Abstract
Foscarnet (trisodium phosphonoformate hexahydrate) is an antiviral agent used to treat cytomegalovirus disease in immunocompromised patients. One common side effect is acute ionized hypocalcemia and hypomagnesemia following intravenous administration. Foscarnet-induced ionized hypomagnesemia might contribute to ionized hypocalcemia by impairing excretion of preformed parathyroid hormone (PTH) or by producing target organ resistance. Prevention of ionized hypomagnesemia following foscarnet administration could blunt the development of ionized hypocalcemia. To determine whether intravenous magnesium ameliorates the decline in ionized calcium and/or magnesium following foscarnet infusions, MgSO4 at doses of 1, 2, and 3 g was administered in a double-blind, placebo-controlled, randomized, crossover trial to 12 patients with AIDS and cytomegalovirus disease. Overall, increasing doses of MgSO4 reduced or eliminated foscarnet-induced acute ionized hypomagnesemia. Supplementation, however, had no discernible effect on foscarnet-induced ionized hypocalcemia despite significant increases in serum PTH levels. No dose-related, clinically significant adverse events were found, suggesting that intravenous supplementation with up to 3 g of MgSO4 was safe in this chronically ill population. Since parenteral MgSO4 did not alter foscarnet-induced ionized hypocalcemia or symptoms associated with foscarnet, routine intravenous supplementation for patients with normal serum magnesium levels is not recommended during treatment with foscarnet.
Foscarnet (trisodium phosphonoformate hexahydrate, Foscavir) is a pyrophosphate analogue that inhibits many viral DNA polymerases (8). It is used to treat cytomegalovirus (CMV) and acyclovir-resistant mucocutaneous herpes simplex virus disease in immunocompromised patients, including those with AIDS (3, 10, 19, 27, 28, 32). Foscarnet is normally administered every 8 or 12 h as an intravenous infusion (21). Reversible nephrotoxicity is a well-recognized side effect and prehydration substantially reduces its incidence (6, 9, 17). Abnormalities of blood chemistries are also common and on occasion severe. These changes include magnesium depletion, acute ionized hypocalcemia and hypomagnesemia, hypokalemia, and hypo- or hyperphosphatemia (13, 15, 17–19, 23, 27, 31, 32; M. S. Youle, J. Clarbour, B. Gazzard, and A. Chanas, Letter, Lancet i:1455–1456, 1988). The mechanisms by which foscarnet induces these changes, especially in ionized cations, are not completely understood.
As a pyrophosphate analogue, foscarnet is a potent chelator of divalent cations. Complexes readily form with calcium and magnesium in solution (17, 18, 36), and a linear relationship exists between ionized hypocalcemia, ionized hypomagnesemia, and circulating plasma foscarnet concentrations (18, 26). Chelation of free calcium and magnesium acutely reduces ionized but not total concentrations of these cations in vitro and in vivo (5, 18; E. Dohin, C. Kindermans, J. C. Souberbielle, C. Sachs, and C. Katlama, presented at Int. Conf. AIDS, 1993). Clinical symptoms associated with foscarnet-induced ionized hypocalcemia include nausea, headache, paresthesias, seizures, generalized neuromuscular irritability, including tetany, and death (13, 17, 18, 24, 34; Youle et al., Letter).
Although calcium complexing with foscarnet may explain, in part, foscarnet-induced ionized hypocalcemia, other mechanisms have been proposed. Measurements of serum parathyroid hormone (PTH) following foscarnet infusions demonstrate inappropriately low levels of this hormone (5; Dohin et al., Int. Conf. AIDS, 1993). Inadequate PTH secretion in response to ionized hypocalcemia could result from concomitant foscarnet-induced ionized hypomagnesemia inhibiting the release of preformed PTH from the parathyroid gland (2, 12). Alternatively, acute ionized hypomagnesemia may contribute to ionized hypocalcemia by altering skeletal responsiveness to the calcium-mobilizing action of PTH (11, 22).
The degree to which foscarnet-induced ionized hypomagnesemia impairs excretion of preformed PTH or produces target organ resistance has not been established. If this mechanism is important, then the prevention of ionized hypomagnesemia following foscarnet administration should blunt the development of ionized hypocalcemia. The effect of parenteral MgSO4 on ionized blood calcium (iCa2+), however, could be unpredictable since magnesium has also been shown to acutely reduce serum calcium through enhanced urinary excretion and by shifting calcium into cells (2, 20). We examined these relationships by intravenously infusing various doses of MgSO4 into patients with AIDS and active CMV disease who were receiving foscarnet. Acute changes in ionized blood magnesium (iMg2+) and iCa2+ were then observed. PTH levels were assayed to determine what effect, if any, MgSO4 has on parathyroid function; other side effects associated with foscarnet administration were tabulated (7; Dohin et al., Int. Conf. AIDS, 1993). Finally, this study addressed the safety of intravenous MgSO4 in this chronically ill population.
MATERIALS AND METHODS
Study subjects.
Patients with AIDS, active CMV infection, and Karnofsky performance status scores of ≥70 were recruited into the study, which was approved by the University of Oklahoma Health Sciences Institutional Review Board. Patient recruitment occurred between March 1995 and January 1997. Patients were excluded if they were receiving potentially nephrotoxic drugs (e.g., aminoglycosides, amphotericin B, or pentamidine), were volume depleted, or were taking magnesium or calcium supplements. A total of 27 patients were screened, and 12 were enrolled in the trial. One patient was enrolled twice, having been removed initially because of a protocol violation. Reasons for nonparticipation among screened patients included refusal (n = 7), a Karnofsky score of <70 (n = 3), inadequate venous access (n = 1), elevated serum creatinine (n = 1), inability to give consent (n = 1), and therapy with amphotericin B (n = 1).
The 12 enrolled patients were all male, with a mean age (± standard deviation) of 35.4 ± 7.6 years and a mean weight of 68.1 ± 11.6 kg. The mean duration of AIDS at time of enrollment was 2.8 ± 1.4 years. Subjects were Caucasian (n = 9), Native American (n = 2), and black (n = 1). Sites of CMV infection included the retina (n = 8), esophagus (n = 2), stomach (n = 1), and adrenal gland (n = 1). Baseline serum magnesium was normal for all patients except one, who had a level of 1.3 mg/dl. Baseline serum calcium, phosphate, and creatinine were normal for all patients. No patient had a prior history of heart block, rhythm abnormality, or prolonged QT interval.
Study design.
This pilot study was a double-blind, placebo-controlled, randomized, crossover, dose-ranging trial that compared the efficacy of three doses of MgSO4 in moderating the fall in iCa2+ and iMg2+ concentrations following foscarnet infusions. Patients were studied during four consecutive morning infusions of foscarnet. MgSO4 in 500 ml of normal saline, at doses of 1, 2, or 3 g, or a placebo (normal saline alone) was administered over 60 min just prior to foscarnet infusion. Hydration prior to evening foscarnet doses consisted of 500 ml of normal saline infused over 60 min. A 24-h washout period between MgSO4 and placebo doses was considered adequate since the half-life of intravenously administered magnesium is short, with nearly all of an administered dose excreted within 4 to 8 h (33). To reduce further the possibility of carryover bias, the sequence of treatments was based on three four-by-four Latin squares. This design was selected so that first-order residual (i.e., carryover) effects could be balanced for the entire study (25). Accordingly, dosing sequences for MgSO4 and placebo differed for each patient (Table 1). Patients failing to complete all assigned doses of MgSO4 or placebo were to be replaced by others in the same sequence group until 12 patients had been recruited.
TABLE 1.
Sequence across study days of MgSO4 and placebo doses for 12 subjects in a crossover design employing three replicated Latin squares
| Subject no. | Dose of MgSO4 (g)
|
|||
|---|---|---|---|---|
| Day 1 | Day 2 | Day 3 | Day 4 | |
| 1 | 1 | 2 | 3 | Placebo |
| 2 | 3 | 1 | Placebo | 2 |
| 3 | Placebo | 3 | 2 | 1 |
| 4 | 2 | Placebo | 1 | 3 |
| 6 | 1 | 3 | 2 | Placebo |
| 7 | Placebo | 2 | 3 | 1 |
| 8 | 3 | Placebo | 1 | 2 |
| 9 | 1 | Placebo | 3 | 2 |
| 10 | 2 | 3 | Placebo | 1 |
| 11 | Placebo | 2 | 1 | 3 |
| 12 | 3 | 1 | 2 | Placebo |
| 13a | 2 | 1 | Placebo | 3 |
Subject no. 5 was removed from the study due to a protocol violation (dose sequence error) and reenrolled after a 2-day washout as subject no. 13 using the same dose sequence; data for subject no. 5 were excluded except those for adverse events.
Randomization codes were supplied by Astra USA, Inc. (Westborough, Mass.), retained by a study pharmacist, and masked from investigators and patients. No codes were broken during the study. Foscarnet was administered for a total of eight doses at 90 mg/kg of body weight over 90 min every 12 h. Prior to infusing study drugs, a 12-h urine collection for determination of creatinine clearance was performed and foscarnet doses were adjusted based on these results (18). All infusions were performed via central venous catheters.
Clinical assessments.
On each day of study drug infusion, vital signs, symptoms, and electrocardiograms were monitored. Blood pressure and heart rate were recorded every 15 min during MgSO4 (or placebo) and foscarnet infusions. A symptom questionnaire was administered prior to, during, and following morning foscarnet doses. Symptoms of nausea, numbness of fingers or lips, twitching or spasms of muscles, anxiety or nervousness, headache, flushing, and fatigue were recorded using a four-point scale (0 = none and 3 = severe). Adverse events spontaneously reported by subjects or observed by investigators were also recorded. Twelve-lead electrocardiograms were performed prior to enrollment and again on each study day prior to and following MgSO4 or placebo doses and following morning foscarnet doses. All enrolled subjects were included in the safety tabulations.
Blood was collected on each study day prior to and immediately following MgSO4 or placebo doses and at 0, 1.5, and 3.5 h following morning foscarnet infusion. Hematocrit, sodium, potassium, iMg2+, iCa2+, and pH were immediately assayed at the bedside using a NOVA 8 analyzer (NOVA Biomedical, Waltham, Mass.). This analyzer utilizes ion-selective calcium and magnesium electrodes to directly determine iCa2+ and iMg2+ concentrations in heparinized whole blood (1). Total serum calcium, magnesium, and phosphate were determined within 2 h using an automated chemistry analyzer (RxL; Dade, Miami, Fla.). Each morning on study days, serum creatinine was also measured with a Dade RxL chemistry analyzer. Quantitative determination of human intact PTH was performed using a radioisotopic assay on serum stored at −20°C (Nichols Institute Diagnostics, San Juan Capistrano, Calif.). Posttreatment serum chemistries and blood counts were determined within 4 days of finishing the study.
Statistical analysis.
Selection of the sample size of 12 was based on variability data from previous studies where the standard deviations for iMg2+ and iCa2+ concentrations at the end of foscarnet infusion were 0.11 and 0.22 mmol/liter, respectively (data on file at Astra USA). Assuming mean reductions of 0.26 and 0.16 mmol/liter for iMg2+ and iCa2+, respectively, following placebo administration and a modest correlation (0.20) between pre- and post-foscarnet infusion values, a sample size of 12 was calculated as providing at least an 80% chance of detecting a difference from placebo if the decline in iMg2+ and/or iCa2+ was abrogated by any of the MgSO4 doses. These considerations were based on a t test for pairwise comparisons with a level of significance of 0.05. Treatments were compared using analysis of covariance for each of the three post-foscarnet infusion times. Magnesium and placebo doses were each considered individual treatments. For laboratory parameters, PROC MIXED (version 6.12; SAS, Cary, N.C.) was used to analyze treatment-related differences from baseline at each post-foscarnet infusion time. The model included pretreatment baseline values as covariates, with baseline data centered prior to inclusion in the model. Treatments were analyzed both by using doses as individual treatments, with all pairwise comparisons tested, and by using regression analysis on the dose levels. Adjustments were not made for multiple comparisons. Results were considered significant at a P value of <0.05.
RESULTS
Magnesium.
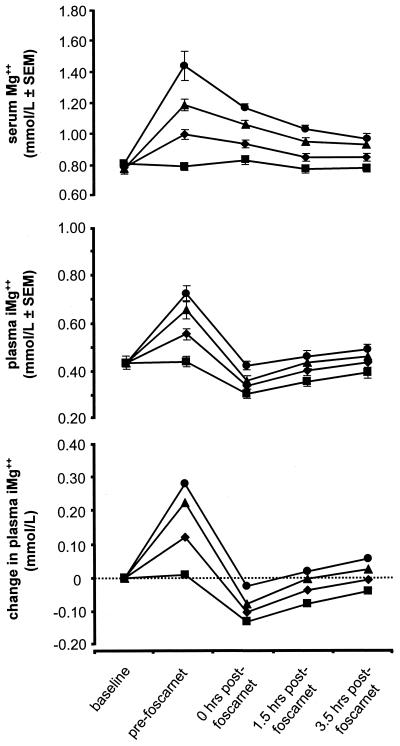
Treatment groups that were administered MgSO4 prior to morning infusion of foscarnet experienced a dose-related increase in mean total serum magnesium and iMg2+ levels (Fig. 1). Decreases in serum magnesium levels occurred following foscarnet infusion in a dose-related manner. Following infusion, serum magnesium levels gradually declined toward baseline over 3.5 h, but levels remained elevated in all three active-dose groups.
FIG. 1.
Changes in total serum Mg2+ and plasma iMg2+ concentrations (mean ± standard error of the mean [SEM]) prior to and following morning foscarnet infusion. All MgSO4 doses showed a significant dose response (P = 0.0001 for linear trend). Changes from baseline for iMg2+ concentration at each post-foscarnet infusion assessment also showed a significant dose response (P = 0.0001). Treatment groups were as follows: ■, placebo; ⧫, 1 g of MgSO4; ▴, 2 g of MgSO4; ●, 3 g of MgSO4.
Levels of iMg2+ dropped abruptly during infusion for all dose groups, so that each dose group achieved a mean value either just below (with the 3-g dose) or more substantially below the baseline by the end of foscarnet infusion. Treatment differences for the changes from baseline in mean iMg2+ concentration among the four groups were highly significant (P < 0.001) and dose related (P < 0.001). All treatment groups had similar average rates of increase in iMg2+ concentration during the 3.5 h after foscarnet infusion. In a dose-dependent manner, each MgSO4 treatment significantly (P < 0.001) reduced the magnitude of foscarnet-induced ionized hypomagnesemia at each post-foscarnet infusion assessment. By the final assessment, all active dose groups had recovered or nearly recovered to the baseline level.
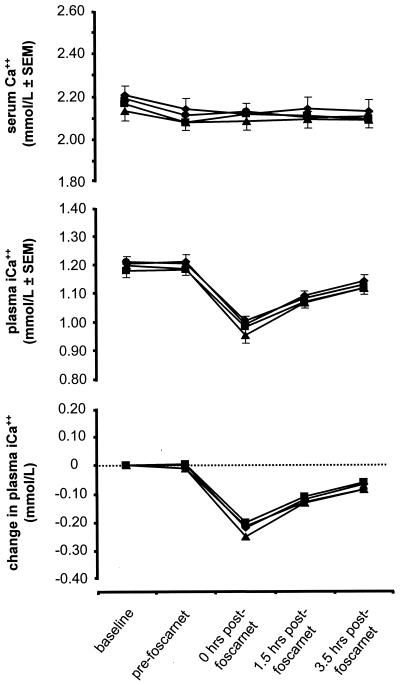
Calcium.
Mean total serum calcium values showed a slight drop after infusion of MgSO4 or placebo (Fig. 2). This decrease was maintained throughout the period of observation and likely reflected the hydration given before foscarnet infusion. With respect to iCa2+, all treatment groups experienced little change due to pre-foscarnet infusion hydration, followed by a substantial decrease after foscarnet infusion. At the completion of the morning foscarnet infusions, low iCa2+ values gradually recovered but remained similar for all groups and were still below baseline after 3.5 h. No significant differences were observed among treatment or placebo groups at any assessment time.
FIG. 2.
Changes in total serum Ca2+ and plasma iCa2+ concentrations (mean ± standard error of the mean [SEM]) prior to and following morning foscarnet infusion. No dose-response trend was noted for the post-foscarnet infusion assessments (P > 0.05 for all tests). Treatment groups are indicated as in Fig. 1.
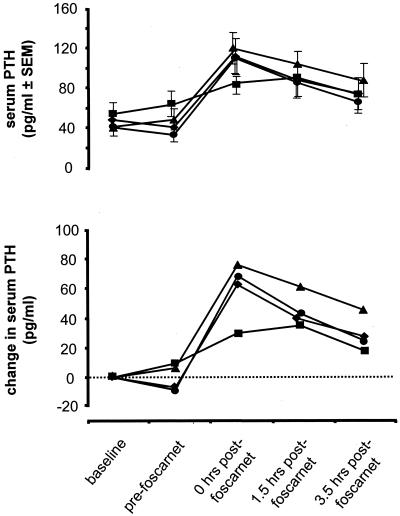
PTH.
MgSO4 supplementation had no discernible effect on mean PTH levels prior to foscarnet infusion (Fig. 3). Compared to placebo, MgSO4 administration increased mean PTH levels post-foscarnet infusion for all treatment groups (P < 0.02). At the immediate post-foscarnet infusion time, the placebo group mean was lower than the mean for each of the other dose groups, but the 2-g group showed greater increases in PTH than either the 1- or 3-g group, suggesting a lack of a true dose-response effect. At 1.5 and 3.5 h post-foscarnet infusion, the overall test for treatment differences and linear trends did not show significance (P > 0.05). The comparisons between the 2-g and placebo groups at these times, however, continued to show significance (P = 0.05 and 0.02, respectively). These data indicate that MgSO4 facilitated release of preformed PTH from the parathyroid gland.
FIG. 3.
Changes in serum PTH levels (mean ± standard error of the mean [SEM]) prior to and following morning foscarnet infusion. Treatment groups are indicated as in Fig. 1.
Other laboratory values.
MgSO4 doses had no observable effects on plasma sodium level, hematocrit, or pH (data not shown). A trend toward a lower plasma phosphate level was found at each of the three measurement intervals following foscarnet infusion. Similarly, a trend toward a lower plasma potassium level was found, but only at the 1.5- and 3.5-h postinfusion times. Changes from baseline were not statistically significantly different among the MgSO4 dose groups at any postinfusion assessment time for either phosphate or potassium (data not shown).
Other clinical parameters.
Symptoms associated with hypocalcemia and/or hypomagnesemia were assessed prior to, during, and immediately following morning foscarnet infusion. Low incidences of nausea, numbness of the fingers or lips, twitching or spasm, anxiety or nervousness, headache, flushing, or fatigue were observed for patients receiving any MgSO4 dose. Overall, no relationships could be ascertained between MgSO4 doses and the incidence of any symptom. A trend was noted for more nervous system symptoms (hyperesthesias, paresthesias, and leg cramps) during and following foscarnet infusion at higher doses of MgSO4 compared to baseline, placebo, or treatment with 1 g of MgSO4 (0% versus 8 to 17%). Adverse events reported by three or more individuals at one or more visits included nausea (7 of 12 [58%]), headache (5 of 12 [42%]), and tachycardia (3 of 12 [25%]).
Electrocardiograms detected no clinically significant dysrhythmias at any MgSO4 dose. Compared to baseline, QTc intervals were slightly prolonged for only two subjects following foscarnet infusion (32 and 22 ms). No significant trends were noted for blood pressure, pulse, temperature, respiration, or body weight (data not shown).
Two serious adverse events occurred during the study. One subject developed acute renal insufficiency which resolved after 2 weeks. During the protocol, the subject had developed a central line infection that led to nausea, volume depletion, and fever, all of which likely helped precipitate foscarnet-induced renal insufficiency. Another subject died from disseminated CMV infection 6 weeks following completion of the study. This death, however, was considered unrelated to foscarnet or MgSO4 infusion. No subject was discontinued from treatment due to an adverse event.
DISCUSSION
Magnesium is the fourth most abundant cation in the body and an essential cofactor in numerous cellular reactions (14, 38). Magnesium exists in blood in an ionized form (55%), complexed to anions such as phosphate, bicarbonate, and citrate (15%), and bound to proteins (30%). Unlike calcium, magnesium homeostasis is regulated not by hormones but instead through the direct actions of nephrons in the kidney (29, 38). Hypomagnesemia is a common side effect of selected drugs such as foscarnet, but other causes include poor oral intake, gastrointestinal losses, kidney disease, and redistribution following trauma, burns, and cardiopulmonary bypass (14, 38). Short- and long-term clinical manifestations of magnesium depletion include muscular spasms, seizures, vertigo, ataxia, weakness, depression, electrocardiogram changes (e.g., widened QRS complex or prolonged PR interval), osteoporosis, osteomalacia, and atherosclerosis (38).
As a therapeutic agent, magnesium is used as an anticonvulsant and potent vasodilator. It has been used for prophylaxis and therapy in a variety of cardiovascular disorders, for treatment of preeclampsia and eclampsia, and to treat stroke (4, 14). Extensive clinical use has confirmed the safety of rapid administration of large doses of MgSO4. Typically, only small decreases in blood pressure are seen (14, 37), although hypotension, decreased respiration, vomiting, neuromuscular blockade, and coma have been reported (14, 30, 37).
This double-blind, placebo-controlled, randomized, crossover study sought to establish the efficacy of parenterally administered MgSO4, included with the hydration before foscarnet infusion, in preventing foscarnet-induced ionized hypocalcemia and ionized hypomagnesemia and symptoms. The study population consisted of 12 patients with AIDS, fair-to-good functional status, and active CMV disease. Overall, doses of MgSO4 reduced or eliminated foscarnet-induced acute ionized hypomagnesemia. Supplementation, however, had no discernible effect on foscarnet-induced ionized hypocalcemia despite a greater increase in serum PTH levels. The lack of effect of higher PTH levels on the fall in iCa2+ is unexplained. Perhaps there are direct or indirect effects of foscarnet, human immunodeficiency virus, or CMV on bone osteoclasts that are yet to be identified, or possibly higher PTH levels were not sustained long enough to mobilize skeletal calcium. Alternatively, a direct hypocalcemic action of magnesium infusion may have offset the influence of higher PTH levels (2, 20).
MgSO4 at doses up to 3 g were generally well tolerated. No associations were detected among any MgSO4 dose and the incidence of any recorded symptoms. Significant blood pressure, pulse, or electrocardiogram changes were not noted. The lack of significant adverse events following MgSO4 infusions is consistent with other reports for patients having received equally large or larger doses of parenteral MgSO4 (4, 14). Although there was a trend toward more nervous system disorders associated with the groups receiving the higher doses of MgSO4, these signs and symptoms were associated with infusions of foscarnet, not MgSO4. These signs and symptoms are normally associated with hypocalcemia and not with hypermagnesemia or administration of MgSO4. Since parenteral MgSO4 did not alter the hypocalcemia or symptoms associated with foscarnet infusion, routine supplementation in patients with normal serum magnesium levels cannot be recommended during treatment with foscarnet.
Foscarnet is indicated for the treatment of immunocompromised patients with CMV or acyclovir-resistant mucocutaneous herpes simplex virus disease (19, 27, 28, 32). Nephrotoxicity and electrolyte abnormalities are the most common adverse side effects associated with its use. In this study only a single case of reversible nephrotoxicity occurred among the 12 subjects (8%). This percentage is comparable to the 15 to 20% incidence of nephrotoxicity per year reported in a recent large controlled trial (35). Electrolyte abnormalities following foscarnet infusion include acute ionized hypocalcemia and hypomagnesemia, hypokalemia, and hypo- or hyperphosphatemia. While these changes are often self-limited, serious adverse sequelae can occur, including seizures, arrhythmias, paresthesias, and changes in the sensorium (13, 17, 18, 24, 34; Youle et al., Letter). In this study foscarnet-associated symptoms were infrequent, and MgSO4 appeared to be of no benefit in preventing them.
Several limitations should be considered while interpreting these data. First, this study was short-term, lasting only for 4 days. Long-term effects of parenteral MgSO4 administration on blood calcium, magnesium, or PTH levels cannot be inferred from these data. Second, the failure to detect any effect of MgSO4 on the fall in iCa2+ concentrations following foscarnet infusion was perhaps due to inadequate dosing. Other studies have used substantially higher MgSO4 doses to achieve >3-fold increases in blood magnesium concentrations compared to baseline values (14, 16, 37). The maximal dose of MgSO4 used in this study, however, did not quite double the mean serum magnesium concentrations (Fig. 1). Any concern for inadequate dosing should be considered in light of the fact that the 3-g MgSO4 dose returned post-foscarnet infusion iMg2+ levels to baseline values, and maximal rises in PTH occurred with the 2-g but not 3-g doses of MgSO4.
Latin square designs for crossover studies, while attractive because of their inherent efficiency, have potential limitations (25). This method permits studies using relatively few subjects. A parallel-group design for three doses of MgSO4 and placebo most likely would have required at least 48 subjects. Recruitment of this number of patients with AIDS and active CMV infection would have required screening at least 100 subjects in a multicenter trial. Instead, the Latin square crossover design allowed a pilot study to be completed at one center in a short time frame. This crossover design assumes the dose sequence for MgSO4 and placebo does not in and of itself produce significant effects. This assumption was considered valid because washout intervals between MgSO4 doses were expected to be short in patients with normal renal function, due to rapid clearance of intravenous magnesium. The return of magnesium, calcium, and PTH levels to baseline each morning before subsequent MgSO4 or placebo doses supported this notion.
Subjects' medical conditions while on treatment are a potential confounder in this analysis. The influence of medical condition on outcome measures could present a serious problem in interpretation if this were a parallel study in which levels of underlying disease were not uniformly distributed across treatment groups. Using the Latin square design over a short (4-day) period, the underlying medical condition of each subject is likely to have been essentially the same during exposure to each treatment. Indeed, only one subject who developed renal insufficiency on the protocol had a significant change in medical condition during the course of the study.
The infusion of MgSO4 modulated the hypomagnesemic effect of foscarnet in a dose-dependent manner. MgSO4 also increased mean PTH levels, apparently restoring the sensitivity of parathyroid cells to the ionized hypocalcemic stimulus, but not in a dose-related fashion. Despite these findings, supplementation with MgSO4 at these doses had no discernible effect on acute ionized hypocalcemia induced by foscarnet. No dose-related, clinically significant adverse events were found, suggesting that intravenous supplementation of magnesium sulfate at doses up to 3 g over 1 h is safe in this chronically ill population. However, given the lack of benefit of parenteral MgSO4 in reducing ionized hypocalcemia or symptoms associated with foscarnet infusion, routine supplementation in individuals who have normal serum magnesium levels cannot be recommended. Given the difficulty of administering parenteral calcium, further studies using combinations of oral calcium, magnesium, and vitamin D are needed to identify regimens that might minimize ionized hypomagnesia and hypocalcemia following foscarnet infusion.
ACKNOWLEDGMENTS
This project was funded in part by Astra USA, Inc.
We thank Anne Hitchcock, Ann Campbell, Cheryl Eaton, Linda Thompson, Wanda Fako, Edna Patatanian, John Ondrasik, Janet Price, and Timothy Tytle for their technical assistance.
REFERENCES
- 1.Altura B T, Shirey T L, Young C C, Hiti J, Dell'Orfano K, Handwerker S M, Altura B M. A new method for the rapid determination of ionized Mg2+ in whole blood, serum and plasma. Methods Find Exp Clin Pharmacol. 1992;14:297–304. [PubMed] [Google Scholar]
- 2.Anast C S, Winnacker J L, Forte L R, Burns T W. Impaired release of parathyroid hormone in magnesium deficiency. J Clin Endocrinol Metab. 1976;42:707–717. doi: 10.1210/jcem-42-4-707. [DOI] [PubMed] [Google Scholar]
- 3.Aweeka F, Gambertoglio J, Mills J, Jacobson M A. Pharmacokinetics of intermittently administered intravenous foscarnet in the treatment of acquired immunodeficiency syndrome patients with serious cytomegalovirus retinitis. Antimicrob Agents Chemother. 1989;33:742–745. doi: 10.1128/aac.33.5.742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Belfort M A, Anthony J, Saade G R. Prevention of eclampsia. Semin Perinatol. 1999;23:65–78. doi: 10.1016/s0146-0005(99)80061-5. [DOI] [PubMed] [Google Scholar]
- 5.Bourdeau A, Dohin E, Kindermans C, Souberbielle J C, Katlama C, Sachs C. Functional hypoparathyroidism induced by foscarnet in AIDS patients treated for CMV related disease is connected with low ionized magnesium: a breakthrough of its electrometric determination. Bone. 1995;16:210S. [Google Scholar]
- 6.Cacoub P, Deray G, Baumelou A, Le Hoang P, Rozenbaum W, Gentilini M, Soubrie C, Rousselie F, Jacobs C. Acute renal failure induced by foscarnet: 4 cases. Clin Nephrol. 1988;29:315–318. [PubMed] [Google Scholar]
- 7.Cholst I N, Steinberg S F, Tropper P J, Fox H E, Segre G V, Bilezikian J P. The influence of hypermagnesemia on serum calcium and parathyroid hormone levels in human subjects. N Engl J Med. 1984;310:1221–1225. doi: 10.1056/NEJM198405103101904. [DOI] [PubMed] [Google Scholar]
- 8.Crumpacker C S. Mechanism of action of foscarnet against viral polymerases. Am J Med. 1992;92(Suppl. 2A):3S–7S. doi: 10.1016/0002-9343(92)90329-a. [DOI] [PubMed] [Google Scholar]
- 9.Deray G, Martinez F, Katlama C, Levaltier B, Beaufils H, Danis M, Rozenheim M, Baumelou A, Dohin E, Gentilini M, Jacobs C. Foscarnet nephrotoxicity: mechanism, incidence and prevention. Am J Nephrol. 1989;9:316–321. doi: 10.1159/000167987. [DOI] [PubMed] [Google Scholar]
- 10.Erlich K S, Jacobson M A, Koehler J E, Follansbee S E, Drennan D P, Gooze L, Safrin S, Mills J. Foscarnet therapy for severe acyclovir-resistant herpes simplex virus type-2 infections in patients with the acquired immunodeficiency syndrome (AIDS): an uncontrolled trial. Ann Intern Med. 1989;110:710–713. doi: 10.7326/0003-4819-110-9-710. [DOI] [PubMed] [Google Scholar]
- 11.Estep H, Shaw W A, Watlington C, Hobe R, Holland W, Tucker S G. Hypocalcemia due to hypomagnesemia and reversible parathyroid hormone unresponsiveness. J Clin Endocrinol Metab. 1969;29:842–848. doi: 10.1210/jcem-29-6-842. [DOI] [PubMed] [Google Scholar]
- 12.Fuss M, Cogan E, Gillet C, Karmali R, Geurts J, Bergans A, Brauman H, Bouillon R, Corvilain J. Magnesium administration reverses the hypocalcaemia secondary to hypomagnesaemia despite low circulating levels of 25-hydroxy vitamin D and 1,25-dihydroxy vitamin D. Clin Endocrinol. 1985;22:807–815. doi: 10.1111/j.1365-2265.1985.tb00171.x. [DOI] [PubMed] [Google Scholar]
- 13.Gearhart M O, Sorg T B. Foscarnet-induced severe hypomagnesemia and other electrolyte disorders. Ann Pharmacother. 1993;27:285–289. doi: 10.1177/106002809302700304. [DOI] [PubMed] [Google Scholar]
- 14.Gomez M N. Magnesium and cardiovascular disease. Anesthesiology. 1998;89:222–240. doi: 10.1097/00000542-199807000-00029. [DOI] [PubMed] [Google Scholar]
- 15.Guillaume M P, Karmali R, Bergmann P, Cogan E. Unusual prolonged hypocalcemia due to foscarnet in a patient with AIDS. Clin Infect Dis. 1997;25:932–933. doi: 10.1086/597645. [DOI] [PubMed] [Google Scholar]
- 16.Idama T O, Lindow S W. Magnesium sulphate: a review of clinical pharmacology applied to obstetrics. Br J Obstet Gynaecol. 1998;105:260–268. doi: 10.1111/j.1471-0528.1998.tb10084.x. [DOI] [PubMed] [Google Scholar]
- 17.Jacobson M. Review of the toxicities of foscarnet. J Acquir Immune Defic Syndr. 1992;5:S11–S17. [PubMed] [Google Scholar]
- 18.Jacobson M A, Gambertoglio J G, Aweeka F T, Causey D M, Portale A A. Foscarnet-induced hypocalcemia and effects of foscarnet on calcium metabolism. J Clin Endocrinol Metab. 1991;72:1130–1135. doi: 10.1210/jcem-72-5-1130. [DOI] [PubMed] [Google Scholar]
- 19.Jacobson M A, O'Donnell J J, Mills J. Foscarnet treatment of cytomegalovirus retinitis in patients with the acquired immunodeficiency syndrome. Antimicrob Agents Chemother. 1989;33:736–741. doi: 10.1128/aac.33.5.736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jones K H, Fourman P. Effects of infusions of magnesium and of calcium in parathyroid insufficiency. Clin Sci. 1966;30:139–146. [PubMed] [Google Scholar]
- 21.Katlama C, Dohin E, Caumes E, Cochereau-Massin I, Brancon C, Robinet M, Rogeaux O, Dahan R, Gentilini M. Foscarnet induction therapy for cytomegalovirus retinitis in AIDS: comparison of twice-daily and three-times-daily regimens. J Acquir Immune Defic Syndr. 1992;5(Suppl. 1):S18–S24. [PubMed] [Google Scholar]
- 22.Levi J, Massry S G, Coburn J W, Llach F, Kleeman C R. Hypocalcemia in magnesium-depleted dogs: evidence for reduced responsiveness to parathyroid hormone and relative failure of parathyroid gland function. Metabolism. 1974;23:323–335. doi: 10.1016/0026-0495(74)90050-x. [DOI] [PubMed] [Google Scholar]
- 23.Loghman-Adham M, Dousa T P. Dual action of phosphonoformic acid on Na+-phosphate cotransport in opossum kidney cells. Am J Physiol. 1992;263:F301–F310. doi: 10.1152/ajprenal.1992.263.2.F301. [DOI] [PubMed] [Google Scholar]
- 24.Lor E, Liu Y Q. Neurologic sequelae associated with foscarnet therapy. Ann Pharmacother. 1994;28:1035–1037. doi: 10.1177/106002809402800908. [DOI] [PubMed] [Google Scholar]
- 25.Neter J, Wasserman W, Kutner M H. Applied linear statistical models. Boston, Mass: Irwin; 1990. pp. 1083–1117. [Google Scholar]
- 26.Noormohamed F H, Youle M S, Tang B, Martin-Munley S, Gazzard B G, Lant A F, Higgs C J, Piner C. Plasma and renal effects of foscarnet in HIV seropositive patients. Antivir Ther. 1996;1:172–179. [PubMed] [Google Scholar]
- 27.Palestine A G, Polis M A, De Smet M D, Baird B F, Falloon J, Kovacs J A, Davey R T, Zurlo J J, Zunich K M, Davis M, Hubbard L, Brothers R, Ferris F L, Chew E, Davis J L, Rubin B I, Mellow S D, Metcalf J A, Manischewitz J, Minor J R, Nussenblatt R B, Masur H, Lane C H. A randomized, controlled trial of foscarnet in the treatment of cytomegalovirus retinitis in patients with AIDS. Ann Intern Med. 1991;115:665–673. doi: 10.7326/0003-4819-115-9-665. [DOI] [PubMed] [Google Scholar]
- 28.Polis M A. Foscarnet and ganciclovir in the treatment of cytomegalovirus retinitis. J Acquir Immune Defic Syndr. 1992;5(Suppl. 1):S3–S10. [PubMed] [Google Scholar]
- 29.Quamme G A. Renal magnesium handling: new insights in understanding old problems. Kidney Int. 1997;52:1180–1195. doi: 10.1038/ki.1997.443. [DOI] [PubMed] [Google Scholar]
- 30.Rizzo M A, Fisher M, Lock J P. Hypermagnesemic pseudocoma. Arch Intern Med. 1993;153:1130–1132. [PubMed] [Google Scholar]
- 31.Ryrfeldt A, Nordgren T, Lundstrom J. Hypocalcemia induced by foscarnet (foscavir) infusion in dogs. Fundam Appl Toxicol. 1992;18:126–130. doi: 10.1016/0272-0590(92)90204-u. [DOI] [PubMed] [Google Scholar]
- 32.Safrin S, Crumpacker C, Chatis P, Daves R, Hafenr R, Rush J, Kessler H, Landry B, Mills J. A controlled trial comparing foscarnet with vidarabine for acyclovir-resistant mucocutaneous herpes simplex in the acquired immunodeficiency syndrome. N Engl J Med. 1991;325:551–555. doi: 10.1056/NEJM199108223250805. [DOI] [PubMed] [Google Scholar]
- 33.Smith P K, Winkler A W, Hopp H E. The pharmacological actions of parenterally administered magnesium salts: a review. Anesthesiology. 1942;3:323–330. [Google Scholar]
- 34.Studies of Ocular Complications of AIDS Research Group in Collaboration with the AIDS Clinical Trials Group. Morbidity and toxic effects associated with ganciclovir or foscarnet therapy in a randomized cytomegalovirus retinitis trial. Arch Intern Med. 1995;155:65–74. [PubMed] [Google Scholar]
- 35.Studies of Ocular Complications of AIDS Research Group in Collaboration with the AIDS Clinical Trials Group. Combination foscarnet and ganciclovir therapy vs monotherapy for the treatment of relapsed cytomegalovirus retinitis in patients with AIDS. The cytomegalovirus retreatment trial. Arch Ophthalmol. 1996;114:23–33. doi: 10.1001/archopht.1996.01100130021004. [DOI] [PubMed] [Google Scholar]
- 36.Stünzi H, Perrin D D. Stability constants of metal complexes of phosphonoacetic acid. J Inorg Chem. 1979;10:309–316. [Google Scholar]
- 37.Sydow M, Crozier T A, Zielmann S, Radke J, Burchardi H. High-dose intravenous magnesium sulfate in the management of life-threatening status asthmaticus. Intensive Care Med. 1993;19:467–471. doi: 10.1007/BF01711089. [DOI] [PubMed] [Google Scholar]
- 38.Weisinger J R, Bellorín-Font E. Magnesium and phosphorus. Lancet. 1998;352:391–396. doi: 10.1016/S0140-6736(97)10535-9. [DOI] [PubMed] [Google Scholar]