Abstract
Ionising radiation damages DNA directly and indirectly through increased production of reactive oxygen species. Although telomeres have been reported as indicators of radiosensitivity, their maintenance in response to occupational exposure to low radiation doses is still a matter of debate. In this work we aimed to investigate telomere length and structure in hospital workers occupationally exposed to X-rays and to relate these findings to oxidation of biomolecules and chromosome aberrations. Blood samples of exposed participants and matching controls were taken during periodical check-ups. Chromosome aberrations and telomere length and structure were analysed in peripheral blood lymphocytes using Q-FISH, whereas oxidative stress parameters [pro/antioxidant balance (PAB), lipid peroxidation, and 8-oxo-dG] were measured in plasma samples. Based on the CA findings we divided the exposed group into two subgroups, of which one had chromosome aberrations in the first division metaphases and the other did not. There was no significant difference in telomere length between any of the groups. However, both subgroups showed significantly higher rate of fragile telomeres and higher lipid peroxidation product and 8-oxo-dG levels than controls. The rate of fragile telomeres significantly correlated with plasma levels of 8-oxo-dG, which suggests that continuous exposure to low radiation doses induces oxidative base damage of guanine resulting in telomere fragility.
Key words: 8-oxo-dG, chromosome aberrations, lipid peroxidation, telomere fragility, telomere length, X-ray
Abstract
Biološki učinci ionizirajućega zračenja (IZ) pripisuju se oštećenjima DNA i indirektnim učincima kroz povećanu proizvodnju reaktivnih vrsta kisika. Iako se telomere rabe kao pokazatelji radioosjetljivosti, o njihovu ponašanju kao odgovoru na ionizirajuće zračenje u uvjetima profesionalne izloženosti i dalje se raspravlja. U ovom radu željeli smo istražiti duljinu i strukturu telomera u bolničkih radnika koji su profesionalno izloženi ionizirajućem zračenju te povezati te nalaze s oksidacijskim biomolekulama i kromosomskim aberacijama. Uzorci krvi izloženih ispitanika i zdravih kontrola uzeti su za analizu tijekom rutinskoga godišnjeg zdravstvenog pregleda. Osim kromosomskih aberacija, u uzorcima plazme izmjereni su i parametri oksidacijskoga stresa [prooksidacijska/antioksidacijska ravnoteža (PAB), lipidna peroksidacija i 8-okso-dG], a procjena duljine i strukture telomera provedena je metodom Q-FISH na metafaznim kromosomima. Analiza kromosomskih aberacija pokazala je da od 34 ispitanika njih 14 ima kromosomske aberacije (skupina 1), a 20 nije imalo aberacije (skupina 2). Nije bilo značajne razlike u spolu ili dobi ni u duljini telomera između skupina. Međutim, incidencija lomljivih telomera bila je značajno veća u objema skupinama ispitanika izloženih IZ-u u usporedbi s kontrolnim ispitanicima. Produkti peroksidacije lipida i 8-okso-dG također su bili značajno viši u objema skupinama. Učestalost lomljivih telomera u pozitivnoj je korelaciji (statistički značajna) s razinama 8-okso-dG u plazmi, što sugerira da kontinuirano izlaganje niskim dozama ionizirajućeg zračenja izaziva oksidacijsko oštećenje baza, koje bi moglo biti uzrok lomljivosti telomera u profesionalno izloženih osoba. Međutim, potrebna su daljnja istraživanja kako bi se razjasnila uloga lomljivih telomera kao potencijalnih biomarkera za izloženost niskim dozama ionizirajućeg zračenja.
Ključne riječi: 8-okso-dG, duljina telomera, ionizirajuće zračenje, lomljivost telomera, kromosomske aberacije, profesionalna izloženost
Biological effects of long-term exposure to ionising radiation are attributed to direct (nuclear and mitochondrial) and indirect DNA damage through increased oxidative changes in biomolecules. Many biomarkers have been studied so far, including chromosome aberrations (CA), micronuclei (MN), single-strand breaks, phosphorylation of histone H2AX (γ-H2AX), premature chromosome condensation, telomere length, and oxidative DNA damage to find the ones most relevant for occupational exposure to low doses of ionising radiation (1). An extensive meta-analysis by Clemence et al. (2) showed that the most accurate bioindicators are chromosome aberrations (CA) and micronuclei (MN). Chromosome aberrations involving telomeres are a reliable tool to assess misrepaired and unrepaired chromosomal damage and radiosensitivity (3, 4), as telomere length alone is not sensitive enough, because it depends on other confounding factors such as age (5), lifestyle (6), and infection with specific pathogens (7, 8). Telomeres, as specific nucleoprotein structures with high guanine content, are more susceptible to oxidative damage generating single-strand breaks and double-strand breaks, as they shorten more quickly and become fragile (8).
To the best of our knowledge, however, there are no studies investigating telomere fragility in persons occupationally exposed to ionising radiation. As occupational exposure to ionising radiation mostly affects healthcare workers (10), the aim of our study was to investigate telomere length and fragility in this population group working in diagnostic radiology units. Our secondary aim was to see if and how telomere parameters are related to oxidative stress parameters.
Participants and methods
Participants
The study included 34 radiologists and technicians exposed to X-rays at their diagnostic radiology units and 30 healthy unexposed controls. All participants were volunteers recruited during periodical check-ups in agreement with current conventions (11), who signed informed consent. The study was approved by the Ethics Committee of the Vinča Institute of Nuclear Sciences, Belgrade, Serbia.
All participants answered a questionnaire asking for basic demographic information, type of work they do, job duration, diet, and smoking. Occupationally exposed participants wore personal dosimeters on the chest, and the absorbed dose was reviewed every three months.
Blood sampling and study design
From each participant we collected 6 mL of peripheral blood into Li-heparin vacutainers (Becton Dickinson, Plymouth, UK). An aliquot of 0.5 mL was used for cell culture, while the rest of heparinised blood was centrifuged at 1.400 g for 15 min to separate plasma, which was then stored at -80 °C and filtered after thawing through a Minisart filter with 0.45 μm pore diameter (Sartorius, Göttingen, Germany) before we measured oxidative stress parameters.
One set of slides with harvested lymphocytes was used for classical chromosome aberration analysis and the other set for telomere quantitative FISH (Q-FISH). Data of exposed participants who showed chromosome aberrations (N=14) were separated from those without chromosome aberrations (N=20) for further statistical analysis and comparison of all data (telomerase length, percent of fragile telomeres, PAB, lipid peroxidation products, and 8-oxo dG) between these two subgroups and control.
Chromosome aberration (CA) assay
Lymphocytes for the chromosome aberration test were obtained by culturing aliquots of heparinised whole blood with the Gibco® PB-MaxTM karyotyping medium (Catalogue No. 12557-021, ThermoFisher Scientific, Life Technologies Corporation, Grand Island, NY, USA) at 37 °C for 48 h. In the last three hours, the cultures were added colchicine (Sigma-Aldrich Co., St. Louis, MO, USA) in the final concentration of 2.5 μg/mL according to the standard procedure described elsewhere (12).
Slides were analysed under a Zeiss Axio Imager A1 microscope (Carl Zeiss, Jena, Germany) with the ISIS imaging software package (MetaSystems Hard & Software GmbH, Altlussheim, Germany). At least 200 metaphase spreads were analysed at 10–100× magnification for chromosome aberrations (chromosome and chromatid breaks, dicentric and ring chromosomes, acentric fragments, stable aberrations, and radial figures) (13).
Telomere quantitative fluorescent in situ hybridisation (Q-FISH)
Even though telomere length can be assessed with quantitative polymerase chain reaction (qPCR) thanks to its high throughput (14), its inter and intra-sample variability may exceed 10 % (15), which is why we opted for Q-FISH as a more consistent method of telomere fragility assessment. We assessed lymphocyte chromosome metaphase spreads on slides using the Cy3-labelled peptide nucleic acid (PNA) telomere probe supplemented with PNA centromere probe for chromosome 2 (DAKO, Glostrup, Denmark) as previously described in detail (16). Denatured slides were hybridised in a dark humidified chamber for 2 h, then washed in 70 % formamide solution (Carlo Erba Reagents, Val-de-Reuil, France), dehydrated in a series with ethanol, and counterstained with 4′,6′-diamidino-2-phenylindole (DAPI)-containing mounting medium (Vector Laboratories, Peterborough, UK). Telomere length and fragility (split, fused, or no signal telomeres) were analysed on a Zeiss Axioimager A1 microscope (Carl Zeiss) equipped with a telomere measurement tool for ISIS software (MetaSystems Hard & Software GmbH) with centromere fluorescence set to 100 %. Telomere length was expressed as the ratio between the actual telomere and set centromere signal intensity (T/C ratio) (17). At least 20 metaphase spreads per slide were analysed. The rate of fragile telomeres was expressed as the ratio between the number of fragile telomeres and total number of analysed metaphase spreads.
Prooxidant/antioxidant balance (PAB) assay
The PAB assay followed the standard protocol described elsewhere in detail (18). The assay measures differences in two ongoing reactions in the same sample: oxidation of chromogen 3,3’,5,5’-tetramethylbenzidine (TMB) by peroxides and reduction of coloured cation by antioxidants. The absorbance was measured colourimetrically on a Sunrise absorbance microplate reader at 450 nm (cut-off at 570 nm) (Tecan Group Ltd, Männedorf, Switzerland). The results are presented as arbitrary Hamidi-Koliakos (HK) units (percentage of hydrogen peroxide in the standard solution).
8-oxo-7,8-dihydro-2′-deoxyguanosine (8-oxo-dG) assay
8-oxo-dG was measured using the HT 8-oxo-dG ELISA Kit (Trevigen®, Gaithersburg, MD, USA) as described by Haghdoost et al. (19). Briefly, the reaction is based on competitive reaction between the sample and 8-oxo-dG monoclonal antibody to bind to the immobilised 8-oxo-dG on pre-coated wells. Plasma levels of 8-oxo-dG are inversely proportional to the product formation detected using a horseradish peroxidase conjugate and colourimetric substrate. The absorbance was measured colourimetrically on a Sunrise absorbance microplate reader at 450 nm (cut-off at 570 nm) and the results presented as nmol/L of 8-oxo-dG.
Lipid peroxidation product (LPP) assay
Lipid peroxidation products were measured according to the method of Tsikas (20). The assay is based on the reaction of the chromogen N-methyl-2-phenylindole with malondialdehyde (MDA) and 4-hydroxyalkenals (HNE). The reaction product was measured colourimetrically on a Sunrise absorbance microplate reader at 586 nm wavelength (cut-off at 570 nm) and the results presented as nmol/mL of plasma LPP using a standard curve of 1,1,3,3-tetramethoxypropane.
Statistical analysis
The results are presented as means ± standard error of the mean (SEM). The groups were compared and relations between parameters analysed with the Mann-Whitney U test and Spearman correlation coefficient using the SPSS 10 for Windows (IBM, Armonk, NY, USA). The level of significance was set to p<0.05.
Results and discussion
Occupationally exposed and control participants did not significantly differ in age, gender, or job duration (Table 1).
Table 1.
General characteristics of study participants (N=64)
| Participants (N) | Annual TLD dose (mSv) | Male | Female | Age Mean±SD (years) | Job duration Mean±SD (years) | Smokers (N) | |
|---|---|---|---|---|---|---|---|
| Occupationally exposed to ionising radiation | 34 | 5.2±3.6 | 8 | 26 | 35.3±8.2 | 20.2±4.2 | 14 |
| Radiologists | 4 | 4.83±4.44 | 1 | 3 | 36±3.1 | 12.7±1.9 | 1 |
| Technicians | 30 | 5.25±3.46 | 7 | 23 | 43±3.7 | 24.6±2.3 | 13 |
| Controls | 30 | - | 7 | 23 | 39.5±6.8 | 21.8±5.1 | 12 |
| Total | 64 | - | 15 | 49 | 37.8±6.7 | - | 26 |
TLD – thermoluminescent dosimeter
Of the 34 exposed participants, 14 had chromosome aberrations in first division metaphases (Table 2), including dicentric chromosomes with accompanying fragments, excesses acentrics, and chromosome and chromatid breaks. Dicentric chromosomes without accompanying fragments and pericentric inversions were also detected, both representing long-lived chromosome aberrations from past exposure (21, 22). Numerous studies support our findings of increased rate of chromosome aberrations and high interindividual variability (23, 24, 25), which is attributed to differences in genetic susceptibility towards ionising radiation (26, 27, 28, 29, 30, 31, 32), activation of DNA repair (33, 34), and anti-inflammatory processes (35). Lymphocyte chromosome aberrations in our occupationally exposed participants whose annual doses did not exceed risk limits can also be attributed to cumulative effects of chronic exposure to low-dose ionising radiation (36).
Table 2.
Chromosome aberrations, telomere length and fragility, and oxidative stress parameters by study groups
| Exposed participants with CAs | Exposed participants without CAs | Controls | |
|---|---|---|---|
| 14 | 20 | 30 | |
| No. of dicentrics* | 4 | ||
| No. of excess acentrics* | 9 | ||
| No. of chromosome breaks* | 9 | ||
| No. of chromatid breaks* | 7 | 3 | 3 |
| No. of stable chromosome aberrations* | 2 | ||
| Average rate of breaks per cell | 0.139 | ||
| Telomere length (T/C ratio) | 40.84±1.63 | 43.54±1.08 | 41.71±1.78 |
| Rate of fragile telomeres/cell | 0.64±0.13c | 0.15±0.01c,d | 0.01±0.004 |
| PAB | 21.7±1.42 | 21.91±0.835 | 21.69±0.34 |
| LPP | 5.88±0.38a | 7.58±0.46b,c | 4.59±0.16 |
| 8-oxo dG | 100.85±7.6c | 122.41±8.15c | 45.15±1.62 |
* per 200 first division metaphases; a significantly different from control at p<0.05; b significantly different from the exposed group with CAs at p<0.05; c significantly different from control at p<0.001; d significantly different from the exposed group with CAs at p<0.001
We found no significant differences in average relative telomere length between the groups. However, the rate of fragile telomeres was significantly higher in both exposed subgroups than control. Figure 1 shows that telomeres of chromosomes 1p, 5p, 4q, and 7q were the longest and of chromosomes Yq, 21p, and 22p the shortest in the exposed subgroup with chromosome aberrations. The rate of split telomere signals (fragile telomeres) was the highest with the longest telomeres 5p, 7q (9.33 % of the total number of either 5p or 7q fragile telomeres), and 2p (8 %), while the involvement of all other chromosomes, including autosomes and sex chromosomes, was below 4 % of the total fragile telomere count. In one participant from the exposed subgroup with chromosome aberrations we noticed telomere fusion between chromosomes 1p and 16q.
Figure 1.
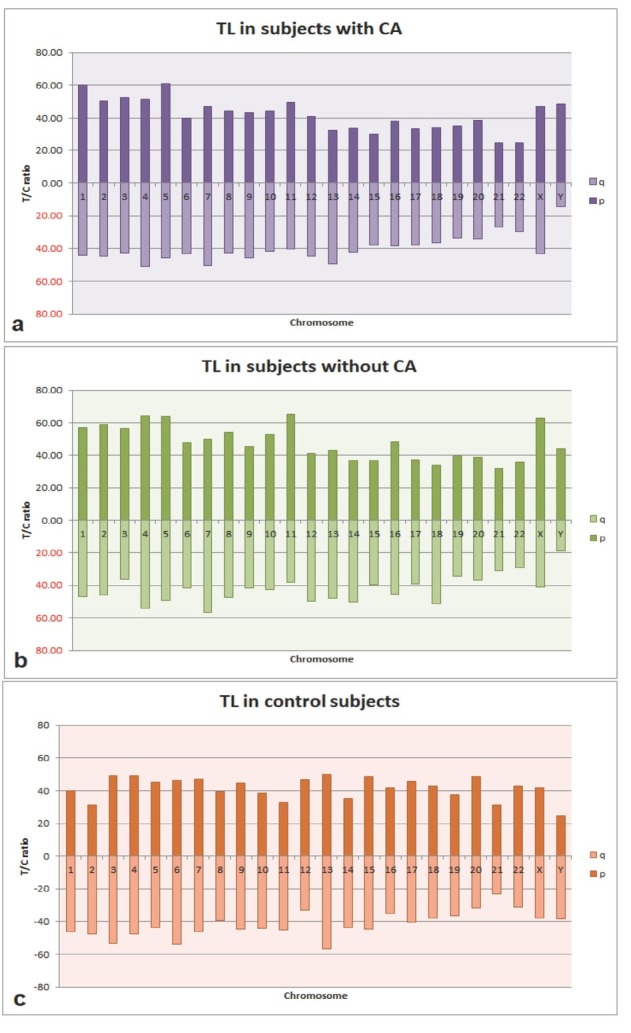
Telomere length of individual chromosomes in the exposed subgroup with chromosome aberrations (a), exposed subgroup without chromosome aberrations (b), and controls (c)
The exposed subgroup without chromosome aberrations had the longest telomeres on chromosomes 4p, 5p, 11p, and Xp. Even though only 0.15±0.01 of fragile telomeres per cell were present, this subgroup showed a similar trend in telomere fragility, i.e., telomere fragility in chromosomes 4q and 11p was 27.27 % and 18.18 %, respectively, while telomere fragility in all other chromosomes was considerably lower. Unlike the exposed subgroups, controls had a much more even distribution of telomere lengths across chromosomes, as we did not observe any extremely long (>60 T/C) or extremely short telomeres (<20 T/C).
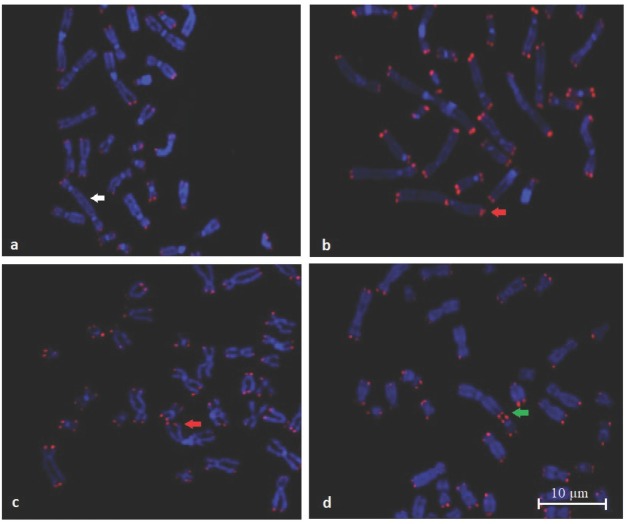
Figure 2 shows photomicrographs representative of telomere fragility, including the lack of telomere signal and fusion as an exchange of two independent double-strand breaks between two different chromosomes.
Figure 2.
Representative photomicrographs of fragile telomeres (Q-FISH). White arrow indicates a dicentric chromosome without telomere signal (a). Red arrow indicates fragile telomeres (b, c). Green arrow indicates telomere fusion between two chromosomes (d)
Accelerated telomere shortening has been reported in Chernobyl clean-up workers (37), whereas in citizens who lived in areas with high natural background radiation no such shortening was found (38).
The most interesting finding in our study is that the most fragile telomeres in participants occupationally exposed to ionising radiation were the longest ones. This phenomenon is perhaps best explained by the activation of a specific homology directed repair pathway known as mitotic DNA synthesis (MiDAS), which is a form of break-induced replication activated in response to replicative stress at loci difficult to replicate, such as telomeres (39, 40, 41). It was recently demonstrated that the highest levels of basal telomere MiDAS occur in cells with the longest telomeres and that 8-oxo-dG in telomere regions activates MiDAS instead of shortening telomeres, which results in their fragility (9, 42). This mechanism in response to radiation-induced oxidative damage might be an attempt to prevent genomic instability (43).
To the best of our knowledge, this is the first study of telomere fragility in persons occupationally exposed to ionising radiation. In view of the fact that dysfunctional telomeres coincide with many late effects or radiation (44, 45), we believe our findings provide one more argument to use telomere fragility as a biomarker of health risks associated with occupational exposure to ionising radiation.
As for oxidative stress parameters, PAB values in the exposed subgroups did not significantly differ from control, whereas the average LPP levels were significantly higher (almost double), which is in line with earlier reports (46, 47). What surprised us, however, was that LPP levels were significantly lower in the exposed subgroup showing chromosome aberrations than in the subgroup without chromosome aberrations. This may be owed to accelerated removal of oxidised nucleotides by base excision repair coupled with other repair pathways (48).
Our findings of significantly higher 8-oxo-dG in the exposed than control participants were expected, as earlier studies have shown that the highest levels of oxidised guanine are expected on telomeres. 8-oxo-dG induces single-strand breaks throughout the genome, which are usually completely repaired except in the telomere regions (41, 42).
While telomere length did not correlate with either LPP or 8-oxo-dG levels, the rate of fragile telomeres correlated with 8-oxo-dG levels (p<0.01, r=0.655), which suggests that oxidative damage of guanine could be the main cause of telomere fragility in people occupationally exposed to ionising radiation. LPP levels also significantly correlated with 8-oxo-dG (r=0.644, p<0.01). El-Benhawy et al. (50) claim to be the first to have established a correlation between 8-oxo-dG and chromosome aberrations in persons occupationally exposed to ionising radiation, but they did not look into telomere fragility. We therefore believe that our study contributes to the current debate about telomere response to occupational radiation exposure and associated health risks by arguing in favour of telomere fragility and lipid peroxidation products as potentially reliable biomarkers of increased health risk in occupations exposed to ionising radiations, but more studies are needed to corroborate our findings.
Conclusion
Our study clearly shows that occupational exposure to low doses of ionising radiation makes telomeres fragile, most likely through oxidative damage of guanine and activation of base excision repair-induced pathways in response to oxidative or replicative stress.
Acknowledgements
This research was funded by the Ministry of Education, Science and Technological Development of the Republic of Serbia (grant No. 451-03-9/2021-14/200017).
Footnotes
Conflicts of interest
None to declare.
References
- 1.Pernot E, Hall J, Baatout S, Benotmane MA, Blanchardon E, Bouffler S, Saghire H El, Gomolka M, Guertler A, Harms-Ringdahl M, Jeggo P, Kreuzer M, Laurier D, Lindholm C, Mkacher R, Quintens R, Rothkamm K, Sabatier L, Tapio S, de Vathaire F, Cardis E. Ionizing radiation biomarkers for potential use in epidemiological studies. Mutat Res. 2012;751:258–86. doi: 10.1016/j.mrrev.2012.05.003. [DOI] [PubMed] [Google Scholar]
- 2.Clemence B, Bernier MO, Klokov D, Andreassi MG. Biomarkers of genotoxicity in medical workers exposed to low-dose ionizing radiation: systematic review and meta-analyses. Int J Mol Sci. 2021;22:7504. doi: 10.3390/ijms22147504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cubby E, Newton C, Joksić G, Woodbine L, Koller B, Jeggo PA, Slijepčević P. Accelerated telomere shortening in radiosensitive cell lines. Radiat Res. 2005;164:53–62. doi: 10.1667/rr3376. [DOI] [PubMed] [Google Scholar]
- 4.Ayouaz A, Raynaud C, Heride C, Revaud D, Sabatier L. Telomeres: hallmarks of radiosensitivity. Biochimie. 2008;90:60–72. doi: 10.1016/j.biochi.2007.09.011. [DOI] [PubMed] [Google Scholar]
- 5.Vaiserman A, Krasnienkov D. Telomere length as a marker of biological age: state-of-the-art, open issues, and future perspectives. Front Genet. 2021;11:630186. doi: 10.3389/fgene.2020.630186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mayer SE, Prather AA, Puterman E, Lin J, Arenander J, Coccia M, Shields GS, George M, Slavich GM, Epel ES. Cumulative lifetime stress exposure and leukocyte telomere length attrition: The unique role of stressor duration and exposure timing. Psychoneuroendocrinology. 2019;104:210–8. doi: 10.1016/j.psyneuen.2019.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bellon M, Nicot C. Telomere dynamics in immune senescence and exhaustion triggered by chronic viral infection. Viruses. 2017;9:289. doi: 10.3390/v9100289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Noppert GA, Feinstein L, Dowd Jennifer B, Stebbins RC, Zang E, Needham BL, Meier HCS, Simanek A, Aiello AE. Pathogen burden and leukocyte telomere length in the United States. Immun Ageing. 2020;17:36. doi: 10.1186/s12979-020-00206-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Barnes RP, de Rosa M, Thosar SA, Detwiler AC, Roginskaya V, Van Houten B, Bruchez MP, Stewart-Ornstein J, Opresko PL. Telomeric 8-oxoguanine drives rapid premature senescence in the absence of telomere shortening. bioRxiv. 2021;2021:442662. doi: 10.1101/2021.05.05.442662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.United Nations, Scientific Committee on the effects of Atomic Radiation. Sources and Effects of Ionizing Radiation. UNSCEAR 2008 Report of the General Assembly with Scientific Annexes. New York (NY): United Nations; 2010. [Google Scholar]
- 11.Zakon o zdravstvenoj zaštiti [Law on health care, in Serbian] Službeni glasnik RS. 2019;25 [Google Scholar]
- 12.Biological Dosimetry: Chromosomal Aberration Analysis for Dose Assessment. Technical Reports Series No. 260. Vienna: IAEA;; 1986. International Atomic Energy Agency (IAEA) [Google Scholar]
- 13.Cytogenetic Analysis for Radiation Dose Assessment. Technical Reports Series No. 405. Vienna: IAEA;; 2001. International Atomic Energy Agency (IAEA) [Google Scholar]
- 14.Lin J, Smith DL, Esteves K, Drury S. Telomere length measurement by qPCR - Summary of critical factors and recommendations for assay design. Psychoneuroendocrinology. 2019;99:271–8. doi: 10.1016/j.psyneuen.2018.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Aubert G, Hills M, Lansdorp PM. Telomere length measurement caveats and critical assessment of the available technologies and tools. Mutat Res. 2012;730:59–67. doi: 10.1016/j.mrfmmm.2011.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Joksic G, Joksic I, Filipović J, Liehr T. Liehr T. Fluorescence in situ hybridization (FISH): application guide. Berlin, Heidelberg: Springer; 2017. Telomere length measurement by FISH; pp. 147–52. –. [Google Scholar]
- 17.Perner S, Brüderlein S, Hasel C, Waibel I, Holdenried A, Ciloglu N, Chopurian H, Vang Nielsen K, Plesch A, Högel J, Möller P. Quantifying telomere lengths of human individual chromosome arms by centromere calibrated fluorescence in situ hybridization and digital imaging. Am J Pathol. 2003;163:1751–6. doi: 10.1016/S0002-9440(10)63534-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Alamdari DH, Paletas K, Pegiou T, Sarigianni M, Befani C, Koliakos G. A novel assay for the evaluation of the prooxidant-antioxidant balance, before and after antioxidant vitamin administration in type II diabetes patients. Clin Biochem. 2007;40:248–54. doi: 10.1016/j.clinbiochem.2006.10.017. [DOI] [PubMed] [Google Scholar]
- 19.Haghdoost S, Czene S, Näslund I, Skog S, Harms-Ringdahl M. Extracellular 8-oxo-dG as a sensitive parameter for oxidative stress in vivo and in vitro. Free Radic Res. 2005;39:153–62. doi: 10.1080/10715760500043132. [DOI] [PubMed] [Google Scholar]
- 20.Tsikas D. Assessment of lipid peroxidation by measuring malondialdehyde (MDA) and relatives in biological samples: Analytical and biological challenges. Anal Biochem. 2017;524:13–30. doi: 10.1016/j.ab.2016.10.021. [DOI] [PubMed] [Google Scholar]
- 21.Natarajan AT, Boei JJ. Formation of chromosome aberrations: insights from FISH. Mutat Res. 2003;544:299–304. doi: 10.1016/j.mrrev.2003.09.005. [DOI] [PubMed] [Google Scholar]
- 22.Tucker JD. Low-dose ionizing radiation and chromosome translocations: A review of the major consideration for human biological dosimetry. Mutat Res. 2008;659:211–20. doi: 10.1016/j.mrrev.2008.04.001. [DOI] [PubMed] [Google Scholar]
- 23.Frequency of chromosomal lesions and damaged lymphocytes of workers occupationally exposed to x-rays. Health Phys. 2005;88:334–9. doi: 10.1097/01.hp.0000149920.09402.1d. Milačić S. [DOI] [PubMed] [Google Scholar]
- 24.Jovicic D, Milacic S, Milic N, Bukvic N, Vukov TD. Chromosomal aberrations in subjects exposed to ionizing radiation. J Environ Pathol Toxicol Oncol. 2009;28:75–82. doi: 10.1615/JEnvironPatholToxicolOncol.v28.i1.80. [DOI] [PubMed] [Google Scholar]
- 25.Zakeri F, Hirobe T. A cytogenetic approach to the effects of low levels of ionizing radiations on occupationally exposed individuals. Eur J Radiol. 2010;73:191–5. doi: 10.1016/j.ejrad.2008.10.015. [DOI] [PubMed] [Google Scholar]
- 26.Lin C-C, Wu LS-H, Lee K-F. The potential effect of different doses of ionizing radiation on genes and disease. Dose-Response. 2019;17:1559325819843375. doi: 10.1177/1559325819843375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Il’yasova D, Kinev A, Melton CD, Davis GF. Donor-specific cell-based assays in studying sensitivity to low-dose radiation: a population-based perspective. Front Public Health. 2014;2:244. doi: 10.3389/fpubh.2014.00244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kato TA, Wilson PF, Nagasaw H, Peng Y, Weil MM, Little JB, Bedford JS. Variations in radiosensitivity among individuals: a potential impact on risk assessment? Health Phys. 2009;97:470–80. doi: 10.1097/HP.0b013e3181b08eee. [DOI] [PubMed] [Google Scholar]
- 29.Wilson PF, Nham PB, Urbin SS, Hinz JM, Jones IM, Thompson LH. Inter-individual variation in DNA double-strand break repair in human fibroblasts before and after exposure to low doses of ionizing radiation. Mutat Res. 2010;683:91–7. doi: 10.1016/j.mrfmmm.2009.10.013. [DOI] [PubMed] [Google Scholar]
- 30.Piotrowski I, Dawid A, Kulcenty K, Suchorska WM. Use of biological dosimetry for monitoring medical workers occupationally exposed to ionizing radiation. Radiation. 2021;1:95–115. doi: 10.3390/radiation1020009. [DOI] [Google Scholar]
- 31.Andreassi MG, Foffa I, Manfredi S, Botto N, Cioppa A, Picano E. Genetic polymorphisms in XRCC1, OGG1, APE1 and XRCC3 DNA repair genes, ionizing radiation exposure and chromosomal DNA damage in interventional cardiologists. Mutat Res. 2009;666:57–63. doi: 10.1016/j.mrfmmm.2009.04.003. [DOI] [PubMed] [Google Scholar]
- 32.Vellingiri B, Shanmugam S, Subramaniam MD, Balasubramanian B, Meyyazhagan A, Alagamuthu K, Prakash V, Shafiahammedkhan M, Kathannan S, Pappuswamy M, Raviganesh B, Anand S, Shahnaz ND, Cho SG, Keshavarao S. Cytogenetic endpoints and xenobiotic gene polymorphism in lymphocytes of hospital workers chronically exposed to ionizing radiation in cardiology, radiology and orthopedic laboratories. Ecotoxicol Environ Saf. 2014;100:266–74. doi: 10.1016/j.ecoenv.2013.09.036. [DOI] [PubMed] [Google Scholar]
- 33.Hou J, Wang F, Kong P, Yu PKN, Wang H, Han W. Gene profiling characteristics of radioadaptive response in AG01522 normal human fibroblasts. PLoS One. 2015;10:e0123316. doi: 10.1371/journal.pone.0123316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Niazi Y, Thomsen H, Smolkova B, Vodickova L, Vodenkova S, Kroupa M, Vymetalkova V, Kazimirova A, Barancokova M, Volkovova K, Staruchova M, Hoffmann P, Nöthen MM, Dusinska M, Musak L, Vodicka P, Försti A, Hemminki K. DNA repair gene polymorphisms and chromosomal aberrations in healthy, nonsmoking population. DNA Repair. 2021;101:103079. doi: 10.1016/j.dnarep.2021.103079. [DOI] [PubMed] [Google Scholar]
- 35.Ebrahimian TG, Beugnies L, Surette J, Priest N, Gueguen Y, Gloaguen C, Benderitter M, Jourdain JR, Tack K. Chronic exposure to external low-dose gamma radiation induces an increase in anti-inflammatory and anti-oxidative parameters resulting in atherosclerotic plaque size reduction in ApoE-/- mice. Radiat Res. 2018;189:187–96. doi: 10.1667/RR14823.1. [DOI] [PubMed] [Google Scholar]
- 36.Fučić A, Želježić D, Kašuba V, Kopjar N, Rozgaj R, Lasan R, Mijić A, Hitrec V, Lucas JN. Stable and unstable chromosome aberrations measured after occupational exposure to ionizing radiation and ultrasound. Croat Med J. 2007;48:371–7. PMCID: PMC2080539. [PMC free article] [PubMed] [Google Scholar]
- 37.Reste J, Zvigule G, Zvagule T, Kurjane N, Eglite M, Gabruseva N, Berzina D, Plonis J, Miklasevics E. Telomere length in Chernobyl accident recovery workers in the late period after the disaster. J Radiat Res. 2014;55:1089–100. doi: 10.1093/jrr/rru060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Movahedi A, Mostajaboddavati M, Rajabibazl M, Mirfakhraie R, Enferadi M. Association of telomere length with chronic exposure to ionizing radiation among inhabitants of natural high background radiation areas of Ramsar, Iran. Int J Radiat Biol. 2019;95:1113–21. doi: 10.1080/09553002.2019.1605460. [DOI] [PubMed] [Google Scholar]
- 39.Blackburn EH. Switching and signaling at the telomere. Cell. 2001;106:661–73. doi: 10.1016/S0092-8674(01)00492-5. [DOI] [PubMed] [Google Scholar]
- 40.Yang Z, Takai KK, Lovejoy CA, de Lange T. Break-induced replication promotes fragile telomere formation. Genes Dev. 2020;34:1392–405. doi: 10.1101/gad.328575.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Flynn RL, Heaphy CM. Surviving telomere attrition with the MiDAS touch. Trends Genet. 2019;35:783–5. doi: 10.1016/j.tig.2019.08.008. [DOI] [PubMed] [Google Scholar]
- 42.Özer Ö, Hickson ID. Pathways for maintenance of telomeres and common fragile sites during DNA replication stress. Open Biol. 2018;8(4):180018. doi: 10.1098/rsob.180018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hayflick L. Living forever and dying in the attempt. Exp Gerontol. 2003;38:1231–41. doi: 10.1016/j.exger.2003.09.003. [DOI] [PubMed] [Google Scholar]
- 44.Bolzan AD. Using telomeric chromosomal aberration to evaluate clastogen-induced genomic instability in mammalian cells. Chromose Res. 2020;28:259–76. doi: 10.1007//s10577-020-09641-2. [DOI] [PubMed] [Google Scholar]
- 45.Luxton JJ, McKenna MJ, Lewis AM, Taylor LE, Jhavar SG, Swanson GP, Bailey SM. Telomere length dynamics and chromosomal instability for predicting individual radiosensitivity and risk via machine learning. J Pers Med. 2021;11(3):188. doi: 10.3390/jpm11030188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ahmad IM, Temme JB, Abdalla MY, Zimmerman MC. Redox status in workers occupationally exposed to long-term low levels of ionizing radiation: A pilot study. Redox Rep. 2016;21:139–45. doi: 10.1080/13510002.2015.1101891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Chen B, Dai Q, Zhang Q, Yan P, Wang A, Qu L, Jin Y, Zhang D. The relationship among occupational irradiation, DNA methylation status and oxidative damage in interventional physicians. Medicine. 2019;98:e17373. doi: 10.1097/MD.0000000000017373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kamiya H. Mutagenicity of oxidized DNA precursors in living cells: Roles of nucleotide pool sanitization and DNA repair enzymes, and translesion synthesis DNA polymerases. Mutat Res G Tox Env Mut. 2010;703:32–6. doi: 10.1016/j.mrgentox.2010.06.003. [DOI] [PubMed] [Google Scholar]
- 49.Fouquerel E, Barnes RP, Uttam S, Watkins SC, Bruchez MP, Opresko PL. Targeted and persistent 8-oxoguanine base damage at telomeres promotes telomere loss and crisis. Mol Cell. 2019;75:117–30. doi: 10.1016/j.molcel.2019.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.El-Benhawy SA, Sadek NA, Behery AK, Issa NM, Ali OK. Chromosomal aberrations and oxidative DNA adduct 8-hydroxy-2-deoxyguanosine as biomarkers of radiotoxicity in radiation workers. J Radiat Res Appl Sci. 2016;9:249–58. doi: 10.1016/j.jrras.2015.12.004. [DOI] [Google Scholar]