Abstract
β-Lactam resistance was studied in 1,072 consecutive P. mirabilis clinical strains isolated at the Clermont-Ferrand teaching hospital between April 1996 and March 1998. The frequency of amoxicillin resistance was 48.5%. Among the 520 amoxicillin-resistant isolates, three resistance phenotypes were detected: penicillinase (407 strains [78.3%]), extended-spectrum β-lactamase (74 strains [14.2%]), and inhibitor resistance (39 strains [7.5%]). The penicillinase phenotype isolates were divided into three groups according to the level of resistance to β-lactams, which was shown to be related to the strength of the promoter. The characterization of the different β-lactamases showed that amoxicillin resistance in P. mirabilis was almost always (97%) associated with TEM or TEM-derived β-lactamases, most of which evolved via TEM-2.
After Escherichia coli, Proteus mirabilis is the most often isolated member of the Enterobacteriaceae in European clinical microbiology laboratories, being isolated more often than Klebsiella pneumoniae (22, 26). Wild-type strains of P. mirabilis are susceptible to all penicillins and cephalosporins. However, since 1990, the resistance of the species to β-lactams has regularly increased (20, 22, 26, 32). Amoxicillin resistance in P. mirabilis is mainly due to the plasmid-mediated penicillinases TEM-1 and TEM-2. TEM-2 is more frequently encountered in this species than in other Enterobacteriaceae (22, 28); TEM-like penicillinase TEM-57 was recently reported by Bonnet et al. (7).
Since 1991, TEM-derived extended-spectrum β-lactamases (ESBL) (TEM-3, TEM-8, TEM-10, TEM-21, TEM-24, TEM-26, and TEM-66) in P. mirabilis have been reported (7, 11, 15, 24, 27, 29; L. Pagani, F. Luzzaro, R. Migliavacca, M. G. Perilli, R. Daturi, G. Lombardi, C. Matti, E. Giacobone, and G. Amicosante, Abstr. 37th Intersci. Conf. Antimicrob. Agents Chemother., abstr. D14, p. 85, 1997). Inhibitor-resistant TEM β-lactamases (TEM-44, TEM-65, TEM-73, and TEM-74) in this species have been recently described (7, 9). Finally, non-TEM-derived β-lactamases (CMY-3, CMY-4, CEP-1, CTX-M-2, and PER-2) in P. mirabilis have also been reported (4, 5, 6, 8, 36). The aim of this 2-year survey was to assess the prevalence of established and newer β-lactamases among amoxicillin-resistant P. mirabilis clinical strains isolated from the teaching hospital of Clermont-Ferrand.
MATERIALS AND METHODS
Bacterial strains.
All nonduplicate P. mirabilis strains isolated from Clermont-Ferrand teaching hospital during a 2-year survey (1 April 1996 to 31 March 1998) were included in this study. Isolates were identified by the Rapid ID 32 E system (BioMérieux, La Balme les Grottes, France).
Susceptibility testing.
Susceptibilities of P. mirabilis isolates were determined by the Rapid ATB E system (BioMérieux). A modified double-disc synergy test (11) was used to detect ESBL. The amoxicillin-resistant isolates were screened for susceptibility to a battery of β-lactam antibiotics chosen to facilitate the recognition of the resistance patterns associated with the different β-lactamase types. Amoxicillin-resistant strains were classified into three different phenotypes according to the following criteria. (i) Penicillinase phenotype isolates were susceptible or intermediate to amoxicillin plus clavulanate and cephalothin and susceptible to ticarcillin plus clavulanate. (ii) ESBL phenotype isolates were resistant to amoxicillin, ticarcillin, and cephalothin and gave a positive result in the modified double-disc synergy test. (iii) Inhibitor-resistant TEM or oxacillinase (IRT/OXA) phenotype isolates were resistant to amoxicillin plus clavulanate and fully susceptible to cephalothin, which eliminates strains that hyperproduce a penicillinase or that produce both a penicillinase and a possibly acquired cephalosporinase. For 217 amoxicillin-resistant strains the disc diffusion method on Mueller-Hinton agar (Sanofi Diagnostics Pasteur, Marnes-la-Coquette, France) was used to determine the inhibition diameters of amoxicillin, amoxicillin plus clavulanate, ticarcillin, ticarcillin plus clavulanate, and cephalothin. MICs were determined by a dilution method on Mueller-Hinton agar (Sanofi Diagnostics Pasteur) with an inoculum of 104 CFU per spot. Antibiotics were provided as powders by SmithKline Beecham, Paris, France (amoxicillin, ticarcillin, and clavulanate) and by Eli Lilly, Paris, France (cephalothin).
Analytical isoelectric focusing.
Isoelectric focusing was performed with polyacrylamide gels containing ampholines (pH range, 3.5 to 10) as previously described (12). β-Lactamases with known pIs (TEM-1, 5.4; TEM-2, 5.6; TEM-3, 6.3; TEM-15, 6.0; TEM-30, 5.2; CARB-2, 5.7; SHV-1, 7.7) were used as standards.
β-Lactamase assays.
Specific β-lactamase activities in crude sonic extracts were determined by the computerized microacidic method described previously (21) with 225 mM benzylpenicillin as the substrate. Enzyme activity was standardized against the total protein concentration in the enzyme preparation, as estimated by the Bio-Rad (Richmond, Calif.) protein assay, with bovine serum albumin (Sigma Chemical Co., St. Louis, Mo.) used as the standard. One unit of β-lactamase activity was defined as the amount of enzyme which hydrolyzes 1 μmol of benzylpenicillin per min at 37°C and pH 7.
Detection of point mutation by ASPCR.
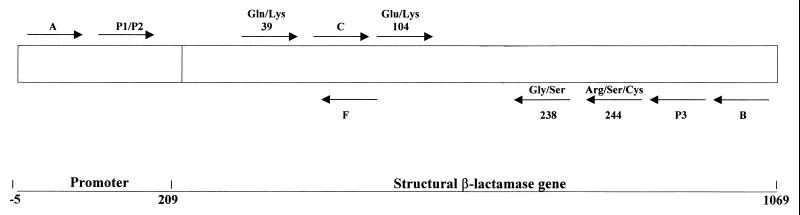
Allele-specific PCR (ASPCR) (37, 38) was used to detect point mutations in the promoter region and the structural TEM genes. The primers used are listed in Table 1, and their positions are shown in Fig. 1. Point mutations were detected by using primers P1 and P2 (position 32 in the promoter region) in conjunction with primer P3, primers Gln and Lys 39 (position 317) in conjunction with primer B, primers Glu and Lys 104 (position 512) in conjunction with primer B, primers Gly and Ser 238 (position 914) in conjunction with primer C, and primers Arg, Ser, and Lys 244 (position 929) in conjunction with primer C. Annealing temperatures were 66, 46, 46, 65, and 58°C for the above primer combinations, respectively.
TABLE 1.
Nucleotide sequences of the oligonucleotides used for amplification and/or sequencing reactions
| Primera | Sequencec | Position 5′b |
|---|---|---|
| Amplification and/or sequencing | ||
| A | 5′-TAAAATTCTTGAAGACG-3′ | −5 |
| C | 5′-GGGCAAGAGCAACTCGG-3′ | 461 |
| F | 5′-TTACTGTCATGCCATCCG-3′ | 560 |
| P3 | 5′-CTGACTCCCCGTCGTGT-3′ | 1003 |
| B | 5′-TTACCAATGCTTAATCA-3′ | 1069 |
| ASPCR | ||
| P1 | 5′-GGGCCTCGTGATACGCC-3′ | 16 |
| P2 | 5′-----------------T-3′ | |
| Gln 39 | 5′-AAAGATGCTGAAGATC-3′ | 302 |
| Lys 39 | 5′----------------A-3′ | |
| Glu 104 | 5′-CTCAGAATGACTTGGTTG-3′ | 495 |
| Lys 104 | 5′------------------A-3′ | |
| Gly 238 | 5′-GCGAGACCCACGCTCACC-3′ | 931 |
| Ser 238 | 5′------------------T-3′ | |
| Arg 244 | 5′-TGCTGCAATGATACCGCG-3′ | 946 |
| Ser 244 | 5′------------------T-3′ | |
| Cys 244 | 5′------------------A-3′ |
FIG. 1.
Strategy for amplification and/or sequencing of blaTEM genes. Arrows, primers (5′ to 3′). Numbering of amino acids is as described by Ambler et al. (1). Numbering of nucleotides is as described by Sutcliffe (35).
DNA amplification and sequencing.
In order to confirm ASPCR results, some representative isolates were selected for DNA amplification and sequencing. Nucleotide sequencing was also performed when ASPCR results were not in agreement with those expected according to the resistance phenotype. Primers A and B (Table 1) were used to amplify the whole TEM genes as previously described (23). Nucleotide sequences were determined as previously described (8) using primers A, B, C, and F.
RESULTS
During this 2-year survey, 1,072 nonrepetitive strains of P. mirabilis were isolated. Among these isolates 520 strains (48.5%) were intermediate or resistant to amoxicillin. According to the criteria established above, the phenotypes of these 520 isolates were classified as penicillinase, ESBL, or IRT/OXA (Table 2). As shown, 407 of 520 isolates (78.3%) presented a penicillinase phenotype, 74 of 520 isolates (14.2%) produced an ESBL phenotype, and 39 of 520 isolates (7.5%) presented an IRT/OXA phenotype.
TABLE 2.
β-Lactam resistance phenotypes of 520 nonduplicate P. mirabilis strains resistant to amoxicillin
| Phenotypeb | Resistancea toc:
|
Synergy test result | No. (%) of strains | ||||
|---|---|---|---|---|---|---|---|
| AMX | AMC | TIC | TCC | CF | |||
| Penicillinase | R | S/I | S/I/R | S | S/I | − | 407 (78.3) |
| ESBL | R | S/I | R | S | R | + | 74 (14.2) |
| IRT/OXA | R | R | S/I | S/I | S | − | 39 (7.5) |
S, susceptible; I, intermediate; R, resistant; S/I, susceptible or intermediate; S/I/R, susceptible, intermediate, or resistant. Based on the recommendations of the Antibiogram Committee of the French Society for Microbiology (2).
See phenotype definitions in Materials and Methods.
AMX, amoxicillin; AMC, amoxicillin-clavulanate; TIC, ticarcillin; TCC, ticarcillin-clavulanate; CF, cephalothin.
All IRT/OXA and ESBL phenotype isolates as well as 104 penicillinase phenotype strains isolated during the last 6 months of the study were retained for further analysis.
Table 3 shows inhibition diameters, MIC ranges, and MICs at which 90% of the isolates are inhibited (MIC90) for five β-lactams obtained for each phenotype. According to their levels of resistance, penicillinase phenotype isolates were divided into three groups: (i) PL (low level), amoxicillin diameter ≥ 10 mm and ticarcillin diameter ≥ 17 mm; (ii) PI (intermediate level), amoxicillin diameter = 6 mm and ticarcillin diameter > 10 mm; (iii) PH (high-level), amoxicillin and ticarcillin diameters 6 mm. MIC results confirmed these different levels of resistance. MIC90 of amoxicillin and ticarcillin were, respectively, 64 and ≤8 μg/ml for the PL phenotype, 512 and 128 μg/ml for the PI phenotype, and >2,048 and 1,024 μg/ml for the PH phenotype. MIC90 of amoxicillin-clavulanate increased from ≤2 μg/ml for the PL phenotype to 32 μg/ml for the PH phenotype. MIC90 of ticarcillin plus clavulanate remained ≤8 μg/ml in the three groups.
TABLE 3.
Inhibition zone diameters and MICs for P. mirabilis isolates with different β-lactamase resistance phenotypes
| β-Lactam phenotype (no. of isolates) | Amoxicillin
|
Amoxicillin + clavulanic acid (2 μg/ml)
|
Ticarcillin
|
Ticarcillin + clavulanic acid (2 μg/ml)
|
Cephalothin
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean zone diam (mm) ± SD | MIC range (μg/ml) | MIC90 (μg/ml) | Mean zone diam (mm) ± SD | MIC range (μg/ml) | MIC90 (μg/ml) | Mean zone diam (mm) ± SD | MIC range (μg/ml) | MIC90 (μg/ml) | Mean zone diam (mm) ± SD | MIC range (μg/ml) | MIC90 (μg/ml) | Mean zone diam (mm) ± SD | MIC range (μg/ml) | MIC90 (μg/ml) | |
| PL (23) | 12.5 ± 2.1 | 16–64 | 64 | 29.0 ± 1.9 | ≤2 | ≤2 | 24.2 ± 3.1 | ≤8–16 | ≤2 | 34.8 ± 1.9 | ≤2 | ≤2 | 25.6 ± 2.0 | ≤2–8 | 4 |
| PI (28) | 6.0 ± 0 | 32–1,024 | 512 | 26.7 ± 2.0 | ≤2–4 | 4 | 15.3 ± 5.8 | ≤8–128 | 4 | 31.2 ± 2.6 | ≤2 | ≤2 | 22.9 ± 2.4 | ≤2–8 | 4 |
| PH (53) | 6.0 ± 0 | 512–>2,048 | >2,048 | 21.1 ± 3.2 | ≤2–128 | 32 | 6.3 ± 0 | 128–1,024 | 32 | 28.4 ± 3.0 | ≤2–16 | 8 | 18.7 ± 2.4 | ≤2–16 | 16 |
| ESBL (74) | 6.0 ± 0 | 256–>2,048 | 1,024 | 26.3 ± 2.3 | ≤2–8 | ≤2 | 7.3 ± 1.8 | 128–>1,024 | ≤2 | 30.5 ± 1.7 | ≤2–8 | ≤2 | 13.9 ± 2.8 | 8–128 | 64 |
| IRT/OXA (39) | 6.1 ± 0.6 | 128–2,048 | 1,024 | 10.9 ± 3.2 | 32–1,024 | 256 | 21.2 ± 5.9 | ≤8–64 | 256 | 28.1 ± 2.2 | ≤2–32 | 16 | 24.1 ± 1.6 | ≤2–8 | 4 |
β-Lactamase-specific activities were determined for 10 isolates each of the PL, PI, and PH phenotypes. The ranges and mean values, respectively, for these phenotypes were as follows: PL, 0.02 to 0.09 and 0.06 U/mg; PI, 0.09 to 1.13 and 0.55 U/mg; PH, 1.65 to 9.72 and 4.49 U/mg.
The ESBL phenotype isolates were characterized by a high level of resistance to amoxicillin (MIC90, 1,024 μg/ml) and ticarcillin (MIC90, 512 μg/ml) and by a high level of susceptibility to β-lactam–β-lactamase inhibitor combinations (MIC90 of amoxicillin plus clavulanate and ticarcillin plus clavulanate, ≤2 μg/ml). MICs of cephalothin ranged from 8 to 128 μg/ml. The IRT/OXA phenotype isolates were characterized by a high level of resistance to amoxicillin plus clavulanate (inhibition zone diameter = 10.9 ± 3.2 mm; MIC90 = 256 μg/ml) and susceptibility to cephalothin (mean zone diameter = 24.1 ± 1; MIC90 = 4 μg/ml).
β-Lactamase characterization.
Results of β-lactamase characterization are presented in Tables 4, 5, and 6.
TABLE 4.
β-Lactamases produced by 104 penicillinase-producing P. mirabilis strains
| pI (n) | ASPCR position 39
|
ASPCR promoter
|
β-Lactamase(s) (promoter), n | ||
|---|---|---|---|---|---|
| Gln | Lys | P3 | Pa + Pb | ||
| Low level (n = 23) | |||||
| 5.4 (21) | + | − | + | − | TEM-1 (P3), 20 |
| + | − | − | + | TEM-1 (Pa + Pb), 1 | |
| 5.7 (1) | − | − | NDa | ND | CARB-2 (PSE-1), 1 |
| 7.7 (1) | ND | ND | ND | ND | SHV-1, 1 |
| Intermediate level (n = 28) | |||||
| 5.4 (21) | + | − | + | − | TEM-1 (P3), 15 |
| + | − | − | + | TEM-1 (Pa + Pb), 6 | |
| 5.6 (3) | − | + | − | + | TEM-2 (Pa + Pb), 3 |
| 5.4 + 5.6 (3) | + | + | − | + | TEM-1 (Pa + Pb) + TEM-2 (Pa + Pb), 1 |
| + | + | + | + | TEM-1 (P3) + TEM-2 (Pa + Pb), 2 | |
| 5.7 (1) | − | − | ND | ND | CARB-2 (PSE-1), 1 |
| High level (n = 53) | |||||
| 5.4 (19) | + | − | − | + | TEM-1 (Pa + Pb), 17 |
| + | − | + | + | TEM-1 (P3) + TEM-1 (Pa + Pb), 1 | |
| + | − | + | − | TEM-1 (P4), 1 | |
| 5.6 (21) | − | + | − | + | TEM-2 (Pa + Pb), 19 |
| − | + | + | − | TEM-2 (P4), 1 | |
| − | + | − | − | TEM-2 (deletion + G162→T), 1 | |
| 5.4 + 5.6 (12) | + | + | + | + | TEM-1 (P3) + TEM-2 (Pa + Pb), 10 |
| + | + | − | + | TEM-1 (Pa + Pb) + TEM-2 (Pa + Pb), 2 | |
| 7.7 (1) | ND | ND | ND | ND | SHV-1, 1 |
ND, not determined.
TABLE 5.
β-Lactamases produced by 39 P. mirabilis strains presenting an inhibitor-resistant phenotype
| pI (n) | ASPCR position 39
|
ASPCR position 244
|
β-Lactamase(s) (n) | |||
|---|---|---|---|---|---|---|
| Gln | Lys | Arg | Ser | Cys | ||
| 5.4 (35) | + | + | + | + | TEM-1 + IRT-13 (18) | |
| − | + | − | + | IRT-13 (14) | ||
| − | + | − | − | + | IRT-16 (2) | |
| + | + | + | − | + | TEM-1 + IRT-16 (1) | |
| 5.2 (1) | + | − | − | + | IRT-2 | |
| 7.1 (3) | NDa | ND | ND | ND | ND | Oxacillinase type |
ND, not determined.
TABLE 6.
β-Lactamases produced by 74 P. mirabilis strains presenting an ESBL phenotype
| pI (n) | ASPCR position 39
|
ASPCR position 104
|
ASPCR position 238
|
β-Lactamase(s) | |||
|---|---|---|---|---|---|---|---|
| Gln | Lys | Glu | Lys | Gly | Ser | ||
| 6.3 (71) | − | + | − | + | − | + | TEM-3 |
| 6.3 + 5.4 (1) | + | + | + | + | + | + | TEM-3 + TEM-1 |
| 6.3 + 5.6 (1) | − | + | + | + | + | + | TEM-3 + TEM-2 |
| 6.0 (1) | − | + | − | + | − | + | TEM-66 |
(i) Penicillinase phenotype (Table 4).
Twenty-one of the 23 strains presenting a PL penicillinase phenotype produced the β-lactamase TEM-1. Twenty isolates had the TEM-1 type promoter P3 (30), and only 1 had the TEM-2 type promoter pair Pa and Pb (13) characterized by a C32→T substitution. One strain produced the β-lactamase SHV-1, and one strain produced the β-lactamase CARB-2. Twenty-one of the 28 strains presenting a PI penicillinase phenotype produced the β-lactamase TEM-1 associated with the promoter P3 in 15 isolates or with the promoter pair Pa and Pb in 6 isolates. Three strains produced TEM-2 associated with the promoter pair Pa and Pb, and three strains produced TEM-1 and TEM-2. One strain produced the β-lactamase CARB-2. Nineteen of the 53 strains presenting a PH penicillinase phenotype produced the TEM-1 β-lactamase. Eighteen strains had the TEM-2 type promoter pair Pa and Pb, and 1 strain had the promoter P4 (16) characterized by a G162→T substitution. Twenty-one strains produced the β-lactamase TEM-2 associated with the promoter pair Pa and Pb in 19 isolates, with the promoter P4 in 1 isolate, and with a promoter associating a 135-bp deletion (between positions 22 and 158) and the G162→T substitution in 1 isolate. Twelve strains produced the two β-lactamases TEM-1 and TEM-2, and 1 strain produced the β-lactamase SHV-1.
(ii) Inhibitor-resistant phenotype (Table 5).
Thirty-five of the 39 inhibitor-resistant strains produced an IRT related to TEM-2 (Lys 39): 32 produced IRT-13/TEM-44, and 3 produced IRT-16/TEM-65. In 19 strains the IRT enzyme was associated with TEM-1. One strain produced IRT-2/TEM-30 related to TEM-1 (Gln 39). Three strains produced an oxacillinase type enzyme (pI 7.1) as suggested by kinetic constant determination (data not shown).
In the 17 isolates producing only an IRT enzyme, ASPCR results showed that all had the strong promoter pair Pa and Pb.
(iii) ESBL phenotype (Table 6).
Seventy-three of the 74 strains produced the TEM-3 β-lactamase associated with TEM-1 in 1 strain and with TEM-2 in 1 strain. One isolate produced TEM-66, a novel ESBL of pI 6.0 (7).
ASPCR results concerning the promoter regions of 24 ESBL-producing representative strains showed that all possessed the strong promoter pair Pa and Pb.
DISCUSSION
This study examined the extent and nature of β-lactamase-mediated resistance among 1,072 consecutively isolated P. mirabilis strains obtained from clinical specimens at the Clermont-Ferrand teaching hospital during a 2-year period from April 1996 to March 1998. The prevalence of amoxicillin resistance was 48.5% (520 of 1,072 strains). This prevalence is similar to that observed in a similar investigation undertaken in the same hospital in 1994, 46.5% (data not shown), but is higher than that reported in previous studies, which produced values ranging from 22, 29.1, and 30% at the beginning of the 1990s (20, 22, 32) to 42.6% in 1996 (26).
Analysis of these resistance patterns and of isoelectric focusing results (Table 2) showed that amoxicillin resistance in P. mirabilis was associated with production of penicillinases (78.3%), ESBL (14.2%), or inhibitor-resistant β-lactamases (7.5%). As reported in Table 4, the isolates presenting a penicillinase phenotype almost always produced TEM-1 and/or TEM-2 enzymes (100 of 104 isolates [96.1%]). TEM-1 was the commonest penicillinase type (76 of 100 isolates), but, as previously reported, TEM-2 was encountered with a high frequency (39 of 100 isolates) similar to data reported by Liu et al. (22) for 25 P. mirabilis strains. These penicillinase-producing isolates could be divided into three groups according to their resistance levels.
Among the 21 TEM-producing strains presenting a low-level resistance phenotype, 95% produced the β-lactamase TEM-1 associated with the weak promoter P3 (30). Only one strain produced the β-lactamase TEM-1 associated with the strong TEM-2 type promoter pair Pa and Pb (13). This TEM-2 type promoter was previously described as being in the promoter region of the TEM-1 genes of 15 clinical isolates of E. coli (38), of “TEM-1B-like” blaIRT genes in E. coli (10), and of ESBL genes (16). The C32→T substitution converts the weak P3 promoter of blaTEM-1 into the two overlapping promoters Pa and Pb and results in a large increase in β-lactamase production (13). The reason for the failure of one strain to exhibit a high level of resistance despite evidence of a strong promoter is not known. As suggested by Wu et al. for E. coli (38), it could be the result of regulational phenomena such as mRNA transcription attenuation, high-level proteases, or export problems.
In contrast 100% of the 52 TEM-producing strains presenting a high-level resistance phenotype possessed a strong promoter associated with either TEM-1, TEM-2 or TEM-1 and TEM-2. The TEM-2 type promoter pair Pa and Pb (C32→T) was present in 49 strains. The strong promoter P4 (16), characterized by the G162→T substitution, was present in one TEM-1-producing strain and one TEM-2-producing strain. This G162→T transversion falls within the functional −10 Pribnow box and consequently renders the −10 consensus region of the β-lactamase gene more similar to the optimal promoter of E. coli 5′-TATAAT-3′ (17). Strong promoter P4 was previously reported as a mechanism of hyperproduction of TEM-1 in Shigella flexneri (34), of “TEM-2-like” IRTs in E. coli (10), and of ESBL in K. pneumoniae, Klebsiella oxytoca, and E. coli (16, 18, 19). In one strain the high-level resistance could result from the presence of the strong promoter described by Arlet et al. for TEM-20, associating a 135-bp deletion and substitution G162→T leading to the association of the −35 region of Pa with the more efficient −10 region of P3 (3).
The results discussed above confirm that, as previously reported for E. coli, the strength of the promoter plays an important role in the production level of, as well as in the resistance level conferred by, TEM β-lactamases in P. mirabilis.
In this study the ESBL phenotype was due to the production of TEM-3 in 98% of strains. This dominance of TEM-3 in ESBL-producing P. mirabilis in France has been previously reported (11, 24; C. De Champs, D. Sirot, C. Chanal, J. Sirot, and the French Study Group, Abstr. 39th Intersci. Conf. Antimicrob. Agents Chemother., abstr. 1485, 1999). This enzyme, initially observed in France in K. pneumoniae (31), appeared in P. mirabilis only since 1994 (11, 24). The ESBL TEM-10, TEM-24, TEM-26, and TEM-66 were next described in P. mirabilis (7, 27, 29). The delay in ESBL arriving in P. mirabilis could be due to a misdetection because of the weak expression of β-lactamases in this species, requiring a modified synergy test for routine detection (7, 11). While ESBL-producing Enterobacteriaceae were initially reported mainly in intensive care units, it is noteworthy that in our study the percentage of ESBL-producing P. mirabilis strains was higher in long-stay care units than in intensive care units (14).
The inhibitor-resistant phenotype was due to the production of an IRT β-lactamase in 92.3% of strains. In this study, all but one of the IRTs were related to TEM-2 (32 IRT-13/TEM-44 and 3 IRT-16/TEM-65). These two enzymes were previously described only for P. mirabilis (7, 9), possibly because of the high frequency of TEM-2 in this species.
In this work the promoter regions of 41 blaTEM genes encoding TEM mutant β-lactamases (17 IRTs and 24 ESBL) were studied. The strong promoter pair Pa plus Pb was always found. These results reinforce the hypothesis that selection of TEM mutant enzymes could only occur if the parent strain produces a high level of β-lactamase (25). In order to study the possible existence of epidemic strains in our hospital, ribotyping was performed for 50 of the 104 penicillinase-producing strains (data not shown) and revealed that these strains were genetically unrelated, which rules out the epidemic dissemination of a clone. Similar results were previously reported for IRT-13-producing strains (9), suggesting an independent emergence of inhibitor resistance under antibiotic selective pressure in P. mirabilis isolates. In contrast, the high prevalence of ESBL-producing P. mirabilis isolates in long-stay and intensive care units (14) was probably due to the dissemination of epidemic strains or plasmids, as previously reported for other TEM-3-producing Enterobacteriaceae (33).
Our results show that in our hospital and during this study the amoxicillin resistance in P. mirabilis was almost always (97%) associated with TEM or TEM-derived β-lactamases. Most of these enzymes (60.3%) evolved via TEM-2 (Gln 39). Finally, the prevalence of the different β-lactamases in amoxicillin-resistant P. mirabilis clinical isolates was as shown in Table 7.
TABLE 7.
β-Lactamase types of 520 P. mirabilis strains resistant to amoxicillin
| Phenotype (n) | Enzyme(s) | % of strains with enzyme |
|---|---|---|
| Penicillinase (407) | TEM-1 | 46 |
| TEM-2 | 18 | |
| TEM-1 + TEM-2 | 11.3 | |
| CARB-2 | 1.5 | |
| SHV-1 | 1.5 | |
| Total | 78.3 | |
| IRT/OXA (39) | IRT-13 | 2.7 |
| IRT-13 + TEM-1 | 3.4 | |
| IRT-16 | 0.4 | |
| IRT-16 + TEM-1 | 0.2 | |
| IRT-2 | 0.2 | |
| OXA | 0.6 | |
| Total | 7.5 | |
| ESBL (74) | TEM-3 | 13.6 |
| TEM-3 + TEM-1 | 0.2 | |
| TEM-3 + TEM-2 | 0.2 | |
| TEM-66 | 0.2 | |
| Total | 14.2 |
Conclusion.
This study emphasizes the diversity of β-lactamases in P. mirabilis. During the last decade, the most significant fact was the appearance of resistance to expanded-spectrum cephalosporins in this species naturally susceptible to β-lactams. Since 1991 TEM ESBL have been reported (7, 13, 16, 28, 29, 32; Pagani et al., 37th ICAAC). Since 1998 AmpC type β-lactamases have been described (6, 8, 36). More recently non-TEM and SHV-derived ESBL in this species have been reported (4, 5).
P. mirabilis is therefore involved in new resistance mechanisms and could constitute a plasmid reservoir for antibiotic resistance, justifying particular vigilance from the microbiologists.
ACKNOWLEDGMENTS
We thank Marlène Jan, Rolande Perroux, and Dominique Rubio for technical assistance.
This study was supported in part by a grant from the Direction de la Recherche et des Etudes Doctorales, Ministère de l'Education Nationale, France.
REFERENCES
- 1.Ambler R P, Coulson A F W, Frere J-M, Ghuysen J M, Joris B, Forsman M, Levesque R C, Tiraby G, Waley S G. A standard numbering scheme for the class A β-lactamases. Biochem J. 1991;276:269–272. doi: 10.1042/bj2760269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Antibiogram Committee of the French Society for Microbiology. Statement. Pathol Biol. 1998;46:1–16. [Google Scholar]
- 3.Arlet G, Goussard S, Courvalin P, Philippon A. Sequence of the genes for the TEM-20, TEM-21, TEM-22, and TEM-29 extended-spectrum β-lactamases. Antimicrob Agents Chemother. 1999;43:969–971. doi: 10.1128/aac.43.4.969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bauernfeind A, Stemplinger I, Jungwirth R, Ernst S, Casellas J M. Sequences of β-lactamase genes encoding CTX-M-1 (MEN-1) and CTX-M-2 and relationship of their amino acid sequences with those of other β-lactamases. Antimicrob Agents Chemother. 1996;40:509–513. doi: 10.1128/aac.40.2.509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bauernfeind A, Stemplinger I, Jungwirth R, Mangold P, Amann S, Akalin E, Ang Ö, Bal C, Casellas J M. Characterization of β-lactamase gene blaPER-2, which encodes an extended-spectrum class A β-lactamase. Antimicrob Agents Chemother. 1996;40:616–620. doi: 10.1128/aac.40.3.616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bobrowski M M, Matthew M, Barth P T, Datta N, Grinter N J, Jacob A E, Kontomichalou P, Dale J W, Smith J T. Plasmid-determined beta-lactamase indistinguishable from the chromosomal beta-lactamase of Escherichia coli. J Bacteriol. 1976;125:149–157. doi: 10.1128/jb.125.1.149-157.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bonnet R, De Champs C, Sirot D, Chanal C, Labia R, Sirot J. Diversity of TEM mutants in Proteus mirabilis. Antimicrob Agents Chemother. 1999;43:2671–2677. doi: 10.1128/aac.43.11.2671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bret L, Chanal-Claris C, Sirot D, Chaibi E B, Labia R, Sirot J. Chromosomally encoded AmpC-type β-lactamase in a clinical isolate of Proteus mirabilis. Antimicrob Agents Chemother. 1998;42:1110–1114. doi: 10.1128/aac.42.5.1110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bret L, Chanal C, Sirot D, Labia R, Sirot J. Characterization of an inhibitor-resistant enzyme IRT-2 derived from TEM-2 β-lactamase produced by Proteus mirabilis strains. J Antimicrob Chemother. 1996;38:183–191. doi: 10.1093/jac/38.2.183. [DOI] [PubMed] [Google Scholar]
- 10.Canica M M, Lu C L, Krishnamoorthy R, Paul G. Molecular diversity and evolution of blaTEM genes encoding β-lactamases resistant to clavulanic acid in clinical E. coli. J Mol Evol. 1997;44:57–65. doi: 10.1007/pl00006121. [DOI] [PubMed] [Google Scholar]
- 11.Chanal C, Sirot D, Romaszko J P, Bret L, Sirot J. Survey of prevalence of extended spectrum β-lactamases among Enterobacteriaceae. J Antimicrob Chemother. 1996;38:127–132. doi: 10.1093/jac/38.1.127. [DOI] [PubMed] [Google Scholar]
- 12.Chanal C M, Sirot D L, Petit A, Labia R, Morand A, Sirot J L, Cluzel R A. Multiplicity of TEM-derived β-lactamases from Klebsiella pneumoniae strains isolated at the same hospital and relationships between the responsible plasmids. Antimicrob Agents Chemother. 1989;33:1915–1920. doi: 10.1128/aac.33.11.1915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen S T, Clowes R C. Two improved promoter sequences for the β-lactamase expression arising from a single base-pair substitution. Nucleic Acids Res. 1984;12:3219–3234. doi: 10.1093/nar/12.7.3219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.De Champs C, Bonnet R, Sirot D, Chanal C, Sirot J. Clinical relevance of Proteus mirabilis in hospital patients: a two-year survey. J Antimicrob Chemother. 2000;45:537–539. doi: 10.1093/jac/45.4.537. [DOI] [PubMed] [Google Scholar]
- 15.Goldstein F W, Pean Y, Rosato A, Gertner J, Gutmann L the Vigil'Roc Study Group. Characterization of ceftriaxone-resistant Enterobacteriaceae: a multicentre study in 26 French hospitals. J Antimicrob Chemother. 1993;32:595–603. doi: 10.1093/jac/32.4.595. [DOI] [PubMed] [Google Scholar]
- 16.Goussard S, Courvalin P. Updated sequence information for TEM β-lactamase genes. Antimicrob Agents Chemother. 1999;43:367–370. doi: 10.1128/aac.43.2.367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hawley D K, McClure W R. Compilation and analysis of Escherichia coli promoter DNA sequences. Nucleic Acids Res. 1983;11:2237–2255. doi: 10.1093/nar/11.8.2237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Heritage J, Hawkey P M, Todd N, Lewis I J. Transposition of the gene encoding a TEM-12 extended-spectrum β-lactamase. Antimicrob Agents Chemother. 1992;36:1981–1986. doi: 10.1128/aac.36.9.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hibbert-Rogers L C F, Heritage J, Todd N, Hawkey P M. Convergent evolution of TEM-26, a β-lactamase with extended spectrum activity. J Antimicrob Chemother. 1994;33:707–720. doi: 10.1093/jac/33.4.707. [DOI] [PubMed] [Google Scholar]
- 20.Jarlier V, Fosse T, Philippon A the ICU Study Group. Antibiotic susceptibility in aerobic gram-negative bacilli isolated in intensive care units in 39 French teaching hospitals (ICU study) Intensive Care Med. 1996;22:1057–1065. doi: 10.1007/BF01699228. [DOI] [PubMed] [Google Scholar]
- 21.Labia R, Andrillon J, Le Goffic F. Computerized microacidimetric determination of β-lactamase Michaelis-Menten constants. FEBS Lett. 1973;33:42–44. doi: 10.1016/0014-5793(73)80154-1. [DOI] [PubMed] [Google Scholar]
- 22.Liu P Y F, Gur D, Hall L M C, Livermore D M. Survey of the prevalence of β-lactamases amongst 1000 Gram-negative bacilli isolated consecutively at the Royal London Hospital. J Antimicrob Chemother. 1992;30:429–447. doi: 10.1093/jac/30.4.429. [DOI] [PubMed] [Google Scholar]
- 23.Mabilat C, Goussard S, Sougakoff W, Spencer R C, Courvalin P. Direct sequencing of the amplified structural gene and promoter for the extended broad-spectrum β-lactamase TEM-9 (RHH-1) of Klebsiella pneumoniae. Plasmid. 1990;23:27–34. doi: 10.1016/0147-619x(90)90041-a. [DOI] [PubMed] [Google Scholar]
- 24.Mariotte S, Nordmann P, Nicolas M H. Extended-spectrum β-lactamase in Proteus mirabilis. J Antimicrob Chemother. 1994;33:925–935. doi: 10.1093/jac/33.5.925. [DOI] [PubMed] [Google Scholar]
- 25.Medeiros A A. Evolution and dissemination of β-lactamases accelerated by generations of β-lactam antibiotics. Clin Infect Dis. 1997;24(Suppl. 1):519–545. doi: 10.1093/clinids/24.supplement_1.s19. [DOI] [PubMed] [Google Scholar]
- 26.Nicolas-Chanoine M H, Chardon H, Avril J L, Cattoen Y, Croix J C, Dabernat H, Etienne J, Fosse T, Ghnassia J C, Lecaillon E, Marmonier A, Roussel-Delvallez M, Soussy J C, Trevoux A, Sirot J. Susceptibility of Enterobacteriaceae to betalactams and fluoroquinolones: a French multicentre study. Clin Microbiol Infect. 1997;3(Suppl. 2):74–75. doi: 10.1046/j.1469-0691.2002.00393.x. [DOI] [PubMed] [Google Scholar]
- 27.Palzkill T, Thomson K S, Sanders C C, Moland E S, Huang W, Milligan T W. New variant of TEM-10 β-lactamase gene produced by a clinical isolate of Proteus mirabilis. Antimicrob Agents Chemother. 1995;39:1199–1200. doi: 10.1128/aac.39.5.1199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Philippon A, Arlet G, Lagrange P H. Origin and impact of plasmid-mediated extended-spectrum beta-lactamases. Eur J Clin Microbiol Infect Dis. 1994;13(Suppl. 1):17–29. doi: 10.1007/BF02390681. [DOI] [PubMed] [Google Scholar]
- 29.Pitout J D D, Thomson K S, Hanson N D, Ehrhardt A F, Moland E S, Sanders C C. β-Lactamases responsible for resistance to expanded-spectrum cephalosporins in Klebsiella pneumoniae, Escherichia coli, and Proteus mirabilis isolates recovered in South Africa. Antimicrob Agents Chemother. 1998;42:1350–1354. doi: 10.1128/aac.42.6.1350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Russel D R, Bennet G N. Characterization of the β-lactamase promoter of pBR322. Nucleic Acids Res. 1981;11:2517–2532. doi: 10.1093/nar/9.11.2517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sirot D, Sirot J, Labia R, Morand A, Courvalin P, Darfeuille-Michaud A, Perroux R, Cluzel R. Transferable resistance to third-generation cephalosporins in clinical isolates of Klebsiella pneumoniae: identification of CTX-1, a novel β-lactamase. J Antimicrob Chemother. 1987;20:323–334. doi: 10.1093/jac/20.3.323. [DOI] [PubMed] [Google Scholar]
- 32.Sirot D L, Golstein F W, Soussy C J, Courtieu A L, Husson M O, Lemozy J, Meyran M, Morel C, Perez R, Quentin-Noury C, Reverdy M E, Scheftel J M, Rosembaum M, Rezvani Y. Resistance to cefotaxime and seven other β-lactams in members of the family Enterobacteriaceae: a 3-year survey in France. Antimicrob Agents Chemother. 1992;36:1677–1681. doi: 10.1128/aac.36.8.1677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sirot J, Chanal C, Petit A, Sirot D, Labia R, Gerbaud G. Klebsiella pneumoniae and other Enterobacteriaceae producing novel plasmid-mediated beta-lactamases markedly active against third-generation cephalosporins: epidemiologic studies. Rev Infect Dis. 1988;10:850–859. doi: 10.1093/clinids/10.4.850. [DOI] [PubMed] [Google Scholar]
- 34.Siu L K, Ho P L, Yuen K Y, Wong S S Y, Chau P Y. Transferable hyperproduction of TEM-1 β-lactamase in Shigella flexneri due to a point mutation in the Pribnow box. Antimicrob Agents Chemother. 1997;41:468–470. doi: 10.1128/aac.41.2.468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sutcliffe J G. Nucleotide sequence of the ampicillin resistance gene of Escherichia coli plasmid pBR322. Proc Natl Acad Sci USA. 1978;75:3737–3741. doi: 10.1073/pnas.75.8.3737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Verdet C, Arlet G, Ben Redjeb S, Ben Hassen A, Lagrange P H, Philippon A. Characterization of CMY-4, an ampC-type plasmid-mediated β-lactamase in a Tunisian clinical isolate of Proteus mirabilis. FEMS Microbiol Lett. 1998;169:235–240. doi: 10.1111/j.1574-6968.1998.tb13323.x. [DOI] [PubMed] [Google Scholar]
- 37.Wu D Y, Ugozzoli L, Pal K B, Wallace R B. Allele-specific enzymatic amplification of beta-globin DNA for diagnosis of sickle cell anemia. Proc Natl Acad Sci USA. 1989;86:2757–2760. doi: 10.1073/pnas.86.8.2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wu P J, Shannon K, Phillips I. Mechanisms of hyperproduction of TEM-1 beta-lactamase by clinical isolates of Escherichia coli. J Antimicrob Chemother. 1995;36:927–939. doi: 10.1093/jac/36.6.927. [DOI] [PubMed] [Google Scholar]