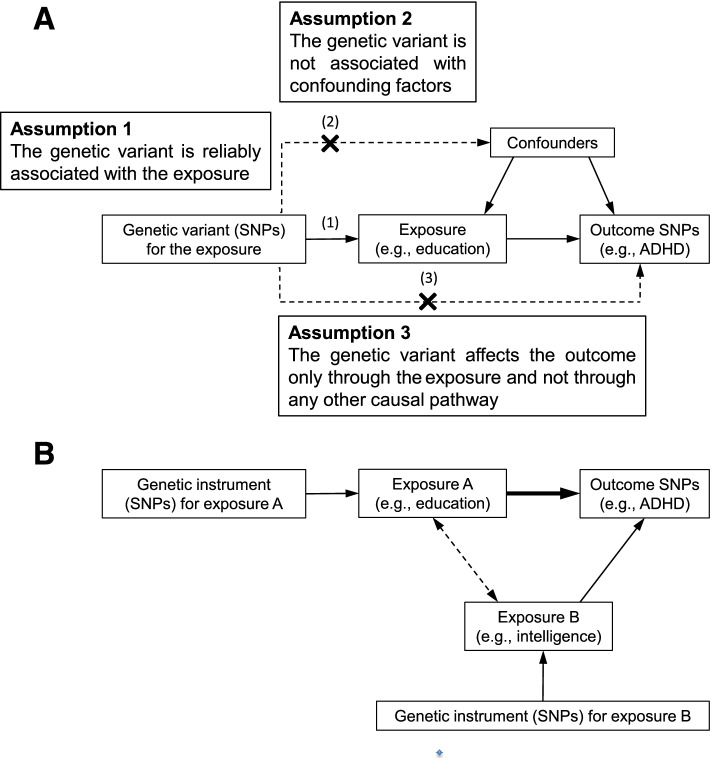
Fig. 1.
Panel A illustrates the assumptions underpinning a Mendelian randomization analysis of the association between an exposure (e.g., education) and an outcome (e.g., ADHD). SNPs indicate single-nucleotide polymorphisms. The arrows represent causal pathways. The dashed arrows represent potential causal associations between variables that would violate the Mendelian assumptions. Panel B displays one such possible violation with inclusion of an exposure B (independent exposure, mediator, or confounding factor), in our example proposed to be intelligence. One method to examine the influence of this possible violation is multivariable Mendelian randomization analysis (MVMR) and the inverse-variance weighted method [32] with markers for education adjusted for intelligence. The remaining direct causal effect of education on ADHD is illustrated by the bold arrow
We report odds ratios (ORs) of ADHD per one SD increase of genetically predicted socioeconomic marker or intelligence. For ADHD as the exposure, effect sizes are expressed as SD changes in the socioeconomic markers or intelligence per unit increase in genetically predicted log odds ratio of ADHD. A log odds ratio of one thus corresponds to an OR of 2.7, and the average change in socioeconomic markers per doubling of the prevalence of an ADHD diagnosis equals 0.693 (=loge 2) times the causal estimate on the untransformed scale [34]. All statistical tests were two-tailed. Associations that were statistically significant at a p-value <0.006 (with Bonferroni correction for 8 main outcomes) were considered to show strong evidence of association. P-values <0.05, but higher than 0.006, were regarded as showing suggestive evidence of associations, requiring confirmation.
In sensitivity analyses, the following approaches were applied: (1) the weighted median method, which provides a causal estimate if at least 50% of the weight in the analysis comes from valid instrumental variables [33]; (2) MR-Egger regression, which can detect and adjust for directional pleiotropy but has low precision [33]; (3) the Pleiotropy RESidual Sum and Outlier (MR-PRESSO) method, which can detect and adjust for horizontal pleiotropy by outlier removal [35]; (4) the contamination mixture method, which performs MR robustly and efficiently in the presence of invalid instrumental variables [36]; the (5) MRMix method, which also aims to estimate causal effects in the presence of invalid instruments [37]; the (6) Generalised Summary-data-based Mendelian Randomisation (GSMR) method, which performs MR analysis with multiple near-independent instruments to test for causal associations [38]; and the (7) MRMode, which has been proposed to provide a single causal effect estimate from multiple genetic instruments [39] but, in accordance with MR-Egger, has only moderate precision [37].
With traditional MR methods there remains a concern that valid instrument selection are often violated, leading to false positive findings through correlated horizontal pleiotropy. To avoid this bias, we adopted a new MR method as the eighth (8) sensitivity analysis, using summary effect estimates (MR-CAUSE), to differentiate correlated pleiotropy from causal effects [40]. MR-CAUSE assumes that the relationship between the genetic instrument’s effect on exposure and on outcome is a mixture of both causal and shared correlated pleiotropy. It estimates posterior distributions of a null effect, a shared effect, and a causal effect of the exposure [40, 41], and model fit comparisons are done by Δ Expected Log Pointwise Posterior Density (ELPD).
The TwoSampleMR [30], MendelianRandomization [42], MRPRESSO [35], and MR-CAUSE [40] packages were used for the statistical analyses. We restricted our analysis to participants with the European ancestry.
Results
Genetic variants with a positive effect on ADHD tended to have a negative association with intelligence and educational attainment: the genetic correlations were −0.37 (95% CI −0.44 to −0.30) and −0.51 (−0.57 to −0.45), respectively. Education and intelligence were genetically related, as expected (r=0.73; 95% CI 0.68 to 0.78)), and education was also correlated with household income (r=0.77; 95% CI 0.70 to 0.84)) and with TID (r=−0.40; 95% CI −0.46 to −0.35). Intelligence was correlated with both household income (r=0.64; 95% CI 0.56 to 0.72) and TID (r=0.20; 95% CI 0.15 to 0.24).
Total and direct effect of genetically predicted SES indicators and intelligence on ADHD
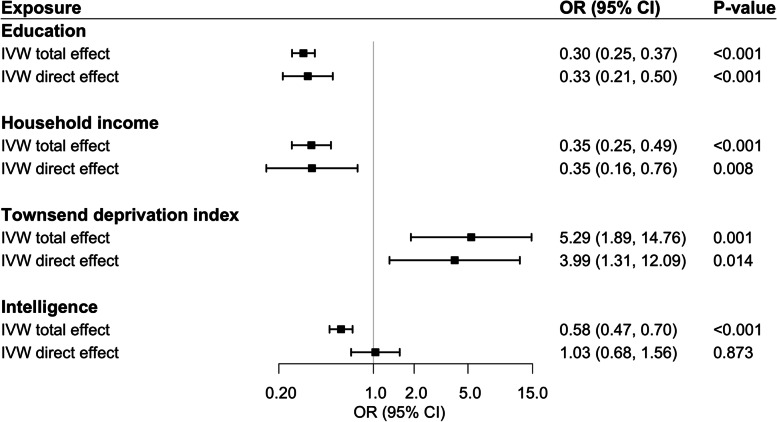
In IVW-random effect models, genetically predicted TID, household income, and educational attainment were all strongly associated with ADHD (Fig. 2). One SD higher genetically predicted duration of education conferred an OR of 0.30 (95% CI 0.25–0.37) (p<0.001), and one SD higher genetically predicted family income gave an OR of 0.35 (95% CI 0.25–0.49; p<0.001). A one SD increment in genetically predicated TID conferred an OR of 5.29 (95% confidence interval [CI] 1.89–14.76) (p<0.001) for ADHD. The direct effects, after adjustment for intelligence, were similar (Fig. 2), with ORs of 0.33 (95% CI 0.21–0.50), OR 0.35 (95% CI 0.16–0.76), and 3.99 (95% CI 1.31–12.09), respectively.
Fig. 2.
Results of the Mendelian randomization analyses of the odds of ADHD conferred by the liability for one standard deviation increase in attained educational level, household income, Townsend deprivation index, or intelligence. Ordinary IVW estimates are provided for the total effect of the exposure, and MVWR IVW estimates for the direct effect of the exposure. Estimates for SES markers are adjusted for intelligence; that for intelligence is adjusted for education
We also observed (Fig. 2) that intelligence was related to ADHD, with a somewhat more moderate OR of 0.58 (95% CI 0.47–0.70; p<0.001) per SD higher predicted intelligence. This lower odds of genetic liability of an ADHD diagnosis with genetically predicted higher intelligence did not remain after adjustment for educational attainment (OR 1.03, 95% CI 0.68–1.56; p=0.87). The estimate was also attenuated by the adjustment for household income (OR 0.71, 95% CI 0.50–0.99; p=0.04 but not by adjustment for TID (OR 0.61, 95% CI 0.50–0.74; p<0.001).
The results were largely the same in sensitivity analyses (Table 1) based on the weighted median, MR-PRESSO methods, and contamination mixture analyses. The MR-Egger analysis showed no clear evidence of horizontal pleiotropy, with the exception of that for household income (p = 0.038). The MRMix analysis indicated independent causal effects of education, intelligence, and income (but not TID) on ADHD, while all four exposures remained statistically significant causes of ADHD when data were analyzed by GSMR. We found also similar patterns with MR-Mode although with lower precision of the estimates. Sensitivity analysis by MR-CAUSE (Table 2) showed that intelligence and income, but especially education, remained as independent causes of ADHD, e.g., the causal model of education on ADHD was superior to the shared education model on ADHD.
Table 1.
Results of the Mendelian randomization sensitivity analyses associating the liability for one standard deviation increase in attained educational level, household income, Townsend deprivation index, and intelligence with the odds of ADHD
| Exposure | Method | OR (95% C)) | p-value |
|---|---|---|---|
| Education (219 SNPs) | Weighted median | 0.34 (0.27, 0.43) | <0.001 |
| MR-Egger | 0.39 (0.18, 0.85) | 0.018 | |
| MR-PRESSO (3 outliers) | 0.30 (0.25, 0.36) | <0.001 | |
| Contamination mixture | 0.23 (0.17, 0.32) | <0.001 | |
| MR-Mode | 0.58 (0.27, 1.26) | 0.168 | |
| MR-Mix | 0.37 (0.23, 0.59) | <0.001 | |
| GSMR | 0.29 (0.25–0.34) | <0.001 | |
| Household income (42 SNPs) | Weighted median | 0.49 (0.34, 0.72) | <0.001 |
| MR-Egger | 1.64 (0.38, 7.00) | 0.508 | |
| MR-PRESSO (1 outlier) | 0.38 (0.29, 0.51) | <0.001 | |
| Contamination mixture | 0.37 (0.25, 0.54) | <0.001 | |
| MR-Mode | 0.69 (0.34, 1.40) | 0.308 | |
| MR-Mix | 0.37 (0.23, 0.60) | <0.001 | |
| GSMR | 0.28 (0.21, 0.38) | <0.001 | |
| Townsend deprivation index (17 SNPs) | Weighted median | 5.51 (2.22, 13.71) | <0.001 |
| MR-Egger | 0.04 (0.00, 229.4) | 0.482 | |
| MR-PRESSO (2 outliers) | 4.86 (2.11, 11.19) | 0.002 | |
| Contamination mixture | 8.25 (3.78, 27.39) | 0.001 | |
| MR-Mode | 8.94 (1.58, 50.69) | 0.025 | |
| MR-Mix | 1.00 (1.00, 1.00) | 0.99 | |
| GSMR | 4.92 (2.95, 8.21) | <0.001 | |
| Intelligence (132 SNPs) | Weighted median | 0.59 (0.48, 0.73) | <0.001 |
| MR-Egger | 0.68 (0.27, 1.72) | 0.416 | |
| MR-PRESSO (4 outliers) | 0.58 (0.48, 0.69) | <0.001 | |
| Contamination mixture | 0.53 (0.44, 0.62) | <0.001 | |
| MR-Mode | 0.59 (0.37, 0.97) | 0.037 | |
| MR-Mix | 0.50 (0.31–0.81) | 0.005 | |
| GSMR | 0.53 (0.46–0.60) | <0.001 |
Table 2.
MR-CAUSE analysis associating the liability for one standard deviation increase in attained educational level, household income, Townsend deprivation index, and intelligence with the odds of ADHD
| Model 1a | Model 2a | Δ ELPDb | s.e. Δ ELPD | z-score | p-value |
|---|---|---|---|---|---|
| Education (SNPs=3733) | |||||
| Null | Sharing | −120.0 | 13 | −8.7 | 2.0e−18 |
| Null | Causal | −120.0 | 14 | −8.6 | 3.0e−18 |
| Sharing | Causal | −7.3 | 1.2 | −6.2 | 2.1e−10 |
| Income (SNPs =73) | |||||
| Null | Sharing | −3.6 | 1.5 | −2.5 | 0.007 |
| Null | Causal | −9.7 | 3.4 | −2.9 | 0.0022 |
| Sharing | Causal | −6.1 | 2.1 | −3.0 | 0.0015 |
| TDI (SNPs =27) | |||||
| Null | Sharing | −0.31 | 0.45 | −0.70 | 0.24 |
| Null | Causal | −1.60 | 2.2 | −0.74 | 0.23 |
| Sharing | Causal | −1.30 | 1.8 | −0.73 | 0.23 |
| Intelligence (SNPs=2945) | |||||
| Null | Sharing | −25.0 | 5.7 | −4.4 | 5.5e−06 |
| Null | Causal | −31.0 | 7.1 | −4.4 | 5.2e−06 |
| Sharing | Causal | −5.9 | 1.4 | −4.1 | 1.8e−05 |
aModel 1 and Model 2 refer to the models being compared (null, sharing, or causal)
bModel fit is measured by Δ Expected Log Pointwise Posterior Density (ELPD); Negative values indicate that model 2 is a better fit
Total effect of genetic liability to ADHD on markers of socio-economic status and intelligence
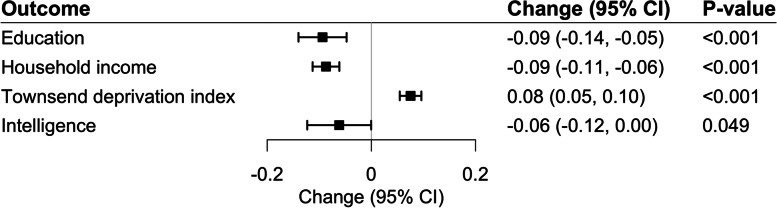
IVW-random effect analysis in the reverse direction indicated that genetic predisposition to ADHD was statistically significantly associated with SES variables but not with intelligence (Fig. 3). However, the strength of the associations in this direction were modest. One log odds ratio of genetically predicted ADHD conferred a 0.09 SD (95% CI 0.05–0.14) lower educational attainment (corresponding to 4.5 months lower education duration), a 0.09 SD (95% CI 0.06–0.11) lower household income, and a 0.08 SD (95% CI 0.05–0.10) higher TID (all p<0.001). Expressed in another way, the average change in adverse socioeconomic markers per genetically predicted doubling in the prevalence of the ADHD diagnosis corresponded to only 0.05–0.06 SD units lower SES measures. The modest impact of ADHD on SES and intelligence can theoretically be a consequence of weak instrument bias given that we only included 9 ADHD SNPs in our analyses. However, the F-statistics with values between 181 and 514 do not indicate such bias.
Fig. 3.
Results of the Mendelian randomization IVW analysis for one standard deviation difference in attained educational level, household income, Townsend deprivation index, or intelligence from genetic liability to ADHD (9 SNPs)
The results were similar in sensitivity analyses based on the weighted median, MR-PRESSO methods, and contamination mixture approach (Table 3). There was limited evidence of unbalanced and correlated horizontal pleiotropy as estimated by MR-Egger regression (p-values from 0.11 to 0.78) as also indicated by analyses using MR-Mode, MRMix (Table 3), and MR-CAUSE (Table 4). The number of SNPs for ADHD (n=9) precluded an analysis by use of GSMR.
Table 3.
Results of the Mendelian randomization sensitivity analyses using summary-level data from genetic liability to ADHD (9 SNPs) to a standard deviation change in attained educational level, household income, Townsend deprivation index, and intelligence
| Outcome | Method | OR (95% CI) | p-value |
|---|---|---|---|
| Education | Weighted median | −0.07 (−0.1, −0.03) | <0.001 |
| MR-Egger | 0.03 (−0.15, 0.21) | 0.760 | |
| MR-PRESSO (4 outliers) | −0.06 (−0.10, −0.02) | 0.046 | |
| Contamination mixture | −0.12 (−0.19, 0.00) | <0.001 | |
| MR-Mode | −0.08 (−0.15, 0.00) | 0.078 | |
| MR-Mix | −0.15 (−0.21, −0.09) | <0.001 | |
| GSMR (<10 SNPs) | NA | ||
| Household income | IVW-random effects | −0.09 (−0.11, −0.06) | <0.001 |
| Weighted median | −0.09 (−0.12, −0.05) | <0.001 | |
| MR-Egger | −0.03 (−0.14, 0.07) | 0.564 | |
| MR-PRESSO (0 outlier) | NA | NA | |
| Contamination mixture | −0.09 (−0.11, −0.07) | <0.001 | |
| MR-Mode | −0.08 (−0.14, −0.02) | 0.026 | |
| MR-Mix | −0.04 (−0.06, −0.02) | 0.001 | |
| GSMR (<10 SNPs) | NA | ||
| Townsend deprivation index | Weighted median | 0.06 (0.04, 0.09) | <0.001 |
| MR-Egger | 0.01 (−0.07, 0.09) | 0.762 | |
| MR-PRESSO (0 outliers) | NA | NA | |
| Contamination mixture | 0.08 (0.05, 0.09) | <0.001 | |
| MR-Mode | 0.07 (0.03, 0.10) | 0.003 | |
| MR-Mix | 0.07 (−0.02, 0.16) | 0.132 | |
| GSMR (<10 SNPs) | NA | ||
| Intelligence | Weighted median | −0.05 (−0.10, −0.01) | 0.030 |
| MR-Egger | −0.24 (−0.49, 0.01) | 0.108 | |
| MR-PRESSO (4 outliers) | −0.06 (−0.13, 0.01) | 0.149 | |
| Contamination mixture | −0.11 (−0.15, −0.08) | 0.020 | |
| MR-Mode | −0.10 (−0.21, 0.02) | 0.130 | |
| MR-Mix | −0.14 (−0.22, −0.06) | 0.001 | |
| GSMR (<10 SNPs) | NA |
NA not applicable given that the genetic instrument for the ADHD exposure included only 9 SNPs, GSMR is not a suitable method
Table 4.
MR-CAUSE analysis displaying that the sharing model has a significantly worse fit than the causal model for the association between ADHD and the outcomes education, income, TDI, as well as intelligence, respectively
| Model 1a | Model 2a | Δ ELPDb | s.e. Δ ELPD | z-score | p-value |
|---|---|---|---|---|---|
| Education (SNPs=1430) | |||||
| Null | Sharing | −6.9 | 2.2 | −3.2 | 0.00072 |
| Null | Causal | −13.0 | 4 | −3.3 | 0.00041 |
| Sharing | Causal | −6.4 | 1.8 | −3.5 | 0.00023 |
| Income (SNPs=1430) | |||||
| Null | Sharing | −3.1 | 1.5 | −2.1 | 0.018 |
| Null | Causal | −8.2 | 3.6 | −2.3 | 0.011 |
| Sharing | Causal | −5.1 | 2.1 | −2.4 | 0.008 |
| TDI (SNPs=1430) | |||||
| Null | Sharing | −5.2 | 2.2 | −2.4 | 0.0092 |
| Null | Causal | −10.0 | 4.1 | −2.5 | 0.007 |
| Sharing | Causal | −4.8 | 1.9 | −2.5 | 0.0063 |
| Intelligence (SNPs=1430) | |||||
| Null | Sharing | −0.91 | 0.77 | −1.2 | 0.12 |
| Null | Causal | −4.60 | 2.7 | −1.7 | 0.043 |
| Sharing | Causal | −3.70 | 1.9 | −1.9 | 0.027 |
aModel 1 and Model 2 refer to the models being compared (null, sharing, or causal)
bModel fit is measured by Δ Expected Log Pointwise Posterior Density (ELPD); Negative values indicate that model 2 is a better fit
Discussion
Previously, the independence and direction of the associations between a diagnosis of ADHD with different markers of socioeconomic and intelligence have been unclear. Our results based on a bidirectional two-sample MR-analysis design show that a genetic predisposition to socioeconomic position including attained educational level, household income, and TID has a direct and strong intelligence-independent association with ADHD. Genetically predicated intelligence per se was not independently associated with ADHD. Genetic liability to ADHD was only modestly inversely associated with SES measures and not clearly with lower intelligence.
Using the bidirectional MR design, our study provides a deeper understanding of previous observational findings [14–18, 29] by clarifying the directional strengths of the associations of ADHD with different aspects of SES as well as with intelligence. Our study also corroborates findings from a recent Mendelian randomization study [25] that reported a strong intelligence-independent association between genetic liability to higher education with lower odds of ADHD, and only a moderately strong association in the reverse direction. We found that the impact of several different aspects of socioeconomic factors on the diagnosis of ADHD was considerably stronger than the influence of an ADHD diagnosis on attained educational level, household income, and TID. The findings also indicate that intelligence is not a major cause or consequence of an ADHD diagnosis, associations suggested in previous observational analyses [4, 43]. It should be emphasized that we are not using the actual measures but genetic instruments for lifetime SES, intelligence, and ADHD with the sequential ordering of events estimated by the bi-directional approach rather than by a timed course of events for the actual exposures and outcomes.
One possible explanation for the fact that we see that SES-related factors are strongly linked to the receipt of an ADHD diagnosis may be that social vulnerability, lack of cultural, and economic capital or immaturity can be a basis for difficulty concentrating and sitting still, e.g., in a school environment. Lack of cultural capital, in the form of the parents’ level of education, can be manifested as shortcomings in language competence, for example. Parental SES is related to language development in multiple domains throughout both childhood and adulthood [44]. Children from lower SES homes show on average lower sustained levels of language and communicative skill than children from higher SES homes, differences beginning in infancy [44]. Low maternal education in general, single parenthood, and social welfare support are thought collectively to account for more than half of all ADHD medication prescriptions in Sweden [45], a country with a tax-supported health care system. In addition, children and young people from more deprived backgrounds in other areas Europe are also more likely to receive medication for ADHD [45, 46].
The resourceful family may also have greater opportunity to protect, stimulate, and train their children to become physically and mentally competent and thus avoid an ADHD diagnosis [47]. In addition to those possible mechanisms for low SES to lead to a diagnosis of ADHD, there are also non-causal possibilities for the association. A possible partial explanation might be that resourceful families, and individuals are reluctant to carry out the investigation leading to an ADHD diagnosis since a diagnosis limits rather than improves the individual’s opportunity for successful choices in life. This may involve a requirement for a medical certificate to obtain a driving license or disqualification from the possibility to obtain specific occupations. An additional aspect may be that an ADHD diagnosis fits poorly with the social practices in which a controlled temper is expected to accompany the physical and mental abilities that characterize a cultivated person [47]. A controlled body and mind with an ability for sustained attention is a classic aristocratic ideal with historical roots from antiquity [48–50].
By some measures, children of lower socioeconomic status score more than 2 years behind their more well-off peers on standardized language development tests by the time they enter school [51]. As a result, an individual from a lower status family may have difficulty following longer instructions, trouble getting started with tasks, and problems with attention in general. A reasonable theoretic interpretation is that these difficulties can lead to frustration and a range of behaviors that comprise ADHD.
Indirect support for the view that immaturity can affect the likelihood to being diagnosed with ADHD, is the fact that children in Norway born in October through December are at 50% higher risk of being diagnosed and treated for ADHD compared with children born the same year but in January through March [52]. Children who are almost a year younger tend also to appear more immature than their classmates, which influences both their academic and physical performance. The youngest children in a grade are often developmentally less mature and are more likely to behave more inattentively, impulsively, and hyperactively than their older classmates [53]. Furthermore, the association between SES with school readiness and maturity is well established and globally observed [54, 55]. Thus, for some ADHD cases, there conceivably could have been a tendency to confuse immaturity with ADHD, a possibility that can be supported by the fact that half of the people who were diagnosed with ADHD as children do not sustain the diagnosis in young adulthood [4].
Collectively, our findings of a strong 3–5 fold change in the odds of ADHD diagnosis per SD change in genetically predicated various dimensions of SES raise questions about whether the current criteria for diagnosis are sufficiently accurate and culturally generalizable tools to correctly identify ADHD, a descriptive diagnosis for which there is not yet any clinically valid biological marker. We definitely acknowledge that ADHD is regarded as a highly debilitating and costly disease [4], but our results suggest that refinement of the diagnosis may be warranted since there may exist subgroups with the diagnosis, especially in those with low SES.
In the Psychiatric Genomics Consortium GWAS, genetic markers for ADHD were identified either by an ADHD diagnosis or by having been prescribed ADHD medications, though without heterogeneity [9]. Therefore, our results can be generalized to both these categories. This is of interest since treatment with ADHD medication alone seems similarly effective in children from families with low and high socioeconomic status whereas combined medication and behavioral treatment showed a superior effect only in children from educated families [56]. Evidence of treatment effects in placebo-controlled randomized clinical trials with amphetamine-like compounds, average a 0.5–0.8 SD reduction in ADHD symptom scoring [4, 57]. A comparable treatment effect on similar measured modalities has also been observed in placebo-controlled trials in healthy young adult volunteers [58].
Strengths and limitations
Using genetic data from large-scale GWASs and genetic consortia, the present study is the first to comprehensively and jointly investigate the causal effects of different aspects of SES and intelligence on the diagnosis of ADHD and vice versa. In addition, we disentangled the independent effect of socioeconomic factors from intelligence using a multivariable MR approach. The analyses were based on data from individuals of the European ancestry thereby limiting the potential for population stratification bias. However, this restriction limits the transferability of the present findings to populations of the non-European ancestries.
Our genetic instruments for social class were not strong, as measured by the explained variance. However, this potential limitation can be regarded of less importance given our two-sample design, a large number of SNPs in the SES instrument variables, and large sample sizes [59, 60]. In any case, any bias due to weak instruments discovered in non-overlapping cohorts will be in the direction of the null [59, 60], supporting our findings of the associations of socio-economic factors leading to a diagnosis of ADHD.
Another conceivable serious limitation of the present study is the possibility of unbalanced horizontal pleiotropy from genetic variants acting through several different biological pathways. Thus, an alternative underlying causal explanations for the SES impact of on the risk of ADHD diagnosis might be factors linked to low SES such as parental mental health, substance abuse, and maternal smoking during pregnancy [14]. Carefully conducted studies have nevertheless not shown a major impact of maternal smoking or substance abuse on ADHD diagnosis [61, 62], and our findings in several different sensitivity analyses do not indicate major pleiotropic or genetically correlated influences that could explain the impact of genetic liabilities of SES on ADHD.
Non-genetic familial influences could not directly be assessed in the present MR study and for obvious reasons, some SES genetic liability markers in our study (household income, car ownership, educational level, and unemployment) pertain to the parent’s circumstances when a child receives an ADHD diagnosis. The genetic liability of low or high SES still pertains to the child. Nonetheless, our design precludes the evaluation of timing of exposure effects and induction periods on the occurrence of the outcomes. The strength of the associations from the analyses in different directions are not directly comparable, but the interpretation of the overall pattern was straightforward. Since identification of socioeconomic patterns with specific ADHD subtypes was not in the scope of our study, we suggest refined evaluation of different disease development pathways between SES and ADHD.
Conclusions
We conclude that there is a strong direct impact of genetically predicted household income, educational attainment, and social deprivation index on the risk of an ADHD diagnosis. Lower intelligence seemed not to be a strong independent cause or a consequence of ADHD. The associated effect of genetic liability of ADHD on genetically predicted socioeconomic markers were modest.
Supplementary Information
Additional file 1: Table S1. Data sources for the genetic instruments.
Acknowledgements
The authors would like to thank the genetic consortia making summary-level data from their respective consortium publicly available.
Abbreviations
- ADHD
Attention-deficit/hyperactivity disorder
- SNP
Single-nucleotide polymorphism
- GWAS
Genome-wide association study
- SES
Socioeconomic status
- MR
Mendelian randomization
- TID
Townsend index of deprivation
- ICD
International Classification of Diseases
- IVW
Inverse-variance weighted
- ORs
Odds ratios
- MR-PRESSO
Pleiotropy RESidual Sum and Outlier
- GSMR
Generalised Summary-data-based Mendelian Randomisation
- ELPD
Expected Log Pointwise Posterior Density
Authors’ contributions
Study concept and design: MM, KM; acquisition, analysis, or interpretation of data: MM, SY, HM, LB, JAB, SL, KM; drafting of the manuscript: MM, KM; critical revision of the manuscript for important intellectual content: MM, SY, HM, LB, JAB, SL, KM; statistical analysis and verification of the underlying data: SY, SL. The authors read and approved the final manuscript.
Funding
Open access funding provided by Uppsala University. The study was supported by grants from the Swedish Research Council (grant no 2017-00644, 2017-06100 and 2019-01291). The funder of the study had no role in the study design, data collection, data analysis, data interpretation, writing of the report, or the decision to submit the article for publication.
Availability of data and materials
All the data supporting the conclusions of this article are included within the article and its supplementary file.
Declarations
Ethics approval and consent to participate
Our study is a secondary analysis of publicly available data. Informed consent was sought for all participants per the original GWAS protocols, and all ethical approvals for the GWAS were obtained by the original GWAS authors.
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Footnotes
Madeleine Michaëlsson and Shuai Yuan shared first authorship of this study.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Madeleine Michaëlsson and Shuai Yuan shared first authors.
References
- 1.Xu G, Strathearn L, Liu B, Yang B, Bao W. Twenty-year trends in diagnosed attention-deficit/hyperactivity disorder among US children and adolescents, 1997-2016. JAMA Netw Open. 2018;1(4):e181471. doi: 10.1001/jamanetworkopen.2018.1471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Deberdt W, Thome J, Lebrec J, Kraemer S, Fregenal I, Ramos-Quiroga JA, et al. Prevalence of ADHD in nonpsychotic adult psychiatric care (ADPSYC): a multinational cross-sectional study in Europe. BMC Psychiatry. 2015;15:242. doi: 10.1186/s12888-015-0624-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Thomas R, Sanders S, Doust J, Beller E, Glasziou P. Prevalence of attention-deficit/hyperactivity disorder: a systematic review and meta-analysis. Pediatrics. 2015;135(4):e994–1001. doi: 10.1542/peds.2014-3482. [DOI] [PubMed] [Google Scholar]
- 4.Posner J, Polanczyk GV, Sonuga-Barke E. Attention-deficit hyperactivity disorder. Lancet. 2020;395(10222):450–462. doi: 10.1016/S0140-6736(19)33004-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Polanczyk G, de Lima MS, Horta BL, Biederman J, Rohde LA. The worldwide prevalence of ADHD: a systematic review and metaregression analysis. Am J Psychiatry. 2007;164(6):942–948. doi: 10.1176/ajp.2007.164.6.942. [DOI] [PubMed] [Google Scholar]
- 6.Rydell M, Lundstrom S, Gillberg C, Lichtenstein P, Larsson H. Has the attention deficit hyperactivity disorder phenotype become more common in children between 2004 and 2014? Trends over 10 years from a Swedish general population sample. J Child Psychol Psychiatry. 2018;59(8):863–871. doi: 10.1111/jcpp.12882. [DOI] [PubMed] [Google Scholar]
- 7.Faraone SV, Perlis RH, Doyle AE, Smoller JW, Goralnick JJ, Holmgren MA, et al. Molecular genetics of attention-deficit/hyperactivity disorder. Biol Psychiatry. 2005;57(11):1313–1323. doi: 10.1016/j.biopsych.2004.11.024. [DOI] [PubMed] [Google Scholar]
- 8.Martin J, Walters RK, Demontis D, Mattheisen M, Lee SH, Robinson E, et al. A genetic investigation of sex bias in the prevalence of attention-deficit/hyperactivity disorder. Biol Psychiatry. 2018;83(12):1044–1053. doi: 10.1016/j.biopsych.2017.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Demontis D, Walters RK, Martin J, Mattheisen M, Als TD, Agerbo E, et al. Discovery of the first genome-wide significant risk loci for attention deficit/hyperactivity disorder. Nat Genet. 2019;51(1):63–75. doi: 10.1038/s41588-018-0269-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yengo L, Sidorenko J, Kemper KE, Zheng Z, Wood AR, Weedon MN, et al. Meta-analysis of genome-wide association studies for height and body mass index in approximately 700000 individuals of European ancestry. Hum Mol Genet. 2018;27(20):3641–3649. doi: 10.1093/hmg/ddy271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hyytinen A, Ilmakunnas P, Johansson E, Toivanen O. Heritability of lifetime earnings. J Econ Inequal. 2019;17(3):319–335. [Google Scholar]
- 12.Silventoinen K, Jelenkovic A, Sund R, Latvala A, Honda C, Inui F, et al. Genetic and environmental variation in educational attainment: an individual-based analysis of 28 twin cohorts. Sci Rep. 2020;10(1):12681. doi: 10.1038/s41598-020-69526-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Plomin R, Deary IJ. Genetics and intelligence differences: five special findings. Mol Psychiatry. 2015;20(1):98–108. doi: 10.1038/mp.2014.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Russell AE, Ford T, Williams R, Russell G. The association between socioeconomic disadvantage and attention deficit/hyperactivity disorder (ADHD): a systematic review. Child Psychiatry Hum Dev. 2016;47(3):440–458. doi: 10.1007/s10578-015-0578-3. [DOI] [PubMed] [Google Scholar]
- 15.Russell AE, Ford T, Russell G. Socioeconomic associations with ADHD: findings from a mediation analysis. PLoS One. 2015;10(6):e0128248. doi: 10.1371/journal.pone.0128248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Choi Y, Shin J, Cho KH, Park E-C. Change in household income and risk for attention deficit hyperactivity disorder during childhood: a nationwide population-based cohort study. J Epidemiol. 2017;27(2):56–62. doi: 10.1016/j.je.2016.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Torvik FA, Eilertsen EM, McAdams TA, Gustavson K, Zachrisson HD, Brandlistuen R, et al. Mechanisms linking parental educational attainment with child ADHD, depression, and academic problems: a study of extended families in The Norwegian Mother, Father and Child Cohort Study. J Child Psychol Psychiatry. 2020;61(9):1009–1018. doi: 10.1111/jcpp.13197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Miller LL, Gustafsson HC, Tipsord J, Song M, Nousen E, Dieckmann N, et al. Is the association of ADHD with socio-economic disadvantage explained by child comorbid externalizing problems or parent ADHD? J Abnorm Child Psychol. 2018;46(5):951–963. doi: 10.1007/s10802-017-0356-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Harpin VA. The effect of ADHD on the life of an individual, their family, and community from preschool to adult life. Arch Dis Child. 2005;90(Suppl 1):i2–i7. doi: 10.1136/adc.2004.059006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kuriyan AB, Pelham WE, Jr, Molina BS, Waschbusch DA, Gnagy EM, Sibley MH, et al. Young adult educational and vocational outcomes of children diagnosed with ADHD. J Abnorm Child Psychol. 2013;41(1):27–41. doi: 10.1007/s10802-012-9658-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bridgett DJ, Walker ME. Intellectual functioning in adults with ADHD: a meta-analytic examination of full scale IQ differences between adults with and without ADHD. Psychol Assess. 2006;18(1):1–14. doi: 10.1037/1040-3590.18.1.1. [DOI] [PubMed] [Google Scholar]
- 22.Usami M. Functional consequences of attention-deficit hyperactivity disorder on children and their families. Psychiatry Clin Neurosci. 2016;70(8):303–317. doi: 10.1111/pcn.12393. [DOI] [PubMed] [Google Scholar]
- 23.Stephen Burgess SGT. Mendelian randomization: methods for using genetic variants in causal estimation: Chapman and Hall/CRC; 2015.
- 24.Zheng J, Baird D, Borges MC, Bowden J, Hemani G, Haycock P, et al. Recent developments in mendelian randomization studies. Curr Epidemiol Rep. 2017;4(4):330–345. doi: 10.1007/s40471-017-0128-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dardani C, Riglin L, Leppert B, Sanderson E, Rai D, Howe LD, et al. Is genetic liability to ADHD and ASD causally linked to educational attainment? Int J Epidemiology. 2022;50(6):2011–23. 10.1093/ije/dyab107. [DOI] [PMC free article] [PubMed]
- 26.Burgess S, Butterworth A, Thompson SG. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol. 2013;37(7):658–665. doi: 10.1002/gepi.21758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Townsend P, Phillimore P, Beattie A. Health and deprivation: inequality and the north. London: Croom Helm; 1987. [Google Scholar]
- 28.Lee JJ, Wedow R, Okbay A, Kong E, Maghzian O, Zacher M, et al. Gene discovery and polygenic prediction from a genome-wide association study of educational attainment in 1.1 million individuals. Nat Genet. 2018;50(8):1112–1121. doi: 10.1038/s41588-018-0147-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Savage JE, Jansen PR, Stringer S, Watanabe K, Bryois J, de Leeuw CA, et al. Genome-wide association meta-analysis in 269,867 individuals identifies new genetic and functional links to intelligence. Nat Genet. 2018;50(7):912–919. doi: 10.1038/s41588-018-0152-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hemani G, Zheng J, Elsworth B, Wade KH, Haberland V, Baird D, Laurin C, Burgess S, Bowden J, Langdon R, Tan VY, Yarmolinsky J, Shihab HA, Timpson NJ, Evans DM, Relton C, Martin RM, Smith GD, Gaunt TR, Haycock PC. The MR-Base platform supports systematic causal inference across the human phenome. Elife. 2018;7:e34408. 10.7554/eLife.34408. [DOI] [PMC free article] [PubMed]
- 31.Bulik-Sullivan B, Finucane HK, Anttila V, Gusev A, Day FR, Loh PR, et al. An atlas of genetic correlations across human diseases and traits. Nat Genet. 2015;47(11):1236–1241. doi: 10.1038/ng.3406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Burgess S, Thompson SG. Multivariable Mendelian randomization: the use of pleiotropic genetic variants to estimate causal effects. Am J Epidemiol. 2015;181(4):251–260. doi: 10.1093/aje/kwu283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Burgess S, Bowden J, Fall T, Ingelsson E, Thompson SG. Sensitivity analyses for robust causal inference from Mendelian randomization analyses with multiple genetic variants. Epidemiology. 2017;28(1):30–42. doi: 10.1097/EDE.0000000000000559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Burgess S, Labrecque JA. Mendelian randomization with a binary exposure variable: interpretation and presentation of causal estimates. Eur J Epidemiol. 2018;33(10):947–952. doi: 10.1007/s10654-018-0424-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Verbanck M, Chen CY, Neale B, Do R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet. 2018;50(5):693–698. doi: 10.1038/s41588-018-0099-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Burgess S, Foley CN, Allara E, Staley JR, Howson JMM. A robust and efficient method for Mendelian randomization with hundreds of genetic variants. Nat Commun. 2020;11(1):376. doi: 10.1038/s41467-019-14156-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Qi G, Chatterjee N. Mendelian randomization analysis using mixture models for robust and efficient estimation of causal effects. Nat Commun. 2019;10(1):1941. doi: 10.1038/s41467-019-09432-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhu Z, Zheng Z, Zhang F, Wu Y, Trzaskowski M, Maier R, et al. Causal associations between risk factors and common diseases inferred from GWAS summary data. Nat Commun. 2018;9(1):224. doi: 10.1038/s41467-017-02317-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hartwig FP, Davey Smith G, Bowden J. Robust inference in summary data Mendelian randomization via the zero modal pleiotropy assumption. Int J Epidemiol. 2017;46(6):1985–1998. doi: 10.1093/ije/dyx102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Morrison J, Knoblauch N, Marcus JH, Stephens M, He X. Mendelian randomization accounting for correlated and uncorrelated pleiotropic effects using genome-wide summary statistics. Nat Genet. 2020;52(7):740–747. doi: 10.1038/s41588-020-0631-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mitchell BL, Thorp JG, Evans DM, Nyholt DR, Martin NG, Lupton MK. Exploring the genetic relationship between hearing impairment and Alzheimer's disease. Alzheimers Dement (Amst) 2020;12(1):e12108. doi: 10.1002/dad2.12108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yavorska OO, Burgess S. MendelianRandomization: an R package for performing Mendelian randomization analyses using summarized data. Int J Epidemiol. 2017;46:1734–1739. doi: 10.1093/ije/dyx034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pievsky MA, McGrath RE. The neurocognitive profile of attention-deficit/hyperactivity disorder: a review of meta-analyses. Arch Clin Neuropsychol. 2018;33(2):143–157. doi: 10.1093/arclin/acx055. [DOI] [PubMed] [Google Scholar]
- 44.Hoff ER, KM. Language development: influence of socio-economic status. In: Wright J, editor. International Encyclopedia of the Social & Behavioral Sciences: Elsevier; 2015. p. 324–8.
- 45.Hjern A, Weitoft GR, Lindblad F. Social adversity predicts ADHD-medication in school children--a national cohort study. Acta Paediatr. 2010;99(6):920–924. doi: 10.1111/j.1651-2227.2009.01638.x. [DOI] [PubMed] [Google Scholar]
- 46.Nunn SPT, Kritsotakis EI, Harpin V, Parker J. Social gradients in the receipt of medication for attention-deficit hyperactivity disorder in children and young people in Sheffield. BJPsych Open. 2020;6(2):e14. doi: 10.1192/bjo.2019.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bourdieu P. Distinction : a social critique of the judgement of taste. Cambridge: Harvard University Press; 1984. p. xiv, 613. [Google Scholar]
- 48.Iamblichus, Dillon JM, Hershbell JP. On the pythagorean way of life. Texts and translations. Atlanta: Scholars Press; 1991. pp. 31–83. [Google Scholar]
- 49.Huffman CA, Philolaus. Philolaus of croton: pythagorean and presocratic: a commentary on the fragments and testimonia with interpretive essays. Cambridge and New York: Cambridge University Press; 1993. p. 44. [Google Scholar]
- 50.Zhmud′ LIA, Windle K, Ireland R. Pythagoras and the early pythagoreans. 1. Oxford: Oxford University Press; 2012. p. 175. [Google Scholar]
- 51.Lee VE, Burkam DT. Inequality at the starting gate: social background differences in achievement as children begin school. Washington, D.C.: Economic Policy Institute; 2002. p. ix, 102. [Google Scholar]
- 52.Karlstad O, Furu K, Stoltenberg C, Haberg SE, Bakken IJ. ADHD treatment and diagnosis in relation to children's birth month: nationwide cohort study from Norway. Scandinavian J Public Health. 2017;45(4):343–349. doi: 10.1177/1403494817708080. [DOI] [PubMed] [Google Scholar]
- 53.Diefenbach C, Schmidt MF, Huss M, König J, Urschitz MS, ikidS Study Group. Age at school entry and reported symptoms of attention-deficit/hyperactivity in first graders: results of the prospective cohort study ikidS. Eur Child Adolesc Psychiatry. 2021. 10.1007/s00787-021-01813-7. Online ahead of print. [DOI] [PMC free article] [PubMed]
- 54.Isaacs JB, Magnuson KA. Income and education as predictors of children's school readiness. The Social Genome Project. 2011. [Google Scholar]
- 55.Ren L, Hu BY, Zhang X. Disentangling the relations between differentcomponents of family socioeconomic status and Chinese preschoolers’ school readiness. Fam Process. 2020;60(1):216–234. doi: 10.1111/famp.12534. [DOI] [PubMed] [Google Scholar]
- 56.Rieppi R, Greenhill LL, Ford RE, Chuang S, Wu M, Davies M, et al. Socioeconomic status as a moderator of ADHD treatment outcomes. J Am Acad Child Adolesc Psychiatry. 2002;41(3):269–277. doi: 10.1097/00004583-200203000-00006. [DOI] [PubMed] [Google Scholar]
- 57.Pereira Ribeiro J, Arthur EJ, Gluud C, Simonsen E, Storebo OJ. Does methylphenidate work in children and adolescents with attention deficit hyperactivity disorder? Pediatr Rep. 2021;13(3):434–443. doi: 10.3390/pediatric13030050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Repantis D, Bovy L, Ohla K, Kuhn S, Dresler M. Cognitive enhancement effects of stimulants: a randomized controlled trial testing methylphenidate, modafinil, and caffeine. Psychopharmacology. 2021;238(2):441–451. doi: 10.1007/s00213-020-05691-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Pierce BL, Burgess S. Efficient design for Mendelian randomization studies: subsample and 2-sample instrumental variable estimators. Am J Epidemiol. 2013;178(7):1177–1184. doi: 10.1093/aje/kwt084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Burgess S, Davies NM, Thompson SG. Bias due to participant overlap in two-sample Mendelian randomization. Genet Epidemiol. 2016;40(7):597–608. doi: 10.1002/gepi.21998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Thapar A, Rice F. Family-based designs that disentangle inherited factors from pre- and postnatal environmental exposures: in vitro fertilization, discordant sibling pairs, maternal versus paternal comparisons, and adoption designs. Cold Spring Harb Perspect Med. 2021;11(3):a038877. 10.1101/cshperspect.a038877. [DOI] [PMC free article] [PubMed]
- 62.Treur JL, Demontis D, Smith GD, Sallis H, Richardson TG, Wiers RW, et al. Investigating causality between liability to ADHD and substance use, and liability to substance use and ADHD risk, using Mendelian randomization. Addict Biol. 2021;26(1):e12849. doi: 10.1111/adb.12849. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Data sources for the genetic instruments.
Data Availability Statement
All the data supporting the conclusions of this article are included within the article and its supplementary file.