Abstract
The bactericidal activities of monotherapy with trovafloxacin (−0.37 ± 0.15 Δlog10 CFU/ml · h), vancomycin (−0.32 ± 0.12 Δlog10 CFU/ml · h), and ceftriaxone (−0.36 ± 0.19 Δlog10 CFU/ml · h) for the treatment of experimental meningitis in rabbits due to a clinical penicillin-resistant pneumococcal strain (MIC, 4 mg/liter) were similar. The combination of ceftriaxone with trovafloxacin considerably improved the killing rates (−0.67 ± 0.16 Δlog10 CFU/ml · h) and was slightly superior to ceftriaxone with vancomycin (killing rate, −0.53 ± 0.22 Δlog10 CFU/ml · h), the regimen most commonly used in clinical practice. In vitro, synergy was demonstrated between ceftriaxone and trovafloxacin by the checkerboard method (fractional inhibitory concentration index, 0.5) and by time-killing assays over 8 h.
The treatment of pneumococcal infections has been jeopardized lately by the worldwide increase in the spread of resistant pneumococci (2). Among recently developed compounds, new quinolones are promising candidates, especially due to their activity against many gram-positive microorganisms, including penicillin-resistant pneumococci. Trovafloxacin showed dose-dependent bactericidal activity against penicillin-resistant pneumococci in an experimental model of meningitis in rabbits (10). However, at doses (15 mg/kg of body weight) that lead to levels achievable in humans, its activity was only moderate (14). These data raise the possibility that trovafloxacin in combination with other antibiotics might play a role in the enhancement of bactericidal activity. We have recently shown that the addition of vancomycin to the trovafloxacin treatment regimen significantly improved the rate of killing of penicillin-resistant pneumococci by trovafloxacin in an experimental model of meningitis and in vitro (14). Little is known about possible interactions between β-lactam antibiotics, e.g., ceftriaxone, and quinolones against penicillin-resistant pneumococci in an experimental model of meningitis. The aim of this study was to test the activity of trovafloxacin (15 mg/kg) in combination with ceftriaxone against penicillin-resistant pneumococci. Ceftriaxone, trovafloxacin, and vancomycin monotherapies and ceftriaxone in combination with vancomycin served as comparison regimens.
Rabbit meningitis model.
The meningitis model, originally described by Dacey and Sande (3), was slightly modified. Briefly, young New Zealand White rabbits that weighed between 2 and 2.5 kg were anesthetized by intramuscular injections of ketamine (30 mg/kg) and xylazine (15 mg/kg) and were immobilized in stereotactic frames for induction of meningitis and sampling of cerebrospinal fluid (CSF). An inoculum that contained approximately 105 CFU of penicillin-resistant pneumococci serotype 6 was directly injected into the cisterna magna. The pneumococcal strain had originally been isolated from a patient with pneumonia at the University Hospital of Bern, Bern, Switzerland. The MICs were as follows: penicillin, 4 mg/liter; ceftriaxone, 0.5 mg/liter; vancomycin, 0.12 to 0.25 mg/liter; and trovafloxacin, 0.12 mg/liter.
A long-acting anesthetic (ethylcarbamate [urethane]; 3.5 g/rabbit) was injected subcutaneously, and the animals were returned to their cages. Fourteen hours later the cisterna magna was punctured again for periodic CSF sampling before and 1, 2, 4, 6, and 8 h after initiation of therapy. The following antibiotics were administered through a peripheral ear vein as bolus injections at the indicated concentrations: alatrofloxacin (prodrug of trovafloxacin), 15 mg/kg; ceftriaxone, 125 mg/kg; vancomycin, 20 mg/kg. Ceftriaxone and alatrofloxacin were injected once at hour 0 and vancomycin was injected at hours 0 and 4, as described by Friedland et al. (6) and Rodoni et al. (14). Untreated controls received saline.
Bacterial titers were measured by 10-fold serial dilution of CSF samples, which were then plated on blood agar plates containing 5% sheep blood, and the plates were incubated overnight at 37°C. In parallel, 20 μl of undiluted CSF sample was plated (limit of detectability, 50 CFU/ml). Comparison of the titers between different dilutions of CSF was used to exclude significant carryover effects during therapy. The antimicrobial activities of the regimens during the 8-h treatment were calculated by linear regression analysis and were expressed as a decrease in the log10 number of CFU per milliliter per hour (Δlog10 CFU/ml · h). A value of 1.7 (the log10 value of the limit of detectability) was assigned to the first sterile CSF sample, and a value of 0 was assigned to any following sterile sample (10, 12). The results were expressed as means ± standard deviations. Statistical significance was determined by the Newman-Keuls test.
Measurement of antibiotic levels in CSF.
Antibiotic concentrations in CSF were determined by the agar diffusion method. Standard curve studies were performed in saline with 5% rabbit serum in order to mimic the protein concentration in CSF during meningitis (10, 12). Escherichia coli (ATCC 25922) was used as the test strain (16) for ceftriaxone, and Bacillus subtilis (ATCC 6633) was used as the test strain for vancomycin and trovafloxacin (15). The intra- and interday variabilities of this method were each less than 10%. The limit of detection was 0.5 mg/liter for vancomycin, 1 mg/liter for ceftriaxone, and 0.25 mg/liter for trovafloxacin.
In vitro assays.
The pneumococcal strain was grown in C+Y medium (11) to an optical density at 590 nm of 0.3 and was then diluted 40-fold to 106 CFU/ml, which corresponded to the bacterial titer in the CSF of the rabbits before initiation of therapy. Antibiotics were added at concentrations that corresponded to 1× the MIC: 0.12 mg/liter for trovafloxacin and 0.5 mg/liter for ceftriaxone. Combinations of ceftriaxone (0.5 mg/liter) with trovafloxacin (0.12 mg/liter) were also tested. Bacterial titers were determined at hours 0, 2, 4, 6, and 8 by serial dilution of samples, which had been plated on agar plates containing 5% sheep blood and incubated at 37°C for 24 h. Experiments were performed in triplicate, and results were expressed as means ± standard deviations. The definition of synergy was a bactericidal effect of a drug combination that significantly exceeded the sum of the bactericidal effects of each agent alone (4, 7).
Determination of FIC index.
The same isolate used in the time-killing experiments and in the animal model was grown in C+Y medium until the logarithmic growth phase and was then diluted. Approximately 0.5 × 106 to 1 × 106 CFU was pipetted into microtiter trays that contained concentrations of each antibiotic which ranged from 1/32 the MIC to 2× the MIC. Microtiter trays were incubated at 37°C for 24 h. After 6, 12, and 24 h the plates were read for detection of inhibition of bacterial growth. The experiments were performed in triplicate and were repeated twice. Fractional inhibitory concentration (FIC) indices were calculated by the method of Eliopoulos and Moellering (4). Synergy was defined as an FIC index of ≤0.5, indifference was defined as an FIC index of >0.5 to ≤4, and antagonism was defined as an FIC index of >4.
Due to its excellent activity against penicillin-resistant isolates trovafloxacin is a valuable alternative for the treatment of pneumococcal infections, especially when resistant strains are suspected. Trovafloxacin was successfully used to treat pneumonia due to penicillin-resistant pneumococci (L. Mandell, D. W. Hopkins, and S. Hopkins. Abstr. 37th Intersci. Conf. Antimicrob. Agents Chemother., abstr. LM-71, p. 377, 1997). Whereas trovafloxacin and ceftriaxone were similarly successful for the treatment of meningococcal meningitis in children (S. Hopkins, D. Williams, M. Dunne, L. Marinovch, M. Edieline, E. Utt, Abstr. 36th Intersci. Conf. Antimicrob. Agents Chemother., abstr. 15, p. 19, 1996), little is known about the efficacy of trovafloxacin for the treatment of pneumococcal meningitis due to penicillin-resistant strains. In the rabbit meningitis model, trovafloxacin showed dose-dependent bactericidal activity against resistant pneumococci. However, at doses that lead to levels comparable to those achieved in humans, trovafloxacin monotherapy was moderately efficient. In contrast, the combination of vancomycin with trovafloxacin was highly effective (14).
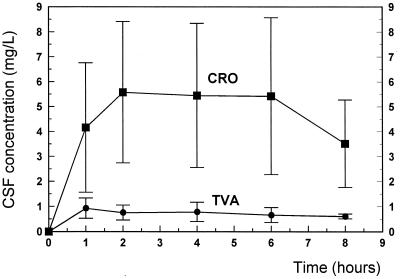
The doses of ceftriaxone (125 mg/kg) and vancomycin (two doses of 20 mg/kg each) used in this study were identical to those used in previous studies of experimental meningitis (6, 7, 8, 14) and produced levels achieved in the CSF of humans (1, 5, 9). In our study, the ceftriaxone levels achieved in the CSF (3.5 to 5.5 mg/liter) (Fig. 1) corresponded to the concentrations described in previous studies (6, 7, 8). Trovafloxacin reached levels in the CSF of rabbits (0.9 to 0.6 mg/liter) comparable to those measured in infants with noninflamed meninges after one injection of trovafloxacin (5 mg/kg) (A. Arguedas-Mohs, S. L. Vargas, J. S. Bradley, C. Loaiza, R. Rivera, J. Vincent, R. Teng, and J. N. Walterspiel. Abstr. 37th Intersci. Conf. Antimicrob. Agents Chemother. abstr. A-105, p. 21, 1997). The levels in CSF remained far above the MICs of both antibiotics during the entire therapy period. The ratio of the concentration in CSF to the MIC ranged between 11 and 7 for ceftriaxone and between 7.5 and 5 for trovafloxacin.
FIG. 1.
Ceftriaxone (CRO) and trovafloxacin (TVA) concentrations in CSF during the 8 h after intravenous injection of 125 mg of ceftriaxone per kg and 15 mg of trovafloxacin per kg. The concentrations of the two substances remained above the MICs during the entire treatment period (MICs, 0.5 and 0.12 mg/liter for ceftriaxone and trovafloxacin, respectively).
In our model, the monotherapies produced similar killing rates, although vancomycin and trovafloxacin seemed to be slightly more efficacious (Table 1). After 8 h, the combination therapies were significantly more effective than the monotherapies (P < 0.05). The combination of trovafloxacin with ceftriaxone was highly bactericidal and was even superior to the standard regimen (ceftriaxone in combination with vancomycin; P < 0.05). Synergistic activity between trovafloxacin and vancomycin has recently been described in the same experimental setting, in which they produced killing rates (−0.60 ± 0.25 Δlog10 CFU/ml · h) comparable to those achieved in the present study (14).
TABLE 1.
Single-drug and combination therapy for experimental meningitis caused by penicillin-resistant Streptococcus pneumoniae
| Antibiotic | No. of rabbits | Initial titer (log10 CFU/ml [mean + SD]) | Killing rate (Δlog10 CFU/ml · h [mean ± SD]) | Killing rate/8 h (log10 CFU/ml [mean ± SD]) |
|---|---|---|---|---|
| Controls | 10 | 6.30 ± 0.64 | +0.04 ± 0.10a | +0.28 ± 0.15a |
| Alatrofloxacin | 9 | 6.36 ± 0.79 | −0.37 ± 0.15 | −3.04 ± 0.19 |
| Ceftriaxone | 9 | 5.64 ± 0.73 | −0.32 ± 0.12 | −2.25 ± 0.50 |
| Vancomycin | 12 | 6.30 ± 1.12 | −0.36 ± 0.19 | −3.25 ± 0.45 |
| Vancomycin + ceftriaxone | 9 | 6.15 ± 0.20 | −0.53 ± 0.22 | −4.00 ± 0.86bc |
| Alatrofloxacin + ceftriaxone | 10 | 6.01 ± 0.74 | −0.67 ± 0.16b | −5.05 ± 0.60bc |
P < 0.05 versus all groups.
P < 0.05 versus all monotherapies.
P < 0.05 for alatrofloxacin + ceftriaxone versus vancomycin + ceftriaxone.
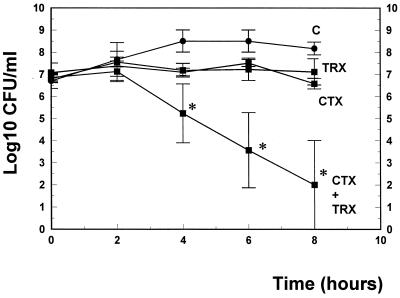
These experimental data confirm the results observed in vitro. The same combinations were tested in time-killing experiments over 8 h. We selected antibiotic concentrations which led to only marginal killing rates with monotherapies. In this experimental setting in liquid cultures over 8 h, trovafloxacin and ceftriaxone acted synergistically (Fig. 2; killing rates over 8 h, 5 versus 0.1 and 0.5 log10 CFU/ml for the combination versus ceftriaxone and trovafloxacin monotherapy, respectively). Synergy was also confirmed by a FIC index of 0.5, as determined by the checkerboard technique. Similar results have recently been described in vitro by Nicolau et al. (13) for 12 of 16 penicillin-resistant pneumococcal isolates.
FIG. 2.
Killing rates of trovafloxacin (TRX), ceftriaxone (CTX), and ceftriaxone in combination with trovafloxacin (CTX+TRX) using concentrations equal to the MIC. Experiments were performed in triplicate, and killing rates were expressed as means ± standard deviation. ∗, P < 0.05.
The underlying mechanism of synergistic activity between a cell wall-active antibiotic (ceftriaxone) and an antibiotic that interferes with DNA synthesis is not clear but is reminiscent of the synergy between β-lactam antibiotics and aminoglycosides described for enterococci. The efficacies of therapies with trovafloxacin in combination with either ceftriaxone or vancomycin (14) suggest a role for quinolones in combination with cell wall-active antibiotics, especially when highly bactericidal activity is required, e.g., for the treatment of meningitis due to penicillin-resistant pneumococci.
REFERENCES
- 1.Ahmed A. A critical evaluation of vancomycin for treatment of bacterial meningitis. Pediatr Infect Dis J. 1997;16:895–903. doi: 10.1097/00006454-199709000-00014. [DOI] [PubMed] [Google Scholar]
- 2.Bradley J, Scheld W M. The challenge of penicillin-resistant Streptococcus pneumoniae meningitis: current antibiotic therapy in the 1990s. Clin Infect Dis. 1997;24(Suppl.):213–221. doi: 10.1093/clinids/24.supplement_2.s213. [DOI] [PubMed] [Google Scholar]
- 3.Dacey R G, Sande M A. Effect of probenecid on cerebrospinal fluid concentrations of penicillin and cephalosporin derivatives. Antimicrob Agents Chemother. 1974;6:437–441. doi: 10.1128/aac.6.4.437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Eliopoulos G M, Moellering R C. Antimicrobials combinations. In: Lorian V, editor. Antibiotics in laboratory medicine. 4th ed. Baltimore, Md: The Williams & Wilkins Co.; 1991. pp. 330–396. [Google Scholar]
- 5.Friedland I R, McCracken G H., Jr Management of infections caused by antibiotic-resistant Streptococcus pneumoniae. N Engl J Med. 1994;331:377–382. doi: 10.1056/NEJM199408113310607. [DOI] [PubMed] [Google Scholar]
- 6.Friedland I R, Paris M, Ehret S, Hickey S, Olsen K, McCracken G H., Jr Evaluation of antimicrobial regimens for treatment of experimental penicillin- and cephalosporin-resistant pneumococcal meningitis. Antimicrob Agents Chemother. 1993;37:1630–1636. doi: 10.1128/aac.37.8.1630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gerber C M, Cottagnoud M, Neftel K A, Täuber M G, Cottagnoud P. Evaluation of cefepime alone and in combination with vancomycin against penicillin-resistant pneumococci in the rabbit meningitis model and in vitro. J Antimicrob Chemother. 2000;45:63–68. doi: 10.1093/jac/45.1.63. [DOI] [PubMed] [Google Scholar]
- 8.Gerber C M, Cottagnoud M, Neftel K A, Täuber M G, Cottagnoud P. Meropenem alone and in combination with vancomycin in experimental meningitis caused by a penicillin-resistant pneumococcal strain. Eur J Clin Microbiol Infect Dis. 1999;18:866–870. doi: 10.1007/s100960050421. [DOI] [PubMed] [Google Scholar]
- 9.Hawley H B, Gump D W. Vancomycin therapy in bacterial meningitis. Am J Dis Child. 1973;126:261–264. doi: 10.1001/archpedi.1973.02110190231025. [DOI] [PubMed] [Google Scholar]
- 10.Kim Y S, Liu Q, Chow L, Täuber M G. Trovafloxacin in treatment of rabbits with experimental meningitis caused by high-level penicillin pneumococci. Antimicrob Agents Chemother. 1997;41:1186–1189. doi: 10.1128/aac.41.5.1186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lack S, Hotchkiss R D. A study of the genetic material determining an enzyme activity in pneumococcus. Biochem Biophys Acta. 1960;39:508–518. doi: 10.1016/0006-3002(60)90205-5. [DOI] [PubMed] [Google Scholar]
- 12.Nau R, Kaye K, Sachdeva M, Sande E, Täuber M G. Rifampin for the therapy of experimental pneumococcal meningitis in rabbits. Antimicrob Agents Chemother. 1995;38:1186–1189. doi: 10.1128/aac.38.5.1186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nicolau D P, Tessier P R, Quintiliani R, Nightingale C H. Synergistic activity of trovafloxacin with ceftriaxone or vancomycin against S. pneumoniae with various penicillin susceptibilities. Antimicrob Agents Chemother. 1998;42:991–992. doi: 10.1128/aac.42.4.991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rodoni D, Hänni F, Gerber C M, Cottagnoud M, Neftel K, Täuber M G, Cottagnoud P. Trovafloxacin in combination with vancomycin against penicillin-resistant pneumococci in the rabbit meningitis model. Antimicrob Agents Chemother. 1999;43:963–965. doi: 10.1128/aac.43.4.963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Simon H J, Yin E Y. Microbioassay of antimicrobial agents. Appl Microbiol. 1970;19:573–579. doi: 10.1128/am.19.4.573-579.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Täuber M G, Hackbarth C J, Scott K G, Rusnak M G, Sande M A. New cephalosporins cefotaxime, cefpimizole, BMY 28142, and HT 810 in experimental pneumococcal meningitis in rabbits. Antimicrob Agents Chemother. 1985;27:340–342. doi: 10.1128/aac.27.3.340. [DOI] [PMC free article] [PubMed] [Google Scholar]