Abstract
In 2012, national recommendations for cervical cancer screening of women aged 30-64 years were quinquennial human papillomavirus and cytology cotesting or triennial cytology. Data from a statewide surveillance program in New Mexico demonstrated 65.2% (95% confidence interval [95% CI] = 64.6% to 65.7%) of women screened in 2019 had a negative cotest within the last 3 years. Percentages of women screened in 2013, 2016, and 2019 with a prior negative cotest more than 5 years and up to 7 years ago were 2.6% (95% CI = 2.2% to 2.9%), 2.1% (95% CI = 1.9% to 2.2%), and 6.5% (95% CI = 6.2% to 6.8%), respectively (2-sided Ptrend < .001). Percentages of women screened in 2013, 2016, and 2019 with a prior negative cytology more than 5 years and up to 7 years ago were 3.8% (95% CI = 3.7% to 3.9%), 9.0% (95% CI = 8.7% to 9.3%), and 14.9% (95% CI = 14.4% to 15.4%), respectively (2-sided Ptrend < .001). Thus, in 2019, only 12.7% (95% CI = 12.4% to 13.1%) of the 30 215 women aged 30-64 years underwent cotesting and 27.7% (95% CI = 27.1% to 28.3%) of the 18 733 underwent cytology at the recommended interval. The observed under- and overscreening could result in increases in cervical cancer incidence and harms and costs, respectively.
Human papillomavirus (HPV) testing for cervical screening has been introduced stepwise into routine practice in the United States in women aged 30-64 years (1-3). Triennial concurrent HPV testing and cytology (“cotesting”) for women was first recommended in 2004 (1). In 2012, cotesting every 5 years was recommended (3,4), and more recently, HPV testing alone (“primary HPV testing”) was recommended every 5 years (2). Triennial cytology-only screening has remained an option because HPV testing has not been available or reimbursed in some clinics.
There is documentation of an increase in and determinants of cotesting uptake nationally (5,6). However, there are no data documenting population-based adherence to 5-year intervals for cotesting or 3-year intervals for cytology-only cervical screening following those 2012 recommendations.
We examined time trends for cervical screening across the state of New Mexico using data from the New Mexico HPV Pap Registry (NMHPVPR), which was established in 2006 to evaluate cervical cancer screening delivery across the continuum of care for New Mexico residents (http://164.64.110.134/parts/title07/07.004.0003.html; https://hpvprevention.unm.edu/nmhpvpr/) (7). The University of New Mexico Human Research Review Committee determined that public health surveillance activities of NMHPVPR were exempt.
We conducted a retrospective examination of cervical screening usage through 2019 for women aged 25-64 years. Percent screened within a given interval and the binomial 95% confidence intervals (CIs) were calculated by age group and screening year. Percent screened within a given interval were compared across screening years using Cochran-Armitage Trend tests. Linear regression with 1-sample t tests were used to test if there was a trend in percent screened as interval lengthened. All statistical tests were 2-sided, and a P value of less than .05 was considered statistically significant. Percentages of underscreened and overscreened by age group and screening year (cotest or cytology) shown in Table 1 were based on guideline recommendations because the screening interval is determined by the antecedent test result (ie, negative cotest or cytology). Exclusions and inclusions and additional details are found in the Supplementary Methods (available online).
Table 1.
Screening intervals among women aged 25-64 years with prior (antecedent) negative cotest (T−1); cytology and HPV negative who underwent a second (index) screening (HPV and cytology cotesting or cytology-alone) (T0) in 2013, 2016, or 2019
| Age group, y | Screening interval,a y | T0 |
P trend b | |||||
|---|---|---|---|---|---|---|---|---|
| 2013 (ntotal = 8537) |
2016 (ntotal = 23 053) |
2019 (ntotal = 31 178) |
||||||
| No. | % (95% CI) | No. | % (95% CI) | No. | % (95% CI) | |||
| 25-29 | 1 | 184 | 44.4 (39.7 to 49.2) | 320 | 36.5 (33.3 to 39.7) | 222 | 23.1 (20.4 to 25.7) | <.001 |
| 2 | 114 | 27.5 (23.2 to 31.8) | 279 | 31.8 (28.8 to 34.9) | 222 | 23.1 (20.4 to 25.7) | .008 | |
| 3 | 64 | 15.5 (12.0 to 18.9) | 180 | 20.5 (17.9 to 23.2) | 268 | 27.8 (25.0 to 30.7) | <.001 | |
| 4 | 26 | 6.3 (3.9 to 8.6) | 67 | 7.6 (5.9 to 9.4) | 146 | 15.2 (12.9 to 17.4) | <.001 | |
| 5 | 16 | 3.9 (2.0 to 5.7) | 16 | 1.8 (0.9 to 2.7) | 75 | 7.8 (6.1 to 9.5) | <.001 | |
| >5-7 | 10 | 2.4 (0.9 to 3.9) | 14 | 1.6 (0.8 to 2.4) | 30 | 3.1 (2.0 to 4.2) | .21 | |
| All | 414 | — | 876 | — | 963 | — | ||
| P c | .004 | <.001 | .03 | |||||
| 30-39 | 1 | 1351 | 49.4 (47.5 to 51.3) | 2221 | 32.8 (31.7 to 33.9) | 1659 | 19.3 (18.5 to 20.1) | <.001 |
| 2 | 809 | 29.6 (27.9 to 31.3) | 2071 | 30.6 (29.5 to 31.7) | 1945 | 22.6 (21.8 to 23.5) | <.001 | |
| 3 | 365 | 13.4 (12.1 to 14.6) | 1726 | 25.5 (24.4 to 26.5) | 2398 | 27.9 (27.0 to 28.9) | <.001 | |
| 4 | 117 | 4.3 (3.5 to 5.0) | 492 | 7.3 (6.6 to 7.9) | 1419 | 16.5 (15.7 to 17.3) | <.001 | |
| 5 (recommended)d | 60 | 2.2 (1.6 to 2.7) | 177 | 2.6 (2.2 to 3.0) | 796 | 9.3 (8.7 to 9.9) | <.001 | |
| >5-7 | 32 | 1.2 (0.8 to 1.6) | 91 | 1.3 (1.1 to 1.6) | 375 | 4.4 (3.9 to 4.8) | <.001 | |
| All | 2734 | — | 6778 | — | 8592 | — | ||
| P c | .006 | .003 | .08 | |||||
| 40-49 | 1 | 1128 | 45.7 (43.7 to 47.7) | 1920 | 28.2 (27.1 to 29.2) | 1618 | 17.1 (16.3 to 17.9) | <.001 |
| 2 | 636 | 25.8 (24.0 to 27.5) | 1927 | 28.3 (27.2 to 29.3) | 1891 | 20.0 (19.2 to 20.8) | <.001 | |
| 3 | 395 | 16.0 (14.6 to 17.5) | 1942 | 28.5 (27.4 to 29.6) | 2541 | 26.8 (26.0 to 27.7) | <.001 | |
| 4 | 155 | 6.3 (5.3 to 7.2) | 621 | 9.1 (8.4 to 9.8) | 1464 | 15.5 (14.7 to 16.2) | <.001 | |
| 5 (recommended)d | 75 | 3.0 (2.4 to 3.7) | 263 | 3.9 (3.4 to 4.3) | 1285 | 13.6 (12.9 to 14.3) | <.001 | |
| >5-7 | 79 | 3.2 (2.5 to 3.9) | 142 | 2.1 (1.7 to 2.4) | 666 | 7.0 (6.5 to 7.6) | <.001 | |
| All | 2468 | — | 6815 | — | 9465 | — | ||
| P c | .006 | .01 | .18 | |||||
| 50-64 | 1 | 1184 | 40.5 (38.8 to 42.3) | 2129 | 24.8 (23.9 to 25.7) | 2130 | 17.5 (16.8 to 18.2) | <.001 |
| 2 | 783 | 26.8 (25.2 to 28.4) | 2489 | 29.0 (28.0 to 30.0) | 2306 | 19.0 (18.3 to 19.7) | <.001 | |
| 3 | 542 | 18.6 (17.1 to 20.0) | 2568 | 29.9 (28.9 to 30.9) | 3206 | 26.4 (25.6 to 27.2) | <.001 | |
| 4 | 206 | 7.1 (6.1 to 8.0) | 767 | 8.9 (8.3 to 9.5) | 1826 | 15.0 (14.4 to 15.7) | <.001 | |
| 5 (recommended)d | 108 | 3.7 (3.0 to 4.4) | 409 | 4.8 (4.3 to 5.2) | 1768 | 14.5 (13.9 to 15.2) | <.001 | |
| >5-7 | 98 | 3.4 (2.7 to 4.0) | 222 | 2.6 (2.3 to 2.9) | 922 | 7.6 (7.1 to 8.1) | <.001 | |
| All | 2921 | — | 8584 | — | 12 158 | — | ||
| P c | .002 | .03 | .19 | |||||
| 30-64 | 1 | 3663 | 45.1 (44.0 to 46.2) | 6270 | 28.3 (27.7 to 28.9) | 5407 | 17.9 (17.5 to 18.3) | <.001 |
| 2 | 2228 | 27.4 (26.5 to 28.4) | 6487 | 29.3 (28.7 to 29.8) | 6142 | 20.3 (19.9 to 20.8) | <.001 | |
| 3 | 1302 | 16.0 (15.2 to 16.8) | 6236 | 28.1 (27.5 to 28.7) | 8145 | 27.0 (26.5 to 27.5) | <.001 | |
| 4 | 478 | 5.9 (5.4 to 6.4) | 1880 | 8.5 (8.1 to 8.8) | 4709 | 15.6 (15.2 to 16.0) | <.001 | |
| 5 (recommended)d | 243 | 3.0 (2.6 to 3.4) | 849 | 3.8 (3.6 to 4.1) | 3849 | 12.7 (12.4 to 13.1) | <.001 | |
| >5-7 | 209 | 2.6 (2.2 to 2.9) | 455 | 2.1 (1.9 to 2.2) | 1963 | 6.5 (6.2 to 6.8) | <.001 | |
| All | 8123 | — | 22 177 | — | 30 215 | — | ||
| P c | .004 | .01 | .14 | |||||
| Overscreened (1 y) | 3663 | 45.1 (44.0 to 46.2) | 6270 | 28.3 (27.7 to 28.9) | 5407 | 17.9 (17.5 to 18.3) | <.001 | |
| Overscreened (1 and 2 y combined) | 5891 | 72.5 (71.6 to 73.5) | 12 757 | 57.5 (56.9 to 58.2) | 11 549 | 38.2 (37.7 to 38.8) | <.001 | |
| Overscreened (1, 2, and 3 y combined) | 7193 | 88.6 (87.9 to 89.2) | 18 993 | 85.6 (85.2 to 86.1) | 19 694 | 65.2 (64.6 to 65.7) | <.001 | |
| Overscreened (1, 2, 3, and 4 y combined) | 7671 | 94.4 (93.9 to 94.9) | 20 873 | 94.1 (93.8 to 94.4) | 24 403 | 80.8 (80.3 to 81.2) | <.001 | |
Screening intervals were defined by T0 to T−1 and were categorized as 11 to less than 18 months (1 year) and in 12-month periods thereafter corresponding to the integer number of years in the interval ±6 months: 1 year = 11-18 months, 2 years = 19-30 months, 3 years = 31-42 months, etc. CI = confidence interval ; HPV = human papillomavirus.
Two-sided Cochran-Armitage Trend tests were used.
Linear regression with 2-sided 1-sample t tests were used.
A 5-year screening interval following a negative cotest was the national recommendation in 2013, 2016, and 2019 for women aged 30-64 years.
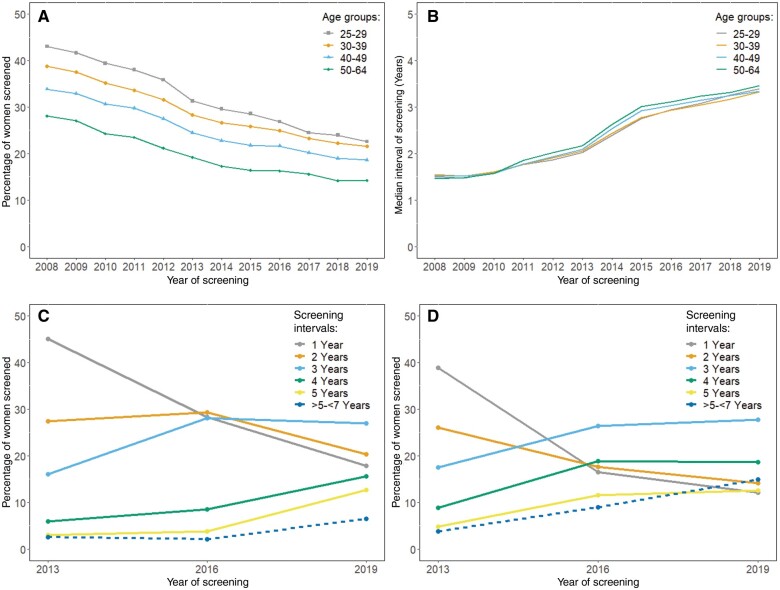
From 2008 to 2019, the percentage of women screened decreased twofold for all age groups, with younger women more likely to be screened than older women (Figure 1, A). There was a concomitant increase in the median screening interval from approximately 1.5 years in 2008 to approximately 3.4 years in 2019 (Figure 1, B).
Figure 1.
Cervical cancer screening use and median screening intervals for women living in New Mexico. Shown are the A) Percentage of women screened and B) Median screening intervals for women aged 25-64 years living in New Mexico undergoing cervical screening by age group and year (irrespective of screening modality). Panels A and B include 600 987 individual women with screening cytology across the period of 2008-2019. Percentages of women included in A use age-specific denominators from the US Census (https://www.census.gov/data/tables/time-series/demo/popest/2010s-state-detail.html). C) Percentage of women aged 30-64 years who had an index (T0) screen (irrespective of screening modality or result; cytology alone or cotesting) in 2013, 2016, or 2019 following an antecedent (T−1) negative cotest (negative HPV and negative cytology) 1, 2, 3, 4, and over than 5-7 years before the index screen. D) Percentage of women aged 30-64 years who had an index (T0) screen (irrespective of screening modality or result; cytology alone or cotesting) in 2013, 2016, or 2019 following an antecedent (T−1) negative cytology 1, 2, 3, 4, and over than 5-7 years before the index screen. The denominators for panels C (antecedent negative cotest) and D (antecedent negative cytology) are shown in Table 1 and Supplementary Table 1 (available online), respectively. Screening intervals are defined by the time between the index screen and the antecedent screen, that is, T0 to T−1. Exclusions defining screening tests are detailed in the Supplementary Methods (available online).
We identified 91 651, 71 300, and 57 532 index (T0) screens in 2013, 2016, and 2019, respectively (total represents Table 1 and Supplementary Table 1, available online). Screening intervals statistically significantly lengthened over time for women with an antecedent (T−1) cotest or a cytology—alone, overall, and in each age group (Ptrend < .01)—with exception only among women aged 25-29 years with intervals of 5-7 years. Notably, some women aged 25-29 years for whom only cytology screening is recommended (Table 1) were being screened by cotesting, with one-quarter of those women undergoing annual cotesting. In 2019, 65.2% (95% CI = 64.6% to 65.7%) of women screened in 2019 had negative cotests within the last 3 years, 17.9% (95% CI = 17.5% to 18.3%) within 1 year, 20.3% (95% CI = 19.9% to 20.8%) within 2 years, and 27.0% (95% CI = 26.5% to 27.5%) within 3 years.
There was an increasing trend across time for women to be screened at intervals longer than those recommended (Table 1). For women aged 30-64 years with an antecedent negative cotest (Figure 1, C; Table 1), the percentage screened at an interval of more than 5 years and up to 7 years (ie, 67-84 months) was 2.6% (95% CI = 2.2% to 2.9%), 2.1% (95% CI = 1.9% to 2.2%), and 6.5% (95% CI = 6.2% to 6.8%) for women with an index screen in 2013, 2016, and 2019, respectively (Ptrend < .001). Only 12.7% (95% CI = 12.4% to 13.1%) of women with an antecedent negative cotest received cervical screening at the recommended 5-year interval in any year.
Among women aged 30-64 years with an antecedent negative cytology test (Figure 1, D; Supplementary Table 1, available online) for whom screening in 3 years was recommended, the percentage screened at an interval of more than 5 years and up to 7 years was 3.8% (95% CI = 3.7% to 3.9%), 9.0% (95% CI = 8.7% to 9.3%), and 14.9% (95% CI = 14.4% to 15.4%) for women with an index screen in 2013, 2016, and 2019, respectively (Ptrend < .001). Only 27.7% (95% CI = 27.1% to 28.3%) of women with an antecedent negative cytology received cervical screening at the recommended 3-year interval in any year.
Of the screen-eligible population served across New Mexico by a diversity of clinical practices, insurers, and health service delivery settings, few women received cervical screening at the recommended intervals. Many were screened too frequently, especially by cotesting, although the percentage of overscreening did decrease for cotesting (94.4%, 95% CI = 93.9% to 94.9% in 2013 to 80.8%, 95% CI = 80.3% to 81.2% in 2019) (Table 1) and cytology (65.1%, 95% CI = 64.7% to 65.4% in 2013 to 26.2%, 95% CI = 25.5% to 26.8% in 2019) (Supplementary Table 1, available online).
A new and alarming observation was the increasing percentage of women being screened at too long of an interval. Most notable was the greater than fourfold increase in women being screened at an interval of more than 5 years and up to 7 years following a negative cytology alone, which does not provide the same reassurance against cancer as a negative HPV test or cotest (8,9). There was also an increasing trend in women being screened at an interval of more than 5 years and up to 7 years following a negative HPV cotest, which may increase the risk of cancer (10 ).
Thus, in 2019, only approximately 13% of women aged 30-64 years underwent cotesting at the recommended 5-year interval (Table 1). In addition, only approximately 28% of women aged 30-64 years underwent cytology screening at the recommended 3-year interval (Supplementary Table 1, available online).
Because of limitations of NMHPVPR data, we were not able to examine the determinants of adherence. One of the main factors related to poor acceptance of longer screening intervals may be a lack of patient and provider knowledge (6,11–14).
Of note, only women with 2 screens are included in Table 1, and we did not account for women with very long screening intervals, only 1 screen, or none at all. Thus, we have underestimated the proportion of women being underscreened. The NMHPVPR was established in 2006, allowing a maximum 7-year retrospective comparison of screening intervals for index screens in 2013, 2016, and 2019. Supplementary Tables 2 and 3 (available online) detail women with screening intervals of more than 5 years (>66 months) following a negative cotest and cytology-alone screen, respectively. Finally, our findings were limited to screening in New Mexico, which may not be generalizable to other settings. Strengths of this analysis include the use of electronic health records from a population-based, statewide registry.
In conclusion, although overscreening is declining, many women are still undergoing cervical cancer screening too frequently, especially by cotesting, thereby increasing its harms and costs. Meanwhile, an increasing number of women are undergoing cervical cancer screening too infrequently, which, as a consequence, may reduce its health benefits (3,10).
Funding
This work was supported by the National Institute of Allergy and Infectious Diseases (NIAID) of the U.S. National Institutes of Health (grant number U19AI113187) and by the National Cancer Institute (NCI) of the U.S. National Institutes of Health (*grant number NCI P30CA118100) through the Biostatistics Shared Resource of the University of New Mexico Comprehensive Cancer Center.
Notes
Role of the funder: The study funder had no role in the design of the study, the collection, analysis, and interpretation of the data and the decision to submit for publication.
Disclosures: PEC has received HPV tests and assays for research at a reduced or no cost from Roche, Cepheid, Becton Dickinson, and Arbor Vita Corporation. JC and CMW have received funds from grants, cooperative agreements or subcontracts related to cervical screening and triage through their respective institutions. JC reports grants and personal fees from Qiagen, grants from Hologic, grants and personal fees from Becton Dickinson (BD) and Genera Biosystems (GB), grants from Gene First, and grants from Trovagene, outside the submitted work. CMW reports receiving reagents and equipment from Roche Molecular Systems, Roche/Ventana Medical Systems, Hologic and GB through her institution and outside of the submitted work, personal fees from BD. All other authors report no potential conflicts of interest.
Author contributions: All authors take responsibility for the manuscript content, contributed to the concept or design, data acquisition, analysis or interpretation of the data, made critical revisions or contributed important intellectual content and took the decision to submit the final manuscript. The authors contributions are as follows: Philip E. Castle: Formal Analysis, Writing—Original Draft Preparation, and Writing—Review and Editing; Walter K. Kinney: Project Administration, Supervision and Writing—Review and Editing; Lu Chen: Formal Analysis and Writing—Review and Editing; Jane J. Kim: Project Administration and Writing—Review and Editing; Steven Jenison: Project Administration and Writing—Review and Editing; Giovanna Rossi: Project Administration and Writing—Review and Editing; Huining Kang: Formal Analysis and Writing—Review and Editing; Jack Cuzick: Investigation, Methodology; Formal Analysis, and Writing—Review and Editing; Cosette M. Wheeler: Funding Acquisition, Project Administration, Supervision, Formal Analysis, Writing—Original Draft Preparation, and Writing—Review and Editing.
Disclaimer: Opinions expressed by the authors are their own and this material should not be interpreted as representing the official viewpoint of the U.S. Department of Health and Human Services, the National Institutes of Health or the National Cancer Institute.
Acknowledgements: New Mexico HPV Pap Registry Steering Committee Collaborators: New Mexico HPV Pap Registry Steering Committee members listed contributed to the content, reviewed and gave input, approved the manuscript and participated in the final decision to submit to the journal. The New Mexico HPV Pap Registry Steering Committee members participating in this effort are as follows: Nancy E. Joste, MD, University of New Mexico Health Sciences Center and Tricore Reference Laboratories, Albuquerque, New Mexico; Walter Kinney, MD, Sacramento California; Cosette M. Wheeler, PhD, University of New Mexico Comprehensive Cancer Center; Charles L. Wiggins, PhD, MSPH, University of New Mexico Comprehensive Cancer Center; Michael Robertson, BS, University of New Mexico Comprehensive Cancer Center; Ruth M. McDonald, MS, University of New Mexico Comprehensive Cancer Center; Alan Waxman, MD, MPH, University of New Mexico Health Sciences Center; Steven Jenison, MD, University of New Mexico Health Sciences Center; Jean Howe, MD, MPH, US Indian Health Service; Debbie Saslow, PhD, American Cancer Society; Jane J. Kim, PhD, Harvard TH Chan School of Public Health; Mark H. Stoler, MD, University of Virginia; Jack Cuzick, PhD, Wolfson Institute of Preventive Medicine, Queen Mary University of London, London, England; Philip E. Castle, PhD, MPH, Albert Einstein College of Medicine (member through October 1, 2019); Rebecca B. Perkins, MD, MS, Boston University; Janice L. Gonzales, MD, MPH, FAAP, New Mexico Department of Health; Salina Torres, PhD, MPH, New Mexico Department of Health; Giovanna Rossi, MSc, Collective Action Strategies, Community Member; and Kevin English, DrPh, MPH, Albuquerque Area Southwest Tribal Epidemiology Center (AASTEC). No compensation was received for contributions to this manuscript by any named authors or by the New Mexico HPV Pap Registry Steering Committee members.
Finally, we thank critical New Mexico HPV Pap Registry team members including Michael Robertson and Ruth McDonald for the curation of all data reported to the state-wide public health surveillance program and Norah Torrez-Martinez and Ann Powell for their critical contributions to data management and validations.
Supplementary Material
Data Availability
Data supporting the investigation reported in this article can be made available in de-identified form subject to establishing a data use agreement with the University of New Mexico Health Sciences Center .
Contributor Information
Philip E Castle, PhD, MPH, Albert Einstein College of Medicine, Bronx, NY, USA; Division of Cancer Prevention and Division of Cancer Epidemiology and Genetics, National Institutes of Health, National Cancer Institute, Rockville, MD, USA; National Institutes of Health, National Cancer Institute, Division of Cancer Prevention and Division of Cancer Epidemiology and Genetics, Rockville, MD, USA.
Walter K Kinney, MD, Sacramento, CA, USA.
Lu Chen, MS, Comprehensive Cancer Center, Biostatistics Shared Resource, University of New Mexico, Albuquerque, NM, USA.
Jane J Kim, PhD, Harvard T.H. Chan, School of Public Health, Boston, MA, USA.
Steven Jenison, MD, School of Medicine, University of New Mexico, Albuquerque, NM, USA.
Giovanna Rossi, MSc, Collective Action Strategies, Albuquerque, NM, USA.
Huining Kang, PhD, Comprehensive Cancer Center, Biostatistics Shared Resource, University of New Mexico, Albuquerque, NM, USA.
Jack Cuzick, PhD, Wolfson Institute of Preventive Medicine, Queen Mary University of London, London, UK.
Cosette M Wheeler, PhD, Comprehensive Cancer Center, Center for HPV Prevention, University of New Mexico, Albuquerque, USA.
the New Mexico HPV Pap Registry Steering Committee, New Mexico HPV Pap Registry Steering Committee Member collaborators: see the Notes.
New Mexico HPV Pap Registry Steering Committee Collaborators:
Nancy E Walter Joste, Cosette M Kinney, Charles L Wheeler, Michael Wiggins, Ruth M Robertson, Alan McDonald, Steven Waxman, Jean Jenison, Debbie Howe, Jane J Saslow, Mark H Kim, Jack Stoler, Philip E Cuzick, Rebecca B Castle, Janice L Perkins, Salina Gonzales, Giovanna Torres, Kevin Rossi, and English
References
- 1. Wright TC Jr, Schiffman M, Solomon D, et al. Interim guidance for the use of human papillomavirus DNA testing as an adjunct to cervical cytology for screening. Obstet Gynecol. 2004;103(2):304–309. [DOI] [PubMed] [Google Scholar]
- 2. Curry SJ, Krist AH, Owens DK, et al. ; US Preventive Services Task Force. Screening for cervical cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;320(7):674–686. [DOI] [PubMed] [Google Scholar]
- 3. Saslow D, Solomon D, Lawson HW, et al. ; American Society for Clinical Pathology. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. Am J Clin Pathol. 2012;137(4):516–542. [DOI] [PubMed] [Google Scholar]
- 4. Moyer VA; U.S. Preventive Services Task Force. Screening for cervical cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2012;156(12):880–891, w312. [DOI] [PubMed] [Google Scholar]
- 5. Goding Sauer A, Bandi P, Saslow D, Islami F, Jemal A, Fedewa SA. Geographic and sociodemographic differences in cervical cancer screening modalities. Prev Med. 2020;133:106014. [DOI] [PubMed] [Google Scholar]
- 6. Watson M, Benard V, King J, Crawford A, Saraiya M. National assessment of HPV and Pap tests: changes in cervical cancer screening, National Health Interview Survey. Prev Med. 2017;100:243–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Cuzick J, Myers O, Hunt WC, et al. ; New Mexico HPV Pap Registry Steering Committee. A population-based evaluation of cervical screening in the United States: 2008-2011. Cancer Epidemiol Biomarkers Prev. 2014;23(5):765–773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Ronco G, Dillner J, Elfström KM, et al. International HPV screening working group. Efficacy of HPV-based screening for prevention of invasive cervical cancer: follow-up of four European randomised controlled trials. Lancet. 2014;383(9916):524–532. [DOI] [PubMed] [Google Scholar]
- 9. Sankaranarayanan R, Nene BM, Shastri SS, et al. HPV screening for cervical cancer in rural India. N Engl J Med. 2009;360(14):1385–1394. [DOI] [PubMed] [Google Scholar]
- 10. Gage JC, Schiffman M, Katki HA, et al. Reassurance against future risk of precancer and cancer conferred by a negative human papillomavirus test. J Natl Cancer Inst. 2014;106(8):dju153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Benard VB, Greek A, Roland KB, Hawkins NA, Lin L, Saraiya M. Change in provider beliefs regarding cervical cancer screening intervals after an educational intervention. J Womens Health (Larchmt). 2016;25(5):422–427. [DOI] [PubMed] [Google Scholar]
- 12. Berkowitz Z, Saraiya M, Sawaya GF. Cervical cancer screening intervals, 2006 to 2009: moving beyond annual testing. JAMA Intern Med. 2013;173(10):922–924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Cooper CP, Saraiya M, Sawaya GF. Acceptable and preferred cervical cancer screening intervals among U.S. women. Am J Prev Med. 2015;49(6):e99-107–e107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Hawkins NA, Benard VB, Greek A, Roland KB, Manninen D, Saraiya M. Patient knowledge and beliefs as barriers to extending cervical cancer screening intervals in Federally Qualified Health Centers. Prev Med. 2013;57(5):641–645. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data supporting the investigation reported in this article can be made available in de-identified form subject to establishing a data use agreement with the University of New Mexico Health Sciences Center .