Abstract
The prevalence of and mortality from non-tuberculous mycobacteria (NTM) infections have been steadily increasing worldwide. Most NTM infections are caused by Mycobacterium avium-intracellulare complex (MAC). MAC can escape from killing by neutrophils, which are professional phagocytes. However, the involvement of neutrophils in the pathogenesis of MAC infection is poorly understood. The present study assessed the roles of neutrophil extracellular trap (NET) formation in neutrophil defense mechanisms against infection with MAC strains, including M. avium isolated from patients with severe or mild lung tissue destruction. Although all MAC induced NET formation, non-pathogenic mycobacteria (M. gordonae and M. smegmatis) slightly but not significantly induced NET formation. Peptidylarginine deiminase 4 (PAD4) inhibitor reduced MAC-induced NET formation but did not affect MAC escape from neutrophils. PAD4 inhibition attenuated the MAC-induced matrix metalloproteinase (MMP)-8 and 9 release to the levels of MMPs from non-pathogenic mycobacteria. MAC also induced interleukin (IL)-8 release by neutrophils, a process independent of MAC-induced NET formation. Taken together, these findings suggest that MAC induce NET formation, IL-8 release and NETs-dependent release of MMP-8 and -9 from neutrophils, leading to neutrophil accumulation and further inflammation, thereby enhancing the progression of infection in the lungs.
Subject terms: Infection, Mechanisms of disease
Introduction
In recent years, the prevalence of1,2 and mortality from3,4 non-tuberculous mycobacteria (NTM) infections have increased worldwide, emphasizing the importance of determining the mechanisms of lung tissue destruction in patients with NTM infection. Most NTM infections are caused by Mycobacterium avium-intracellulare complex (MAC). These bacteria are distributed throughout a broad range of environments, including water, soil, and dust particles5. MAC mainly infects the lungs and induces long-term inflammation5, involving pathological changes to lung tissue, such as granulation, cavitation, and bronchiectasis6,7, and leading to lung tissue destruction. For example, exacerbated lung tissue destruction and CT images showing cavitation and bronchiectasis have been observed in MAC-infected patients8,9. The possibility of rapid lung destruction in some MAC patients has led to the initiation of immediate antimicrobial treatment for patients with pulmonary cavities or extensive disease10,11. In contrast, some MAC patients do not show lung destruction, even without antimicrobial treatments12. These findings suggest that host factors and/or susceptibility to MAC can affect the progression of lung tissue destruction.
Neutrophils are the most abundant leukocytes in blood and play a central role in innate immune responses against mycobacterial infection13,14. These cells are immediately recruited to the site of injury or infection through chemokines such as interleukin-8 (IL-8) and leukotriene B415. Neutrophils kill pathogens via several major pathways, including phagocytosis, superoxide generation, degranulation and the release of neutrophil extracellular traps (NETs)16. NETs manifest as large, extracellular, web-like structures of chromatin filaments composed of histones, proteases, and granule proteins17. In addition to microorganisms, proinflammatory host molecules, such as IL-8, induce NET formation16,18. NET is thought to prevent microbial dissemination by trapping them in DNA web-like structures and killing them through antimicrobial molecules17. NET-associated molecules, however, may induce uncontrolled inflammatory responses causing tissue damage16. Although in vitro studies using human neutrophils19, as well as studies using human lung pathological samples20, blood samples21 and sputum22 have shown that M. tuberculosis induces NET formation, little is known about MAC-induced NET formation.
Interstitial collagen is the most abundant macromolecule constituent of the large airways, blood vessels, and alveolar interstitium in the lungs23. Lung tissue destruction requires the destruction of such collagen. Matrix metalloproteinases (MMPs) are a family of zinc-dependent endoproteinases that degrade various types of collagen24. In human neutrophils, MMP-8 and MMP-9 are stored as latent enzymes (pro-MMP-8 and pro-MMP-9) within specific granules and gelatinase granules, respectively25. MMPs play an important role in many biological processes, including cell proliferation, migration, and differentiation, remodeling of the extracellular matrix, and tissue invasion and vascularization. These biological processes take place multiple times in vivo. However, if not balanced, MMPs can also contribute to harmful pathological conditions24, including the pathogenesis of several respiratory diseases, such as idiopathic pulmonary fibrosis26, emphysema27 and pulmonary tuberculosis28.
The present study was designed to assess the relationship between MAC pathogenicity and NET formation by examining MAC-induced NET formation and the anti-mycobacterial activity of NETs. Furthermore, mechanisms underlying the release of neutrophil granules and cytokine production during MAC-induced NET formation were investigated.
Materials and methods
Reagents and antibodies
Alexa Fluor 633-protein labeling kits (A20170), TO-PRO3 (T3605), Sytox orange (S11368), 3, 3', 5, 5'-tetramethylbenzidine (TMB) and sulfuric acid for use with the TMB substrate were from Invitrogen (Thermo Fisher Scientific, Waltham, MA, USA). Antibodies to human citrullinated histone H3 (cit-H3; ab5103) and myeloperoxidase (MPO; ab25989), Alexa Fluor 488 (ab150077), Alexa Fluor 568 (ab175473), and GSK106 (ab229184) were from abcam (Cambridge, UK). Antibody to human H3 (#4499) was from Cell Signaling Technology (Danvers, MA, USA); antibody to human β-actin antibody was from Sigma-Aldrich (St. Louis, MO, USA), and A23187 (11016) and GSK484 (17488) were from Cayman (Ann Arbor, MI, USA).
Isolation of human neutrophils
Ethical approval for obtaining blood from healthy human volunteers was provided by the Ethics Review Board of Juntendo University Faculty of Medicine (Authorization number: 2019073). All research was performed in accordance with relevant guidelines/regulations, and written informed consent was obtained from all participants.
Human neutrophils were isolated from heparinized peripheral blood of healthy volunteer subjects by Polymorphprep™ (Nycomed Pharma; Oslo, Norway) centrifugation. Neutrophil populations were > 95% pure as determined by Turk's solution staining (FUJIFILM Wako Pure Chemical Corporation; Osaka, Japan).
Bacterial strains and culture conditions
MAC were obtained from stocks clinically isolated from non-HIV-infected patients at Juntendo University Hospital and Juntendo University Urayasu Hospital and identified as M. intracellulare and M. avium by DNA-DNA hybridization methods. The severity of lung tissue destruction was evaluated using a previously described scoring system29. Mycobacteria isolated from patients with severe lung tissue destruction and a critical clinical course was defined M. avium s whereas mycobacteria isolated from patients with mild lung tissue destruction and a favorable clinical course was define M. avium m. M. gordonae GTC 612 was from Gifu University, Japan. Mycobacteria were cultured in Middlebrook 7H9 broth (Difco Laboratories, BD), supplemented with OADC (Becton Dickinson).
Labeling of mycobacteria with Alexa Fluor dye
Mycobacteria were suspended in phosphate-buffered saline (PBS). To each suspension was added a 100 μl aliquot of 1.0 M sodium hydrogen carbonate (NaHCO3), followed by incubation with Alexa Fluor 633 dye for 2 h at room temperature (RT) and extensive washing with PBS.
Examination of NETs by confocal microscopy
Neutrophils, seeded on 0.01% poly L lysine (#0403, ScienCell) coated glass base dishes, were pre-incubated with the indicated inhibitors for 30 min at 37 °C, followed by incubation with mycobacteria at a multiplicity of infection (MOI) of 1 for 4 h at 37 °C under 5% CO2 atmosphere. PMA (20 nM) and A-23187 (1 µM) were used as pharmacological inducers of NET formation. The cells were subsequently fixed with 2% paraformaldehyde, permeabilized with 0.5% Triton X-100, and incubated for 2 h with 2% bovine serum albumin and 5% normal goat serum to prevent nonspecific binding. Then cells were incubated with antibodies against cit-H3 (ab5103, Abcam) and MPO (ab25289, Abcam), followed by incubation with the secondary antibodies (Alexa Fluor 488 (ab150077, Abcam) and Alexa Fluor 568(ab175473, Abcam). DNA was stained with TO-PRO3 (T3605, Invitrogen) or SYTOX orange (S11368, Invitrogen). Cells and NET formation were monitored using a STED microscope Leica TCS SP5(Leica Microsystems GmbH, Wetzlar, Germany).
Quantification of NET formation using confocal images
NET formation was quantitated using confocal images by determining the distribution of the nuclear area30,31 and the percentage of cit-H3 positive cells32. Neutrophils positive for cit-H3 were counted and divided by the number of TO-PRO3-positive neutrophils to determine the percentage of cit-H3 positive cells. Quantification by the distribution of nuclear area is based on changes in nuclear size and neutrophil shape during NET formation. TO-PRO3 images of nuclei were analyzed using ImageJ image processing software (US National Institutes of Health). Each sample consisted of 300 cells, and the TO-PRO3-positive area was individually measured in each sample. The distribution of the number of cells across the range of nuclear area was determined using the frequency function in Excel (Microsoft). The data were plotted as the percentage of cells positive for TO-PRO3 for each DNA area range (Supplementary Fig. 1). TO-PRO3-positive areas ranged from 30–150 µm2 for unstimulated neutrophils, with the positive areas for > 95.6% of cells being < 120 µm2. The TO-PRO3-positive areas of PMA- and A-23187-treated cells were expanded up to 350 µm2. Thus, NET formation was defined as cells with nuclear areas > 120 µm2.
Cytokine/chemokine array
Neutrophils were pre-incubated in the presence or absence of GSK484 and stimulated with mycobacteria at a MOI of 1 for 4 h at 37 °C. The culture supernatants were collected and subjected to cytokine array analysis (ARY005B, R&D Systems, Minneapolis, MN, USA) according to manufacturer's instructions. Briefly, human cytokine array panel A membranes (R&D Systems) were incubated with cell culture supernatants. After washing, the membranes were further incubated with a detection Ab cocktail for 1 h. Spot detection was performed with streptavidin–horseradish peroxidase. The intensity of the spot was measured with ImageJ software (U.S. National Institutes of Health; http://rsb.info.nih.gov/ij/).
ELISA for neutrophil granules and cytokine
The concentrations of MPO, MMPs and the cytokines IL-1β, IL-8, and tumor necrosis factor (TNF)-α in cell culture supernatants were measured using the DuoSet enzyme-linked immunosorbent assay (ELISA) development system (R&D Systems) and ELISA MAX Standard Set Human TNF-α (Cat# 430201; BioLegend, San Diego, CA, USA) according to manufacturer' instructions. The absorbance at 450 nm was measured on a microplate reader (2030 ARVO X4; PerkinElmer Japan; Tokyo).
Western blotting
Samples were resolved by 14% SDS-PAGE and transferred to PVDF membranes. To evaluate the citrullination of histone H3 by mycobacteria, neutrophils were preincubated in the presence or absence of GSK484/109, then stimulated with mycobacteria at MOI of 1 for 4 h at 37 °C. After stimulation, the cells were lysed by Dounce homogenization in lysis buffer; and the proteins were resolved by SDS-PAGE; and subjected to Western blotting. The blots were incubated with primary antibody to citrullinated histone H3, followed by incubation with horseradish peroxidase–conjugated secondary antibodies. Membranes were stripped of antibodies by incubation with stripping buffer [62.5 mM Tris–HCl (pH 6.8), 100 mM β-mercaptoethanol, 2% SDS] for 30 min at 55 °C, followed by incubation with antibodies against total histone H3 and β-actin. Bands detected with SuperSignal reagent (Pierce Chemical) were scanned, and chemiluminescence signal intensities were quantified with ImageJ software (U.S. National Institutes of Health; http://rsb.info.nih.gov/ij/). The extent of histone H3 citrullination was calculated as the ratio of the band intensity of citrullinated histone H3 to that of β-actin.
Killing assay
Neutrophils (2 × 104 cells in 100 μl DMEM/F12) were pre-incubated with indicated inhibitors for 30 min, followed by treatment with mycobacteria at an MOI of 1 in 96-well flat-bottom tissue culture plates (Asahi Techno Glass, Tokyo, Japan) for 24 h at 37 °C. The culture plates were treated with 0.1% Triton X-100-containing PBS for 5 min at RT. Neutrophil lysates were serially diluted tenfold in 0.05% Tween 80-containing PBS and plated on sterile 7H11 Middlebrook agar plates (Difco, BD). Colony forming units (cfu) were counted after 2 weeks of culture at 37 °C. Killing was calculated as the number of these mycobacterial colonies divided by the number after incubation in medium alone.
Statistical analysis
Data were expressed as mean ± SD and analyzed by one-way analysis of variance (ANOVA), followed by Bonferroni’s multiple comparison test with GraphPad Prism software program (San Diego, CA). Differences between means were considered statistically significant at p < 0.05.
Results
MAC induce NET formation
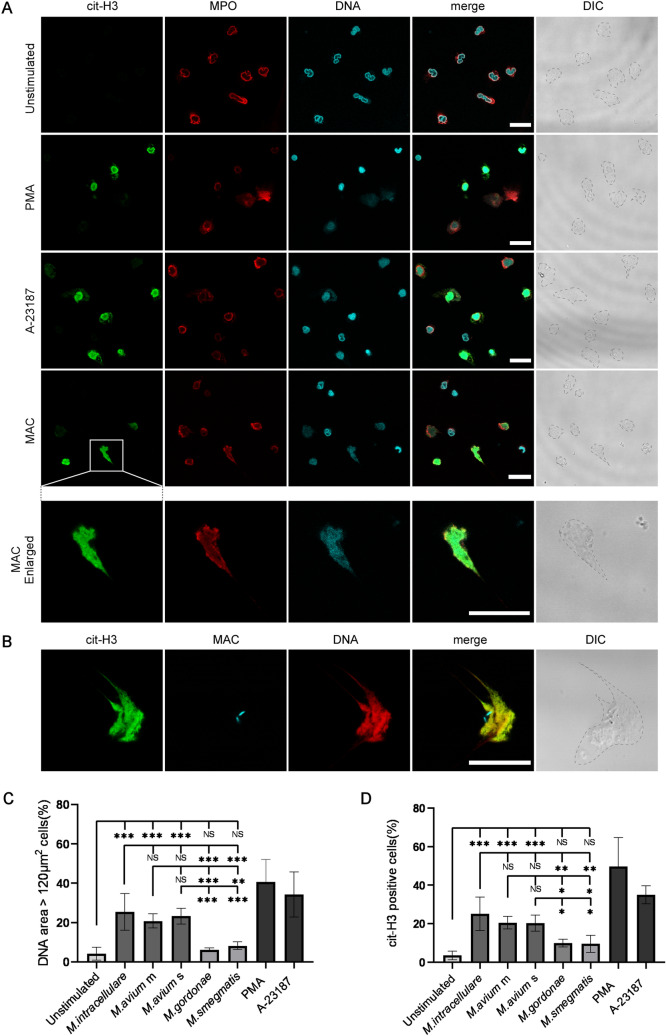
Along with NET formation, histone citrullination has been found to promote chromatin decondensation, resulting in nuclear expansion16. To determine whether mycobacteria induce NET formation in human neutrophils, cells were stained with MPO, which adheres to NETs fibers17, to show the area occupied by NETs. The nuclei in unstimulated neutrophils had a typical lobulated shape, with few cit-H3-positive cells detected (Fig. 1A, Supplementary Fig. 2, 3). In contrast, the reagents PMA (a potent activator of PKC) and A-23187(a potent activator of PKC-ζ), which induce NET formation33,34, markedly reduced nuclear lobular structure, yielding many cit-H3 positive cells. Incubation with MAC induced cit-H3 positive neutrophils, with the cit-H3 positive area also stained with anti-MPO antibody (Fig. 1B, Supplementary Fig. 2, 3). These results suggest that MAC induce NET formation in human neutrophils.
Figure 1.
MAC induce NET formation. (A) Localization of NET-related molecules. Neutrophils were stained to identify nuclei (blue) and areas positive for cit-H3 (green) and MPO (red) to determine the structures of NETs. Neutrophils were treated with PMA and A-23187 as a positive control for NET formation. (B) Localization of M. intracellulare (MAC) and NET-related molecules. Neutrophils were treated with Alexa Fluor 633-conjugated antibody to M. intracellulare (blue) and stained to identify nuclei (red) and areas positive for cit-H3 (green). (A,B) Gray dotted lines in DIC images indicating the outline of neutrophils. Confocal images were acquired on a Leica TCS-SP5 confocal microscope equipped with a 1.4 numerical aperture (NA) × 63 objective lens. DIC; differential interference contrast. white scale bar; 25 μm. (C,D) Assessment of NET formation by DNA area expansion based on the percentage of cells with a nuclear area > 120 µm2 (C) and the percentage of cit-H3 positive neutrophils (D). M. avium m and s were the mycobacteria isolated from patients with mild lung tissue destruction and a favorable clinical course, and from patients with severe lung tissue destruction and a critical clinical course, respectively. We examined the experiments using M. avium m and s isolated from three different patients with triplicate, respectively. Data shown are the means ± standard deviations (SD) of three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001 by one-way analysis of variance (ANOVA) with Bonferroni's multiple comparison test.
Evaluation of pathogenic mycobacteria showed that M. intracellulare, M. avium m and M. avium s induced mean ± SD TO-PRO3 positive areas of 25.4 ± 9.4%, 20.8 ± 3.6% and 23.3 ± 4.0%, representing the percentages of neutrophils producing NETs (Fig. 1C, Supplementary Fig. 4). M. gordonae and M. smegmatis, which are generally considered nonpathogenic although they may cause disease in rare cases35,36, slightly but not significantly induced mean ± SD TO-PRO3 positive areas of 6.2 ± 1.0% and 8.3 ± 1.9%, respectively (Fig. 1C). Evaluation of citrullination, representing mycobacteria-induced NET formation by neutrophils (Fig. 1D, Supplementary Fig. 4), showed that M. intracellulare, M. avium m and M. avium s induced NET formation by 25.2 ± 8.8%, 20.5 ± 3.2% and 20.3 ± 4.2%, respectively, of neutrophils, whereas M. gordonae and M. smegmatis induced NET formation by 10.0 ± 2.1% and 9.6 ± 4.4%, respectively, of neutrophils. Both of these quantification methods revealed that, compared with non-pathogenic mycobacteria, MAC significantly promoted NET formation by neutrophils.
PAD4 mediates MAC-induced NET formation
Peptidylarginine deiminase 4 (PAD4), which converts arginine to citrulline in histones, is highly expressed in human neutrophils. Citrullination by PAD4 reduces the strong positive charge of histones, thereby weakening histone-DNA binding. This weakened interaction subsequently leads to chromatin decondensation and NET formation37. The PAD4 inhibitor GSK484 suppressed induction by MAC of cit-H3 positive neutrophils (Fig. 2A) but had little effect on the percentage of cit-H3 positive neutrophils induced by non-pathogenic mycobacteria. The effect of the PAD4 inhibitor was further evaluated by assessing intracellular cit-H3 protein levels by Western blotting. PAD4 inhibitor significantly decreased intracellular cit-H3 protein band intensity of MAC-treated neutrophils (Fig. 2B,C), whereas GSK106, a negative control of PAD4 inhibitor, had no effect on cit-H3 protein band intensity of neutrophils (Supplementary Fig. 5). These results suggest that MAC-induced NET formation is regulated by PAD4. Evaluation of non-pathogenic mycobacteria showed that neutrophils produced slight amounts of GSK484-inhibitable cit-H3, although these cells rarely formed NETs.
Figure 2.
PAD4 mediates MAC-induced NET formation. (A) The effect of PAD4 inhibitor on the number of cit-H3 positive neutrophils. Data are means ± SD from five independent experiments. (B) The effect of PAD4 inhibitor on the expression levels of cit-H3. Representative protein band are shown. Molecular weights (kDa) are indicated on the left. H3 and β-actin were used as an internal control. Blots were cropped from different parts of the same gel. Original blots are presented in Supplementary Fig. 6. (C) Relative band intensities were calculated on the basis of densitometric analysis using ImageJ processing software. Protein levels of cit-H3 were calculated as the ratio of A-23187 pre-incubated with DMSO. Data are means ± SD from three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001 by one-way analysis of variance (ANOVA) with Bonferroni's multiple comparison test.
NET formation is not involved in neutrophil killing of mycobacteria
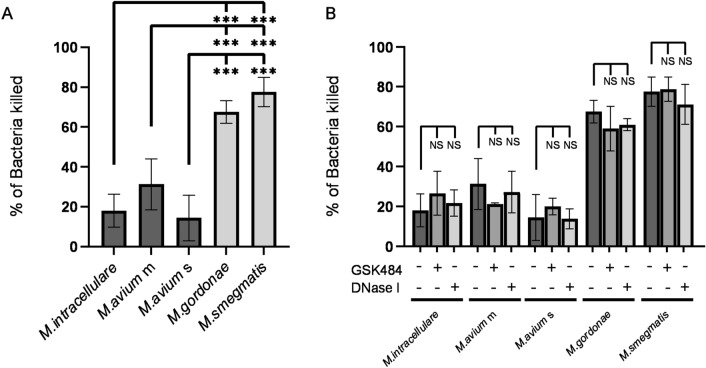
Because NET formation is thought to be involved in neutrophil killing of bacteria17, the present study assessed the involvement of NET formation in neutrophil killing of mycobacteria. In accordance with our previous experiments38, Neutrophils killed 67.5 ± 5.6% of M. gordonae and 77.5 ± 7.3% of M. smegmatis, but killed only 18.0 ± 8.2%, 31.3 ± 12.8% and 14.4 ± 11.5% of M. intracellulare, M. avium m and M. avium s, respectively (Fig. 3A), indicating that all MAC strains escaped from neutrophil killing. PAD4 inhibitor did not affect neutrophil killing of not only non-pathogenic mycobacteria but MAC (Fig. 3B). Although DNase I has been shown to inhibit NETs-dependent S.aureus killing by degrading NETs-associated DNA39, DNase I had little effect on neutrophil killing of any kind of mycobacteria. These results indicate that NET formation is not involved in neutrophil killing of mycobacteria.
Figure 3.
NET formation is not involved in neutrophil killing of mycobacteria. (A) Anti-mycobacterial activity of neutrophils. (B) Effect of inhibitors of NET formation on the anti-mycobacterial activity of neutrophils. Killing rate was calculated as the percentage of control values (bacteria incubated alone in the absence of neutrophils). Data are means ± SD of three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001 by one-way analysis of variance (ANOVA) with Bonferroni's multiple comparison test.
MAC-induced NET formation enhanced neutrophil degranulation
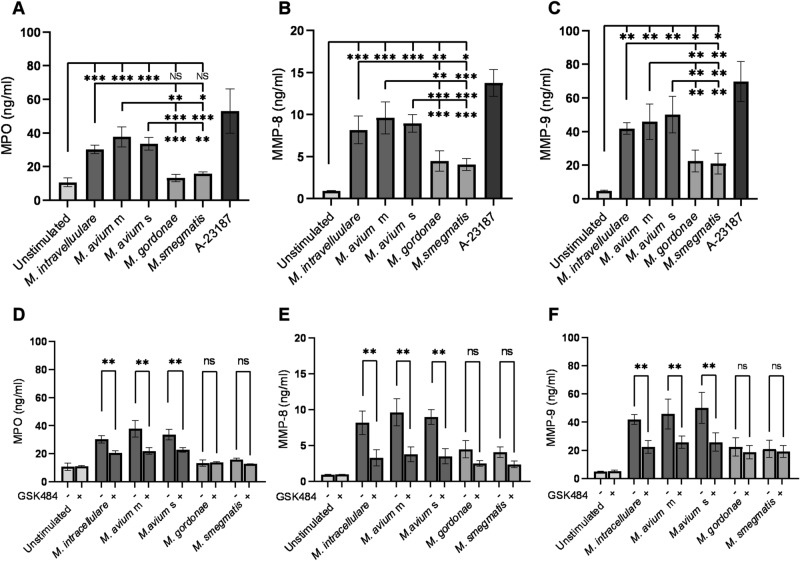
NETs contain proteins from neutrophil granules and cytoplasm17,40, with their components being dependent on the stimulus16. Neutrophil granular enzymes, including MMP-8 and MMP-9, degrade extracellular matrix proteins and lead to lung tissue destruction24. The present study therefore investigated whether MAC induce the release of neutrophil granular enzymes during NET formation. MPO was detected in the culture supernatants of neutrophils treated with MAC but not non-pathogenic mycobacteria (Fig. 4A). MMP-8 and MMP-9 were released into the culture supernatants of all types of mycobacteria-treated neutrophils (Fig. 4B,C), with MAC inducing significantly greater MMP-8 and MMP-9 release than non-pathogenic mycobacteria. PAD4 inhibitor significantly reduced the release of MPO, MMP-8, and MMP-9 from MAC-treated neutrophils (Fig. 4D–F), but did not affect their release from neutrophils treated with non-pathogenic mycobacteria.
Figure 4.
MAC-induced NET formation enhanced neutrophil degranulation. (A–C) Release of MPO (A), MMP-8 (B) and MMP-9 (C) from MAC-treated neutrophils. (D–F) Effect of PAD4 inhibitor on the release of MPO (D), MMP-8 (E) and MMP-9 (F) from MAC-treated neutrophils. MPO, MMP-8 and MMP-9 concentrations in cell culture supernatants were measured by ELISA. Data are means ± SD of three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001 by one-way analysis of variance (ANOVA) with Bonferroni's multiple comparison test.
MAC-induced IL-8 production by neutrophils
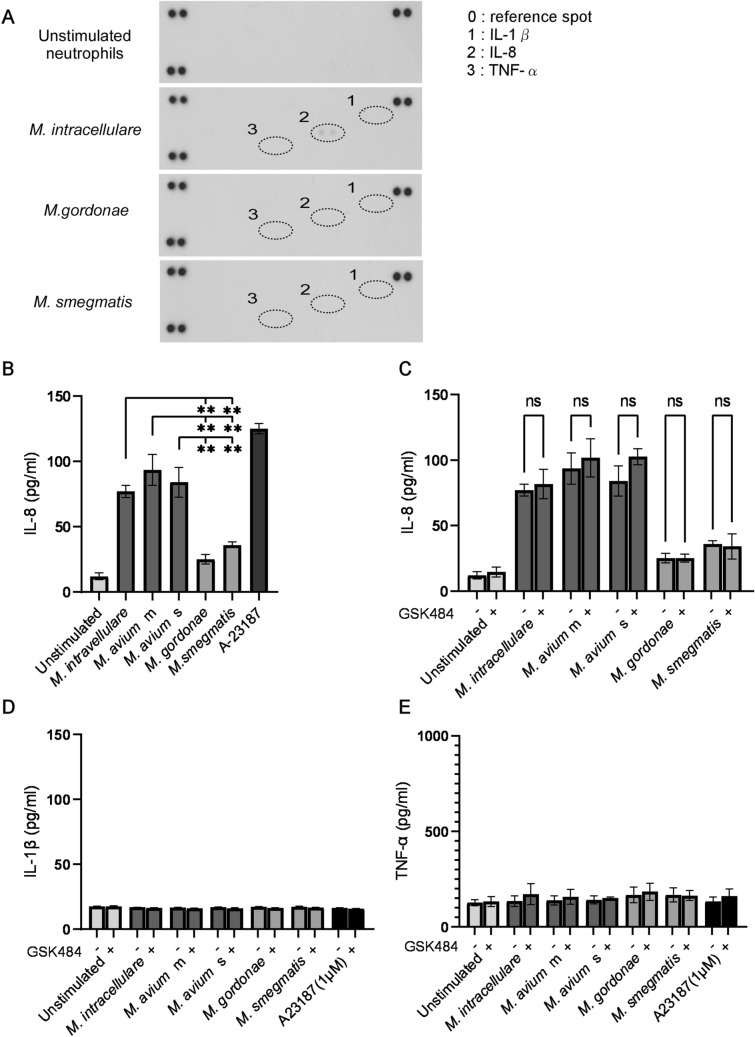
Human neutrophils secrete cytokines and chemokines in response to various stimuli, with these molecules regulating inflammatory and immune responses41. Assessment of cytokines and chemokines secreted by neutrophils treated with M. intracellulare, M. gordonae and M. smegmatis showed that, of the 36 cytokines and chemokines included in array analysis, only IL-8 secretion was observed (Fig. 5A). Because IL-1β and TNF-α are proinflammatory cytokines that promote neutrophilic inflammation42, the secretion of IL-8, IL-1β and TNF-α from mycobacteria-treated neutrophils was analyzed by ELISA. IL-8 was released from mycobacteria-treated neutrophils (Fig. 5B), with the amount secreted by MAC-treated neutrophils significantly higher than secreted by neutrophils treated with non-pathogenic mycobacteria. GSK484 did not affect the mycobacteria-induced IL-8 secretion by neutrophils (Fig. 5C). In agreement with the results of cytokine/chemokine array analysis, mycobacteria did not induce IL-1β or TNF-α release from neutrophils (Fig. 5D,E). These results indicate that the MAC-induced IL-8 production by neutrophils is not associated with NET formation.
Figure 5.
MAC induces IL-8 production by neutrophil. (A) Proteome profile of chemokines and cytokines in neutrophils treated with mycobacteria. Representative results are shown. (B) IL-8 production by neutrophils treated with mycobacteria. (C–E) Effects of PAD4 inhibitor on cytokine production by neutrophils treated with mycobacteria. The production of IL-8 (B,C), IL-1β (D) and TNF-α (E) by neutrophils was measured by ELISA. Data are means ± SD of three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001 by one-way analysis of variance (ANOVA) with Bonferroni’s multiple comparison test.
Discussion
NETs have been identified as the neutrophil preventing tool for microbial dissemination by trapping and killing bacteria17. However, the present study demonstrated that MAC, but not non-pathogenic mycobacteria, induced NET formation (Fig. 1C,D), but that MAC-induced NET formation was not involved in neutrophil killing of MAC (Fig. 3). M. tuberculosis was also demonstrated to escape from neutrophil killing regardless of NET formation19. Thus, NET formation is thought to be not involved in killings of pathogenic mycobacteria. In addition, Streptococcus pneumoniae and some strains of Haemophilus influenzae, which cause pneumonia, were found to escape from the bactericidal effects of NET formation43,44. Some pathogenic microorganisms that infect the lungs are likely to have avidity to avoid disinfection by NETs formation.
MAC and M. tuberculosis cause pathological changes in the lungs, such as bronchiectasis, granulation, and cavitation45,46. Bronchoalveolar lavage fluid (BALF) and plasma from patients with pulmonary tuberculosis contain increased levels of MMP-8 and MMP-947,48. Besides, MMP-8 and MMP-9 were detected in lung tissue samples from patients with pulmonary tuberculosis22, and found to be associated with indications of lung pathogenesis, such as cavitation, granulation and bronchiectasis28,49. In this study, MAC induced MMP-8 and MMP-9 release from neutrophils, and these MMPs release were promoted via NET formation (Fig. 4). MMP-8 is capable of degrading collagen types I to III, which are constituents of the bronchi, blood vessels, pulmonary interstitium and cartilage, whereas MMP-9 mainly degrades collagen types IV and V, which are constituents of basement membrane23. Therefore, it seems that NET formation-promoted MMPs released from neutrophils cause pathological changes in the lungs of MAC infection.
MMP-8 and MMP-9 are released extracellularly as inactive forms and are subsequently activated at sites of inflammation50. The enzymatic activities of MMPs are suppressed by endogenous inhibitors, a class of molecules called tissue inhibitors of metalloproteinases (TIMPs), which bind active and inactive forms of MMPs51. MMP/TIMP imbalances have been associated with the progression of lung tissue destruction in tuberculosis52. The present study used MAC strains, including M. avium, isolated from patients with severe or mild lung tissue destruction. However, MAC strains from patients with severe and mild lung tissue destruction were similarly effective in inducing the release of MMPs (Fig. 4). Activation of MAC-induced MMPs may therefore be associated with the patient-dependent pathogenicity of MAC, although further studies are needed to understand the relationships between MAC pathogenicity and host susceptibility.
MAC patients with neutrophil predominance in BALF exhibit more severe CT imaging and greater progression than patients with lymphocyte predominance in BALF53. Although neutrophils produce a variety of cytokines and chemokines both in vivo and in vitro41,54, neutrophils produce fewer cytokines and chemokines per cell than monocytes/macrophages and lymphocytes41. Of the 36 cytokines assayed, only IL-8 was induced by MAC from neutrophils (Fig. 5A). IL-8, also known as CXCL8, is a CXC type proinflammatory chemokine that attracts and activates neutrophils55. Nuclear factor-kappa B (NF-κB) and c-Jun N-terminal kinase (JNK) have been identified as critical molecules for IL-8 transcription, translation, and expression56, but these cascades have not been associated with NET formation57. In consistent with those previous studies, PAD4 inhibitor had no effect on IL-8 production (Fig. 5C), although MAC-induced NET formation was mediated by PAD4 (Fig. 2). High concentrations of IL-8 (> 100 ng/mL) have been reported to induce NET formation by human neutrophils17,58. Moreover, isolated NETs can induce NET formation and IL-8 production by neutrophils59, suggesting MAC-induced NET formation and IL-8 production can coordinately induce additional neutrophil accumulation and activation at MAC-infected sites, exacerbating pathogenesis. Thus, neutrophils are likely to play detrimental roles in MAC infections via NET formation and IL-8 production.
Taken together, the present results clearly revealed that MAC not only escaped from killing by human neutrophils but also induced NET formation and the production of MMPs and IL-8. NET formation was not involved in MAC killing, suggesting that MAC strains utilize NET formation, release of MMPs and IL-8 production to promote the progression of lung infections. These three processes may therefore be therapeutic targets in patients with MAC infection. Further study of the molecular mechanisms of MAC induced neutrophil activation is needed to elucidate the pathogenesis of MAC infections.
Supplementary Information
Acknowledgements
This study was supported in part by Foundation of Strategic Research Projects in Private Universities (Grants-in-Aid S1311011); AMED (Grant Number JP 20ae0101068h0005 to K.I.); JSPS KAKENHI (Grant Numbers JP17K10031, JP21K06086 to H.N.).
Author contributions
K.N.: Conceptualization, methodology, investigation, formal analysis, writing-original draft, funding acquisition; H.N.: investigation, Conceptualization, writing; S.S.: review and editing; K.T.: review and editing; K.I.: Conceptualization, writing, review and editing, supervision, funding acquisition.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-022-09017-y.
References
- 1.Namkoong H, et al. Epidemiology of pulmonary nontuberculous mycobacterial disease, Japan. Emerg. Infect. Dis. 2016;22:1116–1117. doi: 10.3201/eid2206.151086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Adjemian J, Olivier KN, Seitz AE, Holland SM, Prevots DR. Prevalence of nontuberculous mycobacterial lung disease in US Medicare beneficiaries. Am. J. Respir. Crit. Care Med. 2012;185:881–886. doi: 10.1164/rccm.201111-2016OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Morimoto K, et al. A steady increase in nontuberculous mycobacteriosis mortality and estimated prevalence in Japan. Ann. Am. Thorac. Soc. 2014;11:1–8. doi: 10.1513/AnnalsATS.201303-067OC. [DOI] [PubMed] [Google Scholar]
- 4.Vinnard C, et al. Deaths related to nontuberculous mycobacterial infections in the United States, 1999–2014. Ann. Am. Thorac. Soc. 2016;13:1951–1955. doi: 10.1513/AnnalsATS.201606-474BC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chin KL, Sarmiento ME, Alvarez-Cabrera N, Norazmi MN, Acosta A. Pulmonary non-tuberculous mycobacterial infections: Current state and future management. Eur. J. Clin. Microbiol. Infect. Dis. 2020;39:799–826. doi: 10.1007/s10096-019-03771-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fujita J, et al. Pathological and radiological changes in resected lung specimens in Mycobacterium avium intracellulare complex disease. Eur. Respir. J. 1999;13:535–540. doi: 10.1183/09031936.99.13353599. [DOI] [PubMed] [Google Scholar]
- 7.Oshitani Y, et al. Characteristic chest CT findings for progressive cavities in Mycobacterium avium complex pulmonary disease: a retrospective cohort study. Respir. Res. 2020;21:10. doi: 10.1186/s12931-020-1273-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gochi M, et al. Retrospective study of the predictors of mortality and radiographic deterioration in 782 patients with nodular/bronchiectatic Mycobacterium avium complex lung disease. BMJ Open. 2015;5:e008058. doi: 10.1136/bmjopen-2015-008058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kadota T, et al. Analysis of drug treatment outcome in clarithromycin-resistant Mycobacterium avium complex lung disease. BMC Infect. Dis. 2016;16:31. doi: 10.1186/s12879-016-1384-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Griffith DE, et al. An official ATS/IDSA statement: Diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am. J. Respir. Crit. Care Med. 2007;175:367–416. doi: 10.1164/rccm.200604-571ST. [DOI] [PubMed] [Google Scholar]
- 11.Yamazaki Y, et al. Markers indicating deterioration of pulmonary Mycobacterium avium-intracellulare infection. Am. J. Respir. Crit. Care Med. 1999;160:1851–1855. doi: 10.1164/ajrccm.160.6.9902019. [DOI] [PubMed] [Google Scholar]
- 12.Hwang JA, Kim S, Jo KW, Shim TS. Natural history of Mycobacterium avium complex lung disease in untreated patients with stable course. Eur. Respir. J. 2017;49:2. doi: 10.1183/13993003.00537-2016. [DOI] [PubMed] [Google Scholar]
- 13.Appelberg R, Silva MT. T cell-dependent chronic neutrophilia during mycobacterial infections. Clin. Exp. Immunol. 1989;78:478–483. [PMC free article] [PubMed] [Google Scholar]
- 14.Ulrichs T, Kaufmann SH. New insights into the function of granulomas in human tuberculosis. J. Pathol. 2006;208:261–269. doi: 10.1002/path.1906. [DOI] [PubMed] [Google Scholar]
- 15.Pruchniak MP, Arazna M, Demkow U. Life of neutrophil: From stem cell to neutrophil extracellular trap. Respir. Physiol. Neurobiol. 2013;187:68–73. doi: 10.1016/j.resp.2013.02.023. [DOI] [PubMed] [Google Scholar]
- 16.Papayannopoulos V. Neutrophil extracellular traps in immunity and disease. Nat. Rev. Immunol. 2018;18:134–147. doi: 10.1038/nri.2017.105. [DOI] [PubMed] [Google Scholar]
- 17.Brinkmann V, et al. Neutrophil extracellular traps kill bacteria. Science. 2004;303:1532–1535. doi: 10.1126/science.1092385. [DOI] [PubMed] [Google Scholar]
- 18.von Köckritz-Blickwede M, Nizet V. Innate immunity turned inside-out: Antimicrobial defense by phagocyte extracellular traps. J. Mol. Med. (Berl.) 2009;87:775–783. doi: 10.1007/s00109-009-0481-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ramos-Kichik V, et al. Neutrophil extracellular traps are induced by Mycobacterium tuberculosis. Tuberculosis (Edinb) 2009;89:29–37. doi: 10.1016/j.tube.2008.09.009. [DOI] [PubMed] [Google Scholar]
- 20.Moreira-Teixeira L, et al. Type I IFN exacerbates disease in tuberculosis-susceptible mice by inducing neutrophil-mediated lung inflammation and NETosis. Nat. Commun. 2020;11:5566. doi: 10.1038/s41467-020-19412-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schechter MC, et al. Neutrophil extracellular trap (NET) levels in human plasma are associated with active TB. PLoS ONE. 2017;12:e0182587. doi: 10.1371/journal.pone.0182587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ong CW, et al. Neutrophil-derived MMP-8 drives AMPK-dependent matrix destruction in human pulmonary tuberculosis. PLoS Pathog. 2015;11:e1004917. doi: 10.1371/journal.ppat.1004917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Davidson JM. Biochemistry and turnover of lung interstitium. Eur. Respir. J. 1990;3:1048–1063. [PubMed] [Google Scholar]
- 24.Cui N, Hu M, Khalil RA. Biochemical and biological attributes of matrix metalloproteinases. Prog. Mol. Biol. Transl. Sci. 2017;147:1–73. doi: 10.1016/bs.pmbts.2017.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Murphy G, Reynolds JJ, Bretz U, Baggiolini M. Collagenase is a component of the specific granules of human neutrophil leucocytes. Biochem. J. 1977;162:195–197. doi: 10.1042/bj1620195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Craig VJ, Zhang L, Hagood JS, Owen CA. Matrix metalloproteinases as therapeutic targets for idiopathic pulmonary fibrosis. Am. J. Respir. Cell Mol. Biol. 2015;53:585–600. doi: 10.1165/rcmb.2015-0020TR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gharib SA, Manicone AM, Parks WC. Matrix metalloproteinases in emphysema. Matrix Biol. 2018;73:34–51. doi: 10.1016/j.matbio.2018.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sabir N, Hussain T, Mangi MH, Zhao D, Zhou X. Matrix metalloproteinases: Expression, regulation and role in the immunopathology of tuberculosis. Cell Prolif. 2019;52:e12649. doi: 10.1111/cpr.12649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fowler SJ, et al. Nontuberculous mycobacteria in bronchiectasis: Prevalence and patient characteristics. Eur. Respir. J. 2006;28:1204–1210. doi: 10.1183/09031936.06.00149805. [DOI] [PubMed] [Google Scholar]
- 30.Campos-Garcia L, et al. And non- J. Fungi (Basel) 2019;5:2. doi: 10.3390/jof5020028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Papayannopoulos V, Metzler KD, Hakkim A, Zychlinsky A. Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps. J. Cell Biol. 2010;191:677–691. doi: 10.1083/jcb.201006052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nakazawa D, et al. Enhanced formation and disordered regulation of NETs in myeloperoxidase-ANCA-associated microscopic polyangiitis. J. Am. Soc. Nephrol. 2014;25:990–997. doi: 10.1681/ASN.2013060606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Neeli I, Radic M. Opposition between PKC isoforms regulates histone deimination and neutrophil extracellular chromatin release. Front. Immunol. 2013;4:38. doi: 10.3389/fimmu.2013.00038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Brinkmann V, Goosmann C, Kühn LI, Zychlinsky A. Automatic quantification of in vitro NET formation. Front. Immunol. 2012;3:413. doi: 10.3389/fimmu.2012.00413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Weinberger M, Berg SL, Feuerstein IM, Pizzo PA, Witebsky FG. Disseminated infection with Mycobacterium gordonae: Report of a case and critical review of the literature. Clin. Infect. Dis. 1992;14:1229–1239. doi: 10.1093/clinids/14.6.1229. [DOI] [PubMed] [Google Scholar]
- 36.Best CA, Best TJ. Mycobacterium smegmatis infection of the hand. Hand (N Y) 2009;4:165–166. doi: 10.1007/s11552-008-9147-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Leshner M, et al. PAD4 mediated histone hypercitrullination induces heterochromatin decondensation and chromatin unfolding to form neutrophil extracellular trap-like structures. Front. Immunol. 2012;3:307. doi: 10.3389/fimmu.2012.00307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nakayama H, et al. Lipoarabinomannan binding to lactosylceramide in lipid rafts is essential for the phagocytosis of mycobacteria by human neutrophils. Sci. Signal. 2016;9:101. doi: 10.1126/scisignal.aaf1585. [DOI] [PubMed] [Google Scholar]
- 39.Fuchs TA, et al. Novel cell death program leads to neutrophil extracellular traps. J. Cell Biol. 2007;176:231–241. doi: 10.1083/jcb.200606027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Urban CF, et al. Neutrophil extracellular traps contain calprotectin, a cytosolic protein complex involved in host defense against Candida albicans. PLoS Pathog. 2009;5:e1000639. doi: 10.1371/journal.ppat.1000639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cassatella MA, Östberg NK, Tamassia N, Soehnlein O. Biological roles of neutrophil-derived granule proteins and cytokines. Trends Immunol. 2019;40:648–664. doi: 10.1016/j.it.2019.05.003. [DOI] [PubMed] [Google Scholar]
- 42.Kany S, Vollrath JT, Relja B. Cytokines in inflammatory disease. Int. J. Mol. Sci. 2019;20:2. doi: 10.3390/ijms20236008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hong W, Juneau RA, Pang B, Swords WE. Survival of bacterial biofilms within neutrophil extracellular traps promotes nontypeable Haemophilus influenzae persistence in the chinchilla model for otitis media. J. Innate Immun. 2009;1:215–224. doi: 10.1159/000205937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Beiter K, et al. An endonuclease allows Streptococcus pneumoniae to escape from neutrophil extracellular traps. Curr. Biol. 2006;16:401–407. doi: 10.1016/j.cub.2006.01.056. [DOI] [PubMed] [Google Scholar]
- 45.Miura K, et al. Comparison of the chest computed tomography findings between patients with pulmonary tuberculosis and those with Mycobacterium avium complex lung disease. Respir Investig. 2020;58:137–143. doi: 10.1016/j.resinv.2019.12.006. [DOI] [PubMed] [Google Scholar]
- 46.Jain D, Ghosh S, Teixeira L, Mukhopadhyay S. Pathology of pulmonary tuberculosis and non-tuberculous mycobacterial lung disease: Facts, misconceptions, and practical tips for pathologists. Semin. Diagn. Pathol. 2017;34:518–529. doi: 10.1053/j.semdp.2017.06.003. [DOI] [PubMed] [Google Scholar]
- 47.Chang JC, et al. Effect of Mycobacterium tuberculosis and its components on macrophages and the release of matrix metalloproteinases. Thorax. 1996;51:306–311. doi: 10.1136/thx.51.3.306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sathyamoorthy T, et al. Gender-dependent differences in plasma matrix metalloproteinase-8 elevated in pulmonary tuberculosis. PLoS ONE. 2015;10:e0117605. doi: 10.1371/journal.pone.0117605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Elkington PT, et al. Synergistic up-regulation of epithelial cell matrix metalloproteinase-9 secretion in tuberculosis. Am. J. Respir. Cell Mol. Biol. 2007;37:431–437. doi: 10.1165/rcmb.2007-0011OC. [DOI] [PubMed] [Google Scholar]
- 50.Löffek S, Schilling O, Franzke CW. Series, "matrix metalloproteinases in lung health and disease": Biological role of matrix metalloproteinases: a critical balance. Eur. Respir. J. 2011;38:191–208. doi: 10.1183/09031936.00146510. [DOI] [PubMed] [Google Scholar]
- 51.Bassiouni W, Ali MAM, Schulz R. Multifunctional intracellular matrix metalloproteinases: Implications in disease. FEBS J. 2021 doi: 10.1111/febs.15701. [DOI] [PubMed] [Google Scholar]
- 52.Kübler A, et al. Mycobacterium tuberculosis dysregulates MMP/TIMP balance to drive rapid cavitation and unrestrained bacterial proliferation. J. Pathol. 2015;235:431–444. doi: 10.1002/path.4432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Inomata T, Konno S, Nagai K, Suzuki M, Nishimura M. Neutrophil predominance in bronchoalveolar lavage fluid is associated with disease severity and progression of HRCT findings in pulmonary Mycobacterium avium infection. PLoS ONE. 2018;13:e0190189. doi: 10.1371/journal.pone.0190189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ley K, et al. Neutrophils: New insights and open questions. Sci. Immunol. 2018;3:2. doi: 10.1126/sciimmunol.aat4579. [DOI] [PubMed] [Google Scholar]
- 55.Stillie R, Farooq SM, Gordon JR, Stadnyk AW. The functional significance behind expressing two IL-8 receptor types on PMN. J. Leukoc. Biol. 2009;86:529–543. doi: 10.1189/jlb.0208125. [DOI] [PubMed] [Google Scholar]
- 56.Hoffmann E, Dittrich-Breiholz O, Holtmann H, Kracht M. Multiple control of interleukin-8 gene expression. J. Leukoc. Biol. 2002;72:847–855. [PubMed] [Google Scholar]
- 57.Sollberger G, Amulic B, Zychlinsky A. Neutrophil extracellular trap formation is independent of de novo gene expression. PLoS ONE. 2016;11:e0157454. doi: 10.1371/journal.pone.0157454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.An Z, et al. Neutrophil extracellular traps induced by IL-8 aggravate atherosclerosis via activation NF-κB signaling in macrophages. Cell Cycle. 2019;18:2928–2938. doi: 10.1080/15384101.2019.1662678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Dömer D, Walther T, Möller S, Behnen M, Laskay T. Neutrophil extracellular traps activate proinflammatory functions of human neutrophils. Front. Immunol. 2021;12:636954. doi: 10.3389/fimmu.2021.636954. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.